User login
Non-Invasive Blood and Stool CRC Screening Tests: Available Modalities and Their Clinical Application
Introduction
Colorectal cancer (CRC) screening significantly reduces CRC incidence and mortality, but only 65% of eligible individuals report being up-to-date with screening.1 Colonoscopy is the most widely used opportunistic screening method in the United States and is associated with many barriers to uptake. Providing patients a choice of colonoscopy and/or stool-based tests, improves screening adherence in randomized controlled trials.2,3 Non-invasive screening options have expanded from stool occult blood and multi-target DNA tests, to multi-target stool RNA tests, and novel blood-based tests, the latter only U.S. Food and Drug Administration (FDA) approved for patients who refuse colonoscopy and stool-based tests.
Stool Occult Blood Tests
Guaiac-based fecal occult blood testing (gFOBT) significantly reduces CRC mortality by 33%-35% when implemented on an annual or biennial basis.4,5 Fecal immunochemical testing (FIT) has supplanted gFOBT with advantages including independence from dietary restriction and medication-related interference, use of antibodies specific to human globin, and the need for only a single stool sample.
The most common threshold for a positive FIT in the U.S. is ≥ 20 micrograms (μg) of hemoglobin per gram (g) of stool. FIT is approved by the FDA as a qualitative positive or negative result based on a threshold value.6 A meta-analysis summarized test characteristics of commercially available FITs at various detection thresholds.7 The CRC sensitivity and specificity was 75% and 95% for ≥ 20 ug hemoglobin/g stool, and 91% and 90% for 10 ug hemoglobin/g stool, respectively. The sensitivity for advanced adenomas ranged from 25% at 20 μg/g to 40% at a 10 μg/g. Programmatic use of FIT in adults ages ≥ 50 years at 20 ug/g of stool, in cohort and case control studies, has been shown to significantly reduce CRC mortality by 33%-40% and advanced stage CRC by 34%.8,9
Over 57,000 average-risk individuals ages 50–69 years were randomized to biennial FIT or one-time colonoscopy and followed for 10 years.10 CRC mortality and incidence was similar between the groups: 0.22% with FIT vs. 0.24% with colonoscopy and 1.13% with FIT vs. 1.22% with colonoscopy, respectively. Thus, confirming biennial FIT screening is non-inferior to one-time colonoscopy in important CRC-related outcomes.
Multi-Target Stool Tests
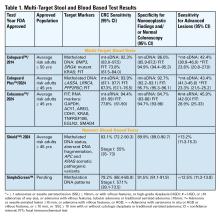
Two multitarget stool DNA tests (mt-sDNA) known as Cologuard™ and Cologuard Plus™ have been approved by the FDA. Both tests include a FIT (with a positivity threshold of 20 μg hemoglobin per gram of stool) combined with DNA methylation markers. The test result is qualitative, reported as a positive or negative. Cologuard™ markers include methylated BMP3, NDRG4, and mutant KRAS while Cologuard Plus™ assesses methylated LASS4, LRRC4, and PPP2R5C. The respective mt-sDNA tests were studied in 9989 of 12,776 and 20,176 of 26,758 average-risk individuals undergoing colonoscopy and the results were compared to a commercially available FIT (with a positivity threshold of 20 μg hemoglobin/gram of stool).11,12 In both trials, the sensitivity for CRC and advanced precancerous lesions was higher with the mt-sDNA tests compared to FIT but had a significantly lower specificity for advanced precancerous lesions versus FIT (see Table 1). An age-related decline in specificity was noted in both trials with mt-sDNA, a trend not observed with FIT. This reduction may be attributed to age-related DNA methylation.
Multi-Target Stool RNA Test
A multi-target stool RNA test (mt-sRNA) commercially available as ColoSense™ is FDA-approved. It combines FIT (at a positivity threshold of 20 μg hemoglobin/gram of stool) with RNA-based stool markers. The combined results of the RNA markers, FIT, and smoking status provide a qualitative single test result. In the trial, 8,920 adults aged ≥45 underwent the mt-sRNA test and FIT followed by colonoscopy (13). The mt-sRNA showed higher sensitivity for CRC than FIT (94.4% versus 77.8%) and advanced adenomas (45.9% versus 28.9%) but lower CRC specificity (84.7% vs 94.7%) (Table 1). Unlike mt-sDNA-based tests, mt-sRNA showed consistent performance across age groups, addressing concerns about age-related declines in specificity attributed to DNA methylation.
Blood-Based Tests
In 2014, the first blood-based (BBT) CRC screening test known as Epi proColon™ was FDA but not Centers for Medicare & Medicaid Services (CMS) approved for average-risk adults ≥50 years of age who are offered and refused other U.S Preventive Services Task Force (USPSTF) endorsed CRC screening tests. It is a qualitative test for detection of circulating methylated Septin 9 (mSeptin9). The accuracy of mSeptin9 to detect CRC was assessed in a subset of 7941 asymptomatic average risk adults undergoing screening colonoscopy.14 The sensitivity and specificity for CRC were 48% and 91.5%, respectively. The sensitivity for advanced adenomas was 11.2%. An increase in sensitivity to 63.9% and reduction in specificity to 88.4% for CRC was demonstrated in a sub-analysis of available samples where an additional (third) polymerase chain replicate was performed. Epi proColon™ is not currently reimbursed by Medicare and not endorsed in the latest USPSTF guidelines.
Technologic advancements have improved the detection of circulating tumor markers in the blood. The Shield™ BBT approved by the FDA in 2024 for average risk adults ≥ 45 years integrates three types of cfDNA data (epigenetic changes resulting in the aberrant methylation or fragmentation patterns, and genomic changes resulting in somatic mutations) into a positive or negative test result. In the trial, 22,877 average-risk, asymptomatic individuals ages 45–84 were enrolled and clinical validation was performed in 7,861 of the participants.15 The sensitivity for CRC was 83.1% which decreased to 55% for stage I tumors (see Table 1). CRC specificity was 89.6% and the sensitivity for advanced adenomas and large sessile serrated lesions was 13.2%.
Another BBT SimpleScreen™, which is not yet FDA-approved, analyzed circulating, cell-free DNA methylation patterns in 27,010 evaluable average-risk, asymptomatic adults ages 45–85 years undergoing screening colonoscopy.16 The sensitivity and specificity for CRC was 79.2% and 91.5%, respectively. Similar to Shield, the sensitivity for stage I CRC was low at 57.1%. The sensitivity for advanced precancerous lesions, a secondary endpoint, was 12.5% which did not meet the prespecified study criteria.
Effectiveness and Cost Effectiveness
Modeling studies have evaluated novel noninvasive CRC screening tests compared to FIT and colonoscopy.17-20 One compared a hypothetical BBT performed every 3 years that meets the minimum CMS threshold CRC sensitivity and specificity of 74% and 90%, respectively, to other established CRC screening tests beginning at age 45.17 Every 3-year BBT reduced CRC incidence and mortality by 40% and 52%, respectively compared to no screening. However, the reductions were much lower than yearly FIT (72% and 76%, respectively), every 10 year colonoscopy (79% and 81%, respectively), and triennial mt-sDNA (68% and 73%, respectively). The BBT resulted in fewer quality-adjusted life-years per person compared to the alternatives.
Additionally, FIT, colonoscopy, and mt-sDNA were less costly and more effective. Advanced precancerous lesion detection was a key measure for a test’s effectiveness. BBT characteristics would require a CRC sensitivity and specificity of >90% and 90%, respectively, and 80% sensitivity for advanced precancerous lesions at a cost of ≤$120–$140 to be cost-effective compared to FIT at comparable participation rates.
Another analysis simulated colorectal neoplasia progression and compared clinical effectiveness and cost between annual FIT, every 3 year stool mt-sRNA, every 3 year stool mt-sDNA tests, every 3 year stool Shield™; these outcomes were compared to colonoscopy every 10 years and no screening in adults ≥ age 45 over different adherence rates.19 At real-world adherence rates of 60%, colonoscopy prevented most CRC cases and associated deaths. FIT was the most cost-effective strategy at all adherence levels. Between the multi-target stool tests and Shield™, mt-sRNA was the most cost-effective. Compared to FIT, mt-sRNA reduced CRC cases and deaths by 1% and 14%.
The third study evaluated CRC incidence and mortality, quality-adjusted life-years and costs with annual FIT, colonoscopy every 10 years, mt- sDNA tests, mt-sRNA test, and BBTs.20 The latest mt-sDNA (Colguard plus™) and mt-sRNA achieved benefits approaching FIT but the Shield™ test was substantially less effective. The authors hypothesized that if 15% of the population substituted Shield™ for current effective CRC screening strategies, an increase in CRC deaths would occur and require 9-10% of the unscreened population to uptake screening with Shield to avert the increases in CRC deaths due to the substitution effect.
Clinical Implications
The effectiveness of non-invasive screening strategies depends on their diagnostic performance, adherence, and ensuring a timely colonoscopy after a positive test. Two claims-based studies found 47.9% and 49% of patients underwent follow-up colonoscopy within 6 months of an abnormal stool or BBT CRC screening test, respectively.21-22
Conclusions
Non-invasive stool mt-sDNA and mt-sRNA have higher effectiveness than the new BBTs. BBTs can lead to increased CRC mortality if substituted for the FDA and CMS-approved, USPSTF-endorsed, CRC screening modalities. If future BBTs increase their sensitivity for CRC (including early-stage CRC) and advanced precancerous lesions and decrease their cost, they may prove to have similar cost-effectiveness to stool-based tests. Currently, BBTs are not a substitute for colonoscopy or other stool tests and should be offered to patients who refuse other CRC screening modalities. A personalized, risk-adapted approach, paired with improved adherence and follow-up are essential to optimize the population-level impact of CRC screening and ensure equitable, effective cancer prevention.
Dr. Gupta is based at the Division of Gastroenterology and Hepatology, Department of Medicine, University of Maryland School of Medicine, Baltimore. Dr. Burke and Dr. Macaron are based at the Department of Gastroenterology, Hepatology, and Nutrition, Cleveland Clinic, Cleveland, Ohio. Dr. Gupta and Dr. Macaron declared no conflicts of interest in regard to this article. Dr. Burke declared research support from Emtora Biosciences. She is a current consultant for Lumabridge, and has been a consultant for Sebela and Almirall. She also disclosed support from Myriad, Genzyme, Ferring, Merck, Sharp and Dohme, Abbvie, Salix, and Natera.
References
1. Benavidez GA, Sedani AE, Felder TM, Asare M, Rogers CR. Rural-urban disparities and trends in cancer screening: an analysis of Behavioral Risk Factor Surveillance System data (2018-2022). JNCI Cancer Spectr. 2024 Nov 1;8(6):pkae113
2. Galoosian A, Dai H, Croymans D, et al. Population Health Colorectal Cancer Screening Strategies in Adults Aged 45 to 49 Years: A Randomized Clinical Trial. JAMA. 2025 Aug 4:e2512049. doi: 10.1001/jama.2025.12049. Epub ahead of print.
3. Pilonis ND, Bugajski M, Wieszczy P, et al. Participation in Competing Strategies for Colorectal Cancer Screening: A Randomized Health Services Study (PICCOLINO Study). Gastroenterology. 2021 Mar;160(4):1097-1105.
4. Shaukat A, Mongin SJ, Geisser MS, et al. Long-term mortality after screening for colorectal cancer. N Engl J Med. 2013;369(12):1106–1114.
5. Kronborg O, Fenger C, Olsen J, Jørgensen OD, Søndergaard O. Randomised study of screening for colorectal cancer with faecal-occult-blood test. Lancet. 1996 Nov 30;348(9040):1467-71. doi: 10.1016/S0140-6736(96)03430-7. PMID: 8942774.
6. Burke CA, Lieberman D, Feuerstein JD. AGA Clinical Practice Update on Approach to the Use of Noninvasive Colorectal Cancer Screening Options: Commentary. Gastroenterology. 2022 Mar;162(3):952-956. doi: 10.1053/j.gastro.2021.09.075. Epub 2022 Jan 28. PMID: 35094786.
7. Imperiale TF, Gruber RN Stump TE, et al. Performance characteristics of fecal immunochemical tests for colorectal cancer and advanced adenomatous polyps: a systematic review and meta-analysis. Ann Intern Med 2019; 170(5):319-329
8. Doubeni CA, Corley DA, Jensen CD, et al. Fecal Immunochemical Test Screening and Risk of Colorectal Cancer Death. JAMA Netw Open. 2024 Jul 1;7(7):e2423671. doi: 10.1001/jamanetworkopen.2024.23671.
9. Chiu HM, Jen GH, Wang YW, et al. Long-term effectiveness of faecal immunochemical test screening for proximal and distal colorectal cancers. Gut. 2021 Dec;70(12):2321-2329. doi: 10.1136/gutjnl-2020-322545. Epub 2021 Jan 25.
10. Castells A, Quintero E, Bujanda L, et al; COLONPREV study investigators. Effect of invitation to colonoscopy versus fecal immunochemical test screening on colorectal cancer mortality (COLONPREV): a pragmatic, randomised, controlled, non-inferiority trial. Lancet. 2025;405(10486):1231–1239
11. Imperiale TF, Ransohoff DF, Itzkowitz SH, et al. Multitarget stool DNA testing for colorectal-cancer screening. N Engl J Med. 2014;370(14):1287-1297
12. Imperiale TF, Porter K, Zella J, et al. Next-Generation Multitarget Stool DNA Test for Colorectal Cancer Screening. N Engl J Med. 2024 Mar 14;390(11):984-993
13. Barnell EK, Wurtzler EM, La Rocca J, et al. Multitarget Stool RNA Test for Colorectal Cancer Screening. JAMA. 2023 Nov 14;330(18):1760-1768.
14. Church TR, Wandell M, Lofton-Day C, et al. Prospective evaluation of methylated SEPT9 in plasma for detection of asymptomatic colorectal cancer. Gut 2014; 63:317–325.
15. Chung DC, Gray DM 2nd, Singh H, et al. A Cell-free DNA Blood-Based Test for Colorectal Cancer Screening. N Engl J Med. 2024 Mar 14;390(11):973-983.
16. Shaukat A, Burke CA, Chan AT, et al. Clinical Validation of a Circulating Tumor DNA-Based Blood Test to Screen for Colorectal Cancer. JAMA. 2025 Jul 1;334(1):56-63.
17. Ladabaum U, Mannalithara A, Weng Y, et al. Comparative Effectiveness and Cost-Effectiveness of Colorectal Cancer Screening with Blood-Based Biomarkers (Liquid Biopsy) vs Fecal Tests or Colonoscopy. Gastroenterology. 2024 Jul;167(2):378-391.
18. van den Puttelaar R, Nascimento de Lima P, Knudsen AB, et al. Effectiveness and cost-effectiveness of colorectal cancer screening with a blood test that meets the Centers for Medicare & Medicaid Services coverage decision. Gastroenterology 2024;167:368–377.
19. Shaukat A, Levin TR, Liang PS. Cost-effectiveness of Novel Noninvasive Screening Tests for Colorectal Neoplasia. Clin Gastroenterol Hepatol. 2025 Jun 23:S1542-3565(25)00525-7. doi: 10.1016/j.cgh.2025.06.006. Epub ahead of print. PMID: 40562290.
20. Ladabaum U, Mannalithara A, Schoen RE, Dominitz JA, Lieberman D. Projected Impact and Cost-Effectiveness of Novel Molecular Blood-Based or Stool-Based Screening Tests for Colorectal Cancer. Ann Intern Med. 2024 Dec;177(12):1610-1620.
20. Ciemins EL, Mohl JT, Moreno CA, Colangelo F, Smith RA, Barton M. Development of a Follow-Up Measure to Ensure Complete Screening for Colorectal Cancer. JAMA Netw Open. 2024 Mar 4;7(3):e242693. doi: 10.1001/jamanetworkopen.2024.2693.
21. Zaki TA, Zhang NJ, Forbes SP, Raymond VM, Das AK, May FP. Colonoscopic Follow-up After Abnormal Blood-Based Colorectal Cancer Screening Results. Gastroenterology. 2025 Jul 21:S0016-5085(25)05775-0. doi: 10.1053/j.gastro.2025.07.019. Epub ahead of print. PMID: 40744392.
Introduction
Colorectal cancer (CRC) screening significantly reduces CRC incidence and mortality, but only 65% of eligible individuals report being up-to-date with screening.1 Colonoscopy is the most widely used opportunistic screening method in the United States and is associated with many barriers to uptake. Providing patients a choice of colonoscopy and/or stool-based tests, improves screening adherence in randomized controlled trials.2,3 Non-invasive screening options have expanded from stool occult blood and multi-target DNA tests, to multi-target stool RNA tests, and novel blood-based tests, the latter only U.S. Food and Drug Administration (FDA) approved for patients who refuse colonoscopy and stool-based tests.
Stool Occult Blood Tests
Guaiac-based fecal occult blood testing (gFOBT) significantly reduces CRC mortality by 33%-35% when implemented on an annual or biennial basis.4,5 Fecal immunochemical testing (FIT) has supplanted gFOBT with advantages including independence from dietary restriction and medication-related interference, use of antibodies specific to human globin, and the need for only a single stool sample.
The most common threshold for a positive FIT in the U.S. is ≥ 20 micrograms (μg) of hemoglobin per gram (g) of stool. FIT is approved by the FDA as a qualitative positive or negative result based on a threshold value.6 A meta-analysis summarized test characteristics of commercially available FITs at various detection thresholds.7 The CRC sensitivity and specificity was 75% and 95% for ≥ 20 ug hemoglobin/g stool, and 91% and 90% for 10 ug hemoglobin/g stool, respectively. The sensitivity for advanced adenomas ranged from 25% at 20 μg/g to 40% at a 10 μg/g. Programmatic use of FIT in adults ages ≥ 50 years at 20 ug/g of stool, in cohort and case control studies, has been shown to significantly reduce CRC mortality by 33%-40% and advanced stage CRC by 34%.8,9
Over 57,000 average-risk individuals ages 50–69 years were randomized to biennial FIT or one-time colonoscopy and followed for 10 years.10 CRC mortality and incidence was similar between the groups: 0.22% with FIT vs. 0.24% with colonoscopy and 1.13% with FIT vs. 1.22% with colonoscopy, respectively. Thus, confirming biennial FIT screening is non-inferior to one-time colonoscopy in important CRC-related outcomes.
Multi-Target Stool Tests
Two multitarget stool DNA tests (mt-sDNA) known as Cologuard™ and Cologuard Plus™ have been approved by the FDA. Both tests include a FIT (with a positivity threshold of 20 μg hemoglobin per gram of stool) combined with DNA methylation markers. The test result is qualitative, reported as a positive or negative. Cologuard™ markers include methylated BMP3, NDRG4, and mutant KRAS while Cologuard Plus™ assesses methylated LASS4, LRRC4, and PPP2R5C. The respective mt-sDNA tests were studied in 9989 of 12,776 and 20,176 of 26,758 average-risk individuals undergoing colonoscopy and the results were compared to a commercially available FIT (with a positivity threshold of 20 μg hemoglobin/gram of stool).11,12 In both trials, the sensitivity for CRC and advanced precancerous lesions was higher with the mt-sDNA tests compared to FIT but had a significantly lower specificity for advanced precancerous lesions versus FIT (see Table 1). An age-related decline in specificity was noted in both trials with mt-sDNA, a trend not observed with FIT. This reduction may be attributed to age-related DNA methylation.
Multi-Target Stool RNA Test
A multi-target stool RNA test (mt-sRNA) commercially available as ColoSense™ is FDA-approved. It combines FIT (at a positivity threshold of 20 μg hemoglobin/gram of stool) with RNA-based stool markers. The combined results of the RNA markers, FIT, and smoking status provide a qualitative single test result. In the trial, 8,920 adults aged ≥45 underwent the mt-sRNA test and FIT followed by colonoscopy (13). The mt-sRNA showed higher sensitivity for CRC than FIT (94.4% versus 77.8%) and advanced adenomas (45.9% versus 28.9%) but lower CRC specificity (84.7% vs 94.7%) (Table 1). Unlike mt-sDNA-based tests, mt-sRNA showed consistent performance across age groups, addressing concerns about age-related declines in specificity attributed to DNA methylation.
Blood-Based Tests
In 2014, the first blood-based (BBT) CRC screening test known as Epi proColon™ was FDA but not Centers for Medicare & Medicaid Services (CMS) approved for average-risk adults ≥50 years of age who are offered and refused other U.S Preventive Services Task Force (USPSTF) endorsed CRC screening tests. It is a qualitative test for detection of circulating methylated Septin 9 (mSeptin9). The accuracy of mSeptin9 to detect CRC was assessed in a subset of 7941 asymptomatic average risk adults undergoing screening colonoscopy.14 The sensitivity and specificity for CRC were 48% and 91.5%, respectively. The sensitivity for advanced adenomas was 11.2%. An increase in sensitivity to 63.9% and reduction in specificity to 88.4% for CRC was demonstrated in a sub-analysis of available samples where an additional (third) polymerase chain replicate was performed. Epi proColon™ is not currently reimbursed by Medicare and not endorsed in the latest USPSTF guidelines.
Technologic advancements have improved the detection of circulating tumor markers in the blood. The Shield™ BBT approved by the FDA in 2024 for average risk adults ≥ 45 years integrates three types of cfDNA data (epigenetic changes resulting in the aberrant methylation or fragmentation patterns, and genomic changes resulting in somatic mutations) into a positive or negative test result. In the trial, 22,877 average-risk, asymptomatic individuals ages 45–84 were enrolled and clinical validation was performed in 7,861 of the participants.15 The sensitivity for CRC was 83.1% which decreased to 55% for stage I tumors (see Table 1). CRC specificity was 89.6% and the sensitivity for advanced adenomas and large sessile serrated lesions was 13.2%.
Another BBT SimpleScreen™, which is not yet FDA-approved, analyzed circulating, cell-free DNA methylation patterns in 27,010 evaluable average-risk, asymptomatic adults ages 45–85 years undergoing screening colonoscopy.16 The sensitivity and specificity for CRC was 79.2% and 91.5%, respectively. Similar to Shield, the sensitivity for stage I CRC was low at 57.1%. The sensitivity for advanced precancerous lesions, a secondary endpoint, was 12.5% which did not meet the prespecified study criteria.
Effectiveness and Cost Effectiveness
Modeling studies have evaluated novel noninvasive CRC screening tests compared to FIT and colonoscopy.17-20 One compared a hypothetical BBT performed every 3 years that meets the minimum CMS threshold CRC sensitivity and specificity of 74% and 90%, respectively, to other established CRC screening tests beginning at age 45.17 Every 3-year BBT reduced CRC incidence and mortality by 40% and 52%, respectively compared to no screening. However, the reductions were much lower than yearly FIT (72% and 76%, respectively), every 10 year colonoscopy (79% and 81%, respectively), and triennial mt-sDNA (68% and 73%, respectively). The BBT resulted in fewer quality-adjusted life-years per person compared to the alternatives.
Additionally, FIT, colonoscopy, and mt-sDNA were less costly and more effective. Advanced precancerous lesion detection was a key measure for a test’s effectiveness. BBT characteristics would require a CRC sensitivity and specificity of >90% and 90%, respectively, and 80% sensitivity for advanced precancerous lesions at a cost of ≤$120–$140 to be cost-effective compared to FIT at comparable participation rates.
Another analysis simulated colorectal neoplasia progression and compared clinical effectiveness and cost between annual FIT, every 3 year stool mt-sRNA, every 3 year stool mt-sDNA tests, every 3 year stool Shield™; these outcomes were compared to colonoscopy every 10 years and no screening in adults ≥ age 45 over different adherence rates.19 At real-world adherence rates of 60%, colonoscopy prevented most CRC cases and associated deaths. FIT was the most cost-effective strategy at all adherence levels. Between the multi-target stool tests and Shield™, mt-sRNA was the most cost-effective. Compared to FIT, mt-sRNA reduced CRC cases and deaths by 1% and 14%.
The third study evaluated CRC incidence and mortality, quality-adjusted life-years and costs with annual FIT, colonoscopy every 10 years, mt- sDNA tests, mt-sRNA test, and BBTs.20 The latest mt-sDNA (Colguard plus™) and mt-sRNA achieved benefits approaching FIT but the Shield™ test was substantially less effective. The authors hypothesized that if 15% of the population substituted Shield™ for current effective CRC screening strategies, an increase in CRC deaths would occur and require 9-10% of the unscreened population to uptake screening with Shield to avert the increases in CRC deaths due to the substitution effect.
Clinical Implications
The effectiveness of non-invasive screening strategies depends on their diagnostic performance, adherence, and ensuring a timely colonoscopy after a positive test. Two claims-based studies found 47.9% and 49% of patients underwent follow-up colonoscopy within 6 months of an abnormal stool or BBT CRC screening test, respectively.21-22
Conclusions
Non-invasive stool mt-sDNA and mt-sRNA have higher effectiveness than the new BBTs. BBTs can lead to increased CRC mortality if substituted for the FDA and CMS-approved, USPSTF-endorsed, CRC screening modalities. If future BBTs increase their sensitivity for CRC (including early-stage CRC) and advanced precancerous lesions and decrease their cost, they may prove to have similar cost-effectiveness to stool-based tests. Currently, BBTs are not a substitute for colonoscopy or other stool tests and should be offered to patients who refuse other CRC screening modalities. A personalized, risk-adapted approach, paired with improved adherence and follow-up are essential to optimize the population-level impact of CRC screening and ensure equitable, effective cancer prevention.
Dr. Gupta is based at the Division of Gastroenterology and Hepatology, Department of Medicine, University of Maryland School of Medicine, Baltimore. Dr. Burke and Dr. Macaron are based at the Department of Gastroenterology, Hepatology, and Nutrition, Cleveland Clinic, Cleveland, Ohio. Dr. Gupta and Dr. Macaron declared no conflicts of interest in regard to this article. Dr. Burke declared research support from Emtora Biosciences. She is a current consultant for Lumabridge, and has been a consultant for Sebela and Almirall. She also disclosed support from Myriad, Genzyme, Ferring, Merck, Sharp and Dohme, Abbvie, Salix, and Natera.
References
1. Benavidez GA, Sedani AE, Felder TM, Asare M, Rogers CR. Rural-urban disparities and trends in cancer screening: an analysis of Behavioral Risk Factor Surveillance System data (2018-2022). JNCI Cancer Spectr. 2024 Nov 1;8(6):pkae113
2. Galoosian A, Dai H, Croymans D, et al. Population Health Colorectal Cancer Screening Strategies in Adults Aged 45 to 49 Years: A Randomized Clinical Trial. JAMA. 2025 Aug 4:e2512049. doi: 10.1001/jama.2025.12049. Epub ahead of print.
3. Pilonis ND, Bugajski M, Wieszczy P, et al. Participation in Competing Strategies for Colorectal Cancer Screening: A Randomized Health Services Study (PICCOLINO Study). Gastroenterology. 2021 Mar;160(4):1097-1105.
4. Shaukat A, Mongin SJ, Geisser MS, et al. Long-term mortality after screening for colorectal cancer. N Engl J Med. 2013;369(12):1106–1114.
5. Kronborg O, Fenger C, Olsen J, Jørgensen OD, Søndergaard O. Randomised study of screening for colorectal cancer with faecal-occult-blood test. Lancet. 1996 Nov 30;348(9040):1467-71. doi: 10.1016/S0140-6736(96)03430-7. PMID: 8942774.
6. Burke CA, Lieberman D, Feuerstein JD. AGA Clinical Practice Update on Approach to the Use of Noninvasive Colorectal Cancer Screening Options: Commentary. Gastroenterology. 2022 Mar;162(3):952-956. doi: 10.1053/j.gastro.2021.09.075. Epub 2022 Jan 28. PMID: 35094786.
7. Imperiale TF, Gruber RN Stump TE, et al. Performance characteristics of fecal immunochemical tests for colorectal cancer and advanced adenomatous polyps: a systematic review and meta-analysis. Ann Intern Med 2019; 170(5):319-329
8. Doubeni CA, Corley DA, Jensen CD, et al. Fecal Immunochemical Test Screening and Risk of Colorectal Cancer Death. JAMA Netw Open. 2024 Jul 1;7(7):e2423671. doi: 10.1001/jamanetworkopen.2024.23671.
9. Chiu HM, Jen GH, Wang YW, et al. Long-term effectiveness of faecal immunochemical test screening for proximal and distal colorectal cancers. Gut. 2021 Dec;70(12):2321-2329. doi: 10.1136/gutjnl-2020-322545. Epub 2021 Jan 25.
10. Castells A, Quintero E, Bujanda L, et al; COLONPREV study investigators. Effect of invitation to colonoscopy versus fecal immunochemical test screening on colorectal cancer mortality (COLONPREV): a pragmatic, randomised, controlled, non-inferiority trial. Lancet. 2025;405(10486):1231–1239
11. Imperiale TF, Ransohoff DF, Itzkowitz SH, et al. Multitarget stool DNA testing for colorectal-cancer screening. N Engl J Med. 2014;370(14):1287-1297
12. Imperiale TF, Porter K, Zella J, et al. Next-Generation Multitarget Stool DNA Test for Colorectal Cancer Screening. N Engl J Med. 2024 Mar 14;390(11):984-993
13. Barnell EK, Wurtzler EM, La Rocca J, et al. Multitarget Stool RNA Test for Colorectal Cancer Screening. JAMA. 2023 Nov 14;330(18):1760-1768.
14. Church TR, Wandell M, Lofton-Day C, et al. Prospective evaluation of methylated SEPT9 in plasma for detection of asymptomatic colorectal cancer. Gut 2014; 63:317–325.
15. Chung DC, Gray DM 2nd, Singh H, et al. A Cell-free DNA Blood-Based Test for Colorectal Cancer Screening. N Engl J Med. 2024 Mar 14;390(11):973-983.
16. Shaukat A, Burke CA, Chan AT, et al. Clinical Validation of a Circulating Tumor DNA-Based Blood Test to Screen for Colorectal Cancer. JAMA. 2025 Jul 1;334(1):56-63.
17. Ladabaum U, Mannalithara A, Weng Y, et al. Comparative Effectiveness and Cost-Effectiveness of Colorectal Cancer Screening with Blood-Based Biomarkers (Liquid Biopsy) vs Fecal Tests or Colonoscopy. Gastroenterology. 2024 Jul;167(2):378-391.
18. van den Puttelaar R, Nascimento de Lima P, Knudsen AB, et al. Effectiveness and cost-effectiveness of colorectal cancer screening with a blood test that meets the Centers for Medicare & Medicaid Services coverage decision. Gastroenterology 2024;167:368–377.
19. Shaukat A, Levin TR, Liang PS. Cost-effectiveness of Novel Noninvasive Screening Tests for Colorectal Neoplasia. Clin Gastroenterol Hepatol. 2025 Jun 23:S1542-3565(25)00525-7. doi: 10.1016/j.cgh.2025.06.006. Epub ahead of print. PMID: 40562290.
20. Ladabaum U, Mannalithara A, Schoen RE, Dominitz JA, Lieberman D. Projected Impact and Cost-Effectiveness of Novel Molecular Blood-Based or Stool-Based Screening Tests for Colorectal Cancer. Ann Intern Med. 2024 Dec;177(12):1610-1620.
20. Ciemins EL, Mohl JT, Moreno CA, Colangelo F, Smith RA, Barton M. Development of a Follow-Up Measure to Ensure Complete Screening for Colorectal Cancer. JAMA Netw Open. 2024 Mar 4;7(3):e242693. doi: 10.1001/jamanetworkopen.2024.2693.
21. Zaki TA, Zhang NJ, Forbes SP, Raymond VM, Das AK, May FP. Colonoscopic Follow-up After Abnormal Blood-Based Colorectal Cancer Screening Results. Gastroenterology. 2025 Jul 21:S0016-5085(25)05775-0. doi: 10.1053/j.gastro.2025.07.019. Epub ahead of print. PMID: 40744392.
Introduction
Colorectal cancer (CRC) screening significantly reduces CRC incidence and mortality, but only 65% of eligible individuals report being up-to-date with screening.1 Colonoscopy is the most widely used opportunistic screening method in the United States and is associated with many barriers to uptake. Providing patients a choice of colonoscopy and/or stool-based tests, improves screening adherence in randomized controlled trials.2,3 Non-invasive screening options have expanded from stool occult blood and multi-target DNA tests, to multi-target stool RNA tests, and novel blood-based tests, the latter only U.S. Food and Drug Administration (FDA) approved for patients who refuse colonoscopy and stool-based tests.
Stool Occult Blood Tests
Guaiac-based fecal occult blood testing (gFOBT) significantly reduces CRC mortality by 33%-35% when implemented on an annual or biennial basis.4,5 Fecal immunochemical testing (FIT) has supplanted gFOBT with advantages including independence from dietary restriction and medication-related interference, use of antibodies specific to human globin, and the need for only a single stool sample.
The most common threshold for a positive FIT in the U.S. is ≥ 20 micrograms (μg) of hemoglobin per gram (g) of stool. FIT is approved by the FDA as a qualitative positive or negative result based on a threshold value.6 A meta-analysis summarized test characteristics of commercially available FITs at various detection thresholds.7 The CRC sensitivity and specificity was 75% and 95% for ≥ 20 ug hemoglobin/g stool, and 91% and 90% for 10 ug hemoglobin/g stool, respectively. The sensitivity for advanced adenomas ranged from 25% at 20 μg/g to 40% at a 10 μg/g. Programmatic use of FIT in adults ages ≥ 50 years at 20 ug/g of stool, in cohort and case control studies, has been shown to significantly reduce CRC mortality by 33%-40% and advanced stage CRC by 34%.8,9
Over 57,000 average-risk individuals ages 50–69 years were randomized to biennial FIT or one-time colonoscopy and followed for 10 years.10 CRC mortality and incidence was similar between the groups: 0.22% with FIT vs. 0.24% with colonoscopy and 1.13% with FIT vs. 1.22% with colonoscopy, respectively. Thus, confirming biennial FIT screening is non-inferior to one-time colonoscopy in important CRC-related outcomes.
Multi-Target Stool Tests
Two multitarget stool DNA tests (mt-sDNA) known as Cologuard™ and Cologuard Plus™ have been approved by the FDA. Both tests include a FIT (with a positivity threshold of 20 μg hemoglobin per gram of stool) combined with DNA methylation markers. The test result is qualitative, reported as a positive or negative. Cologuard™ markers include methylated BMP3, NDRG4, and mutant KRAS while Cologuard Plus™ assesses methylated LASS4, LRRC4, and PPP2R5C. The respective mt-sDNA tests were studied in 9989 of 12,776 and 20,176 of 26,758 average-risk individuals undergoing colonoscopy and the results were compared to a commercially available FIT (with a positivity threshold of 20 μg hemoglobin/gram of stool).11,12 In both trials, the sensitivity for CRC and advanced precancerous lesions was higher with the mt-sDNA tests compared to FIT but had a significantly lower specificity for advanced precancerous lesions versus FIT (see Table 1). An age-related decline in specificity was noted in both trials with mt-sDNA, a trend not observed with FIT. This reduction may be attributed to age-related DNA methylation.
Multi-Target Stool RNA Test
A multi-target stool RNA test (mt-sRNA) commercially available as ColoSense™ is FDA-approved. It combines FIT (at a positivity threshold of 20 μg hemoglobin/gram of stool) with RNA-based stool markers. The combined results of the RNA markers, FIT, and smoking status provide a qualitative single test result. In the trial, 8,920 adults aged ≥45 underwent the mt-sRNA test and FIT followed by colonoscopy (13). The mt-sRNA showed higher sensitivity for CRC than FIT (94.4% versus 77.8%) and advanced adenomas (45.9% versus 28.9%) but lower CRC specificity (84.7% vs 94.7%) (Table 1). Unlike mt-sDNA-based tests, mt-sRNA showed consistent performance across age groups, addressing concerns about age-related declines in specificity attributed to DNA methylation.
Blood-Based Tests
In 2014, the first blood-based (BBT) CRC screening test known as Epi proColon™ was FDA but not Centers for Medicare & Medicaid Services (CMS) approved for average-risk adults ≥50 years of age who are offered and refused other U.S Preventive Services Task Force (USPSTF) endorsed CRC screening tests. It is a qualitative test for detection of circulating methylated Septin 9 (mSeptin9). The accuracy of mSeptin9 to detect CRC was assessed in a subset of 7941 asymptomatic average risk adults undergoing screening colonoscopy.14 The sensitivity and specificity for CRC were 48% and 91.5%, respectively. The sensitivity for advanced adenomas was 11.2%. An increase in sensitivity to 63.9% and reduction in specificity to 88.4% for CRC was demonstrated in a sub-analysis of available samples where an additional (third) polymerase chain replicate was performed. Epi proColon™ is not currently reimbursed by Medicare and not endorsed in the latest USPSTF guidelines.
Technologic advancements have improved the detection of circulating tumor markers in the blood. The Shield™ BBT approved by the FDA in 2024 for average risk adults ≥ 45 years integrates three types of cfDNA data (epigenetic changes resulting in the aberrant methylation or fragmentation patterns, and genomic changes resulting in somatic mutations) into a positive or negative test result. In the trial, 22,877 average-risk, asymptomatic individuals ages 45–84 were enrolled and clinical validation was performed in 7,861 of the participants.15 The sensitivity for CRC was 83.1% which decreased to 55% for stage I tumors (see Table 1). CRC specificity was 89.6% and the sensitivity for advanced adenomas and large sessile serrated lesions was 13.2%.
Another BBT SimpleScreen™, which is not yet FDA-approved, analyzed circulating, cell-free DNA methylation patterns in 27,010 evaluable average-risk, asymptomatic adults ages 45–85 years undergoing screening colonoscopy.16 The sensitivity and specificity for CRC was 79.2% and 91.5%, respectively. Similar to Shield, the sensitivity for stage I CRC was low at 57.1%. The sensitivity for advanced precancerous lesions, a secondary endpoint, was 12.5% which did not meet the prespecified study criteria.
Effectiveness and Cost Effectiveness
Modeling studies have evaluated novel noninvasive CRC screening tests compared to FIT and colonoscopy.17-20 One compared a hypothetical BBT performed every 3 years that meets the minimum CMS threshold CRC sensitivity and specificity of 74% and 90%, respectively, to other established CRC screening tests beginning at age 45.17 Every 3-year BBT reduced CRC incidence and mortality by 40% and 52%, respectively compared to no screening. However, the reductions were much lower than yearly FIT (72% and 76%, respectively), every 10 year colonoscopy (79% and 81%, respectively), and triennial mt-sDNA (68% and 73%, respectively). The BBT resulted in fewer quality-adjusted life-years per person compared to the alternatives.
Additionally, FIT, colonoscopy, and mt-sDNA were less costly and more effective. Advanced precancerous lesion detection was a key measure for a test’s effectiveness. BBT characteristics would require a CRC sensitivity and specificity of >90% and 90%, respectively, and 80% sensitivity for advanced precancerous lesions at a cost of ≤$120–$140 to be cost-effective compared to FIT at comparable participation rates.
Another analysis simulated colorectal neoplasia progression and compared clinical effectiveness and cost between annual FIT, every 3 year stool mt-sRNA, every 3 year stool mt-sDNA tests, every 3 year stool Shield™; these outcomes were compared to colonoscopy every 10 years and no screening in adults ≥ age 45 over different adherence rates.19 At real-world adherence rates of 60%, colonoscopy prevented most CRC cases and associated deaths. FIT was the most cost-effective strategy at all adherence levels. Between the multi-target stool tests and Shield™, mt-sRNA was the most cost-effective. Compared to FIT, mt-sRNA reduced CRC cases and deaths by 1% and 14%.
The third study evaluated CRC incidence and mortality, quality-adjusted life-years and costs with annual FIT, colonoscopy every 10 years, mt- sDNA tests, mt-sRNA test, and BBTs.20 The latest mt-sDNA (Colguard plus™) and mt-sRNA achieved benefits approaching FIT but the Shield™ test was substantially less effective. The authors hypothesized that if 15% of the population substituted Shield™ for current effective CRC screening strategies, an increase in CRC deaths would occur and require 9-10% of the unscreened population to uptake screening with Shield to avert the increases in CRC deaths due to the substitution effect.
Clinical Implications
The effectiveness of non-invasive screening strategies depends on their diagnostic performance, adherence, and ensuring a timely colonoscopy after a positive test. Two claims-based studies found 47.9% and 49% of patients underwent follow-up colonoscopy within 6 months of an abnormal stool or BBT CRC screening test, respectively.21-22
Conclusions
Non-invasive stool mt-sDNA and mt-sRNA have higher effectiveness than the new BBTs. BBTs can lead to increased CRC mortality if substituted for the FDA and CMS-approved, USPSTF-endorsed, CRC screening modalities. If future BBTs increase their sensitivity for CRC (including early-stage CRC) and advanced precancerous lesions and decrease their cost, they may prove to have similar cost-effectiveness to stool-based tests. Currently, BBTs are not a substitute for colonoscopy or other stool tests and should be offered to patients who refuse other CRC screening modalities. A personalized, risk-adapted approach, paired with improved adherence and follow-up are essential to optimize the population-level impact of CRC screening and ensure equitable, effective cancer prevention.
Dr. Gupta is based at the Division of Gastroenterology and Hepatology, Department of Medicine, University of Maryland School of Medicine, Baltimore. Dr. Burke and Dr. Macaron are based at the Department of Gastroenterology, Hepatology, and Nutrition, Cleveland Clinic, Cleveland, Ohio. Dr. Gupta and Dr. Macaron declared no conflicts of interest in regard to this article. Dr. Burke declared research support from Emtora Biosciences. She is a current consultant for Lumabridge, and has been a consultant for Sebela and Almirall. She also disclosed support from Myriad, Genzyme, Ferring, Merck, Sharp and Dohme, Abbvie, Salix, and Natera.
References
1. Benavidez GA, Sedani AE, Felder TM, Asare M, Rogers CR. Rural-urban disparities and trends in cancer screening: an analysis of Behavioral Risk Factor Surveillance System data (2018-2022). JNCI Cancer Spectr. 2024 Nov 1;8(6):pkae113
2. Galoosian A, Dai H, Croymans D, et al. Population Health Colorectal Cancer Screening Strategies in Adults Aged 45 to 49 Years: A Randomized Clinical Trial. JAMA. 2025 Aug 4:e2512049. doi: 10.1001/jama.2025.12049. Epub ahead of print.
3. Pilonis ND, Bugajski M, Wieszczy P, et al. Participation in Competing Strategies for Colorectal Cancer Screening: A Randomized Health Services Study (PICCOLINO Study). Gastroenterology. 2021 Mar;160(4):1097-1105.
4. Shaukat A, Mongin SJ, Geisser MS, et al. Long-term mortality after screening for colorectal cancer. N Engl J Med. 2013;369(12):1106–1114.
5. Kronborg O, Fenger C, Olsen J, Jørgensen OD, Søndergaard O. Randomised study of screening for colorectal cancer with faecal-occult-blood test. Lancet. 1996 Nov 30;348(9040):1467-71. doi: 10.1016/S0140-6736(96)03430-7. PMID: 8942774.
6. Burke CA, Lieberman D, Feuerstein JD. AGA Clinical Practice Update on Approach to the Use of Noninvasive Colorectal Cancer Screening Options: Commentary. Gastroenterology. 2022 Mar;162(3):952-956. doi: 10.1053/j.gastro.2021.09.075. Epub 2022 Jan 28. PMID: 35094786.
7. Imperiale TF, Gruber RN Stump TE, et al. Performance characteristics of fecal immunochemical tests for colorectal cancer and advanced adenomatous polyps: a systematic review and meta-analysis. Ann Intern Med 2019; 170(5):319-329
8. Doubeni CA, Corley DA, Jensen CD, et al. Fecal Immunochemical Test Screening and Risk of Colorectal Cancer Death. JAMA Netw Open. 2024 Jul 1;7(7):e2423671. doi: 10.1001/jamanetworkopen.2024.23671.
9. Chiu HM, Jen GH, Wang YW, et al. Long-term effectiveness of faecal immunochemical test screening for proximal and distal colorectal cancers. Gut. 2021 Dec;70(12):2321-2329. doi: 10.1136/gutjnl-2020-322545. Epub 2021 Jan 25.
10. Castells A, Quintero E, Bujanda L, et al; COLONPREV study investigators. Effect of invitation to colonoscopy versus fecal immunochemical test screening on colorectal cancer mortality (COLONPREV): a pragmatic, randomised, controlled, non-inferiority trial. Lancet. 2025;405(10486):1231–1239
11. Imperiale TF, Ransohoff DF, Itzkowitz SH, et al. Multitarget stool DNA testing for colorectal-cancer screening. N Engl J Med. 2014;370(14):1287-1297
12. Imperiale TF, Porter K, Zella J, et al. Next-Generation Multitarget Stool DNA Test for Colorectal Cancer Screening. N Engl J Med. 2024 Mar 14;390(11):984-993
13. Barnell EK, Wurtzler EM, La Rocca J, et al. Multitarget Stool RNA Test for Colorectal Cancer Screening. JAMA. 2023 Nov 14;330(18):1760-1768.
14. Church TR, Wandell M, Lofton-Day C, et al. Prospective evaluation of methylated SEPT9 in plasma for detection of asymptomatic colorectal cancer. Gut 2014; 63:317–325.
15. Chung DC, Gray DM 2nd, Singh H, et al. A Cell-free DNA Blood-Based Test for Colorectal Cancer Screening. N Engl J Med. 2024 Mar 14;390(11):973-983.
16. Shaukat A, Burke CA, Chan AT, et al. Clinical Validation of a Circulating Tumor DNA-Based Blood Test to Screen for Colorectal Cancer. JAMA. 2025 Jul 1;334(1):56-63.
17. Ladabaum U, Mannalithara A, Weng Y, et al. Comparative Effectiveness and Cost-Effectiveness of Colorectal Cancer Screening with Blood-Based Biomarkers (Liquid Biopsy) vs Fecal Tests or Colonoscopy. Gastroenterology. 2024 Jul;167(2):378-391.
18. van den Puttelaar R, Nascimento de Lima P, Knudsen AB, et al. Effectiveness and cost-effectiveness of colorectal cancer screening with a blood test that meets the Centers for Medicare & Medicaid Services coverage decision. Gastroenterology 2024;167:368–377.
19. Shaukat A, Levin TR, Liang PS. Cost-effectiveness of Novel Noninvasive Screening Tests for Colorectal Neoplasia. Clin Gastroenterol Hepatol. 2025 Jun 23:S1542-3565(25)00525-7. doi: 10.1016/j.cgh.2025.06.006. Epub ahead of print. PMID: 40562290.
20. Ladabaum U, Mannalithara A, Schoen RE, Dominitz JA, Lieberman D. Projected Impact and Cost-Effectiveness of Novel Molecular Blood-Based or Stool-Based Screening Tests for Colorectal Cancer. Ann Intern Med. 2024 Dec;177(12):1610-1620.
20. Ciemins EL, Mohl JT, Moreno CA, Colangelo F, Smith RA, Barton M. Development of a Follow-Up Measure to Ensure Complete Screening for Colorectal Cancer. JAMA Netw Open. 2024 Mar 4;7(3):e242693. doi: 10.1001/jamanetworkopen.2024.2693.
21. Zaki TA, Zhang NJ, Forbes SP, Raymond VM, Das AK, May FP. Colonoscopic Follow-up After Abnormal Blood-Based Colorectal Cancer Screening Results. Gastroenterology. 2025 Jul 21:S0016-5085(25)05775-0. doi: 10.1053/j.gastro.2025.07.019. Epub ahead of print. PMID: 40744392.
Patients With a Positive FIT Fail to Get Follow-Up Colonoscopies
PHOENIX — Patients with or without polyp removal in an index colonoscopy commonly receive follow-up surveillance with a fecal immunochemical test (FIT), yet many of these patients do not receive a recommended colonoscopy after a positive FIT.
“In this large US study, we found interval FITs are frequently performed in patients with and without prior polypectomy,” said first author Natalie J. Wilson, MD, of the University of Minnesota in Minneapolis, while presenting the findings at the American College of Gastroenterology (ACG) 2025 Annual Scientific Meeting.
“ and colorectal cancer, regardless of polypectomy history,” Wilson said.
Guideline recommendations stress the need for follow-up surveillance with a colonoscopy, particularly in patients who have had a prior polypectomy, because of the higher risk.
Reasons patients may instead turn to FIT may include cost or other factors, she said.
To determine just how often that happens, how having a previous polypectomy affects FIT results, and how adherent patients are to follow up if a FIT result is positive, Wilson and her colleagues evaluated data from nearly 4.8 million individuals in the Veterans Health Administration Corporate Data Warehouse who underwent colonoscopy between 2000 and 2024.
Of the patients, 10.9% were found to have subsequently received interval FIT within 10 years of the index colonoscopy, and of those patients, nearly half (49.9%) had received a polypectomy at the index colonoscopy.
The average time from the colonoscopy/polypectomy to the interval FIT was 5.9 years (5.6 years in the polypectomy group vs 6.2 years in the non-polypectomy group).
Among the FIT screenings, results were positive in 17.2% of post-polypectomy patients and 14.1% of patients with no prior polypectomy, indicating a history of polypectomy to be predictive of a positive interval FIT (odds ratio [OR], 1.12; P < .0001).
Notably, while a follow-up colonoscopy is considered essential following a positive FIT result — and having a previous polypectomy should add further urgency to the matter — the study showed only 50.4% of those who had an earlier polypectomy went on to receive the recommended follow-up colonoscopy after a positive follow-up FIT, and the rate was 49.3% among those who had not received a polypectomy (P = .001).
For those who did receive a follow-up colonoscopy after a positive FIT, the duration of time to receiving the colonoscopy was longer among those who had a prior polypectomy, at 2.9 months compared with 2.5 months in the non-polypectomy group (P < .001).
Colonoscopy results following a positive FIT showed higher rates of detections among patients who had prior polypectomies than among those with no prior polypectomy, including tubular adenomas (54.7% vs 45.8%), tubulovillous adenomas (5.6% vs 4.7%), adenomas with high-grade dysplasia (0.8% vs 0.7%), sessile serrated lesions (3.52% vs 2.4%), advanced colorectal neoplasia (9.2% vs 7.9%), and colorectal cancer (3.3% vs 3.0%).
However, a prior polypectomy was not independently predictive of colorectal cancer (OR, 0.96; P = .65) or advanced colorectal neoplasia (OR, 0.97; P = .57) in the post-colonoscopy interval FIT.
The findings underscore that “positive results carried a high risk of advanced neoplasia or cancer, irrespective of prior polypectomy history,” Wilson said.
Clinicians Must ‘Do a Better Job’
Commenting on the study, William D. Chey, MD, AGAF, chief of the Division of Gastroenterology & Hepatology at the University of Michigan in Ann Arbor, noted that the study “addresses one of the biggest challenges we face as a profession, which is making sure that patients who have a positive stool test get a colonoscopy.”
He noted that the low rate of just 50% of recipients of positive FITs going on to receive a colonoscopy is consistent with what is observed in other trials.
“Other data suggests that the rate might even be significantly higher — at 70%-80%, depending upon the population and the test,” Chey told Medscape Medical News.
Reasons for the failure to receive the follow-up testing range from income restrictions (due to the high cost of a colonoscopy, especially if not covered by insurance), education, speaking a foreign language, and other factors, he said.
The relatively high rates of colon cancers detected by FIT in the study, in those with and without a prior polypectomy, along with findings from other studies “should raise questions about whether there might be a role for FIT testing in addition to colonoscopy.” However, much stronger evidence would be needed, Chey noted.
In the meantime, a key issue is “how do we do a better job of making sure that individuals who have a positive FIT test get a colonoscopy,” he said.
“I think a lot of this is going to come down to how it’s done at the primary care level.”
Chey added that in that, and any other setting, “the main message that needs to get out to people who are undergoing stool-based screening is that the stool test is only the first part of the screening process, and if it’s positive, a follow-up colonoscopy must be performed.”
“Otherwise, the stool-based test is of no value.”
Wilson had no disclosures to report. Chey’s disclosures included consulting and/or other relationships with Ardelyx, Atmo, Biomerica, Commonwealth Diagnostics International, Corprata, Dieta, Evinature, Food Marble, Gemelli, Kiwi BioScience, Modify Health, Nestlé, Phathom, Redhill, Salix/Valeant, Takeda, and Vibrant.
A version of this article appeared on Medscape.com .
PHOENIX — Patients with or without polyp removal in an index colonoscopy commonly receive follow-up surveillance with a fecal immunochemical test (FIT), yet many of these patients do not receive a recommended colonoscopy after a positive FIT.
“In this large US study, we found interval FITs are frequently performed in patients with and without prior polypectomy,” said first author Natalie J. Wilson, MD, of the University of Minnesota in Minneapolis, while presenting the findings at the American College of Gastroenterology (ACG) 2025 Annual Scientific Meeting.
“ and colorectal cancer, regardless of polypectomy history,” Wilson said.
Guideline recommendations stress the need for follow-up surveillance with a colonoscopy, particularly in patients who have had a prior polypectomy, because of the higher risk.
Reasons patients may instead turn to FIT may include cost or other factors, she said.
To determine just how often that happens, how having a previous polypectomy affects FIT results, and how adherent patients are to follow up if a FIT result is positive, Wilson and her colleagues evaluated data from nearly 4.8 million individuals in the Veterans Health Administration Corporate Data Warehouse who underwent colonoscopy between 2000 and 2024.
Of the patients, 10.9% were found to have subsequently received interval FIT within 10 years of the index colonoscopy, and of those patients, nearly half (49.9%) had received a polypectomy at the index colonoscopy.
The average time from the colonoscopy/polypectomy to the interval FIT was 5.9 years (5.6 years in the polypectomy group vs 6.2 years in the non-polypectomy group).
Among the FIT screenings, results were positive in 17.2% of post-polypectomy patients and 14.1% of patients with no prior polypectomy, indicating a history of polypectomy to be predictive of a positive interval FIT (odds ratio [OR], 1.12; P < .0001).
Notably, while a follow-up colonoscopy is considered essential following a positive FIT result — and having a previous polypectomy should add further urgency to the matter — the study showed only 50.4% of those who had an earlier polypectomy went on to receive the recommended follow-up colonoscopy after a positive follow-up FIT, and the rate was 49.3% among those who had not received a polypectomy (P = .001).
For those who did receive a follow-up colonoscopy after a positive FIT, the duration of time to receiving the colonoscopy was longer among those who had a prior polypectomy, at 2.9 months compared with 2.5 months in the non-polypectomy group (P < .001).
Colonoscopy results following a positive FIT showed higher rates of detections among patients who had prior polypectomies than among those with no prior polypectomy, including tubular adenomas (54.7% vs 45.8%), tubulovillous adenomas (5.6% vs 4.7%), adenomas with high-grade dysplasia (0.8% vs 0.7%), sessile serrated lesions (3.52% vs 2.4%), advanced colorectal neoplasia (9.2% vs 7.9%), and colorectal cancer (3.3% vs 3.0%).
However, a prior polypectomy was not independently predictive of colorectal cancer (OR, 0.96; P = .65) or advanced colorectal neoplasia (OR, 0.97; P = .57) in the post-colonoscopy interval FIT.
The findings underscore that “positive results carried a high risk of advanced neoplasia or cancer, irrespective of prior polypectomy history,” Wilson said.
Clinicians Must ‘Do a Better Job’
Commenting on the study, William D. Chey, MD, AGAF, chief of the Division of Gastroenterology & Hepatology at the University of Michigan in Ann Arbor, noted that the study “addresses one of the biggest challenges we face as a profession, which is making sure that patients who have a positive stool test get a colonoscopy.”
He noted that the low rate of just 50% of recipients of positive FITs going on to receive a colonoscopy is consistent with what is observed in other trials.
“Other data suggests that the rate might even be significantly higher — at 70%-80%, depending upon the population and the test,” Chey told Medscape Medical News.
Reasons for the failure to receive the follow-up testing range from income restrictions (due to the high cost of a colonoscopy, especially if not covered by insurance), education, speaking a foreign language, and other factors, he said.
The relatively high rates of colon cancers detected by FIT in the study, in those with and without a prior polypectomy, along with findings from other studies “should raise questions about whether there might be a role for FIT testing in addition to colonoscopy.” However, much stronger evidence would be needed, Chey noted.
In the meantime, a key issue is “how do we do a better job of making sure that individuals who have a positive FIT test get a colonoscopy,” he said.
“I think a lot of this is going to come down to how it’s done at the primary care level.”
Chey added that in that, and any other setting, “the main message that needs to get out to people who are undergoing stool-based screening is that the stool test is only the first part of the screening process, and if it’s positive, a follow-up colonoscopy must be performed.”
“Otherwise, the stool-based test is of no value.”
Wilson had no disclosures to report. Chey’s disclosures included consulting and/or other relationships with Ardelyx, Atmo, Biomerica, Commonwealth Diagnostics International, Corprata, Dieta, Evinature, Food Marble, Gemelli, Kiwi BioScience, Modify Health, Nestlé, Phathom, Redhill, Salix/Valeant, Takeda, and Vibrant.
A version of this article appeared on Medscape.com .
PHOENIX — Patients with or without polyp removal in an index colonoscopy commonly receive follow-up surveillance with a fecal immunochemical test (FIT), yet many of these patients do not receive a recommended colonoscopy after a positive FIT.
“In this large US study, we found interval FITs are frequently performed in patients with and without prior polypectomy,” said first author Natalie J. Wilson, MD, of the University of Minnesota in Minneapolis, while presenting the findings at the American College of Gastroenterology (ACG) 2025 Annual Scientific Meeting.
“ and colorectal cancer, regardless of polypectomy history,” Wilson said.
Guideline recommendations stress the need for follow-up surveillance with a colonoscopy, particularly in patients who have had a prior polypectomy, because of the higher risk.
Reasons patients may instead turn to FIT may include cost or other factors, she said.
To determine just how often that happens, how having a previous polypectomy affects FIT results, and how adherent patients are to follow up if a FIT result is positive, Wilson and her colleagues evaluated data from nearly 4.8 million individuals in the Veterans Health Administration Corporate Data Warehouse who underwent colonoscopy between 2000 and 2024.
Of the patients, 10.9% were found to have subsequently received interval FIT within 10 years of the index colonoscopy, and of those patients, nearly half (49.9%) had received a polypectomy at the index colonoscopy.
The average time from the colonoscopy/polypectomy to the interval FIT was 5.9 years (5.6 years in the polypectomy group vs 6.2 years in the non-polypectomy group).
Among the FIT screenings, results were positive in 17.2% of post-polypectomy patients and 14.1% of patients with no prior polypectomy, indicating a history of polypectomy to be predictive of a positive interval FIT (odds ratio [OR], 1.12; P < .0001).
Notably, while a follow-up colonoscopy is considered essential following a positive FIT result — and having a previous polypectomy should add further urgency to the matter — the study showed only 50.4% of those who had an earlier polypectomy went on to receive the recommended follow-up colonoscopy after a positive follow-up FIT, and the rate was 49.3% among those who had not received a polypectomy (P = .001).
For those who did receive a follow-up colonoscopy after a positive FIT, the duration of time to receiving the colonoscopy was longer among those who had a prior polypectomy, at 2.9 months compared with 2.5 months in the non-polypectomy group (P < .001).
Colonoscopy results following a positive FIT showed higher rates of detections among patients who had prior polypectomies than among those with no prior polypectomy, including tubular adenomas (54.7% vs 45.8%), tubulovillous adenomas (5.6% vs 4.7%), adenomas with high-grade dysplasia (0.8% vs 0.7%), sessile serrated lesions (3.52% vs 2.4%), advanced colorectal neoplasia (9.2% vs 7.9%), and colorectal cancer (3.3% vs 3.0%).
However, a prior polypectomy was not independently predictive of colorectal cancer (OR, 0.96; P = .65) or advanced colorectal neoplasia (OR, 0.97; P = .57) in the post-colonoscopy interval FIT.
The findings underscore that “positive results carried a high risk of advanced neoplasia or cancer, irrespective of prior polypectomy history,” Wilson said.
Clinicians Must ‘Do a Better Job’
Commenting on the study, William D. Chey, MD, AGAF, chief of the Division of Gastroenterology & Hepatology at the University of Michigan in Ann Arbor, noted that the study “addresses one of the biggest challenges we face as a profession, which is making sure that patients who have a positive stool test get a colonoscopy.”
He noted that the low rate of just 50% of recipients of positive FITs going on to receive a colonoscopy is consistent with what is observed in other trials.
“Other data suggests that the rate might even be significantly higher — at 70%-80%, depending upon the population and the test,” Chey told Medscape Medical News.
Reasons for the failure to receive the follow-up testing range from income restrictions (due to the high cost of a colonoscopy, especially if not covered by insurance), education, speaking a foreign language, and other factors, he said.
The relatively high rates of colon cancers detected by FIT in the study, in those with and without a prior polypectomy, along with findings from other studies “should raise questions about whether there might be a role for FIT testing in addition to colonoscopy.” However, much stronger evidence would be needed, Chey noted.
In the meantime, a key issue is “how do we do a better job of making sure that individuals who have a positive FIT test get a colonoscopy,” he said.
“I think a lot of this is going to come down to how it’s done at the primary care level.”
Chey added that in that, and any other setting, “the main message that needs to get out to people who are undergoing stool-based screening is that the stool test is only the first part of the screening process, and if it’s positive, a follow-up colonoscopy must be performed.”
“Otherwise, the stool-based test is of no value.”
Wilson had no disclosures to report. Chey’s disclosures included consulting and/or other relationships with Ardelyx, Atmo, Biomerica, Commonwealth Diagnostics International, Corprata, Dieta, Evinature, Food Marble, Gemelli, Kiwi BioScience, Modify Health, Nestlé, Phathom, Redhill, Salix/Valeant, Takeda, and Vibrant.
A version of this article appeared on Medscape.com .
FROM ACG 2025
Polypectomy Best Practices Not Routinely Followed in US
, an analysis of more than 1.8 million colonoscopies found.
“We expected to find some variations in polypectomy technique, but the results were surprising; overall, cold snare usage was much lower than expected, given that this is the recommended method for removing most small polyps,” Seth Crockett, MD, MPH, AGAF, professor of medicine, Division of Gastroenterology and Hepatology, Oregon Health & Science University, Portland, told GI & Hepatology News.
The study was published in the October issue of The American Journal of Gastroenterology.
Using Gastroenterology Quality Improvement Consortium Registry data, Crockett and colleagues analyzed more than 1.8 million colonoscopies performed by 4601 endoscopists between 2019 and 2022 across 702 sites. All colonoscopies involved removal of polyps < 1 cm; lesions of this size are commonly found in screening colonoscopies, and detection is crucial to early cancer prevention.
The researchers found striking variation in polypectomy technique. Guideline-based cold snare polypectomy (CSP) was used in only 58% of cases (and as a single device in only 51%), whereas cold forceps polypectomy (CFP) accounted for 35% and hot snare polypectomy (HSP) for 11%.
The fact that CSP was used in fewer than 60% of cases represents “an important quality gap,” the authors wrote, adding that the fact that more than 10% of colonoscopies used HSP suggests that “some patients harboring low-risk lesions may be exposed to excess risk related to these practice variations.”
And while recommendations around the use of CFP are more nuanced (based largely on forceps type and polyp size), the “high frequency of CFP also suggests nonadherence to best practices,” they noted.
Gastroenterologists More Apt to Follow Guidance
Polypectomy technique varied by polyp type. CFP was more common in cases where only hyperplastic polyps were removed compared with cases with tubular adenomas (45% vs 30%, respectively). CSP use was highest in cases where only sessile serrated lesions were removed (66%) compared with cases with only tubular adenomas (61%) or hyperplastic polyps (37%).
There was also considerable variation by provider specialty.
Gastroenterologists (compared with non-GI specialists) used HSP less (4% vs 8%) and CSP more (40% vs 34%). Colonoscopies performed with GI fellows were more likely to use CFP (31% vs 21%) and less likely to use HSP (1% vs 5%) compared with colonoscopies without fellows.
“It was somewhat reassuring that colonoscopies performed by gastroenterologists were more likely to adhere to guideline recommendations, which suggests that dedicated endoscopy training is likely an important factor driving high-quality colonoscopy,” Crockett told GI & Hepatology News.
“Unexpectedly,” polypectomy technique also differed dramatically by geographic region, he said. CFP was used more than twice as often in the Northeast (31%) as in the Midwest (14%), whereas CSP was used more frequently in the Midwest (52%) than in the Northeast (32%).
“We suspect that much of the variation is related to differences in training, preferences, habits, and evolution of colonoscopy practice over time,” Crockett said. “More research is needed on the underlying drivers of this variation, and how differences in polypectomy technique impact both the safety and efficacy of colonoscopy to prevent colorectal cancer,” he said.
“As a specialty, we need to continue to work on disseminating guideline recommendations regarding colonoscopy quality, monitoring adherence to evidence-based practices, and working to address gaps in quality where they exist,” he added.
‘Concerning, Surprising, and Disappointing’
David Johnson, MD, professor of medicine and chief of gastroenterology at Eastern Virginia Medical School and Old Dominion University in Norfolk, called the results “concerning, surprising, and disappointing” and not consistent with the most current quality recommendations that advocate cold snare for most polyps less than 1 cm in size.
“Cold snare polypectomy has been shown not only to be more effective but also takes less time to perform, relative to cold biopsy,” said Johnson, who wasn’t involved in the study.
Johnson told GI & Hepatology News, “Inadequate lesion resection and variation in resection quality are major issues for colonoscopy quality. Those who perform colonoscopies need to be up-to-date with evidence-based quality standards — as well as held accountable if [there is] discordant practice — if we are to optimize the cancer prevention benefits of quality colonoscopy.”
Limitations of the current analysis include lack of extensive patient information and inability to further stratify polyps < 1 cm by size.
The study had no commercial funding. Crockett had no disclosures. Johnson disclosed serving as a director, officer, partner, employee, advisor, consultant, or trustee for ISOThrive.
A version of this article appeared on Medscape.com.
, an analysis of more than 1.8 million colonoscopies found.
“We expected to find some variations in polypectomy technique, but the results were surprising; overall, cold snare usage was much lower than expected, given that this is the recommended method for removing most small polyps,” Seth Crockett, MD, MPH, AGAF, professor of medicine, Division of Gastroenterology and Hepatology, Oregon Health & Science University, Portland, told GI & Hepatology News.
The study was published in the October issue of The American Journal of Gastroenterology.
Using Gastroenterology Quality Improvement Consortium Registry data, Crockett and colleagues analyzed more than 1.8 million colonoscopies performed by 4601 endoscopists between 2019 and 2022 across 702 sites. All colonoscopies involved removal of polyps < 1 cm; lesions of this size are commonly found in screening colonoscopies, and detection is crucial to early cancer prevention.
The researchers found striking variation in polypectomy technique. Guideline-based cold snare polypectomy (CSP) was used in only 58% of cases (and as a single device in only 51%), whereas cold forceps polypectomy (CFP) accounted for 35% and hot snare polypectomy (HSP) for 11%.
The fact that CSP was used in fewer than 60% of cases represents “an important quality gap,” the authors wrote, adding that the fact that more than 10% of colonoscopies used HSP suggests that “some patients harboring low-risk lesions may be exposed to excess risk related to these practice variations.”
And while recommendations around the use of CFP are more nuanced (based largely on forceps type and polyp size), the “high frequency of CFP also suggests nonadherence to best practices,” they noted.
Gastroenterologists More Apt to Follow Guidance
Polypectomy technique varied by polyp type. CFP was more common in cases where only hyperplastic polyps were removed compared with cases with tubular adenomas (45% vs 30%, respectively). CSP use was highest in cases where only sessile serrated lesions were removed (66%) compared with cases with only tubular adenomas (61%) or hyperplastic polyps (37%).
There was also considerable variation by provider specialty.
Gastroenterologists (compared with non-GI specialists) used HSP less (4% vs 8%) and CSP more (40% vs 34%). Colonoscopies performed with GI fellows were more likely to use CFP (31% vs 21%) and less likely to use HSP (1% vs 5%) compared with colonoscopies without fellows.
“It was somewhat reassuring that colonoscopies performed by gastroenterologists were more likely to adhere to guideline recommendations, which suggests that dedicated endoscopy training is likely an important factor driving high-quality colonoscopy,” Crockett told GI & Hepatology News.
“Unexpectedly,” polypectomy technique also differed dramatically by geographic region, he said. CFP was used more than twice as often in the Northeast (31%) as in the Midwest (14%), whereas CSP was used more frequently in the Midwest (52%) than in the Northeast (32%).
“We suspect that much of the variation is related to differences in training, preferences, habits, and evolution of colonoscopy practice over time,” Crockett said. “More research is needed on the underlying drivers of this variation, and how differences in polypectomy technique impact both the safety and efficacy of colonoscopy to prevent colorectal cancer,” he said.
“As a specialty, we need to continue to work on disseminating guideline recommendations regarding colonoscopy quality, monitoring adherence to evidence-based practices, and working to address gaps in quality where they exist,” he added.
‘Concerning, Surprising, and Disappointing’
David Johnson, MD, professor of medicine and chief of gastroenterology at Eastern Virginia Medical School and Old Dominion University in Norfolk, called the results “concerning, surprising, and disappointing” and not consistent with the most current quality recommendations that advocate cold snare for most polyps less than 1 cm in size.
“Cold snare polypectomy has been shown not only to be more effective but also takes less time to perform, relative to cold biopsy,” said Johnson, who wasn’t involved in the study.
Johnson told GI & Hepatology News, “Inadequate lesion resection and variation in resection quality are major issues for colonoscopy quality. Those who perform colonoscopies need to be up-to-date with evidence-based quality standards — as well as held accountable if [there is] discordant practice — if we are to optimize the cancer prevention benefits of quality colonoscopy.”
Limitations of the current analysis include lack of extensive patient information and inability to further stratify polyps < 1 cm by size.
The study had no commercial funding. Crockett had no disclosures. Johnson disclosed serving as a director, officer, partner, employee, advisor, consultant, or trustee for ISOThrive.
A version of this article appeared on Medscape.com.
, an analysis of more than 1.8 million colonoscopies found.
“We expected to find some variations in polypectomy technique, but the results were surprising; overall, cold snare usage was much lower than expected, given that this is the recommended method for removing most small polyps,” Seth Crockett, MD, MPH, AGAF, professor of medicine, Division of Gastroenterology and Hepatology, Oregon Health & Science University, Portland, told GI & Hepatology News.
The study was published in the October issue of The American Journal of Gastroenterology.
Using Gastroenterology Quality Improvement Consortium Registry data, Crockett and colleagues analyzed more than 1.8 million colonoscopies performed by 4601 endoscopists between 2019 and 2022 across 702 sites. All colonoscopies involved removal of polyps < 1 cm; lesions of this size are commonly found in screening colonoscopies, and detection is crucial to early cancer prevention.
The researchers found striking variation in polypectomy technique. Guideline-based cold snare polypectomy (CSP) was used in only 58% of cases (and as a single device in only 51%), whereas cold forceps polypectomy (CFP) accounted for 35% and hot snare polypectomy (HSP) for 11%.
The fact that CSP was used in fewer than 60% of cases represents “an important quality gap,” the authors wrote, adding that the fact that more than 10% of colonoscopies used HSP suggests that “some patients harboring low-risk lesions may be exposed to excess risk related to these practice variations.”
And while recommendations around the use of CFP are more nuanced (based largely on forceps type and polyp size), the “high frequency of CFP also suggests nonadherence to best practices,” they noted.
Gastroenterologists More Apt to Follow Guidance
Polypectomy technique varied by polyp type. CFP was more common in cases where only hyperplastic polyps were removed compared with cases with tubular adenomas (45% vs 30%, respectively). CSP use was highest in cases where only sessile serrated lesions were removed (66%) compared with cases with only tubular adenomas (61%) or hyperplastic polyps (37%).
There was also considerable variation by provider specialty.
Gastroenterologists (compared with non-GI specialists) used HSP less (4% vs 8%) and CSP more (40% vs 34%). Colonoscopies performed with GI fellows were more likely to use CFP (31% vs 21%) and less likely to use HSP (1% vs 5%) compared with colonoscopies without fellows.
“It was somewhat reassuring that colonoscopies performed by gastroenterologists were more likely to adhere to guideline recommendations, which suggests that dedicated endoscopy training is likely an important factor driving high-quality colonoscopy,” Crockett told GI & Hepatology News.
“Unexpectedly,” polypectomy technique also differed dramatically by geographic region, he said. CFP was used more than twice as often in the Northeast (31%) as in the Midwest (14%), whereas CSP was used more frequently in the Midwest (52%) than in the Northeast (32%).
“We suspect that much of the variation is related to differences in training, preferences, habits, and evolution of colonoscopy practice over time,” Crockett said. “More research is needed on the underlying drivers of this variation, and how differences in polypectomy technique impact both the safety and efficacy of colonoscopy to prevent colorectal cancer,” he said.
“As a specialty, we need to continue to work on disseminating guideline recommendations regarding colonoscopy quality, monitoring adherence to evidence-based practices, and working to address gaps in quality where they exist,” he added.
‘Concerning, Surprising, and Disappointing’
David Johnson, MD, professor of medicine and chief of gastroenterology at Eastern Virginia Medical School and Old Dominion University in Norfolk, called the results “concerning, surprising, and disappointing” and not consistent with the most current quality recommendations that advocate cold snare for most polyps less than 1 cm in size.
“Cold snare polypectomy has been shown not only to be more effective but also takes less time to perform, relative to cold biopsy,” said Johnson, who wasn’t involved in the study.
Johnson told GI & Hepatology News, “Inadequate lesion resection and variation in resection quality are major issues for colonoscopy quality. Those who perform colonoscopies need to be up-to-date with evidence-based quality standards — as well as held accountable if [there is] discordant practice — if we are to optimize the cancer prevention benefits of quality colonoscopy.”
Limitations of the current analysis include lack of extensive patient information and inability to further stratify polyps < 1 cm by size.
The study had no commercial funding. Crockett had no disclosures. Johnson disclosed serving as a director, officer, partner, employee, advisor, consultant, or trustee for ISOThrive.
A version of this article appeared on Medscape.com.
Durvalumab Plus FLOT Ups Survival in Early Upper-GI Cancer
BERLIN — , according to findings presented at the 2025 annual meeting of the European Society for Medical Oncology (ESMO).
Experts said the survival benefit further supports perioperative durvalumab plus FLOT (fluorouracil, leucovorin, oxaliplatin, and docetaxel) as the new standard of care for patients with localized gastric or gastroesophageal adenocarcinoma. Earlier results from the phase 3 MATTERHORN trial, reported at the American Society of Clinical Oncology meeting (ASCO) in June, showed that the addition of durvalumab improved event-free survival compared with FLOT alone.
The findings presented at ESMO show that at 36 months, overall survival was 68.6% among patients who received durvalumab + FLOT vs 61.9% among those given FLOT plus a placebo. After a median of 43 months, the survival advantage in the durvalumab group was statistically significant (hazard ratio [HR], 0.78; 95% CI, 0.63-0.96; P = .021) and “more importantly, clinically meaningful,” said lead investigator Josep Tabernero, MD, PhD, of Vall d’Hebron University Hospital in Barcelona.
The results “strongly support the use of perioperative durvalumab plus chemotherapy with FLOT as a new global standard of care for patients with localized gastric and gastroesophageal adenocarcinoma,” Tabernero said.
Speaking as discussant for the session, Sylvie Lorenzen, MD, PhD, Technische Universität München in Munich, Germany, was enthusiastic that the previously reported trends in MATTERHORN held strong.
“The shape of the curves presented at ASCO was already very positive,” she said. “And now, with a longer follow-up, more events, and a higher overall survival maturity, they reach statistical significance. It looks like the magnitude of the effect increases with longer follow-up, and this is important for our patients.”
The trial randomly assigned 948 patients with resectable gastric or gastroesophageal adenocarcinoma to receive either durvalumab (1500 mg) or placebo every 4 weeks, plus FLOT for 2 cycles before surgery and then again after, followed by durvalumab or placebo every 4 weeks for 10 cycles.
Patients were stratified according to lymph node status, as well as PD-L1 expression (≥ 1% or < 1%, according to the Tumor Area Positivity score.)
The improvement in overall survival with durvalumab was seen regardless of PD-L1 expression, Tabernero said, with the same hazard ratios (0.79) in both the positive and negative subgroups.
However, there was no clear overall survival benefit in certain other subgroups, including women (n = 266; HR, 0.91), those with node-negative disease (n = 277; HR, 1.01), and those with diffuse histology (n = 249; HR, 0.98).
Lorenzen said that clinicians should “pay attention” to those patient subgroups, as they seem to benefit less from the addition of durvalumab. However, she cautioned that the findings were based on small patient numbers and the confidence intervals were wide.
Tabernero also reported additional data on event-free survival (EFS). Overall, the durvalumab/FLOT combination improved EFS among patients with any degree of pathological response and irrespective of lymph node status at surgery.
Regarding nodal staging, which was done in 800 patients, the percentage who achieved negative nodal status was higher in the durvalumab group (58.2%) vs the placebo group (44.8%). However, the improvement in EFS with durvalumab was comparable for node-negative (HR, 0.74) and node-positive (HR, 0.77) patients.
Lorenzen said that overall, the results provide a solid answer to the question, “Is it time to change practice?”
“I think MATTERHORN gives us the largest dataset and answers this question satisfactorily,” she said. Given that overall survival improved regardless of PD-L1 expression, she added, the combination of durvalumab and FLOT should be offered to “all our patient subgroups.”
The study was funded by AstraZeneca. Tabernero made numerous disclosures, including relationships with AstraZeneca, Boehringer Ingelheim, Chugai, and Daichii Sankyo. Lorenzen disclosed financial interests in or serving as an invited speaker for Servier, Lilly, MSD, and BMS.
A version of this article appeared on Medscape.com .
BERLIN — , according to findings presented at the 2025 annual meeting of the European Society for Medical Oncology (ESMO).
Experts said the survival benefit further supports perioperative durvalumab plus FLOT (fluorouracil, leucovorin, oxaliplatin, and docetaxel) as the new standard of care for patients with localized gastric or gastroesophageal adenocarcinoma. Earlier results from the phase 3 MATTERHORN trial, reported at the American Society of Clinical Oncology meeting (ASCO) in June, showed that the addition of durvalumab improved event-free survival compared with FLOT alone.
The findings presented at ESMO show that at 36 months, overall survival was 68.6% among patients who received durvalumab + FLOT vs 61.9% among those given FLOT plus a placebo. After a median of 43 months, the survival advantage in the durvalumab group was statistically significant (hazard ratio [HR], 0.78; 95% CI, 0.63-0.96; P = .021) and “more importantly, clinically meaningful,” said lead investigator Josep Tabernero, MD, PhD, of Vall d’Hebron University Hospital in Barcelona.
The results “strongly support the use of perioperative durvalumab plus chemotherapy with FLOT as a new global standard of care for patients with localized gastric and gastroesophageal adenocarcinoma,” Tabernero said.
Speaking as discussant for the session, Sylvie Lorenzen, MD, PhD, Technische Universität München in Munich, Germany, was enthusiastic that the previously reported trends in MATTERHORN held strong.
“The shape of the curves presented at ASCO was already very positive,” she said. “And now, with a longer follow-up, more events, and a higher overall survival maturity, they reach statistical significance. It looks like the magnitude of the effect increases with longer follow-up, and this is important for our patients.”
The trial randomly assigned 948 patients with resectable gastric or gastroesophageal adenocarcinoma to receive either durvalumab (1500 mg) or placebo every 4 weeks, plus FLOT for 2 cycles before surgery and then again after, followed by durvalumab or placebo every 4 weeks for 10 cycles.
Patients were stratified according to lymph node status, as well as PD-L1 expression (≥ 1% or < 1%, according to the Tumor Area Positivity score.)
The improvement in overall survival with durvalumab was seen regardless of PD-L1 expression, Tabernero said, with the same hazard ratios (0.79) in both the positive and negative subgroups.
However, there was no clear overall survival benefit in certain other subgroups, including women (n = 266; HR, 0.91), those with node-negative disease (n = 277; HR, 1.01), and those with diffuse histology (n = 249; HR, 0.98).
Lorenzen said that clinicians should “pay attention” to those patient subgroups, as they seem to benefit less from the addition of durvalumab. However, she cautioned that the findings were based on small patient numbers and the confidence intervals were wide.
Tabernero also reported additional data on event-free survival (EFS). Overall, the durvalumab/FLOT combination improved EFS among patients with any degree of pathological response and irrespective of lymph node status at surgery.
Regarding nodal staging, which was done in 800 patients, the percentage who achieved negative nodal status was higher in the durvalumab group (58.2%) vs the placebo group (44.8%). However, the improvement in EFS with durvalumab was comparable for node-negative (HR, 0.74) and node-positive (HR, 0.77) patients.
Lorenzen said that overall, the results provide a solid answer to the question, “Is it time to change practice?”
“I think MATTERHORN gives us the largest dataset and answers this question satisfactorily,” she said. Given that overall survival improved regardless of PD-L1 expression, she added, the combination of durvalumab and FLOT should be offered to “all our patient subgroups.”
The study was funded by AstraZeneca. Tabernero made numerous disclosures, including relationships with AstraZeneca, Boehringer Ingelheim, Chugai, and Daichii Sankyo. Lorenzen disclosed financial interests in or serving as an invited speaker for Servier, Lilly, MSD, and BMS.
A version of this article appeared on Medscape.com .
BERLIN — , according to findings presented at the 2025 annual meeting of the European Society for Medical Oncology (ESMO).
Experts said the survival benefit further supports perioperative durvalumab plus FLOT (fluorouracil, leucovorin, oxaliplatin, and docetaxel) as the new standard of care for patients with localized gastric or gastroesophageal adenocarcinoma. Earlier results from the phase 3 MATTERHORN trial, reported at the American Society of Clinical Oncology meeting (ASCO) in June, showed that the addition of durvalumab improved event-free survival compared with FLOT alone.
The findings presented at ESMO show that at 36 months, overall survival was 68.6% among patients who received durvalumab + FLOT vs 61.9% among those given FLOT plus a placebo. After a median of 43 months, the survival advantage in the durvalumab group was statistically significant (hazard ratio [HR], 0.78; 95% CI, 0.63-0.96; P = .021) and “more importantly, clinically meaningful,” said lead investigator Josep Tabernero, MD, PhD, of Vall d’Hebron University Hospital in Barcelona.
The results “strongly support the use of perioperative durvalumab plus chemotherapy with FLOT as a new global standard of care for patients with localized gastric and gastroesophageal adenocarcinoma,” Tabernero said.
Speaking as discussant for the session, Sylvie Lorenzen, MD, PhD, Technische Universität München in Munich, Germany, was enthusiastic that the previously reported trends in MATTERHORN held strong.
“The shape of the curves presented at ASCO was already very positive,” she said. “And now, with a longer follow-up, more events, and a higher overall survival maturity, they reach statistical significance. It looks like the magnitude of the effect increases with longer follow-up, and this is important for our patients.”
The trial randomly assigned 948 patients with resectable gastric or gastroesophageal adenocarcinoma to receive either durvalumab (1500 mg) or placebo every 4 weeks, plus FLOT for 2 cycles before surgery and then again after, followed by durvalumab or placebo every 4 weeks for 10 cycles.
Patients were stratified according to lymph node status, as well as PD-L1 expression (≥ 1% or < 1%, according to the Tumor Area Positivity score.)
The improvement in overall survival with durvalumab was seen regardless of PD-L1 expression, Tabernero said, with the same hazard ratios (0.79) in both the positive and negative subgroups.
However, there was no clear overall survival benefit in certain other subgroups, including women (n = 266; HR, 0.91), those with node-negative disease (n = 277; HR, 1.01), and those with diffuse histology (n = 249; HR, 0.98).
Lorenzen said that clinicians should “pay attention” to those patient subgroups, as they seem to benefit less from the addition of durvalumab. However, she cautioned that the findings were based on small patient numbers and the confidence intervals were wide.
Tabernero also reported additional data on event-free survival (EFS). Overall, the durvalumab/FLOT combination improved EFS among patients with any degree of pathological response and irrespective of lymph node status at surgery.
Regarding nodal staging, which was done in 800 patients, the percentage who achieved negative nodal status was higher in the durvalumab group (58.2%) vs the placebo group (44.8%). However, the improvement in EFS with durvalumab was comparable for node-negative (HR, 0.74) and node-positive (HR, 0.77) patients.
Lorenzen said that overall, the results provide a solid answer to the question, “Is it time to change practice?”
“I think MATTERHORN gives us the largest dataset and answers this question satisfactorily,” she said. Given that overall survival improved regardless of PD-L1 expression, she added, the combination of durvalumab and FLOT should be offered to “all our patient subgroups.”
The study was funded by AstraZeneca. Tabernero made numerous disclosures, including relationships with AstraZeneca, Boehringer Ingelheim, Chugai, and Daichii Sankyo. Lorenzen disclosed financial interests in or serving as an invited speaker for Servier, Lilly, MSD, and BMS.
A version of this article appeared on Medscape.com .
Half of Patients Skip Repeat Stool Tests for CRC Screening
A large real-world study found that
Among those who did repeat the test, the average delay was 3 months before COVID and increased to 5 months during the pandemic, the authors reported in BMJ Public Health.
“Stool tests are relatively easy to complete at home and mailed for testing, and they are inexpensive, but they must be completed annually. In contrast, colonoscopies are more invasive and require more time away from work but only need to be repeated every 5-10 years,” Staci J Wendt, PhD, director, health research accelerator, Providence Research Network, Providence, Rhode Island, told GI & Hepatology News.
In the end, “the best colorectal cancer screening test is the one that gets done,” Wendt said.
“This is why we stress the importance of patients and their doctor having these discussions together and deciding which screening is the most preferred method for the individual patient,” she added.
Stool Tests Gaining Traction
Adults are increasingly turning to at-home stool tests for CRC screening — a trend that accelerated during the pandemic. Yet, there is limited data on whether patients undergo repeat stool tests following initial negative test results.
Wendt and her colleagues documented rates of repeat preventative stool tests by analyzing electronic medical records from Providence St Joseph Health, a large health system with 51 hospitals and over 1000 clinics across seven western US states.
They divided their analysis into two periods based on the onset of the pandemic. The pre-COVID onset period spanned January 2018 to February 2020 and the post-COVID period spanned March 2020 to February 2022.
“The pandemic is a salient time to conduct this study because it resulted in a dramatic decrease in colonoscopies, which were partially replaced by stool tests. This partial replacement of colonoscopies by stool tests has led other studies to conclude that stool tests mitigated gaps in CRC screening during the pandemic. But gaps may persist if patients do not undergo repeat testing,” the study team explained.
Their sample included 403,085 patients. Among those with an initial negative stool test, the share who obtained a timely repeat screening ranged from 38% to 49% across the study years, confirming that “most patients do not undergo the recommended repeat screening after their initial stool test,” the researchers said.
Among adults who do a repeat test, delays were common. The average lag to the follow-up test was 3months on average, increasing to about 5 months amid COVID — almost half as long as the preventative screening period of stool tests (12 months).
“These gaps could delay detection of CRC and subsequent treatment, potentially resulting in higher mortality. These gaps are particularly important as more and more patients use stool tests instead of colonoscopes for CRC screening,” the researchers wrote.
Screening patterns shifted markedly during the pandemic.
Not surprisingly, the volume of colonoscopies declined substantially after the onset of the pandemic and stayed low through the study’s end. In contrast, the volume of at-home stool tests was increasing before the pandemic and accelerated during the pandemic.
“Given this increase in stool tests, it will be increasingly important to focus on improving long-term adherence to screening through outreach, policies and programs,” the researchers said.
A Multilevel Approach
Wendt said health systems that are incorporating proactive measures like sending stool kits to patients who are eligible for screening, should ensure that these screening kits and information are sent annually and that it is stressed that the screening must happen every year.
Reached for comment, Aasma Shaukat, MD, MPH, AGAF, director of outcomes research, Division of Gastroenterology and Hepatology, NYU Langone Health, New York City, who wasn’t involved in the study, said the poor adherence to repeat stool tests for CRC screening seen in this study is “not surprising.”
“We know that adherence goes down with each consecutive screening round and what is really needed is an organized program to keep the level of adherence up,” Shaukat told GI & Hepatology News.
Shaukat agreed that boosting adherence to stool tests requires a “multilevel approach.”
She cited the success of the CRC screening program implemented across Kaiser Permanente Northern California. The program includes proactive and targeted outreach to members who are overdue for screening and mailed fecal immunochemical test kits for at-home use.
As reported previously by GI & Hepatology News, the program has made a huge difference in CRC incidence, deaths, and racial disparities.
The program has doubled the proportion of people up to date with screening. And, within about 10 years, cancer rates were cut by a third, deaths were halved and largely eliminated long-standing differences by race and ethnicity.
The study had no commercial funding. Wendt and Shaukat declared having no relevant disclosures.
A version of this article appeared on Medscape.com.
A large real-world study found that
Among those who did repeat the test, the average delay was 3 months before COVID and increased to 5 months during the pandemic, the authors reported in BMJ Public Health.
“Stool tests are relatively easy to complete at home and mailed for testing, and they are inexpensive, but they must be completed annually. In contrast, colonoscopies are more invasive and require more time away from work but only need to be repeated every 5-10 years,” Staci J Wendt, PhD, director, health research accelerator, Providence Research Network, Providence, Rhode Island, told GI & Hepatology News.
In the end, “the best colorectal cancer screening test is the one that gets done,” Wendt said.
“This is why we stress the importance of patients and their doctor having these discussions together and deciding which screening is the most preferred method for the individual patient,” she added.
Stool Tests Gaining Traction
Adults are increasingly turning to at-home stool tests for CRC screening — a trend that accelerated during the pandemic. Yet, there is limited data on whether patients undergo repeat stool tests following initial negative test results.
Wendt and her colleagues documented rates of repeat preventative stool tests by analyzing electronic medical records from Providence St Joseph Health, a large health system with 51 hospitals and over 1000 clinics across seven western US states.
They divided their analysis into two periods based on the onset of the pandemic. The pre-COVID onset period spanned January 2018 to February 2020 and the post-COVID period spanned March 2020 to February 2022.
“The pandemic is a salient time to conduct this study because it resulted in a dramatic decrease in colonoscopies, which were partially replaced by stool tests. This partial replacement of colonoscopies by stool tests has led other studies to conclude that stool tests mitigated gaps in CRC screening during the pandemic. But gaps may persist if patients do not undergo repeat testing,” the study team explained.
Their sample included 403,085 patients. Among those with an initial negative stool test, the share who obtained a timely repeat screening ranged from 38% to 49% across the study years, confirming that “most patients do not undergo the recommended repeat screening after their initial stool test,” the researchers said.
Among adults who do a repeat test, delays were common. The average lag to the follow-up test was 3months on average, increasing to about 5 months amid COVID — almost half as long as the preventative screening period of stool tests (12 months).
“These gaps could delay detection of CRC and subsequent treatment, potentially resulting in higher mortality. These gaps are particularly important as more and more patients use stool tests instead of colonoscopes for CRC screening,” the researchers wrote.
Screening patterns shifted markedly during the pandemic.
Not surprisingly, the volume of colonoscopies declined substantially after the onset of the pandemic and stayed low through the study’s end. In contrast, the volume of at-home stool tests was increasing before the pandemic and accelerated during the pandemic.
“Given this increase in stool tests, it will be increasingly important to focus on improving long-term adherence to screening through outreach, policies and programs,” the researchers said.
A Multilevel Approach
Wendt said health systems that are incorporating proactive measures like sending stool kits to patients who are eligible for screening, should ensure that these screening kits and information are sent annually and that it is stressed that the screening must happen every year.
Reached for comment, Aasma Shaukat, MD, MPH, AGAF, director of outcomes research, Division of Gastroenterology and Hepatology, NYU Langone Health, New York City, who wasn’t involved in the study, said the poor adherence to repeat stool tests for CRC screening seen in this study is “not surprising.”
“We know that adherence goes down with each consecutive screening round and what is really needed is an organized program to keep the level of adherence up,” Shaukat told GI & Hepatology News.
Shaukat agreed that boosting adherence to stool tests requires a “multilevel approach.”
She cited the success of the CRC screening program implemented across Kaiser Permanente Northern California. The program includes proactive and targeted outreach to members who are overdue for screening and mailed fecal immunochemical test kits for at-home use.
As reported previously by GI & Hepatology News, the program has made a huge difference in CRC incidence, deaths, and racial disparities.
The program has doubled the proportion of people up to date with screening. And, within about 10 years, cancer rates were cut by a third, deaths were halved and largely eliminated long-standing differences by race and ethnicity.
The study had no commercial funding. Wendt and Shaukat declared having no relevant disclosures.
A version of this article appeared on Medscape.com.
A large real-world study found that
Among those who did repeat the test, the average delay was 3 months before COVID and increased to 5 months during the pandemic, the authors reported in BMJ Public Health.
“Stool tests are relatively easy to complete at home and mailed for testing, and they are inexpensive, but they must be completed annually. In contrast, colonoscopies are more invasive and require more time away from work but only need to be repeated every 5-10 years,” Staci J Wendt, PhD, director, health research accelerator, Providence Research Network, Providence, Rhode Island, told GI & Hepatology News.
In the end, “the best colorectal cancer screening test is the one that gets done,” Wendt said.
“This is why we stress the importance of patients and their doctor having these discussions together and deciding which screening is the most preferred method for the individual patient,” she added.
Stool Tests Gaining Traction
Adults are increasingly turning to at-home stool tests for CRC screening — a trend that accelerated during the pandemic. Yet, there is limited data on whether patients undergo repeat stool tests following initial negative test results.
Wendt and her colleagues documented rates of repeat preventative stool tests by analyzing electronic medical records from Providence St Joseph Health, a large health system with 51 hospitals and over 1000 clinics across seven western US states.
They divided their analysis into two periods based on the onset of the pandemic. The pre-COVID onset period spanned January 2018 to February 2020 and the post-COVID period spanned March 2020 to February 2022.
“The pandemic is a salient time to conduct this study because it resulted in a dramatic decrease in colonoscopies, which were partially replaced by stool tests. This partial replacement of colonoscopies by stool tests has led other studies to conclude that stool tests mitigated gaps in CRC screening during the pandemic. But gaps may persist if patients do not undergo repeat testing,” the study team explained.
Their sample included 403,085 patients. Among those with an initial negative stool test, the share who obtained a timely repeat screening ranged from 38% to 49% across the study years, confirming that “most patients do not undergo the recommended repeat screening after their initial stool test,” the researchers said.
Among adults who do a repeat test, delays were common. The average lag to the follow-up test was 3months on average, increasing to about 5 months amid COVID — almost half as long as the preventative screening period of stool tests (12 months).
“These gaps could delay detection of CRC and subsequent treatment, potentially resulting in higher mortality. These gaps are particularly important as more and more patients use stool tests instead of colonoscopes for CRC screening,” the researchers wrote.
Screening patterns shifted markedly during the pandemic.
Not surprisingly, the volume of colonoscopies declined substantially after the onset of the pandemic and stayed low through the study’s end. In contrast, the volume of at-home stool tests was increasing before the pandemic and accelerated during the pandemic.
“Given this increase in stool tests, it will be increasingly important to focus on improving long-term adherence to screening through outreach, policies and programs,” the researchers said.
A Multilevel Approach
Wendt said health systems that are incorporating proactive measures like sending stool kits to patients who are eligible for screening, should ensure that these screening kits and information are sent annually and that it is stressed that the screening must happen every year.
Reached for comment, Aasma Shaukat, MD, MPH, AGAF, director of outcomes research, Division of Gastroenterology and Hepatology, NYU Langone Health, New York City, who wasn’t involved in the study, said the poor adherence to repeat stool tests for CRC screening seen in this study is “not surprising.”
“We know that adherence goes down with each consecutive screening round and what is really needed is an organized program to keep the level of adherence up,” Shaukat told GI & Hepatology News.
Shaukat agreed that boosting adherence to stool tests requires a “multilevel approach.”
She cited the success of the CRC screening program implemented across Kaiser Permanente Northern California. The program includes proactive and targeted outreach to members who are overdue for screening and mailed fecal immunochemical test kits for at-home use.
As reported previously by GI & Hepatology News, the program has made a huge difference in CRC incidence, deaths, and racial disparities.
The program has doubled the proportion of people up to date with screening. And, within about 10 years, cancer rates were cut by a third, deaths were halved and largely eliminated long-standing differences by race and ethnicity.
The study had no commercial funding. Wendt and Shaukat declared having no relevant disclosures.
A version of this article appeared on Medscape.com.
Menopausal Hormone Therapy Lowers Upper GI Cancer Risk
BERLIN — , according to a large population-based study across five Nordic countries. The association appeared strongest for combined estrogen-progestin and systemic formulations.
“This is one of the largest and most comprehensive studies to date supporting the hypothesis of an inverse association between MHT and risk of esophago-gastric cancer,” said Victoria Wocalewski, MD, from the Department of Molecular Medicine and Surgery, Karolinska Institutet, Stockholm, who presented the findings at United European Gastroenterology (UEG) Week 2025.
There was a decreased risk for all investigated cancers in MHT users, but the strongest association was observed for esophageal adenocarcinoma (EAC), said Wocalewski. In addition, “there were discrete dose-dependent results for [EAC] and gastric adenocarcinoma (GAC) but not for esophageal squamous cell carcinoma (ESCC).”
Large Population-Based Study
Previous research has suggested that hormonal changes could partly explain the male predominance in esophageal and gastric cancers, but evidence from large, well-controlled datasets has been limited.
“Cancer rates in women increase significantly after age of 60, so it has been hypothesized that this pattern is linked to declined levels of estrogen that comes with menopause,” said Wocalewski, explaining the rationale for the study.
“Some studies looking at MHT use have indicated a possible protective effect, but with some contradictory results and type-specific variations,” Wocalewski noted. “Our study aimed to investigate these previous findings using a larger study sample.”
The population-based case-control study drew on prospectively collected data from the NordGETS database including national prescription, cancer, and population registries in Denmark, Finland, Iceland, Norway, and Sweden spanning 1994-2020. In total, 19,518 women with esophago-gastric cancer were compared with 195,094 controls randomly selected from the general population, and matched for age, calendar year, and country (in a 1:10 ratio). Women were 45 years or over with a diagnosis of EAC, ESCC, or GAC.
In total there were 5000 cases of EAC, 4401 of ESCC, and 10,117 of GAC, with the median ages being 74, 72, and 75 years, respectively; most cases of EAC and ESCC were found in Denmark, and most cases of GAC were in Sweden.
The investigators categorized participants by defined daily doses (DDDs) of MHT into three equal sized categories: low (< 158 DDDs), intermediate (158-848 DDDs), and high (> 848 DDDs). MHT was defined as systemic or local, and estrogen only or combined with progesterone. Odds ratios (ORs) were calculated for three major cancer outcomes of EAC, ESCC, and GAC, adjusted for known confounders such as age, obesity, smoking, alcohol consumption, reflux disease, Helicobacter pylori eradication, and concomitant use of statins or non-steroidal anti-inflammatory drugs (NSAIDs). However, Wocalewski noted that they did not adjust for socio-economic factors.
Significant Reductions Across Esophago-Gastric Cancers
Compared with nonusers, women with any MHT exposure had a markedly reduced risk of EAC with adjusted ORs (aORs) of 0.74 (95% CI, 0.67-0.81) for low-use, 0.68 (95% CI, 0.61-0.75) for intermediate-use, and 0.68 (95% CI, 0.61-0.75) for high-use groups. Various adjustments were made for obesity, reflux, statins, and NSAIDs, as well as smoking, alcohol use, and H pylori eradication.
Similar inverse associations were seen for ESCC with aORs of 0.69 (95% CI, 0.62-0.77), 0.70 (95% CI, 0.62-0.77), and 0.71 (95% CI, 0.64-0.79) across the dose categories, and for GAC where risk decreased progressively from 0.90 (95% CI, 0.84-0.96) to 0.80 (95% CI, 0.74-0.86) across increasing MHT doses.
When stratified by hormone formulation, combined estrogen-progesterone therapy and systemic MHT conferred the strongest risk reduction. For example, systemic MHT use was associated with aORs of 0.67 (95% CI, 0.61-0.74) for EAC and 0.82 (95% CI, 0.76-0.88) for GAC, while local (vaginal) preparations showed slightly weaker associations at 0.72 (95% CI, 0.66-0.78) and 0.87 (95% CI, 0.83-0.92), respectively.
In EAC, combined estrogen-progesterone therapy led to an OR of 0.68 (95% CI, 0.63-0.73) and 0.77 (95% CI, 0.69-0.87) for women on estrogen alone. Similar results were found for ESCC. For GAC, combination resulted in an aOR of 0.85 (95% CI, 0.80-0.89) and 0.88 (95% CI, 0.81-0.97) in estrogen only therapy respectively.
“Our results reinforce the concept that estrogenic signaling may influence tumor development in the upper GI tract,” said Wocalewski. “Understanding these mechanisms could help identify at-risk populations and inform prevention strategies,” she added, noting that, “hormonal effects on epithelial tight junctions and nitric oxide synthesis in the gastrointestinal tract” would have an influence on smooth muscle cells.
Link Between Hormones and GI Pathology
Commenting on the study for GI & Hepatology News, Jan Bornschein, MD, University of Oxford, UK, who was not involved in the research, said the results are “highly relevant.”
“We’ve seen for a long time a link between hormones and GI pathology, however, it has been poorly investigated and the whole mechanisms are not understood, so it’s welcome that this group is moving forward and investigating this in a structured way,” he said.
Another delegate cautioned that MHT was associated with a risk for other non- gastrointestinal cancers. “I think it’s extremely important, because there are data on associations [of MHT] with breast cancer and also endometrial cancer. It’s good to see that it may help and reduce this cancer, but we have to be really careful about the others.”
Wocalewski reports no relevant conflicts of interest. Bornschein has no disclosures relevant to this study. The study was funded by Karolinska Institutet and supported by national cancer and prescription registry data from Denmark, Finland, Iceland, Norway, and Sweden.
A version of this article appeared on Medscape.com.
BERLIN — , according to a large population-based study across five Nordic countries. The association appeared strongest for combined estrogen-progestin and systemic formulations.
“This is one of the largest and most comprehensive studies to date supporting the hypothesis of an inverse association between MHT and risk of esophago-gastric cancer,” said Victoria Wocalewski, MD, from the Department of Molecular Medicine and Surgery, Karolinska Institutet, Stockholm, who presented the findings at United European Gastroenterology (UEG) Week 2025.
There was a decreased risk for all investigated cancers in MHT users, but the strongest association was observed for esophageal adenocarcinoma (EAC), said Wocalewski. In addition, “there were discrete dose-dependent results for [EAC] and gastric adenocarcinoma (GAC) but not for esophageal squamous cell carcinoma (ESCC).”
Large Population-Based Study
Previous research has suggested that hormonal changes could partly explain the male predominance in esophageal and gastric cancers, but evidence from large, well-controlled datasets has been limited.
“Cancer rates in women increase significantly after age of 60, so it has been hypothesized that this pattern is linked to declined levels of estrogen that comes with menopause,” said Wocalewski, explaining the rationale for the study.
“Some studies looking at MHT use have indicated a possible protective effect, but with some contradictory results and type-specific variations,” Wocalewski noted. “Our study aimed to investigate these previous findings using a larger study sample.”
The population-based case-control study drew on prospectively collected data from the NordGETS database including national prescription, cancer, and population registries in Denmark, Finland, Iceland, Norway, and Sweden spanning 1994-2020. In total, 19,518 women with esophago-gastric cancer were compared with 195,094 controls randomly selected from the general population, and matched for age, calendar year, and country (in a 1:10 ratio). Women were 45 years or over with a diagnosis of EAC, ESCC, or GAC.
In total there were 5000 cases of EAC, 4401 of ESCC, and 10,117 of GAC, with the median ages being 74, 72, and 75 years, respectively; most cases of EAC and ESCC were found in Denmark, and most cases of GAC were in Sweden.
The investigators categorized participants by defined daily doses (DDDs) of MHT into three equal sized categories: low (< 158 DDDs), intermediate (158-848 DDDs), and high (> 848 DDDs). MHT was defined as systemic or local, and estrogen only or combined with progesterone. Odds ratios (ORs) were calculated for three major cancer outcomes of EAC, ESCC, and GAC, adjusted for known confounders such as age, obesity, smoking, alcohol consumption, reflux disease, Helicobacter pylori eradication, and concomitant use of statins or non-steroidal anti-inflammatory drugs (NSAIDs). However, Wocalewski noted that they did not adjust for socio-economic factors.
Significant Reductions Across Esophago-Gastric Cancers
Compared with nonusers, women with any MHT exposure had a markedly reduced risk of EAC with adjusted ORs (aORs) of 0.74 (95% CI, 0.67-0.81) for low-use, 0.68 (95% CI, 0.61-0.75) for intermediate-use, and 0.68 (95% CI, 0.61-0.75) for high-use groups. Various adjustments were made for obesity, reflux, statins, and NSAIDs, as well as smoking, alcohol use, and H pylori eradication.
Similar inverse associations were seen for ESCC with aORs of 0.69 (95% CI, 0.62-0.77), 0.70 (95% CI, 0.62-0.77), and 0.71 (95% CI, 0.64-0.79) across the dose categories, and for GAC where risk decreased progressively from 0.90 (95% CI, 0.84-0.96) to 0.80 (95% CI, 0.74-0.86) across increasing MHT doses.
When stratified by hormone formulation, combined estrogen-progesterone therapy and systemic MHT conferred the strongest risk reduction. For example, systemic MHT use was associated with aORs of 0.67 (95% CI, 0.61-0.74) for EAC and 0.82 (95% CI, 0.76-0.88) for GAC, while local (vaginal) preparations showed slightly weaker associations at 0.72 (95% CI, 0.66-0.78) and 0.87 (95% CI, 0.83-0.92), respectively.
In EAC, combined estrogen-progesterone therapy led to an OR of 0.68 (95% CI, 0.63-0.73) and 0.77 (95% CI, 0.69-0.87) for women on estrogen alone. Similar results were found for ESCC. For GAC, combination resulted in an aOR of 0.85 (95% CI, 0.80-0.89) and 0.88 (95% CI, 0.81-0.97) in estrogen only therapy respectively.
“Our results reinforce the concept that estrogenic signaling may influence tumor development in the upper GI tract,” said Wocalewski. “Understanding these mechanisms could help identify at-risk populations and inform prevention strategies,” she added, noting that, “hormonal effects on epithelial tight junctions and nitric oxide synthesis in the gastrointestinal tract” would have an influence on smooth muscle cells.
Link Between Hormones and GI Pathology
Commenting on the study for GI & Hepatology News, Jan Bornschein, MD, University of Oxford, UK, who was not involved in the research, said the results are “highly relevant.”
“We’ve seen for a long time a link between hormones and GI pathology, however, it has been poorly investigated and the whole mechanisms are not understood, so it’s welcome that this group is moving forward and investigating this in a structured way,” he said.
Another delegate cautioned that MHT was associated with a risk for other non- gastrointestinal cancers. “I think it’s extremely important, because there are data on associations [of MHT] with breast cancer and also endometrial cancer. It’s good to see that it may help and reduce this cancer, but we have to be really careful about the others.”
Wocalewski reports no relevant conflicts of interest. Bornschein has no disclosures relevant to this study. The study was funded by Karolinska Institutet and supported by national cancer and prescription registry data from Denmark, Finland, Iceland, Norway, and Sweden.
A version of this article appeared on Medscape.com.
BERLIN — , according to a large population-based study across five Nordic countries. The association appeared strongest for combined estrogen-progestin and systemic formulations.
“This is one of the largest and most comprehensive studies to date supporting the hypothesis of an inverse association between MHT and risk of esophago-gastric cancer,” said Victoria Wocalewski, MD, from the Department of Molecular Medicine and Surgery, Karolinska Institutet, Stockholm, who presented the findings at United European Gastroenterology (UEG) Week 2025.
There was a decreased risk for all investigated cancers in MHT users, but the strongest association was observed for esophageal adenocarcinoma (EAC), said Wocalewski. In addition, “there were discrete dose-dependent results for [EAC] and gastric adenocarcinoma (GAC) but not for esophageal squamous cell carcinoma (ESCC).”
Large Population-Based Study
Previous research has suggested that hormonal changes could partly explain the male predominance in esophageal and gastric cancers, but evidence from large, well-controlled datasets has been limited.
“Cancer rates in women increase significantly after age of 60, so it has been hypothesized that this pattern is linked to declined levels of estrogen that comes with menopause,” said Wocalewski, explaining the rationale for the study.
“Some studies looking at MHT use have indicated a possible protective effect, but with some contradictory results and type-specific variations,” Wocalewski noted. “Our study aimed to investigate these previous findings using a larger study sample.”
The population-based case-control study drew on prospectively collected data from the NordGETS database including national prescription, cancer, and population registries in Denmark, Finland, Iceland, Norway, and Sweden spanning 1994-2020. In total, 19,518 women with esophago-gastric cancer were compared with 195,094 controls randomly selected from the general population, and matched for age, calendar year, and country (in a 1:10 ratio). Women were 45 years or over with a diagnosis of EAC, ESCC, or GAC.
In total there were 5000 cases of EAC, 4401 of ESCC, and 10,117 of GAC, with the median ages being 74, 72, and 75 years, respectively; most cases of EAC and ESCC were found in Denmark, and most cases of GAC were in Sweden.
The investigators categorized participants by defined daily doses (DDDs) of MHT into three equal sized categories: low (< 158 DDDs), intermediate (158-848 DDDs), and high (> 848 DDDs). MHT was defined as systemic or local, and estrogen only or combined with progesterone. Odds ratios (ORs) were calculated for three major cancer outcomes of EAC, ESCC, and GAC, adjusted for known confounders such as age, obesity, smoking, alcohol consumption, reflux disease, Helicobacter pylori eradication, and concomitant use of statins or non-steroidal anti-inflammatory drugs (NSAIDs). However, Wocalewski noted that they did not adjust for socio-economic factors.
Significant Reductions Across Esophago-Gastric Cancers
Compared with nonusers, women with any MHT exposure had a markedly reduced risk of EAC with adjusted ORs (aORs) of 0.74 (95% CI, 0.67-0.81) for low-use, 0.68 (95% CI, 0.61-0.75) for intermediate-use, and 0.68 (95% CI, 0.61-0.75) for high-use groups. Various adjustments were made for obesity, reflux, statins, and NSAIDs, as well as smoking, alcohol use, and H pylori eradication.
Similar inverse associations were seen for ESCC with aORs of 0.69 (95% CI, 0.62-0.77), 0.70 (95% CI, 0.62-0.77), and 0.71 (95% CI, 0.64-0.79) across the dose categories, and for GAC where risk decreased progressively from 0.90 (95% CI, 0.84-0.96) to 0.80 (95% CI, 0.74-0.86) across increasing MHT doses.
When stratified by hormone formulation, combined estrogen-progesterone therapy and systemic MHT conferred the strongest risk reduction. For example, systemic MHT use was associated with aORs of 0.67 (95% CI, 0.61-0.74) for EAC and 0.82 (95% CI, 0.76-0.88) for GAC, while local (vaginal) preparations showed slightly weaker associations at 0.72 (95% CI, 0.66-0.78) and 0.87 (95% CI, 0.83-0.92), respectively.
In EAC, combined estrogen-progesterone therapy led to an OR of 0.68 (95% CI, 0.63-0.73) and 0.77 (95% CI, 0.69-0.87) for women on estrogen alone. Similar results were found for ESCC. For GAC, combination resulted in an aOR of 0.85 (95% CI, 0.80-0.89) and 0.88 (95% CI, 0.81-0.97) in estrogen only therapy respectively.
“Our results reinforce the concept that estrogenic signaling may influence tumor development in the upper GI tract,” said Wocalewski. “Understanding these mechanisms could help identify at-risk populations and inform prevention strategies,” she added, noting that, “hormonal effects on epithelial tight junctions and nitric oxide synthesis in the gastrointestinal tract” would have an influence on smooth muscle cells.
Link Between Hormones and GI Pathology
Commenting on the study for GI & Hepatology News, Jan Bornschein, MD, University of Oxford, UK, who was not involved in the research, said the results are “highly relevant.”
“We’ve seen for a long time a link between hormones and GI pathology, however, it has been poorly investigated and the whole mechanisms are not understood, so it’s welcome that this group is moving forward and investigating this in a structured way,” he said.
Another delegate cautioned that MHT was associated with a risk for other non- gastrointestinal cancers. “I think it’s extremely important, because there are data on associations [of MHT] with breast cancer and also endometrial cancer. It’s good to see that it may help and reduce this cancer, but we have to be really careful about the others.”
Wocalewski reports no relevant conflicts of interest. Bornschein has no disclosures relevant to this study. The study was funded by Karolinska Institutet and supported by national cancer and prescription registry data from Denmark, Finland, Iceland, Norway, and Sweden.
A version of this article appeared on Medscape.com.
Combining Upper-Lower GI Screening Feasible, Effective
BERLIN — , including malignancies and lesions requiring ongoing surveillance, according to an interim analysis from the TOGAS study.
“There was an abundance of benign but clinically relevant findings,” said lead investigator Jan Bornschein, MD, gastroenterologist at Oxford University Hospitals NHS Foundation Trust, Oxford, England, who presented the interim resuts of the study at United European Gastroenterology (UEG) Week 2025.
While the study found upper GI neoplasia in only 1.4% of participants, 17.8% of individuals were marked for upper GI endoscopic surveillance.
The results may inform how Europe develops gastric cancer prevention programs alongside those for colorectal cancer, said Bornschein. “If we can combine the upper GI endoscopy with other modalities [colonoscopy], the more likelihood there is that you can have a one-stop test package,” he said. “A combination, particularly for bowel and stomach, is more feasible and also more cost-effective. So far, the findings show that it’s definitely a strategy that, in my opinion, is worth implementing.”
Bornschein and the TOGAS study group hope that the combined approach will prove workable across diverse European settings and will help identify a spectrum of upper GI pathology, from cancers and dysplasia to atrophy and intestinal metaplasia, that can meaningfully affect follow-up surveillance.
Mixed Rates of GI Cancers Across Europe and the US
These findings come amid data showing rising rates of early-onset (younger than 50 years) GI cancers in the US, including colorectal, gastric, pancreatic, and esophageal tumors. These trends, previously reported by this news organization, point to environmental and lifestyle drivers, strengthening the case for earlier detection and risk-tailored strategies for upper GI neoplasia and preneoplastic conditions detected during existing colorectal cancer screening pathways.
However, Bornschein noted that prevalence varies considerably across Europe. “There are areas, particularly in the Eastern regions, and in some parts of the West, for example, Portugal, that have a very high incidence of GI cancers. In the UK or in Germany, we have noticed a decline over the years, so the numbers are actually much better than they used to be.”
The study is the second in a series of three TOGAS pilot studies and was conducted across eight centers (France, Germany, Ireland, Latvia, Lithuania, the Netherlands, Portugal, and Spain) in adults aged 50-74 years attending screening or polyp-surveillance colonoscopy.
A European Society of Gastrointestinal Endoscopy-aligned protocol defining image documentation, biopsy sampling, and quality parameters was followed to ensure a standardized approach. “Marked preneoplastic change” was defined as gastric glandular atrophy or intestinal metaplasia at the Operative Link on Gastritis Assessment/Operative Link on Gastric Intestinal Metaplasia Assessment stage III-IV and/or Endoscopic Grading of Gastric Intestinal Metaplasia > 5, triggering a need for endoscopic surveillance.
Data were gathered on colonoscopy findings (including polyp surveillance and family history), EGD findings plus biopsies, serum pepsinogen, and Helicobacter pylori serology. Outcome measures included the prevalence of gastric cancer and preneoplastic conditions, the diagnostic accuracy of pepsinogen testing, comparisons between national settings, the relevance of upper endoscopy in fecal immunochemical test-positive cases, and overall H pylori prevalence.
Neoplasia and Preneoplasia Found
A total of 846 participants were analyzed. At baseline, the mean age was 62 years, 52.2% were men, and 84.2% were White, despite efforts to recruit a more diverse population. Around 390 participants drank alcohol, and 190 smoked tobacco.
A total of 37.8% of participants had undergone prior EGD, of which 94.7% were performed more than 3 years before the study start. The history of GI surgery was 13.7%, and the history of cancer was 14.5%. Around 11% took aspirin, and 14% took proton pump inhibitors (PPIs). “We were surprised at the low prevalence of PPI use,” remarked Bornschein. “It was also good news that around half were never smokers.”
Key results for upper GI neoplasia included six patients (0.7%) with gastric cancers, three (0.4%) with esophageal cancers, and five (0.6%) with duodenal tumors. H pylori positivity was found in 303 patients (35.8%), with an additional 81 (9.6%) reporting a history of eradication.
Colorectal findings included 15 patients (1.8%) with cancers and colon polyps in 503 (59.5%) participants.
Regarding preneoplastic conditions, endoscopy identified intestinal metaplasia in 174 patients (20.6%), of which 65 (7.7%) were multifocal. Atrophy was observed in 220 patients (26.0%), with 59 (7.0%) showing multifocal atrophic changes. Both intestinal metaplasia and atrophy were found together in 105 (12.4%) patients. Barrett’s esophagus was detected in 31 (3.7%) patients.
“I’d really like to highlight these further benign gastric findings,” said Bornschein. These included gastric ulcers in 28 (3.3%) patients, erosive gastritis in 245 (29.0%) patients, esophageal ulcers in three (0.4%) patients, Los Angeles Community College District classification esophagitis in 13 (1.5%) patients, and duodenal ulcers in 10 (1.2%) patients. “These were asymptomatic, but we were able to identify them,” he noted.
“We’ve had a very low rate of complications (0.01%),” he added.” I don’t want to jinx that now. These were basically related to sedation.”
PROSPERO: Early Detection of Upper GI Conditions in a UK Population
Massimiliano di Pietro, MD, consultant gastroenterologist at Addenbrooke’s Hospital, Cambridge, England, and the principal investigator of the PROSPERO study, which aimed to determine the prevalence of premalignant upper GI conditions in routine endoscopy in the UK, commented on the findings. The TOGAS study focuses on asymptomatic individuals referred for colonoscopy and examines the value of performing an upper GI endoscopy at the same time, he explained. “This approach might identify upper GI conditions that require monitoring, in particular early cancer.”
“On the other hand, the PROSPERO study focuses on patients referred for upper GI symptoms and diagnosis,” he said. Preliminary data from that study, presented during the same session as the TOGAS trial, showed a 13.6% prevalence of premalignant upper GI conditions in a symptomatic UK patient population referred for endoscopy.
“In some respects, the findings were similar, particularly the rate of upper GI cancer at 1.4%, although there were differences in the prevalence of premalignant conditions,” he noted. “This may be explained by the fact that TOGAS is a European study, while PROSPERO is UK-based, where the distribution of upper GI cancers differs, with more esophageal adenocarcinoma vs gastric adenocarcinoma.”
Reflecting on both of the studies, Di Pietro said they are “really important in fulfilling an unmet need in the quality of upper GI endoscopy. Currently, there are no diagnostic quality indicators in upper GI endoscopy, so it’s difficult to rate the performance of endoscopists in the same way as we can in lower GI. It’s really important to understand the population prevalence, both in symptomatic and asymptomatic individuals, of premalignant and malignant upper GI conditions.”
TOGAS 2 is recruiting until February 2026, with 1200 of a potential 1600 participants recruited to date. The data will be used for implementation modeling and to inform quality indicators for future screening programs. Final results and plans for a follow-up study are expected in 2026.
Bornschein declared receiving advisory and speaker fees from Flynn Pharma and Juvisé Pharmaceuticals. Di Pietro reported having no disclosures relevant to the studies discussed.
A version of this article first appeared on Medscape.com.
BERLIN — , including malignancies and lesions requiring ongoing surveillance, according to an interim analysis from the TOGAS study.
“There was an abundance of benign but clinically relevant findings,” said lead investigator Jan Bornschein, MD, gastroenterologist at Oxford University Hospitals NHS Foundation Trust, Oxford, England, who presented the interim resuts of the study at United European Gastroenterology (UEG) Week 2025.
While the study found upper GI neoplasia in only 1.4% of participants, 17.8% of individuals were marked for upper GI endoscopic surveillance.
The results may inform how Europe develops gastric cancer prevention programs alongside those for colorectal cancer, said Bornschein. “If we can combine the upper GI endoscopy with other modalities [colonoscopy], the more likelihood there is that you can have a one-stop test package,” he said. “A combination, particularly for bowel and stomach, is more feasible and also more cost-effective. So far, the findings show that it’s definitely a strategy that, in my opinion, is worth implementing.”
Bornschein and the TOGAS study group hope that the combined approach will prove workable across diverse European settings and will help identify a spectrum of upper GI pathology, from cancers and dysplasia to atrophy and intestinal metaplasia, that can meaningfully affect follow-up surveillance.
Mixed Rates of GI Cancers Across Europe and the US
These findings come amid data showing rising rates of early-onset (younger than 50 years) GI cancers in the US, including colorectal, gastric, pancreatic, and esophageal tumors. These trends, previously reported by this news organization, point to environmental and lifestyle drivers, strengthening the case for earlier detection and risk-tailored strategies for upper GI neoplasia and preneoplastic conditions detected during existing colorectal cancer screening pathways.
However, Bornschein noted that prevalence varies considerably across Europe. “There are areas, particularly in the Eastern regions, and in some parts of the West, for example, Portugal, that have a very high incidence of GI cancers. In the UK or in Germany, we have noticed a decline over the years, so the numbers are actually much better than they used to be.”
The study is the second in a series of three TOGAS pilot studies and was conducted across eight centers (France, Germany, Ireland, Latvia, Lithuania, the Netherlands, Portugal, and Spain) in adults aged 50-74 years attending screening or polyp-surveillance colonoscopy.
A European Society of Gastrointestinal Endoscopy-aligned protocol defining image documentation, biopsy sampling, and quality parameters was followed to ensure a standardized approach. “Marked preneoplastic change” was defined as gastric glandular atrophy or intestinal metaplasia at the Operative Link on Gastritis Assessment/Operative Link on Gastric Intestinal Metaplasia Assessment stage III-IV and/or Endoscopic Grading of Gastric Intestinal Metaplasia > 5, triggering a need for endoscopic surveillance.
Data were gathered on colonoscopy findings (including polyp surveillance and family history), EGD findings plus biopsies, serum pepsinogen, and Helicobacter pylori serology. Outcome measures included the prevalence of gastric cancer and preneoplastic conditions, the diagnostic accuracy of pepsinogen testing, comparisons between national settings, the relevance of upper endoscopy in fecal immunochemical test-positive cases, and overall H pylori prevalence.
Neoplasia and Preneoplasia Found
A total of 846 participants were analyzed. At baseline, the mean age was 62 years, 52.2% were men, and 84.2% were White, despite efforts to recruit a more diverse population. Around 390 participants drank alcohol, and 190 smoked tobacco.
A total of 37.8% of participants had undergone prior EGD, of which 94.7% were performed more than 3 years before the study start. The history of GI surgery was 13.7%, and the history of cancer was 14.5%. Around 11% took aspirin, and 14% took proton pump inhibitors (PPIs). “We were surprised at the low prevalence of PPI use,” remarked Bornschein. “It was also good news that around half were never smokers.”
Key results for upper GI neoplasia included six patients (0.7%) with gastric cancers, three (0.4%) with esophageal cancers, and five (0.6%) with duodenal tumors. H pylori positivity was found in 303 patients (35.8%), with an additional 81 (9.6%) reporting a history of eradication.
Colorectal findings included 15 patients (1.8%) with cancers and colon polyps in 503 (59.5%) participants.
Regarding preneoplastic conditions, endoscopy identified intestinal metaplasia in 174 patients (20.6%), of which 65 (7.7%) were multifocal. Atrophy was observed in 220 patients (26.0%), with 59 (7.0%) showing multifocal atrophic changes. Both intestinal metaplasia and atrophy were found together in 105 (12.4%) patients. Barrett’s esophagus was detected in 31 (3.7%) patients.
“I’d really like to highlight these further benign gastric findings,” said Bornschein. These included gastric ulcers in 28 (3.3%) patients, erosive gastritis in 245 (29.0%) patients, esophageal ulcers in three (0.4%) patients, Los Angeles Community College District classification esophagitis in 13 (1.5%) patients, and duodenal ulcers in 10 (1.2%) patients. “These were asymptomatic, but we were able to identify them,” he noted.
“We’ve had a very low rate of complications (0.01%),” he added.” I don’t want to jinx that now. These were basically related to sedation.”
PROSPERO: Early Detection of Upper GI Conditions in a UK Population
Massimiliano di Pietro, MD, consultant gastroenterologist at Addenbrooke’s Hospital, Cambridge, England, and the principal investigator of the PROSPERO study, which aimed to determine the prevalence of premalignant upper GI conditions in routine endoscopy in the UK, commented on the findings. The TOGAS study focuses on asymptomatic individuals referred for colonoscopy and examines the value of performing an upper GI endoscopy at the same time, he explained. “This approach might identify upper GI conditions that require monitoring, in particular early cancer.”
“On the other hand, the PROSPERO study focuses on patients referred for upper GI symptoms and diagnosis,” he said. Preliminary data from that study, presented during the same session as the TOGAS trial, showed a 13.6% prevalence of premalignant upper GI conditions in a symptomatic UK patient population referred for endoscopy.
“In some respects, the findings were similar, particularly the rate of upper GI cancer at 1.4%, although there were differences in the prevalence of premalignant conditions,” he noted. “This may be explained by the fact that TOGAS is a European study, while PROSPERO is UK-based, where the distribution of upper GI cancers differs, with more esophageal adenocarcinoma vs gastric adenocarcinoma.”
Reflecting on both of the studies, Di Pietro said they are “really important in fulfilling an unmet need in the quality of upper GI endoscopy. Currently, there are no diagnostic quality indicators in upper GI endoscopy, so it’s difficult to rate the performance of endoscopists in the same way as we can in lower GI. It’s really important to understand the population prevalence, both in symptomatic and asymptomatic individuals, of premalignant and malignant upper GI conditions.”
TOGAS 2 is recruiting until February 2026, with 1200 of a potential 1600 participants recruited to date. The data will be used for implementation modeling and to inform quality indicators for future screening programs. Final results and plans for a follow-up study are expected in 2026.
Bornschein declared receiving advisory and speaker fees from Flynn Pharma and Juvisé Pharmaceuticals. Di Pietro reported having no disclosures relevant to the studies discussed.
A version of this article first appeared on Medscape.com.
BERLIN — , including malignancies and lesions requiring ongoing surveillance, according to an interim analysis from the TOGAS study.
“There was an abundance of benign but clinically relevant findings,” said lead investigator Jan Bornschein, MD, gastroenterologist at Oxford University Hospitals NHS Foundation Trust, Oxford, England, who presented the interim resuts of the study at United European Gastroenterology (UEG) Week 2025.
While the study found upper GI neoplasia in only 1.4% of participants, 17.8% of individuals were marked for upper GI endoscopic surveillance.
The results may inform how Europe develops gastric cancer prevention programs alongside those for colorectal cancer, said Bornschein. “If we can combine the upper GI endoscopy with other modalities [colonoscopy], the more likelihood there is that you can have a one-stop test package,” he said. “A combination, particularly for bowel and stomach, is more feasible and also more cost-effective. So far, the findings show that it’s definitely a strategy that, in my opinion, is worth implementing.”
Bornschein and the TOGAS study group hope that the combined approach will prove workable across diverse European settings and will help identify a spectrum of upper GI pathology, from cancers and dysplasia to atrophy and intestinal metaplasia, that can meaningfully affect follow-up surveillance.
Mixed Rates of GI Cancers Across Europe and the US
These findings come amid data showing rising rates of early-onset (younger than 50 years) GI cancers in the US, including colorectal, gastric, pancreatic, and esophageal tumors. These trends, previously reported by this news organization, point to environmental and lifestyle drivers, strengthening the case for earlier detection and risk-tailored strategies for upper GI neoplasia and preneoplastic conditions detected during existing colorectal cancer screening pathways.
However, Bornschein noted that prevalence varies considerably across Europe. “There are areas, particularly in the Eastern regions, and in some parts of the West, for example, Portugal, that have a very high incidence of GI cancers. In the UK or in Germany, we have noticed a decline over the years, so the numbers are actually much better than they used to be.”
The study is the second in a series of three TOGAS pilot studies and was conducted across eight centers (France, Germany, Ireland, Latvia, Lithuania, the Netherlands, Portugal, and Spain) in adults aged 50-74 years attending screening or polyp-surveillance colonoscopy.
A European Society of Gastrointestinal Endoscopy-aligned protocol defining image documentation, biopsy sampling, and quality parameters was followed to ensure a standardized approach. “Marked preneoplastic change” was defined as gastric glandular atrophy or intestinal metaplasia at the Operative Link on Gastritis Assessment/Operative Link on Gastric Intestinal Metaplasia Assessment stage III-IV and/or Endoscopic Grading of Gastric Intestinal Metaplasia > 5, triggering a need for endoscopic surveillance.
Data were gathered on colonoscopy findings (including polyp surveillance and family history), EGD findings plus biopsies, serum pepsinogen, and Helicobacter pylori serology. Outcome measures included the prevalence of gastric cancer and preneoplastic conditions, the diagnostic accuracy of pepsinogen testing, comparisons between national settings, the relevance of upper endoscopy in fecal immunochemical test-positive cases, and overall H pylori prevalence.
Neoplasia and Preneoplasia Found
A total of 846 participants were analyzed. At baseline, the mean age was 62 years, 52.2% were men, and 84.2% were White, despite efforts to recruit a more diverse population. Around 390 participants drank alcohol, and 190 smoked tobacco.
A total of 37.8% of participants had undergone prior EGD, of which 94.7% were performed more than 3 years before the study start. The history of GI surgery was 13.7%, and the history of cancer was 14.5%. Around 11% took aspirin, and 14% took proton pump inhibitors (PPIs). “We were surprised at the low prevalence of PPI use,” remarked Bornschein. “It was also good news that around half were never smokers.”
Key results for upper GI neoplasia included six patients (0.7%) with gastric cancers, three (0.4%) with esophageal cancers, and five (0.6%) with duodenal tumors. H pylori positivity was found in 303 patients (35.8%), with an additional 81 (9.6%) reporting a history of eradication.
Colorectal findings included 15 patients (1.8%) with cancers and colon polyps in 503 (59.5%) participants.
Regarding preneoplastic conditions, endoscopy identified intestinal metaplasia in 174 patients (20.6%), of which 65 (7.7%) were multifocal. Atrophy was observed in 220 patients (26.0%), with 59 (7.0%) showing multifocal atrophic changes. Both intestinal metaplasia and atrophy were found together in 105 (12.4%) patients. Barrett’s esophagus was detected in 31 (3.7%) patients.
“I’d really like to highlight these further benign gastric findings,” said Bornschein. These included gastric ulcers in 28 (3.3%) patients, erosive gastritis in 245 (29.0%) patients, esophageal ulcers in three (0.4%) patients, Los Angeles Community College District classification esophagitis in 13 (1.5%) patients, and duodenal ulcers in 10 (1.2%) patients. “These were asymptomatic, but we were able to identify them,” he noted.
“We’ve had a very low rate of complications (0.01%),” he added.” I don’t want to jinx that now. These were basically related to sedation.”
PROSPERO: Early Detection of Upper GI Conditions in a UK Population
Massimiliano di Pietro, MD, consultant gastroenterologist at Addenbrooke’s Hospital, Cambridge, England, and the principal investigator of the PROSPERO study, which aimed to determine the prevalence of premalignant upper GI conditions in routine endoscopy in the UK, commented on the findings. The TOGAS study focuses on asymptomatic individuals referred for colonoscopy and examines the value of performing an upper GI endoscopy at the same time, he explained. “This approach might identify upper GI conditions that require monitoring, in particular early cancer.”
“On the other hand, the PROSPERO study focuses on patients referred for upper GI symptoms and diagnosis,” he said. Preliminary data from that study, presented during the same session as the TOGAS trial, showed a 13.6% prevalence of premalignant upper GI conditions in a symptomatic UK patient population referred for endoscopy.
“In some respects, the findings were similar, particularly the rate of upper GI cancer at 1.4%, although there were differences in the prevalence of premalignant conditions,” he noted. “This may be explained by the fact that TOGAS is a European study, while PROSPERO is UK-based, where the distribution of upper GI cancers differs, with more esophageal adenocarcinoma vs gastric adenocarcinoma.”
Reflecting on both of the studies, Di Pietro said they are “really important in fulfilling an unmet need in the quality of upper GI endoscopy. Currently, there are no diagnostic quality indicators in upper GI endoscopy, so it’s difficult to rate the performance of endoscopists in the same way as we can in lower GI. It’s really important to understand the population prevalence, both in symptomatic and asymptomatic individuals, of premalignant and malignant upper GI conditions.”
TOGAS 2 is recruiting until February 2026, with 1200 of a potential 1600 participants recruited to date. The data will be used for implementation modeling and to inform quality indicators for future screening programs. Final results and plans for a follow-up study are expected in 2026.
Bornschein declared receiving advisory and speaker fees from Flynn Pharma and Juvisé Pharmaceuticals. Di Pietro reported having no disclosures relevant to the studies discussed.
A version of this article first appeared on Medscape.com.
Prevention and Risk-Based Surveillance Key to Curbing HCC
BERLIN — according to a joint statement from United European Gastroenterology (UEG) and the German Society for Gastroenterology, Digestive and Metabolic Diseases (DGVS).
The statement calls on EU and national policymakers to embed a twofold approach into healthcare systems that combines surveillance and prevention, rather than relying on voluntary participation. It also encourages stronger prevention measures, such as improved food labeling and restrictions on marketing unhealthy foods to children. The statement — which was also endorsed by the European Association for the Study of the Liver (EASL) — was presented at UEG Week 2025 .
“Curing HCC in early stages rather than treating the disease in a palliative setting should be the goal for all liver doctors and carers, and this is certainly the goal for patients,” said Thomas Seufferlein, MD, professor of gastroenterology at Ulm University, Germany, and one of the members of the DGVS who initiated the statement.
“We have to take HCC screening seriously which means setting up a structured, nationwide, well-documented, and evaluated program for HCC screening in Germany,” he said in an interview.
HCC is mainly curable in the early stages by local ablation, resection, or liver transplantation, “so early diagnosis is of the utmost importance for improving survival,” added Patrick Michl, MD, gastroenterologist, University of Heidelberg, Germany, DGVS member and co-initiator of the statement.
Risk-Stratified HCC Surveillance
In the face of rising rates worldwide, the UEG/DGVS call on policymakers to recognize liver cancer as a preventable and growing public health priority and to implement structured surveillance programs guided by risk thresholds. In particular, they support the recent policy statement from EASL recommending risk-based screening.
EASL’s key recommendations include:
- Targeted surveillance for individuals with an annual HCC risk exceeding 1.5%, where it is both clinically beneficial and cost-effective
- Risk scoring tools such as the age-male-albumin-bilirubin-platelets score that incorporates age, sex, platelet count, albumin, and bilirubin, to stratify patients by HCC risk, including those without established cirrhosis
- Enhanced surveillance for very high-risk groups, where MRI-based surveillance may be warranted despite higher costs, given its superior sensitivity for early-stage disease
- A de-escalation in low-risk individuals
- Patients with an annual HCC risk < 0.5% may be safely spared surveillance, avoiding unnecessary interventions
Evidence from France, Italy, and the UK showed that structured surveillance in high-risk groups is both clinically beneficial and cost-effective. National models in France have demonstrated higher curative treatment rates and fewer costly late-stage cases with structured surveillance. In the UK, health technology assessments indicate targeted surveillance is an efficient use of National Health Services resources, particularly when uptake is optimized. Italian models show that earlier diagnosis in well-defined high-risk groups can offset downstream treatment costs.
Seufferlein noted that Germany needs a “structured program to be implemented and there is currently little public awareness regarding this surveillance strategy.” However, he added there is a structured hepatitis B vaccination program in Germany, which has been successful. “Studies show that the inclusion of hep B vaccination in infancy and childhood has led to good uptake among young age groups.”
Germany, however, has yet to conduct national studies. “Prospective data on HCC surveillance benefits in Germany are lacking,” said Michl, “but multi-country models incorporating Germany’s cost structures suggest similar benefits would accrue if there were greater adherence to guideline-based recommendations and if publicly funded screening programs were implemented.”
Current recommendations in Germany for surveillance are based on evidence-based guidelines of the DGVS with stronger (‘should’) or weaker (‘may’) evidence-based recommendations. For example, patients with chronic hepatitis B virus infection should be offered regular surveillance once their platelet age gender–hepatitis B risk score is ≥ 10. In patients with advanced fibrosis because of chronic hepatitis C virus infection, regular surveillance should also be offered.
Barriers to Screening Uptake
HCC remains one of the most lethal cancers in Europe, largely because it is often diagnosed too late. Underdiagnosis of chronic liver disease, limited access to imaging, and reimbursement gaps prevent timely intervention.
Maria Buti, MD, consultant hepatologist, Hospital Vall d’Hebron, Barcelona, Spain, who was not involved in drafting the statement, remarked that “Patients with liver cirrhosis, or with advanced fibrosis, and also some high-risk noncirrhotic patients such as those with hepatitis B, clearly benefit from surveillance. Surveillance can change life expectancy and also reduce morbidity.”
However, structural barriers continue to impede uptake. “It is not always easy to identify patients with liver cirrhosis because the majority are completely asymptomatic in the early stages,” she said.
Even when risk factors are identified, adherence to 6-monthly surveillance remains patchy. “Sometimes physicians forget to request ultrasounds, or patients don’t understand the importance of it because they feel well,” Buti told GI & Hepatology News.
Expanded Training and Public Health Measures
The joint statement also advocates for expanded physician training in nutrition and hepatology, equitable access to diagnostic tools including MRI, and EU-wide nutrition labeling systems such as Nutri-Score.
The authors also called for strengthened public health measures to tackle obesity, alcohol misuse, and hepatitis transmission, and fiscal and regulatory measures such as taxation of obesogenic foods, and reducing the cost burden of healthier foods.
“If we decrease the percentage of people with liver cirrhosis through prevention, fewer people will need surveillance,” Buti stated.
Seufferlein, Michl, and Buti all declared no relevant disclosures. All three experts are members of the UEG Public Affairs Group.
A version of this article appeared on Medscape.com.
BERLIN — according to a joint statement from United European Gastroenterology (UEG) and the German Society for Gastroenterology, Digestive and Metabolic Diseases (DGVS).
The statement calls on EU and national policymakers to embed a twofold approach into healthcare systems that combines surveillance and prevention, rather than relying on voluntary participation. It also encourages stronger prevention measures, such as improved food labeling and restrictions on marketing unhealthy foods to children. The statement — which was also endorsed by the European Association for the Study of the Liver (EASL) — was presented at UEG Week 2025 .
“Curing HCC in early stages rather than treating the disease in a palliative setting should be the goal for all liver doctors and carers, and this is certainly the goal for patients,” said Thomas Seufferlein, MD, professor of gastroenterology at Ulm University, Germany, and one of the members of the DGVS who initiated the statement.
“We have to take HCC screening seriously which means setting up a structured, nationwide, well-documented, and evaluated program for HCC screening in Germany,” he said in an interview.
HCC is mainly curable in the early stages by local ablation, resection, or liver transplantation, “so early diagnosis is of the utmost importance for improving survival,” added Patrick Michl, MD, gastroenterologist, University of Heidelberg, Germany, DGVS member and co-initiator of the statement.
Risk-Stratified HCC Surveillance
In the face of rising rates worldwide, the UEG/DGVS call on policymakers to recognize liver cancer as a preventable and growing public health priority and to implement structured surveillance programs guided by risk thresholds. In particular, they support the recent policy statement from EASL recommending risk-based screening.
EASL’s key recommendations include:
- Targeted surveillance for individuals with an annual HCC risk exceeding 1.5%, where it is both clinically beneficial and cost-effective
- Risk scoring tools such as the age-male-albumin-bilirubin-platelets score that incorporates age, sex, platelet count, albumin, and bilirubin, to stratify patients by HCC risk, including those without established cirrhosis
- Enhanced surveillance for very high-risk groups, where MRI-based surveillance may be warranted despite higher costs, given its superior sensitivity for early-stage disease
- A de-escalation in low-risk individuals
- Patients with an annual HCC risk < 0.5% may be safely spared surveillance, avoiding unnecessary interventions
Evidence from France, Italy, and the UK showed that structured surveillance in high-risk groups is both clinically beneficial and cost-effective. National models in France have demonstrated higher curative treatment rates and fewer costly late-stage cases with structured surveillance. In the UK, health technology assessments indicate targeted surveillance is an efficient use of National Health Services resources, particularly when uptake is optimized. Italian models show that earlier diagnosis in well-defined high-risk groups can offset downstream treatment costs.
Seufferlein noted that Germany needs a “structured program to be implemented and there is currently little public awareness regarding this surveillance strategy.” However, he added there is a structured hepatitis B vaccination program in Germany, which has been successful. “Studies show that the inclusion of hep B vaccination in infancy and childhood has led to good uptake among young age groups.”
Germany, however, has yet to conduct national studies. “Prospective data on HCC surveillance benefits in Germany are lacking,” said Michl, “but multi-country models incorporating Germany’s cost structures suggest similar benefits would accrue if there were greater adherence to guideline-based recommendations and if publicly funded screening programs were implemented.”
Current recommendations in Germany for surveillance are based on evidence-based guidelines of the DGVS with stronger (‘should’) or weaker (‘may’) evidence-based recommendations. For example, patients with chronic hepatitis B virus infection should be offered regular surveillance once their platelet age gender–hepatitis B risk score is ≥ 10. In patients with advanced fibrosis because of chronic hepatitis C virus infection, regular surveillance should also be offered.
Barriers to Screening Uptake
HCC remains one of the most lethal cancers in Europe, largely because it is often diagnosed too late. Underdiagnosis of chronic liver disease, limited access to imaging, and reimbursement gaps prevent timely intervention.
Maria Buti, MD, consultant hepatologist, Hospital Vall d’Hebron, Barcelona, Spain, who was not involved in drafting the statement, remarked that “Patients with liver cirrhosis, or with advanced fibrosis, and also some high-risk noncirrhotic patients such as those with hepatitis B, clearly benefit from surveillance. Surveillance can change life expectancy and also reduce morbidity.”
However, structural barriers continue to impede uptake. “It is not always easy to identify patients with liver cirrhosis because the majority are completely asymptomatic in the early stages,” she said.
Even when risk factors are identified, adherence to 6-monthly surveillance remains patchy. “Sometimes physicians forget to request ultrasounds, or patients don’t understand the importance of it because they feel well,” Buti told GI & Hepatology News.
Expanded Training and Public Health Measures
The joint statement also advocates for expanded physician training in nutrition and hepatology, equitable access to diagnostic tools including MRI, and EU-wide nutrition labeling systems such as Nutri-Score.
The authors also called for strengthened public health measures to tackle obesity, alcohol misuse, and hepatitis transmission, and fiscal and regulatory measures such as taxation of obesogenic foods, and reducing the cost burden of healthier foods.
“If we decrease the percentage of people with liver cirrhosis through prevention, fewer people will need surveillance,” Buti stated.
Seufferlein, Michl, and Buti all declared no relevant disclosures. All three experts are members of the UEG Public Affairs Group.
A version of this article appeared on Medscape.com.
BERLIN — according to a joint statement from United European Gastroenterology (UEG) and the German Society for Gastroenterology, Digestive and Metabolic Diseases (DGVS).
The statement calls on EU and national policymakers to embed a twofold approach into healthcare systems that combines surveillance and prevention, rather than relying on voluntary participation. It also encourages stronger prevention measures, such as improved food labeling and restrictions on marketing unhealthy foods to children. The statement — which was also endorsed by the European Association for the Study of the Liver (EASL) — was presented at UEG Week 2025 .
“Curing HCC in early stages rather than treating the disease in a palliative setting should be the goal for all liver doctors and carers, and this is certainly the goal for patients,” said Thomas Seufferlein, MD, professor of gastroenterology at Ulm University, Germany, and one of the members of the DGVS who initiated the statement.
“We have to take HCC screening seriously which means setting up a structured, nationwide, well-documented, and evaluated program for HCC screening in Germany,” he said in an interview.
HCC is mainly curable in the early stages by local ablation, resection, or liver transplantation, “so early diagnosis is of the utmost importance for improving survival,” added Patrick Michl, MD, gastroenterologist, University of Heidelberg, Germany, DGVS member and co-initiator of the statement.
Risk-Stratified HCC Surveillance
In the face of rising rates worldwide, the UEG/DGVS call on policymakers to recognize liver cancer as a preventable and growing public health priority and to implement structured surveillance programs guided by risk thresholds. In particular, they support the recent policy statement from EASL recommending risk-based screening.
EASL’s key recommendations include:
- Targeted surveillance for individuals with an annual HCC risk exceeding 1.5%, where it is both clinically beneficial and cost-effective
- Risk scoring tools such as the age-male-albumin-bilirubin-platelets score that incorporates age, sex, platelet count, albumin, and bilirubin, to stratify patients by HCC risk, including those without established cirrhosis
- Enhanced surveillance for very high-risk groups, where MRI-based surveillance may be warranted despite higher costs, given its superior sensitivity for early-stage disease
- A de-escalation in low-risk individuals
- Patients with an annual HCC risk < 0.5% may be safely spared surveillance, avoiding unnecessary interventions
Evidence from France, Italy, and the UK showed that structured surveillance in high-risk groups is both clinically beneficial and cost-effective. National models in France have demonstrated higher curative treatment rates and fewer costly late-stage cases with structured surveillance. In the UK, health technology assessments indicate targeted surveillance is an efficient use of National Health Services resources, particularly when uptake is optimized. Italian models show that earlier diagnosis in well-defined high-risk groups can offset downstream treatment costs.
Seufferlein noted that Germany needs a “structured program to be implemented and there is currently little public awareness regarding this surveillance strategy.” However, he added there is a structured hepatitis B vaccination program in Germany, which has been successful. “Studies show that the inclusion of hep B vaccination in infancy and childhood has led to good uptake among young age groups.”
Germany, however, has yet to conduct national studies. “Prospective data on HCC surveillance benefits in Germany are lacking,” said Michl, “but multi-country models incorporating Germany’s cost structures suggest similar benefits would accrue if there were greater adherence to guideline-based recommendations and if publicly funded screening programs were implemented.”
Current recommendations in Germany for surveillance are based on evidence-based guidelines of the DGVS with stronger (‘should’) or weaker (‘may’) evidence-based recommendations. For example, patients with chronic hepatitis B virus infection should be offered regular surveillance once their platelet age gender–hepatitis B risk score is ≥ 10. In patients with advanced fibrosis because of chronic hepatitis C virus infection, regular surveillance should also be offered.
Barriers to Screening Uptake
HCC remains one of the most lethal cancers in Europe, largely because it is often diagnosed too late. Underdiagnosis of chronic liver disease, limited access to imaging, and reimbursement gaps prevent timely intervention.
Maria Buti, MD, consultant hepatologist, Hospital Vall d’Hebron, Barcelona, Spain, who was not involved in drafting the statement, remarked that “Patients with liver cirrhosis, or with advanced fibrosis, and also some high-risk noncirrhotic patients such as those with hepatitis B, clearly benefit from surveillance. Surveillance can change life expectancy and also reduce morbidity.”
However, structural barriers continue to impede uptake. “It is not always easy to identify patients with liver cirrhosis because the majority are completely asymptomatic in the early stages,” she said.
Even when risk factors are identified, adherence to 6-monthly surveillance remains patchy. “Sometimes physicians forget to request ultrasounds, or patients don’t understand the importance of it because they feel well,” Buti told GI & Hepatology News.
Expanded Training and Public Health Measures
The joint statement also advocates for expanded physician training in nutrition and hepatology, equitable access to diagnostic tools including MRI, and EU-wide nutrition labeling systems such as Nutri-Score.
The authors also called for strengthened public health measures to tackle obesity, alcohol misuse, and hepatitis transmission, and fiscal and regulatory measures such as taxation of obesogenic foods, and reducing the cost burden of healthier foods.
“If we decrease the percentage of people with liver cirrhosis through prevention, fewer people will need surveillance,” Buti stated.
Seufferlein, Michl, and Buti all declared no relevant disclosures. All three experts are members of the UEG Public Affairs Group.
A version of this article appeared on Medscape.com.
FROM UEG WEEK 2025
Oral Microbes Tied to Pancreatic Cancer Risk
Could oral microbiome profiling help spot people at risk for pancreatic cancer?
It may be possible, according to a recent analysis published in JAMA Oncology.
Researchers found that , which could be a step toward earlier detection of the deadly malignancy.
“We identified 27 individual bacterial and fungal species significantly associated with pancreatic cancer development,” said Jiyoung Ahn, PhD, of NYU Grossman School of Medicine in New York City.
“If validated, oral microbiome profiling could serve as a noninvasive biomarker to identify individuals at elevated risk who might benefit from enhanced surveillance,” Ahn told GI & Hepatology News by email.
Rates of pancreatic cancer are on the rise. But detecting the disease before it becomes unresectable has remained an elusive goal, and the US Preventive Services Task Force discourages screening of asymptomatic adults.
For their study, Ahn and her colleagues analyzed data from 122,000 participants who provided oral wash samples as part of two cohort studies conducted in the US. The researchers used whole-genome shotgun sequencing and internal transcribed spacer sequencing to identity the bacterial and fungal species in the samples, respectively.
Over a median follow-up of nearly 9 years, 445 people developed pancreatic cancer and were matched with 445 who did not. Three oral bacterial periodontal pathogens — Porphyromonas gingivalis (odds ratio [OR], 1.27), Eubacterium nodatum (OR, 1.42), and Parvimonas micra (OR, 1.36) — as well as the fungal genus Candida were all linked to significantly increased odds of developing pancreatic cancer.
In a bacteriome-wide scan, the researchers pinpointed another 20 oral bacteria associated with pancreatic cancer — eight with a decreased risk and 13 with an increased risk for the disease.
The researchers also calculated a microbial risk score, which was the weighted sum of the relative abundance of bacterial and fungal species. In a meta-analysis of data from the two cohorts, the microbial risk score derived from 23 bacterial species and four fungal species, including various Candida species, was associated with pancreatic cancer (multivariate OR per 1-SD increase in the score, 3.44; 95% CI, 2.63-4.51).
“The oral microbiota holds promise as a biomarker to identify individuals at high risk of pancreatic cancer, potentially enabling personalized pancreatic cancer prevention,” Ahn and her colleagues concluded.
But Gil Welch, MD, of Brigham and Women’s Hospital in Boston, who has written about screening for decades, isn’t so sure.
Given the “impressive volume of information” included in the analysis, “it is not surprising that the investigators are able to create a microbial risk score (based on 27 species of bacteria and fungi) that is highly related to pancreatic cancer,” Welch said. “The authors are careful to emphasize these are associations, not causal relationships.”
But even if the relationship were causal, finding more people with the malignancy can also have downsides, said Welch.
In a study out last year, Welch and colleagues found that while the incidence of pancreatic cancer among young Americans has been rising, mortality rates in this demographic haven’t budged, suggesting a potential for overdiagnosis.
“Screening for pancreatic cancer has never been shown to reduce pancreatic cancer mortality,” Welch told GI & Hepatology News. “Why screen large swaths of the population simply to enumerate ‘risk factors’ for an unproven benefit that, at best, could help only a few? Meanwhile, the burdens for everyone else are real: the mental and financial strains of ‘high risk’ labels, false alarms, and endless follow-ups. It’s a recipe to make us all worried sick — and poorer.”
Ahn reported having no disclosures. Welch reported receiving royalties from three books including “Should I be tested for cancer?”
A version of this article appeared on Medscape.com.
Could oral microbiome profiling help spot people at risk for pancreatic cancer?
It may be possible, according to a recent analysis published in JAMA Oncology.
Researchers found that , which could be a step toward earlier detection of the deadly malignancy.
“We identified 27 individual bacterial and fungal species significantly associated with pancreatic cancer development,” said Jiyoung Ahn, PhD, of NYU Grossman School of Medicine in New York City.
“If validated, oral microbiome profiling could serve as a noninvasive biomarker to identify individuals at elevated risk who might benefit from enhanced surveillance,” Ahn told GI & Hepatology News by email.
Rates of pancreatic cancer are on the rise. But detecting the disease before it becomes unresectable has remained an elusive goal, and the US Preventive Services Task Force discourages screening of asymptomatic adults.
For their study, Ahn and her colleagues analyzed data from 122,000 participants who provided oral wash samples as part of two cohort studies conducted in the US. The researchers used whole-genome shotgun sequencing and internal transcribed spacer sequencing to identity the bacterial and fungal species in the samples, respectively.
Over a median follow-up of nearly 9 years, 445 people developed pancreatic cancer and were matched with 445 who did not. Three oral bacterial periodontal pathogens — Porphyromonas gingivalis (odds ratio [OR], 1.27), Eubacterium nodatum (OR, 1.42), and Parvimonas micra (OR, 1.36) — as well as the fungal genus Candida were all linked to significantly increased odds of developing pancreatic cancer.
In a bacteriome-wide scan, the researchers pinpointed another 20 oral bacteria associated with pancreatic cancer — eight with a decreased risk and 13 with an increased risk for the disease.
The researchers also calculated a microbial risk score, which was the weighted sum of the relative abundance of bacterial and fungal species. In a meta-analysis of data from the two cohorts, the microbial risk score derived from 23 bacterial species and four fungal species, including various Candida species, was associated with pancreatic cancer (multivariate OR per 1-SD increase in the score, 3.44; 95% CI, 2.63-4.51).
“The oral microbiota holds promise as a biomarker to identify individuals at high risk of pancreatic cancer, potentially enabling personalized pancreatic cancer prevention,” Ahn and her colleagues concluded.
But Gil Welch, MD, of Brigham and Women’s Hospital in Boston, who has written about screening for decades, isn’t so sure.
Given the “impressive volume of information” included in the analysis, “it is not surprising that the investigators are able to create a microbial risk score (based on 27 species of bacteria and fungi) that is highly related to pancreatic cancer,” Welch said. “The authors are careful to emphasize these are associations, not causal relationships.”
But even if the relationship were causal, finding more people with the malignancy can also have downsides, said Welch.
In a study out last year, Welch and colleagues found that while the incidence of pancreatic cancer among young Americans has been rising, mortality rates in this demographic haven’t budged, suggesting a potential for overdiagnosis.
“Screening for pancreatic cancer has never been shown to reduce pancreatic cancer mortality,” Welch told GI & Hepatology News. “Why screen large swaths of the population simply to enumerate ‘risk factors’ for an unproven benefit that, at best, could help only a few? Meanwhile, the burdens for everyone else are real: the mental and financial strains of ‘high risk’ labels, false alarms, and endless follow-ups. It’s a recipe to make us all worried sick — and poorer.”
Ahn reported having no disclosures. Welch reported receiving royalties from three books including “Should I be tested for cancer?”
A version of this article appeared on Medscape.com.
Could oral microbiome profiling help spot people at risk for pancreatic cancer?
It may be possible, according to a recent analysis published in JAMA Oncology.
Researchers found that , which could be a step toward earlier detection of the deadly malignancy.
“We identified 27 individual bacterial and fungal species significantly associated with pancreatic cancer development,” said Jiyoung Ahn, PhD, of NYU Grossman School of Medicine in New York City.
“If validated, oral microbiome profiling could serve as a noninvasive biomarker to identify individuals at elevated risk who might benefit from enhanced surveillance,” Ahn told GI & Hepatology News by email.
Rates of pancreatic cancer are on the rise. But detecting the disease before it becomes unresectable has remained an elusive goal, and the US Preventive Services Task Force discourages screening of asymptomatic adults.
For their study, Ahn and her colleagues analyzed data from 122,000 participants who provided oral wash samples as part of two cohort studies conducted in the US. The researchers used whole-genome shotgun sequencing and internal transcribed spacer sequencing to identity the bacterial and fungal species in the samples, respectively.
Over a median follow-up of nearly 9 years, 445 people developed pancreatic cancer and were matched with 445 who did not. Three oral bacterial periodontal pathogens — Porphyromonas gingivalis (odds ratio [OR], 1.27), Eubacterium nodatum (OR, 1.42), and Parvimonas micra (OR, 1.36) — as well as the fungal genus Candida were all linked to significantly increased odds of developing pancreatic cancer.
In a bacteriome-wide scan, the researchers pinpointed another 20 oral bacteria associated with pancreatic cancer — eight with a decreased risk and 13 with an increased risk for the disease.
The researchers also calculated a microbial risk score, which was the weighted sum of the relative abundance of bacterial and fungal species. In a meta-analysis of data from the two cohorts, the microbial risk score derived from 23 bacterial species and four fungal species, including various Candida species, was associated with pancreatic cancer (multivariate OR per 1-SD increase in the score, 3.44; 95% CI, 2.63-4.51).
“The oral microbiota holds promise as a biomarker to identify individuals at high risk of pancreatic cancer, potentially enabling personalized pancreatic cancer prevention,” Ahn and her colleagues concluded.
But Gil Welch, MD, of Brigham and Women’s Hospital in Boston, who has written about screening for decades, isn’t so sure.
Given the “impressive volume of information” included in the analysis, “it is not surprising that the investigators are able to create a microbial risk score (based on 27 species of bacteria and fungi) that is highly related to pancreatic cancer,” Welch said. “The authors are careful to emphasize these are associations, not causal relationships.”
But even if the relationship were causal, finding more people with the malignancy can also have downsides, said Welch.
In a study out last year, Welch and colleagues found that while the incidence of pancreatic cancer among young Americans has been rising, mortality rates in this demographic haven’t budged, suggesting a potential for overdiagnosis.
“Screening for pancreatic cancer has never been shown to reduce pancreatic cancer mortality,” Welch told GI & Hepatology News. “Why screen large swaths of the population simply to enumerate ‘risk factors’ for an unproven benefit that, at best, could help only a few? Meanwhile, the burdens for everyone else are real: the mental and financial strains of ‘high risk’ labels, false alarms, and endless follow-ups. It’s a recipe to make us all worried sick — and poorer.”
Ahn reported having no disclosures. Welch reported receiving royalties from three books including “Should I be tested for cancer?”
A version of this article appeared on Medscape.com.
Helicobacter pylori May Shift Gastric Cancer Earlier
Helicobacter pylori May Shift Gastric Cancer Earlier
ORLANDO, Fl — , new data suggested.
H pylori infection is a leading risk factor for gastric carcinoma, accounting for as many as 90% of cases. As the new data show, failure to screen routinely for the bacteria could be leading to younger people developing easily preventable forms of gastric cancer, experts said.
“The most concerning and the most interesting finding for us was we found higher prevalence” of gastric cancer linked to H pylori in the younger group, Neel Patel, MD, MPH, with the Department of Pathology at Staten Island University Hospital in Staten Island, New York, told GI & Hepatology News.
“This does not mean most patients are young. Rather, it means H pylori increases the likelihood of gastric cancer appearing earlier in life compared with non-H pylori cases.”
For the study, Patel and his colleagues, who presented their findings at the annual meeting of the College of American Pathologists (CAP) 2025, used 2016-2020 data from the Nationwide Inpatient Sample, which included records for adults with primary diagnoses of gastric cancer. They looked at outcomes of those whose cancer was associated with H pylori compared with the non-H pylori group.
Among 91,670 adult hospitalizations, 1830 (2%) had gastric cancer linked to H pylori (2016-2020). Patel said the low percentage resulted from focusing solely on diagnostic codes for primary diagnoses of gastric cancer and excluding secondary diagnoses.
These cancers were twice as prevalent in patients aged 18-49 years (3.97%) as in those older than 65 years (1.65%).
Septicemia Odds Higher in H pylori Group
Patients in the H pylori group also had a higher burden of comorbidities such as anemia, chronic blood loss, and metastatic cancer, according to the data. The researcher found these patients also had significantly higher odds of septicemia (odds ratio, 1.62; 95% CI, 1.17-2.24; P = .003) and spent an average of 8 days in the hospital — two more than those with cancers not associated with the infection.
Dipti M. Karamchandani, MD, a professor of pathology at the University of Texas Southwestern Medical Center in Dallas, who was not part of the study, said the longer hospital stays and greater risk for septicemia may be related to increased comorbidities among people who get H pylori infection in general. The infection often is caused by unsanitary conditions, and the groups infected may also be more likely to experience malnutrition, anemia, or lower body reserves, for example, she said.
“Also, H pylori often causes gastric ulcers, even before causing cancer, and those patients may be prone to chronic blood loss,” Karamchandani said. “These are all reasons that these patients may be more prone to longer hospital stay.”
US Guidelines Lacking
H pylori infection is a strong predictor of gastric cancer, but it often goes undetected. “Sometimes we ignore the symptoms,” Patel said.
“There are no standard guidelines for screening for H pylori,” he added. “We need to stop the transition from H pylori to gastric cancer.”
“This abstract highlights an important issue: Gastric cancer is rising among younger adults in the US, particularly in noncardia gastric cancer, which is most often associated with Helicobacter pylori infection,” said Chul S. Hyun, MD, PhD, MPH, director of the Gastric Cancer Prevention and Screening Program at Yale School of Medicine in New Haven, Connecticut.
Hyun said the 2% of patients in the study diagnosed with gastric cancer associated with H pylori likely reflected undercoding and “incomplete capture” in the database and noted that subgroup comparisons “become difficult to interpret reliably.” By extension, the findings also underscore, “We are not adequately capturing H pylori in routine US coding and claims.”
“What we do know is that H pylori is the central, modifiable driver of risk, and that prevention efforts should focus on high prevalence populations — including Asian, Hispanic, and immigrant communities — where systematic H pylori screening remains a major unmet need,” said Hyun, who was not involved in the new research.
Currently no US society guideline recommends systematic screening, Hyun said. “Other high-incidence countries, such as Japan and Korea, already incorporate H pylori and gastroscopy screening into national policy,” he said. “For these reasons, guidelines urgently need to evolve to recommend targeted H pylori screening in high prevalence groups.”
Patel, Karamchandani, and Hyun reported having no relevant financial conflicts of interest.
A version of this article appeared on Medscape.com.
ORLANDO, Fl — , new data suggested.
H pylori infection is a leading risk factor for gastric carcinoma, accounting for as many as 90% of cases. As the new data show, failure to screen routinely for the bacteria could be leading to younger people developing easily preventable forms of gastric cancer, experts said.
“The most concerning and the most interesting finding for us was we found higher prevalence” of gastric cancer linked to H pylori in the younger group, Neel Patel, MD, MPH, with the Department of Pathology at Staten Island University Hospital in Staten Island, New York, told GI & Hepatology News.
“This does not mean most patients are young. Rather, it means H pylori increases the likelihood of gastric cancer appearing earlier in life compared with non-H pylori cases.”
For the study, Patel and his colleagues, who presented their findings at the annual meeting of the College of American Pathologists (CAP) 2025, used 2016-2020 data from the Nationwide Inpatient Sample, which included records for adults with primary diagnoses of gastric cancer. They looked at outcomes of those whose cancer was associated with H pylori compared with the non-H pylori group.
Among 91,670 adult hospitalizations, 1830 (2%) had gastric cancer linked to H pylori (2016-2020). Patel said the low percentage resulted from focusing solely on diagnostic codes for primary diagnoses of gastric cancer and excluding secondary diagnoses.
These cancers were twice as prevalent in patients aged 18-49 years (3.97%) as in those older than 65 years (1.65%).
Septicemia Odds Higher in H pylori Group
Patients in the H pylori group also had a higher burden of comorbidities such as anemia, chronic blood loss, and metastatic cancer, according to the data. The researcher found these patients also had significantly higher odds of septicemia (odds ratio, 1.62; 95% CI, 1.17-2.24; P = .003) and spent an average of 8 days in the hospital — two more than those with cancers not associated with the infection.
Dipti M. Karamchandani, MD, a professor of pathology at the University of Texas Southwestern Medical Center in Dallas, who was not part of the study, said the longer hospital stays and greater risk for septicemia may be related to increased comorbidities among people who get H pylori infection in general. The infection often is caused by unsanitary conditions, and the groups infected may also be more likely to experience malnutrition, anemia, or lower body reserves, for example, she said.
“Also, H pylori often causes gastric ulcers, even before causing cancer, and those patients may be prone to chronic blood loss,” Karamchandani said. “These are all reasons that these patients may be more prone to longer hospital stay.”
US Guidelines Lacking
H pylori infection is a strong predictor of gastric cancer, but it often goes undetected. “Sometimes we ignore the symptoms,” Patel said.
“There are no standard guidelines for screening for H pylori,” he added. “We need to stop the transition from H pylori to gastric cancer.”
“This abstract highlights an important issue: Gastric cancer is rising among younger adults in the US, particularly in noncardia gastric cancer, which is most often associated with Helicobacter pylori infection,” said Chul S. Hyun, MD, PhD, MPH, director of the Gastric Cancer Prevention and Screening Program at Yale School of Medicine in New Haven, Connecticut.
Hyun said the 2% of patients in the study diagnosed with gastric cancer associated with H pylori likely reflected undercoding and “incomplete capture” in the database and noted that subgroup comparisons “become difficult to interpret reliably.” By extension, the findings also underscore, “We are not adequately capturing H pylori in routine US coding and claims.”
“What we do know is that H pylori is the central, modifiable driver of risk, and that prevention efforts should focus on high prevalence populations — including Asian, Hispanic, and immigrant communities — where systematic H pylori screening remains a major unmet need,” said Hyun, who was not involved in the new research.
Currently no US society guideline recommends systematic screening, Hyun said. “Other high-incidence countries, such as Japan and Korea, already incorporate H pylori and gastroscopy screening into national policy,” he said. “For these reasons, guidelines urgently need to evolve to recommend targeted H pylori screening in high prevalence groups.”
Patel, Karamchandani, and Hyun reported having no relevant financial conflicts of interest.
A version of this article appeared on Medscape.com.
ORLANDO, Fl — , new data suggested.
H pylori infection is a leading risk factor for gastric carcinoma, accounting for as many as 90% of cases. As the new data show, failure to screen routinely for the bacteria could be leading to younger people developing easily preventable forms of gastric cancer, experts said.
“The most concerning and the most interesting finding for us was we found higher prevalence” of gastric cancer linked to H pylori in the younger group, Neel Patel, MD, MPH, with the Department of Pathology at Staten Island University Hospital in Staten Island, New York, told GI & Hepatology News.
“This does not mean most patients are young. Rather, it means H pylori increases the likelihood of gastric cancer appearing earlier in life compared with non-H pylori cases.”
For the study, Patel and his colleagues, who presented their findings at the annual meeting of the College of American Pathologists (CAP) 2025, used 2016-2020 data from the Nationwide Inpatient Sample, which included records for adults with primary diagnoses of gastric cancer. They looked at outcomes of those whose cancer was associated with H pylori compared with the non-H pylori group.
Among 91,670 adult hospitalizations, 1830 (2%) had gastric cancer linked to H pylori (2016-2020). Patel said the low percentage resulted from focusing solely on diagnostic codes for primary diagnoses of gastric cancer and excluding secondary diagnoses.
These cancers were twice as prevalent in patients aged 18-49 years (3.97%) as in those older than 65 years (1.65%).
Septicemia Odds Higher in H pylori Group
Patients in the H pylori group also had a higher burden of comorbidities such as anemia, chronic blood loss, and metastatic cancer, according to the data. The researcher found these patients also had significantly higher odds of septicemia (odds ratio, 1.62; 95% CI, 1.17-2.24; P = .003) and spent an average of 8 days in the hospital — two more than those with cancers not associated with the infection.
Dipti M. Karamchandani, MD, a professor of pathology at the University of Texas Southwestern Medical Center in Dallas, who was not part of the study, said the longer hospital stays and greater risk for septicemia may be related to increased comorbidities among people who get H pylori infection in general. The infection often is caused by unsanitary conditions, and the groups infected may also be more likely to experience malnutrition, anemia, or lower body reserves, for example, she said.
“Also, H pylori often causes gastric ulcers, even before causing cancer, and those patients may be prone to chronic blood loss,” Karamchandani said. “These are all reasons that these patients may be more prone to longer hospital stay.”
US Guidelines Lacking
H pylori infection is a strong predictor of gastric cancer, but it often goes undetected. “Sometimes we ignore the symptoms,” Patel said.
“There are no standard guidelines for screening for H pylori,” he added. “We need to stop the transition from H pylori to gastric cancer.”
“This abstract highlights an important issue: Gastric cancer is rising among younger adults in the US, particularly in noncardia gastric cancer, which is most often associated with Helicobacter pylori infection,” said Chul S. Hyun, MD, PhD, MPH, director of the Gastric Cancer Prevention and Screening Program at Yale School of Medicine in New Haven, Connecticut.
Hyun said the 2% of patients in the study diagnosed with gastric cancer associated with H pylori likely reflected undercoding and “incomplete capture” in the database and noted that subgroup comparisons “become difficult to interpret reliably.” By extension, the findings also underscore, “We are not adequately capturing H pylori in routine US coding and claims.”
“What we do know is that H pylori is the central, modifiable driver of risk, and that prevention efforts should focus on high prevalence populations — including Asian, Hispanic, and immigrant communities — where systematic H pylori screening remains a major unmet need,” said Hyun, who was not involved in the new research.
Currently no US society guideline recommends systematic screening, Hyun said. “Other high-incidence countries, such as Japan and Korea, already incorporate H pylori and gastroscopy screening into national policy,” he said. “For these reasons, guidelines urgently need to evolve to recommend targeted H pylori screening in high prevalence groups.”
Patel, Karamchandani, and Hyun reported having no relevant financial conflicts of interest.
A version of this article appeared on Medscape.com.
Helicobacter pylori May Shift Gastric Cancer Earlier
Helicobacter pylori May Shift Gastric Cancer Earlier