User login
Pneumonia is a common reason for hospitalization and major rationale for administration of antibiotics in the United States.[1] Management guidelines for patients hospitalized with community‐acquired pneumonia recommend early antibiotic therapy. Quality measures adopted by the Centers for Medicare and Medicaid Services include antibiotic administration within 6 hours of presentation, based on a relationship between early administration and improved survival.[2, 3, 4, 5] However, this imperative has been associated with an increase in incorrect diagnoses of pneumonia.[6, 7] While pneumonia diagnosis would seem straightforward, clinical findings frequently do not differentiate pneumonia from other respiratory illnesses. Thus, an infiltrate on chest radiographs (CXR) is commonly used to assign an admitting diagnosis of pneumonia.[8, 9, 10] However, radiographic reports are often inconclusive and frequently fail to discriminate pneumonic infiltrates from atelectasis, edema, small pleural effusions, or chronic abnormalities. In cases of uncertainty, physicians invariably initiate antibiotics, even when the illness is more consistent with viral bronchitis or asthma, resulting in unnecessary antibiotic use.[6] Previously considered relatively harmless, antibiotic complications can be lethal, and excessive use promotes antimicrobial resistance.[11, 12]
Procalcitonin (ProCT), a calcitonin precursor normally produced in the thyroid and lungs, is secreted by cells throughout the body in response to bacterial infections.[13, 14] Elevated ProCT is used to screen patients with suspected bacterial infection and predict mortality in critically ill patients.[15, 16] Recently, European investigators have used ProCT to guide more selective antibiotic use in patients with symptoms of lower respiratory tract infection (LRTI), including pneumonia.[17, 18, 19] However, few reports describe the relationship of ProCT with radiographic features in hospitalized patients with LRTI, and none focus on patients with indeterminate radiographic readings.[18, 20, 21]
We sought to determine if elevated serum ProCT in adults hospitalized with LRTI symptoms correlates with the clinical diagnosis of pneumonia and definitive infiltrates on CXRs. We specifically assessed ProCT as a diagnostic marker of pneumonia to augment clinical judgment in patients when radiographic findings are indeterminate.
METHODS
Subjects
The study was performed in a 520‐bed, general medicalsurgical hospital, using subjects participating in a study of the relationship between biomarkers and microbiologic diagnoses (to be described in a separate publication). Adults 21 years of age admitted with a diagnosis compatible with respiratory tract infection (pneumonia, acute exacerbations of chronic obstructive pulmonary disease [AECOPD], acute bronchitis, asthma, upper respiratory infection, viral syndrome, respiratory failure, and congestive heart failure [CHF] with signs of infection) were recruited during winters of 20082009 and 20092010. Patients were screened within 24 hours of admission; immunosupression, lung abscess, witnessed aspiration, or previous antibiotic use were exclusion criteria. Blood for ProCT measurement was collected in the emergency room prior to antibiotics. Subjects or legal guardians provided written informed consent, and institutional review boards approved the study.
Illness Evaluation
At enrollment, demographic, clinical, and laboratory data were collected. To provide consistency of data, 1 investigator (a clinical pulmonary specialist), after interviewing, examining each subject, and considering the radiographic findings, assigned a primary and secondary clinical admitting diagnosis: pneumonia, AECOPD, asthma exacerbation, CHF, acute bronchitis, viral syndrome or influenza, other respiratory illness, or non‐respiratory illness. These diagnoses were used for the analysis but were not available to treating physicians. Discharge diagnoses, based on International Classification of Diseases, Ninth Revision (ICD‐9) codes, and official results of follow‐up chest radiographs were also recorded.
Radiographic Interpretations
All official admission chest radiographic reports were reviewed by one of the investigators. Radiographic descriptions were noted with attention to the following comments: clear or no acute change from prior radiographs, infiltrates without consolidation, consolidation, edema, atelectasis, and pleural effusion. For our analysis, chest radiographs were categorized into 4 groups: (1) No acute disease (NAD) (ie, clear or no change from prior radiographs); (2) Other definitive radiographic abnormality (atelectasis, edema, or pleural effusion); (3) Indeterminate (findings consistent with pneumonia, but also other processes including atelectasis, edema, scarring but with no favored interpretation [often including the phrase pneumonia cannot be excluded]); and (4) infiltrate (ie, an infiltrate or consolidation most consistent with pneumonia). The pulmonary specialist independently categorized CXRs and provided a definitive radiographic interpretation whenever possible.
Microbiology
Blood cultures, sputum Gram stain and culture were performed by the clinical laboratory. Pneumococcal‐specific urinary antigen and serology were performed as previously described.[22]
Procalcitonin Measurement
Serum ProCT was measured using resolved amplified cryptate emission technology (Kryptor PCT, Brahms, Henningsdorf, Germany). Functional sensitivity is 0.06 ng/mL (normal serum levels are 0.033 0.003 ng/mL).[23, 24, 25] ProCT results were not available to investigators at the time of clinical or radiographic assessments.
Statistical Analysis
Categorical and continuous distributions were compared using Fisher's exact and Wilcoxon tests, respectively, and summarized by proportions and medians, and means and standard deviations. Log‐axis boxplots were used to graphically display the skewed distributions of ProCT. Stacked bar charts were used to depict discrete distributions of diagnoses by ProCT group. Receiver operating characteristic (ROC) curves relating ProCT to chest radiographs were computed, and sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) were calculated for ProCT thresholds. Logistic regression was used to estimate odds ratios (OR), and associated P values and confidence intervals, to quantify associations between infiltrate/non‐infiltrate and dichotomized or log‐transformed ProCT. SAS 9.2 (SAS Institute, Inc, Cary, NC) was used for analyses, using 2‐sided 0.05 level tests.
RESULTS
Study Population and Clinical Diagnoses
During 2 winters, 532 subjects admitted for 556 respiratory illnesses were recruited. Two did not have chest radiographs, 16 illnesses were non‐pulmonary, and 10 did not have admission ProCT levels, leaving 528 illnesses for analysis (Table 1). Subjects averaged 65 years of age, predominately lived at home, and a high percentage had underlying diseases. The leading primary admission diagnoses assigned by the pulmonologist were pneumonia and AECOPD at 31% and 27%, respectively. Of the 163 illnesses assigned a primary or secondary admitting diagnosis of pneumonia, infiltrates were identified on the admission radiograph by the pulmonologist in 156 (96%).
| Illnesses N = 528 (%) | |
|---|---|
| |
| Age (mean SD) | 65 17 |
| Female | 278 (53) |
| Race | |
| White | 409 (77) |
| African American | 119 (23) |
| Hispanic | 53 (10) |
| Residence | |
| Community | 488 (92) |
| Assisted living | 27 (5) |
| Nursing home | 13 (2) |
| Medical conditions | |
| Diabetes | 188 (36) |
| CHF | 153 (29) |
| COPD | 216 (41) |
| Chronic renal failure | 21 (4) |
| Past or active smoker | 395 (75) |
| Current influenza vaccine | 371 (70) |
| Prior pneumococcal vaccine | 432 (82) |
| Oral steroids | 69 (13) |
| Inhaled steroids | 201 (38) |
| Home oxygen | 129 (24) |
| Symptoms, No. (%) | |
| Upper respiratory | 350 (66) |
| Cough | 489 (93) |
| Sputum production | 390 (74) |
| Dyspnea | 496 (94) |
| Wheezing | 345 (65) |
| Constitutional | 271 (51) |
| Feverish | 293 (55) |
| Rigors | 118 (22) |
| Physical exam | |
| Wheezing | 279 (53) |
| Rales | 211 (40) |
| Rhonchi | 144 (27) |
| Temperature (mean SD) | 37.2 1.0 |
| Respiratory rate (mean SD) | 25 7 |
| Laboratory data | |
| Oxygen saturation (mean SD) | 92.3 5.5 |
| WBC 103/ml (mean SD) | 9.4 4.8 |
| Clinical diagnosis | |
| Pneumonia | 163 (31) |
| Asthma | 76 (14) |
| CHF | 45 (9) |
| Bronchitis | 83 (16) |
| AECOPD | 142 (27) |
| Other pulmonary | 19 (4) |
| Therapy | |
| Antibacterials | 463 (88) |
| Antivirals | 38 (7) |
| Outcome | |
| Intensive care | 43 (8) |
| Respiratory failure | 29 (6) |
| Death | 6 (1) |
| Length of stay (mean SD) | 5.9 18.7 |
Radiographic Classifications
Based on radiology reports, 213 (40%) were classified as NAD, 76 (14%) as other definitive findings, 75 (14%) as infiltrates, and 164 (31%) as indeterminate. The pulmonologist concurred with the radiology report for most NAD (199/213) and infiltrate (67/75) classifications, but only 4 as indeterminate, assigning approximately half the radiologist's indeterminate CXRs to infiltrate and one‐quarter each to NAD and other categories.
Relationship of ProCT With Clinical and Radiographic Features
The relationship between clinical admitting diagnosis and ProCT is shown in Figure 1A. Subjects with pneumonia had a median ProCT of 0.27 ng/ml (interquartile range [IQR] 1.3), significantly greater than subjects with AECOPD, asthma, acute bronchitis, and viral/emnfluenza. Subjects assigned to the other diagnoses category (ie, skin or urinary infections, empyema) generally had higher ProCT values (median 0.70 ng/ml, IQR 4.6).
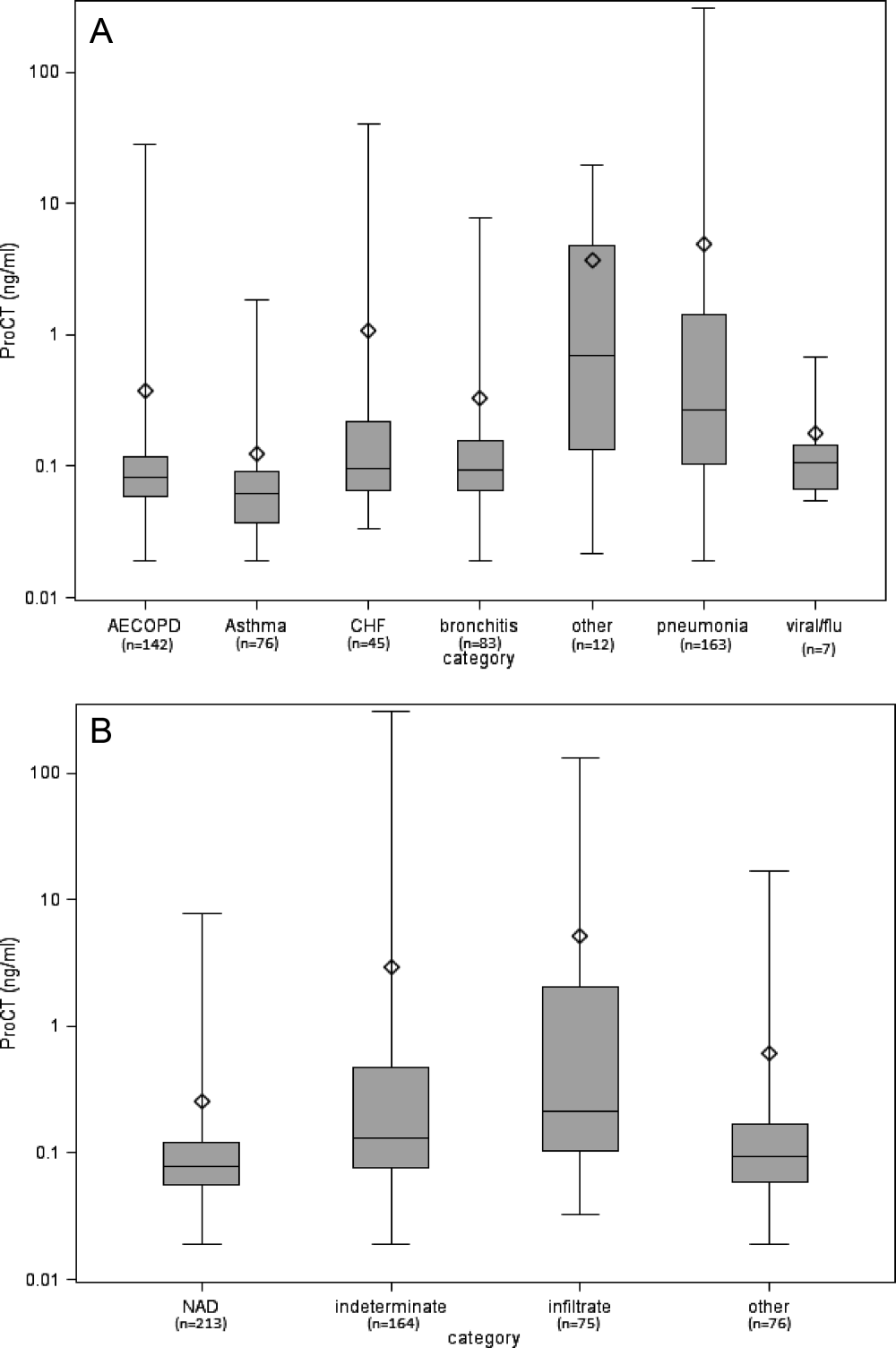
The relationship between the radiologist's classification and ProCT is shown in Figure 1B. Median ProCT levels were significantly lower in subjects with radiographs classified as NAD compared to those showing infiltrates (P < 0.0001). Those classified as indeterminate in radiology reports had a median ProCT value (0.13 ng/ml) midway between those with NAD (0.08 ng/ml) and infiltrates (0.21 ng/ml). Similarly, illnesses with radiographs classified by the pulmonologist as infiltrates also had significantly higher median ProCT levels (0.33 ng/ml, IQR 1.33) than those classified as NAD (0.08 ng/ml, IQR 0.06; P < 0.0001), and other definitive findings (0.11 ng/ml, IQR 0.17). Notably, duration of symptoms prior to evaluation did not affect the relationship between ProCT level and radiographic classification (data not shown). All subjects with infiltrates according to the radiologist's report received early antibiotics, as did 97% and 80% with indeterminate radiographs and NAD, respectively.
Predictive Value of ProCT for Interpreting Indeterminate Radiographs
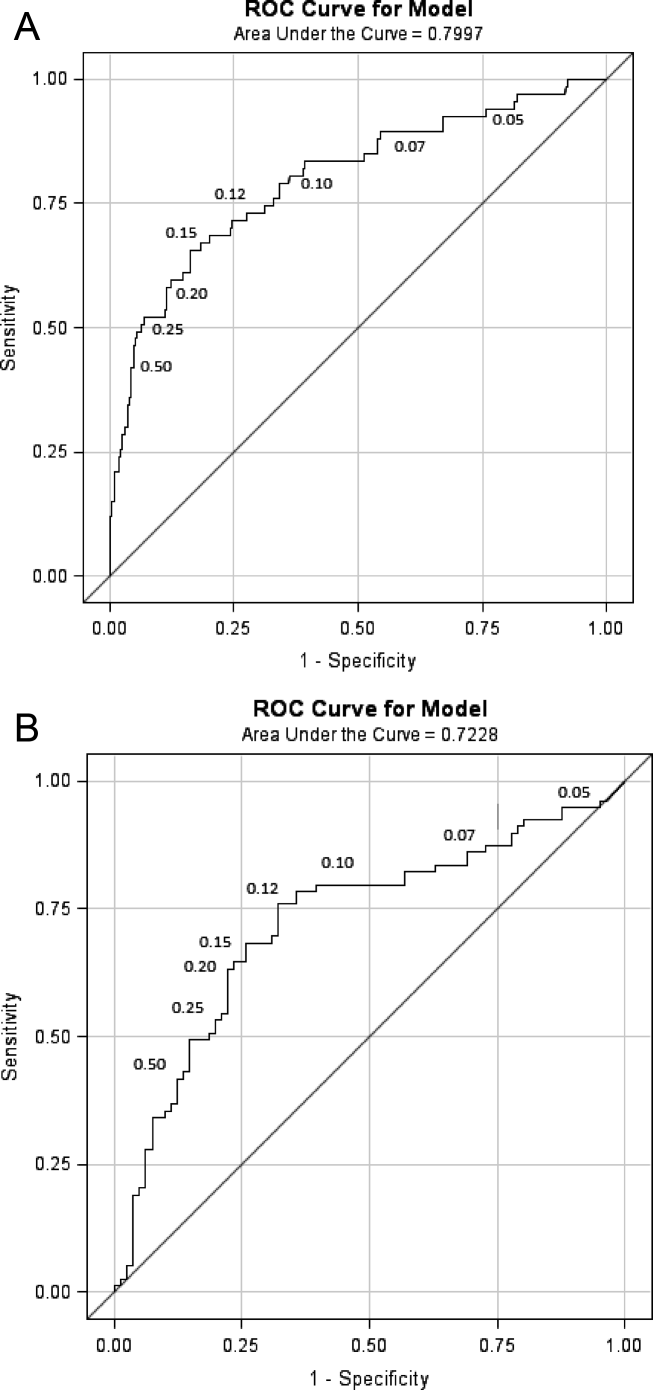
We were specifically interested to learn if ProCT could help clinicians interpret the large number of indeterminate radiographs. As a first step, we evaluated the diagnostic accuracy of ProCT for radiographic infiltrates by calculating the ROC curve (Figure 2A), using cases in which radiologist and pulmonologist concurred on the classification as infiltrate (n = 67) or no infiltrate (ie, NAD or other definitive finding; n = 273). For these cases, the ROC had an area under curve (AUC) of 0.80 (P < 0.0001), indicating moderate predictive accuracy of ProCT for the presence of an infiltrate. The OR for an infiltrate increased steadily with higher ProCT thresholds, quadrupling from 3.9 to 15.6 as the threshold increased from 0.05 to 0.50 ng/ml (Table 2). Using the commonly defined 0.25 ng/ml ProCT threshold for which antibiotics have been recommended for respiratory infections, the PPV for an infiltrate was 61% and the NPV 89%.
| ProCT Threshold (ng/ml) | Sensitivity (%) | Specificity (%) | OR | P Value |
|---|---|---|---|---|
| ||||
| 0.05 | 94 | 20 | 3.9 | 0.012 |
| 0.07 | 90 | 42 | 6.1 | <0.0001 |
| 0.10 | 79 | 65 | 7.1 | <0.0001 |
| 0.12 | 73 | 72 | 7.1 | <0.0001 |
| 0.15* | 67 | 82 | 9.1 | <0.0001 |
| 0.20 | 58 | 88 | 10.5 | <0.0001 |
| 0.25 | 52 | 92 | 12.5 | <0.0001 |
| 0.50 | 42 | 96 | 15.6 | <0.0001 |
Next, we analyzed the 164 illnesses with indeterminate radiographs. Of these, the pulmonologist classified 79 as infiltrate (median ProCT 0.29 ng/ml), 40 as NAD (median ProCT 0.08 ng/ml), 41 as other definitive findings (median ProCT 0.10 ng/ml), and 4 as indeterminate. The admitting diagnosis and corresponding median ProCT for these subjects was pneumonia in 78 (0.28 ng/ml), AECOPD in 26 (0.09 ng/ml), acute bronchitis in 30 (0.09 ng/ml), asthma in 11 (0.08 ng/ml), and CHF in 14 (0.10 ng/ml). The ROC AUC for these cases (excluding 4 considered indeterminate by the pulmonologist) was slightly lower (0.72, P < 0.0001) than the prior analysis (Figure 2B), but ProCT retained moderate predictive value for the presence of infiltrates as diagnosed by the pulmonologist on indeterminate radiographs (Table 3).
| ProCT Threshold | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | OR | P Value | Predicted Probability (%) |
|---|---|---|---|---|---|---|---|
| |||||||
| 0.05 | 95 | 7 | 50 | 60 | 1.5 | 0.54 | 32 |
| 0.07 | 86 | 27 | 54 | 67 | 2.3 | 0.04 | 35 |
| 0.10 | 80 | 59 | 66 | 75 | 5.7 | <0.0001 | 40 |
| 0.12* | 76 | 67 | 69 | 74 | 6.3 | <0.0001 | 42 |
| 0.15 | 67 | 74 | 72 | 70 | 5.8 | <0.0001 | 45 |
| 0.20 | 63 | 78 | 74 | 69 | 6.0 | <0.0001 | 49 |
| 0.25 | 53 | 80 | 72 | 64 | 4.6 | <0.0001 | 52 |
| 0.50 | 37 | 88 | 74 | 59 | 4.1 | 0.0006 | 60 |
Since clinical determinations by a single pulmonologist may be subjective, we sought to objectively assess the accuracy of assigned admission diagnoses by analyzing additional data, including bacterial tests, follow‐up radiographs, and final discharge diagnoses. As a surrogate of invasive bacterial infection, we used identification of S. pneumoniae as the outcome, since multiple complementary diagnostic assays were used, thus minimizing the uncertainty of sputum cultures. Overall, we identified 58 pneumococcal infections (Table 4). The pulmonologist's admitting diagnosis of pneumonia captured a higher proportion (52% for any test positive) of S. pneumoniae cases than identification of a radiographic infiltrate by the radiologist (26%), and equivalent to the pulmonologist's radiographic reading (57%) or elevated ProCT alone (57%). There was an absolute 14% increase (27% relative increase) in the detection of S. pneumoniae infections by addition of ProCT to the pulmonologist's diagnosis. A similar analysis limited to the 164 subjects with indeterminate radiographs found that 13 of 19 (68%) of S. pneumoniae diagnoses had been assigned a clinical diagnosis of pneumonia by the pulmonologist.
| S. pneumonia Confirmed by Diagnostic Assay No. (%) | |||||
|---|---|---|---|---|---|
| Criteria | Blood Culture + (n = 7) | Urine Ag + (n = 27) | Serology + (n = 28) | Sputum + (n = 20) | Any Test + (n = 58) |
| |||||
| Radiologist CXR read as infiltrate | 3 (43) | 9 (33) | 10 (36) | 3 (15) | 15 (26) |
| Pulmonologist CXR read as infiltrate | 6 (86) | 17 (62) | 18 (64) | 10 (50) | 33 (57) |
| Pulmonologist clinical diagnosis of pneumonia | 6 (86) | 16 (60) | 16 (57) | 8 (40) | 30 (52) |
| Pulmonologist clinical diagnosis of AECOPD | 0 (0) | 4 (15) | 5 (18) | 5 (25) | 8 (14) |
| Procalcitonin 0.25 ng/ml | 6 (86) | 18 (67) | 17 (61) | 9 (45) | 33 (57) |
| Pulmonologist diagnosis of pneumonia and/or ProCT 0.25 ng/ml | 7 (100) | 19 (70) | 20 (71) | 10 (50) | 38 (66) |
We also determined how often subsequent radiographs showed evolution to a definitive infiltrate in indeterminate cases that the pulmonologist had classified as either NAD or other finding. Only 3/20 with repeat films showed a definitive infiltrate, none of whom had evidence of bacterial infection, and all 3 had ProCT <0.25 ng/ml. In contrast, 10 of 21 subjects with indeterminate CXRs, that the pulmonologist had classified as an infiltrate, developed definitive infiltrates on follow‐up studies (P = 0.04), and 8 remained indeterminate.
Finally, 93% of those assigned an admitting pneumonia diagnosis by the pulmonologist had primary (71%) or secondary (22%) discharge diagnosis of pneumonia. Of the additional 28 subjects with a discharge diagnosis of pneumonia, for whom the pulmonologist assigned non‐pneumonia admitting diagnoses, none had positive blood cultures or evidence of S. pneumoniae infection, and only one had a follow‐up radiograph showing a definitive infiltrate.
DISCUSSION
Pneumonia can be a serious illness and, if left untreated, result in death. Hence, infectious disease and pulmonary societies advocate timely administration of antibiotics. Unfortunately, clinical diagnosis of pneumonia is difficult, as clinical and radiographic features are often ambiguous. These factors result in liberal use of broad‐spectrum antibiotics, often in patients with viral infection, in whom antibiotic side effects outweigh benefit.[26] Although pneumonia guidelines recommend early antibiotic therapy for patients with infiltrates on CXR, we found that early administration of antibiotics for possible pneumonia is nearly universal.[2, 27] Thus, an objective laboratory test indicative of pneumonia would be a useful adjunct when making treatment decisions.
In an attempt to curb excessive antibiotic use, European investigators have used ProCT to guide therapy for respiratory tract infection, including pneumonia, without increased adverse outcomes.[17, 19, 28, 29] Antibiotics are recommended when ProCT levels are 0.25 ng/ml, rather than based on radiographic or microbiological results. However, ProCT‐guided antibiotic management has not been adopted in the United States, and radiographic findings are a major determinant for diagnosis of pneumonia and antibiotic administration.
Thus, we were interested in correlating ProCT levels with the presence of radiographic infiltrates, and especially how ProCT could assist in interpreting the vexing problem of indeterminate CXRs. We found a direct relationship between an increased ProCT level and definitive infiltrates on CXR, similar to results reported by others.[18, 20, 21] However, these studies did not address illnesses with indeterminate radiographs, which accounted for approximately one‐third of the radiologist's readings in our study. Our situation is likely not unique. In a post hoc analysis of CXRs from subjects with pneumonia in the PORT study, independent radiologists interpreted a similar proportion of CXRs as either no acute disease or possible infiltrates.[30] Since indeterminate readings include the possibility of an infiltrate, it is not surprising that 97% of our subjects received early antibiotic treatment.
Given current pneumonia treatment guidelines, ProCT testing for those with definitive infiltrates would not add to patient management, as all should receive early antibiotics. However, ProCT measurement could have significant impact on management in those with indeterminate films. We found ProCT was moderately predictive for infiltrates when utilizing the pulmonologist's interpretation of indeterminate CXRs. This is concordant with a recent study reporting a similar ROC (0.72) for ProCT for diagnosing pneumonia in patients with dyspnea, and more predictive of pneumonia than any clinical variable.[31]
The major limitation of our study is the lack of a gold standard for diagnosis of pneumonia. We chose to use a pulmonary specialist to provide admission diagnoses, and independently interpret radiographs in the context of clinical findings. Although imperfect, we feel this approach best reflects good medical practice by an experienced clinician evaluating the CXR in context of a patient's history and physical exam. Importantly, analysis of the concurrent microbiology data, follow‐up radiographs, and discharge diagnoses supports the accuracy of the admitting diagnoses of pneumonia and radiographic interpretations by the pulmonologist. Secondly, our findings may not be applicable to immunocompromised or previously treated patients.
How should a busy physician respond to a patient with dyspnea and fever when faced with an indeterminate radiographic report, since no method for the diagnosis of pneumonia is infallible?[8, 32] Clearly, a pulmonary consultation on all such patients is impractical. Our data confirms previous results that high ProCT values are correlated with an infiltrate on CXR and low values have a NPV of nearly 90%. Furthermore, low ProCT values predict low mortality across all levels of standard severity scoring indexes.[33] Thus, a low ProCT may help clinicians feel more confident to delay or stop antibiotics in a patient in whom clinical suspicion for bacterial pneumonia is low, yet the radiographic report states pneumonia cannot be ruled out.[34] Such strategies may assist in reducing unnecessary antibiotic use in the United States.[35]
Disclosures
Funding for this study was provided by NIAID‐1R01AI079446‐01. This work was presented in part at the Infectious Diseases Society of America, Boston, MA, October 2011. The authors report no conflicts of interest.
- , , , et al. The burden of community‐acquired pneumonia in seniors: results of a population‐based study. Clin Infect Dis. 2004;39:1642–1650.
- , , , et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community‐acquired pneumonia in adults. Clin Infect Dis. 2007;44(suppl 2):S27–S72.
- Centers for Medicare and Medicaid Services. The Medicare national pneumonia quality improvement project. Available at: http://www.cms.gov/HospitalQualityInits/. Accessed September 8, 2011.
- , , , , . Timing of antibiotic administration and outcomes for Medicare patients hospitalized with community‐acquired pneumonia. Arch Intern Med. 2004;164:637–644.
- , , , et al. Quality of care, process, and outcomes in elderly patients with pneumonia. JAMA. 1997;278:2080–2084.
- , , , . Misdiagnosis of community‐acquired pneumonia and inappropriate utilization of antibiotics: side effects of the 4‐h antibiotic administration rule. Chest. 2007;131:1865–1869.
- , , . Antibiotic timing and errors in diagnosing pneumonia. Arch Intern Med. 2008;168:351–356.
- , , , . Patients admitted to hospital with suspected pneumonia and normal chest radiographs: epidemiology, microbiology, and outcomes. Am J Med. 2004;117:305–311.
- , , , , . Reliability of radiographic findings and the relation to etiologic agents in community‐acquired pneumonia. Respir Med. 2006;100:926–932.
- , , , . Admission chest radiograph lacks sensitivity in the diagnosis of community‐acquired pneumonia. Am J Med Sci. 2009;337:236–240.
- , . Strategies for reduction in duration of antibiotic use in hospitalized patients. Clin Infect Dis. 2011;52:1232–1240.
- , , , et al. The epidemic of antibiotic‐resistant infections: a call to action for the medical community from the Infectious Diseases Society of America. Clin Infect Dis. 2008;46:155–164.
- , , , , . Clinical review 167: procalcitonin and the calcitonin gene family of peptides in inflammation, infection, and sepsis: a journey from calcitonin back to its precursors. J Clin Endocrinol Metab. 2004;89:1512–1525.
- , , . Procalcitonin assay in systemic inflammation, infection, and sepsis: clinical utility and limitations. Crit Care Med. 2008;36:941–952.
- , , . Procalcitonin in sepsis and systemic inflammation: a harmful biomarker and a therapeutic target. Br J Pharmacol. 2010;159:253–264.
- , , , , . Pneumonitis‐associated hyperprocalcitoninemia. Am J Med Sci. 1996;312:12–18.
- , , , et al. Antibiotic treatment of exacerbations of COPD: a randomized, controlled trial comparing procalcitonin‐guidance with standard therapy. Chest. 2007;131:9–19.
- , , , et al. Procalcitonin versus C‐reactive protein for predicting pneumonia in adults with lower respiratory tract infection in primary care. Br J Gen Pract. 2007;57:555–560.
- , , , et al. Effect of procalcitonin‐based guidelines vs standard guidelines on antibiotic use in lower respiratory tract infections: the ProHOSP randomized controlled trial. JAMA. 2009;302:1059–1066.
- , , , et al. Procalcitonin and C‐reactive protein in hospitalized adult patients with community‐acquired pneumonia or exacerbation of asthma or COPD. Chest. 2011;139:1410–1418.
- , , , et al. Diagnostic and prognostic accuracy of clinical and laboratory parameters in community‐acquired pneumonia. BMC Infect Dis. 2008;7:10.
- , , , et al. Utility of serum procalcitonin values in patients with acute exacerbations of chronic obstructive pulmonary disease: a cautionary note. Int J Chron Obstruct Pulmon Dis. 2112;7:127–135.
- , , . Procalcitonin and its component peptides in systemic inflammation: immunochemical characterization. J Investig Med. 1997;45:552–560.
- , , , . The future diagnostic role of procalcitonin levels: the need for improved sensitivity. Clin Infect Dis. 2003;36:823–824.
- , , , et al. Calcitonin precursor levels in human medullary thyroid carcinoma. Thyroid 2003;13:819–822.
- , , , . Emergency department visits for antibiotic‐associated adverse events. Clin Infect Dis. 2008;47:735–743.
- , , , et al. Guideline tyranny: a response to the article by Baum and Kaltsas. Clin Infect Dis. 2008;47:1117–1118.
- , , , et al. Procalcitonin guidance of antibiotic therapy in community‐acquired pneumonia: a randomized trial. Am J Respir Crit Care Med. 2006;174:84–93.
- , , , et al. Effect of procalcitonin‐guided treatment on antibiotic use and outcome in lower respiratory tract infections: cluster‐randomised, single‐blinded intervention trial. Lancet. 2004;363:600–607.
- , , , et al. Interobserver reliability of the chest radiograph in community‐acquired pneumonia. PORT investigators. Chest. 1996;110:343–350.
- , , , et al. Use of procalcitonin for the diagnosis of pneumonia in patients presenting with a chief complaint of dyspnea: results from the BACH (Biomarkers in Acute Heart Failure) trial. Eur J Heart Fail. 2012;14:278–286.
- , , , , . A multicenter analysis of the ED diagnosis of pneumonia. Am J Emerg Med. 2010;28:862–865.
- , , , et al. Procalcitonin predicts patients at low risk of death from community‐acquired pneumonia across all CRB‐65 classes. Eur Respir J. 2008;31:349–355.
- , , , , , . Antibiotic timing and diagnostic uncertainty in Medicare patients with pneumonia: is it reasonable to expect all patients to receive antibiotics within 4 hours? Chest. 2006;130:16–21.
- . Procalcitonin for triage of patients with respiratory tract symptoms: a case study in the trial design process for approval of a new diagnostic test for lower respiratory tract infection. Clin Infect Dis. 2011;52(S4):S351–S356.
Pneumonia is a common reason for hospitalization and major rationale for administration of antibiotics in the United States.[1] Management guidelines for patients hospitalized with community‐acquired pneumonia recommend early antibiotic therapy. Quality measures adopted by the Centers for Medicare and Medicaid Services include antibiotic administration within 6 hours of presentation, based on a relationship between early administration and improved survival.[2, 3, 4, 5] However, this imperative has been associated with an increase in incorrect diagnoses of pneumonia.[6, 7] While pneumonia diagnosis would seem straightforward, clinical findings frequently do not differentiate pneumonia from other respiratory illnesses. Thus, an infiltrate on chest radiographs (CXR) is commonly used to assign an admitting diagnosis of pneumonia.[8, 9, 10] However, radiographic reports are often inconclusive and frequently fail to discriminate pneumonic infiltrates from atelectasis, edema, small pleural effusions, or chronic abnormalities. In cases of uncertainty, physicians invariably initiate antibiotics, even when the illness is more consistent with viral bronchitis or asthma, resulting in unnecessary antibiotic use.[6] Previously considered relatively harmless, antibiotic complications can be lethal, and excessive use promotes antimicrobial resistance.[11, 12]
Procalcitonin (ProCT), a calcitonin precursor normally produced in the thyroid and lungs, is secreted by cells throughout the body in response to bacterial infections.[13, 14] Elevated ProCT is used to screen patients with suspected bacterial infection and predict mortality in critically ill patients.[15, 16] Recently, European investigators have used ProCT to guide more selective antibiotic use in patients with symptoms of lower respiratory tract infection (LRTI), including pneumonia.[17, 18, 19] However, few reports describe the relationship of ProCT with radiographic features in hospitalized patients with LRTI, and none focus on patients with indeterminate radiographic readings.[18, 20, 21]
We sought to determine if elevated serum ProCT in adults hospitalized with LRTI symptoms correlates with the clinical diagnosis of pneumonia and definitive infiltrates on CXRs. We specifically assessed ProCT as a diagnostic marker of pneumonia to augment clinical judgment in patients when radiographic findings are indeterminate.
METHODS
Subjects
The study was performed in a 520‐bed, general medicalsurgical hospital, using subjects participating in a study of the relationship between biomarkers and microbiologic diagnoses (to be described in a separate publication). Adults 21 years of age admitted with a diagnosis compatible with respiratory tract infection (pneumonia, acute exacerbations of chronic obstructive pulmonary disease [AECOPD], acute bronchitis, asthma, upper respiratory infection, viral syndrome, respiratory failure, and congestive heart failure [CHF] with signs of infection) were recruited during winters of 20082009 and 20092010. Patients were screened within 24 hours of admission; immunosupression, lung abscess, witnessed aspiration, or previous antibiotic use were exclusion criteria. Blood for ProCT measurement was collected in the emergency room prior to antibiotics. Subjects or legal guardians provided written informed consent, and institutional review boards approved the study.
Illness Evaluation
At enrollment, demographic, clinical, and laboratory data were collected. To provide consistency of data, 1 investigator (a clinical pulmonary specialist), after interviewing, examining each subject, and considering the radiographic findings, assigned a primary and secondary clinical admitting diagnosis: pneumonia, AECOPD, asthma exacerbation, CHF, acute bronchitis, viral syndrome or influenza, other respiratory illness, or non‐respiratory illness. These diagnoses were used for the analysis but were not available to treating physicians. Discharge diagnoses, based on International Classification of Diseases, Ninth Revision (ICD‐9) codes, and official results of follow‐up chest radiographs were also recorded.
Radiographic Interpretations
All official admission chest radiographic reports were reviewed by one of the investigators. Radiographic descriptions were noted with attention to the following comments: clear or no acute change from prior radiographs, infiltrates without consolidation, consolidation, edema, atelectasis, and pleural effusion. For our analysis, chest radiographs were categorized into 4 groups: (1) No acute disease (NAD) (ie, clear or no change from prior radiographs); (2) Other definitive radiographic abnormality (atelectasis, edema, or pleural effusion); (3) Indeterminate (findings consistent with pneumonia, but also other processes including atelectasis, edema, scarring but with no favored interpretation [often including the phrase pneumonia cannot be excluded]); and (4) infiltrate (ie, an infiltrate or consolidation most consistent with pneumonia). The pulmonary specialist independently categorized CXRs and provided a definitive radiographic interpretation whenever possible.
Microbiology
Blood cultures, sputum Gram stain and culture were performed by the clinical laboratory. Pneumococcal‐specific urinary antigen and serology were performed as previously described.[22]
Procalcitonin Measurement
Serum ProCT was measured using resolved amplified cryptate emission technology (Kryptor PCT, Brahms, Henningsdorf, Germany). Functional sensitivity is 0.06 ng/mL (normal serum levels are 0.033 0.003 ng/mL).[23, 24, 25] ProCT results were not available to investigators at the time of clinical or radiographic assessments.
Statistical Analysis
Categorical and continuous distributions were compared using Fisher's exact and Wilcoxon tests, respectively, and summarized by proportions and medians, and means and standard deviations. Log‐axis boxplots were used to graphically display the skewed distributions of ProCT. Stacked bar charts were used to depict discrete distributions of diagnoses by ProCT group. Receiver operating characteristic (ROC) curves relating ProCT to chest radiographs were computed, and sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) were calculated for ProCT thresholds. Logistic regression was used to estimate odds ratios (OR), and associated P values and confidence intervals, to quantify associations between infiltrate/non‐infiltrate and dichotomized or log‐transformed ProCT. SAS 9.2 (SAS Institute, Inc, Cary, NC) was used for analyses, using 2‐sided 0.05 level tests.
RESULTS
Study Population and Clinical Diagnoses
During 2 winters, 532 subjects admitted for 556 respiratory illnesses were recruited. Two did not have chest radiographs, 16 illnesses were non‐pulmonary, and 10 did not have admission ProCT levels, leaving 528 illnesses for analysis (Table 1). Subjects averaged 65 years of age, predominately lived at home, and a high percentage had underlying diseases. The leading primary admission diagnoses assigned by the pulmonologist were pneumonia and AECOPD at 31% and 27%, respectively. Of the 163 illnesses assigned a primary or secondary admitting diagnosis of pneumonia, infiltrates were identified on the admission radiograph by the pulmonologist in 156 (96%).
| Illnesses N = 528 (%) | |
|---|---|
| |
| Age (mean SD) | 65 17 |
| Female | 278 (53) |
| Race | |
| White | 409 (77) |
| African American | 119 (23) |
| Hispanic | 53 (10) |
| Residence | |
| Community | 488 (92) |
| Assisted living | 27 (5) |
| Nursing home | 13 (2) |
| Medical conditions | |
| Diabetes | 188 (36) |
| CHF | 153 (29) |
| COPD | 216 (41) |
| Chronic renal failure | 21 (4) |
| Past or active smoker | 395 (75) |
| Current influenza vaccine | 371 (70) |
| Prior pneumococcal vaccine | 432 (82) |
| Oral steroids | 69 (13) |
| Inhaled steroids | 201 (38) |
| Home oxygen | 129 (24) |
| Symptoms, No. (%) | |
| Upper respiratory | 350 (66) |
| Cough | 489 (93) |
| Sputum production | 390 (74) |
| Dyspnea | 496 (94) |
| Wheezing | 345 (65) |
| Constitutional | 271 (51) |
| Feverish | 293 (55) |
| Rigors | 118 (22) |
| Physical exam | |
| Wheezing | 279 (53) |
| Rales | 211 (40) |
| Rhonchi | 144 (27) |
| Temperature (mean SD) | 37.2 1.0 |
| Respiratory rate (mean SD) | 25 7 |
| Laboratory data | |
| Oxygen saturation (mean SD) | 92.3 5.5 |
| WBC 103/ml (mean SD) | 9.4 4.8 |
| Clinical diagnosis | |
| Pneumonia | 163 (31) |
| Asthma | 76 (14) |
| CHF | 45 (9) |
| Bronchitis | 83 (16) |
| AECOPD | 142 (27) |
| Other pulmonary | 19 (4) |
| Therapy | |
| Antibacterials | 463 (88) |
| Antivirals | 38 (7) |
| Outcome | |
| Intensive care | 43 (8) |
| Respiratory failure | 29 (6) |
| Death | 6 (1) |
| Length of stay (mean SD) | 5.9 18.7 |
Radiographic Classifications
Based on radiology reports, 213 (40%) were classified as NAD, 76 (14%) as other definitive findings, 75 (14%) as infiltrates, and 164 (31%) as indeterminate. The pulmonologist concurred with the radiology report for most NAD (199/213) and infiltrate (67/75) classifications, but only 4 as indeterminate, assigning approximately half the radiologist's indeterminate CXRs to infiltrate and one‐quarter each to NAD and other categories.
Relationship of ProCT With Clinical and Radiographic Features
The relationship between clinical admitting diagnosis and ProCT is shown in Figure 1A. Subjects with pneumonia had a median ProCT of 0.27 ng/ml (interquartile range [IQR] 1.3), significantly greater than subjects with AECOPD, asthma, acute bronchitis, and viral/emnfluenza. Subjects assigned to the other diagnoses category (ie, skin or urinary infections, empyema) generally had higher ProCT values (median 0.70 ng/ml, IQR 4.6).

The relationship between the radiologist's classification and ProCT is shown in Figure 1B. Median ProCT levels were significantly lower in subjects with radiographs classified as NAD compared to those showing infiltrates (P < 0.0001). Those classified as indeterminate in radiology reports had a median ProCT value (0.13 ng/ml) midway between those with NAD (0.08 ng/ml) and infiltrates (0.21 ng/ml). Similarly, illnesses with radiographs classified by the pulmonologist as infiltrates also had significantly higher median ProCT levels (0.33 ng/ml, IQR 1.33) than those classified as NAD (0.08 ng/ml, IQR 0.06; P < 0.0001), and other definitive findings (0.11 ng/ml, IQR 0.17). Notably, duration of symptoms prior to evaluation did not affect the relationship between ProCT level and radiographic classification (data not shown). All subjects with infiltrates according to the radiologist's report received early antibiotics, as did 97% and 80% with indeterminate radiographs and NAD, respectively.
Predictive Value of ProCT for Interpreting Indeterminate Radiographs
We were specifically interested to learn if ProCT could help clinicians interpret the large number of indeterminate radiographs. As a first step, we evaluated the diagnostic accuracy of ProCT for radiographic infiltrates by calculating the ROC curve (Figure 2A), using cases in which radiologist and pulmonologist concurred on the classification as infiltrate (n = 67) or no infiltrate (ie, NAD or other definitive finding; n = 273). For these cases, the ROC had an area under curve (AUC) of 0.80 (P < 0.0001), indicating moderate predictive accuracy of ProCT for the presence of an infiltrate. The OR for an infiltrate increased steadily with higher ProCT thresholds, quadrupling from 3.9 to 15.6 as the threshold increased from 0.05 to 0.50 ng/ml (Table 2). Using the commonly defined 0.25 ng/ml ProCT threshold for which antibiotics have been recommended for respiratory infections, the PPV for an infiltrate was 61% and the NPV 89%.

| ProCT Threshold (ng/ml) | Sensitivity (%) | Specificity (%) | OR | P Value |
|---|---|---|---|---|
| ||||
| 0.05 | 94 | 20 | 3.9 | 0.012 |
| 0.07 | 90 | 42 | 6.1 | <0.0001 |
| 0.10 | 79 | 65 | 7.1 | <0.0001 |
| 0.12 | 73 | 72 | 7.1 | <0.0001 |
| 0.15* | 67 | 82 | 9.1 | <0.0001 |
| 0.20 | 58 | 88 | 10.5 | <0.0001 |
| 0.25 | 52 | 92 | 12.5 | <0.0001 |
| 0.50 | 42 | 96 | 15.6 | <0.0001 |
Next, we analyzed the 164 illnesses with indeterminate radiographs. Of these, the pulmonologist classified 79 as infiltrate (median ProCT 0.29 ng/ml), 40 as NAD (median ProCT 0.08 ng/ml), 41 as other definitive findings (median ProCT 0.10 ng/ml), and 4 as indeterminate. The admitting diagnosis and corresponding median ProCT for these subjects was pneumonia in 78 (0.28 ng/ml), AECOPD in 26 (0.09 ng/ml), acute bronchitis in 30 (0.09 ng/ml), asthma in 11 (0.08 ng/ml), and CHF in 14 (0.10 ng/ml). The ROC AUC for these cases (excluding 4 considered indeterminate by the pulmonologist) was slightly lower (0.72, P < 0.0001) than the prior analysis (Figure 2B), but ProCT retained moderate predictive value for the presence of infiltrates as diagnosed by the pulmonologist on indeterminate radiographs (Table 3).
| ProCT Threshold | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | OR | P Value | Predicted Probability (%) |
|---|---|---|---|---|---|---|---|
| |||||||
| 0.05 | 95 | 7 | 50 | 60 | 1.5 | 0.54 | 32 |
| 0.07 | 86 | 27 | 54 | 67 | 2.3 | 0.04 | 35 |
| 0.10 | 80 | 59 | 66 | 75 | 5.7 | <0.0001 | 40 |
| 0.12* | 76 | 67 | 69 | 74 | 6.3 | <0.0001 | 42 |
| 0.15 | 67 | 74 | 72 | 70 | 5.8 | <0.0001 | 45 |
| 0.20 | 63 | 78 | 74 | 69 | 6.0 | <0.0001 | 49 |
| 0.25 | 53 | 80 | 72 | 64 | 4.6 | <0.0001 | 52 |
| 0.50 | 37 | 88 | 74 | 59 | 4.1 | 0.0006 | 60 |
Since clinical determinations by a single pulmonologist may be subjective, we sought to objectively assess the accuracy of assigned admission diagnoses by analyzing additional data, including bacterial tests, follow‐up radiographs, and final discharge diagnoses. As a surrogate of invasive bacterial infection, we used identification of S. pneumoniae as the outcome, since multiple complementary diagnostic assays were used, thus minimizing the uncertainty of sputum cultures. Overall, we identified 58 pneumococcal infections (Table 4). The pulmonologist's admitting diagnosis of pneumonia captured a higher proportion (52% for any test positive) of S. pneumoniae cases than identification of a radiographic infiltrate by the radiologist (26%), and equivalent to the pulmonologist's radiographic reading (57%) or elevated ProCT alone (57%). There was an absolute 14% increase (27% relative increase) in the detection of S. pneumoniae infections by addition of ProCT to the pulmonologist's diagnosis. A similar analysis limited to the 164 subjects with indeterminate radiographs found that 13 of 19 (68%) of S. pneumoniae diagnoses had been assigned a clinical diagnosis of pneumonia by the pulmonologist.
| S. pneumonia Confirmed by Diagnostic Assay No. (%) | |||||
|---|---|---|---|---|---|
| Criteria | Blood Culture + (n = 7) | Urine Ag + (n = 27) | Serology + (n = 28) | Sputum + (n = 20) | Any Test + (n = 58) |
| |||||
| Radiologist CXR read as infiltrate | 3 (43) | 9 (33) | 10 (36) | 3 (15) | 15 (26) |
| Pulmonologist CXR read as infiltrate | 6 (86) | 17 (62) | 18 (64) | 10 (50) | 33 (57) |
| Pulmonologist clinical diagnosis of pneumonia | 6 (86) | 16 (60) | 16 (57) | 8 (40) | 30 (52) |
| Pulmonologist clinical diagnosis of AECOPD | 0 (0) | 4 (15) | 5 (18) | 5 (25) | 8 (14) |
| Procalcitonin 0.25 ng/ml | 6 (86) | 18 (67) | 17 (61) | 9 (45) | 33 (57) |
| Pulmonologist diagnosis of pneumonia and/or ProCT 0.25 ng/ml | 7 (100) | 19 (70) | 20 (71) | 10 (50) | 38 (66) |
We also determined how often subsequent radiographs showed evolution to a definitive infiltrate in indeterminate cases that the pulmonologist had classified as either NAD or other finding. Only 3/20 with repeat films showed a definitive infiltrate, none of whom had evidence of bacterial infection, and all 3 had ProCT <0.25 ng/ml. In contrast, 10 of 21 subjects with indeterminate CXRs, that the pulmonologist had classified as an infiltrate, developed definitive infiltrates on follow‐up studies (P = 0.04), and 8 remained indeterminate.
Finally, 93% of those assigned an admitting pneumonia diagnosis by the pulmonologist had primary (71%) or secondary (22%) discharge diagnosis of pneumonia. Of the additional 28 subjects with a discharge diagnosis of pneumonia, for whom the pulmonologist assigned non‐pneumonia admitting diagnoses, none had positive blood cultures or evidence of S. pneumoniae infection, and only one had a follow‐up radiograph showing a definitive infiltrate.
DISCUSSION
Pneumonia can be a serious illness and, if left untreated, result in death. Hence, infectious disease and pulmonary societies advocate timely administration of antibiotics. Unfortunately, clinical diagnosis of pneumonia is difficult, as clinical and radiographic features are often ambiguous. These factors result in liberal use of broad‐spectrum antibiotics, often in patients with viral infection, in whom antibiotic side effects outweigh benefit.[26] Although pneumonia guidelines recommend early antibiotic therapy for patients with infiltrates on CXR, we found that early administration of antibiotics for possible pneumonia is nearly universal.[2, 27] Thus, an objective laboratory test indicative of pneumonia would be a useful adjunct when making treatment decisions.
In an attempt to curb excessive antibiotic use, European investigators have used ProCT to guide therapy for respiratory tract infection, including pneumonia, without increased adverse outcomes.[17, 19, 28, 29] Antibiotics are recommended when ProCT levels are 0.25 ng/ml, rather than based on radiographic or microbiological results. However, ProCT‐guided antibiotic management has not been adopted in the United States, and radiographic findings are a major determinant for diagnosis of pneumonia and antibiotic administration.
Thus, we were interested in correlating ProCT levels with the presence of radiographic infiltrates, and especially how ProCT could assist in interpreting the vexing problem of indeterminate CXRs. We found a direct relationship between an increased ProCT level and definitive infiltrates on CXR, similar to results reported by others.[18, 20, 21] However, these studies did not address illnesses with indeterminate radiographs, which accounted for approximately one‐third of the radiologist's readings in our study. Our situation is likely not unique. In a post hoc analysis of CXRs from subjects with pneumonia in the PORT study, independent radiologists interpreted a similar proportion of CXRs as either no acute disease or possible infiltrates.[30] Since indeterminate readings include the possibility of an infiltrate, it is not surprising that 97% of our subjects received early antibiotic treatment.
Given current pneumonia treatment guidelines, ProCT testing for those with definitive infiltrates would not add to patient management, as all should receive early antibiotics. However, ProCT measurement could have significant impact on management in those with indeterminate films. We found ProCT was moderately predictive for infiltrates when utilizing the pulmonologist's interpretation of indeterminate CXRs. This is concordant with a recent study reporting a similar ROC (0.72) for ProCT for diagnosing pneumonia in patients with dyspnea, and more predictive of pneumonia than any clinical variable.[31]
The major limitation of our study is the lack of a gold standard for diagnosis of pneumonia. We chose to use a pulmonary specialist to provide admission diagnoses, and independently interpret radiographs in the context of clinical findings. Although imperfect, we feel this approach best reflects good medical practice by an experienced clinician evaluating the CXR in context of a patient's history and physical exam. Importantly, analysis of the concurrent microbiology data, follow‐up radiographs, and discharge diagnoses supports the accuracy of the admitting diagnoses of pneumonia and radiographic interpretations by the pulmonologist. Secondly, our findings may not be applicable to immunocompromised or previously treated patients.
How should a busy physician respond to a patient with dyspnea and fever when faced with an indeterminate radiographic report, since no method for the diagnosis of pneumonia is infallible?[8, 32] Clearly, a pulmonary consultation on all such patients is impractical. Our data confirms previous results that high ProCT values are correlated with an infiltrate on CXR and low values have a NPV of nearly 90%. Furthermore, low ProCT values predict low mortality across all levels of standard severity scoring indexes.[33] Thus, a low ProCT may help clinicians feel more confident to delay or stop antibiotics in a patient in whom clinical suspicion for bacterial pneumonia is low, yet the radiographic report states pneumonia cannot be ruled out.[34] Such strategies may assist in reducing unnecessary antibiotic use in the United States.[35]
Disclosures
Funding for this study was provided by NIAID‐1R01AI079446‐01. This work was presented in part at the Infectious Diseases Society of America, Boston, MA, October 2011. The authors report no conflicts of interest.
Pneumonia is a common reason for hospitalization and major rationale for administration of antibiotics in the United States.[1] Management guidelines for patients hospitalized with community‐acquired pneumonia recommend early antibiotic therapy. Quality measures adopted by the Centers for Medicare and Medicaid Services include antibiotic administration within 6 hours of presentation, based on a relationship between early administration and improved survival.[2, 3, 4, 5] However, this imperative has been associated with an increase in incorrect diagnoses of pneumonia.[6, 7] While pneumonia diagnosis would seem straightforward, clinical findings frequently do not differentiate pneumonia from other respiratory illnesses. Thus, an infiltrate on chest radiographs (CXR) is commonly used to assign an admitting diagnosis of pneumonia.[8, 9, 10] However, radiographic reports are often inconclusive and frequently fail to discriminate pneumonic infiltrates from atelectasis, edema, small pleural effusions, or chronic abnormalities. In cases of uncertainty, physicians invariably initiate antibiotics, even when the illness is more consistent with viral bronchitis or asthma, resulting in unnecessary antibiotic use.[6] Previously considered relatively harmless, antibiotic complications can be lethal, and excessive use promotes antimicrobial resistance.[11, 12]
Procalcitonin (ProCT), a calcitonin precursor normally produced in the thyroid and lungs, is secreted by cells throughout the body in response to bacterial infections.[13, 14] Elevated ProCT is used to screen patients with suspected bacterial infection and predict mortality in critically ill patients.[15, 16] Recently, European investigators have used ProCT to guide more selective antibiotic use in patients with symptoms of lower respiratory tract infection (LRTI), including pneumonia.[17, 18, 19] However, few reports describe the relationship of ProCT with radiographic features in hospitalized patients with LRTI, and none focus on patients with indeterminate radiographic readings.[18, 20, 21]
We sought to determine if elevated serum ProCT in adults hospitalized with LRTI symptoms correlates with the clinical diagnosis of pneumonia and definitive infiltrates on CXRs. We specifically assessed ProCT as a diagnostic marker of pneumonia to augment clinical judgment in patients when radiographic findings are indeterminate.
METHODS
Subjects
The study was performed in a 520‐bed, general medicalsurgical hospital, using subjects participating in a study of the relationship between biomarkers and microbiologic diagnoses (to be described in a separate publication). Adults 21 years of age admitted with a diagnosis compatible with respiratory tract infection (pneumonia, acute exacerbations of chronic obstructive pulmonary disease [AECOPD], acute bronchitis, asthma, upper respiratory infection, viral syndrome, respiratory failure, and congestive heart failure [CHF] with signs of infection) were recruited during winters of 20082009 and 20092010. Patients were screened within 24 hours of admission; immunosupression, lung abscess, witnessed aspiration, or previous antibiotic use were exclusion criteria. Blood for ProCT measurement was collected in the emergency room prior to antibiotics. Subjects or legal guardians provided written informed consent, and institutional review boards approved the study.
Illness Evaluation
At enrollment, demographic, clinical, and laboratory data were collected. To provide consistency of data, 1 investigator (a clinical pulmonary specialist), after interviewing, examining each subject, and considering the radiographic findings, assigned a primary and secondary clinical admitting diagnosis: pneumonia, AECOPD, asthma exacerbation, CHF, acute bronchitis, viral syndrome or influenza, other respiratory illness, or non‐respiratory illness. These diagnoses were used for the analysis but were not available to treating physicians. Discharge diagnoses, based on International Classification of Diseases, Ninth Revision (ICD‐9) codes, and official results of follow‐up chest radiographs were also recorded.
Radiographic Interpretations
All official admission chest radiographic reports were reviewed by one of the investigators. Radiographic descriptions were noted with attention to the following comments: clear or no acute change from prior radiographs, infiltrates without consolidation, consolidation, edema, atelectasis, and pleural effusion. For our analysis, chest radiographs were categorized into 4 groups: (1) No acute disease (NAD) (ie, clear or no change from prior radiographs); (2) Other definitive radiographic abnormality (atelectasis, edema, or pleural effusion); (3) Indeterminate (findings consistent with pneumonia, but also other processes including atelectasis, edema, scarring but with no favored interpretation [often including the phrase pneumonia cannot be excluded]); and (4) infiltrate (ie, an infiltrate or consolidation most consistent with pneumonia). The pulmonary specialist independently categorized CXRs and provided a definitive radiographic interpretation whenever possible.
Microbiology
Blood cultures, sputum Gram stain and culture were performed by the clinical laboratory. Pneumococcal‐specific urinary antigen and serology were performed as previously described.[22]
Procalcitonin Measurement
Serum ProCT was measured using resolved amplified cryptate emission technology (Kryptor PCT, Brahms, Henningsdorf, Germany). Functional sensitivity is 0.06 ng/mL (normal serum levels are 0.033 0.003 ng/mL).[23, 24, 25] ProCT results were not available to investigators at the time of clinical or radiographic assessments.
Statistical Analysis
Categorical and continuous distributions were compared using Fisher's exact and Wilcoxon tests, respectively, and summarized by proportions and medians, and means and standard deviations. Log‐axis boxplots were used to graphically display the skewed distributions of ProCT. Stacked bar charts were used to depict discrete distributions of diagnoses by ProCT group. Receiver operating characteristic (ROC) curves relating ProCT to chest radiographs were computed, and sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) were calculated for ProCT thresholds. Logistic regression was used to estimate odds ratios (OR), and associated P values and confidence intervals, to quantify associations between infiltrate/non‐infiltrate and dichotomized or log‐transformed ProCT. SAS 9.2 (SAS Institute, Inc, Cary, NC) was used for analyses, using 2‐sided 0.05 level tests.
RESULTS
Study Population and Clinical Diagnoses
During 2 winters, 532 subjects admitted for 556 respiratory illnesses were recruited. Two did not have chest radiographs, 16 illnesses were non‐pulmonary, and 10 did not have admission ProCT levels, leaving 528 illnesses for analysis (Table 1). Subjects averaged 65 years of age, predominately lived at home, and a high percentage had underlying diseases. The leading primary admission diagnoses assigned by the pulmonologist were pneumonia and AECOPD at 31% and 27%, respectively. Of the 163 illnesses assigned a primary or secondary admitting diagnosis of pneumonia, infiltrates were identified on the admission radiograph by the pulmonologist in 156 (96%).
| Illnesses N = 528 (%) | |
|---|---|
| |
| Age (mean SD) | 65 17 |
| Female | 278 (53) |
| Race | |
| White | 409 (77) |
| African American | 119 (23) |
| Hispanic | 53 (10) |
| Residence | |
| Community | 488 (92) |
| Assisted living | 27 (5) |
| Nursing home | 13 (2) |
| Medical conditions | |
| Diabetes | 188 (36) |
| CHF | 153 (29) |
| COPD | 216 (41) |
| Chronic renal failure | 21 (4) |
| Past or active smoker | 395 (75) |
| Current influenza vaccine | 371 (70) |
| Prior pneumococcal vaccine | 432 (82) |
| Oral steroids | 69 (13) |
| Inhaled steroids | 201 (38) |
| Home oxygen | 129 (24) |
| Symptoms, No. (%) | |
| Upper respiratory | 350 (66) |
| Cough | 489 (93) |
| Sputum production | 390 (74) |
| Dyspnea | 496 (94) |
| Wheezing | 345 (65) |
| Constitutional | 271 (51) |
| Feverish | 293 (55) |
| Rigors | 118 (22) |
| Physical exam | |
| Wheezing | 279 (53) |
| Rales | 211 (40) |
| Rhonchi | 144 (27) |
| Temperature (mean SD) | 37.2 1.0 |
| Respiratory rate (mean SD) | 25 7 |
| Laboratory data | |
| Oxygen saturation (mean SD) | 92.3 5.5 |
| WBC 103/ml (mean SD) | 9.4 4.8 |
| Clinical diagnosis | |
| Pneumonia | 163 (31) |
| Asthma | 76 (14) |
| CHF | 45 (9) |
| Bronchitis | 83 (16) |
| AECOPD | 142 (27) |
| Other pulmonary | 19 (4) |
| Therapy | |
| Antibacterials | 463 (88) |
| Antivirals | 38 (7) |
| Outcome | |
| Intensive care | 43 (8) |
| Respiratory failure | 29 (6) |
| Death | 6 (1) |
| Length of stay (mean SD) | 5.9 18.7 |
Radiographic Classifications
Based on radiology reports, 213 (40%) were classified as NAD, 76 (14%) as other definitive findings, 75 (14%) as infiltrates, and 164 (31%) as indeterminate. The pulmonologist concurred with the radiology report for most NAD (199/213) and infiltrate (67/75) classifications, but only 4 as indeterminate, assigning approximately half the radiologist's indeterminate CXRs to infiltrate and one‐quarter each to NAD and other categories.
Relationship of ProCT With Clinical and Radiographic Features
The relationship between clinical admitting diagnosis and ProCT is shown in Figure 1A. Subjects with pneumonia had a median ProCT of 0.27 ng/ml (interquartile range [IQR] 1.3), significantly greater than subjects with AECOPD, asthma, acute bronchitis, and viral/emnfluenza. Subjects assigned to the other diagnoses category (ie, skin or urinary infections, empyema) generally had higher ProCT values (median 0.70 ng/ml, IQR 4.6).

The relationship between the radiologist's classification and ProCT is shown in Figure 1B. Median ProCT levels were significantly lower in subjects with radiographs classified as NAD compared to those showing infiltrates (P < 0.0001). Those classified as indeterminate in radiology reports had a median ProCT value (0.13 ng/ml) midway between those with NAD (0.08 ng/ml) and infiltrates (0.21 ng/ml). Similarly, illnesses with radiographs classified by the pulmonologist as infiltrates also had significantly higher median ProCT levels (0.33 ng/ml, IQR 1.33) than those classified as NAD (0.08 ng/ml, IQR 0.06; P < 0.0001), and other definitive findings (0.11 ng/ml, IQR 0.17). Notably, duration of symptoms prior to evaluation did not affect the relationship between ProCT level and radiographic classification (data not shown). All subjects with infiltrates according to the radiologist's report received early antibiotics, as did 97% and 80% with indeterminate radiographs and NAD, respectively.
Predictive Value of ProCT for Interpreting Indeterminate Radiographs
We were specifically interested to learn if ProCT could help clinicians interpret the large number of indeterminate radiographs. As a first step, we evaluated the diagnostic accuracy of ProCT for radiographic infiltrates by calculating the ROC curve (Figure 2A), using cases in which radiologist and pulmonologist concurred on the classification as infiltrate (n = 67) or no infiltrate (ie, NAD or other definitive finding; n = 273). For these cases, the ROC had an area under curve (AUC) of 0.80 (P < 0.0001), indicating moderate predictive accuracy of ProCT for the presence of an infiltrate. The OR for an infiltrate increased steadily with higher ProCT thresholds, quadrupling from 3.9 to 15.6 as the threshold increased from 0.05 to 0.50 ng/ml (Table 2). Using the commonly defined 0.25 ng/ml ProCT threshold for which antibiotics have been recommended for respiratory infections, the PPV for an infiltrate was 61% and the NPV 89%.

| ProCT Threshold (ng/ml) | Sensitivity (%) | Specificity (%) | OR | P Value |
|---|---|---|---|---|
| ||||
| 0.05 | 94 | 20 | 3.9 | 0.012 |
| 0.07 | 90 | 42 | 6.1 | <0.0001 |
| 0.10 | 79 | 65 | 7.1 | <0.0001 |
| 0.12 | 73 | 72 | 7.1 | <0.0001 |
| 0.15* | 67 | 82 | 9.1 | <0.0001 |
| 0.20 | 58 | 88 | 10.5 | <0.0001 |
| 0.25 | 52 | 92 | 12.5 | <0.0001 |
| 0.50 | 42 | 96 | 15.6 | <0.0001 |
Next, we analyzed the 164 illnesses with indeterminate radiographs. Of these, the pulmonologist classified 79 as infiltrate (median ProCT 0.29 ng/ml), 40 as NAD (median ProCT 0.08 ng/ml), 41 as other definitive findings (median ProCT 0.10 ng/ml), and 4 as indeterminate. The admitting diagnosis and corresponding median ProCT for these subjects was pneumonia in 78 (0.28 ng/ml), AECOPD in 26 (0.09 ng/ml), acute bronchitis in 30 (0.09 ng/ml), asthma in 11 (0.08 ng/ml), and CHF in 14 (0.10 ng/ml). The ROC AUC for these cases (excluding 4 considered indeterminate by the pulmonologist) was slightly lower (0.72, P < 0.0001) than the prior analysis (Figure 2B), but ProCT retained moderate predictive value for the presence of infiltrates as diagnosed by the pulmonologist on indeterminate radiographs (Table 3).
| ProCT Threshold | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | OR | P Value | Predicted Probability (%) |
|---|---|---|---|---|---|---|---|
| |||||||
| 0.05 | 95 | 7 | 50 | 60 | 1.5 | 0.54 | 32 |
| 0.07 | 86 | 27 | 54 | 67 | 2.3 | 0.04 | 35 |
| 0.10 | 80 | 59 | 66 | 75 | 5.7 | <0.0001 | 40 |
| 0.12* | 76 | 67 | 69 | 74 | 6.3 | <0.0001 | 42 |
| 0.15 | 67 | 74 | 72 | 70 | 5.8 | <0.0001 | 45 |
| 0.20 | 63 | 78 | 74 | 69 | 6.0 | <0.0001 | 49 |
| 0.25 | 53 | 80 | 72 | 64 | 4.6 | <0.0001 | 52 |
| 0.50 | 37 | 88 | 74 | 59 | 4.1 | 0.0006 | 60 |
Since clinical determinations by a single pulmonologist may be subjective, we sought to objectively assess the accuracy of assigned admission diagnoses by analyzing additional data, including bacterial tests, follow‐up radiographs, and final discharge diagnoses. As a surrogate of invasive bacterial infection, we used identification of S. pneumoniae as the outcome, since multiple complementary diagnostic assays were used, thus minimizing the uncertainty of sputum cultures. Overall, we identified 58 pneumococcal infections (Table 4). The pulmonologist's admitting diagnosis of pneumonia captured a higher proportion (52% for any test positive) of S. pneumoniae cases than identification of a radiographic infiltrate by the radiologist (26%), and equivalent to the pulmonologist's radiographic reading (57%) or elevated ProCT alone (57%). There was an absolute 14% increase (27% relative increase) in the detection of S. pneumoniae infections by addition of ProCT to the pulmonologist's diagnosis. A similar analysis limited to the 164 subjects with indeterminate radiographs found that 13 of 19 (68%) of S. pneumoniae diagnoses had been assigned a clinical diagnosis of pneumonia by the pulmonologist.
| S. pneumonia Confirmed by Diagnostic Assay No. (%) | |||||
|---|---|---|---|---|---|
| Criteria | Blood Culture + (n = 7) | Urine Ag + (n = 27) | Serology + (n = 28) | Sputum + (n = 20) | Any Test + (n = 58) |
| |||||
| Radiologist CXR read as infiltrate | 3 (43) | 9 (33) | 10 (36) | 3 (15) | 15 (26) |
| Pulmonologist CXR read as infiltrate | 6 (86) | 17 (62) | 18 (64) | 10 (50) | 33 (57) |
| Pulmonologist clinical diagnosis of pneumonia | 6 (86) | 16 (60) | 16 (57) | 8 (40) | 30 (52) |
| Pulmonologist clinical diagnosis of AECOPD | 0 (0) | 4 (15) | 5 (18) | 5 (25) | 8 (14) |
| Procalcitonin 0.25 ng/ml | 6 (86) | 18 (67) | 17 (61) | 9 (45) | 33 (57) |
| Pulmonologist diagnosis of pneumonia and/or ProCT 0.25 ng/ml | 7 (100) | 19 (70) | 20 (71) | 10 (50) | 38 (66) |
We also determined how often subsequent radiographs showed evolution to a definitive infiltrate in indeterminate cases that the pulmonologist had classified as either NAD or other finding. Only 3/20 with repeat films showed a definitive infiltrate, none of whom had evidence of bacterial infection, and all 3 had ProCT <0.25 ng/ml. In contrast, 10 of 21 subjects with indeterminate CXRs, that the pulmonologist had classified as an infiltrate, developed definitive infiltrates on follow‐up studies (P = 0.04), and 8 remained indeterminate.
Finally, 93% of those assigned an admitting pneumonia diagnosis by the pulmonologist had primary (71%) or secondary (22%) discharge diagnosis of pneumonia. Of the additional 28 subjects with a discharge diagnosis of pneumonia, for whom the pulmonologist assigned non‐pneumonia admitting diagnoses, none had positive blood cultures or evidence of S. pneumoniae infection, and only one had a follow‐up radiograph showing a definitive infiltrate.
DISCUSSION
Pneumonia can be a serious illness and, if left untreated, result in death. Hence, infectious disease and pulmonary societies advocate timely administration of antibiotics. Unfortunately, clinical diagnosis of pneumonia is difficult, as clinical and radiographic features are often ambiguous. These factors result in liberal use of broad‐spectrum antibiotics, often in patients with viral infection, in whom antibiotic side effects outweigh benefit.[26] Although pneumonia guidelines recommend early antibiotic therapy for patients with infiltrates on CXR, we found that early administration of antibiotics for possible pneumonia is nearly universal.[2, 27] Thus, an objective laboratory test indicative of pneumonia would be a useful adjunct when making treatment decisions.
In an attempt to curb excessive antibiotic use, European investigators have used ProCT to guide therapy for respiratory tract infection, including pneumonia, without increased adverse outcomes.[17, 19, 28, 29] Antibiotics are recommended when ProCT levels are 0.25 ng/ml, rather than based on radiographic or microbiological results. However, ProCT‐guided antibiotic management has not been adopted in the United States, and radiographic findings are a major determinant for diagnosis of pneumonia and antibiotic administration.
Thus, we were interested in correlating ProCT levels with the presence of radiographic infiltrates, and especially how ProCT could assist in interpreting the vexing problem of indeterminate CXRs. We found a direct relationship between an increased ProCT level and definitive infiltrates on CXR, similar to results reported by others.[18, 20, 21] However, these studies did not address illnesses with indeterminate radiographs, which accounted for approximately one‐third of the radiologist's readings in our study. Our situation is likely not unique. In a post hoc analysis of CXRs from subjects with pneumonia in the PORT study, independent radiologists interpreted a similar proportion of CXRs as either no acute disease or possible infiltrates.[30] Since indeterminate readings include the possibility of an infiltrate, it is not surprising that 97% of our subjects received early antibiotic treatment.
Given current pneumonia treatment guidelines, ProCT testing for those with definitive infiltrates would not add to patient management, as all should receive early antibiotics. However, ProCT measurement could have significant impact on management in those with indeterminate films. We found ProCT was moderately predictive for infiltrates when utilizing the pulmonologist's interpretation of indeterminate CXRs. This is concordant with a recent study reporting a similar ROC (0.72) for ProCT for diagnosing pneumonia in patients with dyspnea, and more predictive of pneumonia than any clinical variable.[31]
The major limitation of our study is the lack of a gold standard for diagnosis of pneumonia. We chose to use a pulmonary specialist to provide admission diagnoses, and independently interpret radiographs in the context of clinical findings. Although imperfect, we feel this approach best reflects good medical practice by an experienced clinician evaluating the CXR in context of a patient's history and physical exam. Importantly, analysis of the concurrent microbiology data, follow‐up radiographs, and discharge diagnoses supports the accuracy of the admitting diagnoses of pneumonia and radiographic interpretations by the pulmonologist. Secondly, our findings may not be applicable to immunocompromised or previously treated patients.
How should a busy physician respond to a patient with dyspnea and fever when faced with an indeterminate radiographic report, since no method for the diagnosis of pneumonia is infallible?[8, 32] Clearly, a pulmonary consultation on all such patients is impractical. Our data confirms previous results that high ProCT values are correlated with an infiltrate on CXR and low values have a NPV of nearly 90%. Furthermore, low ProCT values predict low mortality across all levels of standard severity scoring indexes.[33] Thus, a low ProCT may help clinicians feel more confident to delay or stop antibiotics in a patient in whom clinical suspicion for bacterial pneumonia is low, yet the radiographic report states pneumonia cannot be ruled out.[34] Such strategies may assist in reducing unnecessary antibiotic use in the United States.[35]
Disclosures
Funding for this study was provided by NIAID‐1R01AI079446‐01. This work was presented in part at the Infectious Diseases Society of America, Boston, MA, October 2011. The authors report no conflicts of interest.
- , , , et al. The burden of community‐acquired pneumonia in seniors: results of a population‐based study. Clin Infect Dis. 2004;39:1642–1650.
- , , , et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community‐acquired pneumonia in adults. Clin Infect Dis. 2007;44(suppl 2):S27–S72.
- Centers for Medicare and Medicaid Services. The Medicare national pneumonia quality improvement project. Available at: http://www.cms.gov/HospitalQualityInits/. Accessed September 8, 2011.
- , , , , . Timing of antibiotic administration and outcomes for Medicare patients hospitalized with community‐acquired pneumonia. Arch Intern Med. 2004;164:637–644.
- , , , et al. Quality of care, process, and outcomes in elderly patients with pneumonia. JAMA. 1997;278:2080–2084.
- , , , . Misdiagnosis of community‐acquired pneumonia and inappropriate utilization of antibiotics: side effects of the 4‐h antibiotic administration rule. Chest. 2007;131:1865–1869.
- , , . Antibiotic timing and errors in diagnosing pneumonia. Arch Intern Med. 2008;168:351–356.
- , , , . Patients admitted to hospital with suspected pneumonia and normal chest radiographs: epidemiology, microbiology, and outcomes. Am J Med. 2004;117:305–311.
- , , , , . Reliability of radiographic findings and the relation to etiologic agents in community‐acquired pneumonia. Respir Med. 2006;100:926–932.
- , , , . Admission chest radiograph lacks sensitivity in the diagnosis of community‐acquired pneumonia. Am J Med Sci. 2009;337:236–240.
- , . Strategies for reduction in duration of antibiotic use in hospitalized patients. Clin Infect Dis. 2011;52:1232–1240.
- , , , et al. The epidemic of antibiotic‐resistant infections: a call to action for the medical community from the Infectious Diseases Society of America. Clin Infect Dis. 2008;46:155–164.
- , , , , . Clinical review 167: procalcitonin and the calcitonin gene family of peptides in inflammation, infection, and sepsis: a journey from calcitonin back to its precursors. J Clin Endocrinol Metab. 2004;89:1512–1525.
- , , . Procalcitonin assay in systemic inflammation, infection, and sepsis: clinical utility and limitations. Crit Care Med. 2008;36:941–952.
- , , . Procalcitonin in sepsis and systemic inflammation: a harmful biomarker and a therapeutic target. Br J Pharmacol. 2010;159:253–264.
- , , , , . Pneumonitis‐associated hyperprocalcitoninemia. Am J Med Sci. 1996;312:12–18.
- , , , et al. Antibiotic treatment of exacerbations of COPD: a randomized, controlled trial comparing procalcitonin‐guidance with standard therapy. Chest. 2007;131:9–19.
- , , , et al. Procalcitonin versus C‐reactive protein for predicting pneumonia in adults with lower respiratory tract infection in primary care. Br J Gen Pract. 2007;57:555–560.
- , , , et al. Effect of procalcitonin‐based guidelines vs standard guidelines on antibiotic use in lower respiratory tract infections: the ProHOSP randomized controlled trial. JAMA. 2009;302:1059–1066.
- , , , et al. Procalcitonin and C‐reactive protein in hospitalized adult patients with community‐acquired pneumonia or exacerbation of asthma or COPD. Chest. 2011;139:1410–1418.
- , , , et al. Diagnostic and prognostic accuracy of clinical and laboratory parameters in community‐acquired pneumonia. BMC Infect Dis. 2008;7:10.
- , , , et al. Utility of serum procalcitonin values in patients with acute exacerbations of chronic obstructive pulmonary disease: a cautionary note. Int J Chron Obstruct Pulmon Dis. 2112;7:127–135.
- , , . Procalcitonin and its component peptides in systemic inflammation: immunochemical characterization. J Investig Med. 1997;45:552–560.
- , , , . The future diagnostic role of procalcitonin levels: the need for improved sensitivity. Clin Infect Dis. 2003;36:823–824.
- , , , et al. Calcitonin precursor levels in human medullary thyroid carcinoma. Thyroid 2003;13:819–822.
- , , , . Emergency department visits for antibiotic‐associated adverse events. Clin Infect Dis. 2008;47:735–743.
- , , , et al. Guideline tyranny: a response to the article by Baum and Kaltsas. Clin Infect Dis. 2008;47:1117–1118.
- , , , et al. Procalcitonin guidance of antibiotic therapy in community‐acquired pneumonia: a randomized trial. Am J Respir Crit Care Med. 2006;174:84–93.
- , , , et al. Effect of procalcitonin‐guided treatment on antibiotic use and outcome in lower respiratory tract infections: cluster‐randomised, single‐blinded intervention trial. Lancet. 2004;363:600–607.
- , , , et al. Interobserver reliability of the chest radiograph in community‐acquired pneumonia. PORT investigators. Chest. 1996;110:343–350.
- , , , et al. Use of procalcitonin for the diagnosis of pneumonia in patients presenting with a chief complaint of dyspnea: results from the BACH (Biomarkers in Acute Heart Failure) trial. Eur J Heart Fail. 2012;14:278–286.
- , , , , . A multicenter analysis of the ED diagnosis of pneumonia. Am J Emerg Med. 2010;28:862–865.
- , , , et al. Procalcitonin predicts patients at low risk of death from community‐acquired pneumonia across all CRB‐65 classes. Eur Respir J. 2008;31:349–355.
- , , , , , . Antibiotic timing and diagnostic uncertainty in Medicare patients with pneumonia: is it reasonable to expect all patients to receive antibiotics within 4 hours? Chest. 2006;130:16–21.
- . Procalcitonin for triage of patients with respiratory tract symptoms: a case study in the trial design process for approval of a new diagnostic test for lower respiratory tract infection. Clin Infect Dis. 2011;52(S4):S351–S356.
- , , , et al. The burden of community‐acquired pneumonia in seniors: results of a population‐based study. Clin Infect Dis. 2004;39:1642–1650.
- , , , et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community‐acquired pneumonia in adults. Clin Infect Dis. 2007;44(suppl 2):S27–S72.
- Centers for Medicare and Medicaid Services. The Medicare national pneumonia quality improvement project. Available at: http://www.cms.gov/HospitalQualityInits/. Accessed September 8, 2011.
- , , , , . Timing of antibiotic administration and outcomes for Medicare patients hospitalized with community‐acquired pneumonia. Arch Intern Med. 2004;164:637–644.
- , , , et al. Quality of care, process, and outcomes in elderly patients with pneumonia. JAMA. 1997;278:2080–2084.
- , , , . Misdiagnosis of community‐acquired pneumonia and inappropriate utilization of antibiotics: side effects of the 4‐h antibiotic administration rule. Chest. 2007;131:1865–1869.
- , , . Antibiotic timing and errors in diagnosing pneumonia. Arch Intern Med. 2008;168:351–356.
- , , , . Patients admitted to hospital with suspected pneumonia and normal chest radiographs: epidemiology, microbiology, and outcomes. Am J Med. 2004;117:305–311.
- , , , , . Reliability of radiographic findings and the relation to etiologic agents in community‐acquired pneumonia. Respir Med. 2006;100:926–932.
- , , , . Admission chest radiograph lacks sensitivity in the diagnosis of community‐acquired pneumonia. Am J Med Sci. 2009;337:236–240.
- , . Strategies for reduction in duration of antibiotic use in hospitalized patients. Clin Infect Dis. 2011;52:1232–1240.
- , , , et al. The epidemic of antibiotic‐resistant infections: a call to action for the medical community from the Infectious Diseases Society of America. Clin Infect Dis. 2008;46:155–164.
- , , , , . Clinical review 167: procalcitonin and the calcitonin gene family of peptides in inflammation, infection, and sepsis: a journey from calcitonin back to its precursors. J Clin Endocrinol Metab. 2004;89:1512–1525.
- , , . Procalcitonin assay in systemic inflammation, infection, and sepsis: clinical utility and limitations. Crit Care Med. 2008;36:941–952.
- , , . Procalcitonin in sepsis and systemic inflammation: a harmful biomarker and a therapeutic target. Br J Pharmacol. 2010;159:253–264.
- , , , , . Pneumonitis‐associated hyperprocalcitoninemia. Am J Med Sci. 1996;312:12–18.
- , , , et al. Antibiotic treatment of exacerbations of COPD: a randomized, controlled trial comparing procalcitonin‐guidance with standard therapy. Chest. 2007;131:9–19.
- , , , et al. Procalcitonin versus C‐reactive protein for predicting pneumonia in adults with lower respiratory tract infection in primary care. Br J Gen Pract. 2007;57:555–560.
- , , , et al. Effect of procalcitonin‐based guidelines vs standard guidelines on antibiotic use in lower respiratory tract infections: the ProHOSP randomized controlled trial. JAMA. 2009;302:1059–1066.
- , , , et al. Procalcitonin and C‐reactive protein in hospitalized adult patients with community‐acquired pneumonia or exacerbation of asthma or COPD. Chest. 2011;139:1410–1418.
- , , , et al. Diagnostic and prognostic accuracy of clinical and laboratory parameters in community‐acquired pneumonia. BMC Infect Dis. 2008;7:10.
- , , , et al. Utility of serum procalcitonin values in patients with acute exacerbations of chronic obstructive pulmonary disease: a cautionary note. Int J Chron Obstruct Pulmon Dis. 2112;7:127–135.
- , , . Procalcitonin and its component peptides in systemic inflammation: immunochemical characterization. J Investig Med. 1997;45:552–560.
- , , , . The future diagnostic role of procalcitonin levels: the need for improved sensitivity. Clin Infect Dis. 2003;36:823–824.
- , , , et al. Calcitonin precursor levels in human medullary thyroid carcinoma. Thyroid 2003;13:819–822.
- , , , . Emergency department visits for antibiotic‐associated adverse events. Clin Infect Dis. 2008;47:735–743.
- , , , et al. Guideline tyranny: a response to the article by Baum and Kaltsas. Clin Infect Dis. 2008;47:1117–1118.
- , , , et al. Procalcitonin guidance of antibiotic therapy in community‐acquired pneumonia: a randomized trial. Am J Respir Crit Care Med. 2006;174:84–93.
- , , , et al. Effect of procalcitonin‐guided treatment on antibiotic use and outcome in lower respiratory tract infections: cluster‐randomised, single‐blinded intervention trial. Lancet. 2004;363:600–607.
- , , , et al. Interobserver reliability of the chest radiograph in community‐acquired pneumonia. PORT investigators. Chest. 1996;110:343–350.
- , , , et al. Use of procalcitonin for the diagnosis of pneumonia in patients presenting with a chief complaint of dyspnea: results from the BACH (Biomarkers in Acute Heart Failure) trial. Eur J Heart Fail. 2012;14:278–286.
- , , , , . A multicenter analysis of the ED diagnosis of pneumonia. Am J Emerg Med. 2010;28:862–865.
- , , , et al. Procalcitonin predicts patients at low risk of death from community‐acquired pneumonia across all CRB‐65 classes. Eur Respir J. 2008;31:349–355.
- , , , , , . Antibiotic timing and diagnostic uncertainty in Medicare patients with pneumonia: is it reasonable to expect all patients to receive antibiotics within 4 hours? Chest. 2006;130:16–21.
- . Procalcitonin for triage of patients with respiratory tract symptoms: a case study in the trial design process for approval of a new diagnostic test for lower respiratory tract infection. Clin Infect Dis. 2011;52(S4):S351–S356.
Copyright © 2012 Society of Hospital Medicine
