User login
Idarucizumab reverses dabigatran’s anticoagulant effects
TORONTO – Idarucizumab is a promising agent that quickly and safely reverses the anticoagulant effects of dabigatran whether the goal is to control serious bleeding or to permit urgent surgery, according to interim results of a multicenter trial.
Idarucizumab is a monoclonal antibody that binds to dabigatran to reverse its activity. The data, presented by Dr. V. Charles Pollack Jr. at the International Society on Thrombosis and Haemostasis congress, involved the first 90 patients of an ongoing trial with a planned enrollment of 300. The data from this trial, called REVERSE-AD, were published online simultaneously with the June 22 presentation at the congress (N. Engl. J. Med 2015 [doi:10.1056/NEJMoa1502000]).
“Non–vitamin K antagonist oral anticoagulants (NOACs) are generally safer than warfarin, and provide similar or improved efficacy in the prevention of stroke in patients with nonvalvular atrial fibrillation and in the prevention and treatment of venous thromboembolism,” Dr. Pollack said in an interview. “Nonetheless, serious bleeding events may occur with NOAC use, and patients taking one of these agents occasionally require urgent surgery or other intervention for which normal hemostasis is required,” added Dr. Pollack, chair of the department of emergency medicine at Pennsylvania Hospital in Philadelphia.
In RE-VERSE AD (a study of the reversal effects of idarucizumab on active dabigatran), the first 90 patients were divided into two distinct groups. Group A, with 51 patients, included those on dabigatran with serious bleeding. Group B, with 39 patients, required reversal of dabigatran for urgent or emergent procedures. In both, idarucizumab provided a median maximum reversal of 100% (95% confidence interval, 100-100) of the anticoagulation effect within 4 hours.
Clotting assays were normalized almost immediately in almost 90% of patients, and the effect was durable, with 80% having measured dabigatran levels reflecting no significant anticoagulation 24 hours later, Dr. Pollack said.
“Clinical outcomes were quite good in this multimorbid patient population, with restoration of hemostasis as reported by local investigators achieved in less than 12 hours when assessable, and with 92% of surgical patients being reported as having normal hemostasis at the time of the procedure,” he said.
Idarucizumab was generally well tolerated in the patient population. “There were no serious adverse events related to the reversal agent ... and only one patient experienced a thrombotic complication within 72 hours, and that patient had not been restarted on any antithrombotic medications,” Dr. Pollack said.
“The study is ongoing,” he added, “but these interim results show rather convincingly that idarucizumab completely and safely reverses the anticoagulant effects of dabigatran within minutes.”
In addition, Dr. Pollack said the availability of a specific reversal agent for dabigatran would enhance its safety margin, and thus alleviate the fears of providers who may hesitate to use a NOAC because of the lack of an “antidote.”
“In fact, most such cases can already be successfully and safely managed with general support and ‘tincture of time’ (the half-life of dabigatran is much shorter than that of warfarin), but having a specific ‘go-to’ option could streamline the care of the most significantly compromised patients,” he said.
Dr. Pollack emphasized, however, that idarucizumab is a specific reversal agent for dabigatran, not an antidote. “To me, the latter would imply that idarucizumab immediately stops bleeding associated with active use of dabigatran,” he said.
Providers should realize that while idarucizumab seems capable of removing dabigatran-induced coagulopathy from the list of concerns when managing a patient with serious bleeding or before a “sharp” procedure, bleeding is a multifaceted issue that also may be due to traumatized blood vessels, other causes of coagulopathy such as liver disease, or concurrent use of antiplatelet medications, he said.
“The patient with a serious or life-threatening bleed on dabigatran will likely need additional care to investigate and manage such concerns,” Dr. Pollack said. “But at least idarucizumab can specifically, safely, and rapidly address the primary consideration.
“The safety of anticoagulation therapy with dabigatran is further enhanced with idarucizumab, a specific reversal agent that won’t need to be used often, but the availability of which would be reassuring to prescribers,” he concluded.
Boehringer Ingelheim sponsored RE-VERSE AD. Idarucizumab was given a fast-track status by the Food and Drug Administration, and BI submitted a new drug application in March 2015, according to the company.
Dr. Pollack reported receiving personal fees from BI, Janssen, Daiichi-Sankyo, Bristol-Myers Squibb, and Pfizer. Disclosures for all the investigators are available at NEJM.org.
TORONTO – Idarucizumab is a promising agent that quickly and safely reverses the anticoagulant effects of dabigatran whether the goal is to control serious bleeding or to permit urgent surgery, according to interim results of a multicenter trial.
Idarucizumab is a monoclonal antibody that binds to dabigatran to reverse its activity. The data, presented by Dr. V. Charles Pollack Jr. at the International Society on Thrombosis and Haemostasis congress, involved the first 90 patients of an ongoing trial with a planned enrollment of 300. The data from this trial, called REVERSE-AD, were published online simultaneously with the June 22 presentation at the congress (N. Engl. J. Med 2015 [doi:10.1056/NEJMoa1502000]).
“Non–vitamin K antagonist oral anticoagulants (NOACs) are generally safer than warfarin, and provide similar or improved efficacy in the prevention of stroke in patients with nonvalvular atrial fibrillation and in the prevention and treatment of venous thromboembolism,” Dr. Pollack said in an interview. “Nonetheless, serious bleeding events may occur with NOAC use, and patients taking one of these agents occasionally require urgent surgery or other intervention for which normal hemostasis is required,” added Dr. Pollack, chair of the department of emergency medicine at Pennsylvania Hospital in Philadelphia.
In RE-VERSE AD (a study of the reversal effects of idarucizumab on active dabigatran), the first 90 patients were divided into two distinct groups. Group A, with 51 patients, included those on dabigatran with serious bleeding. Group B, with 39 patients, required reversal of dabigatran for urgent or emergent procedures. In both, idarucizumab provided a median maximum reversal of 100% (95% confidence interval, 100-100) of the anticoagulation effect within 4 hours.
Clotting assays were normalized almost immediately in almost 90% of patients, and the effect was durable, with 80% having measured dabigatran levels reflecting no significant anticoagulation 24 hours later, Dr. Pollack said.
“Clinical outcomes were quite good in this multimorbid patient population, with restoration of hemostasis as reported by local investigators achieved in less than 12 hours when assessable, and with 92% of surgical patients being reported as having normal hemostasis at the time of the procedure,” he said.
Idarucizumab was generally well tolerated in the patient population. “There were no serious adverse events related to the reversal agent ... and only one patient experienced a thrombotic complication within 72 hours, and that patient had not been restarted on any antithrombotic medications,” Dr. Pollack said.
“The study is ongoing,” he added, “but these interim results show rather convincingly that idarucizumab completely and safely reverses the anticoagulant effects of dabigatran within minutes.”
In addition, Dr. Pollack said the availability of a specific reversal agent for dabigatran would enhance its safety margin, and thus alleviate the fears of providers who may hesitate to use a NOAC because of the lack of an “antidote.”
“In fact, most such cases can already be successfully and safely managed with general support and ‘tincture of time’ (the half-life of dabigatran is much shorter than that of warfarin), but having a specific ‘go-to’ option could streamline the care of the most significantly compromised patients,” he said.
Dr. Pollack emphasized, however, that idarucizumab is a specific reversal agent for dabigatran, not an antidote. “To me, the latter would imply that idarucizumab immediately stops bleeding associated with active use of dabigatran,” he said.
Providers should realize that while idarucizumab seems capable of removing dabigatran-induced coagulopathy from the list of concerns when managing a patient with serious bleeding or before a “sharp” procedure, bleeding is a multifaceted issue that also may be due to traumatized blood vessels, other causes of coagulopathy such as liver disease, or concurrent use of antiplatelet medications, he said.
“The patient with a serious or life-threatening bleed on dabigatran will likely need additional care to investigate and manage such concerns,” Dr. Pollack said. “But at least idarucizumab can specifically, safely, and rapidly address the primary consideration.
“The safety of anticoagulation therapy with dabigatran is further enhanced with idarucizumab, a specific reversal agent that won’t need to be used often, but the availability of which would be reassuring to prescribers,” he concluded.
Boehringer Ingelheim sponsored RE-VERSE AD. Idarucizumab was given a fast-track status by the Food and Drug Administration, and BI submitted a new drug application in March 2015, according to the company.
Dr. Pollack reported receiving personal fees from BI, Janssen, Daiichi-Sankyo, Bristol-Myers Squibb, and Pfizer. Disclosures for all the investigators are available at NEJM.org.
TORONTO – Idarucizumab is a promising agent that quickly and safely reverses the anticoagulant effects of dabigatran whether the goal is to control serious bleeding or to permit urgent surgery, according to interim results of a multicenter trial.
Idarucizumab is a monoclonal antibody that binds to dabigatran to reverse its activity. The data, presented by Dr. V. Charles Pollack Jr. at the International Society on Thrombosis and Haemostasis congress, involved the first 90 patients of an ongoing trial with a planned enrollment of 300. The data from this trial, called REVERSE-AD, were published online simultaneously with the June 22 presentation at the congress (N. Engl. J. Med 2015 [doi:10.1056/NEJMoa1502000]).
“Non–vitamin K antagonist oral anticoagulants (NOACs) are generally safer than warfarin, and provide similar or improved efficacy in the prevention of stroke in patients with nonvalvular atrial fibrillation and in the prevention and treatment of venous thromboembolism,” Dr. Pollack said in an interview. “Nonetheless, serious bleeding events may occur with NOAC use, and patients taking one of these agents occasionally require urgent surgery or other intervention for which normal hemostasis is required,” added Dr. Pollack, chair of the department of emergency medicine at Pennsylvania Hospital in Philadelphia.
In RE-VERSE AD (a study of the reversal effects of idarucizumab on active dabigatran), the first 90 patients were divided into two distinct groups. Group A, with 51 patients, included those on dabigatran with serious bleeding. Group B, with 39 patients, required reversal of dabigatran for urgent or emergent procedures. In both, idarucizumab provided a median maximum reversal of 100% (95% confidence interval, 100-100) of the anticoagulation effect within 4 hours.
Clotting assays were normalized almost immediately in almost 90% of patients, and the effect was durable, with 80% having measured dabigatran levels reflecting no significant anticoagulation 24 hours later, Dr. Pollack said.
“Clinical outcomes were quite good in this multimorbid patient population, with restoration of hemostasis as reported by local investigators achieved in less than 12 hours when assessable, and with 92% of surgical patients being reported as having normal hemostasis at the time of the procedure,” he said.
Idarucizumab was generally well tolerated in the patient population. “There were no serious adverse events related to the reversal agent ... and only one patient experienced a thrombotic complication within 72 hours, and that patient had not been restarted on any antithrombotic medications,” Dr. Pollack said.
“The study is ongoing,” he added, “but these interim results show rather convincingly that idarucizumab completely and safely reverses the anticoagulant effects of dabigatran within minutes.”
In addition, Dr. Pollack said the availability of a specific reversal agent for dabigatran would enhance its safety margin, and thus alleviate the fears of providers who may hesitate to use a NOAC because of the lack of an “antidote.”
“In fact, most such cases can already be successfully and safely managed with general support and ‘tincture of time’ (the half-life of dabigatran is much shorter than that of warfarin), but having a specific ‘go-to’ option could streamline the care of the most significantly compromised patients,” he said.
Dr. Pollack emphasized, however, that idarucizumab is a specific reversal agent for dabigatran, not an antidote. “To me, the latter would imply that idarucizumab immediately stops bleeding associated with active use of dabigatran,” he said.
Providers should realize that while idarucizumab seems capable of removing dabigatran-induced coagulopathy from the list of concerns when managing a patient with serious bleeding or before a “sharp” procedure, bleeding is a multifaceted issue that also may be due to traumatized blood vessels, other causes of coagulopathy such as liver disease, or concurrent use of antiplatelet medications, he said.
“The patient with a serious or life-threatening bleed on dabigatran will likely need additional care to investigate and manage such concerns,” Dr. Pollack said. “But at least idarucizumab can specifically, safely, and rapidly address the primary consideration.
“The safety of anticoagulation therapy with dabigatran is further enhanced with idarucizumab, a specific reversal agent that won’t need to be used often, but the availability of which would be reassuring to prescribers,” he concluded.
Boehringer Ingelheim sponsored RE-VERSE AD. Idarucizumab was given a fast-track status by the Food and Drug Administration, and BI submitted a new drug application in March 2015, according to the company.
Dr. Pollack reported receiving personal fees from BI, Janssen, Daiichi-Sankyo, Bristol-Myers Squibb, and Pfizer. Disclosures for all the investigators are available at NEJM.org.
AT 2015 ISTH CONGRESS
Key clinical point: The investigational monoclonal antibody idarucizumab reversed the anticoagulant effects of dabigatran.
Major finding: Idarucizumab provided a median maximum dabigatran reversal of 100% (95% CI, 100-100) of the anticoagulation effect within 4 hours in an interim analysis.
Data source: RE-VERSE AD, a prospective cohort study in which 90 patients treated with dabigatran who had uncontrolled bleeding or required emergency surgery or procedures were given 5.0 g idarucizumab.
Disclosures: Boehringer Ingelheim sponsored RE-VERSE AD. Dr. Pollack reported receiving personal fees from Boehringer Ingelheim, Janssen, Daiichi-Sankyo, Bristol-Myers Squibb, and Pfizer. Disclosures for all the investigators are available at NEJM.org.
ICOO: Approach to opioids for cancer pain evolves
BOSTON – Opioid abuse might be as much of a problem in patients with cancer pain as in those who need analgesia for another reason, according to palliative care physicians at the Dana Farber Cancer Institute who outlined their safeguards at the International Conference of Opioids.
“Let’s not lose sight of the fact that the very access to these medications, which can do so much good, is in jeopardy,” said Dr. Douglas E. Brandoff, a palliative-care attending physician at the cancer institute. In the current era of “unprecedented regulation and scrutiny,” Dr. Brandoff said, pain practices in cancer care must evolve “to keep up with the times.”
Evidence that opioid abuse among cancer patients rivals that of other patients prescribed those agents is limited but reasonably consistent, according to Dr. Brandoff. He cited several published studies, including a survey of hospices in which substance abuse and diversion were considered a problem in 38% (J. Palliat. Med. 2013;16:237-42) of patients.
“It’s a little disconcerting. This is hospice, right? This doesn’t happen in hospice, but unfortunately, it does,” Dr. Brandoff reported.
At the cancer institute, a multidisciplinary task force convened in 2013 has now produced numerous specific policies designed to protect patients and institutions from abuse of controlled pain medications. Those steps are not much different from those being increasingly employed in clinics for nonmalignant chronic pain, but they are applied uniformly in essentially every patient – not just those singled out for high risk.
One required step is the implementation of a prescription-monitoring program for every patient started on a narcotic drug in controlled substances schedule II or III, a benzodiazepine, or a department of public health scheduled IV or V controlled substance. Another is the use of a medication management agreement designed to educate patients about the benefits and risks of controlled substances and outline expectations. All patients and their clinicians are required to sign the agreement.
“If we have someone who is imminently dying within hours or days, then, no, I would not impose a management agreement expectation on them or myself,” said Dr. Brandoff, but he said that there are essentially no other exceptions.
The agreement, crafted with nonjudgmental language aimed at clarifying the goals of chronic pain relief, is entered into the medical record. It generally has been well accepted, according to Dr. Lida Nabati, also a palliative care attending physician at the cancer institute, Dr. Nabati, who participated with Dr. Brandoff in presenting the cancer institute’s safeguards, noted that patient resistance to the agreement often is a red flag for potential problems with abuse.
The movement to control opioid abuse in cancer patients is relatively new. At the time that the task force began, Dr. Nabati noted that few other institutions had formal policies in place even though others also were beginning to review their approach. As recently as 2014, a directive from the Department of Veterans Affairs for opioid therapy in chronic pain patients specifically excluded those with cancer, Dr. Nabati reported.
Yet, cancer “does not afford some magical protective effect” from the very same risk factors associated with opioid use in noncancer patients, such as anxiety, depression, or history of substance use, according to Dr. Brandoff. Rather, he suggested that the added stress of a cancer diagnosis could exacerbate those factors.
The implementation of strategies aimed at reducing the risk of opioid abuse in patients with chronic cancer pain is needed and timely, according to Dr. Mellar P. Davis, the co-chair of the 2015 ICOO meeting and director of the palliative medicine fellowship program, Taussig Cancer Institute, Cleveland Clinic.
In an interview, Dr. Davis applauded the types of strategies implemented at the cancer institute, which he believes protect the patient, the physician, and the institution. He believes that the patients might be the greatest beneficiaries when appropriate opioid use permits a gain in quality of life through effective but nondebilitating pain control.
Dr. Brandoff and Dr. Nabati reported having no financial disclosures.
BOSTON – Opioid abuse might be as much of a problem in patients with cancer pain as in those who need analgesia for another reason, according to palliative care physicians at the Dana Farber Cancer Institute who outlined their safeguards at the International Conference of Opioids.
“Let’s not lose sight of the fact that the very access to these medications, which can do so much good, is in jeopardy,” said Dr. Douglas E. Brandoff, a palliative-care attending physician at the cancer institute. In the current era of “unprecedented regulation and scrutiny,” Dr. Brandoff said, pain practices in cancer care must evolve “to keep up with the times.”
Evidence that opioid abuse among cancer patients rivals that of other patients prescribed those agents is limited but reasonably consistent, according to Dr. Brandoff. He cited several published studies, including a survey of hospices in which substance abuse and diversion were considered a problem in 38% (J. Palliat. Med. 2013;16:237-42) of patients.
“It’s a little disconcerting. This is hospice, right? This doesn’t happen in hospice, but unfortunately, it does,” Dr. Brandoff reported.
At the cancer institute, a multidisciplinary task force convened in 2013 has now produced numerous specific policies designed to protect patients and institutions from abuse of controlled pain medications. Those steps are not much different from those being increasingly employed in clinics for nonmalignant chronic pain, but they are applied uniformly in essentially every patient – not just those singled out for high risk.
One required step is the implementation of a prescription-monitoring program for every patient started on a narcotic drug in controlled substances schedule II or III, a benzodiazepine, or a department of public health scheduled IV or V controlled substance. Another is the use of a medication management agreement designed to educate patients about the benefits and risks of controlled substances and outline expectations. All patients and their clinicians are required to sign the agreement.
“If we have someone who is imminently dying within hours or days, then, no, I would not impose a management agreement expectation on them or myself,” said Dr. Brandoff, but he said that there are essentially no other exceptions.
The agreement, crafted with nonjudgmental language aimed at clarifying the goals of chronic pain relief, is entered into the medical record. It generally has been well accepted, according to Dr. Lida Nabati, also a palliative care attending physician at the cancer institute, Dr. Nabati, who participated with Dr. Brandoff in presenting the cancer institute’s safeguards, noted that patient resistance to the agreement often is a red flag for potential problems with abuse.
The movement to control opioid abuse in cancer patients is relatively new. At the time that the task force began, Dr. Nabati noted that few other institutions had formal policies in place even though others also were beginning to review their approach. As recently as 2014, a directive from the Department of Veterans Affairs for opioid therapy in chronic pain patients specifically excluded those with cancer, Dr. Nabati reported.
Yet, cancer “does not afford some magical protective effect” from the very same risk factors associated with opioid use in noncancer patients, such as anxiety, depression, or history of substance use, according to Dr. Brandoff. Rather, he suggested that the added stress of a cancer diagnosis could exacerbate those factors.
The implementation of strategies aimed at reducing the risk of opioid abuse in patients with chronic cancer pain is needed and timely, according to Dr. Mellar P. Davis, the co-chair of the 2015 ICOO meeting and director of the palliative medicine fellowship program, Taussig Cancer Institute, Cleveland Clinic.
In an interview, Dr. Davis applauded the types of strategies implemented at the cancer institute, which he believes protect the patient, the physician, and the institution. He believes that the patients might be the greatest beneficiaries when appropriate opioid use permits a gain in quality of life through effective but nondebilitating pain control.
Dr. Brandoff and Dr. Nabati reported having no financial disclosures.
BOSTON – Opioid abuse might be as much of a problem in patients with cancer pain as in those who need analgesia for another reason, according to palliative care physicians at the Dana Farber Cancer Institute who outlined their safeguards at the International Conference of Opioids.
“Let’s not lose sight of the fact that the very access to these medications, which can do so much good, is in jeopardy,” said Dr. Douglas E. Brandoff, a palliative-care attending physician at the cancer institute. In the current era of “unprecedented regulation and scrutiny,” Dr. Brandoff said, pain practices in cancer care must evolve “to keep up with the times.”
Evidence that opioid abuse among cancer patients rivals that of other patients prescribed those agents is limited but reasonably consistent, according to Dr. Brandoff. He cited several published studies, including a survey of hospices in which substance abuse and diversion were considered a problem in 38% (J. Palliat. Med. 2013;16:237-42) of patients.
“It’s a little disconcerting. This is hospice, right? This doesn’t happen in hospice, but unfortunately, it does,” Dr. Brandoff reported.
At the cancer institute, a multidisciplinary task force convened in 2013 has now produced numerous specific policies designed to protect patients and institutions from abuse of controlled pain medications. Those steps are not much different from those being increasingly employed in clinics for nonmalignant chronic pain, but they are applied uniformly in essentially every patient – not just those singled out for high risk.
One required step is the implementation of a prescription-monitoring program for every patient started on a narcotic drug in controlled substances schedule II or III, a benzodiazepine, or a department of public health scheduled IV or V controlled substance. Another is the use of a medication management agreement designed to educate patients about the benefits and risks of controlled substances and outline expectations. All patients and their clinicians are required to sign the agreement.
“If we have someone who is imminently dying within hours or days, then, no, I would not impose a management agreement expectation on them or myself,” said Dr. Brandoff, but he said that there are essentially no other exceptions.
The agreement, crafted with nonjudgmental language aimed at clarifying the goals of chronic pain relief, is entered into the medical record. It generally has been well accepted, according to Dr. Lida Nabati, also a palliative care attending physician at the cancer institute, Dr. Nabati, who participated with Dr. Brandoff in presenting the cancer institute’s safeguards, noted that patient resistance to the agreement often is a red flag for potential problems with abuse.
The movement to control opioid abuse in cancer patients is relatively new. At the time that the task force began, Dr. Nabati noted that few other institutions had formal policies in place even though others also were beginning to review their approach. As recently as 2014, a directive from the Department of Veterans Affairs for opioid therapy in chronic pain patients specifically excluded those with cancer, Dr. Nabati reported.
Yet, cancer “does not afford some magical protective effect” from the very same risk factors associated with opioid use in noncancer patients, such as anxiety, depression, or history of substance use, according to Dr. Brandoff. Rather, he suggested that the added stress of a cancer diagnosis could exacerbate those factors.
The implementation of strategies aimed at reducing the risk of opioid abuse in patients with chronic cancer pain is needed and timely, according to Dr. Mellar P. Davis, the co-chair of the 2015 ICOO meeting and director of the palliative medicine fellowship program, Taussig Cancer Institute, Cleveland Clinic.
In an interview, Dr. Davis applauded the types of strategies implemented at the cancer institute, which he believes protect the patient, the physician, and the institution. He believes that the patients might be the greatest beneficiaries when appropriate opioid use permits a gain in quality of life through effective but nondebilitating pain control.
Dr. Brandoff and Dr. Nabati reported having no financial disclosures.
EXPERT ANALYSIS AT THE INTERNATIONAL CONFERENCE ON OPIOIDS
Laryngoscopy of vocal cords avoided with ultrasound
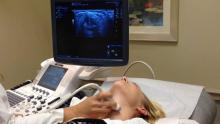
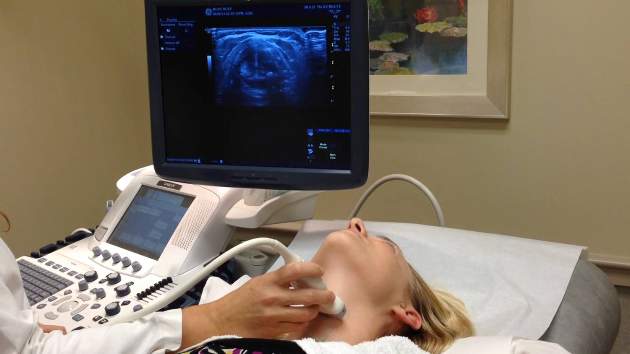
In candidates for thyroid surgery, ultrasound is an effective and convenient method of preoperative vocal cord evaluation, avoiding the costs of laryngoscopy, according to data derived from a retrospective study presented at the annual meeting of the American Association of Endocrine Surgeons in Nashville, Tenn.
In the study, which enrolled only those thyroid or parathyroid patients with risk factors for vocal cord paralysis, who would have been otherwise evaluated with flexible laryngoscopy, vocal cord ultrasonography (VCUS) proved to be an adequate examination in 76%, reported Dr. Denise Carneiro-Pla of the division of oncologic and endocrine surgery at the Medical University of South Carolina, Charleston.
The results have been practice-changing, she said. In thyroid or parathyroid surgical candidates at risk for vocal cord paralysis, flexible laryngoscopy had been the standard of care. When VCUS is adequate for visualization, it permits patients to avoid a separate office visit and an invasive procedure that many experience as uncomfortable. Based on data from this study, cost savings are substantial.
In patients scheduled for thyroid or parathyroid surgery, “ultrasound evaluation is already a routine part of the evaluation,” Dr. Carneiro-Pla explained. An evaluation of the vocal cords is easily performed at the same time, and visualization is typically adequate.
“You either have good visualization or you don’t,” she noted. “In the minority of cases when you do not, the patient can be referred for laryngoscopy.”
This assertion was supported by her study, which included a total of 194 consecutive patients from three institutions who were scheduled for thyroid or parathyroid surgery and had an indication for preoperative evaluation of vocal cord mobility. These indications included previous cervical procedures, a large goiter, significant hoarseness, and thyroid cancer suspected of possible extrathyroidal extension. All patients underwent VCUS, but 52 underwent flexible laryngoscopy regardless of the VCUS findings while 142 only underwent laryngoscopy if the VCUS was considered inadequate.
Overall, VCUS was judged to be adequate for evaluating vocal cord mobility in 164 (85%) of the total study population. In the group of patients for whom laryngoscopy was employed only if VCUS was not considered adequate, 24% did go on to the invasive procedure. The reasons were inadequate visualization in 11%, significant hoarseness despite normal VCUS in 8%, a finding of vocal cord weakness or paresis on VCUS that required further evaluation in 4%, and a surgeon decision or another reason in 1%.
In a comparison to those with adequate VCUS visualization, those without were, on average, older by 4 years and more likely to be male than female. Those with thyroid cartilage calcification were substantially less likely to have adequate visualization on VCUS than those without (57% vs. 92%; P < 0.005). Body mass index did not influence the adequacy of VCUS.
In patients at risk of vocal cord paralysis, Dr. Carneiro-Pla explained that some form of preoperative evaluation is considered essential to reduce the risk of a recurrent laryngeal nerve injury. Several previous studies have suggested that VCUS is accurate for predicting vocal cord paralysis, and this study reinforced this approach in routine patient care.
“An ultrasound evaluation would be performed in most patients anyway, so this avoids an extra step,” said Dr. Carneiro-Pla, whose study included a cost analysis that projected savings for the study group ranging from nearly $8,000 to more than $20,000.
She reported no relevant financial conflicts.
In candidates for thyroid surgery, ultrasound is an effective and convenient method of preoperative vocal cord evaluation, avoiding the costs of laryngoscopy, according to data derived from a retrospective study presented at the annual meeting of the American Association of Endocrine Surgeons in Nashville, Tenn.
In the study, which enrolled only those thyroid or parathyroid patients with risk factors for vocal cord paralysis, who would have been otherwise evaluated with flexible laryngoscopy, vocal cord ultrasonography (VCUS) proved to be an adequate examination in 76%, reported Dr. Denise Carneiro-Pla of the division of oncologic and endocrine surgery at the Medical University of South Carolina, Charleston.
The results have been practice-changing, she said. In thyroid or parathyroid surgical candidates at risk for vocal cord paralysis, flexible laryngoscopy had been the standard of care. When VCUS is adequate for visualization, it permits patients to avoid a separate office visit and an invasive procedure that many experience as uncomfortable. Based on data from this study, cost savings are substantial.
In patients scheduled for thyroid or parathyroid surgery, “ultrasound evaluation is already a routine part of the evaluation,” Dr. Carneiro-Pla explained. An evaluation of the vocal cords is easily performed at the same time, and visualization is typically adequate.
“You either have good visualization or you don’t,” she noted. “In the minority of cases when you do not, the patient can be referred for laryngoscopy.”
This assertion was supported by her study, which included a total of 194 consecutive patients from three institutions who were scheduled for thyroid or parathyroid surgery and had an indication for preoperative evaluation of vocal cord mobility. These indications included previous cervical procedures, a large goiter, significant hoarseness, and thyroid cancer suspected of possible extrathyroidal extension. All patients underwent VCUS, but 52 underwent flexible laryngoscopy regardless of the VCUS findings while 142 only underwent laryngoscopy if the VCUS was considered inadequate.
Overall, VCUS was judged to be adequate for evaluating vocal cord mobility in 164 (85%) of the total study population. In the group of patients for whom laryngoscopy was employed only if VCUS was not considered adequate, 24% did go on to the invasive procedure. The reasons were inadequate visualization in 11%, significant hoarseness despite normal VCUS in 8%, a finding of vocal cord weakness or paresis on VCUS that required further evaluation in 4%, and a surgeon decision or another reason in 1%.
In a comparison to those with adequate VCUS visualization, those without were, on average, older by 4 years and more likely to be male than female. Those with thyroid cartilage calcification were substantially less likely to have adequate visualization on VCUS than those without (57% vs. 92%; P < 0.005). Body mass index did not influence the adequacy of VCUS.
In patients at risk of vocal cord paralysis, Dr. Carneiro-Pla explained that some form of preoperative evaluation is considered essential to reduce the risk of a recurrent laryngeal nerve injury. Several previous studies have suggested that VCUS is accurate for predicting vocal cord paralysis, and this study reinforced this approach in routine patient care.
“An ultrasound evaluation would be performed in most patients anyway, so this avoids an extra step,” said Dr. Carneiro-Pla, whose study included a cost analysis that projected savings for the study group ranging from nearly $8,000 to more than $20,000.
She reported no relevant financial conflicts.
In candidates for thyroid surgery, ultrasound is an effective and convenient method of preoperative vocal cord evaluation, avoiding the costs of laryngoscopy, according to data derived from a retrospective study presented at the annual meeting of the American Association of Endocrine Surgeons in Nashville, Tenn.
In the study, which enrolled only those thyroid or parathyroid patients with risk factors for vocal cord paralysis, who would have been otherwise evaluated with flexible laryngoscopy, vocal cord ultrasonography (VCUS) proved to be an adequate examination in 76%, reported Dr. Denise Carneiro-Pla of the division of oncologic and endocrine surgery at the Medical University of South Carolina, Charleston.
The results have been practice-changing, she said. In thyroid or parathyroid surgical candidates at risk for vocal cord paralysis, flexible laryngoscopy had been the standard of care. When VCUS is adequate for visualization, it permits patients to avoid a separate office visit and an invasive procedure that many experience as uncomfortable. Based on data from this study, cost savings are substantial.
In patients scheduled for thyroid or parathyroid surgery, “ultrasound evaluation is already a routine part of the evaluation,” Dr. Carneiro-Pla explained. An evaluation of the vocal cords is easily performed at the same time, and visualization is typically adequate.
“You either have good visualization or you don’t,” she noted. “In the minority of cases when you do not, the patient can be referred for laryngoscopy.”
This assertion was supported by her study, which included a total of 194 consecutive patients from three institutions who were scheduled for thyroid or parathyroid surgery and had an indication for preoperative evaluation of vocal cord mobility. These indications included previous cervical procedures, a large goiter, significant hoarseness, and thyroid cancer suspected of possible extrathyroidal extension. All patients underwent VCUS, but 52 underwent flexible laryngoscopy regardless of the VCUS findings while 142 only underwent laryngoscopy if the VCUS was considered inadequate.
Overall, VCUS was judged to be adequate for evaluating vocal cord mobility in 164 (85%) of the total study population. In the group of patients for whom laryngoscopy was employed only if VCUS was not considered adequate, 24% did go on to the invasive procedure. The reasons were inadequate visualization in 11%, significant hoarseness despite normal VCUS in 8%, a finding of vocal cord weakness or paresis on VCUS that required further evaluation in 4%, and a surgeon decision or another reason in 1%.
In a comparison to those with adequate VCUS visualization, those without were, on average, older by 4 years and more likely to be male than female. Those with thyroid cartilage calcification were substantially less likely to have adequate visualization on VCUS than those without (57% vs. 92%; P < 0.005). Body mass index did not influence the adequacy of VCUS.
In patients at risk of vocal cord paralysis, Dr. Carneiro-Pla explained that some form of preoperative evaluation is considered essential to reduce the risk of a recurrent laryngeal nerve injury. Several previous studies have suggested that VCUS is accurate for predicting vocal cord paralysis, and this study reinforced this approach in routine patient care.
“An ultrasound evaluation would be performed in most patients anyway, so this avoids an extra step,” said Dr. Carneiro-Pla, whose study included a cost analysis that projected savings for the study group ranging from nearly $8,000 to more than $20,000.
She reported no relevant financial conflicts.
FROM AAES 2015
Key clinical point: If evaluated with ultrasound, surgery patients at risk for vocal cord paralysis can forgo preoperative laryngoscopy.
Major finding: Of candidates for laryngoscopy, 76% avoided this invasive procedure with ultrasound.
Data source: Review of prospectively collected data.
Disclosures: Dr. Carneiro-Pla reported no relevant financial conflicts.
ICOO: Nonspecialist comfort with opioids remains low
BOSTON – The discomfort among primary care physicians managing patients on opioids is not necessarily mitigated with greater support from pain specialists, according to a study that tracked attitudes before and after an initiative specifically designed to increase that support.
The study, presented at the International Conference on Opioids, surveyed 56 primary care clinicians before and after the initiative, according to Robert Jamison, Ph.D., professor in the departments of anesthesia, psychiatry, and physician medicine and rehabilitation at Harvard Medical School in Boston.
The study, according to Dr. Jamison, was based on the observation that primary care physicians are uncomfortable prescribing opioids even after a pain specialist has recommended this therapy.
“A lot of primary care physicians don’t like pain specialists,” Dr. Jamison said. The reason, he explained, is that they are uncomfortable prescribing or managing opioids in patients with chronic pain, and they think that pain specialists provide them with little support.
In a study that involved 56 primary care physicians and 253 chronic pain patients that they referred, a baseline survey showed that less than 20% of the physicians reported formal training in pain management. In addition, 80% reported that they considered treating chronic pain to be a challenge, and nearly 70% reported that they feared prescribing opioids because of the risk of addiction. In addition, 62.5% reported that they were dissatisfied with their past consults with pain specialists.
After the survey, a program was initiated to provide support for opioid management in the referred chronic pain patients. This involved evaluating and initiating a pain management program that included screening patients for risk of opioid misuse. Although all the patients were potential candidates for opioids based on clinical characteristics, only 207 ultimately received opioids.
With patient management, the program also provided the participating primary care physicians with guidance on managing chronic pain with opioids.
The chronic pain patients were contacted monthly by telephone to evaluate compliance and risk of opioid misuse. Follow-up was maintained over 6 months. The 33 primary care physicians linked to the Harvard electronic medical records system were provided with monthly status reports about the opioid patients in their care.
When the primary care physicians were surveyed at the end of 6 months, there was essentially no change in attitude in regard to the challenges of managing patients on opioids, including fear of patient addiction or the physician’s confidence in his or her ability to identify patients at risk of addiction.
Physicians did, however, express improved sense of support from pain specialists. For example, the initial 62.5% rate of dissatisfaction with communication with pain specialists fell to 23.4%. In addition, physician comfort with opioids more than tripled, rising from 7.2% to 23.4%. However, this suggested that 75% of physicians were still uncomfortable.
“So we did get some mixed results on our intervention,” acknowledged Dr. Jamison, who noted that the data from this recently completed study are still being analyzed. He did note that satisfaction overall with pain-specialist communication was greatest in those physicians who received monthly reports.
Based on the data analyzed to date, the findings might be best interpreted “as a heads-up for pain specialists in the room to say that primary care physicians don’t feel that they are getting the support that they need,” Dr. Jamison reported.
The study was investigator initiated. Dr. Jamison reported having no financial disclosures.
BOSTON – The discomfort among primary care physicians managing patients on opioids is not necessarily mitigated with greater support from pain specialists, according to a study that tracked attitudes before and after an initiative specifically designed to increase that support.
The study, presented at the International Conference on Opioids, surveyed 56 primary care clinicians before and after the initiative, according to Robert Jamison, Ph.D., professor in the departments of anesthesia, psychiatry, and physician medicine and rehabilitation at Harvard Medical School in Boston.
The study, according to Dr. Jamison, was based on the observation that primary care physicians are uncomfortable prescribing opioids even after a pain specialist has recommended this therapy.
“A lot of primary care physicians don’t like pain specialists,” Dr. Jamison said. The reason, he explained, is that they are uncomfortable prescribing or managing opioids in patients with chronic pain, and they think that pain specialists provide them with little support.
In a study that involved 56 primary care physicians and 253 chronic pain patients that they referred, a baseline survey showed that less than 20% of the physicians reported formal training in pain management. In addition, 80% reported that they considered treating chronic pain to be a challenge, and nearly 70% reported that they feared prescribing opioids because of the risk of addiction. In addition, 62.5% reported that they were dissatisfied with their past consults with pain specialists.
After the survey, a program was initiated to provide support for opioid management in the referred chronic pain patients. This involved evaluating and initiating a pain management program that included screening patients for risk of opioid misuse. Although all the patients were potential candidates for opioids based on clinical characteristics, only 207 ultimately received opioids.
With patient management, the program also provided the participating primary care physicians with guidance on managing chronic pain with opioids.
The chronic pain patients were contacted monthly by telephone to evaluate compliance and risk of opioid misuse. Follow-up was maintained over 6 months. The 33 primary care physicians linked to the Harvard electronic medical records system were provided with monthly status reports about the opioid patients in their care.
When the primary care physicians were surveyed at the end of 6 months, there was essentially no change in attitude in regard to the challenges of managing patients on opioids, including fear of patient addiction or the physician’s confidence in his or her ability to identify patients at risk of addiction.
Physicians did, however, express improved sense of support from pain specialists. For example, the initial 62.5% rate of dissatisfaction with communication with pain specialists fell to 23.4%. In addition, physician comfort with opioids more than tripled, rising from 7.2% to 23.4%. However, this suggested that 75% of physicians were still uncomfortable.
“So we did get some mixed results on our intervention,” acknowledged Dr. Jamison, who noted that the data from this recently completed study are still being analyzed. He did note that satisfaction overall with pain-specialist communication was greatest in those physicians who received monthly reports.
Based on the data analyzed to date, the findings might be best interpreted “as a heads-up for pain specialists in the room to say that primary care physicians don’t feel that they are getting the support that they need,” Dr. Jamison reported.
The study was investigator initiated. Dr. Jamison reported having no financial disclosures.
BOSTON – The discomfort among primary care physicians managing patients on opioids is not necessarily mitigated with greater support from pain specialists, according to a study that tracked attitudes before and after an initiative specifically designed to increase that support.
The study, presented at the International Conference on Opioids, surveyed 56 primary care clinicians before and after the initiative, according to Robert Jamison, Ph.D., professor in the departments of anesthesia, psychiatry, and physician medicine and rehabilitation at Harvard Medical School in Boston.
The study, according to Dr. Jamison, was based on the observation that primary care physicians are uncomfortable prescribing opioids even after a pain specialist has recommended this therapy.
“A lot of primary care physicians don’t like pain specialists,” Dr. Jamison said. The reason, he explained, is that they are uncomfortable prescribing or managing opioids in patients with chronic pain, and they think that pain specialists provide them with little support.
In a study that involved 56 primary care physicians and 253 chronic pain patients that they referred, a baseline survey showed that less than 20% of the physicians reported formal training in pain management. In addition, 80% reported that they considered treating chronic pain to be a challenge, and nearly 70% reported that they feared prescribing opioids because of the risk of addiction. In addition, 62.5% reported that they were dissatisfied with their past consults with pain specialists.
After the survey, a program was initiated to provide support for opioid management in the referred chronic pain patients. This involved evaluating and initiating a pain management program that included screening patients for risk of opioid misuse. Although all the patients were potential candidates for opioids based on clinical characteristics, only 207 ultimately received opioids.
With patient management, the program also provided the participating primary care physicians with guidance on managing chronic pain with opioids.
The chronic pain patients were contacted monthly by telephone to evaluate compliance and risk of opioid misuse. Follow-up was maintained over 6 months. The 33 primary care physicians linked to the Harvard electronic medical records system were provided with monthly status reports about the opioid patients in their care.
When the primary care physicians were surveyed at the end of 6 months, there was essentially no change in attitude in regard to the challenges of managing patients on opioids, including fear of patient addiction or the physician’s confidence in his or her ability to identify patients at risk of addiction.
Physicians did, however, express improved sense of support from pain specialists. For example, the initial 62.5% rate of dissatisfaction with communication with pain specialists fell to 23.4%. In addition, physician comfort with opioids more than tripled, rising from 7.2% to 23.4%. However, this suggested that 75% of physicians were still uncomfortable.
“So we did get some mixed results on our intervention,” acknowledged Dr. Jamison, who noted that the data from this recently completed study are still being analyzed. He did note that satisfaction overall with pain-specialist communication was greatest in those physicians who received monthly reports.
Based on the data analyzed to date, the findings might be best interpreted “as a heads-up for pain specialists in the room to say that primary care physicians don’t feel that they are getting the support that they need,” Dr. Jamison reported.
The study was investigator initiated. Dr. Jamison reported having no financial disclosures.
AT THE INTERNATIONAL CONFERENCE ON OPIOIDS
Key clinical point: Pain specialists have a long way to go to improve the comfort of primary care specialists with managing patients on opioids, according to a study that tracked one such initiative.
Major finding: After a formal program to provide greater support to primary care physicians, dissatisfaction with pain specialist communication fell from 62.5% to 23.4%, but there was only modest or no improvement in the confidence of physicians in managing chronic pain patients on opioids.
Data source: Prospective study that compared survey responses among 56 participating primary care physicians before and after a support program.
Disclosures: The study was investigator initiated. Dr. Jamison reported having no financial disclosures.
ICOO: Opioid prescribing program yields broad benefits
BOSTON – A patient care process initiated to ensure that opioid prescriptions adhere to high standards of appropriate use and safety has potentially improved the quality of pain management while increasing revenue, according to a detailed analysis of the initiative presented at the International Conference on Opioids.
The process, started 2 years ago in a pilot program, involves a series of steps to evaluate pain patients for their suitability for opioids and then monitor their course of care, said Dr. William G. Brose, an adjunct clinical professor at Stanford (Calif.) University and chief executive officer of the HELP Pain Medical Network.
The process, called the Analgesic Adherence Program (AAP), was constructed out of published guidelines and standards of care, particularly those issued for opioid use by the medical board of the state of California, where the for-profit HELP Pain Medical Network has more than 30 locations. Concern about liability from prescribing opioids was a major impetus for development of the AAP.
“I wanted to try to keep myself safe, to try to keep my patients safe, and to try to keep my practice secure,” Dr. Brose explained.
The process begins with initial risk stratification for which the Medical Board of California specifies the tools. Other states might specify different tools or no tools, although every state has now established recommendations for the use of opioids, except Alaska and Illinois, Dr. Brose said. He said 18 of those guidelines have been updated in the last 4 years, and those following his lead should adhere to state-specific standards.
Subsequent steps include educating patients about the risks and benefits of opioids, seeking informed consent, and then selecting a monitoring program suitable to the patient’s circumstances. Throughout each process, the AAP includes detailed decision trees, including the option to refer patients to specialists if a risk level exceeds the comfort level of the clinician or institution.
Most of these steps, such as risk assessment and seeking informed consent, are billable and performed by nonphysicians. This was captured in an evaluation of 192 new patients and 662 established patients entered into the AAP process over the past 12 months. Relative to usual management, physician time was essentially unchanged, even though time spent by nonphysician clinicians, coders, and billers, rose.
As a result of the AAP, the tasks of the pain specialists have shifted. According to Dr. Brose, his job and the job of other pain specialist physicians in his network is “basically exception management.” This means that Dr. Brose focuses his attention on “the people who are not doing what they are supposed to be doing.”
Those patients, Dr. Brose said, are the more interesting ones, and this approach results in the most efficient use of physician time.
The AAP was created by Dr. Brose for the HELP Pain Medical Network to address growing concern about the medicolegal risks from prescribing opioids. Dr. Brose reported that increased revenue for the network has been a byproduct. Although the implementation of AAP has been associated with some increased costs, average per-year billing for new chronic pain patients evaluated for opioid prescriptions has climbed from $900 to $1,950 per year. For established patients, per-year billing climbed from $600 to $1,300.
“If you go through the effort to establish this kind of process, you’ll increase your revenue and you will be delivering safer, more effective, consistent monitored care,” Dr. Brose said.
Dr. Brose is a stockholder in the HELP Pain Medical Network.
BOSTON – A patient care process initiated to ensure that opioid prescriptions adhere to high standards of appropriate use and safety has potentially improved the quality of pain management while increasing revenue, according to a detailed analysis of the initiative presented at the International Conference on Opioids.
The process, started 2 years ago in a pilot program, involves a series of steps to evaluate pain patients for their suitability for opioids and then monitor their course of care, said Dr. William G. Brose, an adjunct clinical professor at Stanford (Calif.) University and chief executive officer of the HELP Pain Medical Network.
The process, called the Analgesic Adherence Program (AAP), was constructed out of published guidelines and standards of care, particularly those issued for opioid use by the medical board of the state of California, where the for-profit HELP Pain Medical Network has more than 30 locations. Concern about liability from prescribing opioids was a major impetus for development of the AAP.
“I wanted to try to keep myself safe, to try to keep my patients safe, and to try to keep my practice secure,” Dr. Brose explained.
The process begins with initial risk stratification for which the Medical Board of California specifies the tools. Other states might specify different tools or no tools, although every state has now established recommendations for the use of opioids, except Alaska and Illinois, Dr. Brose said. He said 18 of those guidelines have been updated in the last 4 years, and those following his lead should adhere to state-specific standards.
Subsequent steps include educating patients about the risks and benefits of opioids, seeking informed consent, and then selecting a monitoring program suitable to the patient’s circumstances. Throughout each process, the AAP includes detailed decision trees, including the option to refer patients to specialists if a risk level exceeds the comfort level of the clinician or institution.
Most of these steps, such as risk assessment and seeking informed consent, are billable and performed by nonphysicians. This was captured in an evaluation of 192 new patients and 662 established patients entered into the AAP process over the past 12 months. Relative to usual management, physician time was essentially unchanged, even though time spent by nonphysician clinicians, coders, and billers, rose.
As a result of the AAP, the tasks of the pain specialists have shifted. According to Dr. Brose, his job and the job of other pain specialist physicians in his network is “basically exception management.” This means that Dr. Brose focuses his attention on “the people who are not doing what they are supposed to be doing.”
Those patients, Dr. Brose said, are the more interesting ones, and this approach results in the most efficient use of physician time.
The AAP was created by Dr. Brose for the HELP Pain Medical Network to address growing concern about the medicolegal risks from prescribing opioids. Dr. Brose reported that increased revenue for the network has been a byproduct. Although the implementation of AAP has been associated with some increased costs, average per-year billing for new chronic pain patients evaluated for opioid prescriptions has climbed from $900 to $1,950 per year. For established patients, per-year billing climbed from $600 to $1,300.
“If you go through the effort to establish this kind of process, you’ll increase your revenue and you will be delivering safer, more effective, consistent monitored care,” Dr. Brose said.
Dr. Brose is a stockholder in the HELP Pain Medical Network.
BOSTON – A patient care process initiated to ensure that opioid prescriptions adhere to high standards of appropriate use and safety has potentially improved the quality of pain management while increasing revenue, according to a detailed analysis of the initiative presented at the International Conference on Opioids.
The process, started 2 years ago in a pilot program, involves a series of steps to evaluate pain patients for their suitability for opioids and then monitor their course of care, said Dr. William G. Brose, an adjunct clinical professor at Stanford (Calif.) University and chief executive officer of the HELP Pain Medical Network.
The process, called the Analgesic Adherence Program (AAP), was constructed out of published guidelines and standards of care, particularly those issued for opioid use by the medical board of the state of California, where the for-profit HELP Pain Medical Network has more than 30 locations. Concern about liability from prescribing opioids was a major impetus for development of the AAP.
“I wanted to try to keep myself safe, to try to keep my patients safe, and to try to keep my practice secure,” Dr. Brose explained.
The process begins with initial risk stratification for which the Medical Board of California specifies the tools. Other states might specify different tools or no tools, although every state has now established recommendations for the use of opioids, except Alaska and Illinois, Dr. Brose said. He said 18 of those guidelines have been updated in the last 4 years, and those following his lead should adhere to state-specific standards.
Subsequent steps include educating patients about the risks and benefits of opioids, seeking informed consent, and then selecting a monitoring program suitable to the patient’s circumstances. Throughout each process, the AAP includes detailed decision trees, including the option to refer patients to specialists if a risk level exceeds the comfort level of the clinician or institution.
Most of these steps, such as risk assessment and seeking informed consent, are billable and performed by nonphysicians. This was captured in an evaluation of 192 new patients and 662 established patients entered into the AAP process over the past 12 months. Relative to usual management, physician time was essentially unchanged, even though time spent by nonphysician clinicians, coders, and billers, rose.
As a result of the AAP, the tasks of the pain specialists have shifted. According to Dr. Brose, his job and the job of other pain specialist physicians in his network is “basically exception management.” This means that Dr. Brose focuses his attention on “the people who are not doing what they are supposed to be doing.”
Those patients, Dr. Brose said, are the more interesting ones, and this approach results in the most efficient use of physician time.
The AAP was created by Dr. Brose for the HELP Pain Medical Network to address growing concern about the medicolegal risks from prescribing opioids. Dr. Brose reported that increased revenue for the network has been a byproduct. Although the implementation of AAP has been associated with some increased costs, average per-year billing for new chronic pain patients evaluated for opioid prescriptions has climbed from $900 to $1,950 per year. For established patients, per-year billing climbed from $600 to $1,300.
“If you go through the effort to establish this kind of process, you’ll increase your revenue and you will be delivering safer, more effective, consistent monitored care,” Dr. Brose said.
Dr. Brose is a stockholder in the HELP Pain Medical Network.
AT THE INTERNATIONAL CONFERENCE ON OPIOIDS
Key clinical point: A comprehensive, step-by-step program aimed at selecting and managing chronic pain candidates initiating opioids has generated new income for a network of pain clinics even as it improves patient safety and reduces clinician liability.
Major finding: The Analgesia Adherence Program (AAP) has been credited with improving guideline adherence, reducing medicolegal risks, and doubling per-patient revenue.
Data source: Prospective review of pilot analgesia program.
Disclosures: Dr. Brose is a stockholder in the HELP Pain Medical Network.
Pharmacogenomics for pain meds promising but not ready
BOSTON – Genetic tests for pain treatment efficacy are being marketed in the United States and Australia, but one expert argues that they are not ready for use in clinical practice.
There is evidence that variability in patient response to opioids is due to pharmacogenomics that define drug metabolism and receptor activity, but it is just one factor that cannot yet be teased out from others, according to a summary by one expert at the International Conference on Opioids.
“My view is that commercialization of current genetic tests of pain treatment efficacy with opioids is somewhat premature. Evidence is limited, and I think regulation is necessary,” said Dr. Andrew Somogyi, professor in clinical and experimental pharmacology, University of Adelaide (Australia).
Progress has been made in identifying genetic factors that influence response to opioids, but there are two barriers to clinically useful tools, according to Dr. Somogyi. One is that clinical utility has yet to be proven for any test in a well-designed trial. The other is that even if pharmacogenomics features are relevant, the quality of commercial tests must be validated.
There are now at least two companies marketing genetic tests to guide selection of analgesics in the United States as well as one in Australia, but Dr. Somogyi said that neither the U.S. Food and Drug Administration nor the Australian Therapeutic Goods Administration provides any regulatory oversight. He noted that prices for the testing vary, but he believes that there is no evidence that analgesia can be improved by acting on test results.
As an example, he noted that the company in Australia employs genetic predictors of CYP2D6 function, an enzyme important to opioid metabolism, to estimate benefit from this class of analgesics. However, published studies have not yet generated compelling evidence that this is clinically relevant for either improving pain control or avoiding adverse events.
In the case of oxycodone, studies comparing poor metabolizers to ultra-rapid metabolizers do show important differences in experimental models of pain, but “clinical studies have really shown no effect,” Dr. Somogyi said.
He drew the same conclusion about CYP2D6 function in regard to the effects of codeine and the variants in the OPRM1 gene and their effect on the Mu receptor and response to morphine. In all cases, there are studies to suggest genetic differences are meaningful in experimental models, but no evidence from a clinical study to show that acting on this information changes outcome or otherwise improves care.
Although he acknowledged that it is tempting to believe pharmacogenetics can explain the well-known variability in response to analgesics, “other factors are just swamping these pharmacogenetics variables,” Dr. Somogyi maintained. Not least of these other factors are the psychosocial factors that influence response to pain stimuli.
This does not preclude an eventual role for pharmacogenetics in the selection of analgesics. Dr. Somogyi, who has been working in this area for several decades, outlined a broad array of promising areas of research. It is the current clinical application of pharmacogenetics with which Dr. Somogyi took issue. He criticized many of the marketing claims made on behalf of available tests, which he considers unsubstantiated.
Deferring to the expertise of Dr. Somogyi, Dr. Paul A. Sloan, professor and vice chair for research, University of Kentucky, Lexington, said he found the perspective compelling. In an interview, Dr. Sloan, cochair of the 2015 ICOO meeting, reported that he knows of no one using pharmacogenetics testing to guide analgesic choice, and he reiterated Dr. Somogyi’s concerns about the need to validate the quality of tests as a part of an effort to confirm their viability as clinical tools.
Dr. Somogyi reported having no financial disclosures.
BOSTON – Genetic tests for pain treatment efficacy are being marketed in the United States and Australia, but one expert argues that they are not ready for use in clinical practice.
There is evidence that variability in patient response to opioids is due to pharmacogenomics that define drug metabolism and receptor activity, but it is just one factor that cannot yet be teased out from others, according to a summary by one expert at the International Conference on Opioids.
“My view is that commercialization of current genetic tests of pain treatment efficacy with opioids is somewhat premature. Evidence is limited, and I think regulation is necessary,” said Dr. Andrew Somogyi, professor in clinical and experimental pharmacology, University of Adelaide (Australia).
Progress has been made in identifying genetic factors that influence response to opioids, but there are two barriers to clinically useful tools, according to Dr. Somogyi. One is that clinical utility has yet to be proven for any test in a well-designed trial. The other is that even if pharmacogenomics features are relevant, the quality of commercial tests must be validated.
There are now at least two companies marketing genetic tests to guide selection of analgesics in the United States as well as one in Australia, but Dr. Somogyi said that neither the U.S. Food and Drug Administration nor the Australian Therapeutic Goods Administration provides any regulatory oversight. He noted that prices for the testing vary, but he believes that there is no evidence that analgesia can be improved by acting on test results.
As an example, he noted that the company in Australia employs genetic predictors of CYP2D6 function, an enzyme important to opioid metabolism, to estimate benefit from this class of analgesics. However, published studies have not yet generated compelling evidence that this is clinically relevant for either improving pain control or avoiding adverse events.
In the case of oxycodone, studies comparing poor metabolizers to ultra-rapid metabolizers do show important differences in experimental models of pain, but “clinical studies have really shown no effect,” Dr. Somogyi said.
He drew the same conclusion about CYP2D6 function in regard to the effects of codeine and the variants in the OPRM1 gene and their effect on the Mu receptor and response to morphine. In all cases, there are studies to suggest genetic differences are meaningful in experimental models, but no evidence from a clinical study to show that acting on this information changes outcome or otherwise improves care.
Although he acknowledged that it is tempting to believe pharmacogenetics can explain the well-known variability in response to analgesics, “other factors are just swamping these pharmacogenetics variables,” Dr. Somogyi maintained. Not least of these other factors are the psychosocial factors that influence response to pain stimuli.
This does not preclude an eventual role for pharmacogenetics in the selection of analgesics. Dr. Somogyi, who has been working in this area for several decades, outlined a broad array of promising areas of research. It is the current clinical application of pharmacogenetics with which Dr. Somogyi took issue. He criticized many of the marketing claims made on behalf of available tests, which he considers unsubstantiated.
Deferring to the expertise of Dr. Somogyi, Dr. Paul A. Sloan, professor and vice chair for research, University of Kentucky, Lexington, said he found the perspective compelling. In an interview, Dr. Sloan, cochair of the 2015 ICOO meeting, reported that he knows of no one using pharmacogenetics testing to guide analgesic choice, and he reiterated Dr. Somogyi’s concerns about the need to validate the quality of tests as a part of an effort to confirm their viability as clinical tools.
Dr. Somogyi reported having no financial disclosures.
BOSTON – Genetic tests for pain treatment efficacy are being marketed in the United States and Australia, but one expert argues that they are not ready for use in clinical practice.
There is evidence that variability in patient response to opioids is due to pharmacogenomics that define drug metabolism and receptor activity, but it is just one factor that cannot yet be teased out from others, according to a summary by one expert at the International Conference on Opioids.
“My view is that commercialization of current genetic tests of pain treatment efficacy with opioids is somewhat premature. Evidence is limited, and I think regulation is necessary,” said Dr. Andrew Somogyi, professor in clinical and experimental pharmacology, University of Adelaide (Australia).
Progress has been made in identifying genetic factors that influence response to opioids, but there are two barriers to clinically useful tools, according to Dr. Somogyi. One is that clinical utility has yet to be proven for any test in a well-designed trial. The other is that even if pharmacogenomics features are relevant, the quality of commercial tests must be validated.
There are now at least two companies marketing genetic tests to guide selection of analgesics in the United States as well as one in Australia, but Dr. Somogyi said that neither the U.S. Food and Drug Administration nor the Australian Therapeutic Goods Administration provides any regulatory oversight. He noted that prices for the testing vary, but he believes that there is no evidence that analgesia can be improved by acting on test results.
As an example, he noted that the company in Australia employs genetic predictors of CYP2D6 function, an enzyme important to opioid metabolism, to estimate benefit from this class of analgesics. However, published studies have not yet generated compelling evidence that this is clinically relevant for either improving pain control or avoiding adverse events.
In the case of oxycodone, studies comparing poor metabolizers to ultra-rapid metabolizers do show important differences in experimental models of pain, but “clinical studies have really shown no effect,” Dr. Somogyi said.
He drew the same conclusion about CYP2D6 function in regard to the effects of codeine and the variants in the OPRM1 gene and their effect on the Mu receptor and response to morphine. In all cases, there are studies to suggest genetic differences are meaningful in experimental models, but no evidence from a clinical study to show that acting on this information changes outcome or otherwise improves care.
Although he acknowledged that it is tempting to believe pharmacogenetics can explain the well-known variability in response to analgesics, “other factors are just swamping these pharmacogenetics variables,” Dr. Somogyi maintained. Not least of these other factors are the psychosocial factors that influence response to pain stimuli.
This does not preclude an eventual role for pharmacogenetics in the selection of analgesics. Dr. Somogyi, who has been working in this area for several decades, outlined a broad array of promising areas of research. It is the current clinical application of pharmacogenetics with which Dr. Somogyi took issue. He criticized many of the marketing claims made on behalf of available tests, which he considers unsubstantiated.
Deferring to the expertise of Dr. Somogyi, Dr. Paul A. Sloan, professor and vice chair for research, University of Kentucky, Lexington, said he found the perspective compelling. In an interview, Dr. Sloan, cochair of the 2015 ICOO meeting, reported that he knows of no one using pharmacogenetics testing to guide analgesic choice, and he reiterated Dr. Somogyi’s concerns about the need to validate the quality of tests as a part of an effort to confirm their viability as clinical tools.
Dr. Somogyi reported having no financial disclosures.
EXPERT ANALYSIS AT ICOO2015
ICOO: Massachusetts governor takes on opioid epidemic
BOSTON – A campaign promise to create viable strategies aimed at reducing the overuse of prescription opioids will be fulfilled, the governor of Massachusetts said during a keynote address at the International Conference on Opioids.
Without providing many specifics, Gov. Charles D. Baker (R) reported that the task force he created will soon release recommendations around three major components: 1. prevention and education; 2. addiction and intervention; and 3. the treatment of pain. He emphasized the importance of a comprehensive strategy.
“I am smart enough to know or humble enough to know that any one initiative in regard to this type of thing is unlikely to get us to where we need to go,” Gov. Baker said. Decriminalizing addiction will be part of the strategy aimed at reducing roadblocks to treatment, he said.
The opioid epidemic was not an initial part of Gov. Baker’s 2014 platform in his campaign for governor. Rather, he added a promise to address this problem in his inaugural address after voters expressed their concern repeatedly. He reported being “astonished” by how often he heard stories of addiction to prescription opioids.
“It didn’t matter where I was. It didn’t matter what part of the commonwealth I was in. It did not matter what kind of neighborhood I was in. It didn’t matter what the socioeconomic demographic makeup was of the group I was talking to,” Gov. Baker said. “It was literally everywhere.”
He said hard numbers tell the same story. In addition to the often repeated national statistics regarding the rapid increase in opioid prescriptions and its close correlation with opioid-related deaths, he said no area of Massachusetts has been left untouched. According to Gov. Baker, the largest hospital in the town of Plymouth, Mass., which has a population just under 60,000, is now averaging one opioid overdose per day.
“I am one of those people who thinks that you cannot solve this problem if you do not engage the health care community,” Gov. Baker said. One reason to focus on health care providers, the governor said, is that data from the Substance Abuse and Mental Health Services Administration show that a large percentage of patients addicted to heroin started on prescription pain medication.
The thrust of the coming initiatives is therefore likely to advocate pain medicine practice that reduces inappropriate prescriptions, Gov. Baker said. He noted that an initiative started 2 years ago by Blue Cross Blue Shield of Massachusetts resulted in a 25% reduction in opioid prescribing without restricting pain medications for those who need them.
In general, the address, which was heavily covered by the local news media, was well-received by those who attended the International Conference on Opioids. There was one area of disagreement. Many did not believe that most heroin addictions start with a prescription pain medication. Of those who challenged that assertion, Dr. Carol A. Warfield, the Lowenstein Distinguished Professor of Anesthesia at Harvard Medical School, Boston, clarified that those opioids might have been prescription drugs but were not prescribed to the user. Rather, the drugs were diverted, and this is a much more important focus of efforts to halt the epidemic than new restrictions on their use for pain control.
Similarly, Pamela J. Madej, a registered nurse affiliated with the Albany Center for Pain Management, New York, cautioned against overemphasizing the value of regulating opioids. She said plenty of regulations exist in New York but the problem persists. Many people are self-medicating for mental health disorders, she said, including the chronic brain disorder of addiction, for which coverage continues to be limited.
“Patients switch from hydrocodone to heroin, because heroin is cheaper,” Ms. Madej said. “We need expanded coverage for mental health to address the underlying problem.”
BOSTON – A campaign promise to create viable strategies aimed at reducing the overuse of prescription opioids will be fulfilled, the governor of Massachusetts said during a keynote address at the International Conference on Opioids.
Without providing many specifics, Gov. Charles D. Baker (R) reported that the task force he created will soon release recommendations around three major components: 1. prevention and education; 2. addiction and intervention; and 3. the treatment of pain. He emphasized the importance of a comprehensive strategy.
“I am smart enough to know or humble enough to know that any one initiative in regard to this type of thing is unlikely to get us to where we need to go,” Gov. Baker said. Decriminalizing addiction will be part of the strategy aimed at reducing roadblocks to treatment, he said.
The opioid epidemic was not an initial part of Gov. Baker’s 2014 platform in his campaign for governor. Rather, he added a promise to address this problem in his inaugural address after voters expressed their concern repeatedly. He reported being “astonished” by how often he heard stories of addiction to prescription opioids.
“It didn’t matter where I was. It didn’t matter what part of the commonwealth I was in. It did not matter what kind of neighborhood I was in. It didn’t matter what the socioeconomic demographic makeup was of the group I was talking to,” Gov. Baker said. “It was literally everywhere.”
He said hard numbers tell the same story. In addition to the often repeated national statistics regarding the rapid increase in opioid prescriptions and its close correlation with opioid-related deaths, he said no area of Massachusetts has been left untouched. According to Gov. Baker, the largest hospital in the town of Plymouth, Mass., which has a population just under 60,000, is now averaging one opioid overdose per day.
“I am one of those people who thinks that you cannot solve this problem if you do not engage the health care community,” Gov. Baker said. One reason to focus on health care providers, the governor said, is that data from the Substance Abuse and Mental Health Services Administration show that a large percentage of patients addicted to heroin started on prescription pain medication.
The thrust of the coming initiatives is therefore likely to advocate pain medicine practice that reduces inappropriate prescriptions, Gov. Baker said. He noted that an initiative started 2 years ago by Blue Cross Blue Shield of Massachusetts resulted in a 25% reduction in opioid prescribing without restricting pain medications for those who need them.
In general, the address, which was heavily covered by the local news media, was well-received by those who attended the International Conference on Opioids. There was one area of disagreement. Many did not believe that most heroin addictions start with a prescription pain medication. Of those who challenged that assertion, Dr. Carol A. Warfield, the Lowenstein Distinguished Professor of Anesthesia at Harvard Medical School, Boston, clarified that those opioids might have been prescription drugs but were not prescribed to the user. Rather, the drugs were diverted, and this is a much more important focus of efforts to halt the epidemic than new restrictions on their use for pain control.
Similarly, Pamela J. Madej, a registered nurse affiliated with the Albany Center for Pain Management, New York, cautioned against overemphasizing the value of regulating opioids. She said plenty of regulations exist in New York but the problem persists. Many people are self-medicating for mental health disorders, she said, including the chronic brain disorder of addiction, for which coverage continues to be limited.
“Patients switch from hydrocodone to heroin, because heroin is cheaper,” Ms. Madej said. “We need expanded coverage for mental health to address the underlying problem.”
BOSTON – A campaign promise to create viable strategies aimed at reducing the overuse of prescription opioids will be fulfilled, the governor of Massachusetts said during a keynote address at the International Conference on Opioids.
Without providing many specifics, Gov. Charles D. Baker (R) reported that the task force he created will soon release recommendations around three major components: 1. prevention and education; 2. addiction and intervention; and 3. the treatment of pain. He emphasized the importance of a comprehensive strategy.
“I am smart enough to know or humble enough to know that any one initiative in regard to this type of thing is unlikely to get us to where we need to go,” Gov. Baker said. Decriminalizing addiction will be part of the strategy aimed at reducing roadblocks to treatment, he said.
The opioid epidemic was not an initial part of Gov. Baker’s 2014 platform in his campaign for governor. Rather, he added a promise to address this problem in his inaugural address after voters expressed their concern repeatedly. He reported being “astonished” by how often he heard stories of addiction to prescription opioids.
“It didn’t matter where I was. It didn’t matter what part of the commonwealth I was in. It did not matter what kind of neighborhood I was in. It didn’t matter what the socioeconomic demographic makeup was of the group I was talking to,” Gov. Baker said. “It was literally everywhere.”
He said hard numbers tell the same story. In addition to the often repeated national statistics regarding the rapid increase in opioid prescriptions and its close correlation with opioid-related deaths, he said no area of Massachusetts has been left untouched. According to Gov. Baker, the largest hospital in the town of Plymouth, Mass., which has a population just under 60,000, is now averaging one opioid overdose per day.
“I am one of those people who thinks that you cannot solve this problem if you do not engage the health care community,” Gov. Baker said. One reason to focus on health care providers, the governor said, is that data from the Substance Abuse and Mental Health Services Administration show that a large percentage of patients addicted to heroin started on prescription pain medication.
The thrust of the coming initiatives is therefore likely to advocate pain medicine practice that reduces inappropriate prescriptions, Gov. Baker said. He noted that an initiative started 2 years ago by Blue Cross Blue Shield of Massachusetts resulted in a 25% reduction in opioid prescribing without restricting pain medications for those who need them.
In general, the address, which was heavily covered by the local news media, was well-received by those who attended the International Conference on Opioids. There was one area of disagreement. Many did not believe that most heroin addictions start with a prescription pain medication. Of those who challenged that assertion, Dr. Carol A. Warfield, the Lowenstein Distinguished Professor of Anesthesia at Harvard Medical School, Boston, clarified that those opioids might have been prescription drugs but were not prescribed to the user. Rather, the drugs were diverted, and this is a much more important focus of efforts to halt the epidemic than new restrictions on their use for pain control.
Similarly, Pamela J. Madej, a registered nurse affiliated with the Albany Center for Pain Management, New York, cautioned against overemphasizing the value of regulating opioids. She said plenty of regulations exist in New York but the problem persists. Many people are self-medicating for mental health disorders, she said, including the chronic brain disorder of addiction, for which coverage continues to be limited.
“Patients switch from hydrocodone to heroin, because heroin is cheaper,” Ms. Madej said. “We need expanded coverage for mental health to address the underlying problem.”
ICOO: Opioid self-dosing falls short of pain control
BOSTON – Many cancer patients do not pursue or at least do not achieve complete freedom from pain when permitted control over their opioid dose, according to a comprehensive analysis of published studies that evaluated patient-controlled analgesia.
“We do not know why. Patients were encouraged in these studies to titrate opioids until they were pain free or until they had side effects. Although this could be an issue of side effects, another interpretation is that complete pain control is not the goal for many individuals,” reported Dr. Brian H. Wetherington of the University of Kentucky, Lexington.
The data from this analysis were presented at the International Conference on Opioids from a comprehensive literature search that included 905 potentially relevant articles. Of these, 62 met inclusion criteria, particularly an assessment of patient-controlled opioids in patients with cancer pain. The studies also had to assess pain control with a visual analog scale (VAS) or the Neuropathy Pain Scale (NPS) using a 10-point system with 10 being the greatest level of pain imaginable.
“We were interested in evaluating whether patients, when given complete control over their opioids, would take sufficient doses to provide complete pain relief, which is often stated as the goal in pain management,” explained Dr. Wetherington, who was coauthor of a study led by his colleague at University of Kentucky, Dr. Michael Harned.
The answer was no. When the data from the 62 studies, which included 5,251 patients with cancer pain were collated, the average pain score at baseline was 5.4. At the time of assessment of pain control, the mean pain score was 2.7.
“The mean pain score for patients managing their own cancer pain on opioids was reduced from study entry but remained at the moderate to severe pain level or higher than what many health care providers would recommend,” Dr. Wetherington reported.
This review of published studies does not explain why lower pain scores are not reached, but the Dr. Wetherington and his coauthors hypothesized that patients are demonstrating their own benefit-to-risk ratio assessment.
This is thought to be the first systematic review to find that patients do not seek complete control of pain when given access to unrestricted analgesia, but several individual studies have made the same point. In one study cited by the authors, patients on a fentanyl patch only reduced their pain scores to 3.0 on average when given unlimited access to oral morphine for breakthroughs (J. Pain Symptom Manage. 1998;16:102-11).
“We think this deserves further study, because there may be lessons regarding how we think of optimal pain control. While the therapeutic target is often described as complete pain relief, these data suggest that this may not be the goal for patients when they are left to select their own level of pain control,” Dr. Wetherington explained.
The same observation regarding the failure of patients to eliminate all pain on patient-controlled analgesia has been made anecdotally by Dr. William G. Brose of Stanford (Calif.) University. However, he suggested in an interview that patients might be reluctant to rate themselves completely pain free on a subjective scale. He also believes that level of analgesia may not be the most relevant endpoint.
“We are increasingly evaluating change in patient function, which may be a more useful tool for evaluating the efficacy of pain control,” Dr. Brose said.
BOSTON – Many cancer patients do not pursue or at least do not achieve complete freedom from pain when permitted control over their opioid dose, according to a comprehensive analysis of published studies that evaluated patient-controlled analgesia.
“We do not know why. Patients were encouraged in these studies to titrate opioids until they were pain free or until they had side effects. Although this could be an issue of side effects, another interpretation is that complete pain control is not the goal for many individuals,” reported Dr. Brian H. Wetherington of the University of Kentucky, Lexington.
The data from this analysis were presented at the International Conference on Opioids from a comprehensive literature search that included 905 potentially relevant articles. Of these, 62 met inclusion criteria, particularly an assessment of patient-controlled opioids in patients with cancer pain. The studies also had to assess pain control with a visual analog scale (VAS) or the Neuropathy Pain Scale (NPS) using a 10-point system with 10 being the greatest level of pain imaginable.
“We were interested in evaluating whether patients, when given complete control over their opioids, would take sufficient doses to provide complete pain relief, which is often stated as the goal in pain management,” explained Dr. Wetherington, who was coauthor of a study led by his colleague at University of Kentucky, Dr. Michael Harned.
The answer was no. When the data from the 62 studies, which included 5,251 patients with cancer pain were collated, the average pain score at baseline was 5.4. At the time of assessment of pain control, the mean pain score was 2.7.
“The mean pain score for patients managing their own cancer pain on opioids was reduced from study entry but remained at the moderate to severe pain level or higher than what many health care providers would recommend,” Dr. Wetherington reported.
This review of published studies does not explain why lower pain scores are not reached, but the Dr. Wetherington and his coauthors hypothesized that patients are demonstrating their own benefit-to-risk ratio assessment.
This is thought to be the first systematic review to find that patients do not seek complete control of pain when given access to unrestricted analgesia, but several individual studies have made the same point. In one study cited by the authors, patients on a fentanyl patch only reduced their pain scores to 3.0 on average when given unlimited access to oral morphine for breakthroughs (J. Pain Symptom Manage. 1998;16:102-11).
“We think this deserves further study, because there may be lessons regarding how we think of optimal pain control. While the therapeutic target is often described as complete pain relief, these data suggest that this may not be the goal for patients when they are left to select their own level of pain control,” Dr. Wetherington explained.
The same observation regarding the failure of patients to eliminate all pain on patient-controlled analgesia has been made anecdotally by Dr. William G. Brose of Stanford (Calif.) University. However, he suggested in an interview that patients might be reluctant to rate themselves completely pain free on a subjective scale. He also believes that level of analgesia may not be the most relevant endpoint.
“We are increasingly evaluating change in patient function, which may be a more useful tool for evaluating the efficacy of pain control,” Dr. Brose said.
BOSTON – Many cancer patients do not pursue or at least do not achieve complete freedom from pain when permitted control over their opioid dose, according to a comprehensive analysis of published studies that evaluated patient-controlled analgesia.
“We do not know why. Patients were encouraged in these studies to titrate opioids until they were pain free or until they had side effects. Although this could be an issue of side effects, another interpretation is that complete pain control is not the goal for many individuals,” reported Dr. Brian H. Wetherington of the University of Kentucky, Lexington.
The data from this analysis were presented at the International Conference on Opioids from a comprehensive literature search that included 905 potentially relevant articles. Of these, 62 met inclusion criteria, particularly an assessment of patient-controlled opioids in patients with cancer pain. The studies also had to assess pain control with a visual analog scale (VAS) or the Neuropathy Pain Scale (NPS) using a 10-point system with 10 being the greatest level of pain imaginable.
“We were interested in evaluating whether patients, when given complete control over their opioids, would take sufficient doses to provide complete pain relief, which is often stated as the goal in pain management,” explained Dr. Wetherington, who was coauthor of a study led by his colleague at University of Kentucky, Dr. Michael Harned.
The answer was no. When the data from the 62 studies, which included 5,251 patients with cancer pain were collated, the average pain score at baseline was 5.4. At the time of assessment of pain control, the mean pain score was 2.7.
“The mean pain score for patients managing their own cancer pain on opioids was reduced from study entry but remained at the moderate to severe pain level or higher than what many health care providers would recommend,” Dr. Wetherington reported.
This review of published studies does not explain why lower pain scores are not reached, but the Dr. Wetherington and his coauthors hypothesized that patients are demonstrating their own benefit-to-risk ratio assessment.
This is thought to be the first systematic review to find that patients do not seek complete control of pain when given access to unrestricted analgesia, but several individual studies have made the same point. In one study cited by the authors, patients on a fentanyl patch only reduced their pain scores to 3.0 on average when given unlimited access to oral morphine for breakthroughs (J. Pain Symptom Manage. 1998;16:102-11).
“We think this deserves further study, because there may be lessons regarding how we think of optimal pain control. While the therapeutic target is often described as complete pain relief, these data suggest that this may not be the goal for patients when they are left to select their own level of pain control,” Dr. Wetherington explained.
The same observation regarding the failure of patients to eliminate all pain on patient-controlled analgesia has been made anecdotally by Dr. William G. Brose of Stanford (Calif.) University. However, he suggested in an interview that patients might be reluctant to rate themselves completely pain free on a subjective scale. He also believes that level of analgesia may not be the most relevant endpoint.
“We are increasingly evaluating change in patient function, which may be a more useful tool for evaluating the efficacy of pain control,” Dr. Brose said.
AT ICOO 2015
Key clinical point: Given the opportunity, patients do not titrate opioid therapy to a point of complete pain control, according to a comprehensive survey of published studies.
Major finding: In a survey of 62 published studies of cancer patients who were provided unlimited access to opioids for pain control, the average pain control was 2.7 on a scale of 10, indicating that most patients do not seek or are unable to achieve complete control with an acceptable benefit-to-risk ratio.
Data source: Retrospective data review.
Disclosures: The study was investigator initiated. Dr. Wetherington reported having no financial disclosures.
Cancer risk from IBD mitigated with colonoscopy surveillance
PHILADELPHIA - The risk of both colorectal cancer and CRC-related mortality can be reduced in patients with inflammatory bowel disease by adhering to guideline-directed colonoscopy surveillance recommendations, according to a large retrospective cohort study presented at the annual meeting of the American College of Gastroenterology.
After adjustment for age, sex, duration of disease, type of IBD, and coexisting primary cholangitis, the odds ratio (OR) for CRC was reduced by 35% (OR, 0.65; 95% confidence interval, 0.45-0.93), according to Dr. Ashwin Ananthakrishnan, a gastroenterologist at Massachusetts General Hospital, Boston.
Many professional societies, including the ACG, recommend colonoscopy surveillance in IBD patients within 8-10 years of diagnosis, and subsequently at 2- to 3-year intervals, but these recommendations were created without direct evidence of benefit. The aim of the study was to evaluate the effect of colonoscopy surveillance on CRC incidence.
The 6,823 patients who formed the cohort were gathered through electronic medical records from multiple participating institutions. The incidence of CRC was compared in the 2,764 patients who underwent colonoscopy within 3 years prior to a diagnosis of CRC or the end of the follow-up period and the 4,059 without colonoscopy in this period.
Of the 154 cases of CRC observed during follow-up, 43 occurred in the group that had undergone colonoscopy and 111 occurred in the group that had not, producing an incidence of 1.6% and 2.7%, respectively. Although there were potentially relevant differences between the two patient groups – the colonoscopy group had a lower incidence of ulcerative colitis (49% vs. 54%) and a younger age (47 vs. 49 years) – the advantage persisted after adjustment.
In addition, death due to CRC was lower in the group that underwent colonoscopy in the past 3 years (14% vs. 34%; P = .012), producing an odds ratio of 0.34 (95% CI, 0.12-0.95) for this endpoint.
Dr. Ananthakrishnan acknowledged that the limitations of the study included recruitment from a largely tertiary-center population and lack of information on the extent of IBD or the stage of cancer, but he called the overall findings “robust.” He concluded that these data may provide the best support yet for the current guidelines.
“I do not think this suggests we should be doing anything any differently,” Dr. Ananthakrishnan said in an interview, “but it does reinforce the value of the guidelines for those who may not be applying them now.”
Asked for his perspective on these data, Dr. Stephen Hanauer, professor of gastroenterology and hepatology, Northwestern University, Chicago, was more circumspect.
“There is evidence that the incidence of CRC is going down in patients with IBD, and it may be due to better surveillance, but this study has some limitations,” Dr. Hanauer commented. “It could have been just that those who received colonoscopy had less advanced IBD and a lower cancer risk.”
The definitive answer is dependent on a prospective and randomized trial, but there is limited demand for such a study when the increased risk of CRC in patients with IBD is well accepted, making surveillance attractive. Dr. Hanauer did not dispute the logic behind current screening recommendations.
Dr. Ananthakrishnan reported having no relevant financial relationships to disclose.
AGA Resources
The AGA Medical Position Statement on the Diagnosis and Management of Colorectal Neoplasia in Inflammatory Bowel Disease is available online here.
PHILADELPHIA - The risk of both colorectal cancer and CRC-related mortality can be reduced in patients with inflammatory bowel disease by adhering to guideline-directed colonoscopy surveillance recommendations, according to a large retrospective cohort study presented at the annual meeting of the American College of Gastroenterology.
After adjustment for age, sex, duration of disease, type of IBD, and coexisting primary cholangitis, the odds ratio (OR) for CRC was reduced by 35% (OR, 0.65; 95% confidence interval, 0.45-0.93), according to Dr. Ashwin Ananthakrishnan, a gastroenterologist at Massachusetts General Hospital, Boston.
Many professional societies, including the ACG, recommend colonoscopy surveillance in IBD patients within 8-10 years of diagnosis, and subsequently at 2- to 3-year intervals, but these recommendations were created without direct evidence of benefit. The aim of the study was to evaluate the effect of colonoscopy surveillance on CRC incidence.
The 6,823 patients who formed the cohort were gathered through electronic medical records from multiple participating institutions. The incidence of CRC was compared in the 2,764 patients who underwent colonoscopy within 3 years prior to a diagnosis of CRC or the end of the follow-up period and the 4,059 without colonoscopy in this period.
Of the 154 cases of CRC observed during follow-up, 43 occurred in the group that had undergone colonoscopy and 111 occurred in the group that had not, producing an incidence of 1.6% and 2.7%, respectively. Although there were potentially relevant differences between the two patient groups – the colonoscopy group had a lower incidence of ulcerative colitis (49% vs. 54%) and a younger age (47 vs. 49 years) – the advantage persisted after adjustment.
In addition, death due to CRC was lower in the group that underwent colonoscopy in the past 3 years (14% vs. 34%; P = .012), producing an odds ratio of 0.34 (95% CI, 0.12-0.95) for this endpoint.
Dr. Ananthakrishnan acknowledged that the limitations of the study included recruitment from a largely tertiary-center population and lack of information on the extent of IBD or the stage of cancer, but he called the overall findings “robust.” He concluded that these data may provide the best support yet for the current guidelines.
“I do not think this suggests we should be doing anything any differently,” Dr. Ananthakrishnan said in an interview, “but it does reinforce the value of the guidelines for those who may not be applying them now.”
Asked for his perspective on these data, Dr. Stephen Hanauer, professor of gastroenterology and hepatology, Northwestern University, Chicago, was more circumspect.
“There is evidence that the incidence of CRC is going down in patients with IBD, and it may be due to better surveillance, but this study has some limitations,” Dr. Hanauer commented. “It could have been just that those who received colonoscopy had less advanced IBD and a lower cancer risk.”
The definitive answer is dependent on a prospective and randomized trial, but there is limited demand for such a study when the increased risk of CRC in patients with IBD is well accepted, making surveillance attractive. Dr. Hanauer did not dispute the logic behind current screening recommendations.
Dr. Ananthakrishnan reported having no relevant financial relationships to disclose.
AGA Resources
The AGA Medical Position Statement on the Diagnosis and Management of Colorectal Neoplasia in Inflammatory Bowel Disease is available online here.
PHILADELPHIA - The risk of both colorectal cancer and CRC-related mortality can be reduced in patients with inflammatory bowel disease by adhering to guideline-directed colonoscopy surveillance recommendations, according to a large retrospective cohort study presented at the annual meeting of the American College of Gastroenterology.
After adjustment for age, sex, duration of disease, type of IBD, and coexisting primary cholangitis, the odds ratio (OR) for CRC was reduced by 35% (OR, 0.65; 95% confidence interval, 0.45-0.93), according to Dr. Ashwin Ananthakrishnan, a gastroenterologist at Massachusetts General Hospital, Boston.
Many professional societies, including the ACG, recommend colonoscopy surveillance in IBD patients within 8-10 years of diagnosis, and subsequently at 2- to 3-year intervals, but these recommendations were created without direct evidence of benefit. The aim of the study was to evaluate the effect of colonoscopy surveillance on CRC incidence.
The 6,823 patients who formed the cohort were gathered through electronic medical records from multiple participating institutions. The incidence of CRC was compared in the 2,764 patients who underwent colonoscopy within 3 years prior to a diagnosis of CRC or the end of the follow-up period and the 4,059 without colonoscopy in this period.
Of the 154 cases of CRC observed during follow-up, 43 occurred in the group that had undergone colonoscopy and 111 occurred in the group that had not, producing an incidence of 1.6% and 2.7%, respectively. Although there were potentially relevant differences between the two patient groups – the colonoscopy group had a lower incidence of ulcerative colitis (49% vs. 54%) and a younger age (47 vs. 49 years) – the advantage persisted after adjustment.
In addition, death due to CRC was lower in the group that underwent colonoscopy in the past 3 years (14% vs. 34%; P = .012), producing an odds ratio of 0.34 (95% CI, 0.12-0.95) for this endpoint.
Dr. Ananthakrishnan acknowledged that the limitations of the study included recruitment from a largely tertiary-center population and lack of information on the extent of IBD or the stage of cancer, but he called the overall findings “robust.” He concluded that these data may provide the best support yet for the current guidelines.
“I do not think this suggests we should be doing anything any differently,” Dr. Ananthakrishnan said in an interview, “but it does reinforce the value of the guidelines for those who may not be applying them now.”
Asked for his perspective on these data, Dr. Stephen Hanauer, professor of gastroenterology and hepatology, Northwestern University, Chicago, was more circumspect.
“There is evidence that the incidence of CRC is going down in patients with IBD, and it may be due to better surveillance, but this study has some limitations,” Dr. Hanauer commented. “It could have been just that those who received colonoscopy had less advanced IBD and a lower cancer risk.”
The definitive answer is dependent on a prospective and randomized trial, but there is limited demand for such a study when the increased risk of CRC in patients with IBD is well accepted, making surveillance attractive. Dr. Hanauer did not dispute the logic behind current screening recommendations.
Dr. Ananthakrishnan reported having no relevant financial relationships to disclose.
AGA Resources
The AGA Medical Position Statement on the Diagnosis and Management of Colorectal Neoplasia in Inflammatory Bowel Disease is available online here.
AT ACG 2014
Key clinical point: New evidence that surveillance colonoscopy can reduce the risk of colorectal cancer in patients with inflammatory bowel disease validates previously unsupported guidelines.
Major finding: Colonoscopy screening in IBD patients within the previous 3 years reduces the odds ratio of developing cancer by 35% and the risk of death if cancer develops by more than 50%.
Data source: Retrospective cohort analysis of electronic medical records in 6,823 IBD patients.
Disclosures: Dr. Ananthakrishnan reported having no relevant financial relationships to disclose.
Cancer risk from IBD mitigated with colonoscopy surveillance
PHILADELPHIA - The risk of both colorectal cancer and CRC-related mortality can be reduced in patients with inflammatory bowel disease by adhering to guideline-directed colonoscopy surveillance recommendations, according to a large retrospective cohort study presented at the annual meeting of the American College of Gastroenterology.
After adjustment for age, sex, duration of disease, type of IBD, and coexisting primary cholangitis, the odds ratio (OR) for CRC was reduced by 35% (OR, 0.65; 95% confidence interval, 0.45-0.93), according to Dr. Ashwin Ananthakrishnan, a gastroenterologist at Massachusetts General Hospital, Boston.
Many professional societies, including the ACG, recommend colonoscopy surveillance in IBD patients within 8-10 years of diagnosis, and subsequently at 2- to 3-year intervals, but these recommendations were created without direct evidence of benefit. The aim of the study was to evaluate the effect of colonoscopy surveillance on CRC incidence.
The 6,823 patients who formed the cohort were gathered through electronic medical records from multiple participating institutions. The incidence of CRC was compared in the 2,764 patients who underwent colonoscopy within 3 years prior to a diagnosis of CRC or the end of the follow-up period and the 4,059 without colonoscopy in this period.
Of the 154 cases of CRC observed during follow-up, 43 occurred in the group that had undergone colonoscopy and 111 occurred in the group that had not, producing an incidence of 1.6% and 2.7%, respectively. Although there were potentially relevant differences between the two patient groups – the colonoscopy group had a lower incidence of ulcerative colitis (49% vs. 54%) and a younger age (47 vs. 49 years) – the advantage persisted after adjustment.
In addition, death due to CRC was lower in the group that underwent colonoscopy in the past 3 years (14% vs. 34%; P = .012), producing an odds ratio of 0.34 (95% CI, 0.12-0.95) for this endpoint.
Dr. Ananthakrishnan acknowledged that the limitations of the study included recruitment from a largely tertiary-center population and lack of information on the extent of IBD or the stage of cancer, but he called the overall findings “robust.” He concluded that these data may provide the best support yet for the current guidelines.
“I do not think this suggests we should be doing anything any differently,” Dr. Ananthakrishnan said in an interview, “but it does reinforce the value of the guidelines for those who may not be applying them now.”
Asked for his perspective on these data, Dr. Stephen Hanauer, professor of gastroenterology and hepatology, Northwestern University, Chicago, was more circumspect.
“There is evidence that the incidence of CRC is going down in patients with IBD, and it may be due to better surveillance, but this study has some limitations,” Dr. Hanauer commented. “It could have been just that those who received colonoscopy had less advanced IBD and a lower cancer risk.”
The definitive answer is dependent on a prospective and randomized trial, but there is limited demand for such a study when the increased risk of CRC in patients with IBD is well accepted, making surveillance attractive. Dr. Hanauer did not dispute the logic behind current screening recommendations.
Dr. Ananthakrishnan reported having no relevant financial relationships to disclose.
PHILADELPHIA - The risk of both colorectal cancer and CRC-related mortality can be reduced in patients with inflammatory bowel disease by adhering to guideline-directed colonoscopy surveillance recommendations, according to a large retrospective cohort study presented at the annual meeting of the American College of Gastroenterology.
After adjustment for age, sex, duration of disease, type of IBD, and coexisting primary cholangitis, the odds ratio (OR) for CRC was reduced by 35% (OR, 0.65; 95% confidence interval, 0.45-0.93), according to Dr. Ashwin Ananthakrishnan, a gastroenterologist at Massachusetts General Hospital, Boston.
Many professional societies, including the ACG, recommend colonoscopy surveillance in IBD patients within 8-10 years of diagnosis, and subsequently at 2- to 3-year intervals, but these recommendations were created without direct evidence of benefit. The aim of the study was to evaluate the effect of colonoscopy surveillance on CRC incidence.
The 6,823 patients who formed the cohort were gathered through electronic medical records from multiple participating institutions. The incidence of CRC was compared in the 2,764 patients who underwent colonoscopy within 3 years prior to a diagnosis of CRC or the end of the follow-up period and the 4,059 without colonoscopy in this period.
Of the 154 cases of CRC observed during follow-up, 43 occurred in the group that had undergone colonoscopy and 111 occurred in the group that had not, producing an incidence of 1.6% and 2.7%, respectively. Although there were potentially relevant differences between the two patient groups – the colonoscopy group had a lower incidence of ulcerative colitis (49% vs. 54%) and a younger age (47 vs. 49 years) – the advantage persisted after adjustment.
In addition, death due to CRC was lower in the group that underwent colonoscopy in the past 3 years (14% vs. 34%; P = .012), producing an odds ratio of 0.34 (95% CI, 0.12-0.95) for this endpoint.
Dr. Ananthakrishnan acknowledged that the limitations of the study included recruitment from a largely tertiary-center population and lack of information on the extent of IBD or the stage of cancer, but he called the overall findings “robust.” He concluded that these data may provide the best support yet for the current guidelines.
“I do not think this suggests we should be doing anything any differently,” Dr. Ananthakrishnan said in an interview, “but it does reinforce the value of the guidelines for those who may not be applying them now.”
Asked for his perspective on these data, Dr. Stephen Hanauer, professor of gastroenterology and hepatology, Northwestern University, Chicago, was more circumspect.
“There is evidence that the incidence of CRC is going down in patients with IBD, and it may be due to better surveillance, but this study has some limitations,” Dr. Hanauer commented. “It could have been just that those who received colonoscopy had less advanced IBD and a lower cancer risk.”
The definitive answer is dependent on a prospective and randomized trial, but there is limited demand for such a study when the increased risk of CRC in patients with IBD is well accepted, making surveillance attractive. Dr. Hanauer did not dispute the logic behind current screening recommendations.
Dr. Ananthakrishnan reported having no relevant financial relationships to disclose.
PHILADELPHIA - The risk of both colorectal cancer and CRC-related mortality can be reduced in patients with inflammatory bowel disease by adhering to guideline-directed colonoscopy surveillance recommendations, according to a large retrospective cohort study presented at the annual meeting of the American College of Gastroenterology.
After adjustment for age, sex, duration of disease, type of IBD, and coexisting primary cholangitis, the odds ratio (OR) for CRC was reduced by 35% (OR, 0.65; 95% confidence interval, 0.45-0.93), according to Dr. Ashwin Ananthakrishnan, a gastroenterologist at Massachusetts General Hospital, Boston.
Many professional societies, including the ACG, recommend colonoscopy surveillance in IBD patients within 8-10 years of diagnosis, and subsequently at 2- to 3-year intervals, but these recommendations were created without direct evidence of benefit. The aim of the study was to evaluate the effect of colonoscopy surveillance on CRC incidence.
The 6,823 patients who formed the cohort were gathered through electronic medical records from multiple participating institutions. The incidence of CRC was compared in the 2,764 patients who underwent colonoscopy within 3 years prior to a diagnosis of CRC or the end of the follow-up period and the 4,059 without colonoscopy in this period.
Of the 154 cases of CRC observed during follow-up, 43 occurred in the group that had undergone colonoscopy and 111 occurred in the group that had not, producing an incidence of 1.6% and 2.7%, respectively. Although there were potentially relevant differences between the two patient groups – the colonoscopy group had a lower incidence of ulcerative colitis (49% vs. 54%) and a younger age (47 vs. 49 years) – the advantage persisted after adjustment.
In addition, death due to CRC was lower in the group that underwent colonoscopy in the past 3 years (14% vs. 34%; P = .012), producing an odds ratio of 0.34 (95% CI, 0.12-0.95) for this endpoint.
Dr. Ananthakrishnan acknowledged that the limitations of the study included recruitment from a largely tertiary-center population and lack of information on the extent of IBD or the stage of cancer, but he called the overall findings “robust.” He concluded that these data may provide the best support yet for the current guidelines.
“I do not think this suggests we should be doing anything any differently,” Dr. Ananthakrishnan said in an interview, “but it does reinforce the value of the guidelines for those who may not be applying them now.”
Asked for his perspective on these data, Dr. Stephen Hanauer, professor of gastroenterology and hepatology, Northwestern University, Chicago, was more circumspect.
“There is evidence that the incidence of CRC is going down in patients with IBD, and it may be due to better surveillance, but this study has some limitations,” Dr. Hanauer commented. “It could have been just that those who received colonoscopy had less advanced IBD and a lower cancer risk.”
The definitive answer is dependent on a prospective and randomized trial, but there is limited demand for such a study when the increased risk of CRC in patients with IBD is well accepted, making surveillance attractive. Dr. Hanauer did not dispute the logic behind current screening recommendations.
Dr. Ananthakrishnan reported having no relevant financial relationships to disclose.
AT ACG 2014
Key clinical point: New evidence that surveillance colonoscopy can reduce the risk of colorectal cancer in patients with inflammatory bowel disease validates previously unsupported guidelines.
Major finding: Colonoscopy screening in IBD patients within the previous 3 years reduces the odds ratio of developing cancer by 35% and the risk of death if cancer develops by more than 50%.
Data source: Retrospective cohort analysis of electronic medical records in 6,823 IBD patients.
Disclosures: Dr. Ananthakrishnan reported having no relevant financial relationships to disclose.