User login
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
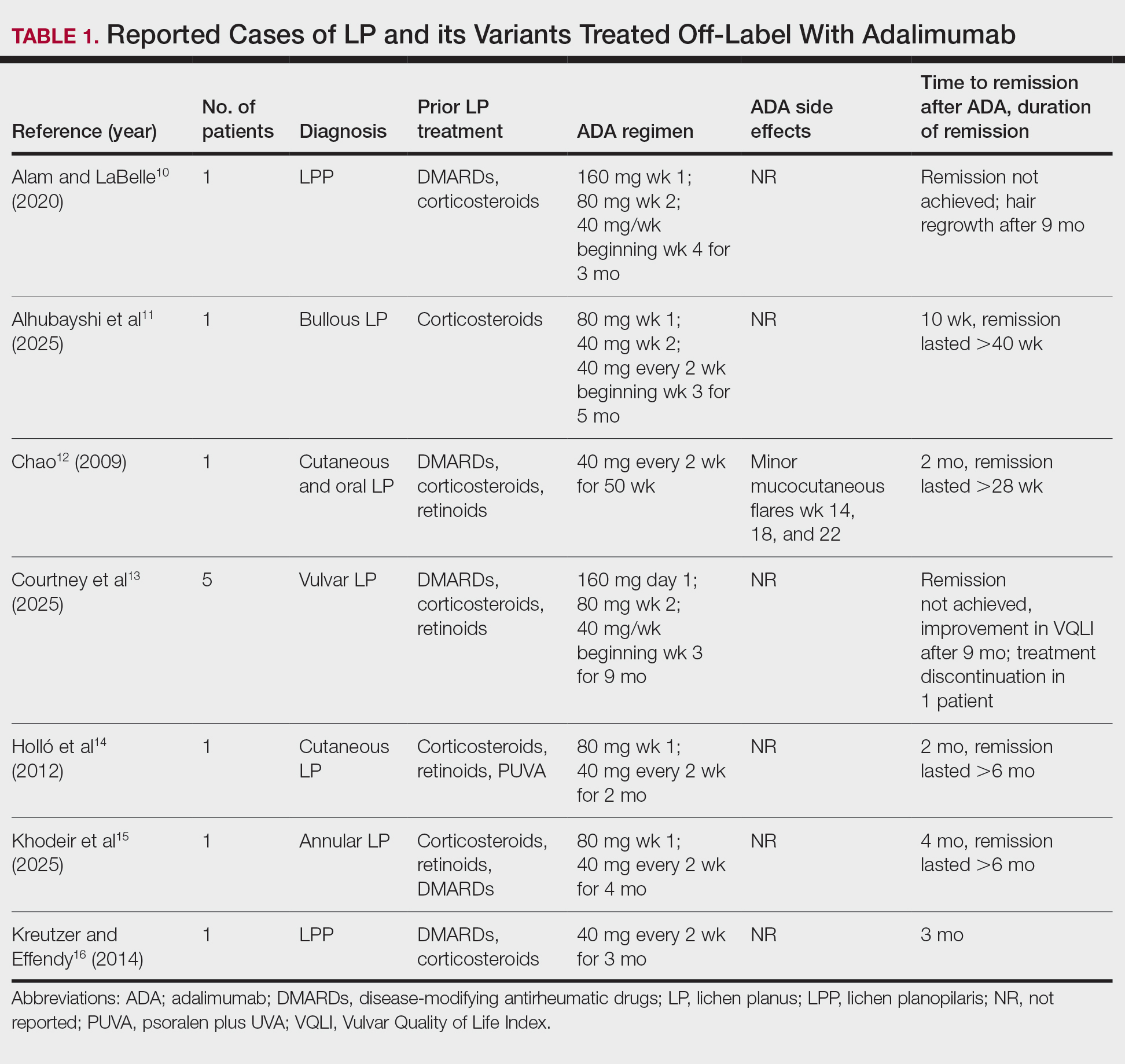
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)
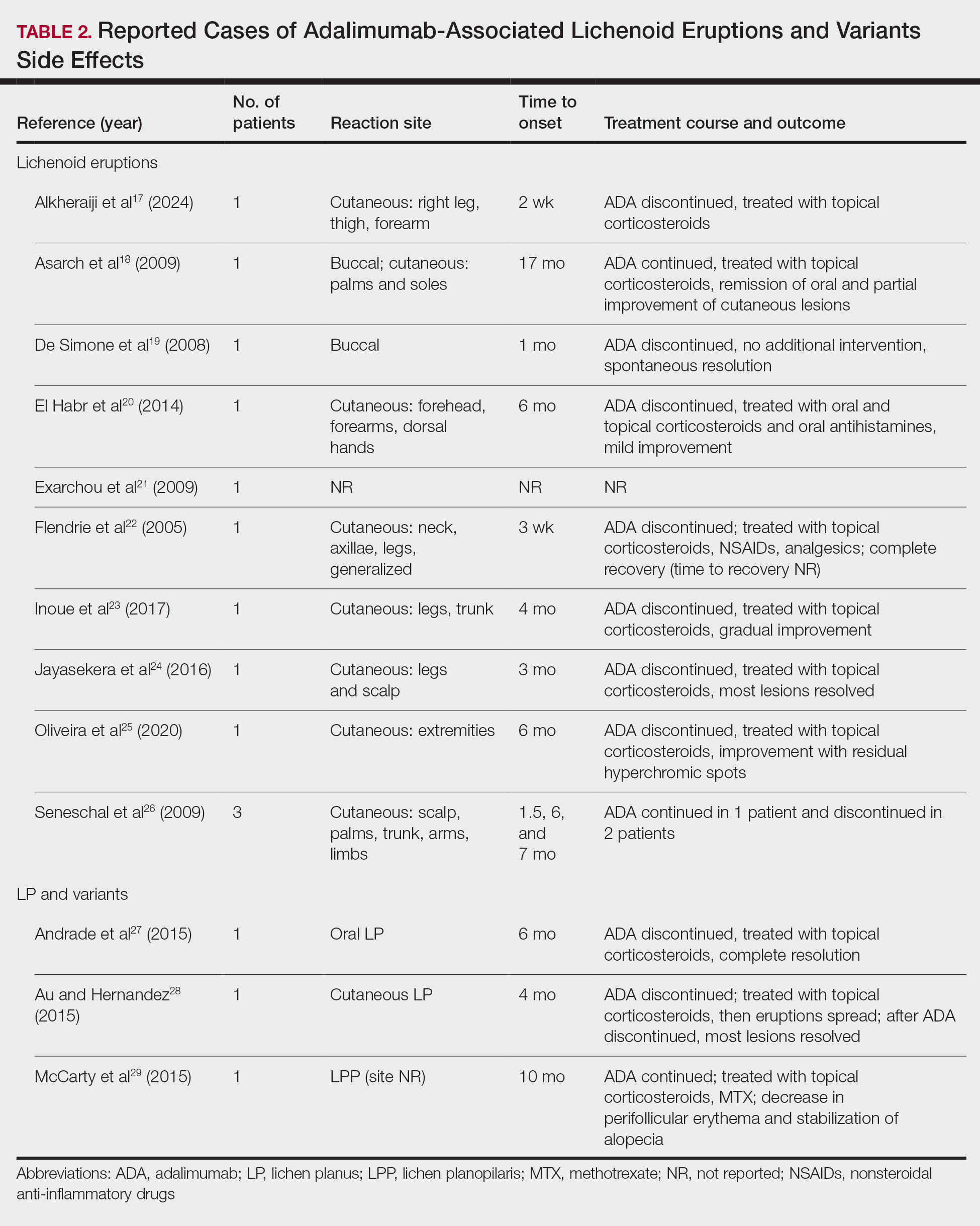
Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22
In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)

Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22

In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)

Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22

In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Practice Points
- Adalimumab can be beneficial when used off label for treatment of lichen planus in patients who do not respond to conventional therapies, including corticosteroids and immunosuppressants.
- Clinicians should be aware that adalimumab could potentially lead to paradoxical lichenoid eruptions and should monitor patients closely during treatment.
Eruptive Erythematous Papules on the Forearms
Eruptive Erythematous Papules on the Forearms
THE DIAGNOSIS: Acral Eruptive Syringoma
Syringomas are small, benign, often asymptomatic eccrine tumors that originate in the intraepidermal portion of eccrine sweat ducts.1 Clinically, they present as multiple symmetric white-to-yellow or discrete flesh-colored papules measuring 1 to 3 mm in diameter, often located on the face (most commonly on the eyelids), with a greater prevalence in middle-aged women. Occasionally, they manifest in other locations such as the cheeks, chest, axillae, abdomen, and groin.2
In 1987, Friedman and Butler3 developed a classification system categorizing syringomas into 4 clinical subtypes: familial syringoma, localized syringoma, Down syndrome–related syringoma, and generalized syringoma. The fourth subtype includes the variant of eruptive syringoma,3 a rare clinical manifestation that often develops before or during puberty with several flesh-colored or lightly pigmented papules on the neck, anterior chest, upper abdomen, axillae, periumbilical region, and/or genital region.1,4,5 The etiology of eruptive syringomas is unclear, although it has been linked to abnormal proliferation of sweat glands due to an underlying local inflammatory process.6
Acral distribution of syringomas is a rare variant that can manifest as part of generalized eruptive syringoma with consequent involvement of the arms and other areas.5,7 There are limited case reports on eruptive syringomas with predominant acral distribution.8 Compared to classic syringomas, the acral variant is associated with an older age of onset as well as a similar prevalence between men and women.9 Acral eruptive syringoma (AES) usually is isolated to the distal arms and legs. The most commonly affected region is the anterior surface of the forearms, although involvement of the dorsal hands, wrists, and feet also has been reported.10-16
The first known case of AES, which was reported in 1977, described eruptive syringomas on the dorsal hands of a healthy 31-year-old man.17 Several cases have been reported since then, mostly in patients aged 30 to 60 years, with predominant involvement of the dorsal hands and forearms.18-24 A review of Embase as well as PubMed articles indexed for MEDLINE using the search terms syringoma OR eccrine ductal tumor and eruptive OR acral OR arms OR forearms OR extremities identified 19 reported cases of AES between 1977 and 2023. For the reported AES cases, the mean (SD) age at diagnosis was 45.1 years (15.96 years), with patient ages ranging from 19 to 76 years. Notably, most cases occurred in individuals aged between 30 and 60 years, which deviates from the typical age of onset of localized syringomas, commonly seen during puberty or early adulthood.
Currently, AES is categorized within the clinical presentation of eruptive syringoma. Nevertheless, some authors have proposed classifying it as a distinct fifth clinical group due to specific features that distinguish it from generalized eruptive syringoma.9 This reclassification has considerable implications for the differential diagnosis, particularly because exclusive acral involvement poses a substantial diagnostic challenge and often requires histologic confirmation.
As shown in the Figure, histopathologic examination revealed tubular structures in the upper dermis with characteristic comma-shaped extensions. Some of these structures were lined with cuboidal cells and contained eosinophilic material within the lumen. There was no involvement of the epidermis or deeper dermis. The histologic features were consistent with syringoma, which is distinguished by its predominant involvement of the upper dermis and the presence of enlarged, dilated eccrine ducts, as observed in our case.
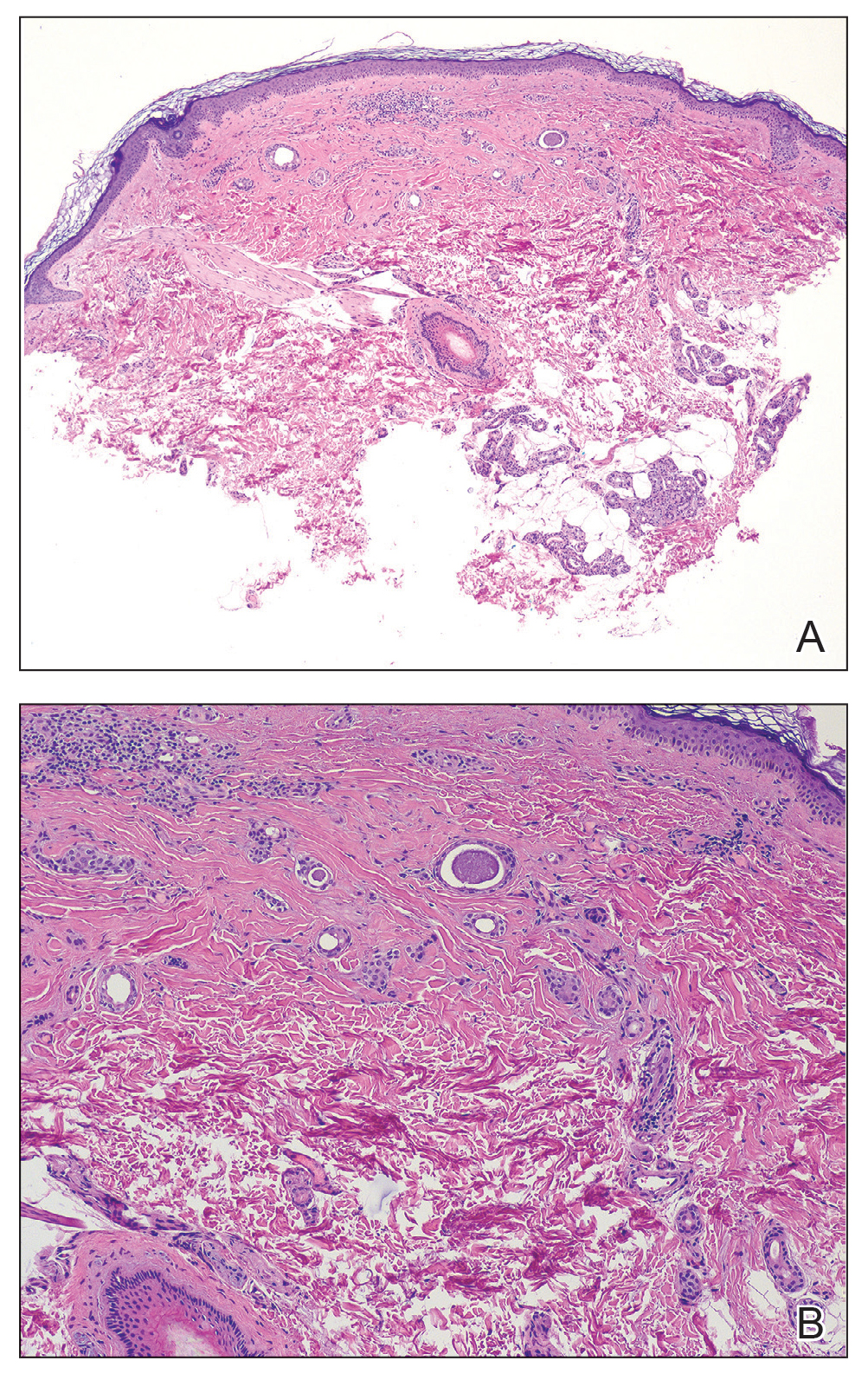
Treatment of syringomas often is challenging due to the high rate of recurrence and the risk for postinflammatory hyperpigmentation. Since the condition is benign, treatment typically is pursued for aesthetic reasons. Various therapeutic approaches have been reported, each with diverse response rates. The most common method involves surgical intervention, either with electrodesiccation or CO2 laser—both of which have shown satisfactory resolution of lesions without recurrence at 1-year follow-up, with no major scarring reported.25,26 Alternatively, topical management with retinoids daily over a 4-month period leads to flattening of the tumors with no further appearance of new lesions.27 Despite the availability of numerous management options, establishing a first-line treatment remains controversial due to the high risk for recurrence and the variability in the number and location of lesions among individual patients. In our case, given the benign nature of syringomas, the asymptomatic nature of the lesions, the involvement of noncritical aesthetic areas, and the limited response to noninvasive therapeutic options, the patient was informed of the diagnosis, and no further pharmacologic or surgical intervention was pursued.
- Williams K, Shinkai K. Evaluation and management of the patient with multiple syringomas: a systematic review of the literature. J Am Acad Dermatol. 2016;74:1234-1240.E9. doi:10.1016 /j.jaad.2015.12.006
- Resende C, Araújo C, Santos R, et al. Late-onset of eruptive syringomas: a diagnostic challenge. An Bras Dermatol. 2015;90(3 suppl 1):239-241. doi:10.1590/abd1806-4841.20153899
- Friedman SJ, Butler DF. Syringoma presenting as milia. J Am Acad Dermatol. 1987;16:310-314.
- Avhad G, Ghuge P, Jerajani HR. Generalized eruptive syringoma. Indian J Dermatol. 2015;60:214. doi:10.4103/0019-5154.152586
- Ning WV, Bashey S, Cole C, et al. Multiple eruptive syringomas on the penis. Cutis. 2019;103:E15-E16.
- Cohen PR, Tschen JA, Rapini RP. Penile syringoma: reports and review of patients with syringoma located on the penis. J Clin Aesthet Dermatol. 2013;6:38-42.
- Jamalipour M, Heidarpour M, Rajabi P. Generalized eruptive syringomas. Indian J Dermatol. 2009;54:65-67. doi:10.4103/0019-5154.48992
- Mohaghegh F, Amiri A, Fatemi Naeini F, et al. Acral eruptive syringoma: an unusual presentation with misdiagnosis. Case Rep Dermatol Med. 2020;2020:5416285. doi:10.1155/2020/5416285
- Valdivielso-Ramos M, de la Cueva P, Gimeno M, et al. Acral syringomas. Actas Dermosifiliogr. 2010;101:458-460.
- Patel K, Lundgren AD, Ahmed AM, et al. Disseminated syringomas of the upper extremities in a young woman. Cureus. 2018;10:E3619. doi:10.7759/cureus.3619
- Balci DD, Atik E, Altintas S. Coexistence of acral syringomas and multiple trichoepitheliomas on the face. J Cutan Med Surg. 2009;13:169-171. doi:10.2310/7750.2008.08011
- Martín-García RF, Muñoz CM. Acral syringomas presenting as a photosensitive papular eruption. Cutis. 2006;77:33-36.
- Varas-Meis E, Prada-García C, Samaniego-González E, et al. Acral syringomas associated with hematological neoplasm. Indian J Dermatol Venereol Leprol. 2017;83:136. doi:10.4103/0378-6323.192961
- Berbis P, Fabre JF, Jancovici E, et al. Late-onset syringomas of the upper extremities associated with a carcinoid tumor. Arch Dermatol. 1989;125:848-849.
- Metze D, Jurecka W, Gebhart W. Disseminated syringomas of the upper extremities. case history and immunohistochemical and ultrastructural study. Dermatologica. 1990;180:228-235. doi:10.1159/000248036
- Gómez-de Castro C, Vivanco Allende B, García-García B. Multiple acral syringomas. siringomas acrales múltiples. Actas Dermosifiliogr (Engl Ed). 2018;109:834-836. doi:10.1016/j.ad.2017.10.014
- Hughes PS, Apisarnthanarax P. Acral syringoma. Arch Dermatol. 1977;113:1435-1436.
- Asai Y, Ishii M, Hamada T. Acral syringoma: electron microscopic studies on its origin. Acta Derm Venereol. 1982;62:64-68.
- van den Broek H, Lundquist CD. Syringomas of the upper extremities with onset in the sixth decade. J Am Acad Dermatol. 1982,6:534-536. doi:10.1016/S0190-9622(82)80368-X
- Garcia C, Krunic AL, Grichnik J, et al. Multiple acral syringomata with uniform involvement of the hands and feet. Cutis. 1997;59:213-214, 216.
- Patrizi A, Neri I, Marzaduri S, et al. Syringoma: a review of twenty-nine cases. Acta Derm Venereol. 1998;78:460-462.
- Iglesias Sancho M, Serra Llobet J, Salleras Redonnet M, et al. Siringomas disem- inados de inicio acral, aparecidos en la octava década. Actas Dermosifiliofr. 1999;90:253-257.
- Muniesa C, Fortuño Y, Moreno A, et al. Papules on the dorsum of the fingers. Actas Dermosifiliogr. 2008;99:812-813. doi:10.1016 /S1578-2190(08)70371-8
- Koh MJ. Multiple acral syringomas involving the hands. Clin Exp Dermatol. 2009;34:E438. doi:10.1111/j.1365-2230.2009.03462.x
- Karam P, Benedetto AV. Syringomas: new approach to an old technique. Int J Dermatol. 1996;35:219-220. doi:10.1111/j.1365-4362 .1996.tb01647.x
- Wang JI, Roenigk HH. Treatment of multiple facial syringomas with the carbon dioxide (CO2) laser. Dermatol Surg. 1999;25:136-139. doi:10.1046/j.1524-4725.1999.08111.x
- Gómez MI, Pérez B, Azaña JM, et al. Eruptive syringoma: treatment with topical tretinoin. Dermatology. 2009;189:105-106. doi:10.1159/000246803
THE DIAGNOSIS: Acral Eruptive Syringoma
Syringomas are small, benign, often asymptomatic eccrine tumors that originate in the intraepidermal portion of eccrine sweat ducts.1 Clinically, they present as multiple symmetric white-to-yellow or discrete flesh-colored papules measuring 1 to 3 mm in diameter, often located on the face (most commonly on the eyelids), with a greater prevalence in middle-aged women. Occasionally, they manifest in other locations such as the cheeks, chest, axillae, abdomen, and groin.2
In 1987, Friedman and Butler3 developed a classification system categorizing syringomas into 4 clinical subtypes: familial syringoma, localized syringoma, Down syndrome–related syringoma, and generalized syringoma. The fourth subtype includes the variant of eruptive syringoma,3 a rare clinical manifestation that often develops before or during puberty with several flesh-colored or lightly pigmented papules on the neck, anterior chest, upper abdomen, axillae, periumbilical region, and/or genital region.1,4,5 The etiology of eruptive syringomas is unclear, although it has been linked to abnormal proliferation of sweat glands due to an underlying local inflammatory process.6
Acral distribution of syringomas is a rare variant that can manifest as part of generalized eruptive syringoma with consequent involvement of the arms and other areas.5,7 There are limited case reports on eruptive syringomas with predominant acral distribution.8 Compared to classic syringomas, the acral variant is associated with an older age of onset as well as a similar prevalence between men and women.9 Acral eruptive syringoma (AES) usually is isolated to the distal arms and legs. The most commonly affected region is the anterior surface of the forearms, although involvement of the dorsal hands, wrists, and feet also has been reported.10-16
The first known case of AES, which was reported in 1977, described eruptive syringomas on the dorsal hands of a healthy 31-year-old man.17 Several cases have been reported since then, mostly in patients aged 30 to 60 years, with predominant involvement of the dorsal hands and forearms.18-24 A review of Embase as well as PubMed articles indexed for MEDLINE using the search terms syringoma OR eccrine ductal tumor and eruptive OR acral OR arms OR forearms OR extremities identified 19 reported cases of AES between 1977 and 2023. For the reported AES cases, the mean (SD) age at diagnosis was 45.1 years (15.96 years), with patient ages ranging from 19 to 76 years. Notably, most cases occurred in individuals aged between 30 and 60 years, which deviates from the typical age of onset of localized syringomas, commonly seen during puberty or early adulthood.
Currently, AES is categorized within the clinical presentation of eruptive syringoma. Nevertheless, some authors have proposed classifying it as a distinct fifth clinical group due to specific features that distinguish it from generalized eruptive syringoma.9 This reclassification has considerable implications for the differential diagnosis, particularly because exclusive acral involvement poses a substantial diagnostic challenge and often requires histologic confirmation.
As shown in the Figure, histopathologic examination revealed tubular structures in the upper dermis with characteristic comma-shaped extensions. Some of these structures were lined with cuboidal cells and contained eosinophilic material within the lumen. There was no involvement of the epidermis or deeper dermis. The histologic features were consistent with syringoma, which is distinguished by its predominant involvement of the upper dermis and the presence of enlarged, dilated eccrine ducts, as observed in our case.

Treatment of syringomas often is challenging due to the high rate of recurrence and the risk for postinflammatory hyperpigmentation. Since the condition is benign, treatment typically is pursued for aesthetic reasons. Various therapeutic approaches have been reported, each with diverse response rates. The most common method involves surgical intervention, either with electrodesiccation or CO2 laser—both of which have shown satisfactory resolution of lesions without recurrence at 1-year follow-up, with no major scarring reported.25,26 Alternatively, topical management with retinoids daily over a 4-month period leads to flattening of the tumors with no further appearance of new lesions.27 Despite the availability of numerous management options, establishing a first-line treatment remains controversial due to the high risk for recurrence and the variability in the number and location of lesions among individual patients. In our case, given the benign nature of syringomas, the asymptomatic nature of the lesions, the involvement of noncritical aesthetic areas, and the limited response to noninvasive therapeutic options, the patient was informed of the diagnosis, and no further pharmacologic or surgical intervention was pursued.
THE DIAGNOSIS: Acral Eruptive Syringoma
Syringomas are small, benign, often asymptomatic eccrine tumors that originate in the intraepidermal portion of eccrine sweat ducts.1 Clinically, they present as multiple symmetric white-to-yellow or discrete flesh-colored papules measuring 1 to 3 mm in diameter, often located on the face (most commonly on the eyelids), with a greater prevalence in middle-aged women. Occasionally, they manifest in other locations such as the cheeks, chest, axillae, abdomen, and groin.2
In 1987, Friedman and Butler3 developed a classification system categorizing syringomas into 4 clinical subtypes: familial syringoma, localized syringoma, Down syndrome–related syringoma, and generalized syringoma. The fourth subtype includes the variant of eruptive syringoma,3 a rare clinical manifestation that often develops before or during puberty with several flesh-colored or lightly pigmented papules on the neck, anterior chest, upper abdomen, axillae, periumbilical region, and/or genital region.1,4,5 The etiology of eruptive syringomas is unclear, although it has been linked to abnormal proliferation of sweat glands due to an underlying local inflammatory process.6
Acral distribution of syringomas is a rare variant that can manifest as part of generalized eruptive syringoma with consequent involvement of the arms and other areas.5,7 There are limited case reports on eruptive syringomas with predominant acral distribution.8 Compared to classic syringomas, the acral variant is associated with an older age of onset as well as a similar prevalence between men and women.9 Acral eruptive syringoma (AES) usually is isolated to the distal arms and legs. The most commonly affected region is the anterior surface of the forearms, although involvement of the dorsal hands, wrists, and feet also has been reported.10-16
The first known case of AES, which was reported in 1977, described eruptive syringomas on the dorsal hands of a healthy 31-year-old man.17 Several cases have been reported since then, mostly in patients aged 30 to 60 years, with predominant involvement of the dorsal hands and forearms.18-24 A review of Embase as well as PubMed articles indexed for MEDLINE using the search terms syringoma OR eccrine ductal tumor and eruptive OR acral OR arms OR forearms OR extremities identified 19 reported cases of AES between 1977 and 2023. For the reported AES cases, the mean (SD) age at diagnosis was 45.1 years (15.96 years), with patient ages ranging from 19 to 76 years. Notably, most cases occurred in individuals aged between 30 and 60 years, which deviates from the typical age of onset of localized syringomas, commonly seen during puberty or early adulthood.
Currently, AES is categorized within the clinical presentation of eruptive syringoma. Nevertheless, some authors have proposed classifying it as a distinct fifth clinical group due to specific features that distinguish it from generalized eruptive syringoma.9 This reclassification has considerable implications for the differential diagnosis, particularly because exclusive acral involvement poses a substantial diagnostic challenge and often requires histologic confirmation.
As shown in the Figure, histopathologic examination revealed tubular structures in the upper dermis with characteristic comma-shaped extensions. Some of these structures were lined with cuboidal cells and contained eosinophilic material within the lumen. There was no involvement of the epidermis or deeper dermis. The histologic features were consistent with syringoma, which is distinguished by its predominant involvement of the upper dermis and the presence of enlarged, dilated eccrine ducts, as observed in our case.

Treatment of syringomas often is challenging due to the high rate of recurrence and the risk for postinflammatory hyperpigmentation. Since the condition is benign, treatment typically is pursued for aesthetic reasons. Various therapeutic approaches have been reported, each with diverse response rates. The most common method involves surgical intervention, either with electrodesiccation or CO2 laser—both of which have shown satisfactory resolution of lesions without recurrence at 1-year follow-up, with no major scarring reported.25,26 Alternatively, topical management with retinoids daily over a 4-month period leads to flattening of the tumors with no further appearance of new lesions.27 Despite the availability of numerous management options, establishing a first-line treatment remains controversial due to the high risk for recurrence and the variability in the number and location of lesions among individual patients. In our case, given the benign nature of syringomas, the asymptomatic nature of the lesions, the involvement of noncritical aesthetic areas, and the limited response to noninvasive therapeutic options, the patient was informed of the diagnosis, and no further pharmacologic or surgical intervention was pursued.
- Williams K, Shinkai K. Evaluation and management of the patient with multiple syringomas: a systematic review of the literature. J Am Acad Dermatol. 2016;74:1234-1240.E9. doi:10.1016 /j.jaad.2015.12.006
- Resende C, Araújo C, Santos R, et al. Late-onset of eruptive syringomas: a diagnostic challenge. An Bras Dermatol. 2015;90(3 suppl 1):239-241. doi:10.1590/abd1806-4841.20153899
- Friedman SJ, Butler DF. Syringoma presenting as milia. J Am Acad Dermatol. 1987;16:310-314.
- Avhad G, Ghuge P, Jerajani HR. Generalized eruptive syringoma. Indian J Dermatol. 2015;60:214. doi:10.4103/0019-5154.152586
- Ning WV, Bashey S, Cole C, et al. Multiple eruptive syringomas on the penis. Cutis. 2019;103:E15-E16.
- Cohen PR, Tschen JA, Rapini RP. Penile syringoma: reports and review of patients with syringoma located on the penis. J Clin Aesthet Dermatol. 2013;6:38-42.
- Jamalipour M, Heidarpour M, Rajabi P. Generalized eruptive syringomas. Indian J Dermatol. 2009;54:65-67. doi:10.4103/0019-5154.48992
- Mohaghegh F, Amiri A, Fatemi Naeini F, et al. Acral eruptive syringoma: an unusual presentation with misdiagnosis. Case Rep Dermatol Med. 2020;2020:5416285. doi:10.1155/2020/5416285
- Valdivielso-Ramos M, de la Cueva P, Gimeno M, et al. Acral syringomas. Actas Dermosifiliogr. 2010;101:458-460.
- Patel K, Lundgren AD, Ahmed AM, et al. Disseminated syringomas of the upper extremities in a young woman. Cureus. 2018;10:E3619. doi:10.7759/cureus.3619
- Balci DD, Atik E, Altintas S. Coexistence of acral syringomas and multiple trichoepitheliomas on the face. J Cutan Med Surg. 2009;13:169-171. doi:10.2310/7750.2008.08011
- Martín-García RF, Muñoz CM. Acral syringomas presenting as a photosensitive papular eruption. Cutis. 2006;77:33-36.
- Varas-Meis E, Prada-García C, Samaniego-González E, et al. Acral syringomas associated with hematological neoplasm. Indian J Dermatol Venereol Leprol. 2017;83:136. doi:10.4103/0378-6323.192961
- Berbis P, Fabre JF, Jancovici E, et al. Late-onset syringomas of the upper extremities associated with a carcinoid tumor. Arch Dermatol. 1989;125:848-849.
- Metze D, Jurecka W, Gebhart W. Disseminated syringomas of the upper extremities. case history and immunohistochemical and ultrastructural study. Dermatologica. 1990;180:228-235. doi:10.1159/000248036
- Gómez-de Castro C, Vivanco Allende B, García-García B. Multiple acral syringomas. siringomas acrales múltiples. Actas Dermosifiliogr (Engl Ed). 2018;109:834-836. doi:10.1016/j.ad.2017.10.014
- Hughes PS, Apisarnthanarax P. Acral syringoma. Arch Dermatol. 1977;113:1435-1436.
- Asai Y, Ishii M, Hamada T. Acral syringoma: electron microscopic studies on its origin. Acta Derm Venereol. 1982;62:64-68.
- van den Broek H, Lundquist CD. Syringomas of the upper extremities with onset in the sixth decade. J Am Acad Dermatol. 1982,6:534-536. doi:10.1016/S0190-9622(82)80368-X
- Garcia C, Krunic AL, Grichnik J, et al. Multiple acral syringomata with uniform involvement of the hands and feet. Cutis. 1997;59:213-214, 216.
- Patrizi A, Neri I, Marzaduri S, et al. Syringoma: a review of twenty-nine cases. Acta Derm Venereol. 1998;78:460-462.
- Iglesias Sancho M, Serra Llobet J, Salleras Redonnet M, et al. Siringomas disem- inados de inicio acral, aparecidos en la octava década. Actas Dermosifiliofr. 1999;90:253-257.
- Muniesa C, Fortuño Y, Moreno A, et al. Papules on the dorsum of the fingers. Actas Dermosifiliogr. 2008;99:812-813. doi:10.1016 /S1578-2190(08)70371-8
- Koh MJ. Multiple acral syringomas involving the hands. Clin Exp Dermatol. 2009;34:E438. doi:10.1111/j.1365-2230.2009.03462.x
- Karam P, Benedetto AV. Syringomas: new approach to an old technique. Int J Dermatol. 1996;35:219-220. doi:10.1111/j.1365-4362 .1996.tb01647.x
- Wang JI, Roenigk HH. Treatment of multiple facial syringomas with the carbon dioxide (CO2) laser. Dermatol Surg. 1999;25:136-139. doi:10.1046/j.1524-4725.1999.08111.x
- Gómez MI, Pérez B, Azaña JM, et al. Eruptive syringoma: treatment with topical tretinoin. Dermatology. 2009;189:105-106. doi:10.1159/000246803
- Williams K, Shinkai K. Evaluation and management of the patient with multiple syringomas: a systematic review of the literature. J Am Acad Dermatol. 2016;74:1234-1240.E9. doi:10.1016 /j.jaad.2015.12.006
- Resende C, Araújo C, Santos R, et al. Late-onset of eruptive syringomas: a diagnostic challenge. An Bras Dermatol. 2015;90(3 suppl 1):239-241. doi:10.1590/abd1806-4841.20153899
- Friedman SJ, Butler DF. Syringoma presenting as milia. J Am Acad Dermatol. 1987;16:310-314.
- Avhad G, Ghuge P, Jerajani HR. Generalized eruptive syringoma. Indian J Dermatol. 2015;60:214. doi:10.4103/0019-5154.152586
- Ning WV, Bashey S, Cole C, et al. Multiple eruptive syringomas on the penis. Cutis. 2019;103:E15-E16.
- Cohen PR, Tschen JA, Rapini RP. Penile syringoma: reports and review of patients with syringoma located on the penis. J Clin Aesthet Dermatol. 2013;6:38-42.
- Jamalipour M, Heidarpour M, Rajabi P. Generalized eruptive syringomas. Indian J Dermatol. 2009;54:65-67. doi:10.4103/0019-5154.48992
- Mohaghegh F, Amiri A, Fatemi Naeini F, et al. Acral eruptive syringoma: an unusual presentation with misdiagnosis. Case Rep Dermatol Med. 2020;2020:5416285. doi:10.1155/2020/5416285
- Valdivielso-Ramos M, de la Cueva P, Gimeno M, et al. Acral syringomas. Actas Dermosifiliogr. 2010;101:458-460.
- Patel K, Lundgren AD, Ahmed AM, et al. Disseminated syringomas of the upper extremities in a young woman. Cureus. 2018;10:E3619. doi:10.7759/cureus.3619
- Balci DD, Atik E, Altintas S. Coexistence of acral syringomas and multiple trichoepitheliomas on the face. J Cutan Med Surg. 2009;13:169-171. doi:10.2310/7750.2008.08011
- Martín-García RF, Muñoz CM. Acral syringomas presenting as a photosensitive papular eruption. Cutis. 2006;77:33-36.
- Varas-Meis E, Prada-García C, Samaniego-González E, et al. Acral syringomas associated with hematological neoplasm. Indian J Dermatol Venereol Leprol. 2017;83:136. doi:10.4103/0378-6323.192961
- Berbis P, Fabre JF, Jancovici E, et al. Late-onset syringomas of the upper extremities associated with a carcinoid tumor. Arch Dermatol. 1989;125:848-849.
- Metze D, Jurecka W, Gebhart W. Disseminated syringomas of the upper extremities. case history and immunohistochemical and ultrastructural study. Dermatologica. 1990;180:228-235. doi:10.1159/000248036
- Gómez-de Castro C, Vivanco Allende B, García-García B. Multiple acral syringomas. siringomas acrales múltiples. Actas Dermosifiliogr (Engl Ed). 2018;109:834-836. doi:10.1016/j.ad.2017.10.014
- Hughes PS, Apisarnthanarax P. Acral syringoma. Arch Dermatol. 1977;113:1435-1436.
- Asai Y, Ishii M, Hamada T. Acral syringoma: electron microscopic studies on its origin. Acta Derm Venereol. 1982;62:64-68.
- van den Broek H, Lundquist CD. Syringomas of the upper extremities with onset in the sixth decade. J Am Acad Dermatol. 1982,6:534-536. doi:10.1016/S0190-9622(82)80368-X
- Garcia C, Krunic AL, Grichnik J, et al. Multiple acral syringomata with uniform involvement of the hands and feet. Cutis. 1997;59:213-214, 216.
- Patrizi A, Neri I, Marzaduri S, et al. Syringoma: a review of twenty-nine cases. Acta Derm Venereol. 1998;78:460-462.
- Iglesias Sancho M, Serra Llobet J, Salleras Redonnet M, et al. Siringomas disem- inados de inicio acral, aparecidos en la octava década. Actas Dermosifiliofr. 1999;90:253-257.
- Muniesa C, Fortuño Y, Moreno A, et al. Papules on the dorsum of the fingers. Actas Dermosifiliogr. 2008;99:812-813. doi:10.1016 /S1578-2190(08)70371-8
- Koh MJ. Multiple acral syringomas involving the hands. Clin Exp Dermatol. 2009;34:E438. doi:10.1111/j.1365-2230.2009.03462.x
- Karam P, Benedetto AV. Syringomas: new approach to an old technique. Int J Dermatol. 1996;35:219-220. doi:10.1111/j.1365-4362 .1996.tb01647.x
- Wang JI, Roenigk HH. Treatment of multiple facial syringomas with the carbon dioxide (CO2) laser. Dermatol Surg. 1999;25:136-139. doi:10.1046/j.1524-4725.1999.08111.x
- Gómez MI, Pérez B, Azaña JM, et al. Eruptive syringoma: treatment with topical tretinoin. Dermatology. 2009;189:105-106. doi:10.1159/000246803
Eruptive Erythematous Papules on the Forearms
Eruptive Erythematous Papules on the Forearms
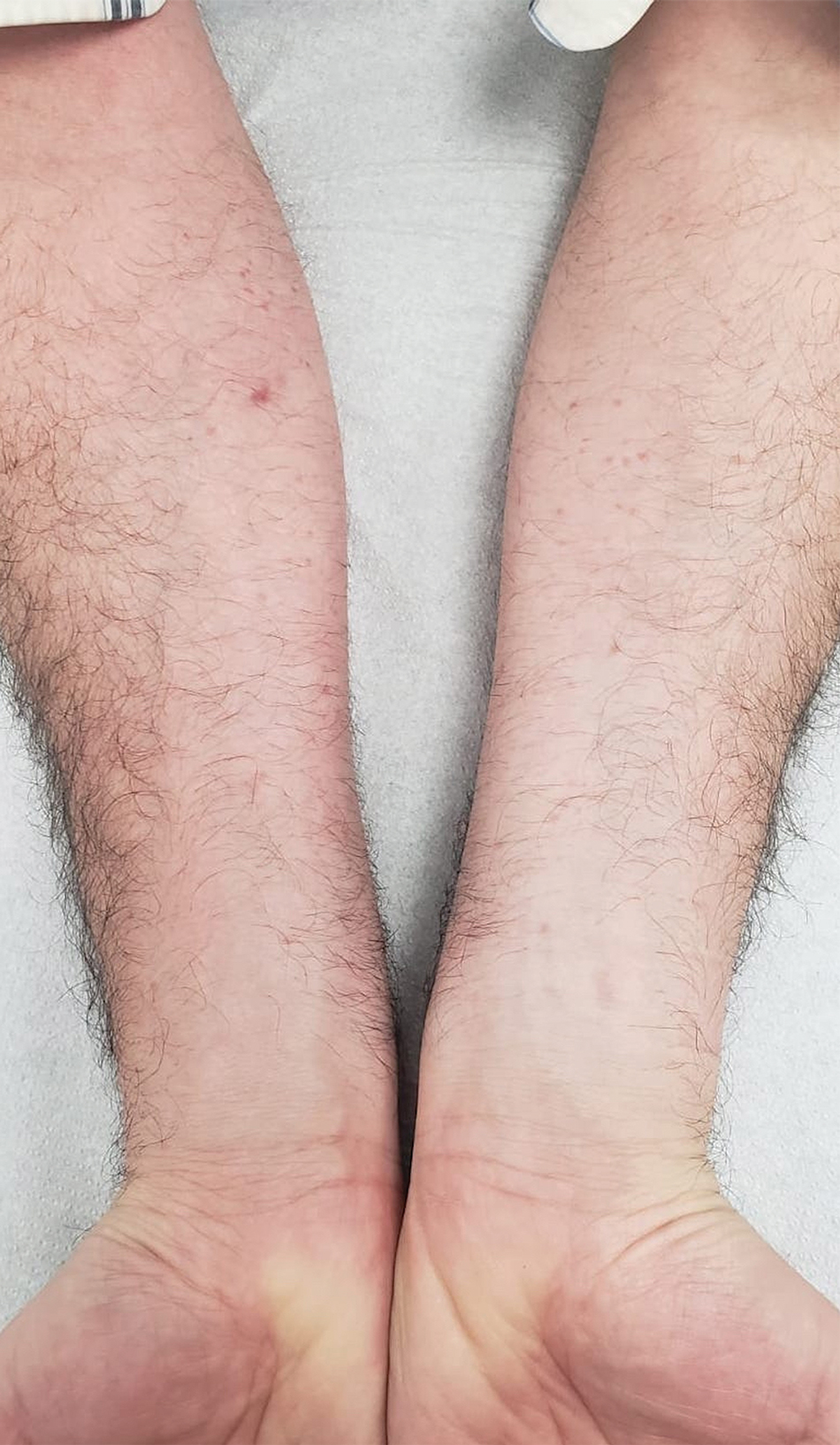
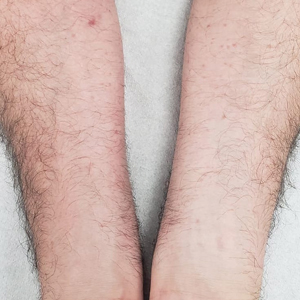
A 44-year-old man presented to the dermatology department with multiple eruptive, nonconfluent, erythematous papules on the anterior forearms of 2 years’ duration. The patient’s medical history was notable for right-sided testicular cancer diagnosed in childhood and 3 excised basal cell carcinomas, the most recent of which was concurrent with the present case. The patient denied any recent pruritus, exposure to irritants, or use of over-the-counter medications. Physical examination was remarkable for numerous monomorphic, symmetric, nonconfluent, flesh-colored to slightly pigmented papules on the dorsal aspect of the forearms. No involvement of the fingers or lower extremities was observed. Two punch biopsies of representative lesions on the right and left forearms were taken. Histopathologic examination revealed eccrine ductal proliferations lined by cuboidal cells embedded within bundles of sclerotic collagen.