User login
Primary Cutaneous Marginal Zone B-Cell Lymphoma Discovered During Mohs Surgery for Basal Cell Carcinoma
To the Editor:
Primary cutaneous B-cell lymphomas (pcBCLs) can clinically mimic basal cell carcinomas (BCCs); however, histopathologic examination typically demonstrates features of lymphoma without evidence of an epithelial tumor. We present the case of a patient who demonstrated histologic features of both pcBCL and BCC in the same lesion, which was discovered during Mohs micrographic surgery.
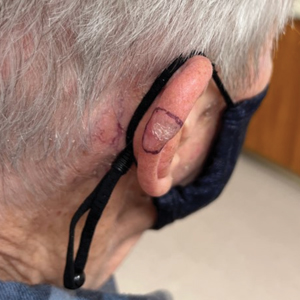
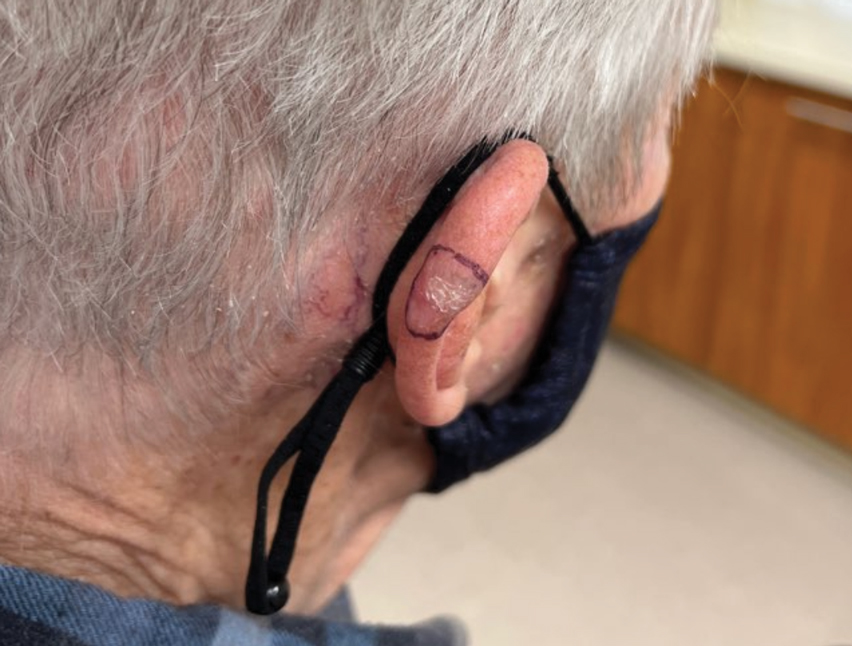
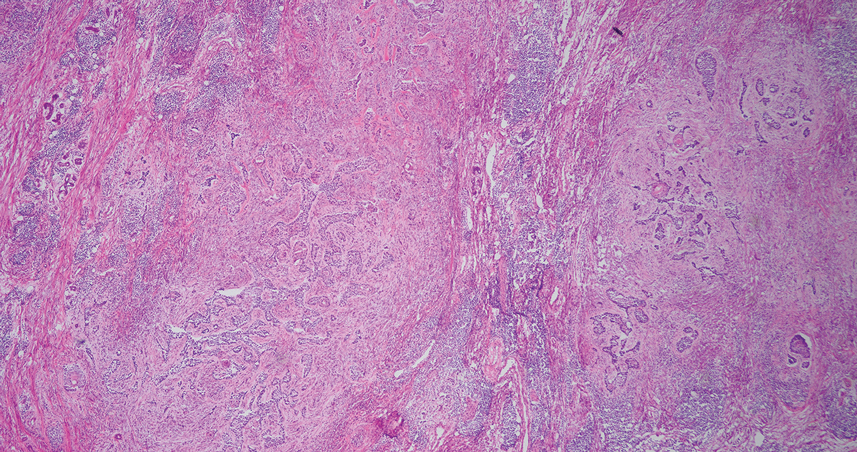
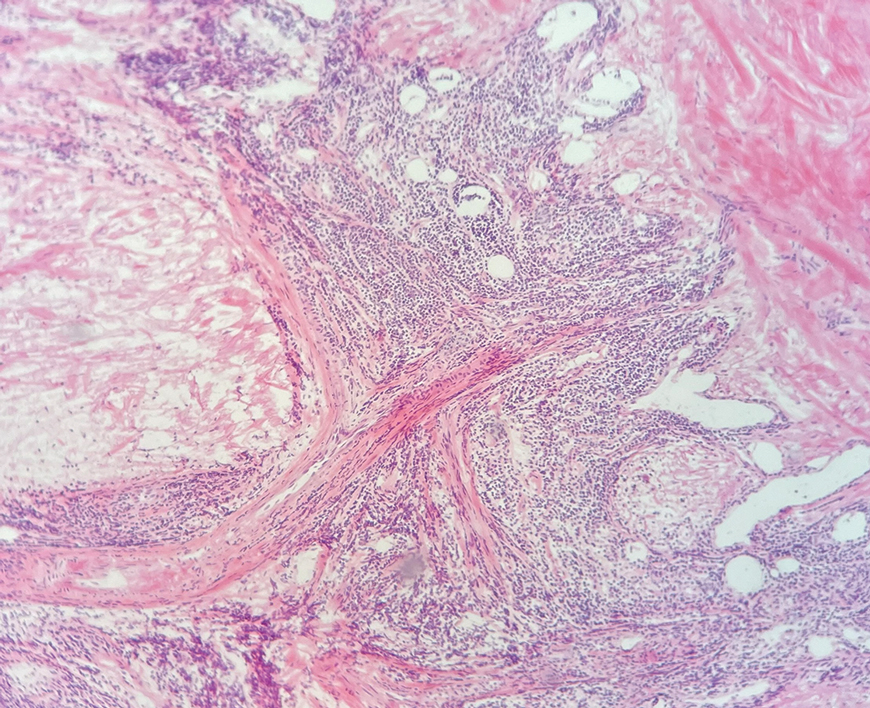
An 84-year-old man presented for Mohs surgery for a biopsy-proven nodular and infiltrative BCC on the right superior helix of the ear of 1 year’s duration. Physical examination of the ear revealed a 1.0×1.3–cm ulcerated indurated plaque with rolled borders and a central hyperkeratotic crust (Figure 1). Frozen sections from the first Mohs stage demonstrated residual superficial, infiltrative, and basosquamous BCC (Figure 2). In addition, there was a brisk inflammatory infiltrate throughout the deep margins. The second stage showed no residual BCC, but there still was a brisk atypical lymphocytic infiltrate, with some areas showing lymphocytes in a linear cordlike distribution (Figure 3). Permanent sections demonstrated infiltration of small to medium lymphoid cells. Immunohistochemistry stains were positive for CD20 and BCL2 and negative for CD5, CD10, BCL6, and CD43; a low Ki-67 proliferation fraction also was observed. B-cell clonality studies and polymerase chain reaction demonstrated rearrangements of the IgH and IgK genes, consistent with primary cutaneous marginal zone lymphoma (pcMZL). Positron emission tomography showed no spread of malignancy; therefore, medical oncology recommended observation and close monitoring.
Primary cutaneous B-cell lymphoma accounts for approximately 25% of all cutaneous lymphomas.1 Three main cutaneous subtypes exist: pcMZL; primary cutaneous follicular center lymphoma; and primary cutaneous diffuse large B-cell lymphoma, leg type. The second most common type of cutaneous lymphoma, pcMZL, accounts for 25% of cases of pcBCL.1 Primary cutaneous follicular center lymphoma makes up 60% of cutaneous lymphomas, and the remainder are primary cutaneous diffuse large B-cell lymphoma, leg type. All share a notable male predominance and onset most commonly in the sixth through eighth decades of life, although they also can occur in younger patients.1
Histologically, pcMZL has 2 distinct subtypes: one resembling mucosal-associated lymphoid tissue lymphomas and a more clinically aggressive subtype with heavy chain class switching, although intermediate forms also exist. Both are characterized by diffuse and/or nodular infiltrates in the subcutis and dermis with sparing of the epidermis. Often, these infiltrates are more prominent in the deeper sections examined, and occasionally they may be accompanied by germinal center follicles. Immunohistochemical stains are key in determining the pcBCL subtype. Primary cutaneous marginal zone lymphoma will most commonly show a BCL2+, BCL6–, CD20+, and CD10– immunophenotype, as in our case. If a majority of cells have undergone plasmacytoid differentiation, loss of CD20 can occur, but retention of other B-cell markers, such as CD79a and CD19, will be seen. Proliferation fraction via Ki-67 commonly is low, reflecting the indolence of this subtype of lymphoma.1
Monoclonal rearrangement of immunoglobulins also can occur, with IgH rearrangements detected in 60% to 80% of cases of pcMZL. Translocations are not a reliable method of diagnosis for pcMZL but can be present in a variable manner, with t(14;18), t(3;14), and t(11;18) reported in a subset of cases.2 Leukemic infiltrates encountered on frozen sections should prompt the Mohs surgeon to consider the possibility of a concomitant leukemia or lymphoma. In one study, 36% (20/55) of patients with chronic lymphocytic leukemia (CLL) were found to have predominantly leukemic B-cell infiltrates on frozen sections.3 Numerous reports also exist of asymptomatic patients being diagnosed with CLL due to leukemic infiltrates identified during Mohs surgery.4,5 Patients with systemic hematologic malignancies, including CLL and non-Hodgkin lymphoma, also are known to be at an increased risk for skin cancers, including keratinocyte cancers, melanoma, and Merkel cell carcinoma. This can be attributed partially to immunosuppression, a well-known risk factor for development of cutaneous malignancies.5 Padgett et al5 speculated that local immune suppression due to underlying pcBCL and reaction of lymphocytes to tumor antigens could have played a role in the development of BCC at this site. If a leukemic infiltrate is demonstrated, the surgeon should consider sending tissue for permanent section and immunostaining. This can be helpful to determine if it is a reactive or neoplastic process and aid in characterizing the leukemic infiltrate if it is suspected to be neoplastic in nature.
There are numerous reports of pcBCL imitating the cutaneous findings of BCC clinically, but this is quite uncommon on histopathology. As in our case, findings of sheets of dense, monomorphic lymphocytes; inability to clear inflammation on deeper Mohs sections; presence of primordial follicles; and atypical cytology, including predominance of blastic forms, plasmacytoid cells, or cleaved lymphocytes, should give the clinician pause to consider further evaluation through permanent sections as well as genetic and immunoglobulin studies by a dermatopathologist. This case highlights the importance of further evaluation when an atypical finding is encountered during Mohs surgery.
- Goyal A, LeBlanc RE, Carter JB. Cutaneous B-cell lymphoma. Hematol Oncol Clin North Am. 2019;33:149-161. doi:10.1016/j.hoc.2018.08.006
- Vitiello P, Sica A, Ronchi A, et al. Primary cutaneous B-cell lymphomas: an update. Front Oncol. 2020;10:651. doi:10.3389/fonc.2020.00651
- Mehrany K, Byrd DR, Roenigk RK, et al. Lymphocytic infiltrates and subclinical epithelial tumor extension in patients with chronic leukemia and solid-organ transplantation. Dermatol Surg. 2003;29:129-134. doi:10.1046/j.1524-4725.2003.29034.x
- Walters M, Chang C, Castillo JR. Diagnosis of chronic lymphocytic leukemia during Mohs micrographic surgery. JAAD Case Rep. 2023;33:1-3. doi:10.1016/j.jdcr.2022.12.012
- Padgett JK, Parlette HL, English JC. A diagnosis of chronic lymphocytic leukemia prompted by cutaneous lymphocytic infiltrates present in mohs micrographic surgery frozen sections. Dermatol Surg. 2003;29:769-771. doi:10.1046/j.1524-4725.2003.29194.x
To the Editor:
Primary cutaneous B-cell lymphomas (pcBCLs) can clinically mimic basal cell carcinomas (BCCs); however, histopathologic examination typically demonstrates features of lymphoma without evidence of an epithelial tumor. We present the case of a patient who demonstrated histologic features of both pcBCL and BCC in the same lesion, which was discovered during Mohs micrographic surgery.
An 84-year-old man presented for Mohs surgery for a biopsy-proven nodular and infiltrative BCC on the right superior helix of the ear of 1 year’s duration. Physical examination of the ear revealed a 1.0×1.3–cm ulcerated indurated plaque with rolled borders and a central hyperkeratotic crust (Figure 1). Frozen sections from the first Mohs stage demonstrated residual superficial, infiltrative, and basosquamous BCC (Figure 2). In addition, there was a brisk inflammatory infiltrate throughout the deep margins. The second stage showed no residual BCC, but there still was a brisk atypical lymphocytic infiltrate, with some areas showing lymphocytes in a linear cordlike distribution (Figure 3). Permanent sections demonstrated infiltration of small to medium lymphoid cells. Immunohistochemistry stains were positive for CD20 and BCL2 and negative for CD5, CD10, BCL6, and CD43; a low Ki-67 proliferation fraction also was observed. B-cell clonality studies and polymerase chain reaction demonstrated rearrangements of the IgH and IgK genes, consistent with primary cutaneous marginal zone lymphoma (pcMZL). Positron emission tomography showed no spread of malignancy; therefore, medical oncology recommended observation and close monitoring.



Primary cutaneous B-cell lymphoma accounts for approximately 25% of all cutaneous lymphomas.1 Three main cutaneous subtypes exist: pcMZL; primary cutaneous follicular center lymphoma; and primary cutaneous diffuse large B-cell lymphoma, leg type. The second most common type of cutaneous lymphoma, pcMZL, accounts for 25% of cases of pcBCL.1 Primary cutaneous follicular center lymphoma makes up 60% of cutaneous lymphomas, and the remainder are primary cutaneous diffuse large B-cell lymphoma, leg type. All share a notable male predominance and onset most commonly in the sixth through eighth decades of life, although they also can occur in younger patients.1
Histologically, pcMZL has 2 distinct subtypes: one resembling mucosal-associated lymphoid tissue lymphomas and a more clinically aggressive subtype with heavy chain class switching, although intermediate forms also exist. Both are characterized by diffuse and/or nodular infiltrates in the subcutis and dermis with sparing of the epidermis. Often, these infiltrates are more prominent in the deeper sections examined, and occasionally they may be accompanied by germinal center follicles. Immunohistochemical stains are key in determining the pcBCL subtype. Primary cutaneous marginal zone lymphoma will most commonly show a BCL2+, BCL6–, CD20+, and CD10– immunophenotype, as in our case. If a majority of cells have undergone plasmacytoid differentiation, loss of CD20 can occur, but retention of other B-cell markers, such as CD79a and CD19, will be seen. Proliferation fraction via Ki-67 commonly is low, reflecting the indolence of this subtype of lymphoma.1
Monoclonal rearrangement of immunoglobulins also can occur, with IgH rearrangements detected in 60% to 80% of cases of pcMZL. Translocations are not a reliable method of diagnosis for pcMZL but can be present in a variable manner, with t(14;18), t(3;14), and t(11;18) reported in a subset of cases.2 Leukemic infiltrates encountered on frozen sections should prompt the Mohs surgeon to consider the possibility of a concomitant leukemia or lymphoma. In one study, 36% (20/55) of patients with chronic lymphocytic leukemia (CLL) were found to have predominantly leukemic B-cell infiltrates on frozen sections.3 Numerous reports also exist of asymptomatic patients being diagnosed with CLL due to leukemic infiltrates identified during Mohs surgery.4,5 Patients with systemic hematologic malignancies, including CLL and non-Hodgkin lymphoma, also are known to be at an increased risk for skin cancers, including keratinocyte cancers, melanoma, and Merkel cell carcinoma. This can be attributed partially to immunosuppression, a well-known risk factor for development of cutaneous malignancies.5 Padgett et al5 speculated that local immune suppression due to underlying pcBCL and reaction of lymphocytes to tumor antigens could have played a role in the development of BCC at this site. If a leukemic infiltrate is demonstrated, the surgeon should consider sending tissue for permanent section and immunostaining. This can be helpful to determine if it is a reactive or neoplastic process and aid in characterizing the leukemic infiltrate if it is suspected to be neoplastic in nature.
There are numerous reports of pcBCL imitating the cutaneous findings of BCC clinically, but this is quite uncommon on histopathology. As in our case, findings of sheets of dense, monomorphic lymphocytes; inability to clear inflammation on deeper Mohs sections; presence of primordial follicles; and atypical cytology, including predominance of blastic forms, plasmacytoid cells, or cleaved lymphocytes, should give the clinician pause to consider further evaluation through permanent sections as well as genetic and immunoglobulin studies by a dermatopathologist. This case highlights the importance of further evaluation when an atypical finding is encountered during Mohs surgery.
To the Editor:
Primary cutaneous B-cell lymphomas (pcBCLs) can clinically mimic basal cell carcinomas (BCCs); however, histopathologic examination typically demonstrates features of lymphoma without evidence of an epithelial tumor. We present the case of a patient who demonstrated histologic features of both pcBCL and BCC in the same lesion, which was discovered during Mohs micrographic surgery.
An 84-year-old man presented for Mohs surgery for a biopsy-proven nodular and infiltrative BCC on the right superior helix of the ear of 1 year’s duration. Physical examination of the ear revealed a 1.0×1.3–cm ulcerated indurated plaque with rolled borders and a central hyperkeratotic crust (Figure 1). Frozen sections from the first Mohs stage demonstrated residual superficial, infiltrative, and basosquamous BCC (Figure 2). In addition, there was a brisk inflammatory infiltrate throughout the deep margins. The second stage showed no residual BCC, but there still was a brisk atypical lymphocytic infiltrate, with some areas showing lymphocytes in a linear cordlike distribution (Figure 3). Permanent sections demonstrated infiltration of small to medium lymphoid cells. Immunohistochemistry stains were positive for CD20 and BCL2 and negative for CD5, CD10, BCL6, and CD43; a low Ki-67 proliferation fraction also was observed. B-cell clonality studies and polymerase chain reaction demonstrated rearrangements of the IgH and IgK genes, consistent with primary cutaneous marginal zone lymphoma (pcMZL). Positron emission tomography showed no spread of malignancy; therefore, medical oncology recommended observation and close monitoring.



Primary cutaneous B-cell lymphoma accounts for approximately 25% of all cutaneous lymphomas.1 Three main cutaneous subtypes exist: pcMZL; primary cutaneous follicular center lymphoma; and primary cutaneous diffuse large B-cell lymphoma, leg type. The second most common type of cutaneous lymphoma, pcMZL, accounts for 25% of cases of pcBCL.1 Primary cutaneous follicular center lymphoma makes up 60% of cutaneous lymphomas, and the remainder are primary cutaneous diffuse large B-cell lymphoma, leg type. All share a notable male predominance and onset most commonly in the sixth through eighth decades of life, although they also can occur in younger patients.1
Histologically, pcMZL has 2 distinct subtypes: one resembling mucosal-associated lymphoid tissue lymphomas and a more clinically aggressive subtype with heavy chain class switching, although intermediate forms also exist. Both are characterized by diffuse and/or nodular infiltrates in the subcutis and dermis with sparing of the epidermis. Often, these infiltrates are more prominent in the deeper sections examined, and occasionally they may be accompanied by germinal center follicles. Immunohistochemical stains are key in determining the pcBCL subtype. Primary cutaneous marginal zone lymphoma will most commonly show a BCL2+, BCL6–, CD20+, and CD10– immunophenotype, as in our case. If a majority of cells have undergone plasmacytoid differentiation, loss of CD20 can occur, but retention of other B-cell markers, such as CD79a and CD19, will be seen. Proliferation fraction via Ki-67 commonly is low, reflecting the indolence of this subtype of lymphoma.1
Monoclonal rearrangement of immunoglobulins also can occur, with IgH rearrangements detected in 60% to 80% of cases of pcMZL. Translocations are not a reliable method of diagnosis for pcMZL but can be present in a variable manner, with t(14;18), t(3;14), and t(11;18) reported in a subset of cases.2 Leukemic infiltrates encountered on frozen sections should prompt the Mohs surgeon to consider the possibility of a concomitant leukemia or lymphoma. In one study, 36% (20/55) of patients with chronic lymphocytic leukemia (CLL) were found to have predominantly leukemic B-cell infiltrates on frozen sections.3 Numerous reports also exist of asymptomatic patients being diagnosed with CLL due to leukemic infiltrates identified during Mohs surgery.4,5 Patients with systemic hematologic malignancies, including CLL and non-Hodgkin lymphoma, also are known to be at an increased risk for skin cancers, including keratinocyte cancers, melanoma, and Merkel cell carcinoma. This can be attributed partially to immunosuppression, a well-known risk factor for development of cutaneous malignancies.5 Padgett et al5 speculated that local immune suppression due to underlying pcBCL and reaction of lymphocytes to tumor antigens could have played a role in the development of BCC at this site. If a leukemic infiltrate is demonstrated, the surgeon should consider sending tissue for permanent section and immunostaining. This can be helpful to determine if it is a reactive or neoplastic process and aid in characterizing the leukemic infiltrate if it is suspected to be neoplastic in nature.
There are numerous reports of pcBCL imitating the cutaneous findings of BCC clinically, but this is quite uncommon on histopathology. As in our case, findings of sheets of dense, monomorphic lymphocytes; inability to clear inflammation on deeper Mohs sections; presence of primordial follicles; and atypical cytology, including predominance of blastic forms, plasmacytoid cells, or cleaved lymphocytes, should give the clinician pause to consider further evaluation through permanent sections as well as genetic and immunoglobulin studies by a dermatopathologist. This case highlights the importance of further evaluation when an atypical finding is encountered during Mohs surgery.
- Goyal A, LeBlanc RE, Carter JB. Cutaneous B-cell lymphoma. Hematol Oncol Clin North Am. 2019;33:149-161. doi:10.1016/j.hoc.2018.08.006
- Vitiello P, Sica A, Ronchi A, et al. Primary cutaneous B-cell lymphomas: an update. Front Oncol. 2020;10:651. doi:10.3389/fonc.2020.00651
- Mehrany K, Byrd DR, Roenigk RK, et al. Lymphocytic infiltrates and subclinical epithelial tumor extension in patients with chronic leukemia and solid-organ transplantation. Dermatol Surg. 2003;29:129-134. doi:10.1046/j.1524-4725.2003.29034.x
- Walters M, Chang C, Castillo JR. Diagnosis of chronic lymphocytic leukemia during Mohs micrographic surgery. JAAD Case Rep. 2023;33:1-3. doi:10.1016/j.jdcr.2022.12.012
- Padgett JK, Parlette HL, English JC. A diagnosis of chronic lymphocytic leukemia prompted by cutaneous lymphocytic infiltrates present in mohs micrographic surgery frozen sections. Dermatol Surg. 2003;29:769-771. doi:10.1046/j.1524-4725.2003.29194.x
- Goyal A, LeBlanc RE, Carter JB. Cutaneous B-cell lymphoma. Hematol Oncol Clin North Am. 2019;33:149-161. doi:10.1016/j.hoc.2018.08.006
- Vitiello P, Sica A, Ronchi A, et al. Primary cutaneous B-cell lymphomas: an update. Front Oncol. 2020;10:651. doi:10.3389/fonc.2020.00651
- Mehrany K, Byrd DR, Roenigk RK, et al. Lymphocytic infiltrates and subclinical epithelial tumor extension in patients with chronic leukemia and solid-organ transplantation. Dermatol Surg. 2003;29:129-134. doi:10.1046/j.1524-4725.2003.29034.x
- Walters M, Chang C, Castillo JR. Diagnosis of chronic lymphocytic leukemia during Mohs micrographic surgery. JAAD Case Rep. 2023;33:1-3. doi:10.1016/j.jdcr.2022.12.012
- Padgett JK, Parlette HL, English JC. A diagnosis of chronic lymphocytic leukemia prompted by cutaneous lymphocytic infiltrates present in mohs micrographic surgery frozen sections. Dermatol Surg. 2003;29:769-771. doi:10.1046/j.1524-4725.2003.29194.x
Primary Cutaneous Marginal Zone B-Cell Lymphoma Discovered During Mohs Surgery for Basal Cell Carcinoma
Primary Cutaneous Marginal Zone B-Cell Lymphoma Discovered During Mohs Surgery for Basal Cell Carcinoma
Practice Points
- Collision tumors of cutaneous B-cell lymphoma and basal cell carcinoma occurring within the same lesion are uncommon findings during Mohs surgery.
- Sheets of atypical monomorphic lymphocytes on deeper Mohs sections should prompt the surgeon to consider further evaluation, including sending tissue for permanent sections.