User login
Weight Loss After Anti-Obesity Medications Linked to Reduced Gout Risk
TOPLINE:
A higher rate of weight loss within 1 year of initiating orlistat is associated with lower risks for incident gout and recurrent gout flares in individuals with body mass index (BMI) > 25, particularly if they have obesity or high baseline serum urate levels.
METHODOLOGY:
- Researchers conducted a population-based cohort study using data from The Health Improvement Network in the United Kingdom to examine the association between weight loss rates after the initiation of anti-obesity medication (orlistat) and the risk for incident gout and recurrent gout flares in patients with overweight or obesity.
- The risk for incident gout was analyzed in 131,000 patients with overweight or obesity (mean age, 45 years; 77.3% women; mean BMI, 37.2) who did not have gout before initiating orlistat.
- The risk for recurrent gout flares was evaluated in 3847 individuals with overweight or obesity (mean age, 56.6 years; 29.4% women; mean BMI, 38.5), who had gout before initiating orlistat.
- Participants were divided into four groups based on their rate of weight loss during the first year of orlistat use: Weight gain or stable (< 2%), slow (2% to < 5%), moderate (5% to < 10%), and fast (≥ 10%).
- The primary outcome was incident gout, and the secondary outcome was the rate of recurrent gout flares during the 5-year follow-up period after initiating orlistat.
TAKEAWAY:
- The 5-year risk for incident gout was the lowest among patients in the fast weight loss group (1.2%) and highest among those in the weight gain or stable weight group (1.6%).
- The risk for incident gout was lower in the fast (hazard ratio [HR], 0.73; 95% CI, 0.62-0.86) and moderate (HR, 0.82; 95% CI, 0.72-0.92) weight loss groups than in the weight gain or stable weight group.
- Similarly, faster weight loss rates were linked to lower rates of recurrent gout flares, with risk ratios of 0.71 (95% CI, 0.60-0.84) and 0.83 (95% CI, 0.71-0.96) in the fast and moderate weight loss groups, respectively.
- This study found that .
IN PRACTICE:
“Pharmacologic treatments, such as orlistat, present an alternative strategy for managing overweight and obesity. Our study provides empirical evidence of a dose-response effect of weight loss after initiating orlistat within 1 year lowers the risk of incident gout and recurrent gout flares,” the authors wrote.
SOURCE:
This study was led by Jie Wei, PhD, Department of Orthopaedics, Xiangya Hospital, Central South University, Changsha, China, and was published online on September 19, 2024, in Arthritis & Rheumatology.
LIMITATIONS:
Despite adjustment for many variables, factors such as disease severity, exercise levels, and diet were not fully captured, which might have influenced the results. The lack of hospitalization data could have resulted in recurrent gout flares being underreported. The current study may have been subjected to bias due to potential exposure misclassification resulting from the timing of weight measurements and missing updated weight data.
DISCLOSURES:
This study was supported by the National Key Research and Development Plan, the National Natural Science Foundation of China, the Project Program of National Clinical Research Center for Geriatric Disorders, and other sources. No disclosures of interest were reported by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
A higher rate of weight loss within 1 year of initiating orlistat is associated with lower risks for incident gout and recurrent gout flares in individuals with body mass index (BMI) > 25, particularly if they have obesity or high baseline serum urate levels.
METHODOLOGY:
- Researchers conducted a population-based cohort study using data from The Health Improvement Network in the United Kingdom to examine the association between weight loss rates after the initiation of anti-obesity medication (orlistat) and the risk for incident gout and recurrent gout flares in patients with overweight or obesity.
- The risk for incident gout was analyzed in 131,000 patients with overweight or obesity (mean age, 45 years; 77.3% women; mean BMI, 37.2) who did not have gout before initiating orlistat.
- The risk for recurrent gout flares was evaluated in 3847 individuals with overweight or obesity (mean age, 56.6 years; 29.4% women; mean BMI, 38.5), who had gout before initiating orlistat.
- Participants were divided into four groups based on their rate of weight loss during the first year of orlistat use: Weight gain or stable (< 2%), slow (2% to < 5%), moderate (5% to < 10%), and fast (≥ 10%).
- The primary outcome was incident gout, and the secondary outcome was the rate of recurrent gout flares during the 5-year follow-up period after initiating orlistat.
TAKEAWAY:
- The 5-year risk for incident gout was the lowest among patients in the fast weight loss group (1.2%) and highest among those in the weight gain or stable weight group (1.6%).
- The risk for incident gout was lower in the fast (hazard ratio [HR], 0.73; 95% CI, 0.62-0.86) and moderate (HR, 0.82; 95% CI, 0.72-0.92) weight loss groups than in the weight gain or stable weight group.
- Similarly, faster weight loss rates were linked to lower rates of recurrent gout flares, with risk ratios of 0.71 (95% CI, 0.60-0.84) and 0.83 (95% CI, 0.71-0.96) in the fast and moderate weight loss groups, respectively.
- This study found that .
IN PRACTICE:
“Pharmacologic treatments, such as orlistat, present an alternative strategy for managing overweight and obesity. Our study provides empirical evidence of a dose-response effect of weight loss after initiating orlistat within 1 year lowers the risk of incident gout and recurrent gout flares,” the authors wrote.
SOURCE:
This study was led by Jie Wei, PhD, Department of Orthopaedics, Xiangya Hospital, Central South University, Changsha, China, and was published online on September 19, 2024, in Arthritis & Rheumatology.
LIMITATIONS:
Despite adjustment for many variables, factors such as disease severity, exercise levels, and diet were not fully captured, which might have influenced the results. The lack of hospitalization data could have resulted in recurrent gout flares being underreported. The current study may have been subjected to bias due to potential exposure misclassification resulting from the timing of weight measurements and missing updated weight data.
DISCLOSURES:
This study was supported by the National Key Research and Development Plan, the National Natural Science Foundation of China, the Project Program of National Clinical Research Center for Geriatric Disorders, and other sources. No disclosures of interest were reported by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
A higher rate of weight loss within 1 year of initiating orlistat is associated with lower risks for incident gout and recurrent gout flares in individuals with body mass index (BMI) > 25, particularly if they have obesity or high baseline serum urate levels.
METHODOLOGY:
- Researchers conducted a population-based cohort study using data from The Health Improvement Network in the United Kingdom to examine the association between weight loss rates after the initiation of anti-obesity medication (orlistat) and the risk for incident gout and recurrent gout flares in patients with overweight or obesity.
- The risk for incident gout was analyzed in 131,000 patients with overweight or obesity (mean age, 45 years; 77.3% women; mean BMI, 37.2) who did not have gout before initiating orlistat.
- The risk for recurrent gout flares was evaluated in 3847 individuals with overweight or obesity (mean age, 56.6 years; 29.4% women; mean BMI, 38.5), who had gout before initiating orlistat.
- Participants were divided into four groups based on their rate of weight loss during the first year of orlistat use: Weight gain or stable (< 2%), slow (2% to < 5%), moderate (5% to < 10%), and fast (≥ 10%).
- The primary outcome was incident gout, and the secondary outcome was the rate of recurrent gout flares during the 5-year follow-up period after initiating orlistat.
TAKEAWAY:
- The 5-year risk for incident gout was the lowest among patients in the fast weight loss group (1.2%) and highest among those in the weight gain or stable weight group (1.6%).
- The risk for incident gout was lower in the fast (hazard ratio [HR], 0.73; 95% CI, 0.62-0.86) and moderate (HR, 0.82; 95% CI, 0.72-0.92) weight loss groups than in the weight gain or stable weight group.
- Similarly, faster weight loss rates were linked to lower rates of recurrent gout flares, with risk ratios of 0.71 (95% CI, 0.60-0.84) and 0.83 (95% CI, 0.71-0.96) in the fast and moderate weight loss groups, respectively.
- This study found that .
IN PRACTICE:
“Pharmacologic treatments, such as orlistat, present an alternative strategy for managing overweight and obesity. Our study provides empirical evidence of a dose-response effect of weight loss after initiating orlistat within 1 year lowers the risk of incident gout and recurrent gout flares,” the authors wrote.
SOURCE:
This study was led by Jie Wei, PhD, Department of Orthopaedics, Xiangya Hospital, Central South University, Changsha, China, and was published online on September 19, 2024, in Arthritis & Rheumatology.
LIMITATIONS:
Despite adjustment for many variables, factors such as disease severity, exercise levels, and diet were not fully captured, which might have influenced the results. The lack of hospitalization data could have resulted in recurrent gout flares being underreported. The current study may have been subjected to bias due to potential exposure misclassification resulting from the timing of weight measurements and missing updated weight data.
DISCLOSURES:
This study was supported by the National Key Research and Development Plan, the National Natural Science Foundation of China, the Project Program of National Clinical Research Center for Geriatric Disorders, and other sources. No disclosures of interest were reported by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Time-Restricted Eating Is Not a Metabolic Magic Bullet
This transcript has been edited for clarity.
One out of three American adults — about 100 million people in this country — have the metabolic syndrome. I’m showing you the official criteria here, but essentially this is a syndrome of insulin resistance and visceral adiposity that predisposes us to a host of chronic diseases such as diabetes, heart disease, and even dementia.
The metabolic syndrome is, fundamentally, a lifestyle disease. There is a direct line between our dietary habits and the wide availability of carbohydrate-rich, highly processed foods, and the rise in the syndrome in the population.
A saying I learned from one of my epidemiology teachers comes to mind: “Lifestyle diseases require lifestyle reinterventions.” But you know what? I’m not so sure anymore.
I’ve been around long enough to see multiple dietary fads come and go with varying efficacy. I grew up in the low-fat era, probably the most detrimental time to our national health as food manufacturers started replacing fats with carbohydrates, driving much of the problem we’re faced with today.
But I was also around for the Atkins diet and the low-carb craze — a healthier approach, all things being equal. And I’ve seen variants of these: the paleo diet (essentially a low-carb, high-protein diet based on minimally processed foods) and the Mediterranean diet, which sought to replace some percentage of fats with healthier fats.
And, of course, there is time-restricted eating.
Time-restricted eating, a variant of intermittent fasting, has the advantage of being very simple. No cookbooks, no recipes. Eat what you want — but limit it to certain hours in the day, ideally a window of less than 10 hours, such as 8 a.m. to 6 p.m.
When it comes to weight loss, the diets that work tend to work because they reduce calorie intake. I know, people will get angry about this, but thermodynamics is not just a good idea, it’s the law.
But weight loss is not the only reason we need to eat healthier. What we eat can impact our health in multiple ways; certain foods lead to more atherosclerosis, more inflammation, increased strain on the kidney and liver, and can affect our glucose homeostasis.
So I was really interested when I saw this article, “Time-Restricted Eating in Adults With Metabolic Syndrome,” appearing in Annals of Internal Medicine October 1, which examined the effect of time-restricted eating on the metabolic syndrome itself. Could this lifestyle intervention cure this lifestyle disease?
In the study, 108 individuals, all of whom had the metabolic syndrome but not full-blown diabetes, were randomized to usual care — basically, nutrition education — vs time-restricted eating. In that group, participants were instructed to reduce their window of eating by at least 4 hours to achieve an 8- to 10-hour eating window. The groups were followed for 3 months.
Now, before we get to the results, it’s important to remember that the success of a lifestyle intervention trial is quite dependent on how well people adhere to the lifestyle intervention. Time-restricted eating is not as easy as taking a pill once a day.
The researchers had participants log their consumption using a smartphone app to confirm whether they were adhering to that restricted eating window.
Broadly speaking, they did. At baseline, both groups had an eating window of about 14 hours a day — think 7 a.m. to 9 p.m. The intervention group reduced that to just under 10 hours, with 10% of days falling outside of the target window.
Lifestyle change achieved, the primary outcome was the change in hemoglobin A1c at 3 months. A1c integrates the serum glucose over time and is thus a good indicator of the success of the intervention in terms of insulin resistance. But the effect was, honestly, disappointing.
Technically, the time-restricted-eating group had a greater A1c change than the control group — by 0.1 percentage points. On average, they went from a baseline A1c of 5.87 to a 3-month A1c of 5.75.
Other metabolic syndrome markers were equally lackluster: no difference in fasting glucose, mean glucose, or fasting insulin.
There was some weight change. The control group, which got that dietary education, lost 1.5% of body weight over the 3 months. The time-restricted-eating group lost 3.3% — about 7 pounds, which is reasonable.
With that weight loss came statistically significant, albeit modest improvements in BMI, body fat percentage, and LDL cholesterol.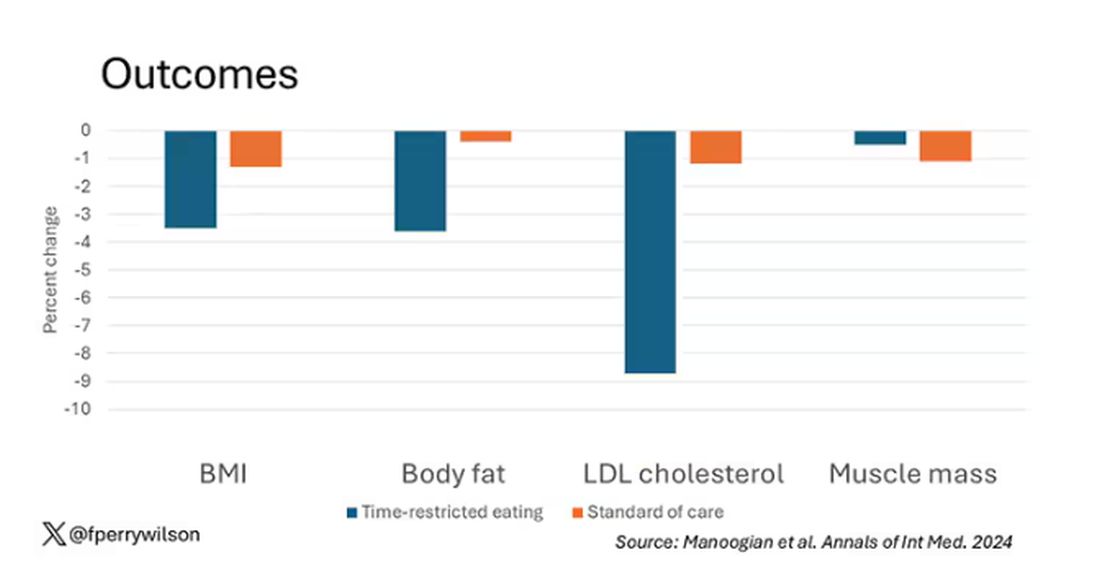
Of interest, despite the larger weight loss in the intermittent-fasting group, there was no difference in muscle mass loss, which is encouraging.
Taken together, we can say that, yes, it seems like time-restricted eating can help people lose some weight. This is essentially due to the fact that people eat fewer calories when they do time-restricted eating, as you can see here.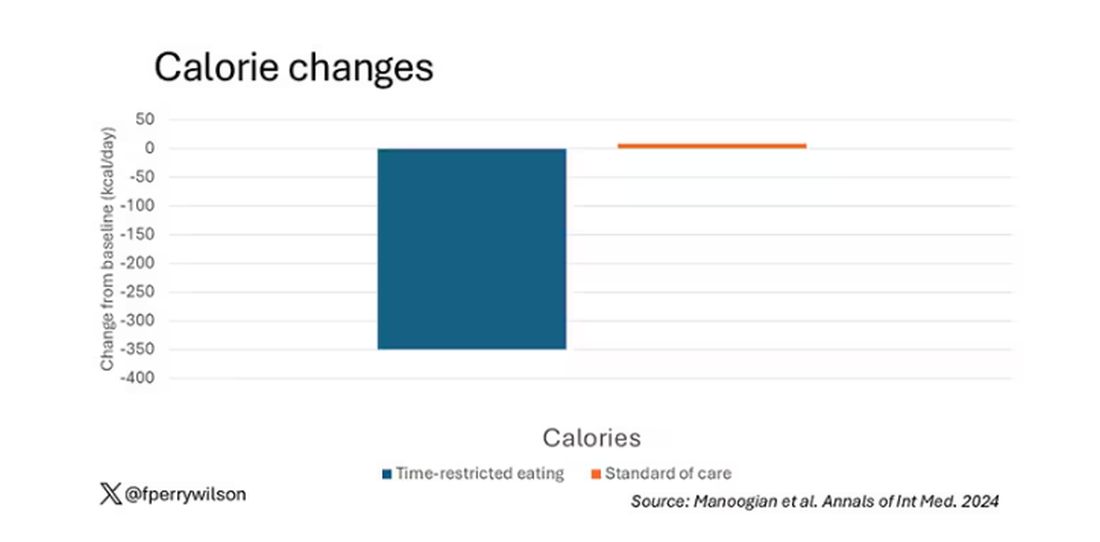
But, in the end, this trial examined whether this relatively straightforward lifestyle intervention would move the needle in terms of metabolic syndrome, and the data are not very compelling for that.
This graph shows how many of those five factors for metabolic syndrome the individuals in this trial had from the start to the end. You see that, over the 3 months, seven people in the time-restricted-eating group moved from having three criteria to two or one — being “cured” of metabolic syndrome, if you will. Nine people in the standard group were cured by that definition. Remember, they had to have at least three to have the syndrome and thus be eligible for the trial. 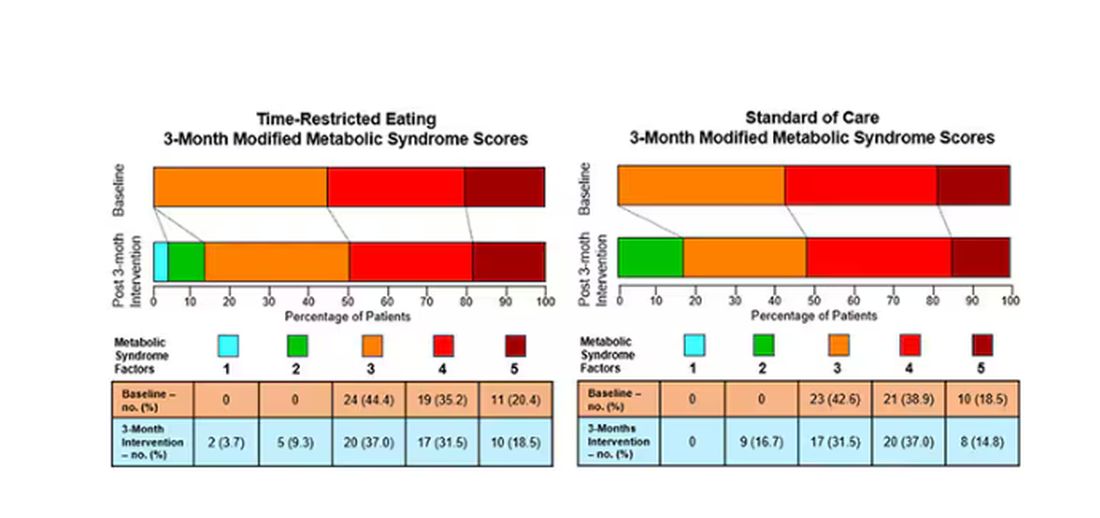
So If it just leads to weight loss by forcing people to consume less calories, then we need to acknowledge that we probably have better methods to achieve this same end. Ten years ago, I would have said that lifestyle change is the only way to end the epidemic of the metabolic syndrome in this country. Today, well, we live in a world of GLP-1 weight loss drugs. It is simply a different world now. Yes, they are expensive. Yes, they have side effects. But we need to evaluate them against the comparison. And so far, lifestyle changes alone are really no comparison.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
One out of three American adults — about 100 million people in this country — have the metabolic syndrome. I’m showing you the official criteria here, but essentially this is a syndrome of insulin resistance and visceral adiposity that predisposes us to a host of chronic diseases such as diabetes, heart disease, and even dementia.
The metabolic syndrome is, fundamentally, a lifestyle disease. There is a direct line between our dietary habits and the wide availability of carbohydrate-rich, highly processed foods, and the rise in the syndrome in the population.
A saying I learned from one of my epidemiology teachers comes to mind: “Lifestyle diseases require lifestyle reinterventions.” But you know what? I’m not so sure anymore.
I’ve been around long enough to see multiple dietary fads come and go with varying efficacy. I grew up in the low-fat era, probably the most detrimental time to our national health as food manufacturers started replacing fats with carbohydrates, driving much of the problem we’re faced with today.
But I was also around for the Atkins diet and the low-carb craze — a healthier approach, all things being equal. And I’ve seen variants of these: the paleo diet (essentially a low-carb, high-protein diet based on minimally processed foods) and the Mediterranean diet, which sought to replace some percentage of fats with healthier fats.
And, of course, there is time-restricted eating.
Time-restricted eating, a variant of intermittent fasting, has the advantage of being very simple. No cookbooks, no recipes. Eat what you want — but limit it to certain hours in the day, ideally a window of less than 10 hours, such as 8 a.m. to 6 p.m.
When it comes to weight loss, the diets that work tend to work because they reduce calorie intake. I know, people will get angry about this, but thermodynamics is not just a good idea, it’s the law.
But weight loss is not the only reason we need to eat healthier. What we eat can impact our health in multiple ways; certain foods lead to more atherosclerosis, more inflammation, increased strain on the kidney and liver, and can affect our glucose homeostasis.
So I was really interested when I saw this article, “Time-Restricted Eating in Adults With Metabolic Syndrome,” appearing in Annals of Internal Medicine October 1, which examined the effect of time-restricted eating on the metabolic syndrome itself. Could this lifestyle intervention cure this lifestyle disease?
In the study, 108 individuals, all of whom had the metabolic syndrome but not full-blown diabetes, were randomized to usual care — basically, nutrition education — vs time-restricted eating. In that group, participants were instructed to reduce their window of eating by at least 4 hours to achieve an 8- to 10-hour eating window. The groups were followed for 3 months.
Now, before we get to the results, it’s important to remember that the success of a lifestyle intervention trial is quite dependent on how well people adhere to the lifestyle intervention. Time-restricted eating is not as easy as taking a pill once a day.
The researchers had participants log their consumption using a smartphone app to confirm whether they were adhering to that restricted eating window.
Broadly speaking, they did. At baseline, both groups had an eating window of about 14 hours a day — think 7 a.m. to 9 p.m. The intervention group reduced that to just under 10 hours, with 10% of days falling outside of the target window.
Lifestyle change achieved, the primary outcome was the change in hemoglobin A1c at 3 months. A1c integrates the serum glucose over time and is thus a good indicator of the success of the intervention in terms of insulin resistance. But the effect was, honestly, disappointing.
Technically, the time-restricted-eating group had a greater A1c change than the control group — by 0.1 percentage points. On average, they went from a baseline A1c of 5.87 to a 3-month A1c of 5.75.
Other metabolic syndrome markers were equally lackluster: no difference in fasting glucose, mean glucose, or fasting insulin.
There was some weight change. The control group, which got that dietary education, lost 1.5% of body weight over the 3 months. The time-restricted-eating group lost 3.3% — about 7 pounds, which is reasonable.
With that weight loss came statistically significant, albeit modest improvements in BMI, body fat percentage, and LDL cholesterol.
Of interest, despite the larger weight loss in the intermittent-fasting group, there was no difference in muscle mass loss, which is encouraging.
Taken together, we can say that, yes, it seems like time-restricted eating can help people lose some weight. This is essentially due to the fact that people eat fewer calories when they do time-restricted eating, as you can see here.
But, in the end, this trial examined whether this relatively straightforward lifestyle intervention would move the needle in terms of metabolic syndrome, and the data are not very compelling for that.
This graph shows how many of those five factors for metabolic syndrome the individuals in this trial had from the start to the end. You see that, over the 3 months, seven people in the time-restricted-eating group moved from having three criteria to two or one — being “cured” of metabolic syndrome, if you will. Nine people in the standard group were cured by that definition. Remember, they had to have at least three to have the syndrome and thus be eligible for the trial. 
So If it just leads to weight loss by forcing people to consume less calories, then we need to acknowledge that we probably have better methods to achieve this same end. Ten years ago, I would have said that lifestyle change is the only way to end the epidemic of the metabolic syndrome in this country. Today, well, we live in a world of GLP-1 weight loss drugs. It is simply a different world now. Yes, they are expensive. Yes, they have side effects. But we need to evaluate them against the comparison. And so far, lifestyle changes alone are really no comparison.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
One out of three American adults — about 100 million people in this country — have the metabolic syndrome. I’m showing you the official criteria here, but essentially this is a syndrome of insulin resistance and visceral adiposity that predisposes us to a host of chronic diseases such as diabetes, heart disease, and even dementia.
The metabolic syndrome is, fundamentally, a lifestyle disease. There is a direct line between our dietary habits and the wide availability of carbohydrate-rich, highly processed foods, and the rise in the syndrome in the population.
A saying I learned from one of my epidemiology teachers comes to mind: “Lifestyle diseases require lifestyle reinterventions.” But you know what? I’m not so sure anymore.
I’ve been around long enough to see multiple dietary fads come and go with varying efficacy. I grew up in the low-fat era, probably the most detrimental time to our national health as food manufacturers started replacing fats with carbohydrates, driving much of the problem we’re faced with today.
But I was also around for the Atkins diet and the low-carb craze — a healthier approach, all things being equal. And I’ve seen variants of these: the paleo diet (essentially a low-carb, high-protein diet based on minimally processed foods) and the Mediterranean diet, which sought to replace some percentage of fats with healthier fats.
And, of course, there is time-restricted eating.
Time-restricted eating, a variant of intermittent fasting, has the advantage of being very simple. No cookbooks, no recipes. Eat what you want — but limit it to certain hours in the day, ideally a window of less than 10 hours, such as 8 a.m. to 6 p.m.
When it comes to weight loss, the diets that work tend to work because they reduce calorie intake. I know, people will get angry about this, but thermodynamics is not just a good idea, it’s the law.
But weight loss is not the only reason we need to eat healthier. What we eat can impact our health in multiple ways; certain foods lead to more atherosclerosis, more inflammation, increased strain on the kidney and liver, and can affect our glucose homeostasis.
So I was really interested when I saw this article, “Time-Restricted Eating in Adults With Metabolic Syndrome,” appearing in Annals of Internal Medicine October 1, which examined the effect of time-restricted eating on the metabolic syndrome itself. Could this lifestyle intervention cure this lifestyle disease?
In the study, 108 individuals, all of whom had the metabolic syndrome but not full-blown diabetes, were randomized to usual care — basically, nutrition education — vs time-restricted eating. In that group, participants were instructed to reduce their window of eating by at least 4 hours to achieve an 8- to 10-hour eating window. The groups were followed for 3 months.
Now, before we get to the results, it’s important to remember that the success of a lifestyle intervention trial is quite dependent on how well people adhere to the lifestyle intervention. Time-restricted eating is not as easy as taking a pill once a day.
The researchers had participants log their consumption using a smartphone app to confirm whether they were adhering to that restricted eating window.
Broadly speaking, they did. At baseline, both groups had an eating window of about 14 hours a day — think 7 a.m. to 9 p.m. The intervention group reduced that to just under 10 hours, with 10% of days falling outside of the target window.
Lifestyle change achieved, the primary outcome was the change in hemoglobin A1c at 3 months. A1c integrates the serum glucose over time and is thus a good indicator of the success of the intervention in terms of insulin resistance. But the effect was, honestly, disappointing.
Technically, the time-restricted-eating group had a greater A1c change than the control group — by 0.1 percentage points. On average, they went from a baseline A1c of 5.87 to a 3-month A1c of 5.75.
Other metabolic syndrome markers were equally lackluster: no difference in fasting glucose, mean glucose, or fasting insulin.
There was some weight change. The control group, which got that dietary education, lost 1.5% of body weight over the 3 months. The time-restricted-eating group lost 3.3% — about 7 pounds, which is reasonable.
With that weight loss came statistically significant, albeit modest improvements in BMI, body fat percentage, and LDL cholesterol.
Of interest, despite the larger weight loss in the intermittent-fasting group, there was no difference in muscle mass loss, which is encouraging.
Taken together, we can say that, yes, it seems like time-restricted eating can help people lose some weight. This is essentially due to the fact that people eat fewer calories when they do time-restricted eating, as you can see here.
But, in the end, this trial examined whether this relatively straightforward lifestyle intervention would move the needle in terms of metabolic syndrome, and the data are not very compelling for that.
This graph shows how many of those five factors for metabolic syndrome the individuals in this trial had from the start to the end. You see that, over the 3 months, seven people in the time-restricted-eating group moved from having three criteria to two or one — being “cured” of metabolic syndrome, if you will. Nine people in the standard group were cured by that definition. Remember, they had to have at least three to have the syndrome and thus be eligible for the trial. 
So If it just leads to weight loss by forcing people to consume less calories, then we need to acknowledge that we probably have better methods to achieve this same end. Ten years ago, I would have said that lifestyle change is the only way to end the epidemic of the metabolic syndrome in this country. Today, well, we live in a world of GLP-1 weight loss drugs. It is simply a different world now. Yes, they are expensive. Yes, they have side effects. But we need to evaluate them against the comparison. And so far, lifestyle changes alone are really no comparison.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
New Guidelines Emphasize Liver Care in T2D, Obesity
MADRID — Individuals with type 2 diabetes and/or obesity plus one or more metabolic risk factors are at a higher risk for metabolic dysfunction–associated steatotic liver disease (MASLD) with fibrosis and progression to more severe liver disease, stated new European guidelines that provide recommendations for diagnosis and management.
“The availability of improved treatment options underlines the need to identify at-risk individuals with MASLD early, as we now possess the tools to positively influence the course of the diseases, which is expected to prevent relevant clinical events,” stated the clinical practice guidelines, updated for the first time since 2016.
“Now we have guidelines that tell clinicians how to monitor the liver,” said Amalia Gastaldelli, PhD, research director at the Institute of Clinical Physiology of the National Research Council in Pisa, Italy, and a member of the panel that developed the guidelines.
Dr. Gastaldelli moderated a session focused on the guidelines at the annual meeting of the European Association for the Study of Diabetes (EASD). In an interview after the session, Dr. Gastaldelli, who leads a cardiometabolic risk research group, stressed the importance of the liver’s role in the body and the need for diabetes specialists to start paying more attention to this vital organ.
“It’s an important organ for monitoring because liver disease is silent, and the patient doesn’t feel unwell until disease is severe,” she said. “Diabetologists already monitor the eye, the heart, the kidney, and so on, but the liver is often neglected,” she said. A 2024 study found that the global pooled prevalence of MASLD among patients with type 2 diabetes was 65.33%.
Dr. Gastaldelli noted the importance of liver status in diabetes care. The liver makes triglycerides and very-low-density lipoprotein cholesterol, which are all major risk factors for atherosclerosis and cardiovascular disease (CVD), she said, as well as producing glucose, which in excess can lead to hyperglycemia.
The guidelines were jointly written by EASD, the European Association for the Study of the Liver, and the European Association for the Study of Obesity, and published in Diabetologia, The Journal of Hepatology, and Obesity Facts.
A Metabolic Condition
In the EASD meeting session, Dr. Gastaldelli discussed the reasons for, and implications of, shifting the name from nonalcoholic fatty liver disease (NAFLD) to MASLD.
“The name change focuses on the fact that this is a metabolic disease, while NAFLD had no mention of this and was considered stigmatizing by patients, especially in relation to the words ‘fatty’ and ‘nonalcoholic,’” she said.
According to the guidelines, MASLD is defined as liver steatosis in the presence of one or more cardiometabolic risk factor(s) and the absence of excess alcohol intake.
MASLD has become the most common chronic liver disease and includes isolated steatosis, metabolic dysfunction-associated steatohepatitis (MASH, previously NASH), MASH-related fibrosis, and cirrhosis.
In the overarching group of steatotic liver disease, a totally new intermediate category has been added: MASLD with moderate (increased) alcohol intake (MetALD), which represents MASLD in people who consume greater amounts of alcohol per week (140-350 g/week and 210-420 g/week for women and men, respectively).
The change in the nomenclature has been incremental and regional, Dr. Gastaldelli said. “The definition first changed from NAFLD to MAFLD, which recognizes the importance of metabolism in the pathophysiology of this disease but does not take into account alcohol intake. MAFLD is still used in Asia, Australasia, and North Africa, while Europe and the Americas have endorsed MASLD.”
Case-Finding and Diagnosis
Identifying MASLD cases in people at risk remains incidental, largely because it is a silent disease and is symptom-free until it becomes severe, said Dr. Gastaldelli.
The guideline recognizes that individuals with type 2 diabetes or obesity with additional metabolic risk factor(s) are at a higher risk for MASLD with fibrosis and progression to MASH.
Assessment strategies for severe liver fibrosis in MASLD include the use of noninvasive tests in people who have cardiometabolic risk factors, abnormal liver enzymes, and/or radiological signs of hepatic steatosis, particularly in the presence of type 2 diabetes or obesity or in the presence of one or more metabolic risk factors.
Dr. Gastaldelli noted that type 2 diabetes, metabolic syndrome, and obesity, including abdominal obesity identified by large waist circumference, are the major risk factors and should be warning signs.
“We need to consider abdominal obesity too — we’ve published data in relatively lean people, body mass index < 25, with MASH but without diabetes. Most of the patients accumulated fat viscerally and in the liver and had hypertriglyceridemia and hypercholesterolemia,” she said.
“The guidelines reflect this because the definition of MASLD includes steatosis plus at least one metabolic factor — waist circumference, for example, which is related to visceral fat, hyperlipidemia, or hyperglycemia. Of note, in both pharmacological and diet-induced weight loss, the decrease in liver fat was associated with the decrease in visceral fat.”
The noninvasive biomarker test, Fibrosis-4 (FIB-4) may be used to assess the risk for liver fibrosis. The FIB-4 index is calculated using a patient’s age and results of three blood tests — aspartate aminotransferase, alanine aminotransferase, and platelet count.
Advanced fibrosis (grade F3-F4) “is a major risk factor for severe outcomes,” said Dr. Gastaldelli. A FIB-4 test result below 1.3 indicates low risk for advanced liver fibrosis, 1.30-2.67 indicates intermediate risk, and above 2.67 indicates high risk.
“When fibrosis increases, then liver enzymes increase and the platelets decrease,” said Dr. Gastaldelli. “It is not a perfect tool, and we need to add in age because at a young age, it is prone to false negatives and when very old — false positives. It’s important to take a global view, especially if the patient has persistent high liver enzymes, but FIB-4 is low.”
“And if they have more than one metabolic risk factor, proceed with more tests, for example, transient elastography,” she advised. Imaging techniques such as transient elastography may rule out or rule in advanced fibrosis, which is predictive of liver-related outcomes.
“However, imaging techniques only diagnose steatosis and fibrosis, and right now, MASH can only be diagnosed with liver biopsy because we do not have any markers of liver inflammation and ballooning. In the future, noninvasive tests based on imaging and blood tests will be used to identify patients with MASH,” she added.
Management of MASLD — Lifestyle and Treatment
“Pharmacological treatments are designed for [patients] with MASH and fibrosis grade F2 or F3, but not MASLD,” Dr. Gastaldelli said. As such, lifestyle interventions are the mainstay of management — including weight loss, dietary changes, physical exercise, and low to no alcohol consumption. “Eating good-quality food and reducing calories are both important because the metabolism responds differently to different nutrients,” Dr. Gastaldelli said.
“In particular, the guidelines advise dietary management because some foods carry liver toxicity, for example, sugary foods with sucrose/fructose especially,” she said, adding that, “complex carbohydrates are less harmful than refined carbohydrates. Processed foods should be avoided if possible because they contain sugars, [as well as] saturated fats and hydrogenated fat, which is particularly bad for the liver. Olive oil is better than butter or margarine, which are rich in saturated fat, and fish and white meat are preferable.”
She added that a diet to help manage type 2 diabetes was not so dissimilar because sugar again needs to be reduced.
If a patient has severe obesity (and MASLD), data show that bariatric surgery is beneficial. “It not only helps weight loss, but it improves liver histology and has been shown to improve or resolve type 2 diabetes and reduce CVD risk. Importantly, regarding fibrosis, nutritional management after the bariatric surgery is the most important thing,” said Dr. Gastaldelli.
Optimal management of comorbidities — including the use of incretin-based therapies such as semaglutide or tirzepatide for type 2 diabetes or obesity, if indicated — is advised, according to the guidelines.
Glucagon-like peptide 1 receptor agonists (GLP-1 RAs) have been shown to have a beneficial effect on MASH, said Dr. Gastaldelli. “They have not shown effectiveness in the resolution of fibrosis, but this might take longer to manifest. However, if the medication is started early enough, it may prevent severe fibrosis. Significant weight loss, both with lifestyle and pharmacological treatment, should lead to an improvement in the liver too.”
There are currently no drugs available in Europe for the treatment of noncirrhotic MASH and severe fibrosis (stage ≥ 2). Resmetirom is the first approved MASH-targeted treatment in noncirrhotic MASH and significant liver fibrosis, with histological effectiveness on steatohepatitis and fibrosis, together with an acceptable safety and tolerability profile, but, for the moment, this agent is only available in United States.
Finally, turning to MASH-related cirrhosis, the guidelines advise adaptations of metabolic drugs, nutritional counseling, and surveillance for portal hypertension and hepatocellular carcinoma, as well as liver transplantation in decompensated cirrhosis.
After the session, this news organization spoke to Tushy Kailayanathan, MBBS BSc, medical director of the liver MRI company, Perspectum, who reviewed the limitations of the FIB-4 test. The FIB-4 test identifies those with advanced fibrosis in the liver, for example, patients with hepatitis C, she noted; however, “it performs worse in type 2 diabetic patients and in the elderly. There is little clinical guidance on the adjustment of FIB-4 thresholds needed for these high cardiometabolic risk groups. The priority patients are missed by FIB-4 because those individuals with early and active disease may not yet have progressed to advanced disease detected by FIB-4.”
These individuals are exactly those amenable to primary care prevention strategies, said Dr. Kailayanathan. Because of the nature of early and active liver disease in patients with high cardiometabolic risk, it would make sense to shift some diagnostic protocols into primary care.
“These individuals are exactly those amenable to primary care prevention strategies at annual diabetic review because they are likely to have modifiable cardiometabolic risk factors such as metabolic syndrome and would benefit from lifestyle and therapeutic intervention, including GLP-1 RAs and SGLT2is [sodium-glucose cotransporter-2 inhibitors],” she said. “Case-finding and detection of early-stage MASLD is a priority in diabetics, and there is an unmet need for accurate biomarkers to measure liver fat and inflammation early.”
Dr. Gastaldelli has been on the advisory board or consulting for Boehringer Ingelheim, Novo Nordisk, Eli Lilly, Fractyl, Pfizer, Merck-MSD, MetaDeq and a speaker for Eli Lilly, Novo Nordisk, and Pfizer. Dr. Kailayanathan is medical director at Perspectum, a UK-based company involved in liver imaging technology.
A version of this article first appeared on Medscape.com.
MADRID — Individuals with type 2 diabetes and/or obesity plus one or more metabolic risk factors are at a higher risk for metabolic dysfunction–associated steatotic liver disease (MASLD) with fibrosis and progression to more severe liver disease, stated new European guidelines that provide recommendations for diagnosis and management.
“The availability of improved treatment options underlines the need to identify at-risk individuals with MASLD early, as we now possess the tools to positively influence the course of the diseases, which is expected to prevent relevant clinical events,” stated the clinical practice guidelines, updated for the first time since 2016.
“Now we have guidelines that tell clinicians how to monitor the liver,” said Amalia Gastaldelli, PhD, research director at the Institute of Clinical Physiology of the National Research Council in Pisa, Italy, and a member of the panel that developed the guidelines.
Dr. Gastaldelli moderated a session focused on the guidelines at the annual meeting of the European Association for the Study of Diabetes (EASD). In an interview after the session, Dr. Gastaldelli, who leads a cardiometabolic risk research group, stressed the importance of the liver’s role in the body and the need for diabetes specialists to start paying more attention to this vital organ.
“It’s an important organ for monitoring because liver disease is silent, and the patient doesn’t feel unwell until disease is severe,” she said. “Diabetologists already monitor the eye, the heart, the kidney, and so on, but the liver is often neglected,” she said. A 2024 study found that the global pooled prevalence of MASLD among patients with type 2 diabetes was 65.33%.
Dr. Gastaldelli noted the importance of liver status in diabetes care. The liver makes triglycerides and very-low-density lipoprotein cholesterol, which are all major risk factors for atherosclerosis and cardiovascular disease (CVD), she said, as well as producing glucose, which in excess can lead to hyperglycemia.
The guidelines were jointly written by EASD, the European Association for the Study of the Liver, and the European Association for the Study of Obesity, and published in Diabetologia, The Journal of Hepatology, and Obesity Facts.
A Metabolic Condition
In the EASD meeting session, Dr. Gastaldelli discussed the reasons for, and implications of, shifting the name from nonalcoholic fatty liver disease (NAFLD) to MASLD.
“The name change focuses on the fact that this is a metabolic disease, while NAFLD had no mention of this and was considered stigmatizing by patients, especially in relation to the words ‘fatty’ and ‘nonalcoholic,’” she said.
According to the guidelines, MASLD is defined as liver steatosis in the presence of one or more cardiometabolic risk factor(s) and the absence of excess alcohol intake.
MASLD has become the most common chronic liver disease and includes isolated steatosis, metabolic dysfunction-associated steatohepatitis (MASH, previously NASH), MASH-related fibrosis, and cirrhosis.
In the overarching group of steatotic liver disease, a totally new intermediate category has been added: MASLD with moderate (increased) alcohol intake (MetALD), which represents MASLD in people who consume greater amounts of alcohol per week (140-350 g/week and 210-420 g/week for women and men, respectively).
The change in the nomenclature has been incremental and regional, Dr. Gastaldelli said. “The definition first changed from NAFLD to MAFLD, which recognizes the importance of metabolism in the pathophysiology of this disease but does not take into account alcohol intake. MAFLD is still used in Asia, Australasia, and North Africa, while Europe and the Americas have endorsed MASLD.”
Case-Finding and Diagnosis
Identifying MASLD cases in people at risk remains incidental, largely because it is a silent disease and is symptom-free until it becomes severe, said Dr. Gastaldelli.
The guideline recognizes that individuals with type 2 diabetes or obesity with additional metabolic risk factor(s) are at a higher risk for MASLD with fibrosis and progression to MASH.
Assessment strategies for severe liver fibrosis in MASLD include the use of noninvasive tests in people who have cardiometabolic risk factors, abnormal liver enzymes, and/or radiological signs of hepatic steatosis, particularly in the presence of type 2 diabetes or obesity or in the presence of one or more metabolic risk factors.
Dr. Gastaldelli noted that type 2 diabetes, metabolic syndrome, and obesity, including abdominal obesity identified by large waist circumference, are the major risk factors and should be warning signs.
“We need to consider abdominal obesity too — we’ve published data in relatively lean people, body mass index < 25, with MASH but without diabetes. Most of the patients accumulated fat viscerally and in the liver and had hypertriglyceridemia and hypercholesterolemia,” she said.
“The guidelines reflect this because the definition of MASLD includes steatosis plus at least one metabolic factor — waist circumference, for example, which is related to visceral fat, hyperlipidemia, or hyperglycemia. Of note, in both pharmacological and diet-induced weight loss, the decrease in liver fat was associated with the decrease in visceral fat.”
The noninvasive biomarker test, Fibrosis-4 (FIB-4) may be used to assess the risk for liver fibrosis. The FIB-4 index is calculated using a patient’s age and results of three blood tests — aspartate aminotransferase, alanine aminotransferase, and platelet count.
Advanced fibrosis (grade F3-F4) “is a major risk factor for severe outcomes,” said Dr. Gastaldelli. A FIB-4 test result below 1.3 indicates low risk for advanced liver fibrosis, 1.30-2.67 indicates intermediate risk, and above 2.67 indicates high risk.
“When fibrosis increases, then liver enzymes increase and the platelets decrease,” said Dr. Gastaldelli. “It is not a perfect tool, and we need to add in age because at a young age, it is prone to false negatives and when very old — false positives. It’s important to take a global view, especially if the patient has persistent high liver enzymes, but FIB-4 is low.”
“And if they have more than one metabolic risk factor, proceed with more tests, for example, transient elastography,” she advised. Imaging techniques such as transient elastography may rule out or rule in advanced fibrosis, which is predictive of liver-related outcomes.
“However, imaging techniques only diagnose steatosis and fibrosis, and right now, MASH can only be diagnosed with liver biopsy because we do not have any markers of liver inflammation and ballooning. In the future, noninvasive tests based on imaging and blood tests will be used to identify patients with MASH,” she added.
Management of MASLD — Lifestyle and Treatment
“Pharmacological treatments are designed for [patients] with MASH and fibrosis grade F2 or F3, but not MASLD,” Dr. Gastaldelli said. As such, lifestyle interventions are the mainstay of management — including weight loss, dietary changes, physical exercise, and low to no alcohol consumption. “Eating good-quality food and reducing calories are both important because the metabolism responds differently to different nutrients,” Dr. Gastaldelli said.
“In particular, the guidelines advise dietary management because some foods carry liver toxicity, for example, sugary foods with sucrose/fructose especially,” she said, adding that, “complex carbohydrates are less harmful than refined carbohydrates. Processed foods should be avoided if possible because they contain sugars, [as well as] saturated fats and hydrogenated fat, which is particularly bad for the liver. Olive oil is better than butter or margarine, which are rich in saturated fat, and fish and white meat are preferable.”
She added that a diet to help manage type 2 diabetes was not so dissimilar because sugar again needs to be reduced.
If a patient has severe obesity (and MASLD), data show that bariatric surgery is beneficial. “It not only helps weight loss, but it improves liver histology and has been shown to improve or resolve type 2 diabetes and reduce CVD risk. Importantly, regarding fibrosis, nutritional management after the bariatric surgery is the most important thing,” said Dr. Gastaldelli.
Optimal management of comorbidities — including the use of incretin-based therapies such as semaglutide or tirzepatide for type 2 diabetes or obesity, if indicated — is advised, according to the guidelines.
Glucagon-like peptide 1 receptor agonists (GLP-1 RAs) have been shown to have a beneficial effect on MASH, said Dr. Gastaldelli. “They have not shown effectiveness in the resolution of fibrosis, but this might take longer to manifest. However, if the medication is started early enough, it may prevent severe fibrosis. Significant weight loss, both with lifestyle and pharmacological treatment, should lead to an improvement in the liver too.”
There are currently no drugs available in Europe for the treatment of noncirrhotic MASH and severe fibrosis (stage ≥ 2). Resmetirom is the first approved MASH-targeted treatment in noncirrhotic MASH and significant liver fibrosis, with histological effectiveness on steatohepatitis and fibrosis, together with an acceptable safety and tolerability profile, but, for the moment, this agent is only available in United States.
Finally, turning to MASH-related cirrhosis, the guidelines advise adaptations of metabolic drugs, nutritional counseling, and surveillance for portal hypertension and hepatocellular carcinoma, as well as liver transplantation in decompensated cirrhosis.
After the session, this news organization spoke to Tushy Kailayanathan, MBBS BSc, medical director of the liver MRI company, Perspectum, who reviewed the limitations of the FIB-4 test. The FIB-4 test identifies those with advanced fibrosis in the liver, for example, patients with hepatitis C, she noted; however, “it performs worse in type 2 diabetic patients and in the elderly. There is little clinical guidance on the adjustment of FIB-4 thresholds needed for these high cardiometabolic risk groups. The priority patients are missed by FIB-4 because those individuals with early and active disease may not yet have progressed to advanced disease detected by FIB-4.”
These individuals are exactly those amenable to primary care prevention strategies, said Dr. Kailayanathan. Because of the nature of early and active liver disease in patients with high cardiometabolic risk, it would make sense to shift some diagnostic protocols into primary care.
“These individuals are exactly those amenable to primary care prevention strategies at annual diabetic review because they are likely to have modifiable cardiometabolic risk factors such as metabolic syndrome and would benefit from lifestyle and therapeutic intervention, including GLP-1 RAs and SGLT2is [sodium-glucose cotransporter-2 inhibitors],” she said. “Case-finding and detection of early-stage MASLD is a priority in diabetics, and there is an unmet need for accurate biomarkers to measure liver fat and inflammation early.”
Dr. Gastaldelli has been on the advisory board or consulting for Boehringer Ingelheim, Novo Nordisk, Eli Lilly, Fractyl, Pfizer, Merck-MSD, MetaDeq and a speaker for Eli Lilly, Novo Nordisk, and Pfizer. Dr. Kailayanathan is medical director at Perspectum, a UK-based company involved in liver imaging technology.
A version of this article first appeared on Medscape.com.
MADRID — Individuals with type 2 diabetes and/or obesity plus one or more metabolic risk factors are at a higher risk for metabolic dysfunction–associated steatotic liver disease (MASLD) with fibrosis and progression to more severe liver disease, stated new European guidelines that provide recommendations for diagnosis and management.
“The availability of improved treatment options underlines the need to identify at-risk individuals with MASLD early, as we now possess the tools to positively influence the course of the diseases, which is expected to prevent relevant clinical events,” stated the clinical practice guidelines, updated for the first time since 2016.
“Now we have guidelines that tell clinicians how to monitor the liver,” said Amalia Gastaldelli, PhD, research director at the Institute of Clinical Physiology of the National Research Council in Pisa, Italy, and a member of the panel that developed the guidelines.
Dr. Gastaldelli moderated a session focused on the guidelines at the annual meeting of the European Association for the Study of Diabetes (EASD). In an interview after the session, Dr. Gastaldelli, who leads a cardiometabolic risk research group, stressed the importance of the liver’s role in the body and the need for diabetes specialists to start paying more attention to this vital organ.
“It’s an important organ for monitoring because liver disease is silent, and the patient doesn’t feel unwell until disease is severe,” she said. “Diabetologists already monitor the eye, the heart, the kidney, and so on, but the liver is often neglected,” she said. A 2024 study found that the global pooled prevalence of MASLD among patients with type 2 diabetes was 65.33%.
Dr. Gastaldelli noted the importance of liver status in diabetes care. The liver makes triglycerides and very-low-density lipoprotein cholesterol, which are all major risk factors for atherosclerosis and cardiovascular disease (CVD), she said, as well as producing glucose, which in excess can lead to hyperglycemia.
The guidelines were jointly written by EASD, the European Association for the Study of the Liver, and the European Association for the Study of Obesity, and published in Diabetologia, The Journal of Hepatology, and Obesity Facts.
A Metabolic Condition
In the EASD meeting session, Dr. Gastaldelli discussed the reasons for, and implications of, shifting the name from nonalcoholic fatty liver disease (NAFLD) to MASLD.
“The name change focuses on the fact that this is a metabolic disease, while NAFLD had no mention of this and was considered stigmatizing by patients, especially in relation to the words ‘fatty’ and ‘nonalcoholic,’” she said.
According to the guidelines, MASLD is defined as liver steatosis in the presence of one or more cardiometabolic risk factor(s) and the absence of excess alcohol intake.
MASLD has become the most common chronic liver disease and includes isolated steatosis, metabolic dysfunction-associated steatohepatitis (MASH, previously NASH), MASH-related fibrosis, and cirrhosis.
In the overarching group of steatotic liver disease, a totally new intermediate category has been added: MASLD with moderate (increased) alcohol intake (MetALD), which represents MASLD in people who consume greater amounts of alcohol per week (140-350 g/week and 210-420 g/week for women and men, respectively).
The change in the nomenclature has been incremental and regional, Dr. Gastaldelli said. “The definition first changed from NAFLD to MAFLD, which recognizes the importance of metabolism in the pathophysiology of this disease but does not take into account alcohol intake. MAFLD is still used in Asia, Australasia, and North Africa, while Europe and the Americas have endorsed MASLD.”
Case-Finding and Diagnosis
Identifying MASLD cases in people at risk remains incidental, largely because it is a silent disease and is symptom-free until it becomes severe, said Dr. Gastaldelli.
The guideline recognizes that individuals with type 2 diabetes or obesity with additional metabolic risk factor(s) are at a higher risk for MASLD with fibrosis and progression to MASH.
Assessment strategies for severe liver fibrosis in MASLD include the use of noninvasive tests in people who have cardiometabolic risk factors, abnormal liver enzymes, and/or radiological signs of hepatic steatosis, particularly in the presence of type 2 diabetes or obesity or in the presence of one or more metabolic risk factors.
Dr. Gastaldelli noted that type 2 diabetes, metabolic syndrome, and obesity, including abdominal obesity identified by large waist circumference, are the major risk factors and should be warning signs.
“We need to consider abdominal obesity too — we’ve published data in relatively lean people, body mass index < 25, with MASH but without diabetes. Most of the patients accumulated fat viscerally and in the liver and had hypertriglyceridemia and hypercholesterolemia,” she said.
“The guidelines reflect this because the definition of MASLD includes steatosis plus at least one metabolic factor — waist circumference, for example, which is related to visceral fat, hyperlipidemia, or hyperglycemia. Of note, in both pharmacological and diet-induced weight loss, the decrease in liver fat was associated with the decrease in visceral fat.”
The noninvasive biomarker test, Fibrosis-4 (FIB-4) may be used to assess the risk for liver fibrosis. The FIB-4 index is calculated using a patient’s age and results of three blood tests — aspartate aminotransferase, alanine aminotransferase, and platelet count.
Advanced fibrosis (grade F3-F4) “is a major risk factor for severe outcomes,” said Dr. Gastaldelli. A FIB-4 test result below 1.3 indicates low risk for advanced liver fibrosis, 1.30-2.67 indicates intermediate risk, and above 2.67 indicates high risk.
“When fibrosis increases, then liver enzymes increase and the platelets decrease,” said Dr. Gastaldelli. “It is not a perfect tool, and we need to add in age because at a young age, it is prone to false negatives and when very old — false positives. It’s important to take a global view, especially if the patient has persistent high liver enzymes, but FIB-4 is low.”
“And if they have more than one metabolic risk factor, proceed with more tests, for example, transient elastography,” she advised. Imaging techniques such as transient elastography may rule out or rule in advanced fibrosis, which is predictive of liver-related outcomes.
“However, imaging techniques only diagnose steatosis and fibrosis, and right now, MASH can only be diagnosed with liver biopsy because we do not have any markers of liver inflammation and ballooning. In the future, noninvasive tests based on imaging and blood tests will be used to identify patients with MASH,” she added.
Management of MASLD — Lifestyle and Treatment
“Pharmacological treatments are designed for [patients] with MASH and fibrosis grade F2 or F3, but not MASLD,” Dr. Gastaldelli said. As such, lifestyle interventions are the mainstay of management — including weight loss, dietary changes, physical exercise, and low to no alcohol consumption. “Eating good-quality food and reducing calories are both important because the metabolism responds differently to different nutrients,” Dr. Gastaldelli said.
“In particular, the guidelines advise dietary management because some foods carry liver toxicity, for example, sugary foods with sucrose/fructose especially,” she said, adding that, “complex carbohydrates are less harmful than refined carbohydrates. Processed foods should be avoided if possible because they contain sugars, [as well as] saturated fats and hydrogenated fat, which is particularly bad for the liver. Olive oil is better than butter or margarine, which are rich in saturated fat, and fish and white meat are preferable.”
She added that a diet to help manage type 2 diabetes was not so dissimilar because sugar again needs to be reduced.
If a patient has severe obesity (and MASLD), data show that bariatric surgery is beneficial. “It not only helps weight loss, but it improves liver histology and has been shown to improve or resolve type 2 diabetes and reduce CVD risk. Importantly, regarding fibrosis, nutritional management after the bariatric surgery is the most important thing,” said Dr. Gastaldelli.
Optimal management of comorbidities — including the use of incretin-based therapies such as semaglutide or tirzepatide for type 2 diabetes or obesity, if indicated — is advised, according to the guidelines.
Glucagon-like peptide 1 receptor agonists (GLP-1 RAs) have been shown to have a beneficial effect on MASH, said Dr. Gastaldelli. “They have not shown effectiveness in the resolution of fibrosis, but this might take longer to manifest. However, if the medication is started early enough, it may prevent severe fibrosis. Significant weight loss, both with lifestyle and pharmacological treatment, should lead to an improvement in the liver too.”
There are currently no drugs available in Europe for the treatment of noncirrhotic MASH and severe fibrosis (stage ≥ 2). Resmetirom is the first approved MASH-targeted treatment in noncirrhotic MASH and significant liver fibrosis, with histological effectiveness on steatohepatitis and fibrosis, together with an acceptable safety and tolerability profile, but, for the moment, this agent is only available in United States.
Finally, turning to MASH-related cirrhosis, the guidelines advise adaptations of metabolic drugs, nutritional counseling, and surveillance for portal hypertension and hepatocellular carcinoma, as well as liver transplantation in decompensated cirrhosis.
After the session, this news organization spoke to Tushy Kailayanathan, MBBS BSc, medical director of the liver MRI company, Perspectum, who reviewed the limitations of the FIB-4 test. The FIB-4 test identifies those with advanced fibrosis in the liver, for example, patients with hepatitis C, she noted; however, “it performs worse in type 2 diabetic patients and in the elderly. There is little clinical guidance on the adjustment of FIB-4 thresholds needed for these high cardiometabolic risk groups. The priority patients are missed by FIB-4 because those individuals with early and active disease may not yet have progressed to advanced disease detected by FIB-4.”
These individuals are exactly those amenable to primary care prevention strategies, said Dr. Kailayanathan. Because of the nature of early and active liver disease in patients with high cardiometabolic risk, it would make sense to shift some diagnostic protocols into primary care.
“These individuals are exactly those amenable to primary care prevention strategies at annual diabetic review because they are likely to have modifiable cardiometabolic risk factors such as metabolic syndrome and would benefit from lifestyle and therapeutic intervention, including GLP-1 RAs and SGLT2is [sodium-glucose cotransporter-2 inhibitors],” she said. “Case-finding and detection of early-stage MASLD is a priority in diabetics, and there is an unmet need for accurate biomarkers to measure liver fat and inflammation early.”
Dr. Gastaldelli has been on the advisory board or consulting for Boehringer Ingelheim, Novo Nordisk, Eli Lilly, Fractyl, Pfizer, Merck-MSD, MetaDeq and a speaker for Eli Lilly, Novo Nordisk, and Pfizer. Dr. Kailayanathan is medical director at Perspectum, a UK-based company involved in liver imaging technology.
A version of this article first appeared on Medscape.com.
FROM EASD 2024
Doctors Seek Additional Obesity Training in Wake of Obesity Patient Boom
Gitanjali Srivastava, MD, professor of medicine, pediatrics, and surgery, and medical director of obesity medicine at Vanderbilt University School of Medicine in Nashville, Tennessee, was nearly 10 years into practicing pediatric medicine when she graduated from the obesity medicine fellowship at Massachusetts General Hospital in Boston in 2013. “We were the very first sort of fellows to speak of then; there were no standards or curriculum,” she said.
Obesity was already epidemic, but stigma and bias were still pervasive in the medical community and within the public. After graduating, Dr. Srivastava spent months vying for a position with hospital CEOs. She traveled across the country explaining the specialty and its value, going into detail about the budget, business model, space requirement, and revenue potential of obesity medicine.
Today marks a very different era.
Obesity medicine is exploding. Patients are spilling into doctors’ offices looking for obesity treatment. Healthcare systems are seeking out obesity specialists and building metabolic health centers. Next month, another 2115 doctors from primary care, surgery, orthopedics, pediatrics, fertility, endocrinology, and beyond will sit for the 2024 exam. The once niche specialty is quickly becoming intertwined with most of modern medicine.
The Need to Treat
It’s no mystery that the rapid expansion of obesity medicine coincides with the US Food and Drug Administration’s approval of GLP-1 injections. The drugs’ radical weight loss properties have captured headlines and driven up patient demand. Meanwhile, doctors are finally able to offer effective treatment for a disease that affects 40% of US adults.
“We are finally treating it as a chronic disease, not as a lifestyle,” said Marcio Griebeler, MD, director of the obesity medicine fellowship at the Cleveland Clinic. And “I think it’s fulfilling for physicians,” he said.
For so long, the advice for obesity was about lifestyle. Move more, eat less, and harness willpower, “which really is a fallacy,” said Kimberly Gudzune, MD, MPH, an obesity medicine specialist and chief medical officer for the American Board of Obesity Medicine (ABOM) Foundation. For people with obesity, “your brain is operating differently,” she said. “Your body really is set up to work against you.”
Brianna Johnson-Rabbett, MD, medical director of the ABOM, told this news organization that with the advent of GLP-1s, “there’s a clearer recognition that obesity is a disease that needs to be treated like other diseases.” Some of that is thanks to clinical trial data showing that just as with other diseases such as high blood pressure or diabetes, obesity can be treated with medication and it resurges when the medication is stopped, she said.
Doctors don’t have to go looking for patients with obesity, dr. Griebeler added. Now that treatment options exist, they’re showing up in droves at the doctor’s office — all the doctors’ offices. In primary care, endocrinology, surgery, pediatrics — a wide variety of doctors are being asked about obesity drugs, Dr. Griebeler noted.
And while doctors are often just as excited as patients about the potential for treatment, many find themselves under-equipped when it comes to obesity. “More physicians are ... recognizing the value in treating this, and some are realizing, “Oh gosh, I never learned how to do this,” said Dr. Gudzune.
Information Patients Have Been Waiting For
Medical training has traditionally devoted minimal, if any, curriculum to obesity and metabolism. “To be honest, we didn’t really cover this at all in my training,” said Nina Paddu, MD, obesity medicine specialist at Maimonides Medical Center in New York City who finished her training only 2 years ago. “The guidance even in residency was ‘let’s send them to nutrition’ and ‘recommend exercising.’ ”
In addition to the medical education gap, until recently there was a “paucity of robust evidence,” Dr. Srivastava said. Leaders in the field wanted to establish standards and guidelines, but there wasn’t enough strong evidence on obesity and its treatments to build them, she said.
Only in the last 5 years or so has the evidence-based understanding of obesity’s pathophysiology truly accelerated: The brain’s driving roles, its interplay with hormones, and its interactions with other diseases. “We are just at the cusp of understanding all the different factors,” Dr. Gudzune said.
But already endocrinologists, surgeons, fertility specialists, gynecologists, and oncologists, to name a few, see the critical overlap with their own field. “Conditions were once suspected of being intertwined [with obesity], and now we have data to connect them,” Dr. Srivastava said. For example, there’s now data connecting semaglutide to a 20% reduction in cardiovascular events for people with obesity. That’s a game changer for multiple specialties, she told this news organization.
Getting Trained in Obesity Management
The recent uptick in obesity insights and increased patient need has doctors from every career stage seeking additional training.
The ABOM offers two board certification pathways: 60 hours of CME credits or a 12-month fellowship. Both paths require doctors to pass the board’s exam.
Many doctors incorporate the training into their existing practice. The CME credit pathway, especially, is designed to help get doctors up to speed without requiring them to upend their lives for a fellowship.
Dr. Srivastava said that the fellowship is more consuming and immersive. While it’s often younger doctors just out of training who apply to fellowship, every year, “I’m astonished at the number of talented physicians with clinical and research experience who want to immerse themselves in a fellowship experience.”
Some doctors return to their previous specialties after fellowship. But many will go on to take obesity medicine–specific roles or set aside clinic hours for obesity medicine. Their credentials are “really attractive to institutions, especially those looking to open up obesity medicine or weight management programs,” said Dr. Srivastava.
Dr. Paddu, who finished her obesity medicine fellowship this year, said there are a variety of obesity medicine jobs to choose from — far different from Dr. Srivastava’s job search 15 years ago. Dr. Paddu’s new role combines 2 days of primary care with 2 days devoted to obesity medicine and 1 day each week set aside for administrative work so she can build up the hospital’s new metabolic health clinic.
Still Not Enough Obesity Specialists
As with all things, rapid growth requires careful oversight. “Part of the responsibility of the board is to think critically of how the field is growing” and conduct ongoing monitoring, Dr. Gudzune said.
This is also why the board’s credentials are time-limited and must be recertified, Dr. Johnson-Rabbett added.
But even with the rise in certified doctors and obesity medicine positions, the 8263 doctors certified by ABOM are only a tiny fraction of US physicians. As a result, there’s genuine likelihood that many patients seeking GLP-1s or other obesity treatment don’t yet have access to the holistic care they need. Plus, doctors may still not have obesity expertise within their networks.
“The field has grown rapidly, but it’s still such a small field relative to the patient need,” said Dr. Gudzune.
A version of this article appeared on Medscape.com.
Gitanjali Srivastava, MD, professor of medicine, pediatrics, and surgery, and medical director of obesity medicine at Vanderbilt University School of Medicine in Nashville, Tennessee, was nearly 10 years into practicing pediatric medicine when she graduated from the obesity medicine fellowship at Massachusetts General Hospital in Boston in 2013. “We were the very first sort of fellows to speak of then; there were no standards or curriculum,” she said.
Obesity was already epidemic, but stigma and bias were still pervasive in the medical community and within the public. After graduating, Dr. Srivastava spent months vying for a position with hospital CEOs. She traveled across the country explaining the specialty and its value, going into detail about the budget, business model, space requirement, and revenue potential of obesity medicine.
Today marks a very different era.
Obesity medicine is exploding. Patients are spilling into doctors’ offices looking for obesity treatment. Healthcare systems are seeking out obesity specialists and building metabolic health centers. Next month, another 2115 doctors from primary care, surgery, orthopedics, pediatrics, fertility, endocrinology, and beyond will sit for the 2024 exam. The once niche specialty is quickly becoming intertwined with most of modern medicine.
The Need to Treat
It’s no mystery that the rapid expansion of obesity medicine coincides with the US Food and Drug Administration’s approval of GLP-1 injections. The drugs’ radical weight loss properties have captured headlines and driven up patient demand. Meanwhile, doctors are finally able to offer effective treatment for a disease that affects 40% of US adults.
“We are finally treating it as a chronic disease, not as a lifestyle,” said Marcio Griebeler, MD, director of the obesity medicine fellowship at the Cleveland Clinic. And “I think it’s fulfilling for physicians,” he said.
For so long, the advice for obesity was about lifestyle. Move more, eat less, and harness willpower, “which really is a fallacy,” said Kimberly Gudzune, MD, MPH, an obesity medicine specialist and chief medical officer for the American Board of Obesity Medicine (ABOM) Foundation. For people with obesity, “your brain is operating differently,” she said. “Your body really is set up to work against you.”
Brianna Johnson-Rabbett, MD, medical director of the ABOM, told this news organization that with the advent of GLP-1s, “there’s a clearer recognition that obesity is a disease that needs to be treated like other diseases.” Some of that is thanks to clinical trial data showing that just as with other diseases such as high blood pressure or diabetes, obesity can be treated with medication and it resurges when the medication is stopped, she said.
Doctors don’t have to go looking for patients with obesity, dr. Griebeler added. Now that treatment options exist, they’re showing up in droves at the doctor’s office — all the doctors’ offices. In primary care, endocrinology, surgery, pediatrics — a wide variety of doctors are being asked about obesity drugs, Dr. Griebeler noted.
And while doctors are often just as excited as patients about the potential for treatment, many find themselves under-equipped when it comes to obesity. “More physicians are ... recognizing the value in treating this, and some are realizing, “Oh gosh, I never learned how to do this,” said Dr. Gudzune.
Information Patients Have Been Waiting For
Medical training has traditionally devoted minimal, if any, curriculum to obesity and metabolism. “To be honest, we didn’t really cover this at all in my training,” said Nina Paddu, MD, obesity medicine specialist at Maimonides Medical Center in New York City who finished her training only 2 years ago. “The guidance even in residency was ‘let’s send them to nutrition’ and ‘recommend exercising.’ ”
In addition to the medical education gap, until recently there was a “paucity of robust evidence,” Dr. Srivastava said. Leaders in the field wanted to establish standards and guidelines, but there wasn’t enough strong evidence on obesity and its treatments to build them, she said.
Only in the last 5 years or so has the evidence-based understanding of obesity’s pathophysiology truly accelerated: The brain’s driving roles, its interplay with hormones, and its interactions with other diseases. “We are just at the cusp of understanding all the different factors,” Dr. Gudzune said.
But already endocrinologists, surgeons, fertility specialists, gynecologists, and oncologists, to name a few, see the critical overlap with their own field. “Conditions were once suspected of being intertwined [with obesity], and now we have data to connect them,” Dr. Srivastava said. For example, there’s now data connecting semaglutide to a 20% reduction in cardiovascular events for people with obesity. That’s a game changer for multiple specialties, she told this news organization.
Getting Trained in Obesity Management
The recent uptick in obesity insights and increased patient need has doctors from every career stage seeking additional training.
The ABOM offers two board certification pathways: 60 hours of CME credits or a 12-month fellowship. Both paths require doctors to pass the board’s exam.
Many doctors incorporate the training into their existing practice. The CME credit pathway, especially, is designed to help get doctors up to speed without requiring them to upend their lives for a fellowship.
Dr. Srivastava said that the fellowship is more consuming and immersive. While it’s often younger doctors just out of training who apply to fellowship, every year, “I’m astonished at the number of talented physicians with clinical and research experience who want to immerse themselves in a fellowship experience.”
Some doctors return to their previous specialties after fellowship. But many will go on to take obesity medicine–specific roles or set aside clinic hours for obesity medicine. Their credentials are “really attractive to institutions, especially those looking to open up obesity medicine or weight management programs,” said Dr. Srivastava.
Dr. Paddu, who finished her obesity medicine fellowship this year, said there are a variety of obesity medicine jobs to choose from — far different from Dr. Srivastava’s job search 15 years ago. Dr. Paddu’s new role combines 2 days of primary care with 2 days devoted to obesity medicine and 1 day each week set aside for administrative work so she can build up the hospital’s new metabolic health clinic.
Still Not Enough Obesity Specialists
As with all things, rapid growth requires careful oversight. “Part of the responsibility of the board is to think critically of how the field is growing” and conduct ongoing monitoring, Dr. Gudzune said.
This is also why the board’s credentials are time-limited and must be recertified, Dr. Johnson-Rabbett added.
But even with the rise in certified doctors and obesity medicine positions, the 8263 doctors certified by ABOM are only a tiny fraction of US physicians. As a result, there’s genuine likelihood that many patients seeking GLP-1s or other obesity treatment don’t yet have access to the holistic care they need. Plus, doctors may still not have obesity expertise within their networks.
“The field has grown rapidly, but it’s still such a small field relative to the patient need,” said Dr. Gudzune.
A version of this article appeared on Medscape.com.
Gitanjali Srivastava, MD, professor of medicine, pediatrics, and surgery, and medical director of obesity medicine at Vanderbilt University School of Medicine in Nashville, Tennessee, was nearly 10 years into practicing pediatric medicine when she graduated from the obesity medicine fellowship at Massachusetts General Hospital in Boston in 2013. “We were the very first sort of fellows to speak of then; there were no standards or curriculum,” she said.
Obesity was already epidemic, but stigma and bias were still pervasive in the medical community and within the public. After graduating, Dr. Srivastava spent months vying for a position with hospital CEOs. She traveled across the country explaining the specialty and its value, going into detail about the budget, business model, space requirement, and revenue potential of obesity medicine.
Today marks a very different era.
Obesity medicine is exploding. Patients are spilling into doctors’ offices looking for obesity treatment. Healthcare systems are seeking out obesity specialists and building metabolic health centers. Next month, another 2115 doctors from primary care, surgery, orthopedics, pediatrics, fertility, endocrinology, and beyond will sit for the 2024 exam. The once niche specialty is quickly becoming intertwined with most of modern medicine.
The Need to Treat
It’s no mystery that the rapid expansion of obesity medicine coincides with the US Food and Drug Administration’s approval of GLP-1 injections. The drugs’ radical weight loss properties have captured headlines and driven up patient demand. Meanwhile, doctors are finally able to offer effective treatment for a disease that affects 40% of US adults.
“We are finally treating it as a chronic disease, not as a lifestyle,” said Marcio Griebeler, MD, director of the obesity medicine fellowship at the Cleveland Clinic. And “I think it’s fulfilling for physicians,” he said.
For so long, the advice for obesity was about lifestyle. Move more, eat less, and harness willpower, “which really is a fallacy,” said Kimberly Gudzune, MD, MPH, an obesity medicine specialist and chief medical officer for the American Board of Obesity Medicine (ABOM) Foundation. For people with obesity, “your brain is operating differently,” she said. “Your body really is set up to work against you.”
Brianna Johnson-Rabbett, MD, medical director of the ABOM, told this news organization that with the advent of GLP-1s, “there’s a clearer recognition that obesity is a disease that needs to be treated like other diseases.” Some of that is thanks to clinical trial data showing that just as with other diseases such as high blood pressure or diabetes, obesity can be treated with medication and it resurges when the medication is stopped, she said.
Doctors don’t have to go looking for patients with obesity, dr. Griebeler added. Now that treatment options exist, they’re showing up in droves at the doctor’s office — all the doctors’ offices. In primary care, endocrinology, surgery, pediatrics — a wide variety of doctors are being asked about obesity drugs, Dr. Griebeler noted.
And while doctors are often just as excited as patients about the potential for treatment, many find themselves under-equipped when it comes to obesity. “More physicians are ... recognizing the value in treating this, and some are realizing, “Oh gosh, I never learned how to do this,” said Dr. Gudzune.
Information Patients Have Been Waiting For
Medical training has traditionally devoted minimal, if any, curriculum to obesity and metabolism. “To be honest, we didn’t really cover this at all in my training,” said Nina Paddu, MD, obesity medicine specialist at Maimonides Medical Center in New York City who finished her training only 2 years ago. “The guidance even in residency was ‘let’s send them to nutrition’ and ‘recommend exercising.’ ”
In addition to the medical education gap, until recently there was a “paucity of robust evidence,” Dr. Srivastava said. Leaders in the field wanted to establish standards and guidelines, but there wasn’t enough strong evidence on obesity and its treatments to build them, she said.
Only in the last 5 years or so has the evidence-based understanding of obesity’s pathophysiology truly accelerated: The brain’s driving roles, its interplay with hormones, and its interactions with other diseases. “We are just at the cusp of understanding all the different factors,” Dr. Gudzune said.
But already endocrinologists, surgeons, fertility specialists, gynecologists, and oncologists, to name a few, see the critical overlap with their own field. “Conditions were once suspected of being intertwined [with obesity], and now we have data to connect them,” Dr. Srivastava said. For example, there’s now data connecting semaglutide to a 20% reduction in cardiovascular events for people with obesity. That’s a game changer for multiple specialties, she told this news organization.
Getting Trained in Obesity Management
The recent uptick in obesity insights and increased patient need has doctors from every career stage seeking additional training.
The ABOM offers two board certification pathways: 60 hours of CME credits or a 12-month fellowship. Both paths require doctors to pass the board’s exam.
Many doctors incorporate the training into their existing practice. The CME credit pathway, especially, is designed to help get doctors up to speed without requiring them to upend their lives for a fellowship.
Dr. Srivastava said that the fellowship is more consuming and immersive. While it’s often younger doctors just out of training who apply to fellowship, every year, “I’m astonished at the number of talented physicians with clinical and research experience who want to immerse themselves in a fellowship experience.”
Some doctors return to their previous specialties after fellowship. But many will go on to take obesity medicine–specific roles or set aside clinic hours for obesity medicine. Their credentials are “really attractive to institutions, especially those looking to open up obesity medicine or weight management programs,” said Dr. Srivastava.
Dr. Paddu, who finished her obesity medicine fellowship this year, said there are a variety of obesity medicine jobs to choose from — far different from Dr. Srivastava’s job search 15 years ago. Dr. Paddu’s new role combines 2 days of primary care with 2 days devoted to obesity medicine and 1 day each week set aside for administrative work so she can build up the hospital’s new metabolic health clinic.
Still Not Enough Obesity Specialists
As with all things, rapid growth requires careful oversight. “Part of the responsibility of the board is to think critically of how the field is growing” and conduct ongoing monitoring, Dr. Gudzune said.
This is also why the board’s credentials are time-limited and must be recertified, Dr. Johnson-Rabbett added.
But even with the rise in certified doctors and obesity medicine positions, the 8263 doctors certified by ABOM are only a tiny fraction of US physicians. As a result, there’s genuine likelihood that many patients seeking GLP-1s or other obesity treatment don’t yet have access to the holistic care they need. Plus, doctors may still not have obesity expertise within their networks.
“The field has grown rapidly, but it’s still such a small field relative to the patient need,” said Dr. Gudzune.
A version of this article appeared on Medscape.com.
Popular Weight Loss Drugs Now for Patients With Cancer?
Demand for new weight loss drugs has surged over the past few years.
Led by the antiobesity drugs semaglutide (Wegovy) and tirzepatide (Zepbound), these popular medications — more commonly known as glucagon-like peptide 1 (GLP-1) agonists — have become game changers for shedding excess pounds.
Aside from obesity indications, both drugs have been approved to treat type 2 diabetes under different brand names and have a growing list of other potential benefits, such as reducing inflammation and depression.
While there’s limited data to support the use of GLP-1 agonists for weight loss in cancer, some oncologists have begun carefully integrating the antiobesity agents into care and studying their effects in this patient population.
The reason: Research suggests that obesity can reduce the effectiveness of cancer therapies, especially in patients with breast cancer, and can increase the risk for treatment-related side effects.
The idea is that managing patients’ weight will improve their cancer outcomes, explained Lajos Pusztai, MD, PhD, a breast cancer specialist and professor of medicine at Yale School of Medicine in New Haven, Connecticut.
Although Dr. Pusztai and his oncology peers at Yale don’t yet use GPL-1 agonists, Neil Iyengar, MD, and colleagues have begun doing so to help some patients with breast cancer manage their weight. Dr. Iyengar estimates that a few hundred — almost 40% — of his patients are on the antiobesity drugs.
“For a patient who has really tried to reduce their weight and who is in the obese range, that’s where I think the use of these medications can be considered,” said Dr. Iyengar, a breast cancer oncologist at Memorial Sloan Kettering Cancer Center in New York City.
Why GLP-1s in Cancer?
GLP-1 is a hormone that the small intestine releases after eating. GLP-1 agonists work by mimicking GLP-1 to trigger the release of insulin and reduce the production of glucagon — two processes that help regulate blood sugar.
These agents, such as Wegovy (or Ozempic when prescribed for diabetes), also slow gastric emptying and can make people feel fuller longer.
Zebound (or Mounjaro for type 2 diabetes) is considered a dual GLP-1 and glucose-dependent insulinotropic polypeptide agonist, which may enhance its weight loss benefits.
In practice, however, these drugs can increase nausea and vomiting from chemotherapy, so Dr. Iyengar typically has patients use them afterwards, during maintenance treatment.
Oncologists don’t prescribe the drugs themselves but instead refer patients to endocrinologists or weight management centers that then write the prescriptions. Taking these drugs involves weekly subcutaneous injections patients can administer themselves.
Endocrinologist Emily Gallagher, MD, PhD, of Mount Sinai Hospital in New York City, estimates she has prescribed the antiobesity drugs to a few hundred patients with cancer and, like Dr. Iyengar, uses the drugs during maintenance treatment with hormone therapy for breast cancer. She also has used these agents in patients with prostate and endometrial cancers and has found the drugs can help counter steroid weight gain in multiple myeloma.
But, to date, the evidence for using GPL-1 agonists in cancer remains limited and the practice has not yet become widespread.
Research largely comes down to a few small retrospective studies in patients with breast cancer receiving aromatase inhibitors. Although no safety issues have emerged so far, these initial reports suggest that the drugs lead to significantly less weight loss in patients with cancer compared to the general population.
Dr. Iyengar led one recent study, presented at the 2024 annual meeting of the American Society of Clinical Oncology, in which he and his team assessed outcomes in 75 women with breast cancer who received a GLP-1 agonist. Almost 80% of patients had diabetes, and 60% received hormone therapy, most commonly an aromatase inhibitor. Patients’ median body mass index (BMI) at baseline was 34 kg/m2 (range, 23-50 kg/m2).
From baseline, patients lost 6.2 kg, on average, or about 5% of their total body weight, 12 months after initiating GLP-1 therapy.
In contrast, phase 3 trials show much higher mean weight loss — about two times — in patients without cancer.
Another recent study also reported modest weight loss results in patients with breast cancer undergoing endocrine therapy. The researchers reported that, at 12 months, Wegovy led to 4.34% reduction in BMI, compared with a 14% change reported in the general population. Zebound, however, was associated with a 2.31% BMI increase overall — though some patients did experience a decrease — compared with a 15% reduction in the general population.
“These findings indicate a substantially reduced weight loss efficacy in breast cancer patients on endocrine therapy compared to the general population,” the authors concluded.
It’s unclear why the drugs appear to not work as well in patients with cancer. It’s possible that hormone therapy or metabolic changes interfere with their effectiveness, given that some cancer therapies lead to weight gain. Steroids and hormone therapies, for instance, often increase appetite, and some treatments can slow patients’ metabolism or lead to fatigue, which can make it harder to exercise.
Patients with cancer may need a higher dose of GLP-1 agonists to achieve similar weight loss to the general population, Dr. Iyengar noted.
However, Dr. Gallagher said, in her own experience, she hasn’t found the drugs to be less effective in patients with cancer, especially the newer agents, like Wegovy and Zepbound.
As for safety, Wegovy and Zepbound both carry a black box warning for thyroid C-cell tumors, including medullary thyroid carcinoma. (Recent research, however, has found that GLP-1 agonists do not increase thyroid cancer risk).
These antiobesity agents are also contraindicated in patients with a personal or family history of medullary thyroid carcinoma and in patients who have multiple endocrine neoplasia syndrome type 2, which is associated with medullary thyroid carcinoma.
Dr. Gallagher hasn’t seen any secondary tumors — thyroid or otherwise — in her patients with cancer, but she follows the labeling contraindications. Dr. Iyengar also noted that more recent and larger data sets have shown no impact on this risk, which may not actually exist, he said
Dr. Gallagher remains cautious about using GPL-1 agonists in patients who have had bariatric surgery because these agents can compound the slower gastric emptying and intestinal transit from surgery, potentially leading to gastrointestinal obstructions.
Looking ahead, GPL-1 manufacturers are interested in adding cancer indications to the drug labeling. Both Dr. Iyengar and Dr. Gallagher said their institutions are in talks with companies to participate in large, multicenter, global phase 3 trials.
Dr. Iyengar welcomes the efforts, not only to test the effectiveness of GPL-1 agonists in oncology but also to “nail down” their safety in cancer.
“I don’t think that there’s mechanistically anything that’s particularly worrisome,” and current observations suggest that these drugs are likely to be safe, Dr. Iyengar said. Even so, “GLP-1 agonists do a lot of things that we don’t fully understand yet.”
The bigger challenge, Dr. Iyengar noted, is that companies will have to show a sizable benefit to using these drugs in patients with cancer to get the Food and Drug Administration’s approval. And to move the needle on cancer-specific outcomes, these antiobesity drugs will need to demonstrate significant, durable weight loss in patients with cancer.
But if these drugs can do that, “I think it’s going to be one of the biggest advances in medicine and oncology given the obesity and cancer epidemic,” Dr. Iyengar said.
Dr. Iyengar has adviser and/or researcher ties with companies that make or are developing GPL-1 agonists, including AstraZeneca, Novartis, Gilead, and Pfizer. Dr. Gallagher is a consultant for Novartis, Flare Therapeutics, Reactive Biosciences, and Seagen.
A version of this article first appeared on Medscape.com.
Demand for new weight loss drugs has surged over the past few years.
Led by the antiobesity drugs semaglutide (Wegovy) and tirzepatide (Zepbound), these popular medications — more commonly known as glucagon-like peptide 1 (GLP-1) agonists — have become game changers for shedding excess pounds.
Aside from obesity indications, both drugs have been approved to treat type 2 diabetes under different brand names and have a growing list of other potential benefits, such as reducing inflammation and depression.
While there’s limited data to support the use of GLP-1 agonists for weight loss in cancer, some oncologists have begun carefully integrating the antiobesity agents into care and studying their effects in this patient population.
The reason: Research suggests that obesity can reduce the effectiveness of cancer therapies, especially in patients with breast cancer, and can increase the risk for treatment-related side effects.
The idea is that managing patients’ weight will improve their cancer outcomes, explained Lajos Pusztai, MD, PhD, a breast cancer specialist and professor of medicine at Yale School of Medicine in New Haven, Connecticut.
Although Dr. Pusztai and his oncology peers at Yale don’t yet use GPL-1 agonists, Neil Iyengar, MD, and colleagues have begun doing so to help some patients with breast cancer manage their weight. Dr. Iyengar estimates that a few hundred — almost 40% — of his patients are on the antiobesity drugs.
“For a patient who has really tried to reduce their weight and who is in the obese range, that’s where I think the use of these medications can be considered,” said Dr. Iyengar, a breast cancer oncologist at Memorial Sloan Kettering Cancer Center in New York City.
Why GLP-1s in Cancer?
GLP-1 is a hormone that the small intestine releases after eating. GLP-1 agonists work by mimicking GLP-1 to trigger the release of insulin and reduce the production of glucagon — two processes that help regulate blood sugar.
These agents, such as Wegovy (or Ozempic when prescribed for diabetes), also slow gastric emptying and can make people feel fuller longer.
Zebound (or Mounjaro for type 2 diabetes) is considered a dual GLP-1 and glucose-dependent insulinotropic polypeptide agonist, which may enhance its weight loss benefits.
In practice, however, these drugs can increase nausea and vomiting from chemotherapy, so Dr. Iyengar typically has patients use them afterwards, during maintenance treatment.
Oncologists don’t prescribe the drugs themselves but instead refer patients to endocrinologists or weight management centers that then write the prescriptions. Taking these drugs involves weekly subcutaneous injections patients can administer themselves.
Endocrinologist Emily Gallagher, MD, PhD, of Mount Sinai Hospital in New York City, estimates she has prescribed the antiobesity drugs to a few hundred patients with cancer and, like Dr. Iyengar, uses the drugs during maintenance treatment with hormone therapy for breast cancer. She also has used these agents in patients with prostate and endometrial cancers and has found the drugs can help counter steroid weight gain in multiple myeloma.
But, to date, the evidence for using GPL-1 agonists in cancer remains limited and the practice has not yet become widespread.
Research largely comes down to a few small retrospective studies in patients with breast cancer receiving aromatase inhibitors. Although no safety issues have emerged so far, these initial reports suggest that the drugs lead to significantly less weight loss in patients with cancer compared to the general population.
Dr. Iyengar led one recent study, presented at the 2024 annual meeting of the American Society of Clinical Oncology, in which he and his team assessed outcomes in 75 women with breast cancer who received a GLP-1 agonist. Almost 80% of patients had diabetes, and 60% received hormone therapy, most commonly an aromatase inhibitor. Patients’ median body mass index (BMI) at baseline was 34 kg/m2 (range, 23-50 kg/m2).
From baseline, patients lost 6.2 kg, on average, or about 5% of their total body weight, 12 months after initiating GLP-1 therapy.
In contrast, phase 3 trials show much higher mean weight loss — about two times — in patients without cancer.
Another recent study also reported modest weight loss results in patients with breast cancer undergoing endocrine therapy. The researchers reported that, at 12 months, Wegovy led to 4.34% reduction in BMI, compared with a 14% change reported in the general population. Zebound, however, was associated with a 2.31% BMI increase overall — though some patients did experience a decrease — compared with a 15% reduction in the general population.
“These findings indicate a substantially reduced weight loss efficacy in breast cancer patients on endocrine therapy compared to the general population,” the authors concluded.
It’s unclear why the drugs appear to not work as well in patients with cancer. It’s possible that hormone therapy or metabolic changes interfere with their effectiveness, given that some cancer therapies lead to weight gain. Steroids and hormone therapies, for instance, often increase appetite, and some treatments can slow patients’ metabolism or lead to fatigue, which can make it harder to exercise.
Patients with cancer may need a higher dose of GLP-1 agonists to achieve similar weight loss to the general population, Dr. Iyengar noted.
However, Dr. Gallagher said, in her own experience, she hasn’t found the drugs to be less effective in patients with cancer, especially the newer agents, like Wegovy and Zepbound.
As for safety, Wegovy and Zepbound both carry a black box warning for thyroid C-cell tumors, including medullary thyroid carcinoma. (Recent research, however, has found that GLP-1 agonists do not increase thyroid cancer risk).
These antiobesity agents are also contraindicated in patients with a personal or family history of medullary thyroid carcinoma and in patients who have multiple endocrine neoplasia syndrome type 2, which is associated with medullary thyroid carcinoma.
Dr. Gallagher hasn’t seen any secondary tumors — thyroid or otherwise — in her patients with cancer, but she follows the labeling contraindications. Dr. Iyengar also noted that more recent and larger data sets have shown no impact on this risk, which may not actually exist, he said
Dr. Gallagher remains cautious about using GPL-1 agonists in patients who have had bariatric surgery because these agents can compound the slower gastric emptying and intestinal transit from surgery, potentially leading to gastrointestinal obstructions.
Looking ahead, GPL-1 manufacturers are interested in adding cancer indications to the drug labeling. Both Dr. Iyengar and Dr. Gallagher said their institutions are in talks with companies to participate in large, multicenter, global phase 3 trials.
Dr. Iyengar welcomes the efforts, not only to test the effectiveness of GPL-1 agonists in oncology but also to “nail down” their safety in cancer.
“I don’t think that there’s mechanistically anything that’s particularly worrisome,” and current observations suggest that these drugs are likely to be safe, Dr. Iyengar said. Even so, “GLP-1 agonists do a lot of things that we don’t fully understand yet.”
The bigger challenge, Dr. Iyengar noted, is that companies will have to show a sizable benefit to using these drugs in patients with cancer to get the Food and Drug Administration’s approval. And to move the needle on cancer-specific outcomes, these antiobesity drugs will need to demonstrate significant, durable weight loss in patients with cancer.
But if these drugs can do that, “I think it’s going to be one of the biggest advances in medicine and oncology given the obesity and cancer epidemic,” Dr. Iyengar said.
Dr. Iyengar has adviser and/or researcher ties with companies that make or are developing GPL-1 agonists, including AstraZeneca, Novartis, Gilead, and Pfizer. Dr. Gallagher is a consultant for Novartis, Flare Therapeutics, Reactive Biosciences, and Seagen.
A version of this article first appeared on Medscape.com.
Demand for new weight loss drugs has surged over the past few years.
Led by the antiobesity drugs semaglutide (Wegovy) and tirzepatide (Zepbound), these popular medications — more commonly known as glucagon-like peptide 1 (GLP-1) agonists — have become game changers for shedding excess pounds.
Aside from obesity indications, both drugs have been approved to treat type 2 diabetes under different brand names and have a growing list of other potential benefits, such as reducing inflammation and depression.
While there’s limited data to support the use of GLP-1 agonists for weight loss in cancer, some oncologists have begun carefully integrating the antiobesity agents into care and studying their effects in this patient population.
The reason: Research suggests that obesity can reduce the effectiveness of cancer therapies, especially in patients with breast cancer, and can increase the risk for treatment-related side effects.
The idea is that managing patients’ weight will improve their cancer outcomes, explained Lajos Pusztai, MD, PhD, a breast cancer specialist and professor of medicine at Yale School of Medicine in New Haven, Connecticut.
Although Dr. Pusztai and his oncology peers at Yale don’t yet use GPL-1 agonists, Neil Iyengar, MD, and colleagues have begun doing so to help some patients with breast cancer manage their weight. Dr. Iyengar estimates that a few hundred — almost 40% — of his patients are on the antiobesity drugs.
“For a patient who has really tried to reduce their weight and who is in the obese range, that’s where I think the use of these medications can be considered,” said Dr. Iyengar, a breast cancer oncologist at Memorial Sloan Kettering Cancer Center in New York City.
Why GLP-1s in Cancer?
GLP-1 is a hormone that the small intestine releases after eating. GLP-1 agonists work by mimicking GLP-1 to trigger the release of insulin and reduce the production of glucagon — two processes that help regulate blood sugar.
These agents, such as Wegovy (or Ozempic when prescribed for diabetes), also slow gastric emptying and can make people feel fuller longer.
Zebound (or Mounjaro for type 2 diabetes) is considered a dual GLP-1 and glucose-dependent insulinotropic polypeptide agonist, which may enhance its weight loss benefits.
In practice, however, these drugs can increase nausea and vomiting from chemotherapy, so Dr. Iyengar typically has patients use them afterwards, during maintenance treatment.
Oncologists don’t prescribe the drugs themselves but instead refer patients to endocrinologists or weight management centers that then write the prescriptions. Taking these drugs involves weekly subcutaneous injections patients can administer themselves.
Endocrinologist Emily Gallagher, MD, PhD, of Mount Sinai Hospital in New York City, estimates she has prescribed the antiobesity drugs to a few hundred patients with cancer and, like Dr. Iyengar, uses the drugs during maintenance treatment with hormone therapy for breast cancer. She also has used these agents in patients with prostate and endometrial cancers and has found the drugs can help counter steroid weight gain in multiple myeloma.
But, to date, the evidence for using GPL-1 agonists in cancer remains limited and the practice has not yet become widespread.
Research largely comes down to a few small retrospective studies in patients with breast cancer receiving aromatase inhibitors. Although no safety issues have emerged so far, these initial reports suggest that the drugs lead to significantly less weight loss in patients with cancer compared to the general population.
Dr. Iyengar led one recent study, presented at the 2024 annual meeting of the American Society of Clinical Oncology, in which he and his team assessed outcomes in 75 women with breast cancer who received a GLP-1 agonist. Almost 80% of patients had diabetes, and 60% received hormone therapy, most commonly an aromatase inhibitor. Patients’ median body mass index (BMI) at baseline was 34 kg/m2 (range, 23-50 kg/m2).
From baseline, patients lost 6.2 kg, on average, or about 5% of their total body weight, 12 months after initiating GLP-1 therapy.
In contrast, phase 3 trials show much higher mean weight loss — about two times — in patients without cancer.
Another recent study also reported modest weight loss results in patients with breast cancer undergoing endocrine therapy. The researchers reported that, at 12 months, Wegovy led to 4.34% reduction in BMI, compared with a 14% change reported in the general population. Zebound, however, was associated with a 2.31% BMI increase overall — though some patients did experience a decrease — compared with a 15% reduction in the general population.
“These findings indicate a substantially reduced weight loss efficacy in breast cancer patients on endocrine therapy compared to the general population,” the authors concluded.
It’s unclear why the drugs appear to not work as well in patients with cancer. It’s possible that hormone therapy or metabolic changes interfere with their effectiveness, given that some cancer therapies lead to weight gain. Steroids and hormone therapies, for instance, often increase appetite, and some treatments can slow patients’ metabolism or lead to fatigue, which can make it harder to exercise.
Patients with cancer may need a higher dose of GLP-1 agonists to achieve similar weight loss to the general population, Dr. Iyengar noted.
However, Dr. Gallagher said, in her own experience, she hasn’t found the drugs to be less effective in patients with cancer, especially the newer agents, like Wegovy and Zepbound.
As for safety, Wegovy and Zepbound both carry a black box warning for thyroid C-cell tumors, including medullary thyroid carcinoma. (Recent research, however, has found that GLP-1 agonists do not increase thyroid cancer risk).
These antiobesity agents are also contraindicated in patients with a personal or family history of medullary thyroid carcinoma and in patients who have multiple endocrine neoplasia syndrome type 2, which is associated with medullary thyroid carcinoma.
Dr. Gallagher hasn’t seen any secondary tumors — thyroid or otherwise — in her patients with cancer, but she follows the labeling contraindications. Dr. Iyengar also noted that more recent and larger data sets have shown no impact on this risk, which may not actually exist, he said
Dr. Gallagher remains cautious about using GPL-1 agonists in patients who have had bariatric surgery because these agents can compound the slower gastric emptying and intestinal transit from surgery, potentially leading to gastrointestinal obstructions.
Looking ahead, GPL-1 manufacturers are interested in adding cancer indications to the drug labeling. Both Dr. Iyengar and Dr. Gallagher said their institutions are in talks with companies to participate in large, multicenter, global phase 3 trials.
Dr. Iyengar welcomes the efforts, not only to test the effectiveness of GPL-1 agonists in oncology but also to “nail down” their safety in cancer.
“I don’t think that there’s mechanistically anything that’s particularly worrisome,” and current observations suggest that these drugs are likely to be safe, Dr. Iyengar said. Even so, “GLP-1 agonists do a lot of things that we don’t fully understand yet.”
The bigger challenge, Dr. Iyengar noted, is that companies will have to show a sizable benefit to using these drugs in patients with cancer to get the Food and Drug Administration’s approval. And to move the needle on cancer-specific outcomes, these antiobesity drugs will need to demonstrate significant, durable weight loss in patients with cancer.
But if these drugs can do that, “I think it’s going to be one of the biggest advances in medicine and oncology given the obesity and cancer epidemic,” Dr. Iyengar said.
Dr. Iyengar has adviser and/or researcher ties with companies that make or are developing GPL-1 agonists, including AstraZeneca, Novartis, Gilead, and Pfizer. Dr. Gallagher is a consultant for Novartis, Flare Therapeutics, Reactive Biosciences, and Seagen.
A version of this article first appeared on Medscape.com.
Five Essential Nutrients for Patients on GLP-1s
Fatigue, nausea, acid reflux, muscle loss, and the dreaded “Ozempic face” are side effects from using glucagon-like peptide 1 (GLP-1) receptor agonists (RAs) such as semaglutide or the dual glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 RA tirzepatide to control blood sugar and promote weight loss.
But what I’ve learned from working with hundreds of patients on these medications, and others, is that most (if not all) of these side effects can be minimized by ensuring proper nutrition.
Setting patients up for success requires dietary education and counseling, along with regular monitoring to determine any nutritional deficiencies. Although adequate intake of all the macro and micronutrients is obviously important,
Protein
My patients are probably sick of hearing me talk about protein, but without the constant reinforcement, many of them wouldn’t consume enough of this macronutrient to maintain their baseline lean body mass. The recommended dietary allowance (RDA) for protein (0.8 g/kg bodyweight) doesn’t cut it, especially for older, obese patients, who need closer to 1.0-1.2 g/kg bodyweight to maintain their muscle mass. For example, for a 250-lb patient, I would recommend 114-136 g protein per day. This is equivalent to roughly 15 oz of cooked animal protein. It’s important to note, though, that individuals with kidney disease must limit their protein intake to 0.6-0.8 g/kg bodyweight per day, to avoid overtaxing their kidneys. In this situation, the benefit of increased protein intake does not outweigh the risk of harming the kidneys.
It’s often challenging for patients with suppressed appetites to even think about eating a large hunk of meat or fish, let alone consume it. Plus, eating more than 3-4 oz of protein in one meal can make some patients extremely uncomfortable, owing to the medication’s effect on gastric emptying. This means that daily protein intake must be spread out over multiple mini-meals.
For patients who need more than 100 g of protein per day, protein powders and premade protein shakes can provide 20-30 g protein to fill in the gaps. Although I always try to promote food first, protein supplements have been game changers for my patients, especially those who find solid food less appealing on the medication, or those who avoid animal protein.
Clinicians should have their patients monitor changes in their lean body mass using a dual-energy x-ray absorptiometry scan or a bioelectrical impedance scale; this can be a helpful tool in assessing whether protein intake is sufficient.
Fiber
Even my most knowledgeable and compliant patients will experience some constipation. Generally speaking, when you eat less, you will have fewer bowel movements. Combine that with delayed gastric emptying and reduced fiber intake, and you have a perfect storm. Many patients are simply not able to get in the recommended 25-35 g fiber per day through food, because fibrous foods are filling. If they are prioritizing the protein in their meal, they will not have enough room for all the vegetables on their plate.
To ensure that patients are getting sufficient fiber, clinicians should push consumption of certain vegetables and fruits, such as carrots, broccoli, Brussels sprouts, raspberries, blackberries, and apples, as well as beans and legumes. (Salads are great, but greens like spinach are not as fibrous as one might think.) If the fruit and veggie intake isn’t up to par, a fiber supplement such as psyllium husk can provide an effective boost.
Vitamin B12
Use of these medications is associated with a reduction in vitamin B12 levels, in part because delayed gastric emptying may affect B12 absorption. Low dietary intake of B12 while on the medications can also be to blame, though. The best food sources are animal proteins, so if possible, patients should prioritize having fish, lean meat, eggs, and dairy daily.
Vegetarians and vegans, who are at an increased risk for deficiency, can incorporate nutritional yeast, an excellent source of vitamin B12, into their daily routine. It is beneficial for patients to get blood work periodically to check on B12 status, because insufficient B12 can contribute to the fatigue patients experience while on the medication.
Calcium
Individuals should have calcium on their radar, because weight loss is associated with a decrease in bone mineral density. Adequate intake of the mineral is crucial for optimal bone health, particularly among postmenopausal women and those who are at risk of developing osteoporosis. The RDA for calcium is 1000-1200 mg/d, which an estimated 50% of obese individuals do not take in.
Although dairy products are well-known for being rich in calcium, there are other great sources. Dark green leafy vegetables, such as cooked collard greens and spinach, provide nearly 300 mg per cup. Tofu and sardines are also calcium powerhouses. Despite the plethora of calcium-rich foods, however, some patients may need a calcium supplement.
Vitamin D
Vitamin D deficiency or insufficiency is common among individuals with obesity, so even before these patients start the medications, supplementation may be warranted. The vitamin’s role in promoting calcium absorption, as well as in bone remodeling, make adequate intake essential for patients experiencing significant weight loss.
Clinicians should emphasize regular consumption of fatty fish, such as salmon, as well as eggs, mushrooms, and vitamin D–fortified milks. But unfortunately, that’s where the list of vitamin D–rich foods ends, so taking a vitamin D supplement will be necessary for many patients.
Regularly monitoring patients on GLP-1 RAs through blood work to check vitamin levels and body composition analysis can be helpful in assessing nutritional status while losing weight. Clinicians can also encourage their patients to work with a registered dietitian who is familiar with these medications, so they can develop optimal eating habits throughout their health journey.
Ms. Hanks, a registered dietitian in New York City, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Fatigue, nausea, acid reflux, muscle loss, and the dreaded “Ozempic face” are side effects from using glucagon-like peptide 1 (GLP-1) receptor agonists (RAs) such as semaglutide or the dual glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 RA tirzepatide to control blood sugar and promote weight loss.
But what I’ve learned from working with hundreds of patients on these medications, and others, is that most (if not all) of these side effects can be minimized by ensuring proper nutrition.
Setting patients up for success requires dietary education and counseling, along with regular monitoring to determine any nutritional deficiencies. Although adequate intake of all the macro and micronutrients is obviously important,
Protein
My patients are probably sick of hearing me talk about protein, but without the constant reinforcement, many of them wouldn’t consume enough of this macronutrient to maintain their baseline lean body mass. The recommended dietary allowance (RDA) for protein (0.8 g/kg bodyweight) doesn’t cut it, especially for older, obese patients, who need closer to 1.0-1.2 g/kg bodyweight to maintain their muscle mass. For example, for a 250-lb patient, I would recommend 114-136 g protein per day. This is equivalent to roughly 15 oz of cooked animal protein. It’s important to note, though, that individuals with kidney disease must limit their protein intake to 0.6-0.8 g/kg bodyweight per day, to avoid overtaxing their kidneys. In this situation, the benefit of increased protein intake does not outweigh the risk of harming the kidneys.
It’s often challenging for patients with suppressed appetites to even think about eating a large hunk of meat or fish, let alone consume it. Plus, eating more than 3-4 oz of protein in one meal can make some patients extremely uncomfortable, owing to the medication’s effect on gastric emptying. This means that daily protein intake must be spread out over multiple mini-meals.
For patients who need more than 100 g of protein per day, protein powders and premade protein shakes can provide 20-30 g protein to fill in the gaps. Although I always try to promote food first, protein supplements have been game changers for my patients, especially those who find solid food less appealing on the medication, or those who avoid animal protein.
Clinicians should have their patients monitor changes in their lean body mass using a dual-energy x-ray absorptiometry scan or a bioelectrical impedance scale; this can be a helpful tool in assessing whether protein intake is sufficient.
Fiber
Even my most knowledgeable and compliant patients will experience some constipation. Generally speaking, when you eat less, you will have fewer bowel movements. Combine that with delayed gastric emptying and reduced fiber intake, and you have a perfect storm. Many patients are simply not able to get in the recommended 25-35 g fiber per day through food, because fibrous foods are filling. If they are prioritizing the protein in their meal, they will not have enough room for all the vegetables on their plate.
To ensure that patients are getting sufficient fiber, clinicians should push consumption of certain vegetables and fruits, such as carrots, broccoli, Brussels sprouts, raspberries, blackberries, and apples, as well as beans and legumes. (Salads are great, but greens like spinach are not as fibrous as one might think.) If the fruit and veggie intake isn’t up to par, a fiber supplement such as psyllium husk can provide an effective boost.
Vitamin B12
Use of these medications is associated with a reduction in vitamin B12 levels, in part because delayed gastric emptying may affect B12 absorption. Low dietary intake of B12 while on the medications can also be to blame, though. The best food sources are animal proteins, so if possible, patients should prioritize having fish, lean meat, eggs, and dairy daily.
Vegetarians and vegans, who are at an increased risk for deficiency, can incorporate nutritional yeast, an excellent source of vitamin B12, into their daily routine. It is beneficial for patients to get blood work periodically to check on B12 status, because insufficient B12 can contribute to the fatigue patients experience while on the medication.
Calcium
Individuals should have calcium on their radar, because weight loss is associated with a decrease in bone mineral density. Adequate intake of the mineral is crucial for optimal bone health, particularly among postmenopausal women and those who are at risk of developing osteoporosis. The RDA for calcium is 1000-1200 mg/d, which an estimated 50% of obese individuals do not take in.
Although dairy products are well-known for being rich in calcium, there are other great sources. Dark green leafy vegetables, such as cooked collard greens and spinach, provide nearly 300 mg per cup. Tofu and sardines are also calcium powerhouses. Despite the plethora of calcium-rich foods, however, some patients may need a calcium supplement.
Vitamin D
Vitamin D deficiency or insufficiency is common among individuals with obesity, so even before these patients start the medications, supplementation may be warranted. The vitamin’s role in promoting calcium absorption, as well as in bone remodeling, make adequate intake essential for patients experiencing significant weight loss.
Clinicians should emphasize regular consumption of fatty fish, such as salmon, as well as eggs, mushrooms, and vitamin D–fortified milks. But unfortunately, that’s where the list of vitamin D–rich foods ends, so taking a vitamin D supplement will be necessary for many patients.
Regularly monitoring patients on GLP-1 RAs through blood work to check vitamin levels and body composition analysis can be helpful in assessing nutritional status while losing weight. Clinicians can also encourage their patients to work with a registered dietitian who is familiar with these medications, so they can develop optimal eating habits throughout their health journey.
Ms. Hanks, a registered dietitian in New York City, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Fatigue, nausea, acid reflux, muscle loss, and the dreaded “Ozempic face” are side effects from using glucagon-like peptide 1 (GLP-1) receptor agonists (RAs) such as semaglutide or the dual glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 RA tirzepatide to control blood sugar and promote weight loss.
But what I’ve learned from working with hundreds of patients on these medications, and others, is that most (if not all) of these side effects can be minimized by ensuring proper nutrition.
Setting patients up for success requires dietary education and counseling, along with regular monitoring to determine any nutritional deficiencies. Although adequate intake of all the macro and micronutrients is obviously important,
Protein
My patients are probably sick of hearing me talk about protein, but without the constant reinforcement, many of them wouldn’t consume enough of this macronutrient to maintain their baseline lean body mass. The recommended dietary allowance (RDA) for protein (0.8 g/kg bodyweight) doesn’t cut it, especially for older, obese patients, who need closer to 1.0-1.2 g/kg bodyweight to maintain their muscle mass. For example, for a 250-lb patient, I would recommend 114-136 g protein per day. This is equivalent to roughly 15 oz of cooked animal protein. It’s important to note, though, that individuals with kidney disease must limit their protein intake to 0.6-0.8 g/kg bodyweight per day, to avoid overtaxing their kidneys. In this situation, the benefit of increased protein intake does not outweigh the risk of harming the kidneys.
It’s often challenging for patients with suppressed appetites to even think about eating a large hunk of meat or fish, let alone consume it. Plus, eating more than 3-4 oz of protein in one meal can make some patients extremely uncomfortable, owing to the medication’s effect on gastric emptying. This means that daily protein intake must be spread out over multiple mini-meals.
For patients who need more than 100 g of protein per day, protein powders and premade protein shakes can provide 20-30 g protein to fill in the gaps. Although I always try to promote food first, protein supplements have been game changers for my patients, especially those who find solid food less appealing on the medication, or those who avoid animal protein.
Clinicians should have their patients monitor changes in their lean body mass using a dual-energy x-ray absorptiometry scan or a bioelectrical impedance scale; this can be a helpful tool in assessing whether protein intake is sufficient.
Fiber
Even my most knowledgeable and compliant patients will experience some constipation. Generally speaking, when you eat less, you will have fewer bowel movements. Combine that with delayed gastric emptying and reduced fiber intake, and you have a perfect storm. Many patients are simply not able to get in the recommended 25-35 g fiber per day through food, because fibrous foods are filling. If they are prioritizing the protein in their meal, they will not have enough room for all the vegetables on their plate.
To ensure that patients are getting sufficient fiber, clinicians should push consumption of certain vegetables and fruits, such as carrots, broccoli, Brussels sprouts, raspberries, blackberries, and apples, as well as beans and legumes. (Salads are great, but greens like spinach are not as fibrous as one might think.) If the fruit and veggie intake isn’t up to par, a fiber supplement such as psyllium husk can provide an effective boost.
Vitamin B12
Use of these medications is associated with a reduction in vitamin B12 levels, in part because delayed gastric emptying may affect B12 absorption. Low dietary intake of B12 while on the medications can also be to blame, though. The best food sources are animal proteins, so if possible, patients should prioritize having fish, lean meat, eggs, and dairy daily.
Vegetarians and vegans, who are at an increased risk for deficiency, can incorporate nutritional yeast, an excellent source of vitamin B12, into their daily routine. It is beneficial for patients to get blood work periodically to check on B12 status, because insufficient B12 can contribute to the fatigue patients experience while on the medication.
Calcium
Individuals should have calcium on their radar, because weight loss is associated with a decrease in bone mineral density. Adequate intake of the mineral is crucial for optimal bone health, particularly among postmenopausal women and those who are at risk of developing osteoporosis. The RDA for calcium is 1000-1200 mg/d, which an estimated 50% of obese individuals do not take in.
Although dairy products are well-known for being rich in calcium, there are other great sources. Dark green leafy vegetables, such as cooked collard greens and spinach, provide nearly 300 mg per cup. Tofu and sardines are also calcium powerhouses. Despite the plethora of calcium-rich foods, however, some patients may need a calcium supplement.
Vitamin D
Vitamin D deficiency or insufficiency is common among individuals with obesity, so even before these patients start the medications, supplementation may be warranted. The vitamin’s role in promoting calcium absorption, as well as in bone remodeling, make adequate intake essential for patients experiencing significant weight loss.
Clinicians should emphasize regular consumption of fatty fish, such as salmon, as well as eggs, mushrooms, and vitamin D–fortified milks. But unfortunately, that’s where the list of vitamin D–rich foods ends, so taking a vitamin D supplement will be necessary for many patients.
Regularly monitoring patients on GLP-1 RAs through blood work to check vitamin levels and body composition analysis can be helpful in assessing nutritional status while losing weight. Clinicians can also encourage their patients to work with a registered dietitian who is familiar with these medications, so they can develop optimal eating habits throughout their health journey.
Ms. Hanks, a registered dietitian in New York City, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Walking App Works Only if Users Think It Does
TOPLINE:
Apps designed to increase physical activity may be useful in increasing daily step counts for users who believe the intervention beneficial, but not for those who do not. The app’s effectiveness is notably influenced by how users perceive its utility.
METHODOLOGY:
- Researchers conducted a randomized controlled trial from February 2021 to May 2022 to evaluate the effectiveness of SNapp, an adaptive app designed to promote walking through tailored coaching content.
- Overall, 176 adults (76% women; mean age, 56 years) were randomly assigned to use the app plus tailored coaching content (SNapp group; n = 87) or only the step counter app (control group; n = 89).
- SNapp’s coaching content provided personalized feedback on step counts and recommendations for increasing walking, while also considering individual preferences for behavior change techniques.
- The primary outcome was the daily step count recorded by the app, which was updated on an hourly basis in a database over an intervention period of 12 months.
- Perceptions of ease of use and usefulness were assessed to determine their effect on the effectiveness of the app.
TAKEAWAY:
- Intervention group participants used the app nearly 30% of days, while those using the app alone showed almost identical use.
- The SNapp intervention did not significantly affect the step counts on average over time (B, −202.30; 95% CI, −889.7 to 485.1).
- Perceived usefulness significantly moderated the intervention effect of SNapp (B, 344.38; 90% CI, 40.4-648.3), but perceived ease of use did not (B, 38.60; 90% CI, −276.5 to 353.7).
- Among participants with a high perceived usefulness, the SNapp group had a higher median step count than the control group (median difference, 1260 steps; 90% CI, −3243.7 to 1298.2); however, this difference was not statistically significant.
IN PRACTICE:
“This study shows that perceived usefulness is also an important factor influencing behavioral effects. Hence, it is essential for apps to be perceived as useful to effectively improve users’ activity levels,” the authors wrote.
SOURCE:
The study was led by Anne L. Vos, PhD, of the Amsterdam School of Communication Research at the University of Amsterdam, in the Netherlands. It was published online on September 16, 2024, in the American Journal of Preventive Medicine.
LIMITATIONS:
The study’s recruitment strategy primarily attracted highly educated individuals, limiting generalizability. The app’s accuracy in measuring steps could be improved, as it sometimes underestimated step counts. Researchers also were unable to check if participants read messages from coaches.
DISCLOSURES:
The study was supported by grants from the Dutch Heart Foundation and the Netherlands Organisation for Health Research and Development. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Apps designed to increase physical activity may be useful in increasing daily step counts for users who believe the intervention beneficial, but not for those who do not. The app’s effectiveness is notably influenced by how users perceive its utility.
METHODOLOGY:
- Researchers conducted a randomized controlled trial from February 2021 to May 2022 to evaluate the effectiveness of SNapp, an adaptive app designed to promote walking through tailored coaching content.
- Overall, 176 adults (76% women; mean age, 56 years) were randomly assigned to use the app plus tailored coaching content (SNapp group; n = 87) or only the step counter app (control group; n = 89).
- SNapp’s coaching content provided personalized feedback on step counts and recommendations for increasing walking, while also considering individual preferences for behavior change techniques.
- The primary outcome was the daily step count recorded by the app, which was updated on an hourly basis in a database over an intervention period of 12 months.
- Perceptions of ease of use and usefulness were assessed to determine their effect on the effectiveness of the app.
TAKEAWAY:
- Intervention group participants used the app nearly 30% of days, while those using the app alone showed almost identical use.
- The SNapp intervention did not significantly affect the step counts on average over time (B, −202.30; 95% CI, −889.7 to 485.1).
- Perceived usefulness significantly moderated the intervention effect of SNapp (B, 344.38; 90% CI, 40.4-648.3), but perceived ease of use did not (B, 38.60; 90% CI, −276.5 to 353.7).
- Among participants with a high perceived usefulness, the SNapp group had a higher median step count than the control group (median difference, 1260 steps; 90% CI, −3243.7 to 1298.2); however, this difference was not statistically significant.
IN PRACTICE:
“This study shows that perceived usefulness is also an important factor influencing behavioral effects. Hence, it is essential for apps to be perceived as useful to effectively improve users’ activity levels,” the authors wrote.
SOURCE:
The study was led by Anne L. Vos, PhD, of the Amsterdam School of Communication Research at the University of Amsterdam, in the Netherlands. It was published online on September 16, 2024, in the American Journal of Preventive Medicine.
LIMITATIONS:
The study’s recruitment strategy primarily attracted highly educated individuals, limiting generalizability. The app’s accuracy in measuring steps could be improved, as it sometimes underestimated step counts. Researchers also were unable to check if participants read messages from coaches.
DISCLOSURES:
The study was supported by grants from the Dutch Heart Foundation and the Netherlands Organisation for Health Research and Development. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Apps designed to increase physical activity may be useful in increasing daily step counts for users who believe the intervention beneficial, but not for those who do not. The app’s effectiveness is notably influenced by how users perceive its utility.
METHODOLOGY:
- Researchers conducted a randomized controlled trial from February 2021 to May 2022 to evaluate the effectiveness of SNapp, an adaptive app designed to promote walking through tailored coaching content.
- Overall, 176 adults (76% women; mean age, 56 years) were randomly assigned to use the app plus tailored coaching content (SNapp group; n = 87) or only the step counter app (control group; n = 89).
- SNapp’s coaching content provided personalized feedback on step counts and recommendations for increasing walking, while also considering individual preferences for behavior change techniques.
- The primary outcome was the daily step count recorded by the app, which was updated on an hourly basis in a database over an intervention period of 12 months.
- Perceptions of ease of use and usefulness were assessed to determine their effect on the effectiveness of the app.
TAKEAWAY:
- Intervention group participants used the app nearly 30% of days, while those using the app alone showed almost identical use.
- The SNapp intervention did not significantly affect the step counts on average over time (B, −202.30; 95% CI, −889.7 to 485.1).
- Perceived usefulness significantly moderated the intervention effect of SNapp (B, 344.38; 90% CI, 40.4-648.3), but perceived ease of use did not (B, 38.60; 90% CI, −276.5 to 353.7).
- Among participants with a high perceived usefulness, the SNapp group had a higher median step count than the control group (median difference, 1260 steps; 90% CI, −3243.7 to 1298.2); however, this difference was not statistically significant.
IN PRACTICE:
“This study shows that perceived usefulness is also an important factor influencing behavioral effects. Hence, it is essential for apps to be perceived as useful to effectively improve users’ activity levels,” the authors wrote.
SOURCE:
The study was led by Anne L. Vos, PhD, of the Amsterdam School of Communication Research at the University of Amsterdam, in the Netherlands. It was published online on September 16, 2024, in the American Journal of Preventive Medicine.
LIMITATIONS:
The study’s recruitment strategy primarily attracted highly educated individuals, limiting generalizability. The app’s accuracy in measuring steps could be improved, as it sometimes underestimated step counts. Researchers also were unable to check if participants read messages from coaches.
DISCLOSURES:
The study was supported by grants from the Dutch Heart Foundation and the Netherlands Organisation for Health Research and Development. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
AACR Cancer Progress Report: Big Strides and Big Gaps
The AACR’s 216-page report — an annual endeavor now in its 14th year — focused on the “tremendous” strides made in cancer care, prevention, and early detection and highlighted areas where more research and attention are warranted.
One key area is funding. For the first time since 2016, federal funding for the National Institutes of Health (NIH) and National Cancer Institute (NCI) decreased in the past year. The cuts followed nearly a decade of funding increases that saw the NIH budget expand by nearly $15 billion, and that allowed for a “rapid pace and broad scope” of advances in cancer, AACR’s chief executive officer Margaret Foti, MD, PhD, said during a press briefing.
These recent cuts “threaten to curtail the medical progress seen in recent years and stymie future advancements,” said Dr. Foti, who called on Congress to commit to funding cancer research at significant and consistent levels to “maintain the momentum of progress against cancer.”
Inside the Report: Big Progress
Overall, advances in prevention, early detection, and treatment have helped catch more cancers earlier and save lives.
According to the AACR report, the age-adjusted overall cancer death rate in the United States fell by 33% between 1991 and 2021, meaning about 4.1 million cancer deaths were averted. The overall cancer death rate for children and adolescents has declined by 24% in the past 2 decades. The 5-year relative survival rate for children diagnosed with cancer in the US has improved from 58% for those diagnosed in the mid-1970s to 85% for those diagnosed between 2013 and 2019.
The past fiscal year has seen many new approvals for cancer drugs, diagnostics, and screening tests. From July 1, 2023, to June 30, 2024, the Food and Drug Administration (FDA) approved 15 new anticancer therapeutics, as well as 15 new indications for previously approved agents, one new imaging agent, several artificial intelligence (AI) tools to improve early cancer detection and diagnosis, and two minimally invasive tests for assessing inherited cancer risk or early cancer detection, according to the report.
“Cancer diagnostics are becoming more sophisticated,” AACR president Patricia M. LoRusso, DO, PhD, said during the briefing. “New technologies, such as spatial transcriptomics, are helping us study tumors at a cellular level, and helping to unveil things that we did not initially even begin to understand or think of. AI-based approaches are beginning to transform cancer detection, diagnosis, clinical decision-making, and treatment response monitoring.”
The report also highlights the significant progress in many childhood and adolescent/young adult cancers, Dr. LoRusso noted. These include FDA approvals for two new molecularly targeted therapeutics: tovorafenib for children with certain types of brain tumor and repotrectinib for children with a wide array of cancer types that have a specific genetic alteration known as NTRK gene fusion. It also includes an expanded approval for eflornithine to reduce the risk for relapse in children with high-risk neuroblastoma.
“Decades — decades — of basic research discoveries, have led to these clinical breakthroughs,” she stressed. “These gains against cancer are because of the rapid progress in our ability to decode the cancer genome, which has opened new and innovative avenues for drug development.”
The Gaps
Even with progress in cancer prevention, early detection, and treatment, cancer remains a significant issue.
“In 2024, it is estimated that more than 2 million new cases of cancer will be diagnosed in the United States. More than 611,000 people will die from the disease,” according to the report.
The 2024 report shows that incidence rates for some cancers are increasing in the United States, including vaccine-preventable cancers such as human papillomavirus (HPV)–associated oral cancers and, in young adults, cervical cancers. A recent analysis also found that overall cervical cancer incidence among women aged 30-34 years increased by 2.5% a year between 2012 and 2019.
Furthermore, despite clear evidence demonstrating that the HPV vaccine reduces cervical cancer incidence, uptake has remained poor, with only 38.6% of US children and adolescents aged 9-17 years receiving at least one dose of the vaccine in 2022.
Early-onset cancers are also increasing. Rates of breast, colorectal, and other cancers are on the rise in adults younger than 50 years, the report noted.
The report also pointed to data that 40% of all cancer cases in the United States can be attributed to preventable factors, such as smoking, excess body weight, and alcohol. However, our understanding of these risk factors has improved. Excessive levels of alcohol consumption have, for instance, been shown to increase the risk for six different types of cancer: certain types of head and neck cancer, esophageal squamous cell carcinoma, and breast, colorectal, liver, and stomach cancers.
Financial toxicity remains prevalent as well.
The report explains that financial hardship following a cancer diagnosis is widespread, and the effects can last for years. In fact, more than 40% of patients can spend their entire life savings within the first 2 years of cancer treatment. Among adult survivors of childhood cancers, 20.7% had trouble paying their medical bills, 29.9% said they had been sent to debt collection for unpaid bills, 14.1% had forgone medical care, and 26.8% could not afford nutritious meals.
For young cancer survivors, the lifetime costs associated with a diagnosis of cancer are substantial, reaching an average of $259,324 per person.
On a global level, it is estimated that from 2020 to 2050, the cumulative economic burden of cancer will be $25.2 trillion.
The Path Forward
Despite these challenges, Dr. LoRusso said, “it is unquestionable that we are in a time of unparalleled opportunities in cancer research.
“I am excited about what the future holds for cancer research, and especially for patient care,” she said.
However, funding commitments are needed to avoid impeding this momentum and losing a “talented and creative young workforce” that has brought new ideas and new technologies to the table.
Continued robust funding will help “to markedly improve cancer care, increase cancer survivorship, spur economic growth, and maintain the United States’ position as the global leader in science and medical research,” she added.
The AACR report specifically calls on Congress to:
- Appropriate at least $51.3 billion in fiscal year 2025 for the base budget of the NIH and at least $7.934 billion for the NCI.
- Provide $3.6 billion in dedicated funding for Cancer Moonshot activities through fiscal year 2026 in addition to other funding, consistent with the President’s fiscal year 2025 budget.
- Appropriate at least $472.4 million in fiscal year 2025 for the CDC’s Division of Cancer Prevention to support comprehensive cancer control, central cancer registries, and screening and awareness programs for specific cancers.
- Allocate $55 million in funding for the Oncology Center of Excellence at FDA in fiscal year 2025 to provide regulators with the staff and tools necessary to conduct expedited review of cancer-related medical products.
By working together with Congress and other stakeholders, “we will be able to accelerate the pace of progress and make major strides toward the lifesaving goal of preventing and curing all cancers at the earliest possible time,” Dr. Foti said. “I believe if we do that ... one day we will win this war on cancer.”
A version of this article first appeared on Medscape.com.
The AACR’s 216-page report — an annual endeavor now in its 14th year — focused on the “tremendous” strides made in cancer care, prevention, and early detection and highlighted areas where more research and attention are warranted.
One key area is funding. For the first time since 2016, federal funding for the National Institutes of Health (NIH) and National Cancer Institute (NCI) decreased in the past year. The cuts followed nearly a decade of funding increases that saw the NIH budget expand by nearly $15 billion, and that allowed for a “rapid pace and broad scope” of advances in cancer, AACR’s chief executive officer Margaret Foti, MD, PhD, said during a press briefing.
These recent cuts “threaten to curtail the medical progress seen in recent years and stymie future advancements,” said Dr. Foti, who called on Congress to commit to funding cancer research at significant and consistent levels to “maintain the momentum of progress against cancer.”
Inside the Report: Big Progress
Overall, advances in prevention, early detection, and treatment have helped catch more cancers earlier and save lives.
According to the AACR report, the age-adjusted overall cancer death rate in the United States fell by 33% between 1991 and 2021, meaning about 4.1 million cancer deaths were averted. The overall cancer death rate for children and adolescents has declined by 24% in the past 2 decades. The 5-year relative survival rate for children diagnosed with cancer in the US has improved from 58% for those diagnosed in the mid-1970s to 85% for those diagnosed between 2013 and 2019.
The past fiscal year has seen many new approvals for cancer drugs, diagnostics, and screening tests. From July 1, 2023, to June 30, 2024, the Food and Drug Administration (FDA) approved 15 new anticancer therapeutics, as well as 15 new indications for previously approved agents, one new imaging agent, several artificial intelligence (AI) tools to improve early cancer detection and diagnosis, and two minimally invasive tests for assessing inherited cancer risk or early cancer detection, according to the report.
“Cancer diagnostics are becoming more sophisticated,” AACR president Patricia M. LoRusso, DO, PhD, said during the briefing. “New technologies, such as spatial transcriptomics, are helping us study tumors at a cellular level, and helping to unveil things that we did not initially even begin to understand or think of. AI-based approaches are beginning to transform cancer detection, diagnosis, clinical decision-making, and treatment response monitoring.”
The report also highlights the significant progress in many childhood and adolescent/young adult cancers, Dr. LoRusso noted. These include FDA approvals for two new molecularly targeted therapeutics: tovorafenib for children with certain types of brain tumor and repotrectinib for children with a wide array of cancer types that have a specific genetic alteration known as NTRK gene fusion. It also includes an expanded approval for eflornithine to reduce the risk for relapse in children with high-risk neuroblastoma.
“Decades — decades — of basic research discoveries, have led to these clinical breakthroughs,” she stressed. “These gains against cancer are because of the rapid progress in our ability to decode the cancer genome, which has opened new and innovative avenues for drug development.”
The Gaps
Even with progress in cancer prevention, early detection, and treatment, cancer remains a significant issue.
“In 2024, it is estimated that more than 2 million new cases of cancer will be diagnosed in the United States. More than 611,000 people will die from the disease,” according to the report.
The 2024 report shows that incidence rates for some cancers are increasing in the United States, including vaccine-preventable cancers such as human papillomavirus (HPV)–associated oral cancers and, in young adults, cervical cancers. A recent analysis also found that overall cervical cancer incidence among women aged 30-34 years increased by 2.5% a year between 2012 and 2019.
Furthermore, despite clear evidence demonstrating that the HPV vaccine reduces cervical cancer incidence, uptake has remained poor, with only 38.6% of US children and adolescents aged 9-17 years receiving at least one dose of the vaccine in 2022.
Early-onset cancers are also increasing. Rates of breast, colorectal, and other cancers are on the rise in adults younger than 50 years, the report noted.
The report also pointed to data that 40% of all cancer cases in the United States can be attributed to preventable factors, such as smoking, excess body weight, and alcohol. However, our understanding of these risk factors has improved. Excessive levels of alcohol consumption have, for instance, been shown to increase the risk for six different types of cancer: certain types of head and neck cancer, esophageal squamous cell carcinoma, and breast, colorectal, liver, and stomach cancers.
Financial toxicity remains prevalent as well.
The report explains that financial hardship following a cancer diagnosis is widespread, and the effects can last for years. In fact, more than 40% of patients can spend their entire life savings within the first 2 years of cancer treatment. Among adult survivors of childhood cancers, 20.7% had trouble paying their medical bills, 29.9% said they had been sent to debt collection for unpaid bills, 14.1% had forgone medical care, and 26.8% could not afford nutritious meals.
For young cancer survivors, the lifetime costs associated with a diagnosis of cancer are substantial, reaching an average of $259,324 per person.
On a global level, it is estimated that from 2020 to 2050, the cumulative economic burden of cancer will be $25.2 trillion.
The Path Forward
Despite these challenges, Dr. LoRusso said, “it is unquestionable that we are in a time of unparalleled opportunities in cancer research.
“I am excited about what the future holds for cancer research, and especially for patient care,” she said.
However, funding commitments are needed to avoid impeding this momentum and losing a “talented and creative young workforce” that has brought new ideas and new technologies to the table.
Continued robust funding will help “to markedly improve cancer care, increase cancer survivorship, spur economic growth, and maintain the United States’ position as the global leader in science and medical research,” she added.
The AACR report specifically calls on Congress to:
- Appropriate at least $51.3 billion in fiscal year 2025 for the base budget of the NIH and at least $7.934 billion for the NCI.
- Provide $3.6 billion in dedicated funding for Cancer Moonshot activities through fiscal year 2026 in addition to other funding, consistent with the President’s fiscal year 2025 budget.
- Appropriate at least $472.4 million in fiscal year 2025 for the CDC’s Division of Cancer Prevention to support comprehensive cancer control, central cancer registries, and screening and awareness programs for specific cancers.
- Allocate $55 million in funding for the Oncology Center of Excellence at FDA in fiscal year 2025 to provide regulators with the staff and tools necessary to conduct expedited review of cancer-related medical products.
By working together with Congress and other stakeholders, “we will be able to accelerate the pace of progress and make major strides toward the lifesaving goal of preventing and curing all cancers at the earliest possible time,” Dr. Foti said. “I believe if we do that ... one day we will win this war on cancer.”
A version of this article first appeared on Medscape.com.
The AACR’s 216-page report — an annual endeavor now in its 14th year — focused on the “tremendous” strides made in cancer care, prevention, and early detection and highlighted areas where more research and attention are warranted.
One key area is funding. For the first time since 2016, federal funding for the National Institutes of Health (NIH) and National Cancer Institute (NCI) decreased in the past year. The cuts followed nearly a decade of funding increases that saw the NIH budget expand by nearly $15 billion, and that allowed for a “rapid pace and broad scope” of advances in cancer, AACR’s chief executive officer Margaret Foti, MD, PhD, said during a press briefing.
These recent cuts “threaten to curtail the medical progress seen in recent years and stymie future advancements,” said Dr. Foti, who called on Congress to commit to funding cancer research at significant and consistent levels to “maintain the momentum of progress against cancer.”
Inside the Report: Big Progress
Overall, advances in prevention, early detection, and treatment have helped catch more cancers earlier and save lives.
According to the AACR report, the age-adjusted overall cancer death rate in the United States fell by 33% between 1991 and 2021, meaning about 4.1 million cancer deaths were averted. The overall cancer death rate for children and adolescents has declined by 24% in the past 2 decades. The 5-year relative survival rate for children diagnosed with cancer in the US has improved from 58% for those diagnosed in the mid-1970s to 85% for those diagnosed between 2013 and 2019.
The past fiscal year has seen many new approvals for cancer drugs, diagnostics, and screening tests. From July 1, 2023, to June 30, 2024, the Food and Drug Administration (FDA) approved 15 new anticancer therapeutics, as well as 15 new indications for previously approved agents, one new imaging agent, several artificial intelligence (AI) tools to improve early cancer detection and diagnosis, and two minimally invasive tests for assessing inherited cancer risk or early cancer detection, according to the report.
“Cancer diagnostics are becoming more sophisticated,” AACR president Patricia M. LoRusso, DO, PhD, said during the briefing. “New technologies, such as spatial transcriptomics, are helping us study tumors at a cellular level, and helping to unveil things that we did not initially even begin to understand or think of. AI-based approaches are beginning to transform cancer detection, diagnosis, clinical decision-making, and treatment response monitoring.”
The report also highlights the significant progress in many childhood and adolescent/young adult cancers, Dr. LoRusso noted. These include FDA approvals for two new molecularly targeted therapeutics: tovorafenib for children with certain types of brain tumor and repotrectinib for children with a wide array of cancer types that have a specific genetic alteration known as NTRK gene fusion. It also includes an expanded approval for eflornithine to reduce the risk for relapse in children with high-risk neuroblastoma.
“Decades — decades — of basic research discoveries, have led to these clinical breakthroughs,” she stressed. “These gains against cancer are because of the rapid progress in our ability to decode the cancer genome, which has opened new and innovative avenues for drug development.”
The Gaps
Even with progress in cancer prevention, early detection, and treatment, cancer remains a significant issue.
“In 2024, it is estimated that more than 2 million new cases of cancer will be diagnosed in the United States. More than 611,000 people will die from the disease,” according to the report.
The 2024 report shows that incidence rates for some cancers are increasing in the United States, including vaccine-preventable cancers such as human papillomavirus (HPV)–associated oral cancers and, in young adults, cervical cancers. A recent analysis also found that overall cervical cancer incidence among women aged 30-34 years increased by 2.5% a year between 2012 and 2019.
Furthermore, despite clear evidence demonstrating that the HPV vaccine reduces cervical cancer incidence, uptake has remained poor, with only 38.6% of US children and adolescents aged 9-17 years receiving at least one dose of the vaccine in 2022.
Early-onset cancers are also increasing. Rates of breast, colorectal, and other cancers are on the rise in adults younger than 50 years, the report noted.
The report also pointed to data that 40% of all cancer cases in the United States can be attributed to preventable factors, such as smoking, excess body weight, and alcohol. However, our understanding of these risk factors has improved. Excessive levels of alcohol consumption have, for instance, been shown to increase the risk for six different types of cancer: certain types of head and neck cancer, esophageal squamous cell carcinoma, and breast, colorectal, liver, and stomach cancers.
Financial toxicity remains prevalent as well.
The report explains that financial hardship following a cancer diagnosis is widespread, and the effects can last for years. In fact, more than 40% of patients can spend their entire life savings within the first 2 years of cancer treatment. Among adult survivors of childhood cancers, 20.7% had trouble paying their medical bills, 29.9% said they had been sent to debt collection for unpaid bills, 14.1% had forgone medical care, and 26.8% could not afford nutritious meals.
For young cancer survivors, the lifetime costs associated with a diagnosis of cancer are substantial, reaching an average of $259,324 per person.
On a global level, it is estimated that from 2020 to 2050, the cumulative economic burden of cancer will be $25.2 trillion.
The Path Forward
Despite these challenges, Dr. LoRusso said, “it is unquestionable that we are in a time of unparalleled opportunities in cancer research.
“I am excited about what the future holds for cancer research, and especially for patient care,” she said.
However, funding commitments are needed to avoid impeding this momentum and losing a “talented and creative young workforce” that has brought new ideas and new technologies to the table.
Continued robust funding will help “to markedly improve cancer care, increase cancer survivorship, spur economic growth, and maintain the United States’ position as the global leader in science and medical research,” she added.
The AACR report specifically calls on Congress to:
- Appropriate at least $51.3 billion in fiscal year 2025 for the base budget of the NIH and at least $7.934 billion for the NCI.
- Provide $3.6 billion in dedicated funding for Cancer Moonshot activities through fiscal year 2026 in addition to other funding, consistent with the President’s fiscal year 2025 budget.
- Appropriate at least $472.4 million in fiscal year 2025 for the CDC’s Division of Cancer Prevention to support comprehensive cancer control, central cancer registries, and screening and awareness programs for specific cancers.
- Allocate $55 million in funding for the Oncology Center of Excellence at FDA in fiscal year 2025 to provide regulators with the staff and tools necessary to conduct expedited review of cancer-related medical products.
By working together with Congress and other stakeholders, “we will be able to accelerate the pace of progress and make major strides toward the lifesaving goal of preventing and curing all cancers at the earliest possible time,” Dr. Foti said. “I believe if we do that ... one day we will win this war on cancer.”
A version of this article first appeared on Medscape.com.
Considering Eating Disorder Potential When Prescribing Weight Loss Drugs to Teens
Now, with the rising popularity of medications like Ozempic for weight loss, she fears she will care for more teens with eating disorders who are seeking glucagon-like peptide 1 (GLP-1) agonists or who have developed a disorder while taking them.
“We haven’t seen patients yet, but I’m sure they are on their way,” said Dr. Dennis, a clinical assistant professor in the Department of Psychiatry at the University of Illinois College of Medicine in Chicago. She is also the cofounder and chief medical officer of SunCloud Health, an outpatient eating disorder treatment center in Illinois.
Dr. Dennis’ concerns reflect a growing unease among eating disorder specialists as obesity medications gain traction for adolescent use. A recent study published in JAMA Pediatrics showed nearly 80% of teens in treatment for obesity reported symptoms of disordered eating at the outset of an intervention. These included signs of binge eating and loss of control.
The randomized clinical trial, conducted from 2018 to 2023, examined 141 adolescents with obesity undergoing interventions like low-energy diets or intermittent energy restriction. Almost half scored as having risk for an undiagnosed eating disorder, as defined by the Eating Disorder Examination Questionnaire (EDE-Q).
At the end of the intervention, many teens continued to have symptoms of disordered eating, while a smaller group was newly scored as having a risk for an eating disorder.
Weight Loss and Eating Disorders: A Balancing Act
The findings illuminate a significant challenge for pediatricians and primary care clinicians: Balancing effective weight management with the risk of exacerbating or triggering eating disorders, said Hiba Jebeile, PhD, a research dietitian at The Children’s Hospital at Westmead in Australia, and the study’s lead author. Adding weight loss medication on top of the equation can further complicate care.
“It is helpful for obesity and eating disorder services to work together, with clear referral pathways, to manage these adolescents,” Dr. Jebeile said.
The US Food and Drug Administration approved semaglutide for weight loss in adolescents aged 12-17 years in December 2020. One study found that the number of adolescents prescribed GLP-1 receptor agonists (GLP-1 RAs) for type 2 diabetes and weight management rose from 8722 to 60,567 between 2020 and 2023 — a nearly sevenfold increase.
“The number of adolescents taking these medications is going up because they work,” said Suzanne Cuda, MD, medical director of Alamo City Healthy Kids and Families, a medical weight management clinic in San Antonio. The medications have been shown to treat type 2 diabetes, lower blood pressure, and reduce the risk for cardiovascular diseases.
“The younger you are, the better the outcome,” Dr. Cuda said.
How GLP-1 agonists may affect adolescents in the long run is not yet clear. Existing studies on GLP-1 medications in patients with eating disorders have shown mixed results. Some studies indicate that the drugs decrease binge episodes for those with binge eating disorder or bulimia nervosa. However, these studies had small sample sizes and measured only short-term effects, leaving long-term outcomes and risks unknown.
Traditional treatments for eating disorders emphasize regular eating patterns, body acceptance, addressing weight stigma, and improving attunement to hunger and fullness cues — approaches that may conflict with the effects of GLP-1 agonists. These drugs suppress appetite, alter metabolic signals, and may unintentionally reinforce weight loss as a primary goal, creating a potential disconnect between the aims of recovery from eating disorders and the biologic effects of the medication, experts said.
Dr. Cuda said she has cared for adolescents with diagnosed eating disorders in her practice who are seeking GLP-1 agonists. She said she first works with patients to treat the underlying disorder before prescribing medication.
“One of the concerns is the extreme reductions in calories that could be induced by GLP-1 RA in children and adolescents,” she said. Unlike adults, adolescents use caloric energy not just for physical activity but also for growing and developing, she said.
“They can’t catch up on that growth and development,” she added.
Advice for Screening and Monitoring
The National Eating Disorders Association raised concerns about the potential misuse of these medications and their potential to exacerbate eating disorder behaviors in people who are already at a higher risk of developing one of the conditions, including those with existing mental health disorders, stress, who have already dieted, and who have experienced weight-centric bullying.
Clinicians should be on the lookout for patients seeking GLP-1s who present with symptoms of an eating disorder that may be less apparent, such as picky eating, insomnia or difficulty sleeping, or, for girls, irregular menstrual periods, Dr. Dennis said. These patients may be more likely to go undiagnosed or misdiagnosed. Research also suggests that people of color are less likely to be diagnosed or receive specialty care for eating disorders.
Discussions between patients and clinicians about obesity treatment prior to prescribing provide a crucial opportunity to screen and monitor for disordered eating, which Dr. Dennis said does not universally occur currently.
Dr. Dennis recommended initial assessments using validated screening tools like the EDE-Q and the Center for Epidemiologic Studies Depression Scale Revised, 10-Item Version.
Ongoing monitoring throughout treatment is essential, with initial monthly check-ins that include dietary counseling to detect subtle changes in eating behaviors or attitudes toward food and body image, Dr. Cuda said.
The Obesity Medicine Association (OMA) has stressed the importance of a collaborative approach involving connections with mental health professionals specializing in eating disorders and dietitians.
“If you have a chance to send them to an obesity medicine specialist, you should do that,” said Dr. Cuda, who coauthored the OMA statement. “It’s impractical to expect a primary care physician to do everything: Screen for dietary disorders, do a full dietary counseling, follow up on their activity.”
For patients showing signs of disordered eating, clinicians should avoid recommending restrictive dietary approaches, like cutting out food groups such as carbohydrates or a restricted calorie goal. Instead, they can suggest focusing on healthier lifestyle habits and referring to a psychotherapist, the experts said. Clinicians also should be prepared to adjust or pause GLP-1 agonists if disordered eating disorder symptoms worsen.
“I think a weight-agnostic approach where the focus of care is not weight loss but increase in health protective behaviors and nutritional intake is safest for all kids, especially those with eating disorders or eating disorder risk factors,” Dr. Dennis said.
Various authors of the eating disorder study reported receiving grants, advisory board fees, and speaker fees from entities including the National Health and Medical Research Council of Australia, Eli Lilly, Novo Nordisk, Nu-Mega Ingredients, and the National Institutes of Health, among others.
A version of this article appeared on Medscape.com.
Now, with the rising popularity of medications like Ozempic for weight loss, she fears she will care for more teens with eating disorders who are seeking glucagon-like peptide 1 (GLP-1) agonists or who have developed a disorder while taking them.
“We haven’t seen patients yet, but I’m sure they are on their way,” said Dr. Dennis, a clinical assistant professor in the Department of Psychiatry at the University of Illinois College of Medicine in Chicago. She is also the cofounder and chief medical officer of SunCloud Health, an outpatient eating disorder treatment center in Illinois.
Dr. Dennis’ concerns reflect a growing unease among eating disorder specialists as obesity medications gain traction for adolescent use. A recent study published in JAMA Pediatrics showed nearly 80% of teens in treatment for obesity reported symptoms of disordered eating at the outset of an intervention. These included signs of binge eating and loss of control.
The randomized clinical trial, conducted from 2018 to 2023, examined 141 adolescents with obesity undergoing interventions like low-energy diets or intermittent energy restriction. Almost half scored as having risk for an undiagnosed eating disorder, as defined by the Eating Disorder Examination Questionnaire (EDE-Q).
At the end of the intervention, many teens continued to have symptoms of disordered eating, while a smaller group was newly scored as having a risk for an eating disorder.
Weight Loss and Eating Disorders: A Balancing Act
The findings illuminate a significant challenge for pediatricians and primary care clinicians: Balancing effective weight management with the risk of exacerbating or triggering eating disorders, said Hiba Jebeile, PhD, a research dietitian at The Children’s Hospital at Westmead in Australia, and the study’s lead author. Adding weight loss medication on top of the equation can further complicate care.
“It is helpful for obesity and eating disorder services to work together, with clear referral pathways, to manage these adolescents,” Dr. Jebeile said.
The US Food and Drug Administration approved semaglutide for weight loss in adolescents aged 12-17 years in December 2020. One study found that the number of adolescents prescribed GLP-1 receptor agonists (GLP-1 RAs) for type 2 diabetes and weight management rose from 8722 to 60,567 between 2020 and 2023 — a nearly sevenfold increase.
“The number of adolescents taking these medications is going up because they work,” said Suzanne Cuda, MD, medical director of Alamo City Healthy Kids and Families, a medical weight management clinic in San Antonio. The medications have been shown to treat type 2 diabetes, lower blood pressure, and reduce the risk for cardiovascular diseases.
“The younger you are, the better the outcome,” Dr. Cuda said.
How GLP-1 agonists may affect adolescents in the long run is not yet clear. Existing studies on GLP-1 medications in patients with eating disorders have shown mixed results. Some studies indicate that the drugs decrease binge episodes for those with binge eating disorder or bulimia nervosa. However, these studies had small sample sizes and measured only short-term effects, leaving long-term outcomes and risks unknown.
Traditional treatments for eating disorders emphasize regular eating patterns, body acceptance, addressing weight stigma, and improving attunement to hunger and fullness cues — approaches that may conflict with the effects of GLP-1 agonists. These drugs suppress appetite, alter metabolic signals, and may unintentionally reinforce weight loss as a primary goal, creating a potential disconnect between the aims of recovery from eating disorders and the biologic effects of the medication, experts said.
Dr. Cuda said she has cared for adolescents with diagnosed eating disorders in her practice who are seeking GLP-1 agonists. She said she first works with patients to treat the underlying disorder before prescribing medication.
“One of the concerns is the extreme reductions in calories that could be induced by GLP-1 RA in children and adolescents,” she said. Unlike adults, adolescents use caloric energy not just for physical activity but also for growing and developing, she said.
“They can’t catch up on that growth and development,” she added.
Advice for Screening and Monitoring
The National Eating Disorders Association raised concerns about the potential misuse of these medications and their potential to exacerbate eating disorder behaviors in people who are already at a higher risk of developing one of the conditions, including those with existing mental health disorders, stress, who have already dieted, and who have experienced weight-centric bullying.
Clinicians should be on the lookout for patients seeking GLP-1s who present with symptoms of an eating disorder that may be less apparent, such as picky eating, insomnia or difficulty sleeping, or, for girls, irregular menstrual periods, Dr. Dennis said. These patients may be more likely to go undiagnosed or misdiagnosed. Research also suggests that people of color are less likely to be diagnosed or receive specialty care for eating disorders.
Discussions between patients and clinicians about obesity treatment prior to prescribing provide a crucial opportunity to screen and monitor for disordered eating, which Dr. Dennis said does not universally occur currently.
Dr. Dennis recommended initial assessments using validated screening tools like the EDE-Q and the Center for Epidemiologic Studies Depression Scale Revised, 10-Item Version.
Ongoing monitoring throughout treatment is essential, with initial monthly check-ins that include dietary counseling to detect subtle changes in eating behaviors or attitudes toward food and body image, Dr. Cuda said.
The Obesity Medicine Association (OMA) has stressed the importance of a collaborative approach involving connections with mental health professionals specializing in eating disorders and dietitians.
“If you have a chance to send them to an obesity medicine specialist, you should do that,” said Dr. Cuda, who coauthored the OMA statement. “It’s impractical to expect a primary care physician to do everything: Screen for dietary disorders, do a full dietary counseling, follow up on their activity.”
For patients showing signs of disordered eating, clinicians should avoid recommending restrictive dietary approaches, like cutting out food groups such as carbohydrates or a restricted calorie goal. Instead, they can suggest focusing on healthier lifestyle habits and referring to a psychotherapist, the experts said. Clinicians also should be prepared to adjust or pause GLP-1 agonists if disordered eating disorder symptoms worsen.
“I think a weight-agnostic approach where the focus of care is not weight loss but increase in health protective behaviors and nutritional intake is safest for all kids, especially those with eating disorders or eating disorder risk factors,” Dr. Dennis said.
Various authors of the eating disorder study reported receiving grants, advisory board fees, and speaker fees from entities including the National Health and Medical Research Council of Australia, Eli Lilly, Novo Nordisk, Nu-Mega Ingredients, and the National Institutes of Health, among others.
A version of this article appeared on Medscape.com.
Now, with the rising popularity of medications like Ozempic for weight loss, she fears she will care for more teens with eating disorders who are seeking glucagon-like peptide 1 (GLP-1) agonists or who have developed a disorder while taking them.
“We haven’t seen patients yet, but I’m sure they are on their way,” said Dr. Dennis, a clinical assistant professor in the Department of Psychiatry at the University of Illinois College of Medicine in Chicago. She is also the cofounder and chief medical officer of SunCloud Health, an outpatient eating disorder treatment center in Illinois.
Dr. Dennis’ concerns reflect a growing unease among eating disorder specialists as obesity medications gain traction for adolescent use. A recent study published in JAMA Pediatrics showed nearly 80% of teens in treatment for obesity reported symptoms of disordered eating at the outset of an intervention. These included signs of binge eating and loss of control.
The randomized clinical trial, conducted from 2018 to 2023, examined 141 adolescents with obesity undergoing interventions like low-energy diets or intermittent energy restriction. Almost half scored as having risk for an undiagnosed eating disorder, as defined by the Eating Disorder Examination Questionnaire (EDE-Q).
At the end of the intervention, many teens continued to have symptoms of disordered eating, while a smaller group was newly scored as having a risk for an eating disorder.
Weight Loss and Eating Disorders: A Balancing Act
The findings illuminate a significant challenge for pediatricians and primary care clinicians: Balancing effective weight management with the risk of exacerbating or triggering eating disorders, said Hiba Jebeile, PhD, a research dietitian at The Children’s Hospital at Westmead in Australia, and the study’s lead author. Adding weight loss medication on top of the equation can further complicate care.
“It is helpful for obesity and eating disorder services to work together, with clear referral pathways, to manage these adolescents,” Dr. Jebeile said.
The US Food and Drug Administration approved semaglutide for weight loss in adolescents aged 12-17 years in December 2020. One study found that the number of adolescents prescribed GLP-1 receptor agonists (GLP-1 RAs) for type 2 diabetes and weight management rose from 8722 to 60,567 between 2020 and 2023 — a nearly sevenfold increase.
“The number of adolescents taking these medications is going up because they work,” said Suzanne Cuda, MD, medical director of Alamo City Healthy Kids and Families, a medical weight management clinic in San Antonio. The medications have been shown to treat type 2 diabetes, lower blood pressure, and reduce the risk for cardiovascular diseases.
“The younger you are, the better the outcome,” Dr. Cuda said.
How GLP-1 agonists may affect adolescents in the long run is not yet clear. Existing studies on GLP-1 medications in patients with eating disorders have shown mixed results. Some studies indicate that the drugs decrease binge episodes for those with binge eating disorder or bulimia nervosa. However, these studies had small sample sizes and measured only short-term effects, leaving long-term outcomes and risks unknown.
Traditional treatments for eating disorders emphasize regular eating patterns, body acceptance, addressing weight stigma, and improving attunement to hunger and fullness cues — approaches that may conflict with the effects of GLP-1 agonists. These drugs suppress appetite, alter metabolic signals, and may unintentionally reinforce weight loss as a primary goal, creating a potential disconnect between the aims of recovery from eating disorders and the biologic effects of the medication, experts said.
Dr. Cuda said she has cared for adolescents with diagnosed eating disorders in her practice who are seeking GLP-1 agonists. She said she first works with patients to treat the underlying disorder before prescribing medication.
“One of the concerns is the extreme reductions in calories that could be induced by GLP-1 RA in children and adolescents,” she said. Unlike adults, adolescents use caloric energy not just for physical activity but also for growing and developing, she said.
“They can’t catch up on that growth and development,” she added.
Advice for Screening and Monitoring
The National Eating Disorders Association raised concerns about the potential misuse of these medications and their potential to exacerbate eating disorder behaviors in people who are already at a higher risk of developing one of the conditions, including those with existing mental health disorders, stress, who have already dieted, and who have experienced weight-centric bullying.
Clinicians should be on the lookout for patients seeking GLP-1s who present with symptoms of an eating disorder that may be less apparent, such as picky eating, insomnia or difficulty sleeping, or, for girls, irregular menstrual periods, Dr. Dennis said. These patients may be more likely to go undiagnosed or misdiagnosed. Research also suggests that people of color are less likely to be diagnosed or receive specialty care for eating disorders.
Discussions between patients and clinicians about obesity treatment prior to prescribing provide a crucial opportunity to screen and monitor for disordered eating, which Dr. Dennis said does not universally occur currently.
Dr. Dennis recommended initial assessments using validated screening tools like the EDE-Q and the Center for Epidemiologic Studies Depression Scale Revised, 10-Item Version.
Ongoing monitoring throughout treatment is essential, with initial monthly check-ins that include dietary counseling to detect subtle changes in eating behaviors or attitudes toward food and body image, Dr. Cuda said.
The Obesity Medicine Association (OMA) has stressed the importance of a collaborative approach involving connections with mental health professionals specializing in eating disorders and dietitians.
“If you have a chance to send them to an obesity medicine specialist, you should do that,” said Dr. Cuda, who coauthored the OMA statement. “It’s impractical to expect a primary care physician to do everything: Screen for dietary disorders, do a full dietary counseling, follow up on their activity.”
For patients showing signs of disordered eating, clinicians should avoid recommending restrictive dietary approaches, like cutting out food groups such as carbohydrates or a restricted calorie goal. Instead, they can suggest focusing on healthier lifestyle habits and referring to a psychotherapist, the experts said. Clinicians also should be prepared to adjust or pause GLP-1 agonists if disordered eating disorder symptoms worsen.
“I think a weight-agnostic approach where the focus of care is not weight loss but increase in health protective behaviors and nutritional intake is safest for all kids, especially those with eating disorders or eating disorder risk factors,” Dr. Dennis said.
Various authors of the eating disorder study reported receiving grants, advisory board fees, and speaker fees from entities including the National Health and Medical Research Council of Australia, Eli Lilly, Novo Nordisk, Nu-Mega Ingredients, and the National Institutes of Health, among others.
A version of this article appeared on Medscape.com.
FROM JAMA PEDIATRICS
Does Preconception BMI Affect Time to Pregnancy and Miscarriage Risk?
TOPLINE:
Higher body mass index (BMI) in both partners is linked to lower fecundability and increased subfertility. Overweight and obesity in women are associated with higher odds of miscarriage.
METHODOLOGY:
- Researchers conducted a population-based prospective cohort study in Rotterdam, the Netherlands, from August 9, 2017, to July 1, 2021.
- A total of 3604 women and their partners were included, with follow-up until birth.
- BMI was measured in preconception or early pregnancy, and outcomes included fecundability, subfertility, and miscarriage.
- Fecundability was defined as the probability of conceiving within 1 month and subfertility as time to pregnancy or duration of actively pursuing pregnancy of more than 12 months or use of assisted reproductive technology.
- Miscarriage was defined as pregnancy loss before 22 weeks of gestation.
TAKEAWAY:
- Higher BMI in women and men was associated with lower fecundability: For every unit increase in BMI, fecundability decreased (women, 0.98; 95% CI, 0.97-0.99; men, 0.99; 95% CI, 0.98-1.00).
- Women with overweight (0.88; 95% CI, 0.80-0.98) and obesity (0.72; 95% CI, 0.63-0.82) had lower fecundability than women with normal weight.
- Overweight (1.35; 95% CI, 1.11-1.63) and obesity (1.67; 95% CI, 1.30-2.13) in women were associated with increased odds of subfertility.
- Obesity in men was associated with increased odds of subfertility (1.69; 95% CI, 1.24-2.31).
IN PRACTICE:
“We observed in this cohort study that BMI outside of the normal category in women and men was associated with lower fecundability, subfertility, and increased odds of miscarriage. Optimizing BMI from the preconception period onward in women and men might be an important strategy to improve fertility and pregnancy outcomes,” wrote the authors of the study.
SOURCE:
The study was led by Aline J. Boxem, MD, and Vincent W. V. Jaddoe, MD, PhD, The Generation R Study Group, Erasmus University Medical Centre in Rotterdam, the Netherlands. It was published online in JAMA Network Open.
LIMITATIONS:
The study’s generalizability may be affected by differences between included and excluded participants, who were younger and had a higher BMI. The accuracy of time-to-pregnancy duration may have been impacted by retrospectively answered questionnaires. Residual confounding might still be an issue due to the observational nature of the study.
DISCLOSURES:
Dr. Boxem and Dr. Jaddoe disclosed receiving grants from the Erasmus University Medical Centre, the Erasmus University Rotterdam, and the Netherlands Organisation for Health Research and Development. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Higher body mass index (BMI) in both partners is linked to lower fecundability and increased subfertility. Overweight and obesity in women are associated with higher odds of miscarriage.
METHODOLOGY:
- Researchers conducted a population-based prospective cohort study in Rotterdam, the Netherlands, from August 9, 2017, to July 1, 2021.
- A total of 3604 women and their partners were included, with follow-up until birth.
- BMI was measured in preconception or early pregnancy, and outcomes included fecundability, subfertility, and miscarriage.
- Fecundability was defined as the probability of conceiving within 1 month and subfertility as time to pregnancy or duration of actively pursuing pregnancy of more than 12 months or use of assisted reproductive technology.
- Miscarriage was defined as pregnancy loss before 22 weeks of gestation.
TAKEAWAY:
- Higher BMI in women and men was associated with lower fecundability: For every unit increase in BMI, fecundability decreased (women, 0.98; 95% CI, 0.97-0.99; men, 0.99; 95% CI, 0.98-1.00).
- Women with overweight (0.88; 95% CI, 0.80-0.98) and obesity (0.72; 95% CI, 0.63-0.82) had lower fecundability than women with normal weight.
- Overweight (1.35; 95% CI, 1.11-1.63) and obesity (1.67; 95% CI, 1.30-2.13) in women were associated with increased odds of subfertility.
- Obesity in men was associated with increased odds of subfertility (1.69; 95% CI, 1.24-2.31).
IN PRACTICE:
“We observed in this cohort study that BMI outside of the normal category in women and men was associated with lower fecundability, subfertility, and increased odds of miscarriage. Optimizing BMI from the preconception period onward in women and men might be an important strategy to improve fertility and pregnancy outcomes,” wrote the authors of the study.
SOURCE:
The study was led by Aline J. Boxem, MD, and Vincent W. V. Jaddoe, MD, PhD, The Generation R Study Group, Erasmus University Medical Centre in Rotterdam, the Netherlands. It was published online in JAMA Network Open.
LIMITATIONS:
The study’s generalizability may be affected by differences between included and excluded participants, who were younger and had a higher BMI. The accuracy of time-to-pregnancy duration may have been impacted by retrospectively answered questionnaires. Residual confounding might still be an issue due to the observational nature of the study.
DISCLOSURES:
Dr. Boxem and Dr. Jaddoe disclosed receiving grants from the Erasmus University Medical Centre, the Erasmus University Rotterdam, and the Netherlands Organisation for Health Research and Development. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Higher body mass index (BMI) in both partners is linked to lower fecundability and increased subfertility. Overweight and obesity in women are associated with higher odds of miscarriage.
METHODOLOGY:
- Researchers conducted a population-based prospective cohort study in Rotterdam, the Netherlands, from August 9, 2017, to July 1, 2021.
- A total of 3604 women and their partners were included, with follow-up until birth.
- BMI was measured in preconception or early pregnancy, and outcomes included fecundability, subfertility, and miscarriage.
- Fecundability was defined as the probability of conceiving within 1 month and subfertility as time to pregnancy or duration of actively pursuing pregnancy of more than 12 months or use of assisted reproductive technology.
- Miscarriage was defined as pregnancy loss before 22 weeks of gestation.
TAKEAWAY:
- Higher BMI in women and men was associated with lower fecundability: For every unit increase in BMI, fecundability decreased (women, 0.98; 95% CI, 0.97-0.99; men, 0.99; 95% CI, 0.98-1.00).
- Women with overweight (0.88; 95% CI, 0.80-0.98) and obesity (0.72; 95% CI, 0.63-0.82) had lower fecundability than women with normal weight.
- Overweight (1.35; 95% CI, 1.11-1.63) and obesity (1.67; 95% CI, 1.30-2.13) in women were associated with increased odds of subfertility.
- Obesity in men was associated with increased odds of subfertility (1.69; 95% CI, 1.24-2.31).
IN PRACTICE:
“We observed in this cohort study that BMI outside of the normal category in women and men was associated with lower fecundability, subfertility, and increased odds of miscarriage. Optimizing BMI from the preconception period onward in women and men might be an important strategy to improve fertility and pregnancy outcomes,” wrote the authors of the study.
SOURCE:
The study was led by Aline J. Boxem, MD, and Vincent W. V. Jaddoe, MD, PhD, The Generation R Study Group, Erasmus University Medical Centre in Rotterdam, the Netherlands. It was published online in JAMA Network Open.
LIMITATIONS:
The study’s generalizability may be affected by differences between included and excluded participants, who were younger and had a higher BMI. The accuracy of time-to-pregnancy duration may have been impacted by retrospectively answered questionnaires. Residual confounding might still be an issue due to the observational nature of the study.
DISCLOSURES:
Dr. Boxem and Dr. Jaddoe disclosed receiving grants from the Erasmus University Medical Centre, the Erasmus University Rotterdam, and the Netherlands Organisation for Health Research and Development. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.

