User login
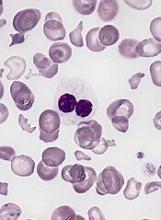
More abnormal cells linked to poorer ASCT outcomes in MDS
NEWPORT BEACH, CALIF. – Researchers say they’ve found an association between the percentage of cytogenetically abnormal cells at allogeneic stem cell transplant (ASCT) and posttransplant outcomes in patients with myelodysplastic syndromes (MDS).
Patients who had more than 60% cytogenetically abnormal cells at ASCT had significantly inferior overall survival (OS) and relapse-free survival (RFS), compared to patients with fewer abnormal cells.
Dipenkumar Modi, MD, of Barbara Ann Karmanos Cancer Institute at Wayne State University in Detroit, and his colleagues conducted this research and presented the results at the Acute Leukemia Forum of Hemedicus.
The researchers studied 109 adult MDS patients who underwent ASCT from January 2000 through December 2016. The patients were divided into three groups based on the percentage of cytogenetically abnormal cells at ASCT:
- Group 1 had less than 30% (n = 22)
- Group 2 had 30%-60% (n = 23)
- Group 3 had greater than 60% (n = 64).
Baseline characteristics were largely similar between the groups. However, patients in group 3 were significantly more likely than those in groups 1 and 2 to have del(5q) and monosomy 5+7 (P = .048).
Patients in group 1 had a significantly higher percentage of bone marrow transplants (as opposed to peripheral blood stem cell transplants) than patients in groups 2 and 3 (P = .039). And patients in group 1 had significantly fewer blasts at ASCT than patients in groups 2 and 3 (P = .011).
The researchers found no significant between-group differences in relapse and nonrelapse mortality, but there were significant differences in OS and RFS.
Patients in group 3 had inferior RFS compared to patients in group 1, which was the reference group. The hazard ratio (HR) was 2.503 (P = .013) in a univariable analysis and 2.196 (P = .049) in a multivariable analysis.
Group 3 also had inferior OS compared to group 1. The hazard ratio was 2.589 (P = .021) in a univariable analysis and 2.478 (P = .040) in a multivariable analysis.
There was no significant difference in RFS or OS between groups 1 and 2. The HR for RFS in group 2 was 1.879 (P = .148) in a univariable analysis and 1.365 (P = .506) in a multivariable analysis. The HR for OS was 1.997 (P = .155) and 1.413 (P = .511), respectively.
Dr. Modi said these results suggest patients with greater than 60% cytogenetically abnormal cells at ASCT should be monitored more closely after transplant, and their immunosuppressive medication should be tapered as soon as possible.
Dr. Modi and his colleagues reported having no conflicts of interest relevant to this research.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
NEWPORT BEACH, CALIF. – Researchers say they’ve found an association between the percentage of cytogenetically abnormal cells at allogeneic stem cell transplant (ASCT) and posttransplant outcomes in patients with myelodysplastic syndromes (MDS).
Patients who had more than 60% cytogenetically abnormal cells at ASCT had significantly inferior overall survival (OS) and relapse-free survival (RFS), compared to patients with fewer abnormal cells.
Dipenkumar Modi, MD, of Barbara Ann Karmanos Cancer Institute at Wayne State University in Detroit, and his colleagues conducted this research and presented the results at the Acute Leukemia Forum of Hemedicus.
The researchers studied 109 adult MDS patients who underwent ASCT from January 2000 through December 2016. The patients were divided into three groups based on the percentage of cytogenetically abnormal cells at ASCT:
- Group 1 had less than 30% (n = 22)
- Group 2 had 30%-60% (n = 23)
- Group 3 had greater than 60% (n = 64).
Baseline characteristics were largely similar between the groups. However, patients in group 3 were significantly more likely than those in groups 1 and 2 to have del(5q) and monosomy 5+7 (P = .048).
Patients in group 1 had a significantly higher percentage of bone marrow transplants (as opposed to peripheral blood stem cell transplants) than patients in groups 2 and 3 (P = .039). And patients in group 1 had significantly fewer blasts at ASCT than patients in groups 2 and 3 (P = .011).
The researchers found no significant between-group differences in relapse and nonrelapse mortality, but there were significant differences in OS and RFS.
Patients in group 3 had inferior RFS compared to patients in group 1, which was the reference group. The hazard ratio (HR) was 2.503 (P = .013) in a univariable analysis and 2.196 (P = .049) in a multivariable analysis.
Group 3 also had inferior OS compared to group 1. The hazard ratio was 2.589 (P = .021) in a univariable analysis and 2.478 (P = .040) in a multivariable analysis.
There was no significant difference in RFS or OS between groups 1 and 2. The HR for RFS in group 2 was 1.879 (P = .148) in a univariable analysis and 1.365 (P = .506) in a multivariable analysis. The HR for OS was 1.997 (P = .155) and 1.413 (P = .511), respectively.
Dr. Modi said these results suggest patients with greater than 60% cytogenetically abnormal cells at ASCT should be monitored more closely after transplant, and their immunosuppressive medication should be tapered as soon as possible.
Dr. Modi and his colleagues reported having no conflicts of interest relevant to this research.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
NEWPORT BEACH, CALIF. – Researchers say they’ve found an association between the percentage of cytogenetically abnormal cells at allogeneic stem cell transplant (ASCT) and posttransplant outcomes in patients with myelodysplastic syndromes (MDS).
Patients who had more than 60% cytogenetically abnormal cells at ASCT had significantly inferior overall survival (OS) and relapse-free survival (RFS), compared to patients with fewer abnormal cells.
Dipenkumar Modi, MD, of Barbara Ann Karmanos Cancer Institute at Wayne State University in Detroit, and his colleagues conducted this research and presented the results at the Acute Leukemia Forum of Hemedicus.
The researchers studied 109 adult MDS patients who underwent ASCT from January 2000 through December 2016. The patients were divided into three groups based on the percentage of cytogenetically abnormal cells at ASCT:
- Group 1 had less than 30% (n = 22)
- Group 2 had 30%-60% (n = 23)
- Group 3 had greater than 60% (n = 64).
Baseline characteristics were largely similar between the groups. However, patients in group 3 were significantly more likely than those in groups 1 and 2 to have del(5q) and monosomy 5+7 (P = .048).
Patients in group 1 had a significantly higher percentage of bone marrow transplants (as opposed to peripheral blood stem cell transplants) than patients in groups 2 and 3 (P = .039). And patients in group 1 had significantly fewer blasts at ASCT than patients in groups 2 and 3 (P = .011).
The researchers found no significant between-group differences in relapse and nonrelapse mortality, but there were significant differences in OS and RFS.
Patients in group 3 had inferior RFS compared to patients in group 1, which was the reference group. The hazard ratio (HR) was 2.503 (P = .013) in a univariable analysis and 2.196 (P = .049) in a multivariable analysis.
Group 3 also had inferior OS compared to group 1. The hazard ratio was 2.589 (P = .021) in a univariable analysis and 2.478 (P = .040) in a multivariable analysis.
There was no significant difference in RFS or OS between groups 1 and 2. The HR for RFS in group 2 was 1.879 (P = .148) in a univariable analysis and 1.365 (P = .506) in a multivariable analysis. The HR for OS was 1.997 (P = .155) and 1.413 (P = .511), respectively.
Dr. Modi said these results suggest patients with greater than 60% cytogenetically abnormal cells at ASCT should be monitored more closely after transplant, and their immunosuppressive medication should be tapered as soon as possible.
Dr. Modi and his colleagues reported having no conflicts of interest relevant to this research.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
REPORTING FROM ALF 2019
Model inspired by Netflix, Amazon may help guide MDS treatment
NEWPORT BEACH, CALIF. — A model that mimics the recommender system used by Netflix and Amazon can help predict outcomes of lenalidomide treatment in patients with non–deletion 5q (non-del[5q]) myelodysplastic syndromes (MDS), according to new research.
The model was used to identify genomic biomarkers that were associated with resistance or response to lenalidomide. Researchers found these associations in 39% of patients with non-del(5q) MDS, and the model predicted response or resistance with 82% accuracy.
Yazan Madanat, MD, of the Cleveland Clinic, and his colleagues presented these findings at the Acute Leukemia Forum of Hemedicus.
Dr. Madanat explained that his group’s model is similar to the recommender system used by Netflix and Amazon, which makes suggestions for new products based on customers’ past behavior. Dr. Madanat and his colleagues used their model to show that patients with certain molecular or cytogenetic abnormalities are likely to respond or not respond to lenalidomide.
The researchers began by looking at 139 patients who had received at least two cycles of lenalidomide treatment. There were 118 patients with MDS, and 108 who had received lenalidomide monotherapy. However, the team focused on the 100 patients who had non-del(5q) MDS, 58 of whom had normal karyotype (NK) and 19 of whom had complex karyotype (CK).
The model revealed several combinations of genomic/cytogenetic abnormalities that could predict resistance to lenalidomide, including the following:
- DNMT3A and SF3B1
- EZH2 and NK
- ASXL1, TET2, and NK
- STAG2, IDH1/2, and NK
- TP53, del(5q), and CK
- BCOR/BCORL1 and NK
- JAK2, TET2, and NK
- U2AF1, +/– ETV6, and NK
However, only the following two combinations could predict response to lenalidomide:
- DDX41 and NK
- MECOM and KDM6A/B
These combinations could be applied to 39% of the patients with non-del(5q) MDS, and the model predicted response or resistance to lenalidomide with 82% accuracy.
Although the biomarkers were found in only a subset of patients, Dr. Madanat said these findings may help physicians tailor therapy for MDS patients, given the high level of accuracy the researchers observed.
“It’s really important to validate the results in a prospective manner and to ensure that we’re able to apply them clinically and potentially change the way we’re treating our patients,” he added.
Dr. Madanat and his colleagues reported having no relevant conflicts of interest.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
NEWPORT BEACH, CALIF. — A model that mimics the recommender system used by Netflix and Amazon can help predict outcomes of lenalidomide treatment in patients with non–deletion 5q (non-del[5q]) myelodysplastic syndromes (MDS), according to new research.
The model was used to identify genomic biomarkers that were associated with resistance or response to lenalidomide. Researchers found these associations in 39% of patients with non-del(5q) MDS, and the model predicted response or resistance with 82% accuracy.
Yazan Madanat, MD, of the Cleveland Clinic, and his colleagues presented these findings at the Acute Leukemia Forum of Hemedicus.
Dr. Madanat explained that his group’s model is similar to the recommender system used by Netflix and Amazon, which makes suggestions for new products based on customers’ past behavior. Dr. Madanat and his colleagues used their model to show that patients with certain molecular or cytogenetic abnormalities are likely to respond or not respond to lenalidomide.
The researchers began by looking at 139 patients who had received at least two cycles of lenalidomide treatment. There were 118 patients with MDS, and 108 who had received lenalidomide monotherapy. However, the team focused on the 100 patients who had non-del(5q) MDS, 58 of whom had normal karyotype (NK) and 19 of whom had complex karyotype (CK).
The model revealed several combinations of genomic/cytogenetic abnormalities that could predict resistance to lenalidomide, including the following:
- DNMT3A and SF3B1
- EZH2 and NK
- ASXL1, TET2, and NK
- STAG2, IDH1/2, and NK
- TP53, del(5q), and CK
- BCOR/BCORL1 and NK
- JAK2, TET2, and NK
- U2AF1, +/– ETV6, and NK
However, only the following two combinations could predict response to lenalidomide:
- DDX41 and NK
- MECOM and KDM6A/B
These combinations could be applied to 39% of the patients with non-del(5q) MDS, and the model predicted response or resistance to lenalidomide with 82% accuracy.
Although the biomarkers were found in only a subset of patients, Dr. Madanat said these findings may help physicians tailor therapy for MDS patients, given the high level of accuracy the researchers observed.
“It’s really important to validate the results in a prospective manner and to ensure that we’re able to apply them clinically and potentially change the way we’re treating our patients,” he added.
Dr. Madanat and his colleagues reported having no relevant conflicts of interest.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
NEWPORT BEACH, CALIF. — A model that mimics the recommender system used by Netflix and Amazon can help predict outcomes of lenalidomide treatment in patients with non–deletion 5q (non-del[5q]) myelodysplastic syndromes (MDS), according to new research.
The model was used to identify genomic biomarkers that were associated with resistance or response to lenalidomide. Researchers found these associations in 39% of patients with non-del(5q) MDS, and the model predicted response or resistance with 82% accuracy.
Yazan Madanat, MD, of the Cleveland Clinic, and his colleagues presented these findings at the Acute Leukemia Forum of Hemedicus.
Dr. Madanat explained that his group’s model is similar to the recommender system used by Netflix and Amazon, which makes suggestions for new products based on customers’ past behavior. Dr. Madanat and his colleagues used their model to show that patients with certain molecular or cytogenetic abnormalities are likely to respond or not respond to lenalidomide.
The researchers began by looking at 139 patients who had received at least two cycles of lenalidomide treatment. There were 118 patients with MDS, and 108 who had received lenalidomide monotherapy. However, the team focused on the 100 patients who had non-del(5q) MDS, 58 of whom had normal karyotype (NK) and 19 of whom had complex karyotype (CK).
The model revealed several combinations of genomic/cytogenetic abnormalities that could predict resistance to lenalidomide, including the following:
- DNMT3A and SF3B1
- EZH2 and NK
- ASXL1, TET2, and NK
- STAG2, IDH1/2, and NK
- TP53, del(5q), and CK
- BCOR/BCORL1 and NK
- JAK2, TET2, and NK
- U2AF1, +/– ETV6, and NK
However, only the following two combinations could predict response to lenalidomide:
- DDX41 and NK
- MECOM and KDM6A/B
These combinations could be applied to 39% of the patients with non-del(5q) MDS, and the model predicted response or resistance to lenalidomide with 82% accuracy.
Although the biomarkers were found in only a subset of patients, Dr. Madanat said these findings may help physicians tailor therapy for MDS patients, given the high level of accuracy the researchers observed.
“It’s really important to validate the results in a prospective manner and to ensure that we’re able to apply them clinically and potentially change the way we’re treating our patients,” he added.
Dr. Madanat and his colleagues reported having no relevant conflicts of interest.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
REPORTING FROM ALF 2019
Sorafenib plus GCLAM held safe in AML, MDS phase-1 study
NEWPORT BEACH, CALIF. – A five-drug regimen was deemed safe in patients with newly diagnosed acute myeloid leukemia (AML) or high-risk myelodysplastic syndromes (MDS), and it appeared to be effective regardless of patients’ FLT3 status.
Researchers tested this regimen – sorafenib plus granulocyte colony–stimulating factor (G-CSF), cladribine, high-dose cytarabine, and mitoxantrone (GCLAM) – in a phase 1 trial.
Kelsey-Leigh Garcia, a clinical research coordinator at Seattle Cancer Care Alliance, and her colleagues presented the results at the Acute Leukemia Forum of Hemedicus.
“The background for doing this study was our institutional results of GCLAM [Leukemia. 2018 Nov;32(11):2352-62] that showed a higher minimal residual disease–negative complete response rate than 7+3 [cytarabine continuously for 7 days, along with short infusions of an anthracycline on each of the first 3 days] and an international study by Röllig that showed the addition of sorafenib to 7+3 increased event-free survival versus [7+3 and] placebo [Lancet Oncol. 2015 Dec;16(16):1691-9],” Ms. Garcia said.
“GCLAM is the standard backbone at our institution, and we wanted to ask the question, ‘If we add sorafenib, can this improve upon the results of GCLAM?’ ” said Anna Halpern, MD, a hematologist-oncologist at the University of Washington, Seattle and principal investigator of the phase 1 trial.
The trial (NCT02728050) included 47 patients, 39 with AML and 8 with MDS. Patients were aged 60 years or younger and had a median age of 48. They had a median treatment-related mortality score of 1.76 (range, 0.19-12.26). A total of 11 patients (23%) had FLT3-ITD, and 4 (9%) had FLT3-TKD.
Treatment and toxicity
For induction, patients received G-CSF at 5 mcg/kg on days 0-5, cladribine at 5 mg/m2 on days 1-5, and cytarabine at 2 g/m2 on days 1-5. Mitoxantrone was given at 10 mg/m2, 12 mg/m2, 15 mg/m2, or 18 mg/m2 on days 1-3. Sorafenib was given at 200 mg twice daily, 400 mg in the morning and 200 mg in the afternoon, or 400 mg b.i.d. on days 10-19.
For consolidation, patients could receive up to four cycles of G-CSF, cladribine, and cytarabine plus sorafenib on days 8-27. Patients who did not proceed to transplant could receive 12 months of sorafenib as maintenance therapy.
There were four dose-limiting toxicities.
- Grade 4 intracranial hemorrhage with mitoxantrone at 12 mg/m2 and sorafenib at 200 mg b.i.d.
- Grade 4 prolonged count recovery with mitoxantrone at 15 mg/m2 and sorafenib at 200 mg b.i.d.
- Grade 4 sepsis, Sweet syndrome, and Bell’s palsy with mitoxantrone at 18 mg/m2 and sorafenib at 200 mg b.i.d.
- Grade 3 cardiomyopathy and acute pericarditis with mitoxantrone at 18 mg/m2 and sorafenib at 400 mg b.i.d.
However, these toxicities did not define the maximum-tolerated dose. Therefore, the recommended phase 2 dose of mitoxantrone is 18 mg/m2, and the recommended phase 2 dose of sorafenib is 400 mg b.i.d.
There were no grade 5 treatment-related adverse events. Grade 3 events included febrile neutropenia (90%), maculopapular rash (20%), infections (10%), hand-foot syndrome (2%), and diarrhea (1%). Grade 4 events included sepsis, intracranial hemorrhage, and oral mucositis (all 1%).
Response and survival
Among the 46 evaluable patients, 83% achieved a complete response, 78% had a minimal residual disease–negative complete response, and 4% had a minimal residual disease–negative complete response with incomplete count recovery. A morphological leukemia-free state was achieved by 4% of patients, and 8% had resistant disease.
Fifty-nine percent of patients went on to transplant. The median overall survival had not been reached at a median follow-up of 10 months.
The researchers compared outcomes in this trial with outcomes in a cohort of patients who had received GCLAM alone, and there were no significant differences in overall survival or event-free survival.
“The trial wasn’t powered, necessarily, for efficacy, but we compared these results to our historical cohort of medically matched and age-matched patients treated with GCLAM alone and, so far, found no differences in survival between the two groups,” Dr. Halpern said.
She noted, however, that follow-up was short in the sorafenib trial, and it included patients treated with all dose levels of sorafenib and mitoxantrone.
A phase 2 study of sorafenib plus GCLAM in newly diagnosed AML or high-risk MDS is now underway.
Dr. Halpern and Ms. Garcia reported that they had no conflicts of interest. The phase 1 trial was sponsored by the University of Washington in collaboration with the National Cancer Institute, and funding was provided by Bayer.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
NEWPORT BEACH, CALIF. – A five-drug regimen was deemed safe in patients with newly diagnosed acute myeloid leukemia (AML) or high-risk myelodysplastic syndromes (MDS), and it appeared to be effective regardless of patients’ FLT3 status.
Researchers tested this regimen – sorafenib plus granulocyte colony–stimulating factor (G-CSF), cladribine, high-dose cytarabine, and mitoxantrone (GCLAM) – in a phase 1 trial.
Kelsey-Leigh Garcia, a clinical research coordinator at Seattle Cancer Care Alliance, and her colleagues presented the results at the Acute Leukemia Forum of Hemedicus.
“The background for doing this study was our institutional results of GCLAM [Leukemia. 2018 Nov;32(11):2352-62] that showed a higher minimal residual disease–negative complete response rate than 7+3 [cytarabine continuously for 7 days, along with short infusions of an anthracycline on each of the first 3 days] and an international study by Röllig that showed the addition of sorafenib to 7+3 increased event-free survival versus [7+3 and] placebo [Lancet Oncol. 2015 Dec;16(16):1691-9],” Ms. Garcia said.
“GCLAM is the standard backbone at our institution, and we wanted to ask the question, ‘If we add sorafenib, can this improve upon the results of GCLAM?’ ” said Anna Halpern, MD, a hematologist-oncologist at the University of Washington, Seattle and principal investigator of the phase 1 trial.
The trial (NCT02728050) included 47 patients, 39 with AML and 8 with MDS. Patients were aged 60 years or younger and had a median age of 48. They had a median treatment-related mortality score of 1.76 (range, 0.19-12.26). A total of 11 patients (23%) had FLT3-ITD, and 4 (9%) had FLT3-TKD.
Treatment and toxicity
For induction, patients received G-CSF at 5 mcg/kg on days 0-5, cladribine at 5 mg/m2 on days 1-5, and cytarabine at 2 g/m2 on days 1-5. Mitoxantrone was given at 10 mg/m2, 12 mg/m2, 15 mg/m2, or 18 mg/m2 on days 1-3. Sorafenib was given at 200 mg twice daily, 400 mg in the morning and 200 mg in the afternoon, or 400 mg b.i.d. on days 10-19.
For consolidation, patients could receive up to four cycles of G-CSF, cladribine, and cytarabine plus sorafenib on days 8-27. Patients who did not proceed to transplant could receive 12 months of sorafenib as maintenance therapy.
There were four dose-limiting toxicities.
- Grade 4 intracranial hemorrhage with mitoxantrone at 12 mg/m2 and sorafenib at 200 mg b.i.d.
- Grade 4 prolonged count recovery with mitoxantrone at 15 mg/m2 and sorafenib at 200 mg b.i.d.
- Grade 4 sepsis, Sweet syndrome, and Bell’s palsy with mitoxantrone at 18 mg/m2 and sorafenib at 200 mg b.i.d.
- Grade 3 cardiomyopathy and acute pericarditis with mitoxantrone at 18 mg/m2 and sorafenib at 400 mg b.i.d.
However, these toxicities did not define the maximum-tolerated dose. Therefore, the recommended phase 2 dose of mitoxantrone is 18 mg/m2, and the recommended phase 2 dose of sorafenib is 400 mg b.i.d.
There were no grade 5 treatment-related adverse events. Grade 3 events included febrile neutropenia (90%), maculopapular rash (20%), infections (10%), hand-foot syndrome (2%), and diarrhea (1%). Grade 4 events included sepsis, intracranial hemorrhage, and oral mucositis (all 1%).
Response and survival
Among the 46 evaluable patients, 83% achieved a complete response, 78% had a minimal residual disease–negative complete response, and 4% had a minimal residual disease–negative complete response with incomplete count recovery. A morphological leukemia-free state was achieved by 4% of patients, and 8% had resistant disease.
Fifty-nine percent of patients went on to transplant. The median overall survival had not been reached at a median follow-up of 10 months.
The researchers compared outcomes in this trial with outcomes in a cohort of patients who had received GCLAM alone, and there were no significant differences in overall survival or event-free survival.
“The trial wasn’t powered, necessarily, for efficacy, but we compared these results to our historical cohort of medically matched and age-matched patients treated with GCLAM alone and, so far, found no differences in survival between the two groups,” Dr. Halpern said.
She noted, however, that follow-up was short in the sorafenib trial, and it included patients treated with all dose levels of sorafenib and mitoxantrone.
A phase 2 study of sorafenib plus GCLAM in newly diagnosed AML or high-risk MDS is now underway.
Dr. Halpern and Ms. Garcia reported that they had no conflicts of interest. The phase 1 trial was sponsored by the University of Washington in collaboration with the National Cancer Institute, and funding was provided by Bayer.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
NEWPORT BEACH, CALIF. – A five-drug regimen was deemed safe in patients with newly diagnosed acute myeloid leukemia (AML) or high-risk myelodysplastic syndromes (MDS), and it appeared to be effective regardless of patients’ FLT3 status.
Researchers tested this regimen – sorafenib plus granulocyte colony–stimulating factor (G-CSF), cladribine, high-dose cytarabine, and mitoxantrone (GCLAM) – in a phase 1 trial.
Kelsey-Leigh Garcia, a clinical research coordinator at Seattle Cancer Care Alliance, and her colleagues presented the results at the Acute Leukemia Forum of Hemedicus.
“The background for doing this study was our institutional results of GCLAM [Leukemia. 2018 Nov;32(11):2352-62] that showed a higher minimal residual disease–negative complete response rate than 7+3 [cytarabine continuously for 7 days, along with short infusions of an anthracycline on each of the first 3 days] and an international study by Röllig that showed the addition of sorafenib to 7+3 increased event-free survival versus [7+3 and] placebo [Lancet Oncol. 2015 Dec;16(16):1691-9],” Ms. Garcia said.
“GCLAM is the standard backbone at our institution, and we wanted to ask the question, ‘If we add sorafenib, can this improve upon the results of GCLAM?’ ” said Anna Halpern, MD, a hematologist-oncologist at the University of Washington, Seattle and principal investigator of the phase 1 trial.
The trial (NCT02728050) included 47 patients, 39 with AML and 8 with MDS. Patients were aged 60 years or younger and had a median age of 48. They had a median treatment-related mortality score of 1.76 (range, 0.19-12.26). A total of 11 patients (23%) had FLT3-ITD, and 4 (9%) had FLT3-TKD.
Treatment and toxicity
For induction, patients received G-CSF at 5 mcg/kg on days 0-5, cladribine at 5 mg/m2 on days 1-5, and cytarabine at 2 g/m2 on days 1-5. Mitoxantrone was given at 10 mg/m2, 12 mg/m2, 15 mg/m2, or 18 mg/m2 on days 1-3. Sorafenib was given at 200 mg twice daily, 400 mg in the morning and 200 mg in the afternoon, or 400 mg b.i.d. on days 10-19.
For consolidation, patients could receive up to four cycles of G-CSF, cladribine, and cytarabine plus sorafenib on days 8-27. Patients who did not proceed to transplant could receive 12 months of sorafenib as maintenance therapy.
There were four dose-limiting toxicities.
- Grade 4 intracranial hemorrhage with mitoxantrone at 12 mg/m2 and sorafenib at 200 mg b.i.d.
- Grade 4 prolonged count recovery with mitoxantrone at 15 mg/m2 and sorafenib at 200 mg b.i.d.
- Grade 4 sepsis, Sweet syndrome, and Bell’s palsy with mitoxantrone at 18 mg/m2 and sorafenib at 200 mg b.i.d.
- Grade 3 cardiomyopathy and acute pericarditis with mitoxantrone at 18 mg/m2 and sorafenib at 400 mg b.i.d.
However, these toxicities did not define the maximum-tolerated dose. Therefore, the recommended phase 2 dose of mitoxantrone is 18 mg/m2, and the recommended phase 2 dose of sorafenib is 400 mg b.i.d.
There were no grade 5 treatment-related adverse events. Grade 3 events included febrile neutropenia (90%), maculopapular rash (20%), infections (10%), hand-foot syndrome (2%), and diarrhea (1%). Grade 4 events included sepsis, intracranial hemorrhage, and oral mucositis (all 1%).
Response and survival
Among the 46 evaluable patients, 83% achieved a complete response, 78% had a minimal residual disease–negative complete response, and 4% had a minimal residual disease–negative complete response with incomplete count recovery. A morphological leukemia-free state was achieved by 4% of patients, and 8% had resistant disease.
Fifty-nine percent of patients went on to transplant. The median overall survival had not been reached at a median follow-up of 10 months.
The researchers compared outcomes in this trial with outcomes in a cohort of patients who had received GCLAM alone, and there were no significant differences in overall survival or event-free survival.
“The trial wasn’t powered, necessarily, for efficacy, but we compared these results to our historical cohort of medically matched and age-matched patients treated with GCLAM alone and, so far, found no differences in survival between the two groups,” Dr. Halpern said.
She noted, however, that follow-up was short in the sorafenib trial, and it included patients treated with all dose levels of sorafenib and mitoxantrone.
A phase 2 study of sorafenib plus GCLAM in newly diagnosed AML or high-risk MDS is now underway.
Dr. Halpern and Ms. Garcia reported that they had no conflicts of interest. The phase 1 trial was sponsored by the University of Washington in collaboration with the National Cancer Institute, and funding was provided by Bayer.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
REPORTING FROM ALF 2019
Chemo for solid tumors and risk of tMDS/AML
Chemotherapy for solid tumors is associated with an increased risk of therapy-related myelodysplastic syndromes or acute myeloid leukemia (tMDS/AML), according to a retrospective analysis.
Long-term, population-based cohort data showed the risk of tMDS/AML was significantly elevated after chemotherapy for 22 solid tumor types.
The relative risk of tMDS/AML was 1.5- to 39.0-fold greater among patients treated for these tumors than among the general population.
Lindsay M. Morton, PhD, of the National Institutes of Health in Rockville, Maryland, and her colleagues reported these findings in JAMA Oncology.
“We undertook an investigation to quantify tMDS/AML risks after chemotherapy for solid tumors in the modern treatment era, 2000-2014, using United States cancer registry data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results Program,” the investigators wrote.
They retrospectively analyzed data from 1619 patients with tMDS/AML who were diagnosed with an initial primary solid tumor from 2000 to 2013.
Patients were given initial chemotherapy and lived for at least 1 year after treatment. Subsequently, Dr. Morton and her colleagues linked patient database records with Medicare insurance claim information to confirm the accuracy of chemotherapy data.
“Because registry data do not include treatment details, we used an alternative database to provide descriptive information on population-based patterns of chemotherapeutic drug use,” the investigators noted.
The team found the risk of developing tMDS/AML was significantly increased following chemotherapy administration for 22 of 23 solid tumor types, excluding colon cancer.
The standardized incidence ratio (SIR) for tMDS/AML ranged from 1.5 to 39.0, and the excess absolute risk (EAR) ranged from 1.4 to 23.6 cases per 10,000 person-years.
SIRs were greatest in patients who received chemotherapy for malignancy of the bone (SIR=39.0, EAR=23.6), testis (SIR, 12.3, EAR=4.4), soft tissue (SIR=10.4, EAR=12.6), fallopian tube (SIR=8.7, EAR=16.0), small cell lung (SIR=8.1, EAR=19.9), peritoneum (SIR=7.5, EAR=15.8), brain or central nervous system (SIR=7.2, EAR=6.0), and ovary (SIR=5.8, EAR=8.2).
The investigators also found that patients who were given chemotherapy at a young age had the highest risk of developing tMDS/AML.
“For patients treated with chemotherapy at the present time, approximately three-quarters of tMDS/AML cases expected to occur within the next 5 years will be attributable to chemotherapy,” the investigators said.
They acknowledged a key limitation of this study was the limited data on patient-specific chemotherapy and dosing information. Given these limitations, Dr. Morton and her colleagues said, “the exact magnitude of our risk estimates, including the proportions of excess cases, should therefore be interpreted cautiously.”
This study was supported by the Intramural Research Program of the National Institutes of Health, National Cancer Institute, and the California Department of Public Health. The authors reported no conflicts of interest.
Chemotherapy for solid tumors is associated with an increased risk of therapy-related myelodysplastic syndromes or acute myeloid leukemia (tMDS/AML), according to a retrospective analysis.
Long-term, population-based cohort data showed the risk of tMDS/AML was significantly elevated after chemotherapy for 22 solid tumor types.
The relative risk of tMDS/AML was 1.5- to 39.0-fold greater among patients treated for these tumors than among the general population.
Lindsay M. Morton, PhD, of the National Institutes of Health in Rockville, Maryland, and her colleagues reported these findings in JAMA Oncology.
“We undertook an investigation to quantify tMDS/AML risks after chemotherapy for solid tumors in the modern treatment era, 2000-2014, using United States cancer registry data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results Program,” the investigators wrote.
They retrospectively analyzed data from 1619 patients with tMDS/AML who were diagnosed with an initial primary solid tumor from 2000 to 2013.
Patients were given initial chemotherapy and lived for at least 1 year after treatment. Subsequently, Dr. Morton and her colleagues linked patient database records with Medicare insurance claim information to confirm the accuracy of chemotherapy data.
“Because registry data do not include treatment details, we used an alternative database to provide descriptive information on population-based patterns of chemotherapeutic drug use,” the investigators noted.
The team found the risk of developing tMDS/AML was significantly increased following chemotherapy administration for 22 of 23 solid tumor types, excluding colon cancer.
The standardized incidence ratio (SIR) for tMDS/AML ranged from 1.5 to 39.0, and the excess absolute risk (EAR) ranged from 1.4 to 23.6 cases per 10,000 person-years.
SIRs were greatest in patients who received chemotherapy for malignancy of the bone (SIR=39.0, EAR=23.6), testis (SIR, 12.3, EAR=4.4), soft tissue (SIR=10.4, EAR=12.6), fallopian tube (SIR=8.7, EAR=16.0), small cell lung (SIR=8.1, EAR=19.9), peritoneum (SIR=7.5, EAR=15.8), brain or central nervous system (SIR=7.2, EAR=6.0), and ovary (SIR=5.8, EAR=8.2).
The investigators also found that patients who were given chemotherapy at a young age had the highest risk of developing tMDS/AML.
“For patients treated with chemotherapy at the present time, approximately three-quarters of tMDS/AML cases expected to occur within the next 5 years will be attributable to chemotherapy,” the investigators said.
They acknowledged a key limitation of this study was the limited data on patient-specific chemotherapy and dosing information. Given these limitations, Dr. Morton and her colleagues said, “the exact magnitude of our risk estimates, including the proportions of excess cases, should therefore be interpreted cautiously.”
This study was supported by the Intramural Research Program of the National Institutes of Health, National Cancer Institute, and the California Department of Public Health. The authors reported no conflicts of interest.
Chemotherapy for solid tumors is associated with an increased risk of therapy-related myelodysplastic syndromes or acute myeloid leukemia (tMDS/AML), according to a retrospective analysis.
Long-term, population-based cohort data showed the risk of tMDS/AML was significantly elevated after chemotherapy for 22 solid tumor types.
The relative risk of tMDS/AML was 1.5- to 39.0-fold greater among patients treated for these tumors than among the general population.
Lindsay M. Morton, PhD, of the National Institutes of Health in Rockville, Maryland, and her colleagues reported these findings in JAMA Oncology.
“We undertook an investigation to quantify tMDS/AML risks after chemotherapy for solid tumors in the modern treatment era, 2000-2014, using United States cancer registry data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results Program,” the investigators wrote.
They retrospectively analyzed data from 1619 patients with tMDS/AML who were diagnosed with an initial primary solid tumor from 2000 to 2013.
Patients were given initial chemotherapy and lived for at least 1 year after treatment. Subsequently, Dr. Morton and her colleagues linked patient database records with Medicare insurance claim information to confirm the accuracy of chemotherapy data.
“Because registry data do not include treatment details, we used an alternative database to provide descriptive information on population-based patterns of chemotherapeutic drug use,” the investigators noted.
The team found the risk of developing tMDS/AML was significantly increased following chemotherapy administration for 22 of 23 solid tumor types, excluding colon cancer.
The standardized incidence ratio (SIR) for tMDS/AML ranged from 1.5 to 39.0, and the excess absolute risk (EAR) ranged from 1.4 to 23.6 cases per 10,000 person-years.
SIRs were greatest in patients who received chemotherapy for malignancy of the bone (SIR=39.0, EAR=23.6), testis (SIR, 12.3, EAR=4.4), soft tissue (SIR=10.4, EAR=12.6), fallopian tube (SIR=8.7, EAR=16.0), small cell lung (SIR=8.1, EAR=19.9), peritoneum (SIR=7.5, EAR=15.8), brain or central nervous system (SIR=7.2, EAR=6.0), and ovary (SIR=5.8, EAR=8.2).
The investigators also found that patients who were given chemotherapy at a young age had the highest risk of developing tMDS/AML.
“For patients treated with chemotherapy at the present time, approximately three-quarters of tMDS/AML cases expected to occur within the next 5 years will be attributable to chemotherapy,” the investigators said.
They acknowledged a key limitation of this study was the limited data on patient-specific chemotherapy and dosing information. Given these limitations, Dr. Morton and her colleagues said, “the exact magnitude of our risk estimates, including the proportions of excess cases, should therefore be interpreted cautiously.”
This study was supported by the Intramural Research Program of the National Institutes of Health, National Cancer Institute, and the California Department of Public Health. The authors reported no conflicts of interest.
Model bests IPSS-R for predicting survival, risk for AML in myelodysplastic syndrome
SAN DIEGO – A newly developed personalized model that “harnesses the power of artificial intelligence” to predict overall survival and transformation to acute myeloid leukemia (AML) in patients with myelodysplastic syndromes outperforms both the original and revised International Prognostic Scoring Systems (IPSS, IPSS-R), according to Aziz Nazha, MD.
The machine learning model, which was built using clinical and genomic data derived from myelodysplastic syndrome (MDS) patients diagnosed according to 2008 World Health Organization criteria, incorporates information beyond that included in the IPSS and IPSS-R, and provides patient-specific survival probabilities at different time points, Dr. Nazha of Cleveland Clinic reported during a press briefing at the annual meeting of the American Society of Hematology.
The model was developed in a combined training cohort of 1,471 patients from the Cleveland Clinic and Munich Leukemia Laboratory and was validated in a separate cohort of 831 patients from the Moffitt Cancer Center in Tampa, Fla.
The concordance index – a measure for comparing the accuracy of the various models – was 0.80 for overall survival (OS), and 0.78 for AML transformation vs. 0.66 and 0.73, respectively, for IPSS, and 0.67 and 0.73, respectively, for IPSS-R, Dr. Nazha said. The new “geno-clinical” model also outperformed mutations-only analysis, mutations plus cytogenetics analysis, and mutations plus cytogenetics plus age analyses for both OS and AML transformation.
Adding mutational variant allelic frequency did not significantly improve prediction accuracy, he noted.
Dr. Nazha and his colleagues are developing a web application tool that can be used to run the trained model to calculate patient-specific, time-specific OS and AML transformation probabilities. He discussed the new model and its implications for personalized prognosis and treatment in this video interview.
Improved risk assessment helps patients understand their disease and “establish expectations about their journey with their disease,” and it is also extremely important for treating physicians, he said.
“All of our consensus guidelines and treatment recommendations are based on risk,” he explained, noting that the approach varies greatly for higher- and lower-risk patients.
This model represents a potential new focus on “personalized prediction” in addition to the increasing focus on personalized treatment and takes into account the heterogeneous outcomes seen in patients with MDS, he said.
Dr. Nazha reported consultancy for Karyopharma and Tolero, and data-monitoring committee membership for MEI.
SOURCE: Nazha A et al. ASH 2018, Abstract 793.
SAN DIEGO – A newly developed personalized model that “harnesses the power of artificial intelligence” to predict overall survival and transformation to acute myeloid leukemia (AML) in patients with myelodysplastic syndromes outperforms both the original and revised International Prognostic Scoring Systems (IPSS, IPSS-R), according to Aziz Nazha, MD.
The machine learning model, which was built using clinical and genomic data derived from myelodysplastic syndrome (MDS) patients diagnosed according to 2008 World Health Organization criteria, incorporates information beyond that included in the IPSS and IPSS-R, and provides patient-specific survival probabilities at different time points, Dr. Nazha of Cleveland Clinic reported during a press briefing at the annual meeting of the American Society of Hematology.
The model was developed in a combined training cohort of 1,471 patients from the Cleveland Clinic and Munich Leukemia Laboratory and was validated in a separate cohort of 831 patients from the Moffitt Cancer Center in Tampa, Fla.
The concordance index – a measure for comparing the accuracy of the various models – was 0.80 for overall survival (OS), and 0.78 for AML transformation vs. 0.66 and 0.73, respectively, for IPSS, and 0.67 and 0.73, respectively, for IPSS-R, Dr. Nazha said. The new “geno-clinical” model also outperformed mutations-only analysis, mutations plus cytogenetics analysis, and mutations plus cytogenetics plus age analyses for both OS and AML transformation.
Adding mutational variant allelic frequency did not significantly improve prediction accuracy, he noted.
Dr. Nazha and his colleagues are developing a web application tool that can be used to run the trained model to calculate patient-specific, time-specific OS and AML transformation probabilities. He discussed the new model and its implications for personalized prognosis and treatment in this video interview.
Improved risk assessment helps patients understand their disease and “establish expectations about their journey with their disease,” and it is also extremely important for treating physicians, he said.
“All of our consensus guidelines and treatment recommendations are based on risk,” he explained, noting that the approach varies greatly for higher- and lower-risk patients.
This model represents a potential new focus on “personalized prediction” in addition to the increasing focus on personalized treatment and takes into account the heterogeneous outcomes seen in patients with MDS, he said.
Dr. Nazha reported consultancy for Karyopharma and Tolero, and data-monitoring committee membership for MEI.
SOURCE: Nazha A et al. ASH 2018, Abstract 793.
SAN DIEGO – A newly developed personalized model that “harnesses the power of artificial intelligence” to predict overall survival and transformation to acute myeloid leukemia (AML) in patients with myelodysplastic syndromes outperforms both the original and revised International Prognostic Scoring Systems (IPSS, IPSS-R), according to Aziz Nazha, MD.
The machine learning model, which was built using clinical and genomic data derived from myelodysplastic syndrome (MDS) patients diagnosed according to 2008 World Health Organization criteria, incorporates information beyond that included in the IPSS and IPSS-R, and provides patient-specific survival probabilities at different time points, Dr. Nazha of Cleveland Clinic reported during a press briefing at the annual meeting of the American Society of Hematology.
The model was developed in a combined training cohort of 1,471 patients from the Cleveland Clinic and Munich Leukemia Laboratory and was validated in a separate cohort of 831 patients from the Moffitt Cancer Center in Tampa, Fla.
The concordance index – a measure for comparing the accuracy of the various models – was 0.80 for overall survival (OS), and 0.78 for AML transformation vs. 0.66 and 0.73, respectively, for IPSS, and 0.67 and 0.73, respectively, for IPSS-R, Dr. Nazha said. The new “geno-clinical” model also outperformed mutations-only analysis, mutations plus cytogenetics analysis, and mutations plus cytogenetics plus age analyses for both OS and AML transformation.
Adding mutational variant allelic frequency did not significantly improve prediction accuracy, he noted.
Dr. Nazha and his colleagues are developing a web application tool that can be used to run the trained model to calculate patient-specific, time-specific OS and AML transformation probabilities. He discussed the new model and its implications for personalized prognosis and treatment in this video interview.
Improved risk assessment helps patients understand their disease and “establish expectations about their journey with their disease,” and it is also extremely important for treating physicians, he said.
“All of our consensus guidelines and treatment recommendations are based on risk,” he explained, noting that the approach varies greatly for higher- and lower-risk patients.
This model represents a potential new focus on “personalized prediction” in addition to the increasing focus on personalized treatment and takes into account the heterogeneous outcomes seen in patients with MDS, he said.
Dr. Nazha reported consultancy for Karyopharma and Tolero, and data-monitoring committee membership for MEI.
SOURCE: Nazha A et al. ASH 2018, Abstract 793.
REPORTING FROM ASH 2018
Luspatercept reduced transfusion burden in MDS patients
SAN DIEGO—Results of the randomized, phase 3 Medalist trial show that the erythroid maturation agent luspatercept can reduce transfusion burden in patients with anemia due to myelodysplastic syndromes (MDS) and ring sideroblasts.
Almost 38% of luspatercept-treated patients achieved red blood cell (RBC) transfusion independence for 8 weeks or more, compared with 13% of patients receiving placebo.
And 28% of luspatercept-treated patients achieved transfusion independence for 12 weeks or more, compared to 8% in the placebo group.
Investigators reported these results as abstract 1 at the 2018 ASH Annual Meeting.
Treatment with luspatercept was “very well tolerated,” and responses were durable, with approximately 40% of patients remaining transfusion-free after 1 year of therapy, said senior investigator Alan F. List, MD, of Moffitt Cancer Center in Tampa, Florida, during a press conference at the meeting.
The first-in-class erythroid maturation agent is being developed as a treatment for anemia related to MDS and beta-thalassemia, Dr. List said.
“Luspatercept is a potential new therapy that we think could be very effective in patients with lower-risk MDS with ring sideroblasts who are red blood cell transfusion-dependent,” Dr. List affirmed.
Luspatercept is a soluble receptor chimera that binds to an array of ligands in the TGF-β superfamily, which is known to be very important in suppressing erythropoiesis in patients with MDS, Dr. List noted.
The Medalist study (NCT02631070) included patients with very low-, low-, or intermediate-risk disease and ring sideroblasts who were RBC transfusion-dependent and were refractory to, unresponsive to, or ineligible for first-line treatment with an erythropoiesis-stimulating agent (ESA).
A total of 153 patients were randomized to receive luspatercept at 1.0 mg/kg, administered subcutaneously every 21 days for at least 24 weeks, while 76 were randomized to placebo every 21 days.
The primary endpoint was the proportion of patients achieving RBC transfusion independence for at least 8 weeks during the first 24 weeks of treatment.
The primary endpoint was achieved by 37.9% of luspatercept-treated patients and 13.2% of placebo-treated patients (P<0.0001).
The luspatercept-treated patients also had a higher rate of erythroid response compared with the placebo group, at 52.9% and 11.8%, respectively (P<0.0001).
The investigators reported no differences in treatment-emergent adverse events, severe adverse events, or frequency of progression of acute myeloid leukemia (AML).
The safety profile was consistent with that of the phase 2 PACE-MDS study, which included treatment-related grade 3 myalgia (2%), increased blast cell count (2%), and general physical health deterioration (2%).
“This was a very clean drug and a very safe drug,” Dr. List said.
The decision to study luspatercept in patients with ring sideroblasts was based on results of the phase 2 PACE study showing a higher response rate in that subset of MDS patients, according to Dr. List.
The PACE study also included a small number of patients who had not previously received an ESA.
Currently underway is a phase 3 trial (NCT03682536) investigating luspatercept in ESA-naïve lower-risk MDS patients with anemia who require RBC transfusions.
Luspatercept would be a useful therapy to have in clinic for patients with ring sideroblasts, which represent about 25% of patients overall, according to MDS expert David Steensma, MD, of Dana-Farber Cancer Institute and Harvard Medical School in Boston, Massachusetts.
“It’s been 12 years since we had an FDA-approved drug in MDS, and there have been 7 in acute myeloid leukemia in the last year and a half, so it’s our turn, I think,” said Dr. Steensma, who moderated the press conference.
The Medalist study was sponsored by Celgene in collaboration with Acceleron Pharma, Inc.
Dr. List reported research funding from Celgene.
SAN DIEGO—Results of the randomized, phase 3 Medalist trial show that the erythroid maturation agent luspatercept can reduce transfusion burden in patients with anemia due to myelodysplastic syndromes (MDS) and ring sideroblasts.
Almost 38% of luspatercept-treated patients achieved red blood cell (RBC) transfusion independence for 8 weeks or more, compared with 13% of patients receiving placebo.
And 28% of luspatercept-treated patients achieved transfusion independence for 12 weeks or more, compared to 8% in the placebo group.
Investigators reported these results as abstract 1 at the 2018 ASH Annual Meeting.
Treatment with luspatercept was “very well tolerated,” and responses were durable, with approximately 40% of patients remaining transfusion-free after 1 year of therapy, said senior investigator Alan F. List, MD, of Moffitt Cancer Center in Tampa, Florida, during a press conference at the meeting.
The first-in-class erythroid maturation agent is being developed as a treatment for anemia related to MDS and beta-thalassemia, Dr. List said.
“Luspatercept is a potential new therapy that we think could be very effective in patients with lower-risk MDS with ring sideroblasts who are red blood cell transfusion-dependent,” Dr. List affirmed.
Luspatercept is a soluble receptor chimera that binds to an array of ligands in the TGF-β superfamily, which is known to be very important in suppressing erythropoiesis in patients with MDS, Dr. List noted.
The Medalist study (NCT02631070) included patients with very low-, low-, or intermediate-risk disease and ring sideroblasts who were RBC transfusion-dependent and were refractory to, unresponsive to, or ineligible for first-line treatment with an erythropoiesis-stimulating agent (ESA).
A total of 153 patients were randomized to receive luspatercept at 1.0 mg/kg, administered subcutaneously every 21 days for at least 24 weeks, while 76 were randomized to placebo every 21 days.
The primary endpoint was the proportion of patients achieving RBC transfusion independence for at least 8 weeks during the first 24 weeks of treatment.
The primary endpoint was achieved by 37.9% of luspatercept-treated patients and 13.2% of placebo-treated patients (P<0.0001).
The luspatercept-treated patients also had a higher rate of erythroid response compared with the placebo group, at 52.9% and 11.8%, respectively (P<0.0001).
The investigators reported no differences in treatment-emergent adverse events, severe adverse events, or frequency of progression of acute myeloid leukemia (AML).
The safety profile was consistent with that of the phase 2 PACE-MDS study, which included treatment-related grade 3 myalgia (2%), increased blast cell count (2%), and general physical health deterioration (2%).
“This was a very clean drug and a very safe drug,” Dr. List said.
The decision to study luspatercept in patients with ring sideroblasts was based on results of the phase 2 PACE study showing a higher response rate in that subset of MDS patients, according to Dr. List.
The PACE study also included a small number of patients who had not previously received an ESA.
Currently underway is a phase 3 trial (NCT03682536) investigating luspatercept in ESA-naïve lower-risk MDS patients with anemia who require RBC transfusions.
Luspatercept would be a useful therapy to have in clinic for patients with ring sideroblasts, which represent about 25% of patients overall, according to MDS expert David Steensma, MD, of Dana-Farber Cancer Institute and Harvard Medical School in Boston, Massachusetts.
“It’s been 12 years since we had an FDA-approved drug in MDS, and there have been 7 in acute myeloid leukemia in the last year and a half, so it’s our turn, I think,” said Dr. Steensma, who moderated the press conference.
The Medalist study was sponsored by Celgene in collaboration with Acceleron Pharma, Inc.
Dr. List reported research funding from Celgene.
SAN DIEGO—Results of the randomized, phase 3 Medalist trial show that the erythroid maturation agent luspatercept can reduce transfusion burden in patients with anemia due to myelodysplastic syndromes (MDS) and ring sideroblasts.
Almost 38% of luspatercept-treated patients achieved red blood cell (RBC) transfusion independence for 8 weeks or more, compared with 13% of patients receiving placebo.
And 28% of luspatercept-treated patients achieved transfusion independence for 12 weeks or more, compared to 8% in the placebo group.
Investigators reported these results as abstract 1 at the 2018 ASH Annual Meeting.
Treatment with luspatercept was “very well tolerated,” and responses were durable, with approximately 40% of patients remaining transfusion-free after 1 year of therapy, said senior investigator Alan F. List, MD, of Moffitt Cancer Center in Tampa, Florida, during a press conference at the meeting.
The first-in-class erythroid maturation agent is being developed as a treatment for anemia related to MDS and beta-thalassemia, Dr. List said.
“Luspatercept is a potential new therapy that we think could be very effective in patients with lower-risk MDS with ring sideroblasts who are red blood cell transfusion-dependent,” Dr. List affirmed.
Luspatercept is a soluble receptor chimera that binds to an array of ligands in the TGF-β superfamily, which is known to be very important in suppressing erythropoiesis in patients with MDS, Dr. List noted.
The Medalist study (NCT02631070) included patients with very low-, low-, or intermediate-risk disease and ring sideroblasts who were RBC transfusion-dependent and were refractory to, unresponsive to, or ineligible for first-line treatment with an erythropoiesis-stimulating agent (ESA).
A total of 153 patients were randomized to receive luspatercept at 1.0 mg/kg, administered subcutaneously every 21 days for at least 24 weeks, while 76 were randomized to placebo every 21 days.
The primary endpoint was the proportion of patients achieving RBC transfusion independence for at least 8 weeks during the first 24 weeks of treatment.
The primary endpoint was achieved by 37.9% of luspatercept-treated patients and 13.2% of placebo-treated patients (P<0.0001).
The luspatercept-treated patients also had a higher rate of erythroid response compared with the placebo group, at 52.9% and 11.8%, respectively (P<0.0001).
The investigators reported no differences in treatment-emergent adverse events, severe adverse events, or frequency of progression of acute myeloid leukemia (AML).
The safety profile was consistent with that of the phase 2 PACE-MDS study, which included treatment-related grade 3 myalgia (2%), increased blast cell count (2%), and general physical health deterioration (2%).
“This was a very clean drug and a very safe drug,” Dr. List said.
The decision to study luspatercept in patients with ring sideroblasts was based on results of the phase 2 PACE study showing a higher response rate in that subset of MDS patients, according to Dr. List.
The PACE study also included a small number of patients who had not previously received an ESA.
Currently underway is a phase 3 trial (NCT03682536) investigating luspatercept in ESA-naïve lower-risk MDS patients with anemia who require RBC transfusions.
Luspatercept would be a useful therapy to have in clinic for patients with ring sideroblasts, which represent about 25% of patients overall, according to MDS expert David Steensma, MD, of Dana-Farber Cancer Institute and Harvard Medical School in Boston, Massachusetts.
“It’s been 12 years since we had an FDA-approved drug in MDS, and there have been 7 in acute myeloid leukemia in the last year and a half, so it’s our turn, I think,” said Dr. Steensma, who moderated the press conference.
The Medalist study was sponsored by Celgene in collaboration with Acceleron Pharma, Inc.
Dr. List reported research funding from Celgene.
MEDALIST: Erythroid maturation agent reduced transfusion burden in MDS
SAN DIEGO – A novel erythroid maturation agent significantly reduced transfusion burden versus placebo in patients with anemia caused by myelodysplastic syndromes (MDS) and ringed sideroblasts, results of a randomized, phase 3 trial demonstrate.
Luspatercept was “very well tolerated” and responses were durable, with approximately 40% of patients remaining transfusion free after 1 year of therapy, said Alan F. List, MD, of Moffitt Cancer Center, Tampa.
“Luspatercept is a potential new therapy that we think could be very effective in patients with lower-risk MDS with ringed sideroblasts who are red blood cell transfusion–dependent,” said Dr. List, senior author of the MEDALIST trial, said in a press conference at the annual meeting of the American Society of Hematology.
The first-in-class erythroid maturation agent is being developed as a treatment for anemia related to MDS and beta-thalassemia, Dr. List said.
In a separate randomized, placebo-controlled, phase 3 study presented at ASH 2018, Maria Domenica Cappellini, MD, of the University of Milan, reported that, in beta-thalassemia patients who were transfusion dependent, luspatercept treatment resulted in a statistically significant reductions in transfusion burden versus placebo, and was generally well tolerated.
Luspatercept is a soluble receptor chimera that binds to an array of ligands in the transforming growth factor–beta superfamily, which is known to be important in suppressing erythropoiesis in patients with MDS, Dr. List said.
The MDS study included patients with very low–, low-, or intermediate-risk disease and ringed sideroblasts who were RBC transfusion–dependent and were refractory to, unresponsive to, or ineligible for first-line treatment with an erythropoiesis-stimulating agent (ESA).
A total of 153 patients were randomly allocated to luspatercept 1.0 mg/kg, administered subcutaneously every 21 days for at least 24 weeks, while 76 were randomized to placebo every 21 days. The primary end point was the proportion of patients achieving RBC transfusion independence for at least 8 weeks during the first 24 weeks of treatment.
A total of 37.9% of luspatercept-treated patients achieved that primary endpoint, compared with 13.2% of placebo-treated patient (P less than .0001), Dr. List reported. The luspatercept-treated patients also had a 52.9% rate of erythroid response, compared with 11.8% in the placebo group (P less than .0001).
There were no differences in treatment-emergent adverse events, severe adverse events, or frequency of progression of acute myeloid leukemia. “This was a very clean drug and a very safe drug,” he said.
The decision to study luspatercept in patients with ringed sideroblasts was based on results of a large, phase 2 European study showing a higher response rate in that subset of MDS patients, according to Dr. List.
That study also included a small number of patients who had not previously received an ESA. Currently underway is a phase 3 trial looking at luspatercept in ESA-naive, lower-risk MDS patients with anemia who require RBC transfusions.
Luspatercept would be a useful therapy to have in clinic for patients with ring sideroblasts, who represent about 25% of patients overall, according to MDS expert David Steensma, MD, of Dana-Farber Cancer Institute and Harvard Medical School, Boston.
“It’s been 12 years since we had an FDA [Food and Drug Administration]-approved drug in MDS, and there have been seven in acute myeloid leukemia in the last year and a half, so it’s our turn, I think,” said Dr. Steensma, who moderated the press conference.
Dr. List reported research funding from Celgene.
SOURCE: List AF et al. ASH 2018, Abstract 1.
SAN DIEGO – A novel erythroid maturation agent significantly reduced transfusion burden versus placebo in patients with anemia caused by myelodysplastic syndromes (MDS) and ringed sideroblasts, results of a randomized, phase 3 trial demonstrate.
Luspatercept was “very well tolerated” and responses were durable, with approximately 40% of patients remaining transfusion free after 1 year of therapy, said Alan F. List, MD, of Moffitt Cancer Center, Tampa.
“Luspatercept is a potential new therapy that we think could be very effective in patients with lower-risk MDS with ringed sideroblasts who are red blood cell transfusion–dependent,” said Dr. List, senior author of the MEDALIST trial, said in a press conference at the annual meeting of the American Society of Hematology.
The first-in-class erythroid maturation agent is being developed as a treatment for anemia related to MDS and beta-thalassemia, Dr. List said.
In a separate randomized, placebo-controlled, phase 3 study presented at ASH 2018, Maria Domenica Cappellini, MD, of the University of Milan, reported that, in beta-thalassemia patients who were transfusion dependent, luspatercept treatment resulted in a statistically significant reductions in transfusion burden versus placebo, and was generally well tolerated.
Luspatercept is a soluble receptor chimera that binds to an array of ligands in the transforming growth factor–beta superfamily, which is known to be important in suppressing erythropoiesis in patients with MDS, Dr. List said.
The MDS study included patients with very low–, low-, or intermediate-risk disease and ringed sideroblasts who were RBC transfusion–dependent and were refractory to, unresponsive to, or ineligible for first-line treatment with an erythropoiesis-stimulating agent (ESA).
A total of 153 patients were randomly allocated to luspatercept 1.0 mg/kg, administered subcutaneously every 21 days for at least 24 weeks, while 76 were randomized to placebo every 21 days. The primary end point was the proportion of patients achieving RBC transfusion independence for at least 8 weeks during the first 24 weeks of treatment.
A total of 37.9% of luspatercept-treated patients achieved that primary endpoint, compared with 13.2% of placebo-treated patient (P less than .0001), Dr. List reported. The luspatercept-treated patients also had a 52.9% rate of erythroid response, compared with 11.8% in the placebo group (P less than .0001).
There were no differences in treatment-emergent adverse events, severe adverse events, or frequency of progression of acute myeloid leukemia. “This was a very clean drug and a very safe drug,” he said.
The decision to study luspatercept in patients with ringed sideroblasts was based on results of a large, phase 2 European study showing a higher response rate in that subset of MDS patients, according to Dr. List.
That study also included a small number of patients who had not previously received an ESA. Currently underway is a phase 3 trial looking at luspatercept in ESA-naive, lower-risk MDS patients with anemia who require RBC transfusions.
Luspatercept would be a useful therapy to have in clinic for patients with ring sideroblasts, who represent about 25% of patients overall, according to MDS expert David Steensma, MD, of Dana-Farber Cancer Institute and Harvard Medical School, Boston.
“It’s been 12 years since we had an FDA [Food and Drug Administration]-approved drug in MDS, and there have been seven in acute myeloid leukemia in the last year and a half, so it’s our turn, I think,” said Dr. Steensma, who moderated the press conference.
Dr. List reported research funding from Celgene.
SOURCE: List AF et al. ASH 2018, Abstract 1.
SAN DIEGO – A novel erythroid maturation agent significantly reduced transfusion burden versus placebo in patients with anemia caused by myelodysplastic syndromes (MDS) and ringed sideroblasts, results of a randomized, phase 3 trial demonstrate.
Luspatercept was “very well tolerated” and responses were durable, with approximately 40% of patients remaining transfusion free after 1 year of therapy, said Alan F. List, MD, of Moffitt Cancer Center, Tampa.
“Luspatercept is a potential new therapy that we think could be very effective in patients with lower-risk MDS with ringed sideroblasts who are red blood cell transfusion–dependent,” said Dr. List, senior author of the MEDALIST trial, said in a press conference at the annual meeting of the American Society of Hematology.
The first-in-class erythroid maturation agent is being developed as a treatment for anemia related to MDS and beta-thalassemia, Dr. List said.
In a separate randomized, placebo-controlled, phase 3 study presented at ASH 2018, Maria Domenica Cappellini, MD, of the University of Milan, reported that, in beta-thalassemia patients who were transfusion dependent, luspatercept treatment resulted in a statistically significant reductions in transfusion burden versus placebo, and was generally well tolerated.
Luspatercept is a soluble receptor chimera that binds to an array of ligands in the transforming growth factor–beta superfamily, which is known to be important in suppressing erythropoiesis in patients with MDS, Dr. List said.
The MDS study included patients with very low–, low-, or intermediate-risk disease and ringed sideroblasts who were RBC transfusion–dependent and were refractory to, unresponsive to, or ineligible for first-line treatment with an erythropoiesis-stimulating agent (ESA).
A total of 153 patients were randomly allocated to luspatercept 1.0 mg/kg, administered subcutaneously every 21 days for at least 24 weeks, while 76 were randomized to placebo every 21 days. The primary end point was the proportion of patients achieving RBC transfusion independence for at least 8 weeks during the first 24 weeks of treatment.
A total of 37.9% of luspatercept-treated patients achieved that primary endpoint, compared with 13.2% of placebo-treated patient (P less than .0001), Dr. List reported. The luspatercept-treated patients also had a 52.9% rate of erythroid response, compared with 11.8% in the placebo group (P less than .0001).
There were no differences in treatment-emergent adverse events, severe adverse events, or frequency of progression of acute myeloid leukemia. “This was a very clean drug and a very safe drug,” he said.
The decision to study luspatercept in patients with ringed sideroblasts was based on results of a large, phase 2 European study showing a higher response rate in that subset of MDS patients, according to Dr. List.
That study also included a small number of patients who had not previously received an ESA. Currently underway is a phase 3 trial looking at luspatercept in ESA-naive, lower-risk MDS patients with anemia who require RBC transfusions.
Luspatercept would be a useful therapy to have in clinic for patients with ring sideroblasts, who represent about 25% of patients overall, according to MDS expert David Steensma, MD, of Dana-Farber Cancer Institute and Harvard Medical School, Boston.
“It’s been 12 years since we had an FDA [Food and Drug Administration]-approved drug in MDS, and there have been seven in acute myeloid leukemia in the last year and a half, so it’s our turn, I think,” said Dr. Steensma, who moderated the press conference.
Dr. List reported research funding from Celgene.
SOURCE: List AF et al. ASH 2018, Abstract 1.
REPORTING FROM ASH 2018
Key clinical point: Luspatercept, a novel erythroid maturation agent, significantly reduced transfusion burden versus placebo in patients with anemia caused by myelodysplastic syndromes and ringed sideroblasts.
Major finding: The proportion of patients achieving RBC transfusion independence for at least 8 weeks during the first 24 weeks of treatment was 37.9% for luspatercept and 13.2% for placebo (P less than .0001).
Study details: A randomized, phase 3 trial including 220 lower-risk myelodysplastic syndromes patients with ringed sideroblasts who were RBC transfusion–dependent.
Disclosures: Dr. List reported research funding from Celgene.
Source: List AF et al. ASH 2018, abstract 1.
Your guide to ASH 2018: Abstracts to watch
With more than 3,000 scientific abstracts at the 2018 annual meeting of the American Society of Hematology, it can be tough to figure out what research is most relevant to practice. But the editorial advisory board of Hematology News is making it easier this year with their picks for what to watch and why.
Lymphomas
Brian T. Hill, MD, of the Cleveland Clinic, offered his top picks in lymphoma research. Results of the phase 3 international Alliance North American Intergroup Study A041202 will be presented during the ASH plenary session at 2 p.m. PT on Sunday, Dec. 2 in Hall AB of the San Diego Convention Center (Abstract 6). The study compared bendamustine plus rituximab with ibrutinib and the combination of ibrutinib plus rituximab to see if the ibrutinib-containing therapies would have superior progression-free survival (PFS) in chronic lymphocytic leukemia (CLL), compared with chemoimmunotherapy. Results indicate that ibrutinib had superior PFS in older patients with CLL and could be a standard of care in this population.
The study is worth watching because it is the first report of a head-to-head trial of chemotherapy versus ibrutinib for first-line treatment of CLL, Dr. Hill said.
Two more studies offer important reports of “real world” experiences with chimeric antigen receptor (CAR) T-cell therapy.
In one multicenter retrospective study, researchers evaluated the outcomes of axicabtagene ciloleucel (axi-cel) CAR T-cell therapy for relapsed/refractory aggressive B-cell lymphoma when it is used a standard care. The researchers will report that 30-day responses in the real-world setting were comparable to the best responses seen in the ZUMA-1 trial. The full results will be reported at 9:30 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 91).
Another retrospective analysis looked at the use of axi-cell and revealed some critical differences from ZUMA-1, specifically the overall response rate (ORR) and complete response (CR) rate were lower than those reported in the pivotal clinical trial. The findings will be reported at 9:45 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 92).
Researchers will also present the unblinded results from the ECHELON-2 study, which compared the efficacy and safety of brentuximab vedotin in combination with cyclophosphamide, doxorubicin, and prednisone (CHP) versus standard CHOP for the treatment of patients with peripheral T-cell lymphoma. The results will be presented at 6:15 p.m. PT on Monday, Dec. 3 in room 6F of the San Diego convention center (Abstract 997).
Previously reported blinded pooled data showed that the treatment was well tolerated with 3-year PFS of 53% and OS of 73%.
“This should be a new standard of care for T-cell lymphomas,” Dr. Hill said.
CAR T-cell therapy
There are a number of abstracts featuring the latest results on CAR T-cell therapy. Helen Heslop, MD, of Baylor College of Medicine, Houston, recommended an updated analysis from the ELIANA study, which looked at the efficacy and safety of tisagenlecleucel in for children and young adults with relapsed/refractory acute lymphoblastic leukemia (ALL).
“Longer-term follow-up of the ELIANA study shows encouraging remission-duration data in pediatric and young adults with ALL without additional therapy,” Dr. Heslop said.
The findings will be presented at 4:30 p.m. PT on Monday, Dec. 3 in room 6A at the San Diego Convention Center (Abstract 895).
Another notable presentation will feature results from a phase 1B/2 trial evaluating infusion of CAR T cells targeting the CD30 molecule and encoding the CD28 endodomain (CD30.CAR-Ts) after lymphodepleting chemotherapy in patients with relapsed or refractory CD30+ Hodgkin lymphoma and non-Hodgkin lymphoma.
The researchers will report that there was a significant PFS advances for who received the highest dose level of the CAR T treatment, combined with bendamustine and fludarabine.
The study will be presented at 11 a.m. PT on Monday, Dec. 3 in room 6F at the San Diego Convention Center (Abstract 681).
Dr. Heslop also recommends another study being presented in the same session, which also shows encouraging results with CD30.CAR-Ts. Dr. Heslop is one of the co-investigators on the phase 1 RELY-30 trial, which is evaluating the efficacy of CD30.CAR-Ts after lymphodepleting chemotherapy. Preliminary results suggest a substantial improvement in efficacy. The findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in room 6F of the San Diego Convention Center (Abstract 680).
MDS/MPN
Vikas Gupta, MD, of Princess Margaret Cancer Center in Toronto, highlighted three abstracts to watch in the areas of myelodysplastic syndrome (MDS) and myeloproliferative neoplasms (MPN).
The phase 3 Medalist trial is a randomized double-blind placebo controlled study of luspatercept to treatment anemia in patients with MDS with ring sideroblasts who require red blood cell transfusion. The researchers will report significantly reduced transfusion burdens for luspatercept, compared with placebo.
“This is a practice-changing, pivotal trial in the field of MDS for the treatment of anemia,” Dr. Gupta said.
The findings will be presented at 2 p.m. PT on Sunday, Dec. 2 during the plenary session in Hall AB in the San Diego Convention Center (Abstract 1).
Also during the Sunday plenary session is a presentation on MPN therapy (Abstract 4). Researchers will present data on secreted mutant calreticulins as rogue cytokines trigger thrombopoietin receptor (TpoR) activation, specifically in CALR-mutated cells.
“This study investigates in to the mechanistic oncogenetic aspects of mutant calreticulin, and has potential for therapeutic approaches in the future,” Dr. Gupta said.
The ASH meeting will also feature the final analysis of the MPN-RC 112 consortium trial of pegylated interferon alfa-2a versus hydroxyurea for the treatment of high-risk polycythemia vera (PV) and essential thrombocythemia (ET). The researchers will report that the CR rates at 12 and 24 months were similar in patients treated with pegylated interferon alfa-2a and hydroxyurea, but pegylated interferon alfa-2a was associated with a higher rate of serious toxicities.
“There is a continuous debate on optimal first-line cytoreductive therapy for high risk PV/ET, and this is one of the first randomized study to answer this question,” Dr. Gupta said.
The findings will be presented at 7 a.m. PT on Monday, Dec. 3 in Grand Hall D at the Manchester Grand Hyatt San Diego (Abstract 577).
AML
For attendees interested in the latest developments in acute myeloid leukemia, Thomas Fischer, MD, of Otto-von-Guericke-University Magdeburg (Germany), highlighted three don’t-miss sessions.
In an analysis of a large cohort of FLT3-ITD mutated AML patients in the RATIFY trial, researchers looked at the prognostic impact of ITD insertion site.
“Interestingly, in this large cohort of 452 FLT3-ITD mutated AML, the negative prognostic impact of beta1-sheet insertion site of FLT3-ITD could be confirmed,” Dr. Fischer said. “Further analysis of a potential predictive effect on outcome of midostaurin treatment is ongoing and will be very interesting.”
The findings will be presented at 5 p.m. PT on Sunday, Dec. 2 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 435).
Another notable presentation features results from the phase 2 RADIUS trial, a randomized study comparing standard of care, with and without midostaurin, after allogeneic stem cell transplant in FLT3-ITD–mutated AML.
“Here, efficacy and toxicity of midostaurin was investigated in a [minimal residual disease] situation post-alloSCT,” Dr. Fischer said. “Interestingly, adding midostaurin to standard of care reduced the risk of relapse at 18 months post-alloSCT by 46%.”
The complete findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 662).
Dr. Fischer singled out another study looking at the efficacy and safety of single-agent quizartinib in patients with FLT3-ITD mutated AML. In this large, randomized trial the researchers noted a significant improvement in CR rates and survival benefit with the single agent FLT3 inhibitors, compared with salvage chemotherapy for patients with relapsed/refractory mutated AML.
The findings will be presented at 8 a.m. on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 563).
Notable posters
Iberia Romina Sosa, MD, PhD, of Baylor College of Medicine in Houston, suggested several posters worth visiting in the areas of thrombosis and bleeding.
Poster 1134 looks at the TNF-alpha driven inflammation and mitochondrial dysfunction in the platelet hyperreactivity of aging and MPN.
How do you know if your therapy for thrombotic thrombocytopenic purpura is working? Poster 3736 examines the measurement of cell-derived microparticles as a possible tool to monitor response to therapy.
You don’t have to be taking aspirin to have a bleeding profile characteristic with consumption of a cyclooxygenase inhibitor. Poster 1156 provides a first report of a platelet function disorder caused by autosomal recessive inheritance of PTGS1.
Poster 2477 takes a closer look at fitusiran, an antithrombin inhibitor, which improves thrombin generation in patients with hemophilia A or B. Protocol amendments for safety monitoring move fitusiran to phase 3 trials, Dr. Sosa said.
With more than 3,000 scientific abstracts at the 2018 annual meeting of the American Society of Hematology, it can be tough to figure out what research is most relevant to practice. But the editorial advisory board of Hematology News is making it easier this year with their picks for what to watch and why.
Lymphomas
Brian T. Hill, MD, of the Cleveland Clinic, offered his top picks in lymphoma research. Results of the phase 3 international Alliance North American Intergroup Study A041202 will be presented during the ASH plenary session at 2 p.m. PT on Sunday, Dec. 2 in Hall AB of the San Diego Convention Center (Abstract 6). The study compared bendamustine plus rituximab with ibrutinib and the combination of ibrutinib plus rituximab to see if the ibrutinib-containing therapies would have superior progression-free survival (PFS) in chronic lymphocytic leukemia (CLL), compared with chemoimmunotherapy. Results indicate that ibrutinib had superior PFS in older patients with CLL and could be a standard of care in this population.
The study is worth watching because it is the first report of a head-to-head trial of chemotherapy versus ibrutinib for first-line treatment of CLL, Dr. Hill said.
Two more studies offer important reports of “real world” experiences with chimeric antigen receptor (CAR) T-cell therapy.
In one multicenter retrospective study, researchers evaluated the outcomes of axicabtagene ciloleucel (axi-cel) CAR T-cell therapy for relapsed/refractory aggressive B-cell lymphoma when it is used a standard care. The researchers will report that 30-day responses in the real-world setting were comparable to the best responses seen in the ZUMA-1 trial. The full results will be reported at 9:30 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 91).
Another retrospective analysis looked at the use of axi-cell and revealed some critical differences from ZUMA-1, specifically the overall response rate (ORR) and complete response (CR) rate were lower than those reported in the pivotal clinical trial. The findings will be reported at 9:45 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 92).
Researchers will also present the unblinded results from the ECHELON-2 study, which compared the efficacy and safety of brentuximab vedotin in combination with cyclophosphamide, doxorubicin, and prednisone (CHP) versus standard CHOP for the treatment of patients with peripheral T-cell lymphoma. The results will be presented at 6:15 p.m. PT on Monday, Dec. 3 in room 6F of the San Diego convention center (Abstract 997).
Previously reported blinded pooled data showed that the treatment was well tolerated with 3-year PFS of 53% and OS of 73%.
“This should be a new standard of care for T-cell lymphomas,” Dr. Hill said.
CAR T-cell therapy
There are a number of abstracts featuring the latest results on CAR T-cell therapy. Helen Heslop, MD, of Baylor College of Medicine, Houston, recommended an updated analysis from the ELIANA study, which looked at the efficacy and safety of tisagenlecleucel in for children and young adults with relapsed/refractory acute lymphoblastic leukemia (ALL).
“Longer-term follow-up of the ELIANA study shows encouraging remission-duration data in pediatric and young adults with ALL without additional therapy,” Dr. Heslop said.
The findings will be presented at 4:30 p.m. PT on Monday, Dec. 3 in room 6A at the San Diego Convention Center (Abstract 895).
Another notable presentation will feature results from a phase 1B/2 trial evaluating infusion of CAR T cells targeting the CD30 molecule and encoding the CD28 endodomain (CD30.CAR-Ts) after lymphodepleting chemotherapy in patients with relapsed or refractory CD30+ Hodgkin lymphoma and non-Hodgkin lymphoma.
The researchers will report that there was a significant PFS advances for who received the highest dose level of the CAR T treatment, combined with bendamustine and fludarabine.
The study will be presented at 11 a.m. PT on Monday, Dec. 3 in room 6F at the San Diego Convention Center (Abstract 681).
Dr. Heslop also recommends another study being presented in the same session, which also shows encouraging results with CD30.CAR-Ts. Dr. Heslop is one of the co-investigators on the phase 1 RELY-30 trial, which is evaluating the efficacy of CD30.CAR-Ts after lymphodepleting chemotherapy. Preliminary results suggest a substantial improvement in efficacy. The findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in room 6F of the San Diego Convention Center (Abstract 680).
MDS/MPN
Vikas Gupta, MD, of Princess Margaret Cancer Center in Toronto, highlighted three abstracts to watch in the areas of myelodysplastic syndrome (MDS) and myeloproliferative neoplasms (MPN).
The phase 3 Medalist trial is a randomized double-blind placebo controlled study of luspatercept to treatment anemia in patients with MDS with ring sideroblasts who require red blood cell transfusion. The researchers will report significantly reduced transfusion burdens for luspatercept, compared with placebo.
“This is a practice-changing, pivotal trial in the field of MDS for the treatment of anemia,” Dr. Gupta said.
The findings will be presented at 2 p.m. PT on Sunday, Dec. 2 during the plenary session in Hall AB in the San Diego Convention Center (Abstract 1).
Also during the Sunday plenary session is a presentation on MPN therapy (Abstract 4). Researchers will present data on secreted mutant calreticulins as rogue cytokines trigger thrombopoietin receptor (TpoR) activation, specifically in CALR-mutated cells.
“This study investigates in to the mechanistic oncogenetic aspects of mutant calreticulin, and has potential for therapeutic approaches in the future,” Dr. Gupta said.
The ASH meeting will also feature the final analysis of the MPN-RC 112 consortium trial of pegylated interferon alfa-2a versus hydroxyurea for the treatment of high-risk polycythemia vera (PV) and essential thrombocythemia (ET). The researchers will report that the CR rates at 12 and 24 months were similar in patients treated with pegylated interferon alfa-2a and hydroxyurea, but pegylated interferon alfa-2a was associated with a higher rate of serious toxicities.
“There is a continuous debate on optimal first-line cytoreductive therapy for high risk PV/ET, and this is one of the first randomized study to answer this question,” Dr. Gupta said.
The findings will be presented at 7 a.m. PT on Monday, Dec. 3 in Grand Hall D at the Manchester Grand Hyatt San Diego (Abstract 577).
AML
For attendees interested in the latest developments in acute myeloid leukemia, Thomas Fischer, MD, of Otto-von-Guericke-University Magdeburg (Germany), highlighted three don’t-miss sessions.
In an analysis of a large cohort of FLT3-ITD mutated AML patients in the RATIFY trial, researchers looked at the prognostic impact of ITD insertion site.
“Interestingly, in this large cohort of 452 FLT3-ITD mutated AML, the negative prognostic impact of beta1-sheet insertion site of FLT3-ITD could be confirmed,” Dr. Fischer said. “Further analysis of a potential predictive effect on outcome of midostaurin treatment is ongoing and will be very interesting.”
The findings will be presented at 5 p.m. PT on Sunday, Dec. 2 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 435).
Another notable presentation features results from the phase 2 RADIUS trial, a randomized study comparing standard of care, with and without midostaurin, after allogeneic stem cell transplant in FLT3-ITD–mutated AML.
“Here, efficacy and toxicity of midostaurin was investigated in a [minimal residual disease] situation post-alloSCT,” Dr. Fischer said. “Interestingly, adding midostaurin to standard of care reduced the risk of relapse at 18 months post-alloSCT by 46%.”
The complete findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 662).
Dr. Fischer singled out another study looking at the efficacy and safety of single-agent quizartinib in patients with FLT3-ITD mutated AML. In this large, randomized trial the researchers noted a significant improvement in CR rates and survival benefit with the single agent FLT3 inhibitors, compared with salvage chemotherapy for patients with relapsed/refractory mutated AML.
The findings will be presented at 8 a.m. on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 563).
Notable posters
Iberia Romina Sosa, MD, PhD, of Baylor College of Medicine in Houston, suggested several posters worth visiting in the areas of thrombosis and bleeding.
Poster 1134 looks at the TNF-alpha driven inflammation and mitochondrial dysfunction in the platelet hyperreactivity of aging and MPN.
How do you know if your therapy for thrombotic thrombocytopenic purpura is working? Poster 3736 examines the measurement of cell-derived microparticles as a possible tool to monitor response to therapy.
You don’t have to be taking aspirin to have a bleeding profile characteristic with consumption of a cyclooxygenase inhibitor. Poster 1156 provides a first report of a platelet function disorder caused by autosomal recessive inheritance of PTGS1.
Poster 2477 takes a closer look at fitusiran, an antithrombin inhibitor, which improves thrombin generation in patients with hemophilia A or B. Protocol amendments for safety monitoring move fitusiran to phase 3 trials, Dr. Sosa said.
With more than 3,000 scientific abstracts at the 2018 annual meeting of the American Society of Hematology, it can be tough to figure out what research is most relevant to practice. But the editorial advisory board of Hematology News is making it easier this year with their picks for what to watch and why.
Lymphomas
Brian T. Hill, MD, of the Cleveland Clinic, offered his top picks in lymphoma research. Results of the phase 3 international Alliance North American Intergroup Study A041202 will be presented during the ASH plenary session at 2 p.m. PT on Sunday, Dec. 2 in Hall AB of the San Diego Convention Center (Abstract 6). The study compared bendamustine plus rituximab with ibrutinib and the combination of ibrutinib plus rituximab to see if the ibrutinib-containing therapies would have superior progression-free survival (PFS) in chronic lymphocytic leukemia (CLL), compared with chemoimmunotherapy. Results indicate that ibrutinib had superior PFS in older patients with CLL and could be a standard of care in this population.
The study is worth watching because it is the first report of a head-to-head trial of chemotherapy versus ibrutinib for first-line treatment of CLL, Dr. Hill said.
Two more studies offer important reports of “real world” experiences with chimeric antigen receptor (CAR) T-cell therapy.
In one multicenter retrospective study, researchers evaluated the outcomes of axicabtagene ciloleucel (axi-cel) CAR T-cell therapy for relapsed/refractory aggressive B-cell lymphoma when it is used a standard care. The researchers will report that 30-day responses in the real-world setting were comparable to the best responses seen in the ZUMA-1 trial. The full results will be reported at 9:30 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 91).
Another retrospective analysis looked at the use of axi-cell and revealed some critical differences from ZUMA-1, specifically the overall response rate (ORR) and complete response (CR) rate were lower than those reported in the pivotal clinical trial. The findings will be reported at 9:45 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 92).
Researchers will also present the unblinded results from the ECHELON-2 study, which compared the efficacy and safety of brentuximab vedotin in combination with cyclophosphamide, doxorubicin, and prednisone (CHP) versus standard CHOP for the treatment of patients with peripheral T-cell lymphoma. The results will be presented at 6:15 p.m. PT on Monday, Dec. 3 in room 6F of the San Diego convention center (Abstract 997).
Previously reported blinded pooled data showed that the treatment was well tolerated with 3-year PFS of 53% and OS of 73%.
“This should be a new standard of care for T-cell lymphomas,” Dr. Hill said.
CAR T-cell therapy
There are a number of abstracts featuring the latest results on CAR T-cell therapy. Helen Heslop, MD, of Baylor College of Medicine, Houston, recommended an updated analysis from the ELIANA study, which looked at the efficacy and safety of tisagenlecleucel in for children and young adults with relapsed/refractory acute lymphoblastic leukemia (ALL).
“Longer-term follow-up of the ELIANA study shows encouraging remission-duration data in pediatric and young adults with ALL without additional therapy,” Dr. Heslop said.
The findings will be presented at 4:30 p.m. PT on Monday, Dec. 3 in room 6A at the San Diego Convention Center (Abstract 895).
Another notable presentation will feature results from a phase 1B/2 trial evaluating infusion of CAR T cells targeting the CD30 molecule and encoding the CD28 endodomain (CD30.CAR-Ts) after lymphodepleting chemotherapy in patients with relapsed or refractory CD30+ Hodgkin lymphoma and non-Hodgkin lymphoma.
The researchers will report that there was a significant PFS advances for who received the highest dose level of the CAR T treatment, combined with bendamustine and fludarabine.
The study will be presented at 11 a.m. PT on Monday, Dec. 3 in room 6F at the San Diego Convention Center (Abstract 681).
Dr. Heslop also recommends another study being presented in the same session, which also shows encouraging results with CD30.CAR-Ts. Dr. Heslop is one of the co-investigators on the phase 1 RELY-30 trial, which is evaluating the efficacy of CD30.CAR-Ts after lymphodepleting chemotherapy. Preliminary results suggest a substantial improvement in efficacy. The findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in room 6F of the San Diego Convention Center (Abstract 680).
MDS/MPN
Vikas Gupta, MD, of Princess Margaret Cancer Center in Toronto, highlighted three abstracts to watch in the areas of myelodysplastic syndrome (MDS) and myeloproliferative neoplasms (MPN).
The phase 3 Medalist trial is a randomized double-blind placebo controlled study of luspatercept to treatment anemia in patients with MDS with ring sideroblasts who require red blood cell transfusion. The researchers will report significantly reduced transfusion burdens for luspatercept, compared with placebo.
“This is a practice-changing, pivotal trial in the field of MDS for the treatment of anemia,” Dr. Gupta said.
The findings will be presented at 2 p.m. PT on Sunday, Dec. 2 during the plenary session in Hall AB in the San Diego Convention Center (Abstract 1).
Also during the Sunday plenary session is a presentation on MPN therapy (Abstract 4). Researchers will present data on secreted mutant calreticulins as rogue cytokines trigger thrombopoietin receptor (TpoR) activation, specifically in CALR-mutated cells.
“This study investigates in to the mechanistic oncogenetic aspects of mutant calreticulin, and has potential for therapeutic approaches in the future,” Dr. Gupta said.
The ASH meeting will also feature the final analysis of the MPN-RC 112 consortium trial of pegylated interferon alfa-2a versus hydroxyurea for the treatment of high-risk polycythemia vera (PV) and essential thrombocythemia (ET). The researchers will report that the CR rates at 12 and 24 months were similar in patients treated with pegylated interferon alfa-2a and hydroxyurea, but pegylated interferon alfa-2a was associated with a higher rate of serious toxicities.
“There is a continuous debate on optimal first-line cytoreductive therapy for high risk PV/ET, and this is one of the first randomized study to answer this question,” Dr. Gupta said.
The findings will be presented at 7 a.m. PT on Monday, Dec. 3 in Grand Hall D at the Manchester Grand Hyatt San Diego (Abstract 577).
AML
For attendees interested in the latest developments in acute myeloid leukemia, Thomas Fischer, MD, of Otto-von-Guericke-University Magdeburg (Germany), highlighted three don’t-miss sessions.
In an analysis of a large cohort of FLT3-ITD mutated AML patients in the RATIFY trial, researchers looked at the prognostic impact of ITD insertion site.
“Interestingly, in this large cohort of 452 FLT3-ITD mutated AML, the negative prognostic impact of beta1-sheet insertion site of FLT3-ITD could be confirmed,” Dr. Fischer said. “Further analysis of a potential predictive effect on outcome of midostaurin treatment is ongoing and will be very interesting.”
The findings will be presented at 5 p.m. PT on Sunday, Dec. 2 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 435).
Another notable presentation features results from the phase 2 RADIUS trial, a randomized study comparing standard of care, with and without midostaurin, after allogeneic stem cell transplant in FLT3-ITD–mutated AML.
“Here, efficacy and toxicity of midostaurin was investigated in a [minimal residual disease] situation post-alloSCT,” Dr. Fischer said. “Interestingly, adding midostaurin to standard of care reduced the risk of relapse at 18 months post-alloSCT by 46%.”
The complete findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 662).
Dr. Fischer singled out another study looking at the efficacy and safety of single-agent quizartinib in patients with FLT3-ITD mutated AML. In this large, randomized trial the researchers noted a significant improvement in CR rates and survival benefit with the single agent FLT3 inhibitors, compared with salvage chemotherapy for patients with relapsed/refractory mutated AML.
The findings will be presented at 8 a.m. on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 563).
Notable posters
Iberia Romina Sosa, MD, PhD, of Baylor College of Medicine in Houston, suggested several posters worth visiting in the areas of thrombosis and bleeding.
Poster 1134 looks at the TNF-alpha driven inflammation and mitochondrial dysfunction in the platelet hyperreactivity of aging and MPN.
How do you know if your therapy for thrombotic thrombocytopenic purpura is working? Poster 3736 examines the measurement of cell-derived microparticles as a possible tool to monitor response to therapy.
You don’t have to be taking aspirin to have a bleeding profile characteristic with consumption of a cyclooxygenase inhibitor. Poster 1156 provides a first report of a platelet function disorder caused by autosomal recessive inheritance of PTGS1.
Poster 2477 takes a closer look at fitusiran, an antithrombin inhibitor, which improves thrombin generation in patients with hemophilia A or B. Protocol amendments for safety monitoring move fitusiran to phase 3 trials, Dr. Sosa said.
FDA approves emapalumab for primary HLH
The (HLH).
Emapalumab, an interferon gamma-blocking antibody, is approved to treat patients of all ages (newborn and older) with primary HLH who have refractory, recurrent, or progressive disease or who cannot tolerate conventional HLH therapy. Emapalumab is the first treatment to be FDA approved for primary HLH, and it is expected to be available in the United States in the first quarter of 2019. The FDA previously granted emapalumab priority review, breakthrough therapy designation, orphan drug designation, and rare pediatric disease designation. The FDA’s approval of emapalumab is based on results from a phase 2/3 trial (NCT01818492).
The trial included 34 patients, 27 of whom had refractory, recurrent, or progressive disease or could not tolerate conventional HLH therapy. Patients received emapalumab in combination with dexamethasone. At the end of treatment, 63% (17/27) of patients had achieved a response, which was defined as complete response (n = 7), partial response (n=8), or HLH improvement (n = 2). A total of 70% (n=19) of patients went on to hematopoietic stem cell transplant. The most common adverse events were infections (56%), hypertension (41%), infusion-related reactions (27%), and pyrexia (24%).
Results also are scheduled to be presented at the 2018 annual meeting of the American Society of Hematology in the late-breaker abstract session (Abstract LBA-6).
Emapalumab was developed by Novimmune SA. Sobi acquired global rights to the drug in August 2018.
The (HLH).
Emapalumab, an interferon gamma-blocking antibody, is approved to treat patients of all ages (newborn and older) with primary HLH who have refractory, recurrent, or progressive disease or who cannot tolerate conventional HLH therapy. Emapalumab is the first treatment to be FDA approved for primary HLH, and it is expected to be available in the United States in the first quarter of 2019. The FDA previously granted emapalumab priority review, breakthrough therapy designation, orphan drug designation, and rare pediatric disease designation. The FDA’s approval of emapalumab is based on results from a phase 2/3 trial (NCT01818492).
The trial included 34 patients, 27 of whom had refractory, recurrent, or progressive disease or could not tolerate conventional HLH therapy. Patients received emapalumab in combination with dexamethasone. At the end of treatment, 63% (17/27) of patients had achieved a response, which was defined as complete response (n = 7), partial response (n=8), or HLH improvement (n = 2). A total of 70% (n=19) of patients went on to hematopoietic stem cell transplant. The most common adverse events were infections (56%), hypertension (41%), infusion-related reactions (27%), and pyrexia (24%).
Results also are scheduled to be presented at the 2018 annual meeting of the American Society of Hematology in the late-breaker abstract session (Abstract LBA-6).
Emapalumab was developed by Novimmune SA. Sobi acquired global rights to the drug in August 2018.
The (HLH).
Emapalumab, an interferon gamma-blocking antibody, is approved to treat patients of all ages (newborn and older) with primary HLH who have refractory, recurrent, or progressive disease or who cannot tolerate conventional HLH therapy. Emapalumab is the first treatment to be FDA approved for primary HLH, and it is expected to be available in the United States in the first quarter of 2019. The FDA previously granted emapalumab priority review, breakthrough therapy designation, orphan drug designation, and rare pediatric disease designation. The FDA’s approval of emapalumab is based on results from a phase 2/3 trial (NCT01818492).
The trial included 34 patients, 27 of whom had refractory, recurrent, or progressive disease or could not tolerate conventional HLH therapy. Patients received emapalumab in combination with dexamethasone. At the end of treatment, 63% (17/27) of patients had achieved a response, which was defined as complete response (n = 7), partial response (n=8), or HLH improvement (n = 2). A total of 70% (n=19) of patients went on to hematopoietic stem cell transplant. The most common adverse events were infections (56%), hypertension (41%), infusion-related reactions (27%), and pyrexia (24%).
Results also are scheduled to be presented at the 2018 annual meeting of the American Society of Hematology in the late-breaker abstract session (Abstract LBA-6).
Emapalumab was developed by Novimmune SA. Sobi acquired global rights to the drug in August 2018.
FDA approves generic decitabine for MDS
The U.S. Food and Drug Administration has approved Lupin’s decitabine product, a generic version of Otsuka Pharmaceutical Co. Ltd.’s Dacogen, to treat patients with myelodysplastic syndromes (MDS).
Lupin’s decitabine for injection (50 mg, single-dose vial) is approved to treat patients with intermediate-1, intermediate-2, and high-risk MDS.
This includes previously treated, untreated, de novo, and secondary MDS of all French-American-British subtypes—refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia.
The U.S. Food and Drug Administration has approved Lupin’s decitabine product, a generic version of Otsuka Pharmaceutical Co. Ltd.’s Dacogen, to treat patients with myelodysplastic syndromes (MDS).
Lupin’s decitabine for injection (50 mg, single-dose vial) is approved to treat patients with intermediate-1, intermediate-2, and high-risk MDS.
This includes previously treated, untreated, de novo, and secondary MDS of all French-American-British subtypes—refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia.
The U.S. Food and Drug Administration has approved Lupin’s decitabine product, a generic version of Otsuka Pharmaceutical Co. Ltd.’s Dacogen, to treat patients with myelodysplastic syndromes (MDS).
Lupin’s decitabine for injection (50 mg, single-dose vial) is approved to treat patients with intermediate-1, intermediate-2, and high-risk MDS.
This includes previously treated, untreated, de novo, and secondary MDS of all French-American-British subtypes—refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia.