User login
More Illnesses Possible Related Linked to Counterfeit Botulinum Toxin Reported
— two in the intensive care unit. None of the cases required intubation, according to an announcement of an investigation into these reports in by the Centers for Disease Control and Prevention (CDC).
The report, published online in the Morbidity and Mortality Weekly Report, notes that the four patients in Tennessee received counterfeit BoNT, while product information was not available for the three cases in New York City. “However, one person reported paying less than US wholesale acquisition cost for the administered product, and another reported that the product had been purchased overseas,” the authors of the report wrote. The development underscores that BoNT injections “should be administered only by licensed and trained providers using recommended doses of FDA [Food and Drug Admininstration]-approved products.”
This report follows a CDC advisory published in April 2024 of at least 22 people from 11 states who reported serious reactions after receiving botulinum toxin injections from unlicensed or untrained individuals or in nonhealthcare settings, such as homes and spas.
The median age of the women in the July report was 48 years, and signs and symptoms included ptosis, dry mouth, dysphagia, shortness of breath, and weakness. Onset occurred between February 23 and March 7, 2024.
“This investigation did not determine why these illnesses occurred after cosmetic BoNT injections; potential reasons might include use of counterfeit BoNT, which might be more potent or contain harmful additional ingredients or higher susceptibility to BoNT effects among some persons,” the investigators wrote. They recommended further studies to describe the clinical spectrum of cosmetic BoNT injection effects such as severity of signs and symptoms.
For cases of suspected systemic botulism, the CDC recommends calling the local or state health department for consultation and antitoxin release (as well as information on reporting adverse events). Alternatively, the 24/7 phone number for the CDC clinical botulism service is 770-488-7100.
A version of this article first appeared on Medscape.com.
— two in the intensive care unit. None of the cases required intubation, according to an announcement of an investigation into these reports in by the Centers for Disease Control and Prevention (CDC).
The report, published online in the Morbidity and Mortality Weekly Report, notes that the four patients in Tennessee received counterfeit BoNT, while product information was not available for the three cases in New York City. “However, one person reported paying less than US wholesale acquisition cost for the administered product, and another reported that the product had been purchased overseas,” the authors of the report wrote. The development underscores that BoNT injections “should be administered only by licensed and trained providers using recommended doses of FDA [Food and Drug Admininstration]-approved products.”
This report follows a CDC advisory published in April 2024 of at least 22 people from 11 states who reported serious reactions after receiving botulinum toxin injections from unlicensed or untrained individuals or in nonhealthcare settings, such as homes and spas.
The median age of the women in the July report was 48 years, and signs and symptoms included ptosis, dry mouth, dysphagia, shortness of breath, and weakness. Onset occurred between February 23 and March 7, 2024.
“This investigation did not determine why these illnesses occurred after cosmetic BoNT injections; potential reasons might include use of counterfeit BoNT, which might be more potent or contain harmful additional ingredients or higher susceptibility to BoNT effects among some persons,” the investigators wrote. They recommended further studies to describe the clinical spectrum of cosmetic BoNT injection effects such as severity of signs and symptoms.
For cases of suspected systemic botulism, the CDC recommends calling the local or state health department for consultation and antitoxin release (as well as information on reporting adverse events). Alternatively, the 24/7 phone number for the CDC clinical botulism service is 770-488-7100.
A version of this article first appeared on Medscape.com.
— two in the intensive care unit. None of the cases required intubation, according to an announcement of an investigation into these reports in by the Centers for Disease Control and Prevention (CDC).
The report, published online in the Morbidity and Mortality Weekly Report, notes that the four patients in Tennessee received counterfeit BoNT, while product information was not available for the three cases in New York City. “However, one person reported paying less than US wholesale acquisition cost for the administered product, and another reported that the product had been purchased overseas,” the authors of the report wrote. The development underscores that BoNT injections “should be administered only by licensed and trained providers using recommended doses of FDA [Food and Drug Admininstration]-approved products.”
This report follows a CDC advisory published in April 2024 of at least 22 people from 11 states who reported serious reactions after receiving botulinum toxin injections from unlicensed or untrained individuals or in nonhealthcare settings, such as homes and spas.
The median age of the women in the July report was 48 years, and signs and symptoms included ptosis, dry mouth, dysphagia, shortness of breath, and weakness. Onset occurred between February 23 and March 7, 2024.
“This investigation did not determine why these illnesses occurred after cosmetic BoNT injections; potential reasons might include use of counterfeit BoNT, which might be more potent or contain harmful additional ingredients or higher susceptibility to BoNT effects among some persons,” the investigators wrote. They recommended further studies to describe the clinical spectrum of cosmetic BoNT injection effects such as severity of signs and symptoms.
For cases of suspected systemic botulism, the CDC recommends calling the local or state health department for consultation and antitoxin release (as well as information on reporting adverse events). Alternatively, the 24/7 phone number for the CDC clinical botulism service is 770-488-7100.
A version of this article first appeared on Medscape.com.
FROM THE MMWR
Study Finds Potential benefits of Spironolactone for Women with HS
TOPLINE:
according to the results of a single-center retrospective study.
METHODOLOGY:
- This retrospective study included 157 women (median age, 36.5 years) with HS who received spironolactone for at least 3 months between 2000 and 2021 at Michigan Medicine outpatient dermatology clinics. The majority of patients were White (59%) or Black (37%) individuals.
- The median prescribed dose was 100 mg/d, the most common dose was 50-100 mg/d, and the median time spironolactone was initiated was 8.8 years after HS was diagnosed.
- Improvement status was classified on the basis of objective clinician assessments, including documented reductions in the lesion count, pain, and symptoms.
TAKEAWAY:
- Overall, 31 patients (20%) showed improvements with spironolactone treatment.
- A shorter duration between the diagnosis of HS and the initiation of spironolactone was associated with improvement (P = .047).
- Axillary involvement (P = .003), the use of intralesional steroids (P = .015), previous treatments (P = .023), and previous treatment failures (P = .030) were linked to a lack of improvement with spironolactone.
- Patients with Hurley stage III were 85% less likely to experience improvement with spironolactone (P = .036).
IN PRACTICE:
Spironolactone, which has antiandrogenic properties, “may be beneficial for patients with mild HS, notably those at Hurley stage I if implemented early as a primary or ancillary treatment,” the authors concluded, adding that prospective, multicenter studies are needed to “elucidate further the safety and efficacy of spironolactone for treating HS.”
SOURCE:
The study was led by Suma V. Gangidi, BS, Carle Illinois College of Medicine, University of Illinois Urbana-Champaign, and was published online in the International Journal of Women’s Dermatology.
LIMITATIONS:
Retrospective design can introduce inherent biases, including the potential for missing or misclassified data. Additionally, the study was conducted at a single center, which may limit the generalizability, and findings were also limited by lack of standardized objective measures for assessing treatment improvement, presence of confounding variables from concomitant treatments, small sample size, and potential multicollinearity.
DISCLOSURES:
This study did not receive any funding. One author declared ties with various pharmaceutical companies. The other authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
according to the results of a single-center retrospective study.
METHODOLOGY:
- This retrospective study included 157 women (median age, 36.5 years) with HS who received spironolactone for at least 3 months between 2000 and 2021 at Michigan Medicine outpatient dermatology clinics. The majority of patients were White (59%) or Black (37%) individuals.
- The median prescribed dose was 100 mg/d, the most common dose was 50-100 mg/d, and the median time spironolactone was initiated was 8.8 years after HS was diagnosed.
- Improvement status was classified on the basis of objective clinician assessments, including documented reductions in the lesion count, pain, and symptoms.
TAKEAWAY:
- Overall, 31 patients (20%) showed improvements with spironolactone treatment.
- A shorter duration between the diagnosis of HS and the initiation of spironolactone was associated with improvement (P = .047).
- Axillary involvement (P = .003), the use of intralesional steroids (P = .015), previous treatments (P = .023), and previous treatment failures (P = .030) were linked to a lack of improvement with spironolactone.
- Patients with Hurley stage III were 85% less likely to experience improvement with spironolactone (P = .036).
IN PRACTICE:
Spironolactone, which has antiandrogenic properties, “may be beneficial for patients with mild HS, notably those at Hurley stage I if implemented early as a primary or ancillary treatment,” the authors concluded, adding that prospective, multicenter studies are needed to “elucidate further the safety and efficacy of spironolactone for treating HS.”
SOURCE:
The study was led by Suma V. Gangidi, BS, Carle Illinois College of Medicine, University of Illinois Urbana-Champaign, and was published online in the International Journal of Women’s Dermatology.
LIMITATIONS:
Retrospective design can introduce inherent biases, including the potential for missing or misclassified data. Additionally, the study was conducted at a single center, which may limit the generalizability, and findings were also limited by lack of standardized objective measures for assessing treatment improvement, presence of confounding variables from concomitant treatments, small sample size, and potential multicollinearity.
DISCLOSURES:
This study did not receive any funding. One author declared ties with various pharmaceutical companies. The other authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
according to the results of a single-center retrospective study.
METHODOLOGY:
- This retrospective study included 157 women (median age, 36.5 years) with HS who received spironolactone for at least 3 months between 2000 and 2021 at Michigan Medicine outpatient dermatology clinics. The majority of patients were White (59%) or Black (37%) individuals.
- The median prescribed dose was 100 mg/d, the most common dose was 50-100 mg/d, and the median time spironolactone was initiated was 8.8 years after HS was diagnosed.
- Improvement status was classified on the basis of objective clinician assessments, including documented reductions in the lesion count, pain, and symptoms.
TAKEAWAY:
- Overall, 31 patients (20%) showed improvements with spironolactone treatment.
- A shorter duration between the diagnosis of HS and the initiation of spironolactone was associated with improvement (P = .047).
- Axillary involvement (P = .003), the use of intralesional steroids (P = .015), previous treatments (P = .023), and previous treatment failures (P = .030) were linked to a lack of improvement with spironolactone.
- Patients with Hurley stage III were 85% less likely to experience improvement with spironolactone (P = .036).
IN PRACTICE:
Spironolactone, which has antiandrogenic properties, “may be beneficial for patients with mild HS, notably those at Hurley stage I if implemented early as a primary or ancillary treatment,” the authors concluded, adding that prospective, multicenter studies are needed to “elucidate further the safety and efficacy of spironolactone for treating HS.”
SOURCE:
The study was led by Suma V. Gangidi, BS, Carle Illinois College of Medicine, University of Illinois Urbana-Champaign, and was published online in the International Journal of Women’s Dermatology.
LIMITATIONS:
Retrospective design can introduce inherent biases, including the potential for missing or misclassified data. Additionally, the study was conducted at a single center, which may limit the generalizability, and findings were also limited by lack of standardized objective measures for assessing treatment improvement, presence of confounding variables from concomitant treatments, small sample size, and potential multicollinearity.
DISCLOSURES:
This study did not receive any funding. One author declared ties with various pharmaceutical companies. The other authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Studies Show Dupilumab Effects In Children with Both Atopic Dermatitis and Alopecia
TOPLINE:
(AA) in a review.
METHODOLOGY:
- Researchers conducted a scoping review of seven studies, a result of a MEDLINE and Embase search on March 1, 2024, which included 31 patients aged 4-17 years with both AD and AA (average age, 11.4 years; 64.5% women).
- The review included four case reports, two case series, and one retrospective chart review.
- Patients had an average duration of AA and AD of 3.31 years and 5.33 years, respectively, before starting dupilumab.
- The type of AA was listed in 22 patients; among these patients, alopecia universalis was the most common (50%), followed by alopecia ophiasis (22.7%), patchy alopecia (18.2%), and alopecia totalis (9.09%).
TAKEAWAY:
- Overall, 77.4% of patients in the trials achieved hair regrowth with dupilumab treatment with a mean 42.6 reduction in SALT score (measuring scalp hair loss on a scale of 0-100) over an average of 3.21 months (P < .01).
- Severity of AD was reduced by an average of 2.14 units to an average of 0.857 (clear or almost clear AD; P < .01) on the AD Investigator Global Assessment dropping from an average of 3 (severe disease) before treatment.
- There were no characteristics that significantly distinguished patients with AA who responded to treatment from those who did not.
- Four patients reported worsening of preexisting AA after starting dupilumab; two of these continued dupilumab and showed improvement at subsequent follow-ups.
IN PRACTICE:
“Our review highlights the efficacy of dupilumab in pediatric AA with concurrent AD,” wrote the authors, noting that “the exact mechanism for this efficacy remains speculative.” Although there have been reports of new or worsening AA with dupilumab, they added, its “favorable safety profile in pediatrics enhances its appeal for AA treatment, as monotherapy or in combination with other AA medications.”
SOURCE:
The study was led by Dea Metko, Michael G. DeGroote School of Medicine in Hamilton, Ontario, Canada. It was published online on July 4, 2024, in Pediatric Dermatology.
LIMITATIONS:
Potential publication bias, inconsistent data reporting, the small number of patients, and short follow-up duration were the main limitations of this study.
DISCLOSURES:
The study funding source was not disclosed. One author received honoraria outside this work. Other authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
(AA) in a review.
METHODOLOGY:
- Researchers conducted a scoping review of seven studies, a result of a MEDLINE and Embase search on March 1, 2024, which included 31 patients aged 4-17 years with both AD and AA (average age, 11.4 years; 64.5% women).
- The review included four case reports, two case series, and one retrospective chart review.
- Patients had an average duration of AA and AD of 3.31 years and 5.33 years, respectively, before starting dupilumab.
- The type of AA was listed in 22 patients; among these patients, alopecia universalis was the most common (50%), followed by alopecia ophiasis (22.7%), patchy alopecia (18.2%), and alopecia totalis (9.09%).
TAKEAWAY:
- Overall, 77.4% of patients in the trials achieved hair regrowth with dupilumab treatment with a mean 42.6 reduction in SALT score (measuring scalp hair loss on a scale of 0-100) over an average of 3.21 months (P < .01).
- Severity of AD was reduced by an average of 2.14 units to an average of 0.857 (clear or almost clear AD; P < .01) on the AD Investigator Global Assessment dropping from an average of 3 (severe disease) before treatment.
- There were no characteristics that significantly distinguished patients with AA who responded to treatment from those who did not.
- Four patients reported worsening of preexisting AA after starting dupilumab; two of these continued dupilumab and showed improvement at subsequent follow-ups.
IN PRACTICE:
“Our review highlights the efficacy of dupilumab in pediatric AA with concurrent AD,” wrote the authors, noting that “the exact mechanism for this efficacy remains speculative.” Although there have been reports of new or worsening AA with dupilumab, they added, its “favorable safety profile in pediatrics enhances its appeal for AA treatment, as monotherapy or in combination with other AA medications.”
SOURCE:
The study was led by Dea Metko, Michael G. DeGroote School of Medicine in Hamilton, Ontario, Canada. It was published online on July 4, 2024, in Pediatric Dermatology.
LIMITATIONS:
Potential publication bias, inconsistent data reporting, the small number of patients, and short follow-up duration were the main limitations of this study.
DISCLOSURES:
The study funding source was not disclosed. One author received honoraria outside this work. Other authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
(AA) in a review.
METHODOLOGY:
- Researchers conducted a scoping review of seven studies, a result of a MEDLINE and Embase search on March 1, 2024, which included 31 patients aged 4-17 years with both AD and AA (average age, 11.4 years; 64.5% women).
- The review included four case reports, two case series, and one retrospective chart review.
- Patients had an average duration of AA and AD of 3.31 years and 5.33 years, respectively, before starting dupilumab.
- The type of AA was listed in 22 patients; among these patients, alopecia universalis was the most common (50%), followed by alopecia ophiasis (22.7%), patchy alopecia (18.2%), and alopecia totalis (9.09%).
TAKEAWAY:
- Overall, 77.4% of patients in the trials achieved hair regrowth with dupilumab treatment with a mean 42.6 reduction in SALT score (measuring scalp hair loss on a scale of 0-100) over an average of 3.21 months (P < .01).
- Severity of AD was reduced by an average of 2.14 units to an average of 0.857 (clear or almost clear AD; P < .01) on the AD Investigator Global Assessment dropping from an average of 3 (severe disease) before treatment.
- There were no characteristics that significantly distinguished patients with AA who responded to treatment from those who did not.
- Four patients reported worsening of preexisting AA after starting dupilumab; two of these continued dupilumab and showed improvement at subsequent follow-ups.
IN PRACTICE:
“Our review highlights the efficacy of dupilumab in pediatric AA with concurrent AD,” wrote the authors, noting that “the exact mechanism for this efficacy remains speculative.” Although there have been reports of new or worsening AA with dupilumab, they added, its “favorable safety profile in pediatrics enhances its appeal for AA treatment, as monotherapy or in combination with other AA medications.”
SOURCE:
The study was led by Dea Metko, Michael G. DeGroote School of Medicine in Hamilton, Ontario, Canada. It was published online on July 4, 2024, in Pediatric Dermatology.
LIMITATIONS:
Potential publication bias, inconsistent data reporting, the small number of patients, and short follow-up duration were the main limitations of this study.
DISCLOSURES:
The study funding source was not disclosed. One author received honoraria outside this work. Other authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Study Estimates Global Prevalence of Seborrheic Dermatitis
TOPLINE:
, according to a meta-analysis that also found a higher prevalence in adults than in children.
METHODOLOGY:
- Researchers conducted a meta-analysis of 121 studies, which included 1,260,163 people with clinician-diagnosed seborrheic dermatitis.
- The included studies represented nine countries; most were from India (n = 18), Turkey (n = 13), and the United States (n = 8).
- The primary outcome was the pooled prevalence of seborrheic dermatitis.
TAKEAWAY:
- The overall pooled prevalence of seborrheic dermatitis was 4.38%, 4.08% in clinical settings, and 4.71% in the studies conducted in the general population.
- The prevalence of seborrheic dermatitis was higher among adults (5.64%) than in children (3.7%) and neonates (0.23%).
- A significant variation was observed across countries, with South Africa having the highest prevalence at 8.82%, followed by the United States at 5.86% and Turkey at 3.74%, while India had the lowest prevalence at 2.62%.
IN PRACTICE:
The global prevalence in this meta-analysis was “higher than previous large-scale global estimates, with notable geographic and sociodemographic variability, highlighting the potential impact of environmental factors and cultural practices,” the authors wrote.
SOURCE:
The study was led by Meredith Tyree Polaskey, MS, Chicago Medical School, Rosalind Franklin University of Medicine and Science, North Chicago, Illinois, and was published online on July 3, 2024, in the JAMA Dermatology.
LIMITATIONS:
Interpretation of the findings is limited by research gaps in Central Asia, much of Sub-Saharan Africa, Eastern Europe, Southeast Asia, Latin America (excluding Brazil), and the Caribbean, along with potential underreporting in regions with restricted healthcare access and significant heterogeneity across studies.
DISCLOSURES:
Funding information was not available. One author reported serving as an advisor, consultant, speaker, and/or investigator for multiple pharmaceutical companies, including AbbVie, Amgen, and Pfizer.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
, according to a meta-analysis that also found a higher prevalence in adults than in children.
METHODOLOGY:
- Researchers conducted a meta-analysis of 121 studies, which included 1,260,163 people with clinician-diagnosed seborrheic dermatitis.
- The included studies represented nine countries; most were from India (n = 18), Turkey (n = 13), and the United States (n = 8).
- The primary outcome was the pooled prevalence of seborrheic dermatitis.
TAKEAWAY:
- The overall pooled prevalence of seborrheic dermatitis was 4.38%, 4.08% in clinical settings, and 4.71% in the studies conducted in the general population.
- The prevalence of seborrheic dermatitis was higher among adults (5.64%) than in children (3.7%) and neonates (0.23%).
- A significant variation was observed across countries, with South Africa having the highest prevalence at 8.82%, followed by the United States at 5.86% and Turkey at 3.74%, while India had the lowest prevalence at 2.62%.
IN PRACTICE:
The global prevalence in this meta-analysis was “higher than previous large-scale global estimates, with notable geographic and sociodemographic variability, highlighting the potential impact of environmental factors and cultural practices,” the authors wrote.
SOURCE:
The study was led by Meredith Tyree Polaskey, MS, Chicago Medical School, Rosalind Franklin University of Medicine and Science, North Chicago, Illinois, and was published online on July 3, 2024, in the JAMA Dermatology.
LIMITATIONS:
Interpretation of the findings is limited by research gaps in Central Asia, much of Sub-Saharan Africa, Eastern Europe, Southeast Asia, Latin America (excluding Brazil), and the Caribbean, along with potential underreporting in regions with restricted healthcare access and significant heterogeneity across studies.
DISCLOSURES:
Funding information was not available. One author reported serving as an advisor, consultant, speaker, and/or investigator for multiple pharmaceutical companies, including AbbVie, Amgen, and Pfizer.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
, according to a meta-analysis that also found a higher prevalence in adults than in children.
METHODOLOGY:
- Researchers conducted a meta-analysis of 121 studies, which included 1,260,163 people with clinician-diagnosed seborrheic dermatitis.
- The included studies represented nine countries; most were from India (n = 18), Turkey (n = 13), and the United States (n = 8).
- The primary outcome was the pooled prevalence of seborrheic dermatitis.
TAKEAWAY:
- The overall pooled prevalence of seborrheic dermatitis was 4.38%, 4.08% in clinical settings, and 4.71% in the studies conducted in the general population.
- The prevalence of seborrheic dermatitis was higher among adults (5.64%) than in children (3.7%) and neonates (0.23%).
- A significant variation was observed across countries, with South Africa having the highest prevalence at 8.82%, followed by the United States at 5.86% and Turkey at 3.74%, while India had the lowest prevalence at 2.62%.
IN PRACTICE:
The global prevalence in this meta-analysis was “higher than previous large-scale global estimates, with notable geographic and sociodemographic variability, highlighting the potential impact of environmental factors and cultural practices,” the authors wrote.
SOURCE:
The study was led by Meredith Tyree Polaskey, MS, Chicago Medical School, Rosalind Franklin University of Medicine and Science, North Chicago, Illinois, and was published online on July 3, 2024, in the JAMA Dermatology.
LIMITATIONS:
Interpretation of the findings is limited by research gaps in Central Asia, much of Sub-Saharan Africa, Eastern Europe, Southeast Asia, Latin America (excluding Brazil), and the Caribbean, along with potential underreporting in regions with restricted healthcare access and significant heterogeneity across studies.
DISCLOSURES:
Funding information was not available. One author reported serving as an advisor, consultant, speaker, and/or investigator for multiple pharmaceutical companies, including AbbVie, Amgen, and Pfizer.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
How Common Are Life-Threatening Infections In Infants with Pustules, Vesicles?
TOPLINE:
, according to the findings from a retrospective study.
METHODOLOGY:
- Researchers reviewed the electronic medical records of infants aged ≤ 60 days who received a pediatric dermatology consultation at six US academic institutions between September 2013 and August 2019.
- Among 879 consults, 183 afebrile infants were identified as having presented with pustules, vesicles, and/or bullae.
- Infectious disease workups included blood cultures, urine cultures, lumbar punctures, and HSV testing using viral skin culture, direct immunofluorescence assay, and/or polymerase chain reaction.
- Patients were categorized by gestational age as preterm (< 37 weeks), full-term (37-42 weeks), and post-term (≥ 42 weeks).
- Overall, 67.8% of infants had pustules, 31.1% had vesicles, and 10.4% had bullae.
TAKEAWAY:
- None of the cases showed positive cerebrospinal fluid or pathogenic blood cultures. In 122 of the cases (66.6%), a noninfectious cause was diagnosed, and an infectious cause was diagnosed in 71 cases (38.8%; some patients had more than one diagnosis).
- Of the 127 newborns evaluated for HSV infection, nine (7.1%) tested positive, of whom seven (5.5%) had disease affecting the skin, eye, and mouth and were full- term infants, and two (1.6%) had disseminated HSV and were preterm infants.
- Angioinvasive fungal infection was diagnosed in five infants (2.7%), all of whom were preterm infants (< 28 weeks gestational age).
- The risk for life-threatening disease was higher in preterm infants born before 32 weeks of gestational age (P < .01) compared with those born after 32 weeks.
IN PRACTICE:
“Full-term, well-appearing, afebrile infants ≤ 60 days of age presenting with pustules or vesicles may not require full SBI [serious bacterial infection] work-up, although larger studies are needed,” the authors concluded. Testing for HSV, they added, “is recommended in all infants with vesicles, grouped pustules, or pustules accompanied by punched out or grouped erosions,” and preterm infants “should be assessed for disseminated fungal infection and HSV in the setting of fluid-filled skin lesions.”
SOURCE:
The study was led by Sonora Yun, BA, Columbia University, New York City, and was published online in Pediatrics.
LIMITATIONS:
The data were limited by the sample size and very low incidence of serious infections. Infants probably had atypical or severe presentations that warranted pediatric dermatology consultation, which may have led to overrepresentation of infectious disease rates. The study inclusion was restricted to those who received a dermatology consult; therefore, the findings may not be generalizable to outpatient primary care.
DISCLOSURES:
This study did not receive any external funding. The authors declared that they had no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
, according to the findings from a retrospective study.
METHODOLOGY:
- Researchers reviewed the electronic medical records of infants aged ≤ 60 days who received a pediatric dermatology consultation at six US academic institutions between September 2013 and August 2019.
- Among 879 consults, 183 afebrile infants were identified as having presented with pustules, vesicles, and/or bullae.
- Infectious disease workups included blood cultures, urine cultures, lumbar punctures, and HSV testing using viral skin culture, direct immunofluorescence assay, and/or polymerase chain reaction.
- Patients were categorized by gestational age as preterm (< 37 weeks), full-term (37-42 weeks), and post-term (≥ 42 weeks).
- Overall, 67.8% of infants had pustules, 31.1% had vesicles, and 10.4% had bullae.
TAKEAWAY:
- None of the cases showed positive cerebrospinal fluid or pathogenic blood cultures. In 122 of the cases (66.6%), a noninfectious cause was diagnosed, and an infectious cause was diagnosed in 71 cases (38.8%; some patients had more than one diagnosis).
- Of the 127 newborns evaluated for HSV infection, nine (7.1%) tested positive, of whom seven (5.5%) had disease affecting the skin, eye, and mouth and were full- term infants, and two (1.6%) had disseminated HSV and were preterm infants.
- Angioinvasive fungal infection was diagnosed in five infants (2.7%), all of whom were preterm infants (< 28 weeks gestational age).
- The risk for life-threatening disease was higher in preterm infants born before 32 weeks of gestational age (P < .01) compared with those born after 32 weeks.
IN PRACTICE:
“Full-term, well-appearing, afebrile infants ≤ 60 days of age presenting with pustules or vesicles may not require full SBI [serious bacterial infection] work-up, although larger studies are needed,” the authors concluded. Testing for HSV, they added, “is recommended in all infants with vesicles, grouped pustules, or pustules accompanied by punched out or grouped erosions,” and preterm infants “should be assessed for disseminated fungal infection and HSV in the setting of fluid-filled skin lesions.”
SOURCE:
The study was led by Sonora Yun, BA, Columbia University, New York City, and was published online in Pediatrics.
LIMITATIONS:
The data were limited by the sample size and very low incidence of serious infections. Infants probably had atypical or severe presentations that warranted pediatric dermatology consultation, which may have led to overrepresentation of infectious disease rates. The study inclusion was restricted to those who received a dermatology consult; therefore, the findings may not be generalizable to outpatient primary care.
DISCLOSURES:
This study did not receive any external funding. The authors declared that they had no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
, according to the findings from a retrospective study.
METHODOLOGY:
- Researchers reviewed the electronic medical records of infants aged ≤ 60 days who received a pediatric dermatology consultation at six US academic institutions between September 2013 and August 2019.
- Among 879 consults, 183 afebrile infants were identified as having presented with pustules, vesicles, and/or bullae.
- Infectious disease workups included blood cultures, urine cultures, lumbar punctures, and HSV testing using viral skin culture, direct immunofluorescence assay, and/or polymerase chain reaction.
- Patients were categorized by gestational age as preterm (< 37 weeks), full-term (37-42 weeks), and post-term (≥ 42 weeks).
- Overall, 67.8% of infants had pustules, 31.1% had vesicles, and 10.4% had bullae.
TAKEAWAY:
- None of the cases showed positive cerebrospinal fluid or pathogenic blood cultures. In 122 of the cases (66.6%), a noninfectious cause was diagnosed, and an infectious cause was diagnosed in 71 cases (38.8%; some patients had more than one diagnosis).
- Of the 127 newborns evaluated for HSV infection, nine (7.1%) tested positive, of whom seven (5.5%) had disease affecting the skin, eye, and mouth and were full- term infants, and two (1.6%) had disseminated HSV and were preterm infants.
- Angioinvasive fungal infection was diagnosed in five infants (2.7%), all of whom were preterm infants (< 28 weeks gestational age).
- The risk for life-threatening disease was higher in preterm infants born before 32 weeks of gestational age (P < .01) compared with those born after 32 weeks.
IN PRACTICE:
“Full-term, well-appearing, afebrile infants ≤ 60 days of age presenting with pustules or vesicles may not require full SBI [serious bacterial infection] work-up, although larger studies are needed,” the authors concluded. Testing for HSV, they added, “is recommended in all infants with vesicles, grouped pustules, or pustules accompanied by punched out or grouped erosions,” and preterm infants “should be assessed for disseminated fungal infection and HSV in the setting of fluid-filled skin lesions.”
SOURCE:
The study was led by Sonora Yun, BA, Columbia University, New York City, and was published online in Pediatrics.
LIMITATIONS:
The data were limited by the sample size and very low incidence of serious infections. Infants probably had atypical or severe presentations that warranted pediatric dermatology consultation, which may have led to overrepresentation of infectious disease rates. The study inclusion was restricted to those who received a dermatology consult; therefore, the findings may not be generalizable to outpatient primary care.
DISCLOSURES:
This study did not receive any external funding. The authors declared that they had no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Topical PDE4 Inhibitor Now Approved for Atopic Dermatitis in Children, Adults
On July 9, the aged 6 years or older.
Roflumilast cream 0.15%, which has been developed by Arcutis Biotherapeutics and is marketed under the brand name Zoryve, is a steroid-free topical phosphodiesterase-4 inhibitor that was previously approved in a higher concentration to treat seborrheic dermatitis and plaque psoriasis.
According to a press release from Arcutis, approval for AD was supported by positive results from three phase 3 studies, a phase 2 dose-ranging study, and two phase 1 pharmacokinetic trials. In two identical phase 3 studies known as INTEGUMENT-1 and INTEGUMENT-2, about 40% of children and adults treated with roflumilast cream 0.15% achieved a Validated Investigator Global Assessment for Atopic Dermatitis score of clear (0) or almost clear (1) at week 4 (INTEGUMENT-1: 41.5% vs 25.2%; P < .0001; INTEGUMENT-2: 39% vs 16.9%; P < .0001), with significant improvement as early as week 1 (P < .0001).
Among children and adults who participated in the INTEGUMENT studies for 28 and 56 weeks, 61.3% and 65.7% achieved a 75% reduction in their Eczema Area and Severity Index scores, respectively. According to the company, there were no adverse reactions in the combined phase 3 pivotal trials that occurred in more than 2.9% of participants in either arm. The most common adverse reactions included headache (2.9%), nausea (1.9%), application-site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
The product is expected to be available commercially at the end of July 2024, according to Arcutis. Roflumilast cream 0.3% is indicated for topical treatment of plaque psoriasis, including intertriginous areas, in adult and pediatric patients aged 6 years or older; roflumilast foam 0.3% is indicated for the treatment of seborrheic dermatitis in adult and pediatric patients aged 9 years or older.
A version of this article first appeared on Medscape.com.
On July 9, the aged 6 years or older.
Roflumilast cream 0.15%, which has been developed by Arcutis Biotherapeutics and is marketed under the brand name Zoryve, is a steroid-free topical phosphodiesterase-4 inhibitor that was previously approved in a higher concentration to treat seborrheic dermatitis and plaque psoriasis.
According to a press release from Arcutis, approval for AD was supported by positive results from three phase 3 studies, a phase 2 dose-ranging study, and two phase 1 pharmacokinetic trials. In two identical phase 3 studies known as INTEGUMENT-1 and INTEGUMENT-2, about 40% of children and adults treated with roflumilast cream 0.15% achieved a Validated Investigator Global Assessment for Atopic Dermatitis score of clear (0) or almost clear (1) at week 4 (INTEGUMENT-1: 41.5% vs 25.2%; P < .0001; INTEGUMENT-2: 39% vs 16.9%; P < .0001), with significant improvement as early as week 1 (P < .0001).
Among children and adults who participated in the INTEGUMENT studies for 28 and 56 weeks, 61.3% and 65.7% achieved a 75% reduction in their Eczema Area and Severity Index scores, respectively. According to the company, there were no adverse reactions in the combined phase 3 pivotal trials that occurred in more than 2.9% of participants in either arm. The most common adverse reactions included headache (2.9%), nausea (1.9%), application-site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
The product is expected to be available commercially at the end of July 2024, according to Arcutis. Roflumilast cream 0.3% is indicated for topical treatment of plaque psoriasis, including intertriginous areas, in adult and pediatric patients aged 6 years or older; roflumilast foam 0.3% is indicated for the treatment of seborrheic dermatitis in adult and pediatric patients aged 9 years or older.
A version of this article first appeared on Medscape.com.
On July 9, the aged 6 years or older.
Roflumilast cream 0.15%, which has been developed by Arcutis Biotherapeutics and is marketed under the brand name Zoryve, is a steroid-free topical phosphodiesterase-4 inhibitor that was previously approved in a higher concentration to treat seborrheic dermatitis and plaque psoriasis.
According to a press release from Arcutis, approval for AD was supported by positive results from three phase 3 studies, a phase 2 dose-ranging study, and two phase 1 pharmacokinetic trials. In two identical phase 3 studies known as INTEGUMENT-1 and INTEGUMENT-2, about 40% of children and adults treated with roflumilast cream 0.15% achieved a Validated Investigator Global Assessment for Atopic Dermatitis score of clear (0) or almost clear (1) at week 4 (INTEGUMENT-1: 41.5% vs 25.2%; P < .0001; INTEGUMENT-2: 39% vs 16.9%; P < .0001), with significant improvement as early as week 1 (P < .0001).
Among children and adults who participated in the INTEGUMENT studies for 28 and 56 weeks, 61.3% and 65.7% achieved a 75% reduction in their Eczema Area and Severity Index scores, respectively. According to the company, there were no adverse reactions in the combined phase 3 pivotal trials that occurred in more than 2.9% of participants in either arm. The most common adverse reactions included headache (2.9%), nausea (1.9%), application-site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
The product is expected to be available commercially at the end of July 2024, according to Arcutis. Roflumilast cream 0.3% is indicated for topical treatment of plaque psoriasis, including intertriginous areas, in adult and pediatric patients aged 6 years or older; roflumilast foam 0.3% is indicated for the treatment of seborrheic dermatitis in adult and pediatric patients aged 9 years or older.
A version of this article first appeared on Medscape.com.
Dupilumab Safe, Effective Over 5 Years in Moderate to Severe Atopic Dermatitis
TOPLINE:
Over 5 years, dupilumab demonstrated acceptable safety and sustained efficacy, with significant improvements in the signs and symptoms of AD, in the treatment of moderate to severe atopic dermatitis (AD).
METHODOLOGY:
- The phase 3 multinational LIBERTY AD open-label extension study evaluated the safety and efficacy of dupilumab in 2677 adults with moderate to severe AD who had previously participated in dupilumab trials over 5 years; 334 patients (12.5%) completed treatment up to 5 years.
- Patients started with subcutaneous dupilumab, initially dosed weekly after a loading dose, then every 2 weeks in 2019.
- The primary outcomes were the incidence and rate of treatment-emergent adverse events (TEAEs).
TAKEAWAY:
- Overall, 14,717 TEAEs were reported over 5 years. The exposure-adjusted incidence rate decreased over time and was 252.48 events per 100 patient-years.
- The most common TEAEs were nasopharyngitis (28.9%), worsening AD (16.7%), upper respiratory tract infection (13.6%), conjunctivitis (10.3%), allergic conjunctivitis (9%), headache (8.1%), oral herpes (7.5%), and injection-site reactions (5.2%).
- Serious and severe TEAE rates were 10.6% and 10.0%, respectively. Exposure-adjusted incidence rates were 6.66 and 6.71 events per 100 patient-years, respectively.
- At week 260, 67.5% of patients had achieved clear or almost clear skin according to the Investigator’s Global Assessment, and 88.9% experienced a 75% or greater improvement in the Eczema Area and Severity Index.
IN PRACTICE:
“Safety and efficacy results from up to 5 years of dupilumab treatment in the LIBERTY AD open-label extension study support dupilumab as a continuous long-term treatment for adults with moderate to severe AD,” the authors concluded.
SOURCE:
The study was led by Lisa A. Beck, MD, University of Rochester, Rochester, New York, and was published online in JAMA Dermatology.
LIMITATIONS:
Study limitations included the absence of a placebo arm and treatment interruptions stemming from protocol changes. The number of patients who received biweekly doses was small. The early conclusion of the trial by the sponsor because of regulatory approval also resulted in a lower number of patients at later stages.
DISCLOSURES:
This study was funded by dupilumab manufacturers Sanofi and Regeneron Pharmaceuticals. Several authors declared ties with various pharmaceutical companies including Sanofi and Regeneron, and several authors were employees of Sanofi or Regeneron. No disclosures were reported by other authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Over 5 years, dupilumab demonstrated acceptable safety and sustained efficacy, with significant improvements in the signs and symptoms of AD, in the treatment of moderate to severe atopic dermatitis (AD).
METHODOLOGY:
- The phase 3 multinational LIBERTY AD open-label extension study evaluated the safety and efficacy of dupilumab in 2677 adults with moderate to severe AD who had previously participated in dupilumab trials over 5 years; 334 patients (12.5%) completed treatment up to 5 years.
- Patients started with subcutaneous dupilumab, initially dosed weekly after a loading dose, then every 2 weeks in 2019.
- The primary outcomes were the incidence and rate of treatment-emergent adverse events (TEAEs).
TAKEAWAY:
- Overall, 14,717 TEAEs were reported over 5 years. The exposure-adjusted incidence rate decreased over time and was 252.48 events per 100 patient-years.
- The most common TEAEs were nasopharyngitis (28.9%), worsening AD (16.7%), upper respiratory tract infection (13.6%), conjunctivitis (10.3%), allergic conjunctivitis (9%), headache (8.1%), oral herpes (7.5%), and injection-site reactions (5.2%).
- Serious and severe TEAE rates were 10.6% and 10.0%, respectively. Exposure-adjusted incidence rates were 6.66 and 6.71 events per 100 patient-years, respectively.
- At week 260, 67.5% of patients had achieved clear or almost clear skin according to the Investigator’s Global Assessment, and 88.9% experienced a 75% or greater improvement in the Eczema Area and Severity Index.
IN PRACTICE:
“Safety and efficacy results from up to 5 years of dupilumab treatment in the LIBERTY AD open-label extension study support dupilumab as a continuous long-term treatment for adults with moderate to severe AD,” the authors concluded.
SOURCE:
The study was led by Lisa A. Beck, MD, University of Rochester, Rochester, New York, and was published online in JAMA Dermatology.
LIMITATIONS:
Study limitations included the absence of a placebo arm and treatment interruptions stemming from protocol changes. The number of patients who received biweekly doses was small. The early conclusion of the trial by the sponsor because of regulatory approval also resulted in a lower number of patients at later stages.
DISCLOSURES:
This study was funded by dupilumab manufacturers Sanofi and Regeneron Pharmaceuticals. Several authors declared ties with various pharmaceutical companies including Sanofi and Regeneron, and several authors were employees of Sanofi or Regeneron. No disclosures were reported by other authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Over 5 years, dupilumab demonstrated acceptable safety and sustained efficacy, with significant improvements in the signs and symptoms of AD, in the treatment of moderate to severe atopic dermatitis (AD).
METHODOLOGY:
- The phase 3 multinational LIBERTY AD open-label extension study evaluated the safety and efficacy of dupilumab in 2677 adults with moderate to severe AD who had previously participated in dupilumab trials over 5 years; 334 patients (12.5%) completed treatment up to 5 years.
- Patients started with subcutaneous dupilumab, initially dosed weekly after a loading dose, then every 2 weeks in 2019.
- The primary outcomes were the incidence and rate of treatment-emergent adverse events (TEAEs).
TAKEAWAY:
- Overall, 14,717 TEAEs were reported over 5 years. The exposure-adjusted incidence rate decreased over time and was 252.48 events per 100 patient-years.
- The most common TEAEs were nasopharyngitis (28.9%), worsening AD (16.7%), upper respiratory tract infection (13.6%), conjunctivitis (10.3%), allergic conjunctivitis (9%), headache (8.1%), oral herpes (7.5%), and injection-site reactions (5.2%).
- Serious and severe TEAE rates were 10.6% and 10.0%, respectively. Exposure-adjusted incidence rates were 6.66 and 6.71 events per 100 patient-years, respectively.
- At week 260, 67.5% of patients had achieved clear or almost clear skin according to the Investigator’s Global Assessment, and 88.9% experienced a 75% or greater improvement in the Eczema Area and Severity Index.
IN PRACTICE:
“Safety and efficacy results from up to 5 years of dupilumab treatment in the LIBERTY AD open-label extension study support dupilumab as a continuous long-term treatment for adults with moderate to severe AD,” the authors concluded.
SOURCE:
The study was led by Lisa A. Beck, MD, University of Rochester, Rochester, New York, and was published online in JAMA Dermatology.
LIMITATIONS:
Study limitations included the absence of a placebo arm and treatment interruptions stemming from protocol changes. The number of patients who received biweekly doses was small. The early conclusion of the trial by the sponsor because of regulatory approval also resulted in a lower number of patients at later stages.
DISCLOSURES:
This study was funded by dupilumab manufacturers Sanofi and Regeneron Pharmaceuticals. Several authors declared ties with various pharmaceutical companies including Sanofi and Regeneron, and several authors were employees of Sanofi or Regeneron. No disclosures were reported by other authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
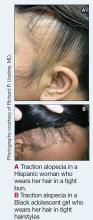
Act Fast With Traction Alopecia to Avoid Permanent Hair Loss
Traction alopecia (TA) is a common type of alopecia that ultimately can result in permanent hair loss. It often is caused or worsened by repetitive and prolonged hairstyling practices such as tight ponytails, braids, or locs, or use of wigs or weaves.1 Use of headwear, as in certain religious or ethnic groups, also can be contributory.2 Individuals participating in or training for occupations involving military service or ballet are at risk for TA due to hairstyling-specific policies. Early stages of TA are reversible with proper treatment and avoidance of exacerbating factors, emphasizing the importance of prompt recognition.3
Epidemiology
Data on the true prevalence of TA are lacking. It can occur in individuals of any race or any hair type. However, it is most common in women of African descent, affecting approximately one-third of this population.4 Other commonly affected groups include ballerinas and active-duty service members due to tight ponytails and buns, as well as the Sikh population due to the use of turbans as a part of their religious practice.2,5,6
Traction alopecia also impacts children, particularly those of African descent. A 2007 study of schoolchildren in South Africa determined that more than 17% of young African girls had evidence of TA—even some as young as 6 years of age.7
Traction alopecia can be caused or exacerbated by the use of hair clips and bobby pins that aid holding styles in place.8 Hair shaft morphology may contribute to the risk for TA, with more tightly coiled hair types being more susceptible.8 Variables such as use of chemical relaxers also increase the risk for disease, especially when combined with high-tension styling methods such as braids.9
Key clinical features
Patients with TA clinically present with hair loss and breakage in areas with tension, most commonly the marginal areas of the scalp as well as the frontal hairline and temporal scalp. Hair loss can result in a “fringe sign,” in which a patient may have preservation of a thin line of hairs at the frontal aspect of the hairline with a band of hair loss behind.10 This presentation may be used to differentiate TA from other forms of alopecia, including frontal fibrosing alopecia and female pattern hair loss. When the hair loss is not marginal, it may mimic other forms of patchy hair loss including alopecia areata and trichotillomania. Other clinical findings in TA may include broken hairs, pustules, and follicular papules.10 Patients also may describe symptoms such as scalp tenderness with specific hairstyles or headaches,11 or they may be completely asymptomatic.
Trichoscopy can be helpful in guiding diagnosis and treatment. Patients with TA often have perifollicular erythema and hair casts (cylindrical structures that encircle the proximal hair shafts) in the earlier stages of the disease, with eventual loss of follicular ostia in the later stages.10,12 Hair casts also may indicate ongoing traction.12 The flambeau sign—white tracks seen on trichoscopy in the direction the hair is pulled—resembles a lit torch.13
Worth noting
Early-stage TA can be reversed by avoiding hair tension. However, patients may not be amenable to this due to personal hairstyling preferences, job duties, or religious practices. Treatment with topical or intralesional steroids or even oral antibiotics such as doxycycline for its anti-inflammatory ability may result in regrowth of lost hair if the follicles are not permanently lost and exacerbating factors are avoided.3,14 Both topical and oral minoxidil have been used with success, with minoxidil thought to increase hair density by extending the anagen (growth) phase of hair follicles.3,15 Culturally sensitive patient counseling on the condition and potential exacerbating factors is critical.16
At later stages of the disease—after loss of follicular ostia has occurred—surgical interventions should be considered,17 such as hair transplantation, which can be successful but remains a technical challenge due to variability in hair shaft curvature.18 Additionally, the cost of the procedure can limit use, and some patients may not be optimal candidates due to the extent of their hair loss. Traction alopecia may not be the only hair loss condition present. Examining the scalp is important even if the chief area of concern is the marginal scalp.
Health disparity highlight
Prevention, early identification, and treatment initiated in a timely fashion are crucial to prevent permanent hair loss. There are added societal and cultural pressures that impact hairstyle and hair care practices, especially for those with tightly coiled hair.19 Historically, tightly coiled hair has been unfairly viewed as “unprofessional,” “unkempt,” and a challenge to “manage” by some. Thus, heat, chemical relaxers, and tight hairstyles holding hair in one position have been used to straighten the hair permanently or temporarily or to keep it maintained in a style that did not necessitate excessive manipulation—often contributing to further tension on the hair.
Military service branches have evaluated and changed some hair-related policies to reflect the diverse hair types of military personnel.20 The CROWN Act (www.thecrownact.com/about)—“Creating a Respectful and Open World for Natural Hair”—is a model law passed by 26 states that prohibits race-based hair discrimination, which is the denial of employment and educational opportunities because of hair texture. Although the law has not been passed in every state, it may help individuals with tightly coiled hair to embrace natural hairstyles. However, even hairstyles with one’s own natural curl pattern can contribute to tension and thus potential development of TA.
1. Larrondo J, McMichael AJ. Traction alopecia. JAMA Dermatol. 2023;159:676. doi:10.1001/jamadermatol.2022.6298
2. James J, Saladi RN, Fox JL. Traction alopecia in Sikh male patients. J Am Board Fam Med. 2007;20:497-498. doi:10.3122/jabfm.2007.05.070076
3. Callender VD, McMichael AJ, Cohen GF. Medical and surgical therapies for alopecias in black women. Dermatol Ther. 2004;17:164-176.
4. Loussouarn G, El Rawadi C, Genain G. Diversity of hair growth profiles. Int J Dermatol. 2005;44(suppl 1):6-9.
5. Samrao AChen CZedek Det al. Traction alopecia in a ballerina: clinicopathologic features. Arch Dermatol. 2010;146:918-935. doi:10.1001/archdermatol.2010.183
6. Korona-Bailey J, Banaag A, Nguyen DR, et al. Free the bun: prevalence of alopecia among active duty service women, fiscal years 2010-2019. Mil Med. 2023;188:e492-e496. doi:10.1093/milmed/usab274
7. Khumalo NP, Jessop S, Gumedze F, et al. Hairdressing is associated with scalp disease in African schoolchildren. Br J Dermatol. 2007;157:106-110. doi:10.1111/j.1365-2133.2007.07987.x
8. Billero V, Miteva M. Traction alopecia: the root of the problem. Clin Cosmet Investig Dermatol. 2018;11:149-159. doi:10.2147/CCID.S137296
9. Haskin A, Aguh C. All hairstyles are not created equal: what the dermatologist needs to know about black hairstyling practices and the risk of traction alopecia (TA). J Am Acad Dermatol. 2016;75:606-611. doi:10.1016/j.jaad.2016.02.1162
10. Samrao A, Price VH, Zedek D, et al. The “fringe sign”—a useful clinical finding in traction alopecia of the marginal hair line. Dermatol Online J. 2011;17:1.
11. Kararizou E, Bougea AM, Giotopoulou D, et al. An update on the less-known group of other primary headaches—a review. Eur Neurol Rev. 2014;9:71-77. doi:10.17925/ENR.2014.09.01.71
12. Tosti A, Miteva M, Torres F, et al. Hair casts are a dermoscopic clue for the diagnosis of traction alopecia. Br J Dermatol. 2010;163:1353-1355.
13. Agrawal S, Daruwalla SB, Dhurat RS. The flambeau sign—a new dermoscopy finding in a case of marginal traction alopecia. Australas J Dermatol. 2020;61:49-50. doi:10.1111/ajd.13187
14. Lawson CN, Hollinger J, Sethi S, et al. Updates in the understanding and treatments of skin & hair disorders in women of color. Int J Womens Dermatol. 2017;3:S21-S37.
15. Awad A, Chim I, Sharma P, et al. Low-dose oral minoxidil improves hair density in traction alopecia. J Am Acad Dermatol. 2023;89:157-159. doi:10.1016/j.jaad.2023.02.024
16. Grayson C, Heath CR. Counseling about traction alopecia: a “compliment, discuss, and suggest” method. Cutis. 2021;108:20-22.
17. Ozçelik D. Extensive traction alopecia attributable to ponytail hairstyle and its treatment with hair transplantation. Aesthetic Plast Surg. 2005;29:325-327. doi:10.1007/s00266-005-0004-5
18. Singh MK, Avram MR. Technical considerations for follicular unit extraction in African-American hair. Dermatol Surg. 2013;39:1282-1284. doi:10.1111/dsu.12229
19. Jones NL, Heath CR. Hair at the intersection of dermatology and anthropology: a conversation on race and relationships. Pediatr Dermatol. 2021;38(suppl 2):158-160.
20. Franklin JMM, Wohltmann WE, Wong EB. From buns to braids and ponytails: entering a new era of female military hair-grooming standards. Cutis. 2021;108:31-35. doi:10.12788/cutis.0296
Traction alopecia (TA) is a common type of alopecia that ultimately can result in permanent hair loss. It often is caused or worsened by repetitive and prolonged hairstyling practices such as tight ponytails, braids, or locs, or use of wigs or weaves.1 Use of headwear, as in certain religious or ethnic groups, also can be contributory.2 Individuals participating in or training for occupations involving military service or ballet are at risk for TA due to hairstyling-specific policies. Early stages of TA are reversible with proper treatment and avoidance of exacerbating factors, emphasizing the importance of prompt recognition.3
Epidemiology
Data on the true prevalence of TA are lacking. It can occur in individuals of any race or any hair type. However, it is most common in women of African descent, affecting approximately one-third of this population.4 Other commonly affected groups include ballerinas and active-duty service members due to tight ponytails and buns, as well as the Sikh population due to the use of turbans as a part of their religious practice.2,5,6
Traction alopecia also impacts children, particularly those of African descent. A 2007 study of schoolchildren in South Africa determined that more than 17% of young African girls had evidence of TA—even some as young as 6 years of age.7
Traction alopecia can be caused or exacerbated by the use of hair clips and bobby pins that aid holding styles in place.8 Hair shaft morphology may contribute to the risk for TA, with more tightly coiled hair types being more susceptible.8 Variables such as use of chemical relaxers also increase the risk for disease, especially when combined with high-tension styling methods such as braids.9
Key clinical features
Patients with TA clinically present with hair loss and breakage in areas with tension, most commonly the marginal areas of the scalp as well as the frontal hairline and temporal scalp. Hair loss can result in a “fringe sign,” in which a patient may have preservation of a thin line of hairs at the frontal aspect of the hairline with a band of hair loss behind.10 This presentation may be used to differentiate TA from other forms of alopecia, including frontal fibrosing alopecia and female pattern hair loss. When the hair loss is not marginal, it may mimic other forms of patchy hair loss including alopecia areata and trichotillomania. Other clinical findings in TA may include broken hairs, pustules, and follicular papules.10 Patients also may describe symptoms such as scalp tenderness with specific hairstyles or headaches,11 or they may be completely asymptomatic.
Trichoscopy can be helpful in guiding diagnosis and treatment. Patients with TA often have perifollicular erythema and hair casts (cylindrical structures that encircle the proximal hair shafts) in the earlier stages of the disease, with eventual loss of follicular ostia in the later stages.10,12 Hair casts also may indicate ongoing traction.12 The flambeau sign—white tracks seen on trichoscopy in the direction the hair is pulled—resembles a lit torch.13
Worth noting
Early-stage TA can be reversed by avoiding hair tension. However, patients may not be amenable to this due to personal hairstyling preferences, job duties, or religious practices. Treatment with topical or intralesional steroids or even oral antibiotics such as doxycycline for its anti-inflammatory ability may result in regrowth of lost hair if the follicles are not permanently lost and exacerbating factors are avoided.3,14 Both topical and oral minoxidil have been used with success, with minoxidil thought to increase hair density by extending the anagen (growth) phase of hair follicles.3,15 Culturally sensitive patient counseling on the condition and potential exacerbating factors is critical.16
At later stages of the disease—after loss of follicular ostia has occurred—surgical interventions should be considered,17 such as hair transplantation, which can be successful but remains a technical challenge due to variability in hair shaft curvature.18 Additionally, the cost of the procedure can limit use, and some patients may not be optimal candidates due to the extent of their hair loss. Traction alopecia may not be the only hair loss condition present. Examining the scalp is important even if the chief area of concern is the marginal scalp.
Health disparity highlight
Prevention, early identification, and treatment initiated in a timely fashion are crucial to prevent permanent hair loss. There are added societal and cultural pressures that impact hairstyle and hair care practices, especially for those with tightly coiled hair.19 Historically, tightly coiled hair has been unfairly viewed as “unprofessional,” “unkempt,” and a challenge to “manage” by some. Thus, heat, chemical relaxers, and tight hairstyles holding hair in one position have been used to straighten the hair permanently or temporarily or to keep it maintained in a style that did not necessitate excessive manipulation—often contributing to further tension on the hair.
Military service branches have evaluated and changed some hair-related policies to reflect the diverse hair types of military personnel.20 The CROWN Act (www.thecrownact.com/about)—“Creating a Respectful and Open World for Natural Hair”—is a model law passed by 26 states that prohibits race-based hair discrimination, which is the denial of employment and educational opportunities because of hair texture. Although the law has not been passed in every state, it may help individuals with tightly coiled hair to embrace natural hairstyles. However, even hairstyles with one’s own natural curl pattern can contribute to tension and thus potential development of TA.
Traction alopecia (TA) is a common type of alopecia that ultimately can result in permanent hair loss. It often is caused or worsened by repetitive and prolonged hairstyling practices such as tight ponytails, braids, or locs, or use of wigs or weaves.1 Use of headwear, as in certain religious or ethnic groups, also can be contributory.2 Individuals participating in or training for occupations involving military service or ballet are at risk for TA due to hairstyling-specific policies. Early stages of TA are reversible with proper treatment and avoidance of exacerbating factors, emphasizing the importance of prompt recognition.3
Epidemiology
Data on the true prevalence of TA are lacking. It can occur in individuals of any race or any hair type. However, it is most common in women of African descent, affecting approximately one-third of this population.4 Other commonly affected groups include ballerinas and active-duty service members due to tight ponytails and buns, as well as the Sikh population due to the use of turbans as a part of their religious practice.2,5,6
Traction alopecia also impacts children, particularly those of African descent. A 2007 study of schoolchildren in South Africa determined that more than 17% of young African girls had evidence of TA—even some as young as 6 years of age.7
Traction alopecia can be caused or exacerbated by the use of hair clips and bobby pins that aid holding styles in place.8 Hair shaft morphology may contribute to the risk for TA, with more tightly coiled hair types being more susceptible.8 Variables such as use of chemical relaxers also increase the risk for disease, especially when combined with high-tension styling methods such as braids.9
Key clinical features
Patients with TA clinically present with hair loss and breakage in areas with tension, most commonly the marginal areas of the scalp as well as the frontal hairline and temporal scalp. Hair loss can result in a “fringe sign,” in which a patient may have preservation of a thin line of hairs at the frontal aspect of the hairline with a band of hair loss behind.10 This presentation may be used to differentiate TA from other forms of alopecia, including frontal fibrosing alopecia and female pattern hair loss. When the hair loss is not marginal, it may mimic other forms of patchy hair loss including alopecia areata and trichotillomania. Other clinical findings in TA may include broken hairs, pustules, and follicular papules.10 Patients also may describe symptoms such as scalp tenderness with specific hairstyles or headaches,11 or they may be completely asymptomatic.
Trichoscopy can be helpful in guiding diagnosis and treatment. Patients with TA often have perifollicular erythema and hair casts (cylindrical structures that encircle the proximal hair shafts) in the earlier stages of the disease, with eventual loss of follicular ostia in the later stages.10,12 Hair casts also may indicate ongoing traction.12 The flambeau sign—white tracks seen on trichoscopy in the direction the hair is pulled—resembles a lit torch.13
Worth noting
Early-stage TA can be reversed by avoiding hair tension. However, patients may not be amenable to this due to personal hairstyling preferences, job duties, or religious practices. Treatment with topical or intralesional steroids or even oral antibiotics such as doxycycline for its anti-inflammatory ability may result in regrowth of lost hair if the follicles are not permanently lost and exacerbating factors are avoided.3,14 Both topical and oral minoxidil have been used with success, with minoxidil thought to increase hair density by extending the anagen (growth) phase of hair follicles.3,15 Culturally sensitive patient counseling on the condition and potential exacerbating factors is critical.16
At later stages of the disease—after loss of follicular ostia has occurred—surgical interventions should be considered,17 such as hair transplantation, which can be successful but remains a technical challenge due to variability in hair shaft curvature.18 Additionally, the cost of the procedure can limit use, and some patients may not be optimal candidates due to the extent of their hair loss. Traction alopecia may not be the only hair loss condition present. Examining the scalp is important even if the chief area of concern is the marginal scalp.
Health disparity highlight
Prevention, early identification, and treatment initiated in a timely fashion are crucial to prevent permanent hair loss. There are added societal and cultural pressures that impact hairstyle and hair care practices, especially for those with tightly coiled hair.19 Historically, tightly coiled hair has been unfairly viewed as “unprofessional,” “unkempt,” and a challenge to “manage” by some. Thus, heat, chemical relaxers, and tight hairstyles holding hair in one position have been used to straighten the hair permanently or temporarily or to keep it maintained in a style that did not necessitate excessive manipulation—often contributing to further tension on the hair.
Military service branches have evaluated and changed some hair-related policies to reflect the diverse hair types of military personnel.20 The CROWN Act (www.thecrownact.com/about)—“Creating a Respectful and Open World for Natural Hair”—is a model law passed by 26 states that prohibits race-based hair discrimination, which is the denial of employment and educational opportunities because of hair texture. Although the law has not been passed in every state, it may help individuals with tightly coiled hair to embrace natural hairstyles. However, even hairstyles with one’s own natural curl pattern can contribute to tension and thus potential development of TA.
1. Larrondo J, McMichael AJ. Traction alopecia. JAMA Dermatol. 2023;159:676. doi:10.1001/jamadermatol.2022.6298
2. James J, Saladi RN, Fox JL. Traction alopecia in Sikh male patients. J Am Board Fam Med. 2007;20:497-498. doi:10.3122/jabfm.2007.05.070076
3. Callender VD, McMichael AJ, Cohen GF. Medical and surgical therapies for alopecias in black women. Dermatol Ther. 2004;17:164-176.
4. Loussouarn G, El Rawadi C, Genain G. Diversity of hair growth profiles. Int J Dermatol. 2005;44(suppl 1):6-9.
5. Samrao AChen CZedek Det al. Traction alopecia in a ballerina: clinicopathologic features. Arch Dermatol. 2010;146:918-935. doi:10.1001/archdermatol.2010.183
6. Korona-Bailey J, Banaag A, Nguyen DR, et al. Free the bun: prevalence of alopecia among active duty service women, fiscal years 2010-2019. Mil Med. 2023;188:e492-e496. doi:10.1093/milmed/usab274
7. Khumalo NP, Jessop S, Gumedze F, et al. Hairdressing is associated with scalp disease in African schoolchildren. Br J Dermatol. 2007;157:106-110. doi:10.1111/j.1365-2133.2007.07987.x
8. Billero V, Miteva M. Traction alopecia: the root of the problem. Clin Cosmet Investig Dermatol. 2018;11:149-159. doi:10.2147/CCID.S137296
9. Haskin A, Aguh C. All hairstyles are not created equal: what the dermatologist needs to know about black hairstyling practices and the risk of traction alopecia (TA). J Am Acad Dermatol. 2016;75:606-611. doi:10.1016/j.jaad.2016.02.1162
10. Samrao A, Price VH, Zedek D, et al. The “fringe sign”—a useful clinical finding in traction alopecia of the marginal hair line. Dermatol Online J. 2011;17:1.
11. Kararizou E, Bougea AM, Giotopoulou D, et al. An update on the less-known group of other primary headaches—a review. Eur Neurol Rev. 2014;9:71-77. doi:10.17925/ENR.2014.09.01.71
12. Tosti A, Miteva M, Torres F, et al. Hair casts are a dermoscopic clue for the diagnosis of traction alopecia. Br J Dermatol. 2010;163:1353-1355.
13. Agrawal S, Daruwalla SB, Dhurat RS. The flambeau sign—a new dermoscopy finding in a case of marginal traction alopecia. Australas J Dermatol. 2020;61:49-50. doi:10.1111/ajd.13187
14. Lawson CN, Hollinger J, Sethi S, et al. Updates in the understanding and treatments of skin & hair disorders in women of color. Int J Womens Dermatol. 2017;3:S21-S37.
15. Awad A, Chim I, Sharma P, et al. Low-dose oral minoxidil improves hair density in traction alopecia. J Am Acad Dermatol. 2023;89:157-159. doi:10.1016/j.jaad.2023.02.024
16. Grayson C, Heath CR. Counseling about traction alopecia: a “compliment, discuss, and suggest” method. Cutis. 2021;108:20-22.
17. Ozçelik D. Extensive traction alopecia attributable to ponytail hairstyle and its treatment with hair transplantation. Aesthetic Plast Surg. 2005;29:325-327. doi:10.1007/s00266-005-0004-5
18. Singh MK, Avram MR. Technical considerations for follicular unit extraction in African-American hair. Dermatol Surg. 2013;39:1282-1284. doi:10.1111/dsu.12229
19. Jones NL, Heath CR. Hair at the intersection of dermatology and anthropology: a conversation on race and relationships. Pediatr Dermatol. 2021;38(suppl 2):158-160.
20. Franklin JMM, Wohltmann WE, Wong EB. From buns to braids and ponytails: entering a new era of female military hair-grooming standards. Cutis. 2021;108:31-35. doi:10.12788/cutis.0296
1. Larrondo J, McMichael AJ. Traction alopecia. JAMA Dermatol. 2023;159:676. doi:10.1001/jamadermatol.2022.6298
2. James J, Saladi RN, Fox JL. Traction alopecia in Sikh male patients. J Am Board Fam Med. 2007;20:497-498. doi:10.3122/jabfm.2007.05.070076
3. Callender VD, McMichael AJ, Cohen GF. Medical and surgical therapies for alopecias in black women. Dermatol Ther. 2004;17:164-176.
4. Loussouarn G, El Rawadi C, Genain G. Diversity of hair growth profiles. Int J Dermatol. 2005;44(suppl 1):6-9.
5. Samrao AChen CZedek Det al. Traction alopecia in a ballerina: clinicopathologic features. Arch Dermatol. 2010;146:918-935. doi:10.1001/archdermatol.2010.183
6. Korona-Bailey J, Banaag A, Nguyen DR, et al. Free the bun: prevalence of alopecia among active duty service women, fiscal years 2010-2019. Mil Med. 2023;188:e492-e496. doi:10.1093/milmed/usab274
7. Khumalo NP, Jessop S, Gumedze F, et al. Hairdressing is associated with scalp disease in African schoolchildren. Br J Dermatol. 2007;157:106-110. doi:10.1111/j.1365-2133.2007.07987.x
8. Billero V, Miteva M. Traction alopecia: the root of the problem. Clin Cosmet Investig Dermatol. 2018;11:149-159. doi:10.2147/CCID.S137296
9. Haskin A, Aguh C. All hairstyles are not created equal: what the dermatologist needs to know about black hairstyling practices and the risk of traction alopecia (TA). J Am Acad Dermatol. 2016;75:606-611. doi:10.1016/j.jaad.2016.02.1162
10. Samrao A, Price VH, Zedek D, et al. The “fringe sign”—a useful clinical finding in traction alopecia of the marginal hair line. Dermatol Online J. 2011;17:1.
11. Kararizou E, Bougea AM, Giotopoulou D, et al. An update on the less-known group of other primary headaches—a review. Eur Neurol Rev. 2014;9:71-77. doi:10.17925/ENR.2014.09.01.71
12. Tosti A, Miteva M, Torres F, et al. Hair casts are a dermoscopic clue for the diagnosis of traction alopecia. Br J Dermatol. 2010;163:1353-1355.
13. Agrawal S, Daruwalla SB, Dhurat RS. The flambeau sign—a new dermoscopy finding in a case of marginal traction alopecia. Australas J Dermatol. 2020;61:49-50. doi:10.1111/ajd.13187
14. Lawson CN, Hollinger J, Sethi S, et al. Updates in the understanding and treatments of skin & hair disorders in women of color. Int J Womens Dermatol. 2017;3:S21-S37.
15. Awad A, Chim I, Sharma P, et al. Low-dose oral minoxidil improves hair density in traction alopecia. J Am Acad Dermatol. 2023;89:157-159. doi:10.1016/j.jaad.2023.02.024
16. Grayson C, Heath CR. Counseling about traction alopecia: a “compliment, discuss, and suggest” method. Cutis. 2021;108:20-22.
17. Ozçelik D. Extensive traction alopecia attributable to ponytail hairstyle and its treatment with hair transplantation. Aesthetic Plast Surg. 2005;29:325-327. doi:10.1007/s00266-005-0004-5
18. Singh MK, Avram MR. Technical considerations for follicular unit extraction in African-American hair. Dermatol Surg. 2013;39:1282-1284. doi:10.1111/dsu.12229
19. Jones NL, Heath CR. Hair at the intersection of dermatology and anthropology: a conversation on race and relationships. Pediatr Dermatol. 2021;38(suppl 2):158-160.
20. Franklin JMM, Wohltmann WE, Wong EB. From buns to braids and ponytails: entering a new era of female military hair-grooming standards. Cutis. 2021;108:31-35. doi:10.12788/cutis.0296
Cosmetic Botulinum Toxin A Doses May Differ in Sunny Climates
findings from a comparative cohort study suggested.
“Botulinum toxin A to the glabella is a popular cosmetic intervention,” researchers led by Kim L. Borsky, MD, MBBS, of the Department of Plastic and Reconstructive Surgery at Stoke Mandeville Hospital, Aylesbury, England, and colleagues wrote in their study, which was published in Plastic and Reconstructive Surgery. “Functional musculature differences may arise from chronic behavioral adjustment to high sun exposure levels, requiring greater doses. This could affect clinical practice globally.”
To investigate the effect of climate on real-world doses of the product, the researchers enrolled 523 women aged 35-60 years who received glabellar botulinum toxin treatment at two centers between 2012 and 2019: one in the United Kingdom and one in Malta. They evaluated data on 292 patients treated during the summer months at the Malta center (classified as the high sun-exposure group), and 231 patients treated during the winter months at the UK center (classified as the low sun-exposure group). The primary outcomes of interest were the required top-up doses and the total dose to achieve full paralysis. Smokers were excluded from the analysis, as were those who did not seek maximal paralysis, those documented as not compliant with posttreatment advice, and those with colds or fevers. They used univariable and multivariable analyses to compare the high vs low sun-exposure groups.
The researchers found that 68.5% of women in the high-sun group required a top-up dose to achieve full paralysis, compared with 61.5% in the low-sun group, a difference that did not reach statistical significance (P = .1032). All patients achieved full paralysis with the treatment protocol used. However, in the high-sun group, the mean top-up dose was significantly higher than that in the low-sun group (a mean of 9.30 vs 7.06 units, respectively; P = .0009), as was the mean total dose (a mean of 29.23 vs 27.25 units; P = .0031).
“Patients subject to less sun exposure require a lower dose than patients with high sun exposure, and this was present and persisted when controlling for potential confounders,” the researchers wrote. “Although robustly demonstrated, the difference in doses seen here was small, and so may not directly impact at a health economic level, as the difference would not necessarily change the number of vials used. However, it may be of relevance to training and protocolization of treatments. Rigid protocols about doses and distributions may lead to undertreatment if applied in sunnier climates.”
They acknowledged certain limitations of their study, including its unblinded design and the fact that they did not evaluate or control for ethnicity. They also characterized the population of Malta as “very homogeneous, mainly made up of Maltese with less than 5% foreigners,” while the demographics of the United Kingdom and especially London, where the injections were performed, “are much more diverse.”
Asked to comment on the results, Pooja Sodha, MD, director of the Center for Laser and Cosmetic Dermatology at George Washington University, Washington, DC, said that the study highlights the importance of tailoring neuromodulator treatment to the individual patient based not just on gender but also on lifestyle and climate. “The conclusion [of the study] is logical, but it’s encouraging that the data supports this,” Dr. Sodha told this news organization. “The potential confounders, such as injection technique (5 point vs 3 point), nonblinding of the evaluator, history of prior treatments, and variation in treatment effect by different botulinum toxin products may be important as well in how we consider this data in practice.”
This study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. Neither the researchers nor Dr. Sodha reported having financial disclosures.
A version of this article appeared on Medscape.com.
findings from a comparative cohort study suggested.
“Botulinum toxin A to the glabella is a popular cosmetic intervention,” researchers led by Kim L. Borsky, MD, MBBS, of the Department of Plastic and Reconstructive Surgery at Stoke Mandeville Hospital, Aylesbury, England, and colleagues wrote in their study, which was published in Plastic and Reconstructive Surgery. “Functional musculature differences may arise from chronic behavioral adjustment to high sun exposure levels, requiring greater doses. This could affect clinical practice globally.”
To investigate the effect of climate on real-world doses of the product, the researchers enrolled 523 women aged 35-60 years who received glabellar botulinum toxin treatment at two centers between 2012 and 2019: one in the United Kingdom and one in Malta. They evaluated data on 292 patients treated during the summer months at the Malta center (classified as the high sun-exposure group), and 231 patients treated during the winter months at the UK center (classified as the low sun-exposure group). The primary outcomes of interest were the required top-up doses and the total dose to achieve full paralysis. Smokers were excluded from the analysis, as were those who did not seek maximal paralysis, those documented as not compliant with posttreatment advice, and those with colds or fevers. They used univariable and multivariable analyses to compare the high vs low sun-exposure groups.
The researchers found that 68.5% of women in the high-sun group required a top-up dose to achieve full paralysis, compared with 61.5% in the low-sun group, a difference that did not reach statistical significance (P = .1032). All patients achieved full paralysis with the treatment protocol used. However, in the high-sun group, the mean top-up dose was significantly higher than that in the low-sun group (a mean of 9.30 vs 7.06 units, respectively; P = .0009), as was the mean total dose (a mean of 29.23 vs 27.25 units; P = .0031).
“Patients subject to less sun exposure require a lower dose than patients with high sun exposure, and this was present and persisted when controlling for potential confounders,” the researchers wrote. “Although robustly demonstrated, the difference in doses seen here was small, and so may not directly impact at a health economic level, as the difference would not necessarily change the number of vials used. However, it may be of relevance to training and protocolization of treatments. Rigid protocols about doses and distributions may lead to undertreatment if applied in sunnier climates.”
They acknowledged certain limitations of their study, including its unblinded design and the fact that they did not evaluate or control for ethnicity. They also characterized the population of Malta as “very homogeneous, mainly made up of Maltese with less than 5% foreigners,” while the demographics of the United Kingdom and especially London, where the injections were performed, “are much more diverse.”
Asked to comment on the results, Pooja Sodha, MD, director of the Center for Laser and Cosmetic Dermatology at George Washington University, Washington, DC, said that the study highlights the importance of tailoring neuromodulator treatment to the individual patient based not just on gender but also on lifestyle and climate. “The conclusion [of the study] is logical, but it’s encouraging that the data supports this,” Dr. Sodha told this news organization. “The potential confounders, such as injection technique (5 point vs 3 point), nonblinding of the evaluator, history of prior treatments, and variation in treatment effect by different botulinum toxin products may be important as well in how we consider this data in practice.”
This study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. Neither the researchers nor Dr. Sodha reported having financial disclosures.
A version of this article appeared on Medscape.com.
findings from a comparative cohort study suggested.
“Botulinum toxin A to the glabella is a popular cosmetic intervention,” researchers led by Kim L. Borsky, MD, MBBS, of the Department of Plastic and Reconstructive Surgery at Stoke Mandeville Hospital, Aylesbury, England, and colleagues wrote in their study, which was published in Plastic and Reconstructive Surgery. “Functional musculature differences may arise from chronic behavioral adjustment to high sun exposure levels, requiring greater doses. This could affect clinical practice globally.”
To investigate the effect of climate on real-world doses of the product, the researchers enrolled 523 women aged 35-60 years who received glabellar botulinum toxin treatment at two centers between 2012 and 2019: one in the United Kingdom and one in Malta. They evaluated data on 292 patients treated during the summer months at the Malta center (classified as the high sun-exposure group), and 231 patients treated during the winter months at the UK center (classified as the low sun-exposure group). The primary outcomes of interest were the required top-up doses and the total dose to achieve full paralysis. Smokers were excluded from the analysis, as were those who did not seek maximal paralysis, those documented as not compliant with posttreatment advice, and those with colds or fevers. They used univariable and multivariable analyses to compare the high vs low sun-exposure groups.
The researchers found that 68.5% of women in the high-sun group required a top-up dose to achieve full paralysis, compared with 61.5% in the low-sun group, a difference that did not reach statistical significance (P = .1032). All patients achieved full paralysis with the treatment protocol used. However, in the high-sun group, the mean top-up dose was significantly higher than that in the low-sun group (a mean of 9.30 vs 7.06 units, respectively; P = .0009), as was the mean total dose (a mean of 29.23 vs 27.25 units; P = .0031).
“Patients subject to less sun exposure require a lower dose than patients with high sun exposure, and this was present and persisted when controlling for potential confounders,” the researchers wrote. “Although robustly demonstrated, the difference in doses seen here was small, and so may not directly impact at a health economic level, as the difference would not necessarily change the number of vials used. However, it may be of relevance to training and protocolization of treatments. Rigid protocols about doses and distributions may lead to undertreatment if applied in sunnier climates.”
They acknowledged certain limitations of their study, including its unblinded design and the fact that they did not evaluate or control for ethnicity. They also characterized the population of Malta as “very homogeneous, mainly made up of Maltese with less than 5% foreigners,” while the demographics of the United Kingdom and especially London, where the injections were performed, “are much more diverse.”
Asked to comment on the results, Pooja Sodha, MD, director of the Center for Laser and Cosmetic Dermatology at George Washington University, Washington, DC, said that the study highlights the importance of tailoring neuromodulator treatment to the individual patient based not just on gender but also on lifestyle and climate. “The conclusion [of the study] is logical, but it’s encouraging that the data supports this,” Dr. Sodha told this news organization. “The potential confounders, such as injection technique (5 point vs 3 point), nonblinding of the evaluator, history of prior treatments, and variation in treatment effect by different botulinum toxin products may be important as well in how we consider this data in practice.”
This study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. Neither the researchers nor Dr. Sodha reported having financial disclosures.
A version of this article appeared on Medscape.com.
FROM PLASTIC AND RECONSTRUCTIVE SURGERY
Urticaria Linked to Higher Cancer Risk, Study Finds
TOPLINE:
which decreased to 6% in subsequent years, in a cohort study using Danish healthcare databases.
METHODOLOGY:
- Researchers conducted a retrospective cohort study using data from Danish healthcare registries and compared the incident cancer risk between patients with urticaria and the risk in the general population.
- They identified 87,507 patients (58% women) with a primary or secondary first-time hospital outpatient clinic, emergency room, or inpatient diagnosis of urticaria between 1980 and 2022, who were followed for a median of 10.1 years.
- Incident cancers, including nonmelanoma skin cancer, were identified using the Danish Cancer Registry and classified by the extent of spread at the time of diagnosis.
- This study computed the absolute cancer risk within the first year of an urticaria diagnosis and standardized incidence ratios (SIRs), with 95% CIs standardized to Danish national cancer rates.
TAKEAWAY:
- For the first year of follow-up, the absolute risk for all cancer types was 0.7%, and it was 29.5% for subsequent years. The overall SIR for all types of cancer was 1.09 (95% CI, 1.06-1.11), which was based on 7788 observed cancer cases compared with 7161 cases expected over the entire follow-up period.
- Within the first year of follow-up, 588 patients with urticaria were diagnosed with cancer, for an SIR of 1.49 (95% CI, 1.38-1.62) for all cancer types.
- After the first year, the SIR for all cancer sites decreased and stabilized at 1.06 (95% CI, 1.04-1.09), with 7200 observed cancer cases.
- The risk was highest for hematological cancers in the first year, particularly Hodgkin lymphoma (SIR, 5.35; 95% CI, 2.56-9.85).
IN PRACTICE:
“Our study suggests that urticaria may be a marker of occult cancer and that it is associated with a slightly increased long-term cancer risk,” the authors wrote.
SOURCE:
The study was led by Sissel B.T. Sørensen, departments of dermatology and rheumatology, Aarhus University Hospital, Aarhus, Denmark. It was published online on June 27, 2024, in the British Journal of Dermatology.
LIMITATIONS:
The study is limited by its observational design and reliance on registry data, which may be subject to misclassification or incomplete information. In addition, the study could not assess individual patient factors such as lifestyle or genetic predispositions that may influence cancer risk, and the results may not be generalizable to other populations. Finally, the exact biologic mechanisms linking urticaria and cancer remain unclear, warranting further investigation.
DISCLOSURES:
The study did not receive any funding. The authors reported that they had no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
which decreased to 6% in subsequent years, in a cohort study using Danish healthcare databases.
METHODOLOGY:
- Researchers conducted a retrospective cohort study using data from Danish healthcare registries and compared the incident cancer risk between patients with urticaria and the risk in the general population.
- They identified 87,507 patients (58% women) with a primary or secondary first-time hospital outpatient clinic, emergency room, or inpatient diagnosis of urticaria between 1980 and 2022, who were followed for a median of 10.1 years.
- Incident cancers, including nonmelanoma skin cancer, were identified using the Danish Cancer Registry and classified by the extent of spread at the time of diagnosis.
- This study computed the absolute cancer risk within the first year of an urticaria diagnosis and standardized incidence ratios (SIRs), with 95% CIs standardized to Danish national cancer rates.
TAKEAWAY:
- For the first year of follow-up, the absolute risk for all cancer types was 0.7%, and it was 29.5% for subsequent years. The overall SIR for all types of cancer was 1.09 (95% CI, 1.06-1.11), which was based on 7788 observed cancer cases compared with 7161 cases expected over the entire follow-up period.
- Within the first year of follow-up, 588 patients with urticaria were diagnosed with cancer, for an SIR of 1.49 (95% CI, 1.38-1.62) for all cancer types.
- After the first year, the SIR for all cancer sites decreased and stabilized at 1.06 (95% CI, 1.04-1.09), with 7200 observed cancer cases.
- The risk was highest for hematological cancers in the first year, particularly Hodgkin lymphoma (SIR, 5.35; 95% CI, 2.56-9.85).
IN PRACTICE:
“Our study suggests that urticaria may be a marker of occult cancer and that it is associated with a slightly increased long-term cancer risk,” the authors wrote.
SOURCE:
The study was led by Sissel B.T. Sørensen, departments of dermatology and rheumatology, Aarhus University Hospital, Aarhus, Denmark. It was published online on June 27, 2024, in the British Journal of Dermatology.
LIMITATIONS:
The study is limited by its observational design and reliance on registry data, which may be subject to misclassification or incomplete information. In addition, the study could not assess individual patient factors such as lifestyle or genetic predispositions that may influence cancer risk, and the results may not be generalizable to other populations. Finally, the exact biologic mechanisms linking urticaria and cancer remain unclear, warranting further investigation.
DISCLOSURES:
The study did not receive any funding. The authors reported that they had no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
which decreased to 6% in subsequent years, in a cohort study using Danish healthcare databases.
METHODOLOGY:
- Researchers conducted a retrospective cohort study using data from Danish healthcare registries and compared the incident cancer risk between patients with urticaria and the risk in the general population.
- They identified 87,507 patients (58% women) with a primary or secondary first-time hospital outpatient clinic, emergency room, or inpatient diagnosis of urticaria between 1980 and 2022, who were followed for a median of 10.1 years.
- Incident cancers, including nonmelanoma skin cancer, were identified using the Danish Cancer Registry and classified by the extent of spread at the time of diagnosis.
- This study computed the absolute cancer risk within the first year of an urticaria diagnosis and standardized incidence ratios (SIRs), with 95% CIs standardized to Danish national cancer rates.
TAKEAWAY:
- For the first year of follow-up, the absolute risk for all cancer types was 0.7%, and it was 29.5% for subsequent years. The overall SIR for all types of cancer was 1.09 (95% CI, 1.06-1.11), which was based on 7788 observed cancer cases compared with 7161 cases expected over the entire follow-up period.
- Within the first year of follow-up, 588 patients with urticaria were diagnosed with cancer, for an SIR of 1.49 (95% CI, 1.38-1.62) for all cancer types.
- After the first year, the SIR for all cancer sites decreased and stabilized at 1.06 (95% CI, 1.04-1.09), with 7200 observed cancer cases.
- The risk was highest for hematological cancers in the first year, particularly Hodgkin lymphoma (SIR, 5.35; 95% CI, 2.56-9.85).
IN PRACTICE:
“Our study suggests that urticaria may be a marker of occult cancer and that it is associated with a slightly increased long-term cancer risk,” the authors wrote.
SOURCE:
The study was led by Sissel B.T. Sørensen, departments of dermatology and rheumatology, Aarhus University Hospital, Aarhus, Denmark. It was published online on June 27, 2024, in the British Journal of Dermatology.
LIMITATIONS:
The study is limited by its observational design and reliance on registry data, which may be subject to misclassification or incomplete information. In addition, the study could not assess individual patient factors such as lifestyle or genetic predispositions that may influence cancer risk, and the results may not be generalizable to other populations. Finally, the exact biologic mechanisms linking urticaria and cancer remain unclear, warranting further investigation.
DISCLOSURES:
The study did not receive any funding. The authors reported that they had no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.