User login
Hepatitis A is on the rise: What FPs can do
In September 2021, a community in Virginia experienced an outbreak of hepatitis A virus (HAV) that was ultimately linked to an infected food handler.1 A total of 149 cases were reported over the next 12 months; 51 were directly related to the food handler and the remainder were the result of sustained community transmission. Of the 51 people who were directly infected by the food handler, 31 were hospitalized and 3 died. This incident offers important reminders about public health surveillance and the role that family physicians can play.
Hepatitis A virus is transmitted through food and drinks that have been contaminated by small amounts of stool that contains the virus or through close contact (including sexual contact) with a person who is infected. The incubation period can range from 15 to 59 days.
HAV generally resolves in a few days to weeks, with no long-term effects. However, recent outbreaks have been associated with high hospitalization and mortality rates because of the underlying comorbidities of those infected.
An increase in incidence. The national rate of HAV infection reached a low of less than 1/100,000 in 2015 but has since increased to almost 6/100,000 in 2019. This increase is mostly due to outbreaks linked to spread among people without a fixed residence, those who use illicit drugs, and men who have sex with men.2
In the Virginia outbreak, the food handler had a risk factor for HAV and was unvaccinated. He worked at 3 different locations of a restaurant chain for a total of 16 days while infectious, preparing ready-to-eat food without using gloves. Furthermore, he delayed seeking medical care for more than 2 weeks—at which time, the nature of his employment was not disclosed.
Prevention is straightforward. HAV infection can be prevented by administration of either HAV vaccine or immune globulin within 2 weeks of exposure.3 During an HAV outbreak, vaccination is recommended for people considered to be at risk, including those without a fixed residence, those who use illicit drugs, those who travel internationally, and men who have sex with men.3
There are 3 HAV vaccines available in the United States: 2 single-antigen vaccines, Havrix and Vaqta, both approved for children and adults, and a combination vaccine (containing both HAV and hepatitis B antigens), Twinrix, which is approved for those ages 18 years and older. All are inactivated vaccines.
What you can do. The Virginia outbreak illustrates the important role that family physicians can and do play in public health. We should:
- Encourage adults with risk factors for HAV to be vaccinated.
- Ask those with an HAV diagnosis about the people they may have exposed through personal contact or occupational exposure.
- Promptly report infectious diseases that are designated “reportable” to the public health department.
- Immediately report (by telephone) when HAV and other enteric infections involve a food handler.
1. Helmick MJ, Morrow CB, White JH, et al. Widespread community transmission of Hepatitis A Virus following an outbreak at a local restaurant—Virginia, September 2021-September 2022. MMWR Morb Mortal Wkly Rep. 2023;72;362-365. doi: 10.15585/mmwr.mm7214a2
2. CDC. Hepatitis A questions and answers for health professionals. Updated July 28, 2020. Accessed April 25, 2023. www.cdc.gov/hepatitis/hav/havfaq.htm
3. Nelson NP, Weng MK, Hofmeister MG, et al. Prevention of hepatitis A virus infection in the United States: recommendations of the Advisory Committee on Immunization Practices, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1-38. doi: 10.15585/mmwr.rr6905a1
In September 2021, a community in Virginia experienced an outbreak of hepatitis A virus (HAV) that was ultimately linked to an infected food handler.1 A total of 149 cases were reported over the next 12 months; 51 were directly related to the food handler and the remainder were the result of sustained community transmission. Of the 51 people who were directly infected by the food handler, 31 were hospitalized and 3 died. This incident offers important reminders about public health surveillance and the role that family physicians can play.
Hepatitis A virus is transmitted through food and drinks that have been contaminated by small amounts of stool that contains the virus or through close contact (including sexual contact) with a person who is infected. The incubation period can range from 15 to 59 days.
HAV generally resolves in a few days to weeks, with no long-term effects. However, recent outbreaks have been associated with high hospitalization and mortality rates because of the underlying comorbidities of those infected.
An increase in incidence. The national rate of HAV infection reached a low of less than 1/100,000 in 2015 but has since increased to almost 6/100,000 in 2019. This increase is mostly due to outbreaks linked to spread among people without a fixed residence, those who use illicit drugs, and men who have sex with men.2
In the Virginia outbreak, the food handler had a risk factor for HAV and was unvaccinated. He worked at 3 different locations of a restaurant chain for a total of 16 days while infectious, preparing ready-to-eat food without using gloves. Furthermore, he delayed seeking medical care for more than 2 weeks—at which time, the nature of his employment was not disclosed.
Prevention is straightforward. HAV infection can be prevented by administration of either HAV vaccine or immune globulin within 2 weeks of exposure.3 During an HAV outbreak, vaccination is recommended for people considered to be at risk, including those without a fixed residence, those who use illicit drugs, those who travel internationally, and men who have sex with men.3
There are 3 HAV vaccines available in the United States: 2 single-antigen vaccines, Havrix and Vaqta, both approved for children and adults, and a combination vaccine (containing both HAV and hepatitis B antigens), Twinrix, which is approved for those ages 18 years and older. All are inactivated vaccines.
What you can do. The Virginia outbreak illustrates the important role that family physicians can and do play in public health. We should:
- Encourage adults with risk factors for HAV to be vaccinated.
- Ask those with an HAV diagnosis about the people they may have exposed through personal contact or occupational exposure.
- Promptly report infectious diseases that are designated “reportable” to the public health department.
- Immediately report (by telephone) when HAV and other enteric infections involve a food handler.
In September 2021, a community in Virginia experienced an outbreak of hepatitis A virus (HAV) that was ultimately linked to an infected food handler.1 A total of 149 cases were reported over the next 12 months; 51 were directly related to the food handler and the remainder were the result of sustained community transmission. Of the 51 people who were directly infected by the food handler, 31 were hospitalized and 3 died. This incident offers important reminders about public health surveillance and the role that family physicians can play.
Hepatitis A virus is transmitted through food and drinks that have been contaminated by small amounts of stool that contains the virus or through close contact (including sexual contact) with a person who is infected. The incubation period can range from 15 to 59 days.
HAV generally resolves in a few days to weeks, with no long-term effects. However, recent outbreaks have been associated with high hospitalization and mortality rates because of the underlying comorbidities of those infected.
An increase in incidence. The national rate of HAV infection reached a low of less than 1/100,000 in 2015 but has since increased to almost 6/100,000 in 2019. This increase is mostly due to outbreaks linked to spread among people without a fixed residence, those who use illicit drugs, and men who have sex with men.2
In the Virginia outbreak, the food handler had a risk factor for HAV and was unvaccinated. He worked at 3 different locations of a restaurant chain for a total of 16 days while infectious, preparing ready-to-eat food without using gloves. Furthermore, he delayed seeking medical care for more than 2 weeks—at which time, the nature of his employment was not disclosed.
Prevention is straightforward. HAV infection can be prevented by administration of either HAV vaccine or immune globulin within 2 weeks of exposure.3 During an HAV outbreak, vaccination is recommended for people considered to be at risk, including those without a fixed residence, those who use illicit drugs, those who travel internationally, and men who have sex with men.3
There are 3 HAV vaccines available in the United States: 2 single-antigen vaccines, Havrix and Vaqta, both approved for children and adults, and a combination vaccine (containing both HAV and hepatitis B antigens), Twinrix, which is approved for those ages 18 years and older. All are inactivated vaccines.
What you can do. The Virginia outbreak illustrates the important role that family physicians can and do play in public health. We should:
- Encourage adults with risk factors for HAV to be vaccinated.
- Ask those with an HAV diagnosis about the people they may have exposed through personal contact or occupational exposure.
- Promptly report infectious diseases that are designated “reportable” to the public health department.
- Immediately report (by telephone) when HAV and other enteric infections involve a food handler.
1. Helmick MJ, Morrow CB, White JH, et al. Widespread community transmission of Hepatitis A Virus following an outbreak at a local restaurant—Virginia, September 2021-September 2022. MMWR Morb Mortal Wkly Rep. 2023;72;362-365. doi: 10.15585/mmwr.mm7214a2
2. CDC. Hepatitis A questions and answers for health professionals. Updated July 28, 2020. Accessed April 25, 2023. www.cdc.gov/hepatitis/hav/havfaq.htm
3. Nelson NP, Weng MK, Hofmeister MG, et al. Prevention of hepatitis A virus infection in the United States: recommendations of the Advisory Committee on Immunization Practices, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1-38. doi: 10.15585/mmwr.rr6905a1
1. Helmick MJ, Morrow CB, White JH, et al. Widespread community transmission of Hepatitis A Virus following an outbreak at a local restaurant—Virginia, September 2021-September 2022. MMWR Morb Mortal Wkly Rep. 2023;72;362-365. doi: 10.15585/mmwr.mm7214a2
2. CDC. Hepatitis A questions and answers for health professionals. Updated July 28, 2020. Accessed April 25, 2023. www.cdc.gov/hepatitis/hav/havfaq.htm
3. Nelson NP, Weng MK, Hofmeister MG, et al. Prevention of hepatitis A virus infection in the United States: recommendations of the Advisory Committee on Immunization Practices, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1-38. doi: 10.15585/mmwr.rr6905a1
Skin Diseases Associated With COVID-19: A Narrative Review
COVID-19 is a potentially severe systemic disease caused by SARS-CoV-2. SARS-CoV and Middle East respiratory syndrome (MERS-CoV) caused fatal epidemics in Asia in 2002 to 2003 and in the Arabian Peninsula in 2012, respectively. In 2019, SARS-CoV-2 was detected in patients with severe, sometimes fatal pneumonia of previously unknown origin; it rapidly spread around the world, and the World Health Organization declared the disease a pandemic on March 11, 2020. SARS-CoV-2 is a β-coronavirus that is genetically related to the bat coronavirus and SARS-CoV; it is a single-stranded RNA virus of which several variants and subvariants exist. The SARS-CoV-2 viral particles bind via their surface spike protein (S protein) to the angiotensin-converting enzyme 2 receptor present on the membrane of several cell types, including epidermal and adnexal keratinocytes.1,2 The α and δ variants, predominant from 2020 to 2021, mainly affected the lower respiratory tract and caused severe, potentially fatal pneumonia, especially in patients older than 65 years and/or with comorbidities, such as obesity, hypertension, diabetes, and (iatrogenic) immunosuppression. The ο variant, which appeared in late 2021, is more contagious than the initial variants, but it causes a less severe disease preferentially affecting the upper respiratory airways.3 As of April 5, 2023, more than 762,000,000 confirmed cases of COVID-19 have been recorded worldwide, causing more than 6,800,000 deaths.4
Early studies from China describing the symptoms of COVID-19 reported a low frequency of skin manifestations (0.2%), probably because they were focused on the most severe disease symptoms.5 Subsequently, when COVID-19 spread to the rest of the world, an increasing number of skin manifestations were reported in association with the disease. After the first publication from northern Italy in spring 2020, which was specifically devoted to skin manifestations of COVID-19,6 an explosive number of publications reported a large number of skin manifestations, and national registries were established in several countries to record these manifestations, such as the American Academy of Dermatology and the International League of Dermatological Societies registry,7,8 the COVIDSKIN registry of the French Dermatology Society,9 and the Italian registry.10 Highlighting the unprecedented number of scientific articles published on this new disease, a PubMed search of articles indexed for MEDLINE search using the terms SARS-CoV-2 or COVID-19, on April 6, 2023, revealed 351,596 articles; that is more than 300 articles published every day in this database alone, with a large number of them concerning the skin.
SKIN DISEASSES ASSOCIATED WITH COVID-19
There are several types of COVID-19–related skin manifestations, depending on the circumstances of onset and the evolution of the pandemic.
Skin Manifestations Associated With SARS-CoV-2 Infection
The estimated incidence varies greatly according to the published series of patients, possibly depending on the geographic location. The estimated incidence seems lower in Asian countries, such as China (0.2%)5 and Japan (0.56%),11 compared with Europe (up to 20%).6 Skin manifestations associated with SARS-CoV-2 infection affect individuals of all ages, slightly more females, and are clinically polymorphous; some of them are associated with the severity of the infection.12 They may precede, accompany, or appear after the symptoms of COVID-19, most often within a month of the infection, of which they rarely are the only manifestation; however, their precise relationship to SARS-CoV-2 is not always well known. They have been classified according to their clinical presentation into several forms.13-15
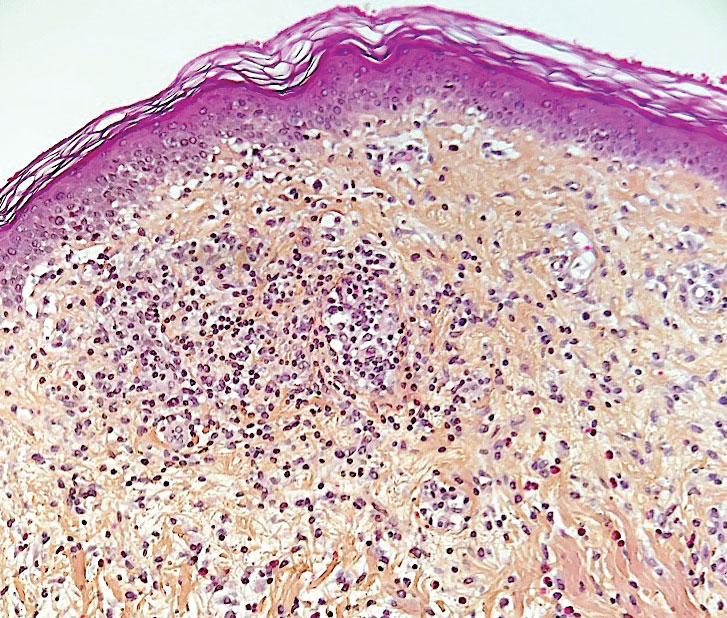
Morbilliform Maculopapular Eruption—Representing 16% to 53% of skin manifestations, morbilliform and maculopapular eruptions usually appear within 15 days of infection; they manifest with more or less confluent erythematous macules that may be hemorrhagic/petechial, and usually are asymptomatic and rarely pruritic. The rash mainly affects the trunk and limbs, sparing the face, palmoplantar regions, and mucous membranes; it appears concomitantly with or a few days after the first symptoms of COVID-19 (eg, fever, respiratory symptoms), regresses within a few days, and does not appear to be associated with disease severity. The distinction from maculopapular drug eruptions may be subtle. Histologically, the rash manifests with a spongiform dermatitis (ie, variable parakeratosis; spongiosis; and a mixed dermal perivascular infiltrate of lymphocytes, eosinophils and histiocytes, depending on the lesion age)(Figure 1). The etiopathogenesis is unknown; it may involve immune complexes to SARS-CoV-2 deposited on skin vessels. Treatment is not mandatory; if necessary, local or systemic corticosteroids may be used.
Vesicular (Pseudovaricella) Rash—This rash accounts for 11% to 18% of all skin manifestations and usually appears within 15 days of COVID-19 onset. It manifests with small monomorphous or varicellalike (pseudopolymorphic) vesicles appearing on the trunk, usually in young patients. The vesicles may be herpetiform, hemorrhagic, or pruritic, and appear before or within 3 days of the onset of mild COVID-19 symptoms; they regress within a few days without scarring. Histologically, the lesions show basal cell vacuolization; multinucleated, dyskeratotic/apoptotic or ballooning/acantholytic epidermal keratinocytes; reticular degeneration of the epidermis; intraepidermal vesicles sometimes resembling herpetic vesicular infections or Grover disease; and mild dermal inflammation. There is no specific treatment.
Urticaria—Urticarial rash, or urticaria, represents 5% to 16% of skin manifestations; usually appears within 15 days of disease onset; and manifests with pruritic, migratory, edematous papules appearing mainly on the trunk and occasionally the face and limbs. The urticarial rash tends to be associated with more severe forms of the disease and regresses within a week, responding to antihistamines. Of note, clinically similar rashes can be caused by drugs. Histologically, the lesions show dermal edema and a mild perivascular lymphocytic infiltrate, sometimes admixed with eosinophils.
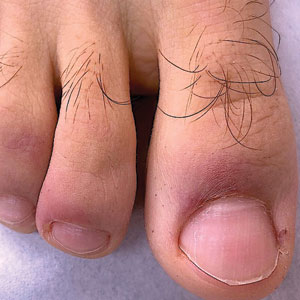
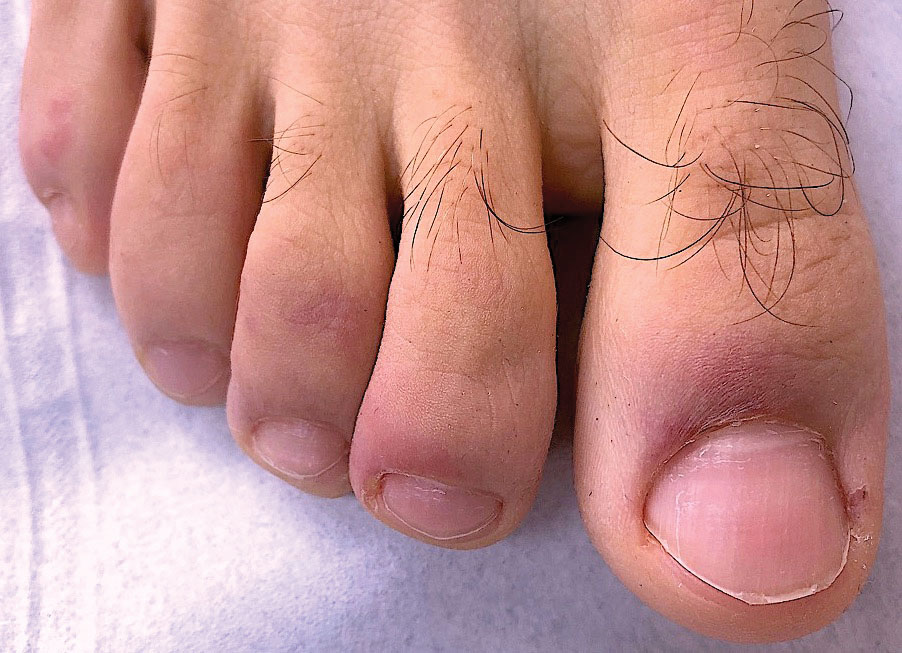
Chilblainlike Lesions—Chilblainlike lesions (CBLLs) account for 19% of skin manifestations associated with COVID-1913 and present as erythematous-purplish, edematous lesions that can be mildly pruritic or painful, appearing on the toes—COVID toes—and more rarely the fingers (Figure 2). They were seen epidemically during the first pandemic wave (2020 lockdown) in several countries, and clinically are very similar to, if not indistinguishable from, idiopathic chilblains, but are not necessarily associated with cold exposure. They appear in young, generally healthy patients or those with mild COVID-19 symptoms 2 to 4 weeks after symptom onset. They regress spontaneously or under local corticosteroid treatment within a few days or weeks. Histologically, CBLLs are indistinguishable from chilblains of other origins, namely idiopathic (seasonal) ones. They manifest with necrosis of epidermal keratinocytes; dermal edema that may be severe, leading to the development of subepidermal pseudobullae; a rather dense perivascular and perieccrine gland lymphocytic infiltrate; and sometimes with vascular lesions (eg, edema of endothelial cells, microthromboses of dermal capillaries and venules, fibrinoid deposits within the wall of dermal venules)(Figure 3).16-18 Most patients (>80%) with CBLLs have negative serologic or polymerase chain reaction tests for SARS-CoV-2,19 which generated a lively debate about the role of SARS-CoV-2 in the genesis of CBLLs. According to some authors, SARS-CoV-2 plays no direct role, and CBLLs would occur in young people who sit or walk barefoot on cold floors at home during confinement.20-23 Remarkably, CBLLs appeared in patients with no history of chilblains during a season that was not particularly cold, namely in France or in southern California, where their incidence was much higher compared to the same time period of prior years. Some reports have supported a direct role for the virus based on questionable observations of the virus within skin lesions (eg, sweat glands, endothelial cells) by immunohistochemistry, electron microscopy, and/or in situ hybridization.17,24,25 A more satisfactory hypothesis would involve the role of a strong innate immunity leading to elimination of the virus before the development of specific antibodies via the increased production of type 1 interferon (IFN-1); this would affect the vessels, causing CBLLs. This mechanism would be similar to the one observed in some interferonopathies (eg, Aicardi-Goutières syndrome), also characterized by IFN-1 hypersecretion and chilblains.26-29 According to this hypothesis, CBLLs should be considered a paraviral rash similar to other skin manifestations associated with COVID-19.30
Acro-ischemia—Acro-ischemia livedoid lesions account for 1% to 6% of skin manifestations and comprise lesions of livedo (either reticulated or racemosa); necrotic acral bullae; and gangrenous necrosis of the extremities, especially the toes. The livedoid lesions most often appear within 15 days of COVID-19 symptom onset, and the purpuric lesions somewhat later (2–4 weeks); they mainly affect adult patients, last about 10 days, and are the hallmark of severe infection, presumably related to microthromboses of the cutaneous capillaries (endothelial dysfunction, prothrombotic state, elevated D-dimers). Histologically, they show capillary thrombosis and dermoepidermal necrosis (Figure 4).
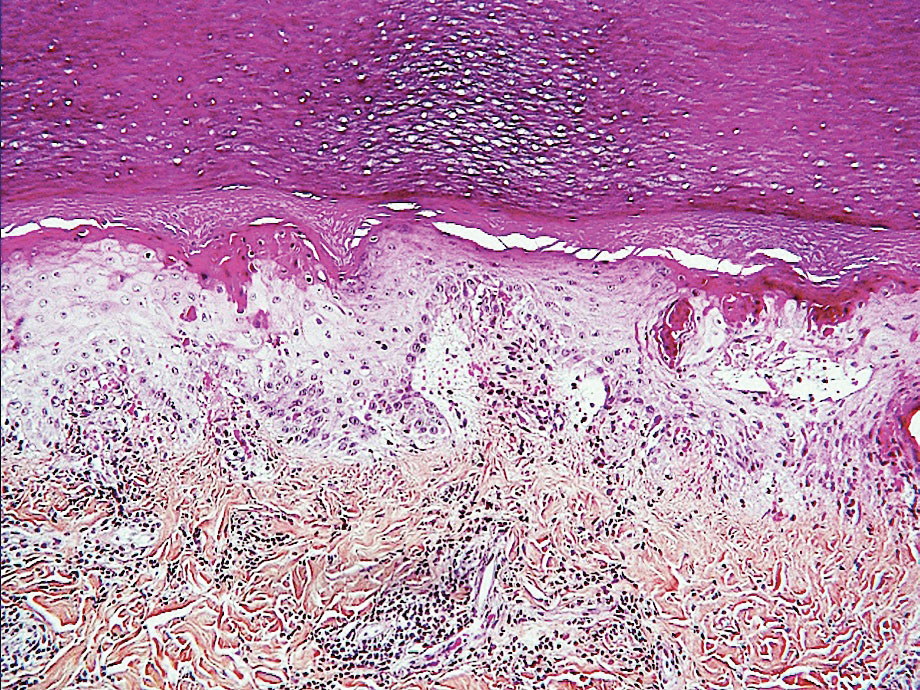
Other Reported Polymorphic or Atypical Rashes—Erythema multiforme–like eruptions may appear before other COVID-19 symptoms and manifest as reddish-purple, nearly symmetric, diffuse, occasionally targetoid bullous or necrotic macules. The eruptions mainly affect adults and most often are seen on the palms, elbows, knees, and sometimes the mucous membranes. The rash regresses in 1 to 3 weeks without scarring and represents a delayed cutaneous hypersensitivity reaction. Histologically, the lesions show vacuolization of basal epidermal keratinocytes, keratinocyte necrosis, dermoepidermal detachment, a variably dense dermal T-lymphocytic infiltrate, and red blood cell extravasation (Figure 5).
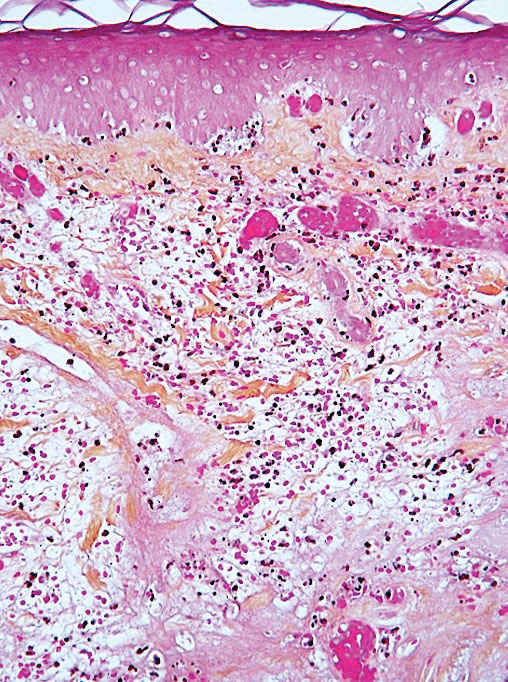
Leukocytoclastic vasculitis may be generalized or localized. It manifests clinically by petechial/purpuric maculopapules, especially on the legs, mainly in elderly patients with COVID-19. Histologically, the lesions show necrotizing changes of dermal postcapillary venules, neutrophilic perivascular inflammation, red blood cell extravasation, and occasionally vascular IgA deposits by direct immunofluorescence examination. The course usually is benign.
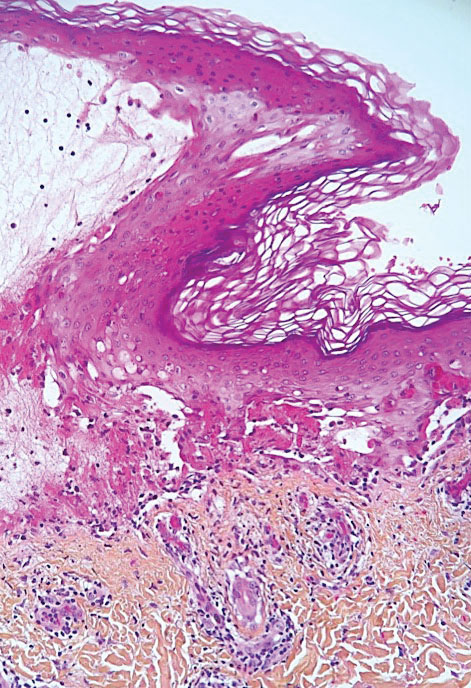
The incidence of pityriasis rosea and of clinically similar rashes (referred to as “pityriasis rosea–like”) increased 5-fold during the COVID-19 pandemic.31,32 These dermatoses manifest with erythematous, scaly, circinate plaques, typically with an initial herald lesion followed a few days later by smaller erythematous macules. Histologically, the lesions comprise a spongiform dermatitis with intraepidermal exocytosis of red blood cells and a mild to moderate dermal lymphocytic infiltrate.
Erythrodysesthesia, or hand-foot syndrome, manifests with edematous erythema and palmoplantar desquamation accompanied by a burning sensation or pain. This syndrome is known as an adverse effect of some chemotherapies because of the associated drug toxicity and sweat gland inflammation; it was observed in 40% of 666 COVID-19–positive patients with mild to moderate pneumonitis.33
“COVID nose” is a rare cutaneous manifestation characterized by nasal pigmentation comprising multiple coalescent frecklelike macules on the tip and wings of the nose and sometimes the malar areas. These lesions predominantly appear in women aged 25 to 65 years and show on average 23 days after onset of COVID-19, which is usually mild. This pigmentation is similar to pigmentary changes after infection with chikungunya; it can be treated with depigmenting products such as azelaic acid and hydroquinone cream with sunscreen use, and it regresses in 2 to 4 months.34
Telogen effluvium (excessive and temporary shedding of normal telogen club hairs of the entire scalp due to the disturbance of the hair cycle) is reportedly frequent in patients (48%) 1 month after COVID-19 infection, but it may appear later (after 12 weeks).35 Alopecia also is frequently reported during long (or postacute) COVID-19 (ie, the symptomatic disease phase past the acute 4 weeks’ stage of the infection) and shows a female predominance36; it likely represents the telogen effluvium seen 90 days after a severe illness. Trichodynia (pruritus, burning, pain, or paresthesia of the scalp) also is reportedly common (developing in more than 58% of patients) and is associated with telogen effluvium in 44% of cases. Several cases of alopecia areata (AA) triggered or aggravated by COVID-19 also have been reported37,38; they could be explained by the “cytokine storm” triggered by the infection, involving T and B lymphocytes; plasmacytoid dendritic cells; natural killer cells with oversecretion of IL-6, IL-4, tumor necrosis factor α, and IFN type I; and a cytotoxic reaction associated with loss of the immune privilege of hair follicles.
Nail Manifestations
The red half-moon nail sign is an asymptomatic purplish-red band around the distal margin of the lunula that affects some adult patients with COVID-19.39 It appears shortly after onset of symptoms, likely the manifestation of vascular inflammation in the nail bed, and regresses slowly after approximately 1 week.40 Beau lines are transverse grooves in the nail plate due to the temporary arrest of the proximal nail matrix growth accompanying systemic illnesses; they appear approximately 2 to 3 weeks after the onset of COVID-19.41 Furthermore, nail alterations can be caused by drugs used to treat COVID-19, such as longitudinal melanonychia due to treatment with hydroxychloroquine or fluorescence of the lunula or nail plate due to treatment with favipiravir.42
Multisystem Inflammatory Syndrome
Multisystem inflammatory syndrome (MIS) is clinically similar to Kawasaki disease; it typically affects children43 and more rarely adults with COVID-19. It manifests with fever, weakness, and biological inflammation and also frequently with skin lesions (72%), which are polymorphous and include morbilliform rash (27%); urticaria (24%); periorbital edema (24%); nonspecific erythema (21.2%); retiform purpura (18%); targetoid lesions (15%); malar rash (15.2%); and periareolar erythema (6%).44 Compared to Kawasaki disease, MIS affects slightly older children (mean age, 8.5 vs 3 years) and more frequently includes cardiac and gastrointestinal manifestations; the mortality rate also is slightly higher (2% vs 0.17%).45
Confirmed COVID-19 Infection
At the beginning of the pandemic, skin manifestations were reported in patients who were suspected of having COVID-19 but did not always have biological confirmation of SARS-CoV-2 infection due to the unavailability of diagnostic tests or the physical impossibility of testing. However, subsequent studies have confirmed that most of these dermatoses were indeed associated with COVID-19 infection.9,46 For example, a study of 655 patients with confirmed COVID-19 infection reported maculopapular (38%), vascular (22%), urticarial (15%), and vesicular (15%) rashes; erythema multiforme or Stevens-Johnson–like syndrome (3%, often related to the use of hydroxychloroquine); generalized pruritus (1%); and MIS (0.5%). The study confirmed that CBLLs were mostly seen in young patients with mild disease, whereas livedo (fixed rash) and retiform purpura occurred in older patients with a guarded prognosis.46
Remarkably, most dermatoses associated with SARS-CoV-2 infection were reported during the initial waves of the pandemic, which were due to the α and δ viral variants. These manifestations were reported more rarely when the ο variant was predominant, even though most patients (63%) who developed CBLLs in the first wave also developed them during the second pandemic wave.47 This decrease in the incidence of COVID-19–associated dermatoses could be because of the lower pathogenicity of the o variant,3 a lower tropism for the skin, and variations in SARS-CoV-2 antigenicity that would induce a different immunologic response, combined with an increasingly stronger herd immunity compared to the first pandemic waves achieved through vaccination and spontaneous infections in the population. Additional reasons may include different baseline characteristics in patients hospitalized with COVID-19 (regarding comorbidities, disease severity, and received treatments), and the possibility that some of the initially reported COVID-19–associated skin manifestations could have been produced by different etiologic agents.48 In the last 2 years, COVID-19–related skin manifestations have been reported mainly as adverse events to COVID-19 vaccination.
CUTANEOUS ADVERSE EFFECTS OF DRUGS USED TO TREAT COVID-19
Prior to the advent of vaccines and specific treatments for SARS-CoV-2, various drugs were used—namely hydroxychloroquine, ivermectin, and tocilizumab—that did not prove efficacious and caused diverse adverse effects, including cutaneous eruptions such as urticaria, maculopapular eruptions, erythema multiforme or Stevens-Johnson syndrome, vasculitis, longitudinal melanonychia, and acute generalized exanthematous pustulosis.49,50 Nirmatrelvir 150 mg–ritonavir 100 mg, which was authorized for emergency use by the US Food and Drug Administration for the treatment of COVID-19, is a viral protease inhibitor blocking the replication of the virus. Ritonavir can induce pruritus, maculopapular rash, acne, Stevens-Johnson syndrome, and toxic epidermal necrolysis; of note, these effects have been observed following administration of ritonavir for treatment of HIV at higher daily doses and for much longer periods of time compared with treatment of COVID-19 (600–1200 mg vs 200 mg/d, respectively). These cutaneous drug side effects are clinically similar to the manifestations caused either directly or indirectly by SARS-CoV-2 infection; therefore, it may be difficult to differentiate them.
DERMATOSES DUE TO PROTECTIVE DEVICES
Dermatoses due to personal protective equipment such as masks or face shields affected the general population and mostly health care professionals51; 54.4% of 879 health care professionals in one study reported such events.52 These dermatoses mainly include contact dermatitis of the face (nose, forehead, and cheeks) of irritant or allergic nature (eg, from preservatives releasing formaldehyde contained in masks and protective goggles). They manifest with skin dryness; desquamation; maceration; fissures; or erosions or ulcerations of the cheeks, forehead, and nose. Cases of pressure urticaria also have been reported. Irritant dermatitis induced by the frequent use of disinfectants (eg, soaps, hydroalcoholic sanitizing gels) also can affect the hands. Allergic hand dermatitis can be caused by medical gloves.
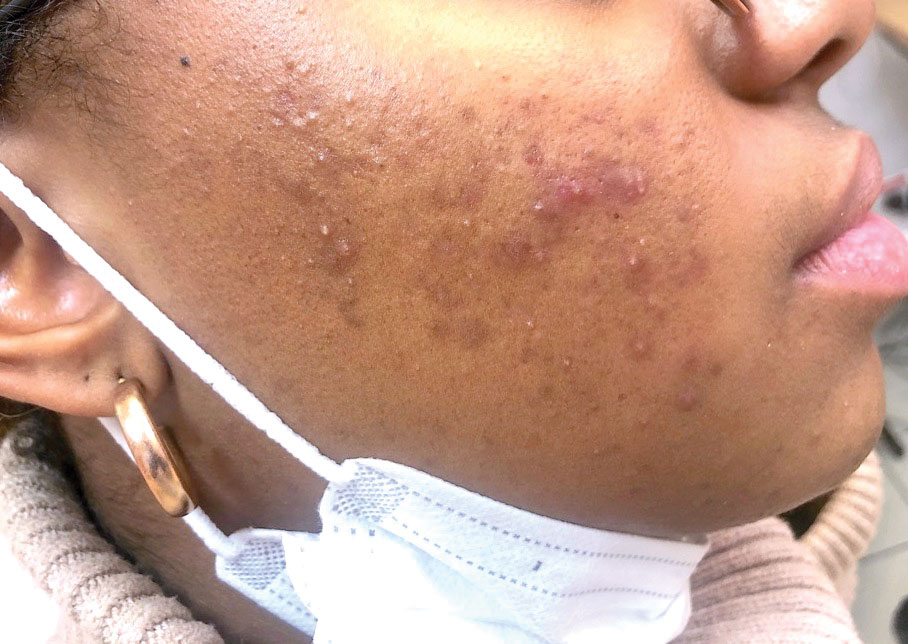
The term maskne (or mask acne) refers to a variety of mechanical acne due to the prolonged use of surgical masks (>4 hours per day for ≥6 weeks); it includes cases of de novo acne and cases of pre-existing acne aggravated by wearing a mask. Maskne is characterized by acne lesions located on the facial area covered by the mask (Figure 6). It is caused by follicular occlusion; increased sebum secretion; mechanical stress (pressure, friction); and dysbiosis of the microbiome induced by changes in heat, pH, and humidity. Preventive measures include application of noncomedogenic moisturizers or gauze before wearing the mask as well as facial cleansing with appropriate nonalcoholic products. Similar to acne, rosacea often is aggravated by prolonged wearing of surgical masks (mask rosacea).53,54
DERMATOSES REVEALED OR AGGRAVATED BY COVID-19
Exacerbation of various skin diseases has been reported after infection with SARS-CoV-2.55 Psoriasis and acrodermatitis continua of Hallopeau,56 which may progress into generalized, pustular, or erythrodermic forms,57 have been reported; the role of hydroxychloroquine and oral corticosteroids used for the treatment of COVID-19 has been suspected.57 Atopic dermatitis patients—26% to 43%—have experienced worsening of their disease after symptomatic COVID-19 infection.58 The incidence of herpesvirus infections, including herpes zoster, increased during the pandemic.59 Alopecia areata relapses occurred in 42.5% of 392 patients with preexisting disease within 2 months of COVID-19 onset in one study,60 possibly favored by the psychological stress; however, some studies have not confirmed the aggravating role of COVID-19 on alopecia areata.61 Lupus erythematosus, which may relapse in the form of Rowell syndrome,62 and livedoid vasculopathy63 also have been reported following COVID-19 infection.
SKIN MANIFESTATIONS ASSOCIATED WITH COVID-19 VACCINES
In parallel with the rapid spread of COVID-19 vaccination,4 an increasing number of skin manifestations has been observed following vaccination; these dermatoses now are more frequently reported than those related to natural SARS-CoV-2 infection.64-70 Vaccine-induced skin manifestations have a reported incidence of approximately 4% and show a female predominance.65 Most of them (79%) have been reported in association with messenger RNA (mRNA)–based vaccines, which have been the most widely used; however, the frequency of side effects would be lower after mRNA vaccines than after inactivated virus-based vaccines. Eighteen percent occurred after the adenoviral vector vaccine, and 3% after the inactivated virus vaccine.70 Fifty-nine percent were observed after the first dose. They are clinically polymorphous and generally benign, regressing spontaneously after a few days, and they should not constitute a contraindication to vaccination.Interestingly, many skin manifestations are similar to those associated with natural SARS-CoV-2 infection; however, their frequency and severity does not seem to depend on whether the patients had developed skin reactions during prior SARS-CoV-2 infection. These reactions have been classified into several types:
• Immediate local reactions at the injection site: pain, erythema, or edema represent the vast majority (96%) of reactions to vaccines. They appear within 7 days after vaccination (average, 1 day), slightly more frequently (59%) after the first dose. They concern mostly young patients and are benign, regressing in 2 to 3 days.70
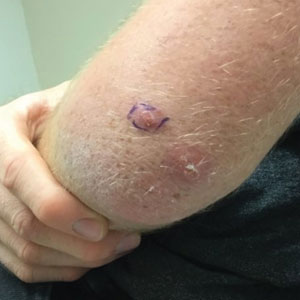
• Delayed local reactions: characterized by pain or pruritus, erythema, and skin induration mimicking cellulitis (COVID arm) and represent 1.7% of postvaccination reactions. They correspond to a delayed hypersensitivity reaction and appear approximately 7 days after vaccination, most often after the first vaccine dose (75% of cases), which is almost invariably mRNA based.70
• Urticarial reactions corresponding to an immediate (type 1) hypersensitivity reaction: constitute 1% of postvaccination reactions, probably due to an allergy to vaccine ingredients. They appear on average 1 day after vaccination, almost always with mRNA vaccines.70
• Angioedema: characterized by mucosal or subcutaneous edema and constitutes 0.5% of postvaccination reactions. It is a potentially serious reaction that appears on average 12 hours after vaccination, always with an mRNA-based vaccine.70
• Morbilliform rash: represents delayed hypersensitivity reactions (0.1% of postvaccination reactions) that appear mostly after the first dose (72%), on average 3 days after vaccination, always with an mRNA-based vaccine.70
• Herpes zoster: usually develops after the first vaccine dose in elderly patients (69% of cases) on average 4 days after vaccination and constitutes 0.1% of postvaccination reactions.71
• Bullous diseases: mainly bullous pemphigoid (90%) and more rarely pemphigus (5%) or bullous erythema pigmentosum (5%). They appear in elderly patients on average 7 days after vaccination and constitute 0.04% of postvaccination reactions.72
• Chilblainlike lesions: several such cases have been reported so far73; they constitute 0.03% of postvaccination reactions.70 Clinically, they are similar to those associated with natural COVID-19; they appear mostly after the first dose (64%), on average 5 days after vaccination with the mRNA or adenovirus vaccine, and show a female predominance. The appearance of these lesions in vaccinated patients, who are a priori not carriers of the virus, strongly suggests that CBLLs are due to the immune reaction against SARS-CoV-2 rather than to a direct effect of this virus on the skin, which also is a likely scenario with regards to other skin manifestations seen during the successive COVID-19 epidemic waves.73-75
• Reactions to hyaluronic acid–containing cosmetic fillers: erythema, edema, and potentially painful induration at the filler injection sites. They constitute 0.04% of postvaccination skin reactions and appear 24 hours after vaccination with mRNA-based vaccines, equally after the first or second dose.76
• Pityriasis rosea–like rash: most occur after the second dose of mRNA-based vaccines (0.023% of postvaccination skin reactions).70
• Severe reactions: these include acute generalized exanthematous pustulosis77 and Stevens-Johnson syndrome.78 One case of each has been reported after the adenoviral vector vaccine 3 days after vaccination.
Other more rarely observed manifestations include reactivation/aggravation or de novo appearance of inflammatory dermatoses such as psoriasis,79,80 leukocytoclastic vasculitis,81,82 lymphocytic83 or urticarial84 vasculitis, Sweet syndrome,85 lupus erythematosus, dermatomyositis,86,87 alopecia,37,88 infection with Trichophyton rubrum,89 Grover disease,90 and lymphomatoid reactions (such as recurrences of cutaneous T-cell lymphomas [CD30+], and de novo development of lymphomatoid papulosis).91
FINAL THOUGHTS
COVID-19 is associated with several skin manifestations, even though the causative role of SARS-CoV-2 has remained elusive. These dermatoses are highly polymorphous, mostly benign, and usually spontaneously regressive, but some of them reflect severe infection. They mostly were described during the first pandemic waves, reported in several national and international registries, which allowed for their morphological classification. Currently, cutaneous adverse effects of vaccines are the most frequently reported dermatoses associated with SARS-CoV-2, and it is likely that they will continue to be observed while COVID-19 vaccination lasts. Hopefully the end of the COVID-19 pandemic is near. In January 2023, the International Health Regulations Emergency Committee of the World Health Organization acknowledged that the COVID-19 pandemic may be approaching an inflexion point, and even though the event continues to constitute a public health emergency of international concern, the higher levels of population immunity achieved globally through infection and/or vaccination may limit the impact of SARS-CoV-2 on morbidity and mortality. However, there is little doubt that this virus will remain a permanently established pathogen in humans and animals for the foreseeable future.92 Therefore, physicians—especially dermatologists—should be aware of the various skin manifestations associated with COVID-19 so they can more efficiently manage their patients.
- Ashraf UM, Abokor AA, Edwards JM, et al. SARS-CoV-2, ACE2 expression, and systemic organ invasion. Physiol Genomics. 2021;53:51-60.
- Ganier C, Harun N, Peplow I, et al. Angiotensin-converting enzyme 2 expression is detectable in keratinocytes, cutaneous appendages, and blood vessels by multiplex RNA in situ hybridization. Adv Skin Wound Care. 2022;35:219-223.
- Ulloa AC, Buchan SA, Daneman N, et al. Estimates of SARS-CoV-2 omicron variant severity in Ontario, Canada. JAMA. 2022;327:1286-1288.
- World Health Organization. Coronavirus (COVID-19) Dashboard. Accessed April 6, 2023. https://covid19.who.int
- Guan WJ, Ni ZY, Hu Y, et al; China Medical Treatment Expert Group for COVID-19. clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708-1720.
- Recalcati S. Cutaneous manifestations in COVID-9: a first perspective. J Eur Acad Dermatol Venereol. 2020;34:E212-E213.
- Freeman EE, McMahon DE, Lipoff JB, et al. The spectrum of COVID-19-associated dermatologic manifestations: an international registry of 716 patients from 31 countries. J Am Acad Dermatol. 2020;83:1118-1129.
- Freeman EE, Chamberlin GC, McMahon DE, et al. Dermatology COVID-19 registries: updates and future directions. Dermatol Clin. 2021;39:575-585.
- Guelimi R, Salle R, Dousset L, et al. Non-acral skin manifestations during the COVID-19 epidemic: COVIDSKIN study by the French Society of Dermatology. J Eur Acad Dermatol Venereol. 2021;35:E539-E541.
- Marzano AV, Genovese G, Moltrasio C, et al; Italian Skin COVID-19 Network of the Italian Society of Dermatology and Sexually Transmitted Diseases. The clinical spectrum of COVID-19 associated cutaneous manifestations: an Italian multicenter study of 200 adult patients. J Am Acad Dermatol. 2021;84:1356-1363.
- Sugai T, Fujita Y, Inamura E, et al. Prevalence and patterns of cutaneous manifestations in 1245 COVID-19 patients in Japan: a single-centre study. J Eur Acad Dermatol Venereol. 2022;36:E522-E524.
- Holmes Z, Courtney A, Lincoln M, et al. Rash morphology as a predictor of COVID‐19 severity: a systematic review of the cutaneous manifestations of COVID‐19. Skin Health Dis. 2022;2:E120. doi:10.1002/ski2.120
- Galván Casas C, Català A, Carretero Hernández G, et al. Classification of the cutaneous manifestations of COVID-19: a rapid prospective nationwide consensus study in Spain with 375 cases. Br J Dermatol. 2020;183:71-77.
- Garduño‑Soto M, Choreño-Parra, Cazarin-Barrientos Dermatological aspects of SARS‑CoV‑2 infection: mechanisms and manifestations. Arch Dermatol Res. 2021;313:611-622.
- Huynh T, Sanchez-Flores X, Yau J, et al. Cutaneous manifestations of SARS-CoV-2 Infection. Am J Clin Dermatol. 2022;23:277-286.
- Kanitakis J, Lesort C, Danset M, et al.
2020; 83:870-875. - Kolivras A, Thompson C, Pastushenko I, et al. A clinicopathological description of COVID-19-induced chilblains (COVID-toes) correlated with a published literature review. J Cutan Pathol. 2022;49:17-28.
- Roca-Ginés J, Torres-Navarro I, Sánchez-Arráez J, et al. Assessment of acute acral lesions in a case series of children and adolescents during the COVID-19 pandemic. 2020;156:992-997.
- Le Cleach L, Dousset L, Assier H, et al; French Society of Dermatology. Most chilblains observed during the COVID-19 outbreak occur in patients who are negative for COVID-19 on polymerase chain reaction and serology testing. Br J Dermatol. 2020;183:866-874.
- Discepolo V, Catzola A, Pierri L, et al. Bilateral chilblain-like lesions of the toes characterized by microvascular remodeling in adolescents during the COVID-19 pandemic. JAMA Netw Open. 2021;4:E2111369.
- Gehlhausen JR, Little AJ, Ko CJ, et al. Lack of association between pandemic chilblains and SARS-CoV-2 infection. Proc Natl Acad Sci U S A. 2022;119:e2122090119.
- Neri, Virdi, Corsini, et al Major cluster of paediatric ‘true’ primary chilblains during the COVID-19 pandemic: a consequence of lifestyle changes due to lockdown. J Eur Acad Dermatol Venereol. 2020;34:2630-2635.
- De Greef A, Choteau M, Herman A, et al. Chilblains observed during the COVID-19 pandemic cannot be distinguished from the classic, cold-related chilblains. Eur J Dermatol. 2022;32:377-383.
- Colmenero I, Santonja C, Alonso-Riaño M, et al. SARS-CoV-2 endothelial infection causes COVID-19 chilblains: histopathological, immunohistochemical and ultrastructural study of seven paediatric cases. Br J Dermatol. 2020;183:729-737.
- Quintero-Bustos G, Aguilar-Leon D, Saeb-Lima M. Histopathological and immunohistochemical characterization of skin biopsies from 41 SARS-CoV-2 (+) patients: experience in a Mexican concentration institute: a case series and literature review. Am J Dermatopathol. 2022;44:327-337.
- Arkin LM, Moon JJ, Tran JM, et al; COVID Human Genetic Effort. From your nose to your toes: a review of severe acute respiratory syndrome coronavirus 2 pandemic-associated pernio. J Invest Dermatol. 2021;141:2791-2796.
- Frumholtz L, Bouaziz JD, Battistella M, et al; Saint-Louis CORE (COvid REsearch). Type I interferon response and vascular alteration in chilblain-like lesions during the COVID-19 outbreak. Br J Dermatol. 2021;185:1176-1185.
- Hubiche T, Cardot-Leccia N, Le Duff F, et al. Clinical, laboratory, and interferon-alpha response characteristics of patients with chilblain-like lesions during the COVID-19 pandemic. JAMA Dermatol. 2021;157:202-206.
- Lesort C, Kanitakis J, Villani A, et al. COVID-19 and outbreak of chilblains: are they related? J Eur Acad Dermatol Venereol. 2020;34:E757-E758.
- Sanchez A, Sohier P, Benghanem S, et al. Digitate papulosquamous eruption associated with severe acute respiratory syndrome coronavirus 2 infection. JAMA Dermatol. 2020;156:819-820.
- Drago F, Broccolo F, Ciccarese G. Pityriasis rosea, pityriasis rosea-like eruptions, and herpes zoster in the setting of COVID-19 and COVID-19 vaccination. Clin Dermatol. 2022;S0738-081X(22)00002-5.
- Dursun R, Temiz SA. The clinics of HHV-6 infection in COVID-19 pandemic: pityriasis rosea and Kawasaki disease. Dermatol Ther. 2020;33:E13730.
- Nuno-Gonzalez A, Magaletsky K, Feito Rodríguez M, et al. Palmoplantar erythrodysesthesia: a diagnostic sign of COVID-19. J Eur Acad Dermatol Venereol. 2021;35:e247-e249.
- Sil A, Panigrahi A, Chandra A, et al. “COVID nose”: a unique post-COVID pigmentary sequelae reminiscing Chik sign: a descriptive case series. J Eur Acad Dermatol Venereol. 2022;36:E419-E421.
- Starace M, Iorizzo M, Sechi A, et al. Trichodynia and telogen effluvium in COVID-19 patients: results of an international expert opinion survey on diagnosis and management. JAAD Int. 2021;5:11-18.
- Wong-Chew RM, Rodríguez Cabrera EX, Rodríguez Valdez CA, et al. Symptom cluster analysis of long COVID-19 in patients discharged from the Temporary COVID-19 Hospital in Mexico City. Ther Adv Infect Dis. 2022;9:20499361211069264.
- Bardazzi F, Guglielmo A, Abbenante D, et al. New insights into alopecia areata during COVID-19 pandemic: when infection or vaccination could play a role. J Cosmet Dermatol. 2022;21:1796-1798.
- Christensen RE, Jafferany M. Association between alopecia areata and COVID-19: a systematic review. JAAD Int. 2022;7:57-61.
- Wollina U, Kanitakis J, Baran R. Nails and COVID-19: a comprehensive review of clinical findings and treatment. Dermatol Ther. 2021;34:E15100.
- Méndez-Flores S, Zaladonis A, Valdes-Rodriguez R. COVID-19 and nail manifestation: be on the lookout for the red half-moon nail sign. Int J Dermatol. 2020;59:1414.
- Alobaida S, Lam JM. Beau lines associated with COVID-19. CMAJ. 2020;192:E1040.
- Durmaz EÖ, Demirciog˘lu D. Fluorescence in the sclera, nails, and teeth secondary to favipiravir use for COVID-19 infections. J Clin Aesthet Dermatol. 2022;15:35-37.
- Brumfiel CM, DiLorenzo AM, Petronic-Rosic VM. Dermatologic manifestations of COVID-19-associated multisystem inflammatory syndrome in children. Clin Dermatol. 2021;39:329-333.
- Akçay N, Topkarcı Z, Menentog˘lu ME, et al. New dermatological findings of MIS-C: can mucocutaneous involvement be associated with severe disease course? Australas J Dermatol. 2022;63:228-234. doi:10.1111/ajd.13819
- Vogel TP, Top KA, Karatzios C, et al. Multisystem inflammatory syndrome in children and adults (MIS-C/A): case definition & guidelines for data collection, analysis, and presentation of immunization safety data. Vaccine. 2021;39:3037-3049.
- Conforti C, Dianzani C, Agozzino M, et al. Cutaneous manifestations in confirmed COVID-19 patients: a systematic review. Biology (Basel). 2020;9:449.
- Hubiche T, Le Duff F, Fontas E, et al. Relapse of chilblain-like lesions during the second wave of the COVID-19 pandemic: a cohort follow-up. Br J Dermatol. 2021;185:858-859.
- Fernandez-Nieto, Ortega-Quijano, Suarez-Valle, et al Lack of skin manifestations in COVID-19 hospitalized patients during the second epidemic wave in Spain: a possible association with a novel SARS-CoV-2 variant: a cross-sectional study. J Eur Acad Dermatol Venereol. 2021;35:E183-E185.
- Martinez-LopezA, Cuenca-Barrales, Montero-Vilchezet al Review of adverse cutaneous reactions of pharmacologic interventions for COVID-19: a guide for the dermatologist. J Am Acad Dermatol. 2020;83:1738-1748.
- Cutaneous side-effects of the potential COVID-19 drugs. Dermatol Ther. 2020;33:E13476.
- Mawhirt SL, Frankel D, Diaz AM. Cutaneous manifestations in adult patients with COVID-19 and dermatologic conditions related to the COVID-19 pandemic in health care workers. Curr Allerg Asthma Rep. 2020;20:75.
- Nguyen C, Young FG, McElroy D, et al. Personal protective equipment and adverse dermatological reactions among healthcare workers: survey observations from the COVID-19 pandemic. Medicine (Baltimore). 2022;101:E29003.
- Rathi SK, Dsouza JM. Maskne: a new acne variant in COVID-19 era. Indian J Dermatol. 2022;67:552-555.
- Damiani G, Girono L, Grada A, et al. COVID-19 related masks increase severity of both acne (maskne) and rosacea (mask rosacea): multi-center, real-life, telemedical, and observational prospective study. Dermatol Ther. 2021;34:E14848.
- Aram K, Patil A, Goldust M, et al. COVID-19 and exacerbation of dermatological diseases: a review of the available literature. Dermatol Ther. 2021;34:E15113.
- Samotij D, Gawron E, Szcze˛ch J, et al. Acrodermatitis continua of Hallopeau evolving into generalized pustular psoriasis following COVID-19: a case report of a successful treatment with infliximab in combination with acitretin. Biologics. 2021;15:107-113.
- Demiri J, Abdo M, Tsianakas A. Erythrodermic psoriasis after COVID-19 [in German]. Hautarzt. 2022;73:156-159.
- de Wijs LEM, Joustra MM, Olydam JI, et al. COVID-19 in patients with cutaneous immune-mediated diseases in the Netherlands: real-world observational data. J Eur Acad Dermatol Venereol. 2021;35:E173-E176.
- Marques NP, Maia CMF, Marques NCT, et al. Continuous increase of herpes zoster cases in Brazil during the COVID-19 pandemic. Oral Surg Oral Med Oral Pathol Oral Radiol. 2022;133:612-614.
- Rinaldi F, Trink A, Giuliani G, et al. Italian survey for the evaluation of the effects of coronavirus disease 2019 (COVID-19) pandemic on alopecia areata recurrence. Dermatol Ther (Heidelb). 2021;11:339-345.
- Rudnicka L, Rakowska A, Waskiel-Burnat A, et al. Mild-to-moderate COVID-19 is not associated with worsening of alopecia areata: a retrospective analysis of 32 patients. J Am Acad Dermatol. 2021;85:723-725.
- Drenovska K, Shahid M, Mateeva V, et al. Case report: Rowell syndrome-like flare of cutaneous lupus erythematosus following COVID-19 infection. Front Med (Lausanne). 2022;9:815743.
- Kawabe R, Tonomura K, Kotobuki Y, et al. Exacerbation of livedoid vasculopathy after coronavirus disease 2019. Eur J Dermatol. 2022;32:129-131. doi:10.1684/ejd.2022.4200
- McMahon DE, Kovarik CL, Damsky W, et al. Clinical and pathologic correlation of cutaneous COVID-19 vaccine reactions including V-REPP: a registry-based study. J Am Acad Dermatol. 2022;86:113-121.
- Avallone G, Quaglino P, Cavallo F, et al. SARS-CoV-2 vaccine-related cutaneous manifestations: a systematic review. Int J Dermatol. 2022;61:1187-1204. doi:10.1111/ijd.16063
- Gambichler T, Boms S, Susok L, et al. Cutaneous findings following COVID-19 vaccination: review of world literature and own experience. J Eur Acad Dermatol Venereol. 2022;36:172-180.
- Kroumpouzos G, Paroikaki ME, Yumeen S, et al. Cutaneous complications of mRNA and AZD1222 COVID-19 vaccines: a worldwide review. Microorganisms. 2022;10:624.
- Robinson L,Fu X,Hashimoto D, et al. Incidence of cutaneous reactions after messenger RNA COVID-19 vaccines. 2021;
- Wollina U, Chiriac A, Kocic H, et al. Cutaneous and hypersensitivity reactions associated with COVID-19 vaccination: a narrative review. Wien Med Wochenschr. 2022;172:63-69.
- Wei TS. Cutaneous reactions to COVID-19 vaccines: a review. JAAD Int. 2022;7:178-186.
- Katsikas Triantafyllidis K, Giannos P, Mian IT, et al. Varicella zoster virus reactivation following COVID-19 vaccination: a systematic review of case reports. Vaccines (Basel). 2021;9:1013.
- Maronese CA, Caproni M, Moltrasio C, et al. Bullous pemphigoid associated with COVID-19 vaccines: an Italian multicentre study. Front Med (Lausanne). 2022;9:841506.
- Cavazos A, Deb A, Sharma U, et al. COVID toes following vaccination. Proc (Bayl Univ Med Cent). 2022;35:476-479.
- Lesort C, Kanitakis J, Danset M, et al. Chilblain-like lesions after BNT162b2 mRNA COVID-19 vaccine: a case report suggesting that ‘COVID toes’ are due to the immune reaction to SARS-CoV-2. J Eur Acad Dermatol Venereol. 2021;35:E630-E632.
- Russo R, Cozzani E, Micalizzi C, et al. Chilblain-like lesions after COVID-19 vaccination: a case series. Acta Derm Venereol. 2022;102:adv00711. doi:10.2340/actadv.v102.2076
- Ortigosa LCM, Lenzoni FC, Suárez MV, et al. Hypersensitivity reaction to hyaluronic acid dermal filler after COVID-19 vaccination: a series of cases in São Paulo, Brazil. Int J Infect Dis. 2022;116:268-270.
- Agaronov A, Makdesi C, Hall CS. Acute generalized exanthematous pustulosis induced by Moderna COVID-19 messenger RNA vaccine. JAAD Case Rep. 2021;16:96-97.
- Dash S, Sirka CS, Mishra S, et al. COVID-19 vaccine-induced Stevens-Johnson syndrome. Clin Exp Dermatol. 2021;46:1615-1617.
- Huang Y, Tsai TF. Exacerbation of psoriasis following COVID-19 vaccination: report from a single center. Front Med (Lausanne). 2021;8:812010.
- Elamin S, Hinds F, Tolland J. De novo generalized pustular psoriasis following Oxford-AstraZeneca COVID-19 vaccine. Clin Exp Dermatol 2022;47:153-155.
- Abdelmaksoud A, Wollina U, Temiz SA, et al. SARS-CoV-2 vaccination-induced cutaneous vasculitis: report of two new cases and literature review. Dermatol Ther. 2022;35:E15458.
- Fritzen M, Funchal GDG, Luiz MO, et al. Leukocytoclastic vasculitis after exposure to COVID-19 vaccine. An Bras Dermatol. 2022;97:118-121.
- Vassallo C, Boveri E, Brazzelli V, et al. Cutaneous lymphocytic vasculitis after administration of COVID-19 mRNA vaccine. Dermatol Ther. 2021;34:E15076.
- Nazzaro G, Maronese CA. Urticarial vasculitis following mRNA anti-COVID-19 vaccine. Dermatol Ther. 2022;35:E15282.
- Hoshina D, Orita A. Sweet syndrome after severe acute respiratory syndrome coronavirus 2 mRNA vaccine: a case report and literature review. J Dermatol. 2022;49:E175-E176.
- Lemoine C, Padilla C, Krampe N, et al. Systemic lupus erythematous after Pfizer COVID-19 vaccine: a case report. Clin Rheumatol. 2022;41:1597-1601.
- Nguyen B, Lalama MJ, Gamret AC, et al. Cutaneous symptoms of connective tissue diseases after COVID-19 vaccination: a systematic review. Int J Dermatol. 2022;61:E238-E241.
- Gallo G, Mastorino L, Tonella L, et al. Alopecia areata after COVID-19 vaccination. Clin Exp Vaccine Res. 2022;11:129-132.
- Norimatsu Y, Norimatsu Y. A severe case of Trichophyton rubrum-caused dermatomycosis exacerbated after COVID-19 vaccination that had to be differentiated from pustular psoriasis. Med Mycol Case Rep. 2022;36:19-22.
- Yang K, Prussick L, Hartman R, et al. Acantholytic dyskeratosis post-COVID vaccination. Am J Dermatopathol. 2022;44:E61-E63.
- Koumaki D, Marinos L, Nikolaou V, et al. Lymphomatoid papulosis (LyP) after AZD1222 and BNT162b2 COVID-19 vaccines. Int J Dermatol. 2022;61:900-902.
- World Health Organization. Statement on the fourteenth meeting of the International Health Regulations (2005) Emergency Committee regarding the coronavirus disease (COVID-19) pandemic. Published January 30, 2023. Accessed April 12, 2023. https://www.who.int/news/item/30-01-2023-statement-on-the-fourteenth-meeting-of-the-international-health-regulations-(2005)-emergency-committee-regarding-the-coronavirus-disease-(covid-19)-pandemic
COVID-19 is a potentially severe systemic disease caused by SARS-CoV-2. SARS-CoV and Middle East respiratory syndrome (MERS-CoV) caused fatal epidemics in Asia in 2002 to 2003 and in the Arabian Peninsula in 2012, respectively. In 2019, SARS-CoV-2 was detected in patients with severe, sometimes fatal pneumonia of previously unknown origin; it rapidly spread around the world, and the World Health Organization declared the disease a pandemic on March 11, 2020. SARS-CoV-2 is a β-coronavirus that is genetically related to the bat coronavirus and SARS-CoV; it is a single-stranded RNA virus of which several variants and subvariants exist. The SARS-CoV-2 viral particles bind via their surface spike protein (S protein) to the angiotensin-converting enzyme 2 receptor present on the membrane of several cell types, including epidermal and adnexal keratinocytes.1,2 The α and δ variants, predominant from 2020 to 2021, mainly affected the lower respiratory tract and caused severe, potentially fatal pneumonia, especially in patients older than 65 years and/or with comorbidities, such as obesity, hypertension, diabetes, and (iatrogenic) immunosuppression. The ο variant, which appeared in late 2021, is more contagious than the initial variants, but it causes a less severe disease preferentially affecting the upper respiratory airways.3 As of April 5, 2023, more than 762,000,000 confirmed cases of COVID-19 have been recorded worldwide, causing more than 6,800,000 deaths.4
Early studies from China describing the symptoms of COVID-19 reported a low frequency of skin manifestations (0.2%), probably because they were focused on the most severe disease symptoms.5 Subsequently, when COVID-19 spread to the rest of the world, an increasing number of skin manifestations were reported in association with the disease. After the first publication from northern Italy in spring 2020, which was specifically devoted to skin manifestations of COVID-19,6 an explosive number of publications reported a large number of skin manifestations, and national registries were established in several countries to record these manifestations, such as the American Academy of Dermatology and the International League of Dermatological Societies registry,7,8 the COVIDSKIN registry of the French Dermatology Society,9 and the Italian registry.10 Highlighting the unprecedented number of scientific articles published on this new disease, a PubMed search of articles indexed for MEDLINE search using the terms SARS-CoV-2 or COVID-19, on April 6, 2023, revealed 351,596 articles; that is more than 300 articles published every day in this database alone, with a large number of them concerning the skin.
SKIN DISEASSES ASSOCIATED WITH COVID-19
There are several types of COVID-19–related skin manifestations, depending on the circumstances of onset and the evolution of the pandemic.
Skin Manifestations Associated With SARS-CoV-2 Infection
The estimated incidence varies greatly according to the published series of patients, possibly depending on the geographic location. The estimated incidence seems lower in Asian countries, such as China (0.2%)5 and Japan (0.56%),11 compared with Europe (up to 20%).6 Skin manifestations associated with SARS-CoV-2 infection affect individuals of all ages, slightly more females, and are clinically polymorphous; some of them are associated with the severity of the infection.12 They may precede, accompany, or appear after the symptoms of COVID-19, most often within a month of the infection, of which they rarely are the only manifestation; however, their precise relationship to SARS-CoV-2 is not always well known. They have been classified according to their clinical presentation into several forms.13-15
Morbilliform Maculopapular Eruption—Representing 16% to 53% of skin manifestations, morbilliform and maculopapular eruptions usually appear within 15 days of infection; they manifest with more or less confluent erythematous macules that may be hemorrhagic/petechial, and usually are asymptomatic and rarely pruritic. The rash mainly affects the trunk and limbs, sparing the face, palmoplantar regions, and mucous membranes; it appears concomitantly with or a few days after the first symptoms of COVID-19 (eg, fever, respiratory symptoms), regresses within a few days, and does not appear to be associated with disease severity. The distinction from maculopapular drug eruptions may be subtle. Histologically, the rash manifests with a spongiform dermatitis (ie, variable parakeratosis; spongiosis; and a mixed dermal perivascular infiltrate of lymphocytes, eosinophils and histiocytes, depending on the lesion age)(Figure 1). The etiopathogenesis is unknown; it may involve immune complexes to SARS-CoV-2 deposited on skin vessels. Treatment is not mandatory; if necessary, local or systemic corticosteroids may be used.

Vesicular (Pseudovaricella) Rash—This rash accounts for 11% to 18% of all skin manifestations and usually appears within 15 days of COVID-19 onset. It manifests with small monomorphous or varicellalike (pseudopolymorphic) vesicles appearing on the trunk, usually in young patients. The vesicles may be herpetiform, hemorrhagic, or pruritic, and appear before or within 3 days of the onset of mild COVID-19 symptoms; they regress within a few days without scarring. Histologically, the lesions show basal cell vacuolization; multinucleated, dyskeratotic/apoptotic or ballooning/acantholytic epidermal keratinocytes; reticular degeneration of the epidermis; intraepidermal vesicles sometimes resembling herpetic vesicular infections or Grover disease; and mild dermal inflammation. There is no specific treatment.
Urticaria—Urticarial rash, or urticaria, represents 5% to 16% of skin manifestations; usually appears within 15 days of disease onset; and manifests with pruritic, migratory, edematous papules appearing mainly on the trunk and occasionally the face and limbs. The urticarial rash tends to be associated with more severe forms of the disease and regresses within a week, responding to antihistamines. Of note, clinically similar rashes can be caused by drugs. Histologically, the lesions show dermal edema and a mild perivascular lymphocytic infiltrate, sometimes admixed with eosinophils.
Chilblainlike Lesions—Chilblainlike lesions (CBLLs) account for 19% of skin manifestations associated with COVID-1913 and present as erythematous-purplish, edematous lesions that can be mildly pruritic or painful, appearing on the toes—COVID toes—and more rarely the fingers (Figure 2). They were seen epidemically during the first pandemic wave (2020 lockdown) in several countries, and clinically are very similar to, if not indistinguishable from, idiopathic chilblains, but are not necessarily associated with cold exposure. They appear in young, generally healthy patients or those with mild COVID-19 symptoms 2 to 4 weeks after symptom onset. They regress spontaneously or under local corticosteroid treatment within a few days or weeks. Histologically, CBLLs are indistinguishable from chilblains of other origins, namely idiopathic (seasonal) ones. They manifest with necrosis of epidermal keratinocytes; dermal edema that may be severe, leading to the development of subepidermal pseudobullae; a rather dense perivascular and perieccrine gland lymphocytic infiltrate; and sometimes with vascular lesions (eg, edema of endothelial cells, microthromboses of dermal capillaries and venules, fibrinoid deposits within the wall of dermal venules)(Figure 3).16-18 Most patients (>80%) with CBLLs have negative serologic or polymerase chain reaction tests for SARS-CoV-2,19 which generated a lively debate about the role of SARS-CoV-2 in the genesis of CBLLs. According to some authors, SARS-CoV-2 plays no direct role, and CBLLs would occur in young people who sit or walk barefoot on cold floors at home during confinement.20-23 Remarkably, CBLLs appeared in patients with no history of chilblains during a season that was not particularly cold, namely in France or in southern California, where their incidence was much higher compared to the same time period of prior years. Some reports have supported a direct role for the virus based on questionable observations of the virus within skin lesions (eg, sweat glands, endothelial cells) by immunohistochemistry, electron microscopy, and/or in situ hybridization.17,24,25 A more satisfactory hypothesis would involve the role of a strong innate immunity leading to elimination of the virus before the development of specific antibodies via the increased production of type 1 interferon (IFN-1); this would affect the vessels, causing CBLLs. This mechanism would be similar to the one observed in some interferonopathies (eg, Aicardi-Goutières syndrome), also characterized by IFN-1 hypersecretion and chilblains.26-29 According to this hypothesis, CBLLs should be considered a paraviral rash similar to other skin manifestations associated with COVID-19.30

Acro-ischemia—Acro-ischemia livedoid lesions account for 1% to 6% of skin manifestations and comprise lesions of livedo (either reticulated or racemosa); necrotic acral bullae; and gangrenous necrosis of the extremities, especially the toes. The livedoid lesions most often appear within 15 days of COVID-19 symptom onset, and the purpuric lesions somewhat later (2–4 weeks); they mainly affect adult patients, last about 10 days, and are the hallmark of severe infection, presumably related to microthromboses of the cutaneous capillaries (endothelial dysfunction, prothrombotic state, elevated D-dimers). Histologically, they show capillary thrombosis and dermoepidermal necrosis (Figure 4).

Other Reported Polymorphic or Atypical Rashes—Erythema multiforme–like eruptions may appear before other COVID-19 symptoms and manifest as reddish-purple, nearly symmetric, diffuse, occasionally targetoid bullous or necrotic macules. The eruptions mainly affect adults and most often are seen on the palms, elbows, knees, and sometimes the mucous membranes. The rash regresses in 1 to 3 weeks without scarring and represents a delayed cutaneous hypersensitivity reaction. Histologically, the lesions show vacuolization of basal epidermal keratinocytes, keratinocyte necrosis, dermoepidermal detachment, a variably dense dermal T-lymphocytic infiltrate, and red blood cell extravasation (Figure 5).

Leukocytoclastic vasculitis may be generalized or localized. It manifests clinically by petechial/purpuric maculopapules, especially on the legs, mainly in elderly patients with COVID-19. Histologically, the lesions show necrotizing changes of dermal postcapillary venules, neutrophilic perivascular inflammation, red blood cell extravasation, and occasionally vascular IgA deposits by direct immunofluorescence examination. The course usually is benign.

The incidence of pityriasis rosea and of clinically similar rashes (referred to as “pityriasis rosea–like”) increased 5-fold during the COVID-19 pandemic.31,32 These dermatoses manifest with erythematous, scaly, circinate plaques, typically with an initial herald lesion followed a few days later by smaller erythematous macules. Histologically, the lesions comprise a spongiform dermatitis with intraepidermal exocytosis of red blood cells and a mild to moderate dermal lymphocytic infiltrate.
Erythrodysesthesia, or hand-foot syndrome, manifests with edematous erythema and palmoplantar desquamation accompanied by a burning sensation or pain. This syndrome is known as an adverse effect of some chemotherapies because of the associated drug toxicity and sweat gland inflammation; it was observed in 40% of 666 COVID-19–positive patients with mild to moderate pneumonitis.33
“COVID nose” is a rare cutaneous manifestation characterized by nasal pigmentation comprising multiple coalescent frecklelike macules on the tip and wings of the nose and sometimes the malar areas. These lesions predominantly appear in women aged 25 to 65 years and show on average 23 days after onset of COVID-19, which is usually mild. This pigmentation is similar to pigmentary changes after infection with chikungunya; it can be treated with depigmenting products such as azelaic acid and hydroquinone cream with sunscreen use, and it regresses in 2 to 4 months.34
Telogen effluvium (excessive and temporary shedding of normal telogen club hairs of the entire scalp due to the disturbance of the hair cycle) is reportedly frequent in patients (48%) 1 month after COVID-19 infection, but it may appear later (after 12 weeks).35 Alopecia also is frequently reported during long (or postacute) COVID-19 (ie, the symptomatic disease phase past the acute 4 weeks’ stage of the infection) and shows a female predominance36; it likely represents the telogen effluvium seen 90 days after a severe illness. Trichodynia (pruritus, burning, pain, or paresthesia of the scalp) also is reportedly common (developing in more than 58% of patients) and is associated with telogen effluvium in 44% of cases. Several cases of alopecia areata (AA) triggered or aggravated by COVID-19 also have been reported37,38; they could be explained by the “cytokine storm” triggered by the infection, involving T and B lymphocytes; plasmacytoid dendritic cells; natural killer cells with oversecretion of IL-6, IL-4, tumor necrosis factor α, and IFN type I; and a cytotoxic reaction associated with loss of the immune privilege of hair follicles.
Nail Manifestations
The red half-moon nail sign is an asymptomatic purplish-red band around the distal margin of the lunula that affects some adult patients with COVID-19.39 It appears shortly after onset of symptoms, likely the manifestation of vascular inflammation in the nail bed, and regresses slowly after approximately 1 week.40 Beau lines are transverse grooves in the nail plate due to the temporary arrest of the proximal nail matrix growth accompanying systemic illnesses; they appear approximately 2 to 3 weeks after the onset of COVID-19.41 Furthermore, nail alterations can be caused by drugs used to treat COVID-19, such as longitudinal melanonychia due to treatment with hydroxychloroquine or fluorescence of the lunula or nail plate due to treatment with favipiravir.42
Multisystem Inflammatory Syndrome
Multisystem inflammatory syndrome (MIS) is clinically similar to Kawasaki disease; it typically affects children43 and more rarely adults with COVID-19. It manifests with fever, weakness, and biological inflammation and also frequently with skin lesions (72%), which are polymorphous and include morbilliform rash (27%); urticaria (24%); periorbital edema (24%); nonspecific erythema (21.2%); retiform purpura (18%); targetoid lesions (15%); malar rash (15.2%); and periareolar erythema (6%).44 Compared to Kawasaki disease, MIS affects slightly older children (mean age, 8.5 vs 3 years) and more frequently includes cardiac and gastrointestinal manifestations; the mortality rate also is slightly higher (2% vs 0.17%).45
Confirmed COVID-19 Infection
At the beginning of the pandemic, skin manifestations were reported in patients who were suspected of having COVID-19 but did not always have biological confirmation of SARS-CoV-2 infection due to the unavailability of diagnostic tests or the physical impossibility of testing. However, subsequent studies have confirmed that most of these dermatoses were indeed associated with COVID-19 infection.9,46 For example, a study of 655 patients with confirmed COVID-19 infection reported maculopapular (38%), vascular (22%), urticarial (15%), and vesicular (15%) rashes; erythema multiforme or Stevens-Johnson–like syndrome (3%, often related to the use of hydroxychloroquine); generalized pruritus (1%); and MIS (0.5%). The study confirmed that CBLLs were mostly seen in young patients with mild disease, whereas livedo (fixed rash) and retiform purpura occurred in older patients with a guarded prognosis.46
Remarkably, most dermatoses associated with SARS-CoV-2 infection were reported during the initial waves of the pandemic, which were due to the α and δ viral variants. These manifestations were reported more rarely when the ο variant was predominant, even though most patients (63%) who developed CBLLs in the first wave also developed them during the second pandemic wave.47 This decrease in the incidence of COVID-19–associated dermatoses could be because of the lower pathogenicity of the o variant,3 a lower tropism for the skin, and variations in SARS-CoV-2 antigenicity that would induce a different immunologic response, combined with an increasingly stronger herd immunity compared to the first pandemic waves achieved through vaccination and spontaneous infections in the population. Additional reasons may include different baseline characteristics in patients hospitalized with COVID-19 (regarding comorbidities, disease severity, and received treatments), and the possibility that some of the initially reported COVID-19–associated skin manifestations could have been produced by different etiologic agents.48 In the last 2 years, COVID-19–related skin manifestations have been reported mainly as adverse events to COVID-19 vaccination.
CUTANEOUS ADVERSE EFFECTS OF DRUGS USED TO TREAT COVID-19
Prior to the advent of vaccines and specific treatments for SARS-CoV-2, various drugs were used—namely hydroxychloroquine, ivermectin, and tocilizumab—that did not prove efficacious and caused diverse adverse effects, including cutaneous eruptions such as urticaria, maculopapular eruptions, erythema multiforme or Stevens-Johnson syndrome, vasculitis, longitudinal melanonychia, and acute generalized exanthematous pustulosis.49,50 Nirmatrelvir 150 mg–ritonavir 100 mg, which was authorized for emergency use by the US Food and Drug Administration for the treatment of COVID-19, is a viral protease inhibitor blocking the replication of the virus. Ritonavir can induce pruritus, maculopapular rash, acne, Stevens-Johnson syndrome, and toxic epidermal necrolysis; of note, these effects have been observed following administration of ritonavir for treatment of HIV at higher daily doses and for much longer periods of time compared with treatment of COVID-19 (600–1200 mg vs 200 mg/d, respectively). These cutaneous drug side effects are clinically similar to the manifestations caused either directly or indirectly by SARS-CoV-2 infection; therefore, it may be difficult to differentiate them.
DERMATOSES DUE TO PROTECTIVE DEVICES
Dermatoses due to personal protective equipment such as masks or face shields affected the general population and mostly health care professionals51; 54.4% of 879 health care professionals in one study reported such events.52 These dermatoses mainly include contact dermatitis of the face (nose, forehead, and cheeks) of irritant or allergic nature (eg, from preservatives releasing formaldehyde contained in masks and protective goggles). They manifest with skin dryness; desquamation; maceration; fissures; or erosions or ulcerations of the cheeks, forehead, and nose. Cases of pressure urticaria also have been reported. Irritant dermatitis induced by the frequent use of disinfectants (eg, soaps, hydroalcoholic sanitizing gels) also can affect the hands. Allergic hand dermatitis can be caused by medical gloves.
The term maskne (or mask acne) refers to a variety of mechanical acne due to the prolonged use of surgical masks (>4 hours per day for ≥6 weeks); it includes cases of de novo acne and cases of pre-existing acne aggravated by wearing a mask. Maskne is characterized by acne lesions located on the facial area covered by the mask (Figure 6). It is caused by follicular occlusion; increased sebum secretion; mechanical stress (pressure, friction); and dysbiosis of the microbiome induced by changes in heat, pH, and humidity. Preventive measures include application of noncomedogenic moisturizers or gauze before wearing the mask as well as facial cleansing with appropriate nonalcoholic products. Similar to acne, rosacea often is aggravated by prolonged wearing of surgical masks (mask rosacea).53,54

DERMATOSES REVEALED OR AGGRAVATED BY COVID-19
Exacerbation of various skin diseases has been reported after infection with SARS-CoV-2.55 Psoriasis and acrodermatitis continua of Hallopeau,56 which may progress into generalized, pustular, or erythrodermic forms,57 have been reported; the role of hydroxychloroquine and oral corticosteroids used for the treatment of COVID-19 has been suspected.57 Atopic dermatitis patients—26% to 43%—have experienced worsening of their disease after symptomatic COVID-19 infection.58 The incidence of herpesvirus infections, including herpes zoster, increased during the pandemic.59 Alopecia areata relapses occurred in 42.5% of 392 patients with preexisting disease within 2 months of COVID-19 onset in one study,60 possibly favored by the psychological stress; however, some studies have not confirmed the aggravating role of COVID-19 on alopecia areata.61 Lupus erythematosus, which may relapse in the form of Rowell syndrome,62 and livedoid vasculopathy63 also have been reported following COVID-19 infection.
SKIN MANIFESTATIONS ASSOCIATED WITH COVID-19 VACCINES
In parallel with the rapid spread of COVID-19 vaccination,4 an increasing number of skin manifestations has been observed following vaccination; these dermatoses now are more frequently reported than those related to natural SARS-CoV-2 infection.64-70 Vaccine-induced skin manifestations have a reported incidence of approximately 4% and show a female predominance.65 Most of them (79%) have been reported in association with messenger RNA (mRNA)–based vaccines, which have been the most widely used; however, the frequency of side effects would be lower after mRNA vaccines than after inactivated virus-based vaccines. Eighteen percent occurred after the adenoviral vector vaccine, and 3% after the inactivated virus vaccine.70 Fifty-nine percent were observed after the first dose. They are clinically polymorphous and generally benign, regressing spontaneously after a few days, and they should not constitute a contraindication to vaccination.Interestingly, many skin manifestations are similar to those associated with natural SARS-CoV-2 infection; however, their frequency and severity does not seem to depend on whether the patients had developed skin reactions during prior SARS-CoV-2 infection. These reactions have been classified into several types:
• Immediate local reactions at the injection site: pain, erythema, or edema represent the vast majority (96%) of reactions to vaccines. They appear within 7 days after vaccination (average, 1 day), slightly more frequently (59%) after the first dose. They concern mostly young patients and are benign, regressing in 2 to 3 days.70
• Delayed local reactions: characterized by pain or pruritus, erythema, and skin induration mimicking cellulitis (COVID arm) and represent 1.7% of postvaccination reactions. They correspond to a delayed hypersensitivity reaction and appear approximately 7 days after vaccination, most often after the first vaccine dose (75% of cases), which is almost invariably mRNA based.70
• Urticarial reactions corresponding to an immediate (type 1) hypersensitivity reaction: constitute 1% of postvaccination reactions, probably due to an allergy to vaccine ingredients. They appear on average 1 day after vaccination, almost always with mRNA vaccines.70
• Angioedema: characterized by mucosal or subcutaneous edema and constitutes 0.5% of postvaccination reactions. It is a potentially serious reaction that appears on average 12 hours after vaccination, always with an mRNA-based vaccine.70
• Morbilliform rash: represents delayed hypersensitivity reactions (0.1% of postvaccination reactions) that appear mostly after the first dose (72%), on average 3 days after vaccination, always with an mRNA-based vaccine.70
• Herpes zoster: usually develops after the first vaccine dose in elderly patients (69% of cases) on average 4 days after vaccination and constitutes 0.1% of postvaccination reactions.71
• Bullous diseases: mainly bullous pemphigoid (90%) and more rarely pemphigus (5%) or bullous erythema pigmentosum (5%). They appear in elderly patients on average 7 days after vaccination and constitute 0.04% of postvaccination reactions.72
• Chilblainlike lesions: several such cases have been reported so far73; they constitute 0.03% of postvaccination reactions.70 Clinically, they are similar to those associated with natural COVID-19; they appear mostly after the first dose (64%), on average 5 days after vaccination with the mRNA or adenovirus vaccine, and show a female predominance. The appearance of these lesions in vaccinated patients, who are a priori not carriers of the virus, strongly suggests that CBLLs are due to the immune reaction against SARS-CoV-2 rather than to a direct effect of this virus on the skin, which also is a likely scenario with regards to other skin manifestations seen during the successive COVID-19 epidemic waves.73-75
• Reactions to hyaluronic acid–containing cosmetic fillers: erythema, edema, and potentially painful induration at the filler injection sites. They constitute 0.04% of postvaccination skin reactions and appear 24 hours after vaccination with mRNA-based vaccines, equally after the first or second dose.76
• Pityriasis rosea–like rash: most occur after the second dose of mRNA-based vaccines (0.023% of postvaccination skin reactions).70
• Severe reactions: these include acute generalized exanthematous pustulosis77 and Stevens-Johnson syndrome.78 One case of each has been reported after the adenoviral vector vaccine 3 days after vaccination.
Other more rarely observed manifestations include reactivation/aggravation or de novo appearance of inflammatory dermatoses such as psoriasis,79,80 leukocytoclastic vasculitis,81,82 lymphocytic83 or urticarial84 vasculitis, Sweet syndrome,85 lupus erythematosus, dermatomyositis,86,87 alopecia,37,88 infection with Trichophyton rubrum,89 Grover disease,90 and lymphomatoid reactions (such as recurrences of cutaneous T-cell lymphomas [CD30+], and de novo development of lymphomatoid papulosis).91
FINAL THOUGHTS
COVID-19 is associated with several skin manifestations, even though the causative role of SARS-CoV-2 has remained elusive. These dermatoses are highly polymorphous, mostly benign, and usually spontaneously regressive, but some of them reflect severe infection. They mostly were described during the first pandemic waves, reported in several national and international registries, which allowed for their morphological classification. Currently, cutaneous adverse effects of vaccines are the most frequently reported dermatoses associated with SARS-CoV-2, and it is likely that they will continue to be observed while COVID-19 vaccination lasts. Hopefully the end of the COVID-19 pandemic is near. In January 2023, the International Health Regulations Emergency Committee of the World Health Organization acknowledged that the COVID-19 pandemic may be approaching an inflexion point, and even though the event continues to constitute a public health emergency of international concern, the higher levels of population immunity achieved globally through infection and/or vaccination may limit the impact of SARS-CoV-2 on morbidity and mortality. However, there is little doubt that this virus will remain a permanently established pathogen in humans and animals for the foreseeable future.92 Therefore, physicians—especially dermatologists—should be aware of the various skin manifestations associated with COVID-19 so they can more efficiently manage their patients.
COVID-19 is a potentially severe systemic disease caused by SARS-CoV-2. SARS-CoV and Middle East respiratory syndrome (MERS-CoV) caused fatal epidemics in Asia in 2002 to 2003 and in the Arabian Peninsula in 2012, respectively. In 2019, SARS-CoV-2 was detected in patients with severe, sometimes fatal pneumonia of previously unknown origin; it rapidly spread around the world, and the World Health Organization declared the disease a pandemic on March 11, 2020. SARS-CoV-2 is a β-coronavirus that is genetically related to the bat coronavirus and SARS-CoV; it is a single-stranded RNA virus of which several variants and subvariants exist. The SARS-CoV-2 viral particles bind via their surface spike protein (S protein) to the angiotensin-converting enzyme 2 receptor present on the membrane of several cell types, including epidermal and adnexal keratinocytes.1,2 The α and δ variants, predominant from 2020 to 2021, mainly affected the lower respiratory tract and caused severe, potentially fatal pneumonia, especially in patients older than 65 years and/or with comorbidities, such as obesity, hypertension, diabetes, and (iatrogenic) immunosuppression. The ο variant, which appeared in late 2021, is more contagious than the initial variants, but it causes a less severe disease preferentially affecting the upper respiratory airways.3 As of April 5, 2023, more than 762,000,000 confirmed cases of COVID-19 have been recorded worldwide, causing more than 6,800,000 deaths.4
Early studies from China describing the symptoms of COVID-19 reported a low frequency of skin manifestations (0.2%), probably because they were focused on the most severe disease symptoms.5 Subsequently, when COVID-19 spread to the rest of the world, an increasing number of skin manifestations were reported in association with the disease. After the first publication from northern Italy in spring 2020, which was specifically devoted to skin manifestations of COVID-19,6 an explosive number of publications reported a large number of skin manifestations, and national registries were established in several countries to record these manifestations, such as the American Academy of Dermatology and the International League of Dermatological Societies registry,7,8 the COVIDSKIN registry of the French Dermatology Society,9 and the Italian registry.10 Highlighting the unprecedented number of scientific articles published on this new disease, a PubMed search of articles indexed for MEDLINE search using the terms SARS-CoV-2 or COVID-19, on April 6, 2023, revealed 351,596 articles; that is more than 300 articles published every day in this database alone, with a large number of them concerning the skin.
SKIN DISEASSES ASSOCIATED WITH COVID-19
There are several types of COVID-19–related skin manifestations, depending on the circumstances of onset and the evolution of the pandemic.
Skin Manifestations Associated With SARS-CoV-2 Infection
The estimated incidence varies greatly according to the published series of patients, possibly depending on the geographic location. The estimated incidence seems lower in Asian countries, such as China (0.2%)5 and Japan (0.56%),11 compared with Europe (up to 20%).6 Skin manifestations associated with SARS-CoV-2 infection affect individuals of all ages, slightly more females, and are clinically polymorphous; some of them are associated with the severity of the infection.12 They may precede, accompany, or appear after the symptoms of COVID-19, most often within a month of the infection, of which they rarely are the only manifestation; however, their precise relationship to SARS-CoV-2 is not always well known. They have been classified according to their clinical presentation into several forms.13-15
Morbilliform Maculopapular Eruption—Representing 16% to 53% of skin manifestations, morbilliform and maculopapular eruptions usually appear within 15 days of infection; they manifest with more or less confluent erythematous macules that may be hemorrhagic/petechial, and usually are asymptomatic and rarely pruritic. The rash mainly affects the trunk and limbs, sparing the face, palmoplantar regions, and mucous membranes; it appears concomitantly with or a few days after the first symptoms of COVID-19 (eg, fever, respiratory symptoms), regresses within a few days, and does not appear to be associated with disease severity. The distinction from maculopapular drug eruptions may be subtle. Histologically, the rash manifests with a spongiform dermatitis (ie, variable parakeratosis; spongiosis; and a mixed dermal perivascular infiltrate of lymphocytes, eosinophils and histiocytes, depending on the lesion age)(Figure 1). The etiopathogenesis is unknown; it may involve immune complexes to SARS-CoV-2 deposited on skin vessels. Treatment is not mandatory; if necessary, local or systemic corticosteroids may be used.

Vesicular (Pseudovaricella) Rash—This rash accounts for 11% to 18% of all skin manifestations and usually appears within 15 days of COVID-19 onset. It manifests with small monomorphous or varicellalike (pseudopolymorphic) vesicles appearing on the trunk, usually in young patients. The vesicles may be herpetiform, hemorrhagic, or pruritic, and appear before or within 3 days of the onset of mild COVID-19 symptoms; they regress within a few days without scarring. Histologically, the lesions show basal cell vacuolization; multinucleated, dyskeratotic/apoptotic or ballooning/acantholytic epidermal keratinocytes; reticular degeneration of the epidermis; intraepidermal vesicles sometimes resembling herpetic vesicular infections or Grover disease; and mild dermal inflammation. There is no specific treatment.
Urticaria—Urticarial rash, or urticaria, represents 5% to 16% of skin manifestations; usually appears within 15 days of disease onset; and manifests with pruritic, migratory, edematous papules appearing mainly on the trunk and occasionally the face and limbs. The urticarial rash tends to be associated with more severe forms of the disease and regresses within a week, responding to antihistamines. Of note, clinically similar rashes can be caused by drugs. Histologically, the lesions show dermal edema and a mild perivascular lymphocytic infiltrate, sometimes admixed with eosinophils.
Chilblainlike Lesions—Chilblainlike lesions (CBLLs) account for 19% of skin manifestations associated with COVID-1913 and present as erythematous-purplish, edematous lesions that can be mildly pruritic or painful, appearing on the toes—COVID toes—and more rarely the fingers (Figure 2). They were seen epidemically during the first pandemic wave (2020 lockdown) in several countries, and clinically are very similar to, if not indistinguishable from, idiopathic chilblains, but are not necessarily associated with cold exposure. They appear in young, generally healthy patients or those with mild COVID-19 symptoms 2 to 4 weeks after symptom onset. They regress spontaneously or under local corticosteroid treatment within a few days or weeks. Histologically, CBLLs are indistinguishable from chilblains of other origins, namely idiopathic (seasonal) ones. They manifest with necrosis of epidermal keratinocytes; dermal edema that may be severe, leading to the development of subepidermal pseudobullae; a rather dense perivascular and perieccrine gland lymphocytic infiltrate; and sometimes with vascular lesions (eg, edema of endothelial cells, microthromboses of dermal capillaries and venules, fibrinoid deposits within the wall of dermal venules)(Figure 3).16-18 Most patients (>80%) with CBLLs have negative serologic or polymerase chain reaction tests for SARS-CoV-2,19 which generated a lively debate about the role of SARS-CoV-2 in the genesis of CBLLs. According to some authors, SARS-CoV-2 plays no direct role, and CBLLs would occur in young people who sit or walk barefoot on cold floors at home during confinement.20-23 Remarkably, CBLLs appeared in patients with no history of chilblains during a season that was not particularly cold, namely in France or in southern California, where their incidence was much higher compared to the same time period of prior years. Some reports have supported a direct role for the virus based on questionable observations of the virus within skin lesions (eg, sweat glands, endothelial cells) by immunohistochemistry, electron microscopy, and/or in situ hybridization.17,24,25 A more satisfactory hypothesis would involve the role of a strong innate immunity leading to elimination of the virus before the development of specific antibodies via the increased production of type 1 interferon (IFN-1); this would affect the vessels, causing CBLLs. This mechanism would be similar to the one observed in some interferonopathies (eg, Aicardi-Goutières syndrome), also characterized by IFN-1 hypersecretion and chilblains.26-29 According to this hypothesis, CBLLs should be considered a paraviral rash similar to other skin manifestations associated with COVID-19.30

Acro-ischemia—Acro-ischemia livedoid lesions account for 1% to 6% of skin manifestations and comprise lesions of livedo (either reticulated or racemosa); necrotic acral bullae; and gangrenous necrosis of the extremities, especially the toes. The livedoid lesions most often appear within 15 days of COVID-19 symptom onset, and the purpuric lesions somewhat later (2–4 weeks); they mainly affect adult patients, last about 10 days, and are the hallmark of severe infection, presumably related to microthromboses of the cutaneous capillaries (endothelial dysfunction, prothrombotic state, elevated D-dimers). Histologically, they show capillary thrombosis and dermoepidermal necrosis (Figure 4).

Other Reported Polymorphic or Atypical Rashes—Erythema multiforme–like eruptions may appear before other COVID-19 symptoms and manifest as reddish-purple, nearly symmetric, diffuse, occasionally targetoid bullous or necrotic macules. The eruptions mainly affect adults and most often are seen on the palms, elbows, knees, and sometimes the mucous membranes. The rash regresses in 1 to 3 weeks without scarring and represents a delayed cutaneous hypersensitivity reaction. Histologically, the lesions show vacuolization of basal epidermal keratinocytes, keratinocyte necrosis, dermoepidermal detachment, a variably dense dermal T-lymphocytic infiltrate, and red blood cell extravasation (Figure 5).

Leukocytoclastic vasculitis may be generalized or localized. It manifests clinically by petechial/purpuric maculopapules, especially on the legs, mainly in elderly patients with COVID-19. Histologically, the lesions show necrotizing changes of dermal postcapillary venules, neutrophilic perivascular inflammation, red blood cell extravasation, and occasionally vascular IgA deposits by direct immunofluorescence examination. The course usually is benign.

The incidence of pityriasis rosea and of clinically similar rashes (referred to as “pityriasis rosea–like”) increased 5-fold during the COVID-19 pandemic.31,32 These dermatoses manifest with erythematous, scaly, circinate plaques, typically with an initial herald lesion followed a few days later by smaller erythematous macules. Histologically, the lesions comprise a spongiform dermatitis with intraepidermal exocytosis of red blood cells and a mild to moderate dermal lymphocytic infiltrate.
Erythrodysesthesia, or hand-foot syndrome, manifests with edematous erythema and palmoplantar desquamation accompanied by a burning sensation or pain. This syndrome is known as an adverse effect of some chemotherapies because of the associated drug toxicity and sweat gland inflammation; it was observed in 40% of 666 COVID-19–positive patients with mild to moderate pneumonitis.33
“COVID nose” is a rare cutaneous manifestation characterized by nasal pigmentation comprising multiple coalescent frecklelike macules on the tip and wings of the nose and sometimes the malar areas. These lesions predominantly appear in women aged 25 to 65 years and show on average 23 days after onset of COVID-19, which is usually mild. This pigmentation is similar to pigmentary changes after infection with chikungunya; it can be treated with depigmenting products such as azelaic acid and hydroquinone cream with sunscreen use, and it regresses in 2 to 4 months.34
Telogen effluvium (excessive and temporary shedding of normal telogen club hairs of the entire scalp due to the disturbance of the hair cycle) is reportedly frequent in patients (48%) 1 month after COVID-19 infection, but it may appear later (after 12 weeks).35 Alopecia also is frequently reported during long (or postacute) COVID-19 (ie, the symptomatic disease phase past the acute 4 weeks’ stage of the infection) and shows a female predominance36; it likely represents the telogen effluvium seen 90 days after a severe illness. Trichodynia (pruritus, burning, pain, or paresthesia of the scalp) also is reportedly common (developing in more than 58% of patients) and is associated with telogen effluvium in 44% of cases. Several cases of alopecia areata (AA) triggered or aggravated by COVID-19 also have been reported37,38; they could be explained by the “cytokine storm” triggered by the infection, involving T and B lymphocytes; plasmacytoid dendritic cells; natural killer cells with oversecretion of IL-6, IL-4, tumor necrosis factor α, and IFN type I; and a cytotoxic reaction associated with loss of the immune privilege of hair follicles.
Nail Manifestations
The red half-moon nail sign is an asymptomatic purplish-red band around the distal margin of the lunula that affects some adult patients with COVID-19.39 It appears shortly after onset of symptoms, likely the manifestation of vascular inflammation in the nail bed, and regresses slowly after approximately 1 week.40 Beau lines are transverse grooves in the nail plate due to the temporary arrest of the proximal nail matrix growth accompanying systemic illnesses; they appear approximately 2 to 3 weeks after the onset of COVID-19.41 Furthermore, nail alterations can be caused by drugs used to treat COVID-19, such as longitudinal melanonychia due to treatment with hydroxychloroquine or fluorescence of the lunula or nail plate due to treatment with favipiravir.42
Multisystem Inflammatory Syndrome
Multisystem inflammatory syndrome (MIS) is clinically similar to Kawasaki disease; it typically affects children43 and more rarely adults with COVID-19. It manifests with fever, weakness, and biological inflammation and also frequently with skin lesions (72%), which are polymorphous and include morbilliform rash (27%); urticaria (24%); periorbital edema (24%); nonspecific erythema (21.2%); retiform purpura (18%); targetoid lesions (15%); malar rash (15.2%); and periareolar erythema (6%).44 Compared to Kawasaki disease, MIS affects slightly older children (mean age, 8.5 vs 3 years) and more frequently includes cardiac and gastrointestinal manifestations; the mortality rate also is slightly higher (2% vs 0.17%).45
Confirmed COVID-19 Infection
At the beginning of the pandemic, skin manifestations were reported in patients who were suspected of having COVID-19 but did not always have biological confirmation of SARS-CoV-2 infection due to the unavailability of diagnostic tests or the physical impossibility of testing. However, subsequent studies have confirmed that most of these dermatoses were indeed associated with COVID-19 infection.9,46 For example, a study of 655 patients with confirmed COVID-19 infection reported maculopapular (38%), vascular (22%), urticarial (15%), and vesicular (15%) rashes; erythema multiforme or Stevens-Johnson–like syndrome (3%, often related to the use of hydroxychloroquine); generalized pruritus (1%); and MIS (0.5%). The study confirmed that CBLLs were mostly seen in young patients with mild disease, whereas livedo (fixed rash) and retiform purpura occurred in older patients with a guarded prognosis.46
Remarkably, most dermatoses associated with SARS-CoV-2 infection were reported during the initial waves of the pandemic, which were due to the α and δ viral variants. These manifestations were reported more rarely when the ο variant was predominant, even though most patients (63%) who developed CBLLs in the first wave also developed them during the second pandemic wave.47 This decrease in the incidence of COVID-19–associated dermatoses could be because of the lower pathogenicity of the o variant,3 a lower tropism for the skin, and variations in SARS-CoV-2 antigenicity that would induce a different immunologic response, combined with an increasingly stronger herd immunity compared to the first pandemic waves achieved through vaccination and spontaneous infections in the population. Additional reasons may include different baseline characteristics in patients hospitalized with COVID-19 (regarding comorbidities, disease severity, and received treatments), and the possibility that some of the initially reported COVID-19–associated skin manifestations could have been produced by different etiologic agents.48 In the last 2 years, COVID-19–related skin manifestations have been reported mainly as adverse events to COVID-19 vaccination.
CUTANEOUS ADVERSE EFFECTS OF DRUGS USED TO TREAT COVID-19
Prior to the advent of vaccines and specific treatments for SARS-CoV-2, various drugs were used—namely hydroxychloroquine, ivermectin, and tocilizumab—that did not prove efficacious and caused diverse adverse effects, including cutaneous eruptions such as urticaria, maculopapular eruptions, erythema multiforme or Stevens-Johnson syndrome, vasculitis, longitudinal melanonychia, and acute generalized exanthematous pustulosis.49,50 Nirmatrelvir 150 mg–ritonavir 100 mg, which was authorized for emergency use by the US Food and Drug Administration for the treatment of COVID-19, is a viral protease inhibitor blocking the replication of the virus. Ritonavir can induce pruritus, maculopapular rash, acne, Stevens-Johnson syndrome, and toxic epidermal necrolysis; of note, these effects have been observed following administration of ritonavir for treatment of HIV at higher daily doses and for much longer periods of time compared with treatment of COVID-19 (600–1200 mg vs 200 mg/d, respectively). These cutaneous drug side effects are clinically similar to the manifestations caused either directly or indirectly by SARS-CoV-2 infection; therefore, it may be difficult to differentiate them.
DERMATOSES DUE TO PROTECTIVE DEVICES
Dermatoses due to personal protective equipment such as masks or face shields affected the general population and mostly health care professionals51; 54.4% of 879 health care professionals in one study reported such events.52 These dermatoses mainly include contact dermatitis of the face (nose, forehead, and cheeks) of irritant or allergic nature (eg, from preservatives releasing formaldehyde contained in masks and protective goggles). They manifest with skin dryness; desquamation; maceration; fissures; or erosions or ulcerations of the cheeks, forehead, and nose. Cases of pressure urticaria also have been reported. Irritant dermatitis induced by the frequent use of disinfectants (eg, soaps, hydroalcoholic sanitizing gels) also can affect the hands. Allergic hand dermatitis can be caused by medical gloves.
The term maskne (or mask acne) refers to a variety of mechanical acne due to the prolonged use of surgical masks (>4 hours per day for ≥6 weeks); it includes cases of de novo acne and cases of pre-existing acne aggravated by wearing a mask. Maskne is characterized by acne lesions located on the facial area covered by the mask (Figure 6). It is caused by follicular occlusion; increased sebum secretion; mechanical stress (pressure, friction); and dysbiosis of the microbiome induced by changes in heat, pH, and humidity. Preventive measures include application of noncomedogenic moisturizers or gauze before wearing the mask as well as facial cleansing with appropriate nonalcoholic products. Similar to acne, rosacea often is aggravated by prolonged wearing of surgical masks (mask rosacea).53,54

DERMATOSES REVEALED OR AGGRAVATED BY COVID-19
Exacerbation of various skin diseases has been reported after infection with SARS-CoV-2.55 Psoriasis and acrodermatitis continua of Hallopeau,56 which may progress into generalized, pustular, or erythrodermic forms,57 have been reported; the role of hydroxychloroquine and oral corticosteroids used for the treatment of COVID-19 has been suspected.57 Atopic dermatitis patients—26% to 43%—have experienced worsening of their disease after symptomatic COVID-19 infection.58 The incidence of herpesvirus infections, including herpes zoster, increased during the pandemic.59 Alopecia areata relapses occurred in 42.5% of 392 patients with preexisting disease within 2 months of COVID-19 onset in one study,60 possibly favored by the psychological stress; however, some studies have not confirmed the aggravating role of COVID-19 on alopecia areata.61 Lupus erythematosus, which may relapse in the form of Rowell syndrome,62 and livedoid vasculopathy63 also have been reported following COVID-19 infection.
SKIN MANIFESTATIONS ASSOCIATED WITH COVID-19 VACCINES
In parallel with the rapid spread of COVID-19 vaccination,4 an increasing number of skin manifestations has been observed following vaccination; these dermatoses now are more frequently reported than those related to natural SARS-CoV-2 infection.64-70 Vaccine-induced skin manifestations have a reported incidence of approximately 4% and show a female predominance.65 Most of them (79%) have been reported in association with messenger RNA (mRNA)–based vaccines, which have been the most widely used; however, the frequency of side effects would be lower after mRNA vaccines than after inactivated virus-based vaccines. Eighteen percent occurred after the adenoviral vector vaccine, and 3% after the inactivated virus vaccine.70 Fifty-nine percent were observed after the first dose. They are clinically polymorphous and generally benign, regressing spontaneously after a few days, and they should not constitute a contraindication to vaccination.Interestingly, many skin manifestations are similar to those associated with natural SARS-CoV-2 infection; however, their frequency and severity does not seem to depend on whether the patients had developed skin reactions during prior SARS-CoV-2 infection. These reactions have been classified into several types:
• Immediate local reactions at the injection site: pain, erythema, or edema represent the vast majority (96%) of reactions to vaccines. They appear within 7 days after vaccination (average, 1 day), slightly more frequently (59%) after the first dose. They concern mostly young patients and are benign, regressing in 2 to 3 days.70
• Delayed local reactions: characterized by pain or pruritus, erythema, and skin induration mimicking cellulitis (COVID arm) and represent 1.7% of postvaccination reactions. They correspond to a delayed hypersensitivity reaction and appear approximately 7 days after vaccination, most often after the first vaccine dose (75% of cases), which is almost invariably mRNA based.70
• Urticarial reactions corresponding to an immediate (type 1) hypersensitivity reaction: constitute 1% of postvaccination reactions, probably due to an allergy to vaccine ingredients. They appear on average 1 day after vaccination, almost always with mRNA vaccines.70
• Angioedema: characterized by mucosal or subcutaneous edema and constitutes 0.5% of postvaccination reactions. It is a potentially serious reaction that appears on average 12 hours after vaccination, always with an mRNA-based vaccine.70
• Morbilliform rash: represents delayed hypersensitivity reactions (0.1% of postvaccination reactions) that appear mostly after the first dose (72%), on average 3 days after vaccination, always with an mRNA-based vaccine.70
• Herpes zoster: usually develops after the first vaccine dose in elderly patients (69% of cases) on average 4 days after vaccination and constitutes 0.1% of postvaccination reactions.71
• Bullous diseases: mainly bullous pemphigoid (90%) and more rarely pemphigus (5%) or bullous erythema pigmentosum (5%). They appear in elderly patients on average 7 days after vaccination and constitute 0.04% of postvaccination reactions.72
• Chilblainlike lesions: several such cases have been reported so far73; they constitute 0.03% of postvaccination reactions.70 Clinically, they are similar to those associated with natural COVID-19; they appear mostly after the first dose (64%), on average 5 days after vaccination with the mRNA or adenovirus vaccine, and show a female predominance. The appearance of these lesions in vaccinated patients, who are a priori not carriers of the virus, strongly suggests that CBLLs are due to the immune reaction against SARS-CoV-2 rather than to a direct effect of this virus on the skin, which also is a likely scenario with regards to other skin manifestations seen during the successive COVID-19 epidemic waves.73-75
• Reactions to hyaluronic acid–containing cosmetic fillers: erythema, edema, and potentially painful induration at the filler injection sites. They constitute 0.04% of postvaccination skin reactions and appear 24 hours after vaccination with mRNA-based vaccines, equally after the first or second dose.76
• Pityriasis rosea–like rash: most occur after the second dose of mRNA-based vaccines (0.023% of postvaccination skin reactions).70
• Severe reactions: these include acute generalized exanthematous pustulosis77 and Stevens-Johnson syndrome.78 One case of each has been reported after the adenoviral vector vaccine 3 days after vaccination.
Other more rarely observed manifestations include reactivation/aggravation or de novo appearance of inflammatory dermatoses such as psoriasis,79,80 leukocytoclastic vasculitis,81,82 lymphocytic83 or urticarial84 vasculitis, Sweet syndrome,85 lupus erythematosus, dermatomyositis,86,87 alopecia,37,88 infection with Trichophyton rubrum,89 Grover disease,90 and lymphomatoid reactions (such as recurrences of cutaneous T-cell lymphomas [CD30+], and de novo development of lymphomatoid papulosis).91
FINAL THOUGHTS
COVID-19 is associated with several skin manifestations, even though the causative role of SARS-CoV-2 has remained elusive. These dermatoses are highly polymorphous, mostly benign, and usually spontaneously regressive, but some of them reflect severe infection. They mostly were described during the first pandemic waves, reported in several national and international registries, which allowed for their morphological classification. Currently, cutaneous adverse effects of vaccines are the most frequently reported dermatoses associated with SARS-CoV-2, and it is likely that they will continue to be observed while COVID-19 vaccination lasts. Hopefully the end of the COVID-19 pandemic is near. In January 2023, the International Health Regulations Emergency Committee of the World Health Organization acknowledged that the COVID-19 pandemic may be approaching an inflexion point, and even though the event continues to constitute a public health emergency of international concern, the higher levels of population immunity achieved globally through infection and/or vaccination may limit the impact of SARS-CoV-2 on morbidity and mortality. However, there is little doubt that this virus will remain a permanently established pathogen in humans and animals for the foreseeable future.92 Therefore, physicians—especially dermatologists—should be aware of the various skin manifestations associated with COVID-19 so they can more efficiently manage their patients.
- Ashraf UM, Abokor AA, Edwards JM, et al. SARS-CoV-2, ACE2 expression, and systemic organ invasion. Physiol Genomics. 2021;53:51-60.
- Ganier C, Harun N, Peplow I, et al. Angiotensin-converting enzyme 2 expression is detectable in keratinocytes, cutaneous appendages, and blood vessels by multiplex RNA in situ hybridization. Adv Skin Wound Care. 2022;35:219-223.
- Ulloa AC, Buchan SA, Daneman N, et al. Estimates of SARS-CoV-2 omicron variant severity in Ontario, Canada. JAMA. 2022;327:1286-1288.
- World Health Organization. Coronavirus (COVID-19) Dashboard. Accessed April 6, 2023. https://covid19.who.int
- Guan WJ, Ni ZY, Hu Y, et al; China Medical Treatment Expert Group for COVID-19. clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708-1720.
- Recalcati S. Cutaneous manifestations in COVID-9: a first perspective. J Eur Acad Dermatol Venereol. 2020;34:E212-E213.
- Freeman EE, McMahon DE, Lipoff JB, et al. The spectrum of COVID-19-associated dermatologic manifestations: an international registry of 716 patients from 31 countries. J Am Acad Dermatol. 2020;83:1118-1129.
- Freeman EE, Chamberlin GC, McMahon DE, et al. Dermatology COVID-19 registries: updates and future directions. Dermatol Clin. 2021;39:575-585.
- Guelimi R, Salle R, Dousset L, et al. Non-acral skin manifestations during the COVID-19 epidemic: COVIDSKIN study by the French Society of Dermatology. J Eur Acad Dermatol Venereol. 2021;35:E539-E541.
- Marzano AV, Genovese G, Moltrasio C, et al; Italian Skin COVID-19 Network of the Italian Society of Dermatology and Sexually Transmitted Diseases. The clinical spectrum of COVID-19 associated cutaneous manifestations: an Italian multicenter study of 200 adult patients. J Am Acad Dermatol. 2021;84:1356-1363.
- Sugai T, Fujita Y, Inamura E, et al. Prevalence and patterns of cutaneous manifestations in 1245 COVID-19 patients in Japan: a single-centre study. J Eur Acad Dermatol Venereol. 2022;36:E522-E524.
- Holmes Z, Courtney A, Lincoln M, et al. Rash morphology as a predictor of COVID‐19 severity: a systematic review of the cutaneous manifestations of COVID‐19. Skin Health Dis. 2022;2:E120. doi:10.1002/ski2.120
- Galván Casas C, Català A, Carretero Hernández G, et al. Classification of the cutaneous manifestations of COVID-19: a rapid prospective nationwide consensus study in Spain with 375 cases. Br J Dermatol. 2020;183:71-77.
- Garduño‑Soto M, Choreño-Parra, Cazarin-Barrientos Dermatological aspects of SARS‑CoV‑2 infection: mechanisms and manifestations. Arch Dermatol Res. 2021;313:611-622.
- Huynh T, Sanchez-Flores X, Yau J, et al. Cutaneous manifestations of SARS-CoV-2 Infection. Am J Clin Dermatol. 2022;23:277-286.
- Kanitakis J, Lesort C, Danset M, et al.
2020; 83:870-875. - Kolivras A, Thompson C, Pastushenko I, et al. A clinicopathological description of COVID-19-induced chilblains (COVID-toes) correlated with a published literature review. J Cutan Pathol. 2022;49:17-28.
- Roca-Ginés J, Torres-Navarro I, Sánchez-Arráez J, et al. Assessment of acute acral lesions in a case series of children and adolescents during the COVID-19 pandemic. 2020;156:992-997.
- Le Cleach L, Dousset L, Assier H, et al; French Society of Dermatology. Most chilblains observed during the COVID-19 outbreak occur in patients who are negative for COVID-19 on polymerase chain reaction and serology testing. Br J Dermatol. 2020;183:866-874.
- Discepolo V, Catzola A, Pierri L, et al. Bilateral chilblain-like lesions of the toes characterized by microvascular remodeling in adolescents during the COVID-19 pandemic. JAMA Netw Open. 2021;4:E2111369.
- Gehlhausen JR, Little AJ, Ko CJ, et al. Lack of association between pandemic chilblains and SARS-CoV-2 infection. Proc Natl Acad Sci U S A. 2022;119:e2122090119.
- Neri, Virdi, Corsini, et al Major cluster of paediatric ‘true’ primary chilblains during the COVID-19 pandemic: a consequence of lifestyle changes due to lockdown. J Eur Acad Dermatol Venereol. 2020;34:2630-2635.
- De Greef A, Choteau M, Herman A, et al. Chilblains observed during the COVID-19 pandemic cannot be distinguished from the classic, cold-related chilblains. Eur J Dermatol. 2022;32:377-383.
- Colmenero I, Santonja C, Alonso-Riaño M, et al. SARS-CoV-2 endothelial infection causes COVID-19 chilblains: histopathological, immunohistochemical and ultrastructural study of seven paediatric cases. Br J Dermatol. 2020;183:729-737.
- Quintero-Bustos G, Aguilar-Leon D, Saeb-Lima M. Histopathological and immunohistochemical characterization of skin biopsies from 41 SARS-CoV-2 (+) patients: experience in a Mexican concentration institute: a case series and literature review. Am J Dermatopathol. 2022;44:327-337.
- Arkin LM, Moon JJ, Tran JM, et al; COVID Human Genetic Effort. From your nose to your toes: a review of severe acute respiratory syndrome coronavirus 2 pandemic-associated pernio. J Invest Dermatol. 2021;141:2791-2796.
- Frumholtz L, Bouaziz JD, Battistella M, et al; Saint-Louis CORE (COvid REsearch). Type I interferon response and vascular alteration in chilblain-like lesions during the COVID-19 outbreak. Br J Dermatol. 2021;185:1176-1185.
- Hubiche T, Cardot-Leccia N, Le Duff F, et al. Clinical, laboratory, and interferon-alpha response characteristics of patients with chilblain-like lesions during the COVID-19 pandemic. JAMA Dermatol. 2021;157:202-206.
- Lesort C, Kanitakis J, Villani A, et al. COVID-19 and outbreak of chilblains: are they related? J Eur Acad Dermatol Venereol. 2020;34:E757-E758.
- Sanchez A, Sohier P, Benghanem S, et al. Digitate papulosquamous eruption associated with severe acute respiratory syndrome coronavirus 2 infection. JAMA Dermatol. 2020;156:819-820.
- Drago F, Broccolo F, Ciccarese G. Pityriasis rosea, pityriasis rosea-like eruptions, and herpes zoster in the setting of COVID-19 and COVID-19 vaccination. Clin Dermatol. 2022;S0738-081X(22)00002-5.
- Dursun R, Temiz SA. The clinics of HHV-6 infection in COVID-19 pandemic: pityriasis rosea and Kawasaki disease. Dermatol Ther. 2020;33:E13730.
- Nuno-Gonzalez A, Magaletsky K, Feito Rodríguez M, et al. Palmoplantar erythrodysesthesia: a diagnostic sign of COVID-19. J Eur Acad Dermatol Venereol. 2021;35:e247-e249.
- Sil A, Panigrahi A, Chandra A, et al. “COVID nose”: a unique post-COVID pigmentary sequelae reminiscing Chik sign: a descriptive case series. J Eur Acad Dermatol Venereol. 2022;36:E419-E421.
- Starace M, Iorizzo M, Sechi A, et al. Trichodynia and telogen effluvium in COVID-19 patients: results of an international expert opinion survey on diagnosis and management. JAAD Int. 2021;5:11-18.
- Wong-Chew RM, Rodríguez Cabrera EX, Rodríguez Valdez CA, et al. Symptom cluster analysis of long COVID-19 in patients discharged from the Temporary COVID-19 Hospital in Mexico City. Ther Adv Infect Dis. 2022;9:20499361211069264.
- Bardazzi F, Guglielmo A, Abbenante D, et al. New insights into alopecia areata during COVID-19 pandemic: when infection or vaccination could play a role. J Cosmet Dermatol. 2022;21:1796-1798.
- Christensen RE, Jafferany M. Association between alopecia areata and COVID-19: a systematic review. JAAD Int. 2022;7:57-61.
- Wollina U, Kanitakis J, Baran R. Nails and COVID-19: a comprehensive review of clinical findings and treatment. Dermatol Ther. 2021;34:E15100.
- Méndez-Flores S, Zaladonis A, Valdes-Rodriguez R. COVID-19 and nail manifestation: be on the lookout for the red half-moon nail sign. Int J Dermatol. 2020;59:1414.
- Alobaida S, Lam JM. Beau lines associated with COVID-19. CMAJ. 2020;192:E1040.
- Durmaz EÖ, Demirciog˘lu D. Fluorescence in the sclera, nails, and teeth secondary to favipiravir use for COVID-19 infections. J Clin Aesthet Dermatol. 2022;15:35-37.
- Brumfiel CM, DiLorenzo AM, Petronic-Rosic VM. Dermatologic manifestations of COVID-19-associated multisystem inflammatory syndrome in children. Clin Dermatol. 2021;39:329-333.
- Akçay N, Topkarcı Z, Menentog˘lu ME, et al. New dermatological findings of MIS-C: can mucocutaneous involvement be associated with severe disease course? Australas J Dermatol. 2022;63:228-234. doi:10.1111/ajd.13819
- Vogel TP, Top KA, Karatzios C, et al. Multisystem inflammatory syndrome in children and adults (MIS-C/A): case definition & guidelines for data collection, analysis, and presentation of immunization safety data. Vaccine. 2021;39:3037-3049.
- Conforti C, Dianzani C, Agozzino M, et al. Cutaneous manifestations in confirmed COVID-19 patients: a systematic review. Biology (Basel). 2020;9:449.
- Hubiche T, Le Duff F, Fontas E, et al. Relapse of chilblain-like lesions during the second wave of the COVID-19 pandemic: a cohort follow-up. Br J Dermatol. 2021;185:858-859.
- Fernandez-Nieto, Ortega-Quijano, Suarez-Valle, et al Lack of skin manifestations in COVID-19 hospitalized patients during the second epidemic wave in Spain: a possible association with a novel SARS-CoV-2 variant: a cross-sectional study. J Eur Acad Dermatol Venereol. 2021;35:E183-E185.
- Martinez-LopezA, Cuenca-Barrales, Montero-Vilchezet al Review of adverse cutaneous reactions of pharmacologic interventions for COVID-19: a guide for the dermatologist. J Am Acad Dermatol. 2020;83:1738-1748.
- Cutaneous side-effects of the potential COVID-19 drugs. Dermatol Ther. 2020;33:E13476.
- Mawhirt SL, Frankel D, Diaz AM. Cutaneous manifestations in adult patients with COVID-19 and dermatologic conditions related to the COVID-19 pandemic in health care workers. Curr Allerg Asthma Rep. 2020;20:75.
- Nguyen C, Young FG, McElroy D, et al. Personal protective equipment and adverse dermatological reactions among healthcare workers: survey observations from the COVID-19 pandemic. Medicine (Baltimore). 2022;101:E29003.
- Rathi SK, Dsouza JM. Maskne: a new acne variant in COVID-19 era. Indian J Dermatol. 2022;67:552-555.
- Damiani G, Girono L, Grada A, et al. COVID-19 related masks increase severity of both acne (maskne) and rosacea (mask rosacea): multi-center, real-life, telemedical, and observational prospective study. Dermatol Ther. 2021;34:E14848.
- Aram K, Patil A, Goldust M, et al. COVID-19 and exacerbation of dermatological diseases: a review of the available literature. Dermatol Ther. 2021;34:E15113.
- Samotij D, Gawron E, Szcze˛ch J, et al. Acrodermatitis continua of Hallopeau evolving into generalized pustular psoriasis following COVID-19: a case report of a successful treatment with infliximab in combination with acitretin. Biologics. 2021;15:107-113.
- Demiri J, Abdo M, Tsianakas A. Erythrodermic psoriasis after COVID-19 [in German]. Hautarzt. 2022;73:156-159.
- de Wijs LEM, Joustra MM, Olydam JI, et al. COVID-19 in patients with cutaneous immune-mediated diseases in the Netherlands: real-world observational data. J Eur Acad Dermatol Venereol. 2021;35:E173-E176.
- Marques NP, Maia CMF, Marques NCT, et al. Continuous increase of herpes zoster cases in Brazil during the COVID-19 pandemic. Oral Surg Oral Med Oral Pathol Oral Radiol. 2022;133:612-614.
- Rinaldi F, Trink A, Giuliani G, et al. Italian survey for the evaluation of the effects of coronavirus disease 2019 (COVID-19) pandemic on alopecia areata recurrence. Dermatol Ther (Heidelb). 2021;11:339-345.
- Rudnicka L, Rakowska A, Waskiel-Burnat A, et al. Mild-to-moderate COVID-19 is not associated with worsening of alopecia areata: a retrospective analysis of 32 patients. J Am Acad Dermatol. 2021;85:723-725.
- Drenovska K, Shahid M, Mateeva V, et al. Case report: Rowell syndrome-like flare of cutaneous lupus erythematosus following COVID-19 infection. Front Med (Lausanne). 2022;9:815743.
- Kawabe R, Tonomura K, Kotobuki Y, et al. Exacerbation of livedoid vasculopathy after coronavirus disease 2019. Eur J Dermatol. 2022;32:129-131. doi:10.1684/ejd.2022.4200
- McMahon DE, Kovarik CL, Damsky W, et al. Clinical and pathologic correlation of cutaneous COVID-19 vaccine reactions including V-REPP: a registry-based study. J Am Acad Dermatol. 2022;86:113-121.
- Avallone G, Quaglino P, Cavallo F, et al. SARS-CoV-2 vaccine-related cutaneous manifestations: a systematic review. Int J Dermatol. 2022;61:1187-1204. doi:10.1111/ijd.16063
- Gambichler T, Boms S, Susok L, et al. Cutaneous findings following COVID-19 vaccination: review of world literature and own experience. J Eur Acad Dermatol Venereol. 2022;36:172-180.
- Kroumpouzos G, Paroikaki ME, Yumeen S, et al. Cutaneous complications of mRNA and AZD1222 COVID-19 vaccines: a worldwide review. Microorganisms. 2022;10:624.
- Robinson L,Fu X,Hashimoto D, et al. Incidence of cutaneous reactions after messenger RNA COVID-19 vaccines. 2021;
- Wollina U, Chiriac A, Kocic H, et al. Cutaneous and hypersensitivity reactions associated with COVID-19 vaccination: a narrative review. Wien Med Wochenschr. 2022;172:63-69.
- Wei TS. Cutaneous reactions to COVID-19 vaccines: a review. JAAD Int. 2022;7:178-186.
- Katsikas Triantafyllidis K, Giannos P, Mian IT, et al. Varicella zoster virus reactivation following COVID-19 vaccination: a systematic review of case reports. Vaccines (Basel). 2021;9:1013.
- Maronese CA, Caproni M, Moltrasio C, et al. Bullous pemphigoid associated with COVID-19 vaccines: an Italian multicentre study. Front Med (Lausanne). 2022;9:841506.
- Cavazos A, Deb A, Sharma U, et al. COVID toes following vaccination. Proc (Bayl Univ Med Cent). 2022;35:476-479.
- Lesort C, Kanitakis J, Danset M, et al. Chilblain-like lesions after BNT162b2 mRNA COVID-19 vaccine: a case report suggesting that ‘COVID toes’ are due to the immune reaction to SARS-CoV-2. J Eur Acad Dermatol Venereol. 2021;35:E630-E632.
- Russo R, Cozzani E, Micalizzi C, et al. Chilblain-like lesions after COVID-19 vaccination: a case series. Acta Derm Venereol. 2022;102:adv00711. doi:10.2340/actadv.v102.2076
- Ortigosa LCM, Lenzoni FC, Suárez MV, et al. Hypersensitivity reaction to hyaluronic acid dermal filler after COVID-19 vaccination: a series of cases in São Paulo, Brazil. Int J Infect Dis. 2022;116:268-270.
- Agaronov A, Makdesi C, Hall CS. Acute generalized exanthematous pustulosis induced by Moderna COVID-19 messenger RNA vaccine. JAAD Case Rep. 2021;16:96-97.
- Dash S, Sirka CS, Mishra S, et al. COVID-19 vaccine-induced Stevens-Johnson syndrome. Clin Exp Dermatol. 2021;46:1615-1617.
- Huang Y, Tsai TF. Exacerbation of psoriasis following COVID-19 vaccination: report from a single center. Front Med (Lausanne). 2021;8:812010.
- Elamin S, Hinds F, Tolland J. De novo generalized pustular psoriasis following Oxford-AstraZeneca COVID-19 vaccine. Clin Exp Dermatol 2022;47:153-155.
- Abdelmaksoud A, Wollina U, Temiz SA, et al. SARS-CoV-2 vaccination-induced cutaneous vasculitis: report of two new cases and literature review. Dermatol Ther. 2022;35:E15458.
- Fritzen M, Funchal GDG, Luiz MO, et al. Leukocytoclastic vasculitis after exposure to COVID-19 vaccine. An Bras Dermatol. 2022;97:118-121.
- Vassallo C, Boveri E, Brazzelli V, et al. Cutaneous lymphocytic vasculitis after administration of COVID-19 mRNA vaccine. Dermatol Ther. 2021;34:E15076.
- Nazzaro G, Maronese CA. Urticarial vasculitis following mRNA anti-COVID-19 vaccine. Dermatol Ther. 2022;35:E15282.
- Hoshina D, Orita A. Sweet syndrome after severe acute respiratory syndrome coronavirus 2 mRNA vaccine: a case report and literature review. J Dermatol. 2022;49:E175-E176.
- Lemoine C, Padilla C, Krampe N, et al. Systemic lupus erythematous after Pfizer COVID-19 vaccine: a case report. Clin Rheumatol. 2022;41:1597-1601.
- Nguyen B, Lalama MJ, Gamret AC, et al. Cutaneous symptoms of connective tissue diseases after COVID-19 vaccination: a systematic review. Int J Dermatol. 2022;61:E238-E241.
- Gallo G, Mastorino L, Tonella L, et al. Alopecia areata after COVID-19 vaccination. Clin Exp Vaccine Res. 2022;11:129-132.
- Norimatsu Y, Norimatsu Y. A severe case of Trichophyton rubrum-caused dermatomycosis exacerbated after COVID-19 vaccination that had to be differentiated from pustular psoriasis. Med Mycol Case Rep. 2022;36:19-22.
- Yang K, Prussick L, Hartman R, et al. Acantholytic dyskeratosis post-COVID vaccination. Am J Dermatopathol. 2022;44:E61-E63.
- Koumaki D, Marinos L, Nikolaou V, et al. Lymphomatoid papulosis (LyP) after AZD1222 and BNT162b2 COVID-19 vaccines. Int J Dermatol. 2022;61:900-902.
- World Health Organization. Statement on the fourteenth meeting of the International Health Regulations (2005) Emergency Committee regarding the coronavirus disease (COVID-19) pandemic. Published January 30, 2023. Accessed April 12, 2023. https://www.who.int/news/item/30-01-2023-statement-on-the-fourteenth-meeting-of-the-international-health-regulations-(2005)-emergency-committee-regarding-the-coronavirus-disease-(covid-19)-pandemic
- Ashraf UM, Abokor AA, Edwards JM, et al. SARS-CoV-2, ACE2 expression, and systemic organ invasion. Physiol Genomics. 2021;53:51-60.
- Ganier C, Harun N, Peplow I, et al. Angiotensin-converting enzyme 2 expression is detectable in keratinocytes, cutaneous appendages, and blood vessels by multiplex RNA in situ hybridization. Adv Skin Wound Care. 2022;35:219-223.
- Ulloa AC, Buchan SA, Daneman N, et al. Estimates of SARS-CoV-2 omicron variant severity in Ontario, Canada. JAMA. 2022;327:1286-1288.
- World Health Organization. Coronavirus (COVID-19) Dashboard. Accessed April 6, 2023. https://covid19.who.int
- Guan WJ, Ni ZY, Hu Y, et al; China Medical Treatment Expert Group for COVID-19. clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708-1720.
- Recalcati S. Cutaneous manifestations in COVID-9: a first perspective. J Eur Acad Dermatol Venereol. 2020;34:E212-E213.
- Freeman EE, McMahon DE, Lipoff JB, et al. The spectrum of COVID-19-associated dermatologic manifestations: an international registry of 716 patients from 31 countries. J Am Acad Dermatol. 2020;83:1118-1129.
- Freeman EE, Chamberlin GC, McMahon DE, et al. Dermatology COVID-19 registries: updates and future directions. Dermatol Clin. 2021;39:575-585.
- Guelimi R, Salle R, Dousset L, et al. Non-acral skin manifestations during the COVID-19 epidemic: COVIDSKIN study by the French Society of Dermatology. J Eur Acad Dermatol Venereol. 2021;35:E539-E541.
- Marzano AV, Genovese G, Moltrasio C, et al; Italian Skin COVID-19 Network of the Italian Society of Dermatology and Sexually Transmitted Diseases. The clinical spectrum of COVID-19 associated cutaneous manifestations: an Italian multicenter study of 200 adult patients. J Am Acad Dermatol. 2021;84:1356-1363.
- Sugai T, Fujita Y, Inamura E, et al. Prevalence and patterns of cutaneous manifestations in 1245 COVID-19 patients in Japan: a single-centre study. J Eur Acad Dermatol Venereol. 2022;36:E522-E524.
- Holmes Z, Courtney A, Lincoln M, et al. Rash morphology as a predictor of COVID‐19 severity: a systematic review of the cutaneous manifestations of COVID‐19. Skin Health Dis. 2022;2:E120. doi:10.1002/ski2.120
- Galván Casas C, Català A, Carretero Hernández G, et al. Classification of the cutaneous manifestations of COVID-19: a rapid prospective nationwide consensus study in Spain with 375 cases. Br J Dermatol. 2020;183:71-77.
- Garduño‑Soto M, Choreño-Parra, Cazarin-Barrientos Dermatological aspects of SARS‑CoV‑2 infection: mechanisms and manifestations. Arch Dermatol Res. 2021;313:611-622.
- Huynh T, Sanchez-Flores X, Yau J, et al. Cutaneous manifestations of SARS-CoV-2 Infection. Am J Clin Dermatol. 2022;23:277-286.
- Kanitakis J, Lesort C, Danset M, et al.
2020; 83:870-875. - Kolivras A, Thompson C, Pastushenko I, et al. A clinicopathological description of COVID-19-induced chilblains (COVID-toes) correlated with a published literature review. J Cutan Pathol. 2022;49:17-28.
- Roca-Ginés J, Torres-Navarro I, Sánchez-Arráez J, et al. Assessment of acute acral lesions in a case series of children and adolescents during the COVID-19 pandemic. 2020;156:992-997.
- Le Cleach L, Dousset L, Assier H, et al; French Society of Dermatology. Most chilblains observed during the COVID-19 outbreak occur in patients who are negative for COVID-19 on polymerase chain reaction and serology testing. Br J Dermatol. 2020;183:866-874.
- Discepolo V, Catzola A, Pierri L, et al. Bilateral chilblain-like lesions of the toes characterized by microvascular remodeling in adolescents during the COVID-19 pandemic. JAMA Netw Open. 2021;4:E2111369.
- Gehlhausen JR, Little AJ, Ko CJ, et al. Lack of association between pandemic chilblains and SARS-CoV-2 infection. Proc Natl Acad Sci U S A. 2022;119:e2122090119.
- Neri, Virdi, Corsini, et al Major cluster of paediatric ‘true’ primary chilblains during the COVID-19 pandemic: a consequence of lifestyle changes due to lockdown. J Eur Acad Dermatol Venereol. 2020;34:2630-2635.
- De Greef A, Choteau M, Herman A, et al. Chilblains observed during the COVID-19 pandemic cannot be distinguished from the classic, cold-related chilblains. Eur J Dermatol. 2022;32:377-383.
- Colmenero I, Santonja C, Alonso-Riaño M, et al. SARS-CoV-2 endothelial infection causes COVID-19 chilblains: histopathological, immunohistochemical and ultrastructural study of seven paediatric cases. Br J Dermatol. 2020;183:729-737.
- Quintero-Bustos G, Aguilar-Leon D, Saeb-Lima M. Histopathological and immunohistochemical characterization of skin biopsies from 41 SARS-CoV-2 (+) patients: experience in a Mexican concentration institute: a case series and literature review. Am J Dermatopathol. 2022;44:327-337.
- Arkin LM, Moon JJ, Tran JM, et al; COVID Human Genetic Effort. From your nose to your toes: a review of severe acute respiratory syndrome coronavirus 2 pandemic-associated pernio. J Invest Dermatol. 2021;141:2791-2796.
- Frumholtz L, Bouaziz JD, Battistella M, et al; Saint-Louis CORE (COvid REsearch). Type I interferon response and vascular alteration in chilblain-like lesions during the COVID-19 outbreak. Br J Dermatol. 2021;185:1176-1185.
- Hubiche T, Cardot-Leccia N, Le Duff F, et al. Clinical, laboratory, and interferon-alpha response characteristics of patients with chilblain-like lesions during the COVID-19 pandemic. JAMA Dermatol. 2021;157:202-206.
- Lesort C, Kanitakis J, Villani A, et al. COVID-19 and outbreak of chilblains: are they related? J Eur Acad Dermatol Venereol. 2020;34:E757-E758.
- Sanchez A, Sohier P, Benghanem S, et al. Digitate papulosquamous eruption associated with severe acute respiratory syndrome coronavirus 2 infection. JAMA Dermatol. 2020;156:819-820.
- Drago F, Broccolo F, Ciccarese G. Pityriasis rosea, pityriasis rosea-like eruptions, and herpes zoster in the setting of COVID-19 and COVID-19 vaccination. Clin Dermatol. 2022;S0738-081X(22)00002-5.
- Dursun R, Temiz SA. The clinics of HHV-6 infection in COVID-19 pandemic: pityriasis rosea and Kawasaki disease. Dermatol Ther. 2020;33:E13730.
- Nuno-Gonzalez A, Magaletsky K, Feito Rodríguez M, et al. Palmoplantar erythrodysesthesia: a diagnostic sign of COVID-19. J Eur Acad Dermatol Venereol. 2021;35:e247-e249.
- Sil A, Panigrahi A, Chandra A, et al. “COVID nose”: a unique post-COVID pigmentary sequelae reminiscing Chik sign: a descriptive case series. J Eur Acad Dermatol Venereol. 2022;36:E419-E421.
- Starace M, Iorizzo M, Sechi A, et al. Trichodynia and telogen effluvium in COVID-19 patients: results of an international expert opinion survey on diagnosis and management. JAAD Int. 2021;5:11-18.
- Wong-Chew RM, Rodríguez Cabrera EX, Rodríguez Valdez CA, et al. Symptom cluster analysis of long COVID-19 in patients discharged from the Temporary COVID-19 Hospital in Mexico City. Ther Adv Infect Dis. 2022;9:20499361211069264.
- Bardazzi F, Guglielmo A, Abbenante D, et al. New insights into alopecia areata during COVID-19 pandemic: when infection or vaccination could play a role. J Cosmet Dermatol. 2022;21:1796-1798.
- Christensen RE, Jafferany M. Association between alopecia areata and COVID-19: a systematic review. JAAD Int. 2022;7:57-61.
- Wollina U, Kanitakis J, Baran R. Nails and COVID-19: a comprehensive review of clinical findings and treatment. Dermatol Ther. 2021;34:E15100.
- Méndez-Flores S, Zaladonis A, Valdes-Rodriguez R. COVID-19 and nail manifestation: be on the lookout for the red half-moon nail sign. Int J Dermatol. 2020;59:1414.
- Alobaida S, Lam JM. Beau lines associated with COVID-19. CMAJ. 2020;192:E1040.
- Durmaz EÖ, Demirciog˘lu D. Fluorescence in the sclera, nails, and teeth secondary to favipiravir use for COVID-19 infections. J Clin Aesthet Dermatol. 2022;15:35-37.
- Brumfiel CM, DiLorenzo AM, Petronic-Rosic VM. Dermatologic manifestations of COVID-19-associated multisystem inflammatory syndrome in children. Clin Dermatol. 2021;39:329-333.
- Akçay N, Topkarcı Z, Menentog˘lu ME, et al. New dermatological findings of MIS-C: can mucocutaneous involvement be associated with severe disease course? Australas J Dermatol. 2022;63:228-234. doi:10.1111/ajd.13819
- Vogel TP, Top KA, Karatzios C, et al. Multisystem inflammatory syndrome in children and adults (MIS-C/A): case definition & guidelines for data collection, analysis, and presentation of immunization safety data. Vaccine. 2021;39:3037-3049.
- Conforti C, Dianzani C, Agozzino M, et al. Cutaneous manifestations in confirmed COVID-19 patients: a systematic review. Biology (Basel). 2020;9:449.
- Hubiche T, Le Duff F, Fontas E, et al. Relapse of chilblain-like lesions during the second wave of the COVID-19 pandemic: a cohort follow-up. Br J Dermatol. 2021;185:858-859.
- Fernandez-Nieto, Ortega-Quijano, Suarez-Valle, et al Lack of skin manifestations in COVID-19 hospitalized patients during the second epidemic wave in Spain: a possible association with a novel SARS-CoV-2 variant: a cross-sectional study. J Eur Acad Dermatol Venereol. 2021;35:E183-E185.
- Martinez-LopezA, Cuenca-Barrales, Montero-Vilchezet al Review of adverse cutaneous reactions of pharmacologic interventions for COVID-19: a guide for the dermatologist. J Am Acad Dermatol. 2020;83:1738-1748.
- Cutaneous side-effects of the potential COVID-19 drugs. Dermatol Ther. 2020;33:E13476.
- Mawhirt SL, Frankel D, Diaz AM. Cutaneous manifestations in adult patients with COVID-19 and dermatologic conditions related to the COVID-19 pandemic in health care workers. Curr Allerg Asthma Rep. 2020;20:75.
- Nguyen C, Young FG, McElroy D, et al. Personal protective equipment and adverse dermatological reactions among healthcare workers: survey observations from the COVID-19 pandemic. Medicine (Baltimore). 2022;101:E29003.
- Rathi SK, Dsouza JM. Maskne: a new acne variant in COVID-19 era. Indian J Dermatol. 2022;67:552-555.
- Damiani G, Girono L, Grada A, et al. COVID-19 related masks increase severity of both acne (maskne) and rosacea (mask rosacea): multi-center, real-life, telemedical, and observational prospective study. Dermatol Ther. 2021;34:E14848.
- Aram K, Patil A, Goldust M, et al. COVID-19 and exacerbation of dermatological diseases: a review of the available literature. Dermatol Ther. 2021;34:E15113.
- Samotij D, Gawron E, Szcze˛ch J, et al. Acrodermatitis continua of Hallopeau evolving into generalized pustular psoriasis following COVID-19: a case report of a successful treatment with infliximab in combination with acitretin. Biologics. 2021;15:107-113.
- Demiri J, Abdo M, Tsianakas A. Erythrodermic psoriasis after COVID-19 [in German]. Hautarzt. 2022;73:156-159.
- de Wijs LEM, Joustra MM, Olydam JI, et al. COVID-19 in patients with cutaneous immune-mediated diseases in the Netherlands: real-world observational data. J Eur Acad Dermatol Venereol. 2021;35:E173-E176.
- Marques NP, Maia CMF, Marques NCT, et al. Continuous increase of herpes zoster cases in Brazil during the COVID-19 pandemic. Oral Surg Oral Med Oral Pathol Oral Radiol. 2022;133:612-614.
- Rinaldi F, Trink A, Giuliani G, et al. Italian survey for the evaluation of the effects of coronavirus disease 2019 (COVID-19) pandemic on alopecia areata recurrence. Dermatol Ther (Heidelb). 2021;11:339-345.
- Rudnicka L, Rakowska A, Waskiel-Burnat A, et al. Mild-to-moderate COVID-19 is not associated with worsening of alopecia areata: a retrospective analysis of 32 patients. J Am Acad Dermatol. 2021;85:723-725.
- Drenovska K, Shahid M, Mateeva V, et al. Case report: Rowell syndrome-like flare of cutaneous lupus erythematosus following COVID-19 infection. Front Med (Lausanne). 2022;9:815743.
- Kawabe R, Tonomura K, Kotobuki Y, et al. Exacerbation of livedoid vasculopathy after coronavirus disease 2019. Eur J Dermatol. 2022;32:129-131. doi:10.1684/ejd.2022.4200
- McMahon DE, Kovarik CL, Damsky W, et al. Clinical and pathologic correlation of cutaneous COVID-19 vaccine reactions including V-REPP: a registry-based study. J Am Acad Dermatol. 2022;86:113-121.
- Avallone G, Quaglino P, Cavallo F, et al. SARS-CoV-2 vaccine-related cutaneous manifestations: a systematic review. Int J Dermatol. 2022;61:1187-1204. doi:10.1111/ijd.16063
- Gambichler T, Boms S, Susok L, et al. Cutaneous findings following COVID-19 vaccination: review of world literature and own experience. J Eur Acad Dermatol Venereol. 2022;36:172-180.
- Kroumpouzos G, Paroikaki ME, Yumeen S, et al. Cutaneous complications of mRNA and AZD1222 COVID-19 vaccines: a worldwide review. Microorganisms. 2022;10:624.
- Robinson L,Fu X,Hashimoto D, et al. Incidence of cutaneous reactions after messenger RNA COVID-19 vaccines. 2021;
- Wollina U, Chiriac A, Kocic H, et al. Cutaneous and hypersensitivity reactions associated with COVID-19 vaccination: a narrative review. Wien Med Wochenschr. 2022;172:63-69.
- Wei TS. Cutaneous reactions to COVID-19 vaccines: a review. JAAD Int. 2022;7:178-186.
- Katsikas Triantafyllidis K, Giannos P, Mian IT, et al. Varicella zoster virus reactivation following COVID-19 vaccination: a systematic review of case reports. Vaccines (Basel). 2021;9:1013.
- Maronese CA, Caproni M, Moltrasio C, et al. Bullous pemphigoid associated with COVID-19 vaccines: an Italian multicentre study. Front Med (Lausanne). 2022;9:841506.
- Cavazos A, Deb A, Sharma U, et al. COVID toes following vaccination. Proc (Bayl Univ Med Cent). 2022;35:476-479.
- Lesort C, Kanitakis J, Danset M, et al. Chilblain-like lesions after BNT162b2 mRNA COVID-19 vaccine: a case report suggesting that ‘COVID toes’ are due to the immune reaction to SARS-CoV-2. J Eur Acad Dermatol Venereol. 2021;35:E630-E632.
- Russo R, Cozzani E, Micalizzi C, et al. Chilblain-like lesions after COVID-19 vaccination: a case series. Acta Derm Venereol. 2022;102:adv00711. doi:10.2340/actadv.v102.2076
- Ortigosa LCM, Lenzoni FC, Suárez MV, et al. Hypersensitivity reaction to hyaluronic acid dermal filler after COVID-19 vaccination: a series of cases in São Paulo, Brazil. Int J Infect Dis. 2022;116:268-270.
- Agaronov A, Makdesi C, Hall CS. Acute generalized exanthematous pustulosis induced by Moderna COVID-19 messenger RNA vaccine. JAAD Case Rep. 2021;16:96-97.
- Dash S, Sirka CS, Mishra S, et al. COVID-19 vaccine-induced Stevens-Johnson syndrome. Clin Exp Dermatol. 2021;46:1615-1617.
- Huang Y, Tsai TF. Exacerbation of psoriasis following COVID-19 vaccination: report from a single center. Front Med (Lausanne). 2021;8:812010.
- Elamin S, Hinds F, Tolland J. De novo generalized pustular psoriasis following Oxford-AstraZeneca COVID-19 vaccine. Clin Exp Dermatol 2022;47:153-155.
- Abdelmaksoud A, Wollina U, Temiz SA, et al. SARS-CoV-2 vaccination-induced cutaneous vasculitis: report of two new cases and literature review. Dermatol Ther. 2022;35:E15458.
- Fritzen M, Funchal GDG, Luiz MO, et al. Leukocytoclastic vasculitis after exposure to COVID-19 vaccine. An Bras Dermatol. 2022;97:118-121.
- Vassallo C, Boveri E, Brazzelli V, et al. Cutaneous lymphocytic vasculitis after administration of COVID-19 mRNA vaccine. Dermatol Ther. 2021;34:E15076.
- Nazzaro G, Maronese CA. Urticarial vasculitis following mRNA anti-COVID-19 vaccine. Dermatol Ther. 2022;35:E15282.
- Hoshina D, Orita A. Sweet syndrome after severe acute respiratory syndrome coronavirus 2 mRNA vaccine: a case report and literature review. J Dermatol. 2022;49:E175-E176.
- Lemoine C, Padilla C, Krampe N, et al. Systemic lupus erythematous after Pfizer COVID-19 vaccine: a case report. Clin Rheumatol. 2022;41:1597-1601.
- Nguyen B, Lalama MJ, Gamret AC, et al. Cutaneous symptoms of connective tissue diseases after COVID-19 vaccination: a systematic review. Int J Dermatol. 2022;61:E238-E241.
- Gallo G, Mastorino L, Tonella L, et al. Alopecia areata after COVID-19 vaccination. Clin Exp Vaccine Res. 2022;11:129-132.
- Norimatsu Y, Norimatsu Y. A severe case of Trichophyton rubrum-caused dermatomycosis exacerbated after COVID-19 vaccination that had to be differentiated from pustular psoriasis. Med Mycol Case Rep. 2022;36:19-22.
- Yang K, Prussick L, Hartman R, et al. Acantholytic dyskeratosis post-COVID vaccination. Am J Dermatopathol. 2022;44:E61-E63.
- Koumaki D, Marinos L, Nikolaou V, et al. Lymphomatoid papulosis (LyP) after AZD1222 and BNT162b2 COVID-19 vaccines. Int J Dermatol. 2022;61:900-902.
- World Health Organization. Statement on the fourteenth meeting of the International Health Regulations (2005) Emergency Committee regarding the coronavirus disease (COVID-19) pandemic. Published January 30, 2023. Accessed April 12, 2023. https://www.who.int/news/item/30-01-2023-statement-on-the-fourteenth-meeting-of-the-international-health-regulations-(2005)-emergency-committee-regarding-the-coronavirus-disease-(covid-19)-pandemic
Practice Points
- During the COVID-19 pandemic, several skin diseases were reported in association with this new infectious disease and were classified mainly according to their morphologic aspect. However, the pathogenetic mechanisms often are unclear and the causal link of the culprit virus (SARS-CoV-2) not always well established.
- Currently, most skin manifestations related to COVID-19 are reported after vaccination against COVID-19; remarkably, many of them are similar to those attributed to the natural infection.
Painful Nodules With a Crawling Sensation
The Diagnosis: Cutaneous Furuncular Myiasis
Histopathology of the punch biopsy showed an undulating chitinous exoskeleton and pigmented spines (setae) protruding from the exoskeleton with associated superficial perivascular lymphohistiocytic infiltrates on hematoxylin and eosin stain (Figure 1). Live insect larvae were observed and extracted, which immediately relieved the crawling sensation (Figure 2). Light microscopy of the larva showed a row of hooks surrounding a tapered body with a head attached anteriorly (Figure 3).
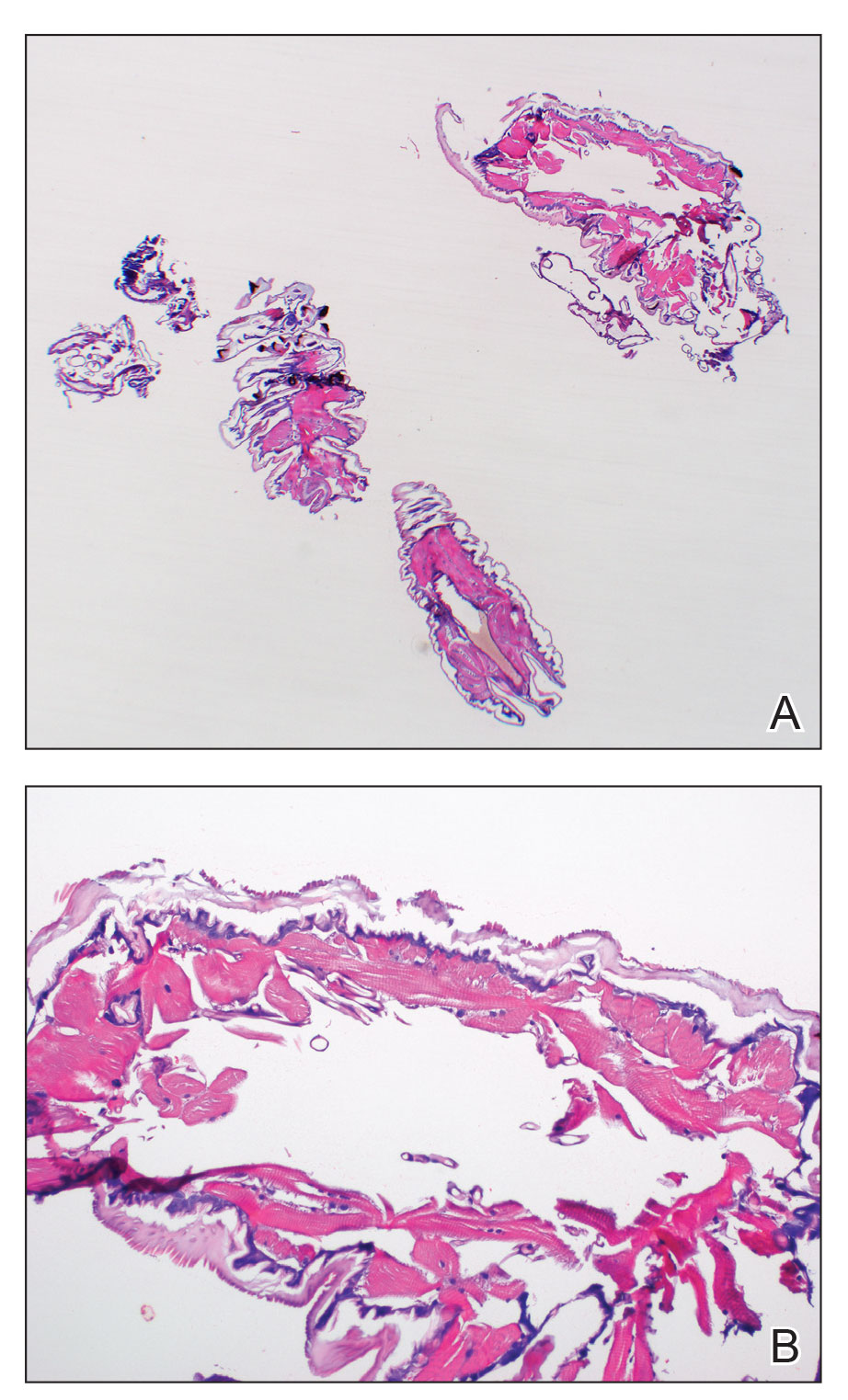
Myiasis is a parasitic infestation of the dipterous fly’s larvae in the host organ and tissue. There are 5 types of myiasis based on the location of the infestation: wound myiasis occurs with egg infestations on an open wound; furuncular myiasis results from egg placement by penetration of healthy skin by a mosquito vector; plaque myiasis comprises the placement of eggs on clothing through several maggots and flies; creeping myiasis involves the Gasterophilus fly delivering the larva intradermally; and body cavity myiasis may develop in the orbit, nasal cavity, urogenital system, and gastrointestinal tract.1-3

Furuncular myiasis infestation occurs via a complex life cycle in which mosquitoes act as a vector and transfer the eggs to the human or animal host.1-3 Botfly larvae then penetrate the skin and reside within the subdermis to mature. Adults then emerge after 1 month to repeat the cycle.1 Dermatobia hominis and Cordylobia anthropophaga are the most common causes of furuncular myiasis.2,3 Furuncular myiasis commonly presents in travelers that are returning from tropical countries. Initially, an itching erythematous papule develops. After the larvae mature, they can appear as boil-like lesions with a small central punctum.1-3 Dermoscopy can be utilized for visualization of different larvae anatomy such as a furuncularlike lesion, spines, and posterior breathing spiracle from the central punctum.4
Our patient’s recent travel to the Amazon in Brazil, clinical history, and histopathologic findings ruled out other differential diagnoses such as cutaneous larva migrans, gnathostomiasis, loiasis, and tungiasis.
Treatment is curative with the extraction of the intact larva from the nodule. Localized skin anesthetic injection can be used to bulge the larva outward for easier extraction. A single dose of ivermectin 15 mg can treat the parasitic infestation of myiasis.1-3
- John DT, Petri WA, Markell EK, et al. Markell and Voge’s Medical Parasitology. 9th ed. Saunders Elsevier; 2006.
- Caissie R, Beaulieu F, Giroux M, et al. Cutaneous myiasis: diagnosis, treatment, and prevention. J Oral Maxillofac Surg. 2008;66:560-568.
- Lachish T, Marhoom E, Mumcuoglu KY, et al. Myiasis in travelers. J Travel Med. 2015;22:232-236.
- Mello C, Magalhães R. Triangular black dots in dermoscopy of furuncular myiasis. JAAD Case Rep. 2021;12:49-50.
The Diagnosis: Cutaneous Furuncular Myiasis
Histopathology of the punch biopsy showed an undulating chitinous exoskeleton and pigmented spines (setae) protruding from the exoskeleton with associated superficial perivascular lymphohistiocytic infiltrates on hematoxylin and eosin stain (Figure 1). Live insect larvae were observed and extracted, which immediately relieved the crawling sensation (Figure 2). Light microscopy of the larva showed a row of hooks surrounding a tapered body with a head attached anteriorly (Figure 3).

Myiasis is a parasitic infestation of the dipterous fly’s larvae in the host organ and tissue. There are 5 types of myiasis based on the location of the infestation: wound myiasis occurs with egg infestations on an open wound; furuncular myiasis results from egg placement by penetration of healthy skin by a mosquito vector; plaque myiasis comprises the placement of eggs on clothing through several maggots and flies; creeping myiasis involves the Gasterophilus fly delivering the larva intradermally; and body cavity myiasis may develop in the orbit, nasal cavity, urogenital system, and gastrointestinal tract.1-3

Furuncular myiasis infestation occurs via a complex life cycle in which mosquitoes act as a vector and transfer the eggs to the human or animal host.1-3 Botfly larvae then penetrate the skin and reside within the subdermis to mature. Adults then emerge after 1 month to repeat the cycle.1 Dermatobia hominis and Cordylobia anthropophaga are the most common causes of furuncular myiasis.2,3 Furuncular myiasis commonly presents in travelers that are returning from tropical countries. Initially, an itching erythematous papule develops. After the larvae mature, they can appear as boil-like lesions with a small central punctum.1-3 Dermoscopy can be utilized for visualization of different larvae anatomy such as a furuncularlike lesion, spines, and posterior breathing spiracle from the central punctum.4

Our patient’s recent travel to the Amazon in Brazil, clinical history, and histopathologic findings ruled out other differential diagnoses such as cutaneous larva migrans, gnathostomiasis, loiasis, and tungiasis.
Treatment is curative with the extraction of the intact larva from the nodule. Localized skin anesthetic injection can be used to bulge the larva outward for easier extraction. A single dose of ivermectin 15 mg can treat the parasitic infestation of myiasis.1-3
The Diagnosis: Cutaneous Furuncular Myiasis
Histopathology of the punch biopsy showed an undulating chitinous exoskeleton and pigmented spines (setae) protruding from the exoskeleton with associated superficial perivascular lymphohistiocytic infiltrates on hematoxylin and eosin stain (Figure 1). Live insect larvae were observed and extracted, which immediately relieved the crawling sensation (Figure 2). Light microscopy of the larva showed a row of hooks surrounding a tapered body with a head attached anteriorly (Figure 3).

Myiasis is a parasitic infestation of the dipterous fly’s larvae in the host organ and tissue. There are 5 types of myiasis based on the location of the infestation: wound myiasis occurs with egg infestations on an open wound; furuncular myiasis results from egg placement by penetration of healthy skin by a mosquito vector; plaque myiasis comprises the placement of eggs on clothing through several maggots and flies; creeping myiasis involves the Gasterophilus fly delivering the larva intradermally; and body cavity myiasis may develop in the orbit, nasal cavity, urogenital system, and gastrointestinal tract.1-3

Furuncular myiasis infestation occurs via a complex life cycle in which mosquitoes act as a vector and transfer the eggs to the human or animal host.1-3 Botfly larvae then penetrate the skin and reside within the subdermis to mature. Adults then emerge after 1 month to repeat the cycle.1 Dermatobia hominis and Cordylobia anthropophaga are the most common causes of furuncular myiasis.2,3 Furuncular myiasis commonly presents in travelers that are returning from tropical countries. Initially, an itching erythematous papule develops. After the larvae mature, they can appear as boil-like lesions with a small central punctum.1-3 Dermoscopy can be utilized for visualization of different larvae anatomy such as a furuncularlike lesion, spines, and posterior breathing spiracle from the central punctum.4

Our patient’s recent travel to the Amazon in Brazil, clinical history, and histopathologic findings ruled out other differential diagnoses such as cutaneous larva migrans, gnathostomiasis, loiasis, and tungiasis.
Treatment is curative with the extraction of the intact larva from the nodule. Localized skin anesthetic injection can be used to bulge the larva outward for easier extraction. A single dose of ivermectin 15 mg can treat the parasitic infestation of myiasis.1-3
- John DT, Petri WA, Markell EK, et al. Markell and Voge’s Medical Parasitology. 9th ed. Saunders Elsevier; 2006.
- Caissie R, Beaulieu F, Giroux M, et al. Cutaneous myiasis: diagnosis, treatment, and prevention. J Oral Maxillofac Surg. 2008;66:560-568.
- Lachish T, Marhoom E, Mumcuoglu KY, et al. Myiasis in travelers. J Travel Med. 2015;22:232-236.
- Mello C, Magalhães R. Triangular black dots in dermoscopy of furuncular myiasis. JAAD Case Rep. 2021;12:49-50.
- John DT, Petri WA, Markell EK, et al. Markell and Voge’s Medical Parasitology. 9th ed. Saunders Elsevier; 2006.
- Caissie R, Beaulieu F, Giroux M, et al. Cutaneous myiasis: diagnosis, treatment, and prevention. J Oral Maxillofac Surg. 2008;66:560-568.
- Lachish T, Marhoom E, Mumcuoglu KY, et al. Myiasis in travelers. J Travel Med. 2015;22:232-236.
- Mello C, Magalhães R. Triangular black dots in dermoscopy of furuncular myiasis. JAAD Case Rep. 2021;12:49-50.
A 20-year-old man presented with progressively enlarging, painful lesions on the arm with a crawling sensation of 3 weeks’ duration. The lesions appeared after a recent trip to Brazil where he was hiking in the Amazon. He noted that the pain occurred suddenly and there was some serous drainage from the lesions. He denied any trauma to the area and reported no history of similar eruptions, treatments, or systemic symptoms. Physical examination revealed 2 tender erythematous nodules, each measuring 0.6 cm in diameter, with associated crust and a reported crawling sensation on the posterior aspect of the left arm. No drainage was seen. A punch biopsy was performed.
Diagnosis by dog: Canines detect COVID in schoolchildren with no symptoms
Scent-detecting dogs have long been used to sniff out medical conditions ranging from low blood sugar and cancer to malaria, impending seizures, and migraines – not to mention explosives and narcotics.
Recently, the sensitivity of the canine nose has been tested as a strategy for screening for SARS-CoV-2 infection in schoolchildren showing no outward symptoms of the virus. A pilot study led by Carol A. Glaser, DVM, MD, of the California Department of Public Health in Richmond, found that trained dogs had an accuracy of more than 95% for detecting the odor of volatile organic compounds, or VOCs, produced by COVID-infected individuals.
The authors believe that odor-based diagnosis with dogs could eventually provide a rapid, inexpensive, and noninvasive way to screen large groups for COVID-19 without the need for antigen testing.
“This is a new program with research ongoing, so it would be premature to consider it from a consumer’s perspective,” Dr. Glaser said in an interview. “However, the data look promising and we are hopeful we can continue to pilot various programs in various settings to see where, and if, dogs can be used for biomedical detection.”
In the lab and in the field
In a study published online in JAMA Pediatrics, Dr. Glaser’s group found that after 2 months’ training on COVID-19 scent samples in the laboratory, the dogs detected the presence of the virus more than 95% of the time. Antigen tests were used as a comparative reference.
In medical terms, the dogs achieved a greater than 95% accuracy on two important measures of effectiveness: sensitivity – a test’s ability to correctly detect the positive presence of disease – and specificity – the ability of a test to accurately rule out the presence of disease and identify as negative an uninfected person.
Next, the researchers piloted field tests in 50 visits at 27 schools from April 1 to May 25, 2022, to compare dogs’ detection ability with that of standard laboratory antigen testing. Participants in the completely voluntary screening numbered 1,558 and ranged in age from 9 to 17 years. Of these, 56% were girls and 89% were students. Almost 70% were screened at least twice.
Overall, the field test compared 3,897 paired antigen-vs.-dog screenings. The dogs accurately signaled the presence of 85 infections and ruled out 3,411 infections, for an overall accuracy of 90%. In 383 cases, however, they inaccurately signaled the presence of infection (false positives) and missed 18 actual infections (false negatives). That translated to a sensitivity in the field of 83%, considerably lower than that of their lab performance.
Direct screening of individuals with dogs outside of the lab involved circumstantial factors that likely contributed to decreased sensitivity and specificity, the authors acknowledged. These included such distractions as noise and the presence of excitable young children as well environmental conditions such as wind and other odors. What about dog phobia and dog hair allergy? “Dog screening takes only a few seconds per student and the dogs do not generally touch the participant as they run a line and sniff at ankles,” Dr. Glaser explained.
As for allergies, the rapid, ankle-level screening occurred in outdoor settings. “The chance of allergies is very low. This would be similar to someone who is out walking on the sidewalk and walks by a dog,” Dr. Glaser said.
Last year, a British trial of almost 4,000 adults tested six dogs trained to detect differences in VOCs between COVID-infected and uninfected individuals. Given samples from both groups, the dogs were able to distinguish between infected and uninfected samples with a sensitivity for detecting the virus ranging from 82% to 94% and a specificity for ruling it out of 76% to 92%. And they were able to smell the VOCs even when the viral load was low. The study also tested organic sensors, which proved even more accurate than the canines.
According to lead author James G. Logan, PhD, a disease control expert at the London School of Hygiene & Tropical Medicine in London, “Odour-based diagnostics using dogs and/or sensors may prove a rapid and effective tool for screening large numbers of people. Mathematical modelling suggests that dog screening plus a confirmatory PCR test could detect up to 89% of SARS-CoV-2 infections, averting up to 2.2 times as much transmission compared to isolation of symptomatic individuals only.”
Funding was provided by the Centers for Disease Control and Prevention Foundation (CDCF) to Early Alert Canines for the purchase and care of the dogs and the support of the handlers and trainers. The CDCF had no other role in the study. Coauthor Carol A. Edwards of Early Alert Canines reported receiving grants from the CDCF.
Scent-detecting dogs have long been used to sniff out medical conditions ranging from low blood sugar and cancer to malaria, impending seizures, and migraines – not to mention explosives and narcotics.
Recently, the sensitivity of the canine nose has been tested as a strategy for screening for SARS-CoV-2 infection in schoolchildren showing no outward symptoms of the virus. A pilot study led by Carol A. Glaser, DVM, MD, of the California Department of Public Health in Richmond, found that trained dogs had an accuracy of more than 95% for detecting the odor of volatile organic compounds, or VOCs, produced by COVID-infected individuals.
The authors believe that odor-based diagnosis with dogs could eventually provide a rapid, inexpensive, and noninvasive way to screen large groups for COVID-19 without the need for antigen testing.
“This is a new program with research ongoing, so it would be premature to consider it from a consumer’s perspective,” Dr. Glaser said in an interview. “However, the data look promising and we are hopeful we can continue to pilot various programs in various settings to see where, and if, dogs can be used for biomedical detection.”
In the lab and in the field
In a study published online in JAMA Pediatrics, Dr. Glaser’s group found that after 2 months’ training on COVID-19 scent samples in the laboratory, the dogs detected the presence of the virus more than 95% of the time. Antigen tests were used as a comparative reference.
In medical terms, the dogs achieved a greater than 95% accuracy on two important measures of effectiveness: sensitivity – a test’s ability to correctly detect the positive presence of disease – and specificity – the ability of a test to accurately rule out the presence of disease and identify as negative an uninfected person.
Next, the researchers piloted field tests in 50 visits at 27 schools from April 1 to May 25, 2022, to compare dogs’ detection ability with that of standard laboratory antigen testing. Participants in the completely voluntary screening numbered 1,558 and ranged in age from 9 to 17 years. Of these, 56% were girls and 89% were students. Almost 70% were screened at least twice.
Overall, the field test compared 3,897 paired antigen-vs.-dog screenings. The dogs accurately signaled the presence of 85 infections and ruled out 3,411 infections, for an overall accuracy of 90%. In 383 cases, however, they inaccurately signaled the presence of infection (false positives) and missed 18 actual infections (false negatives). That translated to a sensitivity in the field of 83%, considerably lower than that of their lab performance.
Direct screening of individuals with dogs outside of the lab involved circumstantial factors that likely contributed to decreased sensitivity and specificity, the authors acknowledged. These included such distractions as noise and the presence of excitable young children as well environmental conditions such as wind and other odors. What about dog phobia and dog hair allergy? “Dog screening takes only a few seconds per student and the dogs do not generally touch the participant as they run a line and sniff at ankles,” Dr. Glaser explained.
As for allergies, the rapid, ankle-level screening occurred in outdoor settings. “The chance of allergies is very low. This would be similar to someone who is out walking on the sidewalk and walks by a dog,” Dr. Glaser said.
Last year, a British trial of almost 4,000 adults tested six dogs trained to detect differences in VOCs between COVID-infected and uninfected individuals. Given samples from both groups, the dogs were able to distinguish between infected and uninfected samples with a sensitivity for detecting the virus ranging from 82% to 94% and a specificity for ruling it out of 76% to 92%. And they were able to smell the VOCs even when the viral load was low. The study also tested organic sensors, which proved even more accurate than the canines.
According to lead author James G. Logan, PhD, a disease control expert at the London School of Hygiene & Tropical Medicine in London, “Odour-based diagnostics using dogs and/or sensors may prove a rapid and effective tool for screening large numbers of people. Mathematical modelling suggests that dog screening plus a confirmatory PCR test could detect up to 89% of SARS-CoV-2 infections, averting up to 2.2 times as much transmission compared to isolation of symptomatic individuals only.”
Funding was provided by the Centers for Disease Control and Prevention Foundation (CDCF) to Early Alert Canines for the purchase and care of the dogs and the support of the handlers and trainers. The CDCF had no other role in the study. Coauthor Carol A. Edwards of Early Alert Canines reported receiving grants from the CDCF.
Scent-detecting dogs have long been used to sniff out medical conditions ranging from low blood sugar and cancer to malaria, impending seizures, and migraines – not to mention explosives and narcotics.
Recently, the sensitivity of the canine nose has been tested as a strategy for screening for SARS-CoV-2 infection in schoolchildren showing no outward symptoms of the virus. A pilot study led by Carol A. Glaser, DVM, MD, of the California Department of Public Health in Richmond, found that trained dogs had an accuracy of more than 95% for detecting the odor of volatile organic compounds, or VOCs, produced by COVID-infected individuals.
The authors believe that odor-based diagnosis with dogs could eventually provide a rapid, inexpensive, and noninvasive way to screen large groups for COVID-19 without the need for antigen testing.
“This is a new program with research ongoing, so it would be premature to consider it from a consumer’s perspective,” Dr. Glaser said in an interview. “However, the data look promising and we are hopeful we can continue to pilot various programs in various settings to see where, and if, dogs can be used for biomedical detection.”
In the lab and in the field
In a study published online in JAMA Pediatrics, Dr. Glaser’s group found that after 2 months’ training on COVID-19 scent samples in the laboratory, the dogs detected the presence of the virus more than 95% of the time. Antigen tests were used as a comparative reference.
In medical terms, the dogs achieved a greater than 95% accuracy on two important measures of effectiveness: sensitivity – a test’s ability to correctly detect the positive presence of disease – and specificity – the ability of a test to accurately rule out the presence of disease and identify as negative an uninfected person.
Next, the researchers piloted field tests in 50 visits at 27 schools from April 1 to May 25, 2022, to compare dogs’ detection ability with that of standard laboratory antigen testing. Participants in the completely voluntary screening numbered 1,558 and ranged in age from 9 to 17 years. Of these, 56% were girls and 89% were students. Almost 70% were screened at least twice.
Overall, the field test compared 3,897 paired antigen-vs.-dog screenings. The dogs accurately signaled the presence of 85 infections and ruled out 3,411 infections, for an overall accuracy of 90%. In 383 cases, however, they inaccurately signaled the presence of infection (false positives) and missed 18 actual infections (false negatives). That translated to a sensitivity in the field of 83%, considerably lower than that of their lab performance.
Direct screening of individuals with dogs outside of the lab involved circumstantial factors that likely contributed to decreased sensitivity and specificity, the authors acknowledged. These included such distractions as noise and the presence of excitable young children as well environmental conditions such as wind and other odors. What about dog phobia and dog hair allergy? “Dog screening takes only a few seconds per student and the dogs do not generally touch the participant as they run a line and sniff at ankles,” Dr. Glaser explained.
As for allergies, the rapid, ankle-level screening occurred in outdoor settings. “The chance of allergies is very low. This would be similar to someone who is out walking on the sidewalk and walks by a dog,” Dr. Glaser said.
Last year, a British trial of almost 4,000 adults tested six dogs trained to detect differences in VOCs between COVID-infected and uninfected individuals. Given samples from both groups, the dogs were able to distinguish between infected and uninfected samples with a sensitivity for detecting the virus ranging from 82% to 94% and a specificity for ruling it out of 76% to 92%. And they were able to smell the VOCs even when the viral load was low. The study also tested organic sensors, which proved even more accurate than the canines.
According to lead author James G. Logan, PhD, a disease control expert at the London School of Hygiene & Tropical Medicine in London, “Odour-based diagnostics using dogs and/or sensors may prove a rapid and effective tool for screening large numbers of people. Mathematical modelling suggests that dog screening plus a confirmatory PCR test could detect up to 89% of SARS-CoV-2 infections, averting up to 2.2 times as much transmission compared to isolation of symptomatic individuals only.”
Funding was provided by the Centers for Disease Control and Prevention Foundation (CDCF) to Early Alert Canines for the purchase and care of the dogs and the support of the handlers and trainers. The CDCF had no other role in the study. Coauthor Carol A. Edwards of Early Alert Canines reported receiving grants from the CDCF.
FROM JAMA PEDIATRICS
Long COVID mobile monitoring study hunts for answers
A new federal research project aims to answer lingering questions about long COVID using mobile monitoring devices to help track the condition.
The federally funded RECOVER Initiative expects to give out 10,000 sensors to people with long COVID to collect data in real time.
The hope is that researchers will be able to provide doctors and patients with a wealth of information to address gaps in knowledge about long COVID.
The project takes advantage of the approach other researchers have used to track patients’ health data on heart rate, exercise, and more using mobile monitoring devices such as Fitbits, smartwatches, and other remote sensors.
Researchers believe the initiative could be particularly useful for people with long COVID – whose symptoms come and go. They can use a wristband sensor to passively collect data in real time.
For a condition defined by its symptoms, that kind of data promises to be useful, experts said.
But not everyone has room in their budget for a smartwatch or a fitness tracker. Until recently, most clinical trials were BYOD: Bring your own device. At a time when researchers are trying to make sure that clinical trials reflect the diversity of the population, that leaves a lot of people out.
So, researchers are starting to supply subjects with their own monitors. The RECOVER Initiative expects to give out 10,000 sensors to people who are eligible based on race/ethnicity, income, and other demographic factors (rural residents for example). After 2 months, all people in the RECOVER study over the age of 13 will be eligible for the sensors.
The federal program builds on earlier research at places like The Scripps Institute, a center of research into remote monitoring. The institute supplied 7,000 monitors to people in an arm of the All of Us study, a 5-year-old multisite cohort that aims to collect medical information from 1 million people.
The devices went to people who have been historically underrepresented in biomedical research, said Scripps researchers, who plan to give out more this year.
In March of 2023, Scripps researchers published a study on the tracking data that found a significant post-COVID-19 drop in physical activity. But the data are incomplete because many people can’t always afford these devices. Most of the people in the study were “White, young, and active,” they wrote.
Researchers at an All of Us site at Vanderbilt University, which also used a BYOD approach, realized that they produced biased results. They reported their findings at the Pacific Symposium on Biocomputing in January.
“[The] majority of participants who provided Fitbit data reported being White and employed for wages,” they said. “However, these data represent participants who had their own Fitbit devices and consented to share EHR [electronic health record] data.”
Their solution: The program has begun providing Fitbit devices to all study participants who do not own one or cannot afford one.
Now, the web page for the All of Us study asks visitors to “Learn about the All of Us WEAR study. You could get a Fitbit at no cost! … As a part of the WEAR Study, you could receive a new Fitbit to wear at no cost to you. All of Us will be able to get the data the Fitbit collects. This data may help us understand how behavior impacts health.”
Jennifer Radin, PhD, an epidemiologist at Scripps Research Translational Institute, is heading up the DETECT study, which is a remote monitoring research project that has enrolled over 40,000 people who have their own sensors – be it a smartwatch or Fitbit. She was looking at remote monitoring for disease before COVID emerged.
Dr. Radin said she began researching remote sensing after working in public health and dealing with outdated data collection systems.
“They typically rely on case reports that are recorded by pen and paper and faxed or mailed in,” she said. “Then, they have to be entered into a database. “
In addition to offering objective data on a subject’s physical response to the infection, she said, the data collection can be long-term and continuous.
DETECT collects data on resting heart rate, which is unique to every person, and activity levels. Both measures are meaningful for those with long COVID. Her research found differences in sleep, heart rate, and activity between those with COVID and those without.
Joseph Kvedar, MD, is a Harvard Medical School researcher and the editor of NPJ Digital Medicine. He’s been studying digital health systems and called clinical research a “beachhead” for the use of data from monitors. But he also said problems remain that need to be worked out. The quality of the devices and their Bluetooth connections are better. But different devices measure different things, and a counted step can vary from person to person, he said. And the problems of the early days of electronic health records have not been fully resolved.
“We haven’t gotten to this universal language to connect all these things and make them relevant,” he said.
The All of Us researchers are working with the RECOVER project to address some of those issues. Usually not focused on a single condition, the All of Us researchers are testing a machine-learning approach for identifying long COVID.
A version of this article originally appeared on WebMD.com.
A new federal research project aims to answer lingering questions about long COVID using mobile monitoring devices to help track the condition.
The federally funded RECOVER Initiative expects to give out 10,000 sensors to people with long COVID to collect data in real time.
The hope is that researchers will be able to provide doctors and patients with a wealth of information to address gaps in knowledge about long COVID.
The project takes advantage of the approach other researchers have used to track patients’ health data on heart rate, exercise, and more using mobile monitoring devices such as Fitbits, smartwatches, and other remote sensors.
Researchers believe the initiative could be particularly useful for people with long COVID – whose symptoms come and go. They can use a wristband sensor to passively collect data in real time.
For a condition defined by its symptoms, that kind of data promises to be useful, experts said.
But not everyone has room in their budget for a smartwatch or a fitness tracker. Until recently, most clinical trials were BYOD: Bring your own device. At a time when researchers are trying to make sure that clinical trials reflect the diversity of the population, that leaves a lot of people out.
So, researchers are starting to supply subjects with their own monitors. The RECOVER Initiative expects to give out 10,000 sensors to people who are eligible based on race/ethnicity, income, and other demographic factors (rural residents for example). After 2 months, all people in the RECOVER study over the age of 13 will be eligible for the sensors.
The federal program builds on earlier research at places like The Scripps Institute, a center of research into remote monitoring. The institute supplied 7,000 monitors to people in an arm of the All of Us study, a 5-year-old multisite cohort that aims to collect medical information from 1 million people.
The devices went to people who have been historically underrepresented in biomedical research, said Scripps researchers, who plan to give out more this year.
In March of 2023, Scripps researchers published a study on the tracking data that found a significant post-COVID-19 drop in physical activity. But the data are incomplete because many people can’t always afford these devices. Most of the people in the study were “White, young, and active,” they wrote.
Researchers at an All of Us site at Vanderbilt University, which also used a BYOD approach, realized that they produced biased results. They reported their findings at the Pacific Symposium on Biocomputing in January.
“[The] majority of participants who provided Fitbit data reported being White and employed for wages,” they said. “However, these data represent participants who had their own Fitbit devices and consented to share EHR [electronic health record] data.”
Their solution: The program has begun providing Fitbit devices to all study participants who do not own one or cannot afford one.
Now, the web page for the All of Us study asks visitors to “Learn about the All of Us WEAR study. You could get a Fitbit at no cost! … As a part of the WEAR Study, you could receive a new Fitbit to wear at no cost to you. All of Us will be able to get the data the Fitbit collects. This data may help us understand how behavior impacts health.”
Jennifer Radin, PhD, an epidemiologist at Scripps Research Translational Institute, is heading up the DETECT study, which is a remote monitoring research project that has enrolled over 40,000 people who have their own sensors – be it a smartwatch or Fitbit. She was looking at remote monitoring for disease before COVID emerged.
Dr. Radin said she began researching remote sensing after working in public health and dealing with outdated data collection systems.
“They typically rely on case reports that are recorded by pen and paper and faxed or mailed in,” she said. “Then, they have to be entered into a database. “
In addition to offering objective data on a subject’s physical response to the infection, she said, the data collection can be long-term and continuous.
DETECT collects data on resting heart rate, which is unique to every person, and activity levels. Both measures are meaningful for those with long COVID. Her research found differences in sleep, heart rate, and activity between those with COVID and those without.
Joseph Kvedar, MD, is a Harvard Medical School researcher and the editor of NPJ Digital Medicine. He’s been studying digital health systems and called clinical research a “beachhead” for the use of data from monitors. But he also said problems remain that need to be worked out. The quality of the devices and their Bluetooth connections are better. But different devices measure different things, and a counted step can vary from person to person, he said. And the problems of the early days of electronic health records have not been fully resolved.
“We haven’t gotten to this universal language to connect all these things and make them relevant,” he said.
The All of Us researchers are working with the RECOVER project to address some of those issues. Usually not focused on a single condition, the All of Us researchers are testing a machine-learning approach for identifying long COVID.
A version of this article originally appeared on WebMD.com.
A new federal research project aims to answer lingering questions about long COVID using mobile monitoring devices to help track the condition.
The federally funded RECOVER Initiative expects to give out 10,000 sensors to people with long COVID to collect data in real time.
The hope is that researchers will be able to provide doctors and patients with a wealth of information to address gaps in knowledge about long COVID.
The project takes advantage of the approach other researchers have used to track patients’ health data on heart rate, exercise, and more using mobile monitoring devices such as Fitbits, smartwatches, and other remote sensors.
Researchers believe the initiative could be particularly useful for people with long COVID – whose symptoms come and go. They can use a wristband sensor to passively collect data in real time.
For a condition defined by its symptoms, that kind of data promises to be useful, experts said.
But not everyone has room in their budget for a smartwatch or a fitness tracker. Until recently, most clinical trials were BYOD: Bring your own device. At a time when researchers are trying to make sure that clinical trials reflect the diversity of the population, that leaves a lot of people out.
So, researchers are starting to supply subjects with their own monitors. The RECOVER Initiative expects to give out 10,000 sensors to people who are eligible based on race/ethnicity, income, and other demographic factors (rural residents for example). After 2 months, all people in the RECOVER study over the age of 13 will be eligible for the sensors.
The federal program builds on earlier research at places like The Scripps Institute, a center of research into remote monitoring. The institute supplied 7,000 monitors to people in an arm of the All of Us study, a 5-year-old multisite cohort that aims to collect medical information from 1 million people.
The devices went to people who have been historically underrepresented in biomedical research, said Scripps researchers, who plan to give out more this year.
In March of 2023, Scripps researchers published a study on the tracking data that found a significant post-COVID-19 drop in physical activity. But the data are incomplete because many people can’t always afford these devices. Most of the people in the study were “White, young, and active,” they wrote.
Researchers at an All of Us site at Vanderbilt University, which also used a BYOD approach, realized that they produced biased results. They reported their findings at the Pacific Symposium on Biocomputing in January.
“[The] majority of participants who provided Fitbit data reported being White and employed for wages,” they said. “However, these data represent participants who had their own Fitbit devices and consented to share EHR [electronic health record] data.”
Their solution: The program has begun providing Fitbit devices to all study participants who do not own one or cannot afford one.
Now, the web page for the All of Us study asks visitors to “Learn about the All of Us WEAR study. You could get a Fitbit at no cost! … As a part of the WEAR Study, you could receive a new Fitbit to wear at no cost to you. All of Us will be able to get the data the Fitbit collects. This data may help us understand how behavior impacts health.”
Jennifer Radin, PhD, an epidemiologist at Scripps Research Translational Institute, is heading up the DETECT study, which is a remote monitoring research project that has enrolled over 40,000 people who have their own sensors – be it a smartwatch or Fitbit. She was looking at remote monitoring for disease before COVID emerged.
Dr. Radin said she began researching remote sensing after working in public health and dealing with outdated data collection systems.
“They typically rely on case reports that are recorded by pen and paper and faxed or mailed in,” she said. “Then, they have to be entered into a database. “
In addition to offering objective data on a subject’s physical response to the infection, she said, the data collection can be long-term and continuous.
DETECT collects data on resting heart rate, which is unique to every person, and activity levels. Both measures are meaningful for those with long COVID. Her research found differences in sleep, heart rate, and activity between those with COVID and those without.
Joseph Kvedar, MD, is a Harvard Medical School researcher and the editor of NPJ Digital Medicine. He’s been studying digital health systems and called clinical research a “beachhead” for the use of data from monitors. But he also said problems remain that need to be worked out. The quality of the devices and their Bluetooth connections are better. But different devices measure different things, and a counted step can vary from person to person, he said. And the problems of the early days of electronic health records have not been fully resolved.
“We haven’t gotten to this universal language to connect all these things and make them relevant,” he said.
The All of Us researchers are working with the RECOVER project to address some of those issues. Usually not focused on a single condition, the All of Us researchers are testing a machine-learning approach for identifying long COVID.
A version of this article originally appeared on WebMD.com.
Dried blood spot test validated for HIV, hep B, and hep C
A test that uses a single drop of dried blood to detect HIV, hepatitis B virus, and HCV has been validated and is now in use in some high-risk settings in Denmark, according to research presented at the annual European Congress of Clinical Microbiology & Infectious Diseases.
Molecular biologist Stephen Nilsson-Møller, MSc, and colleagues at the department of clinical microbiology, Copenhagen University Hospital, developed and validated the test, known as the Dried Blood Spot (DBS), for HIV, HBV, and HCV.
The “test that can detect low viral loads for all three viruses from a single drop of blood, and can be done using existing hospital equipment,” Mr. Nilsson-Møller said in an interview. “Importantly, it does not require venipuncture, but can be done from a drop of dried blood from the finger.”
He highlighted the utility of the new test in more challenging settings. “This method is particularly useful in high-risk settings such as homeless shelters, drug rehabilitation centers, and prisons, where needles might be misused, and it can be difficult to convince people to have the more invasive test.”
“Also, in some places – such as in low- and middle-income settings – there is a distinct risk of ruining blood samples before analysis due to limited refrigeration for transit and storage,” he added. “[Standard] blood samples need to be analyzed within 6 hours when kept at room temperature, while dried blood spots can last for 9 months at room temperature and can be mailed to a laboratory with the right equipment to analyze it.”
Tiny amounts of virus detected
Mr. Nilsson-Møller was tasked with developing a test for use by the university’s department of infectious diseases to screen people in high-risk settings in the capital region of Copenhagen. The work forms part of a PhD project by Jonas Demant at the University of Copenhagen, for which he is screening for HIV, HBV, and HCV in drug rehabilitation centers, prisons, and homeless shelters.
The study is the first to use the Hologic Panther system (a nucleic acid amplification test) combining all three viruses, Mr. Nilsson-Møller pointed out. “A tiny amount of virus can be detected because it is a very sensitive platform using transcription-mediated amplification.”
“If it detects low amounts of virus, it will create many copies very quickly, creating a signal that tells us that the sample is positive,” he explained.
The researchers collected whole blood from a finger prick, dried it out on a protein saver card (filter paper), and cut out a 1.2-cm diameter dry blood spot which was then prepared for analysis.
Twenty blood samples with known amounts of HIV, HBV, and HCV were analyzed via the DBS method (60 in total) and the viruses were detected in all of the samples.
To validate the method, the researchers used plasma with a known viral load, and a series of dilutions were performed to determine the lower limit for positive detection of all three viruses.
“Untreated patients typically have above 1 million IU/mL of viral loads in their plasma, and we found that we can detect much lower levels,” said Mr. Nilsson-Møller. “Ideally, 40 mcL of blood is good, but less should be sufficient if the test is on untreated patients.”
Early testing and treatment reduces morbidity and mortality
Elimination of HBV, HCV, and HIV by 2030 is a global health strategy set by the World Health Organization, but to meet this goal, new approaches for diagnostic testing are required. The DBS test for HIV, HBV, and HCV promises to make a significant contribution toward this goal.
“One in two people currently living with HIV is diagnosed late in the course of their infection, and an even larger proportion of the estimated 6 million Europeans living with chronic hepatitis B or C are not aware that they are infected,” said Anastasia Pharris, PhD, from the European Center for Disease Prevention and Control Principal Expert Infectious Diseases.
“Increasing testing coverage and uptake, especially for those most at risk, is an essential element of any strategy to eliminate HBV, HCV, and HIV in the European Union and European Economic Area,” she pointed out.
Dr. Pharris also highlighted that, while HIV, and often HBV infection, require lifelong treatment, HCV infection is now curable within a few weeks. “To maximize the benefits of individual treatment for all three infections, it is critical to test and diagnose people as soon as possible – in itself a challenge given that these infections can typically be asymptomatic for years.
“Early diagnosis of HBV, HCV, or HIV is vital as it allows people to access treatment, which significantly reduces associated long-term morbidity and mortality.
“In many cases, those most at risk of one of these infections are also more vulnerable to infection with one or both of the other viruses, making the argument for integrated testing even stronger,” she said in an interview.
Mr. Nilsson-Møller and Dr. Pharris reported no relevant financial relationships. Aptima kits for validation were provided by Hologic.
A version of this article first appeared on Medscape.com.
A test that uses a single drop of dried blood to detect HIV, hepatitis B virus, and HCV has been validated and is now in use in some high-risk settings in Denmark, according to research presented at the annual European Congress of Clinical Microbiology & Infectious Diseases.
Molecular biologist Stephen Nilsson-Møller, MSc, and colleagues at the department of clinical microbiology, Copenhagen University Hospital, developed and validated the test, known as the Dried Blood Spot (DBS), for HIV, HBV, and HCV.
The “test that can detect low viral loads for all three viruses from a single drop of blood, and can be done using existing hospital equipment,” Mr. Nilsson-Møller said in an interview. “Importantly, it does not require venipuncture, but can be done from a drop of dried blood from the finger.”
He highlighted the utility of the new test in more challenging settings. “This method is particularly useful in high-risk settings such as homeless shelters, drug rehabilitation centers, and prisons, where needles might be misused, and it can be difficult to convince people to have the more invasive test.”
“Also, in some places – such as in low- and middle-income settings – there is a distinct risk of ruining blood samples before analysis due to limited refrigeration for transit and storage,” he added. “[Standard] blood samples need to be analyzed within 6 hours when kept at room temperature, while dried blood spots can last for 9 months at room temperature and can be mailed to a laboratory with the right equipment to analyze it.”
Tiny amounts of virus detected
Mr. Nilsson-Møller was tasked with developing a test for use by the university’s department of infectious diseases to screen people in high-risk settings in the capital region of Copenhagen. The work forms part of a PhD project by Jonas Demant at the University of Copenhagen, for which he is screening for HIV, HBV, and HCV in drug rehabilitation centers, prisons, and homeless shelters.
The study is the first to use the Hologic Panther system (a nucleic acid amplification test) combining all three viruses, Mr. Nilsson-Møller pointed out. “A tiny amount of virus can be detected because it is a very sensitive platform using transcription-mediated amplification.”
“If it detects low amounts of virus, it will create many copies very quickly, creating a signal that tells us that the sample is positive,” he explained.
The researchers collected whole blood from a finger prick, dried it out on a protein saver card (filter paper), and cut out a 1.2-cm diameter dry blood spot which was then prepared for analysis.
Twenty blood samples with known amounts of HIV, HBV, and HCV were analyzed via the DBS method (60 in total) and the viruses were detected in all of the samples.
To validate the method, the researchers used plasma with a known viral load, and a series of dilutions were performed to determine the lower limit for positive detection of all three viruses.
“Untreated patients typically have above 1 million IU/mL of viral loads in their plasma, and we found that we can detect much lower levels,” said Mr. Nilsson-Møller. “Ideally, 40 mcL of blood is good, but less should be sufficient if the test is on untreated patients.”
Early testing and treatment reduces morbidity and mortality
Elimination of HBV, HCV, and HIV by 2030 is a global health strategy set by the World Health Organization, but to meet this goal, new approaches for diagnostic testing are required. The DBS test for HIV, HBV, and HCV promises to make a significant contribution toward this goal.
“One in two people currently living with HIV is diagnosed late in the course of their infection, and an even larger proportion of the estimated 6 million Europeans living with chronic hepatitis B or C are not aware that they are infected,” said Anastasia Pharris, PhD, from the European Center for Disease Prevention and Control Principal Expert Infectious Diseases.
“Increasing testing coverage and uptake, especially for those most at risk, is an essential element of any strategy to eliminate HBV, HCV, and HIV in the European Union and European Economic Area,” she pointed out.
Dr. Pharris also highlighted that, while HIV, and often HBV infection, require lifelong treatment, HCV infection is now curable within a few weeks. “To maximize the benefits of individual treatment for all three infections, it is critical to test and diagnose people as soon as possible – in itself a challenge given that these infections can typically be asymptomatic for years.
“Early diagnosis of HBV, HCV, or HIV is vital as it allows people to access treatment, which significantly reduces associated long-term morbidity and mortality.
“In many cases, those most at risk of one of these infections are also more vulnerable to infection with one or both of the other viruses, making the argument for integrated testing even stronger,” she said in an interview.
Mr. Nilsson-Møller and Dr. Pharris reported no relevant financial relationships. Aptima kits for validation were provided by Hologic.
A version of this article first appeared on Medscape.com.
A test that uses a single drop of dried blood to detect HIV, hepatitis B virus, and HCV has been validated and is now in use in some high-risk settings in Denmark, according to research presented at the annual European Congress of Clinical Microbiology & Infectious Diseases.
Molecular biologist Stephen Nilsson-Møller, MSc, and colleagues at the department of clinical microbiology, Copenhagen University Hospital, developed and validated the test, known as the Dried Blood Spot (DBS), for HIV, HBV, and HCV.
The “test that can detect low viral loads for all three viruses from a single drop of blood, and can be done using existing hospital equipment,” Mr. Nilsson-Møller said in an interview. “Importantly, it does not require venipuncture, but can be done from a drop of dried blood from the finger.”
He highlighted the utility of the new test in more challenging settings. “This method is particularly useful in high-risk settings such as homeless shelters, drug rehabilitation centers, and prisons, where needles might be misused, and it can be difficult to convince people to have the more invasive test.”
“Also, in some places – such as in low- and middle-income settings – there is a distinct risk of ruining blood samples before analysis due to limited refrigeration for transit and storage,” he added. “[Standard] blood samples need to be analyzed within 6 hours when kept at room temperature, while dried blood spots can last for 9 months at room temperature and can be mailed to a laboratory with the right equipment to analyze it.”
Tiny amounts of virus detected
Mr. Nilsson-Møller was tasked with developing a test for use by the university’s department of infectious diseases to screen people in high-risk settings in the capital region of Copenhagen. The work forms part of a PhD project by Jonas Demant at the University of Copenhagen, for which he is screening for HIV, HBV, and HCV in drug rehabilitation centers, prisons, and homeless shelters.
The study is the first to use the Hologic Panther system (a nucleic acid amplification test) combining all three viruses, Mr. Nilsson-Møller pointed out. “A tiny amount of virus can be detected because it is a very sensitive platform using transcription-mediated amplification.”
“If it detects low amounts of virus, it will create many copies very quickly, creating a signal that tells us that the sample is positive,” he explained.
The researchers collected whole blood from a finger prick, dried it out on a protein saver card (filter paper), and cut out a 1.2-cm diameter dry blood spot which was then prepared for analysis.
Twenty blood samples with known amounts of HIV, HBV, and HCV were analyzed via the DBS method (60 in total) and the viruses were detected in all of the samples.
To validate the method, the researchers used plasma with a known viral load, and a series of dilutions were performed to determine the lower limit for positive detection of all three viruses.
“Untreated patients typically have above 1 million IU/mL of viral loads in their plasma, and we found that we can detect much lower levels,” said Mr. Nilsson-Møller. “Ideally, 40 mcL of blood is good, but less should be sufficient if the test is on untreated patients.”
Early testing and treatment reduces morbidity and mortality
Elimination of HBV, HCV, and HIV by 2030 is a global health strategy set by the World Health Organization, but to meet this goal, new approaches for diagnostic testing are required. The DBS test for HIV, HBV, and HCV promises to make a significant contribution toward this goal.
“One in two people currently living with HIV is diagnosed late in the course of their infection, and an even larger proportion of the estimated 6 million Europeans living with chronic hepatitis B or C are not aware that they are infected,” said Anastasia Pharris, PhD, from the European Center for Disease Prevention and Control Principal Expert Infectious Diseases.
“Increasing testing coverage and uptake, especially for those most at risk, is an essential element of any strategy to eliminate HBV, HCV, and HIV in the European Union and European Economic Area,” she pointed out.
Dr. Pharris also highlighted that, while HIV, and often HBV infection, require lifelong treatment, HCV infection is now curable within a few weeks. “To maximize the benefits of individual treatment for all three infections, it is critical to test and diagnose people as soon as possible – in itself a challenge given that these infections can typically be asymptomatic for years.
“Early diagnosis of HBV, HCV, or HIV is vital as it allows people to access treatment, which significantly reduces associated long-term morbidity and mortality.
“In many cases, those most at risk of one of these infections are also more vulnerable to infection with one or both of the other viruses, making the argument for integrated testing even stronger,” she said in an interview.
Mr. Nilsson-Møller and Dr. Pharris reported no relevant financial relationships. Aptima kits for validation were provided by Hologic.
A version of this article first appeared on Medscape.com.
FROM ECCMID 2023
Are delayed antibiotic prescriptions futile?
I recently posted a case about a smoker who became angry when I hesitated to prescribe antibiotics for his self-diagnosed bronchitis. He even threatened to retaliate by posting negative online reviews of my practice. In the end, I decided to use the strategy of a delayed prescription for antibiotics, instructing him to fill the prescription only if his symptoms worsened. I asked whether readers agreed with this approach. Thank you for the thoughtful comments regarding a case that certainly seemed familiar to many of you. I very much appreciate the chance to interact and share perspectives in a challenging clinical dilemma.
One theme that emerged through several comments was the perceived futility of the delayed prescriptions for antibiotics. To summarize, the collective logic stated that there is no point in delaying a prescription, because the patient will be very likely to fill that prescription right away despite counseling from the health care provider (HCP).
However, studies of delayed antibiotic prescriptions show that patients generally honor the advice to only fill the prescription if they are not improving clinically. In a study comparing immediate, delayed, or no antibiotic prescriptions among a cohort of children with uncomplicated respiratory infections, the overall rates of use of antibiotics in the three respective groups were 96%, 25.3%, and 12.0%. In another randomized trial exploring different strategies for delayed prescriptions among adults with upper respiratory infections, the rate of antibiotic use was 37% with delayed prescription strategies vs. 97% of patients prescribed antibiotics immediately. Neither of these prospective studies found a significant difference in clinical symptoms or complications in comparing the delayed and immediate antibiotic prescription groups.
Another common theme in the comments on this case focused on the challenge of online reviews of HCPs by patients. Multiple popular websites are devoted to patients’ unedited comments on HCPs and their practices, but there are still certain patterns to the comments. Some reviews describe the professionalism or empathy of the HCP, but others might focus more attention on the overall practice or office. These latter comments might emphasize issues such as timeliness of appointments, interactions with staff, or even parking and traffic. These are issues over which the HCP usually has little control.
HCPs are quite human, and therefore we might feel great about positive comments and dispirited or even angry with negative comments. So what is the best practice for HCPs in managing these online comments? A review by Dr Rebekah Bernard, which was published in the Sept. 25, 2018, issue of Medical Economics, offered some pragmatic advice:
Do not perseverate on one or two negative reviews. In fact, they might help! Dr. Bernard describes the psychological theory of the “pratfall effect,” in which people are more likely to prefer someone who is generally very good but not perfect to someone with nothing but exceptional reviews. HCPs with perfect reviews every time may be seen as intimidating or unapproachable.
Satisfied patients will frequently rally to support an HCP with an unfavorable review. This group may not be very motivated to complete online reviews until they see a comment which does at all match their own experience with the HCP.
Most importantly, HCPs can take an active role in minimizing the impact of negative online reviews while also enhancing their business model. Increasing your presence on the Internet and social media can help dilute negative reviews and push them down the list when someone performs a search on your name or practice. Creating a website for your practice is an effective means to be first on search engine lists, and HCPs should seek search-engine optimization features that promote this outcome. Adding social media contacts for yourself and/or your practice, as many as you can tolerate and maintain, allows HCPs to further control the narrative regarding their practice and central messaging to patients and the community.
In conclusion, delayed antibiotic prescriptions can reduce the use of unnecessary antibiotics for upper respiratory infections among children and adults, and they are not associated with worse clinical outcomes vs. immediate antibiotic prescriptions. They can also improve patient satisfaction for these visits, which can minimize the challenging issue of negative reviews of HCPs. HCPs should therefore consider delayed prescriptions as a strong option among patients without an indication for an antibiotic prescription.
A version of this article first appeared on Medscape.com.
I recently posted a case about a smoker who became angry when I hesitated to prescribe antibiotics for his self-diagnosed bronchitis. He even threatened to retaliate by posting negative online reviews of my practice. In the end, I decided to use the strategy of a delayed prescription for antibiotics, instructing him to fill the prescription only if his symptoms worsened. I asked whether readers agreed with this approach. Thank you for the thoughtful comments regarding a case that certainly seemed familiar to many of you. I very much appreciate the chance to interact and share perspectives in a challenging clinical dilemma.
One theme that emerged through several comments was the perceived futility of the delayed prescriptions for antibiotics. To summarize, the collective logic stated that there is no point in delaying a prescription, because the patient will be very likely to fill that prescription right away despite counseling from the health care provider (HCP).
However, studies of delayed antibiotic prescriptions show that patients generally honor the advice to only fill the prescription if they are not improving clinically. In a study comparing immediate, delayed, or no antibiotic prescriptions among a cohort of children with uncomplicated respiratory infections, the overall rates of use of antibiotics in the three respective groups were 96%, 25.3%, and 12.0%. In another randomized trial exploring different strategies for delayed prescriptions among adults with upper respiratory infections, the rate of antibiotic use was 37% with delayed prescription strategies vs. 97% of patients prescribed antibiotics immediately. Neither of these prospective studies found a significant difference in clinical symptoms or complications in comparing the delayed and immediate antibiotic prescription groups.
Another common theme in the comments on this case focused on the challenge of online reviews of HCPs by patients. Multiple popular websites are devoted to patients’ unedited comments on HCPs and their practices, but there are still certain patterns to the comments. Some reviews describe the professionalism or empathy of the HCP, but others might focus more attention on the overall practice or office. These latter comments might emphasize issues such as timeliness of appointments, interactions with staff, or even parking and traffic. These are issues over which the HCP usually has little control.
HCPs are quite human, and therefore we might feel great about positive comments and dispirited or even angry with negative comments. So what is the best practice for HCPs in managing these online comments? A review by Dr Rebekah Bernard, which was published in the Sept. 25, 2018, issue of Medical Economics, offered some pragmatic advice:
Do not perseverate on one or two negative reviews. In fact, they might help! Dr. Bernard describes the psychological theory of the “pratfall effect,” in which people are more likely to prefer someone who is generally very good but not perfect to someone with nothing but exceptional reviews. HCPs with perfect reviews every time may be seen as intimidating or unapproachable.
Satisfied patients will frequently rally to support an HCP with an unfavorable review. This group may not be very motivated to complete online reviews until they see a comment which does at all match their own experience with the HCP.
Most importantly, HCPs can take an active role in minimizing the impact of negative online reviews while also enhancing their business model. Increasing your presence on the Internet and social media can help dilute negative reviews and push them down the list when someone performs a search on your name or practice. Creating a website for your practice is an effective means to be first on search engine lists, and HCPs should seek search-engine optimization features that promote this outcome. Adding social media contacts for yourself and/or your practice, as many as you can tolerate and maintain, allows HCPs to further control the narrative regarding their practice and central messaging to patients and the community.
In conclusion, delayed antibiotic prescriptions can reduce the use of unnecessary antibiotics for upper respiratory infections among children and adults, and they are not associated with worse clinical outcomes vs. immediate antibiotic prescriptions. They can also improve patient satisfaction for these visits, which can minimize the challenging issue of negative reviews of HCPs. HCPs should therefore consider delayed prescriptions as a strong option among patients without an indication for an antibiotic prescription.
A version of this article first appeared on Medscape.com.
I recently posted a case about a smoker who became angry when I hesitated to prescribe antibiotics for his self-diagnosed bronchitis. He even threatened to retaliate by posting negative online reviews of my practice. In the end, I decided to use the strategy of a delayed prescription for antibiotics, instructing him to fill the prescription only if his symptoms worsened. I asked whether readers agreed with this approach. Thank you for the thoughtful comments regarding a case that certainly seemed familiar to many of you. I very much appreciate the chance to interact and share perspectives in a challenging clinical dilemma.
One theme that emerged through several comments was the perceived futility of the delayed prescriptions for antibiotics. To summarize, the collective logic stated that there is no point in delaying a prescription, because the patient will be very likely to fill that prescription right away despite counseling from the health care provider (HCP).
However, studies of delayed antibiotic prescriptions show that patients generally honor the advice to only fill the prescription if they are not improving clinically. In a study comparing immediate, delayed, or no antibiotic prescriptions among a cohort of children with uncomplicated respiratory infections, the overall rates of use of antibiotics in the three respective groups were 96%, 25.3%, and 12.0%. In another randomized trial exploring different strategies for delayed prescriptions among adults with upper respiratory infections, the rate of antibiotic use was 37% with delayed prescription strategies vs. 97% of patients prescribed antibiotics immediately. Neither of these prospective studies found a significant difference in clinical symptoms or complications in comparing the delayed and immediate antibiotic prescription groups.
Another common theme in the comments on this case focused on the challenge of online reviews of HCPs by patients. Multiple popular websites are devoted to patients’ unedited comments on HCPs and their practices, but there are still certain patterns to the comments. Some reviews describe the professionalism or empathy of the HCP, but others might focus more attention on the overall practice or office. These latter comments might emphasize issues such as timeliness of appointments, interactions with staff, or even parking and traffic. These are issues over which the HCP usually has little control.
HCPs are quite human, and therefore we might feel great about positive comments and dispirited or even angry with negative comments. So what is the best practice for HCPs in managing these online comments? A review by Dr Rebekah Bernard, which was published in the Sept. 25, 2018, issue of Medical Economics, offered some pragmatic advice:
Do not perseverate on one or two negative reviews. In fact, they might help! Dr. Bernard describes the psychological theory of the “pratfall effect,” in which people are more likely to prefer someone who is generally very good but not perfect to someone with nothing but exceptional reviews. HCPs with perfect reviews every time may be seen as intimidating or unapproachable.
Satisfied patients will frequently rally to support an HCP with an unfavorable review. This group may not be very motivated to complete online reviews until they see a comment which does at all match their own experience with the HCP.
Most importantly, HCPs can take an active role in minimizing the impact of negative online reviews while also enhancing their business model. Increasing your presence on the Internet and social media can help dilute negative reviews and push them down the list when someone performs a search on your name or practice. Creating a website for your practice is an effective means to be first on search engine lists, and HCPs should seek search-engine optimization features that promote this outcome. Adding social media contacts for yourself and/or your practice, as many as you can tolerate and maintain, allows HCPs to further control the narrative regarding their practice and central messaging to patients and the community.
In conclusion, delayed antibiotic prescriptions can reduce the use of unnecessary antibiotics for upper respiratory infections among children and adults, and they are not associated with worse clinical outcomes vs. immediate antibiotic prescriptions. They can also improve patient satisfaction for these visits, which can minimize the challenging issue of negative reviews of HCPs. HCPs should therefore consider delayed prescriptions as a strong option among patients without an indication for an antibiotic prescription.
A version of this article first appeared on Medscape.com.
CDC backs FDA’s call for second COVID booster for those at high risk
This backs the Food and Drug Administration’s authorization April 18 of the additional shot.
“Following FDA regulatory action, CDC has taken steps to simplify COVID-19 vaccine recommendations and allow more flexibility for people at higher risk who want the option of added protection from additional COVID-19 vaccine doses,” the CDC said in a statement.
The agency is following the recommendations made by its Advisory Committee on Immunization Practices (ACIP). While there was no vote, the group reaffirmed its commitment to boosters overall, proposing that all Americans over age 6 who have not had a bivalent mRNA COVID-19 booster vaccine go ahead and get one.
But most others who’ve already had the bivalent shot – which targets the original COVID strain and the two Omicron variants BA.4 and BA.5 – should wait until the fall to get whatever updated vaccine is available.
The panel did carve out exceptions for people over age 65 and those who are immunocompromised because they are at higher risk for severe COVID-19 complications, Evelyn Twentyman, MD, MPH, the lead official in the CDC’s COVID-19 Vaccine Policy Unit, said during the meeting.
People over 65 can now choose to get a second bivalent mRNA booster shot as long as it has been at least 4 months since the last one, she said, and people who are immunocompromised also should have the flexibility to receive one or more additional bivalent boosters at least 2 months after an initial dose.
Regardless of whether someone is unvaccinated, and regardless of how many single-strain COVID vaccines an individual has previously received, they should get a mRNA bivalent shot, Dr. Twentyman said.
If an individual has already received a bivalent mRNA booster – made by either Pfizer/BioNTech or Moderna – “your vaccination is complete,” she said. “No doses indicated at this time, come back and see us in autumn of 2023.”
The CDC is trying to encourage more people to get the updated COVID shot, as just 17% of Americans of any age have received a bivalent booster and only 43% of those age 65 and over.
The CDC followed the FDA’s lead in its statement, phasing out the original single-strain COVID vaccine, saying it will no longer be recommended for use in the United States.
‘Unnecessary drama’ over children’s recs
The CDC panel mostly followed the FDA’s guidance on who should get a booster, but many ACIP members expressed consternation and confusion about what was being recommended for children.
For children aged 6 months to 4 years, the CDC will offer tables to help physicians determine how many bivalent doses to give, depending on the child’s vaccination history.
All children those ages should get at least two vaccine doses, one of which is bivalent, Dr. Twentyman said. For children in that age group who have already received a monovalent series and a bivalent dose, “their vaccination is complete,” she said.
For 5-year-olds, the recommendations will be similar if they received a Pfizer monovalent series, but the shot regimen will have to be customized if they had previously received a Moderna shot, because of differences in the dosages.
ACIP member Sarah S. Long, MD, professor of pediatrics, Drexel University, Philadelphia, said that it was unclear why a set age couldn’t be established for COVID-19 vaccination as it had been for other immunizations.
“We picked 60 months for most immunizations in children,” Dr. Long said. “Immunologically there is not a difference between a 4-, a 5- and a 6-year-old.
“There isn’t a reason to have all this unnecessary drama around those ages,” she said, adding that having the different ages would make it harder for pediatricians to appropriately stock vaccines.
Dr. Twentyman said that the CDC would be providing more detailed guidance on its COVID-19 website soon and would be holding a call with health care professionals to discuss the updated recommendations on May 11.
New vaccine by fall
CDC and ACIP members both said they hoped to have an even simpler vaccine schedule by the fall, when it is anticipated that the FDA may have authorized a new, updated bivalent vaccine that targets other COVID variants.
“We all recognize this is a work in progress,” said ACIP Chair Grace M. Lee, MD, MPH, acknowledging that there is continued confusion over COVID-19 vaccination.
“The goal really is to try to simplify things over time to be able to help communicate with our provider community, and our patients and families what vaccine is right for them, when do they need it, and how often should they get it,” said Dr. Lee, professor of pediatrics, Stanford (Calif.) University.
A version of this article originally appeared on Medscape.com .
This backs the Food and Drug Administration’s authorization April 18 of the additional shot.
“Following FDA regulatory action, CDC has taken steps to simplify COVID-19 vaccine recommendations and allow more flexibility for people at higher risk who want the option of added protection from additional COVID-19 vaccine doses,” the CDC said in a statement.
The agency is following the recommendations made by its Advisory Committee on Immunization Practices (ACIP). While there was no vote, the group reaffirmed its commitment to boosters overall, proposing that all Americans over age 6 who have not had a bivalent mRNA COVID-19 booster vaccine go ahead and get one.
But most others who’ve already had the bivalent shot – which targets the original COVID strain and the two Omicron variants BA.4 and BA.5 – should wait until the fall to get whatever updated vaccine is available.
The panel did carve out exceptions for people over age 65 and those who are immunocompromised because they are at higher risk for severe COVID-19 complications, Evelyn Twentyman, MD, MPH, the lead official in the CDC’s COVID-19 Vaccine Policy Unit, said during the meeting.
People over 65 can now choose to get a second bivalent mRNA booster shot as long as it has been at least 4 months since the last one, she said, and people who are immunocompromised also should have the flexibility to receive one or more additional bivalent boosters at least 2 months after an initial dose.
Regardless of whether someone is unvaccinated, and regardless of how many single-strain COVID vaccines an individual has previously received, they should get a mRNA bivalent shot, Dr. Twentyman said.
If an individual has already received a bivalent mRNA booster – made by either Pfizer/BioNTech or Moderna – “your vaccination is complete,” she said. “No doses indicated at this time, come back and see us in autumn of 2023.”
The CDC is trying to encourage more people to get the updated COVID shot, as just 17% of Americans of any age have received a bivalent booster and only 43% of those age 65 and over.
The CDC followed the FDA’s lead in its statement, phasing out the original single-strain COVID vaccine, saying it will no longer be recommended for use in the United States.
‘Unnecessary drama’ over children’s recs
The CDC panel mostly followed the FDA’s guidance on who should get a booster, but many ACIP members expressed consternation and confusion about what was being recommended for children.
For children aged 6 months to 4 years, the CDC will offer tables to help physicians determine how many bivalent doses to give, depending on the child’s vaccination history.
All children those ages should get at least two vaccine doses, one of which is bivalent, Dr. Twentyman said. For children in that age group who have already received a monovalent series and a bivalent dose, “their vaccination is complete,” she said.
For 5-year-olds, the recommendations will be similar if they received a Pfizer monovalent series, but the shot regimen will have to be customized if they had previously received a Moderna shot, because of differences in the dosages.
ACIP member Sarah S. Long, MD, professor of pediatrics, Drexel University, Philadelphia, said that it was unclear why a set age couldn’t be established for COVID-19 vaccination as it had been for other immunizations.
“We picked 60 months for most immunizations in children,” Dr. Long said. “Immunologically there is not a difference between a 4-, a 5- and a 6-year-old.
“There isn’t a reason to have all this unnecessary drama around those ages,” she said, adding that having the different ages would make it harder for pediatricians to appropriately stock vaccines.
Dr. Twentyman said that the CDC would be providing more detailed guidance on its COVID-19 website soon and would be holding a call with health care professionals to discuss the updated recommendations on May 11.
New vaccine by fall
CDC and ACIP members both said they hoped to have an even simpler vaccine schedule by the fall, when it is anticipated that the FDA may have authorized a new, updated bivalent vaccine that targets other COVID variants.
“We all recognize this is a work in progress,” said ACIP Chair Grace M. Lee, MD, MPH, acknowledging that there is continued confusion over COVID-19 vaccination.
“The goal really is to try to simplify things over time to be able to help communicate with our provider community, and our patients and families what vaccine is right for them, when do they need it, and how often should they get it,” said Dr. Lee, professor of pediatrics, Stanford (Calif.) University.
A version of this article originally appeared on Medscape.com .
This backs the Food and Drug Administration’s authorization April 18 of the additional shot.
“Following FDA regulatory action, CDC has taken steps to simplify COVID-19 vaccine recommendations and allow more flexibility for people at higher risk who want the option of added protection from additional COVID-19 vaccine doses,” the CDC said in a statement.
The agency is following the recommendations made by its Advisory Committee on Immunization Practices (ACIP). While there was no vote, the group reaffirmed its commitment to boosters overall, proposing that all Americans over age 6 who have not had a bivalent mRNA COVID-19 booster vaccine go ahead and get one.
But most others who’ve already had the bivalent shot – which targets the original COVID strain and the two Omicron variants BA.4 and BA.5 – should wait until the fall to get whatever updated vaccine is available.
The panel did carve out exceptions for people over age 65 and those who are immunocompromised because they are at higher risk for severe COVID-19 complications, Evelyn Twentyman, MD, MPH, the lead official in the CDC’s COVID-19 Vaccine Policy Unit, said during the meeting.
People over 65 can now choose to get a second bivalent mRNA booster shot as long as it has been at least 4 months since the last one, she said, and people who are immunocompromised also should have the flexibility to receive one or more additional bivalent boosters at least 2 months after an initial dose.
Regardless of whether someone is unvaccinated, and regardless of how many single-strain COVID vaccines an individual has previously received, they should get a mRNA bivalent shot, Dr. Twentyman said.
If an individual has already received a bivalent mRNA booster – made by either Pfizer/BioNTech or Moderna – “your vaccination is complete,” she said. “No doses indicated at this time, come back and see us in autumn of 2023.”
The CDC is trying to encourage more people to get the updated COVID shot, as just 17% of Americans of any age have received a bivalent booster and only 43% of those age 65 and over.
The CDC followed the FDA’s lead in its statement, phasing out the original single-strain COVID vaccine, saying it will no longer be recommended for use in the United States.
‘Unnecessary drama’ over children’s recs
The CDC panel mostly followed the FDA’s guidance on who should get a booster, but many ACIP members expressed consternation and confusion about what was being recommended for children.
For children aged 6 months to 4 years, the CDC will offer tables to help physicians determine how many bivalent doses to give, depending on the child’s vaccination history.
All children those ages should get at least two vaccine doses, one of which is bivalent, Dr. Twentyman said. For children in that age group who have already received a monovalent series and a bivalent dose, “their vaccination is complete,” she said.
For 5-year-olds, the recommendations will be similar if they received a Pfizer monovalent series, but the shot regimen will have to be customized if they had previously received a Moderna shot, because of differences in the dosages.
ACIP member Sarah S. Long, MD, professor of pediatrics, Drexel University, Philadelphia, said that it was unclear why a set age couldn’t be established for COVID-19 vaccination as it had been for other immunizations.
“We picked 60 months for most immunizations in children,” Dr. Long said. “Immunologically there is not a difference between a 4-, a 5- and a 6-year-old.
“There isn’t a reason to have all this unnecessary drama around those ages,” she said, adding that having the different ages would make it harder for pediatricians to appropriately stock vaccines.
Dr. Twentyman said that the CDC would be providing more detailed guidance on its COVID-19 website soon and would be holding a call with health care professionals to discuss the updated recommendations on May 11.
New vaccine by fall
CDC and ACIP members both said they hoped to have an even simpler vaccine schedule by the fall, when it is anticipated that the FDA may have authorized a new, updated bivalent vaccine that targets other COVID variants.
“We all recognize this is a work in progress,” said ACIP Chair Grace M. Lee, MD, MPH, acknowledging that there is continued confusion over COVID-19 vaccination.
“The goal really is to try to simplify things over time to be able to help communicate with our provider community, and our patients and families what vaccine is right for them, when do they need it, and how often should they get it,” said Dr. Lee, professor of pediatrics, Stanford (Calif.) University.
A version of this article originally appeared on Medscape.com .
Rabies: How to respond to parents’ questions
When most families hear the word rabies, they envision a dog foaming at the mouth and think about receiving multiple painful, often intra-abdominal injections. However, the epidemiology of rabies has changed in the United States. Postexposure prophylaxis (PEP) may not always be indicated and for certain persons preexposure prophylaxis (PrEP) is available and recommended.
Rabies is a Lyssavirus that is transmitted through saliva most often from the bite or scratch of an infected animal. Sometimes it’s via direct contact with mucous membranes. Although rare, cases have been described in which an undiagnosed donor passed the virus via transplant to recipients and four cases of aerosolized transmission were documented in two spelunkers and two laboratory technicians working with the virus. Worldwide it’s estimated that rabies causes 59,000 deaths annually.
Most cases (98%) are secondary to canine rabies. Prior to 1960, dogs were the major reservoir in the United States; however, after introduction of leash laws and animal vaccination in 1947, there was a drastic decline in cases caused by the canine rabies virus variant (CRVV). By 2004, CRVV was eliminated in the United States.
However, the proportion of strains associated with wildlife including raccoons, skunks, foxes, bats, coyotes, and mongoose now account for most of the cases in humans. Wildlife rabies is found in all states except Hawaii. Between 1960 and 2018, 89 cases were acquired in the United States and 62 (70%) were from bat exposure. Dog bites acquired during international travel were the cause of 36 cases.
Once signs and symptoms of disease develop there is no treatment. Regardless of the species variant, rabies virus infection is fatal in over 99% of cases. However, disease can be prevented with prompt initiation of PEP, which includes administration of rabies immune globulin (RIG) and rabies vaccine. Let’s look at a few different scenarios.
1. A delivery person is bitten by your neighbor’s dog while making a delivery. He was told to get rabies vaccine. What should we advise?
Canine rabies has been eliminated in the United States. However, unvaccinated canines can acquire rabies from wildlife. In this situation, you can determine the immunization status of the dog. Contact your local/state health department to assist with enforcement and management. Bites by cats and ferrets should be managed similarly.
Healthy dog:
1. Observe for 10 days.
2. PEP is not indicated unless the animal develops signs/symptoms of rabies. Then euthanize and begin PEP.
Dog appears rabid or suspected to be rabid:
1. Begin PEP.
2. Animal should be euthanized. If immunofluorescent test is negative discontinue PEP.
Dog unavailable:
Contact local/state health department. They are more familiar with rabies surveillance data.
2. Patient relocating to Malaysia for 3-4 years. Rabies PrEP was recommended but the family wants your opinion before receiving the vaccine. What would you advise?
Canine rabies is felt to be the primary cause of rabies outside of the United States. Canines are not routinely vaccinated in many foreign destinations, and the availability of RIG and rabies vaccine is not guaranteed in developing countries. As noted above, dog bites during international travel accounted for 28% of U.S. cases between 1960 and 2018.
In May 2022 recommendations for a modified two-dose PrEP schedule was published that identifies five risk groups and includes specific timing for checking rabies titers. The third rabies dose can now be administered up until year 3 (Morb Mortal Wkly Rep. 2022 May 6;71[18]:619-27). For individuals relocating to countries where CRVV is present, I prefer the traditional three-dose PrEP schedule administered between 21 and 28 days. However, we now have options. If exposure occurs any time after completion of a three-dose PrEP series or within 3 years after completion of a two-dose PrEP series, RIG would not be required. All patients would receive two doses of rabies vaccine (days 0, 3). If exposure occurs after 3 years in a person who received two doses of PrEP who did not have documentation of a protective rabies titer (> 5 IU/mL), treatment will include RIG plus four doses of vaccine (days 0, 3, 7, 14).
For this relocating patient, supporting PrEP would be strongly recommended.
3. A mother tells you she sees bats flying around her home at night and a few have even gotten into the home. This morning she saw one in her child’s room. He was still sleeping. Is there anything she needs to do?
Bats have become the predominant source of rabies in the United States. In addition to the cases noted above, three fatal cases occurred between Sept. 28 and Nov. 10, 2021, after bat exposures in August 2021 (MMWR Morb Mortal Wkly Rep. 2022 Jan 7;71:31-2). All had recognized contact with a bat 3-7 weeks prior to onset of symptoms and died 2-3 weeks after symptom onset. One declined PEP and the other two did not realize the risk for rabies from their exposure or did not notice a scratch or bite. Bites from bats may be small and unnoticed. Exposure to a bat in a closed room while sleeping is considered an exposure. Hawaii is the only state not reporting rabid bats.
PEP is recommended for her child. She should identify potential areas bats may enter the home and seal them in addition to removal of any bat roosts.
4. A parent realizes a house guest has been feeding raccoons in the backyard. What’s your response?
While bat rabies is the predominant variant associated with disease in the United States, as illustrated in Figure 1, other species of wildlife including raccoons are a major source of rabies. The geographic spread of the raccoon variant of rabies has been limited by oral vaccination via bait. In the situation noted here, the raccoons have returned because food was being offered thus increasing the families chance of a potential rabies exposure. Wildlife including skunks, raccoons, coyotes, foxes, and mongooses are always considered rabid until proven negative by laboratory testing.
You recommend to stop feeding wildlife and never to approach them. Have them contact the local rabies control unit and/or state wildlife services to assist with removal of the raccoons. Depending on the locale, pest control may be required at the owners expense. Inform the family to seek PEP if anyone is bitten or scratched by the raccoons.
As per the Centers for Disease Control and Prevention, about 55,000 residents receive PEP annually with health-associated expenditures including diagnostics, prevention, and control estimated between $245 and $510 million annually. Rabies is one of the most fatal diseases that can be prevented by avoiding contact with wild animals, maintenance of high immunization rates in pets, and keeping people informed of potential sources including bats. One can’t determine if an animal has rabies by looking at it. Rabies remains an urgent disease that we have to remember to address with our patients and their families. For additional information go to www.CDC.gov/rabies.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She has no relevant financial disclosures.
When most families hear the word rabies, they envision a dog foaming at the mouth and think about receiving multiple painful, often intra-abdominal injections. However, the epidemiology of rabies has changed in the United States. Postexposure prophylaxis (PEP) may not always be indicated and for certain persons preexposure prophylaxis (PrEP) is available and recommended.
Rabies is a Lyssavirus that is transmitted through saliva most often from the bite or scratch of an infected animal. Sometimes it’s via direct contact with mucous membranes. Although rare, cases have been described in which an undiagnosed donor passed the virus via transplant to recipients and four cases of aerosolized transmission were documented in two spelunkers and two laboratory technicians working with the virus. Worldwide it’s estimated that rabies causes 59,000 deaths annually.

Most cases (98%) are secondary to canine rabies. Prior to 1960, dogs were the major reservoir in the United States; however, after introduction of leash laws and animal vaccination in 1947, there was a drastic decline in cases caused by the canine rabies virus variant (CRVV). By 2004, CRVV was eliminated in the United States.
However, the proportion of strains associated with wildlife including raccoons, skunks, foxes, bats, coyotes, and mongoose now account for most of the cases in humans. Wildlife rabies is found in all states except Hawaii. Between 1960 and 2018, 89 cases were acquired in the United States and 62 (70%) were from bat exposure. Dog bites acquired during international travel were the cause of 36 cases.
Once signs and symptoms of disease develop there is no treatment. Regardless of the species variant, rabies virus infection is fatal in over 99% of cases. However, disease can be prevented with prompt initiation of PEP, which includes administration of rabies immune globulin (RIG) and rabies vaccine. Let’s look at a few different scenarios.
1. A delivery person is bitten by your neighbor’s dog while making a delivery. He was told to get rabies vaccine. What should we advise?
Canine rabies has been eliminated in the United States. However, unvaccinated canines can acquire rabies from wildlife. In this situation, you can determine the immunization status of the dog. Contact your local/state health department to assist with enforcement and management. Bites by cats and ferrets should be managed similarly.
Healthy dog:
1. Observe for 10 days.
2. PEP is not indicated unless the animal develops signs/symptoms of rabies. Then euthanize and begin PEP.
Dog appears rabid or suspected to be rabid:
1. Begin PEP.
2. Animal should be euthanized. If immunofluorescent test is negative discontinue PEP.
Dog unavailable:
Contact local/state health department. They are more familiar with rabies surveillance data.
2. Patient relocating to Malaysia for 3-4 years. Rabies PrEP was recommended but the family wants your opinion before receiving the vaccine. What would you advise?
Canine rabies is felt to be the primary cause of rabies outside of the United States. Canines are not routinely vaccinated in many foreign destinations, and the availability of RIG and rabies vaccine is not guaranteed in developing countries. As noted above, dog bites during international travel accounted for 28% of U.S. cases between 1960 and 2018.
In May 2022 recommendations for a modified two-dose PrEP schedule was published that identifies five risk groups and includes specific timing for checking rabies titers. The third rabies dose can now be administered up until year 3 (Morb Mortal Wkly Rep. 2022 May 6;71[18]:619-27). For individuals relocating to countries where CRVV is present, I prefer the traditional three-dose PrEP schedule administered between 21 and 28 days. However, we now have options. If exposure occurs any time after completion of a three-dose PrEP series or within 3 years after completion of a two-dose PrEP series, RIG would not be required. All patients would receive two doses of rabies vaccine (days 0, 3). If exposure occurs after 3 years in a person who received two doses of PrEP who did not have documentation of a protective rabies titer (> 5 IU/mL), treatment will include RIG plus four doses of vaccine (days 0, 3, 7, 14).
For this relocating patient, supporting PrEP would be strongly recommended.
3. A mother tells you she sees bats flying around her home at night and a few have even gotten into the home. This morning she saw one in her child’s room. He was still sleeping. Is there anything she needs to do?
Bats have become the predominant source of rabies in the United States. In addition to the cases noted above, three fatal cases occurred between Sept. 28 and Nov. 10, 2021, after bat exposures in August 2021 (MMWR Morb Mortal Wkly Rep. 2022 Jan 7;71:31-2). All had recognized contact with a bat 3-7 weeks prior to onset of symptoms and died 2-3 weeks after symptom onset. One declined PEP and the other two did not realize the risk for rabies from their exposure or did not notice a scratch or bite. Bites from bats may be small and unnoticed. Exposure to a bat in a closed room while sleeping is considered an exposure. Hawaii is the only state not reporting rabid bats.
PEP is recommended for her child. She should identify potential areas bats may enter the home and seal them in addition to removal of any bat roosts.
4. A parent realizes a house guest has been feeding raccoons in the backyard. What’s your response?
While bat rabies is the predominant variant associated with disease in the United States, as illustrated in Figure 1, other species of wildlife including raccoons are a major source of rabies. The geographic spread of the raccoon variant of rabies has been limited by oral vaccination via bait. In the situation noted here, the raccoons have returned because food was being offered thus increasing the families chance of a potential rabies exposure. Wildlife including skunks, raccoons, coyotes, foxes, and mongooses are always considered rabid until proven negative by laboratory testing.

You recommend to stop feeding wildlife and never to approach them. Have them contact the local rabies control unit and/or state wildlife services to assist with removal of the raccoons. Depending on the locale, pest control may be required at the owners expense. Inform the family to seek PEP if anyone is bitten or scratched by the raccoons.
As per the Centers for Disease Control and Prevention, about 55,000 residents receive PEP annually with health-associated expenditures including diagnostics, prevention, and control estimated between $245 and $510 million annually. Rabies is one of the most fatal diseases that can be prevented by avoiding contact with wild animals, maintenance of high immunization rates in pets, and keeping people informed of potential sources including bats. One can’t determine if an animal has rabies by looking at it. Rabies remains an urgent disease that we have to remember to address with our patients and their families. For additional information go to www.CDC.gov/rabies.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She has no relevant financial disclosures.
When most families hear the word rabies, they envision a dog foaming at the mouth and think about receiving multiple painful, often intra-abdominal injections. However, the epidemiology of rabies has changed in the United States. Postexposure prophylaxis (PEP) may not always be indicated and for certain persons preexposure prophylaxis (PrEP) is available and recommended.
Rabies is a Lyssavirus that is transmitted through saliva most often from the bite or scratch of an infected animal. Sometimes it’s via direct contact with mucous membranes. Although rare, cases have been described in which an undiagnosed donor passed the virus via transplant to recipients and four cases of aerosolized transmission were documented in two spelunkers and two laboratory technicians working with the virus. Worldwide it’s estimated that rabies causes 59,000 deaths annually.

Most cases (98%) are secondary to canine rabies. Prior to 1960, dogs were the major reservoir in the United States; however, after introduction of leash laws and animal vaccination in 1947, there was a drastic decline in cases caused by the canine rabies virus variant (CRVV). By 2004, CRVV was eliminated in the United States.
However, the proportion of strains associated with wildlife including raccoons, skunks, foxes, bats, coyotes, and mongoose now account for most of the cases in humans. Wildlife rabies is found in all states except Hawaii. Between 1960 and 2018, 89 cases were acquired in the United States and 62 (70%) were from bat exposure. Dog bites acquired during international travel were the cause of 36 cases.
Once signs and symptoms of disease develop there is no treatment. Regardless of the species variant, rabies virus infection is fatal in over 99% of cases. However, disease can be prevented with prompt initiation of PEP, which includes administration of rabies immune globulin (RIG) and rabies vaccine. Let’s look at a few different scenarios.
1. A delivery person is bitten by your neighbor’s dog while making a delivery. He was told to get rabies vaccine. What should we advise?
Canine rabies has been eliminated in the United States. However, unvaccinated canines can acquire rabies from wildlife. In this situation, you can determine the immunization status of the dog. Contact your local/state health department to assist with enforcement and management. Bites by cats and ferrets should be managed similarly.
Healthy dog:
1. Observe for 10 days.
2. PEP is not indicated unless the animal develops signs/symptoms of rabies. Then euthanize and begin PEP.
Dog appears rabid or suspected to be rabid:
1. Begin PEP.
2. Animal should be euthanized. If immunofluorescent test is negative discontinue PEP.
Dog unavailable:
Contact local/state health department. They are more familiar with rabies surveillance data.
2. Patient relocating to Malaysia for 3-4 years. Rabies PrEP was recommended but the family wants your opinion before receiving the vaccine. What would you advise?
Canine rabies is felt to be the primary cause of rabies outside of the United States. Canines are not routinely vaccinated in many foreign destinations, and the availability of RIG and rabies vaccine is not guaranteed in developing countries. As noted above, dog bites during international travel accounted for 28% of U.S. cases between 1960 and 2018.
In May 2022 recommendations for a modified two-dose PrEP schedule was published that identifies five risk groups and includes specific timing for checking rabies titers. The third rabies dose can now be administered up until year 3 (Morb Mortal Wkly Rep. 2022 May 6;71[18]:619-27). For individuals relocating to countries where CRVV is present, I prefer the traditional three-dose PrEP schedule administered between 21 and 28 days. However, we now have options. If exposure occurs any time after completion of a three-dose PrEP series or within 3 years after completion of a two-dose PrEP series, RIG would not be required. All patients would receive two doses of rabies vaccine (days 0, 3). If exposure occurs after 3 years in a person who received two doses of PrEP who did not have documentation of a protective rabies titer (> 5 IU/mL), treatment will include RIG plus four doses of vaccine (days 0, 3, 7, 14).
For this relocating patient, supporting PrEP would be strongly recommended.
3. A mother tells you she sees bats flying around her home at night and a few have even gotten into the home. This morning she saw one in her child’s room. He was still sleeping. Is there anything she needs to do?
Bats have become the predominant source of rabies in the United States. In addition to the cases noted above, three fatal cases occurred between Sept. 28 and Nov. 10, 2021, after bat exposures in August 2021 (MMWR Morb Mortal Wkly Rep. 2022 Jan 7;71:31-2). All had recognized contact with a bat 3-7 weeks prior to onset of symptoms and died 2-3 weeks after symptom onset. One declined PEP and the other two did not realize the risk for rabies from their exposure or did not notice a scratch or bite. Bites from bats may be small and unnoticed. Exposure to a bat in a closed room while sleeping is considered an exposure. Hawaii is the only state not reporting rabid bats.
PEP is recommended for her child. She should identify potential areas bats may enter the home and seal them in addition to removal of any bat roosts.
4. A parent realizes a house guest has been feeding raccoons in the backyard. What’s your response?
While bat rabies is the predominant variant associated with disease in the United States, as illustrated in Figure 1, other species of wildlife including raccoons are a major source of rabies. The geographic spread of the raccoon variant of rabies has been limited by oral vaccination via bait. In the situation noted here, the raccoons have returned because food was being offered thus increasing the families chance of a potential rabies exposure. Wildlife including skunks, raccoons, coyotes, foxes, and mongooses are always considered rabid until proven negative by laboratory testing.

You recommend to stop feeding wildlife and never to approach them. Have them contact the local rabies control unit and/or state wildlife services to assist with removal of the raccoons. Depending on the locale, pest control may be required at the owners expense. Inform the family to seek PEP if anyone is bitten or scratched by the raccoons.
As per the Centers for Disease Control and Prevention, about 55,000 residents receive PEP annually with health-associated expenditures including diagnostics, prevention, and control estimated between $245 and $510 million annually. Rabies is one of the most fatal diseases that can be prevented by avoiding contact with wild animals, maintenance of high immunization rates in pets, and keeping people informed of potential sources including bats. One can’t determine if an animal has rabies by looking at it. Rabies remains an urgent disease that we have to remember to address with our patients and their families. For additional information go to www.CDC.gov/rabies.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She has no relevant financial disclosures.
Perinatal HIV nearly eradicated in U.S.
, with less than 1 baby for every 100,000 live births having the virus, a new study released by researchers at the Centers for Disease Control and Prevention finds.
The report marks significant progress on the U.S. government’s goal to eradicate perinatal HIV, an immune-weakening and potentially deadly virus that is passed from mother to baby during pregnancy. Just 32 children in the country were diagnosed in 2019, compared with twice as many in 2010, according to the CDC.
Mothers who are HIV positive can prevent transmission of the infection by receiving antiretroviral therapy, according to Monica Gandhi, MD, MPH, a professor of medicine at University of California, San Francisco’s division of HIV, infectious disease and global medicine.
Dr. Gandhi said she could recall only one case of perinatal HIV in the San Francisco area over the last decade.
“This country has been really aggressive about counseling women who are pregnant and getting mothers in care,” Dr. Gandhi said.
The treatment method was discovered more than 30 years ago. Prior to the therapy and ensuing awareness campaigns to prevent transmission, mothers with HIV would typically pass the virus to their child in utero, during delivery, or while breastfeeding.
“There should be zero children born with HIV, given that we’ve had these drugs for so long,” Dr. Ghandi said.
Disparities persist
But challenges remain in some communities, where babies born to Black mothers are disproportionately affected by the disease, the new study found. “Racial and ethnic differences in perinatal HIV diagnoses persisted through the 10-year period,” the report’s authors concluded. “The highest rates of perinatal HIV diagnoses were seen among infants born to Black women.”
Although rates of perinatal HIV declined for babies born to Black mothers over the decade-long study, the diagnosis rate was above the goal of elimination at 3.1 for every 100,000 live births, according to the data.
Meanwhile, transmission rates hovered around 1%-2% for Latinx and Hispanic women and mothers who identified as “other races,” including Native American.
Despite the availability of medication, expectant mothers may face several hurdles to getting the daily treatment they need to prevent transmission to their fetus, according to Jennifer Jao, MD, MPH, a physician of infectious diseases at Lurie Children’s Hospital of Chicago.
They might have trouble securing health insurance or finding transportation to doctor’s appointments, or face other problems like lacking secure housing or food – all factors that prevent them from prioritizing the care.
“All of those things play into the mix,” Dr. Jao said. “We see over and over again that closing the gap means you’ve got to reach the women who are pregnant and who don’t have resources.”
Progress in ‘danger’
Experts said they’re not sure what the impact of the COVID-19 pandemic, accompanied by a recent uptick in sexually transmitted diseases, will be on rates of perinatal HIV. Some women were unable to access prenatal health care during the pandemic because they couldn’t access public transportation or childcare, the U.S. Government Accountability Office said in 2022.
Globally, a decline in rates of HIV and AIDS rates has slowed, prompting the World Health Organization to warn last year that progress on the disease is in danger. Researchers only included HIV rates in the United States through 2019, so the data are outdated, Dr. Gandhi noted.
“All of this put together means we don’t know where we are with perinatal transmission over the last 3 years,” she said.
In an accompanying editorial, coauthors Nahida Chakhtoura, MD, MsGH, and Bill Kapogiannis, MD, both with the National Institutes of Health, urge health care professionals to take an active role in eliminating these racial and ethnic disparities in an effort to – as the title of their editorial proclaims – achieve a “road to zero perinatal HIV transmission” in the United States.
“The more proactive we are in identifying and promptly addressing systematic deficiencies that exacerbate health inequities in cutting-edge research innovations and optimal clinical service provision,” they write, “the less reactive we will need to be when new transmissible infections appear at our doorstep.”
A version of this article first appeared on Medscape.com.
, with less than 1 baby for every 100,000 live births having the virus, a new study released by researchers at the Centers for Disease Control and Prevention finds.
The report marks significant progress on the U.S. government’s goal to eradicate perinatal HIV, an immune-weakening and potentially deadly virus that is passed from mother to baby during pregnancy. Just 32 children in the country were diagnosed in 2019, compared with twice as many in 2010, according to the CDC.
Mothers who are HIV positive can prevent transmission of the infection by receiving antiretroviral therapy, according to Monica Gandhi, MD, MPH, a professor of medicine at University of California, San Francisco’s division of HIV, infectious disease and global medicine.
Dr. Gandhi said she could recall only one case of perinatal HIV in the San Francisco area over the last decade.
“This country has been really aggressive about counseling women who are pregnant and getting mothers in care,” Dr. Gandhi said.
The treatment method was discovered more than 30 years ago. Prior to the therapy and ensuing awareness campaigns to prevent transmission, mothers with HIV would typically pass the virus to their child in utero, during delivery, or while breastfeeding.
“There should be zero children born with HIV, given that we’ve had these drugs for so long,” Dr. Ghandi said.
Disparities persist
But challenges remain in some communities, where babies born to Black mothers are disproportionately affected by the disease, the new study found. “Racial and ethnic differences in perinatal HIV diagnoses persisted through the 10-year period,” the report’s authors concluded. “The highest rates of perinatal HIV diagnoses were seen among infants born to Black women.”
Although rates of perinatal HIV declined for babies born to Black mothers over the decade-long study, the diagnosis rate was above the goal of elimination at 3.1 for every 100,000 live births, according to the data.
Meanwhile, transmission rates hovered around 1%-2% for Latinx and Hispanic women and mothers who identified as “other races,” including Native American.
Despite the availability of medication, expectant mothers may face several hurdles to getting the daily treatment they need to prevent transmission to their fetus, according to Jennifer Jao, MD, MPH, a physician of infectious diseases at Lurie Children’s Hospital of Chicago.
They might have trouble securing health insurance or finding transportation to doctor’s appointments, or face other problems like lacking secure housing or food – all factors that prevent them from prioritizing the care.
“All of those things play into the mix,” Dr. Jao said. “We see over and over again that closing the gap means you’ve got to reach the women who are pregnant and who don’t have resources.”
Progress in ‘danger’
Experts said they’re not sure what the impact of the COVID-19 pandemic, accompanied by a recent uptick in sexually transmitted diseases, will be on rates of perinatal HIV. Some women were unable to access prenatal health care during the pandemic because they couldn’t access public transportation or childcare, the U.S. Government Accountability Office said in 2022.
Globally, a decline in rates of HIV and AIDS rates has slowed, prompting the World Health Organization to warn last year that progress on the disease is in danger. Researchers only included HIV rates in the United States through 2019, so the data are outdated, Dr. Gandhi noted.
“All of this put together means we don’t know where we are with perinatal transmission over the last 3 years,” she said.
In an accompanying editorial, coauthors Nahida Chakhtoura, MD, MsGH, and Bill Kapogiannis, MD, both with the National Institutes of Health, urge health care professionals to take an active role in eliminating these racial and ethnic disparities in an effort to – as the title of their editorial proclaims – achieve a “road to zero perinatal HIV transmission” in the United States.
“The more proactive we are in identifying and promptly addressing systematic deficiencies that exacerbate health inequities in cutting-edge research innovations and optimal clinical service provision,” they write, “the less reactive we will need to be when new transmissible infections appear at our doorstep.”
A version of this article first appeared on Medscape.com.
, with less than 1 baby for every 100,000 live births having the virus, a new study released by researchers at the Centers for Disease Control and Prevention finds.
The report marks significant progress on the U.S. government’s goal to eradicate perinatal HIV, an immune-weakening and potentially deadly virus that is passed from mother to baby during pregnancy. Just 32 children in the country were diagnosed in 2019, compared with twice as many in 2010, according to the CDC.
Mothers who are HIV positive can prevent transmission of the infection by receiving antiretroviral therapy, according to Monica Gandhi, MD, MPH, a professor of medicine at University of California, San Francisco’s division of HIV, infectious disease and global medicine.
Dr. Gandhi said she could recall only one case of perinatal HIV in the San Francisco area over the last decade.
“This country has been really aggressive about counseling women who are pregnant and getting mothers in care,” Dr. Gandhi said.
The treatment method was discovered more than 30 years ago. Prior to the therapy and ensuing awareness campaigns to prevent transmission, mothers with HIV would typically pass the virus to their child in utero, during delivery, or while breastfeeding.
“There should be zero children born with HIV, given that we’ve had these drugs for so long,” Dr. Ghandi said.
Disparities persist
But challenges remain in some communities, where babies born to Black mothers are disproportionately affected by the disease, the new study found. “Racial and ethnic differences in perinatal HIV diagnoses persisted through the 10-year period,” the report’s authors concluded. “The highest rates of perinatal HIV diagnoses were seen among infants born to Black women.”
Although rates of perinatal HIV declined for babies born to Black mothers over the decade-long study, the diagnosis rate was above the goal of elimination at 3.1 for every 100,000 live births, according to the data.
Meanwhile, transmission rates hovered around 1%-2% for Latinx and Hispanic women and mothers who identified as “other races,” including Native American.
Despite the availability of medication, expectant mothers may face several hurdles to getting the daily treatment they need to prevent transmission to their fetus, according to Jennifer Jao, MD, MPH, a physician of infectious diseases at Lurie Children’s Hospital of Chicago.
They might have trouble securing health insurance or finding transportation to doctor’s appointments, or face other problems like lacking secure housing or food – all factors that prevent them from prioritizing the care.
“All of those things play into the mix,” Dr. Jao said. “We see over and over again that closing the gap means you’ve got to reach the women who are pregnant and who don’t have resources.”
Progress in ‘danger’
Experts said they’re not sure what the impact of the COVID-19 pandemic, accompanied by a recent uptick in sexually transmitted diseases, will be on rates of perinatal HIV. Some women were unable to access prenatal health care during the pandemic because they couldn’t access public transportation or childcare, the U.S. Government Accountability Office said in 2022.
Globally, a decline in rates of HIV and AIDS rates has slowed, prompting the World Health Organization to warn last year that progress on the disease is in danger. Researchers only included HIV rates in the United States through 2019, so the data are outdated, Dr. Gandhi noted.
“All of this put together means we don’t know where we are with perinatal transmission over the last 3 years,” she said.
In an accompanying editorial, coauthors Nahida Chakhtoura, MD, MsGH, and Bill Kapogiannis, MD, both with the National Institutes of Health, urge health care professionals to take an active role in eliminating these racial and ethnic disparities in an effort to – as the title of their editorial proclaims – achieve a “road to zero perinatal HIV transmission” in the United States.
“The more proactive we are in identifying and promptly addressing systematic deficiencies that exacerbate health inequities in cutting-edge research innovations and optimal clinical service provision,” they write, “the less reactive we will need to be when new transmissible infections appear at our doorstep.”
A version of this article first appeared on Medscape.com.