User login
Can we reduce the rate of scheduled births that occur earlier than 39 weeks of gestation?
This interesting study from the Ohio Perinatal Quality Collaborative (OPQC) tackles an important topic—reducing the number of scheduled deliveries that take place before 39 weeks’ gestation. Infants born before 39 weeks have a higher rate of NICU admission in addition to measurable morbidity. The March of Dimes and other organizations have recognized the reduction of these births as an important goal, and ACOG recently refined and clarified the medical indications for them.1
Details of the study
Twenty maternity hospitals in Ohio collected baseline data on the rate of scheduled delivery between 36.0 and 38.6 weeks’ gestation over a 60-day period. Members of the OPQC (which included perinatologists) then visited each participating hospital to introduce the project and explain how it would be sustained using techniques from the Institute for Healthcare Improvement Breakthrough Series.2 Each hospital developed a three-person team—a physician, a nurse, and a data manager—to lead the effort. OPQC faculty developed a list of interventions, including the following broad techniques:
- promote optimal determination of gestational age using ultrasonography (US)
- use ACOG criteria for the indication and timing of scheduled births
- educate physicians, nurses, and pregnant women of the risks and benefits of birth between 36.0 and 38.6 weeks’ gestation
- improve communication between obstetricians and pediatricians through chart documentation of clear patient hand-offs and monthly reporting of statistics to physicians, nurses, and administrators
- include scheduled births in an overall “culture of safety,” with regular discussion of the matter at department and quality meetings.
I offer five key points about any program to reduce the number of scheduled births between 36.0 and 38.6 weeks’ gestation:
- it requires a lot of work and commitment on the part of all members of the health care team to change a behavior. Educate team members about the goal and the reasons behind it, and encourage them to contribute ideas to achieve it
- consider cooperating with state agencies to obtain data that can be used as baseline information and to measure the effect of an intervention
- a “one size fits all” approach is unlikely to be effective. Each hospital and physician is unique, and interventions should be individualized
- although the result of this 14-month study is impressive, real change must be maintained over the long term. Keep this in mind when you are planning interventions
- efforts such as this one have a financial cost associated with them, and I would hope that the government would contribute funding.
—GEORGE A. MACONES, MD, MSCE
Staff at participating hospitals had much latitude in selecting and refining interventions for their institution. After implementation of specific initiatives, each hospital tracked and reported relevant outcomes, especially the rate of scheduled deliveries between 36.0 and 38.6 weeks’ gestation. Each hospital received a monthly report, and staff members were encouraged to share the results internally.
Dramatic result was validated
The result of this initiative is impressive: The rate of inappropriate scheduled births was reduced from 25% to less than 5% over 14 months. This finding was validated in two ways:
- by checking Ohio birth certificate data
- by determining whether there was a concomitant rise in the rate of deliveries between 39 and 41 weeks’ gestation in the same hospitals.
Both validations confirmed that the OPQC interventions were significantly effective.
Project was not truly “statewide”
No study is perfect. A potential limitation of this study is the fact that it did not include all hospitals in the state of Ohio—only a sampling of larger hospitals. This makes it unclear whether the approach would be as effective in smaller institutions. Moreover, it is possible that physicians simply got better at documenting a reason for scheduled delivery as the study progressed—although I doubt that this reason alone would explain the striking findings.
1. American College of Obstetricians and Gynecologists Committee on Practice Bulletins—Obstetrics. ACOG Practice Bulletin#107, August 2009: Induction of labor. Obstet Gynecol. 2009;114(2, part 1):386-397.
2. The Breakthrough Series: IHI’s Collaborative Model for Achieving Breakthrough Improvement. IHI Innovation Series white paper. Boston: Institute for Healthcare Improvement Website. 2003. http://www.ihi.org/IHI/Results/WhitePapers/TheBreakthroughSeriesIHIsCollaborativeModelforAchieving+BreakthroughImprovement.htm. Accessed May 4, 2010.
This interesting study from the Ohio Perinatal Quality Collaborative (OPQC) tackles an important topic—reducing the number of scheduled deliveries that take place before 39 weeks’ gestation. Infants born before 39 weeks have a higher rate of NICU admission in addition to measurable morbidity. The March of Dimes and other organizations have recognized the reduction of these births as an important goal, and ACOG recently refined and clarified the medical indications for them.1
Details of the study
Twenty maternity hospitals in Ohio collected baseline data on the rate of scheduled delivery between 36.0 and 38.6 weeks’ gestation over a 60-day period. Members of the OPQC (which included perinatologists) then visited each participating hospital to introduce the project and explain how it would be sustained using techniques from the Institute for Healthcare Improvement Breakthrough Series.2 Each hospital developed a three-person team—a physician, a nurse, and a data manager—to lead the effort. OPQC faculty developed a list of interventions, including the following broad techniques:
- promote optimal determination of gestational age using ultrasonography (US)
- use ACOG criteria for the indication and timing of scheduled births
- educate physicians, nurses, and pregnant women of the risks and benefits of birth between 36.0 and 38.6 weeks’ gestation
- improve communication between obstetricians and pediatricians through chart documentation of clear patient hand-offs and monthly reporting of statistics to physicians, nurses, and administrators
- include scheduled births in an overall “culture of safety,” with regular discussion of the matter at department and quality meetings.
I offer five key points about any program to reduce the number of scheduled births between 36.0 and 38.6 weeks’ gestation:
- it requires a lot of work and commitment on the part of all members of the health care team to change a behavior. Educate team members about the goal and the reasons behind it, and encourage them to contribute ideas to achieve it
- consider cooperating with state agencies to obtain data that can be used as baseline information and to measure the effect of an intervention
- a “one size fits all” approach is unlikely to be effective. Each hospital and physician is unique, and interventions should be individualized
- although the result of this 14-month study is impressive, real change must be maintained over the long term. Keep this in mind when you are planning interventions
- efforts such as this one have a financial cost associated with them, and I would hope that the government would contribute funding.
—GEORGE A. MACONES, MD, MSCE
Staff at participating hospitals had much latitude in selecting and refining interventions for their institution. After implementation of specific initiatives, each hospital tracked and reported relevant outcomes, especially the rate of scheduled deliveries between 36.0 and 38.6 weeks’ gestation. Each hospital received a monthly report, and staff members were encouraged to share the results internally.
Dramatic result was validated
The result of this initiative is impressive: The rate of inappropriate scheduled births was reduced from 25% to less than 5% over 14 months. This finding was validated in two ways:
- by checking Ohio birth certificate data
- by determining whether there was a concomitant rise in the rate of deliveries between 39 and 41 weeks’ gestation in the same hospitals.
Both validations confirmed that the OPQC interventions were significantly effective.
Project was not truly “statewide”
No study is perfect. A potential limitation of this study is the fact that it did not include all hospitals in the state of Ohio—only a sampling of larger hospitals. This makes it unclear whether the approach would be as effective in smaller institutions. Moreover, it is possible that physicians simply got better at documenting a reason for scheduled delivery as the study progressed—although I doubt that this reason alone would explain the striking findings.
This interesting study from the Ohio Perinatal Quality Collaborative (OPQC) tackles an important topic—reducing the number of scheduled deliveries that take place before 39 weeks’ gestation. Infants born before 39 weeks have a higher rate of NICU admission in addition to measurable morbidity. The March of Dimes and other organizations have recognized the reduction of these births as an important goal, and ACOG recently refined and clarified the medical indications for them.1
Details of the study
Twenty maternity hospitals in Ohio collected baseline data on the rate of scheduled delivery between 36.0 and 38.6 weeks’ gestation over a 60-day period. Members of the OPQC (which included perinatologists) then visited each participating hospital to introduce the project and explain how it would be sustained using techniques from the Institute for Healthcare Improvement Breakthrough Series.2 Each hospital developed a three-person team—a physician, a nurse, and a data manager—to lead the effort. OPQC faculty developed a list of interventions, including the following broad techniques:
- promote optimal determination of gestational age using ultrasonography (US)
- use ACOG criteria for the indication and timing of scheduled births
- educate physicians, nurses, and pregnant women of the risks and benefits of birth between 36.0 and 38.6 weeks’ gestation
- improve communication between obstetricians and pediatricians through chart documentation of clear patient hand-offs and monthly reporting of statistics to physicians, nurses, and administrators
- include scheduled births in an overall “culture of safety,” with regular discussion of the matter at department and quality meetings.
I offer five key points about any program to reduce the number of scheduled births between 36.0 and 38.6 weeks’ gestation:
- it requires a lot of work and commitment on the part of all members of the health care team to change a behavior. Educate team members about the goal and the reasons behind it, and encourage them to contribute ideas to achieve it
- consider cooperating with state agencies to obtain data that can be used as baseline information and to measure the effect of an intervention
- a “one size fits all” approach is unlikely to be effective. Each hospital and physician is unique, and interventions should be individualized
- although the result of this 14-month study is impressive, real change must be maintained over the long term. Keep this in mind when you are planning interventions
- efforts such as this one have a financial cost associated with them, and I would hope that the government would contribute funding.
—GEORGE A. MACONES, MD, MSCE
Staff at participating hospitals had much latitude in selecting and refining interventions for their institution. After implementation of specific initiatives, each hospital tracked and reported relevant outcomes, especially the rate of scheduled deliveries between 36.0 and 38.6 weeks’ gestation. Each hospital received a monthly report, and staff members were encouraged to share the results internally.
Dramatic result was validated
The result of this initiative is impressive: The rate of inappropriate scheduled births was reduced from 25% to less than 5% over 14 months. This finding was validated in two ways:
- by checking Ohio birth certificate data
- by determining whether there was a concomitant rise in the rate of deliveries between 39 and 41 weeks’ gestation in the same hospitals.
Both validations confirmed that the OPQC interventions were significantly effective.
Project was not truly “statewide”
No study is perfect. A potential limitation of this study is the fact that it did not include all hospitals in the state of Ohio—only a sampling of larger hospitals. This makes it unclear whether the approach would be as effective in smaller institutions. Moreover, it is possible that physicians simply got better at documenting a reason for scheduled delivery as the study progressed—although I doubt that this reason alone would explain the striking findings.
1. American College of Obstetricians and Gynecologists Committee on Practice Bulletins—Obstetrics. ACOG Practice Bulletin#107, August 2009: Induction of labor. Obstet Gynecol. 2009;114(2, part 1):386-397.
2. The Breakthrough Series: IHI’s Collaborative Model for Achieving Breakthrough Improvement. IHI Innovation Series white paper. Boston: Institute for Healthcare Improvement Website. 2003. http://www.ihi.org/IHI/Results/WhitePapers/TheBreakthroughSeriesIHIsCollaborativeModelforAchieving+BreakthroughImprovement.htm. Accessed May 4, 2010.
1. American College of Obstetricians and Gynecologists Committee on Practice Bulletins—Obstetrics. ACOG Practice Bulletin#107, August 2009: Induction of labor. Obstet Gynecol. 2009;114(2, part 1):386-397.
2. The Breakthrough Series: IHI’s Collaborative Model for Achieving Breakthrough Improvement. IHI Innovation Series white paper. Boston: Institute for Healthcare Improvement Website. 2003. http://www.ihi.org/IHI/Results/WhitePapers/TheBreakthroughSeriesIHIsCollaborativeModelforAchieving+BreakthroughImprovement.htm. Accessed May 4, 2010.
A new treatment for menorrhagia enters the US market
CASE: Heavy bleeding and apprehension over hormones
Your patient, R.L., 34 years old, has been bothered by gradually worsening menorrhagia for 2 years when she schedules an appointment to discuss treatment options. She prefers to avoid surgery, if at all possible, and is reluctant to use hormonal medications because she’s concerned about side effects. Nonsteroidal anti-inflammatory drugs provide inadequate relief.
R.L. asks if there is any other agent that can reduce her menstrual blood loss.
What can you offer to her?
Heavy menstrual bleeding (menorrhagia) impairs quality of life and prompts many women such as R.L. to seek a gynecologic consultation—many of them hoping to avoid surgery.
In the United States, medical-management options for the treatment of heavy menstrual bleeding include nonsteroidal anti-inflammatory drugs (NSAIDs) and off-label use of oral contraceptives, injectable depot medroxyprogesterone acetate, and high-dose oral progestin (e.g., norethindrone acetate, 5-mg tablets). The levonorgestrel intrauterine system (LNG-IUS; Mirena) is also widely used to treat heavy menstrual bleeding in women and was approved for this indication in October 2009. FDA approval of the LNG-IUS for heavy menstrual bleeding was based on a pivotal randomized trial demonstrating high efficacy.1,2
Very soon an additional option will be available—the antifibrinolytic agent tranexamic acid (to be sold by Ferring Pharmaceuticals under the brand name Lysteda). Although the LNG-IUS remains first-line treatment for heavy menstrual bleeding, tranexamic acid is appropriate for women who need to, or prefer to, avoid hormones.
How tranexamic acid works
Women who experience heavy menstrual bleeding have an elevated level of plasminogen activators in the endometrium, enzymes that facilitate the dissolution of clots. These enzymes can be inhibited by antifibrinolytic agents such as tranexamic acid, which competitively blocks the conversion of plasminogen to plasmin, thereby reducing fibrinolysis (FIGURE).3
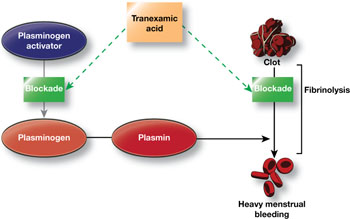
FIGURE How tranexamic acid stanches heavy menstrual bleeding
Tranexamic acid (Lysteda) competitively blocks the conversion of plasminogen to plasmin, thereby reducing fibrinolysis.Clinical trials of tranexamic acid to treat menorrhagia were first performed in the 1960s. The agent proved to be superior to placebo, NSAIDs, and cyclic oral progestin.4 However, the LNG-IUS has been found to be more effective than tranexamic acid in reducing menstrual blood loss.5
In the United States, until very recently, tranexamic acid was FDA-indicated only as an injectable agent for use at the time of dental extraction in hemophilic patients. However, an oral formulation has been widely used abroad, including in Canada, to treat heavy menstrual bleeding. In Sweden, the agent is available over the counter.
In November 2009, the FDA approved 650-mg tablets of tranexamic acid as a treatment for heavy menstrual bleeding.6
Recommended dosage
In women who have normal renal function, the recommended dosage of tranexamic acid is 1,300 mg (two 650-mg tablets) three times daily (a total of 3,900 mg) for as long as 5 days during menstruation. In women who have impaired renal function, the dosage should be adjusted according to the package insert.
In randomized clinical trials, tranexamic acid did not produce more side effects than placebo did. However, because of concerns that tranexamic acid may increase risk of venous thromboembolism (VTE), the agent is contraindicated in women who have a history of VTE.
Although women who use hormonal contraception have not been included in clinical trials of tranexamic acid, the FDA recommends that they take tranexamic acid only if there is “a strong medical need and the benefit of treatment will outweigh the potential increased risk of a thrombotic event.”6
Tranexamic acid will likely be of particular utility for women who desire treatment for heavy menstrual bleeding but who should, or would prefer to, avoid hormonal agents. British guidelines recommend that, when medical management is appropriate in the treatment of heavy menstrual bleeding, the LNG-IUS be considered first-line treatment, with tranexamic acid representing a second-line strategy.7
CASE: Resolved
When she hears about tranexamic acid, R.L. is eager to try it. She is pleased that it does not contain hormones and needs to be taken only 5 days or fewer each month. You give her a prescription and schedule a follow-up visit 3 months later, at which time she reports being satisfied with the drug.
1. FDA approves additional use for IUD Mirena to treat heavy menstrual bleeding in IUD users [press release]. Rockville, Md: FDA; Oct. 1, 2009.http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm184747.htm. Accessed May 3, 2010.
2. Kaunitz AM, Bissonette F, Monteiro I, Lukkari-Lax E, Muysers C, Jensen J. Levonorgestrel intrauterine system and medroxyprogesterone acetate for treatment of heavy menstrual bleeding. Poster No. 49 Presented at Reproductive Health 2009, Association of Reproductive Health Professionals annual meeting, September 30–October 3, 2009, Los Angeles, Calif. Contraception. 2009;80(2):212.-
3. Grimes DA, Davis AR, Ramos DE. Heavy menstrual bleeding: Assessing impact, evaluating management options. OBG Management. 2009;21(10)(supple).-http://www.obgmanagement.com/article_pages.asp?filename="2206OBG_Article2" aid=7971. Accessed May 3, 2010.
4. Lethaby A, Farquhar C, Cooke I. Antifibrinolytics for heavy menstrual bleeding. Cochrane Database Syst Rev. 2000;(4):CD000249.-
5. Milsom I, Andersson K, Andersch B, Rybo G. A comparison of flurbiprofen, tranexamic acid, and a levonorgestrel-releasing intrauterine contraceptive device in the treatment of idiopathic menorrhagia. Am J Obstet Gynecol. 1991;164(3):879-883.
6. FDA approves Lysteda to treat heavy menstrual bleeding [press release]. Rockville, Md: FDA; Nov. 13, 2009.http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm190551.htm. Accessed May 3, 2010.
7. National Institute for Health and Clinical Excellence. Heavy menstrual bleeding. January 2007.http://www.nice.org.uk/CG44. Accessed May 3, 2010.
CASE: Heavy bleeding and apprehension over hormones
Your patient, R.L., 34 years old, has been bothered by gradually worsening menorrhagia for 2 years when she schedules an appointment to discuss treatment options. She prefers to avoid surgery, if at all possible, and is reluctant to use hormonal medications because she’s concerned about side effects. Nonsteroidal anti-inflammatory drugs provide inadequate relief.
R.L. asks if there is any other agent that can reduce her menstrual blood loss.
What can you offer to her?
Heavy menstrual bleeding (menorrhagia) impairs quality of life and prompts many women such as R.L. to seek a gynecologic consultation—many of them hoping to avoid surgery.
In the United States, medical-management options for the treatment of heavy menstrual bleeding include nonsteroidal anti-inflammatory drugs (NSAIDs) and off-label use of oral contraceptives, injectable depot medroxyprogesterone acetate, and high-dose oral progestin (e.g., norethindrone acetate, 5-mg tablets). The levonorgestrel intrauterine system (LNG-IUS; Mirena) is also widely used to treat heavy menstrual bleeding in women and was approved for this indication in October 2009. FDA approval of the LNG-IUS for heavy menstrual bleeding was based on a pivotal randomized trial demonstrating high efficacy.1,2
Very soon an additional option will be available—the antifibrinolytic agent tranexamic acid (to be sold by Ferring Pharmaceuticals under the brand name Lysteda). Although the LNG-IUS remains first-line treatment for heavy menstrual bleeding, tranexamic acid is appropriate for women who need to, or prefer to, avoid hormones.
How tranexamic acid works
Women who experience heavy menstrual bleeding have an elevated level of plasminogen activators in the endometrium, enzymes that facilitate the dissolution of clots. These enzymes can be inhibited by antifibrinolytic agents such as tranexamic acid, which competitively blocks the conversion of plasminogen to plasmin, thereby reducing fibrinolysis (FIGURE).3

FIGURE How tranexamic acid stanches heavy menstrual bleeding
Tranexamic acid (Lysteda) competitively blocks the conversion of plasminogen to plasmin, thereby reducing fibrinolysis.Clinical trials of tranexamic acid to treat menorrhagia were first performed in the 1960s. The agent proved to be superior to placebo, NSAIDs, and cyclic oral progestin.4 However, the LNG-IUS has been found to be more effective than tranexamic acid in reducing menstrual blood loss.5
In the United States, until very recently, tranexamic acid was FDA-indicated only as an injectable agent for use at the time of dental extraction in hemophilic patients. However, an oral formulation has been widely used abroad, including in Canada, to treat heavy menstrual bleeding. In Sweden, the agent is available over the counter.
In November 2009, the FDA approved 650-mg tablets of tranexamic acid as a treatment for heavy menstrual bleeding.6
Recommended dosage
In women who have normal renal function, the recommended dosage of tranexamic acid is 1,300 mg (two 650-mg tablets) three times daily (a total of 3,900 mg) for as long as 5 days during menstruation. In women who have impaired renal function, the dosage should be adjusted according to the package insert.
In randomized clinical trials, tranexamic acid did not produce more side effects than placebo did. However, because of concerns that tranexamic acid may increase risk of venous thromboembolism (VTE), the agent is contraindicated in women who have a history of VTE.
Although women who use hormonal contraception have not been included in clinical trials of tranexamic acid, the FDA recommends that they take tranexamic acid only if there is “a strong medical need and the benefit of treatment will outweigh the potential increased risk of a thrombotic event.”6
Tranexamic acid will likely be of particular utility for women who desire treatment for heavy menstrual bleeding but who should, or would prefer to, avoid hormonal agents. British guidelines recommend that, when medical management is appropriate in the treatment of heavy menstrual bleeding, the LNG-IUS be considered first-line treatment, with tranexamic acid representing a second-line strategy.7
CASE: Resolved
When she hears about tranexamic acid, R.L. is eager to try it. She is pleased that it does not contain hormones and needs to be taken only 5 days or fewer each month. You give her a prescription and schedule a follow-up visit 3 months later, at which time she reports being satisfied with the drug.
CASE: Heavy bleeding and apprehension over hormones
Your patient, R.L., 34 years old, has been bothered by gradually worsening menorrhagia for 2 years when she schedules an appointment to discuss treatment options. She prefers to avoid surgery, if at all possible, and is reluctant to use hormonal medications because she’s concerned about side effects. Nonsteroidal anti-inflammatory drugs provide inadequate relief.
R.L. asks if there is any other agent that can reduce her menstrual blood loss.
What can you offer to her?
Heavy menstrual bleeding (menorrhagia) impairs quality of life and prompts many women such as R.L. to seek a gynecologic consultation—many of them hoping to avoid surgery.
In the United States, medical-management options for the treatment of heavy menstrual bleeding include nonsteroidal anti-inflammatory drugs (NSAIDs) and off-label use of oral contraceptives, injectable depot medroxyprogesterone acetate, and high-dose oral progestin (e.g., norethindrone acetate, 5-mg tablets). The levonorgestrel intrauterine system (LNG-IUS; Mirena) is also widely used to treat heavy menstrual bleeding in women and was approved for this indication in October 2009. FDA approval of the LNG-IUS for heavy menstrual bleeding was based on a pivotal randomized trial demonstrating high efficacy.1,2
Very soon an additional option will be available—the antifibrinolytic agent tranexamic acid (to be sold by Ferring Pharmaceuticals under the brand name Lysteda). Although the LNG-IUS remains first-line treatment for heavy menstrual bleeding, tranexamic acid is appropriate for women who need to, or prefer to, avoid hormones.
How tranexamic acid works
Women who experience heavy menstrual bleeding have an elevated level of plasminogen activators in the endometrium, enzymes that facilitate the dissolution of clots. These enzymes can be inhibited by antifibrinolytic agents such as tranexamic acid, which competitively blocks the conversion of plasminogen to plasmin, thereby reducing fibrinolysis (FIGURE).3

FIGURE How tranexamic acid stanches heavy menstrual bleeding
Tranexamic acid (Lysteda) competitively blocks the conversion of plasminogen to plasmin, thereby reducing fibrinolysis.Clinical trials of tranexamic acid to treat menorrhagia were first performed in the 1960s. The agent proved to be superior to placebo, NSAIDs, and cyclic oral progestin.4 However, the LNG-IUS has been found to be more effective than tranexamic acid in reducing menstrual blood loss.5
In the United States, until very recently, tranexamic acid was FDA-indicated only as an injectable agent for use at the time of dental extraction in hemophilic patients. However, an oral formulation has been widely used abroad, including in Canada, to treat heavy menstrual bleeding. In Sweden, the agent is available over the counter.
In November 2009, the FDA approved 650-mg tablets of tranexamic acid as a treatment for heavy menstrual bleeding.6
Recommended dosage
In women who have normal renal function, the recommended dosage of tranexamic acid is 1,300 mg (two 650-mg tablets) three times daily (a total of 3,900 mg) for as long as 5 days during menstruation. In women who have impaired renal function, the dosage should be adjusted according to the package insert.
In randomized clinical trials, tranexamic acid did not produce more side effects than placebo did. However, because of concerns that tranexamic acid may increase risk of venous thromboembolism (VTE), the agent is contraindicated in women who have a history of VTE.
Although women who use hormonal contraception have not been included in clinical trials of tranexamic acid, the FDA recommends that they take tranexamic acid only if there is “a strong medical need and the benefit of treatment will outweigh the potential increased risk of a thrombotic event.”6
Tranexamic acid will likely be of particular utility for women who desire treatment for heavy menstrual bleeding but who should, or would prefer to, avoid hormonal agents. British guidelines recommend that, when medical management is appropriate in the treatment of heavy menstrual bleeding, the LNG-IUS be considered first-line treatment, with tranexamic acid representing a second-line strategy.7
CASE: Resolved
When she hears about tranexamic acid, R.L. is eager to try it. She is pleased that it does not contain hormones and needs to be taken only 5 days or fewer each month. You give her a prescription and schedule a follow-up visit 3 months later, at which time she reports being satisfied with the drug.
1. FDA approves additional use for IUD Mirena to treat heavy menstrual bleeding in IUD users [press release]. Rockville, Md: FDA; Oct. 1, 2009.http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm184747.htm. Accessed May 3, 2010.
2. Kaunitz AM, Bissonette F, Monteiro I, Lukkari-Lax E, Muysers C, Jensen J. Levonorgestrel intrauterine system and medroxyprogesterone acetate for treatment of heavy menstrual bleeding. Poster No. 49 Presented at Reproductive Health 2009, Association of Reproductive Health Professionals annual meeting, September 30–October 3, 2009, Los Angeles, Calif. Contraception. 2009;80(2):212.-
3. Grimes DA, Davis AR, Ramos DE. Heavy menstrual bleeding: Assessing impact, evaluating management options. OBG Management. 2009;21(10)(supple).-http://www.obgmanagement.com/article_pages.asp?filename="2206OBG_Article2" aid=7971. Accessed May 3, 2010.
4. Lethaby A, Farquhar C, Cooke I. Antifibrinolytics for heavy menstrual bleeding. Cochrane Database Syst Rev. 2000;(4):CD000249.-
5. Milsom I, Andersson K, Andersch B, Rybo G. A comparison of flurbiprofen, tranexamic acid, and a levonorgestrel-releasing intrauterine contraceptive device in the treatment of idiopathic menorrhagia. Am J Obstet Gynecol. 1991;164(3):879-883.
6. FDA approves Lysteda to treat heavy menstrual bleeding [press release]. Rockville, Md: FDA; Nov. 13, 2009.http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm190551.htm. Accessed May 3, 2010.
7. National Institute for Health and Clinical Excellence. Heavy menstrual bleeding. January 2007.http://www.nice.org.uk/CG44. Accessed May 3, 2010.
1. FDA approves additional use for IUD Mirena to treat heavy menstrual bleeding in IUD users [press release]. Rockville, Md: FDA; Oct. 1, 2009.http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm184747.htm. Accessed May 3, 2010.
2. Kaunitz AM, Bissonette F, Monteiro I, Lukkari-Lax E, Muysers C, Jensen J. Levonorgestrel intrauterine system and medroxyprogesterone acetate for treatment of heavy menstrual bleeding. Poster No. 49 Presented at Reproductive Health 2009, Association of Reproductive Health Professionals annual meeting, September 30–October 3, 2009, Los Angeles, Calif. Contraception. 2009;80(2):212.-
3. Grimes DA, Davis AR, Ramos DE. Heavy menstrual bleeding: Assessing impact, evaluating management options. OBG Management. 2009;21(10)(supple).-http://www.obgmanagement.com/article_pages.asp?filename="2206OBG_Article2" aid=7971. Accessed May 3, 2010.
4. Lethaby A, Farquhar C, Cooke I. Antifibrinolytics for heavy menstrual bleeding. Cochrane Database Syst Rev. 2000;(4):CD000249.-
5. Milsom I, Andersson K, Andersch B, Rybo G. A comparison of flurbiprofen, tranexamic acid, and a levonorgestrel-releasing intrauterine contraceptive device in the treatment of idiopathic menorrhagia. Am J Obstet Gynecol. 1991;164(3):879-883.
6. FDA approves Lysteda to treat heavy menstrual bleeding [press release]. Rockville, Md: FDA; Nov. 13, 2009.http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm190551.htm. Accessed May 3, 2010.
7. National Institute for Health and Clinical Excellence. Heavy menstrual bleeding. January 2007.http://www.nice.org.uk/CG44. Accessed May 3, 2010.
IN THIS ARTICLE
6 skin disorders of pregnancy: A management guide
The dermatoses of pregnancy are a poorly understood group of conditions. Their only common feature is a tendency to appear during pregnancy.
Only three of these conditions are considered unique to pregnancy, however; the others are probably exacerbations of preexisting conditions triggered by pregnancy. There isn’t even complete agreement on what to call them. To make management even more complex, two patients—mother and fetus—need to be considered in decisions about care.
Who manages these patients is another matter. These conditions fall into overlapping areas of health care, where family physicians, obstetricians, and dermatologists all might have some share in responsibility for diagnosis and treatment. You need to be sufficiently familiar with these conditions so that you can differentiate those that can be treated symptomatically and those that require referral to a specialist. This review and the handy TABLE, will help you toward that end.
TABLE
Skin disorders of pregnancy: What you’ll see, how to treat
| Disorder | Lesions | Diagnosis and sequelae | Treatment | Recurrence |
|---|---|---|---|---|
| Pemphigoid gestationis3,5 | Erythematous papules that progress to vesicles and bullae, in a periumbilical distribution that spares the face, palms, and soles |
|
| Frequent; skips a pregnancy 8% of the time |
| Pruritic urticarial papules and plaques of pregnancy8-10 | Urticarial papules and plaques on the abdomen, legs, arms, buttocks, chest, and back |
| Topical corticosteroids and antihistamines | Uncommon |
| Intrahepatic cholestasis of pregnancy14,17,19-22 | No primary lesions; secondary excoriations in any area that the patient can reach |
| Ursodeoxycholic acid, 450-1,200 mg/d | Frequent |
| Eczema of pregnancy/pruritus of pregnancy4,10,24 | Grouped, crusted erythematous papules, patches, and plaques, most often on extensor surfaces of the arms and legs or on the abdomen |
| Symptomatic treatment with topical corticosteroids or antihistamines | Frequent |
| Acute pustular psoriasis of pregnancy26-28 | Erythematous plaques and pustules that start on the inner thighs and groin and spread to the trunk and extremities |
|
| Unknown |
| Pruritic folliculitis of pregnancy24,28 | Papules and pustules concentrated around hair follicles, often beginning on the abdomen and spreading to the extremities |
| Topical corticosteroids | Unknown |
DERMATOSES UNIQUE TO PREGNANCY
1. Pemphigoid gestationis
Years ago, this disorder was referred to as herpes gestationis, because the lesions are herpetiform. Pemphigoid gestationis (PG) has an incidence of approximately 1 in 10,000 pregnancies.1,2 Time of onset is usually about the 21st week of gestation, although, in about 20% of cases, the eruption appears immediately postpartum.3
Presentation. The disease usually begins with urticarial papules and plaques around the umbilicus and extremities. Bullous lesions tend to develop as the disease progresses, and are often not present on first presentation (FIGURE 1). Lesions of PG tend to spare the face, palms, and soles. Mucosal surfaces are involved in fewer than 20% of cases. In about 75% of cases, PG flares around the time of delivery, regressing spontaneously after the baby is born.4
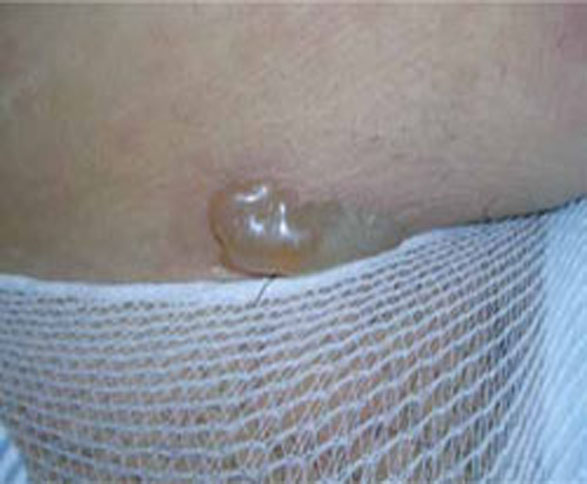
FIGURE 1 Pemphigoid gestationis
As the disease progresses, bullous lesions tend to develop.
Pathophysiology. The pathophysiology of PG is nearly identical to that of bullous pemphigoid, a blistering skin disorder seen more often in elderly patients.5 Pemphigoid disorders are immune processes, involving an immunoglobulin G (IgG) immune response directed at a 180-kDa hemidesmosome transmembrane glycoprotein. This protein is the common target in several subepidermal blistering diseases.
Differential diagnosis. Disorders that may have some of the same features as PG include pruritic urticarial papules and plaques of pregnancy (PUPPP), erythema multiforme, intrahepatic cholestasis of pregnancy (ICP), contact dermatitis, and drug reactions.
Diagnosis. A biopsy is necessary for definitive diagnosis. Direct immunofluorescence (DIF) microscopy of a sample of perilesional skin can show tissue-bound immunoreactants. Linear deposition of the complement component protein C3 along the basement membrane zone is diagnostic for PG. IgG is also deposited about 40% of the time.3
Serum enzyme-linked immunosorbent assay (ELISA) studies are also helpful in diagnosis. They have excellent sensitivity and specificity, as well as the capacity to monitor levels of antibody, which correlate with the severity of disease.1
Treatment. Oral corticosteroids are the first-line treatment for PG, typically 20 to 60 mg/d of prednisone. Oral corticosteroids are generally most effective at ameliorating symptoms. Prednisone at a dosage of 40 to 80 mg/d for a short time has not been associated with congenital abnormalities.6 PG patients can also be treated successfully with intravenous immunoglobulin (IVIG) and cyclosporine in refractory cases.7
Pruritus associated with this condition can interfere with day-to-day activities and with the patient’s ability to sleep. Patients may also complain that the rash is painful, particularly if bullae rupture, leading to superficial ulcerations. Fortunately, the patient’s quality of life can be dramatically improved with systemic corticosteroids—with no significant risk to the fetus.
Sequelae. PG uniformly resolves within a few weeks, but the mother’s autoantibodies can be passively transferred to the fetus, causing vesicles and bullae in the newborn.8 An increased incidence of small-for-gestational age (SGA) infants has also been noted in PG, although no lasting morbidity or mortality in the offspring has been noted.5 The disease tends to recur in future pregnancies.
2. Pruritic urticarial papules and plaques of pregnancy
This condition is known by many names besides its acronym PUPPP: polymorphic eruption of pregnancy, toxemic erythema of pregnancy, and late prurigo of pregnancy.1 It is a pruritic, inflammatory skin disorder that has been variously estimated to occur in anywhere from 1 in 120 to 1 in 240 pregnancies.8 PUPPP is second only to eczema as the most common dermatosis of pregnancy.
Presentation. As the name suggests, the lesions of PUPPP are itchy, red papules that often coalesce into plaques (FIGURE 2). Lesions usually occur in primigravidas after the 34th week of gestation, although they may be seen at any time from the first trimester through the postpartum period.9
Lesions are classically found on the abdomen, sparing the umbilical area, and are found primarily in the striae. This distribution helps you to differentiate PUPPP from PG, in which lesions typically cluster around the umbilicus. Most PUPPP lesions (80% in one study) are dispersed on the abdomen, legs, arms, buttocks, chest, and back. Another 17% appear only on the abdomen and proximal thighs, and the remaining 3% on the limbs.10 Nearly 50% of the time, lesions also include discrete vesicles.11 There are no reported cases of mucosal involvement.
Patients with this condition are often very uncomfortable. The associated pruritus is severe enough to interfere with sleep. Despite the itching, however, lesions are seldom excoriated.

FIGURE 2 Pruritic urticarial papules and plaques of pregnancy
The itchy, red papules of PUPP often coalesce into plaques.
Pathophysiology. The disorder has been strongly associated with maternal weight gain and multiple gestations. One working hypothesis is that rapid abdominal distention observed in the third trimester leads to damage of the connective tissue, which then releases antigenic molecules, causing an inflammatory reaction.12 Another hypothesis is that increased levels of fetal DNA that have been detected in the skin of PUPPP patients may contribute to the pathology. One study detected male DNA in six of 10 PUPPP sufferers, but found none in any of 26 controls—pregnant women without PUPPP pathology.5 There is some evidence that patients with atopy may be predisposed to PUPPP, as well as patients who are hypertensive or obese.10,13
Differential diagnosis. Initially, PUPPP lesions can be difficult to differentiate from urticarial PG lesions. The distribution of the lesions is the best clue: PG lesions cluster around the umbilicus, whereas PUPPP lesions uniformly spare the umbilical area. Additional disorders in the PUPPP differential are atopic dermatitis, superficial urticarial allergic eruption, viral exanthema, and contact or irritant dermatitis.
Diagnosis. PUPPP can be diagnosed only by clinical observation. None of the available laboratory tests—immunofluorescence, histology, serology—yield findings specific for PUPPP, although histology and immunofluorescence can readily differentiate between this condition and PG.
Treatment. Because the disease holds no real danger for mother or fetus, treatment can be aimed solely at symptomatic relief. Mild-to-potent topical corticosteroids (consider triamcinolone or fluocinonide) should relieve pruritus within 48 to 72 hours.8 Antihistamines and, occasionally, low-dose systemic corticosteroids may also be used. Consider hydroxyzine, although diphenhydramine has the more proven safety profile in pregnancy.
Nonpharmaceutical treatments such as oil baths and emollients should also be considered. If the condition appears classic for PUPPP, it can be managed symptomatically. If there is any question about the diagnosis, however, referral to a dermatologist is prudent.
Sequelae. No increase in maternal or fetal morbidity or mortality is associated with PUPPP. Recurrence is fairly uncommon, as the disease primarily affects women during their first pregnancy.
3. Intrahepatic cholestasis of pregnancy
This condition is also called recurrent or idiopathic jaundice of pregnancy, obstetric cholestasis, and pruritus gravidarum. Intrahepatic cholestasis of pregnancy (ICP) is caused by disruption of hepatic bile flow during pregnancy. It has been recorded at a rate of approximately 10 to 150 of every 10,000 pregnancies in Europe and 70 of every 10,000 in the United States.12 In 80% of patients, time of onset is after the 30th week.14
Although this disorder is not primarily a dermatosis of pregnancy, it is a pruritic condition that often presents with excoriations in pregnant women and is associated with fetal morbidity and mortality. It’s important to be able to identify this disease early to minimize sequelae.
Presentation. There are no primary lesions with ICP. The primary presenting symptom is a generalized pruritus affecting the palms and soles, and sometimes extending to the legs and abdomen (FIGURE 3). This itching is often so severe that it leads to chronic insomnia. You may see secondary skin lesions, such as erythema and excoriations. Observable jaundice occurs in 10% to 20% of patients.3 These patients do not develop the encephalopathy that is associated with cholestasis in the nonpregnant state, however.14
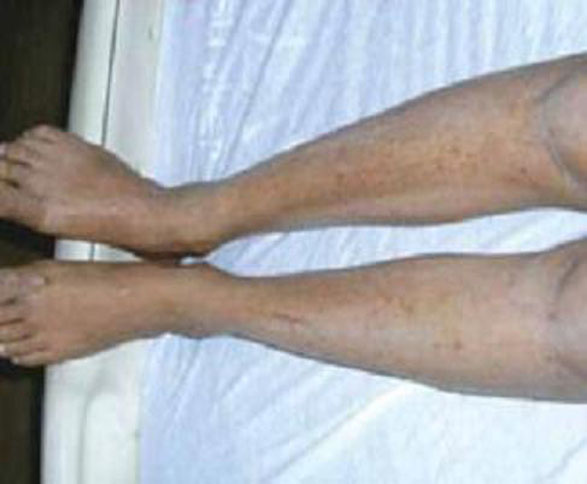
FIGURE 3 Intrahepatic cholestasis of pregnancy
ICP lacks primary lesions. Shown here are the secondary erythema and excoriations that results from scratching the intense pruritis.
Pathophysiology. The genesis of this condition is thought to be a combination of genetic and environmental factors. A family history of the disorder is present in one half of cases; cases with a familial component tend to be more severe.15 ICP may be an exaggerated response to increased estrogen levels in pregnancy, but the mechanism of this response is unknown.16
Differential diagnosis. Other conditions that must be considered in making the diagnosis are viral hepatitis, gallbladder disease, PG, PUPPP, drug hepatotoxicity, primary biliary cirrhosis, and uremia.
Diagnosis. Laboratory values are the definitive diagnostic tool in this condition. Increased levels of serum bile acids are the single most sensitive test. Average levels of serum bile acids in pregnancy are 6.6 µmol/L, with an upper limit of 11 µmol/L. The average value in women who have ICP is 47 µmol/L.17
Although serum bile acids remain the gold standard, a recent study showed that elevated urine bile acids have 100% sensitivity and 83% specificity for ICP.18 In 55% to 60% of cases, the liver enzymes aspartate aminotransferase (AST) and alanine aminotransferase (ALT) are mildly increased. Steatorrhea is often noted by the patient, and is followed by vitamin K deficiency.17
Treatment. The current standard of care for ICP is treatment with ursodeoxycholic acid (UDCA). In four controlled trials, UDCA caused a sustained decrease in serum bile acids.19-22 The dosage used in these trials ranged from 450 to 1,200 mg/d.
Before UDCA treatment was available, ICP was treated with cholestyramine, which could bring about a 70% rate of response. The drawback to cholestyramine treatment is that it precipitates vitamin K, which is already compromised by the disease process. Further, the onset of action of cholestyramine is slow.3
Elective delivery is indicated for ICP, particularly in patients who have a significant clinical presentation.12 Delivery for ICP should be performed around weeks 37 to 38, as stillbirths tend to cluster around weeks 37 to 39.14 Given the significant fetal mortality associated with ICP (see the next paragraph), the condition should be managed by a clinician experienced with the disease—likely, a gastroenterologist.
Sequelae. The impact of this maternal disorder on the fetus can be disastrous: a 10% to 15% rate of perinatal death, and a 30% to 40% rate of premature labor.14 Fortunately, the rate of preterm labor correlates strongly with the level of bile acids, so that as bile acid levels are reduced with UDCA treatment, the rate of preterm labor also falls. Management of ICP has reduced the rate of perinatal death to 3.5%. No evidence of fetal growth retardation has been noted.14
DERMATOSES TRIGGERED BY PREGNANCY
4. Eczema of pregnancy/prurigo of pregnancy
Eczema of pregnancy/prurigo of pregnancy (EP/PP) may not actually be correlated with the pregnant state. Both conditions manifest as eczematous lesions in an atopic distribution. Although they have been described in the literature as separate entities, the lack of clinical distinction between them led Ambros-Rudolph and colleagues to combine them under the umbrella term, atopic eruption of pregnancy.23
In some patients, at least, EP/PP may be preexisting conditions that are exacerbated by pregnancy. One study of 255 patients with the condition found that 20% had had the lesions before they became pregnant.23 The tendency of the condition to be made markedly worse by pregnancy, however, leads us to include it here.
PP has an estimated incidence of 1 in 450 pregnancies.11 Although many authorities consider EP to be the most common dermatosis of pregnancy, no clear estimation of its prevalence has been established.23,24 Taken together, the two conditions have the highest prevalence of all pregnancy-induced dermatoses.
PP is also known as popular dermatitis of Spangler, Nurse’s early prurigo of pregnancy, and linear IgM disease of pregnancy.3,4,23
Presentation. The typical presentation is grouped, crusted, erythematous papules, patches, and plaques—frequently with excoriations. Lesions typically present on the extensor surfaces of the arms and legs or on the abdomen (FIGURE 4).4 Recurrence in later pregnancies is common.
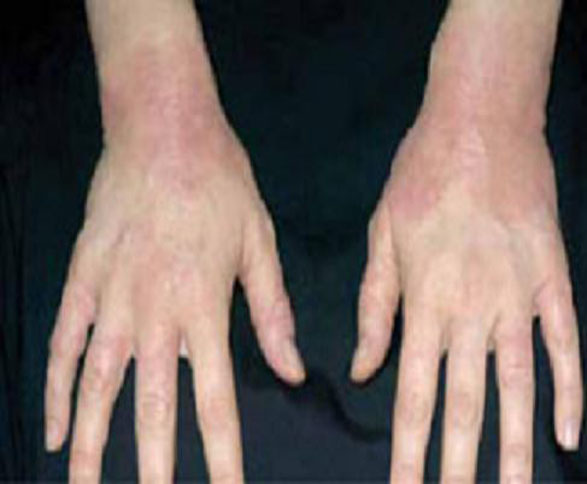
FIGURE 4 Eczema of pregnancy
EP/PP typically manifests as grouped, crusted, erythematous papules, patches, and plaques, often with excoriations.
Pathophysiology. The pathophysiology of EP/PP is not understood. Many patients who develop EP/PP have a history of atopy.10
Differential diagnosis. Conditions that need to be considered in making the diagnosis include Tinea infection, scabies, contact dermatitis, ICP, pruritic folliculitis of pregnancy (PFP), and PG.
Diagnosis. The history and the physical examination determine the diagnosis. Serology, histopathology, and immunofluorescence are nonspecific. Correlation of EP/PP with increased IgE is marginal, at best.24,25
Treatment. These conditions are treated symptomatically with topical corticosteroids or systemic antihistamines.
Sequelae. No increase in maternal or fetal morbidity or mortality is associated with EP/PP.
5. Acute pustular psoriasis of pregnancy
Whether or not acute pustular psoriasis of pregnancy (APPP) is actually a pregnancy-induced dermatosis is subject to debate.
There is evidence that APPP is not unique to pregnancy but simply a manifestation of ordinary psoriasis. Clinically and histologically, APPP is indistinguishable from pustular psoriasis. Unlike most cases of acute psoriasis, however, APPP often appears in pregnancy without any personal or family history of psoriasis and usually ceases when the pregnancy ends. This fact, combined with reports of increased fetal and maternal morbidity and mortality associated with APPP, leads us to include it here.26
Presentation. APPP is a rare condition that may have an onset at any point in pregnancy. Characteristic lesions begin as erythematous plaques with pustules on the inner thighs, flexural areas, and groin and spread to the trunk and extremities. As plaques enlarge, their center becomes eroded and crusted.
The nails may become onycholytic. Hands, feet, and face are usually spared. Oral and esophageal erosions can occur. Pruritus is typically mild, although the lesions are often painful. Flu-like symptoms are often present.27
Pathophysiology. The pathophysiology of APPP is unknown.
Differential diagnosis. Conditions with similar presentations include an adverse drug reaction, pityriasis rosea, lichen simplex chronicus, eczema, lupus, and pityriasis rubra pilaris.
Diagnosis. The clinical history and an association with systemic illness are the basis for a diagnosis of APPP. Cultures of pustules are negative for any infective pathology, although, as the disease progresses, pustules may become superinfected. Laboratory testing may show an increased erythrocyte sedimentation rate, hypocalcemia, and a low level of vitamin D.
Treatment. Prednisone, 15 to 60 mg/d, is often sufficient to control the disease.27 Cyclosporine, 100 mg twice daily, has also been shown to be useful.28 Cyclosporine in pregnancy is a Category C drug. Data on fetal malformation associated with cyclosporine therapy are limited, but risk appears minimal.6
Maternal hypocalcemia should be monitored and treated appropriately. If disease progression is judged serious enough, early induction of labor is indicated, because delivery will almost always lead to swift resolution.
Sequelae. A number of case reports link APPP to serious sequelae, including fetal growth retardation, hypocalcemia, and stillbirth.27,29,30 The condition is too rare, however, for good data on specific sequelae. Although APPP does give significant cause for concern, it appears that some of the traditional apprehension comes from older publications reporting a rate of maternal mortality of 70% to 90%.31 This statistic has not been borne out in practice. It does appear that the mother will frequently suffer systemic symptoms, including fever and malaise.
6. Pruritic folliculitis of pregnancy
Accounts of the prevalence of pruritic folliculitis of pregnancy (PFP) vary widely. Some sources report fewer than 30 cases in all of the literature; others indicate that the prevalence is equivalent to that of PG—one in every 10,000 pregnancies.3,11 PFP most often presents in the third trimester. It often resolves before delivery, but uniformly clears within 2 weeks of delivery.
Presentation. PFP presents as papules and pustules concentrated around hair follicles (FIGURE 5). Often, lesions begin on the abdomen and spread to the extremities.24,28 The condition is often, but not always, pruritic. Patients are more likely to be concerned about what the condition means for their health than distressed by the symptoms.
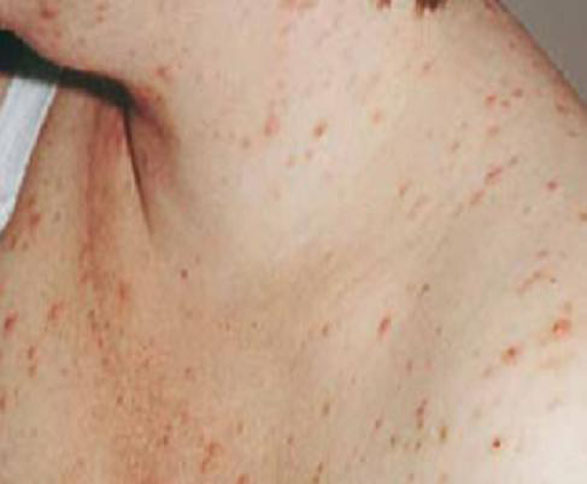
FIGURE 5 Pruritic folliculitis of pregnancy
The papules and pustules of PFP are concentrated around hair follicles.
Pathophysiology. Like many other dermatoses of pregnancy, the pathophysiology of PFP is unknown. There is little evidence that the condition is immunologically or hormonally mediated, and there is no evidence of an infectious component.24,28
Differential diagnosis. PFP must be distinguished from infectious folliculitis, acneiform disorders, HIV-associated eosinophilic folliculitis, and a drug reaction.
Diagnosis. The clinical diagnosis is based on presenting symptoms and third-trimester onset. No specific laboratory or histologic analysis can be used to make a definitive diagnosis.
Treatment. As the condition is, by definition, a nonmicrobial folliculitis, the most effective therapy tends to be with a low- or midpotency topical corticosteroid, such as triamcinolone or desonide. A benzoyl peroxide wash can also be effective.
Sequelae. One study reports an increased incidence of low birth weight, but no associated morbidity or mortality has been reported in recent studies.24
- Pemphigoid gestationis is best managed with oral prednisone at doses from 20 to 60 mg per day to control symptoms
- The pruritus associated with pruritic urticarial papules and plaques of pregnancy can be safely and effectively managed with topical corticosteroids and oral antihistamines
- Treat intrahepatic cholestasis of pregnancy with ursodeoxycholic acid, which likely reduces serum bile acids as well as associated fetal morbidity and mortality
1. Sitaru C, Powell J, Messer G, Bröcker EB, Wojnarowska F, Zillikens D. Immunoblotting and enzyme linked immunosorbent assay for the diagnosis of pemphigoid gestationis. Obstet Gynecol. 2004;103(4):757-763.
2. Engineer L, Bohl K, Ahmed AR. Pemphigoid gestationis: a review. Am J Obstet Gynecol. 2000;183(2):483-491.
3. Kroumpouzos G, Cohen LM. Specific dermatoses of pregnancy: an evidence-based systematic review. Am J Obstet Gynecol. 2003;188(4):1083-1092.
4. Shornick JK. Dermatoses of pregnancy. Semin Cutan Med Surg. 1998;17(3):172-181.
5. Shornick JK, Bangert JL, Freeman RG, Gilliam JN. Herpes gestationis: clinical and histological features in 28 cases. J Am Acad Dermatol. 1983;8(2):214-224.
6. Leachman SA, Reed BR. The use of dermatologic drugs in pregnancy and lactation. Dermatol Clin. 2006;24(2):167-197, vi.
7. Hern S, Harman K, Bhogal BS, Black MM. A severe persistent case of pemphigoid gestationis treated with intravenous immunoglobulins and cyclosporine. Clin Exp Dermatol. 1998;23(4):185-188.
8. Fitzpatrick TP. Diseases in pregnancy. Color Atlas and Synopsis of Clinical Dermatology. New York, NY: McGraw Hill; 1997:414–419.
9. Aractingi D, Berkane N, Bertheau P, et al. Fetal DNA in skin of polymorphic eruptions of pregnancy. Lancet. 1998;352(9144):1898-1901.
10. Rudolph CM, Al-Fares S, Vaughan-Jones SA, et al. Polymorphic eruption of pregnancy: clinicopathology and potential trigger factors in 181 patients. Br J Dermatol. 2006;154(1):54-60.
11. Roger D, Vaillant L, Fignon A, et al. Specific pruritic diseases of pregnancy. A prospective study of 3192 pregnant women. Arch Dermatol. 1994;130(6):734-739.
12. McDonald JA. Cholestasis of pregnancy. J Gastroenterol Hepatol. 1999;14(6):515-518.
13. Ohel I, Levy A, Silberstein T, Holcberg G, Sheiner E. Pregnancy outcome of patients with pruritic urticarial papules and plaques of pregnancy. J Maternal Fetal Neonat Med. 2006;19(5):305-308.
14. Geenes V, Williamson C. Intrahepatic cholestasis of pregnancy. World J Gastroenterol. 2009;15(17):2049-2066.
15. Shaw D, Frohlich J, Wittmann BA, Willms M. A prospective study of 18 patients with cholestasis of pregnancy. Am J Obstet Gynecol. 1982;142(6 Pt 1):621-625.
16. Reyes H. The spectrum of liver and gastrointestinal disease seen in cholestasis of pregnancy. Gastroenterol Clin North Am. 1992;21(4):905-921.
17. Lammert F, Marschall H, Glantz A, Matern S. Intrahepatic cholestasis of pregnancy; molecular pathogenesis, diagnosis and management. J Hepatol. 2000;33(6):1012-1021.
18. Huang WM, Seubert DE, Donnelly JG, Liu M, Javitt NB. Intrahepatic cholestasis of pregnancy: detection with urinary bile acid assays. J Perinat Med. 2007;35(6):486-491.
19. Palma J, Reyes H, Ribalta J, et al. Ursodeoxycholic acid in the treatment of intrahepatic cholestasis of pregnancy: a randomized, double-blind study controlled with placebo. J Hepatol. 1997;27(6):1022-1028.
20. Diaferia A, Nicastri PL, Tartagni M, et al. Ursodeoxycholic acid therapy in pregnant women with cholestasis. Int J Gynaecol Obstet. 1996;52:133-140.
21. Nicastri PL, Diaferia A, Tartagni M, Loizzi P, Fanelli M. A randomised placebo-controlled trial of ursodeoxycholic acid and S-adenosylmethionine in the treatment of intrahepatic cholestasis of pregnancy. Br J Obstet Gynaecol. 1998;105(11):1205-1207.
22. Glantz A, Marschall HU, Lammert F, Mattsson LA. Intrahepatic cholestasis in pregnancy: a randomized controlled trial comparing dexamethasone and ursodeoxycholic acid. Hepatology. 2005;42(6):1399-1405.
23. Ambros-Rudolph C, Müllegger M, Vaughan-Jones S, et al. The specific dermatoses of pregnancy revisited and reclassified: results of a retrospective two-center study on 505 pregnant patients. J Am Acad Dermatol. 2006;54(3):395-404.
24. Vaughan Jones SA, Hern S, Nelson-Piercy C, Seed PT, Black MM. A prospective study of 200 women with dermatoses of pregnancy correlating clinical findings with hormonal and immunopathological profiles. Br J Dermatol. 1999;141(1):71-81.
25. Holmes RC, Black MM. The specific dermatoses of pregnancy. J Am Acad Dermatol. 1983;8(3):405-412.
26. Bukhari IA. Impetigo herpetiformis in a primagravida: successful treatment with etretinate. J Drugs Dermatol. 2004;3(4):449-451.
27. Oumeish OY, Parish JL. Impetigo herpetiformis. Clin Dermatol. 2006;24(2):101-104.
28. Kroumpouzos G, Cohen LM. Pruritic folliculitis of pregnancy. J Am Acad Dermatol. 2000;43(1 Pt 1):132-134.
29. Sahin HG, Sahin HA, Metin A, Zeteroglu S, Ugras S. Recurrent impetigo herpetiformis in a pregnant adolescent: case report. Eur J Obstet Gynecol Reprod Biol. 2002;101:201-203.
30. Aka N, Kuscu NK, Yazicioglu E. Impetigo herpetiformis at the 36th week of gestation. Int J Gynecol Obstet. 2000;69(2):153-154.
31. Wade T, Wade S, Jones H. Skin changes and diseases associated with pregnancy. Obstet Gynecol. 1978;52(2):233-242.
The dermatoses of pregnancy are a poorly understood group of conditions. Their only common feature is a tendency to appear during pregnancy.
Only three of these conditions are considered unique to pregnancy, however; the others are probably exacerbations of preexisting conditions triggered by pregnancy. There isn’t even complete agreement on what to call them. To make management even more complex, two patients—mother and fetus—need to be considered in decisions about care.
Who manages these patients is another matter. These conditions fall into overlapping areas of health care, where family physicians, obstetricians, and dermatologists all might have some share in responsibility for diagnosis and treatment. You need to be sufficiently familiar with these conditions so that you can differentiate those that can be treated symptomatically and those that require referral to a specialist. This review and the handy TABLE, will help you toward that end.
TABLE
Skin disorders of pregnancy: What you’ll see, how to treat
| Disorder | Lesions | Diagnosis and sequelae | Treatment | Recurrence |
|---|---|---|---|---|
| Pemphigoid gestationis3,5 | Erythematous papules that progress to vesicles and bullae, in a periumbilical distribution that spares the face, palms, and soles |
|
| Frequent; skips a pregnancy 8% of the time |
| Pruritic urticarial papules and plaques of pregnancy8-10 | Urticarial papules and plaques on the abdomen, legs, arms, buttocks, chest, and back |
| Topical corticosteroids and antihistamines | Uncommon |
| Intrahepatic cholestasis of pregnancy14,17,19-22 | No primary lesions; secondary excoriations in any area that the patient can reach |
| Ursodeoxycholic acid, 450-1,200 mg/d | Frequent |
| Eczema of pregnancy/pruritus of pregnancy4,10,24 | Grouped, crusted erythematous papules, patches, and plaques, most often on extensor surfaces of the arms and legs or on the abdomen |
| Symptomatic treatment with topical corticosteroids or antihistamines | Frequent |
| Acute pustular psoriasis of pregnancy26-28 | Erythematous plaques and pustules that start on the inner thighs and groin and spread to the trunk and extremities |
|
| Unknown |
| Pruritic folliculitis of pregnancy24,28 | Papules and pustules concentrated around hair follicles, often beginning on the abdomen and spreading to the extremities |
| Topical corticosteroids | Unknown |
DERMATOSES UNIQUE TO PREGNANCY
1. Pemphigoid gestationis
Years ago, this disorder was referred to as herpes gestationis, because the lesions are herpetiform. Pemphigoid gestationis (PG) has an incidence of approximately 1 in 10,000 pregnancies.1,2 Time of onset is usually about the 21st week of gestation, although, in about 20% of cases, the eruption appears immediately postpartum.3
Presentation. The disease usually begins with urticarial papules and plaques around the umbilicus and extremities. Bullous lesions tend to develop as the disease progresses, and are often not present on first presentation (FIGURE 1). Lesions of PG tend to spare the face, palms, and soles. Mucosal surfaces are involved in fewer than 20% of cases. In about 75% of cases, PG flares around the time of delivery, regressing spontaneously after the baby is born.4

FIGURE 1 Pemphigoid gestationis
As the disease progresses, bullous lesions tend to develop.
Pathophysiology. The pathophysiology of PG is nearly identical to that of bullous pemphigoid, a blistering skin disorder seen more often in elderly patients.5 Pemphigoid disorders are immune processes, involving an immunoglobulin G (IgG) immune response directed at a 180-kDa hemidesmosome transmembrane glycoprotein. This protein is the common target in several subepidermal blistering diseases.
Differential diagnosis. Disorders that may have some of the same features as PG include pruritic urticarial papules and plaques of pregnancy (PUPPP), erythema multiforme, intrahepatic cholestasis of pregnancy (ICP), contact dermatitis, and drug reactions.
Diagnosis. A biopsy is necessary for definitive diagnosis. Direct immunofluorescence (DIF) microscopy of a sample of perilesional skin can show tissue-bound immunoreactants. Linear deposition of the complement component protein C3 along the basement membrane zone is diagnostic for PG. IgG is also deposited about 40% of the time.3
Serum enzyme-linked immunosorbent assay (ELISA) studies are also helpful in diagnosis. They have excellent sensitivity and specificity, as well as the capacity to monitor levels of antibody, which correlate with the severity of disease.1
Treatment. Oral corticosteroids are the first-line treatment for PG, typically 20 to 60 mg/d of prednisone. Oral corticosteroids are generally most effective at ameliorating symptoms. Prednisone at a dosage of 40 to 80 mg/d for a short time has not been associated with congenital abnormalities.6 PG patients can also be treated successfully with intravenous immunoglobulin (IVIG) and cyclosporine in refractory cases.7
Pruritus associated with this condition can interfere with day-to-day activities and with the patient’s ability to sleep. Patients may also complain that the rash is painful, particularly if bullae rupture, leading to superficial ulcerations. Fortunately, the patient’s quality of life can be dramatically improved with systemic corticosteroids—with no significant risk to the fetus.
Sequelae. PG uniformly resolves within a few weeks, but the mother’s autoantibodies can be passively transferred to the fetus, causing vesicles and bullae in the newborn.8 An increased incidence of small-for-gestational age (SGA) infants has also been noted in PG, although no lasting morbidity or mortality in the offspring has been noted.5 The disease tends to recur in future pregnancies.
2. Pruritic urticarial papules and plaques of pregnancy
This condition is known by many names besides its acronym PUPPP: polymorphic eruption of pregnancy, toxemic erythema of pregnancy, and late prurigo of pregnancy.1 It is a pruritic, inflammatory skin disorder that has been variously estimated to occur in anywhere from 1 in 120 to 1 in 240 pregnancies.8 PUPPP is second only to eczema as the most common dermatosis of pregnancy.
Presentation. As the name suggests, the lesions of PUPPP are itchy, red papules that often coalesce into plaques (FIGURE 2). Lesions usually occur in primigravidas after the 34th week of gestation, although they may be seen at any time from the first trimester through the postpartum period.9
Lesions are classically found on the abdomen, sparing the umbilical area, and are found primarily in the striae. This distribution helps you to differentiate PUPPP from PG, in which lesions typically cluster around the umbilicus. Most PUPPP lesions (80% in one study) are dispersed on the abdomen, legs, arms, buttocks, chest, and back. Another 17% appear only on the abdomen and proximal thighs, and the remaining 3% on the limbs.10 Nearly 50% of the time, lesions also include discrete vesicles.11 There are no reported cases of mucosal involvement.
Patients with this condition are often very uncomfortable. The associated pruritus is severe enough to interfere with sleep. Despite the itching, however, lesions are seldom excoriated.

FIGURE 2 Pruritic urticarial papules and plaques of pregnancy
The itchy, red papules of PUPP often coalesce into plaques.
Pathophysiology. The disorder has been strongly associated with maternal weight gain and multiple gestations. One working hypothesis is that rapid abdominal distention observed in the third trimester leads to damage of the connective tissue, which then releases antigenic molecules, causing an inflammatory reaction.12 Another hypothesis is that increased levels of fetal DNA that have been detected in the skin of PUPPP patients may contribute to the pathology. One study detected male DNA in six of 10 PUPPP sufferers, but found none in any of 26 controls—pregnant women without PUPPP pathology.5 There is some evidence that patients with atopy may be predisposed to PUPPP, as well as patients who are hypertensive or obese.10,13
Differential diagnosis. Initially, PUPPP lesions can be difficult to differentiate from urticarial PG lesions. The distribution of the lesions is the best clue: PG lesions cluster around the umbilicus, whereas PUPPP lesions uniformly spare the umbilical area. Additional disorders in the PUPPP differential are atopic dermatitis, superficial urticarial allergic eruption, viral exanthema, and contact or irritant dermatitis.
Diagnosis. PUPPP can be diagnosed only by clinical observation. None of the available laboratory tests—immunofluorescence, histology, serology—yield findings specific for PUPPP, although histology and immunofluorescence can readily differentiate between this condition and PG.
Treatment. Because the disease holds no real danger for mother or fetus, treatment can be aimed solely at symptomatic relief. Mild-to-potent topical corticosteroids (consider triamcinolone or fluocinonide) should relieve pruritus within 48 to 72 hours.8 Antihistamines and, occasionally, low-dose systemic corticosteroids may also be used. Consider hydroxyzine, although diphenhydramine has the more proven safety profile in pregnancy.
Nonpharmaceutical treatments such as oil baths and emollients should also be considered. If the condition appears classic for PUPPP, it can be managed symptomatically. If there is any question about the diagnosis, however, referral to a dermatologist is prudent.
Sequelae. No increase in maternal or fetal morbidity or mortality is associated with PUPPP. Recurrence is fairly uncommon, as the disease primarily affects women during their first pregnancy.
3. Intrahepatic cholestasis of pregnancy
This condition is also called recurrent or idiopathic jaundice of pregnancy, obstetric cholestasis, and pruritus gravidarum. Intrahepatic cholestasis of pregnancy (ICP) is caused by disruption of hepatic bile flow during pregnancy. It has been recorded at a rate of approximately 10 to 150 of every 10,000 pregnancies in Europe and 70 of every 10,000 in the United States.12 In 80% of patients, time of onset is after the 30th week.14
Although this disorder is not primarily a dermatosis of pregnancy, it is a pruritic condition that often presents with excoriations in pregnant women and is associated with fetal morbidity and mortality. It’s important to be able to identify this disease early to minimize sequelae.
Presentation. There are no primary lesions with ICP. The primary presenting symptom is a generalized pruritus affecting the palms and soles, and sometimes extending to the legs and abdomen (FIGURE 3). This itching is often so severe that it leads to chronic insomnia. You may see secondary skin lesions, such as erythema and excoriations. Observable jaundice occurs in 10% to 20% of patients.3 These patients do not develop the encephalopathy that is associated with cholestasis in the nonpregnant state, however.14

FIGURE 3 Intrahepatic cholestasis of pregnancy
ICP lacks primary lesions. Shown here are the secondary erythema and excoriations that results from scratching the intense pruritis.
Pathophysiology. The genesis of this condition is thought to be a combination of genetic and environmental factors. A family history of the disorder is present in one half of cases; cases with a familial component tend to be more severe.15 ICP may be an exaggerated response to increased estrogen levels in pregnancy, but the mechanism of this response is unknown.16
Differential diagnosis. Other conditions that must be considered in making the diagnosis are viral hepatitis, gallbladder disease, PG, PUPPP, drug hepatotoxicity, primary biliary cirrhosis, and uremia.
Diagnosis. Laboratory values are the definitive diagnostic tool in this condition. Increased levels of serum bile acids are the single most sensitive test. Average levels of serum bile acids in pregnancy are 6.6 µmol/L, with an upper limit of 11 µmol/L. The average value in women who have ICP is 47 µmol/L.17
Although serum bile acids remain the gold standard, a recent study showed that elevated urine bile acids have 100% sensitivity and 83% specificity for ICP.18 In 55% to 60% of cases, the liver enzymes aspartate aminotransferase (AST) and alanine aminotransferase (ALT) are mildly increased. Steatorrhea is often noted by the patient, and is followed by vitamin K deficiency.17
Treatment. The current standard of care for ICP is treatment with ursodeoxycholic acid (UDCA). In four controlled trials, UDCA caused a sustained decrease in serum bile acids.19-22 The dosage used in these trials ranged from 450 to 1,200 mg/d.
Before UDCA treatment was available, ICP was treated with cholestyramine, which could bring about a 70% rate of response. The drawback to cholestyramine treatment is that it precipitates vitamin K, which is already compromised by the disease process. Further, the onset of action of cholestyramine is slow.3
Elective delivery is indicated for ICP, particularly in patients who have a significant clinical presentation.12 Delivery for ICP should be performed around weeks 37 to 38, as stillbirths tend to cluster around weeks 37 to 39.14 Given the significant fetal mortality associated with ICP (see the next paragraph), the condition should be managed by a clinician experienced with the disease—likely, a gastroenterologist.
Sequelae. The impact of this maternal disorder on the fetus can be disastrous: a 10% to 15% rate of perinatal death, and a 30% to 40% rate of premature labor.14 Fortunately, the rate of preterm labor correlates strongly with the level of bile acids, so that as bile acid levels are reduced with UDCA treatment, the rate of preterm labor also falls. Management of ICP has reduced the rate of perinatal death to 3.5%. No evidence of fetal growth retardation has been noted.14
DERMATOSES TRIGGERED BY PREGNANCY
4. Eczema of pregnancy/prurigo of pregnancy
Eczema of pregnancy/prurigo of pregnancy (EP/PP) may not actually be correlated with the pregnant state. Both conditions manifest as eczematous lesions in an atopic distribution. Although they have been described in the literature as separate entities, the lack of clinical distinction between them led Ambros-Rudolph and colleagues to combine them under the umbrella term, atopic eruption of pregnancy.23
In some patients, at least, EP/PP may be preexisting conditions that are exacerbated by pregnancy. One study of 255 patients with the condition found that 20% had had the lesions before they became pregnant.23 The tendency of the condition to be made markedly worse by pregnancy, however, leads us to include it here.
PP has an estimated incidence of 1 in 450 pregnancies.11 Although many authorities consider EP to be the most common dermatosis of pregnancy, no clear estimation of its prevalence has been established.23,24 Taken together, the two conditions have the highest prevalence of all pregnancy-induced dermatoses.
PP is also known as popular dermatitis of Spangler, Nurse’s early prurigo of pregnancy, and linear IgM disease of pregnancy.3,4,23
Presentation. The typical presentation is grouped, crusted, erythematous papules, patches, and plaques—frequently with excoriations. Lesions typically present on the extensor surfaces of the arms and legs or on the abdomen (FIGURE 4).4 Recurrence in later pregnancies is common.

FIGURE 4 Eczema of pregnancy
EP/PP typically manifests as grouped, crusted, erythematous papules, patches, and plaques, often with excoriations.
Pathophysiology. The pathophysiology of EP/PP is not understood. Many patients who develop EP/PP have a history of atopy.10
Differential diagnosis. Conditions that need to be considered in making the diagnosis include Tinea infection, scabies, contact dermatitis, ICP, pruritic folliculitis of pregnancy (PFP), and PG.
Diagnosis. The history and the physical examination determine the diagnosis. Serology, histopathology, and immunofluorescence are nonspecific. Correlation of EP/PP with increased IgE is marginal, at best.24,25
Treatment. These conditions are treated symptomatically with topical corticosteroids or systemic antihistamines.
Sequelae. No increase in maternal or fetal morbidity or mortality is associated with EP/PP.
5. Acute pustular psoriasis of pregnancy
Whether or not acute pustular psoriasis of pregnancy (APPP) is actually a pregnancy-induced dermatosis is subject to debate.
There is evidence that APPP is not unique to pregnancy but simply a manifestation of ordinary psoriasis. Clinically and histologically, APPP is indistinguishable from pustular psoriasis. Unlike most cases of acute psoriasis, however, APPP often appears in pregnancy without any personal or family history of psoriasis and usually ceases when the pregnancy ends. This fact, combined with reports of increased fetal and maternal morbidity and mortality associated with APPP, leads us to include it here.26
Presentation. APPP is a rare condition that may have an onset at any point in pregnancy. Characteristic lesions begin as erythematous plaques with pustules on the inner thighs, flexural areas, and groin and spread to the trunk and extremities. As plaques enlarge, their center becomes eroded and crusted.
The nails may become onycholytic. Hands, feet, and face are usually spared. Oral and esophageal erosions can occur. Pruritus is typically mild, although the lesions are often painful. Flu-like symptoms are often present.27
Pathophysiology. The pathophysiology of APPP is unknown.
Differential diagnosis. Conditions with similar presentations include an adverse drug reaction, pityriasis rosea, lichen simplex chronicus, eczema, lupus, and pityriasis rubra pilaris.
Diagnosis. The clinical history and an association with systemic illness are the basis for a diagnosis of APPP. Cultures of pustules are negative for any infective pathology, although, as the disease progresses, pustules may become superinfected. Laboratory testing may show an increased erythrocyte sedimentation rate, hypocalcemia, and a low level of vitamin D.
Treatment. Prednisone, 15 to 60 mg/d, is often sufficient to control the disease.27 Cyclosporine, 100 mg twice daily, has also been shown to be useful.28 Cyclosporine in pregnancy is a Category C drug. Data on fetal malformation associated with cyclosporine therapy are limited, but risk appears minimal.6
Maternal hypocalcemia should be monitored and treated appropriately. If disease progression is judged serious enough, early induction of labor is indicated, because delivery will almost always lead to swift resolution.
Sequelae. A number of case reports link APPP to serious sequelae, including fetal growth retardation, hypocalcemia, and stillbirth.27,29,30 The condition is too rare, however, for good data on specific sequelae. Although APPP does give significant cause for concern, it appears that some of the traditional apprehension comes from older publications reporting a rate of maternal mortality of 70% to 90%.31 This statistic has not been borne out in practice. It does appear that the mother will frequently suffer systemic symptoms, including fever and malaise.
6. Pruritic folliculitis of pregnancy
Accounts of the prevalence of pruritic folliculitis of pregnancy (PFP) vary widely. Some sources report fewer than 30 cases in all of the literature; others indicate that the prevalence is equivalent to that of PG—one in every 10,000 pregnancies.3,11 PFP most often presents in the third trimester. It often resolves before delivery, but uniformly clears within 2 weeks of delivery.
Presentation. PFP presents as papules and pustules concentrated around hair follicles (FIGURE 5). Often, lesions begin on the abdomen and spread to the extremities.24,28 The condition is often, but not always, pruritic. Patients are more likely to be concerned about what the condition means for their health than distressed by the symptoms.

FIGURE 5 Pruritic folliculitis of pregnancy
The papules and pustules of PFP are concentrated around hair follicles.
Pathophysiology. Like many other dermatoses of pregnancy, the pathophysiology of PFP is unknown. There is little evidence that the condition is immunologically or hormonally mediated, and there is no evidence of an infectious component.24,28
Differential diagnosis. PFP must be distinguished from infectious folliculitis, acneiform disorders, HIV-associated eosinophilic folliculitis, and a drug reaction.
Diagnosis. The clinical diagnosis is based on presenting symptoms and third-trimester onset. No specific laboratory or histologic analysis can be used to make a definitive diagnosis.
Treatment. As the condition is, by definition, a nonmicrobial folliculitis, the most effective therapy tends to be with a low- or midpotency topical corticosteroid, such as triamcinolone or desonide. A benzoyl peroxide wash can also be effective.
Sequelae. One study reports an increased incidence of low birth weight, but no associated morbidity or mortality has been reported in recent studies.24
- Pemphigoid gestationis is best managed with oral prednisone at doses from 20 to 60 mg per day to control symptoms
- The pruritus associated with pruritic urticarial papules and plaques of pregnancy can be safely and effectively managed with topical corticosteroids and oral antihistamines
- Treat intrahepatic cholestasis of pregnancy with ursodeoxycholic acid, which likely reduces serum bile acids as well as associated fetal morbidity and mortality
The dermatoses of pregnancy are a poorly understood group of conditions. Their only common feature is a tendency to appear during pregnancy.
Only three of these conditions are considered unique to pregnancy, however; the others are probably exacerbations of preexisting conditions triggered by pregnancy. There isn’t even complete agreement on what to call them. To make management even more complex, two patients—mother and fetus—need to be considered in decisions about care.
Who manages these patients is another matter. These conditions fall into overlapping areas of health care, where family physicians, obstetricians, and dermatologists all might have some share in responsibility for diagnosis and treatment. You need to be sufficiently familiar with these conditions so that you can differentiate those that can be treated symptomatically and those that require referral to a specialist. This review and the handy TABLE, will help you toward that end.
TABLE
Skin disorders of pregnancy: What you’ll see, how to treat
| Disorder | Lesions | Diagnosis and sequelae | Treatment | Recurrence |
|---|---|---|---|---|
| Pemphigoid gestationis3,5 | Erythematous papules that progress to vesicles and bullae, in a periumbilical distribution that spares the face, palms, and soles |
|
| Frequent; skips a pregnancy 8% of the time |
| Pruritic urticarial papules and plaques of pregnancy8-10 | Urticarial papules and plaques on the abdomen, legs, arms, buttocks, chest, and back |
| Topical corticosteroids and antihistamines | Uncommon |
| Intrahepatic cholestasis of pregnancy14,17,19-22 | No primary lesions; secondary excoriations in any area that the patient can reach |
| Ursodeoxycholic acid, 450-1,200 mg/d | Frequent |
| Eczema of pregnancy/pruritus of pregnancy4,10,24 | Grouped, crusted erythematous papules, patches, and plaques, most often on extensor surfaces of the arms and legs or on the abdomen |
| Symptomatic treatment with topical corticosteroids or antihistamines | Frequent |
| Acute pustular psoriasis of pregnancy26-28 | Erythematous plaques and pustules that start on the inner thighs and groin and spread to the trunk and extremities |
|
| Unknown |
| Pruritic folliculitis of pregnancy24,28 | Papules and pustules concentrated around hair follicles, often beginning on the abdomen and spreading to the extremities |
| Topical corticosteroids | Unknown |
DERMATOSES UNIQUE TO PREGNANCY
1. Pemphigoid gestationis
Years ago, this disorder was referred to as herpes gestationis, because the lesions are herpetiform. Pemphigoid gestationis (PG) has an incidence of approximately 1 in 10,000 pregnancies.1,2 Time of onset is usually about the 21st week of gestation, although, in about 20% of cases, the eruption appears immediately postpartum.3
Presentation. The disease usually begins with urticarial papules and plaques around the umbilicus and extremities. Bullous lesions tend to develop as the disease progresses, and are often not present on first presentation (FIGURE 1). Lesions of PG tend to spare the face, palms, and soles. Mucosal surfaces are involved in fewer than 20% of cases. In about 75% of cases, PG flares around the time of delivery, regressing spontaneously after the baby is born.4

FIGURE 1 Pemphigoid gestationis
As the disease progresses, bullous lesions tend to develop.
Pathophysiology. The pathophysiology of PG is nearly identical to that of bullous pemphigoid, a blistering skin disorder seen more often in elderly patients.5 Pemphigoid disorders are immune processes, involving an immunoglobulin G (IgG) immune response directed at a 180-kDa hemidesmosome transmembrane glycoprotein. This protein is the common target in several subepidermal blistering diseases.
Differential diagnosis. Disorders that may have some of the same features as PG include pruritic urticarial papules and plaques of pregnancy (PUPPP), erythema multiforme, intrahepatic cholestasis of pregnancy (ICP), contact dermatitis, and drug reactions.
Diagnosis. A biopsy is necessary for definitive diagnosis. Direct immunofluorescence (DIF) microscopy of a sample of perilesional skin can show tissue-bound immunoreactants. Linear deposition of the complement component protein C3 along the basement membrane zone is diagnostic for PG. IgG is also deposited about 40% of the time.3
Serum enzyme-linked immunosorbent assay (ELISA) studies are also helpful in diagnosis. They have excellent sensitivity and specificity, as well as the capacity to monitor levels of antibody, which correlate with the severity of disease.1
Treatment. Oral corticosteroids are the first-line treatment for PG, typically 20 to 60 mg/d of prednisone. Oral corticosteroids are generally most effective at ameliorating symptoms. Prednisone at a dosage of 40 to 80 mg/d for a short time has not been associated with congenital abnormalities.6 PG patients can also be treated successfully with intravenous immunoglobulin (IVIG) and cyclosporine in refractory cases.7
Pruritus associated with this condition can interfere with day-to-day activities and with the patient’s ability to sleep. Patients may also complain that the rash is painful, particularly if bullae rupture, leading to superficial ulcerations. Fortunately, the patient’s quality of life can be dramatically improved with systemic corticosteroids—with no significant risk to the fetus.
Sequelae. PG uniformly resolves within a few weeks, but the mother’s autoantibodies can be passively transferred to the fetus, causing vesicles and bullae in the newborn.8 An increased incidence of small-for-gestational age (SGA) infants has also been noted in PG, although no lasting morbidity or mortality in the offspring has been noted.5 The disease tends to recur in future pregnancies.
2. Pruritic urticarial papules and plaques of pregnancy
This condition is known by many names besides its acronym PUPPP: polymorphic eruption of pregnancy, toxemic erythema of pregnancy, and late prurigo of pregnancy.1 It is a pruritic, inflammatory skin disorder that has been variously estimated to occur in anywhere from 1 in 120 to 1 in 240 pregnancies.8 PUPPP is second only to eczema as the most common dermatosis of pregnancy.
Presentation. As the name suggests, the lesions of PUPPP are itchy, red papules that often coalesce into plaques (FIGURE 2). Lesions usually occur in primigravidas after the 34th week of gestation, although they may be seen at any time from the first trimester through the postpartum period.9
Lesions are classically found on the abdomen, sparing the umbilical area, and are found primarily in the striae. This distribution helps you to differentiate PUPPP from PG, in which lesions typically cluster around the umbilicus. Most PUPPP lesions (80% in one study) are dispersed on the abdomen, legs, arms, buttocks, chest, and back. Another 17% appear only on the abdomen and proximal thighs, and the remaining 3% on the limbs.10 Nearly 50% of the time, lesions also include discrete vesicles.11 There are no reported cases of mucosal involvement.
Patients with this condition are often very uncomfortable. The associated pruritus is severe enough to interfere with sleep. Despite the itching, however, lesions are seldom excoriated.

FIGURE 2 Pruritic urticarial papules and plaques of pregnancy
The itchy, red papules of PUPP often coalesce into plaques.
Pathophysiology. The disorder has been strongly associated with maternal weight gain and multiple gestations. One working hypothesis is that rapid abdominal distention observed in the third trimester leads to damage of the connective tissue, which then releases antigenic molecules, causing an inflammatory reaction.12 Another hypothesis is that increased levels of fetal DNA that have been detected in the skin of PUPPP patients may contribute to the pathology. One study detected male DNA in six of 10 PUPPP sufferers, but found none in any of 26 controls—pregnant women without PUPPP pathology.5 There is some evidence that patients with atopy may be predisposed to PUPPP, as well as patients who are hypertensive or obese.10,13
Differential diagnosis. Initially, PUPPP lesions can be difficult to differentiate from urticarial PG lesions. The distribution of the lesions is the best clue: PG lesions cluster around the umbilicus, whereas PUPPP lesions uniformly spare the umbilical area. Additional disorders in the PUPPP differential are atopic dermatitis, superficial urticarial allergic eruption, viral exanthema, and contact or irritant dermatitis.
Diagnosis. PUPPP can be diagnosed only by clinical observation. None of the available laboratory tests—immunofluorescence, histology, serology—yield findings specific for PUPPP, although histology and immunofluorescence can readily differentiate between this condition and PG.
Treatment. Because the disease holds no real danger for mother or fetus, treatment can be aimed solely at symptomatic relief. Mild-to-potent topical corticosteroids (consider triamcinolone or fluocinonide) should relieve pruritus within 48 to 72 hours.8 Antihistamines and, occasionally, low-dose systemic corticosteroids may also be used. Consider hydroxyzine, although diphenhydramine has the more proven safety profile in pregnancy.
Nonpharmaceutical treatments such as oil baths and emollients should also be considered. If the condition appears classic for PUPPP, it can be managed symptomatically. If there is any question about the diagnosis, however, referral to a dermatologist is prudent.
Sequelae. No increase in maternal or fetal morbidity or mortality is associated with PUPPP. Recurrence is fairly uncommon, as the disease primarily affects women during their first pregnancy.
3. Intrahepatic cholestasis of pregnancy
This condition is also called recurrent or idiopathic jaundice of pregnancy, obstetric cholestasis, and pruritus gravidarum. Intrahepatic cholestasis of pregnancy (ICP) is caused by disruption of hepatic bile flow during pregnancy. It has been recorded at a rate of approximately 10 to 150 of every 10,000 pregnancies in Europe and 70 of every 10,000 in the United States.12 In 80% of patients, time of onset is after the 30th week.14
Although this disorder is not primarily a dermatosis of pregnancy, it is a pruritic condition that often presents with excoriations in pregnant women and is associated with fetal morbidity and mortality. It’s important to be able to identify this disease early to minimize sequelae.
Presentation. There are no primary lesions with ICP. The primary presenting symptom is a generalized pruritus affecting the palms and soles, and sometimes extending to the legs and abdomen (FIGURE 3). This itching is often so severe that it leads to chronic insomnia. You may see secondary skin lesions, such as erythema and excoriations. Observable jaundice occurs in 10% to 20% of patients.3 These patients do not develop the encephalopathy that is associated with cholestasis in the nonpregnant state, however.14

FIGURE 3 Intrahepatic cholestasis of pregnancy
ICP lacks primary lesions. Shown here are the secondary erythema and excoriations that results from scratching the intense pruritis.
Pathophysiology. The genesis of this condition is thought to be a combination of genetic and environmental factors. A family history of the disorder is present in one half of cases; cases with a familial component tend to be more severe.15 ICP may be an exaggerated response to increased estrogen levels in pregnancy, but the mechanism of this response is unknown.16
Differential diagnosis. Other conditions that must be considered in making the diagnosis are viral hepatitis, gallbladder disease, PG, PUPPP, drug hepatotoxicity, primary biliary cirrhosis, and uremia.
Diagnosis. Laboratory values are the definitive diagnostic tool in this condition. Increased levels of serum bile acids are the single most sensitive test. Average levels of serum bile acids in pregnancy are 6.6 µmol/L, with an upper limit of 11 µmol/L. The average value in women who have ICP is 47 µmol/L.17
Although serum bile acids remain the gold standard, a recent study showed that elevated urine bile acids have 100% sensitivity and 83% specificity for ICP.18 In 55% to 60% of cases, the liver enzymes aspartate aminotransferase (AST) and alanine aminotransferase (ALT) are mildly increased. Steatorrhea is often noted by the patient, and is followed by vitamin K deficiency.17
Treatment. The current standard of care for ICP is treatment with ursodeoxycholic acid (UDCA). In four controlled trials, UDCA caused a sustained decrease in serum bile acids.19-22 The dosage used in these trials ranged from 450 to 1,200 mg/d.
Before UDCA treatment was available, ICP was treated with cholestyramine, which could bring about a 70% rate of response. The drawback to cholestyramine treatment is that it precipitates vitamin K, which is already compromised by the disease process. Further, the onset of action of cholestyramine is slow.3
Elective delivery is indicated for ICP, particularly in patients who have a significant clinical presentation.12 Delivery for ICP should be performed around weeks 37 to 38, as stillbirths tend to cluster around weeks 37 to 39.14 Given the significant fetal mortality associated with ICP (see the next paragraph), the condition should be managed by a clinician experienced with the disease—likely, a gastroenterologist.
Sequelae. The impact of this maternal disorder on the fetus can be disastrous: a 10% to 15% rate of perinatal death, and a 30% to 40% rate of premature labor.14 Fortunately, the rate of preterm labor correlates strongly with the level of bile acids, so that as bile acid levels are reduced with UDCA treatment, the rate of preterm labor also falls. Management of ICP has reduced the rate of perinatal death to 3.5%. No evidence of fetal growth retardation has been noted.14
DERMATOSES TRIGGERED BY PREGNANCY
4. Eczema of pregnancy/prurigo of pregnancy
Eczema of pregnancy/prurigo of pregnancy (EP/PP) may not actually be correlated with the pregnant state. Both conditions manifest as eczematous lesions in an atopic distribution. Although they have been described in the literature as separate entities, the lack of clinical distinction between them led Ambros-Rudolph and colleagues to combine them under the umbrella term, atopic eruption of pregnancy.23
In some patients, at least, EP/PP may be preexisting conditions that are exacerbated by pregnancy. One study of 255 patients with the condition found that 20% had had the lesions before they became pregnant.23 The tendency of the condition to be made markedly worse by pregnancy, however, leads us to include it here.
PP has an estimated incidence of 1 in 450 pregnancies.11 Although many authorities consider EP to be the most common dermatosis of pregnancy, no clear estimation of its prevalence has been established.23,24 Taken together, the two conditions have the highest prevalence of all pregnancy-induced dermatoses.
PP is also known as popular dermatitis of Spangler, Nurse’s early prurigo of pregnancy, and linear IgM disease of pregnancy.3,4,23
Presentation. The typical presentation is grouped, crusted, erythematous papules, patches, and plaques—frequently with excoriations. Lesions typically present on the extensor surfaces of the arms and legs or on the abdomen (FIGURE 4).4 Recurrence in later pregnancies is common.

FIGURE 4 Eczema of pregnancy
EP/PP typically manifests as grouped, crusted, erythematous papules, patches, and plaques, often with excoriations.
Pathophysiology. The pathophysiology of EP/PP is not understood. Many patients who develop EP/PP have a history of atopy.10
Differential diagnosis. Conditions that need to be considered in making the diagnosis include Tinea infection, scabies, contact dermatitis, ICP, pruritic folliculitis of pregnancy (PFP), and PG.
Diagnosis. The history and the physical examination determine the diagnosis. Serology, histopathology, and immunofluorescence are nonspecific. Correlation of EP/PP with increased IgE is marginal, at best.24,25
Treatment. These conditions are treated symptomatically with topical corticosteroids or systemic antihistamines.
Sequelae. No increase in maternal or fetal morbidity or mortality is associated with EP/PP.
5. Acute pustular psoriasis of pregnancy
Whether or not acute pustular psoriasis of pregnancy (APPP) is actually a pregnancy-induced dermatosis is subject to debate.
There is evidence that APPP is not unique to pregnancy but simply a manifestation of ordinary psoriasis. Clinically and histologically, APPP is indistinguishable from pustular psoriasis. Unlike most cases of acute psoriasis, however, APPP often appears in pregnancy without any personal or family history of psoriasis and usually ceases when the pregnancy ends. This fact, combined with reports of increased fetal and maternal morbidity and mortality associated with APPP, leads us to include it here.26
Presentation. APPP is a rare condition that may have an onset at any point in pregnancy. Characteristic lesions begin as erythematous plaques with pustules on the inner thighs, flexural areas, and groin and spread to the trunk and extremities. As plaques enlarge, their center becomes eroded and crusted.
The nails may become onycholytic. Hands, feet, and face are usually spared. Oral and esophageal erosions can occur. Pruritus is typically mild, although the lesions are often painful. Flu-like symptoms are often present.27
Pathophysiology. The pathophysiology of APPP is unknown.
Differential diagnosis. Conditions with similar presentations include an adverse drug reaction, pityriasis rosea, lichen simplex chronicus, eczema, lupus, and pityriasis rubra pilaris.
Diagnosis. The clinical history and an association with systemic illness are the basis for a diagnosis of APPP. Cultures of pustules are negative for any infective pathology, although, as the disease progresses, pustules may become superinfected. Laboratory testing may show an increased erythrocyte sedimentation rate, hypocalcemia, and a low level of vitamin D.
Treatment. Prednisone, 15 to 60 mg/d, is often sufficient to control the disease.27 Cyclosporine, 100 mg twice daily, has also been shown to be useful.28 Cyclosporine in pregnancy is a Category C drug. Data on fetal malformation associated with cyclosporine therapy are limited, but risk appears minimal.6
Maternal hypocalcemia should be monitored and treated appropriately. If disease progression is judged serious enough, early induction of labor is indicated, because delivery will almost always lead to swift resolution.
Sequelae. A number of case reports link APPP to serious sequelae, including fetal growth retardation, hypocalcemia, and stillbirth.27,29,30 The condition is too rare, however, for good data on specific sequelae. Although APPP does give significant cause for concern, it appears that some of the traditional apprehension comes from older publications reporting a rate of maternal mortality of 70% to 90%.31 This statistic has not been borne out in practice. It does appear that the mother will frequently suffer systemic symptoms, including fever and malaise.
6. Pruritic folliculitis of pregnancy
Accounts of the prevalence of pruritic folliculitis of pregnancy (PFP) vary widely. Some sources report fewer than 30 cases in all of the literature; others indicate that the prevalence is equivalent to that of PG—one in every 10,000 pregnancies.3,11 PFP most often presents in the third trimester. It often resolves before delivery, but uniformly clears within 2 weeks of delivery.
Presentation. PFP presents as papules and pustules concentrated around hair follicles (FIGURE 5). Often, lesions begin on the abdomen and spread to the extremities.24,28 The condition is often, but not always, pruritic. Patients are more likely to be concerned about what the condition means for their health than distressed by the symptoms.

FIGURE 5 Pruritic folliculitis of pregnancy
The papules and pustules of PFP are concentrated around hair follicles.
Pathophysiology. Like many other dermatoses of pregnancy, the pathophysiology of PFP is unknown. There is little evidence that the condition is immunologically or hormonally mediated, and there is no evidence of an infectious component.24,28
Differential diagnosis. PFP must be distinguished from infectious folliculitis, acneiform disorders, HIV-associated eosinophilic folliculitis, and a drug reaction.
Diagnosis. The clinical diagnosis is based on presenting symptoms and third-trimester onset. No specific laboratory or histologic analysis can be used to make a definitive diagnosis.
Treatment. As the condition is, by definition, a nonmicrobial folliculitis, the most effective therapy tends to be with a low- or midpotency topical corticosteroid, such as triamcinolone or desonide. A benzoyl peroxide wash can also be effective.
Sequelae. One study reports an increased incidence of low birth weight, but no associated morbidity or mortality has been reported in recent studies.24
- Pemphigoid gestationis is best managed with oral prednisone at doses from 20 to 60 mg per day to control symptoms
- The pruritus associated with pruritic urticarial papules and plaques of pregnancy can be safely and effectively managed with topical corticosteroids and oral antihistamines
- Treat intrahepatic cholestasis of pregnancy with ursodeoxycholic acid, which likely reduces serum bile acids as well as associated fetal morbidity and mortality
1. Sitaru C, Powell J, Messer G, Bröcker EB, Wojnarowska F, Zillikens D. Immunoblotting and enzyme linked immunosorbent assay for the diagnosis of pemphigoid gestationis. Obstet Gynecol. 2004;103(4):757-763.
2. Engineer L, Bohl K, Ahmed AR. Pemphigoid gestationis: a review. Am J Obstet Gynecol. 2000;183(2):483-491.
3. Kroumpouzos G, Cohen LM. Specific dermatoses of pregnancy: an evidence-based systematic review. Am J Obstet Gynecol. 2003;188(4):1083-1092.
4. Shornick JK. Dermatoses of pregnancy. Semin Cutan Med Surg. 1998;17(3):172-181.
5. Shornick JK, Bangert JL, Freeman RG, Gilliam JN. Herpes gestationis: clinical and histological features in 28 cases. J Am Acad Dermatol. 1983;8(2):214-224.
6. Leachman SA, Reed BR. The use of dermatologic drugs in pregnancy and lactation. Dermatol Clin. 2006;24(2):167-197, vi.
7. Hern S, Harman K, Bhogal BS, Black MM. A severe persistent case of pemphigoid gestationis treated with intravenous immunoglobulins and cyclosporine. Clin Exp Dermatol. 1998;23(4):185-188.
8. Fitzpatrick TP. Diseases in pregnancy. Color Atlas and Synopsis of Clinical Dermatology. New York, NY: McGraw Hill; 1997:414–419.
9. Aractingi D, Berkane N, Bertheau P, et al. Fetal DNA in skin of polymorphic eruptions of pregnancy. Lancet. 1998;352(9144):1898-1901.
10. Rudolph CM, Al-Fares S, Vaughan-Jones SA, et al. Polymorphic eruption of pregnancy: clinicopathology and potential trigger factors in 181 patients. Br J Dermatol. 2006;154(1):54-60.
11. Roger D, Vaillant L, Fignon A, et al. Specific pruritic diseases of pregnancy. A prospective study of 3192 pregnant women. Arch Dermatol. 1994;130(6):734-739.
12. McDonald JA. Cholestasis of pregnancy. J Gastroenterol Hepatol. 1999;14(6):515-518.
13. Ohel I, Levy A, Silberstein T, Holcberg G, Sheiner E. Pregnancy outcome of patients with pruritic urticarial papules and plaques of pregnancy. J Maternal Fetal Neonat Med. 2006;19(5):305-308.
14. Geenes V, Williamson C. Intrahepatic cholestasis of pregnancy. World J Gastroenterol. 2009;15(17):2049-2066.
15. Shaw D, Frohlich J, Wittmann BA, Willms M. A prospective study of 18 patients with cholestasis of pregnancy. Am J Obstet Gynecol. 1982;142(6 Pt 1):621-625.
16. Reyes H. The spectrum of liver and gastrointestinal disease seen in cholestasis of pregnancy. Gastroenterol Clin North Am. 1992;21(4):905-921.
17. Lammert F, Marschall H, Glantz A, Matern S. Intrahepatic cholestasis of pregnancy; molecular pathogenesis, diagnosis and management. J Hepatol. 2000;33(6):1012-1021.
18. Huang WM, Seubert DE, Donnelly JG, Liu M, Javitt NB. Intrahepatic cholestasis of pregnancy: detection with urinary bile acid assays. J Perinat Med. 2007;35(6):486-491.
19. Palma J, Reyes H, Ribalta J, et al. Ursodeoxycholic acid in the treatment of intrahepatic cholestasis of pregnancy: a randomized, double-blind study controlled with placebo. J Hepatol. 1997;27(6):1022-1028.
20. Diaferia A, Nicastri PL, Tartagni M, et al. Ursodeoxycholic acid therapy in pregnant women with cholestasis. Int J Gynaecol Obstet. 1996;52:133-140.
21. Nicastri PL, Diaferia A, Tartagni M, Loizzi P, Fanelli M. A randomised placebo-controlled trial of ursodeoxycholic acid and S-adenosylmethionine in the treatment of intrahepatic cholestasis of pregnancy. Br J Obstet Gynaecol. 1998;105(11):1205-1207.
22. Glantz A, Marschall HU, Lammert F, Mattsson LA. Intrahepatic cholestasis in pregnancy: a randomized controlled trial comparing dexamethasone and ursodeoxycholic acid. Hepatology. 2005;42(6):1399-1405.
23. Ambros-Rudolph C, Müllegger M, Vaughan-Jones S, et al. The specific dermatoses of pregnancy revisited and reclassified: results of a retrospective two-center study on 505 pregnant patients. J Am Acad Dermatol. 2006;54(3):395-404.
24. Vaughan Jones SA, Hern S, Nelson-Piercy C, Seed PT, Black MM. A prospective study of 200 women with dermatoses of pregnancy correlating clinical findings with hormonal and immunopathological profiles. Br J Dermatol. 1999;141(1):71-81.
25. Holmes RC, Black MM. The specific dermatoses of pregnancy. J Am Acad Dermatol. 1983;8(3):405-412.
26. Bukhari IA. Impetigo herpetiformis in a primagravida: successful treatment with etretinate. J Drugs Dermatol. 2004;3(4):449-451.
27. Oumeish OY, Parish JL. Impetigo herpetiformis. Clin Dermatol. 2006;24(2):101-104.
28. Kroumpouzos G, Cohen LM. Pruritic folliculitis of pregnancy. J Am Acad Dermatol. 2000;43(1 Pt 1):132-134.
29. Sahin HG, Sahin HA, Metin A, Zeteroglu S, Ugras S. Recurrent impetigo herpetiformis in a pregnant adolescent: case report. Eur J Obstet Gynecol Reprod Biol. 2002;101:201-203.
30. Aka N, Kuscu NK, Yazicioglu E. Impetigo herpetiformis at the 36th week of gestation. Int J Gynecol Obstet. 2000;69(2):153-154.
31. Wade T, Wade S, Jones H. Skin changes and diseases associated with pregnancy. Obstet Gynecol. 1978;52(2):233-242.
1. Sitaru C, Powell J, Messer G, Bröcker EB, Wojnarowska F, Zillikens D. Immunoblotting and enzyme linked immunosorbent assay for the diagnosis of pemphigoid gestationis. Obstet Gynecol. 2004;103(4):757-763.
2. Engineer L, Bohl K, Ahmed AR. Pemphigoid gestationis: a review. Am J Obstet Gynecol. 2000;183(2):483-491.
3. Kroumpouzos G, Cohen LM. Specific dermatoses of pregnancy: an evidence-based systematic review. Am J Obstet Gynecol. 2003;188(4):1083-1092.
4. Shornick JK. Dermatoses of pregnancy. Semin Cutan Med Surg. 1998;17(3):172-181.
5. Shornick JK, Bangert JL, Freeman RG, Gilliam JN. Herpes gestationis: clinical and histological features in 28 cases. J Am Acad Dermatol. 1983;8(2):214-224.
6. Leachman SA, Reed BR. The use of dermatologic drugs in pregnancy and lactation. Dermatol Clin. 2006;24(2):167-197, vi.
7. Hern S, Harman K, Bhogal BS, Black MM. A severe persistent case of pemphigoid gestationis treated with intravenous immunoglobulins and cyclosporine. Clin Exp Dermatol. 1998;23(4):185-188.
8. Fitzpatrick TP. Diseases in pregnancy. Color Atlas and Synopsis of Clinical Dermatology. New York, NY: McGraw Hill; 1997:414–419.
9. Aractingi D, Berkane N, Bertheau P, et al. Fetal DNA in skin of polymorphic eruptions of pregnancy. Lancet. 1998;352(9144):1898-1901.
10. Rudolph CM, Al-Fares S, Vaughan-Jones SA, et al. Polymorphic eruption of pregnancy: clinicopathology and potential trigger factors in 181 patients. Br J Dermatol. 2006;154(1):54-60.
11. Roger D, Vaillant L, Fignon A, et al. Specific pruritic diseases of pregnancy. A prospective study of 3192 pregnant women. Arch Dermatol. 1994;130(6):734-739.
12. McDonald JA. Cholestasis of pregnancy. J Gastroenterol Hepatol. 1999;14(6):515-518.
13. Ohel I, Levy A, Silberstein T, Holcberg G, Sheiner E. Pregnancy outcome of patients with pruritic urticarial papules and plaques of pregnancy. J Maternal Fetal Neonat Med. 2006;19(5):305-308.
14. Geenes V, Williamson C. Intrahepatic cholestasis of pregnancy. World J Gastroenterol. 2009;15(17):2049-2066.
15. Shaw D, Frohlich J, Wittmann BA, Willms M. A prospective study of 18 patients with cholestasis of pregnancy. Am J Obstet Gynecol. 1982;142(6 Pt 1):621-625.
16. Reyes H. The spectrum of liver and gastrointestinal disease seen in cholestasis of pregnancy. Gastroenterol Clin North Am. 1992;21(4):905-921.
17. Lammert F, Marschall H, Glantz A, Matern S. Intrahepatic cholestasis of pregnancy; molecular pathogenesis, diagnosis and management. J Hepatol. 2000;33(6):1012-1021.
18. Huang WM, Seubert DE, Donnelly JG, Liu M, Javitt NB. Intrahepatic cholestasis of pregnancy: detection with urinary bile acid assays. J Perinat Med. 2007;35(6):486-491.
19. Palma J, Reyes H, Ribalta J, et al. Ursodeoxycholic acid in the treatment of intrahepatic cholestasis of pregnancy: a randomized, double-blind study controlled with placebo. J Hepatol. 1997;27(6):1022-1028.
20. Diaferia A, Nicastri PL, Tartagni M, et al. Ursodeoxycholic acid therapy in pregnant women with cholestasis. Int J Gynaecol Obstet. 1996;52:133-140.
21. Nicastri PL, Diaferia A, Tartagni M, Loizzi P, Fanelli M. A randomised placebo-controlled trial of ursodeoxycholic acid and S-adenosylmethionine in the treatment of intrahepatic cholestasis of pregnancy. Br J Obstet Gynaecol. 1998;105(11):1205-1207.
22. Glantz A, Marschall HU, Lammert F, Mattsson LA. Intrahepatic cholestasis in pregnancy: a randomized controlled trial comparing dexamethasone and ursodeoxycholic acid. Hepatology. 2005;42(6):1399-1405.
23. Ambros-Rudolph C, Müllegger M, Vaughan-Jones S, et al. The specific dermatoses of pregnancy revisited and reclassified: results of a retrospective two-center study on 505 pregnant patients. J Am Acad Dermatol. 2006;54(3):395-404.
24. Vaughan Jones SA, Hern S, Nelson-Piercy C, Seed PT, Black MM. A prospective study of 200 women with dermatoses of pregnancy correlating clinical findings with hormonal and immunopathological profiles. Br J Dermatol. 1999;141(1):71-81.
25. Holmes RC, Black MM. The specific dermatoses of pregnancy. J Am Acad Dermatol. 1983;8(3):405-412.
26. Bukhari IA. Impetigo herpetiformis in a primagravida: successful treatment with etretinate. J Drugs Dermatol. 2004;3(4):449-451.
27. Oumeish OY, Parish JL. Impetigo herpetiformis. Clin Dermatol. 2006;24(2):101-104.
28. Kroumpouzos G, Cohen LM. Pruritic folliculitis of pregnancy. J Am Acad Dermatol. 2000;43(1 Pt 1):132-134.
29. Sahin HG, Sahin HA, Metin A, Zeteroglu S, Ugras S. Recurrent impetigo herpetiformis in a pregnant adolescent: case report. Eur J Obstet Gynecol Reprod Biol. 2002;101:201-203.
30. Aka N, Kuscu NK, Yazicioglu E. Impetigo herpetiformis at the 36th week of gestation. Int J Gynecol Obstet. 2000;69(2):153-154.
31. Wade T, Wade S, Jones H. Skin changes and diseases associated with pregnancy. Obstet Gynecol. 1978;52(2):233-242.
The Earlier, the Better
Most hospitals work hard to increase the portion of discharges that occur early in the workday and decrease the number that occur in the afternoon or evening. In every case, hospitalists have an important role in making this happen.
In my April 2009 column (“Top O’ the Morning,” p. 53), I wrote about why this is important to hospitals and which strategies hospitalists could adopt. But this is still such a big issue for hospitalists that I thought I would elaborate on a few of the really simple ideas. Your HM group could implement most of the following strategies beginning next week, and you wouldn’t need months of meetings with other hospital departments.
But before I get to the ideas, I want to mention a couple of other things. First, I can’t resist pointing out that giving hospitalists a financial incentive for writing the majority of discharge orders by a certain time in the morning has met with mixed results, despite the fact that some institutions believe this approach is valuable. In the absence of computerized physician order entry (CPOE), it can be really difficult to track exactly when the doctor wrote the discharge order. And, more importantly, a financial incentive might discourage a hospitalist from discharging a patient this afternoon, and they might instead wait to discharge tomorrow morning—adding to length of stay and defeating the goal of the incentive.
It turns out that a lot has been written about throughput; just do an Internet search and pair “throughput” with terms like “hospital,” “hospitalist,” “ED,” etc. Remarkably, I haven’t been able to dig up much material that specifically addresses early-morning discharges, which is an important component of throughput.
Let’s turn our attention to some specific recommendations for increasing morning discharges. Remember, I’ve selected these because they’re easy to implement and won’t require HM groups to negotiate with others at the hospital.
Write “Probable Discharge Tomorrow” Orders
Letting other staff know the anticipated discharge date via an order in the chart typically is more effective than writing the same information in the progress note section of the chart. Although a hospitalist should verbally communicate the anticipated discharge date with the patient’s nurse and discharge planner, it still is worthwhile to write an order, because it increases the chance all, or nearly all, staff (e.g., night nurses) will be aware of the plan and communicate the same message to the family.
Your group could establish a rule or financial incentive, such that all charts will be reviewed after discharge, and a certain portion (e.g., 85%) must have such an order written sometime prior to discharge. It doesn’t always need to be written on the day prior to discharge; instead, an order written on Monday could say “likely discharge on Wednesday or Thursday.” And, of course, there shouldn’t be a requirement that the patient actually be discharged on the day that was forecast.
Prepare the Day Prior
Typically, hospitalized-patient discharges are very time-consuming. Most discharges are complicated by last-minute medical or social loose ends that require attention. Routinely trying to uncover and address these on the evening prior to discharge will ensure that a larger percentage of patients will be discharged—and vacate their room—earlier the next day. Here is what this might look like:
On Tuesday, Dr. Guaraldi is wrapping up most work for the day. He stops by to see his patient, Mr. Schultz, to see if he is improving as expected. Indeed, Mr. Schultz is looking better and probably will be ready for discharge Wednesday morning. So, Dr. Guaraldi talks with Mr. Schultz and calls the patient’s daughter to answer any questions and concerns, ensuring no surprises by the Wednesday-morning discharge. When the daughter asks (as nearly all family members do) what time she should plan to pick up her dad, Dr. Guaraldi can suggest a time based on when he will be able to round in the morning. He also can arrange to have the discharge planning staff alerted if there are more complicated issues (e.g., arranging for professional transport home).
Dr. Guaraldi then dictates the discharge summary, addresses the discharge medicine reconciliation, and writes the prescriptions. In doing so, he might uncover some loose ends and might end up ordering a lab or imaging test to be done in the evening so the results will be available early Wednesday morning and won’t delay the routine discharge.
On Wednesday morning, Dr. Guaraldi rounds on Mr. Schultz early, finds the patient is improving as expected, and writes the discharge order. The whole visit takes only a few minutes, as most of the time-consuming work was completed the prior evening. In fact, because it is a relatively short visit, it is a lot easier for Dr. Guaraldi to arrange to round on Mr. Schultz early in the day (e.g., even on the way to see ICU patients), as the hospital’s chief medical officer is always asking him to do.
I hope this scenario doesn’t sound too difficult. (Another benefit of dictating discharge summaries the evening before discharge is that the typed document should be available the next morning, so the patient can have a copy to take with him at discharge.) Of course, it won’t apply to all patients, such as those patients whose discharges can’t be predicted.
Many hospitalists think arranging for discharge the evening before is impossible because “I’m just too spent at the end of a long day to stay late getting patients ready for discharge tomorrow!” But realize you won’t be doing any more work; you’re rearranging when you do the work. The time you spend arranging for discharge in advance will save you time and stress tomorrow. My own experience is that it is much easier to do all the discharge work the evening before than in the morning when I’m so busy and am being pulled in 10 different directions. Most morning discharge visits are relatively quick and painless, which is really valuable for increasing the efficiency and decreasing the stress of morning rounds.
The alert reader already has figured out there is a pretty big cost to doing the discharge work the evening before. Some patients won’t be able to discharge as planned (e.g., they have a fever overnight) and the preparations will have been in vain. My experience is that such “failed” discharges are reasonably common, but even when they occur, it is usually reasonable to use most of the original prescriptions and discharge summary, with an addendum as required. For example, Dr. Guaraldi could dictate an addendum stating:
“The patient originally was planned for discharge on Wednesday but had a temperature of 38.6 degrees Celsius the night before, so stayed in the hospital for two more days for … ”
Start Rounds Earlier
This strategy might be the most difficult for you and your HM group to arrange, but I propose it because you could do it without having to negotiate with a lot of other departments in the hospital. If your group currently has a day shift that starts at 8 a.m. with a team conference, you could instead start at 7 a.m. Your group could try to shorten the duration of the morning team conference, or eliminate it. Whether the need to get patients discharged early in the day is worth the complexity of rearranging your schedule will depend on the circumstances of your hospital and your group. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Most hospitals work hard to increase the portion of discharges that occur early in the workday and decrease the number that occur in the afternoon or evening. In every case, hospitalists have an important role in making this happen.
In my April 2009 column (“Top O’ the Morning,” p. 53), I wrote about why this is important to hospitals and which strategies hospitalists could adopt. But this is still such a big issue for hospitalists that I thought I would elaborate on a few of the really simple ideas. Your HM group could implement most of the following strategies beginning next week, and you wouldn’t need months of meetings with other hospital departments.
But before I get to the ideas, I want to mention a couple of other things. First, I can’t resist pointing out that giving hospitalists a financial incentive for writing the majority of discharge orders by a certain time in the morning has met with mixed results, despite the fact that some institutions believe this approach is valuable. In the absence of computerized physician order entry (CPOE), it can be really difficult to track exactly when the doctor wrote the discharge order. And, more importantly, a financial incentive might discourage a hospitalist from discharging a patient this afternoon, and they might instead wait to discharge tomorrow morning—adding to length of stay and defeating the goal of the incentive.
It turns out that a lot has been written about throughput; just do an Internet search and pair “throughput” with terms like “hospital,” “hospitalist,” “ED,” etc. Remarkably, I haven’t been able to dig up much material that specifically addresses early-morning discharges, which is an important component of throughput.
Let’s turn our attention to some specific recommendations for increasing morning discharges. Remember, I’ve selected these because they’re easy to implement and won’t require HM groups to negotiate with others at the hospital.
Write “Probable Discharge Tomorrow” Orders
Letting other staff know the anticipated discharge date via an order in the chart typically is more effective than writing the same information in the progress note section of the chart. Although a hospitalist should verbally communicate the anticipated discharge date with the patient’s nurse and discharge planner, it still is worthwhile to write an order, because it increases the chance all, or nearly all, staff (e.g., night nurses) will be aware of the plan and communicate the same message to the family.
Your group could establish a rule or financial incentive, such that all charts will be reviewed after discharge, and a certain portion (e.g., 85%) must have such an order written sometime prior to discharge. It doesn’t always need to be written on the day prior to discharge; instead, an order written on Monday could say “likely discharge on Wednesday or Thursday.” And, of course, there shouldn’t be a requirement that the patient actually be discharged on the day that was forecast.
Prepare the Day Prior
Typically, hospitalized-patient discharges are very time-consuming. Most discharges are complicated by last-minute medical or social loose ends that require attention. Routinely trying to uncover and address these on the evening prior to discharge will ensure that a larger percentage of patients will be discharged—and vacate their room—earlier the next day. Here is what this might look like:
On Tuesday, Dr. Guaraldi is wrapping up most work for the day. He stops by to see his patient, Mr. Schultz, to see if he is improving as expected. Indeed, Mr. Schultz is looking better and probably will be ready for discharge Wednesday morning. So, Dr. Guaraldi talks with Mr. Schultz and calls the patient’s daughter to answer any questions and concerns, ensuring no surprises by the Wednesday-morning discharge. When the daughter asks (as nearly all family members do) what time she should plan to pick up her dad, Dr. Guaraldi can suggest a time based on when he will be able to round in the morning. He also can arrange to have the discharge planning staff alerted if there are more complicated issues (e.g., arranging for professional transport home).
Dr. Guaraldi then dictates the discharge summary, addresses the discharge medicine reconciliation, and writes the prescriptions. In doing so, he might uncover some loose ends and might end up ordering a lab or imaging test to be done in the evening so the results will be available early Wednesday morning and won’t delay the routine discharge.
On Wednesday morning, Dr. Guaraldi rounds on Mr. Schultz early, finds the patient is improving as expected, and writes the discharge order. The whole visit takes only a few minutes, as most of the time-consuming work was completed the prior evening. In fact, because it is a relatively short visit, it is a lot easier for Dr. Guaraldi to arrange to round on Mr. Schultz early in the day (e.g., even on the way to see ICU patients), as the hospital’s chief medical officer is always asking him to do.
I hope this scenario doesn’t sound too difficult. (Another benefit of dictating discharge summaries the evening before discharge is that the typed document should be available the next morning, so the patient can have a copy to take with him at discharge.) Of course, it won’t apply to all patients, such as those patients whose discharges can’t be predicted.
Many hospitalists think arranging for discharge the evening before is impossible because “I’m just too spent at the end of a long day to stay late getting patients ready for discharge tomorrow!” But realize you won’t be doing any more work; you’re rearranging when you do the work. The time you spend arranging for discharge in advance will save you time and stress tomorrow. My own experience is that it is much easier to do all the discharge work the evening before than in the morning when I’m so busy and am being pulled in 10 different directions. Most morning discharge visits are relatively quick and painless, which is really valuable for increasing the efficiency and decreasing the stress of morning rounds.
The alert reader already has figured out there is a pretty big cost to doing the discharge work the evening before. Some patients won’t be able to discharge as planned (e.g., they have a fever overnight) and the preparations will have been in vain. My experience is that such “failed” discharges are reasonably common, but even when they occur, it is usually reasonable to use most of the original prescriptions and discharge summary, with an addendum as required. For example, Dr. Guaraldi could dictate an addendum stating:
“The patient originally was planned for discharge on Wednesday but had a temperature of 38.6 degrees Celsius the night before, so stayed in the hospital for two more days for … ”
Start Rounds Earlier
This strategy might be the most difficult for you and your HM group to arrange, but I propose it because you could do it without having to negotiate with a lot of other departments in the hospital. If your group currently has a day shift that starts at 8 a.m. with a team conference, you could instead start at 7 a.m. Your group could try to shorten the duration of the morning team conference, or eliminate it. Whether the need to get patients discharged early in the day is worth the complexity of rearranging your schedule will depend on the circumstances of your hospital and your group. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Most hospitals work hard to increase the portion of discharges that occur early in the workday and decrease the number that occur in the afternoon or evening. In every case, hospitalists have an important role in making this happen.
In my April 2009 column (“Top O’ the Morning,” p. 53), I wrote about why this is important to hospitals and which strategies hospitalists could adopt. But this is still such a big issue for hospitalists that I thought I would elaborate on a few of the really simple ideas. Your HM group could implement most of the following strategies beginning next week, and you wouldn’t need months of meetings with other hospital departments.
But before I get to the ideas, I want to mention a couple of other things. First, I can’t resist pointing out that giving hospitalists a financial incentive for writing the majority of discharge orders by a certain time in the morning has met with mixed results, despite the fact that some institutions believe this approach is valuable. In the absence of computerized physician order entry (CPOE), it can be really difficult to track exactly when the doctor wrote the discharge order. And, more importantly, a financial incentive might discourage a hospitalist from discharging a patient this afternoon, and they might instead wait to discharge tomorrow morning—adding to length of stay and defeating the goal of the incentive.
It turns out that a lot has been written about throughput; just do an Internet search and pair “throughput” with terms like “hospital,” “hospitalist,” “ED,” etc. Remarkably, I haven’t been able to dig up much material that specifically addresses early-morning discharges, which is an important component of throughput.
Let’s turn our attention to some specific recommendations for increasing morning discharges. Remember, I’ve selected these because they’re easy to implement and won’t require HM groups to negotiate with others at the hospital.
Write “Probable Discharge Tomorrow” Orders
Letting other staff know the anticipated discharge date via an order in the chart typically is more effective than writing the same information in the progress note section of the chart. Although a hospitalist should verbally communicate the anticipated discharge date with the patient’s nurse and discharge planner, it still is worthwhile to write an order, because it increases the chance all, or nearly all, staff (e.g., night nurses) will be aware of the plan and communicate the same message to the family.
Your group could establish a rule or financial incentive, such that all charts will be reviewed after discharge, and a certain portion (e.g., 85%) must have such an order written sometime prior to discharge. It doesn’t always need to be written on the day prior to discharge; instead, an order written on Monday could say “likely discharge on Wednesday or Thursday.” And, of course, there shouldn’t be a requirement that the patient actually be discharged on the day that was forecast.
Prepare the Day Prior
Typically, hospitalized-patient discharges are very time-consuming. Most discharges are complicated by last-minute medical or social loose ends that require attention. Routinely trying to uncover and address these on the evening prior to discharge will ensure that a larger percentage of patients will be discharged—and vacate their room—earlier the next day. Here is what this might look like:
On Tuesday, Dr. Guaraldi is wrapping up most work for the day. He stops by to see his patient, Mr. Schultz, to see if he is improving as expected. Indeed, Mr. Schultz is looking better and probably will be ready for discharge Wednesday morning. So, Dr. Guaraldi talks with Mr. Schultz and calls the patient’s daughter to answer any questions and concerns, ensuring no surprises by the Wednesday-morning discharge. When the daughter asks (as nearly all family members do) what time she should plan to pick up her dad, Dr. Guaraldi can suggest a time based on when he will be able to round in the morning. He also can arrange to have the discharge planning staff alerted if there are more complicated issues (e.g., arranging for professional transport home).
Dr. Guaraldi then dictates the discharge summary, addresses the discharge medicine reconciliation, and writes the prescriptions. In doing so, he might uncover some loose ends and might end up ordering a lab or imaging test to be done in the evening so the results will be available early Wednesday morning and won’t delay the routine discharge.
On Wednesday morning, Dr. Guaraldi rounds on Mr. Schultz early, finds the patient is improving as expected, and writes the discharge order. The whole visit takes only a few minutes, as most of the time-consuming work was completed the prior evening. In fact, because it is a relatively short visit, it is a lot easier for Dr. Guaraldi to arrange to round on Mr. Schultz early in the day (e.g., even on the way to see ICU patients), as the hospital’s chief medical officer is always asking him to do.
I hope this scenario doesn’t sound too difficult. (Another benefit of dictating discharge summaries the evening before discharge is that the typed document should be available the next morning, so the patient can have a copy to take with him at discharge.) Of course, it won’t apply to all patients, such as those patients whose discharges can’t be predicted.
Many hospitalists think arranging for discharge the evening before is impossible because “I’m just too spent at the end of a long day to stay late getting patients ready for discharge tomorrow!” But realize you won’t be doing any more work; you’re rearranging when you do the work. The time you spend arranging for discharge in advance will save you time and stress tomorrow. My own experience is that it is much easier to do all the discharge work the evening before than in the morning when I’m so busy and am being pulled in 10 different directions. Most morning discharge visits are relatively quick and painless, which is really valuable for increasing the efficiency and decreasing the stress of morning rounds.
The alert reader already has figured out there is a pretty big cost to doing the discharge work the evening before. Some patients won’t be able to discharge as planned (e.g., they have a fever overnight) and the preparations will have been in vain. My experience is that such “failed” discharges are reasonably common, but even when they occur, it is usually reasonable to use most of the original prescriptions and discharge summary, with an addendum as required. For example, Dr. Guaraldi could dictate an addendum stating:
“The patient originally was planned for discharge on Wednesday but had a temperature of 38.6 degrees Celsius the night before, so stayed in the hospital for two more days for … ”
Start Rounds Earlier
This strategy might be the most difficult for you and your HM group to arrange, but I propose it because you could do it without having to negotiate with a lot of other departments in the hospital. If your group currently has a day shift that starts at 8 a.m. with a team conference, you could instead start at 7 a.m. Your group could try to shorten the duration of the morning team conference, or eliminate it. Whether the need to get patients discharged early in the day is worth the complexity of rearranging your schedule will depend on the circumstances of your hospital and your group. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Pessary and pelvic floor exercises for incontinence—are two better than one?
Because we will all be seeing more patients with stress urinary incontinence and other urogynecologic issues, it is critical that we keep abreast of the treatment options available—and their relative effectiveness.1
In this exploration of nonsurgical approaches to stress incontinence, Richter and colleagues started with the premise that a combination of instructed pelvic floor exercises and an incontinence pessary would be better than either treatment alone. They (very appropriately) designated the following as primary outcome measures:
- patient-reported improvement
- symptoms of stress incontinence
- patient satisfaction, as measured using validated instruments.
As reported above, combination therapy did not prove to be superior to single-modality intervention. And although behavioral therapy was superior to a pessary at 3 months, by 12 months the modalities were roughly equivalent, and only about half of patients were still using the prescribed therapy: pessary (45%) or pelvic floor exercises (57%).
This is not a real-world study
Most women who have stress incontinence and who select nonsurgical therapy choose only one option—pelvic floor exercises (if very motivated), a vaginal pessary or other device (if not so motivated), or another conservative option such as radiofrequency therapy (if even less motivated). In this study, women enrolled in behavioral therapy paid four visits (at roughly 2-week intervals) to approved “interventionists,” who instructed them in the technique for pelvic floor exercise and explained other skills and strategies to prevent urge and stress incontinence.
Many women find it difficult to attend the four to eight physiotherapy sessions that are necessary for behavioral intervention and are unwilling to devote 1 year to a therapy that they don’t find effective early on. (Physiotherapy is effective but requires a motivated patient.) Other women dislike inserting a vaginal device on a regular basis. 2
What’s more, very safe minimally invasive slings are available that offer more definitive therapy to patients who have stress incontinence. That said, a sling procedure should not be undertaken lightly. Patient selection should be based on preoperative testing, including an assessment of urethral function, for the transobturator sling.3 A retropubic sling requires a greater degree of expertise to tension appropriately but is suitable for a wider range of severity, including intrinsic sphincteric deficiency. The role of single-incision slings is unclear.
Bottom line: individualize care
The authors’ concluding statements are right on the money: “Individualization of care should continue to be the cornerstone of our approach to [stress incontinent] patients.” These women have several effective options available. We should help them make an educated choice based on symptom severity, lifestyle, and willingness to enroll in self-help intervention versus surgical therapy.
Many patients seek to avoid surgery, either because they believe that their stress incontinence is not severe enough to warrant it, or because they are unwilling to take the 6 to 8 weeks of relative inactivity required for the sling to settle in.
In the absence of approved pharmacotherapy for stress incontinence, I tell patients that they 1) can expect their symptoms to become worse over time and 2) should designate a period of time for a trial of conservative therapy—usually, 3 months. If their condition has not improved to their satisfaction over that period, I recommend that they identify a 6-week window during which they can avoid the gym and the golf course, as well as sexual activity, to allow for unstressed healing from a sling procedure.—G. WILLY DAVILA, MD
1. Wu JM, Hundley AF, Fulton RG, Myers ER. Forecasting the prevalence of pelvic floor disorders in US women: 2010 to 2050. Obstet Gynecol. 2009;114(6):1278-1283.
2. Davila GW, Bernier F. Multimodality pelvic physiotherapy treatment of urinary incontinence in adult women. Int Urogynecol J Pelvic Floor Dysfunct. 1995;6(4):187-194.
3. Guerette N, Bena J, Davila GW. Transobturator slings for stress incontinence: using urodynamic parameters to predict outcomes. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19(1):97-102.
Because we will all be seeing more patients with stress urinary incontinence and other urogynecologic issues, it is critical that we keep abreast of the treatment options available—and their relative effectiveness.1
In this exploration of nonsurgical approaches to stress incontinence, Richter and colleagues started with the premise that a combination of instructed pelvic floor exercises and an incontinence pessary would be better than either treatment alone. They (very appropriately) designated the following as primary outcome measures:
- patient-reported improvement
- symptoms of stress incontinence
- patient satisfaction, as measured using validated instruments.
As reported above, combination therapy did not prove to be superior to single-modality intervention. And although behavioral therapy was superior to a pessary at 3 months, by 12 months the modalities were roughly equivalent, and only about half of patients were still using the prescribed therapy: pessary (45%) or pelvic floor exercises (57%).
This is not a real-world study
Most women who have stress incontinence and who select nonsurgical therapy choose only one option—pelvic floor exercises (if very motivated), a vaginal pessary or other device (if not so motivated), or another conservative option such as radiofrequency therapy (if even less motivated). In this study, women enrolled in behavioral therapy paid four visits (at roughly 2-week intervals) to approved “interventionists,” who instructed them in the technique for pelvic floor exercise and explained other skills and strategies to prevent urge and stress incontinence.
Many women find it difficult to attend the four to eight physiotherapy sessions that are necessary for behavioral intervention and are unwilling to devote 1 year to a therapy that they don’t find effective early on. (Physiotherapy is effective but requires a motivated patient.) Other women dislike inserting a vaginal device on a regular basis. 2
What’s more, very safe minimally invasive slings are available that offer more definitive therapy to patients who have stress incontinence. That said, a sling procedure should not be undertaken lightly. Patient selection should be based on preoperative testing, including an assessment of urethral function, for the transobturator sling.3 A retropubic sling requires a greater degree of expertise to tension appropriately but is suitable for a wider range of severity, including intrinsic sphincteric deficiency. The role of single-incision slings is unclear.
Bottom line: individualize care
The authors’ concluding statements are right on the money: “Individualization of care should continue to be the cornerstone of our approach to [stress incontinent] patients.” These women have several effective options available. We should help them make an educated choice based on symptom severity, lifestyle, and willingness to enroll in self-help intervention versus surgical therapy.
Many patients seek to avoid surgery, either because they believe that their stress incontinence is not severe enough to warrant it, or because they are unwilling to take the 6 to 8 weeks of relative inactivity required for the sling to settle in.
In the absence of approved pharmacotherapy for stress incontinence, I tell patients that they 1) can expect their symptoms to become worse over time and 2) should designate a period of time for a trial of conservative therapy—usually, 3 months. If their condition has not improved to their satisfaction over that period, I recommend that they identify a 6-week window during which they can avoid the gym and the golf course, as well as sexual activity, to allow for unstressed healing from a sling procedure.—G. WILLY DAVILA, MD
Because we will all be seeing more patients with stress urinary incontinence and other urogynecologic issues, it is critical that we keep abreast of the treatment options available—and their relative effectiveness.1
In this exploration of nonsurgical approaches to stress incontinence, Richter and colleagues started with the premise that a combination of instructed pelvic floor exercises and an incontinence pessary would be better than either treatment alone. They (very appropriately) designated the following as primary outcome measures:
- patient-reported improvement
- symptoms of stress incontinence
- patient satisfaction, as measured using validated instruments.
As reported above, combination therapy did not prove to be superior to single-modality intervention. And although behavioral therapy was superior to a pessary at 3 months, by 12 months the modalities were roughly equivalent, and only about half of patients were still using the prescribed therapy: pessary (45%) or pelvic floor exercises (57%).
This is not a real-world study
Most women who have stress incontinence and who select nonsurgical therapy choose only one option—pelvic floor exercises (if very motivated), a vaginal pessary or other device (if not so motivated), or another conservative option such as radiofrequency therapy (if even less motivated). In this study, women enrolled in behavioral therapy paid four visits (at roughly 2-week intervals) to approved “interventionists,” who instructed them in the technique for pelvic floor exercise and explained other skills and strategies to prevent urge and stress incontinence.
Many women find it difficult to attend the four to eight physiotherapy sessions that are necessary for behavioral intervention and are unwilling to devote 1 year to a therapy that they don’t find effective early on. (Physiotherapy is effective but requires a motivated patient.) Other women dislike inserting a vaginal device on a regular basis. 2
What’s more, very safe minimally invasive slings are available that offer more definitive therapy to patients who have stress incontinence. That said, a sling procedure should not be undertaken lightly. Patient selection should be based on preoperative testing, including an assessment of urethral function, for the transobturator sling.3 A retropubic sling requires a greater degree of expertise to tension appropriately but is suitable for a wider range of severity, including intrinsic sphincteric deficiency. The role of single-incision slings is unclear.
Bottom line: individualize care
The authors’ concluding statements are right on the money: “Individualization of care should continue to be the cornerstone of our approach to [stress incontinent] patients.” These women have several effective options available. We should help them make an educated choice based on symptom severity, lifestyle, and willingness to enroll in self-help intervention versus surgical therapy.
Many patients seek to avoid surgery, either because they believe that their stress incontinence is not severe enough to warrant it, or because they are unwilling to take the 6 to 8 weeks of relative inactivity required for the sling to settle in.
In the absence of approved pharmacotherapy for stress incontinence, I tell patients that they 1) can expect their symptoms to become worse over time and 2) should designate a period of time for a trial of conservative therapy—usually, 3 months. If their condition has not improved to their satisfaction over that period, I recommend that they identify a 6-week window during which they can avoid the gym and the golf course, as well as sexual activity, to allow for unstressed healing from a sling procedure.—G. WILLY DAVILA, MD
1. Wu JM, Hundley AF, Fulton RG, Myers ER. Forecasting the prevalence of pelvic floor disorders in US women: 2010 to 2050. Obstet Gynecol. 2009;114(6):1278-1283.
2. Davila GW, Bernier F. Multimodality pelvic physiotherapy treatment of urinary incontinence in adult women. Int Urogynecol J Pelvic Floor Dysfunct. 1995;6(4):187-194.
3. Guerette N, Bena J, Davila GW. Transobturator slings for stress incontinence: using urodynamic parameters to predict outcomes. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19(1):97-102.
1. Wu JM, Hundley AF, Fulton RG, Myers ER. Forecasting the prevalence of pelvic floor disorders in US women: 2010 to 2050. Obstet Gynecol. 2009;114(6):1278-1283.
2. Davila GW, Bernier F. Multimodality pelvic physiotherapy treatment of urinary incontinence in adult women. Int Urogynecol J Pelvic Floor Dysfunct. 1995;6(4):187-194.
3. Guerette N, Bena J, Davila GW. Transobturator slings for stress incontinence: using urodynamic parameters to predict outcomes. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19(1):97-102.
What can be safer than having a baby in the USA?
A recent report1 by the National Highway Traffic Safety Administration revealed that deaths from automobile accidents have declined strikingly since 1954, reaching the lowest level ever recorded today: 1.16 fatalities for every 100 million vehicle-miles traveled. Why this improvement? Reasons include an increase in seat belt use and a lower rate of drunk driving, both secondary effects of better law enforcement, safer roads, and people just driving less.
There are lessons to be learned from this success that can be applied to obstetrical care and, potentially, to lower the unacceptably high rate of maternal mortality in this country.
Things aren’t working as we need them to
Truly, it is a reflection of a broken health care system that we, as health care providers, cannot say that we have seen a dramatic decrease in maternal mortality. In 2006, the latest year for which statistics are available, maternal mortality in the United States was at the highest level recorded since 1991: 13.3 deaths for every 100,000 live births (in California, the rate is 16.9 for every 100,000!).
Remember the Healthy People 2010 benchmark of 4.3 maternal deaths for every 100,000 live births? Clearly, we will not see that number any time in the near future; achieving it is as likely to happen as getting the national cesarean delivery rate to fall below 15% from the current level above 30%, or the return of vaginal breech deliveries!
Maternal mortality numbers are even more distressing when you appreciate that 1) African American women have three to four times the death rate from pregnancy complications that white women do and 2) minorities (African Americans, Latinos) are having the majority of pregnancies.

In a society supplied with well-trained obstetricians, marked improvements in the safety of anesthesia, and a plethora of maternal-fetal medicine specialists, we must do some serious introspection and ask ourselves: Why are so many young women dying? And what can we do to prevent their deaths?
Why the rise in maternal deaths?
Multiple reasons are cited for the rise in maternal mortality. They include:
- difficult access to care
- racial discrimination
- increase in substance abuse
- barriers to communication with non–English-speaking patients
- lack of health insurance
- inflexible appointment hours
- lack of transportation to visits
- serious comorbidities (e.g., diabetes).
Each of these has some validity. But my opinion (and many of you will disagree with me) is that our society has not addressed the real issues driving the problem—issues that are difficult to face:
First, some women have too many pregnancies.
Second, certain women who have serious medical conditions, including morbid obesity and substance abuse, are having pregnancies when they should not have any at all.
It is an interesting observation that a person needs a license to drive a car or hunt deer but only a simple act of intercourse with no contraception to become pregnant.
What I’ve learned from my work
I have spent more than 35 years caring for pregnant women of low socioeconomic status. I am proud to have played that role. The two constant themes in my work with these patients are that 1) they feel a lack of control over most aspects of their life and 2) they lack self-esteem.
Pregnancy, however, is the one thing that they can control. It is the one time when others become interested in their welfare, and their best opportunity to have a something (a baby) that is all theirs.
But their adherence to appointments and laboratory testing is often poor. And their use of contraception between pregnancies is sporadic.
The 18-year-old woman who is in her second pregnancy often has a mother who is in her early 30s and a grandmother around 50. For these women, and for their children and grandchildren, the cycle of poverty is repeated endlessly. No matter how much money or effort is put into providing resources to care for them, little has changed in the past 30 years, and there is, I believe, little hope for change in the future. The only solutions that I can discern that will decisively break the cycle of poverty, lower the rate of maternal mortality, and improve the well being of women and their newborns are education and contraception.
My three-pronged proposal
Several actions can improve the outcome of a pregnancy for mothers of low socioeconomic status and their babies. I understand that many people will find these actions coercive and prejudicial, which truly they are not.
- All forms of contraception should be free for women, and access to contraception should be easy. Such a policy would result in fewer unintended pregnancies and abortions and better pregnancy spacing. The expense of free and universal contraception offers a great return on investment compared to the cost of care for a premature baby who spends any length of time in the neonatal intensive care unit.
Young women should be compensated for using whatever contraceptive they choose by being given vouchers that can be redeemed for material goods at select venues or free minutes for their cellular phones.
- All women who qualify for Medicaid coverage of pregnancy should be aggressively encouraged to have a preconception visit at least 3 to 6 months before they plan to become pregnant to assess any risks to themselves or their fetus and have a program put in place to maximize outcomes. This encouragement can take place at any visit in which contraception is discussed.
At this visit, an attempt can be made to optimize their clinical condition by:
- counseling them about drug and alcohol use and smoking
- assessing their risk of genetic disorders
- initiating folic acid and dietary modifications
- performing appropriate screening tests (i.e., blood glucose).
Appropriate consideration should be given, and discussion held, during the visit about whether the patient should even consider becoming pregnant. She should be given a realistic assessment of the risk of pregnancy and childbirth to her and the baby—including the potential for death.
The preconception assessment should be conducted by a physician, a midwife, or a nurse practitioner who has not been involved in the care of the patient. Doing so will minimize the introduction of any bias into the conversation by a treating physician.
When it is in a woman’s medical best interest not to conceive, she should become eligible for expedited adoption and be compensated for each reproductive year in which she does not conceive.
Last, a strong financial incentive should be offered to women who complete this preconception evaluation.
Many will say that such a program is unfair and prejudicial to women of lower socioeconomic status. But a precedent exists: Medicaid prohibits the performance of a sterilization procedure unless a signed permit has been in place 30 days or longer.
- Pregnant women who adhere to a prenatal care plan should be compensated with vouchers that can be redeemed for baby items, maternity clothes, or food for the family at select venues. They should also be compensated for keeping prenatal appointments; obtaining timely laboratory tests; attending prenatal classes; avoiding drugs, alcohol and smoking; returning for postpartum assessment; and using reliable contraception.
Education + contraception = fewer deaths?
Would such a plan work? I am convinced that it is worth trying. What do you think? Send your comments to me at [email protected]!
Reference
1. Traffic safety facts. Early estimate of motor vehicle traffic fatalities in 2009. National Highway Traffic Safety Administration.http://www-nrd.nhtsa.dot.gov/Pubs/811291.PDF. March 2010.
A recent report1 by the National Highway Traffic Safety Administration revealed that deaths from automobile accidents have declined strikingly since 1954, reaching the lowest level ever recorded today: 1.16 fatalities for every 100 million vehicle-miles traveled. Why this improvement? Reasons include an increase in seat belt use and a lower rate of drunk driving, both secondary effects of better law enforcement, safer roads, and people just driving less.
There are lessons to be learned from this success that can be applied to obstetrical care and, potentially, to lower the unacceptably high rate of maternal mortality in this country.
Things aren’t working as we need them to
Truly, it is a reflection of a broken health care system that we, as health care providers, cannot say that we have seen a dramatic decrease in maternal mortality. In 2006, the latest year for which statistics are available, maternal mortality in the United States was at the highest level recorded since 1991: 13.3 deaths for every 100,000 live births (in California, the rate is 16.9 for every 100,000!).
Remember the Healthy People 2010 benchmark of 4.3 maternal deaths for every 100,000 live births? Clearly, we will not see that number any time in the near future; achieving it is as likely to happen as getting the national cesarean delivery rate to fall below 15% from the current level above 30%, or the return of vaginal breech deliveries!
Maternal mortality numbers are even more distressing when you appreciate that 1) African American women have three to four times the death rate from pregnancy complications that white women do and 2) minorities (African Americans, Latinos) are having the majority of pregnancies.

In a society supplied with well-trained obstetricians, marked improvements in the safety of anesthesia, and a plethora of maternal-fetal medicine specialists, we must do some serious introspection and ask ourselves: Why are so many young women dying? And what can we do to prevent their deaths?
Why the rise in maternal deaths?
Multiple reasons are cited for the rise in maternal mortality. They include:
- difficult access to care
- racial discrimination
- increase in substance abuse
- barriers to communication with non–English-speaking patients
- lack of health insurance
- inflexible appointment hours
- lack of transportation to visits
- serious comorbidities (e.g., diabetes).
Each of these has some validity. But my opinion (and many of you will disagree with me) is that our society has not addressed the real issues driving the problem—issues that are difficult to face:
First, some women have too many pregnancies.
Second, certain women who have serious medical conditions, including morbid obesity and substance abuse, are having pregnancies when they should not have any at all.
It is an interesting observation that a person needs a license to drive a car or hunt deer but only a simple act of intercourse with no contraception to become pregnant.
What I’ve learned from my work
I have spent more than 35 years caring for pregnant women of low socioeconomic status. I am proud to have played that role. The two constant themes in my work with these patients are that 1) they feel a lack of control over most aspects of their life and 2) they lack self-esteem.
Pregnancy, however, is the one thing that they can control. It is the one time when others become interested in their welfare, and their best opportunity to have a something (a baby) that is all theirs.
But their adherence to appointments and laboratory testing is often poor. And their use of contraception between pregnancies is sporadic.
The 18-year-old woman who is in her second pregnancy often has a mother who is in her early 30s and a grandmother around 50. For these women, and for their children and grandchildren, the cycle of poverty is repeated endlessly. No matter how much money or effort is put into providing resources to care for them, little has changed in the past 30 years, and there is, I believe, little hope for change in the future. The only solutions that I can discern that will decisively break the cycle of poverty, lower the rate of maternal mortality, and improve the well being of women and their newborns are education and contraception.
My three-pronged proposal
Several actions can improve the outcome of a pregnancy for mothers of low socioeconomic status and their babies. I understand that many people will find these actions coercive and prejudicial, which truly they are not.
- All forms of contraception should be free for women, and access to contraception should be easy. Such a policy would result in fewer unintended pregnancies and abortions and better pregnancy spacing. The expense of free and universal contraception offers a great return on investment compared to the cost of care for a premature baby who spends any length of time in the neonatal intensive care unit.
Young women should be compensated for using whatever contraceptive they choose by being given vouchers that can be redeemed for material goods at select venues or free minutes for their cellular phones.
- All women who qualify for Medicaid coverage of pregnancy should be aggressively encouraged to have a preconception visit at least 3 to 6 months before they plan to become pregnant to assess any risks to themselves or their fetus and have a program put in place to maximize outcomes. This encouragement can take place at any visit in which contraception is discussed.
At this visit, an attempt can be made to optimize their clinical condition by:
- counseling them about drug and alcohol use and smoking
- assessing their risk of genetic disorders
- initiating folic acid and dietary modifications
- performing appropriate screening tests (i.e., blood glucose).
Appropriate consideration should be given, and discussion held, during the visit about whether the patient should even consider becoming pregnant. She should be given a realistic assessment of the risk of pregnancy and childbirth to her and the baby—including the potential for death.
The preconception assessment should be conducted by a physician, a midwife, or a nurse practitioner who has not been involved in the care of the patient. Doing so will minimize the introduction of any bias into the conversation by a treating physician.
When it is in a woman’s medical best interest not to conceive, she should become eligible for expedited adoption and be compensated for each reproductive year in which she does not conceive.
Last, a strong financial incentive should be offered to women who complete this preconception evaluation.
Many will say that such a program is unfair and prejudicial to women of lower socioeconomic status. But a precedent exists: Medicaid prohibits the performance of a sterilization procedure unless a signed permit has been in place 30 days or longer.
- Pregnant women who adhere to a prenatal care plan should be compensated with vouchers that can be redeemed for baby items, maternity clothes, or food for the family at select venues. They should also be compensated for keeping prenatal appointments; obtaining timely laboratory tests; attending prenatal classes; avoiding drugs, alcohol and smoking; returning for postpartum assessment; and using reliable contraception.
Education + contraception = fewer deaths?
Would such a plan work? I am convinced that it is worth trying. What do you think? Send your comments to me at [email protected]!
A recent report1 by the National Highway Traffic Safety Administration revealed that deaths from automobile accidents have declined strikingly since 1954, reaching the lowest level ever recorded today: 1.16 fatalities for every 100 million vehicle-miles traveled. Why this improvement? Reasons include an increase in seat belt use and a lower rate of drunk driving, both secondary effects of better law enforcement, safer roads, and people just driving less.
There are lessons to be learned from this success that can be applied to obstetrical care and, potentially, to lower the unacceptably high rate of maternal mortality in this country.
Things aren’t working as we need them to
Truly, it is a reflection of a broken health care system that we, as health care providers, cannot say that we have seen a dramatic decrease in maternal mortality. In 2006, the latest year for which statistics are available, maternal mortality in the United States was at the highest level recorded since 1991: 13.3 deaths for every 100,000 live births (in California, the rate is 16.9 for every 100,000!).
Remember the Healthy People 2010 benchmark of 4.3 maternal deaths for every 100,000 live births? Clearly, we will not see that number any time in the near future; achieving it is as likely to happen as getting the national cesarean delivery rate to fall below 15% from the current level above 30%, or the return of vaginal breech deliveries!
Maternal mortality numbers are even more distressing when you appreciate that 1) African American women have three to four times the death rate from pregnancy complications that white women do and 2) minorities (African Americans, Latinos) are having the majority of pregnancies.

In a society supplied with well-trained obstetricians, marked improvements in the safety of anesthesia, and a plethora of maternal-fetal medicine specialists, we must do some serious introspection and ask ourselves: Why are so many young women dying? And what can we do to prevent their deaths?
Why the rise in maternal deaths?
Multiple reasons are cited for the rise in maternal mortality. They include:
- difficult access to care
- racial discrimination
- increase in substance abuse
- barriers to communication with non–English-speaking patients
- lack of health insurance
- inflexible appointment hours
- lack of transportation to visits
- serious comorbidities (e.g., diabetes).
Each of these has some validity. But my opinion (and many of you will disagree with me) is that our society has not addressed the real issues driving the problem—issues that are difficult to face:
First, some women have too many pregnancies.
Second, certain women who have serious medical conditions, including morbid obesity and substance abuse, are having pregnancies when they should not have any at all.
It is an interesting observation that a person needs a license to drive a car or hunt deer but only a simple act of intercourse with no contraception to become pregnant.
What I’ve learned from my work
I have spent more than 35 years caring for pregnant women of low socioeconomic status. I am proud to have played that role. The two constant themes in my work with these patients are that 1) they feel a lack of control over most aspects of their life and 2) they lack self-esteem.
Pregnancy, however, is the one thing that they can control. It is the one time when others become interested in their welfare, and their best opportunity to have a something (a baby) that is all theirs.
But their adherence to appointments and laboratory testing is often poor. And their use of contraception between pregnancies is sporadic.
The 18-year-old woman who is in her second pregnancy often has a mother who is in her early 30s and a grandmother around 50. For these women, and for their children and grandchildren, the cycle of poverty is repeated endlessly. No matter how much money or effort is put into providing resources to care for them, little has changed in the past 30 years, and there is, I believe, little hope for change in the future. The only solutions that I can discern that will decisively break the cycle of poverty, lower the rate of maternal mortality, and improve the well being of women and their newborns are education and contraception.
My three-pronged proposal
Several actions can improve the outcome of a pregnancy for mothers of low socioeconomic status and their babies. I understand that many people will find these actions coercive and prejudicial, which truly they are not.
- All forms of contraception should be free for women, and access to contraception should be easy. Such a policy would result in fewer unintended pregnancies and abortions and better pregnancy spacing. The expense of free and universal contraception offers a great return on investment compared to the cost of care for a premature baby who spends any length of time in the neonatal intensive care unit.
Young women should be compensated for using whatever contraceptive they choose by being given vouchers that can be redeemed for material goods at select venues or free minutes for their cellular phones.
- All women who qualify for Medicaid coverage of pregnancy should be aggressively encouraged to have a preconception visit at least 3 to 6 months before they plan to become pregnant to assess any risks to themselves or their fetus and have a program put in place to maximize outcomes. This encouragement can take place at any visit in which contraception is discussed.
At this visit, an attempt can be made to optimize their clinical condition by:
- counseling them about drug and alcohol use and smoking
- assessing their risk of genetic disorders
- initiating folic acid and dietary modifications
- performing appropriate screening tests (i.e., blood glucose).
Appropriate consideration should be given, and discussion held, during the visit about whether the patient should even consider becoming pregnant. She should be given a realistic assessment of the risk of pregnancy and childbirth to her and the baby—including the potential for death.
The preconception assessment should be conducted by a physician, a midwife, or a nurse practitioner who has not been involved in the care of the patient. Doing so will minimize the introduction of any bias into the conversation by a treating physician.
When it is in a woman’s medical best interest not to conceive, she should become eligible for expedited adoption and be compensated for each reproductive year in which she does not conceive.
Last, a strong financial incentive should be offered to women who complete this preconception evaluation.
Many will say that such a program is unfair and prejudicial to women of lower socioeconomic status. But a precedent exists: Medicaid prohibits the performance of a sterilization procedure unless a signed permit has been in place 30 days or longer.
- Pregnant women who adhere to a prenatal care plan should be compensated with vouchers that can be redeemed for baby items, maternity clothes, or food for the family at select venues. They should also be compensated for keeping prenatal appointments; obtaining timely laboratory tests; attending prenatal classes; avoiding drugs, alcohol and smoking; returning for postpartum assessment; and using reliable contraception.
Education + contraception = fewer deaths?
Would such a plan work? I am convinced that it is worth trying. What do you think? Send your comments to me at [email protected]!
Reference
1. Traffic safety facts. Early estimate of motor vehicle traffic fatalities in 2009. National Highway Traffic Safety Administration.http://www-nrd.nhtsa.dot.gov/Pubs/811291.PDF. March 2010.
Reference
1. Traffic safety facts. Early estimate of motor vehicle traffic fatalities in 2009. National Highway Traffic Safety Administration.http://www-nrd.nhtsa.dot.gov/Pubs/811291.PDF. March 2010.
Continuity Conundrum
Editor’s note: Third of a three-part series.
In the two monthly columns preceding this one, I’ve provided an overview of some ways hospitalist groups distribute new referrals among the providers. This month, I’ll review things that cause some groups to make exceptions to their typical method of distributing patients, and turn from how patients are distributed over 24 hours to thoughts about how they might be assigned over the course of consecutive days worked by a doctor.
Equitable Exceptions
There are a number of reasons groups decide to depart from their typical method of assigning patients. These include:
- “Bouncebacks”;
- One hospitalist is at the cap, others aren’t;
- Consult requested of a specific hospitalist;
- Hospitalists with unique skills (e.g., ICU expertise); and
- A patient “fires” the hospitalist.
There isn’t a standard “hospitalist way” of dealing with these issues, and each group will need to work out its own system. The most common of these issues is “bouncebacks.” Every group should try to have patients readmitted within three or four days of discharge go back to the discharging hospitalist. However, this proves difficult in many cases for several reasons, most commonly because the original discharging doctor might not be working when the patient returns.
The Alpha & Omega
Nearly every hospitalist practice makes some effort to maximize continuity between a single hospitalist and patient over the course of a hospital stay. But the effect of the method of patient assignment on continuity often is overlooked.
A reasonable way to think about or measure continuity is to estimate the portion of patients seen by the group that see the same hospitalist for each daytime visit over the course of their stay. (Assume that in most HM groups the same hospitalist can’t make both day and night visits over the course of the hospital stay. So, just for simplicity, I’ve intentionally left night visits, including an initial admission visit at night, out of the continuity calculation.) Plug the numbers for your practice into the formula (see Figure 1, right) and see what you get.
If a hospitalist always works seven consecutive day shifts (e.g., a seven-on/seven-off schedule) and the hospitalist’s patients have an average LOS of 4.2 days, then 54% of patients will see the same hospitalist for all daytime visits, and 46% will experience at least one handoff. (To keep things simple, I’m ignoring the effect on continuity of patients being admitted by an “admitter” or nocturnist who doesn’t see the patient subsequently.)
Changing the number of consecutive day shifts a hospitalist works has the most significant impact on continuity, but just how many consecutive days can one work routinely before fatigue and burnout—not too mention increased errors and decreased patient satisfaction—become a problem? (Many hospitalists make the mistake of trying to stuff what might be a reasonable annual workload into the smallest number of shifts possible with the goal of maximizing the number of days off. That means each worked day will be very busy, making it really hard to work many consecutive days. But you always have the option of titrating out that same annual workload over more days so that each day is less busy and it becomes easier to work more consecutive days.)
An often-overlooked way to improve continuity without having to work more consecutive day shifts is to have a hospitalist who is early in their series of worked days take on more new admissions and consults, and perhaps exempt that doctor from taking on new referrals for the last day or two he or she is on service. Eric Howell, MD, FHM, an SHM board member, calls this method “slam and dwindle.” This has been the approach I’ve experienced my whole career, and it is hard for me to imagine doing it any other way.
Here’s how it might work: Let’s say Dr. Petty always works seven consecutive day shifts, and on the first day he picks up a list of patients remaining from the doctor he’s replacing. To keep things simple, let’s assume he’s not in a large group, and during his first day of seven days on service he accepts and “keeps” all new referrals to the practice. On each successive day, he might assume the care of some new patients, but none on days six and seven. This means he takes on a disproportionately large number of new referrals at the beginning of his consecutive worked days, or “front-loads” new referrals. And because many of these patients will discharge before the end of his seven days and he takes on no new patients on days six and seven, his census will drop a lot before he rotates off, which in turn means there will be few patients who will have to get to know a new doctor on the first day Dr. Petty starts his seven-off schedule.
This system of patient distribution means continuity improved without requiring Dr. Petty to work more consecutive day shifts. Even though he works seven consecutive days and his average (or median) LOS is 4.2, as in the example above, his continuity will be much better than 54%. In fact, as many as 70% to 80% of Dr. Petty’s patients will see him for every daytime visit during their stay.
Other benefits of assigning more patients early and none late in a series of worked days are that on his last day of service, he will have more time to “tee up” patients for the next doctor, including preparing for patients anticipated to discharge the next day (e.g., dictate discharge summary, complete paperwork, etc.), and might be able to wrap up a little earlier that day. And when rotating back on service, he will pick up a small list of patients left by Dr. Tench, maybe fewer than eight, rather than the group’s average daily load of 15 patients per doctor, so he will have the capacity to admit a lot of patients that day.
I think there are three main reasons this isn’t a more common approach:
- Many HM groups just haven’t considered it.
- HM groups might have a schedule that has all doctors rotate off/on the same days each week. For example, all doctors rotate off on Tuesdays and are replaced by new doctors on Wednesday. That makes it impossible to exempt a doctor from taking on new referrals on the last day of service because all of the group’s doctors have their last day on Tuesday. These groups could stagger the day each doctor rotates off—one on Monday, one on Tuesday, and so on.
- Every doctor is so busy each day that it wouldn’t be feasible to exempt any individual doctor from taking on new patients, even if they are off the next day.
Despite the difficulties implementing a system of front-loading new referrals, I think most hospitalists would find that they like it. Because it reduces handoffs, it reduces, at least modestly, the group’s overall workload and probably benefits the group’s quality and patient satisfaction. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Editor’s note: Third of a three-part series.
In the two monthly columns preceding this one, I’ve provided an overview of some ways hospitalist groups distribute new referrals among the providers. This month, I’ll review things that cause some groups to make exceptions to their typical method of distributing patients, and turn from how patients are distributed over 24 hours to thoughts about how they might be assigned over the course of consecutive days worked by a doctor.
Equitable Exceptions
There are a number of reasons groups decide to depart from their typical method of assigning patients. These include:
- “Bouncebacks”;
- One hospitalist is at the cap, others aren’t;
- Consult requested of a specific hospitalist;
- Hospitalists with unique skills (e.g., ICU expertise); and
- A patient “fires” the hospitalist.
There isn’t a standard “hospitalist way” of dealing with these issues, and each group will need to work out its own system. The most common of these issues is “bouncebacks.” Every group should try to have patients readmitted within three or four days of discharge go back to the discharging hospitalist. However, this proves difficult in many cases for several reasons, most commonly because the original discharging doctor might not be working when the patient returns.
The Alpha & Omega
Nearly every hospitalist practice makes some effort to maximize continuity between a single hospitalist and patient over the course of a hospital stay. But the effect of the method of patient assignment on continuity often is overlooked.
A reasonable way to think about or measure continuity is to estimate the portion of patients seen by the group that see the same hospitalist for each daytime visit over the course of their stay. (Assume that in most HM groups the same hospitalist can’t make both day and night visits over the course of the hospital stay. So, just for simplicity, I’ve intentionally left night visits, including an initial admission visit at night, out of the continuity calculation.) Plug the numbers for your practice into the formula (see Figure 1, right) and see what you get.
If a hospitalist always works seven consecutive day shifts (e.g., a seven-on/seven-off schedule) and the hospitalist’s patients have an average LOS of 4.2 days, then 54% of patients will see the same hospitalist for all daytime visits, and 46% will experience at least one handoff. (To keep things simple, I’m ignoring the effect on continuity of patients being admitted by an “admitter” or nocturnist who doesn’t see the patient subsequently.)
Changing the number of consecutive day shifts a hospitalist works has the most significant impact on continuity, but just how many consecutive days can one work routinely before fatigue and burnout—not too mention increased errors and decreased patient satisfaction—become a problem? (Many hospitalists make the mistake of trying to stuff what might be a reasonable annual workload into the smallest number of shifts possible with the goal of maximizing the number of days off. That means each worked day will be very busy, making it really hard to work many consecutive days. But you always have the option of titrating out that same annual workload over more days so that each day is less busy and it becomes easier to work more consecutive days.)
An often-overlooked way to improve continuity without having to work more consecutive day shifts is to have a hospitalist who is early in their series of worked days take on more new admissions and consults, and perhaps exempt that doctor from taking on new referrals for the last day or two he or she is on service. Eric Howell, MD, FHM, an SHM board member, calls this method “slam and dwindle.” This has been the approach I’ve experienced my whole career, and it is hard for me to imagine doing it any other way.
Here’s how it might work: Let’s say Dr. Petty always works seven consecutive day shifts, and on the first day he picks up a list of patients remaining from the doctor he’s replacing. To keep things simple, let’s assume he’s not in a large group, and during his first day of seven days on service he accepts and “keeps” all new referrals to the practice. On each successive day, he might assume the care of some new patients, but none on days six and seven. This means he takes on a disproportionately large number of new referrals at the beginning of his consecutive worked days, or “front-loads” new referrals. And because many of these patients will discharge before the end of his seven days and he takes on no new patients on days six and seven, his census will drop a lot before he rotates off, which in turn means there will be few patients who will have to get to know a new doctor on the first day Dr. Petty starts his seven-off schedule.
This system of patient distribution means continuity improved without requiring Dr. Petty to work more consecutive day shifts. Even though he works seven consecutive days and his average (or median) LOS is 4.2, as in the example above, his continuity will be much better than 54%. In fact, as many as 70% to 80% of Dr. Petty’s patients will see him for every daytime visit during their stay.
Other benefits of assigning more patients early and none late in a series of worked days are that on his last day of service, he will have more time to “tee up” patients for the next doctor, including preparing for patients anticipated to discharge the next day (e.g., dictate discharge summary, complete paperwork, etc.), and might be able to wrap up a little earlier that day. And when rotating back on service, he will pick up a small list of patients left by Dr. Tench, maybe fewer than eight, rather than the group’s average daily load of 15 patients per doctor, so he will have the capacity to admit a lot of patients that day.
I think there are three main reasons this isn’t a more common approach:
- Many HM groups just haven’t considered it.
- HM groups might have a schedule that has all doctors rotate off/on the same days each week. For example, all doctors rotate off on Tuesdays and are replaced by new doctors on Wednesday. That makes it impossible to exempt a doctor from taking on new referrals on the last day of service because all of the group’s doctors have their last day on Tuesday. These groups could stagger the day each doctor rotates off—one on Monday, one on Tuesday, and so on.
- Every doctor is so busy each day that it wouldn’t be feasible to exempt any individual doctor from taking on new patients, even if they are off the next day.
Despite the difficulties implementing a system of front-loading new referrals, I think most hospitalists would find that they like it. Because it reduces handoffs, it reduces, at least modestly, the group’s overall workload and probably benefits the group’s quality and patient satisfaction. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Editor’s note: Third of a three-part series.
In the two monthly columns preceding this one, I’ve provided an overview of some ways hospitalist groups distribute new referrals among the providers. This month, I’ll review things that cause some groups to make exceptions to their typical method of distributing patients, and turn from how patients are distributed over 24 hours to thoughts about how they might be assigned over the course of consecutive days worked by a doctor.
Equitable Exceptions
There are a number of reasons groups decide to depart from their typical method of assigning patients. These include:
- “Bouncebacks”;
- One hospitalist is at the cap, others aren’t;
- Consult requested of a specific hospitalist;
- Hospitalists with unique skills (e.g., ICU expertise); and
- A patient “fires” the hospitalist.
There isn’t a standard “hospitalist way” of dealing with these issues, and each group will need to work out its own system. The most common of these issues is “bouncebacks.” Every group should try to have patients readmitted within three or four days of discharge go back to the discharging hospitalist. However, this proves difficult in many cases for several reasons, most commonly because the original discharging doctor might not be working when the patient returns.
The Alpha & Omega
Nearly every hospitalist practice makes some effort to maximize continuity between a single hospitalist and patient over the course of a hospital stay. But the effect of the method of patient assignment on continuity often is overlooked.
A reasonable way to think about or measure continuity is to estimate the portion of patients seen by the group that see the same hospitalist for each daytime visit over the course of their stay. (Assume that in most HM groups the same hospitalist can’t make both day and night visits over the course of the hospital stay. So, just for simplicity, I’ve intentionally left night visits, including an initial admission visit at night, out of the continuity calculation.) Plug the numbers for your practice into the formula (see Figure 1, right) and see what you get.
If a hospitalist always works seven consecutive day shifts (e.g., a seven-on/seven-off schedule) and the hospitalist’s patients have an average LOS of 4.2 days, then 54% of patients will see the same hospitalist for all daytime visits, and 46% will experience at least one handoff. (To keep things simple, I’m ignoring the effect on continuity of patients being admitted by an “admitter” or nocturnist who doesn’t see the patient subsequently.)
Changing the number of consecutive day shifts a hospitalist works has the most significant impact on continuity, but just how many consecutive days can one work routinely before fatigue and burnout—not too mention increased errors and decreased patient satisfaction—become a problem? (Many hospitalists make the mistake of trying to stuff what might be a reasonable annual workload into the smallest number of shifts possible with the goal of maximizing the number of days off. That means each worked day will be very busy, making it really hard to work many consecutive days. But you always have the option of titrating out that same annual workload over more days so that each day is less busy and it becomes easier to work more consecutive days.)
An often-overlooked way to improve continuity without having to work more consecutive day shifts is to have a hospitalist who is early in their series of worked days take on more new admissions and consults, and perhaps exempt that doctor from taking on new referrals for the last day or two he or she is on service. Eric Howell, MD, FHM, an SHM board member, calls this method “slam and dwindle.” This has been the approach I’ve experienced my whole career, and it is hard for me to imagine doing it any other way.
Here’s how it might work: Let’s say Dr. Petty always works seven consecutive day shifts, and on the first day he picks up a list of patients remaining from the doctor he’s replacing. To keep things simple, let’s assume he’s not in a large group, and during his first day of seven days on service he accepts and “keeps” all new referrals to the practice. On each successive day, he might assume the care of some new patients, but none on days six and seven. This means he takes on a disproportionately large number of new referrals at the beginning of his consecutive worked days, or “front-loads” new referrals. And because many of these patients will discharge before the end of his seven days and he takes on no new patients on days six and seven, his census will drop a lot before he rotates off, which in turn means there will be few patients who will have to get to know a new doctor on the first day Dr. Petty starts his seven-off schedule.
This system of patient distribution means continuity improved without requiring Dr. Petty to work more consecutive day shifts. Even though he works seven consecutive days and his average (or median) LOS is 4.2, as in the example above, his continuity will be much better than 54%. In fact, as many as 70% to 80% of Dr. Petty’s patients will see him for every daytime visit during their stay.
Other benefits of assigning more patients early and none late in a series of worked days are that on his last day of service, he will have more time to “tee up” patients for the next doctor, including preparing for patients anticipated to discharge the next day (e.g., dictate discharge summary, complete paperwork, etc.), and might be able to wrap up a little earlier that day. And when rotating back on service, he will pick up a small list of patients left by Dr. Tench, maybe fewer than eight, rather than the group’s average daily load of 15 patients per doctor, so he will have the capacity to admit a lot of patients that day.
I think there are three main reasons this isn’t a more common approach:
- Many HM groups just haven’t considered it.
- HM groups might have a schedule that has all doctors rotate off/on the same days each week. For example, all doctors rotate off on Tuesdays and are replaced by new doctors on Wednesday. That makes it impossible to exempt a doctor from taking on new referrals on the last day of service because all of the group’s doctors have their last day on Tuesday. These groups could stagger the day each doctor rotates off—one on Monday, one on Tuesday, and so on.
- Every doctor is so busy each day that it wouldn’t be feasible to exempt any individual doctor from taking on new patients, even if they are off the next day.
Despite the difficulties implementing a system of front-loading new referrals, I think most hospitalists would find that they like it. Because it reduces handoffs, it reduces, at least modestly, the group’s overall workload and probably benefits the group’s quality and patient satisfaction. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Do certain SSRIs reduce the benefits of tamoxifen in breast cancer survivors?
Tamoxifen is widely used by women who have receptor-positive breast cancer to lower their risk of recurrence. Tamoxifen is a prodrug converted by the hepatic cytochrome P450 enzyme system (particularly isoenzyme CYP2D6) to active metabolites, which have much higher affinity for the estrogen receptor than the parent prodrug has. Selective serotonin reuptake inhibitors (SSRIs) inhibit CYP2D6 to variable degrees, with paroxetine thought to be the most potent inhibitor.
The fact that SSRIs inhibit CYP2D6 is important because as many as one in every four women who have breast cancer experiences depression as well.1 SSRIs are widely prescribed for these patients—not only as treatment for their depressive disorder but also because the drugs have been shown to offer some relief from hot flashes.
In this cohort study from Ontario, where health care is delivered in a centralized fashion, Kelly and colleagues assessed the survival of breast cancer survivors 66 years and older who were treated with both tamoxifen and an SSRI, or with both tamoxifen and venlafaxine (which inhibits reuptake of both serotonin and norepinephrine).
The median age of the 2,430 eligible and evaluable women at the time of tamoxifen initiation was 74 years. These women began using tamoxifen between 1993 and 2005, with a median duration of use of 4 years. Paroxetine was the most commonly used SSRI in this study (25.9%), followed by sertraline (Zoloft, 22.3%), citalopram (Celexa, 19.2%), venlafaxine (Effexor, 15.0%), fluoxetine (Prozac, Sarafem, 10.4%), and fluvoxamine (Luvox, 7.2%).
Neither duloxetine (Cymbalta) nor escitalopram (Lexapro) were included in this analysis.
By the end of follow-up, 1,074 women (44.2%) in the cohort had died, 374 of them from breast cancer.
When taken with tamoxifen, paroxetine increased breast cancer mortality
Concomitant use of paroxetine and tamoxifen was associated with a significantly increased risk of death from breast cancer. In addition, as the proportion of concomitant use of the drugs increased to 25%, 50%, and 75% of total tamoxifen use, the adjusted risk of death increased 24%, 54%, and 91%, respectively. Among women taking tamoxifen, concomitant use of other SSRIs or venlafaxine was not associated with an increased risk of death.
Based on their data, the investigators determined that use of paroxetine during 41% of tamoxifen treatment (the median overlap observed in this study) would result in one additional breast cancer death at 5 years for every 19.7 women treated in this fashion. If paroxetine were used for the entire duration of tamoxifen treatment—an overlap of 100%—an additional death would occur for every 6.9 women treated.
The authors also noted that fluoxetine is a “strong inhibitor” of tamoxifen, although it was not associated with an increased risk of breast cancer death in this trial. One reason may be the small number of women who took tamoxifen and fluoxetine concomitantly in this study.
“Our results should not be viewed as evidence that fluoxetine can be safely used in combination with tamoxifen,” Kelly and coworkers write. “Similarly, we cannot exclude the possibility that insufficient sample size explains the nonsignificant mortality results with other SSRIs.”
Because fluoxetine is known to strongly inhibit CYP2D6, other antidepressants are more prudent choices for tamoxifen users.
In addition, switch any tamoxifen user who is taking paroxetine to an alternative antidepressant. Venlafaxine may be a sound choice in this setting, given the data indicating its efficacy as a nonhormonal treatment for vasomotor symptoms in breast cancer survivors.—ANDREW M. KAUNITZ, MD
Reference
1. Fann JR, Thomas-Rich AM, Katon WJ, et al. Major depression after breast cancer: a review of epidemiology and treatment. Gen Hosp Psychiatry. 2008;30(2):112-126.
Tamoxifen is widely used by women who have receptor-positive breast cancer to lower their risk of recurrence. Tamoxifen is a prodrug converted by the hepatic cytochrome P450 enzyme system (particularly isoenzyme CYP2D6) to active metabolites, which have much higher affinity for the estrogen receptor than the parent prodrug has. Selective serotonin reuptake inhibitors (SSRIs) inhibit CYP2D6 to variable degrees, with paroxetine thought to be the most potent inhibitor.
The fact that SSRIs inhibit CYP2D6 is important because as many as one in every four women who have breast cancer experiences depression as well.1 SSRIs are widely prescribed for these patients—not only as treatment for their depressive disorder but also because the drugs have been shown to offer some relief from hot flashes.
In this cohort study from Ontario, where health care is delivered in a centralized fashion, Kelly and colleagues assessed the survival of breast cancer survivors 66 years and older who were treated with both tamoxifen and an SSRI, or with both tamoxifen and venlafaxine (which inhibits reuptake of both serotonin and norepinephrine).
The median age of the 2,430 eligible and evaluable women at the time of tamoxifen initiation was 74 years. These women began using tamoxifen between 1993 and 2005, with a median duration of use of 4 years. Paroxetine was the most commonly used SSRI in this study (25.9%), followed by sertraline (Zoloft, 22.3%), citalopram (Celexa, 19.2%), venlafaxine (Effexor, 15.0%), fluoxetine (Prozac, Sarafem, 10.4%), and fluvoxamine (Luvox, 7.2%).
Neither duloxetine (Cymbalta) nor escitalopram (Lexapro) were included in this analysis.
By the end of follow-up, 1,074 women (44.2%) in the cohort had died, 374 of them from breast cancer.
When taken with tamoxifen, paroxetine increased breast cancer mortality
Concomitant use of paroxetine and tamoxifen was associated with a significantly increased risk of death from breast cancer. In addition, as the proportion of concomitant use of the drugs increased to 25%, 50%, and 75% of total tamoxifen use, the adjusted risk of death increased 24%, 54%, and 91%, respectively. Among women taking tamoxifen, concomitant use of other SSRIs or venlafaxine was not associated with an increased risk of death.
Based on their data, the investigators determined that use of paroxetine during 41% of tamoxifen treatment (the median overlap observed in this study) would result in one additional breast cancer death at 5 years for every 19.7 women treated in this fashion. If paroxetine were used for the entire duration of tamoxifen treatment—an overlap of 100%—an additional death would occur for every 6.9 women treated.
The authors also noted that fluoxetine is a “strong inhibitor” of tamoxifen, although it was not associated with an increased risk of breast cancer death in this trial. One reason may be the small number of women who took tamoxifen and fluoxetine concomitantly in this study.
“Our results should not be viewed as evidence that fluoxetine can be safely used in combination with tamoxifen,” Kelly and coworkers write. “Similarly, we cannot exclude the possibility that insufficient sample size explains the nonsignificant mortality results with other SSRIs.”
Because fluoxetine is known to strongly inhibit CYP2D6, other antidepressants are more prudent choices for tamoxifen users.
In addition, switch any tamoxifen user who is taking paroxetine to an alternative antidepressant. Venlafaxine may be a sound choice in this setting, given the data indicating its efficacy as a nonhormonal treatment for vasomotor symptoms in breast cancer survivors.—ANDREW M. KAUNITZ, MD
Tamoxifen is widely used by women who have receptor-positive breast cancer to lower their risk of recurrence. Tamoxifen is a prodrug converted by the hepatic cytochrome P450 enzyme system (particularly isoenzyme CYP2D6) to active metabolites, which have much higher affinity for the estrogen receptor than the parent prodrug has. Selective serotonin reuptake inhibitors (SSRIs) inhibit CYP2D6 to variable degrees, with paroxetine thought to be the most potent inhibitor.
The fact that SSRIs inhibit CYP2D6 is important because as many as one in every four women who have breast cancer experiences depression as well.1 SSRIs are widely prescribed for these patients—not only as treatment for their depressive disorder but also because the drugs have been shown to offer some relief from hot flashes.
In this cohort study from Ontario, where health care is delivered in a centralized fashion, Kelly and colleagues assessed the survival of breast cancer survivors 66 years and older who were treated with both tamoxifen and an SSRI, or with both tamoxifen and venlafaxine (which inhibits reuptake of both serotonin and norepinephrine).
The median age of the 2,430 eligible and evaluable women at the time of tamoxifen initiation was 74 years. These women began using tamoxifen between 1993 and 2005, with a median duration of use of 4 years. Paroxetine was the most commonly used SSRI in this study (25.9%), followed by sertraline (Zoloft, 22.3%), citalopram (Celexa, 19.2%), venlafaxine (Effexor, 15.0%), fluoxetine (Prozac, Sarafem, 10.4%), and fluvoxamine (Luvox, 7.2%).
Neither duloxetine (Cymbalta) nor escitalopram (Lexapro) were included in this analysis.
By the end of follow-up, 1,074 women (44.2%) in the cohort had died, 374 of them from breast cancer.
When taken with tamoxifen, paroxetine increased breast cancer mortality
Concomitant use of paroxetine and tamoxifen was associated with a significantly increased risk of death from breast cancer. In addition, as the proportion of concomitant use of the drugs increased to 25%, 50%, and 75% of total tamoxifen use, the adjusted risk of death increased 24%, 54%, and 91%, respectively. Among women taking tamoxifen, concomitant use of other SSRIs or venlafaxine was not associated with an increased risk of death.
Based on their data, the investigators determined that use of paroxetine during 41% of tamoxifen treatment (the median overlap observed in this study) would result in one additional breast cancer death at 5 years for every 19.7 women treated in this fashion. If paroxetine were used for the entire duration of tamoxifen treatment—an overlap of 100%—an additional death would occur for every 6.9 women treated.
The authors also noted that fluoxetine is a “strong inhibitor” of tamoxifen, although it was not associated with an increased risk of breast cancer death in this trial. One reason may be the small number of women who took tamoxifen and fluoxetine concomitantly in this study.
“Our results should not be viewed as evidence that fluoxetine can be safely used in combination with tamoxifen,” Kelly and coworkers write. “Similarly, we cannot exclude the possibility that insufficient sample size explains the nonsignificant mortality results with other SSRIs.”
Because fluoxetine is known to strongly inhibit CYP2D6, other antidepressants are more prudent choices for tamoxifen users.
In addition, switch any tamoxifen user who is taking paroxetine to an alternative antidepressant. Venlafaxine may be a sound choice in this setting, given the data indicating its efficacy as a nonhormonal treatment for vasomotor symptoms in breast cancer survivors.—ANDREW M. KAUNITZ, MD
Reference
1. Fann JR, Thomas-Rich AM, Katon WJ, et al. Major depression after breast cancer: a review of epidemiology and treatment. Gen Hosp Psychiatry. 2008;30(2):112-126.
Reference
1. Fann JR, Thomas-Rich AM, Katon WJ, et al. Major depression after breast cancer: a review of epidemiology and treatment. Gen Hosp Psychiatry. 2008;30(2):112-126.
OBG Management ©2010 Dowden Health Media
New Referral Distribution
Editor’s note: Second of a three-part series.
As I mentioned last month, there isn’t a proven best method to use when distributing new referrals among your group’s providers. The popular methods fall along a continuum of being focused on daily, or continuous, leveling of patient loads between providers (“load leveling”) at one end; at the other end of the continuum is having a doctor be “on” for all new referrals for a predetermined time period, and accepting that patient volumes might be uneven day to day but tend to even out over long periods.
There might not be any reason to change your group’s approach to patient assignment, but you should always be thinking about how your own methods might be changed or improved. I have shared (“Bigger Isn’t Always Better,” June 2009, p. 46) my concern that some groups invest far too much time in a morning load-leveling and handoff conference. Make sure your group is using only as much time as needed.
Air-Traffic Controllers
Many large groups (e.g., more than 15 full-time equivalents) that assign patients to providers in sequence, like dealing a deck of cards, have a designated provider who holds the triage pager and serves as “air-traffic controller.” This person typically takes incoming calls about all new referrals, jots down the relevant clinical data, keeps track of which hospitalist is due to take the next patient, pages that person, and repeats the clinical information. As I’ve written before (“How to Hire and Use Clerical Staff,” June 2007, p. 73), many practices have found that during business hours, they can hand this role to a clerical person who simply takes down the name and phone number of the doctor making the referral, then pages that information to the hospitalist due to get the next patient. The hospitalist then calls and speaks directly with the referring doctor.
Small- to medium-sized groups can eliminate entirely the need for any such “air-traffic control” function if they assign all new referrals to a single doctor for specified periods of time. For example, from 7 a.m. to 3 p.m. today, all new referrals go to Dr. Glass, and from 3 p.m. to 11 p.m., they go to Dr. Cage.
Admitter-Rounder Duties
Many—maybe most?—large groups separate daytime admitter and rounder functions so that on any given day, a hospitalist does one but not both. The principal advantages of this approach are reducing the stress on, and possibly increasing the efficiency of, rounding doctors by shielding them from the unpredictable and time-consuming interruption of needing to admit a new patient. And a daytime doctor who only does admissions might be able to start seeing a patient in the ED more quickly than one who is busy making rounds.
Any increased availability of admitters to the ED could be offset by their lack of surge capacity leading to a bottleneck in ED throughput when there are many patients to admit at the same time and a limited number of admitters (often only one). Such a bottleneck would be much less likely if all daytime doctors (i.e., the rounders) were available to see admissions rather than just admitters.
Continuity of care suffers when a group has separate admitters and rounders, because no patients will be seen by the same doctor on the day of admission and the day following. This method requires a handoff from the day of admission to the next day. Such a handoff might be unavoidable for patients admitted during the night, but this doesn’t have to occur when patients are admitted during the daytime.
Who’s Seeing this Patient?
It seems to make sense to wait until each morning to distribute patients. That allows the practice to know just how many new patients there are, and they can be distributed according to complexity and whether a hospitalist has formed a previous relationship with that patient. But it means that no one at the hospital will know which hospitalist is caring for the patient until later in the morning. For example, if the radiologist is over-reading a study done during the night and finds something worthy of a phone call to the hospitalist, no one is sure who should get the call. A patient might develop hypoglycemia shortly after the hospitalist night shift is over, but the nurse doesn’t know which hospitalist to call.
And, perhaps most importantly, if patients aren’t distributed until the start of the day shift, the night hospitalist can’t tell the patient and family which hospitalist to expect the next morning. To test the significance of this issue, I conducted an experiment while working our group’s late-evening admitter shift. I concluded my visit with each admitted patient by explaining, “I am on-call for our group tonight, so I will be off recovering tomorrow. Therefore, I won’t see you again, but one of my partners will take over in the morning. Do you have any questions for me?” Every patient I admitted had the same question. “What is that doctor’s name?”
How does your group answer a patient who asks which hospitalist will be in the next day? If your method is load-leveling in the morning, then the best answer your night admitting doctor can give is probably to say: “I don’t know which of my partners will be in. There are several working tomorrow, and at the start of the day, they will divide up the patients who come in tonight depending on how busy each of them is. But all the doctors in our group are terrific and will take good care of you.”
I’m told the same thing when I get my hair cut: You’ll get whichever “hair artist” is up next. I put up with it at the hair place because it costs less than $15. But I still find it a little irritating. I’m sure all the barbers aren’t equally skilled or diligent, and I want the best one. (Maybe I shouldn’t care since there isn’t much that can be done with my hair.) I’m pretty sure patients feel the same way about which doctor they get. The public is convinced there is a wide variety in the quality of doctors, and they want a good one. If you have to tell them theirs is being assigned by lottery, they won’t be as happy than if you can provide the name and a little information about the doctor they can expect to see the next day.
When the patients I admit late last evening ask who would see them the next day, I’m glad when I can provide a name and a little more information. I say something like, “I won’t see you after tonight, but my partner, Dr. Shawn Lee, will be instead. That means you’re getting an upgrade! Not only is he a really nice guy, he’s voted one of Seattle’s best doctors every year. He’ll do a great job for you.”
To make this communication effective, the night doctor has to know which hospitalist takes over the next morning and has a list indicating which day doctor will get the first, second, third new patient, and so on, admitted during the night. This is possible if patients are assigned by a predetermined algorithm, or if the day doctors have their load-leveling meeting at the end of each day shift, rather than in the morning, to create a list telling the night doctor which day hospitalist he should admit the first and subsequent patents to. That way, the night doctor can write in the admitting orders at 1 a.m. “admit to Dr. X.” This eliminates confusion on the part of other hospital staff who need to know who to call about a patient after the start of the day shift.
Next month I will look at special circumstances, and some pros and cons of having an individual hospitalist take on the care of more patients at the beginning of consecutive day shifts, and exempting them from taking on new patients on the last day or two before rotating off. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Editor’s note: Second of a three-part series.
As I mentioned last month, there isn’t a proven best method to use when distributing new referrals among your group’s providers. The popular methods fall along a continuum of being focused on daily, or continuous, leveling of patient loads between providers (“load leveling”) at one end; at the other end of the continuum is having a doctor be “on” for all new referrals for a predetermined time period, and accepting that patient volumes might be uneven day to day but tend to even out over long periods.
There might not be any reason to change your group’s approach to patient assignment, but you should always be thinking about how your own methods might be changed or improved. I have shared (“Bigger Isn’t Always Better,” June 2009, p. 46) my concern that some groups invest far too much time in a morning load-leveling and handoff conference. Make sure your group is using only as much time as needed.
Air-Traffic Controllers
Many large groups (e.g., more than 15 full-time equivalents) that assign patients to providers in sequence, like dealing a deck of cards, have a designated provider who holds the triage pager and serves as “air-traffic controller.” This person typically takes incoming calls about all new referrals, jots down the relevant clinical data, keeps track of which hospitalist is due to take the next patient, pages that person, and repeats the clinical information. As I’ve written before (“How to Hire and Use Clerical Staff,” June 2007, p. 73), many practices have found that during business hours, they can hand this role to a clerical person who simply takes down the name and phone number of the doctor making the referral, then pages that information to the hospitalist due to get the next patient. The hospitalist then calls and speaks directly with the referring doctor.
Small- to medium-sized groups can eliminate entirely the need for any such “air-traffic control” function if they assign all new referrals to a single doctor for specified periods of time. For example, from 7 a.m. to 3 p.m. today, all new referrals go to Dr. Glass, and from 3 p.m. to 11 p.m., they go to Dr. Cage.
Admitter-Rounder Duties
Many—maybe most?—large groups separate daytime admitter and rounder functions so that on any given day, a hospitalist does one but not both. The principal advantages of this approach are reducing the stress on, and possibly increasing the efficiency of, rounding doctors by shielding them from the unpredictable and time-consuming interruption of needing to admit a new patient. And a daytime doctor who only does admissions might be able to start seeing a patient in the ED more quickly than one who is busy making rounds.
Any increased availability of admitters to the ED could be offset by their lack of surge capacity leading to a bottleneck in ED throughput when there are many patients to admit at the same time and a limited number of admitters (often only one). Such a bottleneck would be much less likely if all daytime doctors (i.e., the rounders) were available to see admissions rather than just admitters.
Continuity of care suffers when a group has separate admitters and rounders, because no patients will be seen by the same doctor on the day of admission and the day following. This method requires a handoff from the day of admission to the next day. Such a handoff might be unavoidable for patients admitted during the night, but this doesn’t have to occur when patients are admitted during the daytime.
Who’s Seeing this Patient?
It seems to make sense to wait until each morning to distribute patients. That allows the practice to know just how many new patients there are, and they can be distributed according to complexity and whether a hospitalist has formed a previous relationship with that patient. But it means that no one at the hospital will know which hospitalist is caring for the patient until later in the morning. For example, if the radiologist is over-reading a study done during the night and finds something worthy of a phone call to the hospitalist, no one is sure who should get the call. A patient might develop hypoglycemia shortly after the hospitalist night shift is over, but the nurse doesn’t know which hospitalist to call.
And, perhaps most importantly, if patients aren’t distributed until the start of the day shift, the night hospitalist can’t tell the patient and family which hospitalist to expect the next morning. To test the significance of this issue, I conducted an experiment while working our group’s late-evening admitter shift. I concluded my visit with each admitted patient by explaining, “I am on-call for our group tonight, so I will be off recovering tomorrow. Therefore, I won’t see you again, but one of my partners will take over in the morning. Do you have any questions for me?” Every patient I admitted had the same question. “What is that doctor’s name?”
How does your group answer a patient who asks which hospitalist will be in the next day? If your method is load-leveling in the morning, then the best answer your night admitting doctor can give is probably to say: “I don’t know which of my partners will be in. There are several working tomorrow, and at the start of the day, they will divide up the patients who come in tonight depending on how busy each of them is. But all the doctors in our group are terrific and will take good care of you.”
I’m told the same thing when I get my hair cut: You’ll get whichever “hair artist” is up next. I put up with it at the hair place because it costs less than $15. But I still find it a little irritating. I’m sure all the barbers aren’t equally skilled or diligent, and I want the best one. (Maybe I shouldn’t care since there isn’t much that can be done with my hair.) I’m pretty sure patients feel the same way about which doctor they get. The public is convinced there is a wide variety in the quality of doctors, and they want a good one. If you have to tell them theirs is being assigned by lottery, they won’t be as happy than if you can provide the name and a little information about the doctor they can expect to see the next day.
When the patients I admit late last evening ask who would see them the next day, I’m glad when I can provide a name and a little more information. I say something like, “I won’t see you after tonight, but my partner, Dr. Shawn Lee, will be instead. That means you’re getting an upgrade! Not only is he a really nice guy, he’s voted one of Seattle’s best doctors every year. He’ll do a great job for you.”
To make this communication effective, the night doctor has to know which hospitalist takes over the next morning and has a list indicating which day doctor will get the first, second, third new patient, and so on, admitted during the night. This is possible if patients are assigned by a predetermined algorithm, or if the day doctors have their load-leveling meeting at the end of each day shift, rather than in the morning, to create a list telling the night doctor which day hospitalist he should admit the first and subsequent patents to. That way, the night doctor can write in the admitting orders at 1 a.m. “admit to Dr. X.” This eliminates confusion on the part of other hospital staff who need to know who to call about a patient after the start of the day shift.
Next month I will look at special circumstances, and some pros and cons of having an individual hospitalist take on the care of more patients at the beginning of consecutive day shifts, and exempting them from taking on new patients on the last day or two before rotating off. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Editor’s note: Second of a three-part series.
As I mentioned last month, there isn’t a proven best method to use when distributing new referrals among your group’s providers. The popular methods fall along a continuum of being focused on daily, or continuous, leveling of patient loads between providers (“load leveling”) at one end; at the other end of the continuum is having a doctor be “on” for all new referrals for a predetermined time period, and accepting that patient volumes might be uneven day to day but tend to even out over long periods.
There might not be any reason to change your group’s approach to patient assignment, but you should always be thinking about how your own methods might be changed or improved. I have shared (“Bigger Isn’t Always Better,” June 2009, p. 46) my concern that some groups invest far too much time in a morning load-leveling and handoff conference. Make sure your group is using only as much time as needed.
Air-Traffic Controllers
Many large groups (e.g., more than 15 full-time equivalents) that assign patients to providers in sequence, like dealing a deck of cards, have a designated provider who holds the triage pager and serves as “air-traffic controller.” This person typically takes incoming calls about all new referrals, jots down the relevant clinical data, keeps track of which hospitalist is due to take the next patient, pages that person, and repeats the clinical information. As I’ve written before (“How to Hire and Use Clerical Staff,” June 2007, p. 73), many practices have found that during business hours, they can hand this role to a clerical person who simply takes down the name and phone number of the doctor making the referral, then pages that information to the hospitalist due to get the next patient. The hospitalist then calls and speaks directly with the referring doctor.
Small- to medium-sized groups can eliminate entirely the need for any such “air-traffic control” function if they assign all new referrals to a single doctor for specified periods of time. For example, from 7 a.m. to 3 p.m. today, all new referrals go to Dr. Glass, and from 3 p.m. to 11 p.m., they go to Dr. Cage.
Admitter-Rounder Duties
Many—maybe most?—large groups separate daytime admitter and rounder functions so that on any given day, a hospitalist does one but not both. The principal advantages of this approach are reducing the stress on, and possibly increasing the efficiency of, rounding doctors by shielding them from the unpredictable and time-consuming interruption of needing to admit a new patient. And a daytime doctor who only does admissions might be able to start seeing a patient in the ED more quickly than one who is busy making rounds.
Any increased availability of admitters to the ED could be offset by their lack of surge capacity leading to a bottleneck in ED throughput when there are many patients to admit at the same time and a limited number of admitters (often only one). Such a bottleneck would be much less likely if all daytime doctors (i.e., the rounders) were available to see admissions rather than just admitters.
Continuity of care suffers when a group has separate admitters and rounders, because no patients will be seen by the same doctor on the day of admission and the day following. This method requires a handoff from the day of admission to the next day. Such a handoff might be unavoidable for patients admitted during the night, but this doesn’t have to occur when patients are admitted during the daytime.
Who’s Seeing this Patient?
It seems to make sense to wait until each morning to distribute patients. That allows the practice to know just how many new patients there are, and they can be distributed according to complexity and whether a hospitalist has formed a previous relationship with that patient. But it means that no one at the hospital will know which hospitalist is caring for the patient until later in the morning. For example, if the radiologist is over-reading a study done during the night and finds something worthy of a phone call to the hospitalist, no one is sure who should get the call. A patient might develop hypoglycemia shortly after the hospitalist night shift is over, but the nurse doesn’t know which hospitalist to call.
And, perhaps most importantly, if patients aren’t distributed until the start of the day shift, the night hospitalist can’t tell the patient and family which hospitalist to expect the next morning. To test the significance of this issue, I conducted an experiment while working our group’s late-evening admitter shift. I concluded my visit with each admitted patient by explaining, “I am on-call for our group tonight, so I will be off recovering tomorrow. Therefore, I won’t see you again, but one of my partners will take over in the morning. Do you have any questions for me?” Every patient I admitted had the same question. “What is that doctor’s name?”
How does your group answer a patient who asks which hospitalist will be in the next day? If your method is load-leveling in the morning, then the best answer your night admitting doctor can give is probably to say: “I don’t know which of my partners will be in. There are several working tomorrow, and at the start of the day, they will divide up the patients who come in tonight depending on how busy each of them is. But all the doctors in our group are terrific and will take good care of you.”
I’m told the same thing when I get my hair cut: You’ll get whichever “hair artist” is up next. I put up with it at the hair place because it costs less than $15. But I still find it a little irritating. I’m sure all the barbers aren’t equally skilled or diligent, and I want the best one. (Maybe I shouldn’t care since there isn’t much that can be done with my hair.) I’m pretty sure patients feel the same way about which doctor they get. The public is convinced there is a wide variety in the quality of doctors, and they want a good one. If you have to tell them theirs is being assigned by lottery, they won’t be as happy than if you can provide the name and a little information about the doctor they can expect to see the next day.
When the patients I admit late last evening ask who would see them the next day, I’m glad when I can provide a name and a little more information. I say something like, “I won’t see you after tonight, but my partner, Dr. Shawn Lee, will be instead. That means you’re getting an upgrade! Not only is he a really nice guy, he’s voted one of Seattle’s best doctors every year. He’ll do a great job for you.”
To make this communication effective, the night doctor has to know which hospitalist takes over the next morning and has a list indicating which day doctor will get the first, second, third new patient, and so on, admitted during the night. This is possible if patients are assigned by a predetermined algorithm, or if the day doctors have their load-leveling meeting at the end of each day shift, rather than in the morning, to create a list telling the night doctor which day hospitalist he should admit the first and subsequent patents to. That way, the night doctor can write in the admitting orders at 1 a.m. “admit to Dr. X.” This eliminates confusion on the part of other hospital staff who need to know who to call about a patient after the start of the day shift.
Next month I will look at special circumstances, and some pros and cons of having an individual hospitalist take on the care of more patients at the beginning of consecutive day shifts, and exempting them from taking on new patients on the last day or two before rotating off. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is also course co-director and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
You may not have noticed, but your workload is lighter. So is your wallet.
A new study reveals a trend that most of us on the front line of medical education see daily: The number of hours that any given physician works in a week has decreased dramatically over the past decade, along with the fees that he or she collects.1
The retrospective analysis used data from the US Census Bureau Current Population Survey from 1976 through 2008 (n = 116,733). Trends were estimated among all US physicians and by residency status, sex, age, and work setting. As the authors point out, the decrease in work hours was “broad-based,” occurring in different demographic groups and in different settings.1
Among the findings:
- The mean weekly workload remained steady through the early 1990s, but declined 7.2% between 1996 and 2008 among all physicians, from 54.9 hours in 1996–1998 to 51.0 hours in 2006–2008 (95% confidence interval [CI], 5.3%–9.0%; P < .001).
- The hours worked by nonresident physicians decreased by 5.7% (95% CI, 3.8%–7.7%; P < .001), versus 9.8% among residents (95% CI, 5.8%–13.7%; P < .001).
- Overall, among nonresident physicians, those younger than 45 years experienced the biggest decrease in mean work hours (7.4%; 95% CI, 4.7%–10.2%; P < .001). Non-resident physicians who worked outside a hospital setting also saw a significant decrease (6.4%; 95% CI, 4.1%–8.7%; P < .001).
- Overall, nonresident physicians older than 45 years had the smallest decrease in mean work hours (3.7%; 95% CI, 1.0%–6.5%; P < .008). Nonresident physicians who worked in a hospital setting also experienced a slim reduction (4.0%; 95% CI, 0.4%–7.6%; P =.03).
- Mean physician fees declined 25% (after adjustment for inflation) between 1995 and 2006, coincident with diminishing work hours.
- The largest reduction in mean work hours—9.8% among resident physicians—came after the imposition of limits on resident work hours by the American Council of Graduate Medical Education in 2003.1
Which came first—lower reimbursement or fewer hours?
“The observed decrease in physician fees is a potential economic factor behind the decrease in physician hours,” the authors write.1 For example, hours diminished first in metropolitan statistical areas where fees were lowest in 2001.
The General Accounting Office found that these areas also tended to feature greater competition among physicians, as well as greater penetration by managed care, which may have contributed to lower fees. However, these conditions also could have “weakened physician autonomy and reduced job satisfaction, which in turn could have led to fewer hours.”1
Study sheds light on gender shift, generational differences
In this study, Staiger and colleagues documented the reality of the “gender shift” in medicine: Women constituted 20.6% of respondents and worked a mean of 44.4 hours a week in 2006–2008, compared with 51.7 hours for men.1 However, the authors point out, “[a]lthough female physicians work fewer mean hours and represent an increasing proportion of physicians, decreases in hours were observed for both male and female physicians.”1
Although physicians of all ages worked fewer hours than before, the reduction was greatest among physicians younger than 45 years.1
The study also demonstrated that physicians work significantly longer hours than lawyers, engineers, and nurses. The work hours of these other professions have remained relatively stable since 1976.1
How do fewer hours affect the supply of physicians?
Over the past 10 years, much has been written about a projected shortage in the physician workforce and what should be done about it. In response, the Association of American Medical Colleges requested an expansion in medical school capacity, and many medical schools increased class size. In most cases, however, the reason for the increase in class size was financial: a desire for more tuition dollars, one of the few sources of income under the control of a medical school.
In addition, medical school tuition has continued to rise, and the increase in tuition has markedly outpaced the rate of inflation. The result: Medical students graduate with a large financial debt that influences their choice of specialty. Specialties that have high remuneration such as radiology, dermatology, anesthesiology, and orthopedics have become prime choices for graduating medical students.
The system needs to catch up to reality
As Staiger and his coauthors point out, a 5.7% decrease in the mean number of hours worked among approximately 630,000 physicians is equivalent to a loss of 36,000 physicians from the workforce.1 If the current system is sincere about increasing the physician workforce, why haven’t there been 1) a decrease in the length of medical school, 2) control of tuition escalation, and 3) an increase in the number of residency positions? And why has nothing been done to bring some of the lost physicians back in to the workplace by offering part time or flexible positions?
We won’t solve the physician shortage simply by increasing enrollment and building new medical schools. It’s time to realize that physicians want a work-lifestyle balance similar to that of other professionals. If we don’t make the practice of medicine more enjoyable and align it with the needs and desires of Generation X and Generation Y physicians, we’ll fall short of the number of physicians that we need to care for a growing, and an aging, population.
Reference
1. Staiger DO, Auerbach DI, Buerhaus PI. Trends in the work hours of physicians in the United States. JAMA. 2010;303(8):747-753.
Recommended reading
Weinstein L, Wolfe HM. The downward spiral of physician satisfaction: an attempt to avert a crisis within the medical profession. Obstet Gynecol. 2007;109(5):1181–1183.
Weinstein L. The unbearable unhappiness of the ObGyn: A crisis looms. OBG Management. 2008;20(12):34–42. http://www.obgmanagement.com/article_pages.asp?ref=2012OBGM_Article3. Accessed February 23, 2010.
Weinstein L, Wofle HM. A unique solution to solve the pending medical school tuition crisis. Am J Obstet Gynecol. In press.
A new study reveals a trend that most of us on the front line of medical education see daily: The number of hours that any given physician works in a week has decreased dramatically over the past decade, along with the fees that he or she collects.1
The retrospective analysis used data from the US Census Bureau Current Population Survey from 1976 through 2008 (n = 116,733). Trends were estimated among all US physicians and by residency status, sex, age, and work setting. As the authors point out, the decrease in work hours was “broad-based,” occurring in different demographic groups and in different settings.1
Among the findings:
- The mean weekly workload remained steady through the early 1990s, but declined 7.2% between 1996 and 2008 among all physicians, from 54.9 hours in 1996–1998 to 51.0 hours in 2006–2008 (95% confidence interval [CI], 5.3%–9.0%; P < .001).
- The hours worked by nonresident physicians decreased by 5.7% (95% CI, 3.8%–7.7%; P < .001), versus 9.8% among residents (95% CI, 5.8%–13.7%; P < .001).
- Overall, among nonresident physicians, those younger than 45 years experienced the biggest decrease in mean work hours (7.4%; 95% CI, 4.7%–10.2%; P < .001). Non-resident physicians who worked outside a hospital setting also saw a significant decrease (6.4%; 95% CI, 4.1%–8.7%; P < .001).
- Overall, nonresident physicians older than 45 years had the smallest decrease in mean work hours (3.7%; 95% CI, 1.0%–6.5%; P < .008). Nonresident physicians who worked in a hospital setting also experienced a slim reduction (4.0%; 95% CI, 0.4%–7.6%; P =.03).
- Mean physician fees declined 25% (after adjustment for inflation) between 1995 and 2006, coincident with diminishing work hours.
- The largest reduction in mean work hours—9.8% among resident physicians—came after the imposition of limits on resident work hours by the American Council of Graduate Medical Education in 2003.1
Which came first—lower reimbursement or fewer hours?
“The observed decrease in physician fees is a potential economic factor behind the decrease in physician hours,” the authors write.1 For example, hours diminished first in metropolitan statistical areas where fees were lowest in 2001.
The General Accounting Office found that these areas also tended to feature greater competition among physicians, as well as greater penetration by managed care, which may have contributed to lower fees. However, these conditions also could have “weakened physician autonomy and reduced job satisfaction, which in turn could have led to fewer hours.”1
Study sheds light on gender shift, generational differences
In this study, Staiger and colleagues documented the reality of the “gender shift” in medicine: Women constituted 20.6% of respondents and worked a mean of 44.4 hours a week in 2006–2008, compared with 51.7 hours for men.1 However, the authors point out, “[a]lthough female physicians work fewer mean hours and represent an increasing proportion of physicians, decreases in hours were observed for both male and female physicians.”1
Although physicians of all ages worked fewer hours than before, the reduction was greatest among physicians younger than 45 years.1
The study also demonstrated that physicians work significantly longer hours than lawyers, engineers, and nurses. The work hours of these other professions have remained relatively stable since 1976.1
How do fewer hours affect the supply of physicians?
Over the past 10 years, much has been written about a projected shortage in the physician workforce and what should be done about it. In response, the Association of American Medical Colleges requested an expansion in medical school capacity, and many medical schools increased class size. In most cases, however, the reason for the increase in class size was financial: a desire for more tuition dollars, one of the few sources of income under the control of a medical school.
In addition, medical school tuition has continued to rise, and the increase in tuition has markedly outpaced the rate of inflation. The result: Medical students graduate with a large financial debt that influences their choice of specialty. Specialties that have high remuneration such as radiology, dermatology, anesthesiology, and orthopedics have become prime choices for graduating medical students.
The system needs to catch up to reality
As Staiger and his coauthors point out, a 5.7% decrease in the mean number of hours worked among approximately 630,000 physicians is equivalent to a loss of 36,000 physicians from the workforce.1 If the current system is sincere about increasing the physician workforce, why haven’t there been 1) a decrease in the length of medical school, 2) control of tuition escalation, and 3) an increase in the number of residency positions? And why has nothing been done to bring some of the lost physicians back in to the workplace by offering part time or flexible positions?
We won’t solve the physician shortage simply by increasing enrollment and building new medical schools. It’s time to realize that physicians want a work-lifestyle balance similar to that of other professionals. If we don’t make the practice of medicine more enjoyable and align it with the needs and desires of Generation X and Generation Y physicians, we’ll fall short of the number of physicians that we need to care for a growing, and an aging, population.
A new study reveals a trend that most of us on the front line of medical education see daily: The number of hours that any given physician works in a week has decreased dramatically over the past decade, along with the fees that he or she collects.1
The retrospective analysis used data from the US Census Bureau Current Population Survey from 1976 through 2008 (n = 116,733). Trends were estimated among all US physicians and by residency status, sex, age, and work setting. As the authors point out, the decrease in work hours was “broad-based,” occurring in different demographic groups and in different settings.1
Among the findings:
- The mean weekly workload remained steady through the early 1990s, but declined 7.2% between 1996 and 2008 among all physicians, from 54.9 hours in 1996–1998 to 51.0 hours in 2006–2008 (95% confidence interval [CI], 5.3%–9.0%; P < .001).
- The hours worked by nonresident physicians decreased by 5.7% (95% CI, 3.8%–7.7%; P < .001), versus 9.8% among residents (95% CI, 5.8%–13.7%; P < .001).
- Overall, among nonresident physicians, those younger than 45 years experienced the biggest decrease in mean work hours (7.4%; 95% CI, 4.7%–10.2%; P < .001). Non-resident physicians who worked outside a hospital setting also saw a significant decrease (6.4%; 95% CI, 4.1%–8.7%; P < .001).
- Overall, nonresident physicians older than 45 years had the smallest decrease in mean work hours (3.7%; 95% CI, 1.0%–6.5%; P < .008). Nonresident physicians who worked in a hospital setting also experienced a slim reduction (4.0%; 95% CI, 0.4%–7.6%; P =.03).
- Mean physician fees declined 25% (after adjustment for inflation) between 1995 and 2006, coincident with diminishing work hours.
- The largest reduction in mean work hours—9.8% among resident physicians—came after the imposition of limits on resident work hours by the American Council of Graduate Medical Education in 2003.1
Which came first—lower reimbursement or fewer hours?
“The observed decrease in physician fees is a potential economic factor behind the decrease in physician hours,” the authors write.1 For example, hours diminished first in metropolitan statistical areas where fees were lowest in 2001.
The General Accounting Office found that these areas also tended to feature greater competition among physicians, as well as greater penetration by managed care, which may have contributed to lower fees. However, these conditions also could have “weakened physician autonomy and reduced job satisfaction, which in turn could have led to fewer hours.”1
Study sheds light on gender shift, generational differences
In this study, Staiger and colleagues documented the reality of the “gender shift” in medicine: Women constituted 20.6% of respondents and worked a mean of 44.4 hours a week in 2006–2008, compared with 51.7 hours for men.1 However, the authors point out, “[a]lthough female physicians work fewer mean hours and represent an increasing proportion of physicians, decreases in hours were observed for both male and female physicians.”1
Although physicians of all ages worked fewer hours than before, the reduction was greatest among physicians younger than 45 years.1
The study also demonstrated that physicians work significantly longer hours than lawyers, engineers, and nurses. The work hours of these other professions have remained relatively stable since 1976.1
How do fewer hours affect the supply of physicians?
Over the past 10 years, much has been written about a projected shortage in the physician workforce and what should be done about it. In response, the Association of American Medical Colleges requested an expansion in medical school capacity, and many medical schools increased class size. In most cases, however, the reason for the increase in class size was financial: a desire for more tuition dollars, one of the few sources of income under the control of a medical school.
In addition, medical school tuition has continued to rise, and the increase in tuition has markedly outpaced the rate of inflation. The result: Medical students graduate with a large financial debt that influences their choice of specialty. Specialties that have high remuneration such as radiology, dermatology, anesthesiology, and orthopedics have become prime choices for graduating medical students.
The system needs to catch up to reality
As Staiger and his coauthors point out, a 5.7% decrease in the mean number of hours worked among approximately 630,000 physicians is equivalent to a loss of 36,000 physicians from the workforce.1 If the current system is sincere about increasing the physician workforce, why haven’t there been 1) a decrease in the length of medical school, 2) control of tuition escalation, and 3) an increase in the number of residency positions? And why has nothing been done to bring some of the lost physicians back in to the workplace by offering part time or flexible positions?
We won’t solve the physician shortage simply by increasing enrollment and building new medical schools. It’s time to realize that physicians want a work-lifestyle balance similar to that of other professionals. If we don’t make the practice of medicine more enjoyable and align it with the needs and desires of Generation X and Generation Y physicians, we’ll fall short of the number of physicians that we need to care for a growing, and an aging, population.
Reference
1. Staiger DO, Auerbach DI, Buerhaus PI. Trends in the work hours of physicians in the United States. JAMA. 2010;303(8):747-753.
Recommended reading
Weinstein L, Wolfe HM. The downward spiral of physician satisfaction: an attempt to avert a crisis within the medical profession. Obstet Gynecol. 2007;109(5):1181–1183.
Weinstein L. The unbearable unhappiness of the ObGyn: A crisis looms. OBG Management. 2008;20(12):34–42. http://www.obgmanagement.com/article_pages.asp?ref=2012OBGM_Article3. Accessed February 23, 2010.
Weinstein L, Wofle HM. A unique solution to solve the pending medical school tuition crisis. Am J Obstet Gynecol. In press.
Reference
1. Staiger DO, Auerbach DI, Buerhaus PI. Trends in the work hours of physicians in the United States. JAMA. 2010;303(8):747-753.
Recommended reading
Weinstein L, Wolfe HM. The downward spiral of physician satisfaction: an attempt to avert a crisis within the medical profession. Obstet Gynecol. 2007;109(5):1181–1183.
Weinstein L. The unbearable unhappiness of the ObGyn: A crisis looms. OBG Management. 2008;20(12):34–42. http://www.obgmanagement.com/article_pages.asp?ref=2012OBGM_Article3. Accessed February 23, 2010.
Weinstein L, Wofle HM. A unique solution to solve the pending medical school tuition crisis. Am J Obstet Gynecol. In press.
OBG Management ©2010 Dowden Health Media





