User login
Q.Does cervical dysplasia raise the risk of preterm birth?
Expert Commentary
Preterm birth and cervical dysplasia share many risk factors, most of which are the progeny of low socioeconomic status. It is not surprising, therefore, that cervical dysplasia is a risk factor for pre-term birth independent of the treatment modality chosen for the precancerous condition. This large cohort study linking outpatient gynecologic records with childbirths from Australia found exactly that. It is the largest study so far to focus on pregnancy outcomes in women following diagnosis and treatment of dysplasia. Frustrating to both the obstetrician and the gynecologist is the fact that smoking is the only readily modifiable risk factor for preterm birth or cervical dysplasia.
Ablative procedures produce better pregnancy outcomes than excision
Beyond epidemiology, this paper bears an important message for clinicians and patients: Procedures that remove portions of the cervix, such as LEEP, diathermy, and cone biopsy, raise the risk of subsequent preterm birth, compared with less destructive ablative procedures such as laser ablation (as reported in the Up-date on Cervical Disease, by Thomas C. Wright, MD, in the March issue of this journal). This was also demonstrated in a large review of the subject.1 Therefore, for an optimal obstetrical outcome, ablative procedures are preferable to excisional ones in women who have not yet completed childbearing. Given that success rates are only slightly lower for ablative procedures than for destructive ones, a clinician can recommend ablation without fear of dysplasia progressing to invasive cancer.
Widespread HPV vaccination could help reduce preterm birth rate
This study highlights how a systematic program of human papillomavirus (HPV) vaccination in adolescents (male and female) before their coital debut could reduce the rate of preterm birth by eliminating the need for women to undergo excisional treatment for cervical dysplasia. The possibility for such improvement in birth outcomes should motivate those of us working to prevent preterm birth to advocate for societal investment in this approach. It also might alleviate concerns that HPV vaccination has the potential to disrupt family life by encouraging promiscuity. How can anyone argue against a program that will prevent cancer and preterm birth?
1. Kyrgiou M, Koliopoulos G, Martin-Hirsch P, Arbyn M, Prendiville W, Paraskevaidis E. Obstetric outcomes after conservative treatment for intraepithelial or early invasive lesions: a systematic review and meta-analysis of the literature. Lancet. 2006;367:489-498.
Expert Commentary
Preterm birth and cervical dysplasia share many risk factors, most of which are the progeny of low socioeconomic status. It is not surprising, therefore, that cervical dysplasia is a risk factor for pre-term birth independent of the treatment modality chosen for the precancerous condition. This large cohort study linking outpatient gynecologic records with childbirths from Australia found exactly that. It is the largest study so far to focus on pregnancy outcomes in women following diagnosis and treatment of dysplasia. Frustrating to both the obstetrician and the gynecologist is the fact that smoking is the only readily modifiable risk factor for preterm birth or cervical dysplasia.
Ablative procedures produce better pregnancy outcomes than excision
Beyond epidemiology, this paper bears an important message for clinicians and patients: Procedures that remove portions of the cervix, such as LEEP, diathermy, and cone biopsy, raise the risk of subsequent preterm birth, compared with less destructive ablative procedures such as laser ablation (as reported in the Up-date on Cervical Disease, by Thomas C. Wright, MD, in the March issue of this journal). This was also demonstrated in a large review of the subject.1 Therefore, for an optimal obstetrical outcome, ablative procedures are preferable to excisional ones in women who have not yet completed childbearing. Given that success rates are only slightly lower for ablative procedures than for destructive ones, a clinician can recommend ablation without fear of dysplasia progressing to invasive cancer.
Widespread HPV vaccination could help reduce preterm birth rate
This study highlights how a systematic program of human papillomavirus (HPV) vaccination in adolescents (male and female) before their coital debut could reduce the rate of preterm birth by eliminating the need for women to undergo excisional treatment for cervical dysplasia. The possibility for such improvement in birth outcomes should motivate those of us working to prevent preterm birth to advocate for societal investment in this approach. It also might alleviate concerns that HPV vaccination has the potential to disrupt family life by encouraging promiscuity. How can anyone argue against a program that will prevent cancer and preterm birth?
Expert Commentary
Preterm birth and cervical dysplasia share many risk factors, most of which are the progeny of low socioeconomic status. It is not surprising, therefore, that cervical dysplasia is a risk factor for pre-term birth independent of the treatment modality chosen for the precancerous condition. This large cohort study linking outpatient gynecologic records with childbirths from Australia found exactly that. It is the largest study so far to focus on pregnancy outcomes in women following diagnosis and treatment of dysplasia. Frustrating to both the obstetrician and the gynecologist is the fact that smoking is the only readily modifiable risk factor for preterm birth or cervical dysplasia.
Ablative procedures produce better pregnancy outcomes than excision
Beyond epidemiology, this paper bears an important message for clinicians and patients: Procedures that remove portions of the cervix, such as LEEP, diathermy, and cone biopsy, raise the risk of subsequent preterm birth, compared with less destructive ablative procedures such as laser ablation (as reported in the Up-date on Cervical Disease, by Thomas C. Wright, MD, in the March issue of this journal). This was also demonstrated in a large review of the subject.1 Therefore, for an optimal obstetrical outcome, ablative procedures are preferable to excisional ones in women who have not yet completed childbearing. Given that success rates are only slightly lower for ablative procedures than for destructive ones, a clinician can recommend ablation without fear of dysplasia progressing to invasive cancer.
Widespread HPV vaccination could help reduce preterm birth rate
This study highlights how a systematic program of human papillomavirus (HPV) vaccination in adolescents (male and female) before their coital debut could reduce the rate of preterm birth by eliminating the need for women to undergo excisional treatment for cervical dysplasia. The possibility for such improvement in birth outcomes should motivate those of us working to prevent preterm birth to advocate for societal investment in this approach. It also might alleviate concerns that HPV vaccination has the potential to disrupt family life by encouraging promiscuity. How can anyone argue against a program that will prevent cancer and preterm birth?
1. Kyrgiou M, Koliopoulos G, Martin-Hirsch P, Arbyn M, Prendiville W, Paraskevaidis E. Obstetric outcomes after conservative treatment for intraepithelial or early invasive lesions: a systematic review and meta-analysis of the literature. Lancet. 2006;367:489-498.
1. Kyrgiou M, Koliopoulos G, Martin-Hirsch P, Arbyn M, Prendiville W, Paraskevaidis E. Obstetric outcomes after conservative treatment for intraepithelial or early invasive lesions: a systematic review and meta-analysis of the literature. Lancet. 2006;367:489-498.
How patients benefit when you add an HPV test to screening for cervical Ca
CASE: Erratic screening history with at least 1 abnormality
G.A., 40, vaguely remembers having at least 1 abnormal Pap test about 20 years ago. She is not sure exactly what the result was, but does recall that she underwent colposcopy. Her physician at the time told her that nothing important was detected and encouraged her to get “regular Pap tests” in the future. She followed this advice for several years, but her Pap tests became much less frequent after her 2 children were born. Today, she reports that her last Pap test was at least 5 years ago, but she does not remember exactly when. She also remembers being notified that she needed to repeat it, but is not sure why. Her records are unavailable because she has moved a lot and cannot remember the name of the doctor who saw her.
Today, G.A. is screened with both a Pap test and a human papillomavirus (HPV) test for high-risk viral types. The physician outlines the benefits of doing both tests and explains the “commonness” of HPV, offering reassuring facts about its natural history to “soften the concern” in the event she is found to be positive.
The Pap test comes back as “negative for intraepithelial neoplasia or malignancy” (ie, normal), but her HPV test is positive.
What questions is G.A. likely to raise? How can her risk of cervical cancer be quantified? And how should she be managed?
Why do an HPV test with the Pap?
G.A.’s situation is not unusual. Many women provide a vague history that includes 1 or more abnormal Pap tests in the distant past, with “probably normal” results on infrequent, irregular screening in more recent years.
It has been estimated that annual lifetime screening with a Pap test sensitivity of 70% for cervical intraepithelial neoplasia (CIN) 2 or 3 reduces the lifetime risk of cervical cancer by 93%. That means that even diligent lifetime screening will leave some women unprotected.1
The risk increases for women who are not screened regularly, especially those with a history of an abnormal Pap test. Approximately 10% of cervical cancers occur in women who have been screened in the past but not within the past 5 years.2
Pap test alone has poor sensitivity
A number of meta-analyses have documented the sensitivity of the conventional Pap smear for the detection of CIN 2,3 to be between 51% and 67%.3,4 And although liquid-based cytology offers many advantages, early reports of improved sensitivity over conventional cytology were not substantiated in a 2006 meta-analysis of a large number of studies (as reported in Update on Cervical Disease, by Thomas C. Wright, MD, in the March issue of OBG Management).5
Despite the low sensitivity of cervical cytology, Pap test screening has been extremely successful in detecting precancerous cervical changes and allowing timely treatment. The success is directly attributable to repeated screening of women during the relatively slow progression from initial HPV infection to CIN 3 (typically, about 10 years) and from CIN 3 to cancer (typically, 10 or more years).6 Poor sensitivity raises concern, however, when screening attendance is not ideal.
Together, the tests lower the risk of missing CIN or cancer to 1 in 1,000
Surely, G.A. would benefit by having the most reassuring screening available. Guidelines from the American Cancer Society (2002) and 2 practice bulletins from the American College of Obstetricians and Gynecologists (ACOG) (2003, 2005) recommend as 1 of 2 options screening women age 30 and over with both the Pap test and the HPV test for high-risk types. 7-9
A 2005 ACOG practice bulletin on HPV10 noted the reassurance offered by combined testing and observed that, based on Level A evidence, “HPV testing is more sensitive than cervical cytology in detecting CIN 2 and CIN 3, [so] women with concurrent negative test results can be reassured that their risk of unidentified CIN 2, CIN 3, or cervical cancer is approximately 1 in 1,000.”
Most women would benefit from a screening test that provides a reassurance of 1 in 1,000. Given G.A.’s infrequent screening history, previous abnormal cervical cytology, and unknown result on her last screen over 5 years ago, using the most reassuring combination of tests would seem to be imperative.
Likely questions
Because HPV testing plays an ever-increasing role in cervical screening, it presents a new set of educational challenges.6 Until recently, clinicians generally avoided discussing the cause of abnormal cervical cytology, CIN, and cervical cancer. However, when a woman is tested for high-risk HPV along with the Pap test in primary screening, the subject can no longer be skirted. Already the HPV vaccine and widespread use of the Web by patients have changed the information base on HPV. Education can begin at the time of the Pap and HPV tests, need not be extensive, and often can deflect undue anxiety and many of the patient’s questions about a positive result.
G.A. has been married for nearly 22 years. Her husband has been her only partner, but he was sexually active before they were married. She is naturally concerned about how long she has had HPV and what this means for her relationship. Her questions are universal: How and when did I become infected? What is my risk and that of my partner? How will I be managed? Will I always have HPV?
These questions may seem daunting, but the answers can be kept simple and short and still provide enough information to be reassuring and to prepare the patient for a possible positive test result.
Quantify risk before you select management
Most women with normal cytology but a first-time positive HPV test do not have CIN 2,3 or greater.11-14 A National Cancer Institute prospective 10-year follow-up of more than 20,000 women screened at enrollment with both the HPV test and cytology demonstrated that only 4.4% of the HPV-positive, Pap-negative women had CIN 3 or cancer detected over the following 3 to 5 years, and only 7% did by 10 years.13,14 These rates are similar to those found in other studies and are about half the risk represented by a Pap result of atypical squamous cells of undetermined significance (ASC-US).15
Therefore, because of the low immediate risk for high-grade cervical neoplasia and the extremely rare occurrence of cervical cancer in this setting, immediate referral to colposcopy is not recommended in routine cases (FIGURE 1).9,15 Instead, repeating Pap and HPV testing in 6 to 12 months yields a more accurate picture of risk by determining whether the HPV is only transient or is persistent. Only persistent HPV is associated with significant risk for CIN 2,3. Therefore, repeating the tests, rather than sending the patient to immediate colposcopy, allows the 45% to 70% of HPV infections destined to be transient to resolve spontaneously, but will still detect most significant lesions within a reasonable period of time.15
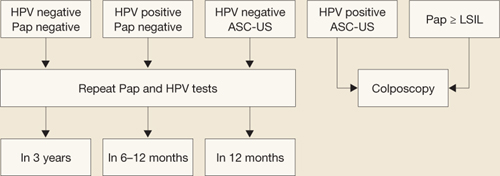
FIGURE 1 How to interpret the HPV test and cytology combined: NCI–ASCCP Interim Guidance
SOURCE: National Cancer Institute and the American Society for Colposcopy and Cervical Pathology. Adapted from Wright et al15
ASC-US=atypical squamous cells of undetermined significance; HPV=human papillomavirus; LSIL=low-grade squamous intraepithelial lesions.If G.A. is managed in this way and, at the 6- and 12-month repeats of both tests, has a positive HPV test (regardless of the cytology finding) or any abnormal Pap test result other than HPV-negative/ASC-US, she should undergo colposcopy.15 A test designated as HPV-negative/ASC-US can be managed by repeat testing in 12 months.
Remember: Even though a repeat positive HPV test increases the patient’s risk of CIN 2,3 significantly, it is specifically not recommended to treat the cervix solely on the basis of a persistently positive HPV test without evidence of CIN or cervical cancer.
This patient warrants a different approach—here’s why
Although repeat Pap and HPV testing in 6 to 12 months is the standard recommendation for women with a normal Pap test and positive HPV results, extenuating circumstances may exist. Clinical judgment always trumps routine recommendations in these cases.
The progression of CIN 3 to cervical cancer is usually a slow process that occurs over many years.6 Therefore, delaying colposcopy for 6 to 12 months will probably not increase risk significantly even if a high-grade lesion is already present. But G.A.’s case involves a number of variables that may increase her risk enough to justify immediate colposcopy:
- an abnormal Pap test more than 20 years ago
- a history of irregular screening
- no screening within the past 5 years until the current testing
- concern that her last Pap result was either minimally abnormal or of limited quality.
Lack of access to any earlier records further limits the physician’s ability to adequately judge G.A.’s risk. Because of these concerns, the physician asks G.A. to come in for colposcopy, at which time a 2-quadrant CIN 3 lesion is found (FIGURE 2). The patient is treated by loop electrosurgical excision procedure and has normal cytology and a negative HPV test result on follow-up.
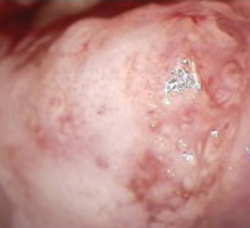
FIGURE 2 Two-quadrant CIN 2,3
High-grade lesions begin as a monoclonal cell change that enlarges centrifugally. Hence, increasing size is suspect for increasing duration of presence and increasing risk for cancer, because risk of invasion is proportional to lesion size.
Did you read Dr. Thomas C. Wright’s Update on Cervical Disease in the March Issue of OBG Management? If not, visit www.obgmanagement.com and follow the PAST ISSUES link on the navigation bar to the March issue.
The author reports no financial relationships relevant to this article.
1. Screening for squamous cervical cancer: duration of low risk after negative results of cervical cytology and its implication for screening policies. IARC Working Group on Evaluation of Cervical Cancer Screening Programmes. Br Med J (J Clin Res Ed). 1986;293:659-664.
2. Sung HY, Kearney KA, Miller M, Kinney W, Sawaya GF, Hiatt RA. Papanicolaou smear history and diagnosis of invasive cervical carcinoma among members of a large prepaid health plan. Cancer. 2000;88:2283-2289.
3. Nanda K, McCrory DC, Myers ER, et al. Accuracy of the Papanicolaou test in screening for and follow-up of cervical cytologic abnormalities: a systematic review. Ann Intern Med. 2000;132:810-819.
4. Fahey MT, Irwig L, Macaskill P. Meta-analysis of Pap test accuracy. Am J Epidemiol. 1995;141:680-689.
5. Davey E, Barratt A, Irwig L, et al. Effect of study design and quality on unsatisfactory rates, cytology classifications, and accuracy in liquid-based versus conventional cervical cytology: a systematic review. Lancet. 2006;367:122-132.
6. Kitchener HC, Castle PE, Cox JT. Chapter 7: Achievements and limitations of cervical cytology screening. Vaccine. 2006;24 Suppl 3:S63-S70.
7. Saslow D, Runowicz CD, Solomon D, et al. For the American Cancer Society. American Cancer Society guideline for the early detection of cervical neoplasia and cancer. CA Cancer J Clin. 2002;52:342-362.
8. Cervical cytology screening. American College of Obstetricians and Gynecologists Practice Bulletin #45. Washington, DC: ACOG; 2003.
9. Management of abnormal cervical cytology and histology. Clinical management guidelines for the Obstetrician and Gynecologist. American College of Obstetricians and Gynecologists Practice Bulletin #66. Washington, DC: ACOG; 2005.
10. Human papillomavirus. American College of Obstetricians and Gynecologists Practice Bulletin #61. Washington, DC: ACOG; 2005.
11. Clavel C, Masure M, Bory JP, et al. Human papillomavirus testing in primary screening for the detection of high-grade cervical lesions: a study of 7,932 women. Br J Cancer. 2001;84:1616-1623.
12. Cuzick J, Szarewski A, Cubie H, et al. Management of women who test positive for high-risk types of human papillomavirus: the HART study. Lancet. 2003;362:1871-1876.
13. Sherman ME, Lorincz AT, Scott DR, et al. Baseline cytology, human papillomavirus testing, and risk for cervical neoplasia: 10-year cohort analysis. J Natl Cancer Inst. 2003;95:46-52.
14. Khan MJ, Castle PE, Lorincz AT, et al. The elevated 10-year risk of cervical precancer and cancer in women with human papillomavirus (HPV) type 16 or 18 and the possible utility of type-specific HPV testing in clinical practice. J Natl Cancer Inst. 2005;97:1072-1079.
15. Wright TC, Jr, Schiffman M, Solomon D, et al. Interim guidance for the use of human papillomavirus DNA testing as an adjunct to cervical cytology for screening. Obstet Gynecol. 2004;103:304-309.
CASE: Erratic screening history with at least 1 abnormality
G.A., 40, vaguely remembers having at least 1 abnormal Pap test about 20 years ago. She is not sure exactly what the result was, but does recall that she underwent colposcopy. Her physician at the time told her that nothing important was detected and encouraged her to get “regular Pap tests” in the future. She followed this advice for several years, but her Pap tests became much less frequent after her 2 children were born. Today, she reports that her last Pap test was at least 5 years ago, but she does not remember exactly when. She also remembers being notified that she needed to repeat it, but is not sure why. Her records are unavailable because she has moved a lot and cannot remember the name of the doctor who saw her.
Today, G.A. is screened with both a Pap test and a human papillomavirus (HPV) test for high-risk viral types. The physician outlines the benefits of doing both tests and explains the “commonness” of HPV, offering reassuring facts about its natural history to “soften the concern” in the event she is found to be positive.
The Pap test comes back as “negative for intraepithelial neoplasia or malignancy” (ie, normal), but her HPV test is positive.
What questions is G.A. likely to raise? How can her risk of cervical cancer be quantified? And how should she be managed?
Why do an HPV test with the Pap?
G.A.’s situation is not unusual. Many women provide a vague history that includes 1 or more abnormal Pap tests in the distant past, with “probably normal” results on infrequent, irregular screening in more recent years.
It has been estimated that annual lifetime screening with a Pap test sensitivity of 70% for cervical intraepithelial neoplasia (CIN) 2 or 3 reduces the lifetime risk of cervical cancer by 93%. That means that even diligent lifetime screening will leave some women unprotected.1
The risk increases for women who are not screened regularly, especially those with a history of an abnormal Pap test. Approximately 10% of cervical cancers occur in women who have been screened in the past but not within the past 5 years.2
Pap test alone has poor sensitivity
A number of meta-analyses have documented the sensitivity of the conventional Pap smear for the detection of CIN 2,3 to be between 51% and 67%.3,4 And although liquid-based cytology offers many advantages, early reports of improved sensitivity over conventional cytology were not substantiated in a 2006 meta-analysis of a large number of studies (as reported in Update on Cervical Disease, by Thomas C. Wright, MD, in the March issue of OBG Management).5
Despite the low sensitivity of cervical cytology, Pap test screening has been extremely successful in detecting precancerous cervical changes and allowing timely treatment. The success is directly attributable to repeated screening of women during the relatively slow progression from initial HPV infection to CIN 3 (typically, about 10 years) and from CIN 3 to cancer (typically, 10 or more years).6 Poor sensitivity raises concern, however, when screening attendance is not ideal.
Together, the tests lower the risk of missing CIN or cancer to 1 in 1,000
Surely, G.A. would benefit by having the most reassuring screening available. Guidelines from the American Cancer Society (2002) and 2 practice bulletins from the American College of Obstetricians and Gynecologists (ACOG) (2003, 2005) recommend as 1 of 2 options screening women age 30 and over with both the Pap test and the HPV test for high-risk types. 7-9
A 2005 ACOG practice bulletin on HPV10 noted the reassurance offered by combined testing and observed that, based on Level A evidence, “HPV testing is more sensitive than cervical cytology in detecting CIN 2 and CIN 3, [so] women with concurrent negative test results can be reassured that their risk of unidentified CIN 2, CIN 3, or cervical cancer is approximately 1 in 1,000.”
Most women would benefit from a screening test that provides a reassurance of 1 in 1,000. Given G.A.’s infrequent screening history, previous abnormal cervical cytology, and unknown result on her last screen over 5 years ago, using the most reassuring combination of tests would seem to be imperative.
Likely questions
Because HPV testing plays an ever-increasing role in cervical screening, it presents a new set of educational challenges.6 Until recently, clinicians generally avoided discussing the cause of abnormal cervical cytology, CIN, and cervical cancer. However, when a woman is tested for high-risk HPV along with the Pap test in primary screening, the subject can no longer be skirted. Already the HPV vaccine and widespread use of the Web by patients have changed the information base on HPV. Education can begin at the time of the Pap and HPV tests, need not be extensive, and often can deflect undue anxiety and many of the patient’s questions about a positive result.
G.A. has been married for nearly 22 years. Her husband has been her only partner, but he was sexually active before they were married. She is naturally concerned about how long she has had HPV and what this means for her relationship. Her questions are universal: How and when did I become infected? What is my risk and that of my partner? How will I be managed? Will I always have HPV?
These questions may seem daunting, but the answers can be kept simple and short and still provide enough information to be reassuring and to prepare the patient for a possible positive test result.
Quantify risk before you select management
Most women with normal cytology but a first-time positive HPV test do not have CIN 2,3 or greater.11-14 A National Cancer Institute prospective 10-year follow-up of more than 20,000 women screened at enrollment with both the HPV test and cytology demonstrated that only 4.4% of the HPV-positive, Pap-negative women had CIN 3 or cancer detected over the following 3 to 5 years, and only 7% did by 10 years.13,14 These rates are similar to those found in other studies and are about half the risk represented by a Pap result of atypical squamous cells of undetermined significance (ASC-US).15
Therefore, because of the low immediate risk for high-grade cervical neoplasia and the extremely rare occurrence of cervical cancer in this setting, immediate referral to colposcopy is not recommended in routine cases (FIGURE 1).9,15 Instead, repeating Pap and HPV testing in 6 to 12 months yields a more accurate picture of risk by determining whether the HPV is only transient or is persistent. Only persistent HPV is associated with significant risk for CIN 2,3. Therefore, repeating the tests, rather than sending the patient to immediate colposcopy, allows the 45% to 70% of HPV infections destined to be transient to resolve spontaneously, but will still detect most significant lesions within a reasonable period of time.15

FIGURE 1 How to interpret the HPV test and cytology combined: NCI–ASCCP Interim Guidance
SOURCE: National Cancer Institute and the American Society for Colposcopy and Cervical Pathology. Adapted from Wright et al15
ASC-US=atypical squamous cells of undetermined significance; HPV=human papillomavirus; LSIL=low-grade squamous intraepithelial lesions.If G.A. is managed in this way and, at the 6- and 12-month repeats of both tests, has a positive HPV test (regardless of the cytology finding) or any abnormal Pap test result other than HPV-negative/ASC-US, she should undergo colposcopy.15 A test designated as HPV-negative/ASC-US can be managed by repeat testing in 12 months.
Remember: Even though a repeat positive HPV test increases the patient’s risk of CIN 2,3 significantly, it is specifically not recommended to treat the cervix solely on the basis of a persistently positive HPV test without evidence of CIN or cervical cancer.
This patient warrants a different approach—here’s why
Although repeat Pap and HPV testing in 6 to 12 months is the standard recommendation for women with a normal Pap test and positive HPV results, extenuating circumstances may exist. Clinical judgment always trumps routine recommendations in these cases.
The progression of CIN 3 to cervical cancer is usually a slow process that occurs over many years.6 Therefore, delaying colposcopy for 6 to 12 months will probably not increase risk significantly even if a high-grade lesion is already present. But G.A.’s case involves a number of variables that may increase her risk enough to justify immediate colposcopy:
- an abnormal Pap test more than 20 years ago
- a history of irregular screening
- no screening within the past 5 years until the current testing
- concern that her last Pap result was either minimally abnormal or of limited quality.
Lack of access to any earlier records further limits the physician’s ability to adequately judge G.A.’s risk. Because of these concerns, the physician asks G.A. to come in for colposcopy, at which time a 2-quadrant CIN 3 lesion is found (FIGURE 2). The patient is treated by loop electrosurgical excision procedure and has normal cytology and a negative HPV test result on follow-up.

FIGURE 2 Two-quadrant CIN 2,3
High-grade lesions begin as a monoclonal cell change that enlarges centrifugally. Hence, increasing size is suspect for increasing duration of presence and increasing risk for cancer, because risk of invasion is proportional to lesion size.
Did you read Dr. Thomas C. Wright’s Update on Cervical Disease in the March Issue of OBG Management? If not, visit www.obgmanagement.com and follow the PAST ISSUES link on the navigation bar to the March issue.
The author reports no financial relationships relevant to this article.
CASE: Erratic screening history with at least 1 abnormality
G.A., 40, vaguely remembers having at least 1 abnormal Pap test about 20 years ago. She is not sure exactly what the result was, but does recall that she underwent colposcopy. Her physician at the time told her that nothing important was detected and encouraged her to get “regular Pap tests” in the future. She followed this advice for several years, but her Pap tests became much less frequent after her 2 children were born. Today, she reports that her last Pap test was at least 5 years ago, but she does not remember exactly when. She also remembers being notified that she needed to repeat it, but is not sure why. Her records are unavailable because she has moved a lot and cannot remember the name of the doctor who saw her.
Today, G.A. is screened with both a Pap test and a human papillomavirus (HPV) test for high-risk viral types. The physician outlines the benefits of doing both tests and explains the “commonness” of HPV, offering reassuring facts about its natural history to “soften the concern” in the event she is found to be positive.
The Pap test comes back as “negative for intraepithelial neoplasia or malignancy” (ie, normal), but her HPV test is positive.
What questions is G.A. likely to raise? How can her risk of cervical cancer be quantified? And how should she be managed?
Why do an HPV test with the Pap?
G.A.’s situation is not unusual. Many women provide a vague history that includes 1 or more abnormal Pap tests in the distant past, with “probably normal” results on infrequent, irregular screening in more recent years.
It has been estimated that annual lifetime screening with a Pap test sensitivity of 70% for cervical intraepithelial neoplasia (CIN) 2 or 3 reduces the lifetime risk of cervical cancer by 93%. That means that even diligent lifetime screening will leave some women unprotected.1
The risk increases for women who are not screened regularly, especially those with a history of an abnormal Pap test. Approximately 10% of cervical cancers occur in women who have been screened in the past but not within the past 5 years.2
Pap test alone has poor sensitivity
A number of meta-analyses have documented the sensitivity of the conventional Pap smear for the detection of CIN 2,3 to be between 51% and 67%.3,4 And although liquid-based cytology offers many advantages, early reports of improved sensitivity over conventional cytology were not substantiated in a 2006 meta-analysis of a large number of studies (as reported in Update on Cervical Disease, by Thomas C. Wright, MD, in the March issue of OBG Management).5
Despite the low sensitivity of cervical cytology, Pap test screening has been extremely successful in detecting precancerous cervical changes and allowing timely treatment. The success is directly attributable to repeated screening of women during the relatively slow progression from initial HPV infection to CIN 3 (typically, about 10 years) and from CIN 3 to cancer (typically, 10 or more years).6 Poor sensitivity raises concern, however, when screening attendance is not ideal.
Together, the tests lower the risk of missing CIN or cancer to 1 in 1,000
Surely, G.A. would benefit by having the most reassuring screening available. Guidelines from the American Cancer Society (2002) and 2 practice bulletins from the American College of Obstetricians and Gynecologists (ACOG) (2003, 2005) recommend as 1 of 2 options screening women age 30 and over with both the Pap test and the HPV test for high-risk types. 7-9
A 2005 ACOG practice bulletin on HPV10 noted the reassurance offered by combined testing and observed that, based on Level A evidence, “HPV testing is more sensitive than cervical cytology in detecting CIN 2 and CIN 3, [so] women with concurrent negative test results can be reassured that their risk of unidentified CIN 2, CIN 3, or cervical cancer is approximately 1 in 1,000.”
Most women would benefit from a screening test that provides a reassurance of 1 in 1,000. Given G.A.’s infrequent screening history, previous abnormal cervical cytology, and unknown result on her last screen over 5 years ago, using the most reassuring combination of tests would seem to be imperative.
Likely questions
Because HPV testing plays an ever-increasing role in cervical screening, it presents a new set of educational challenges.6 Until recently, clinicians generally avoided discussing the cause of abnormal cervical cytology, CIN, and cervical cancer. However, when a woman is tested for high-risk HPV along with the Pap test in primary screening, the subject can no longer be skirted. Already the HPV vaccine and widespread use of the Web by patients have changed the information base on HPV. Education can begin at the time of the Pap and HPV tests, need not be extensive, and often can deflect undue anxiety and many of the patient’s questions about a positive result.
G.A. has been married for nearly 22 years. Her husband has been her only partner, but he was sexually active before they were married. She is naturally concerned about how long she has had HPV and what this means for her relationship. Her questions are universal: How and when did I become infected? What is my risk and that of my partner? How will I be managed? Will I always have HPV?
These questions may seem daunting, but the answers can be kept simple and short and still provide enough information to be reassuring and to prepare the patient for a possible positive test result.
Quantify risk before you select management
Most women with normal cytology but a first-time positive HPV test do not have CIN 2,3 or greater.11-14 A National Cancer Institute prospective 10-year follow-up of more than 20,000 women screened at enrollment with both the HPV test and cytology demonstrated that only 4.4% of the HPV-positive, Pap-negative women had CIN 3 or cancer detected over the following 3 to 5 years, and only 7% did by 10 years.13,14 These rates are similar to those found in other studies and are about half the risk represented by a Pap result of atypical squamous cells of undetermined significance (ASC-US).15
Therefore, because of the low immediate risk for high-grade cervical neoplasia and the extremely rare occurrence of cervical cancer in this setting, immediate referral to colposcopy is not recommended in routine cases (FIGURE 1).9,15 Instead, repeating Pap and HPV testing in 6 to 12 months yields a more accurate picture of risk by determining whether the HPV is only transient or is persistent. Only persistent HPV is associated with significant risk for CIN 2,3. Therefore, repeating the tests, rather than sending the patient to immediate colposcopy, allows the 45% to 70% of HPV infections destined to be transient to resolve spontaneously, but will still detect most significant lesions within a reasonable period of time.15

FIGURE 1 How to interpret the HPV test and cytology combined: NCI–ASCCP Interim Guidance
SOURCE: National Cancer Institute and the American Society for Colposcopy and Cervical Pathology. Adapted from Wright et al15
ASC-US=atypical squamous cells of undetermined significance; HPV=human papillomavirus; LSIL=low-grade squamous intraepithelial lesions.If G.A. is managed in this way and, at the 6- and 12-month repeats of both tests, has a positive HPV test (regardless of the cytology finding) or any abnormal Pap test result other than HPV-negative/ASC-US, she should undergo colposcopy.15 A test designated as HPV-negative/ASC-US can be managed by repeat testing in 12 months.
Remember: Even though a repeat positive HPV test increases the patient’s risk of CIN 2,3 significantly, it is specifically not recommended to treat the cervix solely on the basis of a persistently positive HPV test without evidence of CIN or cervical cancer.
This patient warrants a different approach—here’s why
Although repeat Pap and HPV testing in 6 to 12 months is the standard recommendation for women with a normal Pap test and positive HPV results, extenuating circumstances may exist. Clinical judgment always trumps routine recommendations in these cases.
The progression of CIN 3 to cervical cancer is usually a slow process that occurs over many years.6 Therefore, delaying colposcopy for 6 to 12 months will probably not increase risk significantly even if a high-grade lesion is already present. But G.A.’s case involves a number of variables that may increase her risk enough to justify immediate colposcopy:
- an abnormal Pap test more than 20 years ago
- a history of irregular screening
- no screening within the past 5 years until the current testing
- concern that her last Pap result was either minimally abnormal or of limited quality.
Lack of access to any earlier records further limits the physician’s ability to adequately judge G.A.’s risk. Because of these concerns, the physician asks G.A. to come in for colposcopy, at which time a 2-quadrant CIN 3 lesion is found (FIGURE 2). The patient is treated by loop electrosurgical excision procedure and has normal cytology and a negative HPV test result on follow-up.

FIGURE 2 Two-quadrant CIN 2,3
High-grade lesions begin as a monoclonal cell change that enlarges centrifugally. Hence, increasing size is suspect for increasing duration of presence and increasing risk for cancer, because risk of invasion is proportional to lesion size.
Did you read Dr. Thomas C. Wright’s Update on Cervical Disease in the March Issue of OBG Management? If not, visit www.obgmanagement.com and follow the PAST ISSUES link on the navigation bar to the March issue.
The author reports no financial relationships relevant to this article.
1. Screening for squamous cervical cancer: duration of low risk after negative results of cervical cytology and its implication for screening policies. IARC Working Group on Evaluation of Cervical Cancer Screening Programmes. Br Med J (J Clin Res Ed). 1986;293:659-664.
2. Sung HY, Kearney KA, Miller M, Kinney W, Sawaya GF, Hiatt RA. Papanicolaou smear history and diagnosis of invasive cervical carcinoma among members of a large prepaid health plan. Cancer. 2000;88:2283-2289.
3. Nanda K, McCrory DC, Myers ER, et al. Accuracy of the Papanicolaou test in screening for and follow-up of cervical cytologic abnormalities: a systematic review. Ann Intern Med. 2000;132:810-819.
4. Fahey MT, Irwig L, Macaskill P. Meta-analysis of Pap test accuracy. Am J Epidemiol. 1995;141:680-689.
5. Davey E, Barratt A, Irwig L, et al. Effect of study design and quality on unsatisfactory rates, cytology classifications, and accuracy in liquid-based versus conventional cervical cytology: a systematic review. Lancet. 2006;367:122-132.
6. Kitchener HC, Castle PE, Cox JT. Chapter 7: Achievements and limitations of cervical cytology screening. Vaccine. 2006;24 Suppl 3:S63-S70.
7. Saslow D, Runowicz CD, Solomon D, et al. For the American Cancer Society. American Cancer Society guideline for the early detection of cervical neoplasia and cancer. CA Cancer J Clin. 2002;52:342-362.
8. Cervical cytology screening. American College of Obstetricians and Gynecologists Practice Bulletin #45. Washington, DC: ACOG; 2003.
9. Management of abnormal cervical cytology and histology. Clinical management guidelines for the Obstetrician and Gynecologist. American College of Obstetricians and Gynecologists Practice Bulletin #66. Washington, DC: ACOG; 2005.
10. Human papillomavirus. American College of Obstetricians and Gynecologists Practice Bulletin #61. Washington, DC: ACOG; 2005.
11. Clavel C, Masure M, Bory JP, et al. Human papillomavirus testing in primary screening for the detection of high-grade cervical lesions: a study of 7,932 women. Br J Cancer. 2001;84:1616-1623.
12. Cuzick J, Szarewski A, Cubie H, et al. Management of women who test positive for high-risk types of human papillomavirus: the HART study. Lancet. 2003;362:1871-1876.
13. Sherman ME, Lorincz AT, Scott DR, et al. Baseline cytology, human papillomavirus testing, and risk for cervical neoplasia: 10-year cohort analysis. J Natl Cancer Inst. 2003;95:46-52.
14. Khan MJ, Castle PE, Lorincz AT, et al. The elevated 10-year risk of cervical precancer and cancer in women with human papillomavirus (HPV) type 16 or 18 and the possible utility of type-specific HPV testing in clinical practice. J Natl Cancer Inst. 2005;97:1072-1079.
15. Wright TC, Jr, Schiffman M, Solomon D, et al. Interim guidance for the use of human papillomavirus DNA testing as an adjunct to cervical cytology for screening. Obstet Gynecol. 2004;103:304-309.
1. Screening for squamous cervical cancer: duration of low risk after negative results of cervical cytology and its implication for screening policies. IARC Working Group on Evaluation of Cervical Cancer Screening Programmes. Br Med J (J Clin Res Ed). 1986;293:659-664.
2. Sung HY, Kearney KA, Miller M, Kinney W, Sawaya GF, Hiatt RA. Papanicolaou smear history and diagnosis of invasive cervical carcinoma among members of a large prepaid health plan. Cancer. 2000;88:2283-2289.
3. Nanda K, McCrory DC, Myers ER, et al. Accuracy of the Papanicolaou test in screening for and follow-up of cervical cytologic abnormalities: a systematic review. Ann Intern Med. 2000;132:810-819.
4. Fahey MT, Irwig L, Macaskill P. Meta-analysis of Pap test accuracy. Am J Epidemiol. 1995;141:680-689.
5. Davey E, Barratt A, Irwig L, et al. Effect of study design and quality on unsatisfactory rates, cytology classifications, and accuracy in liquid-based versus conventional cervical cytology: a systematic review. Lancet. 2006;367:122-132.
6. Kitchener HC, Castle PE, Cox JT. Chapter 7: Achievements and limitations of cervical cytology screening. Vaccine. 2006;24 Suppl 3:S63-S70.
7. Saslow D, Runowicz CD, Solomon D, et al. For the American Cancer Society. American Cancer Society guideline for the early detection of cervical neoplasia and cancer. CA Cancer J Clin. 2002;52:342-362.
8. Cervical cytology screening. American College of Obstetricians and Gynecologists Practice Bulletin #45. Washington, DC: ACOG; 2003.
9. Management of abnormal cervical cytology and histology. Clinical management guidelines for the Obstetrician and Gynecologist. American College of Obstetricians and Gynecologists Practice Bulletin #66. Washington, DC: ACOG; 2005.
10. Human papillomavirus. American College of Obstetricians and Gynecologists Practice Bulletin #61. Washington, DC: ACOG; 2005.
11. Clavel C, Masure M, Bory JP, et al. Human papillomavirus testing in primary screening for the detection of high-grade cervical lesions: a study of 7,932 women. Br J Cancer. 2001;84:1616-1623.
12. Cuzick J, Szarewski A, Cubie H, et al. Management of women who test positive for high-risk types of human papillomavirus: the HART study. Lancet. 2003;362:1871-1876.
13. Sherman ME, Lorincz AT, Scott DR, et al. Baseline cytology, human papillomavirus testing, and risk for cervical neoplasia: 10-year cohort analysis. J Natl Cancer Inst. 2003;95:46-52.
14. Khan MJ, Castle PE, Lorincz AT, et al. The elevated 10-year risk of cervical precancer and cancer in women with human papillomavirus (HPV) type 16 or 18 and the possible utility of type-specific HPV testing in clinical practice. J Natl Cancer Inst. 2005;97:1072-1079.
15. Wright TC, Jr, Schiffman M, Solomon D, et al. Interim guidance for the use of human papillomavirus DNA testing as an adjunct to cervical cytology for screening. Obstet Gynecol. 2004;103:304-309.
IN THIS ARTICLE
QDoes postmenopausal use of unopposed estrogen increase the risk of breast cancer?
<huc>A</huc>Yes, if the estrogen is oral estradiol and it is used for 5 years or longer. When Lyytinen and colleagues studied different estrogen doses, constituents, and routes of administration in a cohort representing the entire postmenopausal population of Finland, they found an additional 2 to 3 cases of breast cancer for every 1,000 women when oral estradiol was used for 5 years or more. When it was used for a shorter time, or when the estrogen was oral estriol or a vaginal formulation, there was no increase in risk.
Expert Commentary
Statistical modeling cannot adequately account for the multiple variables involved in complex conditions such as breast cancer. For example, the GAIL model, which is used to predict an individual’s chance of having breast cancer, will only correctly score 59% of women with cancer; 41% of women with cancer will have a lower score estimate than their cancer-free cohorts. In short, “current breast cancer risk prediction models perform well for populations but poorly for individuals.”1
Breast cancer is not a single disease
Estrogen-related breast cancer is preceded histologically by atypical epithelial hyperplasia that progresses to invasive disease in some but not all women. Women who develop breast cancer while taking estrogen are more likely to have immature duct epithelium that is predominantly estrogen receptor-alpha (ERα). They are also likely to be genetically susceptible to modified physiologic cell growth and estrogen-metabolizing pathways in response to various environmental carcinogens or oncogenic promoters. In this context, exogenous estrogen may be a promoter, but not an instigator, of breast cancer.
A deficiency of vitamin D also plays a role. Vitamin D has potent antiproliferative effects that include the differentiation of breast tissue, enhanced apoptosis, and inhibition of cancer cell growth.
Untreated women with breast cancer have higher tissue levels of estrogen, which are correlated with increased breast-tissue enzymatic activity (aromatase, sulfatase, and 17β-OH dehydrogenase), especially in women with a genetic predisposition to increased or aberrant breast-tissue estrogen synthesis and metabolism. Other inherent factors include gene mutation involving cell-cycle growth (BRCA1, BRCA2, p53), and the ratio and expression of estrogen receptors; estrogen-therapy-associated breast cancer is more prevalent in women with a predominant ERα/ERβ ratio.
Mammographic density reflects the breast’s hormonal environment, the influence of background genetics, and the effect of various types, dosages, and routes of exogenous estrogen.
Not all estrogen is bioavailable
About 95% of orally administered estradiol is metabolized to estrone, estrone sulfate, and estradiol glucoronide. The bioconversion of these pro-hormones to more potent estradiol is dependent on the estrogen-metabolizing enzymes noted previously, the dose of estrogen, and the route of administration. Only 5% of orally administered estradiol is bioavailable. Because of the hepatic first-pass effect, 1 mg of oral 17β-estradiol and 25 μg of transdermal estrogen yield equivalent levels of free serum estradiol.
Variability in these and unknown factors account for the differing results of population-based studies and meta-analyses. It also may explain why, in a randomly selected group of Finnish women, only 2 to 3 extra cases of breast cancer for every 1,000 women were detected after 10 years of estrogen therapy, and at a dose twice that currently recommended.
Timing is critical, too
In the estrogen-alone arm of the Women’s Health Initiative, women aged 50 to 59 years—who are most likely to be treated with estrogen in everyday clinical practice—derived cardiovascular protection (hazard ratio [HR], 0.56), reduced colorectal cancer incidence, and a reduction in breast cancer (HR, 0.72). In contrast, most of the women in the study by Lyytinen and colleagues were over age 60. Other important risk factors not noted in their study include parity (pregnancy induces differentiation and maturation of breast ductal epithelium), pretreatment mammographic density, and vitamin D status.
Clinical recommendations
- Conduct a full clinical evaluation before initiating estrogen therapy
- Assess mammographic density before and after initiation of estrogen therapy. If density increases, stop therapy or reduce the dosage and repeat mammography in 3 to 6 months
- Measure high-sensitivity serum estradiol in women at high risk. Values in excess of 10 pg/dL may reflect an increased risk of breast cancer in untreated women—although no particular level of concern has been definitively identified
- Individualize dose and length of therapy according to age and indication.
Arbitrary restriction of estrogen therapy to 5 years is not biologically rational or clinically justifiable.
1. Elmore JG, Fletcher SW. The risk of cancer risk predisposition: “What is my risk of getting breast cancer?” J Natl Cancer Inst. 2006;98:1673-1675.
<huc>A</huc>Yes, if the estrogen is oral estradiol and it is used for 5 years or longer. When Lyytinen and colleagues studied different estrogen doses, constituents, and routes of administration in a cohort representing the entire postmenopausal population of Finland, they found an additional 2 to 3 cases of breast cancer for every 1,000 women when oral estradiol was used for 5 years or more. When it was used for a shorter time, or when the estrogen was oral estriol or a vaginal formulation, there was no increase in risk.
Expert Commentary
Statistical modeling cannot adequately account for the multiple variables involved in complex conditions such as breast cancer. For example, the GAIL model, which is used to predict an individual’s chance of having breast cancer, will only correctly score 59% of women with cancer; 41% of women with cancer will have a lower score estimate than their cancer-free cohorts. In short, “current breast cancer risk prediction models perform well for populations but poorly for individuals.”1
Breast cancer is not a single disease
Estrogen-related breast cancer is preceded histologically by atypical epithelial hyperplasia that progresses to invasive disease in some but not all women. Women who develop breast cancer while taking estrogen are more likely to have immature duct epithelium that is predominantly estrogen receptor-alpha (ERα). They are also likely to be genetically susceptible to modified physiologic cell growth and estrogen-metabolizing pathways in response to various environmental carcinogens or oncogenic promoters. In this context, exogenous estrogen may be a promoter, but not an instigator, of breast cancer.
A deficiency of vitamin D also plays a role. Vitamin D has potent antiproliferative effects that include the differentiation of breast tissue, enhanced apoptosis, and inhibition of cancer cell growth.
Untreated women with breast cancer have higher tissue levels of estrogen, which are correlated with increased breast-tissue enzymatic activity (aromatase, sulfatase, and 17β-OH dehydrogenase), especially in women with a genetic predisposition to increased or aberrant breast-tissue estrogen synthesis and metabolism. Other inherent factors include gene mutation involving cell-cycle growth (BRCA1, BRCA2, p53), and the ratio and expression of estrogen receptors; estrogen-therapy-associated breast cancer is more prevalent in women with a predominant ERα/ERβ ratio.
Mammographic density reflects the breast’s hormonal environment, the influence of background genetics, and the effect of various types, dosages, and routes of exogenous estrogen.
Not all estrogen is bioavailable
About 95% of orally administered estradiol is metabolized to estrone, estrone sulfate, and estradiol glucoronide. The bioconversion of these pro-hormones to more potent estradiol is dependent on the estrogen-metabolizing enzymes noted previously, the dose of estrogen, and the route of administration. Only 5% of orally administered estradiol is bioavailable. Because of the hepatic first-pass effect, 1 mg of oral 17β-estradiol and 25 μg of transdermal estrogen yield equivalent levels of free serum estradiol.
Variability in these and unknown factors account for the differing results of population-based studies and meta-analyses. It also may explain why, in a randomly selected group of Finnish women, only 2 to 3 extra cases of breast cancer for every 1,000 women were detected after 10 years of estrogen therapy, and at a dose twice that currently recommended.
Timing is critical, too
In the estrogen-alone arm of the Women’s Health Initiative, women aged 50 to 59 years—who are most likely to be treated with estrogen in everyday clinical practice—derived cardiovascular protection (hazard ratio [HR], 0.56), reduced colorectal cancer incidence, and a reduction in breast cancer (HR, 0.72). In contrast, most of the women in the study by Lyytinen and colleagues were over age 60. Other important risk factors not noted in their study include parity (pregnancy induces differentiation and maturation of breast ductal epithelium), pretreatment mammographic density, and vitamin D status.
Clinical recommendations
- Conduct a full clinical evaluation before initiating estrogen therapy
- Assess mammographic density before and after initiation of estrogen therapy. If density increases, stop therapy or reduce the dosage and repeat mammography in 3 to 6 months
- Measure high-sensitivity serum estradiol in women at high risk. Values in excess of 10 pg/dL may reflect an increased risk of breast cancer in untreated women—although no particular level of concern has been definitively identified
- Individualize dose and length of therapy according to age and indication.
Arbitrary restriction of estrogen therapy to 5 years is not biologically rational or clinically justifiable.
<huc>A</huc>Yes, if the estrogen is oral estradiol and it is used for 5 years or longer. When Lyytinen and colleagues studied different estrogen doses, constituents, and routes of administration in a cohort representing the entire postmenopausal population of Finland, they found an additional 2 to 3 cases of breast cancer for every 1,000 women when oral estradiol was used for 5 years or more. When it was used for a shorter time, or when the estrogen was oral estriol or a vaginal formulation, there was no increase in risk.
Expert Commentary
Statistical modeling cannot adequately account for the multiple variables involved in complex conditions such as breast cancer. For example, the GAIL model, which is used to predict an individual’s chance of having breast cancer, will only correctly score 59% of women with cancer; 41% of women with cancer will have a lower score estimate than their cancer-free cohorts. In short, “current breast cancer risk prediction models perform well for populations but poorly for individuals.”1
Breast cancer is not a single disease
Estrogen-related breast cancer is preceded histologically by atypical epithelial hyperplasia that progresses to invasive disease in some but not all women. Women who develop breast cancer while taking estrogen are more likely to have immature duct epithelium that is predominantly estrogen receptor-alpha (ERα). They are also likely to be genetically susceptible to modified physiologic cell growth and estrogen-metabolizing pathways in response to various environmental carcinogens or oncogenic promoters. In this context, exogenous estrogen may be a promoter, but not an instigator, of breast cancer.
A deficiency of vitamin D also plays a role. Vitamin D has potent antiproliferative effects that include the differentiation of breast tissue, enhanced apoptosis, and inhibition of cancer cell growth.
Untreated women with breast cancer have higher tissue levels of estrogen, which are correlated with increased breast-tissue enzymatic activity (aromatase, sulfatase, and 17β-OH dehydrogenase), especially in women with a genetic predisposition to increased or aberrant breast-tissue estrogen synthesis and metabolism. Other inherent factors include gene mutation involving cell-cycle growth (BRCA1, BRCA2, p53), and the ratio and expression of estrogen receptors; estrogen-therapy-associated breast cancer is more prevalent in women with a predominant ERα/ERβ ratio.
Mammographic density reflects the breast’s hormonal environment, the influence of background genetics, and the effect of various types, dosages, and routes of exogenous estrogen.
Not all estrogen is bioavailable
About 95% of orally administered estradiol is metabolized to estrone, estrone sulfate, and estradiol glucoronide. The bioconversion of these pro-hormones to more potent estradiol is dependent on the estrogen-metabolizing enzymes noted previously, the dose of estrogen, and the route of administration. Only 5% of orally administered estradiol is bioavailable. Because of the hepatic first-pass effect, 1 mg of oral 17β-estradiol and 25 μg of transdermal estrogen yield equivalent levels of free serum estradiol.
Variability in these and unknown factors account for the differing results of population-based studies and meta-analyses. It also may explain why, in a randomly selected group of Finnish women, only 2 to 3 extra cases of breast cancer for every 1,000 women were detected after 10 years of estrogen therapy, and at a dose twice that currently recommended.
Timing is critical, too
In the estrogen-alone arm of the Women’s Health Initiative, women aged 50 to 59 years—who are most likely to be treated with estrogen in everyday clinical practice—derived cardiovascular protection (hazard ratio [HR], 0.56), reduced colorectal cancer incidence, and a reduction in breast cancer (HR, 0.72). In contrast, most of the women in the study by Lyytinen and colleagues were over age 60. Other important risk factors not noted in their study include parity (pregnancy induces differentiation and maturation of breast ductal epithelium), pretreatment mammographic density, and vitamin D status.
Clinical recommendations
- Conduct a full clinical evaluation before initiating estrogen therapy
- Assess mammographic density before and after initiation of estrogen therapy. If density increases, stop therapy or reduce the dosage and repeat mammography in 3 to 6 months
- Measure high-sensitivity serum estradiol in women at high risk. Values in excess of 10 pg/dL may reflect an increased risk of breast cancer in untreated women—although no particular level of concern has been definitively identified
- Individualize dose and length of therapy according to age and indication.
Arbitrary restriction of estrogen therapy to 5 years is not biologically rational or clinically justifiable.
1. Elmore JG, Fletcher SW. The risk of cancer risk predisposition: “What is my risk of getting breast cancer?” J Natl Cancer Inst. 2006;98:1673-1675.
1. Elmore JG, Fletcher SW. The risk of cancer risk predisposition: “What is my risk of getting breast cancer?” J Natl Cancer Inst. 2006;98:1673-1675.
Does bone loss resume when alendronate is discontinued?
Yes. But the amount of bone loss is clinically small (2% to 3%) in women who stop taking alendronate after 5 years of therapy. At 10 years after initiation of alendronate (5 years after discontinuation), bone mineral density remained well above baseline value.
Expert Commentary
This large, multicenter trial will help us better define clinical management with the bisphosphonates—although this study looked specifically at alendronate. An earlier and smaller study of 226 subjects by Greenspan and colleagues demonstrated that bone mineral density (BMD) maintains itself for 15 months after discontinuation of alendronate.1 This trial by Black and colleagues—the FLEX trial—is a 5-year extension of the Fracture Intervention Trial (FIT).2 It randomized 1,099 women who had taken alendronate for 5 years in FIT to alendronate 5 mg daily (n=329), 10 mg daily (n=333), or placebo (n=437) for 5 additional years. Women were excluded from FLEX if their T-score was less than -3.5 or their BMD was lower than at entry into FIT.
In the FLEX trial, women who switched to placebo after 5 years of alendronate lost a statistically significant but clinically small amount of BMD—approximately 2% to 3%—compared with those who continued taking alendronate for a full 10 years. In all groups, however, BMD levels remained well above baseline at the time of entry into FIT.
Similarities in fracture rates, too
Despite the small difference in BMD measurements between groups, there was no increase in overall clinical fractures or radiographically detected vertebral fractures among women in the placebo group. However, there was a statistically significant 2.9% increase in absolute risk for clinically detected vertebral fractures. One reason for these somewhat surprising findings may be that the trial was powered to detect BMD changes rather than fractures. Nevertheless, it appears that, for some women, 5 years of bisphosphonate therapy may be enough to realize fracture-reduction benefits.
The magnitude of the absolute reduction in clinical vertebral fractures was greatest in women with T-scores worse than -2.5 at the beginning of FLEX, as well as in those with a baseline vertebral fracture at entry. The authors conclude that women at high risk of vertebral fracture because of previous vertebral fractures may be considered for continued therapy. Obviously, a long-term study powered for fractures rather than BMD measurement would be ideal, if extraordinarily expensive.
Who can take a ‘drug holiday’?
Women who have a good response to 5 years of bisphosphonate therapy (ie, a 3–5% increase in hip BMD, 8–10% increase in spine BMD, and a T-score better than -3.5) do not appear to be at increased risk of vertebral fracture after a “drug holiday” of up to 5 years. Such an approach would clearly improve the cost-effectiveness of bisphosphonate therapy. However, it would also necessitate careful BMD monitoring because the BMD values listed above are mean findings. Close monitoring would identify women who might be rapidly losing BMD and who need to resume bisphosphonate therapy or an alternative. Therefore, the treatment center should be reliable, with use of the same dual-energy x-ray absorptiometry (DXA) machine whenever possible.
Today, almost all patients are treated with once-weekly dosing. Although this regimen appears to be equivalent to daily dosing,3 it could confound the findings of FLEX.
Bottom line: Consider stopping alendronate in selected patients
Findings from FIT and similar trials established that the initiation of bisphosphonate therapy in postmenopausal women with osteoporosis or a previous nontraumatic fracture substantially reduces their risk of vertebral and nonvertebral fractures.4 These new data from the FLEX trial will allow us to discontinue bisphosphonate therapy in some women after 5 years without exposing them to additional risk.5
1. Greenspan SL. Significant differential effects of alendronate, estrogen, or combination therapy on the rate of bone loss after discontinuation of treatment of postmenopausal osteoporosis. A randomized, double-blind, placebo-controlled trial. Ann Intern Med. 2002;137:875-883.
2. Black DM, Cummings SR, Karpt DB, et al. Randomized trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Lancet. 1996;348:1535-1541.
3. Emkey R. Alendronate and risedronate for the treatment of postmenopausal osteoporosis: clinical profiles of the once-weekly and once-daily dosing formulations. Med Gen Med. 2004;204(3):6.-
4. Fink HA, Ensrud KE, Nelson DB, et al. Disability after clinical fracture in postmenopausal women with low bone density: the Fracture Intervention Trial (FIT). Osteoporosis Int. 2003;14:69-76.
5. Colon-Emerie CS. Ten vs five years of bisphosphonate treatment for postmenopausal osteoporosis. Enough of a good thing. JAMA. 2006;296:2968-2969.
Yes. But the amount of bone loss is clinically small (2% to 3%) in women who stop taking alendronate after 5 years of therapy. At 10 years after initiation of alendronate (5 years after discontinuation), bone mineral density remained well above baseline value.
Expert Commentary
This large, multicenter trial will help us better define clinical management with the bisphosphonates—although this study looked specifically at alendronate. An earlier and smaller study of 226 subjects by Greenspan and colleagues demonstrated that bone mineral density (BMD) maintains itself for 15 months after discontinuation of alendronate.1 This trial by Black and colleagues—the FLEX trial—is a 5-year extension of the Fracture Intervention Trial (FIT).2 It randomized 1,099 women who had taken alendronate for 5 years in FIT to alendronate 5 mg daily (n=329), 10 mg daily (n=333), or placebo (n=437) for 5 additional years. Women were excluded from FLEX if their T-score was less than -3.5 or their BMD was lower than at entry into FIT.
In the FLEX trial, women who switched to placebo after 5 years of alendronate lost a statistically significant but clinically small amount of BMD—approximately 2% to 3%—compared with those who continued taking alendronate for a full 10 years. In all groups, however, BMD levels remained well above baseline at the time of entry into FIT.
Similarities in fracture rates, too
Despite the small difference in BMD measurements between groups, there was no increase in overall clinical fractures or radiographically detected vertebral fractures among women in the placebo group. However, there was a statistically significant 2.9% increase in absolute risk for clinically detected vertebral fractures. One reason for these somewhat surprising findings may be that the trial was powered to detect BMD changes rather than fractures. Nevertheless, it appears that, for some women, 5 years of bisphosphonate therapy may be enough to realize fracture-reduction benefits.
The magnitude of the absolute reduction in clinical vertebral fractures was greatest in women with T-scores worse than -2.5 at the beginning of FLEX, as well as in those with a baseline vertebral fracture at entry. The authors conclude that women at high risk of vertebral fracture because of previous vertebral fractures may be considered for continued therapy. Obviously, a long-term study powered for fractures rather than BMD measurement would be ideal, if extraordinarily expensive.
Who can take a ‘drug holiday’?
Women who have a good response to 5 years of bisphosphonate therapy (ie, a 3–5% increase in hip BMD, 8–10% increase in spine BMD, and a T-score better than -3.5) do not appear to be at increased risk of vertebral fracture after a “drug holiday” of up to 5 years. Such an approach would clearly improve the cost-effectiveness of bisphosphonate therapy. However, it would also necessitate careful BMD monitoring because the BMD values listed above are mean findings. Close monitoring would identify women who might be rapidly losing BMD and who need to resume bisphosphonate therapy or an alternative. Therefore, the treatment center should be reliable, with use of the same dual-energy x-ray absorptiometry (DXA) machine whenever possible.
Today, almost all patients are treated with once-weekly dosing. Although this regimen appears to be equivalent to daily dosing,3 it could confound the findings of FLEX.
Bottom line: Consider stopping alendronate in selected patients
Findings from FIT and similar trials established that the initiation of bisphosphonate therapy in postmenopausal women with osteoporosis or a previous nontraumatic fracture substantially reduces their risk of vertebral and nonvertebral fractures.4 These new data from the FLEX trial will allow us to discontinue bisphosphonate therapy in some women after 5 years without exposing them to additional risk.5
Yes. But the amount of bone loss is clinically small (2% to 3%) in women who stop taking alendronate after 5 years of therapy. At 10 years after initiation of alendronate (5 years after discontinuation), bone mineral density remained well above baseline value.
Expert Commentary
This large, multicenter trial will help us better define clinical management with the bisphosphonates—although this study looked specifically at alendronate. An earlier and smaller study of 226 subjects by Greenspan and colleagues demonstrated that bone mineral density (BMD) maintains itself for 15 months after discontinuation of alendronate.1 This trial by Black and colleagues—the FLEX trial—is a 5-year extension of the Fracture Intervention Trial (FIT).2 It randomized 1,099 women who had taken alendronate for 5 years in FIT to alendronate 5 mg daily (n=329), 10 mg daily (n=333), or placebo (n=437) for 5 additional years. Women were excluded from FLEX if their T-score was less than -3.5 or their BMD was lower than at entry into FIT.
In the FLEX trial, women who switched to placebo after 5 years of alendronate lost a statistically significant but clinically small amount of BMD—approximately 2% to 3%—compared with those who continued taking alendronate for a full 10 years. In all groups, however, BMD levels remained well above baseline at the time of entry into FIT.
Similarities in fracture rates, too
Despite the small difference in BMD measurements between groups, there was no increase in overall clinical fractures or radiographically detected vertebral fractures among women in the placebo group. However, there was a statistically significant 2.9% increase in absolute risk for clinically detected vertebral fractures. One reason for these somewhat surprising findings may be that the trial was powered to detect BMD changes rather than fractures. Nevertheless, it appears that, for some women, 5 years of bisphosphonate therapy may be enough to realize fracture-reduction benefits.
The magnitude of the absolute reduction in clinical vertebral fractures was greatest in women with T-scores worse than -2.5 at the beginning of FLEX, as well as in those with a baseline vertebral fracture at entry. The authors conclude that women at high risk of vertebral fracture because of previous vertebral fractures may be considered for continued therapy. Obviously, a long-term study powered for fractures rather than BMD measurement would be ideal, if extraordinarily expensive.
Who can take a ‘drug holiday’?
Women who have a good response to 5 years of bisphosphonate therapy (ie, a 3–5% increase in hip BMD, 8–10% increase in spine BMD, and a T-score better than -3.5) do not appear to be at increased risk of vertebral fracture after a “drug holiday” of up to 5 years. Such an approach would clearly improve the cost-effectiveness of bisphosphonate therapy. However, it would also necessitate careful BMD monitoring because the BMD values listed above are mean findings. Close monitoring would identify women who might be rapidly losing BMD and who need to resume bisphosphonate therapy or an alternative. Therefore, the treatment center should be reliable, with use of the same dual-energy x-ray absorptiometry (DXA) machine whenever possible.
Today, almost all patients are treated with once-weekly dosing. Although this regimen appears to be equivalent to daily dosing,3 it could confound the findings of FLEX.
Bottom line: Consider stopping alendronate in selected patients
Findings from FIT and similar trials established that the initiation of bisphosphonate therapy in postmenopausal women with osteoporosis or a previous nontraumatic fracture substantially reduces their risk of vertebral and nonvertebral fractures.4 These new data from the FLEX trial will allow us to discontinue bisphosphonate therapy in some women after 5 years without exposing them to additional risk.5
1. Greenspan SL. Significant differential effects of alendronate, estrogen, or combination therapy on the rate of bone loss after discontinuation of treatment of postmenopausal osteoporosis. A randomized, double-blind, placebo-controlled trial. Ann Intern Med. 2002;137:875-883.
2. Black DM, Cummings SR, Karpt DB, et al. Randomized trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Lancet. 1996;348:1535-1541.
3. Emkey R. Alendronate and risedronate for the treatment of postmenopausal osteoporosis: clinical profiles of the once-weekly and once-daily dosing formulations. Med Gen Med. 2004;204(3):6.-
4. Fink HA, Ensrud KE, Nelson DB, et al. Disability after clinical fracture in postmenopausal women with low bone density: the Fracture Intervention Trial (FIT). Osteoporosis Int. 2003;14:69-76.
5. Colon-Emerie CS. Ten vs five years of bisphosphonate treatment for postmenopausal osteoporosis. Enough of a good thing. JAMA. 2006;296:2968-2969.
1. Greenspan SL. Significant differential effects of alendronate, estrogen, or combination therapy on the rate of bone loss after discontinuation of treatment of postmenopausal osteoporosis. A randomized, double-blind, placebo-controlled trial. Ann Intern Med. 2002;137:875-883.
2. Black DM, Cummings SR, Karpt DB, et al. Randomized trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Lancet. 1996;348:1535-1541.
3. Emkey R. Alendronate and risedronate for the treatment of postmenopausal osteoporosis: clinical profiles of the once-weekly and once-daily dosing formulations. Med Gen Med. 2004;204(3):6.-
4. Fink HA, Ensrud KE, Nelson DB, et al. Disability after clinical fracture in postmenopausal women with low bone density: the Fracture Intervention Trial (FIT). Osteoporosis Int. 2003;14:69-76.
5. Colon-Emerie CS. Ten vs five years of bisphosphonate treatment for postmenopausal osteoporosis. Enough of a good thing. JAMA. 2006;296:2968-2969.
Treating the range of lower-tract symptoms in prolapse
To watch a demonstration of how pelvic organ prolapse is repaired, visit the Video Library.
Lower urinary tract symptoms are common in women who have pelvic organ prolapse (POP). For some, these symptoms resolve or improve after surgery for prolapse; for others, symptoms remain unchanged or become worse. These clinical pearls can help you decide how to counsel, evaluate, and treat patients who have POP and coexisting lower-tract symptoms.
How are POP and lower-tract symptoms related?
Lower-tract symptoms that result from, or coexist with, POP include urinary incontinence (stress, urge, mixed), irritative symptoms (frequency, urgency, nocturia), and difficulty voiding (hesitancy, weak or intermittent stream). Prolapse can produce lower-tract symptoms by:
- causing urethral obstruction
- dissipating the effects of abdominal pressure during Valsalva voiding, which makes voiding more difficult
- masking sphincteric incontinence.
Is prolapse causing symptoms or masking stress incontinence?
Some clues to answering this question can be obtained from the history:
- If the patient says that she voids better when the prolapse is reduced, prolapse is probably causing urethral obstruction
- If the patient says that she experienced stress incontinence previously but that it has subsided and she now only has difficulty voiding, she probably has occult stress incontinence and, possibly, urethral obstruction.
How is treatment established for lower-tract symptoms?
Prolapse is graded by any of several classifications that are based on the severity and extent of the condition.
Mild degrees of prolapse rarely, if ever, cause urethral obstruction or mask stress incontinence; you can manage lower-tract symptoms in these patients as if they did not have prolapse. Stress incontinence, which is common among these women, can be corrected either in isolation or in conjunction with repair of the prolapse, if such repair is indicated.
More advanced degrees of prolapse, defined as prolapse that extends to or beyond the hymen, are commonly associated with urethral obstruction or occult sphincteric incontinence, or both. This makes it important to diagnose these conditions (by means described earlier) before you intervene surgically to repair the prolapse.
Can surgery for POP affect lower-tract symptoms?
Paradoxically, surgical treatment of prolapse can treat lower-tract symptoms successfully in some patients but cause them in others. How can this be?
- Surgery works when prolapse has caused obstruction and the obstruction is relieved when you resupport the pelvic floor
- Surgery can cause symptoms when occult stress incontinence goes unrecognized and is unmasked after repair of prolapse without concomitant anti-incontinence surgery
- De novo irritative symptoms and OAB can arise secondary to placement of a sling.
Treat all prolapse surgery patients with prophylactic anti-incontinence surgery?
Some experts recommend that practice. But anti-incontinence surgery carries its own risk of complications, so we believe that the need for anti-incontinence surgery should be individualized—based on symptoms, anatomy, the results of diagnostic testing, and the patient’s quality-of-life priorities. Of course, when there is pre-existing or occult stress incontinence, you should routinely consider concomitant anti-incontinence surgery.
When is POP surgery effective for OAB symptoms?
The literature is scant on this question. We believe that, in patients who have an advanced degree of prolapse (especially when urethral obstruction has been documented), symptoms of OAB subside most of the time after effective prolapse surgery.
We do not recommend surgery for mild degrees of prolapse or when there is no pre-existing obstruction.
Drs. Blaivas and Karram co-chair the 6th Annual International Symposium on Female Urology & Urogynecology, to be held April 26–28, 2007, in Las Vegas (www.urogyn-cme.org).
To watch a demonstration of how pelvic organ prolapse is repaired, visit the Video Library.
Lower urinary tract symptoms are common in women who have pelvic organ prolapse (POP). For some, these symptoms resolve or improve after surgery for prolapse; for others, symptoms remain unchanged or become worse. These clinical pearls can help you decide how to counsel, evaluate, and treat patients who have POP and coexisting lower-tract symptoms.
How are POP and lower-tract symptoms related?
Lower-tract symptoms that result from, or coexist with, POP include urinary incontinence (stress, urge, mixed), irritative symptoms (frequency, urgency, nocturia), and difficulty voiding (hesitancy, weak or intermittent stream). Prolapse can produce lower-tract symptoms by:
- causing urethral obstruction
- dissipating the effects of abdominal pressure during Valsalva voiding, which makes voiding more difficult
- masking sphincteric incontinence.
Is prolapse causing symptoms or masking stress incontinence?
Some clues to answering this question can be obtained from the history:
- If the patient says that she voids better when the prolapse is reduced, prolapse is probably causing urethral obstruction
- If the patient says that she experienced stress incontinence previously but that it has subsided and she now only has difficulty voiding, she probably has occult stress incontinence and, possibly, urethral obstruction.
How is treatment established for lower-tract symptoms?
Prolapse is graded by any of several classifications that are based on the severity and extent of the condition.
Mild degrees of prolapse rarely, if ever, cause urethral obstruction or mask stress incontinence; you can manage lower-tract symptoms in these patients as if they did not have prolapse. Stress incontinence, which is common among these women, can be corrected either in isolation or in conjunction with repair of the prolapse, if such repair is indicated.
More advanced degrees of prolapse, defined as prolapse that extends to or beyond the hymen, are commonly associated with urethral obstruction or occult sphincteric incontinence, or both. This makes it important to diagnose these conditions (by means described earlier) before you intervene surgically to repair the prolapse.
Can surgery for POP affect lower-tract symptoms?
Paradoxically, surgical treatment of prolapse can treat lower-tract symptoms successfully in some patients but cause them in others. How can this be?
- Surgery works when prolapse has caused obstruction and the obstruction is relieved when you resupport the pelvic floor
- Surgery can cause symptoms when occult stress incontinence goes unrecognized and is unmasked after repair of prolapse without concomitant anti-incontinence surgery
- De novo irritative symptoms and OAB can arise secondary to placement of a sling.
Treat all prolapse surgery patients with prophylactic anti-incontinence surgery?
Some experts recommend that practice. But anti-incontinence surgery carries its own risk of complications, so we believe that the need for anti-incontinence surgery should be individualized—based on symptoms, anatomy, the results of diagnostic testing, and the patient’s quality-of-life priorities. Of course, when there is pre-existing or occult stress incontinence, you should routinely consider concomitant anti-incontinence surgery.
When is POP surgery effective for OAB symptoms?
The literature is scant on this question. We believe that, in patients who have an advanced degree of prolapse (especially when urethral obstruction has been documented), symptoms of OAB subside most of the time after effective prolapse surgery.
We do not recommend surgery for mild degrees of prolapse or when there is no pre-existing obstruction.
To watch a demonstration of how pelvic organ prolapse is repaired, visit the Video Library.
Lower urinary tract symptoms are common in women who have pelvic organ prolapse (POP). For some, these symptoms resolve or improve after surgery for prolapse; for others, symptoms remain unchanged or become worse. These clinical pearls can help you decide how to counsel, evaluate, and treat patients who have POP and coexisting lower-tract symptoms.
How are POP and lower-tract symptoms related?
Lower-tract symptoms that result from, or coexist with, POP include urinary incontinence (stress, urge, mixed), irritative symptoms (frequency, urgency, nocturia), and difficulty voiding (hesitancy, weak or intermittent stream). Prolapse can produce lower-tract symptoms by:
- causing urethral obstruction
- dissipating the effects of abdominal pressure during Valsalva voiding, which makes voiding more difficult
- masking sphincteric incontinence.
Is prolapse causing symptoms or masking stress incontinence?
Some clues to answering this question can be obtained from the history:
- If the patient says that she voids better when the prolapse is reduced, prolapse is probably causing urethral obstruction
- If the patient says that she experienced stress incontinence previously but that it has subsided and she now only has difficulty voiding, she probably has occult stress incontinence and, possibly, urethral obstruction.
How is treatment established for lower-tract symptoms?
Prolapse is graded by any of several classifications that are based on the severity and extent of the condition.
Mild degrees of prolapse rarely, if ever, cause urethral obstruction or mask stress incontinence; you can manage lower-tract symptoms in these patients as if they did not have prolapse. Stress incontinence, which is common among these women, can be corrected either in isolation or in conjunction with repair of the prolapse, if such repair is indicated.
More advanced degrees of prolapse, defined as prolapse that extends to or beyond the hymen, are commonly associated with urethral obstruction or occult sphincteric incontinence, or both. This makes it important to diagnose these conditions (by means described earlier) before you intervene surgically to repair the prolapse.
Can surgery for POP affect lower-tract symptoms?
Paradoxically, surgical treatment of prolapse can treat lower-tract symptoms successfully in some patients but cause them in others. How can this be?
- Surgery works when prolapse has caused obstruction and the obstruction is relieved when you resupport the pelvic floor
- Surgery can cause symptoms when occult stress incontinence goes unrecognized and is unmasked after repair of prolapse without concomitant anti-incontinence surgery
- De novo irritative symptoms and OAB can arise secondary to placement of a sling.
Treat all prolapse surgery patients with prophylactic anti-incontinence surgery?
Some experts recommend that practice. But anti-incontinence surgery carries its own risk of complications, so we believe that the need for anti-incontinence surgery should be individualized—based on symptoms, anatomy, the results of diagnostic testing, and the patient’s quality-of-life priorities. Of course, when there is pre-existing or occult stress incontinence, you should routinely consider concomitant anti-incontinence surgery.
When is POP surgery effective for OAB symptoms?
The literature is scant on this question. We believe that, in patients who have an advanced degree of prolapse (especially when urethral obstruction has been documented), symptoms of OAB subside most of the time after effective prolapse surgery.
We do not recommend surgery for mild degrees of prolapse or when there is no pre-existing obstruction.
Drs. Blaivas and Karram co-chair the 6th Annual International Symposium on Female Urology & Urogynecology, to be held April 26–28, 2007, in Las Vegas (www.urogyn-cme.org).
Drs. Blaivas and Karram co-chair the 6th Annual International Symposium on Female Urology & Urogynecology, to be held April 26–28, 2007, in Las Vegas (www.urogyn-cme.org).
Q Should all obese women be screened for PCOS?
Expert Commentary
Polycystic ovary syndrome (PCOS) is often accompanied by obesity, and the obesity epidemic appears to have been accompanied by a PCOS epidemic. Rather than focus on obesity’s effects on PCOS, Àlvarez-Blasco and colleagues looked for stigmata of PCOS in an unselected obese population.
Findings in line with earlier studies
This study adds credence to other investigations that have found women with a metabolic abnormality more likely than an unselected sample of the same population to have PCOS. Another study found a similar prevalence of PCOS—26.7%—among premenopausal women with type 2 diabetes.2
Obesity per se is associated with metabolic abnormalities, and the investigators showed an increasing prevalence of the metabolic syndrome and its components with increasing obesity among the study cohort. The components of metabolic syndrome are:
- waist circumference >88 cm
- triglyceride level >150 mg/dL
- HDL cholesterol ≤50 mg/dL
- blood pressure ≥130/85 mm Hg
- fasting glucose ≥100 mg/dL
Strengths and weaknesses
The prospective design, size of the cohort, and full phenotyping performed on all subjects are strengths of this study.
The major weakness is the referral bias of a university-based endocrine clinic that is likely to attract women who are obese and also have endocrine abnormalities such as PCOS. (Endocrinology and nutrition are a single medical specialty in Spain.)
The best prevalence study of PCOS in the US general population involved asymptomatic women applying for employment at a university medical center.3 A similar study design and findings would strengthen the investigators’ recommendations to routinely screen for PCOS in an obese population.
This study did not use the revised Rotterdam criteria, which incorporate ultrasonographic size and morphology of the ovaries into the diagnosis. Preliminary studies show that these revised criteria tend to increase the prevalence of PCOS by about 50% among women with oligomenorrhea,4 so Álvarez-Blasco and colleagues likely underdetected PCOS by these criteria.
Bottom line: Screen obese patients for PCOS and metabolic syndrome
This study adds evidence of obesity’s adverse effects on reproduction, and suggests that routine screening of obese women for both PCOS and the metabolic syndrome is a high-yield procedure (25–30% for both).
1. Escobar-Morreale HF, Roldan B, Barrio R, et al. High prevalence of the polycystic ovary syndrome and hirsutism in women with type 1 diabetes mellitus. J Clin Endocrinol Metab. 2000;85:4182-4187.
2. Peppard HR, Marfori J, Iuorno MJ, et al. Prevalence of polycystic ovary syndrome among premenopausal women with type 2 diabetes. Diabetes Care. 2001;24:1050-1052.
3. Azziz R, Woods KS, Reyna R, Key TJ, Knochenhauer ES, Yildiz BO. The prevalence and features of the polycystic ovary syndrome in an unselected population. J Clin Endocrinol Metab. 2004;89:2745-2749.
4. Broekmans FJ, Knauff EA, Valkenburg O, Laven JS, Eijkemans MJ, Fauser BC. PCOS according to the Rotterdam consensus criteria: change in prevalence among WHO-II anovulation and association with metabolic factors. BJOG. 2006;113:1210-1217.
Expert Commentary
Polycystic ovary syndrome (PCOS) is often accompanied by obesity, and the obesity epidemic appears to have been accompanied by a PCOS epidemic. Rather than focus on obesity’s effects on PCOS, Àlvarez-Blasco and colleagues looked for stigmata of PCOS in an unselected obese population.
Findings in line with earlier studies
This study adds credence to other investigations that have found women with a metabolic abnormality more likely than an unselected sample of the same population to have PCOS. Another study found a similar prevalence of PCOS—26.7%—among premenopausal women with type 2 diabetes.2
Obesity per se is associated with metabolic abnormalities, and the investigators showed an increasing prevalence of the metabolic syndrome and its components with increasing obesity among the study cohort. The components of metabolic syndrome are:
- waist circumference >88 cm
- triglyceride level >150 mg/dL
- HDL cholesterol ≤50 mg/dL
- blood pressure ≥130/85 mm Hg
- fasting glucose ≥100 mg/dL
Strengths and weaknesses
The prospective design, size of the cohort, and full phenotyping performed on all subjects are strengths of this study.
The major weakness is the referral bias of a university-based endocrine clinic that is likely to attract women who are obese and also have endocrine abnormalities such as PCOS. (Endocrinology and nutrition are a single medical specialty in Spain.)
The best prevalence study of PCOS in the US general population involved asymptomatic women applying for employment at a university medical center.3 A similar study design and findings would strengthen the investigators’ recommendations to routinely screen for PCOS in an obese population.
This study did not use the revised Rotterdam criteria, which incorporate ultrasonographic size and morphology of the ovaries into the diagnosis. Preliminary studies show that these revised criteria tend to increase the prevalence of PCOS by about 50% among women with oligomenorrhea,4 so Álvarez-Blasco and colleagues likely underdetected PCOS by these criteria.
Bottom line: Screen obese patients for PCOS and metabolic syndrome
This study adds evidence of obesity’s adverse effects on reproduction, and suggests that routine screening of obese women for both PCOS and the metabolic syndrome is a high-yield procedure (25–30% for both).
Expert Commentary
Polycystic ovary syndrome (PCOS) is often accompanied by obesity, and the obesity epidemic appears to have been accompanied by a PCOS epidemic. Rather than focus on obesity’s effects on PCOS, Àlvarez-Blasco and colleagues looked for stigmata of PCOS in an unselected obese population.
Findings in line with earlier studies
This study adds credence to other investigations that have found women with a metabolic abnormality more likely than an unselected sample of the same population to have PCOS. Another study found a similar prevalence of PCOS—26.7%—among premenopausal women with type 2 diabetes.2
Obesity per se is associated with metabolic abnormalities, and the investigators showed an increasing prevalence of the metabolic syndrome and its components with increasing obesity among the study cohort. The components of metabolic syndrome are:
- waist circumference >88 cm
- triglyceride level >150 mg/dL
- HDL cholesterol ≤50 mg/dL
- blood pressure ≥130/85 mm Hg
- fasting glucose ≥100 mg/dL
Strengths and weaknesses
The prospective design, size of the cohort, and full phenotyping performed on all subjects are strengths of this study.
The major weakness is the referral bias of a university-based endocrine clinic that is likely to attract women who are obese and also have endocrine abnormalities such as PCOS. (Endocrinology and nutrition are a single medical specialty in Spain.)
The best prevalence study of PCOS in the US general population involved asymptomatic women applying for employment at a university medical center.3 A similar study design and findings would strengthen the investigators’ recommendations to routinely screen for PCOS in an obese population.
This study did not use the revised Rotterdam criteria, which incorporate ultrasonographic size and morphology of the ovaries into the diagnosis. Preliminary studies show that these revised criteria tend to increase the prevalence of PCOS by about 50% among women with oligomenorrhea,4 so Álvarez-Blasco and colleagues likely underdetected PCOS by these criteria.
Bottom line: Screen obese patients for PCOS and metabolic syndrome
This study adds evidence of obesity’s adverse effects on reproduction, and suggests that routine screening of obese women for both PCOS and the metabolic syndrome is a high-yield procedure (25–30% for both).
1. Escobar-Morreale HF, Roldan B, Barrio R, et al. High prevalence of the polycystic ovary syndrome and hirsutism in women with type 1 diabetes mellitus. J Clin Endocrinol Metab. 2000;85:4182-4187.
2. Peppard HR, Marfori J, Iuorno MJ, et al. Prevalence of polycystic ovary syndrome among premenopausal women with type 2 diabetes. Diabetes Care. 2001;24:1050-1052.
3. Azziz R, Woods KS, Reyna R, Key TJ, Knochenhauer ES, Yildiz BO. The prevalence and features of the polycystic ovary syndrome in an unselected population. J Clin Endocrinol Metab. 2004;89:2745-2749.
4. Broekmans FJ, Knauff EA, Valkenburg O, Laven JS, Eijkemans MJ, Fauser BC. PCOS according to the Rotterdam consensus criteria: change in prevalence among WHO-II anovulation and association with metabolic factors. BJOG. 2006;113:1210-1217.
1. Escobar-Morreale HF, Roldan B, Barrio R, et al. High prevalence of the polycystic ovary syndrome and hirsutism in women with type 1 diabetes mellitus. J Clin Endocrinol Metab. 2000;85:4182-4187.
2. Peppard HR, Marfori J, Iuorno MJ, et al. Prevalence of polycystic ovary syndrome among premenopausal women with type 2 diabetes. Diabetes Care. 2001;24:1050-1052.
3. Azziz R, Woods KS, Reyna R, Key TJ, Knochenhauer ES, Yildiz BO. The prevalence and features of the polycystic ovary syndrome in an unselected population. J Clin Endocrinol Metab. 2004;89:2745-2749.
4. Broekmans FJ, Knauff EA, Valkenburg O, Laven JS, Eijkemans MJ, Fauser BC. PCOS according to the Rotterdam consensus criteria: change in prevalence among WHO-II anovulation and association with metabolic factors. BJOG. 2006;113:1210-1217.
Q Does inducing labor raise the risk of amniotic fluid embolism?
Expert Commentary
Although earlier studies suggested an association between AFE and medical induction of labor, as well as an association between AFE and the risk factors listed above, no controlled trials had confirmed or refuted this assumption until now. A population-based study by Gilbert in 19991 looked at pregnancy complications and mortality associated with AFE, rather than risk factors per se.
Incidence of AFE is hard to define
Worldwide, the incidence of AFE is estimated to range from 1 in 8,000 to 1 in 83,000 live births.2,3 In the United States, the estimate is 1 in 20,000 to 1 in 30,000 live births.4,5 In the study by Kramer and colleagues, the incidence of AFE was approximately 1 case per 17,000 singleton pregnancies, or 6 cases per 100,000—slightly higher than the 4.8 cases per 100,000 reported by Gilbert.1
The fatality rate varies, too
Overall, the reported mortality rate of AFE ranges from 26% to 90%.2,6 AFE is the fifth most common cause of maternal death in the world.7,8 In Canada, maternal deaths from AFE rank third behind cerebrovascular and hypertensive disorders.
Kramer and colleagues found a comparatively low mortality rate of only 13%. They reasoned that higher mortality rates in earlier uncontrolled case series may have been caused by a bias arising from selective reporting of more severe cases of AFE, such as fatal cases. They also postulated that the relatively constant annual rate of fatal AFE in their population argues against significant misdiagnosis.
AFE is linked to risk factors
The association between AFE and specific risk factors such as labor induction, cesarean section, and operative vaginal delivery was strengthened considerably when Kramer and colleagues restricted their analysis to fatal cases. The authors acknowledged that the link between AFE and cesarean section, instrumental delivery, and the presence of fetal distress may reflect the difficult labors that led to operative delivery—that is, reverse causality. If that is the case, it would substantiate the current belief that AFE is propagated by the tearing and shearing of fetal membranes and uterine vessels, which occurs more frequently in difficult and augmented labors with strong contractions. This theory is consistent with the authors’ finding that dystocia (probably associated with weaker contractions and early rupture of membranes) resulted in a significant reduction in the risk of AFE.
Nonfatal cases were hard to identify
The identification and analysis of nonfatal AFE cases were more problematic. Kramer and colleagues pointed out that the lower mortality rate found in their study may be secondary to some degree of overdiagnosis of nonfatal AFE, which can be difficult to identify. The lack of an absolute gold standard and the lack of specificity of the signs and symptoms of AFE may contribute to both overreporting and underreporting of this syndrome.
Compounding the dilemma is the fact that many signs and symptoms associated with AFE are also clinical syndromes that can occur in its absence, such as anaphylactic shock, left ventricular failure, sepsis, adult respiratory distress syndrome, and diffuse intravascular coagulation. Most commonly, AFE is a diagnosis of exclusion, made during pregnancy or the immediate postpartum period. However, even this temporal association lends little support to the diagnosis of AFE, because any and all of these other clinical syndromes can and do occur in isolation during and after pregnancy.
The constellation of many of the clinical sequelae, rather than isolated signs and symptoms, is what many recognize as the sine qua non of AFE. However, this hypothesis is also slightly problematic because not all cases of AFE—especially nonfatal cases—present with multiorgan disease involving cardiac, respiratory, renal, neurologic, and hematologic failure.
Choose elective induction wisely
Despite the relative rarity of AFE, the phenomenon is worthy of attention because the incidence of elective labor induction has been increasing steadily. The absolute increase in the risk of AFE among women who undergo medical induction is very small: 4 or 5 cases of AFE and 1 or 2 cases of fatal AFE for every 100,000 labors induced. However, with nearly 4 million births annually and induction rates of 20% in the United States, the incidence of AFE could escalate to 30 to 40 cases a year and 10 to 15 deaths. This statistic is alarming, especially given the increasing use of elective medical induction. Extra caution in choosing elective induction is therefore justified.
1. Gilbert WM, Danielson B. Amniotic fluid embolism: decreased mortality in a population-based study. Obstet Gynecol. 1999;93:973-977.
2. Clark SL, Hankins GD, Dudley DA, et al. Amniotic fluid embolism: analysis of the national registry. Am J Obstet Gynecol. 1995;172:1158-1167.
3. Tuffnell DJ. Amniotic fluid embolism. Curr Opin Obstet Gynecol. 2003;15:119-122.
4. Killam A. Amniotic fluid embolism. Clin Obstet Gynecol. 1985;28:32-36.
5. Sperry K. Landmark perspective: amniotic fluid embolism. To understand an enigma. JAMA. 1986;255:2183-2186.
6. Tuffnell DJ. United Kingdom amniotic fluid embolism register. BJOG. 2005;112:1625-1629.
7. Moore J, Baldisseri MR. Amniotic fluid embolism. Crit Care Med. 2005;33:S279-285.
8. Turner LA, Kramer MS, Liu S. for the Maternal Mortality and Morbidity Study Group of the Canadian Perinatal Surveillance System. Cause-specific mortality during and after pregnancy and the definition of maternal death. Chronic Dis Can. 2005;23:31-36.
Expert Commentary
Although earlier studies suggested an association between AFE and medical induction of labor, as well as an association between AFE and the risk factors listed above, no controlled trials had confirmed or refuted this assumption until now. A population-based study by Gilbert in 19991 looked at pregnancy complications and mortality associated with AFE, rather than risk factors per se.
Incidence of AFE is hard to define
Worldwide, the incidence of AFE is estimated to range from 1 in 8,000 to 1 in 83,000 live births.2,3 In the United States, the estimate is 1 in 20,000 to 1 in 30,000 live births.4,5 In the study by Kramer and colleagues, the incidence of AFE was approximately 1 case per 17,000 singleton pregnancies, or 6 cases per 100,000—slightly higher than the 4.8 cases per 100,000 reported by Gilbert.1
The fatality rate varies, too
Overall, the reported mortality rate of AFE ranges from 26% to 90%.2,6 AFE is the fifth most common cause of maternal death in the world.7,8 In Canada, maternal deaths from AFE rank third behind cerebrovascular and hypertensive disorders.
Kramer and colleagues found a comparatively low mortality rate of only 13%. They reasoned that higher mortality rates in earlier uncontrolled case series may have been caused by a bias arising from selective reporting of more severe cases of AFE, such as fatal cases. They also postulated that the relatively constant annual rate of fatal AFE in their population argues against significant misdiagnosis.
AFE is linked to risk factors
The association between AFE and specific risk factors such as labor induction, cesarean section, and operative vaginal delivery was strengthened considerably when Kramer and colleagues restricted their analysis to fatal cases. The authors acknowledged that the link between AFE and cesarean section, instrumental delivery, and the presence of fetal distress may reflect the difficult labors that led to operative delivery—that is, reverse causality. If that is the case, it would substantiate the current belief that AFE is propagated by the tearing and shearing of fetal membranes and uterine vessels, which occurs more frequently in difficult and augmented labors with strong contractions. This theory is consistent with the authors’ finding that dystocia (probably associated with weaker contractions and early rupture of membranes) resulted in a significant reduction in the risk of AFE.
Nonfatal cases were hard to identify
The identification and analysis of nonfatal AFE cases were more problematic. Kramer and colleagues pointed out that the lower mortality rate found in their study may be secondary to some degree of overdiagnosis of nonfatal AFE, which can be difficult to identify. The lack of an absolute gold standard and the lack of specificity of the signs and symptoms of AFE may contribute to both overreporting and underreporting of this syndrome.
Compounding the dilemma is the fact that many signs and symptoms associated with AFE are also clinical syndromes that can occur in its absence, such as anaphylactic shock, left ventricular failure, sepsis, adult respiratory distress syndrome, and diffuse intravascular coagulation. Most commonly, AFE is a diagnosis of exclusion, made during pregnancy or the immediate postpartum period. However, even this temporal association lends little support to the diagnosis of AFE, because any and all of these other clinical syndromes can and do occur in isolation during and after pregnancy.
The constellation of many of the clinical sequelae, rather than isolated signs and symptoms, is what many recognize as the sine qua non of AFE. However, this hypothesis is also slightly problematic because not all cases of AFE—especially nonfatal cases—present with multiorgan disease involving cardiac, respiratory, renal, neurologic, and hematologic failure.
Choose elective induction wisely
Despite the relative rarity of AFE, the phenomenon is worthy of attention because the incidence of elective labor induction has been increasing steadily. The absolute increase in the risk of AFE among women who undergo medical induction is very small: 4 or 5 cases of AFE and 1 or 2 cases of fatal AFE for every 100,000 labors induced. However, with nearly 4 million births annually and induction rates of 20% in the United States, the incidence of AFE could escalate to 30 to 40 cases a year and 10 to 15 deaths. This statistic is alarming, especially given the increasing use of elective medical induction. Extra caution in choosing elective induction is therefore justified.
Expert Commentary
Although earlier studies suggested an association between AFE and medical induction of labor, as well as an association between AFE and the risk factors listed above, no controlled trials had confirmed or refuted this assumption until now. A population-based study by Gilbert in 19991 looked at pregnancy complications and mortality associated with AFE, rather than risk factors per se.
Incidence of AFE is hard to define
Worldwide, the incidence of AFE is estimated to range from 1 in 8,000 to 1 in 83,000 live births.2,3 In the United States, the estimate is 1 in 20,000 to 1 in 30,000 live births.4,5 In the study by Kramer and colleagues, the incidence of AFE was approximately 1 case per 17,000 singleton pregnancies, or 6 cases per 100,000—slightly higher than the 4.8 cases per 100,000 reported by Gilbert.1
The fatality rate varies, too
Overall, the reported mortality rate of AFE ranges from 26% to 90%.2,6 AFE is the fifth most common cause of maternal death in the world.7,8 In Canada, maternal deaths from AFE rank third behind cerebrovascular and hypertensive disorders.
Kramer and colleagues found a comparatively low mortality rate of only 13%. They reasoned that higher mortality rates in earlier uncontrolled case series may have been caused by a bias arising from selective reporting of more severe cases of AFE, such as fatal cases. They also postulated that the relatively constant annual rate of fatal AFE in their population argues against significant misdiagnosis.
AFE is linked to risk factors
The association between AFE and specific risk factors such as labor induction, cesarean section, and operative vaginal delivery was strengthened considerably when Kramer and colleagues restricted their analysis to fatal cases. The authors acknowledged that the link between AFE and cesarean section, instrumental delivery, and the presence of fetal distress may reflect the difficult labors that led to operative delivery—that is, reverse causality. If that is the case, it would substantiate the current belief that AFE is propagated by the tearing and shearing of fetal membranes and uterine vessels, which occurs more frequently in difficult and augmented labors with strong contractions. This theory is consistent with the authors’ finding that dystocia (probably associated with weaker contractions and early rupture of membranes) resulted in a significant reduction in the risk of AFE.
Nonfatal cases were hard to identify
The identification and analysis of nonfatal AFE cases were more problematic. Kramer and colleagues pointed out that the lower mortality rate found in their study may be secondary to some degree of overdiagnosis of nonfatal AFE, which can be difficult to identify. The lack of an absolute gold standard and the lack of specificity of the signs and symptoms of AFE may contribute to both overreporting and underreporting of this syndrome.
Compounding the dilemma is the fact that many signs and symptoms associated with AFE are also clinical syndromes that can occur in its absence, such as anaphylactic shock, left ventricular failure, sepsis, adult respiratory distress syndrome, and diffuse intravascular coagulation. Most commonly, AFE is a diagnosis of exclusion, made during pregnancy or the immediate postpartum period. However, even this temporal association lends little support to the diagnosis of AFE, because any and all of these other clinical syndromes can and do occur in isolation during and after pregnancy.
The constellation of many of the clinical sequelae, rather than isolated signs and symptoms, is what many recognize as the sine qua non of AFE. However, this hypothesis is also slightly problematic because not all cases of AFE—especially nonfatal cases—present with multiorgan disease involving cardiac, respiratory, renal, neurologic, and hematologic failure.
Choose elective induction wisely
Despite the relative rarity of AFE, the phenomenon is worthy of attention because the incidence of elective labor induction has been increasing steadily. The absolute increase in the risk of AFE among women who undergo medical induction is very small: 4 or 5 cases of AFE and 1 or 2 cases of fatal AFE for every 100,000 labors induced. However, with nearly 4 million births annually and induction rates of 20% in the United States, the incidence of AFE could escalate to 30 to 40 cases a year and 10 to 15 deaths. This statistic is alarming, especially given the increasing use of elective medical induction. Extra caution in choosing elective induction is therefore justified.
1. Gilbert WM, Danielson B. Amniotic fluid embolism: decreased mortality in a population-based study. Obstet Gynecol. 1999;93:973-977.
2. Clark SL, Hankins GD, Dudley DA, et al. Amniotic fluid embolism: analysis of the national registry. Am J Obstet Gynecol. 1995;172:1158-1167.
3. Tuffnell DJ. Amniotic fluid embolism. Curr Opin Obstet Gynecol. 2003;15:119-122.
4. Killam A. Amniotic fluid embolism. Clin Obstet Gynecol. 1985;28:32-36.
5. Sperry K. Landmark perspective: amniotic fluid embolism. To understand an enigma. JAMA. 1986;255:2183-2186.
6. Tuffnell DJ. United Kingdom amniotic fluid embolism register. BJOG. 2005;112:1625-1629.
7. Moore J, Baldisseri MR. Amniotic fluid embolism. Crit Care Med. 2005;33:S279-285.
8. Turner LA, Kramer MS, Liu S. for the Maternal Mortality and Morbidity Study Group of the Canadian Perinatal Surveillance System. Cause-specific mortality during and after pregnancy and the definition of maternal death. Chronic Dis Can. 2005;23:31-36.
1. Gilbert WM, Danielson B. Amniotic fluid embolism: decreased mortality in a population-based study. Obstet Gynecol. 1999;93:973-977.
2. Clark SL, Hankins GD, Dudley DA, et al. Amniotic fluid embolism: analysis of the national registry. Am J Obstet Gynecol. 1995;172:1158-1167.
3. Tuffnell DJ. Amniotic fluid embolism. Curr Opin Obstet Gynecol. 2003;15:119-122.
4. Killam A. Amniotic fluid embolism. Clin Obstet Gynecol. 1985;28:32-36.
5. Sperry K. Landmark perspective: amniotic fluid embolism. To understand an enigma. JAMA. 1986;255:2183-2186.
6. Tuffnell DJ. United Kingdom amniotic fluid embolism register. BJOG. 2005;112:1625-1629.
7. Moore J, Baldisseri MR. Amniotic fluid embolism. Crit Care Med. 2005;33:S279-285.
8. Turner LA, Kramer MS, Liu S. for the Maternal Mortality and Morbidity Study Group of the Canadian Perinatal Surveillance System. Cause-specific mortality during and after pregnancy and the definition of maternal death. Chronic Dis Can. 2005;23:31-36.
Managing troublesome urethral diverticula
Urethral diverticula are often overlooked as a source of recurrent urinary tract infection, voiding dysfunction, dyspareunia, and chronic pelvic pain. Here, in brief, is how to diagnose and manage this condition, including a look at surgical options.
What are the common complaints?
Urethral diverticula present in myriad ways—most often, as recurrent urinary tract infection, overactive bladder, stress urinary incontinence, and pelvic pain. Other common presenting symptoms include voiding dysfunction, a painful or palpable mass, and postvoid dribbling.
What can be done routinely during a pelvic exam to make the Dx?
Become accustomed to massaging the anterior vaginal wall underneath the urethra. Any discharge or excretion of fluid that you observe from the external urethral meatus as you massage is pathognomonic for urethral diverticulum. In addition, palpate the anterior vaginal wall for paraurethral masses. Sometimes, a diverticulum is ballotable but not palpable.
Which test is best?
Imaging has been used in different ways, with variable success.
- Most diverticula are well visualized by voiding cystourethrography or magnetic resonance imaging (MRI); we view these as complementary techniques, in fact, because some diverticula are visualized only by one modality or the other. MRI provides a superior examination for surgical planning because it defines urethral and diverticular anatomy most clearly
- Ultrasonography has been used with some success
- Positive-pressure urethrography, using a Tratner or double balloon catheter, is difficult to perform and uncomfortable for the patient.
What is the role of urethroscopy?
We find urethroscopy very helpful. One caveat: Inability to visualize a diverticulum or its opening does not, by any means, exclude a urethral diverticulum.
How should you manage a urethral diverticulum?
- Urinary tract infection should be treated with a culture-specific antibiotic; in some cases, the patient will become asymptomatic afterwards
- Overactive bladder symptoms can be treated with an anticholinergic
- In most cases, surgery proves necessary
- When you identify a urethral diverticulum during pregnancy, manage the patient conservatively during the antenatal period
- A patient who has an asymptomatic urethral diverticulum can be managed expectantly, but perform a pelvic exam periodically.
When is surgery appropriate? By what method?
Several observations are useful:
- Hardness or induration of the diverticular mass is extremely rare; such a finding should prompt surgical excision because it may signal cancer
- Marsupialization has been demonstrated to be successful for very distal and small urethral diverticula
- Most diverticula at the level of the midurethra and proximal urethra require some form of excision, broadly classified as partial ablation or complete excision
- Placement of a suburethral sling is controversial, but some experts believe that, to prevent stress incontinence, this intervention should be undertaken simultaneously with any other surgical treatment for diverticula of the proximal urethra
- Sometimes a Martius fat pad must be brought into the field to avoid devascularization and breakdown of the repair. When a suburethral sling is necessary, we routinely place a Martius flap between the urethra and the sling.
Drs. Karram and Blaivas cochair the 6th Annual International Symposium on Female Urology & Urogynecology, to be held April 26–28, 2007 in Las Vegas (www.urogyn-cme.org).
Urethral diverticula are often overlooked as a source of recurrent urinary tract infection, voiding dysfunction, dyspareunia, and chronic pelvic pain. Here, in brief, is how to diagnose and manage this condition, including a look at surgical options.
What are the common complaints?
Urethral diverticula present in myriad ways—most often, as recurrent urinary tract infection, overactive bladder, stress urinary incontinence, and pelvic pain. Other common presenting symptoms include voiding dysfunction, a painful or palpable mass, and postvoid dribbling.
What can be done routinely during a pelvic exam to make the Dx?
Become accustomed to massaging the anterior vaginal wall underneath the urethra. Any discharge or excretion of fluid that you observe from the external urethral meatus as you massage is pathognomonic for urethral diverticulum. In addition, palpate the anterior vaginal wall for paraurethral masses. Sometimes, a diverticulum is ballotable but not palpable.
Which test is best?
Imaging has been used in different ways, with variable success.
- Most diverticula are well visualized by voiding cystourethrography or magnetic resonance imaging (MRI); we view these as complementary techniques, in fact, because some diverticula are visualized only by one modality or the other. MRI provides a superior examination for surgical planning because it defines urethral and diverticular anatomy most clearly
- Ultrasonography has been used with some success
- Positive-pressure urethrography, using a Tratner or double balloon catheter, is difficult to perform and uncomfortable for the patient.
What is the role of urethroscopy?
We find urethroscopy very helpful. One caveat: Inability to visualize a diverticulum or its opening does not, by any means, exclude a urethral diverticulum.
How should you manage a urethral diverticulum?
- Urinary tract infection should be treated with a culture-specific antibiotic; in some cases, the patient will become asymptomatic afterwards
- Overactive bladder symptoms can be treated with an anticholinergic
- In most cases, surgery proves necessary
- When you identify a urethral diverticulum during pregnancy, manage the patient conservatively during the antenatal period
- A patient who has an asymptomatic urethral diverticulum can be managed expectantly, but perform a pelvic exam periodically.
When is surgery appropriate? By what method?
Several observations are useful:
- Hardness or induration of the diverticular mass is extremely rare; such a finding should prompt surgical excision because it may signal cancer
- Marsupialization has been demonstrated to be successful for very distal and small urethral diverticula
- Most diverticula at the level of the midurethra and proximal urethra require some form of excision, broadly classified as partial ablation or complete excision
- Placement of a suburethral sling is controversial, but some experts believe that, to prevent stress incontinence, this intervention should be undertaken simultaneously with any other surgical treatment for diverticula of the proximal urethra
- Sometimes a Martius fat pad must be brought into the field to avoid devascularization and breakdown of the repair. When a suburethral sling is necessary, we routinely place a Martius flap between the urethra and the sling.
Urethral diverticula are often overlooked as a source of recurrent urinary tract infection, voiding dysfunction, dyspareunia, and chronic pelvic pain. Here, in brief, is how to diagnose and manage this condition, including a look at surgical options.
What are the common complaints?
Urethral diverticula present in myriad ways—most often, as recurrent urinary tract infection, overactive bladder, stress urinary incontinence, and pelvic pain. Other common presenting symptoms include voiding dysfunction, a painful or palpable mass, and postvoid dribbling.
What can be done routinely during a pelvic exam to make the Dx?
Become accustomed to massaging the anterior vaginal wall underneath the urethra. Any discharge or excretion of fluid that you observe from the external urethral meatus as you massage is pathognomonic for urethral diverticulum. In addition, palpate the anterior vaginal wall for paraurethral masses. Sometimes, a diverticulum is ballotable but not palpable.
Which test is best?
Imaging has been used in different ways, with variable success.
- Most diverticula are well visualized by voiding cystourethrography or magnetic resonance imaging (MRI); we view these as complementary techniques, in fact, because some diverticula are visualized only by one modality or the other. MRI provides a superior examination for surgical planning because it defines urethral and diverticular anatomy most clearly
- Ultrasonography has been used with some success
- Positive-pressure urethrography, using a Tratner or double balloon catheter, is difficult to perform and uncomfortable for the patient.
What is the role of urethroscopy?
We find urethroscopy very helpful. One caveat: Inability to visualize a diverticulum or its opening does not, by any means, exclude a urethral diverticulum.
How should you manage a urethral diverticulum?
- Urinary tract infection should be treated with a culture-specific antibiotic; in some cases, the patient will become asymptomatic afterwards
- Overactive bladder symptoms can be treated with an anticholinergic
- In most cases, surgery proves necessary
- When you identify a urethral diverticulum during pregnancy, manage the patient conservatively during the antenatal period
- A patient who has an asymptomatic urethral diverticulum can be managed expectantly, but perform a pelvic exam periodically.
When is surgery appropriate? By what method?
Several observations are useful:
- Hardness or induration of the diverticular mass is extremely rare; such a finding should prompt surgical excision because it may signal cancer
- Marsupialization has been demonstrated to be successful for very distal and small urethral diverticula
- Most diverticula at the level of the midurethra and proximal urethra require some form of excision, broadly classified as partial ablation or complete excision
- Placement of a suburethral sling is controversial, but some experts believe that, to prevent stress incontinence, this intervention should be undertaken simultaneously with any other surgical treatment for diverticula of the proximal urethra
- Sometimes a Martius fat pad must be brought into the field to avoid devascularization and breakdown of the repair. When a suburethral sling is necessary, we routinely place a Martius flap between the urethra and the sling.
Drs. Karram and Blaivas cochair the 6th Annual International Symposium on Female Urology & Urogynecology, to be held April 26–28, 2007 in Las Vegas (www.urogyn-cme.org).
Drs. Karram and Blaivas cochair the 6th Annual International Symposium on Female Urology & Urogynecology, to be held April 26–28, 2007 in Las Vegas (www.urogyn-cme.org).
Q Does prophylactic oophorectomy raise the risk of death?
Expert Commentary
A longstanding controversy in gynecologic practice is whether the ovaries should be removed at the time of abdominal hysterectomy. Depending on the patient’s age, this question requires the clinician to weigh the risk of subsequent ovarian cancer against the benefit of protection against cardiovascular disease and osteoporosis. During my training in the late 1970s, prophylactic oophorectomy was recommended at the time of hysterectomy if the patient was older than 40 years. The most recent guidelines (1999) from the American College of Obstetricians and Gynecologists1 state: “The decision to perform prophylactic oophorectomy should not be based only on age; it should be a highly individualized decision that takes into account several patient factors and choices.”
High risk of heart disease versus low risk of ovarian cancer
Given that the number of women older than 40 who will die of heart disease is vastly greater than the number who will die of ovarian cancer, even a small protective effect against heart disease from the retention of estrogen-producing ovaries might outweigh the potential risk of ovarian cancer. Other variables influence the outcome—eg, ovarian conservation reduces hip fracture but increases breast cancer—but the main drivers of overall outcome are heart disease and ovarian cancer.
How this study explored the issue
One way to address the question of risk is to model various outcomes using simulation methods, as discussed in a recent issue of Obg Management.2 Another way is to analyze retrospectively the survival across time of women who did or did not undergo oophorectomy. The study by Rocca and colleagues is such an analysis, using data from women residing in Olmstead County, Minnesota, between 1950 and 1987.
Women were included as cases if they had oophorectomy between 40 years and menopause; controls were age-matched women in the database who had survived without oophorectomy to the same index year as the case. Using these criteria, almost 1,100 women were identified who underwent bilateral oophorectomy between 40 years and menopause.
Life-table analysis revealed no difference between cases and controls in survival across time (hazard ratio, 1.05; 95% confidence interval, 0.92–1.20). However, a subgroup of 79 women who had bilateral prophylactic oophorectomy between 40 and 45 years, but who were not given estrogen, were estimated to have twice the mortality of their controls across time. Similarly, a group of 183 women who underwent bilateral oophorectomy for benign disease between 40 and 45 years had a 50% higher mortality. None of the subgroups of women who underwent bilateral oophorectomy after 45 years had increased mortality in comparison with controls.
What this means for clinical practice
It is safe to say that data from a randomized, controlled trial will not be forthcoming anytime soon. Therefore, we must rely on the careful analysis of retrospective data, as in the study by Rocca et al. From this analysis, 2 recommendations can be drawn:
- After age 45. It appears that oophorectomy after age 45 will not alter the subsequent overall mortality risk. For this age group, the decision to remove or retain the ovaries should be made on an individual basis depending on the risk profile and informed patient choice, as suggested by American College of Obstetricians and Gynecologists guidelines.1
- Prior to age 45. If bilateral oophorectomy is performed, estrogen replacement should be strongly considered. In contemporary practice, if alternatives to estrogen are desired, the patient should be monitored for evidence of preclinical cardiovascular disease and osteoporosis, and appropriate treatment should be initiated, if indicated.
Expert Commentary
A longstanding controversy in gynecologic practice is whether the ovaries should be removed at the time of abdominal hysterectomy. Depending on the patient’s age, this question requires the clinician to weigh the risk of subsequent ovarian cancer against the benefit of protection against cardiovascular disease and osteoporosis. During my training in the late 1970s, prophylactic oophorectomy was recommended at the time of hysterectomy if the patient was older than 40 years. The most recent guidelines (1999) from the American College of Obstetricians and Gynecologists1 state: “The decision to perform prophylactic oophorectomy should not be based only on age; it should be a highly individualized decision that takes into account several patient factors and choices.”
High risk of heart disease versus low risk of ovarian cancer
Given that the number of women older than 40 who will die of heart disease is vastly greater than the number who will die of ovarian cancer, even a small protective effect against heart disease from the retention of estrogen-producing ovaries might outweigh the potential risk of ovarian cancer. Other variables influence the outcome—eg, ovarian conservation reduces hip fracture but increases breast cancer—but the main drivers of overall outcome are heart disease and ovarian cancer.
How this study explored the issue
One way to address the question of risk is to model various outcomes using simulation methods, as discussed in a recent issue of Obg Management.2 Another way is to analyze retrospectively the survival across time of women who did or did not undergo oophorectomy. The study by Rocca and colleagues is such an analysis, using data from women residing in Olmstead County, Minnesota, between 1950 and 1987.
Women were included as cases if they had oophorectomy between 40 years and menopause; controls were age-matched women in the database who had survived without oophorectomy to the same index year as the case. Using these criteria, almost 1,100 women were identified who underwent bilateral oophorectomy between 40 years and menopause.
Life-table analysis revealed no difference between cases and controls in survival across time (hazard ratio, 1.05; 95% confidence interval, 0.92–1.20). However, a subgroup of 79 women who had bilateral prophylactic oophorectomy between 40 and 45 years, but who were not given estrogen, were estimated to have twice the mortality of their controls across time. Similarly, a group of 183 women who underwent bilateral oophorectomy for benign disease between 40 and 45 years had a 50% higher mortality. None of the subgroups of women who underwent bilateral oophorectomy after 45 years had increased mortality in comparison with controls.
What this means for clinical practice
It is safe to say that data from a randomized, controlled trial will not be forthcoming anytime soon. Therefore, we must rely on the careful analysis of retrospective data, as in the study by Rocca et al. From this analysis, 2 recommendations can be drawn:
- After age 45. It appears that oophorectomy after age 45 will not alter the subsequent overall mortality risk. For this age group, the decision to remove or retain the ovaries should be made on an individual basis depending on the risk profile and informed patient choice, as suggested by American College of Obstetricians and Gynecologists guidelines.1
- Prior to age 45. If bilateral oophorectomy is performed, estrogen replacement should be strongly considered. In contemporary practice, if alternatives to estrogen are desired, the patient should be monitored for evidence of preclinical cardiovascular disease and osteoporosis, and appropriate treatment should be initiated, if indicated.
Expert Commentary
A longstanding controversy in gynecologic practice is whether the ovaries should be removed at the time of abdominal hysterectomy. Depending on the patient’s age, this question requires the clinician to weigh the risk of subsequent ovarian cancer against the benefit of protection against cardiovascular disease and osteoporosis. During my training in the late 1970s, prophylactic oophorectomy was recommended at the time of hysterectomy if the patient was older than 40 years. The most recent guidelines (1999) from the American College of Obstetricians and Gynecologists1 state: “The decision to perform prophylactic oophorectomy should not be based only on age; it should be a highly individualized decision that takes into account several patient factors and choices.”
High risk of heart disease versus low risk of ovarian cancer
Given that the number of women older than 40 who will die of heart disease is vastly greater than the number who will die of ovarian cancer, even a small protective effect against heart disease from the retention of estrogen-producing ovaries might outweigh the potential risk of ovarian cancer. Other variables influence the outcome—eg, ovarian conservation reduces hip fracture but increases breast cancer—but the main drivers of overall outcome are heart disease and ovarian cancer.
How this study explored the issue
One way to address the question of risk is to model various outcomes using simulation methods, as discussed in a recent issue of Obg Management.2 Another way is to analyze retrospectively the survival across time of women who did or did not undergo oophorectomy. The study by Rocca and colleagues is such an analysis, using data from women residing in Olmstead County, Minnesota, between 1950 and 1987.
Women were included as cases if they had oophorectomy between 40 years and menopause; controls were age-matched women in the database who had survived without oophorectomy to the same index year as the case. Using these criteria, almost 1,100 women were identified who underwent bilateral oophorectomy between 40 years and menopause.
Life-table analysis revealed no difference between cases and controls in survival across time (hazard ratio, 1.05; 95% confidence interval, 0.92–1.20). However, a subgroup of 79 women who had bilateral prophylactic oophorectomy between 40 and 45 years, but who were not given estrogen, were estimated to have twice the mortality of their controls across time. Similarly, a group of 183 women who underwent bilateral oophorectomy for benign disease between 40 and 45 years had a 50% higher mortality. None of the subgroups of women who underwent bilateral oophorectomy after 45 years had increased mortality in comparison with controls.
What this means for clinical practice
It is safe to say that data from a randomized, controlled trial will not be forthcoming anytime soon. Therefore, we must rely on the careful analysis of retrospective data, as in the study by Rocca et al. From this analysis, 2 recommendations can be drawn:
- After age 45. It appears that oophorectomy after age 45 will not alter the subsequent overall mortality risk. For this age group, the decision to remove or retain the ovaries should be made on an individual basis depending on the risk profile and informed patient choice, as suggested by American College of Obstetricians and Gynecologists guidelines.1
- Prior to age 45. If bilateral oophorectomy is performed, estrogen replacement should be strongly considered. In contemporary practice, if alternatives to estrogen are desired, the patient should be monitored for evidence of preclinical cardiovascular disease and osteoporosis, and appropriate treatment should be initiated, if indicated.
Q Does Botox relieve urinary urgency and urge incontinence?
<huc>A</huc> Yes, but improvements are time-limited. In this series of 100 cases of idiopathic detrusor overactivity treated with botulinum toxin A (Botox), symptoms resolved in 74% of patients with urge incontinence by 4 weeks after treatment, and in 80% of patients by 12 weeks after treatment. Urgency resolved in 72% at 4 weeks and 66% at 12 weeks.
Expert Commentary
This fairly large case series helps clarify the potential of Botox to ease symptoms in patients who do not respond to current therapies for idiopathic detrusor overactivity.
Among the first authors to report successful use of Botox for neurogenic detrusor overactivity was a research team from Switzerland led by Brigitte Schurch—the same group that published this case series. Participants met detailed criteria for “overactive bladder syndrome,” as defined by the International Continence Society, with either (1) urodynamically demonstrated nonneurogenic (idiopathic) detrusor overactivity with or without incontinence or (2) hypersensitive bladder with premature filling sensation (even when maximum bladder capacity was normal) and more than 8 voids per 24 hours (urgency–frequency syndrome with or without incontinence).
Participants included 77 women and 23 men, each of whom received 100 U of Botox diluted with saline and injected at 30 sites in the bladder under cystoscopic guidance, sparing the trigone.
Risks versus benefits
Adverse events reported in this study included urinary tract infections in 10 patients (10%) and urinary retention requiring intermittent self-catheterization in 4 patients (4%). However, retention was otherwise not well characterized. Mean postvoid residual did not return to baseline values until 9 months after injection.
Why these data are imperfect
Although the study included both women and men (ratio about 3:1), data were not reported by sex. Moreover, according to the International Continence Society definition, patients with overactive bladder syndrome constitute a heterogeneous group based on urinary symptoms (which may or may not include urge incontinence) and urodynamic findings (which may or may not include detrusor overactivity).
A small percentage of patients had apparent detrusor hypocontractility or acontractility with elevated postvoid residual urine volumes—a group clinically distinct from patients with otherwise “normal” bladder emptying despite symptoms attributed to the detrusor muscle.
A robust placebo effect is likely
In this open-label series, as in clinical practice, both patients and clinicians expected treatment to be beneficial—raising the possibility of a placebo effect that may explain part (or most) of the improvement. In many placebo-controlled drug trials involving patients with detrusor overactivity, a relatively large portion of the “treatment effect” may be attributed to the placebo effect; as many as 40% of patients taking placebo report some relief of symptoms.
Beyond beauty: Botox for the bladder
Under cystoscopic guidance, Schmid et al injected 100 U of saline-diluted Botox (tinted with indigo carmine to aid in spacing) at 30 sites in the bladder, excluding the trigone. Most women reported temporary relief of symptoms.
More data are needed
To address these gaps in knowledge, especially as use of Botox is being rapidly incorporated into clinical practice, investigators in the NIH-sponsored Pelvic Floor Disorders Network are conducting a randomized, placebo-controlled trial in women with idiopathic detrusor overactivity incontinence. The aim of the trial: to describe the percentage of women whose symptoms resolve, followed by time to recurrence, after treatment with Botox versus placebo, with 1 year of follow-up.
To obtain focused data, inclusion and exclusion criteria were developed to ensure that the population of this triple-blinded study is relatively homogeneous. As the occurrence and consequences of urinary retention have not been well described in prior studies, subjects in the NIH trial are being carefully assessed for urinary retention. Enrollment should be complete by mid-2007, and follow-up by mid-2008.
Should ObGyns consider Botox for women with intractable symptoms?
For clinicians experienced with cystoscopically guided injections, Botox might be a reasonable option for highly selected women who are truly refractory to all other treatments for detrusor overactivity incontinence. Such women should understand that Botox is not FDA-approved for this indication and that its use is experimental. No evidence identifies the optimal dose of Botox to be injected cystoscopically for incontinence related to detrusor overactivity. However, dosing information may be forthcoming from a Phase II study being performed by the company that markets Botox (Allergan).
Greatest risk is urinary retention
The most important immediate or short-term risk is probably urinary retention. Although experienced clinicians have estimated a low risk of retention, a much higher rate can be found when the postvoid residual is measured routinely after Botox injection. It still seems likely that Botox-associated retention is temporary, but it may last as long as the effect on symptoms, on the order of several months. Women should be counseled carefully to be sure they understand this.
Further, because urinary sensation may be altered after Botox, women may not experience bothersome symptoms of retention. Nevertheless, more data on possible short- and long-term consequences are needed before changing clinical management of this type of retention (ie, I recommend continuing to treat retention rather than watchful waiting).
Until more data are available on risks versus benefits, I recommend against using Botox in women at high risk for complications associated with partial urinary retention. I would be particularly concerned about elderly women at risk of urinary tract infection that may lead to urosepsis.
<huc>A</huc> Yes, but improvements are time-limited. In this series of 100 cases of idiopathic detrusor overactivity treated with botulinum toxin A (Botox), symptoms resolved in 74% of patients with urge incontinence by 4 weeks after treatment, and in 80% of patients by 12 weeks after treatment. Urgency resolved in 72% at 4 weeks and 66% at 12 weeks.
Expert Commentary
This fairly large case series helps clarify the potential of Botox to ease symptoms in patients who do not respond to current therapies for idiopathic detrusor overactivity.
Among the first authors to report successful use of Botox for neurogenic detrusor overactivity was a research team from Switzerland led by Brigitte Schurch—the same group that published this case series. Participants met detailed criteria for “overactive bladder syndrome,” as defined by the International Continence Society, with either (1) urodynamically demonstrated nonneurogenic (idiopathic) detrusor overactivity with or without incontinence or (2) hypersensitive bladder with premature filling sensation (even when maximum bladder capacity was normal) and more than 8 voids per 24 hours (urgency–frequency syndrome with or without incontinence).
Participants included 77 women and 23 men, each of whom received 100 U of Botox diluted with saline and injected at 30 sites in the bladder under cystoscopic guidance, sparing the trigone.
Risks versus benefits
Adverse events reported in this study included urinary tract infections in 10 patients (10%) and urinary retention requiring intermittent self-catheterization in 4 patients (4%). However, retention was otherwise not well characterized. Mean postvoid residual did not return to baseline values until 9 months after injection.
Why these data are imperfect
Although the study included both women and men (ratio about 3:1), data were not reported by sex. Moreover, according to the International Continence Society definition, patients with overactive bladder syndrome constitute a heterogeneous group based on urinary symptoms (which may or may not include urge incontinence) and urodynamic findings (which may or may not include detrusor overactivity).
A small percentage of patients had apparent detrusor hypocontractility or acontractility with elevated postvoid residual urine volumes—a group clinically distinct from patients with otherwise “normal” bladder emptying despite symptoms attributed to the detrusor muscle.
A robust placebo effect is likely
In this open-label series, as in clinical practice, both patients and clinicians expected treatment to be beneficial—raising the possibility of a placebo effect that may explain part (or most) of the improvement. In many placebo-controlled drug trials involving patients with detrusor overactivity, a relatively large portion of the “treatment effect” may be attributed to the placebo effect; as many as 40% of patients taking placebo report some relief of symptoms.
Beyond beauty: Botox for the bladder
Under cystoscopic guidance, Schmid et al injected 100 U of saline-diluted Botox (tinted with indigo carmine to aid in spacing) at 30 sites in the bladder, excluding the trigone. Most women reported temporary relief of symptoms.
More data are needed
To address these gaps in knowledge, especially as use of Botox is being rapidly incorporated into clinical practice, investigators in the NIH-sponsored Pelvic Floor Disorders Network are conducting a randomized, placebo-controlled trial in women with idiopathic detrusor overactivity incontinence. The aim of the trial: to describe the percentage of women whose symptoms resolve, followed by time to recurrence, after treatment with Botox versus placebo, with 1 year of follow-up.
To obtain focused data, inclusion and exclusion criteria were developed to ensure that the population of this triple-blinded study is relatively homogeneous. As the occurrence and consequences of urinary retention have not been well described in prior studies, subjects in the NIH trial are being carefully assessed for urinary retention. Enrollment should be complete by mid-2007, and follow-up by mid-2008.
Should ObGyns consider Botox for women with intractable symptoms?
For clinicians experienced with cystoscopically guided injections, Botox might be a reasonable option for highly selected women who are truly refractory to all other treatments for detrusor overactivity incontinence. Such women should understand that Botox is not FDA-approved for this indication and that its use is experimental. No evidence identifies the optimal dose of Botox to be injected cystoscopically for incontinence related to detrusor overactivity. However, dosing information may be forthcoming from a Phase II study being performed by the company that markets Botox (Allergan).
Greatest risk is urinary retention
The most important immediate or short-term risk is probably urinary retention. Although experienced clinicians have estimated a low risk of retention, a much higher rate can be found when the postvoid residual is measured routinely after Botox injection. It still seems likely that Botox-associated retention is temporary, but it may last as long as the effect on symptoms, on the order of several months. Women should be counseled carefully to be sure they understand this.
Further, because urinary sensation may be altered after Botox, women may not experience bothersome symptoms of retention. Nevertheless, more data on possible short- and long-term consequences are needed before changing clinical management of this type of retention (ie, I recommend continuing to treat retention rather than watchful waiting).
Until more data are available on risks versus benefits, I recommend against using Botox in women at high risk for complications associated with partial urinary retention. I would be particularly concerned about elderly women at risk of urinary tract infection that may lead to urosepsis.
<huc>A</huc> Yes, but improvements are time-limited. In this series of 100 cases of idiopathic detrusor overactivity treated with botulinum toxin A (Botox), symptoms resolved in 74% of patients with urge incontinence by 4 weeks after treatment, and in 80% of patients by 12 weeks after treatment. Urgency resolved in 72% at 4 weeks and 66% at 12 weeks.
Expert Commentary
This fairly large case series helps clarify the potential of Botox to ease symptoms in patients who do not respond to current therapies for idiopathic detrusor overactivity.
Among the first authors to report successful use of Botox for neurogenic detrusor overactivity was a research team from Switzerland led by Brigitte Schurch—the same group that published this case series. Participants met detailed criteria for “overactive bladder syndrome,” as defined by the International Continence Society, with either (1) urodynamically demonstrated nonneurogenic (idiopathic) detrusor overactivity with or without incontinence or (2) hypersensitive bladder with premature filling sensation (even when maximum bladder capacity was normal) and more than 8 voids per 24 hours (urgency–frequency syndrome with or without incontinence).
Participants included 77 women and 23 men, each of whom received 100 U of Botox diluted with saline and injected at 30 sites in the bladder under cystoscopic guidance, sparing the trigone.
Risks versus benefits
Adverse events reported in this study included urinary tract infections in 10 patients (10%) and urinary retention requiring intermittent self-catheterization in 4 patients (4%). However, retention was otherwise not well characterized. Mean postvoid residual did not return to baseline values until 9 months after injection.
Why these data are imperfect
Although the study included both women and men (ratio about 3:1), data were not reported by sex. Moreover, according to the International Continence Society definition, patients with overactive bladder syndrome constitute a heterogeneous group based on urinary symptoms (which may or may not include urge incontinence) and urodynamic findings (which may or may not include detrusor overactivity).
A small percentage of patients had apparent detrusor hypocontractility or acontractility with elevated postvoid residual urine volumes—a group clinically distinct from patients with otherwise “normal” bladder emptying despite symptoms attributed to the detrusor muscle.
A robust placebo effect is likely
In this open-label series, as in clinical practice, both patients and clinicians expected treatment to be beneficial—raising the possibility of a placebo effect that may explain part (or most) of the improvement. In many placebo-controlled drug trials involving patients with detrusor overactivity, a relatively large portion of the “treatment effect” may be attributed to the placebo effect; as many as 40% of patients taking placebo report some relief of symptoms.
Beyond beauty: Botox for the bladder
Under cystoscopic guidance, Schmid et al injected 100 U of saline-diluted Botox (tinted with indigo carmine to aid in spacing) at 30 sites in the bladder, excluding the trigone. Most women reported temporary relief of symptoms.
More data are needed
To address these gaps in knowledge, especially as use of Botox is being rapidly incorporated into clinical practice, investigators in the NIH-sponsored Pelvic Floor Disorders Network are conducting a randomized, placebo-controlled trial in women with idiopathic detrusor overactivity incontinence. The aim of the trial: to describe the percentage of women whose symptoms resolve, followed by time to recurrence, after treatment with Botox versus placebo, with 1 year of follow-up.
To obtain focused data, inclusion and exclusion criteria were developed to ensure that the population of this triple-blinded study is relatively homogeneous. As the occurrence and consequences of urinary retention have not been well described in prior studies, subjects in the NIH trial are being carefully assessed for urinary retention. Enrollment should be complete by mid-2007, and follow-up by mid-2008.
Should ObGyns consider Botox for women with intractable symptoms?
For clinicians experienced with cystoscopically guided injections, Botox might be a reasonable option for highly selected women who are truly refractory to all other treatments for detrusor overactivity incontinence. Such women should understand that Botox is not FDA-approved for this indication and that its use is experimental. No evidence identifies the optimal dose of Botox to be injected cystoscopically for incontinence related to detrusor overactivity. However, dosing information may be forthcoming from a Phase II study being performed by the company that markets Botox (Allergan).
Greatest risk is urinary retention
The most important immediate or short-term risk is probably urinary retention. Although experienced clinicians have estimated a low risk of retention, a much higher rate can be found when the postvoid residual is measured routinely after Botox injection. It still seems likely that Botox-associated retention is temporary, but it may last as long as the effect on symptoms, on the order of several months. Women should be counseled carefully to be sure they understand this.
Further, because urinary sensation may be altered after Botox, women may not experience bothersome symptoms of retention. Nevertheless, more data on possible short- and long-term consequences are needed before changing clinical management of this type of retention (ie, I recommend continuing to treat retention rather than watchful waiting).
Until more data are available on risks versus benefits, I recommend against using Botox in women at high risk for complications associated with partial urinary retention. I would be particularly concerned about elderly women at risk of urinary tract infection that may lead to urosepsis.