User login
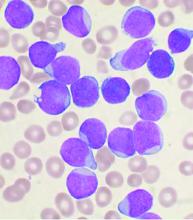
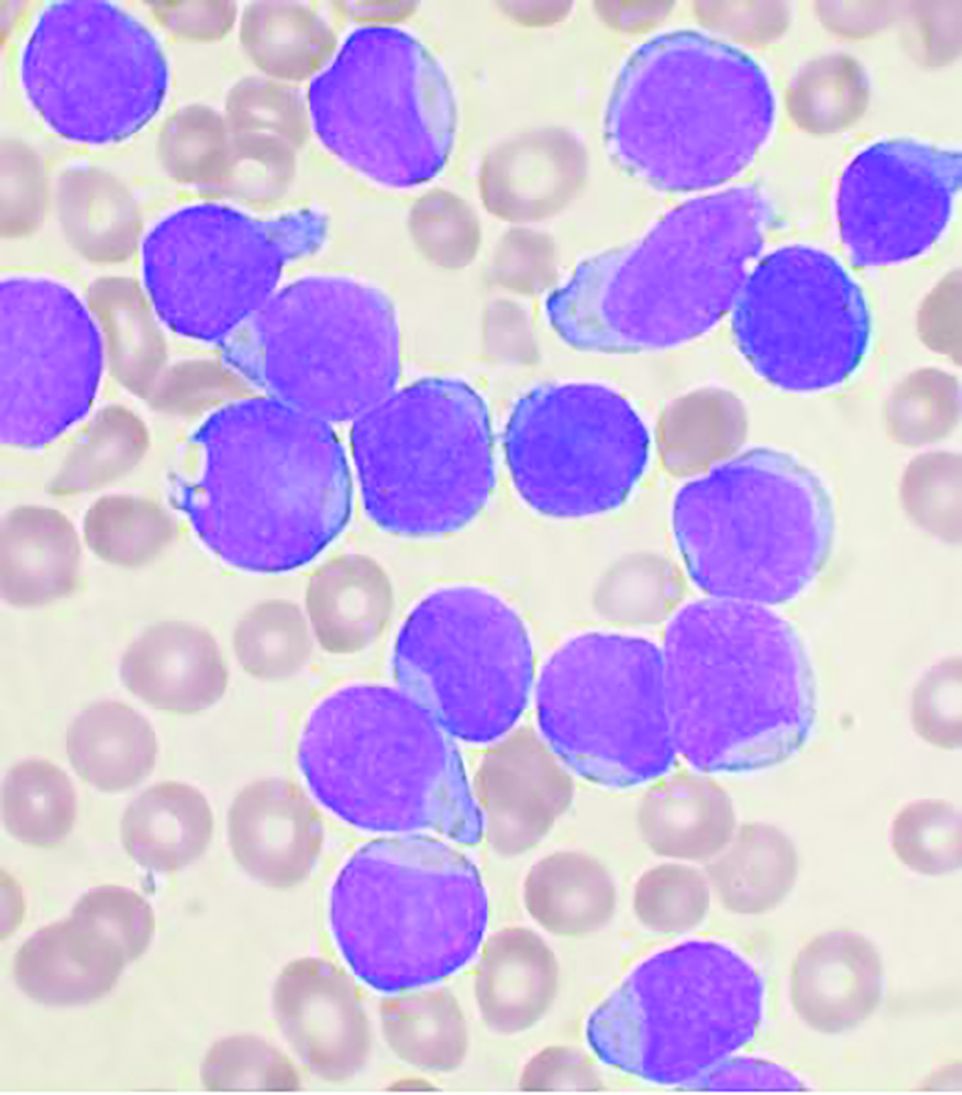
Allogeneic hematopoietic stem-cell transplantation (AHSCT) improved disease-free survival (DFS), compared with pediatric-inspired Berlin-Frankfurt-Münster (BFM-95) chemotherapy in adults with acute lymphoblastic leukemia (ALL), according to the results of retrospective study published in Clinical Lymphoma, Myeloma & Leukemia. However, overall survival (OS) was not significantly different between the two groups, as reported by Elifcan Aladag, MD, of the Hacettepe University Faculty of Medicine, Ankara, Turkey, and colleagues.
Despite this, “AHSCT is recommended for all patients with suitable donors, but the risk of transplant-related mortality should be kept in mind,” according to the researchers.
The multicenter study compared two different treatment approaches (BFM-95 chemotherapy regimen and AHSCT). The BFM-95 chemotherapy group comprised 47 newly diagnosed ALL patients. The transplant cohort comprised 83 patients with ALL in first complete remission who received AHSCT from fully matched human leukocyte antigen (HLA)-identical siblings. Thirty-five of the AHSCT patients (42.1%) received chemotherapy at least until the M stage of the BFM-95 protocol.
The primary endpoints of the study were OS and duration of DFS. OS was defined from the day of starting BFM-95 chemotherapy until death from any cause, and DFS was calculated from the date of complete remission until the date of first relapse or death from any cause, whichever occurred first, according to the authors.
Study results
The median OS was 68 months in patients who underwent AHSCT and 46 months in patients treated only with BFM-95 (P = .3). Two- and 5-year OS rates were 78% and 60% , respectively, in the AHSCT group, and 69% and 64% in the BFM-95 group (P = .06 and .13, respectively).
The median DFS was 36.6 months in patients who underwent AHSCT and 28 months in patients treated with BFM-95 (P = .033). Two- and 5-year DFS rates were 68.5% and 57%, respectively, in the AHSCT group, and 63% and 38% respectively, in the BFM-95 group (P = .12 and .029, respectively).
Mortality in the BFM-95 group was the result of sepsis due to infections (fungal infection in two patients, resistant bacterial infections in four patients). In the AHSCT group, respectively, three and seven patients died of graft-versus-host disease and bacterial infections (with fungal infections in four patients and resistant bacterial infections in three patients), according to the researchers.
“In our study, no 2-year OS and DFS difference was observed in any treatment group; however, a significant difference occurred in 5-year DFS in favor of AHSCT. This may be due to transplant-related mortality in the first 2 years, which led to no statistically significant difference,” the authors stated.
“In order to further elucidate the role of AHSCT when pediatric-derived regimens are used for the treatment of adult lymphoblastic leukemia, higher-powered randomized prospective studies are needed,” they concluded.
The authors reported that they had no conflicts of interest.
Allogeneic hematopoietic stem-cell transplantation (AHSCT) improved disease-free survival (DFS), compared with pediatric-inspired Berlin-Frankfurt-Münster (BFM-95) chemotherapy in adults with acute lymphoblastic leukemia (ALL), according to the results of retrospective study published in Clinical Lymphoma, Myeloma & Leukemia. However, overall survival (OS) was not significantly different between the two groups, as reported by Elifcan Aladag, MD, of the Hacettepe University Faculty of Medicine, Ankara, Turkey, and colleagues.
Despite this, “AHSCT is recommended for all patients with suitable donors, but the risk of transplant-related mortality should be kept in mind,” according to the researchers.
The multicenter study compared two different treatment approaches (BFM-95 chemotherapy regimen and AHSCT). The BFM-95 chemotherapy group comprised 47 newly diagnosed ALL patients. The transplant cohort comprised 83 patients with ALL in first complete remission who received AHSCT from fully matched human leukocyte antigen (HLA)-identical siblings. Thirty-five of the AHSCT patients (42.1%) received chemotherapy at least until the M stage of the BFM-95 protocol.
The primary endpoints of the study were OS and duration of DFS. OS was defined from the day of starting BFM-95 chemotherapy until death from any cause, and DFS was calculated from the date of complete remission until the date of first relapse or death from any cause, whichever occurred first, according to the authors.
Study results
The median OS was 68 months in patients who underwent AHSCT and 46 months in patients treated only with BFM-95 (P = .3). Two- and 5-year OS rates were 78% and 60% , respectively, in the AHSCT group, and 69% and 64% in the BFM-95 group (P = .06 and .13, respectively).
The median DFS was 36.6 months in patients who underwent AHSCT and 28 months in patients treated with BFM-95 (P = .033). Two- and 5-year DFS rates were 68.5% and 57%, respectively, in the AHSCT group, and 63% and 38% respectively, in the BFM-95 group (P = .12 and .029, respectively).
Mortality in the BFM-95 group was the result of sepsis due to infections (fungal infection in two patients, resistant bacterial infections in four patients). In the AHSCT group, respectively, three and seven patients died of graft-versus-host disease and bacterial infections (with fungal infections in four patients and resistant bacterial infections in three patients), according to the researchers.
“In our study, no 2-year OS and DFS difference was observed in any treatment group; however, a significant difference occurred in 5-year DFS in favor of AHSCT. This may be due to transplant-related mortality in the first 2 years, which led to no statistically significant difference,” the authors stated.
“In order to further elucidate the role of AHSCT when pediatric-derived regimens are used for the treatment of adult lymphoblastic leukemia, higher-powered randomized prospective studies are needed,” they concluded.
The authors reported that they had no conflicts of interest.
Allogeneic hematopoietic stem-cell transplantation (AHSCT) improved disease-free survival (DFS), compared with pediatric-inspired Berlin-Frankfurt-Münster (BFM-95) chemotherapy in adults with acute lymphoblastic leukemia (ALL), according to the results of retrospective study published in Clinical Lymphoma, Myeloma & Leukemia. However, overall survival (OS) was not significantly different between the two groups, as reported by Elifcan Aladag, MD, of the Hacettepe University Faculty of Medicine, Ankara, Turkey, and colleagues.
Despite this, “AHSCT is recommended for all patients with suitable donors, but the risk of transplant-related mortality should be kept in mind,” according to the researchers.
The multicenter study compared two different treatment approaches (BFM-95 chemotherapy regimen and AHSCT). The BFM-95 chemotherapy group comprised 47 newly diagnosed ALL patients. The transplant cohort comprised 83 patients with ALL in first complete remission who received AHSCT from fully matched human leukocyte antigen (HLA)-identical siblings. Thirty-five of the AHSCT patients (42.1%) received chemotherapy at least until the M stage of the BFM-95 protocol.
The primary endpoints of the study were OS and duration of DFS. OS was defined from the day of starting BFM-95 chemotherapy until death from any cause, and DFS was calculated from the date of complete remission until the date of first relapse or death from any cause, whichever occurred first, according to the authors.
Study results
The median OS was 68 months in patients who underwent AHSCT and 46 months in patients treated only with BFM-95 (P = .3). Two- and 5-year OS rates were 78% and 60% , respectively, in the AHSCT group, and 69% and 64% in the BFM-95 group (P = .06 and .13, respectively).
The median DFS was 36.6 months in patients who underwent AHSCT and 28 months in patients treated with BFM-95 (P = .033). Two- and 5-year DFS rates were 68.5% and 57%, respectively, in the AHSCT group, and 63% and 38% respectively, in the BFM-95 group (P = .12 and .029, respectively).
Mortality in the BFM-95 group was the result of sepsis due to infections (fungal infection in two patients, resistant bacterial infections in four patients). In the AHSCT group, respectively, three and seven patients died of graft-versus-host disease and bacterial infections (with fungal infections in four patients and resistant bacterial infections in three patients), according to the researchers.
“In our study, no 2-year OS and DFS difference was observed in any treatment group; however, a significant difference occurred in 5-year DFS in favor of AHSCT. This may be due to transplant-related mortality in the first 2 years, which led to no statistically significant difference,” the authors stated.
“In order to further elucidate the role of AHSCT when pediatric-derived regimens are used for the treatment of adult lymphoblastic leukemia, higher-powered randomized prospective studies are needed,” they concluded.
The authors reported that they had no conflicts of interest.
FROM CLINICAL LYMPHOMA, MYELOMA & LEUKEMIA