User login
Improving Colorectal Cancer Screening via Mailed Fecal Immunochemical Testing in a Veterans Affairs Health System
Colorectal cancer (CRC) is among the most common cancers and causes of cancer-related deaths in the United States.1 Reflective of a nationwide trend, CRC screening rates at the Veterans Affairs Connecticut Healthcare System (VACHS) decreased during the COVID-19 pandemic.2-5 Contributing factors to this decrease included cancellations of elective colonoscopies during the initial phase of the pandemic and concurrent turnover of endoscopists. In 2021, the US Preventive Services Task Force lowered the recommended initial CRC screening age from 50 years to 45 years, further increasing the backlog of unscreened patients.6
Fecal immunochemical testing (FIT) is a noninvasive screening method in which antibodies are used to detect hemoglobin in the stool. The sensitivity and specificity of 1-time FIT are 79% to 80% and 94%, respectively, for the detection of CRC, with sensitivity improving with successive testing.7,8 Annual FIT is recognized as a tier 1 preferred screening method by the US Multi-Society Task Force on Colorectal Cancer.7,9 Programs that mail FIT kits to eligible patients outside of physician visits have been successfully implemented in health care systems.10,11
The VACHS designed and implemented a mailed FIT program using existing infrastructure and staffing.
Program Description
A team of local stakeholders comprised of VACHS leadership, primary care, nursing, and gastroenterology staff, as well as representatives from laboratory, informatics, mail services, and group practice management, was established to execute the project. The team met monthly to plan the project.
The team developed a dataset consisting of patients aged 45 to 75 years who were at average risk for CRC and due for CRC screening. Patients were defined as due for CRC screening if they had not had a colonoscopy in the previous 9 years or a FIT or fecal occult blood test in the previous 11 months. Average risk for CRC was defined by excluding patients with associated diagnosis codes for CRC, colectomy, inflammatory bowel disease, and anemia. The program also excluded patients with diagnosis codes associated with dementia, deferring discussions about cancer screening to their primary care practitioners (PCPs). Patients with invalid mailing addresses were also excluded, as well as those whose PCPs had indicated in the electronic health record that the patient received CRC screening outside the US Department of Veterans Affairs (VA) system.
Letter Templates
Two patient letter electronic health record templates were developed. The first was a primer letter, which was mailed to patients 2 to 3 weeks before the mailed FIT kit as an introduction to the program.12 The purpose of the primer letter was to give advance notice to patients that they could expect a FIT kit to arrive in the mail. The goal was to prepare patients to complete FIT when the kit arrived and prompt them to call the VA to opt out of the mailed FIT program if they were up to date with CRC screening or if they had a condition which made them at high risk for CRC.
The second FIT letter arrived with the FIT kit, introduced FIT and described the importance of CRC screening. The letter detailed instructions for completing FIT and automatically created a FIT order. It also included a list of common conditions that may exclude patients, with a recommendation for patients to contact their medical team if they felt they were not candidates for FIT.
Staff Education
A previous VACHS pilot project demonstrated the success of a mailed FIT program to increase FIT use. Implemented as part of the pilot program, staff education consisted of a session for clinicians about the role of FIT in CRC screening and an all-staff education session. An additional education session about CRC and FIT for all staff was repeated with the program launch.
Program Launch
The mailed FIT program was introduced during a VACHS primary care all-staff meeting. After the meeting, each patient aligned care team (PACT) received an encrypted email that included a list of the patients on their team who were candidates for the program, a patient-facing FIT instruction sheet, detailed instructions on how to send the FIT primer letter, and a FIT package consisting of the labeled FIT kit, FIT letter, and patient instruction sheet. A reminder letter was sent to each patient 3 weeks after the FIT package was mailed. The patient lists were populated into a shared, encrypted Microsoft Teams folder that was edited in real time by PACT teams and viewed by VACHS leadership to track progress.
Program Metrics
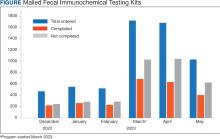
At program launch, the VACHS had 4642 patients due for CRC screening who were eligible for the mailed FIT program. On March 7, 2023, the data consisting of FIT tests ordered between December 2022 and May 2023—3 months before and after the launch of the program—were reviewed and categorized. In the 3 months before program launch, 1528 FIT were ordered and 714 were returned (46.7%). In the 3 months after the launch of the program, 4383 FIT were ordered and 1712 were returned (39.1%) (Figure). Test orders increased 287% from the preintervention to the postintervention period. The mean (SD) number of monthly FIT tests prelaunch was 509 (32.7), which increased to 1461 (331.6) postlaunch.
At the VACHS, 61.4% of patients aged 45 to 75 years were up to date with CRC screening before the program launch. In the 3 months after program launch, the rate increased to 63.8% among patients aged 45 to 75 years, the highest rate in our Veterans Integrated Services Network and exceeding the VA national average CRC screening rate, according to unpublished VA Monthly Management Report data.
In the 3 months following the program launch, 139 FIT kits tested positive for potential CRC. Of these, 79 (56.8%) patients had completed a diagnostic colonoscopy. PACT PCPs and nurses received reports on patients with positive FIT tests and those with no colonoscopy scheduled or completed and were asked to follow up.
Discussion
Through a proactive, population-based CRC screening program centered on mailed FIT kits outside of the traditional patient visit, the VACHS increased the use of FIT and rates of CRC screening. The numbers of FIT kits ordered and completed substantially increased in the 3 months after program launch.
Compared to mailed FIT programs described in the literature that rely on centralized processes in that a separate team operates the mailed FIT program for the entire organization, this program used existing PACT infrastructure and staff.10,11 This strategy allowed VACHS to design and implement the program in several months. Not needing to hire new staff or create a central team for the sole purpose of implementing the program allowed us to save on any organizational funding and efforts that would have accompanied the additional staff. The program described in this article may be more attainable for primary care practices or smaller health systems that do not have the capacity for the creation of a centralized process.
Limitations
Although the total number of FIT completions substantially increased during the program, the rate of FIT completion during the mailed FIT program was lower than the rate of completion prior to program launch. This decreased rate of FIT kit completion may be related to separation from a patient visit and potential loss of real-time education with a clinician. The program’s decentralized design increased the existing workload for primary care staff, and as a result, consideration must be given to local staffing levels. Additionally, the report of eligible patients depended on diagnosis codes and may have captured patients with higher-than-average risk of CRC, such as patients with prior history of adenomatous polyps, family history of CRC, or other medical or genetic conditions. We attempted to mitigate this by including a list of conditions that would exclude patients from FIT eligibility in the FIT letter and giving them the option to opt out.
Conclusions
CRC screening rates improved following implementation of a primary care team-centered quality improvement process to proactively identify patients appropriate for FIT and mail them FIT kits. This project highlights that population-health interventions around CRC screening via use of FIT can be successful within a primary care patient-centered medical home model, considering the increases in both CRC screening rates and increase in FIT tests ordered.
1. American Cancer Society. Key statistics for colorectal cancer. Revised January 29, 2024. Accessed June 11, 2024. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
2. Chen RC, Haynes K, Du S, Barron J, Katz AJ. Association of cancer screening deficit in the United States with the COVID-19 pandemic. JAMA Oncol. 2021;7(6):878-884. doi:10.1001/jamaoncol.2021.0884
3. Mazidimoradi A, Tiznobaik A, Salehiniya H. Impact of the COVID-19 pandemic on colorectal cancer screening: a systematic review. J Gastrointest Cancer. 2022;53(3):730-744. doi:10.1007/s12029-021-00679-x
4. Adams MA, Kurlander JE, Gao Y, Yankey N, Saini SD. Impact of coronavirus disease 2019 on screening colonoscopy utilization in a large integrated health system. Gastroenterology. 2022;162(7):2098-2100.e2. doi:10.1053/j.gastro.2022.02.034
5. Sundaram S, Olson S, Sharma P, Rajendra S. A review of the impact of the COVID-19 pandemic on colorectal cancer screening: implications and solutions. Pathogens. 2021;10(11):558. doi:10.3390/pathogens10111508
6. US Preventive Services Task Force. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(19):1965-1977. doi:10.1001/jama.2021.6238
7. Robertson DJ, Lee JK, Boland CR, et al. Recommendations on fecal immunochemical testing to screen for colorectal neoplasia: a consensus statement by the US Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc. 2017;85(1):2-21.e3. doi:10.1016/j.gie.2016.09.025
8. Lee JK, Liles EG, Bent S, Levin TR, Corley DA. Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis. Ann Intern Med. 2014;160(3):171. doi:10.7326/M13-1484
9. Rex DK, Boland CR, Dominitz JA, et al. Colorectal cancer screening: recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2017;153(1):307-323. doi:10.1053/j.gastro.2017.05.013
10. Deeds SA, Moore CB, Gunnink EJ, et al. Implementation of a mailed faecal immunochemical test programme for colorectal cancer screening among veterans. BMJ Open Qual. 2022;11(4):e001927. doi:10.1136/bmjoq-2022-001927
11. Selby K, Jensen CD, Levin TR, et al. Program components and results from an organized colorectal cancer screening program using annual fecal immunochemical testing. Clin Gastroenterol Hepatol. 2022;20(1):145-152. doi:10.1016/j.cgh.2020.09.042
12. Deeds S, Liu T, Schuttner L, et al. A postcard primer prior to mailed fecal immunochemical test among veterans: a randomized controlled trial. J Gen Intern Med. 2023:38(14):3235-3241. doi:10.1007/s11606-023-08248-7
Colorectal cancer (CRC) is among the most common cancers and causes of cancer-related deaths in the United States.1 Reflective of a nationwide trend, CRC screening rates at the Veterans Affairs Connecticut Healthcare System (VACHS) decreased during the COVID-19 pandemic.2-5 Contributing factors to this decrease included cancellations of elective colonoscopies during the initial phase of the pandemic and concurrent turnover of endoscopists. In 2021, the US Preventive Services Task Force lowered the recommended initial CRC screening age from 50 years to 45 years, further increasing the backlog of unscreened patients.6
Fecal immunochemical testing (FIT) is a noninvasive screening method in which antibodies are used to detect hemoglobin in the stool. The sensitivity and specificity of 1-time FIT are 79% to 80% and 94%, respectively, for the detection of CRC, with sensitivity improving with successive testing.7,8 Annual FIT is recognized as a tier 1 preferred screening method by the US Multi-Society Task Force on Colorectal Cancer.7,9 Programs that mail FIT kits to eligible patients outside of physician visits have been successfully implemented in health care systems.10,11
The VACHS designed and implemented a mailed FIT program using existing infrastructure and staffing.
Program Description
A team of local stakeholders comprised of VACHS leadership, primary care, nursing, and gastroenterology staff, as well as representatives from laboratory, informatics, mail services, and group practice management, was established to execute the project. The team met monthly to plan the project.
The team developed a dataset consisting of patients aged 45 to 75 years who were at average risk for CRC and due for CRC screening. Patients were defined as due for CRC screening if they had not had a colonoscopy in the previous 9 years or a FIT or fecal occult blood test in the previous 11 months. Average risk for CRC was defined by excluding patients with associated diagnosis codes for CRC, colectomy, inflammatory bowel disease, and anemia. The program also excluded patients with diagnosis codes associated with dementia, deferring discussions about cancer screening to their primary care practitioners (PCPs). Patients with invalid mailing addresses were also excluded, as well as those whose PCPs had indicated in the electronic health record that the patient received CRC screening outside the US Department of Veterans Affairs (VA) system.
Letter Templates
Two patient letter electronic health record templates were developed. The first was a primer letter, which was mailed to patients 2 to 3 weeks before the mailed FIT kit as an introduction to the program.12 The purpose of the primer letter was to give advance notice to patients that they could expect a FIT kit to arrive in the mail. The goal was to prepare patients to complete FIT when the kit arrived and prompt them to call the VA to opt out of the mailed FIT program if they were up to date with CRC screening or if they had a condition which made them at high risk for CRC.
The second FIT letter arrived with the FIT kit, introduced FIT and described the importance of CRC screening. The letter detailed instructions for completing FIT and automatically created a FIT order. It also included a list of common conditions that may exclude patients, with a recommendation for patients to contact their medical team if they felt they were not candidates for FIT.
Staff Education
A previous VACHS pilot project demonstrated the success of a mailed FIT program to increase FIT use. Implemented as part of the pilot program, staff education consisted of a session for clinicians about the role of FIT in CRC screening and an all-staff education session. An additional education session about CRC and FIT for all staff was repeated with the program launch.
Program Launch
The mailed FIT program was introduced during a VACHS primary care all-staff meeting. After the meeting, each patient aligned care team (PACT) received an encrypted email that included a list of the patients on their team who were candidates for the program, a patient-facing FIT instruction sheet, detailed instructions on how to send the FIT primer letter, and a FIT package consisting of the labeled FIT kit, FIT letter, and patient instruction sheet. A reminder letter was sent to each patient 3 weeks after the FIT package was mailed. The patient lists were populated into a shared, encrypted Microsoft Teams folder that was edited in real time by PACT teams and viewed by VACHS leadership to track progress.
Program Metrics
At program launch, the VACHS had 4642 patients due for CRC screening who were eligible for the mailed FIT program. On March 7, 2023, the data consisting of FIT tests ordered between December 2022 and May 2023—3 months before and after the launch of the program—were reviewed and categorized. In the 3 months before program launch, 1528 FIT were ordered and 714 were returned (46.7%). In the 3 months after the launch of the program, 4383 FIT were ordered and 1712 were returned (39.1%) (Figure). Test orders increased 287% from the preintervention to the postintervention period. The mean (SD) number of monthly FIT tests prelaunch was 509 (32.7), which increased to 1461 (331.6) postlaunch.
At the VACHS, 61.4% of patients aged 45 to 75 years were up to date with CRC screening before the program launch. In the 3 months after program launch, the rate increased to 63.8% among patients aged 45 to 75 years, the highest rate in our Veterans Integrated Services Network and exceeding the VA national average CRC screening rate, according to unpublished VA Monthly Management Report data.
In the 3 months following the program launch, 139 FIT kits tested positive for potential CRC. Of these, 79 (56.8%) patients had completed a diagnostic colonoscopy. PACT PCPs and nurses received reports on patients with positive FIT tests and those with no colonoscopy scheduled or completed and were asked to follow up.
Discussion
Through a proactive, population-based CRC screening program centered on mailed FIT kits outside of the traditional patient visit, the VACHS increased the use of FIT and rates of CRC screening. The numbers of FIT kits ordered and completed substantially increased in the 3 months after program launch.
Compared to mailed FIT programs described in the literature that rely on centralized processes in that a separate team operates the mailed FIT program for the entire organization, this program used existing PACT infrastructure and staff.10,11 This strategy allowed VACHS to design and implement the program in several months. Not needing to hire new staff or create a central team for the sole purpose of implementing the program allowed us to save on any organizational funding and efforts that would have accompanied the additional staff. The program described in this article may be more attainable for primary care practices or smaller health systems that do not have the capacity for the creation of a centralized process.
Limitations
Although the total number of FIT completions substantially increased during the program, the rate of FIT completion during the mailed FIT program was lower than the rate of completion prior to program launch. This decreased rate of FIT kit completion may be related to separation from a patient visit and potential loss of real-time education with a clinician. The program’s decentralized design increased the existing workload for primary care staff, and as a result, consideration must be given to local staffing levels. Additionally, the report of eligible patients depended on diagnosis codes and may have captured patients with higher-than-average risk of CRC, such as patients with prior history of adenomatous polyps, family history of CRC, or other medical or genetic conditions. We attempted to mitigate this by including a list of conditions that would exclude patients from FIT eligibility in the FIT letter and giving them the option to opt out.
Conclusions
CRC screening rates improved following implementation of a primary care team-centered quality improvement process to proactively identify patients appropriate for FIT and mail them FIT kits. This project highlights that population-health interventions around CRC screening via use of FIT can be successful within a primary care patient-centered medical home model, considering the increases in both CRC screening rates and increase in FIT tests ordered.
Colorectal cancer (CRC) is among the most common cancers and causes of cancer-related deaths in the United States.1 Reflective of a nationwide trend, CRC screening rates at the Veterans Affairs Connecticut Healthcare System (VACHS) decreased during the COVID-19 pandemic.2-5 Contributing factors to this decrease included cancellations of elective colonoscopies during the initial phase of the pandemic and concurrent turnover of endoscopists. In 2021, the US Preventive Services Task Force lowered the recommended initial CRC screening age from 50 years to 45 years, further increasing the backlog of unscreened patients.6
Fecal immunochemical testing (FIT) is a noninvasive screening method in which antibodies are used to detect hemoglobin in the stool. The sensitivity and specificity of 1-time FIT are 79% to 80% and 94%, respectively, for the detection of CRC, with sensitivity improving with successive testing.7,8 Annual FIT is recognized as a tier 1 preferred screening method by the US Multi-Society Task Force on Colorectal Cancer.7,9 Programs that mail FIT kits to eligible patients outside of physician visits have been successfully implemented in health care systems.10,11
The VACHS designed and implemented a mailed FIT program using existing infrastructure and staffing.
Program Description
A team of local stakeholders comprised of VACHS leadership, primary care, nursing, and gastroenterology staff, as well as representatives from laboratory, informatics, mail services, and group practice management, was established to execute the project. The team met monthly to plan the project.
The team developed a dataset consisting of patients aged 45 to 75 years who were at average risk for CRC and due for CRC screening. Patients were defined as due for CRC screening if they had not had a colonoscopy in the previous 9 years or a FIT or fecal occult blood test in the previous 11 months. Average risk for CRC was defined by excluding patients with associated diagnosis codes for CRC, colectomy, inflammatory bowel disease, and anemia. The program also excluded patients with diagnosis codes associated with dementia, deferring discussions about cancer screening to their primary care practitioners (PCPs). Patients with invalid mailing addresses were also excluded, as well as those whose PCPs had indicated in the electronic health record that the patient received CRC screening outside the US Department of Veterans Affairs (VA) system.
Letter Templates
Two patient letter electronic health record templates were developed. The first was a primer letter, which was mailed to patients 2 to 3 weeks before the mailed FIT kit as an introduction to the program.12 The purpose of the primer letter was to give advance notice to patients that they could expect a FIT kit to arrive in the mail. The goal was to prepare patients to complete FIT when the kit arrived and prompt them to call the VA to opt out of the mailed FIT program if they were up to date with CRC screening or if they had a condition which made them at high risk for CRC.
The second FIT letter arrived with the FIT kit, introduced FIT and described the importance of CRC screening. The letter detailed instructions for completing FIT and automatically created a FIT order. It also included a list of common conditions that may exclude patients, with a recommendation for patients to contact their medical team if they felt they were not candidates for FIT.
Staff Education
A previous VACHS pilot project demonstrated the success of a mailed FIT program to increase FIT use. Implemented as part of the pilot program, staff education consisted of a session for clinicians about the role of FIT in CRC screening and an all-staff education session. An additional education session about CRC and FIT for all staff was repeated with the program launch.
Program Launch
The mailed FIT program was introduced during a VACHS primary care all-staff meeting. After the meeting, each patient aligned care team (PACT) received an encrypted email that included a list of the patients on their team who were candidates for the program, a patient-facing FIT instruction sheet, detailed instructions on how to send the FIT primer letter, and a FIT package consisting of the labeled FIT kit, FIT letter, and patient instruction sheet. A reminder letter was sent to each patient 3 weeks after the FIT package was mailed. The patient lists were populated into a shared, encrypted Microsoft Teams folder that was edited in real time by PACT teams and viewed by VACHS leadership to track progress.
Program Metrics
At program launch, the VACHS had 4642 patients due for CRC screening who were eligible for the mailed FIT program. On March 7, 2023, the data consisting of FIT tests ordered between December 2022 and May 2023—3 months before and after the launch of the program—were reviewed and categorized. In the 3 months before program launch, 1528 FIT were ordered and 714 were returned (46.7%). In the 3 months after the launch of the program, 4383 FIT were ordered and 1712 were returned (39.1%) (Figure). Test orders increased 287% from the preintervention to the postintervention period. The mean (SD) number of monthly FIT tests prelaunch was 509 (32.7), which increased to 1461 (331.6) postlaunch.
At the VACHS, 61.4% of patients aged 45 to 75 years were up to date with CRC screening before the program launch. In the 3 months after program launch, the rate increased to 63.8% among patients aged 45 to 75 years, the highest rate in our Veterans Integrated Services Network and exceeding the VA national average CRC screening rate, according to unpublished VA Monthly Management Report data.
In the 3 months following the program launch, 139 FIT kits tested positive for potential CRC. Of these, 79 (56.8%) patients had completed a diagnostic colonoscopy. PACT PCPs and nurses received reports on patients with positive FIT tests and those with no colonoscopy scheduled or completed and were asked to follow up.
Discussion
Through a proactive, population-based CRC screening program centered on mailed FIT kits outside of the traditional patient visit, the VACHS increased the use of FIT and rates of CRC screening. The numbers of FIT kits ordered and completed substantially increased in the 3 months after program launch.
Compared to mailed FIT programs described in the literature that rely on centralized processes in that a separate team operates the mailed FIT program for the entire organization, this program used existing PACT infrastructure and staff.10,11 This strategy allowed VACHS to design and implement the program in several months. Not needing to hire new staff or create a central team for the sole purpose of implementing the program allowed us to save on any organizational funding and efforts that would have accompanied the additional staff. The program described in this article may be more attainable for primary care practices or smaller health systems that do not have the capacity for the creation of a centralized process.
Limitations
Although the total number of FIT completions substantially increased during the program, the rate of FIT completion during the mailed FIT program was lower than the rate of completion prior to program launch. This decreased rate of FIT kit completion may be related to separation from a patient visit and potential loss of real-time education with a clinician. The program’s decentralized design increased the existing workload for primary care staff, and as a result, consideration must be given to local staffing levels. Additionally, the report of eligible patients depended on diagnosis codes and may have captured patients with higher-than-average risk of CRC, such as patients with prior history of adenomatous polyps, family history of CRC, or other medical or genetic conditions. We attempted to mitigate this by including a list of conditions that would exclude patients from FIT eligibility in the FIT letter and giving them the option to opt out.
Conclusions
CRC screening rates improved following implementation of a primary care team-centered quality improvement process to proactively identify patients appropriate for FIT and mail them FIT kits. This project highlights that population-health interventions around CRC screening via use of FIT can be successful within a primary care patient-centered medical home model, considering the increases in both CRC screening rates and increase in FIT tests ordered.
1. American Cancer Society. Key statistics for colorectal cancer. Revised January 29, 2024. Accessed June 11, 2024. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
2. Chen RC, Haynes K, Du S, Barron J, Katz AJ. Association of cancer screening deficit in the United States with the COVID-19 pandemic. JAMA Oncol. 2021;7(6):878-884. doi:10.1001/jamaoncol.2021.0884
3. Mazidimoradi A, Tiznobaik A, Salehiniya H. Impact of the COVID-19 pandemic on colorectal cancer screening: a systematic review. J Gastrointest Cancer. 2022;53(3):730-744. doi:10.1007/s12029-021-00679-x
4. Adams MA, Kurlander JE, Gao Y, Yankey N, Saini SD. Impact of coronavirus disease 2019 on screening colonoscopy utilization in a large integrated health system. Gastroenterology. 2022;162(7):2098-2100.e2. doi:10.1053/j.gastro.2022.02.034
5. Sundaram S, Olson S, Sharma P, Rajendra S. A review of the impact of the COVID-19 pandemic on colorectal cancer screening: implications and solutions. Pathogens. 2021;10(11):558. doi:10.3390/pathogens10111508
6. US Preventive Services Task Force. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(19):1965-1977. doi:10.1001/jama.2021.6238
7. Robertson DJ, Lee JK, Boland CR, et al. Recommendations on fecal immunochemical testing to screen for colorectal neoplasia: a consensus statement by the US Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc. 2017;85(1):2-21.e3. doi:10.1016/j.gie.2016.09.025
8. Lee JK, Liles EG, Bent S, Levin TR, Corley DA. Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis. Ann Intern Med. 2014;160(3):171. doi:10.7326/M13-1484
9. Rex DK, Boland CR, Dominitz JA, et al. Colorectal cancer screening: recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2017;153(1):307-323. doi:10.1053/j.gastro.2017.05.013
10. Deeds SA, Moore CB, Gunnink EJ, et al. Implementation of a mailed faecal immunochemical test programme for colorectal cancer screening among veterans. BMJ Open Qual. 2022;11(4):e001927. doi:10.1136/bmjoq-2022-001927
11. Selby K, Jensen CD, Levin TR, et al. Program components and results from an organized colorectal cancer screening program using annual fecal immunochemical testing. Clin Gastroenterol Hepatol. 2022;20(1):145-152. doi:10.1016/j.cgh.2020.09.042
12. Deeds S, Liu T, Schuttner L, et al. A postcard primer prior to mailed fecal immunochemical test among veterans: a randomized controlled trial. J Gen Intern Med. 2023:38(14):3235-3241. doi:10.1007/s11606-023-08248-7
1. American Cancer Society. Key statistics for colorectal cancer. Revised January 29, 2024. Accessed June 11, 2024. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
2. Chen RC, Haynes K, Du S, Barron J, Katz AJ. Association of cancer screening deficit in the United States with the COVID-19 pandemic. JAMA Oncol. 2021;7(6):878-884. doi:10.1001/jamaoncol.2021.0884
3. Mazidimoradi A, Tiznobaik A, Salehiniya H. Impact of the COVID-19 pandemic on colorectal cancer screening: a systematic review. J Gastrointest Cancer. 2022;53(3):730-744. doi:10.1007/s12029-021-00679-x
4. Adams MA, Kurlander JE, Gao Y, Yankey N, Saini SD. Impact of coronavirus disease 2019 on screening colonoscopy utilization in a large integrated health system. Gastroenterology. 2022;162(7):2098-2100.e2. doi:10.1053/j.gastro.2022.02.034
5. Sundaram S, Olson S, Sharma P, Rajendra S. A review of the impact of the COVID-19 pandemic on colorectal cancer screening: implications and solutions. Pathogens. 2021;10(11):558. doi:10.3390/pathogens10111508
6. US Preventive Services Task Force. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(19):1965-1977. doi:10.1001/jama.2021.6238
7. Robertson DJ, Lee JK, Boland CR, et al. Recommendations on fecal immunochemical testing to screen for colorectal neoplasia: a consensus statement by the US Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc. 2017;85(1):2-21.e3. doi:10.1016/j.gie.2016.09.025
8. Lee JK, Liles EG, Bent S, Levin TR, Corley DA. Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis. Ann Intern Med. 2014;160(3):171. doi:10.7326/M13-1484
9. Rex DK, Boland CR, Dominitz JA, et al. Colorectal cancer screening: recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2017;153(1):307-323. doi:10.1053/j.gastro.2017.05.013
10. Deeds SA, Moore CB, Gunnink EJ, et al. Implementation of a mailed faecal immunochemical test programme for colorectal cancer screening among veterans. BMJ Open Qual. 2022;11(4):e001927. doi:10.1136/bmjoq-2022-001927
11. Selby K, Jensen CD, Levin TR, et al. Program components and results from an organized colorectal cancer screening program using annual fecal immunochemical testing. Clin Gastroenterol Hepatol. 2022;20(1):145-152. doi:10.1016/j.cgh.2020.09.042
12. Deeds S, Liu T, Schuttner L, et al. A postcard primer prior to mailed fecal immunochemical test among veterans: a randomized controlled trial. J Gen Intern Med. 2023:38(14):3235-3241. doi:10.1007/s11606-023-08248-7
VHA CRC Screening Has Blind Spots, Disparities
TOPLINE:
More than 1 in 8 colorectal cancer (CRC) cases among veterans occur outside the standard screening age of 50-75 years or those with high-risk personal or family history. High-risk patients face > 6 times the risk for CRC compared with average-risk patients aged 50-75 years who are up to date with screening, while Black patients have > 50% higher risk compared with White patients.
METHODOLOGY:
Researchers conducted a case-control analysis using Veterans Health Administration (VHA) Corporate Data Warehouse data from 2012-2018 at 2 sites: Veterans Affairs (VA) New York Harbor Health Care System and VA Puget Sound Health Care System.
Participants included 3714 cases among veterans with CRC matched to 14,856 controls (4:1), with matching on age (± 3 years), sex, and facility site; each control was used once.
Screening categories included 5 groups by age (50-75 years vs < 50 years or > 75 years), screening up-to-date status, and high-risk status (inflammatory bowel disease, hereditary cancer syndromes, or family history).
CRC screening was considered up to date if US Preventive Services Task Force-recommended tests were completed on time (colonoscopy ≤ 10 years; guaiac-based fecal occult blood test or fecal immunochemical test ≤ 1 year).
TAKEAWAY:
Compared with category 1 (age 50-75 years and up-to-date with screening), CRC was associated with category 4 (age < 50 years or > 75 years and not up to date) (odds ratio [OR], 1.40; 95% CI, 1.11-1.78), and category 5 (high risk) (OR, 6.23; 95% CI, 5.06-7.66).
Race and comorbidity associations included higher CRC risk for Black vs White patients (OR, 1.54; 95% CI, 1.37-1.73), and higher CRC risk with diabetes (OR, 1.65; 95% CI, 1.51-1.81) and alcohol use disorder (OR, 1.53; 95% CI, 1.35-1.73).
Among 3714 CRC cases, 71.1% occurred in individuals aged 50-75 years not up to date with screening.
A total of 12.5% of CRC cases occurred in people outside age 50-75 or with high-risk personal or family history, suggesting that conventional screening-adherence metrics may miss a clinically relevant minority.
IN PRACTICE:
“The conventional measure of CRC screening, focused on average-risk individuals aged 50 to 75, does not reflect screening status in an important minority of CRC patients," the authors wrote.
SOURCE:
The study was led by researchers at NYU Grossman School of Medicine and Veterans Affairs New York Harbor Health Care System, and published online July 9, 2026 in Medicine.
LIMITATIONS:
The study population consisted predominantly of male veterans (97.1%), who tend to be older and have more comorbidities compared with the US population, which may limit the generalizability of findings to other populations. Researchers defined screening status cross-sectionally relative to a single point in time rather than assessing longitudinal screening adherence, which may not fully capture the consistency of screening over time that is likely important for defining CRC risk. Veterans may receive screening at non-VA medical facilities, potentially leading to incomplete documentation of screening status and important covariates such as race, ethnicity, and comorbidities. The possibility of residual confounding cannot be excluded despite adjustment for multiple risk factors in the analysis.
DISCLOSURES:
This study received support from NIH grant K08 CA230162 and the AGA Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer, both awarded to Peter S. Liang. Liang disclosed receiving research support from Freenome and serving on the advisory boards for Guardant Health and Natera. The remaining authors reported no funding or conflicts of interest to disclose.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE:
More than 1 in 8 colorectal cancer (CRC) cases among veterans occur outside the standard screening age of 50-75 years or those with high-risk personal or family history. High-risk patients face > 6 times the risk for CRC compared with average-risk patients aged 50-75 years who are up to date with screening, while Black patients have > 50% higher risk compared with White patients.
METHODOLOGY:
Researchers conducted a case-control analysis using Veterans Health Administration (VHA) Corporate Data Warehouse data from 2012-2018 at 2 sites: Veterans Affairs (VA) New York Harbor Health Care System and VA Puget Sound Health Care System.
Participants included 3714 cases among veterans with CRC matched to 14,856 controls (4:1), with matching on age (± 3 years), sex, and facility site; each control was used once.
Screening categories included 5 groups by age (50-75 years vs < 50 years or > 75 years), screening up-to-date status, and high-risk status (inflammatory bowel disease, hereditary cancer syndromes, or family history).
CRC screening was considered up to date if US Preventive Services Task Force-recommended tests were completed on time (colonoscopy ≤ 10 years; guaiac-based fecal occult blood test or fecal immunochemical test ≤ 1 year).
TAKEAWAY:
Compared with category 1 (age 50-75 years and up-to-date with screening), CRC was associated with category 4 (age < 50 years or > 75 years and not up to date) (odds ratio [OR], 1.40; 95% CI, 1.11-1.78), and category 5 (high risk) (OR, 6.23; 95% CI, 5.06-7.66).
Race and comorbidity associations included higher CRC risk for Black vs White patients (OR, 1.54; 95% CI, 1.37-1.73), and higher CRC risk with diabetes (OR, 1.65; 95% CI, 1.51-1.81) and alcohol use disorder (OR, 1.53; 95% CI, 1.35-1.73).
Among 3714 CRC cases, 71.1% occurred in individuals aged 50-75 years not up to date with screening.
A total of 12.5% of CRC cases occurred in people outside age 50-75 or with high-risk personal or family history, suggesting that conventional screening-adherence metrics may miss a clinically relevant minority.
IN PRACTICE:
“The conventional measure of CRC screening, focused on average-risk individuals aged 50 to 75, does not reflect screening status in an important minority of CRC patients," the authors wrote.
SOURCE:
The study was led by researchers at NYU Grossman School of Medicine and Veterans Affairs New York Harbor Health Care System, and published online July 9, 2026 in Medicine.
LIMITATIONS:
The study population consisted predominantly of male veterans (97.1%), who tend to be older and have more comorbidities compared with the US population, which may limit the generalizability of findings to other populations. Researchers defined screening status cross-sectionally relative to a single point in time rather than assessing longitudinal screening adherence, which may not fully capture the consistency of screening over time that is likely important for defining CRC risk. Veterans may receive screening at non-VA medical facilities, potentially leading to incomplete documentation of screening status and important covariates such as race, ethnicity, and comorbidities. The possibility of residual confounding cannot be excluded despite adjustment for multiple risk factors in the analysis.
DISCLOSURES:
This study received support from NIH grant K08 CA230162 and the AGA Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer, both awarded to Peter S. Liang. Liang disclosed receiving research support from Freenome and serving on the advisory boards for Guardant Health and Natera. The remaining authors reported no funding or conflicts of interest to disclose.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE:
More than 1 in 8 colorectal cancer (CRC) cases among veterans occur outside the standard screening age of 50-75 years or those with high-risk personal or family history. High-risk patients face > 6 times the risk for CRC compared with average-risk patients aged 50-75 years who are up to date with screening, while Black patients have > 50% higher risk compared with White patients.
METHODOLOGY:
Researchers conducted a case-control analysis using Veterans Health Administration (VHA) Corporate Data Warehouse data from 2012-2018 at 2 sites: Veterans Affairs (VA) New York Harbor Health Care System and VA Puget Sound Health Care System.
Participants included 3714 cases among veterans with CRC matched to 14,856 controls (4:1), with matching on age (± 3 years), sex, and facility site; each control was used once.
Screening categories included 5 groups by age (50-75 years vs < 50 years or > 75 years), screening up-to-date status, and high-risk status (inflammatory bowel disease, hereditary cancer syndromes, or family history).
CRC screening was considered up to date if US Preventive Services Task Force-recommended tests were completed on time (colonoscopy ≤ 10 years; guaiac-based fecal occult blood test or fecal immunochemical test ≤ 1 year).
TAKEAWAY:
Compared with category 1 (age 50-75 years and up-to-date with screening), CRC was associated with category 4 (age < 50 years or > 75 years and not up to date) (odds ratio [OR], 1.40; 95% CI, 1.11-1.78), and category 5 (high risk) (OR, 6.23; 95% CI, 5.06-7.66).
Race and comorbidity associations included higher CRC risk for Black vs White patients (OR, 1.54; 95% CI, 1.37-1.73), and higher CRC risk with diabetes (OR, 1.65; 95% CI, 1.51-1.81) and alcohol use disorder (OR, 1.53; 95% CI, 1.35-1.73).
Among 3714 CRC cases, 71.1% occurred in individuals aged 50-75 years not up to date with screening.
A total of 12.5% of CRC cases occurred in people outside age 50-75 or with high-risk personal or family history, suggesting that conventional screening-adherence metrics may miss a clinically relevant minority.
IN PRACTICE:
“The conventional measure of CRC screening, focused on average-risk individuals aged 50 to 75, does not reflect screening status in an important minority of CRC patients," the authors wrote.
SOURCE:
The study was led by researchers at NYU Grossman School of Medicine and Veterans Affairs New York Harbor Health Care System, and published online July 9, 2026 in Medicine.
LIMITATIONS:
The study population consisted predominantly of male veterans (97.1%), who tend to be older and have more comorbidities compared with the US population, which may limit the generalizability of findings to other populations. Researchers defined screening status cross-sectionally relative to a single point in time rather than assessing longitudinal screening adherence, which may not fully capture the consistency of screening over time that is likely important for defining CRC risk. Veterans may receive screening at non-VA medical facilities, potentially leading to incomplete documentation of screening status and important covariates such as race, ethnicity, and comorbidities. The possibility of residual confounding cannot be excluded despite adjustment for multiple risk factors in the analysis.
DISCLOSURES:
This study received support from NIH grant K08 CA230162 and the AGA Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer, both awarded to Peter S. Liang. Liang disclosed receiving research support from Freenome and serving on the advisory boards for Guardant Health and Natera. The remaining authors reported no funding or conflicts of interest to disclose.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
TOPLINE:
In a meta-analysis, patients with hidradenitis suppurativa (HS) faced a more than 80% higher risk for cancer overall than the general population, with particularly elevated risks for gastrointestinal, head and neck, hematologic, and respiratory system cancers.
METHODOLOGY:
- Researchers conducted a meta-analysis including 11 studies from PubMed, Embase, and Web of Science databases published between 2001 and 2024; these studies examined the risk for cancer in patients with HS compared with that in the general population.
- These studies included 624,721 patients diagnosed with HS (mean age, 33.6-43.8 years) and 393,691,636 control individuals from the general population.
- Researchers performed an inverse variance-weighted random-effects analysis to calculate pooled odds ratios (ORs) for cancer overall and specific cancer subtypes.
- Cancer types were categorized into 11 groups for subgroup analysis: bone and soft tissue cancers, breast cancer, central nervous system cancers, endocrine-related cancers, gastrointestinal cancers, head and neck cancers, hematologic cancers, respiratory system cancers, skin cancers, urogenital cancers, and unspecified cancers.
TAKEAWAY:
- Patients with HS demonstrated a significantly higher risk for cancer overall than control individuals (crude OR, 1.82; P = .018).
- Patients with HS showed an increased risk for gastrointestinal cancers (crude OR, 1.61; P = .0002), head and neck cancers (crude OR, 2.41; P = .00001), hematologic cancers (crude OR, 1.71; P = .00005), and respiratory system cancers (crude OR, 1.81; P = .04).
- Patients with HS demonstrated significantly elevated risks for both Hodgkin lymphoma (OR, 2.44; P = .0001) and non-Hodgkin lymphoma (OR, 1.15; P = .012).
- A non-significant increased risk for skin cancer was observed in patients with HS (crude OR, 1.48; P = .08). No increased risks for bone and soft tissue cancers, central nervous system cancers, breast cancer, or urogenital cancers were observed in patients with HS.
IN PRACTICE:
"HS was associated with an increased overall risk of cancer, including several specific subtypes, compared with controls," the authors wrote, suggesting that "studies are adjusting for confounders and assess long-term associations between HS and cancer risk are highly needed to investigate which factors contribute to this cancer risk."
SOURCE:
This study was led by Daniel Isufi, Department of Dermatology and Allergy, Copenhagen University Hospital-Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark. It was published online on March 11, 2026, in Dermatology and Therapy.
LIMITATIONS:
Limited data on cancer subtypes hindered meta-analyses of rare cancers, and the lack of reporting on anti‑inflammatory treatment and disease severity prevented subgroup analyses. Most studies originated from North America, introducing potential geographic bias. No study reported BMI, and ethnicity was poorly documented. Only few studies adjusted for key confounders (smoking, obesity, and alcohol intake), limiting the determination of whether the increased risk for cancer was due to HS itself or shared lifestyle and metabolic factors.
DISCLOSURES:
This study did not receive any funding or sponsorship. Two authors reported receiving research grant funding from the LEO Foundation and having other ties with various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
In a meta-analysis, patients with hidradenitis suppurativa (HS) faced a more than 80% higher risk for cancer overall than the general population, with particularly elevated risks for gastrointestinal, head and neck, hematologic, and respiratory system cancers.
METHODOLOGY:
- Researchers conducted a meta-analysis including 11 studies from PubMed, Embase, and Web of Science databases published between 2001 and 2024; these studies examined the risk for cancer in patients with HS compared with that in the general population.
- These studies included 624,721 patients diagnosed with HS (mean age, 33.6-43.8 years) and 393,691,636 control individuals from the general population.
- Researchers performed an inverse variance-weighted random-effects analysis to calculate pooled odds ratios (ORs) for cancer overall and specific cancer subtypes.
- Cancer types were categorized into 11 groups for subgroup analysis: bone and soft tissue cancers, breast cancer, central nervous system cancers, endocrine-related cancers, gastrointestinal cancers, head and neck cancers, hematologic cancers, respiratory system cancers, skin cancers, urogenital cancers, and unspecified cancers.
TAKEAWAY:
- Patients with HS demonstrated a significantly higher risk for cancer overall than control individuals (crude OR, 1.82; P = .018).
- Patients with HS showed an increased risk for gastrointestinal cancers (crude OR, 1.61; P = .0002), head and neck cancers (crude OR, 2.41; P = .00001), hematologic cancers (crude OR, 1.71; P = .00005), and respiratory system cancers (crude OR, 1.81; P = .04).
- Patients with HS demonstrated significantly elevated risks for both Hodgkin lymphoma (OR, 2.44; P = .0001) and non-Hodgkin lymphoma (OR, 1.15; P = .012).
- A non-significant increased risk for skin cancer was observed in patients with HS (crude OR, 1.48; P = .08). No increased risks for bone and soft tissue cancers, central nervous system cancers, breast cancer, or urogenital cancers were observed in patients with HS.
IN PRACTICE:
"HS was associated with an increased overall risk of cancer, including several specific subtypes, compared with controls," the authors wrote, suggesting that "studies are adjusting for confounders and assess long-term associations between HS and cancer risk are highly needed to investigate which factors contribute to this cancer risk."
SOURCE:
This study was led by Daniel Isufi, Department of Dermatology and Allergy, Copenhagen University Hospital-Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark. It was published online on March 11, 2026, in Dermatology and Therapy.
LIMITATIONS:
Limited data on cancer subtypes hindered meta-analyses of rare cancers, and the lack of reporting on anti‑inflammatory treatment and disease severity prevented subgroup analyses. Most studies originated from North America, introducing potential geographic bias. No study reported BMI, and ethnicity was poorly documented. Only few studies adjusted for key confounders (smoking, obesity, and alcohol intake), limiting the determination of whether the increased risk for cancer was due to HS itself or shared lifestyle and metabolic factors.
DISCLOSURES:
This study did not receive any funding or sponsorship. Two authors reported receiving research grant funding from the LEO Foundation and having other ties with various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
In a meta-analysis, patients with hidradenitis suppurativa (HS) faced a more than 80% higher risk for cancer overall than the general population, with particularly elevated risks for gastrointestinal, head and neck, hematologic, and respiratory system cancers.
METHODOLOGY:
- Researchers conducted a meta-analysis including 11 studies from PubMed, Embase, and Web of Science databases published between 2001 and 2024; these studies examined the risk for cancer in patients with HS compared with that in the general population.
- These studies included 624,721 patients diagnosed with HS (mean age, 33.6-43.8 years) and 393,691,636 control individuals from the general population.
- Researchers performed an inverse variance-weighted random-effects analysis to calculate pooled odds ratios (ORs) for cancer overall and specific cancer subtypes.
- Cancer types were categorized into 11 groups for subgroup analysis: bone and soft tissue cancers, breast cancer, central nervous system cancers, endocrine-related cancers, gastrointestinal cancers, head and neck cancers, hematologic cancers, respiratory system cancers, skin cancers, urogenital cancers, and unspecified cancers.
TAKEAWAY:
- Patients with HS demonstrated a significantly higher risk for cancer overall than control individuals (crude OR, 1.82; P = .018).
- Patients with HS showed an increased risk for gastrointestinal cancers (crude OR, 1.61; P = .0002), head and neck cancers (crude OR, 2.41; P = .00001), hematologic cancers (crude OR, 1.71; P = .00005), and respiratory system cancers (crude OR, 1.81; P = .04).
- Patients with HS demonstrated significantly elevated risks for both Hodgkin lymphoma (OR, 2.44; P = .0001) and non-Hodgkin lymphoma (OR, 1.15; P = .012).
- A non-significant increased risk for skin cancer was observed in patients with HS (crude OR, 1.48; P = .08). No increased risks for bone and soft tissue cancers, central nervous system cancers, breast cancer, or urogenital cancers were observed in patients with HS.
IN PRACTICE:
"HS was associated with an increased overall risk of cancer, including several specific subtypes, compared with controls," the authors wrote, suggesting that "studies are adjusting for confounders and assess long-term associations between HS and cancer risk are highly needed to investigate which factors contribute to this cancer risk."
SOURCE:
This study was led by Daniel Isufi, Department of Dermatology and Allergy, Copenhagen University Hospital-Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark. It was published online on March 11, 2026, in Dermatology and Therapy.
LIMITATIONS:
Limited data on cancer subtypes hindered meta-analyses of rare cancers, and the lack of reporting on anti‑inflammatory treatment and disease severity prevented subgroup analyses. Most studies originated from North America, introducing potential geographic bias. No study reported BMI, and ethnicity was poorly documented. Only few studies adjusted for key confounders (smoking, obesity, and alcohol intake), limiting the determination of whether the increased risk for cancer was due to HS itself or shared lifestyle and metabolic factors.
DISCLOSURES:
This study did not receive any funding or sponsorship. Two authors reported receiving research grant funding from the LEO Foundation and having other ties with various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
“Colon Age” Tool Evaluates Early CRC Risk in Male Vets
TOPLINE: Interviews with 23 male veterans (aged 35-49 years) at average-risk for colorectal cancer (CRC) and 8 primary care practitioners (PCPs) found broad acceptability of the Colon Age concept, with 96% of patients agreeing to calculation. PCPs describe its potential use to support screening discussions (fecal immunochemical test [FIT] vs colonoscopy) but emphasize workflow barriers, requesting electronic medical record integration and “time neutral” implementation.
METHODOLOGY:
Researchers conducted semistructured qualitative interviews with 31 participants (23 male veteran patients aged 35-49 years and 8 PCPs) at the Richard L. Roudebush Veterans Affairs Medical Center between June and September 2022.
Patients were eligible if they were at average risk for CRC, had no prior screening (colonoscopy or fecal immunochemical test [FIT]), no inflammatory bowel disease, and no significant family history of CRC.
Interviews explored participants' experiences with CRC screening, understanding of the Colon Age tool, and perceived clinical use.
Audio-recorded interviews were transcribed, deidentified, and analyzed using the constant comparison method with open and focused coding phases until saturation was reached.
TAKEAWAY:
Among 23 male veteran patients (mean age 47 years), 96% agreed to have their Colon Age calculated; 68% had a Colon Age below their biological age, 14% higher than their biological age, and 18% equal to their biological age.
Patients accepted the Colon Age concept, finding it easy to understand and helpful for being informed about their health, though most were unaware of screening options beyond colonoscopy prior to the interview.
The 8 PCPs (mean age 53 years, 50% female, mean 29 years in practice) interviewed found the tool acceptable and useful for screening conversations, improving uptake, and facilitating shared decision-making, particularly in gray zone cases where screening decisions are unclear.
PCPs emphasized the need for the tool to be integrated into the electronic medical record system and expressed concerns about time commitment, consistency with practice guidelines, and the validation process, stating they would only use the tool if it were time neutral and evidence-based.
IN PRACTICE: “Although the age at which to begin colorectal cancer screening in the US was lowered to 45 years in 2018, uptake of screening in persons aged 45 to 49 has been slow,” wrote the authors of the study.
SOURCE:The study was led by researchers at the VA Center for Health Information and Communication. It was published online on July 15 in BMC Primary Care.
LIMITATIONS: The study was conducted at a single VA medical center in the Midwest and all patient participants were male, which may limit generalizability to nonveteran patients, female patients, and non-VA clinicians. The Colon Age tool has limitations, as it was based on a risk prediction model with modest discrimination, and the linkage to screening recommendations was based on arbitrary Surveillance, Epidemiology and End Results thresholds chosen by the tool developers. Additionally, the qualitative nature of the study with a small sample size may not capture the full range of perspectives across diverse health care settings and patient populations.
DISCLOSURES: The primary author received support from Health Services Research and Development, Veterans Administration. Funding for this project was provided by Richard L. Roudebush VA Medical Center Indianapolis, Indiana Center for Health Information, and Communication COIN funds. The authors reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: Interviews with 23 male veterans (aged 35-49 years) at average-risk for colorectal cancer (CRC) and 8 primary care practitioners (PCPs) found broad acceptability of the Colon Age concept, with 96% of patients agreeing to calculation. PCPs describe its potential use to support screening discussions (fecal immunochemical test [FIT] vs colonoscopy) but emphasize workflow barriers, requesting electronic medical record integration and “time neutral” implementation.
METHODOLOGY:
Researchers conducted semistructured qualitative interviews with 31 participants (23 male veteran patients aged 35-49 years and 8 PCPs) at the Richard L. Roudebush Veterans Affairs Medical Center between June and September 2022.
Patients were eligible if they were at average risk for CRC, had no prior screening (colonoscopy or fecal immunochemical test [FIT]), no inflammatory bowel disease, and no significant family history of CRC.
Interviews explored participants' experiences with CRC screening, understanding of the Colon Age tool, and perceived clinical use.
Audio-recorded interviews were transcribed, deidentified, and analyzed using the constant comparison method with open and focused coding phases until saturation was reached.
TAKEAWAY:
Among 23 male veteran patients (mean age 47 years), 96% agreed to have their Colon Age calculated; 68% had a Colon Age below their biological age, 14% higher than their biological age, and 18% equal to their biological age.
Patients accepted the Colon Age concept, finding it easy to understand and helpful for being informed about their health, though most were unaware of screening options beyond colonoscopy prior to the interview.
The 8 PCPs (mean age 53 years, 50% female, mean 29 years in practice) interviewed found the tool acceptable and useful for screening conversations, improving uptake, and facilitating shared decision-making, particularly in gray zone cases where screening decisions are unclear.
PCPs emphasized the need for the tool to be integrated into the electronic medical record system and expressed concerns about time commitment, consistency with practice guidelines, and the validation process, stating they would only use the tool if it were time neutral and evidence-based.
IN PRACTICE: “Although the age at which to begin colorectal cancer screening in the US was lowered to 45 years in 2018, uptake of screening in persons aged 45 to 49 has been slow,” wrote the authors of the study.
SOURCE:The study was led by researchers at the VA Center for Health Information and Communication. It was published online on July 15 in BMC Primary Care.
LIMITATIONS: The study was conducted at a single VA medical center in the Midwest and all patient participants were male, which may limit generalizability to nonveteran patients, female patients, and non-VA clinicians. The Colon Age tool has limitations, as it was based on a risk prediction model with modest discrimination, and the linkage to screening recommendations was based on arbitrary Surveillance, Epidemiology and End Results thresholds chosen by the tool developers. Additionally, the qualitative nature of the study with a small sample size may not capture the full range of perspectives across diverse health care settings and patient populations.
DISCLOSURES: The primary author received support from Health Services Research and Development, Veterans Administration. Funding for this project was provided by Richard L. Roudebush VA Medical Center Indianapolis, Indiana Center for Health Information, and Communication COIN funds. The authors reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: Interviews with 23 male veterans (aged 35-49 years) at average-risk for colorectal cancer (CRC) and 8 primary care practitioners (PCPs) found broad acceptability of the Colon Age concept, with 96% of patients agreeing to calculation. PCPs describe its potential use to support screening discussions (fecal immunochemical test [FIT] vs colonoscopy) but emphasize workflow barriers, requesting electronic medical record integration and “time neutral” implementation.
METHODOLOGY:
Researchers conducted semistructured qualitative interviews with 31 participants (23 male veteran patients aged 35-49 years and 8 PCPs) at the Richard L. Roudebush Veterans Affairs Medical Center between June and September 2022.
Patients were eligible if they were at average risk for CRC, had no prior screening (colonoscopy or fecal immunochemical test [FIT]), no inflammatory bowel disease, and no significant family history of CRC.
Interviews explored participants' experiences with CRC screening, understanding of the Colon Age tool, and perceived clinical use.
Audio-recorded interviews were transcribed, deidentified, and analyzed using the constant comparison method with open and focused coding phases until saturation was reached.
TAKEAWAY:
Among 23 male veteran patients (mean age 47 years), 96% agreed to have their Colon Age calculated; 68% had a Colon Age below their biological age, 14% higher than their biological age, and 18% equal to their biological age.
Patients accepted the Colon Age concept, finding it easy to understand and helpful for being informed about their health, though most were unaware of screening options beyond colonoscopy prior to the interview.
The 8 PCPs (mean age 53 years, 50% female, mean 29 years in practice) interviewed found the tool acceptable and useful for screening conversations, improving uptake, and facilitating shared decision-making, particularly in gray zone cases where screening decisions are unclear.
PCPs emphasized the need for the tool to be integrated into the electronic medical record system and expressed concerns about time commitment, consistency with practice guidelines, and the validation process, stating they would only use the tool if it were time neutral and evidence-based.
IN PRACTICE: “Although the age at which to begin colorectal cancer screening in the US was lowered to 45 years in 2018, uptake of screening in persons aged 45 to 49 has been slow,” wrote the authors of the study.
SOURCE:The study was led by researchers at the VA Center for Health Information and Communication. It was published online on July 15 in BMC Primary Care.
LIMITATIONS: The study was conducted at a single VA medical center in the Midwest and all patient participants were male, which may limit generalizability to nonveteran patients, female patients, and non-VA clinicians. The Colon Age tool has limitations, as it was based on a risk prediction model with modest discrimination, and the linkage to screening recommendations was based on arbitrary Surveillance, Epidemiology and End Results thresholds chosen by the tool developers. Additionally, the qualitative nature of the study with a small sample size may not capture the full range of perspectives across diverse health care settings and patient populations.
DISCLOSURES: The primary author received support from Health Services Research and Development, Veterans Administration. Funding for this project was provided by Richard L. Roudebush VA Medical Center Indianapolis, Indiana Center for Health Information, and Communication COIN funds. The authors reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Veteran Testicular Cancer Survivors Face High Mental Health Burden
Anxiety, depression, and suicide rates are elevated for veterans who are survivors of testicular cancer (TC) compared with veterans without cancer, a retrospective analysis finds.
Over 5 years, the cumulative incidence of anxiety and depression was 53.4% in veterans with TC vs 35.0% in matched controls (P < .001; hazard ratio [HR], 1.66), reported Aditya Bagrodia, MD, professor of urology and radiation oncology at the University of California San Diego, et al in Cancer Medicine. The cumulative incidence of suicidality was 5.0% and 0.1%, respectively (P < .001; HR, 22.99).
“More than half of men with testicular cancer contend with these diagnoses,” Bagrodia told Federal Practitioner. “There are risk factors, including chemotherapy, being single or divorced, or unemployed.”
Patients in these groups warrant aggressive screening and intervention, Bagrodia said. TC is the most common cancer in men in the military and the most common malignancy in men aged 18 to 45 years, Bagrodia said: “The vast majority of men who have testicular cancer are curable.”
Patients, however, face an intense burden.
“One theme that comes up consistently from patients and caregivers is centered around mental health impact, brain fog, anxiety, depression, and difficulty concentrating,” Bagrodia said. “We wanted to dig into this a little bit further. The idea is to shed light on how common these diagnoses are on these young cancer survivors and intervene so we could positively impact their quality of life.”
The study analyzed 2022 patients with TC and 6375 matched controls enrolled at the US Department of Veterans Affairs (VA) from 1990 through 2016. In the cancer cohort, the mean age at diagnosis was 42.46 years, and ages ranged from 18 to 88 years; 89.7% of patients were White, 6.0% were Black, 2.4% were other race, 1.2% were Asian/Pacific Islander, and 0.7% were Native; 6.2% were Hispanic; and 19.9% were diagnosed between 1990 and 1999.
Factors linked to higher rates of anxiety/depression among patients with TC included divorce (HR 1.15, P = .044), unemployment (HR 1.68, P < .001), and receipt of chemotherapy (HR 1.20, P < .001).
The incidence of de novo anxiety/depression was 30.1% for patients with TC vs 16.7% for controls (P < .001), and the incidence of de novo suicidality was 2.4% for patients and 0.1% for controls.
“These are men who are going to beat their cancer and go on to live for decades and decades,” Bagrodia said. “We found that the impact of a diagnosis and chemotherapy can persist beyond the initial time frame.”
It’s not clear, however, why chemotherapy boosts the risk, Bagrodia said. Clinicians who treat patients with TC should let them know that anxiety, depression, and suicidality are common and treatable concerns.
“We've got some wonderful support services, therapy, and medications that can help out with those diagnoses,” Bagrodia said.
The study authors noted limitations such as the retrospective study design and limited consideration of factors that may affect mental health.
“Additionally, the baseline rates of anxiety, depression, and suicidality are high in the VA population, which may limit ability to apply results to the civilian population,” Bagrodia said.
Genitourinary oncologist Philippe Spiess, MD, of Moffitt Cancer Center in Tampa, praised the study in an interview, saying it provides stronger evidence than previous research.
"It's not only about screening but surveillance, because you never know what kind of challenges they have in their lives,” Spiess told Federal Practitioner, emphasizing the need for clinicians to continue to monitor patients. “They're young, they're vulnerable. Don’t assume they're going to get help somewhere else. You need to be that source that facilitates it.”
No funding is reported. Bagrodia and other authors have no disclosures. Spiess has no disclosures.
Anxiety, depression, and suicide rates are elevated for veterans who are survivors of testicular cancer (TC) compared with veterans without cancer, a retrospective analysis finds.
Over 5 years, the cumulative incidence of anxiety and depression was 53.4% in veterans with TC vs 35.0% in matched controls (P < .001; hazard ratio [HR], 1.66), reported Aditya Bagrodia, MD, professor of urology and radiation oncology at the University of California San Diego, et al in Cancer Medicine. The cumulative incidence of suicidality was 5.0% and 0.1%, respectively (P < .001; HR, 22.99).
“More than half of men with testicular cancer contend with these diagnoses,” Bagrodia told Federal Practitioner. “There are risk factors, including chemotherapy, being single or divorced, or unemployed.”
Patients in these groups warrant aggressive screening and intervention, Bagrodia said. TC is the most common cancer in men in the military and the most common malignancy in men aged 18 to 45 years, Bagrodia said: “The vast majority of men who have testicular cancer are curable.”
Patients, however, face an intense burden.
“One theme that comes up consistently from patients and caregivers is centered around mental health impact, brain fog, anxiety, depression, and difficulty concentrating,” Bagrodia said. “We wanted to dig into this a little bit further. The idea is to shed light on how common these diagnoses are on these young cancer survivors and intervene so we could positively impact their quality of life.”
The study analyzed 2022 patients with TC and 6375 matched controls enrolled at the US Department of Veterans Affairs (VA) from 1990 through 2016. In the cancer cohort, the mean age at diagnosis was 42.46 years, and ages ranged from 18 to 88 years; 89.7% of patients were White, 6.0% were Black, 2.4% were other race, 1.2% were Asian/Pacific Islander, and 0.7% were Native; 6.2% were Hispanic; and 19.9% were diagnosed between 1990 and 1999.
Factors linked to higher rates of anxiety/depression among patients with TC included divorce (HR 1.15, P = .044), unemployment (HR 1.68, P < .001), and receipt of chemotherapy (HR 1.20, P < .001).
The incidence of de novo anxiety/depression was 30.1% for patients with TC vs 16.7% for controls (P < .001), and the incidence of de novo suicidality was 2.4% for patients and 0.1% for controls.
“These are men who are going to beat their cancer and go on to live for decades and decades,” Bagrodia said. “We found that the impact of a diagnosis and chemotherapy can persist beyond the initial time frame.”
It’s not clear, however, why chemotherapy boosts the risk, Bagrodia said. Clinicians who treat patients with TC should let them know that anxiety, depression, and suicidality are common and treatable concerns.
“We've got some wonderful support services, therapy, and medications that can help out with those diagnoses,” Bagrodia said.
The study authors noted limitations such as the retrospective study design and limited consideration of factors that may affect mental health.
“Additionally, the baseline rates of anxiety, depression, and suicidality are high in the VA population, which may limit ability to apply results to the civilian population,” Bagrodia said.
Genitourinary oncologist Philippe Spiess, MD, of Moffitt Cancer Center in Tampa, praised the study in an interview, saying it provides stronger evidence than previous research.
"It's not only about screening but surveillance, because you never know what kind of challenges they have in their lives,” Spiess told Federal Practitioner, emphasizing the need for clinicians to continue to monitor patients. “They're young, they're vulnerable. Don’t assume they're going to get help somewhere else. You need to be that source that facilitates it.”
No funding is reported. Bagrodia and other authors have no disclosures. Spiess has no disclosures.
Anxiety, depression, and suicide rates are elevated for veterans who are survivors of testicular cancer (TC) compared with veterans without cancer, a retrospective analysis finds.
Over 5 years, the cumulative incidence of anxiety and depression was 53.4% in veterans with TC vs 35.0% in matched controls (P < .001; hazard ratio [HR], 1.66), reported Aditya Bagrodia, MD, professor of urology and radiation oncology at the University of California San Diego, et al in Cancer Medicine. The cumulative incidence of suicidality was 5.0% and 0.1%, respectively (P < .001; HR, 22.99).
“More than half of men with testicular cancer contend with these diagnoses,” Bagrodia told Federal Practitioner. “There are risk factors, including chemotherapy, being single or divorced, or unemployed.”
Patients in these groups warrant aggressive screening and intervention, Bagrodia said. TC is the most common cancer in men in the military and the most common malignancy in men aged 18 to 45 years, Bagrodia said: “The vast majority of men who have testicular cancer are curable.”
Patients, however, face an intense burden.
“One theme that comes up consistently from patients and caregivers is centered around mental health impact, brain fog, anxiety, depression, and difficulty concentrating,” Bagrodia said. “We wanted to dig into this a little bit further. The idea is to shed light on how common these diagnoses are on these young cancer survivors and intervene so we could positively impact their quality of life.”
The study analyzed 2022 patients with TC and 6375 matched controls enrolled at the US Department of Veterans Affairs (VA) from 1990 through 2016. In the cancer cohort, the mean age at diagnosis was 42.46 years, and ages ranged from 18 to 88 years; 89.7% of patients were White, 6.0% were Black, 2.4% were other race, 1.2% were Asian/Pacific Islander, and 0.7% were Native; 6.2% were Hispanic; and 19.9% were diagnosed between 1990 and 1999.
Factors linked to higher rates of anxiety/depression among patients with TC included divorce (HR 1.15, P = .044), unemployment (HR 1.68, P < .001), and receipt of chemotherapy (HR 1.20, P < .001).
The incidence of de novo anxiety/depression was 30.1% for patients with TC vs 16.7% for controls (P < .001), and the incidence of de novo suicidality was 2.4% for patients and 0.1% for controls.
“These are men who are going to beat their cancer and go on to live for decades and decades,” Bagrodia said. “We found that the impact of a diagnosis and chemotherapy can persist beyond the initial time frame.”
It’s not clear, however, why chemotherapy boosts the risk, Bagrodia said. Clinicians who treat patients with TC should let them know that anxiety, depression, and suicidality are common and treatable concerns.
“We've got some wonderful support services, therapy, and medications that can help out with those diagnoses,” Bagrodia said.
The study authors noted limitations such as the retrospective study design and limited consideration of factors that may affect mental health.
“Additionally, the baseline rates of anxiety, depression, and suicidality are high in the VA population, which may limit ability to apply results to the civilian population,” Bagrodia said.
Genitourinary oncologist Philippe Spiess, MD, of Moffitt Cancer Center in Tampa, praised the study in an interview, saying it provides stronger evidence than previous research.
"It's not only about screening but surveillance, because you never know what kind of challenges they have in their lives,” Spiess told Federal Practitioner, emphasizing the need for clinicians to continue to monitor patients. “They're young, they're vulnerable. Don’t assume they're going to get help somewhere else. You need to be that source that facilitates it.”
No funding is reported. Bagrodia and other authors have no disclosures. Spiess has no disclosures.
Rising Lung Cancer Burden Among Women
Rising Lung Cancer Burden Among Women
While the incidence of lung cancer is decreasing in men, it continues to rise in women. With more than 19,000 new cases in France each year, lung cancer is now the third most commonly diagnosed cancer among women. This trend is also seen in other European countries but appears to be region-specific because other continents report a decline in incidence among women. Moreover, although overall prognosis remains better in the female population, the trend is worrying: Mortality associated with the disease is increasing in women, unlike in men with lung cancer. A session at the French-Language Pneumology Congress held from January 30 to February 1, 2026, in Lille, France, provided an opportunity to review the situation.
Efficacy and Toxicity
Lung tumors in women have a distinct tumor profile: Women have a higher proportion of adenocarcinomas than men and a higher frequency of somatic mutations (EGFR, BRAF, or HER2), including in nonsmokers. In addition, 65% of lung cancers in women are associated with smoking compared with 87% of those in men.
The role of estrogens is central because they interact directly with tumor growth signaling pathways. Moreover, “sex is the second leading factor of variability in drug pharmacokinetics after weight and accounts for 28% of anticancer drug kinetics,” emphasized Julien Mazières, pulmonologist, Toulouse University Hospital, Toulouse, France. Also involved in this equation are a higher body fat percentage, lower gastric acidity, and, above all, reduced renal and hepatic clearance.
As a result, exposure to drugs — represented by the area under the curve — is often greater in women and translates into not only improved progression-free survival with targeted therapies and chemotherapy but also increased toxicity. Carboplatin and paclitaxel are among the drugs whose kinetics are most affected by clearance. There are differences in clearance of more than 20% for these drugs in women vs men, though dosages are not systematically adjusted except for weight-based dosing. This vulnerability to adverse effects is particularly pronounced with targeted therapies, with more neuropsychiatric and gastrointestinal disorders. Data on the efficacy of immunotherapy in lung cancer by sex are contradictory. However, endocrine-related adverse effects and pneumonitis are more frequent in women, especially before menopause.
Women remain underrepresented in clinical trials, and sex-specific analyses of results are too rarely performed, which limits understanding of mechanisms and prevents tailoring management recommendations according to sex.
Impaired Quality of Life
Lung cancer most severely impairs physical functioning in women. “In the absence of sex-stratified studies, psycho-oncologists’ experience suggests that women have more cognitive disorders, anxiety, and depression associated with this disease. Its impact on quality of life is major, with deterioration of social relationships and reduced treatment adherence,” summarized Céline Mascaux, MD, PhD, pulmonologist, Strasbourg University Hospital, Strasbourg, France. Women also face social and family pressure — a mental burden that pushes them to “hold on” for their loved ones. Regarding sexual health, women with lung cancer who are sexually active often report dissatisfaction with the quality of their sexual relations because of fatigue, lack of energy, sadness, and shortness of breath, not to mention treatment-related sexual dysfunction. These problems are often not given sufficient attention by physicians.
Finally, fertility requires greater attention from the medical community: According to the VICAN study conducted by France’s National Health Insurance Fund, a discussion about fertility preservation did not take place at the time of cancer diagnosis for 60% of men and 67% of women of childbearing age. “In lung cancer specifically, the desire for children nevertheless exists in nearly 40% of patients of childbearing age,” regretted Jacques Cadranel, pulmonologist, Tenon Hospital, Paris, France. This desire does not appear to have influenced therapeutic strategy, and fertility preservation was ultimately proposed in only a third of cases and was carried out in only 3% of women compared with21% of men.
This story has been translated from Univadis France, part of the Medscape Professional Network.
A version of this story first appeared on Medscape.com
While the incidence of lung cancer is decreasing in men, it continues to rise in women. With more than 19,000 new cases in France each year, lung cancer is now the third most commonly diagnosed cancer among women. This trend is also seen in other European countries but appears to be region-specific because other continents report a decline in incidence among women. Moreover, although overall prognosis remains better in the female population, the trend is worrying: Mortality associated with the disease is increasing in women, unlike in men with lung cancer. A session at the French-Language Pneumology Congress held from January 30 to February 1, 2026, in Lille, France, provided an opportunity to review the situation.
Efficacy and Toxicity
Lung tumors in women have a distinct tumor profile: Women have a higher proportion of adenocarcinomas than men and a higher frequency of somatic mutations (EGFR, BRAF, or HER2), including in nonsmokers. In addition, 65% of lung cancers in women are associated with smoking compared with 87% of those in men.
The role of estrogens is central because they interact directly with tumor growth signaling pathways. Moreover, “sex is the second leading factor of variability in drug pharmacokinetics after weight and accounts for 28% of anticancer drug kinetics,” emphasized Julien Mazières, pulmonologist, Toulouse University Hospital, Toulouse, France. Also involved in this equation are a higher body fat percentage, lower gastric acidity, and, above all, reduced renal and hepatic clearance.
As a result, exposure to drugs — represented by the area under the curve — is often greater in women and translates into not only improved progression-free survival with targeted therapies and chemotherapy but also increased toxicity. Carboplatin and paclitaxel are among the drugs whose kinetics are most affected by clearance. There are differences in clearance of more than 20% for these drugs in women vs men, though dosages are not systematically adjusted except for weight-based dosing. This vulnerability to adverse effects is particularly pronounced with targeted therapies, with more neuropsychiatric and gastrointestinal disorders. Data on the efficacy of immunotherapy in lung cancer by sex are contradictory. However, endocrine-related adverse effects and pneumonitis are more frequent in women, especially before menopause.
Women remain underrepresented in clinical trials, and sex-specific analyses of results are too rarely performed, which limits understanding of mechanisms and prevents tailoring management recommendations according to sex.
Impaired Quality of Life
Lung cancer most severely impairs physical functioning in women. “In the absence of sex-stratified studies, psycho-oncologists’ experience suggests that women have more cognitive disorders, anxiety, and depression associated with this disease. Its impact on quality of life is major, with deterioration of social relationships and reduced treatment adherence,” summarized Céline Mascaux, MD, PhD, pulmonologist, Strasbourg University Hospital, Strasbourg, France. Women also face social and family pressure — a mental burden that pushes them to “hold on” for their loved ones. Regarding sexual health, women with lung cancer who are sexually active often report dissatisfaction with the quality of their sexual relations because of fatigue, lack of energy, sadness, and shortness of breath, not to mention treatment-related sexual dysfunction. These problems are often not given sufficient attention by physicians.
Finally, fertility requires greater attention from the medical community: According to the VICAN study conducted by France’s National Health Insurance Fund, a discussion about fertility preservation did not take place at the time of cancer diagnosis for 60% of men and 67% of women of childbearing age. “In lung cancer specifically, the desire for children nevertheless exists in nearly 40% of patients of childbearing age,” regretted Jacques Cadranel, pulmonologist, Tenon Hospital, Paris, France. This desire does not appear to have influenced therapeutic strategy, and fertility preservation was ultimately proposed in only a third of cases and was carried out in only 3% of women compared with21% of men.
This story has been translated from Univadis France, part of the Medscape Professional Network.
A version of this story first appeared on Medscape.com
While the incidence of lung cancer is decreasing in men, it continues to rise in women. With more than 19,000 new cases in France each year, lung cancer is now the third most commonly diagnosed cancer among women. This trend is also seen in other European countries but appears to be region-specific because other continents report a decline in incidence among women. Moreover, although overall prognosis remains better in the female population, the trend is worrying: Mortality associated with the disease is increasing in women, unlike in men with lung cancer. A session at the French-Language Pneumology Congress held from January 30 to February 1, 2026, in Lille, France, provided an opportunity to review the situation.
Efficacy and Toxicity
Lung tumors in women have a distinct tumor profile: Women have a higher proportion of adenocarcinomas than men and a higher frequency of somatic mutations (EGFR, BRAF, or HER2), including in nonsmokers. In addition, 65% of lung cancers in women are associated with smoking compared with 87% of those in men.
The role of estrogens is central because they interact directly with tumor growth signaling pathways. Moreover, “sex is the second leading factor of variability in drug pharmacokinetics after weight and accounts for 28% of anticancer drug kinetics,” emphasized Julien Mazières, pulmonologist, Toulouse University Hospital, Toulouse, France. Also involved in this equation are a higher body fat percentage, lower gastric acidity, and, above all, reduced renal and hepatic clearance.
As a result, exposure to drugs — represented by the area under the curve — is often greater in women and translates into not only improved progression-free survival with targeted therapies and chemotherapy but also increased toxicity. Carboplatin and paclitaxel are among the drugs whose kinetics are most affected by clearance. There are differences in clearance of more than 20% for these drugs in women vs men, though dosages are not systematically adjusted except for weight-based dosing. This vulnerability to adverse effects is particularly pronounced with targeted therapies, with more neuropsychiatric and gastrointestinal disorders. Data on the efficacy of immunotherapy in lung cancer by sex are contradictory. However, endocrine-related adverse effects and pneumonitis are more frequent in women, especially before menopause.
Women remain underrepresented in clinical trials, and sex-specific analyses of results are too rarely performed, which limits understanding of mechanisms and prevents tailoring management recommendations according to sex.
Impaired Quality of Life
Lung cancer most severely impairs physical functioning in women. “In the absence of sex-stratified studies, psycho-oncologists’ experience suggests that women have more cognitive disorders, anxiety, and depression associated with this disease. Its impact on quality of life is major, with deterioration of social relationships and reduced treatment adherence,” summarized Céline Mascaux, MD, PhD, pulmonologist, Strasbourg University Hospital, Strasbourg, France. Women also face social and family pressure — a mental burden that pushes them to “hold on” for their loved ones. Regarding sexual health, women with lung cancer who are sexually active often report dissatisfaction with the quality of their sexual relations because of fatigue, lack of energy, sadness, and shortness of breath, not to mention treatment-related sexual dysfunction. These problems are often not given sufficient attention by physicians.
Finally, fertility requires greater attention from the medical community: According to the VICAN study conducted by France’s National Health Insurance Fund, a discussion about fertility preservation did not take place at the time of cancer diagnosis for 60% of men and 67% of women of childbearing age. “In lung cancer specifically, the desire for children nevertheless exists in nearly 40% of patients of childbearing age,” regretted Jacques Cadranel, pulmonologist, Tenon Hospital, Paris, France. This desire does not appear to have influenced therapeutic strategy, and fertility preservation was ultimately proposed in only a third of cases and was carried out in only 3% of women compared with21% of men.
This story has been translated from Univadis France, part of the Medscape Professional Network.
A version of this story first appeared on Medscape.com
Rising Lung Cancer Burden Among Women
Rising Lung Cancer Burden Among Women
Remote Program Doubles Metastatic Prostate Cancer Germline Testing
A pilot program appeared to more than double the rate of germline genetic testing among veterans with metastatic prostate cancer (mPC) by using remote communication rather than relying on clinicians for in-person outreach to patients.
Of 1952 veterans with mPC, 681 (34.9%) provided consent and 459 (23.5%) completed testing, exceeding the usual 10% to 12% of patients who undergo testing, reported Bruce Montgomery, MD, et al in Cancer.
Although testing is recommended for all patients with mPC to guide therapy and alert relatives who may be at risk, 23.5% is still an impressive number, Montgomery, an oncologist with Veterans Affairs (VA) Puget Sound Health Care System in Seattle told Federal Practitioner: “With a letter and very little money and very little real time from clinicians, we could get testing done at 3 times the rate happening out there in the big wide world,” he said. “For 2000 patients, we needed one research coordinator and a small part of a genetic counselor's time.”
According to the study, germline genetic testing—which examines inherited DNA—is now recommended for all men with mPC by the National Comprehensive Cancer Network, the American Society of Clinical Oncology, and the American Urological Association. Germline genetic testing differs from somatic testing, which seeks genetic changes in the tumors themselves.
In the VA and community at large, the percentage of men with mPC who undergo germline genetic testing is low, Montgomery said. Research suggests < 40% of patients undergo somatic testing.
Germline genetic testing only costs about 10% compared with somatic testing, Montgomery said, and can be conducted at any time. In about 10% of mPC cases, the testing provides insight into the best treatment, he said.
Montgomery noted another benefit to germline genetic testing: It can raise the alarm about pathogenic variants that could boost cancer risk in family members, allowing them to get screened and take action.
There are many reasons veterans do not get tested, Montgomery said. The process is not automatic because patient consent is needed, and clinicians often fail to ask. In some cases, veterans worry about privacy or whether they will lose service-connected benefits if their cancer is blamed on genetics.
The study focused on 2104 veterans with mPC who had already agreed to take part in the Million Veteran Program, a prospective cohort study examining genetic and nongenetic risk for disease. The genetic analysis from that project did not provide guidance about mPC, so researchers approached the veterans directly.
Patients were enrolled from February 2021 to October 2023. A total of 1952 veterans did not opt out when contacted by mail (median age, 75 years; 63% White, 25% Black; 74% urban and 24% rural). The median age of those who consented and completed testing after phone contact was 74 years; 67% of patients were White and 22% were Black; 78% of patients lived in urban communities and 20% lived in rural communities.
Fifty-nine patients (13%) had pathogenic variants, and 37 of those had variants that indicated treatment with targeted therapies. Of the 37, 14 received targeted therapy, 18 were not at the point where targeted therapy was indicated, and 5 were not treated with targeted therapy for various reasons before they died.
Twelve of the 59 patients with pathogenic variants agreed to let the study team contact their first-degree relatives. Thirty relatives underwent testing, and 10 of them were positive for the variants.
Following completion of the study, researchers examined electronic records for the 59 patients with pathogenic variants and found that 19% did not have documentation of the germline finding in the medical record. The authors cited an “urgent need” to standardize where genetic information is included in the records.
While “it seems like a very small number of patients took up testing,” Montgomery said, the study findings are promising: “If we did the same thing nationally in the VA, there would be 15,000 men with metastatic disease, and we’d be testing 5000 of them with almost no effort.”
In an interview, Susan Vadaparampil, PhD, MPH, associate center director of Community Outreach and Engagement at Moffitt Cancer Center, who studies genetic testing, praised the strengths of the study. Vadaparampil, who did not take part in the research, told Federal Practitioner that the study relies on “an intervention that could likely be incorporated into routine clinical practice, a less resource-intensive model that provides posttest counseling for those who test positive, and support to share results with family members.”
However, she said, “testing uptake was uneven based on participant sociodemographic characteristics. It's important to consider how discussions and resources to facilitate testing may need to be adapted to meet the needs of all patients.
“Strategies that facilitate clinicians’ knowledge, comfort, and consistency in discussing testing with all mPC patients are essential,” Vadaparampil added. “Simultaneously using multiple strategies targeted to different levels can further help boost uptake.”
The study was funded by the VA Office of Research and Development, Prostate Cancer Foundation, Pacific Northwest Prostate Cancer SPORE, Institute for Prostate Cancer Research, Congressionally Directed Medical Research Programs (CDMRP), and Put VA Data to Work for Veterans.
Montgomery discloses relationships with Daiichi Sankyo, INmune Bio, Clovis, Janssen Pharmaceuticals, Johnson and Johnson, and Merck. Some other authors report various disclosures. Vadaparampil has no disclosures.
A pilot program appeared to more than double the rate of germline genetic testing among veterans with metastatic prostate cancer (mPC) by using remote communication rather than relying on clinicians for in-person outreach to patients.
Of 1952 veterans with mPC, 681 (34.9%) provided consent and 459 (23.5%) completed testing, exceeding the usual 10% to 12% of patients who undergo testing, reported Bruce Montgomery, MD, et al in Cancer.
Although testing is recommended for all patients with mPC to guide therapy and alert relatives who may be at risk, 23.5% is still an impressive number, Montgomery, an oncologist with Veterans Affairs (VA) Puget Sound Health Care System in Seattle told Federal Practitioner: “With a letter and very little money and very little real time from clinicians, we could get testing done at 3 times the rate happening out there in the big wide world,” he said. “For 2000 patients, we needed one research coordinator and a small part of a genetic counselor's time.”
According to the study, germline genetic testing—which examines inherited DNA—is now recommended for all men with mPC by the National Comprehensive Cancer Network, the American Society of Clinical Oncology, and the American Urological Association. Germline genetic testing differs from somatic testing, which seeks genetic changes in the tumors themselves.
In the VA and community at large, the percentage of men with mPC who undergo germline genetic testing is low, Montgomery said. Research suggests < 40% of patients undergo somatic testing.
Germline genetic testing only costs about 10% compared with somatic testing, Montgomery said, and can be conducted at any time. In about 10% of mPC cases, the testing provides insight into the best treatment, he said.
Montgomery noted another benefit to germline genetic testing: It can raise the alarm about pathogenic variants that could boost cancer risk in family members, allowing them to get screened and take action.
There are many reasons veterans do not get tested, Montgomery said. The process is not automatic because patient consent is needed, and clinicians often fail to ask. In some cases, veterans worry about privacy or whether they will lose service-connected benefits if their cancer is blamed on genetics.
The study focused on 2104 veterans with mPC who had already agreed to take part in the Million Veteran Program, a prospective cohort study examining genetic and nongenetic risk for disease. The genetic analysis from that project did not provide guidance about mPC, so researchers approached the veterans directly.
Patients were enrolled from February 2021 to October 2023. A total of 1952 veterans did not opt out when contacted by mail (median age, 75 years; 63% White, 25% Black; 74% urban and 24% rural). The median age of those who consented and completed testing after phone contact was 74 years; 67% of patients were White and 22% were Black; 78% of patients lived in urban communities and 20% lived in rural communities.
Fifty-nine patients (13%) had pathogenic variants, and 37 of those had variants that indicated treatment with targeted therapies. Of the 37, 14 received targeted therapy, 18 were not at the point where targeted therapy was indicated, and 5 were not treated with targeted therapy for various reasons before they died.
Twelve of the 59 patients with pathogenic variants agreed to let the study team contact their first-degree relatives. Thirty relatives underwent testing, and 10 of them were positive for the variants.
Following completion of the study, researchers examined electronic records for the 59 patients with pathogenic variants and found that 19% did not have documentation of the germline finding in the medical record. The authors cited an “urgent need” to standardize where genetic information is included in the records.
While “it seems like a very small number of patients took up testing,” Montgomery said, the study findings are promising: “If we did the same thing nationally in the VA, there would be 15,000 men with metastatic disease, and we’d be testing 5000 of them with almost no effort.”
In an interview, Susan Vadaparampil, PhD, MPH, associate center director of Community Outreach and Engagement at Moffitt Cancer Center, who studies genetic testing, praised the strengths of the study. Vadaparampil, who did not take part in the research, told Federal Practitioner that the study relies on “an intervention that could likely be incorporated into routine clinical practice, a less resource-intensive model that provides posttest counseling for those who test positive, and support to share results with family members.”
However, she said, “testing uptake was uneven based on participant sociodemographic characteristics. It's important to consider how discussions and resources to facilitate testing may need to be adapted to meet the needs of all patients.
“Strategies that facilitate clinicians’ knowledge, comfort, and consistency in discussing testing with all mPC patients are essential,” Vadaparampil added. “Simultaneously using multiple strategies targeted to different levels can further help boost uptake.”
The study was funded by the VA Office of Research and Development, Prostate Cancer Foundation, Pacific Northwest Prostate Cancer SPORE, Institute for Prostate Cancer Research, Congressionally Directed Medical Research Programs (CDMRP), and Put VA Data to Work for Veterans.
Montgomery discloses relationships with Daiichi Sankyo, INmune Bio, Clovis, Janssen Pharmaceuticals, Johnson and Johnson, and Merck. Some other authors report various disclosures. Vadaparampil has no disclosures.
A pilot program appeared to more than double the rate of germline genetic testing among veterans with metastatic prostate cancer (mPC) by using remote communication rather than relying on clinicians for in-person outreach to patients.
Of 1952 veterans with mPC, 681 (34.9%) provided consent and 459 (23.5%) completed testing, exceeding the usual 10% to 12% of patients who undergo testing, reported Bruce Montgomery, MD, et al in Cancer.
Although testing is recommended for all patients with mPC to guide therapy and alert relatives who may be at risk, 23.5% is still an impressive number, Montgomery, an oncologist with Veterans Affairs (VA) Puget Sound Health Care System in Seattle told Federal Practitioner: “With a letter and very little money and very little real time from clinicians, we could get testing done at 3 times the rate happening out there in the big wide world,” he said. “For 2000 patients, we needed one research coordinator and a small part of a genetic counselor's time.”
According to the study, germline genetic testing—which examines inherited DNA—is now recommended for all men with mPC by the National Comprehensive Cancer Network, the American Society of Clinical Oncology, and the American Urological Association. Germline genetic testing differs from somatic testing, which seeks genetic changes in the tumors themselves.
In the VA and community at large, the percentage of men with mPC who undergo germline genetic testing is low, Montgomery said. Research suggests < 40% of patients undergo somatic testing.
Germline genetic testing only costs about 10% compared with somatic testing, Montgomery said, and can be conducted at any time. In about 10% of mPC cases, the testing provides insight into the best treatment, he said.
Montgomery noted another benefit to germline genetic testing: It can raise the alarm about pathogenic variants that could boost cancer risk in family members, allowing them to get screened and take action.
There are many reasons veterans do not get tested, Montgomery said. The process is not automatic because patient consent is needed, and clinicians often fail to ask. In some cases, veterans worry about privacy or whether they will lose service-connected benefits if their cancer is blamed on genetics.
The study focused on 2104 veterans with mPC who had already agreed to take part in the Million Veteran Program, a prospective cohort study examining genetic and nongenetic risk for disease. The genetic analysis from that project did not provide guidance about mPC, so researchers approached the veterans directly.
Patients were enrolled from February 2021 to October 2023. A total of 1952 veterans did not opt out when contacted by mail (median age, 75 years; 63% White, 25% Black; 74% urban and 24% rural). The median age of those who consented and completed testing after phone contact was 74 years; 67% of patients were White and 22% were Black; 78% of patients lived in urban communities and 20% lived in rural communities.
Fifty-nine patients (13%) had pathogenic variants, and 37 of those had variants that indicated treatment with targeted therapies. Of the 37, 14 received targeted therapy, 18 were not at the point where targeted therapy was indicated, and 5 were not treated with targeted therapy for various reasons before they died.
Twelve of the 59 patients with pathogenic variants agreed to let the study team contact their first-degree relatives. Thirty relatives underwent testing, and 10 of them were positive for the variants.
Following completion of the study, researchers examined electronic records for the 59 patients with pathogenic variants and found that 19% did not have documentation of the germline finding in the medical record. The authors cited an “urgent need” to standardize where genetic information is included in the records.
While “it seems like a very small number of patients took up testing,” Montgomery said, the study findings are promising: “If we did the same thing nationally in the VA, there would be 15,000 men with metastatic disease, and we’d be testing 5000 of them with almost no effort.”
In an interview, Susan Vadaparampil, PhD, MPH, associate center director of Community Outreach and Engagement at Moffitt Cancer Center, who studies genetic testing, praised the strengths of the study. Vadaparampil, who did not take part in the research, told Federal Practitioner that the study relies on “an intervention that could likely be incorporated into routine clinical practice, a less resource-intensive model that provides posttest counseling for those who test positive, and support to share results with family members.”
However, she said, “testing uptake was uneven based on participant sociodemographic characteristics. It's important to consider how discussions and resources to facilitate testing may need to be adapted to meet the needs of all patients.
“Strategies that facilitate clinicians’ knowledge, comfort, and consistency in discussing testing with all mPC patients are essential,” Vadaparampil added. “Simultaneously using multiple strategies targeted to different levels can further help boost uptake.”
The study was funded by the VA Office of Research and Development, Prostate Cancer Foundation, Pacific Northwest Prostate Cancer SPORE, Institute for Prostate Cancer Research, Congressionally Directed Medical Research Programs (CDMRP), and Put VA Data to Work for Veterans.
Montgomery discloses relationships with Daiichi Sankyo, INmune Bio, Clovis, Janssen Pharmaceuticals, Johnson and Johnson, and Merck. Some other authors report various disclosures. Vadaparampil has no disclosures.
Diet and Cancer: Here's What I Tell Patients
Diet and Cancer: Here's What I Tell Patients
One of the most common questions my patients ask is, “What diet can help me beat this cancer?” It is a profoundly important question that is worthy of our efforts to answer. In this brief essay, I will take a deep dive into this question in depth and explore the broader clinical and scientific themes it brings into play.
Low-Hanging Fruit: Nutrition Science
A cancer diagnosis can be a deeply disempowering experience. Although I have not lived with cancer myself, I have seen this play out repeatedly over the past 5 years in my role as an oncologist treating patients with hematologic malignancies.
Our diet is an important part of our personal identity, culturally and spiritually. If lifestyle changes, such as a modified diet or more exercise, can contribute to cancer treatment, it may help us regain a sense of control over our lives, one that cancer so often cruelly strips away. I hypothesize that, among other factors, this is why diet is so important to our patients.
Another factor is exposure to a compelling diet-cancer narrative. Nearly every day, a media headline appears claiming that eating a particular food, or drinking coffee, can either increase or decrease your risk for a certain disease.
These claims, however, are often based on studies of large observational datasets where individuals fill out surveys about their dietary habits and are subsequently assessed for disease outcomes. In these studies, people aren’t asked to eat a particular diet; instead, their dietary habits are analyzed by researchers who have endless permutations to explore. This, in a nutshell, is the field of nutritional epidemiology.
In my opinion, nutritional epidemiology represents the collision of the well-intentioned effort to answer clinically meaningful questions with the ease — and near-infinite permutations — of dietary questions that can be asked from an increasingly larger number of different datasets.
Now, factor in the never-ending appetite (pun intended) of journalism and the public’s desire for dietary studies, and you create the perfect storm of incentives that drives a flood of low-quality nutritional science. These studies are highly malleable to analytical choices and can essentially produce results consistent with your prior beliefs, regardless of the philosophical inclination you have (pro keto-diet, pro-vegan, etc.). I love quoting this study to my trainees that, depending on what variables are included and how the analysis is conducted, the same dataset could be used to show that red meat either increases, decreases, or has no effect on all-cause mortality. Unfortunately, much of the evidence base for diet in cancer comes from similarly confounded, low-quality studies.
Diet and Cancer
So, what do randomized trials show for diet and cancer?
The highest-quality evidence is generated from randomized controlled trials. One of their key advantages is the ability to control both measured and unmeasured confounders.
Unfortunately, the evidence supporting diet as an anticancer modality in randomized trials in patients with cancer is bleak. We did a systematic review of all randomized trials of dietary intervention ever done in patients with cancer. Most of the trials measured outcomes such as feasibility (often small pilot studies that measure variables such as weight changes or lab values). The trials that measure clinical endpoints, such as survival, were largely negative and demonstrated no meaningful effect of diet on outcomes. Take trials exploring whether a Mediterranean diet helps prevent breast cancer recurrence, or whether a diet rich in fruits and vegetables improves prostate cancer outcomes. Although these diets may offer benefits, these studies found that specific diets did not change the natural history of cancer.
Myeloma and Diet
In my specialty, multiple myeloma, I am thankful that some trials are beginning to shed light on whether diet influences cancer outcomes.
One study, which was recently published in Cancer Discovery, explored whether a high-fiber, plant-based diet could potentially slow or delay progression from myeloma precursor conditions toward full-blown multiple myeloma. The trial enrolled 23 participants, with the primary endpoints of dietary adherence and changes to BMI. Measures of progression to multiple myeloma were exploratory at best. Yet, the media coverage, as well as the majority of the discussion and results sections of this study manuscript, claimed that the diet changes can prevent progression to myeloma.
However, the study design and conclusions were flawed. The paper focused on two patients who had some improvement in disease trajectory, while descriptions of patients who had an increase in their bone marrow plasma cell percentage were relegated to the supplemental section.
As a primary investigator of a trial in smoldering myeloma where we use advanced imaging as an alternative to pharmacologic treatment, I frequently see myeloma markers fluctuate and often decrease. I attribute these changes to random variation, or possibly regression to the mean, rather than the effect of any intervention.
Future randomized studies by this group used primary endpoints of stool butyrate level and implement dietary interventions for a limited period— 2 weeks in one study and 12 weeks in another — to again assess the impact of a high-fiber, plant-based diet on progression to myeloma. Although there are no data yet, the limited timeframes in these studies severely limits generalizability for outcomes that would truly matter, such as cancer control and longevity. There is also no evidence that changes in stool butyrate levels influence patient outcomes.
High-quality science — whether it is evaluating diet or other interventions—requires high-quality data, effort, funding, and time. It is not impossible.
We can draw inspiration from the CHALLENGE trial. This large, randomized trial, which took over a decade to complete, assessed the benefit of a structured exercise program in the adjuvant setting for colon cancer. The endpoint of this study was disease-free survival, and the intervention was deployed over a much longer period: 3 years, as opposed to a 2-week intervention. This trial took years from inception to completion, but it yielded a conclusive result and will probably lead to more dedicated efforts to facilitate exercise programs for patients with cancer.
Our patients deserve the same effort as the CHALLENGE trial to answer their important dietary questions. Until such trials are completed, we must acknowledge, with humility, that despite the common sense and feel-good factor that many diets offer us, their impact on cancer remains uncertain.
Conversely, we must recognize that even if diet does not cure or alter the course of a certain cancer, it can still impact quality of life, treatment tolerance, and other supportive care outcomes, making it an important factor in patient care.
This is what I tell my patients that it is unlikely any one diet will change the trajectory of your cancer. Focus on eating healthy, and remember that most things in moderation are fine. Your diet remains an important risk factor and determinant for health outcomes beyond cancer. Eat what makes you happy. You are going through a tough time, and this is not the moment to impose stringent restrictions on yourself.
A version of this article first appeared on Medscape.com.
One of the most common questions my patients ask is, “What diet can help me beat this cancer?” It is a profoundly important question that is worthy of our efforts to answer. In this brief essay, I will take a deep dive into this question in depth and explore the broader clinical and scientific themes it brings into play.
Low-Hanging Fruit: Nutrition Science
A cancer diagnosis can be a deeply disempowering experience. Although I have not lived with cancer myself, I have seen this play out repeatedly over the past 5 years in my role as an oncologist treating patients with hematologic malignancies.
Our diet is an important part of our personal identity, culturally and spiritually. If lifestyle changes, such as a modified diet or more exercise, can contribute to cancer treatment, it may help us regain a sense of control over our lives, one that cancer so often cruelly strips away. I hypothesize that, among other factors, this is why diet is so important to our patients.
Another factor is exposure to a compelling diet-cancer narrative. Nearly every day, a media headline appears claiming that eating a particular food, or drinking coffee, can either increase or decrease your risk for a certain disease.
These claims, however, are often based on studies of large observational datasets where individuals fill out surveys about their dietary habits and are subsequently assessed for disease outcomes. In these studies, people aren’t asked to eat a particular diet; instead, their dietary habits are analyzed by researchers who have endless permutations to explore. This, in a nutshell, is the field of nutritional epidemiology.
In my opinion, nutritional epidemiology represents the collision of the well-intentioned effort to answer clinically meaningful questions with the ease — and near-infinite permutations — of dietary questions that can be asked from an increasingly larger number of different datasets.
Now, factor in the never-ending appetite (pun intended) of journalism and the public’s desire for dietary studies, and you create the perfect storm of incentives that drives a flood of low-quality nutritional science. These studies are highly malleable to analytical choices and can essentially produce results consistent with your prior beliefs, regardless of the philosophical inclination you have (pro keto-diet, pro-vegan, etc.). I love quoting this study to my trainees that, depending on what variables are included and how the analysis is conducted, the same dataset could be used to show that red meat either increases, decreases, or has no effect on all-cause mortality. Unfortunately, much of the evidence base for diet in cancer comes from similarly confounded, low-quality studies.
Diet and Cancer
So, what do randomized trials show for diet and cancer?
The highest-quality evidence is generated from randomized controlled trials. One of their key advantages is the ability to control both measured and unmeasured confounders.
Unfortunately, the evidence supporting diet as an anticancer modality in randomized trials in patients with cancer is bleak. We did a systematic review of all randomized trials of dietary intervention ever done in patients with cancer. Most of the trials measured outcomes such as feasibility (often small pilot studies that measure variables such as weight changes or lab values). The trials that measure clinical endpoints, such as survival, were largely negative and demonstrated no meaningful effect of diet on outcomes. Take trials exploring whether a Mediterranean diet helps prevent breast cancer recurrence, or whether a diet rich in fruits and vegetables improves prostate cancer outcomes. Although these diets may offer benefits, these studies found that specific diets did not change the natural history of cancer.
Myeloma and Diet
In my specialty, multiple myeloma, I am thankful that some trials are beginning to shed light on whether diet influences cancer outcomes.
One study, which was recently published in Cancer Discovery, explored whether a high-fiber, plant-based diet could potentially slow or delay progression from myeloma precursor conditions toward full-blown multiple myeloma. The trial enrolled 23 participants, with the primary endpoints of dietary adherence and changes to BMI. Measures of progression to multiple myeloma were exploratory at best. Yet, the media coverage, as well as the majority of the discussion and results sections of this study manuscript, claimed that the diet changes can prevent progression to myeloma.
However, the study design and conclusions were flawed. The paper focused on two patients who had some improvement in disease trajectory, while descriptions of patients who had an increase in their bone marrow plasma cell percentage were relegated to the supplemental section.
As a primary investigator of a trial in smoldering myeloma where we use advanced imaging as an alternative to pharmacologic treatment, I frequently see myeloma markers fluctuate and often decrease. I attribute these changes to random variation, or possibly regression to the mean, rather than the effect of any intervention.
Future randomized studies by this group used primary endpoints of stool butyrate level and implement dietary interventions for a limited period— 2 weeks in one study and 12 weeks in another — to again assess the impact of a high-fiber, plant-based diet on progression to myeloma. Although there are no data yet, the limited timeframes in these studies severely limits generalizability for outcomes that would truly matter, such as cancer control and longevity. There is also no evidence that changes in stool butyrate levels influence patient outcomes.
High-quality science — whether it is evaluating diet or other interventions—requires high-quality data, effort, funding, and time. It is not impossible.
We can draw inspiration from the CHALLENGE trial. This large, randomized trial, which took over a decade to complete, assessed the benefit of a structured exercise program in the adjuvant setting for colon cancer. The endpoint of this study was disease-free survival, and the intervention was deployed over a much longer period: 3 years, as opposed to a 2-week intervention. This trial took years from inception to completion, but it yielded a conclusive result and will probably lead to more dedicated efforts to facilitate exercise programs for patients with cancer.
Our patients deserve the same effort as the CHALLENGE trial to answer their important dietary questions. Until such trials are completed, we must acknowledge, with humility, that despite the common sense and feel-good factor that many diets offer us, their impact on cancer remains uncertain.
Conversely, we must recognize that even if diet does not cure or alter the course of a certain cancer, it can still impact quality of life, treatment tolerance, and other supportive care outcomes, making it an important factor in patient care.
This is what I tell my patients that it is unlikely any one diet will change the trajectory of your cancer. Focus on eating healthy, and remember that most things in moderation are fine. Your diet remains an important risk factor and determinant for health outcomes beyond cancer. Eat what makes you happy. You are going through a tough time, and this is not the moment to impose stringent restrictions on yourself.
A version of this article first appeared on Medscape.com.
One of the most common questions my patients ask is, “What diet can help me beat this cancer?” It is a profoundly important question that is worthy of our efforts to answer. In this brief essay, I will take a deep dive into this question in depth and explore the broader clinical and scientific themes it brings into play.
Low-Hanging Fruit: Nutrition Science
A cancer diagnosis can be a deeply disempowering experience. Although I have not lived with cancer myself, I have seen this play out repeatedly over the past 5 years in my role as an oncologist treating patients with hematologic malignancies.
Our diet is an important part of our personal identity, culturally and spiritually. If lifestyle changes, such as a modified diet or more exercise, can contribute to cancer treatment, it may help us regain a sense of control over our lives, one that cancer so often cruelly strips away. I hypothesize that, among other factors, this is why diet is so important to our patients.
Another factor is exposure to a compelling diet-cancer narrative. Nearly every day, a media headline appears claiming that eating a particular food, or drinking coffee, can either increase or decrease your risk for a certain disease.
These claims, however, are often based on studies of large observational datasets where individuals fill out surveys about their dietary habits and are subsequently assessed for disease outcomes. In these studies, people aren’t asked to eat a particular diet; instead, their dietary habits are analyzed by researchers who have endless permutations to explore. This, in a nutshell, is the field of nutritional epidemiology.
In my opinion, nutritional epidemiology represents the collision of the well-intentioned effort to answer clinically meaningful questions with the ease — and near-infinite permutations — of dietary questions that can be asked from an increasingly larger number of different datasets.
Now, factor in the never-ending appetite (pun intended) of journalism and the public’s desire for dietary studies, and you create the perfect storm of incentives that drives a flood of low-quality nutritional science. These studies are highly malleable to analytical choices and can essentially produce results consistent with your prior beliefs, regardless of the philosophical inclination you have (pro keto-diet, pro-vegan, etc.). I love quoting this study to my trainees that, depending on what variables are included and how the analysis is conducted, the same dataset could be used to show that red meat either increases, decreases, or has no effect on all-cause mortality. Unfortunately, much of the evidence base for diet in cancer comes from similarly confounded, low-quality studies.
Diet and Cancer
So, what do randomized trials show for diet and cancer?
The highest-quality evidence is generated from randomized controlled trials. One of their key advantages is the ability to control both measured and unmeasured confounders.
Unfortunately, the evidence supporting diet as an anticancer modality in randomized trials in patients with cancer is bleak. We did a systematic review of all randomized trials of dietary intervention ever done in patients with cancer. Most of the trials measured outcomes such as feasibility (often small pilot studies that measure variables such as weight changes or lab values). The trials that measure clinical endpoints, such as survival, were largely negative and demonstrated no meaningful effect of diet on outcomes. Take trials exploring whether a Mediterranean diet helps prevent breast cancer recurrence, or whether a diet rich in fruits and vegetables improves prostate cancer outcomes. Although these diets may offer benefits, these studies found that specific diets did not change the natural history of cancer.
Myeloma and Diet
In my specialty, multiple myeloma, I am thankful that some trials are beginning to shed light on whether diet influences cancer outcomes.
One study, which was recently published in Cancer Discovery, explored whether a high-fiber, plant-based diet could potentially slow or delay progression from myeloma precursor conditions toward full-blown multiple myeloma. The trial enrolled 23 participants, with the primary endpoints of dietary adherence and changes to BMI. Measures of progression to multiple myeloma were exploratory at best. Yet, the media coverage, as well as the majority of the discussion and results sections of this study manuscript, claimed that the diet changes can prevent progression to myeloma.
However, the study design and conclusions were flawed. The paper focused on two patients who had some improvement in disease trajectory, while descriptions of patients who had an increase in their bone marrow plasma cell percentage were relegated to the supplemental section.
As a primary investigator of a trial in smoldering myeloma where we use advanced imaging as an alternative to pharmacologic treatment, I frequently see myeloma markers fluctuate and often decrease. I attribute these changes to random variation, or possibly regression to the mean, rather than the effect of any intervention.
Future randomized studies by this group used primary endpoints of stool butyrate level and implement dietary interventions for a limited period— 2 weeks in one study and 12 weeks in another — to again assess the impact of a high-fiber, plant-based diet on progression to myeloma. Although there are no data yet, the limited timeframes in these studies severely limits generalizability for outcomes that would truly matter, such as cancer control and longevity. There is also no evidence that changes in stool butyrate levels influence patient outcomes.
High-quality science — whether it is evaluating diet or other interventions—requires high-quality data, effort, funding, and time. It is not impossible.
We can draw inspiration from the CHALLENGE trial. This large, randomized trial, which took over a decade to complete, assessed the benefit of a structured exercise program in the adjuvant setting for colon cancer. The endpoint of this study was disease-free survival, and the intervention was deployed over a much longer period: 3 years, as opposed to a 2-week intervention. This trial took years from inception to completion, but it yielded a conclusive result and will probably lead to more dedicated efforts to facilitate exercise programs for patients with cancer.
Our patients deserve the same effort as the CHALLENGE trial to answer their important dietary questions. Until such trials are completed, we must acknowledge, with humility, that despite the common sense and feel-good factor that many diets offer us, their impact on cancer remains uncertain.
Conversely, we must recognize that even if diet does not cure or alter the course of a certain cancer, it can still impact quality of life, treatment tolerance, and other supportive care outcomes, making it an important factor in patient care.
This is what I tell my patients that it is unlikely any one diet will change the trajectory of your cancer. Focus on eating healthy, and remember that most things in moderation are fine. Your diet remains an important risk factor and determinant for health outcomes beyond cancer. Eat what makes you happy. You are going through a tough time, and this is not the moment to impose stringent restrictions on yourself.
A version of this article first appeared on Medscape.com.
Diet and Cancer: Here's What I Tell Patients
Diet and Cancer: Here's What I Tell Patients
Housing Support May Boost CRC Screening in Vets Experiencing Homelessness
TOPLINE: Among Veterans Health Administration (VHA) patients experiencing homelessness, gaining housing is linked to higher 24-month colorectal (CRC) and breast cancer screening completion. In cohorts of 117,619 veterans eligible for colorectal screening and 6517 veterans eligible for breast cancer screening veterans, screening occurs in 36.1% and 47.9% after housing gain vs 18.8% and 23.7% if homelessness persists.
METHODOLOGY
A retrospective cohort study examined all veterans experiencing homelessness who received care at the VHA from 2011 to 2021 and were eligible for but not up to date on CRC and breast cancer screening.
117,619 veterans experiencing homelessness were eligible for but not up to date on CRC screening (aged 50-75 years without prior cancer diagnosis, inflammatory bowel disease, or colectomy) and 6517 veterans experiencing homelessness were eligible for but not up to date on breast cancer screening (women aged 50-75 years without prior cancer diagnosis, lumpectomy, or mastectomy) were included at their index clinic visit.
Exposure was defined as gaining housing within 24 months following index clinic visit, identified through the Homeless Screening Clinical Reminder, US Department of Veterans Affairs (VA) Homeless Operations, Management, and Evaluation System assessments, or US Department of Housing and Urban Development—VA Supportive Housing program move-in dates.
Primary outcome were undergoing screening for CRC (colonoscopy, flexible sigmoidoscopy, computed tomography colonography, barium enema, or stool-based study) or breast cancer (mammogram) that was at a VHA facility or paid by VA within 24 months following index clinic visit.
TAKEAWAY
Among veterans who gained housing, 36.1% underwent CRC screening and 47.9% underwent breast cancer screening during the 24-month observation period, compared with 18.8% and 23.7% of veterans, respectively, among those who remained homeless.
Veterans who gained housing had 2.3 times the adjusted hazard ratio (aHR) of undergoing CRC screening compared with those who remained homeless (AHR, 2.3; 95% CI, 2.2-2.3; P < .001).
Veterans who gained housing had 2.4 times the adjusted hazard of undergoing breast cancer screening compared with those who remained homeless (AHR, 2.4; 95% CI, 2.2-2.7; P < .001).
Median (interquartile range [IQR]) time from index visit to cancer screening was 8 months (4-15) for CRC screening and 8 months (3-14) for breast cancer screening; median (IQR) time from gaining housing to screening was 4 months (1-9) and 3 months (1-8), respectively.
IN PRACTICE: Veterans experiencing homelessness who gain housing have higher rates of cancer screening. “This finding supports promotion of housing to improve health outcomes for homeless individuals," wrote the authors of the study.
SOURCE: The study was led by researchers at the University of California, San Francisco. It was published online in Annals of Family Medicine.
LIMITATIONS: Residual unmeasured confounding was likely due to the observational design of this study, because veterans able to navigate services to obtain housing may also be more likely to complete preventive care. Housing transitions may be misclassified because the Homeless Screening Clinical Reminder was not designed to track changes and may not be administered to veterans already identified as experiencing homelessness. The study did not capture data for screening completed outside VHA or that was not paid for by it. The study cohort only includes veterans with VHA contact, which may limit generalizability.
DISCLOSURES: Benioff Homelessness and Housing Initiative provided grant support for the work; Project Grant K24AG046372 was also awarded to Kushel for the study. Decker is a National Clinician Scholar with salary support from the US Department of Veterans Affairs and reported receiving personal fees from Moon Surgical. Kanzaria and Kushel are faculty members of the Benioff Homelessness and Housing Initiative; Kanzaria also reported advisory work for Amae Health. Kushel is listed as serving on boards including Housing California, National Homelessness Law Center, and Steinberg Institute; other authors reported no conflicts.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: Among Veterans Health Administration (VHA) patients experiencing homelessness, gaining housing is linked to higher 24-month colorectal (CRC) and breast cancer screening completion. In cohorts of 117,619 veterans eligible for colorectal screening and 6517 veterans eligible for breast cancer screening veterans, screening occurs in 36.1% and 47.9% after housing gain vs 18.8% and 23.7% if homelessness persists.
METHODOLOGY
A retrospective cohort study examined all veterans experiencing homelessness who received care at the VHA from 2011 to 2021 and were eligible for but not up to date on CRC and breast cancer screening.
117,619 veterans experiencing homelessness were eligible for but not up to date on CRC screening (aged 50-75 years without prior cancer diagnosis, inflammatory bowel disease, or colectomy) and 6517 veterans experiencing homelessness were eligible for but not up to date on breast cancer screening (women aged 50-75 years without prior cancer diagnosis, lumpectomy, or mastectomy) were included at their index clinic visit.
Exposure was defined as gaining housing within 24 months following index clinic visit, identified through the Homeless Screening Clinical Reminder, US Department of Veterans Affairs (VA) Homeless Operations, Management, and Evaluation System assessments, or US Department of Housing and Urban Development—VA Supportive Housing program move-in dates.
Primary outcome were undergoing screening for CRC (colonoscopy, flexible sigmoidoscopy, computed tomography colonography, barium enema, or stool-based study) or breast cancer (mammogram) that was at a VHA facility or paid by VA within 24 months following index clinic visit.
TAKEAWAY
Among veterans who gained housing, 36.1% underwent CRC screening and 47.9% underwent breast cancer screening during the 24-month observation period, compared with 18.8% and 23.7% of veterans, respectively, among those who remained homeless.
Veterans who gained housing had 2.3 times the adjusted hazard ratio (aHR) of undergoing CRC screening compared with those who remained homeless (AHR, 2.3; 95% CI, 2.2-2.3; P < .001).
Veterans who gained housing had 2.4 times the adjusted hazard of undergoing breast cancer screening compared with those who remained homeless (AHR, 2.4; 95% CI, 2.2-2.7; P < .001).
Median (interquartile range [IQR]) time from index visit to cancer screening was 8 months (4-15) for CRC screening and 8 months (3-14) for breast cancer screening; median (IQR) time from gaining housing to screening was 4 months (1-9) and 3 months (1-8), respectively.
IN PRACTICE: Veterans experiencing homelessness who gain housing have higher rates of cancer screening. “This finding supports promotion of housing to improve health outcomes for homeless individuals," wrote the authors of the study.
SOURCE: The study was led by researchers at the University of California, San Francisco. It was published online in Annals of Family Medicine.
LIMITATIONS: Residual unmeasured confounding was likely due to the observational design of this study, because veterans able to navigate services to obtain housing may also be more likely to complete preventive care. Housing transitions may be misclassified because the Homeless Screening Clinical Reminder was not designed to track changes and may not be administered to veterans already identified as experiencing homelessness. The study did not capture data for screening completed outside VHA or that was not paid for by it. The study cohort only includes veterans with VHA contact, which may limit generalizability.
DISCLOSURES: Benioff Homelessness and Housing Initiative provided grant support for the work; Project Grant K24AG046372 was also awarded to Kushel for the study. Decker is a National Clinician Scholar with salary support from the US Department of Veterans Affairs and reported receiving personal fees from Moon Surgical. Kanzaria and Kushel are faculty members of the Benioff Homelessness and Housing Initiative; Kanzaria also reported advisory work for Amae Health. Kushel is listed as serving on boards including Housing California, National Homelessness Law Center, and Steinberg Institute; other authors reported no conflicts.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: Among Veterans Health Administration (VHA) patients experiencing homelessness, gaining housing is linked to higher 24-month colorectal (CRC) and breast cancer screening completion. In cohorts of 117,619 veterans eligible for colorectal screening and 6517 veterans eligible for breast cancer screening veterans, screening occurs in 36.1% and 47.9% after housing gain vs 18.8% and 23.7% if homelessness persists.
METHODOLOGY
A retrospective cohort study examined all veterans experiencing homelessness who received care at the VHA from 2011 to 2021 and were eligible for but not up to date on CRC and breast cancer screening.
117,619 veterans experiencing homelessness were eligible for but not up to date on CRC screening (aged 50-75 years without prior cancer diagnosis, inflammatory bowel disease, or colectomy) and 6517 veterans experiencing homelessness were eligible for but not up to date on breast cancer screening (women aged 50-75 years without prior cancer diagnosis, lumpectomy, or mastectomy) were included at their index clinic visit.
Exposure was defined as gaining housing within 24 months following index clinic visit, identified through the Homeless Screening Clinical Reminder, US Department of Veterans Affairs (VA) Homeless Operations, Management, and Evaluation System assessments, or US Department of Housing and Urban Development—VA Supportive Housing program move-in dates.
Primary outcome were undergoing screening for CRC (colonoscopy, flexible sigmoidoscopy, computed tomography colonography, barium enema, or stool-based study) or breast cancer (mammogram) that was at a VHA facility or paid by VA within 24 months following index clinic visit.
TAKEAWAY
Among veterans who gained housing, 36.1% underwent CRC screening and 47.9% underwent breast cancer screening during the 24-month observation period, compared with 18.8% and 23.7% of veterans, respectively, among those who remained homeless.
Veterans who gained housing had 2.3 times the adjusted hazard ratio (aHR) of undergoing CRC screening compared with those who remained homeless (AHR, 2.3; 95% CI, 2.2-2.3; P < .001).
Veterans who gained housing had 2.4 times the adjusted hazard of undergoing breast cancer screening compared with those who remained homeless (AHR, 2.4; 95% CI, 2.2-2.7; P < .001).
Median (interquartile range [IQR]) time from index visit to cancer screening was 8 months (4-15) for CRC screening and 8 months (3-14) for breast cancer screening; median (IQR) time from gaining housing to screening was 4 months (1-9) and 3 months (1-8), respectively.
IN PRACTICE: Veterans experiencing homelessness who gain housing have higher rates of cancer screening. “This finding supports promotion of housing to improve health outcomes for homeless individuals," wrote the authors of the study.
SOURCE: The study was led by researchers at the University of California, San Francisco. It was published online in Annals of Family Medicine.
LIMITATIONS: Residual unmeasured confounding was likely due to the observational design of this study, because veterans able to navigate services to obtain housing may also be more likely to complete preventive care. Housing transitions may be misclassified because the Homeless Screening Clinical Reminder was not designed to track changes and may not be administered to veterans already identified as experiencing homelessness. The study did not capture data for screening completed outside VHA or that was not paid for by it. The study cohort only includes veterans with VHA contact, which may limit generalizability.
DISCLOSURES: Benioff Homelessness and Housing Initiative provided grant support for the work; Project Grant K24AG046372 was also awarded to Kushel for the study. Decker is a National Clinician Scholar with salary support from the US Department of Veterans Affairs and reported receiving personal fees from Moon Surgical. Kanzaria and Kushel are faculty members of the Benioff Homelessness and Housing Initiative; Kanzaria also reported advisory work for Amae Health. Kushel is listed as serving on boards including Housing California, National Homelessness Law Center, and Steinberg Institute; other authors reported no conflicts.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
US Cancer Institute Studying Ivermectin's 'Ability to Kill Cancer Cells'
US Cancer Institute Studying Ivermectin's 'Ability to Kill Cancer Cells'
The National Cancer Institute (NCI), the federal research agency charged with leading the war against the nation’s second-largest killer, is studying ivermectin as a potential cancer treatment, according to its top official.
“There are enough reports of it, enough interest in it, that we actually did — ivermectin, in particular — did engage in sort of a better preclinical study of its properties and its ability to kill cancer cells,” said Anthony Letai, a physician the Trump administration appointed as NCI director in September.
Letai did not cite new evidence that might have prompted the institute to research the effectiveness of the antiparasitic drug against cancer. The drug, largely used to treat people or animals for infections caused by parasites, is a popular dewormer for horses.
“We’ll probably have those results in a few months,” Letai said. “So we are taking it seriously.”
He spoke about ivermectin at a January 30 event, “Reclaiming Science: The People’s NIH,” with National Institutes of Health (NIH) Director Jay Bhattacharya and other senior agency officials at Washington, DC’s Willard Hotel. The MAHA Institute hosted the discussion, framed by the “Make America Healthy Again” agenda of Health and Human Services (HSS) Secretary Robert F. Kennedy Jr. The National Cancer Institute is the largest of the NIH’s 27 branches.
During the COVID pandemic, ivermectin’s popularity surged as fringe medical groups promoted it as an effective treatment. Clinical trials have found it isn’t effective against COVID.
Ivermectin has become a symbol of resistance against the medical establishment among MAHA adherents and conservatives. Like-minded commentators and wellness and other online influencers have hyped — without evidence — ivermectin as a miracle cure for a host of diseases, including cancer. Trump officials have pointed to research on ivermectin as an example of the administration’s receptiveness to ideas the scientific establishment has rejected.
“If lots of people believe it and it’s moving public health, we as NIH have an obligation, again, to treat it seriously,” Bhattacharya said at the event. According to The Chronicle at Duke University, Bhattacharya recently said he wants the NIH to be “the research arm of MAHA.”
The decision by the world’s premier cancer research institute to study ivermectin as a cancer treatment has alarmed career scientists at the agency.
“I am shocked and appalled,” one NCI scientist said. “We are moving funds away from so much promising research in order to do a preclinical study based on nonscientific ideas. It’s absurd.”
KFF Health News granted the scientist and other NCI workers anonymity because they are not authorized to speak to the press and fear retaliation.
HHS and the National Cancer Institute did not answer KFF Health News’ questions on the amount of money the cancer institute is spending on the study, who is carrying it out, and whether there was new evidence that prompted NCI to look into ivermectin as an anticancer therapy. Emily Hilliard, an HHS spokesperson, said NIH is dedicated to “rigorous, gold-standard research,” something the administration has repeatedly professed.
A preclinical study is an early phase of research conducted in a lab to test whether a drug or treatment may be useful and to assess potential harms. These studies take place before human clinical trials.
The scientist questioned whether there is enough initial evidence to warrant NCI’s spending of taxpayer funds to investigate the drug’s potential as a cancer treatment.
The FDA has approved ivermectin for certain uses in humans and animals. Tablets are used to treat conditions caused by parasitic worms, and the FDA has approved ivermectin lotions to treat lice and rosacea. Two scientists involved in its discovery won the Nobel Prize in 2015, tied to the drug’s success in treating certain parasitic diseases.
The FDA has warned that large doses of ivermectin can be dangerous. Overdoses can cause seizures, comas, or death.
Kennedy, supporters of the MAHA movement, and some conservative commentators have promoted the idea that the government and pharmaceutical companies quashed ivermectin and other inexpensive, off-patent drugs because they’re not profitable for the drug industry.
“FDA’s war on public health is about to end,” Kennedy wrote in an October 2024 X post that has since gone viral. “This includes its aggressive suppression of psychedelics, peptides, stem cells, raw milk, hyperbaric therapies, chelating compounds, ivermectin, hydroxychloroquine, vitamins, clean foods, sunshine, exercise, nutraceuticals and anything else that advances human health and can’t be patented by Pharma.”
Previous laboratory research has shown that ivermectin could have anticancer effects because it promotes cell death and inhibits the growth of tumor cells. “It actually has been studied both with NIH funds and outside of NIH funds,” Letai said.
However, there is no evidence that ivermectin is safe and effective in treating cancer in humans. Preliminary data from a small clinical trial that gave ivermectin to patients with one type of metastatic breast cancer, in combination with immunotherapy, found no significant benefit from the addition of ivermectin.
Some physicians are concerned that patients will delay or forgo effective cancer treatments, or be harmed in other ways, if they believe unfounded claims that ivermectin can treat their disease.
“Many, many, many things work in a test tube. Quite a few things work in a mouse or a monkey. It still doesn’t mean it’s going to work in people,” said Jeffery Edenfield, executive medical director of oncology for the South Carolina-based Prisma Health Cancer Institute.
Edenfield said cancer patients ask him about ivermectin “regularly,” mostly because of what they see on social media. He said he persuaded a patient to stop using it, and a colleague recently had a patient who decided “to forgo highly effective standard therapy in favor of ivermectin.”
“People come to the discussion having largely already made up their mind,” Edenfield said. “We’re in this delicate time when there’s sort of a fundamental mistrust of medicine,” he added. “Some people are just not going to believe me. I just have to keep trying.”
A June letter by clinicians at Cincinnati Children’s Hospital Medical Center in Ohio detailed how an adolescent patient with metastatic bone cancer started taking ivermectin “after encountering social media posts touting its benefits.” The patient — who hadn’t been given a prescription by a clinician — experienced ivermectin-related neurotoxicity and had to seek emergency care because of nausea, fatigue, and other symptoms.
“We urge the pediatric oncology community to advocate for sensible health policy that prioritizes the well-being of our patients,” the clinicians wrote. The lack of evidence about ivermectin and cancer hasn’t stopped celebrities and online influencers from promoting the notion that the drug is a cure-all. On a January 2025 episode of Joe Rogan’s podcast, actor Mel Gibson claimed that a combination of drugs that included ivermectin cured 3friends with stage IV cancer. The episode has been viewed > 12 million times.
Lawmakers in a handful of states have made the drug available over the counter. And Florida — which, under Republican Governor Ron DeSantis, has become a hotbed for anti-vaccine policies and the spread of public health misinformation — announced last fall that the state plans to fund research to study the drug as a potential cancer treatment.
The Florida Department of Health did not respond to questions about that effort.
Letai, previously a Dana-Farber Cancer Institute oncologist, started at the National Cancer Institute after months of upheaval caused by Trump administration policies.
“What you’re hearing at the NIH now is an openness to ideas — even ideas that scientists would say, ‘Oh, there’s no way it could work’ — but nevertheless applying rigorous scientific methods to those ideas,” Bhattacharya said at the January 30 event.
A second NCI scientist, who was granted anonymity due to fear of retaliation, said the notion that NIH was not open to investigating the value of off-label drugs in cancer is “ridiculous.”
“This is not a new idea they came up with,” the scientist said.
Letai didn’t elaborate on whether NCI scientists are conducting the research or if it has directed funding to an outside institution. Three-fourths of the cancer institute’s research dollars go to outside scientists.
He also aimed to temper expectations.
“At least on a population level,” Letai said, “it’s not going to be a cure-all for cancer.”
The National Cancer Institute (NCI), the federal research agency charged with leading the war against the nation’s second-largest killer, is studying ivermectin as a potential cancer treatment, according to its top official.
“There are enough reports of it, enough interest in it, that we actually did — ivermectin, in particular — did engage in sort of a better preclinical study of its properties and its ability to kill cancer cells,” said Anthony Letai, a physician the Trump administration appointed as NCI director in September.
Letai did not cite new evidence that might have prompted the institute to research the effectiveness of the antiparasitic drug against cancer. The drug, largely used to treat people or animals for infections caused by parasites, is a popular dewormer for horses.
“We’ll probably have those results in a few months,” Letai said. “So we are taking it seriously.”
He spoke about ivermectin at a January 30 event, “Reclaiming Science: The People’s NIH,” with National Institutes of Health (NIH) Director Jay Bhattacharya and other senior agency officials at Washington, DC’s Willard Hotel. The MAHA Institute hosted the discussion, framed by the “Make America Healthy Again” agenda of Health and Human Services (HSS) Secretary Robert F. Kennedy Jr. The National Cancer Institute is the largest of the NIH’s 27 branches.
During the COVID pandemic, ivermectin’s popularity surged as fringe medical groups promoted it as an effective treatment. Clinical trials have found it isn’t effective against COVID.
Ivermectin has become a symbol of resistance against the medical establishment among MAHA adherents and conservatives. Like-minded commentators and wellness and other online influencers have hyped — without evidence — ivermectin as a miracle cure for a host of diseases, including cancer. Trump officials have pointed to research on ivermectin as an example of the administration’s receptiveness to ideas the scientific establishment has rejected.
“If lots of people believe it and it’s moving public health, we as NIH have an obligation, again, to treat it seriously,” Bhattacharya said at the event. According to The Chronicle at Duke University, Bhattacharya recently said he wants the NIH to be “the research arm of MAHA.”
The decision by the world’s premier cancer research institute to study ivermectin as a cancer treatment has alarmed career scientists at the agency.
“I am shocked and appalled,” one NCI scientist said. “We are moving funds away from so much promising research in order to do a preclinical study based on nonscientific ideas. It’s absurd.”
KFF Health News granted the scientist and other NCI workers anonymity because they are not authorized to speak to the press and fear retaliation.
HHS and the National Cancer Institute did not answer KFF Health News’ questions on the amount of money the cancer institute is spending on the study, who is carrying it out, and whether there was new evidence that prompted NCI to look into ivermectin as an anticancer therapy. Emily Hilliard, an HHS spokesperson, said NIH is dedicated to “rigorous, gold-standard research,” something the administration has repeatedly professed.
A preclinical study is an early phase of research conducted in a lab to test whether a drug or treatment may be useful and to assess potential harms. These studies take place before human clinical trials.
The scientist questioned whether there is enough initial evidence to warrant NCI’s spending of taxpayer funds to investigate the drug’s potential as a cancer treatment.
The FDA has approved ivermectin for certain uses in humans and animals. Tablets are used to treat conditions caused by parasitic worms, and the FDA has approved ivermectin lotions to treat lice and rosacea. Two scientists involved in its discovery won the Nobel Prize in 2015, tied to the drug’s success in treating certain parasitic diseases.
The FDA has warned that large doses of ivermectin can be dangerous. Overdoses can cause seizures, comas, or death.
Kennedy, supporters of the MAHA movement, and some conservative commentators have promoted the idea that the government and pharmaceutical companies quashed ivermectin and other inexpensive, off-patent drugs because they’re not profitable for the drug industry.
“FDA’s war on public health is about to end,” Kennedy wrote in an October 2024 X post that has since gone viral. “This includes its aggressive suppression of psychedelics, peptides, stem cells, raw milk, hyperbaric therapies, chelating compounds, ivermectin, hydroxychloroquine, vitamins, clean foods, sunshine, exercise, nutraceuticals and anything else that advances human health and can’t be patented by Pharma.”
Previous laboratory research has shown that ivermectin could have anticancer effects because it promotes cell death and inhibits the growth of tumor cells. “It actually has been studied both with NIH funds and outside of NIH funds,” Letai said.
However, there is no evidence that ivermectin is safe and effective in treating cancer in humans. Preliminary data from a small clinical trial that gave ivermectin to patients with one type of metastatic breast cancer, in combination with immunotherapy, found no significant benefit from the addition of ivermectin.
Some physicians are concerned that patients will delay or forgo effective cancer treatments, or be harmed in other ways, if they believe unfounded claims that ivermectin can treat their disease.
“Many, many, many things work in a test tube. Quite a few things work in a mouse or a monkey. It still doesn’t mean it’s going to work in people,” said Jeffery Edenfield, executive medical director of oncology for the South Carolina-based Prisma Health Cancer Institute.
Edenfield said cancer patients ask him about ivermectin “regularly,” mostly because of what they see on social media. He said he persuaded a patient to stop using it, and a colleague recently had a patient who decided “to forgo highly effective standard therapy in favor of ivermectin.”
“People come to the discussion having largely already made up their mind,” Edenfield said. “We’re in this delicate time when there’s sort of a fundamental mistrust of medicine,” he added. “Some people are just not going to believe me. I just have to keep trying.”
A June letter by clinicians at Cincinnati Children’s Hospital Medical Center in Ohio detailed how an adolescent patient with metastatic bone cancer started taking ivermectin “after encountering social media posts touting its benefits.” The patient — who hadn’t been given a prescription by a clinician — experienced ivermectin-related neurotoxicity and had to seek emergency care because of nausea, fatigue, and other symptoms.
“We urge the pediatric oncology community to advocate for sensible health policy that prioritizes the well-being of our patients,” the clinicians wrote. The lack of evidence about ivermectin and cancer hasn’t stopped celebrities and online influencers from promoting the notion that the drug is a cure-all. On a January 2025 episode of Joe Rogan’s podcast, actor Mel Gibson claimed that a combination of drugs that included ivermectin cured 3friends with stage IV cancer. The episode has been viewed > 12 million times.
Lawmakers in a handful of states have made the drug available over the counter. And Florida — which, under Republican Governor Ron DeSantis, has become a hotbed for anti-vaccine policies and the spread of public health misinformation — announced last fall that the state plans to fund research to study the drug as a potential cancer treatment.
The Florida Department of Health did not respond to questions about that effort.
Letai, previously a Dana-Farber Cancer Institute oncologist, started at the National Cancer Institute after months of upheaval caused by Trump administration policies.
“What you’re hearing at the NIH now is an openness to ideas — even ideas that scientists would say, ‘Oh, there’s no way it could work’ — but nevertheless applying rigorous scientific methods to those ideas,” Bhattacharya said at the January 30 event.
A second NCI scientist, who was granted anonymity due to fear of retaliation, said the notion that NIH was not open to investigating the value of off-label drugs in cancer is “ridiculous.”
“This is not a new idea they came up with,” the scientist said.
Letai didn’t elaborate on whether NCI scientists are conducting the research or if it has directed funding to an outside institution. Three-fourths of the cancer institute’s research dollars go to outside scientists.
He also aimed to temper expectations.
“At least on a population level,” Letai said, “it’s not going to be a cure-all for cancer.”
The National Cancer Institute (NCI), the federal research agency charged with leading the war against the nation’s second-largest killer, is studying ivermectin as a potential cancer treatment, according to its top official.
“There are enough reports of it, enough interest in it, that we actually did — ivermectin, in particular — did engage in sort of a better preclinical study of its properties and its ability to kill cancer cells,” said Anthony Letai, a physician the Trump administration appointed as NCI director in September.
Letai did not cite new evidence that might have prompted the institute to research the effectiveness of the antiparasitic drug against cancer. The drug, largely used to treat people or animals for infections caused by parasites, is a popular dewormer for horses.
“We’ll probably have those results in a few months,” Letai said. “So we are taking it seriously.”
He spoke about ivermectin at a January 30 event, “Reclaiming Science: The People’s NIH,” with National Institutes of Health (NIH) Director Jay Bhattacharya and other senior agency officials at Washington, DC’s Willard Hotel. The MAHA Institute hosted the discussion, framed by the “Make America Healthy Again” agenda of Health and Human Services (HSS) Secretary Robert F. Kennedy Jr. The National Cancer Institute is the largest of the NIH’s 27 branches.
During the COVID pandemic, ivermectin’s popularity surged as fringe medical groups promoted it as an effective treatment. Clinical trials have found it isn’t effective against COVID.
Ivermectin has become a symbol of resistance against the medical establishment among MAHA adherents and conservatives. Like-minded commentators and wellness and other online influencers have hyped — without evidence — ivermectin as a miracle cure for a host of diseases, including cancer. Trump officials have pointed to research on ivermectin as an example of the administration’s receptiveness to ideas the scientific establishment has rejected.
“If lots of people believe it and it’s moving public health, we as NIH have an obligation, again, to treat it seriously,” Bhattacharya said at the event. According to The Chronicle at Duke University, Bhattacharya recently said he wants the NIH to be “the research arm of MAHA.”
The decision by the world’s premier cancer research institute to study ivermectin as a cancer treatment has alarmed career scientists at the agency.
“I am shocked and appalled,” one NCI scientist said. “We are moving funds away from so much promising research in order to do a preclinical study based on nonscientific ideas. It’s absurd.”
KFF Health News granted the scientist and other NCI workers anonymity because they are not authorized to speak to the press and fear retaliation.
HHS and the National Cancer Institute did not answer KFF Health News’ questions on the amount of money the cancer institute is spending on the study, who is carrying it out, and whether there was new evidence that prompted NCI to look into ivermectin as an anticancer therapy. Emily Hilliard, an HHS spokesperson, said NIH is dedicated to “rigorous, gold-standard research,” something the administration has repeatedly professed.
A preclinical study is an early phase of research conducted in a lab to test whether a drug or treatment may be useful and to assess potential harms. These studies take place before human clinical trials.
The scientist questioned whether there is enough initial evidence to warrant NCI’s spending of taxpayer funds to investigate the drug’s potential as a cancer treatment.
The FDA has approved ivermectin for certain uses in humans and animals. Tablets are used to treat conditions caused by parasitic worms, and the FDA has approved ivermectin lotions to treat lice and rosacea. Two scientists involved in its discovery won the Nobel Prize in 2015, tied to the drug’s success in treating certain parasitic diseases.
The FDA has warned that large doses of ivermectin can be dangerous. Overdoses can cause seizures, comas, or death.
Kennedy, supporters of the MAHA movement, and some conservative commentators have promoted the idea that the government and pharmaceutical companies quashed ivermectin and other inexpensive, off-patent drugs because they’re not profitable for the drug industry.
“FDA’s war on public health is about to end,” Kennedy wrote in an October 2024 X post that has since gone viral. “This includes its aggressive suppression of psychedelics, peptides, stem cells, raw milk, hyperbaric therapies, chelating compounds, ivermectin, hydroxychloroquine, vitamins, clean foods, sunshine, exercise, nutraceuticals and anything else that advances human health and can’t be patented by Pharma.”
Previous laboratory research has shown that ivermectin could have anticancer effects because it promotes cell death and inhibits the growth of tumor cells. “It actually has been studied both with NIH funds and outside of NIH funds,” Letai said.
However, there is no evidence that ivermectin is safe and effective in treating cancer in humans. Preliminary data from a small clinical trial that gave ivermectin to patients with one type of metastatic breast cancer, in combination with immunotherapy, found no significant benefit from the addition of ivermectin.
Some physicians are concerned that patients will delay or forgo effective cancer treatments, or be harmed in other ways, if they believe unfounded claims that ivermectin can treat their disease.
“Many, many, many things work in a test tube. Quite a few things work in a mouse or a monkey. It still doesn’t mean it’s going to work in people,” said Jeffery Edenfield, executive medical director of oncology for the South Carolina-based Prisma Health Cancer Institute.
Edenfield said cancer patients ask him about ivermectin “regularly,” mostly because of what they see on social media. He said he persuaded a patient to stop using it, and a colleague recently had a patient who decided “to forgo highly effective standard therapy in favor of ivermectin.”
“People come to the discussion having largely already made up their mind,” Edenfield said. “We’re in this delicate time when there’s sort of a fundamental mistrust of medicine,” he added. “Some people are just not going to believe me. I just have to keep trying.”
A June letter by clinicians at Cincinnati Children’s Hospital Medical Center in Ohio detailed how an adolescent patient with metastatic bone cancer started taking ivermectin “after encountering social media posts touting its benefits.” The patient — who hadn’t been given a prescription by a clinician — experienced ivermectin-related neurotoxicity and had to seek emergency care because of nausea, fatigue, and other symptoms.
“We urge the pediatric oncology community to advocate for sensible health policy that prioritizes the well-being of our patients,” the clinicians wrote. The lack of evidence about ivermectin and cancer hasn’t stopped celebrities and online influencers from promoting the notion that the drug is a cure-all. On a January 2025 episode of Joe Rogan’s podcast, actor Mel Gibson claimed that a combination of drugs that included ivermectin cured 3friends with stage IV cancer. The episode has been viewed > 12 million times.
Lawmakers in a handful of states have made the drug available over the counter. And Florida — which, under Republican Governor Ron DeSantis, has become a hotbed for anti-vaccine policies and the spread of public health misinformation — announced last fall that the state plans to fund research to study the drug as a potential cancer treatment.
The Florida Department of Health did not respond to questions about that effort.
Letai, previously a Dana-Farber Cancer Institute oncologist, started at the National Cancer Institute after months of upheaval caused by Trump administration policies.
“What you’re hearing at the NIH now is an openness to ideas — even ideas that scientists would say, ‘Oh, there’s no way it could work’ — but nevertheless applying rigorous scientific methods to those ideas,” Bhattacharya said at the January 30 event.
A second NCI scientist, who was granted anonymity due to fear of retaliation, said the notion that NIH was not open to investigating the value of off-label drugs in cancer is “ridiculous.”
“This is not a new idea they came up with,” the scientist said.
Letai didn’t elaborate on whether NCI scientists are conducting the research or if it has directed funding to an outside institution. Three-fourths of the cancer institute’s research dollars go to outside scientists.
He also aimed to temper expectations.
“At least on a population level,” Letai said, “it’s not going to be a cure-all for cancer.”
US Cancer Institute Studying Ivermectin's 'Ability to Kill Cancer Cells'
US Cancer Institute Studying Ivermectin's 'Ability to Kill Cancer Cells'