User login
D-Mannose as UTI Treatment Offers No Benefit
TOPLINE:
A natural sugar used to treat recurring urinary tract infections (rUTIs) did not reduce future episodes, outpatient visits, the use of antibiotics, or symptoms compared with a placebo, according to a new study.
METHODOLOGY:
- D-Mannose is recommended as a natural alternative to antibiotics and sold as a dietary supplement; research showing the efficacy of D-mannose in treating UTIs is mixed.
- The double-blind, randomized controlled trial followed 598 women older than 18 years (median age, 61.3; range 18.2-93.5 years) with a history of rUTIs over 6 months from nearly 100 primary care clinics in the United Kingdom.
- Patients took 2 g of D-mannose or placebo powder daily and recorded their symptoms using a daily diary or through responses to health surveys, weekly questionnaires, and phone calls.
- Researchers checked medical records for urine culture results, antibiotic prescriptions, hospitalizations, and outpatient visits for UTIs.
TAKEAWAY:
- Approximately 51% of participants who took D-mannose and 55.7% of those who took a placebo contacted a healthcare professional reporting a UTI (relative risk, 0.92; 95% CI, 0.80-1.05; P = .22).
- Women in both groups reported similar durations of “moderately bad” or “worse” symptoms, and the number of antibiotic courses, instances of clinically suspected UTIs, and hospital admissions were similar between the two groups.
- Some studies have reported that synthetic mannosides are promising alternatives to D-mannose.
IN PRACTICE:
“D-Mannose should not be recommended to prevent future episodes of medically attended UTI in women with recurrent UTI in primary care,” the study authors wrote.
SOURCE:
The study was led by Gail Hayward, DPhil, associate professor at the Nuffield Department of Primary Care Health Sciences at the University of Oxford in England, and was published online in JAMA Internal Medicine.
LIMITATIONS:
Some participants may have taken less than 2 mg/d or skipped days. Because researchers used powder rather than capsules, dosing could have been inconsistent. Researchers did not obtain a microbiologic confirmation for each rUTI. A small percentage of women were taking antibiotics.
DISCLOSURES:
The study was funded by the National Institute for Health and Care Research (NIHR) School for Primary Care Research and the NIHR Oxford Biomedical Research Centre. Various authors reported receiving support from the NIHR Health Protection Research Unit on Healthcare-Associated Infections and Antimicrobial Resistance and were NIHR investigators.
A version of this article first appeared on Medscape.com.
TOPLINE:
A natural sugar used to treat recurring urinary tract infections (rUTIs) did not reduce future episodes, outpatient visits, the use of antibiotics, or symptoms compared with a placebo, according to a new study.
METHODOLOGY:
- D-Mannose is recommended as a natural alternative to antibiotics and sold as a dietary supplement; research showing the efficacy of D-mannose in treating UTIs is mixed.
- The double-blind, randomized controlled trial followed 598 women older than 18 years (median age, 61.3; range 18.2-93.5 years) with a history of rUTIs over 6 months from nearly 100 primary care clinics in the United Kingdom.
- Patients took 2 g of D-mannose or placebo powder daily and recorded their symptoms using a daily diary or through responses to health surveys, weekly questionnaires, and phone calls.
- Researchers checked medical records for urine culture results, antibiotic prescriptions, hospitalizations, and outpatient visits for UTIs.
TAKEAWAY:
- Approximately 51% of participants who took D-mannose and 55.7% of those who took a placebo contacted a healthcare professional reporting a UTI (relative risk, 0.92; 95% CI, 0.80-1.05; P = .22).
- Women in both groups reported similar durations of “moderately bad” or “worse” symptoms, and the number of antibiotic courses, instances of clinically suspected UTIs, and hospital admissions were similar between the two groups.
- Some studies have reported that synthetic mannosides are promising alternatives to D-mannose.
IN PRACTICE:
“D-Mannose should not be recommended to prevent future episodes of medically attended UTI in women with recurrent UTI in primary care,” the study authors wrote.
SOURCE:
The study was led by Gail Hayward, DPhil, associate professor at the Nuffield Department of Primary Care Health Sciences at the University of Oxford in England, and was published online in JAMA Internal Medicine.
LIMITATIONS:
Some participants may have taken less than 2 mg/d or skipped days. Because researchers used powder rather than capsules, dosing could have been inconsistent. Researchers did not obtain a microbiologic confirmation for each rUTI. A small percentage of women were taking antibiotics.
DISCLOSURES:
The study was funded by the National Institute for Health and Care Research (NIHR) School for Primary Care Research and the NIHR Oxford Biomedical Research Centre. Various authors reported receiving support from the NIHR Health Protection Research Unit on Healthcare-Associated Infections and Antimicrobial Resistance and were NIHR investigators.
A version of this article first appeared on Medscape.com.
TOPLINE:
A natural sugar used to treat recurring urinary tract infections (rUTIs) did not reduce future episodes, outpatient visits, the use of antibiotics, or symptoms compared with a placebo, according to a new study.
METHODOLOGY:
- D-Mannose is recommended as a natural alternative to antibiotics and sold as a dietary supplement; research showing the efficacy of D-mannose in treating UTIs is mixed.
- The double-blind, randomized controlled trial followed 598 women older than 18 years (median age, 61.3; range 18.2-93.5 years) with a history of rUTIs over 6 months from nearly 100 primary care clinics in the United Kingdom.
- Patients took 2 g of D-mannose or placebo powder daily and recorded their symptoms using a daily diary or through responses to health surveys, weekly questionnaires, and phone calls.
- Researchers checked medical records for urine culture results, antibiotic prescriptions, hospitalizations, and outpatient visits for UTIs.
TAKEAWAY:
- Approximately 51% of participants who took D-mannose and 55.7% of those who took a placebo contacted a healthcare professional reporting a UTI (relative risk, 0.92; 95% CI, 0.80-1.05; P = .22).
- Women in both groups reported similar durations of “moderately bad” or “worse” symptoms, and the number of antibiotic courses, instances of clinically suspected UTIs, and hospital admissions were similar between the two groups.
- Some studies have reported that synthetic mannosides are promising alternatives to D-mannose.
IN PRACTICE:
“D-Mannose should not be recommended to prevent future episodes of medically attended UTI in women with recurrent UTI in primary care,” the study authors wrote.
SOURCE:
The study was led by Gail Hayward, DPhil, associate professor at the Nuffield Department of Primary Care Health Sciences at the University of Oxford in England, and was published online in JAMA Internal Medicine.
LIMITATIONS:
Some participants may have taken less than 2 mg/d or skipped days. Because researchers used powder rather than capsules, dosing could have been inconsistent. Researchers did not obtain a microbiologic confirmation for each rUTI. A small percentage of women were taking antibiotics.
DISCLOSURES:
The study was funded by the National Institute for Health and Care Research (NIHR) School for Primary Care Research and the NIHR Oxford Biomedical Research Centre. Various authors reported receiving support from the NIHR Health Protection Research Unit on Healthcare-Associated Infections and Antimicrobial Resistance and were NIHR investigators.
A version of this article first appeared on Medscape.com.
Worldwide Uptick in Invasive Group A Streptococcus Disease Post Pandemic — What Should We Know?
Invasive group A streptococcus (iGAS) infections are rare (4-9 cases/100,000 US population annually) but potentially devastating (approximately 2,300 deaths annually in US), and affect all ages. Cases increase in winter-spring, paralleling the “season” of increased noninvasive GAS, e.g., pharyngitis and scarlet fever. iGAS case rates are lower in children than adults. That said, one well-known pediatric iGAS scenario has been deep cellulitis and necrotizing fasciitis during the healing phase of varicella. Other forms of iGAS include bacteremia, pneumonia (particularly when empyema is present), lymphangitis, erysipelas, and toxic shock syndrome. iGAS can occur with/after influenza but has also occurred concurrently with other viral respiratory infections.
Persons with underlying conditions (cancer or immune compromised status; chronic diseases of the heart, kidney or lung; diabetes mellitus) are at higher risk. Other subpopulations at risk for iGAS are illicit drug users, the elderly, homeless persons, nursing home residents, American Indian persons, and Alaska Native persons. Most experts feel that highly toxigenic strains of GAS are responsible for most iGAS. Indeed, most iGAS isolates produce (sometimes hyper-produce) superantigens that cause exaggerated innate immune responses, higher levels of inflammation, and often times tissue destruction, e.g., “flesh eating bacteria.” And who can forget that Jim Henson, creator of the Muppets, died of iGAS?
But why discuss iGAS in 2024? The pattern for iGAS has fluctuated more than usual in the last decade. So much so that the recent upsurge has caught the collective eye of the lay press. So, patients and friends may have questions about why and how iGAS is increasing lately. The bottom line is that no one knows for sure. However, the most recent 2 years of uptick may reflect GAS circulating at relatively high levels even when taking into account that GAS season occurs in winter-spring most years. Yet it seems likely that additional factors may have played a role in the fluctuations noted this past decade, e.g., temporary changes in societal behavior, a new GAS strain with over two dozen mutations, and possibly rapid waning of protection against GAS exotoxins.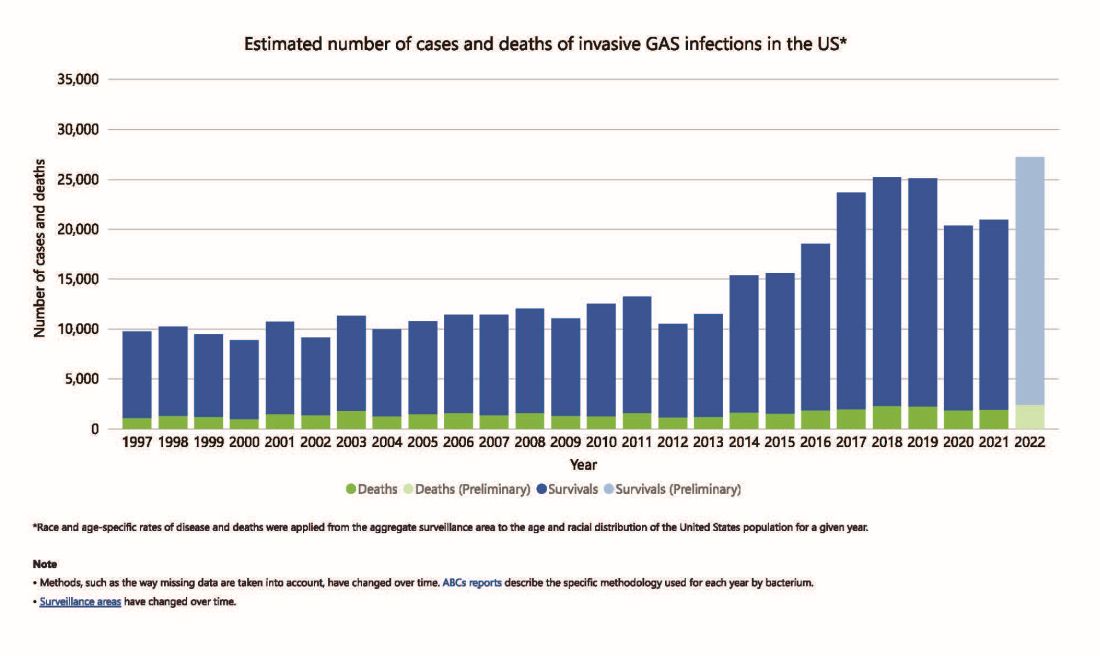
Social Behavior Factor
The SARS-CoV-2 pandemic brought extremes of disease and death to the world and dramatic changes in social behavior. A byproduct was dramatic decreases in nearly all infectious diseases, with numerous reports of near absence of many respiratory and gastrointestinal viruses in the 2020-2021 seasons. Interestingly, we did not see a drop in human rhinovirus infections, justifying its nickname as the cockroach of viruses. Reports also emerged about drops in bacterial diseases during 2020-2021 (although not so much for STIs), including noninvasive and invasive GAS disease, and also GAS-associated deaths (lowest since 2016).1 The drop in iGAS during social restrictions makes sense because GAS is spread by direct contact with infected persons or their secretions, and social contact had dramatically decreased particularly in the first 6 months of the pandemic.
However, since 2022 and the return to “normal” social behaviors, both viral diseases (e.g., RSV, influenza, and Norovirus), and some bacterial diseases have rebounded. That said, something else must be contributing, because iGAS rates had increased 4-5 years pre pandemic. In fact, the fluctuating pattern included “normal” annual rates in the early 2000s rising in ~2015 followed by the explainable pandemic drop (by nearly 25%), and not-too-unexpected 2-year postpandemic rise. But interestingly enough, the rebound is higher than might be expected for iGAS and children were overrepresented in first year’s rise (2022 rate for pediatric iGAS was the highest since 1997) while those older than 65 were overrepresented in second year (2023).1
Emergence of M1UK
One potential factor for the prepandemic rise in iGAS infections worldwide is the emergence and worldwide spread of a new GAS emm type variant designated M1UK.2 GAS isolates can be typed into categories designated as emm types based on DNA sequence. There are more than 240 emm types, with 6 being most common — M1, M3, and M28 (each up to 20% of GAS isolates) and M12, M82, and M89 (each up to 10%). M1, M3 and M28 have also been particularly associated with invasive disease. While emm types vary year to year and region by region, the overall emm type distribution among iGAS isolates in the United States had not been unusual since the turn of the century and the US M1 strain was the same as that which had been predominant worldwide (designated M1GLOBAL). This new M1UK sublineage had emerged around 2010 and had been increasing pre pandemic. The M1UK sequence contained a specific set of 27 SNPs (single nucleoside polymorphisms, i.e., single base mutations) and was associated with an uptick in scarlet fever in the United Kingdom starting around 2010. Its prevalence increased up to around 2015 while spreading internationally. It also had enhanced expression of SpeA, a phage-encoded superantigen. Some of the M1UK mutations also appear to alter GAS metabolic processes to allow better survival (better “fitness”) compared with other GAS. So, a more virulent hardier GAS had arisen and seems a reasonable candidate for contributing to the increased iGAS rates.
Waning Antibody to GAS As Potential Factor in Rebound
No consensus exists on correlates of protection from iGAS. However, adults seem to have less noninvasive GAS than children. One potential reason is that frequent GAS re-exposure, regardless of whether disease results, likely boosts anti-GAS antibodies. Pandemic social restrictions temporarily prevented such boosts. In children with developing antibody repertoires, anti-GAS antibodies may have waned below protective levels faster during a year without frequent boosting. Thus, children were iGAS susceptible soon after pandemic restrictions were dropped (2022). Increased iGAS rates in the elderly in 2023 may have occurred because of diminished GAS exposures accelerating immune senescence with anti-GAS antibodies dropping, but less quickly than in children. These speculations are simply hypotheses until future studies can test them.
All that said, how do we use information on increased iGAS in our daily practices? In addition to standard preventive strategies for viral coinfections (e.g., varicella and influenza vaccine), reminding families about rigorous attention to wound care is the one high-risk scenario we have not yet discussed. During 2024, a time of expected increased prevalence of iGAS, early wound care needs to be fastidious. Further, share warning signs with families (e.g., rapidly expanding painful erythema), “streaks” ascending from extremity wounds, fever and a highly painful wound, darkening almost purple color within cellulitis or soft tissue infection, or loss of sensation in the middle of an otherwise painful soft tissue infection. These presentations require immediate medical attention.
If such a patient presents, the Centers for Disease Control and Prevention (CDC) recommends admission along with blood and, where possible, wound cultures. If in the context of pneumonia with pleural effusion, culturing pleural fluid is also important. Remember, leading edge cultures are not often positive for GAS, seemingly because GAS exotoxins are found at erythema’s leading edge, not the bacteria. The bacteria are somewhere more central in the inflammatory process. Despite not being prominent among recent iGAS cases, another scenario that could sneak up on you is the infected surgical wound as nascent iGAS.
Finally, remember that nationally increasing numbers of iGAS isolates are resistant to erythromycin and clindamycin, the latter usually recommended to reduce tissue damage in iGAS.3 So, it is important to be aware of susceptibility patterns in your locale and consider an ID consultation. My hope is that you do not see an iGAS case this year, but we all need to remain alert. With a high index of suspicion and rapid diagnosis, you can minimize long-term sequelae and potential fatalities.
While it is too early to tell how the rest of 2024 will turn out, preliminary indications are that GAS is circulating at higher than usual levels (30%-35% GAS positive throat swabs in early April 2024 in Kansas City area) and iGAS rates will likely also be relatively high, particularly if Ontario, Canada, data are any indication.4 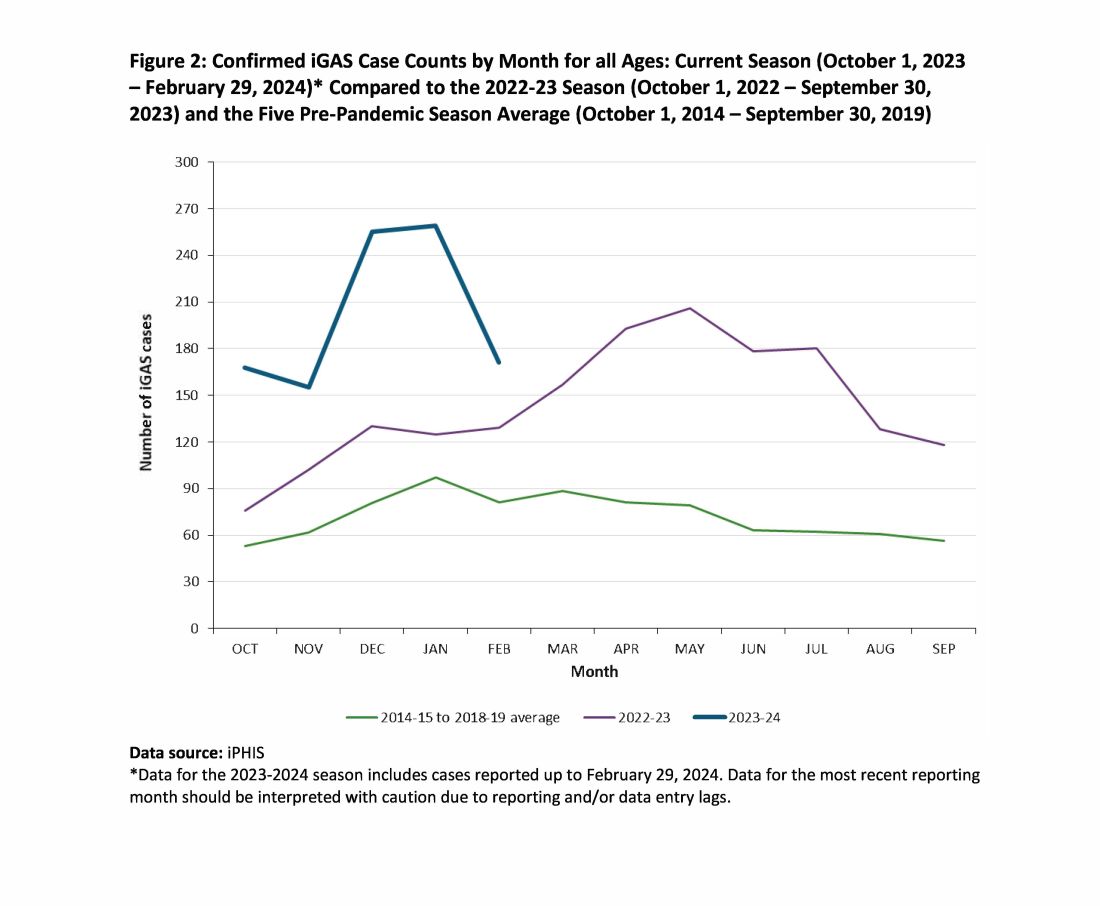
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Mo. He said he had no relevant financial disclosures. Email him at [email protected].
References
1. Current Group A Strep Activity, Centers for Disease Control and Prevention. April 2024. CDC webpage on current invasive GAS disease. April 2024.
2. Li Y et al. Expansion of Invasive Group A Streptococcus M1UK Lineage in Active Bacterial Core Surveillance, United States, 2019-2021 Emerg Infect Dis. 2023;29(10):2116-2120. doi: 10.3201/eid2910.230675.
3. Andreoni F et al. Clindamycin Affects Group A Streptococcus Virulence Factors and Improves Clinical Outcome. J Infect Dis. 2017 Jan 15;215(2):269-277. doi: 10.1093/infdis/jiw229.
4. Group A Streptococcal Disease, Invasive (iGAS), Public Health Ontario.
Invasive group A streptococcus (iGAS) infections are rare (4-9 cases/100,000 US population annually) but potentially devastating (approximately 2,300 deaths annually in US), and affect all ages. Cases increase in winter-spring, paralleling the “season” of increased noninvasive GAS, e.g., pharyngitis and scarlet fever. iGAS case rates are lower in children than adults. That said, one well-known pediatric iGAS scenario has been deep cellulitis and necrotizing fasciitis during the healing phase of varicella. Other forms of iGAS include bacteremia, pneumonia (particularly when empyema is present), lymphangitis, erysipelas, and toxic shock syndrome. iGAS can occur with/after influenza but has also occurred concurrently with other viral respiratory infections.
Persons with underlying conditions (cancer or immune compromised status; chronic diseases of the heart, kidney or lung; diabetes mellitus) are at higher risk. Other subpopulations at risk for iGAS are illicit drug users, the elderly, homeless persons, nursing home residents, American Indian persons, and Alaska Native persons. Most experts feel that highly toxigenic strains of GAS are responsible for most iGAS. Indeed, most iGAS isolates produce (sometimes hyper-produce) superantigens that cause exaggerated innate immune responses, higher levels of inflammation, and often times tissue destruction, e.g., “flesh eating bacteria.” And who can forget that Jim Henson, creator of the Muppets, died of iGAS?
But why discuss iGAS in 2024? The pattern for iGAS has fluctuated more than usual in the last decade. So much so that the recent upsurge has caught the collective eye of the lay press. So, patients and friends may have questions about why and how iGAS is increasing lately. The bottom line is that no one knows for sure. However, the most recent 2 years of uptick may reflect GAS circulating at relatively high levels even when taking into account that GAS season occurs in winter-spring most years. Yet it seems likely that additional factors may have played a role in the fluctuations noted this past decade, e.g., temporary changes in societal behavior, a new GAS strain with over two dozen mutations, and possibly rapid waning of protection against GAS exotoxins.
Social Behavior Factor
The SARS-CoV-2 pandemic brought extremes of disease and death to the world and dramatic changes in social behavior. A byproduct was dramatic decreases in nearly all infectious diseases, with numerous reports of near absence of many respiratory and gastrointestinal viruses in the 2020-2021 seasons. Interestingly, we did not see a drop in human rhinovirus infections, justifying its nickname as the cockroach of viruses. Reports also emerged about drops in bacterial diseases during 2020-2021 (although not so much for STIs), including noninvasive and invasive GAS disease, and also GAS-associated deaths (lowest since 2016).1 The drop in iGAS during social restrictions makes sense because GAS is spread by direct contact with infected persons or their secretions, and social contact had dramatically decreased particularly in the first 6 months of the pandemic.
However, since 2022 and the return to “normal” social behaviors, both viral diseases (e.g., RSV, influenza, and Norovirus), and some bacterial diseases have rebounded. That said, something else must be contributing, because iGAS rates had increased 4-5 years pre pandemic. In fact, the fluctuating pattern included “normal” annual rates in the early 2000s rising in ~2015 followed by the explainable pandemic drop (by nearly 25%), and not-too-unexpected 2-year postpandemic rise. But interestingly enough, the rebound is higher than might be expected for iGAS and children were overrepresented in first year’s rise (2022 rate for pediatric iGAS was the highest since 1997) while those older than 65 were overrepresented in second year (2023).1
Emergence of M1UK
One potential factor for the prepandemic rise in iGAS infections worldwide is the emergence and worldwide spread of a new GAS emm type variant designated M1UK.2 GAS isolates can be typed into categories designated as emm types based on DNA sequence. There are more than 240 emm types, with 6 being most common — M1, M3, and M28 (each up to 20% of GAS isolates) and M12, M82, and M89 (each up to 10%). M1, M3 and M28 have also been particularly associated with invasive disease. While emm types vary year to year and region by region, the overall emm type distribution among iGAS isolates in the United States had not been unusual since the turn of the century and the US M1 strain was the same as that which had been predominant worldwide (designated M1GLOBAL). This new M1UK sublineage had emerged around 2010 and had been increasing pre pandemic. The M1UK sequence contained a specific set of 27 SNPs (single nucleoside polymorphisms, i.e., single base mutations) and was associated with an uptick in scarlet fever in the United Kingdom starting around 2010. Its prevalence increased up to around 2015 while spreading internationally. It also had enhanced expression of SpeA, a phage-encoded superantigen. Some of the M1UK mutations also appear to alter GAS metabolic processes to allow better survival (better “fitness”) compared with other GAS. So, a more virulent hardier GAS had arisen and seems a reasonable candidate for contributing to the increased iGAS rates.
Waning Antibody to GAS As Potential Factor in Rebound
No consensus exists on correlates of protection from iGAS. However, adults seem to have less noninvasive GAS than children. One potential reason is that frequent GAS re-exposure, regardless of whether disease results, likely boosts anti-GAS antibodies. Pandemic social restrictions temporarily prevented such boosts. In children with developing antibody repertoires, anti-GAS antibodies may have waned below protective levels faster during a year without frequent boosting. Thus, children were iGAS susceptible soon after pandemic restrictions were dropped (2022). Increased iGAS rates in the elderly in 2023 may have occurred because of diminished GAS exposures accelerating immune senescence with anti-GAS antibodies dropping, but less quickly than in children. These speculations are simply hypotheses until future studies can test them.
All that said, how do we use information on increased iGAS in our daily practices? In addition to standard preventive strategies for viral coinfections (e.g., varicella and influenza vaccine), reminding families about rigorous attention to wound care is the one high-risk scenario we have not yet discussed. During 2024, a time of expected increased prevalence of iGAS, early wound care needs to be fastidious. Further, share warning signs with families (e.g., rapidly expanding painful erythema), “streaks” ascending from extremity wounds, fever and a highly painful wound, darkening almost purple color within cellulitis or soft tissue infection, or loss of sensation in the middle of an otherwise painful soft tissue infection. These presentations require immediate medical attention.
If such a patient presents, the Centers for Disease Control and Prevention (CDC) recommends admission along with blood and, where possible, wound cultures. If in the context of pneumonia with pleural effusion, culturing pleural fluid is also important. Remember, leading edge cultures are not often positive for GAS, seemingly because GAS exotoxins are found at erythema’s leading edge, not the bacteria. The bacteria are somewhere more central in the inflammatory process. Despite not being prominent among recent iGAS cases, another scenario that could sneak up on you is the infected surgical wound as nascent iGAS.
Finally, remember that nationally increasing numbers of iGAS isolates are resistant to erythromycin and clindamycin, the latter usually recommended to reduce tissue damage in iGAS.3 So, it is important to be aware of susceptibility patterns in your locale and consider an ID consultation. My hope is that you do not see an iGAS case this year, but we all need to remain alert. With a high index of suspicion and rapid diagnosis, you can minimize long-term sequelae and potential fatalities.
While it is too early to tell how the rest of 2024 will turn out, preliminary indications are that GAS is circulating at higher than usual levels (30%-35% GAS positive throat swabs in early April 2024 in Kansas City area) and iGAS rates will likely also be relatively high, particularly if Ontario, Canada, data are any indication.4 
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Mo. He said he had no relevant financial disclosures. Email him at [email protected].
References
1. Current Group A Strep Activity, Centers for Disease Control and Prevention. April 2024. CDC webpage on current invasive GAS disease. April 2024.
2. Li Y et al. Expansion of Invasive Group A Streptococcus M1UK Lineage in Active Bacterial Core Surveillance, United States, 2019-2021 Emerg Infect Dis. 2023;29(10):2116-2120. doi: 10.3201/eid2910.230675.
3. Andreoni F et al. Clindamycin Affects Group A Streptococcus Virulence Factors and Improves Clinical Outcome. J Infect Dis. 2017 Jan 15;215(2):269-277. doi: 10.1093/infdis/jiw229.
4. Group A Streptococcal Disease, Invasive (iGAS), Public Health Ontario.
Invasive group A streptococcus (iGAS) infections are rare (4-9 cases/100,000 US population annually) but potentially devastating (approximately 2,300 deaths annually in US), and affect all ages. Cases increase in winter-spring, paralleling the “season” of increased noninvasive GAS, e.g., pharyngitis and scarlet fever. iGAS case rates are lower in children than adults. That said, one well-known pediatric iGAS scenario has been deep cellulitis and necrotizing fasciitis during the healing phase of varicella. Other forms of iGAS include bacteremia, pneumonia (particularly when empyema is present), lymphangitis, erysipelas, and toxic shock syndrome. iGAS can occur with/after influenza but has also occurred concurrently with other viral respiratory infections.
Persons with underlying conditions (cancer or immune compromised status; chronic diseases of the heart, kidney or lung; diabetes mellitus) are at higher risk. Other subpopulations at risk for iGAS are illicit drug users, the elderly, homeless persons, nursing home residents, American Indian persons, and Alaska Native persons. Most experts feel that highly toxigenic strains of GAS are responsible for most iGAS. Indeed, most iGAS isolates produce (sometimes hyper-produce) superantigens that cause exaggerated innate immune responses, higher levels of inflammation, and often times tissue destruction, e.g., “flesh eating bacteria.” And who can forget that Jim Henson, creator of the Muppets, died of iGAS?
But why discuss iGAS in 2024? The pattern for iGAS has fluctuated more than usual in the last decade. So much so that the recent upsurge has caught the collective eye of the lay press. So, patients and friends may have questions about why and how iGAS is increasing lately. The bottom line is that no one knows for sure. However, the most recent 2 years of uptick may reflect GAS circulating at relatively high levels even when taking into account that GAS season occurs in winter-spring most years. Yet it seems likely that additional factors may have played a role in the fluctuations noted this past decade, e.g., temporary changes in societal behavior, a new GAS strain with over two dozen mutations, and possibly rapid waning of protection against GAS exotoxins.
Social Behavior Factor
The SARS-CoV-2 pandemic brought extremes of disease and death to the world and dramatic changes in social behavior. A byproduct was dramatic decreases in nearly all infectious diseases, with numerous reports of near absence of many respiratory and gastrointestinal viruses in the 2020-2021 seasons. Interestingly, we did not see a drop in human rhinovirus infections, justifying its nickname as the cockroach of viruses. Reports also emerged about drops in bacterial diseases during 2020-2021 (although not so much for STIs), including noninvasive and invasive GAS disease, and also GAS-associated deaths (lowest since 2016).1 The drop in iGAS during social restrictions makes sense because GAS is spread by direct contact with infected persons or their secretions, and social contact had dramatically decreased particularly in the first 6 months of the pandemic.
However, since 2022 and the return to “normal” social behaviors, both viral diseases (e.g., RSV, influenza, and Norovirus), and some bacterial diseases have rebounded. That said, something else must be contributing, because iGAS rates had increased 4-5 years pre pandemic. In fact, the fluctuating pattern included “normal” annual rates in the early 2000s rising in ~2015 followed by the explainable pandemic drop (by nearly 25%), and not-too-unexpected 2-year postpandemic rise. But interestingly enough, the rebound is higher than might be expected for iGAS and children were overrepresented in first year’s rise (2022 rate for pediatric iGAS was the highest since 1997) while those older than 65 were overrepresented in second year (2023).1
Emergence of M1UK
One potential factor for the prepandemic rise in iGAS infections worldwide is the emergence and worldwide spread of a new GAS emm type variant designated M1UK.2 GAS isolates can be typed into categories designated as emm types based on DNA sequence. There are more than 240 emm types, with 6 being most common — M1, M3, and M28 (each up to 20% of GAS isolates) and M12, M82, and M89 (each up to 10%). M1, M3 and M28 have also been particularly associated with invasive disease. While emm types vary year to year and region by region, the overall emm type distribution among iGAS isolates in the United States had not been unusual since the turn of the century and the US M1 strain was the same as that which had been predominant worldwide (designated M1GLOBAL). This new M1UK sublineage had emerged around 2010 and had been increasing pre pandemic. The M1UK sequence contained a specific set of 27 SNPs (single nucleoside polymorphisms, i.e., single base mutations) and was associated with an uptick in scarlet fever in the United Kingdom starting around 2010. Its prevalence increased up to around 2015 while spreading internationally. It also had enhanced expression of SpeA, a phage-encoded superantigen. Some of the M1UK mutations also appear to alter GAS metabolic processes to allow better survival (better “fitness”) compared with other GAS. So, a more virulent hardier GAS had arisen and seems a reasonable candidate for contributing to the increased iGAS rates.
Waning Antibody to GAS As Potential Factor in Rebound
No consensus exists on correlates of protection from iGAS. However, adults seem to have less noninvasive GAS than children. One potential reason is that frequent GAS re-exposure, regardless of whether disease results, likely boosts anti-GAS antibodies. Pandemic social restrictions temporarily prevented such boosts. In children with developing antibody repertoires, anti-GAS antibodies may have waned below protective levels faster during a year without frequent boosting. Thus, children were iGAS susceptible soon after pandemic restrictions were dropped (2022). Increased iGAS rates in the elderly in 2023 may have occurred because of diminished GAS exposures accelerating immune senescence with anti-GAS antibodies dropping, but less quickly than in children. These speculations are simply hypotheses until future studies can test them.
All that said, how do we use information on increased iGAS in our daily practices? In addition to standard preventive strategies for viral coinfections (e.g., varicella and influenza vaccine), reminding families about rigorous attention to wound care is the one high-risk scenario we have not yet discussed. During 2024, a time of expected increased prevalence of iGAS, early wound care needs to be fastidious. Further, share warning signs with families (e.g., rapidly expanding painful erythema), “streaks” ascending from extremity wounds, fever and a highly painful wound, darkening almost purple color within cellulitis or soft tissue infection, or loss of sensation in the middle of an otherwise painful soft tissue infection. These presentations require immediate medical attention.
If such a patient presents, the Centers for Disease Control and Prevention (CDC) recommends admission along with blood and, where possible, wound cultures. If in the context of pneumonia with pleural effusion, culturing pleural fluid is also important. Remember, leading edge cultures are not often positive for GAS, seemingly because GAS exotoxins are found at erythema’s leading edge, not the bacteria. The bacteria are somewhere more central in the inflammatory process. Despite not being prominent among recent iGAS cases, another scenario that could sneak up on you is the infected surgical wound as nascent iGAS.
Finally, remember that nationally increasing numbers of iGAS isolates are resistant to erythromycin and clindamycin, the latter usually recommended to reduce tissue damage in iGAS.3 So, it is important to be aware of susceptibility patterns in your locale and consider an ID consultation. My hope is that you do not see an iGAS case this year, but we all need to remain alert. With a high index of suspicion and rapid diagnosis, you can minimize long-term sequelae and potential fatalities.
While it is too early to tell how the rest of 2024 will turn out, preliminary indications are that GAS is circulating at higher than usual levels (30%-35% GAS positive throat swabs in early April 2024 in Kansas City area) and iGAS rates will likely also be relatively high, particularly if Ontario, Canada, data are any indication.4 
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Mo. He said he had no relevant financial disclosures. Email him at [email protected].
References
1. Current Group A Strep Activity, Centers for Disease Control and Prevention. April 2024. CDC webpage on current invasive GAS disease. April 2024.
2. Li Y et al. Expansion of Invasive Group A Streptococcus M1UK Lineage in Active Bacterial Core Surveillance, United States, 2019-2021 Emerg Infect Dis. 2023;29(10):2116-2120. doi: 10.3201/eid2910.230675.
3. Andreoni F et al. Clindamycin Affects Group A Streptococcus Virulence Factors and Improves Clinical Outcome. J Infect Dis. 2017 Jan 15;215(2):269-277. doi: 10.1093/infdis/jiw229.
4. Group A Streptococcal Disease, Invasive (iGAS), Public Health Ontario.
Antibiotics of Little Benefit in Lower Respiratory Tract Infection
Antibiotics had no measurable effect on the severity or duration of coughs due to acute lower respiratory tract infection (LRTI, or acute bronchitis), a large prospective study found.
In fact, those receiving an antibiotic in the primary- and urgent-care setting had a small but significant increase in overall length of illness (17.5 vs 15.9 days; P = .05) — largely because patients with longer illness before the index visit were more likely to receive these drugs. The study adds further support for reducing the prescription of antibiotics for LRTIs.
“Importantly, the pathogen data demonstrated that the length of time until illness resolution for those with bacterial infection was the same as for those not receiving an antibiotic versus those receiving one (17.3 vs 17.4 days),” researchers led by Daniel J. Merenstein, MD, a professor and director of research programs, family medicine, at Georgetown University Medical Center in Washington, wrote in the Journal of General Internal Medicine (doi: 10.1007/s11606-024-08758-y).
Patients believed an antibiotic would shorten their illness by an average of about 4 days, from 13.4 days to 9.7 days, whereas the average duration of all coughs was more than 2 weeks regardless of pathogen type or receipt of an antibiotic.
“Patients had unrealistic expectations regarding the duration of LRTI and the effect of antibiotics, which should be the target of antibiotic stewardship efforts,” the group wrote.
LRTIs can, however, be dangerous, with 3%-5% progressing to pneumonia, “but not everyone has easy access at an initial visit to an x-ray, which may be the reason clinicians still give antibiotics without any other evidence of a bacterial infection,” Dr. Merenstein said in a news release. “Patients have come to expect antibiotics for a cough, even if it doesn’t help. Basic symptom-relieving medications plus time bring a resolution to most people’s infections.”
The authors noted that cough is the most common reason for an ambulatory care visit, accounting for 2.7 million outpatient visits and more than 4 million emergency department visits annually.
Risks
Overuse of antibiotics can result in dizziness, nausea, diarrhea, and rash, along with a roughly 4% chance of serious adverse effects including anaphylaxis; Stevens-Johnson syndrome, a serious skin and mucous membrane disorder; and Clostridioides difficile-associated diarrhea.
An estimated half of all antibiotic prescriptions for acute respiratory conditions are unnecessary. Before the COVID-19 pandemic, antibiotics were prescribed about 70% of the time for a diagnosis of uncomplicated cough and LRTI. The viral pandemic did not change this practice according to a meta-analysis of 130 studies showing that 78% of COVID-19 patients were prescribed an antibiotic.
The study
The study looked at a cohort of 718 patients, with a mean age of 38.9 years, 65.3% female, of whom 207 received an antibiotic and 511 did not. Of those with baseline data, 29% had an antibiotic prescribed at baseline, the most common (in 85%) being amoxicillin-clavulanate, azithromycin, doxycycline, and amoxicillin. Antibiotics had no effect on the duration or overall severity of cough in viral, bacterial, or mixed infections. Receipt of an antibiotic did, however, reduce the likelihood of a follow-up visit: 14.1% vs 8.2% (adjusted odds ratio, 0.47; 95% confidence interval, 0.26-0.84) — perhaps because it removed the motivation for seeking another consultation. Antibiotic recipients were more likely to receive a systemic corticosteroid (31.9% vs 4.5%, P <.001) and were also more likely to receive an albuterol inhaler (22.7% vs 7.6%, P <.001).
Jeffrey A. Linder, MD, MPH, a primary care physician and chief of internal medicine and geriatrics at Northwestern University Feinberg School of Medicine in Chicago, agrees that in the vast majority of LRTIs — usually acute bronchitis — antibiotics do not speed the healing process. “Forty years of research show that antibiotics do not make acute bronchitis go away any faster,” Dr. Linder, who was not involved in the current study, said in an interview. “There’s even growing evidence that a lot of pneumonia is viral as well, and 10 or 20 years from now we may often not be giving antibiotics for pneumonia because we’ll be able to see better if it’s caused by a virus.”
A large 2018 review by Dr. Linder and associates reported that 46% of antibiotics were prescribed without any infection-related diagnosis code and 20% without an office visit.
Dr. Linder routinely informs patients requesting an antibiotic about the risks of putting an ineffective chemical into their body. “I stress that it can cause rash and other allergic reactions, and even promote C diff infection,” he said. “And I also say it messes with the good bacteria in the microbiome, and they usually come around.”
Patients need to know, Dr. Linder added, that the normal course of healing the respiratory tract after acute bronchitis takes weeks. While a wet cough with sputum or phlegm will last a few days, it’s replaced with a dry annoying cough that persists for up to 3 weeks. “As long as they’re feeling generally better, that cough is normal,” he said. “A virus has run roughshod over their airways and they need a long time to heal and the cough is part of the healing process. Think how long it takes to heal a cut on a finger.”
In an era of escalating antimicrobial resistance fueled by antibiotic overuse, it’s become increasingly important to reserve antibiotics for necessary cases. According to a recent World Health Organization call to action, “Uncontrolled antimicrobial resistance is expected to lower life expectancy and lead to unprecedented health expenditure and economic losses.”
That said, there is important clinical work to be done to determine if there is a limited role for antibiotics in patients with cough, perhaps based on age and baseline severity. “Serious cough symptoms and how to treat them properly needs to be studied more, perhaps in a randomized clinical trial as this study was observational and there haven’t been any randomized trials looking at this issue since about 2012,” Dr. Merenstein said.
This research was funded by the Agency for Healthcare Research and Quality. The authors have no conflicts of interest to declare. Dr. Linder reported stock ownership in pharmaceutical companies but none that make antibiotics or other infectious disease drugs.
Antibiotics had no measurable effect on the severity or duration of coughs due to acute lower respiratory tract infection (LRTI, or acute bronchitis), a large prospective study found.
In fact, those receiving an antibiotic in the primary- and urgent-care setting had a small but significant increase in overall length of illness (17.5 vs 15.9 days; P = .05) — largely because patients with longer illness before the index visit were more likely to receive these drugs. The study adds further support for reducing the prescription of antibiotics for LRTIs.
“Importantly, the pathogen data demonstrated that the length of time until illness resolution for those with bacterial infection was the same as for those not receiving an antibiotic versus those receiving one (17.3 vs 17.4 days),” researchers led by Daniel J. Merenstein, MD, a professor and director of research programs, family medicine, at Georgetown University Medical Center in Washington, wrote in the Journal of General Internal Medicine (doi: 10.1007/s11606-024-08758-y).
Patients believed an antibiotic would shorten their illness by an average of about 4 days, from 13.4 days to 9.7 days, whereas the average duration of all coughs was more than 2 weeks regardless of pathogen type or receipt of an antibiotic.
“Patients had unrealistic expectations regarding the duration of LRTI and the effect of antibiotics, which should be the target of antibiotic stewardship efforts,” the group wrote.
LRTIs can, however, be dangerous, with 3%-5% progressing to pneumonia, “but not everyone has easy access at an initial visit to an x-ray, which may be the reason clinicians still give antibiotics without any other evidence of a bacterial infection,” Dr. Merenstein said in a news release. “Patients have come to expect antibiotics for a cough, even if it doesn’t help. Basic symptom-relieving medications plus time bring a resolution to most people’s infections.”
The authors noted that cough is the most common reason for an ambulatory care visit, accounting for 2.7 million outpatient visits and more than 4 million emergency department visits annually.
Risks
Overuse of antibiotics can result in dizziness, nausea, diarrhea, and rash, along with a roughly 4% chance of serious adverse effects including anaphylaxis; Stevens-Johnson syndrome, a serious skin and mucous membrane disorder; and Clostridioides difficile-associated diarrhea.
An estimated half of all antibiotic prescriptions for acute respiratory conditions are unnecessary. Before the COVID-19 pandemic, antibiotics were prescribed about 70% of the time for a diagnosis of uncomplicated cough and LRTI. The viral pandemic did not change this practice according to a meta-analysis of 130 studies showing that 78% of COVID-19 patients were prescribed an antibiotic.
The study
The study looked at a cohort of 718 patients, with a mean age of 38.9 years, 65.3% female, of whom 207 received an antibiotic and 511 did not. Of those with baseline data, 29% had an antibiotic prescribed at baseline, the most common (in 85%) being amoxicillin-clavulanate, azithromycin, doxycycline, and amoxicillin. Antibiotics had no effect on the duration or overall severity of cough in viral, bacterial, or mixed infections. Receipt of an antibiotic did, however, reduce the likelihood of a follow-up visit: 14.1% vs 8.2% (adjusted odds ratio, 0.47; 95% confidence interval, 0.26-0.84) — perhaps because it removed the motivation for seeking another consultation. Antibiotic recipients were more likely to receive a systemic corticosteroid (31.9% vs 4.5%, P <.001) and were also more likely to receive an albuterol inhaler (22.7% vs 7.6%, P <.001).
Jeffrey A. Linder, MD, MPH, a primary care physician and chief of internal medicine and geriatrics at Northwestern University Feinberg School of Medicine in Chicago, agrees that in the vast majority of LRTIs — usually acute bronchitis — antibiotics do not speed the healing process. “Forty years of research show that antibiotics do not make acute bronchitis go away any faster,” Dr. Linder, who was not involved in the current study, said in an interview. “There’s even growing evidence that a lot of pneumonia is viral as well, and 10 or 20 years from now we may often not be giving antibiotics for pneumonia because we’ll be able to see better if it’s caused by a virus.”
A large 2018 review by Dr. Linder and associates reported that 46% of antibiotics were prescribed without any infection-related diagnosis code and 20% without an office visit.
Dr. Linder routinely informs patients requesting an antibiotic about the risks of putting an ineffective chemical into their body. “I stress that it can cause rash and other allergic reactions, and even promote C diff infection,” he said. “And I also say it messes with the good bacteria in the microbiome, and they usually come around.”
Patients need to know, Dr. Linder added, that the normal course of healing the respiratory tract after acute bronchitis takes weeks. While a wet cough with sputum or phlegm will last a few days, it’s replaced with a dry annoying cough that persists for up to 3 weeks. “As long as they’re feeling generally better, that cough is normal,” he said. “A virus has run roughshod over their airways and they need a long time to heal and the cough is part of the healing process. Think how long it takes to heal a cut on a finger.”
In an era of escalating antimicrobial resistance fueled by antibiotic overuse, it’s become increasingly important to reserve antibiotics for necessary cases. According to a recent World Health Organization call to action, “Uncontrolled antimicrobial resistance is expected to lower life expectancy and lead to unprecedented health expenditure and economic losses.”
That said, there is important clinical work to be done to determine if there is a limited role for antibiotics in patients with cough, perhaps based on age and baseline severity. “Serious cough symptoms and how to treat them properly needs to be studied more, perhaps in a randomized clinical trial as this study was observational and there haven’t been any randomized trials looking at this issue since about 2012,” Dr. Merenstein said.
This research was funded by the Agency for Healthcare Research and Quality. The authors have no conflicts of interest to declare. Dr. Linder reported stock ownership in pharmaceutical companies but none that make antibiotics or other infectious disease drugs.
Antibiotics had no measurable effect on the severity or duration of coughs due to acute lower respiratory tract infection (LRTI, or acute bronchitis), a large prospective study found.
In fact, those receiving an antibiotic in the primary- and urgent-care setting had a small but significant increase in overall length of illness (17.5 vs 15.9 days; P = .05) — largely because patients with longer illness before the index visit were more likely to receive these drugs. The study adds further support for reducing the prescription of antibiotics for LRTIs.
“Importantly, the pathogen data demonstrated that the length of time until illness resolution for those with bacterial infection was the same as for those not receiving an antibiotic versus those receiving one (17.3 vs 17.4 days),” researchers led by Daniel J. Merenstein, MD, a professor and director of research programs, family medicine, at Georgetown University Medical Center in Washington, wrote in the Journal of General Internal Medicine (doi: 10.1007/s11606-024-08758-y).
Patients believed an antibiotic would shorten their illness by an average of about 4 days, from 13.4 days to 9.7 days, whereas the average duration of all coughs was more than 2 weeks regardless of pathogen type or receipt of an antibiotic.
“Patients had unrealistic expectations regarding the duration of LRTI and the effect of antibiotics, which should be the target of antibiotic stewardship efforts,” the group wrote.
LRTIs can, however, be dangerous, with 3%-5% progressing to pneumonia, “but not everyone has easy access at an initial visit to an x-ray, which may be the reason clinicians still give antibiotics without any other evidence of a bacterial infection,” Dr. Merenstein said in a news release. “Patients have come to expect antibiotics for a cough, even if it doesn’t help. Basic symptom-relieving medications plus time bring a resolution to most people’s infections.”
The authors noted that cough is the most common reason for an ambulatory care visit, accounting for 2.7 million outpatient visits and more than 4 million emergency department visits annually.
Risks
Overuse of antibiotics can result in dizziness, nausea, diarrhea, and rash, along with a roughly 4% chance of serious adverse effects including anaphylaxis; Stevens-Johnson syndrome, a serious skin and mucous membrane disorder; and Clostridioides difficile-associated diarrhea.
An estimated half of all antibiotic prescriptions for acute respiratory conditions are unnecessary. Before the COVID-19 pandemic, antibiotics were prescribed about 70% of the time for a diagnosis of uncomplicated cough and LRTI. The viral pandemic did not change this practice according to a meta-analysis of 130 studies showing that 78% of COVID-19 patients were prescribed an antibiotic.
The study
The study looked at a cohort of 718 patients, with a mean age of 38.9 years, 65.3% female, of whom 207 received an antibiotic and 511 did not. Of those with baseline data, 29% had an antibiotic prescribed at baseline, the most common (in 85%) being amoxicillin-clavulanate, azithromycin, doxycycline, and amoxicillin. Antibiotics had no effect on the duration or overall severity of cough in viral, bacterial, or mixed infections. Receipt of an antibiotic did, however, reduce the likelihood of a follow-up visit: 14.1% vs 8.2% (adjusted odds ratio, 0.47; 95% confidence interval, 0.26-0.84) — perhaps because it removed the motivation for seeking another consultation. Antibiotic recipients were more likely to receive a systemic corticosteroid (31.9% vs 4.5%, P <.001) and were also more likely to receive an albuterol inhaler (22.7% vs 7.6%, P <.001).
Jeffrey A. Linder, MD, MPH, a primary care physician and chief of internal medicine and geriatrics at Northwestern University Feinberg School of Medicine in Chicago, agrees that in the vast majority of LRTIs — usually acute bronchitis — antibiotics do not speed the healing process. “Forty years of research show that antibiotics do not make acute bronchitis go away any faster,” Dr. Linder, who was not involved in the current study, said in an interview. “There’s even growing evidence that a lot of pneumonia is viral as well, and 10 or 20 years from now we may often not be giving antibiotics for pneumonia because we’ll be able to see better if it’s caused by a virus.”
A large 2018 review by Dr. Linder and associates reported that 46% of antibiotics were prescribed without any infection-related diagnosis code and 20% without an office visit.
Dr. Linder routinely informs patients requesting an antibiotic about the risks of putting an ineffective chemical into their body. “I stress that it can cause rash and other allergic reactions, and even promote C diff infection,” he said. “And I also say it messes with the good bacteria in the microbiome, and they usually come around.”
Patients need to know, Dr. Linder added, that the normal course of healing the respiratory tract after acute bronchitis takes weeks. While a wet cough with sputum or phlegm will last a few days, it’s replaced with a dry annoying cough that persists for up to 3 weeks. “As long as they’re feeling generally better, that cough is normal,” he said. “A virus has run roughshod over their airways and they need a long time to heal and the cough is part of the healing process. Think how long it takes to heal a cut on a finger.”
In an era of escalating antimicrobial resistance fueled by antibiotic overuse, it’s become increasingly important to reserve antibiotics for necessary cases. According to a recent World Health Organization call to action, “Uncontrolled antimicrobial resistance is expected to lower life expectancy and lead to unprecedented health expenditure and economic losses.”
That said, there is important clinical work to be done to determine if there is a limited role for antibiotics in patients with cough, perhaps based on age and baseline severity. “Serious cough symptoms and how to treat them properly needs to be studied more, perhaps in a randomized clinical trial as this study was observational and there haven’t been any randomized trials looking at this issue since about 2012,” Dr. Merenstein said.
This research was funded by the Agency for Healthcare Research and Quality. The authors have no conflicts of interest to declare. Dr. Linder reported stock ownership in pharmaceutical companies but none that make antibiotics or other infectious disease drugs.
FROM JOURNAL OF GENERAL INTERNAL MEDICINE
European Scientists Assess Avian Flu Pandemic Risk
As avian influenza continues to spread among wild bird populations in the European Union (EU), scientists have described a wide range of factors that could drive the virus to spread efficiently among humans, thereby increasing its pandemic potential.
Although transmission of avian influenza A(H5N1) from infected birds to humans is rare, “new strains carrying potential mutations for mammalian adaptation” could occur, according to a report issued on April 3 by the European Centre for Disease Prevention and Control and the European Food Safety Authority. The analysis identified a threat of strains currently circulating outside Europe that could enter the EU and the wider European Economic Area (EEA).
“If avian A(H5N1) influenza viruses acquire the ability to spread efficiently among humans, large-scale transmission could occur due to the lack of immune defenses against H5 viruses in humans,” the report warned.
Evolution of Avian Influenza Remains Hard to Predict
However, despite many occurrences of human exposure to avian influenza since 2020, “no symptomatic or productive infection in a human has been identified in the EU/EEA,” the scientists stated. Furthermore, after almost three decades of human exposure to the A(H5N1) virus of the Gs/GD lineage, the virus has not yet acquired the mutations required for airborne transmissibility between humans. However, it remains “difficult to predict the evolutionary direction the virus will take in the future,” the scientists assessed.
“Clearly, humans are being exposed in the current USA cattle outbreak,” Professor James Wood, infectious disease epidemiologist at the University of Cambridge, United Kingdom, told this news organization. “But, arguably, what is more significant is how few cases there have been with this virus lineage and its close relatives, despite massive global exposures over the last 3 years. All diagnosed human cases seem to have been singletons, with no evidence of human-to-human transmission.”
Ian Jones, professor of virology at the University of Reading, United Kingdom, sees no evidence of an imminent spillover of avian influenza from birds. But he told this news organization: “The trouble is, the clock resets every minute. Every time the virus has come out of a bird and gone somewhere, the clock is reset. So you can never say that just because it hasn’t happened since whenever, it’s never going to happen.”
Preventive Measures Recommended
The European report recommended a range of cautionary measures that included enhanced surveillance, access to rapid diagnostics, and sharing of genetic sequence data. It urged EU authorities to work together, adopting a One Health perspective, to limit the exposure of mammals, including humans, to avian influenza viruses.
Sarah Pitt, a microbiologist at the University of Brighton, United Kingdom, said the emphasis on authorities taking a One Health approach was sound. “You’re looking at humans, animals, plants, and the environment and how they’re all closely interacted,” she told this news organization. “Putting all those things together is actually going to be good for human health. So they’ve mentioned One Health a lot and I’m sure that’s on purpose because it’s the latest buzzword, and presumably it’s a way of getting governments to take it seriously.”
Overall, Dr. Pitt believes the document is designed to move zoonotic infectious diseases a bit higher up the agenda. “They should have been higher up the agenda before COVID,” she said.
The report also called for consideration of preventative measures, such as vaccination of poultry flocks.
Overall, Dr. Jones assesses the European report as “a reworking of what’s been pretty well covered over the years.” Despite extensive work by scientists in the field, he said: “I’m not sure we’re any better at predicting an emerging virus than we’ve ever been. I would point out that we didn’t spot SARS-CoV-2 coming, even though we had SARS-CoV-1 a few years earlier. Nobody spotted the 2009 pandemic from influenza, even though there was a lot of surveillance around at the time.”
A version of this article appeared on Medscape.com.
As avian influenza continues to spread among wild bird populations in the European Union (EU), scientists have described a wide range of factors that could drive the virus to spread efficiently among humans, thereby increasing its pandemic potential.
Although transmission of avian influenza A(H5N1) from infected birds to humans is rare, “new strains carrying potential mutations for mammalian adaptation” could occur, according to a report issued on April 3 by the European Centre for Disease Prevention and Control and the European Food Safety Authority. The analysis identified a threat of strains currently circulating outside Europe that could enter the EU and the wider European Economic Area (EEA).
“If avian A(H5N1) influenza viruses acquire the ability to spread efficiently among humans, large-scale transmission could occur due to the lack of immune defenses against H5 viruses in humans,” the report warned.
Evolution of Avian Influenza Remains Hard to Predict
However, despite many occurrences of human exposure to avian influenza since 2020, “no symptomatic or productive infection in a human has been identified in the EU/EEA,” the scientists stated. Furthermore, after almost three decades of human exposure to the A(H5N1) virus of the Gs/GD lineage, the virus has not yet acquired the mutations required for airborne transmissibility between humans. However, it remains “difficult to predict the evolutionary direction the virus will take in the future,” the scientists assessed.
“Clearly, humans are being exposed in the current USA cattle outbreak,” Professor James Wood, infectious disease epidemiologist at the University of Cambridge, United Kingdom, told this news organization. “But, arguably, what is more significant is how few cases there have been with this virus lineage and its close relatives, despite massive global exposures over the last 3 years. All diagnosed human cases seem to have been singletons, with no evidence of human-to-human transmission.”
Ian Jones, professor of virology at the University of Reading, United Kingdom, sees no evidence of an imminent spillover of avian influenza from birds. But he told this news organization: “The trouble is, the clock resets every minute. Every time the virus has come out of a bird and gone somewhere, the clock is reset. So you can never say that just because it hasn’t happened since whenever, it’s never going to happen.”
Preventive Measures Recommended
The European report recommended a range of cautionary measures that included enhanced surveillance, access to rapid diagnostics, and sharing of genetic sequence data. It urged EU authorities to work together, adopting a One Health perspective, to limit the exposure of mammals, including humans, to avian influenza viruses.
Sarah Pitt, a microbiologist at the University of Brighton, United Kingdom, said the emphasis on authorities taking a One Health approach was sound. “You’re looking at humans, animals, plants, and the environment and how they’re all closely interacted,” she told this news organization. “Putting all those things together is actually going to be good for human health. So they’ve mentioned One Health a lot and I’m sure that’s on purpose because it’s the latest buzzword, and presumably it’s a way of getting governments to take it seriously.”
Overall, Dr. Pitt believes the document is designed to move zoonotic infectious diseases a bit higher up the agenda. “They should have been higher up the agenda before COVID,” she said.
The report also called for consideration of preventative measures, such as vaccination of poultry flocks.
Overall, Dr. Jones assesses the European report as “a reworking of what’s been pretty well covered over the years.” Despite extensive work by scientists in the field, he said: “I’m not sure we’re any better at predicting an emerging virus than we’ve ever been. I would point out that we didn’t spot SARS-CoV-2 coming, even though we had SARS-CoV-1 a few years earlier. Nobody spotted the 2009 pandemic from influenza, even though there was a lot of surveillance around at the time.”
A version of this article appeared on Medscape.com.
As avian influenza continues to spread among wild bird populations in the European Union (EU), scientists have described a wide range of factors that could drive the virus to spread efficiently among humans, thereby increasing its pandemic potential.
Although transmission of avian influenza A(H5N1) from infected birds to humans is rare, “new strains carrying potential mutations for mammalian adaptation” could occur, according to a report issued on April 3 by the European Centre for Disease Prevention and Control and the European Food Safety Authority. The analysis identified a threat of strains currently circulating outside Europe that could enter the EU and the wider European Economic Area (EEA).
“If avian A(H5N1) influenza viruses acquire the ability to spread efficiently among humans, large-scale transmission could occur due to the lack of immune defenses against H5 viruses in humans,” the report warned.
Evolution of Avian Influenza Remains Hard to Predict
However, despite many occurrences of human exposure to avian influenza since 2020, “no symptomatic or productive infection in a human has been identified in the EU/EEA,” the scientists stated. Furthermore, after almost three decades of human exposure to the A(H5N1) virus of the Gs/GD lineage, the virus has not yet acquired the mutations required for airborne transmissibility between humans. However, it remains “difficult to predict the evolutionary direction the virus will take in the future,” the scientists assessed.
“Clearly, humans are being exposed in the current USA cattle outbreak,” Professor James Wood, infectious disease epidemiologist at the University of Cambridge, United Kingdom, told this news organization. “But, arguably, what is more significant is how few cases there have been with this virus lineage and its close relatives, despite massive global exposures over the last 3 years. All diagnosed human cases seem to have been singletons, with no evidence of human-to-human transmission.”
Ian Jones, professor of virology at the University of Reading, United Kingdom, sees no evidence of an imminent spillover of avian influenza from birds. But he told this news organization: “The trouble is, the clock resets every minute. Every time the virus has come out of a bird and gone somewhere, the clock is reset. So you can never say that just because it hasn’t happened since whenever, it’s never going to happen.”
Preventive Measures Recommended
The European report recommended a range of cautionary measures that included enhanced surveillance, access to rapid diagnostics, and sharing of genetic sequence data. It urged EU authorities to work together, adopting a One Health perspective, to limit the exposure of mammals, including humans, to avian influenza viruses.
Sarah Pitt, a microbiologist at the University of Brighton, United Kingdom, said the emphasis on authorities taking a One Health approach was sound. “You’re looking at humans, animals, plants, and the environment and how they’re all closely interacted,” she told this news organization. “Putting all those things together is actually going to be good for human health. So they’ve mentioned One Health a lot and I’m sure that’s on purpose because it’s the latest buzzword, and presumably it’s a way of getting governments to take it seriously.”
Overall, Dr. Pitt believes the document is designed to move zoonotic infectious diseases a bit higher up the agenda. “They should have been higher up the agenda before COVID,” she said.
The report also called for consideration of preventative measures, such as vaccination of poultry flocks.
Overall, Dr. Jones assesses the European report as “a reworking of what’s been pretty well covered over the years.” Despite extensive work by scientists in the field, he said: “I’m not sure we’re any better at predicting an emerging virus than we’ve ever been. I would point out that we didn’t spot SARS-CoV-2 coming, even though we had SARS-CoV-1 a few years earlier. Nobody spotted the 2009 pandemic from influenza, even though there was a lot of surveillance around at the time.”
A version of this article appeared on Medscape.com.
Hepatitis E Vaccine Shows Long-Term Efficacy
The hepatitis E virus (HEV) is among the leading global causes of acute viral hepatitis. Molecular studies of HEV strains have identified four main genotypes. Genotypes 1 and 2 are limited to humans and are transmitted through contaminated water in resource-limited countries, mainly in Asia. Genotypes 3 and 4 are zoonotic, causing sporadic indigenous hepatitis E in nearly all countries.
Each year, approximately 20 million HEV infections occur worldwide, resulting in around 3.3 million symptomatic infections and 70,000 deaths. Despite this toll, HEV infection remains underestimated, and Western countries are likely not immune to the virus. To date, two recombinant vaccines against hepatitis E, based on genotype 1, have been developed and approved in China, but further studies are needed to determine the duration of vaccination protection.
Ten-Year Results
This study is an extension of a randomized, double-blind, placebo-controlled phase 3 clinical trial of the Hecolin hepatitis E vaccine that was conducted in Dongtai County, Jiangsu, China. In the initial trial, healthy adults aged 16-65 years were recruited, stratified by age and sex, and randomly assigned in a 1:1 ratio to receive three doses of intramuscular hepatitis E vaccine or placebo at months 0, 1, and 6.
A hepatitis E surveillance system, including 205 clinical sentinels covering the entire study region, was established before the study began and maintained for 10 years after vaccination to identify individuals with suspected hepatitis. In addition, an external control cohort was formed to assess vaccine efficacy. The primary endpoint was the vaccine’s efficacy in preventing confirmed hepatitis E occurring at least 30 days after the administration of the third vaccine dose.
Follow-up occurred every 3 months. Participants with hepatitis symptoms for 3 days or more underwent alanine aminotransferase (ALT) concentration measurement. Patients with ALT concentrations ≥ 2.5 times the upper limit of normal were considered to have acute hepatitis. A diagnosis of HEV-confirmed infection was made for patients with acute hepatitis presenting with at least two of the following markers: Presence of HEV RNA, presence of positive anti-HEV immunoglobulin (Ig) M antibodies, and at least fourfold increase in anti-HEV IgG concentrations.
For the efficacy analysis, a Poisson regression model was used to estimate the relative risk and its 95% CI of incidence between groups. Incidence was reported as the number of patients with hepatitis E per 10,000 person-years.
Immunogenicity persistence was assessed by measuring anti-HEV IgG in participants. Serum samples were collected at months 0, 7, 13, 19, 31, 43, 55, 79, and 103 for Qingdao district participants and at months 0, 7, 19, 31, 43, 67, and 91 for Anfeng district participants.
Efficacy and Duration
The follow-up period extended from 2007 to 2017. In total, 97,356 participants completed the three-dose regimen and were included in the per-protocol population (48,693 in the vaccine group and 48,663 in the placebo group), and 178,236 residents from the study region participated in the external control cohort. During the study period, 90 cases of hepatitis E were identified, with 13 in the vaccine group (0.2 per 10,000 person-years) and 77 in the placebo group (1.4 per 10,000 person-years). This indicated a vaccine efficacy of 86.6% in the per-protocol analysis.
In the subgroups evaluated for immunogenicity persistence, among those who were initially seronegative and received three doses of hepatitis E vaccine, 254 out of 291 vaccinated participants (87.3%) in Qingdao after 8.5 years and 1270 (73.0%) out of 1740 vaccinated participants in Anfeng after 7.5 years maintained detectable antibody concentrations.
The identification of infections despite vaccination is notable, especially with eight cases occurring beyond the fourth year following the last dose. This information is crucial for understanding potential immunity decline over time and highlights the importance of exploring various vaccination strategies to optimize protection.
An ongoing phase 4 clinical trial in Bangladesh, exploring different administration schedules and target populations, could help optimize vaccination strategies. The remarkable efficacy (100%) observed over a 30-month period for the two-dose schedule (doses are administered 1 month apart) is promising.
The observation of higher IgG antibody avidity in participants with infections despite vaccination underscores the importance of robust antibody responses to mitigate disease severity and duration. Several study limitations, such as lack of data on deaths and emigrations, a single-center study design, predominance of genotype 4 infections, and the risk for bias in the external control cohort, should be acknowledged.
In conclusion, this study provides compelling evidence of sustained protection of the hepatitis E vaccine over a decade. The observed persistence of induced antibodies for at least 8.5 years supports the long-term efficacy of the vaccine. Diverse global trials, further investigation into the impact of natural infections on vaccine-induced antibodies, and confirmation of inter-genotypic protection are needed.
This story was translated from JIM, which is part of the Medscape professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
The hepatitis E virus (HEV) is among the leading global causes of acute viral hepatitis. Molecular studies of HEV strains have identified four main genotypes. Genotypes 1 and 2 are limited to humans and are transmitted through contaminated water in resource-limited countries, mainly in Asia. Genotypes 3 and 4 are zoonotic, causing sporadic indigenous hepatitis E in nearly all countries.
Each year, approximately 20 million HEV infections occur worldwide, resulting in around 3.3 million symptomatic infections and 70,000 deaths. Despite this toll, HEV infection remains underestimated, and Western countries are likely not immune to the virus. To date, two recombinant vaccines against hepatitis E, based on genotype 1, have been developed and approved in China, but further studies are needed to determine the duration of vaccination protection.
Ten-Year Results
This study is an extension of a randomized, double-blind, placebo-controlled phase 3 clinical trial of the Hecolin hepatitis E vaccine that was conducted in Dongtai County, Jiangsu, China. In the initial trial, healthy adults aged 16-65 years were recruited, stratified by age and sex, and randomly assigned in a 1:1 ratio to receive three doses of intramuscular hepatitis E vaccine or placebo at months 0, 1, and 6.
A hepatitis E surveillance system, including 205 clinical sentinels covering the entire study region, was established before the study began and maintained for 10 years after vaccination to identify individuals with suspected hepatitis. In addition, an external control cohort was formed to assess vaccine efficacy. The primary endpoint was the vaccine’s efficacy in preventing confirmed hepatitis E occurring at least 30 days after the administration of the third vaccine dose.
Follow-up occurred every 3 months. Participants with hepatitis symptoms for 3 days or more underwent alanine aminotransferase (ALT) concentration measurement. Patients with ALT concentrations ≥ 2.5 times the upper limit of normal were considered to have acute hepatitis. A diagnosis of HEV-confirmed infection was made for patients with acute hepatitis presenting with at least two of the following markers: Presence of HEV RNA, presence of positive anti-HEV immunoglobulin (Ig) M antibodies, and at least fourfold increase in anti-HEV IgG concentrations.
For the efficacy analysis, a Poisson regression model was used to estimate the relative risk and its 95% CI of incidence between groups. Incidence was reported as the number of patients with hepatitis E per 10,000 person-years.
Immunogenicity persistence was assessed by measuring anti-HEV IgG in participants. Serum samples were collected at months 0, 7, 13, 19, 31, 43, 55, 79, and 103 for Qingdao district participants and at months 0, 7, 19, 31, 43, 67, and 91 for Anfeng district participants.
Efficacy and Duration
The follow-up period extended from 2007 to 2017. In total, 97,356 participants completed the three-dose regimen and were included in the per-protocol population (48,693 in the vaccine group and 48,663 in the placebo group), and 178,236 residents from the study region participated in the external control cohort. During the study period, 90 cases of hepatitis E were identified, with 13 in the vaccine group (0.2 per 10,000 person-years) and 77 in the placebo group (1.4 per 10,000 person-years). This indicated a vaccine efficacy of 86.6% in the per-protocol analysis.
In the subgroups evaluated for immunogenicity persistence, among those who were initially seronegative and received three doses of hepatitis E vaccine, 254 out of 291 vaccinated participants (87.3%) in Qingdao after 8.5 years and 1270 (73.0%) out of 1740 vaccinated participants in Anfeng after 7.5 years maintained detectable antibody concentrations.
The identification of infections despite vaccination is notable, especially with eight cases occurring beyond the fourth year following the last dose. This information is crucial for understanding potential immunity decline over time and highlights the importance of exploring various vaccination strategies to optimize protection.
An ongoing phase 4 clinical trial in Bangladesh, exploring different administration schedules and target populations, could help optimize vaccination strategies. The remarkable efficacy (100%) observed over a 30-month period for the two-dose schedule (doses are administered 1 month apart) is promising.
The observation of higher IgG antibody avidity in participants with infections despite vaccination underscores the importance of robust antibody responses to mitigate disease severity and duration. Several study limitations, such as lack of data on deaths and emigrations, a single-center study design, predominance of genotype 4 infections, and the risk for bias in the external control cohort, should be acknowledged.
In conclusion, this study provides compelling evidence of sustained protection of the hepatitis E vaccine over a decade. The observed persistence of induced antibodies for at least 8.5 years supports the long-term efficacy of the vaccine. Diverse global trials, further investigation into the impact of natural infections on vaccine-induced antibodies, and confirmation of inter-genotypic protection are needed.
This story was translated from JIM, which is part of the Medscape professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
The hepatitis E virus (HEV) is among the leading global causes of acute viral hepatitis. Molecular studies of HEV strains have identified four main genotypes. Genotypes 1 and 2 are limited to humans and are transmitted through contaminated water in resource-limited countries, mainly in Asia. Genotypes 3 and 4 are zoonotic, causing sporadic indigenous hepatitis E in nearly all countries.
Each year, approximately 20 million HEV infections occur worldwide, resulting in around 3.3 million symptomatic infections and 70,000 deaths. Despite this toll, HEV infection remains underestimated, and Western countries are likely not immune to the virus. To date, two recombinant vaccines against hepatitis E, based on genotype 1, have been developed and approved in China, but further studies are needed to determine the duration of vaccination protection.
Ten-Year Results
This study is an extension of a randomized, double-blind, placebo-controlled phase 3 clinical trial of the Hecolin hepatitis E vaccine that was conducted in Dongtai County, Jiangsu, China. In the initial trial, healthy adults aged 16-65 years were recruited, stratified by age and sex, and randomly assigned in a 1:1 ratio to receive three doses of intramuscular hepatitis E vaccine or placebo at months 0, 1, and 6.
A hepatitis E surveillance system, including 205 clinical sentinels covering the entire study region, was established before the study began and maintained for 10 years after vaccination to identify individuals with suspected hepatitis. In addition, an external control cohort was formed to assess vaccine efficacy. The primary endpoint was the vaccine’s efficacy in preventing confirmed hepatitis E occurring at least 30 days after the administration of the third vaccine dose.
Follow-up occurred every 3 months. Participants with hepatitis symptoms for 3 days or more underwent alanine aminotransferase (ALT) concentration measurement. Patients with ALT concentrations ≥ 2.5 times the upper limit of normal were considered to have acute hepatitis. A diagnosis of HEV-confirmed infection was made for patients with acute hepatitis presenting with at least two of the following markers: Presence of HEV RNA, presence of positive anti-HEV immunoglobulin (Ig) M antibodies, and at least fourfold increase in anti-HEV IgG concentrations.
For the efficacy analysis, a Poisson regression model was used to estimate the relative risk and its 95% CI of incidence between groups. Incidence was reported as the number of patients with hepatitis E per 10,000 person-years.
Immunogenicity persistence was assessed by measuring anti-HEV IgG in participants. Serum samples were collected at months 0, 7, 13, 19, 31, 43, 55, 79, and 103 for Qingdao district participants and at months 0, 7, 19, 31, 43, 67, and 91 for Anfeng district participants.
Efficacy and Duration
The follow-up period extended from 2007 to 2017. In total, 97,356 participants completed the three-dose regimen and were included in the per-protocol population (48,693 in the vaccine group and 48,663 in the placebo group), and 178,236 residents from the study region participated in the external control cohort. During the study period, 90 cases of hepatitis E were identified, with 13 in the vaccine group (0.2 per 10,000 person-years) and 77 in the placebo group (1.4 per 10,000 person-years). This indicated a vaccine efficacy of 86.6% in the per-protocol analysis.
In the subgroups evaluated for immunogenicity persistence, among those who were initially seronegative and received three doses of hepatitis E vaccine, 254 out of 291 vaccinated participants (87.3%) in Qingdao after 8.5 years and 1270 (73.0%) out of 1740 vaccinated participants in Anfeng after 7.5 years maintained detectable antibody concentrations.
The identification of infections despite vaccination is notable, especially with eight cases occurring beyond the fourth year following the last dose. This information is crucial for understanding potential immunity decline over time and highlights the importance of exploring various vaccination strategies to optimize protection.
An ongoing phase 4 clinical trial in Bangladesh, exploring different administration schedules and target populations, could help optimize vaccination strategies. The remarkable efficacy (100%) observed over a 30-month period for the two-dose schedule (doses are administered 1 month apart) is promising.
The observation of higher IgG antibody avidity in participants with infections despite vaccination underscores the importance of robust antibody responses to mitigate disease severity and duration. Several study limitations, such as lack of data on deaths and emigrations, a single-center study design, predominance of genotype 4 infections, and the risk for bias in the external control cohort, should be acknowledged.
In conclusion, this study provides compelling evidence of sustained protection of the hepatitis E vaccine over a decade. The observed persistence of induced antibodies for at least 8.5 years supports the long-term efficacy of the vaccine. Diverse global trials, further investigation into the impact of natural infections on vaccine-induced antibodies, and confirmation of inter-genotypic protection are needed.
This story was translated from JIM, which is part of the Medscape professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Is It Time to Stop Using the Term AIDS?
The acronym AIDS is redundant, loaded with stigma, and potentially harmful, according to a group of specialists who suggest replacing the term with “advanced HIV.”
Mexico, and colleagues.
People generally associate the acronym AIDS with patients who have no available treatment options and a short life expectancy, said Dr. Núñez. That mischaracterization may affect treatment decisions by patients and clinicians and could result in exaggerated infection-control measures.
Using the HIV/AIDS combination erroneously implies equivalence and can mislead the public and clinicians, which the authors explained in their Viewpoint article published in The Lancet HIV.
Original Reason for the Term
AIDS, which stands for acquired immunodeficiency syndrome, was coined in 1982 by the US Centers for Disease Control and Prevention (CDC) to name a disease with an unknown cause that affected people with weakened cell-mediated immunity.
“When HIV was found to be the cause of the disease (labeled HIV in 1986), the term AIDS, strictly speaking, became unnecessary,” Dr. Núñez said.
AIDS was originally intended as a case definition for surveillance purposes, and treatment decisions were based on whether patients met the case definition for AIDS, he pointed out.
“The fact that some people still do so in this day and age shows that this is not only unhelpful, but misleading and even harmful,” he noted. Without the label AIDS, clinicians can focus on whether and for how long people have been on treatment, whether they recently switched treatment, and other factors that will help determine appropriate care.
Some Organizations Removed AIDS From Their Names
Some organizations have already removed AIDS from their names. For example, the International AIDS Society–USA, which issues guidelines on antiretroviral treatment, changed its name to the International Antiviral Society–USA.
In 2017, the name of AIDS.gov was changed to HIV.gov. In its explanation, the group wrote, “Today, people with HIV who are diagnosed early, linked to care, start antiretroviral therapy, and take it as prescribed can achieve life-long viral suppression that prevents HIV infection from progressing to AIDS.”
A different view on the term AIDS comes from Greg Millett, MPH, vice president at the Foundation for AIDS Research (amfAR) and the director of amfAR’s Public Policy Office.
Although he believes that AIDS is an anachronistic term, as a researcher for more than 30 years in the field; a policy director in Washington; a scientist; and a person living with HIV, “it feels like a distinction without a difference. At least from where I sit, there are far more pressing issues that we’re facing as an HIV community,” Millett shared.
For instance, “we’re seeing that global, as well as domestic, HIV funding is in, by far, the most precarious position that I’ve ever seen in the field. Calling it AIDS or HIV makes no difference in trying to alleviate that jeopardy,” he said.
Millett also said that the stigma and persecution and, in some cases, criminalization of people living with HIV or AIDS is pervasive and won’t go away with a name change, which is a point the authors also acknowledged.
“We need to focus on the social determinants of health,” he said. “That is the thing that is going to move the needle among people living with HIV, not nomenclature.”
Millett likens the argument to the one between Black and African American. “As a Black American, I remember fierce debates in the early ‘90s over whether we should be called African Americans or Blacks. Some argued that African American carried greater dignity and would help with self-esteem and address inequities by emphasizing that we are American. Many others said that it doesn’t make a difference.”
“It is clear that being called African American has not fixed intractable issues like poverty, structural racism, or inequities in incarceration,” he pointed out.
End the Epidemic, Not the Name
The authors misinterpret the impact of the term on stigma, said James W. Curran, MD, MPH, dean emeritus of the Rollins School of Public Health and professor of epidemiology and global health at Emory University, both in Atlanta, Georgia. The term AIDS “is more likely attributed to the fatal nature of the infection itself,” without treatment, he explained, and the mode of transmission, exacerbated by homophobia.
“The term has been in widespread use for 40 years and recognized worldwide,” Dr. Curran, who led the nation’s efforts in the battle against HIV and AIDS at the CDC for 15 years before joining Emory as dean, said.
He also worries about the continued trajectory of lives lost: “Over 35 million people worldwide have perished from HIV/AIDS, including over 500,000 per year now.”
Meanwhile, “global programs such as PEPFAR [the US President’s Emergency Plan for AIDS Relief] are under fire and threatened by Congress as no longer necessary. Removing AIDS from the terminology may add to confusion,” making people think “that the epidemic is over,” he said.
Although the authors argue that keeping the term may cause harm, eliminating it might worsen a different kind of harm. “There is a risk that abolishing the term will further de-emphasize the importance of the problem, with no significant impact on stigma,” Dr. Curran added.
A version of this article appeared on Medscape.com.
The acronym AIDS is redundant, loaded with stigma, and potentially harmful, according to a group of specialists who suggest replacing the term with “advanced HIV.”
Mexico, and colleagues.
People generally associate the acronym AIDS with patients who have no available treatment options and a short life expectancy, said Dr. Núñez. That mischaracterization may affect treatment decisions by patients and clinicians and could result in exaggerated infection-control measures.
Using the HIV/AIDS combination erroneously implies equivalence and can mislead the public and clinicians, which the authors explained in their Viewpoint article published in The Lancet HIV.
Original Reason for the Term
AIDS, which stands for acquired immunodeficiency syndrome, was coined in 1982 by the US Centers for Disease Control and Prevention (CDC) to name a disease with an unknown cause that affected people with weakened cell-mediated immunity.
“When HIV was found to be the cause of the disease (labeled HIV in 1986), the term AIDS, strictly speaking, became unnecessary,” Dr. Núñez said.
AIDS was originally intended as a case definition for surveillance purposes, and treatment decisions were based on whether patients met the case definition for AIDS, he pointed out.
“The fact that some people still do so in this day and age shows that this is not only unhelpful, but misleading and even harmful,” he noted. Without the label AIDS, clinicians can focus on whether and for how long people have been on treatment, whether they recently switched treatment, and other factors that will help determine appropriate care.
Some Organizations Removed AIDS From Their Names
Some organizations have already removed AIDS from their names. For example, the International AIDS Society–USA, which issues guidelines on antiretroviral treatment, changed its name to the International Antiviral Society–USA.
In 2017, the name of AIDS.gov was changed to HIV.gov. In its explanation, the group wrote, “Today, people with HIV who are diagnosed early, linked to care, start antiretroviral therapy, and take it as prescribed can achieve life-long viral suppression that prevents HIV infection from progressing to AIDS.”
A different view on the term AIDS comes from Greg Millett, MPH, vice president at the Foundation for AIDS Research (amfAR) and the director of amfAR’s Public Policy Office.
Although he believes that AIDS is an anachronistic term, as a researcher for more than 30 years in the field; a policy director in Washington; a scientist; and a person living with HIV, “it feels like a distinction without a difference. At least from where I sit, there are far more pressing issues that we’re facing as an HIV community,” Millett shared.
For instance, “we’re seeing that global, as well as domestic, HIV funding is in, by far, the most precarious position that I’ve ever seen in the field. Calling it AIDS or HIV makes no difference in trying to alleviate that jeopardy,” he said.
Millett also said that the stigma and persecution and, in some cases, criminalization of people living with HIV or AIDS is pervasive and won’t go away with a name change, which is a point the authors also acknowledged.
“We need to focus on the social determinants of health,” he said. “That is the thing that is going to move the needle among people living with HIV, not nomenclature.”
Millett likens the argument to the one between Black and African American. “As a Black American, I remember fierce debates in the early ‘90s over whether we should be called African Americans or Blacks. Some argued that African American carried greater dignity and would help with self-esteem and address inequities by emphasizing that we are American. Many others said that it doesn’t make a difference.”
“It is clear that being called African American has not fixed intractable issues like poverty, structural racism, or inequities in incarceration,” he pointed out.
End the Epidemic, Not the Name
The authors misinterpret the impact of the term on stigma, said James W. Curran, MD, MPH, dean emeritus of the Rollins School of Public Health and professor of epidemiology and global health at Emory University, both in Atlanta, Georgia. The term AIDS “is more likely attributed to the fatal nature of the infection itself,” without treatment, he explained, and the mode of transmission, exacerbated by homophobia.
“The term has been in widespread use for 40 years and recognized worldwide,” Dr. Curran, who led the nation’s efforts in the battle against HIV and AIDS at the CDC for 15 years before joining Emory as dean, said.
He also worries about the continued trajectory of lives lost: “Over 35 million people worldwide have perished from HIV/AIDS, including over 500,000 per year now.”
Meanwhile, “global programs such as PEPFAR [the US President’s Emergency Plan for AIDS Relief] are under fire and threatened by Congress as no longer necessary. Removing AIDS from the terminology may add to confusion,” making people think “that the epidemic is over,” he said.
Although the authors argue that keeping the term may cause harm, eliminating it might worsen a different kind of harm. “There is a risk that abolishing the term will further de-emphasize the importance of the problem, with no significant impact on stigma,” Dr. Curran added.
A version of this article appeared on Medscape.com.
The acronym AIDS is redundant, loaded with stigma, and potentially harmful, according to a group of specialists who suggest replacing the term with “advanced HIV.”
Mexico, and colleagues.
People generally associate the acronym AIDS with patients who have no available treatment options and a short life expectancy, said Dr. Núñez. That mischaracterization may affect treatment decisions by patients and clinicians and could result in exaggerated infection-control measures.
Using the HIV/AIDS combination erroneously implies equivalence and can mislead the public and clinicians, which the authors explained in their Viewpoint article published in The Lancet HIV.
Original Reason for the Term
AIDS, which stands for acquired immunodeficiency syndrome, was coined in 1982 by the US Centers for Disease Control and Prevention (CDC) to name a disease with an unknown cause that affected people with weakened cell-mediated immunity.
“When HIV was found to be the cause of the disease (labeled HIV in 1986), the term AIDS, strictly speaking, became unnecessary,” Dr. Núñez said.
AIDS was originally intended as a case definition for surveillance purposes, and treatment decisions were based on whether patients met the case definition for AIDS, he pointed out.
“The fact that some people still do so in this day and age shows that this is not only unhelpful, but misleading and even harmful,” he noted. Without the label AIDS, clinicians can focus on whether and for how long people have been on treatment, whether they recently switched treatment, and other factors that will help determine appropriate care.
Some Organizations Removed AIDS From Their Names
Some organizations have already removed AIDS from their names. For example, the International AIDS Society–USA, which issues guidelines on antiretroviral treatment, changed its name to the International Antiviral Society–USA.
In 2017, the name of AIDS.gov was changed to HIV.gov. In its explanation, the group wrote, “Today, people with HIV who are diagnosed early, linked to care, start antiretroviral therapy, and take it as prescribed can achieve life-long viral suppression that prevents HIV infection from progressing to AIDS.”
A different view on the term AIDS comes from Greg Millett, MPH, vice president at the Foundation for AIDS Research (amfAR) and the director of amfAR’s Public Policy Office.
Although he believes that AIDS is an anachronistic term, as a researcher for more than 30 years in the field; a policy director in Washington; a scientist; and a person living with HIV, “it feels like a distinction without a difference. At least from where I sit, there are far more pressing issues that we’re facing as an HIV community,” Millett shared.
For instance, “we’re seeing that global, as well as domestic, HIV funding is in, by far, the most precarious position that I’ve ever seen in the field. Calling it AIDS or HIV makes no difference in trying to alleviate that jeopardy,” he said.
Millett also said that the stigma and persecution and, in some cases, criminalization of people living with HIV or AIDS is pervasive and won’t go away with a name change, which is a point the authors also acknowledged.
“We need to focus on the social determinants of health,” he said. “That is the thing that is going to move the needle among people living with HIV, not nomenclature.”
Millett likens the argument to the one between Black and African American. “As a Black American, I remember fierce debates in the early ‘90s over whether we should be called African Americans or Blacks. Some argued that African American carried greater dignity and would help with self-esteem and address inequities by emphasizing that we are American. Many others said that it doesn’t make a difference.”
“It is clear that being called African American has not fixed intractable issues like poverty, structural racism, or inequities in incarceration,” he pointed out.
End the Epidemic, Not the Name
The authors misinterpret the impact of the term on stigma, said James W. Curran, MD, MPH, dean emeritus of the Rollins School of Public Health and professor of epidemiology and global health at Emory University, both in Atlanta, Georgia. The term AIDS “is more likely attributed to the fatal nature of the infection itself,” without treatment, he explained, and the mode of transmission, exacerbated by homophobia.
“The term has been in widespread use for 40 years and recognized worldwide,” Dr. Curran, who led the nation’s efforts in the battle against HIV and AIDS at the CDC for 15 years before joining Emory as dean, said.
He also worries about the continued trajectory of lives lost: “Over 35 million people worldwide have perished from HIV/AIDS, including over 500,000 per year now.”
Meanwhile, “global programs such as PEPFAR [the US President’s Emergency Plan for AIDS Relief] are under fire and threatened by Congress as no longer necessary. Removing AIDS from the terminology may add to confusion,” making people think “that the epidemic is over,” he said.
Although the authors argue that keeping the term may cause harm, eliminating it might worsen a different kind of harm. “There is a risk that abolishing the term will further de-emphasize the importance of the problem, with no significant impact on stigma,” Dr. Curran added.
A version of this article appeared on Medscape.com.
FROM THE LANCET HIV
Tuberculosis Prevention Brings Economic Gains, Says WHO
A new study conducted by the World Health Organization (WHO) suggests that in addition to providing significant improvements in public health, investment in the diagnosis and prevention of tuberculosis also generates economic benefits.
According to a survey conducted by governments and researchers from Brazil, Georgia, Kenya, and South Africa, even modest increases in funding for measures against tuberculosis can bring gains. Every $1 invested produces returns of as much as $39, it found.
The findings may remind governments and policymakers about the importance of investing in public health policies. According to the WHO, the study “provides strong economic arguments” about the true costs of tuberculosis and proves the benefits of increasing funding to accelerate the diagnosis and preventive treatment of the disease.
UN Targets Tuberculosis
In September 2023, during the last meeting of the United Nations General Assembly, following a widespread worsening of disease indicators because of the COVID-19 pandemic, world leaders signed a declaration committing to the expansion of efforts to combat tuberculosis during the next 5 years. The current WHO study was developed to provide a road map for the implementation of key measures against the disease.
The survey highlights two fundamental actions: The expansion of screening, especially in populations considered more vulnerable, and the provision of tuberculosis preventive treatment (TPT), which entails administering drugs to people who have been exposed to the disease but have not yet developed it.
“TPT is a proven and effective intervention to prevent the development of tuberculosis among exposed people, reducing the risk of developing the disease by about 60%-90% compared with individuals who did not receive it,” the document emphasized.
Investments Yield Returns
To achieve the necessary coverage levels, the study estimated that Brazil would need to increase annual per capita investment by $0.28 (about R$1.41) between 2024 and 2050. Brazilian society, in turn, would receive a return of $11 (R$55.27) for every dollar invested.
For South Africa, whose per capita investment increase is estimated at $1.10 per year, the return would be even more significant: $39 for every dollar allocated.
The WHO emphasized that funding for combating the disease is much lower than the value of the damage it causes to nations. “Tuberculosis has high costs for society. Only a small proportion of these costs go directly to the health system (ranging from 1.7% in South Africa to 7.8% in Kenya). Most are costs for patients and society.”
The study projected that between 2024 and 2050, the total cost of tuberculosis to Brazilian society would be $81.2 billion, with an average annual value of $3.01 billion. This figure represents, in 2024, 0.16% of the country’s gross domestic product.
Achieving screening and preventive treatment goals in Brazil would lead to a reduction of as much as 18% in the national incidence of the disease, as well as 1.9 million fewer deaths, between 2024 and 2050.
Although treatable and preventable, tuberculosis remains the leading cause of death from infectious agents worldwide. It is estimated that over 1.3 million people died from the disease in 2022.
The document provides the “health and economic justification for investing in evidence-based interventions recommended by WHO in tuberculosis screening and prevention,” according to WHO Director-General Tedros Adhanom Ghebreyesus, PhD.
“Today we have the knowledge, tools, and political commitment that can end this age-old disease that continues to be one of the leading causes of death from infectious diseases worldwide,” he added.
Emerging Concerns
Although the WHO highlighted the global increase in access to tuberculosis diagnosis and treatment in 2022, which coincided with the recovery of healthcare systems in several countries after the beginning of the pandemic, it emphasized that the implementation of preventive treatment for exposed individuals and high-vulnerability populations remains slow.
Another concern is the increase in drug resistance. Multidrug-resistant tuberculosis is considered a public health crisis. It is estimated that about 410,000 people had multidrug-resistant tuberculosis or rifampicin-resistant tuberculosis in 2022, but only two of every five patients had access to treatment.This story was translated from the Medscape Portuguese edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com .
A new study conducted by the World Health Organization (WHO) suggests that in addition to providing significant improvements in public health, investment in the diagnosis and prevention of tuberculosis also generates economic benefits.
According to a survey conducted by governments and researchers from Brazil, Georgia, Kenya, and South Africa, even modest increases in funding for measures against tuberculosis can bring gains. Every $1 invested produces returns of as much as $39, it found.
The findings may remind governments and policymakers about the importance of investing in public health policies. According to the WHO, the study “provides strong economic arguments” about the true costs of tuberculosis and proves the benefits of increasing funding to accelerate the diagnosis and preventive treatment of the disease.
UN Targets Tuberculosis
In September 2023, during the last meeting of the United Nations General Assembly, following a widespread worsening of disease indicators because of the COVID-19 pandemic, world leaders signed a declaration committing to the expansion of efforts to combat tuberculosis during the next 5 years. The current WHO study was developed to provide a road map for the implementation of key measures against the disease.
The survey highlights two fundamental actions: The expansion of screening, especially in populations considered more vulnerable, and the provision of tuberculosis preventive treatment (TPT), which entails administering drugs to people who have been exposed to the disease but have not yet developed it.
“TPT is a proven and effective intervention to prevent the development of tuberculosis among exposed people, reducing the risk of developing the disease by about 60%-90% compared with individuals who did not receive it,” the document emphasized.
Investments Yield Returns
To achieve the necessary coverage levels, the study estimated that Brazil would need to increase annual per capita investment by $0.28 (about R$1.41) between 2024 and 2050. Brazilian society, in turn, would receive a return of $11 (R$55.27) for every dollar invested.
For South Africa, whose per capita investment increase is estimated at $1.10 per year, the return would be even more significant: $39 for every dollar allocated.
The WHO emphasized that funding for combating the disease is much lower than the value of the damage it causes to nations. “Tuberculosis has high costs for society. Only a small proportion of these costs go directly to the health system (ranging from 1.7% in South Africa to 7.8% in Kenya). Most are costs for patients and society.”
The study projected that between 2024 and 2050, the total cost of tuberculosis to Brazilian society would be $81.2 billion, with an average annual value of $3.01 billion. This figure represents, in 2024, 0.16% of the country’s gross domestic product.
Achieving screening and preventive treatment goals in Brazil would lead to a reduction of as much as 18% in the national incidence of the disease, as well as 1.9 million fewer deaths, between 2024 and 2050.
Although treatable and preventable, tuberculosis remains the leading cause of death from infectious agents worldwide. It is estimated that over 1.3 million people died from the disease in 2022.
The document provides the “health and economic justification for investing in evidence-based interventions recommended by WHO in tuberculosis screening and prevention,” according to WHO Director-General Tedros Adhanom Ghebreyesus, PhD.
“Today we have the knowledge, tools, and political commitment that can end this age-old disease that continues to be one of the leading causes of death from infectious diseases worldwide,” he added.
Emerging Concerns
Although the WHO highlighted the global increase in access to tuberculosis diagnosis and treatment in 2022, which coincided with the recovery of healthcare systems in several countries after the beginning of the pandemic, it emphasized that the implementation of preventive treatment for exposed individuals and high-vulnerability populations remains slow.
Another concern is the increase in drug resistance. Multidrug-resistant tuberculosis is considered a public health crisis. It is estimated that about 410,000 people had multidrug-resistant tuberculosis or rifampicin-resistant tuberculosis in 2022, but only two of every five patients had access to treatment.This story was translated from the Medscape Portuguese edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com .
A new study conducted by the World Health Organization (WHO) suggests that in addition to providing significant improvements in public health, investment in the diagnosis and prevention of tuberculosis also generates economic benefits.
According to a survey conducted by governments and researchers from Brazil, Georgia, Kenya, and South Africa, even modest increases in funding for measures against tuberculosis can bring gains. Every $1 invested produces returns of as much as $39, it found.
The findings may remind governments and policymakers about the importance of investing in public health policies. According to the WHO, the study “provides strong economic arguments” about the true costs of tuberculosis and proves the benefits of increasing funding to accelerate the diagnosis and preventive treatment of the disease.
UN Targets Tuberculosis
In September 2023, during the last meeting of the United Nations General Assembly, following a widespread worsening of disease indicators because of the COVID-19 pandemic, world leaders signed a declaration committing to the expansion of efforts to combat tuberculosis during the next 5 years. The current WHO study was developed to provide a road map for the implementation of key measures against the disease.
The survey highlights two fundamental actions: The expansion of screening, especially in populations considered more vulnerable, and the provision of tuberculosis preventive treatment (TPT), which entails administering drugs to people who have been exposed to the disease but have not yet developed it.
“TPT is a proven and effective intervention to prevent the development of tuberculosis among exposed people, reducing the risk of developing the disease by about 60%-90% compared with individuals who did not receive it,” the document emphasized.
Investments Yield Returns
To achieve the necessary coverage levels, the study estimated that Brazil would need to increase annual per capita investment by $0.28 (about R$1.41) between 2024 and 2050. Brazilian society, in turn, would receive a return of $11 (R$55.27) for every dollar invested.
For South Africa, whose per capita investment increase is estimated at $1.10 per year, the return would be even more significant: $39 for every dollar allocated.
The WHO emphasized that funding for combating the disease is much lower than the value of the damage it causes to nations. “Tuberculosis has high costs for society. Only a small proportion of these costs go directly to the health system (ranging from 1.7% in South Africa to 7.8% in Kenya). Most are costs for patients and society.”
The study projected that between 2024 and 2050, the total cost of tuberculosis to Brazilian society would be $81.2 billion, with an average annual value of $3.01 billion. This figure represents, in 2024, 0.16% of the country’s gross domestic product.
Achieving screening and preventive treatment goals in Brazil would lead to a reduction of as much as 18% in the national incidence of the disease, as well as 1.9 million fewer deaths, between 2024 and 2050.
Although treatable and preventable, tuberculosis remains the leading cause of death from infectious agents worldwide. It is estimated that over 1.3 million people died from the disease in 2022.
The document provides the “health and economic justification for investing in evidence-based interventions recommended by WHO in tuberculosis screening and prevention,” according to WHO Director-General Tedros Adhanom Ghebreyesus, PhD.
“Today we have the knowledge, tools, and political commitment that can end this age-old disease that continues to be one of the leading causes of death from infectious diseases worldwide,” he added.
Emerging Concerns
Although the WHO highlighted the global increase in access to tuberculosis diagnosis and treatment in 2022, which coincided with the recovery of healthcare systems in several countries after the beginning of the pandemic, it emphasized that the implementation of preventive treatment for exposed individuals and high-vulnerability populations remains slow.
Another concern is the increase in drug resistance. Multidrug-resistant tuberculosis is considered a public health crisis. It is estimated that about 410,000 people had multidrug-resistant tuberculosis or rifampicin-resistant tuberculosis in 2022, but only two of every five patients had access to treatment.This story was translated from the Medscape Portuguese edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com .
Bone Infections Increase After S. aureus Bacteremia in Patients With Rheumatoid Arthritis
TOPLINE:
After Staphylococcus aureus bacteremia, patients with rheumatoid arthritis (RA) face nearly double the risk for osteoarticular infections compared with those without RA, with similar mortality risks in both groups.
METHODOLOGY:
- The contraction of S aureus bacteremia is linked to poor clinical outcomes in patients with RA; however, no well-sized studies have evaluated the risk for osteoarticular infections and mortality outcomes in patients with RA following S aureus bacteremia.
- This Danish nationwide cohort study aimed to explore whether the cumulative incidence of osteoarticular infections and death would be higher in patients with RA than in those without RA after contracting S aureus bacteremia.
- The study cohort included 18,274 patients with a first episode of S aureus bacteremia between 2006 and 2018, of whom 367 had been diagnosed with RA before contracting S aureus bacteremia.
- The RA cohort had more women (62%) and a higher median age of participants (73 years) than the non-RA cohort (37% women; median age of participants, 70 years).
TAKEAWAY:
- The 90-day cumulative incidence of osteoarticular infections (septic arthritis, spondylitis, osteomyelitis, psoas muscle abscess, or prosthetic joint infection) was nearly double in patients with RA compared with in those without RA (23.1% vs 12.5%; hazard ratio [HR], 1.93; 95% CI, 1.54-2.41).
- In patients with RA, the risk for osteoarticular infections increased with tumor necrosis factor inhibitor use (HR, 2.27; 95% CI, 1.29-3.98) and orthopedic implants (HR, 1.75; 95% CI, 1.08-2.85).
- Moreover, 90-day all-cause mortality was comparable in the RA (35.4%) and non-RA cohorts (33.9%).
IN PRACTICE:
“Our findings stress the need for vigilance in patients with RA who present with S aureus bacteremia to ensure timely identification and treatment of osteoarticular infections, especially in current TNFi [tumor necrosis factor inhibitor] users and patients with orthopedic implants,” the authors wrote.
SOURCE:
This study, led by Sabine S. Dieperink, MD, of the Centre of Head and Orthopaedics, Copenhagen University Rigshospitalet Glostrup, Denmark, was published online March 9 in Rheumatology (Oxford).
LIMITATIONS:
There might have been chances of misclassification of metastatic S aureus infections owing to the lack of specificity in diagnoses or procedure codes. This study relied on administrative data to record osteoarticular infections, which might have led investigators to underestimate the true cumulative incidence of osteoarticular infections. Also, some patients might have passed away before being diagnosed with osteoarticular infection owing to the high mortality.
DISCLOSURES:
This work was supported by grants from The Danish Rheumatism Association and Beckett Fonden. Some of the authors, including the lead author, declared receiving grants from various funding agencies and other sources, including pharmaceutical companies.
A version of this article appeared on Medscape.com.
TOPLINE:
After Staphylococcus aureus bacteremia, patients with rheumatoid arthritis (RA) face nearly double the risk for osteoarticular infections compared with those without RA, with similar mortality risks in both groups.
METHODOLOGY:
- The contraction of S aureus bacteremia is linked to poor clinical outcomes in patients with RA; however, no well-sized studies have evaluated the risk for osteoarticular infections and mortality outcomes in patients with RA following S aureus bacteremia.
- This Danish nationwide cohort study aimed to explore whether the cumulative incidence of osteoarticular infections and death would be higher in patients with RA than in those without RA after contracting S aureus bacteremia.
- The study cohort included 18,274 patients with a first episode of S aureus bacteremia between 2006 and 2018, of whom 367 had been diagnosed with RA before contracting S aureus bacteremia.
- The RA cohort had more women (62%) and a higher median age of participants (73 years) than the non-RA cohort (37% women; median age of participants, 70 years).
TAKEAWAY:
- The 90-day cumulative incidence of osteoarticular infections (septic arthritis, spondylitis, osteomyelitis, psoas muscle abscess, or prosthetic joint infection) was nearly double in patients with RA compared with in those without RA (23.1% vs 12.5%; hazard ratio [HR], 1.93; 95% CI, 1.54-2.41).
- In patients with RA, the risk for osteoarticular infections increased with tumor necrosis factor inhibitor use (HR, 2.27; 95% CI, 1.29-3.98) and orthopedic implants (HR, 1.75; 95% CI, 1.08-2.85).
- Moreover, 90-day all-cause mortality was comparable in the RA (35.4%) and non-RA cohorts (33.9%).
IN PRACTICE:
“Our findings stress the need for vigilance in patients with RA who present with S aureus bacteremia to ensure timely identification and treatment of osteoarticular infections, especially in current TNFi [tumor necrosis factor inhibitor] users and patients with orthopedic implants,” the authors wrote.
SOURCE:
This study, led by Sabine S. Dieperink, MD, of the Centre of Head and Orthopaedics, Copenhagen University Rigshospitalet Glostrup, Denmark, was published online March 9 in Rheumatology (Oxford).
LIMITATIONS:
There might have been chances of misclassification of metastatic S aureus infections owing to the lack of specificity in diagnoses or procedure codes. This study relied on administrative data to record osteoarticular infections, which might have led investigators to underestimate the true cumulative incidence of osteoarticular infections. Also, some patients might have passed away before being diagnosed with osteoarticular infection owing to the high mortality.
DISCLOSURES:
This work was supported by grants from The Danish Rheumatism Association and Beckett Fonden. Some of the authors, including the lead author, declared receiving grants from various funding agencies and other sources, including pharmaceutical companies.
A version of this article appeared on Medscape.com.
TOPLINE:
After Staphylococcus aureus bacteremia, patients with rheumatoid arthritis (RA) face nearly double the risk for osteoarticular infections compared with those without RA, with similar mortality risks in both groups.
METHODOLOGY:
- The contraction of S aureus bacteremia is linked to poor clinical outcomes in patients with RA; however, no well-sized studies have evaluated the risk for osteoarticular infections and mortality outcomes in patients with RA following S aureus bacteremia.
- This Danish nationwide cohort study aimed to explore whether the cumulative incidence of osteoarticular infections and death would be higher in patients with RA than in those without RA after contracting S aureus bacteremia.
- The study cohort included 18,274 patients with a first episode of S aureus bacteremia between 2006 and 2018, of whom 367 had been diagnosed with RA before contracting S aureus bacteremia.
- The RA cohort had more women (62%) and a higher median age of participants (73 years) than the non-RA cohort (37% women; median age of participants, 70 years).
TAKEAWAY:
- The 90-day cumulative incidence of osteoarticular infections (septic arthritis, spondylitis, osteomyelitis, psoas muscle abscess, or prosthetic joint infection) was nearly double in patients with RA compared with in those without RA (23.1% vs 12.5%; hazard ratio [HR], 1.93; 95% CI, 1.54-2.41).
- In patients with RA, the risk for osteoarticular infections increased with tumor necrosis factor inhibitor use (HR, 2.27; 95% CI, 1.29-3.98) and orthopedic implants (HR, 1.75; 95% CI, 1.08-2.85).
- Moreover, 90-day all-cause mortality was comparable in the RA (35.4%) and non-RA cohorts (33.9%).
IN PRACTICE:
“Our findings stress the need for vigilance in patients with RA who present with S aureus bacteremia to ensure timely identification and treatment of osteoarticular infections, especially in current TNFi [tumor necrosis factor inhibitor] users and patients with orthopedic implants,” the authors wrote.
SOURCE:
This study, led by Sabine S. Dieperink, MD, of the Centre of Head and Orthopaedics, Copenhagen University Rigshospitalet Glostrup, Denmark, was published online March 9 in Rheumatology (Oxford).
LIMITATIONS:
There might have been chances of misclassification of metastatic S aureus infections owing to the lack of specificity in diagnoses or procedure codes. This study relied on administrative data to record osteoarticular infections, which might have led investigators to underestimate the true cumulative incidence of osteoarticular infections. Also, some patients might have passed away before being diagnosed with osteoarticular infection owing to the high mortality.
DISCLOSURES:
This work was supported by grants from The Danish Rheumatism Association and Beckett Fonden. Some of the authors, including the lead author, declared receiving grants from various funding agencies and other sources, including pharmaceutical companies.
A version of this article appeared on Medscape.com.
Study Shows Nirmatrelvir–Ritonavir No More Effective Than Placebo for COVID-19 Symptom Relief
Paxlovid does not significantly alleviate symptoms of COVID-19 compared with placebo among nonhospitalized adults, a new study published April 3 in The New England Journal of Medicine found.
The results suggest that the drug, a combination of nirmatrelvir and ritonavir, may not be particularly helpful for patients who are not at high risk for severe COVID-19. However, although the rate of hospitalization and death from any cause was low overall, the group that received Paxlovid had a reduced rate compared with people in the placebo group, according to the researchers.
“Clearly, the benefit observed among unvaccinated high-risk persons does not extend to those at lower risk for severe COVID-19,” Rajesh T. Gandhi, MD, and Martin Hirsch, MD, of Massachusetts General Hospital in Boston, wrote in an editorial accompanying the journal article. “This result supports guidelines that recommend nirmatrelvir–ritonavir only for persons who are at high risk for disease progression.”
The time from onset to relief of COVID-19 symptoms — including cough, shortness of breath, body aches, and chills — did not differ significantly between the two study groups, the researchers reported. The median time to sustained alleviation of symptoms was 12 days for the Paxlovid group compared with 13 days in the placebo group (P = .60).
However, the phase 2/3 trial found a 57.6% relative reduction in the risk for hospitalizations or death among people who took Paxlovid and were vaccinated but were at high risk for poor outcomes, according to Jennifer Hammond, PhD, head of antiviral development for Pfizer, which makes the drug, and the corresponding author on the study.
Paxlovid has “an increasing body of evidence supporting the strong clinical value of the treatment in preventing hospitalization and death among eligible patients across age groups, vaccination status, and predominant variants,” Dr. Hammond said.
She and her colleagues analyzed data from 1250 adults with symptomatic COVID-19. Participants were fully vaccinated and had a high risk for progression to severe disease or were never vaccinated or had not been in the previous year and had no risk factors for progression to severe disease.
More than half of participants were women, 78.5% were White and 41.4% identified as Hispanic or Latinx. Almost three quarters underwent randomization within 3 days of the start of symptoms, and a little over half had previously received a COVID-19 vaccination. Almost half had one risk factor for severe illness, the most common of these being hypertension (12.3%).
In a subgroup analysis of high-risk participants, hospitalization or death occurred in 0.9% of patients in the Paxlovid group and 2.2% in the placebo group (95% CI, -3.3 to 0.7).
The study’s limitations include that the statistical analysis of COVID-19–related hospitalizations or death from any cause was only descriptive, “because the results for the primary efficacy end point were not significant,” the authors wrote.
Participants who were vaccinated and at high risk were also enrolled regardless of when they had last had a vaccine dose. Furthermore, Paxlovid has a telltale taste, which may have affected the blinding. Finally, the trial was started when the B.1.617.2 (Delta) variant was predominant.
Dr. Gandhi and Dr. Hirsch pointed out that only 5% of participants in the trial were older than 65 years and that other than risk factors such as obesity and smoking, just 2% of people had heart or lung disease.
“As with many medical interventions, there is likely to be a gradient of benefit for nirmatrelvir–ritonavir, with the patients at highest risk for progression most likely to derive the greatest benefit,” Dr. Gandhi and Dr. Hirsch wrote in the editorial. “Thus, it appears reasonable to recommend nirmatrelvir–ritonavir primarily for the treatment of COVID-19 in older patients (particularly those ≥ 65 years of age), those who are immunocompromised, and those who have conditions that substantially increase the risk of severe COVID-19, regardless of previous vaccination or infection status.”
The study was supported by Pfizer.
A version of this article appeared on Medscape.com .
Paxlovid does not significantly alleviate symptoms of COVID-19 compared with placebo among nonhospitalized adults, a new study published April 3 in The New England Journal of Medicine found.
The results suggest that the drug, a combination of nirmatrelvir and ritonavir, may not be particularly helpful for patients who are not at high risk for severe COVID-19. However, although the rate of hospitalization and death from any cause was low overall, the group that received Paxlovid had a reduced rate compared with people in the placebo group, according to the researchers.
“Clearly, the benefit observed among unvaccinated high-risk persons does not extend to those at lower risk for severe COVID-19,” Rajesh T. Gandhi, MD, and Martin Hirsch, MD, of Massachusetts General Hospital in Boston, wrote in an editorial accompanying the journal article. “This result supports guidelines that recommend nirmatrelvir–ritonavir only for persons who are at high risk for disease progression.”
The time from onset to relief of COVID-19 symptoms — including cough, shortness of breath, body aches, and chills — did not differ significantly between the two study groups, the researchers reported. The median time to sustained alleviation of symptoms was 12 days for the Paxlovid group compared with 13 days in the placebo group (P = .60).
However, the phase 2/3 trial found a 57.6% relative reduction in the risk for hospitalizations or death among people who took Paxlovid and were vaccinated but were at high risk for poor outcomes, according to Jennifer Hammond, PhD, head of antiviral development for Pfizer, which makes the drug, and the corresponding author on the study.
Paxlovid has “an increasing body of evidence supporting the strong clinical value of the treatment in preventing hospitalization and death among eligible patients across age groups, vaccination status, and predominant variants,” Dr. Hammond said.
She and her colleagues analyzed data from 1250 adults with symptomatic COVID-19. Participants were fully vaccinated and had a high risk for progression to severe disease or were never vaccinated or had not been in the previous year and had no risk factors for progression to severe disease.
More than half of participants were women, 78.5% were White and 41.4% identified as Hispanic or Latinx. Almost three quarters underwent randomization within 3 days of the start of symptoms, and a little over half had previously received a COVID-19 vaccination. Almost half had one risk factor for severe illness, the most common of these being hypertension (12.3%).
In a subgroup analysis of high-risk participants, hospitalization or death occurred in 0.9% of patients in the Paxlovid group and 2.2% in the placebo group (95% CI, -3.3 to 0.7).
The study’s limitations include that the statistical analysis of COVID-19–related hospitalizations or death from any cause was only descriptive, “because the results for the primary efficacy end point were not significant,” the authors wrote.
Participants who were vaccinated and at high risk were also enrolled regardless of when they had last had a vaccine dose. Furthermore, Paxlovid has a telltale taste, which may have affected the blinding. Finally, the trial was started when the B.1.617.2 (Delta) variant was predominant.
Dr. Gandhi and Dr. Hirsch pointed out that only 5% of participants in the trial were older than 65 years and that other than risk factors such as obesity and smoking, just 2% of people had heart or lung disease.
“As with many medical interventions, there is likely to be a gradient of benefit for nirmatrelvir–ritonavir, with the patients at highest risk for progression most likely to derive the greatest benefit,” Dr. Gandhi and Dr. Hirsch wrote in the editorial. “Thus, it appears reasonable to recommend nirmatrelvir–ritonavir primarily for the treatment of COVID-19 in older patients (particularly those ≥ 65 years of age), those who are immunocompromised, and those who have conditions that substantially increase the risk of severe COVID-19, regardless of previous vaccination or infection status.”
The study was supported by Pfizer.
A version of this article appeared on Medscape.com .
Paxlovid does not significantly alleviate symptoms of COVID-19 compared with placebo among nonhospitalized adults, a new study published April 3 in The New England Journal of Medicine found.
The results suggest that the drug, a combination of nirmatrelvir and ritonavir, may not be particularly helpful for patients who are not at high risk for severe COVID-19. However, although the rate of hospitalization and death from any cause was low overall, the group that received Paxlovid had a reduced rate compared with people in the placebo group, according to the researchers.
“Clearly, the benefit observed among unvaccinated high-risk persons does not extend to those at lower risk for severe COVID-19,” Rajesh T. Gandhi, MD, and Martin Hirsch, MD, of Massachusetts General Hospital in Boston, wrote in an editorial accompanying the journal article. “This result supports guidelines that recommend nirmatrelvir–ritonavir only for persons who are at high risk for disease progression.”
The time from onset to relief of COVID-19 symptoms — including cough, shortness of breath, body aches, and chills — did not differ significantly between the two study groups, the researchers reported. The median time to sustained alleviation of symptoms was 12 days for the Paxlovid group compared with 13 days in the placebo group (P = .60).
However, the phase 2/3 trial found a 57.6% relative reduction in the risk for hospitalizations or death among people who took Paxlovid and were vaccinated but were at high risk for poor outcomes, according to Jennifer Hammond, PhD, head of antiviral development for Pfizer, which makes the drug, and the corresponding author on the study.
Paxlovid has “an increasing body of evidence supporting the strong clinical value of the treatment in preventing hospitalization and death among eligible patients across age groups, vaccination status, and predominant variants,” Dr. Hammond said.
She and her colleagues analyzed data from 1250 adults with symptomatic COVID-19. Participants were fully vaccinated and had a high risk for progression to severe disease or were never vaccinated or had not been in the previous year and had no risk factors for progression to severe disease.
More than half of participants were women, 78.5% were White and 41.4% identified as Hispanic or Latinx. Almost three quarters underwent randomization within 3 days of the start of symptoms, and a little over half had previously received a COVID-19 vaccination. Almost half had one risk factor for severe illness, the most common of these being hypertension (12.3%).
In a subgroup analysis of high-risk participants, hospitalization or death occurred in 0.9% of patients in the Paxlovid group and 2.2% in the placebo group (95% CI, -3.3 to 0.7).
The study’s limitations include that the statistical analysis of COVID-19–related hospitalizations or death from any cause was only descriptive, “because the results for the primary efficacy end point were not significant,” the authors wrote.
Participants who were vaccinated and at high risk were also enrolled regardless of when they had last had a vaccine dose. Furthermore, Paxlovid has a telltale taste, which may have affected the blinding. Finally, the trial was started when the B.1.617.2 (Delta) variant was predominant.
Dr. Gandhi and Dr. Hirsch pointed out that only 5% of participants in the trial were older than 65 years and that other than risk factors such as obesity and smoking, just 2% of people had heart or lung disease.
“As with many medical interventions, there is likely to be a gradient of benefit for nirmatrelvir–ritonavir, with the patients at highest risk for progression most likely to derive the greatest benefit,” Dr. Gandhi and Dr. Hirsch wrote in the editorial. “Thus, it appears reasonable to recommend nirmatrelvir–ritonavir primarily for the treatment of COVID-19 in older patients (particularly those ≥ 65 years of age), those who are immunocompromised, and those who have conditions that substantially increase the risk of severe COVID-19, regardless of previous vaccination or infection status.”
The study was supported by Pfizer.
A version of this article appeared on Medscape.com .
Can VAP be prevented? New data suggest so
Chest Infections and Disaster Response Network
Chest Infections Section
The efficacy of prophylactic antibiotics in the prevention of VAP has been the subject of several studies in recent years. Three large randomized controlled trials, all published since late 2022, have investigated whether antibiotics can prevent VAP and the optimal method of antibiotic administration.
In the AMIKINHAL trial, patients intubated for at least 72 hours in 19 ICUs in France received inhaled amikacin at a dose of 20 mg/kg/day for 3 days.1 Compared with placebo, there was a statistically significant, 7% absolute risk reduction in rate of VAP at 28 days.
In the SUDDICU trial, patients suspected to be intubated for at least 48 hours in 19 ICUs in Australia received a combination of oral paste and gastric suspension containing colistin, tobramycin, and nystatin every 6 hours along with 4 days of intravenous antibiotics.2 There was no difference in the primary outcome of 90-day all-cause mortality; however, there was a statistically significant, 12% reduction in the isolation of antibiotic-resistant organisms in cultures.
In the PROPHY-VAP trial, patients with acute brain injury (Glasgow Coma Scale score [GCS ] ≤12) intubated for at least 48 hours in 9 ICUs in France received a single dose of intravenous ceftriaxone (2 g) within 12 hours of intubation.3 There was an 18% absolute risk reduction in VAP from days 2 to 7 post-ventilation.
These trials, involving distinct patient populations and interventions, indicate that antibiotic prophylaxis may reduce VAP risk under specific circumstances, but its effect on overall outcomes is still uncertain. The understanding of prophylactic antibiotics in VAP prevention is rapidly evolving.
References
1. Ehrmann S, et al. N Engl J Med. 2023;389(22):2052-2062.
2. Myburgh JA, et al. JAMA. 2022;328(19):1911-1921.
3. Dahyot-Fizelier C, et al. Lancet Respir Med. 2024;S2213-2600(23):00471-X.
Chest Infections and Disaster Response Network
Chest Infections Section
The efficacy of prophylactic antibiotics in the prevention of VAP has been the subject of several studies in recent years. Three large randomized controlled trials, all published since late 2022, have investigated whether antibiotics can prevent VAP and the optimal method of antibiotic administration.
In the AMIKINHAL trial, patients intubated for at least 72 hours in 19 ICUs in France received inhaled amikacin at a dose of 20 mg/kg/day for 3 days.1 Compared with placebo, there was a statistically significant, 7% absolute risk reduction in rate of VAP at 28 days.
In the SUDDICU trial, patients suspected to be intubated for at least 48 hours in 19 ICUs in Australia received a combination of oral paste and gastric suspension containing colistin, tobramycin, and nystatin every 6 hours along with 4 days of intravenous antibiotics.2 There was no difference in the primary outcome of 90-day all-cause mortality; however, there was a statistically significant, 12% reduction in the isolation of antibiotic-resistant organisms in cultures.
In the PROPHY-VAP trial, patients with acute brain injury (Glasgow Coma Scale score [GCS ] ≤12) intubated for at least 48 hours in 9 ICUs in France received a single dose of intravenous ceftriaxone (2 g) within 12 hours of intubation.3 There was an 18% absolute risk reduction in VAP from days 2 to 7 post-ventilation.
These trials, involving distinct patient populations and interventions, indicate that antibiotic prophylaxis may reduce VAP risk under specific circumstances, but its effect on overall outcomes is still uncertain. The understanding of prophylactic antibiotics in VAP prevention is rapidly evolving.
References
1. Ehrmann S, et al. N Engl J Med. 2023;389(22):2052-2062.
2. Myburgh JA, et al. JAMA. 2022;328(19):1911-1921.
3. Dahyot-Fizelier C, et al. Lancet Respir Med. 2024;S2213-2600(23):00471-X.
Chest Infections and Disaster Response Network
Chest Infections Section
The efficacy of prophylactic antibiotics in the prevention of VAP has been the subject of several studies in recent years. Three large randomized controlled trials, all published since late 2022, have investigated whether antibiotics can prevent VAP and the optimal method of antibiotic administration.
In the AMIKINHAL trial, patients intubated for at least 72 hours in 19 ICUs in France received inhaled amikacin at a dose of 20 mg/kg/day for 3 days.1 Compared with placebo, there was a statistically significant, 7% absolute risk reduction in rate of VAP at 28 days.
In the SUDDICU trial, patients suspected to be intubated for at least 48 hours in 19 ICUs in Australia received a combination of oral paste and gastric suspension containing colistin, tobramycin, and nystatin every 6 hours along with 4 days of intravenous antibiotics.2 There was no difference in the primary outcome of 90-day all-cause mortality; however, there was a statistically significant, 12% reduction in the isolation of antibiotic-resistant organisms in cultures.
In the PROPHY-VAP trial, patients with acute brain injury (Glasgow Coma Scale score [GCS ] ≤12) intubated for at least 48 hours in 9 ICUs in France received a single dose of intravenous ceftriaxone (2 g) within 12 hours of intubation.3 There was an 18% absolute risk reduction in VAP from days 2 to 7 post-ventilation.
These trials, involving distinct patient populations and interventions, indicate that antibiotic prophylaxis may reduce VAP risk under specific circumstances, but its effect on overall outcomes is still uncertain. The understanding of prophylactic antibiotics in VAP prevention is rapidly evolving.
References
1. Ehrmann S, et al. N Engl J Med. 2023;389(22):2052-2062.
2. Myburgh JA, et al. JAMA. 2022;328(19):1911-1921.
3. Dahyot-Fizelier C, et al. Lancet Respir Med. 2024;S2213-2600(23):00471-X.





