User login
When it’s time for ‘the talk’: Sexuality and your geriatric patient
Recent studies suggest that most older adults maintain sexual interest well into late life; many, however, experience sexual dysfunction. This article provides psychiatric practitioners with current information regarding sexuality and aging, as well as psychiatric and systemic medical comorbidities and sexual side effects of medications. Practice guidelines for assessing and managing sexual dysfunction have been developed for use in many medical specialties, and such guidance would be welcome in psychiatric practice.
This article addresses the myth of “geriatric asexuality” and its potential impact on clinical practice, the effects of age-related physiological changes on sexual activity, the importance of sexuality in the lives of older adults, and sensitive questions clinicians can pose about geriatric sexuality. We also will discuss:
• the importance of including a sexual assessment in the comprehensive psychiatric evaluation
• recognizing sexual dysfunction
• providing appropriate management within a multi-disciplinary, collaborative approach.
Sexuality after 65
Regardless of age, sexual activity can provide a sense of comfort and elicit a positive emotional and physical response.1 Hillman2 defined human sexuality as any combination of sexual behavior, emotional intimacy, and sense of sexual identity.
Sexuality in the aging population generally is an understudied area, obscured by the myth of “geriatric asexuality” and subject to numerous psychosocial variables.1 Previous research, focused on a biological perspective of sexuality, has largely overlooked psychological and social influences.3 It has been assumed that, with age, physical and hormonal changes or chronic illness ordinarily reduce or eliminate sexual desire and sexual behavior.3 However, the majority of older adults (defined as age ≥65) report a moderate-to-high level of sexual interest well into late life.1,3
Sexual function remains a subject often neglected in psychiatry. Sexual dysfunctions, as described in the DSM-5,4 do not include age-related changes in sexual function. In addition to physiological changes, sexual difficulties can result from relationship strain, systemic medical or psychiatric disorders, and sexual side effects of medications.
CASE REPORT
Mr. C, age 71 and married, is being treated for a major depressive episode that followed a course of shingles and persistent postherpetic neuralgia. Medications are: escitalopram, 20 mg/d; pregabalin, 150 mg/d; and ramipril, 5 mg/d. Mr. C is physically active and involved in social activities; he has no substance use history. He attends clinic visits with his wife.
Mr. C reports that despite significant improvement of his depressive and pain symptoms, he now experiences sexual difficulties, which he seems hesitant to discuss in detail. According to his wife, Mr. C appears to lack sexual desire and has difficulty initiating and maintaining an erection. She asks Mr. C’s psychiatrist whether she should stop her estrogen treatment, intended to enhance her sexual function, given that the couple is no longer engaging in sexual intercourse.
Mr. C admits to missing physical intimacy; however, he states, “If I have to make a choice between having sex with my wife and getting this depression out of my head, I’m going to pick getting rid of the depression.” Mrs. C says she is becoming dissatisfied with their marriage and the limited time she and her husband now spend together. Mr. C’s psychiatrist suggests that Mr. C and his wife undergo couples counseling.
Physiological changes with aging
In both women and men, the reproductive system undergoes age-related physiological changes.
Women. In women, the phase of decline in ovarian function and resulting decline in sex steroid production (estradiol and progesterone) is referred to as the climacteric, with menopause being determined retrospectively by the cessation of a menstrual period for 1 year.5
Menopausal symptoms typically occur between age 40 and 58; the average age of menopause is 51.6,7 Both estradiol and progesterone levels decline with menopause, and anovulation and ovarian failure ensue. A more gradual decline of female testosterone levels also occurs with aging, starting in the fourth decade of life.8
Clinical manifestations of menopause include vasomotor symptoms (ie, “hot flushes”), sleep disturbances, anxiety and depressive symptoms, decreased bone mineral density, and increased risk of cardiovascular disease.6,7 Loss of estrogen as well as continued loss of testosterone can result in dyspareunia because of atrophy and decreased vulvar and vaginal lubrication, with sexual excitement achieved less quickly, and a decreased intensity of orgasm.7
Men. Research has shown that testosterone levels are highest in men in the second and third decades, with a subsequent gradual decline.9 Older men with a low testosterone level are described as experiencing “late-onset hypogonadism,” also known by the popularized term “andropause.”10 This is attributed to decreased activity at the testicular and hypothalamic levels.10
Nonetheless, only a small fraction of older men with confirmed androgen deficiency are clinically symptomatic.11,12 Low testosterone is associated with decreased libido; it can hinder morning erections, contribute to erectile dysfunction, and result in erections that require physical stimulation.13
Notably, erectile dysfunction involves several other etiologic factors: psychiatric (eg, relationship difficulties, depression), neurogenic (eg, spinal cord injury), endocrine (eg, hyperprolactinemia), arteriogenic (eg, hypertension, type 2 diabetes mellitus), and drug-induced (eg, antidepressants, antihypertensives).14 A low testosterone level also has been associated with potential cognitive changes, decreased bone mineral density, metabolic syndrome (eg, increased risk of type 2 diabetes mellitus), and cardiovascular mortality.10
Effects on sexual activity. How much age-related physiological changes impact sexual practices in the geriatric population is uncertain. A study following 3,302 women through menopause over 6 years found some decline in sexual activity; however, this decline was not associated with increased sexual pain, decreased desire, or lack of arousal.15 Men continue to find ways to remain sexually active despite physiological changes (eg, erectile difficulties), but the etiology of sexual dysfunction in later life remains multi-modal, involving physical, psychological, and relational factors.16,17
Sexual practices in older adults
Researchers for the National Social Life, Health, and Aging Project (NSHAP) have examined sexual activities, behaviors, and problems in >3,000 older community-dwelling men and women across the United States, using information collected from in-home interviews.18 This study found that sexual activity, defined as any mutually voluntary sexual contact with another person, declines with age; however, even in the oldest age group (age 75 to 85), 39% of men and 17% of women reported being sexually active in the last 12 months. Among these persons, 54% reported sexual activity at least 2 times per month; 23% reported having sex at least once a week; and 32% reported engaging in oral sex. Partner availability predicted sexual activity.
Respondents with self-reported poor physical health were more likely to experience sexual problems (eg, difficulty with erection or lubrication, dyspareunia, and lack of pleasure). The most commonly reported reason for sexual inactivity in those with a spouse or other intimate partner was the male partner’s poor physical health.18
A longitudinal study, part of the Women’s Healthy Ageing Project, examined changes in sexual function at late menopause compared with early menopause. Although the researchers also found an age-related decrease in sexual activity, 50% of their late-menopause respondents (mean age, 70; range, 64 to 77) still reported sexual activity in the previous month, with 35% of this subgroup reporting sexual activity at least once a week; 83% reported sexual thoughts or fantasies.19 Availability of a partner, absence of a history of depression, moderate (compared with no) alcohol consumption, and better cognitive function were significantly associated with a higher level of sexual activity.19
In the Successful Aging Evaluation study, conducted in San Diego County, California, community-dwelling older partnered adults age 50 to 99 (mean age, 75) were surveyed about their sexual health after a cognitive screen by telephone20; rating scales for depression, anxiety, and physical function also were included. Results included 41% of men and 35% of women reporting sexual activity at least once a week, and only 21% of men and 24% of women reporting no sexual activity in the previous year. Depressive symptoms were most highly correlated with lack of sexual activity.20
Overall, these studies reveal that positive physical and mental health, access to a healthy partner, and a positive attitude toward sex are correlated with sexual activity in later life, whereas barriers to sexual activity include lack of a healthy sexual partner, depression, and chronic systemic medical illnesses, such as coronary artery disease or type 2 diabetes mellitus.13,17,21-24 Sexual activity and satisfaction have been positively associated with perceived general well-being and self-esteem.25,26 Conversely, sexual difficulties secondary to disease can be a source of distress for couples.27
Possibly overlooked? It is important to note that sexuality itself is a subjective area. Psychological intimacy is a universal phenomenon, and its physical expression may evolve as couples accommodate to age-related bodily changes. Means of achieving physical closeness, other than intercourse (eg, intimate touching, hand holding, kissing, or even acts of caretaking), may not be adequately captured in studies that look specifically at sexual activity.
Taking a sexual history in a geriatric patient
Because sexuality can be an uncomfortable topic for geriatric patients to discuss, sexual problems in this population often go unrecognized. It has been suggested that psychiatrists are more likely to inquire about sexual activity in middle-aged patients than geriatric patients with the same psychiatric presentation—perhaps illustrating a bias against taking a sexual history from a geriatric patient.28 However, because many older patients can experience depression or anxiety disorders in relation to normal sexual changes or sexual dysfunction within the context of their intimate relationships, it is essential to bring these issues to light.
Although a sexual history may not be the focus of a first clinical encounter, consider making such an assessment at a relatively early stage of patient care. The importance of such dialogue is 2-fold:
• It demonstrates to the patient that talking about sexuality in a respectful and empathic manner is appropriate and can encourage patients to communicate more effectively about sexuality with clinicians and with sexual partners.
• It helps elicit medical information needed to make an accurate diagnosis and provide adequate management.
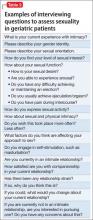
How to begin. As a starting point to taking a sexual history, an open-ended invitation for the geriatric patient to share information may be best, such as “What would you like to tell me about your sexual life?” See further suggestions (Table 1) and examples of more detailed questions to ask once a dialogue has been initiated (Table 2). Additional factors that may contribute to sexual dysfunction are presented in Table 3.1,27,29,30
CASE CONTINUED
In Mr. C’s case, an assessment of his sexual history, including risk factors for sexual dysfunction, is completed. Results from laboratory investigations, including a total testosterone level, are within normal limits.
Mr. C asks about using medications with fewer sexual side effects (he has been taking 3 medications that can contribute to sexual dysfunction). A gradual cross-taper of escitalopram, 20 mg/d, to mirtazapine, 45 mg/d, is implemented, along with tapering pregabalin to 50 mg/d.
Mr. C’s psychiatric and pain symptom improvement is maintained. He notices a boost in his sexual desire but has minimal improvement in erectile dysfunction. He is encouraged to speak with his primary care physician about an antihypertensive agent with less impact on sexual function, as well as therapeutic agents for erectile dysfunction; these, he declines.
At a subsequent visit, Mr. C reports feeling less apprehension about sexual performance. He is now willing to consider further medication options with his primary care physician, and agrees to a recommendation for couples psychotherapy.
As illustrated in Mr. C’s case, the recommended sexual assessment and management strategies to consider at a minimum in psychiatric practice are listed in Table 4.
STI risk in geriatric patients
The risk of sexually transmitted infections (STIs), including human immunodeficiency virus (HIV), often is overlooked in sexually active older adults. Although STIs are more common among younger adults, there is recent evidence of increased incidence in the geriatric population31 (with the highest risk of incident HIV and some STIs in older men who have sex with men32). These increased rates can be explained, at least in part, by:
• older men being less likely to use a condom during sexual activity
• promotion of viral entry in older women through a drier, thinner vaginal wall
• increased longevity of HIV-positive persons.31
Routine STI screening is not warranted in all older adults, but education and prevention strategies in sexually active seniors who are at greater risk of STIs are recommended. Particularly, clinicians should seek opportunities to discuss risk factors and safe sex practices (eg, using condoms, limiting number of sexual partners, practicing good hygiene, engaging in preventive care), and provide behavioral counseling where appropriate.31,33
Additional considerations in geriatric sexuality
Because psychiatric and systemic medical conditions can hinder sexual function, it is essential to identify and manage these conditions. Several neuropsychiatric disorders, including mood and neurocognitive disorders, can not only cause sexual dysfunction, but also can raise ethical dilemmas for clinicians, such as reduced decisional capacity in cognitively impaired patients to consent to sexual activity.1,34
In some patients, psychological, environmental, and pharmacological treatment options may help. A phosphodiesterase type 5 inhibitor for erectile dysfunction can be prescribed by the primary care physician, a psychiatrist, or another specialist, depending on the physician’s expertise and comfort level.
Sequencing of sexual dysfunction. Notably, there is a common paradox in mood disorders. Decreased sexual interest or performance may represent an aspect of anhedonia associated with depression, whereas sexual dysfunction could also result from medication use (particularly that of serotonergic antidepressants, such as selective serotonin reuptake inhibitors and serotonin-norepinephrine inhibitors), even as other depressive symptoms improve. Therefore, it is critical to analyze sequencing of sexual dysfunction—as part of the presenting mood symptoms or dysfunction induced by antidepressant treatment.
Geriatric sexuality in the digital age. Because older adults represent a rapidly growing segment of digital device users,35 Internet use is likely to play a role in the future of sexuality and “digital intimacy,” in that older adults can engage in online sexual activities. The Internet also can be a tool to access medical education.
Related Resources
• Burghardt KJ, Gardner KN. Sildenafil for SSRI-induced sexual dysfunction. Current Psychiatry. 2013;12(4):29-32,A.
• Maciel M, Laganà L. Older women’s sexual desire problems: biopsychosocial factors impacting them and barriers to their clinical assessment [published online January 5, 2014]. Biomed Res Int. 2014;2014:107217. doi: 10.1155/2014/107217.
Drug Brand Names
Bupropion • Wellbutrin, Zyban Mirtazapine • Remeron
Carbamazepine • Tegretol Oxcarbazepine • Trileptal
Clonidine • Catapres Phenobarbital • Luminal
Donepezil • Aricept Phenytoin • Dilantin
Escitalopram • Lexapro Pregabalin • Lyrica
Gabapentin • Neurontin Ramipril • Altace
Lamotrigine • Lamictal Rivastigmine • Exelon
Lithium • Eskalith, Lithobid Trazodone • Desyrel
Memantine • Namenda Valproic acid • Depakote
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Jagus CE, Benbow SM. Sexuality in older men with mental health problems. Sex Relation Ther. 2002;17(3):271-279.
2. Hillman JL. Clinical perspectives on elderly sexuality. New York, NY: Springer; 2000.
3. DeLamater JD, Sill M. Sexual desire in later life. J Sex Res. 2005;42(2):138-149.
4. Diagnostic and statistical manual of mental disorders. 5th ed. Washington, DC: American Psychiatric Association; 2013.
5. Laufer LR, Gambone JC. Climacteric: menopause and peri-and postmenopause. In: Hacker NF, Gambone JC, Hobel CJ. Hacker and Moore’s essentials of obstetrics and gynecology. 5th ed. Philadelphia, PA: Saunders/Elsevier; 2010:379-385.
6. Wilson MM. Menopause. Clin Geriatr Med. 2003;19(3): 483-506.
7. Reid R, Abramson BL, Blake J, et al. Managing menopause. J Obstet Gynaecol Can. 2014;36(9):830-838.
8. Horstman AM, Dillon EL, Urban RJ, et al. The role of androgens and estrogens on healthy aging and longevity. J Gerontol A Biol Sci Med Sci. 2012;67(11):1140-1152.
9. Wu FC, Tajar A, Pye SR, et al. Hypothalamic-pituitary-testicular axis disruptions in older men are differentially linked to age and modifiable risk factors: the European Male Aging Study. J Clin Endocrinol Metab. 2008;93(7):2737-2745.
10. Basaria S. Reproductive aging in men. Endocrinol Metab Clin North Am. 2013;42(2):255-270.
11. Wu FC, Tajar A, Beynon JM, et al. Identification of late-onset hypogonadism in middle-aged and elderly men. N Engl J Med. 2010;363(2):123-135.
12. Araujo AB, Esche GR, Kupelian V, et al. Prevalence of symptomatic androgen deficiency in men. J Clin Endocrinol Metab. 2007;92(11):4241-4247.
13. Lochlainn MN, Kenny RA. Sexual activity and aging. J Am Med Dir Assoc. 2013;14(8):565-572.
14. McMahon CG. Erectile dysfunction. Intern Med J. 2014;44(1):18-26.
15. Avis NE, Brockwell S, Randolph JF Jr, et al. Longitudinal changes in sexual functioning as women transition through menopause: results from the Study of Women’s Health Across the Nation. Menopause. 2009;16(3):442-452.
16. Perelman M, Shabsigh R, Seftel A, et al. Attitudes of men with erectile dysfunction: a cross-national survey. J Sex Med. 2005;2(3):397-406.
17. Corona G, Rastrelli G, Maseroli E, et al. Sexual function of the ageing male. Best Pract Res Clin Endocrinol Metab. 2013;27(4):581-601.
18. Lindau ST, Schumm LP, Laumann EO, et al. A study of sexuality and health among older adults in the United States. N Engl J Med. 2007;357(8):762-774.
19. Lonnèe-Hoffmann RA, Dennerstein L, Lehert P, et al. Sexual function in the late postmenopause: a decade of follow-up in a population-based cohort of Australian women. J Sex Med. 2014;11(8):2029-2038.
20. Wang V, Depp CA, Ceglowski J, et al. Sexual health and function in later life: a population-based study of 606 older adults with a partner. Am J Geriatr Psychiatry. 2015;23(3):227-233.
21. Garrett D. Psychosocial barriers to sexual intimacy for older people. Br J Nurs. 2014;23(6):327-331.
22. DeLamater J, Karraker A. Sexual functioning in older adults. Curr Psychiatry Rep. 2009;11(1):6-11.
23. DeLamater J. Sexual expression in later life: a review and synthesis. J Sex Res. 2012;49(2-3):125-141.
24. Inelmen EM, Sergi G, Girardi A, et al. The importance of sexual health in the elderly: breaking down barriers and taboos. Aging Clin Exp Res. 2012;24(suppl 3):31-34.
25. Choi KB, Jang SH, Lee MY, et al. Sexual life and self-esteem in married elderly. Arch Gerontol Geriatr. 2011;53(1):e17-e20.
26. Davison SL, Bell RJ, LaChina M, et al. The relationship between self-reported sexual satisfaction and general well-being in women. J Sex Med. 2009;6(10):2690-2697.
27. Morley JE, Tariq SH. Sexuality and disease. Clin Geriatr Med. 2003;19(3):563-573.
28. Bouman WP, Arcelus J. Are psychiatrists guilty of “ageism” when it comes to taking a sexual history? Int J Geriatr Psychiatry. 2001;16(1):27-31.
29. La Torre A, Giupponi G, Duffy DM, et al. Sexual dysfunction related to psychotropic drugs: a critical review. Part III: mood stabilizers and anxiolytic drugs. Pharmacopsychiatry. 2014;47(1):1-6.
30. Tucker I. Management of inappropriate sexual behaviors in dementia: a literature review. Int Psychogeriatr. 2010; 22(5):683-692.
31. Imparato T, Sanders D. STD prevalence demands clinical awareness. Aging Well. 2012;5(1):14.
32. Poynten IM, Grulich AE, Templeton DJ. Sexually transmitted infections in older populations. Curr Opin Infect Dis. 2013;26(1):80-85.
33. Talashek ML, Tichy AM, Epping H. Sexually transmitted diseases in the elderly—issues and recommendations. J Gerontol Nurs. 1990;16(4):33-40.
34. Benbow SM, Jagus CE. Sexuality in older women with mental health problems. Sex Relation Ther. 2002;17(3):261-270.
35. Veenhof B, Timusk P. Online activities of Canadian boomers and seniors. http://www.statcan.gc.ca/pub/ 11-008-x/2009002/article/10910-eng.htm#tphp. Accessed March 26, 2015.
Recent studies suggest that most older adults maintain sexual interest well into late life; many, however, experience sexual dysfunction. This article provides psychiatric practitioners with current information regarding sexuality and aging, as well as psychiatric and systemic medical comorbidities and sexual side effects of medications. Practice guidelines for assessing and managing sexual dysfunction have been developed for use in many medical specialties, and such guidance would be welcome in psychiatric practice.
This article addresses the myth of “geriatric asexuality” and its potential impact on clinical practice, the effects of age-related physiological changes on sexual activity, the importance of sexuality in the lives of older adults, and sensitive questions clinicians can pose about geriatric sexuality. We also will discuss:
• the importance of including a sexual assessment in the comprehensive psychiatric evaluation
• recognizing sexual dysfunction
• providing appropriate management within a multi-disciplinary, collaborative approach.
Sexuality after 65
Regardless of age, sexual activity can provide a sense of comfort and elicit a positive emotional and physical response.1 Hillman2 defined human sexuality as any combination of sexual behavior, emotional intimacy, and sense of sexual identity.
Sexuality in the aging population generally is an understudied area, obscured by the myth of “geriatric asexuality” and subject to numerous psychosocial variables.1 Previous research, focused on a biological perspective of sexuality, has largely overlooked psychological and social influences.3 It has been assumed that, with age, physical and hormonal changes or chronic illness ordinarily reduce or eliminate sexual desire and sexual behavior.3 However, the majority of older adults (defined as age ≥65) report a moderate-to-high level of sexual interest well into late life.1,3
Sexual function remains a subject often neglected in psychiatry. Sexual dysfunctions, as described in the DSM-5,4 do not include age-related changes in sexual function. In addition to physiological changes, sexual difficulties can result from relationship strain, systemic medical or psychiatric disorders, and sexual side effects of medications.
CASE REPORT
Mr. C, age 71 and married, is being treated for a major depressive episode that followed a course of shingles and persistent postherpetic neuralgia. Medications are: escitalopram, 20 mg/d; pregabalin, 150 mg/d; and ramipril, 5 mg/d. Mr. C is physically active and involved in social activities; he has no substance use history. He attends clinic visits with his wife.
Mr. C reports that despite significant improvement of his depressive and pain symptoms, he now experiences sexual difficulties, which he seems hesitant to discuss in detail. According to his wife, Mr. C appears to lack sexual desire and has difficulty initiating and maintaining an erection. She asks Mr. C’s psychiatrist whether she should stop her estrogen treatment, intended to enhance her sexual function, given that the couple is no longer engaging in sexual intercourse.
Mr. C admits to missing physical intimacy; however, he states, “If I have to make a choice between having sex with my wife and getting this depression out of my head, I’m going to pick getting rid of the depression.” Mrs. C says she is becoming dissatisfied with their marriage and the limited time she and her husband now spend together. Mr. C’s psychiatrist suggests that Mr. C and his wife undergo couples counseling.
Physiological changes with aging
In both women and men, the reproductive system undergoes age-related physiological changes.
Women. In women, the phase of decline in ovarian function and resulting decline in sex steroid production (estradiol and progesterone) is referred to as the climacteric, with menopause being determined retrospectively by the cessation of a menstrual period for 1 year.5
Menopausal symptoms typically occur between age 40 and 58; the average age of menopause is 51.6,7 Both estradiol and progesterone levels decline with menopause, and anovulation and ovarian failure ensue. A more gradual decline of female testosterone levels also occurs with aging, starting in the fourth decade of life.8
Clinical manifestations of menopause include vasomotor symptoms (ie, “hot flushes”), sleep disturbances, anxiety and depressive symptoms, decreased bone mineral density, and increased risk of cardiovascular disease.6,7 Loss of estrogen as well as continued loss of testosterone can result in dyspareunia because of atrophy and decreased vulvar and vaginal lubrication, with sexual excitement achieved less quickly, and a decreased intensity of orgasm.7
Men. Research has shown that testosterone levels are highest in men in the second and third decades, with a subsequent gradual decline.9 Older men with a low testosterone level are described as experiencing “late-onset hypogonadism,” also known by the popularized term “andropause.”10 This is attributed to decreased activity at the testicular and hypothalamic levels.10
Nonetheless, only a small fraction of older men with confirmed androgen deficiency are clinically symptomatic.11,12 Low testosterone is associated with decreased libido; it can hinder morning erections, contribute to erectile dysfunction, and result in erections that require physical stimulation.13
Notably, erectile dysfunction involves several other etiologic factors: psychiatric (eg, relationship difficulties, depression), neurogenic (eg, spinal cord injury), endocrine (eg, hyperprolactinemia), arteriogenic (eg, hypertension, type 2 diabetes mellitus), and drug-induced (eg, antidepressants, antihypertensives).14 A low testosterone level also has been associated with potential cognitive changes, decreased bone mineral density, metabolic syndrome (eg, increased risk of type 2 diabetes mellitus), and cardiovascular mortality.10
Effects on sexual activity. How much age-related physiological changes impact sexual practices in the geriatric population is uncertain. A study following 3,302 women through menopause over 6 years found some decline in sexual activity; however, this decline was not associated with increased sexual pain, decreased desire, or lack of arousal.15 Men continue to find ways to remain sexually active despite physiological changes (eg, erectile difficulties), but the etiology of sexual dysfunction in later life remains multi-modal, involving physical, psychological, and relational factors.16,17
Sexual practices in older adults
Researchers for the National Social Life, Health, and Aging Project (NSHAP) have examined sexual activities, behaviors, and problems in >3,000 older community-dwelling men and women across the United States, using information collected from in-home interviews.18 This study found that sexual activity, defined as any mutually voluntary sexual contact with another person, declines with age; however, even in the oldest age group (age 75 to 85), 39% of men and 17% of women reported being sexually active in the last 12 months. Among these persons, 54% reported sexual activity at least 2 times per month; 23% reported having sex at least once a week; and 32% reported engaging in oral sex. Partner availability predicted sexual activity.
Respondents with self-reported poor physical health were more likely to experience sexual problems (eg, difficulty with erection or lubrication, dyspareunia, and lack of pleasure). The most commonly reported reason for sexual inactivity in those with a spouse or other intimate partner was the male partner’s poor physical health.18
A longitudinal study, part of the Women’s Healthy Ageing Project, examined changes in sexual function at late menopause compared with early menopause. Although the researchers also found an age-related decrease in sexual activity, 50% of their late-menopause respondents (mean age, 70; range, 64 to 77) still reported sexual activity in the previous month, with 35% of this subgroup reporting sexual activity at least once a week; 83% reported sexual thoughts or fantasies.19 Availability of a partner, absence of a history of depression, moderate (compared with no) alcohol consumption, and better cognitive function were significantly associated with a higher level of sexual activity.19
In the Successful Aging Evaluation study, conducted in San Diego County, California, community-dwelling older partnered adults age 50 to 99 (mean age, 75) were surveyed about their sexual health after a cognitive screen by telephone20; rating scales for depression, anxiety, and physical function also were included. Results included 41% of men and 35% of women reporting sexual activity at least once a week, and only 21% of men and 24% of women reporting no sexual activity in the previous year. Depressive symptoms were most highly correlated with lack of sexual activity.20
Overall, these studies reveal that positive physical and mental health, access to a healthy partner, and a positive attitude toward sex are correlated with sexual activity in later life, whereas barriers to sexual activity include lack of a healthy sexual partner, depression, and chronic systemic medical illnesses, such as coronary artery disease or type 2 diabetes mellitus.13,17,21-24 Sexual activity and satisfaction have been positively associated with perceived general well-being and self-esteem.25,26 Conversely, sexual difficulties secondary to disease can be a source of distress for couples.27
Possibly overlooked? It is important to note that sexuality itself is a subjective area. Psychological intimacy is a universal phenomenon, and its physical expression may evolve as couples accommodate to age-related bodily changes. Means of achieving physical closeness, other than intercourse (eg, intimate touching, hand holding, kissing, or even acts of caretaking), may not be adequately captured in studies that look specifically at sexual activity.
Taking a sexual history in a geriatric patient
Because sexuality can be an uncomfortable topic for geriatric patients to discuss, sexual problems in this population often go unrecognized. It has been suggested that psychiatrists are more likely to inquire about sexual activity in middle-aged patients than geriatric patients with the same psychiatric presentation—perhaps illustrating a bias against taking a sexual history from a geriatric patient.28 However, because many older patients can experience depression or anxiety disorders in relation to normal sexual changes or sexual dysfunction within the context of their intimate relationships, it is essential to bring these issues to light.
Although a sexual history may not be the focus of a first clinical encounter, consider making such an assessment at a relatively early stage of patient care. The importance of such dialogue is 2-fold:
• It demonstrates to the patient that talking about sexuality in a respectful and empathic manner is appropriate and can encourage patients to communicate more effectively about sexuality with clinicians and with sexual partners.
• It helps elicit medical information needed to make an accurate diagnosis and provide adequate management.
How to begin. As a starting point to taking a sexual history, an open-ended invitation for the geriatric patient to share information may be best, such as “What would you like to tell me about your sexual life?” See further suggestions (Table 1) and examples of more detailed questions to ask once a dialogue has been initiated (Table 2). Additional factors that may contribute to sexual dysfunction are presented in Table 3.1,27,29,30
CASE CONTINUED
In Mr. C’s case, an assessment of his sexual history, including risk factors for sexual dysfunction, is completed. Results from laboratory investigations, including a total testosterone level, are within normal limits.
Mr. C asks about using medications with fewer sexual side effects (he has been taking 3 medications that can contribute to sexual dysfunction). A gradual cross-taper of escitalopram, 20 mg/d, to mirtazapine, 45 mg/d, is implemented, along with tapering pregabalin to 50 mg/d.
Mr. C’s psychiatric and pain symptom improvement is maintained. He notices a boost in his sexual desire but has minimal improvement in erectile dysfunction. He is encouraged to speak with his primary care physician about an antihypertensive agent with less impact on sexual function, as well as therapeutic agents for erectile dysfunction; these, he declines.
At a subsequent visit, Mr. C reports feeling less apprehension about sexual performance. He is now willing to consider further medication options with his primary care physician, and agrees to a recommendation for couples psychotherapy.
As illustrated in Mr. C’s case, the recommended sexual assessment and management strategies to consider at a minimum in psychiatric practice are listed in Table 4.
STI risk in geriatric patients
The risk of sexually transmitted infections (STIs), including human immunodeficiency virus (HIV), often is overlooked in sexually active older adults. Although STIs are more common among younger adults, there is recent evidence of increased incidence in the geriatric population31 (with the highest risk of incident HIV and some STIs in older men who have sex with men32). These increased rates can be explained, at least in part, by:
• older men being less likely to use a condom during sexual activity
• promotion of viral entry in older women through a drier, thinner vaginal wall
• increased longevity of HIV-positive persons.31
Routine STI screening is not warranted in all older adults, but education and prevention strategies in sexually active seniors who are at greater risk of STIs are recommended. Particularly, clinicians should seek opportunities to discuss risk factors and safe sex practices (eg, using condoms, limiting number of sexual partners, practicing good hygiene, engaging in preventive care), and provide behavioral counseling where appropriate.31,33
Additional considerations in geriatric sexuality
Because psychiatric and systemic medical conditions can hinder sexual function, it is essential to identify and manage these conditions. Several neuropsychiatric disorders, including mood and neurocognitive disorders, can not only cause sexual dysfunction, but also can raise ethical dilemmas for clinicians, such as reduced decisional capacity in cognitively impaired patients to consent to sexual activity.1,34
In some patients, psychological, environmental, and pharmacological treatment options may help. A phosphodiesterase type 5 inhibitor for erectile dysfunction can be prescribed by the primary care physician, a psychiatrist, or another specialist, depending on the physician’s expertise and comfort level.
Sequencing of sexual dysfunction. Notably, there is a common paradox in mood disorders. Decreased sexual interest or performance may represent an aspect of anhedonia associated with depression, whereas sexual dysfunction could also result from medication use (particularly that of serotonergic antidepressants, such as selective serotonin reuptake inhibitors and serotonin-norepinephrine inhibitors), even as other depressive symptoms improve. Therefore, it is critical to analyze sequencing of sexual dysfunction—as part of the presenting mood symptoms or dysfunction induced by antidepressant treatment.
Geriatric sexuality in the digital age. Because older adults represent a rapidly growing segment of digital device users,35 Internet use is likely to play a role in the future of sexuality and “digital intimacy,” in that older adults can engage in online sexual activities. The Internet also can be a tool to access medical education.
Related Resources
• Burghardt KJ, Gardner KN. Sildenafil for SSRI-induced sexual dysfunction. Current Psychiatry. 2013;12(4):29-32,A.
• Maciel M, Laganà L. Older women’s sexual desire problems: biopsychosocial factors impacting them and barriers to their clinical assessment [published online January 5, 2014]. Biomed Res Int. 2014;2014:107217. doi: 10.1155/2014/107217.
Drug Brand Names
Bupropion • Wellbutrin, Zyban Mirtazapine • Remeron
Carbamazepine • Tegretol Oxcarbazepine • Trileptal
Clonidine • Catapres Phenobarbital • Luminal
Donepezil • Aricept Phenytoin • Dilantin
Escitalopram • Lexapro Pregabalin • Lyrica
Gabapentin • Neurontin Ramipril • Altace
Lamotrigine • Lamictal Rivastigmine • Exelon
Lithium • Eskalith, Lithobid Trazodone • Desyrel
Memantine • Namenda Valproic acid • Depakote
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Recent studies suggest that most older adults maintain sexual interest well into late life; many, however, experience sexual dysfunction. This article provides psychiatric practitioners with current information regarding sexuality and aging, as well as psychiatric and systemic medical comorbidities and sexual side effects of medications. Practice guidelines for assessing and managing sexual dysfunction have been developed for use in many medical specialties, and such guidance would be welcome in psychiatric practice.
This article addresses the myth of “geriatric asexuality” and its potential impact on clinical practice, the effects of age-related physiological changes on sexual activity, the importance of sexuality in the lives of older adults, and sensitive questions clinicians can pose about geriatric sexuality. We also will discuss:
• the importance of including a sexual assessment in the comprehensive psychiatric evaluation
• recognizing sexual dysfunction
• providing appropriate management within a multi-disciplinary, collaborative approach.
Sexuality after 65
Regardless of age, sexual activity can provide a sense of comfort and elicit a positive emotional and physical response.1 Hillman2 defined human sexuality as any combination of sexual behavior, emotional intimacy, and sense of sexual identity.
Sexuality in the aging population generally is an understudied area, obscured by the myth of “geriatric asexuality” and subject to numerous psychosocial variables.1 Previous research, focused on a biological perspective of sexuality, has largely overlooked psychological and social influences.3 It has been assumed that, with age, physical and hormonal changes or chronic illness ordinarily reduce or eliminate sexual desire and sexual behavior.3 However, the majority of older adults (defined as age ≥65) report a moderate-to-high level of sexual interest well into late life.1,3
Sexual function remains a subject often neglected in psychiatry. Sexual dysfunctions, as described in the DSM-5,4 do not include age-related changes in sexual function. In addition to physiological changes, sexual difficulties can result from relationship strain, systemic medical or psychiatric disorders, and sexual side effects of medications.
CASE REPORT
Mr. C, age 71 and married, is being treated for a major depressive episode that followed a course of shingles and persistent postherpetic neuralgia. Medications are: escitalopram, 20 mg/d; pregabalin, 150 mg/d; and ramipril, 5 mg/d. Mr. C is physically active and involved in social activities; he has no substance use history. He attends clinic visits with his wife.
Mr. C reports that despite significant improvement of his depressive and pain symptoms, he now experiences sexual difficulties, which he seems hesitant to discuss in detail. According to his wife, Mr. C appears to lack sexual desire and has difficulty initiating and maintaining an erection. She asks Mr. C’s psychiatrist whether she should stop her estrogen treatment, intended to enhance her sexual function, given that the couple is no longer engaging in sexual intercourse.
Mr. C admits to missing physical intimacy; however, he states, “If I have to make a choice between having sex with my wife and getting this depression out of my head, I’m going to pick getting rid of the depression.” Mrs. C says she is becoming dissatisfied with their marriage and the limited time she and her husband now spend together. Mr. C’s psychiatrist suggests that Mr. C and his wife undergo couples counseling.
Physiological changes with aging
In both women and men, the reproductive system undergoes age-related physiological changes.
Women. In women, the phase of decline in ovarian function and resulting decline in sex steroid production (estradiol and progesterone) is referred to as the climacteric, with menopause being determined retrospectively by the cessation of a menstrual period for 1 year.5
Menopausal symptoms typically occur between age 40 and 58; the average age of menopause is 51.6,7 Both estradiol and progesterone levels decline with menopause, and anovulation and ovarian failure ensue. A more gradual decline of female testosterone levels also occurs with aging, starting in the fourth decade of life.8
Clinical manifestations of menopause include vasomotor symptoms (ie, “hot flushes”), sleep disturbances, anxiety and depressive symptoms, decreased bone mineral density, and increased risk of cardiovascular disease.6,7 Loss of estrogen as well as continued loss of testosterone can result in dyspareunia because of atrophy and decreased vulvar and vaginal lubrication, with sexual excitement achieved less quickly, and a decreased intensity of orgasm.7
Men. Research has shown that testosterone levels are highest in men in the second and third decades, with a subsequent gradual decline.9 Older men with a low testosterone level are described as experiencing “late-onset hypogonadism,” also known by the popularized term “andropause.”10 This is attributed to decreased activity at the testicular and hypothalamic levels.10
Nonetheless, only a small fraction of older men with confirmed androgen deficiency are clinically symptomatic.11,12 Low testosterone is associated with decreased libido; it can hinder morning erections, contribute to erectile dysfunction, and result in erections that require physical stimulation.13
Notably, erectile dysfunction involves several other etiologic factors: psychiatric (eg, relationship difficulties, depression), neurogenic (eg, spinal cord injury), endocrine (eg, hyperprolactinemia), arteriogenic (eg, hypertension, type 2 diabetes mellitus), and drug-induced (eg, antidepressants, antihypertensives).14 A low testosterone level also has been associated with potential cognitive changes, decreased bone mineral density, metabolic syndrome (eg, increased risk of type 2 diabetes mellitus), and cardiovascular mortality.10
Effects on sexual activity. How much age-related physiological changes impact sexual practices in the geriatric population is uncertain. A study following 3,302 women through menopause over 6 years found some decline in sexual activity; however, this decline was not associated with increased sexual pain, decreased desire, or lack of arousal.15 Men continue to find ways to remain sexually active despite physiological changes (eg, erectile difficulties), but the etiology of sexual dysfunction in later life remains multi-modal, involving physical, psychological, and relational factors.16,17
Sexual practices in older adults
Researchers for the National Social Life, Health, and Aging Project (NSHAP) have examined sexual activities, behaviors, and problems in >3,000 older community-dwelling men and women across the United States, using information collected from in-home interviews.18 This study found that sexual activity, defined as any mutually voluntary sexual contact with another person, declines with age; however, even in the oldest age group (age 75 to 85), 39% of men and 17% of women reported being sexually active in the last 12 months. Among these persons, 54% reported sexual activity at least 2 times per month; 23% reported having sex at least once a week; and 32% reported engaging in oral sex. Partner availability predicted sexual activity.
Respondents with self-reported poor physical health were more likely to experience sexual problems (eg, difficulty with erection or lubrication, dyspareunia, and lack of pleasure). The most commonly reported reason for sexual inactivity in those with a spouse or other intimate partner was the male partner’s poor physical health.18
A longitudinal study, part of the Women’s Healthy Ageing Project, examined changes in sexual function at late menopause compared with early menopause. Although the researchers also found an age-related decrease in sexual activity, 50% of their late-menopause respondents (mean age, 70; range, 64 to 77) still reported sexual activity in the previous month, with 35% of this subgroup reporting sexual activity at least once a week; 83% reported sexual thoughts or fantasies.19 Availability of a partner, absence of a history of depression, moderate (compared with no) alcohol consumption, and better cognitive function were significantly associated with a higher level of sexual activity.19
In the Successful Aging Evaluation study, conducted in San Diego County, California, community-dwelling older partnered adults age 50 to 99 (mean age, 75) were surveyed about their sexual health after a cognitive screen by telephone20; rating scales for depression, anxiety, and physical function also were included. Results included 41% of men and 35% of women reporting sexual activity at least once a week, and only 21% of men and 24% of women reporting no sexual activity in the previous year. Depressive symptoms were most highly correlated with lack of sexual activity.20
Overall, these studies reveal that positive physical and mental health, access to a healthy partner, and a positive attitude toward sex are correlated with sexual activity in later life, whereas barriers to sexual activity include lack of a healthy sexual partner, depression, and chronic systemic medical illnesses, such as coronary artery disease or type 2 diabetes mellitus.13,17,21-24 Sexual activity and satisfaction have been positively associated with perceived general well-being and self-esteem.25,26 Conversely, sexual difficulties secondary to disease can be a source of distress for couples.27
Possibly overlooked? It is important to note that sexuality itself is a subjective area. Psychological intimacy is a universal phenomenon, and its physical expression may evolve as couples accommodate to age-related bodily changes. Means of achieving physical closeness, other than intercourse (eg, intimate touching, hand holding, kissing, or even acts of caretaking), may not be adequately captured in studies that look specifically at sexual activity.
Taking a sexual history in a geriatric patient
Because sexuality can be an uncomfortable topic for geriatric patients to discuss, sexual problems in this population often go unrecognized. It has been suggested that psychiatrists are more likely to inquire about sexual activity in middle-aged patients than geriatric patients with the same psychiatric presentation—perhaps illustrating a bias against taking a sexual history from a geriatric patient.28 However, because many older patients can experience depression or anxiety disorders in relation to normal sexual changes or sexual dysfunction within the context of their intimate relationships, it is essential to bring these issues to light.
Although a sexual history may not be the focus of a first clinical encounter, consider making such an assessment at a relatively early stage of patient care. The importance of such dialogue is 2-fold:
• It demonstrates to the patient that talking about sexuality in a respectful and empathic manner is appropriate and can encourage patients to communicate more effectively about sexuality with clinicians and with sexual partners.
• It helps elicit medical information needed to make an accurate diagnosis and provide adequate management.
How to begin. As a starting point to taking a sexual history, an open-ended invitation for the geriatric patient to share information may be best, such as “What would you like to tell me about your sexual life?” See further suggestions (Table 1) and examples of more detailed questions to ask once a dialogue has been initiated (Table 2). Additional factors that may contribute to sexual dysfunction are presented in Table 3.1,27,29,30
CASE CONTINUED
In Mr. C’s case, an assessment of his sexual history, including risk factors for sexual dysfunction, is completed. Results from laboratory investigations, including a total testosterone level, are within normal limits.
Mr. C asks about using medications with fewer sexual side effects (he has been taking 3 medications that can contribute to sexual dysfunction). A gradual cross-taper of escitalopram, 20 mg/d, to mirtazapine, 45 mg/d, is implemented, along with tapering pregabalin to 50 mg/d.
Mr. C’s psychiatric and pain symptom improvement is maintained. He notices a boost in his sexual desire but has minimal improvement in erectile dysfunction. He is encouraged to speak with his primary care physician about an antihypertensive agent with less impact on sexual function, as well as therapeutic agents for erectile dysfunction; these, he declines.
At a subsequent visit, Mr. C reports feeling less apprehension about sexual performance. He is now willing to consider further medication options with his primary care physician, and agrees to a recommendation for couples psychotherapy.
As illustrated in Mr. C’s case, the recommended sexual assessment and management strategies to consider at a minimum in psychiatric practice are listed in Table 4.
STI risk in geriatric patients
The risk of sexually transmitted infections (STIs), including human immunodeficiency virus (HIV), often is overlooked in sexually active older adults. Although STIs are more common among younger adults, there is recent evidence of increased incidence in the geriatric population31 (with the highest risk of incident HIV and some STIs in older men who have sex with men32). These increased rates can be explained, at least in part, by:
• older men being less likely to use a condom during sexual activity
• promotion of viral entry in older women through a drier, thinner vaginal wall
• increased longevity of HIV-positive persons.31
Routine STI screening is not warranted in all older adults, but education and prevention strategies in sexually active seniors who are at greater risk of STIs are recommended. Particularly, clinicians should seek opportunities to discuss risk factors and safe sex practices (eg, using condoms, limiting number of sexual partners, practicing good hygiene, engaging in preventive care), and provide behavioral counseling where appropriate.31,33
Additional considerations in geriatric sexuality
Because psychiatric and systemic medical conditions can hinder sexual function, it is essential to identify and manage these conditions. Several neuropsychiatric disorders, including mood and neurocognitive disorders, can not only cause sexual dysfunction, but also can raise ethical dilemmas for clinicians, such as reduced decisional capacity in cognitively impaired patients to consent to sexual activity.1,34
In some patients, psychological, environmental, and pharmacological treatment options may help. A phosphodiesterase type 5 inhibitor for erectile dysfunction can be prescribed by the primary care physician, a psychiatrist, or another specialist, depending on the physician’s expertise and comfort level.
Sequencing of sexual dysfunction. Notably, there is a common paradox in mood disorders. Decreased sexual interest or performance may represent an aspect of anhedonia associated with depression, whereas sexual dysfunction could also result from medication use (particularly that of serotonergic antidepressants, such as selective serotonin reuptake inhibitors and serotonin-norepinephrine inhibitors), even as other depressive symptoms improve. Therefore, it is critical to analyze sequencing of sexual dysfunction—as part of the presenting mood symptoms or dysfunction induced by antidepressant treatment.
Geriatric sexuality in the digital age. Because older adults represent a rapidly growing segment of digital device users,35 Internet use is likely to play a role in the future of sexuality and “digital intimacy,” in that older adults can engage in online sexual activities. The Internet also can be a tool to access medical education.
Related Resources
• Burghardt KJ, Gardner KN. Sildenafil for SSRI-induced sexual dysfunction. Current Psychiatry. 2013;12(4):29-32,A.
• Maciel M, Laganà L. Older women’s sexual desire problems: biopsychosocial factors impacting them and barriers to their clinical assessment [published online January 5, 2014]. Biomed Res Int. 2014;2014:107217. doi: 10.1155/2014/107217.
Drug Brand Names
Bupropion • Wellbutrin, Zyban Mirtazapine • Remeron
Carbamazepine • Tegretol Oxcarbazepine • Trileptal
Clonidine • Catapres Phenobarbital • Luminal
Donepezil • Aricept Phenytoin • Dilantin
Escitalopram • Lexapro Pregabalin • Lyrica
Gabapentin • Neurontin Ramipril • Altace
Lamotrigine • Lamictal Rivastigmine • Exelon
Lithium • Eskalith, Lithobid Trazodone • Desyrel
Memantine • Namenda Valproic acid • Depakote
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Jagus CE, Benbow SM. Sexuality in older men with mental health problems. Sex Relation Ther. 2002;17(3):271-279.
2. Hillman JL. Clinical perspectives on elderly sexuality. New York, NY: Springer; 2000.
3. DeLamater JD, Sill M. Sexual desire in later life. J Sex Res. 2005;42(2):138-149.
4. Diagnostic and statistical manual of mental disorders. 5th ed. Washington, DC: American Psychiatric Association; 2013.
5. Laufer LR, Gambone JC. Climacteric: menopause and peri-and postmenopause. In: Hacker NF, Gambone JC, Hobel CJ. Hacker and Moore’s essentials of obstetrics and gynecology. 5th ed. Philadelphia, PA: Saunders/Elsevier; 2010:379-385.
6. Wilson MM. Menopause. Clin Geriatr Med. 2003;19(3): 483-506.
7. Reid R, Abramson BL, Blake J, et al. Managing menopause. J Obstet Gynaecol Can. 2014;36(9):830-838.
8. Horstman AM, Dillon EL, Urban RJ, et al. The role of androgens and estrogens on healthy aging and longevity. J Gerontol A Biol Sci Med Sci. 2012;67(11):1140-1152.
9. Wu FC, Tajar A, Pye SR, et al. Hypothalamic-pituitary-testicular axis disruptions in older men are differentially linked to age and modifiable risk factors: the European Male Aging Study. J Clin Endocrinol Metab. 2008;93(7):2737-2745.
10. Basaria S. Reproductive aging in men. Endocrinol Metab Clin North Am. 2013;42(2):255-270.
11. Wu FC, Tajar A, Beynon JM, et al. Identification of late-onset hypogonadism in middle-aged and elderly men. N Engl J Med. 2010;363(2):123-135.
12. Araujo AB, Esche GR, Kupelian V, et al. Prevalence of symptomatic androgen deficiency in men. J Clin Endocrinol Metab. 2007;92(11):4241-4247.
13. Lochlainn MN, Kenny RA. Sexual activity and aging. J Am Med Dir Assoc. 2013;14(8):565-572.
14. McMahon CG. Erectile dysfunction. Intern Med J. 2014;44(1):18-26.
15. Avis NE, Brockwell S, Randolph JF Jr, et al. Longitudinal changes in sexual functioning as women transition through menopause: results from the Study of Women’s Health Across the Nation. Menopause. 2009;16(3):442-452.
16. Perelman M, Shabsigh R, Seftel A, et al. Attitudes of men with erectile dysfunction: a cross-national survey. J Sex Med. 2005;2(3):397-406.
17. Corona G, Rastrelli G, Maseroli E, et al. Sexual function of the ageing male. Best Pract Res Clin Endocrinol Metab. 2013;27(4):581-601.
18. Lindau ST, Schumm LP, Laumann EO, et al. A study of sexuality and health among older adults in the United States. N Engl J Med. 2007;357(8):762-774.
19. Lonnèe-Hoffmann RA, Dennerstein L, Lehert P, et al. Sexual function in the late postmenopause: a decade of follow-up in a population-based cohort of Australian women. J Sex Med. 2014;11(8):2029-2038.
20. Wang V, Depp CA, Ceglowski J, et al. Sexual health and function in later life: a population-based study of 606 older adults with a partner. Am J Geriatr Psychiatry. 2015;23(3):227-233.
21. Garrett D. Psychosocial barriers to sexual intimacy for older people. Br J Nurs. 2014;23(6):327-331.
22. DeLamater J, Karraker A. Sexual functioning in older adults. Curr Psychiatry Rep. 2009;11(1):6-11.
23. DeLamater J. Sexual expression in later life: a review and synthesis. J Sex Res. 2012;49(2-3):125-141.
24. Inelmen EM, Sergi G, Girardi A, et al. The importance of sexual health in the elderly: breaking down barriers and taboos. Aging Clin Exp Res. 2012;24(suppl 3):31-34.
25. Choi KB, Jang SH, Lee MY, et al. Sexual life and self-esteem in married elderly. Arch Gerontol Geriatr. 2011;53(1):e17-e20.
26. Davison SL, Bell RJ, LaChina M, et al. The relationship between self-reported sexual satisfaction and general well-being in women. J Sex Med. 2009;6(10):2690-2697.
27. Morley JE, Tariq SH. Sexuality and disease. Clin Geriatr Med. 2003;19(3):563-573.
28. Bouman WP, Arcelus J. Are psychiatrists guilty of “ageism” when it comes to taking a sexual history? Int J Geriatr Psychiatry. 2001;16(1):27-31.
29. La Torre A, Giupponi G, Duffy DM, et al. Sexual dysfunction related to psychotropic drugs: a critical review. Part III: mood stabilizers and anxiolytic drugs. Pharmacopsychiatry. 2014;47(1):1-6.
30. Tucker I. Management of inappropriate sexual behaviors in dementia: a literature review. Int Psychogeriatr. 2010; 22(5):683-692.
31. Imparato T, Sanders D. STD prevalence demands clinical awareness. Aging Well. 2012;5(1):14.
32. Poynten IM, Grulich AE, Templeton DJ. Sexually transmitted infections in older populations. Curr Opin Infect Dis. 2013;26(1):80-85.
33. Talashek ML, Tichy AM, Epping H. Sexually transmitted diseases in the elderly—issues and recommendations. J Gerontol Nurs. 1990;16(4):33-40.
34. Benbow SM, Jagus CE. Sexuality in older women with mental health problems. Sex Relation Ther. 2002;17(3):261-270.
35. Veenhof B, Timusk P. Online activities of Canadian boomers and seniors. http://www.statcan.gc.ca/pub/ 11-008-x/2009002/article/10910-eng.htm#tphp. Accessed March 26, 2015.
1. Jagus CE, Benbow SM. Sexuality in older men with mental health problems. Sex Relation Ther. 2002;17(3):271-279.
2. Hillman JL. Clinical perspectives on elderly sexuality. New York, NY: Springer; 2000.
3. DeLamater JD, Sill M. Sexual desire in later life. J Sex Res. 2005;42(2):138-149.
4. Diagnostic and statistical manual of mental disorders. 5th ed. Washington, DC: American Psychiatric Association; 2013.
5. Laufer LR, Gambone JC. Climacteric: menopause and peri-and postmenopause. In: Hacker NF, Gambone JC, Hobel CJ. Hacker and Moore’s essentials of obstetrics and gynecology. 5th ed. Philadelphia, PA: Saunders/Elsevier; 2010:379-385.
6. Wilson MM. Menopause. Clin Geriatr Med. 2003;19(3): 483-506.
7. Reid R, Abramson BL, Blake J, et al. Managing menopause. J Obstet Gynaecol Can. 2014;36(9):830-838.
8. Horstman AM, Dillon EL, Urban RJ, et al. The role of androgens and estrogens on healthy aging and longevity. J Gerontol A Biol Sci Med Sci. 2012;67(11):1140-1152.
9. Wu FC, Tajar A, Pye SR, et al. Hypothalamic-pituitary-testicular axis disruptions in older men are differentially linked to age and modifiable risk factors: the European Male Aging Study. J Clin Endocrinol Metab. 2008;93(7):2737-2745.
10. Basaria S. Reproductive aging in men. Endocrinol Metab Clin North Am. 2013;42(2):255-270.
11. Wu FC, Tajar A, Beynon JM, et al. Identification of late-onset hypogonadism in middle-aged and elderly men. N Engl J Med. 2010;363(2):123-135.
12. Araujo AB, Esche GR, Kupelian V, et al. Prevalence of symptomatic androgen deficiency in men. J Clin Endocrinol Metab. 2007;92(11):4241-4247.
13. Lochlainn MN, Kenny RA. Sexual activity and aging. J Am Med Dir Assoc. 2013;14(8):565-572.
14. McMahon CG. Erectile dysfunction. Intern Med J. 2014;44(1):18-26.
15. Avis NE, Brockwell S, Randolph JF Jr, et al. Longitudinal changes in sexual functioning as women transition through menopause: results from the Study of Women’s Health Across the Nation. Menopause. 2009;16(3):442-452.
16. Perelman M, Shabsigh R, Seftel A, et al. Attitudes of men with erectile dysfunction: a cross-national survey. J Sex Med. 2005;2(3):397-406.
17. Corona G, Rastrelli G, Maseroli E, et al. Sexual function of the ageing male. Best Pract Res Clin Endocrinol Metab. 2013;27(4):581-601.
18. Lindau ST, Schumm LP, Laumann EO, et al. A study of sexuality and health among older adults in the United States. N Engl J Med. 2007;357(8):762-774.
19. Lonnèe-Hoffmann RA, Dennerstein L, Lehert P, et al. Sexual function in the late postmenopause: a decade of follow-up in a population-based cohort of Australian women. J Sex Med. 2014;11(8):2029-2038.
20. Wang V, Depp CA, Ceglowski J, et al. Sexual health and function in later life: a population-based study of 606 older adults with a partner. Am J Geriatr Psychiatry. 2015;23(3):227-233.
21. Garrett D. Psychosocial barriers to sexual intimacy for older people. Br J Nurs. 2014;23(6):327-331.
22. DeLamater J, Karraker A. Sexual functioning in older adults. Curr Psychiatry Rep. 2009;11(1):6-11.
23. DeLamater J. Sexual expression in later life: a review and synthesis. J Sex Res. 2012;49(2-3):125-141.
24. Inelmen EM, Sergi G, Girardi A, et al. The importance of sexual health in the elderly: breaking down barriers and taboos. Aging Clin Exp Res. 2012;24(suppl 3):31-34.
25. Choi KB, Jang SH, Lee MY, et al. Sexual life and self-esteem in married elderly. Arch Gerontol Geriatr. 2011;53(1):e17-e20.
26. Davison SL, Bell RJ, LaChina M, et al. The relationship between self-reported sexual satisfaction and general well-being in women. J Sex Med. 2009;6(10):2690-2697.
27. Morley JE, Tariq SH. Sexuality and disease. Clin Geriatr Med. 2003;19(3):563-573.
28. Bouman WP, Arcelus J. Are psychiatrists guilty of “ageism” when it comes to taking a sexual history? Int J Geriatr Psychiatry. 2001;16(1):27-31.
29. La Torre A, Giupponi G, Duffy DM, et al. Sexual dysfunction related to psychotropic drugs: a critical review. Part III: mood stabilizers and anxiolytic drugs. Pharmacopsychiatry. 2014;47(1):1-6.
30. Tucker I. Management of inappropriate sexual behaviors in dementia: a literature review. Int Psychogeriatr. 2010; 22(5):683-692.
31. Imparato T, Sanders D. STD prevalence demands clinical awareness. Aging Well. 2012;5(1):14.
32. Poynten IM, Grulich AE, Templeton DJ. Sexually transmitted infections in older populations. Curr Opin Infect Dis. 2013;26(1):80-85.
33. Talashek ML, Tichy AM, Epping H. Sexually transmitted diseases in the elderly—issues and recommendations. J Gerontol Nurs. 1990;16(4):33-40.
34. Benbow SM, Jagus CE. Sexuality in older women with mental health problems. Sex Relation Ther. 2002;17(3):261-270.
35. Veenhof B, Timusk P. Online activities of Canadian boomers and seniors. http://www.statcan.gc.ca/pub/ 11-008-x/2009002/article/10910-eng.htm#tphp. Accessed March 26, 2015.
Should lithium and ECT be used concurrently in geriatric patients?
Delirium has been described as a potential complication of concurrent lithium and electroconvulsive therapy (ECT) for depression, in association with a range of serum lithium levels. Although debate persists about the safety of continuing previously established lithium therapy during a course of ECT for mood symptoms, withholding lithium for 24 hours before administering ECT and measuring the serum lithium level before ECT were found to decrease the risk of post-ECT neurocognitive effects.1
We have found that the conventional practice of holding lithium for 24 hours before ECT might need to be re-evaluated in geriatric patients, as the following case demonstrates. Only 24 hours of holding lithium therapy might result in a lithium level sufficient to contribute to delirium after ECT.
CASE REPORT
An older woman with recurrent unipolar psychotic depression
Mrs. A, age 81, was admitted to the hospital with a 1-week history of depressed mood, anhedonia, insomnia, anergia, anorexia, and nihilistic somatic delusions that her organs were “rotting and shutting down.” Treatment included nortriptyline, 40 mg/d; lithium, 150 mg/d; and haloperidol, 0.5 mg/d. Her serum lithium level was 0.3 mEq/L (reference range, 0.6 to 1.2 mEq/L); the serum nortriptyline level was 68 ng/mL (reference range, 50 to 150 ng/mL). CT of the head and an electrocardiogram were unremarkable.
A twice-weekly course of ECT was initiated.
The day before Treatment 1 of ECT, the serum lithium level (drawn 12 hours after the last dose) was 0.4 mEq/L. Lithium was withheld 24 hours before ECT; nortriptyline and haloperidol were continued at prescribed dosages.
Right unilateral stimulation was used at 50%/mC energy (Thymatron DG, with methohexital anesthesia, and succinylcholine for muscle relaxation). Seizure duration, measured by EEG, was 57 seconds.
Mrs. A developed postictal delirium after the first 2 ECT sessions. The serum lithium level was unchanged. Subsequently, lithium treatment was discontinued and ECT was continued; once lithium was stopped, delirium resolved. ECT sessions 3 and 4 were uneventful, with no post-treatment delirium. Seizure duration for Treatment 4 was 58 seconds. She started breathing easily after all ECT sessions.
After Treatment 4, Mrs. A experienced full remission of depressive and psychotic symptoms. Repeat CT of head, after Treatment 4, was unchanged from baseline.
What is the role of lithium?
Mrs. A did not exhibit typical signs of lithium intoxication (diarrhea, vomiting, tremor). Notably, lithium has an intrinsic anticholinergic activity2; concurrent nortriptyline, a secondary amine tricyclic antidepressant with fewer anticholinergic side effects than other tricyclics,2 could precipitate delirium in a vulnerable patient secondary to excessive cumulative anticholinergic exposure.
No prolonged time-to-respiration or time-to-awakening occurred during treatments in which concurrent lithium and ECT were used; seizure duration with and without concurrent lithium was relatively similar.
There are potential complications of concurrent use of lithium and ECT:
• prolongation of the duration of muscle paralysis and apnea induced by commonly used neuromuscular-blocking agents (eg, succinylcholine)
• post-ECT cognitive disturbance.1,3,4
There is debate about the safety of continuing lithium during, or in close proximity to, ECT. In a case series of 12 patients who underwent combined lithium therapy and ECT, the authors concluded that this combination can be safe, regardless of age, as long as appropriate clinical monitoring is provided.4 In Mrs. A’s case, once post-ECT delirium was noted, lithium was discontinued for subsequent ECT sessions.
Because further ECT was uneventful without lithium, and no other clear acute cause of delirium could be identified, we concluded that lithium likely played a role in Mrs. A’s delirium. Notably, nortriptyline had been continued, suggesting that the degree of anticholinergic blockade provided by nortriptyline was insufficient to provoke delirium post-ECT in the absence of potentiation of this effect, as it had been when lithium also was used initially.
Guidelines for dosing and serum lithium concentrations in geriatric patients are not well-established; the current traditional range of 0.6 to 1.2 mEq/L, is too high for geriatric patients and can result in episodes of lithium toxicity, including delirium.5 Although our patient’s lithium level was below the reference range for all patients, a level of 0.3 mEq/L can be considered at the low end of the reference range for geriatric patients.5 Inasmuch as the lithium-assisted post-ECT delirium could represent a clinical sign of lithium toxicity, perhaps even a subtherapeutic level in a certain patient could be paradoxically “toxic.”
Although the serum lithium level in our patient remained below the toxic level for the general population (>1.5 mEq/L), delirium in a geriatric patient could result from:
• age-related changes in the pharmacokinetics of lithium, a water-soluble drug; these changes reduce renal clearance of the drug and extend plasma elimination half-life of a single dose to 36 hours, with the result that lithium remains in the body longer and necessitating a lower dosage (ie, a dosage that yields a serum level of approximately 0.5 mEq/L)
• the CNS tissue concentration of lithium, which can be high even though the serum level is not toxic
• an age-related increase in blood-brain barrier permeability, making the barrier more porous for drugs
• changes in blood-brain barrier permeability by post-ECT biochemical induction, with subsequent increased drug availability in the CNS.5,6
What we recommend
Possible interactions between lithium and ECT that lead to ECT-associated delirium need further elucidation, but discontinuing lithium during the course of ECT in a geriatric patient warrants your consideration. Following a safe interval after the last ECT session, lithium likely can be safely re-introduced 1) if there is clinical need and 2) as long as clinical surveillance for cognitive side effects is provided— especially if ECT will need to be reconsidered in the future.
Two additional considerations:
• Actively reassess lithium dosing in all geriatric psychiatric patients, especially those with renal insufficiency and other systemic metabolic considerations.
• Actively examine the use of all other anticholinergic agents in the course of evaluating a patient’s candidacy for ECT.
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. American Psychiatric Association. The practice of electroconvulsive therapy: recommendations for treatment, training, and privileging. A task force report of the American Psychiatric Association. 2nd ed. Washington, DC: American Psychiatric Publishing; 2001.
2. Chew ML, Mulsant BH, Pollock BG, et al. Anticholinergic activity of 107 medications commonly used by older adults. J Am Geriatr Soc. 2008;56(7):1333-1341.
3. Hill GE, Wong KC, Hodges MR. Potentiation of succinylcholine neuromuscular blockade by lithium carbonate. Anesthesiology. 1976;44(5):439-442.
4. Dolenc TJ, Rasmussen KG. The safety of electroconvulsive therapy and lithium in combination: a case series and review of the literature. J ECT. 2005;21(3):165-170.
5. Shulman KI. Lithium for older adults with bipolar disorder: should it still be considered a first line agent? Drugs Aging. 2010;27(8):607-615.
6. Grandjean EM, Aubry JM. Lithium: updated human knowledge using an evidence-based approach. Part II: clinical pharmacology and therapeutic monitoring. CNS Drugs. 2009;23(4):331-349.
Delirium has been described as a potential complication of concurrent lithium and electroconvulsive therapy (ECT) for depression, in association with a range of serum lithium levels. Although debate persists about the safety of continuing previously established lithium therapy during a course of ECT for mood symptoms, withholding lithium for 24 hours before administering ECT and measuring the serum lithium level before ECT were found to decrease the risk of post-ECT neurocognitive effects.1
We have found that the conventional practice of holding lithium for 24 hours before ECT might need to be re-evaluated in geriatric patients, as the following case demonstrates. Only 24 hours of holding lithium therapy might result in a lithium level sufficient to contribute to delirium after ECT.
CASE REPORT
An older woman with recurrent unipolar psychotic depression
Mrs. A, age 81, was admitted to the hospital with a 1-week history of depressed mood, anhedonia, insomnia, anergia, anorexia, and nihilistic somatic delusions that her organs were “rotting and shutting down.” Treatment included nortriptyline, 40 mg/d; lithium, 150 mg/d; and haloperidol, 0.5 mg/d. Her serum lithium level was 0.3 mEq/L (reference range, 0.6 to 1.2 mEq/L); the serum nortriptyline level was 68 ng/mL (reference range, 50 to 150 ng/mL). CT of the head and an electrocardiogram were unremarkable.
A twice-weekly course of ECT was initiated.
The day before Treatment 1 of ECT, the serum lithium level (drawn 12 hours after the last dose) was 0.4 mEq/L. Lithium was withheld 24 hours before ECT; nortriptyline and haloperidol were continued at prescribed dosages.
Right unilateral stimulation was used at 50%/mC energy (Thymatron DG, with methohexital anesthesia, and succinylcholine for muscle relaxation). Seizure duration, measured by EEG, was 57 seconds.
Mrs. A developed postictal delirium after the first 2 ECT sessions. The serum lithium level was unchanged. Subsequently, lithium treatment was discontinued and ECT was continued; once lithium was stopped, delirium resolved. ECT sessions 3 and 4 were uneventful, with no post-treatment delirium. Seizure duration for Treatment 4 was 58 seconds. She started breathing easily after all ECT sessions.
After Treatment 4, Mrs. A experienced full remission of depressive and psychotic symptoms. Repeat CT of head, after Treatment 4, was unchanged from baseline.
What is the role of lithium?
Mrs. A did not exhibit typical signs of lithium intoxication (diarrhea, vomiting, tremor). Notably, lithium has an intrinsic anticholinergic activity2; concurrent nortriptyline, a secondary amine tricyclic antidepressant with fewer anticholinergic side effects than other tricyclics,2 could precipitate delirium in a vulnerable patient secondary to excessive cumulative anticholinergic exposure.
No prolonged time-to-respiration or time-to-awakening occurred during treatments in which concurrent lithium and ECT were used; seizure duration with and without concurrent lithium was relatively similar.
There are potential complications of concurrent use of lithium and ECT:
• prolongation of the duration of muscle paralysis and apnea induced by commonly used neuromuscular-blocking agents (eg, succinylcholine)
• post-ECT cognitive disturbance.1,3,4
There is debate about the safety of continuing lithium during, or in close proximity to, ECT. In a case series of 12 patients who underwent combined lithium therapy and ECT, the authors concluded that this combination can be safe, regardless of age, as long as appropriate clinical monitoring is provided.4 In Mrs. A’s case, once post-ECT delirium was noted, lithium was discontinued for subsequent ECT sessions.
Because further ECT was uneventful without lithium, and no other clear acute cause of delirium could be identified, we concluded that lithium likely played a role in Mrs. A’s delirium. Notably, nortriptyline had been continued, suggesting that the degree of anticholinergic blockade provided by nortriptyline was insufficient to provoke delirium post-ECT in the absence of potentiation of this effect, as it had been when lithium also was used initially.
Guidelines for dosing and serum lithium concentrations in geriatric patients are not well-established; the current traditional range of 0.6 to 1.2 mEq/L, is too high for geriatric patients and can result in episodes of lithium toxicity, including delirium.5 Although our patient’s lithium level was below the reference range for all patients, a level of 0.3 mEq/L can be considered at the low end of the reference range for geriatric patients.5 Inasmuch as the lithium-assisted post-ECT delirium could represent a clinical sign of lithium toxicity, perhaps even a subtherapeutic level in a certain patient could be paradoxically “toxic.”
Although the serum lithium level in our patient remained below the toxic level for the general population (>1.5 mEq/L), delirium in a geriatric patient could result from:
• age-related changes in the pharmacokinetics of lithium, a water-soluble drug; these changes reduce renal clearance of the drug and extend plasma elimination half-life of a single dose to 36 hours, with the result that lithium remains in the body longer and necessitating a lower dosage (ie, a dosage that yields a serum level of approximately 0.5 mEq/L)
• the CNS tissue concentration of lithium, which can be high even though the serum level is not toxic
• an age-related increase in blood-brain barrier permeability, making the barrier more porous for drugs
• changes in blood-brain barrier permeability by post-ECT biochemical induction, with subsequent increased drug availability in the CNS.5,6
What we recommend
Possible interactions between lithium and ECT that lead to ECT-associated delirium need further elucidation, but discontinuing lithium during the course of ECT in a geriatric patient warrants your consideration. Following a safe interval after the last ECT session, lithium likely can be safely re-introduced 1) if there is clinical need and 2) as long as clinical surveillance for cognitive side effects is provided— especially if ECT will need to be reconsidered in the future.
Two additional considerations:
• Actively reassess lithium dosing in all geriatric psychiatric patients, especially those with renal insufficiency and other systemic metabolic considerations.
• Actively examine the use of all other anticholinergic agents in the course of evaluating a patient’s candidacy for ECT.
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Delirium has been described as a potential complication of concurrent lithium and electroconvulsive therapy (ECT) for depression, in association with a range of serum lithium levels. Although debate persists about the safety of continuing previously established lithium therapy during a course of ECT for mood symptoms, withholding lithium for 24 hours before administering ECT and measuring the serum lithium level before ECT were found to decrease the risk of post-ECT neurocognitive effects.1
We have found that the conventional practice of holding lithium for 24 hours before ECT might need to be re-evaluated in geriatric patients, as the following case demonstrates. Only 24 hours of holding lithium therapy might result in a lithium level sufficient to contribute to delirium after ECT.
CASE REPORT
An older woman with recurrent unipolar psychotic depression
Mrs. A, age 81, was admitted to the hospital with a 1-week history of depressed mood, anhedonia, insomnia, anergia, anorexia, and nihilistic somatic delusions that her organs were “rotting and shutting down.” Treatment included nortriptyline, 40 mg/d; lithium, 150 mg/d; and haloperidol, 0.5 mg/d. Her serum lithium level was 0.3 mEq/L (reference range, 0.6 to 1.2 mEq/L); the serum nortriptyline level was 68 ng/mL (reference range, 50 to 150 ng/mL). CT of the head and an electrocardiogram were unremarkable.
A twice-weekly course of ECT was initiated.
The day before Treatment 1 of ECT, the serum lithium level (drawn 12 hours after the last dose) was 0.4 mEq/L. Lithium was withheld 24 hours before ECT; nortriptyline and haloperidol were continued at prescribed dosages.
Right unilateral stimulation was used at 50%/mC energy (Thymatron DG, with methohexital anesthesia, and succinylcholine for muscle relaxation). Seizure duration, measured by EEG, was 57 seconds.
Mrs. A developed postictal delirium after the first 2 ECT sessions. The serum lithium level was unchanged. Subsequently, lithium treatment was discontinued and ECT was continued; once lithium was stopped, delirium resolved. ECT sessions 3 and 4 were uneventful, with no post-treatment delirium. Seizure duration for Treatment 4 was 58 seconds. She started breathing easily after all ECT sessions.
After Treatment 4, Mrs. A experienced full remission of depressive and psychotic symptoms. Repeat CT of head, after Treatment 4, was unchanged from baseline.
What is the role of lithium?
Mrs. A did not exhibit typical signs of lithium intoxication (diarrhea, vomiting, tremor). Notably, lithium has an intrinsic anticholinergic activity2; concurrent nortriptyline, a secondary amine tricyclic antidepressant with fewer anticholinergic side effects than other tricyclics,2 could precipitate delirium in a vulnerable patient secondary to excessive cumulative anticholinergic exposure.
No prolonged time-to-respiration or time-to-awakening occurred during treatments in which concurrent lithium and ECT were used; seizure duration with and without concurrent lithium was relatively similar.
There are potential complications of concurrent use of lithium and ECT:
• prolongation of the duration of muscle paralysis and apnea induced by commonly used neuromuscular-blocking agents (eg, succinylcholine)
• post-ECT cognitive disturbance.1,3,4
There is debate about the safety of continuing lithium during, or in close proximity to, ECT. In a case series of 12 patients who underwent combined lithium therapy and ECT, the authors concluded that this combination can be safe, regardless of age, as long as appropriate clinical monitoring is provided.4 In Mrs. A’s case, once post-ECT delirium was noted, lithium was discontinued for subsequent ECT sessions.
Because further ECT was uneventful without lithium, and no other clear acute cause of delirium could be identified, we concluded that lithium likely played a role in Mrs. A’s delirium. Notably, nortriptyline had been continued, suggesting that the degree of anticholinergic blockade provided by nortriptyline was insufficient to provoke delirium post-ECT in the absence of potentiation of this effect, as it had been when lithium also was used initially.
Guidelines for dosing and serum lithium concentrations in geriatric patients are not well-established; the current traditional range of 0.6 to 1.2 mEq/L, is too high for geriatric patients and can result in episodes of lithium toxicity, including delirium.5 Although our patient’s lithium level was below the reference range for all patients, a level of 0.3 mEq/L can be considered at the low end of the reference range for geriatric patients.5 Inasmuch as the lithium-assisted post-ECT delirium could represent a clinical sign of lithium toxicity, perhaps even a subtherapeutic level in a certain patient could be paradoxically “toxic.”
Although the serum lithium level in our patient remained below the toxic level for the general population (>1.5 mEq/L), delirium in a geriatric patient could result from:
• age-related changes in the pharmacokinetics of lithium, a water-soluble drug; these changes reduce renal clearance of the drug and extend plasma elimination half-life of a single dose to 36 hours, with the result that lithium remains in the body longer and necessitating a lower dosage (ie, a dosage that yields a serum level of approximately 0.5 mEq/L)
• the CNS tissue concentration of lithium, which can be high even though the serum level is not toxic
• an age-related increase in blood-brain barrier permeability, making the barrier more porous for drugs
• changes in blood-brain barrier permeability by post-ECT biochemical induction, with subsequent increased drug availability in the CNS.5,6
What we recommend
Possible interactions between lithium and ECT that lead to ECT-associated delirium need further elucidation, but discontinuing lithium during the course of ECT in a geriatric patient warrants your consideration. Following a safe interval after the last ECT session, lithium likely can be safely re-introduced 1) if there is clinical need and 2) as long as clinical surveillance for cognitive side effects is provided— especially if ECT will need to be reconsidered in the future.
Two additional considerations:
• Actively reassess lithium dosing in all geriatric psychiatric patients, especially those with renal insufficiency and other systemic metabolic considerations.
• Actively examine the use of all other anticholinergic agents in the course of evaluating a patient’s candidacy for ECT.
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. American Psychiatric Association. The practice of electroconvulsive therapy: recommendations for treatment, training, and privileging. A task force report of the American Psychiatric Association. 2nd ed. Washington, DC: American Psychiatric Publishing; 2001.
2. Chew ML, Mulsant BH, Pollock BG, et al. Anticholinergic activity of 107 medications commonly used by older adults. J Am Geriatr Soc. 2008;56(7):1333-1341.
3. Hill GE, Wong KC, Hodges MR. Potentiation of succinylcholine neuromuscular blockade by lithium carbonate. Anesthesiology. 1976;44(5):439-442.
4. Dolenc TJ, Rasmussen KG. The safety of electroconvulsive therapy and lithium in combination: a case series and review of the literature. J ECT. 2005;21(3):165-170.
5. Shulman KI. Lithium for older adults with bipolar disorder: should it still be considered a first line agent? Drugs Aging. 2010;27(8):607-615.
6. Grandjean EM, Aubry JM. Lithium: updated human knowledge using an evidence-based approach. Part II: clinical pharmacology and therapeutic monitoring. CNS Drugs. 2009;23(4):331-349.
1. American Psychiatric Association. The practice of electroconvulsive therapy: recommendations for treatment, training, and privileging. A task force report of the American Psychiatric Association. 2nd ed. Washington, DC: American Psychiatric Publishing; 2001.
2. Chew ML, Mulsant BH, Pollock BG, et al. Anticholinergic activity of 107 medications commonly used by older adults. J Am Geriatr Soc. 2008;56(7):1333-1341.
3. Hill GE, Wong KC, Hodges MR. Potentiation of succinylcholine neuromuscular blockade by lithium carbonate. Anesthesiology. 1976;44(5):439-442.
4. Dolenc TJ, Rasmussen KG. The safety of electroconvulsive therapy and lithium in combination: a case series and review of the literature. J ECT. 2005;21(3):165-170.
5. Shulman KI. Lithium for older adults with bipolar disorder: should it still be considered a first line agent? Drugs Aging. 2010;27(8):607-615.
6. Grandjean EM, Aubry JM. Lithium: updated human knowledge using an evidence-based approach. Part II: clinical pharmacology and therapeutic monitoring. CNS Drugs. 2009;23(4):331-349.
Delirium in the hospital: Emphasis on the management of geriatric patients
Although delirium has many descriptive terms (Table 1), a common unifying term is “acute global cognitive dysfunction,” now recognized as delirium; a consensus supported by DSM-51 and ICD-102 (Table 2). According to DSM-5, the essential feature is a disturbance of attention or awareness that is accompanied by a change in baseline cognition that cannot be explained by another preexisting, established, or evolving neurocognitive disorder (the newly named DSM-5 entity for dementia syndromes).1 Because delirium affects the cortex diffusely, psychiatric symptoms can include cognitive, mood, anxiety, or psychotic symptoms. Because many systemic illnesses can induce delirium, the differential diagnosis spans all organ systems.
Three subtypes
Delirium can be classified, based on symptoms,3,4 into 3 subtypes: hyperactive-hyperalert, hypoactive-hypoalert, and mixed delirium. Hyperactive patients present with restlessness and agitation. Hypoactive patients are lethargic, confused, slow to respond to questions, and often appear depressed. The differential prognostic significance of these subtypes has been examined in the literature, with conflicting results. Rabinowitz5 reported that hypoactive delirium has the worst prognosis, while Marcantonio et al6 indicated that the hyperactive subtype is associated with the highest mortality rate. Mixed delirium, with periods of both hyperactivity and hypoactivity, is the most common type of delirium.7
A prodromal phase, characterized by anxiety, frequent requests for nursing and medical assistance, decreased attention, restlessness, vivid dreams, disorientation immediately after awakening, and hallucinations, can occur before an episode of full-spectrum delirium; this prodromal state often is identified retrospectively —after the patient is in an episode of delirium.8,9
Evidence-based guidelines aim to improve recognition and clinical management.10-13 Disruptive behavior is the main reason for psychiatric referral in delirium.14,15 Delayed psychiatric consultation because of non-recognition of delirium is related to variables such as older age; history of a pre-existing, comorbid neurocognitive disorder; and the clinical appearance of hypoactive delirium.14
The case of Mr. D (Box),16 illustrates how the emergence of antipsychotic-associated neuroleptic malignant syndrome (NMS) can complicate antipsychotic treatment of delirium in a geriatric medical patient, although delirium also is a common presentation in NMS.17 Delirium developed after an increase in carbidopa/levodopa, which has central dopaminergic effects that can precipitate delirium, particularly in a geriatric patient with preexisting comorbid neurocognitive disorder. Further complicating Mr. D’s delirium presentation was the development of NMS, which had a multifactorial causation, such as the use of dopamine antagonists (ie, quetiapine, metoclopramide), and an abrupt decrease of a dopaminergic agent (ie, carbidopa/levodopa), all inducing a central dopamine relative hypoactivity.
Epidemiology
Delirium is more common in older patients,15 and is seen in 30% to 40% of hospitalized geriatric patients.18 Delirium in older patients, compared with other adults, is associated with more severe cognitive impairment.19 It is common among geriatric surgical patients (15% to 62%)20 with a peak 2 to 5 days postoperatively for hip fracture,21 and often is seen in ICU patients (70% to 87%).20 However, Spronk et al22 found that delirium is significantly under-recognized in the ICU. Nearly 90% of terminally ill patients become delirious before death.23 Terminal delirium often is unrecognized and can interfere with assessment of other clinical problems.24 A preexisting history of comorbid neurocognitive disorder was evident in as many as two-thirds of delirium cases.25
Pathophysiology and risk factors
The pathophysiology of delirium has been characterized as an imbalance of CNS metabolism, including decreased blood flow in various regions of the brain that may normalize once delirium resolves.26 Studies describe the simultaneous decrease of cholinergic transmission and dopaminergic excess.27,28 Predisposing and precipitating factors for delirium that are of particular importance in geriatric patients include:
• advanced age
• CNS disease
• infection
• cognitive impairment
• male sex
• poor nutrition
• dehydration and other metabolic abnormalities
• cardiovascular events
• substance use
• medication
• sensory deprivation (eg, impaired vision or hearing)
• sleep deprivation
• low level of physical activity.27,29,30
Table 3 lists the most common delirium-provocative medications.27
Evaluation and psychometric scales
The EEG can be useful in evaluating delirium, especially in clinically ambiguous cases. EEG findings may indicate generalized slowing or dropout of the posterior dominant rhythm, and generalized slow theta and delta waves, findings that are more common in delirium than in other neurocognitive disorders and other psychiatric illnesses. The EEG must be interpreted in the context of the delirium diagnostic workup, because abnormalities seen in other neurocognitive disorders can overlap with those of delirium.31
The EEG referral should specify the clinical suspicion of delirium to help interpret the results. Delirium cases in which the patient’s previous cognitive status is unknown may benefit from EEG evaluation, such as:
• in possible status epilepticus
• when delirium improvement has reached a plateau at a lower level of cognitive function than before onset of delirium
• when the patient is unable or unwilling to complete a psychiatric interview.27
Assessment instruments are available to diagnose and monitor delirium (Table 4). Typically, delirium assessment includes examining levels of arousal, psychomotor activity, cognition (ie, orientation, attention, and memory), and perceptual disturbances.
Psychometrically, a review of Table 4 suggests that validity appeared stable with adequate specificity (64% to 99%) but more variable sensitivity (36% to 100%). These reliability parameters also will be affected by the classification system (ie, DSM vs ICD) and the cut-off score employed.32 Most measures (eg, Confusion Assessment Method [CAM], CAM-ICU) provide an adequate sample of behavioral (ie, level of alertness), motor (ie, psychomotor activity), and cognitive (ie, orientation, attention, memory, and receptive language) function, with the exception of the Global Attentiveness Rating, which is a 2-minute open conversation protocol between physician and patient.
Some measures are stand-alone instruments, such as the Memorial Delirium Assessment Scale, whereas the CAM requires administration of separate cognitive screens, including the Mini-Mental State Examination (MMSE) and Digit Span.33 Instruments to detect delirium in critically ill patients are a more recent development. Wong et al34 reported that the most widely studied tool was the CAM. Obtaining collateral information from family, caregivers, and hospital staff is essential, particularly given the fluctuating nature of delirium.
Management
Prevention. Identify patients at high risk of delirium so that preventive strategies can be employed. Multi-component, nonpharmacotherapeutic interventions are used in clinical settings but few randomized trials have been conducted. The contributing effectiveness of individual components is not well-studied, but most include staff education to increase awareness of delirium. Of 3 multi-component intervention randomized trials, 2 reported a significantly lower incidence of delirium in the intervention group.35-37 Implementation of a multi-component protocol in medical/ surgical units was associated with a significant reduction in use of restraints.38
As in Mr. D’s case, complex drug regimens, particularly for CNS illness, can increase the risk of delirium. Considering the medication profile for patients with complex systemic illness—in particular, minimizing the use anticholinergics and dopamine agonists— may be crucial in preventing delirium.
Prophylactic administration of antipsychotics may reduce the risk of developing postoperative delirium.39 Studies of the use of these agents were characterized by small sample sizes and selected groups of patient populations. Of the 4 randomized studies evaluating prophylactic antipsychotics (vs placebo), 3 found a lower incidence of delirium in the intervention groups.39-41
A study of haloperidol in post-GI surgery patients showed a reduced occurrence of delirium,40 whereas its prophylactic use in patients undergoing hip surgery42 did not reduce the incidence of delirium compared with placebo, but did decrease severity when delirium occurred.42
Risperidone39 in post-cardiac surgery and olanzapine41 perioperatively in patients undergoing total knee or hip replacement have been shown to decrease delirium severity and duration. Targeted prophylaxis with risperidone43 in post-cardiac surgery patients who showed disturbed cognition but did not meet criteria for delirium reduced the number of patients requiring medication, compared with placebo.43
Dexmedetomidine, an α-2 adrenergic receptor agonist, compared with propofol or midazolam in post-cardiac valve surgery patients, resulted in a decreased incidence of delirium but no difference in delirium duration, hospital length of stay, or use of other medications.44 However, other studies have shown that dexmedetomidine reduces ICU length of stay and duration of mechanical ventilation.45
Treatment. Management of hospitalized medically ill geriatric patients with delirium is challenging and requires a comprehensive approach. The first step in delirium management is prompt identification and management of systemic medical disturbances associated with the delirium episode. First-line, nonpharmacotherapeutic strategies for patients with delirium include:
• reorientation
• behavioral interventions (eg, use of clear instructions and frequent eye contact with patients)
• environmental interventions (eg, minimal noise, adequate lighting, and limited room and staff changes)
• avoidance of physical restraints.46
Consider employing family members or hospital staff sitters to stay with the patient and to reassure, reorient, and watch for agitation and other unsafe behaviors (eg, attempted elopement). Psychoeducation for the patient and family on the phenomenology of delirium can be helpful.
The use of drug treatment strategies should be integrated into a comprehensive approach that includes the routine use of nondrug measures.46 Using medications for treating hypoactive delirium, formerly controversial, now has wider acceptance.47,48 A few high-quality randomized trials have been performed.25,49,50
Pharmacotherapy, especially in frail patients, should be initiated at the lowest starting dosage and titrated cautiously to clinical effect and for the shortest period of time necessary. Antipsychotics are preferred agents for treating all subtypes of delirium; haloperidol is widely used.46,51,52 However, antipsychotics, including haloperidol, can be associated with adverse neurologic effects such as extrapyramidal symptoms (EPS) and NMS.
Although reported less frequently than with haloperidol, other agents have been implicated in development of EPS and NMS, including atypical antipsychotics and antiemetic dopamine antagonists, particularly in parkinsonism-prone patients.53 Strategies that can minimize such risks in geriatric inpatients with delirium include oral, rather than parenteral, use of antipsychotics—preferential use of atypical over typical antipsychotics— and lowest effective dosages.54
In controlled trials, atypical antipsychotics for delirium showed efficacy compared with haloperidol.52,55 However, there is no research that demonstrates any advantage of one atypical over another.25
In Mr. D’s case, the most important intervention for managing delirium caused by NMS is to discontinue all dopamine antagonists and treat agitation with judicious doses of a benzodiazepine, with supportive care.17 In cases of sudden discontinuation or a dosage decrease of dopamine agonists, these medications should be resumed or optimized to minimize the risk of NMS-associated rhabdomyolysis and subsequent renal failure.17 Antipsychotics carry an increased risk of stroke and mortality in older patients with established or evolving neurocognitive disorders56,57 and can cause prolongation of the QTc interval.57
Other medications that could be used for delirium include cholinesterase inhibitors58,59 (although larger trials and a systematic review did not support this use60), and 5-HT receptor antagonists,61 such as trazodone. Benzodiazepines, such as lorazepam, are first-line treatment for delirium associated with seizures or withdrawal from alcohol, sedatives, hypnotics, and anxiolytics and for delirium caused by NMS. Be cautious about using benzodiazepines in geriatric patients because of a risk of respiratory depression, falls, sedation, and amnesia.
Geriatric patients with alcoholism and those with malnutrition are prone to thiamine and vitamin B12 deficiencies, which can induce delirium. Laboratory assessment and consideration of supplementation is recommended. Despite high occurrence of delirium in hospitalized older adults with preexisting comorbid neurocognitive disorders, there is no standard care for delirium comorbid with another neurocognitive disorder.62 Clinical practice guidelines for older patients receiving palliative care have been developed63; the goal is to minimize suffering and discomfort in patients in palliative care.64
Post-delirium prophylaxis. Medications for delirium usually can be tapered and discontinued once the episode has resolved and the patient is stable; it is common to discontinue medications when the patient has been symptom-free for 1 week.65 Some patients (eg, with end-stage liver disease, disseminated cancer) are prone to recurrent or to prolonged or chronic delirium. A period of post-recovery treatment with antipsychotics—even indefinite treatment in some cases—should be considered.
Post-delirium debriefing and aftercare. The psychological complications of delirium are distressing for the patient and his (her) caregivers. Psychiatric complications associated with delirium, including acute stress disorder—which might predict posttraumatic stress disorder—have been explored; early recognition and treatment may improve long-term outcomes.66 After recovery from acute delirium, cognitive assessment (eg, MMSE67 or Montreal Cognitive Assessment68) is recommended to validate current cognitive status because patients may have persistent decrement in cognitive function compared with pre-delirium condition, even after recovery from the acute episode.
Post-delirium debriefing may help patients who have recovered from a delirium episode. Patients may fear that their brief period of hallucinations might represent the onset of a chronic-relapsing psychotic disorder. Allow patients to communicate their distress about the delirium episode and give them the opportunity to talk through the experience. Brief them on the possibility that delirium will recur and advise them to seek emergency medical care in case of recurrence. Advise patients to monitor and maintain a normal sleep-wake cycle.
Family members can watch for syndromal recurrence of delirium. They should be encouraged to discuss their reaction to having seen their relative in a delirious state.
Health care systems with integrated electronic medical records should list “delirium, resolved” on the patient’s illness profile or problem list and alert the patient’s primary care provider to the delirium history to avoid future exposure to delirium-provocative medications, and to prompt the provider to assume an active role in post-delirium care, including delirium recurrence surveillance, medication adjustment, risk factor management, and post-recovery cognitive assessment.
Bottom Line
Evaluation of delirium in geriatric patients includes clinical vigilance and screening, differentiating delirium from other neurocognitive disorders, and identifying and treating underlying causes. Perioperative use of antipsychotics may reduce the incidence of delirium, although hospital length of stay generally has not been reduced with prophylaxis. Management interventions include staff education, systematic screening, use of multicomponent interventions, and pharmacologic interventions.
Related Resources
• Downing LJ, Caprio TV, Lyness JM. Geriatric psychiatry review: differential diagnosis and treatment of the 3 D’s - delirium, dementia, and depression. Curr Psychiatry Rep. 2013;15(6):365.
• Brooks PB. Postoperative delirium in elderly patients. Am J Nurs. 2012;112(9):38-49.
Drug Brand Names
Carbidopa/levodopa • Sinemet Midazolam • Versed
Dexmedetomidine • Precedex Olanzapine • Zyprexa
Haloperidol • Haldol Propofol • Diprivan
Lithium • Eskalith, Lithobid Quetiapine • Seroquel
Lorazepam • Ativan Risperidone • Risperdal
Metoclopramide • Reglan Trazodone • Desyrel
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Diagnostic and statistical manual of mental disorders, fifth edition. Washington, DC: American Psychiatric Association; 2013.
2. World Health Organization. The ICD-10 classification of mental and behavioural disorders. Diagnostic criteria for research. Geneva, Switzerland: WHO; 1993.
3. Lipowski ZJ. Delirium in the elderly patient. N Engl J Med. 1989;320(9):578-582.
4. Meagher DJ, Trzepacz PT. Motoric subtypes of delirium. Semin Clin Neuropsychiatry. 2000;5(2):75-85.
5. Rabinowitz T. Delirium: an important (but often unrecognized) clinical syndrome. Curr Psychiatry Rep. 2002;4(3):202-208.
6. Marcantonio ER, Ta T, Duthie E, et al. Delirium severity and psychomotor types: their relationship with outcomes after hip fracture repair. Am J Geriatr Soc. 2002;50(5):850-857.
7. Ely EW, Inouye SK, Bernard GR, et al. Delirium in mechanically ventilated patients: validity and reliability of the confusion assessment method for the intensive care unit (CAM-ICU). JAMA. 2001;286(21):2703-2710.
8. Duppils GS, Wikblad K. Delirium: behavioural changes before and during the prodromal phase. J Clin Nurs. 2004;13(5):609-616.
9. de Jonghe JF, Kalisvaart KJ, Dijkstra M, et al. Early symptoms in the prodromal phase of delirium: a prospective cohort study in elderly patients undergoing hip surgery. Am J Geriatr Psychiatry. 2007;15(2):112-121.
10. Cook IA. Guideline watch: practice guideline for the treatment of patients with delirium. Arlington, VA: American Psychiatric Publishing; 2004.
11. Hogan D, Gage L, Bruto V, et al. National guidelines for seniors’ mental health: the assessment and treatment of delirium. Canadian Journal of Geriatrics. 2006;9(suppl 2):S42-51.
12. Leentjens AF, Diefenbacher A. A survey of delirium guidelines in Europe. J Psychosom Res. 2006;61(1):123-128.
13. Tropea J, Slee JA, Brand CA, et al. Clinical practice guidelines for the management of delirium in older people in Australia. Australas J Ageing. 2008;27(3):150-156.
14. Mittal D, Majithia D, Kennedy R, et al. Differences in characteristics and outcome of delirium as based on referral patterns. Psychosomatics. 2006;47(5):367-375.
15. Grover S, Subodh BN, Avasthi A, et al. Prevalence and clinical profile of delirium: a study from a tertiary-care hospital in north India. Gen Hosp Psychiatry. 2009;31(1): 25-29.
16. Inouye SK, van Dyck CH, Alessi CA, et al. Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann Intern Med. 1990;113(12): 941-948.
17. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164(6):870-876.
18. Dobmejer K. Delirium in elderly medical patients. Clinical Geriatrics. 1996;4:43-68.
19. Leentjens AF, Maclullich AM, Meagher DJ. Delirium, Cinderella no more...? J Psychosom Res. 2008;65(3):205.
20. Fong TG, Tulebaev SR, Inouye SK. Delirium in elderly adults: diagnosis, prevention and treatment. Nat Rev Neurol. 2009;5(4):210-220.
21. Streubel PN, Ricci WM, Gardner MJ. Fragility fractures: preoperative, perioperative, and postoperative management. Current Orthopaedic Practice. 2009;20(5):482-489.
22. Spronk PE, Riekerk B, Hofhuis J, et al. Occurrence of delirium is severely underestimated in the ICU during daily care. Intensive Care Med. 2009;35(7):1276-1280.
23. Lawlor PG, Gagnon B, Mancini IL, et al. Occurrence, causes, and outcome of delirium in patients with advanced cancer: a prospective study. Arch Intern Med. 2000;160(6):786-794.
24. Ganzini L. Care of patients with delirium at the end of life. Annals of Long-Term Care. 2007;15(3):35-40.
25. Bourne RS, Tahir TA, Borthwick M, et al. Drug treatment of delirium: past, present and future. J Psychosom Res. 2008;65(3):273-282.
26. Yokota H, Ogawa S, Kurokawa A, et al. Regional cerebral blood flow in delirium patients. Psychiatry Clin Neurosci. 2003;57(3):337-339.
27. Maldonado JR. Pathoetiological model of delirium: a comprehensive understanding of the neurobiology of delirium and an evidence-based approach to prevention and treatment. Crit Care Clin. 2008;24(4):789-856, ix.
28. Trzepacz PT. Is there a final common neural pathway in delirium? Focus on acetylcholine and dopamine. Semin Clin Neuropsychiatry. 2000;5(2):132-148.
29. Inouye SK. The dilemma of delirium: clinical and research controversies regarding diagnosis and evaluation of delirium in hospitalized elderly medical patients. Am J Med. 1994;97(3):278-288.
30. Laurila JV, Laakkonen ML, Tilvis RS, et al. Predisposing and precipitating factors for delirium in a frail geriatric population. J Psychosom Res. 2008;65(3):249-254.
31. Morandi A, McCurley J, Vasilevskis EE, et al. Tools to detect delirium superimposed on dementia: a systematic review. J Am Geriatr Soc. 2012;60(11):2005-2013.
32. Kazmierski J, Kowman M, Banach M, et al. The use of DSM-IV and ICD-10 criteria and diagnostic scales for delirium among cardiac surgery patients: results from the IPDACS study. J Neuropsychiatry Clin Neurosci. 2010; 22(4):426-432.
33. Breitbart W, Rosenfeld B, Roth A, et al. The Memorial Delirium Rating Scale. J Pain Symptom Manage. 1997;13(3):128-137.
34. Wong CL, Holroyd-Leduc J, Simel DL, et al. Does this patient have delirium?: value of bedside instruments. JAMA. 2010;304(7):779-786.
35. Marcantonio ER, Flacker JM, Wright RJ, et al. Reducing delirium after hip fracture: a randomized trial. J Am Geriatr Soc. 2011;49(5):516-522.
36. Lundström M, Edlund A, Karlsson S, et al. A multifactorial intervention program reduces the duration of delirium, length of hospitalization, and mortality in delirious patients. J Am Geriatr Soc. 2005;53(4):622-628.
37. Lundström M, Olofsson B, Stenvall M, et al. Postoperative delirium in old patients with femoral neck fracture: a randomized intervention study. Aging Clin Exp Res. 2007; 19(3):178-186.
38. Kratz A. Use of the acute confusion protocol: a research utilization project. J Nurs Care Qual. 2008;23(4):331-337.
39. Prakanrattana U, Prapaitrakool S. Efficacy of risperidone for prevention of postoperative delirium in cardiac surgery. Anaesth Intensive Care. 2007;35(5):714-719.
40. Kaneko T, Cai J, Ishikura T, et al. Prophylactic consecutive administration of haloperidol can reduce the occurrence of postoperative delirium in gastrointestinal surgery. Yonago Acta Medica. 1999;42:179-184.
41. Larsen KA, Kelly SE, Stern TA, et al. Administration of olanzapine to prevent postoperative delirium in elderly joint-replacement patients: a randomized, controlled trial. Psychosomatics. 2010;51(5):409-418.
42. Kalisvaart KJ, de Jonghe JF, Bogaards MJ, et al. Haloperidol prophylaxis for elderly hip-surgery patients at risk for delirium: a randomized placebo-controlled study. J Am Geriatr Soc. 2005;53(10):1658-1666.
43. Hakim SM, Othman AI, Naoum DO. Early treatment with risperidone for subsyndromal delirium after on-pump cardiac surgery in the elderly: a randomized trial. Anesthesiology. 2012;116(5):987-997.
44. Maldonado JR, Wysong A, van der Starre PJ, et al. Dexmedetomidine and the reduction of postoperative delirium after cardiac surgery. Psychosomatics. 2009;50(3): 206-217.
45. Short J. Use of dexmedetomidine for primary sedation in a general intensive care unit. Crit Care Nurse. 2010;30(1): 29-38; quiz 39.
46. Practice guideline for the treatment of patients with delirium. American Psychiatric Association [Comment in: Treatment of patients with delirium. Am J Psychiatry. 2000.]. Am J Psychiatry. 1999;156(suppl 5):1-20.
47. Maldonado JR. Delirium in the acute care setting: characteristics, diagnosis, and treatment. Crit Care Clin. 2008;24(4):657-722, vii.
48. Platt MM, Breitbart W, Smith M, et al. Efficacy of neuroleptics for hypoactive delirium. J Neuropsychiatry Clin Neurosci. 1994;6(1):66-67.
49. Lonergan E, Britton AM, Luxenberg J, et al. Antipsychotics for delirium. Cochrane Database Syst Rev. 2007;(2):CD005594.
50. Seitz DP, Gill SS, van Zyl LT. Antipsychotics in the treatment of delirium: a systematic review. J Clin Psychiatry. 2007;68(1):11-21.
51. Breitbart W, Marotta R, Platt MM, et al. A double-blind trial of haloperidol, chlorpromazine, and lorazepam in the treatment of delirium in hospitalized AIDS patients. Am J Psychiatry. 1996;153(2):231-237.
52. Hu H, Deng W, Yang H, et al. Olanzapine and haloperidol for senile delirium: a randomized controlled observation. Chinese Journal of Clinical Rehabilitation. 2006;10(42): 188-190.
53. Friedman JH, Fernandez HH. Atypical antipsychotics in Parkinson-sensitive populations. J Geriatr Psychiatry Neurol. 2002;15(3):156-170.
54. Seitz DP, Gill SS. Neuroleptic malignant syndrome complicating antipsychotic treatment of delirium or agitation in medical and surgical patients: case reports and a review of the literature. Psychosomatics. 2009; 50(1):8-15.
55. Han CS, Kim YK. A double-blind trial of risperidone and haloperidol for the treatment of delirium. Psychosomatics. 2004;45(4):297-301.
56. Sink KM, Holden KF, Yaffe K. Pharmacological treatment of neuropsychiatric symptoms of dementia: a review of the evidence. JAMA. 2005;293(5):596-608.
57. Hermann N, Lanctôt KL. Atypical antipsychotics for neuropsychiatric symptoms of dementia: malignant or maligned? Drug Saf. 2006;29(10):833-843.
58. Noyan MA, Elbi H, Aksu H. Donepezil for anticholinergic drug intoxication: a case report. Prog Neuropsychopharmacol Biol Psychiatry. 2003;27(5):885-887.
59. Gleason OC. Donepezil for postoperative delirium. Psychosomatics. 2003;44(5):437-438.
60. Overshott R, Karim S, Burns A. Cholinesterase inhibitors for delirium. Cochrane Database Syst Rev. 2008;(1): CD005317.
61. Davis MP. Does trazodone have a role in palliating symptoms? Support Care Cancer. 2007;15(2):221-224.
62. Fick DM, Agostini JV, Inouye SK. Delirium superimposed on dementia: a systematic review. J Am Geriatr Soc. 2002; 50(10):1723-1732.
63. Brajtman S, Wright D, Hogan D, et al. Developing guidelines for the assessment and treatment of delirium in older adults at the end of life. Can Geriatr J. 2011;14(2):40-50.
64. Caraceni A, Simonetti F. Palliating delirium in patients with cancer. Lancet Oncol. 2009;10(2):164-172.
65. Alexopoulos GS, Streim J, Carpenter D, et al; Expert Consensus Panel for using Antipsychotic Drugs in Older Patients. Using antipsychotic agents in older patients. J Clin Psychiatry. 2004;65(suppl 2):5-99; discussion 100-102; quiz 103-104.
66. Granja C, Gomes E, Amaro A, et al. Understanding posttraumatic stress disorder-related symptoms after critical care: the early illness amnesia hypothesis. Crit Care Med. 2008;36(10):2801-2809.
67. Ringdal GI, Ringdal K, Juliebø V, et al. Using the Mini- Mental State Examination to screen for delirium in elderly patients with hip fracture. Dement Geriatr Cogn Disord. 2011;32(6):394-400.
68. Olson RA, Chhanabhai T, McKenzie M. Feasibility study of the Montreal Cognitive Assessment (MoCA) in patients with brain metastases. Support Care Cancer. 2008;16(11):1273-1278.
Although delirium has many descriptive terms (Table 1), a common unifying term is “acute global cognitive dysfunction,” now recognized as delirium; a consensus supported by DSM-51 and ICD-102 (Table 2). According to DSM-5, the essential feature is a disturbance of attention or awareness that is accompanied by a change in baseline cognition that cannot be explained by another preexisting, established, or evolving neurocognitive disorder (the newly named DSM-5 entity for dementia syndromes).1 Because delirium affects the cortex diffusely, psychiatric symptoms can include cognitive, mood, anxiety, or psychotic symptoms. Because many systemic illnesses can induce delirium, the differential diagnosis spans all organ systems.
Three subtypes
Delirium can be classified, based on symptoms,3,4 into 3 subtypes: hyperactive-hyperalert, hypoactive-hypoalert, and mixed delirium. Hyperactive patients present with restlessness and agitation. Hypoactive patients are lethargic, confused, slow to respond to questions, and often appear depressed. The differential prognostic significance of these subtypes has been examined in the literature, with conflicting results. Rabinowitz5 reported that hypoactive delirium has the worst prognosis, while Marcantonio et al6 indicated that the hyperactive subtype is associated with the highest mortality rate. Mixed delirium, with periods of both hyperactivity and hypoactivity, is the most common type of delirium.7
A prodromal phase, characterized by anxiety, frequent requests for nursing and medical assistance, decreased attention, restlessness, vivid dreams, disorientation immediately after awakening, and hallucinations, can occur before an episode of full-spectrum delirium; this prodromal state often is identified retrospectively —after the patient is in an episode of delirium.8,9
Evidence-based guidelines aim to improve recognition and clinical management.10-13 Disruptive behavior is the main reason for psychiatric referral in delirium.14,15 Delayed psychiatric consultation because of non-recognition of delirium is related to variables such as older age; history of a pre-existing, comorbid neurocognitive disorder; and the clinical appearance of hypoactive delirium.14
The case of Mr. D (Box),16 illustrates how the emergence of antipsychotic-associated neuroleptic malignant syndrome (NMS) can complicate antipsychotic treatment of delirium in a geriatric medical patient, although delirium also is a common presentation in NMS.17 Delirium developed after an increase in carbidopa/levodopa, which has central dopaminergic effects that can precipitate delirium, particularly in a geriatric patient with preexisting comorbid neurocognitive disorder. Further complicating Mr. D’s delirium presentation was the development of NMS, which had a multifactorial causation, such as the use of dopamine antagonists (ie, quetiapine, metoclopramide), and an abrupt decrease of a dopaminergic agent (ie, carbidopa/levodopa), all inducing a central dopamine relative hypoactivity.
Epidemiology
Delirium is more common in older patients,15 and is seen in 30% to 40% of hospitalized geriatric patients.18 Delirium in older patients, compared with other adults, is associated with more severe cognitive impairment.19 It is common among geriatric surgical patients (15% to 62%)20 with a peak 2 to 5 days postoperatively for hip fracture,21 and often is seen in ICU patients (70% to 87%).20 However, Spronk et al22 found that delirium is significantly under-recognized in the ICU. Nearly 90% of terminally ill patients become delirious before death.23 Terminal delirium often is unrecognized and can interfere with assessment of other clinical problems.24 A preexisting history of comorbid neurocognitive disorder was evident in as many as two-thirds of delirium cases.25
Pathophysiology and risk factors
The pathophysiology of delirium has been characterized as an imbalance of CNS metabolism, including decreased blood flow in various regions of the brain that may normalize once delirium resolves.26 Studies describe the simultaneous decrease of cholinergic transmission and dopaminergic excess.27,28 Predisposing and precipitating factors for delirium that are of particular importance in geriatric patients include:
• advanced age
• CNS disease
• infection
• cognitive impairment
• male sex
• poor nutrition
• dehydration and other metabolic abnormalities
• cardiovascular events
• substance use
• medication
• sensory deprivation (eg, impaired vision or hearing)
• sleep deprivation
• low level of physical activity.27,29,30
Table 3 lists the most common delirium-provocative medications.27
Evaluation and psychometric scales
The EEG can be useful in evaluating delirium, especially in clinically ambiguous cases. EEG findings may indicate generalized slowing or dropout of the posterior dominant rhythm, and generalized slow theta and delta waves, findings that are more common in delirium than in other neurocognitive disorders and other psychiatric illnesses. The EEG must be interpreted in the context of the delirium diagnostic workup, because abnormalities seen in other neurocognitive disorders can overlap with those of delirium.31
The EEG referral should specify the clinical suspicion of delirium to help interpret the results. Delirium cases in which the patient’s previous cognitive status is unknown may benefit from EEG evaluation, such as:
• in possible status epilepticus
• when delirium improvement has reached a plateau at a lower level of cognitive function than before onset of delirium
• when the patient is unable or unwilling to complete a psychiatric interview.27
Assessment instruments are available to diagnose and monitor delirium (Table 4). Typically, delirium assessment includes examining levels of arousal, psychomotor activity, cognition (ie, orientation, attention, and memory), and perceptual disturbances.
Psychometrically, a review of Table 4 suggests that validity appeared stable with adequate specificity (64% to 99%) but more variable sensitivity (36% to 100%). These reliability parameters also will be affected by the classification system (ie, DSM vs ICD) and the cut-off score employed.32 Most measures (eg, Confusion Assessment Method [CAM], CAM-ICU) provide an adequate sample of behavioral (ie, level of alertness), motor (ie, psychomotor activity), and cognitive (ie, orientation, attention, memory, and receptive language) function, with the exception of the Global Attentiveness Rating, which is a 2-minute open conversation protocol between physician and patient.
Some measures are stand-alone instruments, such as the Memorial Delirium Assessment Scale, whereas the CAM requires administration of separate cognitive screens, including the Mini-Mental State Examination (MMSE) and Digit Span.33 Instruments to detect delirium in critically ill patients are a more recent development. Wong et al34 reported that the most widely studied tool was the CAM. Obtaining collateral information from family, caregivers, and hospital staff is essential, particularly given the fluctuating nature of delirium.
Management
Prevention. Identify patients at high risk of delirium so that preventive strategies can be employed. Multi-component, nonpharmacotherapeutic interventions are used in clinical settings but few randomized trials have been conducted. The contributing effectiveness of individual components is not well-studied, but most include staff education to increase awareness of delirium. Of 3 multi-component intervention randomized trials, 2 reported a significantly lower incidence of delirium in the intervention group.35-37 Implementation of a multi-component protocol in medical/ surgical units was associated with a significant reduction in use of restraints.38
As in Mr. D’s case, complex drug regimens, particularly for CNS illness, can increase the risk of delirium. Considering the medication profile for patients with complex systemic illness—in particular, minimizing the use anticholinergics and dopamine agonists— may be crucial in preventing delirium.
Prophylactic administration of antipsychotics may reduce the risk of developing postoperative delirium.39 Studies of the use of these agents were characterized by small sample sizes and selected groups of patient populations. Of the 4 randomized studies evaluating prophylactic antipsychotics (vs placebo), 3 found a lower incidence of delirium in the intervention groups.39-41
A study of haloperidol in post-GI surgery patients showed a reduced occurrence of delirium,40 whereas its prophylactic use in patients undergoing hip surgery42 did not reduce the incidence of delirium compared with placebo, but did decrease severity when delirium occurred.42
Risperidone39 in post-cardiac surgery and olanzapine41 perioperatively in patients undergoing total knee or hip replacement have been shown to decrease delirium severity and duration. Targeted prophylaxis with risperidone43 in post-cardiac surgery patients who showed disturbed cognition but did not meet criteria for delirium reduced the number of patients requiring medication, compared with placebo.43
Dexmedetomidine, an α-2 adrenergic receptor agonist, compared with propofol or midazolam in post-cardiac valve surgery patients, resulted in a decreased incidence of delirium but no difference in delirium duration, hospital length of stay, or use of other medications.44 However, other studies have shown that dexmedetomidine reduces ICU length of stay and duration of mechanical ventilation.45
Treatment. Management of hospitalized medically ill geriatric patients with delirium is challenging and requires a comprehensive approach. The first step in delirium management is prompt identification and management of systemic medical disturbances associated with the delirium episode. First-line, nonpharmacotherapeutic strategies for patients with delirium include:
• reorientation
• behavioral interventions (eg, use of clear instructions and frequent eye contact with patients)
• environmental interventions (eg, minimal noise, adequate lighting, and limited room and staff changes)
• avoidance of physical restraints.46
Consider employing family members or hospital staff sitters to stay with the patient and to reassure, reorient, and watch for agitation and other unsafe behaviors (eg, attempted elopement). Psychoeducation for the patient and family on the phenomenology of delirium can be helpful.
The use of drug treatment strategies should be integrated into a comprehensive approach that includes the routine use of nondrug measures.46 Using medications for treating hypoactive delirium, formerly controversial, now has wider acceptance.47,48 A few high-quality randomized trials have been performed.25,49,50
Pharmacotherapy, especially in frail patients, should be initiated at the lowest starting dosage and titrated cautiously to clinical effect and for the shortest period of time necessary. Antipsychotics are preferred agents for treating all subtypes of delirium; haloperidol is widely used.46,51,52 However, antipsychotics, including haloperidol, can be associated with adverse neurologic effects such as extrapyramidal symptoms (EPS) and NMS.
Although reported less frequently than with haloperidol, other agents have been implicated in development of EPS and NMS, including atypical antipsychotics and antiemetic dopamine antagonists, particularly in parkinsonism-prone patients.53 Strategies that can minimize such risks in geriatric inpatients with delirium include oral, rather than parenteral, use of antipsychotics—preferential use of atypical over typical antipsychotics— and lowest effective dosages.54
In controlled trials, atypical antipsychotics for delirium showed efficacy compared with haloperidol.52,55 However, there is no research that demonstrates any advantage of one atypical over another.25
In Mr. D’s case, the most important intervention for managing delirium caused by NMS is to discontinue all dopamine antagonists and treat agitation with judicious doses of a benzodiazepine, with supportive care.17 In cases of sudden discontinuation or a dosage decrease of dopamine agonists, these medications should be resumed or optimized to minimize the risk of NMS-associated rhabdomyolysis and subsequent renal failure.17 Antipsychotics carry an increased risk of stroke and mortality in older patients with established or evolving neurocognitive disorders56,57 and can cause prolongation of the QTc interval.57
Other medications that could be used for delirium include cholinesterase inhibitors58,59 (although larger trials and a systematic review did not support this use60), and 5-HT receptor antagonists,61 such as trazodone. Benzodiazepines, such as lorazepam, are first-line treatment for delirium associated with seizures or withdrawal from alcohol, sedatives, hypnotics, and anxiolytics and for delirium caused by NMS. Be cautious about using benzodiazepines in geriatric patients because of a risk of respiratory depression, falls, sedation, and amnesia.
Geriatric patients with alcoholism and those with malnutrition are prone to thiamine and vitamin B12 deficiencies, which can induce delirium. Laboratory assessment and consideration of supplementation is recommended. Despite high occurrence of delirium in hospitalized older adults with preexisting comorbid neurocognitive disorders, there is no standard care for delirium comorbid with another neurocognitive disorder.62 Clinical practice guidelines for older patients receiving palliative care have been developed63; the goal is to minimize suffering and discomfort in patients in palliative care.64
Post-delirium prophylaxis. Medications for delirium usually can be tapered and discontinued once the episode has resolved and the patient is stable; it is common to discontinue medications when the patient has been symptom-free for 1 week.65 Some patients (eg, with end-stage liver disease, disseminated cancer) are prone to recurrent or to prolonged or chronic delirium. A period of post-recovery treatment with antipsychotics—even indefinite treatment in some cases—should be considered.
Post-delirium debriefing and aftercare. The psychological complications of delirium are distressing for the patient and his (her) caregivers. Psychiatric complications associated with delirium, including acute stress disorder—which might predict posttraumatic stress disorder—have been explored; early recognition and treatment may improve long-term outcomes.66 After recovery from acute delirium, cognitive assessment (eg, MMSE67 or Montreal Cognitive Assessment68) is recommended to validate current cognitive status because patients may have persistent decrement in cognitive function compared with pre-delirium condition, even after recovery from the acute episode.
Post-delirium debriefing may help patients who have recovered from a delirium episode. Patients may fear that their brief period of hallucinations might represent the onset of a chronic-relapsing psychotic disorder. Allow patients to communicate their distress about the delirium episode and give them the opportunity to talk through the experience. Brief them on the possibility that delirium will recur and advise them to seek emergency medical care in case of recurrence. Advise patients to monitor and maintain a normal sleep-wake cycle.
Family members can watch for syndromal recurrence of delirium. They should be encouraged to discuss their reaction to having seen their relative in a delirious state.
Health care systems with integrated electronic medical records should list “delirium, resolved” on the patient’s illness profile or problem list and alert the patient’s primary care provider to the delirium history to avoid future exposure to delirium-provocative medications, and to prompt the provider to assume an active role in post-delirium care, including delirium recurrence surveillance, medication adjustment, risk factor management, and post-recovery cognitive assessment.
Bottom Line
Evaluation of delirium in geriatric patients includes clinical vigilance and screening, differentiating delirium from other neurocognitive disorders, and identifying and treating underlying causes. Perioperative use of antipsychotics may reduce the incidence of delirium, although hospital length of stay generally has not been reduced with prophylaxis. Management interventions include staff education, systematic screening, use of multicomponent interventions, and pharmacologic interventions.
Related Resources
• Downing LJ, Caprio TV, Lyness JM. Geriatric psychiatry review: differential diagnosis and treatment of the 3 D’s - delirium, dementia, and depression. Curr Psychiatry Rep. 2013;15(6):365.
• Brooks PB. Postoperative delirium in elderly patients. Am J Nurs. 2012;112(9):38-49.
Drug Brand Names
Carbidopa/levodopa • Sinemet Midazolam • Versed
Dexmedetomidine • Precedex Olanzapine • Zyprexa
Haloperidol • Haldol Propofol • Diprivan
Lithium • Eskalith, Lithobid Quetiapine • Seroquel
Lorazepam • Ativan Risperidone • Risperdal
Metoclopramide • Reglan Trazodone • Desyrel
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Although delirium has many descriptive terms (Table 1), a common unifying term is “acute global cognitive dysfunction,” now recognized as delirium; a consensus supported by DSM-51 and ICD-102 (Table 2). According to DSM-5, the essential feature is a disturbance of attention or awareness that is accompanied by a change in baseline cognition that cannot be explained by another preexisting, established, or evolving neurocognitive disorder (the newly named DSM-5 entity for dementia syndromes).1 Because delirium affects the cortex diffusely, psychiatric symptoms can include cognitive, mood, anxiety, or psychotic symptoms. Because many systemic illnesses can induce delirium, the differential diagnosis spans all organ systems.
Three subtypes
Delirium can be classified, based on symptoms,3,4 into 3 subtypes: hyperactive-hyperalert, hypoactive-hypoalert, and mixed delirium. Hyperactive patients present with restlessness and agitation. Hypoactive patients are lethargic, confused, slow to respond to questions, and often appear depressed. The differential prognostic significance of these subtypes has been examined in the literature, with conflicting results. Rabinowitz5 reported that hypoactive delirium has the worst prognosis, while Marcantonio et al6 indicated that the hyperactive subtype is associated with the highest mortality rate. Mixed delirium, with periods of both hyperactivity and hypoactivity, is the most common type of delirium.7
A prodromal phase, characterized by anxiety, frequent requests for nursing and medical assistance, decreased attention, restlessness, vivid dreams, disorientation immediately after awakening, and hallucinations, can occur before an episode of full-spectrum delirium; this prodromal state often is identified retrospectively —after the patient is in an episode of delirium.8,9
Evidence-based guidelines aim to improve recognition and clinical management.10-13 Disruptive behavior is the main reason for psychiatric referral in delirium.14,15 Delayed psychiatric consultation because of non-recognition of delirium is related to variables such as older age; history of a pre-existing, comorbid neurocognitive disorder; and the clinical appearance of hypoactive delirium.14
The case of Mr. D (Box),16 illustrates how the emergence of antipsychotic-associated neuroleptic malignant syndrome (NMS) can complicate antipsychotic treatment of delirium in a geriatric medical patient, although delirium also is a common presentation in NMS.17 Delirium developed after an increase in carbidopa/levodopa, which has central dopaminergic effects that can precipitate delirium, particularly in a geriatric patient with preexisting comorbid neurocognitive disorder. Further complicating Mr. D’s delirium presentation was the development of NMS, which had a multifactorial causation, such as the use of dopamine antagonists (ie, quetiapine, metoclopramide), and an abrupt decrease of a dopaminergic agent (ie, carbidopa/levodopa), all inducing a central dopamine relative hypoactivity.
Epidemiology
Delirium is more common in older patients,15 and is seen in 30% to 40% of hospitalized geriatric patients.18 Delirium in older patients, compared with other adults, is associated with more severe cognitive impairment.19 It is common among geriatric surgical patients (15% to 62%)20 with a peak 2 to 5 days postoperatively for hip fracture,21 and often is seen in ICU patients (70% to 87%).20 However, Spronk et al22 found that delirium is significantly under-recognized in the ICU. Nearly 90% of terminally ill patients become delirious before death.23 Terminal delirium often is unrecognized and can interfere with assessment of other clinical problems.24 A preexisting history of comorbid neurocognitive disorder was evident in as many as two-thirds of delirium cases.25
Pathophysiology and risk factors
The pathophysiology of delirium has been characterized as an imbalance of CNS metabolism, including decreased blood flow in various regions of the brain that may normalize once delirium resolves.26 Studies describe the simultaneous decrease of cholinergic transmission and dopaminergic excess.27,28 Predisposing and precipitating factors for delirium that are of particular importance in geriatric patients include:
• advanced age
• CNS disease
• infection
• cognitive impairment
• male sex
• poor nutrition
• dehydration and other metabolic abnormalities
• cardiovascular events
• substance use
• medication
• sensory deprivation (eg, impaired vision or hearing)
• sleep deprivation
• low level of physical activity.27,29,30
Table 3 lists the most common delirium-provocative medications.27
Evaluation and psychometric scales
The EEG can be useful in evaluating delirium, especially in clinically ambiguous cases. EEG findings may indicate generalized slowing or dropout of the posterior dominant rhythm, and generalized slow theta and delta waves, findings that are more common in delirium than in other neurocognitive disorders and other psychiatric illnesses. The EEG must be interpreted in the context of the delirium diagnostic workup, because abnormalities seen in other neurocognitive disorders can overlap with those of delirium.31
The EEG referral should specify the clinical suspicion of delirium to help interpret the results. Delirium cases in which the patient’s previous cognitive status is unknown may benefit from EEG evaluation, such as:
• in possible status epilepticus
• when delirium improvement has reached a plateau at a lower level of cognitive function than before onset of delirium
• when the patient is unable or unwilling to complete a psychiatric interview.27
Assessment instruments are available to diagnose and monitor delirium (Table 4). Typically, delirium assessment includes examining levels of arousal, psychomotor activity, cognition (ie, orientation, attention, and memory), and perceptual disturbances.
Psychometrically, a review of Table 4 suggests that validity appeared stable with adequate specificity (64% to 99%) but more variable sensitivity (36% to 100%). These reliability parameters also will be affected by the classification system (ie, DSM vs ICD) and the cut-off score employed.32 Most measures (eg, Confusion Assessment Method [CAM], CAM-ICU) provide an adequate sample of behavioral (ie, level of alertness), motor (ie, psychomotor activity), and cognitive (ie, orientation, attention, memory, and receptive language) function, with the exception of the Global Attentiveness Rating, which is a 2-minute open conversation protocol between physician and patient.
Some measures are stand-alone instruments, such as the Memorial Delirium Assessment Scale, whereas the CAM requires administration of separate cognitive screens, including the Mini-Mental State Examination (MMSE) and Digit Span.33 Instruments to detect delirium in critically ill patients are a more recent development. Wong et al34 reported that the most widely studied tool was the CAM. Obtaining collateral information from family, caregivers, and hospital staff is essential, particularly given the fluctuating nature of delirium.
Management
Prevention. Identify patients at high risk of delirium so that preventive strategies can be employed. Multi-component, nonpharmacotherapeutic interventions are used in clinical settings but few randomized trials have been conducted. The contributing effectiveness of individual components is not well-studied, but most include staff education to increase awareness of delirium. Of 3 multi-component intervention randomized trials, 2 reported a significantly lower incidence of delirium in the intervention group.35-37 Implementation of a multi-component protocol in medical/ surgical units was associated with a significant reduction in use of restraints.38
As in Mr. D’s case, complex drug regimens, particularly for CNS illness, can increase the risk of delirium. Considering the medication profile for patients with complex systemic illness—in particular, minimizing the use anticholinergics and dopamine agonists— may be crucial in preventing delirium.
Prophylactic administration of antipsychotics may reduce the risk of developing postoperative delirium.39 Studies of the use of these agents were characterized by small sample sizes and selected groups of patient populations. Of the 4 randomized studies evaluating prophylactic antipsychotics (vs placebo), 3 found a lower incidence of delirium in the intervention groups.39-41
A study of haloperidol in post-GI surgery patients showed a reduced occurrence of delirium,40 whereas its prophylactic use in patients undergoing hip surgery42 did not reduce the incidence of delirium compared with placebo, but did decrease severity when delirium occurred.42
Risperidone39 in post-cardiac surgery and olanzapine41 perioperatively in patients undergoing total knee or hip replacement have been shown to decrease delirium severity and duration. Targeted prophylaxis with risperidone43 in post-cardiac surgery patients who showed disturbed cognition but did not meet criteria for delirium reduced the number of patients requiring medication, compared with placebo.43
Dexmedetomidine, an α-2 adrenergic receptor agonist, compared with propofol or midazolam in post-cardiac valve surgery patients, resulted in a decreased incidence of delirium but no difference in delirium duration, hospital length of stay, or use of other medications.44 However, other studies have shown that dexmedetomidine reduces ICU length of stay and duration of mechanical ventilation.45
Treatment. Management of hospitalized medically ill geriatric patients with delirium is challenging and requires a comprehensive approach. The first step in delirium management is prompt identification and management of systemic medical disturbances associated with the delirium episode. First-line, nonpharmacotherapeutic strategies for patients with delirium include:
• reorientation
• behavioral interventions (eg, use of clear instructions and frequent eye contact with patients)
• environmental interventions (eg, minimal noise, adequate lighting, and limited room and staff changes)
• avoidance of physical restraints.46
Consider employing family members or hospital staff sitters to stay with the patient and to reassure, reorient, and watch for agitation and other unsafe behaviors (eg, attempted elopement). Psychoeducation for the patient and family on the phenomenology of delirium can be helpful.
The use of drug treatment strategies should be integrated into a comprehensive approach that includes the routine use of nondrug measures.46 Using medications for treating hypoactive delirium, formerly controversial, now has wider acceptance.47,48 A few high-quality randomized trials have been performed.25,49,50
Pharmacotherapy, especially in frail patients, should be initiated at the lowest starting dosage and titrated cautiously to clinical effect and for the shortest period of time necessary. Antipsychotics are preferred agents for treating all subtypes of delirium; haloperidol is widely used.46,51,52 However, antipsychotics, including haloperidol, can be associated with adverse neurologic effects such as extrapyramidal symptoms (EPS) and NMS.
Although reported less frequently than with haloperidol, other agents have been implicated in development of EPS and NMS, including atypical antipsychotics and antiemetic dopamine antagonists, particularly in parkinsonism-prone patients.53 Strategies that can minimize such risks in geriatric inpatients with delirium include oral, rather than parenteral, use of antipsychotics—preferential use of atypical over typical antipsychotics— and lowest effective dosages.54
In controlled trials, atypical antipsychotics for delirium showed efficacy compared with haloperidol.52,55 However, there is no research that demonstrates any advantage of one atypical over another.25
In Mr. D’s case, the most important intervention for managing delirium caused by NMS is to discontinue all dopamine antagonists and treat agitation with judicious doses of a benzodiazepine, with supportive care.17 In cases of sudden discontinuation or a dosage decrease of dopamine agonists, these medications should be resumed or optimized to minimize the risk of NMS-associated rhabdomyolysis and subsequent renal failure.17 Antipsychotics carry an increased risk of stroke and mortality in older patients with established or evolving neurocognitive disorders56,57 and can cause prolongation of the QTc interval.57
Other medications that could be used for delirium include cholinesterase inhibitors58,59 (although larger trials and a systematic review did not support this use60), and 5-HT receptor antagonists,61 such as trazodone. Benzodiazepines, such as lorazepam, are first-line treatment for delirium associated with seizures or withdrawal from alcohol, sedatives, hypnotics, and anxiolytics and for delirium caused by NMS. Be cautious about using benzodiazepines in geriatric patients because of a risk of respiratory depression, falls, sedation, and amnesia.
Geriatric patients with alcoholism and those with malnutrition are prone to thiamine and vitamin B12 deficiencies, which can induce delirium. Laboratory assessment and consideration of supplementation is recommended. Despite high occurrence of delirium in hospitalized older adults with preexisting comorbid neurocognitive disorders, there is no standard care for delirium comorbid with another neurocognitive disorder.62 Clinical practice guidelines for older patients receiving palliative care have been developed63; the goal is to minimize suffering and discomfort in patients in palliative care.64
Post-delirium prophylaxis. Medications for delirium usually can be tapered and discontinued once the episode has resolved and the patient is stable; it is common to discontinue medications when the patient has been symptom-free for 1 week.65 Some patients (eg, with end-stage liver disease, disseminated cancer) are prone to recurrent or to prolonged or chronic delirium. A period of post-recovery treatment with antipsychotics—even indefinite treatment in some cases—should be considered.
Post-delirium debriefing and aftercare. The psychological complications of delirium are distressing for the patient and his (her) caregivers. Psychiatric complications associated with delirium, including acute stress disorder—which might predict posttraumatic stress disorder—have been explored; early recognition and treatment may improve long-term outcomes.66 After recovery from acute delirium, cognitive assessment (eg, MMSE67 or Montreal Cognitive Assessment68) is recommended to validate current cognitive status because patients may have persistent decrement in cognitive function compared with pre-delirium condition, even after recovery from the acute episode.
Post-delirium debriefing may help patients who have recovered from a delirium episode. Patients may fear that their brief period of hallucinations might represent the onset of a chronic-relapsing psychotic disorder. Allow patients to communicate their distress about the delirium episode and give them the opportunity to talk through the experience. Brief them on the possibility that delirium will recur and advise them to seek emergency medical care in case of recurrence. Advise patients to monitor and maintain a normal sleep-wake cycle.
Family members can watch for syndromal recurrence of delirium. They should be encouraged to discuss their reaction to having seen their relative in a delirious state.
Health care systems with integrated electronic medical records should list “delirium, resolved” on the patient’s illness profile or problem list and alert the patient’s primary care provider to the delirium history to avoid future exposure to delirium-provocative medications, and to prompt the provider to assume an active role in post-delirium care, including delirium recurrence surveillance, medication adjustment, risk factor management, and post-recovery cognitive assessment.
Bottom Line
Evaluation of delirium in geriatric patients includes clinical vigilance and screening, differentiating delirium from other neurocognitive disorders, and identifying and treating underlying causes. Perioperative use of antipsychotics may reduce the incidence of delirium, although hospital length of stay generally has not been reduced with prophylaxis. Management interventions include staff education, systematic screening, use of multicomponent interventions, and pharmacologic interventions.
Related Resources
• Downing LJ, Caprio TV, Lyness JM. Geriatric psychiatry review: differential diagnosis and treatment of the 3 D’s - delirium, dementia, and depression. Curr Psychiatry Rep. 2013;15(6):365.
• Brooks PB. Postoperative delirium in elderly patients. Am J Nurs. 2012;112(9):38-49.
Drug Brand Names
Carbidopa/levodopa • Sinemet Midazolam • Versed
Dexmedetomidine • Precedex Olanzapine • Zyprexa
Haloperidol • Haldol Propofol • Diprivan
Lithium • Eskalith, Lithobid Quetiapine • Seroquel
Lorazepam • Ativan Risperidone • Risperdal
Metoclopramide • Reglan Trazodone • Desyrel
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Diagnostic and statistical manual of mental disorders, fifth edition. Washington, DC: American Psychiatric Association; 2013.
2. World Health Organization. The ICD-10 classification of mental and behavioural disorders. Diagnostic criteria for research. Geneva, Switzerland: WHO; 1993.
3. Lipowski ZJ. Delirium in the elderly patient. N Engl J Med. 1989;320(9):578-582.
4. Meagher DJ, Trzepacz PT. Motoric subtypes of delirium. Semin Clin Neuropsychiatry. 2000;5(2):75-85.
5. Rabinowitz T. Delirium: an important (but often unrecognized) clinical syndrome. Curr Psychiatry Rep. 2002;4(3):202-208.
6. Marcantonio ER, Ta T, Duthie E, et al. Delirium severity and psychomotor types: their relationship with outcomes after hip fracture repair. Am J Geriatr Soc. 2002;50(5):850-857.
7. Ely EW, Inouye SK, Bernard GR, et al. Delirium in mechanically ventilated patients: validity and reliability of the confusion assessment method for the intensive care unit (CAM-ICU). JAMA. 2001;286(21):2703-2710.
8. Duppils GS, Wikblad K. Delirium: behavioural changes before and during the prodromal phase. J Clin Nurs. 2004;13(5):609-616.
9. de Jonghe JF, Kalisvaart KJ, Dijkstra M, et al. Early symptoms in the prodromal phase of delirium: a prospective cohort study in elderly patients undergoing hip surgery. Am J Geriatr Psychiatry. 2007;15(2):112-121.
10. Cook IA. Guideline watch: practice guideline for the treatment of patients with delirium. Arlington, VA: American Psychiatric Publishing; 2004.
11. Hogan D, Gage L, Bruto V, et al. National guidelines for seniors’ mental health: the assessment and treatment of delirium. Canadian Journal of Geriatrics. 2006;9(suppl 2):S42-51.
12. Leentjens AF, Diefenbacher A. A survey of delirium guidelines in Europe. J Psychosom Res. 2006;61(1):123-128.
13. Tropea J, Slee JA, Brand CA, et al. Clinical practice guidelines for the management of delirium in older people in Australia. Australas J Ageing. 2008;27(3):150-156.
14. Mittal D, Majithia D, Kennedy R, et al. Differences in characteristics and outcome of delirium as based on referral patterns. Psychosomatics. 2006;47(5):367-375.
15. Grover S, Subodh BN, Avasthi A, et al. Prevalence and clinical profile of delirium: a study from a tertiary-care hospital in north India. Gen Hosp Psychiatry. 2009;31(1): 25-29.
16. Inouye SK, van Dyck CH, Alessi CA, et al. Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann Intern Med. 1990;113(12): 941-948.
17. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164(6):870-876.
18. Dobmejer K. Delirium in elderly medical patients. Clinical Geriatrics. 1996;4:43-68.
19. Leentjens AF, Maclullich AM, Meagher DJ. Delirium, Cinderella no more...? J Psychosom Res. 2008;65(3):205.
20. Fong TG, Tulebaev SR, Inouye SK. Delirium in elderly adults: diagnosis, prevention and treatment. Nat Rev Neurol. 2009;5(4):210-220.
21. Streubel PN, Ricci WM, Gardner MJ. Fragility fractures: preoperative, perioperative, and postoperative management. Current Orthopaedic Practice. 2009;20(5):482-489.
22. Spronk PE, Riekerk B, Hofhuis J, et al. Occurrence of delirium is severely underestimated in the ICU during daily care. Intensive Care Med. 2009;35(7):1276-1280.
23. Lawlor PG, Gagnon B, Mancini IL, et al. Occurrence, causes, and outcome of delirium in patients with advanced cancer: a prospective study. Arch Intern Med. 2000;160(6):786-794.
24. Ganzini L. Care of patients with delirium at the end of life. Annals of Long-Term Care. 2007;15(3):35-40.
25. Bourne RS, Tahir TA, Borthwick M, et al. Drug treatment of delirium: past, present and future. J Psychosom Res. 2008;65(3):273-282.
26. Yokota H, Ogawa S, Kurokawa A, et al. Regional cerebral blood flow in delirium patients. Psychiatry Clin Neurosci. 2003;57(3):337-339.
27. Maldonado JR. Pathoetiological model of delirium: a comprehensive understanding of the neurobiology of delirium and an evidence-based approach to prevention and treatment. Crit Care Clin. 2008;24(4):789-856, ix.
28. Trzepacz PT. Is there a final common neural pathway in delirium? Focus on acetylcholine and dopamine. Semin Clin Neuropsychiatry. 2000;5(2):132-148.
29. Inouye SK. The dilemma of delirium: clinical and research controversies regarding diagnosis and evaluation of delirium in hospitalized elderly medical patients. Am J Med. 1994;97(3):278-288.
30. Laurila JV, Laakkonen ML, Tilvis RS, et al. Predisposing and precipitating factors for delirium in a frail geriatric population. J Psychosom Res. 2008;65(3):249-254.
31. Morandi A, McCurley J, Vasilevskis EE, et al. Tools to detect delirium superimposed on dementia: a systematic review. J Am Geriatr Soc. 2012;60(11):2005-2013.
32. Kazmierski J, Kowman M, Banach M, et al. The use of DSM-IV and ICD-10 criteria and diagnostic scales for delirium among cardiac surgery patients: results from the IPDACS study. J Neuropsychiatry Clin Neurosci. 2010; 22(4):426-432.
33. Breitbart W, Rosenfeld B, Roth A, et al. The Memorial Delirium Rating Scale. J Pain Symptom Manage. 1997;13(3):128-137.
34. Wong CL, Holroyd-Leduc J, Simel DL, et al. Does this patient have delirium?: value of bedside instruments. JAMA. 2010;304(7):779-786.
35. Marcantonio ER, Flacker JM, Wright RJ, et al. Reducing delirium after hip fracture: a randomized trial. J Am Geriatr Soc. 2011;49(5):516-522.
36. Lundström M, Edlund A, Karlsson S, et al. A multifactorial intervention program reduces the duration of delirium, length of hospitalization, and mortality in delirious patients. J Am Geriatr Soc. 2005;53(4):622-628.
37. Lundström M, Olofsson B, Stenvall M, et al. Postoperative delirium in old patients with femoral neck fracture: a randomized intervention study. Aging Clin Exp Res. 2007; 19(3):178-186.
38. Kratz A. Use of the acute confusion protocol: a research utilization project. J Nurs Care Qual. 2008;23(4):331-337.
39. Prakanrattana U, Prapaitrakool S. Efficacy of risperidone for prevention of postoperative delirium in cardiac surgery. Anaesth Intensive Care. 2007;35(5):714-719.
40. Kaneko T, Cai J, Ishikura T, et al. Prophylactic consecutive administration of haloperidol can reduce the occurrence of postoperative delirium in gastrointestinal surgery. Yonago Acta Medica. 1999;42:179-184.
41. Larsen KA, Kelly SE, Stern TA, et al. Administration of olanzapine to prevent postoperative delirium in elderly joint-replacement patients: a randomized, controlled trial. Psychosomatics. 2010;51(5):409-418.
42. Kalisvaart KJ, de Jonghe JF, Bogaards MJ, et al. Haloperidol prophylaxis for elderly hip-surgery patients at risk for delirium: a randomized placebo-controlled study. J Am Geriatr Soc. 2005;53(10):1658-1666.
43. Hakim SM, Othman AI, Naoum DO. Early treatment with risperidone for subsyndromal delirium after on-pump cardiac surgery in the elderly: a randomized trial. Anesthesiology. 2012;116(5):987-997.
44. Maldonado JR, Wysong A, van der Starre PJ, et al. Dexmedetomidine and the reduction of postoperative delirium after cardiac surgery. Psychosomatics. 2009;50(3): 206-217.
45. Short J. Use of dexmedetomidine for primary sedation in a general intensive care unit. Crit Care Nurse. 2010;30(1): 29-38; quiz 39.
46. Practice guideline for the treatment of patients with delirium. American Psychiatric Association [Comment in: Treatment of patients with delirium. Am J Psychiatry. 2000.]. Am J Psychiatry. 1999;156(suppl 5):1-20.
47. Maldonado JR. Delirium in the acute care setting: characteristics, diagnosis, and treatment. Crit Care Clin. 2008;24(4):657-722, vii.
48. Platt MM, Breitbart W, Smith M, et al. Efficacy of neuroleptics for hypoactive delirium. J Neuropsychiatry Clin Neurosci. 1994;6(1):66-67.
49. Lonergan E, Britton AM, Luxenberg J, et al. Antipsychotics for delirium. Cochrane Database Syst Rev. 2007;(2):CD005594.
50. Seitz DP, Gill SS, van Zyl LT. Antipsychotics in the treatment of delirium: a systematic review. J Clin Psychiatry. 2007;68(1):11-21.
51. Breitbart W, Marotta R, Platt MM, et al. A double-blind trial of haloperidol, chlorpromazine, and lorazepam in the treatment of delirium in hospitalized AIDS patients. Am J Psychiatry. 1996;153(2):231-237.
52. Hu H, Deng W, Yang H, et al. Olanzapine and haloperidol for senile delirium: a randomized controlled observation. Chinese Journal of Clinical Rehabilitation. 2006;10(42): 188-190.
53. Friedman JH, Fernandez HH. Atypical antipsychotics in Parkinson-sensitive populations. J Geriatr Psychiatry Neurol. 2002;15(3):156-170.
54. Seitz DP, Gill SS. Neuroleptic malignant syndrome complicating antipsychotic treatment of delirium or agitation in medical and surgical patients: case reports and a review of the literature. Psychosomatics. 2009; 50(1):8-15.
55. Han CS, Kim YK. A double-blind trial of risperidone and haloperidol for the treatment of delirium. Psychosomatics. 2004;45(4):297-301.
56. Sink KM, Holden KF, Yaffe K. Pharmacological treatment of neuropsychiatric symptoms of dementia: a review of the evidence. JAMA. 2005;293(5):596-608.
57. Hermann N, Lanctôt KL. Atypical antipsychotics for neuropsychiatric symptoms of dementia: malignant or maligned? Drug Saf. 2006;29(10):833-843.
58. Noyan MA, Elbi H, Aksu H. Donepezil for anticholinergic drug intoxication: a case report. Prog Neuropsychopharmacol Biol Psychiatry. 2003;27(5):885-887.
59. Gleason OC. Donepezil for postoperative delirium. Psychosomatics. 2003;44(5):437-438.
60. Overshott R, Karim S, Burns A. Cholinesterase inhibitors for delirium. Cochrane Database Syst Rev. 2008;(1): CD005317.
61. Davis MP. Does trazodone have a role in palliating symptoms? Support Care Cancer. 2007;15(2):221-224.
62. Fick DM, Agostini JV, Inouye SK. Delirium superimposed on dementia: a systematic review. J Am Geriatr Soc. 2002; 50(10):1723-1732.
63. Brajtman S, Wright D, Hogan D, et al. Developing guidelines for the assessment and treatment of delirium in older adults at the end of life. Can Geriatr J. 2011;14(2):40-50.
64. Caraceni A, Simonetti F. Palliating delirium in patients with cancer. Lancet Oncol. 2009;10(2):164-172.
65. Alexopoulos GS, Streim J, Carpenter D, et al; Expert Consensus Panel for using Antipsychotic Drugs in Older Patients. Using antipsychotic agents in older patients. J Clin Psychiatry. 2004;65(suppl 2):5-99; discussion 100-102; quiz 103-104.
66. Granja C, Gomes E, Amaro A, et al. Understanding posttraumatic stress disorder-related symptoms after critical care: the early illness amnesia hypothesis. Crit Care Med. 2008;36(10):2801-2809.
67. Ringdal GI, Ringdal K, Juliebø V, et al. Using the Mini- Mental State Examination to screen for delirium in elderly patients with hip fracture. Dement Geriatr Cogn Disord. 2011;32(6):394-400.
68. Olson RA, Chhanabhai T, McKenzie M. Feasibility study of the Montreal Cognitive Assessment (MoCA) in patients with brain metastases. Support Care Cancer. 2008;16(11):1273-1278.
1. Diagnostic and statistical manual of mental disorders, fifth edition. Washington, DC: American Psychiatric Association; 2013.
2. World Health Organization. The ICD-10 classification of mental and behavioural disorders. Diagnostic criteria for research. Geneva, Switzerland: WHO; 1993.
3. Lipowski ZJ. Delirium in the elderly patient. N Engl J Med. 1989;320(9):578-582.
4. Meagher DJ, Trzepacz PT. Motoric subtypes of delirium. Semin Clin Neuropsychiatry. 2000;5(2):75-85.
5. Rabinowitz T. Delirium: an important (but often unrecognized) clinical syndrome. Curr Psychiatry Rep. 2002;4(3):202-208.
6. Marcantonio ER, Ta T, Duthie E, et al. Delirium severity and psychomotor types: their relationship with outcomes after hip fracture repair. Am J Geriatr Soc. 2002;50(5):850-857.
7. Ely EW, Inouye SK, Bernard GR, et al. Delirium in mechanically ventilated patients: validity and reliability of the confusion assessment method for the intensive care unit (CAM-ICU). JAMA. 2001;286(21):2703-2710.
8. Duppils GS, Wikblad K. Delirium: behavioural changes before and during the prodromal phase. J Clin Nurs. 2004;13(5):609-616.
9. de Jonghe JF, Kalisvaart KJ, Dijkstra M, et al. Early symptoms in the prodromal phase of delirium: a prospective cohort study in elderly patients undergoing hip surgery. Am J Geriatr Psychiatry. 2007;15(2):112-121.
10. Cook IA. Guideline watch: practice guideline for the treatment of patients with delirium. Arlington, VA: American Psychiatric Publishing; 2004.
11. Hogan D, Gage L, Bruto V, et al. National guidelines for seniors’ mental health: the assessment and treatment of delirium. Canadian Journal of Geriatrics. 2006;9(suppl 2):S42-51.
12. Leentjens AF, Diefenbacher A. A survey of delirium guidelines in Europe. J Psychosom Res. 2006;61(1):123-128.
13. Tropea J, Slee JA, Brand CA, et al. Clinical practice guidelines for the management of delirium in older people in Australia. Australas J Ageing. 2008;27(3):150-156.
14. Mittal D, Majithia D, Kennedy R, et al. Differences in characteristics and outcome of delirium as based on referral patterns. Psychosomatics. 2006;47(5):367-375.
15. Grover S, Subodh BN, Avasthi A, et al. Prevalence and clinical profile of delirium: a study from a tertiary-care hospital in north India. Gen Hosp Psychiatry. 2009;31(1): 25-29.
16. Inouye SK, van Dyck CH, Alessi CA, et al. Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann Intern Med. 1990;113(12): 941-948.
17. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164(6):870-876.
18. Dobmejer K. Delirium in elderly medical patients. Clinical Geriatrics. 1996;4:43-68.
19. Leentjens AF, Maclullich AM, Meagher DJ. Delirium, Cinderella no more...? J Psychosom Res. 2008;65(3):205.
20. Fong TG, Tulebaev SR, Inouye SK. Delirium in elderly adults: diagnosis, prevention and treatment. Nat Rev Neurol. 2009;5(4):210-220.
21. Streubel PN, Ricci WM, Gardner MJ. Fragility fractures: preoperative, perioperative, and postoperative management. Current Orthopaedic Practice. 2009;20(5):482-489.
22. Spronk PE, Riekerk B, Hofhuis J, et al. Occurrence of delirium is severely underestimated in the ICU during daily care. Intensive Care Med. 2009;35(7):1276-1280.
23. Lawlor PG, Gagnon B, Mancini IL, et al. Occurrence, causes, and outcome of delirium in patients with advanced cancer: a prospective study. Arch Intern Med. 2000;160(6):786-794.
24. Ganzini L. Care of patients with delirium at the end of life. Annals of Long-Term Care. 2007;15(3):35-40.
25. Bourne RS, Tahir TA, Borthwick M, et al. Drug treatment of delirium: past, present and future. J Psychosom Res. 2008;65(3):273-282.
26. Yokota H, Ogawa S, Kurokawa A, et al. Regional cerebral blood flow in delirium patients. Psychiatry Clin Neurosci. 2003;57(3):337-339.
27. Maldonado JR. Pathoetiological model of delirium: a comprehensive understanding of the neurobiology of delirium and an evidence-based approach to prevention and treatment. Crit Care Clin. 2008;24(4):789-856, ix.
28. Trzepacz PT. Is there a final common neural pathway in delirium? Focus on acetylcholine and dopamine. Semin Clin Neuropsychiatry. 2000;5(2):132-148.
29. Inouye SK. The dilemma of delirium: clinical and research controversies regarding diagnosis and evaluation of delirium in hospitalized elderly medical patients. Am J Med. 1994;97(3):278-288.
30. Laurila JV, Laakkonen ML, Tilvis RS, et al. Predisposing and precipitating factors for delirium in a frail geriatric population. J Psychosom Res. 2008;65(3):249-254.
31. Morandi A, McCurley J, Vasilevskis EE, et al. Tools to detect delirium superimposed on dementia: a systematic review. J Am Geriatr Soc. 2012;60(11):2005-2013.
32. Kazmierski J, Kowman M, Banach M, et al. The use of DSM-IV and ICD-10 criteria and diagnostic scales for delirium among cardiac surgery patients: results from the IPDACS study. J Neuropsychiatry Clin Neurosci. 2010; 22(4):426-432.
33. Breitbart W, Rosenfeld B, Roth A, et al. The Memorial Delirium Rating Scale. J Pain Symptom Manage. 1997;13(3):128-137.
34. Wong CL, Holroyd-Leduc J, Simel DL, et al. Does this patient have delirium?: value of bedside instruments. JAMA. 2010;304(7):779-786.
35. Marcantonio ER, Flacker JM, Wright RJ, et al. Reducing delirium after hip fracture: a randomized trial. J Am Geriatr Soc. 2011;49(5):516-522.
36. Lundström M, Edlund A, Karlsson S, et al. A multifactorial intervention program reduces the duration of delirium, length of hospitalization, and mortality in delirious patients. J Am Geriatr Soc. 2005;53(4):622-628.
37. Lundström M, Olofsson B, Stenvall M, et al. Postoperative delirium in old patients with femoral neck fracture: a randomized intervention study. Aging Clin Exp Res. 2007; 19(3):178-186.
38. Kratz A. Use of the acute confusion protocol: a research utilization project. J Nurs Care Qual. 2008;23(4):331-337.
39. Prakanrattana U, Prapaitrakool S. Efficacy of risperidone for prevention of postoperative delirium in cardiac surgery. Anaesth Intensive Care. 2007;35(5):714-719.
40. Kaneko T, Cai J, Ishikura T, et al. Prophylactic consecutive administration of haloperidol can reduce the occurrence of postoperative delirium in gastrointestinal surgery. Yonago Acta Medica. 1999;42:179-184.
41. Larsen KA, Kelly SE, Stern TA, et al. Administration of olanzapine to prevent postoperative delirium in elderly joint-replacement patients: a randomized, controlled trial. Psychosomatics. 2010;51(5):409-418.
42. Kalisvaart KJ, de Jonghe JF, Bogaards MJ, et al. Haloperidol prophylaxis for elderly hip-surgery patients at risk for delirium: a randomized placebo-controlled study. J Am Geriatr Soc. 2005;53(10):1658-1666.
43. Hakim SM, Othman AI, Naoum DO. Early treatment with risperidone for subsyndromal delirium after on-pump cardiac surgery in the elderly: a randomized trial. Anesthesiology. 2012;116(5):987-997.
44. Maldonado JR, Wysong A, van der Starre PJ, et al. Dexmedetomidine and the reduction of postoperative delirium after cardiac surgery. Psychosomatics. 2009;50(3): 206-217.
45. Short J. Use of dexmedetomidine for primary sedation in a general intensive care unit. Crit Care Nurse. 2010;30(1): 29-38; quiz 39.
46. Practice guideline for the treatment of patients with delirium. American Psychiatric Association [Comment in: Treatment of patients with delirium. Am J Psychiatry. 2000.]. Am J Psychiatry. 1999;156(suppl 5):1-20.
47. Maldonado JR. Delirium in the acute care setting: characteristics, diagnosis, and treatment. Crit Care Clin. 2008;24(4):657-722, vii.
48. Platt MM, Breitbart W, Smith M, et al. Efficacy of neuroleptics for hypoactive delirium. J Neuropsychiatry Clin Neurosci. 1994;6(1):66-67.
49. Lonergan E, Britton AM, Luxenberg J, et al. Antipsychotics for delirium. Cochrane Database Syst Rev. 2007;(2):CD005594.
50. Seitz DP, Gill SS, van Zyl LT. Antipsychotics in the treatment of delirium: a systematic review. J Clin Psychiatry. 2007;68(1):11-21.
51. Breitbart W, Marotta R, Platt MM, et al. A double-blind trial of haloperidol, chlorpromazine, and lorazepam in the treatment of delirium in hospitalized AIDS patients. Am J Psychiatry. 1996;153(2):231-237.
52. Hu H, Deng W, Yang H, et al. Olanzapine and haloperidol for senile delirium: a randomized controlled observation. Chinese Journal of Clinical Rehabilitation. 2006;10(42): 188-190.
53. Friedman JH, Fernandez HH. Atypical antipsychotics in Parkinson-sensitive populations. J Geriatr Psychiatry Neurol. 2002;15(3):156-170.
54. Seitz DP, Gill SS. Neuroleptic malignant syndrome complicating antipsychotic treatment of delirium or agitation in medical and surgical patients: case reports and a review of the literature. Psychosomatics. 2009; 50(1):8-15.
55. Han CS, Kim YK. A double-blind trial of risperidone and haloperidol for the treatment of delirium. Psychosomatics. 2004;45(4):297-301.
56. Sink KM, Holden KF, Yaffe K. Pharmacological treatment of neuropsychiatric symptoms of dementia: a review of the evidence. JAMA. 2005;293(5):596-608.
57. Hermann N, Lanctôt KL. Atypical antipsychotics for neuropsychiatric symptoms of dementia: malignant or maligned? Drug Saf. 2006;29(10):833-843.
58. Noyan MA, Elbi H, Aksu H. Donepezil for anticholinergic drug intoxication: a case report. Prog Neuropsychopharmacol Biol Psychiatry. 2003;27(5):885-887.
59. Gleason OC. Donepezil for postoperative delirium. Psychosomatics. 2003;44(5):437-438.
60. Overshott R, Karim S, Burns A. Cholinesterase inhibitors for delirium. Cochrane Database Syst Rev. 2008;(1): CD005317.
61. Davis MP. Does trazodone have a role in palliating symptoms? Support Care Cancer. 2007;15(2):221-224.
62. Fick DM, Agostini JV, Inouye SK. Delirium superimposed on dementia: a systematic review. J Am Geriatr Soc. 2002; 50(10):1723-1732.
63. Brajtman S, Wright D, Hogan D, et al. Developing guidelines for the assessment and treatment of delirium in older adults at the end of life. Can Geriatr J. 2011;14(2):40-50.
64. Caraceni A, Simonetti F. Palliating delirium in patients with cancer. Lancet Oncol. 2009;10(2):164-172.
65. Alexopoulos GS, Streim J, Carpenter D, et al; Expert Consensus Panel for using Antipsychotic Drugs in Older Patients. Using antipsychotic agents in older patients. J Clin Psychiatry. 2004;65(suppl 2):5-99; discussion 100-102; quiz 103-104.
66. Granja C, Gomes E, Amaro A, et al. Understanding posttraumatic stress disorder-related symptoms after critical care: the early illness amnesia hypothesis. Crit Care Med. 2008;36(10):2801-2809.
67. Ringdal GI, Ringdal K, Juliebø V, et al. Using the Mini- Mental State Examination to screen for delirium in elderly patients with hip fracture. Dement Geriatr Cogn Disord. 2011;32(6):394-400.
68. Olson RA, Chhanabhai T, McKenzie M. Feasibility study of the Montreal Cognitive Assessment (MoCA) in patients with brain metastases. Support Care Cancer. 2008;16(11):1273-1278.
Obtaining informed consent for research in an acute inpatient psychiatric setting
Conducting clinical research with patients in an acute inpatient psychiatric setting raises possible ethical difficulties, in part because of concern about patients’ ability to give informed consent to participate in research.
We propose the acronym CHECK (for capacity, heredity, ethics, coercion-free, and knowledge) to provide researchers with guidance on the process of addressing informed consent in an acute inpatient setting.
Capacity. Ensure that the patient has the decisional capacity to:
• understand disclosed information about proposed research
• appreciate the impact of participation and nonparticipation
• reason about risks and benefits of participation
• communicate a consistent choice.1
The standards for disclosing information to a potential participant are higher for research than in clinical practice, because patients must understand and accept randomization, placebo control, blinding, and possible exposure to non-approved treatment interventions—yet there is a balance regarding how much information is necessary for consent in a given situation.2
Be mindful that the severity of the patient’s psychiatric illness can impair understanding and insight that might preclude giving informed consent (eg, major depression can produce a slowing of intellectual processes; mania can display distractibility; schizophrenia can compromise decisional capacity because of disorganized thinking or delusions; and neurocognitive disorders can affect the ability to process information).
The MacArthur Competence Assessment Tool for Clinical Research, designed as an aid to assessing capacity, has the most empirical support, although other instruments might be equally or better suited to some situations.1
Heredity. When undertaking human genetic and genomic research, create a precise, robust consent process. Genome sequencing studies can reveal information about the health of patients and their families, provoking discussion about appropriate protections for such data. Informed consent should include:
• how the data will be used now and in the future
• the extent to which patients can control future use of the data
• benefits and risks of participation, including the potential for unknown future risks
• what information, including incidental findings, will be returned to the patient
• what methods will be used to safeguard genetic testing data.3
Ethics. Researchers are bound by a code of ethics:
• Patients have the right to decline participation in research and to withdraw at any stage without prejudice; exclusion recognizes the need to protect those who may be incapable of exercising that right.2 Avoid research with dissenting patients, whether or not they are considered capable.2 Do not routinely invite treatment-refusing patients to participate in research projects, other than in extraordinary circumstances; eg, treatment refusing patients who have been adjudicated as “incompetent,” in which case the court-appointed surrogate decision-maker could be approached for informed consent. You should routinely seek a legal opinion in such a circumstance.
• Unless the research is examining interventions for acute and disabling psychiatric illness, consent should not be sought until patients are well enough to make an informed decision. However, clinical assessment is always needed (despite psychiatric illness category) because it cannot be assumed that psychiatric patients are unable to make such a decision (eg, in some cases, substance abuse should not automatically eliminate a participant, as long as the patient retains adequate cognitive status for informed consent).
• Capacity for consent is not “all-or-nothing,” but is specific to the research paradigm. In cases of impaired decisional capacity, researchers can obtain informed consent by obtaining agreement of family, legal representative, or caregiver; therefore, research with assenting adults, who are nonetheless incapable, is unlikely to be regarded as unethical.2
Coercion-free. Avoid covert pressures:
• Ensure that consent is given freely without coercion or duress. This is important if the participant has a physician-patient relationship with a member of the research team. Exercise caution when research methods involve physical contact. Such contact, in incapable patients—even those who assent— could create a medico-legal conflict (eg, taking a blood sample specific for research purposes without consent could result in a charge of battery).2 When in doubt, seek a legal opinion before enrolling decisionally incapable patients (and/or those adjudicated as incompetent) in research trials.
• Consider that participation be initiated by a third party (eg, an approach from a staff member who is not part of their care team and not involved in the research to ask if the potential participant has made a decision that he wants to have communicated to the researcher4).
• Require that a family member, legal representative, or caregiver be present at the time of consent with decisionally incapacitated patients.
Knowledge. The participant must be given adequate information about the project. Understand consent as an ongoing process occurring within a specific context:
• Give participants a fair explanation of the proposed project, the risks and benefits that might ensue, and, when applicable, what appropriate procedures may be offered if the participant experiences discomfort. If a study is to be blinded, patients must understand and appreciate that they could receive no benefit at all.
• Consider the importance of using appropriate language, repeating information, ensuring adequate time for questions and answers, and providing written material to the patient.2 Avoid leaving the patient alone with an information sheet to avoid coercion, because this risks denying patients the opportunity to participate because they lack the occasion to receive information and ask questions.4 Rather, go over the research consent document item by item with the patient in an iterative process, encouraging questions. Ensure private individual discussion between study team members and the patient to address questions related to the study.4
• Reapproach patients to discuss or revisit consent as needed, because their capacity to provide informed consent may vary over time. This is especially important in CNS illnesses, in which the level of cognitive function is variable. An item such as “consent status” for each encounter can be added to the checklist.
Disclosure
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Dunn LB, Nowrangi MA, Palmer BW, et al. Assessing decisional capacity for clinical research or treatment: a review of instruments. Am J Psychiatry. 2006;163(8): 1323-1334.
2. Fulford KW, Howse K. Ethics of research with psychiatric patients: principles, problems and the primary responsibilities of researchers. J Med Ethics. 1993;19(2):85-91.
3. Kuehn BM. Growing use of genomic data reveals need to improve consent and privacy standards. JAMA. 2013; 309(20):2083-2084.
4. Cameron J, Hart A. Ethical issues in obtaining informed consent for research from those recovering from acute mental health problems: a commentary. Research Ethics Review. 2007;3(4):127-129.
Conducting clinical research with patients in an acute inpatient psychiatric setting raises possible ethical difficulties, in part because of concern about patients’ ability to give informed consent to participate in research.
We propose the acronym CHECK (for capacity, heredity, ethics, coercion-free, and knowledge) to provide researchers with guidance on the process of addressing informed consent in an acute inpatient setting.
Capacity. Ensure that the patient has the decisional capacity to:
• understand disclosed information about proposed research
• appreciate the impact of participation and nonparticipation
• reason about risks and benefits of participation
• communicate a consistent choice.1
The standards for disclosing information to a potential participant are higher for research than in clinical practice, because patients must understand and accept randomization, placebo control, blinding, and possible exposure to non-approved treatment interventions—yet there is a balance regarding how much information is necessary for consent in a given situation.2
Be mindful that the severity of the patient’s psychiatric illness can impair understanding and insight that might preclude giving informed consent (eg, major depression can produce a slowing of intellectual processes; mania can display distractibility; schizophrenia can compromise decisional capacity because of disorganized thinking or delusions; and neurocognitive disorders can affect the ability to process information).
The MacArthur Competence Assessment Tool for Clinical Research, designed as an aid to assessing capacity, has the most empirical support, although other instruments might be equally or better suited to some situations.1
Heredity. When undertaking human genetic and genomic research, create a precise, robust consent process. Genome sequencing studies can reveal information about the health of patients and their families, provoking discussion about appropriate protections for such data. Informed consent should include:
• how the data will be used now and in the future
• the extent to which patients can control future use of the data
• benefits and risks of participation, including the potential for unknown future risks
• what information, including incidental findings, will be returned to the patient
• what methods will be used to safeguard genetic testing data.3
Ethics. Researchers are bound by a code of ethics:
• Patients have the right to decline participation in research and to withdraw at any stage without prejudice; exclusion recognizes the need to protect those who may be incapable of exercising that right.2 Avoid research with dissenting patients, whether or not they are considered capable.2 Do not routinely invite treatment-refusing patients to participate in research projects, other than in extraordinary circumstances; eg, treatment refusing patients who have been adjudicated as “incompetent,” in which case the court-appointed surrogate decision-maker could be approached for informed consent. You should routinely seek a legal opinion in such a circumstance.
• Unless the research is examining interventions for acute and disabling psychiatric illness, consent should not be sought until patients are well enough to make an informed decision. However, clinical assessment is always needed (despite psychiatric illness category) because it cannot be assumed that psychiatric patients are unable to make such a decision (eg, in some cases, substance abuse should not automatically eliminate a participant, as long as the patient retains adequate cognitive status for informed consent).
• Capacity for consent is not “all-or-nothing,” but is specific to the research paradigm. In cases of impaired decisional capacity, researchers can obtain informed consent by obtaining agreement of family, legal representative, or caregiver; therefore, research with assenting adults, who are nonetheless incapable, is unlikely to be regarded as unethical.2
Coercion-free. Avoid covert pressures:
• Ensure that consent is given freely without coercion or duress. This is important if the participant has a physician-patient relationship with a member of the research team. Exercise caution when research methods involve physical contact. Such contact, in incapable patients—even those who assent— could create a medico-legal conflict (eg, taking a blood sample specific for research purposes without consent could result in a charge of battery).2 When in doubt, seek a legal opinion before enrolling decisionally incapable patients (and/or those adjudicated as incompetent) in research trials.
• Consider that participation be initiated by a third party (eg, an approach from a staff member who is not part of their care team and not involved in the research to ask if the potential participant has made a decision that he wants to have communicated to the researcher4).
• Require that a family member, legal representative, or caregiver be present at the time of consent with decisionally incapacitated patients.
Knowledge. The participant must be given adequate information about the project. Understand consent as an ongoing process occurring within a specific context:
• Give participants a fair explanation of the proposed project, the risks and benefits that might ensue, and, when applicable, what appropriate procedures may be offered if the participant experiences discomfort. If a study is to be blinded, patients must understand and appreciate that they could receive no benefit at all.
• Consider the importance of using appropriate language, repeating information, ensuring adequate time for questions and answers, and providing written material to the patient.2 Avoid leaving the patient alone with an information sheet to avoid coercion, because this risks denying patients the opportunity to participate because they lack the occasion to receive information and ask questions.4 Rather, go over the research consent document item by item with the patient in an iterative process, encouraging questions. Ensure private individual discussion between study team members and the patient to address questions related to the study.4
• Reapproach patients to discuss or revisit consent as needed, because their capacity to provide informed consent may vary over time. This is especially important in CNS illnesses, in which the level of cognitive function is variable. An item such as “consent status” for each encounter can be added to the checklist.
Disclosure
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Conducting clinical research with patients in an acute inpatient psychiatric setting raises possible ethical difficulties, in part because of concern about patients’ ability to give informed consent to participate in research.
We propose the acronym CHECK (for capacity, heredity, ethics, coercion-free, and knowledge) to provide researchers with guidance on the process of addressing informed consent in an acute inpatient setting.
Capacity. Ensure that the patient has the decisional capacity to:
• understand disclosed information about proposed research
• appreciate the impact of participation and nonparticipation
• reason about risks and benefits of participation
• communicate a consistent choice.1
The standards for disclosing information to a potential participant are higher for research than in clinical practice, because patients must understand and accept randomization, placebo control, blinding, and possible exposure to non-approved treatment interventions—yet there is a balance regarding how much information is necessary for consent in a given situation.2
Be mindful that the severity of the patient’s psychiatric illness can impair understanding and insight that might preclude giving informed consent (eg, major depression can produce a slowing of intellectual processes; mania can display distractibility; schizophrenia can compromise decisional capacity because of disorganized thinking or delusions; and neurocognitive disorders can affect the ability to process information).
The MacArthur Competence Assessment Tool for Clinical Research, designed as an aid to assessing capacity, has the most empirical support, although other instruments might be equally or better suited to some situations.1
Heredity. When undertaking human genetic and genomic research, create a precise, robust consent process. Genome sequencing studies can reveal information about the health of patients and their families, provoking discussion about appropriate protections for such data. Informed consent should include:
• how the data will be used now and in the future
• the extent to which patients can control future use of the data
• benefits and risks of participation, including the potential for unknown future risks
• what information, including incidental findings, will be returned to the patient
• what methods will be used to safeguard genetic testing data.3
Ethics. Researchers are bound by a code of ethics:
• Patients have the right to decline participation in research and to withdraw at any stage without prejudice; exclusion recognizes the need to protect those who may be incapable of exercising that right.2 Avoid research with dissenting patients, whether or not they are considered capable.2 Do not routinely invite treatment-refusing patients to participate in research projects, other than in extraordinary circumstances; eg, treatment refusing patients who have been adjudicated as “incompetent,” in which case the court-appointed surrogate decision-maker could be approached for informed consent. You should routinely seek a legal opinion in such a circumstance.
• Unless the research is examining interventions for acute and disabling psychiatric illness, consent should not be sought until patients are well enough to make an informed decision. However, clinical assessment is always needed (despite psychiatric illness category) because it cannot be assumed that psychiatric patients are unable to make such a decision (eg, in some cases, substance abuse should not automatically eliminate a participant, as long as the patient retains adequate cognitive status for informed consent).
• Capacity for consent is not “all-or-nothing,” but is specific to the research paradigm. In cases of impaired decisional capacity, researchers can obtain informed consent by obtaining agreement of family, legal representative, or caregiver; therefore, research with assenting adults, who are nonetheless incapable, is unlikely to be regarded as unethical.2
Coercion-free. Avoid covert pressures:
• Ensure that consent is given freely without coercion or duress. This is important if the participant has a physician-patient relationship with a member of the research team. Exercise caution when research methods involve physical contact. Such contact, in incapable patients—even those who assent— could create a medico-legal conflict (eg, taking a blood sample specific for research purposes without consent could result in a charge of battery).2 When in doubt, seek a legal opinion before enrolling decisionally incapable patients (and/or those adjudicated as incompetent) in research trials.
• Consider that participation be initiated by a third party (eg, an approach from a staff member who is not part of their care team and not involved in the research to ask if the potential participant has made a decision that he wants to have communicated to the researcher4).
• Require that a family member, legal representative, or caregiver be present at the time of consent with decisionally incapacitated patients.
Knowledge. The participant must be given adequate information about the project. Understand consent as an ongoing process occurring within a specific context:
• Give participants a fair explanation of the proposed project, the risks and benefits that might ensue, and, when applicable, what appropriate procedures may be offered if the participant experiences discomfort. If a study is to be blinded, patients must understand and appreciate that they could receive no benefit at all.
• Consider the importance of using appropriate language, repeating information, ensuring adequate time for questions and answers, and providing written material to the patient.2 Avoid leaving the patient alone with an information sheet to avoid coercion, because this risks denying patients the opportunity to participate because they lack the occasion to receive information and ask questions.4 Rather, go over the research consent document item by item with the patient in an iterative process, encouraging questions. Ensure private individual discussion between study team members and the patient to address questions related to the study.4
• Reapproach patients to discuss or revisit consent as needed, because their capacity to provide informed consent may vary over time. This is especially important in CNS illnesses, in which the level of cognitive function is variable. An item such as “consent status” for each encounter can be added to the checklist.
Disclosure
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Dunn LB, Nowrangi MA, Palmer BW, et al. Assessing decisional capacity for clinical research or treatment: a review of instruments. Am J Psychiatry. 2006;163(8): 1323-1334.
2. Fulford KW, Howse K. Ethics of research with psychiatric patients: principles, problems and the primary responsibilities of researchers. J Med Ethics. 1993;19(2):85-91.
3. Kuehn BM. Growing use of genomic data reveals need to improve consent and privacy standards. JAMA. 2013; 309(20):2083-2084.
4. Cameron J, Hart A. Ethical issues in obtaining informed consent for research from those recovering from acute mental health problems: a commentary. Research Ethics Review. 2007;3(4):127-129.
1. Dunn LB, Nowrangi MA, Palmer BW, et al. Assessing decisional capacity for clinical research or treatment: a review of instruments. Am J Psychiatry. 2006;163(8): 1323-1334.
2. Fulford KW, Howse K. Ethics of research with psychiatric patients: principles, problems and the primary responsibilities of researchers. J Med Ethics. 1993;19(2):85-91.
3. Kuehn BM. Growing use of genomic data reveals need to improve consent and privacy standards. JAMA. 2013; 309(20):2083-2084.
4. Cameron J, Hart A. Ethical issues in obtaining informed consent for research from those recovering from acute mental health problems: a commentary. Research Ethics Review. 2007;3(4):127-129.
Taking the spice route: Psychoactive properties of culinary spices
Many substances that are not typically thought of as “substances of abuse” possess—when adequately dosed—clinically meaningful psychoactive properties. In addition to the more familiar effects of alcohol, psychostimulants, opioids, Cannabis, and hallucinogens, you may encounter psychiatric phenomena resulting from abuse of more obscure substances, including culinary spices.
The clinician treating a patient in an apparent intoxicated state who has a negative drug screen might ask that patient if he (she) abuses spices. This might be particularly relevant when treating patients thought to have limited access to illicit substances or those with ready access to large amounts of spices, such as prisoners, young patients, and those working in the food service industry.
Abuse of spices can be a problematic diagnosis
Patients may misuse culinary spices to achieve euphoria, or a “natural high.” They may present with medical or psychiatric symptoms, including acute altered mental status, but the psychoactive substances are not identified on routine toxicology studies. In addition, patients may not attribute their use of spices for psychoactive effect to “drugs,” because these materials are legal and readily available. This may lead to misdiagnosis of a systemic medical disorder or a primary psychiatric illness to explain the patient’s symptoms and initiating a psychotropic agent and other psychiatric services when a substance abuse program might be a more appropriate clinical intervention.
Some spices contain psychoactive compounds that can alter CNS function (Table1-7), might be abused for recreational purposes, and can be toxic in an excessive amount. Internet resources, including anonymous web-based communications, and anecdotal materials about non-traditional recreational drugs, are available to anyone with Internet access.8 However, little research has been conducted into the prevalence of abuse (Box)9 and spices’ psychoactive properties. The lack of toxicology detection of spices in the medical setting presents a diagnostic challenge.
The psychoactive plants used in “natural high” products mainly are psychoactively inactive in their natural form, but extracts or alkaloids obtained from them might induce 1 or more of 3 classifications of psychoactivity:
• stimulant
• sedative
• hallucinogenic.
Many of these substances are considered to be aphrodisiac, and some may be abused to increase sexual function.
The following is a review of common spices that have been reported to possess potential psychoactive properties.
Nutmeg
Nutmeg (Myristica fragrans) is a common and easily accessible means of reaching euphoria in adults.10 The aromatic oil of nutmeg contains myristicin, a psychoactive substance that is chemically similar to hallucinogenic compounds such as mescaline. Its psychoactive effects could be attributed to metabolic formation of amphetamine derivatives from its core ingredients, elemicin, myristicin, and safrole.11,12
Nutmeg and its active component, myristicin, produce central monoamine oxidase (MAO) inhibition as evidenced by the ability to lower the convulsive dose of IV tryptamine in mice and to increase brain 5-hydroxytryptamine concentrations.13,14 Although myristicin’s potency is not comparable to that of the more potent MAO inhibitors such as tranylcypromine and iproniazid (which is not available in the United States), it seems adequate when compared with its low toxicity.14 Nutmeg extract is associated with a significant antidepressant effect in mice, which seemed to be mediated by interaction with the adrenergic, dopaminergic, and serotonergic systems.13 Nutmeg is associated with sustained increase in sexual activity in animal studies, with no evidence of adverse effects and toxicity, suggesting that nutmeg possesses clinically significant aphrodisiac activity.15
Psychoactive effects can be achieved by ingesting 5 to 15 g of nutmeg.11 Acute nutmeg intoxication produces palpitations, dizziness, anxiety, and hallucinations, mostly resolving within 24 hours, while effects of chronic abuse are reported to be similar to Cannabis use, including euphoria, giddiness, anxiety, fear, sense of impending doom, detachment, confabulation, and hallucinations.11,16 Urine drug screens are negative unless other psychoactive substances have been ingested.17
Suspected nutmeg intoxication or poisoning should be treated with supportive treatment. Use sedatives with caution because of alternating periods of delirium and obtundation during nutmeg intoxication.17
In case reports, myristicin poisoning induced CNS neuromodulatory signs that mimicked an anticholinergic hyperstimulation state.12,18 Fatal myristicin poisoning is rare; 2 cases have been reported, 1 in combination with flunitrazepam (not available in the United States).19,20 Nutmeg also has sedative properties and can cause GI symptoms when ingesting excessive amounts.1,20,21 Grover et al21 described no harmful effects on blood pressure and electrocardiogram; however, Shah et al22 reported palpitations and dry mouth.
Vanilla
Vanilla (species of the genus Vanilla) contains piperonal, also known as heliotropin.1 Piperonal has aromatherapeutic qualities that might elevate mood and well-being. In the early 1990s, the Memorial Sloan- Kettering Cancer Center in New York City described heliotropin as a powerful aromatherapy tool. Patients who were undergoing an MRI in an environment scented with heliotropin demonstrated a 63% reduction in anxiety compared with those who were not exposed to fragrance.23 The Smell and Taste Treatment and Research Foundation in Chicago found that vanilla can promote sexual arousal.24
Short-term effects of vanillin—a major component of vanilla—include a feeling of relaxation and reduced stress; long-term use can produce an antidepressant effect.1 There are no reports of vanilla abuse to achieve these effects; however, patients might abuse vanilla extract because of its alcohol content (up to 35% ethanol).25
Fennel
The essential oil of fennel (Foeniculum vulgare) can be neurotoxic and epileptogenic. Skalli and colleagues recently reported a case of seizure induction in a young woman after ingesting cakes containing fennel oil.26 Fennel oil also has been reported to have significant interaction with the fluoroquinolone-type antibiotics. Be aware of adverse effects associated with fennel ingestion; question patients if atypical seizures or reactions to antibiotics occur.27
Spices such as fennel, dill, cinnamon, saffron, and anise also contain psychoactive substances that are chemically similar to myristicin, which can induce sedation, stimulation, or hallucinations.7
Black pepper
Piperine, which gives black pepper (Piper nigrum) its spiciness, enhances thermogenesis of lipid metabolism, accelerates energy metabolism, and increases serotonin and endorphin production in the brain.28 Black pepper is reported to potentiate γ-aminobutyric acid A receptor subtypes,29 and could present possible applications for treating insomnia, epilepsy, and anxiety disorders.
Cloves
Non-culinary uses of clove (Syzygium aromaticum, a tree in the myrtle family) include flavored cigarettes. However, in 2009 clove cigarettes were banned in the United States as part of a public policy to reduce the number of children who start smoking.30 Eugenol, which constitutes as much as 90% of the essential oil extracted from cloves (and is responsible for the aroma), can cause hepatotoxicity31 and palpitations32; it can be toxic in quantities as low as 5 mL.33 Eugenol is present in other spices, such as nutmeg and cinnamon, and has been reported to have sedative properties.1
Mace
Mace is made from the covering of nutmeg (Myristica fragrans) seeds. It has a strong aroma resembling that of nutmeg. Whole mace contains 4% to 14% of a volatile oil similar to that found in nutmeg. Because mace contains the same oils that make nutmeg psychoactive1 in excessive amounts—although nutmeg seeds are more potent—be aware of the psychoactive potential of mace.
CinnamonCassia cinnamon (Cinnamomum aromaticum) is spicier and tarter than Ceylon cinnamon (Cinnamomum zeylanicum), which has a more flowery aroma. The 2 types of cinnamon can be distinguished by their different chemical composition. Ceylon cinnamon contains eugenol and benzyl benzoate; cassia cinnamon contains coumarin.3 Eugenol is reported to have sedative effects.1 Coumarin is a precursor molecule in the synthesis of a number of synthetic anticoagulant pharmaceuticals, including coumadin. Because of the toxic component of coumarin, European health agencies have warned against consuming high amounts of cassia.34 There are no reports of side effects arising from the occasional use of cinnamon as a spice.
In a study by Frydman-Marom et al,35 cinnamon extract (CEppt) was found to act on the CNS by inhibiting development of Alzheimer’s disease in animal models.
Asarone
Asarone is found in the Asarum family of spices that includes Acorus calamus. Asarone is chemically similar to mescaline. Although anecdotal reports indicate that A. calamus is a hallucinogen, research shows no evidence that it contains hallucinogenic substances.36 Han et al37 reported an antidepressant effect with the essential oil and asarones for the rhizomes of Acorus tatarinowii. In animal studies, asarone was found to reduce spontaneous motor activity, and even in low doses, reduced anxiety without decreasing acuity of perception.38
Ginger
Ginger (Zingiber officinale) is regarded as a sedative, general stimulant, and aphrodisiac.1,4,5 Its main constituents are phenolic compounds such as gingerols and shogaols, and sesquiterpenes such as zingiberene.4 Ginger is an inhibitor of thromboxane synthetase, a property shared by tricyclic antidepressants.39
Research indicates that 9 compounds found in ginger may interact with the serotonin 5-HT1A receptor, suggesting a possible mechanism for reducing anxiety.40 A study by Nievergelt et al41 indicates that by binding to human serotonin receptors, ginger might influence GI function. Ginger extract contains a cholinergic and spasmogenic component, which provides a mechanistic insight for the prokinetic action of ginger.40
Turmeric
Turmeric (Curcuma longa) has been investigated for possible benefit in Alzheimer’s disease42; research into curcumin, the active substance of turmeric, is increasing. Although the original report was retracted after publication, curcumin was reported to selectively bind to human cannabinoid receptors type 1 (CB1) with nanomolar affinities and to function as an antagonist/inverse agonist.43 However, Gertsch et al44 found that curcumin did not interact functionally with the CB1 receptor, although this compound appears to share ability of the CB1 receptor inverse agonist.
Galangal
Major constituents identified in the galangal (or galanga) rhizome and leaf oil were 1,8-cineole, and β-pinene and camphor.6 Galangal, a member of the ginger (Zingiberaceae) family, interacts with MAO inhibitors, H2 receptor antagonists, and proton-pump inhibitors.1 Anxiolytic, hallucinogenic, and stimulant properties have been reported.1 An excessive amount can induce diarrhea, dizziness, nausea, and vomiting.1
Saffron
Stigma of saffron (a member of the family Iridaceae) was found to be significantly more effective than placebo and equally as efficacious as fluoxetine and imipramine in treating depression. Saffron petal was found to be significantly more effective than placebo and as effective as fluoxetine and saffron stigma in a recent systematic review.45-48
Asafetida
Asafetida (Ferula assa-foetida), when combined with valerian root, is used as a sedative to treat hyperactivity.2 The active ingredients of asafetida are the resin, endogenous gum, essential oil, propenyl-isobutylsulfide, umbelliferone, and vanillin. Several of the volatile constituents produce a sedative effect.2 Additive effects can occur between the hypotensive property of asafetida and dopamine receptor agonists such as bromocriptine mesylate. Use caution when combining asafetida in conjunction with a CNS depressant or a stimulant.2
Recommendations for treating spice-abusers
Patients may present to psychiatry services with psychological and physiological evidence of intoxication with culinary spices that may mimic 1) abuse of other substances, 2) primary psychiatric illness, and 3) primary medical illness. When you encounter a patient with a new psychiatric symptom, consider inquiring about the abuse of spices.
Patients might abuse more than 1 spice; a comprehensive screening approach might therefore be useful. Caution patients that ingesting these substance to excess can have harmful effects. Consider appropriate psychopharmacotherapy for underlying psychiatric symptoms to help patients who use spices maladaptively to self-medicate psychiatric symptoms.
Consider abuse of culinary spices in clinical presentations of psychiatric symptoms that do not seem adequate for a diagnosis of a primary anxiety, mood, or psychotic disorder, or in cases atypical psychiatric presentations that are—perhaps to your surprise—associated with negative toxicology studies for common, more familiar substances of abuse.
Physicians practicing in an environment where street drugs are difficult to obtain (eg, prisons) should consider monitoring for possible abuse of spices. Based on the available, albeit limited, literature, it appears that most culinary spice–associated intoxication can be managed:
• with an elevated level of clinical suspicion
• by ruling out other causes of intoxication
• using targeted, empirical psychopharmacotherapy to manage symptoms
• with supportive care that includes close psychiatric follow-up.
Consider comorbid abuse of other, more familiar substances of abuse in patients who misuse spices. As with inhalant abuse, the concept of “substance abuse” in clinical practice may need to be further expanded to include patients who abuse culinary spices. Patients could be screened for psychiatric illnesses known to increase the risk of substance abuse. These might include—but are not limited to:
• comorbid psychotic disorders
• mood disorders, particularly bipolar disorders
• trauma- and stressor-related disorders, particularly posttraumatic stress disorder
• personality disorders, particularly antisocial, borderline, and narcissistic personality disorders.
Pending the availability of population-based studies on abuse of culinary spices, the usual cautions regarding substance abuse seem to be appropriate when caring for these patients. Assessment for and management of comorbid psychiatric conditions is essential in the comprehensive psychiatric care of patients who abuse substances.
Last, general consideration of a 12-step recovery program appears warranted for these patients; the self-reflection and group support of such programs can be useful in helping patients control their use of these substances.
Bottom Line
Presentation of culinary spice intoxication can parallel that of other medical or psychiatric illnesses, or other drugs of abuse. Consideration and questioning for abuse of spices is necessary to ascertain the psychoactive effects of these substances when used surreptitiously. Management should follow substance abuse treatment protocols: inquiry into patterns of problematic use and readiness to change, assessment and management of psychiatric comorbidity, and referral to a recovery program.
Related Resources
• Srinivasan K. Role of spices beyond food flavoring: nutraceuticals with multiple health effects. Food Reviews International. 2005;21(2):167-188.
• Parthasarathi U, Hategan A, Bourgeois JA. Out of the cupboard and into the clinic: Nutmeg-induced mood disorder. Current Psychiatry. 2013;12(12):E1-E2.
Drug Brand Names
Bromocriptine mesylate • Parlodel Imipramine • Tofrani
Flunitrazepam • Rohypnol Iproniazid • Marsilid
Fluoxetine • Prozac Tranylcypromine • Parnate
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. O’Mahony Carey S. Psychoactive substances. A guide to ethnobotanical plants and herbs, synthetic chemicals, compounds and products. http://www.drugs.ie/ resourcesfiles/guides/Psychoactive_substances_low_res. pdf. Accessed March 4, 2014.
2. Asafetida. Applied Health. http://www.appliedhealth.com/index.php?option=com _content&view=article&id= 108207. Accessed March 4, 2014.
3. Jayatilaka A, Poole SK, Poole CF, et al. Simultaneous micro steam distillation/solvent extraction for the isolation of semivolatile flavor compounds from cinnamon and their separation by series coupled-column gas chromatography. Analytica Chimica Acta. 1995;302(2-3):147-162.
4. Spices. History & Special Collections UCLA Louise M. Darling Biomedical Library. http://unitproj.library.ucla. edu/biomed/spice/index.cfm?displayID=15. Accessed March 4, 2014.
5. Ginger action and uses. Ginger extract. Gingerols. MDidea Web site. http://www.mdidea.com/products/new/ new02108.html. Accessed March 4, 2014.
6. Raina VK, Srivastava SK, Syamasunder KV. The essential oil of ‘greater galangal’ [Alpinia galanga (L.) Willd.] from the lower Himalayan region of India. Flavour and Fragrance Journal. 2002;17(5):358-360.
7. Wenk G. Psychoactive spices - Bon appetite! http://www.psychologytoday.com/blog/your-brain-food/201008/ psychoactive-spices-bon-appetite. Published August 4, 2010. Accessed March 4, 2014.
8. Wax PM. Just a click away: recreational drug Web sites on the Internet. Pediatrics.2002;109(6):e96.
9. Forrester MB. Nutmeg intoxication in Texas, 1998-2004. Hum Exp Toxicol. 2005;24(11):563-566.
10. Abernethy MK, Becker LB. Acute nutmeg intoxication. Am J Emerg Med. 1992;10(5):429-430.
11. Brenner N, Frank OS, Knight E. Chronic nutmeg psychosis. J R Soc Med. 1993;86(3):179-180.
12. McKenna A, Nordt SP, Ryan J. Acute nutmeg poisoning. Eur J Emerg Med. 2004;11(4):240-241.
13. Dhingra D, Sharma A. Antidepressant-like activity of n-hexane extract of nutmeg (Myristica fragrans) seeds in mice. J Med Food. 2006;9(1):84-89.
14. Truitt EB Jr, Duritz G, Ebersberger EM. Evidence of monoamine oxidase inhibition by myristicin and nutmeg. Proc Soc Exp Biol Med. 1963;112:647-650.
15. Tajuddin, Ahmad S, Latif A, et al. An experimental study of sexual function improving effect of Myristica fragrans Houtt. (nutmeg). BMC Complement Altern Med. 2005;5:16.
16. Quin GI, Fanning NF, Plunkett PK. Nutmeg intoxication. J Accid Emerg Med. 1998;15(4):287-288.
17. Barceloux DG. Nutmeg (Myristica fragrans Houtt.) Dis Mon. 2009;55(6):373-379.
18. Demetriades AK, Wallman PD, McGuiness A, et al. Low cost, high risk: accidental nutmeg intoxication. Emerg Med J. 2005;22(3):223-225.
19. Weil A. The use of nutmeg as a psychotropic agent. Bull Narc. 1966;18(4):15-23. http://www.unodc.org/unodc/en/data-and-analysis/bulletin/bulletin_1966-01-01_4_ page003.html. Accessed March 5, 2013.
20. Stein U, Greyer H, Hentschel H. Nutmeg (myristicin) poisoning - report on a fatal case and a series of cases recorded by a poison information centre. Forensic Sci Int. 2001;118(1):87-90.
21. Grover JK, Khandkar S, Vats V, et al. Pharmacological studies on Myristica fragrans—antidiarrheal, hypnotic, analgesic and hemodynamic (blood pressure) parameters. Methods Find Exp Clin Pharmacol. 2002;24(10):675-680.
22. Shah AM, Calello DP, Quintero-Solivan J, et al. The not-so-nice spice: a teenage girl with palpitations and dry mouth. Pediatr Emerg Care. 2011;27(12):1205-1207.
23. Heliotropin. Polarized light microscopy digital image gallery. http://micro.magnet.fsu.edu/primer/techniques/ polarized/gallery/pages/heliotropinsmall.html. Accessed March 5, 2014.
24. Gage E. Romancing the bean. Budget Travel. http://articles.cnn.com/2007-09-11/travel/vanilla_1_vanilla-orchid-totonaca?_s=PM:TRAVEL. Published September 11, 2007. Updated September 16, 2012. Accessed March 5, 2014.
25. Mazor S, DesLauriers CA, Mycyk MB. Adolescent ethanol intoxication from vanilla extract ingestion: a case report. The Internet Journal of Family Practice. 2005;4(1). doi: 10.5580/bc.
26. Skalli S, Soulaymani Bencheikh R. Epileptic seizure induced by fennel essential oil. Epileptic Disord. 2011;13(3):345-347.
27. Zhu M, Wong PY, Li RC. Effect of oral administration of fennel (Foeniculum vulgare) on ciprofloxacin absorption and disposition in the rat. J Pharm Pharmacol. 1999;51(12):1391-1396.
28. Malini T, Arunakaran J, Aruldhas MM, et al. Effects of piperine on the lipid composition and enzymes of the pyruvate-malate cycle in the testis of the rat in vivo. Biochem Mol Biol Int. 1999;47(3):537-545.
29. Zaugg J, Baburin I, Hering S, et al. Identifying GABAA receptor ligands in black pepper by activity profiling, LC-TOFMS, and offline microprobe NMR. Planta Med. 2009; 75(9):888-889. doi: 10.1055/s-0029-1234276.
30. Flavored tobacco. FDA.gov. http://www.fda.gov/TobaccoProducts/ProtectingKidsfromTobacco/ FlavoredTobacco/default.htm. Published September 22, 2009. Updated March 21, 2013. Accessed March 18, 2014.
31. Fujisawa S, Atsumi T, Kadoma Y, et al. Antioxidant and prooxidant action of eugenol-related compounds and their cytotoxicity. Toxicology. 2002;177(1):39-54.
32. Eugenol oil overdose. New York Times Health Guide. http://health.nytimes.com/health/guides/poison/ eugenol-oil-overdose/overview.html. Accessed March 5, 2014.
33. Hartnoll G, Moore D, Douek D. Near fatal ingestion of oil of cloves. Arch Dis Child. 1993;69(3):392-393.
34. Harris E. NPR. German Christmas cookies pose health danger. http://www.npr.org/templates/story/story.php? storyId=6672644. Published December 25, 2006. Accessed March 5, 2014.
35. Frydman-Marom A, Levin A, Farfara D, et al. Orally administrated cinnamon extract reduces β-amyloid oligomerization and corrects cognitive impairment in Alzheimer’s disease animal models. PLoS One. 2011; 6(1):e16564. doi:10.1371/journal.pone.001656453.
36. Björnstad K, Helander A, Hultén P, et al. Bioanalytical investigation of asarone in connection with Acorus calamus oil intoxications. J Anal Toxicol. 2009;33(9):604-609.
37. Han P, Han T, Peng W, et al. Antidepressant-like effects of essential oil and asarone, a major essential oil component from the rhizome of Acorus tatarinowii. Pharm Biol. 2013;51(5):589-594.
38. Dandiya PC, Menon MK. Actions of asarone on behavior, stress, and hyperpyrexia, and its interaction with central stimulants. J Pharmacol Exp Ther. 1964;145:42-46.
39. Bockon J. Ginger: inhibition of thromboxane synthetase and stimulation of prostacyclin: relevance for medicine and psychiatry. Med Hypotheses. 1986;20(3):271-278.
40. Ghayur MN, Gilani AH. Pharmacological basis for the medicinal use of ginger in gastrointestinal disorders. Dig Dis Sci. 2005;50(10):1889-1897.
41. Nievergelt A, Huonker P, Schoop R, et al. Identification of serotonin 5-HT1A receptor partial agonists in ginger. Bioorg Med Chem. 2010;18(9):3345-3351.
42. Mishra A, Palanivelu K. The effect of curcumin (turmeric) on Alzheimer’s disease: an overview. Ann Indian Acad Neurol. 2008;11(1):13-19.
43. Seely KA, Levi MS, Prather PL. The dietary polyphenols trans-resveratrol and curcumin selectively bind human CB1 cannabinoid receptors with nanomolar affinities and function as antagonists/inverse agonists [retracted in: J Pharmacol Exp Ther. 2009;331(3):1147]. J Pharmacol Exp Ther. 2009;330(1): 31-39.
44. Gertsch J, Pertwee RG, Di Marzo V. Phytocannabinoids beyond the Cannabis plant – do they exist? Br J Pharmacol. 2010;160(3):523-529.
45. Dwyer AV, Whitten DL, Hawrelak JA. Herbal medicines, other than St. John’s Wort, in the treatment of depression: a systematic review. Altern Med Rev. 2011;16(1):40-49.
46. Moshiri E, Basti AA, Noorbala AA, et al. Crocus sativus L. (petal) in the treatment of mild-to-moderate depression: a double-blind, randomized and placebo controlled trial. Phytomedicine. 2006;13(9-10):607-611.
47. Noorbala AA, Akhondzadeh S, Tahmacebi-Pour N, et al. Hydro-alcoholic extract of Crocus sativus L. versus fluoxetine in the treatment of mild to moderate depression: a double-blind, randomized pilot trial. J Ethnopharmacol. 2005;97(2):281-284.
48. Akhondzadeh S, Tahmacebi-Pour N, Noorbala AA, et al. Crocus sativus L. in the treatment of mild to moderate depression: a double-blind, randomized, and placebo-controlled trial. Phytother Res. 2005;19(2):148-151.
Many substances that are not typically thought of as “substances of abuse” possess—when adequately dosed—clinically meaningful psychoactive properties. In addition to the more familiar effects of alcohol, psychostimulants, opioids, Cannabis, and hallucinogens, you may encounter psychiatric phenomena resulting from abuse of more obscure substances, including culinary spices.
The clinician treating a patient in an apparent intoxicated state who has a negative drug screen might ask that patient if he (she) abuses spices. This might be particularly relevant when treating patients thought to have limited access to illicit substances or those with ready access to large amounts of spices, such as prisoners, young patients, and those working in the food service industry.
Abuse of spices can be a problematic diagnosis
Patients may misuse culinary spices to achieve euphoria, or a “natural high.” They may present with medical or psychiatric symptoms, including acute altered mental status, but the psychoactive substances are not identified on routine toxicology studies. In addition, patients may not attribute their use of spices for psychoactive effect to “drugs,” because these materials are legal and readily available. This may lead to misdiagnosis of a systemic medical disorder or a primary psychiatric illness to explain the patient’s symptoms and initiating a psychotropic agent and other psychiatric services when a substance abuse program might be a more appropriate clinical intervention.
Some spices contain psychoactive compounds that can alter CNS function (Table1-7), might be abused for recreational purposes, and can be toxic in an excessive amount. Internet resources, including anonymous web-based communications, and anecdotal materials about non-traditional recreational drugs, are available to anyone with Internet access.8 However, little research has been conducted into the prevalence of abuse (Box)9 and spices’ psychoactive properties. The lack of toxicology detection of spices in the medical setting presents a diagnostic challenge.
The psychoactive plants used in “natural high” products mainly are psychoactively inactive in their natural form, but extracts or alkaloids obtained from them might induce 1 or more of 3 classifications of psychoactivity:
• stimulant
• sedative
• hallucinogenic.
Many of these substances are considered to be aphrodisiac, and some may be abused to increase sexual function.
The following is a review of common spices that have been reported to possess potential psychoactive properties.
Nutmeg
Nutmeg (Myristica fragrans) is a common and easily accessible means of reaching euphoria in adults.10 The aromatic oil of nutmeg contains myristicin, a psychoactive substance that is chemically similar to hallucinogenic compounds such as mescaline. Its psychoactive effects could be attributed to metabolic formation of amphetamine derivatives from its core ingredients, elemicin, myristicin, and safrole.11,12
Nutmeg and its active component, myristicin, produce central monoamine oxidase (MAO) inhibition as evidenced by the ability to lower the convulsive dose of IV tryptamine in mice and to increase brain 5-hydroxytryptamine concentrations.13,14 Although myristicin’s potency is not comparable to that of the more potent MAO inhibitors such as tranylcypromine and iproniazid (which is not available in the United States), it seems adequate when compared with its low toxicity.14 Nutmeg extract is associated with a significant antidepressant effect in mice, which seemed to be mediated by interaction with the adrenergic, dopaminergic, and serotonergic systems.13 Nutmeg is associated with sustained increase in sexual activity in animal studies, with no evidence of adverse effects and toxicity, suggesting that nutmeg possesses clinically significant aphrodisiac activity.15
Psychoactive effects can be achieved by ingesting 5 to 15 g of nutmeg.11 Acute nutmeg intoxication produces palpitations, dizziness, anxiety, and hallucinations, mostly resolving within 24 hours, while effects of chronic abuse are reported to be similar to Cannabis use, including euphoria, giddiness, anxiety, fear, sense of impending doom, detachment, confabulation, and hallucinations.11,16 Urine drug screens are negative unless other psychoactive substances have been ingested.17
Suspected nutmeg intoxication or poisoning should be treated with supportive treatment. Use sedatives with caution because of alternating periods of delirium and obtundation during nutmeg intoxication.17
In case reports, myristicin poisoning induced CNS neuromodulatory signs that mimicked an anticholinergic hyperstimulation state.12,18 Fatal myristicin poisoning is rare; 2 cases have been reported, 1 in combination with flunitrazepam (not available in the United States).19,20 Nutmeg also has sedative properties and can cause GI symptoms when ingesting excessive amounts.1,20,21 Grover et al21 described no harmful effects on blood pressure and electrocardiogram; however, Shah et al22 reported palpitations and dry mouth.
Vanilla
Vanilla (species of the genus Vanilla) contains piperonal, also known as heliotropin.1 Piperonal has aromatherapeutic qualities that might elevate mood and well-being. In the early 1990s, the Memorial Sloan- Kettering Cancer Center in New York City described heliotropin as a powerful aromatherapy tool. Patients who were undergoing an MRI in an environment scented with heliotropin demonstrated a 63% reduction in anxiety compared with those who were not exposed to fragrance.23 The Smell and Taste Treatment and Research Foundation in Chicago found that vanilla can promote sexual arousal.24
Short-term effects of vanillin—a major component of vanilla—include a feeling of relaxation and reduced stress; long-term use can produce an antidepressant effect.1 There are no reports of vanilla abuse to achieve these effects; however, patients might abuse vanilla extract because of its alcohol content (up to 35% ethanol).25
Fennel
The essential oil of fennel (Foeniculum vulgare) can be neurotoxic and epileptogenic. Skalli and colleagues recently reported a case of seizure induction in a young woman after ingesting cakes containing fennel oil.26 Fennel oil also has been reported to have significant interaction with the fluoroquinolone-type antibiotics. Be aware of adverse effects associated with fennel ingestion; question patients if atypical seizures or reactions to antibiotics occur.27
Spices such as fennel, dill, cinnamon, saffron, and anise also contain psychoactive substances that are chemically similar to myristicin, which can induce sedation, stimulation, or hallucinations.7
Black pepper
Piperine, which gives black pepper (Piper nigrum) its spiciness, enhances thermogenesis of lipid metabolism, accelerates energy metabolism, and increases serotonin and endorphin production in the brain.28 Black pepper is reported to potentiate γ-aminobutyric acid A receptor subtypes,29 and could present possible applications for treating insomnia, epilepsy, and anxiety disorders.
Cloves
Non-culinary uses of clove (Syzygium aromaticum, a tree in the myrtle family) include flavored cigarettes. However, in 2009 clove cigarettes were banned in the United States as part of a public policy to reduce the number of children who start smoking.30 Eugenol, which constitutes as much as 90% of the essential oil extracted from cloves (and is responsible for the aroma), can cause hepatotoxicity31 and palpitations32; it can be toxic in quantities as low as 5 mL.33 Eugenol is present in other spices, such as nutmeg and cinnamon, and has been reported to have sedative properties.1
Mace
Mace is made from the covering of nutmeg (Myristica fragrans) seeds. It has a strong aroma resembling that of nutmeg. Whole mace contains 4% to 14% of a volatile oil similar to that found in nutmeg. Because mace contains the same oils that make nutmeg psychoactive1 in excessive amounts—although nutmeg seeds are more potent—be aware of the psychoactive potential of mace.
CinnamonCassia cinnamon (Cinnamomum aromaticum) is spicier and tarter than Ceylon cinnamon (Cinnamomum zeylanicum), which has a more flowery aroma. The 2 types of cinnamon can be distinguished by their different chemical composition. Ceylon cinnamon contains eugenol and benzyl benzoate; cassia cinnamon contains coumarin.3 Eugenol is reported to have sedative effects.1 Coumarin is a precursor molecule in the synthesis of a number of synthetic anticoagulant pharmaceuticals, including coumadin. Because of the toxic component of coumarin, European health agencies have warned against consuming high amounts of cassia.34 There are no reports of side effects arising from the occasional use of cinnamon as a spice.
In a study by Frydman-Marom et al,35 cinnamon extract (CEppt) was found to act on the CNS by inhibiting development of Alzheimer’s disease in animal models.
Asarone
Asarone is found in the Asarum family of spices that includes Acorus calamus. Asarone is chemically similar to mescaline. Although anecdotal reports indicate that A. calamus is a hallucinogen, research shows no evidence that it contains hallucinogenic substances.36 Han et al37 reported an antidepressant effect with the essential oil and asarones for the rhizomes of Acorus tatarinowii. In animal studies, asarone was found to reduce spontaneous motor activity, and even in low doses, reduced anxiety without decreasing acuity of perception.38
Ginger
Ginger (Zingiber officinale) is regarded as a sedative, general stimulant, and aphrodisiac.1,4,5 Its main constituents are phenolic compounds such as gingerols and shogaols, and sesquiterpenes such as zingiberene.4 Ginger is an inhibitor of thromboxane synthetase, a property shared by tricyclic antidepressants.39
Research indicates that 9 compounds found in ginger may interact with the serotonin 5-HT1A receptor, suggesting a possible mechanism for reducing anxiety.40 A study by Nievergelt et al41 indicates that by binding to human serotonin receptors, ginger might influence GI function. Ginger extract contains a cholinergic and spasmogenic component, which provides a mechanistic insight for the prokinetic action of ginger.40
Turmeric
Turmeric (Curcuma longa) has been investigated for possible benefit in Alzheimer’s disease42; research into curcumin, the active substance of turmeric, is increasing. Although the original report was retracted after publication, curcumin was reported to selectively bind to human cannabinoid receptors type 1 (CB1) with nanomolar affinities and to function as an antagonist/inverse agonist.43 However, Gertsch et al44 found that curcumin did not interact functionally with the CB1 receptor, although this compound appears to share ability of the CB1 receptor inverse agonist.
Galangal
Major constituents identified in the galangal (or galanga) rhizome and leaf oil were 1,8-cineole, and β-pinene and camphor.6 Galangal, a member of the ginger (Zingiberaceae) family, interacts with MAO inhibitors, H2 receptor antagonists, and proton-pump inhibitors.1 Anxiolytic, hallucinogenic, and stimulant properties have been reported.1 An excessive amount can induce diarrhea, dizziness, nausea, and vomiting.1
Saffron
Stigma of saffron (a member of the family Iridaceae) was found to be significantly more effective than placebo and equally as efficacious as fluoxetine and imipramine in treating depression. Saffron petal was found to be significantly more effective than placebo and as effective as fluoxetine and saffron stigma in a recent systematic review.45-48
Asafetida
Asafetida (Ferula assa-foetida), when combined with valerian root, is used as a sedative to treat hyperactivity.2 The active ingredients of asafetida are the resin, endogenous gum, essential oil, propenyl-isobutylsulfide, umbelliferone, and vanillin. Several of the volatile constituents produce a sedative effect.2 Additive effects can occur between the hypotensive property of asafetida and dopamine receptor agonists such as bromocriptine mesylate. Use caution when combining asafetida in conjunction with a CNS depressant or a stimulant.2
Recommendations for treating spice-abusers
Patients may present to psychiatry services with psychological and physiological evidence of intoxication with culinary spices that may mimic 1) abuse of other substances, 2) primary psychiatric illness, and 3) primary medical illness. When you encounter a patient with a new psychiatric symptom, consider inquiring about the abuse of spices.
Patients might abuse more than 1 spice; a comprehensive screening approach might therefore be useful. Caution patients that ingesting these substance to excess can have harmful effects. Consider appropriate psychopharmacotherapy for underlying psychiatric symptoms to help patients who use spices maladaptively to self-medicate psychiatric symptoms.
Consider abuse of culinary spices in clinical presentations of psychiatric symptoms that do not seem adequate for a diagnosis of a primary anxiety, mood, or psychotic disorder, or in cases atypical psychiatric presentations that are—perhaps to your surprise—associated with negative toxicology studies for common, more familiar substances of abuse.
Physicians practicing in an environment where street drugs are difficult to obtain (eg, prisons) should consider monitoring for possible abuse of spices. Based on the available, albeit limited, literature, it appears that most culinary spice–associated intoxication can be managed:
• with an elevated level of clinical suspicion
• by ruling out other causes of intoxication
• using targeted, empirical psychopharmacotherapy to manage symptoms
• with supportive care that includes close psychiatric follow-up.
Consider comorbid abuse of other, more familiar substances of abuse in patients who misuse spices. As with inhalant abuse, the concept of “substance abuse” in clinical practice may need to be further expanded to include patients who abuse culinary spices. Patients could be screened for psychiatric illnesses known to increase the risk of substance abuse. These might include—but are not limited to:
• comorbid psychotic disorders
• mood disorders, particularly bipolar disorders
• trauma- and stressor-related disorders, particularly posttraumatic stress disorder
• personality disorders, particularly antisocial, borderline, and narcissistic personality disorders.
Pending the availability of population-based studies on abuse of culinary spices, the usual cautions regarding substance abuse seem to be appropriate when caring for these patients. Assessment for and management of comorbid psychiatric conditions is essential in the comprehensive psychiatric care of patients who abuse substances.
Last, general consideration of a 12-step recovery program appears warranted for these patients; the self-reflection and group support of such programs can be useful in helping patients control their use of these substances.
Bottom Line
Presentation of culinary spice intoxication can parallel that of other medical or psychiatric illnesses, or other drugs of abuse. Consideration and questioning for abuse of spices is necessary to ascertain the psychoactive effects of these substances when used surreptitiously. Management should follow substance abuse treatment protocols: inquiry into patterns of problematic use and readiness to change, assessment and management of psychiatric comorbidity, and referral to a recovery program.
Related Resources
• Srinivasan K. Role of spices beyond food flavoring: nutraceuticals with multiple health effects. Food Reviews International. 2005;21(2):167-188.
• Parthasarathi U, Hategan A, Bourgeois JA. Out of the cupboard and into the clinic: Nutmeg-induced mood disorder. Current Psychiatry. 2013;12(12):E1-E2.
Drug Brand Names
Bromocriptine mesylate • Parlodel Imipramine • Tofrani
Flunitrazepam • Rohypnol Iproniazid • Marsilid
Fluoxetine • Prozac Tranylcypromine • Parnate
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Many substances that are not typically thought of as “substances of abuse” possess—when adequately dosed—clinically meaningful psychoactive properties. In addition to the more familiar effects of alcohol, psychostimulants, opioids, Cannabis, and hallucinogens, you may encounter psychiatric phenomena resulting from abuse of more obscure substances, including culinary spices.
The clinician treating a patient in an apparent intoxicated state who has a negative drug screen might ask that patient if he (she) abuses spices. This might be particularly relevant when treating patients thought to have limited access to illicit substances or those with ready access to large amounts of spices, such as prisoners, young patients, and those working in the food service industry.
Abuse of spices can be a problematic diagnosis
Patients may misuse culinary spices to achieve euphoria, or a “natural high.” They may present with medical or psychiatric symptoms, including acute altered mental status, but the psychoactive substances are not identified on routine toxicology studies. In addition, patients may not attribute their use of spices for psychoactive effect to “drugs,” because these materials are legal and readily available. This may lead to misdiagnosis of a systemic medical disorder or a primary psychiatric illness to explain the patient’s symptoms and initiating a psychotropic agent and other psychiatric services when a substance abuse program might be a more appropriate clinical intervention.
Some spices contain psychoactive compounds that can alter CNS function (Table1-7), might be abused for recreational purposes, and can be toxic in an excessive amount. Internet resources, including anonymous web-based communications, and anecdotal materials about non-traditional recreational drugs, are available to anyone with Internet access.8 However, little research has been conducted into the prevalence of abuse (Box)9 and spices’ psychoactive properties. The lack of toxicology detection of spices in the medical setting presents a diagnostic challenge.
The psychoactive plants used in “natural high” products mainly are psychoactively inactive in their natural form, but extracts or alkaloids obtained from them might induce 1 or more of 3 classifications of psychoactivity:
• stimulant
• sedative
• hallucinogenic.
Many of these substances are considered to be aphrodisiac, and some may be abused to increase sexual function.
The following is a review of common spices that have been reported to possess potential psychoactive properties.
Nutmeg
Nutmeg (Myristica fragrans) is a common and easily accessible means of reaching euphoria in adults.10 The aromatic oil of nutmeg contains myristicin, a psychoactive substance that is chemically similar to hallucinogenic compounds such as mescaline. Its psychoactive effects could be attributed to metabolic formation of amphetamine derivatives from its core ingredients, elemicin, myristicin, and safrole.11,12
Nutmeg and its active component, myristicin, produce central monoamine oxidase (MAO) inhibition as evidenced by the ability to lower the convulsive dose of IV tryptamine in mice and to increase brain 5-hydroxytryptamine concentrations.13,14 Although myristicin’s potency is not comparable to that of the more potent MAO inhibitors such as tranylcypromine and iproniazid (which is not available in the United States), it seems adequate when compared with its low toxicity.14 Nutmeg extract is associated with a significant antidepressant effect in mice, which seemed to be mediated by interaction with the adrenergic, dopaminergic, and serotonergic systems.13 Nutmeg is associated with sustained increase in sexual activity in animal studies, with no evidence of adverse effects and toxicity, suggesting that nutmeg possesses clinically significant aphrodisiac activity.15
Psychoactive effects can be achieved by ingesting 5 to 15 g of nutmeg.11 Acute nutmeg intoxication produces palpitations, dizziness, anxiety, and hallucinations, mostly resolving within 24 hours, while effects of chronic abuse are reported to be similar to Cannabis use, including euphoria, giddiness, anxiety, fear, sense of impending doom, detachment, confabulation, and hallucinations.11,16 Urine drug screens are negative unless other psychoactive substances have been ingested.17
Suspected nutmeg intoxication or poisoning should be treated with supportive treatment. Use sedatives with caution because of alternating periods of delirium and obtundation during nutmeg intoxication.17
In case reports, myristicin poisoning induced CNS neuromodulatory signs that mimicked an anticholinergic hyperstimulation state.12,18 Fatal myristicin poisoning is rare; 2 cases have been reported, 1 in combination with flunitrazepam (not available in the United States).19,20 Nutmeg also has sedative properties and can cause GI symptoms when ingesting excessive amounts.1,20,21 Grover et al21 described no harmful effects on blood pressure and electrocardiogram; however, Shah et al22 reported palpitations and dry mouth.
Vanilla
Vanilla (species of the genus Vanilla) contains piperonal, also known as heliotropin.1 Piperonal has aromatherapeutic qualities that might elevate mood and well-being. In the early 1990s, the Memorial Sloan- Kettering Cancer Center in New York City described heliotropin as a powerful aromatherapy tool. Patients who were undergoing an MRI in an environment scented with heliotropin demonstrated a 63% reduction in anxiety compared with those who were not exposed to fragrance.23 The Smell and Taste Treatment and Research Foundation in Chicago found that vanilla can promote sexual arousal.24
Short-term effects of vanillin—a major component of vanilla—include a feeling of relaxation and reduced stress; long-term use can produce an antidepressant effect.1 There are no reports of vanilla abuse to achieve these effects; however, patients might abuse vanilla extract because of its alcohol content (up to 35% ethanol).25
Fennel
The essential oil of fennel (Foeniculum vulgare) can be neurotoxic and epileptogenic. Skalli and colleagues recently reported a case of seizure induction in a young woman after ingesting cakes containing fennel oil.26 Fennel oil also has been reported to have significant interaction with the fluoroquinolone-type antibiotics. Be aware of adverse effects associated with fennel ingestion; question patients if atypical seizures or reactions to antibiotics occur.27
Spices such as fennel, dill, cinnamon, saffron, and anise also contain psychoactive substances that are chemically similar to myristicin, which can induce sedation, stimulation, or hallucinations.7
Black pepper
Piperine, which gives black pepper (Piper nigrum) its spiciness, enhances thermogenesis of lipid metabolism, accelerates energy metabolism, and increases serotonin and endorphin production in the brain.28 Black pepper is reported to potentiate γ-aminobutyric acid A receptor subtypes,29 and could present possible applications for treating insomnia, epilepsy, and anxiety disorders.
Cloves
Non-culinary uses of clove (Syzygium aromaticum, a tree in the myrtle family) include flavored cigarettes. However, in 2009 clove cigarettes were banned in the United States as part of a public policy to reduce the number of children who start smoking.30 Eugenol, which constitutes as much as 90% of the essential oil extracted from cloves (and is responsible for the aroma), can cause hepatotoxicity31 and palpitations32; it can be toxic in quantities as low as 5 mL.33 Eugenol is present in other spices, such as nutmeg and cinnamon, and has been reported to have sedative properties.1
Mace
Mace is made from the covering of nutmeg (Myristica fragrans) seeds. It has a strong aroma resembling that of nutmeg. Whole mace contains 4% to 14% of a volatile oil similar to that found in nutmeg. Because mace contains the same oils that make nutmeg psychoactive1 in excessive amounts—although nutmeg seeds are more potent—be aware of the psychoactive potential of mace.
CinnamonCassia cinnamon (Cinnamomum aromaticum) is spicier and tarter than Ceylon cinnamon (Cinnamomum zeylanicum), which has a more flowery aroma. The 2 types of cinnamon can be distinguished by their different chemical composition. Ceylon cinnamon contains eugenol and benzyl benzoate; cassia cinnamon contains coumarin.3 Eugenol is reported to have sedative effects.1 Coumarin is a precursor molecule in the synthesis of a number of synthetic anticoagulant pharmaceuticals, including coumadin. Because of the toxic component of coumarin, European health agencies have warned against consuming high amounts of cassia.34 There are no reports of side effects arising from the occasional use of cinnamon as a spice.
In a study by Frydman-Marom et al,35 cinnamon extract (CEppt) was found to act on the CNS by inhibiting development of Alzheimer’s disease in animal models.
Asarone
Asarone is found in the Asarum family of spices that includes Acorus calamus. Asarone is chemically similar to mescaline. Although anecdotal reports indicate that A. calamus is a hallucinogen, research shows no evidence that it contains hallucinogenic substances.36 Han et al37 reported an antidepressant effect with the essential oil and asarones for the rhizomes of Acorus tatarinowii. In animal studies, asarone was found to reduce spontaneous motor activity, and even in low doses, reduced anxiety without decreasing acuity of perception.38
Ginger
Ginger (Zingiber officinale) is regarded as a sedative, general stimulant, and aphrodisiac.1,4,5 Its main constituents are phenolic compounds such as gingerols and shogaols, and sesquiterpenes such as zingiberene.4 Ginger is an inhibitor of thromboxane synthetase, a property shared by tricyclic antidepressants.39
Research indicates that 9 compounds found in ginger may interact with the serotonin 5-HT1A receptor, suggesting a possible mechanism for reducing anxiety.40 A study by Nievergelt et al41 indicates that by binding to human serotonin receptors, ginger might influence GI function. Ginger extract contains a cholinergic and spasmogenic component, which provides a mechanistic insight for the prokinetic action of ginger.40
Turmeric
Turmeric (Curcuma longa) has been investigated for possible benefit in Alzheimer’s disease42; research into curcumin, the active substance of turmeric, is increasing. Although the original report was retracted after publication, curcumin was reported to selectively bind to human cannabinoid receptors type 1 (CB1) with nanomolar affinities and to function as an antagonist/inverse agonist.43 However, Gertsch et al44 found that curcumin did not interact functionally with the CB1 receptor, although this compound appears to share ability of the CB1 receptor inverse agonist.
Galangal
Major constituents identified in the galangal (or galanga) rhizome and leaf oil were 1,8-cineole, and β-pinene and camphor.6 Galangal, a member of the ginger (Zingiberaceae) family, interacts with MAO inhibitors, H2 receptor antagonists, and proton-pump inhibitors.1 Anxiolytic, hallucinogenic, and stimulant properties have been reported.1 An excessive amount can induce diarrhea, dizziness, nausea, and vomiting.1
Saffron
Stigma of saffron (a member of the family Iridaceae) was found to be significantly more effective than placebo and equally as efficacious as fluoxetine and imipramine in treating depression. Saffron petal was found to be significantly more effective than placebo and as effective as fluoxetine and saffron stigma in a recent systematic review.45-48
Asafetida
Asafetida (Ferula assa-foetida), when combined with valerian root, is used as a sedative to treat hyperactivity.2 The active ingredients of asafetida are the resin, endogenous gum, essential oil, propenyl-isobutylsulfide, umbelliferone, and vanillin. Several of the volatile constituents produce a sedative effect.2 Additive effects can occur between the hypotensive property of asafetida and dopamine receptor agonists such as bromocriptine mesylate. Use caution when combining asafetida in conjunction with a CNS depressant or a stimulant.2
Recommendations for treating spice-abusers
Patients may present to psychiatry services with psychological and physiological evidence of intoxication with culinary spices that may mimic 1) abuse of other substances, 2) primary psychiatric illness, and 3) primary medical illness. When you encounter a patient with a new psychiatric symptom, consider inquiring about the abuse of spices.
Patients might abuse more than 1 spice; a comprehensive screening approach might therefore be useful. Caution patients that ingesting these substance to excess can have harmful effects. Consider appropriate psychopharmacotherapy for underlying psychiatric symptoms to help patients who use spices maladaptively to self-medicate psychiatric symptoms.
Consider abuse of culinary spices in clinical presentations of psychiatric symptoms that do not seem adequate for a diagnosis of a primary anxiety, mood, or psychotic disorder, or in cases atypical psychiatric presentations that are—perhaps to your surprise—associated with negative toxicology studies for common, more familiar substances of abuse.
Physicians practicing in an environment where street drugs are difficult to obtain (eg, prisons) should consider monitoring for possible abuse of spices. Based on the available, albeit limited, literature, it appears that most culinary spice–associated intoxication can be managed:
• with an elevated level of clinical suspicion
• by ruling out other causes of intoxication
• using targeted, empirical psychopharmacotherapy to manage symptoms
• with supportive care that includes close psychiatric follow-up.
Consider comorbid abuse of other, more familiar substances of abuse in patients who misuse spices. As with inhalant abuse, the concept of “substance abuse” in clinical practice may need to be further expanded to include patients who abuse culinary spices. Patients could be screened for psychiatric illnesses known to increase the risk of substance abuse. These might include—but are not limited to:
• comorbid psychotic disorders
• mood disorders, particularly bipolar disorders
• trauma- and stressor-related disorders, particularly posttraumatic stress disorder
• personality disorders, particularly antisocial, borderline, and narcissistic personality disorders.
Pending the availability of population-based studies on abuse of culinary spices, the usual cautions regarding substance abuse seem to be appropriate when caring for these patients. Assessment for and management of comorbid psychiatric conditions is essential in the comprehensive psychiatric care of patients who abuse substances.
Last, general consideration of a 12-step recovery program appears warranted for these patients; the self-reflection and group support of such programs can be useful in helping patients control their use of these substances.
Bottom Line
Presentation of culinary spice intoxication can parallel that of other medical or psychiatric illnesses, or other drugs of abuse. Consideration and questioning for abuse of spices is necessary to ascertain the psychoactive effects of these substances when used surreptitiously. Management should follow substance abuse treatment protocols: inquiry into patterns of problematic use and readiness to change, assessment and management of psychiatric comorbidity, and referral to a recovery program.
Related Resources
• Srinivasan K. Role of spices beyond food flavoring: nutraceuticals with multiple health effects. Food Reviews International. 2005;21(2):167-188.
• Parthasarathi U, Hategan A, Bourgeois JA. Out of the cupboard and into the clinic: Nutmeg-induced mood disorder. Current Psychiatry. 2013;12(12):E1-E2.
Drug Brand Names
Bromocriptine mesylate • Parlodel Imipramine • Tofrani
Flunitrazepam • Rohypnol Iproniazid • Marsilid
Fluoxetine • Prozac Tranylcypromine • Parnate
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. O’Mahony Carey S. Psychoactive substances. A guide to ethnobotanical plants and herbs, synthetic chemicals, compounds and products. http://www.drugs.ie/ resourcesfiles/guides/Psychoactive_substances_low_res. pdf. Accessed March 4, 2014.
2. Asafetida. Applied Health. http://www.appliedhealth.com/index.php?option=com _content&view=article&id= 108207. Accessed March 4, 2014.
3. Jayatilaka A, Poole SK, Poole CF, et al. Simultaneous micro steam distillation/solvent extraction for the isolation of semivolatile flavor compounds from cinnamon and their separation by series coupled-column gas chromatography. Analytica Chimica Acta. 1995;302(2-3):147-162.
4. Spices. History & Special Collections UCLA Louise M. Darling Biomedical Library. http://unitproj.library.ucla. edu/biomed/spice/index.cfm?displayID=15. Accessed March 4, 2014.
5. Ginger action and uses. Ginger extract. Gingerols. MDidea Web site. http://www.mdidea.com/products/new/ new02108.html. Accessed March 4, 2014.
6. Raina VK, Srivastava SK, Syamasunder KV. The essential oil of ‘greater galangal’ [Alpinia galanga (L.) Willd.] from the lower Himalayan region of India. Flavour and Fragrance Journal. 2002;17(5):358-360.
7. Wenk G. Psychoactive spices - Bon appetite! http://www.psychologytoday.com/blog/your-brain-food/201008/ psychoactive-spices-bon-appetite. Published August 4, 2010. Accessed March 4, 2014.
8. Wax PM. Just a click away: recreational drug Web sites on the Internet. Pediatrics.2002;109(6):e96.
9. Forrester MB. Nutmeg intoxication in Texas, 1998-2004. Hum Exp Toxicol. 2005;24(11):563-566.
10. Abernethy MK, Becker LB. Acute nutmeg intoxication. Am J Emerg Med. 1992;10(5):429-430.
11. Brenner N, Frank OS, Knight E. Chronic nutmeg psychosis. J R Soc Med. 1993;86(3):179-180.
12. McKenna A, Nordt SP, Ryan J. Acute nutmeg poisoning. Eur J Emerg Med. 2004;11(4):240-241.
13. Dhingra D, Sharma A. Antidepressant-like activity of n-hexane extract of nutmeg (Myristica fragrans) seeds in mice. J Med Food. 2006;9(1):84-89.
14. Truitt EB Jr, Duritz G, Ebersberger EM. Evidence of monoamine oxidase inhibition by myristicin and nutmeg. Proc Soc Exp Biol Med. 1963;112:647-650.
15. Tajuddin, Ahmad S, Latif A, et al. An experimental study of sexual function improving effect of Myristica fragrans Houtt. (nutmeg). BMC Complement Altern Med. 2005;5:16.
16. Quin GI, Fanning NF, Plunkett PK. Nutmeg intoxication. J Accid Emerg Med. 1998;15(4):287-288.
17. Barceloux DG. Nutmeg (Myristica fragrans Houtt.) Dis Mon. 2009;55(6):373-379.
18. Demetriades AK, Wallman PD, McGuiness A, et al. Low cost, high risk: accidental nutmeg intoxication. Emerg Med J. 2005;22(3):223-225.
19. Weil A. The use of nutmeg as a psychotropic agent. Bull Narc. 1966;18(4):15-23. http://www.unodc.org/unodc/en/data-and-analysis/bulletin/bulletin_1966-01-01_4_ page003.html. Accessed March 5, 2013.
20. Stein U, Greyer H, Hentschel H. Nutmeg (myristicin) poisoning - report on a fatal case and a series of cases recorded by a poison information centre. Forensic Sci Int. 2001;118(1):87-90.
21. Grover JK, Khandkar S, Vats V, et al. Pharmacological studies on Myristica fragrans—antidiarrheal, hypnotic, analgesic and hemodynamic (blood pressure) parameters. Methods Find Exp Clin Pharmacol. 2002;24(10):675-680.
22. Shah AM, Calello DP, Quintero-Solivan J, et al. The not-so-nice spice: a teenage girl with palpitations and dry mouth. Pediatr Emerg Care. 2011;27(12):1205-1207.
23. Heliotropin. Polarized light microscopy digital image gallery. http://micro.magnet.fsu.edu/primer/techniques/ polarized/gallery/pages/heliotropinsmall.html. Accessed March 5, 2014.
24. Gage E. Romancing the bean. Budget Travel. http://articles.cnn.com/2007-09-11/travel/vanilla_1_vanilla-orchid-totonaca?_s=PM:TRAVEL. Published September 11, 2007. Updated September 16, 2012. Accessed March 5, 2014.
25. Mazor S, DesLauriers CA, Mycyk MB. Adolescent ethanol intoxication from vanilla extract ingestion: a case report. The Internet Journal of Family Practice. 2005;4(1). doi: 10.5580/bc.
26. Skalli S, Soulaymani Bencheikh R. Epileptic seizure induced by fennel essential oil. Epileptic Disord. 2011;13(3):345-347.
27. Zhu M, Wong PY, Li RC. Effect of oral administration of fennel (Foeniculum vulgare) on ciprofloxacin absorption and disposition in the rat. J Pharm Pharmacol. 1999;51(12):1391-1396.
28. Malini T, Arunakaran J, Aruldhas MM, et al. Effects of piperine on the lipid composition and enzymes of the pyruvate-malate cycle in the testis of the rat in vivo. Biochem Mol Biol Int. 1999;47(3):537-545.
29. Zaugg J, Baburin I, Hering S, et al. Identifying GABAA receptor ligands in black pepper by activity profiling, LC-TOFMS, and offline microprobe NMR. Planta Med. 2009; 75(9):888-889. doi: 10.1055/s-0029-1234276.
30. Flavored tobacco. FDA.gov. http://www.fda.gov/TobaccoProducts/ProtectingKidsfromTobacco/ FlavoredTobacco/default.htm. Published September 22, 2009. Updated March 21, 2013. Accessed March 18, 2014.
31. Fujisawa S, Atsumi T, Kadoma Y, et al. Antioxidant and prooxidant action of eugenol-related compounds and their cytotoxicity. Toxicology. 2002;177(1):39-54.
32. Eugenol oil overdose. New York Times Health Guide. http://health.nytimes.com/health/guides/poison/ eugenol-oil-overdose/overview.html. Accessed March 5, 2014.
33. Hartnoll G, Moore D, Douek D. Near fatal ingestion of oil of cloves. Arch Dis Child. 1993;69(3):392-393.
34. Harris E. NPR. German Christmas cookies pose health danger. http://www.npr.org/templates/story/story.php? storyId=6672644. Published December 25, 2006. Accessed March 5, 2014.
35. Frydman-Marom A, Levin A, Farfara D, et al. Orally administrated cinnamon extract reduces β-amyloid oligomerization and corrects cognitive impairment in Alzheimer’s disease animal models. PLoS One. 2011; 6(1):e16564. doi:10.1371/journal.pone.001656453.
36. Björnstad K, Helander A, Hultén P, et al. Bioanalytical investigation of asarone in connection with Acorus calamus oil intoxications. J Anal Toxicol. 2009;33(9):604-609.
37. Han P, Han T, Peng W, et al. Antidepressant-like effects of essential oil and asarone, a major essential oil component from the rhizome of Acorus tatarinowii. Pharm Biol. 2013;51(5):589-594.
38. Dandiya PC, Menon MK. Actions of asarone on behavior, stress, and hyperpyrexia, and its interaction with central stimulants. J Pharmacol Exp Ther. 1964;145:42-46.
39. Bockon J. Ginger: inhibition of thromboxane synthetase and stimulation of prostacyclin: relevance for medicine and psychiatry. Med Hypotheses. 1986;20(3):271-278.
40. Ghayur MN, Gilani AH. Pharmacological basis for the medicinal use of ginger in gastrointestinal disorders. Dig Dis Sci. 2005;50(10):1889-1897.
41. Nievergelt A, Huonker P, Schoop R, et al. Identification of serotonin 5-HT1A receptor partial agonists in ginger. Bioorg Med Chem. 2010;18(9):3345-3351.
42. Mishra A, Palanivelu K. The effect of curcumin (turmeric) on Alzheimer’s disease: an overview. Ann Indian Acad Neurol. 2008;11(1):13-19.
43. Seely KA, Levi MS, Prather PL. The dietary polyphenols trans-resveratrol and curcumin selectively bind human CB1 cannabinoid receptors with nanomolar affinities and function as antagonists/inverse agonists [retracted in: J Pharmacol Exp Ther. 2009;331(3):1147]. J Pharmacol Exp Ther. 2009;330(1): 31-39.
44. Gertsch J, Pertwee RG, Di Marzo V. Phytocannabinoids beyond the Cannabis plant – do they exist? Br J Pharmacol. 2010;160(3):523-529.
45. Dwyer AV, Whitten DL, Hawrelak JA. Herbal medicines, other than St. John’s Wort, in the treatment of depression: a systematic review. Altern Med Rev. 2011;16(1):40-49.
46. Moshiri E, Basti AA, Noorbala AA, et al. Crocus sativus L. (petal) in the treatment of mild-to-moderate depression: a double-blind, randomized and placebo controlled trial. Phytomedicine. 2006;13(9-10):607-611.
47. Noorbala AA, Akhondzadeh S, Tahmacebi-Pour N, et al. Hydro-alcoholic extract of Crocus sativus L. versus fluoxetine in the treatment of mild to moderate depression: a double-blind, randomized pilot trial. J Ethnopharmacol. 2005;97(2):281-284.
48. Akhondzadeh S, Tahmacebi-Pour N, Noorbala AA, et al. Crocus sativus L. in the treatment of mild to moderate depression: a double-blind, randomized, and placebo-controlled trial. Phytother Res. 2005;19(2):148-151.
1. O’Mahony Carey S. Psychoactive substances. A guide to ethnobotanical plants and herbs, synthetic chemicals, compounds and products. http://www.drugs.ie/ resourcesfiles/guides/Psychoactive_substances_low_res. pdf. Accessed March 4, 2014.
2. Asafetida. Applied Health. http://www.appliedhealth.com/index.php?option=com _content&view=article&id= 108207. Accessed March 4, 2014.
3. Jayatilaka A, Poole SK, Poole CF, et al. Simultaneous micro steam distillation/solvent extraction for the isolation of semivolatile flavor compounds from cinnamon and their separation by series coupled-column gas chromatography. Analytica Chimica Acta. 1995;302(2-3):147-162.
4. Spices. History & Special Collections UCLA Louise M. Darling Biomedical Library. http://unitproj.library.ucla. edu/biomed/spice/index.cfm?displayID=15. Accessed March 4, 2014.
5. Ginger action and uses. Ginger extract. Gingerols. MDidea Web site. http://www.mdidea.com/products/new/ new02108.html. Accessed March 4, 2014.
6. Raina VK, Srivastava SK, Syamasunder KV. The essential oil of ‘greater galangal’ [Alpinia galanga (L.) Willd.] from the lower Himalayan region of India. Flavour and Fragrance Journal. 2002;17(5):358-360.
7. Wenk G. Psychoactive spices - Bon appetite! http://www.psychologytoday.com/blog/your-brain-food/201008/ psychoactive-spices-bon-appetite. Published August 4, 2010. Accessed March 4, 2014.
8. Wax PM. Just a click away: recreational drug Web sites on the Internet. Pediatrics.2002;109(6):e96.
9. Forrester MB. Nutmeg intoxication in Texas, 1998-2004. Hum Exp Toxicol. 2005;24(11):563-566.
10. Abernethy MK, Becker LB. Acute nutmeg intoxication. Am J Emerg Med. 1992;10(5):429-430.
11. Brenner N, Frank OS, Knight E. Chronic nutmeg psychosis. J R Soc Med. 1993;86(3):179-180.
12. McKenna A, Nordt SP, Ryan J. Acute nutmeg poisoning. Eur J Emerg Med. 2004;11(4):240-241.
13. Dhingra D, Sharma A. Antidepressant-like activity of n-hexane extract of nutmeg (Myristica fragrans) seeds in mice. J Med Food. 2006;9(1):84-89.
14. Truitt EB Jr, Duritz G, Ebersberger EM. Evidence of monoamine oxidase inhibition by myristicin and nutmeg. Proc Soc Exp Biol Med. 1963;112:647-650.
15. Tajuddin, Ahmad S, Latif A, et al. An experimental study of sexual function improving effect of Myristica fragrans Houtt. (nutmeg). BMC Complement Altern Med. 2005;5:16.
16. Quin GI, Fanning NF, Plunkett PK. Nutmeg intoxication. J Accid Emerg Med. 1998;15(4):287-288.
17. Barceloux DG. Nutmeg (Myristica fragrans Houtt.) Dis Mon. 2009;55(6):373-379.
18. Demetriades AK, Wallman PD, McGuiness A, et al. Low cost, high risk: accidental nutmeg intoxication. Emerg Med J. 2005;22(3):223-225.
19. Weil A. The use of nutmeg as a psychotropic agent. Bull Narc. 1966;18(4):15-23. http://www.unodc.org/unodc/en/data-and-analysis/bulletin/bulletin_1966-01-01_4_ page003.html. Accessed March 5, 2013.
20. Stein U, Greyer H, Hentschel H. Nutmeg (myristicin) poisoning - report on a fatal case and a series of cases recorded by a poison information centre. Forensic Sci Int. 2001;118(1):87-90.
21. Grover JK, Khandkar S, Vats V, et al. Pharmacological studies on Myristica fragrans—antidiarrheal, hypnotic, analgesic and hemodynamic (blood pressure) parameters. Methods Find Exp Clin Pharmacol. 2002;24(10):675-680.
22. Shah AM, Calello DP, Quintero-Solivan J, et al. The not-so-nice spice: a teenage girl with palpitations and dry mouth. Pediatr Emerg Care. 2011;27(12):1205-1207.
23. Heliotropin. Polarized light microscopy digital image gallery. http://micro.magnet.fsu.edu/primer/techniques/ polarized/gallery/pages/heliotropinsmall.html. Accessed March 5, 2014.
24. Gage E. Romancing the bean. Budget Travel. http://articles.cnn.com/2007-09-11/travel/vanilla_1_vanilla-orchid-totonaca?_s=PM:TRAVEL. Published September 11, 2007. Updated September 16, 2012. Accessed March 5, 2014.
25. Mazor S, DesLauriers CA, Mycyk MB. Adolescent ethanol intoxication from vanilla extract ingestion: a case report. The Internet Journal of Family Practice. 2005;4(1). doi: 10.5580/bc.
26. Skalli S, Soulaymani Bencheikh R. Epileptic seizure induced by fennel essential oil. Epileptic Disord. 2011;13(3):345-347.
27. Zhu M, Wong PY, Li RC. Effect of oral administration of fennel (Foeniculum vulgare) on ciprofloxacin absorption and disposition in the rat. J Pharm Pharmacol. 1999;51(12):1391-1396.
28. Malini T, Arunakaran J, Aruldhas MM, et al. Effects of piperine on the lipid composition and enzymes of the pyruvate-malate cycle in the testis of the rat in vivo. Biochem Mol Biol Int. 1999;47(3):537-545.
29. Zaugg J, Baburin I, Hering S, et al. Identifying GABAA receptor ligands in black pepper by activity profiling, LC-TOFMS, and offline microprobe NMR. Planta Med. 2009; 75(9):888-889. doi: 10.1055/s-0029-1234276.
30. Flavored tobacco. FDA.gov. http://www.fda.gov/TobaccoProducts/ProtectingKidsfromTobacco/ FlavoredTobacco/default.htm. Published September 22, 2009. Updated March 21, 2013. Accessed March 18, 2014.
31. Fujisawa S, Atsumi T, Kadoma Y, et al. Antioxidant and prooxidant action of eugenol-related compounds and their cytotoxicity. Toxicology. 2002;177(1):39-54.
32. Eugenol oil overdose. New York Times Health Guide. http://health.nytimes.com/health/guides/poison/ eugenol-oil-overdose/overview.html. Accessed March 5, 2014.
33. Hartnoll G, Moore D, Douek D. Near fatal ingestion of oil of cloves. Arch Dis Child. 1993;69(3):392-393.
34. Harris E. NPR. German Christmas cookies pose health danger. http://www.npr.org/templates/story/story.php? storyId=6672644. Published December 25, 2006. Accessed March 5, 2014.
35. Frydman-Marom A, Levin A, Farfara D, et al. Orally administrated cinnamon extract reduces β-amyloid oligomerization and corrects cognitive impairment in Alzheimer’s disease animal models. PLoS One. 2011; 6(1):e16564. doi:10.1371/journal.pone.001656453.
36. Björnstad K, Helander A, Hultén P, et al. Bioanalytical investigation of asarone in connection with Acorus calamus oil intoxications. J Anal Toxicol. 2009;33(9):604-609.
37. Han P, Han T, Peng W, et al. Antidepressant-like effects of essential oil and asarone, a major essential oil component from the rhizome of Acorus tatarinowii. Pharm Biol. 2013;51(5):589-594.
38. Dandiya PC, Menon MK. Actions of asarone on behavior, stress, and hyperpyrexia, and its interaction with central stimulants. J Pharmacol Exp Ther. 1964;145:42-46.
39. Bockon J. Ginger: inhibition of thromboxane synthetase and stimulation of prostacyclin: relevance for medicine and psychiatry. Med Hypotheses. 1986;20(3):271-278.
40. Ghayur MN, Gilani AH. Pharmacological basis for the medicinal use of ginger in gastrointestinal disorders. Dig Dis Sci. 2005;50(10):1889-1897.
41. Nievergelt A, Huonker P, Schoop R, et al. Identification of serotonin 5-HT1A receptor partial agonists in ginger. Bioorg Med Chem. 2010;18(9):3345-3351.
42. Mishra A, Palanivelu K. The effect of curcumin (turmeric) on Alzheimer’s disease: an overview. Ann Indian Acad Neurol. 2008;11(1):13-19.
43. Seely KA, Levi MS, Prather PL. The dietary polyphenols trans-resveratrol and curcumin selectively bind human CB1 cannabinoid receptors with nanomolar affinities and function as antagonists/inverse agonists [retracted in: J Pharmacol Exp Ther. 2009;331(3):1147]. J Pharmacol Exp Ther. 2009;330(1): 31-39.
44. Gertsch J, Pertwee RG, Di Marzo V. Phytocannabinoids beyond the Cannabis plant – do they exist? Br J Pharmacol. 2010;160(3):523-529.
45. Dwyer AV, Whitten DL, Hawrelak JA. Herbal medicines, other than St. John’s Wort, in the treatment of depression: a systematic review. Altern Med Rev. 2011;16(1):40-49.
46. Moshiri E, Basti AA, Noorbala AA, et al. Crocus sativus L. (petal) in the treatment of mild-to-moderate depression: a double-blind, randomized and placebo controlled trial. Phytomedicine. 2006;13(9-10):607-611.
47. Noorbala AA, Akhondzadeh S, Tahmacebi-Pour N, et al. Hydro-alcoholic extract of Crocus sativus L. versus fluoxetine in the treatment of mild to moderate depression: a double-blind, randomized pilot trial. J Ethnopharmacol. 2005;97(2):281-284.
48. Akhondzadeh S, Tahmacebi-Pour N, Noorbala AA, et al. Crocus sativus L. in the treatment of mild to moderate depression: a double-blind, randomized, and placebo-controlled trial. Phytother Res. 2005;19(2):148-151.
Out of the cupboard and into the clinic: Nutmeg-induced mood disorder
Clinicians often are unaware of a patient’s misuse or abuse of easily accessible substances such as spices, herbs, and natural supplements. This can lead to misdiagnosed severe psychiatric disorders and, more alarmingly, unnecessary use of long-term psychotropics and psychiatric services.
Excessive ingestion of nutmeg (Myristica fragrans) can produce psychiatric symptoms because it contains myristicin, a psychoactive substance, in its aromatic oil.1 It is structurally similar to other hallucinogenic compounds such as mescaline. The effects of nutmeg could be attributed to metabolic formation of amphetamine derivatives from its core ingredients: elemicin, myristicin, and safrole.1-3 However, neither amphetamine derivatives nor core ingredients are detected in the urine of patients suspected of abusing nutmeg, which makes diagnosis challenging.
We present a case of nutmeg abuse leading to psychotic depression.
Nutmeg and depression
Mr. D, age 50, is admitted to our inpatient psychiatric unit with severe dysphoria, hopelessness, persecutory delusions, suicidal ideation, and a sense of impending doom for the third time in 2 years. At previous admissions, he was diagnosed with bipolar disorder.
During his first admission, Mr. D reported an intentional overdose by water intoxication; laboratory studies revealed hyponatremia, liver dysfunction, abnormal cardiac markers, increased creatine kinase, and leukocytosis with neutrophilia. With supportive treatment, all parameters returned to the normal range within 7 days.
At his third admission, Mr. D describes an extensive history of nutmeg abuse. He reports achieving desirable psychoactive effects such as excitement, euphoria, enhanced sensory perceptions, and racing thoughts within a half hour of ingesting 1 teaspoon (5 g) of nutmeg; effects lasted for 6 hours. He reports that consuming 2 teaspoons (10 g) produced a “stronger” effect, and that 1 tablespoon (15 g) was associated with severe dysphoria, fear, psychosis, suicidal ideation, and behavior, which led to his psychiatric admissions. He denies any other substance use.
Urine drug screen and other routine laboratory investigations are negative. Symptoms resolve spontaneously within 3 days and he is managed without pharmacotherapy. The diagnosis is revised to substance-induced mood disorder with psychotic features. We provide psychoeducation about nutmeg’s psychoactive effects, and Mr. D is motivated to stop abusing nutmeg. Three years later he remains in good health.
Effects of nutmeg
Acute nutmeg intoxication produces anxiety, fear, and hallucinations, and generally is self-limited, with most symptoms resolving within 24 hours.2 Chronic effects of nutmeg abuse resemble those of marijuana abuse (Table 1). Acute and long-term physical effects are listed in Table 2.
Be alert for presentations of a ‘natural high’
Nutmeg, other spices, and herbs can be used by persons looking for a ”natural high”; as we saw with Mr. D, nutmeg abuse can present as a mood disorder resembling bipolar disorder. An acute, atypical presentation of mood changes or suicidal ideation should prompt you to investigate causes other than primary mood or psychotic disorders, and should include consideration of the effects of atypical drugs—and spices—of abuse.
Disclosure
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Weiss G. Hallucinogenic and narcotic-like effects of powdered Myristica (nutmeg). Psychiatr Q. 1960;34:346-356.
2. McKenna A, Nordt SP, Ryan J. Acute nutmeg poisoning. Eur J Emerg Med. 2004;11(4):240-241.
3. Brenner N, Frank OS, Knight E. Chronic nutmeg psychosis. J R Soc Med. 1993;86(3):179-180.
Clinicians often are unaware of a patient’s misuse or abuse of easily accessible substances such as spices, herbs, and natural supplements. This can lead to misdiagnosed severe psychiatric disorders and, more alarmingly, unnecessary use of long-term psychotropics and psychiatric services.
Excessive ingestion of nutmeg (Myristica fragrans) can produce psychiatric symptoms because it contains myristicin, a psychoactive substance, in its aromatic oil.1 It is structurally similar to other hallucinogenic compounds such as mescaline. The effects of nutmeg could be attributed to metabolic formation of amphetamine derivatives from its core ingredients: elemicin, myristicin, and safrole.1-3 However, neither amphetamine derivatives nor core ingredients are detected in the urine of patients suspected of abusing nutmeg, which makes diagnosis challenging.
We present a case of nutmeg abuse leading to psychotic depression.
Nutmeg and depression
Mr. D, age 50, is admitted to our inpatient psychiatric unit with severe dysphoria, hopelessness, persecutory delusions, suicidal ideation, and a sense of impending doom for the third time in 2 years. At previous admissions, he was diagnosed with bipolar disorder.
During his first admission, Mr. D reported an intentional overdose by water intoxication; laboratory studies revealed hyponatremia, liver dysfunction, abnormal cardiac markers, increased creatine kinase, and leukocytosis with neutrophilia. With supportive treatment, all parameters returned to the normal range within 7 days.
At his third admission, Mr. D describes an extensive history of nutmeg abuse. He reports achieving desirable psychoactive effects such as excitement, euphoria, enhanced sensory perceptions, and racing thoughts within a half hour of ingesting 1 teaspoon (5 g) of nutmeg; effects lasted for 6 hours. He reports that consuming 2 teaspoons (10 g) produced a “stronger” effect, and that 1 tablespoon (15 g) was associated with severe dysphoria, fear, psychosis, suicidal ideation, and behavior, which led to his psychiatric admissions. He denies any other substance use.
Urine drug screen and other routine laboratory investigations are negative. Symptoms resolve spontaneously within 3 days and he is managed without pharmacotherapy. The diagnosis is revised to substance-induced mood disorder with psychotic features. We provide psychoeducation about nutmeg’s psychoactive effects, and Mr. D is motivated to stop abusing nutmeg. Three years later he remains in good health.
Effects of nutmeg
Acute nutmeg intoxication produces anxiety, fear, and hallucinations, and generally is self-limited, with most symptoms resolving within 24 hours.2 Chronic effects of nutmeg abuse resemble those of marijuana abuse (Table 1). Acute and long-term physical effects are listed in Table 2.
Be alert for presentations of a ‘natural high’
Nutmeg, other spices, and herbs can be used by persons looking for a ”natural high”; as we saw with Mr. D, nutmeg abuse can present as a mood disorder resembling bipolar disorder. An acute, atypical presentation of mood changes or suicidal ideation should prompt you to investigate causes other than primary mood or psychotic disorders, and should include consideration of the effects of atypical drugs—and spices—of abuse.
Disclosure
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Clinicians often are unaware of a patient’s misuse or abuse of easily accessible substances such as spices, herbs, and natural supplements. This can lead to misdiagnosed severe psychiatric disorders and, more alarmingly, unnecessary use of long-term psychotropics and psychiatric services.
Excessive ingestion of nutmeg (Myristica fragrans) can produce psychiatric symptoms because it contains myristicin, a psychoactive substance, in its aromatic oil.1 It is structurally similar to other hallucinogenic compounds such as mescaline. The effects of nutmeg could be attributed to metabolic formation of amphetamine derivatives from its core ingredients: elemicin, myristicin, and safrole.1-3 However, neither amphetamine derivatives nor core ingredients are detected in the urine of patients suspected of abusing nutmeg, which makes diagnosis challenging.
We present a case of nutmeg abuse leading to psychotic depression.
Nutmeg and depression
Mr. D, age 50, is admitted to our inpatient psychiatric unit with severe dysphoria, hopelessness, persecutory delusions, suicidal ideation, and a sense of impending doom for the third time in 2 years. At previous admissions, he was diagnosed with bipolar disorder.
During his first admission, Mr. D reported an intentional overdose by water intoxication; laboratory studies revealed hyponatremia, liver dysfunction, abnormal cardiac markers, increased creatine kinase, and leukocytosis with neutrophilia. With supportive treatment, all parameters returned to the normal range within 7 days.
At his third admission, Mr. D describes an extensive history of nutmeg abuse. He reports achieving desirable psychoactive effects such as excitement, euphoria, enhanced sensory perceptions, and racing thoughts within a half hour of ingesting 1 teaspoon (5 g) of nutmeg; effects lasted for 6 hours. He reports that consuming 2 teaspoons (10 g) produced a “stronger” effect, and that 1 tablespoon (15 g) was associated with severe dysphoria, fear, psychosis, suicidal ideation, and behavior, which led to his psychiatric admissions. He denies any other substance use.
Urine drug screen and other routine laboratory investigations are negative. Symptoms resolve spontaneously within 3 days and he is managed without pharmacotherapy. The diagnosis is revised to substance-induced mood disorder with psychotic features. We provide psychoeducation about nutmeg’s psychoactive effects, and Mr. D is motivated to stop abusing nutmeg. Three years later he remains in good health.
Effects of nutmeg
Acute nutmeg intoxication produces anxiety, fear, and hallucinations, and generally is self-limited, with most symptoms resolving within 24 hours.2 Chronic effects of nutmeg abuse resemble those of marijuana abuse (Table 1). Acute and long-term physical effects are listed in Table 2.
Be alert for presentations of a ‘natural high’
Nutmeg, other spices, and herbs can be used by persons looking for a ”natural high”; as we saw with Mr. D, nutmeg abuse can present as a mood disorder resembling bipolar disorder. An acute, atypical presentation of mood changes or suicidal ideation should prompt you to investigate causes other than primary mood or psychotic disorders, and should include consideration of the effects of atypical drugs—and spices—of abuse.
Disclosure
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Weiss G. Hallucinogenic and narcotic-like effects of powdered Myristica (nutmeg). Psychiatr Q. 1960;34:346-356.
2. McKenna A, Nordt SP, Ryan J. Acute nutmeg poisoning. Eur J Emerg Med. 2004;11(4):240-241.
3. Brenner N, Frank OS, Knight E. Chronic nutmeg psychosis. J R Soc Med. 1993;86(3):179-180.
1. Weiss G. Hallucinogenic and narcotic-like effects of powdered Myristica (nutmeg). Psychiatr Q. 1960;34:346-356.
2. McKenna A, Nordt SP, Ryan J. Acute nutmeg poisoning. Eur J Emerg Med. 2004;11(4):240-241.
3. Brenner N, Frank OS, Knight E. Chronic nutmeg psychosis. J R Soc Med. 1993;86(3):179-180.
Sleep in hospitalized medical patients, Part 2: Behavioral and pharmacological management of sleep disturbances
In Part 1, we reviewed normal sleep architecture, and discussed the numerous factors that often disrupt the sleep of hospitalized medical patients. Effective management of sleep complaints among acutely ill patients includes a thorough assessment of medical and psychiatric conditions, medications and other psychosocial factors that may be directly or indirectly impairing sleep. In Part 2, we review and introduce an algorithm for assessing and managing sleep complaints in acutely ill hospitalized patients.
ASSESSMENT AND EVALUATION OF SLEEP COMPLAINTS
Assessment and evaluation of a sleep complaint begins with (Figure 1) an initial review of the medical record for documentation of the signs and symptoms of an underlying primary sleep disorder, which may be exacerbated during an acute medical illness. Common sleep disorders that are often overlooked include obstructive sleep apnea (OSA), restless leg syndrome (RLS), and periodic limb movement disorder (PLMD). Predisposing factors, characteristic clinical features, and differential diagnoses of these disorders are described in Table 1.
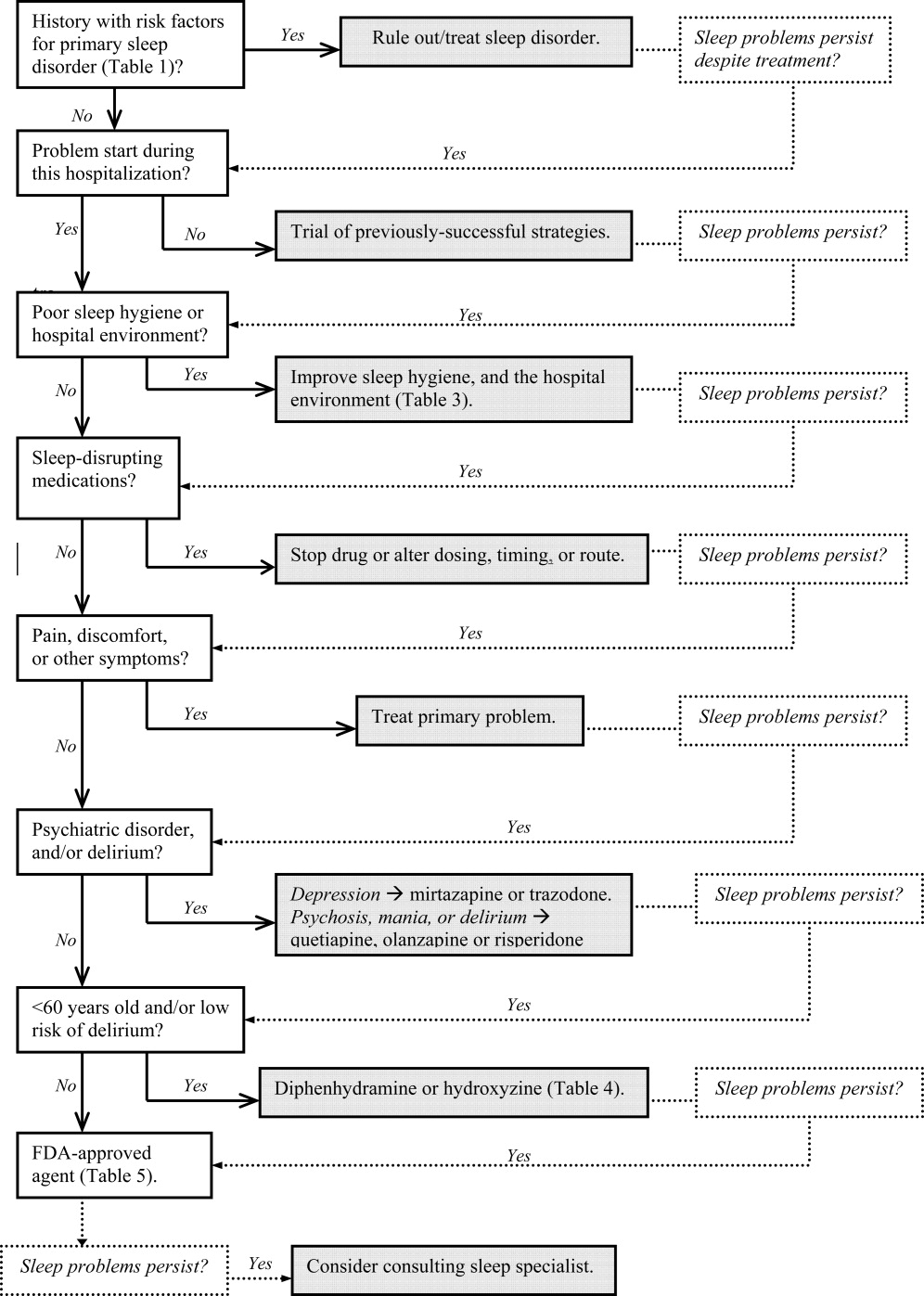
| Sleep Disorder | Predisposing Factors | Clinical Features | Differential Diagnosis |
|---|---|---|---|
| |||
| Obstructive sleep apnea (OSA) | Nasopharyngeal abnormalities, craniofacial abnormalities, obesity, >40 years old, men > women (2:1), neurologic disorder (eg, recent stroke) | Repetitive episodes of upper airway obstruction that occur during sleep, usually associated with oxygen desaturation. Episodes include loud snoring or gasps lasting 2030 seconds. Associated with morning headaches and dry mouth. | Sleep‐related laryngospasm, nocturnal gastroesophageal reflux, narcolepsy, hypersomnia, PLMD, central alveolar hypoventilation, paroxysmal nocturnal dyspnea, primary snoring, Cheyne‐Stokes ventilation, nocturnal asthma |
| Periodic limb movement disorder (PLMD) | OSA. RLS, or narcolepsy; aging; chronic uremia; TCAs or MAOIs; withdrawal from antiepileptic agents, or other sedating agents | Periodic episodes of repetitive and stereotyped limb movements: extension of the big toe with partial flexion of the ankles, knees, or hips. Muscle contractions last 0.5 to 5 seconds, with 20‐second to 40‐second intervals between them. | Sleep starts (occur just prior to, not during, sleep, and do not have a regular periodicity like PLMD), nocturnal epileptic seizures, myoclonic epilepsy |
| Restless leg syndrome (RLS) | Pregnancy (>20 weeks gestation), uremia, anemia, rheumatoid arthritis, peak onset is middle age | Uncomfortable leg sensations that occur prior to sleep onset that leads to an irresistible urge to move the legs. Described as achy, crawling, pulling, prickling, or tingling, and disrupts sleep onset. | Chronic myelopathy, peripheral neuropathy, akathisia, fasciculation syndromes, anemia |
| Sleep starts | Can worsen with anxiety, caffeine or other stimulants, daytime physical exertion | Sudden, brief contraction of the legs that occurs at sleep onset. Usually benign, but may worsen during hospitalization, and interfere with sleep. | PLMD, RLS, hyperekplexia syndrome, in which generalized myoclonus is readily elicited by stimuli |
Obtain a focused history by using questions listed in Table 2 to characterize the onset, duration, frequency, and specific characteristics of the patient's current sleep patterns. Next, establish whether the onset of the patient's sleep complaint began with the time of hospitalization. Subsequent questions can then focus on factors that may be impairing sleep such as the hospital environment and sleep hygiene behaviors by comparing the patient's home sleep habits with those during hospitalization. Inquire about the use or abuse of substances such as sedatives, antidepressants, sedatives, antiepileptic drugs (AEDs), and opioids. Ask questions about the presence of pain syndromes and other comorbidities that often impact sleep.
| Focus | Examples of Questions |
|---|---|
| |
| Sleep pattern | Do you have problems falling asleep or staying asleep? How often do you wake up during the night? How long does it take you to fall back asleep? When did the problem start? What can we do to help you sleep? What time do you try to go to sleep, and what time do you wake up? |
| Behavioral factors | Compare your bedtime routine at home, and in the hospital. |
| Environment | Does the lighting or noise level in the hospital disrupt your sleep? How so? Are you awoken from sleep for laboratory work, monitoring, bathing, or other nursing/medical procedures? |
| Patient comfort | Is your pain adequately controlled at night? If not, are you on a scheduled analgesic regimen, or do you have to ask for pain medications? Do you have breathing problems, gastroesophageal reflux, or other type of discomfort that keeps you from sleeping well? |
| Substances | Do you drink alcohol? How much, and how often? When was your last alcoholic beverage? Inquire about cocaine, methamphetamine, marijuana, and medically‐unsupervised use of opioids. |
| Psychosocial | How was your mood just prior to being hospitalized? How has your mood been since you were admitted? Have you experienced any emotionally or physically traumatic event prior to, or during, this hospitalization that continues to bother you (eg, intubation, resuscitation, surgery, blood draws, MRI scanning)? |
MANAGEMENT OF SLEEP COMPLAINTS
Management of sleep disturbance is multifactorial and consists of nonpharmacologic as well as pharmacologic therapies. A stepwise approach is suggested and begins with nonpharmacologic strategies.
Nonpharmacologic Interventions
Before using sedative/hypnotic agents, address sleep hygiene and other factors that disrupt sleep during a hospitalization such as those listed in Table 3.
| Barriers to Sleep | Strategies To Optimize Sleep in the Hospital |
|---|---|
| |
| Noise | Limit the volume level of television sets, and do not allow patients or visitors to increase the volume. |
| Promptly respond to alarm monitors, and consider liberalizing the monitor alarm setting, if appropriate. | |
| Keep patients' doors closed, if possible. | |
| Post signs to remind staff and visitors to minimize conversations at or near the bedside. | |
| Adhere strictly to visiting hours. | |
| Encourage staff to switch their beepers and other electronic devices to vibrate at night. | |
| Limit the number of visitors at a time and/or if appropriate, have the patient meet with visitors in another location (eg, conference room, cafeteria). | |
| Offer earplugs. | |
| Ask patients to turn their phone ringers off when visiting hours are over. | |
| Anxiety | Encourage visitors to minimize discussing emotionally difficult topics with patients near bedtime. |
| Lighting | Offer eye masks. |
| Encourage exposure to brighter light during the day (turn on the lights, open the curtains), and turn off the lights by 9 PM. | |
| Poor sleep hygiene | Encourage regular nocturnal sleep time, and discourage lengthy naps during the day. |
| Medications and substances | Minimize BzRAs for sleep. Try to wean patients off BzRAs prior to discharge. At discharge, provide the minimum number of pills until they are scheduled to see their primary care clinician posthospitalization, and do not provide refills. |
|
Avoid starting multiple medications at one time. Minimize use of sleep‐disrupting medications (see Part 1, Table 3). |
|
| Change medication regimens to promote sleep; eg, avoid night‐time diuretics if possible. | |
| No caffeine or cigarette smoking after 6 PM. | |
| Effects of treatments | Minimize bathing, dressing changes, room switches, and other activities at night. |
| Regularly review nighttime orders to see if you could decrease the frequency of overnight monitoring (eg, fingersticks, labdraws, checking vitals). | |
| Delirium | Provide an updated calendar to facilitate cognitive orientation. |
| Discontinue nonessential medications. Minimize use of BzRAs, barbiturates, opiates, antihistamines, and anticholinergic agents. | |
| Regularly provide verbal and other cues to orient patients to the date, time, location, and circumstances. | |
| Nocturnal discomfort | Optimize nighttime glycemic control, and maximize pain management. |
| For patients with reflux: No oral intake after 8 PM, and keep head of bed elevated 30 degrees. | |
| Provide nocturnal O2, CPAP, and/or other medications, as appropriate. If patient is on CPAP, assess the mask's fit and comfort. | |
Pharmacologic (Sedative/Hypnotic) Interventions
Pharmacologic therapy may be necessary to treat disordered sleep. The ideal sleep aid would reduce sleep latency or time to fall asleep, increase total sleep time (TST), not cause next‐day sedation, improve daytime functioning, and minimize the development of tolerance. Unfortunately, no single agent meets all these independent criteria. In the past 10 years, newer benzodiazepines (BzRAs) with shorter half‐lives have been shown to be efficacious in reducing sleep latency, but the problem of sleep maintenance without next‐day sedation persists.1 To choose an appropriate sleep agent, evaluate the drug's efficacy, mechanism of action, and side‐effect profile. Then, match these characteristics with the patient's clinical condition(s). In patients with comorbid sleep and psychiatric problems, consider using a sedating psychotropic at bedtime to promote sleep.
Non‐Food and Drug AdministrationApproved (Off‐Label) Sleep Aids: Psychotropic Medications
Limited data exist on the efficacy of non‐Food and Drug Administration (FDA)approved medications for insomnia,2 such as antidepressants and atypical antipsychotics (AAPs), and antihistamines; examples of which are listed in Table 4. The administration of antihistamines, barbiturates, chloral hydrate, and alternative/herbal therapies has been discouraged, because the benefits rarely outweigh the risks associated with their use. Currently, trazodone is the most commonly prescribed antidepressant for the treatment of insomnia, despite the relative lack of data regarding its use for insomnia.3 Prescription data suggest that trazodoneat hypnotic doses, which are lower than the full antidepressant doseis more commonly prescribed for insomnia rather than for its FDA‐approved use for depression.4 In general, sleep specialists refrain from recommending sedating antidepressants for primary insomnia due to insufficient data regarding efficacy and safety. In addition, trazodone has been associated with arrhythmias in patients with preexisting cardiac conduction system disease. Curry et al.3 speculated that trazodone is popular among prescribers because, unlike most BzRAs, trazodone does not have a recommended limited duration of use and is perceived as being safer than BzRAs. Walsh et al.5 conducted a randomized double‐blind, placebo‐controlled trial (n = 589) that compared the hypnotic efficacy and other sleep‐associated variables of trazodone (50 mg) and zolpidem (10 mg). During the first week of treatment, the subjects on trazodone or zolpidem decreased their time to fall asleep, or sleep latency, by 22% and 35%, respectively, compared to placebo. Sleep latency was significantly shorter on zolpidem (57.75 2.7 minutes) than for trazodone (57.7 + 4.0 minutes). By the second week, subjects on zolpidem continued to have a reduction in the time to fall asleep, but there was no significant difference between subjects on trazodone and placebo.5 Trazodone may be an acceptable short‐term alternative to BzRAs for patients with hypercapnia or hypoxemia, and in those with a history of drug abuse or dependence. At doses of 150 to 450 mg, trazodone may be an appropriate medication in patients with major depressive disorder and problems with sleep maintenance.6 Tolerance to trazodone's sedating property tends to develop after 2 weeks of treatment, however, so other treatments may need to be considered if sleep problems persist. The available data address relatively short‐term use of trazodone, so questions of safety and efficacy for chronic insomnia remain unanswered.
| Drug | Pertinent Side Effects | Comments |
|---|---|---|
| ||
| Antidepressants | ||
| Mirtazapine (Remeron) | Somnolence, appetite, weight, dry mouth | May be beneficial for comorbid depression and insomnia. Lower doses (15 mg) increase sedation. |
| Trazodone | Residual daytime sedation, headache, orthostatic hypotension, priapism, cardiac arrhythmias | May be beneficial for comorbid depression and insomnia. Not recommended as first‐line agent for insomnia.3 May be an alternative if BzRAs are contraindicated (severe hypercapnia or hypoxemia or history of substance abuse). Tolerance usually develops within 2 weeks. Lower doses (50100 mg) than when used for depression (400 mg). |
| TCAs | Delirium, cognition, seizure threshold, orthostatic hypotension, tachycardia, acquired prolonged QT syndrome, heart block, acute hepatitis | Avoid in hospitalized patients due to their anticholinergic, antihistaminic, and cardiovascular side effects. May be beneficial for comorbid depression and insomnia. |
| Antihistamines | ||
| Diphenhydramine (Benadryl) | Residual daytime sedation, delirium, orthostatic hypotension, psychomotor function, prolonged QT syndrome, blurred vision, urinary retention | Better than placebo to treat insomnia,12 but data is lacking to definitively endorse diphenhydramine for insomnia.13 Tolerance to antihistamines develops within a few days. Avoid in patients >60 years old.18 |
| Hydroxyzine | Drowsiness, dry mouth, dizziness, agitation, cognitive function | Efficacy as anxiolytic for >4 months use not established. Not FDA‐approved for insomnia. Avoid in patients >60 years old, closed‐angle glaucoma, prostatic hypertrophy, severe asthma, and COPD. |
| Antipsychotics | ||
| Quetiapine (Seroquel) | Sedation, orthostatic hypotension, hyperglycemia, appetite, weight, hyperlipidemia | The most sedating of the atypical antipsychotics, it is frequently used as a sleep aid. Not recommended for insomnia or other sleep problems unless there is a comorbid psychiatric disorder. Dosed lower (25100 mg) when used for insomnia versus for FDA‐approved indications (600 mg). |
| Olanzapine (Zyprexa) | Sedation, hyperglycemia, appetite, weight, hyperlipidemia | Of atypical antipsychotics, olanzapine is the most likely to cause metabolic complications. Should not be used solely for insomnia. |
| Barbiturate | ||
| Chloral hydrate | Oversedation, respiratory depression, nausea, vomiting, diarrhea, drowsiness, cognitive function, psychotic symptoms (paranoia, hallucinations), vertigo, dizziness, headache | Chloral hydrate has been used for the short‐term (<2 weeks) treatment of insomnia, but is currently not FDA‐approved for that indication. Additive CNS depression may occur if given with other sedative‐hypnotics. Caution in patients with severe cardiac disease. Contraindicated in marked hepatic or renal impairment. Highly lethal in overdose, and should be avoided in patients with risk of suicide. |
Mirtazapine (Remeron), which promotes both sleep and appetite, may be particularly helpful for patients with cancer, acquired immunodeficiency syndrome (AIDS), and other conditions in which the triad of poor sleep, anorexia, and depression are common. Mirtazapine is a noradrenergic and specific serotonergic agent that causes inverse, dose‐dependent sedation (doses 15 mg are less sedating).7 To target sleeplessness, start with a dose between 7.5 and 15 mg. If ineffective at this dose, it is unlikely that increasing the dose will be of benefit for sleep. A small randomized, double‐blind, placebo‐controlled trial found that low‐dose mirtazapine reduced the apnea‐hypopnea index (API) by half in newly‐diagnosed subjects with OSA (n = 12).8 The results were promising in terms of the use of mixed‐profile serotonergic drugs in treating OSA. However, as pointed out by the researchers, mirtazapine's tendency to cause weight gain, is problematic in this patient population.
Although sedating, tricyclic antidepressants (TCAs) should not be used to promote sleep in hospitalized patients. TCAs increase the risk of cardiac conduction abnormalities, decrease seizure threshold, and have significant anticholinergic and anti‐alpha‐adrenergic effects. In dementia patients, the anticholinergic effect of TCAs may precipitate delirium.
AAPs should not be used routinely as first‐line agents for insomnia, except in patients who are in the midst of acute manic or psychotic episodes.9 With chronic use of AAPs, the risks of hyperglycemia, hyperlipidemia, and weight gain outweigh the potential sleep benefits of these agents. AAPs, especially risperidone, may cause extrapyramidal syndrome (EPS). Risperidone, ziprasidone and quetiapine have been associated with prolonged QTc interval, but the relatively low doses of AAPs that are used purely for sedative purposes makes this risk relatively low. If a patient has a history of Parkinsonism or other EPS, risperidone should generally be avoided. If a patient treated with risperidone develops EPS, another AAP should be considered. A reasonable precaution is to obtain a pretreatment 12‐lead electrocardiogram. If the QTc is greater than 450 msec, consider using olanzapine rather than ziprasidone, risperidone, or quetiapine. Sedating AAPs include risperidone (Risperdal), olanzapine (Zyprexa), and quetiapine (Seroquel), with the latter 2 being especially sedating. Quetiapine may also cause orthostatic hypotension. The recent practice of using AAPs for delirium has not been reported to be associated with significant safety risks, probably because delirium treatment is typically of short duration under a period of close clinical observation. These agents should not be used indefinitely for insomnia without close monitoring of metabolic, psychiatric, and neurologic status. However, recent data suggest that the risk of serious adverse effects of AAPs may outweigh the potential benefits for the treatment of aggression or agitation in patients with Alzheimer's disease.10
A meta‐analysis of randomized placebo‐controlled trials of AAP use among dementia patients showed that overall, the use of AAP drugs for periods of less than 8 to 12 weeks was associated with a small increased risk for death compared with placebo.11 Data indicated that most patients' behaviors improved substantially during the first 1 to 4 weeks of treatment. In a double‐blind, placebo‐controlled trial, 421 patients with Alzheimer's disease and psychosis, aggression or agitation were randomly assigned to receive olanzapine (mean dose, 5.5 mg per day), quetiapine (mean dose, 56.5 mg per day), risperidone (mean dose, 1.0 mg per day), or placebo. Improvement was observed in 32% of patients assigned to olanzapine, 26% of patients assigned to quetiapine, 29% of patients assigned to risperidone, and 21% of patients assigned to placebo. A lower, but significant, proportion of the patients (24%, 16%, 18%, and 5%, respectively) discontinued these medications due to intolerable side effects. Thus, if minimal improvement is observed even after 8 weeks of treatment, prescribers should consider discontinuing the AAP. The management of agitation in dementia, particularly in the elderly, calls for an integrative and creative psychopharmacological approach, including the use of antidepressants, nonbenzodiazepine anxiolytics such as buspirone, and mood stabilizers such as divalproex sodium (Depakote) before exposing patients to the risks of AAPs.
Antihistamines are the most commonly used over‐the‐counter agents for chronic insomnia.1 Diphenhydramine (Benadryl) has been shown to be better than placebo to treat insomnia,12 but data is lacking to definitively endorse its use to promote sleep.13 Diphenhydramine is also limited by the development of tolerance within a few days of daily use. The anticholinergic action of antihistamines may lead to orthostatic hypotension, urinary retention, and may induce delirium in vulnerable patients. Therefore, diphenhydramine should be avoided in hospitalized patients.
Recent data suggest that hydroxyzine, an antihistamine, may be an appropriate sleep aid for patients with hepatic encephalopathy in whom BzRAs are contraindicated.14 Subjective improvement in sleep was observed in 40% of hydroxyzine‐treated patients with hepatic encephalopathy compared to placebo.
Chloral hydrate is one of the Western world's oldest known sedative‐hypnotics and was commonly used as a sleep aid through the 1970s.15 Chloral hydrate was eventually supplanted by BzRAs,16 and fell out of favor as a sleep aid due to its relatively high tolerance rate, drug‐drug interaction profile, and the high risk of death in an overdose. Doses of 500 to 1000 mg sufficed to promote sleep in most of the hospitalized subjects. More recent data regarding its use for treating insomnia are not available, but chloral hydrate may be an alternative short‐term treatment for insomnia in selected hospitalized patients. Because of its high‐risk profile, chloral hydrate would be used as a last‐resort medication, preferably with input from critical care and/or sleep medicine specialists.
FDA‐Approved Sleep Aids
As shown in Table 5, the FDA has approved 3 classes of medications for the treatment of insomnia: benzodiazepine gamma‐aminobutyric acid (GABA)A receptor agonists (BzRAs), nonbenzodiazepine GABAA receptor agonists (non‐BzRAs), and melatonin‐receptor agonists.17 BzRAs include estazolam (ProSom), flurazepam (Dalmane), quazepam (Doral), temazepam (Restoril), and triazolam (Halcion). Though BzRAs decrease sleep latency, increase TST, and decrease slow wave or deep sleep, they also have adverse side effects such as daytime sedation, anterograde amnesia, cognitive impairment, motor incoordination, dependence, tolerance, and rebound insomnia.18 Because of these side effects, BzRAs should be limited to generally healthy, young (ie, <45 years old) patients who are expected to have brief hospital stays.
| Drugs | Adult Dose (mg) | Half‐Life (hours)* | Onset (minutes) | Peak Effect (hours) | Major Effects/Clinical Comments |
|---|---|---|---|---|---|
| |||||
| BzRAs | Caution in elderly patients. Tolerance to BzRAs develop to the sedative, hypnotic, and anticonvulsant effects. | ||||
| Estazolam (ProSom) | 12 | 1024 | 60 | 0.51.5 | Short‐term (710 days) treatment for frequent arousals, early morning awakening. Not as useful for sleep onset. Avoid in patients with OSA. Caution in elderly patients, liver disease. High doses can cause respiratory depression. |
| Flurazepam (Dalmane) | 1530 | 47100 | 1520 | 36 | In general, avoid in hospitalized medical patients, especially elderly patients. |
| Quazepam (Doral) | 7.515 | 25114 | 1.5 | In general, avoid in hospitalized medical patients, especially elderly patients. | |
| Temazepam (Restoril) | 1530 | 616 | 23 | Short‐term (710 days) treatment for sleep onset and maintenance. Doses 30 mg/day: morning grogginess, nausea, headache, and vivid dreaming. | |
| Triazolam (Halcion) | 0.1250.25 | 1.55.5 | 1530 | 1.75 | Maximum dose is 0.5 mg. Short‐term (710 days) treatment. Rapid onset; should be in bed when taking medication. Contraindicated with atazanavir, ketoconazole, itraconazole, nefazodone, ritonavir. |
| Non‐BzRAs | |||||
| Eszopiclone (Lunesta) | 23 | 69 | 1 | In elderly: difficulty falling asleep, then initial: 1 mg; maximum 2 mg. Difficulty staying asleep: 2 mg. Rapid onset; should be in bed when taking medication. For faster sleep onset, do not ingest with high‐fat foods. No tolerance after 6 months. | |
| Zaleplon (Sonata) | 520 | 1 | Rapid | 1 | Short‐term (710 days) treatment for falling asleep and/or next‐day wakefulness is crucial (eg, shift workers). |
| Zopiclone (Imovane) | 515 | 3.86.5 (510 in elderly) | 30 | <2 | Transient and short‐term (710 days) treatment. Contraindicated in severe respiratory impairment. Caution in liver disease and depression; elderly prone to side effects. Anticholinergic agents may plasma level. |
| Zolpidem (Ambien) | 520 | 1.44.5 | 30 | 2 | Short‐term (710 days) treatment for sleep onset and maintenance. Rapid onset; should be in bed when taking medication. For faster sleep onset, do not ingest with food. No tolerance after 50 weeks. |
| Melatonin agonist | |||||
| Ramelton (Rozerem) | 8 | 12 | 30 | 11.5 | For sleep onset. For faster sleep onset, do not ingest with high‐fat foods. No tolerance. Contraindicated with fluvoxamine. |
Efficacy and safety studies have generally been limited to healthy, younger individuals without a history of primary sleep disorder. Potential adverse effects of BzRAs may become even more pronounced in hospitalized medical patients due to older age, acute illness, cointeraction drugs, and multidrug regimens. Although BzRAs are FDA‐approved for the treatment of insomnia, flurazepam and quazepam should generally be avoided in hospitalized patients. These agents' long half‐lives increase the risk of drug‐drug interactions and adverse events such as respiratory depression, cognitive decline, and delirium in acutely ill patients. For similar reasons, other long‐acting BzRAs such as clonazepam (Klonopin) and diazepam (Valium) should also not be used to treat insomnia in hospitalized patients. An exception to this is a patient with RLS, in which clonazepam is an approved treatment. However, now that ropinirole HCl (Requip) is FDA‐approved for RLS, BzARs may be able to be avoided. Lorazepam (Ativan), due to its relatively short half‐life and its anxiolytic property, is frequently used to treat insomnia in hospitalized medical patients.18 Start with the lowest dose possible (eg, 0.5 mg) as a one‐time‐only order, or on a as needed basis for 3 days. Alprazolam (Xanax), a potent, fast‐acting BzRA with a relatively short half‐life, has developed a reputation as being notoriously addictive, and experts feel alprazolam has similar potential for withdrawal and rebound.19, 20
The use of BzRAs should be minimized in all patients, and avoided in the elderly or those with a particularly high risk for delirium (eg, traumatic brain injury, stroke, multiple new medications). All BzRAs should be avoided in patients with a prior history of sedative‐hypnotic and/or alcohol dependence unless medically indicated, such as in alcohol withdrawal. Refrain from ordering nightly scheduled BzRAs without a specific time limit to ensure that sedative‐hypnotic use is closely monitored.
For the past 2 decades, physicians have been advised against using long‐acting BzRAs in the elderly (>65 years old) due to the increased risks of hip fractures, falls, motor vehicle accidents, daytime sedation, and adverse cognitive events such as delirium.2124 A large 5‐year prospective study in Quebec found that the risk of injury varied by the BzRA, and was independent of half‐life.25 Importantly, the risk of injury was dose‐dependent: the higher the dose of oxazepam, flurazepam, or chlordiazepoxide, the higher the risk of injury in the elderly.
Non‐BzRAs seem to have a superior side‐effect profile when compared to BzRAs, but should also be used with caution in the elderly. Non‐BzRAs include eszopiclone (Lunesta), zaleplon (Sonata), zolpidem (Ambien), and zolpidem extended‐release. The number of comparison studies is limited, but the available data reveal that: (1) zolpidem (Ambien) may be better than temazepam (Restoril) in terms of sleep latency and quality; and (2) zaleplon (Sonata) may lead to a shorter sleep latency than zolpidem (Ambien), but the latter is associated with longer sleep duration.26 Non‐BzRAs have less next‐day sedation, psychomotor dysfunction, tolerance/withdrawal, and rapid‐eye‐movement (REM) sleep rebound; and lower abuse potential than BzRAs.27
The most commonly prescribed hypnotic, zolpidem has a short half‐life, and seems to reduce sleep latency with minimal residual side effects when compared to BzRAs. The results of a recent multicenter, randomized, double‐blind, placebo‐controlled trial indicated that zolpidem extended‐release may be efficacious for up to 6 months in outpatients with chronic insomnia.28
The sole melatonin‐receptor agonist, ramelteon (Rozerem), also reduces time to fall asleep without next‐day psychomotor and memory effects.29 Ramelteon is believed to target receptors melatonin 1 and 2 receptors located in the brain's suprachiasmatic nucleus to stabilize circadian rhythms and stabilize the sleep‐wake cycle.30
CONCLUSION
Hospitalization is often associated with disrupted sleep, which can affect recovery from illness. Understanding the major factors that impair sleep during hospitalization allows clinicians to systemically evaluate and treat sleep problems. More than just prescribing a sedative/hypnotic, the treatment for sleep disruption includes addressing sleep hygiene and hospital environment issues, identifying medications that could disrupt sleep, and treating specific syndromes that impair sleep. We suggest a practical algorithm to guide clinical assessment, treatment options, and selection of appropriate sleeping medications. Critical to optimizing recovery from illness, sleep may be considered as the sixth vital sign, and should be part of the routine evaluation of every hospitalized patient.
- .Sleep maintenance insomnia: strengths and weaknesses of current pharmacologic therapies.Ann Clin Psychiatry.2006;18(1):49–56.
- ,.Treatment of insomnia.Prim Psychiatry.2005;12(8):47–56.
- ,,.Pharmacologic management of insomnia: past, present, and future.Psychiatr Clin North Am.2006;29:871–893.
- ,.“Hypnotic” prescription patterns in a large managed‐care population.Sleep Med.2004;5(5):463–466.
- ,,, et al.Subjective hypnotic efficacy of trazodone and zolpidem in DSM III‐R primary insomnia.Hum Psychopharmacol.1998;13:191–198.
- ,,, et al.Mirtazapine is more effective than trazodone: a double‐blind controlled study in hospitalized patients with major depression.Int Clin Psychopharmacol.1995;10:3–9.
- ,,.Mirtazapine: an antidepressant with noradrenergic and specific serotonergic effects.Pharmacotherapy.1997;17:10–21.
- ,,,.Efficacy of mirtazapine in obstructive sleep apnea syndrome.Sleep.2007;30(1):35–41.
- ,.Atypical antipsychotics in bipolar disorder: systematic review of randomised trials.BMC Psychiatry.2007;7:40:1–17.
- ,,, et al.Effectiveness of atypical antipsychotic drugs in patients with Alzheimer's Disease.N Engl J Med.2006;355:1525–1538.
- ,,.Risk of death with atypical antipsychotic drug treatment for dementia: meta‐analysis of randomized placebo‐controlled trials.JAMA.2005;294(15):1934–1943.
- ,.Clinical evaluation of diphenhydramine hydrochloride for the treatment of insomnia in psychiatric patients: a double‐blind study.J Clin Pharmacol.1983;23:234–242.
- .Diagnosis and treatment of chronic insomnia: a review.Psychiatr Serv.2005;56:332–343.
- ,,,,.Histamine H1 blocker hydroxyzine improves sleep in patients with cirrhosis and minimal hepatic encephalopathy: a randomized controlled pilot trial.Am J Gastroenterol.2007;102:744–753.
- ,.Clinical effects of chloral hydrate in hospitalized medical patients.J Clin Pharmacol.1979;19(10):669–674.
- Miller RD, editor.Miller's Anesthesia.6th ed.Philadelphia, PA:Elsevier;2005.
- .State‐of‐the‐art sleep management. Awakening insomnia management. Proceedings from a satellite symposium at SLEEP 2006: 20th Anniversary Meeting of the Associated Professional Sleep Societies, Salt Lake City, UT.2006:6–12.
- ,,.Use of a computer‐based reminder to improve sedative‐hypnotic prescribing in older hospitalized patients.J Am Geriatr Soc.2007;55:43–47.
- ,,,.Topiramate use in alprazolam addiction.World J Biol Psychiatry.2006;7(4):265–267.
- ,,,.Trends in recommendations for the pharmacotherapy of anxiety disorders by an international expert panel, 1992–1997.Eur Neuropsychopharmacol.1999;9(Suppl 6):S393–S398.
- ,,,,.Benzodiazepine use and the risk of motor vehicle crash in the elderly.JAMA.1997;278:27–31.
- ,,,,.Psychotropic drug use and the risk of hip fracture.NEngl J Med.1987;316:363–369.
- ,,,,.Sedative hypnotics in older people with insomnia: meta‐analysis of risks and benefits.BMJ.2005;331:1169–1175.
- ,,,,,.Delirium in hospitalized older persons: outcomes and predictors.J Am Geriatr Soc.1994;42:809–815.
- ,,,,.A 5‐year prospective assessment of the risk associated with individual benzodiazdepines and doses in new elderly users.J Am Geriatr Soc.2005;53:233–241.
- ,,, et al.Newer hypnotic drugs for the short‐term management of insomnia: a systematic review and economic evaluation.Health Technol Assess.2004;19:305–322.
- .Medications and their effect on sleep.Prim Care Clin Off Pract.2005;32:401–509.
- ,,,,.Long‐term efficacy and safety of zolpidem extended‐release 12.5 mg, administered 3 to 7 nights per week for 24 weeks, in patients with chronic primary insomnia: a 6‐month, randomized, double‐blind, placebo‐controlled, parallel‐group, multicenter study.Sleep.2008;31(1):79–90.
- ,,,.An efficacy, safety, and dose‐response study of Ramelteon in patients with chronic primary insomnia.Sleep Med.2006;7(1):17–24.
- ,.Melatonin, sleep, and circadian rhythms: rationale for development of specific melatonin agonists.Sleep Med.2004;5(6):523–532.
In Part 1, we reviewed normal sleep architecture, and discussed the numerous factors that often disrupt the sleep of hospitalized medical patients. Effective management of sleep complaints among acutely ill patients includes a thorough assessment of medical and psychiatric conditions, medications and other psychosocial factors that may be directly or indirectly impairing sleep. In Part 2, we review and introduce an algorithm for assessing and managing sleep complaints in acutely ill hospitalized patients.
ASSESSMENT AND EVALUATION OF SLEEP COMPLAINTS
Assessment and evaluation of a sleep complaint begins with (Figure 1) an initial review of the medical record for documentation of the signs and symptoms of an underlying primary sleep disorder, which may be exacerbated during an acute medical illness. Common sleep disorders that are often overlooked include obstructive sleep apnea (OSA), restless leg syndrome (RLS), and periodic limb movement disorder (PLMD). Predisposing factors, characteristic clinical features, and differential diagnoses of these disorders are described in Table 1.

| Sleep Disorder | Predisposing Factors | Clinical Features | Differential Diagnosis |
|---|---|---|---|
| |||
| Obstructive sleep apnea (OSA) | Nasopharyngeal abnormalities, craniofacial abnormalities, obesity, >40 years old, men > women (2:1), neurologic disorder (eg, recent stroke) | Repetitive episodes of upper airway obstruction that occur during sleep, usually associated with oxygen desaturation. Episodes include loud snoring or gasps lasting 2030 seconds. Associated with morning headaches and dry mouth. | Sleep‐related laryngospasm, nocturnal gastroesophageal reflux, narcolepsy, hypersomnia, PLMD, central alveolar hypoventilation, paroxysmal nocturnal dyspnea, primary snoring, Cheyne‐Stokes ventilation, nocturnal asthma |
| Periodic limb movement disorder (PLMD) | OSA. RLS, or narcolepsy; aging; chronic uremia; TCAs or MAOIs; withdrawal from antiepileptic agents, or other sedating agents | Periodic episodes of repetitive and stereotyped limb movements: extension of the big toe with partial flexion of the ankles, knees, or hips. Muscle contractions last 0.5 to 5 seconds, with 20‐second to 40‐second intervals between them. | Sleep starts (occur just prior to, not during, sleep, and do not have a regular periodicity like PLMD), nocturnal epileptic seizures, myoclonic epilepsy |
| Restless leg syndrome (RLS) | Pregnancy (>20 weeks gestation), uremia, anemia, rheumatoid arthritis, peak onset is middle age | Uncomfortable leg sensations that occur prior to sleep onset that leads to an irresistible urge to move the legs. Described as achy, crawling, pulling, prickling, or tingling, and disrupts sleep onset. | Chronic myelopathy, peripheral neuropathy, akathisia, fasciculation syndromes, anemia |
| Sleep starts | Can worsen with anxiety, caffeine or other stimulants, daytime physical exertion | Sudden, brief contraction of the legs that occurs at sleep onset. Usually benign, but may worsen during hospitalization, and interfere with sleep. | PLMD, RLS, hyperekplexia syndrome, in which generalized myoclonus is readily elicited by stimuli |
Obtain a focused history by using questions listed in Table 2 to characterize the onset, duration, frequency, and specific characteristics of the patient's current sleep patterns. Next, establish whether the onset of the patient's sleep complaint began with the time of hospitalization. Subsequent questions can then focus on factors that may be impairing sleep such as the hospital environment and sleep hygiene behaviors by comparing the patient's home sleep habits with those during hospitalization. Inquire about the use or abuse of substances such as sedatives, antidepressants, sedatives, antiepileptic drugs (AEDs), and opioids. Ask questions about the presence of pain syndromes and other comorbidities that often impact sleep.
| Focus | Examples of Questions |
|---|---|
| |
| Sleep pattern | Do you have problems falling asleep or staying asleep? How often do you wake up during the night? How long does it take you to fall back asleep? When did the problem start? What can we do to help you sleep? What time do you try to go to sleep, and what time do you wake up? |
| Behavioral factors | Compare your bedtime routine at home, and in the hospital. |
| Environment | Does the lighting or noise level in the hospital disrupt your sleep? How so? Are you awoken from sleep for laboratory work, monitoring, bathing, or other nursing/medical procedures? |
| Patient comfort | Is your pain adequately controlled at night? If not, are you on a scheduled analgesic regimen, or do you have to ask for pain medications? Do you have breathing problems, gastroesophageal reflux, or other type of discomfort that keeps you from sleeping well? |
| Substances | Do you drink alcohol? How much, and how often? When was your last alcoholic beverage? Inquire about cocaine, methamphetamine, marijuana, and medically‐unsupervised use of opioids. |
| Psychosocial | How was your mood just prior to being hospitalized? How has your mood been since you were admitted? Have you experienced any emotionally or physically traumatic event prior to, or during, this hospitalization that continues to bother you (eg, intubation, resuscitation, surgery, blood draws, MRI scanning)? |
MANAGEMENT OF SLEEP COMPLAINTS
Management of sleep disturbance is multifactorial and consists of nonpharmacologic as well as pharmacologic therapies. A stepwise approach is suggested and begins with nonpharmacologic strategies.
Nonpharmacologic Interventions
Before using sedative/hypnotic agents, address sleep hygiene and other factors that disrupt sleep during a hospitalization such as those listed in Table 3.
| Barriers to Sleep | Strategies To Optimize Sleep in the Hospital |
|---|---|
| |
| Noise | Limit the volume level of television sets, and do not allow patients or visitors to increase the volume. |
| Promptly respond to alarm monitors, and consider liberalizing the monitor alarm setting, if appropriate. | |
| Keep patients' doors closed, if possible. | |
| Post signs to remind staff and visitors to minimize conversations at or near the bedside. | |
| Adhere strictly to visiting hours. | |
| Encourage staff to switch their beepers and other electronic devices to vibrate at night. | |
| Limit the number of visitors at a time and/or if appropriate, have the patient meet with visitors in another location (eg, conference room, cafeteria). | |
| Offer earplugs. | |
| Ask patients to turn their phone ringers off when visiting hours are over. | |
| Anxiety | Encourage visitors to minimize discussing emotionally difficult topics with patients near bedtime. |
| Lighting | Offer eye masks. |
| Encourage exposure to brighter light during the day (turn on the lights, open the curtains), and turn off the lights by 9 PM. | |
| Poor sleep hygiene | Encourage regular nocturnal sleep time, and discourage lengthy naps during the day. |
| Medications and substances | Minimize BzRAs for sleep. Try to wean patients off BzRAs prior to discharge. At discharge, provide the minimum number of pills until they are scheduled to see their primary care clinician posthospitalization, and do not provide refills. |
|
Avoid starting multiple medications at one time. Minimize use of sleep‐disrupting medications (see Part 1, Table 3). |
|
| Change medication regimens to promote sleep; eg, avoid night‐time diuretics if possible. | |
| No caffeine or cigarette smoking after 6 PM. | |
| Effects of treatments | Minimize bathing, dressing changes, room switches, and other activities at night. |
| Regularly review nighttime orders to see if you could decrease the frequency of overnight monitoring (eg, fingersticks, labdraws, checking vitals). | |
| Delirium | Provide an updated calendar to facilitate cognitive orientation. |
| Discontinue nonessential medications. Minimize use of BzRAs, barbiturates, opiates, antihistamines, and anticholinergic agents. | |
| Regularly provide verbal and other cues to orient patients to the date, time, location, and circumstances. | |
| Nocturnal discomfort | Optimize nighttime glycemic control, and maximize pain management. |
| For patients with reflux: No oral intake after 8 PM, and keep head of bed elevated 30 degrees. | |
| Provide nocturnal O2, CPAP, and/or other medications, as appropriate. If patient is on CPAP, assess the mask's fit and comfort. | |
Pharmacologic (Sedative/Hypnotic) Interventions
Pharmacologic therapy may be necessary to treat disordered sleep. The ideal sleep aid would reduce sleep latency or time to fall asleep, increase total sleep time (TST), not cause next‐day sedation, improve daytime functioning, and minimize the development of tolerance. Unfortunately, no single agent meets all these independent criteria. In the past 10 years, newer benzodiazepines (BzRAs) with shorter half‐lives have been shown to be efficacious in reducing sleep latency, but the problem of sleep maintenance without next‐day sedation persists.1 To choose an appropriate sleep agent, evaluate the drug's efficacy, mechanism of action, and side‐effect profile. Then, match these characteristics with the patient's clinical condition(s). In patients with comorbid sleep and psychiatric problems, consider using a sedating psychotropic at bedtime to promote sleep.
Non‐Food and Drug AdministrationApproved (Off‐Label) Sleep Aids: Psychotropic Medications
Limited data exist on the efficacy of non‐Food and Drug Administration (FDA)approved medications for insomnia,2 such as antidepressants and atypical antipsychotics (AAPs), and antihistamines; examples of which are listed in Table 4. The administration of antihistamines, barbiturates, chloral hydrate, and alternative/herbal therapies has been discouraged, because the benefits rarely outweigh the risks associated with their use. Currently, trazodone is the most commonly prescribed antidepressant for the treatment of insomnia, despite the relative lack of data regarding its use for insomnia.3 Prescription data suggest that trazodoneat hypnotic doses, which are lower than the full antidepressant doseis more commonly prescribed for insomnia rather than for its FDA‐approved use for depression.4 In general, sleep specialists refrain from recommending sedating antidepressants for primary insomnia due to insufficient data regarding efficacy and safety. In addition, trazodone has been associated with arrhythmias in patients with preexisting cardiac conduction system disease. Curry et al.3 speculated that trazodone is popular among prescribers because, unlike most BzRAs, trazodone does not have a recommended limited duration of use and is perceived as being safer than BzRAs. Walsh et al.5 conducted a randomized double‐blind, placebo‐controlled trial (n = 589) that compared the hypnotic efficacy and other sleep‐associated variables of trazodone (50 mg) and zolpidem (10 mg). During the first week of treatment, the subjects on trazodone or zolpidem decreased their time to fall asleep, or sleep latency, by 22% and 35%, respectively, compared to placebo. Sleep latency was significantly shorter on zolpidem (57.75 2.7 minutes) than for trazodone (57.7 + 4.0 minutes). By the second week, subjects on zolpidem continued to have a reduction in the time to fall asleep, but there was no significant difference between subjects on trazodone and placebo.5 Trazodone may be an acceptable short‐term alternative to BzRAs for patients with hypercapnia or hypoxemia, and in those with a history of drug abuse or dependence. At doses of 150 to 450 mg, trazodone may be an appropriate medication in patients with major depressive disorder and problems with sleep maintenance.6 Tolerance to trazodone's sedating property tends to develop after 2 weeks of treatment, however, so other treatments may need to be considered if sleep problems persist. The available data address relatively short‐term use of trazodone, so questions of safety and efficacy for chronic insomnia remain unanswered.
| Drug | Pertinent Side Effects | Comments |
|---|---|---|
| ||
| Antidepressants | ||
| Mirtazapine (Remeron) | Somnolence, appetite, weight, dry mouth | May be beneficial for comorbid depression and insomnia. Lower doses (15 mg) increase sedation. |
| Trazodone | Residual daytime sedation, headache, orthostatic hypotension, priapism, cardiac arrhythmias | May be beneficial for comorbid depression and insomnia. Not recommended as first‐line agent for insomnia.3 May be an alternative if BzRAs are contraindicated (severe hypercapnia or hypoxemia or history of substance abuse). Tolerance usually develops within 2 weeks. Lower doses (50100 mg) than when used for depression (400 mg). |
| TCAs | Delirium, cognition, seizure threshold, orthostatic hypotension, tachycardia, acquired prolonged QT syndrome, heart block, acute hepatitis | Avoid in hospitalized patients due to their anticholinergic, antihistaminic, and cardiovascular side effects. May be beneficial for comorbid depression and insomnia. |
| Antihistamines | ||
| Diphenhydramine (Benadryl) | Residual daytime sedation, delirium, orthostatic hypotension, psychomotor function, prolonged QT syndrome, blurred vision, urinary retention | Better than placebo to treat insomnia,12 but data is lacking to definitively endorse diphenhydramine for insomnia.13 Tolerance to antihistamines develops within a few days. Avoid in patients >60 years old.18 |
| Hydroxyzine | Drowsiness, dry mouth, dizziness, agitation, cognitive function | Efficacy as anxiolytic for >4 months use not established. Not FDA‐approved for insomnia. Avoid in patients >60 years old, closed‐angle glaucoma, prostatic hypertrophy, severe asthma, and COPD. |
| Antipsychotics | ||
| Quetiapine (Seroquel) | Sedation, orthostatic hypotension, hyperglycemia, appetite, weight, hyperlipidemia | The most sedating of the atypical antipsychotics, it is frequently used as a sleep aid. Not recommended for insomnia or other sleep problems unless there is a comorbid psychiatric disorder. Dosed lower (25100 mg) when used for insomnia versus for FDA‐approved indications (600 mg). |
| Olanzapine (Zyprexa) | Sedation, hyperglycemia, appetite, weight, hyperlipidemia | Of atypical antipsychotics, olanzapine is the most likely to cause metabolic complications. Should not be used solely for insomnia. |
| Barbiturate | ||
| Chloral hydrate | Oversedation, respiratory depression, nausea, vomiting, diarrhea, drowsiness, cognitive function, psychotic symptoms (paranoia, hallucinations), vertigo, dizziness, headache | Chloral hydrate has been used for the short‐term (<2 weeks) treatment of insomnia, but is currently not FDA‐approved for that indication. Additive CNS depression may occur if given with other sedative‐hypnotics. Caution in patients with severe cardiac disease. Contraindicated in marked hepatic or renal impairment. Highly lethal in overdose, and should be avoided in patients with risk of suicide. |
Mirtazapine (Remeron), which promotes both sleep and appetite, may be particularly helpful for patients with cancer, acquired immunodeficiency syndrome (AIDS), and other conditions in which the triad of poor sleep, anorexia, and depression are common. Mirtazapine is a noradrenergic and specific serotonergic agent that causes inverse, dose‐dependent sedation (doses 15 mg are less sedating).7 To target sleeplessness, start with a dose between 7.5 and 15 mg. If ineffective at this dose, it is unlikely that increasing the dose will be of benefit for sleep. A small randomized, double‐blind, placebo‐controlled trial found that low‐dose mirtazapine reduced the apnea‐hypopnea index (API) by half in newly‐diagnosed subjects with OSA (n = 12).8 The results were promising in terms of the use of mixed‐profile serotonergic drugs in treating OSA. However, as pointed out by the researchers, mirtazapine's tendency to cause weight gain, is problematic in this patient population.
Although sedating, tricyclic antidepressants (TCAs) should not be used to promote sleep in hospitalized patients. TCAs increase the risk of cardiac conduction abnormalities, decrease seizure threshold, and have significant anticholinergic and anti‐alpha‐adrenergic effects. In dementia patients, the anticholinergic effect of TCAs may precipitate delirium.
AAPs should not be used routinely as first‐line agents for insomnia, except in patients who are in the midst of acute manic or psychotic episodes.9 With chronic use of AAPs, the risks of hyperglycemia, hyperlipidemia, and weight gain outweigh the potential sleep benefits of these agents. AAPs, especially risperidone, may cause extrapyramidal syndrome (EPS). Risperidone, ziprasidone and quetiapine have been associated with prolonged QTc interval, but the relatively low doses of AAPs that are used purely for sedative purposes makes this risk relatively low. If a patient has a history of Parkinsonism or other EPS, risperidone should generally be avoided. If a patient treated with risperidone develops EPS, another AAP should be considered. A reasonable precaution is to obtain a pretreatment 12‐lead electrocardiogram. If the QTc is greater than 450 msec, consider using olanzapine rather than ziprasidone, risperidone, or quetiapine. Sedating AAPs include risperidone (Risperdal), olanzapine (Zyprexa), and quetiapine (Seroquel), with the latter 2 being especially sedating. Quetiapine may also cause orthostatic hypotension. The recent practice of using AAPs for delirium has not been reported to be associated with significant safety risks, probably because delirium treatment is typically of short duration under a period of close clinical observation. These agents should not be used indefinitely for insomnia without close monitoring of metabolic, psychiatric, and neurologic status. However, recent data suggest that the risk of serious adverse effects of AAPs may outweigh the potential benefits for the treatment of aggression or agitation in patients with Alzheimer's disease.10
A meta‐analysis of randomized placebo‐controlled trials of AAP use among dementia patients showed that overall, the use of AAP drugs for periods of less than 8 to 12 weeks was associated with a small increased risk for death compared with placebo.11 Data indicated that most patients' behaviors improved substantially during the first 1 to 4 weeks of treatment. In a double‐blind, placebo‐controlled trial, 421 patients with Alzheimer's disease and psychosis, aggression or agitation were randomly assigned to receive olanzapine (mean dose, 5.5 mg per day), quetiapine (mean dose, 56.5 mg per day), risperidone (mean dose, 1.0 mg per day), or placebo. Improvement was observed in 32% of patients assigned to olanzapine, 26% of patients assigned to quetiapine, 29% of patients assigned to risperidone, and 21% of patients assigned to placebo. A lower, but significant, proportion of the patients (24%, 16%, 18%, and 5%, respectively) discontinued these medications due to intolerable side effects. Thus, if minimal improvement is observed even after 8 weeks of treatment, prescribers should consider discontinuing the AAP. The management of agitation in dementia, particularly in the elderly, calls for an integrative and creative psychopharmacological approach, including the use of antidepressants, nonbenzodiazepine anxiolytics such as buspirone, and mood stabilizers such as divalproex sodium (Depakote) before exposing patients to the risks of AAPs.
Antihistamines are the most commonly used over‐the‐counter agents for chronic insomnia.1 Diphenhydramine (Benadryl) has been shown to be better than placebo to treat insomnia,12 but data is lacking to definitively endorse its use to promote sleep.13 Diphenhydramine is also limited by the development of tolerance within a few days of daily use. The anticholinergic action of antihistamines may lead to orthostatic hypotension, urinary retention, and may induce delirium in vulnerable patients. Therefore, diphenhydramine should be avoided in hospitalized patients.
Recent data suggest that hydroxyzine, an antihistamine, may be an appropriate sleep aid for patients with hepatic encephalopathy in whom BzRAs are contraindicated.14 Subjective improvement in sleep was observed in 40% of hydroxyzine‐treated patients with hepatic encephalopathy compared to placebo.
Chloral hydrate is one of the Western world's oldest known sedative‐hypnotics and was commonly used as a sleep aid through the 1970s.15 Chloral hydrate was eventually supplanted by BzRAs,16 and fell out of favor as a sleep aid due to its relatively high tolerance rate, drug‐drug interaction profile, and the high risk of death in an overdose. Doses of 500 to 1000 mg sufficed to promote sleep in most of the hospitalized subjects. More recent data regarding its use for treating insomnia are not available, but chloral hydrate may be an alternative short‐term treatment for insomnia in selected hospitalized patients. Because of its high‐risk profile, chloral hydrate would be used as a last‐resort medication, preferably with input from critical care and/or sleep medicine specialists.
FDA‐Approved Sleep Aids
As shown in Table 5, the FDA has approved 3 classes of medications for the treatment of insomnia: benzodiazepine gamma‐aminobutyric acid (GABA)A receptor agonists (BzRAs), nonbenzodiazepine GABAA receptor agonists (non‐BzRAs), and melatonin‐receptor agonists.17 BzRAs include estazolam (ProSom), flurazepam (Dalmane), quazepam (Doral), temazepam (Restoril), and triazolam (Halcion). Though BzRAs decrease sleep latency, increase TST, and decrease slow wave or deep sleep, they also have adverse side effects such as daytime sedation, anterograde amnesia, cognitive impairment, motor incoordination, dependence, tolerance, and rebound insomnia.18 Because of these side effects, BzRAs should be limited to generally healthy, young (ie, <45 years old) patients who are expected to have brief hospital stays.
| Drugs | Adult Dose (mg) | Half‐Life (hours)* | Onset (minutes) | Peak Effect (hours) | Major Effects/Clinical Comments |
|---|---|---|---|---|---|
| |||||
| BzRAs | Caution in elderly patients. Tolerance to BzRAs develop to the sedative, hypnotic, and anticonvulsant effects. | ||||
| Estazolam (ProSom) | 12 | 1024 | 60 | 0.51.5 | Short‐term (710 days) treatment for frequent arousals, early morning awakening. Not as useful for sleep onset. Avoid in patients with OSA. Caution in elderly patients, liver disease. High doses can cause respiratory depression. |
| Flurazepam (Dalmane) | 1530 | 47100 | 1520 | 36 | In general, avoid in hospitalized medical patients, especially elderly patients. |
| Quazepam (Doral) | 7.515 | 25114 | 1.5 | In general, avoid in hospitalized medical patients, especially elderly patients. | |
| Temazepam (Restoril) | 1530 | 616 | 23 | Short‐term (710 days) treatment for sleep onset and maintenance. Doses 30 mg/day: morning grogginess, nausea, headache, and vivid dreaming. | |
| Triazolam (Halcion) | 0.1250.25 | 1.55.5 | 1530 | 1.75 | Maximum dose is 0.5 mg. Short‐term (710 days) treatment. Rapid onset; should be in bed when taking medication. Contraindicated with atazanavir, ketoconazole, itraconazole, nefazodone, ritonavir. |
| Non‐BzRAs | |||||
| Eszopiclone (Lunesta) | 23 | 69 | 1 | In elderly: difficulty falling asleep, then initial: 1 mg; maximum 2 mg. Difficulty staying asleep: 2 mg. Rapid onset; should be in bed when taking medication. For faster sleep onset, do not ingest with high‐fat foods. No tolerance after 6 months. | |
| Zaleplon (Sonata) | 520 | 1 | Rapid | 1 | Short‐term (710 days) treatment for falling asleep and/or next‐day wakefulness is crucial (eg, shift workers). |
| Zopiclone (Imovane) | 515 | 3.86.5 (510 in elderly) | 30 | <2 | Transient and short‐term (710 days) treatment. Contraindicated in severe respiratory impairment. Caution in liver disease and depression; elderly prone to side effects. Anticholinergic agents may plasma level. |
| Zolpidem (Ambien) | 520 | 1.44.5 | 30 | 2 | Short‐term (710 days) treatment for sleep onset and maintenance. Rapid onset; should be in bed when taking medication. For faster sleep onset, do not ingest with food. No tolerance after 50 weeks. |
| Melatonin agonist | |||||
| Ramelton (Rozerem) | 8 | 12 | 30 | 11.5 | For sleep onset. For faster sleep onset, do not ingest with high‐fat foods. No tolerance. Contraindicated with fluvoxamine. |
Efficacy and safety studies have generally been limited to healthy, younger individuals without a history of primary sleep disorder. Potential adverse effects of BzRAs may become even more pronounced in hospitalized medical patients due to older age, acute illness, cointeraction drugs, and multidrug regimens. Although BzRAs are FDA‐approved for the treatment of insomnia, flurazepam and quazepam should generally be avoided in hospitalized patients. These agents' long half‐lives increase the risk of drug‐drug interactions and adverse events such as respiratory depression, cognitive decline, and delirium in acutely ill patients. For similar reasons, other long‐acting BzRAs such as clonazepam (Klonopin) and diazepam (Valium) should also not be used to treat insomnia in hospitalized patients. An exception to this is a patient with RLS, in which clonazepam is an approved treatment. However, now that ropinirole HCl (Requip) is FDA‐approved for RLS, BzARs may be able to be avoided. Lorazepam (Ativan), due to its relatively short half‐life and its anxiolytic property, is frequently used to treat insomnia in hospitalized medical patients.18 Start with the lowest dose possible (eg, 0.5 mg) as a one‐time‐only order, or on a as needed basis for 3 days. Alprazolam (Xanax), a potent, fast‐acting BzRA with a relatively short half‐life, has developed a reputation as being notoriously addictive, and experts feel alprazolam has similar potential for withdrawal and rebound.19, 20
The use of BzRAs should be minimized in all patients, and avoided in the elderly or those with a particularly high risk for delirium (eg, traumatic brain injury, stroke, multiple new medications). All BzRAs should be avoided in patients with a prior history of sedative‐hypnotic and/or alcohol dependence unless medically indicated, such as in alcohol withdrawal. Refrain from ordering nightly scheduled BzRAs without a specific time limit to ensure that sedative‐hypnotic use is closely monitored.
For the past 2 decades, physicians have been advised against using long‐acting BzRAs in the elderly (>65 years old) due to the increased risks of hip fractures, falls, motor vehicle accidents, daytime sedation, and adverse cognitive events such as delirium.2124 A large 5‐year prospective study in Quebec found that the risk of injury varied by the BzRA, and was independent of half‐life.25 Importantly, the risk of injury was dose‐dependent: the higher the dose of oxazepam, flurazepam, or chlordiazepoxide, the higher the risk of injury in the elderly.
Non‐BzRAs seem to have a superior side‐effect profile when compared to BzRAs, but should also be used with caution in the elderly. Non‐BzRAs include eszopiclone (Lunesta), zaleplon (Sonata), zolpidem (Ambien), and zolpidem extended‐release. The number of comparison studies is limited, but the available data reveal that: (1) zolpidem (Ambien) may be better than temazepam (Restoril) in terms of sleep latency and quality; and (2) zaleplon (Sonata) may lead to a shorter sleep latency than zolpidem (Ambien), but the latter is associated with longer sleep duration.26 Non‐BzRAs have less next‐day sedation, psychomotor dysfunction, tolerance/withdrawal, and rapid‐eye‐movement (REM) sleep rebound; and lower abuse potential than BzRAs.27
The most commonly prescribed hypnotic, zolpidem has a short half‐life, and seems to reduce sleep latency with minimal residual side effects when compared to BzRAs. The results of a recent multicenter, randomized, double‐blind, placebo‐controlled trial indicated that zolpidem extended‐release may be efficacious for up to 6 months in outpatients with chronic insomnia.28
The sole melatonin‐receptor agonist, ramelteon (Rozerem), also reduces time to fall asleep without next‐day psychomotor and memory effects.29 Ramelteon is believed to target receptors melatonin 1 and 2 receptors located in the brain's suprachiasmatic nucleus to stabilize circadian rhythms and stabilize the sleep‐wake cycle.30
CONCLUSION
Hospitalization is often associated with disrupted sleep, which can affect recovery from illness. Understanding the major factors that impair sleep during hospitalization allows clinicians to systemically evaluate and treat sleep problems. More than just prescribing a sedative/hypnotic, the treatment for sleep disruption includes addressing sleep hygiene and hospital environment issues, identifying medications that could disrupt sleep, and treating specific syndromes that impair sleep. We suggest a practical algorithm to guide clinical assessment, treatment options, and selection of appropriate sleeping medications. Critical to optimizing recovery from illness, sleep may be considered as the sixth vital sign, and should be part of the routine evaluation of every hospitalized patient.
In Part 1, we reviewed normal sleep architecture, and discussed the numerous factors that often disrupt the sleep of hospitalized medical patients. Effective management of sleep complaints among acutely ill patients includes a thorough assessment of medical and psychiatric conditions, medications and other psychosocial factors that may be directly or indirectly impairing sleep. In Part 2, we review and introduce an algorithm for assessing and managing sleep complaints in acutely ill hospitalized patients.
ASSESSMENT AND EVALUATION OF SLEEP COMPLAINTS
Assessment and evaluation of a sleep complaint begins with (Figure 1) an initial review of the medical record for documentation of the signs and symptoms of an underlying primary sleep disorder, which may be exacerbated during an acute medical illness. Common sleep disorders that are often overlooked include obstructive sleep apnea (OSA), restless leg syndrome (RLS), and periodic limb movement disorder (PLMD). Predisposing factors, characteristic clinical features, and differential diagnoses of these disorders are described in Table 1.

| Sleep Disorder | Predisposing Factors | Clinical Features | Differential Diagnosis |
|---|---|---|---|
| |||
| Obstructive sleep apnea (OSA) | Nasopharyngeal abnormalities, craniofacial abnormalities, obesity, >40 years old, men > women (2:1), neurologic disorder (eg, recent stroke) | Repetitive episodes of upper airway obstruction that occur during sleep, usually associated with oxygen desaturation. Episodes include loud snoring or gasps lasting 2030 seconds. Associated with morning headaches and dry mouth. | Sleep‐related laryngospasm, nocturnal gastroesophageal reflux, narcolepsy, hypersomnia, PLMD, central alveolar hypoventilation, paroxysmal nocturnal dyspnea, primary snoring, Cheyne‐Stokes ventilation, nocturnal asthma |
| Periodic limb movement disorder (PLMD) | OSA. RLS, or narcolepsy; aging; chronic uremia; TCAs or MAOIs; withdrawal from antiepileptic agents, or other sedating agents | Periodic episodes of repetitive and stereotyped limb movements: extension of the big toe with partial flexion of the ankles, knees, or hips. Muscle contractions last 0.5 to 5 seconds, with 20‐second to 40‐second intervals between them. | Sleep starts (occur just prior to, not during, sleep, and do not have a regular periodicity like PLMD), nocturnal epileptic seizures, myoclonic epilepsy |
| Restless leg syndrome (RLS) | Pregnancy (>20 weeks gestation), uremia, anemia, rheumatoid arthritis, peak onset is middle age | Uncomfortable leg sensations that occur prior to sleep onset that leads to an irresistible urge to move the legs. Described as achy, crawling, pulling, prickling, or tingling, and disrupts sleep onset. | Chronic myelopathy, peripheral neuropathy, akathisia, fasciculation syndromes, anemia |
| Sleep starts | Can worsen with anxiety, caffeine or other stimulants, daytime physical exertion | Sudden, brief contraction of the legs that occurs at sleep onset. Usually benign, but may worsen during hospitalization, and interfere with sleep. | PLMD, RLS, hyperekplexia syndrome, in which generalized myoclonus is readily elicited by stimuli |
Obtain a focused history by using questions listed in Table 2 to characterize the onset, duration, frequency, and specific characteristics of the patient's current sleep patterns. Next, establish whether the onset of the patient's sleep complaint began with the time of hospitalization. Subsequent questions can then focus on factors that may be impairing sleep such as the hospital environment and sleep hygiene behaviors by comparing the patient's home sleep habits with those during hospitalization. Inquire about the use or abuse of substances such as sedatives, antidepressants, sedatives, antiepileptic drugs (AEDs), and opioids. Ask questions about the presence of pain syndromes and other comorbidities that often impact sleep.
| Focus | Examples of Questions |
|---|---|
| |
| Sleep pattern | Do you have problems falling asleep or staying asleep? How often do you wake up during the night? How long does it take you to fall back asleep? When did the problem start? What can we do to help you sleep? What time do you try to go to sleep, and what time do you wake up? |
| Behavioral factors | Compare your bedtime routine at home, and in the hospital. |
| Environment | Does the lighting or noise level in the hospital disrupt your sleep? How so? Are you awoken from sleep for laboratory work, monitoring, bathing, or other nursing/medical procedures? |
| Patient comfort | Is your pain adequately controlled at night? If not, are you on a scheduled analgesic regimen, or do you have to ask for pain medications? Do you have breathing problems, gastroesophageal reflux, or other type of discomfort that keeps you from sleeping well? |
| Substances | Do you drink alcohol? How much, and how often? When was your last alcoholic beverage? Inquire about cocaine, methamphetamine, marijuana, and medically‐unsupervised use of opioids. |
| Psychosocial | How was your mood just prior to being hospitalized? How has your mood been since you were admitted? Have you experienced any emotionally or physically traumatic event prior to, or during, this hospitalization that continues to bother you (eg, intubation, resuscitation, surgery, blood draws, MRI scanning)? |
MANAGEMENT OF SLEEP COMPLAINTS
Management of sleep disturbance is multifactorial and consists of nonpharmacologic as well as pharmacologic therapies. A stepwise approach is suggested and begins with nonpharmacologic strategies.
Nonpharmacologic Interventions
Before using sedative/hypnotic agents, address sleep hygiene and other factors that disrupt sleep during a hospitalization such as those listed in Table 3.
| Barriers to Sleep | Strategies To Optimize Sleep in the Hospital |
|---|---|
| |
| Noise | Limit the volume level of television sets, and do not allow patients or visitors to increase the volume. |
| Promptly respond to alarm monitors, and consider liberalizing the monitor alarm setting, if appropriate. | |
| Keep patients' doors closed, if possible. | |
| Post signs to remind staff and visitors to minimize conversations at or near the bedside. | |
| Adhere strictly to visiting hours. | |
| Encourage staff to switch their beepers and other electronic devices to vibrate at night. | |
| Limit the number of visitors at a time and/or if appropriate, have the patient meet with visitors in another location (eg, conference room, cafeteria). | |
| Offer earplugs. | |
| Ask patients to turn their phone ringers off when visiting hours are over. | |
| Anxiety | Encourage visitors to minimize discussing emotionally difficult topics with patients near bedtime. |
| Lighting | Offer eye masks. |
| Encourage exposure to brighter light during the day (turn on the lights, open the curtains), and turn off the lights by 9 PM. | |
| Poor sleep hygiene | Encourage regular nocturnal sleep time, and discourage lengthy naps during the day. |
| Medications and substances | Minimize BzRAs for sleep. Try to wean patients off BzRAs prior to discharge. At discharge, provide the minimum number of pills until they are scheduled to see their primary care clinician posthospitalization, and do not provide refills. |
|
Avoid starting multiple medications at one time. Minimize use of sleep‐disrupting medications (see Part 1, Table 3). |
|
| Change medication regimens to promote sleep; eg, avoid night‐time diuretics if possible. | |
| No caffeine or cigarette smoking after 6 PM. | |
| Effects of treatments | Minimize bathing, dressing changes, room switches, and other activities at night. |
| Regularly review nighttime orders to see if you could decrease the frequency of overnight monitoring (eg, fingersticks, labdraws, checking vitals). | |
| Delirium | Provide an updated calendar to facilitate cognitive orientation. |
| Discontinue nonessential medications. Minimize use of BzRAs, barbiturates, opiates, antihistamines, and anticholinergic agents. | |
| Regularly provide verbal and other cues to orient patients to the date, time, location, and circumstances. | |
| Nocturnal discomfort | Optimize nighttime glycemic control, and maximize pain management. |
| For patients with reflux: No oral intake after 8 PM, and keep head of bed elevated 30 degrees. | |
| Provide nocturnal O2, CPAP, and/or other medications, as appropriate. If patient is on CPAP, assess the mask's fit and comfort. | |
Pharmacologic (Sedative/Hypnotic) Interventions
Pharmacologic therapy may be necessary to treat disordered sleep. The ideal sleep aid would reduce sleep latency or time to fall asleep, increase total sleep time (TST), not cause next‐day sedation, improve daytime functioning, and minimize the development of tolerance. Unfortunately, no single agent meets all these independent criteria. In the past 10 years, newer benzodiazepines (BzRAs) with shorter half‐lives have been shown to be efficacious in reducing sleep latency, but the problem of sleep maintenance without next‐day sedation persists.1 To choose an appropriate sleep agent, evaluate the drug's efficacy, mechanism of action, and side‐effect profile. Then, match these characteristics with the patient's clinical condition(s). In patients with comorbid sleep and psychiatric problems, consider using a sedating psychotropic at bedtime to promote sleep.
Non‐Food and Drug AdministrationApproved (Off‐Label) Sleep Aids: Psychotropic Medications
Limited data exist on the efficacy of non‐Food and Drug Administration (FDA)approved medications for insomnia,2 such as antidepressants and atypical antipsychotics (AAPs), and antihistamines; examples of which are listed in Table 4. The administration of antihistamines, barbiturates, chloral hydrate, and alternative/herbal therapies has been discouraged, because the benefits rarely outweigh the risks associated with their use. Currently, trazodone is the most commonly prescribed antidepressant for the treatment of insomnia, despite the relative lack of data regarding its use for insomnia.3 Prescription data suggest that trazodoneat hypnotic doses, which are lower than the full antidepressant doseis more commonly prescribed for insomnia rather than for its FDA‐approved use for depression.4 In general, sleep specialists refrain from recommending sedating antidepressants for primary insomnia due to insufficient data regarding efficacy and safety. In addition, trazodone has been associated with arrhythmias in patients with preexisting cardiac conduction system disease. Curry et al.3 speculated that trazodone is popular among prescribers because, unlike most BzRAs, trazodone does not have a recommended limited duration of use and is perceived as being safer than BzRAs. Walsh et al.5 conducted a randomized double‐blind, placebo‐controlled trial (n = 589) that compared the hypnotic efficacy and other sleep‐associated variables of trazodone (50 mg) and zolpidem (10 mg). During the first week of treatment, the subjects on trazodone or zolpidem decreased their time to fall asleep, or sleep latency, by 22% and 35%, respectively, compared to placebo. Sleep latency was significantly shorter on zolpidem (57.75 2.7 minutes) than for trazodone (57.7 + 4.0 minutes). By the second week, subjects on zolpidem continued to have a reduction in the time to fall asleep, but there was no significant difference between subjects on trazodone and placebo.5 Trazodone may be an acceptable short‐term alternative to BzRAs for patients with hypercapnia or hypoxemia, and in those with a history of drug abuse or dependence. At doses of 150 to 450 mg, trazodone may be an appropriate medication in patients with major depressive disorder and problems with sleep maintenance.6 Tolerance to trazodone's sedating property tends to develop after 2 weeks of treatment, however, so other treatments may need to be considered if sleep problems persist. The available data address relatively short‐term use of trazodone, so questions of safety and efficacy for chronic insomnia remain unanswered.
| Drug | Pertinent Side Effects | Comments |
|---|---|---|
| ||
| Antidepressants | ||
| Mirtazapine (Remeron) | Somnolence, appetite, weight, dry mouth | May be beneficial for comorbid depression and insomnia. Lower doses (15 mg) increase sedation. |
| Trazodone | Residual daytime sedation, headache, orthostatic hypotension, priapism, cardiac arrhythmias | May be beneficial for comorbid depression and insomnia. Not recommended as first‐line agent for insomnia.3 May be an alternative if BzRAs are contraindicated (severe hypercapnia or hypoxemia or history of substance abuse). Tolerance usually develops within 2 weeks. Lower doses (50100 mg) than when used for depression (400 mg). |
| TCAs | Delirium, cognition, seizure threshold, orthostatic hypotension, tachycardia, acquired prolonged QT syndrome, heart block, acute hepatitis | Avoid in hospitalized patients due to their anticholinergic, antihistaminic, and cardiovascular side effects. May be beneficial for comorbid depression and insomnia. |
| Antihistamines | ||
| Diphenhydramine (Benadryl) | Residual daytime sedation, delirium, orthostatic hypotension, psychomotor function, prolonged QT syndrome, blurred vision, urinary retention | Better than placebo to treat insomnia,12 but data is lacking to definitively endorse diphenhydramine for insomnia.13 Tolerance to antihistamines develops within a few days. Avoid in patients >60 years old.18 |
| Hydroxyzine | Drowsiness, dry mouth, dizziness, agitation, cognitive function | Efficacy as anxiolytic for >4 months use not established. Not FDA‐approved for insomnia. Avoid in patients >60 years old, closed‐angle glaucoma, prostatic hypertrophy, severe asthma, and COPD. |
| Antipsychotics | ||
| Quetiapine (Seroquel) | Sedation, orthostatic hypotension, hyperglycemia, appetite, weight, hyperlipidemia | The most sedating of the atypical antipsychotics, it is frequently used as a sleep aid. Not recommended for insomnia or other sleep problems unless there is a comorbid psychiatric disorder. Dosed lower (25100 mg) when used for insomnia versus for FDA‐approved indications (600 mg). |
| Olanzapine (Zyprexa) | Sedation, hyperglycemia, appetite, weight, hyperlipidemia | Of atypical antipsychotics, olanzapine is the most likely to cause metabolic complications. Should not be used solely for insomnia. |
| Barbiturate | ||
| Chloral hydrate | Oversedation, respiratory depression, nausea, vomiting, diarrhea, drowsiness, cognitive function, psychotic symptoms (paranoia, hallucinations), vertigo, dizziness, headache | Chloral hydrate has been used for the short‐term (<2 weeks) treatment of insomnia, but is currently not FDA‐approved for that indication. Additive CNS depression may occur if given with other sedative‐hypnotics. Caution in patients with severe cardiac disease. Contraindicated in marked hepatic or renal impairment. Highly lethal in overdose, and should be avoided in patients with risk of suicide. |
Mirtazapine (Remeron), which promotes both sleep and appetite, may be particularly helpful for patients with cancer, acquired immunodeficiency syndrome (AIDS), and other conditions in which the triad of poor sleep, anorexia, and depression are common. Mirtazapine is a noradrenergic and specific serotonergic agent that causes inverse, dose‐dependent sedation (doses 15 mg are less sedating).7 To target sleeplessness, start with a dose between 7.5 and 15 mg. If ineffective at this dose, it is unlikely that increasing the dose will be of benefit for sleep. A small randomized, double‐blind, placebo‐controlled trial found that low‐dose mirtazapine reduced the apnea‐hypopnea index (API) by half in newly‐diagnosed subjects with OSA (n = 12).8 The results were promising in terms of the use of mixed‐profile serotonergic drugs in treating OSA. However, as pointed out by the researchers, mirtazapine's tendency to cause weight gain, is problematic in this patient population.
Although sedating, tricyclic antidepressants (TCAs) should not be used to promote sleep in hospitalized patients. TCAs increase the risk of cardiac conduction abnormalities, decrease seizure threshold, and have significant anticholinergic and anti‐alpha‐adrenergic effects. In dementia patients, the anticholinergic effect of TCAs may precipitate delirium.
AAPs should not be used routinely as first‐line agents for insomnia, except in patients who are in the midst of acute manic or psychotic episodes.9 With chronic use of AAPs, the risks of hyperglycemia, hyperlipidemia, and weight gain outweigh the potential sleep benefits of these agents. AAPs, especially risperidone, may cause extrapyramidal syndrome (EPS). Risperidone, ziprasidone and quetiapine have been associated with prolonged QTc interval, but the relatively low doses of AAPs that are used purely for sedative purposes makes this risk relatively low. If a patient has a history of Parkinsonism or other EPS, risperidone should generally be avoided. If a patient treated with risperidone develops EPS, another AAP should be considered. A reasonable precaution is to obtain a pretreatment 12‐lead electrocardiogram. If the QTc is greater than 450 msec, consider using olanzapine rather than ziprasidone, risperidone, or quetiapine. Sedating AAPs include risperidone (Risperdal), olanzapine (Zyprexa), and quetiapine (Seroquel), with the latter 2 being especially sedating. Quetiapine may also cause orthostatic hypotension. The recent practice of using AAPs for delirium has not been reported to be associated with significant safety risks, probably because delirium treatment is typically of short duration under a period of close clinical observation. These agents should not be used indefinitely for insomnia without close monitoring of metabolic, psychiatric, and neurologic status. However, recent data suggest that the risk of serious adverse effects of AAPs may outweigh the potential benefits for the treatment of aggression or agitation in patients with Alzheimer's disease.10
A meta‐analysis of randomized placebo‐controlled trials of AAP use among dementia patients showed that overall, the use of AAP drugs for periods of less than 8 to 12 weeks was associated with a small increased risk for death compared with placebo.11 Data indicated that most patients' behaviors improved substantially during the first 1 to 4 weeks of treatment. In a double‐blind, placebo‐controlled trial, 421 patients with Alzheimer's disease and psychosis, aggression or agitation were randomly assigned to receive olanzapine (mean dose, 5.5 mg per day), quetiapine (mean dose, 56.5 mg per day), risperidone (mean dose, 1.0 mg per day), or placebo. Improvement was observed in 32% of patients assigned to olanzapine, 26% of patients assigned to quetiapine, 29% of patients assigned to risperidone, and 21% of patients assigned to placebo. A lower, but significant, proportion of the patients (24%, 16%, 18%, and 5%, respectively) discontinued these medications due to intolerable side effects. Thus, if minimal improvement is observed even after 8 weeks of treatment, prescribers should consider discontinuing the AAP. The management of agitation in dementia, particularly in the elderly, calls for an integrative and creative psychopharmacological approach, including the use of antidepressants, nonbenzodiazepine anxiolytics such as buspirone, and mood stabilizers such as divalproex sodium (Depakote) before exposing patients to the risks of AAPs.
Antihistamines are the most commonly used over‐the‐counter agents for chronic insomnia.1 Diphenhydramine (Benadryl) has been shown to be better than placebo to treat insomnia,12 but data is lacking to definitively endorse its use to promote sleep.13 Diphenhydramine is also limited by the development of tolerance within a few days of daily use. The anticholinergic action of antihistamines may lead to orthostatic hypotension, urinary retention, and may induce delirium in vulnerable patients. Therefore, diphenhydramine should be avoided in hospitalized patients.
Recent data suggest that hydroxyzine, an antihistamine, may be an appropriate sleep aid for patients with hepatic encephalopathy in whom BzRAs are contraindicated.14 Subjective improvement in sleep was observed in 40% of hydroxyzine‐treated patients with hepatic encephalopathy compared to placebo.
Chloral hydrate is one of the Western world's oldest known sedative‐hypnotics and was commonly used as a sleep aid through the 1970s.15 Chloral hydrate was eventually supplanted by BzRAs,16 and fell out of favor as a sleep aid due to its relatively high tolerance rate, drug‐drug interaction profile, and the high risk of death in an overdose. Doses of 500 to 1000 mg sufficed to promote sleep in most of the hospitalized subjects. More recent data regarding its use for treating insomnia are not available, but chloral hydrate may be an alternative short‐term treatment for insomnia in selected hospitalized patients. Because of its high‐risk profile, chloral hydrate would be used as a last‐resort medication, preferably with input from critical care and/or sleep medicine specialists.
FDA‐Approved Sleep Aids
As shown in Table 5, the FDA has approved 3 classes of medications for the treatment of insomnia: benzodiazepine gamma‐aminobutyric acid (GABA)A receptor agonists (BzRAs), nonbenzodiazepine GABAA receptor agonists (non‐BzRAs), and melatonin‐receptor agonists.17 BzRAs include estazolam (ProSom), flurazepam (Dalmane), quazepam (Doral), temazepam (Restoril), and triazolam (Halcion). Though BzRAs decrease sleep latency, increase TST, and decrease slow wave or deep sleep, they also have adverse side effects such as daytime sedation, anterograde amnesia, cognitive impairment, motor incoordination, dependence, tolerance, and rebound insomnia.18 Because of these side effects, BzRAs should be limited to generally healthy, young (ie, <45 years old) patients who are expected to have brief hospital stays.
| Drugs | Adult Dose (mg) | Half‐Life (hours)* | Onset (minutes) | Peak Effect (hours) | Major Effects/Clinical Comments |
|---|---|---|---|---|---|
| |||||
| BzRAs | Caution in elderly patients. Tolerance to BzRAs develop to the sedative, hypnotic, and anticonvulsant effects. | ||||
| Estazolam (ProSom) | 12 | 1024 | 60 | 0.51.5 | Short‐term (710 days) treatment for frequent arousals, early morning awakening. Not as useful for sleep onset. Avoid in patients with OSA. Caution in elderly patients, liver disease. High doses can cause respiratory depression. |
| Flurazepam (Dalmane) | 1530 | 47100 | 1520 | 36 | In general, avoid in hospitalized medical patients, especially elderly patients. |
| Quazepam (Doral) | 7.515 | 25114 | 1.5 | In general, avoid in hospitalized medical patients, especially elderly patients. | |
| Temazepam (Restoril) | 1530 | 616 | 23 | Short‐term (710 days) treatment for sleep onset and maintenance. Doses 30 mg/day: morning grogginess, nausea, headache, and vivid dreaming. | |
| Triazolam (Halcion) | 0.1250.25 | 1.55.5 | 1530 | 1.75 | Maximum dose is 0.5 mg. Short‐term (710 days) treatment. Rapid onset; should be in bed when taking medication. Contraindicated with atazanavir, ketoconazole, itraconazole, nefazodone, ritonavir. |
| Non‐BzRAs | |||||
| Eszopiclone (Lunesta) | 23 | 69 | 1 | In elderly: difficulty falling asleep, then initial: 1 mg; maximum 2 mg. Difficulty staying asleep: 2 mg. Rapid onset; should be in bed when taking medication. For faster sleep onset, do not ingest with high‐fat foods. No tolerance after 6 months. | |
| Zaleplon (Sonata) | 520 | 1 | Rapid | 1 | Short‐term (710 days) treatment for falling asleep and/or next‐day wakefulness is crucial (eg, shift workers). |
| Zopiclone (Imovane) | 515 | 3.86.5 (510 in elderly) | 30 | <2 | Transient and short‐term (710 days) treatment. Contraindicated in severe respiratory impairment. Caution in liver disease and depression; elderly prone to side effects. Anticholinergic agents may plasma level. |
| Zolpidem (Ambien) | 520 | 1.44.5 | 30 | 2 | Short‐term (710 days) treatment for sleep onset and maintenance. Rapid onset; should be in bed when taking medication. For faster sleep onset, do not ingest with food. No tolerance after 50 weeks. |
| Melatonin agonist | |||||
| Ramelton (Rozerem) | 8 | 12 | 30 | 11.5 | For sleep onset. For faster sleep onset, do not ingest with high‐fat foods. No tolerance. Contraindicated with fluvoxamine. |
Efficacy and safety studies have generally been limited to healthy, younger individuals without a history of primary sleep disorder. Potential adverse effects of BzRAs may become even more pronounced in hospitalized medical patients due to older age, acute illness, cointeraction drugs, and multidrug regimens. Although BzRAs are FDA‐approved for the treatment of insomnia, flurazepam and quazepam should generally be avoided in hospitalized patients. These agents' long half‐lives increase the risk of drug‐drug interactions and adverse events such as respiratory depression, cognitive decline, and delirium in acutely ill patients. For similar reasons, other long‐acting BzRAs such as clonazepam (Klonopin) and diazepam (Valium) should also not be used to treat insomnia in hospitalized patients. An exception to this is a patient with RLS, in which clonazepam is an approved treatment. However, now that ropinirole HCl (Requip) is FDA‐approved for RLS, BzARs may be able to be avoided. Lorazepam (Ativan), due to its relatively short half‐life and its anxiolytic property, is frequently used to treat insomnia in hospitalized medical patients.18 Start with the lowest dose possible (eg, 0.5 mg) as a one‐time‐only order, or on a as needed basis for 3 days. Alprazolam (Xanax), a potent, fast‐acting BzRA with a relatively short half‐life, has developed a reputation as being notoriously addictive, and experts feel alprazolam has similar potential for withdrawal and rebound.19, 20
The use of BzRAs should be minimized in all patients, and avoided in the elderly or those with a particularly high risk for delirium (eg, traumatic brain injury, stroke, multiple new medications). All BzRAs should be avoided in patients with a prior history of sedative‐hypnotic and/or alcohol dependence unless medically indicated, such as in alcohol withdrawal. Refrain from ordering nightly scheduled BzRAs without a specific time limit to ensure that sedative‐hypnotic use is closely monitored.
For the past 2 decades, physicians have been advised against using long‐acting BzRAs in the elderly (>65 years old) due to the increased risks of hip fractures, falls, motor vehicle accidents, daytime sedation, and adverse cognitive events such as delirium.2124 A large 5‐year prospective study in Quebec found that the risk of injury varied by the BzRA, and was independent of half‐life.25 Importantly, the risk of injury was dose‐dependent: the higher the dose of oxazepam, flurazepam, or chlordiazepoxide, the higher the risk of injury in the elderly.
Non‐BzRAs seem to have a superior side‐effect profile when compared to BzRAs, but should also be used with caution in the elderly. Non‐BzRAs include eszopiclone (Lunesta), zaleplon (Sonata), zolpidem (Ambien), and zolpidem extended‐release. The number of comparison studies is limited, but the available data reveal that: (1) zolpidem (Ambien) may be better than temazepam (Restoril) in terms of sleep latency and quality; and (2) zaleplon (Sonata) may lead to a shorter sleep latency than zolpidem (Ambien), but the latter is associated with longer sleep duration.26 Non‐BzRAs have less next‐day sedation, psychomotor dysfunction, tolerance/withdrawal, and rapid‐eye‐movement (REM) sleep rebound; and lower abuse potential than BzRAs.27
The most commonly prescribed hypnotic, zolpidem has a short half‐life, and seems to reduce sleep latency with minimal residual side effects when compared to BzRAs. The results of a recent multicenter, randomized, double‐blind, placebo‐controlled trial indicated that zolpidem extended‐release may be efficacious for up to 6 months in outpatients with chronic insomnia.28
The sole melatonin‐receptor agonist, ramelteon (Rozerem), also reduces time to fall asleep without next‐day psychomotor and memory effects.29 Ramelteon is believed to target receptors melatonin 1 and 2 receptors located in the brain's suprachiasmatic nucleus to stabilize circadian rhythms and stabilize the sleep‐wake cycle.30
CONCLUSION
Hospitalization is often associated with disrupted sleep, which can affect recovery from illness. Understanding the major factors that impair sleep during hospitalization allows clinicians to systemically evaluate and treat sleep problems. More than just prescribing a sedative/hypnotic, the treatment for sleep disruption includes addressing sleep hygiene and hospital environment issues, identifying medications that could disrupt sleep, and treating specific syndromes that impair sleep. We suggest a practical algorithm to guide clinical assessment, treatment options, and selection of appropriate sleeping medications. Critical to optimizing recovery from illness, sleep may be considered as the sixth vital sign, and should be part of the routine evaluation of every hospitalized patient.
- .Sleep maintenance insomnia: strengths and weaknesses of current pharmacologic therapies.Ann Clin Psychiatry.2006;18(1):49–56.
- ,.Treatment of insomnia.Prim Psychiatry.2005;12(8):47–56.
- ,,.Pharmacologic management of insomnia: past, present, and future.Psychiatr Clin North Am.2006;29:871–893.
- ,.“Hypnotic” prescription patterns in a large managed‐care population.Sleep Med.2004;5(5):463–466.
- ,,, et al.Subjective hypnotic efficacy of trazodone and zolpidem in DSM III‐R primary insomnia.Hum Psychopharmacol.1998;13:191–198.
- ,,, et al.Mirtazapine is more effective than trazodone: a double‐blind controlled study in hospitalized patients with major depression.Int Clin Psychopharmacol.1995;10:3–9.
- ,,.Mirtazapine: an antidepressant with noradrenergic and specific serotonergic effects.Pharmacotherapy.1997;17:10–21.
- ,,,.Efficacy of mirtazapine in obstructive sleep apnea syndrome.Sleep.2007;30(1):35–41.
- ,.Atypical antipsychotics in bipolar disorder: systematic review of randomised trials.BMC Psychiatry.2007;7:40:1–17.
- ,,, et al.Effectiveness of atypical antipsychotic drugs in patients with Alzheimer's Disease.N Engl J Med.2006;355:1525–1538.
- ,,.Risk of death with atypical antipsychotic drug treatment for dementia: meta‐analysis of randomized placebo‐controlled trials.JAMA.2005;294(15):1934–1943.
- ,.Clinical evaluation of diphenhydramine hydrochloride for the treatment of insomnia in psychiatric patients: a double‐blind study.J Clin Pharmacol.1983;23:234–242.
- .Diagnosis and treatment of chronic insomnia: a review.Psychiatr Serv.2005;56:332–343.
- ,,,,.Histamine H1 blocker hydroxyzine improves sleep in patients with cirrhosis and minimal hepatic encephalopathy: a randomized controlled pilot trial.Am J Gastroenterol.2007;102:744–753.
- ,.Clinical effects of chloral hydrate in hospitalized medical patients.J Clin Pharmacol.1979;19(10):669–674.
- Miller RD, editor.Miller's Anesthesia.6th ed.Philadelphia, PA:Elsevier;2005.
- .State‐of‐the‐art sleep management. Awakening insomnia management. Proceedings from a satellite symposium at SLEEP 2006: 20th Anniversary Meeting of the Associated Professional Sleep Societies, Salt Lake City, UT.2006:6–12.
- ,,.Use of a computer‐based reminder to improve sedative‐hypnotic prescribing in older hospitalized patients.J Am Geriatr Soc.2007;55:43–47.
- ,,,.Topiramate use in alprazolam addiction.World J Biol Psychiatry.2006;7(4):265–267.
- ,,,.Trends in recommendations for the pharmacotherapy of anxiety disorders by an international expert panel, 1992–1997.Eur Neuropsychopharmacol.1999;9(Suppl 6):S393–S398.
- ,,,,.Benzodiazepine use and the risk of motor vehicle crash in the elderly.JAMA.1997;278:27–31.
- ,,,,.Psychotropic drug use and the risk of hip fracture.NEngl J Med.1987;316:363–369.
- ,,,,.Sedative hypnotics in older people with insomnia: meta‐analysis of risks and benefits.BMJ.2005;331:1169–1175.
- ,,,,,.Delirium in hospitalized older persons: outcomes and predictors.J Am Geriatr Soc.1994;42:809–815.
- ,,,,.A 5‐year prospective assessment of the risk associated with individual benzodiazdepines and doses in new elderly users.J Am Geriatr Soc.2005;53:233–241.
- ,,, et al.Newer hypnotic drugs for the short‐term management of insomnia: a systematic review and economic evaluation.Health Technol Assess.2004;19:305–322.
- .Medications and their effect on sleep.Prim Care Clin Off Pract.2005;32:401–509.
- ,,,,.Long‐term efficacy and safety of zolpidem extended‐release 12.5 mg, administered 3 to 7 nights per week for 24 weeks, in patients with chronic primary insomnia: a 6‐month, randomized, double‐blind, placebo‐controlled, parallel‐group, multicenter study.Sleep.2008;31(1):79–90.
- ,,,.An efficacy, safety, and dose‐response study of Ramelteon in patients with chronic primary insomnia.Sleep Med.2006;7(1):17–24.
- ,.Melatonin, sleep, and circadian rhythms: rationale for development of specific melatonin agonists.Sleep Med.2004;5(6):523–532.
- .Sleep maintenance insomnia: strengths and weaknesses of current pharmacologic therapies.Ann Clin Psychiatry.2006;18(1):49–56.
- ,.Treatment of insomnia.Prim Psychiatry.2005;12(8):47–56.
- ,,.Pharmacologic management of insomnia: past, present, and future.Psychiatr Clin North Am.2006;29:871–893.
- ,.“Hypnotic” prescription patterns in a large managed‐care population.Sleep Med.2004;5(5):463–466.
- ,,, et al.Subjective hypnotic efficacy of trazodone and zolpidem in DSM III‐R primary insomnia.Hum Psychopharmacol.1998;13:191–198.
- ,,, et al.Mirtazapine is more effective than trazodone: a double‐blind controlled study in hospitalized patients with major depression.Int Clin Psychopharmacol.1995;10:3–9.
- ,,.Mirtazapine: an antidepressant with noradrenergic and specific serotonergic effects.Pharmacotherapy.1997;17:10–21.
- ,,,.Efficacy of mirtazapine in obstructive sleep apnea syndrome.Sleep.2007;30(1):35–41.
- ,.Atypical antipsychotics in bipolar disorder: systematic review of randomised trials.BMC Psychiatry.2007;7:40:1–17.
- ,,, et al.Effectiveness of atypical antipsychotic drugs in patients with Alzheimer's Disease.N Engl J Med.2006;355:1525–1538.
- ,,.Risk of death with atypical antipsychotic drug treatment for dementia: meta‐analysis of randomized placebo‐controlled trials.JAMA.2005;294(15):1934–1943.
- ,.Clinical evaluation of diphenhydramine hydrochloride for the treatment of insomnia in psychiatric patients: a double‐blind study.J Clin Pharmacol.1983;23:234–242.
- .Diagnosis and treatment of chronic insomnia: a review.Psychiatr Serv.2005;56:332–343.
- ,,,,.Histamine H1 blocker hydroxyzine improves sleep in patients with cirrhosis and minimal hepatic encephalopathy: a randomized controlled pilot trial.Am J Gastroenterol.2007;102:744–753.
- ,.Clinical effects of chloral hydrate in hospitalized medical patients.J Clin Pharmacol.1979;19(10):669–674.
- Miller RD, editor.Miller's Anesthesia.6th ed.Philadelphia, PA:Elsevier;2005.
- .State‐of‐the‐art sleep management. Awakening insomnia management. Proceedings from a satellite symposium at SLEEP 2006: 20th Anniversary Meeting of the Associated Professional Sleep Societies, Salt Lake City, UT.2006:6–12.
- ,,.Use of a computer‐based reminder to improve sedative‐hypnotic prescribing in older hospitalized patients.J Am Geriatr Soc.2007;55:43–47.
- ,,,.Topiramate use in alprazolam addiction.World J Biol Psychiatry.2006;7(4):265–267.
- ,,,.Trends in recommendations for the pharmacotherapy of anxiety disorders by an international expert panel, 1992–1997.Eur Neuropsychopharmacol.1999;9(Suppl 6):S393–S398.
- ,,,,.Benzodiazepine use and the risk of motor vehicle crash in the elderly.JAMA.1997;278:27–31.
- ,,,,.Psychotropic drug use and the risk of hip fracture.NEngl J Med.1987;316:363–369.
- ,,,,.Sedative hypnotics in older people with insomnia: meta‐analysis of risks and benefits.BMJ.2005;331:1169–1175.
- ,,,,,.Delirium in hospitalized older persons: outcomes and predictors.J Am Geriatr Soc.1994;42:809–815.
- ,,,,.A 5‐year prospective assessment of the risk associated with individual benzodiazdepines and doses in new elderly users.J Am Geriatr Soc.2005;53:233–241.
- ,,, et al.Newer hypnotic drugs for the short‐term management of insomnia: a systematic review and economic evaluation.Health Technol Assess.2004;19:305–322.
- .Medications and their effect on sleep.Prim Care Clin Off Pract.2005;32:401–509.
- ,,,,.Long‐term efficacy and safety of zolpidem extended‐release 12.5 mg, administered 3 to 7 nights per week for 24 weeks, in patients with chronic primary insomnia: a 6‐month, randomized, double‐blind, placebo‐controlled, parallel‐group, multicenter study.Sleep.2008;31(1):79–90.
- ,,,.An efficacy, safety, and dose‐response study of Ramelteon in patients with chronic primary insomnia.Sleep Med.2006;7(1):17–24.
- ,.Melatonin, sleep, and circadian rhythms: rationale for development of specific melatonin agonists.Sleep Med.2004;5(6):523–532.
Sleep in Hospitalized Medical Patients: Part 1
Hospitalized patients often have difficulty initiating and maintaining sleep, or complain of early awakening and nonrestorative sleep.1 The etiology of sleep disruption is multifactorial and includes the patient's underlying illness(es), medical treatments, and the hospital environment. Often unrecognized and untreated during hospitalization, sleep disruption may lead to sleep deprivation, or a chronic lack of restorative sleep.
Even in healthy individuals, sleep deprivation can result in numerous physical and psychological consequences. Sleep deprivation is associated with hypertension,2, 3 impaired postural control,4 decreased ventilatory drive,5 increased sympathetic cardiovascular activation,6 blunted hypothalamic‐pituitary‐adrenal axis,7 impaired host defenses, and possibly diabetes mellitus and obesity.810 The lack of restorative sleep increases the risk of developing anxiety and mood disorders and delirium, especially in acutely ill older patients.11 In the presence of acute physical infirmity, inadequate sleep may further compound illness and impair recovery. We provide an overview of normal sleep architecture and discuss factors that lead to sleep disruption in hospitalized medical patients.
NORMAL SLEEP ARCHITECTURE AND REGULATION
Normal sleep architecture refers to a characteristic pattern of sleep, and consists of two major stages: nonrapid eye movement (NREM, pronounced non‐rem) and rapid eye movement (REM). (For a table of pertinent abbreviations and terms, see Table 1) Sleep is quantified by polysomnography (PSG), which includes an electroencephalogram (EEG), electromyogram (EMG), and electrooculogram (EOG). A PSG also includes an electrocardiogram (ECG), and measures of airflow, oxygen saturation, and body position. NREM sleep comprises 75% to 80% of total sleep time (TST), and is characterized by relatively quiescent brain activity and decreased metabolic rate.12 NREM sleep consists of four stages (S1‐S4), with each stage leading to a progressively deeper sleep (Figure 1). REM sleep follows slow wave sleep (SWS), or deep sleep, and increases over the night, comprising 20% to 25% of TST. REM sleep is characterized by an activated EEG pattern, muscle atonia, and episodic bursts of rapid eye movements.
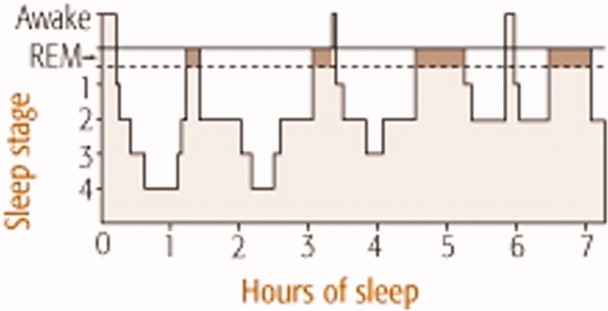
| Acronym | Term |
|---|---|
| BiPAP | Bilevel positive airway pressure |
| CHF | Congestive heart failure |
| CPAP | Continuous positive airway pressure |
| COPD | Chronic obstructive pulmonary disease |
| EEG | Electroencephalogram |
| EOG | Electroculogram |
| EMG | Electromyogram |
| ESRD | End‐stage renal disease |
| NPPV | Noninvasive positive pressure ventilation |
| NREM | Nonrapid eye movement |
| OSA | Obstructive sleep apnea |
| PLMD | Periodic limb movement disorder |
| PSG | Polysomnography |
| RBD | REM sleep behavior disorder |
| REM | Rapid eye movement |
| RLS | Restless leg syndrome |
| S1‐S4 | 4 Stages of sleep in NREM |
| SE | Sleep efficiency; TST divided by total time in bed |
| SWS | Slow wave sleep |
| TBI | Traumatic brain injury |
| TST | Total sleep time |
Normal sleep provides a period of physiologic and mental rest. During sleep, sympathetic tone decreases and parasympathetic tone increases, leading to a reduction in heart rate, arterial blood pressure, and cardiac output.13 Deep sleep is theorized to be necessary for physiologic restoration. REM sleep is associated with dreaming, and is essential for maintaining emotional and cognitive well‐being. Sleep architecture undergoes characteristic changes as people age.14 The duration of SWS peaks in childhood and decreases with age. Consequently, people >60 years old tend to have lower arousal thresholds and to have more frequent awakenings. The results of the Sleep Heart Health Study found that increased age was associated with decreased percentage of REM sleep, worse sleep efficiency (SE, which is TST divided by total time in bed), and lower arousal thresholds.14 With the reduction of SE, older people need to spend more hours in bed to achieve the same amount of restorative sleep as when they were younger. Although sleep tends to become more disrupted as people age, insomnia should not be considered a normal part of aging, and needs to be addressed clinically.15 The results of a National Sleep Foundation telephone survey of subjects between the ages of 55 and 84 years old (n = 1,506) suggested that sleep complaints in older adults are frequently secondary to comorbid medical conditions.16
Multiple anatomic structures, pathways, and neurotransmitter systems are involved in controlling wakefulness and sleep. Neurotransmitters that promote wakefulness include acetylcholine, histamine, noradrenaline (norepinephrine), serotonin, dopamine, and hypocretin (orexin). Sleep‐promoting neurotransmitters include gamma aminobutyric acid (GABA), adenosine, and melatonin. Specific stages of sleep are regulated by the turning on and off of various neurons. REM on cells use GABA, acetylcholine, and glutamine, whereas REM off cells use norepinephrine and serotonin. SWS is promoted by GABA and serotonin.17
Sleep regulation is a balance between a homeostatic sleep need and an intrinsic body clock, or circadian pacemaker. Located in the suprachiasmic nucleus, the circadian pacemaker determines the onset and termination of sleep, and is partially regulated by environmental cues such as light and ambient temperature. Melatonin, a physiologic sleep promoter, is inhibited by ambient light, and its circulation is decreased during daylight hours. The adrenal secretion of cortisol, which is associated with wakefulness, follows a circadian pattern. Regulated by the hypothalamic‐pituitary axis, cortisol levels peak in the early morning hours in preparation for the increased metabolic demands during wakefulness.
SLEEP PROBLEMS IN HOSPITALIZED PATIENTS
Insomnia, which is characterized by difficulty initiating or maintaining sleep, is the most common sleep disorder in the United States. About one‐third of the adult population in the United States experiences insomnia at some point in their lives,18 and it is a persistent problem in approximately 10% of U.S. adults.19 Insomnia can be exacerbated during hospitalization.
Studies investigating sleep in hospitalized patients using PSG have been limited primarily to the setting of the intensive care unit (ICU). Critically ill patients, particularly those requiring mechanical ventilation, are prone to sleep disturbances and an associated delirium.2022 Critically ill patients have fragmented sleep, with decreased SE and SWS, and increased S1 and S2.23 Physician awareness of the impact of sleep disturbance in hospitalized patients is vital. Surveys reveal that approximately one‐half of patients admitted on general medical wards complain of sleep disruption.24, 25 Meissner et al.25 examined the prevalence of sleep complaints and physician recognition of these complaints among general medical patients admitted to a Veterans Affairs tertiary care center. Results showed that 47% (n = 222) of patients had complaints of either insomnia and/or excessive daytime sleepiness.
FACTORS AFFECTING SLEEP DURING HOSPITALIZATION
Many medical and neurologic illnesses, psychiatric disorders, pain, medication therapy and the hospital environment may impair sleep, and hinder recovery from illness.
General Medical Disorders
Primary sleep disorders, such as obstructive sleep apnea (OSA) and numerous other medical illnesses, can directly impair sleep physiology, leading to a cyclical interaction (Figure 2). Other conditions that disrupt sleep include congestive heart failure (CHF), diabetes mellitus, chronic obstructive pulmonary disease (COPD), gastroesophageal reflux, cardiovascular disease, thyroid disorders, renal disease, and severe liver disease.26 Table 2 lists selected medical and neurological conditions, their associated sleep‐related problems, and suggestions on how to ameliorate these problems.
| Disease | Problem | Clinical Implications and Strategies to Improve Sleep |
|---|---|---|
| ||
| Asthma | Nocturnal exacerbation, nocturnal GER | Inhaled corticosteroids and/or long‐acting inhaled beta‐adrenergic agents |
| CHF | Orthopnea, paroxysmal nocturnal dyspnea, sleep‐disordered breathing, increased sympathetic tone, nighttime diuresis, Cheyne‐Stokes respiration | Keep the head of bed elevated 30 degrees. Nocturnal O2 to keep O2 saturation >88%. Daytime diuresis. Optimize cardiac function to treat Cheyne‐Stokes respiration. Consider CPAP for CHF |
| COPD | Persistent nocturnal hypoxemia with complications (e.g., cor pulmonale, polycythemia) | O2 for COPD and persistent hypoxemia (PaO2 55‐60 mm Hg) |
| Sporadic nighttime desaturations | PaO2 55 mm Hg monitor O2 saturation by pulse oximetry. If patient desaturates to 88% at night consistently, start nocturnal O2. For hypercapnia, adjust O2 to maintain O2 saturation at 88% to 90% | |
| Early‐morning airflow obstruction | Consider bedtime tiotropium and inhaled long‐acting beta‐adrenergic agonist agents | |
| Inhibition of respiratory muscles in REM | Avoid sedative‐hypnotics that cause respiratory depression | |
| Decreased functional residual capacity from recumbent position during sleep | Minimize recumbancy by keeping the head of bed up at 30 degrees | |
| End‐stage renal disease | Pruritus, nausea; increased risk of RLS and PLMD | Ambulation may help with RLS. Consider ropinirole and pramipexole. Correct hyperphosphatemia and uremia. Consider antipruritic and antiemetic agents |
| Nocturnal GER | Nocturnal GER decreased sleep, heartburn, coughing, asthma | Avoid eating or drinking 2 hours before bedtime, especially those that delay gastric emptying, increase acid secretion, or decrease lower esophageal sphincter pressure; e.g., high‐fat foods, ethanol, chocolate, peppers, peppermint. Keep head of bed 30 degrees. Minimize medications that could worsen nocturnal GER; e.g., theophylline, calcium channel blockers, prostaglandins, bisphosphonates |
| OSA | Snoring with upper airway obstruction | No ethanol 2 hours before bedtime. Minimize CNS depressants. Avoid supine position. Consider CPAP, oral mandibular advancement device, and/or surgical correction. Long‐term plan should include weight loss |
| Stroke | Focal neurologic deficits (e.g., dysphagia, weakness or paralysis) | Keep head of bed 30 degrees. Regularly suction secretions. Post‐stroke patients have an increased risk of hypersomnia, insomnia, and/or OSA |
Affecting approximately 24% of men and 9% of women in the United States, OSA is the most common primary sleep disorder,27, 28 and causes significant mental and physical morbidity. Risk factors for OSA include obesity, hypothyroidism‐induced muscle weakness, and structural abnormalities in the oropharynx region such as acromegaly, micrognathia, or retrognathia. OSA is characterized by episodes of complete or partial pharyngeal obstruction during sleep that cause snoring, apneic episodes, choking, dyspnea, and restlessness.28 These episodes are associated with intermittent nocturnal sympathetic activation leading to nocturnal awakenings and cortical arousals, all of which lead to daytime symptoms of fatigue, sleepiness, and cognitive impairment (Figure 2). In addition, chronic sympathetic activation causes numerous derangements in the vascular endothelium and platelet activation.29, 30 Sleep‐disordered breathing has been independently associated with cardiovascular diseases such as hypertension, CHF, ischemic heart disease, atrial fibrillation, and cerebrovascular disease.31, 32
OSA is also associated with sleep‐related gastroesophageal reflux, which is characterized by pain and nocturnal cough, and can induce nocturnal asthma attacks and laryngospasm.33 Green et al.29 found that OSA patients treated with continuous positive airway pressure (CPAP) had a 48% improvement in nocturnal reflux symptoms. Although the pathophysiology connecting OSA to the renal system is unknown, OSA has been found in up to 60% of patients with end‐stage renal disease and chronic renal failure.34
Patients with pulmonary disorders can be profoundly affected by the normal physiologic changes during sleep, particularly in REM sleep. During REM sleep, all respiratory muscles except the diaphragm become paralyzed. Thus, episodes of marked oxygen desaturation can occur in patients who rely on their accessory muscles for respiration. COPD patients have decreased TST, SWS, and REM sleep. Shortness of breath, nocturnal cough, and wheezing worsen sleep.35 The resulting fatigue and sleep deprivation negatively impact the work of breathing and impair gas exchange. Airflow obstruction tends to worsen in the early morning hours in patients with COPD and asthma, and may be related to the effect of REM on the accessory muscles for respiration. Although used to target CO2 retention, investigations using bilevel positive airway pressure ventilators (BiPAP) for improving sleep in COPD patients have been limited. Noninvasive positive pressure ventilation (NPPV) appears to acutely improve SE and TST in patients with hypercapnic COPD without significantly improving gas exchange. Other sleep parameters such as sleep architecture and the number of arousals during the night, remain unchanged during NPPV.36
CPAP has several side effects that could worsen sleep, which may explain its poor adherence rate among ambulatory patients.37 Side effects include nasal bridge discomfort, nasal congestion, swallowing air, dry nose, dry or red eyes, noise, ear pain, and rhinitis.38 During hospitalization, efforts should be made to improve patient comfort by resizing ill‐fitting masks, adding heated humidification or nasal steroids to alleviate nasal congestion, or adding a chin strap to reduce air leak and ingestion of air.
Endocrine disorders have also been associated with sleep disruption. Studies suggest that patients with diabetes mellitus have decreased TST and impaired sleep quality due to nocturia and neuropathic pain.39 Inadequate sleep may also affect glucose control. Inadequate quality or quantity of sleep has been shown to be a risk factor for developing Type 2 diabetes mellitus in large prospective studies.40 Sleep duration and quality were significant predictors of increased levels of glycosylated hemoglobin (HbA1c) in patients with Type 2 diabetes mellitus. Thyroid diseases often coexist with diabetes mellitus. Both hypo‐ and hyperthyroidism have been associated with sleep disruption. Hypothyroidism is associated with daytime somnolence and fatigue. Patients with hypothyroidism tend to have reduced SWS. Hyperthyroid patients often complain of insomnia, which has been attributed to a hypermetabolic state.
Approximately 50% of patients with chronic end‐stage renal disease (ESRD) have insomnia and other sleep disorders.41 Patients often complain of restless leg syndrome (RLS), periodic limb movement disorder (PLMD), bone pain, nausea, and pruritus. The etiology of sleep disorders appears to be related to metabolic derangements associated with ESRD or from coexisting diabetes mellitus.
RLS and PLMD are distinct problems that affect sleep differently. RLS is characterized by an unpleasant crampy, creeping or crawling sensation in the lower extremities that is relieved by movement of the legs.42 RLS symptoms typically occur soon after going to bed, and therefore tend to disrupt sleep onset. The requisite bed rest during hospitalization can worsen RLS, further exacerbating sleep problems.43 Since RLS may partially be caused by disrupted iron metabolism, serum ferritin levels should be evaluated.44 Other conditions associated with RLS include pregnancy, rheumatoid arthritis, fibromyalgia, multiple sclerosis, ESRD, and Parkinson's disease. The differential diagnosis for RLS and PLMD includes neuroleptic‐induced akathisia, peripheral neuropathy, and positional or nocturnal leg cramps. PLMD occurs in about 80% of those with RLS, and is characterized by involuntary limb movements that occur every 20 to 40 seconds during NREM sleep. Unaware of these movements, patients often experience frequent arousals throughout the night, and complain of daytime somnolence and fatigue.42
A pilot study of 35 patients with minimal hepatic encephalopathy found that nearly 50% complained of sleep difficulties.45 Hypothesizing that a dysregulation of histaminergic neurotransmission in cirrhosis alters the sleep‐wake cycle, Spahr et al.46 found that 40% of their patients reported subjective improvement in sleep when administered 25 mg of hydroxyzine, compared to none who received placebo.
Neurologic Disorders
Since the brain and its various neurotransmitter systems are critical in regulating sleep and wakefulness, patients with neurologic disorders have an increased risk of developing sleep disorders. Patients with dementia, other neurodegenerative disorders, epilepsy, and traumatic brain injury (TBI) have a higher prevalence of sleep disturbance and sleep disorders.47 Poststroke patients can develop insomnia or hypersomnia, a reduction in sleep latency, increased sleep, or excessive daytime sleepiness, and are at higher risk for OSA during the first several months after a stroke.48 Specific neurologic lesions may lead to uncommon problems such as inversion of the sleep‐wake cycle, parasomnias, and hallucinatory dream‐like states.
Both Parkinson's disease and Alzheimer's disease are associated with multiple sleep disturbances, which tend to worsen with disease progression.14 Common problems include increased sleep fragmentation and wakefulness, with increases of stage 1 sleep and reductions of SWS and REM. Patients with neurodegenerative disorders also have an increased risk of REM sleep behavior disorder, or RBD.49 RBD is characterized by vivid and unusual dreams, and physically vigorous sleep behaviors that may result in ecchymoses, lacerations, and fractures.50 Fifty percent of patients with TBI reported insomnia symptoms.51 Disorders in initiating and maintaining sleep were the most common complaints among hospitalized patients with TBI. Some patients with TBI may develop circadian rhythm disturbances.52
Pain
A majority of patients with chronic pain, 50% to 70%, complain of impaired sleep.53 Sleep disruption is so common in fibromyalgia (75%) that it is considered to be a key diagnostic symptom.54 In a study investigating the affect of pain on sleep in burn patients, pain was associated with increased intermittent awakenings and prolonged periods of wake time during the night.55 The following day, these patients had poorer pain tolerance and greater pain intensity. Pain causes sleep fragmentation by increasing cortical arousals. Recent evidence suggests that sleep deprivation can increase pain sensitivity by inhibiting opioid protein synthesis or reducing opioid receptor affinity.56
Psychiatric Disorders
Sleep problems are so common in psychiatric conditions that the Diagnostic and Statistical Manual of Mental Disorders (DMS‐IV‐TR) includes sleep disturbance as a diagnostic criterion for a manic episode, and for various depressive, anxiety, and substance abuse disorders.57 The presence of sleep disturbance in hospitalized patients may suggest the presence of an underlying psychiatric disorder that would otherwise go unrecognized. In a survey of 200 general medical patients in a Brazilian hospital, Rocha et al.58 found that 112 (56.5%) complained of insomnia, and 100 (50%) met criteria for at least 1 psychiatric disorder. However, only 3 out of the total number of 200 surveyed (1.5%) were identified as having psychiatric diagnoses in the medical record, and sleep history was not noted in the clinical evaluation. An episode of major depressive disorder was the most common psychiatric diagnosis (35%). In this study, hospitalized patients with insomnia had a 3.6 times higher risk of having major depressive disorder than inpatients without insomnia.
Insomnia has a profound effect on mental health by worsening health‐related quality of life. In a study of outpatients at family medicine, internal medicine, endocrinology, cardiology, and psychiatry clinics in 3 U.S. cities (n = 3,445), insomnia worsened health‐related quality of life nearly as much as CHF or major depressive disorder did.59 Another survey of outpatients found that those with chronic insomnia were nearly 40 times more likely to have major depression and 6 times more likely to have an anxiety disorder compared to those without insomnia.60 Longitudinal studies have found that prior insomnia was associated with 2‐ to 5‐fold increase in the odds of mood and anxiety disorders and suicide.61, 62 Examining prodromes and precursors to mental disorders, Eaton et al.63 found that 47% of those with onset of depression at the 1‐year follow‐up had sleep problems at baseline.
An estimated 65% of patients with major depression have difficulty falling asleep, frequent awakenings, or early morning awakenings.64 Three patterns of sleep architecture abnormalities have been observed in patients with major depression: 1) sleep continuity disturbances characterized by prolonged sleep‐onset, increased wake time during sleep, increased early morning wake time, and decreased TST; 2) decreased proportion and length of SWS; and 3) REM sleep abnormalities such as reduced time to REM sleep, prolonged first REM sleep episode, and increased REM sleep percentage.65 Sleep during a manic episode has been less studied than in depression, but the data suggest that abnormal sleep in mania includes disrupted sleep continuity, shortened REM latency, and increased REM density (REM eye movement activity/total REM sleep time).65
Substance use disorders are also associated with sleep problems. In a survey by Brower et al.66 of patients who were undergoing alcohol rehabilitation, 61% (n = 172) had symptoms of insomnia such as increased sleep latency during the 6 months prior to entering treatment. Approximately 45% of these patients reported using alcohol for the purpose of initiating sleep. Alcohol and illicit substance intoxication and withdrawal are known to be associated with disrupted sleep. However, sleep disturbances may persist long after withdrawal symptoms have abated. Drummond et al. found that some patients continued to have alcohol‐associated sleep problems even after 27 months of abstinence.67 Evidence also suggests that untreated insomnia and other sleep problems may increase the risk of developing substance abuse problems due to self‐medicating with alcohol and other substances to help with sleep.68
Drugs that Affect Sleep
Numerous drugs can alter sleep quantity and quality. Sedatives and opioids may initially help with sleep onset, but impair sleep architecture. Medications used to treat medical and psychiatric illnesses also disrupt sleep (Table 3). The most common agents that impair sleep include antiepileptic drugs, selective serotonin reuptake inhibitors, monoamine oxidase inhibitors, tricyclic antidepressants, antihypertensives, antihistamines, and corticosteroids.
| Drug Class | Examples of Drugs | Affect on Sleep Architecture | Potential Mechanism | Clinical Implications |
|---|---|---|---|---|
| ||||
| CNS | ||||
| AEDs | Phenobarbital, carbamazepine, phenytoin | Very sedating. AEDs tend to TST, sleep latency | Inhibit neuronal calcium influx, adenosine, or 5HT activity | Sedation is dose‐dependent, and tends to occur with acute use |
| TCAs | Amoxapine, amitriptyline, imipramine, nortriptyline, desipramine, doxepin, clomipramine | Very sedating. Suppresses REM sleep, TST, stage‐2 sleep | Stimulate antimuscarinic‐receptor and alpha1‐receptor | Suppressed REM sleep motor inhibition restlessness, psychomotor agitation during sleep subjectively sleep quality, daytime sleepiness |
| BzRAs | Alprazolam, lorazepam, chlordiazepoxide, diazepam, oxazepam | Very sedating. TST, sleep latency, SWS duration, REM, stage‐2 sleep | Stimulate GABA type A receptor | Minimize daytime use. Chronic BzRAs SWS long‐term sequelae unknown |
| MAOIs | Phenylzine, tranylcypromine | Very sedating. TST, REM, REM rebound if stop MAOIs | Mechanism unknown | Daytime sleepiness; dosing time does not affect daytime somnolence |
| SSRIs | Sedating: paroxetine, fluvoxamine. Activating: fluoxetine, sertraline, citalopram | TST, are less sedating than TCAs and MAOIs. May REM, TWT, TST, SE | 5HT activity | Some patients get the opposite reaction |
| SNRI | Venlafaxine, duloxetine | Activating in some patients; sedating in 12% to 31%. TST | 5HT and NE activity | If activating, switch to AM dosing. If sedating, switch to PM dosing |
| Mood stabilizer | Lithium | Sedating. TST, SWS, REM, REM latency | daytime sedation. Dose at night | |
| Stimulants | Ephedrine, pseudoephedrine, modafinil | Activating. TST, SWS, sleep latency | DOPA, NE, and 5HT activity | Avoid after 6 PM |
| Anti‐Parkinson | Bromocriptine, levodopa | Sedating. Nightmares, SWS | DOPA | Dose at night, if possible |
| Cardiac | ||||
| Lipophilic beta‐blockers | Propranolol, pindolol, metoprolol, timolol. Hydrophilic agents (atenolol and sotalol) lack these effects | Activating. awakenings, TWT, REM, nightmares | CNS beta‐blockade | Lipophilic beta‐blockers daytime sleep when dosed in AM |
| CNS agents | Norepinephrine, epinephrine | Activating. REM, SWS | Stimulate alpha1‐receptor | Minimize use at night |
| Dopamine | Activating. REM, SWS | Stimulate dopamine2‐receptor and alpha1‐receptor | Minimize use at night | |
| Ca++ channel blockers | Amlodipine, verapamil, nifedipine | Exacerbate underlying medical condition | Lower esophageal sphincter tone nocturnal GER sleep disturbance | |
| Alpha2‐receptor agonist | Clonidine | Stage 1, REM, nightmares | Stimulate alpha2‐receptor | Alpha2‐agonists daytime sleep and sleepiness directly. Dose at night |
| Alpha1‐receptor blockers | Doxazosin, prazosin, terazosin | Inhibit alpha1‐receptor | Alpha1‐receptor blockers daytime sleepiness | |
| Diuretics | HCTZ, furosemide | Sedating. | PM diuresis frequent awakenings | |
| Other | ||||
| Opioids | Codeine, morphine | Sedating. SWS, REM | Stimulate mu‐receptor | Minimize use at night |
| NSAIDs | Ibuprofen, indomethcin, celecoxib | TST, SE | Inhibit prostaglandin synthesis | Minimize use at night |
| Methylxanthine | Theophylline | Activating. stage 1, REM | Causes less restful sleep | |
| Antihistamines | Diphenhydramine, promethazine | Sedating | H1 receptor blockade | Minimize use at night |
| Corticosteroids | Dexamethasone, prednisone | Activating. REM, SWS, nightmares | Melatonin secretion | Can disrupt sleep, anxiety, induce mania or psychosis |
| H2 blockers | Cimetidine, ranitidine, famotidine | Sedating. TST | H2 receptor blockade | Sedating if >60 years old, renal impairment |
| Quinolone | Ciprofloxacin, sparfloxacin, ofloxacin, grepafloxacin, levofloxacin | Activating | Stimulate GABA type A receptor | Consider sleep agent after maximizing sleep hygiene. Linezolid rarely causes sleep disturbances |
Lipophilic beta antagonists such as propranolol and timolol can increase total wake time, decrease REM sleep, and increase the incidence of nightmares and insomnia.69 Anabolic steroids and beta‐agonist bronchodilator therapy can cause severe anxiety, sleeplessness, and even psychosis. Vasopressor agents such as dopamine can cause cortical activation, leading to increased arousal and reduced SWS.
Hospital Environment
Environmental noise and patient care activities often interfere with sleep in the hospital. They account for about 30% of patient awakenings in ICU patients.70 Noise levels in the ICU have average sound peaks of 150 to 200 dB, and evening peaks >80 dB between midnight and 6 AM.71 By comparison, the front row seats at a rock concert have sound levels of 110 dB. The high noise level in hospitals has long been implicated as a sleep disruptor,72 but studies in the past decade have found that patient care activities probably contribute more to awakenings than does environmental noise.73 An analysis of critical care nursing routines found that activities such as taking vital signs and giving baths occurred a mean 42.6 times a night per patient.74 Tamburri et al.74 found that patients experienced 2 to 3 hours without interruption on only 6% of the 147 nights studied. Routine daily baths were provided on 55 of the 147 study nights between 2 AM and 5 AM, which is unlikely to be an opportune time for most patients.
CONCLUSION
Hospitalization often prevents patients from achieving adequate sleep and can affect recovery from illness. Understanding the major factors that impair sleep during hospitalization allows clinicians to systemically evaluate and treat sleep problems. More than just prescribing sedative/hypnotic agents, the treatment for sleep disruption includes addressing multiple medical, behavioral, and environmental factors, which will be discussed in Part 2 of this article.
- NIH State‐of‐the Science Conference Statement on Manifestations and Management of Chronic Insomnia in Adults.NIH Consens Sci Statements.2005;22(2):1–30.
- ,,, et al.Short sleep duration as a risk factor for hypertension: analyses of the first National Health and Nutrition Examination Survey.Hypertension.2006;47(5):833–839.
- ,,, et al.Total sleep deprivation elevates blood pressure through arterial baroreflex resetting: a study with microneurographic technique.Sleep.2003;26(8):986–989.
- ,,,,.Postural control after a night without sleep.Neuropsychologia.2006;44(12):2520–2525.
- .Sleep deprivation decreases ventilatory responses to CO2 but not load compensation.Chest.1983;84(6):695–698.
- ,,, et al.Increased sympathetic and decreased parasympathetic cardiovascular modulation in normal humans with acute sleep deprivation.J Appl Physiol.2005;98(6):2024–2032.
- .Reductions in circulating anabolic hormones induced by sustained sleep deprivation in rats.Am J Physiol Endocrinol Metab.2004;286(6):E1060–E1070.
- ,,,.The metabolic consequences of sleep deprivation.Sleep Med Rev.2007;11(3):163–178.
- ,,,,.Sleep deprivation and activation of morning levels of cellular and genomic markers of inflammation.Arch Int Med.2006;166:1756–1752.
- ,,,.Effects of sleep and sleep deprivation on immunoglobulins and complement in humans.Brain Behav Immun.2007;21:308–310.
- ,,,,.The effects of sleep deprivation on symptoms of psychopathology in healthy adults.Sleep Med.2007;8:215–221.
- ,.Normal human sleep. In:Kryger MH,Roth T,Dement WC, editors.Principles and Practice of Sleep Medicine.3rd ed.Philadelphia:W.B. Saunders;2000:15–16.
- .Update on the Science, Diagnosis and Management of Insomnia.International Congress and Symposium Series 262.London:Royal Society of Medicine Press Ltd;2006.
- ,,,,,.The effects of age, sex, ethnicity, and sleep‐disordered breathing on sleep architecture.Arch Intern Med.2004;164:406–18.
- ,.Sleep disruption in older adults.Am J Nurs.2007;107(5):40–49.
- ,,,.Sleep disturbances and chronic disease in older adults: results of the 2003 National Sleep Foundation Sleep in America Survey.J Psychosom Res.2004;56(5):497–502.
- .Sleep in patients with neurologic and psychiatric disorders.Prim Care.2005;32:535–548.
- ,,.Insomnia and its treatment: prevalence and correlates.Arch Gen Psychiatry.1985;42(3):225–232.
- ,.Epidemiologic study of sleep disturbances and psychiatric disorders: an opportunity for prevention?JAMA.1989;262:1479–1484.
- ,,,,,.Sleep in critically ill patients requiring mechanical ventilation.Chest.2000;117:809–818.
- ,.Sedative and analgesic medications: risk factors for delirium and sleep disturbances in the critically ill.Crit Care Med.2006;22:313–327.
- ,,,,.Quantity and quality of sleep in the surgical intensive care unit: are our patients sleeping?J Trauma.2007;63:1210–1214.
- .Sleep in acute care units.Sleep Breath.2006;10:6–15.
- ,,,,,.An assessment of quality of sleep and the use of drugs with sedating properties in hospitalized adult patients.Health Qual Life Outcomes.2004;2:17.
- ,,,,,.Failure of physician documentation of sleep complaints in hospitalized patients.West J Med.1998;169:146–149.
- .Sleep and medical disorders.Prim Care.2005;35:511–533.
- ,,,,,.The occurrence of sleep‐disordered breathing among middle‐aged adults.N Engl J Med.1993;328:1230–1235.
- ,.Clinical features and evaluation of obstructive sleep apnea‐hypopnea syndrome. In:Kryger MH,Roth T,Dement WC, editors.Principles and Practice of Sleep Medicine.3rd ed.Philadelphia:W.B. Saunders;2000:869–872.
- ,,.Marked improvement in nocturnal gastroesophageal reflux in a large cohort of patients with obstructive sleep apnea.Arch Intern Med.2003;163:41–45.
- ,.Sleep and medical disorders.Med Clin North Am.2004;88:679–703.
- ,,,.Relation of sleep‐disordered breathing to carotid plaque and intima‐media thickness.Atherosclerosis.2008;197(1):125– 131.
- ,,, et al.Sleep‐disordered breathing and cardiovascular disease: cross‐sectional results of the Sleep Heart Health Study.Am J Respir Crit Care Med.2001;163:19–25.
- .Gastroesophageal reflux during sleep.Sleep Med Clin.2007;2:41–50.
- ,.Sleep apnea in renal failure.Adv Perit Dial.1997;13:88–92.
- .Sleep in chronic obstructive pulmonary disease.Sleep Med Clin.2007;2:1–8.
- ,,,.Effects of non‐invasive positive pressure ventilation on gas exchange and sleep in COPD patients.Chest.1997;112:623–628.
- ,,, et al.Night‐to‐night variability in CPAP use over the first three months of treatment.Sleep.1997;20(4):278–283.
- ,.Improving CPAP use by patients with the sleep apnoea/hypopnoea syndrome (SAHS).Sleep Med Rev.2003;7(1):81–99.
- ,,.Factors predicting sleep disruption in type II diabetes.Sleep.2000;23:415–416.
- ,,.Sleep duration as a risk factor for the development of type 2 diabetes.Diabetes Care.2006;29:657–661.
- .Sleep disorders and end‐stage renal disease.Sleep Med Clin.2007;2:59–66.
- ,,,.Sleep and aging: 1. Sleep disorders commonly found in older people.CMAJ.2007;176(9):1299–1304.
- ,,,,,.Impact, diagnosis and treatment of restless legs syndrome (RLS) in a primary care population: The REST (RLS Epidemiology, Symptoms, and Treatment) primary care study.Sleep Med.2004;5(3):237–246.
- ,.Epidemiology and clinical findings of restless leg syndrome.Sleep Med.2004;5(3):293–299.
- ,,,,,.High prevalence of sleep disturbance in cirrhosis.Hepatology.1998;27:339–345.
- ,,,,.Histamine H1 blocker hydroxyzine improves sleep in patients with cirrhosis and minimal hepatic encephalopathy: a randomized controlled pilot trial.Am J Gastroenterol.2007;102:744–753.
- .Sleep in patients with neurologic and psychiatric disorders.Prim Care.2005;32:535–548.
- ,,.Obstructive sleep apnea: implications for cardiac and vascular disease.JAMA.2003;290(14):1906–1914.
- ,,, et al.Pathophysiology of REM sleep behaviour disorder and relevance to neurodegenerative disease.Brain.2007;130(Pt 11):2770–2788.
- ,.REM sleep parasomnias. In:Kryger MH,Roth T,Dement WC, editors.Principles and Practice of Sleep Medicine.3rd ed.Philadelphia:W.B. Saunders;2000:724–725.
- ,,.Insomnia in patients with traumatic brain injury.J Head Trauma Rehabil.2006;21(3):199–212.
- ,,,,.Circadian rhythm sleep disorders following mild traumatic brain injury.Neurology.2007;68(14):1136–1140.
- ,.Comorbidities: psychiatric, medical, medications, and substances.Sleep Med Clin.2006;231–245.
- ,,.Sleep disturbance and fibromyalgia.Sleep Med Clin.2007;2:31–39.
- ,,.Sleep disturbances, pain and analgesia in adults hospitalized for burn injuries.Sleep Med.2004;5:551–559.
- ,,.Sleep deprivation and pain perception.Sleep Med Rev.2006;10:357–369.
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders.4th ed. Text Revision.Washington, DC:American Psychiatric Association;2000.
- ,,, et al.Is insomnia a marker for psychiatric disorders in general hospitals?Sleep Med.2005;6:549–553.
- ,.The relationship between insomnia and health‐related quality of life in patients with chronic illness.J Fam Pract.2002;51(3):229–235.
- ,.Epidemiologic study of sleep disturbances and psychiatric disorders: an opportunity for prevention?JAMA.1989;262:1479–1484.
- ,,,.Sleep disturbance and psychiatric disorders: a longitudinal epidemiological study of young adults.Biol Psychiatry.1996;39:411–418.
- ,,,.The morbidity of insomnia uncomplicated by psychiatric disorders.Gen Hosp Psychiatry.1997;19:245–250.
- ,,.Prodromes and precursors: epidemiologic data for primary prevention of disorders with slow onset.Am J Psychiatry.1995;152:967–972.
- ,,,,,.Which depressive symptoms are related to which sleep electroencephalographic variables?Biol Psychol.1997;42:904–913.
- ,.Sleep in mood disorders.Psychiatr Clin North Am.2006;29:1009–1032.
- ,,,,.Insomnia, self‐medication, and relapse to alcoholism.Am J Psychiatry.2001;158:399–404.
- ,,,.The sleep of abstinent pure primary alcoholic patients: natural course and relationship to relapse.Alcohol Clin Exp Res.1998;22:1796–1802.
- ,,,,.Screening for substance use patterns among patients referred for a variety of sleep complaints.Am J Drug Alcohol Abuse.2006;32:111–120.
- .Drugs that disturb sleep and wakefulness. In:Kryger MH,Roth T,Dement WC, editors.Principles and Practice of Sleep Medicine.3rd ed.Philadelphia:W.B. Saunders;2000:441–462.
- ,,, et al.Contribution of the intensive care unit environment to sleep disruption in mechanically ventilated patients and healthy subjects.Am J Respir Crit Care Med.2003;167(5):708–715.
- ,,.Adverse environmental conditions in the respiratory and medical ICU settings.Chest.1994;105:1211–1216.
- ,,,,,.Noise levels in Johns Hopkins Hospital.J Acoust Soc Am.2005;118(6):3629–3645.
- ,,.Patient perception of sleep quality and etiology of sleep disruption in the intensive care unit.Am J Respir Crit Care Med.1999;159:1155–1162.
- ,,,.Nocturnal care interactions with patients in critical care units.Am J Crit Care.2004;13(2):102–115.
Hospitalized patients often have difficulty initiating and maintaining sleep, or complain of early awakening and nonrestorative sleep.1 The etiology of sleep disruption is multifactorial and includes the patient's underlying illness(es), medical treatments, and the hospital environment. Often unrecognized and untreated during hospitalization, sleep disruption may lead to sleep deprivation, or a chronic lack of restorative sleep.
Even in healthy individuals, sleep deprivation can result in numerous physical and psychological consequences. Sleep deprivation is associated with hypertension,2, 3 impaired postural control,4 decreased ventilatory drive,5 increased sympathetic cardiovascular activation,6 blunted hypothalamic‐pituitary‐adrenal axis,7 impaired host defenses, and possibly diabetes mellitus and obesity.810 The lack of restorative sleep increases the risk of developing anxiety and mood disorders and delirium, especially in acutely ill older patients.11 In the presence of acute physical infirmity, inadequate sleep may further compound illness and impair recovery. We provide an overview of normal sleep architecture and discuss factors that lead to sleep disruption in hospitalized medical patients.
NORMAL SLEEP ARCHITECTURE AND REGULATION
Normal sleep architecture refers to a characteristic pattern of sleep, and consists of two major stages: nonrapid eye movement (NREM, pronounced non‐rem) and rapid eye movement (REM). (For a table of pertinent abbreviations and terms, see Table 1) Sleep is quantified by polysomnography (PSG), which includes an electroencephalogram (EEG), electromyogram (EMG), and electrooculogram (EOG). A PSG also includes an electrocardiogram (ECG), and measures of airflow, oxygen saturation, and body position. NREM sleep comprises 75% to 80% of total sleep time (TST), and is characterized by relatively quiescent brain activity and decreased metabolic rate.12 NREM sleep consists of four stages (S1‐S4), with each stage leading to a progressively deeper sleep (Figure 1). REM sleep follows slow wave sleep (SWS), or deep sleep, and increases over the night, comprising 20% to 25% of TST. REM sleep is characterized by an activated EEG pattern, muscle atonia, and episodic bursts of rapid eye movements.

| Acronym | Term |
|---|---|
| BiPAP | Bilevel positive airway pressure |
| CHF | Congestive heart failure |
| CPAP | Continuous positive airway pressure |
| COPD | Chronic obstructive pulmonary disease |
| EEG | Electroencephalogram |
| EOG | Electroculogram |
| EMG | Electromyogram |
| ESRD | End‐stage renal disease |
| NPPV | Noninvasive positive pressure ventilation |
| NREM | Nonrapid eye movement |
| OSA | Obstructive sleep apnea |
| PLMD | Periodic limb movement disorder |
| PSG | Polysomnography |
| RBD | REM sleep behavior disorder |
| REM | Rapid eye movement |
| RLS | Restless leg syndrome |
| S1‐S4 | 4 Stages of sleep in NREM |
| SE | Sleep efficiency; TST divided by total time in bed |
| SWS | Slow wave sleep |
| TBI | Traumatic brain injury |
| TST | Total sleep time |
Normal sleep provides a period of physiologic and mental rest. During sleep, sympathetic tone decreases and parasympathetic tone increases, leading to a reduction in heart rate, arterial blood pressure, and cardiac output.13 Deep sleep is theorized to be necessary for physiologic restoration. REM sleep is associated with dreaming, and is essential for maintaining emotional and cognitive well‐being. Sleep architecture undergoes characteristic changes as people age.14 The duration of SWS peaks in childhood and decreases with age. Consequently, people >60 years old tend to have lower arousal thresholds and to have more frequent awakenings. The results of the Sleep Heart Health Study found that increased age was associated with decreased percentage of REM sleep, worse sleep efficiency (SE, which is TST divided by total time in bed), and lower arousal thresholds.14 With the reduction of SE, older people need to spend more hours in bed to achieve the same amount of restorative sleep as when they were younger. Although sleep tends to become more disrupted as people age, insomnia should not be considered a normal part of aging, and needs to be addressed clinically.15 The results of a National Sleep Foundation telephone survey of subjects between the ages of 55 and 84 years old (n = 1,506) suggested that sleep complaints in older adults are frequently secondary to comorbid medical conditions.16
Multiple anatomic structures, pathways, and neurotransmitter systems are involved in controlling wakefulness and sleep. Neurotransmitters that promote wakefulness include acetylcholine, histamine, noradrenaline (norepinephrine), serotonin, dopamine, and hypocretin (orexin). Sleep‐promoting neurotransmitters include gamma aminobutyric acid (GABA), adenosine, and melatonin. Specific stages of sleep are regulated by the turning on and off of various neurons. REM on cells use GABA, acetylcholine, and glutamine, whereas REM off cells use norepinephrine and serotonin. SWS is promoted by GABA and serotonin.17
Sleep regulation is a balance between a homeostatic sleep need and an intrinsic body clock, or circadian pacemaker. Located in the suprachiasmic nucleus, the circadian pacemaker determines the onset and termination of sleep, and is partially regulated by environmental cues such as light and ambient temperature. Melatonin, a physiologic sleep promoter, is inhibited by ambient light, and its circulation is decreased during daylight hours. The adrenal secretion of cortisol, which is associated with wakefulness, follows a circadian pattern. Regulated by the hypothalamic‐pituitary axis, cortisol levels peak in the early morning hours in preparation for the increased metabolic demands during wakefulness.
SLEEP PROBLEMS IN HOSPITALIZED PATIENTS
Insomnia, which is characterized by difficulty initiating or maintaining sleep, is the most common sleep disorder in the United States. About one‐third of the adult population in the United States experiences insomnia at some point in their lives,18 and it is a persistent problem in approximately 10% of U.S. adults.19 Insomnia can be exacerbated during hospitalization.
Studies investigating sleep in hospitalized patients using PSG have been limited primarily to the setting of the intensive care unit (ICU). Critically ill patients, particularly those requiring mechanical ventilation, are prone to sleep disturbances and an associated delirium.2022 Critically ill patients have fragmented sleep, with decreased SE and SWS, and increased S1 and S2.23 Physician awareness of the impact of sleep disturbance in hospitalized patients is vital. Surveys reveal that approximately one‐half of patients admitted on general medical wards complain of sleep disruption.24, 25 Meissner et al.25 examined the prevalence of sleep complaints and physician recognition of these complaints among general medical patients admitted to a Veterans Affairs tertiary care center. Results showed that 47% (n = 222) of patients had complaints of either insomnia and/or excessive daytime sleepiness.
FACTORS AFFECTING SLEEP DURING HOSPITALIZATION
Many medical and neurologic illnesses, psychiatric disorders, pain, medication therapy and the hospital environment may impair sleep, and hinder recovery from illness.
General Medical Disorders
Primary sleep disorders, such as obstructive sleep apnea (OSA) and numerous other medical illnesses, can directly impair sleep physiology, leading to a cyclical interaction (Figure 2). Other conditions that disrupt sleep include congestive heart failure (CHF), diabetes mellitus, chronic obstructive pulmonary disease (COPD), gastroesophageal reflux, cardiovascular disease, thyroid disorders, renal disease, and severe liver disease.26 Table 2 lists selected medical and neurological conditions, their associated sleep‐related problems, and suggestions on how to ameliorate these problems.

| Disease | Problem | Clinical Implications and Strategies to Improve Sleep |
|---|---|---|
| ||
| Asthma | Nocturnal exacerbation, nocturnal GER | Inhaled corticosteroids and/or long‐acting inhaled beta‐adrenergic agents |
| CHF | Orthopnea, paroxysmal nocturnal dyspnea, sleep‐disordered breathing, increased sympathetic tone, nighttime diuresis, Cheyne‐Stokes respiration | Keep the head of bed elevated 30 degrees. Nocturnal O2 to keep O2 saturation >88%. Daytime diuresis. Optimize cardiac function to treat Cheyne‐Stokes respiration. Consider CPAP for CHF |
| COPD | Persistent nocturnal hypoxemia with complications (e.g., cor pulmonale, polycythemia) | O2 for COPD and persistent hypoxemia (PaO2 55‐60 mm Hg) |
| Sporadic nighttime desaturations | PaO2 55 mm Hg monitor O2 saturation by pulse oximetry. If patient desaturates to 88% at night consistently, start nocturnal O2. For hypercapnia, adjust O2 to maintain O2 saturation at 88% to 90% | |
| Early‐morning airflow obstruction | Consider bedtime tiotropium and inhaled long‐acting beta‐adrenergic agonist agents | |
| Inhibition of respiratory muscles in REM | Avoid sedative‐hypnotics that cause respiratory depression | |
| Decreased functional residual capacity from recumbent position during sleep | Minimize recumbancy by keeping the head of bed up at 30 degrees | |
| End‐stage renal disease | Pruritus, nausea; increased risk of RLS and PLMD | Ambulation may help with RLS. Consider ropinirole and pramipexole. Correct hyperphosphatemia and uremia. Consider antipruritic and antiemetic agents |
| Nocturnal GER | Nocturnal GER decreased sleep, heartburn, coughing, asthma | Avoid eating or drinking 2 hours before bedtime, especially those that delay gastric emptying, increase acid secretion, or decrease lower esophageal sphincter pressure; e.g., high‐fat foods, ethanol, chocolate, peppers, peppermint. Keep head of bed 30 degrees. Minimize medications that could worsen nocturnal GER; e.g., theophylline, calcium channel blockers, prostaglandins, bisphosphonates |
| OSA | Snoring with upper airway obstruction | No ethanol 2 hours before bedtime. Minimize CNS depressants. Avoid supine position. Consider CPAP, oral mandibular advancement device, and/or surgical correction. Long‐term plan should include weight loss |
| Stroke | Focal neurologic deficits (e.g., dysphagia, weakness or paralysis) | Keep head of bed 30 degrees. Regularly suction secretions. Post‐stroke patients have an increased risk of hypersomnia, insomnia, and/or OSA |
Affecting approximately 24% of men and 9% of women in the United States, OSA is the most common primary sleep disorder,27, 28 and causes significant mental and physical morbidity. Risk factors for OSA include obesity, hypothyroidism‐induced muscle weakness, and structural abnormalities in the oropharynx region such as acromegaly, micrognathia, or retrognathia. OSA is characterized by episodes of complete or partial pharyngeal obstruction during sleep that cause snoring, apneic episodes, choking, dyspnea, and restlessness.28 These episodes are associated with intermittent nocturnal sympathetic activation leading to nocturnal awakenings and cortical arousals, all of which lead to daytime symptoms of fatigue, sleepiness, and cognitive impairment (Figure 2). In addition, chronic sympathetic activation causes numerous derangements in the vascular endothelium and platelet activation.29, 30 Sleep‐disordered breathing has been independently associated with cardiovascular diseases such as hypertension, CHF, ischemic heart disease, atrial fibrillation, and cerebrovascular disease.31, 32
OSA is also associated with sleep‐related gastroesophageal reflux, which is characterized by pain and nocturnal cough, and can induce nocturnal asthma attacks and laryngospasm.33 Green et al.29 found that OSA patients treated with continuous positive airway pressure (CPAP) had a 48% improvement in nocturnal reflux symptoms. Although the pathophysiology connecting OSA to the renal system is unknown, OSA has been found in up to 60% of patients with end‐stage renal disease and chronic renal failure.34
Patients with pulmonary disorders can be profoundly affected by the normal physiologic changes during sleep, particularly in REM sleep. During REM sleep, all respiratory muscles except the diaphragm become paralyzed. Thus, episodes of marked oxygen desaturation can occur in patients who rely on their accessory muscles for respiration. COPD patients have decreased TST, SWS, and REM sleep. Shortness of breath, nocturnal cough, and wheezing worsen sleep.35 The resulting fatigue and sleep deprivation negatively impact the work of breathing and impair gas exchange. Airflow obstruction tends to worsen in the early morning hours in patients with COPD and asthma, and may be related to the effect of REM on the accessory muscles for respiration. Although used to target CO2 retention, investigations using bilevel positive airway pressure ventilators (BiPAP) for improving sleep in COPD patients have been limited. Noninvasive positive pressure ventilation (NPPV) appears to acutely improve SE and TST in patients with hypercapnic COPD without significantly improving gas exchange. Other sleep parameters such as sleep architecture and the number of arousals during the night, remain unchanged during NPPV.36
CPAP has several side effects that could worsen sleep, which may explain its poor adherence rate among ambulatory patients.37 Side effects include nasal bridge discomfort, nasal congestion, swallowing air, dry nose, dry or red eyes, noise, ear pain, and rhinitis.38 During hospitalization, efforts should be made to improve patient comfort by resizing ill‐fitting masks, adding heated humidification or nasal steroids to alleviate nasal congestion, or adding a chin strap to reduce air leak and ingestion of air.
Endocrine disorders have also been associated with sleep disruption. Studies suggest that patients with diabetes mellitus have decreased TST and impaired sleep quality due to nocturia and neuropathic pain.39 Inadequate sleep may also affect glucose control. Inadequate quality or quantity of sleep has been shown to be a risk factor for developing Type 2 diabetes mellitus in large prospective studies.40 Sleep duration and quality were significant predictors of increased levels of glycosylated hemoglobin (HbA1c) in patients with Type 2 diabetes mellitus. Thyroid diseases often coexist with diabetes mellitus. Both hypo‐ and hyperthyroidism have been associated with sleep disruption. Hypothyroidism is associated with daytime somnolence and fatigue. Patients with hypothyroidism tend to have reduced SWS. Hyperthyroid patients often complain of insomnia, which has been attributed to a hypermetabolic state.
Approximately 50% of patients with chronic end‐stage renal disease (ESRD) have insomnia and other sleep disorders.41 Patients often complain of restless leg syndrome (RLS), periodic limb movement disorder (PLMD), bone pain, nausea, and pruritus. The etiology of sleep disorders appears to be related to metabolic derangements associated with ESRD or from coexisting diabetes mellitus.
RLS and PLMD are distinct problems that affect sleep differently. RLS is characterized by an unpleasant crampy, creeping or crawling sensation in the lower extremities that is relieved by movement of the legs.42 RLS symptoms typically occur soon after going to bed, and therefore tend to disrupt sleep onset. The requisite bed rest during hospitalization can worsen RLS, further exacerbating sleep problems.43 Since RLS may partially be caused by disrupted iron metabolism, serum ferritin levels should be evaluated.44 Other conditions associated with RLS include pregnancy, rheumatoid arthritis, fibromyalgia, multiple sclerosis, ESRD, and Parkinson's disease. The differential diagnosis for RLS and PLMD includes neuroleptic‐induced akathisia, peripheral neuropathy, and positional or nocturnal leg cramps. PLMD occurs in about 80% of those with RLS, and is characterized by involuntary limb movements that occur every 20 to 40 seconds during NREM sleep. Unaware of these movements, patients often experience frequent arousals throughout the night, and complain of daytime somnolence and fatigue.42
A pilot study of 35 patients with minimal hepatic encephalopathy found that nearly 50% complained of sleep difficulties.45 Hypothesizing that a dysregulation of histaminergic neurotransmission in cirrhosis alters the sleep‐wake cycle, Spahr et al.46 found that 40% of their patients reported subjective improvement in sleep when administered 25 mg of hydroxyzine, compared to none who received placebo.
Neurologic Disorders
Since the brain and its various neurotransmitter systems are critical in regulating sleep and wakefulness, patients with neurologic disorders have an increased risk of developing sleep disorders. Patients with dementia, other neurodegenerative disorders, epilepsy, and traumatic brain injury (TBI) have a higher prevalence of sleep disturbance and sleep disorders.47 Poststroke patients can develop insomnia or hypersomnia, a reduction in sleep latency, increased sleep, or excessive daytime sleepiness, and are at higher risk for OSA during the first several months after a stroke.48 Specific neurologic lesions may lead to uncommon problems such as inversion of the sleep‐wake cycle, parasomnias, and hallucinatory dream‐like states.
Both Parkinson's disease and Alzheimer's disease are associated with multiple sleep disturbances, which tend to worsen with disease progression.14 Common problems include increased sleep fragmentation and wakefulness, with increases of stage 1 sleep and reductions of SWS and REM. Patients with neurodegenerative disorders also have an increased risk of REM sleep behavior disorder, or RBD.49 RBD is characterized by vivid and unusual dreams, and physically vigorous sleep behaviors that may result in ecchymoses, lacerations, and fractures.50 Fifty percent of patients with TBI reported insomnia symptoms.51 Disorders in initiating and maintaining sleep were the most common complaints among hospitalized patients with TBI. Some patients with TBI may develop circadian rhythm disturbances.52
Pain
A majority of patients with chronic pain, 50% to 70%, complain of impaired sleep.53 Sleep disruption is so common in fibromyalgia (75%) that it is considered to be a key diagnostic symptom.54 In a study investigating the affect of pain on sleep in burn patients, pain was associated with increased intermittent awakenings and prolonged periods of wake time during the night.55 The following day, these patients had poorer pain tolerance and greater pain intensity. Pain causes sleep fragmentation by increasing cortical arousals. Recent evidence suggests that sleep deprivation can increase pain sensitivity by inhibiting opioid protein synthesis or reducing opioid receptor affinity.56
Psychiatric Disorders
Sleep problems are so common in psychiatric conditions that the Diagnostic and Statistical Manual of Mental Disorders (DMS‐IV‐TR) includes sleep disturbance as a diagnostic criterion for a manic episode, and for various depressive, anxiety, and substance abuse disorders.57 The presence of sleep disturbance in hospitalized patients may suggest the presence of an underlying psychiatric disorder that would otherwise go unrecognized. In a survey of 200 general medical patients in a Brazilian hospital, Rocha et al.58 found that 112 (56.5%) complained of insomnia, and 100 (50%) met criteria for at least 1 psychiatric disorder. However, only 3 out of the total number of 200 surveyed (1.5%) were identified as having psychiatric diagnoses in the medical record, and sleep history was not noted in the clinical evaluation. An episode of major depressive disorder was the most common psychiatric diagnosis (35%). In this study, hospitalized patients with insomnia had a 3.6 times higher risk of having major depressive disorder than inpatients without insomnia.
Insomnia has a profound effect on mental health by worsening health‐related quality of life. In a study of outpatients at family medicine, internal medicine, endocrinology, cardiology, and psychiatry clinics in 3 U.S. cities (n = 3,445), insomnia worsened health‐related quality of life nearly as much as CHF or major depressive disorder did.59 Another survey of outpatients found that those with chronic insomnia were nearly 40 times more likely to have major depression and 6 times more likely to have an anxiety disorder compared to those without insomnia.60 Longitudinal studies have found that prior insomnia was associated with 2‐ to 5‐fold increase in the odds of mood and anxiety disorders and suicide.61, 62 Examining prodromes and precursors to mental disorders, Eaton et al.63 found that 47% of those with onset of depression at the 1‐year follow‐up had sleep problems at baseline.
An estimated 65% of patients with major depression have difficulty falling asleep, frequent awakenings, or early morning awakenings.64 Three patterns of sleep architecture abnormalities have been observed in patients with major depression: 1) sleep continuity disturbances characterized by prolonged sleep‐onset, increased wake time during sleep, increased early morning wake time, and decreased TST; 2) decreased proportion and length of SWS; and 3) REM sleep abnormalities such as reduced time to REM sleep, prolonged first REM sleep episode, and increased REM sleep percentage.65 Sleep during a manic episode has been less studied than in depression, but the data suggest that abnormal sleep in mania includes disrupted sleep continuity, shortened REM latency, and increased REM density (REM eye movement activity/total REM sleep time).65
Substance use disorders are also associated with sleep problems. In a survey by Brower et al.66 of patients who were undergoing alcohol rehabilitation, 61% (n = 172) had symptoms of insomnia such as increased sleep latency during the 6 months prior to entering treatment. Approximately 45% of these patients reported using alcohol for the purpose of initiating sleep. Alcohol and illicit substance intoxication and withdrawal are known to be associated with disrupted sleep. However, sleep disturbances may persist long after withdrawal symptoms have abated. Drummond et al. found that some patients continued to have alcohol‐associated sleep problems even after 27 months of abstinence.67 Evidence also suggests that untreated insomnia and other sleep problems may increase the risk of developing substance abuse problems due to self‐medicating with alcohol and other substances to help with sleep.68
Drugs that Affect Sleep
Numerous drugs can alter sleep quantity and quality. Sedatives and opioids may initially help with sleep onset, but impair sleep architecture. Medications used to treat medical and psychiatric illnesses also disrupt sleep (Table 3). The most common agents that impair sleep include antiepileptic drugs, selective serotonin reuptake inhibitors, monoamine oxidase inhibitors, tricyclic antidepressants, antihypertensives, antihistamines, and corticosteroids.
| Drug Class | Examples of Drugs | Affect on Sleep Architecture | Potential Mechanism | Clinical Implications |
|---|---|---|---|---|
| ||||
| CNS | ||||
| AEDs | Phenobarbital, carbamazepine, phenytoin | Very sedating. AEDs tend to TST, sleep latency | Inhibit neuronal calcium influx, adenosine, or 5HT activity | Sedation is dose‐dependent, and tends to occur with acute use |
| TCAs | Amoxapine, amitriptyline, imipramine, nortriptyline, desipramine, doxepin, clomipramine | Very sedating. Suppresses REM sleep, TST, stage‐2 sleep | Stimulate antimuscarinic‐receptor and alpha1‐receptor | Suppressed REM sleep motor inhibition restlessness, psychomotor agitation during sleep subjectively sleep quality, daytime sleepiness |
| BzRAs | Alprazolam, lorazepam, chlordiazepoxide, diazepam, oxazepam | Very sedating. TST, sleep latency, SWS duration, REM, stage‐2 sleep | Stimulate GABA type A receptor | Minimize daytime use. Chronic BzRAs SWS long‐term sequelae unknown |
| MAOIs | Phenylzine, tranylcypromine | Very sedating. TST, REM, REM rebound if stop MAOIs | Mechanism unknown | Daytime sleepiness; dosing time does not affect daytime somnolence |
| SSRIs | Sedating: paroxetine, fluvoxamine. Activating: fluoxetine, sertraline, citalopram | TST, are less sedating than TCAs and MAOIs. May REM, TWT, TST, SE | 5HT activity | Some patients get the opposite reaction |
| SNRI | Venlafaxine, duloxetine | Activating in some patients; sedating in 12% to 31%. TST | 5HT and NE activity | If activating, switch to AM dosing. If sedating, switch to PM dosing |
| Mood stabilizer | Lithium | Sedating. TST, SWS, REM, REM latency | daytime sedation. Dose at night | |
| Stimulants | Ephedrine, pseudoephedrine, modafinil | Activating. TST, SWS, sleep latency | DOPA, NE, and 5HT activity | Avoid after 6 PM |
| Anti‐Parkinson | Bromocriptine, levodopa | Sedating. Nightmares, SWS | DOPA | Dose at night, if possible |
| Cardiac | ||||
| Lipophilic beta‐blockers | Propranolol, pindolol, metoprolol, timolol. Hydrophilic agents (atenolol and sotalol) lack these effects | Activating. awakenings, TWT, REM, nightmares | CNS beta‐blockade | Lipophilic beta‐blockers daytime sleep when dosed in AM |
| CNS agents | Norepinephrine, epinephrine | Activating. REM, SWS | Stimulate alpha1‐receptor | Minimize use at night |
| Dopamine | Activating. REM, SWS | Stimulate dopamine2‐receptor and alpha1‐receptor | Minimize use at night | |
| Ca++ channel blockers | Amlodipine, verapamil, nifedipine | Exacerbate underlying medical condition | Lower esophageal sphincter tone nocturnal GER sleep disturbance | |
| Alpha2‐receptor agonist | Clonidine | Stage 1, REM, nightmares | Stimulate alpha2‐receptor | Alpha2‐agonists daytime sleep and sleepiness directly. Dose at night |
| Alpha1‐receptor blockers | Doxazosin, prazosin, terazosin | Inhibit alpha1‐receptor | Alpha1‐receptor blockers daytime sleepiness | |
| Diuretics | HCTZ, furosemide | Sedating. | PM diuresis frequent awakenings | |
| Other | ||||
| Opioids | Codeine, morphine | Sedating. SWS, REM | Stimulate mu‐receptor | Minimize use at night |
| NSAIDs | Ibuprofen, indomethcin, celecoxib | TST, SE | Inhibit prostaglandin synthesis | Minimize use at night |
| Methylxanthine | Theophylline | Activating. stage 1, REM | Causes less restful sleep | |
| Antihistamines | Diphenhydramine, promethazine | Sedating | H1 receptor blockade | Minimize use at night |
| Corticosteroids | Dexamethasone, prednisone | Activating. REM, SWS, nightmares | Melatonin secretion | Can disrupt sleep, anxiety, induce mania or psychosis |
| H2 blockers | Cimetidine, ranitidine, famotidine | Sedating. TST | H2 receptor blockade | Sedating if >60 years old, renal impairment |
| Quinolone | Ciprofloxacin, sparfloxacin, ofloxacin, grepafloxacin, levofloxacin | Activating | Stimulate GABA type A receptor | Consider sleep agent after maximizing sleep hygiene. Linezolid rarely causes sleep disturbances |
Lipophilic beta antagonists such as propranolol and timolol can increase total wake time, decrease REM sleep, and increase the incidence of nightmares and insomnia.69 Anabolic steroids and beta‐agonist bronchodilator therapy can cause severe anxiety, sleeplessness, and even psychosis. Vasopressor agents such as dopamine can cause cortical activation, leading to increased arousal and reduced SWS.
Hospital Environment
Environmental noise and patient care activities often interfere with sleep in the hospital. They account for about 30% of patient awakenings in ICU patients.70 Noise levels in the ICU have average sound peaks of 150 to 200 dB, and evening peaks >80 dB between midnight and 6 AM.71 By comparison, the front row seats at a rock concert have sound levels of 110 dB. The high noise level in hospitals has long been implicated as a sleep disruptor,72 but studies in the past decade have found that patient care activities probably contribute more to awakenings than does environmental noise.73 An analysis of critical care nursing routines found that activities such as taking vital signs and giving baths occurred a mean 42.6 times a night per patient.74 Tamburri et al.74 found that patients experienced 2 to 3 hours without interruption on only 6% of the 147 nights studied. Routine daily baths were provided on 55 of the 147 study nights between 2 AM and 5 AM, which is unlikely to be an opportune time for most patients.
CONCLUSION
Hospitalization often prevents patients from achieving adequate sleep and can affect recovery from illness. Understanding the major factors that impair sleep during hospitalization allows clinicians to systemically evaluate and treat sleep problems. More than just prescribing sedative/hypnotic agents, the treatment for sleep disruption includes addressing multiple medical, behavioral, and environmental factors, which will be discussed in Part 2 of this article.
Hospitalized patients often have difficulty initiating and maintaining sleep, or complain of early awakening and nonrestorative sleep.1 The etiology of sleep disruption is multifactorial and includes the patient's underlying illness(es), medical treatments, and the hospital environment. Often unrecognized and untreated during hospitalization, sleep disruption may lead to sleep deprivation, or a chronic lack of restorative sleep.
Even in healthy individuals, sleep deprivation can result in numerous physical and psychological consequences. Sleep deprivation is associated with hypertension,2, 3 impaired postural control,4 decreased ventilatory drive,5 increased sympathetic cardiovascular activation,6 blunted hypothalamic‐pituitary‐adrenal axis,7 impaired host defenses, and possibly diabetes mellitus and obesity.810 The lack of restorative sleep increases the risk of developing anxiety and mood disorders and delirium, especially in acutely ill older patients.11 In the presence of acute physical infirmity, inadequate sleep may further compound illness and impair recovery. We provide an overview of normal sleep architecture and discuss factors that lead to sleep disruption in hospitalized medical patients.
NORMAL SLEEP ARCHITECTURE AND REGULATION
Normal sleep architecture refers to a characteristic pattern of sleep, and consists of two major stages: nonrapid eye movement (NREM, pronounced non‐rem) and rapid eye movement (REM). (For a table of pertinent abbreviations and terms, see Table 1) Sleep is quantified by polysomnography (PSG), which includes an electroencephalogram (EEG), electromyogram (EMG), and electrooculogram (EOG). A PSG also includes an electrocardiogram (ECG), and measures of airflow, oxygen saturation, and body position. NREM sleep comprises 75% to 80% of total sleep time (TST), and is characterized by relatively quiescent brain activity and decreased metabolic rate.12 NREM sleep consists of four stages (S1‐S4), with each stage leading to a progressively deeper sleep (Figure 1). REM sleep follows slow wave sleep (SWS), or deep sleep, and increases over the night, comprising 20% to 25% of TST. REM sleep is characterized by an activated EEG pattern, muscle atonia, and episodic bursts of rapid eye movements.

| Acronym | Term |
|---|---|
| BiPAP | Bilevel positive airway pressure |
| CHF | Congestive heart failure |
| CPAP | Continuous positive airway pressure |
| COPD | Chronic obstructive pulmonary disease |
| EEG | Electroencephalogram |
| EOG | Electroculogram |
| EMG | Electromyogram |
| ESRD | End‐stage renal disease |
| NPPV | Noninvasive positive pressure ventilation |
| NREM | Nonrapid eye movement |
| OSA | Obstructive sleep apnea |
| PLMD | Periodic limb movement disorder |
| PSG | Polysomnography |
| RBD | REM sleep behavior disorder |
| REM | Rapid eye movement |
| RLS | Restless leg syndrome |
| S1‐S4 | 4 Stages of sleep in NREM |
| SE | Sleep efficiency; TST divided by total time in bed |
| SWS | Slow wave sleep |
| TBI | Traumatic brain injury |
| TST | Total sleep time |
Normal sleep provides a period of physiologic and mental rest. During sleep, sympathetic tone decreases and parasympathetic tone increases, leading to a reduction in heart rate, arterial blood pressure, and cardiac output.13 Deep sleep is theorized to be necessary for physiologic restoration. REM sleep is associated with dreaming, and is essential for maintaining emotional and cognitive well‐being. Sleep architecture undergoes characteristic changes as people age.14 The duration of SWS peaks in childhood and decreases with age. Consequently, people >60 years old tend to have lower arousal thresholds and to have more frequent awakenings. The results of the Sleep Heart Health Study found that increased age was associated with decreased percentage of REM sleep, worse sleep efficiency (SE, which is TST divided by total time in bed), and lower arousal thresholds.14 With the reduction of SE, older people need to spend more hours in bed to achieve the same amount of restorative sleep as when they were younger. Although sleep tends to become more disrupted as people age, insomnia should not be considered a normal part of aging, and needs to be addressed clinically.15 The results of a National Sleep Foundation telephone survey of subjects between the ages of 55 and 84 years old (n = 1,506) suggested that sleep complaints in older adults are frequently secondary to comorbid medical conditions.16
Multiple anatomic structures, pathways, and neurotransmitter systems are involved in controlling wakefulness and sleep. Neurotransmitters that promote wakefulness include acetylcholine, histamine, noradrenaline (norepinephrine), serotonin, dopamine, and hypocretin (orexin). Sleep‐promoting neurotransmitters include gamma aminobutyric acid (GABA), adenosine, and melatonin. Specific stages of sleep are regulated by the turning on and off of various neurons. REM on cells use GABA, acetylcholine, and glutamine, whereas REM off cells use norepinephrine and serotonin. SWS is promoted by GABA and serotonin.17
Sleep regulation is a balance between a homeostatic sleep need and an intrinsic body clock, or circadian pacemaker. Located in the suprachiasmic nucleus, the circadian pacemaker determines the onset and termination of sleep, and is partially regulated by environmental cues such as light and ambient temperature. Melatonin, a physiologic sleep promoter, is inhibited by ambient light, and its circulation is decreased during daylight hours. The adrenal secretion of cortisol, which is associated with wakefulness, follows a circadian pattern. Regulated by the hypothalamic‐pituitary axis, cortisol levels peak in the early morning hours in preparation for the increased metabolic demands during wakefulness.
SLEEP PROBLEMS IN HOSPITALIZED PATIENTS
Insomnia, which is characterized by difficulty initiating or maintaining sleep, is the most common sleep disorder in the United States. About one‐third of the adult population in the United States experiences insomnia at some point in their lives,18 and it is a persistent problem in approximately 10% of U.S. adults.19 Insomnia can be exacerbated during hospitalization.
Studies investigating sleep in hospitalized patients using PSG have been limited primarily to the setting of the intensive care unit (ICU). Critically ill patients, particularly those requiring mechanical ventilation, are prone to sleep disturbances and an associated delirium.2022 Critically ill patients have fragmented sleep, with decreased SE and SWS, and increased S1 and S2.23 Physician awareness of the impact of sleep disturbance in hospitalized patients is vital. Surveys reveal that approximately one‐half of patients admitted on general medical wards complain of sleep disruption.24, 25 Meissner et al.25 examined the prevalence of sleep complaints and physician recognition of these complaints among general medical patients admitted to a Veterans Affairs tertiary care center. Results showed that 47% (n = 222) of patients had complaints of either insomnia and/or excessive daytime sleepiness.
FACTORS AFFECTING SLEEP DURING HOSPITALIZATION
Many medical and neurologic illnesses, psychiatric disorders, pain, medication therapy and the hospital environment may impair sleep, and hinder recovery from illness.
General Medical Disorders
Primary sleep disorders, such as obstructive sleep apnea (OSA) and numerous other medical illnesses, can directly impair sleep physiology, leading to a cyclical interaction (Figure 2). Other conditions that disrupt sleep include congestive heart failure (CHF), diabetes mellitus, chronic obstructive pulmonary disease (COPD), gastroesophageal reflux, cardiovascular disease, thyroid disorders, renal disease, and severe liver disease.26 Table 2 lists selected medical and neurological conditions, their associated sleep‐related problems, and suggestions on how to ameliorate these problems.

| Disease | Problem | Clinical Implications and Strategies to Improve Sleep |
|---|---|---|
| ||
| Asthma | Nocturnal exacerbation, nocturnal GER | Inhaled corticosteroids and/or long‐acting inhaled beta‐adrenergic agents |
| CHF | Orthopnea, paroxysmal nocturnal dyspnea, sleep‐disordered breathing, increased sympathetic tone, nighttime diuresis, Cheyne‐Stokes respiration | Keep the head of bed elevated 30 degrees. Nocturnal O2 to keep O2 saturation >88%. Daytime diuresis. Optimize cardiac function to treat Cheyne‐Stokes respiration. Consider CPAP for CHF |
| COPD | Persistent nocturnal hypoxemia with complications (e.g., cor pulmonale, polycythemia) | O2 for COPD and persistent hypoxemia (PaO2 55‐60 mm Hg) |
| Sporadic nighttime desaturations | PaO2 55 mm Hg monitor O2 saturation by pulse oximetry. If patient desaturates to 88% at night consistently, start nocturnal O2. For hypercapnia, adjust O2 to maintain O2 saturation at 88% to 90% | |
| Early‐morning airflow obstruction | Consider bedtime tiotropium and inhaled long‐acting beta‐adrenergic agonist agents | |
| Inhibition of respiratory muscles in REM | Avoid sedative‐hypnotics that cause respiratory depression | |
| Decreased functional residual capacity from recumbent position during sleep | Minimize recumbancy by keeping the head of bed up at 30 degrees | |
| End‐stage renal disease | Pruritus, nausea; increased risk of RLS and PLMD | Ambulation may help with RLS. Consider ropinirole and pramipexole. Correct hyperphosphatemia and uremia. Consider antipruritic and antiemetic agents |
| Nocturnal GER | Nocturnal GER decreased sleep, heartburn, coughing, asthma | Avoid eating or drinking 2 hours before bedtime, especially those that delay gastric emptying, increase acid secretion, or decrease lower esophageal sphincter pressure; e.g., high‐fat foods, ethanol, chocolate, peppers, peppermint. Keep head of bed 30 degrees. Minimize medications that could worsen nocturnal GER; e.g., theophylline, calcium channel blockers, prostaglandins, bisphosphonates |
| OSA | Snoring with upper airway obstruction | No ethanol 2 hours before bedtime. Minimize CNS depressants. Avoid supine position. Consider CPAP, oral mandibular advancement device, and/or surgical correction. Long‐term plan should include weight loss |
| Stroke | Focal neurologic deficits (e.g., dysphagia, weakness or paralysis) | Keep head of bed 30 degrees. Regularly suction secretions. Post‐stroke patients have an increased risk of hypersomnia, insomnia, and/or OSA |
Affecting approximately 24% of men and 9% of women in the United States, OSA is the most common primary sleep disorder,27, 28 and causes significant mental and physical morbidity. Risk factors for OSA include obesity, hypothyroidism‐induced muscle weakness, and structural abnormalities in the oropharynx region such as acromegaly, micrognathia, or retrognathia. OSA is characterized by episodes of complete or partial pharyngeal obstruction during sleep that cause snoring, apneic episodes, choking, dyspnea, and restlessness.28 These episodes are associated with intermittent nocturnal sympathetic activation leading to nocturnal awakenings and cortical arousals, all of which lead to daytime symptoms of fatigue, sleepiness, and cognitive impairment (Figure 2). In addition, chronic sympathetic activation causes numerous derangements in the vascular endothelium and platelet activation.29, 30 Sleep‐disordered breathing has been independently associated with cardiovascular diseases such as hypertension, CHF, ischemic heart disease, atrial fibrillation, and cerebrovascular disease.31, 32
OSA is also associated with sleep‐related gastroesophageal reflux, which is characterized by pain and nocturnal cough, and can induce nocturnal asthma attacks and laryngospasm.33 Green et al.29 found that OSA patients treated with continuous positive airway pressure (CPAP) had a 48% improvement in nocturnal reflux symptoms. Although the pathophysiology connecting OSA to the renal system is unknown, OSA has been found in up to 60% of patients with end‐stage renal disease and chronic renal failure.34
Patients with pulmonary disorders can be profoundly affected by the normal physiologic changes during sleep, particularly in REM sleep. During REM sleep, all respiratory muscles except the diaphragm become paralyzed. Thus, episodes of marked oxygen desaturation can occur in patients who rely on their accessory muscles for respiration. COPD patients have decreased TST, SWS, and REM sleep. Shortness of breath, nocturnal cough, and wheezing worsen sleep.35 The resulting fatigue and sleep deprivation negatively impact the work of breathing and impair gas exchange. Airflow obstruction tends to worsen in the early morning hours in patients with COPD and asthma, and may be related to the effect of REM on the accessory muscles for respiration. Although used to target CO2 retention, investigations using bilevel positive airway pressure ventilators (BiPAP) for improving sleep in COPD patients have been limited. Noninvasive positive pressure ventilation (NPPV) appears to acutely improve SE and TST in patients with hypercapnic COPD without significantly improving gas exchange. Other sleep parameters such as sleep architecture and the number of arousals during the night, remain unchanged during NPPV.36
CPAP has several side effects that could worsen sleep, which may explain its poor adherence rate among ambulatory patients.37 Side effects include nasal bridge discomfort, nasal congestion, swallowing air, dry nose, dry or red eyes, noise, ear pain, and rhinitis.38 During hospitalization, efforts should be made to improve patient comfort by resizing ill‐fitting masks, adding heated humidification or nasal steroids to alleviate nasal congestion, or adding a chin strap to reduce air leak and ingestion of air.
Endocrine disorders have also been associated with sleep disruption. Studies suggest that patients with diabetes mellitus have decreased TST and impaired sleep quality due to nocturia and neuropathic pain.39 Inadequate sleep may also affect glucose control. Inadequate quality or quantity of sleep has been shown to be a risk factor for developing Type 2 diabetes mellitus in large prospective studies.40 Sleep duration and quality were significant predictors of increased levels of glycosylated hemoglobin (HbA1c) in patients with Type 2 diabetes mellitus. Thyroid diseases often coexist with diabetes mellitus. Both hypo‐ and hyperthyroidism have been associated with sleep disruption. Hypothyroidism is associated with daytime somnolence and fatigue. Patients with hypothyroidism tend to have reduced SWS. Hyperthyroid patients often complain of insomnia, which has been attributed to a hypermetabolic state.
Approximately 50% of patients with chronic end‐stage renal disease (ESRD) have insomnia and other sleep disorders.41 Patients often complain of restless leg syndrome (RLS), periodic limb movement disorder (PLMD), bone pain, nausea, and pruritus. The etiology of sleep disorders appears to be related to metabolic derangements associated with ESRD or from coexisting diabetes mellitus.
RLS and PLMD are distinct problems that affect sleep differently. RLS is characterized by an unpleasant crampy, creeping or crawling sensation in the lower extremities that is relieved by movement of the legs.42 RLS symptoms typically occur soon after going to bed, and therefore tend to disrupt sleep onset. The requisite bed rest during hospitalization can worsen RLS, further exacerbating sleep problems.43 Since RLS may partially be caused by disrupted iron metabolism, serum ferritin levels should be evaluated.44 Other conditions associated with RLS include pregnancy, rheumatoid arthritis, fibromyalgia, multiple sclerosis, ESRD, and Parkinson's disease. The differential diagnosis for RLS and PLMD includes neuroleptic‐induced akathisia, peripheral neuropathy, and positional or nocturnal leg cramps. PLMD occurs in about 80% of those with RLS, and is characterized by involuntary limb movements that occur every 20 to 40 seconds during NREM sleep. Unaware of these movements, patients often experience frequent arousals throughout the night, and complain of daytime somnolence and fatigue.42
A pilot study of 35 patients with minimal hepatic encephalopathy found that nearly 50% complained of sleep difficulties.45 Hypothesizing that a dysregulation of histaminergic neurotransmission in cirrhosis alters the sleep‐wake cycle, Spahr et al.46 found that 40% of their patients reported subjective improvement in sleep when administered 25 mg of hydroxyzine, compared to none who received placebo.
Neurologic Disorders
Since the brain and its various neurotransmitter systems are critical in regulating sleep and wakefulness, patients with neurologic disorders have an increased risk of developing sleep disorders. Patients with dementia, other neurodegenerative disorders, epilepsy, and traumatic brain injury (TBI) have a higher prevalence of sleep disturbance and sleep disorders.47 Poststroke patients can develop insomnia or hypersomnia, a reduction in sleep latency, increased sleep, or excessive daytime sleepiness, and are at higher risk for OSA during the first several months after a stroke.48 Specific neurologic lesions may lead to uncommon problems such as inversion of the sleep‐wake cycle, parasomnias, and hallucinatory dream‐like states.
Both Parkinson's disease and Alzheimer's disease are associated with multiple sleep disturbances, which tend to worsen with disease progression.14 Common problems include increased sleep fragmentation and wakefulness, with increases of stage 1 sleep and reductions of SWS and REM. Patients with neurodegenerative disorders also have an increased risk of REM sleep behavior disorder, or RBD.49 RBD is characterized by vivid and unusual dreams, and physically vigorous sleep behaviors that may result in ecchymoses, lacerations, and fractures.50 Fifty percent of patients with TBI reported insomnia symptoms.51 Disorders in initiating and maintaining sleep were the most common complaints among hospitalized patients with TBI. Some patients with TBI may develop circadian rhythm disturbances.52
Pain
A majority of patients with chronic pain, 50% to 70%, complain of impaired sleep.53 Sleep disruption is so common in fibromyalgia (75%) that it is considered to be a key diagnostic symptom.54 In a study investigating the affect of pain on sleep in burn patients, pain was associated with increased intermittent awakenings and prolonged periods of wake time during the night.55 The following day, these patients had poorer pain tolerance and greater pain intensity. Pain causes sleep fragmentation by increasing cortical arousals. Recent evidence suggests that sleep deprivation can increase pain sensitivity by inhibiting opioid protein synthesis or reducing opioid receptor affinity.56
Psychiatric Disorders
Sleep problems are so common in psychiatric conditions that the Diagnostic and Statistical Manual of Mental Disorders (DMS‐IV‐TR) includes sleep disturbance as a diagnostic criterion for a manic episode, and for various depressive, anxiety, and substance abuse disorders.57 The presence of sleep disturbance in hospitalized patients may suggest the presence of an underlying psychiatric disorder that would otherwise go unrecognized. In a survey of 200 general medical patients in a Brazilian hospital, Rocha et al.58 found that 112 (56.5%) complained of insomnia, and 100 (50%) met criteria for at least 1 psychiatric disorder. However, only 3 out of the total number of 200 surveyed (1.5%) were identified as having psychiatric diagnoses in the medical record, and sleep history was not noted in the clinical evaluation. An episode of major depressive disorder was the most common psychiatric diagnosis (35%). In this study, hospitalized patients with insomnia had a 3.6 times higher risk of having major depressive disorder than inpatients without insomnia.
Insomnia has a profound effect on mental health by worsening health‐related quality of life. In a study of outpatients at family medicine, internal medicine, endocrinology, cardiology, and psychiatry clinics in 3 U.S. cities (n = 3,445), insomnia worsened health‐related quality of life nearly as much as CHF or major depressive disorder did.59 Another survey of outpatients found that those with chronic insomnia were nearly 40 times more likely to have major depression and 6 times more likely to have an anxiety disorder compared to those without insomnia.60 Longitudinal studies have found that prior insomnia was associated with 2‐ to 5‐fold increase in the odds of mood and anxiety disorders and suicide.61, 62 Examining prodromes and precursors to mental disorders, Eaton et al.63 found that 47% of those with onset of depression at the 1‐year follow‐up had sleep problems at baseline.
An estimated 65% of patients with major depression have difficulty falling asleep, frequent awakenings, or early morning awakenings.64 Three patterns of sleep architecture abnormalities have been observed in patients with major depression: 1) sleep continuity disturbances characterized by prolonged sleep‐onset, increased wake time during sleep, increased early morning wake time, and decreased TST; 2) decreased proportion and length of SWS; and 3) REM sleep abnormalities such as reduced time to REM sleep, prolonged first REM sleep episode, and increased REM sleep percentage.65 Sleep during a manic episode has been less studied than in depression, but the data suggest that abnormal sleep in mania includes disrupted sleep continuity, shortened REM latency, and increased REM density (REM eye movement activity/total REM sleep time).65
Substance use disorders are also associated with sleep problems. In a survey by Brower et al.66 of patients who were undergoing alcohol rehabilitation, 61% (n = 172) had symptoms of insomnia such as increased sleep latency during the 6 months prior to entering treatment. Approximately 45% of these patients reported using alcohol for the purpose of initiating sleep. Alcohol and illicit substance intoxication and withdrawal are known to be associated with disrupted sleep. However, sleep disturbances may persist long after withdrawal symptoms have abated. Drummond et al. found that some patients continued to have alcohol‐associated sleep problems even after 27 months of abstinence.67 Evidence also suggests that untreated insomnia and other sleep problems may increase the risk of developing substance abuse problems due to self‐medicating with alcohol and other substances to help with sleep.68
Drugs that Affect Sleep
Numerous drugs can alter sleep quantity and quality. Sedatives and opioids may initially help with sleep onset, but impair sleep architecture. Medications used to treat medical and psychiatric illnesses also disrupt sleep (Table 3). The most common agents that impair sleep include antiepileptic drugs, selective serotonin reuptake inhibitors, monoamine oxidase inhibitors, tricyclic antidepressants, antihypertensives, antihistamines, and corticosteroids.
| Drug Class | Examples of Drugs | Affect on Sleep Architecture | Potential Mechanism | Clinical Implications |
|---|---|---|---|---|
| ||||
| CNS | ||||
| AEDs | Phenobarbital, carbamazepine, phenytoin | Very sedating. AEDs tend to TST, sleep latency | Inhibit neuronal calcium influx, adenosine, or 5HT activity | Sedation is dose‐dependent, and tends to occur with acute use |
| TCAs | Amoxapine, amitriptyline, imipramine, nortriptyline, desipramine, doxepin, clomipramine | Very sedating. Suppresses REM sleep, TST, stage‐2 sleep | Stimulate antimuscarinic‐receptor and alpha1‐receptor | Suppressed REM sleep motor inhibition restlessness, psychomotor agitation during sleep subjectively sleep quality, daytime sleepiness |
| BzRAs | Alprazolam, lorazepam, chlordiazepoxide, diazepam, oxazepam | Very sedating. TST, sleep latency, SWS duration, REM, stage‐2 sleep | Stimulate GABA type A receptor | Minimize daytime use. Chronic BzRAs SWS long‐term sequelae unknown |
| MAOIs | Phenylzine, tranylcypromine | Very sedating. TST, REM, REM rebound if stop MAOIs | Mechanism unknown | Daytime sleepiness; dosing time does not affect daytime somnolence |
| SSRIs | Sedating: paroxetine, fluvoxamine. Activating: fluoxetine, sertraline, citalopram | TST, are less sedating than TCAs and MAOIs. May REM, TWT, TST, SE | 5HT activity | Some patients get the opposite reaction |
| SNRI | Venlafaxine, duloxetine | Activating in some patients; sedating in 12% to 31%. TST | 5HT and NE activity | If activating, switch to AM dosing. If sedating, switch to PM dosing |
| Mood stabilizer | Lithium | Sedating. TST, SWS, REM, REM latency | daytime sedation. Dose at night | |
| Stimulants | Ephedrine, pseudoephedrine, modafinil | Activating. TST, SWS, sleep latency | DOPA, NE, and 5HT activity | Avoid after 6 PM |
| Anti‐Parkinson | Bromocriptine, levodopa | Sedating. Nightmares, SWS | DOPA | Dose at night, if possible |
| Cardiac | ||||
| Lipophilic beta‐blockers | Propranolol, pindolol, metoprolol, timolol. Hydrophilic agents (atenolol and sotalol) lack these effects | Activating. awakenings, TWT, REM, nightmares | CNS beta‐blockade | Lipophilic beta‐blockers daytime sleep when dosed in AM |
| CNS agents | Norepinephrine, epinephrine | Activating. REM, SWS | Stimulate alpha1‐receptor | Minimize use at night |
| Dopamine | Activating. REM, SWS | Stimulate dopamine2‐receptor and alpha1‐receptor | Minimize use at night | |
| Ca++ channel blockers | Amlodipine, verapamil, nifedipine | Exacerbate underlying medical condition | Lower esophageal sphincter tone nocturnal GER sleep disturbance | |
| Alpha2‐receptor agonist | Clonidine | Stage 1, REM, nightmares | Stimulate alpha2‐receptor | Alpha2‐agonists daytime sleep and sleepiness directly. Dose at night |
| Alpha1‐receptor blockers | Doxazosin, prazosin, terazosin | Inhibit alpha1‐receptor | Alpha1‐receptor blockers daytime sleepiness | |
| Diuretics | HCTZ, furosemide | Sedating. | PM diuresis frequent awakenings | |
| Other | ||||
| Opioids | Codeine, morphine | Sedating. SWS, REM | Stimulate mu‐receptor | Minimize use at night |
| NSAIDs | Ibuprofen, indomethcin, celecoxib | TST, SE | Inhibit prostaglandin synthesis | Minimize use at night |
| Methylxanthine | Theophylline | Activating. stage 1, REM | Causes less restful sleep | |
| Antihistamines | Diphenhydramine, promethazine | Sedating | H1 receptor blockade | Minimize use at night |
| Corticosteroids | Dexamethasone, prednisone | Activating. REM, SWS, nightmares | Melatonin secretion | Can disrupt sleep, anxiety, induce mania or psychosis |
| H2 blockers | Cimetidine, ranitidine, famotidine | Sedating. TST | H2 receptor blockade | Sedating if >60 years old, renal impairment |
| Quinolone | Ciprofloxacin, sparfloxacin, ofloxacin, grepafloxacin, levofloxacin | Activating | Stimulate GABA type A receptor | Consider sleep agent after maximizing sleep hygiene. Linezolid rarely causes sleep disturbances |
Lipophilic beta antagonists such as propranolol and timolol can increase total wake time, decrease REM sleep, and increase the incidence of nightmares and insomnia.69 Anabolic steroids and beta‐agonist bronchodilator therapy can cause severe anxiety, sleeplessness, and even psychosis. Vasopressor agents such as dopamine can cause cortical activation, leading to increased arousal and reduced SWS.
Hospital Environment
Environmental noise and patient care activities often interfere with sleep in the hospital. They account for about 30% of patient awakenings in ICU patients.70 Noise levels in the ICU have average sound peaks of 150 to 200 dB, and evening peaks >80 dB between midnight and 6 AM.71 By comparison, the front row seats at a rock concert have sound levels of 110 dB. The high noise level in hospitals has long been implicated as a sleep disruptor,72 but studies in the past decade have found that patient care activities probably contribute more to awakenings than does environmental noise.73 An analysis of critical care nursing routines found that activities such as taking vital signs and giving baths occurred a mean 42.6 times a night per patient.74 Tamburri et al.74 found that patients experienced 2 to 3 hours without interruption on only 6% of the 147 nights studied. Routine daily baths were provided on 55 of the 147 study nights between 2 AM and 5 AM, which is unlikely to be an opportune time for most patients.
CONCLUSION
Hospitalization often prevents patients from achieving adequate sleep and can affect recovery from illness. Understanding the major factors that impair sleep during hospitalization allows clinicians to systemically evaluate and treat sleep problems. More than just prescribing sedative/hypnotic agents, the treatment for sleep disruption includes addressing multiple medical, behavioral, and environmental factors, which will be discussed in Part 2 of this article.
- NIH State‐of‐the Science Conference Statement on Manifestations and Management of Chronic Insomnia in Adults.NIH Consens Sci Statements.2005;22(2):1–30.
- ,,, et al.Short sleep duration as a risk factor for hypertension: analyses of the first National Health and Nutrition Examination Survey.Hypertension.2006;47(5):833–839.
- ,,, et al.Total sleep deprivation elevates blood pressure through arterial baroreflex resetting: a study with microneurographic technique.Sleep.2003;26(8):986–989.
- ,,,,.Postural control after a night without sleep.Neuropsychologia.2006;44(12):2520–2525.
- .Sleep deprivation decreases ventilatory responses to CO2 but not load compensation.Chest.1983;84(6):695–698.
- ,,, et al.Increased sympathetic and decreased parasympathetic cardiovascular modulation in normal humans with acute sleep deprivation.J Appl Physiol.2005;98(6):2024–2032.
- .Reductions in circulating anabolic hormones induced by sustained sleep deprivation in rats.Am J Physiol Endocrinol Metab.2004;286(6):E1060–E1070.
- ,,,.The metabolic consequences of sleep deprivation.Sleep Med Rev.2007;11(3):163–178.
- ,,,,.Sleep deprivation and activation of morning levels of cellular and genomic markers of inflammation.Arch Int Med.2006;166:1756–1752.
- ,,,.Effects of sleep and sleep deprivation on immunoglobulins and complement in humans.Brain Behav Immun.2007;21:308–310.
- ,,,,.The effects of sleep deprivation on symptoms of psychopathology in healthy adults.Sleep Med.2007;8:215–221.
- ,.Normal human sleep. In:Kryger MH,Roth T,Dement WC, editors.Principles and Practice of Sleep Medicine.3rd ed.Philadelphia:W.B. Saunders;2000:15–16.
- .Update on the Science, Diagnosis and Management of Insomnia.International Congress and Symposium Series 262.London:Royal Society of Medicine Press Ltd;2006.
- ,,,,,.The effects of age, sex, ethnicity, and sleep‐disordered breathing on sleep architecture.Arch Intern Med.2004;164:406–18.
- ,.Sleep disruption in older adults.Am J Nurs.2007;107(5):40–49.
- ,,,.Sleep disturbances and chronic disease in older adults: results of the 2003 National Sleep Foundation Sleep in America Survey.J Psychosom Res.2004;56(5):497–502.
- .Sleep in patients with neurologic and psychiatric disorders.Prim Care.2005;32:535–548.
- ,,.Insomnia and its treatment: prevalence and correlates.Arch Gen Psychiatry.1985;42(3):225–232.
- ,.Epidemiologic study of sleep disturbances and psychiatric disorders: an opportunity for prevention?JAMA.1989;262:1479–1484.
- ,,,,,.Sleep in critically ill patients requiring mechanical ventilation.Chest.2000;117:809–818.
- ,.Sedative and analgesic medications: risk factors for delirium and sleep disturbances in the critically ill.Crit Care Med.2006;22:313–327.
- ,,,,.Quantity and quality of sleep in the surgical intensive care unit: are our patients sleeping?J Trauma.2007;63:1210–1214.
- .Sleep in acute care units.Sleep Breath.2006;10:6–15.
- ,,,,,.An assessment of quality of sleep and the use of drugs with sedating properties in hospitalized adult patients.Health Qual Life Outcomes.2004;2:17.
- ,,,,,.Failure of physician documentation of sleep complaints in hospitalized patients.West J Med.1998;169:146–149.
- .Sleep and medical disorders.Prim Care.2005;35:511–533.
- ,,,,,.The occurrence of sleep‐disordered breathing among middle‐aged adults.N Engl J Med.1993;328:1230–1235.
- ,.Clinical features and evaluation of obstructive sleep apnea‐hypopnea syndrome. In:Kryger MH,Roth T,Dement WC, editors.Principles and Practice of Sleep Medicine.3rd ed.Philadelphia:W.B. Saunders;2000:869–872.
- ,,.Marked improvement in nocturnal gastroesophageal reflux in a large cohort of patients with obstructive sleep apnea.Arch Intern Med.2003;163:41–45.
- ,.Sleep and medical disorders.Med Clin North Am.2004;88:679–703.
- ,,,.Relation of sleep‐disordered breathing to carotid plaque and intima‐media thickness.Atherosclerosis.2008;197(1):125– 131.
- ,,, et al.Sleep‐disordered breathing and cardiovascular disease: cross‐sectional results of the Sleep Heart Health Study.Am J Respir Crit Care Med.2001;163:19–25.
- .Gastroesophageal reflux during sleep.Sleep Med Clin.2007;2:41–50.
- ,.Sleep apnea in renal failure.Adv Perit Dial.1997;13:88–92.
- .Sleep in chronic obstructive pulmonary disease.Sleep Med Clin.2007;2:1–8.
- ,,,.Effects of non‐invasive positive pressure ventilation on gas exchange and sleep in COPD patients.Chest.1997;112:623–628.
- ,,, et al.Night‐to‐night variability in CPAP use over the first three months of treatment.Sleep.1997;20(4):278–283.
- ,.Improving CPAP use by patients with the sleep apnoea/hypopnoea syndrome (SAHS).Sleep Med Rev.2003;7(1):81–99.
- ,,.Factors predicting sleep disruption in type II diabetes.Sleep.2000;23:415–416.
- ,,.Sleep duration as a risk factor for the development of type 2 diabetes.Diabetes Care.2006;29:657–661.
- .Sleep disorders and end‐stage renal disease.Sleep Med Clin.2007;2:59–66.
- ,,,.Sleep and aging: 1. Sleep disorders commonly found in older people.CMAJ.2007;176(9):1299–1304.
- ,,,,,.Impact, diagnosis and treatment of restless legs syndrome (RLS) in a primary care population: The REST (RLS Epidemiology, Symptoms, and Treatment) primary care study.Sleep Med.2004;5(3):237–246.
- ,.Epidemiology and clinical findings of restless leg syndrome.Sleep Med.2004;5(3):293–299.
- ,,,,,.High prevalence of sleep disturbance in cirrhosis.Hepatology.1998;27:339–345.
- ,,,,.Histamine H1 blocker hydroxyzine improves sleep in patients with cirrhosis and minimal hepatic encephalopathy: a randomized controlled pilot trial.Am J Gastroenterol.2007;102:744–753.
- .Sleep in patients with neurologic and psychiatric disorders.Prim Care.2005;32:535–548.
- ,,.Obstructive sleep apnea: implications for cardiac and vascular disease.JAMA.2003;290(14):1906–1914.
- ,,, et al.Pathophysiology of REM sleep behaviour disorder and relevance to neurodegenerative disease.Brain.2007;130(Pt 11):2770–2788.
- ,.REM sleep parasomnias. In:Kryger MH,Roth T,Dement WC, editors.Principles and Practice of Sleep Medicine.3rd ed.Philadelphia:W.B. Saunders;2000:724–725.
- ,,.Insomnia in patients with traumatic brain injury.J Head Trauma Rehabil.2006;21(3):199–212.
- ,,,,.Circadian rhythm sleep disorders following mild traumatic brain injury.Neurology.2007;68(14):1136–1140.
- ,.Comorbidities: psychiatric, medical, medications, and substances.Sleep Med Clin.2006;231–245.
- ,,.Sleep disturbance and fibromyalgia.Sleep Med Clin.2007;2:31–39.
- ,,.Sleep disturbances, pain and analgesia in adults hospitalized for burn injuries.Sleep Med.2004;5:551–559.
- ,,.Sleep deprivation and pain perception.Sleep Med Rev.2006;10:357–369.
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders.4th ed. Text Revision.Washington, DC:American Psychiatric Association;2000.
- ,,, et al.Is insomnia a marker for psychiatric disorders in general hospitals?Sleep Med.2005;6:549–553.
- ,.The relationship between insomnia and health‐related quality of life in patients with chronic illness.J Fam Pract.2002;51(3):229–235.
- ,.Epidemiologic study of sleep disturbances and psychiatric disorders: an opportunity for prevention?JAMA.1989;262:1479–1484.
- ,,,.Sleep disturbance and psychiatric disorders: a longitudinal epidemiological study of young adults.Biol Psychiatry.1996;39:411–418.
- ,,,.The morbidity of insomnia uncomplicated by psychiatric disorders.Gen Hosp Psychiatry.1997;19:245–250.
- ,,.Prodromes and precursors: epidemiologic data for primary prevention of disorders with slow onset.Am J Psychiatry.1995;152:967–972.
- ,,,,,.Which depressive symptoms are related to which sleep electroencephalographic variables?Biol Psychol.1997;42:904–913.
- ,.Sleep in mood disorders.Psychiatr Clin North Am.2006;29:1009–1032.
- ,,,,.Insomnia, self‐medication, and relapse to alcoholism.Am J Psychiatry.2001;158:399–404.
- ,,,.The sleep of abstinent pure primary alcoholic patients: natural course and relationship to relapse.Alcohol Clin Exp Res.1998;22:1796–1802.
- ,,,,.Screening for substance use patterns among patients referred for a variety of sleep complaints.Am J Drug Alcohol Abuse.2006;32:111–120.
- .Drugs that disturb sleep and wakefulness. In:Kryger MH,Roth T,Dement WC, editors.Principles and Practice of Sleep Medicine.3rd ed.Philadelphia:W.B. Saunders;2000:441–462.
- ,,, et al.Contribution of the intensive care unit environment to sleep disruption in mechanically ventilated patients and healthy subjects.Am J Respir Crit Care Med.2003;167(5):708–715.
- ,,.Adverse environmental conditions in the respiratory and medical ICU settings.Chest.1994;105:1211–1216.
- ,,,,,.Noise levels in Johns Hopkins Hospital.J Acoust Soc Am.2005;118(6):3629–3645.
- ,,.Patient perception of sleep quality and etiology of sleep disruption in the intensive care unit.Am J Respir Crit Care Med.1999;159:1155–1162.
- ,,,.Nocturnal care interactions with patients in critical care units.Am J Crit Care.2004;13(2):102–115.
- NIH State‐of‐the Science Conference Statement on Manifestations and Management of Chronic Insomnia in Adults.NIH Consens Sci Statements.2005;22(2):1–30.
- ,,, et al.Short sleep duration as a risk factor for hypertension: analyses of the first National Health and Nutrition Examination Survey.Hypertension.2006;47(5):833–839.
- ,,, et al.Total sleep deprivation elevates blood pressure through arterial baroreflex resetting: a study with microneurographic technique.Sleep.2003;26(8):986–989.
- ,,,,.Postural control after a night without sleep.Neuropsychologia.2006;44(12):2520–2525.
- .Sleep deprivation decreases ventilatory responses to CO2 but not load compensation.Chest.1983;84(6):695–698.
- ,,, et al.Increased sympathetic and decreased parasympathetic cardiovascular modulation in normal humans with acute sleep deprivation.J Appl Physiol.2005;98(6):2024–2032.
- .Reductions in circulating anabolic hormones induced by sustained sleep deprivation in rats.Am J Physiol Endocrinol Metab.2004;286(6):E1060–E1070.
- ,,,.The metabolic consequences of sleep deprivation.Sleep Med Rev.2007;11(3):163–178.
- ,,,,.Sleep deprivation and activation of morning levels of cellular and genomic markers of inflammation.Arch Int Med.2006;166:1756–1752.
- ,,,.Effects of sleep and sleep deprivation on immunoglobulins and complement in humans.Brain Behav Immun.2007;21:308–310.
- ,,,,.The effects of sleep deprivation on symptoms of psychopathology in healthy adults.Sleep Med.2007;8:215–221.
- ,.Normal human sleep. In:Kryger MH,Roth T,Dement WC, editors.Principles and Practice of Sleep Medicine.3rd ed.Philadelphia:W.B. Saunders;2000:15–16.
- .Update on the Science, Diagnosis and Management of Insomnia.International Congress and Symposium Series 262.London:Royal Society of Medicine Press Ltd;2006.
- ,,,,,.The effects of age, sex, ethnicity, and sleep‐disordered breathing on sleep architecture.Arch Intern Med.2004;164:406–18.
- ,.Sleep disruption in older adults.Am J Nurs.2007;107(5):40–49.
- ,,,.Sleep disturbances and chronic disease in older adults: results of the 2003 National Sleep Foundation Sleep in America Survey.J Psychosom Res.2004;56(5):497–502.
- .Sleep in patients with neurologic and psychiatric disorders.Prim Care.2005;32:535–548.
- ,,.Insomnia and its treatment: prevalence and correlates.Arch Gen Psychiatry.1985;42(3):225–232.
- ,.Epidemiologic study of sleep disturbances and psychiatric disorders: an opportunity for prevention?JAMA.1989;262:1479–1484.
- ,,,,,.Sleep in critically ill patients requiring mechanical ventilation.Chest.2000;117:809–818.
- ,.Sedative and analgesic medications: risk factors for delirium and sleep disturbances in the critically ill.Crit Care Med.2006;22:313–327.
- ,,,,.Quantity and quality of sleep in the surgical intensive care unit: are our patients sleeping?J Trauma.2007;63:1210–1214.
- .Sleep in acute care units.Sleep Breath.2006;10:6–15.
- ,,,,,.An assessment of quality of sleep and the use of drugs with sedating properties in hospitalized adult patients.Health Qual Life Outcomes.2004;2:17.
- ,,,,,.Failure of physician documentation of sleep complaints in hospitalized patients.West J Med.1998;169:146–149.
- .Sleep and medical disorders.Prim Care.2005;35:511–533.
- ,,,,,.The occurrence of sleep‐disordered breathing among middle‐aged adults.N Engl J Med.1993;328:1230–1235.
- ,.Clinical features and evaluation of obstructive sleep apnea‐hypopnea syndrome. In:Kryger MH,Roth T,Dement WC, editors.Principles and Practice of Sleep Medicine.3rd ed.Philadelphia:W.B. Saunders;2000:869–872.
- ,,.Marked improvement in nocturnal gastroesophageal reflux in a large cohort of patients with obstructive sleep apnea.Arch Intern Med.2003;163:41–45.
- ,.Sleep and medical disorders.Med Clin North Am.2004;88:679–703.
- ,,,.Relation of sleep‐disordered breathing to carotid plaque and intima‐media thickness.Atherosclerosis.2008;197(1):125– 131.
- ,,, et al.Sleep‐disordered breathing and cardiovascular disease: cross‐sectional results of the Sleep Heart Health Study.Am J Respir Crit Care Med.2001;163:19–25.
- .Gastroesophageal reflux during sleep.Sleep Med Clin.2007;2:41–50.
- ,.Sleep apnea in renal failure.Adv Perit Dial.1997;13:88–92.
- .Sleep in chronic obstructive pulmonary disease.Sleep Med Clin.2007;2:1–8.
- ,,,.Effects of non‐invasive positive pressure ventilation on gas exchange and sleep in COPD patients.Chest.1997;112:623–628.
- ,,, et al.Night‐to‐night variability in CPAP use over the first three months of treatment.Sleep.1997;20(4):278–283.
- ,.Improving CPAP use by patients with the sleep apnoea/hypopnoea syndrome (SAHS).Sleep Med Rev.2003;7(1):81–99.
- ,,.Factors predicting sleep disruption in type II diabetes.Sleep.2000;23:415–416.
- ,,.Sleep duration as a risk factor for the development of type 2 diabetes.Diabetes Care.2006;29:657–661.
- .Sleep disorders and end‐stage renal disease.Sleep Med Clin.2007;2:59–66.
- ,,,.Sleep and aging: 1. Sleep disorders commonly found in older people.CMAJ.2007;176(9):1299–1304.
- ,,,,,.Impact, diagnosis and treatment of restless legs syndrome (RLS) in a primary care population: The REST (RLS Epidemiology, Symptoms, and Treatment) primary care study.Sleep Med.2004;5(3):237–246.
- ,.Epidemiology and clinical findings of restless leg syndrome.Sleep Med.2004;5(3):293–299.
- ,,,,,.High prevalence of sleep disturbance in cirrhosis.Hepatology.1998;27:339–345.
- ,,,,.Histamine H1 blocker hydroxyzine improves sleep in patients with cirrhosis and minimal hepatic encephalopathy: a randomized controlled pilot trial.Am J Gastroenterol.2007;102:744–753.
- .Sleep in patients with neurologic and psychiatric disorders.Prim Care.2005;32:535–548.
- ,,.Obstructive sleep apnea: implications for cardiac and vascular disease.JAMA.2003;290(14):1906–1914.
- ,,, et al.Pathophysiology of REM sleep behaviour disorder and relevance to neurodegenerative disease.Brain.2007;130(Pt 11):2770–2788.
- ,.REM sleep parasomnias. In:Kryger MH,Roth T,Dement WC, editors.Principles and Practice of Sleep Medicine.3rd ed.Philadelphia:W.B. Saunders;2000:724–725.
- ,,.Insomnia in patients with traumatic brain injury.J Head Trauma Rehabil.2006;21(3):199–212.
- ,,,,.Circadian rhythm sleep disorders following mild traumatic brain injury.Neurology.2007;68(14):1136–1140.
- ,.Comorbidities: psychiatric, medical, medications, and substances.Sleep Med Clin.2006;231–245.
- ,,.Sleep disturbance and fibromyalgia.Sleep Med Clin.2007;2:31–39.
- ,,.Sleep disturbances, pain and analgesia in adults hospitalized for burn injuries.Sleep Med.2004;5:551–559.
- ,,.Sleep deprivation and pain perception.Sleep Med Rev.2006;10:357–369.
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders.4th ed. Text Revision.Washington, DC:American Psychiatric Association;2000.
- ,,, et al.Is insomnia a marker for psychiatric disorders in general hospitals?Sleep Med.2005;6:549–553.
- ,.The relationship between insomnia and health‐related quality of life in patients with chronic illness.J Fam Pract.2002;51(3):229–235.
- ,.Epidemiologic study of sleep disturbances and psychiatric disorders: an opportunity for prevention?JAMA.1989;262:1479–1484.
- ,,,.Sleep disturbance and psychiatric disorders: a longitudinal epidemiological study of young adults.Biol Psychiatry.1996;39:411–418.
- ,,,.The morbidity of insomnia uncomplicated by psychiatric disorders.Gen Hosp Psychiatry.1997;19:245–250.
- ,,.Prodromes and precursors: epidemiologic data for primary prevention of disorders with slow onset.Am J Psychiatry.1995;152:967–972.
- ,,,,,.Which depressive symptoms are related to which sleep electroencephalographic variables?Biol Psychol.1997;42:904–913.
- ,.Sleep in mood disorders.Psychiatr Clin North Am.2006;29:1009–1032.
- ,,,,.Insomnia, self‐medication, and relapse to alcoholism.Am J Psychiatry.2001;158:399–404.
- ,,,.The sleep of abstinent pure primary alcoholic patients: natural course and relationship to relapse.Alcohol Clin Exp Res.1998;22:1796–1802.
- ,,,,.Screening for substance use patterns among patients referred for a variety of sleep complaints.Am J Drug Alcohol Abuse.2006;32:111–120.
- .Drugs that disturb sleep and wakefulness. In:Kryger MH,Roth T,Dement WC, editors.Principles and Practice of Sleep Medicine.3rd ed.Philadelphia:W.B. Saunders;2000:441–462.
- ,,, et al.Contribution of the intensive care unit environment to sleep disruption in mechanically ventilated patients and healthy subjects.Am J Respir Crit Care Med.2003;167(5):708–715.
- ,,.Adverse environmental conditions in the respiratory and medical ICU settings.Chest.1994;105:1211–1216.
- ,,,,,.Noise levels in Johns Hopkins Hospital.J Acoust Soc Am.2005;118(6):3629–3645.
- ,,.Patient perception of sleep quality and etiology of sleep disruption in the intensive care unit.Am J Respir Crit Care Med.1999;159:1155–1162.
- ,,,.Nocturnal care interactions with patients in critical care units.Am J Crit Care.2004;13(2):102–115.
Did antismoking therapy make him sick?
Presentation: unconscious on the street
Emergency medical personnel bring Mr. M, age 66, to the ER after passers-by find him supine on the sidewalk. On arrival, he is comatose as confirmed by a Glasgow Coma Scale score of 8 (eye opening 3, verbal response 2, motor response 3). Systolic blood pressure is 108 mm Hg on palpation, pulse is 135 beats per minute, and temperature is 105 °F. Minor abrasions cover his face and arms, and his hands and feet are rigid.
Mr. M has lived at a board-and-care facility for 30 years. The facility’s operator tells us that Mr. M has had schizophrenia for 40 years and has been taking:
- olanzapine, 7.5 mg each morning and 10 mg at bedtime
- chlorpromazine, 50 mg nightly
- lithium carbonate, 300 mg tid
- and benztropine, 2 mg bid.
Three weeks ago, Mr. M was hospitalized for 6 days with pneumonia. In 3 months, he will undergo surgery for prostate cancer. He is taking no medication for the prostate cancer.
Creatine phosphokinase (CPK) is 2,939 IU/L, indicating neuroleptic malignant syndrome (NMS). Other laboratory test results suggest diabetes or renal failure (Table 1). Lumbar puncture shows protein at 91 mg/dL, glucose at 74 mg/dL, and red- and white-blood-cell counts at 0 and 1, respectively. CSF Gram’s stain and brain CT are unremarkable. ECG is normal except for sinus tachycardia. Serum lithium is normal (1.1 mmol/L).
Mr. M undergoes tracheal intubation and receives ceftazidime, dose unknown, because chest radiograph shows lower lung opacities, suggesting aspiration. He receives morphine, 2 to 4 mg hourly as needed, to calm him during intubation. He is then transferred to the intensive care unit.
Table 1
Diabetes, renal failure, or NMS? The story behind Mr. M’s laboratory values
| Mr. M’s reading | Normal range | Might suggest | |
|---|---|---|---|
| CPK | 2,939 IU/L | 8-150 IU/L | NMS |
| Serum creatinine | 1.9 mg/dL | 0.6-1.5 mg/dL | Renal failure, a complication from elevated CPK |
| Serum glucose | 143 mg/dL | 66-99 mg/dL | Diabetes mellitus |
| NMS: Neuroleptic malignant syndrome | |||
| CPK: Creatine phosphokinase | |||
The authors’ observations
NMS, a potentially fatal side effect of antipsychotics, is characterized by rigidity, hyperthermia, and autonomic instability1—as seen with Mr. M.
The patient’s rigidity, elevated creatine kinase, and face and arm abrasions could suggest a seizure. Mr. M’s EEG is negative, however, and he has no history of seizures or head trauma, so seizure is ruled out.
Researchers have associated bupropion with a small risk of developing seizures. Richmond and Zwar2 reported a 0.1% risk with bupropion, ≥300 mg/d, but Mr. M was taking 150 mg/d. Dunner et al3 estimated the risk of developing seizure while taking standard-release bupropion—the form Mr. M used—at 0.06%, but patients in this study who developed seizures typically had a past seizure disorder or head trauma.
The combination of hyperthermia, tachycardia, altered mental status, and positive chest X-ray suggest pneumonia, which was addressed with antibiotics. Pneumonia, however, does not solely account for Mr. M’s fever, rigidity, and profoundly increased CPK. These findings suggest NMS.
The Glasgow Coma Scale (GCS) is used to quantitatively rate degree of responsiveness in critically ill or injured patients (Table 2). Total scores range from 3 to 15 based on the patient’s best eye, motor, and verbal responses. Total score ≤8 indicates a probable coma. Serial GCS scores can measure clinical course in comatose patients.
Table 2
Using Glasgow Coma Scale to determine level of consciousness
| Component | Response | Score |
|---|---|---|
| Best eye response | No eye opening | 1 |
| Eye opening to pain | 2 | |
| Eye opening to verbal command | 3 | |
| Eyes open spontaneously | 4 | |
| Best verbal response | No verbal response | 1 |
| Incomprehensible sounds | 2 | |
| Inappropriate words | 3 | |
| Confused | 4 | |
| Oriented | 5 | |
| Best motor response | No motor response | 1 |
| Extension to pain | 2 | |
| Flexion to pain | 3 | |
| Withdrawal from pain | 4 | |
| Localizing pain | 5 | |
| Obeys commands | 6 | |
| Total score ≤8 is severe, and 90% of patients with scores ≤8 are in a coma). Coma is defined as not opening eyes, not obeying commands, and not saying understandable words. Composite scores listing eye, verbal, and motor responses (such as E3V3M5) are clinically more useful than totals. | ||
| Source: Reprinted from Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet 1974;304(7872):81-4, with permission from Elsevier. | ||
Treatment: slow progress
In the ICU, we diagnose NMS and stop all psychotropics, fearing that interactions between any of them might be causing NMS. We give midazolam, 1 to 2 mg hourly as needed for agitation, and continue morphine, 2 to 4 mg hourly as needed for pain. We stop ceftazidime after ruling out aspiration risk.
On day 2 of hospitalization, we call the neurology and consultation-liaison (C-L) psychiatry services. The C-L psychiatrist attempts a mental status examination, but Mr. M is too frail and sedated to communicate. Neurologic exam shows increased foot rigidity, and follow-up studies show negative EEG, normal head and neck MRIs and MRAs, a peak in CPK at 5,487 IU/L, and normal chest films.
We taper and discontinue midazolam and morphine, and Mr. M’s consciousness improves as the dosages decrease. We add lorazepam, 1 mg tid, to address Mr. M’s agitation. He also starts physical therapy to address potential movement problems caused by laying static for 3 days. By day 7, he is extubated and transferred to the general medical unit.
On day 9, Mr. M’s recall and concentration are diminished, and he cannot follow a 3-step command. His Mini-Mental State Examination (MMSE) score of 17 points to a cognitive impairment.
By day 12, residual psychosis is increasing Mr. M’s confusion, paranoia, and agitation. Despite this complication, he is able to work with his occupational and physical therapists.
By day 20, Mr. M becomes more paranoid, with tangential and loose associations. To address these symptoms, we stop lorazepam and start aripiprazole, 15 mg each morning. Because aripiprazole is a partial dopamine agonist and antagonist, it is less likely than other antipsychotics to cause recurrence of NMS symptoms.
Four days later, Mr. M is medically cleared for transfer to the county psychiatric hospital. Creatinine and CPK elevations, metabolic acidosis, and anemia have resolved.
Treatment: new facility, new drugs
On initial evaluation at the psychiatric hospital, Mr. M is cooperative and aware of person, place, and time. His thought processes range from tangential to disorganized, and his paranoia persists.
The attending psychiatrist stops aripiprazole and starts risperidone, 1 mg bid, possibly because he is less familiar with aripiprazole—a newer antipsychotic— than with risperidone. Laboratory results within 3 days of starting risperidone show normal serum levels, blood counts, liver enzymes, and CPK.
On day 2 at the psychiatric hospital, Mr. M’s behavior worsens; he frequently disrobes in front of others, yells at staff, and requires verbal redirection. His MMSE score has fallen to 15. The attending psychiatrist modifies risperidone to 2 mg nightly and adds donepezil, 10 mg each morning, to try to reverse his cognitive decline.
By day 8, Mr. M is more cooperative and his behavior improves. He is transferred back to his board-and-care facility on risperidone and donepezil at the above dosages.
The following month, Mr. M presents to his outpatient psychiatrist with improved cognitive function, but he is still delusional. The psychiatrist stops risperidone and donepezil and resumes olanzapine, 7.5 mg each morning and 10 mg nightly, and chlorpromazine, 50 mg nightly, to try to restore the patient’s pre-NMS function.
Mr. M undergoes successful prostate cancer surgery before his 3-month psychiatry follow-up, at which the psychiatrist adds lithium carbonate, 300 mg tid, for residual irritability. Serum lithium levels are normal; bupropion is not restarted.
One year after presentation, Mr. M is minimally delusional but functioning well. No symptoms suggesting NMS recurrence have been reported.
The authors’ observations
Though the precise mechanism is unknown, NMS has been linked with use of FGAs such as chlorpromazine, which can trigger excessive dopamine blockade.4 Studies increasingly associate SGAs such as olanzapine, risperidone, and aripiprazole with NMS onset.4-6 Mood stabilizers such as lithium carbonate also have been implicated, especially when used with antipsychotics.6-9 No association between antibiotics and NMS has been found.
For years, Mr. M has been taking FGAs and concomitant olanzapine and lithium carbonate without developing NMS symptoms until now. Since discharge, he has been free of NMS symptoms despite taking two SGAs (aripiprazole and risperidone) at different times and later resuming chlorpromazine, olanzapine, and lithium carbonate.
Of note, bupropion—the last psychotropic added before NMS onset—has not been restarted. The literature does not link bupropion to NMS, although one case report10 suggests an association between fluoxetine and NMS after the patient had taken several antipsychotic/antidepressant combinations.
As a dopamine agonist, bupropion should protect against NMS. Case reports,11,12 however, have described patients who developed NMS after antipsychotics were discontinued, and stopping an antipsychotic essentially mimics bupropion’s action by eliminating the dopamine blockade. Additionally, bupropion’s norepinephrine modulation could have precipitated NMS by dysregulating the sympathetic nervous system.13
Mr. M’s board-and-care operator indicated that the patient’s tobacco consumption decreased—from about a pack to a half-pack of cigarettes daily—after bupropion was added. Alternatively, the effects of pneumonia could have curtailed Mr. M’s smoking. Because nicotine increases metabolism of neuroleptics,14,15 decreased nicotine consumption might have increased dopamine blockade to the point of causing NMS.
Other possibilities. Mr. M’s pneumonia might have caused dehydration, which can also lead to NMS.
Bupropion also reportedly alters metabolism of chlorpromazine and other phenothiazine antipsychotics by inhibiting the cytochrome P-450 2D6 isoenzyme. This pharmacokinetic interaction could have precipitated Mr. M’s NMS episode independent of an antipsychotic dosage increase.16
Because this case is so complex, pinpointing a specific cause for Mr. M’s apparent NMS symptoms is difficult. Be aware that combining psychotropics can lead to NMS. Patients who present with mental status changes, hyperthermia, rigidity, and/or increased creatine kinase while taking psychotropics should be promptly evaluated and managed.
Treating NMS
A review of NMS treatment by Davis et al17 suggests that you:
- consider NMS in the differential diagnosis of an acutely delirious patient who has used antipsychotics, no matter how long he or she has been taking the medication(s) or how stable the dosage
- check for other signs of NMS—such as rigidity or autonomic instability—during the physical examination.
- consider NMS as a possible cause of dysarthria, diaphoresis, dysphagia, sialorrhea, and myoclonus, although these are less common signs of the disorder
- include CPK levels, chemistry panel, CBC, and liver enzyme assessment in the early evaluation of laboratory results. Consider performing a urine drug screen to check for illicit substance use. Head CT results might also help confirm NMS diagnosis.
If sedation becomes necessary, use benzodiazepines cautiously. Serial CPKs and daily reassessment of clinical degree of rigidity are essential; continued rigidity may indicate use of dopamine agonists and dantrolene.17
Related resources
- Neuroleptic Malignant Syndrome Information Service. Archive of articles addressing NMS diagnosis and treatment, and listing of psychotropics associated with NMS. www.nmsis.org.
- Aripiprazole • Abilify
- Benztropine • Cogentin
- Bupropion SR • Wellbutrin, Zyban
- Ceftazidime • various
- Chlorpromazine • Thorazine
- Dantrolene • Dantrium
- Donepezil • Aricept
- Lithium carbonate • various
- Lorazepam • Ativan
- Olanzapine • Zyprexa
- Risperidone • Risperdal
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Diagnostic and statistical manual of mental disorders, 4th ed., text rev. Washington, DC: American Psychiatric Association, 2000.
2. Richmond R, Zwar N. Review of bupropion for smoking cessation. Drug Alcohol Rev 2003;22:203-20.
3. Dunner DL, Zisook S, Billow A, et al. A prospective safety study for bupropion sustained-release in the treatment of depression. J Clin Psychiatry 1998;59:366-73.
4. Caroff SN, Mann SC, Campbell EC. Atypical antipsychotics and neuroleptic malignant syndrome. Psychiatr Ann 2000;30:314-21.
5. Berry N, Pradhan S, Sagar R, Gupta SK. Neuroleptic malignant syndrome in an adolescent receiving olanzapine-lithium combination therapy. Pharmacotherapy 2003;23:255-9
6. Ananth J, Parameswaran S, Gunatilake S, et al. Neuroleptic malignant syndrome and atypical antipsychotic drugs. J Clin Psychiatry 2004;65:464-70.
7. Caroff SN, Mann SC. Neuroleptic malignant syndrome. Med Clin North Am 1993;77:185-202.
8. Bourgeois JA, Kahn DR. Neuroleptic malignant syndrome following administration of risperidone and lithium. J Clin Psychopharmacol 2003;23:315-6.
9. Gill J, Singh H, Nugent K. Acute lithium intoxication and neuroleptic malignant syndrome. Pharmacotherapy 2003;23:811-15.
10. Halman M, Goldbloom DS. Fluoxetine and neuroleptic malignant syndrome. Biol Psychiatry 1990;28:518-21.
11. Spivak B, Gonen N, Mester R, et al. Neuroleptic malignant syndrome associated with abrupt withdrawal of anticholinergic agents. Int Clin Psychopharmacol 1996;11:207-9.
12. Rosse R, Ciolino C. Dopamine agonists and neuroleptic malignant syndrome. Am J Psychiatry 1985;142:270-1.
13. Gurrera RJ. Sympathoadrenal hyperactivity and the etiology of neuroleptic malignant syndrome. Am J Psychiatry 1999;156:169-80.
14. Ereshefsky L, Jann MW, Saklad SR, et al. Effects of smoking on fluphenazine clearance in psychiatric inpatients. Biol Psychiatry 1985;20:329-32.
15. Jann MW, Saklad SR, Ereshefsky L, et al. Effects of smoking on haloperidol and reduced haloperidol plasma concentrations and haloperidol clearance. Psychopharmacology (Berl) 1986;90:468-70.
16. Sandson NB, Armstrong SC, Cozza KL. An overview of psychotropic drug-drug interactions. Psychosomatics 2005;46:464-94.
17. Davis JM, Caroff SN, Mann SC. Treatment of neuroleptic malignant syndrome. Psychiatr Ann 2000;30:325-31.
Presentation: unconscious on the street
Emergency medical personnel bring Mr. M, age 66, to the ER after passers-by find him supine on the sidewalk. On arrival, he is comatose as confirmed by a Glasgow Coma Scale score of 8 (eye opening 3, verbal response 2, motor response 3). Systolic blood pressure is 108 mm Hg on palpation, pulse is 135 beats per minute, and temperature is 105 °F. Minor abrasions cover his face and arms, and his hands and feet are rigid.
Mr. M has lived at a board-and-care facility for 30 years. The facility’s operator tells us that Mr. M has had schizophrenia for 40 years and has been taking:
- olanzapine, 7.5 mg each morning and 10 mg at bedtime
- chlorpromazine, 50 mg nightly
- lithium carbonate, 300 mg tid
- and benztropine, 2 mg bid.
Three weeks ago, Mr. M was hospitalized for 6 days with pneumonia. In 3 months, he will undergo surgery for prostate cancer. He is taking no medication for the prostate cancer.
Creatine phosphokinase (CPK) is 2,939 IU/L, indicating neuroleptic malignant syndrome (NMS). Other laboratory test results suggest diabetes or renal failure (Table 1). Lumbar puncture shows protein at 91 mg/dL, glucose at 74 mg/dL, and red- and white-blood-cell counts at 0 and 1, respectively. CSF Gram’s stain and brain CT are unremarkable. ECG is normal except for sinus tachycardia. Serum lithium is normal (1.1 mmol/L).
Mr. M undergoes tracheal intubation and receives ceftazidime, dose unknown, because chest radiograph shows lower lung opacities, suggesting aspiration. He receives morphine, 2 to 4 mg hourly as needed, to calm him during intubation. He is then transferred to the intensive care unit.
Table 1
Diabetes, renal failure, or NMS? The story behind Mr. M’s laboratory values
| Mr. M’s reading | Normal range | Might suggest | |
|---|---|---|---|
| CPK | 2,939 IU/L | 8-150 IU/L | NMS |
| Serum creatinine | 1.9 mg/dL | 0.6-1.5 mg/dL | Renal failure, a complication from elevated CPK |
| Serum glucose | 143 mg/dL | 66-99 mg/dL | Diabetes mellitus |
| NMS: Neuroleptic malignant syndrome | |||
| CPK: Creatine phosphokinase | |||
The authors’ observations
NMS, a potentially fatal side effect of antipsychotics, is characterized by rigidity, hyperthermia, and autonomic instability1—as seen with Mr. M.
The patient’s rigidity, elevated creatine kinase, and face and arm abrasions could suggest a seizure. Mr. M’s EEG is negative, however, and he has no history of seizures or head trauma, so seizure is ruled out.
Researchers have associated bupropion with a small risk of developing seizures. Richmond and Zwar2 reported a 0.1% risk with bupropion, ≥300 mg/d, but Mr. M was taking 150 mg/d. Dunner et al3 estimated the risk of developing seizure while taking standard-release bupropion—the form Mr. M used—at 0.06%, but patients in this study who developed seizures typically had a past seizure disorder or head trauma.
The combination of hyperthermia, tachycardia, altered mental status, and positive chest X-ray suggest pneumonia, which was addressed with antibiotics. Pneumonia, however, does not solely account for Mr. M’s fever, rigidity, and profoundly increased CPK. These findings suggest NMS.
The Glasgow Coma Scale (GCS) is used to quantitatively rate degree of responsiveness in critically ill or injured patients (Table 2). Total scores range from 3 to 15 based on the patient’s best eye, motor, and verbal responses. Total score ≤8 indicates a probable coma. Serial GCS scores can measure clinical course in comatose patients.
Table 2
Using Glasgow Coma Scale to determine level of consciousness
| Component | Response | Score |
|---|---|---|
| Best eye response | No eye opening | 1 |
| Eye opening to pain | 2 | |
| Eye opening to verbal command | 3 | |
| Eyes open spontaneously | 4 | |
| Best verbal response | No verbal response | 1 |
| Incomprehensible sounds | 2 | |
| Inappropriate words | 3 | |
| Confused | 4 | |
| Oriented | 5 | |
| Best motor response | No motor response | 1 |
| Extension to pain | 2 | |
| Flexion to pain | 3 | |
| Withdrawal from pain | 4 | |
| Localizing pain | 5 | |
| Obeys commands | 6 | |
| Total score ≤8 is severe, and 90% of patients with scores ≤8 are in a coma). Coma is defined as not opening eyes, not obeying commands, and not saying understandable words. Composite scores listing eye, verbal, and motor responses (such as E3V3M5) are clinically more useful than totals. | ||
| Source: Reprinted from Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet 1974;304(7872):81-4, with permission from Elsevier. | ||
Treatment: slow progress
In the ICU, we diagnose NMS and stop all psychotropics, fearing that interactions between any of them might be causing NMS. We give midazolam, 1 to 2 mg hourly as needed for agitation, and continue morphine, 2 to 4 mg hourly as needed for pain. We stop ceftazidime after ruling out aspiration risk.
On day 2 of hospitalization, we call the neurology and consultation-liaison (C-L) psychiatry services. The C-L psychiatrist attempts a mental status examination, but Mr. M is too frail and sedated to communicate. Neurologic exam shows increased foot rigidity, and follow-up studies show negative EEG, normal head and neck MRIs and MRAs, a peak in CPK at 5,487 IU/L, and normal chest films.
We taper and discontinue midazolam and morphine, and Mr. M’s consciousness improves as the dosages decrease. We add lorazepam, 1 mg tid, to address Mr. M’s agitation. He also starts physical therapy to address potential movement problems caused by laying static for 3 days. By day 7, he is extubated and transferred to the general medical unit.
On day 9, Mr. M’s recall and concentration are diminished, and he cannot follow a 3-step command. His Mini-Mental State Examination (MMSE) score of 17 points to a cognitive impairment.
By day 12, residual psychosis is increasing Mr. M’s confusion, paranoia, and agitation. Despite this complication, he is able to work with his occupational and physical therapists.
By day 20, Mr. M becomes more paranoid, with tangential and loose associations. To address these symptoms, we stop lorazepam and start aripiprazole, 15 mg each morning. Because aripiprazole is a partial dopamine agonist and antagonist, it is less likely than other antipsychotics to cause recurrence of NMS symptoms.
Four days later, Mr. M is medically cleared for transfer to the county psychiatric hospital. Creatinine and CPK elevations, metabolic acidosis, and anemia have resolved.
Treatment: new facility, new drugs
On initial evaluation at the psychiatric hospital, Mr. M is cooperative and aware of person, place, and time. His thought processes range from tangential to disorganized, and his paranoia persists.
The attending psychiatrist stops aripiprazole and starts risperidone, 1 mg bid, possibly because he is less familiar with aripiprazole—a newer antipsychotic— than with risperidone. Laboratory results within 3 days of starting risperidone show normal serum levels, blood counts, liver enzymes, and CPK.
On day 2 at the psychiatric hospital, Mr. M’s behavior worsens; he frequently disrobes in front of others, yells at staff, and requires verbal redirection. His MMSE score has fallen to 15. The attending psychiatrist modifies risperidone to 2 mg nightly and adds donepezil, 10 mg each morning, to try to reverse his cognitive decline.
By day 8, Mr. M is more cooperative and his behavior improves. He is transferred back to his board-and-care facility on risperidone and donepezil at the above dosages.
The following month, Mr. M presents to his outpatient psychiatrist with improved cognitive function, but he is still delusional. The psychiatrist stops risperidone and donepezil and resumes olanzapine, 7.5 mg each morning and 10 mg nightly, and chlorpromazine, 50 mg nightly, to try to restore the patient’s pre-NMS function.
Mr. M undergoes successful prostate cancer surgery before his 3-month psychiatry follow-up, at which the psychiatrist adds lithium carbonate, 300 mg tid, for residual irritability. Serum lithium levels are normal; bupropion is not restarted.
One year after presentation, Mr. M is minimally delusional but functioning well. No symptoms suggesting NMS recurrence have been reported.
The authors’ observations
Though the precise mechanism is unknown, NMS has been linked with use of FGAs such as chlorpromazine, which can trigger excessive dopamine blockade.4 Studies increasingly associate SGAs such as olanzapine, risperidone, and aripiprazole with NMS onset.4-6 Mood stabilizers such as lithium carbonate also have been implicated, especially when used with antipsychotics.6-9 No association between antibiotics and NMS has been found.
For years, Mr. M has been taking FGAs and concomitant olanzapine and lithium carbonate without developing NMS symptoms until now. Since discharge, he has been free of NMS symptoms despite taking two SGAs (aripiprazole and risperidone) at different times and later resuming chlorpromazine, olanzapine, and lithium carbonate.
Of note, bupropion—the last psychotropic added before NMS onset—has not been restarted. The literature does not link bupropion to NMS, although one case report10 suggests an association between fluoxetine and NMS after the patient had taken several antipsychotic/antidepressant combinations.
As a dopamine agonist, bupropion should protect against NMS. Case reports,11,12 however, have described patients who developed NMS after antipsychotics were discontinued, and stopping an antipsychotic essentially mimics bupropion’s action by eliminating the dopamine blockade. Additionally, bupropion’s norepinephrine modulation could have precipitated NMS by dysregulating the sympathetic nervous system.13
Mr. M’s board-and-care operator indicated that the patient’s tobacco consumption decreased—from about a pack to a half-pack of cigarettes daily—after bupropion was added. Alternatively, the effects of pneumonia could have curtailed Mr. M’s smoking. Because nicotine increases metabolism of neuroleptics,14,15 decreased nicotine consumption might have increased dopamine blockade to the point of causing NMS.
Other possibilities. Mr. M’s pneumonia might have caused dehydration, which can also lead to NMS.
Bupropion also reportedly alters metabolism of chlorpromazine and other phenothiazine antipsychotics by inhibiting the cytochrome P-450 2D6 isoenzyme. This pharmacokinetic interaction could have precipitated Mr. M’s NMS episode independent of an antipsychotic dosage increase.16
Because this case is so complex, pinpointing a specific cause for Mr. M’s apparent NMS symptoms is difficult. Be aware that combining psychotropics can lead to NMS. Patients who present with mental status changes, hyperthermia, rigidity, and/or increased creatine kinase while taking psychotropics should be promptly evaluated and managed.
Treating NMS
A review of NMS treatment by Davis et al17 suggests that you:
- consider NMS in the differential diagnosis of an acutely delirious patient who has used antipsychotics, no matter how long he or she has been taking the medication(s) or how stable the dosage
- check for other signs of NMS—such as rigidity or autonomic instability—during the physical examination.
- consider NMS as a possible cause of dysarthria, diaphoresis, dysphagia, sialorrhea, and myoclonus, although these are less common signs of the disorder
- include CPK levels, chemistry panel, CBC, and liver enzyme assessment in the early evaluation of laboratory results. Consider performing a urine drug screen to check for illicit substance use. Head CT results might also help confirm NMS diagnosis.
If sedation becomes necessary, use benzodiazepines cautiously. Serial CPKs and daily reassessment of clinical degree of rigidity are essential; continued rigidity may indicate use of dopamine agonists and dantrolene.17
Related resources
- Neuroleptic Malignant Syndrome Information Service. Archive of articles addressing NMS diagnosis and treatment, and listing of psychotropics associated with NMS. www.nmsis.org.
- Aripiprazole • Abilify
- Benztropine • Cogentin
- Bupropion SR • Wellbutrin, Zyban
- Ceftazidime • various
- Chlorpromazine • Thorazine
- Dantrolene • Dantrium
- Donepezil • Aricept
- Lithium carbonate • various
- Lorazepam • Ativan
- Olanzapine • Zyprexa
- Risperidone • Risperdal
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Presentation: unconscious on the street
Emergency medical personnel bring Mr. M, age 66, to the ER after passers-by find him supine on the sidewalk. On arrival, he is comatose as confirmed by a Glasgow Coma Scale score of 8 (eye opening 3, verbal response 2, motor response 3). Systolic blood pressure is 108 mm Hg on palpation, pulse is 135 beats per minute, and temperature is 105 °F. Minor abrasions cover his face and arms, and his hands and feet are rigid.
Mr. M has lived at a board-and-care facility for 30 years. The facility’s operator tells us that Mr. M has had schizophrenia for 40 years and has been taking:
- olanzapine, 7.5 mg each morning and 10 mg at bedtime
- chlorpromazine, 50 mg nightly
- lithium carbonate, 300 mg tid
- and benztropine, 2 mg bid.
Three weeks ago, Mr. M was hospitalized for 6 days with pneumonia. In 3 months, he will undergo surgery for prostate cancer. He is taking no medication for the prostate cancer.
Creatine phosphokinase (CPK) is 2,939 IU/L, indicating neuroleptic malignant syndrome (NMS). Other laboratory test results suggest diabetes or renal failure (Table 1). Lumbar puncture shows protein at 91 mg/dL, glucose at 74 mg/dL, and red- and white-blood-cell counts at 0 and 1, respectively. CSF Gram’s stain and brain CT are unremarkable. ECG is normal except for sinus tachycardia. Serum lithium is normal (1.1 mmol/L).
Mr. M undergoes tracheal intubation and receives ceftazidime, dose unknown, because chest radiograph shows lower lung opacities, suggesting aspiration. He receives morphine, 2 to 4 mg hourly as needed, to calm him during intubation. He is then transferred to the intensive care unit.
Table 1
Diabetes, renal failure, or NMS? The story behind Mr. M’s laboratory values
| Mr. M’s reading | Normal range | Might suggest | |
|---|---|---|---|
| CPK | 2,939 IU/L | 8-150 IU/L | NMS |
| Serum creatinine | 1.9 mg/dL | 0.6-1.5 mg/dL | Renal failure, a complication from elevated CPK |
| Serum glucose | 143 mg/dL | 66-99 mg/dL | Diabetes mellitus |
| NMS: Neuroleptic malignant syndrome | |||
| CPK: Creatine phosphokinase | |||
The authors’ observations
NMS, a potentially fatal side effect of antipsychotics, is characterized by rigidity, hyperthermia, and autonomic instability1—as seen with Mr. M.
The patient’s rigidity, elevated creatine kinase, and face and arm abrasions could suggest a seizure. Mr. M’s EEG is negative, however, and he has no history of seizures or head trauma, so seizure is ruled out.
Researchers have associated bupropion with a small risk of developing seizures. Richmond and Zwar2 reported a 0.1% risk with bupropion, ≥300 mg/d, but Mr. M was taking 150 mg/d. Dunner et al3 estimated the risk of developing seizure while taking standard-release bupropion—the form Mr. M used—at 0.06%, but patients in this study who developed seizures typically had a past seizure disorder or head trauma.
The combination of hyperthermia, tachycardia, altered mental status, and positive chest X-ray suggest pneumonia, which was addressed with antibiotics. Pneumonia, however, does not solely account for Mr. M’s fever, rigidity, and profoundly increased CPK. These findings suggest NMS.
The Glasgow Coma Scale (GCS) is used to quantitatively rate degree of responsiveness in critically ill or injured patients (Table 2). Total scores range from 3 to 15 based on the patient’s best eye, motor, and verbal responses. Total score ≤8 indicates a probable coma. Serial GCS scores can measure clinical course in comatose patients.
Table 2
Using Glasgow Coma Scale to determine level of consciousness
| Component | Response | Score |
|---|---|---|
| Best eye response | No eye opening | 1 |
| Eye opening to pain | 2 | |
| Eye opening to verbal command | 3 | |
| Eyes open spontaneously | 4 | |
| Best verbal response | No verbal response | 1 |
| Incomprehensible sounds | 2 | |
| Inappropriate words | 3 | |
| Confused | 4 | |
| Oriented | 5 | |
| Best motor response | No motor response | 1 |
| Extension to pain | 2 | |
| Flexion to pain | 3 | |
| Withdrawal from pain | 4 | |
| Localizing pain | 5 | |
| Obeys commands | 6 | |
| Total score ≤8 is severe, and 90% of patients with scores ≤8 are in a coma). Coma is defined as not opening eyes, not obeying commands, and not saying understandable words. Composite scores listing eye, verbal, and motor responses (such as E3V3M5) are clinically more useful than totals. | ||
| Source: Reprinted from Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet 1974;304(7872):81-4, with permission from Elsevier. | ||
Treatment: slow progress
In the ICU, we diagnose NMS and stop all psychotropics, fearing that interactions between any of them might be causing NMS. We give midazolam, 1 to 2 mg hourly as needed for agitation, and continue morphine, 2 to 4 mg hourly as needed for pain. We stop ceftazidime after ruling out aspiration risk.
On day 2 of hospitalization, we call the neurology and consultation-liaison (C-L) psychiatry services. The C-L psychiatrist attempts a mental status examination, but Mr. M is too frail and sedated to communicate. Neurologic exam shows increased foot rigidity, and follow-up studies show negative EEG, normal head and neck MRIs and MRAs, a peak in CPK at 5,487 IU/L, and normal chest films.
We taper and discontinue midazolam and morphine, and Mr. M’s consciousness improves as the dosages decrease. We add lorazepam, 1 mg tid, to address Mr. M’s agitation. He also starts physical therapy to address potential movement problems caused by laying static for 3 days. By day 7, he is extubated and transferred to the general medical unit.
On day 9, Mr. M’s recall and concentration are diminished, and he cannot follow a 3-step command. His Mini-Mental State Examination (MMSE) score of 17 points to a cognitive impairment.
By day 12, residual psychosis is increasing Mr. M’s confusion, paranoia, and agitation. Despite this complication, he is able to work with his occupational and physical therapists.
By day 20, Mr. M becomes more paranoid, with tangential and loose associations. To address these symptoms, we stop lorazepam and start aripiprazole, 15 mg each morning. Because aripiprazole is a partial dopamine agonist and antagonist, it is less likely than other antipsychotics to cause recurrence of NMS symptoms.
Four days later, Mr. M is medically cleared for transfer to the county psychiatric hospital. Creatinine and CPK elevations, metabolic acidosis, and anemia have resolved.
Treatment: new facility, new drugs
On initial evaluation at the psychiatric hospital, Mr. M is cooperative and aware of person, place, and time. His thought processes range from tangential to disorganized, and his paranoia persists.
The attending psychiatrist stops aripiprazole and starts risperidone, 1 mg bid, possibly because he is less familiar with aripiprazole—a newer antipsychotic— than with risperidone. Laboratory results within 3 days of starting risperidone show normal serum levels, blood counts, liver enzymes, and CPK.
On day 2 at the psychiatric hospital, Mr. M’s behavior worsens; he frequently disrobes in front of others, yells at staff, and requires verbal redirection. His MMSE score has fallen to 15. The attending psychiatrist modifies risperidone to 2 mg nightly and adds donepezil, 10 mg each morning, to try to reverse his cognitive decline.
By day 8, Mr. M is more cooperative and his behavior improves. He is transferred back to his board-and-care facility on risperidone and donepezil at the above dosages.
The following month, Mr. M presents to his outpatient psychiatrist with improved cognitive function, but he is still delusional. The psychiatrist stops risperidone and donepezil and resumes olanzapine, 7.5 mg each morning and 10 mg nightly, and chlorpromazine, 50 mg nightly, to try to restore the patient’s pre-NMS function.
Mr. M undergoes successful prostate cancer surgery before his 3-month psychiatry follow-up, at which the psychiatrist adds lithium carbonate, 300 mg tid, for residual irritability. Serum lithium levels are normal; bupropion is not restarted.
One year after presentation, Mr. M is minimally delusional but functioning well. No symptoms suggesting NMS recurrence have been reported.
The authors’ observations
Though the precise mechanism is unknown, NMS has been linked with use of FGAs such as chlorpromazine, which can trigger excessive dopamine blockade.4 Studies increasingly associate SGAs such as olanzapine, risperidone, and aripiprazole with NMS onset.4-6 Mood stabilizers such as lithium carbonate also have been implicated, especially when used with antipsychotics.6-9 No association between antibiotics and NMS has been found.
For years, Mr. M has been taking FGAs and concomitant olanzapine and lithium carbonate without developing NMS symptoms until now. Since discharge, he has been free of NMS symptoms despite taking two SGAs (aripiprazole and risperidone) at different times and later resuming chlorpromazine, olanzapine, and lithium carbonate.
Of note, bupropion—the last psychotropic added before NMS onset—has not been restarted. The literature does not link bupropion to NMS, although one case report10 suggests an association between fluoxetine and NMS after the patient had taken several antipsychotic/antidepressant combinations.
As a dopamine agonist, bupropion should protect against NMS. Case reports,11,12 however, have described patients who developed NMS after antipsychotics were discontinued, and stopping an antipsychotic essentially mimics bupropion’s action by eliminating the dopamine blockade. Additionally, bupropion’s norepinephrine modulation could have precipitated NMS by dysregulating the sympathetic nervous system.13
Mr. M’s board-and-care operator indicated that the patient’s tobacco consumption decreased—from about a pack to a half-pack of cigarettes daily—after bupropion was added. Alternatively, the effects of pneumonia could have curtailed Mr. M’s smoking. Because nicotine increases metabolism of neuroleptics,14,15 decreased nicotine consumption might have increased dopamine blockade to the point of causing NMS.
Other possibilities. Mr. M’s pneumonia might have caused dehydration, which can also lead to NMS.
Bupropion also reportedly alters metabolism of chlorpromazine and other phenothiazine antipsychotics by inhibiting the cytochrome P-450 2D6 isoenzyme. This pharmacokinetic interaction could have precipitated Mr. M’s NMS episode independent of an antipsychotic dosage increase.16
Because this case is so complex, pinpointing a specific cause for Mr. M’s apparent NMS symptoms is difficult. Be aware that combining psychotropics can lead to NMS. Patients who present with mental status changes, hyperthermia, rigidity, and/or increased creatine kinase while taking psychotropics should be promptly evaluated and managed.
Treating NMS
A review of NMS treatment by Davis et al17 suggests that you:
- consider NMS in the differential diagnosis of an acutely delirious patient who has used antipsychotics, no matter how long he or she has been taking the medication(s) or how stable the dosage
- check for other signs of NMS—such as rigidity or autonomic instability—during the physical examination.
- consider NMS as a possible cause of dysarthria, diaphoresis, dysphagia, sialorrhea, and myoclonus, although these are less common signs of the disorder
- include CPK levels, chemistry panel, CBC, and liver enzyme assessment in the early evaluation of laboratory results. Consider performing a urine drug screen to check for illicit substance use. Head CT results might also help confirm NMS diagnosis.
If sedation becomes necessary, use benzodiazepines cautiously. Serial CPKs and daily reassessment of clinical degree of rigidity are essential; continued rigidity may indicate use of dopamine agonists and dantrolene.17
Related resources
- Neuroleptic Malignant Syndrome Information Service. Archive of articles addressing NMS diagnosis and treatment, and listing of psychotropics associated with NMS. www.nmsis.org.
- Aripiprazole • Abilify
- Benztropine • Cogentin
- Bupropion SR • Wellbutrin, Zyban
- Ceftazidime • various
- Chlorpromazine • Thorazine
- Dantrolene • Dantrium
- Donepezil • Aricept
- Lithium carbonate • various
- Lorazepam • Ativan
- Olanzapine • Zyprexa
- Risperidone • Risperdal
The authors report no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Diagnostic and statistical manual of mental disorders, 4th ed., text rev. Washington, DC: American Psychiatric Association, 2000.
2. Richmond R, Zwar N. Review of bupropion for smoking cessation. Drug Alcohol Rev 2003;22:203-20.
3. Dunner DL, Zisook S, Billow A, et al. A prospective safety study for bupropion sustained-release in the treatment of depression. J Clin Psychiatry 1998;59:366-73.
4. Caroff SN, Mann SC, Campbell EC. Atypical antipsychotics and neuroleptic malignant syndrome. Psychiatr Ann 2000;30:314-21.
5. Berry N, Pradhan S, Sagar R, Gupta SK. Neuroleptic malignant syndrome in an adolescent receiving olanzapine-lithium combination therapy. Pharmacotherapy 2003;23:255-9
6. Ananth J, Parameswaran S, Gunatilake S, et al. Neuroleptic malignant syndrome and atypical antipsychotic drugs. J Clin Psychiatry 2004;65:464-70.
7. Caroff SN, Mann SC. Neuroleptic malignant syndrome. Med Clin North Am 1993;77:185-202.
8. Bourgeois JA, Kahn DR. Neuroleptic malignant syndrome following administration of risperidone and lithium. J Clin Psychopharmacol 2003;23:315-6.
9. Gill J, Singh H, Nugent K. Acute lithium intoxication and neuroleptic malignant syndrome. Pharmacotherapy 2003;23:811-15.
10. Halman M, Goldbloom DS. Fluoxetine and neuroleptic malignant syndrome. Biol Psychiatry 1990;28:518-21.
11. Spivak B, Gonen N, Mester R, et al. Neuroleptic malignant syndrome associated with abrupt withdrawal of anticholinergic agents. Int Clin Psychopharmacol 1996;11:207-9.
12. Rosse R, Ciolino C. Dopamine agonists and neuroleptic malignant syndrome. Am J Psychiatry 1985;142:270-1.
13. Gurrera RJ. Sympathoadrenal hyperactivity and the etiology of neuroleptic malignant syndrome. Am J Psychiatry 1999;156:169-80.
14. Ereshefsky L, Jann MW, Saklad SR, et al. Effects of smoking on fluphenazine clearance in psychiatric inpatients. Biol Psychiatry 1985;20:329-32.
15. Jann MW, Saklad SR, Ereshefsky L, et al. Effects of smoking on haloperidol and reduced haloperidol plasma concentrations and haloperidol clearance. Psychopharmacology (Berl) 1986;90:468-70.
16. Sandson NB, Armstrong SC, Cozza KL. An overview of psychotropic drug-drug interactions. Psychosomatics 2005;46:464-94.
17. Davis JM, Caroff SN, Mann SC. Treatment of neuroleptic malignant syndrome. Psychiatr Ann 2000;30:325-31.
1. Diagnostic and statistical manual of mental disorders, 4th ed., text rev. Washington, DC: American Psychiatric Association, 2000.
2. Richmond R, Zwar N. Review of bupropion for smoking cessation. Drug Alcohol Rev 2003;22:203-20.
3. Dunner DL, Zisook S, Billow A, et al. A prospective safety study for bupropion sustained-release in the treatment of depression. J Clin Psychiatry 1998;59:366-73.
4. Caroff SN, Mann SC, Campbell EC. Atypical antipsychotics and neuroleptic malignant syndrome. Psychiatr Ann 2000;30:314-21.
5. Berry N, Pradhan S, Sagar R, Gupta SK. Neuroleptic malignant syndrome in an adolescent receiving olanzapine-lithium combination therapy. Pharmacotherapy 2003;23:255-9
6. Ananth J, Parameswaran S, Gunatilake S, et al. Neuroleptic malignant syndrome and atypical antipsychotic drugs. J Clin Psychiatry 2004;65:464-70.
7. Caroff SN, Mann SC. Neuroleptic malignant syndrome. Med Clin North Am 1993;77:185-202.
8. Bourgeois JA, Kahn DR. Neuroleptic malignant syndrome following administration of risperidone and lithium. J Clin Psychopharmacol 2003;23:315-6.
9. Gill J, Singh H, Nugent K. Acute lithium intoxication and neuroleptic malignant syndrome. Pharmacotherapy 2003;23:811-15.
10. Halman M, Goldbloom DS. Fluoxetine and neuroleptic malignant syndrome. Biol Psychiatry 1990;28:518-21.
11. Spivak B, Gonen N, Mester R, et al. Neuroleptic malignant syndrome associated with abrupt withdrawal of anticholinergic agents. Int Clin Psychopharmacol 1996;11:207-9.
12. Rosse R, Ciolino C. Dopamine agonists and neuroleptic malignant syndrome. Am J Psychiatry 1985;142:270-1.
13. Gurrera RJ. Sympathoadrenal hyperactivity and the etiology of neuroleptic malignant syndrome. Am J Psychiatry 1999;156:169-80.
14. Ereshefsky L, Jann MW, Saklad SR, et al. Effects of smoking on fluphenazine clearance in psychiatric inpatients. Biol Psychiatry 1985;20:329-32.
15. Jann MW, Saklad SR, Ereshefsky L, et al. Effects of smoking on haloperidol and reduced haloperidol plasma concentrations and haloperidol clearance. Psychopharmacology (Berl) 1986;90:468-70.
16. Sandson NB, Armstrong SC, Cozza KL. An overview of psychotropic drug-drug interactions. Psychosomatics 2005;46:464-94.
17. Davis JM, Caroff SN, Mann SC. Treatment of neuroleptic malignant syndrome. Psychiatr Ann 2000;30:325-31.