User login
The Hospitalist only
By the Numbers: 3,000
Approximate number of million-dollar hospitalization bills in Northern California hospitals in 2010, up from 430 a decade earlier, according to statistics from the Office of Statewide Health Planning and Development.1 More than 20% of the giant bills—typically for complicated, life-saving procedures and lengthy hospital stays following critical illness or trauma—went to the parents of newborn babies with catastrophic illnesses. Organ transplants were also associated with extended hospital stays and million-dollar price tags.
Medical bills are a major factor in two-thirds of personal bankruptcy cases, although most of the largest bills will be lowered significantly, as much as 80%, after negotiations between hospitals and insurers.
Reference
Approximate number of million-dollar hospitalization bills in Northern California hospitals in 2010, up from 430 a decade earlier, according to statistics from the Office of Statewide Health Planning and Development.1 More than 20% of the giant bills—typically for complicated, life-saving procedures and lengthy hospital stays following critical illness or trauma—went to the parents of newborn babies with catastrophic illnesses. Organ transplants were also associated with extended hospital stays and million-dollar price tags.
Medical bills are a major factor in two-thirds of personal bankruptcy cases, although most of the largest bills will be lowered significantly, as much as 80%, after negotiations between hospitals and insurers.
Reference
Approximate number of million-dollar hospitalization bills in Northern California hospitals in 2010, up from 430 a decade earlier, according to statistics from the Office of Statewide Health Planning and Development.1 More than 20% of the giant bills—typically for complicated, life-saving procedures and lengthy hospital stays following critical illness or trauma—went to the parents of newborn babies with catastrophic illnesses. Organ transplants were also associated with extended hospital stays and million-dollar price tags.
Medical bills are a major factor in two-thirds of personal bankruptcy cases, although most of the largest bills will be lowered significantly, as much as 80%, after negotiations between hospitals and insurers.
Reference
C. Diff Deaths at All-Time High
Both incidence and deaths related to the bacterial infection Clostridium difficile have reached an all-time high, according to the federal Centers for Disease Control and Prevention (CDC), which calls C. diff “a formidable opponent” and a widespread safety issue. A total of 14,000 deaths related to C. diff were reported during the period of 2006-2007, compared with 3,000 in 1999-2000. A quarter of infections now appear first in hospitalized patients, with the rest in nursing home residents or patients in doctors’ offices.
L. Clifford McDonald, MD, CDC epidemiologist and lead author of a recent CDC Vital Signs report on the subject, recommended several steps for reducing C. diff infections, starting with better antibiotic stewardship, early detection and isolation for those who test positive, use of gloves and gowns when treating them, and informing the receiving medical team when those patients are transferred.1 Dr. McDonald also noted that state-led hospital collaboratives in Illinois, Massachusetts, and New York have reduced infection rates by 20%.
Another recent example of successful antibiotic stewardship comes from the University of Maryland Medical Center (UMMC) and Health Day News, summarizing a study in the journal Infection Control and Hospital Epidemiology.2
A seven-year stewardship program using an anti-microbial monitoring team generated a $3 million reduction in the hospital’s annual budget for antibiotics by its third year, with no increase in death rates, hospital readmissions, or length of stay. After seven years, antibiotic spending per patient per day had been cut by nearly half. The antibiotic stewardship program was canceled in 2008 in favor of providing more infectious-disease consulting as an alternative mode of stewardship. Over the subsequent two years, antibiotic costs went back up 32%, reports lead author Harold Standiford, MD.
“Our results clearly show that an antimicrobial stewardship program like the one at UMMC is safe, effective, and makes good financial sense,” he says, adding physicians should eliminate wasteful healthcare spending.
References
- Centers for Disease Control and Prevention. Vital signs: preventing Clostridium difficile infections. Centers for Disease Control and Prevention website. Available at: http://www.cdc.gov/mmwr/preview/mmwrhtml/mm6109a3.htm. Accessed March 29, 2012.
- Standiford HC, Chan S, Tripoli M, Weekes E, Forrest GN. Antimicrobial stewardship at a large tertiary care academic medical center: cost analysis before, during, and after a 7-year program. Infect Control Hosp Epidemiol. 2012;33(4);338-345.
Both incidence and deaths related to the bacterial infection Clostridium difficile have reached an all-time high, according to the federal Centers for Disease Control and Prevention (CDC), which calls C. diff “a formidable opponent” and a widespread safety issue. A total of 14,000 deaths related to C. diff were reported during the period of 2006-2007, compared with 3,000 in 1999-2000. A quarter of infections now appear first in hospitalized patients, with the rest in nursing home residents or patients in doctors’ offices.
L. Clifford McDonald, MD, CDC epidemiologist and lead author of a recent CDC Vital Signs report on the subject, recommended several steps for reducing C. diff infections, starting with better antibiotic stewardship, early detection and isolation for those who test positive, use of gloves and gowns when treating them, and informing the receiving medical team when those patients are transferred.1 Dr. McDonald also noted that state-led hospital collaboratives in Illinois, Massachusetts, and New York have reduced infection rates by 20%.
Another recent example of successful antibiotic stewardship comes from the University of Maryland Medical Center (UMMC) and Health Day News, summarizing a study in the journal Infection Control and Hospital Epidemiology.2
A seven-year stewardship program using an anti-microbial monitoring team generated a $3 million reduction in the hospital’s annual budget for antibiotics by its third year, with no increase in death rates, hospital readmissions, or length of stay. After seven years, antibiotic spending per patient per day had been cut by nearly half. The antibiotic stewardship program was canceled in 2008 in favor of providing more infectious-disease consulting as an alternative mode of stewardship. Over the subsequent two years, antibiotic costs went back up 32%, reports lead author Harold Standiford, MD.
“Our results clearly show that an antimicrobial stewardship program like the one at UMMC is safe, effective, and makes good financial sense,” he says, adding physicians should eliminate wasteful healthcare spending.
References
- Centers for Disease Control and Prevention. Vital signs: preventing Clostridium difficile infections. Centers for Disease Control and Prevention website. Available at: http://www.cdc.gov/mmwr/preview/mmwrhtml/mm6109a3.htm. Accessed March 29, 2012.
- Standiford HC, Chan S, Tripoli M, Weekes E, Forrest GN. Antimicrobial stewardship at a large tertiary care academic medical center: cost analysis before, during, and after a 7-year program. Infect Control Hosp Epidemiol. 2012;33(4);338-345.
Both incidence and deaths related to the bacterial infection Clostridium difficile have reached an all-time high, according to the federal Centers for Disease Control and Prevention (CDC), which calls C. diff “a formidable opponent” and a widespread safety issue. A total of 14,000 deaths related to C. diff were reported during the period of 2006-2007, compared with 3,000 in 1999-2000. A quarter of infections now appear first in hospitalized patients, with the rest in nursing home residents or patients in doctors’ offices.
L. Clifford McDonald, MD, CDC epidemiologist and lead author of a recent CDC Vital Signs report on the subject, recommended several steps for reducing C. diff infections, starting with better antibiotic stewardship, early detection and isolation for those who test positive, use of gloves and gowns when treating them, and informing the receiving medical team when those patients are transferred.1 Dr. McDonald also noted that state-led hospital collaboratives in Illinois, Massachusetts, and New York have reduced infection rates by 20%.
Another recent example of successful antibiotic stewardship comes from the University of Maryland Medical Center (UMMC) and Health Day News, summarizing a study in the journal Infection Control and Hospital Epidemiology.2
A seven-year stewardship program using an anti-microbial monitoring team generated a $3 million reduction in the hospital’s annual budget for antibiotics by its third year, with no increase in death rates, hospital readmissions, or length of stay. After seven years, antibiotic spending per patient per day had been cut by nearly half. The antibiotic stewardship program was canceled in 2008 in favor of providing more infectious-disease consulting as an alternative mode of stewardship. Over the subsequent two years, antibiotic costs went back up 32%, reports lead author Harold Standiford, MD.
“Our results clearly show that an antimicrobial stewardship program like the one at UMMC is safe, effective, and makes good financial sense,” he says, adding physicians should eliminate wasteful healthcare spending.
References
- Centers for Disease Control and Prevention. Vital signs: preventing Clostridium difficile infections. Centers for Disease Control and Prevention website. Available at: http://www.cdc.gov/mmwr/preview/mmwrhtml/mm6109a3.htm. Accessed March 29, 2012.
- Standiford HC, Chan S, Tripoli M, Weekes E, Forrest GN. Antimicrobial stewardship at a large tertiary care academic medical center: cost analysis before, during, and after a 7-year program. Infect Control Hosp Epidemiol. 2012;33(4);338-345.
AMA Microsite Offers New Practice-Management Resources
In January, the American Medical Association (AMA) announced a redesigned website for its Practice Management Center (PMC), a resource designed to help physicians manage their practice more effectively. The PMC includes an online library of tools to help physicians streamline their administrative and business practices, with new sections on practice operations, claims revenue cycle management, and health insurer relations.
An estimated 7,000 visitors access the center each month. Physicians can sign up for practice management alerts or join an online community.
One particular resource that might be useful to hospitalists is a sample hospital-physician employment agreement.
In January, the American Medical Association (AMA) announced a redesigned website for its Practice Management Center (PMC), a resource designed to help physicians manage their practice more effectively. The PMC includes an online library of tools to help physicians streamline their administrative and business practices, with new sections on practice operations, claims revenue cycle management, and health insurer relations.
An estimated 7,000 visitors access the center each month. Physicians can sign up for practice management alerts or join an online community.
One particular resource that might be useful to hospitalists is a sample hospital-physician employment agreement.
In January, the American Medical Association (AMA) announced a redesigned website for its Practice Management Center (PMC), a resource designed to help physicians manage their practice more effectively. The PMC includes an online library of tools to help physicians streamline their administrative and business practices, with new sections on practice operations, claims revenue cycle management, and health insurer relations.
An estimated 7,000 visitors access the center each month. Physicians can sign up for practice management alerts or join an online community.
One particular resource that might be useful to hospitalists is a sample hospital-physician employment agreement.
Hospital Quality Reporting Fails to Impact Death Rates
A study in the March issue of Health Affairs concludes that Hospital Compare, the government’s national hospital quality data reporting system, has produced no reductions in 30-day mortality rates for heart attacks and pneumonia, beyond what would be expected due to existing trends and ongoing innovations in care.1 The data were inconclusive on the impact of reporting on heart failure.
The survey covers Medicare claims data from 2000 to 2008. Its authors say it is one of the strongest studies yet, suggesting that Medicare’s public reporting has little or no impact on actual hospital quality—at least according to current metrics. The jury is still out on Medicare’s efforts to improve hospital quality of care with public reporting, with more study needed, according to lead author Andrew Ryan, MD, of Weill Cornell Medical College in New York City.
Reference
A study in the March issue of Health Affairs concludes that Hospital Compare, the government’s national hospital quality data reporting system, has produced no reductions in 30-day mortality rates for heart attacks and pneumonia, beyond what would be expected due to existing trends and ongoing innovations in care.1 The data were inconclusive on the impact of reporting on heart failure.
The survey covers Medicare claims data from 2000 to 2008. Its authors say it is one of the strongest studies yet, suggesting that Medicare’s public reporting has little or no impact on actual hospital quality—at least according to current metrics. The jury is still out on Medicare’s efforts to improve hospital quality of care with public reporting, with more study needed, according to lead author Andrew Ryan, MD, of Weill Cornell Medical College in New York City.
Reference
A study in the March issue of Health Affairs concludes that Hospital Compare, the government’s national hospital quality data reporting system, has produced no reductions in 30-day mortality rates for heart attacks and pneumonia, beyond what would be expected due to existing trends and ongoing innovations in care.1 The data were inconclusive on the impact of reporting on heart failure.
The survey covers Medicare claims data from 2000 to 2008. Its authors say it is one of the strongest studies yet, suggesting that Medicare’s public reporting has little or no impact on actual hospital quality—at least according to current metrics. The jury is still out on Medicare’s efforts to improve hospital quality of care with public reporting, with more study needed, according to lead author Andrew Ryan, MD, of Weill Cornell Medical College in New York City.
Reference
SHM Should Create Task Force to Explore Reimbursement Methodology Change
One of the The Hospitalist newsmagazine’s features over the years is frequent articles educating physicians about Medicare reimbursement. Nothing wrong with that. Reimbursement for “sepsis” is better than for “urosepsis,” and that’s important for us to know.
However, I think it’s about time that we physicians take a stand and try to change some of the ridiculous word games going on in Medicare reimbursement circles. Why do we stand idly by and let some bureaucrat decree that if we write “urosepsis,” Medicare reimburses markedly less, even though we are treating exactly the same thing if we write “sepsis from UTI”? Why are we not fighting the asinine “bullet” system, in which we get substantially “downcoded” if we miss one trivial bullet in our physical exam that has no bearing on our assessment or plan for the patient? Why have we allowed this travesty to pass through unchallenged? Sure, this is the system now and has been, but do we need to continue to accept these inane reimbursement mechanisms?
I propose we develop a team from within SHM, maybe with some help from other physician organizations, to infiltrate, badger, or whatever it takes to advocate change to a reimbursement system that is more intuitive to physicians. The system should reimburse us for the value we add to the patient encounter. For example, how about reimbursement for nonprocedural visits measured not on history/exam bullets but on the complexity of your assessment and plan, including patient/family counseling and/ or care coordination?
Then we can read articles showing what we are doing to change/improve the Medicare reimbursement landscape instead of just learning how to comply with the current system.
Jim Fulmer, MD, site medical director,
Baptist Primary Care Hospitalist System,
Baptist Medical Center— Downtown Campus,
One of the The Hospitalist newsmagazine’s features over the years is frequent articles educating physicians about Medicare reimbursement. Nothing wrong with that. Reimbursement for “sepsis” is better than for “urosepsis,” and that’s important for us to know.
However, I think it’s about time that we physicians take a stand and try to change some of the ridiculous word games going on in Medicare reimbursement circles. Why do we stand idly by and let some bureaucrat decree that if we write “urosepsis,” Medicare reimburses markedly less, even though we are treating exactly the same thing if we write “sepsis from UTI”? Why are we not fighting the asinine “bullet” system, in which we get substantially “downcoded” if we miss one trivial bullet in our physical exam that has no bearing on our assessment or plan for the patient? Why have we allowed this travesty to pass through unchallenged? Sure, this is the system now and has been, but do we need to continue to accept these inane reimbursement mechanisms?
I propose we develop a team from within SHM, maybe with some help from other physician organizations, to infiltrate, badger, or whatever it takes to advocate change to a reimbursement system that is more intuitive to physicians. The system should reimburse us for the value we add to the patient encounter. For example, how about reimbursement for nonprocedural visits measured not on history/exam bullets but on the complexity of your assessment and plan, including patient/family counseling and/ or care coordination?
Then we can read articles showing what we are doing to change/improve the Medicare reimbursement landscape instead of just learning how to comply with the current system.
Jim Fulmer, MD, site medical director,
Baptist Primary Care Hospitalist System,
Baptist Medical Center— Downtown Campus,
One of the The Hospitalist newsmagazine’s features over the years is frequent articles educating physicians about Medicare reimbursement. Nothing wrong with that. Reimbursement for “sepsis” is better than for “urosepsis,” and that’s important for us to know.
However, I think it’s about time that we physicians take a stand and try to change some of the ridiculous word games going on in Medicare reimbursement circles. Why do we stand idly by and let some bureaucrat decree that if we write “urosepsis,” Medicare reimburses markedly less, even though we are treating exactly the same thing if we write “sepsis from UTI”? Why are we not fighting the asinine “bullet” system, in which we get substantially “downcoded” if we miss one trivial bullet in our physical exam that has no bearing on our assessment or plan for the patient? Why have we allowed this travesty to pass through unchallenged? Sure, this is the system now and has been, but do we need to continue to accept these inane reimbursement mechanisms?
I propose we develop a team from within SHM, maybe with some help from other physician organizations, to infiltrate, badger, or whatever it takes to advocate change to a reimbursement system that is more intuitive to physicians. The system should reimburse us for the value we add to the patient encounter. For example, how about reimbursement for nonprocedural visits measured not on history/exam bullets but on the complexity of your assessment and plan, including patient/family counseling and/ or care coordination?
Then we can read articles showing what we are doing to change/improve the Medicare reimbursement landscape instead of just learning how to comply with the current system.
Jim Fulmer, MD, site medical director,
Baptist Primary Care Hospitalist System,
Baptist Medical Center— Downtown Campus,
Do Not Swab Any Superficial Dermatologic Wound Without Sterilizing the Surface
I would broaden the comment in the April 2012 article “10 Things Hospitalists Should Know about Infectious Diseases” regarding culturing decubitus ulcers to: “Do not swab any superficial dermatologic wound without sterilizing the surface and then subsequently expressing a sample from the underlying tissue.” I tell my trainees that, unless they sterilize, they might as well culture their ties and send that instead.
Clifford A. Kaye, MD, assistant professor,
Wayne State University SOM,
associate medical director of quality and hospital utilization,
Henry Ford Hospital, Detroit
I would broaden the comment in the April 2012 article “10 Things Hospitalists Should Know about Infectious Diseases” regarding culturing decubitus ulcers to: “Do not swab any superficial dermatologic wound without sterilizing the surface and then subsequently expressing a sample from the underlying tissue.” I tell my trainees that, unless they sterilize, they might as well culture their ties and send that instead.
Clifford A. Kaye, MD, assistant professor,
Wayne State University SOM,
associate medical director of quality and hospital utilization,
Henry Ford Hospital, Detroit
I would broaden the comment in the April 2012 article “10 Things Hospitalists Should Know about Infectious Diseases” regarding culturing decubitus ulcers to: “Do not swab any superficial dermatologic wound without sterilizing the surface and then subsequently expressing a sample from the underlying tissue.” I tell my trainees that, unless they sterilize, they might as well culture their ties and send that instead.
Clifford A. Kaye, MD, assistant professor,
Wayne State University SOM,
associate medical director of quality and hospital utilization,
Henry Ford Hospital, Detroit
In the Literature: Physician Reviews of HM-Related Research
In This Edition
Literature At A Glance
A guide to this month’s studies
- IDSA/ATS guidelines for community-acquired pneumonia
- Improved asthma with IL-13 antibody
- Rivaroxaban vs. warfarin for stroke prevention in atrial fibrillation
- Apixaban vs. warfarin for stroke prevention in atrial fibrillation
- Ultrasonography more sensitive than chest radiograph for pneumothorax
- Current readmission risk models inadequate
- Optimal fluid volume for acute pancreatitis
- Low mortality in saddle pulmonary embolism
Triage Decisions for Patients with Severe Community-Acquired Pneumonia Should Be Based on IDSA/ATS Guidelines, Not Inflammatory Biomarkers
Clinical question: Can C-reactive protein levels (CRP), procalcitonin, TNF-alpha, and cytokine levels predict the need for intensive-care admission more accurately than IDSA/ATS guidelines in patients with severe community-acquired pneumonia (CAP)?
Background: Inflammatory biomarkers, such as CRP and procalcitonin, have diagnostic and prognostic utility in patients with CAP. Whether these inflammatory biomarkers can help triage patients to the appropriate level of care is unknown.
Study design: Prospective case control study.
Setting: Two university hospitals in Spain.
Synopsis: The study included 685 patients with severe CAP who did not require mechanical ventilation or vasopressor support. Serum levels of CRP, procalcitonin, TNF-alpha, IL-1, IL-6, IL-8, and IL-10, as well as Infectious Diseases Society of American/American Thoracic Society (IDSA/ATS) minor severity criteria data, were collected on admission. After controlling for age, comorbidities, and PSI risk class, serum levels of CRP and procalcitonin were found to be significantly higher in ICU patients compared with non-ICU patients. Despite this, these inflammatory biomarkers did not augment the IDSA/ATS guidelines, suggesting that patients who have three or more minor criteria be considered for ICU admission.
The study did suggest that patients with severe CAP and low levels of IL-6 and procalcitonin could potentially be managed safely outside of the ICU. However, hospitalists should be wary of applying the study results due to the small number of ICU patients in this study and the lack of real-time availability of these biomarkers at most institutions.
Bottom line: More studies of inflammatory biomarkers are needed before using them to determine the level of care required for patients with CAP. Until these data are available, physicians should use the IDSA/ATS guidelines to triage patients to the appropriate level of care.
Citation: Ramirez P, Ferrer M, Torres A, et al. Inflammatory biomarkers and prediction for intensive care unit admission pneumonia. Crit Care Med. 2011;39:2211-2217.
IL-13 Antibody Lebrikizumab Shows Promise as a New Therapy for Adults with Uncontrolled Asthma
Clinical question: Can lebrikizumab, an IL-13 antibody, improve asthma control in patients with uncontrolled asthma?
Background: Asthma is a complex disease, with varied patient response to treatment. Some patients have uncontrolled asthma despite inhaled glucocorticoids. It is postulated that IL-13 may account for this variability and that some patients with uncontrolled asthma are poorly controlled due to glucocorticoid resistance mediated by IL-13. Lebrikizumab is an IgG4 monoclonal antibody that binds to and inhibits the function of IL-13. This study was performed to see if this antibody would be effective in patients with uncontrolled asthma despite inhaled glucocorticoid therapy.
Study design: Randomized double-blinded placebo-controlled trial.
Setting: Multiple centers.
Synopsis: The study randomized 219 adult asthma patients who were inadequately controlled despite inhaled corticosteroids to a placebo or lebrikizumab. The primary outcome was improvement in prebronchodilator FEV1 from baseline. Secondary outcomes were exacerbations, use of rescue medications, and symptom scores. Patients were also stratified and analyzed based on surrogate markers for IL-13, which included serum IGE levels, eosinophil counts, and periostin levels.
In patients who were randomized to the lebrikizumab treatment, there was a statistically significant improvement in FEV1 of 5.5%, which occurred almost immediately and was sustained for the entire 32 weeks of the study. The improvement was more significant in patients who had high surrogate markers for IL-13. Despite this improvement in FEV1, there were no differences in secondary outcomes except in patients who had surrogate markers for high IL-13 levels.
Bottom line: In adults with asthma who remained uncontrolled despite inhaled corticosteroid therapy, IL-13 antagonism with lebrikizumab improved FEV1. However, the clinical relevance of these modest improvements remains unclear.
Citation: Corren J, Lemanske R, Matthews J, et al. Lebrikizumab treatment in adults with asthma. N Engl J Med. 2011;365:1088-1098.
Rivaroxaban Is Noninferior to Warfarin for Stroke Prevention in Atrial Fibrillation
Clinical question: How does rivaroxaban compare with warfarin in the prevention of stroke or systemic embolism in patients with nonvalvular atrial fibrillation?
Background: Warfarin is effective for the prevention of stroke in atrial fibrillation, but it requires close monitoring and adjustment. Rivaroxaban, an oral Xa inhibitor, may be safer, easier, and more effective than warfarin.
Study design: Multicenter, randomized, double-blind, double-dummy trial.
Setting: 1,178 sites in 45 countries.
Synopsis: The study included 14,264 patients with nonvalvular atrial fibrillation who were randomized to either fixed-dose rivaroxaban (20 mg daily or 15 mg daily for CrCl 30-49 mL/min) plus placebo or adjusted-dose warfarin (target INR 2.0 to 3.0) plus placebo. The mean CHADS2 score was 3.5. The primary endpoint (stroke or systemic embolism) occurred in 1.7% of patients per year in the rivaroxaban group and 2.2% per year in the warfarin group (hazard ratio for rivaroxaban 0.79; 95% CI: 0.66 to 0.96, P<0.001 for noninferiority). There was no difference in major or nonmajor clinically significant bleeding between the two groups (14.9% rivaroxaban vs. 14.5% warfarin, hazard ratio=1.03, 95% CI: 0.96 to 1.11, P=0.44). There were fewer intracranial hemorrhages (0.5% vs. 0.7%, P=0.02) and fatal bleeding (0.2% vs. 0.5%, P=0.003) in the rivaroxaban group.
Bottom line: In patients with atrial fibrillation, rivaroxaban was noninferior to warfarin for the prevention of stroke or systemic embolization, with a similar risk of major bleeding and a lower risk of intracranial hemorrhage or fatal bleeding.
Citation: Patel MR, Mahaffey K, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365:883-891.
Apixaban More Effective and Safer than Warfarin for Stroke Prevention in Atrial Fibrillation
Clinical question: How does the effectiveness and safety of apixaban compare with warfarin for stroke prevention in atrial fibrillation?
Background: Until recently, warfarin has been the only available oral anticoagulant for stroke prevention in patients with atrial fibrillation (AF). The oral factor Xa inhibitors have shown similar efficacy and safety, without the monitoring requirement and drug interactions associated with warfarin.
Study design: Prospective randomized double-blind controlled trial.
Setting: More than 1,000 clinical sites in 39 countries.
Synopsis: This study randomized 18,201 patients with atrial fibrillation or flutter and at least one CHADS2 risk factor for stroke to receive oral apixaban or warfarin therapy. Exclusion criteria were prosthetic valves and severe kidney disease. The median duration of follow-up was 1.8 years, and the major endpoints were incidence of stroke, systemic embolism, bleeding complications, and mortality.
Compared with warfarin, apixaban reduced the annual incidence of stroke and systemic embolism from 1.6% to 1.3% (HR 0.79, 95%: CI 0.66 to 0.95, P=0.01 for superiority), and reduced mortality (HR: 0.89, 95% CI: 0.80 to 0.998). For the combined endpoint of stroke, systemic embolism, MI, or death, the annual rate was reduced from 5.5% to 4.9% (HR: 0.88, 95% CI: 0.80 to 0.97). All measures of bleeding were less frequent with apixaban: major 2.1% vs. 3.1% (HR: 0.69, 95% CI: 0.60 to 0.80), and combined major and minor bleeding 4.1% vs. 6.0% (HR: 0.68, 95% CI: 0.61 to 0.75). The annual rate for the net outcome of stroke, embolism, or major bleeding was 3.2% with apixaban and 4.1% with warfarin (HR: 0.77, 95% CI: 0.69 to 0.86).
Bottom line: Compared with warfarin therapy, apixaban is more effective and safer for stroke prevention in patients with atrial fibrillation.
Citation: Granger CB, Alexander JH, McMurray JJ, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011;365:981-992.
Ultrasonography Is Useful in Diagnosis of Pneumothorax
Clinical question: Is transthoracic ultrasonography a useful tool to diagnose pneumothorax?
Background: CT is the diagnostic gold standard for pneumothorax, but it is associated with radiation exposure and requires patient transport. Chest radiograph is easy to perform but may be too insensitive for adequate diagnosis. Ultrasonography’s diagnostic performance for detecting pneumothorax needs further evaluation.
Study design: Systematic review and meta-analysis.
Setting: Critically ill, trauma, or post-biopsy patients were identified in each of the studies.
Synopsis: The meta-analysis of 20 eligible studies found a pooled sensitivity of ultrasound for the detection of pneumothorax of 0.88 (95% CI: 0.85 to 0.91) and specificity of 0.99 (0.98 to 0.99) compared with sensitivity of 0.52 (0.49 to 0.55) and specificity of 1.00 (1.00 to 1.00) for chest radiograph. Although the overall ROC curve was not significantly different between these modalities, the accuracy of ultrasonography was highly dependent on the skill of the operator.
Bottom line: When performed by a skilled operator, transthoracic ultrasonography is as specific, and more sensitive, than chest radiograph in diagnosing pneumothorax.
Citation: Ding W, Shen Y, Yang J, He X, Zhang M. Diagnosis of pneumothorax by radiography and ultrasonography: a meta-analysis. Chest. 2011;140:859-866.
Risk Prediction for Hospital Readmission Remains Challenging
Clinical question: Can readmission risk assessment be used to identify which patients would benefit most from care-transition interventions, or to risk-adjust readmission rates for hospital comparison?
Background: Multiple models to predict hospital readmission have been described and validated. Identifying patients at high risk for readmission could allow for customized care-transition interventions, or could be used to risk-adjust readmission rates to compare publicly reported rates by hospital.
Study design: Systematic review with qualitative synthesis of results.
Setting: Thirty studies (23 from the U.S.) tested 26 unique readmission models.
Synopsis: Each model had been tested in both a derivation and validation cohort. Fourteen models (nine from the U.S.), using retrospective administrative data to compare risk-adjusted rates between hospitals, had poor discriminative capacity (c statistic range: 0.55 to 0.65). Seven models could be used to identify high-risk patients early in the hospitalization (c statistic range: 0.56 to 0.72) and five could be used to identify high-risk patients at discharge (c statistic range: 0.68 to 0.83), but these also had poor to moderate discriminative capacity. Multiple variables were considered in each of the models; most incorporated medical comorbidities and prior use of healthcare services.
Bottom line: Current readmission risk prediction models do not perform adequately for comparative or clinical purposes.
Citation: Kansagara D, Englander H, Salanitro A, et. al. Risk prediction models for hospital readmission: a systematic review. JAMA. 2011;306:1688-1698.
Intravenous Fluids for Acute Pancreatitis: More May Be Less
Clinical question: What is the optimal volume of fluid administration for treatment of acute pancreatitis?
Background: Current guidelines for management of acute pancreatitis emphasize vigorous administration of intravenous fluids to reduce the risk of pancreatic necrosis and organ failure. This recommendation is based upon animal studies, and has not been subjected to clinical evaluation in humans.
Study design: Prospective observational cohort.
Setting: University-affiliated tertiary-care public hospital in Spain.
Synopsis: This study enrolled 247 patients admitted with acute pancreatitis to determine the association between the volume of fluid administered during the first 24 hours and the development of persistent organ failure, pancreatic fluid collection or necrosis, and mortality. The volume and rate of fluid administered were determined by the treating physician. Patients were classified into three groups: those receiving a volume <3.1 L, 3.1 to 4.1 L, or >4.1 L.
After multivariate adjustment, those receiving <3.1 L had no increased risk of necrosis or any other adverse outcome, compared with those who received the middle range of fluid volume.
Patients receiving >4.1 L had a higher risk of persistent organ failure (OR: 7.7, 95% CI: 1.5 to 38.7), particularly renal and respiratory insufficiency, and fluid collection development (OR: 1.9, 95% CI: 1 to 3.7) independent of disease severity. Pancreatic necrosis and mortality were similar in the three groups.
Bottom line: Administration of large-volume intravenous fluids (>4.1 L) in
the first 24 hours was associated with worse outcomes, although residual confounding cannot be excluded in this nonrandomized study.
Citation: de-Madaria E, Soler-Sala G, Sanchez-Paya J, et al. Influence of fluid therapy on the prognosis of acute pancreatitis: a prospective cohort study. Am J Gastroenterol. 2011;106:1843-1850.
Clinical Outcomes in Saddle Pulmonary Embolism
Clinical question: What are the treatments used and outcomes associated with saddle pulmonary embolism?
Background: Saddle pulmonary embolism is a risk for right ventricular dysfunction and sudden hemodynamic collapse. There are limited data on the clinical presentation and outcomes in these patients.
Study design: Retrospective case review.
Setting: Single academic medical center.
Synopsis: In this retrospective review of 680 patients diagnosed with pulmonary embolism on CT at a single academic medical center from 2004 to 2009, 5.4% (37 patients) had a saddle pulmonary embolism.
Most patients with saddle pulmonary embolism were hemodynamically stable and responded to standard therapy with unfractionated heparin. The mean length of stay was nine days, 46% received an inferior vena cava filter, 41% were treated in an ICU, and 5.4% (two patients) died in the hospital. Thrombolytics were used in only 11% of patients, most of which had sustained hypotension and/or were mechanically ventilated.
Bottom line: Most patients with saddle pulmonary embolus in this single institution study did not receive thrombolytics and had overall low mortality.
Citation: Sardi A, Gluskin J, Guttentag A, Kotler MN, Braitman LE, Lippmann M. Saddle pulmonary embolism: is it as bad as it looks? A community hospital experience. Crit Care Med. 2011;39:2413-2418.
In This Edition
Literature At A Glance
A guide to this month’s studies
- IDSA/ATS guidelines for community-acquired pneumonia
- Improved asthma with IL-13 antibody
- Rivaroxaban vs. warfarin for stroke prevention in atrial fibrillation
- Apixaban vs. warfarin for stroke prevention in atrial fibrillation
- Ultrasonography more sensitive than chest radiograph for pneumothorax
- Current readmission risk models inadequate
- Optimal fluid volume for acute pancreatitis
- Low mortality in saddle pulmonary embolism
Triage Decisions for Patients with Severe Community-Acquired Pneumonia Should Be Based on IDSA/ATS Guidelines, Not Inflammatory Biomarkers
Clinical question: Can C-reactive protein levels (CRP), procalcitonin, TNF-alpha, and cytokine levels predict the need for intensive-care admission more accurately than IDSA/ATS guidelines in patients with severe community-acquired pneumonia (CAP)?
Background: Inflammatory biomarkers, such as CRP and procalcitonin, have diagnostic and prognostic utility in patients with CAP. Whether these inflammatory biomarkers can help triage patients to the appropriate level of care is unknown.
Study design: Prospective case control study.
Setting: Two university hospitals in Spain.
Synopsis: The study included 685 patients with severe CAP who did not require mechanical ventilation or vasopressor support. Serum levels of CRP, procalcitonin, TNF-alpha, IL-1, IL-6, IL-8, and IL-10, as well as Infectious Diseases Society of American/American Thoracic Society (IDSA/ATS) minor severity criteria data, were collected on admission. After controlling for age, comorbidities, and PSI risk class, serum levels of CRP and procalcitonin were found to be significantly higher in ICU patients compared with non-ICU patients. Despite this, these inflammatory biomarkers did not augment the IDSA/ATS guidelines, suggesting that patients who have three or more minor criteria be considered for ICU admission.
The study did suggest that patients with severe CAP and low levels of IL-6 and procalcitonin could potentially be managed safely outside of the ICU. However, hospitalists should be wary of applying the study results due to the small number of ICU patients in this study and the lack of real-time availability of these biomarkers at most institutions.
Bottom line: More studies of inflammatory biomarkers are needed before using them to determine the level of care required for patients with CAP. Until these data are available, physicians should use the IDSA/ATS guidelines to triage patients to the appropriate level of care.
Citation: Ramirez P, Ferrer M, Torres A, et al. Inflammatory biomarkers and prediction for intensive care unit admission pneumonia. Crit Care Med. 2011;39:2211-2217.
IL-13 Antibody Lebrikizumab Shows Promise as a New Therapy for Adults with Uncontrolled Asthma
Clinical question: Can lebrikizumab, an IL-13 antibody, improve asthma control in patients with uncontrolled asthma?
Background: Asthma is a complex disease, with varied patient response to treatment. Some patients have uncontrolled asthma despite inhaled glucocorticoids. It is postulated that IL-13 may account for this variability and that some patients with uncontrolled asthma are poorly controlled due to glucocorticoid resistance mediated by IL-13. Lebrikizumab is an IgG4 monoclonal antibody that binds to and inhibits the function of IL-13. This study was performed to see if this antibody would be effective in patients with uncontrolled asthma despite inhaled glucocorticoid therapy.
Study design: Randomized double-blinded placebo-controlled trial.
Setting: Multiple centers.
Synopsis: The study randomized 219 adult asthma patients who were inadequately controlled despite inhaled corticosteroids to a placebo or lebrikizumab. The primary outcome was improvement in prebronchodilator FEV1 from baseline. Secondary outcomes were exacerbations, use of rescue medications, and symptom scores. Patients were also stratified and analyzed based on surrogate markers for IL-13, which included serum IGE levels, eosinophil counts, and periostin levels.
In patients who were randomized to the lebrikizumab treatment, there was a statistically significant improvement in FEV1 of 5.5%, which occurred almost immediately and was sustained for the entire 32 weeks of the study. The improvement was more significant in patients who had high surrogate markers for IL-13. Despite this improvement in FEV1, there were no differences in secondary outcomes except in patients who had surrogate markers for high IL-13 levels.
Bottom line: In adults with asthma who remained uncontrolled despite inhaled corticosteroid therapy, IL-13 antagonism with lebrikizumab improved FEV1. However, the clinical relevance of these modest improvements remains unclear.
Citation: Corren J, Lemanske R, Matthews J, et al. Lebrikizumab treatment in adults with asthma. N Engl J Med. 2011;365:1088-1098.
Rivaroxaban Is Noninferior to Warfarin for Stroke Prevention in Atrial Fibrillation
Clinical question: How does rivaroxaban compare with warfarin in the prevention of stroke or systemic embolism in patients with nonvalvular atrial fibrillation?
Background: Warfarin is effective for the prevention of stroke in atrial fibrillation, but it requires close monitoring and adjustment. Rivaroxaban, an oral Xa inhibitor, may be safer, easier, and more effective than warfarin.
Study design: Multicenter, randomized, double-blind, double-dummy trial.
Setting: 1,178 sites in 45 countries.
Synopsis: The study included 14,264 patients with nonvalvular atrial fibrillation who were randomized to either fixed-dose rivaroxaban (20 mg daily or 15 mg daily for CrCl 30-49 mL/min) plus placebo or adjusted-dose warfarin (target INR 2.0 to 3.0) plus placebo. The mean CHADS2 score was 3.5. The primary endpoint (stroke or systemic embolism) occurred in 1.7% of patients per year in the rivaroxaban group and 2.2% per year in the warfarin group (hazard ratio for rivaroxaban 0.79; 95% CI: 0.66 to 0.96, P<0.001 for noninferiority). There was no difference in major or nonmajor clinically significant bleeding between the two groups (14.9% rivaroxaban vs. 14.5% warfarin, hazard ratio=1.03, 95% CI: 0.96 to 1.11, P=0.44). There were fewer intracranial hemorrhages (0.5% vs. 0.7%, P=0.02) and fatal bleeding (0.2% vs. 0.5%, P=0.003) in the rivaroxaban group.
Bottom line: In patients with atrial fibrillation, rivaroxaban was noninferior to warfarin for the prevention of stroke or systemic embolization, with a similar risk of major bleeding and a lower risk of intracranial hemorrhage or fatal bleeding.
Citation: Patel MR, Mahaffey K, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365:883-891.
Apixaban More Effective and Safer than Warfarin for Stroke Prevention in Atrial Fibrillation
Clinical question: How does the effectiveness and safety of apixaban compare with warfarin for stroke prevention in atrial fibrillation?
Background: Until recently, warfarin has been the only available oral anticoagulant for stroke prevention in patients with atrial fibrillation (AF). The oral factor Xa inhibitors have shown similar efficacy and safety, without the monitoring requirement and drug interactions associated with warfarin.
Study design: Prospective randomized double-blind controlled trial.
Setting: More than 1,000 clinical sites in 39 countries.
Synopsis: This study randomized 18,201 patients with atrial fibrillation or flutter and at least one CHADS2 risk factor for stroke to receive oral apixaban or warfarin therapy. Exclusion criteria were prosthetic valves and severe kidney disease. The median duration of follow-up was 1.8 years, and the major endpoints were incidence of stroke, systemic embolism, bleeding complications, and mortality.
Compared with warfarin, apixaban reduced the annual incidence of stroke and systemic embolism from 1.6% to 1.3% (HR 0.79, 95%: CI 0.66 to 0.95, P=0.01 for superiority), and reduced mortality (HR: 0.89, 95% CI: 0.80 to 0.998). For the combined endpoint of stroke, systemic embolism, MI, or death, the annual rate was reduced from 5.5% to 4.9% (HR: 0.88, 95% CI: 0.80 to 0.97). All measures of bleeding were less frequent with apixaban: major 2.1% vs. 3.1% (HR: 0.69, 95% CI: 0.60 to 0.80), and combined major and minor bleeding 4.1% vs. 6.0% (HR: 0.68, 95% CI: 0.61 to 0.75). The annual rate for the net outcome of stroke, embolism, or major bleeding was 3.2% with apixaban and 4.1% with warfarin (HR: 0.77, 95% CI: 0.69 to 0.86).
Bottom line: Compared with warfarin therapy, apixaban is more effective and safer for stroke prevention in patients with atrial fibrillation.
Citation: Granger CB, Alexander JH, McMurray JJ, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011;365:981-992.
Ultrasonography Is Useful in Diagnosis of Pneumothorax
Clinical question: Is transthoracic ultrasonography a useful tool to diagnose pneumothorax?
Background: CT is the diagnostic gold standard for pneumothorax, but it is associated with radiation exposure and requires patient transport. Chest radiograph is easy to perform but may be too insensitive for adequate diagnosis. Ultrasonography’s diagnostic performance for detecting pneumothorax needs further evaluation.
Study design: Systematic review and meta-analysis.
Setting: Critically ill, trauma, or post-biopsy patients were identified in each of the studies.
Synopsis: The meta-analysis of 20 eligible studies found a pooled sensitivity of ultrasound for the detection of pneumothorax of 0.88 (95% CI: 0.85 to 0.91) and specificity of 0.99 (0.98 to 0.99) compared with sensitivity of 0.52 (0.49 to 0.55) and specificity of 1.00 (1.00 to 1.00) for chest radiograph. Although the overall ROC curve was not significantly different between these modalities, the accuracy of ultrasonography was highly dependent on the skill of the operator.
Bottom line: When performed by a skilled operator, transthoracic ultrasonography is as specific, and more sensitive, than chest radiograph in diagnosing pneumothorax.
Citation: Ding W, Shen Y, Yang J, He X, Zhang M. Diagnosis of pneumothorax by radiography and ultrasonography: a meta-analysis. Chest. 2011;140:859-866.
Risk Prediction for Hospital Readmission Remains Challenging
Clinical question: Can readmission risk assessment be used to identify which patients would benefit most from care-transition interventions, or to risk-adjust readmission rates for hospital comparison?
Background: Multiple models to predict hospital readmission have been described and validated. Identifying patients at high risk for readmission could allow for customized care-transition interventions, or could be used to risk-adjust readmission rates to compare publicly reported rates by hospital.
Study design: Systematic review with qualitative synthesis of results.
Setting: Thirty studies (23 from the U.S.) tested 26 unique readmission models.
Synopsis: Each model had been tested in both a derivation and validation cohort. Fourteen models (nine from the U.S.), using retrospective administrative data to compare risk-adjusted rates between hospitals, had poor discriminative capacity (c statistic range: 0.55 to 0.65). Seven models could be used to identify high-risk patients early in the hospitalization (c statistic range: 0.56 to 0.72) and five could be used to identify high-risk patients at discharge (c statistic range: 0.68 to 0.83), but these also had poor to moderate discriminative capacity. Multiple variables were considered in each of the models; most incorporated medical comorbidities and prior use of healthcare services.
Bottom line: Current readmission risk prediction models do not perform adequately for comparative or clinical purposes.
Citation: Kansagara D, Englander H, Salanitro A, et. al. Risk prediction models for hospital readmission: a systematic review. JAMA. 2011;306:1688-1698.
Intravenous Fluids for Acute Pancreatitis: More May Be Less
Clinical question: What is the optimal volume of fluid administration for treatment of acute pancreatitis?
Background: Current guidelines for management of acute pancreatitis emphasize vigorous administration of intravenous fluids to reduce the risk of pancreatic necrosis and organ failure. This recommendation is based upon animal studies, and has not been subjected to clinical evaluation in humans.
Study design: Prospective observational cohort.
Setting: University-affiliated tertiary-care public hospital in Spain.
Synopsis: This study enrolled 247 patients admitted with acute pancreatitis to determine the association between the volume of fluid administered during the first 24 hours and the development of persistent organ failure, pancreatic fluid collection or necrosis, and mortality. The volume and rate of fluid administered were determined by the treating physician. Patients were classified into three groups: those receiving a volume <3.1 L, 3.1 to 4.1 L, or >4.1 L.
After multivariate adjustment, those receiving <3.1 L had no increased risk of necrosis or any other adverse outcome, compared with those who received the middle range of fluid volume.
Patients receiving >4.1 L had a higher risk of persistent organ failure (OR: 7.7, 95% CI: 1.5 to 38.7), particularly renal and respiratory insufficiency, and fluid collection development (OR: 1.9, 95% CI: 1 to 3.7) independent of disease severity. Pancreatic necrosis and mortality were similar in the three groups.
Bottom line: Administration of large-volume intravenous fluids (>4.1 L) in
the first 24 hours was associated with worse outcomes, although residual confounding cannot be excluded in this nonrandomized study.
Citation: de-Madaria E, Soler-Sala G, Sanchez-Paya J, et al. Influence of fluid therapy on the prognosis of acute pancreatitis: a prospective cohort study. Am J Gastroenterol. 2011;106:1843-1850.
Clinical Outcomes in Saddle Pulmonary Embolism
Clinical question: What are the treatments used and outcomes associated with saddle pulmonary embolism?
Background: Saddle pulmonary embolism is a risk for right ventricular dysfunction and sudden hemodynamic collapse. There are limited data on the clinical presentation and outcomes in these patients.
Study design: Retrospective case review.
Setting: Single academic medical center.
Synopsis: In this retrospective review of 680 patients diagnosed with pulmonary embolism on CT at a single academic medical center from 2004 to 2009, 5.4% (37 patients) had a saddle pulmonary embolism.
Most patients with saddle pulmonary embolism were hemodynamically stable and responded to standard therapy with unfractionated heparin. The mean length of stay was nine days, 46% received an inferior vena cava filter, 41% were treated in an ICU, and 5.4% (two patients) died in the hospital. Thrombolytics were used in only 11% of patients, most of which had sustained hypotension and/or were mechanically ventilated.
Bottom line: Most patients with saddle pulmonary embolus in this single institution study did not receive thrombolytics and had overall low mortality.
Citation: Sardi A, Gluskin J, Guttentag A, Kotler MN, Braitman LE, Lippmann M. Saddle pulmonary embolism: is it as bad as it looks? A community hospital experience. Crit Care Med. 2011;39:2413-2418.
In This Edition
Literature At A Glance
A guide to this month’s studies
- IDSA/ATS guidelines for community-acquired pneumonia
- Improved asthma with IL-13 antibody
- Rivaroxaban vs. warfarin for stroke prevention in atrial fibrillation
- Apixaban vs. warfarin for stroke prevention in atrial fibrillation
- Ultrasonography more sensitive than chest radiograph for pneumothorax
- Current readmission risk models inadequate
- Optimal fluid volume for acute pancreatitis
- Low mortality in saddle pulmonary embolism
Triage Decisions for Patients with Severe Community-Acquired Pneumonia Should Be Based on IDSA/ATS Guidelines, Not Inflammatory Biomarkers
Clinical question: Can C-reactive protein levels (CRP), procalcitonin, TNF-alpha, and cytokine levels predict the need for intensive-care admission more accurately than IDSA/ATS guidelines in patients with severe community-acquired pneumonia (CAP)?
Background: Inflammatory biomarkers, such as CRP and procalcitonin, have diagnostic and prognostic utility in patients with CAP. Whether these inflammatory biomarkers can help triage patients to the appropriate level of care is unknown.
Study design: Prospective case control study.
Setting: Two university hospitals in Spain.
Synopsis: The study included 685 patients with severe CAP who did not require mechanical ventilation or vasopressor support. Serum levels of CRP, procalcitonin, TNF-alpha, IL-1, IL-6, IL-8, and IL-10, as well as Infectious Diseases Society of American/American Thoracic Society (IDSA/ATS) minor severity criteria data, were collected on admission. After controlling for age, comorbidities, and PSI risk class, serum levels of CRP and procalcitonin were found to be significantly higher in ICU patients compared with non-ICU patients. Despite this, these inflammatory biomarkers did not augment the IDSA/ATS guidelines, suggesting that patients who have three or more minor criteria be considered for ICU admission.
The study did suggest that patients with severe CAP and low levels of IL-6 and procalcitonin could potentially be managed safely outside of the ICU. However, hospitalists should be wary of applying the study results due to the small number of ICU patients in this study and the lack of real-time availability of these biomarkers at most institutions.
Bottom line: More studies of inflammatory biomarkers are needed before using them to determine the level of care required for patients with CAP. Until these data are available, physicians should use the IDSA/ATS guidelines to triage patients to the appropriate level of care.
Citation: Ramirez P, Ferrer M, Torres A, et al. Inflammatory biomarkers and prediction for intensive care unit admission pneumonia. Crit Care Med. 2011;39:2211-2217.
IL-13 Antibody Lebrikizumab Shows Promise as a New Therapy for Adults with Uncontrolled Asthma
Clinical question: Can lebrikizumab, an IL-13 antibody, improve asthma control in patients with uncontrolled asthma?
Background: Asthma is a complex disease, with varied patient response to treatment. Some patients have uncontrolled asthma despite inhaled glucocorticoids. It is postulated that IL-13 may account for this variability and that some patients with uncontrolled asthma are poorly controlled due to glucocorticoid resistance mediated by IL-13. Lebrikizumab is an IgG4 monoclonal antibody that binds to and inhibits the function of IL-13. This study was performed to see if this antibody would be effective in patients with uncontrolled asthma despite inhaled glucocorticoid therapy.
Study design: Randomized double-blinded placebo-controlled trial.
Setting: Multiple centers.
Synopsis: The study randomized 219 adult asthma patients who were inadequately controlled despite inhaled corticosteroids to a placebo or lebrikizumab. The primary outcome was improvement in prebronchodilator FEV1 from baseline. Secondary outcomes were exacerbations, use of rescue medications, and symptom scores. Patients were also stratified and analyzed based on surrogate markers for IL-13, which included serum IGE levels, eosinophil counts, and periostin levels.
In patients who were randomized to the lebrikizumab treatment, there was a statistically significant improvement in FEV1 of 5.5%, which occurred almost immediately and was sustained for the entire 32 weeks of the study. The improvement was more significant in patients who had high surrogate markers for IL-13. Despite this improvement in FEV1, there were no differences in secondary outcomes except in patients who had surrogate markers for high IL-13 levels.
Bottom line: In adults with asthma who remained uncontrolled despite inhaled corticosteroid therapy, IL-13 antagonism with lebrikizumab improved FEV1. However, the clinical relevance of these modest improvements remains unclear.
Citation: Corren J, Lemanske R, Matthews J, et al. Lebrikizumab treatment in adults with asthma. N Engl J Med. 2011;365:1088-1098.
Rivaroxaban Is Noninferior to Warfarin for Stroke Prevention in Atrial Fibrillation
Clinical question: How does rivaroxaban compare with warfarin in the prevention of stroke or systemic embolism in patients with nonvalvular atrial fibrillation?
Background: Warfarin is effective for the prevention of stroke in atrial fibrillation, but it requires close monitoring and adjustment. Rivaroxaban, an oral Xa inhibitor, may be safer, easier, and more effective than warfarin.
Study design: Multicenter, randomized, double-blind, double-dummy trial.
Setting: 1,178 sites in 45 countries.
Synopsis: The study included 14,264 patients with nonvalvular atrial fibrillation who were randomized to either fixed-dose rivaroxaban (20 mg daily or 15 mg daily for CrCl 30-49 mL/min) plus placebo or adjusted-dose warfarin (target INR 2.0 to 3.0) plus placebo. The mean CHADS2 score was 3.5. The primary endpoint (stroke or systemic embolism) occurred in 1.7% of patients per year in the rivaroxaban group and 2.2% per year in the warfarin group (hazard ratio for rivaroxaban 0.79; 95% CI: 0.66 to 0.96, P<0.001 for noninferiority). There was no difference in major or nonmajor clinically significant bleeding between the two groups (14.9% rivaroxaban vs. 14.5% warfarin, hazard ratio=1.03, 95% CI: 0.96 to 1.11, P=0.44). There were fewer intracranial hemorrhages (0.5% vs. 0.7%, P=0.02) and fatal bleeding (0.2% vs. 0.5%, P=0.003) in the rivaroxaban group.
Bottom line: In patients with atrial fibrillation, rivaroxaban was noninferior to warfarin for the prevention of stroke or systemic embolization, with a similar risk of major bleeding and a lower risk of intracranial hemorrhage or fatal bleeding.
Citation: Patel MR, Mahaffey K, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365:883-891.
Apixaban More Effective and Safer than Warfarin for Stroke Prevention in Atrial Fibrillation
Clinical question: How does the effectiveness and safety of apixaban compare with warfarin for stroke prevention in atrial fibrillation?
Background: Until recently, warfarin has been the only available oral anticoagulant for stroke prevention in patients with atrial fibrillation (AF). The oral factor Xa inhibitors have shown similar efficacy and safety, without the monitoring requirement and drug interactions associated with warfarin.
Study design: Prospective randomized double-blind controlled trial.
Setting: More than 1,000 clinical sites in 39 countries.
Synopsis: This study randomized 18,201 patients with atrial fibrillation or flutter and at least one CHADS2 risk factor for stroke to receive oral apixaban or warfarin therapy. Exclusion criteria were prosthetic valves and severe kidney disease. The median duration of follow-up was 1.8 years, and the major endpoints were incidence of stroke, systemic embolism, bleeding complications, and mortality.
Compared with warfarin, apixaban reduced the annual incidence of stroke and systemic embolism from 1.6% to 1.3% (HR 0.79, 95%: CI 0.66 to 0.95, P=0.01 for superiority), and reduced mortality (HR: 0.89, 95% CI: 0.80 to 0.998). For the combined endpoint of stroke, systemic embolism, MI, or death, the annual rate was reduced from 5.5% to 4.9% (HR: 0.88, 95% CI: 0.80 to 0.97). All measures of bleeding were less frequent with apixaban: major 2.1% vs. 3.1% (HR: 0.69, 95% CI: 0.60 to 0.80), and combined major and minor bleeding 4.1% vs. 6.0% (HR: 0.68, 95% CI: 0.61 to 0.75). The annual rate for the net outcome of stroke, embolism, or major bleeding was 3.2% with apixaban and 4.1% with warfarin (HR: 0.77, 95% CI: 0.69 to 0.86).
Bottom line: Compared with warfarin therapy, apixaban is more effective and safer for stroke prevention in patients with atrial fibrillation.
Citation: Granger CB, Alexander JH, McMurray JJ, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011;365:981-992.
Ultrasonography Is Useful in Diagnosis of Pneumothorax
Clinical question: Is transthoracic ultrasonography a useful tool to diagnose pneumothorax?
Background: CT is the diagnostic gold standard for pneumothorax, but it is associated with radiation exposure and requires patient transport. Chest radiograph is easy to perform but may be too insensitive for adequate diagnosis. Ultrasonography’s diagnostic performance for detecting pneumothorax needs further evaluation.
Study design: Systematic review and meta-analysis.
Setting: Critically ill, trauma, or post-biopsy patients were identified in each of the studies.
Synopsis: The meta-analysis of 20 eligible studies found a pooled sensitivity of ultrasound for the detection of pneumothorax of 0.88 (95% CI: 0.85 to 0.91) and specificity of 0.99 (0.98 to 0.99) compared with sensitivity of 0.52 (0.49 to 0.55) and specificity of 1.00 (1.00 to 1.00) for chest radiograph. Although the overall ROC curve was not significantly different between these modalities, the accuracy of ultrasonography was highly dependent on the skill of the operator.
Bottom line: When performed by a skilled operator, transthoracic ultrasonography is as specific, and more sensitive, than chest radiograph in diagnosing pneumothorax.
Citation: Ding W, Shen Y, Yang J, He X, Zhang M. Diagnosis of pneumothorax by radiography and ultrasonography: a meta-analysis. Chest. 2011;140:859-866.
Risk Prediction for Hospital Readmission Remains Challenging
Clinical question: Can readmission risk assessment be used to identify which patients would benefit most from care-transition interventions, or to risk-adjust readmission rates for hospital comparison?
Background: Multiple models to predict hospital readmission have been described and validated. Identifying patients at high risk for readmission could allow for customized care-transition interventions, or could be used to risk-adjust readmission rates to compare publicly reported rates by hospital.
Study design: Systematic review with qualitative synthesis of results.
Setting: Thirty studies (23 from the U.S.) tested 26 unique readmission models.
Synopsis: Each model had been tested in both a derivation and validation cohort. Fourteen models (nine from the U.S.), using retrospective administrative data to compare risk-adjusted rates between hospitals, had poor discriminative capacity (c statistic range: 0.55 to 0.65). Seven models could be used to identify high-risk patients early in the hospitalization (c statistic range: 0.56 to 0.72) and five could be used to identify high-risk patients at discharge (c statistic range: 0.68 to 0.83), but these also had poor to moderate discriminative capacity. Multiple variables were considered in each of the models; most incorporated medical comorbidities and prior use of healthcare services.
Bottom line: Current readmission risk prediction models do not perform adequately for comparative or clinical purposes.
Citation: Kansagara D, Englander H, Salanitro A, et. al. Risk prediction models for hospital readmission: a systematic review. JAMA. 2011;306:1688-1698.
Intravenous Fluids for Acute Pancreatitis: More May Be Less
Clinical question: What is the optimal volume of fluid administration for treatment of acute pancreatitis?
Background: Current guidelines for management of acute pancreatitis emphasize vigorous administration of intravenous fluids to reduce the risk of pancreatic necrosis and organ failure. This recommendation is based upon animal studies, and has not been subjected to clinical evaluation in humans.
Study design: Prospective observational cohort.
Setting: University-affiliated tertiary-care public hospital in Spain.
Synopsis: This study enrolled 247 patients admitted with acute pancreatitis to determine the association between the volume of fluid administered during the first 24 hours and the development of persistent organ failure, pancreatic fluid collection or necrosis, and mortality. The volume and rate of fluid administered were determined by the treating physician. Patients were classified into three groups: those receiving a volume <3.1 L, 3.1 to 4.1 L, or >4.1 L.
After multivariate adjustment, those receiving <3.1 L had no increased risk of necrosis or any other adverse outcome, compared with those who received the middle range of fluid volume.
Patients receiving >4.1 L had a higher risk of persistent organ failure (OR: 7.7, 95% CI: 1.5 to 38.7), particularly renal and respiratory insufficiency, and fluid collection development (OR: 1.9, 95% CI: 1 to 3.7) independent of disease severity. Pancreatic necrosis and mortality were similar in the three groups.
Bottom line: Administration of large-volume intravenous fluids (>4.1 L) in
the first 24 hours was associated with worse outcomes, although residual confounding cannot be excluded in this nonrandomized study.
Citation: de-Madaria E, Soler-Sala G, Sanchez-Paya J, et al. Influence of fluid therapy on the prognosis of acute pancreatitis: a prospective cohort study. Am J Gastroenterol. 2011;106:1843-1850.
Clinical Outcomes in Saddle Pulmonary Embolism
Clinical question: What are the treatments used and outcomes associated with saddle pulmonary embolism?
Background: Saddle pulmonary embolism is a risk for right ventricular dysfunction and sudden hemodynamic collapse. There are limited data on the clinical presentation and outcomes in these patients.
Study design: Retrospective case review.
Setting: Single academic medical center.
Synopsis: In this retrospective review of 680 patients diagnosed with pulmonary embolism on CT at a single academic medical center from 2004 to 2009, 5.4% (37 patients) had a saddle pulmonary embolism.
Most patients with saddle pulmonary embolism were hemodynamically stable and responded to standard therapy with unfractionated heparin. The mean length of stay was nine days, 46% received an inferior vena cava filter, 41% were treated in an ICU, and 5.4% (two patients) died in the hospital. Thrombolytics were used in only 11% of patients, most of which had sustained hypotension and/or were mechanically ventilated.
Bottom line: Most patients with saddle pulmonary embolus in this single institution study did not receive thrombolytics and had overall low mortality.
Citation: Sardi A, Gluskin J, Guttentag A, Kotler MN, Braitman LE, Lippmann M. Saddle pulmonary embolism: is it as bad as it looks? A community hospital experience. Crit Care Med. 2011;39:2413-2418.
Early Fluids Might Decrease Renal Morbidity in Hemolytic Uremic Syndrome
Clinical question: Does intravenous volume expansion during diarrheal illness mitigate the nephrotoxic effects of hemolytic uremic syndrome (HUS)?
Background: HUS often results in significant morbidity, particularly when oligoanuria is also present. Shiga-toxin-producing bacteria, notoriously Escherichia coli O157:H7 in the context of a diarrheal illness, are the most common cause, and worldwide outbreaks have been increasingly described. One prior report suggests that early IV fluid administration results in improved outcomes.
Study design: Prospective cohort study.
Setting: Eleven pediatric hospitals in the U.S. and Scotland.
Synopsis: Fifty children with diarrhea-associated HUS were enrolled and received clinical care at the discretion of the local provider, independent of the study. A family questionnaire (to define initial illness) and chart review were subsequently performed. Oligoanuria, defined as a urine output of less than 0.5 mL/kg/hr for at least one calendar day after HUS onset, was present in 34 (68%) patients. Oligoanuric and nonoligoanuric patients were similar at baseline; however, there was a significant association between less fluid administration in the first four days of illness and oligoanuria. Specifically, lack of IV fluids portended a 1.6 times higher likelihood of oligoanuria (95% confidence interval, 1.1-2.4; P=0.02).
The authors also suggest a dose-response relationship to their findings, which potentially strengthens their findings. However, the practical applicability of these findings appears limited. Many of the patients who did not receive IV fluids early on were also not admitted to a hospital, likely signifying mild illness without notable dehydration. Replicability of the benefits of early volume expansion in HUS will depend on the ability to accurately identify patients with Shiga-toxin-producing diarrheal illnesses at presentation. If this is feasible, it would be interesting to examine the details of oral hydration as well, particularly in those who are not dehydrated enough to require hospitalization.
Bottom line: Early IV fluids might be nephroprotective in diarrhea-associated HUS.
Citation: Hickey CA, Beattie J, Cowieson J, et al. Early volume expansion during diarrhea and relative nephroprotection during subsequent hemolytic uremic syndrome. Arch Pediatr Adolesc Med. 2011;165:884-889.
Reviewed by Pediatric Editor Mark Shen, MD, FHM, medical director of hospital medicine at Dell Children’s Medical Center, Austin, Texas.
Clinical question: Does intravenous volume expansion during diarrheal illness mitigate the nephrotoxic effects of hemolytic uremic syndrome (HUS)?
Background: HUS often results in significant morbidity, particularly when oligoanuria is also present. Shiga-toxin-producing bacteria, notoriously Escherichia coli O157:H7 in the context of a diarrheal illness, are the most common cause, and worldwide outbreaks have been increasingly described. One prior report suggests that early IV fluid administration results in improved outcomes.
Study design: Prospective cohort study.
Setting: Eleven pediatric hospitals in the U.S. and Scotland.
Synopsis: Fifty children with diarrhea-associated HUS were enrolled and received clinical care at the discretion of the local provider, independent of the study. A family questionnaire (to define initial illness) and chart review were subsequently performed. Oligoanuria, defined as a urine output of less than 0.5 mL/kg/hr for at least one calendar day after HUS onset, was present in 34 (68%) patients. Oligoanuric and nonoligoanuric patients were similar at baseline; however, there was a significant association between less fluid administration in the first four days of illness and oligoanuria. Specifically, lack of IV fluids portended a 1.6 times higher likelihood of oligoanuria (95% confidence interval, 1.1-2.4; P=0.02).
The authors also suggest a dose-response relationship to their findings, which potentially strengthens their findings. However, the practical applicability of these findings appears limited. Many of the patients who did not receive IV fluids early on were also not admitted to a hospital, likely signifying mild illness without notable dehydration. Replicability of the benefits of early volume expansion in HUS will depend on the ability to accurately identify patients with Shiga-toxin-producing diarrheal illnesses at presentation. If this is feasible, it would be interesting to examine the details of oral hydration as well, particularly in those who are not dehydrated enough to require hospitalization.
Bottom line: Early IV fluids might be nephroprotective in diarrhea-associated HUS.
Citation: Hickey CA, Beattie J, Cowieson J, et al. Early volume expansion during diarrhea and relative nephroprotection during subsequent hemolytic uremic syndrome. Arch Pediatr Adolesc Med. 2011;165:884-889.
Reviewed by Pediatric Editor Mark Shen, MD, FHM, medical director of hospital medicine at Dell Children’s Medical Center, Austin, Texas.
Clinical question: Does intravenous volume expansion during diarrheal illness mitigate the nephrotoxic effects of hemolytic uremic syndrome (HUS)?
Background: HUS often results in significant morbidity, particularly when oligoanuria is also present. Shiga-toxin-producing bacteria, notoriously Escherichia coli O157:H7 in the context of a diarrheal illness, are the most common cause, and worldwide outbreaks have been increasingly described. One prior report suggests that early IV fluid administration results in improved outcomes.
Study design: Prospective cohort study.
Setting: Eleven pediatric hospitals in the U.S. and Scotland.
Synopsis: Fifty children with diarrhea-associated HUS were enrolled and received clinical care at the discretion of the local provider, independent of the study. A family questionnaire (to define initial illness) and chart review were subsequently performed. Oligoanuria, defined as a urine output of less than 0.5 mL/kg/hr for at least one calendar day after HUS onset, was present in 34 (68%) patients. Oligoanuric and nonoligoanuric patients were similar at baseline; however, there was a significant association between less fluid administration in the first four days of illness and oligoanuria. Specifically, lack of IV fluids portended a 1.6 times higher likelihood of oligoanuria (95% confidence interval, 1.1-2.4; P=0.02).
The authors also suggest a dose-response relationship to their findings, which potentially strengthens their findings. However, the practical applicability of these findings appears limited. Many of the patients who did not receive IV fluids early on were also not admitted to a hospital, likely signifying mild illness without notable dehydration. Replicability of the benefits of early volume expansion in HUS will depend on the ability to accurately identify patients with Shiga-toxin-producing diarrheal illnesses at presentation. If this is feasible, it would be interesting to examine the details of oral hydration as well, particularly in those who are not dehydrated enough to require hospitalization.
Bottom line: Early IV fluids might be nephroprotective in diarrhea-associated HUS.
Citation: Hickey CA, Beattie J, Cowieson J, et al. Early volume expansion during diarrhea and relative nephroprotection during subsequent hemolytic uremic syndrome. Arch Pediatr Adolesc Med. 2011;165:884-889.
Reviewed by Pediatric Editor Mark Shen, MD, FHM, medical director of hospital medicine at Dell Children’s Medical Center, Austin, Texas.
What Are Common Causes of Glomerular Disease in Adults?
The Case
A 52-year-old man presents with abdominal pain. His temperature is 100.8°F, his blood pressure is 170/90 mm/Hg, and his pulse is 110 beats per minute. On exam, he has 2+ lower extremity edema, periorbital edema, and left-sided flank tenderness. His BUN is 42 mg/dL, his creatinine is 2.5 mg/dL, and his albumin is 1.4 g/dL. Urinalysis shows 2+ protein, large blood, and red blood cells (RBCs). What are the next steps in his diagnosis?
Overview
Glomerular diseases involve a wide spectrum of disease processes. They can result from an acute illness, such as an upper respiratory infection that self-resolves, or from chronic disease states, such as HIV. In some instances, such illnesses as systemic lupus erythematosus (SLE) can cause rapidly progressive renal failure, requiring prompt intervention. While glomerular diseases can be daunting, it is essential for hospitalists to be familiar with fundamental concepts and key features unique to each syndrome.
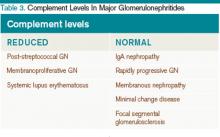
The approach to glomerulonephritis (GN) can be simplified by summarizing various types into the two broad categories of nephrotic and nephritic syndromes, and identifying the key clinical findings (see Table 1, p. below).
The major subtypes of nephrotic syndrome are minimal change disease (MCD), focal segmental glomerulosclerosis (FSGS), membranous nephropathy (MN), and membranoproliferative glomerulonephritis (MPGN). The clinical manifestations of nephrotic syndrome are edema, hyperlipidemia, lipiduria, and hypoalbuminemia.1 The urinalysis is significant for >3.5 g/day of proteinuria showing fatty casts or oval fat bodies.2 The loss of other proteins, such as anti-thrombin III, may put patients at higher risk for developing venous thromboses.1
The major subtypes of nephritic syndrome are post-streptococcal glomerulonephritis (PSGS), IgA nephropathy, Henoch-Schonlein Purpura (HSP), and rapidly progressive GN (RPGN types I, II, and III). The clinical manifestations of nephritic syndrome are hypertension (HTN) and hematuria.1 Nephritic syndromes may present with more rapidly progressive renal failure when compared with nephrotic syndrome.1 The urinalysis is significant for hematuria with RBC casts, and variable levels of proteinuria (typically, less than 3.5 g/day is seen in nephritic syndrome).1
Review of the Data
Nephrotic Syndromes
Minimal change disease. MCD is more common in children than adults, and only accounts for 10% to 15% of glomerular disease cases in adults.3 It is associated with Hodgkin’s lymphoma, NSAID use, and allergic conditions. There usually is an absence of hypertension (HTN). There are no glomerular basement membrane abnormalities seen on light microscopy. Electron microscopy shows effacement of podocytes. On urinalysis, oval fat bodies are seen, which are characteristic of heavy proteinuria. Complement levels are normal. Steroids are first-line treatment, but in adults with relapses or steroid resistance, immunosuppressive agents have also been used.2
Focal segment glomerlosclerosis.
FSGS is the most common primary glomerular disorder in the United States and is the most common cause of nephrotic syndrome among blacks.4,5 It is associated with HIV (collapsing variant), parvovirus B19, heroin use, sickle-cell disease, obesity, chronic vesicoureteral reflux, and HTN.4,6 Sclerosis of segmental glomeruli is seen on light microscopy.
Electron microscopy shows effacement of podocytes. Complement levels are normal. Treatment of primary idiopathic FSGS includes use of renin-angiotensin inhibitors and steroids.4 Immunosuppressives are reserved for relapses. Treatment of secondary FSGS involves identifying the underlying cause.
Membranous nephropathy. MN is twice as common in males as in females and is the most common cause of adult-onset idiopathic nephrotic syndrome, with the average presentation in the fifth or sixth decade of life.7,8 Aside from its idiopathic form, up to 25% of MN cases have an underlying disease process, such as solid organ tumors or hepatitis B.7,9 While nephrotic syndrome overall can increase the risk of thromboembolic complications, MN is the most common nephrotic disorder predisposing the development of renal vein thrombosis.7 Diffuse capillary wall thickening is seen on light microscopy, and electron microscopy shows sub-epithelial immune deposits. Complement levels are normal. Steroids and immunosuppressive agents are used for treatment.10
Membranoproliferative glomerulonephritis. MPGN is a nephrotic syndrome that is more common in children and young adults and can present with features of nephritic syndrome.1,11 It is associated with hepatitis C, SLE, and cryoglobulinemia.11 Light microscopy shows mesangial and endocapillary proliferation, as well as glomerular basement membrane thickening and splitting (“tram track” appearance). Electron microscopy shows subendothelial and dense deposits. It presents with reduced complement levels (C3 and C4).11 Treatment depends on the associated disease.
Nephritic Syndromes
Post-streptococcal glomerulonephritis. PSGN is seen in children and young adults and is associated with skin (impetigo) and throat infections (pharyngitis).12 Hematuria usually presents two to three weeks after a streptococcal infection. The urine is classically dark and smoky-colored. Levels of C3 and CH50 are low, but C4 levels are normal.1 In addition, there are positive antibody titers for ASO and anti-DNase. Light microscopy shows hypercellularity of glomeruli. Electron microscopy shows dome-shaped sub-endothelial deposits. Treatment is usually supportive.
IgA nephropathy. IgA nephropathy is the most common form of glomerular disease worldwide and the most common form of glomerular-related microscopic hematuria in all age groups.2,13 It occurs in all ages but more frequently in males.14 It occurs during or immediately after an upper respiratory infection. Light microscopy shows mesangial cell proliferation and crescentic GN. Electron microscopy shows immune deposits in the mesangium. Complement levels are normal. There has been no proven therapy, but ACE inhibitors (ACE-Is), angiotensin receptor blockers (ARBs), fish oil, steroids, and tonsillectomy have been used with some success.14 The clinical course of IgA nephropathy can be highly variable, with the potential for a benign course to rapidly progressive renal failure, with 15% to 40% of patients developing end-stage renal disease.14
Henoch-Schonlein purpura. HSP affects children more than adults and is the systemic form of IgA nephropathy.14 Most cases are idiopathic. Clinical
manifestations include: HTN; purpuric palpable rash on buttocks, ankles, and legs; bloody diarrhea with abdominal cramps; and pain in wrist, ankle, and knee joints.15 Light microscopy shows mesangial cell proliferation. Immune deposits in the mesangium are seen on electron microscopy. Complement levels are normal. Treatment is supportive.
Rapidly progressive glomerulonephritis types I, II, and III. RPGN represents a wide variety of disease states in which rapid progression to renal failure is seen within days to weeks.16 They are categorized into three sub-categories: I, II, and III.
Type I is an anti-GBM disease, an example being Goodpasture’s syndrome. This condition presents with hemoptysis, pulmonary infiltrates, and hematuria with RBC casts. Anti-GBM antibodies are classically found.1 Complement levels are normal and a linear immunofluorescence pattern is seen. Treatment is steroids, immunosuppressive agents, and plasmapheresis.17
Type II is an immune complex deposition disease, such as HSP, SLE, or post-streptococcal GN, in which granular complex deposits are seen. Treatment is directed toward treating the underlying cause.
Type III is pauci-immune (no immune deposits), showing necrotizing crescentic GN on biopsy, and is associated with a positive ANCA.1,18 They are associated with systemic small-vessel vasculitis, such as granulomatosis with polyangiitis (formerly known as Wegener’s granulomatosis), microscopic polyangiitis, and Churg-Strauss syndrome, or can be limited to renal involvement.1,18 Complement levels are normal. Treatment is steroids and immunosuppressive agents, such as cyclophosphamide.
A summary of the findings found in the glomerulonephritides and how complement levels are affected are found in Table 2 and Table 3, respectively.
Secondary Causes of Nephrotic Diseases
Diabetic nephropathy. Diabetic nephropathy is the single most common cause of progressive renal failure in the United States.3 Up to 50% of patients with diabetes present with diabetic nephropathy.19 Current recommendations are to screen yearly for microalbuminuria at the time of diagnosis.3 Treatment involves use of ACE-Is or ARBs to reduce proteinuria and slow the progression of renal disease.
HIV-associated nephropathy. HIV-associated nephropathy commonly presents as the collapsing variant of FSGS. However, it can present as other forms of glomerulopathy, such as MPGN or IgA nephropathy, as well as an immune complex GN with “lupus-like” features without evidence of SLE.19,20 Therefore, HIV nephropathy has now been categorized as a separate entity.3 ACE-Is, HAART therapy, and corticosteroids are the mainstays of treatment.
Amyloidosis. Renal involvement is seen in both primary (AL) and secondary (AA) amyloidosis. Eighty percent of patients with AL have renal disease, and 25% of these patients have nephrotic syndrome.16 Diagnosis is made with Congo Red stain, which shows fibrillary amyloid deposits within the mesangium and capillary walls. Treatment is directed at the underlying process.
Systemic lupus erythematosus. SLE is divided into six classes (I-VI) based on the involvement and severity of renal disease, and steroids and immunosuppressive agents are used for treatment, also based on the severity of the disease.21
Back to the Case
Our patient presented to the hospital with abdominal pain, low-grade fever, HTN, edema, hypoalbuminemia, and new-onset renal failure with gross hematuria and proteinuria. The presence of proteinuria and hypoalbuminemia, combined with peripheral and periorbital edema, suggests glomerular loss of albumin, such as in nephrotic syndrome. His renal failure in the setting of the sudden development of gross hematuria with flank pain is concerning for a renal vein thrombosis, and an abdominal magnetic resonance venography did in fact visualize a renal vein thrombosis.
He was admitted to the hospital and was started on therapeutic intravenous heparin, and bridged to warfarin. Subsequent renal biopsy confirmed the findings of membranous nephropathy, which was suspected due to his renal vein thrombosis. Therapy was initiated with corticosteroids after the biopsy, and he responded well. Because of his risk factors for further thromboembolic events, lifelong anticoagulation therapy was recommended.
Bottom Line
For patients with glomerular disease, differentiating between nephrotic and nephritic syndromes and understanding key clinical and laboratory differences can lead to easier identification and treatment.
Drs. Khan and Smith are assistant professors of medicine, and Dr. Ansari is associate division director, in the Division of Hospital Medicine at Loyola University Medical Center, Maywood, Ill.
References
- Donegio RGB, Salant DJ. Nephrology: Glomerular Diseases. In: ACP Medicine. Dale D, Federman D, eds. Available at: http://www.acpmedicine.com/acpmedicine/institutional/instHtmlReader.action?readerFlag=chapt&chapId=part10_ch05. Accessed Feb. 15, 2012.
- Orth SR, Ritz E. The nephrotic syndrome. N Engl J Med. 1998;338:1202-1211.
- Lewis JB, Neilson EG. Chapter 283. Glomerular Diseases. In: Longo DL, Fauci AS, Kasper DL, Hauser SL, Jameson JL, Loscalzo J, eds. Harrison’s Principles of Internal Medicine. 18th ed. New York: McGraw-Hill; 2012.
- D’Agati VD, Kaskel FJ, Falk RJ. Focal segmental glomerulosclerosis. N Engl J Med. 2011;365:2398-23411.
- Kitiyakara C, Eggers P, Kopp JB. Twenty-one-year trend in ESRD due to focal segmental glomerulosclerosis in the United States. Am J Kidney Dis. 2004;44:815-825.
- Balow JE. Nephropathy in the context of HIV infection. Kidney Int. 2005;67:1632-1633.
- Glassock RJ. Diagnosis and natural course of membranous nephropathy. Semin Nephrol. 2003;23:324-332.
- Nickolas TL, Radhakrishnan J, Appel GB. Hyperlipidemia and thrombotic complications in patients with membranous nephropathy. Semin Nephrol. 2003;23:406-411.
- Burstein DM, Korbet SM, Schwartz MM. Membranous glomerulonephritis and malignancy. Am J Kidney Dis. 1993;22:5-10.
- Hofstra JM, Wetzels JF. Management of patients with membranous nephropathy. Nephrol Dial Transplant. 2012;27:6-9.
- Alchi B, Jayne D. Membranoproliferative glomerulonephritis. Pediatr Nephrol. 2010;25:1409-1418.
- Eison TM, Ault BH, Jones DP, Chesney RW, Wyatt RJ. Post-streptococcal acute glomerulonephritis in children: clinical features and pathogenesis. Pediatr Nephrol. 2011;26:165-180.
- Cohen RA, Brown RS. Clinical practice. Microscopic hematuria. N Engl J Med. 2003;348:2330-2338.
- Donadio JV, Grande JP. IgA nephropathy. N Engl J Med. 2002;347:738-748.
- McCarthy HJ, Tizard EJ. Clinical practice: Diagnosis and management of henoch-schonlein purpura. Eur J Pediatr. 2010169:643-650.
- Appel GB, Radhakrishnan J. Cecil Medicine: Volume 1: Chapter 123: Glomerular Disorders and Nephrotic Syndromes. MD Consult Preview website. Available at: http://www.mdconsult.com/books/page.do?eid=4-u1.0-B978-1-4377-1604-7..00123-8&isbn=978-1-4377-1604-7&uniqId=313771243-2#4-u1.0-B978-1-4377-1604-7..00123-8. Accessed Feb. 16, 2012.
- Walters G, Willis NS, Craig JC. Interventions for renal vasculitis in adults. Cochrane Database Syst Rev. 2008;(3)(3):CD003232.
- Jennette JC. Rapidly progressive crescentic glomerulonephritis. Kidney Int. 2003;63:1164-1177.
- Falk, RJ: Medical Knowledge Self Assessment Program 14. Nephrology: 2006.
- Haas M, Kaul S, Eustace JA. HIV-associated immune complex glomerulonephritis with “lupus-like” features: a clinicopathologic study of 14 cases. Kidney Int. 2005;67:1381.
- Dooley MA, Hogan S, Jennette C, Falk R. Cyclophosphamide therapy for lupus nephritis: poor renal survival in black americans. glomerular disease collaborative network. Kidney Int. 1997;51:1188-1195.
The Case
A 52-year-old man presents with abdominal pain. His temperature is 100.8°F, his blood pressure is 170/90 mm/Hg, and his pulse is 110 beats per minute. On exam, he has 2+ lower extremity edema, periorbital edema, and left-sided flank tenderness. His BUN is 42 mg/dL, his creatinine is 2.5 mg/dL, and his albumin is 1.4 g/dL. Urinalysis shows 2+ protein, large blood, and red blood cells (RBCs). What are the next steps in his diagnosis?
Overview
Glomerular diseases involve a wide spectrum of disease processes. They can result from an acute illness, such as an upper respiratory infection that self-resolves, or from chronic disease states, such as HIV. In some instances, such illnesses as systemic lupus erythematosus (SLE) can cause rapidly progressive renal failure, requiring prompt intervention. While glomerular diseases can be daunting, it is essential for hospitalists to be familiar with fundamental concepts and key features unique to each syndrome.
The approach to glomerulonephritis (GN) can be simplified by summarizing various types into the two broad categories of nephrotic and nephritic syndromes, and identifying the key clinical findings (see Table 1, p. below).
The major subtypes of nephrotic syndrome are minimal change disease (MCD), focal segmental glomerulosclerosis (FSGS), membranous nephropathy (MN), and membranoproliferative glomerulonephritis (MPGN). The clinical manifestations of nephrotic syndrome are edema, hyperlipidemia, lipiduria, and hypoalbuminemia.1 The urinalysis is significant for >3.5 g/day of proteinuria showing fatty casts or oval fat bodies.2 The loss of other proteins, such as anti-thrombin III, may put patients at higher risk for developing venous thromboses.1
The major subtypes of nephritic syndrome are post-streptococcal glomerulonephritis (PSGS), IgA nephropathy, Henoch-Schonlein Purpura (HSP), and rapidly progressive GN (RPGN types I, II, and III). The clinical manifestations of nephritic syndrome are hypertension (HTN) and hematuria.1 Nephritic syndromes may present with more rapidly progressive renal failure when compared with nephrotic syndrome.1 The urinalysis is significant for hematuria with RBC casts, and variable levels of proteinuria (typically, less than 3.5 g/day is seen in nephritic syndrome).1
Review of the Data
Nephrotic Syndromes
Minimal change disease. MCD is more common in children than adults, and only accounts for 10% to 15% of glomerular disease cases in adults.3 It is associated with Hodgkin’s lymphoma, NSAID use, and allergic conditions. There usually is an absence of hypertension (HTN). There are no glomerular basement membrane abnormalities seen on light microscopy. Electron microscopy shows effacement of podocytes. On urinalysis, oval fat bodies are seen, which are characteristic of heavy proteinuria. Complement levels are normal. Steroids are first-line treatment, but in adults with relapses or steroid resistance, immunosuppressive agents have also been used.2
Focal segment glomerlosclerosis.
FSGS is the most common primary glomerular disorder in the United States and is the most common cause of nephrotic syndrome among blacks.4,5 It is associated with HIV (collapsing variant), parvovirus B19, heroin use, sickle-cell disease, obesity, chronic vesicoureteral reflux, and HTN.4,6 Sclerosis of segmental glomeruli is seen on light microscopy.
Electron microscopy shows effacement of podocytes. Complement levels are normal. Treatment of primary idiopathic FSGS includes use of renin-angiotensin inhibitors and steroids.4 Immunosuppressives are reserved for relapses. Treatment of secondary FSGS involves identifying the underlying cause.
Membranous nephropathy. MN is twice as common in males as in females and is the most common cause of adult-onset idiopathic nephrotic syndrome, with the average presentation in the fifth or sixth decade of life.7,8 Aside from its idiopathic form, up to 25% of MN cases have an underlying disease process, such as solid organ tumors or hepatitis B.7,9 While nephrotic syndrome overall can increase the risk of thromboembolic complications, MN is the most common nephrotic disorder predisposing the development of renal vein thrombosis.7 Diffuse capillary wall thickening is seen on light microscopy, and electron microscopy shows sub-epithelial immune deposits. Complement levels are normal. Steroids and immunosuppressive agents are used for treatment.10
Membranoproliferative glomerulonephritis. MPGN is a nephrotic syndrome that is more common in children and young adults and can present with features of nephritic syndrome.1,11 It is associated with hepatitis C, SLE, and cryoglobulinemia.11 Light microscopy shows mesangial and endocapillary proliferation, as well as glomerular basement membrane thickening and splitting (“tram track” appearance). Electron microscopy shows subendothelial and dense deposits. It presents with reduced complement levels (C3 and C4).11 Treatment depends on the associated disease.
Nephritic Syndromes
Post-streptococcal glomerulonephritis. PSGN is seen in children and young adults and is associated with skin (impetigo) and throat infections (pharyngitis).12 Hematuria usually presents two to three weeks after a streptococcal infection. The urine is classically dark and smoky-colored. Levels of C3 and CH50 are low, but C4 levels are normal.1 In addition, there are positive antibody titers for ASO and anti-DNase. Light microscopy shows hypercellularity of glomeruli. Electron microscopy shows dome-shaped sub-endothelial deposits. Treatment is usually supportive.
IgA nephropathy. IgA nephropathy is the most common form of glomerular disease worldwide and the most common form of glomerular-related microscopic hematuria in all age groups.2,13 It occurs in all ages but more frequently in males.14 It occurs during or immediately after an upper respiratory infection. Light microscopy shows mesangial cell proliferation and crescentic GN. Electron microscopy shows immune deposits in the mesangium. Complement levels are normal. There has been no proven therapy, but ACE inhibitors (ACE-Is), angiotensin receptor blockers (ARBs), fish oil, steroids, and tonsillectomy have been used with some success.14 The clinical course of IgA nephropathy can be highly variable, with the potential for a benign course to rapidly progressive renal failure, with 15% to 40% of patients developing end-stage renal disease.14
Henoch-Schonlein purpura. HSP affects children more than adults and is the systemic form of IgA nephropathy.14 Most cases are idiopathic. Clinical
manifestations include: HTN; purpuric palpable rash on buttocks, ankles, and legs; bloody diarrhea with abdominal cramps; and pain in wrist, ankle, and knee joints.15 Light microscopy shows mesangial cell proliferation. Immune deposits in the mesangium are seen on electron microscopy. Complement levels are normal. Treatment is supportive.
Rapidly progressive glomerulonephritis types I, II, and III. RPGN represents a wide variety of disease states in which rapid progression to renal failure is seen within days to weeks.16 They are categorized into three sub-categories: I, II, and III.
Type I is an anti-GBM disease, an example being Goodpasture’s syndrome. This condition presents with hemoptysis, pulmonary infiltrates, and hematuria with RBC casts. Anti-GBM antibodies are classically found.1 Complement levels are normal and a linear immunofluorescence pattern is seen. Treatment is steroids, immunosuppressive agents, and plasmapheresis.17
Type II is an immune complex deposition disease, such as HSP, SLE, or post-streptococcal GN, in which granular complex deposits are seen. Treatment is directed toward treating the underlying cause.
Type III is pauci-immune (no immune deposits), showing necrotizing crescentic GN on biopsy, and is associated with a positive ANCA.1,18 They are associated with systemic small-vessel vasculitis, such as granulomatosis with polyangiitis (formerly known as Wegener’s granulomatosis), microscopic polyangiitis, and Churg-Strauss syndrome, or can be limited to renal involvement.1,18 Complement levels are normal. Treatment is steroids and immunosuppressive agents, such as cyclophosphamide.
A summary of the findings found in the glomerulonephritides and how complement levels are affected are found in Table 2 and Table 3, respectively.
Secondary Causes of Nephrotic Diseases
Diabetic nephropathy. Diabetic nephropathy is the single most common cause of progressive renal failure in the United States.3 Up to 50% of patients with diabetes present with diabetic nephropathy.19 Current recommendations are to screen yearly for microalbuminuria at the time of diagnosis.3 Treatment involves use of ACE-Is or ARBs to reduce proteinuria and slow the progression of renal disease.
HIV-associated nephropathy. HIV-associated nephropathy commonly presents as the collapsing variant of FSGS. However, it can present as other forms of glomerulopathy, such as MPGN or IgA nephropathy, as well as an immune complex GN with “lupus-like” features without evidence of SLE.19,20 Therefore, HIV nephropathy has now been categorized as a separate entity.3 ACE-Is, HAART therapy, and corticosteroids are the mainstays of treatment.
Amyloidosis. Renal involvement is seen in both primary (AL) and secondary (AA) amyloidosis. Eighty percent of patients with AL have renal disease, and 25% of these patients have nephrotic syndrome.16 Diagnosis is made with Congo Red stain, which shows fibrillary amyloid deposits within the mesangium and capillary walls. Treatment is directed at the underlying process.
Systemic lupus erythematosus. SLE is divided into six classes (I-VI) based on the involvement and severity of renal disease, and steroids and immunosuppressive agents are used for treatment, also based on the severity of the disease.21
Back to the Case
Our patient presented to the hospital with abdominal pain, low-grade fever, HTN, edema, hypoalbuminemia, and new-onset renal failure with gross hematuria and proteinuria. The presence of proteinuria and hypoalbuminemia, combined with peripheral and periorbital edema, suggests glomerular loss of albumin, such as in nephrotic syndrome. His renal failure in the setting of the sudden development of gross hematuria with flank pain is concerning for a renal vein thrombosis, and an abdominal magnetic resonance venography did in fact visualize a renal vein thrombosis.
He was admitted to the hospital and was started on therapeutic intravenous heparin, and bridged to warfarin. Subsequent renal biopsy confirmed the findings of membranous nephropathy, which was suspected due to his renal vein thrombosis. Therapy was initiated with corticosteroids after the biopsy, and he responded well. Because of his risk factors for further thromboembolic events, lifelong anticoagulation therapy was recommended.
Bottom Line
For patients with glomerular disease, differentiating between nephrotic and nephritic syndromes and understanding key clinical and laboratory differences can lead to easier identification and treatment.
Drs. Khan and Smith are assistant professors of medicine, and Dr. Ansari is associate division director, in the Division of Hospital Medicine at Loyola University Medical Center, Maywood, Ill.
References
- Donegio RGB, Salant DJ. Nephrology: Glomerular Diseases. In: ACP Medicine. Dale D, Federman D, eds. Available at: http://www.acpmedicine.com/acpmedicine/institutional/instHtmlReader.action?readerFlag=chapt&chapId=part10_ch05. Accessed Feb. 15, 2012.
- Orth SR, Ritz E. The nephrotic syndrome. N Engl J Med. 1998;338:1202-1211.
- Lewis JB, Neilson EG. Chapter 283. Glomerular Diseases. In: Longo DL, Fauci AS, Kasper DL, Hauser SL, Jameson JL, Loscalzo J, eds. Harrison’s Principles of Internal Medicine. 18th ed. New York: McGraw-Hill; 2012.
- D’Agati VD, Kaskel FJ, Falk RJ. Focal segmental glomerulosclerosis. N Engl J Med. 2011;365:2398-23411.
- Kitiyakara C, Eggers P, Kopp JB. Twenty-one-year trend in ESRD due to focal segmental glomerulosclerosis in the United States. Am J Kidney Dis. 2004;44:815-825.
- Balow JE. Nephropathy in the context of HIV infection. Kidney Int. 2005;67:1632-1633.
- Glassock RJ. Diagnosis and natural course of membranous nephropathy. Semin Nephrol. 2003;23:324-332.
- Nickolas TL, Radhakrishnan J, Appel GB. Hyperlipidemia and thrombotic complications in patients with membranous nephropathy. Semin Nephrol. 2003;23:406-411.
- Burstein DM, Korbet SM, Schwartz MM. Membranous glomerulonephritis and malignancy. Am J Kidney Dis. 1993;22:5-10.
- Hofstra JM, Wetzels JF. Management of patients with membranous nephropathy. Nephrol Dial Transplant. 2012;27:6-9.
- Alchi B, Jayne D. Membranoproliferative glomerulonephritis. Pediatr Nephrol. 2010;25:1409-1418.
- Eison TM, Ault BH, Jones DP, Chesney RW, Wyatt RJ. Post-streptococcal acute glomerulonephritis in children: clinical features and pathogenesis. Pediatr Nephrol. 2011;26:165-180.
- Cohen RA, Brown RS. Clinical practice. Microscopic hematuria. N Engl J Med. 2003;348:2330-2338.
- Donadio JV, Grande JP. IgA nephropathy. N Engl J Med. 2002;347:738-748.
- McCarthy HJ, Tizard EJ. Clinical practice: Diagnosis and management of henoch-schonlein purpura. Eur J Pediatr. 2010169:643-650.
- Appel GB, Radhakrishnan J. Cecil Medicine: Volume 1: Chapter 123: Glomerular Disorders and Nephrotic Syndromes. MD Consult Preview website. Available at: http://www.mdconsult.com/books/page.do?eid=4-u1.0-B978-1-4377-1604-7..00123-8&isbn=978-1-4377-1604-7&uniqId=313771243-2#4-u1.0-B978-1-4377-1604-7..00123-8. Accessed Feb. 16, 2012.
- Walters G, Willis NS, Craig JC. Interventions for renal vasculitis in adults. Cochrane Database Syst Rev. 2008;(3)(3):CD003232.
- Jennette JC. Rapidly progressive crescentic glomerulonephritis. Kidney Int. 2003;63:1164-1177.
- Falk, RJ: Medical Knowledge Self Assessment Program 14. Nephrology: 2006.
- Haas M, Kaul S, Eustace JA. HIV-associated immune complex glomerulonephritis with “lupus-like” features: a clinicopathologic study of 14 cases. Kidney Int. 2005;67:1381.
- Dooley MA, Hogan S, Jennette C, Falk R. Cyclophosphamide therapy for lupus nephritis: poor renal survival in black americans. glomerular disease collaborative network. Kidney Int. 1997;51:1188-1195.
The Case
A 52-year-old man presents with abdominal pain. His temperature is 100.8°F, his blood pressure is 170/90 mm/Hg, and his pulse is 110 beats per minute. On exam, he has 2+ lower extremity edema, periorbital edema, and left-sided flank tenderness. His BUN is 42 mg/dL, his creatinine is 2.5 mg/dL, and his albumin is 1.4 g/dL. Urinalysis shows 2+ protein, large blood, and red blood cells (RBCs). What are the next steps in his diagnosis?
Overview
Glomerular diseases involve a wide spectrum of disease processes. They can result from an acute illness, such as an upper respiratory infection that self-resolves, or from chronic disease states, such as HIV. In some instances, such illnesses as systemic lupus erythematosus (SLE) can cause rapidly progressive renal failure, requiring prompt intervention. While glomerular diseases can be daunting, it is essential for hospitalists to be familiar with fundamental concepts and key features unique to each syndrome.
The approach to glomerulonephritis (GN) can be simplified by summarizing various types into the two broad categories of nephrotic and nephritic syndromes, and identifying the key clinical findings (see Table 1, p. below).
The major subtypes of nephrotic syndrome are minimal change disease (MCD), focal segmental glomerulosclerosis (FSGS), membranous nephropathy (MN), and membranoproliferative glomerulonephritis (MPGN). The clinical manifestations of nephrotic syndrome are edema, hyperlipidemia, lipiduria, and hypoalbuminemia.1 The urinalysis is significant for >3.5 g/day of proteinuria showing fatty casts or oval fat bodies.2 The loss of other proteins, such as anti-thrombin III, may put patients at higher risk for developing venous thromboses.1
The major subtypes of nephritic syndrome are post-streptococcal glomerulonephritis (PSGS), IgA nephropathy, Henoch-Schonlein Purpura (HSP), and rapidly progressive GN (RPGN types I, II, and III). The clinical manifestations of nephritic syndrome are hypertension (HTN) and hematuria.1 Nephritic syndromes may present with more rapidly progressive renal failure when compared with nephrotic syndrome.1 The urinalysis is significant for hematuria with RBC casts, and variable levels of proteinuria (typically, less than 3.5 g/day is seen in nephritic syndrome).1
Review of the Data
Nephrotic Syndromes
Minimal change disease. MCD is more common in children than adults, and only accounts for 10% to 15% of glomerular disease cases in adults.3 It is associated with Hodgkin’s lymphoma, NSAID use, and allergic conditions. There usually is an absence of hypertension (HTN). There are no glomerular basement membrane abnormalities seen on light microscopy. Electron microscopy shows effacement of podocytes. On urinalysis, oval fat bodies are seen, which are characteristic of heavy proteinuria. Complement levels are normal. Steroids are first-line treatment, but in adults with relapses or steroid resistance, immunosuppressive agents have also been used.2
Focal segment glomerlosclerosis.
FSGS is the most common primary glomerular disorder in the United States and is the most common cause of nephrotic syndrome among blacks.4,5 It is associated with HIV (collapsing variant), parvovirus B19, heroin use, sickle-cell disease, obesity, chronic vesicoureteral reflux, and HTN.4,6 Sclerosis of segmental glomeruli is seen on light microscopy.
Electron microscopy shows effacement of podocytes. Complement levels are normal. Treatment of primary idiopathic FSGS includes use of renin-angiotensin inhibitors and steroids.4 Immunosuppressives are reserved for relapses. Treatment of secondary FSGS involves identifying the underlying cause.
Membranous nephropathy. MN is twice as common in males as in females and is the most common cause of adult-onset idiopathic nephrotic syndrome, with the average presentation in the fifth or sixth decade of life.7,8 Aside from its idiopathic form, up to 25% of MN cases have an underlying disease process, such as solid organ tumors or hepatitis B.7,9 While nephrotic syndrome overall can increase the risk of thromboembolic complications, MN is the most common nephrotic disorder predisposing the development of renal vein thrombosis.7 Diffuse capillary wall thickening is seen on light microscopy, and electron microscopy shows sub-epithelial immune deposits. Complement levels are normal. Steroids and immunosuppressive agents are used for treatment.10
Membranoproliferative glomerulonephritis. MPGN is a nephrotic syndrome that is more common in children and young adults and can present with features of nephritic syndrome.1,11 It is associated with hepatitis C, SLE, and cryoglobulinemia.11 Light microscopy shows mesangial and endocapillary proliferation, as well as glomerular basement membrane thickening and splitting (“tram track” appearance). Electron microscopy shows subendothelial and dense deposits. It presents with reduced complement levels (C3 and C4).11 Treatment depends on the associated disease.
Nephritic Syndromes
Post-streptococcal glomerulonephritis. PSGN is seen in children and young adults and is associated with skin (impetigo) and throat infections (pharyngitis).12 Hematuria usually presents two to three weeks after a streptococcal infection. The urine is classically dark and smoky-colored. Levels of C3 and CH50 are low, but C4 levels are normal.1 In addition, there are positive antibody titers for ASO and anti-DNase. Light microscopy shows hypercellularity of glomeruli. Electron microscopy shows dome-shaped sub-endothelial deposits. Treatment is usually supportive.
IgA nephropathy. IgA nephropathy is the most common form of glomerular disease worldwide and the most common form of glomerular-related microscopic hematuria in all age groups.2,13 It occurs in all ages but more frequently in males.14 It occurs during or immediately after an upper respiratory infection. Light microscopy shows mesangial cell proliferation and crescentic GN. Electron microscopy shows immune deposits in the mesangium. Complement levels are normal. There has been no proven therapy, but ACE inhibitors (ACE-Is), angiotensin receptor blockers (ARBs), fish oil, steroids, and tonsillectomy have been used with some success.14 The clinical course of IgA nephropathy can be highly variable, with the potential for a benign course to rapidly progressive renal failure, with 15% to 40% of patients developing end-stage renal disease.14
Henoch-Schonlein purpura. HSP affects children more than adults and is the systemic form of IgA nephropathy.14 Most cases are idiopathic. Clinical
manifestations include: HTN; purpuric palpable rash on buttocks, ankles, and legs; bloody diarrhea with abdominal cramps; and pain in wrist, ankle, and knee joints.15 Light microscopy shows mesangial cell proliferation. Immune deposits in the mesangium are seen on electron microscopy. Complement levels are normal. Treatment is supportive.
Rapidly progressive glomerulonephritis types I, II, and III. RPGN represents a wide variety of disease states in which rapid progression to renal failure is seen within days to weeks.16 They are categorized into three sub-categories: I, II, and III.
Type I is an anti-GBM disease, an example being Goodpasture’s syndrome. This condition presents with hemoptysis, pulmonary infiltrates, and hematuria with RBC casts. Anti-GBM antibodies are classically found.1 Complement levels are normal and a linear immunofluorescence pattern is seen. Treatment is steroids, immunosuppressive agents, and plasmapheresis.17
Type II is an immune complex deposition disease, such as HSP, SLE, or post-streptococcal GN, in which granular complex deposits are seen. Treatment is directed toward treating the underlying cause.
Type III is pauci-immune (no immune deposits), showing necrotizing crescentic GN on biopsy, and is associated with a positive ANCA.1,18 They are associated with systemic small-vessel vasculitis, such as granulomatosis with polyangiitis (formerly known as Wegener’s granulomatosis), microscopic polyangiitis, and Churg-Strauss syndrome, or can be limited to renal involvement.1,18 Complement levels are normal. Treatment is steroids and immunosuppressive agents, such as cyclophosphamide.
A summary of the findings found in the glomerulonephritides and how complement levels are affected are found in Table 2 and Table 3, respectively.
Secondary Causes of Nephrotic Diseases
Diabetic nephropathy. Diabetic nephropathy is the single most common cause of progressive renal failure in the United States.3 Up to 50% of patients with diabetes present with diabetic nephropathy.19 Current recommendations are to screen yearly for microalbuminuria at the time of diagnosis.3 Treatment involves use of ACE-Is or ARBs to reduce proteinuria and slow the progression of renal disease.
HIV-associated nephropathy. HIV-associated nephropathy commonly presents as the collapsing variant of FSGS. However, it can present as other forms of glomerulopathy, such as MPGN or IgA nephropathy, as well as an immune complex GN with “lupus-like” features without evidence of SLE.19,20 Therefore, HIV nephropathy has now been categorized as a separate entity.3 ACE-Is, HAART therapy, and corticosteroids are the mainstays of treatment.
Amyloidosis. Renal involvement is seen in both primary (AL) and secondary (AA) amyloidosis. Eighty percent of patients with AL have renal disease, and 25% of these patients have nephrotic syndrome.16 Diagnosis is made with Congo Red stain, which shows fibrillary amyloid deposits within the mesangium and capillary walls. Treatment is directed at the underlying process.
Systemic lupus erythematosus. SLE is divided into six classes (I-VI) based on the involvement and severity of renal disease, and steroids and immunosuppressive agents are used for treatment, also based on the severity of the disease.21
Back to the Case
Our patient presented to the hospital with abdominal pain, low-grade fever, HTN, edema, hypoalbuminemia, and new-onset renal failure with gross hematuria and proteinuria. The presence of proteinuria and hypoalbuminemia, combined with peripheral and periorbital edema, suggests glomerular loss of albumin, such as in nephrotic syndrome. His renal failure in the setting of the sudden development of gross hematuria with flank pain is concerning for a renal vein thrombosis, and an abdominal magnetic resonance venography did in fact visualize a renal vein thrombosis.
He was admitted to the hospital and was started on therapeutic intravenous heparin, and bridged to warfarin. Subsequent renal biopsy confirmed the findings of membranous nephropathy, which was suspected due to his renal vein thrombosis. Therapy was initiated with corticosteroids after the biopsy, and he responded well. Because of his risk factors for further thromboembolic events, lifelong anticoagulation therapy was recommended.
Bottom Line
For patients with glomerular disease, differentiating between nephrotic and nephritic syndromes and understanding key clinical and laboratory differences can lead to easier identification and treatment.
Drs. Khan and Smith are assistant professors of medicine, and Dr. Ansari is associate division director, in the Division of Hospital Medicine at Loyola University Medical Center, Maywood, Ill.
References
- Donegio RGB, Salant DJ. Nephrology: Glomerular Diseases. In: ACP Medicine. Dale D, Federman D, eds. Available at: http://www.acpmedicine.com/acpmedicine/institutional/instHtmlReader.action?readerFlag=chapt&chapId=part10_ch05. Accessed Feb. 15, 2012.
- Orth SR, Ritz E. The nephrotic syndrome. N Engl J Med. 1998;338:1202-1211.
- Lewis JB, Neilson EG. Chapter 283. Glomerular Diseases. In: Longo DL, Fauci AS, Kasper DL, Hauser SL, Jameson JL, Loscalzo J, eds. Harrison’s Principles of Internal Medicine. 18th ed. New York: McGraw-Hill; 2012.
- D’Agati VD, Kaskel FJ, Falk RJ. Focal segmental glomerulosclerosis. N Engl J Med. 2011;365:2398-23411.
- Kitiyakara C, Eggers P, Kopp JB. Twenty-one-year trend in ESRD due to focal segmental glomerulosclerosis in the United States. Am J Kidney Dis. 2004;44:815-825.
- Balow JE. Nephropathy in the context of HIV infection. Kidney Int. 2005;67:1632-1633.
- Glassock RJ. Diagnosis and natural course of membranous nephropathy. Semin Nephrol. 2003;23:324-332.
- Nickolas TL, Radhakrishnan J, Appel GB. Hyperlipidemia and thrombotic complications in patients with membranous nephropathy. Semin Nephrol. 2003;23:406-411.
- Burstein DM, Korbet SM, Schwartz MM. Membranous glomerulonephritis and malignancy. Am J Kidney Dis. 1993;22:5-10.
- Hofstra JM, Wetzels JF. Management of patients with membranous nephropathy. Nephrol Dial Transplant. 2012;27:6-9.
- Alchi B, Jayne D. Membranoproliferative glomerulonephritis. Pediatr Nephrol. 2010;25:1409-1418.
- Eison TM, Ault BH, Jones DP, Chesney RW, Wyatt RJ. Post-streptococcal acute glomerulonephritis in children: clinical features and pathogenesis. Pediatr Nephrol. 2011;26:165-180.
- Cohen RA, Brown RS. Clinical practice. Microscopic hematuria. N Engl J Med. 2003;348:2330-2338.
- Donadio JV, Grande JP. IgA nephropathy. N Engl J Med. 2002;347:738-748.
- McCarthy HJ, Tizard EJ. Clinical practice: Diagnosis and management of henoch-schonlein purpura. Eur J Pediatr. 2010169:643-650.
- Appel GB, Radhakrishnan J. Cecil Medicine: Volume 1: Chapter 123: Glomerular Disorders and Nephrotic Syndromes. MD Consult Preview website. Available at: http://www.mdconsult.com/books/page.do?eid=4-u1.0-B978-1-4377-1604-7..00123-8&isbn=978-1-4377-1604-7&uniqId=313771243-2#4-u1.0-B978-1-4377-1604-7..00123-8. Accessed Feb. 16, 2012.
- Walters G, Willis NS, Craig JC. Interventions for renal vasculitis in adults. Cochrane Database Syst Rev. 2008;(3)(3):CD003232.
- Jennette JC. Rapidly progressive crescentic glomerulonephritis. Kidney Int. 2003;63:1164-1177.
- Falk, RJ: Medical Knowledge Self Assessment Program 14. Nephrology: 2006.
- Haas M, Kaul S, Eustace JA. HIV-associated immune complex glomerulonephritis with “lupus-like” features: a clinicopathologic study of 14 cases. Kidney Int. 2005;67:1381.
- Dooley MA, Hogan S, Jennette C, Falk R. Cyclophosphamide therapy for lupus nephritis: poor renal survival in black americans. glomerular disease collaborative network. Kidney Int. 1997;51:1188-1195.
Three Earn Master of Hospital Medicine Designation

—Joseph Ming-Wah Li, MD, SFHM, SHM president
If the SHM Award of Excellence winners are the MVPs of HM, then the specialty’s new Masters in Hospital Medicine are this year’s Hall of Famers.
SHM is proud to announce that Patrick J. Cawley, MD, MBA, MHM, Peter K. Lindenauer, MD, MSc, FACP, MHM, and Mark V. Williams, MD, FACP, MHM, have earned the Master in Hospital Medicine designation. They are the eighth, ninth, and 10th SHM members to receive the exclusive designation. In addition to being honored at HM12, they have earned the right to use the “MHM” credential.
“Hospitalists of all stripes owe our new Masters in Hospital Medicine a great debt for their commitment to the specialty, to their patients, and to healthcare,” says Joseph Ming Wah Li, MD, SFHM, SHM president. “Their innovation, leadership, and vision have helped grow the influence and credibility of the hospital medicine specialty.”
Dr. Cawley is a charter member and past president of SHM who has served on the Ethics and Public Policy committees. He is a frequent speaker at the society’s annual meetings. Dr. Cawley received his medical degree from Georgetown University School of Medicine and trained as a resident at Duke University Medical Center, where he also served as an assistant chief resident. He has been a contributing writer to The Hospitalist and several other journals and books.
Dr. Lindenauer is a board-certified internist and founding SHM board member. His research focuses on measuring the quality and outcomes of hospital care for patients with common medical conditions. He is supported by grants from the Agency for Healthcare Research and Quality and the NIH’s National Heart Lung and Blood Institute. His research has appeared in the New England Journal of Medicine, JAMA, Annals of Internal Medicine, Health Affairs, Medical Care, and other general internal medicine and subspecialty journals.
Dr. Williams is professor and chief of the Division of Hospital Medicine at Northwestern University Feinberg School of Medicine in Chicago. He is the principal investigator for SHM’s Project BOOST and is a former SHM president and board member. He has served in numerous leadership roles since the society’s establishment in 1996. In 1998, Dr. Williams established the first hospitalist program at a public hospital, Grady Memorial Hospital in Atlanta.
Drs. Cawley, Lindenauer, and Williams represent the third year of the Masters program. This marks the first year that nominations could be submitted to the SHM Masters Selection committee.
Joining the new masters in earning a new designation are 120 new Fellows in Hospital Medicine (FHM) and 52 Senior Fellows in Hospital Medicine (SFHM). The new members push the fellows program totals to 947 FHMs, 243 SFHMs, and 10 MHMs.
Brendon Shank is SHM’s associate vice president of communications.

—Joseph Ming-Wah Li, MD, SFHM, SHM president
If the SHM Award of Excellence winners are the MVPs of HM, then the specialty’s new Masters in Hospital Medicine are this year’s Hall of Famers.
SHM is proud to announce that Patrick J. Cawley, MD, MBA, MHM, Peter K. Lindenauer, MD, MSc, FACP, MHM, and Mark V. Williams, MD, FACP, MHM, have earned the Master in Hospital Medicine designation. They are the eighth, ninth, and 10th SHM members to receive the exclusive designation. In addition to being honored at HM12, they have earned the right to use the “MHM” credential.
“Hospitalists of all stripes owe our new Masters in Hospital Medicine a great debt for their commitment to the specialty, to their patients, and to healthcare,” says Joseph Ming Wah Li, MD, SFHM, SHM president. “Their innovation, leadership, and vision have helped grow the influence and credibility of the hospital medicine specialty.”
Dr. Cawley is a charter member and past president of SHM who has served on the Ethics and Public Policy committees. He is a frequent speaker at the society’s annual meetings. Dr. Cawley received his medical degree from Georgetown University School of Medicine and trained as a resident at Duke University Medical Center, where he also served as an assistant chief resident. He has been a contributing writer to The Hospitalist and several other journals and books.
Dr. Lindenauer is a board-certified internist and founding SHM board member. His research focuses on measuring the quality and outcomes of hospital care for patients with common medical conditions. He is supported by grants from the Agency for Healthcare Research and Quality and the NIH’s National Heart Lung and Blood Institute. His research has appeared in the New England Journal of Medicine, JAMA, Annals of Internal Medicine, Health Affairs, Medical Care, and other general internal medicine and subspecialty journals.
Dr. Williams is professor and chief of the Division of Hospital Medicine at Northwestern University Feinberg School of Medicine in Chicago. He is the principal investigator for SHM’s Project BOOST and is a former SHM president and board member. He has served in numerous leadership roles since the society’s establishment in 1996. In 1998, Dr. Williams established the first hospitalist program at a public hospital, Grady Memorial Hospital in Atlanta.
Drs. Cawley, Lindenauer, and Williams represent the third year of the Masters program. This marks the first year that nominations could be submitted to the SHM Masters Selection committee.
Joining the new masters in earning a new designation are 120 new Fellows in Hospital Medicine (FHM) and 52 Senior Fellows in Hospital Medicine (SFHM). The new members push the fellows program totals to 947 FHMs, 243 SFHMs, and 10 MHMs.
Brendon Shank is SHM’s associate vice president of communications.

—Joseph Ming-Wah Li, MD, SFHM, SHM president
If the SHM Award of Excellence winners are the MVPs of HM, then the specialty’s new Masters in Hospital Medicine are this year’s Hall of Famers.
SHM is proud to announce that Patrick J. Cawley, MD, MBA, MHM, Peter K. Lindenauer, MD, MSc, FACP, MHM, and Mark V. Williams, MD, FACP, MHM, have earned the Master in Hospital Medicine designation. They are the eighth, ninth, and 10th SHM members to receive the exclusive designation. In addition to being honored at HM12, they have earned the right to use the “MHM” credential.
“Hospitalists of all stripes owe our new Masters in Hospital Medicine a great debt for their commitment to the specialty, to their patients, and to healthcare,” says Joseph Ming Wah Li, MD, SFHM, SHM president. “Their innovation, leadership, and vision have helped grow the influence and credibility of the hospital medicine specialty.”
Dr. Cawley is a charter member and past president of SHM who has served on the Ethics and Public Policy committees. He is a frequent speaker at the society’s annual meetings. Dr. Cawley received his medical degree from Georgetown University School of Medicine and trained as a resident at Duke University Medical Center, where he also served as an assistant chief resident. He has been a contributing writer to The Hospitalist and several other journals and books.
Dr. Lindenauer is a board-certified internist and founding SHM board member. His research focuses on measuring the quality and outcomes of hospital care for patients with common medical conditions. He is supported by grants from the Agency for Healthcare Research and Quality and the NIH’s National Heart Lung and Blood Institute. His research has appeared in the New England Journal of Medicine, JAMA, Annals of Internal Medicine, Health Affairs, Medical Care, and other general internal medicine and subspecialty journals.
Dr. Williams is professor and chief of the Division of Hospital Medicine at Northwestern University Feinberg School of Medicine in Chicago. He is the principal investigator for SHM’s Project BOOST and is a former SHM president and board member. He has served in numerous leadership roles since the society’s establishment in 1996. In 1998, Dr. Williams established the first hospitalist program at a public hospital, Grady Memorial Hospital in Atlanta.
Drs. Cawley, Lindenauer, and Williams represent the third year of the Masters program. This marks the first year that nominations could be submitted to the SHM Masters Selection committee.
Joining the new masters in earning a new designation are 120 new Fellows in Hospital Medicine (FHM) and 52 Senior Fellows in Hospital Medicine (SFHM). The new members push the fellows program totals to 947 FHMs, 243 SFHMs, and 10 MHMs.
Brendon Shank is SHM’s associate vice president of communications.