User login
Topical Ruxolitinib Effective for AD in Study of Children Ages 2-11 years
) affecting ≥ 35% or more of their body surface area (BSA), results from a small open-label maximum-use trial showed.
When approved for this age group, ruxolitinib cream will provide a topical nonsteroidal option for patients aged 2-11, which will “simplify the treatment regimen,” one of the study investigators, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said in an interview after the Revolutionizing Atopic Dermatitis conference, where the study was presented during a late-breaking abstract session.
A topical formulation of the selective Janus kinase (JAK) 1/JAK2 inhibitor, ruxolitinib cream 1.5% is currently approved by the Food and Drug Administration for the short-term and noncontinuous chronic treatment of mild to moderate AD in non-immunocompromised adult and pediatric patients aged 12 years and older, whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable.
In previous reports of this trial in children aged 2-11 years with ≥ 35% affected BSA, ruxolitinib cream 1.5% was generally well tolerated, with rapid anti-inflammatory and antipruritic effects and improvements in patient-reported outcomes observed with ≤ 4 weeks of continuous treatment and maintained with as-needed treatment from 4 to 8 weeks.
For the current trial, investigators evaluated data on tolerability, safety, systemic exposure, and clinical and patient-reported outcomes through 52 weeks to determine whether clinical benefits and tolerability observed through 8 weeks were sustained.
Dr. Stein Gold and colleagues reported results from 29 children who received ruxolitinib cream 1.5% from baseline through week 8. Of these, 22 continued into the long-term safety period from week 8 through 52. From baseline through week 8, patients applied a mean of 6.5 g per day of ruxolitinib cream; this dropped to a mean of 3.2 g per day from weeks 8 through 52. The mean steady-state plasma concentration of ruxolitinib throughout the study was 98.2 nM, which is “well below half-maximal concentration of JAK-mediated myelosuppression in adults (281 nM),” the researchers stated in their abstract.
No treatment-related interruptions, discontinuations, or serious adverse events were observed between baseline and week 52. One patient (3.4%) had two treatment-related application site reactions (paresthesia and folliculitis). At weeks 4 and 52, 53.8% of patients achieved treatment success, which was defined as an Investigator Global Assessment of 0/1 with a ≥ 2-grade improvement from baseline. The mean affected BSA decreased from 58.0% at baseline to 11.4% at week 4 and continued to decrease to 2.2% through week 52. “I was surprised that patients could maintain control over the long-term using the medication as needed,” Dr. Stein Gold told this news organization. “I was also pleased to see that there was low systemic exposure even when used on large body surface areas.”
In other findings, the mean total Patient Oriented Eczema Measure score dropped from a baseline of 19.4 to a mean of 4.5 at week 8 and 3.6 at week 52 and the mean total Children’s Dermatology Life Quality Index score fell from a baseline of 15.4 to a mean of 5.3 at week 8 and a mean of 2.1 at week 52. Meanwhile, the mean total Infants’ Dermatology Quality of Life Index score fell from a mean of 12.3 at baseline to a mean of 2.8 at week 8 and a mean of 0.7 at week 52.
Dr. Stein Gold noted certain limitations of the study, including the fact that it did not study children aged younger than 2 years.
The study was funded by Incyte, which markets ruxolitinib cream 1.5% as Opzelura. Dr. Stein Gold disclosed that she has served as an investigator, advisor, and/or speaker for several pharmaceutical companies, including Incyte.
A version of this article appeared on Medscape.com.
) affecting ≥ 35% or more of their body surface area (BSA), results from a small open-label maximum-use trial showed.
When approved for this age group, ruxolitinib cream will provide a topical nonsteroidal option for patients aged 2-11, which will “simplify the treatment regimen,” one of the study investigators, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said in an interview after the Revolutionizing Atopic Dermatitis conference, where the study was presented during a late-breaking abstract session.
A topical formulation of the selective Janus kinase (JAK) 1/JAK2 inhibitor, ruxolitinib cream 1.5% is currently approved by the Food and Drug Administration for the short-term and noncontinuous chronic treatment of mild to moderate AD in non-immunocompromised adult and pediatric patients aged 12 years and older, whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable.
In previous reports of this trial in children aged 2-11 years with ≥ 35% affected BSA, ruxolitinib cream 1.5% was generally well tolerated, with rapid anti-inflammatory and antipruritic effects and improvements in patient-reported outcomes observed with ≤ 4 weeks of continuous treatment and maintained with as-needed treatment from 4 to 8 weeks.
For the current trial, investigators evaluated data on tolerability, safety, systemic exposure, and clinical and patient-reported outcomes through 52 weeks to determine whether clinical benefits and tolerability observed through 8 weeks were sustained.
Dr. Stein Gold and colleagues reported results from 29 children who received ruxolitinib cream 1.5% from baseline through week 8. Of these, 22 continued into the long-term safety period from week 8 through 52. From baseline through week 8, patients applied a mean of 6.5 g per day of ruxolitinib cream; this dropped to a mean of 3.2 g per day from weeks 8 through 52. The mean steady-state plasma concentration of ruxolitinib throughout the study was 98.2 nM, which is “well below half-maximal concentration of JAK-mediated myelosuppression in adults (281 nM),” the researchers stated in their abstract.
No treatment-related interruptions, discontinuations, or serious adverse events were observed between baseline and week 52. One patient (3.4%) had two treatment-related application site reactions (paresthesia and folliculitis). At weeks 4 and 52, 53.8% of patients achieved treatment success, which was defined as an Investigator Global Assessment of 0/1 with a ≥ 2-grade improvement from baseline. The mean affected BSA decreased from 58.0% at baseline to 11.4% at week 4 and continued to decrease to 2.2% through week 52. “I was surprised that patients could maintain control over the long-term using the medication as needed,” Dr. Stein Gold told this news organization. “I was also pleased to see that there was low systemic exposure even when used on large body surface areas.”
In other findings, the mean total Patient Oriented Eczema Measure score dropped from a baseline of 19.4 to a mean of 4.5 at week 8 and 3.6 at week 52 and the mean total Children’s Dermatology Life Quality Index score fell from a baseline of 15.4 to a mean of 5.3 at week 8 and a mean of 2.1 at week 52. Meanwhile, the mean total Infants’ Dermatology Quality of Life Index score fell from a mean of 12.3 at baseline to a mean of 2.8 at week 8 and a mean of 0.7 at week 52.
Dr. Stein Gold noted certain limitations of the study, including the fact that it did not study children aged younger than 2 years.
The study was funded by Incyte, which markets ruxolitinib cream 1.5% as Opzelura. Dr. Stein Gold disclosed that she has served as an investigator, advisor, and/or speaker for several pharmaceutical companies, including Incyte.
A version of this article appeared on Medscape.com.
) affecting ≥ 35% or more of their body surface area (BSA), results from a small open-label maximum-use trial showed.
When approved for this age group, ruxolitinib cream will provide a topical nonsteroidal option for patients aged 2-11, which will “simplify the treatment regimen,” one of the study investigators, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said in an interview after the Revolutionizing Atopic Dermatitis conference, where the study was presented during a late-breaking abstract session.
A topical formulation of the selective Janus kinase (JAK) 1/JAK2 inhibitor, ruxolitinib cream 1.5% is currently approved by the Food and Drug Administration for the short-term and noncontinuous chronic treatment of mild to moderate AD in non-immunocompromised adult and pediatric patients aged 12 years and older, whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable.
In previous reports of this trial in children aged 2-11 years with ≥ 35% affected BSA, ruxolitinib cream 1.5% was generally well tolerated, with rapid anti-inflammatory and antipruritic effects and improvements in patient-reported outcomes observed with ≤ 4 weeks of continuous treatment and maintained with as-needed treatment from 4 to 8 weeks.
For the current trial, investigators evaluated data on tolerability, safety, systemic exposure, and clinical and patient-reported outcomes through 52 weeks to determine whether clinical benefits and tolerability observed through 8 weeks were sustained.
Dr. Stein Gold and colleagues reported results from 29 children who received ruxolitinib cream 1.5% from baseline through week 8. Of these, 22 continued into the long-term safety period from week 8 through 52. From baseline through week 8, patients applied a mean of 6.5 g per day of ruxolitinib cream; this dropped to a mean of 3.2 g per day from weeks 8 through 52. The mean steady-state plasma concentration of ruxolitinib throughout the study was 98.2 nM, which is “well below half-maximal concentration of JAK-mediated myelosuppression in adults (281 nM),” the researchers stated in their abstract.
No treatment-related interruptions, discontinuations, or serious adverse events were observed between baseline and week 52. One patient (3.4%) had two treatment-related application site reactions (paresthesia and folliculitis). At weeks 4 and 52, 53.8% of patients achieved treatment success, which was defined as an Investigator Global Assessment of 0/1 with a ≥ 2-grade improvement from baseline. The mean affected BSA decreased from 58.0% at baseline to 11.4% at week 4 and continued to decrease to 2.2% through week 52. “I was surprised that patients could maintain control over the long-term using the medication as needed,” Dr. Stein Gold told this news organization. “I was also pleased to see that there was low systemic exposure even when used on large body surface areas.”
In other findings, the mean total Patient Oriented Eczema Measure score dropped from a baseline of 19.4 to a mean of 4.5 at week 8 and 3.6 at week 52 and the mean total Children’s Dermatology Life Quality Index score fell from a baseline of 15.4 to a mean of 5.3 at week 8 and a mean of 2.1 at week 52. Meanwhile, the mean total Infants’ Dermatology Quality of Life Index score fell from a mean of 12.3 at baseline to a mean of 2.8 at week 8 and a mean of 0.7 at week 52.
Dr. Stein Gold noted certain limitations of the study, including the fact that it did not study children aged younger than 2 years.
The study was funded by Incyte, which markets ruxolitinib cream 1.5% as Opzelura. Dr. Stein Gold disclosed that she has served as an investigator, advisor, and/or speaker for several pharmaceutical companies, including Incyte.
A version of this article appeared on Medscape.com.
Pediatric Atopic Dermatitis: Study Suggests Treatment May Impact Atopic March
TOPLINE:
METHODOLOGY:
- Researchers conducted a retrospective cohort study using data from the US Collaborative Network, focusing on pediatric patients aged 18 years and younger with two AD diagnoses at least 30 days apart.
- Patients were divided into two cohorts: Those treated with dupilumab (n = 2192) and those who received conventional therapies (n = 2192), including systemic corticosteroids or conventional immunomodulators. They were stratified into three age groups: Preschoolers (< 6 years), school-aged children (6 to < 12 years), and adolescents (12-18 years).
- Both cohorts underwent 1:1 propensity score matching based on current age, age at index (first prescription of dupilumab or conventional therapy), sex, race, comorbidities, laboratory measurements, and prior medications. The primary outcome was atopic march progression, defined by incident asthma or allergic rhinitis.
TAKEAWAY:
- Over 3 years, the dupilumab-treated cohort had a significantly lower cumulative incidence of atopic march progression (20.09% vs 27.22%; P < .001), asthma (9.43% vs 14.64%; P = .001), and allergic rhinitis (13.57% vs 20.52%; P = .003) than the conventional therapy cohort.
- The risk for atopic march progression, asthma, and allergic rhinitis was also significantly reduced by 32%, 40%, and 31%, respectively, in the dupilumab vs conventional therapy cohort.
- Age-specific analyses found that the protective effect of dupilumab against allergic rhinitis was the most pronounced in adolescents (hazard ratio [HR], 0.503; 95% CI, 0.322-0.784), followed by school-aged children (HR, 0.577; 95% CI, 0.399-0.834), and preschoolers (HR, 0.623; 95% CI, 0.412-0.942).
- However, dupilumab was associated with reduced risk for asthma only in preschoolers (HR, 0.427; 95% CI, 0.247-0.738) and not in school-aged children or adolescents.
IN PRACTICE:
“Dupilumab in AD not only treats the disease but may influence atopic march mechanisms, suggesting its role as a disease-modifying atopic march drug,” the authors wrote, adding that more research “with extended follow-up and proof-of-concept is warranted.”
SOURCE:
The study was led by Teng-Li Lin, MD, Department of Dermatology, Dalin Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, Chiayi, Taiwan, and was published online on June 13, 2024, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The observational nature of the study limited the ability to infer direct causality between dupilumab use and reduced atopic march risk. Lack of detailed information on AD severity, total dosage, and duration of medication treatment may affect the interpretation of the study’s findings. The demographic data suggest that the dupilumab cohort had more severe AD, so the observed risk reduction may be greater than that reported in this study.
DISCLOSURES:
The study was supported in part by the National Science and Technology Council, Taiwan, and Taichung Veterans General Hospital. The authors had no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com .
TOPLINE:
METHODOLOGY:
- Researchers conducted a retrospective cohort study using data from the US Collaborative Network, focusing on pediatric patients aged 18 years and younger with two AD diagnoses at least 30 days apart.
- Patients were divided into two cohorts: Those treated with dupilumab (n = 2192) and those who received conventional therapies (n = 2192), including systemic corticosteroids or conventional immunomodulators. They were stratified into three age groups: Preschoolers (< 6 years), school-aged children (6 to < 12 years), and adolescents (12-18 years).
- Both cohorts underwent 1:1 propensity score matching based on current age, age at index (first prescription of dupilumab or conventional therapy), sex, race, comorbidities, laboratory measurements, and prior medications. The primary outcome was atopic march progression, defined by incident asthma or allergic rhinitis.
TAKEAWAY:
- Over 3 years, the dupilumab-treated cohort had a significantly lower cumulative incidence of atopic march progression (20.09% vs 27.22%; P < .001), asthma (9.43% vs 14.64%; P = .001), and allergic rhinitis (13.57% vs 20.52%; P = .003) than the conventional therapy cohort.
- The risk for atopic march progression, asthma, and allergic rhinitis was also significantly reduced by 32%, 40%, and 31%, respectively, in the dupilumab vs conventional therapy cohort.
- Age-specific analyses found that the protective effect of dupilumab against allergic rhinitis was the most pronounced in adolescents (hazard ratio [HR], 0.503; 95% CI, 0.322-0.784), followed by school-aged children (HR, 0.577; 95% CI, 0.399-0.834), and preschoolers (HR, 0.623; 95% CI, 0.412-0.942).
- However, dupilumab was associated with reduced risk for asthma only in preschoolers (HR, 0.427; 95% CI, 0.247-0.738) and not in school-aged children or adolescents.
IN PRACTICE:
“Dupilumab in AD not only treats the disease but may influence atopic march mechanisms, suggesting its role as a disease-modifying atopic march drug,” the authors wrote, adding that more research “with extended follow-up and proof-of-concept is warranted.”
SOURCE:
The study was led by Teng-Li Lin, MD, Department of Dermatology, Dalin Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, Chiayi, Taiwan, and was published online on June 13, 2024, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The observational nature of the study limited the ability to infer direct causality between dupilumab use and reduced atopic march risk. Lack of detailed information on AD severity, total dosage, and duration of medication treatment may affect the interpretation of the study’s findings. The demographic data suggest that the dupilumab cohort had more severe AD, so the observed risk reduction may be greater than that reported in this study.
DISCLOSURES:
The study was supported in part by the National Science and Technology Council, Taiwan, and Taichung Veterans General Hospital. The authors had no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com .
TOPLINE:
METHODOLOGY:
- Researchers conducted a retrospective cohort study using data from the US Collaborative Network, focusing on pediatric patients aged 18 years and younger with two AD diagnoses at least 30 days apart.
- Patients were divided into two cohorts: Those treated with dupilumab (n = 2192) and those who received conventional therapies (n = 2192), including systemic corticosteroids or conventional immunomodulators. They were stratified into three age groups: Preschoolers (< 6 years), school-aged children (6 to < 12 years), and adolescents (12-18 years).
- Both cohorts underwent 1:1 propensity score matching based on current age, age at index (first prescription of dupilumab or conventional therapy), sex, race, comorbidities, laboratory measurements, and prior medications. The primary outcome was atopic march progression, defined by incident asthma or allergic rhinitis.
TAKEAWAY:
- Over 3 years, the dupilumab-treated cohort had a significantly lower cumulative incidence of atopic march progression (20.09% vs 27.22%; P < .001), asthma (9.43% vs 14.64%; P = .001), and allergic rhinitis (13.57% vs 20.52%; P = .003) than the conventional therapy cohort.
- The risk for atopic march progression, asthma, and allergic rhinitis was also significantly reduced by 32%, 40%, and 31%, respectively, in the dupilumab vs conventional therapy cohort.
- Age-specific analyses found that the protective effect of dupilumab against allergic rhinitis was the most pronounced in adolescents (hazard ratio [HR], 0.503; 95% CI, 0.322-0.784), followed by school-aged children (HR, 0.577; 95% CI, 0.399-0.834), and preschoolers (HR, 0.623; 95% CI, 0.412-0.942).
- However, dupilumab was associated with reduced risk for asthma only in preschoolers (HR, 0.427; 95% CI, 0.247-0.738) and not in school-aged children or adolescents.
IN PRACTICE:
“Dupilumab in AD not only treats the disease but may influence atopic march mechanisms, suggesting its role as a disease-modifying atopic march drug,” the authors wrote, adding that more research “with extended follow-up and proof-of-concept is warranted.”
SOURCE:
The study was led by Teng-Li Lin, MD, Department of Dermatology, Dalin Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, Chiayi, Taiwan, and was published online on June 13, 2024, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The observational nature of the study limited the ability to infer direct causality between dupilumab use and reduced atopic march risk. Lack of detailed information on AD severity, total dosage, and duration of medication treatment may affect the interpretation of the study’s findings. The demographic data suggest that the dupilumab cohort had more severe AD, so the observed risk reduction may be greater than that reported in this study.
DISCLOSURES:
The study was supported in part by the National Science and Technology Council, Taiwan, and Taichung Veterans General Hospital. The authors had no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com .
Meta-Analysis Finds Combination Cream Plus Tranexamic Acid Effective for Melasma
TOPLINE:
A meta-analysis showed that .
METHODOLOGY:
- Current treatments for melasma focus on inducing remission and preventing relapse. Tranexamic acid, an antifibrinolytic drug, has shown promise in recent studies, but its optimal use, either alone or as an adjunct to TCC, remains unclear.
- Researchers conducted a meta-analysis of four randomized controlled trials patients that compared oral tranexamic acid plus TCC (hydroquinone, retinoic acid, and hydrocortisone) and TCC alone in 480 patients with melasma, divided almost evenly into the two treatment groups.
- The main outcome was the change in the Melasma Severity Area Index (MASI) score and recurrence rate from baseline.
TAKEAWAY:
- Patients treated with oral tranexamic acid plus TCC showed a greater reduction in MASI scores compared with those who received TCC alone (mean difference, −3.10; P = .03).
- The recurrence rate of melasma was significantly lower in the tranexamic acid plus TCC group (risk ratio [RR], 0.28; P < .001).
- There was no significant difference in the incidences of erythema (RR, 0.63; P = .147) and burning (RR, 0.59; P = .131).
IN PRACTICE:
“Evidence indicates that oral tranexamic acid confers clinical benefits, contributing to the enhancement of treatment outcomes in melasma when used in conjunction with TCC therapy,” and results are promising with regards to minimizing recurrence, the authors concluded.
SOURCE:
The study was led by Ocílio Ribeiro Gonçalves, MS, of the Federal University of Piauí, Teresina, Brazil, and was published online on June 8, 2024, in Clinical and Experimental Dermatology.
LIMITATIONS:
There was heterogeneity across studies, including different methods of administration, treatment protocols (including dosage), and timing of treatment.
DISCLOSURES:
The study reported receiving no funding. The authors declared no conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
A meta-analysis showed that .
METHODOLOGY:
- Current treatments for melasma focus on inducing remission and preventing relapse. Tranexamic acid, an antifibrinolytic drug, has shown promise in recent studies, but its optimal use, either alone or as an adjunct to TCC, remains unclear.
- Researchers conducted a meta-analysis of four randomized controlled trials patients that compared oral tranexamic acid plus TCC (hydroquinone, retinoic acid, and hydrocortisone) and TCC alone in 480 patients with melasma, divided almost evenly into the two treatment groups.
- The main outcome was the change in the Melasma Severity Area Index (MASI) score and recurrence rate from baseline.
TAKEAWAY:
- Patients treated with oral tranexamic acid plus TCC showed a greater reduction in MASI scores compared with those who received TCC alone (mean difference, −3.10; P = .03).
- The recurrence rate of melasma was significantly lower in the tranexamic acid plus TCC group (risk ratio [RR], 0.28; P < .001).
- There was no significant difference in the incidences of erythema (RR, 0.63; P = .147) and burning (RR, 0.59; P = .131).
IN PRACTICE:
“Evidence indicates that oral tranexamic acid confers clinical benefits, contributing to the enhancement of treatment outcomes in melasma when used in conjunction with TCC therapy,” and results are promising with regards to minimizing recurrence, the authors concluded.
SOURCE:
The study was led by Ocílio Ribeiro Gonçalves, MS, of the Federal University of Piauí, Teresina, Brazil, and was published online on June 8, 2024, in Clinical and Experimental Dermatology.
LIMITATIONS:
There was heterogeneity across studies, including different methods of administration, treatment protocols (including dosage), and timing of treatment.
DISCLOSURES:
The study reported receiving no funding. The authors declared no conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
A meta-analysis showed that .
METHODOLOGY:
- Current treatments for melasma focus on inducing remission and preventing relapse. Tranexamic acid, an antifibrinolytic drug, has shown promise in recent studies, but its optimal use, either alone or as an adjunct to TCC, remains unclear.
- Researchers conducted a meta-analysis of four randomized controlled trials patients that compared oral tranexamic acid plus TCC (hydroquinone, retinoic acid, and hydrocortisone) and TCC alone in 480 patients with melasma, divided almost evenly into the two treatment groups.
- The main outcome was the change in the Melasma Severity Area Index (MASI) score and recurrence rate from baseline.
TAKEAWAY:
- Patients treated with oral tranexamic acid plus TCC showed a greater reduction in MASI scores compared with those who received TCC alone (mean difference, −3.10; P = .03).
- The recurrence rate of melasma was significantly lower in the tranexamic acid plus TCC group (risk ratio [RR], 0.28; P < .001).
- There was no significant difference in the incidences of erythema (RR, 0.63; P = .147) and burning (RR, 0.59; P = .131).
IN PRACTICE:
“Evidence indicates that oral tranexamic acid confers clinical benefits, contributing to the enhancement of treatment outcomes in melasma when used in conjunction with TCC therapy,” and results are promising with regards to minimizing recurrence, the authors concluded.
SOURCE:
The study was led by Ocílio Ribeiro Gonçalves, MS, of the Federal University of Piauí, Teresina, Brazil, and was published online on June 8, 2024, in Clinical and Experimental Dermatology.
LIMITATIONS:
There was heterogeneity across studies, including different methods of administration, treatment protocols (including dosage), and timing of treatment.
DISCLOSURES:
The study reported receiving no funding. The authors declared no conflicts of interest.
A version of this article appeared on Medscape.com.
OTC Supplement Linked to Hyperpigmentation
CHICAGO —The .
“This is something we will see more and more,” Heather Woolery-Lloyd, MD, director of the Skin of Color Division at the University of Miami Department of Dermatology, said at the Pigmentary Disorders Exchange Symposium. The key marker of this hyperpigmentation, she said, is that “it’s strongly photoaccentuated,” affecting areas exposed to the sun — but it also tends to spare the knuckles on patients’ hands.
Used Like an Opioid, But It’s Not Regulated
Kratom is a plant common in southeast Asia and is used as an analgesic. It’s marketed as a “legal opioid” or “legal high” and is sold in 2- or 3-ounce containers of extract or sold as a powder, Dr. Woolery-Lloyd said. The leaves may be boiled into a tea, smoked, chewed, or put into capsules, according to a case report published in February in the Journal of Integrative Dermatology. It is used worldwide and is not regulated in the United States.
“Many of our patients think kratom is a safe, herbal supplement” but often don’t know it can have several side effects and can be addictive, Dr. Woolery-Lloyd said. Its popularity is increasing as reflected by the number of posts related to kratom on social media platforms.
In the February case report, Shaina Patel, BA, and Nathaniel Phelan, MD, from Kansas City University, Kansas City, Missouri, wrote that side effects of kratom include drowsiness, tachycardia, vomiting, respiratory depression, and cardiac arrest, in addition to confusion and hallucinations.
Kratom also has many different effects on the psyche, Dr. Woolery-Lloyd said at the meeting. At low doses, it blocks the reuptake of norepinephrine, serotonin, and dopamine, producing a motivational effect, and at high doses, it creates an analgesic, calming effect. And people who chronically consume high doses of kratom may be susceptible to hyperpigmentation.
Kratom-associated hyperpigmentation should be considered as a diagnosis when evaluating patients for other drug-associated pigmentary disorders, “especially if pigment is photodistributed,” she said. “If you see new-onset hyperpigmentation or onset over several months and it’s very photoaccentuated, definitely ask about use of kratom.”
Case Reports Show Patterns of Presentation
A 2022 report from Landon R. Powell, BS, with the department of biology, Whitworth University in Spokane, Washington, and coauthors, published in JAAD Case Reports, noted that kratom use in the United States has increased dramatically. “As measured by call reports to the United States National Poison Data System, in 2011, there were 11 reported kratom exposures, and in the first 7 months of 2018, there were 357 reported exposures,” they wrote.
An estimated 1.7 million Americans aged ≥ 12 years said they had used kratom in the previous year, according to the Substance Abuse and Mental Health Services Administration 2021 National Survey on Drug Use and Health.
In the case report, Mr. Powell and coauthors described a 54-year-old White male patient who had been using kratom for the previous four to five years to reduce opioid use. During this period, he consumed kratom powder mixed with orange juice three to four times a day. He presented with “diffuse hyperpigmented patches on his arms and face in a photodistributed manner, with notable sparing of the knuckles on both hands.”
Dark Gray-Blue Skin
In the more recent case report, Ms. Patel and Dr. Phelan described a 30-year-old White male patient who presented with dark gray-blue skin coloring on his cheeks, back of his neck, and the backs of his hands and forearms. He had no other medical conditions and did not take any medications or supplements that cause hyperpigmentation while using kratom.
The patient had been taking kratom for years in the wake of an opioid addiction following medications for a high school injury. He developed an opioid use disorder and tried to replace his pain medications with kratom.
“The patient stopped using kratom in May 2022, but the discoloration remains. It has not regressed in the following 16 months after discontinuing kratom use,” the authors wrote, noting that “whether or not the hyperpigmentation is able to regress is unknown.”
Dr. Woolery-Lloyd is a consultant for AbbVie, Incyte, Johnson & Johnson Consumer, LivDerm, and L’Oreal; a speaker for Eli Lilly, Incyte, L’Oreal, and Ortho Dermatologics; and a researcher/investigator for AbbVie, Allergan, Eirion Therapeutics, Galderma, Pfizer, Sanofi, and Vyne Therapeutics.
According to an information page on kratom on the Food and Drug Administration website, health care professionals and consumers can report adverse reactions associated with kratom to the FDA’s MedWatch program.
A version of this article appeared on Medscape.com.
CHICAGO —The .
“This is something we will see more and more,” Heather Woolery-Lloyd, MD, director of the Skin of Color Division at the University of Miami Department of Dermatology, said at the Pigmentary Disorders Exchange Symposium. The key marker of this hyperpigmentation, she said, is that “it’s strongly photoaccentuated,” affecting areas exposed to the sun — but it also tends to spare the knuckles on patients’ hands.
Used Like an Opioid, But It’s Not Regulated
Kratom is a plant common in southeast Asia and is used as an analgesic. It’s marketed as a “legal opioid” or “legal high” and is sold in 2- or 3-ounce containers of extract or sold as a powder, Dr. Woolery-Lloyd said. The leaves may be boiled into a tea, smoked, chewed, or put into capsules, according to a case report published in February in the Journal of Integrative Dermatology. It is used worldwide and is not regulated in the United States.
“Many of our patients think kratom is a safe, herbal supplement” but often don’t know it can have several side effects and can be addictive, Dr. Woolery-Lloyd said. Its popularity is increasing as reflected by the number of posts related to kratom on social media platforms.
In the February case report, Shaina Patel, BA, and Nathaniel Phelan, MD, from Kansas City University, Kansas City, Missouri, wrote that side effects of kratom include drowsiness, tachycardia, vomiting, respiratory depression, and cardiac arrest, in addition to confusion and hallucinations.
Kratom also has many different effects on the psyche, Dr. Woolery-Lloyd said at the meeting. At low doses, it blocks the reuptake of norepinephrine, serotonin, and dopamine, producing a motivational effect, and at high doses, it creates an analgesic, calming effect. And people who chronically consume high doses of kratom may be susceptible to hyperpigmentation.
Kratom-associated hyperpigmentation should be considered as a diagnosis when evaluating patients for other drug-associated pigmentary disorders, “especially if pigment is photodistributed,” she said. “If you see new-onset hyperpigmentation or onset over several months and it’s very photoaccentuated, definitely ask about use of kratom.”
Case Reports Show Patterns of Presentation
A 2022 report from Landon R. Powell, BS, with the department of biology, Whitworth University in Spokane, Washington, and coauthors, published in JAAD Case Reports, noted that kratom use in the United States has increased dramatically. “As measured by call reports to the United States National Poison Data System, in 2011, there were 11 reported kratom exposures, and in the first 7 months of 2018, there were 357 reported exposures,” they wrote.
An estimated 1.7 million Americans aged ≥ 12 years said they had used kratom in the previous year, according to the Substance Abuse and Mental Health Services Administration 2021 National Survey on Drug Use and Health.
In the case report, Mr. Powell and coauthors described a 54-year-old White male patient who had been using kratom for the previous four to five years to reduce opioid use. During this period, he consumed kratom powder mixed with orange juice three to four times a day. He presented with “diffuse hyperpigmented patches on his arms and face in a photodistributed manner, with notable sparing of the knuckles on both hands.”
Dark Gray-Blue Skin
In the more recent case report, Ms. Patel and Dr. Phelan described a 30-year-old White male patient who presented with dark gray-blue skin coloring on his cheeks, back of his neck, and the backs of his hands and forearms. He had no other medical conditions and did not take any medications or supplements that cause hyperpigmentation while using kratom.
The patient had been taking kratom for years in the wake of an opioid addiction following medications for a high school injury. He developed an opioid use disorder and tried to replace his pain medications with kratom.
“The patient stopped using kratom in May 2022, but the discoloration remains. It has not regressed in the following 16 months after discontinuing kratom use,” the authors wrote, noting that “whether or not the hyperpigmentation is able to regress is unknown.”
Dr. Woolery-Lloyd is a consultant for AbbVie, Incyte, Johnson & Johnson Consumer, LivDerm, and L’Oreal; a speaker for Eli Lilly, Incyte, L’Oreal, and Ortho Dermatologics; and a researcher/investigator for AbbVie, Allergan, Eirion Therapeutics, Galderma, Pfizer, Sanofi, and Vyne Therapeutics.
According to an information page on kratom on the Food and Drug Administration website, health care professionals and consumers can report adverse reactions associated with kratom to the FDA’s MedWatch program.
A version of this article appeared on Medscape.com.
CHICAGO —The .
“This is something we will see more and more,” Heather Woolery-Lloyd, MD, director of the Skin of Color Division at the University of Miami Department of Dermatology, said at the Pigmentary Disorders Exchange Symposium. The key marker of this hyperpigmentation, she said, is that “it’s strongly photoaccentuated,” affecting areas exposed to the sun — but it also tends to spare the knuckles on patients’ hands.
Used Like an Opioid, But It’s Not Regulated
Kratom is a plant common in southeast Asia and is used as an analgesic. It’s marketed as a “legal opioid” or “legal high” and is sold in 2- or 3-ounce containers of extract or sold as a powder, Dr. Woolery-Lloyd said. The leaves may be boiled into a tea, smoked, chewed, or put into capsules, according to a case report published in February in the Journal of Integrative Dermatology. It is used worldwide and is not regulated in the United States.
“Many of our patients think kratom is a safe, herbal supplement” but often don’t know it can have several side effects and can be addictive, Dr. Woolery-Lloyd said. Its popularity is increasing as reflected by the number of posts related to kratom on social media platforms.
In the February case report, Shaina Patel, BA, and Nathaniel Phelan, MD, from Kansas City University, Kansas City, Missouri, wrote that side effects of kratom include drowsiness, tachycardia, vomiting, respiratory depression, and cardiac arrest, in addition to confusion and hallucinations.
Kratom also has many different effects on the psyche, Dr. Woolery-Lloyd said at the meeting. At low doses, it blocks the reuptake of norepinephrine, serotonin, and dopamine, producing a motivational effect, and at high doses, it creates an analgesic, calming effect. And people who chronically consume high doses of kratom may be susceptible to hyperpigmentation.
Kratom-associated hyperpigmentation should be considered as a diagnosis when evaluating patients for other drug-associated pigmentary disorders, “especially if pigment is photodistributed,” she said. “If you see new-onset hyperpigmentation or onset over several months and it’s very photoaccentuated, definitely ask about use of kratom.”
Case Reports Show Patterns of Presentation
A 2022 report from Landon R. Powell, BS, with the department of biology, Whitworth University in Spokane, Washington, and coauthors, published in JAAD Case Reports, noted that kratom use in the United States has increased dramatically. “As measured by call reports to the United States National Poison Data System, in 2011, there were 11 reported kratom exposures, and in the first 7 months of 2018, there were 357 reported exposures,” they wrote.
An estimated 1.7 million Americans aged ≥ 12 years said they had used kratom in the previous year, according to the Substance Abuse and Mental Health Services Administration 2021 National Survey on Drug Use and Health.
In the case report, Mr. Powell and coauthors described a 54-year-old White male patient who had been using kratom for the previous four to five years to reduce opioid use. During this period, he consumed kratom powder mixed with orange juice three to four times a day. He presented with “diffuse hyperpigmented patches on his arms and face in a photodistributed manner, with notable sparing of the knuckles on both hands.”
Dark Gray-Blue Skin
In the more recent case report, Ms. Patel and Dr. Phelan described a 30-year-old White male patient who presented with dark gray-blue skin coloring on his cheeks, back of his neck, and the backs of his hands and forearms. He had no other medical conditions and did not take any medications or supplements that cause hyperpigmentation while using kratom.
The patient had been taking kratom for years in the wake of an opioid addiction following medications for a high school injury. He developed an opioid use disorder and tried to replace his pain medications with kratom.
“The patient stopped using kratom in May 2022, but the discoloration remains. It has not regressed in the following 16 months after discontinuing kratom use,” the authors wrote, noting that “whether or not the hyperpigmentation is able to regress is unknown.”
Dr. Woolery-Lloyd is a consultant for AbbVie, Incyte, Johnson & Johnson Consumer, LivDerm, and L’Oreal; a speaker for Eli Lilly, Incyte, L’Oreal, and Ortho Dermatologics; and a researcher/investigator for AbbVie, Allergan, Eirion Therapeutics, Galderma, Pfizer, Sanofi, and Vyne Therapeutics.
According to an information page on kratom on the Food and Drug Administration website, health care professionals and consumers can report adverse reactions associated with kratom to the FDA’s MedWatch program.
A version of this article appeared on Medscape.com.
VEXAS Syndrome: Study Highlights Cutaneous Symptoms
Additionally, the most common histologic findings include leukocytoclastic vasculitis, neutrophilic dermatosis, and perivascular dermatitis; different variants in the UBA1 gene are associated with specific skin manifestations.
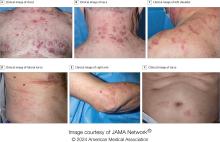
Those are key findings from a cohort study of 112 patients with VEXAS published online in JAMA Dermatology. The study, conducted by researchers at the National Institutes of Health (NIH) and several other institutions, aimed to define the spectrum of cutaneous manifestations in VEXAS in association with genetic, histologic, and other clinical findings.
First described in 2020, VEXAS syndrome is an adult-onset multisystem disease that can pose a diagnostic challenge to clinicians, the study’s corresponding author, Edward W. Cowen, MD, MHSc, of the dermatology branch at the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), said in an interview. The disease is caused by pathogenic variants in the UBA1 gene, located on the X chromosome. Affected individuals exhibit a wide range of manifestations, including cytopenia/myelodysplasia, multiorgan systemic inflammation, and cutaneous involvement.
“Patients may present to a variety of disease specialists depending on their symptoms and providers may not immediately consider a genetic etiology in an older individual,” Dr. Cowen said in an interview. “Although skin involvement occurs in more than 80% of patients, it is pleomorphic and may resemble a variety of other conditions such as vasculitis and Sweet syndrome.”
To better understand the cutaneous manifestations of VEXAS syndrome, the researchers evaluated data from 112 patients with VEXAS-defining genetic variants in the UBA1 gene between 2019 and 2023. Of the 112 patients, 73 underwent medical record review only, and 39 were prospectively evaluated at NIH. All but one of the patients were men, 94% were White individuals, and their mean age was 64 years. Skin involvement occurred in 83% of cases and was the most common presenting feature of VEXAS in 61% of cases.
Of the 64 histopathologic reports available from 60 patients, the main skin histopathologic findings were leukocytoclastic vasculitis in 23 patients (36%), neutrophilic dermatosis in 22 patients (34%), and perivascular dermatitis in 19 patients (30%). According to Dr. Cowen, one key histologic finding was a distinct pattern of “histiocytoid” dermal neutrophilic inflammation, which was present in 13 of 15 specimens (86%) that underwent central re-review. “This pattern can occasionally also be seen in patients with Sweet syndrome, unrelated to VEXAS, but was a hallmark feature found in the majority of skin biopsies of patients with VEXAS,” he said.
“Together with another pathologic finding, leukocytoclasia, these features can be useful clues to alert the pathologist to a potential diagnosis of VEXAS. This myeloid predominant pattern of skin inflammation was also most strongly associated with the leucine pathogenic variant of the UBA1 gene.” In contrast, cutaneous vasculitis was most strongly associated with the valine pathogenic variant of UBA1. “This is important because the valine variant has been previously independently linked to decreased survival,” he said.
In findings related to pathogenic genetic variants, the researchers observed that the p.Met41Leu variant was most frequently associated with neutrophilic dermal infiltrates in 14 of 17 patients (82%) with this variant and often resembled histiocytoid Sweet syndrome. In addition, the p.Met41Val variant was associated with vasculitic lesions in 11 of 20 patients (55%) with this variant and with a mixed leukocytic infiltrate in 17 of these 20 patients (85%).
Treatment Outcomes
In the realm of therapies, skin manifestations improved in 67 of 73 patients (92%) treated with oral prednisone, while treatment with the interleukin-1 receptor antagonist anakinra improved cutaneous disease in 9 of the 16 (56%) who received it. However, 12 (75%) of those who received anakinra developed severe injection-site reactions, including ulceration in two patients and abscess formation in one patient.
Dr. Cowen noted that VEXAS is associated with high mortality (22% in this cohort), and a high degree of suspicion is required to diagnose patients with VEXAS before significant end organ damage has occurred. “This diagnosis should be considered in all older male patients who present with neutrophilic dermatosis — particularly histiocytoid Sweet syndrome, vasculitis, or leukocytoclasia without vasculitis. Patients who appear to have isolated skin involvement may have cytopenias and acute phase reactants. Therefore, complete blood count with differential and ESR and CRP should be considered to investigate for macrocytosis, cytopenias, and systemic inflammation.”
He acknowledged certain limitations of the study, including the fact that many patients were first evaluated at the NIH after having disease symptoms for many months or years. “It is possible that patients with VEXAS referred to the NIH, either for genetic testing or in person evaluation, represent a population with more aggressive disease.”
Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Connecticut, who was asked to comment on the study, emphasized the importance of the UBA1 mutation in the diagnosis of this complex syndrome. “Dermatologists should be aware of VEXAS syndrome as the majority of patients present with skin lesions, which can range from urticarial to Sweet syndrome–like to palpable purpura,” Dr. Ko said.
“Chondritis and periorbital edema, sometimes unilateral, are also associated. Histopathologic clues include a predominantly histiocytoid infiltrate,” she noted. In addition, “the prominent myxoid stroma around blood vessels and adnexal structures as a clue to VEXAS syndrome surprised me; I had not read that before.”
The study was supported by the Intramural Research Program of NIAMS. One of the study authors reported personal fees from Alexion, Novartis, and Sobi outside of the submitted work. No other disclosures were reported. Dr. Ko reported having no disclosures.
A version of this article appeared on Medscape.com .
Additionally, the most common histologic findings include leukocytoclastic vasculitis, neutrophilic dermatosis, and perivascular dermatitis; different variants in the UBA1 gene are associated with specific skin manifestations.
Those are key findings from a cohort study of 112 patients with VEXAS published online in JAMA Dermatology. The study, conducted by researchers at the National Institutes of Health (NIH) and several other institutions, aimed to define the spectrum of cutaneous manifestations in VEXAS in association with genetic, histologic, and other clinical findings.
First described in 2020, VEXAS syndrome is an adult-onset multisystem disease that can pose a diagnostic challenge to clinicians, the study’s corresponding author, Edward W. Cowen, MD, MHSc, of the dermatology branch at the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), said in an interview. The disease is caused by pathogenic variants in the UBA1 gene, located on the X chromosome. Affected individuals exhibit a wide range of manifestations, including cytopenia/myelodysplasia, multiorgan systemic inflammation, and cutaneous involvement.
“Patients may present to a variety of disease specialists depending on their symptoms and providers may not immediately consider a genetic etiology in an older individual,” Dr. Cowen said in an interview. “Although skin involvement occurs in more than 80% of patients, it is pleomorphic and may resemble a variety of other conditions such as vasculitis and Sweet syndrome.”
To better understand the cutaneous manifestations of VEXAS syndrome, the researchers evaluated data from 112 patients with VEXAS-defining genetic variants in the UBA1 gene between 2019 and 2023. Of the 112 patients, 73 underwent medical record review only, and 39 were prospectively evaluated at NIH. All but one of the patients were men, 94% were White individuals, and their mean age was 64 years. Skin involvement occurred in 83% of cases and was the most common presenting feature of VEXAS in 61% of cases.
Of the 64 histopathologic reports available from 60 patients, the main skin histopathologic findings were leukocytoclastic vasculitis in 23 patients (36%), neutrophilic dermatosis in 22 patients (34%), and perivascular dermatitis in 19 patients (30%). According to Dr. Cowen, one key histologic finding was a distinct pattern of “histiocytoid” dermal neutrophilic inflammation, which was present in 13 of 15 specimens (86%) that underwent central re-review. “This pattern can occasionally also be seen in patients with Sweet syndrome, unrelated to VEXAS, but was a hallmark feature found in the majority of skin biopsies of patients with VEXAS,” he said.
“Together with another pathologic finding, leukocytoclasia, these features can be useful clues to alert the pathologist to a potential diagnosis of VEXAS. This myeloid predominant pattern of skin inflammation was also most strongly associated with the leucine pathogenic variant of the UBA1 gene.” In contrast, cutaneous vasculitis was most strongly associated with the valine pathogenic variant of UBA1. “This is important because the valine variant has been previously independently linked to decreased survival,” he said.
In findings related to pathogenic genetic variants, the researchers observed that the p.Met41Leu variant was most frequently associated with neutrophilic dermal infiltrates in 14 of 17 patients (82%) with this variant and often resembled histiocytoid Sweet syndrome. In addition, the p.Met41Val variant was associated with vasculitic lesions in 11 of 20 patients (55%) with this variant and with a mixed leukocytic infiltrate in 17 of these 20 patients (85%).
Treatment Outcomes
In the realm of therapies, skin manifestations improved in 67 of 73 patients (92%) treated with oral prednisone, while treatment with the interleukin-1 receptor antagonist anakinra improved cutaneous disease in 9 of the 16 (56%) who received it. However, 12 (75%) of those who received anakinra developed severe injection-site reactions, including ulceration in two patients and abscess formation in one patient.
Dr. Cowen noted that VEXAS is associated with high mortality (22% in this cohort), and a high degree of suspicion is required to diagnose patients with VEXAS before significant end organ damage has occurred. “This diagnosis should be considered in all older male patients who present with neutrophilic dermatosis — particularly histiocytoid Sweet syndrome, vasculitis, or leukocytoclasia without vasculitis. Patients who appear to have isolated skin involvement may have cytopenias and acute phase reactants. Therefore, complete blood count with differential and ESR and CRP should be considered to investigate for macrocytosis, cytopenias, and systemic inflammation.”
He acknowledged certain limitations of the study, including the fact that many patients were first evaluated at the NIH after having disease symptoms for many months or years. “It is possible that patients with VEXAS referred to the NIH, either for genetic testing or in person evaluation, represent a population with more aggressive disease.”
Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Connecticut, who was asked to comment on the study, emphasized the importance of the UBA1 mutation in the diagnosis of this complex syndrome. “Dermatologists should be aware of VEXAS syndrome as the majority of patients present with skin lesions, which can range from urticarial to Sweet syndrome–like to palpable purpura,” Dr. Ko said.
“Chondritis and periorbital edema, sometimes unilateral, are also associated. Histopathologic clues include a predominantly histiocytoid infiltrate,” she noted. In addition, “the prominent myxoid stroma around blood vessels and adnexal structures as a clue to VEXAS syndrome surprised me; I had not read that before.”
The study was supported by the Intramural Research Program of NIAMS. One of the study authors reported personal fees from Alexion, Novartis, and Sobi outside of the submitted work. No other disclosures were reported. Dr. Ko reported having no disclosures.
A version of this article appeared on Medscape.com .
Additionally, the most common histologic findings include leukocytoclastic vasculitis, neutrophilic dermatosis, and perivascular dermatitis; different variants in the UBA1 gene are associated with specific skin manifestations.
Those are key findings from a cohort study of 112 patients with VEXAS published online in JAMA Dermatology. The study, conducted by researchers at the National Institutes of Health (NIH) and several other institutions, aimed to define the spectrum of cutaneous manifestations in VEXAS in association with genetic, histologic, and other clinical findings.
First described in 2020, VEXAS syndrome is an adult-onset multisystem disease that can pose a diagnostic challenge to clinicians, the study’s corresponding author, Edward W. Cowen, MD, MHSc, of the dermatology branch at the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), said in an interview. The disease is caused by pathogenic variants in the UBA1 gene, located on the X chromosome. Affected individuals exhibit a wide range of manifestations, including cytopenia/myelodysplasia, multiorgan systemic inflammation, and cutaneous involvement.
“Patients may present to a variety of disease specialists depending on their symptoms and providers may not immediately consider a genetic etiology in an older individual,” Dr. Cowen said in an interview. “Although skin involvement occurs in more than 80% of patients, it is pleomorphic and may resemble a variety of other conditions such as vasculitis and Sweet syndrome.”
To better understand the cutaneous manifestations of VEXAS syndrome, the researchers evaluated data from 112 patients with VEXAS-defining genetic variants in the UBA1 gene between 2019 and 2023. Of the 112 patients, 73 underwent medical record review only, and 39 were prospectively evaluated at NIH. All but one of the patients were men, 94% were White individuals, and their mean age was 64 years. Skin involvement occurred in 83% of cases and was the most common presenting feature of VEXAS in 61% of cases.
Of the 64 histopathologic reports available from 60 patients, the main skin histopathologic findings were leukocytoclastic vasculitis in 23 patients (36%), neutrophilic dermatosis in 22 patients (34%), and perivascular dermatitis in 19 patients (30%). According to Dr. Cowen, one key histologic finding was a distinct pattern of “histiocytoid” dermal neutrophilic inflammation, which was present in 13 of 15 specimens (86%) that underwent central re-review. “This pattern can occasionally also be seen in patients with Sweet syndrome, unrelated to VEXAS, but was a hallmark feature found in the majority of skin biopsies of patients with VEXAS,” he said.
“Together with another pathologic finding, leukocytoclasia, these features can be useful clues to alert the pathologist to a potential diagnosis of VEXAS. This myeloid predominant pattern of skin inflammation was also most strongly associated with the leucine pathogenic variant of the UBA1 gene.” In contrast, cutaneous vasculitis was most strongly associated with the valine pathogenic variant of UBA1. “This is important because the valine variant has been previously independently linked to decreased survival,” he said.
In findings related to pathogenic genetic variants, the researchers observed that the p.Met41Leu variant was most frequently associated with neutrophilic dermal infiltrates in 14 of 17 patients (82%) with this variant and often resembled histiocytoid Sweet syndrome. In addition, the p.Met41Val variant was associated with vasculitic lesions in 11 of 20 patients (55%) with this variant and with a mixed leukocytic infiltrate in 17 of these 20 patients (85%).
Treatment Outcomes
In the realm of therapies, skin manifestations improved in 67 of 73 patients (92%) treated with oral prednisone, while treatment with the interleukin-1 receptor antagonist anakinra improved cutaneous disease in 9 of the 16 (56%) who received it. However, 12 (75%) of those who received anakinra developed severe injection-site reactions, including ulceration in two patients and abscess formation in one patient.
Dr. Cowen noted that VEXAS is associated with high mortality (22% in this cohort), and a high degree of suspicion is required to diagnose patients with VEXAS before significant end organ damage has occurred. “This diagnosis should be considered in all older male patients who present with neutrophilic dermatosis — particularly histiocytoid Sweet syndrome, vasculitis, or leukocytoclasia without vasculitis. Patients who appear to have isolated skin involvement may have cytopenias and acute phase reactants. Therefore, complete blood count with differential and ESR and CRP should be considered to investigate for macrocytosis, cytopenias, and systemic inflammation.”
He acknowledged certain limitations of the study, including the fact that many patients were first evaluated at the NIH after having disease symptoms for many months or years. “It is possible that patients with VEXAS referred to the NIH, either for genetic testing or in person evaluation, represent a population with more aggressive disease.”
Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Connecticut, who was asked to comment on the study, emphasized the importance of the UBA1 mutation in the diagnosis of this complex syndrome. “Dermatologists should be aware of VEXAS syndrome as the majority of patients present with skin lesions, which can range from urticarial to Sweet syndrome–like to palpable purpura,” Dr. Ko said.
“Chondritis and periorbital edema, sometimes unilateral, are also associated. Histopathologic clues include a predominantly histiocytoid infiltrate,” she noted. In addition, “the prominent myxoid stroma around blood vessels and adnexal structures as a clue to VEXAS syndrome surprised me; I had not read that before.”
The study was supported by the Intramural Research Program of NIAMS. One of the study authors reported personal fees from Alexion, Novartis, and Sobi outside of the submitted work. No other disclosures were reported. Dr. Ko reported having no disclosures.
A version of this article appeared on Medscape.com .
FROM JAMA DERMATOLOGY
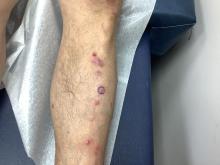
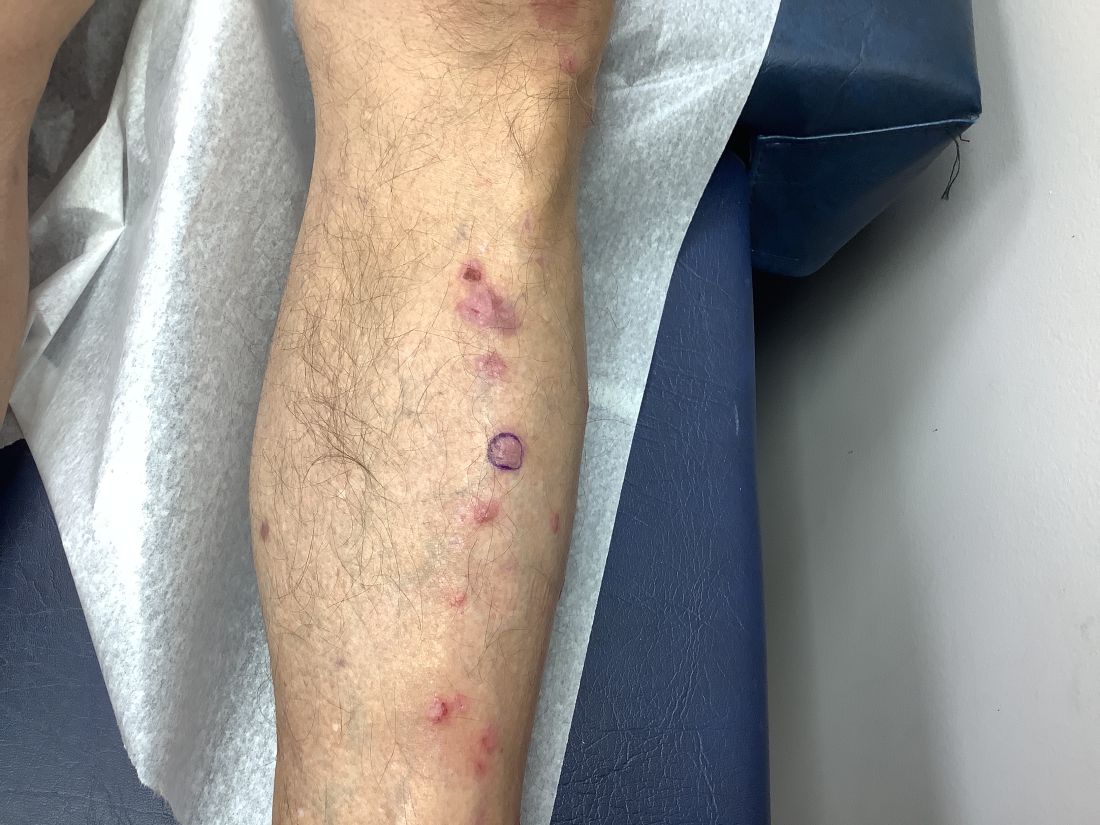
Pruritic, violaceous papules in a patient with renal cell carcinoma
Pembrolizumab (Keytruda) is a programmed cell death protein 1 (PD-1) blocking antibody used to treat different malignancies including melanoma, non–small cell lung cancer, and other advanced solid tumors and hematologic malignancies. and drug rash with eosinophilia and systemic symptoms (DRESS).
Lichen planus-like adverse drug reactions, as seen in this patient, are also referred to as lichenoid drug eruption or drug-induced lichen planus. This cutaneous reaction is one of the more rare side effects of pembrolizumab. It should be noted that in lichenoid reactions, keratinocytes expressing PD-L1 are particularly affected, leading to a dense CD4/CD8 positive lymphocytic infiltration in the basal layer, necrosis of keratinocytes, acanthosis, and hypergranulosis. Subsequently, the cutaneous adverse reaction is a target effect of the PD-1/PD-L1 pathway and not a general hypersensitivity reaction. Clinically, both lichen planus and lichenoid drug eruptions exhibit erythematous papules and plaques. Lichenoid drug eruptions, however, can be scaly, pruritic, and heal with more hyperpigmentation.
A skin biopsy revealed irregular epidermal hyperplasia with jagged rete ridges. Within the dermis, there was a lichenoid inflammatory cell infiltrate obscuring the dermal-epidermal junction. The inflammatory cell infiltrate contained lymphocytes, histiocytes, and eosinophils. A diagnosis of a lichen planus-like adverse drug reaction to pembrolizumab was favored.
If the reaction is mild, topical corticosteroids and oral antihistamines can help with the drug-induced lichen planus. For more severe cases, systemic steroids can be given to help ease the reaction. Physicians should be aware of potential adverse drug effects that can mimic other medical conditions.
The case and photo were submitted by Ms. Towe, Nova Southeastern University College of Osteopathic Medicine, Davie, Florida, and Dr. Berke, Three Rivers Dermatology, Coraopolis, Pennsylvania. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Bansal A et al. Indian Dermatol Online J. 2023 Apr 4;14(3):391-4. doi: 10.4103/idoj.idoj_377_22.
Sethi A, Raj M. Cureus. 2021 Mar 8;13(3):e13768. doi: 10.7759/cureus.13768.
Pembrolizumab (Keytruda) is a programmed cell death protein 1 (PD-1) blocking antibody used to treat different malignancies including melanoma, non–small cell lung cancer, and other advanced solid tumors and hematologic malignancies. and drug rash with eosinophilia and systemic symptoms (DRESS).
Lichen planus-like adverse drug reactions, as seen in this patient, are also referred to as lichenoid drug eruption or drug-induced lichen planus. This cutaneous reaction is one of the more rare side effects of pembrolizumab. It should be noted that in lichenoid reactions, keratinocytes expressing PD-L1 are particularly affected, leading to a dense CD4/CD8 positive lymphocytic infiltration in the basal layer, necrosis of keratinocytes, acanthosis, and hypergranulosis. Subsequently, the cutaneous adverse reaction is a target effect of the PD-1/PD-L1 pathway and not a general hypersensitivity reaction. Clinically, both lichen planus and lichenoid drug eruptions exhibit erythematous papules and plaques. Lichenoid drug eruptions, however, can be scaly, pruritic, and heal with more hyperpigmentation.
A skin biopsy revealed irregular epidermal hyperplasia with jagged rete ridges. Within the dermis, there was a lichenoid inflammatory cell infiltrate obscuring the dermal-epidermal junction. The inflammatory cell infiltrate contained lymphocytes, histiocytes, and eosinophils. A diagnosis of a lichen planus-like adverse drug reaction to pembrolizumab was favored.
If the reaction is mild, topical corticosteroids and oral antihistamines can help with the drug-induced lichen planus. For more severe cases, systemic steroids can be given to help ease the reaction. Physicians should be aware of potential adverse drug effects that can mimic other medical conditions.
The case and photo were submitted by Ms. Towe, Nova Southeastern University College of Osteopathic Medicine, Davie, Florida, and Dr. Berke, Three Rivers Dermatology, Coraopolis, Pennsylvania. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Bansal A et al. Indian Dermatol Online J. 2023 Apr 4;14(3):391-4. doi: 10.4103/idoj.idoj_377_22.
Sethi A, Raj M. Cureus. 2021 Mar 8;13(3):e13768. doi: 10.7759/cureus.13768.
Pembrolizumab (Keytruda) is a programmed cell death protein 1 (PD-1) blocking antibody used to treat different malignancies including melanoma, non–small cell lung cancer, and other advanced solid tumors and hematologic malignancies. and drug rash with eosinophilia and systemic symptoms (DRESS).
Lichen planus-like adverse drug reactions, as seen in this patient, are also referred to as lichenoid drug eruption or drug-induced lichen planus. This cutaneous reaction is one of the more rare side effects of pembrolizumab. It should be noted that in lichenoid reactions, keratinocytes expressing PD-L1 are particularly affected, leading to a dense CD4/CD8 positive lymphocytic infiltration in the basal layer, necrosis of keratinocytes, acanthosis, and hypergranulosis. Subsequently, the cutaneous adverse reaction is a target effect of the PD-1/PD-L1 pathway and not a general hypersensitivity reaction. Clinically, both lichen planus and lichenoid drug eruptions exhibit erythematous papules and plaques. Lichenoid drug eruptions, however, can be scaly, pruritic, and heal with more hyperpigmentation.
A skin biopsy revealed irregular epidermal hyperplasia with jagged rete ridges. Within the dermis, there was a lichenoid inflammatory cell infiltrate obscuring the dermal-epidermal junction. The inflammatory cell infiltrate contained lymphocytes, histiocytes, and eosinophils. A diagnosis of a lichen planus-like adverse drug reaction to pembrolizumab was favored.
If the reaction is mild, topical corticosteroids and oral antihistamines can help with the drug-induced lichen planus. For more severe cases, systemic steroids can be given to help ease the reaction. Physicians should be aware of potential adverse drug effects that can mimic other medical conditions.
The case and photo were submitted by Ms. Towe, Nova Southeastern University College of Osteopathic Medicine, Davie, Florida, and Dr. Berke, Three Rivers Dermatology, Coraopolis, Pennsylvania. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Bansal A et al. Indian Dermatol Online J. 2023 Apr 4;14(3):391-4. doi: 10.4103/idoj.idoj_377_22.
Sethi A, Raj M. Cureus. 2021 Mar 8;13(3):e13768. doi: 10.7759/cureus.13768.
Photoprotection: Benefits of Sunscreens With Iron Oxide
CHICAGO — One of the more recent developments in sunscreen technology is the addition of iron oxide to mineral sunscreens.
Iron oxide is “an excellent pigment” that absorbs and blocks visible light, which is particularly important in individuals with Fitzpatrick skin types III-VI, Zoe D. Draelos, MD, consulting professor of dermatology at Duke University, Durham, North Carolina, said at the Pigmentation Disorders Exchange symposium.
Susan C. Taylor, MD, professor of dermatology at the University of Pennsylvania, Philadelphia, who spoke at the conference, also recommended tinted sunscreen with iron oxide for patients with skin of color. “It still needs to be broad spectrum,” she said, “and at least an SPF [Sun Protection Factor] 30.”
When blended with mineral sunscreens, iron oxide can reduce transmission of visible light by 90% and can protect patients from hyperpigmentation. Iron oxide comes in different colors blended together for various degrees of tinting.
Dr. Taylor noted that iron oxide is listed under the inactive ingredients. “The literature indicates a 3% concentration to aim for, but we don’t know the concentration in most of the products,” she added.
During her presentation, Dr. Draelos noted that inorganic sunscreens, such as zinc oxide and titanium oxides, are highly effective but make the skin white and pasty. To address this issue, many companies are now grinding these materials into such small particles that they are transparent.
“That’s great, except the smaller the particle is, the less UV [ultraviolet] radiation it reflects and that lowers the [SPF],” she said.
In addition to providing photoprotection, sunscreens in general provide protection from nanoparticles in tobacco and combustion, such as traffic exhaust, which can harm skin over time. “Moisturizers and sunscreens are the best way to protect against pollution and tobacco nanoparticle damage, which can contribute to inflammation,” she noted. They create a film over the skin and trap the nanoparticles.
Start the Patient Visit With a Photoprotection Talk
At the meeting, Dr. Taylor recommended that for all patients with hypopigmentation and hyperpigmentation disorders, “treatment really begins with photoprotection.”
She acknowledged that photoprotection discussions, including the basics of seeking shade, wearing protective clothing, and avoiding midday sun, often come at the end of the patient visit but she urged dermatologists to make that the first topic instead.
Dr. Taylor said a question often asked of patients of color about prolonged sun exposure — whether their skin turns bright red after too much sun — may get a negative reply. The better question is whether the patient has experienced tender skin after too much sun — which can signify a sunburn, she said.
Dr. Draelos reported no relevant financial relationships. Dr. Taylor reported financial relationships and grant support from multiple pharmaceutical companies.
A version of this article first appeared on Medscape.com.
CHICAGO — One of the more recent developments in sunscreen technology is the addition of iron oxide to mineral sunscreens.
Iron oxide is “an excellent pigment” that absorbs and blocks visible light, which is particularly important in individuals with Fitzpatrick skin types III-VI, Zoe D. Draelos, MD, consulting professor of dermatology at Duke University, Durham, North Carolina, said at the Pigmentation Disorders Exchange symposium.
Susan C. Taylor, MD, professor of dermatology at the University of Pennsylvania, Philadelphia, who spoke at the conference, also recommended tinted sunscreen with iron oxide for patients with skin of color. “It still needs to be broad spectrum,” she said, “and at least an SPF [Sun Protection Factor] 30.”
When blended with mineral sunscreens, iron oxide can reduce transmission of visible light by 90% and can protect patients from hyperpigmentation. Iron oxide comes in different colors blended together for various degrees of tinting.
Dr. Taylor noted that iron oxide is listed under the inactive ingredients. “The literature indicates a 3% concentration to aim for, but we don’t know the concentration in most of the products,” she added.
During her presentation, Dr. Draelos noted that inorganic sunscreens, such as zinc oxide and titanium oxides, are highly effective but make the skin white and pasty. To address this issue, many companies are now grinding these materials into such small particles that they are transparent.
“That’s great, except the smaller the particle is, the less UV [ultraviolet] radiation it reflects and that lowers the [SPF],” she said.
In addition to providing photoprotection, sunscreens in general provide protection from nanoparticles in tobacco and combustion, such as traffic exhaust, which can harm skin over time. “Moisturizers and sunscreens are the best way to protect against pollution and tobacco nanoparticle damage, which can contribute to inflammation,” she noted. They create a film over the skin and trap the nanoparticles.
Start the Patient Visit With a Photoprotection Talk
At the meeting, Dr. Taylor recommended that for all patients with hypopigmentation and hyperpigmentation disorders, “treatment really begins with photoprotection.”
She acknowledged that photoprotection discussions, including the basics of seeking shade, wearing protective clothing, and avoiding midday sun, often come at the end of the patient visit but she urged dermatologists to make that the first topic instead.
Dr. Taylor said a question often asked of patients of color about prolonged sun exposure — whether their skin turns bright red after too much sun — may get a negative reply. The better question is whether the patient has experienced tender skin after too much sun — which can signify a sunburn, she said.
Dr. Draelos reported no relevant financial relationships. Dr. Taylor reported financial relationships and grant support from multiple pharmaceutical companies.
A version of this article first appeared on Medscape.com.
CHICAGO — One of the more recent developments in sunscreen technology is the addition of iron oxide to mineral sunscreens.
Iron oxide is “an excellent pigment” that absorbs and blocks visible light, which is particularly important in individuals with Fitzpatrick skin types III-VI, Zoe D. Draelos, MD, consulting professor of dermatology at Duke University, Durham, North Carolina, said at the Pigmentation Disorders Exchange symposium.
Susan C. Taylor, MD, professor of dermatology at the University of Pennsylvania, Philadelphia, who spoke at the conference, also recommended tinted sunscreen with iron oxide for patients with skin of color. “It still needs to be broad spectrum,” she said, “and at least an SPF [Sun Protection Factor] 30.”
When blended with mineral sunscreens, iron oxide can reduce transmission of visible light by 90% and can protect patients from hyperpigmentation. Iron oxide comes in different colors blended together for various degrees of tinting.
Dr. Taylor noted that iron oxide is listed under the inactive ingredients. “The literature indicates a 3% concentration to aim for, but we don’t know the concentration in most of the products,” she added.
During her presentation, Dr. Draelos noted that inorganic sunscreens, such as zinc oxide and titanium oxides, are highly effective but make the skin white and pasty. To address this issue, many companies are now grinding these materials into such small particles that they are transparent.
“That’s great, except the smaller the particle is, the less UV [ultraviolet] radiation it reflects and that lowers the [SPF],” she said.
In addition to providing photoprotection, sunscreens in general provide protection from nanoparticles in tobacco and combustion, such as traffic exhaust, which can harm skin over time. “Moisturizers and sunscreens are the best way to protect against pollution and tobacco nanoparticle damage, which can contribute to inflammation,” she noted. They create a film over the skin and trap the nanoparticles.
Start the Patient Visit With a Photoprotection Talk
At the meeting, Dr. Taylor recommended that for all patients with hypopigmentation and hyperpigmentation disorders, “treatment really begins with photoprotection.”
She acknowledged that photoprotection discussions, including the basics of seeking shade, wearing protective clothing, and avoiding midday sun, often come at the end of the patient visit but she urged dermatologists to make that the first topic instead.
Dr. Taylor said a question often asked of patients of color about prolonged sun exposure — whether their skin turns bright red after too much sun — may get a negative reply. The better question is whether the patient has experienced tender skin after too much sun — which can signify a sunburn, she said.
Dr. Draelos reported no relevant financial relationships. Dr. Taylor reported financial relationships and grant support from multiple pharmaceutical companies.
A version of this article first appeared on Medscape.com.
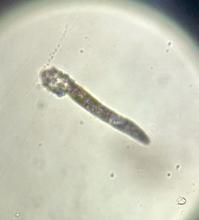
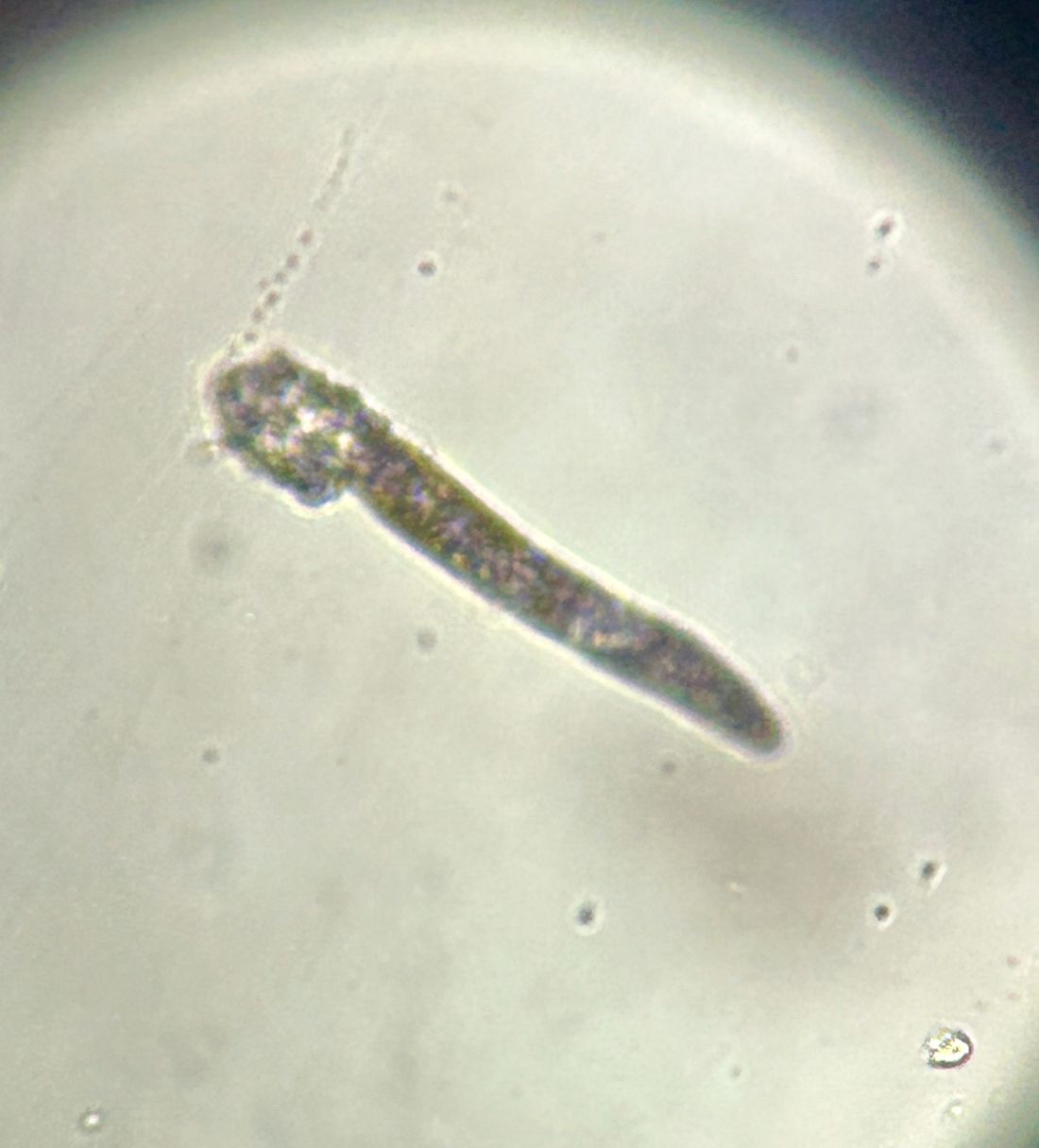
A 7-year-old female presents with persistent pimples on the nose and cheeks for approximately 1 year
Diagnosis
During the visit, skin scrapings were performed, revealing several Demodex mites, confirming the diagnosis of demodicosis.
Demodex folliculorum and Demodex brevis are the two common species implicated. The life cycle of Demodex spp. occurs in the sebaceous glands, leading to mechanical and chemical irritation of the skin.
Various immune responses are also triggered, such as a keratinocyte response via Toll-like receptor 2. Patients usually present with non-specific symptoms such as skin erythema, irritation, peeling, and dryness on the cheeks, eyelids, and paranasal areas. Patients may develop a maculopapular or rosacea-like rash.
Diagnosis is often made through microscopic examination of a skin sample in KOH solution. In rare occasions, a skin surface standardization biopsy method may be used, which determines the density of mites per 1 cm2. Dermoscopy may identify spiky white structures. Molecular methods such as PCR can be used but are not standard.
The differential diagnosis may include acne, rosacea, folliculitis, and Candida infection. Demodicosis can be differentiated by history and further studies including dermoscopy.
Acne vulgaris is an inflammatory disease of the skin’s pilosebaceous unit, primarily involving the face and trunk. It can present with comedones, papules, pustules, and nodules. Secondary signs suggestive of acne vulgaris include scars, erythema, and hyperpigmentation. All forms of acne share a common pathogenesis resulting in the formation of microcomedones, precursors for all clinical acne lesions. In this patient, the absence of microcomedones and the presence of primary inflammatory papules localized to the nose and cheeks suggested an alternative diagnosis.
Rosacea was also considered in the differential diagnosis. Rosacea is an inflammatory dermatosis characterized by erythema, telangiectasia, recurrent flushing, and inflammatory lesions including papulopustules and swelling, primarily affecting the face. The pathogenesis of rosacea is not fully understood but is suggested to involve immune-mediated responses. Vascular dysregulation and reactive oxygen species damage keratinocytes, fibroblasts, and endothelial cells. A higher incidence of rosacea in those with a family history and UV exposure is a known trigger. Demodex folliculorum and Helicobacter pylori are also implicated. Occasionally, Demodex infestation and rosacea may co-occur, and treatment with topical metronidazole can be helpful.
Folliculitis is an infection and inflammation of the hair follicles, forming pustules or erythematous papules over hair-covered skin. It is commonly caused by bacterial infection but can also be due to fungi, viruses, and noninfectious causes such as eosinophilic folliculitis. Diagnosis is clinical, based on physical exam and history, such as recent increased sweating or scratching. KOH prep can be used for Malassezia folliculitis and skin biopsy for eosinophilic folliculitis. Treatment targets the underlying cause. Most bacterial folliculitis cases resolve without treatment, but topical antibiotics may be used. Fungal folliculitis requires oral antifungals, and herpes simplex folliculitis can be treated with antiviral medications.
Cutaneous candidiasis is an infection of the skin by various Candida species, commonly C. albicans. Superficial infections of the skin and mucous membranes, such as intertrigo, are common types. Risk factors include immunosuppression, endocrine disorders, or compromised blood flow. Increased humidity, occlusion, broken skin barriers, and altered skin microbial flora contribute to Candida infection. Diagnosis is clinical but can be confirmed by KOH prep, microscopy, and culture. Treatment involves anti-inflammatory, antibacterial, and antifungal medications. Topical clotrimazole, nystatin, and miconazole are commonly used. Recurrence is prevented by keeping the affected area dry with barrier creams.
Therapeutic goals include arresting mite reproduction, elimination, and preventing recurrent infestations. Treatment may last several months, and the choice of drug depends on patient factors. There have been no standardized treatment studies or long-term effectiveness analyses. Antibiotics such as tetracycline, metronidazole, doxycycline, and ivermectin may be used to prevent proliferation. Permethrin, benzyl benzoate, crotamiton, lindane, and sulfur have also been used. Metronidazole is a common treatment for demodicosis, as was used in our patient for several weeks until the lesions cleared. Systemic metronidazole therapy may be indicated for reducing Demodex spp. density. Severe cases, particularly in immunocompromised individuals, may require oral ivermectin. Appropriate hygiene is important for prevention, such as washing the face with non-oily cleansers and laundering linens regularly.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Mr. Lee is a medical student at the University of California San Diego.
Suggested Reading
Chudzicka-Strugała I et al. Demodicosis in different age groups and alternative treatment options—A review. J Clin Med. 2023 Feb 19;12(4):1649. doi: 10.3390/jcm12041649.
Eichenfield DZ et al. Management of acne vulgaris: A review. JAMA. 2021 Nov 23;326(20):2055-2067. doi: 10.1001/jama.2021.17633.
Sharma A et al. Rosacea management: A comprehensive review. J Cosmet Dermatol. 2022 May;21(5):1895-1904. doi: 10.1111/jocd.14816.
Taudorf EH et al. Cutaneous candidiasis — an evidence-based review of topical and systemic treatments to inform clinical practice. J Eur Acad Dermatol Venereol. 2019 Oct;33(10):1863-1873. doi: 10.1111/jdv.15782.
Winters RD, Mitchell M. Folliculitis. [Updated 2023 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547754/
Diagnosis
During the visit, skin scrapings were performed, revealing several Demodex mites, confirming the diagnosis of demodicosis.
Demodex folliculorum and Demodex brevis are the two common species implicated. The life cycle of Demodex spp. occurs in the sebaceous glands, leading to mechanical and chemical irritation of the skin.
Various immune responses are also triggered, such as a keratinocyte response via Toll-like receptor 2. Patients usually present with non-specific symptoms such as skin erythema, irritation, peeling, and dryness on the cheeks, eyelids, and paranasal areas. Patients may develop a maculopapular or rosacea-like rash.
Diagnosis is often made through microscopic examination of a skin sample in KOH solution. In rare occasions, a skin surface standardization biopsy method may be used, which determines the density of mites per 1 cm2. Dermoscopy may identify spiky white structures. Molecular methods such as PCR can be used but are not standard.
The differential diagnosis may include acne, rosacea, folliculitis, and Candida infection. Demodicosis can be differentiated by history and further studies including dermoscopy.
Acne vulgaris is an inflammatory disease of the skin’s pilosebaceous unit, primarily involving the face and trunk. It can present with comedones, papules, pustules, and nodules. Secondary signs suggestive of acne vulgaris include scars, erythema, and hyperpigmentation. All forms of acne share a common pathogenesis resulting in the formation of microcomedones, precursors for all clinical acne lesions. In this patient, the absence of microcomedones and the presence of primary inflammatory papules localized to the nose and cheeks suggested an alternative diagnosis.
Rosacea was also considered in the differential diagnosis. Rosacea is an inflammatory dermatosis characterized by erythema, telangiectasia, recurrent flushing, and inflammatory lesions including papulopustules and swelling, primarily affecting the face. The pathogenesis of rosacea is not fully understood but is suggested to involve immune-mediated responses. Vascular dysregulation and reactive oxygen species damage keratinocytes, fibroblasts, and endothelial cells. A higher incidence of rosacea in those with a family history and UV exposure is a known trigger. Demodex folliculorum and Helicobacter pylori are also implicated. Occasionally, Demodex infestation and rosacea may co-occur, and treatment with topical metronidazole can be helpful.
Folliculitis is an infection and inflammation of the hair follicles, forming pustules or erythematous papules over hair-covered skin. It is commonly caused by bacterial infection but can also be due to fungi, viruses, and noninfectious causes such as eosinophilic folliculitis. Diagnosis is clinical, based on physical exam and history, such as recent increased sweating or scratching. KOH prep can be used for Malassezia folliculitis and skin biopsy for eosinophilic folliculitis. Treatment targets the underlying cause. Most bacterial folliculitis cases resolve without treatment, but topical antibiotics may be used. Fungal folliculitis requires oral antifungals, and herpes simplex folliculitis can be treated with antiviral medications.
Cutaneous candidiasis is an infection of the skin by various Candida species, commonly C. albicans. Superficial infections of the skin and mucous membranes, such as intertrigo, are common types. Risk factors include immunosuppression, endocrine disorders, or compromised blood flow. Increased humidity, occlusion, broken skin barriers, and altered skin microbial flora contribute to Candida infection. Diagnosis is clinical but can be confirmed by KOH prep, microscopy, and culture. Treatment involves anti-inflammatory, antibacterial, and antifungal medications. Topical clotrimazole, nystatin, and miconazole are commonly used. Recurrence is prevented by keeping the affected area dry with barrier creams.
Therapeutic goals include arresting mite reproduction, elimination, and preventing recurrent infestations. Treatment may last several months, and the choice of drug depends on patient factors. There have been no standardized treatment studies or long-term effectiveness analyses. Antibiotics such as tetracycline, metronidazole, doxycycline, and ivermectin may be used to prevent proliferation. Permethrin, benzyl benzoate, crotamiton, lindane, and sulfur have also been used. Metronidazole is a common treatment for demodicosis, as was used in our patient for several weeks until the lesions cleared. Systemic metronidazole therapy may be indicated for reducing Demodex spp. density. Severe cases, particularly in immunocompromised individuals, may require oral ivermectin. Appropriate hygiene is important for prevention, such as washing the face with non-oily cleansers and laundering linens regularly.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Mr. Lee is a medical student at the University of California San Diego.
Suggested Reading
Chudzicka-Strugała I et al. Demodicosis in different age groups and alternative treatment options—A review. J Clin Med. 2023 Feb 19;12(4):1649. doi: 10.3390/jcm12041649.
Eichenfield DZ et al. Management of acne vulgaris: A review. JAMA. 2021 Nov 23;326(20):2055-2067. doi: 10.1001/jama.2021.17633.
Sharma A et al. Rosacea management: A comprehensive review. J Cosmet Dermatol. 2022 May;21(5):1895-1904. doi: 10.1111/jocd.14816.
Taudorf EH et al. Cutaneous candidiasis — an evidence-based review of topical and systemic treatments to inform clinical practice. J Eur Acad Dermatol Venereol. 2019 Oct;33(10):1863-1873. doi: 10.1111/jdv.15782.
Winters RD, Mitchell M. Folliculitis. [Updated 2023 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547754/
Diagnosis
During the visit, skin scrapings were performed, revealing several Demodex mites, confirming the diagnosis of demodicosis.
Demodex folliculorum and Demodex brevis are the two common species implicated. The life cycle of Demodex spp. occurs in the sebaceous glands, leading to mechanical and chemical irritation of the skin.
Various immune responses are also triggered, such as a keratinocyte response via Toll-like receptor 2. Patients usually present with non-specific symptoms such as skin erythema, irritation, peeling, and dryness on the cheeks, eyelids, and paranasal areas. Patients may develop a maculopapular or rosacea-like rash.
Diagnosis is often made through microscopic examination of a skin sample in KOH solution. In rare occasions, a skin surface standardization biopsy method may be used, which determines the density of mites per 1 cm2. Dermoscopy may identify spiky white structures. Molecular methods such as PCR can be used but are not standard.
The differential diagnosis may include acne, rosacea, folliculitis, and Candida infection. Demodicosis can be differentiated by history and further studies including dermoscopy.
Acne vulgaris is an inflammatory disease of the skin’s pilosebaceous unit, primarily involving the face and trunk. It can present with comedones, papules, pustules, and nodules. Secondary signs suggestive of acne vulgaris include scars, erythema, and hyperpigmentation. All forms of acne share a common pathogenesis resulting in the formation of microcomedones, precursors for all clinical acne lesions. In this patient, the absence of microcomedones and the presence of primary inflammatory papules localized to the nose and cheeks suggested an alternative diagnosis.
Rosacea was also considered in the differential diagnosis. Rosacea is an inflammatory dermatosis characterized by erythema, telangiectasia, recurrent flushing, and inflammatory lesions including papulopustules and swelling, primarily affecting the face. The pathogenesis of rosacea is not fully understood but is suggested to involve immune-mediated responses. Vascular dysregulation and reactive oxygen species damage keratinocytes, fibroblasts, and endothelial cells. A higher incidence of rosacea in those with a family history and UV exposure is a known trigger. Demodex folliculorum and Helicobacter pylori are also implicated. Occasionally, Demodex infestation and rosacea may co-occur, and treatment with topical metronidazole can be helpful.
Folliculitis is an infection and inflammation of the hair follicles, forming pustules or erythematous papules over hair-covered skin. It is commonly caused by bacterial infection but can also be due to fungi, viruses, and noninfectious causes such as eosinophilic folliculitis. Diagnosis is clinical, based on physical exam and history, such as recent increased sweating or scratching. KOH prep can be used for Malassezia folliculitis and skin biopsy for eosinophilic folliculitis. Treatment targets the underlying cause. Most bacterial folliculitis cases resolve without treatment, but topical antibiotics may be used. Fungal folliculitis requires oral antifungals, and herpes simplex folliculitis can be treated with antiviral medications.
Cutaneous candidiasis is an infection of the skin by various Candida species, commonly C. albicans. Superficial infections of the skin and mucous membranes, such as intertrigo, are common types. Risk factors include immunosuppression, endocrine disorders, or compromised blood flow. Increased humidity, occlusion, broken skin barriers, and altered skin microbial flora contribute to Candida infection. Diagnosis is clinical but can be confirmed by KOH prep, microscopy, and culture. Treatment involves anti-inflammatory, antibacterial, and antifungal medications. Topical clotrimazole, nystatin, and miconazole are commonly used. Recurrence is prevented by keeping the affected area dry with barrier creams.
Therapeutic goals include arresting mite reproduction, elimination, and preventing recurrent infestations. Treatment may last several months, and the choice of drug depends on patient factors. There have been no standardized treatment studies or long-term effectiveness analyses. Antibiotics such as tetracycline, metronidazole, doxycycline, and ivermectin may be used to prevent proliferation. Permethrin, benzyl benzoate, crotamiton, lindane, and sulfur have also been used. Metronidazole is a common treatment for demodicosis, as was used in our patient for several weeks until the lesions cleared. Systemic metronidazole therapy may be indicated for reducing Demodex spp. density. Severe cases, particularly in immunocompromised individuals, may require oral ivermectin. Appropriate hygiene is important for prevention, such as washing the face with non-oily cleansers and laundering linens regularly.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Mr. Lee is a medical student at the University of California San Diego.
Suggested Reading
Chudzicka-Strugała I et al. Demodicosis in different age groups and alternative treatment options—A review. J Clin Med. 2023 Feb 19;12(4):1649. doi: 10.3390/jcm12041649.
Eichenfield DZ et al. Management of acne vulgaris: A review. JAMA. 2021 Nov 23;326(20):2055-2067. doi: 10.1001/jama.2021.17633.
Sharma A et al. Rosacea management: A comprehensive review. J Cosmet Dermatol. 2022 May;21(5):1895-1904. doi: 10.1111/jocd.14816.
Taudorf EH et al. Cutaneous candidiasis — an evidence-based review of topical and systemic treatments to inform clinical practice. J Eur Acad Dermatol Venereol. 2019 Oct;33(10):1863-1873. doi: 10.1111/jdv.15782.
Winters RD, Mitchell M. Folliculitis. [Updated 2023 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547754/
A 7-year-old female presents with persistent pimples on the nose and cheeks for approximately 1 year. She had been treated with several topical antibiotics and acne washes without resolution of the lesions. There were no signs of early puberty, and the child had no history of medical conditions. Her mother has a history of rosacea. Physical examination revealed erythematous papules on the nose and cheeks bilaterally.
Cosmetic Tattoo Ingredients Associated With Contact Dermatitis
TOPLINE:
, but the ability to identify these allergies in patients is limited.
METHODOLOGY:
- While the allergenic potential of pigments in traditional tattoos has been documented, there is less clarity about pigments used in inks contained in cosmetic tattoos, also known as permanent makeup, and their association with ACD.
- Researchers conducted an Internet search and identified 974 individual permanent makeup ink products sold in the United States and also identified 79 unique pigments in those products.
- They evaluated the safety data sheets of these products and performed a PubMed search to identify documented ACD cases related to these pigments.
TAKEAWAY:
- Of the 79 pigments, 20 contained inorganic metals, which included iron, aluminum, silicone, chromium, copper, titanium, molybdenum, and manganese.
- Organic pigments were more common: 59 of the remaining pigments were organic compounds, mostly azo, quinacridone, or anthraquinone dyes, including 4 black pigments made from carbon only.
- A literature search identified 29 cases where patients had developed ACD thought to be caused by at least one of the 79 pigments identified by the authors of the current study and included 10 of the 79 pigments (12%).
- In 18 of the 29 cases in the literature, patch testing to the suspected pigment had been performed; in 3 cases, ACD was suspected without confirmatory testing.
IN PRACTICE:
Permanent makeup is becoming more popular, and there have been reports of ACD related to pigments contained in the inks, the authors wrote. “Traditional patch testing methods may not be useful in confirming the presence of a pigment allergy, even if one is suspect,” they added. “Consumers and patch testing physicians would benefit from better labeling of tattoo inks and the development of protocols designed to specifically test for tattoo pigment allergies.”
SOURCE:
The study was led by Sarah Rigali, MS, of Rosalind Franklin University, Chicago Medical School, Chicago, and coauthors from the Department of Dermatology, Northwestern University, Chicago, published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The study is limited by incomplete safety data sheets. So, many brands of permanent makeup ink could not be investigated. In addition, some pigments may not be fully disclosed in ingredient lists and precise ink content measurements were not available.
DISCLOSURES:
The study reported receiving no funding. The authors declared no conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
, but the ability to identify these allergies in patients is limited.
METHODOLOGY:
- While the allergenic potential of pigments in traditional tattoos has been documented, there is less clarity about pigments used in inks contained in cosmetic tattoos, also known as permanent makeup, and their association with ACD.
- Researchers conducted an Internet search and identified 974 individual permanent makeup ink products sold in the United States and also identified 79 unique pigments in those products.
- They evaluated the safety data sheets of these products and performed a PubMed search to identify documented ACD cases related to these pigments.
TAKEAWAY:
- Of the 79 pigments, 20 contained inorganic metals, which included iron, aluminum, silicone, chromium, copper, titanium, molybdenum, and manganese.
- Organic pigments were more common: 59 of the remaining pigments were organic compounds, mostly azo, quinacridone, or anthraquinone dyes, including 4 black pigments made from carbon only.
- A literature search identified 29 cases where patients had developed ACD thought to be caused by at least one of the 79 pigments identified by the authors of the current study and included 10 of the 79 pigments (12%).
- In 18 of the 29 cases in the literature, patch testing to the suspected pigment had been performed; in 3 cases, ACD was suspected without confirmatory testing.
IN PRACTICE:
Permanent makeup is becoming more popular, and there have been reports of ACD related to pigments contained in the inks, the authors wrote. “Traditional patch testing methods may not be useful in confirming the presence of a pigment allergy, even if one is suspect,” they added. “Consumers and patch testing physicians would benefit from better labeling of tattoo inks and the development of protocols designed to specifically test for tattoo pigment allergies.”
SOURCE:
The study was led by Sarah Rigali, MS, of Rosalind Franklin University, Chicago Medical School, Chicago, and coauthors from the Department of Dermatology, Northwestern University, Chicago, published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The study is limited by incomplete safety data sheets. So, many brands of permanent makeup ink could not be investigated. In addition, some pigments may not be fully disclosed in ingredient lists and precise ink content measurements were not available.
DISCLOSURES:
The study reported receiving no funding. The authors declared no conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
, but the ability to identify these allergies in patients is limited.
METHODOLOGY:
- While the allergenic potential of pigments in traditional tattoos has been documented, there is less clarity about pigments used in inks contained in cosmetic tattoos, also known as permanent makeup, and their association with ACD.
- Researchers conducted an Internet search and identified 974 individual permanent makeup ink products sold in the United States and also identified 79 unique pigments in those products.
- They evaluated the safety data sheets of these products and performed a PubMed search to identify documented ACD cases related to these pigments.
TAKEAWAY:
- Of the 79 pigments, 20 contained inorganic metals, which included iron, aluminum, silicone, chromium, copper, titanium, molybdenum, and manganese.
- Organic pigments were more common: 59 of the remaining pigments were organic compounds, mostly azo, quinacridone, or anthraquinone dyes, including 4 black pigments made from carbon only.
- A literature search identified 29 cases where patients had developed ACD thought to be caused by at least one of the 79 pigments identified by the authors of the current study and included 10 of the 79 pigments (12%).
- In 18 of the 29 cases in the literature, patch testing to the suspected pigment had been performed; in 3 cases, ACD was suspected without confirmatory testing.
IN PRACTICE:
Permanent makeup is becoming more popular, and there have been reports of ACD related to pigments contained in the inks, the authors wrote. “Traditional patch testing methods may not be useful in confirming the presence of a pigment allergy, even if one is suspect,” they added. “Consumers and patch testing physicians would benefit from better labeling of tattoo inks and the development of protocols designed to specifically test for tattoo pigment allergies.”
SOURCE:
The study was led by Sarah Rigali, MS, of Rosalind Franklin University, Chicago Medical School, Chicago, and coauthors from the Department of Dermatology, Northwestern University, Chicago, published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The study is limited by incomplete safety data sheets. So, many brands of permanent makeup ink could not be investigated. In addition, some pigments may not be fully disclosed in ingredient lists and precise ink content measurements were not available.
DISCLOSURES:
The study reported receiving no funding. The authors declared no conflicts of interest.
A version of this article appeared on Medscape.com.
Study Highlights Melanoma Survival Disparities in Rural vs Urban Settings
, results from an analysis of data from the National Cancer Institute showed.
“Melanoma is currently the fifth most common malignancy in the United States, with approximately 106,000 new cases and 7180 reported deaths occurring in 2021,” the study’s first author, Mitchell Taylor, MD, a dermatology research fellow at the University of Nebraska, Omaha, and colleagues wrote in the abstract, which was presented during a poster session at the annual meeting of the Society for Investigative Dermatology. “Rural areas have been shown to bear a higher melanoma disease burden, yet there is a paucity of national-level studies examining these disparities.”
To characterize the rural population diagnosed with cutaneous melanoma and assess associated disparities in the United States, the researchers queried the NCI’s Surveillance, Epidemiology, and End Results database to identify individuals diagnosed with cutaneous melanoma from 2000 to 2020 (International Classification of Diseases, 3rd Edition, 8720/3 — 8780/3; Primary Site codes C44.0-C44.9). They drew from US Office of Management and Budget terminology to define and categorize rural and urban communities.
Among 391,047 patients included during the study period, binary logistic regression analysis revealed that patients in rural areas had a greater odds of being older, from ages 50 to 75 years (odds ratio [OR], 1.10; P < .001); had annual incomes < $70,000 (OR, 16.80; P < .001); had tumors located on the head and neck (OR, 1.24; P < .001); and presented with regional/distant disease (OR, 1.13; P < .001).
As for disease-specific survival, patients living in rural areas had significantly reduced survival compared with those living in urban areas (a mean of 207.3 vs 216.3 months, respectively; P < .001). Multivariate Cox regression revealed that living in a rural setting was significantly associated with reduced disease-specific survival (hazard ratio [HR], 1.10; P < .001), as was having head and neck tumors (HR, 1.41; P < .001).“Overall, this study underscores a significant decrease in disease-specific survival among rural patients diagnosed with cutaneous melanoma and establishes a significant association between rural living and high-risk primary tumor locations, particularly the head and neck,” the authors concluded.
Lucinda Kohn, MD, assistant professor of dermatology in the Centers for American Indian and Alaska Native Health at the University of Colorado at Denver, Aurora, Colorado, who was asked to comment on the results, said the findings echo the results of a recent study which characterized melanoma rates among non-Hispanic American Indian/Alaska Native individuals from 1999 to 2019.
“I suspect this decreased disease-specific survival highlights the issues our rural-residing patients face with access to dermatology care,” Dr. Kohn told this news organization. “Dermatologists are able to detect thinner melanomas than patients [and] are preferentially concentrated in metropolitan areas. Dermatologists are also the most skilled and knowledgeable to screen, diagnose, and manage melanomas. Having fewer dermatologists in rural areas impedes melanoma care for our rural-residing patients.”
Neither the researchers nor Dr. Kohn reported any relevant disclosures.
A version of this article first appeared on Medscape.com.
, results from an analysis of data from the National Cancer Institute showed.
“Melanoma is currently the fifth most common malignancy in the United States, with approximately 106,000 new cases and 7180 reported deaths occurring in 2021,” the study’s first author, Mitchell Taylor, MD, a dermatology research fellow at the University of Nebraska, Omaha, and colleagues wrote in the abstract, which was presented during a poster session at the annual meeting of the Society for Investigative Dermatology. “Rural areas have been shown to bear a higher melanoma disease burden, yet there is a paucity of national-level studies examining these disparities.”
To characterize the rural population diagnosed with cutaneous melanoma and assess associated disparities in the United States, the researchers queried the NCI’s Surveillance, Epidemiology, and End Results database to identify individuals diagnosed with cutaneous melanoma from 2000 to 2020 (International Classification of Diseases, 3rd Edition, 8720/3 — 8780/3; Primary Site codes C44.0-C44.9). They drew from US Office of Management and Budget terminology to define and categorize rural and urban communities.
Among 391,047 patients included during the study period, binary logistic regression analysis revealed that patients in rural areas had a greater odds of being older, from ages 50 to 75 years (odds ratio [OR], 1.10; P < .001); had annual incomes < $70,000 (OR, 16.80; P < .001); had tumors located on the head and neck (OR, 1.24; P < .001); and presented with regional/distant disease (OR, 1.13; P < .001).
As for disease-specific survival, patients living in rural areas had significantly reduced survival compared with those living in urban areas (a mean of 207.3 vs 216.3 months, respectively; P < .001). Multivariate Cox regression revealed that living in a rural setting was significantly associated with reduced disease-specific survival (hazard ratio [HR], 1.10; P < .001), as was having head and neck tumors (HR, 1.41; P < .001).“Overall, this study underscores a significant decrease in disease-specific survival among rural patients diagnosed with cutaneous melanoma and establishes a significant association between rural living and high-risk primary tumor locations, particularly the head and neck,” the authors concluded.
Lucinda Kohn, MD, assistant professor of dermatology in the Centers for American Indian and Alaska Native Health at the University of Colorado at Denver, Aurora, Colorado, who was asked to comment on the results, said the findings echo the results of a recent study which characterized melanoma rates among non-Hispanic American Indian/Alaska Native individuals from 1999 to 2019.
“I suspect this decreased disease-specific survival highlights the issues our rural-residing patients face with access to dermatology care,” Dr. Kohn told this news organization. “Dermatologists are able to detect thinner melanomas than patients [and] are preferentially concentrated in metropolitan areas. Dermatologists are also the most skilled and knowledgeable to screen, diagnose, and manage melanomas. Having fewer dermatologists in rural areas impedes melanoma care for our rural-residing patients.”
Neither the researchers nor Dr. Kohn reported any relevant disclosures.
A version of this article first appeared on Medscape.com.
, results from an analysis of data from the National Cancer Institute showed.
“Melanoma is currently the fifth most common malignancy in the United States, with approximately 106,000 new cases and 7180 reported deaths occurring in 2021,” the study’s first author, Mitchell Taylor, MD, a dermatology research fellow at the University of Nebraska, Omaha, and colleagues wrote in the abstract, which was presented during a poster session at the annual meeting of the Society for Investigative Dermatology. “Rural areas have been shown to bear a higher melanoma disease burden, yet there is a paucity of national-level studies examining these disparities.”
To characterize the rural population diagnosed with cutaneous melanoma and assess associated disparities in the United States, the researchers queried the NCI’s Surveillance, Epidemiology, and End Results database to identify individuals diagnosed with cutaneous melanoma from 2000 to 2020 (International Classification of Diseases, 3rd Edition, 8720/3 — 8780/3; Primary Site codes C44.0-C44.9). They drew from US Office of Management and Budget terminology to define and categorize rural and urban communities.
Among 391,047 patients included during the study period, binary logistic regression analysis revealed that patients in rural areas had a greater odds of being older, from ages 50 to 75 years (odds ratio [OR], 1.10; P < .001); had annual incomes < $70,000 (OR, 16.80; P < .001); had tumors located on the head and neck (OR, 1.24; P < .001); and presented with regional/distant disease (OR, 1.13; P < .001).
As for disease-specific survival, patients living in rural areas had significantly reduced survival compared with those living in urban areas (a mean of 207.3 vs 216.3 months, respectively; P < .001). Multivariate Cox regression revealed that living in a rural setting was significantly associated with reduced disease-specific survival (hazard ratio [HR], 1.10; P < .001), as was having head and neck tumors (HR, 1.41; P < .001).“Overall, this study underscores a significant decrease in disease-specific survival among rural patients diagnosed with cutaneous melanoma and establishes a significant association between rural living and high-risk primary tumor locations, particularly the head and neck,” the authors concluded.
Lucinda Kohn, MD, assistant professor of dermatology in the Centers for American Indian and Alaska Native Health at the University of Colorado at Denver, Aurora, Colorado, who was asked to comment on the results, said the findings echo the results of a recent study which characterized melanoma rates among non-Hispanic American Indian/Alaska Native individuals from 1999 to 2019.
“I suspect this decreased disease-specific survival highlights the issues our rural-residing patients face with access to dermatology care,” Dr. Kohn told this news organization. “Dermatologists are able to detect thinner melanomas than patients [and] are preferentially concentrated in metropolitan areas. Dermatologists are also the most skilled and knowledgeable to screen, diagnose, and manage melanomas. Having fewer dermatologists in rural areas impedes melanoma care for our rural-residing patients.”
Neither the researchers nor Dr. Kohn reported any relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM SID 2024