User login
Creating a Planned Gift That’s Meaningful To You
The AGA Research Foundation has helped make significant strides in advancing the treatment and cure of digestive diseases by funding talented investigators.
Planning your gift to benefit AGA Research Foundation in the future is an opportunity to express what matters to you. As an AGA member, you can work with the AGA Research Foundation to ensure that your planned gift is designated for a purpose that meets your goals for leaving a legacy—such as research awards, support for specific programs, or unrestricted gifts to help meet the Foundation’s mission.
In as little as one sentence in your will and/or trust, you can complete your gift: “I give to AGA Research Foundation, a nonprofit corporation currently located at 4930 Del Ray Avenue, Bethesda, MD 20814, or its successor thereto, _________ [written amount or percentage of the estate or description of property] for its unrestricted charitable use and purpose.”
If you have named the AGA Research Foundation in your will or trust, please let us know so we can ensure that your gift is used according to your wishes. Notifying us of your plans will enable us to plan for the use of your future gift. However, if you prefer to remain anonymous, we will keep your name and gift in strict confidence.
Please contact [email protected] for more information. If you are considering a planned gift, consult with your own legal and tax advisors.
The AGA Research Foundation has helped make significant strides in advancing the treatment and cure of digestive diseases by funding talented investigators.
Planning your gift to benefit AGA Research Foundation in the future is an opportunity to express what matters to you. As an AGA member, you can work with the AGA Research Foundation to ensure that your planned gift is designated for a purpose that meets your goals for leaving a legacy—such as research awards, support for specific programs, or unrestricted gifts to help meet the Foundation’s mission.
In as little as one sentence in your will and/or trust, you can complete your gift: “I give to AGA Research Foundation, a nonprofit corporation currently located at 4930 Del Ray Avenue, Bethesda, MD 20814, or its successor thereto, _________ [written amount or percentage of the estate or description of property] for its unrestricted charitable use and purpose.”
If you have named the AGA Research Foundation in your will or trust, please let us know so we can ensure that your gift is used according to your wishes. Notifying us of your plans will enable us to plan for the use of your future gift. However, if you prefer to remain anonymous, we will keep your name and gift in strict confidence.
Please contact [email protected] for more information. If you are considering a planned gift, consult with your own legal and tax advisors.
The AGA Research Foundation has helped make significant strides in advancing the treatment and cure of digestive diseases by funding talented investigators.
Planning your gift to benefit AGA Research Foundation in the future is an opportunity to express what matters to you. As an AGA member, you can work with the AGA Research Foundation to ensure that your planned gift is designated for a purpose that meets your goals for leaving a legacy—such as research awards, support for specific programs, or unrestricted gifts to help meet the Foundation’s mission.
In as little as one sentence in your will and/or trust, you can complete your gift: “I give to AGA Research Foundation, a nonprofit corporation currently located at 4930 Del Ray Avenue, Bethesda, MD 20814, or its successor thereto, _________ [written amount or percentage of the estate or description of property] for its unrestricted charitable use and purpose.”
If you have named the AGA Research Foundation in your will or trust, please let us know so we can ensure that your gift is used according to your wishes. Notifying us of your plans will enable us to plan for the use of your future gift. However, if you prefer to remain anonymous, we will keep your name and gift in strict confidence.
Please contact [email protected] for more information. If you are considering a planned gift, consult with your own legal and tax advisors.
Hospital to home tracheostomy care
SLEEP MEDICINE NETWORK
Home-Based Mechanical Ventilation and Neuromuscular Section
Technological improvement has enhanced our ability to support these patients with complex conditions in their home settings. However, clinical practice guidelines are lacking, and current practice relies on a consensus of expert opinions.1-3
Once a patient who has had a tracheostomy begins transitioning care to home, identifying caregivers is vital.
Caregivers need to be educated on daily tracheostomy care, airway clearance, and ventilator management.
Protocols to standardize this transition, such as the “Trach Trail” protocol, help reduce ICU readmissions with new tracheostomies (P = .05), eliminate predischarge mortality (P = .05), and may decrease ICU length of stay (P = 0.72).4 Standardized protocols for aspects of tracheostomy care, such as the “Go-Bag” from Boston Children’s Hospital, ensure that a consistent approach keeps providers, families, and patients familiar with their equipment and safety procedures, improving outcomes and decreasing tracheostomy-related adverse events.4-6
Understanding the landscape surrounding which equipment companies have trained field respiratory therapists is crucial. Airway clearance is key to improving ventilation and oxygenation and maintaining tracheostomy patency. Knowing the types of airway clearance modalities used for each patient remains critical.
Trach care may look substantially different for some populations, like patients in the neonatal ICU. Trach changes may happen more frequently. Speaking valve times may be gradually increased while planning for possible decannulation. Skin care involving granulation tissue and stoma complications is particularly important for this population. Active infants need well-fitting trach ties to balance enough support to maintain their trach without causing skin breakdown or discomfort. Securing the trach to prevent pulling or dislodgement as infants become more active is crucial as developmental milestones are achieved.
We hope national societies prioritize standardizing care for this vulnerable population while promoting additional high-quality, patient-centered outcomes in research studies. Implementation strategies to promote interprofessional teams to enhance education, communication, and outcomes will reduce health care disparities.
References
1. Am J Respir Crit Care Med Vol 161. pp Sherman JM, Davis S, Albamonte-Petrick S, et al. Care of the child with a chronic tracheostomy. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med. 2000;161(1):297-308. doi: 10.1164/ajrccm.161.1.ats1-00 297-308, 2000
2. Mitchell RB, Hussey HM, Setzen G, et al. Clinical consensus statement: tracheostomy care. Otolaryngol Head Neck Surg. 2013;148(1):6-20. Preprint. Posted online September 18, 2012. PMID: 22990518. doi: 10.1177/0194599812460376
3. Sterni LM, Collaco JM, Baker CD, et al; ATS Pediatric Chronic Home Ventilation Workgroup. An official American Thoracic Society clinical practice guideline: pediatric chronic home invasive ventilation. Am J Respir Crit Care Med. 2016;193(8):e16-35. PMID: 27082538; PMCID: PMC5439679. doi: 10.1164/rccm.201602-0276ST
4. Cherney RL, Pandian V, Ninan A, et al. The Trach Trail: a systems-based pathway to improve quality of tracheostomy care and interdisciplinary collaboration. Otolaryngol Head Neck Surg. 2020;163(2):232-243. doi: 10.1177/0194599820917427
5. Brown J. Tracheostomy to noninvasive ventilation: from acute care to home. Sleep Med Clin. 2020;15(4):593-598. doi: 10.1016/j.jsmc.2020.08.003
6. Kohn J, McKeon M, Munhall D, Blanchette S, Wells S, Watters K. Standardization of pediatric tracheostomy care with “Go-bags.” Int J Pediatr Otorhinolaryngol. 2019;121:154-156. doi: 10.1016/j.ijporl.2019.03.022
SLEEP MEDICINE NETWORK
Home-Based Mechanical Ventilation and Neuromuscular Section
Technological improvement has enhanced our ability to support these patients with complex conditions in their home settings. However, clinical practice guidelines are lacking, and current practice relies on a consensus of expert opinions.1-3
Once a patient who has had a tracheostomy begins transitioning care to home, identifying caregivers is vital.
Caregivers need to be educated on daily tracheostomy care, airway clearance, and ventilator management.
Protocols to standardize this transition, such as the “Trach Trail” protocol, help reduce ICU readmissions with new tracheostomies (P = .05), eliminate predischarge mortality (P = .05), and may decrease ICU length of stay (P = 0.72).4 Standardized protocols for aspects of tracheostomy care, such as the “Go-Bag” from Boston Children’s Hospital, ensure that a consistent approach keeps providers, families, and patients familiar with their equipment and safety procedures, improving outcomes and decreasing tracheostomy-related adverse events.4-6
Understanding the landscape surrounding which equipment companies have trained field respiratory therapists is crucial. Airway clearance is key to improving ventilation and oxygenation and maintaining tracheostomy patency. Knowing the types of airway clearance modalities used for each patient remains critical.
Trach care may look substantially different for some populations, like patients in the neonatal ICU. Trach changes may happen more frequently. Speaking valve times may be gradually increased while planning for possible decannulation. Skin care involving granulation tissue and stoma complications is particularly important for this population. Active infants need well-fitting trach ties to balance enough support to maintain their trach without causing skin breakdown or discomfort. Securing the trach to prevent pulling or dislodgement as infants become more active is crucial as developmental milestones are achieved.
We hope national societies prioritize standardizing care for this vulnerable population while promoting additional high-quality, patient-centered outcomes in research studies. Implementation strategies to promote interprofessional teams to enhance education, communication, and outcomes will reduce health care disparities.
References
1. Am J Respir Crit Care Med Vol 161. pp Sherman JM, Davis S, Albamonte-Petrick S, et al. Care of the child with a chronic tracheostomy. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med. 2000;161(1):297-308. doi: 10.1164/ajrccm.161.1.ats1-00 297-308, 2000
2. Mitchell RB, Hussey HM, Setzen G, et al. Clinical consensus statement: tracheostomy care. Otolaryngol Head Neck Surg. 2013;148(1):6-20. Preprint. Posted online September 18, 2012. PMID: 22990518. doi: 10.1177/0194599812460376
3. Sterni LM, Collaco JM, Baker CD, et al; ATS Pediatric Chronic Home Ventilation Workgroup. An official American Thoracic Society clinical practice guideline: pediatric chronic home invasive ventilation. Am J Respir Crit Care Med. 2016;193(8):e16-35. PMID: 27082538; PMCID: PMC5439679. doi: 10.1164/rccm.201602-0276ST
4. Cherney RL, Pandian V, Ninan A, et al. The Trach Trail: a systems-based pathway to improve quality of tracheostomy care and interdisciplinary collaboration. Otolaryngol Head Neck Surg. 2020;163(2):232-243. doi: 10.1177/0194599820917427
5. Brown J. Tracheostomy to noninvasive ventilation: from acute care to home. Sleep Med Clin. 2020;15(4):593-598. doi: 10.1016/j.jsmc.2020.08.003
6. Kohn J, McKeon M, Munhall D, Blanchette S, Wells S, Watters K. Standardization of pediatric tracheostomy care with “Go-bags.” Int J Pediatr Otorhinolaryngol. 2019;121:154-156. doi: 10.1016/j.ijporl.2019.03.022
SLEEP MEDICINE NETWORK
Home-Based Mechanical Ventilation and Neuromuscular Section
Technological improvement has enhanced our ability to support these patients with complex conditions in their home settings. However, clinical practice guidelines are lacking, and current practice relies on a consensus of expert opinions.1-3
Once a patient who has had a tracheostomy begins transitioning care to home, identifying caregivers is vital.
Caregivers need to be educated on daily tracheostomy care, airway clearance, and ventilator management.
Protocols to standardize this transition, such as the “Trach Trail” protocol, help reduce ICU readmissions with new tracheostomies (P = .05), eliminate predischarge mortality (P = .05), and may decrease ICU length of stay (P = 0.72).4 Standardized protocols for aspects of tracheostomy care, such as the “Go-Bag” from Boston Children’s Hospital, ensure that a consistent approach keeps providers, families, and patients familiar with their equipment and safety procedures, improving outcomes and decreasing tracheostomy-related adverse events.4-6
Understanding the landscape surrounding which equipment companies have trained field respiratory therapists is crucial. Airway clearance is key to improving ventilation and oxygenation and maintaining tracheostomy patency. Knowing the types of airway clearance modalities used for each patient remains critical.
Trach care may look substantially different for some populations, like patients in the neonatal ICU. Trach changes may happen more frequently. Speaking valve times may be gradually increased while planning for possible decannulation. Skin care involving granulation tissue and stoma complications is particularly important for this population. Active infants need well-fitting trach ties to balance enough support to maintain their trach without causing skin breakdown or discomfort. Securing the trach to prevent pulling or dislodgement as infants become more active is crucial as developmental milestones are achieved.
We hope national societies prioritize standardizing care for this vulnerable population while promoting additional high-quality, patient-centered outcomes in research studies. Implementation strategies to promote interprofessional teams to enhance education, communication, and outcomes will reduce health care disparities.
References
1. Am J Respir Crit Care Med Vol 161. pp Sherman JM, Davis S, Albamonte-Petrick S, et al. Care of the child with a chronic tracheostomy. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med. 2000;161(1):297-308. doi: 10.1164/ajrccm.161.1.ats1-00 297-308, 2000
2. Mitchell RB, Hussey HM, Setzen G, et al. Clinical consensus statement: tracheostomy care. Otolaryngol Head Neck Surg. 2013;148(1):6-20. Preprint. Posted online September 18, 2012. PMID: 22990518. doi: 10.1177/0194599812460376
3. Sterni LM, Collaco JM, Baker CD, et al; ATS Pediatric Chronic Home Ventilation Workgroup. An official American Thoracic Society clinical practice guideline: pediatric chronic home invasive ventilation. Am J Respir Crit Care Med. 2016;193(8):e16-35. PMID: 27082538; PMCID: PMC5439679. doi: 10.1164/rccm.201602-0276ST
4. Cherney RL, Pandian V, Ninan A, et al. The Trach Trail: a systems-based pathway to improve quality of tracheostomy care and interdisciplinary collaboration. Otolaryngol Head Neck Surg. 2020;163(2):232-243. doi: 10.1177/0194599820917427
5. Brown J. Tracheostomy to noninvasive ventilation: from acute care to home. Sleep Med Clin. 2020;15(4):593-598. doi: 10.1016/j.jsmc.2020.08.003
6. Kohn J, McKeon M, Munhall D, Blanchette S, Wells S, Watters K. Standardization of pediatric tracheostomy care with “Go-bags.” Int J Pediatr Otorhinolaryngol. 2019;121:154-156. doi: 10.1016/j.ijporl.2019.03.022
HALT early recognition is key
DIFFUSE LUNG DISEASE AND LUNG TRANSPLANT NETWORK
Lung Transplant Section
Hyperammonemia after lung transplantation (HALT) is a rare but serious complication occurring in 1% to 4% of patients with high morbidity and mortality. Early recognition is crucial, as mortality rates can reach 75%.1
HALT arises from excess ammonia production or decreased clearance and is often linked to infections by urea-splitting organisms, including mycoplasma and ureaplasma. Prompt, aggressive treatment is essential and typically includes dietary protein restriction, renal replacement therapy (ideally intermittent hemodialysis), bowel decontamination (lactulose, rifaximin, metronidazole, or neomycin), amino acids (arginine and levocarnitine), nitrogen scavengers (sodium phenylbutyrate or glycerol phenylbutyrate), and empiric antimicrobial coverage for urea-splitting organisms.2 Given concerns for calcineurin inhibitor-induced hyperammonemia, transition to an alternative agent may be considered.
Given the severe risks associated with HALT, vigilance is vital, particularly in intubated and sedated patients where monitoring of neurologic status is more challenging. Protocols may involve routine serum ammonia monitoring, polymerase chain reaction testing for mycoplasma and ureaplasma at the time of transplant or with postoperative bronchoscopy, and empiric antimicrobial treatment. No definitive ammonia threshold exists, but altered sensorium with elevated levels warrants immediate and more aggressive treatment with levels >75 μmol/L. Early testing and symptom recognition can significantly improve survival rates in this potentially devastating condition.
References
1. Leger RF, Silverman MS, Hauck ES, Guvakova KD. Hyperammonemia post lung transplantation: a review. Clin Med Insights Circ Respir Pulm Med. 2020;14:1179548420966234. doi:10.1177/1179548420966234
2. Chen C, Bain KB, Luppa JA. Hyperammonemia syndrome after lung transplantation: a single center experience. Transplantation. 2016;100(3):678-684. doi:10.1097/TP.0000000000000868
DIFFUSE LUNG DISEASE AND LUNG TRANSPLANT NETWORK
Lung Transplant Section
Hyperammonemia after lung transplantation (HALT) is a rare but serious complication occurring in 1% to 4% of patients with high morbidity and mortality. Early recognition is crucial, as mortality rates can reach 75%.1
HALT arises from excess ammonia production or decreased clearance and is often linked to infections by urea-splitting organisms, including mycoplasma and ureaplasma. Prompt, aggressive treatment is essential and typically includes dietary protein restriction, renal replacement therapy (ideally intermittent hemodialysis), bowel decontamination (lactulose, rifaximin, metronidazole, or neomycin), amino acids (arginine and levocarnitine), nitrogen scavengers (sodium phenylbutyrate or glycerol phenylbutyrate), and empiric antimicrobial coverage for urea-splitting organisms.2 Given concerns for calcineurin inhibitor-induced hyperammonemia, transition to an alternative agent may be considered.
Given the severe risks associated with HALT, vigilance is vital, particularly in intubated and sedated patients where monitoring of neurologic status is more challenging. Protocols may involve routine serum ammonia monitoring, polymerase chain reaction testing for mycoplasma and ureaplasma at the time of transplant or with postoperative bronchoscopy, and empiric antimicrobial treatment. No definitive ammonia threshold exists, but altered sensorium with elevated levels warrants immediate and more aggressive treatment with levels >75 μmol/L. Early testing and symptom recognition can significantly improve survival rates in this potentially devastating condition.
References
1. Leger RF, Silverman MS, Hauck ES, Guvakova KD. Hyperammonemia post lung transplantation: a review. Clin Med Insights Circ Respir Pulm Med. 2020;14:1179548420966234. doi:10.1177/1179548420966234
2. Chen C, Bain KB, Luppa JA. Hyperammonemia syndrome after lung transplantation: a single center experience. Transplantation. 2016;100(3):678-684. doi:10.1097/TP.0000000000000868
DIFFUSE LUNG DISEASE AND LUNG TRANSPLANT NETWORK
Lung Transplant Section
Hyperammonemia after lung transplantation (HALT) is a rare but serious complication occurring in 1% to 4% of patients with high morbidity and mortality. Early recognition is crucial, as mortality rates can reach 75%.1
HALT arises from excess ammonia production or decreased clearance and is often linked to infections by urea-splitting organisms, including mycoplasma and ureaplasma. Prompt, aggressive treatment is essential and typically includes dietary protein restriction, renal replacement therapy (ideally intermittent hemodialysis), bowel decontamination (lactulose, rifaximin, metronidazole, or neomycin), amino acids (arginine and levocarnitine), nitrogen scavengers (sodium phenylbutyrate or glycerol phenylbutyrate), and empiric antimicrobial coverage for urea-splitting organisms.2 Given concerns for calcineurin inhibitor-induced hyperammonemia, transition to an alternative agent may be considered.
Given the severe risks associated with HALT, vigilance is vital, particularly in intubated and sedated patients where monitoring of neurologic status is more challenging. Protocols may involve routine serum ammonia monitoring, polymerase chain reaction testing for mycoplasma and ureaplasma at the time of transplant or with postoperative bronchoscopy, and empiric antimicrobial treatment. No definitive ammonia threshold exists, but altered sensorium with elevated levels warrants immediate and more aggressive treatment with levels >75 μmol/L. Early testing and symptom recognition can significantly improve survival rates in this potentially devastating condition.
References
1. Leger RF, Silverman MS, Hauck ES, Guvakova KD. Hyperammonemia post lung transplantation: a review. Clin Med Insights Circ Respir Pulm Med. 2020;14:1179548420966234. doi:10.1177/1179548420966234
2. Chen C, Bain KB, Luppa JA. Hyperammonemia syndrome after lung transplantation: a single center experience. Transplantation. 2016;100(3):678-684. doi:10.1097/TP.0000000000000868
Advancements in nutritional management for critically ill patients
CRITICAL CARE NETWORK
Nonrespiratory Critical Care Section
Nutrition plays an important role in the management and recovery of critically ill patients admitted to the ICU. Major guidelines recommend that critically ill patients should receive 1.2 to 2.0 g/kg/day of protein, with an emphasis on early (within 48 hours of ICU admission) enteral nutrition.1-3
In a randomized controlled trial involving 173 critically ill patients who stayed in the ICU in Zhejiang, China, Wang and colleagues studied the impact of early high protein intake (1.5 g/kg/day vs 0.8 g/kg/day).4 The primary outcome of 28-day mortality was lower among the high protein intake group (8.14% vs 19.54%). Still, this intention-to-treat analysis did not reach a statistical significance (P = .051). However, a time-to-event analysis using the Cox proportional hazard model showed that the high protein intake group had a significantly lower 28-day mortality rate, shorter ICU stays, and improved nutritional status, particularly in patients with sepsis (P = .045).
In a systematic review and meta-analysis involving 19 randomized controlled trials and 1,731 patients, there was no definitive evidence that higher protein intake significantly reduces mortality. However, it may improve specific clinical outcomes like muscle mass retention and shorter duration of mechanical ventilation.5 Similarly, a post hoc analysis on the EFFORT Protein Trial focusing on critically ill patients with acute kidney injury (AKI) showed that higher protein intake did not significantly impact the duration of kidney replacement therapy but was associated with higher serum urea levels and slower time-to-discharge-alive among patients with AKI.6
For critically ill patients, increasing early protein intake to 1.5 g/kg/day is safe and may be beneficial. We still need more data to guide the best approach to determining the protein intake.
References
1. Taylor BE, McClave SA, Martindale RG, et al. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (ASPEN). Crit Care Med. 2016;44(2):390-438. doi:10.1097/CCM.0000000000001525
2. Singer P, Blaser AR, Berger MM, et al. ESPEN guideline on clinical nutrition in the intensive care unit. Clin Nutr. 2019;38(1):48-79. doi:10.1016/j.clnu.2018.08.037
3. McClave SA, Taylor BE, Martindale RG, et al. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (ASPEN). JJPEN J Parenter Enteral Nutr. 2016;40(2):159-211. doi:10.1177/0148607115621863
4. Wang Y, Ye Y, Xuan L, et al. Impact of early high protein intake in critically ill patients: a randomized controlled trial. Nutr Metab. 2024;21(1):39. doi.org/10.1186/s12986-024-00818-8
5. Lee ZY, Yap CSL, Hasan MS, et al. The effect of higher versus lower protein delivery in critically ill patients: a systematic review and meta-analysis of randomized controlled trials. Crit Care. 2021;25(1):260. doi.org/10.1186/s13054-021-03693-4
6. Stoppe C, Patel JJ, Zarbock A, et al. The impact of higher protein dosing on outcomes in critically ill patients with acute kidney injury: a post hoc analysis of the EFFORT protein trial. Crit Care. 2023;27(1):399. doi.org/10.1186/s13054-023-04663-8
CRITICAL CARE NETWORK
Nonrespiratory Critical Care Section
Nutrition plays an important role in the management and recovery of critically ill patients admitted to the ICU. Major guidelines recommend that critically ill patients should receive 1.2 to 2.0 g/kg/day of protein, with an emphasis on early (within 48 hours of ICU admission) enteral nutrition.1-3
In a randomized controlled trial involving 173 critically ill patients who stayed in the ICU in Zhejiang, China, Wang and colleagues studied the impact of early high protein intake (1.5 g/kg/day vs 0.8 g/kg/day).4 The primary outcome of 28-day mortality was lower among the high protein intake group (8.14% vs 19.54%). Still, this intention-to-treat analysis did not reach a statistical significance (P = .051). However, a time-to-event analysis using the Cox proportional hazard model showed that the high protein intake group had a significantly lower 28-day mortality rate, shorter ICU stays, and improved nutritional status, particularly in patients with sepsis (P = .045).
In a systematic review and meta-analysis involving 19 randomized controlled trials and 1,731 patients, there was no definitive evidence that higher protein intake significantly reduces mortality. However, it may improve specific clinical outcomes like muscle mass retention and shorter duration of mechanical ventilation.5 Similarly, a post hoc analysis on the EFFORT Protein Trial focusing on critically ill patients with acute kidney injury (AKI) showed that higher protein intake did not significantly impact the duration of kidney replacement therapy but was associated with higher serum urea levels and slower time-to-discharge-alive among patients with AKI.6
For critically ill patients, increasing early protein intake to 1.5 g/kg/day is safe and may be beneficial. We still need more data to guide the best approach to determining the protein intake.
References
1. Taylor BE, McClave SA, Martindale RG, et al. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (ASPEN). Crit Care Med. 2016;44(2):390-438. doi:10.1097/CCM.0000000000001525
2. Singer P, Blaser AR, Berger MM, et al. ESPEN guideline on clinical nutrition in the intensive care unit. Clin Nutr. 2019;38(1):48-79. doi:10.1016/j.clnu.2018.08.037
3. McClave SA, Taylor BE, Martindale RG, et al. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (ASPEN). JJPEN J Parenter Enteral Nutr. 2016;40(2):159-211. doi:10.1177/0148607115621863
4. Wang Y, Ye Y, Xuan L, et al. Impact of early high protein intake in critically ill patients: a randomized controlled trial. Nutr Metab. 2024;21(1):39. doi.org/10.1186/s12986-024-00818-8
5. Lee ZY, Yap CSL, Hasan MS, et al. The effect of higher versus lower protein delivery in critically ill patients: a systematic review and meta-analysis of randomized controlled trials. Crit Care. 2021;25(1):260. doi.org/10.1186/s13054-021-03693-4
6. Stoppe C, Patel JJ, Zarbock A, et al. The impact of higher protein dosing on outcomes in critically ill patients with acute kidney injury: a post hoc analysis of the EFFORT protein trial. Crit Care. 2023;27(1):399. doi.org/10.1186/s13054-023-04663-8
CRITICAL CARE NETWORK
Nonrespiratory Critical Care Section
Nutrition plays an important role in the management and recovery of critically ill patients admitted to the ICU. Major guidelines recommend that critically ill patients should receive 1.2 to 2.0 g/kg/day of protein, with an emphasis on early (within 48 hours of ICU admission) enteral nutrition.1-3
In a randomized controlled trial involving 173 critically ill patients who stayed in the ICU in Zhejiang, China, Wang and colleagues studied the impact of early high protein intake (1.5 g/kg/day vs 0.8 g/kg/day).4 The primary outcome of 28-day mortality was lower among the high protein intake group (8.14% vs 19.54%). Still, this intention-to-treat analysis did not reach a statistical significance (P = .051). However, a time-to-event analysis using the Cox proportional hazard model showed that the high protein intake group had a significantly lower 28-day mortality rate, shorter ICU stays, and improved nutritional status, particularly in patients with sepsis (P = .045).
In a systematic review and meta-analysis involving 19 randomized controlled trials and 1,731 patients, there was no definitive evidence that higher protein intake significantly reduces mortality. However, it may improve specific clinical outcomes like muscle mass retention and shorter duration of mechanical ventilation.5 Similarly, a post hoc analysis on the EFFORT Protein Trial focusing on critically ill patients with acute kidney injury (AKI) showed that higher protein intake did not significantly impact the duration of kidney replacement therapy but was associated with higher serum urea levels and slower time-to-discharge-alive among patients with AKI.6
For critically ill patients, increasing early protein intake to 1.5 g/kg/day is safe and may be beneficial. We still need more data to guide the best approach to determining the protein intake.
References
1. Taylor BE, McClave SA, Martindale RG, et al. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (ASPEN). Crit Care Med. 2016;44(2):390-438. doi:10.1097/CCM.0000000000001525
2. Singer P, Blaser AR, Berger MM, et al. ESPEN guideline on clinical nutrition in the intensive care unit. Clin Nutr. 2019;38(1):48-79. doi:10.1016/j.clnu.2018.08.037
3. McClave SA, Taylor BE, Martindale RG, et al. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (ASPEN). JJPEN J Parenter Enteral Nutr. 2016;40(2):159-211. doi:10.1177/0148607115621863
4. Wang Y, Ye Y, Xuan L, et al. Impact of early high protein intake in critically ill patients: a randomized controlled trial. Nutr Metab. 2024;21(1):39. doi.org/10.1186/s12986-024-00818-8
5. Lee ZY, Yap CSL, Hasan MS, et al. The effect of higher versus lower protein delivery in critically ill patients: a systematic review and meta-analysis of randomized controlled trials. Crit Care. 2021;25(1):260. doi.org/10.1186/s13054-021-03693-4
6. Stoppe C, Patel JJ, Zarbock A, et al. The impact of higher protein dosing on outcomes in critically ill patients with acute kidney injury: a post hoc analysis of the EFFORT protein trial. Crit Care. 2023;27(1):399. doi.org/10.1186/s13054-023-04663-8
New developments on the forefront of intermediate-risk pulmonary embolism
PULMONARY VASCULAR AND CARDIOVASCULAR NETWORK
Cardiovascular Medicine and Surgery Section
Patients with intermediate-risk pulmonary embolism (IRPE), or those with right ventricular dysfunction without overt hemodynamic instability, represent a heterogenous population with short-term mortality ranging from 2% to 17%.1 While systemic anticoagulation is the mainstay therapy, select individuals may benefit from more immediate reperfusion. Unfortunately, only small, randomized trials exploring surrogate outcomes are available to guide modality and patient selection.2
To better define which patients with IRPE are best managed with which therapy, several large-scale randomized controlled trials are underway. PE-TRACT, a study funded by the National Institutes of Health, aims to randomize 500 patients with IRPE to anticoagulation alone vs one of several modalities of CBT with a focus on long-term functional outcomes, including peak oxygen consumption at 3 months and functional class at 1 year. Aspiration thrombectomy with the FlowTriever® device is being compared with anticoagulation alone in a study of 1,200 patients examining short-term composite end points.
While full-dose thrombolysis may decrease the composite outcome of death or hemodynamic deterioration in this population, the benefit is counterbalanced by the risk of significant bleeding. Whether reduced-dose thrombolysis is associated with improved outcomes has been questioned in several small studies. The PEITHO-3 trial plans to randomize 650 patients with IRPE to reduced-dose thrombolytics vs placebo, exploring several outcomes at 30 days. With multiple large trials ongoing, we anticipate important changes to the landscape of IRPE care over the coming years.
References
1. Fernández C, Bova C, Sanchez O, et al. Validation of a model for identification of patients at intermediate to high risk for complications associated with acute symptomatic pulmonary embolism. Chest. 2015;148(1):211-218. doi:10.1378/chest.14-2551
2. Yuriditsky E, Horowitz JM. The role of the PERT in the management and therapeutic decision-making in pulmonary embolism. Eur Heart J Acute Cardiovasc Care. 2022;11(9):693-694. doi:10.1093/ehjacc/zuac102
3. National Library of Medicine (US). A Randomized Trial of Ultrasound-facilitated, Catheter-directed, Thrombolysis Versus Anticoagulation for Acute Intermediate-high Risk Pulmonary Embolism: The Higher-risk Pulmonary Embolism Thrombolysis Study. Updated July 16, 2024. https://clinicaltrials.gov/study/NCT04790370
4. National Library of Medicine (US). PEERLESS II: RCT of FlowTriever vs. Anticoagulation Alone in Pulmonary Embolism. Updated July 17, 2024. https://clinicaltrials.gov/study/NCT06055920
5. National Library of Medicine (US). A Reduced Dose of Thrombolytic Treatment for Patients With Intermediate High-risk Acute Pulmonary Embolism: a Randomized Controled Trial. Updated July 17, 2024. https://clinicaltrials.gov/study/NCT04430569
PULMONARY VASCULAR AND CARDIOVASCULAR NETWORK
Cardiovascular Medicine and Surgery Section
Patients with intermediate-risk pulmonary embolism (IRPE), or those with right ventricular dysfunction without overt hemodynamic instability, represent a heterogenous population with short-term mortality ranging from 2% to 17%.1 While systemic anticoagulation is the mainstay therapy, select individuals may benefit from more immediate reperfusion. Unfortunately, only small, randomized trials exploring surrogate outcomes are available to guide modality and patient selection.2
To better define which patients with IRPE are best managed with which therapy, several large-scale randomized controlled trials are underway. PE-TRACT, a study funded by the National Institutes of Health, aims to randomize 500 patients with IRPE to anticoagulation alone vs one of several modalities of CBT with a focus on long-term functional outcomes, including peak oxygen consumption at 3 months and functional class at 1 year. Aspiration thrombectomy with the FlowTriever® device is being compared with anticoagulation alone in a study of 1,200 patients examining short-term composite end points.
While full-dose thrombolysis may decrease the composite outcome of death or hemodynamic deterioration in this population, the benefit is counterbalanced by the risk of significant bleeding. Whether reduced-dose thrombolysis is associated with improved outcomes has been questioned in several small studies. The PEITHO-3 trial plans to randomize 650 patients with IRPE to reduced-dose thrombolytics vs placebo, exploring several outcomes at 30 days. With multiple large trials ongoing, we anticipate important changes to the landscape of IRPE care over the coming years.
References
1. Fernández C, Bova C, Sanchez O, et al. Validation of a model for identification of patients at intermediate to high risk for complications associated with acute symptomatic pulmonary embolism. Chest. 2015;148(1):211-218. doi:10.1378/chest.14-2551
2. Yuriditsky E, Horowitz JM. The role of the PERT in the management and therapeutic decision-making in pulmonary embolism. Eur Heart J Acute Cardiovasc Care. 2022;11(9):693-694. doi:10.1093/ehjacc/zuac102
3. National Library of Medicine (US). A Randomized Trial of Ultrasound-facilitated, Catheter-directed, Thrombolysis Versus Anticoagulation for Acute Intermediate-high Risk Pulmonary Embolism: The Higher-risk Pulmonary Embolism Thrombolysis Study. Updated July 16, 2024. https://clinicaltrials.gov/study/NCT04790370
4. National Library of Medicine (US). PEERLESS II: RCT of FlowTriever vs. Anticoagulation Alone in Pulmonary Embolism. Updated July 17, 2024. https://clinicaltrials.gov/study/NCT06055920
5. National Library of Medicine (US). A Reduced Dose of Thrombolytic Treatment for Patients With Intermediate High-risk Acute Pulmonary Embolism: a Randomized Controled Trial. Updated July 17, 2024. https://clinicaltrials.gov/study/NCT04430569
PULMONARY VASCULAR AND CARDIOVASCULAR NETWORK
Cardiovascular Medicine and Surgery Section
Patients with intermediate-risk pulmonary embolism (IRPE), or those with right ventricular dysfunction without overt hemodynamic instability, represent a heterogenous population with short-term mortality ranging from 2% to 17%.1 While systemic anticoagulation is the mainstay therapy, select individuals may benefit from more immediate reperfusion. Unfortunately, only small, randomized trials exploring surrogate outcomes are available to guide modality and patient selection.2
To better define which patients with IRPE are best managed with which therapy, several large-scale randomized controlled trials are underway. PE-TRACT, a study funded by the National Institutes of Health, aims to randomize 500 patients with IRPE to anticoagulation alone vs one of several modalities of CBT with a focus on long-term functional outcomes, including peak oxygen consumption at 3 months and functional class at 1 year. Aspiration thrombectomy with the FlowTriever® device is being compared with anticoagulation alone in a study of 1,200 patients examining short-term composite end points.
While full-dose thrombolysis may decrease the composite outcome of death or hemodynamic deterioration in this population, the benefit is counterbalanced by the risk of significant bleeding. Whether reduced-dose thrombolysis is associated with improved outcomes has been questioned in several small studies. The PEITHO-3 trial plans to randomize 650 patients with IRPE to reduced-dose thrombolytics vs placebo, exploring several outcomes at 30 days. With multiple large trials ongoing, we anticipate important changes to the landscape of IRPE care over the coming years.
References
1. Fernández C, Bova C, Sanchez O, et al. Validation of a model for identification of patients at intermediate to high risk for complications associated with acute symptomatic pulmonary embolism. Chest. 2015;148(1):211-218. doi:10.1378/chest.14-2551
2. Yuriditsky E, Horowitz JM. The role of the PERT in the management and therapeutic decision-making in pulmonary embolism. Eur Heart J Acute Cardiovasc Care. 2022;11(9):693-694. doi:10.1093/ehjacc/zuac102
3. National Library of Medicine (US). A Randomized Trial of Ultrasound-facilitated, Catheter-directed, Thrombolysis Versus Anticoagulation for Acute Intermediate-high Risk Pulmonary Embolism: The Higher-risk Pulmonary Embolism Thrombolysis Study. Updated July 16, 2024. https://clinicaltrials.gov/study/NCT04790370
4. National Library of Medicine (US). PEERLESS II: RCT of FlowTriever vs. Anticoagulation Alone in Pulmonary Embolism. Updated July 17, 2024. https://clinicaltrials.gov/study/NCT06055920
5. National Library of Medicine (US). A Reduced Dose of Thrombolytic Treatment for Patients With Intermediate High-risk Acute Pulmonary Embolism: a Randomized Controled Trial. Updated July 17, 2024. https://clinicaltrials.gov/study/NCT04430569
Bronchiectasis: A call to action
AIRWAYS DISORDERS NETWORK
Bronchiectasis Section
For years, the noncystic fibrosis (CF) bronchiectasis community has been trying to organize to provide better care for more than half a million adults with bronchiectasis in the United States. Internationally, the Europeans created the European Bronchiectasis Registry, which has been a powerful tool including nearly 20,000 patients, to answer important epidemiologic and management questions. We must do more for the bronchiectasis community.
Clinicaltrials.gov indicates that there are 8 international phase 3 or 4 clinical trials that are currently enrolling; 3 of those have enrollment sites in the United States. One such study from University of North Carolina at Chapel Hill is looking at the use of nebulized hypertonic saline in patients with non-CF bronchiectasis to understand the effect it has on mucociliary clearance. Emory University is looking at the use of elexacaftor/tezacaftor/ivacaftor (Trikafta) in patients with non-CF bronchiectasis; these patients have only 1 targetable mutation and a phenotype that resembles CF. This 8-week, open-label, single-center study aims to measure both clinical and biomarker outcomes after treatment with Trikafta. Finally, a phase 3 trial out of Florida, the ICoN-1 study, is examining the efficacy and safety of inhaled clofazimine in the treatment of nontuberculous mycobacteria (NTM). This double-blind, randomized trial will look at culture conversion and quality of life measures. Additionally, the COPD Foundation has created the Bronchiectasis and NTM Research Registry, an American cohort containing more than 5,000 patients and data from 22 different sites, to answer some of the most important questions for clinicians and patients.
We have made significant progress in bronchiectasis research; however, there is still much to learn. Together, we must make a concerted effort to enroll patients in clinical trials. Doing so will allow us to define our epidemiologic profile more precisely and explore new treatments and airway clearance techniques.
AIRWAYS DISORDERS NETWORK
Bronchiectasis Section
For years, the noncystic fibrosis (CF) bronchiectasis community has been trying to organize to provide better care for more than half a million adults with bronchiectasis in the United States. Internationally, the Europeans created the European Bronchiectasis Registry, which has been a powerful tool including nearly 20,000 patients, to answer important epidemiologic and management questions. We must do more for the bronchiectasis community.
Clinicaltrials.gov indicates that there are 8 international phase 3 or 4 clinical trials that are currently enrolling; 3 of those have enrollment sites in the United States. One such study from University of North Carolina at Chapel Hill is looking at the use of nebulized hypertonic saline in patients with non-CF bronchiectasis to understand the effect it has on mucociliary clearance. Emory University is looking at the use of elexacaftor/tezacaftor/ivacaftor (Trikafta) in patients with non-CF bronchiectasis; these patients have only 1 targetable mutation and a phenotype that resembles CF. This 8-week, open-label, single-center study aims to measure both clinical and biomarker outcomes after treatment with Trikafta. Finally, a phase 3 trial out of Florida, the ICoN-1 study, is examining the efficacy and safety of inhaled clofazimine in the treatment of nontuberculous mycobacteria (NTM). This double-blind, randomized trial will look at culture conversion and quality of life measures. Additionally, the COPD Foundation has created the Bronchiectasis and NTM Research Registry, an American cohort containing more than 5,000 patients and data from 22 different sites, to answer some of the most important questions for clinicians and patients.
We have made significant progress in bronchiectasis research; however, there is still much to learn. Together, we must make a concerted effort to enroll patients in clinical trials. Doing so will allow us to define our epidemiologic profile more precisely and explore new treatments and airway clearance techniques.
AIRWAYS DISORDERS NETWORK
Bronchiectasis Section
For years, the noncystic fibrosis (CF) bronchiectasis community has been trying to organize to provide better care for more than half a million adults with bronchiectasis in the United States. Internationally, the Europeans created the European Bronchiectasis Registry, which has been a powerful tool including nearly 20,000 patients, to answer important epidemiologic and management questions. We must do more for the bronchiectasis community.
Clinicaltrials.gov indicates that there are 8 international phase 3 or 4 clinical trials that are currently enrolling; 3 of those have enrollment sites in the United States. One such study from University of North Carolina at Chapel Hill is looking at the use of nebulized hypertonic saline in patients with non-CF bronchiectasis to understand the effect it has on mucociliary clearance. Emory University is looking at the use of elexacaftor/tezacaftor/ivacaftor (Trikafta) in patients with non-CF bronchiectasis; these patients have only 1 targetable mutation and a phenotype that resembles CF. This 8-week, open-label, single-center study aims to measure both clinical and biomarker outcomes after treatment with Trikafta. Finally, a phase 3 trial out of Florida, the ICoN-1 study, is examining the efficacy and safety of inhaled clofazimine in the treatment of nontuberculous mycobacteria (NTM). This double-blind, randomized trial will look at culture conversion and quality of life measures. Additionally, the COPD Foundation has created the Bronchiectasis and NTM Research Registry, an American cohort containing more than 5,000 patients and data from 22 different sites, to answer some of the most important questions for clinicians and patients.
We have made significant progress in bronchiectasis research; however, there is still much to learn. Together, we must make a concerted effort to enroll patients in clinical trials. Doing so will allow us to define our epidemiologic profile more precisely and explore new treatments and airway clearance techniques.
The countdown to CHEST 2024 begins
As we find ourselves in September, I cannot help but dedicate my column to the upcoming CHEST Annual Meeting quickly approaching, October 6 to 9, in Boston.
If you haven’t yet been to a CHEST Annual Meeting, it’s an unmatched experience.
For those who have attended, there’s always something new to see. Every year is different, with the culture of the location guiding the way and new opportunities to network while engaging in activity. No matter how many times you have been, attending the CHEST Annual Meeting never gets old.
Leveraging CHEST 2024’s location, we’ll be hosting a Grand Rounds event days before the meeting starts with pulmonary and critical care medicine fellows from the regional Boston programs to learn from visiting CHEST leadership on a variety of influential topics. These fellowship programs held events like this prepandemic, so I’m truly excited we could help restart the tradition and give the local fellows an opportunity to interact with each other from both an academic and social perspective. Personally, I am very much looking forward to meeting and getting to know the fellows from the Boston area.
The meeting has a lot of notable opportunities lined up (see my official “President’s checklist”), including the third year of CHEST After Hours (Monday, October 7)—a unique storytelling event focusing on the humanities of medicine in partnership with The Nocturnists podcast. And for the first time in recent years, CHEST 2024 will feature a 5K run/walk (Tuesday, October 8) in support of CHEST philanthropy and its work to fuel breakthroughs, empower innovation, and drive toward a future where every patient’s well-being is safeguarded. I encourage you to register in advance of the meeting to secure your space and snag a souvenir T-shirt.
First thing Sunday morning (October 6), the meeting kicks off with the Opening Session where we will be celebrating the new fellows of the college (FCCP), honoring trailblazers in chest medicine, and welcoming this year’s keynote speaker.
This year’s keynote address will come from Vanessa Kerry, MD, who will speak on environmental issues and her work to raise awareness of the impact of climate change on health.
With so many things to look forward to, this meeting will be one to remember for all in attendance.
I look forward to seeing you in Boston,
Jack
As we find ourselves in September, I cannot help but dedicate my column to the upcoming CHEST Annual Meeting quickly approaching, October 6 to 9, in Boston.
If you haven’t yet been to a CHEST Annual Meeting, it’s an unmatched experience.
For those who have attended, there’s always something new to see. Every year is different, with the culture of the location guiding the way and new opportunities to network while engaging in activity. No matter how many times you have been, attending the CHEST Annual Meeting never gets old.
Leveraging CHEST 2024’s location, we’ll be hosting a Grand Rounds event days before the meeting starts with pulmonary and critical care medicine fellows from the regional Boston programs to learn from visiting CHEST leadership on a variety of influential topics. These fellowship programs held events like this prepandemic, so I’m truly excited we could help restart the tradition and give the local fellows an opportunity to interact with each other from both an academic and social perspective. Personally, I am very much looking forward to meeting and getting to know the fellows from the Boston area.
The meeting has a lot of notable opportunities lined up (see my official “President’s checklist”), including the third year of CHEST After Hours (Monday, October 7)—a unique storytelling event focusing on the humanities of medicine in partnership with The Nocturnists podcast. And for the first time in recent years, CHEST 2024 will feature a 5K run/walk (Tuesday, October 8) in support of CHEST philanthropy and its work to fuel breakthroughs, empower innovation, and drive toward a future where every patient’s well-being is safeguarded. I encourage you to register in advance of the meeting to secure your space and snag a souvenir T-shirt.
First thing Sunday morning (October 6), the meeting kicks off with the Opening Session where we will be celebrating the new fellows of the college (FCCP), honoring trailblazers in chest medicine, and welcoming this year’s keynote speaker.
This year’s keynote address will come from Vanessa Kerry, MD, who will speak on environmental issues and her work to raise awareness of the impact of climate change on health.
With so many things to look forward to, this meeting will be one to remember for all in attendance.
I look forward to seeing you in Boston,
Jack
As we find ourselves in September, I cannot help but dedicate my column to the upcoming CHEST Annual Meeting quickly approaching, October 6 to 9, in Boston.
If you haven’t yet been to a CHEST Annual Meeting, it’s an unmatched experience.
For those who have attended, there’s always something new to see. Every year is different, with the culture of the location guiding the way and new opportunities to network while engaging in activity. No matter how many times you have been, attending the CHEST Annual Meeting never gets old.
Leveraging CHEST 2024’s location, we’ll be hosting a Grand Rounds event days before the meeting starts with pulmonary and critical care medicine fellows from the regional Boston programs to learn from visiting CHEST leadership on a variety of influential topics. These fellowship programs held events like this prepandemic, so I’m truly excited we could help restart the tradition and give the local fellows an opportunity to interact with each other from both an academic and social perspective. Personally, I am very much looking forward to meeting and getting to know the fellows from the Boston area.
The meeting has a lot of notable opportunities lined up (see my official “President’s checklist”), including the third year of CHEST After Hours (Monday, October 7)—a unique storytelling event focusing on the humanities of medicine in partnership with The Nocturnists podcast. And for the first time in recent years, CHEST 2024 will feature a 5K run/walk (Tuesday, October 8) in support of CHEST philanthropy and its work to fuel breakthroughs, empower innovation, and drive toward a future where every patient’s well-being is safeguarded. I encourage you to register in advance of the meeting to secure your space and snag a souvenir T-shirt.
First thing Sunday morning (October 6), the meeting kicks off with the Opening Session where we will be celebrating the new fellows of the college (FCCP), honoring trailblazers in chest medicine, and welcoming this year’s keynote speaker.
This year’s keynote address will come from Vanessa Kerry, MD, who will speak on environmental issues and her work to raise awareness of the impact of climate change on health.
With so many things to look forward to, this meeting will be one to remember for all in attendance.
I look forward to seeing you in Boston,
Jack
Top reads from the CHEST journal portfolio
Covering the frailty scale in ILD, diagnosis of peripheral pulmonary nodules, and platelet mitochondrial function in sepsis.
Journal CHEST®
By Guler, MD, and colleagues
Life expectancy is a very important factor for patients with interstitial lung disease (ILD) and their caregivers. The discussion surrounding prognosis is often wrought with uncertainty and is inherently painful for both patients and clinicians when faced with nonmodifiable traits. This study illustrates the significance of employing a method that succinctly and systematically communicates the degree of functional impairment in patients with fibrotic lung disease. The authors have highlighted the importance of identifying and improving health factors associated with frailty to enhance the survival and quality of life of patients with chronic noncurable fibrotic lung disease. It also presents hope that interventions aimed at improving functional capacity may improve frailty and thus modify prognosis. In the future, longitudinal trends of frailty assessments following interventions aimed at improving both exercise and functional capacity, like pulmonary rehab, should be explored.
– Commentary by Priya Balakrishnan, MD, MS, FCCP, Member of the CHEST Physician Editorial Board
CHEST® Pulmonary
By Michael V. Brown, MD, and colleagues
Brown and colleagues provide a systemic review and meta-analysis of the diagnostic yield of cone beam computed tomography (CBCT) scan combined with radial-endobronchial ultrasound (r-EBUS) for the diagnosis of peripheral pulmonary nodules. They included 14 studies (865 patients with 882 lesions) with pooled diagnostic yield from CBCT scan and r-EBUS for peripheral pulmonary nodules of 80% (95% CI, 76% to 84%) with complication rates of 2.01% for pneumothorax and 1.08% for bleeding. Amongst the studies selected, confounders (including study design, definition of diagnostic yield, use of ROSE, additional equipment, etc) existed. The important takeaway is that 3D imaging guidance with CBCT scan can corroborate “tool in lesion” and thus potentially improve the outcomes of the different bronchoscopic modalities utilized to diagnose peripheral pulmonary nodules. Future prospective investigations with less heterogeneity in study design and outcomes, as well as comparison with newer technologies such as robotic bronchoscopy, are necessary to corroborate these findings.
– Commentary by Saadia A. Faiz, MD, FCCP, Member of the CHEST Physician Editorial Board
CHEST® Critical Care
Platelet Bioenergetics and Associations With Delirium and Coma in Patients With Sepsis
By Chukwudi A. Onyemekwu, DO, and colleagues
The study by Onyemekwu and colleagues explores the link between platelet mitochondrial function and acute brain dysfunction (delirium and coma) in patients with sepsis. The investigators measured various parameters of platelet mitochondrial respiratory bioenergetics and found that increased spare respiratory capacity was significantly associated with coma but not delirium. These findings suggest that systemic mitochondrial function could influence brain health and indicate a potential link between mitochondrial bioenergetics and coma during sepsis. The study did not find a significant association between platelet bioenergetics and delirium, suggesting that coma and delirium may have different underlying pathophysiologic mechanisms. We must interpret the results with caution, as the associations identified in this observational study do not prove causation. It is possible that the changes seen in platelet mitochondria may be a result of coma rather than a mechanism. Nonetheless, the study provides a foundation for future research to explore the mechanistic role of mitochondria in acute brain dysfunction during sepsis and the potential for developing mitochondrial-targeted therapies as a possible treatment approach for patients with sepsis-induced coma.
– Commentary by Angel O. Coz, MD, FCCP, Editor in Chief of CHEST Physician
Covering the frailty scale in ILD, diagnosis of peripheral pulmonary nodules, and platelet mitochondrial function in sepsis.
Covering the frailty scale in ILD, diagnosis of peripheral pulmonary nodules, and platelet mitochondrial function in sepsis.
Journal CHEST®
By Guler, MD, and colleagues
Life expectancy is a very important factor for patients with interstitial lung disease (ILD) and their caregivers. The discussion surrounding prognosis is often wrought with uncertainty and is inherently painful for both patients and clinicians when faced with nonmodifiable traits. This study illustrates the significance of employing a method that succinctly and systematically communicates the degree of functional impairment in patients with fibrotic lung disease. The authors have highlighted the importance of identifying and improving health factors associated with frailty to enhance the survival and quality of life of patients with chronic noncurable fibrotic lung disease. It also presents hope that interventions aimed at improving functional capacity may improve frailty and thus modify prognosis. In the future, longitudinal trends of frailty assessments following interventions aimed at improving both exercise and functional capacity, like pulmonary rehab, should be explored.
– Commentary by Priya Balakrishnan, MD, MS, FCCP, Member of the CHEST Physician Editorial Board
CHEST® Pulmonary
By Michael V. Brown, MD, and colleagues
Brown and colleagues provide a systemic review and meta-analysis of the diagnostic yield of cone beam computed tomography (CBCT) scan combined with radial-endobronchial ultrasound (r-EBUS) for the diagnosis of peripheral pulmonary nodules. They included 14 studies (865 patients with 882 lesions) with pooled diagnostic yield from CBCT scan and r-EBUS for peripheral pulmonary nodules of 80% (95% CI, 76% to 84%) with complication rates of 2.01% for pneumothorax and 1.08% for bleeding. Amongst the studies selected, confounders (including study design, definition of diagnostic yield, use of ROSE, additional equipment, etc) existed. The important takeaway is that 3D imaging guidance with CBCT scan can corroborate “tool in lesion” and thus potentially improve the outcomes of the different bronchoscopic modalities utilized to diagnose peripheral pulmonary nodules. Future prospective investigations with less heterogeneity in study design and outcomes, as well as comparison with newer technologies such as robotic bronchoscopy, are necessary to corroborate these findings.
– Commentary by Saadia A. Faiz, MD, FCCP, Member of the CHEST Physician Editorial Board
CHEST® Critical Care
Platelet Bioenergetics and Associations With Delirium and Coma in Patients With Sepsis
By Chukwudi A. Onyemekwu, DO, and colleagues
The study by Onyemekwu and colleagues explores the link between platelet mitochondrial function and acute brain dysfunction (delirium and coma) in patients with sepsis. The investigators measured various parameters of platelet mitochondrial respiratory bioenergetics and found that increased spare respiratory capacity was significantly associated with coma but not delirium. These findings suggest that systemic mitochondrial function could influence brain health and indicate a potential link between mitochondrial bioenergetics and coma during sepsis. The study did not find a significant association between platelet bioenergetics and delirium, suggesting that coma and delirium may have different underlying pathophysiologic mechanisms. We must interpret the results with caution, as the associations identified in this observational study do not prove causation. It is possible that the changes seen in platelet mitochondria may be a result of coma rather than a mechanism. Nonetheless, the study provides a foundation for future research to explore the mechanistic role of mitochondria in acute brain dysfunction during sepsis and the potential for developing mitochondrial-targeted therapies as a possible treatment approach for patients with sepsis-induced coma.
– Commentary by Angel O. Coz, MD, FCCP, Editor in Chief of CHEST Physician
Journal CHEST®
By Guler, MD, and colleagues
Life expectancy is a very important factor for patients with interstitial lung disease (ILD) and their caregivers. The discussion surrounding prognosis is often wrought with uncertainty and is inherently painful for both patients and clinicians when faced with nonmodifiable traits. This study illustrates the significance of employing a method that succinctly and systematically communicates the degree of functional impairment in patients with fibrotic lung disease. The authors have highlighted the importance of identifying and improving health factors associated with frailty to enhance the survival and quality of life of patients with chronic noncurable fibrotic lung disease. It also presents hope that interventions aimed at improving functional capacity may improve frailty and thus modify prognosis. In the future, longitudinal trends of frailty assessments following interventions aimed at improving both exercise and functional capacity, like pulmonary rehab, should be explored.
– Commentary by Priya Balakrishnan, MD, MS, FCCP, Member of the CHEST Physician Editorial Board
CHEST® Pulmonary
By Michael V. Brown, MD, and colleagues
Brown and colleagues provide a systemic review and meta-analysis of the diagnostic yield of cone beam computed tomography (CBCT) scan combined with radial-endobronchial ultrasound (r-EBUS) for the diagnosis of peripheral pulmonary nodules. They included 14 studies (865 patients with 882 lesions) with pooled diagnostic yield from CBCT scan and r-EBUS for peripheral pulmonary nodules of 80% (95% CI, 76% to 84%) with complication rates of 2.01% for pneumothorax and 1.08% for bleeding. Amongst the studies selected, confounders (including study design, definition of diagnostic yield, use of ROSE, additional equipment, etc) existed. The important takeaway is that 3D imaging guidance with CBCT scan can corroborate “tool in lesion” and thus potentially improve the outcomes of the different bronchoscopic modalities utilized to diagnose peripheral pulmonary nodules. Future prospective investigations with less heterogeneity in study design and outcomes, as well as comparison with newer technologies such as robotic bronchoscopy, are necessary to corroborate these findings.
– Commentary by Saadia A. Faiz, MD, FCCP, Member of the CHEST Physician Editorial Board
CHEST® Critical Care
Platelet Bioenergetics and Associations With Delirium and Coma in Patients With Sepsis
By Chukwudi A. Onyemekwu, DO, and colleagues
The study by Onyemekwu and colleagues explores the link between platelet mitochondrial function and acute brain dysfunction (delirium and coma) in patients with sepsis. The investigators measured various parameters of platelet mitochondrial respiratory bioenergetics and found that increased spare respiratory capacity was significantly associated with coma but not delirium. These findings suggest that systemic mitochondrial function could influence brain health and indicate a potential link between mitochondrial bioenergetics and coma during sepsis. The study did not find a significant association between platelet bioenergetics and delirium, suggesting that coma and delirium may have different underlying pathophysiologic mechanisms. We must interpret the results with caution, as the associations identified in this observational study do not prove causation. It is possible that the changes seen in platelet mitochondria may be a result of coma rather than a mechanism. Nonetheless, the study provides a foundation for future research to explore the mechanistic role of mitochondria in acute brain dysfunction during sepsis and the potential for developing mitochondrial-targeted therapies as a possible treatment approach for patients with sepsis-induced coma.
– Commentary by Angel O. Coz, MD, FCCP, Editor in Chief of CHEST Physician
The language of AI and its applications in health care
AI is a group of nonhuman techniques that utilize automated learning methods to extract information from datasets through generalization, classification, prediction, and association. In other words, AI is the simulation of human intelligence processes by machines. The branches of AI include natural language processing, speech recognition, machine vision, and expert systems. AI can make clinical care more efficient; however, many find its confusing terminology to be a barrier.1 This article provides concise definitions of AI terms and is intended to help physicians better understand how AI methods can be applied to clinical care. The clinical application of natural language processing and machine vision applications are more clinically intuitive than the roles of machine learning algorithms.
Machine learning and algorithms
Machine learning is a branch of AI that uses data and algorithms to mimic human reasoning through classification, pattern recognition, and prediction. Supervised and unsupervised machine-learning algorithms can analyze data and recognize undetected associations and relationships.
Supervised learning involves training models to make predictions using data sets that have correct outcome parameters called labels using predictive fields called features. Machine learning uses iterative analysis including random forest, decision tree, and gradient boosting methods that minimize predictive error metrics (see Table 1). This approach is widely used to improve diagnoses, predict disease progression or exacerbation, and personalize treatment plan modifications.
Supervised machine learning methods can be particularly effective for processing large volumes of medical information to identify patterns and make accurate predictions. In contrast, unsupervised learning techniques can analyze unlabeled data and help clinicians uncover hidden patterns or undetected groupings. Techniques including clustering, exploratory analysis, and anomaly detection are common applications. Both of these machine-learning approaches can be used to extract novel and helpful insights.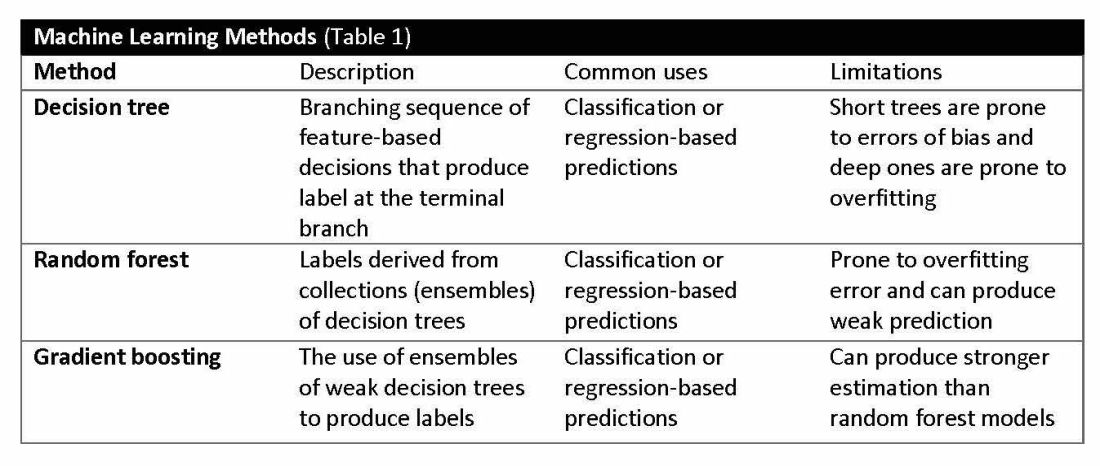
The utility of machine learning analyses depends on the size and accuracy of the available datasets. Small datasets can limit usability, while large datasets require substantial computational power. Predictive models are generated using training datasets and evaluated using separate evaluation datasets. Deep learning models, a subset of machine learning, can automatically readjust themselves to maintain or improve accuracy when analyzing new observations that include accurate labels.
Challenges of algorithms and calibration
Machine learning algorithms vary in complexity and accuracy. For example, a simple logistic regression model using time, date, latitude, and indoor/outdoor location can accurately recommend sunscreen application. This model identifies when solar radiation is high enough to warrant sunscreen use, avoiding unnecessary recommendations during nighttime hours or indoor locations. A more complex model might suffer from model overfitting and inappropriately suggest sunscreen before a tanning salon visit.
Complex machine learning models, like support vector machine (SVM) and decision tree methods, are useful when many features have predictive power. SVMs are useful for small but complex datasets. Features are manipulated in a multidimensional space to maximize the “margins” separating 2 groups. Decision tree analyses are useful when more than 2 groups are being analyzed. SVM and decision tree models can also lose accuracy by data overfitting.
Consider the development of an SVM analysis to predict whether an individual is a fellow or a senior faculty member. One could use high gray hair density feature values to identify senior faculty. When this algorithm is applied to an individual with alopecia, no amount of model adjustment can achieve high levels of discrimination because no hair is present. Rather than overfitting the model by adding more nonpredictive features, individuals with alopecia are analyzed by their own algorithm (tree) that uses the skin wrinkle/solar damage rather than the gray hair density feature.
Decision tree ensemble algorithms like random forest and gradient boosting use feature-based decision trees to process and classify data. Random forests are robust, scalable, and versatile, providing classifications and predictions while protecting against inaccurate data and outliers and have the advantage of being able to handle both categorical and continuous features. Gradient boosting, which uses an ensemble of weak decision trees, often outperforms random forests when individual trees perform only slightly better than random chance. This method incrementally builds the model by optimizing the residual errors of previous trees, leading to more accurate predictions.
In practice, gradient boosting can be used to fine-tune diagnostic models, improving their precision and reliability. A recent example of how gradient boosting of random forest predictions yielded highly accurate predictions for unplanned vasopressor initiation and intubation events 2 to 4 hours before an ICU adult became unstable.2
Assessing the accuracy of algorithms
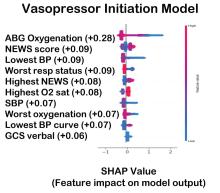
The value of the data set is directly related to the accuracy of its labels. Traditional methods that measure model performance, such as sensitivity, specificity, and predictive values (PPV and NPV), have important limitations. They provide little insight into how a complex model made its prediction. Understanding which individual features drive model accuracy is key to fostering trust in model predictions. This can be done by comparing model output with and without including individual features. The results of all possible combinations are aggregated according to feature importance, which is summarized in the Shapley value for each model feature. Higher values indicate greater relative importance. SHAP plots help identify how much and how often specific features change the model output, presenting values of individual model estimates with and without a specific feature (see Figure 1).
Promoting AI use
AI and machine learning algorithms are coming to patient care. Understanding the language of AI helps caregivers integrate these tools into their practices. The science of AI faces serious challenges. Algorithms must be recalibrated to keep pace as therapies advance, disease prevalence changes, and our population ages. AI must address new challenges as they confront those suffering from respiratory diseases. This resource encourages clinicians with novel approaches by using AI methodologies to advance their development. We can better address future health care needs by promoting the equitable use of AI technologies, especially among socially disadvantaged developers.
References
1. Lilly CM, Soni AV, Dunlap D, et al. Advancing point of care testing by application of machine learning techniques and artificial intelligence. Chest. 2024 (in press).
2. Lilly CM, Kirk D, Pessach IM, et al. Application of machine learning models to biomedical and information system signals from critically ill adults. Chest. 2024;165(5):1139-1148.
AI is a group of nonhuman techniques that utilize automated learning methods to extract information from datasets through generalization, classification, prediction, and association. In other words, AI is the simulation of human intelligence processes by machines. The branches of AI include natural language processing, speech recognition, machine vision, and expert systems. AI can make clinical care more efficient; however, many find its confusing terminology to be a barrier.1 This article provides concise definitions of AI terms and is intended to help physicians better understand how AI methods can be applied to clinical care. The clinical application of natural language processing and machine vision applications are more clinically intuitive than the roles of machine learning algorithms.
Machine learning and algorithms
Machine learning is a branch of AI that uses data and algorithms to mimic human reasoning through classification, pattern recognition, and prediction. Supervised and unsupervised machine-learning algorithms can analyze data and recognize undetected associations and relationships.
Supervised learning involves training models to make predictions using data sets that have correct outcome parameters called labels using predictive fields called features. Machine learning uses iterative analysis including random forest, decision tree, and gradient boosting methods that minimize predictive error metrics (see Table 1). This approach is widely used to improve diagnoses, predict disease progression or exacerbation, and personalize treatment plan modifications.
Supervised machine learning methods can be particularly effective for processing large volumes of medical information to identify patterns and make accurate predictions. In contrast, unsupervised learning techniques can analyze unlabeled data and help clinicians uncover hidden patterns or undetected groupings. Techniques including clustering, exploratory analysis, and anomaly detection are common applications. Both of these machine-learning approaches can be used to extract novel and helpful insights.
The utility of machine learning analyses depends on the size and accuracy of the available datasets. Small datasets can limit usability, while large datasets require substantial computational power. Predictive models are generated using training datasets and evaluated using separate evaluation datasets. Deep learning models, a subset of machine learning, can automatically readjust themselves to maintain or improve accuracy when analyzing new observations that include accurate labels.
Challenges of algorithms and calibration
Machine learning algorithms vary in complexity and accuracy. For example, a simple logistic regression model using time, date, latitude, and indoor/outdoor location can accurately recommend sunscreen application. This model identifies when solar radiation is high enough to warrant sunscreen use, avoiding unnecessary recommendations during nighttime hours or indoor locations. A more complex model might suffer from model overfitting and inappropriately suggest sunscreen before a tanning salon visit.
Complex machine learning models, like support vector machine (SVM) and decision tree methods, are useful when many features have predictive power. SVMs are useful for small but complex datasets. Features are manipulated in a multidimensional space to maximize the “margins” separating 2 groups. Decision tree analyses are useful when more than 2 groups are being analyzed. SVM and decision tree models can also lose accuracy by data overfitting.
Consider the development of an SVM analysis to predict whether an individual is a fellow or a senior faculty member. One could use high gray hair density feature values to identify senior faculty. When this algorithm is applied to an individual with alopecia, no amount of model adjustment can achieve high levels of discrimination because no hair is present. Rather than overfitting the model by adding more nonpredictive features, individuals with alopecia are analyzed by their own algorithm (tree) that uses the skin wrinkle/solar damage rather than the gray hair density feature.
Decision tree ensemble algorithms like random forest and gradient boosting use feature-based decision trees to process and classify data. Random forests are robust, scalable, and versatile, providing classifications and predictions while protecting against inaccurate data and outliers and have the advantage of being able to handle both categorical and continuous features. Gradient boosting, which uses an ensemble of weak decision trees, often outperforms random forests when individual trees perform only slightly better than random chance. This method incrementally builds the model by optimizing the residual errors of previous trees, leading to more accurate predictions.
In practice, gradient boosting can be used to fine-tune diagnostic models, improving their precision and reliability. A recent example of how gradient boosting of random forest predictions yielded highly accurate predictions for unplanned vasopressor initiation and intubation events 2 to 4 hours before an ICU adult became unstable.2
Assessing the accuracy of algorithms
The value of the data set is directly related to the accuracy of its labels. Traditional methods that measure model performance, such as sensitivity, specificity, and predictive values (PPV and NPV), have important limitations. They provide little insight into how a complex model made its prediction. Understanding which individual features drive model accuracy is key to fostering trust in model predictions. This can be done by comparing model output with and without including individual features. The results of all possible combinations are aggregated according to feature importance, which is summarized in the Shapley value for each model feature. Higher values indicate greater relative importance. SHAP plots help identify how much and how often specific features change the model output, presenting values of individual model estimates with and without a specific feature (see Figure 1).
Promoting AI use
AI and machine learning algorithms are coming to patient care. Understanding the language of AI helps caregivers integrate these tools into their practices. The science of AI faces serious challenges. Algorithms must be recalibrated to keep pace as therapies advance, disease prevalence changes, and our population ages. AI must address new challenges as they confront those suffering from respiratory diseases. This resource encourages clinicians with novel approaches by using AI methodologies to advance their development. We can better address future health care needs by promoting the equitable use of AI technologies, especially among socially disadvantaged developers.
References
1. Lilly CM, Soni AV, Dunlap D, et al. Advancing point of care testing by application of machine learning techniques and artificial intelligence. Chest. 2024 (in press).
2. Lilly CM, Kirk D, Pessach IM, et al. Application of machine learning models to biomedical and information system signals from critically ill adults. Chest. 2024;165(5):1139-1148.
AI is a group of nonhuman techniques that utilize automated learning methods to extract information from datasets through generalization, classification, prediction, and association. In other words, AI is the simulation of human intelligence processes by machines. The branches of AI include natural language processing, speech recognition, machine vision, and expert systems. AI can make clinical care more efficient; however, many find its confusing terminology to be a barrier.1 This article provides concise definitions of AI terms and is intended to help physicians better understand how AI methods can be applied to clinical care. The clinical application of natural language processing and machine vision applications are more clinically intuitive than the roles of machine learning algorithms.
Machine learning and algorithms
Machine learning is a branch of AI that uses data and algorithms to mimic human reasoning through classification, pattern recognition, and prediction. Supervised and unsupervised machine-learning algorithms can analyze data and recognize undetected associations and relationships.
Supervised learning involves training models to make predictions using data sets that have correct outcome parameters called labels using predictive fields called features. Machine learning uses iterative analysis including random forest, decision tree, and gradient boosting methods that minimize predictive error metrics (see Table 1). This approach is widely used to improve diagnoses, predict disease progression or exacerbation, and personalize treatment plan modifications.
Supervised machine learning methods can be particularly effective for processing large volumes of medical information to identify patterns and make accurate predictions. In contrast, unsupervised learning techniques can analyze unlabeled data and help clinicians uncover hidden patterns or undetected groupings. Techniques including clustering, exploratory analysis, and anomaly detection are common applications. Both of these machine-learning approaches can be used to extract novel and helpful insights.
The utility of machine learning analyses depends on the size and accuracy of the available datasets. Small datasets can limit usability, while large datasets require substantial computational power. Predictive models are generated using training datasets and evaluated using separate evaluation datasets. Deep learning models, a subset of machine learning, can automatically readjust themselves to maintain or improve accuracy when analyzing new observations that include accurate labels.
Challenges of algorithms and calibration
Machine learning algorithms vary in complexity and accuracy. For example, a simple logistic regression model using time, date, latitude, and indoor/outdoor location can accurately recommend sunscreen application. This model identifies when solar radiation is high enough to warrant sunscreen use, avoiding unnecessary recommendations during nighttime hours or indoor locations. A more complex model might suffer from model overfitting and inappropriately suggest sunscreen before a tanning salon visit.
Complex machine learning models, like support vector machine (SVM) and decision tree methods, are useful when many features have predictive power. SVMs are useful for small but complex datasets. Features are manipulated in a multidimensional space to maximize the “margins” separating 2 groups. Decision tree analyses are useful when more than 2 groups are being analyzed. SVM and decision tree models can also lose accuracy by data overfitting.
Consider the development of an SVM analysis to predict whether an individual is a fellow or a senior faculty member. One could use high gray hair density feature values to identify senior faculty. When this algorithm is applied to an individual with alopecia, no amount of model adjustment can achieve high levels of discrimination because no hair is present. Rather than overfitting the model by adding more nonpredictive features, individuals with alopecia are analyzed by their own algorithm (tree) that uses the skin wrinkle/solar damage rather than the gray hair density feature.
Decision tree ensemble algorithms like random forest and gradient boosting use feature-based decision trees to process and classify data. Random forests are robust, scalable, and versatile, providing classifications and predictions while protecting against inaccurate data and outliers and have the advantage of being able to handle both categorical and continuous features. Gradient boosting, which uses an ensemble of weak decision trees, often outperforms random forests when individual trees perform only slightly better than random chance. This method incrementally builds the model by optimizing the residual errors of previous trees, leading to more accurate predictions.
In practice, gradient boosting can be used to fine-tune diagnostic models, improving their precision and reliability. A recent example of how gradient boosting of random forest predictions yielded highly accurate predictions for unplanned vasopressor initiation and intubation events 2 to 4 hours before an ICU adult became unstable.2
Assessing the accuracy of algorithms
The value of the data set is directly related to the accuracy of its labels. Traditional methods that measure model performance, such as sensitivity, specificity, and predictive values (PPV and NPV), have important limitations. They provide little insight into how a complex model made its prediction. Understanding which individual features drive model accuracy is key to fostering trust in model predictions. This can be done by comparing model output with and without including individual features. The results of all possible combinations are aggregated according to feature importance, which is summarized in the Shapley value for each model feature. Higher values indicate greater relative importance. SHAP plots help identify how much and how often specific features change the model output, presenting values of individual model estimates with and without a specific feature (see Figure 1).
Promoting AI use
AI and machine learning algorithms are coming to patient care. Understanding the language of AI helps caregivers integrate these tools into their practices. The science of AI faces serious challenges. Algorithms must be recalibrated to keep pace as therapies advance, disease prevalence changes, and our population ages. AI must address new challenges as they confront those suffering from respiratory diseases. This resource encourages clinicians with novel approaches by using AI methodologies to advance their development. We can better address future health care needs by promoting the equitable use of AI technologies, especially among socially disadvantaged developers.
References
1. Lilly CM, Soni AV, Dunlap D, et al. Advancing point of care testing by application of machine learning techniques and artificial intelligence. Chest. 2024 (in press).
2. Lilly CM, Kirk D, Pessach IM, et al. Application of machine learning models to biomedical and information system signals from critically ill adults. Chest. 2024;165(5):1139-1148.
CHEST releases new guideline on management of central airway obstruction
CHEST recently released a new clinical guideline on central airway obstruction (CAO).
“Central airway obstruction is associated with a poor prognosis, and the management of CAO is highly variable dependent on the provider expertise and local resources. By releasing this guideline, the panel hopes to standardize the definition of CAO and provide guidance for the management of patients to optimize care and improve outcomes,” said Kamran Mahmood, MD, MPH, FCCP, lead author on the guideline. “The guideline recommendations are developed using GRADE methodology and based on thorough evidence review and expert input. But the quality of overall evidence is very low, and the panel calls for well-designed studies and randomized controlled trials for the management of CAO.”
CAO can be caused by a variety of malignant and nonmalignant disorders, and multiple specialists may be involved in the care, including pulmonologists, interventional pulmonologists, radiologists, anesthesiologists, oncologists, radiation oncologists, thoracic surgeons and otolaryngologists, etc. The panel recommends shared decision-making with the patients and a multidisciplinary approach to manage CAO.
Below, you’ll find flowcharts outlining the suggested treatments for patients with a malignant or nonmalignant CAO.
Visit www.chestnet.org/CAO-guideline to download the flowcharts and access the complete guideline.
CHEST recently released a new clinical guideline on central airway obstruction (CAO).
“Central airway obstruction is associated with a poor prognosis, and the management of CAO is highly variable dependent on the provider expertise and local resources. By releasing this guideline, the panel hopes to standardize the definition of CAO and provide guidance for the management of patients to optimize care and improve outcomes,” said Kamran Mahmood, MD, MPH, FCCP, lead author on the guideline. “The guideline recommendations are developed using GRADE methodology and based on thorough evidence review and expert input. But the quality of overall evidence is very low, and the panel calls for well-designed studies and randomized controlled trials for the management of CAO.”
CAO can be caused by a variety of malignant and nonmalignant disorders, and multiple specialists may be involved in the care, including pulmonologists, interventional pulmonologists, radiologists, anesthesiologists, oncologists, radiation oncologists, thoracic surgeons and otolaryngologists, etc. The panel recommends shared decision-making with the patients and a multidisciplinary approach to manage CAO.
Below, you’ll find flowcharts outlining the suggested treatments for patients with a malignant or nonmalignant CAO.

Visit www.chestnet.org/CAO-guideline to download the flowcharts and access the complete guideline.
CHEST recently released a new clinical guideline on central airway obstruction (CAO).
“Central airway obstruction is associated with a poor prognosis, and the management of CAO is highly variable dependent on the provider expertise and local resources. By releasing this guideline, the panel hopes to standardize the definition of CAO and provide guidance for the management of patients to optimize care and improve outcomes,” said Kamran Mahmood, MD, MPH, FCCP, lead author on the guideline. “The guideline recommendations are developed using GRADE methodology and based on thorough evidence review and expert input. But the quality of overall evidence is very low, and the panel calls for well-designed studies and randomized controlled trials for the management of CAO.”
CAO can be caused by a variety of malignant and nonmalignant disorders, and multiple specialists may be involved in the care, including pulmonologists, interventional pulmonologists, radiologists, anesthesiologists, oncologists, radiation oncologists, thoracic surgeons and otolaryngologists, etc. The panel recommends shared decision-making with the patients and a multidisciplinary approach to manage CAO.
Below, you’ll find flowcharts outlining the suggested treatments for patients with a malignant or nonmalignant CAO.

Visit www.chestnet.org/CAO-guideline to download the flowcharts and access the complete guideline.