User login
Whole blood viscosity as a biomarker for distant metastasis and survival in HCC
Key clinical point: High diastolic whole blood viscosity (WBV) may serve as a new independent factor associated with extrahepatic metastasis and poor survival in patients with hepatocellular carcinoma (HCC).
Main finding: After adjusting for confounding variables, high diastolic WBV was independently associated with extrahepatic metastasis (adjusted odds ratio, 23.41; P < .001) and poor survival (adjusted hazard ratio, 3.81; P < .001) and significantly predicted extrahepatic metastasis at an optimal cutoff of 16 cP (area under the receiver operating characteristic curve, 0.768; P < .001).
Study details: Findings are from a pilot retrospective study including 181 patients with HCC, of which 148 were treatment-naïve having preserved liver function and 33 received nivolumab.
Disclosures: The study was sponsored by Young Medical Scientist Research Grant through the Daewoong Foundation and the Research Fund of Seoul St. Mary’s Hospital, The Catholic University of Korea. The authors did not declare any conflict of interests.
Source: Han JW et al. PLoS ONE. 2021 Dec 2. doi: 10.1371/journal.pone.0260311.
Key clinical point: High diastolic whole blood viscosity (WBV) may serve as a new independent factor associated with extrahepatic metastasis and poor survival in patients with hepatocellular carcinoma (HCC).
Main finding: After adjusting for confounding variables, high diastolic WBV was independently associated with extrahepatic metastasis (adjusted odds ratio, 23.41; P < .001) and poor survival (adjusted hazard ratio, 3.81; P < .001) and significantly predicted extrahepatic metastasis at an optimal cutoff of 16 cP (area under the receiver operating characteristic curve, 0.768; P < .001).
Study details: Findings are from a pilot retrospective study including 181 patients with HCC, of which 148 were treatment-naïve having preserved liver function and 33 received nivolumab.
Disclosures: The study was sponsored by Young Medical Scientist Research Grant through the Daewoong Foundation and the Research Fund of Seoul St. Mary’s Hospital, The Catholic University of Korea. The authors did not declare any conflict of interests.
Source: Han JW et al. PLoS ONE. 2021 Dec 2. doi: 10.1371/journal.pone.0260311.
Key clinical point: High diastolic whole blood viscosity (WBV) may serve as a new independent factor associated with extrahepatic metastasis and poor survival in patients with hepatocellular carcinoma (HCC).
Main finding: After adjusting for confounding variables, high diastolic WBV was independently associated with extrahepatic metastasis (adjusted odds ratio, 23.41; P < .001) and poor survival (adjusted hazard ratio, 3.81; P < .001) and significantly predicted extrahepatic metastasis at an optimal cutoff of 16 cP (area under the receiver operating characteristic curve, 0.768; P < .001).
Study details: Findings are from a pilot retrospective study including 181 patients with HCC, of which 148 were treatment-naïve having preserved liver function and 33 received nivolumab.
Disclosures: The study was sponsored by Young Medical Scientist Research Grant through the Daewoong Foundation and the Research Fund of Seoul St. Mary’s Hospital, The Catholic University of Korea. The authors did not declare any conflict of interests.
Source: Han JW et al. PLoS ONE. 2021 Dec 2. doi: 10.1371/journal.pone.0260311.
Radiologic response to TACE-RT as a prognostic factor in advanced HCC with macroscopic vascular invasion
Key clinical point: The modified Response Evaluation Criteria in Solid Tumors (mRECIST)-determined radiologic response rate of transarterial chemoembolization (TACE) plus radiotherapy (RT) among patients with advanced hepatocellular carcinoma (HCC) showing macroscopic vascular invasion (MVI) is an independent prognosticator for overall survival (OS).
Main finding: Responders vs nonresponders had significantly longer median OS at 2 months (23.1 months vs 8.0 months; adjusted hazard ratio [aHR], 3.194; P < .001) and 4 months (responders vs nonresponders: 26.5 months vs 9.3 months; aHR, 4.534; P < .001).
Study details: This was a retrospective review study including 427 patients with advanced HCC and MVI who received first-line treatment with TACE plus respiratory-gated 3-dimensional conformal RT in the 2-month analysis, whereas the patient number reduced to 355 in the 4-month analysis.
Disclosures: The study was supported by the Asan Institute for Life Sciences of Asan Medical Center, Seoul, Republic of Korea. The authors declared no conflict of interests.
Source: Jung J et al. Liver Cancer. 2021 Dec 7. doi: 10.1159/000521227.
Key clinical point: The modified Response Evaluation Criteria in Solid Tumors (mRECIST)-determined radiologic response rate of transarterial chemoembolization (TACE) plus radiotherapy (RT) among patients with advanced hepatocellular carcinoma (HCC) showing macroscopic vascular invasion (MVI) is an independent prognosticator for overall survival (OS).
Main finding: Responders vs nonresponders had significantly longer median OS at 2 months (23.1 months vs 8.0 months; adjusted hazard ratio [aHR], 3.194; P < .001) and 4 months (responders vs nonresponders: 26.5 months vs 9.3 months; aHR, 4.534; P < .001).
Study details: This was a retrospective review study including 427 patients with advanced HCC and MVI who received first-line treatment with TACE plus respiratory-gated 3-dimensional conformal RT in the 2-month analysis, whereas the patient number reduced to 355 in the 4-month analysis.
Disclosures: The study was supported by the Asan Institute for Life Sciences of Asan Medical Center, Seoul, Republic of Korea. The authors declared no conflict of interests.
Source: Jung J et al. Liver Cancer. 2021 Dec 7. doi: 10.1159/000521227.
Key clinical point: The modified Response Evaluation Criteria in Solid Tumors (mRECIST)-determined radiologic response rate of transarterial chemoembolization (TACE) plus radiotherapy (RT) among patients with advanced hepatocellular carcinoma (HCC) showing macroscopic vascular invasion (MVI) is an independent prognosticator for overall survival (OS).
Main finding: Responders vs nonresponders had significantly longer median OS at 2 months (23.1 months vs 8.0 months; adjusted hazard ratio [aHR], 3.194; P < .001) and 4 months (responders vs nonresponders: 26.5 months vs 9.3 months; aHR, 4.534; P < .001).
Study details: This was a retrospective review study including 427 patients with advanced HCC and MVI who received first-line treatment with TACE plus respiratory-gated 3-dimensional conformal RT in the 2-month analysis, whereas the patient number reduced to 355 in the 4-month analysis.
Disclosures: The study was supported by the Asan Institute for Life Sciences of Asan Medical Center, Seoul, Republic of Korea. The authors declared no conflict of interests.
Source: Jung J et al. Liver Cancer. 2021 Dec 7. doi: 10.1159/000521227.
HAIC-FO outperforms sorafenib against advanced HCC in phase 3
Key clinical point: Hepatic arterial infusion chemotherapy of infusional fluorouracil, leucovorin, and oxaliplatin (HAIC-FO) is better than sorafenib at improving survival in patients with locally advanced hepatocellular carcinoma (HCC).
Main finding: At a median follow-up of 17.1 and 19.8 months, HAIC-FO- and sorafenib-treated patients showed a median overall survival (OS) of 13.9 months (95% CI, 10.6-17.2) and 8.2 months (95% CI, 7.5-9.0), respectively (hazard ratio [HR], 0.408; P = .001), with OS improvements favoring HAIC-FO vs sorafenib in even high-risk patients (10.8 months vs 5.7 months; HR, 0.343; P < .001). Grade 3/4 adverse events were more frequent with sorafenib vs HAIC-FO (48.1% vs20.3%).
Study details: The data come from the open-label, phase 3 FOHAIC-1 trial, which included 262 systemic therapy-naive patients with locally advanced or unresectable HCC who were randomly assigned to receive either HAIC-FO (n=130) or sorafenib (n=132).
Disclosures: The National Natural Science Foundation of China sponsored the study. The authors did not report any potential conflict of interests.
Source: Lyu N et al. J Clin Oncol. 2021 Dec 14. doi: 10.1200/JCO.21.01963.
Key clinical point: Hepatic arterial infusion chemotherapy of infusional fluorouracil, leucovorin, and oxaliplatin (HAIC-FO) is better than sorafenib at improving survival in patients with locally advanced hepatocellular carcinoma (HCC).
Main finding: At a median follow-up of 17.1 and 19.8 months, HAIC-FO- and sorafenib-treated patients showed a median overall survival (OS) of 13.9 months (95% CI, 10.6-17.2) and 8.2 months (95% CI, 7.5-9.0), respectively (hazard ratio [HR], 0.408; P = .001), with OS improvements favoring HAIC-FO vs sorafenib in even high-risk patients (10.8 months vs 5.7 months; HR, 0.343; P < .001). Grade 3/4 adverse events were more frequent with sorafenib vs HAIC-FO (48.1% vs20.3%).
Study details: The data come from the open-label, phase 3 FOHAIC-1 trial, which included 262 systemic therapy-naive patients with locally advanced or unresectable HCC who were randomly assigned to receive either HAIC-FO (n=130) or sorafenib (n=132).
Disclosures: The National Natural Science Foundation of China sponsored the study. The authors did not report any potential conflict of interests.
Source: Lyu N et al. J Clin Oncol. 2021 Dec 14. doi: 10.1200/JCO.21.01963.
Key clinical point: Hepatic arterial infusion chemotherapy of infusional fluorouracil, leucovorin, and oxaliplatin (HAIC-FO) is better than sorafenib at improving survival in patients with locally advanced hepatocellular carcinoma (HCC).
Main finding: At a median follow-up of 17.1 and 19.8 months, HAIC-FO- and sorafenib-treated patients showed a median overall survival (OS) of 13.9 months (95% CI, 10.6-17.2) and 8.2 months (95% CI, 7.5-9.0), respectively (hazard ratio [HR], 0.408; P = .001), with OS improvements favoring HAIC-FO vs sorafenib in even high-risk patients (10.8 months vs 5.7 months; HR, 0.343; P < .001). Grade 3/4 adverse events were more frequent with sorafenib vs HAIC-FO (48.1% vs20.3%).
Study details: The data come from the open-label, phase 3 FOHAIC-1 trial, which included 262 systemic therapy-naive patients with locally advanced or unresectable HCC who were randomly assigned to receive either HAIC-FO (n=130) or sorafenib (n=132).
Disclosures: The National Natural Science Foundation of China sponsored the study. The authors did not report any potential conflict of interests.
Source: Lyu N et al. J Clin Oncol. 2021 Dec 14. doi: 10.1200/JCO.21.01963.
Atezolizumab + bevacizumab shows long-term benefits over sorafenib for unresectable HCC
Key clinical point: Longer follow-up results confirm the survival benefits and consistent safety of first-line atezolizumab + bevacizumab vs sorafenib in patients with locally advanced or metastatic and/or unresectable hepatocellular carcinoma (HCC).
Major finding: After a 15.6-month median follow-up, the median overall survival (19.2 months vs 13.4 months; stratified hazard ratio [HR] for death, 0.66; P < .001) and progression-free survival (6.9 months vs 4.3 months; HR for death/progression, 0.65; P < .001) were higher with atezolizumab + bevacizumab vs sorafenib. Treatment-related grade 3/4 adverse events occurred in 43% vs 46% of patients receiving atezolizumab + bevacizumab vs sorafenib.
Study details: Findings are from a post hoc analysis of the phase 3 IMbrave150 trial including 501 treatment-naïve patients with locally advanced or metastatic and/or unresectable HCC. Patients were randomly assigned to atezolizumab + bevacizumab or sorafenib.
Disclosures: This study was funded by F. Hoffman-La Roche (FHLR)/Genentech. All investigators reported receiving financial or nonfinancial support, providing expert testimony, being an employee of, or holding shares/stocks in various pharmaceutical companies including FHLR/Genentech.
Source: Cheng AL et al. J Hepatol. 2021 Dec 10. doi: 10.1016/j.jhep.2021.11.030.
Key clinical point: Longer follow-up results confirm the survival benefits and consistent safety of first-line atezolizumab + bevacizumab vs sorafenib in patients with locally advanced or metastatic and/or unresectable hepatocellular carcinoma (HCC).
Major finding: After a 15.6-month median follow-up, the median overall survival (19.2 months vs 13.4 months; stratified hazard ratio [HR] for death, 0.66; P < .001) and progression-free survival (6.9 months vs 4.3 months; HR for death/progression, 0.65; P < .001) were higher with atezolizumab + bevacizumab vs sorafenib. Treatment-related grade 3/4 adverse events occurred in 43% vs 46% of patients receiving atezolizumab + bevacizumab vs sorafenib.
Study details: Findings are from a post hoc analysis of the phase 3 IMbrave150 trial including 501 treatment-naïve patients with locally advanced or metastatic and/or unresectable HCC. Patients were randomly assigned to atezolizumab + bevacizumab or sorafenib.
Disclosures: This study was funded by F. Hoffman-La Roche (FHLR)/Genentech. All investigators reported receiving financial or nonfinancial support, providing expert testimony, being an employee of, or holding shares/stocks in various pharmaceutical companies including FHLR/Genentech.
Source: Cheng AL et al. J Hepatol. 2021 Dec 10. doi: 10.1016/j.jhep.2021.11.030.
Key clinical point: Longer follow-up results confirm the survival benefits and consistent safety of first-line atezolizumab + bevacizumab vs sorafenib in patients with locally advanced or metastatic and/or unresectable hepatocellular carcinoma (HCC).
Major finding: After a 15.6-month median follow-up, the median overall survival (19.2 months vs 13.4 months; stratified hazard ratio [HR] for death, 0.66; P < .001) and progression-free survival (6.9 months vs 4.3 months; HR for death/progression, 0.65; P < .001) were higher with atezolizumab + bevacizumab vs sorafenib. Treatment-related grade 3/4 adverse events occurred in 43% vs 46% of patients receiving atezolizumab + bevacizumab vs sorafenib.
Study details: Findings are from a post hoc analysis of the phase 3 IMbrave150 trial including 501 treatment-naïve patients with locally advanced or metastatic and/or unresectable HCC. Patients were randomly assigned to atezolizumab + bevacizumab or sorafenib.
Disclosures: This study was funded by F. Hoffman-La Roche (FHLR)/Genentech. All investigators reported receiving financial or nonfinancial support, providing expert testimony, being an employee of, or holding shares/stocks in various pharmaceutical companies including FHLR/Genentech.
Source: Cheng AL et al. J Hepatol. 2021 Dec 10. doi: 10.1016/j.jhep.2021.11.030.
No survival benefits with first-line nivolumab vs sorafenib in advanced HCC
Key clinical point: First-line nivolumab treatment did not improve overall survival in patients with advanced hepatocellular carcinoma (HCC) compared with sorafenib.
Major finding: At a minimum follow-up of 22.8 months, nivolumab vs sorafenib did not meet the prespecified significance boundary for superior overall survival (16.4 months vs 14.7 months; hazard ratio, 0.85; P = .075).
Study details: Findings are from the phase 3 CheckMate 459 trial including 743 adult patients with advanced HCC randomly assigned to receive either nivolumab (n=371) or sorafenib (n=372).
Disclosures: This study was supported by Bristol Myers Squibb (BMS) and Ono Pharmaceutical. Some investigators including the lead author reported receiving grants and fees from, participation on data safety monitoring board or advisory boards for, owning stocks in, and being an employee of various pharmaceutical companies, including BMS and Ono Pharmaceutical.
Source: Yau T et al. Lancet Oncol. 2021 Dec 13. doi: 10.1016/S1470-2045(21)00604-5.
Key clinical point: First-line nivolumab treatment did not improve overall survival in patients with advanced hepatocellular carcinoma (HCC) compared with sorafenib.
Major finding: At a minimum follow-up of 22.8 months, nivolumab vs sorafenib did not meet the prespecified significance boundary for superior overall survival (16.4 months vs 14.7 months; hazard ratio, 0.85; P = .075).
Study details: Findings are from the phase 3 CheckMate 459 trial including 743 adult patients with advanced HCC randomly assigned to receive either nivolumab (n=371) or sorafenib (n=372).
Disclosures: This study was supported by Bristol Myers Squibb (BMS) and Ono Pharmaceutical. Some investigators including the lead author reported receiving grants and fees from, participation on data safety monitoring board or advisory boards for, owning stocks in, and being an employee of various pharmaceutical companies, including BMS and Ono Pharmaceutical.
Source: Yau T et al. Lancet Oncol. 2021 Dec 13. doi: 10.1016/S1470-2045(21)00604-5.
Key clinical point: First-line nivolumab treatment did not improve overall survival in patients with advanced hepatocellular carcinoma (HCC) compared with sorafenib.
Major finding: At a minimum follow-up of 22.8 months, nivolumab vs sorafenib did not meet the prespecified significance boundary for superior overall survival (16.4 months vs 14.7 months; hazard ratio, 0.85; P = .075).
Study details: Findings are from the phase 3 CheckMate 459 trial including 743 adult patients with advanced HCC randomly assigned to receive either nivolumab (n=371) or sorafenib (n=372).
Disclosures: This study was supported by Bristol Myers Squibb (BMS) and Ono Pharmaceutical. Some investigators including the lead author reported receiving grants and fees from, participation on data safety monitoring board or advisory boards for, owning stocks in, and being an employee of various pharmaceutical companies, including BMS and Ono Pharmaceutical.
Source: Yau T et al. Lancet Oncol. 2021 Dec 13. doi: 10.1016/S1470-2045(21)00604-5.
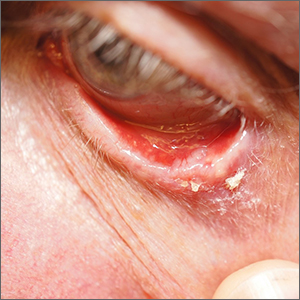
Growth on eyelid
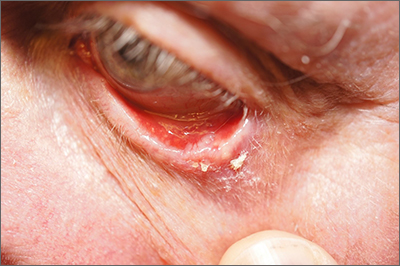
A shave biopsy (performed carefully to avoid caustic hemostatic agents irritating the conjunctiva) confirmed the diagnosis of a micronodular basal cell carcinoma (BCC).
BCC is a common tumor occurring on the eyelids and in the periocular region. Any new growing papule on the eyelids, history of focal bleeding, irritation, or focal loss of eyelashes should cause suspicion for BCC. Patients are often unaware of any symptoms when lesions begin, highlighting the importance of close inspection of the eyelids when skin or eye exams are performed. The differential diagnosis includes benign lesions such as hidrocystomas and nevi, as well as malignancies, including sebaceous carcinoma and squamous cell carcinoma.1
Factors that come into play when exploring eyelid BCC treatment options include tumor removal, eyelid function, and appearance. The potential morbidity associated with tumor spread in the periorbital region highlights the importance of early detection of eyelid cancers. Mohs micrographic surgery (MMS) is a first choice for tumor removal of an eyelid BCC and offers a high cure rate with minimal tissue removal.
Removal of an eyelid BCC may be a multidisciplinary endeavor with MMS achieving a clear margin, and Ophthalmology or Oculoplastics following with repair and closure soon after. Patients who can’t tolerate surgery should consider vismodegib, a targeted chemotherapy, or radiotherapy.
The patient in this case opted for a single staged excision and repair with Oculoplastics and has had no recurrence. He subsequently underwent a revision procedure to improve ectropion.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Shi Y, Jia R, Fan X. Ocular basal cell carcinoma: a brief literature review of clinical diagnosis and treatment. Onco Targets Ther. 2017;10:2483-2489. doi: 10.2147/OTT.S130371

A shave biopsy (performed carefully to avoid caustic hemostatic agents irritating the conjunctiva) confirmed the diagnosis of a micronodular basal cell carcinoma (BCC).
BCC is a common tumor occurring on the eyelids and in the periocular region. Any new growing papule on the eyelids, history of focal bleeding, irritation, or focal loss of eyelashes should cause suspicion for BCC. Patients are often unaware of any symptoms when lesions begin, highlighting the importance of close inspection of the eyelids when skin or eye exams are performed. The differential diagnosis includes benign lesions such as hidrocystomas and nevi, as well as malignancies, including sebaceous carcinoma and squamous cell carcinoma.1
Factors that come into play when exploring eyelid BCC treatment options include tumor removal, eyelid function, and appearance. The potential morbidity associated with tumor spread in the periorbital region highlights the importance of early detection of eyelid cancers. Mohs micrographic surgery (MMS) is a first choice for tumor removal of an eyelid BCC and offers a high cure rate with minimal tissue removal.
Removal of an eyelid BCC may be a multidisciplinary endeavor with MMS achieving a clear margin, and Ophthalmology or Oculoplastics following with repair and closure soon after. Patients who can’t tolerate surgery should consider vismodegib, a targeted chemotherapy, or radiotherapy.
The patient in this case opted for a single staged excision and repair with Oculoplastics and has had no recurrence. He subsequently underwent a revision procedure to improve ectropion.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

A shave biopsy (performed carefully to avoid caustic hemostatic agents irritating the conjunctiva) confirmed the diagnosis of a micronodular basal cell carcinoma (BCC).
BCC is a common tumor occurring on the eyelids and in the periocular region. Any new growing papule on the eyelids, history of focal bleeding, irritation, or focal loss of eyelashes should cause suspicion for BCC. Patients are often unaware of any symptoms when lesions begin, highlighting the importance of close inspection of the eyelids when skin or eye exams are performed. The differential diagnosis includes benign lesions such as hidrocystomas and nevi, as well as malignancies, including sebaceous carcinoma and squamous cell carcinoma.1
Factors that come into play when exploring eyelid BCC treatment options include tumor removal, eyelid function, and appearance. The potential morbidity associated with tumor spread in the periorbital region highlights the importance of early detection of eyelid cancers. Mohs micrographic surgery (MMS) is a first choice for tumor removal of an eyelid BCC and offers a high cure rate with minimal tissue removal.
Removal of an eyelid BCC may be a multidisciplinary endeavor with MMS achieving a clear margin, and Ophthalmology or Oculoplastics following with repair and closure soon after. Patients who can’t tolerate surgery should consider vismodegib, a targeted chemotherapy, or radiotherapy.
The patient in this case opted for a single staged excision and repair with Oculoplastics and has had no recurrence. He subsequently underwent a revision procedure to improve ectropion.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Shi Y, Jia R, Fan X. Ocular basal cell carcinoma: a brief literature review of clinical diagnosis and treatment. Onco Targets Ther. 2017;10:2483-2489. doi: 10.2147/OTT.S130371
1. Shi Y, Jia R, Fan X. Ocular basal cell carcinoma: a brief literature review of clinical diagnosis and treatment. Onco Targets Ther. 2017;10:2483-2489. doi: 10.2147/OTT.S130371
Chronic Lymphocytic Leukemia Updates From ASH 2021
Jennifer Brown, MD, PhD, from the Dana-Farber Cancer Institute, highlights findings from chronic lymphocytic leukemia (CLL) studies presented at the 2021 American Society of Hematology (ASH) Annual Meeting.
Dr Brown begins with several studies in the front-line setting. The CLL13 trial compared three venetoclax CD20 antibody regimens in young, fit patients. Most notably, obinutuzumab plus venetoclax demonstrated superiority over chemoimmunotherapy.
Next, Dr Brown shares results from the FLAIR trial, in which oral ibrutinib plus intravenous rituximab showed superior progression-free survival over oral fludarabine, oral cyclophosphamide, and intravenous rituximab (FCR).
She also discusses long-term results from a study of ibrutinib plus FCR in younger patients. The rate of undetectable minimal residual disease was sustained and the rate of complete remission increased compared with the initial analysis.
Dr Brown also reports that in the SEQUOIA trial, zanubrutinib demonstrated superiority in progression-free survival, even in high-risk subgroups.
In the relapsed/refractory setting, Dr Brown looks at the BRUIN study, in which pirtobrutinib demonstrated promising efficacy in patients who were previously treated with Bruton tyrosine kinase (BTK) inhibitors, as well as promising early data on the novel covalent inhibitor MK-1026.
Dr Brown concludes with a review of two studies of humoral and T-cell responses to COVID-19 vaccines in patients with CLL, which both underscored the importance of vaccinations, boosters, and follow-up doses in this group.
--
Jennifer Brown, MD, PhD, Worthington and Margaret Collette Professor of Medicine, Harvard Medical School; Institute Physician, Department of Medical Oncology, Dana-Farber Cancer Institute, Brigham & Women's Hospital, Boston, Massachusetts
Jennifer Brown, MD, PhD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; Acerta/AstraZeneca; BeiGene; Catapult; Genentech/Roche; Hutchmed; Janssen; MEI Pharma
Received research grant from: Gilead; Loxo/Lilly; TG Therapeutics; Verastem/SecuraBio
Jennifer Brown, MD, PhD, from the Dana-Farber Cancer Institute, highlights findings from chronic lymphocytic leukemia (CLL) studies presented at the 2021 American Society of Hematology (ASH) Annual Meeting.
Dr Brown begins with several studies in the front-line setting. The CLL13 trial compared three venetoclax CD20 antibody regimens in young, fit patients. Most notably, obinutuzumab plus venetoclax demonstrated superiority over chemoimmunotherapy.
Next, Dr Brown shares results from the FLAIR trial, in which oral ibrutinib plus intravenous rituximab showed superior progression-free survival over oral fludarabine, oral cyclophosphamide, and intravenous rituximab (FCR).
She also discusses long-term results from a study of ibrutinib plus FCR in younger patients. The rate of undetectable minimal residual disease was sustained and the rate of complete remission increased compared with the initial analysis.
Dr Brown also reports that in the SEQUOIA trial, zanubrutinib demonstrated superiority in progression-free survival, even in high-risk subgroups.
In the relapsed/refractory setting, Dr Brown looks at the BRUIN study, in which pirtobrutinib demonstrated promising efficacy in patients who were previously treated with Bruton tyrosine kinase (BTK) inhibitors, as well as promising early data on the novel covalent inhibitor MK-1026.
Dr Brown concludes with a review of two studies of humoral and T-cell responses to COVID-19 vaccines in patients with CLL, which both underscored the importance of vaccinations, boosters, and follow-up doses in this group.
--
Jennifer Brown, MD, PhD, Worthington and Margaret Collette Professor of Medicine, Harvard Medical School; Institute Physician, Department of Medical Oncology, Dana-Farber Cancer Institute, Brigham & Women's Hospital, Boston, Massachusetts
Jennifer Brown, MD, PhD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; Acerta/AstraZeneca; BeiGene; Catapult; Genentech/Roche; Hutchmed; Janssen; MEI Pharma
Received research grant from: Gilead; Loxo/Lilly; TG Therapeutics; Verastem/SecuraBio
Jennifer Brown, MD, PhD, from the Dana-Farber Cancer Institute, highlights findings from chronic lymphocytic leukemia (CLL) studies presented at the 2021 American Society of Hematology (ASH) Annual Meeting.
Dr Brown begins with several studies in the front-line setting. The CLL13 trial compared three venetoclax CD20 antibody regimens in young, fit patients. Most notably, obinutuzumab plus venetoclax demonstrated superiority over chemoimmunotherapy.
Next, Dr Brown shares results from the FLAIR trial, in which oral ibrutinib plus intravenous rituximab showed superior progression-free survival over oral fludarabine, oral cyclophosphamide, and intravenous rituximab (FCR).
She also discusses long-term results from a study of ibrutinib plus FCR in younger patients. The rate of undetectable minimal residual disease was sustained and the rate of complete remission increased compared with the initial analysis.
Dr Brown also reports that in the SEQUOIA trial, zanubrutinib demonstrated superiority in progression-free survival, even in high-risk subgroups.
In the relapsed/refractory setting, Dr Brown looks at the BRUIN study, in which pirtobrutinib demonstrated promising efficacy in patients who were previously treated with Bruton tyrosine kinase (BTK) inhibitors, as well as promising early data on the novel covalent inhibitor MK-1026.
Dr Brown concludes with a review of two studies of humoral and T-cell responses to COVID-19 vaccines in patients with CLL, which both underscored the importance of vaccinations, boosters, and follow-up doses in this group.
--
Jennifer Brown, MD, PhD, Worthington and Margaret Collette Professor of Medicine, Harvard Medical School; Institute Physician, Department of Medical Oncology, Dana-Farber Cancer Institute, Brigham & Women's Hospital, Boston, Massachusetts
Jennifer Brown, MD, PhD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; Acerta/AstraZeneca; BeiGene; Catapult; Genentech/Roche; Hutchmed; Janssen; MEI Pharma
Received research grant from: Gilead; Loxo/Lilly; TG Therapeutics; Verastem/SecuraBio

Non-Hodgkin Lymphoma Updates From ASH 2021
Brad Kahl, MD, shares results from non-Hodgkin lymphoma clinical trials that were presented at the 2021 American Society of Hematology (ASH) Annual Meeting.
Dr Kahl looks first at a frontline study examining a new combination therapy. The POLARIX study compared polatuzumab vedotin plus rituximab, cyclophosphamide, doxorubicin, and prednisone (Pola-R-CHP) with standard of care in patients with untreated diffuse large B-cell lymphoma (DLBCL). Pola-R-CHP demonstrated significant improvement in progression-free survival.
In relapsed/refractory non-Hodgkin lymphoma, Dr Kahl highlights several studies in chimeric antigen receptor (CAR) T-cell therapy. He starts with a primary analysis of the ZUMA-7 trial, in which axicabtagene ciloleucel (axi-cel) demonstrated improved survival compared with standard of care in patients with relapsed/refractory DLBCL.
Next, he reports on the TRANSFORM study, which compared lisocabtagene maraleucel (liso-cel) with standard of care in the second-line setting for patients with high-risk relapsed/refractory DLBCL. Liso-cel demonstrated favorable outcomes, with improved event-free survival and no new safety concerns.
The third CAR T-cell study he discusses is an updated analysis from ZUMA-5 that shows longer-term data for axi-cel in patients with relapsed/refractory follicular lymphoma or marginal zone lymphoma. Consistent with the primary analysis, this study demonstrated positive survival and safety outcomes in both groups.
Finally, Dr Kahl examines a phase 1/2 study of mosunetuzumab monotherapy for patients with relapsed/refractory follicular lymphoma who have received at least two lines of therapy. The study demonstrated improved response rates and favorable safety results.
--
Brad Kahl, MD, Professor of Medicine, Department of Medical Oncology; Director, Lymphoma Program, Washington University School of Medicine, St. Louis, Missouri
Brad Kahl, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; ADC Therapeutics; AstraZeneca; BeiGene; Celgene: Epizyme; Genentech; Pharmacyclics; Roche; TG Therapeutics
Received income in an amount equal to or greater than $250 from: Genentech; AbbVie; Janssen
Brad Kahl, MD, shares results from non-Hodgkin lymphoma clinical trials that were presented at the 2021 American Society of Hematology (ASH) Annual Meeting.
Dr Kahl looks first at a frontline study examining a new combination therapy. The POLARIX study compared polatuzumab vedotin plus rituximab, cyclophosphamide, doxorubicin, and prednisone (Pola-R-CHP) with standard of care in patients with untreated diffuse large B-cell lymphoma (DLBCL). Pola-R-CHP demonstrated significant improvement in progression-free survival.
In relapsed/refractory non-Hodgkin lymphoma, Dr Kahl highlights several studies in chimeric antigen receptor (CAR) T-cell therapy. He starts with a primary analysis of the ZUMA-7 trial, in which axicabtagene ciloleucel (axi-cel) demonstrated improved survival compared with standard of care in patients with relapsed/refractory DLBCL.
Next, he reports on the TRANSFORM study, which compared lisocabtagene maraleucel (liso-cel) with standard of care in the second-line setting for patients with high-risk relapsed/refractory DLBCL. Liso-cel demonstrated favorable outcomes, with improved event-free survival and no new safety concerns.
The third CAR T-cell study he discusses is an updated analysis from ZUMA-5 that shows longer-term data for axi-cel in patients with relapsed/refractory follicular lymphoma or marginal zone lymphoma. Consistent with the primary analysis, this study demonstrated positive survival and safety outcomes in both groups.
Finally, Dr Kahl examines a phase 1/2 study of mosunetuzumab monotherapy for patients with relapsed/refractory follicular lymphoma who have received at least two lines of therapy. The study demonstrated improved response rates and favorable safety results.
--
Brad Kahl, MD, Professor of Medicine, Department of Medical Oncology; Director, Lymphoma Program, Washington University School of Medicine, St. Louis, Missouri
Brad Kahl, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; ADC Therapeutics; AstraZeneca; BeiGene; Celgene: Epizyme; Genentech; Pharmacyclics; Roche; TG Therapeutics
Received income in an amount equal to or greater than $250 from: Genentech; AbbVie; Janssen
Brad Kahl, MD, shares results from non-Hodgkin lymphoma clinical trials that were presented at the 2021 American Society of Hematology (ASH) Annual Meeting.
Dr Kahl looks first at a frontline study examining a new combination therapy. The POLARIX study compared polatuzumab vedotin plus rituximab, cyclophosphamide, doxorubicin, and prednisone (Pola-R-CHP) with standard of care in patients with untreated diffuse large B-cell lymphoma (DLBCL). Pola-R-CHP demonstrated significant improvement in progression-free survival.
In relapsed/refractory non-Hodgkin lymphoma, Dr Kahl highlights several studies in chimeric antigen receptor (CAR) T-cell therapy. He starts with a primary analysis of the ZUMA-7 trial, in which axicabtagene ciloleucel (axi-cel) demonstrated improved survival compared with standard of care in patients with relapsed/refractory DLBCL.
Next, he reports on the TRANSFORM study, which compared lisocabtagene maraleucel (liso-cel) with standard of care in the second-line setting for patients with high-risk relapsed/refractory DLBCL. Liso-cel demonstrated favorable outcomes, with improved event-free survival and no new safety concerns.
The third CAR T-cell study he discusses is an updated analysis from ZUMA-5 that shows longer-term data for axi-cel in patients with relapsed/refractory follicular lymphoma or marginal zone lymphoma. Consistent with the primary analysis, this study demonstrated positive survival and safety outcomes in both groups.
Finally, Dr Kahl examines a phase 1/2 study of mosunetuzumab monotherapy for patients with relapsed/refractory follicular lymphoma who have received at least two lines of therapy. The study demonstrated improved response rates and favorable safety results.
--
Brad Kahl, MD, Professor of Medicine, Department of Medical Oncology; Director, Lymphoma Program, Washington University School of Medicine, St. Louis, Missouri
Brad Kahl, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; ADC Therapeutics; AstraZeneca; BeiGene; Celgene: Epizyme; Genentech; Pharmacyclics; Roche; TG Therapeutics
Received income in an amount equal to or greater than $250 from: Genentech; AbbVie; Janssen

Hodgkin Lymphoma Updates From ASH 2021
Ian Flinn, MD, PhD, director of lymphoma research at the Sarah Cannon Cancer Institute, highlights findings in Hodgkin lymphoma presented at the 2021 meeting of the American Society of Hematology.
The first study that Dr Flinn discusses combined pembrolizumab with AVD in patients with untreated Hodgkin lymphoma. This regimen was found to be both safe and effective, with high overall response, progression-free survival, and overall survival rates.
Another study he examines combined pembrolizumab with ICE chemotherapy prior to autologous hematologic stem cell transplant. The primary endpoint of complete metabolic response was met and the regimen demonstrated a tolerable safety profile.
Next, Dr Flinn looks at a study comparing seven novel and conventional salvage therapies. Most notably, brentuximab vedotin plus nivolumab demonstrated a higher complete remission rate and better post-autologous stem cell transplant progression-free survival compared with chemotherapy.
Dr Flinn concludes with a phase 2 study that evaluated first-line nivolumab in older frail patients. The primary objective of complete metabolic response rate was not reached, with safety findings comparable to those of published data in this population.
--
Ian W. Flinn, MD, PhD, Director, Lymphoma Research; Principal Investigator, Sarah Cannon Cancer Institute, Nashville, Tennessee
Ian W. Flinn, MD, PhD, has disclosed the following relevant financial relationships:
Serve(d) as a consultant: AbbVie; AstraZeneca; BeiGene; Century Therapeutics; Genentech; Gilead Sciences; Great Point Partners; Hutchison MediPharma; Iksuda Therapeutics; Janssen; Juno Therapeutics; Kite Pharma; MorphoSys; Novartis; Nurix Therapeutics; Pharmacyclics; Roche; Seattle Genetics; Takeda; TG Therapeutics; Unum Therapeutics; Verastem; Vincerx Pharma; Yingli Pharmaceuticals
Institute received research grant from: AbbVie; Acerta Pharma; Agios; ArQule; AstraZeneca; BeiGene; Calithera Biosciences; Celgene; Constellation Pharmaceuticals; Curis; Forma Therapeutics; Forty Seven; Genentech; Gilead Sciences; IGM Biosciences; Incyte; Infinity Pharmaceuticals; Janssen; Juno Therapeutics; Karyopharm Therapeutics; Kite Pharma; Loxo; Merck; MorphoSys; Novartis; Pfizer; Pharmacyclics; Portola Pharmaceuticals; Rhizen Pharmaceuticals; Roche; Seattle Genetics; Takeda; Teva Pharmaceuticals; TG Therapeutics; Trillium Therapeutics; Triphase Research & Development; Unum Therapeutics; Verastem
Ian Flinn, MD, PhD, director of lymphoma research at the Sarah Cannon Cancer Institute, highlights findings in Hodgkin lymphoma presented at the 2021 meeting of the American Society of Hematology.
The first study that Dr Flinn discusses combined pembrolizumab with AVD in patients with untreated Hodgkin lymphoma. This regimen was found to be both safe and effective, with high overall response, progression-free survival, and overall survival rates.
Another study he examines combined pembrolizumab with ICE chemotherapy prior to autologous hematologic stem cell transplant. The primary endpoint of complete metabolic response was met and the regimen demonstrated a tolerable safety profile.
Next, Dr Flinn looks at a study comparing seven novel and conventional salvage therapies. Most notably, brentuximab vedotin plus nivolumab demonstrated a higher complete remission rate and better post-autologous stem cell transplant progression-free survival compared with chemotherapy.
Dr Flinn concludes with a phase 2 study that evaluated first-line nivolumab in older frail patients. The primary objective of complete metabolic response rate was not reached, with safety findings comparable to those of published data in this population.
--
Ian W. Flinn, MD, PhD, Director, Lymphoma Research; Principal Investigator, Sarah Cannon Cancer Institute, Nashville, Tennessee
Ian W. Flinn, MD, PhD, has disclosed the following relevant financial relationships:
Serve(d) as a consultant: AbbVie; AstraZeneca; BeiGene; Century Therapeutics; Genentech; Gilead Sciences; Great Point Partners; Hutchison MediPharma; Iksuda Therapeutics; Janssen; Juno Therapeutics; Kite Pharma; MorphoSys; Novartis; Nurix Therapeutics; Pharmacyclics; Roche; Seattle Genetics; Takeda; TG Therapeutics; Unum Therapeutics; Verastem; Vincerx Pharma; Yingli Pharmaceuticals
Institute received research grant from: AbbVie; Acerta Pharma; Agios; ArQule; AstraZeneca; BeiGene; Calithera Biosciences; Celgene; Constellation Pharmaceuticals; Curis; Forma Therapeutics; Forty Seven; Genentech; Gilead Sciences; IGM Biosciences; Incyte; Infinity Pharmaceuticals; Janssen; Juno Therapeutics; Karyopharm Therapeutics; Kite Pharma; Loxo; Merck; MorphoSys; Novartis; Pfizer; Pharmacyclics; Portola Pharmaceuticals; Rhizen Pharmaceuticals; Roche; Seattle Genetics; Takeda; Teva Pharmaceuticals; TG Therapeutics; Trillium Therapeutics; Triphase Research & Development; Unum Therapeutics; Verastem
Ian Flinn, MD, PhD, director of lymphoma research at the Sarah Cannon Cancer Institute, highlights findings in Hodgkin lymphoma presented at the 2021 meeting of the American Society of Hematology.
The first study that Dr Flinn discusses combined pembrolizumab with AVD in patients with untreated Hodgkin lymphoma. This regimen was found to be both safe and effective, with high overall response, progression-free survival, and overall survival rates.
Another study he examines combined pembrolizumab with ICE chemotherapy prior to autologous hematologic stem cell transplant. The primary endpoint of complete metabolic response was met and the regimen demonstrated a tolerable safety profile.
Next, Dr Flinn looks at a study comparing seven novel and conventional salvage therapies. Most notably, brentuximab vedotin plus nivolumab demonstrated a higher complete remission rate and better post-autologous stem cell transplant progression-free survival compared with chemotherapy.
Dr Flinn concludes with a phase 2 study that evaluated first-line nivolumab in older frail patients. The primary objective of complete metabolic response rate was not reached, with safety findings comparable to those of published data in this population.
--
Ian W. Flinn, MD, PhD, Director, Lymphoma Research; Principal Investigator, Sarah Cannon Cancer Institute, Nashville, Tennessee
Ian W. Flinn, MD, PhD, has disclosed the following relevant financial relationships:
Serve(d) as a consultant: AbbVie; AstraZeneca; BeiGene; Century Therapeutics; Genentech; Gilead Sciences; Great Point Partners; Hutchison MediPharma; Iksuda Therapeutics; Janssen; Juno Therapeutics; Kite Pharma; MorphoSys; Novartis; Nurix Therapeutics; Pharmacyclics; Roche; Seattle Genetics; Takeda; TG Therapeutics; Unum Therapeutics; Verastem; Vincerx Pharma; Yingli Pharmaceuticals
Institute received research grant from: AbbVie; Acerta Pharma; Agios; ArQule; AstraZeneca; BeiGene; Calithera Biosciences; Celgene; Constellation Pharmaceuticals; Curis; Forma Therapeutics; Forty Seven; Genentech; Gilead Sciences; IGM Biosciences; Incyte; Infinity Pharmaceuticals; Janssen; Juno Therapeutics; Karyopharm Therapeutics; Kite Pharma; Loxo; Merck; MorphoSys; Novartis; Pfizer; Pharmacyclics; Portola Pharmaceuticals; Rhizen Pharmaceuticals; Roche; Seattle Genetics; Takeda; Teva Pharmaceuticals; TG Therapeutics; Trillium Therapeutics; Triphase Research & Development; Unum Therapeutics; Verastem

Infectious disease pop quiz: Clinical challenge #9 for the ObGyn
For uncomplicated chlamydia infection in a pregnant woman, what is the most appropriate treatment?
Continue to the answer...
Uncomplicated chlamydia infection in a pregnant woman should be treated with a single 1,000-mg oral dose of azithromycin. An acceptable alternative is amoxicillin 500 mg orally 3 times daily for 7 days.
In a nonpregnant patient, doxycycline 100 mg orally twice daily for 7 days is also an appropriate alternative. However, doxycycline is relatively expensive and may not be well tolerated because of gastrointestinal adverse effects. (Workowski KA, Bolan GA. Sexually transmitted diseases treatment guidelines, 2015. MMWR Morbid Mortal Wkly Rep. 2015;64[RR3]:1-137.)
- Duff P. Maternal and perinatal infections: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al. Gabbe’s Obstetrics: Normal and Problem Pregnancies. 8th ed. Elsevier; 2021:1124-1146.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TJ, et al. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862-919.
For uncomplicated chlamydia infection in a pregnant woman, what is the most appropriate treatment?
Continue to the answer...
Uncomplicated chlamydia infection in a pregnant woman should be treated with a single 1,000-mg oral dose of azithromycin. An acceptable alternative is amoxicillin 500 mg orally 3 times daily for 7 days.
In a nonpregnant patient, doxycycline 100 mg orally twice daily for 7 days is also an appropriate alternative. However, doxycycline is relatively expensive and may not be well tolerated because of gastrointestinal adverse effects. (Workowski KA, Bolan GA. Sexually transmitted diseases treatment guidelines, 2015. MMWR Morbid Mortal Wkly Rep. 2015;64[RR3]:1-137.)
For uncomplicated chlamydia infection in a pregnant woman, what is the most appropriate treatment?
Continue to the answer...
Uncomplicated chlamydia infection in a pregnant woman should be treated with a single 1,000-mg oral dose of azithromycin. An acceptable alternative is amoxicillin 500 mg orally 3 times daily for 7 days.
In a nonpregnant patient, doxycycline 100 mg orally twice daily for 7 days is also an appropriate alternative. However, doxycycline is relatively expensive and may not be well tolerated because of gastrointestinal adverse effects. (Workowski KA, Bolan GA. Sexually transmitted diseases treatment guidelines, 2015. MMWR Morbid Mortal Wkly Rep. 2015;64[RR3]:1-137.)
- Duff P. Maternal and perinatal infections: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al. Gabbe’s Obstetrics: Normal and Problem Pregnancies. 8th ed. Elsevier; 2021:1124-1146.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TJ, et al. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862-919.
- Duff P. Maternal and perinatal infections: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al. Gabbe’s Obstetrics: Normal and Problem Pregnancies. 8th ed. Elsevier; 2021:1124-1146.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TJ, et al. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862-919.