User login
Generic clofarabine injection hits the market
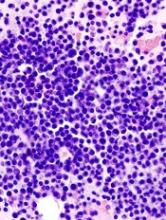
The generic clofarabine injection for treatment of children with relapsed or refractory acute lymphoblastic leukemia (ALL) is now available on the U.S. market.
The generic version of the drug was approved by the Food and Drug Administration in May 2017 for children up to age 21 years with relapsed or refractory ALL after at least two prior regimens.
The injection, marketed by Mylan N.V., is available in 20 mg/20 mL (1 mg/mL) single-dose vials. It is a generic version of Genzyme’s Clolar.
[email protected]
On Twitter @maryellenny
The generic clofarabine injection for treatment of children with relapsed or refractory acute lymphoblastic leukemia (ALL) is now available on the U.S. market.
The generic version of the drug was approved by the Food and Drug Administration in May 2017 for children up to age 21 years with relapsed or refractory ALL after at least two prior regimens.
The injection, marketed by Mylan N.V., is available in 20 mg/20 mL (1 mg/mL) single-dose vials. It is a generic version of Genzyme’s Clolar.
[email protected]
On Twitter @maryellenny
The generic clofarabine injection for treatment of children with relapsed or refractory acute lymphoblastic leukemia (ALL) is now available on the U.S. market.
The generic version of the drug was approved by the Food and Drug Administration in May 2017 for children up to age 21 years with relapsed or refractory ALL after at least two prior regimens.
The injection, marketed by Mylan N.V., is available in 20 mg/20 mL (1 mg/mL) single-dose vials. It is a generic version of Genzyme’s Clolar.
[email protected]
On Twitter @maryellenny
Cancer drug costs increasing despite competition
Cancer drug costs in the US increase substantially after launch, regardless of competition, according to a study published in the Journal of Clinical Oncology.*
Researchers studied 24 cancer drugs approved over the last 20 years and found a mean cumulative cost increase of about 37%, or 19% when adjusted for inflation.
Among drugs approved to treat hematologic malignancies, the greatest inflation-adjusted price increases were for arsenic trioxide (57%), nelarabine (55%), and rituximab (49%).
The lowest inflation-adjusted price increases were for ofatumumab (8%), clofarabine (8%), and liposomal vincristine (18%).
For this study, Daniel A. Goldstein, MD, of Emory University in Atlanta, Georgia, and his colleagues measured the monthly price trajectories of 24 cancer drugs approved by the US Food and Drug Administration. This included 10 drugs approved to treat hematologic malignancies between 1997 and 2011.
To account for discounts and rebates, the researchers used the average sales prices published by the Centers for Medicare and Medicaid Services and adjusted to general and health-related inflation rates. For each drug, the researchers calculated the cumulative and annual drug cost changes.
Results
The mean follow-up was 8 years. The mean cumulative cost increase for all 24 drugs was +36.5% (95% CI, 24.7% to 48.3%).
The general inflation-adjusted increase was +19.1% (95% CI, 11.0% to 27.2%), and the health-related inflation-adjusted increase was +8.4% (95% CI, 1.4% to 15.4%).
Only 1 of the 24 drugs studied had a price decrease over time. That drug is ziv-aflibercept, which was approved to treat metastatic colorectal cancer in 2012.
Ziv-aflibercept was launched with an annual price exceeding $110,000. After public outcry, the drug’s manufacturer, Sanofi, cut the price in half. By the end of the study’s follow-up period in 2017, the cost of ziv-aflibercept had decreased 13% (inflation-adjusted decrease of 15%, health-related inflation-adjusted decrease of 20%).
Cost changes for the drugs approved to treat hematologic malignancies are listed in the following table.
| Drug (indication, approval date, years of follow-up) | Mean monthly cost at launch | Mean annual cost change (SD) | Cumulative cost change | General and health-related inflation-adjusted change, respectively |
| Arsenic trioxide (APL, 2000, 12) | $11,455 | +6% (4) | +95% | +57%, +39% |
| Bendamustine (CLL, NHL, 2008, 8) | $6924 | +5% (5) | +50% | +32%, +21% |
| Bortezomib (MM, MCL, 2003, 12) | $5490 | +4% (3) | +63% | +31%, +16% |
| Brentuximab (lymphoma, 2011, 4) | $19,482 | +8% (0.1) | +35% | +29%, +22% |
| Clofarabine (ALL, 2004, 11) | $56,486 | +3% (3) | +31% | +8%, -4% |
| Liposomal vincristine (ALL, 2012, 3) | $34,602 | +8% (0.5) | +21% | +18%, +14% |
| Nelarabine (ALL, lymphoma, 2005, 10) | $18,513 | +6% (2) | +83% | +55%, +39% |
| Ofatumumab (CLL, 2009, 6) | $4538 | +3% (2) | +17% | +8%, -0.5% |
| Pralatrexate (lymphoma, 2009, 6) | $31,684 | +6% (4) | +43% | +31%, +21% |
| Rituximab (NHL, CLL, 1997, 12) | $4111 | +5% (0.5) | +85% | +49%, +32% |
Abbreviations: ALL, acute lymphoblastic leukemia; APL, acute promyelocytic leukemia; CLL, chronic lymphocytic leukemia; MCL, mantle cell lymphoma; MM, multiple myeloma; NHL, non-Hodgkin lymphoma; SD, standard deviation.
The researchers noted that there was a steady increase in drug costs over the study period, regardless of whether a drug was granted a new supplemental indication, the drug had a new off-label indication, or a competitor drug was approved.
The only variable that was significantly associated with price change was the amount of time that had elapsed from a drug’s launch.
This association was significant in models in which the researchers used prices adjusted to inflation (P=0.002) and health-related inflation (P=0.023). However, it was not significant when the researchers used the actual drug price (P=0.085). ![]()
*Data in the abstract differ from data in the body of the JCO paper. This article includes data from the body of the JCO paper.
Cancer drug costs in the US increase substantially after launch, regardless of competition, according to a study published in the Journal of Clinical Oncology.*
Researchers studied 24 cancer drugs approved over the last 20 years and found a mean cumulative cost increase of about 37%, or 19% when adjusted for inflation.
Among drugs approved to treat hematologic malignancies, the greatest inflation-adjusted price increases were for arsenic trioxide (57%), nelarabine (55%), and rituximab (49%).
The lowest inflation-adjusted price increases were for ofatumumab (8%), clofarabine (8%), and liposomal vincristine (18%).
For this study, Daniel A. Goldstein, MD, of Emory University in Atlanta, Georgia, and his colleagues measured the monthly price trajectories of 24 cancer drugs approved by the US Food and Drug Administration. This included 10 drugs approved to treat hematologic malignancies between 1997 and 2011.
To account for discounts and rebates, the researchers used the average sales prices published by the Centers for Medicare and Medicaid Services and adjusted to general and health-related inflation rates. For each drug, the researchers calculated the cumulative and annual drug cost changes.
Results
The mean follow-up was 8 years. The mean cumulative cost increase for all 24 drugs was +36.5% (95% CI, 24.7% to 48.3%).
The general inflation-adjusted increase was +19.1% (95% CI, 11.0% to 27.2%), and the health-related inflation-adjusted increase was +8.4% (95% CI, 1.4% to 15.4%).
Only 1 of the 24 drugs studied had a price decrease over time. That drug is ziv-aflibercept, which was approved to treat metastatic colorectal cancer in 2012.
Ziv-aflibercept was launched with an annual price exceeding $110,000. After public outcry, the drug’s manufacturer, Sanofi, cut the price in half. By the end of the study’s follow-up period in 2017, the cost of ziv-aflibercept had decreased 13% (inflation-adjusted decrease of 15%, health-related inflation-adjusted decrease of 20%).
Cost changes for the drugs approved to treat hematologic malignancies are listed in the following table.
| Drug (indication, approval date, years of follow-up) | Mean monthly cost at launch | Mean annual cost change (SD) | Cumulative cost change | General and health-related inflation-adjusted change, respectively |
| Arsenic trioxide (APL, 2000, 12) | $11,455 | +6% (4) | +95% | +57%, +39% |
| Bendamustine (CLL, NHL, 2008, 8) | $6924 | +5% (5) | +50% | +32%, +21% |
| Bortezomib (MM, MCL, 2003, 12) | $5490 | +4% (3) | +63% | +31%, +16% |
| Brentuximab (lymphoma, 2011, 4) | $19,482 | +8% (0.1) | +35% | +29%, +22% |
| Clofarabine (ALL, 2004, 11) | $56,486 | +3% (3) | +31% | +8%, -4% |
| Liposomal vincristine (ALL, 2012, 3) | $34,602 | +8% (0.5) | +21% | +18%, +14% |
| Nelarabine (ALL, lymphoma, 2005, 10) | $18,513 | +6% (2) | +83% | +55%, +39% |
| Ofatumumab (CLL, 2009, 6) | $4538 | +3% (2) | +17% | +8%, -0.5% |
| Pralatrexate (lymphoma, 2009, 6) | $31,684 | +6% (4) | +43% | +31%, +21% |
| Rituximab (NHL, CLL, 1997, 12) | $4111 | +5% (0.5) | +85% | +49%, +32% |
Abbreviations: ALL, acute lymphoblastic leukemia; APL, acute promyelocytic leukemia; CLL, chronic lymphocytic leukemia; MCL, mantle cell lymphoma; MM, multiple myeloma; NHL, non-Hodgkin lymphoma; SD, standard deviation.
The researchers noted that there was a steady increase in drug costs over the study period, regardless of whether a drug was granted a new supplemental indication, the drug had a new off-label indication, or a competitor drug was approved.
The only variable that was significantly associated with price change was the amount of time that had elapsed from a drug’s launch.
This association was significant in models in which the researchers used prices adjusted to inflation (P=0.002) and health-related inflation (P=0.023). However, it was not significant when the researchers used the actual drug price (P=0.085). ![]()
*Data in the abstract differ from data in the body of the JCO paper. This article includes data from the body of the JCO paper.
Cancer drug costs in the US increase substantially after launch, regardless of competition, according to a study published in the Journal of Clinical Oncology.*
Researchers studied 24 cancer drugs approved over the last 20 years and found a mean cumulative cost increase of about 37%, or 19% when adjusted for inflation.
Among drugs approved to treat hematologic malignancies, the greatest inflation-adjusted price increases were for arsenic trioxide (57%), nelarabine (55%), and rituximab (49%).
The lowest inflation-adjusted price increases were for ofatumumab (8%), clofarabine (8%), and liposomal vincristine (18%).
For this study, Daniel A. Goldstein, MD, of Emory University in Atlanta, Georgia, and his colleagues measured the monthly price trajectories of 24 cancer drugs approved by the US Food and Drug Administration. This included 10 drugs approved to treat hematologic malignancies between 1997 and 2011.
To account for discounts and rebates, the researchers used the average sales prices published by the Centers for Medicare and Medicaid Services and adjusted to general and health-related inflation rates. For each drug, the researchers calculated the cumulative and annual drug cost changes.
Results
The mean follow-up was 8 years. The mean cumulative cost increase for all 24 drugs was +36.5% (95% CI, 24.7% to 48.3%).
The general inflation-adjusted increase was +19.1% (95% CI, 11.0% to 27.2%), and the health-related inflation-adjusted increase was +8.4% (95% CI, 1.4% to 15.4%).
Only 1 of the 24 drugs studied had a price decrease over time. That drug is ziv-aflibercept, which was approved to treat metastatic colorectal cancer in 2012.
Ziv-aflibercept was launched with an annual price exceeding $110,000. After public outcry, the drug’s manufacturer, Sanofi, cut the price in half. By the end of the study’s follow-up period in 2017, the cost of ziv-aflibercept had decreased 13% (inflation-adjusted decrease of 15%, health-related inflation-adjusted decrease of 20%).
Cost changes for the drugs approved to treat hematologic malignancies are listed in the following table.
| Drug (indication, approval date, years of follow-up) | Mean monthly cost at launch | Mean annual cost change (SD) | Cumulative cost change | General and health-related inflation-adjusted change, respectively |
| Arsenic trioxide (APL, 2000, 12) | $11,455 | +6% (4) | +95% | +57%, +39% |
| Bendamustine (CLL, NHL, 2008, 8) | $6924 | +5% (5) | +50% | +32%, +21% |
| Bortezomib (MM, MCL, 2003, 12) | $5490 | +4% (3) | +63% | +31%, +16% |
| Brentuximab (lymphoma, 2011, 4) | $19,482 | +8% (0.1) | +35% | +29%, +22% |
| Clofarabine (ALL, 2004, 11) | $56,486 | +3% (3) | +31% | +8%, -4% |
| Liposomal vincristine (ALL, 2012, 3) | $34,602 | +8% (0.5) | +21% | +18%, +14% |
| Nelarabine (ALL, lymphoma, 2005, 10) | $18,513 | +6% (2) | +83% | +55%, +39% |
| Ofatumumab (CLL, 2009, 6) | $4538 | +3% (2) | +17% | +8%, -0.5% |
| Pralatrexate (lymphoma, 2009, 6) | $31,684 | +6% (4) | +43% | +31%, +21% |
| Rituximab (NHL, CLL, 1997, 12) | $4111 | +5% (0.5) | +85% | +49%, +32% |
Abbreviations: ALL, acute lymphoblastic leukemia; APL, acute promyelocytic leukemia; CLL, chronic lymphocytic leukemia; MCL, mantle cell lymphoma; MM, multiple myeloma; NHL, non-Hodgkin lymphoma; SD, standard deviation.
The researchers noted that there was a steady increase in drug costs over the study period, regardless of whether a drug was granted a new supplemental indication, the drug had a new off-label indication, or a competitor drug was approved.
The only variable that was significantly associated with price change was the amount of time that had elapsed from a drug’s launch.
This association was significant in models in which the researchers used prices adjusted to inflation (P=0.002) and health-related inflation (P=0.023). However, it was not significant when the researchers used the actual drug price (P=0.085). ![]()
*Data in the abstract differ from data in the body of the JCO paper. This article includes data from the body of the JCO paper.
Overcoming resistance to proteasome inhibitors
Preclinical research has revealed a potential method of overcoming resistance to proteasome inhibitors.
By studying a rare genetic disease known as NGLY1 deficiency, researchers have gained new understanding of resistance to proteasome inhibitors.
The team found that treatment with a NGLY1 inhibitor can enhance the activity of the proteasome inhibitor carfilzomib against multiple myeloma (MM) and T-cell acute lymphoblastic leukemia (T-ALL).
Carolyn Bertozzi, PhD, of Stanford University in California, and her colleagues reported these findings in ACS Central Science.
Previous studies have suggested that proteasome inhibitor resistance could be linked to a protein called Nrf1. When proteasome inhibitors swing into action, Nrf1 is spurred into overdrive to restore cancer cells’ normal activities and keep them alive.
Researchers theorized that, if they could block Nrf1, they might be able to overcome proteasome inhibitor resistance.
Through studying NGLY1 deficiency, Dr Bertozzi and her colleagues may have hit upon an approach to do just that.
The researchers were investigating how lacking NGLY1 causes a host of debilitating symptoms, and they found that NGLY1 is responsible for activating Nrf1.
Further testing revealed that inhibiting NGLY1 eliminated interference from Nrf1 and enhanced the cytotoxicity of carfilzomib in MM and T-ALL cell lines.
The researchers treated the MM cell lines U266 and H929 with the NGLY1 inhibitor, known as WRR139, and carfilzomib and observed a significant decrease in cell survival when compared to treatment with carfilzomib alone. The team observed the same results when testing the T-ALL Jurkat cell line.
The addition of WRR139 resulted in a 2.6-fold reduction in carfilzomib’s LD50 for U266, a 2.0-fold reduction for H929, and a 1.5-fold reduction for Jurkat cells.
The researchers said these findings hold promise for the development of combination therapies for hematologic malignancies. ![]()
Preclinical research has revealed a potential method of overcoming resistance to proteasome inhibitors.
By studying a rare genetic disease known as NGLY1 deficiency, researchers have gained new understanding of resistance to proteasome inhibitors.
The team found that treatment with a NGLY1 inhibitor can enhance the activity of the proteasome inhibitor carfilzomib against multiple myeloma (MM) and T-cell acute lymphoblastic leukemia (T-ALL).
Carolyn Bertozzi, PhD, of Stanford University in California, and her colleagues reported these findings in ACS Central Science.
Previous studies have suggested that proteasome inhibitor resistance could be linked to a protein called Nrf1. When proteasome inhibitors swing into action, Nrf1 is spurred into overdrive to restore cancer cells’ normal activities and keep them alive.
Researchers theorized that, if they could block Nrf1, they might be able to overcome proteasome inhibitor resistance.
Through studying NGLY1 deficiency, Dr Bertozzi and her colleagues may have hit upon an approach to do just that.
The researchers were investigating how lacking NGLY1 causes a host of debilitating symptoms, and they found that NGLY1 is responsible for activating Nrf1.
Further testing revealed that inhibiting NGLY1 eliminated interference from Nrf1 and enhanced the cytotoxicity of carfilzomib in MM and T-ALL cell lines.
The researchers treated the MM cell lines U266 and H929 with the NGLY1 inhibitor, known as WRR139, and carfilzomib and observed a significant decrease in cell survival when compared to treatment with carfilzomib alone. The team observed the same results when testing the T-ALL Jurkat cell line.
The addition of WRR139 resulted in a 2.6-fold reduction in carfilzomib’s LD50 for U266, a 2.0-fold reduction for H929, and a 1.5-fold reduction for Jurkat cells.
The researchers said these findings hold promise for the development of combination therapies for hematologic malignancies. ![]()
Preclinical research has revealed a potential method of overcoming resistance to proteasome inhibitors.
By studying a rare genetic disease known as NGLY1 deficiency, researchers have gained new understanding of resistance to proteasome inhibitors.
The team found that treatment with a NGLY1 inhibitor can enhance the activity of the proteasome inhibitor carfilzomib against multiple myeloma (MM) and T-cell acute lymphoblastic leukemia (T-ALL).
Carolyn Bertozzi, PhD, of Stanford University in California, and her colleagues reported these findings in ACS Central Science.
Previous studies have suggested that proteasome inhibitor resistance could be linked to a protein called Nrf1. When proteasome inhibitors swing into action, Nrf1 is spurred into overdrive to restore cancer cells’ normal activities and keep them alive.
Researchers theorized that, if they could block Nrf1, they might be able to overcome proteasome inhibitor resistance.
Through studying NGLY1 deficiency, Dr Bertozzi and her colleagues may have hit upon an approach to do just that.
The researchers were investigating how lacking NGLY1 causes a host of debilitating symptoms, and they found that NGLY1 is responsible for activating Nrf1.
Further testing revealed that inhibiting NGLY1 eliminated interference from Nrf1 and enhanced the cytotoxicity of carfilzomib in MM and T-ALL cell lines.
The researchers treated the MM cell lines U266 and H929 with the NGLY1 inhibitor, known as WRR139, and carfilzomib and observed a significant decrease in cell survival when compared to treatment with carfilzomib alone. The team observed the same results when testing the T-ALL Jurkat cell line.
The addition of WRR139 resulted in a 2.6-fold reduction in carfilzomib’s LD50 for U266, a 2.0-fold reduction for H929, and a 1.5-fold reduction for Jurkat cells.
The researchers said these findings hold promise for the development of combination therapies for hematologic malignancies. ![]()
ATLG fights GVHD but reduces PFS, OS
Results of a phase 3 trial suggest rabbit anti-T lymphocyte globulin (ATLG) can reduce graft-versus-host disease (GVHD) but also decrease survival in patients who have received a hematopoietic stem cell transplant (HSCT) from a matched, unrelated donor.
In this randomized trial, ATLG significantly decreased the incidence of moderate-to-severe chronic GVHD and acute grade 2-4 GVHD, when compared to placebo.
However, patients who received ATLG also had significantly lower progression-free survival (PFS) and overall survival (OS) than placebo-treated patients.
On the other hand, the data also suggest that patients who receive conditioning regimens that do not lower absolute lymphocyte counts (ALCs) substantially may not experience a significant decrease in survival with ATLG.
These results were published in the Journal of Clinical Oncology. The study was sponsored by Neovii Pharmaceuticals AG, which is developing ATLG as Grafalon®.
The study was a prospective, randomized, double-blind trial conducted in North America and Australia (NCT01295710). It enrolled 254 patients, ages 18 to 65, who had acute lymphoblastic leukemia, acute myeloid leukemia, or myelodysplastic syndromes. All patients were undergoing myeloablative, HLA-matched, unrelated HSCT.
Patients were randomized in a 1:1 fashion to receive ATLG (given at 20 mg/kg/day, n=126) or placebo (250 ml of normal saline, n=128) on days -3, -2, and -1 prior to HSCT.
In addition, all patients received antihistamine and methylprednisolone (at 2 mg/kg on day -3 and 1 mg/kg on days -2 and -1).
Patients also received GVHD prophylaxis in the form of tacrolimus (with a target serum trough level of 5 to 15 ng/mL) and methotrexate (15 mg/m2 on day 1, then 10 mg/m2 on days 3, 6, and 11). If patients did not develop clinical GVHD, tacrolimus was tapered starting on day 50 or later over a minimum of 26 weeks and ultimately discontinued.
Patients received 1 of 3 conditioning regimens, which were declared prior to randomization and included:
- Cyclophosphamide at 120 mg/kg intravenously (IV) and fractionated total body irradiation (TBI, ≥12 Gy)
- Busulfan at 16 mg/kg orally or 12.8 mg/kg IV and cyclophosphamide at 120 mg/kg IV
- Busulfan at 16 mg/kg orally or 12.8 mg/kg IV and fludarabine at 120 mg/m2 IV.
Overall results
Compared to placebo-treated patients, those who received ATLG had a significant reduction in grade 2-4 acute GVHD—23% and 40%, respectively (P=0.004)—and moderate-to-severe chronic GVHD—12% and 33%, respectively (P<0.001).
However, there was no significant difference between the ATLG and placebo arms with regard to moderate-severe chronic GVHD-free survival. The 2-year estimate was 48% and 44%, respectively (P=0.47).
In addition, PFS and OS were significantly lower in patients who received ATLG. The estimated 2-year PFS was 47% in the ATLG arm and 65% in the placebo arm (P=0.04). The estimated 2-year OS was 59% and 74%, respectively (P=0.034).
In a multivariable analysis, ATLG remained significantly associated with inferior PFS (hazard ratio [HR]=1.55, P=0.026) and OS (hazard ratio=1.74, P=0.01).
Role of conditioning, ALC
The researchers found evidence to suggest that conditioning regimen and ALC played a role in patient outcomes.
For patients who received cyclophosphamide and TBI, 2-year moderate-severe chronic GVHD-free survival was 61% in the placebo arm and 38% in the ATLG arm (P=0.080). Two-year OS was 88% and 48%, respectively (P=0.006). And 2-year PFS was 75% and 29%, respectively (P=0.007).
For patients who received busulfan and cyclophosphamide, 2-year moderate-severe chronic GVHD-free survival was 47% in the placebo arm and 53% in the ATLG arm (P=0.650). Two-year OS was 77% and 71%, respectively (P=0.350). And 2-year PFS was 73% and 60%, respectively (P=0.460).
For patients who received busulfan and fludarabine, 2-year moderate-severe chronic GVHD-free survival was 33% in the placebo arm and 49% in the ATLG arm (P=0.047). Two-year OS was 66% and 53%, respectively (P=0.520). And 2-year PFS was 58% and 48%, respectively (P=0.540).
The researchers noted that the choice of conditioning regimen had a “profound effect” on ALC at day -3 (the time of ATLG/placebo initiation). More than 70% of patients who received TBI had an ALC <0.1 x 109/L, compared to less than 35% of patients who received busulfan-based conditioning.
ALC, in turn, had an impact on PFS and OS. In patients with an ALC ≥ 0.1 x 109/L on day -3, ATLG did not compromise PFS or OS, but PFS and OS were negatively affected in patients with an ALC < 0.1.
ATLG recipients with an ALC < 0.1 had significantly worse OS (HR=4.13, P<0.001) and PFS (HR=3.19, P<0.001) than patients with an ALC ≥ 0.1. ![]()
Results of a phase 3 trial suggest rabbit anti-T lymphocyte globulin (ATLG) can reduce graft-versus-host disease (GVHD) but also decrease survival in patients who have received a hematopoietic stem cell transplant (HSCT) from a matched, unrelated donor.
In this randomized trial, ATLG significantly decreased the incidence of moderate-to-severe chronic GVHD and acute grade 2-4 GVHD, when compared to placebo.
However, patients who received ATLG also had significantly lower progression-free survival (PFS) and overall survival (OS) than placebo-treated patients.
On the other hand, the data also suggest that patients who receive conditioning regimens that do not lower absolute lymphocyte counts (ALCs) substantially may not experience a significant decrease in survival with ATLG.
These results were published in the Journal of Clinical Oncology. The study was sponsored by Neovii Pharmaceuticals AG, which is developing ATLG as Grafalon®.
The study was a prospective, randomized, double-blind trial conducted in North America and Australia (NCT01295710). It enrolled 254 patients, ages 18 to 65, who had acute lymphoblastic leukemia, acute myeloid leukemia, or myelodysplastic syndromes. All patients were undergoing myeloablative, HLA-matched, unrelated HSCT.
Patients were randomized in a 1:1 fashion to receive ATLG (given at 20 mg/kg/day, n=126) or placebo (250 ml of normal saline, n=128) on days -3, -2, and -1 prior to HSCT.
In addition, all patients received antihistamine and methylprednisolone (at 2 mg/kg on day -3 and 1 mg/kg on days -2 and -1).
Patients also received GVHD prophylaxis in the form of tacrolimus (with a target serum trough level of 5 to 15 ng/mL) and methotrexate (15 mg/m2 on day 1, then 10 mg/m2 on days 3, 6, and 11). If patients did not develop clinical GVHD, tacrolimus was tapered starting on day 50 or later over a minimum of 26 weeks and ultimately discontinued.
Patients received 1 of 3 conditioning regimens, which were declared prior to randomization and included:
- Cyclophosphamide at 120 mg/kg intravenously (IV) and fractionated total body irradiation (TBI, ≥12 Gy)
- Busulfan at 16 mg/kg orally or 12.8 mg/kg IV and cyclophosphamide at 120 mg/kg IV
- Busulfan at 16 mg/kg orally or 12.8 mg/kg IV and fludarabine at 120 mg/m2 IV.
Overall results
Compared to placebo-treated patients, those who received ATLG had a significant reduction in grade 2-4 acute GVHD—23% and 40%, respectively (P=0.004)—and moderate-to-severe chronic GVHD—12% and 33%, respectively (P<0.001).
However, there was no significant difference between the ATLG and placebo arms with regard to moderate-severe chronic GVHD-free survival. The 2-year estimate was 48% and 44%, respectively (P=0.47).
In addition, PFS and OS were significantly lower in patients who received ATLG. The estimated 2-year PFS was 47% in the ATLG arm and 65% in the placebo arm (P=0.04). The estimated 2-year OS was 59% and 74%, respectively (P=0.034).
In a multivariable analysis, ATLG remained significantly associated with inferior PFS (hazard ratio [HR]=1.55, P=0.026) and OS (hazard ratio=1.74, P=0.01).
Role of conditioning, ALC
The researchers found evidence to suggest that conditioning regimen and ALC played a role in patient outcomes.
For patients who received cyclophosphamide and TBI, 2-year moderate-severe chronic GVHD-free survival was 61% in the placebo arm and 38% in the ATLG arm (P=0.080). Two-year OS was 88% and 48%, respectively (P=0.006). And 2-year PFS was 75% and 29%, respectively (P=0.007).
For patients who received busulfan and cyclophosphamide, 2-year moderate-severe chronic GVHD-free survival was 47% in the placebo arm and 53% in the ATLG arm (P=0.650). Two-year OS was 77% and 71%, respectively (P=0.350). And 2-year PFS was 73% and 60%, respectively (P=0.460).
For patients who received busulfan and fludarabine, 2-year moderate-severe chronic GVHD-free survival was 33% in the placebo arm and 49% in the ATLG arm (P=0.047). Two-year OS was 66% and 53%, respectively (P=0.520). And 2-year PFS was 58% and 48%, respectively (P=0.540).
The researchers noted that the choice of conditioning regimen had a “profound effect” on ALC at day -3 (the time of ATLG/placebo initiation). More than 70% of patients who received TBI had an ALC <0.1 x 109/L, compared to less than 35% of patients who received busulfan-based conditioning.
ALC, in turn, had an impact on PFS and OS. In patients with an ALC ≥ 0.1 x 109/L on day -3, ATLG did not compromise PFS or OS, but PFS and OS were negatively affected in patients with an ALC < 0.1.
ATLG recipients with an ALC < 0.1 had significantly worse OS (HR=4.13, P<0.001) and PFS (HR=3.19, P<0.001) than patients with an ALC ≥ 0.1. ![]()
Results of a phase 3 trial suggest rabbit anti-T lymphocyte globulin (ATLG) can reduce graft-versus-host disease (GVHD) but also decrease survival in patients who have received a hematopoietic stem cell transplant (HSCT) from a matched, unrelated donor.
In this randomized trial, ATLG significantly decreased the incidence of moderate-to-severe chronic GVHD and acute grade 2-4 GVHD, when compared to placebo.
However, patients who received ATLG also had significantly lower progression-free survival (PFS) and overall survival (OS) than placebo-treated patients.
On the other hand, the data also suggest that patients who receive conditioning regimens that do not lower absolute lymphocyte counts (ALCs) substantially may not experience a significant decrease in survival with ATLG.
These results were published in the Journal of Clinical Oncology. The study was sponsored by Neovii Pharmaceuticals AG, which is developing ATLG as Grafalon®.
The study was a prospective, randomized, double-blind trial conducted in North America and Australia (NCT01295710). It enrolled 254 patients, ages 18 to 65, who had acute lymphoblastic leukemia, acute myeloid leukemia, or myelodysplastic syndromes. All patients were undergoing myeloablative, HLA-matched, unrelated HSCT.
Patients were randomized in a 1:1 fashion to receive ATLG (given at 20 mg/kg/day, n=126) or placebo (250 ml of normal saline, n=128) on days -3, -2, and -1 prior to HSCT.
In addition, all patients received antihistamine and methylprednisolone (at 2 mg/kg on day -3 and 1 mg/kg on days -2 and -1).
Patients also received GVHD prophylaxis in the form of tacrolimus (with a target serum trough level of 5 to 15 ng/mL) and methotrexate (15 mg/m2 on day 1, then 10 mg/m2 on days 3, 6, and 11). If patients did not develop clinical GVHD, tacrolimus was tapered starting on day 50 or later over a minimum of 26 weeks and ultimately discontinued.
Patients received 1 of 3 conditioning regimens, which were declared prior to randomization and included:
- Cyclophosphamide at 120 mg/kg intravenously (IV) and fractionated total body irradiation (TBI, ≥12 Gy)
- Busulfan at 16 mg/kg orally or 12.8 mg/kg IV and cyclophosphamide at 120 mg/kg IV
- Busulfan at 16 mg/kg orally or 12.8 mg/kg IV and fludarabine at 120 mg/m2 IV.
Overall results
Compared to placebo-treated patients, those who received ATLG had a significant reduction in grade 2-4 acute GVHD—23% and 40%, respectively (P=0.004)—and moderate-to-severe chronic GVHD—12% and 33%, respectively (P<0.001).
However, there was no significant difference between the ATLG and placebo arms with regard to moderate-severe chronic GVHD-free survival. The 2-year estimate was 48% and 44%, respectively (P=0.47).
In addition, PFS and OS were significantly lower in patients who received ATLG. The estimated 2-year PFS was 47% in the ATLG arm and 65% in the placebo arm (P=0.04). The estimated 2-year OS was 59% and 74%, respectively (P=0.034).
In a multivariable analysis, ATLG remained significantly associated with inferior PFS (hazard ratio [HR]=1.55, P=0.026) and OS (hazard ratio=1.74, P=0.01).
Role of conditioning, ALC
The researchers found evidence to suggest that conditioning regimen and ALC played a role in patient outcomes.
For patients who received cyclophosphamide and TBI, 2-year moderate-severe chronic GVHD-free survival was 61% in the placebo arm and 38% in the ATLG arm (P=0.080). Two-year OS was 88% and 48%, respectively (P=0.006). And 2-year PFS was 75% and 29%, respectively (P=0.007).
For patients who received busulfan and cyclophosphamide, 2-year moderate-severe chronic GVHD-free survival was 47% in the placebo arm and 53% in the ATLG arm (P=0.650). Two-year OS was 77% and 71%, respectively (P=0.350). And 2-year PFS was 73% and 60%, respectively (P=0.460).
For patients who received busulfan and fludarabine, 2-year moderate-severe chronic GVHD-free survival was 33% in the placebo arm and 49% in the ATLG arm (P=0.047). Two-year OS was 66% and 53%, respectively (P=0.520). And 2-year PFS was 58% and 48%, respectively (P=0.540).
The researchers noted that the choice of conditioning regimen had a “profound effect” on ALC at day -3 (the time of ATLG/placebo initiation). More than 70% of patients who received TBI had an ALC <0.1 x 109/L, compared to less than 35% of patients who received busulfan-based conditioning.
ALC, in turn, had an impact on PFS and OS. In patients with an ALC ≥ 0.1 x 109/L on day -3, ATLG did not compromise PFS or OS, but PFS and OS were negatively affected in patients with an ALC < 0.1.
ATLG recipients with an ALC < 0.1 had significantly worse OS (HR=4.13, P<0.001) and PFS (HR=3.19, P<0.001) than patients with an ALC ≥ 0.1. ![]()
Cascade of costs could push new gene therapy above $1 million per patient
Outrage over the high cost of cancer care has focused on skyrocketing drug prices, including the $475,000 price tag for the country’s first gene therapy, Novartis’ Kymriah (tisagenlecleucel), a leukemia treatment approved in August.
But the total costs of tisagenlecleucel and the 21 similar drugs in development – known as CAR T-cell therapies – will be far higher than many have imagined, reaching $1 million or more per patient, according to leading cancer experts. The next CAR T-cell drug could be approved as soon as November.
Although Kymriah’s price tag has “shattered oncology drug pricing norms,” said Leonard Saltz, MD, chief of gastrointestinal oncology at Memorial Sloan Kettering Cancer Center in New York, “the sticker price is just the starting point.”
These therapies lead to a cascade of costs, propelled by serious side effects that require sophisticated management, Dr. Saltz said. For this class of drugs, Dr, Saltz advised consumers to “think of the $475,000 as parts, not labor.”
Hagop Kantarjian, MD, leukemia specialist and professor at the University of Texas MD Anderson Cancer Center, estimates tisagenlecleucel’s total cost could reach $1.5 million.
CAR T-cell therapy is expensive because of the unique way that it works. Doctors harvest patients’ immune cells, genetically alter them to rev up their ability to fight cancer, then reinfuse them into patients.
Taking the brakes off the immune system, Dr. Kantarjian said, can lead to life-threatening complications that require lengthy hospitalizations and expensive medications, which are prescribed in addition to conventional cancer therapy, rather than in place of it.
Keith D. Eaton, MD, a Seattle oncologist, said he ran up medical bills of $500,000 when he participated in a clinical trial of CAR T cells in 2013, even though all patients in the study received the medication for free. Dr. Eaton, who was diagnosed with acute lymphoblastic leukemia (ALL), spent nearly 2 months in the hospital.
Like Dr. Eaton, nearly half of patients who receive CAR T cells develop cytokine storm. Other serious side effects include strokelike symptoms and coma.
The cytokine storm felt like “the worst flu of your life,” said Dr. Eaton, now aged 51 years. His fever spiked so high that a hospital nurse assumed the thermometer was broken. Dr. Eaton replied, “It’s not broken. My temperature is too high to register on the thermometer.”
Although Dr. Eaton recovered, he wasn’t done with treatment. His doctors recommended a bone marrow transplant, another harrowing procedure, at a cost of hundreds of thousands of dollars.
Dr. Eaton said he feels fortunate to be healthy today, with tests showing no evidence of leukemia. His insurer paid for almost everything.
Kymriah’s sticker price is especially “outrageous” given its relatively low manufacturing costs, said Walid F. Gellad, MD, codirector of the Center for Pharmaceutical Policy and Prescribing at the University of Pittsburgh.
The gene therapy process used to create tisagenlecleucel costs about $15,000, according to a 2012 presentation by Carl H. June, MD, who pioneered CAR T-cell research at the University of Pennsylvania in Philadelphia. Dr. June could not be reached for comment.
To quell unrest about price, Novartis has offered patients and insurers a new twist on the money-back guarantee.
Novartis will charge for the drug only if patients go into remission within 1 month of treatment. In a key clinical trial, 83% of the children and young adults treated with tisagenlecleucel went into remission within 3 months. Novartis calls the plan “outcomes-based pricing.”
Novartis is “working through the specific details” of how the pricing plan will affect the Centers for Medicare & Medicaid Services, which pays for care for many cancer patients, company spokesperson Julie Masow said. “There are many hurdles” to this type of pricing plan but, Ms. Masow said, “Novartis is committed to making this happen.”
She also said that Kymriah’s manufacturing costs are much higher than $15,000, although she didn’t cite a specific dollar amount. She noted that Novartis has invested heavily in the technology, designing “an innovative manufacturing facility and process specifically for cellular therapies.”
As for Kymriah-related hospital and medication charges, “costs will vary from patient to patient and treatment center to treatment center, based on the level of care each patient requires,” Ms. Masow said. “Kymriah is a one-time treatment that has shown remarkable early, deep, and durable responses in these children who are very sick and often out of options.”
Some doctors said tisagenlecleucel, which could be used by about 600 patients a year, offers an incalculable benefit for desperately ill young people. The drug is approved for children and young adults with B-cell ALL who already have been treated with at least two other cancer therapies.
“A kid’s life is priceless,” said Michelle Hermiston, MD, director of pediatric immunotherapy at Benioff Children’s Hospital, at the University of California, San Francisco. “Any given kid has the potential to make financial impacts over a lifetime that far outweigh the cost of their cure. From this perspective, every child in my mind deserves the best curative therapy we can offer.”
Other cancer doctors say the Novartis plan is no bargain.
About 36% of patients who go into remission with tisagenlecleucel relapse within 1 year, said Vinay Prasad, MD, of Oregon Health & Science University, Portland. Many of these patients will need additional treatment, said Dr. Prasad, who wrote an editorial about tisagenlecleucel’s price Oct. 4 in Nature.
“If you’ve paid half a million dollars for drugs and half a million dollars for care, and a year later your cancer is back, is that a good deal?” asked Dr. Saltz, who cowrote a recent editorial on tisagenlecleucel’s price in JAMA.
Steve Miller, MD, chief medical officer for Express Scripts, said it would be more fair to judge Kymriah’s success after 6 months of treatment, rather than 1 month. Dr. Prasad goes even further. He said Novartis should issue refunds for any patient who relapses within 3 years.
A consumer-advocate group called Patients for Affordable Drugs also has said that tisagenlecleucel costs too much, given that the federal government spent more than $200 million over 2 decades to support the basic research into CAR T-cell therapy, long before Novartis bought the rights.
Rep. Lloyd Doggett (D-Texas) wrote a letter to the Medicare program’s director last month asking for details on how the Novartis payment deal will work.
“As Big Pharma continues to put price gouging before patient access, companies will point more and more proudly at their pricing agreements,” Rep. Doggett wrote. “But taxpayers deserve to know more about how these agreements will work – whether they will actually save the government money, defray these massive costs, and ensure that they can access lifesaving medications.”
KHN’s coverage related to aging & improving care of older adults is supported by The John A. Hartford Foundation. Kaiser Health News is a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation.
Outrage over the high cost of cancer care has focused on skyrocketing drug prices, including the $475,000 price tag for the country’s first gene therapy, Novartis’ Kymriah (tisagenlecleucel), a leukemia treatment approved in August.
But the total costs of tisagenlecleucel and the 21 similar drugs in development – known as CAR T-cell therapies – will be far higher than many have imagined, reaching $1 million or more per patient, according to leading cancer experts. The next CAR T-cell drug could be approved as soon as November.
Although Kymriah’s price tag has “shattered oncology drug pricing norms,” said Leonard Saltz, MD, chief of gastrointestinal oncology at Memorial Sloan Kettering Cancer Center in New York, “the sticker price is just the starting point.”
These therapies lead to a cascade of costs, propelled by serious side effects that require sophisticated management, Dr. Saltz said. For this class of drugs, Dr, Saltz advised consumers to “think of the $475,000 as parts, not labor.”
Hagop Kantarjian, MD, leukemia specialist and professor at the University of Texas MD Anderson Cancer Center, estimates tisagenlecleucel’s total cost could reach $1.5 million.
CAR T-cell therapy is expensive because of the unique way that it works. Doctors harvest patients’ immune cells, genetically alter them to rev up their ability to fight cancer, then reinfuse them into patients.
Taking the brakes off the immune system, Dr. Kantarjian said, can lead to life-threatening complications that require lengthy hospitalizations and expensive medications, which are prescribed in addition to conventional cancer therapy, rather than in place of it.
Keith D. Eaton, MD, a Seattle oncologist, said he ran up medical bills of $500,000 when he participated in a clinical trial of CAR T cells in 2013, even though all patients in the study received the medication for free. Dr. Eaton, who was diagnosed with acute lymphoblastic leukemia (ALL), spent nearly 2 months in the hospital.
Like Dr. Eaton, nearly half of patients who receive CAR T cells develop cytokine storm. Other serious side effects include strokelike symptoms and coma.
The cytokine storm felt like “the worst flu of your life,” said Dr. Eaton, now aged 51 years. His fever spiked so high that a hospital nurse assumed the thermometer was broken. Dr. Eaton replied, “It’s not broken. My temperature is too high to register on the thermometer.”
Although Dr. Eaton recovered, he wasn’t done with treatment. His doctors recommended a bone marrow transplant, another harrowing procedure, at a cost of hundreds of thousands of dollars.
Dr. Eaton said he feels fortunate to be healthy today, with tests showing no evidence of leukemia. His insurer paid for almost everything.
Kymriah’s sticker price is especially “outrageous” given its relatively low manufacturing costs, said Walid F. Gellad, MD, codirector of the Center for Pharmaceutical Policy and Prescribing at the University of Pittsburgh.
The gene therapy process used to create tisagenlecleucel costs about $15,000, according to a 2012 presentation by Carl H. June, MD, who pioneered CAR T-cell research at the University of Pennsylvania in Philadelphia. Dr. June could not be reached for comment.
To quell unrest about price, Novartis has offered patients and insurers a new twist on the money-back guarantee.
Novartis will charge for the drug only if patients go into remission within 1 month of treatment. In a key clinical trial, 83% of the children and young adults treated with tisagenlecleucel went into remission within 3 months. Novartis calls the plan “outcomes-based pricing.”
Novartis is “working through the specific details” of how the pricing plan will affect the Centers for Medicare & Medicaid Services, which pays for care for many cancer patients, company spokesperson Julie Masow said. “There are many hurdles” to this type of pricing plan but, Ms. Masow said, “Novartis is committed to making this happen.”
She also said that Kymriah’s manufacturing costs are much higher than $15,000, although she didn’t cite a specific dollar amount. She noted that Novartis has invested heavily in the technology, designing “an innovative manufacturing facility and process specifically for cellular therapies.”
As for Kymriah-related hospital and medication charges, “costs will vary from patient to patient and treatment center to treatment center, based on the level of care each patient requires,” Ms. Masow said. “Kymriah is a one-time treatment that has shown remarkable early, deep, and durable responses in these children who are very sick and often out of options.”
Some doctors said tisagenlecleucel, which could be used by about 600 patients a year, offers an incalculable benefit for desperately ill young people. The drug is approved for children and young adults with B-cell ALL who already have been treated with at least two other cancer therapies.
“A kid’s life is priceless,” said Michelle Hermiston, MD, director of pediatric immunotherapy at Benioff Children’s Hospital, at the University of California, San Francisco. “Any given kid has the potential to make financial impacts over a lifetime that far outweigh the cost of their cure. From this perspective, every child in my mind deserves the best curative therapy we can offer.”
Other cancer doctors say the Novartis plan is no bargain.
About 36% of patients who go into remission with tisagenlecleucel relapse within 1 year, said Vinay Prasad, MD, of Oregon Health & Science University, Portland. Many of these patients will need additional treatment, said Dr. Prasad, who wrote an editorial about tisagenlecleucel’s price Oct. 4 in Nature.
“If you’ve paid half a million dollars for drugs and half a million dollars for care, and a year later your cancer is back, is that a good deal?” asked Dr. Saltz, who cowrote a recent editorial on tisagenlecleucel’s price in JAMA.
Steve Miller, MD, chief medical officer for Express Scripts, said it would be more fair to judge Kymriah’s success after 6 months of treatment, rather than 1 month. Dr. Prasad goes even further. He said Novartis should issue refunds for any patient who relapses within 3 years.
A consumer-advocate group called Patients for Affordable Drugs also has said that tisagenlecleucel costs too much, given that the federal government spent more than $200 million over 2 decades to support the basic research into CAR T-cell therapy, long before Novartis bought the rights.
Rep. Lloyd Doggett (D-Texas) wrote a letter to the Medicare program’s director last month asking for details on how the Novartis payment deal will work.
“As Big Pharma continues to put price gouging before patient access, companies will point more and more proudly at their pricing agreements,” Rep. Doggett wrote. “But taxpayers deserve to know more about how these agreements will work – whether they will actually save the government money, defray these massive costs, and ensure that they can access lifesaving medications.”
KHN’s coverage related to aging & improving care of older adults is supported by The John A. Hartford Foundation. Kaiser Health News is a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation.
Outrage over the high cost of cancer care has focused on skyrocketing drug prices, including the $475,000 price tag for the country’s first gene therapy, Novartis’ Kymriah (tisagenlecleucel), a leukemia treatment approved in August.
But the total costs of tisagenlecleucel and the 21 similar drugs in development – known as CAR T-cell therapies – will be far higher than many have imagined, reaching $1 million or more per patient, according to leading cancer experts. The next CAR T-cell drug could be approved as soon as November.
Although Kymriah’s price tag has “shattered oncology drug pricing norms,” said Leonard Saltz, MD, chief of gastrointestinal oncology at Memorial Sloan Kettering Cancer Center in New York, “the sticker price is just the starting point.”
These therapies lead to a cascade of costs, propelled by serious side effects that require sophisticated management, Dr. Saltz said. For this class of drugs, Dr, Saltz advised consumers to “think of the $475,000 as parts, not labor.”
Hagop Kantarjian, MD, leukemia specialist and professor at the University of Texas MD Anderson Cancer Center, estimates tisagenlecleucel’s total cost could reach $1.5 million.
CAR T-cell therapy is expensive because of the unique way that it works. Doctors harvest patients’ immune cells, genetically alter them to rev up their ability to fight cancer, then reinfuse them into patients.
Taking the brakes off the immune system, Dr. Kantarjian said, can lead to life-threatening complications that require lengthy hospitalizations and expensive medications, which are prescribed in addition to conventional cancer therapy, rather than in place of it.
Keith D. Eaton, MD, a Seattle oncologist, said he ran up medical bills of $500,000 when he participated in a clinical trial of CAR T cells in 2013, even though all patients in the study received the medication for free. Dr. Eaton, who was diagnosed with acute lymphoblastic leukemia (ALL), spent nearly 2 months in the hospital.
Like Dr. Eaton, nearly half of patients who receive CAR T cells develop cytokine storm. Other serious side effects include strokelike symptoms and coma.
The cytokine storm felt like “the worst flu of your life,” said Dr. Eaton, now aged 51 years. His fever spiked so high that a hospital nurse assumed the thermometer was broken. Dr. Eaton replied, “It’s not broken. My temperature is too high to register on the thermometer.”
Although Dr. Eaton recovered, he wasn’t done with treatment. His doctors recommended a bone marrow transplant, another harrowing procedure, at a cost of hundreds of thousands of dollars.
Dr. Eaton said he feels fortunate to be healthy today, with tests showing no evidence of leukemia. His insurer paid for almost everything.
Kymriah’s sticker price is especially “outrageous” given its relatively low manufacturing costs, said Walid F. Gellad, MD, codirector of the Center for Pharmaceutical Policy and Prescribing at the University of Pittsburgh.
The gene therapy process used to create tisagenlecleucel costs about $15,000, according to a 2012 presentation by Carl H. June, MD, who pioneered CAR T-cell research at the University of Pennsylvania in Philadelphia. Dr. June could not be reached for comment.
To quell unrest about price, Novartis has offered patients and insurers a new twist on the money-back guarantee.
Novartis will charge for the drug only if patients go into remission within 1 month of treatment. In a key clinical trial, 83% of the children and young adults treated with tisagenlecleucel went into remission within 3 months. Novartis calls the plan “outcomes-based pricing.”
Novartis is “working through the specific details” of how the pricing plan will affect the Centers for Medicare & Medicaid Services, which pays for care for many cancer patients, company spokesperson Julie Masow said. “There are many hurdles” to this type of pricing plan but, Ms. Masow said, “Novartis is committed to making this happen.”
She also said that Kymriah’s manufacturing costs are much higher than $15,000, although she didn’t cite a specific dollar amount. She noted that Novartis has invested heavily in the technology, designing “an innovative manufacturing facility and process specifically for cellular therapies.”
As for Kymriah-related hospital and medication charges, “costs will vary from patient to patient and treatment center to treatment center, based on the level of care each patient requires,” Ms. Masow said. “Kymriah is a one-time treatment that has shown remarkable early, deep, and durable responses in these children who are very sick and often out of options.”
Some doctors said tisagenlecleucel, which could be used by about 600 patients a year, offers an incalculable benefit for desperately ill young people. The drug is approved for children and young adults with B-cell ALL who already have been treated with at least two other cancer therapies.
“A kid’s life is priceless,” said Michelle Hermiston, MD, director of pediatric immunotherapy at Benioff Children’s Hospital, at the University of California, San Francisco. “Any given kid has the potential to make financial impacts over a lifetime that far outweigh the cost of their cure. From this perspective, every child in my mind deserves the best curative therapy we can offer.”
Other cancer doctors say the Novartis plan is no bargain.
About 36% of patients who go into remission with tisagenlecleucel relapse within 1 year, said Vinay Prasad, MD, of Oregon Health & Science University, Portland. Many of these patients will need additional treatment, said Dr. Prasad, who wrote an editorial about tisagenlecleucel’s price Oct. 4 in Nature.
“If you’ve paid half a million dollars for drugs and half a million dollars for care, and a year later your cancer is back, is that a good deal?” asked Dr. Saltz, who cowrote a recent editorial on tisagenlecleucel’s price in JAMA.
Steve Miller, MD, chief medical officer for Express Scripts, said it would be more fair to judge Kymriah’s success after 6 months of treatment, rather than 1 month. Dr. Prasad goes even further. He said Novartis should issue refunds for any patient who relapses within 3 years.
A consumer-advocate group called Patients for Affordable Drugs also has said that tisagenlecleucel costs too much, given that the federal government spent more than $200 million over 2 decades to support the basic research into CAR T-cell therapy, long before Novartis bought the rights.
Rep. Lloyd Doggett (D-Texas) wrote a letter to the Medicare program’s director last month asking for details on how the Novartis payment deal will work.
“As Big Pharma continues to put price gouging before patient access, companies will point more and more proudly at their pricing agreements,” Rep. Doggett wrote. “But taxpayers deserve to know more about how these agreements will work – whether they will actually save the government money, defray these massive costs, and ensure that they can access lifesaving medications.”
KHN’s coverage related to aging & improving care of older adults is supported by The John A. Hartford Foundation. Kaiser Health News is a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation.
Flu vaccine appears ineffective in young leukemia patients
Vaccination may fail to protect young leukemia patients from developing influenza during cancer treatment, according to research published in the Journal of Pediatrics.
Researchers found that young patients with acute leukemia who received flu shots were just as likely as their unvaccinated peers to develop the flu.
The team said these results are preliminary, but they suggest a need for more research and additional efforts to prevent flu in young patients with leukemia.
“The annual flu shot, whose side effects are generally mild and short-lived, is still recommended for patients with acute leukemia who are being treated for their disease,” said study author Elisabeth Adderson, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“However, the results do highlight the need for additional research in this area and for us to redouble our efforts to protect our patients through other means.”
In this retrospective study, Dr Adderson and her colleagues looked at rates of flu infection during 3 successive flu seasons (2010-2013) in 498 patients treated for acute leukemia at St. Jude.
The patients’ median age was 6 years (range, 1-21). Most patients had acute lymphoblastic leukemia (ALL, 94%), though some had acute myeloid leukemia (4.8%) or mixed-lineage leukemia (1.2%).
Most patients (n=354) received flu shots, including 98 patients who received booster doses. The remaining 144 patients were not vaccinated.
The vaccinated patients received the trivalent vaccine, which is designed to protect against 3 flu strains predicted to be in wide circulation during a particular flu season. The vaccine was a fairly good match for circulating flu viruses during the flu seasons included in this analysis.
Demographic characteristic were largely similar between vaccinated and unvaccinated patients. The exceptions were that more vaccinated patients had ALL (95.5% vs 90.3%; P=0.034) and vaccinated patients were more likely to be in a low-intensity phase of cancer therapy (90.7% vs 73.6%, P<0.0001).
Results
There were no significant differences between vaccinated and unvaccinated patients when it came to flu rates or rates of flu-like illnesses.
There were 37 episodes of flu in vaccinated patients and 16 episodes in unvaccinated patients. The rates (per 1000 patient days) were 0.73 and 0.70, respectively (P=0.874).
There were 123 cases of flu-like illnesses in vaccinated patients and 55 cases in unvaccinated patients. The rates were 2.44 and 2.41, respectively (P=0.932).
Likewise, there was no significant difference in the rates of flu or flu-like illnesses between patients who received 1 dose of flu vaccine and those who received 2 doses.
The flu rates were 0.60 and 1.02, respectively (P=0.107). And the rates of flu-like illnesses were 2.42 and 2.73, respectively (P=0.529).
Dr Adderson said additional research is needed to determine if a subset of young leukemia patients may benefit from vaccination.
She added that patients at risk of flu should practice good hand hygiene and avoid crowds during the flu season. Patients may also benefit from “cocooning,” a process that focuses on getting family members, healthcare providers, and others in close contact with at-risk patients vaccinated. ![]()
Vaccination may fail to protect young leukemia patients from developing influenza during cancer treatment, according to research published in the Journal of Pediatrics.
Researchers found that young patients with acute leukemia who received flu shots were just as likely as their unvaccinated peers to develop the flu.
The team said these results are preliminary, but they suggest a need for more research and additional efforts to prevent flu in young patients with leukemia.
“The annual flu shot, whose side effects are generally mild and short-lived, is still recommended for patients with acute leukemia who are being treated for their disease,” said study author Elisabeth Adderson, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“However, the results do highlight the need for additional research in this area and for us to redouble our efforts to protect our patients through other means.”
In this retrospective study, Dr Adderson and her colleagues looked at rates of flu infection during 3 successive flu seasons (2010-2013) in 498 patients treated for acute leukemia at St. Jude.
The patients’ median age was 6 years (range, 1-21). Most patients had acute lymphoblastic leukemia (ALL, 94%), though some had acute myeloid leukemia (4.8%) or mixed-lineage leukemia (1.2%).
Most patients (n=354) received flu shots, including 98 patients who received booster doses. The remaining 144 patients were not vaccinated.
The vaccinated patients received the trivalent vaccine, which is designed to protect against 3 flu strains predicted to be in wide circulation during a particular flu season. The vaccine was a fairly good match for circulating flu viruses during the flu seasons included in this analysis.
Demographic characteristic were largely similar between vaccinated and unvaccinated patients. The exceptions were that more vaccinated patients had ALL (95.5% vs 90.3%; P=0.034) and vaccinated patients were more likely to be in a low-intensity phase of cancer therapy (90.7% vs 73.6%, P<0.0001).
Results
There were no significant differences between vaccinated and unvaccinated patients when it came to flu rates or rates of flu-like illnesses.
There were 37 episodes of flu in vaccinated patients and 16 episodes in unvaccinated patients. The rates (per 1000 patient days) were 0.73 and 0.70, respectively (P=0.874).
There were 123 cases of flu-like illnesses in vaccinated patients and 55 cases in unvaccinated patients. The rates were 2.44 and 2.41, respectively (P=0.932).
Likewise, there was no significant difference in the rates of flu or flu-like illnesses between patients who received 1 dose of flu vaccine and those who received 2 doses.
The flu rates were 0.60 and 1.02, respectively (P=0.107). And the rates of flu-like illnesses were 2.42 and 2.73, respectively (P=0.529).
Dr Adderson said additional research is needed to determine if a subset of young leukemia patients may benefit from vaccination.
She added that patients at risk of flu should practice good hand hygiene and avoid crowds during the flu season. Patients may also benefit from “cocooning,” a process that focuses on getting family members, healthcare providers, and others in close contact with at-risk patients vaccinated. ![]()
Vaccination may fail to protect young leukemia patients from developing influenza during cancer treatment, according to research published in the Journal of Pediatrics.
Researchers found that young patients with acute leukemia who received flu shots were just as likely as their unvaccinated peers to develop the flu.
The team said these results are preliminary, but they suggest a need for more research and additional efforts to prevent flu in young patients with leukemia.
“The annual flu shot, whose side effects are generally mild and short-lived, is still recommended for patients with acute leukemia who are being treated for their disease,” said study author Elisabeth Adderson, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“However, the results do highlight the need for additional research in this area and for us to redouble our efforts to protect our patients through other means.”
In this retrospective study, Dr Adderson and her colleagues looked at rates of flu infection during 3 successive flu seasons (2010-2013) in 498 patients treated for acute leukemia at St. Jude.
The patients’ median age was 6 years (range, 1-21). Most patients had acute lymphoblastic leukemia (ALL, 94%), though some had acute myeloid leukemia (4.8%) or mixed-lineage leukemia (1.2%).
Most patients (n=354) received flu shots, including 98 patients who received booster doses. The remaining 144 patients were not vaccinated.
The vaccinated patients received the trivalent vaccine, which is designed to protect against 3 flu strains predicted to be in wide circulation during a particular flu season. The vaccine was a fairly good match for circulating flu viruses during the flu seasons included in this analysis.
Demographic characteristic were largely similar between vaccinated and unvaccinated patients. The exceptions were that more vaccinated patients had ALL (95.5% vs 90.3%; P=0.034) and vaccinated patients were more likely to be in a low-intensity phase of cancer therapy (90.7% vs 73.6%, P<0.0001).
Results
There were no significant differences between vaccinated and unvaccinated patients when it came to flu rates or rates of flu-like illnesses.
There were 37 episodes of flu in vaccinated patients and 16 episodes in unvaccinated patients. The rates (per 1000 patient days) were 0.73 and 0.70, respectively (P=0.874).
There were 123 cases of flu-like illnesses in vaccinated patients and 55 cases in unvaccinated patients. The rates were 2.44 and 2.41, respectively (P=0.932).
Likewise, there was no significant difference in the rates of flu or flu-like illnesses between patients who received 1 dose of flu vaccine and those who received 2 doses.
The flu rates were 0.60 and 1.02, respectively (P=0.107). And the rates of flu-like illnesses were 2.42 and 2.73, respectively (P=0.529).
Dr Adderson said additional research is needed to determine if a subset of young leukemia patients may benefit from vaccination.
She added that patients at risk of flu should practice good hand hygiene and avoid crowds during the flu season. Patients may also benefit from “cocooning,” a process that focuses on getting family members, healthcare providers, and others in close contact with at-risk patients vaccinated. ![]()
CHMP recommends new formulation of pegaspargase
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) is recommending marketing authorization for lyophilized pegaspargase (ONCASPAR).
If approved, the product would be used as a component of antineoplastic therapy in patients of all ages who have acute lymphoblastic leukemia (ALL).
The product is a freeze-dried formulation of liquid pegaspargase, which is already approved for the aforementioned indication.
The CHMP’s recommendation regarding lyophilized pegaspargase will be submitted to the European Commission (EC).
The EC typically adheres to the CHMP’s recommendations and delivers its final decision within 67 days of the CHMP’s recommendation.
The EC’s decision will be applicable to the entire European Economic Area—all member states of the European Union plus Iceland, Liechtenstein, and Norway.
The CHMP’s recommendation regarding lyophilized pegaspargase is based on analytical and nonclinical studies, which indicate that lyophilized pegaspargase is comparable to the liquid formulation.
Once reconstituted, lyophilized pegaspargase demonstrates similar pharmacokinetics and pharmacodynamics as liquid pegaspargase.
“Lyophilized ONCASPAR builds on more than a decade of data and research with liquid ONCASPAR, and, with no change in dosing regimen, it offers a 3-times longer shelf life,” said Howard B. Mayer, MD, of Shire, the company that developed lyophilized pegaspargase.
“Prolonging shelf life to 24 months for this critically important therapy facilitates management of product inventory by enabling greater flexibility and longer-term planning. Once approved, with the extended shelf life of lyophilized ONCASPAR, we also hope to improve access to the medicine for ALL patients in countries currently not offering liquid ONCASPAR.”
Lyophilized pegaspargase works in the same way as the liquid formulation. It rapidly depletes serum L-asparagine levels and interferes with protein synthesis, thereby depriving lymphoblasts of asparaginase and resulting in cell death. ![]()
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) is recommending marketing authorization for lyophilized pegaspargase (ONCASPAR).
If approved, the product would be used as a component of antineoplastic therapy in patients of all ages who have acute lymphoblastic leukemia (ALL).
The product is a freeze-dried formulation of liquid pegaspargase, which is already approved for the aforementioned indication.
The CHMP’s recommendation regarding lyophilized pegaspargase will be submitted to the European Commission (EC).
The EC typically adheres to the CHMP’s recommendations and delivers its final decision within 67 days of the CHMP’s recommendation.
The EC’s decision will be applicable to the entire European Economic Area—all member states of the European Union plus Iceland, Liechtenstein, and Norway.
The CHMP’s recommendation regarding lyophilized pegaspargase is based on analytical and nonclinical studies, which indicate that lyophilized pegaspargase is comparable to the liquid formulation.
Once reconstituted, lyophilized pegaspargase demonstrates similar pharmacokinetics and pharmacodynamics as liquid pegaspargase.
“Lyophilized ONCASPAR builds on more than a decade of data and research with liquid ONCASPAR, and, with no change in dosing regimen, it offers a 3-times longer shelf life,” said Howard B. Mayer, MD, of Shire, the company that developed lyophilized pegaspargase.
“Prolonging shelf life to 24 months for this critically important therapy facilitates management of product inventory by enabling greater flexibility and longer-term planning. Once approved, with the extended shelf life of lyophilized ONCASPAR, we also hope to improve access to the medicine for ALL patients in countries currently not offering liquid ONCASPAR.”
Lyophilized pegaspargase works in the same way as the liquid formulation. It rapidly depletes serum L-asparagine levels and interferes with protein synthesis, thereby depriving lymphoblasts of asparaginase and resulting in cell death. ![]()
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) is recommending marketing authorization for lyophilized pegaspargase (ONCASPAR).
If approved, the product would be used as a component of antineoplastic therapy in patients of all ages who have acute lymphoblastic leukemia (ALL).
The product is a freeze-dried formulation of liquid pegaspargase, which is already approved for the aforementioned indication.
The CHMP’s recommendation regarding lyophilized pegaspargase will be submitted to the European Commission (EC).
The EC typically adheres to the CHMP’s recommendations and delivers its final decision within 67 days of the CHMP’s recommendation.
The EC’s decision will be applicable to the entire European Economic Area—all member states of the European Union plus Iceland, Liechtenstein, and Norway.
The CHMP’s recommendation regarding lyophilized pegaspargase is based on analytical and nonclinical studies, which indicate that lyophilized pegaspargase is comparable to the liquid formulation.
Once reconstituted, lyophilized pegaspargase demonstrates similar pharmacokinetics and pharmacodynamics as liquid pegaspargase.
“Lyophilized ONCASPAR builds on more than a decade of data and research with liquid ONCASPAR, and, with no change in dosing regimen, it offers a 3-times longer shelf life,” said Howard B. Mayer, MD, of Shire, the company that developed lyophilized pegaspargase.
“Prolonging shelf life to 24 months for this critically important therapy facilitates management of product inventory by enabling greater flexibility and longer-term planning. Once approved, with the extended shelf life of lyophilized ONCASPAR, we also hope to improve access to the medicine for ALL patients in countries currently not offering liquid ONCASPAR.”
Lyophilized pegaspargase works in the same way as the liquid formulation. It rapidly depletes serum L-asparagine levels and interferes with protein synthesis, thereby depriving lymphoblasts of asparaginase and resulting in cell death. ![]()
Natural selection opportunities tied to cancer rates
Countries with the lowest opportunities for natural selection have higher cancer rates than countries with the highest opportunities for natural selection, according to a study published in Evolutionary Applications.
Researchers said this is because modern medicine is enabling people to survive cancers, and their genetic backgrounds are passing from one generation to the next.
The team said the rate of some cancers has doubled and even quadrupled over the past 100 to 150 years, and human evolution has moved away from “survival of the fittest.”
“Modern medicine has enabled the human species to live much longer than would otherwise be expected in the natural world,” said study author Maciej Henneberg, PhD, DSc, of the University of Adelaide in South Australia.
“Besides the obvious benefits that modern medicine gives, it also brings with it an unexpected side-effect—allowing genetic material to be passed from one generation to the next that predisposes people to have poor health, such as type 1 diabetes or cancer.”
“Because of the quality of our healthcare in western society, we have almost removed natural selection as the ‘janitor of the gene pool.’ Unfortunately, the accumulation of genetic mutations over time and across multiple generations is like a delayed death sentence.”
Country comparison
The researchers studied global cancer data from the World Health Organization as well as other health and socioeconomic data from the United Nations and the World Bank of 173 countries. The team compared the top 10 countries with the highest opportunities for natural selection to the 10 countries with the lowest opportunities for natural selection.
“We looked at countries that offered the greatest opportunity to survive cancer compared with those that didn’t,” said study author Wenpeng You, a PhD student at the University of Adelaide. “This does not only take into account factors such as socioeconomic status, urbanization, and quality of medical services but also low mortality and fertility rates, which are the 2 distinguishing features in the ‘better’ world.”
“Countries with low mortality rates may allow more people with cancer genetic background to reproduce and pass cancer genes/mutations to the next generation. Meanwhile, low fertility rates in these countries may not be able to have diverse biological variations to provide the opportunity for selecting a naturally fit population—for example, people without or with less cancer genetic background. Low mortality rate and low fertility rate in the ‘better’ world may have formed a self-reinforcing cycle which has accumulated cancer genetic background at a greater rate than previously thought.”
Based on the researchers’ analysis, the 20 countries are:
| Lowest opportunities for natural selection | Highest opportunities for natural selection |
| Iceland | Burkina Faso |
| Singapore | Chad |
| Japan | Central African Republic |
| Switzerland | Afghanistan |
| Sweden | Somalia |
| Luxembourg | Sierra Leone |
| Germany | Democratic Republic of the Congo |
| Italy | Guinea-Bissau |
| Cyprus | Burundi |
| Andorra | Cameroon |
Cancer incidence
The researchers found the rates of most cancers were higher in the 10 countries with the lowest opportunities for natural selection. The incidence of all cancers was 2.326 times higher in the low-opportunity countries than the high-opportunity ones.
The increased incidences of hematologic malignancies were as follows:
- Non-Hodgkin lymphoma—2.019 times higher in the low-opportunity countries
- Hodgkin lymphoma—3.314 times higher in the low-opportunity countries
- Leukemia—3.574 times higher in the low-opportunity countries
- Multiple myeloma—4.257 times higher in the low-opportunity countries .
Dr Henneberg said that, having removed natural selection as the “janitor of the gene pool,” our modern society is faced with a controversial issue.
“It may be that the only way humankind can be rid of cancer once and for all is through genetic engineering—to repair our genes and take cancer out of the equation,” he said. ![]()
Countries with the lowest opportunities for natural selection have higher cancer rates than countries with the highest opportunities for natural selection, according to a study published in Evolutionary Applications.
Researchers said this is because modern medicine is enabling people to survive cancers, and their genetic backgrounds are passing from one generation to the next.
The team said the rate of some cancers has doubled and even quadrupled over the past 100 to 150 years, and human evolution has moved away from “survival of the fittest.”
“Modern medicine has enabled the human species to live much longer than would otherwise be expected in the natural world,” said study author Maciej Henneberg, PhD, DSc, of the University of Adelaide in South Australia.
“Besides the obvious benefits that modern medicine gives, it also brings with it an unexpected side-effect—allowing genetic material to be passed from one generation to the next that predisposes people to have poor health, such as type 1 diabetes or cancer.”
“Because of the quality of our healthcare in western society, we have almost removed natural selection as the ‘janitor of the gene pool.’ Unfortunately, the accumulation of genetic mutations over time and across multiple generations is like a delayed death sentence.”
Country comparison
The researchers studied global cancer data from the World Health Organization as well as other health and socioeconomic data from the United Nations and the World Bank of 173 countries. The team compared the top 10 countries with the highest opportunities for natural selection to the 10 countries with the lowest opportunities for natural selection.
“We looked at countries that offered the greatest opportunity to survive cancer compared with those that didn’t,” said study author Wenpeng You, a PhD student at the University of Adelaide. “This does not only take into account factors such as socioeconomic status, urbanization, and quality of medical services but also low mortality and fertility rates, which are the 2 distinguishing features in the ‘better’ world.”
“Countries with low mortality rates may allow more people with cancer genetic background to reproduce and pass cancer genes/mutations to the next generation. Meanwhile, low fertility rates in these countries may not be able to have diverse biological variations to provide the opportunity for selecting a naturally fit population—for example, people without or with less cancer genetic background. Low mortality rate and low fertility rate in the ‘better’ world may have formed a self-reinforcing cycle which has accumulated cancer genetic background at a greater rate than previously thought.”
Based on the researchers’ analysis, the 20 countries are:
| Lowest opportunities for natural selection | Highest opportunities for natural selection |
| Iceland | Burkina Faso |
| Singapore | Chad |
| Japan | Central African Republic |
| Switzerland | Afghanistan |
| Sweden | Somalia |
| Luxembourg | Sierra Leone |
| Germany | Democratic Republic of the Congo |
| Italy | Guinea-Bissau |
| Cyprus | Burundi |
| Andorra | Cameroon |
Cancer incidence
The researchers found the rates of most cancers were higher in the 10 countries with the lowest opportunities for natural selection. The incidence of all cancers was 2.326 times higher in the low-opportunity countries than the high-opportunity ones.
The increased incidences of hematologic malignancies were as follows:
- Non-Hodgkin lymphoma—2.019 times higher in the low-opportunity countries
- Hodgkin lymphoma—3.314 times higher in the low-opportunity countries
- Leukemia—3.574 times higher in the low-opportunity countries
- Multiple myeloma—4.257 times higher in the low-opportunity countries .
Dr Henneberg said that, having removed natural selection as the “janitor of the gene pool,” our modern society is faced with a controversial issue.
“It may be that the only way humankind can be rid of cancer once and for all is through genetic engineering—to repair our genes and take cancer out of the equation,” he said. ![]()
Countries with the lowest opportunities for natural selection have higher cancer rates than countries with the highest opportunities for natural selection, according to a study published in Evolutionary Applications.
Researchers said this is because modern medicine is enabling people to survive cancers, and their genetic backgrounds are passing from one generation to the next.
The team said the rate of some cancers has doubled and even quadrupled over the past 100 to 150 years, and human evolution has moved away from “survival of the fittest.”
“Modern medicine has enabled the human species to live much longer than would otherwise be expected in the natural world,” said study author Maciej Henneberg, PhD, DSc, of the University of Adelaide in South Australia.
“Besides the obvious benefits that modern medicine gives, it also brings with it an unexpected side-effect—allowing genetic material to be passed from one generation to the next that predisposes people to have poor health, such as type 1 diabetes or cancer.”
“Because of the quality of our healthcare in western society, we have almost removed natural selection as the ‘janitor of the gene pool.’ Unfortunately, the accumulation of genetic mutations over time and across multiple generations is like a delayed death sentence.”
Country comparison
The researchers studied global cancer data from the World Health Organization as well as other health and socioeconomic data from the United Nations and the World Bank of 173 countries. The team compared the top 10 countries with the highest opportunities for natural selection to the 10 countries with the lowest opportunities for natural selection.
“We looked at countries that offered the greatest opportunity to survive cancer compared with those that didn’t,” said study author Wenpeng You, a PhD student at the University of Adelaide. “This does not only take into account factors such as socioeconomic status, urbanization, and quality of medical services but also low mortality and fertility rates, which are the 2 distinguishing features in the ‘better’ world.”
“Countries with low mortality rates may allow more people with cancer genetic background to reproduce and pass cancer genes/mutations to the next generation. Meanwhile, low fertility rates in these countries may not be able to have diverse biological variations to provide the opportunity for selecting a naturally fit population—for example, people without or with less cancer genetic background. Low mortality rate and low fertility rate in the ‘better’ world may have formed a self-reinforcing cycle which has accumulated cancer genetic background at a greater rate than previously thought.”
Based on the researchers’ analysis, the 20 countries are:
| Lowest opportunities for natural selection | Highest opportunities for natural selection |
| Iceland | Burkina Faso |
| Singapore | Chad |
| Japan | Central African Republic |
| Switzerland | Afghanistan |
| Sweden | Somalia |
| Luxembourg | Sierra Leone |
| Germany | Democratic Republic of the Congo |
| Italy | Guinea-Bissau |
| Cyprus | Burundi |
| Andorra | Cameroon |
Cancer incidence
The researchers found the rates of most cancers were higher in the 10 countries with the lowest opportunities for natural selection. The incidence of all cancers was 2.326 times higher in the low-opportunity countries than the high-opportunity ones.
The increased incidences of hematologic malignancies were as follows:
- Non-Hodgkin lymphoma—2.019 times higher in the low-opportunity countries
- Hodgkin lymphoma—3.314 times higher in the low-opportunity countries
- Leukemia—3.574 times higher in the low-opportunity countries
- Multiple myeloma—4.257 times higher in the low-opportunity countries .
Dr Henneberg said that, having removed natural selection as the “janitor of the gene pool,” our modern society is faced with a controversial issue.
“It may be that the only way humankind can be rid of cancer once and for all is through genetic engineering—to repair our genes and take cancer out of the equation,” he said. ![]()
Study supports prophylaxis in kids with ALL
Results of an observational study support targeted antibacterial prophylaxis in children undergoing induction therapy for acute lymphoblastic leukemia (ALL).
Prophylaxis effectively prevented febrile neutropenia and systemic infection in the children studied.
Prophylaxis with the drug levofloxacin reduced the use of treatment antibiotics and the incidence of Clostridium difficile infection.
“This research provides the first major evidence supporting targeted use of antibacterial prophylaxis for at-risk pediatric ALL patients, particularly use of the broad-spectrum antibiotic levofloxacin,” said study author Joshua Wolf, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“Prophylactic antibiotic therapy with levofloxacin is routine for at-risk adult ALL patients, but it has remained controversial in children. Until this study, evidence supporting the safety and efficacy of prophylactic antibiotic therapy in children with ALL has been sparse.”
Dr Wolf and his colleagues described their study in Clinical Infectious Diseases.
The study included 344 patients newly diagnosed with ALL who were enrolled in the St. Jude Total XVI clinical trial (NCT00549848). Patients were enrolled from 2007 to 2016.
Until July 2014, the patients received prophylactic antibiotic therapy at the discretion of their physicians. Patients typically received cefepime, ciprofloxacin, or vancomycin plus cefepime or ciprofloxacin. And prophylaxis was typically started at the onset of neutropenia after chemotherapy.
Beginning in August 2014, hospital treatment guidelines changed to recommend prophylactic levofloxacin during induction for ALL patients who develop neutropenia expected to last at least 7 days.
Dr Wolf and his colleagues used the change to compare infection rates and other questions in the following patient groups.
| Patient characteristics | No prophylaxis (n=173) | Levofloxacin prophylaxis (n=69) | Other prophylaxis (n=102) |
| Median age in years (range) | 5.8 (3-11.9) | 6.8 (3.9-11.1) | 7 (3.6-11.9) |
| B-ALL | 83% | 78% | 79% |
| Low-risk ALL | 51% | 54% | 50% |
| Standard-risk ALL | 47% | 41% | 42% |
| High-risk ALL | 2% | 6% | 8% |
| Median duration of neutropenia in days (range) | 17 (11-24) | 18 (12-23) | 20 (17-25) |
| Median duration of profound neutropenia in days (range) | 6 (2-13) | 7 (4-12) | 11 (5-16) |
Results
Researchers reported that patients with neutropenia who received any prophylactic therapy were far less likely than those who did not to develop fever, documented or likely infections, or bloodstream infections.
In a multivariate analysis, the adjusted odds ratios in patients who received prophylaxis, compared to those who did not, were as follows.
- Febrile neutropenia—0.23, P<0.001
- Febrile neutropenia with clinically documented infection—0.30, P=0.002
- Febrile neutropenia with microbiologically documented infection—0.25, P<0.001
- Clinically documented infection—0.54, P=0.02
- Microbiologically documented infection—0.40, P<0.001
- Bloodstream infection—0.30, P=0.008
- C difficile infection—0.38, P=0.04
- Likely bacterial infection—0.26, P<0.001
- Any enterocolitis—0.44, P=0.03.
Analysis also revealed that patients who received levofloxacin had a greater reduction in C difficile infection than patients who received other prophylaxis. The adjusted odds ratio was 0.04 (P<0.001).
However, there was no significant difference between the prophylaxis groups when it came to other infections.
Patients who received levofloxacin prophylaxis had significantly less exposure to other antibiotics than patients who received other prophylaxis or no prophylaxis.
This included exposure to cefepime/ceftazidime (P<0.001 for both comparisons), vancomycin (P<0.001 for both), meropenem (P<0.001 for both), and aminoglycosides (P=0.002 for no prophylaxis, P=0.04 for other prophylaxis).
The reduction in exposure to other antibiotics may partly explain why C difficile infections declined in levofloxacin-treated patients, Dr Wolf said.
He also noted that antibiotic resistance did not significantly increase in this study, despite the greater use of levofloxacin to prevent infections.
“We are cautiously optimistic that any impact of levofloxacin on antibacterial resistance will be balanced by the reduction in use of other antibiotics,” Dr Wolf said, “but long-term monitoring of antibiotic resistance patterns in young ALL patients will be needed to prove this.” ![]()
Results of an observational study support targeted antibacterial prophylaxis in children undergoing induction therapy for acute lymphoblastic leukemia (ALL).
Prophylaxis effectively prevented febrile neutropenia and systemic infection in the children studied.
Prophylaxis with the drug levofloxacin reduced the use of treatment antibiotics and the incidence of Clostridium difficile infection.
“This research provides the first major evidence supporting targeted use of antibacterial prophylaxis for at-risk pediatric ALL patients, particularly use of the broad-spectrum antibiotic levofloxacin,” said study author Joshua Wolf, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“Prophylactic antibiotic therapy with levofloxacin is routine for at-risk adult ALL patients, but it has remained controversial in children. Until this study, evidence supporting the safety and efficacy of prophylactic antibiotic therapy in children with ALL has been sparse.”
Dr Wolf and his colleagues described their study in Clinical Infectious Diseases.
The study included 344 patients newly diagnosed with ALL who were enrolled in the St. Jude Total XVI clinical trial (NCT00549848). Patients were enrolled from 2007 to 2016.
Until July 2014, the patients received prophylactic antibiotic therapy at the discretion of their physicians. Patients typically received cefepime, ciprofloxacin, or vancomycin plus cefepime or ciprofloxacin. And prophylaxis was typically started at the onset of neutropenia after chemotherapy.
Beginning in August 2014, hospital treatment guidelines changed to recommend prophylactic levofloxacin during induction for ALL patients who develop neutropenia expected to last at least 7 days.
Dr Wolf and his colleagues used the change to compare infection rates and other questions in the following patient groups.
| Patient characteristics | No prophylaxis (n=173) | Levofloxacin prophylaxis (n=69) | Other prophylaxis (n=102) |
| Median age in years (range) | 5.8 (3-11.9) | 6.8 (3.9-11.1) | 7 (3.6-11.9) |
| B-ALL | 83% | 78% | 79% |
| Low-risk ALL | 51% | 54% | 50% |
| Standard-risk ALL | 47% | 41% | 42% |
| High-risk ALL | 2% | 6% | 8% |
| Median duration of neutropenia in days (range) | 17 (11-24) | 18 (12-23) | 20 (17-25) |
| Median duration of profound neutropenia in days (range) | 6 (2-13) | 7 (4-12) | 11 (5-16) |
Results
Researchers reported that patients with neutropenia who received any prophylactic therapy were far less likely than those who did not to develop fever, documented or likely infections, or bloodstream infections.
In a multivariate analysis, the adjusted odds ratios in patients who received prophylaxis, compared to those who did not, were as follows.
- Febrile neutropenia—0.23, P<0.001
- Febrile neutropenia with clinically documented infection—0.30, P=0.002
- Febrile neutropenia with microbiologically documented infection—0.25, P<0.001
- Clinically documented infection—0.54, P=0.02
- Microbiologically documented infection—0.40, P<0.001
- Bloodstream infection—0.30, P=0.008
- C difficile infection—0.38, P=0.04
- Likely bacterial infection—0.26, P<0.001
- Any enterocolitis—0.44, P=0.03.
Analysis also revealed that patients who received levofloxacin had a greater reduction in C difficile infection than patients who received other prophylaxis. The adjusted odds ratio was 0.04 (P<0.001).
However, there was no significant difference between the prophylaxis groups when it came to other infections.
Patients who received levofloxacin prophylaxis had significantly less exposure to other antibiotics than patients who received other prophylaxis or no prophylaxis.
This included exposure to cefepime/ceftazidime (P<0.001 for both comparisons), vancomycin (P<0.001 for both), meropenem (P<0.001 for both), and aminoglycosides (P=0.002 for no prophylaxis, P=0.04 for other prophylaxis).
The reduction in exposure to other antibiotics may partly explain why C difficile infections declined in levofloxacin-treated patients, Dr Wolf said.
He also noted that antibiotic resistance did not significantly increase in this study, despite the greater use of levofloxacin to prevent infections.
“We are cautiously optimistic that any impact of levofloxacin on antibacterial resistance will be balanced by the reduction in use of other antibiotics,” Dr Wolf said, “but long-term monitoring of antibiotic resistance patterns in young ALL patients will be needed to prove this.” ![]()
Results of an observational study support targeted antibacterial prophylaxis in children undergoing induction therapy for acute lymphoblastic leukemia (ALL).
Prophylaxis effectively prevented febrile neutropenia and systemic infection in the children studied.
Prophylaxis with the drug levofloxacin reduced the use of treatment antibiotics and the incidence of Clostridium difficile infection.
“This research provides the first major evidence supporting targeted use of antibacterial prophylaxis for at-risk pediatric ALL patients, particularly use of the broad-spectrum antibiotic levofloxacin,” said study author Joshua Wolf, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“Prophylactic antibiotic therapy with levofloxacin is routine for at-risk adult ALL patients, but it has remained controversial in children. Until this study, evidence supporting the safety and efficacy of prophylactic antibiotic therapy in children with ALL has been sparse.”
Dr Wolf and his colleagues described their study in Clinical Infectious Diseases.
The study included 344 patients newly diagnosed with ALL who were enrolled in the St. Jude Total XVI clinical trial (NCT00549848). Patients were enrolled from 2007 to 2016.
Until July 2014, the patients received prophylactic antibiotic therapy at the discretion of their physicians. Patients typically received cefepime, ciprofloxacin, or vancomycin plus cefepime or ciprofloxacin. And prophylaxis was typically started at the onset of neutropenia after chemotherapy.
Beginning in August 2014, hospital treatment guidelines changed to recommend prophylactic levofloxacin during induction for ALL patients who develop neutropenia expected to last at least 7 days.
Dr Wolf and his colleagues used the change to compare infection rates and other questions in the following patient groups.
| Patient characteristics | No prophylaxis (n=173) | Levofloxacin prophylaxis (n=69) | Other prophylaxis (n=102) |
| Median age in years (range) | 5.8 (3-11.9) | 6.8 (3.9-11.1) | 7 (3.6-11.9) |
| B-ALL | 83% | 78% | 79% |
| Low-risk ALL | 51% | 54% | 50% |
| Standard-risk ALL | 47% | 41% | 42% |
| High-risk ALL | 2% | 6% | 8% |
| Median duration of neutropenia in days (range) | 17 (11-24) | 18 (12-23) | 20 (17-25) |
| Median duration of profound neutropenia in days (range) | 6 (2-13) | 7 (4-12) | 11 (5-16) |
Results
Researchers reported that patients with neutropenia who received any prophylactic therapy were far less likely than those who did not to develop fever, documented or likely infections, or bloodstream infections.
In a multivariate analysis, the adjusted odds ratios in patients who received prophylaxis, compared to those who did not, were as follows.
- Febrile neutropenia—0.23, P<0.001
- Febrile neutropenia with clinically documented infection—0.30, P=0.002
- Febrile neutropenia with microbiologically documented infection—0.25, P<0.001
- Clinically documented infection—0.54, P=0.02
- Microbiologically documented infection—0.40, P<0.001
- Bloodstream infection—0.30, P=0.008
- C difficile infection—0.38, P=0.04
- Likely bacterial infection—0.26, P<0.001
- Any enterocolitis—0.44, P=0.03.
Analysis also revealed that patients who received levofloxacin had a greater reduction in C difficile infection than patients who received other prophylaxis. The adjusted odds ratio was 0.04 (P<0.001).
However, there was no significant difference between the prophylaxis groups when it came to other infections.
Patients who received levofloxacin prophylaxis had significantly less exposure to other antibiotics than patients who received other prophylaxis or no prophylaxis.
This included exposure to cefepime/ceftazidime (P<0.001 for both comparisons), vancomycin (P<0.001 for both), meropenem (P<0.001 for both), and aminoglycosides (P=0.002 for no prophylaxis, P=0.04 for other prophylaxis).
The reduction in exposure to other antibiotics may partly explain why C difficile infections declined in levofloxacin-treated patients, Dr Wolf said.
He also noted that antibiotic resistance did not significantly increase in this study, despite the greater use of levofloxacin to prevent infections.
“We are cautiously optimistic that any impact of levofloxacin on antibacterial resistance will be balanced by the reduction in use of other antibiotics,” Dr Wolf said, “but long-term monitoring of antibiotic resistance patterns in young ALL patients will be needed to prove this.”
NCCN completes resource on radiation therapy
The National Comprehensive Cancer Network® (NCCN) has announced the release of the newly completed NCCN Radiation Therapy Compendium™.
This resource includes information designed to support clinical decision-making regarding the use of radiation therapy in cancer patients.
The content is based on the NCCN Clinical Practice Guidelines in Oncology and includes information from the 41 guidelines that reference radiation therapy.
“By compiling every recommendation for radiation therapy in one place, we’ve made it significantly easier for specialists . . . to stay up-to-date on the very latest recommendations, regardless of how many different cancer types they treat,” said Robert W. Carlson, MD, chief executive officer of NCCN.
“This targeted content provides radiation oncologists with the specific, cutting-edge information they need, without forcing them to sift through any extraneous information. It’s part of our ongoing effort to always provide the most pertinent data on emerging treatment practices in the clearest, most efficient way possible.”
The NCCN Radiation Therapy Compendium includes a full complement of radiation therapy recommendations found in the current NCCN guidelines, including specific treatment modalities such as 2D/3D conformal external beam radiation therapy, intensity modulated radiation therapy, intra-operative radiation therapy, stereotactic radiosurgery/stereotactic body radiotherapy/stereotactic ablative body radiotherapy, image-guided radiation therapy, low dose-rate/high dose-rate brachytherapy, radioisotope, and particle therapy.
NCCN first announced the launch of the Radiation Therapy Compendium in March at the NCCN Annual Conference: Improving the Quality, Effectiveness, and Efficiency of Cancer Care.
At the time, the NCCN released a preliminary version of the compendium featuring 24 cancer types. The newly completed version now contains all 41 disease sites that are currently being treated using radiation therapy.
The compendium will be updated on a continual basis in conjunction with the library of clinical guidelines.
For more information and to access the NCCN Radiation Therapy Compendium, visit NCCN.org/RTCompendium. The compendium is available free-of-charge through March 2018.
The National Comprehensive Cancer Network® (NCCN) has announced the release of the newly completed NCCN Radiation Therapy Compendium™.
This resource includes information designed to support clinical decision-making regarding the use of radiation therapy in cancer patients.
The content is based on the NCCN Clinical Practice Guidelines in Oncology and includes information from the 41 guidelines that reference radiation therapy.
“By compiling every recommendation for radiation therapy in one place, we’ve made it significantly easier for specialists . . . to stay up-to-date on the very latest recommendations, regardless of how many different cancer types they treat,” said Robert W. Carlson, MD, chief executive officer of NCCN.
“This targeted content provides radiation oncologists with the specific, cutting-edge information they need, without forcing them to sift through any extraneous information. It’s part of our ongoing effort to always provide the most pertinent data on emerging treatment practices in the clearest, most efficient way possible.”
The NCCN Radiation Therapy Compendium includes a full complement of radiation therapy recommendations found in the current NCCN guidelines, including specific treatment modalities such as 2D/3D conformal external beam radiation therapy, intensity modulated radiation therapy, intra-operative radiation therapy, stereotactic radiosurgery/stereotactic body radiotherapy/stereotactic ablative body radiotherapy, image-guided radiation therapy, low dose-rate/high dose-rate brachytherapy, radioisotope, and particle therapy.
NCCN first announced the launch of the Radiation Therapy Compendium in March at the NCCN Annual Conference: Improving the Quality, Effectiveness, and Efficiency of Cancer Care.
At the time, the NCCN released a preliminary version of the compendium featuring 24 cancer types. The newly completed version now contains all 41 disease sites that are currently being treated using radiation therapy.
The compendium will be updated on a continual basis in conjunction with the library of clinical guidelines.
For more information and to access the NCCN Radiation Therapy Compendium, visit NCCN.org/RTCompendium. The compendium is available free-of-charge through March 2018.
The National Comprehensive Cancer Network® (NCCN) has announced the release of the newly completed NCCN Radiation Therapy Compendium™.
This resource includes information designed to support clinical decision-making regarding the use of radiation therapy in cancer patients.
The content is based on the NCCN Clinical Practice Guidelines in Oncology and includes information from the 41 guidelines that reference radiation therapy.
“By compiling every recommendation for radiation therapy in one place, we’ve made it significantly easier for specialists . . . to stay up-to-date on the very latest recommendations, regardless of how many different cancer types they treat,” said Robert W. Carlson, MD, chief executive officer of NCCN.
“This targeted content provides radiation oncologists with the specific, cutting-edge information they need, without forcing them to sift through any extraneous information. It’s part of our ongoing effort to always provide the most pertinent data on emerging treatment practices in the clearest, most efficient way possible.”
The NCCN Radiation Therapy Compendium includes a full complement of radiation therapy recommendations found in the current NCCN guidelines, including specific treatment modalities such as 2D/3D conformal external beam radiation therapy, intensity modulated radiation therapy, intra-operative radiation therapy, stereotactic radiosurgery/stereotactic body radiotherapy/stereotactic ablative body radiotherapy, image-guided radiation therapy, low dose-rate/high dose-rate brachytherapy, radioisotope, and particle therapy.
NCCN first announced the launch of the Radiation Therapy Compendium in March at the NCCN Annual Conference: Improving the Quality, Effectiveness, and Efficiency of Cancer Care.
At the time, the NCCN released a preliminary version of the compendium featuring 24 cancer types. The newly completed version now contains all 41 disease sites that are currently being treated using radiation therapy.
The compendium will be updated on a continual basis in conjunction with the library of clinical guidelines.
For more information and to access the NCCN Radiation Therapy Compendium, visit NCCN.org/RTCompendium. The compendium is available free-of-charge through March 2018.