User login
Keeping an open mind about functional medicine
Considering the controversy surrounding functional medicine, you may be wondering why JFP published an article about it last month.1 David Gorski, MD, PhD, FACS, a vocal critic of functional medicine, commented: “Functional medicine. It sounds so … scientific and reasonable. It’s anything but. In fact, functional medicine combines the worst features of conventional medicine with a heapin’ helpin’ of quackery.”2 On its website, however, The Institute for Functional Medicine claims that “functional medicine determines how and why illness occurs and restores health by addressing the root causes of disease for each individual.”3
I suspect the truth lies somewhere in between.
Because functional medicine has gained a certain degree of popularity, I felt it was important for family physicians and other primary care clinicians to know enough about this alternative healing method to discuss it with patients who express interest.
In their review article in JFP, Orlando and colleagues tell us there are 7 defining characteristics of functional medicine.1 It is patient centered rather than disease centered, uses a “systems biology” approach, considers the dynamic balance of gene-environment interactions, is personalized based on biochemical individuality, promotes organ reserve and sustained health span, sees health as a positive vitality (not merely the absence of disease), and focuses on function rather than pathology.
Most of these statements about functional medicine apply to traditional family medicine. The clinical approach stressing lifestyle changes is mainstream, not unique. The focus on digestion and the microbiome as an important determinant of health is based on interesting basic science studies and associations noted between certain microbiome profiles and diseases.
But association is not causation. So far there is scant evidence that changing the microbiome results in better health, although some preliminary case series have generated intriguing hypotheses. And there is evidence that probiotics improve some symptoms. Ongoing research into the microbiome and health will, no doubt, be illuminating. We have much to learn.
What does seem unique, but suspect, about functional medicine is its focus on biochemical testing of unproven value and the prescribing of diets and supplements based on the test results. There are no sound scientific studies showing the benefit of this approach.
I suggest you read Orlando et al’s article. Functional medicine is an interesting, mostly unproven, approach to patient care. But I will keep an open mind until we see better research that either does—or doesn’t—support the validity of its practices.
1. Orlando FA, Chang KL, Estores IM. Functional medicine: focusing on imbalances in core metabolic processes. J Fam Pract. 2021;70:482-488,498.
2. Gorski D. Functional medicine: the ultimate misnomer in the world of integrative medicine. Science-Based Medicine. April 11, 2016. Accessed January 4, 2022. https://sciencebasedmedicine.org/functional-medicine-the-ultimate-misnomer-in-the-world-of-integrative-medicine/
3. The Institute for Functional Medicine. Accessed January 4, 2022. www.ifm.org
Considering the controversy surrounding functional medicine, you may be wondering why JFP published an article about it last month.1 David Gorski, MD, PhD, FACS, a vocal critic of functional medicine, commented: “Functional medicine. It sounds so … scientific and reasonable. It’s anything but. In fact, functional medicine combines the worst features of conventional medicine with a heapin’ helpin’ of quackery.”2 On its website, however, The Institute for Functional Medicine claims that “functional medicine determines how and why illness occurs and restores health by addressing the root causes of disease for each individual.”3
I suspect the truth lies somewhere in between.
Because functional medicine has gained a certain degree of popularity, I felt it was important for family physicians and other primary care clinicians to know enough about this alternative healing method to discuss it with patients who express interest.
In their review article in JFP, Orlando and colleagues tell us there are 7 defining characteristics of functional medicine.1 It is patient centered rather than disease centered, uses a “systems biology” approach, considers the dynamic balance of gene-environment interactions, is personalized based on biochemical individuality, promotes organ reserve and sustained health span, sees health as a positive vitality (not merely the absence of disease), and focuses on function rather than pathology.
Most of these statements about functional medicine apply to traditional family medicine. The clinical approach stressing lifestyle changes is mainstream, not unique. The focus on digestion and the microbiome as an important determinant of health is based on interesting basic science studies and associations noted between certain microbiome profiles and diseases.
But association is not causation. So far there is scant evidence that changing the microbiome results in better health, although some preliminary case series have generated intriguing hypotheses. And there is evidence that probiotics improve some symptoms. Ongoing research into the microbiome and health will, no doubt, be illuminating. We have much to learn.
What does seem unique, but suspect, about functional medicine is its focus on biochemical testing of unproven value and the prescribing of diets and supplements based on the test results. There are no sound scientific studies showing the benefit of this approach.
I suggest you read Orlando et al’s article. Functional medicine is an interesting, mostly unproven, approach to patient care. But I will keep an open mind until we see better research that either does—or doesn’t—support the validity of its practices.
Considering the controversy surrounding functional medicine, you may be wondering why JFP published an article about it last month.1 David Gorski, MD, PhD, FACS, a vocal critic of functional medicine, commented: “Functional medicine. It sounds so … scientific and reasonable. It’s anything but. In fact, functional medicine combines the worst features of conventional medicine with a heapin’ helpin’ of quackery.”2 On its website, however, The Institute for Functional Medicine claims that “functional medicine determines how and why illness occurs and restores health by addressing the root causes of disease for each individual.”3
I suspect the truth lies somewhere in between.
Because functional medicine has gained a certain degree of popularity, I felt it was important for family physicians and other primary care clinicians to know enough about this alternative healing method to discuss it with patients who express interest.
In their review article in JFP, Orlando and colleagues tell us there are 7 defining characteristics of functional medicine.1 It is patient centered rather than disease centered, uses a “systems biology” approach, considers the dynamic balance of gene-environment interactions, is personalized based on biochemical individuality, promotes organ reserve and sustained health span, sees health as a positive vitality (not merely the absence of disease), and focuses on function rather than pathology.
Most of these statements about functional medicine apply to traditional family medicine. The clinical approach stressing lifestyle changes is mainstream, not unique. The focus on digestion and the microbiome as an important determinant of health is based on interesting basic science studies and associations noted between certain microbiome profiles and diseases.
But association is not causation. So far there is scant evidence that changing the microbiome results in better health, although some preliminary case series have generated intriguing hypotheses. And there is evidence that probiotics improve some symptoms. Ongoing research into the microbiome and health will, no doubt, be illuminating. We have much to learn.
What does seem unique, but suspect, about functional medicine is its focus on biochemical testing of unproven value and the prescribing of diets and supplements based on the test results. There are no sound scientific studies showing the benefit of this approach.
I suggest you read Orlando et al’s article. Functional medicine is an interesting, mostly unproven, approach to patient care. But I will keep an open mind until we see better research that either does—or doesn’t—support the validity of its practices.
1. Orlando FA, Chang KL, Estores IM. Functional medicine: focusing on imbalances in core metabolic processes. J Fam Pract. 2021;70:482-488,498.
2. Gorski D. Functional medicine: the ultimate misnomer in the world of integrative medicine. Science-Based Medicine. April 11, 2016. Accessed January 4, 2022. https://sciencebasedmedicine.org/functional-medicine-the-ultimate-misnomer-in-the-world-of-integrative-medicine/
3. The Institute for Functional Medicine. Accessed January 4, 2022. www.ifm.org
1. Orlando FA, Chang KL, Estores IM. Functional medicine: focusing on imbalances in core metabolic processes. J Fam Pract. 2021;70:482-488,498.
2. Gorski D. Functional medicine: the ultimate misnomer in the world of integrative medicine. Science-Based Medicine. April 11, 2016. Accessed January 4, 2022. https://sciencebasedmedicine.org/functional-medicine-the-ultimate-misnomer-in-the-world-of-integrative-medicine/
3. The Institute for Functional Medicine. Accessed January 4, 2022. www.ifm.org
Nodule on the left cheek
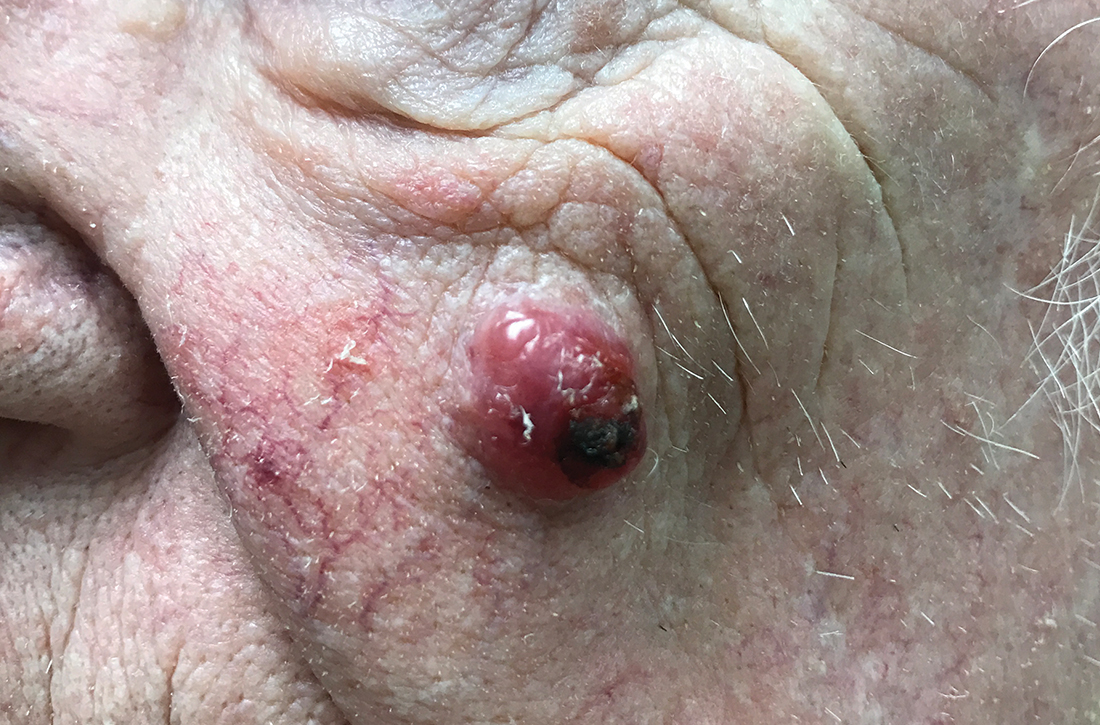
An 85-year-old man with a history of skin cancer presented to my dermatology practice (NT) for evaluation of a “pimple” on his left cheek that failed to resolve after 2 months (FIGURE). The patient noted that the lesion had grown, but that he otherwise felt well.
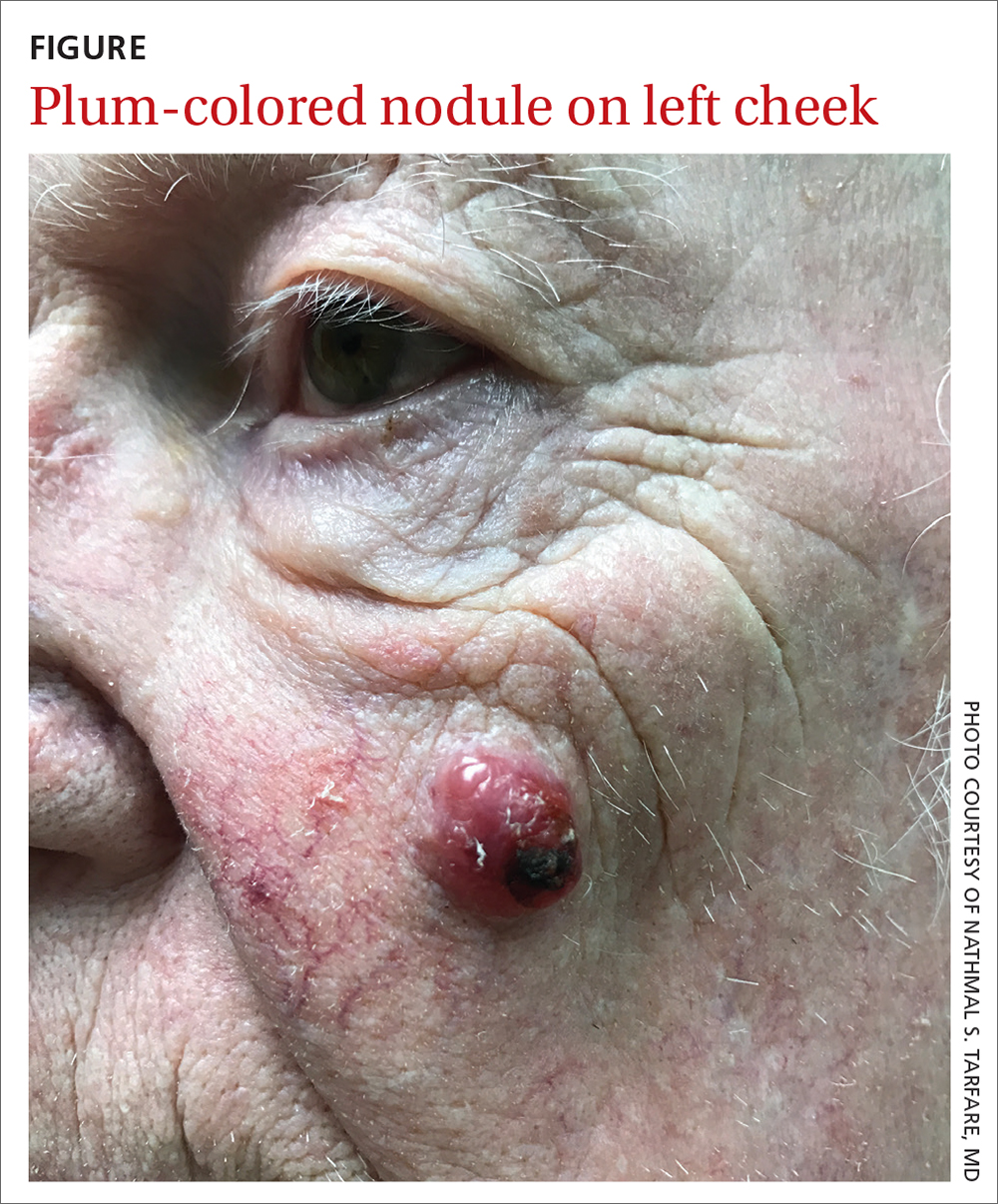
On examination, the lesion was plum colored, and the area was firm and nontender to palpation. The patient was referred to a plastic surgeon for an excisional biopsy to clarify the nature of the lesion.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Merkel cell carcinoma
A biopsy performed 2 weeks after the initial visit confirmed the clinical suspicion for Merkel cell carcinoma (MCC).
MCC is a cutaneous neuroendocrine malignancy. Although its name acknowledges similarities between the tumor cells and Merkel cells, it is now considered unlikely that Merkel cells are the actual cells of origin.1
The majority of MCCs are asymptomatic despite rapid growth and are typically red or pink and occur on UV-exposed areas, as in our patient.2 A cyst or acneiform lesion is the single most common diagnosis given at the time of biopsy.2
The incidence of MCC is greatest in people of advanced age and in those who are immunosuppressed. In the United States, the estimated annual incidence rate rose from 0.5 cases per 100,000 people in 2000 to 0.7 cases per 100,000 people in 2013.3 MCC increases exponentially with advancing age, from 0.1 (per 100,000) in those ages 40 to 44 years to 9.8 in those older than 85 years.3 The growing cohort of ageing baby boomers and the increased number of immunosuppressed individuals in the community suggest that clinicians are now more likely to encounter MCC than in the past.
While UV radiation is highly associated with MCC, the major causative factor is considered to be Merkel cell polyomavirus (MCPyV).1 In fact, MCPyV has been linked to 80% of MCC cases.1,3 Most people have positive serology for MCPyV in early childhood, but the association between MCC and old age highlights the impact of immunosuppression on MCPyV activity and MCC development.1
Clinical suspicion is the first step in diagnosing MCC
The mnemonic AEIOU highlights the key clinical features of this aggressive tumor2,4:
- Asymptomatic
- Expanding rapidly (often grows in less than 3 months)
- Immune suppression (eg, chronic lymphocytic leukemia, solid organ transplant patient)
- Older than 50
- UV exposure on fair skin.
If a lesion is suspected to be MCC, the next step includes biopsy so that a definitive diagnosis can be made. A firm, nontender nodule that lacks fluctuance should raise suspicion for a neoplastic process.
Continue to: The differential is broad, ranging from cysts to melanoma
The differential is broad, ranging from cysts to melanoma
The differential diagnosis for an enlarging, plum-colored nodule on sun-exposed skin includes an abscess, a ruptured or inflamed epidermoid cyst, basal cell carcinoma, squamous cell carcinoma, and malignant melanoma.
An abscess is typically tender and expands within a matter of days rather than months.
A cyst can be ruled out by the clinical appearance and lack of an overlying pore.
Basal cell carcinoma can be characterized by a rolled border and central ulceration.
Squamous cell carcinomas often exhibit a verrucous surface with marked hyperkeratosis.
Continue to: Melanoma
Melanoma manifests with brown or irregular pigmentation and may be associated with a precursor lesion.
Tx includes excision and consistent follow-up
Complete excision is the critical first step to successful therapy. Sentinel lymph node studies are typically performed because of the high incidence of lymph node metastasis. Frequent follow-up is required because of the high risk of recurrent or persistent disease.
Local recurrence usually occurs within 1 year of diagnosis in more than 40% of patients.5 Distant metastasis can be treated with a programmed cell death ligand 1 blocking agent (avelumab) or a programmed cell death protein 1 inhibitor (nivolumab or pembrolizumab).6
Our patient was referred to a regional cancer center for sentinel lymph node evaluation, where he was found to have nodal disease. The patient was put on pembrolizumab and received radiation therapy but showed only limited response. Seven months after diagnosis, he passed away from metastatic MCC.
1. Pietropaolo V, Prezioso C, Moens U. Merkel cell polyomavirus and Merkel cell carcinoma. Cancers (Basel). 2020;12:1774. doi: 10.3390/cancers12071774
2. Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375-381. doi: 10.1016/j.jaad.2007.11.020
3. Paulson KG, Park SY, Vandeven NA, et al. Merkel cell carcinoma: current US incidence and projected increases based on changing demographics. J Am Acad Dermatol. 2018;78:457-463.e2. doi: 10.1016/j.jaad.2017.10.028
4. Voelker R. Why Merkel cell cancer is garnering more attention. JAMA. 2018;320:18-20. doi: 10.1001/jama.2018.7042
5. Allen PJ, Browne WB, Jacques DP, et al. Merkel cell carcinoma: prognosis and treatment of patients from a single institution. J Clin Oncol. 2005;23:2300-2309. doi: 10.1200/JCO.2005.02.329
6. D’Angelo SP, Russell J, Lebbé C, et al. Efficacy and safety of first-line avelumab treatment in patients with stage IV metastatic Merkel cell carcinoma: a preplanned interim analysis of a clinical trial. JAMA Oncol. 2018;4:e180077. doi: 10.1001/jamaoncol.2018.0077
An 85-year-old man with a history of skin cancer presented to my dermatology practice (NT) for evaluation of a “pimple” on his left cheek that failed to resolve after 2 months (FIGURE). The patient noted that the lesion had grown, but that he otherwise felt well.

On examination, the lesion was plum colored, and the area was firm and nontender to palpation. The patient was referred to a plastic surgeon for an excisional biopsy to clarify the nature of the lesion.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Merkel cell carcinoma
A biopsy performed 2 weeks after the initial visit confirmed the clinical suspicion for Merkel cell carcinoma (MCC).
MCC is a cutaneous neuroendocrine malignancy. Although its name acknowledges similarities between the tumor cells and Merkel cells, it is now considered unlikely that Merkel cells are the actual cells of origin.1
The majority of MCCs are asymptomatic despite rapid growth and are typically red or pink and occur on UV-exposed areas, as in our patient.2 A cyst or acneiform lesion is the single most common diagnosis given at the time of biopsy.2
The incidence of MCC is greatest in people of advanced age and in those who are immunosuppressed. In the United States, the estimated annual incidence rate rose from 0.5 cases per 100,000 people in 2000 to 0.7 cases per 100,000 people in 2013.3 MCC increases exponentially with advancing age, from 0.1 (per 100,000) in those ages 40 to 44 years to 9.8 in those older than 85 years.3 The growing cohort of ageing baby boomers and the increased number of immunosuppressed individuals in the community suggest that clinicians are now more likely to encounter MCC than in the past.
While UV radiation is highly associated with MCC, the major causative factor is considered to be Merkel cell polyomavirus (MCPyV).1 In fact, MCPyV has been linked to 80% of MCC cases.1,3 Most people have positive serology for MCPyV in early childhood, but the association between MCC and old age highlights the impact of immunosuppression on MCPyV activity and MCC development.1
Clinical suspicion is the first step in diagnosing MCC
The mnemonic AEIOU highlights the key clinical features of this aggressive tumor2,4:
- Asymptomatic
- Expanding rapidly (often grows in less than 3 months)
- Immune suppression (eg, chronic lymphocytic leukemia, solid organ transplant patient)
- Older than 50
- UV exposure on fair skin.
If a lesion is suspected to be MCC, the next step includes biopsy so that a definitive diagnosis can be made. A firm, nontender nodule that lacks fluctuance should raise suspicion for a neoplastic process.
Continue to: The differential is broad, ranging from cysts to melanoma
The differential is broad, ranging from cysts to melanoma
The differential diagnosis for an enlarging, plum-colored nodule on sun-exposed skin includes an abscess, a ruptured or inflamed epidermoid cyst, basal cell carcinoma, squamous cell carcinoma, and malignant melanoma.
An abscess is typically tender and expands within a matter of days rather than months.
A cyst can be ruled out by the clinical appearance and lack of an overlying pore.
Basal cell carcinoma can be characterized by a rolled border and central ulceration.
Squamous cell carcinomas often exhibit a verrucous surface with marked hyperkeratosis.
Continue to: Melanoma
Melanoma manifests with brown or irregular pigmentation and may be associated with a precursor lesion.
Tx includes excision and consistent follow-up
Complete excision is the critical first step to successful therapy. Sentinel lymph node studies are typically performed because of the high incidence of lymph node metastasis. Frequent follow-up is required because of the high risk of recurrent or persistent disease.
Local recurrence usually occurs within 1 year of diagnosis in more than 40% of patients.5 Distant metastasis can be treated with a programmed cell death ligand 1 blocking agent (avelumab) or a programmed cell death protein 1 inhibitor (nivolumab or pembrolizumab).6
Our patient was referred to a regional cancer center for sentinel lymph node evaluation, where he was found to have nodal disease. The patient was put on pembrolizumab and received radiation therapy but showed only limited response. Seven months after diagnosis, he passed away from metastatic MCC.
An 85-year-old man with a history of skin cancer presented to my dermatology practice (NT) for evaluation of a “pimple” on his left cheek that failed to resolve after 2 months (FIGURE). The patient noted that the lesion had grown, but that he otherwise felt well.

On examination, the lesion was plum colored, and the area was firm and nontender to palpation. The patient was referred to a plastic surgeon for an excisional biopsy to clarify the nature of the lesion.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Merkel cell carcinoma
A biopsy performed 2 weeks after the initial visit confirmed the clinical suspicion for Merkel cell carcinoma (MCC).
MCC is a cutaneous neuroendocrine malignancy. Although its name acknowledges similarities between the tumor cells and Merkel cells, it is now considered unlikely that Merkel cells are the actual cells of origin.1
The majority of MCCs are asymptomatic despite rapid growth and are typically red or pink and occur on UV-exposed areas, as in our patient.2 A cyst or acneiform lesion is the single most common diagnosis given at the time of biopsy.2
The incidence of MCC is greatest in people of advanced age and in those who are immunosuppressed. In the United States, the estimated annual incidence rate rose from 0.5 cases per 100,000 people in 2000 to 0.7 cases per 100,000 people in 2013.3 MCC increases exponentially with advancing age, from 0.1 (per 100,000) in those ages 40 to 44 years to 9.8 in those older than 85 years.3 The growing cohort of ageing baby boomers and the increased number of immunosuppressed individuals in the community suggest that clinicians are now more likely to encounter MCC than in the past.
While UV radiation is highly associated with MCC, the major causative factor is considered to be Merkel cell polyomavirus (MCPyV).1 In fact, MCPyV has been linked to 80% of MCC cases.1,3 Most people have positive serology for MCPyV in early childhood, but the association between MCC and old age highlights the impact of immunosuppression on MCPyV activity and MCC development.1
Clinical suspicion is the first step in diagnosing MCC
The mnemonic AEIOU highlights the key clinical features of this aggressive tumor2,4:
- Asymptomatic
- Expanding rapidly (often grows in less than 3 months)
- Immune suppression (eg, chronic lymphocytic leukemia, solid organ transplant patient)
- Older than 50
- UV exposure on fair skin.
If a lesion is suspected to be MCC, the next step includes biopsy so that a definitive diagnosis can be made. A firm, nontender nodule that lacks fluctuance should raise suspicion for a neoplastic process.
Continue to: The differential is broad, ranging from cysts to melanoma
The differential is broad, ranging from cysts to melanoma
The differential diagnosis for an enlarging, plum-colored nodule on sun-exposed skin includes an abscess, a ruptured or inflamed epidermoid cyst, basal cell carcinoma, squamous cell carcinoma, and malignant melanoma.
An abscess is typically tender and expands within a matter of days rather than months.
A cyst can be ruled out by the clinical appearance and lack of an overlying pore.
Basal cell carcinoma can be characterized by a rolled border and central ulceration.
Squamous cell carcinomas often exhibit a verrucous surface with marked hyperkeratosis.
Continue to: Melanoma
Melanoma manifests with brown or irregular pigmentation and may be associated with a precursor lesion.
Tx includes excision and consistent follow-up
Complete excision is the critical first step to successful therapy. Sentinel lymph node studies are typically performed because of the high incidence of lymph node metastasis. Frequent follow-up is required because of the high risk of recurrent or persistent disease.
Local recurrence usually occurs within 1 year of diagnosis in more than 40% of patients.5 Distant metastasis can be treated with a programmed cell death ligand 1 blocking agent (avelumab) or a programmed cell death protein 1 inhibitor (nivolumab or pembrolizumab).6
Our patient was referred to a regional cancer center for sentinel lymph node evaluation, where he was found to have nodal disease. The patient was put on pembrolizumab and received radiation therapy but showed only limited response. Seven months after diagnosis, he passed away from metastatic MCC.
1. Pietropaolo V, Prezioso C, Moens U. Merkel cell polyomavirus and Merkel cell carcinoma. Cancers (Basel). 2020;12:1774. doi: 10.3390/cancers12071774
2. Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375-381. doi: 10.1016/j.jaad.2007.11.020
3. Paulson KG, Park SY, Vandeven NA, et al. Merkel cell carcinoma: current US incidence and projected increases based on changing demographics. J Am Acad Dermatol. 2018;78:457-463.e2. doi: 10.1016/j.jaad.2017.10.028
4. Voelker R. Why Merkel cell cancer is garnering more attention. JAMA. 2018;320:18-20. doi: 10.1001/jama.2018.7042
5. Allen PJ, Browne WB, Jacques DP, et al. Merkel cell carcinoma: prognosis and treatment of patients from a single institution. J Clin Oncol. 2005;23:2300-2309. doi: 10.1200/JCO.2005.02.329
6. D’Angelo SP, Russell J, Lebbé C, et al. Efficacy and safety of first-line avelumab treatment in patients with stage IV metastatic Merkel cell carcinoma: a preplanned interim analysis of a clinical trial. JAMA Oncol. 2018;4:e180077. doi: 10.1001/jamaoncol.2018.0077
1. Pietropaolo V, Prezioso C, Moens U. Merkel cell polyomavirus and Merkel cell carcinoma. Cancers (Basel). 2020;12:1774. doi: 10.3390/cancers12071774
2. Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375-381. doi: 10.1016/j.jaad.2007.11.020
3. Paulson KG, Park SY, Vandeven NA, et al. Merkel cell carcinoma: current US incidence and projected increases based on changing demographics. J Am Acad Dermatol. 2018;78:457-463.e2. doi: 10.1016/j.jaad.2017.10.028
4. Voelker R. Why Merkel cell cancer is garnering more attention. JAMA. 2018;320:18-20. doi: 10.1001/jama.2018.7042
5. Allen PJ, Browne WB, Jacques DP, et al. Merkel cell carcinoma: prognosis and treatment of patients from a single institution. J Clin Oncol. 2005;23:2300-2309. doi: 10.1200/JCO.2005.02.329
6. D’Angelo SP, Russell J, Lebbé C, et al. Efficacy and safety of first-line avelumab treatment in patients with stage IV metastatic Merkel cell carcinoma: a preplanned interim analysis of a clinical trial. JAMA Oncol. 2018;4:e180077. doi: 10.1001/jamaoncol.2018.0077
Can extended anticoagulation prophylaxis after discharge prevent thromboembolism?
ILLUSTRATIVE CASE
A 67-year-old man with a history of type 2 diabetes, hypertension, and chronic congestive heart failure (ejection fraction = 30%) was admitted to the intensive care unit with a diagnosis of acute hypoxic respiratory failure. He was discharged after 10 days of inpatient treatment that included daily VTE prophylaxis with low-molecular-weight heparin (LMWH). Should he go home on VTE prophylaxis?
Patients hospitalized with nonsurgical conditions such as congestive heart failure, chronic obstructive pulmonary disease, sepsis, inflammatory bowel disease, or active cancers are at increased risk for VTE due to inflammation and immobility. In a US study of 158,325 hospitalized nonsurgical patients, including those with cancer, infections, congestive heart failure, or respiratory failure, 4% of patients developed
However, use of DOACs for short-term VTE prophylaxis as an alternative to LMWH in hospitalized patients is supported by a meta-analysis showing equivalent efficacy, safety, and cost-effectiveness.1 The current study examined DOACs for extended postdischarge use.1
STUDY SUMMARY
Significant benefit of DOACs demonstrated across 4 large trials
This meta-analysis of 4 large randomized controlled trials examined the safety and efficacy of 6 weeks of postdischarge DOAC thromboprophylaxis compared with placebo in 26,408 high-risk nonsurgical hospitalized patients.1 Patients at least 40 years old were admitted with diagnoses that included New York Heart Association (NYHA) class III or IV congestive heart failure, active cancer, acute ischemic stroke, acute respiratory failure, or infectious or inflammatory disease. Study patients also had risk factors for VTE, including age 75 and older, obesity, chronic venous insufficiency, history of VTE, history of NYHA class III or IV congestive heart failure, history of cancer, thrombophilia, hormone replacement therapy, or major surgery within the 6 to 12 weeks before current medical hospitalization.
Patients were excluded if DOACs were contraindicated or if they had active or recent bleeding, renal failure, abnormal liver values, an upcoming need for surgery, or an indication for ongoing anticoagulation. Patients in 3 studies received 6 to 10 days of enoxaparin as prophylaxis during their inpatient stay. (The fourth study did not specify length of inpatient prophylaxis or drug used.) After discharge, patients were assigned to placebo or a regimen of rivaroxaban 10 mg daily, apixaban 2.5 mg twice daily, or betrixaban 80 mg daily for a range of 30 to 45 days. The primary outcome was the composite of total VTE and VTE-related death. A secondary outcome was the occurrence of nonfatal symptomatic VTE, and the primary safety outcome was the incidence of major bleeding.
The primary outcome occurred in 2.9% of the patients in the DOAC group compared with 3.6% of patients in the placebo group (odds ratio [OR] = 0.79; 95% CI, 0.69-0.91; number needed to treat [NNT] = 143). The secondary outcome occurred in 0.48% of patients in the DOAC group compared with 0.77% of patients in the placebo group (OR = 0.62; 95% CI, 0.47-0.83; NNT = 345). Major bleeding resulting in a decrease in hemoglobin concentration of more than 2 g/L, requiring transfusion of at least 2 units of packed red blood cells, reintervention at a previous surgical site, or bleeding in a critical organ or that was fatal, occurred in 0.58% of patients in the DOAC group compared with 0.3% of patients in the placebo group (OR = 1.9; 95% CI, 1.4-2.7; number needed to harm [NNH] = 357). Nonmajor bleeding was increased in the DOAC group compared with placebo (2.2% vs 1.2%; OR = 1.8; 95% CI, 1.5-2.1; NNH = 110).
The NNT to prevent a fatal VTE was 899 patients
Continue to: WHAT'S NEW
WHAT’S NEW
Mortality and morbidity benefit with small bleeding risk
Based on this study, for every 300 high-risk patients hospitalized with nonsurgical diagnoses who are given 6 weeks of DOAC prophylaxis, there will be 2 fewer cases of VTE and VTE-related death. In this same group of patients, there will be approximately 1 major bleeding event and 3 less serious bleeds.
Patients with preexisting medical conditions such as congestive heart failure, cancer, and sepsis and those admitted to an intensive care unit are at increased risk for DVT after discharge.5 Extending DOAC prophylaxis in nonsurgical patients with serious medical conditions for 6 weeks after discharge reduces the risk of VTE or VTE-related death by 0.7% compared with placebo. Treatment in this population does incur a small increased risk of major bleeding by 0.3% in the DOAC group compared with placebo.
CAVEATS
Results cannot be generalized to all patient populations
Many high-risk patients have chronic kidney disease, and because DOACs (including apixaban, rivaroxaban, and dabigatran) are renally cleared, there are limited data to establish their safety in patients with creatinine clearance ≤ 30 mL/min. Benefits seen with DOACs cannot be extrapolated to other anticoagulation agents, including warfarin or LMWH.
In accordance with new guidelines, some of the patients in this study would now receive antiplatelet therapy, eg, poststroke patients, cancer patients, and—with the ease of DOAC use—patients with atrial fibrillation. If these patients were excluded, it is not known whether the benefit would remain. Patients included in these trials were at particularly high risk for VTE, and the benefits seen in this study cannot be generalized to a patient population with fewer VTE risk factors.
CHALLENGES TO IMPLEMENTATION
High cost and lack of updated guidelines may limit DOAC thromboprophylaxis
Cost is a concern. All the new DOACs are expensive; for example, rivaroxaban costs a little less than $500 per month.6 Obtaining insurance coverage for a novel indication may be challenging. The American Society of Hematology and others have not yet endorsed extended posthospital thromboprophylaxis in nonsurgical patients, although the use of DOACs has expanded since the last guideline revisions.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Bhalla V, Lamping OF, Abdel-Latif A, et al. Contemporary meta-analysis of extended direct-acting oral anticoagulant thromboprophylaxis to prevent venous thromboembolism. Am J Med. 2020;133:1074-1081.e8. doi: 10.1016/j.amjmed.2020.01.037
2. Spyropoulos AC, Hussein M, Lin J, et al. Rates of venous thromboembolism occurrence in medical patients among the insured population. Thromb Haemost. 2009;102:951-957. doi: 10.1160/TH09-02-0073
3. Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl):e195S-226S. doi: 10.1378/chest.11-2296
4. Schünemann HJ, Cushman M, Burnett AE, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018;2:3198-3225. doi: 10.1182/bloodadvances.2018022954
5. White RH. The epidemiology of venous thromboembolism. Circulation. 2003;107(suppl):I-4-I-8. doi: 10.1161/01.CIR.0000078468.11849.66
6. Rivaroxaban . GoodRx. Accessed August 10, 2021. www.goodrx.com/rivaroxaban
ILLUSTRATIVE CASE
A 67-year-old man with a history of type 2 diabetes, hypertension, and chronic congestive heart failure (ejection fraction = 30%) was admitted to the intensive care unit with a diagnosis of acute hypoxic respiratory failure. He was discharged after 10 days of inpatient treatment that included daily VTE prophylaxis with low-molecular-weight heparin (LMWH). Should he go home on VTE prophylaxis?
Patients hospitalized with nonsurgical conditions such as congestive heart failure, chronic obstructive pulmonary disease, sepsis, inflammatory bowel disease, or active cancers are at increased risk for VTE due to inflammation and immobility. In a US study of 158,325 hospitalized nonsurgical patients, including those with cancer, infections, congestive heart failure, or respiratory failure, 4% of patients developed
However, use of DOACs for short-term VTE prophylaxis as an alternative to LMWH in hospitalized patients is supported by a meta-analysis showing equivalent efficacy, safety, and cost-effectiveness.1 The current study examined DOACs for extended postdischarge use.1
STUDY SUMMARY
Significant benefit of DOACs demonstrated across 4 large trials
This meta-analysis of 4 large randomized controlled trials examined the safety and efficacy of 6 weeks of postdischarge DOAC thromboprophylaxis compared with placebo in 26,408 high-risk nonsurgical hospitalized patients.1 Patients at least 40 years old were admitted with diagnoses that included New York Heart Association (NYHA) class III or IV congestive heart failure, active cancer, acute ischemic stroke, acute respiratory failure, or infectious or inflammatory disease. Study patients also had risk factors for VTE, including age 75 and older, obesity, chronic venous insufficiency, history of VTE, history of NYHA class III or IV congestive heart failure, history of cancer, thrombophilia, hormone replacement therapy, or major surgery within the 6 to 12 weeks before current medical hospitalization.
Patients were excluded if DOACs were contraindicated or if they had active or recent bleeding, renal failure, abnormal liver values, an upcoming need for surgery, or an indication for ongoing anticoagulation. Patients in 3 studies received 6 to 10 days of enoxaparin as prophylaxis during their inpatient stay. (The fourth study did not specify length of inpatient prophylaxis or drug used.) After discharge, patients were assigned to placebo or a regimen of rivaroxaban 10 mg daily, apixaban 2.5 mg twice daily, or betrixaban 80 mg daily for a range of 30 to 45 days. The primary outcome was the composite of total VTE and VTE-related death. A secondary outcome was the occurrence of nonfatal symptomatic VTE, and the primary safety outcome was the incidence of major bleeding.
The primary outcome occurred in 2.9% of the patients in the DOAC group compared with 3.6% of patients in the placebo group (odds ratio [OR] = 0.79; 95% CI, 0.69-0.91; number needed to treat [NNT] = 143). The secondary outcome occurred in 0.48% of patients in the DOAC group compared with 0.77% of patients in the placebo group (OR = 0.62; 95% CI, 0.47-0.83; NNT = 345). Major bleeding resulting in a decrease in hemoglobin concentration of more than 2 g/L, requiring transfusion of at least 2 units of packed red blood cells, reintervention at a previous surgical site, or bleeding in a critical organ or that was fatal, occurred in 0.58% of patients in the DOAC group compared with 0.3% of patients in the placebo group (OR = 1.9; 95% CI, 1.4-2.7; number needed to harm [NNH] = 357). Nonmajor bleeding was increased in the DOAC group compared with placebo (2.2% vs 1.2%; OR = 1.8; 95% CI, 1.5-2.1; NNH = 110).
The NNT to prevent a fatal VTE was 899 patients
Continue to: WHAT'S NEW
WHAT’S NEW
Mortality and morbidity benefit with small bleeding risk
Based on this study, for every 300 high-risk patients hospitalized with nonsurgical diagnoses who are given 6 weeks of DOAC prophylaxis, there will be 2 fewer cases of VTE and VTE-related death. In this same group of patients, there will be approximately 1 major bleeding event and 3 less serious bleeds.
Patients with preexisting medical conditions such as congestive heart failure, cancer, and sepsis and those admitted to an intensive care unit are at increased risk for DVT after discharge.5 Extending DOAC prophylaxis in nonsurgical patients with serious medical conditions for 6 weeks after discharge reduces the risk of VTE or VTE-related death by 0.7% compared with placebo. Treatment in this population does incur a small increased risk of major bleeding by 0.3% in the DOAC group compared with placebo.
CAVEATS
Results cannot be generalized to all patient populations
Many high-risk patients have chronic kidney disease, and because DOACs (including apixaban, rivaroxaban, and dabigatran) are renally cleared, there are limited data to establish their safety in patients with creatinine clearance ≤ 30 mL/min. Benefits seen with DOACs cannot be extrapolated to other anticoagulation agents, including warfarin or LMWH.
In accordance with new guidelines, some of the patients in this study would now receive antiplatelet therapy, eg, poststroke patients, cancer patients, and—with the ease of DOAC use—patients with atrial fibrillation. If these patients were excluded, it is not known whether the benefit would remain. Patients included in these trials were at particularly high risk for VTE, and the benefits seen in this study cannot be generalized to a patient population with fewer VTE risk factors.
CHALLENGES TO IMPLEMENTATION
High cost and lack of updated guidelines may limit DOAC thromboprophylaxis
Cost is a concern. All the new DOACs are expensive; for example, rivaroxaban costs a little less than $500 per month.6 Obtaining insurance coverage for a novel indication may be challenging. The American Society of Hematology and others have not yet endorsed extended posthospital thromboprophylaxis in nonsurgical patients, although the use of DOACs has expanded since the last guideline revisions.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 67-year-old man with a history of type 2 diabetes, hypertension, and chronic congestive heart failure (ejection fraction = 30%) was admitted to the intensive care unit with a diagnosis of acute hypoxic respiratory failure. He was discharged after 10 days of inpatient treatment that included daily VTE prophylaxis with low-molecular-weight heparin (LMWH). Should he go home on VTE prophylaxis?
Patients hospitalized with nonsurgical conditions such as congestive heart failure, chronic obstructive pulmonary disease, sepsis, inflammatory bowel disease, or active cancers are at increased risk for VTE due to inflammation and immobility. In a US study of 158,325 hospitalized nonsurgical patients, including those with cancer, infections, congestive heart failure, or respiratory failure, 4% of patients developed
However, use of DOACs for short-term VTE prophylaxis as an alternative to LMWH in hospitalized patients is supported by a meta-analysis showing equivalent efficacy, safety, and cost-effectiveness.1 The current study examined DOACs for extended postdischarge use.1
STUDY SUMMARY
Significant benefit of DOACs demonstrated across 4 large trials
This meta-analysis of 4 large randomized controlled trials examined the safety and efficacy of 6 weeks of postdischarge DOAC thromboprophylaxis compared with placebo in 26,408 high-risk nonsurgical hospitalized patients.1 Patients at least 40 years old were admitted with diagnoses that included New York Heart Association (NYHA) class III or IV congestive heart failure, active cancer, acute ischemic stroke, acute respiratory failure, or infectious or inflammatory disease. Study patients also had risk factors for VTE, including age 75 and older, obesity, chronic venous insufficiency, history of VTE, history of NYHA class III or IV congestive heart failure, history of cancer, thrombophilia, hormone replacement therapy, or major surgery within the 6 to 12 weeks before current medical hospitalization.
Patients were excluded if DOACs were contraindicated or if they had active or recent bleeding, renal failure, abnormal liver values, an upcoming need for surgery, or an indication for ongoing anticoagulation. Patients in 3 studies received 6 to 10 days of enoxaparin as prophylaxis during their inpatient stay. (The fourth study did not specify length of inpatient prophylaxis or drug used.) After discharge, patients were assigned to placebo or a regimen of rivaroxaban 10 mg daily, apixaban 2.5 mg twice daily, or betrixaban 80 mg daily for a range of 30 to 45 days. The primary outcome was the composite of total VTE and VTE-related death. A secondary outcome was the occurrence of nonfatal symptomatic VTE, and the primary safety outcome was the incidence of major bleeding.
The primary outcome occurred in 2.9% of the patients in the DOAC group compared with 3.6% of patients in the placebo group (odds ratio [OR] = 0.79; 95% CI, 0.69-0.91; number needed to treat [NNT] = 143). The secondary outcome occurred in 0.48% of patients in the DOAC group compared with 0.77% of patients in the placebo group (OR = 0.62; 95% CI, 0.47-0.83; NNT = 345). Major bleeding resulting in a decrease in hemoglobin concentration of more than 2 g/L, requiring transfusion of at least 2 units of packed red blood cells, reintervention at a previous surgical site, or bleeding in a critical organ or that was fatal, occurred in 0.58% of patients in the DOAC group compared with 0.3% of patients in the placebo group (OR = 1.9; 95% CI, 1.4-2.7; number needed to harm [NNH] = 357). Nonmajor bleeding was increased in the DOAC group compared with placebo (2.2% vs 1.2%; OR = 1.8; 95% CI, 1.5-2.1; NNH = 110).
The NNT to prevent a fatal VTE was 899 patients
Continue to: WHAT'S NEW
WHAT’S NEW
Mortality and morbidity benefit with small bleeding risk
Based on this study, for every 300 high-risk patients hospitalized with nonsurgical diagnoses who are given 6 weeks of DOAC prophylaxis, there will be 2 fewer cases of VTE and VTE-related death. In this same group of patients, there will be approximately 1 major bleeding event and 3 less serious bleeds.
Patients with preexisting medical conditions such as congestive heart failure, cancer, and sepsis and those admitted to an intensive care unit are at increased risk for DVT after discharge.5 Extending DOAC prophylaxis in nonsurgical patients with serious medical conditions for 6 weeks after discharge reduces the risk of VTE or VTE-related death by 0.7% compared with placebo. Treatment in this population does incur a small increased risk of major bleeding by 0.3% in the DOAC group compared with placebo.
CAVEATS
Results cannot be generalized to all patient populations
Many high-risk patients have chronic kidney disease, and because DOACs (including apixaban, rivaroxaban, and dabigatran) are renally cleared, there are limited data to establish their safety in patients with creatinine clearance ≤ 30 mL/min. Benefits seen with DOACs cannot be extrapolated to other anticoagulation agents, including warfarin or LMWH.
In accordance with new guidelines, some of the patients in this study would now receive antiplatelet therapy, eg, poststroke patients, cancer patients, and—with the ease of DOAC use—patients with atrial fibrillation. If these patients were excluded, it is not known whether the benefit would remain. Patients included in these trials were at particularly high risk for VTE, and the benefits seen in this study cannot be generalized to a patient population with fewer VTE risk factors.
CHALLENGES TO IMPLEMENTATION
High cost and lack of updated guidelines may limit DOAC thromboprophylaxis
Cost is a concern. All the new DOACs are expensive; for example, rivaroxaban costs a little less than $500 per month.6 Obtaining insurance coverage for a novel indication may be challenging. The American Society of Hematology and others have not yet endorsed extended posthospital thromboprophylaxis in nonsurgical patients, although the use of DOACs has expanded since the last guideline revisions.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Bhalla V, Lamping OF, Abdel-Latif A, et al. Contemporary meta-analysis of extended direct-acting oral anticoagulant thromboprophylaxis to prevent venous thromboembolism. Am J Med. 2020;133:1074-1081.e8. doi: 10.1016/j.amjmed.2020.01.037
2. Spyropoulos AC, Hussein M, Lin J, et al. Rates of venous thromboembolism occurrence in medical patients among the insured population. Thromb Haemost. 2009;102:951-957. doi: 10.1160/TH09-02-0073
3. Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl):e195S-226S. doi: 10.1378/chest.11-2296
4. Schünemann HJ, Cushman M, Burnett AE, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018;2:3198-3225. doi: 10.1182/bloodadvances.2018022954
5. White RH. The epidemiology of venous thromboembolism. Circulation. 2003;107(suppl):I-4-I-8. doi: 10.1161/01.CIR.0000078468.11849.66
6. Rivaroxaban . GoodRx. Accessed August 10, 2021. www.goodrx.com/rivaroxaban
1. Bhalla V, Lamping OF, Abdel-Latif A, et al. Contemporary meta-analysis of extended direct-acting oral anticoagulant thromboprophylaxis to prevent venous thromboembolism. Am J Med. 2020;133:1074-1081.e8. doi: 10.1016/j.amjmed.2020.01.037
2. Spyropoulos AC, Hussein M, Lin J, et al. Rates of venous thromboembolism occurrence in medical patients among the insured population. Thromb Haemost. 2009;102:951-957. doi: 10.1160/TH09-02-0073
3. Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl):e195S-226S. doi: 10.1378/chest.11-2296
4. Schünemann HJ, Cushman M, Burnett AE, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018;2:3198-3225. doi: 10.1182/bloodadvances.2018022954
5. White RH. The epidemiology of venous thromboembolism. Circulation. 2003;107(suppl):I-4-I-8. doi: 10.1161/01.CIR.0000078468.11849.66
6. Rivaroxaban . GoodRx. Accessed August 10, 2021. www.goodrx.com/rivaroxaban
PRACTICE CHANGER
Treat seriously ill patients with a
STRENGTH OF RECOMMENDATION
A: Meta-analysis of randomized clinical trials1
Bhalla V, Lamping OF, Abdel-Latif A, et al. Contemporary meta-analysis of extended direct-acting oral anticoagulant thromboprophylaxis to prevent venous thromboembolism. Am J Med. 2020;133:1074-1081.e8. doi: 10.1016/j.amjmed.2020.01.037
52-year-old man • syncopal episode • chest pain • mild lightheadedness • Dx?
THE CASE
A 52-year-old man with a history of hypertension and gastroesophageal reflux disease (GERD) presented to the emergency department (ED) after an episode of syncope. He reported that the syncope occurred soon after he stood up to go to the kitchen to make dinner but was without prodrome or associated symptoms. He recalled little of the event, and the episode was unwitnessed. He had a few bruises on his arms but no significant injuries.
On questioning, he reported occasional palpitations but no changes in his normal exercise tolerance. His only medication was lisinopril 10 mg/d.
In the ED, his vital signs, physical exam (including orthostatic vital signs), basic labs (including troponin I), and a 12-lead EKG were normal. After a cardiology consultation, he was discharged home with a 30-day ambulatory rhythm monitor.
A few days later, while walking up and down some hills, he experienced about 15 seconds of chest pain accompanied by mild lightheadedness. Thinking it might be related to his GERD, he took some over-the-counter antacids when he returned home, since these had been effective for him in the past.
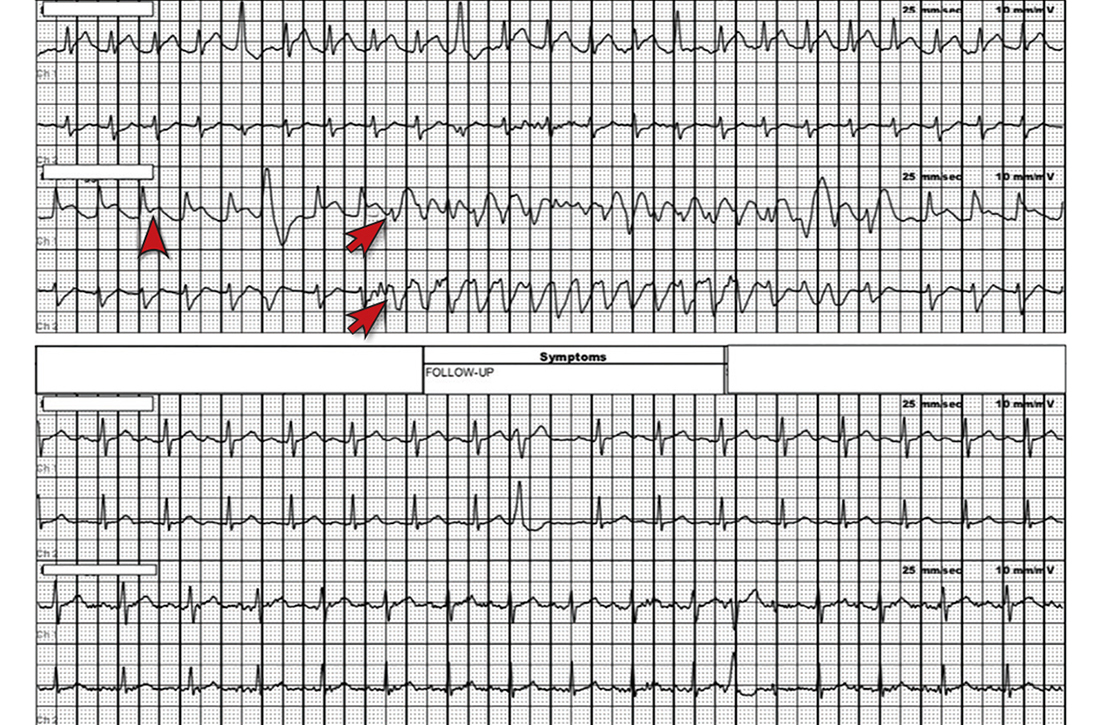
However, the rhythm monitoring company contacted the EKG lab to transmit a concerning strip (FIGURE). They also reported that the patient had been contacted and reported no further symptoms.
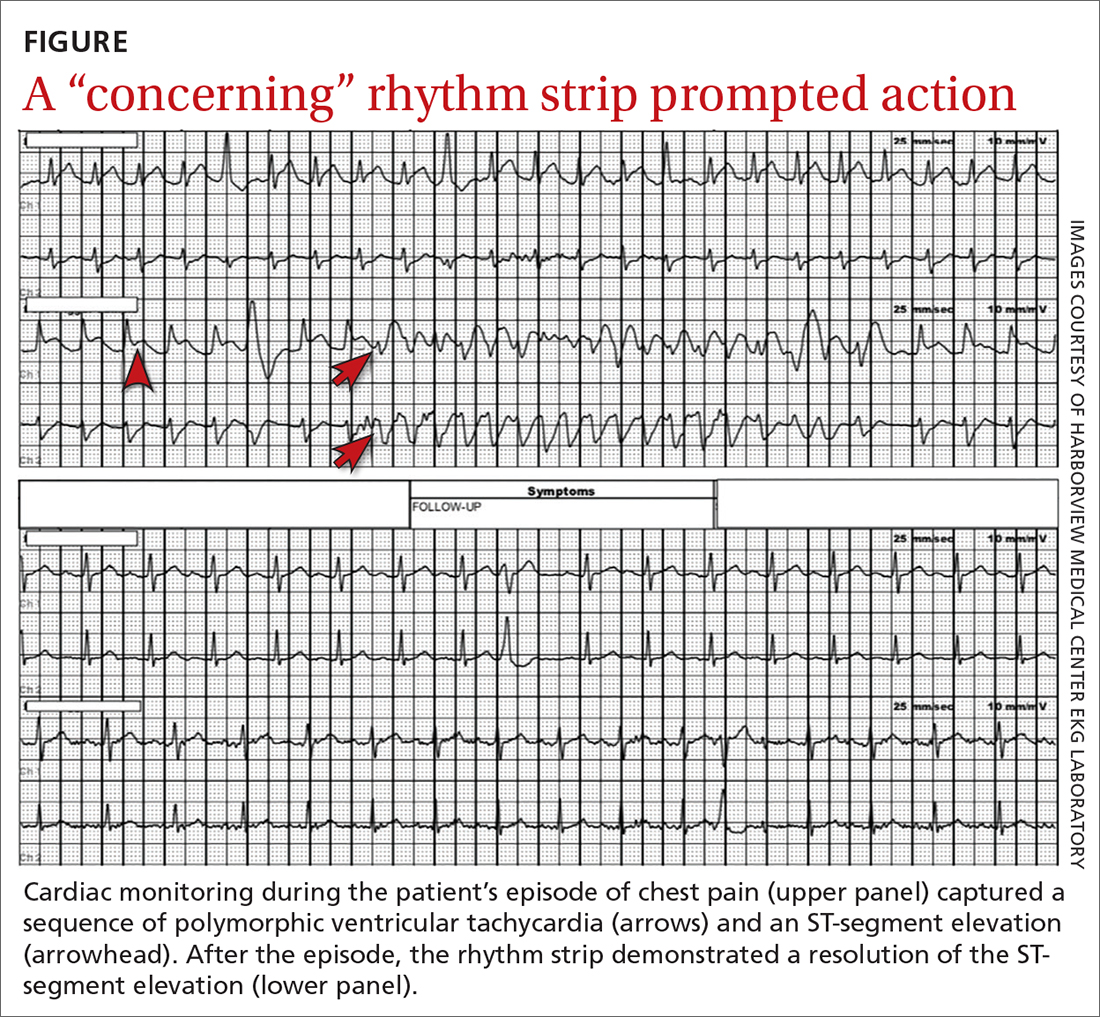
THE DIAGNOSIS
Most notable on the patient’s rhythm strip was a continuously varying QRS complex, which was indicative of polymorphic ventricular tachycardia and consistent with the patient’s syncope and other symptoms. Less obvious at first glance was an ST-segment elevation in the preceding beats. Comparison to a post-episode tracing (FIGURE) highlights the abnormality. Polymorphic ventricular tachycardia resolves in 1 of 2 ways: It will either stop on its own (causing syncope if it lasts more than a few seconds) or it will devolve into ventricular fibrillation, causing cardiac arrest.1
The combination of these findings and the clinical scenario prompted a recommendation that the patient report to the ED for admission (his wife drove him). He was admitted to the intensive care unit (ICU) for continuous telemetry monitoring, and a cardiac catheterization was ordered. The procedure revealed a 99% thrombotic mid-right coronary artery lesion, for which aspiration thrombectomy and uncomplicated stenting were performed.
Continue to: DISCUSSION
DISCUSSION
Guidelines from the American College of Cardiology/American Heart Association/Heart Rhythm Society recommend a detailed history and physical exam, as well as an EKG, for the initial evaluation of syncope.2 If this does not point to a diagnosis (and depending on the presentation and other factors), an ambulatory rhythm monitor can be considered. Other possible testing modalities include stress testing, resting transthoracic echocardiography, electrophysiologic testing, and cardiac magnetic resonance imaging or computed tomography.
Is the cause cardiac? The guidelines suggest that a cardiac cause of syncope is more likely if several of the following factors are present: age > 60 years; male sex; presence of known heart disease (acquired or congenital); brief prodrome (eg, palpitations) or no prodrome; exertional or supine syncope; 1 to 2 episodes; an abnormal cardiac exam; and a family history of premature sudden death.2 A noncardiac cause is suggested by other factors: younger age; no known cardiac disease; standing or a position change from supine to sitting/standing; prodrome; specific triggers (eg, dehydration, pain); and frequent and prolonged stereotypic episodes.2
While the guidelines do not specify the number of factors or endorse a specific scoring system, such tools have been developed. For example, the EGSYS (Evaluation of Guidelines in Syncope Study) Score assigns 1 point for each of 6 factors: palpitations; heart disease and/or abnormal EKG; effort syncope; supine syncope; precipitating or predisposing factors; and autonomic prodromes. A score ≥ 3 identified cardiac syncope with a sensitivity of 95%, but with a specificity of only 61%. In the derivation study, patients with a score ≥ 3 had higher mortality than those with a lower score (17 vs 3%; P < .001).3
Myocardial ischemia can trigger ventricular arrhythmias. In the GUSTO-1 trial of fibrinolytic therapy in patients with acute ST-segment elevation myocardial infarction (n = 40,895), the incidence of ventricular tachycardia or ventricular fibrillation was 10.2%.4 In a pooled analysis (4 trials; n = 26,416) of patients who were treated for non–ST-segment elevation or unstable angina-type acute coronary syndromes, the rate of these arrhythmias was markedly lower (2.1%).5 The risk of ventricular arrhythmia is one reason close monitoring (eg, continuous telemetry, ICU admission) is the standard of care for patients with acute coronary syndromes.
Our patient experienced syncope upon standing, which suggested a noncardiac cause (usually orthostatic hypotension). However, the history of palpitations increased the suspicion for a cardiac cause, and thus the rhythm monitor was ordered.
THE TAKEAWAY
This case was unusual in that ambulatory monitoring captured electrocardiographic evidence of myocardial ischemia leading directly to a ventricular arrhythmia. In the evaluation of syncope, a detailed history, physical exam, and a baseline 12-lead EKG can sometimes give clues to an arrhythmic cause of syncope (eg, Brugada syndrome, prior infarct pattern, prolonged QTc, bradycardia, heart block, arrhythmogenic right ventricular cardiomyopathy)—but prolonged rhythm monitoring is sometimes needed to identify a cause.
Michael A. Chen, MD, PhD, Harborview Medical Center, University of Washington School of Medicine, 325 9th Avenue, Box 359748 (Cardiology), Seattle, WA 98104; [email protected]
1. Viskin S, Chorin E, Viskin D, et al. Polymorphic ventricular tachycardia: terminology, mechanism, diagnosis, and emergency therapy. Circulation. 2021;144:823-839. doi: 10.1161/CIRCULATIONAHA.121.055783
2. Shen W-K, Sheldon RS, Benditt DG, et al. 2017 ACC/AHA/HRS guideline for the evaluation and management of patients with syncope: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2017;70:620-663. doi: 10.1016/j.jacc.2017.03.002
3. Del Rosso A, Ungar A, Maggi R, et al. Clinical predictors of cardiac syncope at initial evaluation in patients referred urgently to a general hospital: the EGSYS score. Heart. 2008;94:1528-1529. doi: 10.1136/hrt.2008.143123
4. Newby KH, Thompson T, Stebbins A, et al. Sustained ventricular arrhythmias in patients receiving thrombolytic therapy: incidence and outcomes. The GUSTO Investigators. Circulation. 1998;98:2567-2573. doi: 10.1161/01.cir.98.23.2567
5. Al-Khatib SM, Granger CB, Huang Y, et al. Sustained ventricular arrhythmias among patients with acute coronary syndromes with no ST-segment elevation: incidence, predictors, and outcomes. Circulation. 2002;106:309-12. doi: 10.1161/01.cir.0000022692.49934.e3
THE CASE
A 52-year-old man with a history of hypertension and gastroesophageal reflux disease (GERD) presented to the emergency department (ED) after an episode of syncope. He reported that the syncope occurred soon after he stood up to go to the kitchen to make dinner but was without prodrome or associated symptoms. He recalled little of the event, and the episode was unwitnessed. He had a few bruises on his arms but no significant injuries.
On questioning, he reported occasional palpitations but no changes in his normal exercise tolerance. His only medication was lisinopril 10 mg/d.
In the ED, his vital signs, physical exam (including orthostatic vital signs), basic labs (including troponin I), and a 12-lead EKG were normal. After a cardiology consultation, he was discharged home with a 30-day ambulatory rhythm monitor.
A few days later, while walking up and down some hills, he experienced about 15 seconds of chest pain accompanied by mild lightheadedness. Thinking it might be related to his GERD, he took some over-the-counter antacids when he returned home, since these had been effective for him in the past.
However, the rhythm monitoring company contacted the EKG lab to transmit a concerning strip (FIGURE). They also reported that the patient had been contacted and reported no further symptoms.

THE DIAGNOSIS
Most notable on the patient’s rhythm strip was a continuously varying QRS complex, which was indicative of polymorphic ventricular tachycardia and consistent with the patient’s syncope and other symptoms. Less obvious at first glance was an ST-segment elevation in the preceding beats. Comparison to a post-episode tracing (FIGURE) highlights the abnormality. Polymorphic ventricular tachycardia resolves in 1 of 2 ways: It will either stop on its own (causing syncope if it lasts more than a few seconds) or it will devolve into ventricular fibrillation, causing cardiac arrest.1
The combination of these findings and the clinical scenario prompted a recommendation that the patient report to the ED for admission (his wife drove him). He was admitted to the intensive care unit (ICU) for continuous telemetry monitoring, and a cardiac catheterization was ordered. The procedure revealed a 99% thrombotic mid-right coronary artery lesion, for which aspiration thrombectomy and uncomplicated stenting were performed.
Continue to: DISCUSSION
DISCUSSION
Guidelines from the American College of Cardiology/American Heart Association/Heart Rhythm Society recommend a detailed history and physical exam, as well as an EKG, for the initial evaluation of syncope.2 If this does not point to a diagnosis (and depending on the presentation and other factors), an ambulatory rhythm monitor can be considered. Other possible testing modalities include stress testing, resting transthoracic echocardiography, electrophysiologic testing, and cardiac magnetic resonance imaging or computed tomography.
Is the cause cardiac? The guidelines suggest that a cardiac cause of syncope is more likely if several of the following factors are present: age > 60 years; male sex; presence of known heart disease (acquired or congenital); brief prodrome (eg, palpitations) or no prodrome; exertional or supine syncope; 1 to 2 episodes; an abnormal cardiac exam; and a family history of premature sudden death.2 A noncardiac cause is suggested by other factors: younger age; no known cardiac disease; standing or a position change from supine to sitting/standing; prodrome; specific triggers (eg, dehydration, pain); and frequent and prolonged stereotypic episodes.2
While the guidelines do not specify the number of factors or endorse a specific scoring system, such tools have been developed. For example, the EGSYS (Evaluation of Guidelines in Syncope Study) Score assigns 1 point for each of 6 factors: palpitations; heart disease and/or abnormal EKG; effort syncope; supine syncope; precipitating or predisposing factors; and autonomic prodromes. A score ≥ 3 identified cardiac syncope with a sensitivity of 95%, but with a specificity of only 61%. In the derivation study, patients with a score ≥ 3 had higher mortality than those with a lower score (17 vs 3%; P < .001).3
Myocardial ischemia can trigger ventricular arrhythmias. In the GUSTO-1 trial of fibrinolytic therapy in patients with acute ST-segment elevation myocardial infarction (n = 40,895), the incidence of ventricular tachycardia or ventricular fibrillation was 10.2%.4 In a pooled analysis (4 trials; n = 26,416) of patients who were treated for non–ST-segment elevation or unstable angina-type acute coronary syndromes, the rate of these arrhythmias was markedly lower (2.1%).5 The risk of ventricular arrhythmia is one reason close monitoring (eg, continuous telemetry, ICU admission) is the standard of care for patients with acute coronary syndromes.
Our patient experienced syncope upon standing, which suggested a noncardiac cause (usually orthostatic hypotension). However, the history of palpitations increased the suspicion for a cardiac cause, and thus the rhythm monitor was ordered.
THE TAKEAWAY
This case was unusual in that ambulatory monitoring captured electrocardiographic evidence of myocardial ischemia leading directly to a ventricular arrhythmia. In the evaluation of syncope, a detailed history, physical exam, and a baseline 12-lead EKG can sometimes give clues to an arrhythmic cause of syncope (eg, Brugada syndrome, prior infarct pattern, prolonged QTc, bradycardia, heart block, arrhythmogenic right ventricular cardiomyopathy)—but prolonged rhythm monitoring is sometimes needed to identify a cause.
Michael A. Chen, MD, PhD, Harborview Medical Center, University of Washington School of Medicine, 325 9th Avenue, Box 359748 (Cardiology), Seattle, WA 98104; [email protected]
THE CASE
A 52-year-old man with a history of hypertension and gastroesophageal reflux disease (GERD) presented to the emergency department (ED) after an episode of syncope. He reported that the syncope occurred soon after he stood up to go to the kitchen to make dinner but was without prodrome or associated symptoms. He recalled little of the event, and the episode was unwitnessed. He had a few bruises on his arms but no significant injuries.
On questioning, he reported occasional palpitations but no changes in his normal exercise tolerance. His only medication was lisinopril 10 mg/d.
In the ED, his vital signs, physical exam (including orthostatic vital signs), basic labs (including troponin I), and a 12-lead EKG were normal. After a cardiology consultation, he was discharged home with a 30-day ambulatory rhythm monitor.
A few days later, while walking up and down some hills, he experienced about 15 seconds of chest pain accompanied by mild lightheadedness. Thinking it might be related to his GERD, he took some over-the-counter antacids when he returned home, since these had been effective for him in the past.
However, the rhythm monitoring company contacted the EKG lab to transmit a concerning strip (FIGURE). They also reported that the patient had been contacted and reported no further symptoms.

THE DIAGNOSIS
Most notable on the patient’s rhythm strip was a continuously varying QRS complex, which was indicative of polymorphic ventricular tachycardia and consistent with the patient’s syncope and other symptoms. Less obvious at first glance was an ST-segment elevation in the preceding beats. Comparison to a post-episode tracing (FIGURE) highlights the abnormality. Polymorphic ventricular tachycardia resolves in 1 of 2 ways: It will either stop on its own (causing syncope if it lasts more than a few seconds) or it will devolve into ventricular fibrillation, causing cardiac arrest.1
The combination of these findings and the clinical scenario prompted a recommendation that the patient report to the ED for admission (his wife drove him). He was admitted to the intensive care unit (ICU) for continuous telemetry monitoring, and a cardiac catheterization was ordered. The procedure revealed a 99% thrombotic mid-right coronary artery lesion, for which aspiration thrombectomy and uncomplicated stenting were performed.
Continue to: DISCUSSION
DISCUSSION
Guidelines from the American College of Cardiology/American Heart Association/Heart Rhythm Society recommend a detailed history and physical exam, as well as an EKG, for the initial evaluation of syncope.2 If this does not point to a diagnosis (and depending on the presentation and other factors), an ambulatory rhythm monitor can be considered. Other possible testing modalities include stress testing, resting transthoracic echocardiography, electrophysiologic testing, and cardiac magnetic resonance imaging or computed tomography.
Is the cause cardiac? The guidelines suggest that a cardiac cause of syncope is more likely if several of the following factors are present: age > 60 years; male sex; presence of known heart disease (acquired or congenital); brief prodrome (eg, palpitations) or no prodrome; exertional or supine syncope; 1 to 2 episodes; an abnormal cardiac exam; and a family history of premature sudden death.2 A noncardiac cause is suggested by other factors: younger age; no known cardiac disease; standing or a position change from supine to sitting/standing; prodrome; specific triggers (eg, dehydration, pain); and frequent and prolonged stereotypic episodes.2
While the guidelines do not specify the number of factors or endorse a specific scoring system, such tools have been developed. For example, the EGSYS (Evaluation of Guidelines in Syncope Study) Score assigns 1 point for each of 6 factors: palpitations; heart disease and/or abnormal EKG; effort syncope; supine syncope; precipitating or predisposing factors; and autonomic prodromes. A score ≥ 3 identified cardiac syncope with a sensitivity of 95%, but with a specificity of only 61%. In the derivation study, patients with a score ≥ 3 had higher mortality than those with a lower score (17 vs 3%; P < .001).3
Myocardial ischemia can trigger ventricular arrhythmias. In the GUSTO-1 trial of fibrinolytic therapy in patients with acute ST-segment elevation myocardial infarction (n = 40,895), the incidence of ventricular tachycardia or ventricular fibrillation was 10.2%.4 In a pooled analysis (4 trials; n = 26,416) of patients who were treated for non–ST-segment elevation or unstable angina-type acute coronary syndromes, the rate of these arrhythmias was markedly lower (2.1%).5 The risk of ventricular arrhythmia is one reason close monitoring (eg, continuous telemetry, ICU admission) is the standard of care for patients with acute coronary syndromes.
Our patient experienced syncope upon standing, which suggested a noncardiac cause (usually orthostatic hypotension). However, the history of palpitations increased the suspicion for a cardiac cause, and thus the rhythm monitor was ordered.
THE TAKEAWAY
This case was unusual in that ambulatory monitoring captured electrocardiographic evidence of myocardial ischemia leading directly to a ventricular arrhythmia. In the evaluation of syncope, a detailed history, physical exam, and a baseline 12-lead EKG can sometimes give clues to an arrhythmic cause of syncope (eg, Brugada syndrome, prior infarct pattern, prolonged QTc, bradycardia, heart block, arrhythmogenic right ventricular cardiomyopathy)—but prolonged rhythm monitoring is sometimes needed to identify a cause.
Michael A. Chen, MD, PhD, Harborview Medical Center, University of Washington School of Medicine, 325 9th Avenue, Box 359748 (Cardiology), Seattle, WA 98104; [email protected]
1. Viskin S, Chorin E, Viskin D, et al. Polymorphic ventricular tachycardia: terminology, mechanism, diagnosis, and emergency therapy. Circulation. 2021;144:823-839. doi: 10.1161/CIRCULATIONAHA.121.055783
2. Shen W-K, Sheldon RS, Benditt DG, et al. 2017 ACC/AHA/HRS guideline for the evaluation and management of patients with syncope: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2017;70:620-663. doi: 10.1016/j.jacc.2017.03.002
3. Del Rosso A, Ungar A, Maggi R, et al. Clinical predictors of cardiac syncope at initial evaluation in patients referred urgently to a general hospital: the EGSYS score. Heart. 2008;94:1528-1529. doi: 10.1136/hrt.2008.143123
4. Newby KH, Thompson T, Stebbins A, et al. Sustained ventricular arrhythmias in patients receiving thrombolytic therapy: incidence and outcomes. The GUSTO Investigators. Circulation. 1998;98:2567-2573. doi: 10.1161/01.cir.98.23.2567
5. Al-Khatib SM, Granger CB, Huang Y, et al. Sustained ventricular arrhythmias among patients with acute coronary syndromes with no ST-segment elevation: incidence, predictors, and outcomes. Circulation. 2002;106:309-12. doi: 10.1161/01.cir.0000022692.49934.e3
1. Viskin S, Chorin E, Viskin D, et al. Polymorphic ventricular tachycardia: terminology, mechanism, diagnosis, and emergency therapy. Circulation. 2021;144:823-839. doi: 10.1161/CIRCULATIONAHA.121.055783
2. Shen W-K, Sheldon RS, Benditt DG, et al. 2017 ACC/AHA/HRS guideline for the evaluation and management of patients with syncope: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2017;70:620-663. doi: 10.1016/j.jacc.2017.03.002
3. Del Rosso A, Ungar A, Maggi R, et al. Clinical predictors of cardiac syncope at initial evaluation in patients referred urgently to a general hospital: the EGSYS score. Heart. 2008;94:1528-1529. doi: 10.1136/hrt.2008.143123
4. Newby KH, Thompson T, Stebbins A, et al. Sustained ventricular arrhythmias in patients receiving thrombolytic therapy: incidence and outcomes. The GUSTO Investigators. Circulation. 1998;98:2567-2573. doi: 10.1161/01.cir.98.23.2567
5. Al-Khatib SM, Granger CB, Huang Y, et al. Sustained ventricular arrhythmias among patients with acute coronary syndromes with no ST-segment elevation: incidence, predictors, and outcomes. Circulation. 2002;106:309-12. doi: 10.1161/01.cir.0000022692.49934.e3
How to screen for and treat teen alcohol use
THE CASE
Paul F* is a 16-year-old White boy who lives with his mother and spends some weekends with his father who has shared custody. He recently presented to the clinic for treatment due to an arrest for disorderly conduct at school. He and a friend were found drinking liquor outside the school building when they were scheduled to be in class. Paul reported that he and his friends often drink at school and at extracurricular functions. He has been using alcohol for the past 2 years, with escalating consumption (5 or more drinks per episode) in the past year. Paul has been drinking most days of the week and has even driven under the influence at times. He said, “I just feel happier when I am drinking.” An accomplished soccer player recruited by colleges, Paul recently was suspended from the team due to his poor grades. His response was, “It’s stupid anyway. What’s the point of playing?”
●
* The patient’s name and some personal details have been changed to protect his identity.
Alcohol is the number 1 substance of abuse for adolescents, used more than tobacco or drugs.1-3 In 2007 and again in 2016, the Surgeon General of the United States issued reports to highlight this important topic,1,2 noting that early and repeated exposure to alcohol during this crucial time of brain development increases the risk for future problems, including addiction.2
Adolescent alcohol use is often underestimated by parents and physicians, including misjudging how much, how often, and how young children are when they begin to drink.1 Boys and girls tend to start drinking at similar ages (13.9 and 14.4 years, respectively),3 but as girls age, they tend to drink more and binge more.4 In 2019, 1 in 4 adolescents reported drinking and more than 4 million reported at least 1 episode of binge drinking in the prior month.4 These numbers have further ramifications: early drinking is associated with alcohol dependence, relapse, use of other substances, risky sexual behaviors, injurious behaviors, suicide, motor vehicle accidents, and dating violence.4-6
Diagnosing alcohol use disorder
The range of alcohol use includes consumption, bingeing, abuse, and dependence.7,8 Consumption is defined as the drinking of alcoholic beverages. Bingeing is the consumption of more than 5 drinks for men or 4 drinks for women in 2 hours, according to the National Institute on Alcohol Abuse and Alcoholism.7 However, the criterion is slightly different for the Substance Abuse and Mental Health Services Administration, which broadens the timeframe to “on the same occasion.”9 While previously known as separate disorders, alcohol abuse (or misuse) and alcohol dependence are now diagnostically classified together as alcohol use disorders (AUDs), per the Diagnostic and Statistical Manual of Mental Disorders-5 (DSM-5).8 AUD is further stratified as mild, moderate, or severe, depending on the number of criteria that are met by the patient (TABLE).8,10

Alcohol screening
Currently, the US Preventive Services Task Force (USPSTF) does not recommend screening adolescents ages 12 to 17 for AUD, and has instead issued an “I” statement (insufficient evidence).11 While the USPSTF recognizes the potential burdens of adolescent alcohol use, the potential harms of screening include “stigma, anxiety, labeling, discrimination, privacy concerns, and interference with the patient–clinician relationship.”11 The USPSTF also notes that it “did not find any evidence that specifically examined the harms of screening for alcohol use in adolescents.”11
This is at odds with recommendations from the American Academy of Pediatrics (AAP), which in 2011 released a policy statement advocating screening, brief intervention, and referral to treatment for adolescent substance use.12 In the United States, even though 83% of adolescents see a physician at least once each year,12,13 alcohol misuse screening still varies, occurring in the range of 50% to 86% of office visits.12 When screening does occur, it is often based on clinical impression only.12 Studies have shown that when a screening tool is not used, up to two-thirds of substance use disorders may be missed.12-15
Continue to: A full and complete biopsychosocial interview
A full and complete biopsychosocial interview with adolescents is a necessity, and should include queries about alcohol, drugs, and other substances. Acknowledgment of use should trigger further investigation into the substance use areas. Interviews may start with open-ended questions about alcohol use at home or at school before moving to more personalized and detailed questioning and use of screening tools.16
While various screening instruments exist, for the sake of brevity we provide as an example the Screening to Brief Intervention (S2BI) tool. It is an efficient, single-page tool that can help clinicians in their routine care of adolescents to quickly stratify the patient risk of substance use disorder as none/low, moderate, or severe.12 It can be found here: www.mcpap.com/pdf/S2Bi%20Toolkit.pdf (see page 10).
For all patients, but particularly for adolescents, confidentiality is important, and many specialty societies have created language to address this issue.12 Discuss confidentiality with both the adolescent patient and the patient’s caregiver simultaneously, with dialogue that includes: (a) the need to speak with adolescents alone during the office visit, (b) the benefits of confidentiality in the physician–patient relationship, and (c) the need to disclose selected information to keep patients safe.12 Describing the process for required disclosures is essential. Benefits of disclosure include further support for the adolescent patient as well as appropriate parental participation and support for possible referrals.12
Treating AUD
Treatment for AUD should be multifaceted. Screen for comorbid mood disorders, such as generalized anxiety,17,18 social anxiety,18 and depression,19 as well as for insomnia.18 Studies have demonstrated a strong link between insomnia and anxiety, and again between anxiety and AUD.17-19 Finally, screen for adverse childhood events such as trauma, victimization, and abuse.20 Addressing issues discovered in screening allows for more targeted and personalized treatment of AUD.
The National Institute on Drug Abuse categorizes evidence-based treatment into 3 areas: behavioral therapies, family therapies, and medications.21
Continue to: Behavioral therapies
Behavioral therapies can include group therapy, cognitive behavioral therapy (CBT), motivational enhancement therapy, 12-Step facilitation, and contingency management, in which small rewards or incentives are given for participation in treatment to reinforce positive behaviors.21
Family-based therapies, such as brief strategic family therapy, functional family therapy, and multisystem therapy recognize that adolescents exist in systems of families in communities, and that the patient’s success in treatment may be supported by these relationships.21
Some medications may achieve modest benefit for treatment of adolescents with AUD. Naltrexone, acamprosate, and disulfiram have all been used successfully to treat AUD in adults21; some physicians may choose to use these medications “off label” in adolescents. Bupropion has been used successfully in the treatment of nicotine use disorder,21 and a small study in 2005 showed some success with bupropion in treating adolescents with attention-deficit/hyperactivity disorder, comorbid depression, and substance use disorder.22 Naltrexone has also been studied in adolescents with opioid use disorder, although these were not large studies.23
Adolescents with serious, sustained issues with AUD may require more in-depth treatments such as an intensive outpatient program, a partial hospitalization program, or a residential treatment program.15 The least-restrictive environment is preferable.15 Families are generally included as part of the treatment and recovery process in those settings.21 Some patients may require detoxification prior to referral to residential treatment settings; the American Society of Addiction Medicine has published a comprehensive guideline on alcohol withdrawal.24
Paul’s family physician diagnosed his condition as AUD and referred him for CBT with a psychologist, who treated him for both the AUD and an underlying depressive disorder that was later identified. CBT focused on cognitive restructuring of depressive thoughts as well as support for continued abstinence from alcohol. The patient, with family support, declined antidepressant medication.
After 6 months of treatment, Paul and his parents were pleased with his progress. His grades improved to the point that he was permitted to play soccer again, and he was seriously looking at his future college options.
CORRESPONDENCE
Scott A. Fields, PhD, 3200 MacCorkle Avenue Southeast, 5th Floor, Robert C. Byrd Clinical Teaching Center, Department of Family Medicine, Charleston, WV 25304; [email protected]
1. US Department of Health and Human Services. The Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking. Washington, DC; US Department of Health and Human Services, Office of the Surgeon General. 2007.
2. US Department of Health and Human Services. Facing Addiction in America: The Surgeon General’s Report on Alcohol, Drugs, and Health. Washington, DC; US Department of Health and Human Services, Office of the Surgeon General. 2016.
3. Hingson R, White A. New research findings since the 2007 Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking: A review. J Stud Alcohol Drugs Suppl. 2014; 75:158-169.
4. National Institute on Alcohol Abuse and Alcoholism. Underage drinking. National Institute of Health. Accessed December 22, 2021. www.niaaa.nih.gov/publications/brochures-and-fact-sheets/underage-drinking.
5. Hingson R, Zha W, Iannotti R, et al. Physician advice to adolescents about drinking and other health behaviors. Pediatrics. 2013;131:249-257.
6. Schaus JF, Sole ML, McCoy TP, et al. Screening for high-risk drinking in a college student health center: characterizing students based on quantity, frequency, and harms. J Stud Alcohol Drugs Suppl. 2009;16:34-44.
7. National Institute on Alcohol Abuse and Alcoholism. Drinking levels defined. Accessed December 27, 2021. www.niaaa.nih.gov/alcohol-health/overview-alcohol-consumption/moderate-binge-drinking
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5). Arlington, VA; American Psychiatric Association. 2013.
9. Substance Abuse and Mental Health Services Administration. Bringing down binge drinking. Accessed December 27, 2021. www.samhsa.gov/sites/default/files/programs_campaigns/nation_prevention_week/data-binge-drinking.pdf
10. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 Alcohol Use Disorder: results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72:757-766.
11. USPSTF. Screening and behavioral counseling interventions to reduce unhealthy alcohol use in adolescents and adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;320:1899-1909.
12. Levy SJ, Williams JF, Committee on Substance Use and Prevention. Substance use screening, brief intervention, and referral to treatment. Pediatrics. 2016;138:e20161211.
13. MacKay AP, Duran CP. Adolescent Health in the United States. National Center for Health Statistics, Centers for Disease Control and Prevention. 2007.
14. Haller DM, Meynard A, Lefebvre D, et al. Effectiveness of training family physicians to deliver a brief intervention to address excessive substance use among young patients: a cluster randomized controlled trial. CMAJ. 2014;186:E263-E272.
15. Borus J, Parhami I, Levy S. Screening, brief intervention, and referral to treatment. Child Adolesc Psychiatric Clin N Am. 2016;25:579-601.
16. Knight J, Roberts T, Gabrielli J, et al. Adolescent alcohol and substance use and abuse. Performing preventive services: A bright futures handbook. Accessed December 22, 2021. American Academy of Pediatrics. https://ocfcpacourts.us/wp-content/uploads/2020/06/Adolescent_Alcohol_and_Substance_Abuse_001005.pdf
17. Dyer ML, Heron J, Hickman M, et al. Alcohol use in late adolescence and early adulthood: the role of generalized anxiety disorder and drinking to cope motives. Drug Alcohol Depend. 2019;204:107480.
18. Blumenthal H, Taylor DJ, Cloutier RM, et al. The links between social anxiety disorder, insomnia symptoms, and alcohol use disorders: findings from a large sample of adolescents in the United States. Behav Ther. 2019;50:50-59.
19. Pedrelli P, Shapero B, Archibald A, et al. Alcohol use and depression during adolescence and young adulthood: a summary and interpretation of mixed findings. Curr Addict Rep. 2016;3:91-97.
20. Davis JP, Dworkin ER, Helton J, et al. Extending poly-victimization theory: differential effects of adolescents’ experiences of victimization on substance use disorder diagnoses upon treatment entry. Child Abuse Negl. 2019; 89:165-177.
21. NIDA. Principles of adolescent substance use disorder treatment: a research-based guide. Accessed December 22, 2021. www.drugabuse.gov/publications/principles-adolescent-substance-use-disorder-treatment-research-based-guide
22. Solhkhah R, Wilens TE, Daly J, et al. Bupropion SR for the treatment of substance-abusing outpatient adolescents with attention-deficit/hyperactivity disorder and mood disorders. J Child Adolesc Psychopharmacol. 2005:15:777-786.
23. Camenga DR, Colon-Rivera HA, Muvvala SB. Medications for maintenance treatment of opioid use disorder in adolescents. J Stud Alcohol Drugs. 2019;80:393-402.
24. American Society of Addiction Medicine. The ASAM clinical practice guideline on alcohol withdrawal management. Accessed December 22, 2021. www.asam.org/quality-care/clinical-guidelines/alcohol-withdrawal-management-guideline
THE CASE
Paul F* is a 16-year-old White boy who lives with his mother and spends some weekends with his father who has shared custody. He recently presented to the clinic for treatment due to an arrest for disorderly conduct at school. He and a friend were found drinking liquor outside the school building when they were scheduled to be in class. Paul reported that he and his friends often drink at school and at extracurricular functions. He has been using alcohol for the past 2 years, with escalating consumption (5 or more drinks per episode) in the past year. Paul has been drinking most days of the week and has even driven under the influence at times. He said, “I just feel happier when I am drinking.” An accomplished soccer player recruited by colleges, Paul recently was suspended from the team due to his poor grades. His response was, “It’s stupid anyway. What’s the point of playing?”
●
* The patient’s name and some personal details have been changed to protect his identity.
Alcohol is the number 1 substance of abuse for adolescents, used more than tobacco or drugs.1-3 In 2007 and again in 2016, the Surgeon General of the United States issued reports to highlight this important topic,1,2 noting that early and repeated exposure to alcohol during this crucial time of brain development increases the risk for future problems, including addiction.2
Adolescent alcohol use is often underestimated by parents and physicians, including misjudging how much, how often, and how young children are when they begin to drink.1 Boys and girls tend to start drinking at similar ages (13.9 and 14.4 years, respectively),3 but as girls age, they tend to drink more and binge more.4 In 2019, 1 in 4 adolescents reported drinking and more than 4 million reported at least 1 episode of binge drinking in the prior month.4 These numbers have further ramifications: early drinking is associated with alcohol dependence, relapse, use of other substances, risky sexual behaviors, injurious behaviors, suicide, motor vehicle accidents, and dating violence.4-6
Diagnosing alcohol use disorder
The range of alcohol use includes consumption, bingeing, abuse, and dependence.7,8 Consumption is defined as the drinking of alcoholic beverages. Bingeing is the consumption of more than 5 drinks for men or 4 drinks for women in 2 hours, according to the National Institute on Alcohol Abuse and Alcoholism.7 However, the criterion is slightly different for the Substance Abuse and Mental Health Services Administration, which broadens the timeframe to “on the same occasion.”9 While previously known as separate disorders, alcohol abuse (or misuse) and alcohol dependence are now diagnostically classified together as alcohol use disorders (AUDs), per the Diagnostic and Statistical Manual of Mental Disorders-5 (DSM-5).8 AUD is further stratified as mild, moderate, or severe, depending on the number of criteria that are met by the patient (TABLE).8,10

Alcohol screening
Currently, the US Preventive Services Task Force (USPSTF) does not recommend screening adolescents ages 12 to 17 for AUD, and has instead issued an “I” statement (insufficient evidence).11 While the USPSTF recognizes the potential burdens of adolescent alcohol use, the potential harms of screening include “stigma, anxiety, labeling, discrimination, privacy concerns, and interference with the patient–clinician relationship.”11 The USPSTF also notes that it “did not find any evidence that specifically examined the harms of screening for alcohol use in adolescents.”11
This is at odds with recommendations from the American Academy of Pediatrics (AAP), which in 2011 released a policy statement advocating screening, brief intervention, and referral to treatment for adolescent substance use.12 In the United States, even though 83% of adolescents see a physician at least once each year,12,13 alcohol misuse screening still varies, occurring in the range of 50% to 86% of office visits.12 When screening does occur, it is often based on clinical impression only.12 Studies have shown that when a screening tool is not used, up to two-thirds of substance use disorders may be missed.12-15
Continue to: A full and complete biopsychosocial interview
A full and complete biopsychosocial interview with adolescents is a necessity, and should include queries about alcohol, drugs, and other substances. Acknowledgment of use should trigger further investigation into the substance use areas. Interviews may start with open-ended questions about alcohol use at home or at school before moving to more personalized and detailed questioning and use of screening tools.16
While various screening instruments exist, for the sake of brevity we provide as an example the Screening to Brief Intervention (S2BI) tool. It is an efficient, single-page tool that can help clinicians in their routine care of adolescents to quickly stratify the patient risk of substance use disorder as none/low, moderate, or severe.12 It can be found here: www.mcpap.com/pdf/S2Bi%20Toolkit.pdf (see page 10).
For all patients, but particularly for adolescents, confidentiality is important, and many specialty societies have created language to address this issue.12 Discuss confidentiality with both the adolescent patient and the patient’s caregiver simultaneously, with dialogue that includes: (a) the need to speak with adolescents alone during the office visit, (b) the benefits of confidentiality in the physician–patient relationship, and (c) the need to disclose selected information to keep patients safe.12 Describing the process for required disclosures is essential. Benefits of disclosure include further support for the adolescent patient as well as appropriate parental participation and support for possible referrals.12
Treating AUD
Treatment for AUD should be multifaceted. Screen for comorbid mood disorders, such as generalized anxiety,17,18 social anxiety,18 and depression,19 as well as for insomnia.18 Studies have demonstrated a strong link between insomnia and anxiety, and again between anxiety and AUD.17-19 Finally, screen for adverse childhood events such as trauma, victimization, and abuse.20 Addressing issues discovered in screening allows for more targeted and personalized treatment of AUD.
The National Institute on Drug Abuse categorizes evidence-based treatment into 3 areas: behavioral therapies, family therapies, and medications.21
Continue to: Behavioral therapies
Behavioral therapies can include group therapy, cognitive behavioral therapy (CBT), motivational enhancement therapy, 12-Step facilitation, and contingency management, in which small rewards or incentives are given for participation in treatment to reinforce positive behaviors.21
Family-based therapies, such as brief strategic family therapy, functional family therapy, and multisystem therapy recognize that adolescents exist in systems of families in communities, and that the patient’s success in treatment may be supported by these relationships.21
Some medications may achieve modest benefit for treatment of adolescents with AUD. Naltrexone, acamprosate, and disulfiram have all been used successfully to treat AUD in adults21; some physicians may choose to use these medications “off label” in adolescents. Bupropion has been used successfully in the treatment of nicotine use disorder,21 and a small study in 2005 showed some success with bupropion in treating adolescents with attention-deficit/hyperactivity disorder, comorbid depression, and substance use disorder.22 Naltrexone has also been studied in adolescents with opioid use disorder, although these were not large studies.23
Adolescents with serious, sustained issues with AUD may require more in-depth treatments such as an intensive outpatient program, a partial hospitalization program, or a residential treatment program.15 The least-restrictive environment is preferable.15 Families are generally included as part of the treatment and recovery process in those settings.21 Some patients may require detoxification prior to referral to residential treatment settings; the American Society of Addiction Medicine has published a comprehensive guideline on alcohol withdrawal.24
Paul’s family physician diagnosed his condition as AUD and referred him for CBT with a psychologist, who treated him for both the AUD and an underlying depressive disorder that was later identified. CBT focused on cognitive restructuring of depressive thoughts as well as support for continued abstinence from alcohol. The patient, with family support, declined antidepressant medication.
After 6 months of treatment, Paul and his parents were pleased with his progress. His grades improved to the point that he was permitted to play soccer again, and he was seriously looking at his future college options.
CORRESPONDENCE
Scott A. Fields, PhD, 3200 MacCorkle Avenue Southeast, 5th Floor, Robert C. Byrd Clinical Teaching Center, Department of Family Medicine, Charleston, WV 25304; [email protected]
THE CASE
Paul F* is a 16-year-old White boy who lives with his mother and spends some weekends with his father who has shared custody. He recently presented to the clinic for treatment due to an arrest for disorderly conduct at school. He and a friend were found drinking liquor outside the school building when they were scheduled to be in class. Paul reported that he and his friends often drink at school and at extracurricular functions. He has been using alcohol for the past 2 years, with escalating consumption (5 or more drinks per episode) in the past year. Paul has been drinking most days of the week and has even driven under the influence at times. He said, “I just feel happier when I am drinking.” An accomplished soccer player recruited by colleges, Paul recently was suspended from the team due to his poor grades. His response was, “It’s stupid anyway. What’s the point of playing?”
●
* The patient’s name and some personal details have been changed to protect his identity.
Alcohol is the number 1 substance of abuse for adolescents, used more than tobacco or drugs.1-3 In 2007 and again in 2016, the Surgeon General of the United States issued reports to highlight this important topic,1,2 noting that early and repeated exposure to alcohol during this crucial time of brain development increases the risk for future problems, including addiction.2
Adolescent alcohol use is often underestimated by parents and physicians, including misjudging how much, how often, and how young children are when they begin to drink.1 Boys and girls tend to start drinking at similar ages (13.9 and 14.4 years, respectively),3 but as girls age, they tend to drink more and binge more.4 In 2019, 1 in 4 adolescents reported drinking and more than 4 million reported at least 1 episode of binge drinking in the prior month.4 These numbers have further ramifications: early drinking is associated with alcohol dependence, relapse, use of other substances, risky sexual behaviors, injurious behaviors, suicide, motor vehicle accidents, and dating violence.4-6
Diagnosing alcohol use disorder
The range of alcohol use includes consumption, bingeing, abuse, and dependence.7,8 Consumption is defined as the drinking of alcoholic beverages. Bingeing is the consumption of more than 5 drinks for men or 4 drinks for women in 2 hours, according to the National Institute on Alcohol Abuse and Alcoholism.7 However, the criterion is slightly different for the Substance Abuse and Mental Health Services Administration, which broadens the timeframe to “on the same occasion.”9 While previously known as separate disorders, alcohol abuse (or misuse) and alcohol dependence are now diagnostically classified together as alcohol use disorders (AUDs), per the Diagnostic and Statistical Manual of Mental Disorders-5 (DSM-5).8 AUD is further stratified as mild, moderate, or severe, depending on the number of criteria that are met by the patient (TABLE).8,10

Alcohol screening
Currently, the US Preventive Services Task Force (USPSTF) does not recommend screening adolescents ages 12 to 17 for AUD, and has instead issued an “I” statement (insufficient evidence).11 While the USPSTF recognizes the potential burdens of adolescent alcohol use, the potential harms of screening include “stigma, anxiety, labeling, discrimination, privacy concerns, and interference with the patient–clinician relationship.”11 The USPSTF also notes that it “did not find any evidence that specifically examined the harms of screening for alcohol use in adolescents.”11
This is at odds with recommendations from the American Academy of Pediatrics (AAP), which in 2011 released a policy statement advocating screening, brief intervention, and referral to treatment for adolescent substance use.12 In the United States, even though 83% of adolescents see a physician at least once each year,12,13 alcohol misuse screening still varies, occurring in the range of 50% to 86% of office visits.12 When screening does occur, it is often based on clinical impression only.12 Studies have shown that when a screening tool is not used, up to two-thirds of substance use disorders may be missed.12-15
Continue to: A full and complete biopsychosocial interview
A full and complete biopsychosocial interview with adolescents is a necessity, and should include queries about alcohol, drugs, and other substances. Acknowledgment of use should trigger further investigation into the substance use areas. Interviews may start with open-ended questions about alcohol use at home or at school before moving to more personalized and detailed questioning and use of screening tools.16
While various screening instruments exist, for the sake of brevity we provide as an example the Screening to Brief Intervention (S2BI) tool. It is an efficient, single-page tool that can help clinicians in their routine care of adolescents to quickly stratify the patient risk of substance use disorder as none/low, moderate, or severe.12 It can be found here: www.mcpap.com/pdf/S2Bi%20Toolkit.pdf (see page 10).
For all patients, but particularly for adolescents, confidentiality is important, and many specialty societies have created language to address this issue.12 Discuss confidentiality with both the adolescent patient and the patient’s caregiver simultaneously, with dialogue that includes: (a) the need to speak with adolescents alone during the office visit, (b) the benefits of confidentiality in the physician–patient relationship, and (c) the need to disclose selected information to keep patients safe.12 Describing the process for required disclosures is essential. Benefits of disclosure include further support for the adolescent patient as well as appropriate parental participation and support for possible referrals.12
Treating AUD
Treatment for AUD should be multifaceted. Screen for comorbid mood disorders, such as generalized anxiety,17,18 social anxiety,18 and depression,19 as well as for insomnia.18 Studies have demonstrated a strong link between insomnia and anxiety, and again between anxiety and AUD.17-19 Finally, screen for adverse childhood events such as trauma, victimization, and abuse.20 Addressing issues discovered in screening allows for more targeted and personalized treatment of AUD.
The National Institute on Drug Abuse categorizes evidence-based treatment into 3 areas: behavioral therapies, family therapies, and medications.21
Continue to: Behavioral therapies
Behavioral therapies can include group therapy, cognitive behavioral therapy (CBT), motivational enhancement therapy, 12-Step facilitation, and contingency management, in which small rewards or incentives are given for participation in treatment to reinforce positive behaviors.21
Family-based therapies, such as brief strategic family therapy, functional family therapy, and multisystem therapy recognize that adolescents exist in systems of families in communities, and that the patient’s success in treatment may be supported by these relationships.21
Some medications may achieve modest benefit for treatment of adolescents with AUD. Naltrexone, acamprosate, and disulfiram have all been used successfully to treat AUD in adults21; some physicians may choose to use these medications “off label” in adolescents. Bupropion has been used successfully in the treatment of nicotine use disorder,21 and a small study in 2005 showed some success with bupropion in treating adolescents with attention-deficit/hyperactivity disorder, comorbid depression, and substance use disorder.22 Naltrexone has also been studied in adolescents with opioid use disorder, although these were not large studies.23
Adolescents with serious, sustained issues with AUD may require more in-depth treatments such as an intensive outpatient program, a partial hospitalization program, or a residential treatment program.15 The least-restrictive environment is preferable.15 Families are generally included as part of the treatment and recovery process in those settings.21 Some patients may require detoxification prior to referral to residential treatment settings; the American Society of Addiction Medicine has published a comprehensive guideline on alcohol withdrawal.24
Paul’s family physician diagnosed his condition as AUD and referred him for CBT with a psychologist, who treated him for both the AUD and an underlying depressive disorder that was later identified. CBT focused on cognitive restructuring of depressive thoughts as well as support for continued abstinence from alcohol. The patient, with family support, declined antidepressant medication.
After 6 months of treatment, Paul and his parents were pleased with his progress. His grades improved to the point that he was permitted to play soccer again, and he was seriously looking at his future college options.
CORRESPONDENCE
Scott A. Fields, PhD, 3200 MacCorkle Avenue Southeast, 5th Floor, Robert C. Byrd Clinical Teaching Center, Department of Family Medicine, Charleston, WV 25304; [email protected]
1. US Department of Health and Human Services. The Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking. Washington, DC; US Department of Health and Human Services, Office of the Surgeon General. 2007.
2. US Department of Health and Human Services. Facing Addiction in America: The Surgeon General’s Report on Alcohol, Drugs, and Health. Washington, DC; US Department of Health and Human Services, Office of the Surgeon General. 2016.
3. Hingson R, White A. New research findings since the 2007 Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking: A review. J Stud Alcohol Drugs Suppl. 2014; 75:158-169.
4. National Institute on Alcohol Abuse and Alcoholism. Underage drinking. National Institute of Health. Accessed December 22, 2021. www.niaaa.nih.gov/publications/brochures-and-fact-sheets/underage-drinking.
5. Hingson R, Zha W, Iannotti R, et al. Physician advice to adolescents about drinking and other health behaviors. Pediatrics. 2013;131:249-257.
6. Schaus JF, Sole ML, McCoy TP, et al. Screening for high-risk drinking in a college student health center: characterizing students based on quantity, frequency, and harms. J Stud Alcohol Drugs Suppl. 2009;16:34-44.
7. National Institute on Alcohol Abuse and Alcoholism. Drinking levels defined. Accessed December 27, 2021. www.niaaa.nih.gov/alcohol-health/overview-alcohol-consumption/moderate-binge-drinking
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5). Arlington, VA; American Psychiatric Association. 2013.
9. Substance Abuse and Mental Health Services Administration. Bringing down binge drinking. Accessed December 27, 2021. www.samhsa.gov/sites/default/files/programs_campaigns/nation_prevention_week/data-binge-drinking.pdf
10. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 Alcohol Use Disorder: results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72:757-766.
11. USPSTF. Screening and behavioral counseling interventions to reduce unhealthy alcohol use in adolescents and adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;320:1899-1909.
12. Levy SJ, Williams JF, Committee on Substance Use and Prevention. Substance use screening, brief intervention, and referral to treatment. Pediatrics. 2016;138:e20161211.
13. MacKay AP, Duran CP. Adolescent Health in the United States. National Center for Health Statistics, Centers for Disease Control and Prevention. 2007.
14. Haller DM, Meynard A, Lefebvre D, et al. Effectiveness of training family physicians to deliver a brief intervention to address excessive substance use among young patients: a cluster randomized controlled trial. CMAJ. 2014;186:E263-E272.
15. Borus J, Parhami I, Levy S. Screening, brief intervention, and referral to treatment. Child Adolesc Psychiatric Clin N Am. 2016;25:579-601.
16. Knight J, Roberts T, Gabrielli J, et al. Adolescent alcohol and substance use and abuse. Performing preventive services: A bright futures handbook. Accessed December 22, 2021. American Academy of Pediatrics. https://ocfcpacourts.us/wp-content/uploads/2020/06/Adolescent_Alcohol_and_Substance_Abuse_001005.pdf
17. Dyer ML, Heron J, Hickman M, et al. Alcohol use in late adolescence and early adulthood: the role of generalized anxiety disorder and drinking to cope motives. Drug Alcohol Depend. 2019;204:107480.
18. Blumenthal H, Taylor DJ, Cloutier RM, et al. The links between social anxiety disorder, insomnia symptoms, and alcohol use disorders: findings from a large sample of adolescents in the United States. Behav Ther. 2019;50:50-59.
19. Pedrelli P, Shapero B, Archibald A, et al. Alcohol use and depression during adolescence and young adulthood: a summary and interpretation of mixed findings. Curr Addict Rep. 2016;3:91-97.
20. Davis JP, Dworkin ER, Helton J, et al. Extending poly-victimization theory: differential effects of adolescents’ experiences of victimization on substance use disorder diagnoses upon treatment entry. Child Abuse Negl. 2019; 89:165-177.
21. NIDA. Principles of adolescent substance use disorder treatment: a research-based guide. Accessed December 22, 2021. www.drugabuse.gov/publications/principles-adolescent-substance-use-disorder-treatment-research-based-guide
22. Solhkhah R, Wilens TE, Daly J, et al. Bupropion SR for the treatment of substance-abusing outpatient adolescents with attention-deficit/hyperactivity disorder and mood disorders. J Child Adolesc Psychopharmacol. 2005:15:777-786.
23. Camenga DR, Colon-Rivera HA, Muvvala SB. Medications for maintenance treatment of opioid use disorder in adolescents. J Stud Alcohol Drugs. 2019;80:393-402.
24. American Society of Addiction Medicine. The ASAM clinical practice guideline on alcohol withdrawal management. Accessed December 22, 2021. www.asam.org/quality-care/clinical-guidelines/alcohol-withdrawal-management-guideline
1. US Department of Health and Human Services. The Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking. Washington, DC; US Department of Health and Human Services, Office of the Surgeon General. 2007.
2. US Department of Health and Human Services. Facing Addiction in America: The Surgeon General’s Report on Alcohol, Drugs, and Health. Washington, DC; US Department of Health and Human Services, Office of the Surgeon General. 2016.
3. Hingson R, White A. New research findings since the 2007 Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking: A review. J Stud Alcohol Drugs Suppl. 2014; 75:158-169.
4. National Institute on Alcohol Abuse and Alcoholism. Underage drinking. National Institute of Health. Accessed December 22, 2021. www.niaaa.nih.gov/publications/brochures-and-fact-sheets/underage-drinking.
5. Hingson R, Zha W, Iannotti R, et al. Physician advice to adolescents about drinking and other health behaviors. Pediatrics. 2013;131:249-257.
6. Schaus JF, Sole ML, McCoy TP, et al. Screening for high-risk drinking in a college student health center: characterizing students based on quantity, frequency, and harms. J Stud Alcohol Drugs Suppl. 2009;16:34-44.
7. National Institute on Alcohol Abuse and Alcoholism. Drinking levels defined. Accessed December 27, 2021. www.niaaa.nih.gov/alcohol-health/overview-alcohol-consumption/moderate-binge-drinking
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5). Arlington, VA; American Psychiatric Association. 2013.
9. Substance Abuse and Mental Health Services Administration. Bringing down binge drinking. Accessed December 27, 2021. www.samhsa.gov/sites/default/files/programs_campaigns/nation_prevention_week/data-binge-drinking.pdf
10. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 Alcohol Use Disorder: results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72:757-766.
11. USPSTF. Screening and behavioral counseling interventions to reduce unhealthy alcohol use in adolescents and adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;320:1899-1909.
12. Levy SJ, Williams JF, Committee on Substance Use and Prevention. Substance use screening, brief intervention, and referral to treatment. Pediatrics. 2016;138:e20161211.
13. MacKay AP, Duran CP. Adolescent Health in the United States. National Center for Health Statistics, Centers for Disease Control and Prevention. 2007.
14. Haller DM, Meynard A, Lefebvre D, et al. Effectiveness of training family physicians to deliver a brief intervention to address excessive substance use among young patients: a cluster randomized controlled trial. CMAJ. 2014;186:E263-E272.
15. Borus J, Parhami I, Levy S. Screening, brief intervention, and referral to treatment. Child Adolesc Psychiatric Clin N Am. 2016;25:579-601.
16. Knight J, Roberts T, Gabrielli J, et al. Adolescent alcohol and substance use and abuse. Performing preventive services: A bright futures handbook. Accessed December 22, 2021. American Academy of Pediatrics. https://ocfcpacourts.us/wp-content/uploads/2020/06/Adolescent_Alcohol_and_Substance_Abuse_001005.pdf
17. Dyer ML, Heron J, Hickman M, et al. Alcohol use in late adolescence and early adulthood: the role of generalized anxiety disorder and drinking to cope motives. Drug Alcohol Depend. 2019;204:107480.
18. Blumenthal H, Taylor DJ, Cloutier RM, et al. The links between social anxiety disorder, insomnia symptoms, and alcohol use disorders: findings from a large sample of adolescents in the United States. Behav Ther. 2019;50:50-59.
19. Pedrelli P, Shapero B, Archibald A, et al. Alcohol use and depression during adolescence and young adulthood: a summary and interpretation of mixed findings. Curr Addict Rep. 2016;3:91-97.
20. Davis JP, Dworkin ER, Helton J, et al. Extending poly-victimization theory: differential effects of adolescents’ experiences of victimization on substance use disorder diagnoses upon treatment entry. Child Abuse Negl. 2019; 89:165-177.
21. NIDA. Principles of adolescent substance use disorder treatment: a research-based guide. Accessed December 22, 2021. www.drugabuse.gov/publications/principles-adolescent-substance-use-disorder-treatment-research-based-guide
22. Solhkhah R, Wilens TE, Daly J, et al. Bupropion SR for the treatment of substance-abusing outpatient adolescents with attention-deficit/hyperactivity disorder and mood disorders. J Child Adolesc Psychopharmacol. 2005:15:777-786.
23. Camenga DR, Colon-Rivera HA, Muvvala SB. Medications for maintenance treatment of opioid use disorder in adolescents. J Stud Alcohol Drugs. 2019;80:393-402.
24. American Society of Addiction Medicine. The ASAM clinical practice guideline on alcohol withdrawal management. Accessed December 22, 2021. www.asam.org/quality-care/clinical-guidelines/alcohol-withdrawal-management-guideline
A practical guide to appendicitis evaluation and treatment
CASE
A 35-year-old man with a body mass index of 20 presented to the emergency department after 24 hours of abdominal pain that began in the periumbilical region and then migrated to the right lower quadrant. The pain was exacerbated during ambulation and was intense when the car transporting him to the hospital encountered bumps in the road. After his pain started, he had associated anorexia, followed by nausea and emesis. He reported fever and chills. On examination, his temperature was 100.8 °F (38.2 °C), and palpation of the right and left lower quadrants elicited right lower quadrant pain. Laboratory evaluation revealed a white blood cell (WBC) count of 14,000 cells/mcL with 85% neutrophils, C-reactive protein of 40 mg/L, and a negative urinalysis.
How would you proceed with this patient?
Acute appendicitis is the most common cause of abdominal pain resulting in the need for surgical treatment; lifetime risk of appendicitis is 6% to 7%.1 Appendicitis is caused by intraluminal obstruction in the appendix from enlarged lymphoid tissue or a fecalith. The obstruction leads to elevated intraluminal pressure due to persistent mucus and gas production by bacteria, ultimately leading to ischemia and perforation.1 Additionally, obstruction leads to bacterial overgrowth, most commonly colonic flora such as Escherichia coli, Bacteroides fragilis, Streptococcus viridans, Enterococcus sp., Pseudomonas aeruginosa, and Klebsiella pneumoniaei.1,2
The following review provides a look at how 3 clinical scoring systems compare in the identification of acute appendicitis and details which imaging studies you should order—and when. But first, we’ll quickly detail the relevant physical findings and lab values that point to a diagnosis of acute appendicitis.
Physical findings. The patient typically first experiences vague abdominal pain that then localizes to the right lower quadrant due to peritoneal inflammation. Anorexia and nausea typically follow the abdominal pain. On examination, the patient often appears ill and exhibits abdominal guarding due to peritonitis. Tachycardia and fever are common; however, the absence of either does not exclude appendicitis. Classically, on palpation, the patient will have pain at McBurney’s point (one-third the distance from the anterior iliac spine to the umbilicus). The exact point of maximal tenderness can differ because of the varying anatomy of the appendix (retrocecal, paracolic, pelvic, pre/post ileal, promontoric, or subcecal).1 Right lower quadrant pain, abdominal rigidity, and radiation of periumbilical pain to the right lower quadrant are the most accurate findings in adults to rule in appendicitis.3 For children, physical exam findings have the highest likelihood in predicting appendicitis and include a positive Obturator sign, positive Rovsing sign, or a positive Psoas sign, and absent or decreased bowel sounds.4
Laboratory studies can support a diagnosis of appendicitis but cannot exclude it. Leukocytosis with neutrophil predominance is present in 90% of cases.5 An elevated C-reactive protein level renders the highest diagnostic accuracy.5 Perform a pregnancy test for any woman of child-bearing age, to assist in the diagnosis and guide imaging choices for evaluation. Additional laboratory tests are not needed unless there are concerns about volume depletion.
Clinical scoring systems
Several clinical scoring systems (TABLE6-10) have been validated to aid clinicians in evaluating patients with possible appendicitis, to decrease unnecessary exposure to ionizing radiation from computed tomography (CT) scans, to identify and reassure patients with low likelihoods of appendicitis, and to conduct outpatient follow-up.
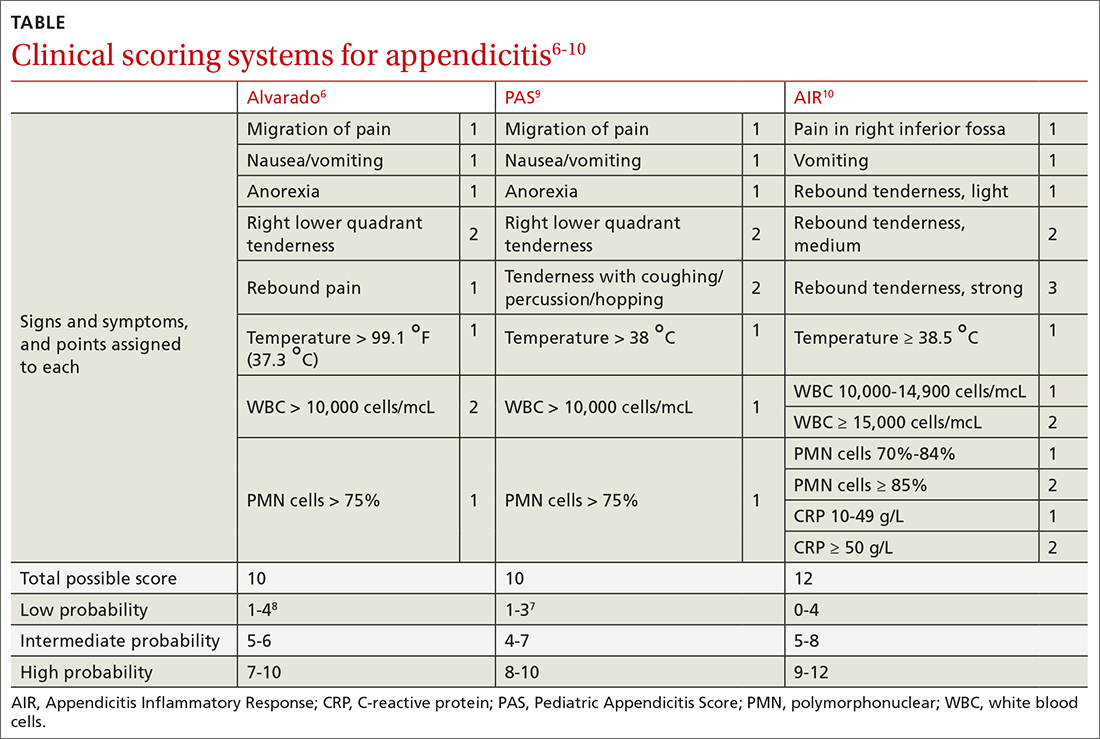
Continue to: The Alvarado score
The Alvarado score is the oldest scoring rule, developed in 1986; it entails 8 clinical and laboratory variables.6 Ebell et al altered the proposed cutoff values of the Alvarado score to be low risk (< 4), intermediate risk (4-8), and high risk (≥ 9), effectively improving the sensitivity and specificity rates.7
In a meta-analysis of the Alvarado score that included 42 studies of men, women, and children, the sensitivity for “ruling out” appendicitis with a cutoff of 5 points was 96% for men, 99% for women, and 99% for children.8 The accuracy of a high-risk score (> 7) for “ruling in” appendicitis was less with an overall specificity of 82%.8 The Alvarado score did seem to overestimate appendicitis in women in all score categories.8
The Pediatric Appendicitis Score (PAS) is similar to Alvarado and was prospectively validated in 1170 children in 2002 for more specific guidance in this age group.9 The PAS had excellent specificity in the study; those with a score of ≥ 6 had a high probability of appendicitis. In a study comparing Alvarado with PAS in 311 patients, insignificant differences were noted at a score of ≥ 7 for both tests (sensitivity 86% vs 89%, and specificity 59% vs 50%, respectively).11 No scoring system has been found to be sufficiently accurate for use in children 4 years of age and younger.12
The Appendicitis Inflammatory Response (AIR) Score was prospectively validated in 545 patients representing all age groups.10 Subsequently, in a larger prospective multicenter study of 3878 patients older than 5 years, the original cut points were altered, thereby improving test sensitivity and negative predictive value to 99% for those with low probability (0 to 3), and test specificity to 98% for those with high-probability (9 to 12).13 Compared with the Alvarado Score, the AIR Score has higher specificity for those in the high-probability range, and similar exclusion rates in the low-probability range.14
Caveats with clinical decision scores. These tools are accepted and often used. However, challenges that affect generalizability of study data include differences in patient selection for each study (undifferentiated abdominal pain vs appendicitis), prospective vs retrospective designs, and age and gender variations in the patient populations. Despite the numerous scoring systems developed, none can accurately be used to rule in appendicitis. They are best used to assist in ruling out appendicitis and to aid in deciding for or against imaging.
Continue to: A look at the imaging options
A look at the imaging options
Abdominal CT has sensitivity and specificity rates between 76% and 100% and 83% and 100%, respectively.15,20,21 Ultrasonography has sensitivity and specificity rates of 71% to 94% and 81% to 98%, respectively.15,20,21 Formal US is reliable to confirm appendicitis, but less so to rule out appendicitis. Special considerations for imagining in pregnant patients and children are discussed in a bit.
Timing of surgical consultation
Surgical consultation is paramount once the diagnosis of appendicitis is probable. Imaging is best obtained prior to surgical consultation to streamline evaluation and enhance decision- making. Typically, patients will be categorized as complicated or uncomplicated based on the presence or absence of perforation, a gangrenous appendix, an intra-abdominal abscess (IAA), or purulent peritonitis. Active continuous surgical involvement (co-management or assumption of care) is recommended in all cases of appendicitis, especially if nonoperative management is selected, given that some cases must convert to immediate operative treatment or may be selected for delayed future (interval) appendectomy.22
Management
Uncomplicated appendicitis
Prompt appendectomy has been the gold standard of care for uncomplicated acute appendicitis for 60 years. However, several studies have investigated an antibiotic-based strategy rather than surgical treatment for uncomplicated appendicitis.
Antibiotics vs appendectomy. In 2020, the CODA Collaborative published a randomized trial comparing a 10-day course of antibiotics with appendectomy in patients with uncomplicated appendicitis. In this multicenter study based in the United States, 1552 patients 18 years of age or older were randomized to receive antibiotics or undergo appendectomy (95% performed laparoscopically). The antibiotic treatment consisted of at least 24 hours of IV antibiotics, with or without admission to the hospital. Antibiotic choice was individualized according to guidelines for intra-abdominal infection published by the Infectious Diseases Society of America, with the most common IV medications being ertapenem, cefoxitin, or metronidazole plus one of the following: ceftriaxone, cefazolin, or levofloxacin. For the remaining 10 days, oral metronidazole plus ciprofloxacin or cefdinir were used.22
Continue to: The primary endpoint...
The primary endpoint was the European Quality of Life-5 Dimensions (EQ-5D) questionnaire, with secondary outcomes including appendectomy in the antibiotics group and complications through 90 days. Exclusion criteria included pregnancy, sepsis, peritonitis, recurrent appendicitis, severe phlegmon on imaging, or evidence of neoplasm.22
Antibiotics were noninferior to appendectomy for the 30-day study. However, antibiotics failed in 29%, who then proceeded to appendectomy by 90 days; these patients also accounted for 41% of those with an appendicolith. Overall complications were more common in the antibiotics group than in the appendectomy group (8.1 vs 3.5 per 100 participants; 95% CI, 1.3-3.98). Also more common in the antibiotic group were serious adverse events (4 vs 3 per 100 participants; hazard ratio [HR] = 1.29; 95% CI, 0.67-2.50). The presence of an appendicolith in the antibiotics group increased the conversion risk to appendectomy, as well as adverse events risk.22
The takeaway. Antibiotic treatment is a noninferior method to treat acute uncomplicated appendicitis. However, the informed consent process is important, given the ~30% failure rate. Patient factors such as continued access to care should help inform the decision.
Two main surgical approaches exist for appendectomy: open and minimally invasive. At this time, the minimally invasive options include laparoscopic, single incision laparoscopic surgery (SILS), and robotic appendectomy. A study comparing cost, availability, or complications of these options has not been conducted at this time.
A large Cochrane review of 67 studies examining open vs laparoscopic appendectomy in adults and children completed in 2018 revealed that the laparoscopic approach reduced early postoperative pain intensity and led to a shorter hospital stay, earlier return to work or usual activities, and a decrease in wound infections.23 The odds of IAA occurring with laparoscopic appendectomy increased by 65% compared with an open procedure; however, postoperative bowel obstruction and incisional hernias were less likely to occur.23 Additionally, following laparoscopic surgery, postoperative bowel obstruction and incisional hernias are less likely to occur. The laparoscopic approach is preferred due to overall increased patient satisfaction and a reduction in most, if not all, complications.
Continue to: Complicated appendicitis
Complicated appendicitis
Excluding patients with severe sepsis or purulent peritonitis requiring resuscitation and immediate surgical intervention of intra-abdominal infection, the approach to patients with complicated appendicitis varies between aggressive surgical intervention and nonoperative management.
In a 2007 meta-analysis reviewing nonsurgical treatment of appendiceal abscess/phlegmon, immediate surgery was associated with higher morbidity.24 Within the nonoperative management group 7.2% (CI, 4.0-10.5) required surgical intervention and 19.7% (CI, 11.0-28.3) required abscess drainage. Malignant disease was detected in 1.2% (CI, 0.6-1.7).24 Small subsequent studies concluded different results.25
Ultimately, the 2015 European Association of Endoscopic Surgery guidelines recommend a new systematic review; but with current data, initial nonoperative management is preferred.15 After initial nonoperative treatment, the only benefits from interval appendectomy are identification of an underlying malignancy (6% to 20%) and mitigating the risk of recurrent appendicitis (5% to 44%).15,25-30
Multiple single institutional series found increased neoplasm incidence (9% to 20%) in complicated appendicitis in patients 40 years and older.26-30 Prior to interval appendectomy in patients 45 years and older, ensuring they have an up-to-date screening colonoscopy is important. This is in line with 2021 US Preventive Services Task Force (Grade “B” recommendation), 2018 American Cancer Society (qualified recommendation), and 2021 American College of Gastroenterology (conditional recommendation) guidelines for colorectal cancer screening to start at age 45 in average-risk patients.31 Patients younger than 45 can consider screening through shared decision-making.
Special populations
Pregnant patients
In pregnancy, challenges exist with the presence of traditional signs and symptoms of appendicitis, with the most predictive sign being a WBC count higher than 18,000.32 The American College of Radiology’s (ACR) Appropriateness Criteria recommend US as the imaging modality of choice in pregnancy, with MRI as the best option when US is inconclusive.33 Two meta-analyses demonstrated high sensitivity (91.8%-96.6%) and specificity (95.9%-97.9%) of MRI in diagnosing appendicitis.34,35 CT scan is not the preferred initial imagining modality in pregnancy unless urgent information is needed and other modalities are insufficient or unavailable.36
Continue to: The most common...
The most common nonobstetric surgical intervention during pregnancy is appendectomy, at a rate of 6.3/10,000 person-years, which increases to 9.9/10,000 in the postpartum period.37 Two large population studies demonstrate the rate of appendicitis varies over the course of pregnancy, with the lowest rates in the third trimester,38,39 and a significant rebound lasting for 2 years postpartum.39 Peritonitis, septic shock, pneumonia, postoperative infection, and longer hospital stays occur more frequently in pregnant women than in nonpregnant women with appendicitis.40 Fetal loss is higher in the first trimester.32
In a 14-year review of 63,145 appendicitis cases, an increased risk of fetal loss and maternal death was noted across ages and ethnicities, with the largest risk of maternal death occurring in Hispanics and fetal death in non-Hispanic Blacks.41 In a large study of 1018 adverse events after appendectomy or cholecystectomy, the 3 most common events were preterm delivery (35.4%), preterm labor without preterm delivery (26.4%), and miscarriage (25.7%).42 The surgery itself was not a major risk factor for adverse events. Major risk factors included cervical incompetence (odds ratio [OR] = 24.3), preterm labor in current pregnancy (OR = 18.3), and presence of vulvovaginitis (OR = 5.2).42
Nonoperative management in pregnancy is not recommended; only 1 prospective trial has been done, with 20 patients, showing a 25% failure rate.43 Two meta-analyses published in 2019 highlight the potential increase of fetal loss with laparoscopic approaches to appendectomy.44,45 However, recently published literature demonstrates no significant maternal-fetal morbidity. Current guidelines of the Society of American Gastrointestinal and Endoscopic Surgeons agree that laparoscopy is the operative choice in pregnancy.36
Children
Acute appendicitis is the most common surgical emergency in children.4 Physical exam findings and laboratory results are not classic in this population, obtaining an accurate history can be challenging, and results of clinical scoring systems can be inconclusive.4 Additional serum biomarkers, procalcitonin and calprotectin, are gaining evidence for use in improving scoring systems to refine low-risk groups. Unavailability of timely, reliable biomarker testing in rural practice locations limits definitive recommendations at this time.46 ACR recommends no imaging in a pediatric patient whose risk of having appendicitis is low based on any of several scoring systems.47 For those assessed as having higher risk, US is the recommended initial modality,with CT with IV contrast or MRI without contrast equally recommended if the US is equivocal.47
Despite promising data from trials of nonoperative treatment for adults with appendicitis, no definitive evidence and recommendations are available for children. Two systematic reviews show nonoperative treatment is safe, with an efficacy rate of 76% to 82% at long-term follow-up,48,49 although the success of antibiotic regimens varies. Within the nonoperative treatment group, 16% of patients had appendectomy during the follow-up period, which varied from 8 weeks to 4 years.48 A randomized controlled trial is needed for final guidance.
Continue to: CASE
CASE
The patient had an Alvarado score of 9 (high probability) and an AIR score of 6 (intermediate probability). A CT with IV contrast showed a 9-mm fluid-filled appendix with periappendiceal fluid. During surgical consultation, he was offered laparoscopic appendectomy or nonoperative treatment with antibiotics. He opted for a preoperative dose of piperacillin-tazobactam 3.375 g IV and laparoscopic appendectomy. The patient was discharged home 6 hours after his procedure.
CORRESPONDENCE
Jessica Servey, MD, MHPE, 4301 Jones Bridge Road, Bethesda, MD 20814; [email protected]
1. Prystowsky JB, Pugh CM, Nagle AP. Current problems in surgery. Appendicitis. Curr Probl Surg. 2005;42:688-742.
2. Song DW, Park BK, Suh SW, et al. Bacterial culture and antibiotic susceptibility in patients with acute appendicitis. Int J Colorectal Dis. 2018;33:441-447.
3. Wagner JM, McKinney WP, Carpenter JL. Does this patient have appendicitis? JAMA. 1996;276:1589-1594.
4. Benabbas R, Hanna M, Shah J, et al. Diagnostic accuracy of history, physical examination, laboratory tests, and point-of-care ultrasound for pediatric acute appendicitis in the emergency department: a systematic review and meta-analysis. Acad Emerg Med. 2017;24:523-551.
5. Andersson RE. Meta-analysis of the clinical and laboratory diagnosis of appendicitis. Br J Surg. 2004;91:28-37.
6. Alvarado A. A practical score for the early diagnosis of acute appendicitis. Ann Emerg Med. 1986;15:557-564.
7. Ebell MH, Shinholser J. What are the most clinically useful cutoffs for the Alvarado and Pediatric Appendicitis Scores? A systematic review. Ann Emerg Med. 2014;64:365-372.e2.
8. Ohle R, O’Reilly F, O’Brien KK, et al. The Alvarado score for predicting acute appendicitis: a systematic review. BMC Med. 2011;9:139.
9. Samuel M. Pediatric appendicitis score. J Pediatr Surg. 2002;37:877-881.
10. Andersson M, Andersson RE. The appendicitis inflammatory response score: a tool for the diagnosis of acute appendicitis that outperforms the Alvarado score. World J Surg. 2008;32:1843-1849.
11. Pogorelić Z, Rak S, Mrklić I, et al. Prospective validation of Alvarado score and Pediatric Appendicitis Score for the diagnosis of acute appendicitis in children. Pediatr Emerg Care. 2015;31:164-168.
12. Rassi R, Muse F, Sánchez-Martínez J, et al. Diagnostic value of clinical prediction scores for acute appendicitis in children younger than 4 years. Eur J Pediatr Surg. 2021. [Online ahead of print]
13. Andersson M, Kolodziej B, Andersson RE. Validation of the Appendicitis Inflammatory Response (AIR) score. World J Surg. 2021;45:2081-2091.
14. Kollár D, McCartan DP, Bourke M, et al. Predicting acute appendicitis? A comparison of the Alvarado score, the Appendicitis Inflammatory Response Score and clinical assessment. World J Surg. 2015;39:104-109.
15. Gorter RR, Eker HH, Gorter-Stam MA, et al. Diagnosis and management of acute appendicitis. EAES consensus development conference 2015. Surg Endosc. 2016;30:4668-4690.
16. Matthew Fields J, Davis J, Alsup C, et al. Accuracy of point-of-care ultrasonography for diagnosing acute appendicitis: a systematic review and meta-analysis. Acad Emerg Med. 2017;24:1124-1136.
17. Sharif S, Skitch S, Vlahaki D, et al. Point-of-care ultrasound to diagnose appendicitis in a Canadian emergency department. CJEM. 2018;20:732-735.
18. Doniger SJ, Kornblith A. Point-of-care ultrasound integrated into a staged diagnostic algorithm for pediatric appendicitis. Pediatr Emerg Care. 2018;34:109-115.
19. Menon N, Kumar S, Keeler B, et al. A systematic review of point-of-care abdominal ultrasound scans performed by general surgeons. Surgeon. 2021. [Online ahead of print]
20. Doria AS, Moineddin R, Kellenberger CJ, et al. US or CT for diagnosis of appendicitis in children and adults? A meta-analysis. Radiology. 2006;241:83-94.
21. van Randen A, Laméris W, van Es HW, et al. A comparison of the accuracy of ultrasound and computed tomography in common diagnoses causing acute abdominal pain. Eur Radiol. 2011;21:1535-1545.
22. Flum DR, Davidson GH, Monsell SE, et al. A randomized trial comparing antibiotics with appendectomy for appendicitis. N Engl J Med. 2020;383:1907-1919.
23. Jaschinski T, Mosch CG, Eikermann M, et al. Laparoscopic versus open surgery for suspected appendicitis. Cochrane Database Syst Rev. 2018;11:CD001546.
24. Andersson RE, Petzold MG. Nonsurgical treatment of appendiceal abscess or phlegmon: a systematic review and meta-analysis. Ann Surg. 2007;246:741-748.
25. Deelder JD, Richir MC, Schoorl T, et al. How to treat an appendiceal inflammatory mass: operatively or nonoperatively? J Gastrointest Surg. 2014;18:641-645.
26. Carpenter SG, Chapital AB, Merritt MV, et al. Increased risk of neoplasm in appendicitis treated with interval appendectomy: single-institution experience and literature review. Am Surg. 2012;78:339-343.
27. Hayes D, Reiter S, Hagen E, et al. Is interval appendectomy really needed? A closer look at neoplasm rates in adult patients undergoing interval appendectomy after complicated appendicitis. Surg Endosc. 2021;35:3855-3860.
28. Peltrini R, Cantoni V, Green R, et al. Risk of appendiceal neoplasm after interval appendectomy for complicated appendicitis: a systematic review and meta-analysis. Surgeon. 2021. [Online ahead of print.]
29. Mällinen J, Rautio T, Grönroos J, et al. Risk of appendiceal neoplasm in periappendicular abscess in patients treated with interval appendectomy vs follow-up with magnetic resonance imaging: 1-year outcomes of the peri-appendicitis acuta randomized clinical trial. JAMA Surg. 2019;154:200-207.
30. Son J, Park YJ, Lee SR, et al. Increased risk of neoplasms in adult patients undergoing interval appendectomy. Ann Coloproctol. 2020;36:311-315.
31. Davidson KW, Barry MJ, Mangione CM, et al. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325:1965-1977.
32. Theilen LH, Mellnick VM, Shanks AL, et al. Acute appendicitis in pregnancy: predictive clinical factors and pregnancy outcomes. Am J Perinatol. 2017;34:523-528.
33. Garcia EM, Camacho MA, Karolyi DR, et al. ACR Appropriateness Criteria right lower quadrant pain-suspected appendicitis. J Am Coll Radiol. 2018;15:S373-s387.
34. Kave M, Parooie F, Salarzaei M. Pregnancy and appendicitis: a systematic review and meta-analysis on the clinical use of MRI in diagnosis of appendicitis in pregnant women. World J Emerg Surg. 2019;14:37.
35. Repplinger MD, Levy JF, Peethumnongsin E, et al. Systematic review and meta-analysis of the accuracy of MRI to diagnose appendicitis in the general population. J Magn Reson Imaging. 2016;43:1346-1354.
36. Pearl JP, Price RR, Tonkin AE, et al. SAGES guidelines for the use of laparoscopy during pregnancy. Surg Endosc. 2017;31:3767-3782.
37. Zingone F, Sultan AA, Humes DJ, et al. Risk of acute appendicitis in and around pregnancy: a population-based cohort study from England. Ann Surg. 2015;261:332-337.
38. Andersson RE, Lambe M. Incidence of appendicitis during pregnancy. Int J Epidemiol. 2001;30:1281-1285.
39. Moltubak E, Landerholm K, Blomberg M, et al. Major variation in the incidence of appendicitis before, during and after pregnancy: a population-based cohort study. World J Surg. 2020;44:2601-2608.
40. Abbasi N, Patenaude V, Abenhaim HA. Management and outcomes of acute appendicitis in pregnancy-population-based study of over 7000 cases. BJOG. 2014;121:1509-1514.
41. Dongarwar D, Taylor J, Ajewole V, et al. Trends in appendicitis among pregnant women, the risk for cardiac arrest, and maternal-fetal mortality. World J Surg. 2020;44:3999-4005.
42. Sachs A, Guglielminotti J, Miller R, et al. Risk factors and risk stratification for adverse obstetrical outcomes after appendectomy or cholecystectomy during pregnancy. JAMA Surg. 2017;152:436-441.
43. Joo JI, Park HC, Kim MJ, et al. Outcomes of antibiotic therapy for uncomplicated appendicitis in pregnancy. Am J Med. 2017;130:1467-1469.
44. Lee SH, Lee JY, Choi YY, Lee JG. Laparoscopic appendectomy versus open appendectomy for suspected appendicitis during pregnancy: a systematic review and updated meta-analysis. BMC Surg. 2019;19:41.
45. Frountzas M, Nikolaou C, Stergios K, et al. Is the laparoscopic approach a safe choice for the management of acute appendicitis in pregnant women? A meta-analysis of observational studies. Ann R Coll Surg Engl. 2019;101:235-248.
46. Di Saverio S, Podda M, De Simone B, et al. Diagnosis and treatment of acute appendicitis: 2020 update of the WSES Jerusalem guidelines. World J Emerg Surg. 2020;15:27.
47. Koberlein GC, Trout AT, Rigsby CK, et al. ACR Appropriateness Criteria suspected appendicitis-child. J Am Coll Radiol. 2019;16:S252-S263.
48. Maita S, Andersson B, Svensson JF, et al. Nonoperative treatment for nonperforated appendicitis in children: a systematic review and meta-analysis. Pediatr Surg Int. 2020;36:261-269.
49. Georgiou R, Eaton S, Stanton MP, et al. Efficacy and safety of nonoperative treatment for acute appendicitis: a meta-analysis. Pediatrics. 2017;139:e20163003.
CASE
A 35-year-old man with a body mass index of 20 presented to the emergency department after 24 hours of abdominal pain that began in the periumbilical region and then migrated to the right lower quadrant. The pain was exacerbated during ambulation and was intense when the car transporting him to the hospital encountered bumps in the road. After his pain started, he had associated anorexia, followed by nausea and emesis. He reported fever and chills. On examination, his temperature was 100.8 °F (38.2 °C), and palpation of the right and left lower quadrants elicited right lower quadrant pain. Laboratory evaluation revealed a white blood cell (WBC) count of 14,000 cells/mcL with 85% neutrophils, C-reactive protein of 40 mg/L, and a negative urinalysis.
How would you proceed with this patient?
Acute appendicitis is the most common cause of abdominal pain resulting in the need for surgical treatment; lifetime risk of appendicitis is 6% to 7%.1 Appendicitis is caused by intraluminal obstruction in the appendix from enlarged lymphoid tissue or a fecalith. The obstruction leads to elevated intraluminal pressure due to persistent mucus and gas production by bacteria, ultimately leading to ischemia and perforation.1 Additionally, obstruction leads to bacterial overgrowth, most commonly colonic flora such as Escherichia coli, Bacteroides fragilis, Streptococcus viridans, Enterococcus sp., Pseudomonas aeruginosa, and Klebsiella pneumoniaei.1,2
The following review provides a look at how 3 clinical scoring systems compare in the identification of acute appendicitis and details which imaging studies you should order—and when. But first, we’ll quickly detail the relevant physical findings and lab values that point to a diagnosis of acute appendicitis.
Physical findings. The patient typically first experiences vague abdominal pain that then localizes to the right lower quadrant due to peritoneal inflammation. Anorexia and nausea typically follow the abdominal pain. On examination, the patient often appears ill and exhibits abdominal guarding due to peritonitis. Tachycardia and fever are common; however, the absence of either does not exclude appendicitis. Classically, on palpation, the patient will have pain at McBurney’s point (one-third the distance from the anterior iliac spine to the umbilicus). The exact point of maximal tenderness can differ because of the varying anatomy of the appendix (retrocecal, paracolic, pelvic, pre/post ileal, promontoric, or subcecal).1 Right lower quadrant pain, abdominal rigidity, and radiation of periumbilical pain to the right lower quadrant are the most accurate findings in adults to rule in appendicitis.3 For children, physical exam findings have the highest likelihood in predicting appendicitis and include a positive Obturator sign, positive Rovsing sign, or a positive Psoas sign, and absent or decreased bowel sounds.4
Laboratory studies can support a diagnosis of appendicitis but cannot exclude it. Leukocytosis with neutrophil predominance is present in 90% of cases.5 An elevated C-reactive protein level renders the highest diagnostic accuracy.5 Perform a pregnancy test for any woman of child-bearing age, to assist in the diagnosis and guide imaging choices for evaluation. Additional laboratory tests are not needed unless there are concerns about volume depletion.
Clinical scoring systems
Several clinical scoring systems (TABLE6-10) have been validated to aid clinicians in evaluating patients with possible appendicitis, to decrease unnecessary exposure to ionizing radiation from computed tomography (CT) scans, to identify and reassure patients with low likelihoods of appendicitis, and to conduct outpatient follow-up.

Continue to: The Alvarado score
The Alvarado score is the oldest scoring rule, developed in 1986; it entails 8 clinical and laboratory variables.6 Ebell et al altered the proposed cutoff values of the Alvarado score to be low risk (< 4), intermediate risk (4-8), and high risk (≥ 9), effectively improving the sensitivity and specificity rates.7
In a meta-analysis of the Alvarado score that included 42 studies of men, women, and children, the sensitivity for “ruling out” appendicitis with a cutoff of 5 points was 96% for men, 99% for women, and 99% for children.8 The accuracy of a high-risk score (> 7) for “ruling in” appendicitis was less with an overall specificity of 82%.8 The Alvarado score did seem to overestimate appendicitis in women in all score categories.8
The Pediatric Appendicitis Score (PAS) is similar to Alvarado and was prospectively validated in 1170 children in 2002 for more specific guidance in this age group.9 The PAS had excellent specificity in the study; those with a score of ≥ 6 had a high probability of appendicitis. In a study comparing Alvarado with PAS in 311 patients, insignificant differences were noted at a score of ≥ 7 for both tests (sensitivity 86% vs 89%, and specificity 59% vs 50%, respectively).11 No scoring system has been found to be sufficiently accurate for use in children 4 years of age and younger.12
The Appendicitis Inflammatory Response (AIR) Score was prospectively validated in 545 patients representing all age groups.10 Subsequently, in a larger prospective multicenter study of 3878 patients older than 5 years, the original cut points were altered, thereby improving test sensitivity and negative predictive value to 99% for those with low probability (0 to 3), and test specificity to 98% for those with high-probability (9 to 12).13 Compared with the Alvarado Score, the AIR Score has higher specificity for those in the high-probability range, and similar exclusion rates in the low-probability range.14
Caveats with clinical decision scores. These tools are accepted and often used. However, challenges that affect generalizability of study data include differences in patient selection for each study (undifferentiated abdominal pain vs appendicitis), prospective vs retrospective designs, and age and gender variations in the patient populations. Despite the numerous scoring systems developed, none can accurately be used to rule in appendicitis. They are best used to assist in ruling out appendicitis and to aid in deciding for or against imaging.
Continue to: A look at the imaging options
A look at the imaging options
Abdominal CT has sensitivity and specificity rates between 76% and 100% and 83% and 100%, respectively.15,20,21 Ultrasonography has sensitivity and specificity rates of 71% to 94% and 81% to 98%, respectively.15,20,21 Formal US is reliable to confirm appendicitis, but less so to rule out appendicitis. Special considerations for imagining in pregnant patients and children are discussed in a bit.
Timing of surgical consultation
Surgical consultation is paramount once the diagnosis of appendicitis is probable. Imaging is best obtained prior to surgical consultation to streamline evaluation and enhance decision- making. Typically, patients will be categorized as complicated or uncomplicated based on the presence or absence of perforation, a gangrenous appendix, an intra-abdominal abscess (IAA), or purulent peritonitis. Active continuous surgical involvement (co-management or assumption of care) is recommended in all cases of appendicitis, especially if nonoperative management is selected, given that some cases must convert to immediate operative treatment or may be selected for delayed future (interval) appendectomy.22
Management
Uncomplicated appendicitis
Prompt appendectomy has been the gold standard of care for uncomplicated acute appendicitis for 60 years. However, several studies have investigated an antibiotic-based strategy rather than surgical treatment for uncomplicated appendicitis.
Antibiotics vs appendectomy. In 2020, the CODA Collaborative published a randomized trial comparing a 10-day course of antibiotics with appendectomy in patients with uncomplicated appendicitis. In this multicenter study based in the United States, 1552 patients 18 years of age or older were randomized to receive antibiotics or undergo appendectomy (95% performed laparoscopically). The antibiotic treatment consisted of at least 24 hours of IV antibiotics, with or without admission to the hospital. Antibiotic choice was individualized according to guidelines for intra-abdominal infection published by the Infectious Diseases Society of America, with the most common IV medications being ertapenem, cefoxitin, or metronidazole plus one of the following: ceftriaxone, cefazolin, or levofloxacin. For the remaining 10 days, oral metronidazole plus ciprofloxacin or cefdinir were used.22
Continue to: The primary endpoint...
The primary endpoint was the European Quality of Life-5 Dimensions (EQ-5D) questionnaire, with secondary outcomes including appendectomy in the antibiotics group and complications through 90 days. Exclusion criteria included pregnancy, sepsis, peritonitis, recurrent appendicitis, severe phlegmon on imaging, or evidence of neoplasm.22
Antibiotics were noninferior to appendectomy for the 30-day study. However, antibiotics failed in 29%, who then proceeded to appendectomy by 90 days; these patients also accounted for 41% of those with an appendicolith. Overall complications were more common in the antibiotics group than in the appendectomy group (8.1 vs 3.5 per 100 participants; 95% CI, 1.3-3.98). Also more common in the antibiotic group were serious adverse events (4 vs 3 per 100 participants; hazard ratio [HR] = 1.29; 95% CI, 0.67-2.50). The presence of an appendicolith in the antibiotics group increased the conversion risk to appendectomy, as well as adverse events risk.22
The takeaway. Antibiotic treatment is a noninferior method to treat acute uncomplicated appendicitis. However, the informed consent process is important, given the ~30% failure rate. Patient factors such as continued access to care should help inform the decision.
Two main surgical approaches exist for appendectomy: open and minimally invasive. At this time, the minimally invasive options include laparoscopic, single incision laparoscopic surgery (SILS), and robotic appendectomy. A study comparing cost, availability, or complications of these options has not been conducted at this time.
A large Cochrane review of 67 studies examining open vs laparoscopic appendectomy in adults and children completed in 2018 revealed that the laparoscopic approach reduced early postoperative pain intensity and led to a shorter hospital stay, earlier return to work or usual activities, and a decrease in wound infections.23 The odds of IAA occurring with laparoscopic appendectomy increased by 65% compared with an open procedure; however, postoperative bowel obstruction and incisional hernias were less likely to occur.23 Additionally, following laparoscopic surgery, postoperative bowel obstruction and incisional hernias are less likely to occur. The laparoscopic approach is preferred due to overall increased patient satisfaction and a reduction in most, if not all, complications.
Continue to: Complicated appendicitis
Complicated appendicitis
Excluding patients with severe sepsis or purulent peritonitis requiring resuscitation and immediate surgical intervention of intra-abdominal infection, the approach to patients with complicated appendicitis varies between aggressive surgical intervention and nonoperative management.
In a 2007 meta-analysis reviewing nonsurgical treatment of appendiceal abscess/phlegmon, immediate surgery was associated with higher morbidity.24 Within the nonoperative management group 7.2% (CI, 4.0-10.5) required surgical intervention and 19.7% (CI, 11.0-28.3) required abscess drainage. Malignant disease was detected in 1.2% (CI, 0.6-1.7).24 Small subsequent studies concluded different results.25
Ultimately, the 2015 European Association of Endoscopic Surgery guidelines recommend a new systematic review; but with current data, initial nonoperative management is preferred.15 After initial nonoperative treatment, the only benefits from interval appendectomy are identification of an underlying malignancy (6% to 20%) and mitigating the risk of recurrent appendicitis (5% to 44%).15,25-30
Multiple single institutional series found increased neoplasm incidence (9% to 20%) in complicated appendicitis in patients 40 years and older.26-30 Prior to interval appendectomy in patients 45 years and older, ensuring they have an up-to-date screening colonoscopy is important. This is in line with 2021 US Preventive Services Task Force (Grade “B” recommendation), 2018 American Cancer Society (qualified recommendation), and 2021 American College of Gastroenterology (conditional recommendation) guidelines for colorectal cancer screening to start at age 45 in average-risk patients.31 Patients younger than 45 can consider screening through shared decision-making.
Special populations
Pregnant patients
In pregnancy, challenges exist with the presence of traditional signs and symptoms of appendicitis, with the most predictive sign being a WBC count higher than 18,000.32 The American College of Radiology’s (ACR) Appropriateness Criteria recommend US as the imaging modality of choice in pregnancy, with MRI as the best option when US is inconclusive.33 Two meta-analyses demonstrated high sensitivity (91.8%-96.6%) and specificity (95.9%-97.9%) of MRI in diagnosing appendicitis.34,35 CT scan is not the preferred initial imagining modality in pregnancy unless urgent information is needed and other modalities are insufficient or unavailable.36
Continue to: The most common...
The most common nonobstetric surgical intervention during pregnancy is appendectomy, at a rate of 6.3/10,000 person-years, which increases to 9.9/10,000 in the postpartum period.37 Two large population studies demonstrate the rate of appendicitis varies over the course of pregnancy, with the lowest rates in the third trimester,38,39 and a significant rebound lasting for 2 years postpartum.39 Peritonitis, septic shock, pneumonia, postoperative infection, and longer hospital stays occur more frequently in pregnant women than in nonpregnant women with appendicitis.40 Fetal loss is higher in the first trimester.32
In a 14-year review of 63,145 appendicitis cases, an increased risk of fetal loss and maternal death was noted across ages and ethnicities, with the largest risk of maternal death occurring in Hispanics and fetal death in non-Hispanic Blacks.41 In a large study of 1018 adverse events after appendectomy or cholecystectomy, the 3 most common events were preterm delivery (35.4%), preterm labor without preterm delivery (26.4%), and miscarriage (25.7%).42 The surgery itself was not a major risk factor for adverse events. Major risk factors included cervical incompetence (odds ratio [OR] = 24.3), preterm labor in current pregnancy (OR = 18.3), and presence of vulvovaginitis (OR = 5.2).42
Nonoperative management in pregnancy is not recommended; only 1 prospective trial has been done, with 20 patients, showing a 25% failure rate.43 Two meta-analyses published in 2019 highlight the potential increase of fetal loss with laparoscopic approaches to appendectomy.44,45 However, recently published literature demonstrates no significant maternal-fetal morbidity. Current guidelines of the Society of American Gastrointestinal and Endoscopic Surgeons agree that laparoscopy is the operative choice in pregnancy.36
Children
Acute appendicitis is the most common surgical emergency in children.4 Physical exam findings and laboratory results are not classic in this population, obtaining an accurate history can be challenging, and results of clinical scoring systems can be inconclusive.4 Additional serum biomarkers, procalcitonin and calprotectin, are gaining evidence for use in improving scoring systems to refine low-risk groups. Unavailability of timely, reliable biomarker testing in rural practice locations limits definitive recommendations at this time.46 ACR recommends no imaging in a pediatric patient whose risk of having appendicitis is low based on any of several scoring systems.47 For those assessed as having higher risk, US is the recommended initial modality,with CT with IV contrast or MRI without contrast equally recommended if the US is equivocal.47
Despite promising data from trials of nonoperative treatment for adults with appendicitis, no definitive evidence and recommendations are available for children. Two systematic reviews show nonoperative treatment is safe, with an efficacy rate of 76% to 82% at long-term follow-up,48,49 although the success of antibiotic regimens varies. Within the nonoperative treatment group, 16% of patients had appendectomy during the follow-up period, which varied from 8 weeks to 4 years.48 A randomized controlled trial is needed for final guidance.
Continue to: CASE
CASE
The patient had an Alvarado score of 9 (high probability) and an AIR score of 6 (intermediate probability). A CT with IV contrast showed a 9-mm fluid-filled appendix with periappendiceal fluid. During surgical consultation, he was offered laparoscopic appendectomy or nonoperative treatment with antibiotics. He opted for a preoperative dose of piperacillin-tazobactam 3.375 g IV and laparoscopic appendectomy. The patient was discharged home 6 hours after his procedure.
CORRESPONDENCE
Jessica Servey, MD, MHPE, 4301 Jones Bridge Road, Bethesda, MD 20814; [email protected]
CASE
A 35-year-old man with a body mass index of 20 presented to the emergency department after 24 hours of abdominal pain that began in the periumbilical region and then migrated to the right lower quadrant. The pain was exacerbated during ambulation and was intense when the car transporting him to the hospital encountered bumps in the road. After his pain started, he had associated anorexia, followed by nausea and emesis. He reported fever and chills. On examination, his temperature was 100.8 °F (38.2 °C), and palpation of the right and left lower quadrants elicited right lower quadrant pain. Laboratory evaluation revealed a white blood cell (WBC) count of 14,000 cells/mcL with 85% neutrophils, C-reactive protein of 40 mg/L, and a negative urinalysis.
How would you proceed with this patient?
Acute appendicitis is the most common cause of abdominal pain resulting in the need for surgical treatment; lifetime risk of appendicitis is 6% to 7%.1 Appendicitis is caused by intraluminal obstruction in the appendix from enlarged lymphoid tissue or a fecalith. The obstruction leads to elevated intraluminal pressure due to persistent mucus and gas production by bacteria, ultimately leading to ischemia and perforation.1 Additionally, obstruction leads to bacterial overgrowth, most commonly colonic flora such as Escherichia coli, Bacteroides fragilis, Streptococcus viridans, Enterococcus sp., Pseudomonas aeruginosa, and Klebsiella pneumoniaei.1,2
The following review provides a look at how 3 clinical scoring systems compare in the identification of acute appendicitis and details which imaging studies you should order—and when. But first, we’ll quickly detail the relevant physical findings and lab values that point to a diagnosis of acute appendicitis.
Physical findings. The patient typically first experiences vague abdominal pain that then localizes to the right lower quadrant due to peritoneal inflammation. Anorexia and nausea typically follow the abdominal pain. On examination, the patient often appears ill and exhibits abdominal guarding due to peritonitis. Tachycardia and fever are common; however, the absence of either does not exclude appendicitis. Classically, on palpation, the patient will have pain at McBurney’s point (one-third the distance from the anterior iliac spine to the umbilicus). The exact point of maximal tenderness can differ because of the varying anatomy of the appendix (retrocecal, paracolic, pelvic, pre/post ileal, promontoric, or subcecal).1 Right lower quadrant pain, abdominal rigidity, and radiation of periumbilical pain to the right lower quadrant are the most accurate findings in adults to rule in appendicitis.3 For children, physical exam findings have the highest likelihood in predicting appendicitis and include a positive Obturator sign, positive Rovsing sign, or a positive Psoas sign, and absent or decreased bowel sounds.4
Laboratory studies can support a diagnosis of appendicitis but cannot exclude it. Leukocytosis with neutrophil predominance is present in 90% of cases.5 An elevated C-reactive protein level renders the highest diagnostic accuracy.5 Perform a pregnancy test for any woman of child-bearing age, to assist in the diagnosis and guide imaging choices for evaluation. Additional laboratory tests are not needed unless there are concerns about volume depletion.
Clinical scoring systems
Several clinical scoring systems (TABLE6-10) have been validated to aid clinicians in evaluating patients with possible appendicitis, to decrease unnecessary exposure to ionizing radiation from computed tomography (CT) scans, to identify and reassure patients with low likelihoods of appendicitis, and to conduct outpatient follow-up.

Continue to: The Alvarado score
The Alvarado score is the oldest scoring rule, developed in 1986; it entails 8 clinical and laboratory variables.6 Ebell et al altered the proposed cutoff values of the Alvarado score to be low risk (< 4), intermediate risk (4-8), and high risk (≥ 9), effectively improving the sensitivity and specificity rates.7
In a meta-analysis of the Alvarado score that included 42 studies of men, women, and children, the sensitivity for “ruling out” appendicitis with a cutoff of 5 points was 96% for men, 99% for women, and 99% for children.8 The accuracy of a high-risk score (> 7) for “ruling in” appendicitis was less with an overall specificity of 82%.8 The Alvarado score did seem to overestimate appendicitis in women in all score categories.8
The Pediatric Appendicitis Score (PAS) is similar to Alvarado and was prospectively validated in 1170 children in 2002 for more specific guidance in this age group.9 The PAS had excellent specificity in the study; those with a score of ≥ 6 had a high probability of appendicitis. In a study comparing Alvarado with PAS in 311 patients, insignificant differences were noted at a score of ≥ 7 for both tests (sensitivity 86% vs 89%, and specificity 59% vs 50%, respectively).11 No scoring system has been found to be sufficiently accurate for use in children 4 years of age and younger.12
The Appendicitis Inflammatory Response (AIR) Score was prospectively validated in 545 patients representing all age groups.10 Subsequently, in a larger prospective multicenter study of 3878 patients older than 5 years, the original cut points were altered, thereby improving test sensitivity and negative predictive value to 99% for those with low probability (0 to 3), and test specificity to 98% for those with high-probability (9 to 12).13 Compared with the Alvarado Score, the AIR Score has higher specificity for those in the high-probability range, and similar exclusion rates in the low-probability range.14
Caveats with clinical decision scores. These tools are accepted and often used. However, challenges that affect generalizability of study data include differences in patient selection for each study (undifferentiated abdominal pain vs appendicitis), prospective vs retrospective designs, and age and gender variations in the patient populations. Despite the numerous scoring systems developed, none can accurately be used to rule in appendicitis. They are best used to assist in ruling out appendicitis and to aid in deciding for or against imaging.
Continue to: A look at the imaging options
A look at the imaging options
Abdominal CT has sensitivity and specificity rates between 76% and 100% and 83% and 100%, respectively.15,20,21 Ultrasonography has sensitivity and specificity rates of 71% to 94% and 81% to 98%, respectively.15,20,21 Formal US is reliable to confirm appendicitis, but less so to rule out appendicitis. Special considerations for imagining in pregnant patients and children are discussed in a bit.
Timing of surgical consultation
Surgical consultation is paramount once the diagnosis of appendicitis is probable. Imaging is best obtained prior to surgical consultation to streamline evaluation and enhance decision- making. Typically, patients will be categorized as complicated or uncomplicated based on the presence or absence of perforation, a gangrenous appendix, an intra-abdominal abscess (IAA), or purulent peritonitis. Active continuous surgical involvement (co-management or assumption of care) is recommended in all cases of appendicitis, especially if nonoperative management is selected, given that some cases must convert to immediate operative treatment or may be selected for delayed future (interval) appendectomy.22
Management
Uncomplicated appendicitis
Prompt appendectomy has been the gold standard of care for uncomplicated acute appendicitis for 60 years. However, several studies have investigated an antibiotic-based strategy rather than surgical treatment for uncomplicated appendicitis.
Antibiotics vs appendectomy. In 2020, the CODA Collaborative published a randomized trial comparing a 10-day course of antibiotics with appendectomy in patients with uncomplicated appendicitis. In this multicenter study based in the United States, 1552 patients 18 years of age or older were randomized to receive antibiotics or undergo appendectomy (95% performed laparoscopically). The antibiotic treatment consisted of at least 24 hours of IV antibiotics, with or without admission to the hospital. Antibiotic choice was individualized according to guidelines for intra-abdominal infection published by the Infectious Diseases Society of America, with the most common IV medications being ertapenem, cefoxitin, or metronidazole plus one of the following: ceftriaxone, cefazolin, or levofloxacin. For the remaining 10 days, oral metronidazole plus ciprofloxacin or cefdinir were used.22
Continue to: The primary endpoint...
The primary endpoint was the European Quality of Life-5 Dimensions (EQ-5D) questionnaire, with secondary outcomes including appendectomy in the antibiotics group and complications through 90 days. Exclusion criteria included pregnancy, sepsis, peritonitis, recurrent appendicitis, severe phlegmon on imaging, or evidence of neoplasm.22
Antibiotics were noninferior to appendectomy for the 30-day study. However, antibiotics failed in 29%, who then proceeded to appendectomy by 90 days; these patients also accounted for 41% of those with an appendicolith. Overall complications were more common in the antibiotics group than in the appendectomy group (8.1 vs 3.5 per 100 participants; 95% CI, 1.3-3.98). Also more common in the antibiotic group were serious adverse events (4 vs 3 per 100 participants; hazard ratio [HR] = 1.29; 95% CI, 0.67-2.50). The presence of an appendicolith in the antibiotics group increased the conversion risk to appendectomy, as well as adverse events risk.22
The takeaway. Antibiotic treatment is a noninferior method to treat acute uncomplicated appendicitis. However, the informed consent process is important, given the ~30% failure rate. Patient factors such as continued access to care should help inform the decision.
Two main surgical approaches exist for appendectomy: open and minimally invasive. At this time, the minimally invasive options include laparoscopic, single incision laparoscopic surgery (SILS), and robotic appendectomy. A study comparing cost, availability, or complications of these options has not been conducted at this time.
A large Cochrane review of 67 studies examining open vs laparoscopic appendectomy in adults and children completed in 2018 revealed that the laparoscopic approach reduced early postoperative pain intensity and led to a shorter hospital stay, earlier return to work or usual activities, and a decrease in wound infections.23 The odds of IAA occurring with laparoscopic appendectomy increased by 65% compared with an open procedure; however, postoperative bowel obstruction and incisional hernias were less likely to occur.23 Additionally, following laparoscopic surgery, postoperative bowel obstruction and incisional hernias are less likely to occur. The laparoscopic approach is preferred due to overall increased patient satisfaction and a reduction in most, if not all, complications.
Continue to: Complicated appendicitis
Complicated appendicitis
Excluding patients with severe sepsis or purulent peritonitis requiring resuscitation and immediate surgical intervention of intra-abdominal infection, the approach to patients with complicated appendicitis varies between aggressive surgical intervention and nonoperative management.
In a 2007 meta-analysis reviewing nonsurgical treatment of appendiceal abscess/phlegmon, immediate surgery was associated with higher morbidity.24 Within the nonoperative management group 7.2% (CI, 4.0-10.5) required surgical intervention and 19.7% (CI, 11.0-28.3) required abscess drainage. Malignant disease was detected in 1.2% (CI, 0.6-1.7).24 Small subsequent studies concluded different results.25
Ultimately, the 2015 European Association of Endoscopic Surgery guidelines recommend a new systematic review; but with current data, initial nonoperative management is preferred.15 After initial nonoperative treatment, the only benefits from interval appendectomy are identification of an underlying malignancy (6% to 20%) and mitigating the risk of recurrent appendicitis (5% to 44%).15,25-30
Multiple single institutional series found increased neoplasm incidence (9% to 20%) in complicated appendicitis in patients 40 years and older.26-30 Prior to interval appendectomy in patients 45 years and older, ensuring they have an up-to-date screening colonoscopy is important. This is in line with 2021 US Preventive Services Task Force (Grade “B” recommendation), 2018 American Cancer Society (qualified recommendation), and 2021 American College of Gastroenterology (conditional recommendation) guidelines for colorectal cancer screening to start at age 45 in average-risk patients.31 Patients younger than 45 can consider screening through shared decision-making.
Special populations
Pregnant patients
In pregnancy, challenges exist with the presence of traditional signs and symptoms of appendicitis, with the most predictive sign being a WBC count higher than 18,000.32 The American College of Radiology’s (ACR) Appropriateness Criteria recommend US as the imaging modality of choice in pregnancy, with MRI as the best option when US is inconclusive.33 Two meta-analyses demonstrated high sensitivity (91.8%-96.6%) and specificity (95.9%-97.9%) of MRI in diagnosing appendicitis.34,35 CT scan is not the preferred initial imagining modality in pregnancy unless urgent information is needed and other modalities are insufficient or unavailable.36
Continue to: The most common...
The most common nonobstetric surgical intervention during pregnancy is appendectomy, at a rate of 6.3/10,000 person-years, which increases to 9.9/10,000 in the postpartum period.37 Two large population studies demonstrate the rate of appendicitis varies over the course of pregnancy, with the lowest rates in the third trimester,38,39 and a significant rebound lasting for 2 years postpartum.39 Peritonitis, septic shock, pneumonia, postoperative infection, and longer hospital stays occur more frequently in pregnant women than in nonpregnant women with appendicitis.40 Fetal loss is higher in the first trimester.32
In a 14-year review of 63,145 appendicitis cases, an increased risk of fetal loss and maternal death was noted across ages and ethnicities, with the largest risk of maternal death occurring in Hispanics and fetal death in non-Hispanic Blacks.41 In a large study of 1018 adverse events after appendectomy or cholecystectomy, the 3 most common events were preterm delivery (35.4%), preterm labor without preterm delivery (26.4%), and miscarriage (25.7%).42 The surgery itself was not a major risk factor for adverse events. Major risk factors included cervical incompetence (odds ratio [OR] = 24.3), preterm labor in current pregnancy (OR = 18.3), and presence of vulvovaginitis (OR = 5.2).42
Nonoperative management in pregnancy is not recommended; only 1 prospective trial has been done, with 20 patients, showing a 25% failure rate.43 Two meta-analyses published in 2019 highlight the potential increase of fetal loss with laparoscopic approaches to appendectomy.44,45 However, recently published literature demonstrates no significant maternal-fetal morbidity. Current guidelines of the Society of American Gastrointestinal and Endoscopic Surgeons agree that laparoscopy is the operative choice in pregnancy.36
Children
Acute appendicitis is the most common surgical emergency in children.4 Physical exam findings and laboratory results are not classic in this population, obtaining an accurate history can be challenging, and results of clinical scoring systems can be inconclusive.4 Additional serum biomarkers, procalcitonin and calprotectin, are gaining evidence for use in improving scoring systems to refine low-risk groups. Unavailability of timely, reliable biomarker testing in rural practice locations limits definitive recommendations at this time.46 ACR recommends no imaging in a pediatric patient whose risk of having appendicitis is low based on any of several scoring systems.47 For those assessed as having higher risk, US is the recommended initial modality,with CT with IV contrast or MRI without contrast equally recommended if the US is equivocal.47
Despite promising data from trials of nonoperative treatment for adults with appendicitis, no definitive evidence and recommendations are available for children. Two systematic reviews show nonoperative treatment is safe, with an efficacy rate of 76% to 82% at long-term follow-up,48,49 although the success of antibiotic regimens varies. Within the nonoperative treatment group, 16% of patients had appendectomy during the follow-up period, which varied from 8 weeks to 4 years.48 A randomized controlled trial is needed for final guidance.
Continue to: CASE
CASE
The patient had an Alvarado score of 9 (high probability) and an AIR score of 6 (intermediate probability). A CT with IV contrast showed a 9-mm fluid-filled appendix with periappendiceal fluid. During surgical consultation, he was offered laparoscopic appendectomy or nonoperative treatment with antibiotics. He opted for a preoperative dose of piperacillin-tazobactam 3.375 g IV and laparoscopic appendectomy. The patient was discharged home 6 hours after his procedure.
CORRESPONDENCE
Jessica Servey, MD, MHPE, 4301 Jones Bridge Road, Bethesda, MD 20814; [email protected]
1. Prystowsky JB, Pugh CM, Nagle AP. Current problems in surgery. Appendicitis. Curr Probl Surg. 2005;42:688-742.
2. Song DW, Park BK, Suh SW, et al. Bacterial culture and antibiotic susceptibility in patients with acute appendicitis. Int J Colorectal Dis. 2018;33:441-447.
3. Wagner JM, McKinney WP, Carpenter JL. Does this patient have appendicitis? JAMA. 1996;276:1589-1594.
4. Benabbas R, Hanna M, Shah J, et al. Diagnostic accuracy of history, physical examination, laboratory tests, and point-of-care ultrasound for pediatric acute appendicitis in the emergency department: a systematic review and meta-analysis. Acad Emerg Med. 2017;24:523-551.
5. Andersson RE. Meta-analysis of the clinical and laboratory diagnosis of appendicitis. Br J Surg. 2004;91:28-37.
6. Alvarado A. A practical score for the early diagnosis of acute appendicitis. Ann Emerg Med. 1986;15:557-564.
7. Ebell MH, Shinholser J. What are the most clinically useful cutoffs for the Alvarado and Pediatric Appendicitis Scores? A systematic review. Ann Emerg Med. 2014;64:365-372.e2.
8. Ohle R, O’Reilly F, O’Brien KK, et al. The Alvarado score for predicting acute appendicitis: a systematic review. BMC Med. 2011;9:139.
9. Samuel M. Pediatric appendicitis score. J Pediatr Surg. 2002;37:877-881.
10. Andersson M, Andersson RE. The appendicitis inflammatory response score: a tool for the diagnosis of acute appendicitis that outperforms the Alvarado score. World J Surg. 2008;32:1843-1849.
11. Pogorelić Z, Rak S, Mrklić I, et al. Prospective validation of Alvarado score and Pediatric Appendicitis Score for the diagnosis of acute appendicitis in children. Pediatr Emerg Care. 2015;31:164-168.
12. Rassi R, Muse F, Sánchez-Martínez J, et al. Diagnostic value of clinical prediction scores for acute appendicitis in children younger than 4 years. Eur J Pediatr Surg. 2021. [Online ahead of print]
13. Andersson M, Kolodziej B, Andersson RE. Validation of the Appendicitis Inflammatory Response (AIR) score. World J Surg. 2021;45:2081-2091.
14. Kollár D, McCartan DP, Bourke M, et al. Predicting acute appendicitis? A comparison of the Alvarado score, the Appendicitis Inflammatory Response Score and clinical assessment. World J Surg. 2015;39:104-109.
15. Gorter RR, Eker HH, Gorter-Stam MA, et al. Diagnosis and management of acute appendicitis. EAES consensus development conference 2015. Surg Endosc. 2016;30:4668-4690.
16. Matthew Fields J, Davis J, Alsup C, et al. Accuracy of point-of-care ultrasonography for diagnosing acute appendicitis: a systematic review and meta-analysis. Acad Emerg Med. 2017;24:1124-1136.
17. Sharif S, Skitch S, Vlahaki D, et al. Point-of-care ultrasound to diagnose appendicitis in a Canadian emergency department. CJEM. 2018;20:732-735.
18. Doniger SJ, Kornblith A. Point-of-care ultrasound integrated into a staged diagnostic algorithm for pediatric appendicitis. Pediatr Emerg Care. 2018;34:109-115.
19. Menon N, Kumar S, Keeler B, et al. A systematic review of point-of-care abdominal ultrasound scans performed by general surgeons. Surgeon. 2021. [Online ahead of print]
20. Doria AS, Moineddin R, Kellenberger CJ, et al. US or CT for diagnosis of appendicitis in children and adults? A meta-analysis. Radiology. 2006;241:83-94.
21. van Randen A, Laméris W, van Es HW, et al. A comparison of the accuracy of ultrasound and computed tomography in common diagnoses causing acute abdominal pain. Eur Radiol. 2011;21:1535-1545.
22. Flum DR, Davidson GH, Monsell SE, et al. A randomized trial comparing antibiotics with appendectomy for appendicitis. N Engl J Med. 2020;383:1907-1919.
23. Jaschinski T, Mosch CG, Eikermann M, et al. Laparoscopic versus open surgery for suspected appendicitis. Cochrane Database Syst Rev. 2018;11:CD001546.
24. Andersson RE, Petzold MG. Nonsurgical treatment of appendiceal abscess or phlegmon: a systematic review and meta-analysis. Ann Surg. 2007;246:741-748.
25. Deelder JD, Richir MC, Schoorl T, et al. How to treat an appendiceal inflammatory mass: operatively or nonoperatively? J Gastrointest Surg. 2014;18:641-645.
26. Carpenter SG, Chapital AB, Merritt MV, et al. Increased risk of neoplasm in appendicitis treated with interval appendectomy: single-institution experience and literature review. Am Surg. 2012;78:339-343.
27. Hayes D, Reiter S, Hagen E, et al. Is interval appendectomy really needed? A closer look at neoplasm rates in adult patients undergoing interval appendectomy after complicated appendicitis. Surg Endosc. 2021;35:3855-3860.
28. Peltrini R, Cantoni V, Green R, et al. Risk of appendiceal neoplasm after interval appendectomy for complicated appendicitis: a systematic review and meta-analysis. Surgeon. 2021. [Online ahead of print.]
29. Mällinen J, Rautio T, Grönroos J, et al. Risk of appendiceal neoplasm in periappendicular abscess in patients treated with interval appendectomy vs follow-up with magnetic resonance imaging: 1-year outcomes of the peri-appendicitis acuta randomized clinical trial. JAMA Surg. 2019;154:200-207.
30. Son J, Park YJ, Lee SR, et al. Increased risk of neoplasms in adult patients undergoing interval appendectomy. Ann Coloproctol. 2020;36:311-315.
31. Davidson KW, Barry MJ, Mangione CM, et al. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325:1965-1977.
32. Theilen LH, Mellnick VM, Shanks AL, et al. Acute appendicitis in pregnancy: predictive clinical factors and pregnancy outcomes. Am J Perinatol. 2017;34:523-528.
33. Garcia EM, Camacho MA, Karolyi DR, et al. ACR Appropriateness Criteria right lower quadrant pain-suspected appendicitis. J Am Coll Radiol. 2018;15:S373-s387.
34. Kave M, Parooie F, Salarzaei M. Pregnancy and appendicitis: a systematic review and meta-analysis on the clinical use of MRI in diagnosis of appendicitis in pregnant women. World J Emerg Surg. 2019;14:37.
35. Repplinger MD, Levy JF, Peethumnongsin E, et al. Systematic review and meta-analysis of the accuracy of MRI to diagnose appendicitis in the general population. J Magn Reson Imaging. 2016;43:1346-1354.
36. Pearl JP, Price RR, Tonkin AE, et al. SAGES guidelines for the use of laparoscopy during pregnancy. Surg Endosc. 2017;31:3767-3782.
37. Zingone F, Sultan AA, Humes DJ, et al. Risk of acute appendicitis in and around pregnancy: a population-based cohort study from England. Ann Surg. 2015;261:332-337.
38. Andersson RE, Lambe M. Incidence of appendicitis during pregnancy. Int J Epidemiol. 2001;30:1281-1285.
39. Moltubak E, Landerholm K, Blomberg M, et al. Major variation in the incidence of appendicitis before, during and after pregnancy: a population-based cohort study. World J Surg. 2020;44:2601-2608.
40. Abbasi N, Patenaude V, Abenhaim HA. Management and outcomes of acute appendicitis in pregnancy-population-based study of over 7000 cases. BJOG. 2014;121:1509-1514.
41. Dongarwar D, Taylor J, Ajewole V, et al. Trends in appendicitis among pregnant women, the risk for cardiac arrest, and maternal-fetal mortality. World J Surg. 2020;44:3999-4005.
42. Sachs A, Guglielminotti J, Miller R, et al. Risk factors and risk stratification for adverse obstetrical outcomes after appendectomy or cholecystectomy during pregnancy. JAMA Surg. 2017;152:436-441.
43. Joo JI, Park HC, Kim MJ, et al. Outcomes of antibiotic therapy for uncomplicated appendicitis in pregnancy. Am J Med. 2017;130:1467-1469.
44. Lee SH, Lee JY, Choi YY, Lee JG. Laparoscopic appendectomy versus open appendectomy for suspected appendicitis during pregnancy: a systematic review and updated meta-analysis. BMC Surg. 2019;19:41.
45. Frountzas M, Nikolaou C, Stergios K, et al. Is the laparoscopic approach a safe choice for the management of acute appendicitis in pregnant women? A meta-analysis of observational studies. Ann R Coll Surg Engl. 2019;101:235-248.
46. Di Saverio S, Podda M, De Simone B, et al. Diagnosis and treatment of acute appendicitis: 2020 update of the WSES Jerusalem guidelines. World J Emerg Surg. 2020;15:27.
47. Koberlein GC, Trout AT, Rigsby CK, et al. ACR Appropriateness Criteria suspected appendicitis-child. J Am Coll Radiol. 2019;16:S252-S263.
48. Maita S, Andersson B, Svensson JF, et al. Nonoperative treatment for nonperforated appendicitis in children: a systematic review and meta-analysis. Pediatr Surg Int. 2020;36:261-269.
49. Georgiou R, Eaton S, Stanton MP, et al. Efficacy and safety of nonoperative treatment for acute appendicitis: a meta-analysis. Pediatrics. 2017;139:e20163003.
1. Prystowsky JB, Pugh CM, Nagle AP. Current problems in surgery. Appendicitis. Curr Probl Surg. 2005;42:688-742.
2. Song DW, Park BK, Suh SW, et al. Bacterial culture and antibiotic susceptibility in patients with acute appendicitis. Int J Colorectal Dis. 2018;33:441-447.
3. Wagner JM, McKinney WP, Carpenter JL. Does this patient have appendicitis? JAMA. 1996;276:1589-1594.
4. Benabbas R, Hanna M, Shah J, et al. Diagnostic accuracy of history, physical examination, laboratory tests, and point-of-care ultrasound for pediatric acute appendicitis in the emergency department: a systematic review and meta-analysis. Acad Emerg Med. 2017;24:523-551.
5. Andersson RE. Meta-analysis of the clinical and laboratory diagnosis of appendicitis. Br J Surg. 2004;91:28-37.
6. Alvarado A. A practical score for the early diagnosis of acute appendicitis. Ann Emerg Med. 1986;15:557-564.
7. Ebell MH, Shinholser J. What are the most clinically useful cutoffs for the Alvarado and Pediatric Appendicitis Scores? A systematic review. Ann Emerg Med. 2014;64:365-372.e2.
8. Ohle R, O’Reilly F, O’Brien KK, et al. The Alvarado score for predicting acute appendicitis: a systematic review. BMC Med. 2011;9:139.
9. Samuel M. Pediatric appendicitis score. J Pediatr Surg. 2002;37:877-881.
10. Andersson M, Andersson RE. The appendicitis inflammatory response score: a tool for the diagnosis of acute appendicitis that outperforms the Alvarado score. World J Surg. 2008;32:1843-1849.
11. Pogorelić Z, Rak S, Mrklić I, et al. Prospective validation of Alvarado score and Pediatric Appendicitis Score for the diagnosis of acute appendicitis in children. Pediatr Emerg Care. 2015;31:164-168.
12. Rassi R, Muse F, Sánchez-Martínez J, et al. Diagnostic value of clinical prediction scores for acute appendicitis in children younger than 4 years. Eur J Pediatr Surg. 2021. [Online ahead of print]
13. Andersson M, Kolodziej B, Andersson RE. Validation of the Appendicitis Inflammatory Response (AIR) score. World J Surg. 2021;45:2081-2091.
14. Kollár D, McCartan DP, Bourke M, et al. Predicting acute appendicitis? A comparison of the Alvarado score, the Appendicitis Inflammatory Response Score and clinical assessment. World J Surg. 2015;39:104-109.
15. Gorter RR, Eker HH, Gorter-Stam MA, et al. Diagnosis and management of acute appendicitis. EAES consensus development conference 2015. Surg Endosc. 2016;30:4668-4690.
16. Matthew Fields J, Davis J, Alsup C, et al. Accuracy of point-of-care ultrasonography for diagnosing acute appendicitis: a systematic review and meta-analysis. Acad Emerg Med. 2017;24:1124-1136.
17. Sharif S, Skitch S, Vlahaki D, et al. Point-of-care ultrasound to diagnose appendicitis in a Canadian emergency department. CJEM. 2018;20:732-735.
18. Doniger SJ, Kornblith A. Point-of-care ultrasound integrated into a staged diagnostic algorithm for pediatric appendicitis. Pediatr Emerg Care. 2018;34:109-115.
19. Menon N, Kumar S, Keeler B, et al. A systematic review of point-of-care abdominal ultrasound scans performed by general surgeons. Surgeon. 2021. [Online ahead of print]
20. Doria AS, Moineddin R, Kellenberger CJ, et al. US or CT for diagnosis of appendicitis in children and adults? A meta-analysis. Radiology. 2006;241:83-94.
21. van Randen A, Laméris W, van Es HW, et al. A comparison of the accuracy of ultrasound and computed tomography in common diagnoses causing acute abdominal pain. Eur Radiol. 2011;21:1535-1545.
22. Flum DR, Davidson GH, Monsell SE, et al. A randomized trial comparing antibiotics with appendectomy for appendicitis. N Engl J Med. 2020;383:1907-1919.
23. Jaschinski T, Mosch CG, Eikermann M, et al. Laparoscopic versus open surgery for suspected appendicitis. Cochrane Database Syst Rev. 2018;11:CD001546.
24. Andersson RE, Petzold MG. Nonsurgical treatment of appendiceal abscess or phlegmon: a systematic review and meta-analysis. Ann Surg. 2007;246:741-748.
25. Deelder JD, Richir MC, Schoorl T, et al. How to treat an appendiceal inflammatory mass: operatively or nonoperatively? J Gastrointest Surg. 2014;18:641-645.
26. Carpenter SG, Chapital AB, Merritt MV, et al. Increased risk of neoplasm in appendicitis treated with interval appendectomy: single-institution experience and literature review. Am Surg. 2012;78:339-343.
27. Hayes D, Reiter S, Hagen E, et al. Is interval appendectomy really needed? A closer look at neoplasm rates in adult patients undergoing interval appendectomy after complicated appendicitis. Surg Endosc. 2021;35:3855-3860.
28. Peltrini R, Cantoni V, Green R, et al. Risk of appendiceal neoplasm after interval appendectomy for complicated appendicitis: a systematic review and meta-analysis. Surgeon. 2021. [Online ahead of print.]
29. Mällinen J, Rautio T, Grönroos J, et al. Risk of appendiceal neoplasm in periappendicular abscess in patients treated with interval appendectomy vs follow-up with magnetic resonance imaging: 1-year outcomes of the peri-appendicitis acuta randomized clinical trial. JAMA Surg. 2019;154:200-207.
30. Son J, Park YJ, Lee SR, et al. Increased risk of neoplasms in adult patients undergoing interval appendectomy. Ann Coloproctol. 2020;36:311-315.
31. Davidson KW, Barry MJ, Mangione CM, et al. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325:1965-1977.
32. Theilen LH, Mellnick VM, Shanks AL, et al. Acute appendicitis in pregnancy: predictive clinical factors and pregnancy outcomes. Am J Perinatol. 2017;34:523-528.
33. Garcia EM, Camacho MA, Karolyi DR, et al. ACR Appropriateness Criteria right lower quadrant pain-suspected appendicitis. J Am Coll Radiol. 2018;15:S373-s387.
34. Kave M, Parooie F, Salarzaei M. Pregnancy and appendicitis: a systematic review and meta-analysis on the clinical use of MRI in diagnosis of appendicitis in pregnant women. World J Emerg Surg. 2019;14:37.
35. Repplinger MD, Levy JF, Peethumnongsin E, et al. Systematic review and meta-analysis of the accuracy of MRI to diagnose appendicitis in the general population. J Magn Reson Imaging. 2016;43:1346-1354.
36. Pearl JP, Price RR, Tonkin AE, et al. SAGES guidelines for the use of laparoscopy during pregnancy. Surg Endosc. 2017;31:3767-3782.
37. Zingone F, Sultan AA, Humes DJ, et al. Risk of acute appendicitis in and around pregnancy: a population-based cohort study from England. Ann Surg. 2015;261:332-337.
38. Andersson RE, Lambe M. Incidence of appendicitis during pregnancy. Int J Epidemiol. 2001;30:1281-1285.
39. Moltubak E, Landerholm K, Blomberg M, et al. Major variation in the incidence of appendicitis before, during and after pregnancy: a population-based cohort study. World J Surg. 2020;44:2601-2608.
40. Abbasi N, Patenaude V, Abenhaim HA. Management and outcomes of acute appendicitis in pregnancy-population-based study of over 7000 cases. BJOG. 2014;121:1509-1514.
41. Dongarwar D, Taylor J, Ajewole V, et al. Trends in appendicitis among pregnant women, the risk for cardiac arrest, and maternal-fetal mortality. World J Surg. 2020;44:3999-4005.
42. Sachs A, Guglielminotti J, Miller R, et al. Risk factors and risk stratification for adverse obstetrical outcomes after appendectomy or cholecystectomy during pregnancy. JAMA Surg. 2017;152:436-441.
43. Joo JI, Park HC, Kim MJ, et al. Outcomes of antibiotic therapy for uncomplicated appendicitis in pregnancy. Am J Med. 2017;130:1467-1469.
44. Lee SH, Lee JY, Choi YY, Lee JG. Laparoscopic appendectomy versus open appendectomy for suspected appendicitis during pregnancy: a systematic review and updated meta-analysis. BMC Surg. 2019;19:41.
45. Frountzas M, Nikolaou C, Stergios K, et al. Is the laparoscopic approach a safe choice for the management of acute appendicitis in pregnant women? A meta-analysis of observational studies. Ann R Coll Surg Engl. 2019;101:235-248.
46. Di Saverio S, Podda M, De Simone B, et al. Diagnosis and treatment of acute appendicitis: 2020 update of the WSES Jerusalem guidelines. World J Emerg Surg. 2020;15:27.
47. Koberlein GC, Trout AT, Rigsby CK, et al. ACR Appropriateness Criteria suspected appendicitis-child. J Am Coll Radiol. 2019;16:S252-S263.
48. Maita S, Andersson B, Svensson JF, et al. Nonoperative treatment for nonperforated appendicitis in children: a systematic review and meta-analysis. Pediatr Surg Int. 2020;36:261-269.
49. Georgiou R, Eaton S, Stanton MP, et al. Efficacy and safety of nonoperative treatment for acute appendicitis: a meta-analysis. Pediatrics. 2017;139:e20163003.
PRACTICE RECOMMENDATIONS
› Use the Alvarado Score, Pediatric Appendicitis Score, or Appendicitis Inflammatory Response Score to help rule out appendicitis and thereby reduce unnecessary imaging. A
› Choose ultrasound first as the imaging procedure for children and pregnant women, followed by magnetic resonance imaging if needed, to reduce ionizing radiation in these populations. B
› Consider an antibiotic-based strategy under the care of a surgeon in lieu of immediate surgery for uncomplicated appendicitis. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Fibroids: Growing management options for a prevalent problem
OBG Manag. 33(12). | doi 10.12788/obgm.0169
Migraine Signs and Symptoms
COMMENT & CONTROVERSY
HOW TO CHOOSE THE RIGHT VAGINAL MOISTURIZER OR LUBRICANT FOR YOUR PATIENT
JOHN PENNYCUFF, MD, MSPH, AND CHERYL IGLESIA, MD (JUNE 2021)
Which vaginal products to recommend
We applaud Drs. Pennycuff and Iglesia for providing education on lubricants and vaginal moisturizers in their recent article, and agree that ObGyns, urogynecologists, and primary care providers should be aware of the types of products available. However, the authors underplayed the health risks associated with the use of poor-quality lubricants and moisturizers.
Women often turn to lubricants or vaginal moisturizers because they experience vaginal dryness during intercourse, related to menopause, and from certain medications. Vaginal fluid is primarily composed of exudate from capillaries in the vaginal wall. During sexual arousal, blood flow to the vaginal wall increases, and in turn, this should increase exudate. But chronic inflammation can suppress these increases in vaginal blood flow, preventing adequate vaginal fluid production. One such cause of chronic inflammation is using hyperosmolar lubricants, as this has been shown to negatively affect the vaginal epithelium.1,2 In this way, use of hyperosmolar lubricants can actually worsen symptoms, creating a vicious circle of dryness, lubricant use, and worsening dryness.
In addition, hyperosmolar lubricants have been shown to reduce the epithelial barrier properties of the vaginal epithelium, increasing susceptibility to microbes associated with bacterial vaginosis and to true pathogens, including herpes simplex virus type 2.3 In fact, hyperosmolar lubricants are a serious enough problem that the World Health Organization has weighed in, recommending osmolality of personal lubricants be under 380 mOsm/kg to prevent damage to the vaginal epithelium.4
Appropriately acidic pH is just as critical as osmolality. Using products with a pH higher than 4.5 will reduce amounts of protective lactobacilli and other commensal vaginal bacteria, encouraging growth of opportunistic bacteria and yeast already present. This can lead to bacterial vaginosis, aerobic vaginitis, and candidiasis. Bacterial vaginosis can lead to other serious sequelae such as increased risk in acquisition of HIV infection and preterm birth in pregnancy. Unfortunately, much of the data cited in Drs. Pennycuff and Iglesia’s article were sourced from another study (by Edwards and Panay published in Climacteric in 2016), which measured product pH values with an inappropriately calibrated device; the study’s supplemental information stated that calibration was between 5 and 9, and so any measurement below 5 was invalid and subject to error. For example, the Good Clean Love lubricant is listed as having a pH of 4.7, but its pH is never higher than 4.4.
The products on the market that meet the dual criteria of appropriate pH and isotonicity to vaginal epithelial cells may be less well known to consumers. But this should not be a reason to encourage use of hyperosmolar products whose main selling point is that they are the “leading brand.” Educating women on their choices in personal lubricants should include a full discussion of product ingredients and properties, based upon the available literature to help them select a product that supports the health of their intimate tissues.
Members of the Scientific Advisory Board for the Sexual Health and Wellness Institute: Jill Krapf, MD, MEd, IF; Cathy Chung Hwa Yi, MD; Christine Enzmann, MD, PhD, NMCP; Susan Kellogg-Spadt, PhD, CRNP, IF, CSC, FCST; Betsy Greenleaf, DO, MBA; Elizabeth DuPriest, PhD
References
- Dezzutti CS, Brown ER, Moncla B, et al. Is wetter better? An evaluation of over-the-counter personal lubricants for safety and anti-HIV-1 activity. PLoS One. 2012;7:e48328. doi: 10.1371/journal .pone.0048328.
- Ayehunie S, Wang YY, Landry T, et al. Hyperosmolal vaginal lubricants markedly reduce epithelial barrier properties in a threedimensional vaginal epithelium model. Toxicol Rep. 2017;5:134-140. doi: 10.1016 /j.toxrep.2017.12.011.
- Moench TR, Mumper RJ, Hoen TE, et al. Microbicide excipients can greatly increase susceptibility to genital herpes transmission in the mouse. BMC Infect Dis. 2010;10:331. doi: 10.1186/1471 -2334-10-331.
- Use and procurement of additional lubricants for male and female condoms: WHO/UNFPA /FHI360 Advisory note. World Health Organization, 2012. http://apps.who.int/iris/bitstream /handle/10665/76580/WHO_RHR_12.33_eng .pdf?sequence=1. Accessed December 27, 2021.
Drs. Pennycuff And Iglesia Respond
We thank the members of the scientific advisory board for the Sexual Health and Wellness Institute for their thoughtful and insightful comments to our article. We agree with their comments on the importance of both pH and osmolality for vaginal moisturizers and lubricants. We also agree that selection of an incorrectly formulated product may lead to worsening of vulvovaginal symptoms as well as dysbiosis and all of its sequelae as the letter writers mentioned.
In writing the review article, we attempted to address the role that pH and osmolality play in vaginal moisturizers and lubricants and make clinicians more aware of the importance of these factors in product formulation. Our goal was to help to improve patient counseling. We tried to amass as much of the available literature as we could to act as a resource for practitioners, such as the table included in the article as well as the supplemental table included online. We hoped that by writing this article we would heighten awareness among female health practitioners about vaginal health products and encourage them to consider those products that may be better suited for their patients based on pH and osmolality.
While there remains a paucity of research on vaginal moisturizers and lubricants, there is even less consumer knowledge regarding ingredients and formulations of these products. We wholeheartedly agree with the scientific advisory board that we as health providers need to help educate women on the full spectrum of products available beyond the “leading brands.” Furthermore, we advocate that there be continued research on these products as well as more manufacturer transparency regarding not only the ingredients contained within these products but also the pH and osmolality. Simple steps such as these would ensure that providers could help counsel patients to make informed decisions regarding products for their pelvic health.
Continue to: DISMANTLING RACISM IN YOUR PERSONAL AND PROFESSIONAL SPHERES...
DISMANTLING RACISM IN YOUR PERSONAL AND PROFESSIONAL SPHERES
CASSANDRA CARBERRY, MD, MS; ANNETTA MADSEN, MD; OLIVIA CARDENAS-TROWERS, MD; OLUWATENIOLA BROWN, MD; MOIURI SIDDIQUE, MD; AND BLAIR WASHINGTON, MD, MHA (AUGUST 2021)
Dissenting opinion
“Race is real but it’s not biologic.” “Race is not based on genetic or biologic inheritance.” Am I the only one with a dissenting voice of opinion when it comes to these types of statements?
Scott Peters, MD
Oak Ridge, Tennessee
The Authors Respond
Thank you for your opinion, Dr. Peters. Although it is not completely clear what your question is, it seems that it concerns the validity of the idea that race is a social construct. We will address this question with the assumption that this letter was an effort to invite discussion and increase understanding.
The National Human Genome Research Institute describes race in this way: “Race is a fluid concept used to group people according to various factors, including ancestral background and social identity. Race is also used to group people that share a set of visible characteristics, such as skin color and facial features. Though these visible traits are influenced by genes, the vast majority of genetic variation exists within racial groups and not between them.”1
The understanding that race is a social construct has been upheld by numerous medical organizations. In August 2020, a Joint Statement was published by the American College of Obstetricians and Gynecologists, the American Board of Obstetricians and Gynecologists, and 22 other organizations representing our specialty. This document states: “Recognizing that race is a social construct, not biologically based, is important to understanding that racism, not race, impacts health care, health, and health outcomes.”2
This idea is also endorsed by the AMA, who in November 2020 adopted the following policies3:
- “Recognize that race is a social construct and is distinct from ethnicity, genetic ancestry, or biology
- Support ending the practice of using race as a proxy for biology or genetics in medical education, research, and clinical practice.”
There are numerous sources that further illuminate why race is a social construct. Here are a few:
- https://www.racepowerofanillusion .org/resources/
- https ://www.pewresearch.org /fact-tank/2020/02/25/the-changing -categories-the-u-s-has-used-to -measure-race/
- Roberts D. Fatal Invention: How Science, Politics and Big Business Re-create Race in the Twenty-First Century. The New Press. 2011.
- Yudell M, Roberts D, DeSalle R, et al. Science and society. Taking race out of human genetics. Science. 2016;351(6273):564-5. doi: 10.1126/science.aac4951.
References
- National Human Genome Research Institute. Race. https://www.genome.gov/genetic-glossary /Race. Accessed December 27, 2021.
- The American College of Obstetricians and Gynecologists. Joint Statement: Collective Action Addressing Racism. https://www.acog.org /news/news-articles/2020/08/joint-statementobstetrics-and-gynecology-collective-actionaddressing-racism.
- O’Reilly KB. AMA: Racism is a threat to public health. November 16, 2020. https://www.ama -assn.org/delivering-care/health-equity/ama -racism-threat-public-health. Accessed December 27, 2021.
HOW TO CHOOSE THE RIGHT VAGINAL MOISTURIZER OR LUBRICANT FOR YOUR PATIENT
JOHN PENNYCUFF, MD, MSPH, AND CHERYL IGLESIA, MD (JUNE 2021)
Which vaginal products to recommend
We applaud Drs. Pennycuff and Iglesia for providing education on lubricants and vaginal moisturizers in their recent article, and agree that ObGyns, urogynecologists, and primary care providers should be aware of the types of products available. However, the authors underplayed the health risks associated with the use of poor-quality lubricants and moisturizers.
Women often turn to lubricants or vaginal moisturizers because they experience vaginal dryness during intercourse, related to menopause, and from certain medications. Vaginal fluid is primarily composed of exudate from capillaries in the vaginal wall. During sexual arousal, blood flow to the vaginal wall increases, and in turn, this should increase exudate. But chronic inflammation can suppress these increases in vaginal blood flow, preventing adequate vaginal fluid production. One such cause of chronic inflammation is using hyperosmolar lubricants, as this has been shown to negatively affect the vaginal epithelium.1,2 In this way, use of hyperosmolar lubricants can actually worsen symptoms, creating a vicious circle of dryness, lubricant use, and worsening dryness.
In addition, hyperosmolar lubricants have been shown to reduce the epithelial barrier properties of the vaginal epithelium, increasing susceptibility to microbes associated with bacterial vaginosis and to true pathogens, including herpes simplex virus type 2.3 In fact, hyperosmolar lubricants are a serious enough problem that the World Health Organization has weighed in, recommending osmolality of personal lubricants be under 380 mOsm/kg to prevent damage to the vaginal epithelium.4
Appropriately acidic pH is just as critical as osmolality. Using products with a pH higher than 4.5 will reduce amounts of protective lactobacilli and other commensal vaginal bacteria, encouraging growth of opportunistic bacteria and yeast already present. This can lead to bacterial vaginosis, aerobic vaginitis, and candidiasis. Bacterial vaginosis can lead to other serious sequelae such as increased risk in acquisition of HIV infection and preterm birth in pregnancy. Unfortunately, much of the data cited in Drs. Pennycuff and Iglesia’s article were sourced from another study (by Edwards and Panay published in Climacteric in 2016), which measured product pH values with an inappropriately calibrated device; the study’s supplemental information stated that calibration was between 5 and 9, and so any measurement below 5 was invalid and subject to error. For example, the Good Clean Love lubricant is listed as having a pH of 4.7, but its pH is never higher than 4.4.
The products on the market that meet the dual criteria of appropriate pH and isotonicity to vaginal epithelial cells may be less well known to consumers. But this should not be a reason to encourage use of hyperosmolar products whose main selling point is that they are the “leading brand.” Educating women on their choices in personal lubricants should include a full discussion of product ingredients and properties, based upon the available literature to help them select a product that supports the health of their intimate tissues.
Members of the Scientific Advisory Board for the Sexual Health and Wellness Institute: Jill Krapf, MD, MEd, IF; Cathy Chung Hwa Yi, MD; Christine Enzmann, MD, PhD, NMCP; Susan Kellogg-Spadt, PhD, CRNP, IF, CSC, FCST; Betsy Greenleaf, DO, MBA; Elizabeth DuPriest, PhD
References
- Dezzutti CS, Brown ER, Moncla B, et al. Is wetter better? An evaluation of over-the-counter personal lubricants for safety and anti-HIV-1 activity. PLoS One. 2012;7:e48328. doi: 10.1371/journal .pone.0048328.
- Ayehunie S, Wang YY, Landry T, et al. Hyperosmolal vaginal lubricants markedly reduce epithelial barrier properties in a threedimensional vaginal epithelium model. Toxicol Rep. 2017;5:134-140. doi: 10.1016 /j.toxrep.2017.12.011.
- Moench TR, Mumper RJ, Hoen TE, et al. Microbicide excipients can greatly increase susceptibility to genital herpes transmission in the mouse. BMC Infect Dis. 2010;10:331. doi: 10.1186/1471 -2334-10-331.
- Use and procurement of additional lubricants for male and female condoms: WHO/UNFPA /FHI360 Advisory note. World Health Organization, 2012. http://apps.who.int/iris/bitstream /handle/10665/76580/WHO_RHR_12.33_eng .pdf?sequence=1. Accessed December 27, 2021.
Drs. Pennycuff And Iglesia Respond
We thank the members of the scientific advisory board for the Sexual Health and Wellness Institute for their thoughtful and insightful comments to our article. We agree with their comments on the importance of both pH and osmolality for vaginal moisturizers and lubricants. We also agree that selection of an incorrectly formulated product may lead to worsening of vulvovaginal symptoms as well as dysbiosis and all of its sequelae as the letter writers mentioned.
In writing the review article, we attempted to address the role that pH and osmolality play in vaginal moisturizers and lubricants and make clinicians more aware of the importance of these factors in product formulation. Our goal was to help to improve patient counseling. We tried to amass as much of the available literature as we could to act as a resource for practitioners, such as the table included in the article as well as the supplemental table included online. We hoped that by writing this article we would heighten awareness among female health practitioners about vaginal health products and encourage them to consider those products that may be better suited for their patients based on pH and osmolality.
While there remains a paucity of research on vaginal moisturizers and lubricants, there is even less consumer knowledge regarding ingredients and formulations of these products. We wholeheartedly agree with the scientific advisory board that we as health providers need to help educate women on the full spectrum of products available beyond the “leading brands.” Furthermore, we advocate that there be continued research on these products as well as more manufacturer transparency regarding not only the ingredients contained within these products but also the pH and osmolality. Simple steps such as these would ensure that providers could help counsel patients to make informed decisions regarding products for their pelvic health.
Continue to: DISMANTLING RACISM IN YOUR PERSONAL AND PROFESSIONAL SPHERES...
DISMANTLING RACISM IN YOUR PERSONAL AND PROFESSIONAL SPHERES
CASSANDRA CARBERRY, MD, MS; ANNETTA MADSEN, MD; OLIVIA CARDENAS-TROWERS, MD; OLUWATENIOLA BROWN, MD; MOIURI SIDDIQUE, MD; AND BLAIR WASHINGTON, MD, MHA (AUGUST 2021)
Dissenting opinion
“Race is real but it’s not biologic.” “Race is not based on genetic or biologic inheritance.” Am I the only one with a dissenting voice of opinion when it comes to these types of statements?
Scott Peters, MD
Oak Ridge, Tennessee
The Authors Respond
Thank you for your opinion, Dr. Peters. Although it is not completely clear what your question is, it seems that it concerns the validity of the idea that race is a social construct. We will address this question with the assumption that this letter was an effort to invite discussion and increase understanding.
The National Human Genome Research Institute describes race in this way: “Race is a fluid concept used to group people according to various factors, including ancestral background and social identity. Race is also used to group people that share a set of visible characteristics, such as skin color and facial features. Though these visible traits are influenced by genes, the vast majority of genetic variation exists within racial groups and not between them.”1
The understanding that race is a social construct has been upheld by numerous medical organizations. In August 2020, a Joint Statement was published by the American College of Obstetricians and Gynecologists, the American Board of Obstetricians and Gynecologists, and 22 other organizations representing our specialty. This document states: “Recognizing that race is a social construct, not biologically based, is important to understanding that racism, not race, impacts health care, health, and health outcomes.”2
This idea is also endorsed by the AMA, who in November 2020 adopted the following policies3:
- “Recognize that race is a social construct and is distinct from ethnicity, genetic ancestry, or biology
- Support ending the practice of using race as a proxy for biology or genetics in medical education, research, and clinical practice.”
There are numerous sources that further illuminate why race is a social construct. Here are a few:
- https://www.racepowerofanillusion .org/resources/
- https ://www.pewresearch.org /fact-tank/2020/02/25/the-changing -categories-the-u-s-has-used-to -measure-race/
- Roberts D. Fatal Invention: How Science, Politics and Big Business Re-create Race in the Twenty-First Century. The New Press. 2011.
- Yudell M, Roberts D, DeSalle R, et al. Science and society. Taking race out of human genetics. Science. 2016;351(6273):564-5. doi: 10.1126/science.aac4951.
References
- National Human Genome Research Institute. Race. https://www.genome.gov/genetic-glossary /Race. Accessed December 27, 2021.
- The American College of Obstetricians and Gynecologists. Joint Statement: Collective Action Addressing Racism. https://www.acog.org /news/news-articles/2020/08/joint-statementobstetrics-and-gynecology-collective-actionaddressing-racism.
- O’Reilly KB. AMA: Racism is a threat to public health. November 16, 2020. https://www.ama -assn.org/delivering-care/health-equity/ama -racism-threat-public-health. Accessed December 27, 2021.
HOW TO CHOOSE THE RIGHT VAGINAL MOISTURIZER OR LUBRICANT FOR YOUR PATIENT
JOHN PENNYCUFF, MD, MSPH, AND CHERYL IGLESIA, MD (JUNE 2021)
Which vaginal products to recommend
We applaud Drs. Pennycuff and Iglesia for providing education on lubricants and vaginal moisturizers in their recent article, and agree that ObGyns, urogynecologists, and primary care providers should be aware of the types of products available. However, the authors underplayed the health risks associated with the use of poor-quality lubricants and moisturizers.
Women often turn to lubricants or vaginal moisturizers because they experience vaginal dryness during intercourse, related to menopause, and from certain medications. Vaginal fluid is primarily composed of exudate from capillaries in the vaginal wall. During sexual arousal, blood flow to the vaginal wall increases, and in turn, this should increase exudate. But chronic inflammation can suppress these increases in vaginal blood flow, preventing adequate vaginal fluid production. One such cause of chronic inflammation is using hyperosmolar lubricants, as this has been shown to negatively affect the vaginal epithelium.1,2 In this way, use of hyperosmolar lubricants can actually worsen symptoms, creating a vicious circle of dryness, lubricant use, and worsening dryness.
In addition, hyperosmolar lubricants have been shown to reduce the epithelial barrier properties of the vaginal epithelium, increasing susceptibility to microbes associated with bacterial vaginosis and to true pathogens, including herpes simplex virus type 2.3 In fact, hyperosmolar lubricants are a serious enough problem that the World Health Organization has weighed in, recommending osmolality of personal lubricants be under 380 mOsm/kg to prevent damage to the vaginal epithelium.4
Appropriately acidic pH is just as critical as osmolality. Using products with a pH higher than 4.5 will reduce amounts of protective lactobacilli and other commensal vaginal bacteria, encouraging growth of opportunistic bacteria and yeast already present. This can lead to bacterial vaginosis, aerobic vaginitis, and candidiasis. Bacterial vaginosis can lead to other serious sequelae such as increased risk in acquisition of HIV infection and preterm birth in pregnancy. Unfortunately, much of the data cited in Drs. Pennycuff and Iglesia’s article were sourced from another study (by Edwards and Panay published in Climacteric in 2016), which measured product pH values with an inappropriately calibrated device; the study’s supplemental information stated that calibration was between 5 and 9, and so any measurement below 5 was invalid and subject to error. For example, the Good Clean Love lubricant is listed as having a pH of 4.7, but its pH is never higher than 4.4.
The products on the market that meet the dual criteria of appropriate pH and isotonicity to vaginal epithelial cells may be less well known to consumers. But this should not be a reason to encourage use of hyperosmolar products whose main selling point is that they are the “leading brand.” Educating women on their choices in personal lubricants should include a full discussion of product ingredients and properties, based upon the available literature to help them select a product that supports the health of their intimate tissues.
Members of the Scientific Advisory Board for the Sexual Health and Wellness Institute: Jill Krapf, MD, MEd, IF; Cathy Chung Hwa Yi, MD; Christine Enzmann, MD, PhD, NMCP; Susan Kellogg-Spadt, PhD, CRNP, IF, CSC, FCST; Betsy Greenleaf, DO, MBA; Elizabeth DuPriest, PhD
References
- Dezzutti CS, Brown ER, Moncla B, et al. Is wetter better? An evaluation of over-the-counter personal lubricants for safety and anti-HIV-1 activity. PLoS One. 2012;7:e48328. doi: 10.1371/journal .pone.0048328.
- Ayehunie S, Wang YY, Landry T, et al. Hyperosmolal vaginal lubricants markedly reduce epithelial barrier properties in a threedimensional vaginal epithelium model. Toxicol Rep. 2017;5:134-140. doi: 10.1016 /j.toxrep.2017.12.011.
- Moench TR, Mumper RJ, Hoen TE, et al. Microbicide excipients can greatly increase susceptibility to genital herpes transmission in the mouse. BMC Infect Dis. 2010;10:331. doi: 10.1186/1471 -2334-10-331.
- Use and procurement of additional lubricants for male and female condoms: WHO/UNFPA /FHI360 Advisory note. World Health Organization, 2012. http://apps.who.int/iris/bitstream /handle/10665/76580/WHO_RHR_12.33_eng .pdf?sequence=1. Accessed December 27, 2021.
Drs. Pennycuff And Iglesia Respond
We thank the members of the scientific advisory board for the Sexual Health and Wellness Institute for their thoughtful and insightful comments to our article. We agree with their comments on the importance of both pH and osmolality for vaginal moisturizers and lubricants. We also agree that selection of an incorrectly formulated product may lead to worsening of vulvovaginal symptoms as well as dysbiosis and all of its sequelae as the letter writers mentioned.
In writing the review article, we attempted to address the role that pH and osmolality play in vaginal moisturizers and lubricants and make clinicians more aware of the importance of these factors in product formulation. Our goal was to help to improve patient counseling. We tried to amass as much of the available literature as we could to act as a resource for practitioners, such as the table included in the article as well as the supplemental table included online. We hoped that by writing this article we would heighten awareness among female health practitioners about vaginal health products and encourage them to consider those products that may be better suited for their patients based on pH and osmolality.
While there remains a paucity of research on vaginal moisturizers and lubricants, there is even less consumer knowledge regarding ingredients and formulations of these products. We wholeheartedly agree with the scientific advisory board that we as health providers need to help educate women on the full spectrum of products available beyond the “leading brands.” Furthermore, we advocate that there be continued research on these products as well as more manufacturer transparency regarding not only the ingredients contained within these products but also the pH and osmolality. Simple steps such as these would ensure that providers could help counsel patients to make informed decisions regarding products for their pelvic health.
Continue to: DISMANTLING RACISM IN YOUR PERSONAL AND PROFESSIONAL SPHERES...
DISMANTLING RACISM IN YOUR PERSONAL AND PROFESSIONAL SPHERES
CASSANDRA CARBERRY, MD, MS; ANNETTA MADSEN, MD; OLIVIA CARDENAS-TROWERS, MD; OLUWATENIOLA BROWN, MD; MOIURI SIDDIQUE, MD; AND BLAIR WASHINGTON, MD, MHA (AUGUST 2021)
Dissenting opinion
“Race is real but it’s not biologic.” “Race is not based on genetic or biologic inheritance.” Am I the only one with a dissenting voice of opinion when it comes to these types of statements?
Scott Peters, MD
Oak Ridge, Tennessee
The Authors Respond
Thank you for your opinion, Dr. Peters. Although it is not completely clear what your question is, it seems that it concerns the validity of the idea that race is a social construct. We will address this question with the assumption that this letter was an effort to invite discussion and increase understanding.
The National Human Genome Research Institute describes race in this way: “Race is a fluid concept used to group people according to various factors, including ancestral background and social identity. Race is also used to group people that share a set of visible characteristics, such as skin color and facial features. Though these visible traits are influenced by genes, the vast majority of genetic variation exists within racial groups and not between them.”1
The understanding that race is a social construct has been upheld by numerous medical organizations. In August 2020, a Joint Statement was published by the American College of Obstetricians and Gynecologists, the American Board of Obstetricians and Gynecologists, and 22 other organizations representing our specialty. This document states: “Recognizing that race is a social construct, not biologically based, is important to understanding that racism, not race, impacts health care, health, and health outcomes.”2
This idea is also endorsed by the AMA, who in November 2020 adopted the following policies3:
- “Recognize that race is a social construct and is distinct from ethnicity, genetic ancestry, or biology
- Support ending the practice of using race as a proxy for biology or genetics in medical education, research, and clinical practice.”
There are numerous sources that further illuminate why race is a social construct. Here are a few:
- https://www.racepowerofanillusion .org/resources/
- https ://www.pewresearch.org /fact-tank/2020/02/25/the-changing -categories-the-u-s-has-used-to -measure-race/
- Roberts D. Fatal Invention: How Science, Politics and Big Business Re-create Race in the Twenty-First Century. The New Press. 2011.
- Yudell M, Roberts D, DeSalle R, et al. Science and society. Taking race out of human genetics. Science. 2016;351(6273):564-5. doi: 10.1126/science.aac4951.
References
- National Human Genome Research Institute. Race. https://www.genome.gov/genetic-glossary /Race. Accessed December 27, 2021.
- The American College of Obstetricians and Gynecologists. Joint Statement: Collective Action Addressing Racism. https://www.acog.org /news/news-articles/2020/08/joint-statementobstetrics-and-gynecology-collective-actionaddressing-racism.
- O’Reilly KB. AMA: Racism is a threat to public health. November 16, 2020. https://www.ama -assn.org/delivering-care/health-equity/ama -racism-threat-public-health. Accessed December 27, 2021.
How to identify balance disorders and reduce fall risk
CASE Mr. J, a 75-year-old man, presents to your family practice reporting that he feels increasingly unsteady and slow while walking. He fell twice last year, without resulting injury. He now worries about tripping while walking around the house and relies on his spouse to run errands.
Clearly, Mr. J is experiencing a problem with balance. What management approach should you undertake to prevent him from falling?

Balance disorders are common in older people and drastically hinder quality of life.1-4 Patients often describe imbalance as vague symptoms: dizziness, unsteadiness, faintness, spinning sensations.5,6 Importantly, balance disorders disrupt normal gait and contribute to falls that are a major cause of disability and morbidity in older people. Almost 30% of people older than 65 years report 1 or more falls annually.7 Factors that increase the risk of falls include impaired mobility, previously reported falls, reduced psychological functioning, chronic medical conditions, and polypharmacy.7,8
The cause of any single case of imbalance is often multifactorial, resulting from dysfunction of multiple body systems (TABLE 17-56); in our clinical experience, most patients with imbalance and who are at risk of falls do not have a detectable deficit of the vestibular system. These alterations in function arise in 3 key systems—vision, proprioception, and vestibular function—which signal to, and are incorporated by, the cerebellum to mediate balance. Cognitive and neurologic decline are also factors in imbalance.
Considering that 20% of falls result in serious injury in older populations, it is important to identify balance disorders and implement preventive strategies to mitigate harmful consequences of falls on patients’ health and independence.7,57 In this article, we answer the question that the case presentation raises about the proper management approach to imbalance in family practice, including assessment of risk and rehabilitation strategies to reduce the risk of falls. Our insights and recommendations are based on our clinical experience and a review of the medical literature from the past 40 years.
CASE Mr. J has a history of hypertension, age-related hearing loss, and osteoarthritis of the knees; he has not had surgery for the arthritis. His medications are antihypertensives and extra-strength acetaminophen for knee pain.

Making the diagnosis of a balance disorder
History
A thorough clinical history, often including a collateral history from caregivers, narrows the differential diagnosis. Information regarding onset, duration, timing, character, and previous episodes of imbalance is essential. Symptoms of imbalance are often challenging for the patient to describe: They might use terms such as vertigo or dizziness, when, in fact, on further questioning, they are describing balance difficulties. Inquiry into (1) their use of assistive walking devices and (2) development or exacerbation of neurologic, musculoskeletal, auditory, visual, and mood symptoms is necessary. Note the current level of their mobility, episodes of pain or fatigue, previous falls and associated injuries, fear of falling, balance confidence, and sensations that precede falls.58
Continue to: The medical and surgical histories
The medical and surgical histories are key pieces of information. The history of smoking, alcohol habits, and substance use is relevant.
A robust medication history is essential to evaluate a patient’s risk of falling. Polypharmacy—typically, defined as taking 4 or more medications—has been repeatedly associated with a heightened risk of falls.53,59-61 Moreover, a dose-dependent association between polypharmacy and hospitalization following falls has been identified, and demonstrates that taking 10 or more medications greatly increases the risk of hospitalization.59 Studies of polypharmacy cement the importance of inquiring about medication use when assessing imbalance, particularly in older patients.
Physical examination
A focused and detailed physical examination provides insight into systems that should be investigated:
- Obtain vital signs, including orthostatic vitals to test for orthostatic hypotension62; keep in mind that symptoms of orthostatic dizziness can occur without orthostatic hypotension.
- Examine gait, which can distinguish between causes of imbalance (TABLE 2).21,40,63-70
- Perform a cardiac examination.
- Assess visual acuity and visual fields; test for nystagmus and identify any optic-nerve and retinal abnormalities.
- Evaluate lower-limb sensation, proprioception, and motor function.
- Evaluate suspected vestibular dysfunction, including dysfunction with positional testing (the Dix-Hallpike maneuver71). The patient is taken from sitting to supine while the head is rotated 45° to the tested side by the examiner. As the patient moves into a supine position, the neck is extended 30° off the table and held for at least 30 seconds. The maneuver is positive if torsional nystagmus is noted while the head is held rotated during neck extension. The maneuver is negative if the patient reports dizziness, vertigo, unsteadiness, or “pressure in the head.” Torsional nystagmus must be present to confirm a diagnosis of benign paroxysmal positional vertigo.
- If you suspect a central nervous system cause of imbalance, assess the cranial nerves, coordination, strength, and, of course, balance.
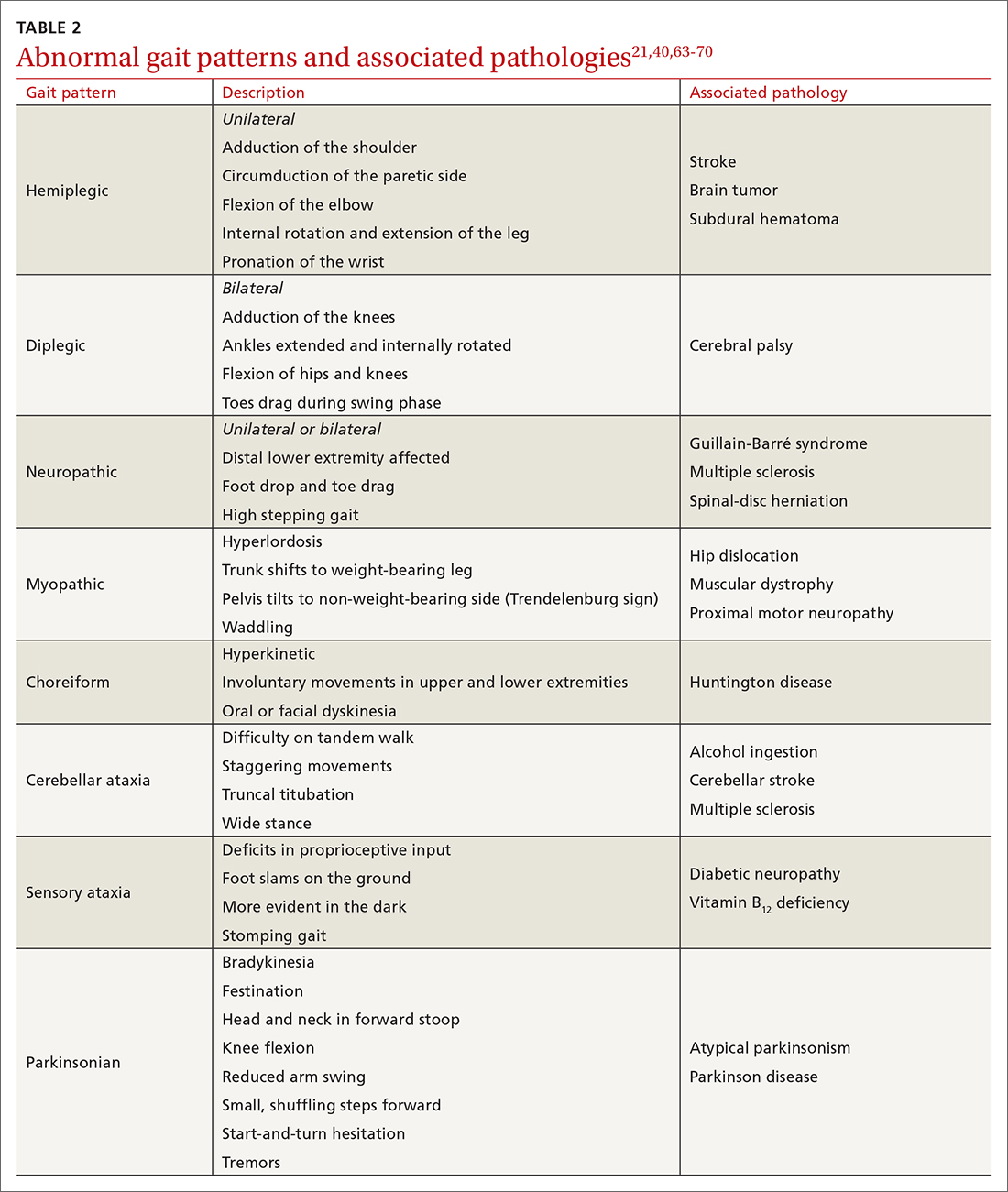
CASE
Mr. J’s physical examination showed normal vital signs without significant postural changes in blood pressure. Gait analysis revealed a slowed gait, with reduced range of motion in both knees over the entire gait cycle. Audiometry revealed symmetric moderate sensorineural hearing loss characteristic of presbycusis.
Diagnostic investigations
Consider focused investigations into imbalance based on the history and physical examination. We discourage overly broad testing and imaging; in primary care, cost and limited access to technology can bar robust investigations into causes of imbalance. However, identification of acute pathologies should prompt immediate referral to the emergency department. Furthermore, specific symptoms (TABLE 17-56) should prompt referral to specialists for assessment.
Continue to: In the emergency department...
In the emergency department and academic hospitals, key investigations can identify causes of imbalance:
- Electrocardiography and Holter monitoring test for cardiac arrhythmias.
- Echocardiography identifies structural abnormalities.
- Radiography and computed tomography are useful for detecting musculoskeletal abnormalities.
- Bone densitometry can identify osteoporosis.
- Head and spinal cord magnetic resonance imaging can be used to identify lesions of the central nervous system.
- Computed tomographic angiography of the head and neck is useful for identifying stroke, cerebral atrophy, and stenotic lesions of the carotid and vertebral arteries.
- Nerve conduction studies and levels of serum vitamin B12, hemoglobin A1C, thyroid-stimulating hormone, and random cortisol can uncover causes of peripheral neuropathy.
- Bedside cognitive screening tests can be used to measure cognitive decline.72
- Suspicion of vestibular disease requires audiometry and vestibular testing, including videonystagmography, head impulse testing, and vestibular evoked myogenic potentials.
In many cases of imbalance, no specific underlying correctable cause is discovered.
Management of imbalance
Pharmacotherapy
Targeted pharmacotherapy can be utilized in select clinical scenarios:
- Medical treatment of peripheral neuropathy should target the underlying condition.
- Cognitive behavioral therapy and antidepressants are useful for treating anxiety and depressive disorders.73
- Musculoskeletal pain can be managed with acetaminophen and topical nonsteroidal anti-inflammatory drugs (NSAIDs), using a short course of an oral NSAID when needed.74
- Cardiovascular disease management might include any of several classes of pharmacotherapy, including antiplatelet and lipid-lowering medications, antiarrhythmic drugs, and antihypertensive agents.
- Acute episodes of vertigo due to vestibular neuritis or labyrinthitis can be managed with an antiemetic.46
Surgical treatment
Surgery is infrequently considered for patients with imbalance. Examples of indications include microsurgical resection of vestibular schwannoma, resection of central nervous system tumors, lens replacement surgery for cataract, surgical management of severe spinal fracture, and hip or knee arthroplasty in select patients.
Tools for assessing the risk of falls
Scoring systems called falls risk assessment tools, or FRAT, have been developed to gauge a patient’s risk of falling. The various FRATs differ in specificity and sensitivity for predicting the risk of falls, and are typically designed for specific clinical environments, such as hospital inpatient care or long-term care facilities. Specifically, FRATs attempt to classify risk using sets of risk factors known to be associated with falls.
Continue to: Research abounds into...
Research abounds into the validity of commonly used FRATs across institutions, patient populations, and clinical environments:
The Johns Hopkins FRATa determines risk using metrics such as age, fall history, incontinence, cognition, mobility, and medications75; it is predominantly used for assessment in hospital inpatient units. This tool has been validated repeatedly.76,77
Peninsula Health FRATb stratifies patients in subacute and residential aged-care settings, based on risk factors that include recent falls, medications, psychological status, and cognition.78
FRAT-upc is a web-based tool that generates falls risk using risk factors that users input. This tool has been studied in the context of patients older than 65 years living in the community.79
Although FRATs are reasonably useful for predicting falls, their utility varies by patient population and clinical context. Moreover, it has been suggested that FRATs neglect environmental and personal factors when assessing risk by focusing primarily on bodily factors.80 Implementing a FRAT requires extensive consideration of the target population and should be accompanied by clinical judgment that is grounded in an individual patient’s circumstances.81
Continue to: Preventing falls in primary care
Preventing falls in primary care
An approach to preventing falls includes the development of individualized programs that account for frailty, a syndrome of physiologic decline associated with aging. Because frailty leads to diminished balance and mobility, a patient’s frailty index—determined using the 5 frailty phenotype criteria (exhaustion, weight loss, low physical activity, weakness, slowness)82 or the Canadian Study of Health and Aging Clinical Frailty Scale83—is a useful tool for predicting falls risk and readmission for falls following trauma-related injury. Prevention of falls in communities is critical for reducing mortality and allowing older people to maintain their independence and quality of life.
Exercise. In some areas, exercise and falls prevention programs are accessible to seniors.84 Community exercise programs that focus on balance retraining and muscle strengthening can reduce the risk of falls.73,85 The Choosing Wisely initiative of the ABIM [American Board of Internal Medicine] Foundation recommends that exercise programs be designed around an accurate functional baseline of the patient to avoid underdosed strength training.54
Multifactorial risk assessment in high-risk patients can reduce the rate of falls. Such an assessment includes examination of orthostatic blood pressure, vision and hearing, bone health, gait, activities of daily living, cognition, and environmental hazards, and enables provision of necessary interventions.73,86 Hearing amplification, specifically, correlates with enhanced postural control, slowed cognitive decline, and a reduced likelihood of falls.87-93 The mechanism behind improved balance performance might be reduced cognitive load through supporting a patient’s listening needs.88-90
Pharmacotherapy. Optimizing medications and performing a complete medication review before prescribing new medications is highly recommended to avoid unnecessary polypharmacy7,8,18,53-56 (TABLE 17-56).
Management of comorbidities associated with a higher risk of falls, including arthritis, cancer, stroke, diabetes, depression, kidney disease, chronic obstructive pulmonary disease, cognitive impairment, hypertension, and atrial fibrillation, is essential.94-96
Continue to: Home safety interventions
Home safety interventions, through occupational therapy, are important. These include removing unsafe mats and step-overs and installing nonslip strips on stairs, double-sided tape under mats, and handrails.73-97
Screening for risk of falls. The Centers for Disease Control and Prevention recommends that (1) all patients older than 65 years and (2) any patient presenting with an acute fall undergo screening for their risk of falls.98 When a patient is identified as at risk of falling, you can, when appropriate, assess modifiable risk factors and facilitate interventions.98 This strategy is supported by a 2018 statement from the US Preventive Services Task Force99 that recommends identifying high-risk patients who have:
- a history of falling
- a balance disturbance that causes a deficit of mobility or function
- poor performance on clinical tests, such as the 3-meter Timed Up and Go (TUG) assessment (www.cdc.gov/steadi/pdf/TUG_test-print.pdf).
An increased risk of falls should prompt you to refer the patient to community programs and physiotherapy in accordance with the individual’s personal goals99; a balance and vestibular physiotherapist is ideally positioned to accurately assess and manage patients at risk of falls. Specifically, the Task Force identified exercise programs and multifactorial interventions as being beneficial in preventing falls in high-risk older people.99
Balance assessment and rehabilitation in specialty centers
An individualized rehabilitation program aims to restore safe mobility by testing and addressing specific balance deficits, improving functional balance, and increasing balance confidence. Collaboration with colleagues from physiotherapy and occupational therapy aids in tailoring individualized programs.
Many tests are available to assess balance, determine the risk of falls, and guide rehabilitation:
- The timed 10-meter walk testd and the TUG test are simple assessments that measure functional mobility; both have normalized values for the risk of falls. A TUG time of ≥ 12 seconds suggests a high risk of falls.
- The 30-second chair stande evaluates functional lower-extremity strength in older patients. The test can indicate if lower-extremity strength is contributing to a patient’s imbalance.
- The modified clinical test of sensory interaction in balancef is a static balance test that measures the integrity of sensory inputs. The test can suggest if 1 or more sensory systems are compromised.
- The mini balance evaluation systems testg is similar: It can differentiate balance deficits by underlying system and allows individualization of a rehabilitation program.
- The functional gait assessmenth is a modification of the dynamic gait index that assesses postural stability during everyday dynamic activities, including tasks such as walking with head turns and pivots.
- The Berg Balance Scalei continues to be used extensively to assess balance.
Continue to: The mini balance evaluation systems test...
The mini balance evaluation systems test, functional gait index, and Berg Balance Scale all have normative age-graded values to predict fall risk.
CASE
Mr. J was referred for balance assessment and to a rehabilitation program. He underwent balance physiotherapy, including multifactorial balance assessment, joined a community exercise program, was fitted with hearing aids, and had his home environment optimized by an occupational therapist. (See examples of “home safety interventions” under “Preventing falls in primary care.”)
3 months later. Mr. J says he feels stronger on his feet. His knee pain has eased, and he is more confident walking around his home. He continues to engage in exercise programs and is comfortable running errands with his spouse.
CORRESPONDENCE
Jason A. Beyea, MD, PhD, FRCSC, Division of OtolaryngologyHead and Neck Surgery, Queen’s University, 144 Brock Street, Kingston, Ontario, Canada, K7L 5G2; [email protected]
a www.hopkinsmedicine.org/institute_nursing/models_tools/jhfrat_acute%20care%20original_6_22_17.pdf
c www.ncbi.nlm.nih.gov/pmc/articles/PMC4376110/figure/figure14/?report=objectonly
e www.cdc.gov/steadi/pdf/STEADI-Assessment-30Sec-508.pdf
f www.mdapp.co/mctsib-modified-clinical-test-of-sensory-interaction-in-balance-calculator-404/
g www.sralab.org/sites/default/files/2017-07/MiniBEST_revised_final_3_8_13.pdf
1. Larocca NG. Impact of walking impairment in multiple sclerosis: perspectives of patients and care partners. Patient. 2011;4:189-201. doi: 10.2165/11591150-000000000-00000
2. TB, ZF, ES, et al. The relationship of balance disorders with falling, the effect of health problems, and social life on postural balance in the elderly living in a district in Turkey. Geriatrics (Basel). 2019;4:37. doi: 10.3390/geriatrics4020037
3. R, Sixt E, Landahl S, et al. Prevalence of dizziness and vertigo in an urban elderly population. J Vestib Res. 2004;14:47-52.
4. Sturnieks DL, St George R, Lord SR. Balance disorders in the elderly. Neurophysiol Clin. 2008;38:467-478. doi: 10.1016/j.neucli.2008.09.001
5. Boult C, Murphy J, Sloane P, et al. The relation of dizziness to functional decline. J Am Geriatr Soc. 1991;39:858-861. doi: 10.1111/j.1532-5415.1991.tb04451.x
6. Lin HW, Bhattacharyya N. Balance disorders in the elderly: epidemiology and functional impact. Laryngoscope. 2012;122:1858-1861. doi: 10.1002/lary.23376
7. Jia H, Lubetkin EI, DeMichele K, et al. Prevalence, risk factors, and burden of disease for falls and balance or walking problems among older adults in the U.S. Prev Med. 2019;126:105737. doi: 10.1016/j.ypmed.2019.05.025
8. Al-Momani M, Al-Momani F, Alghadir AH, et al. Factors related to gait and balance deficits in older adults. Clin Interv Aging. 2016;11:1043-1049. doi: 10.2147/CIA.S112282
9. Agrawal Y, Ward BK, Minor LB. Vestibular dysfunction: prevalence, impact and need for targeted treatment. J Vestib Res. 2013;23:113-117. doi: 10.3233/VES-130498
10. Altinsoy B, Erboy F, Tanriverdi H, et al. Syncope as a presentation of acute pulmonary embolism. Ther Clin Risk Manag. 2016;12:1023-1028. doi: 10.2147/TCRM.S105722
11. Belvederi Murri M, Triolo F, Coni A, et al. Instrumental assessment of balance and gait in depression: a systematic review. Psychiatry Res. 2020;284:112687. doi: 10.1016/j.psychres.2019.112687
12. Bhattacharyya N, Gubbels SP, Schwartz SR, et al. Clinical practice guideline: benign paroxysmal positional vertigo (update). Otolaryngol Head Neck Surg. 2017;156(suppl 3):S1-S47. doi: 10.1177/0194599816689667
13. S, Schwarm S, Grevenrath P, et al. Prevalence, aetiologies and prognosis of the symptom dizziness in primary care - a systematic review. BMC Fam Pract. 2018;19:33. doi: 10.1186/s12875-017-0695-0
14. Brouwer MC, Tunkel AR, van de Beek D. Epidemiology, diagnosis, and antimicrobial treatment of acute bacterial meningitis. Clin Microbiol Rev. 2010;23:467-492. doi: 10.1128/CMR.00070-09
15. Chad DA. Lumbar spinal stenosis. Neurol Clin. 2007;25:407-418. doi: 10.1016/j.ncl.2007.01.003
16. Conrad BP, Shokat MS, Abbasi AZ, et al. Associations of self-report measures with gait, range of motion and proprioception in patients with lumbar spinal stenosis. Gait Posture. 2013;38:987-992. doi: 10.1016/j.gaitpost.2013.05.010
17. de Luna RA, Mihailovic A, Nguyen AM, et al. The association of glaucomatous visual field loss and balance. Transl Vis Sci Technol. 2017;6:8. doi: 10.1167/tvst.6.3.8
18. DiSogra RM. Common aminoglycosides and platinum-based ototoxic drugs: cochlear/vestibular side effects and incidence. Semin Hear. 2019;40:104-107. doi: 10.1055/s-0039-1684040
19. Ebersbach G, Moreau C, Gandor F, et al. Clinical syndromes: parkinsonian gait. Mov Disord. 2013;28:1552-1559. doi: 10.1002/mds.25675
20. Evans WJ. Skeletal muscle loss: cachexia, sarcopenia, and inactivity. Am J Clin Nutr. 2010;91:1123S-1127S. doi: 10.3945/ajcn.2010.28608A
21. Filli L, Sutter T, Easthope CS, et al. Profiling walking dysfunction in multiple sclerosis: characterisation, classification and progression over time. Sci Rep. 2018;8:4984. doi: 10.1038/s41598-018-22676-0
22. Fritz NE, Kegelmeyer DA, Kloos AD, et al. Motor performance differentiates individuals with Lewy body dementia, Parkinson’s and Alzheimer’s disease. Gait Posture. 2016;50:1-7. doi: 10.1016/j.gaitpost.2016.08.009
23. Furman JM, Jacob RG. A clinical taxonomy of dizziness and anxiety in the otoneurological setting. J Anxiety Disord. 2001;15:9-26. doi: 10.1016/s0887-6185(00)00040-2
24. Furman JM, Marcus DA, Balaban CD. Vestibular migraine: clinical aspects and pathophysiology. Lancet Neurol. 2013;12:706-715. doi: 10.1016/S1474-4422(13)70107-8
25. Gerson LW, Jarjoura D, McCord G. Risk of imbalance in elderly people with impaired hearing or vision. Age Ageing. 1989;18:31-34. doi: 10.1093/ageing/18.1.31
26. Goudakos JK, Markou KD, Franco-Vidal V, et al. Corticosteroids in the treatment of vestibular neuritis: a systematic review and meta-analysis. Otol Neurotol. 2010;31:183-189. doi: 10.1097/MAO.0b013e3181ca843d
27. Green AD, CS, Bastian L, et al. Does this woman have osteoporosis? JAMA. 2004;292:2890-2900. doi: 10.1001/jama.292.23.2890
28. Hallemans A, Ortibus E, Meire F, et al. Low vision affects dynamic stability of gait. Gait Posture. 2010;32:547-551. doi: 10.1016/j.gaitpost.2010.07.018
29. Handelsman JA. Vestibulotoxicity: strategies for clinical diagnosis and rehabilitation. Int J Audiol. 2018;57(suppl 4):S99-S107. doi: 10.1080/14992027.2018.1468092
30. Head VA, Wakerley BR. Guillain-Barré syndrome in general practice: clinical features suggestive of early diagnosis. Br J Gen Pract. 2016;66:218-219. doi: 10.3399/bjgp16X684733
31. Helbostad JL, Vereijken B, Hesseberg K, et al. Altered vision destabilizes gait in older persons. Gait Posture. 2009;30:233-238. doi: 10.1016/j.gaitpost.2009.05.004
32. Hsu W-L, Chen C-Y, Tsauo J-Y, et al. Balance control in elderly people with osteoporosis. J Formos Med Assoc. 2014;113:334-339. doi: 10.1016/j.jfma.2014.02.006
33. Kim H-S, Yun DH, Yoo SD, et al. Balance control and knee osteoarthritis severity. Ann Rehabil Med. 2011;35:701-709. doi: 10.5535/arm.2011.35.5.701
34. Li L, Simonsick EM, Ferrucci L, et al. Hearing loss and gait speed among older adults in the United States. Gait Posture. 2013;38:25-29.
35. McKeith IG, Boeve BF, Dickson DW, et al. Diagnosis and management of dementia with Lewy bodies: fourth consensus report of the DLB Consortium. Neurology. 2017;89:88-100. doi: 10.1212/WNL.0000000000004058
36. Milner KA, Funk M, Richards S, et al. Gender differences in symptom presentation associated with coronary heart disease. Am J Cardiol. 1999;84:396-399. doi: 10.1016/s0002-9149(99)00322-7
37. Paillard T, F, Bru N, et al. The impact of time of day on the gait and balance control of Alzheimer’s patients. Chronobiol Int. 2016;33:161-168. doi: 10.3109/07420528.2015.1124885
38. Paldor I, Chen AS, Kaye AH. Growth rate of vestibular schwannoma. J Clin Neurosci. 2016;32:1-8. doi: 10.1016/j.jocn.2016.05.003
39. Picorelli AMA, Hatton AL, Gane EM, et al. Balance performance in older adults with hip osteoarthritis: a systematic review. Gait Posture. 2018;65:89-99. doi: 10.1016/j.gaitpost.2018.07.001
40. Raccagni C, Nonnekes J, Bloem BR, et al. Gait and postural disorders in parkinsonism: a clinical approach. J Neurol. 2020;267:3169-3176. doi: 10.1007/s00415-019-09382-1
41. Shanmugarajah PD, Hoggard N, Currie S, et al. Alcohol-related cerebellar degeneration: not all down to toxicity? Cerebellum Ataxias. 2016;3:17. doi: 10.1186/s40673-016-0055-1
42. Shih RY, Smirniotopoulos JG. Posterior fossa tumors in adult patients. Neuroimaging Clin N Am. 2016;26:493-510. doi: 10.1016/j.nic.2016.06.003
43. Smith EE. Clinical presentations and epidemiology of vascular dementia. Clin Sci (Lond). 2017;131:1059-1068. doi: 10.1042/CS20160607
44. Streur M, Ratcliffe SJ, Ball J, et al. Symptom clusters in adults with chronic atrial fibrillation. J Cardiovasc Nurs. 2017;32:296-303. doi: 10.1097/JCN.0000000000000344
45. Strupp M, M, JA. Peripheral vestibular disorders: an update. Curr Opin Neurol. 2019;32:165-173. doi: 10.1097/WCO.0000000000000649
46. Thompson TL, Amedee R. Vertigo: a review of common peripheral and central vestibular disorders. Ochsner J. 2009;9:20-26.
47. Timar B, Timar R, L, et al. The impact of diabetic neuropathy on balance and on the risk of falls in patients with type 2 diabetes mellitus: a cross-sectional study. PLoS One. 2016;11:e0154654. doi: 10.1371/journal.pone.0154654
48. Walls R, Hockberger R, Gausche-Hill M. Peripheral nerve disorders. In: Rosen’s Emergency Medicine: Concepts and Clinical Practice. 9th ed. Elsevier, Inc; 2018:1307-1320.
49. Watson JC, Dyck PJB. Peripheral neuropathy: a practical approach to diagnosis and symptom management. Mayo Clin Proc. 2015;90:940-951. doi: 10.1016/j.mayocp.2015.05.004
50. Whitfield KC, Bourassa MW, Adamolekun B, et al. Thiamine deficiency disorders: diagnosis, prevalence, and a roadmap for global control programs. Ann N Y Acad Sci. 2018;1430:3-43. doi: 10.1111/nyas.13919
51. Wu V, Sykes EA, Beyea MM, et al. Approach to Meniere disease management. Can Fam Physician. 2019;65:463-467.
52. Yew KS, Cheng EM. Diagnosis of acute stroke. Am Fam Physician. 2015;91:528-536.
53. Seppala LJ, van de Glind EMM, Daams JG, et al; . Fall-risk-increasing drugs: a systematic review and meta-analysis: III. Others. J Am Med Dir Assoc. 2018;19:372.e1-372.e8. doi: 10.1016/j.jamda.2017.12.099
54. ABIM Foundation. Choosing wisely. Choosing Wisely website. 2021. Accessed November 11. 2021. www.choosingwisely.org/
55. Berlie HD, Garwood CL. Diabetes medications related to an increased risk of falls and fall-related morbidity in the elderly. Ann Pharmacother. 2010;44:712-717. doi: 10.1345/aph.1M551
56. Hartikainen S, E, Louhivuori K. Medication as a risk factor for falls: critical systematic review. J Gerontol A Biol Sci Med Sci. 2007;62:1172-1181. doi: 10.1093/gerona/62.10.1172
57. Khanuja K, Joki J, Bachmann G, et al. Gait and balance in the aging population: Fall prevention using innovation and technology. Maturitas. 2018;110:51-56. doi: 10.1016/j.maturitas.2018.01.021
58. Salzman B. Gait and balance disorders in older adults. Am Fam Physician. 2010;82:61-68.
59. Zaninotto P, Huang YT, Di Gessa G, et al. Polypharmacy is a risk factor for hospital admission due to a fall: evidence from the English Longitudinal Study of Ageing. BMC Public Health. 2020;20:1804. doi: 10.1186/s12889-020-09920-x
60. Morin L, Calderon A, Welmer AK, et al. Polypharmacy and injurious falls in older adults: a nationwide nested case-control study. Clin Epidemiol. 2019;11:483-493. doi: 10.2147/CLEP.S201614
61. Dhalwani NN, Fahami R, Sathanapally H, et al. Association between polypharmacy and falls in older adults: a longitudinal study from England. BMJ Open. 2017;7:e016358. doi: 10.1136/bmjopen-2017-016358
62. Arnold AC, Raj SR. Orthostatic hypotension: a practical approach to investigation and management. Can J Cardiol. 2017;33:1725-1728. doi: 10.1016/j.cjca.2017.05.007
63. Alexander NB. Differential diagnosis of gait disorders in older adults. Clin Geriatr Med. 1996;12:689-703.
64. Baker JM. Gait disorders. Am J Med. 2018;131:602-607. doi: 10.1016/j.amjmed.2017.11.051
65. Cameron MH, Wagner JM. Gait abnormalities in multiple sclerosis: pathogenesis, evaluation, and advances in treatment. Curr Neurol Neurosci Rep. 2011;11:507-515. doi: 10.1007/s11910-011-0214-y
66. Chen C-L, Chen H-C, Tang SF-T, et al. Gait performance with compensatory adaptations in stroke patients with different degrees of motor recovery. Am J Phys Med Rehabil. 2003;82:925-935. doi: 10.1097/01.PHM.0000098040.13355.B5
67. Marsden J, Harris C. Cerebellar ataxia: pathophysiology and rehabilitation. Clin Rehabil. 2011;25:195-216. doi: 10.1177/0269215510382495
68. Mirek E, Filip M, W, et al. Three-dimensional trunk and lower limbs characteristics during gait in patients with Huntington’s disease. Front Neurosci. 2017;11:566. doi: 10.3389/fnins.2017.00566
69. Paramanandam V, Lizarraga KJ, Soh D, et al. Unusual gait disorders: a phenomenological approach and classification. Expert Rev Neurother. 2019;19:119-132. doi: 10.1080/14737175.2019.1562337
70. Sahyouni R, Goshtasbi K, Mahmoodi A, et al. Chronic subdural hematoma: a historical and clinical perspective. World Neurosurg. 2017;108:948-953. doi: 10.1016/j.wneu.2017.09.064
71. Talmud JD, Coffey R, Edemekong PF. Dix Hallpike maneuver. StatPearls [Internet]. StatPearls Publishing Updated September 5, 2021. Accessed December 6, 2021. www.ncbi.nlm.nih.gov/books/NBK459307/
72. Molnar FJ, Benjamin S, Hawkins SA, et al. One size does not fit all: choosing practical cognitive screening tools for your practice. J Am Geriatr Soc. 2020;68:2207-2213. doi: 10.1111/jgs.16713
73. Gillespie LD, Robertson MC, Gillespie WJ, et al. Interventions for preventing falls in older people living in the community. Cochrane Database Syst Rev. 2012:CD007146. doi: 10.1002/14651858.CD007146.pub3
74. Wongrakpanich S, Wongrakpanich A, Melhado K, Rangaswami J. A comprehensive review of non-steroidal anti-inflammatory drug use in the elderly. Aging Dis. 2018;9:143-150. doi: 10.14336/AD.2017.0306
75. Poe SS, Cvach M, Dawson PB, Straus H, Hill EE. The Johns Hopkins Fall Risk Assessment Tool: postimplementation evaluation. J Nurs Care Qual. 2007;22:293-298. doi: 10.1097/01.NCQ.0000290408.74027.39
76. Poe SS, Dawson PB, Cvach M, et al. The Johns Hopkins Fall Risk Assessment Tool: a study of reliability and validity. J Nurs Care Qual. 2018;33:10-19. doi: 10.1097/NCQ.0000000000000301
77. Klinkenberg WD, Potter P. Validity of the Johns Hopkins Fall Risk Assessment Tool for predicting falls on inpatient medicine services. J Nurs Care Qual. 2017;32:108-113. doi: 10.1097/NCQ.0000000000000210
78. Stapleton C, Hough P, Oldmeadow L, et al. Four-item fall risk screening tool for subacute and residential aged care: the first step in fall prevention. Australas J Ageing. 2009;28:139-143. doi: 10.1111/j.1741-6612.2009.00375.x
79. Cattelani L, Palumbo P, Palmerini L, et al. FRAT-up, a Web-based fall-risk assessment tool for elderly people living in the community. J Med Internet Res. 2015;17:e41. doi: 10.2196/jmir.4064
80. De Clercq H, Naudé A, Bornman J. Factors included in adult fall risk assessment tools (FRATs): a systematic review. Ageing Soc. 2020;41:2558-2582. doi: 10.1017/S0144686X2000046X
81. Yap G, Melder A. Accuracy of validated falls risk assessment tools and clinical judgement. Centre for Clinical Effectiveness, Monash Innovation and Quality. Monash Health. February 5, 2020. Accessed November 11, 2021. https://monashhealth.org/wp-content/uploads/2019/01/Rapid-Review_Falls-risk-tools-FINAL.pdf
82. Chittrakul J, Siviroj P, Sungkarat S, et al. Physical frailty and fall risk in community-dwelling older adults: a cross-sectional study. J Aging Res. 2020;2020:3964973. doi: 10.1155/2020/3964973
83. Hatcher VH, Galet C, Lilienthal M, et al. Association of clinical frailty scores with hospital readmission for falls after index admission for trauma-related injury. JAMA Netw Open. 2019;2:e1912409. doi: 10.1001/jamanetworkopen.2019.12409
84. Exercise and fall prevention programs. Government of Ontario Ministry of Health. Updated April 9, 2019. Accessed November 11. 2021. www.ontario.ca/page/exercise-and-falls-prevention-programs
85. Sherrington C, Fairhall NJ, Wallbank GK, et al. Exercise for preventing falls in older people living in the community. Cochrane Database Syst Rev. 2019;1:CD012424. doi: 10.1002/14651858.CD012424.pub2
86. Hopewell S, Copsey B, Nicolson P, et al. Multifactorial interventions for preventing falls in older people living in the community: a systematic review and meta-analysis of 41 trials and almost 20 000 participants. Br J Sports Med. 2020;54:1340-1350. doi: 10.1136/bjsports-2019-100732
87. Jafari Z, Kolb BE, Mohajerani MH. Age-related hearing loss and tinnitus, dementia risk, and auditory amplification outcomes. Ageing Res Rev. 2019;56:100963. doi: 10.1016/j.arr.2019.100963
88. Griffiths TD, Lad M, Kumar S, et al. How can hearing loss cause dementia? Neuron. 2020;108:401-412. doi: 10.1016/j.neuron.2020.08.003
89. Martini A, Castiglione A, Bovo R, et al. Aging, cognitive load, dementia and hearing loss. Audiol Neurootol. 2014;19(suppl 1):2-5. doi: 10.1159/000371593
90. Vitkovic J, Le C, Lee S-L, et al. The contribution of hearing and hearing loss to balance control. Audiol Neurootol. 2016;21:195-202. doi: 10.1159/000445100
91. Maheu M, Behtani L, Nooristani M, et al. Vestibular function modulates the benefit of hearing aids in people with hearing loss during static postural control. Ear Hear. 2019;40:1418-1424. doi: 10.1097/AUD.0000000000000720
92. Negahban H, Bavarsad Cheshmeh Ali M, Nassadj G. Effect of hearing aids on static balance function in elderly with hearing loss. Gait Posture. 2017;58:126-129. doi: 10.1016/j.gaitpost.2017.07.112
93. Mahmoudi E, Basu T, Langa K, et al. Can hearing aids delay time to diagnosis of dementia, depression, or falls in older adults? J Am Geriatr Soc. 2019;67:2362-2369. doi: 10.1111/jgs.16109
94. Paliwal Y, Slattum PW, Ratliff SM. Chronic health conditions as a risk factor for falls among the community-dwelling US older adults: a zero-inflated regression modeling approach. Biomed Res Int. 2017;2017:5146378. doi: 10.1155/2017/5146378
95. Deandrea S, Lucenteforte E, Bravi F, et al. Risk factors for falls in community-dwelling older people: a systematic review and meta-analysis. Epidemiology. 2010;21:658-668. doi: 10.1097/EDE.0b013e3181e89905
96. Ambrose AF, Paul G, Hausdorff JM. Risk factors for falls among older adults: a review of the literature. Maturitas. 2013;75:51-61. doi: 10.1016/j.maturitas.2013.02.009
97. Stevens M, Holman CD, Bennett N. Preventing falls in older people: impact of an intervention to reduce environmental hazards in the home. J Am Geriatr Soc. 2001;49:1442-1447. doi: 10.1046/j.1532-5415.2001.4911235.x
98. Clinical resources. Centers for Disease Control and Prevention STEADI-Older Adult Fall Prevention website. 2020. Accessed November 12, 2021. www.cdc.gov/steadi/materials.html
99. ; Grossman DC, Curry SJ, Owens DK, et al. Interventions to prevent falls in community-dwelling older adults: US Preventive Services Task Force recommendation statement. JAMA. 2018;319:1696-1704. doi: 10.1001/jama.2018.3097
CASE Mr. J, a 75-year-old man, presents to your family practice reporting that he feels increasingly unsteady and slow while walking. He fell twice last year, without resulting injury. He now worries about tripping while walking around the house and relies on his spouse to run errands.
Clearly, Mr. J is experiencing a problem with balance. What management approach should you undertake to prevent him from falling?

Balance disorders are common in older people and drastically hinder quality of life.1-4 Patients often describe imbalance as vague symptoms: dizziness, unsteadiness, faintness, spinning sensations.5,6 Importantly, balance disorders disrupt normal gait and contribute to falls that are a major cause of disability and morbidity in older people. Almost 30% of people older than 65 years report 1 or more falls annually.7 Factors that increase the risk of falls include impaired mobility, previously reported falls, reduced psychological functioning, chronic medical conditions, and polypharmacy.7,8
The cause of any single case of imbalance is often multifactorial, resulting from dysfunction of multiple body systems (TABLE 17-56); in our clinical experience, most patients with imbalance and who are at risk of falls do not have a detectable deficit of the vestibular system. These alterations in function arise in 3 key systems—vision, proprioception, and vestibular function—which signal to, and are incorporated by, the cerebellum to mediate balance. Cognitive and neurologic decline are also factors in imbalance.

Considering that 20% of falls result in serious injury in older populations, it is important to identify balance disorders and implement preventive strategies to mitigate harmful consequences of falls on patients’ health and independence.7,57 In this article, we answer the question that the case presentation raises about the proper management approach to imbalance in family practice, including assessment of risk and rehabilitation strategies to reduce the risk of falls. Our insights and recommendations are based on our clinical experience and a review of the medical literature from the past 40 years.

CASE Mr. J has a history of hypertension, age-related hearing loss, and osteoarthritis of the knees; he has not had surgery for the arthritis. His medications are antihypertensives and extra-strength acetaminophen for knee pain.

Making the diagnosis of a balance disorder
History
A thorough clinical history, often including a collateral history from caregivers, narrows the differential diagnosis. Information regarding onset, duration, timing, character, and previous episodes of imbalance is essential. Symptoms of imbalance are often challenging for the patient to describe: They might use terms such as vertigo or dizziness, when, in fact, on further questioning, they are describing balance difficulties. Inquiry into (1) their use of assistive walking devices and (2) development or exacerbation of neurologic, musculoskeletal, auditory, visual, and mood symptoms is necessary. Note the current level of their mobility, episodes of pain or fatigue, previous falls and associated injuries, fear of falling, balance confidence, and sensations that precede falls.58
Continue to: The medical and surgical histories
The medical and surgical histories are key pieces of information. The history of smoking, alcohol habits, and substance use is relevant.
A robust medication history is essential to evaluate a patient’s risk of falling. Polypharmacy—typically, defined as taking 4 or more medications—has been repeatedly associated with a heightened risk of falls.53,59-61 Moreover, a dose-dependent association between polypharmacy and hospitalization following falls has been identified, and demonstrates that taking 10 or more medications greatly increases the risk of hospitalization.59 Studies of polypharmacy cement the importance of inquiring about medication use when assessing imbalance, particularly in older patients.
Physical examination
A focused and detailed physical examination provides insight into systems that should be investigated:
- Obtain vital signs, including orthostatic vitals to test for orthostatic hypotension62; keep in mind that symptoms of orthostatic dizziness can occur without orthostatic hypotension.
- Examine gait, which can distinguish between causes of imbalance (TABLE 2).21,40,63-70
- Perform a cardiac examination.
- Assess visual acuity and visual fields; test for nystagmus and identify any optic-nerve and retinal abnormalities.
- Evaluate lower-limb sensation, proprioception, and motor function.
- Evaluate suspected vestibular dysfunction, including dysfunction with positional testing (the Dix-Hallpike maneuver71). The patient is taken from sitting to supine while the head is rotated 45° to the tested side by the examiner. As the patient moves into a supine position, the neck is extended 30° off the table and held for at least 30 seconds. The maneuver is positive if torsional nystagmus is noted while the head is held rotated during neck extension. The maneuver is negative if the patient reports dizziness, vertigo, unsteadiness, or “pressure in the head.” Torsional nystagmus must be present to confirm a diagnosis of benign paroxysmal positional vertigo.
- If you suspect a central nervous system cause of imbalance, assess the cranial nerves, coordination, strength, and, of course, balance.

CASE
Mr. J’s physical examination showed normal vital signs without significant postural changes in blood pressure. Gait analysis revealed a slowed gait, with reduced range of motion in both knees over the entire gait cycle. Audiometry revealed symmetric moderate sensorineural hearing loss characteristic of presbycusis.
Diagnostic investigations
Consider focused investigations into imbalance based on the history and physical examination. We discourage overly broad testing and imaging; in primary care, cost and limited access to technology can bar robust investigations into causes of imbalance. However, identification of acute pathologies should prompt immediate referral to the emergency department. Furthermore, specific symptoms (TABLE 17-56) should prompt referral to specialists for assessment.
Continue to: In the emergency department...
In the emergency department and academic hospitals, key investigations can identify causes of imbalance:
- Electrocardiography and Holter monitoring test for cardiac arrhythmias.
- Echocardiography identifies structural abnormalities.
- Radiography and computed tomography are useful for detecting musculoskeletal abnormalities.
- Bone densitometry can identify osteoporosis.
- Head and spinal cord magnetic resonance imaging can be used to identify lesions of the central nervous system.
- Computed tomographic angiography of the head and neck is useful for identifying stroke, cerebral atrophy, and stenotic lesions of the carotid and vertebral arteries.
- Nerve conduction studies and levels of serum vitamin B12, hemoglobin A1C, thyroid-stimulating hormone, and random cortisol can uncover causes of peripheral neuropathy.
- Bedside cognitive screening tests can be used to measure cognitive decline.72
- Suspicion of vestibular disease requires audiometry and vestibular testing, including videonystagmography, head impulse testing, and vestibular evoked myogenic potentials.
In many cases of imbalance, no specific underlying correctable cause is discovered.
Management of imbalance
Pharmacotherapy
Targeted pharmacotherapy can be utilized in select clinical scenarios:
- Medical treatment of peripheral neuropathy should target the underlying condition.
- Cognitive behavioral therapy and antidepressants are useful for treating anxiety and depressive disorders.73
- Musculoskeletal pain can be managed with acetaminophen and topical nonsteroidal anti-inflammatory drugs (NSAIDs), using a short course of an oral NSAID when needed.74
- Cardiovascular disease management might include any of several classes of pharmacotherapy, including antiplatelet and lipid-lowering medications, antiarrhythmic drugs, and antihypertensive agents.
- Acute episodes of vertigo due to vestibular neuritis or labyrinthitis can be managed with an antiemetic.46
Surgical treatment
Surgery is infrequently considered for patients with imbalance. Examples of indications include microsurgical resection of vestibular schwannoma, resection of central nervous system tumors, lens replacement surgery for cataract, surgical management of severe spinal fracture, and hip or knee arthroplasty in select patients.
Tools for assessing the risk of falls
Scoring systems called falls risk assessment tools, or FRAT, have been developed to gauge a patient’s risk of falling. The various FRATs differ in specificity and sensitivity for predicting the risk of falls, and are typically designed for specific clinical environments, such as hospital inpatient care or long-term care facilities. Specifically, FRATs attempt to classify risk using sets of risk factors known to be associated with falls.
Continue to: Research abounds into...
Research abounds into the validity of commonly used FRATs across institutions, patient populations, and clinical environments:
The Johns Hopkins FRATa determines risk using metrics such as age, fall history, incontinence, cognition, mobility, and medications75; it is predominantly used for assessment in hospital inpatient units. This tool has been validated repeatedly.76,77
Peninsula Health FRATb stratifies patients in subacute and residential aged-care settings, based on risk factors that include recent falls, medications, psychological status, and cognition.78
FRAT-upc is a web-based tool that generates falls risk using risk factors that users input. This tool has been studied in the context of patients older than 65 years living in the community.79
Although FRATs are reasonably useful for predicting falls, their utility varies by patient population and clinical context. Moreover, it has been suggested that FRATs neglect environmental and personal factors when assessing risk by focusing primarily on bodily factors.80 Implementing a FRAT requires extensive consideration of the target population and should be accompanied by clinical judgment that is grounded in an individual patient’s circumstances.81
Continue to: Preventing falls in primary care
Preventing falls in primary care
An approach to preventing falls includes the development of individualized programs that account for frailty, a syndrome of physiologic decline associated with aging. Because frailty leads to diminished balance and mobility, a patient’s frailty index—determined using the 5 frailty phenotype criteria (exhaustion, weight loss, low physical activity, weakness, slowness)82 or the Canadian Study of Health and Aging Clinical Frailty Scale83—is a useful tool for predicting falls risk and readmission for falls following trauma-related injury. Prevention of falls in communities is critical for reducing mortality and allowing older people to maintain their independence and quality of life.
Exercise. In some areas, exercise and falls prevention programs are accessible to seniors.84 Community exercise programs that focus on balance retraining and muscle strengthening can reduce the risk of falls.73,85 The Choosing Wisely initiative of the ABIM [American Board of Internal Medicine] Foundation recommends that exercise programs be designed around an accurate functional baseline of the patient to avoid underdosed strength training.54
Multifactorial risk assessment in high-risk patients can reduce the rate of falls. Such an assessment includes examination of orthostatic blood pressure, vision and hearing, bone health, gait, activities of daily living, cognition, and environmental hazards, and enables provision of necessary interventions.73,86 Hearing amplification, specifically, correlates with enhanced postural control, slowed cognitive decline, and a reduced likelihood of falls.87-93 The mechanism behind improved balance performance might be reduced cognitive load through supporting a patient’s listening needs.88-90
Pharmacotherapy. Optimizing medications and performing a complete medication review before prescribing new medications is highly recommended to avoid unnecessary polypharmacy7,8,18,53-56 (TABLE 17-56).
Management of comorbidities associated with a higher risk of falls, including arthritis, cancer, stroke, diabetes, depression, kidney disease, chronic obstructive pulmonary disease, cognitive impairment, hypertension, and atrial fibrillation, is essential.94-96
Continue to: Home safety interventions
Home safety interventions, through occupational therapy, are important. These include removing unsafe mats and step-overs and installing nonslip strips on stairs, double-sided tape under mats, and handrails.73-97
Screening for risk of falls. The Centers for Disease Control and Prevention recommends that (1) all patients older than 65 years and (2) any patient presenting with an acute fall undergo screening for their risk of falls.98 When a patient is identified as at risk of falling, you can, when appropriate, assess modifiable risk factors and facilitate interventions.98 This strategy is supported by a 2018 statement from the US Preventive Services Task Force99 that recommends identifying high-risk patients who have:
- a history of falling
- a balance disturbance that causes a deficit of mobility or function
- poor performance on clinical tests, such as the 3-meter Timed Up and Go (TUG) assessment (www.cdc.gov/steadi/pdf/TUG_test-print.pdf).
An increased risk of falls should prompt you to refer the patient to community programs and physiotherapy in accordance with the individual’s personal goals99; a balance and vestibular physiotherapist is ideally positioned to accurately assess and manage patients at risk of falls. Specifically, the Task Force identified exercise programs and multifactorial interventions as being beneficial in preventing falls in high-risk older people.99
Balance assessment and rehabilitation in specialty centers
An individualized rehabilitation program aims to restore safe mobility by testing and addressing specific balance deficits, improving functional balance, and increasing balance confidence. Collaboration with colleagues from physiotherapy and occupational therapy aids in tailoring individualized programs.
Many tests are available to assess balance, determine the risk of falls, and guide rehabilitation:
- The timed 10-meter walk testd and the TUG test are simple assessments that measure functional mobility; both have normalized values for the risk of falls. A TUG time of ≥ 12 seconds suggests a high risk of falls.
- The 30-second chair stande evaluates functional lower-extremity strength in older patients. The test can indicate if lower-extremity strength is contributing to a patient’s imbalance.
- The modified clinical test of sensory interaction in balancef is a static balance test that measures the integrity of sensory inputs. The test can suggest if 1 or more sensory systems are compromised.
- The mini balance evaluation systems testg is similar: It can differentiate balance deficits by underlying system and allows individualization of a rehabilitation program.
- The functional gait assessmenth is a modification of the dynamic gait index that assesses postural stability during everyday dynamic activities, including tasks such as walking with head turns and pivots.
- The Berg Balance Scalei continues to be used extensively to assess balance.
Continue to: The mini balance evaluation systems test...
The mini balance evaluation systems test, functional gait index, and Berg Balance Scale all have normative age-graded values to predict fall risk.
CASE
Mr. J was referred for balance assessment and to a rehabilitation program. He underwent balance physiotherapy, including multifactorial balance assessment, joined a community exercise program, was fitted with hearing aids, and had his home environment optimized by an occupational therapist. (See examples of “home safety interventions” under “Preventing falls in primary care.”)
3 months later. Mr. J says he feels stronger on his feet. His knee pain has eased, and he is more confident walking around his home. He continues to engage in exercise programs and is comfortable running errands with his spouse.
CORRESPONDENCE
Jason A. Beyea, MD, PhD, FRCSC, Division of OtolaryngologyHead and Neck Surgery, Queen’s University, 144 Brock Street, Kingston, Ontario, Canada, K7L 5G2; [email protected]
a www.hopkinsmedicine.org/institute_nursing/models_tools/jhfrat_acute%20care%20original_6_22_17.pdf
c www.ncbi.nlm.nih.gov/pmc/articles/PMC4376110/figure/figure14/?report=objectonly
e www.cdc.gov/steadi/pdf/STEADI-Assessment-30Sec-508.pdf
f www.mdapp.co/mctsib-modified-clinical-test-of-sensory-interaction-in-balance-calculator-404/
g www.sralab.org/sites/default/files/2017-07/MiniBEST_revised_final_3_8_13.pdf
CASE Mr. J, a 75-year-old man, presents to your family practice reporting that he feels increasingly unsteady and slow while walking. He fell twice last year, without resulting injury. He now worries about tripping while walking around the house and relies on his spouse to run errands.
Clearly, Mr. J is experiencing a problem with balance. What management approach should you undertake to prevent him from falling?

Balance disorders are common in older people and drastically hinder quality of life.1-4 Patients often describe imbalance as vague symptoms: dizziness, unsteadiness, faintness, spinning sensations.5,6 Importantly, balance disorders disrupt normal gait and contribute to falls that are a major cause of disability and morbidity in older people. Almost 30% of people older than 65 years report 1 or more falls annually.7 Factors that increase the risk of falls include impaired mobility, previously reported falls, reduced psychological functioning, chronic medical conditions, and polypharmacy.7,8
The cause of any single case of imbalance is often multifactorial, resulting from dysfunction of multiple body systems (TABLE 17-56); in our clinical experience, most patients with imbalance and who are at risk of falls do not have a detectable deficit of the vestibular system. These alterations in function arise in 3 key systems—vision, proprioception, and vestibular function—which signal to, and are incorporated by, the cerebellum to mediate balance. Cognitive and neurologic decline are also factors in imbalance.

Considering that 20% of falls result in serious injury in older populations, it is important to identify balance disorders and implement preventive strategies to mitigate harmful consequences of falls on patients’ health and independence.7,57 In this article, we answer the question that the case presentation raises about the proper management approach to imbalance in family practice, including assessment of risk and rehabilitation strategies to reduce the risk of falls. Our insights and recommendations are based on our clinical experience and a review of the medical literature from the past 40 years.

CASE Mr. J has a history of hypertension, age-related hearing loss, and osteoarthritis of the knees; he has not had surgery for the arthritis. His medications are antihypertensives and extra-strength acetaminophen for knee pain.

Making the diagnosis of a balance disorder
History
A thorough clinical history, often including a collateral history from caregivers, narrows the differential diagnosis. Information regarding onset, duration, timing, character, and previous episodes of imbalance is essential. Symptoms of imbalance are often challenging for the patient to describe: They might use terms such as vertigo or dizziness, when, in fact, on further questioning, they are describing balance difficulties. Inquiry into (1) their use of assistive walking devices and (2) development or exacerbation of neurologic, musculoskeletal, auditory, visual, and mood symptoms is necessary. Note the current level of their mobility, episodes of pain or fatigue, previous falls and associated injuries, fear of falling, balance confidence, and sensations that precede falls.58
Continue to: The medical and surgical histories
The medical and surgical histories are key pieces of information. The history of smoking, alcohol habits, and substance use is relevant.
A robust medication history is essential to evaluate a patient’s risk of falling. Polypharmacy—typically, defined as taking 4 or more medications—has been repeatedly associated with a heightened risk of falls.53,59-61 Moreover, a dose-dependent association between polypharmacy and hospitalization following falls has been identified, and demonstrates that taking 10 or more medications greatly increases the risk of hospitalization.59 Studies of polypharmacy cement the importance of inquiring about medication use when assessing imbalance, particularly in older patients.
Physical examination
A focused and detailed physical examination provides insight into systems that should be investigated:
- Obtain vital signs, including orthostatic vitals to test for orthostatic hypotension62; keep in mind that symptoms of orthostatic dizziness can occur without orthostatic hypotension.
- Examine gait, which can distinguish between causes of imbalance (TABLE 2).21,40,63-70
- Perform a cardiac examination.
- Assess visual acuity and visual fields; test for nystagmus and identify any optic-nerve and retinal abnormalities.
- Evaluate lower-limb sensation, proprioception, and motor function.
- Evaluate suspected vestibular dysfunction, including dysfunction with positional testing (the Dix-Hallpike maneuver71). The patient is taken from sitting to supine while the head is rotated 45° to the tested side by the examiner. As the patient moves into a supine position, the neck is extended 30° off the table and held for at least 30 seconds. The maneuver is positive if torsional nystagmus is noted while the head is held rotated during neck extension. The maneuver is negative if the patient reports dizziness, vertigo, unsteadiness, or “pressure in the head.” Torsional nystagmus must be present to confirm a diagnosis of benign paroxysmal positional vertigo.
- If you suspect a central nervous system cause of imbalance, assess the cranial nerves, coordination, strength, and, of course, balance.

CASE
Mr. J’s physical examination showed normal vital signs without significant postural changes in blood pressure. Gait analysis revealed a slowed gait, with reduced range of motion in both knees over the entire gait cycle. Audiometry revealed symmetric moderate sensorineural hearing loss characteristic of presbycusis.
Diagnostic investigations
Consider focused investigations into imbalance based on the history and physical examination. We discourage overly broad testing and imaging; in primary care, cost and limited access to technology can bar robust investigations into causes of imbalance. However, identification of acute pathologies should prompt immediate referral to the emergency department. Furthermore, specific symptoms (TABLE 17-56) should prompt referral to specialists for assessment.
Continue to: In the emergency department...
In the emergency department and academic hospitals, key investigations can identify causes of imbalance:
- Electrocardiography and Holter monitoring test for cardiac arrhythmias.
- Echocardiography identifies structural abnormalities.
- Radiography and computed tomography are useful for detecting musculoskeletal abnormalities.
- Bone densitometry can identify osteoporosis.
- Head and spinal cord magnetic resonance imaging can be used to identify lesions of the central nervous system.
- Computed tomographic angiography of the head and neck is useful for identifying stroke, cerebral atrophy, and stenotic lesions of the carotid and vertebral arteries.
- Nerve conduction studies and levels of serum vitamin B12, hemoglobin A1C, thyroid-stimulating hormone, and random cortisol can uncover causes of peripheral neuropathy.
- Bedside cognitive screening tests can be used to measure cognitive decline.72
- Suspicion of vestibular disease requires audiometry and vestibular testing, including videonystagmography, head impulse testing, and vestibular evoked myogenic potentials.
In many cases of imbalance, no specific underlying correctable cause is discovered.
Management of imbalance
Pharmacotherapy
Targeted pharmacotherapy can be utilized in select clinical scenarios:
- Medical treatment of peripheral neuropathy should target the underlying condition.
- Cognitive behavioral therapy and antidepressants are useful for treating anxiety and depressive disorders.73
- Musculoskeletal pain can be managed with acetaminophen and topical nonsteroidal anti-inflammatory drugs (NSAIDs), using a short course of an oral NSAID when needed.74
- Cardiovascular disease management might include any of several classes of pharmacotherapy, including antiplatelet and lipid-lowering medications, antiarrhythmic drugs, and antihypertensive agents.
- Acute episodes of vertigo due to vestibular neuritis or labyrinthitis can be managed with an antiemetic.46
Surgical treatment
Surgery is infrequently considered for patients with imbalance. Examples of indications include microsurgical resection of vestibular schwannoma, resection of central nervous system tumors, lens replacement surgery for cataract, surgical management of severe spinal fracture, and hip or knee arthroplasty in select patients.
Tools for assessing the risk of falls
Scoring systems called falls risk assessment tools, or FRAT, have been developed to gauge a patient’s risk of falling. The various FRATs differ in specificity and sensitivity for predicting the risk of falls, and are typically designed for specific clinical environments, such as hospital inpatient care or long-term care facilities. Specifically, FRATs attempt to classify risk using sets of risk factors known to be associated with falls.
Continue to: Research abounds into...
Research abounds into the validity of commonly used FRATs across institutions, patient populations, and clinical environments:
The Johns Hopkins FRATa determines risk using metrics such as age, fall history, incontinence, cognition, mobility, and medications75; it is predominantly used for assessment in hospital inpatient units. This tool has been validated repeatedly.76,77
Peninsula Health FRATb stratifies patients in subacute and residential aged-care settings, based on risk factors that include recent falls, medications, psychological status, and cognition.78
FRAT-upc is a web-based tool that generates falls risk using risk factors that users input. This tool has been studied in the context of patients older than 65 years living in the community.79
Although FRATs are reasonably useful for predicting falls, their utility varies by patient population and clinical context. Moreover, it has been suggested that FRATs neglect environmental and personal factors when assessing risk by focusing primarily on bodily factors.80 Implementing a FRAT requires extensive consideration of the target population and should be accompanied by clinical judgment that is grounded in an individual patient’s circumstances.81
Continue to: Preventing falls in primary care
Preventing falls in primary care
An approach to preventing falls includes the development of individualized programs that account for frailty, a syndrome of physiologic decline associated with aging. Because frailty leads to diminished balance and mobility, a patient’s frailty index—determined using the 5 frailty phenotype criteria (exhaustion, weight loss, low physical activity, weakness, slowness)82 or the Canadian Study of Health and Aging Clinical Frailty Scale83—is a useful tool for predicting falls risk and readmission for falls following trauma-related injury. Prevention of falls in communities is critical for reducing mortality and allowing older people to maintain their independence and quality of life.
Exercise. In some areas, exercise and falls prevention programs are accessible to seniors.84 Community exercise programs that focus on balance retraining and muscle strengthening can reduce the risk of falls.73,85 The Choosing Wisely initiative of the ABIM [American Board of Internal Medicine] Foundation recommends that exercise programs be designed around an accurate functional baseline of the patient to avoid underdosed strength training.54
Multifactorial risk assessment in high-risk patients can reduce the rate of falls. Such an assessment includes examination of orthostatic blood pressure, vision and hearing, bone health, gait, activities of daily living, cognition, and environmental hazards, and enables provision of necessary interventions.73,86 Hearing amplification, specifically, correlates with enhanced postural control, slowed cognitive decline, and a reduced likelihood of falls.87-93 The mechanism behind improved balance performance might be reduced cognitive load through supporting a patient’s listening needs.88-90
Pharmacotherapy. Optimizing medications and performing a complete medication review before prescribing new medications is highly recommended to avoid unnecessary polypharmacy7,8,18,53-56 (TABLE 17-56).
Management of comorbidities associated with a higher risk of falls, including arthritis, cancer, stroke, diabetes, depression, kidney disease, chronic obstructive pulmonary disease, cognitive impairment, hypertension, and atrial fibrillation, is essential.94-96
Continue to: Home safety interventions
Home safety interventions, through occupational therapy, are important. These include removing unsafe mats and step-overs and installing nonslip strips on stairs, double-sided tape under mats, and handrails.73-97
Screening for risk of falls. The Centers for Disease Control and Prevention recommends that (1) all patients older than 65 years and (2) any patient presenting with an acute fall undergo screening for their risk of falls.98 When a patient is identified as at risk of falling, you can, when appropriate, assess modifiable risk factors and facilitate interventions.98 This strategy is supported by a 2018 statement from the US Preventive Services Task Force99 that recommends identifying high-risk patients who have:
- a history of falling
- a balance disturbance that causes a deficit of mobility or function
- poor performance on clinical tests, such as the 3-meter Timed Up and Go (TUG) assessment (www.cdc.gov/steadi/pdf/TUG_test-print.pdf).
An increased risk of falls should prompt you to refer the patient to community programs and physiotherapy in accordance with the individual’s personal goals99; a balance and vestibular physiotherapist is ideally positioned to accurately assess and manage patients at risk of falls. Specifically, the Task Force identified exercise programs and multifactorial interventions as being beneficial in preventing falls in high-risk older people.99
Balance assessment and rehabilitation in specialty centers
An individualized rehabilitation program aims to restore safe mobility by testing and addressing specific balance deficits, improving functional balance, and increasing balance confidence. Collaboration with colleagues from physiotherapy and occupational therapy aids in tailoring individualized programs.
Many tests are available to assess balance, determine the risk of falls, and guide rehabilitation:
- The timed 10-meter walk testd and the TUG test are simple assessments that measure functional mobility; both have normalized values for the risk of falls. A TUG time of ≥ 12 seconds suggests a high risk of falls.
- The 30-second chair stande evaluates functional lower-extremity strength in older patients. The test can indicate if lower-extremity strength is contributing to a patient’s imbalance.
- The modified clinical test of sensory interaction in balancef is a static balance test that measures the integrity of sensory inputs. The test can suggest if 1 or more sensory systems are compromised.
- The mini balance evaluation systems testg is similar: It can differentiate balance deficits by underlying system and allows individualization of a rehabilitation program.
- The functional gait assessmenth is a modification of the dynamic gait index that assesses postural stability during everyday dynamic activities, including tasks such as walking with head turns and pivots.
- The Berg Balance Scalei continues to be used extensively to assess balance.
Continue to: The mini balance evaluation systems test...
The mini balance evaluation systems test, functional gait index, and Berg Balance Scale all have normative age-graded values to predict fall risk.
CASE
Mr. J was referred for balance assessment and to a rehabilitation program. He underwent balance physiotherapy, including multifactorial balance assessment, joined a community exercise program, was fitted with hearing aids, and had his home environment optimized by an occupational therapist. (See examples of “home safety interventions” under “Preventing falls in primary care.”)
3 months later. Mr. J says he feels stronger on his feet. His knee pain has eased, and he is more confident walking around his home. He continues to engage in exercise programs and is comfortable running errands with his spouse.
CORRESPONDENCE
Jason A. Beyea, MD, PhD, FRCSC, Division of OtolaryngologyHead and Neck Surgery, Queen’s University, 144 Brock Street, Kingston, Ontario, Canada, K7L 5G2; [email protected]
a www.hopkinsmedicine.org/institute_nursing/models_tools/jhfrat_acute%20care%20original_6_22_17.pdf
c www.ncbi.nlm.nih.gov/pmc/articles/PMC4376110/figure/figure14/?report=objectonly
e www.cdc.gov/steadi/pdf/STEADI-Assessment-30Sec-508.pdf
f www.mdapp.co/mctsib-modified-clinical-test-of-sensory-interaction-in-balance-calculator-404/
g www.sralab.org/sites/default/files/2017-07/MiniBEST_revised_final_3_8_13.pdf
1. Larocca NG. Impact of walking impairment in multiple sclerosis: perspectives of patients and care partners. Patient. 2011;4:189-201. doi: 10.2165/11591150-000000000-00000
2. TB, ZF, ES, et al. The relationship of balance disorders with falling, the effect of health problems, and social life on postural balance in the elderly living in a district in Turkey. Geriatrics (Basel). 2019;4:37. doi: 10.3390/geriatrics4020037
3. R, Sixt E, Landahl S, et al. Prevalence of dizziness and vertigo in an urban elderly population. J Vestib Res. 2004;14:47-52.
4. Sturnieks DL, St George R, Lord SR. Balance disorders in the elderly. Neurophysiol Clin. 2008;38:467-478. doi: 10.1016/j.neucli.2008.09.001
5. Boult C, Murphy J, Sloane P, et al. The relation of dizziness to functional decline. J Am Geriatr Soc. 1991;39:858-861. doi: 10.1111/j.1532-5415.1991.tb04451.x
6. Lin HW, Bhattacharyya N. Balance disorders in the elderly: epidemiology and functional impact. Laryngoscope. 2012;122:1858-1861. doi: 10.1002/lary.23376
7. Jia H, Lubetkin EI, DeMichele K, et al. Prevalence, risk factors, and burden of disease for falls and balance or walking problems among older adults in the U.S. Prev Med. 2019;126:105737. doi: 10.1016/j.ypmed.2019.05.025
8. Al-Momani M, Al-Momani F, Alghadir AH, et al. Factors related to gait and balance deficits in older adults. Clin Interv Aging. 2016;11:1043-1049. doi: 10.2147/CIA.S112282
9. Agrawal Y, Ward BK, Minor LB. Vestibular dysfunction: prevalence, impact and need for targeted treatment. J Vestib Res. 2013;23:113-117. doi: 10.3233/VES-130498
10. Altinsoy B, Erboy F, Tanriverdi H, et al. Syncope as a presentation of acute pulmonary embolism. Ther Clin Risk Manag. 2016;12:1023-1028. doi: 10.2147/TCRM.S105722
11. Belvederi Murri M, Triolo F, Coni A, et al. Instrumental assessment of balance and gait in depression: a systematic review. Psychiatry Res. 2020;284:112687. doi: 10.1016/j.psychres.2019.112687
12. Bhattacharyya N, Gubbels SP, Schwartz SR, et al. Clinical practice guideline: benign paroxysmal positional vertigo (update). Otolaryngol Head Neck Surg. 2017;156(suppl 3):S1-S47. doi: 10.1177/0194599816689667
13. S, Schwarm S, Grevenrath P, et al. Prevalence, aetiologies and prognosis of the symptom dizziness in primary care - a systematic review. BMC Fam Pract. 2018;19:33. doi: 10.1186/s12875-017-0695-0
14. Brouwer MC, Tunkel AR, van de Beek D. Epidemiology, diagnosis, and antimicrobial treatment of acute bacterial meningitis. Clin Microbiol Rev. 2010;23:467-492. doi: 10.1128/CMR.00070-09
15. Chad DA. Lumbar spinal stenosis. Neurol Clin. 2007;25:407-418. doi: 10.1016/j.ncl.2007.01.003
16. Conrad BP, Shokat MS, Abbasi AZ, et al. Associations of self-report measures with gait, range of motion and proprioception in patients with lumbar spinal stenosis. Gait Posture. 2013;38:987-992. doi: 10.1016/j.gaitpost.2013.05.010
17. de Luna RA, Mihailovic A, Nguyen AM, et al. The association of glaucomatous visual field loss and balance. Transl Vis Sci Technol. 2017;6:8. doi: 10.1167/tvst.6.3.8
18. DiSogra RM. Common aminoglycosides and platinum-based ototoxic drugs: cochlear/vestibular side effects and incidence. Semin Hear. 2019;40:104-107. doi: 10.1055/s-0039-1684040
19. Ebersbach G, Moreau C, Gandor F, et al. Clinical syndromes: parkinsonian gait. Mov Disord. 2013;28:1552-1559. doi: 10.1002/mds.25675
20. Evans WJ. Skeletal muscle loss: cachexia, sarcopenia, and inactivity. Am J Clin Nutr. 2010;91:1123S-1127S. doi: 10.3945/ajcn.2010.28608A
21. Filli L, Sutter T, Easthope CS, et al. Profiling walking dysfunction in multiple sclerosis: characterisation, classification and progression over time. Sci Rep. 2018;8:4984. doi: 10.1038/s41598-018-22676-0
22. Fritz NE, Kegelmeyer DA, Kloos AD, et al. Motor performance differentiates individuals with Lewy body dementia, Parkinson’s and Alzheimer’s disease. Gait Posture. 2016;50:1-7. doi: 10.1016/j.gaitpost.2016.08.009
23. Furman JM, Jacob RG. A clinical taxonomy of dizziness and anxiety in the otoneurological setting. J Anxiety Disord. 2001;15:9-26. doi: 10.1016/s0887-6185(00)00040-2
24. Furman JM, Marcus DA, Balaban CD. Vestibular migraine: clinical aspects and pathophysiology. Lancet Neurol. 2013;12:706-715. doi: 10.1016/S1474-4422(13)70107-8
25. Gerson LW, Jarjoura D, McCord G. Risk of imbalance in elderly people with impaired hearing or vision. Age Ageing. 1989;18:31-34. doi: 10.1093/ageing/18.1.31
26. Goudakos JK, Markou KD, Franco-Vidal V, et al. Corticosteroids in the treatment of vestibular neuritis: a systematic review and meta-analysis. Otol Neurotol. 2010;31:183-189. doi: 10.1097/MAO.0b013e3181ca843d
27. Green AD, CS, Bastian L, et al. Does this woman have osteoporosis? JAMA. 2004;292:2890-2900. doi: 10.1001/jama.292.23.2890
28. Hallemans A, Ortibus E, Meire F, et al. Low vision affects dynamic stability of gait. Gait Posture. 2010;32:547-551. doi: 10.1016/j.gaitpost.2010.07.018
29. Handelsman JA. Vestibulotoxicity: strategies for clinical diagnosis and rehabilitation. Int J Audiol. 2018;57(suppl 4):S99-S107. doi: 10.1080/14992027.2018.1468092
30. Head VA, Wakerley BR. Guillain-Barré syndrome in general practice: clinical features suggestive of early diagnosis. Br J Gen Pract. 2016;66:218-219. doi: 10.3399/bjgp16X684733
31. Helbostad JL, Vereijken B, Hesseberg K, et al. Altered vision destabilizes gait in older persons. Gait Posture. 2009;30:233-238. doi: 10.1016/j.gaitpost.2009.05.004
32. Hsu W-L, Chen C-Y, Tsauo J-Y, et al. Balance control in elderly people with osteoporosis. J Formos Med Assoc. 2014;113:334-339. doi: 10.1016/j.jfma.2014.02.006
33. Kim H-S, Yun DH, Yoo SD, et al. Balance control and knee osteoarthritis severity. Ann Rehabil Med. 2011;35:701-709. doi: 10.5535/arm.2011.35.5.701
34. Li L, Simonsick EM, Ferrucci L, et al. Hearing loss and gait speed among older adults in the United States. Gait Posture. 2013;38:25-29.
35. McKeith IG, Boeve BF, Dickson DW, et al. Diagnosis and management of dementia with Lewy bodies: fourth consensus report of the DLB Consortium. Neurology. 2017;89:88-100. doi: 10.1212/WNL.0000000000004058
36. Milner KA, Funk M, Richards S, et al. Gender differences in symptom presentation associated with coronary heart disease. Am J Cardiol. 1999;84:396-399. doi: 10.1016/s0002-9149(99)00322-7
37. Paillard T, F, Bru N, et al. The impact of time of day on the gait and balance control of Alzheimer’s patients. Chronobiol Int. 2016;33:161-168. doi: 10.3109/07420528.2015.1124885
38. Paldor I, Chen AS, Kaye AH. Growth rate of vestibular schwannoma. J Clin Neurosci. 2016;32:1-8. doi: 10.1016/j.jocn.2016.05.003
39. Picorelli AMA, Hatton AL, Gane EM, et al. Balance performance in older adults with hip osteoarthritis: a systematic review. Gait Posture. 2018;65:89-99. doi: 10.1016/j.gaitpost.2018.07.001
40. Raccagni C, Nonnekes J, Bloem BR, et al. Gait and postural disorders in parkinsonism: a clinical approach. J Neurol. 2020;267:3169-3176. doi: 10.1007/s00415-019-09382-1
41. Shanmugarajah PD, Hoggard N, Currie S, et al. Alcohol-related cerebellar degeneration: not all down to toxicity? Cerebellum Ataxias. 2016;3:17. doi: 10.1186/s40673-016-0055-1
42. Shih RY, Smirniotopoulos JG. Posterior fossa tumors in adult patients. Neuroimaging Clin N Am. 2016;26:493-510. doi: 10.1016/j.nic.2016.06.003
43. Smith EE. Clinical presentations and epidemiology of vascular dementia. Clin Sci (Lond). 2017;131:1059-1068. doi: 10.1042/CS20160607
44. Streur M, Ratcliffe SJ, Ball J, et al. Symptom clusters in adults with chronic atrial fibrillation. J Cardiovasc Nurs. 2017;32:296-303. doi: 10.1097/JCN.0000000000000344
45. Strupp M, M, JA. Peripheral vestibular disorders: an update. Curr Opin Neurol. 2019;32:165-173. doi: 10.1097/WCO.0000000000000649
46. Thompson TL, Amedee R. Vertigo: a review of common peripheral and central vestibular disorders. Ochsner J. 2009;9:20-26.
47. Timar B, Timar R, L, et al. The impact of diabetic neuropathy on balance and on the risk of falls in patients with type 2 diabetes mellitus: a cross-sectional study. PLoS One. 2016;11:e0154654. doi: 10.1371/journal.pone.0154654
48. Walls R, Hockberger R, Gausche-Hill M. Peripheral nerve disorders. In: Rosen’s Emergency Medicine: Concepts and Clinical Practice. 9th ed. Elsevier, Inc; 2018:1307-1320.
49. Watson JC, Dyck PJB. Peripheral neuropathy: a practical approach to diagnosis and symptom management. Mayo Clin Proc. 2015;90:940-951. doi: 10.1016/j.mayocp.2015.05.004
50. Whitfield KC, Bourassa MW, Adamolekun B, et al. Thiamine deficiency disorders: diagnosis, prevalence, and a roadmap for global control programs. Ann N Y Acad Sci. 2018;1430:3-43. doi: 10.1111/nyas.13919
51. Wu V, Sykes EA, Beyea MM, et al. Approach to Meniere disease management. Can Fam Physician. 2019;65:463-467.
52. Yew KS, Cheng EM. Diagnosis of acute stroke. Am Fam Physician. 2015;91:528-536.
53. Seppala LJ, van de Glind EMM, Daams JG, et al; . Fall-risk-increasing drugs: a systematic review and meta-analysis: III. Others. J Am Med Dir Assoc. 2018;19:372.e1-372.e8. doi: 10.1016/j.jamda.2017.12.099
54. ABIM Foundation. Choosing wisely. Choosing Wisely website. 2021. Accessed November 11. 2021. www.choosingwisely.org/
55. Berlie HD, Garwood CL. Diabetes medications related to an increased risk of falls and fall-related morbidity in the elderly. Ann Pharmacother. 2010;44:712-717. doi: 10.1345/aph.1M551
56. Hartikainen S, E, Louhivuori K. Medication as a risk factor for falls: critical systematic review. J Gerontol A Biol Sci Med Sci. 2007;62:1172-1181. doi: 10.1093/gerona/62.10.1172
57. Khanuja K, Joki J, Bachmann G, et al. Gait and balance in the aging population: Fall prevention using innovation and technology. Maturitas. 2018;110:51-56. doi: 10.1016/j.maturitas.2018.01.021
58. Salzman B. Gait and balance disorders in older adults. Am Fam Physician. 2010;82:61-68.
59. Zaninotto P, Huang YT, Di Gessa G, et al. Polypharmacy is a risk factor for hospital admission due to a fall: evidence from the English Longitudinal Study of Ageing. BMC Public Health. 2020;20:1804. doi: 10.1186/s12889-020-09920-x
60. Morin L, Calderon A, Welmer AK, et al. Polypharmacy and injurious falls in older adults: a nationwide nested case-control study. Clin Epidemiol. 2019;11:483-493. doi: 10.2147/CLEP.S201614
61. Dhalwani NN, Fahami R, Sathanapally H, et al. Association between polypharmacy and falls in older adults: a longitudinal study from England. BMJ Open. 2017;7:e016358. doi: 10.1136/bmjopen-2017-016358
62. Arnold AC, Raj SR. Orthostatic hypotension: a practical approach to investigation and management. Can J Cardiol. 2017;33:1725-1728. doi: 10.1016/j.cjca.2017.05.007
63. Alexander NB. Differential diagnosis of gait disorders in older adults. Clin Geriatr Med. 1996;12:689-703.
64. Baker JM. Gait disorders. Am J Med. 2018;131:602-607. doi: 10.1016/j.amjmed.2017.11.051
65. Cameron MH, Wagner JM. Gait abnormalities in multiple sclerosis: pathogenesis, evaluation, and advances in treatment. Curr Neurol Neurosci Rep. 2011;11:507-515. doi: 10.1007/s11910-011-0214-y
66. Chen C-L, Chen H-C, Tang SF-T, et al. Gait performance with compensatory adaptations in stroke patients with different degrees of motor recovery. Am J Phys Med Rehabil. 2003;82:925-935. doi: 10.1097/01.PHM.0000098040.13355.B5
67. Marsden J, Harris C. Cerebellar ataxia: pathophysiology and rehabilitation. Clin Rehabil. 2011;25:195-216. doi: 10.1177/0269215510382495
68. Mirek E, Filip M, W, et al. Three-dimensional trunk and lower limbs characteristics during gait in patients with Huntington’s disease. Front Neurosci. 2017;11:566. doi: 10.3389/fnins.2017.00566
69. Paramanandam V, Lizarraga KJ, Soh D, et al. Unusual gait disorders: a phenomenological approach and classification. Expert Rev Neurother. 2019;19:119-132. doi: 10.1080/14737175.2019.1562337
70. Sahyouni R, Goshtasbi K, Mahmoodi A, et al. Chronic subdural hematoma: a historical and clinical perspective. World Neurosurg. 2017;108:948-953. doi: 10.1016/j.wneu.2017.09.064
71. Talmud JD, Coffey R, Edemekong PF. Dix Hallpike maneuver. StatPearls [Internet]. StatPearls Publishing Updated September 5, 2021. Accessed December 6, 2021. www.ncbi.nlm.nih.gov/books/NBK459307/
72. Molnar FJ, Benjamin S, Hawkins SA, et al. One size does not fit all: choosing practical cognitive screening tools for your practice. J Am Geriatr Soc. 2020;68:2207-2213. doi: 10.1111/jgs.16713
73. Gillespie LD, Robertson MC, Gillespie WJ, et al. Interventions for preventing falls in older people living in the community. Cochrane Database Syst Rev. 2012:CD007146. doi: 10.1002/14651858.CD007146.pub3
74. Wongrakpanich S, Wongrakpanich A, Melhado K, Rangaswami J. A comprehensive review of non-steroidal anti-inflammatory drug use in the elderly. Aging Dis. 2018;9:143-150. doi: 10.14336/AD.2017.0306
75. Poe SS, Cvach M, Dawson PB, Straus H, Hill EE. The Johns Hopkins Fall Risk Assessment Tool: postimplementation evaluation. J Nurs Care Qual. 2007;22:293-298. doi: 10.1097/01.NCQ.0000290408.74027.39
76. Poe SS, Dawson PB, Cvach M, et al. The Johns Hopkins Fall Risk Assessment Tool: a study of reliability and validity. J Nurs Care Qual. 2018;33:10-19. doi: 10.1097/NCQ.0000000000000301
77. Klinkenberg WD, Potter P. Validity of the Johns Hopkins Fall Risk Assessment Tool for predicting falls on inpatient medicine services. J Nurs Care Qual. 2017;32:108-113. doi: 10.1097/NCQ.0000000000000210
78. Stapleton C, Hough P, Oldmeadow L, et al. Four-item fall risk screening tool for subacute and residential aged care: the first step in fall prevention. Australas J Ageing. 2009;28:139-143. doi: 10.1111/j.1741-6612.2009.00375.x
79. Cattelani L, Palumbo P, Palmerini L, et al. FRAT-up, a Web-based fall-risk assessment tool for elderly people living in the community. J Med Internet Res. 2015;17:e41. doi: 10.2196/jmir.4064
80. De Clercq H, Naudé A, Bornman J. Factors included in adult fall risk assessment tools (FRATs): a systematic review. Ageing Soc. 2020;41:2558-2582. doi: 10.1017/S0144686X2000046X
81. Yap G, Melder A. Accuracy of validated falls risk assessment tools and clinical judgement. Centre for Clinical Effectiveness, Monash Innovation and Quality. Monash Health. February 5, 2020. Accessed November 11, 2021. https://monashhealth.org/wp-content/uploads/2019/01/Rapid-Review_Falls-risk-tools-FINAL.pdf
82. Chittrakul J, Siviroj P, Sungkarat S, et al. Physical frailty and fall risk in community-dwelling older adults: a cross-sectional study. J Aging Res. 2020;2020:3964973. doi: 10.1155/2020/3964973
83. Hatcher VH, Galet C, Lilienthal M, et al. Association of clinical frailty scores with hospital readmission for falls after index admission for trauma-related injury. JAMA Netw Open. 2019;2:e1912409. doi: 10.1001/jamanetworkopen.2019.12409
84. Exercise and fall prevention programs. Government of Ontario Ministry of Health. Updated April 9, 2019. Accessed November 11. 2021. www.ontario.ca/page/exercise-and-falls-prevention-programs
85. Sherrington C, Fairhall NJ, Wallbank GK, et al. Exercise for preventing falls in older people living in the community. Cochrane Database Syst Rev. 2019;1:CD012424. doi: 10.1002/14651858.CD012424.pub2
86. Hopewell S, Copsey B, Nicolson P, et al. Multifactorial interventions for preventing falls in older people living in the community: a systematic review and meta-analysis of 41 trials and almost 20 000 participants. Br J Sports Med. 2020;54:1340-1350. doi: 10.1136/bjsports-2019-100732
87. Jafari Z, Kolb BE, Mohajerani MH. Age-related hearing loss and tinnitus, dementia risk, and auditory amplification outcomes. Ageing Res Rev. 2019;56:100963. doi: 10.1016/j.arr.2019.100963
88. Griffiths TD, Lad M, Kumar S, et al. How can hearing loss cause dementia? Neuron. 2020;108:401-412. doi: 10.1016/j.neuron.2020.08.003
89. Martini A, Castiglione A, Bovo R, et al. Aging, cognitive load, dementia and hearing loss. Audiol Neurootol. 2014;19(suppl 1):2-5. doi: 10.1159/000371593
90. Vitkovic J, Le C, Lee S-L, et al. The contribution of hearing and hearing loss to balance control. Audiol Neurootol. 2016;21:195-202. doi: 10.1159/000445100
91. Maheu M, Behtani L, Nooristani M, et al. Vestibular function modulates the benefit of hearing aids in people with hearing loss during static postural control. Ear Hear. 2019;40:1418-1424. doi: 10.1097/AUD.0000000000000720
92. Negahban H, Bavarsad Cheshmeh Ali M, Nassadj G. Effect of hearing aids on static balance function in elderly with hearing loss. Gait Posture. 2017;58:126-129. doi: 10.1016/j.gaitpost.2017.07.112
93. Mahmoudi E, Basu T, Langa K, et al. Can hearing aids delay time to diagnosis of dementia, depression, or falls in older adults? J Am Geriatr Soc. 2019;67:2362-2369. doi: 10.1111/jgs.16109
94. Paliwal Y, Slattum PW, Ratliff SM. Chronic health conditions as a risk factor for falls among the community-dwelling US older adults: a zero-inflated regression modeling approach. Biomed Res Int. 2017;2017:5146378. doi: 10.1155/2017/5146378
95. Deandrea S, Lucenteforte E, Bravi F, et al. Risk factors for falls in community-dwelling older people: a systematic review and meta-analysis. Epidemiology. 2010;21:658-668. doi: 10.1097/EDE.0b013e3181e89905
96. Ambrose AF, Paul G, Hausdorff JM. Risk factors for falls among older adults: a review of the literature. Maturitas. 2013;75:51-61. doi: 10.1016/j.maturitas.2013.02.009
97. Stevens M, Holman CD, Bennett N. Preventing falls in older people: impact of an intervention to reduce environmental hazards in the home. J Am Geriatr Soc. 2001;49:1442-1447. doi: 10.1046/j.1532-5415.2001.4911235.x
98. Clinical resources. Centers for Disease Control and Prevention STEADI-Older Adult Fall Prevention website. 2020. Accessed November 12, 2021. www.cdc.gov/steadi/materials.html
99. ; Grossman DC, Curry SJ, Owens DK, et al. Interventions to prevent falls in community-dwelling older adults: US Preventive Services Task Force recommendation statement. JAMA. 2018;319:1696-1704. doi: 10.1001/jama.2018.3097
1. Larocca NG. Impact of walking impairment in multiple sclerosis: perspectives of patients and care partners. Patient. 2011;4:189-201. doi: 10.2165/11591150-000000000-00000
2. TB, ZF, ES, et al. The relationship of balance disorders with falling, the effect of health problems, and social life on postural balance in the elderly living in a district in Turkey. Geriatrics (Basel). 2019;4:37. doi: 10.3390/geriatrics4020037
3. R, Sixt E, Landahl S, et al. Prevalence of dizziness and vertigo in an urban elderly population. J Vestib Res. 2004;14:47-52.
4. Sturnieks DL, St George R, Lord SR. Balance disorders in the elderly. Neurophysiol Clin. 2008;38:467-478. doi: 10.1016/j.neucli.2008.09.001
5. Boult C, Murphy J, Sloane P, et al. The relation of dizziness to functional decline. J Am Geriatr Soc. 1991;39:858-861. doi: 10.1111/j.1532-5415.1991.tb04451.x
6. Lin HW, Bhattacharyya N. Balance disorders in the elderly: epidemiology and functional impact. Laryngoscope. 2012;122:1858-1861. doi: 10.1002/lary.23376
7. Jia H, Lubetkin EI, DeMichele K, et al. Prevalence, risk factors, and burden of disease for falls and balance or walking problems among older adults in the U.S. Prev Med. 2019;126:105737. doi: 10.1016/j.ypmed.2019.05.025
8. Al-Momani M, Al-Momani F, Alghadir AH, et al. Factors related to gait and balance deficits in older adults. Clin Interv Aging. 2016;11:1043-1049. doi: 10.2147/CIA.S112282
9. Agrawal Y, Ward BK, Minor LB. Vestibular dysfunction: prevalence, impact and need for targeted treatment. J Vestib Res. 2013;23:113-117. doi: 10.3233/VES-130498
10. Altinsoy B, Erboy F, Tanriverdi H, et al. Syncope as a presentation of acute pulmonary embolism. Ther Clin Risk Manag. 2016;12:1023-1028. doi: 10.2147/TCRM.S105722
11. Belvederi Murri M, Triolo F, Coni A, et al. Instrumental assessment of balance and gait in depression: a systematic review. Psychiatry Res. 2020;284:112687. doi: 10.1016/j.psychres.2019.112687
12. Bhattacharyya N, Gubbels SP, Schwartz SR, et al. Clinical practice guideline: benign paroxysmal positional vertigo (update). Otolaryngol Head Neck Surg. 2017;156(suppl 3):S1-S47. doi: 10.1177/0194599816689667
13. S, Schwarm S, Grevenrath P, et al. Prevalence, aetiologies and prognosis of the symptom dizziness in primary care - a systematic review. BMC Fam Pract. 2018;19:33. doi: 10.1186/s12875-017-0695-0
14. Brouwer MC, Tunkel AR, van de Beek D. Epidemiology, diagnosis, and antimicrobial treatment of acute bacterial meningitis. Clin Microbiol Rev. 2010;23:467-492. doi: 10.1128/CMR.00070-09
15. Chad DA. Lumbar spinal stenosis. Neurol Clin. 2007;25:407-418. doi: 10.1016/j.ncl.2007.01.003
16. Conrad BP, Shokat MS, Abbasi AZ, et al. Associations of self-report measures with gait, range of motion and proprioception in patients with lumbar spinal stenosis. Gait Posture. 2013;38:987-992. doi: 10.1016/j.gaitpost.2013.05.010
17. de Luna RA, Mihailovic A, Nguyen AM, et al. The association of glaucomatous visual field loss and balance. Transl Vis Sci Technol. 2017;6:8. doi: 10.1167/tvst.6.3.8
18. DiSogra RM. Common aminoglycosides and platinum-based ototoxic drugs: cochlear/vestibular side effects and incidence. Semin Hear. 2019;40:104-107. doi: 10.1055/s-0039-1684040
19. Ebersbach G, Moreau C, Gandor F, et al. Clinical syndromes: parkinsonian gait. Mov Disord. 2013;28:1552-1559. doi: 10.1002/mds.25675
20. Evans WJ. Skeletal muscle loss: cachexia, sarcopenia, and inactivity. Am J Clin Nutr. 2010;91:1123S-1127S. doi: 10.3945/ajcn.2010.28608A
21. Filli L, Sutter T, Easthope CS, et al. Profiling walking dysfunction in multiple sclerosis: characterisation, classification and progression over time. Sci Rep. 2018;8:4984. doi: 10.1038/s41598-018-22676-0
22. Fritz NE, Kegelmeyer DA, Kloos AD, et al. Motor performance differentiates individuals with Lewy body dementia, Parkinson’s and Alzheimer’s disease. Gait Posture. 2016;50:1-7. doi: 10.1016/j.gaitpost.2016.08.009
23. Furman JM, Jacob RG. A clinical taxonomy of dizziness and anxiety in the otoneurological setting. J Anxiety Disord. 2001;15:9-26. doi: 10.1016/s0887-6185(00)00040-2
24. Furman JM, Marcus DA, Balaban CD. Vestibular migraine: clinical aspects and pathophysiology. Lancet Neurol. 2013;12:706-715. doi: 10.1016/S1474-4422(13)70107-8
25. Gerson LW, Jarjoura D, McCord G. Risk of imbalance in elderly people with impaired hearing or vision. Age Ageing. 1989;18:31-34. doi: 10.1093/ageing/18.1.31
26. Goudakos JK, Markou KD, Franco-Vidal V, et al. Corticosteroids in the treatment of vestibular neuritis: a systematic review and meta-analysis. Otol Neurotol. 2010;31:183-189. doi: 10.1097/MAO.0b013e3181ca843d
27. Green AD, CS, Bastian L, et al. Does this woman have osteoporosis? JAMA. 2004;292:2890-2900. doi: 10.1001/jama.292.23.2890
28. Hallemans A, Ortibus E, Meire F, et al. Low vision affects dynamic stability of gait. Gait Posture. 2010;32:547-551. doi: 10.1016/j.gaitpost.2010.07.018
29. Handelsman JA. Vestibulotoxicity: strategies for clinical diagnosis and rehabilitation. Int J Audiol. 2018;57(suppl 4):S99-S107. doi: 10.1080/14992027.2018.1468092
30. Head VA, Wakerley BR. Guillain-Barré syndrome in general practice: clinical features suggestive of early diagnosis. Br J Gen Pract. 2016;66:218-219. doi: 10.3399/bjgp16X684733
31. Helbostad JL, Vereijken B, Hesseberg K, et al. Altered vision destabilizes gait in older persons. Gait Posture. 2009;30:233-238. doi: 10.1016/j.gaitpost.2009.05.004
32. Hsu W-L, Chen C-Y, Tsauo J-Y, et al. Balance control in elderly people with osteoporosis. J Formos Med Assoc. 2014;113:334-339. doi: 10.1016/j.jfma.2014.02.006
33. Kim H-S, Yun DH, Yoo SD, et al. Balance control and knee osteoarthritis severity. Ann Rehabil Med. 2011;35:701-709. doi: 10.5535/arm.2011.35.5.701
34. Li L, Simonsick EM, Ferrucci L, et al. Hearing loss and gait speed among older adults in the United States. Gait Posture. 2013;38:25-29.
35. McKeith IG, Boeve BF, Dickson DW, et al. Diagnosis and management of dementia with Lewy bodies: fourth consensus report of the DLB Consortium. Neurology. 2017;89:88-100. doi: 10.1212/WNL.0000000000004058
36. Milner KA, Funk M, Richards S, et al. Gender differences in symptom presentation associated with coronary heart disease. Am J Cardiol. 1999;84:396-399. doi: 10.1016/s0002-9149(99)00322-7
37. Paillard T, F, Bru N, et al. The impact of time of day on the gait and balance control of Alzheimer’s patients. Chronobiol Int. 2016;33:161-168. doi: 10.3109/07420528.2015.1124885
38. Paldor I, Chen AS, Kaye AH. Growth rate of vestibular schwannoma. J Clin Neurosci. 2016;32:1-8. doi: 10.1016/j.jocn.2016.05.003
39. Picorelli AMA, Hatton AL, Gane EM, et al. Balance performance in older adults with hip osteoarthritis: a systematic review. Gait Posture. 2018;65:89-99. doi: 10.1016/j.gaitpost.2018.07.001
40. Raccagni C, Nonnekes J, Bloem BR, et al. Gait and postural disorders in parkinsonism: a clinical approach. J Neurol. 2020;267:3169-3176. doi: 10.1007/s00415-019-09382-1
41. Shanmugarajah PD, Hoggard N, Currie S, et al. Alcohol-related cerebellar degeneration: not all down to toxicity? Cerebellum Ataxias. 2016;3:17. doi: 10.1186/s40673-016-0055-1
42. Shih RY, Smirniotopoulos JG. Posterior fossa tumors in adult patients. Neuroimaging Clin N Am. 2016;26:493-510. doi: 10.1016/j.nic.2016.06.003
43. Smith EE. Clinical presentations and epidemiology of vascular dementia. Clin Sci (Lond). 2017;131:1059-1068. doi: 10.1042/CS20160607
44. Streur M, Ratcliffe SJ, Ball J, et al. Symptom clusters in adults with chronic atrial fibrillation. J Cardiovasc Nurs. 2017;32:296-303. doi: 10.1097/JCN.0000000000000344
45. Strupp M, M, JA. Peripheral vestibular disorders: an update. Curr Opin Neurol. 2019;32:165-173. doi: 10.1097/WCO.0000000000000649
46. Thompson TL, Amedee R. Vertigo: a review of common peripheral and central vestibular disorders. Ochsner J. 2009;9:20-26.
47. Timar B, Timar R, L, et al. The impact of diabetic neuropathy on balance and on the risk of falls in patients with type 2 diabetes mellitus: a cross-sectional study. PLoS One. 2016;11:e0154654. doi: 10.1371/journal.pone.0154654
48. Walls R, Hockberger R, Gausche-Hill M. Peripheral nerve disorders. In: Rosen’s Emergency Medicine: Concepts and Clinical Practice. 9th ed. Elsevier, Inc; 2018:1307-1320.
49. Watson JC, Dyck PJB. Peripheral neuropathy: a practical approach to diagnosis and symptom management. Mayo Clin Proc. 2015;90:940-951. doi: 10.1016/j.mayocp.2015.05.004
50. Whitfield KC, Bourassa MW, Adamolekun B, et al. Thiamine deficiency disorders: diagnosis, prevalence, and a roadmap for global control programs. Ann N Y Acad Sci. 2018;1430:3-43. doi: 10.1111/nyas.13919
51. Wu V, Sykes EA, Beyea MM, et al. Approach to Meniere disease management. Can Fam Physician. 2019;65:463-467.
52. Yew KS, Cheng EM. Diagnosis of acute stroke. Am Fam Physician. 2015;91:528-536.
53. Seppala LJ, van de Glind EMM, Daams JG, et al; . Fall-risk-increasing drugs: a systematic review and meta-analysis: III. Others. J Am Med Dir Assoc. 2018;19:372.e1-372.e8. doi: 10.1016/j.jamda.2017.12.099
54. ABIM Foundation. Choosing wisely. Choosing Wisely website. 2021. Accessed November 11. 2021. www.choosingwisely.org/
55. Berlie HD, Garwood CL. Diabetes medications related to an increased risk of falls and fall-related morbidity in the elderly. Ann Pharmacother. 2010;44:712-717. doi: 10.1345/aph.1M551
56. Hartikainen S, E, Louhivuori K. Medication as a risk factor for falls: critical systematic review. J Gerontol A Biol Sci Med Sci. 2007;62:1172-1181. doi: 10.1093/gerona/62.10.1172
57. Khanuja K, Joki J, Bachmann G, et al. Gait and balance in the aging population: Fall prevention using innovation and technology. Maturitas. 2018;110:51-56. doi: 10.1016/j.maturitas.2018.01.021
58. Salzman B. Gait and balance disorders in older adults. Am Fam Physician. 2010;82:61-68.
59. Zaninotto P, Huang YT, Di Gessa G, et al. Polypharmacy is a risk factor for hospital admission due to a fall: evidence from the English Longitudinal Study of Ageing. BMC Public Health. 2020;20:1804. doi: 10.1186/s12889-020-09920-x
60. Morin L, Calderon A, Welmer AK, et al. Polypharmacy and injurious falls in older adults: a nationwide nested case-control study. Clin Epidemiol. 2019;11:483-493. doi: 10.2147/CLEP.S201614
61. Dhalwani NN, Fahami R, Sathanapally H, et al. Association between polypharmacy and falls in older adults: a longitudinal study from England. BMJ Open. 2017;7:e016358. doi: 10.1136/bmjopen-2017-016358
62. Arnold AC, Raj SR. Orthostatic hypotension: a practical approach to investigation and management. Can J Cardiol. 2017;33:1725-1728. doi: 10.1016/j.cjca.2017.05.007
63. Alexander NB. Differential diagnosis of gait disorders in older adults. Clin Geriatr Med. 1996;12:689-703.
64. Baker JM. Gait disorders. Am J Med. 2018;131:602-607. doi: 10.1016/j.amjmed.2017.11.051
65. Cameron MH, Wagner JM. Gait abnormalities in multiple sclerosis: pathogenesis, evaluation, and advances in treatment. Curr Neurol Neurosci Rep. 2011;11:507-515. doi: 10.1007/s11910-011-0214-y
66. Chen C-L, Chen H-C, Tang SF-T, et al. Gait performance with compensatory adaptations in stroke patients with different degrees of motor recovery. Am J Phys Med Rehabil. 2003;82:925-935. doi: 10.1097/01.PHM.0000098040.13355.B5
67. Marsden J, Harris C. Cerebellar ataxia: pathophysiology and rehabilitation. Clin Rehabil. 2011;25:195-216. doi: 10.1177/0269215510382495
68. Mirek E, Filip M, W, et al. Three-dimensional trunk and lower limbs characteristics during gait in patients with Huntington’s disease. Front Neurosci. 2017;11:566. doi: 10.3389/fnins.2017.00566
69. Paramanandam V, Lizarraga KJ, Soh D, et al. Unusual gait disorders: a phenomenological approach and classification. Expert Rev Neurother. 2019;19:119-132. doi: 10.1080/14737175.2019.1562337
70. Sahyouni R, Goshtasbi K, Mahmoodi A, et al. Chronic subdural hematoma: a historical and clinical perspective. World Neurosurg. 2017;108:948-953. doi: 10.1016/j.wneu.2017.09.064
71. Talmud JD, Coffey R, Edemekong PF. Dix Hallpike maneuver. StatPearls [Internet]. StatPearls Publishing Updated September 5, 2021. Accessed December 6, 2021. www.ncbi.nlm.nih.gov/books/NBK459307/
72. Molnar FJ, Benjamin S, Hawkins SA, et al. One size does not fit all: choosing practical cognitive screening tools for your practice. J Am Geriatr Soc. 2020;68:2207-2213. doi: 10.1111/jgs.16713
73. Gillespie LD, Robertson MC, Gillespie WJ, et al. Interventions for preventing falls in older people living in the community. Cochrane Database Syst Rev. 2012:CD007146. doi: 10.1002/14651858.CD007146.pub3
74. Wongrakpanich S, Wongrakpanich A, Melhado K, Rangaswami J. A comprehensive review of non-steroidal anti-inflammatory drug use in the elderly. Aging Dis. 2018;9:143-150. doi: 10.14336/AD.2017.0306
75. Poe SS, Cvach M, Dawson PB, Straus H, Hill EE. The Johns Hopkins Fall Risk Assessment Tool: postimplementation evaluation. J Nurs Care Qual. 2007;22:293-298. doi: 10.1097/01.NCQ.0000290408.74027.39
76. Poe SS, Dawson PB, Cvach M, et al. The Johns Hopkins Fall Risk Assessment Tool: a study of reliability and validity. J Nurs Care Qual. 2018;33:10-19. doi: 10.1097/NCQ.0000000000000301
77. Klinkenberg WD, Potter P. Validity of the Johns Hopkins Fall Risk Assessment Tool for predicting falls on inpatient medicine services. J Nurs Care Qual. 2017;32:108-113. doi: 10.1097/NCQ.0000000000000210
78. Stapleton C, Hough P, Oldmeadow L, et al. Four-item fall risk screening tool for subacute and residential aged care: the first step in fall prevention. Australas J Ageing. 2009;28:139-143. doi: 10.1111/j.1741-6612.2009.00375.x
79. Cattelani L, Palumbo P, Palmerini L, et al. FRAT-up, a Web-based fall-risk assessment tool for elderly people living in the community. J Med Internet Res. 2015;17:e41. doi: 10.2196/jmir.4064
80. De Clercq H, Naudé A, Bornman J. Factors included in adult fall risk assessment tools (FRATs): a systematic review. Ageing Soc. 2020;41:2558-2582. doi: 10.1017/S0144686X2000046X
81. Yap G, Melder A. Accuracy of validated falls risk assessment tools and clinical judgement. Centre for Clinical Effectiveness, Monash Innovation and Quality. Monash Health. February 5, 2020. Accessed November 11, 2021. https://monashhealth.org/wp-content/uploads/2019/01/Rapid-Review_Falls-risk-tools-FINAL.pdf
82. Chittrakul J, Siviroj P, Sungkarat S, et al. Physical frailty and fall risk in community-dwelling older adults: a cross-sectional study. J Aging Res. 2020;2020:3964973. doi: 10.1155/2020/3964973
83. Hatcher VH, Galet C, Lilienthal M, et al. Association of clinical frailty scores with hospital readmission for falls after index admission for trauma-related injury. JAMA Netw Open. 2019;2:e1912409. doi: 10.1001/jamanetworkopen.2019.12409
84. Exercise and fall prevention programs. Government of Ontario Ministry of Health. Updated April 9, 2019. Accessed November 11. 2021. www.ontario.ca/page/exercise-and-falls-prevention-programs
85. Sherrington C, Fairhall NJ, Wallbank GK, et al. Exercise for preventing falls in older people living in the community. Cochrane Database Syst Rev. 2019;1:CD012424. doi: 10.1002/14651858.CD012424.pub2
86. Hopewell S, Copsey B, Nicolson P, et al. Multifactorial interventions for preventing falls in older people living in the community: a systematic review and meta-analysis of 41 trials and almost 20 000 participants. Br J Sports Med. 2020;54:1340-1350. doi: 10.1136/bjsports-2019-100732
87. Jafari Z, Kolb BE, Mohajerani MH. Age-related hearing loss and tinnitus, dementia risk, and auditory amplification outcomes. Ageing Res Rev. 2019;56:100963. doi: 10.1016/j.arr.2019.100963
88. Griffiths TD, Lad M, Kumar S, et al. How can hearing loss cause dementia? Neuron. 2020;108:401-412. doi: 10.1016/j.neuron.2020.08.003
89. Martini A, Castiglione A, Bovo R, et al. Aging, cognitive load, dementia and hearing loss. Audiol Neurootol. 2014;19(suppl 1):2-5. doi: 10.1159/000371593
90. Vitkovic J, Le C, Lee S-L, et al. The contribution of hearing and hearing loss to balance control. Audiol Neurootol. 2016;21:195-202. doi: 10.1159/000445100
91. Maheu M, Behtani L, Nooristani M, et al. Vestibular function modulates the benefit of hearing aids in people with hearing loss during static postural control. Ear Hear. 2019;40:1418-1424. doi: 10.1097/AUD.0000000000000720
92. Negahban H, Bavarsad Cheshmeh Ali M, Nassadj G. Effect of hearing aids on static balance function in elderly with hearing loss. Gait Posture. 2017;58:126-129. doi: 10.1016/j.gaitpost.2017.07.112
93. Mahmoudi E, Basu T, Langa K, et al. Can hearing aids delay time to diagnosis of dementia, depression, or falls in older adults? J Am Geriatr Soc. 2019;67:2362-2369. doi: 10.1111/jgs.16109
94. Paliwal Y, Slattum PW, Ratliff SM. Chronic health conditions as a risk factor for falls among the community-dwelling US older adults: a zero-inflated regression modeling approach. Biomed Res Int. 2017;2017:5146378. doi: 10.1155/2017/5146378
95. Deandrea S, Lucenteforte E, Bravi F, et al. Risk factors for falls in community-dwelling older people: a systematic review and meta-analysis. Epidemiology. 2010;21:658-668. doi: 10.1097/EDE.0b013e3181e89905
96. Ambrose AF, Paul G, Hausdorff JM. Risk factors for falls among older adults: a review of the literature. Maturitas. 2013;75:51-61. doi: 10.1016/j.maturitas.2013.02.009
97. Stevens M, Holman CD, Bennett N. Preventing falls in older people: impact of an intervention to reduce environmental hazards in the home. J Am Geriatr Soc. 2001;49:1442-1447. doi: 10.1046/j.1532-5415.2001.4911235.x
98. Clinical resources. Centers for Disease Control and Prevention STEADI-Older Adult Fall Prevention website. 2020. Accessed November 12, 2021. www.cdc.gov/steadi/materials.html
99. ; Grossman DC, Curry SJ, Owens DK, et al. Interventions to prevent falls in community-dwelling older adults: US Preventive Services Task Force recommendation statement. JAMA. 2018;319:1696-1704. doi: 10.1001/jama.2018.3097
PRACTICE RECOMMENDATIONS
› Utilize a falls-prevention program for older patients that focuses on balance and functional exercises. A
› Perform a multifactorial assessment of the risk of falls in older patients that includes optimizing medications, managing comorbidities, and addressing environmental hazards. B
› Use a systems-based approach to presentations of imbalance to direct your clinical judgment and highlight the need for referral to specialists for management and rehabilitation. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series