User login
Patient with tachycardia
On the basis of the patient's personal and family history together with his presentation, the likely diagnosis is latent autoimmune diabetes in adults (LADA). LADA is characterized by beta-cell loss and insulin resistance. This slowly evolving form of autoimmune diabetes comprises 2%-12% of all patients with adult-onset diabetes. Patients with LADA present with evidence of autoimmunity and varying C-peptide levels, which decrease more slowly in this subgroup than in patients with type 1 diabetes (T1D). They also have immunogenic markers associated with T1D, primarily anti-glutamic acid decarboxylase (GAD) antibodies.
Patients with LADA are often misdiagnosed as having T2D. The clinical picture of LADA overlaps with that of T2D, with patients being insulin resistant and often overweight. In addition, presenting symptoms of LADA — excessive thirst, blurred vision, and high blood glucose — are also seen in T2D. Although LADA is technically classified as T1D, some groups posit that the condition exists on a spectrum between T1D and T2D. Compared with patients with T2D, those with LADA are generally younger at diagnosis (often in their 30s), have lower BMI, and report a personal or family history of autoimmune diseases, such as the patient in this quiz. Throughout the disease course, individuals with LADA show a reduced frequency of metabolic syndrome compared with those with T2D.
Key to diagnosis is the absence of insulin requirement for at least 6 months. Anti-GAD antibodies are the most sensitive marker for LADA; other autoantibodies that occur less frequently include ICA, IA-2A, ZnT8A, and tetraspanin 7 autoantibodies. With a paucity of large-scale clinical trials in LADA, current treatment strategies are not based on consensus guidelines, though an expert panel has published management recommendations. Category 1 patients (defined as a C-peptide level < 0.3 nmol/L) are treated with intensive insulin therapy. The recommendation for category 2 patients (defined as C-peptide values ≥ 0.3 and ≤ 0.7 nmol/L) is a modified American Diabetes Association/European Association for the Study of Diabetes algorithm for T2D. However, patients with category 2 LADA may need to initiate insulin therapy earlier to combat beta-cell failure (ostensibly because LADA is an autoimmune disease beta-cell function declines much faster than in T2D). For category 3 patients (defined as C-peptide values > 0.7 nmol/L), treatment decisions are made in response to changing C-peptide levels.
Romesh K. Khardori, MD, PhD, Professor, Department of Internal Medicine, Division of Diabetes, Endocrine, and Metabolic Disorders, Eastern Virginia Medical School; EVMS Medical Group, Norfolk, Virginia.
Romesh K. Khardori, MD, PhD, has disclosed no relevant financial relationships.
On the basis of the patient's personal and family history together with his presentation, the likely diagnosis is latent autoimmune diabetes in adults (LADA). LADA is characterized by beta-cell loss and insulin resistance. This slowly evolving form of autoimmune diabetes comprises 2%-12% of all patients with adult-onset diabetes. Patients with LADA present with evidence of autoimmunity and varying C-peptide levels, which decrease more slowly in this subgroup than in patients with type 1 diabetes (T1D). They also have immunogenic markers associated with T1D, primarily anti-glutamic acid decarboxylase (GAD) antibodies.
Patients with LADA are often misdiagnosed as having T2D. The clinical picture of LADA overlaps with that of T2D, with patients being insulin resistant and often overweight. In addition, presenting symptoms of LADA — excessive thirst, blurred vision, and high blood glucose — are also seen in T2D. Although LADA is technically classified as T1D, some groups posit that the condition exists on a spectrum between T1D and T2D. Compared with patients with T2D, those with LADA are generally younger at diagnosis (often in their 30s), have lower BMI, and report a personal or family history of autoimmune diseases, such as the patient in this quiz. Throughout the disease course, individuals with LADA show a reduced frequency of metabolic syndrome compared with those with T2D.
Key to diagnosis is the absence of insulin requirement for at least 6 months. Anti-GAD antibodies are the most sensitive marker for LADA; other autoantibodies that occur less frequently include ICA, IA-2A, ZnT8A, and tetraspanin 7 autoantibodies. With a paucity of large-scale clinical trials in LADA, current treatment strategies are not based on consensus guidelines, though an expert panel has published management recommendations. Category 1 patients (defined as a C-peptide level < 0.3 nmol/L) are treated with intensive insulin therapy. The recommendation for category 2 patients (defined as C-peptide values ≥ 0.3 and ≤ 0.7 nmol/L) is a modified American Diabetes Association/European Association for the Study of Diabetes algorithm for T2D. However, patients with category 2 LADA may need to initiate insulin therapy earlier to combat beta-cell failure (ostensibly because LADA is an autoimmune disease beta-cell function declines much faster than in T2D). For category 3 patients (defined as C-peptide values > 0.7 nmol/L), treatment decisions are made in response to changing C-peptide levels.
Romesh K. Khardori, MD, PhD, Professor, Department of Internal Medicine, Division of Diabetes, Endocrine, and Metabolic Disorders, Eastern Virginia Medical School; EVMS Medical Group, Norfolk, Virginia.
Romesh K. Khardori, MD, PhD, has disclosed no relevant financial relationships.
On the basis of the patient's personal and family history together with his presentation, the likely diagnosis is latent autoimmune diabetes in adults (LADA). LADA is characterized by beta-cell loss and insulin resistance. This slowly evolving form of autoimmune diabetes comprises 2%-12% of all patients with adult-onset diabetes. Patients with LADA present with evidence of autoimmunity and varying C-peptide levels, which decrease more slowly in this subgroup than in patients with type 1 diabetes (T1D). They also have immunogenic markers associated with T1D, primarily anti-glutamic acid decarboxylase (GAD) antibodies.
Patients with LADA are often misdiagnosed as having T2D. The clinical picture of LADA overlaps with that of T2D, with patients being insulin resistant and often overweight. In addition, presenting symptoms of LADA — excessive thirst, blurred vision, and high blood glucose — are also seen in T2D. Although LADA is technically classified as T1D, some groups posit that the condition exists on a spectrum between T1D and T2D. Compared with patients with T2D, those with LADA are generally younger at diagnosis (often in their 30s), have lower BMI, and report a personal or family history of autoimmune diseases, such as the patient in this quiz. Throughout the disease course, individuals with LADA show a reduced frequency of metabolic syndrome compared with those with T2D.
Key to diagnosis is the absence of insulin requirement for at least 6 months. Anti-GAD antibodies are the most sensitive marker for LADA; other autoantibodies that occur less frequently include ICA, IA-2A, ZnT8A, and tetraspanin 7 autoantibodies. With a paucity of large-scale clinical trials in LADA, current treatment strategies are not based on consensus guidelines, though an expert panel has published management recommendations. Category 1 patients (defined as a C-peptide level < 0.3 nmol/L) are treated with intensive insulin therapy. The recommendation for category 2 patients (defined as C-peptide values ≥ 0.3 and ≤ 0.7 nmol/L) is a modified American Diabetes Association/European Association for the Study of Diabetes algorithm for T2D. However, patients with category 2 LADA may need to initiate insulin therapy earlier to combat beta-cell failure (ostensibly because LADA is an autoimmune disease beta-cell function declines much faster than in T2D). For category 3 patients (defined as C-peptide values > 0.7 nmol/L), treatment decisions are made in response to changing C-peptide levels.
Romesh K. Khardori, MD, PhD, Professor, Department of Internal Medicine, Division of Diabetes, Endocrine, and Metabolic Disorders, Eastern Virginia Medical School; EVMS Medical Group, Norfolk, Virginia.
Romesh K. Khardori, MD, PhD, has disclosed no relevant financial relationships.
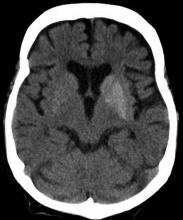
A 33-year-old man presents with blurred vision and tachycardia. Physical examination is remarkable for a BMI of 27 kg/m2. The patient explains that he feels he has lost weight. However, he attributes this change to a new exercise regimen he undertook when he was diagnosed with type 2 diabetes (T2D) about 8 months ago. The patient also notes polydipsia over a series of weeks. He reports that his first cousin may have lupus, though her diagnosis is still uncertain. Axial noncontrast CT demonstrates hyperattenuation that is more pronounced on the left side and involves the lentiform and caudate nuclei bilaterally.
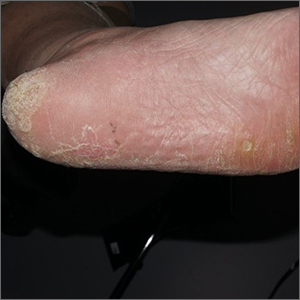
Plaque on heel
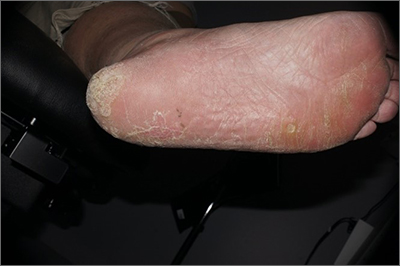
Physical exam revealed a plaque with multiple verrucous projections and clusters of smaller circular papules, all with associated thrombosed vessels. The plaque interrupted normal skin lines, consistent with a large, benign, plantar wart, also termed a mosaic wart when clusters of individual plantar warts form a single plaque.
Mosaic warts are caused by infection with human papillomavirus (HPV). They begin as individual papules or macules with a rough surface and small pinpoint capillaries. Plantar warts can be painful if located over a weight-bearing area of the foot. Plantar warts spread by autoinoculation from microtrauma to the foot. Picking at the wart, having it rub against a shoe insert, or exposing it to contaminated surfaces (such as a shower floor) can lead to the wart’s spread. Usually, the diagnosis of a plantar wart is based on clinical examination, with the main differential including a corn or callus. However, rare instances of squamous cell carcinoma or arsenical keratoses can mimic a plantar wart.
Although plantar warts can resolve spontaneously over months or years, patients often seek treatment. Warts may require multiple treatments and various therapies. Common first-line therapies include over-the-counter (OTC) salicylic acid and cryotherapy. The list of other therapies is lengthy, with no single agent credited with high cure rates in well-controlled trials. These therapies include intralesional candida antigen, topical 5 fluorouracil, and topical imiquimod, among many others.
Salicylic acid is available in several forms including 40% acid pads that may be cut to size and applied daily to affected areas. These pads may need to be reinforced with tape to improve adherence. Salicylic acid is also available as a 17% paint-on formulation that can be applied daily, with or without occlusion. This treatment usually requires 2 to 3 months of daily application.
When treated in the office, cryotherapy with liquid nitrogen (LN2) is a first-line therapy, with a cure rate of approximately 65%—similar to that of OTC salicylic acid.1 Application of LN2 via a spray cannister every 2 to 4 weeks until clear is a common strategy. Freezing the area, letting it thaw, and repeating the freeze again in 1 sitting improves clearance. Pain from LN2 can be significant and not all patients can tolerate it. However, for a motivated patient, this can be more convenient than home treatments or a good option when home treatment has failed.
This patient chose cryotherapy, and his foot cleared completely after several rounds of in-office treatments.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Lipke MM. An armamentarium of wart treatments. Clin Med Res. 2006;4:273-293. doi: 10.3121/cmr.4.4.273

Physical exam revealed a plaque with multiple verrucous projections and clusters of smaller circular papules, all with associated thrombosed vessels. The plaque interrupted normal skin lines, consistent with a large, benign, plantar wart, also termed a mosaic wart when clusters of individual plantar warts form a single plaque.
Mosaic warts are caused by infection with human papillomavirus (HPV). They begin as individual papules or macules with a rough surface and small pinpoint capillaries. Plantar warts can be painful if located over a weight-bearing area of the foot. Plantar warts spread by autoinoculation from microtrauma to the foot. Picking at the wart, having it rub against a shoe insert, or exposing it to contaminated surfaces (such as a shower floor) can lead to the wart’s spread. Usually, the diagnosis of a plantar wart is based on clinical examination, with the main differential including a corn or callus. However, rare instances of squamous cell carcinoma or arsenical keratoses can mimic a plantar wart.
Although plantar warts can resolve spontaneously over months or years, patients often seek treatment. Warts may require multiple treatments and various therapies. Common first-line therapies include over-the-counter (OTC) salicylic acid and cryotherapy. The list of other therapies is lengthy, with no single agent credited with high cure rates in well-controlled trials. These therapies include intralesional candida antigen, topical 5 fluorouracil, and topical imiquimod, among many others.
Salicylic acid is available in several forms including 40% acid pads that may be cut to size and applied daily to affected areas. These pads may need to be reinforced with tape to improve adherence. Salicylic acid is also available as a 17% paint-on formulation that can be applied daily, with or without occlusion. This treatment usually requires 2 to 3 months of daily application.
When treated in the office, cryotherapy with liquid nitrogen (LN2) is a first-line therapy, with a cure rate of approximately 65%—similar to that of OTC salicylic acid.1 Application of LN2 via a spray cannister every 2 to 4 weeks until clear is a common strategy. Freezing the area, letting it thaw, and repeating the freeze again in 1 sitting improves clearance. Pain from LN2 can be significant and not all patients can tolerate it. However, for a motivated patient, this can be more convenient than home treatments or a good option when home treatment has failed.
This patient chose cryotherapy, and his foot cleared completely after several rounds of in-office treatments.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

Physical exam revealed a plaque with multiple verrucous projections and clusters of smaller circular papules, all with associated thrombosed vessels. The plaque interrupted normal skin lines, consistent with a large, benign, plantar wart, also termed a mosaic wart when clusters of individual plantar warts form a single plaque.
Mosaic warts are caused by infection with human papillomavirus (HPV). They begin as individual papules or macules with a rough surface and small pinpoint capillaries. Plantar warts can be painful if located over a weight-bearing area of the foot. Plantar warts spread by autoinoculation from microtrauma to the foot. Picking at the wart, having it rub against a shoe insert, or exposing it to contaminated surfaces (such as a shower floor) can lead to the wart’s spread. Usually, the diagnosis of a plantar wart is based on clinical examination, with the main differential including a corn or callus. However, rare instances of squamous cell carcinoma or arsenical keratoses can mimic a plantar wart.
Although plantar warts can resolve spontaneously over months or years, patients often seek treatment. Warts may require multiple treatments and various therapies. Common first-line therapies include over-the-counter (OTC) salicylic acid and cryotherapy. The list of other therapies is lengthy, with no single agent credited with high cure rates in well-controlled trials. These therapies include intralesional candida antigen, topical 5 fluorouracil, and topical imiquimod, among many others.
Salicylic acid is available in several forms including 40% acid pads that may be cut to size and applied daily to affected areas. These pads may need to be reinforced with tape to improve adherence. Salicylic acid is also available as a 17% paint-on formulation that can be applied daily, with or without occlusion. This treatment usually requires 2 to 3 months of daily application.
When treated in the office, cryotherapy with liquid nitrogen (LN2) is a first-line therapy, with a cure rate of approximately 65%—similar to that of OTC salicylic acid.1 Application of LN2 via a spray cannister every 2 to 4 weeks until clear is a common strategy. Freezing the area, letting it thaw, and repeating the freeze again in 1 sitting improves clearance. Pain from LN2 can be significant and not all patients can tolerate it. However, for a motivated patient, this can be more convenient than home treatments or a good option when home treatment has failed.
This patient chose cryotherapy, and his foot cleared completely after several rounds of in-office treatments.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Lipke MM. An armamentarium of wart treatments. Clin Med Res. 2006;4:273-293. doi: 10.3121/cmr.4.4.273
1. Lipke MM. An armamentarium of wart treatments. Clin Med Res. 2006;4:273-293. doi: 10.3121/cmr.4.4.273
The troubling trend of repackaging feminine hygiene products for the next generation
Feminine hygiene products have been commercially available for decades. They are commonly marketed to reduce odor or clean vaginal discharge and menses. Multiple formulas are available as topical washes, wipes, creams, sprays, powders, deodorants, and douches.1 Products on the market range from those used externally on the vulva, such as wipes and sprays, to liquid solutions used intravaginally, such as washes and douches.
Who uses feminine hygiene products?
According to a 2006 study, the majority of women who use douches started using them between age 15 and 19 years, but some women initiate this practice habit as early as age 10 to 14.1 Predictably, women who douche are more likely to perceive douche products as safe.1
Demographic data on douche utilization are mixed: Some studies show that there are no significant racial differences in douching practices,2 while others have found that Black and African American women are more likely to practice douching than White and Hispanic women.1,3 Studies have shown a significant difference in attitudes toward douching and knowledge of normal vaginal symptoms among US racial demographics, although this must be examined through the historical context of racism and the lens of medical anthropology.4
Women cite that common reasons they use feminine hygiene products are to feel clean, to control odor, and to use after menses and intercourse.1,2
Modern marketing approaches
From wipes to soaps to douches, feminine hygiene products often are advertised to promote “funk-free periods”5 and “freshness,” fostering an environment in which women and men develop unrealistic standards for what is considered normal genital odor and resulting in poor body image.6
Recently, Vagisil (Combe Incorporated) marketing efforts faced backlash from the ObGyn community for targeting younger populations with a specific product line for adolescents called OMV! In addition, attention has been drawn to VCF vaginal odor eliminating film (Apothecus Pharmaceutical Corp), small stamp-sized dissolving films that are placed in the vaginal canal in contact with the epithelium. This product has entered the market of feminine hygiene products accompanied by slogans of eliminating “feminine odor” and providing “confidence for women to be intimate.”
Continue to: Effects of feminine hygiene products on the vaginal microbiome...
Effects of feminine hygiene products on the vaginal microbiome
Frequent use of feminine hygiene products has been associated with recurrent vaginitis, bacterial vaginosis, and general irritation/itch,7,8 which can cause more discharge and odor. Ironically, this may result in women using the product more frequently since they often seek out these products to eliminate odor and discharge.1,2
The pH of the vagina changes during a woman’s lifetime, but in the reproductive years, the normal pH range is typically 3.8 to 4.4.9 This range allows for a normal vaginal flora to form with bacteria such as Lactobacillus species and Gardnerella vaginalis, while feminine hygiene products have a wide range of pH.9,10
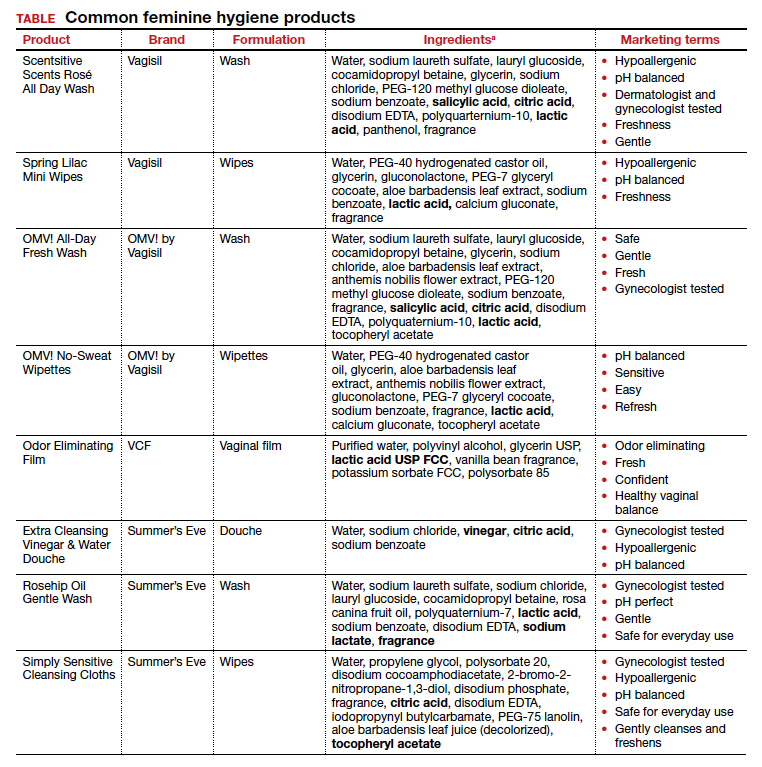
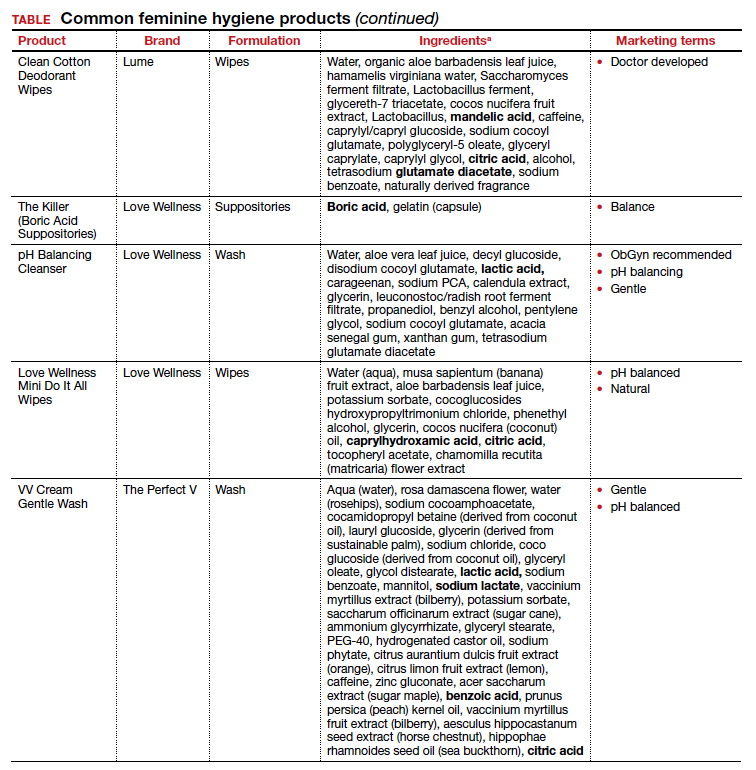
Regardless of the formulation, most feminine hygiene products contain ingredients and compositions that potentially are detrimental to the health of the vulva and vagina. Many products contain acidic ingredients, such as citric acid, lactic acid, and dehydroacetic acid, that can alter the vaginal pH and weaken the vaginal barrier by wiping out normal vaginal flora10 despite being advertised for use on “sensitive areas” (TABLE). Lactic acid also has been found to increase diverse anaerobic bacteria in the vaginal microbiome.11 Some feminine hygiene products have been shown to suppress Lactobacillus growth at 2 hours after use and to kill all lactobacilli at 24 hours.10 Shifts in microbiota numbers often occur when the vaginal pH has been altered, as is frequently the case with feminine hygiene products. In the absence of microbiome bacteria, the presence of vaginal hygiene products has been shown to increase interleukin-8 (IL-8), suggesting a proinflammatory reaction.10

A study in the United Kingdom found that women who used bubble bath, antiseptics, or douche products had a higher incidence of bacterial vaginosis compared with women who did not use such products.7 Women in Canada who used feminine hygiene products were more likely to report adverse conditions, including yeast infections, bacterial vaginosis, urinary tract infections, and sexually transmitted diseases.8 Furthermore, a significant association exists between vaginal douching and endometrial infection by bacterial vaginosis–associated organisms.12
Additionally, a study that analyzed volatile organic compound levels in the blood with the use of feminine hygiene products revealed a significant positive dose-exposure relationship between the frequency of vaginal douching in the last 6 months and concentrations of 1,4-dichloromethane, one of the volatile organic compounds.3 This points to the issue of not only disruption of pH and vaginal flora but also to the introduction of harmful substances that can further disrupt the vaginal barrier.
Understand the products to help educate patients
Use of feminine hygiene products is common among women. While women depend on the market to filter out products that are considered unsafe or may have harmful side effects,1 unfortunately that is not necessarily the case. With increasingly more feminine products on the market and the target demographic becoming younger, women of all ages are susceptible to misinformation that could affect their vaginal health long term.
It is vital that clinicians understand the topical effects of these products in order to properly educate and counsel patients. Ultimately, research on feminine hygiene products is limited and, as more products come to market, we must continue to reassess the effects of topical products on the vaginal epithelium and vulvar tissues. ●
- Grimley DM, Annang L, Foushee HR, et al. Vaginal douches and other feminine hygiene products: women’s practices and perceptions of product safety. Matern Child Health J. 2006;10:303-310. doi: 10.1007/s10995-005-0054-y.
- Foch BJ, McDaniel ND, Chacko MR. Racial differences in vaginal douching knowledge, attitude, and practices among sexually active adolescents. J Pediatr Adolesc Gynecol. 2001;14:29-33. doi: 10.1016/S1083-3188(00)00080-2.
- Lin N, Ding N, Meza-Wilson E, et al. Volatile organic compounds in feminine hygiene products sold in the US market: a survey of products and health risks. Environ Int. 2020;144:105740. doi: 10.1016/j.envint.2020.105740.
- Wayne State University Digital Commons. Guy-Lee AK. Rituals reproducing race: African American women’s feminine hygiene practices, shared experiences, and power. 2017. http://digitalcommons.wayne.edu/oa_dissertations/1806. Accessed December 13, 2021.
- YouTube. OMV! by Vagisil—Intimate care products designed by teens. July 10, 2020. www.youtube.com/ watch?v=VkVsCagrAw0. Accessed December 13, 2021.
- Jenkins A, O’Doherty KC. The clean vagina, the healthy vagina, and the dirty vagina: exploring women’s portrayals of the vagina in relation to vaginal cleansing product use. Fem Psychol. 2021;31:192-211. doi: 10.1177/0959353520944144.
- Rajamanoharan S, Low N, Jones SB, et al. Bacterial vaginosis, ethnicity, and the use of genital cleansing agents: a case control study. Sex Transm Dis. 1999;26:404-409.
- Crann SE, Cunningham S, Albert A, et al. Vaginal health and hygiene practices and product use in Canada: a national cross-sectional survey. BMC Womens Health. 2018;18:52. doi: 10.1186/s12905-018-0543-y.
- Chen Y, Bruning E, Rubino J, et al. Role of female intimate hygiene in vulvovaginal health: global hygiene practices and product usage. Womens Health (London). 2017;13:58-67. doi: 10.1177/1745505717731011.
- Fashemi B, Delaney ML, Onderdonk AB, et al. Effects of feminine hygiene products on the vaginal mucosal biome. Microb Ecol Health Dis. 2013;24. doi: 10.3402/mehd. v24i0.19703.
- Van der Veer C, Bruisten SM, Van Houdt R, et al. Effects of an over-the-counter lactic-acid containing intra-vaginal douching product on the vaginal microbiota. BMC Microbiol. 2019;19:168. doi: 10.1186/s12866-019-1545-0.
- Gondwe T, Ness R, Totten PA, et al. Novel bacterial vaginosis-associated organisms mediate the relationship between vaginal douching and pelvic inflammatory disease. Sex Transm Infect. 2020;96:439-444. doi: 10.1136/ sextrans-2019-054191.
Feminine hygiene products have been commercially available for decades. They are commonly marketed to reduce odor or clean vaginal discharge and menses. Multiple formulas are available as topical washes, wipes, creams, sprays, powders, deodorants, and douches.1 Products on the market range from those used externally on the vulva, such as wipes and sprays, to liquid solutions used intravaginally, such as washes and douches.
Who uses feminine hygiene products?
According to a 2006 study, the majority of women who use douches started using them between age 15 and 19 years, but some women initiate this practice habit as early as age 10 to 14.1 Predictably, women who douche are more likely to perceive douche products as safe.1
Demographic data on douche utilization are mixed: Some studies show that there are no significant racial differences in douching practices,2 while others have found that Black and African American women are more likely to practice douching than White and Hispanic women.1,3 Studies have shown a significant difference in attitudes toward douching and knowledge of normal vaginal symptoms among US racial demographics, although this must be examined through the historical context of racism and the lens of medical anthropology.4
Women cite that common reasons they use feminine hygiene products are to feel clean, to control odor, and to use after menses and intercourse.1,2
Modern marketing approaches
From wipes to soaps to douches, feminine hygiene products often are advertised to promote “funk-free periods”5 and “freshness,” fostering an environment in which women and men develop unrealistic standards for what is considered normal genital odor and resulting in poor body image.6
Recently, Vagisil (Combe Incorporated) marketing efforts faced backlash from the ObGyn community for targeting younger populations with a specific product line for adolescents called OMV! In addition, attention has been drawn to VCF vaginal odor eliminating film (Apothecus Pharmaceutical Corp), small stamp-sized dissolving films that are placed in the vaginal canal in contact with the epithelium. This product has entered the market of feminine hygiene products accompanied by slogans of eliminating “feminine odor” and providing “confidence for women to be intimate.”
Continue to: Effects of feminine hygiene products on the vaginal microbiome...
Effects of feminine hygiene products on the vaginal microbiome
Frequent use of feminine hygiene products has been associated with recurrent vaginitis, bacterial vaginosis, and general irritation/itch,7,8 which can cause more discharge and odor. Ironically, this may result in women using the product more frequently since they often seek out these products to eliminate odor and discharge.1,2
The pH of the vagina changes during a woman’s lifetime, but in the reproductive years, the normal pH range is typically 3.8 to 4.4.9 This range allows for a normal vaginal flora to form with bacteria such as Lactobacillus species and Gardnerella vaginalis, while feminine hygiene products have a wide range of pH.9,10
Regardless of the formulation, most feminine hygiene products contain ingredients and compositions that potentially are detrimental to the health of the vulva and vagina. Many products contain acidic ingredients, such as citric acid, lactic acid, and dehydroacetic acid, that can alter the vaginal pH and weaken the vaginal barrier by wiping out normal vaginal flora10 despite being advertised for use on “sensitive areas” (TABLE). Lactic acid also has been found to increase diverse anaerobic bacteria in the vaginal microbiome.11 Some feminine hygiene products have been shown to suppress Lactobacillus growth at 2 hours after use and to kill all lactobacilli at 24 hours.10 Shifts in microbiota numbers often occur when the vaginal pH has been altered, as is frequently the case with feminine hygiene products. In the absence of microbiome bacteria, the presence of vaginal hygiene products has been shown to increase interleukin-8 (IL-8), suggesting a proinflammatory reaction.10



A study in the United Kingdom found that women who used bubble bath, antiseptics, or douche products had a higher incidence of bacterial vaginosis compared with women who did not use such products.7 Women in Canada who used feminine hygiene products were more likely to report adverse conditions, including yeast infections, bacterial vaginosis, urinary tract infections, and sexually transmitted diseases.8 Furthermore, a significant association exists between vaginal douching and endometrial infection by bacterial vaginosis–associated organisms.12
Additionally, a study that analyzed volatile organic compound levels in the blood with the use of feminine hygiene products revealed a significant positive dose-exposure relationship between the frequency of vaginal douching in the last 6 months and concentrations of 1,4-dichloromethane, one of the volatile organic compounds.3 This points to the issue of not only disruption of pH and vaginal flora but also to the introduction of harmful substances that can further disrupt the vaginal barrier.
Understand the products to help educate patients
Use of feminine hygiene products is common among women. While women depend on the market to filter out products that are considered unsafe or may have harmful side effects,1 unfortunately that is not necessarily the case. With increasingly more feminine products on the market and the target demographic becoming younger, women of all ages are susceptible to misinformation that could affect their vaginal health long term.
It is vital that clinicians understand the topical effects of these products in order to properly educate and counsel patients. Ultimately, research on feminine hygiene products is limited and, as more products come to market, we must continue to reassess the effects of topical products on the vaginal epithelium and vulvar tissues. ●
Feminine hygiene products have been commercially available for decades. They are commonly marketed to reduce odor or clean vaginal discharge and menses. Multiple formulas are available as topical washes, wipes, creams, sprays, powders, deodorants, and douches.1 Products on the market range from those used externally on the vulva, such as wipes and sprays, to liquid solutions used intravaginally, such as washes and douches.
Who uses feminine hygiene products?
According to a 2006 study, the majority of women who use douches started using them between age 15 and 19 years, but some women initiate this practice habit as early as age 10 to 14.1 Predictably, women who douche are more likely to perceive douche products as safe.1
Demographic data on douche utilization are mixed: Some studies show that there are no significant racial differences in douching practices,2 while others have found that Black and African American women are more likely to practice douching than White and Hispanic women.1,3 Studies have shown a significant difference in attitudes toward douching and knowledge of normal vaginal symptoms among US racial demographics, although this must be examined through the historical context of racism and the lens of medical anthropology.4
Women cite that common reasons they use feminine hygiene products are to feel clean, to control odor, and to use after menses and intercourse.1,2
Modern marketing approaches
From wipes to soaps to douches, feminine hygiene products often are advertised to promote “funk-free periods”5 and “freshness,” fostering an environment in which women and men develop unrealistic standards for what is considered normal genital odor and resulting in poor body image.6
Recently, Vagisil (Combe Incorporated) marketing efforts faced backlash from the ObGyn community for targeting younger populations with a specific product line for adolescents called OMV! In addition, attention has been drawn to VCF vaginal odor eliminating film (Apothecus Pharmaceutical Corp), small stamp-sized dissolving films that are placed in the vaginal canal in contact with the epithelium. This product has entered the market of feminine hygiene products accompanied by slogans of eliminating “feminine odor” and providing “confidence for women to be intimate.”
Continue to: Effects of feminine hygiene products on the vaginal microbiome...
Effects of feminine hygiene products on the vaginal microbiome
Frequent use of feminine hygiene products has been associated with recurrent vaginitis, bacterial vaginosis, and general irritation/itch,7,8 which can cause more discharge and odor. Ironically, this may result in women using the product more frequently since they often seek out these products to eliminate odor and discharge.1,2
The pH of the vagina changes during a woman’s lifetime, but in the reproductive years, the normal pH range is typically 3.8 to 4.4.9 This range allows for a normal vaginal flora to form with bacteria such as Lactobacillus species and Gardnerella vaginalis, while feminine hygiene products have a wide range of pH.9,10
Regardless of the formulation, most feminine hygiene products contain ingredients and compositions that potentially are detrimental to the health of the vulva and vagina. Many products contain acidic ingredients, such as citric acid, lactic acid, and dehydroacetic acid, that can alter the vaginal pH and weaken the vaginal barrier by wiping out normal vaginal flora10 despite being advertised for use on “sensitive areas” (TABLE). Lactic acid also has been found to increase diverse anaerobic bacteria in the vaginal microbiome.11 Some feminine hygiene products have been shown to suppress Lactobacillus growth at 2 hours after use and to kill all lactobacilli at 24 hours.10 Shifts in microbiota numbers often occur when the vaginal pH has been altered, as is frequently the case with feminine hygiene products. In the absence of microbiome bacteria, the presence of vaginal hygiene products has been shown to increase interleukin-8 (IL-8), suggesting a proinflammatory reaction.10



A study in the United Kingdom found that women who used bubble bath, antiseptics, or douche products had a higher incidence of bacterial vaginosis compared with women who did not use such products.7 Women in Canada who used feminine hygiene products were more likely to report adverse conditions, including yeast infections, bacterial vaginosis, urinary tract infections, and sexually transmitted diseases.8 Furthermore, a significant association exists between vaginal douching and endometrial infection by bacterial vaginosis–associated organisms.12
Additionally, a study that analyzed volatile organic compound levels in the blood with the use of feminine hygiene products revealed a significant positive dose-exposure relationship between the frequency of vaginal douching in the last 6 months and concentrations of 1,4-dichloromethane, one of the volatile organic compounds.3 This points to the issue of not only disruption of pH and vaginal flora but also to the introduction of harmful substances that can further disrupt the vaginal barrier.
Understand the products to help educate patients
Use of feminine hygiene products is common among women. While women depend on the market to filter out products that are considered unsafe or may have harmful side effects,1 unfortunately that is not necessarily the case. With increasingly more feminine products on the market and the target demographic becoming younger, women of all ages are susceptible to misinformation that could affect their vaginal health long term.
It is vital that clinicians understand the topical effects of these products in order to properly educate and counsel patients. Ultimately, research on feminine hygiene products is limited and, as more products come to market, we must continue to reassess the effects of topical products on the vaginal epithelium and vulvar tissues. ●
- Grimley DM, Annang L, Foushee HR, et al. Vaginal douches and other feminine hygiene products: women’s practices and perceptions of product safety. Matern Child Health J. 2006;10:303-310. doi: 10.1007/s10995-005-0054-y.
- Foch BJ, McDaniel ND, Chacko MR. Racial differences in vaginal douching knowledge, attitude, and practices among sexually active adolescents. J Pediatr Adolesc Gynecol. 2001;14:29-33. doi: 10.1016/S1083-3188(00)00080-2.
- Lin N, Ding N, Meza-Wilson E, et al. Volatile organic compounds in feminine hygiene products sold in the US market: a survey of products and health risks. Environ Int. 2020;144:105740. doi: 10.1016/j.envint.2020.105740.
- Wayne State University Digital Commons. Guy-Lee AK. Rituals reproducing race: African American women’s feminine hygiene practices, shared experiences, and power. 2017. http://digitalcommons.wayne.edu/oa_dissertations/1806. Accessed December 13, 2021.
- YouTube. OMV! by Vagisil—Intimate care products designed by teens. July 10, 2020. www.youtube.com/ watch?v=VkVsCagrAw0. Accessed December 13, 2021.
- Jenkins A, O’Doherty KC. The clean vagina, the healthy vagina, and the dirty vagina: exploring women’s portrayals of the vagina in relation to vaginal cleansing product use. Fem Psychol. 2021;31:192-211. doi: 10.1177/0959353520944144.
- Rajamanoharan S, Low N, Jones SB, et al. Bacterial vaginosis, ethnicity, and the use of genital cleansing agents: a case control study. Sex Transm Dis. 1999;26:404-409.
- Crann SE, Cunningham S, Albert A, et al. Vaginal health and hygiene practices and product use in Canada: a national cross-sectional survey. BMC Womens Health. 2018;18:52. doi: 10.1186/s12905-018-0543-y.
- Chen Y, Bruning E, Rubino J, et al. Role of female intimate hygiene in vulvovaginal health: global hygiene practices and product usage. Womens Health (London). 2017;13:58-67. doi: 10.1177/1745505717731011.
- Fashemi B, Delaney ML, Onderdonk AB, et al. Effects of feminine hygiene products on the vaginal mucosal biome. Microb Ecol Health Dis. 2013;24. doi: 10.3402/mehd. v24i0.19703.
- Van der Veer C, Bruisten SM, Van Houdt R, et al. Effects of an over-the-counter lactic-acid containing intra-vaginal douching product on the vaginal microbiota. BMC Microbiol. 2019;19:168. doi: 10.1186/s12866-019-1545-0.
- Gondwe T, Ness R, Totten PA, et al. Novel bacterial vaginosis-associated organisms mediate the relationship between vaginal douching and pelvic inflammatory disease. Sex Transm Infect. 2020;96:439-444. doi: 10.1136/ sextrans-2019-054191.
- Grimley DM, Annang L, Foushee HR, et al. Vaginal douches and other feminine hygiene products: women’s practices and perceptions of product safety. Matern Child Health J. 2006;10:303-310. doi: 10.1007/s10995-005-0054-y.
- Foch BJ, McDaniel ND, Chacko MR. Racial differences in vaginal douching knowledge, attitude, and practices among sexually active adolescents. J Pediatr Adolesc Gynecol. 2001;14:29-33. doi: 10.1016/S1083-3188(00)00080-2.
- Lin N, Ding N, Meza-Wilson E, et al. Volatile organic compounds in feminine hygiene products sold in the US market: a survey of products and health risks. Environ Int. 2020;144:105740. doi: 10.1016/j.envint.2020.105740.
- Wayne State University Digital Commons. Guy-Lee AK. Rituals reproducing race: African American women’s feminine hygiene practices, shared experiences, and power. 2017. http://digitalcommons.wayne.edu/oa_dissertations/1806. Accessed December 13, 2021.
- YouTube. OMV! by Vagisil—Intimate care products designed by teens. July 10, 2020. www.youtube.com/ watch?v=VkVsCagrAw0. Accessed December 13, 2021.
- Jenkins A, O’Doherty KC. The clean vagina, the healthy vagina, and the dirty vagina: exploring women’s portrayals of the vagina in relation to vaginal cleansing product use. Fem Psychol. 2021;31:192-211. doi: 10.1177/0959353520944144.
- Rajamanoharan S, Low N, Jones SB, et al. Bacterial vaginosis, ethnicity, and the use of genital cleansing agents: a case control study. Sex Transm Dis. 1999;26:404-409.
- Crann SE, Cunningham S, Albert A, et al. Vaginal health and hygiene practices and product use in Canada: a national cross-sectional survey. BMC Womens Health. 2018;18:52. doi: 10.1186/s12905-018-0543-y.
- Chen Y, Bruning E, Rubino J, et al. Role of female intimate hygiene in vulvovaginal health: global hygiene practices and product usage. Womens Health (London). 2017;13:58-67. doi: 10.1177/1745505717731011.
- Fashemi B, Delaney ML, Onderdonk AB, et al. Effects of feminine hygiene products on the vaginal mucosal biome. Microb Ecol Health Dis. 2013;24. doi: 10.3402/mehd. v24i0.19703.
- Van der Veer C, Bruisten SM, Van Houdt R, et al. Effects of an over-the-counter lactic-acid containing intra-vaginal douching product on the vaginal microbiota. BMC Microbiol. 2019;19:168. doi: 10.1186/s12866-019-1545-0.
- Gondwe T, Ness R, Totten PA, et al. Novel bacterial vaginosis-associated organisms mediate the relationship between vaginal douching and pelvic inflammatory disease. Sex Transm Infect. 2020;96:439-444. doi: 10.1136/ sextrans-2019-054191.
Prior authorization abuse: It’s time for health insurance CEOs and their proxies to cease and desist the practice once and for all!
Before reading this editorial and concluding that the author (me) has lost his grip on reality, I would ask that you consider the facts I provide below and the ramifications incurred by your patients and practices, due to the misbehaviors adopted by the health insurance industry.
- Two of the most common issues discussed in today’s health care environment are revenue generation and provider/staff burnout.
While these issues are impacted by several factors, one of the most common denominators is increasing administrative workloads driven by non–revenue-generating activities. Consider this:
- A recent American Medical Association survey pointed out that during the course of the average workweek, a physician completes an average of 37 prior authorization requests. Physicians and their staff spend an average of 16.4 hours per week completing prior authorization requirements for patient medicines, procedures, and medical services that they may need.1
- While physicians report that about 65% of prior authorizations take only 1 day, they report that 26% take 3 or more days.2
The potential significance of the generated delays
While this may not seem like a long time (other than the impact it has on staff workload), consider the impact this can have on the patient if the medication being requested is: PrEP, the morning after pill, or other contraceptives? The consequences of the delay or denial could be a lifetime living with HIV, or an unintended pregnancy. This is to say nothing on the larger impact to family, partners, and the potential social stigma faced by all.
Beyond the personal costs and costs within your practice associated with the additional workload, consider the financial costs. The average cost to complete a prior authorization remains the single highest cost for the health care industry at $13.40 per manual transaction, and $7.19 per partially electronic web portal transaction,3 meaning that if I did only one prescription per week, I probably would not mind, but at $13.40 per prior authorization, this burden amounts to millions, actually $767 million by recent estimates.3 Additionally, if you factor in the number of denials and potential follow-ups, this creates a significant amount of waste and spending.
Ultimately, in my experience, I have found that most prior authorizations are simply unnecessary. Here, I’ve picked key examples from just my own recent experiences:
- My patient was denied access to a particular birth control pill she had been on successfully before, and my office was told she needed to try and fail on 5 different generic pills before she could be approved. However, the Affordable Care Act’s (ACA; aka Obamacare) Contraceptive Mandate requires coverage of all contraceptives determined to be most appropriate between a patient and their provider (see below).
- A menopausal patient was denied coverage twice (electronically) for generic micronized progesterone, and I was asked to write a letter of appeal because the insurance company wanted me to use medroxyprogesterone acetate instead. Polling my nearby retail independent pharmacy, the total cost difference per year was $19.96 savings/year ($47.01 ‒ $27.05 = $19.96). My pharmacist did note it could have been a different amount at a large chain pharmacy. Really? I had to write a letter, following two denials, to save less than $20, for a full year!
- A 78-year-old patient using Prolia for severe osteoporosis and preexisting fractures was delayed in getting her next Prolia injection due to a prior authorization snafu. She ended up with multiple additional fractures, a well-described effect of the increase in bone turnover when stopping or delaying this medication. She is now disabled.
- A 94-year-old patient was sent an email reminder to get the medical practice to authorize a refill of ileostomy bags. The email went to spam, and the patient ran out of bags prior to a holiday weekend. I got them in 2 days on Amazon Prime. But who emails a 94-year-old? And ileostomy bags! When does anyone stop needing ileostomy bags?
- I requested a prior authorization for Orilissa (clearly off label) because a severely progestogen-sensitive patient (augmented depression) with severe premenstrual dysphoric disorder requiring hospitalization was thought by her psychiatrist to be better off without menstrual periods. I completed the proper paperwork, two electronic appeals, and a letter of explanation including available references on the use of gonadotropin-releasing hormone analogues for such patients. I was then told I would need to have a peer-to-peer discussion, so I filled out that paperwork, which clearly noted that I am a board-certified reproductive endocrinologist. I got a phone call a few days later by a pleasant, young-sounding pediatric rheumatologist. Our interaction did not go well for him. This was not peer-to-peer!
Let us be clear, prior authorizations have nothing to do with patient care. In fact, they are solely about the money. We in ObGyn have mostly inexpensive and generic products, but even that fact has not lowered the excessive burden of the prior authorization process. In the case of contraception, whether you like the ACA or not it is the law, and it contains specific provisions regarding contraception. With the goals of providing broad access to patients and incentives to developers for new and novel contraceptive methods, these provisions require insurers to cover, without cost-sharing, women’s preventive services including the full range of FDA-approved contraceptives (currently 18 different method categories), and additional methods identified by the FDA as they become available. Further, providers must have an easily accessible, transparent, and sufficiently expedient exceptions process that is not unduly burdensome on the individual or a provider (or other individuals acting as a patient’s authorized representative).
And while I can regale you with chapter and verse and citations of the legal precedent and language, it boils down to this:
- The AMA reported that medical practices spend an average of 2 business days a week per physician to comply with health plans’ inefficient and overused prior-authorization protocols.4 To keep up with the administrative burden, 2 out of 5 physicians (40%) employ staff members who work exclusively on tasks associated with prior authorization.4
- About 86% of practices reported an increased burden of prior authorizations in the last 5 years.5
Continue to: What is to be done?
What is to be done?
I do have suggested solutions. Given the insurance industry’s complete lack of progress in voluntarily reducing the burdens of prior authorizations agreed to in their consensus statement with the AMA, American Hospital Association, America’s Health Insurance Plans, American Pharmacists Association, Blue Cross Blue Shield Association, and the Medical Group Management Association, I say, why not fine them? The AMA is calling on Congress to pass legislation that would codify much of the agreement, in which the above parties had already agreed that reforms were needed to reduce prior authorization burdens and enhance patient-centered care.6
A good model for enforcement via fines could be based on the old “incident to” rules of Medicare. These state that a physician needs to be “in the space” when advanced practice nurses or physician assistants see Medicare recipients. If they are not actually “in the space” they are subject to a fine. As a completely theoretical example, let’s say the claim was for $100. The practitioner would have to pay it back plus triple that amount in damages, or $400. They can also be fined up to $11,000 per claim and kick you out of Medicare and Medicaid. Take my example of Prolia from above…a single shot of Prolia is about $1,000. The insurer would theoretically have to pay $14,000/claim (the claim + triple damages + $11,000) if it was determined that the prior authorization was unnecessary. Seems about right to me. Or we could just sit the health insurance CEOs and their proxies in the corner on 2-foot-tall plastic Little Tikes® chairs for a “timeout” (dunce cap optional), like the outset of the article says.
Until the detrimental prior authorization process is challenged at all levels, we will continue to see and feel the effects of the harm it causes. Being able to drive change through advocacy and education is the best way we as clinicians can impact not just the future of health care but provide for the daily care of our patients who depend on and trust us to provide for their medical needs. We must be the impactors of change for ourselves, colleagues, staff, and profession if we are to really make advancements into the future.
Oh…and health insurance CEOs and their proxies, to get out of their “time-out” would still be entitled to one phone call to beg forgiveness from their mommies/daddies, priest/ rabbi/pastor, psychologist/psychiatrist/mystic healer, etc., but alas, the average wait time is an hour, and if anyone answers the phone, they have a grade school education used in following an irrelevant algorithm. ●
- Corder JC. Streamlining the insurance prior authorization debacle. Mo Med. 2018;115:312-314.
- Prior authorization hurdles have led to serious adverse events. American Medical Association website. February 5, 2019. https://www.ama-assn .org/press-center/press-releases/prior-author ization-hurdles-have-led-serious-adverse -events. Accessed November 29, 2021.
- Council for Affordable Quality Healthcare. 2020 CAQH INDEX. https://www.caqh.org/sites /default/files/explorations/index/2020-caqh -index.pdf. Accessed November 22, 2021.
- Most physicians had little relief from prior authorization as COVID cases soared. American Medical Association website. April 7, 2021. https:// www.ama-assn.org/press-center/press-releases /most-physicians-had-little-relief-prior-author ization-covid-cases. Accessed November 29, 2021.
- Robeznieks A. 1 in 4 doctors say prior authorization has led to a serious adverse event. American Medical Association website. February 5, 2019. https://www .ama-assn.org/practice-management/sustainability /1-4-doctors-say-prior-authorization-has-led-serious -adverse. Accessed November 29, 2021.
- Physicians call on Congress to address prior authorization reform. American Medical Association website. May 14, 2021. https://www .ama-assn.org/press-center/press-releases /physicians-call-congress-address-prior-author ization-reform. Accessed November 29, 2021.
Before reading this editorial and concluding that the author (me) has lost his grip on reality, I would ask that you consider the facts I provide below and the ramifications incurred by your patients and practices, due to the misbehaviors adopted by the health insurance industry.
- Two of the most common issues discussed in today’s health care environment are revenue generation and provider/staff burnout.
While these issues are impacted by several factors, one of the most common denominators is increasing administrative workloads driven by non–revenue-generating activities. Consider this:
- A recent American Medical Association survey pointed out that during the course of the average workweek, a physician completes an average of 37 prior authorization requests. Physicians and their staff spend an average of 16.4 hours per week completing prior authorization requirements for patient medicines, procedures, and medical services that they may need.1
- While physicians report that about 65% of prior authorizations take only 1 day, they report that 26% take 3 or more days.2
The potential significance of the generated delays
While this may not seem like a long time (other than the impact it has on staff workload), consider the impact this can have on the patient if the medication being requested is: PrEP, the morning after pill, or other contraceptives? The consequences of the delay or denial could be a lifetime living with HIV, or an unintended pregnancy. This is to say nothing on the larger impact to family, partners, and the potential social stigma faced by all.
Beyond the personal costs and costs within your practice associated with the additional workload, consider the financial costs. The average cost to complete a prior authorization remains the single highest cost for the health care industry at $13.40 per manual transaction, and $7.19 per partially electronic web portal transaction,3 meaning that if I did only one prescription per week, I probably would not mind, but at $13.40 per prior authorization, this burden amounts to millions, actually $767 million by recent estimates.3 Additionally, if you factor in the number of denials and potential follow-ups, this creates a significant amount of waste and spending.
Ultimately, in my experience, I have found that most prior authorizations are simply unnecessary. Here, I’ve picked key examples from just my own recent experiences:
- My patient was denied access to a particular birth control pill she had been on successfully before, and my office was told she needed to try and fail on 5 different generic pills before she could be approved. However, the Affordable Care Act’s (ACA; aka Obamacare) Contraceptive Mandate requires coverage of all contraceptives determined to be most appropriate between a patient and their provider (see below).
- A menopausal patient was denied coverage twice (electronically) for generic micronized progesterone, and I was asked to write a letter of appeal because the insurance company wanted me to use medroxyprogesterone acetate instead. Polling my nearby retail independent pharmacy, the total cost difference per year was $19.96 savings/year ($47.01 ‒ $27.05 = $19.96). My pharmacist did note it could have been a different amount at a large chain pharmacy. Really? I had to write a letter, following two denials, to save less than $20, for a full year!
- A 78-year-old patient using Prolia for severe osteoporosis and preexisting fractures was delayed in getting her next Prolia injection due to a prior authorization snafu. She ended up with multiple additional fractures, a well-described effect of the increase in bone turnover when stopping or delaying this medication. She is now disabled.
- A 94-year-old patient was sent an email reminder to get the medical practice to authorize a refill of ileostomy bags. The email went to spam, and the patient ran out of bags prior to a holiday weekend. I got them in 2 days on Amazon Prime. But who emails a 94-year-old? And ileostomy bags! When does anyone stop needing ileostomy bags?
- I requested a prior authorization for Orilissa (clearly off label) because a severely progestogen-sensitive patient (augmented depression) with severe premenstrual dysphoric disorder requiring hospitalization was thought by her psychiatrist to be better off without menstrual periods. I completed the proper paperwork, two electronic appeals, and a letter of explanation including available references on the use of gonadotropin-releasing hormone analogues for such patients. I was then told I would need to have a peer-to-peer discussion, so I filled out that paperwork, which clearly noted that I am a board-certified reproductive endocrinologist. I got a phone call a few days later by a pleasant, young-sounding pediatric rheumatologist. Our interaction did not go well for him. This was not peer-to-peer!
Let us be clear, prior authorizations have nothing to do with patient care. In fact, they are solely about the money. We in ObGyn have mostly inexpensive and generic products, but even that fact has not lowered the excessive burden of the prior authorization process. In the case of contraception, whether you like the ACA or not it is the law, and it contains specific provisions regarding contraception. With the goals of providing broad access to patients and incentives to developers for new and novel contraceptive methods, these provisions require insurers to cover, without cost-sharing, women’s preventive services including the full range of FDA-approved contraceptives (currently 18 different method categories), and additional methods identified by the FDA as they become available. Further, providers must have an easily accessible, transparent, and sufficiently expedient exceptions process that is not unduly burdensome on the individual or a provider (or other individuals acting as a patient’s authorized representative).
And while I can regale you with chapter and verse and citations of the legal precedent and language, it boils down to this:
- The AMA reported that medical practices spend an average of 2 business days a week per physician to comply with health plans’ inefficient and overused prior-authorization protocols.4 To keep up with the administrative burden, 2 out of 5 physicians (40%) employ staff members who work exclusively on tasks associated with prior authorization.4
- About 86% of practices reported an increased burden of prior authorizations in the last 5 years.5
Continue to: What is to be done?
What is to be done?
I do have suggested solutions. Given the insurance industry’s complete lack of progress in voluntarily reducing the burdens of prior authorizations agreed to in their consensus statement with the AMA, American Hospital Association, America’s Health Insurance Plans, American Pharmacists Association, Blue Cross Blue Shield Association, and the Medical Group Management Association, I say, why not fine them? The AMA is calling on Congress to pass legislation that would codify much of the agreement, in which the above parties had already agreed that reforms were needed to reduce prior authorization burdens and enhance patient-centered care.6
A good model for enforcement via fines could be based on the old “incident to” rules of Medicare. These state that a physician needs to be “in the space” when advanced practice nurses or physician assistants see Medicare recipients. If they are not actually “in the space” they are subject to a fine. As a completely theoretical example, let’s say the claim was for $100. The practitioner would have to pay it back plus triple that amount in damages, or $400. They can also be fined up to $11,000 per claim and kick you out of Medicare and Medicaid. Take my example of Prolia from above…a single shot of Prolia is about $1,000. The insurer would theoretically have to pay $14,000/claim (the claim + triple damages + $11,000) if it was determined that the prior authorization was unnecessary. Seems about right to me. Or we could just sit the health insurance CEOs and their proxies in the corner on 2-foot-tall plastic Little Tikes® chairs for a “timeout” (dunce cap optional), like the outset of the article says.
Until the detrimental prior authorization process is challenged at all levels, we will continue to see and feel the effects of the harm it causes. Being able to drive change through advocacy and education is the best way we as clinicians can impact not just the future of health care but provide for the daily care of our patients who depend on and trust us to provide for their medical needs. We must be the impactors of change for ourselves, colleagues, staff, and profession if we are to really make advancements into the future.
Oh…and health insurance CEOs and their proxies, to get out of their “time-out” would still be entitled to one phone call to beg forgiveness from their mommies/daddies, priest/ rabbi/pastor, psychologist/psychiatrist/mystic healer, etc., but alas, the average wait time is an hour, and if anyone answers the phone, they have a grade school education used in following an irrelevant algorithm. ●
Before reading this editorial and concluding that the author (me) has lost his grip on reality, I would ask that you consider the facts I provide below and the ramifications incurred by your patients and practices, due to the misbehaviors adopted by the health insurance industry.
- Two of the most common issues discussed in today’s health care environment are revenue generation and provider/staff burnout.
While these issues are impacted by several factors, one of the most common denominators is increasing administrative workloads driven by non–revenue-generating activities. Consider this:
- A recent American Medical Association survey pointed out that during the course of the average workweek, a physician completes an average of 37 prior authorization requests. Physicians and their staff spend an average of 16.4 hours per week completing prior authorization requirements for patient medicines, procedures, and medical services that they may need.1
- While physicians report that about 65% of prior authorizations take only 1 day, they report that 26% take 3 or more days.2
The potential significance of the generated delays
While this may not seem like a long time (other than the impact it has on staff workload), consider the impact this can have on the patient if the medication being requested is: PrEP, the morning after pill, or other contraceptives? The consequences of the delay or denial could be a lifetime living with HIV, or an unintended pregnancy. This is to say nothing on the larger impact to family, partners, and the potential social stigma faced by all.
Beyond the personal costs and costs within your practice associated with the additional workload, consider the financial costs. The average cost to complete a prior authorization remains the single highest cost for the health care industry at $13.40 per manual transaction, and $7.19 per partially electronic web portal transaction,3 meaning that if I did only one prescription per week, I probably would not mind, but at $13.40 per prior authorization, this burden amounts to millions, actually $767 million by recent estimates.3 Additionally, if you factor in the number of denials and potential follow-ups, this creates a significant amount of waste and spending.
Ultimately, in my experience, I have found that most prior authorizations are simply unnecessary. Here, I’ve picked key examples from just my own recent experiences:
- My patient was denied access to a particular birth control pill she had been on successfully before, and my office was told she needed to try and fail on 5 different generic pills before she could be approved. However, the Affordable Care Act’s (ACA; aka Obamacare) Contraceptive Mandate requires coverage of all contraceptives determined to be most appropriate between a patient and their provider (see below).
- A menopausal patient was denied coverage twice (electronically) for generic micronized progesterone, and I was asked to write a letter of appeal because the insurance company wanted me to use medroxyprogesterone acetate instead. Polling my nearby retail independent pharmacy, the total cost difference per year was $19.96 savings/year ($47.01 ‒ $27.05 = $19.96). My pharmacist did note it could have been a different amount at a large chain pharmacy. Really? I had to write a letter, following two denials, to save less than $20, for a full year!
- A 78-year-old patient using Prolia for severe osteoporosis and preexisting fractures was delayed in getting her next Prolia injection due to a prior authorization snafu. She ended up with multiple additional fractures, a well-described effect of the increase in bone turnover when stopping or delaying this medication. She is now disabled.
- A 94-year-old patient was sent an email reminder to get the medical practice to authorize a refill of ileostomy bags. The email went to spam, and the patient ran out of bags prior to a holiday weekend. I got them in 2 days on Amazon Prime. But who emails a 94-year-old? And ileostomy bags! When does anyone stop needing ileostomy bags?
- I requested a prior authorization for Orilissa (clearly off label) because a severely progestogen-sensitive patient (augmented depression) with severe premenstrual dysphoric disorder requiring hospitalization was thought by her psychiatrist to be better off without menstrual periods. I completed the proper paperwork, two electronic appeals, and a letter of explanation including available references on the use of gonadotropin-releasing hormone analogues for such patients. I was then told I would need to have a peer-to-peer discussion, so I filled out that paperwork, which clearly noted that I am a board-certified reproductive endocrinologist. I got a phone call a few days later by a pleasant, young-sounding pediatric rheumatologist. Our interaction did not go well for him. This was not peer-to-peer!
Let us be clear, prior authorizations have nothing to do with patient care. In fact, they are solely about the money. We in ObGyn have mostly inexpensive and generic products, but even that fact has not lowered the excessive burden of the prior authorization process. In the case of contraception, whether you like the ACA or not it is the law, and it contains specific provisions regarding contraception. With the goals of providing broad access to patients and incentives to developers for new and novel contraceptive methods, these provisions require insurers to cover, without cost-sharing, women’s preventive services including the full range of FDA-approved contraceptives (currently 18 different method categories), and additional methods identified by the FDA as they become available. Further, providers must have an easily accessible, transparent, and sufficiently expedient exceptions process that is not unduly burdensome on the individual or a provider (or other individuals acting as a patient’s authorized representative).
And while I can regale you with chapter and verse and citations of the legal precedent and language, it boils down to this:
- The AMA reported that medical practices spend an average of 2 business days a week per physician to comply with health plans’ inefficient and overused prior-authorization protocols.4 To keep up with the administrative burden, 2 out of 5 physicians (40%) employ staff members who work exclusively on tasks associated with prior authorization.4
- About 86% of practices reported an increased burden of prior authorizations in the last 5 years.5
Continue to: What is to be done?
What is to be done?
I do have suggested solutions. Given the insurance industry’s complete lack of progress in voluntarily reducing the burdens of prior authorizations agreed to in their consensus statement with the AMA, American Hospital Association, America’s Health Insurance Plans, American Pharmacists Association, Blue Cross Blue Shield Association, and the Medical Group Management Association, I say, why not fine them? The AMA is calling on Congress to pass legislation that would codify much of the agreement, in which the above parties had already agreed that reforms were needed to reduce prior authorization burdens and enhance patient-centered care.6
A good model for enforcement via fines could be based on the old “incident to” rules of Medicare. These state that a physician needs to be “in the space” when advanced practice nurses or physician assistants see Medicare recipients. If they are not actually “in the space” they are subject to a fine. As a completely theoretical example, let’s say the claim was for $100. The practitioner would have to pay it back plus triple that amount in damages, or $400. They can also be fined up to $11,000 per claim and kick you out of Medicare and Medicaid. Take my example of Prolia from above…a single shot of Prolia is about $1,000. The insurer would theoretically have to pay $14,000/claim (the claim + triple damages + $11,000) if it was determined that the prior authorization was unnecessary. Seems about right to me. Or we could just sit the health insurance CEOs and their proxies in the corner on 2-foot-tall plastic Little Tikes® chairs for a “timeout” (dunce cap optional), like the outset of the article says.
Until the detrimental prior authorization process is challenged at all levels, we will continue to see and feel the effects of the harm it causes. Being able to drive change through advocacy and education is the best way we as clinicians can impact not just the future of health care but provide for the daily care of our patients who depend on and trust us to provide for their medical needs. We must be the impactors of change for ourselves, colleagues, staff, and profession if we are to really make advancements into the future.
Oh…and health insurance CEOs and their proxies, to get out of their “time-out” would still be entitled to one phone call to beg forgiveness from their mommies/daddies, priest/ rabbi/pastor, psychologist/psychiatrist/mystic healer, etc., but alas, the average wait time is an hour, and if anyone answers the phone, they have a grade school education used in following an irrelevant algorithm. ●
- Corder JC. Streamlining the insurance prior authorization debacle. Mo Med. 2018;115:312-314.
- Prior authorization hurdles have led to serious adverse events. American Medical Association website. February 5, 2019. https://www.ama-assn .org/press-center/press-releases/prior-author ization-hurdles-have-led-serious-adverse -events. Accessed November 29, 2021.
- Council for Affordable Quality Healthcare. 2020 CAQH INDEX. https://www.caqh.org/sites /default/files/explorations/index/2020-caqh -index.pdf. Accessed November 22, 2021.
- Most physicians had little relief from prior authorization as COVID cases soared. American Medical Association website. April 7, 2021. https:// www.ama-assn.org/press-center/press-releases /most-physicians-had-little-relief-prior-author ization-covid-cases. Accessed November 29, 2021.
- Robeznieks A. 1 in 4 doctors say prior authorization has led to a serious adverse event. American Medical Association website. February 5, 2019. https://www .ama-assn.org/practice-management/sustainability /1-4-doctors-say-prior-authorization-has-led-serious -adverse. Accessed November 29, 2021.
- Physicians call on Congress to address prior authorization reform. American Medical Association website. May 14, 2021. https://www .ama-assn.org/press-center/press-releases /physicians-call-congress-address-prior-author ization-reform. Accessed November 29, 2021.
- Corder JC. Streamlining the insurance prior authorization debacle. Mo Med. 2018;115:312-314.
- Prior authorization hurdles have led to serious adverse events. American Medical Association website. February 5, 2019. https://www.ama-assn .org/press-center/press-releases/prior-author ization-hurdles-have-led-serious-adverse -events. Accessed November 29, 2021.
- Council for Affordable Quality Healthcare. 2020 CAQH INDEX. https://www.caqh.org/sites /default/files/explorations/index/2020-caqh -index.pdf. Accessed November 22, 2021.
- Most physicians had little relief from prior authorization as COVID cases soared. American Medical Association website. April 7, 2021. https:// www.ama-assn.org/press-center/press-releases /most-physicians-had-little-relief-prior-author ization-covid-cases. Accessed November 29, 2021.
- Robeznieks A. 1 in 4 doctors say prior authorization has led to a serious adverse event. American Medical Association website. February 5, 2019. https://www .ama-assn.org/practice-management/sustainability /1-4-doctors-say-prior-authorization-has-led-serious -adverse. Accessed November 29, 2021.
- Physicians call on Congress to address prior authorization reform. American Medical Association website. May 14, 2021. https://www .ama-assn.org/press-center/press-releases /physicians-call-congress-address-prior-author ization-reform. Accessed November 29, 2021.
Atopic Dermatitis: Phenotypes
Clinical Edge Journal Scan Commentary: Migraine January 2022
Ferrari et al1 provided information on an open label extension to the “LIBERTY” study which investigated the use of erenumab in subjects with episodic migraine that have failed multiple prior preventive medications. The initial Calcitonin gene-related peptide (CGRP) monoclonal antibody (mAb) studies excluded more refractory patients. Most commercial insurances in the United States have a “step” policy that relates to use of these and other newer medications, meaning that the majority of patient in the US who receive these medications have previously tried other preventive medications. This raised the question whether migraine refractoriness is a negative predictive factor for erenumab.
This long-term open label study is more like the real-world use of erenumab, and as such the results are similar to what many practitioners are seeing in their clinical experience. Approximately 25% of subjects discontinued erenumab, mostly due to ineffectiveness. Adverse events were mild, and although erenumab has warnings for constipation and hypertension, this study did not show either as increasing over 2 years. Erenumab appeared to be tolerable over time. There were no newly noted safety signals in this study.
The efficacy of erenumab also appeared to be stable over time, without the development of tolerance to the medication. There is a slight decrease in the 50% responder rate at 2 years when these more refractory patients are compared to those that did not have multiple treatment failures. This study also looked at “functional parameters,” such as Migraine Disability Assessment (MIDAS) and Headache Impact Test (HIT-6), both of which were significantly improved over time.
Although there are some significant limitations in this study-primarily the fact that it is open label—this does give a more representative and real-world sample of patients who will be prescribed erenumab in the United States. Most practitioners will be glad to find that the long-term use of erenumab appears safe, and the efficacy remains stable, even in a more difficult-to-treat population.
A randomized controlled international study investigated the preventive use of occipital nerve blocks in migraine without aura.2 The majority of the literature for the use of occipital nerve blocks is for acute treatment, and arguably the most significant study prior to this was Friedman et al3 investigating the use of this procedure in the emergency ward. Prior occipital nerve studies have been inconclusive, and although occipital nerve blocks are considered standard of care for specific conditions in most headache centers, reimbursement is usually very limited. Insurance companies have quoted prior preventive occipital nerve studies to justify non-coverage of these procedures, making access to them for many patients very limited.
Occipital nerve blocks are not performed uniformly, both regarding the medications used—some practitioners use no steroids, some use lidocaine and bupivocaine—and regarding the placement of the injections. In this a small cohort study, 55 subjects were divided into four groups for intervention—one of which was a control group of saline—and all were given one 2.5 mL injection at a point in between the occipital protuberance and the mastoid process bilaterally. Due to adverse events (alopecia and cutaneous atrophy) in two of the triamcinolone groups, recruitment was halted for those two groups. Patients were assessed based on headache duration, frequency, and severity over a 4-week course.
Compared to baseline all interventional groups had significantly decreased headache severity, which did return closer to baseline during the final week. Headache duration was decreased in the first 2 weeks post-injection. Headache frequency was seen to return to baseline at week 4, but prior to that the groups injected with lidocaine had a significant decrease in migraine frequency, with an average decrease in headache days.
Occipital nerve blocks are performed frequently for migraine, occipital neuralgia, cervicogenic headache, and many other conditions with noted tenderness over the occiput. As noted above, they are not performed uniformly—sometimes they are given for acute headache pain or status migranosus, and other times they are used in regular intervals for prevention. This data does finally show a preventive benefit with occipital nerve blocks, and this may allow for modifications in how occipital nerve blocks are currently performed. Based on this study, if given preventively, occipital nerve blocks should only contain topical anesthetics, not steroids, and should be performed on an every 2-3 week basis.
The limitations of this study are significant as well. This is a very small cohort, and the injections were performed in only one manner (one bilateral injection), whereas many practitioners will target the greater and lesser branches of the occipital nerve individually. There were no exclusion criteria for subjects that already had occipital nerve blocks performed—those patients would be unblinded as there is a different sensation when injected with a topical anesthetic versus normal saline (normal saline does not cause burning subcutaneously).
These results should pave the way for further investigations in the use of occipital and other nerve blocks in the prevention of migraine. This should allow better access for our patients and the possibility of performing these procedures more uniformly in the future.
It can be challenging for many practitioners to determine which medication is ideal for individual situations. This is especially true when treating chronic migraine, where many potential complicating factors can influence positive to negative responses to treatment. The investigators here sought to determine which factors may potentially predict a positive response to galcanezumab.4
This is an observational study, where 156 subjects with a diagnosis of chronic migraine were enrolled. There was a 1-month run-in period where the following characteristics were collected: monthly headache days, monthly abortive medication intake, clinical features of migraine, and disability scores (MIDAS and HIT-6). These were tracked over a 3-month period after starting glacanezumab.
Approximately 40% of subjects experienced a 50% reduction in headache frequency. The better responders had a lower body mass index, fewer previously failed preventive medications, unilateral headache pain, and previous good response to triptan use. Surprisingly, the presence of medication overuse was associated with persistent improvement at 3 months as well, with over 60% of subjects with medication overuse no longer overusing acute medications at 3 months.
This study is helpful in identifying specific features that may allow a practitioner to better recommend CGRP mAb medications, such as galcanezumab. Chronic migraine can offer a challenge to even the best trained clinicians. Patients will often have multiple factors that have led to a conversion from episodic to chronic migraine, and a history of medication failures or intolerances. These patients are often referred specifically due to these challenges.
When deciding on a preventive medication for patients with chronic migraine, we often first consider which oral preventive medications may allow us to treat migraine in addition to another underlying issue—such as insomnia, depression, or hypertension. Although the oral class can improve other comorbidities, intolerance is significantly higher for most of these medications as well. The CGRP mAb class is somewhat more ideal for prevention of migraine; the focus when using this class is for migraine prevention alone, and the side effect profile is more tolerable for most patients. That said, if predictive factors were known a more individualized approach to migraine prevention would be possible.
The authors’ recognition of the factors associated with improvement in patients using glacanezumab allows this better individualization. Based on these results, patients with more unilateral pain, lower BMI, and good response to triptans could be recommended glacanuzumab with a great degree of confidence. This should be irrespective of even high frequency use of acute medications, as most of subjects in this study with medication overuse reverted after 3 months.
There is never a single ideal preventive or acute treatment for migraine in any population, however, recognizing factors that allow for an individualized approach improves the quality of life for our patients, and leaves them less disabled by migraine.
References
- Ferrari MD et al. Two-year efficacy and safety of erenumab in participants with episodic migraine and 2–4 prior preventive treatment failures: results from the LIBERTY study. J Neurol Neurosurg Psychiatry. 2021(Nov 29).
- Malekian N et al. Preventive effect of greater occipital nerve block on patients with episodic migraine: A randomized double‐blind placebo‐controlled clinical trial. Cephalalgia. 2021(Nov 17).
- Friedman BW et al. A Randomized, Sham-Controlled Trial of Bilateral Greater Occipital Nerve Blocks With Bupivacaine for Acute Migraine Patients Refractory to Standard Emergency Department Treatment With Metoclopramide. Headache. 2018(Oct);58(9):1427-34. https://doi.org/10.1111/head.13395.
- Vernieri F et al. Rapid response to galcanezumab and predictive factors in chronic migraine patients: A 3-month observational, longitudinal, cohort, multicenter, Italian real-life study. Eur J Neurol. 2021(Nov 26).
Ferrari et al1 provided information on an open label extension to the “LIBERTY” study which investigated the use of erenumab in subjects with episodic migraine that have failed multiple prior preventive medications. The initial Calcitonin gene-related peptide (CGRP) monoclonal antibody (mAb) studies excluded more refractory patients. Most commercial insurances in the United States have a “step” policy that relates to use of these and other newer medications, meaning that the majority of patient in the US who receive these medications have previously tried other preventive medications. This raised the question whether migraine refractoriness is a negative predictive factor for erenumab.
This long-term open label study is more like the real-world use of erenumab, and as such the results are similar to what many practitioners are seeing in their clinical experience. Approximately 25% of subjects discontinued erenumab, mostly due to ineffectiveness. Adverse events were mild, and although erenumab has warnings for constipation and hypertension, this study did not show either as increasing over 2 years. Erenumab appeared to be tolerable over time. There were no newly noted safety signals in this study.
The efficacy of erenumab also appeared to be stable over time, without the development of tolerance to the medication. There is a slight decrease in the 50% responder rate at 2 years when these more refractory patients are compared to those that did not have multiple treatment failures. This study also looked at “functional parameters,” such as Migraine Disability Assessment (MIDAS) and Headache Impact Test (HIT-6), both of which were significantly improved over time.
Although there are some significant limitations in this study-primarily the fact that it is open label—this does give a more representative and real-world sample of patients who will be prescribed erenumab in the United States. Most practitioners will be glad to find that the long-term use of erenumab appears safe, and the efficacy remains stable, even in a more difficult-to-treat population.
A randomized controlled international study investigated the preventive use of occipital nerve blocks in migraine without aura.2 The majority of the literature for the use of occipital nerve blocks is for acute treatment, and arguably the most significant study prior to this was Friedman et al3 investigating the use of this procedure in the emergency ward. Prior occipital nerve studies have been inconclusive, and although occipital nerve blocks are considered standard of care for specific conditions in most headache centers, reimbursement is usually very limited. Insurance companies have quoted prior preventive occipital nerve studies to justify non-coverage of these procedures, making access to them for many patients very limited.
Occipital nerve blocks are not performed uniformly, both regarding the medications used—some practitioners use no steroids, some use lidocaine and bupivocaine—and regarding the placement of the injections. In this a small cohort study, 55 subjects were divided into four groups for intervention—one of which was a control group of saline—and all were given one 2.5 mL injection at a point in between the occipital protuberance and the mastoid process bilaterally. Due to adverse events (alopecia and cutaneous atrophy) in two of the triamcinolone groups, recruitment was halted for those two groups. Patients were assessed based on headache duration, frequency, and severity over a 4-week course.
Compared to baseline all interventional groups had significantly decreased headache severity, which did return closer to baseline during the final week. Headache duration was decreased in the first 2 weeks post-injection. Headache frequency was seen to return to baseline at week 4, but prior to that the groups injected with lidocaine had a significant decrease in migraine frequency, with an average decrease in headache days.
Occipital nerve blocks are performed frequently for migraine, occipital neuralgia, cervicogenic headache, and many other conditions with noted tenderness over the occiput. As noted above, they are not performed uniformly—sometimes they are given for acute headache pain or status migranosus, and other times they are used in regular intervals for prevention. This data does finally show a preventive benefit with occipital nerve blocks, and this may allow for modifications in how occipital nerve blocks are currently performed. Based on this study, if given preventively, occipital nerve blocks should only contain topical anesthetics, not steroids, and should be performed on an every 2-3 week basis.
The limitations of this study are significant as well. This is a very small cohort, and the injections were performed in only one manner (one bilateral injection), whereas many practitioners will target the greater and lesser branches of the occipital nerve individually. There were no exclusion criteria for subjects that already had occipital nerve blocks performed—those patients would be unblinded as there is a different sensation when injected with a topical anesthetic versus normal saline (normal saline does not cause burning subcutaneously).
These results should pave the way for further investigations in the use of occipital and other nerve blocks in the prevention of migraine. This should allow better access for our patients and the possibility of performing these procedures more uniformly in the future.
It can be challenging for many practitioners to determine which medication is ideal for individual situations. This is especially true when treating chronic migraine, where many potential complicating factors can influence positive to negative responses to treatment. The investigators here sought to determine which factors may potentially predict a positive response to galcanezumab.4
This is an observational study, where 156 subjects with a diagnosis of chronic migraine were enrolled. There was a 1-month run-in period where the following characteristics were collected: monthly headache days, monthly abortive medication intake, clinical features of migraine, and disability scores (MIDAS and HIT-6). These were tracked over a 3-month period after starting glacanezumab.
Approximately 40% of subjects experienced a 50% reduction in headache frequency. The better responders had a lower body mass index, fewer previously failed preventive medications, unilateral headache pain, and previous good response to triptan use. Surprisingly, the presence of medication overuse was associated with persistent improvement at 3 months as well, with over 60% of subjects with medication overuse no longer overusing acute medications at 3 months.
This study is helpful in identifying specific features that may allow a practitioner to better recommend CGRP mAb medications, such as galcanezumab. Chronic migraine can offer a challenge to even the best trained clinicians. Patients will often have multiple factors that have led to a conversion from episodic to chronic migraine, and a history of medication failures or intolerances. These patients are often referred specifically due to these challenges.
When deciding on a preventive medication for patients with chronic migraine, we often first consider which oral preventive medications may allow us to treat migraine in addition to another underlying issue—such as insomnia, depression, or hypertension. Although the oral class can improve other comorbidities, intolerance is significantly higher for most of these medications as well. The CGRP mAb class is somewhat more ideal for prevention of migraine; the focus when using this class is for migraine prevention alone, and the side effect profile is more tolerable for most patients. That said, if predictive factors were known a more individualized approach to migraine prevention would be possible.
The authors’ recognition of the factors associated with improvement in patients using glacanezumab allows this better individualization. Based on these results, patients with more unilateral pain, lower BMI, and good response to triptans could be recommended glacanuzumab with a great degree of confidence. This should be irrespective of even high frequency use of acute medications, as most of subjects in this study with medication overuse reverted after 3 months.
There is never a single ideal preventive or acute treatment for migraine in any population, however, recognizing factors that allow for an individualized approach improves the quality of life for our patients, and leaves them less disabled by migraine.
References
- Ferrari MD et al. Two-year efficacy and safety of erenumab in participants with episodic migraine and 2–4 prior preventive treatment failures: results from the LIBERTY study. J Neurol Neurosurg Psychiatry. 2021(Nov 29).
- Malekian N et al. Preventive effect of greater occipital nerve block on patients with episodic migraine: A randomized double‐blind placebo‐controlled clinical trial. Cephalalgia. 2021(Nov 17).
- Friedman BW et al. A Randomized, Sham-Controlled Trial of Bilateral Greater Occipital Nerve Blocks With Bupivacaine for Acute Migraine Patients Refractory to Standard Emergency Department Treatment With Metoclopramide. Headache. 2018(Oct);58(9):1427-34. https://doi.org/10.1111/head.13395.
- Vernieri F et al. Rapid response to galcanezumab and predictive factors in chronic migraine patients: A 3-month observational, longitudinal, cohort, multicenter, Italian real-life study. Eur J Neurol. 2021(Nov 26).
Ferrari et al1 provided information on an open label extension to the “LIBERTY” study which investigated the use of erenumab in subjects with episodic migraine that have failed multiple prior preventive medications. The initial Calcitonin gene-related peptide (CGRP) monoclonal antibody (mAb) studies excluded more refractory patients. Most commercial insurances in the United States have a “step” policy that relates to use of these and other newer medications, meaning that the majority of patient in the US who receive these medications have previously tried other preventive medications. This raised the question whether migraine refractoriness is a negative predictive factor for erenumab.
This long-term open label study is more like the real-world use of erenumab, and as such the results are similar to what many practitioners are seeing in their clinical experience. Approximately 25% of subjects discontinued erenumab, mostly due to ineffectiveness. Adverse events were mild, and although erenumab has warnings for constipation and hypertension, this study did not show either as increasing over 2 years. Erenumab appeared to be tolerable over time. There were no newly noted safety signals in this study.
The efficacy of erenumab also appeared to be stable over time, without the development of tolerance to the medication. There is a slight decrease in the 50% responder rate at 2 years when these more refractory patients are compared to those that did not have multiple treatment failures. This study also looked at “functional parameters,” such as Migraine Disability Assessment (MIDAS) and Headache Impact Test (HIT-6), both of which were significantly improved over time.
Although there are some significant limitations in this study-primarily the fact that it is open label—this does give a more representative and real-world sample of patients who will be prescribed erenumab in the United States. Most practitioners will be glad to find that the long-term use of erenumab appears safe, and the efficacy remains stable, even in a more difficult-to-treat population.
A randomized controlled international study investigated the preventive use of occipital nerve blocks in migraine without aura.2 The majority of the literature for the use of occipital nerve blocks is for acute treatment, and arguably the most significant study prior to this was Friedman et al3 investigating the use of this procedure in the emergency ward. Prior occipital nerve studies have been inconclusive, and although occipital nerve blocks are considered standard of care for specific conditions in most headache centers, reimbursement is usually very limited. Insurance companies have quoted prior preventive occipital nerve studies to justify non-coverage of these procedures, making access to them for many patients very limited.
Occipital nerve blocks are not performed uniformly, both regarding the medications used—some practitioners use no steroids, some use lidocaine and bupivocaine—and regarding the placement of the injections. In this a small cohort study, 55 subjects were divided into four groups for intervention—one of which was a control group of saline—and all were given one 2.5 mL injection at a point in between the occipital protuberance and the mastoid process bilaterally. Due to adverse events (alopecia and cutaneous atrophy) in two of the triamcinolone groups, recruitment was halted for those two groups. Patients were assessed based on headache duration, frequency, and severity over a 4-week course.
Compared to baseline all interventional groups had significantly decreased headache severity, which did return closer to baseline during the final week. Headache duration was decreased in the first 2 weeks post-injection. Headache frequency was seen to return to baseline at week 4, but prior to that the groups injected with lidocaine had a significant decrease in migraine frequency, with an average decrease in headache days.
Occipital nerve blocks are performed frequently for migraine, occipital neuralgia, cervicogenic headache, and many other conditions with noted tenderness over the occiput. As noted above, they are not performed uniformly—sometimes they are given for acute headache pain or status migranosus, and other times they are used in regular intervals for prevention. This data does finally show a preventive benefit with occipital nerve blocks, and this may allow for modifications in how occipital nerve blocks are currently performed. Based on this study, if given preventively, occipital nerve blocks should only contain topical anesthetics, not steroids, and should be performed on an every 2-3 week basis.
The limitations of this study are significant as well. This is a very small cohort, and the injections were performed in only one manner (one bilateral injection), whereas many practitioners will target the greater and lesser branches of the occipital nerve individually. There were no exclusion criteria for subjects that already had occipital nerve blocks performed—those patients would be unblinded as there is a different sensation when injected with a topical anesthetic versus normal saline (normal saline does not cause burning subcutaneously).
These results should pave the way for further investigations in the use of occipital and other nerve blocks in the prevention of migraine. This should allow better access for our patients and the possibility of performing these procedures more uniformly in the future.
It can be challenging for many practitioners to determine which medication is ideal for individual situations. This is especially true when treating chronic migraine, where many potential complicating factors can influence positive to negative responses to treatment. The investigators here sought to determine which factors may potentially predict a positive response to galcanezumab.4
This is an observational study, where 156 subjects with a diagnosis of chronic migraine were enrolled. There was a 1-month run-in period where the following characteristics were collected: monthly headache days, monthly abortive medication intake, clinical features of migraine, and disability scores (MIDAS and HIT-6). These were tracked over a 3-month period after starting glacanezumab.
Approximately 40% of subjects experienced a 50% reduction in headache frequency. The better responders had a lower body mass index, fewer previously failed preventive medications, unilateral headache pain, and previous good response to triptan use. Surprisingly, the presence of medication overuse was associated with persistent improvement at 3 months as well, with over 60% of subjects with medication overuse no longer overusing acute medications at 3 months.
This study is helpful in identifying specific features that may allow a practitioner to better recommend CGRP mAb medications, such as galcanezumab. Chronic migraine can offer a challenge to even the best trained clinicians. Patients will often have multiple factors that have led to a conversion from episodic to chronic migraine, and a history of medication failures or intolerances. These patients are often referred specifically due to these challenges.
When deciding on a preventive medication for patients with chronic migraine, we often first consider which oral preventive medications may allow us to treat migraine in addition to another underlying issue—such as insomnia, depression, or hypertension. Although the oral class can improve other comorbidities, intolerance is significantly higher for most of these medications as well. The CGRP mAb class is somewhat more ideal for prevention of migraine; the focus when using this class is for migraine prevention alone, and the side effect profile is more tolerable for most patients. That said, if predictive factors were known a more individualized approach to migraine prevention would be possible.
The authors’ recognition of the factors associated with improvement in patients using glacanezumab allows this better individualization. Based on these results, patients with more unilateral pain, lower BMI, and good response to triptans could be recommended glacanuzumab with a great degree of confidence. This should be irrespective of even high frequency use of acute medications, as most of subjects in this study with medication overuse reverted after 3 months.
There is never a single ideal preventive or acute treatment for migraine in any population, however, recognizing factors that allow for an individualized approach improves the quality of life for our patients, and leaves them less disabled by migraine.
References
- Ferrari MD et al. Two-year efficacy and safety of erenumab in participants with episodic migraine and 2–4 prior preventive treatment failures: results from the LIBERTY study. J Neurol Neurosurg Psychiatry. 2021(Nov 29).
- Malekian N et al. Preventive effect of greater occipital nerve block on patients with episodic migraine: A randomized double‐blind placebo‐controlled clinical trial. Cephalalgia. 2021(Nov 17).
- Friedman BW et al. A Randomized, Sham-Controlled Trial of Bilateral Greater Occipital Nerve Blocks With Bupivacaine for Acute Migraine Patients Refractory to Standard Emergency Department Treatment With Metoclopramide. Headache. 2018(Oct);58(9):1427-34. https://doi.org/10.1111/head.13395.
- Vernieri F et al. Rapid response to galcanezumab and predictive factors in chronic migraine patients: A 3-month observational, longitudinal, cohort, multicenter, Italian real-life study. Eur J Neurol. 2021(Nov 26).
Responding to the Pandemic: How is the VA doing?
The VA’s Coronavirus Disease 2019 Response Report is now in its third iteration as the pandemic continues. On the bright side, as Steven Lieberman, MD, deputy under secretary for health at the US Department of Veterans Affairs (VA), writes in the report’s introduction, “we have learned a great deal about mounting a national response to a public health crisis.”
“Annex B” covers January 1, 2021 to July 31, 2021, building on the 2 previous reports. All 3 have sought to capture and share lessons learned, with updated information on vaccination, elder care, health equity, mental health, health care ethics, preparedness, and other topics.
As the pandemic evolved, so did the VA efforts to cope with it. This iteration, for instance, deals with details of the campaign that vaccinated more than 2.5 million people “while sustaining all other aspects of the pandemic response and veteran health services,” and how the VA implemented a vaccine mandate for all VA employees in health care roles—the first federal agency to do so. In addition to vaccinating veterans, the Strengthening and Amplifying Vaccination Efforts to Locally Immunize All Veterans and Every Spouse (SAVE LIVES) Act led to nearly 80,000 other vaccinations among families, caregivers, and veterans who do not use VHA services.
The VA also conducted extensive COVID-19 testing, processing as many as 70,000 to 90,000 tests per week. It enhanced telehealth services to reach home-based and rural veterans, for an almost 2,500% increase in home-based primary care. Recognizing the added stress the pandemic put on people at risk for suicide, the VHA used predictive analytic tools specific to veterans with COVID-19 and monitored “high-risk flags,” using them to identify veterans for tailored outreach.
The response also included carrying out 158 Federal Emergency Management Agency Fourth Mission assignments. The report highlights the contributions of the more than 1,600 Veterans Health Administration (VHA) employees who volunteered to deploy across the country, often multiple times.
In addition to active response, more than 300 studies on COVID-19 have been published by VA researchers.
The current status report discusses how to expand what worked and to improve what did not. For instance, one unsurprising finding was that “the sustained pandemic response has imposed stress on the workforce, most evident in the nursing workforce.” The recommendation: Develop a comprehensive strategy with metrics and actions to monitor and mitigate stress on the health care workforce, facilitate wellness, and enhance retention.
The finding that VHA has demonstrated that telehealth usage for care to elderly veterans is “beneficial and feasible with the right technical support” led to recommendations for expanded research to identify effective COVID-19 prevention and intervention measures for elderly veterans residing at home or in long-term care facilities.
The research found that VHA processes for protecting community living center (CLC) residents during the pandemic “have succeeded in keeping rates of CLC-onset COVID-19 at the same rate as for the population of enrolled veterans over 65,” the report says. The recommendation based on that finding is to develop an information system to facilitate monitoring of state-run veterans homes for indicators of infectious disease risk, combining periodic assessment results with epidemiologic community data.
However, the report also acknowledges unexpected detours or blocks. “Planning for the mass vaccination campaign was highly effective, but did not anticipate the complexity of interagency support.” And “[t[he inability to access state vaccination data left VHA with an incomplete picture of the vaccination status of enrolled veterans.” In response, the VA recommends incorporating interagency support into planning templates and pursuing legislative action to enable the VA to obtain vaccination data from states.
Overall, the report gives the VA high marks for managing a “well-coordinated response” to an overwhelming crisis. But the lessons are not over.
“As we continue to address the pandemic and as new variants arise,” Dr. Lieberman said in comments, “it is clear that continuous learning and improvement are essential to a successful COVID-19 response. We will continue to update this report to document our efforts so veterans, doctors, and the public can understand and learn from what we’ve discovered to better serve our veterans and communities.” Stay tuned for Annex C.
The VA’s Coronavirus Disease 2019 Response Report is now in its third iteration as the pandemic continues. On the bright side, as Steven Lieberman, MD, deputy under secretary for health at the US Department of Veterans Affairs (VA), writes in the report’s introduction, “we have learned a great deal about mounting a national response to a public health crisis.”
“Annex B” covers January 1, 2021 to July 31, 2021, building on the 2 previous reports. All 3 have sought to capture and share lessons learned, with updated information on vaccination, elder care, health equity, mental health, health care ethics, preparedness, and other topics.
As the pandemic evolved, so did the VA efforts to cope with it. This iteration, for instance, deals with details of the campaign that vaccinated more than 2.5 million people “while sustaining all other aspects of the pandemic response and veteran health services,” and how the VA implemented a vaccine mandate for all VA employees in health care roles—the first federal agency to do so. In addition to vaccinating veterans, the Strengthening and Amplifying Vaccination Efforts to Locally Immunize All Veterans and Every Spouse (SAVE LIVES) Act led to nearly 80,000 other vaccinations among families, caregivers, and veterans who do not use VHA services.
The VA also conducted extensive COVID-19 testing, processing as many as 70,000 to 90,000 tests per week. It enhanced telehealth services to reach home-based and rural veterans, for an almost 2,500% increase in home-based primary care. Recognizing the added stress the pandemic put on people at risk for suicide, the VHA used predictive analytic tools specific to veterans with COVID-19 and monitored “high-risk flags,” using them to identify veterans for tailored outreach.
The response also included carrying out 158 Federal Emergency Management Agency Fourth Mission assignments. The report highlights the contributions of the more than 1,600 Veterans Health Administration (VHA) employees who volunteered to deploy across the country, often multiple times.
In addition to active response, more than 300 studies on COVID-19 have been published by VA researchers.
The current status report discusses how to expand what worked and to improve what did not. For instance, one unsurprising finding was that “the sustained pandemic response has imposed stress on the workforce, most evident in the nursing workforce.” The recommendation: Develop a comprehensive strategy with metrics and actions to monitor and mitigate stress on the health care workforce, facilitate wellness, and enhance retention.
The finding that VHA has demonstrated that telehealth usage for care to elderly veterans is “beneficial and feasible with the right technical support” led to recommendations for expanded research to identify effective COVID-19 prevention and intervention measures for elderly veterans residing at home or in long-term care facilities.
The research found that VHA processes for protecting community living center (CLC) residents during the pandemic “have succeeded in keeping rates of CLC-onset COVID-19 at the same rate as for the population of enrolled veterans over 65,” the report says. The recommendation based on that finding is to develop an information system to facilitate monitoring of state-run veterans homes for indicators of infectious disease risk, combining periodic assessment results with epidemiologic community data.
However, the report also acknowledges unexpected detours or blocks. “Planning for the mass vaccination campaign was highly effective, but did not anticipate the complexity of interagency support.” And “[t[he inability to access state vaccination data left VHA with an incomplete picture of the vaccination status of enrolled veterans.” In response, the VA recommends incorporating interagency support into planning templates and pursuing legislative action to enable the VA to obtain vaccination data from states.
Overall, the report gives the VA high marks for managing a “well-coordinated response” to an overwhelming crisis. But the lessons are not over.
“As we continue to address the pandemic and as new variants arise,” Dr. Lieberman said in comments, “it is clear that continuous learning and improvement are essential to a successful COVID-19 response. We will continue to update this report to document our efforts so veterans, doctors, and the public can understand and learn from what we’ve discovered to better serve our veterans and communities.” Stay tuned for Annex C.
The VA’s Coronavirus Disease 2019 Response Report is now in its third iteration as the pandemic continues. On the bright side, as Steven Lieberman, MD, deputy under secretary for health at the US Department of Veterans Affairs (VA), writes in the report’s introduction, “we have learned a great deal about mounting a national response to a public health crisis.”
“Annex B” covers January 1, 2021 to July 31, 2021, building on the 2 previous reports. All 3 have sought to capture and share lessons learned, with updated information on vaccination, elder care, health equity, mental health, health care ethics, preparedness, and other topics.
As the pandemic evolved, so did the VA efforts to cope with it. This iteration, for instance, deals with details of the campaign that vaccinated more than 2.5 million people “while sustaining all other aspects of the pandemic response and veteran health services,” and how the VA implemented a vaccine mandate for all VA employees in health care roles—the first federal agency to do so. In addition to vaccinating veterans, the Strengthening and Amplifying Vaccination Efforts to Locally Immunize All Veterans and Every Spouse (SAVE LIVES) Act led to nearly 80,000 other vaccinations among families, caregivers, and veterans who do not use VHA services.
The VA also conducted extensive COVID-19 testing, processing as many as 70,000 to 90,000 tests per week. It enhanced telehealth services to reach home-based and rural veterans, for an almost 2,500% increase in home-based primary care. Recognizing the added stress the pandemic put on people at risk for suicide, the VHA used predictive analytic tools specific to veterans with COVID-19 and monitored “high-risk flags,” using them to identify veterans for tailored outreach.
The response also included carrying out 158 Federal Emergency Management Agency Fourth Mission assignments. The report highlights the contributions of the more than 1,600 Veterans Health Administration (VHA) employees who volunteered to deploy across the country, often multiple times.
In addition to active response, more than 300 studies on COVID-19 have been published by VA researchers.
The current status report discusses how to expand what worked and to improve what did not. For instance, one unsurprising finding was that “the sustained pandemic response has imposed stress on the workforce, most evident in the nursing workforce.” The recommendation: Develop a comprehensive strategy with metrics and actions to monitor and mitigate stress on the health care workforce, facilitate wellness, and enhance retention.
The finding that VHA has demonstrated that telehealth usage for care to elderly veterans is “beneficial and feasible with the right technical support” led to recommendations for expanded research to identify effective COVID-19 prevention and intervention measures for elderly veterans residing at home or in long-term care facilities.
The research found that VHA processes for protecting community living center (CLC) residents during the pandemic “have succeeded in keeping rates of CLC-onset COVID-19 at the same rate as for the population of enrolled veterans over 65,” the report says. The recommendation based on that finding is to develop an information system to facilitate monitoring of state-run veterans homes for indicators of infectious disease risk, combining periodic assessment results with epidemiologic community data.
However, the report also acknowledges unexpected detours or blocks. “Planning for the mass vaccination campaign was highly effective, but did not anticipate the complexity of interagency support.” And “[t[he inability to access state vaccination data left VHA with an incomplete picture of the vaccination status of enrolled veterans.” In response, the VA recommends incorporating interagency support into planning templates and pursuing legislative action to enable the VA to obtain vaccination data from states.
Overall, the report gives the VA high marks for managing a “well-coordinated response” to an overwhelming crisis. But the lessons are not over.
“As we continue to address the pandemic and as new variants arise,” Dr. Lieberman said in comments, “it is clear that continuous learning and improvement are essential to a successful COVID-19 response. We will continue to update this report to document our efforts so veterans, doctors, and the public can understand and learn from what we’ve discovered to better serve our veterans and communities.” Stay tuned for Annex C.
What if the National Guard Can’t Help?
What if the National Guard Can’t Help?
In early January, Ohio not only set a state record for COVID-19 hospitalizations—it had the fourth highest rate in the country, with 6,747 hospitalized coronavirus patients on January 10, a 40% increase over the previous 21 days. Most were unvaccinated. To help overwhelmed hospitals cope, Ohio Gov. Mike DeWine turned to the National Guard. Unfortunately, nearly half of the Ohio National Guard also were unvaccinated.
By US Department of Defense (DoD) directive, National Guard members must have a COVID-19 vaccination to be deployed on hospital missions. Thus, in COVID hotspots across the nation, governors are on the horns of a dilemma. They want and need to deploy the National Guard to give medical and nonclinical support but aren’t sure whether they will be able to or, indeed, whether they should.
So far, vaccinated teams are already on the ground in a number of states. In Indiana, where hospitalizations jumped 50% over 2 weeks in December, the National Guard sent 6-person teams, all fully vaccinated. In New Hampshire, 70 guards are being deployed to help hospitals with food service, clerical work, and other nonmedical functions. New York Governor Kathy Hochul has deployed guard members for help to ease the strain on nursing homes. Massachusetts Governor Charlie Baker has activated up to 500 guard members; some will be supporting 55 acute care hospital and 12 ambulance services. In Maine, where cases have peaked, Governor Janet Mills activated guard members to support nursing facilities and administer monoclonal antibodies. The Louisiana National Guard has administered more than 542,000 COVID-19 tests and 206,300 vaccines. As many as 1,000 Maryland Air and Army National Guardsmen are being activated to help with testing and other missions.
However, as in Ohio, other states are facing problematic scenarios. For instance, about 40% of the more than 20,000 Texas National Guard are refusing to get vaccinated, challenging the Biden Administration vaccine requirement for all military.
And a court showdown over federal vaccine mandates, started by Governor Kevin Stitt of Oklahoma and joined by the Republican governors of Wyoming, Iowa, Alaska, Nebraska, and Mississippi, came to a head in December. Last November, Stitt asked Defense Secretary Lloyd Austin to exempt Oklahoma’s National Guard from the vaccine mandate. He claimed the requirement violated the personal freedoms of many Oklahomans and could cause them to “potentially sacrifice their personal beliefs.” But in a memo to the Joint Chiefs chairmen, the service secretaries and the head of the National Guard Bureau, Austin wrote that Pentagon funds could not be used to pay for duties performed under Title 32 for members of the Guard who do not comply with the military’s vaccine requirement. (Title 32 refers to Guard operations under state orders.) Austin also said National Guard members must be vaccinated to participate in drills, training, and other duty conducted under Title 32.
Stitt, maintaining that he is commander in chief of the Oklahoma National Guard as long as it operates under Title 32 orders, put out his own memo stipulating that no Guard member was required to get vaccinated. He also ordered Brig. Gen. Thomas Mancino, newly appointed commander of the Oklahoma National Guard, to not enforce the mandate. Subsequently, Mancino issued a statement pointing out that current state law is limited in protecting troops who opt out of the shot. Moreover, if the Guard were called up under federal orders, he said, he would enforce the mandate. Training events, schools, and mobilizations were going to “eventually force you out of that safe harbor,” he wrote, “…This is reality.”
In late December, a federal judge denied Oklahoma’s motion to enjoin the mandate. The Oklahoma Attorney General’s office responded, “We will not be surprised if the President’s vaccine mandate actually reduces the nation’s military readiness instead of promoting it.”
In a press briefing, Pentagon press secretary John Kirby said, “The Secretary has the authorities he needs to require this vaccine across the force, including the National Guard. …[E]ven when they’re in a Title 32 status.” He added, “It is a lawful order for National Guardsmen to receive the COVID vaccine. It’s a lawful order, and refusing to do that, absent of an improved exemption, puts them in the same potential [position] as active-duty members who refuse the vaccine.” That could mean, for instance, loss of pay and membership in the National Guard.
A core rationale for the mandate, according to Secretary Austin, is the need for military readiness—meaning Guard members must be healthy and fit for duty. And that extends to being healthy and fit for missions like transporting at-risk patients. Ohio National Guard Adjutant General Major General John Harris Jr. said, “I would never put a soldier or airman in harm’s way without the best protection we could put on them—body armor, helmets. And this medical readiness is the exact same thing. We’re putting folks into harm’s way.” He has moved the deadline from the Pentagon’s June 30 date to March 31—a move that boosted the vaccination rate from 53% to 56% in one week.
Ohio Governor DeWine has expressed frustration that almost half of the Ohio Army National Guard personnel can’t be deployed on this mission because they’re unvaccinated. “In some of our testing places, 40 to 50% of the people are testing positive,” he said. “So this is a high-risk operation. You need to be protected. The best way for you to be protected is to get the vaccination.”
As of December 2021, according to the National Guard Bureau, the National Guard as a whole was 66% fully vaccinated. The percentages vary according to service; for instance, nearly 90% of airmen have been vaccinated, compared with only 40% of Army Guardsmen. Among the states challenging the mandate, the vaccinated rates have been moving upward: In Alaska, about 92% of the Air National Guard have been vaccinated—leaving roughly 11,000 troops who had not met the December 2 deadline. In Iowa, as of Nov. 30, 91% of Air National Guard and 80% of Army National Guard members had been vaccinated, but about 9,000 soldiers had been directed to get the vaccination or risk disciplinary action. Almost 2,200 of the more than 2,800-strong Wyoming National Guard (77%) have received at least 1 dose. Nebraska Air National Guard’s force of 1,000 was 94% fully vaccinated as of December 1. (Maj Scott Ingalsbe, public affairs officer, said, “Vaccinations are tied to individual medical readiness. They provide service members with the best protection available so they can perform missions across the globe.”).
In most states, Army National Guard members have until June 30, 2022, to comply. “Our soldiers …have until [the DoD’s deadline], and some of them are just going to wait close to the deadline,” John Goheen of the National Guard Association of the United States said in a discussion on NPR. “That’s human nature.”
Earlier this month, Texas Governor Greg Abbott told National Guard members they can ignore the Pentagon’s COVID-19 vaccine mandate: “President Biden is not your commander-in-chief.” He has also sued the Biden administration over the requirement.
In the meantime, the hospitals at breaking point must hope for the best and take as much help as they can get.
In early January, Ohio not only set a state record for COVID-19 hospitalizations—it had the fourth highest rate in the country, with 6,747 hospitalized coronavirus patients on January 10, a 40% increase over the previous 21 days. Most were unvaccinated. To help overwhelmed hospitals cope, Ohio Gov. Mike DeWine turned to the National Guard. Unfortunately, nearly half of the Ohio National Guard also were unvaccinated.
By US Department of Defense (DoD) directive, National Guard members must have a COVID-19 vaccination to be deployed on hospital missions. Thus, in COVID hotspots across the nation, governors are on the horns of a dilemma. They want and need to deploy the National Guard to give medical and nonclinical support but aren’t sure whether they will be able to or, indeed, whether they should.
So far, vaccinated teams are already on the ground in a number of states. In Indiana, where hospitalizations jumped 50% over 2 weeks in December, the National Guard sent 6-person teams, all fully vaccinated. In New Hampshire, 70 guards are being deployed to help hospitals with food service, clerical work, and other nonmedical functions. New York Governor Kathy Hochul has deployed guard members for help to ease the strain on nursing homes. Massachusetts Governor Charlie Baker has activated up to 500 guard members; some will be supporting 55 acute care hospital and 12 ambulance services. In Maine, where cases have peaked, Governor Janet Mills activated guard members to support nursing facilities and administer monoclonal antibodies. The Louisiana National Guard has administered more than 542,000 COVID-19 tests and 206,300 vaccines. As many as 1,000 Maryland Air and Army National Guardsmen are being activated to help with testing and other missions.
However, as in Ohio, other states are facing problematic scenarios. For instance, about 40% of the more than 20,000 Texas National Guard are refusing to get vaccinated, challenging the Biden Administration vaccine requirement for all military.
And a court showdown over federal vaccine mandates, started by Governor Kevin Stitt of Oklahoma and joined by the Republican governors of Wyoming, Iowa, Alaska, Nebraska, and Mississippi, came to a head in December. Last November, Stitt asked Defense Secretary Lloyd Austin to exempt Oklahoma’s National Guard from the vaccine mandate. He claimed the requirement violated the personal freedoms of many Oklahomans and could cause them to “potentially sacrifice their personal beliefs.” But in a memo to the Joint Chiefs chairmen, the service secretaries and the head of the National Guard Bureau, Austin wrote that Pentagon funds could not be used to pay for duties performed under Title 32 for members of the Guard who do not comply with the military’s vaccine requirement. (Title 32 refers to Guard operations under state orders.) Austin also said National Guard members must be vaccinated to participate in drills, training, and other duty conducted under Title 32.
Stitt, maintaining that he is commander in chief of the Oklahoma National Guard as long as it operates under Title 32 orders, put out his own memo stipulating that no Guard member was required to get vaccinated. He also ordered Brig. Gen. Thomas Mancino, newly appointed commander of the Oklahoma National Guard, to not enforce the mandate. Subsequently, Mancino issued a statement pointing out that current state law is limited in protecting troops who opt out of the shot. Moreover, if the Guard were called up under federal orders, he said, he would enforce the mandate. Training events, schools, and mobilizations were going to “eventually force you out of that safe harbor,” he wrote, “…This is reality.”
In late December, a federal judge denied Oklahoma’s motion to enjoin the mandate. The Oklahoma Attorney General’s office responded, “We will not be surprised if the President’s vaccine mandate actually reduces the nation’s military readiness instead of promoting it.”
In a press briefing, Pentagon press secretary John Kirby said, “The Secretary has the authorities he needs to require this vaccine across the force, including the National Guard. …[E]ven when they’re in a Title 32 status.” He added, “It is a lawful order for National Guardsmen to receive the COVID vaccine. It’s a lawful order, and refusing to do that, absent of an improved exemption, puts them in the same potential [position] as active-duty members who refuse the vaccine.” That could mean, for instance, loss of pay and membership in the National Guard.
A core rationale for the mandate, according to Secretary Austin, is the need for military readiness—meaning Guard members must be healthy and fit for duty. And that extends to being healthy and fit for missions like transporting at-risk patients. Ohio National Guard Adjutant General Major General John Harris Jr. said, “I would never put a soldier or airman in harm’s way without the best protection we could put on them—body armor, helmets. And this medical readiness is the exact same thing. We’re putting folks into harm’s way.” He has moved the deadline from the Pentagon’s June 30 date to March 31—a move that boosted the vaccination rate from 53% to 56% in one week.
Ohio Governor DeWine has expressed frustration that almost half of the Ohio Army National Guard personnel can’t be deployed on this mission because they’re unvaccinated. “In some of our testing places, 40 to 50% of the people are testing positive,” he said. “So this is a high-risk operation. You need to be protected. The best way for you to be protected is to get the vaccination.”
As of December 2021, according to the National Guard Bureau, the National Guard as a whole was 66% fully vaccinated. The percentages vary according to service; for instance, nearly 90% of airmen have been vaccinated, compared with only 40% of Army Guardsmen. Among the states challenging the mandate, the vaccinated rates have been moving upward: In Alaska, about 92% of the Air National Guard have been vaccinated—leaving roughly 11,000 troops who had not met the December 2 deadline. In Iowa, as of Nov. 30, 91% of Air National Guard and 80% of Army National Guard members had been vaccinated, but about 9,000 soldiers had been directed to get the vaccination or risk disciplinary action. Almost 2,200 of the more than 2,800-strong Wyoming National Guard (77%) have received at least 1 dose. Nebraska Air National Guard’s force of 1,000 was 94% fully vaccinated as of December 1. (Maj Scott Ingalsbe, public affairs officer, said, “Vaccinations are tied to individual medical readiness. They provide service members with the best protection available so they can perform missions across the globe.”).
In most states, Army National Guard members have until June 30, 2022, to comply. “Our soldiers …have until [the DoD’s deadline], and some of them are just going to wait close to the deadline,” John Goheen of the National Guard Association of the United States said in a discussion on NPR. “That’s human nature.”
Earlier this month, Texas Governor Greg Abbott told National Guard members they can ignore the Pentagon’s COVID-19 vaccine mandate: “President Biden is not your commander-in-chief.” He has also sued the Biden administration over the requirement.
In the meantime, the hospitals at breaking point must hope for the best and take as much help as they can get.
In early January, Ohio not only set a state record for COVID-19 hospitalizations—it had the fourth highest rate in the country, with 6,747 hospitalized coronavirus patients on January 10, a 40% increase over the previous 21 days. Most were unvaccinated. To help overwhelmed hospitals cope, Ohio Gov. Mike DeWine turned to the National Guard. Unfortunately, nearly half of the Ohio National Guard also were unvaccinated.
By US Department of Defense (DoD) directive, National Guard members must have a COVID-19 vaccination to be deployed on hospital missions. Thus, in COVID hotspots across the nation, governors are on the horns of a dilemma. They want and need to deploy the National Guard to give medical and nonclinical support but aren’t sure whether they will be able to or, indeed, whether they should.
So far, vaccinated teams are already on the ground in a number of states. In Indiana, where hospitalizations jumped 50% over 2 weeks in December, the National Guard sent 6-person teams, all fully vaccinated. In New Hampshire, 70 guards are being deployed to help hospitals with food service, clerical work, and other nonmedical functions. New York Governor Kathy Hochul has deployed guard members for help to ease the strain on nursing homes. Massachusetts Governor Charlie Baker has activated up to 500 guard members; some will be supporting 55 acute care hospital and 12 ambulance services. In Maine, where cases have peaked, Governor Janet Mills activated guard members to support nursing facilities and administer monoclonal antibodies. The Louisiana National Guard has administered more than 542,000 COVID-19 tests and 206,300 vaccines. As many as 1,000 Maryland Air and Army National Guardsmen are being activated to help with testing and other missions.
However, as in Ohio, other states are facing problematic scenarios. For instance, about 40% of the more than 20,000 Texas National Guard are refusing to get vaccinated, challenging the Biden Administration vaccine requirement for all military.
And a court showdown over federal vaccine mandates, started by Governor Kevin Stitt of Oklahoma and joined by the Republican governors of Wyoming, Iowa, Alaska, Nebraska, and Mississippi, came to a head in December. Last November, Stitt asked Defense Secretary Lloyd Austin to exempt Oklahoma’s National Guard from the vaccine mandate. He claimed the requirement violated the personal freedoms of many Oklahomans and could cause them to “potentially sacrifice their personal beliefs.” But in a memo to the Joint Chiefs chairmen, the service secretaries and the head of the National Guard Bureau, Austin wrote that Pentagon funds could not be used to pay for duties performed under Title 32 for members of the Guard who do not comply with the military’s vaccine requirement. (Title 32 refers to Guard operations under state orders.) Austin also said National Guard members must be vaccinated to participate in drills, training, and other duty conducted under Title 32.
Stitt, maintaining that he is commander in chief of the Oklahoma National Guard as long as it operates under Title 32 orders, put out his own memo stipulating that no Guard member was required to get vaccinated. He also ordered Brig. Gen. Thomas Mancino, newly appointed commander of the Oklahoma National Guard, to not enforce the mandate. Subsequently, Mancino issued a statement pointing out that current state law is limited in protecting troops who opt out of the shot. Moreover, if the Guard were called up under federal orders, he said, he would enforce the mandate. Training events, schools, and mobilizations were going to “eventually force you out of that safe harbor,” he wrote, “…This is reality.”
In late December, a federal judge denied Oklahoma’s motion to enjoin the mandate. The Oklahoma Attorney General’s office responded, “We will not be surprised if the President’s vaccine mandate actually reduces the nation’s military readiness instead of promoting it.”
In a press briefing, Pentagon press secretary John Kirby said, “The Secretary has the authorities he needs to require this vaccine across the force, including the National Guard. …[E]ven when they’re in a Title 32 status.” He added, “It is a lawful order for National Guardsmen to receive the COVID vaccine. It’s a lawful order, and refusing to do that, absent of an improved exemption, puts them in the same potential [position] as active-duty members who refuse the vaccine.” That could mean, for instance, loss of pay and membership in the National Guard.
A core rationale for the mandate, according to Secretary Austin, is the need for military readiness—meaning Guard members must be healthy and fit for duty. And that extends to being healthy and fit for missions like transporting at-risk patients. Ohio National Guard Adjutant General Major General John Harris Jr. said, “I would never put a soldier or airman in harm’s way without the best protection we could put on them—body armor, helmets. And this medical readiness is the exact same thing. We’re putting folks into harm’s way.” He has moved the deadline from the Pentagon’s June 30 date to March 31—a move that boosted the vaccination rate from 53% to 56% in one week.
Ohio Governor DeWine has expressed frustration that almost half of the Ohio Army National Guard personnel can’t be deployed on this mission because they’re unvaccinated. “In some of our testing places, 40 to 50% of the people are testing positive,” he said. “So this is a high-risk operation. You need to be protected. The best way for you to be protected is to get the vaccination.”
As of December 2021, according to the National Guard Bureau, the National Guard as a whole was 66% fully vaccinated. The percentages vary according to service; for instance, nearly 90% of airmen have been vaccinated, compared with only 40% of Army Guardsmen. Among the states challenging the mandate, the vaccinated rates have been moving upward: In Alaska, about 92% of the Air National Guard have been vaccinated—leaving roughly 11,000 troops who had not met the December 2 deadline. In Iowa, as of Nov. 30, 91% of Air National Guard and 80% of Army National Guard members had been vaccinated, but about 9,000 soldiers had been directed to get the vaccination or risk disciplinary action. Almost 2,200 of the more than 2,800-strong Wyoming National Guard (77%) have received at least 1 dose. Nebraska Air National Guard’s force of 1,000 was 94% fully vaccinated as of December 1. (Maj Scott Ingalsbe, public affairs officer, said, “Vaccinations are tied to individual medical readiness. They provide service members with the best protection available so they can perform missions across the globe.”).
In most states, Army National Guard members have until June 30, 2022, to comply. “Our soldiers …have until [the DoD’s deadline], and some of them are just going to wait close to the deadline,” John Goheen of the National Guard Association of the United States said in a discussion on NPR. “That’s human nature.”
Earlier this month, Texas Governor Greg Abbott told National Guard members they can ignore the Pentagon’s COVID-19 vaccine mandate: “President Biden is not your commander-in-chief.” He has also sued the Biden administration over the requirement.
In the meantime, the hospitals at breaking point must hope for the best and take as much help as they can get.
What if the National Guard Can’t Help?
What if the National Guard Can’t Help?
Are there perinatal benefits to pregnant patients after bariatric surgery?
Getahun D, Fassett MJ, Jacobsen SJ, et al. Perinatal outcomes after bariatric surgery. Am J Obstet Gynecol. 2021;S0002-9378(21)00771-7. doi: 10.1016/j.ajog.2021 .06.087.
EXPERT COMMENTARY
Prepregnancy obesity continues to rise in the United States, with a prevalence of 29% among reproductive-age women in 2019, an 11% increase from 2016.1 Pregnant patients with obesity are at increased risk for multiple adverse perinatal outcomes, including gestational diabetes and preeclampsia. Bariatric surgery is effective for weight loss and has been shown to improve comorbidities associated with obesity,2 and it may have potential benefits for pregnancy outcomes, such as reducing rates of gestational diabetes and preeclampsia.3-5 However, little was known about other outcomes as well as other potential factors before a recent study in which investigators examined perinatal outcomes after bariatric surgery.
Details of the study
Getahun and colleagues conducted a population-based, retrospective study of pregnant patients who were eligible for bariatric surgery (body mass index [BMI] ≥40 kg/m2 with no comorbidities or a BMI between 35 and 40 kg/m2 with obesity-related comorbidities, such as diabetes). They aimed to evaluate the association of bariatric surgery with adverse perinatal outcomes.
Results. In a large sample of pregnant patients eligible for bariatric surgery (N = 20,213), the authors found that patients who had bariatric surgery (n = 1,886) had a reduced risk of macrosomia (aOR, 0.24), preeclampsia (aOR, 0.53), gestational diabetes (aOR, 0.60), and cesarean delivery (aOR, 0.65) compared with those who did not have bariatric surgery (n = 18,327). They also found that patients who had bariatric surgery had an increased risk of small-for-gestational age neonates (aOR, 2.46) and postpartum hemorrhage (aOR, 1.79).
These results remained after adjusting for other potential confounders. The authors evaluated the outcomes based on the timing of surgery and the patients’ pregnancy (<1 year, 1-1.5 years, 1.5-2 years, >2 years). The outcomes were more favorable among the patients who had the bariatric surgery regardless of the time interval of surgery to pregnancy than those who did not have the surgery. In addition, the benefits of bariatric surgery did not differ between the 2 most common types of bariatric surgery (Roux-en-Y gastric bypass and vertical sleeve gastrectomy) performed in this study, and both had better outcomes than those who did not have the surgery. Finally, patients with chronic hypertension and pregestational diabetes who had bariatric surgery also had lower risks of adverse outcomes than those without bariatric surgery.
Study strengths and limitations
Given the study’s retrospective design, uncertainties and important confounders could not be addressed, such as why certain eligible patients had the surgery and others did not. However, with its large sample size and an appropriate comparison group, the study findings further support the perinatal benefits of bariatric surgery in obese patients. Of note, this study also had a large sample of Black and Hispanic patients, populations known to have higher rates of obesity1 and pregnancy complications. Subgroup analyses within each racial/ethnic group revealed that those who had the surgery had lower risks of adverse perinatal outcomes than those who did not.
Patients who had the bariatric surgery had an increased risk of postpartum hemorrhage; however, there is no physiologic basis or theory to explain this finding, so further studies are needed. Lastly, although patients who had bariatric surgery had an increased risk of small-for-gestational-age babies and the study was not powered for the risk of stillbirth, the patients who had the surgery had a reduced risk of neonates admitted to the neonatal intensive care unit. More data would have been beneficial to assess if these small-for-gestational-age babies were healthy. In general, obese patients tend to have larger and unhealthy babies; thus, healthier babies, even if small for gestational age, would not be an adverse outcome.
Benefits of bariatric surgery extend to perinatal outcomes
This study reinforces current practice that includes eligible patients being counseled about the health-related benefits of bariatric surgery, which now includes more perinatal outcomes. The finding of the increased risk of small-for-gestational-age fetuses supports the practice of a screening growth ultrasound exam in patients who had bariatric surgery. ●
An important, modifiable risk factor for adverse perinatal outcomes is the patient’s prepregnancy BMI at the time of pregnancy. Bariatric surgery is an effective procedure for weight loss. There are many perinatal benefits for eligible patients who have bariatric surgery before pregnancy. Clinicians should counsel their obese patients who are considering or planning pregnancy about the benefits of bariatric surgery.
RODNEY A. MCLAREN, JR, MD, AND VINCENZO BERGHELLA, MD
- Driscoll AK, Gregory ECW. Increases in prepregnancy obesity: United States, 2016-2019. NCHS Data Brief. 2020 Nov;(392):1-8.
- Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA. 2004;292:1724- 1737. doi: 10.1001/jama.292.14.1724.
- Maggard MA, Yermilov I, Li Z, et al. Pregnancy and fertility following bariatric surgery: a systematic review. JAMA. 2008;300:2286-2296. doi: 10.1001/jama.2008.641.
- Watanabe A, Seki Y, Haruta H, et al. Maternal impacts and perinatal outcomes after three types of bariatric surgery at a single institution. Arch Gynecol Obstet. 2019;300:145-152. doi: 10.1007/s00404-019-05195-9.
- Balestrin B, Urbanetz AA, Barbieri MM, et al. Pregnancy after bariatric surgery: a comparative study of post-bariatric pregnant women versus non-bariatric obese pregnant women. Obes Surg. 2019;29:3142-3148. doi: 10.1007/s11695- 019-03961-x.
Getahun D, Fassett MJ, Jacobsen SJ, et al. Perinatal outcomes after bariatric surgery. Am J Obstet Gynecol. 2021;S0002-9378(21)00771-7. doi: 10.1016/j.ajog.2021 .06.087.
EXPERT COMMENTARY
Prepregnancy obesity continues to rise in the United States, with a prevalence of 29% among reproductive-age women in 2019, an 11% increase from 2016.1 Pregnant patients with obesity are at increased risk for multiple adverse perinatal outcomes, including gestational diabetes and preeclampsia. Bariatric surgery is effective for weight loss and has been shown to improve comorbidities associated with obesity,2 and it may have potential benefits for pregnancy outcomes, such as reducing rates of gestational diabetes and preeclampsia.3-5 However, little was known about other outcomes as well as other potential factors before a recent study in which investigators examined perinatal outcomes after bariatric surgery.
Details of the study
Getahun and colleagues conducted a population-based, retrospective study of pregnant patients who were eligible for bariatric surgery (body mass index [BMI] ≥40 kg/m2 with no comorbidities or a BMI between 35 and 40 kg/m2 with obesity-related comorbidities, such as diabetes). They aimed to evaluate the association of bariatric surgery with adverse perinatal outcomes.
Results. In a large sample of pregnant patients eligible for bariatric surgery (N = 20,213), the authors found that patients who had bariatric surgery (n = 1,886) had a reduced risk of macrosomia (aOR, 0.24), preeclampsia (aOR, 0.53), gestational diabetes (aOR, 0.60), and cesarean delivery (aOR, 0.65) compared with those who did not have bariatric surgery (n = 18,327). They also found that patients who had bariatric surgery had an increased risk of small-for-gestational age neonates (aOR, 2.46) and postpartum hemorrhage (aOR, 1.79).
These results remained after adjusting for other potential confounders. The authors evaluated the outcomes based on the timing of surgery and the patients’ pregnancy (<1 year, 1-1.5 years, 1.5-2 years, >2 years). The outcomes were more favorable among the patients who had the bariatric surgery regardless of the time interval of surgery to pregnancy than those who did not have the surgery. In addition, the benefits of bariatric surgery did not differ between the 2 most common types of bariatric surgery (Roux-en-Y gastric bypass and vertical sleeve gastrectomy) performed in this study, and both had better outcomes than those who did not have the surgery. Finally, patients with chronic hypertension and pregestational diabetes who had bariatric surgery also had lower risks of adverse outcomes than those without bariatric surgery.
Study strengths and limitations
Given the study’s retrospective design, uncertainties and important confounders could not be addressed, such as why certain eligible patients had the surgery and others did not. However, with its large sample size and an appropriate comparison group, the study findings further support the perinatal benefits of bariatric surgery in obese patients. Of note, this study also had a large sample of Black and Hispanic patients, populations known to have higher rates of obesity1 and pregnancy complications. Subgroup analyses within each racial/ethnic group revealed that those who had the surgery had lower risks of adverse perinatal outcomes than those who did not.
Patients who had the bariatric surgery had an increased risk of postpartum hemorrhage; however, there is no physiologic basis or theory to explain this finding, so further studies are needed. Lastly, although patients who had bariatric surgery had an increased risk of small-for-gestational-age babies and the study was not powered for the risk of stillbirth, the patients who had the surgery had a reduced risk of neonates admitted to the neonatal intensive care unit. More data would have been beneficial to assess if these small-for-gestational-age babies were healthy. In general, obese patients tend to have larger and unhealthy babies; thus, healthier babies, even if small for gestational age, would not be an adverse outcome.
Benefits of bariatric surgery extend to perinatal outcomes
This study reinforces current practice that includes eligible patients being counseled about the health-related benefits of bariatric surgery, which now includes more perinatal outcomes. The finding of the increased risk of small-for-gestational-age fetuses supports the practice of a screening growth ultrasound exam in patients who had bariatric surgery. ●
An important, modifiable risk factor for adverse perinatal outcomes is the patient’s prepregnancy BMI at the time of pregnancy. Bariatric surgery is an effective procedure for weight loss. There are many perinatal benefits for eligible patients who have bariatric surgery before pregnancy. Clinicians should counsel their obese patients who are considering or planning pregnancy about the benefits of bariatric surgery.
RODNEY A. MCLAREN, JR, MD, AND VINCENZO BERGHELLA, MD
Getahun D, Fassett MJ, Jacobsen SJ, et al. Perinatal outcomes after bariatric surgery. Am J Obstet Gynecol. 2021;S0002-9378(21)00771-7. doi: 10.1016/j.ajog.2021 .06.087.
EXPERT COMMENTARY
Prepregnancy obesity continues to rise in the United States, with a prevalence of 29% among reproductive-age women in 2019, an 11% increase from 2016.1 Pregnant patients with obesity are at increased risk for multiple adverse perinatal outcomes, including gestational diabetes and preeclampsia. Bariatric surgery is effective for weight loss and has been shown to improve comorbidities associated with obesity,2 and it may have potential benefits for pregnancy outcomes, such as reducing rates of gestational diabetes and preeclampsia.3-5 However, little was known about other outcomes as well as other potential factors before a recent study in which investigators examined perinatal outcomes after bariatric surgery.
Details of the study
Getahun and colleagues conducted a population-based, retrospective study of pregnant patients who were eligible for bariatric surgery (body mass index [BMI] ≥40 kg/m2 with no comorbidities or a BMI between 35 and 40 kg/m2 with obesity-related comorbidities, such as diabetes). They aimed to evaluate the association of bariatric surgery with adverse perinatal outcomes.
Results. In a large sample of pregnant patients eligible for bariatric surgery (N = 20,213), the authors found that patients who had bariatric surgery (n = 1,886) had a reduced risk of macrosomia (aOR, 0.24), preeclampsia (aOR, 0.53), gestational diabetes (aOR, 0.60), and cesarean delivery (aOR, 0.65) compared with those who did not have bariatric surgery (n = 18,327). They also found that patients who had bariatric surgery had an increased risk of small-for-gestational age neonates (aOR, 2.46) and postpartum hemorrhage (aOR, 1.79).
These results remained after adjusting for other potential confounders. The authors evaluated the outcomes based on the timing of surgery and the patients’ pregnancy (<1 year, 1-1.5 years, 1.5-2 years, >2 years). The outcomes were more favorable among the patients who had the bariatric surgery regardless of the time interval of surgery to pregnancy than those who did not have the surgery. In addition, the benefits of bariatric surgery did not differ between the 2 most common types of bariatric surgery (Roux-en-Y gastric bypass and vertical sleeve gastrectomy) performed in this study, and both had better outcomes than those who did not have the surgery. Finally, patients with chronic hypertension and pregestational diabetes who had bariatric surgery also had lower risks of adverse outcomes than those without bariatric surgery.
Study strengths and limitations
Given the study’s retrospective design, uncertainties and important confounders could not be addressed, such as why certain eligible patients had the surgery and others did not. However, with its large sample size and an appropriate comparison group, the study findings further support the perinatal benefits of bariatric surgery in obese patients. Of note, this study also had a large sample of Black and Hispanic patients, populations known to have higher rates of obesity1 and pregnancy complications. Subgroup analyses within each racial/ethnic group revealed that those who had the surgery had lower risks of adverse perinatal outcomes than those who did not.
Patients who had the bariatric surgery had an increased risk of postpartum hemorrhage; however, there is no physiologic basis or theory to explain this finding, so further studies are needed. Lastly, although patients who had bariatric surgery had an increased risk of small-for-gestational-age babies and the study was not powered for the risk of stillbirth, the patients who had the surgery had a reduced risk of neonates admitted to the neonatal intensive care unit. More data would have been beneficial to assess if these small-for-gestational-age babies were healthy. In general, obese patients tend to have larger and unhealthy babies; thus, healthier babies, even if small for gestational age, would not be an adverse outcome.
Benefits of bariatric surgery extend to perinatal outcomes
This study reinforces current practice that includes eligible patients being counseled about the health-related benefits of bariatric surgery, which now includes more perinatal outcomes. The finding of the increased risk of small-for-gestational-age fetuses supports the practice of a screening growth ultrasound exam in patients who had bariatric surgery. ●
An important, modifiable risk factor for adverse perinatal outcomes is the patient’s prepregnancy BMI at the time of pregnancy. Bariatric surgery is an effective procedure for weight loss. There are many perinatal benefits for eligible patients who have bariatric surgery before pregnancy. Clinicians should counsel their obese patients who are considering or planning pregnancy about the benefits of bariatric surgery.
RODNEY A. MCLAREN, JR, MD, AND VINCENZO BERGHELLA, MD
- Driscoll AK, Gregory ECW. Increases in prepregnancy obesity: United States, 2016-2019. NCHS Data Brief. 2020 Nov;(392):1-8.
- Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA. 2004;292:1724- 1737. doi: 10.1001/jama.292.14.1724.
- Maggard MA, Yermilov I, Li Z, et al. Pregnancy and fertility following bariatric surgery: a systematic review. JAMA. 2008;300:2286-2296. doi: 10.1001/jama.2008.641.
- Watanabe A, Seki Y, Haruta H, et al. Maternal impacts and perinatal outcomes after three types of bariatric surgery at a single institution. Arch Gynecol Obstet. 2019;300:145-152. doi: 10.1007/s00404-019-05195-9.
- Balestrin B, Urbanetz AA, Barbieri MM, et al. Pregnancy after bariatric surgery: a comparative study of post-bariatric pregnant women versus non-bariatric obese pregnant women. Obes Surg. 2019;29:3142-3148. doi: 10.1007/s11695- 019-03961-x.
- Driscoll AK, Gregory ECW. Increases in prepregnancy obesity: United States, 2016-2019. NCHS Data Brief. 2020 Nov;(392):1-8.
- Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA. 2004;292:1724- 1737. doi: 10.1001/jama.292.14.1724.
- Maggard MA, Yermilov I, Li Z, et al. Pregnancy and fertility following bariatric surgery: a systematic review. JAMA. 2008;300:2286-2296. doi: 10.1001/jama.2008.641.
- Watanabe A, Seki Y, Haruta H, et al. Maternal impacts and perinatal outcomes after three types of bariatric surgery at a single institution. Arch Gynecol Obstet. 2019;300:145-152. doi: 10.1007/s00404-019-05195-9.
- Balestrin B, Urbanetz AA, Barbieri MM, et al. Pregnancy after bariatric surgery: a comparative study of post-bariatric pregnant women versus non-bariatric obese pregnant women. Obes Surg. 2019;29:3142-3148. doi: 10.1007/s11695- 019-03961-x.
Ubrogepant beneficial in migraine regardless of prior preventive medication use
Key clinical point: Ubrogepant was effective and safe in patients with migraine, regardless of prior or concomitant preventive medication use.
Major finding: Ubrogepant vs. placebo was associated with significantly higher responder rates for pain freedom (P £ .005), absence of most bothersome symptom (50 mg ubrogepant vs. placebo; P £ .001), and pain relief (P £ .011) at 2 hours, with the responder rates not being significantly different among patients with vs. without preventive medication use (all P > .05). No serious or treatment-related adverse events were reported.
Study details: Findings are from a pooled analysis of ACHIEVE I and ACHIEVE II phase 3 trials (n = 2,247) and the long-term safety extension trial (n = 813) including patients with migraine with or without a history of preventive medication use.
Disclosures: This study was funded by Allergan (before its acquisition by AbbVie). Some investigators, including the lead author, reported advising or consulting for; being a speaker, on the board of directors, or a contributing author for; receiving grants, research support, and honoraria from; holding stocks or patents with; or employment with various sources including Allergan and AbbVie.
Source: Blumenfeld AM et al. Add Ther. 2021 (Dec 7). Doi: 10.1007/s12325-021-01923-3.
Key clinical point: Ubrogepant was effective and safe in patients with migraine, regardless of prior or concomitant preventive medication use.
Major finding: Ubrogepant vs. placebo was associated with significantly higher responder rates for pain freedom (P £ .005), absence of most bothersome symptom (50 mg ubrogepant vs. placebo; P £ .001), and pain relief (P £ .011) at 2 hours, with the responder rates not being significantly different among patients with vs. without preventive medication use (all P > .05). No serious or treatment-related adverse events were reported.
Study details: Findings are from a pooled analysis of ACHIEVE I and ACHIEVE II phase 3 trials (n = 2,247) and the long-term safety extension trial (n = 813) including patients with migraine with or without a history of preventive medication use.
Disclosures: This study was funded by Allergan (before its acquisition by AbbVie). Some investigators, including the lead author, reported advising or consulting for; being a speaker, on the board of directors, or a contributing author for; receiving grants, research support, and honoraria from; holding stocks or patents with; or employment with various sources including Allergan and AbbVie.
Source: Blumenfeld AM et al. Add Ther. 2021 (Dec 7). Doi: 10.1007/s12325-021-01923-3.
Key clinical point: Ubrogepant was effective and safe in patients with migraine, regardless of prior or concomitant preventive medication use.
Major finding: Ubrogepant vs. placebo was associated with significantly higher responder rates for pain freedom (P £ .005), absence of most bothersome symptom (50 mg ubrogepant vs. placebo; P £ .001), and pain relief (P £ .011) at 2 hours, with the responder rates not being significantly different among patients with vs. without preventive medication use (all P > .05). No serious or treatment-related adverse events were reported.
Study details: Findings are from a pooled analysis of ACHIEVE I and ACHIEVE II phase 3 trials (n = 2,247) and the long-term safety extension trial (n = 813) including patients with migraine with or without a history of preventive medication use.
Disclosures: This study was funded by Allergan (before its acquisition by AbbVie). Some investigators, including the lead author, reported advising or consulting for; being a speaker, on the board of directors, or a contributing author for; receiving grants, research support, and honoraria from; holding stocks or patents with; or employment with various sources including Allergan and AbbVie.
Source: Blumenfeld AM et al. Add Ther. 2021 (Dec 7). Doi: 10.1007/s12325-021-01923-3.