User login
Can an endoscopic procedure treat type 2 diabetes?
Called recellularization via electroporation therapy (ReCET), the technology, manufactured by Endogenex, uses a specialized catheter to deliver alternating electric pulses to the duodenum to induce cellular regeneration. This process is thought to improve insulin sensitivity, in part, by altering gut hormones and nutritional sensing, principal investigator Jacques Bergman, MD, PhD, said in a press briefing held in conjunction with the annual Digestive Disease Week® (DDW), where he will present the data on May 9.
In the first-in-human study of ReCET, 12 of 14 patients were able to come off insulin for up to a year following the procedure when combined with the use of the glucagonlike peptide–1 agonist semaglutide.
“This might be a game changer in the management of type 2 diabetes because a single outpatient endoscopic intervention was suggested to have a pretty long therapeutic effect, which is compliance-free, as opposed to drug therapy that relies on patients taking the drugs on a daily basis,” said Dr. Bergman, professor of gastrointestinal endoscopy at Amsterdam University Medical Center.
Moreover, he added, “this technique is disease-modifying, so it goes to the root cause of type 2 diabetes and tackles the insulin resistance, as opposed to drug therapy, which at best, is disease-controlling, and the effect is immediately gone if you stop the medication.”
ReCET is similar to another product, Fractyl’s Revita DMR, for which Dr. Bergman was involved in a randomized clinical trial. He said in an interview that the two technologies differ in that the Revita uses heat with submucosal lifting to avoid deeper heat penetration, whereas ReCET is nonthermal. He is also involved in a second randomized trial of the Revita.
Is semaglutide muddying the findings?
Asked to comment about the current study with ReCET, Ali Aminian, MD, professor of surgery and director of the Bariatric and Metabolic Institute at the Cleveland Clinic, said that the treatment effect is certainly plausible.
“The observation that hyperglycemia rapidly and substantially improves after bariatric surgery has prompted innovators to search for novel endoscopic procedures targeting the GI tract to improve diabetes and metabolic disease. Over the years, we learned that in addition to its role in digestion and absorption, the GI tract is actually a large endocrine organ which contributes to development of diabetes and metabolic disease.”
However, Dr. Aminian said that, “while these preliminary findings on a very small number of patients with a very short follow-up time are interesting,” he faulted the study design for including semaglutide. “When patients are treated with a combination of therapies, it will be hard to understand the true effect of each therapy,” and particularly, “when we add a strong diabetes medication like semaglutide.”
Dr. Bergman said semaglutide was used to “boost the insulin-resistant effect of the endoscopic treatment,” and that a planned double-blind, randomized trial will “show how much semaglutide actually contributed to the effect.” The ultimate goal, he noted, is to eliminate the need for all medications.
Moreover, when people with type 2 diabetes add semaglutide to insulin treatment, only about 20% typically are able to quit taking the insulin, in contrast to the 86% seen in this study, lead author Celine Busch, MBBS, a PhD candidate in gastroenterology at Amsterdam University, said in a DDW statement.
Dr. Aminian said, “I’m looking forward to better quality data ... from studies with a stronger design to prove safety, efficacy, and durability of this endoscopic intervention in patients with diabetes.”
But, he also cautioned, “in the past few years, other endoscopic procedures targeting the duodenum were introduced with exciting initial findings based on a small series [with a] short-term follow-up time. However, their safety, efficacy, and durability were not proven in subsequent studies.”
All patients stopped insulin, most for a year
The single-arm, single-center study involved 14 patients with type 2 diabetes taking basal but not premeal insulin. All underwent the 1-hour outpatient ReCET procedure, which involved placing a catheter into the first part of the small bowel and delivering electrical pulses to the duodenum.
Patients adhered to a calorie-controlled liquid diet for 2 weeks, after which they were initiated on semaglutide. All 14 patients were able to come off insulin for 3 months while maintaining glycemic control, and 12 were able to come off insulin for 12 months. They also experienced a 50% reduction in liver fat.
Dr. Bergman said a randomized, double-blind study using a sham procedure for controls is expected to start in about 2 months. “But for now, we are very encouraged by the potential for controlling type 2 diabetes with a single endoscopic treatment.”
Dr. Bergman has reported serving on the advisory board for Endogenex. Dr. Aminian has reported receiving research support and honorarium from Medtronic and Ethicon.
The meeting is sponsored by the American Gastroenterological Association, the American Association for the Study of Liver Diseases, the American Society for Gastrointestinal Endoscopy, and the Society for Surgery of the Alimentary Tract.
A version of this article first appeared on Medscape.com.
Called recellularization via electroporation therapy (ReCET), the technology, manufactured by Endogenex, uses a specialized catheter to deliver alternating electric pulses to the duodenum to induce cellular regeneration. This process is thought to improve insulin sensitivity, in part, by altering gut hormones and nutritional sensing, principal investigator Jacques Bergman, MD, PhD, said in a press briefing held in conjunction with the annual Digestive Disease Week® (DDW), where he will present the data on May 9.
In the first-in-human study of ReCET, 12 of 14 patients were able to come off insulin for up to a year following the procedure when combined with the use of the glucagonlike peptide–1 agonist semaglutide.
“This might be a game changer in the management of type 2 diabetes because a single outpatient endoscopic intervention was suggested to have a pretty long therapeutic effect, which is compliance-free, as opposed to drug therapy that relies on patients taking the drugs on a daily basis,” said Dr. Bergman, professor of gastrointestinal endoscopy at Amsterdam University Medical Center.
Moreover, he added, “this technique is disease-modifying, so it goes to the root cause of type 2 diabetes and tackles the insulin resistance, as opposed to drug therapy, which at best, is disease-controlling, and the effect is immediately gone if you stop the medication.”
ReCET is similar to another product, Fractyl’s Revita DMR, for which Dr. Bergman was involved in a randomized clinical trial. He said in an interview that the two technologies differ in that the Revita uses heat with submucosal lifting to avoid deeper heat penetration, whereas ReCET is nonthermal. He is also involved in a second randomized trial of the Revita.
Is semaglutide muddying the findings?
Asked to comment about the current study with ReCET, Ali Aminian, MD, professor of surgery and director of the Bariatric and Metabolic Institute at the Cleveland Clinic, said that the treatment effect is certainly plausible.
“The observation that hyperglycemia rapidly and substantially improves after bariatric surgery has prompted innovators to search for novel endoscopic procedures targeting the GI tract to improve diabetes and metabolic disease. Over the years, we learned that in addition to its role in digestion and absorption, the GI tract is actually a large endocrine organ which contributes to development of diabetes and metabolic disease.”
However, Dr. Aminian said that, “while these preliminary findings on a very small number of patients with a very short follow-up time are interesting,” he faulted the study design for including semaglutide. “When patients are treated with a combination of therapies, it will be hard to understand the true effect of each therapy,” and particularly, “when we add a strong diabetes medication like semaglutide.”
Dr. Bergman said semaglutide was used to “boost the insulin-resistant effect of the endoscopic treatment,” and that a planned double-blind, randomized trial will “show how much semaglutide actually contributed to the effect.” The ultimate goal, he noted, is to eliminate the need for all medications.
Moreover, when people with type 2 diabetes add semaglutide to insulin treatment, only about 20% typically are able to quit taking the insulin, in contrast to the 86% seen in this study, lead author Celine Busch, MBBS, a PhD candidate in gastroenterology at Amsterdam University, said in a DDW statement.
Dr. Aminian said, “I’m looking forward to better quality data ... from studies with a stronger design to prove safety, efficacy, and durability of this endoscopic intervention in patients with diabetes.”
But, he also cautioned, “in the past few years, other endoscopic procedures targeting the duodenum were introduced with exciting initial findings based on a small series [with a] short-term follow-up time. However, their safety, efficacy, and durability were not proven in subsequent studies.”
All patients stopped insulin, most for a year
The single-arm, single-center study involved 14 patients with type 2 diabetes taking basal but not premeal insulin. All underwent the 1-hour outpatient ReCET procedure, which involved placing a catheter into the first part of the small bowel and delivering electrical pulses to the duodenum.
Patients adhered to a calorie-controlled liquid diet for 2 weeks, after which they were initiated on semaglutide. All 14 patients were able to come off insulin for 3 months while maintaining glycemic control, and 12 were able to come off insulin for 12 months. They also experienced a 50% reduction in liver fat.
Dr. Bergman said a randomized, double-blind study using a sham procedure for controls is expected to start in about 2 months. “But for now, we are very encouraged by the potential for controlling type 2 diabetes with a single endoscopic treatment.”
Dr. Bergman has reported serving on the advisory board for Endogenex. Dr. Aminian has reported receiving research support and honorarium from Medtronic and Ethicon.
The meeting is sponsored by the American Gastroenterological Association, the American Association for the Study of Liver Diseases, the American Society for Gastrointestinal Endoscopy, and the Society for Surgery of the Alimentary Tract.
A version of this article first appeared on Medscape.com.
Called recellularization via electroporation therapy (ReCET), the technology, manufactured by Endogenex, uses a specialized catheter to deliver alternating electric pulses to the duodenum to induce cellular regeneration. This process is thought to improve insulin sensitivity, in part, by altering gut hormones and nutritional sensing, principal investigator Jacques Bergman, MD, PhD, said in a press briefing held in conjunction with the annual Digestive Disease Week® (DDW), where he will present the data on May 9.
In the first-in-human study of ReCET, 12 of 14 patients were able to come off insulin for up to a year following the procedure when combined with the use of the glucagonlike peptide–1 agonist semaglutide.
“This might be a game changer in the management of type 2 diabetes because a single outpatient endoscopic intervention was suggested to have a pretty long therapeutic effect, which is compliance-free, as opposed to drug therapy that relies on patients taking the drugs on a daily basis,” said Dr. Bergman, professor of gastrointestinal endoscopy at Amsterdam University Medical Center.
Moreover, he added, “this technique is disease-modifying, so it goes to the root cause of type 2 diabetes and tackles the insulin resistance, as opposed to drug therapy, which at best, is disease-controlling, and the effect is immediately gone if you stop the medication.”
ReCET is similar to another product, Fractyl’s Revita DMR, for which Dr. Bergman was involved in a randomized clinical trial. He said in an interview that the two technologies differ in that the Revita uses heat with submucosal lifting to avoid deeper heat penetration, whereas ReCET is nonthermal. He is also involved in a second randomized trial of the Revita.
Is semaglutide muddying the findings?
Asked to comment about the current study with ReCET, Ali Aminian, MD, professor of surgery and director of the Bariatric and Metabolic Institute at the Cleveland Clinic, said that the treatment effect is certainly plausible.
“The observation that hyperglycemia rapidly and substantially improves after bariatric surgery has prompted innovators to search for novel endoscopic procedures targeting the GI tract to improve diabetes and metabolic disease. Over the years, we learned that in addition to its role in digestion and absorption, the GI tract is actually a large endocrine organ which contributes to development of diabetes and metabolic disease.”
However, Dr. Aminian said that, “while these preliminary findings on a very small number of patients with a very short follow-up time are interesting,” he faulted the study design for including semaglutide. “When patients are treated with a combination of therapies, it will be hard to understand the true effect of each therapy,” and particularly, “when we add a strong diabetes medication like semaglutide.”
Dr. Bergman said semaglutide was used to “boost the insulin-resistant effect of the endoscopic treatment,” and that a planned double-blind, randomized trial will “show how much semaglutide actually contributed to the effect.” The ultimate goal, he noted, is to eliminate the need for all medications.
Moreover, when people with type 2 diabetes add semaglutide to insulin treatment, only about 20% typically are able to quit taking the insulin, in contrast to the 86% seen in this study, lead author Celine Busch, MBBS, a PhD candidate in gastroenterology at Amsterdam University, said in a DDW statement.
Dr. Aminian said, “I’m looking forward to better quality data ... from studies with a stronger design to prove safety, efficacy, and durability of this endoscopic intervention in patients with diabetes.”
But, he also cautioned, “in the past few years, other endoscopic procedures targeting the duodenum were introduced with exciting initial findings based on a small series [with a] short-term follow-up time. However, their safety, efficacy, and durability were not proven in subsequent studies.”
All patients stopped insulin, most for a year
The single-arm, single-center study involved 14 patients with type 2 diabetes taking basal but not premeal insulin. All underwent the 1-hour outpatient ReCET procedure, which involved placing a catheter into the first part of the small bowel and delivering electrical pulses to the duodenum.
Patients adhered to a calorie-controlled liquid diet for 2 weeks, after which they were initiated on semaglutide. All 14 patients were able to come off insulin for 3 months while maintaining glycemic control, and 12 were able to come off insulin for 12 months. They also experienced a 50% reduction in liver fat.
Dr. Bergman said a randomized, double-blind study using a sham procedure for controls is expected to start in about 2 months. “But for now, we are very encouraged by the potential for controlling type 2 diabetes with a single endoscopic treatment.”
Dr. Bergman has reported serving on the advisory board for Endogenex. Dr. Aminian has reported receiving research support and honorarium from Medtronic and Ethicon.
The meeting is sponsored by the American Gastroenterological Association, the American Association for the Study of Liver Diseases, the American Society for Gastrointestinal Endoscopy, and the Society for Surgery of the Alimentary Tract.
A version of this article first appeared on Medscape.com.
FROM DDW 2023
Personalizing treatment plans for older patients with T2D
In the United States, type 2 diabetes (T2D) more commonly affects people older than 40 years, but it is most prevalent among adults over age 65, affecting more than 29% of this population. The heterogeneity in the health and functional status of older adults presents a challenge in the management and treatment of older patients with T2D. Moreover, there is an increased risk for health-related comorbidities and complications from diabetes treatment (for example, hypoglycemia) in older adults. Physiologic changes, such as decreased renal function, cognitive decline, and sarcopenia, may lead to an increased risk for adverse reactions to medications and require an individualized treatment approach. Although there have been a limited number of randomized controlled studies targeting older adults with multiple comorbidities and poor health status, subanalyses of diabetes trials with a subpopulation of older adults have provided additional evidence to better guide therapeutic approaches in caring for older patients with T2D.
Here’s a guide to developing personalized therapeutic regimens for older patients with T2D using lifestyle interventions, pharmacotherapy, and diabetes technology.
Determining an optimal glycemic target
An important first step in diabetes treatment is to determine the optimal glycemic target for patients. Although data support intensive glycemic control (hemoglobin A1c < 7%) to prevent complications from diabetes in younger patients with recently diagnosed disease, the data are less compelling in trials involving older populations with longer durations of T2D. One observational study with 71,092 older adults over age 60 reported a U-shaped correlation between A1c and mortality, with higher risks for mortality in those with A1c levels < 6% and ≥ 11%, compared with those with A1c levels of 6%-9%. Risks for any diabetes complications were higher at an A1c level ≥ 8%. Another observational study reported a U-shaped association between A1c and mortality, with the lowest hazard ratio for mortality at an A1c level of about 7.5%. Similarly, the ACCORD trial, which included older and middle-aged patients with T2D who had or were at risk for atherosclerotic cardiovascular disease, found that mortality followed a U-shaped curve at the low (A1c < 7%) and high (A1c > 8%) ends in patients who were given standard glycemic therapy. Hence, there has been a general trend to recommend less strict glycemic control in older adults.
However, it is important to remember that older patients with T2D are a heterogeneous group. The spectrum includes adults with recent-onset diabetes with no or few complications, those with long-standing diabetes and many complications, and frail older adults with multiple comorbidities and complications. Determining the optimal glycemic target for an older patient with T2D requires assessment not only of the patient’s medical status and comorbidities but also functional status, cognitive and psychological health, social situation, individual preferences, and life expectancy. The American Diabetes Association Standards of Medical Care in Diabetes provides the following guidance in determining the optimal glycemic control for older adults:
- Healthy adults with few coexisting chronic illnesses and intact cognitive and functional status should have an A1c level < 7.0%-7.5%.
- Adults with complex or intermediate comorbidities (multiple coexisting chronic illnesses, or two or more instrumental activities of daily living impairments, or mild to moderate cognitive impairment) should have an A1c level < 8.0%.
- Patients with poor health (long-term care or end-stage chronic illnesses or moderate to severe cognitive impairment or two or more activities of daily living impairments) should avoid reliance on A1c, and the goal is to avoid hypoglycemia and symptomatic hyperglycemia.
Because older patients are at a higher risk for complications and adverse effects from polypharmacy, regular assessments are recommended and treatment plans should be routinely reviewed and modified to avoid overtreatment.
Lifestyle interventions and pharmacotherapy
Lifestyle interventions, such as exercise, optimal nutrition, and protein intake, are integral in treating older patients with T2D. Older adults should engage in regular exercise (that is, aerobic activity, weight-bearing exercise, or resistance training), and the activity should be customized to frailty status. Regular exercise improves insulin sensitivity and glucose control, enhances functional status, and provides cardiometabolic benefits. Optimal nutrition and adequate protein intake are also important to prevent the development or worsening of sarcopenia and frailty.
Several factors must be considered when choosing pharmacotherapy for T2D treatment in older adults. These patients are at higher risk for adverse reactions to medications that can trigger hypoglycemia and serious cardiovascular events, and worsen cognitive function. Therefore, side effects should always be reviewed when choosing antidiabetic drugs. The complexity of treatment plans needs to be matched with the patients’ self-management abilities and available social support. Medication costs and insurance coverage should be considered because many older adults live on a fixed income. Although limited, data exist on the safety and efficacy of some glucose-lowering agents in older adults, which can provide guidance for choosing the optimal therapy for these patients.
Among the insulin sensitizers, metformin is most commonly used because of its efficacy, low risk for hypoglycemia, and affordability. Metformin can be safely used in the setting of reduced renal function down to the estimated glomerular filtration rate ≥ 30 mL/min per 1.73 m2. However, metformin should be avoided in patients with more advanced renal disease, liver failure, or heart failure. In older patients with T2D, potential concerns of metformin include gastrointestinal side effects, leading to reduced appetite, mild weight loss, and risk for vitamin B12 deficiency.
Pioglitazone, an oral antidiabetic in the thiazolidinedione (TZD) class, also targets insulin resistance and may provide some cardiovascular benefits. However, these agents are not commonly used in treating older patients with T2D owing to associated risk for edema, heart failure, osteoporosis/fractures, and bladder cancer.
Sulfonylureas and meglitinides are insulin secretagogues, which can promote insulin release independent of glucose levels. Sulfonylureas are typically avoided in older patients because they are associated with high risk for hypoglycemia. Meglitinides have a lower hypoglycemia risk than sulfonylureas because of their short duration of action; however, they are more expensive and require multiple daily administration, which can lead to issues with adherence.
Since 2008, there have been numerous cardiovascular outcomes trials assessing the safety and efficacy of T2D therapies that included a subpopulation of older patients either with cardiovascular disease or at high risk for cardiovascular disease. Post hoc analysis of data from these trials and smaller studies dedicated to older adults demonstrated the safety and efficacy of most incretin-based therapies and sodium-glucose cotransporter 2 (SGLT2) inhibitors in these patients. These newer medications have low hypoglycemia risk if not used in combination with insulin or insulin secretagogues.
Dipeptidyl peptidase 4 (DPP-4) inhibitors have the mildest side effect profile. However, they can be expensive and not reduce major adverse cardiovascular outcomes, and one agent, saxagliptin, has been associated with increased risk for heart failure hospitalization. Some glucagon-like peptide 1 (GLP-1) receptor agonists are effective in reducing major adverse cardiovascular events (cardiovascular deaths, stroke, and myocardial infarction) in patients older and younger than age 65. However, the gastrointestinal side effects and weight loss associated with this medication can be problematic for older patients. Most of the GLP-1 receptor agonists are injectables, which require good visual, motor, and cognitive skills for administration. SGLT2 inhibitors offer benefits for patients with T2D who have established cardiovascular disease, heart failure, and chronic kidney disease, with possible greater cardiovascular benefits in older adults. Adverse effects associated with SGLT2 inhibitors, such as weight loss, volume depletion, urinary incontinence, and genitourinary infections, may be a concern in older patients with T2D who are using these medications.
Because the insulin-secreting capacity of the pancreas declines with age, insulin therapy may be required for treatment of T2D in older patients. Insulin therapy can be complex and consideration must be given to patients’ social circumstances, as well as their physical and cognitive abilities. Older adults may need adaptive strategies, such as additional lighting, magnification glass, and premixed syringes. Simplification of complex insulin therapy (discontinuation of prandial insulin or sliding scale, changing timing of basal insulin) and use of insulin analogs with lower hypoglycemia risks should be considered. Weight gain as a result of insulin therapy may be beneficial in older adults with sarcopenia or frailty.
T2D technology for glycemic improvement
There have been major technological advancements in diabetes therapy. Continuous glucose monitors (CGMs) and automated insulin delivery systems can improve glycemic control, decrease the rate of hypoglycemia, and enhance the quality of life of older patients. Most of the studies evaluating the use of automated insulin delivery systems in older patients have focused on those with type 1 diabetes and demonstrated improvement in glycemic control and/or reduced hypoglycemia. The DIAMOND trial demonstrated improved A1c and reduced glycemic variability with the use of CGM in adults older than 60 years with either type 1 or type 2 diabetes on multiple daily injections. Bluetooth-enabled “smart” insulin pens, which record the time and dose of insulin administrations, can also be a great asset in caring for older patients, especially those with cognitive impairment. With better insurance coverage, diabetes technologies may become more accessible and an asset in treating older patients with T2D.
In conclusion, management of T2D in older adults requires an individualized approach because of the heterogeneity in their health and functional status. Because cardiovascular disease is the leading cause of mortality in older patients with T2D, treatment plans should also address frequently coexisting cardiovascular risk factors, such as hypertension and hyperlipidemia. Clinicians should consider patients’ overall health, comorbidities, cognitive and functional status, social support systems, preferences, and life expectancy when developing individualized therapeutic plans.
Dr. Gunawan is an assistant professor in the department of internal medicine at UT Southwestern Medical Center, Dallas. She reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
In the United States, type 2 diabetes (T2D) more commonly affects people older than 40 years, but it is most prevalent among adults over age 65, affecting more than 29% of this population. The heterogeneity in the health and functional status of older adults presents a challenge in the management and treatment of older patients with T2D. Moreover, there is an increased risk for health-related comorbidities and complications from diabetes treatment (for example, hypoglycemia) in older adults. Physiologic changes, such as decreased renal function, cognitive decline, and sarcopenia, may lead to an increased risk for adverse reactions to medications and require an individualized treatment approach. Although there have been a limited number of randomized controlled studies targeting older adults with multiple comorbidities and poor health status, subanalyses of diabetes trials with a subpopulation of older adults have provided additional evidence to better guide therapeutic approaches in caring for older patients with T2D.
Here’s a guide to developing personalized therapeutic regimens for older patients with T2D using lifestyle interventions, pharmacotherapy, and diabetes technology.
Determining an optimal glycemic target
An important first step in diabetes treatment is to determine the optimal glycemic target for patients. Although data support intensive glycemic control (hemoglobin A1c < 7%) to prevent complications from diabetes in younger patients with recently diagnosed disease, the data are less compelling in trials involving older populations with longer durations of T2D. One observational study with 71,092 older adults over age 60 reported a U-shaped correlation between A1c and mortality, with higher risks for mortality in those with A1c levels < 6% and ≥ 11%, compared with those with A1c levels of 6%-9%. Risks for any diabetes complications were higher at an A1c level ≥ 8%. Another observational study reported a U-shaped association between A1c and mortality, with the lowest hazard ratio for mortality at an A1c level of about 7.5%. Similarly, the ACCORD trial, which included older and middle-aged patients with T2D who had or were at risk for atherosclerotic cardiovascular disease, found that mortality followed a U-shaped curve at the low (A1c < 7%) and high (A1c > 8%) ends in patients who were given standard glycemic therapy. Hence, there has been a general trend to recommend less strict glycemic control in older adults.
However, it is important to remember that older patients with T2D are a heterogeneous group. The spectrum includes adults with recent-onset diabetes with no or few complications, those with long-standing diabetes and many complications, and frail older adults with multiple comorbidities and complications. Determining the optimal glycemic target for an older patient with T2D requires assessment not only of the patient’s medical status and comorbidities but also functional status, cognitive and psychological health, social situation, individual preferences, and life expectancy. The American Diabetes Association Standards of Medical Care in Diabetes provides the following guidance in determining the optimal glycemic control for older adults:
- Healthy adults with few coexisting chronic illnesses and intact cognitive and functional status should have an A1c level < 7.0%-7.5%.
- Adults with complex or intermediate comorbidities (multiple coexisting chronic illnesses, or two or more instrumental activities of daily living impairments, or mild to moderate cognitive impairment) should have an A1c level < 8.0%.
- Patients with poor health (long-term care or end-stage chronic illnesses or moderate to severe cognitive impairment or two or more activities of daily living impairments) should avoid reliance on A1c, and the goal is to avoid hypoglycemia and symptomatic hyperglycemia.
Because older patients are at a higher risk for complications and adverse effects from polypharmacy, regular assessments are recommended and treatment plans should be routinely reviewed and modified to avoid overtreatment.
Lifestyle interventions and pharmacotherapy
Lifestyle interventions, such as exercise, optimal nutrition, and protein intake, are integral in treating older patients with T2D. Older adults should engage in regular exercise (that is, aerobic activity, weight-bearing exercise, or resistance training), and the activity should be customized to frailty status. Regular exercise improves insulin sensitivity and glucose control, enhances functional status, and provides cardiometabolic benefits. Optimal nutrition and adequate protein intake are also important to prevent the development or worsening of sarcopenia and frailty.
Several factors must be considered when choosing pharmacotherapy for T2D treatment in older adults. These patients are at higher risk for adverse reactions to medications that can trigger hypoglycemia and serious cardiovascular events, and worsen cognitive function. Therefore, side effects should always be reviewed when choosing antidiabetic drugs. The complexity of treatment plans needs to be matched with the patients’ self-management abilities and available social support. Medication costs and insurance coverage should be considered because many older adults live on a fixed income. Although limited, data exist on the safety and efficacy of some glucose-lowering agents in older adults, which can provide guidance for choosing the optimal therapy for these patients.
Among the insulin sensitizers, metformin is most commonly used because of its efficacy, low risk for hypoglycemia, and affordability. Metformin can be safely used in the setting of reduced renal function down to the estimated glomerular filtration rate ≥ 30 mL/min per 1.73 m2. However, metformin should be avoided in patients with more advanced renal disease, liver failure, or heart failure. In older patients with T2D, potential concerns of metformin include gastrointestinal side effects, leading to reduced appetite, mild weight loss, and risk for vitamin B12 deficiency.
Pioglitazone, an oral antidiabetic in the thiazolidinedione (TZD) class, also targets insulin resistance and may provide some cardiovascular benefits. However, these agents are not commonly used in treating older patients with T2D owing to associated risk for edema, heart failure, osteoporosis/fractures, and bladder cancer.
Sulfonylureas and meglitinides are insulin secretagogues, which can promote insulin release independent of glucose levels. Sulfonylureas are typically avoided in older patients because they are associated with high risk for hypoglycemia. Meglitinides have a lower hypoglycemia risk than sulfonylureas because of their short duration of action; however, they are more expensive and require multiple daily administration, which can lead to issues with adherence.
Since 2008, there have been numerous cardiovascular outcomes trials assessing the safety and efficacy of T2D therapies that included a subpopulation of older patients either with cardiovascular disease or at high risk for cardiovascular disease. Post hoc analysis of data from these trials and smaller studies dedicated to older adults demonstrated the safety and efficacy of most incretin-based therapies and sodium-glucose cotransporter 2 (SGLT2) inhibitors in these patients. These newer medications have low hypoglycemia risk if not used in combination with insulin or insulin secretagogues.
Dipeptidyl peptidase 4 (DPP-4) inhibitors have the mildest side effect profile. However, they can be expensive and not reduce major adverse cardiovascular outcomes, and one agent, saxagliptin, has been associated with increased risk for heart failure hospitalization. Some glucagon-like peptide 1 (GLP-1) receptor agonists are effective in reducing major adverse cardiovascular events (cardiovascular deaths, stroke, and myocardial infarction) in patients older and younger than age 65. However, the gastrointestinal side effects and weight loss associated with this medication can be problematic for older patients. Most of the GLP-1 receptor agonists are injectables, which require good visual, motor, and cognitive skills for administration. SGLT2 inhibitors offer benefits for patients with T2D who have established cardiovascular disease, heart failure, and chronic kidney disease, with possible greater cardiovascular benefits in older adults. Adverse effects associated with SGLT2 inhibitors, such as weight loss, volume depletion, urinary incontinence, and genitourinary infections, may be a concern in older patients with T2D who are using these medications.
Because the insulin-secreting capacity of the pancreas declines with age, insulin therapy may be required for treatment of T2D in older patients. Insulin therapy can be complex and consideration must be given to patients’ social circumstances, as well as their physical and cognitive abilities. Older adults may need adaptive strategies, such as additional lighting, magnification glass, and premixed syringes. Simplification of complex insulin therapy (discontinuation of prandial insulin or sliding scale, changing timing of basal insulin) and use of insulin analogs with lower hypoglycemia risks should be considered. Weight gain as a result of insulin therapy may be beneficial in older adults with sarcopenia or frailty.
T2D technology for glycemic improvement
There have been major technological advancements in diabetes therapy. Continuous glucose monitors (CGMs) and automated insulin delivery systems can improve glycemic control, decrease the rate of hypoglycemia, and enhance the quality of life of older patients. Most of the studies evaluating the use of automated insulin delivery systems in older patients have focused on those with type 1 diabetes and demonstrated improvement in glycemic control and/or reduced hypoglycemia. The DIAMOND trial demonstrated improved A1c and reduced glycemic variability with the use of CGM in adults older than 60 years with either type 1 or type 2 diabetes on multiple daily injections. Bluetooth-enabled “smart” insulin pens, which record the time and dose of insulin administrations, can also be a great asset in caring for older patients, especially those with cognitive impairment. With better insurance coverage, diabetes technologies may become more accessible and an asset in treating older patients with T2D.
In conclusion, management of T2D in older adults requires an individualized approach because of the heterogeneity in their health and functional status. Because cardiovascular disease is the leading cause of mortality in older patients with T2D, treatment plans should also address frequently coexisting cardiovascular risk factors, such as hypertension and hyperlipidemia. Clinicians should consider patients’ overall health, comorbidities, cognitive and functional status, social support systems, preferences, and life expectancy when developing individualized therapeutic plans.
Dr. Gunawan is an assistant professor in the department of internal medicine at UT Southwestern Medical Center, Dallas. She reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
In the United States, type 2 diabetes (T2D) more commonly affects people older than 40 years, but it is most prevalent among adults over age 65, affecting more than 29% of this population. The heterogeneity in the health and functional status of older adults presents a challenge in the management and treatment of older patients with T2D. Moreover, there is an increased risk for health-related comorbidities and complications from diabetes treatment (for example, hypoglycemia) in older adults. Physiologic changes, such as decreased renal function, cognitive decline, and sarcopenia, may lead to an increased risk for adverse reactions to medications and require an individualized treatment approach. Although there have been a limited number of randomized controlled studies targeting older adults with multiple comorbidities and poor health status, subanalyses of diabetes trials with a subpopulation of older adults have provided additional evidence to better guide therapeutic approaches in caring for older patients with T2D.
Here’s a guide to developing personalized therapeutic regimens for older patients with T2D using lifestyle interventions, pharmacotherapy, and diabetes technology.
Determining an optimal glycemic target
An important first step in diabetes treatment is to determine the optimal glycemic target for patients. Although data support intensive glycemic control (hemoglobin A1c < 7%) to prevent complications from diabetes in younger patients with recently diagnosed disease, the data are less compelling in trials involving older populations with longer durations of T2D. One observational study with 71,092 older adults over age 60 reported a U-shaped correlation between A1c and mortality, with higher risks for mortality in those with A1c levels < 6% and ≥ 11%, compared with those with A1c levels of 6%-9%. Risks for any diabetes complications were higher at an A1c level ≥ 8%. Another observational study reported a U-shaped association between A1c and mortality, with the lowest hazard ratio for mortality at an A1c level of about 7.5%. Similarly, the ACCORD trial, which included older and middle-aged patients with T2D who had or were at risk for atherosclerotic cardiovascular disease, found that mortality followed a U-shaped curve at the low (A1c < 7%) and high (A1c > 8%) ends in patients who were given standard glycemic therapy. Hence, there has been a general trend to recommend less strict glycemic control in older adults.
However, it is important to remember that older patients with T2D are a heterogeneous group. The spectrum includes adults with recent-onset diabetes with no or few complications, those with long-standing diabetes and many complications, and frail older adults with multiple comorbidities and complications. Determining the optimal glycemic target for an older patient with T2D requires assessment not only of the patient’s medical status and comorbidities but also functional status, cognitive and psychological health, social situation, individual preferences, and life expectancy. The American Diabetes Association Standards of Medical Care in Diabetes provides the following guidance in determining the optimal glycemic control for older adults:
- Healthy adults with few coexisting chronic illnesses and intact cognitive and functional status should have an A1c level < 7.0%-7.5%.
- Adults with complex or intermediate comorbidities (multiple coexisting chronic illnesses, or two or more instrumental activities of daily living impairments, or mild to moderate cognitive impairment) should have an A1c level < 8.0%.
- Patients with poor health (long-term care or end-stage chronic illnesses or moderate to severe cognitive impairment or two or more activities of daily living impairments) should avoid reliance on A1c, and the goal is to avoid hypoglycemia and symptomatic hyperglycemia.
Because older patients are at a higher risk for complications and adverse effects from polypharmacy, regular assessments are recommended and treatment plans should be routinely reviewed and modified to avoid overtreatment.
Lifestyle interventions and pharmacotherapy
Lifestyle interventions, such as exercise, optimal nutrition, and protein intake, are integral in treating older patients with T2D. Older adults should engage in regular exercise (that is, aerobic activity, weight-bearing exercise, or resistance training), and the activity should be customized to frailty status. Regular exercise improves insulin sensitivity and glucose control, enhances functional status, and provides cardiometabolic benefits. Optimal nutrition and adequate protein intake are also important to prevent the development or worsening of sarcopenia and frailty.
Several factors must be considered when choosing pharmacotherapy for T2D treatment in older adults. These patients are at higher risk for adverse reactions to medications that can trigger hypoglycemia and serious cardiovascular events, and worsen cognitive function. Therefore, side effects should always be reviewed when choosing antidiabetic drugs. The complexity of treatment plans needs to be matched with the patients’ self-management abilities and available social support. Medication costs and insurance coverage should be considered because many older adults live on a fixed income. Although limited, data exist on the safety and efficacy of some glucose-lowering agents in older adults, which can provide guidance for choosing the optimal therapy for these patients.
Among the insulin sensitizers, metformin is most commonly used because of its efficacy, low risk for hypoglycemia, and affordability. Metformin can be safely used in the setting of reduced renal function down to the estimated glomerular filtration rate ≥ 30 mL/min per 1.73 m2. However, metformin should be avoided in patients with more advanced renal disease, liver failure, or heart failure. In older patients with T2D, potential concerns of metformin include gastrointestinal side effects, leading to reduced appetite, mild weight loss, and risk for vitamin B12 deficiency.
Pioglitazone, an oral antidiabetic in the thiazolidinedione (TZD) class, also targets insulin resistance and may provide some cardiovascular benefits. However, these agents are not commonly used in treating older patients with T2D owing to associated risk for edema, heart failure, osteoporosis/fractures, and bladder cancer.
Sulfonylureas and meglitinides are insulin secretagogues, which can promote insulin release independent of glucose levels. Sulfonylureas are typically avoided in older patients because they are associated with high risk for hypoglycemia. Meglitinides have a lower hypoglycemia risk than sulfonylureas because of their short duration of action; however, they are more expensive and require multiple daily administration, which can lead to issues with adherence.
Since 2008, there have been numerous cardiovascular outcomes trials assessing the safety and efficacy of T2D therapies that included a subpopulation of older patients either with cardiovascular disease or at high risk for cardiovascular disease. Post hoc analysis of data from these trials and smaller studies dedicated to older adults demonstrated the safety and efficacy of most incretin-based therapies and sodium-glucose cotransporter 2 (SGLT2) inhibitors in these patients. These newer medications have low hypoglycemia risk if not used in combination with insulin or insulin secretagogues.
Dipeptidyl peptidase 4 (DPP-4) inhibitors have the mildest side effect profile. However, they can be expensive and not reduce major adverse cardiovascular outcomes, and one agent, saxagliptin, has been associated with increased risk for heart failure hospitalization. Some glucagon-like peptide 1 (GLP-1) receptor agonists are effective in reducing major adverse cardiovascular events (cardiovascular deaths, stroke, and myocardial infarction) in patients older and younger than age 65. However, the gastrointestinal side effects and weight loss associated with this medication can be problematic for older patients. Most of the GLP-1 receptor agonists are injectables, which require good visual, motor, and cognitive skills for administration. SGLT2 inhibitors offer benefits for patients with T2D who have established cardiovascular disease, heart failure, and chronic kidney disease, with possible greater cardiovascular benefits in older adults. Adverse effects associated with SGLT2 inhibitors, such as weight loss, volume depletion, urinary incontinence, and genitourinary infections, may be a concern in older patients with T2D who are using these medications.
Because the insulin-secreting capacity of the pancreas declines with age, insulin therapy may be required for treatment of T2D in older patients. Insulin therapy can be complex and consideration must be given to patients’ social circumstances, as well as their physical and cognitive abilities. Older adults may need adaptive strategies, such as additional lighting, magnification glass, and premixed syringes. Simplification of complex insulin therapy (discontinuation of prandial insulin or sliding scale, changing timing of basal insulin) and use of insulin analogs with lower hypoglycemia risks should be considered. Weight gain as a result of insulin therapy may be beneficial in older adults with sarcopenia or frailty.
T2D technology for glycemic improvement
There have been major technological advancements in diabetes therapy. Continuous glucose monitors (CGMs) and automated insulin delivery systems can improve glycemic control, decrease the rate of hypoglycemia, and enhance the quality of life of older patients. Most of the studies evaluating the use of automated insulin delivery systems in older patients have focused on those with type 1 diabetes and demonstrated improvement in glycemic control and/or reduced hypoglycemia. The DIAMOND trial demonstrated improved A1c and reduced glycemic variability with the use of CGM in adults older than 60 years with either type 1 or type 2 diabetes on multiple daily injections. Bluetooth-enabled “smart” insulin pens, which record the time and dose of insulin administrations, can also be a great asset in caring for older patients, especially those with cognitive impairment. With better insurance coverage, diabetes technologies may become more accessible and an asset in treating older patients with T2D.
In conclusion, management of T2D in older adults requires an individualized approach because of the heterogeneity in their health and functional status. Because cardiovascular disease is the leading cause of mortality in older patients with T2D, treatment plans should also address frequently coexisting cardiovascular risk factors, such as hypertension and hyperlipidemia. Clinicians should consider patients’ overall health, comorbidities, cognitive and functional status, social support systems, preferences, and life expectancy when developing individualized therapeutic plans.
Dr. Gunawan is an assistant professor in the department of internal medicine at UT Southwestern Medical Center, Dallas. She reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Smoking cessation has many benefits in diabetes
MONTPELLIER, FRANCE – The first expert consensus on smoking and diabetes, coauthored by the Francophone Diabetes Society (SFD) and the French Society for the Study of Nicotine Addiction (SFT), was presented at the SFD’s annual conference.
Alexia Rouland, MD, an endocrinologist at Dijon Bourgogne University Hospital, Dijon, France, took the conference as an opportunity to list the many benefits of smoking cessation for patients with diabetes, despite the “slight and temporary” risk for blood sugar imbalance.
Societies target smoking
Diabetes societies around Europe have set their sights on the topic of smoking. Indeed, the guidelines published in 2019 by the European Association for the Study of Diabetes and the European Society of Cardiology state that “smoking cessation is obligatory for all prediabetic and diabetic patients” (class I, level A).
This year, the France-based SFD and SFT dedicated an expert consensus to the major problem of smoking in patients with diabetes. The aim was to provide health care professionals with convincing, well-supported arguments in favor of smoking cessation in their patients with type 1 and type 2 diabetes.
“Before anything else, diabetic patients need to be made aware of the risks of smoking,” said Dr. Rouland. “It’s not just about the fear factor, though. It’s also about providing a positive incentive – they need to be told about the ways they’ll benefit from quitting smoking. For example, you have all-cause mortality, macro- and microangiopathic complications, and so on.”
Duration of abstinence
“Diabetic patients who have stopped smoking have a relative risk for all-cause mortality of 1.28 (1.09-1.51), which is less than what you see in active smokers (relative risk = 1.58; 1.42-1.77), but still above that of nonsmokers,” said Dr. Rouland.
A previous study revealed that although the risk does indeed go down after stopping smoking, it is linked to how long ago the person stopped. Patients who stopped smoking less than 10 years ago still had a slightly raised all-cause mortality risk, and this was even higher if they had smoked for 20 years or more.
After 10 years of not smoking, however, the greater all-cause mortality risk was no longer significant in any of the groups monitored (smoking duration, number of cigarettes/day). Concrete evidence of the link between all-cause mortality and the length of time since a person stopped smoking also emerged from the large cohort in the American Nurses’ Health Study.
The relative risk for all-cause mortality in women who stopped smoking less than 5 years ago remained high (RR = 1.96, 1.47-2.67), then decreased over time. After 10 years, it was no longer significant (RR = 1.11, 0.92-1.35).
Macro- and microangiopathic risks
Smoking cessation also has a real benefit in terms of the increased macro- and microangiopathic risks. In type 2 diabetes, a study found an increased relative risk for macro- and microalbuminuria of 1.86 (95% confidence interval, 1.37-2.52) in former smokers, compared with an increased relative risk of 2.61 (95% CI, 1.86-2.64) in current smokers.
In type 1 diabetes, the cumulative risk for microalbuminuria in former smokers was 15.1% vs. 18.9% in smokers and 10% in nonsmokers.
A 2019 meta-analysis of prospective cohort studies determined that smoking is an independent risk factor for diabetic nephropathy, especially in patients with type 1 diabetes.
Yet, most of the data for this condition come from subjects with type 2 diabetes. One publication estimated its prevalence after a 1-year follow-up of the smoking cessation program as 10.9% in former smokers and 15% in those who continued smoking.
In regard to macroangiopathy in the context of type 2 diabetes, the aforementioned 2019 meta-analysis focused on coronary artery disease, cerebrovascular accident (CVA), cardiovascular mortality, and myocardial infarction (MI). It found that smokers face an increased risk for all these outcomes.
The relative risks wavered between 1.53 and 1.66 and decreased after smoking cessation. For coronary artery disease and MI, they became insignificant. There was still a risk for CVA (RR = 1.34; 1.07-1.67) and fatal cardiovascular events (RR = 1.19; 0.02-1.39).
The data are slightly more heterogeneous for type 1 diabetes, where, despite smoking cessation, the increased risk for heart failure and CVA persists in men, yet the same risk for coronary heart disease and CVA drops in women.
Risk for weight gain
Dr. Rouland tried to reassure patients about the risk for gaining weight. “Weight gain is not inevitable. There is a risk for this, but it’s temporary. And, even with some weight gain, the cardiovascular benefits are still indisputable.”
A study carried out in 2013 focused specifically on this point, with an average post-cessation weight gain of 3.8 kg (8.4 lb) seen in diabetic individuals in the first 4 years after stopping smoking and of 0.1 kg (0.2 lb) thereafter. A time-based effect was observed with regulation of excess weight post-cessation over time, as seen in the general population (3 kg [6.6 lb] on average in nondiabetic individuals).
Weight gain tends to occur mainly in the immediate post-cessation period, essentially in the first 3 months, and there is a large variation in weight change. Some people gain a lot (from 5 to 10 kg [11 to 22 lb], or even more than 10 kg); others lose weight (20% of diabetic former smokers in the first month, 7% after 12 months), and 25% gain less than 5 kg (11 lb).
Blood glucose imbalance
“A risk for blood glucose imbalance has been reported after smoking cessation, although this is very slight and only temporary,” said Dr. Rouland.
A British retrospective study examined this question, focusing on glycated hemoglobin in patients with type 2 diabetes. Hemoglobin A1c increased by 0.21% (95% CI, 0.17-0.25; P < .001) within the first year after quitting. A1c decreased as abstinence continued and became comparable to that of continual smokers after 3 years. This increase in A1c was not mediated by weight change.
Another study published in 2018 on the topic of type 2 diabetes also reported on the risk for poor glycemic control (defined as A1c > 7%) persisting for 10 years after smoking cessation (odds ratio, 1.23; 95% CI, 1.06-1.42). Thereafter, between 10 and 19 years post-cessation, the OR decreased to 0.97 (95% CI, 0.80-1.19, NS). Beyond 20 years post-cessation, the OR was 1.14 (95% CI, 0.89-1.44, NS) and was therefore no longer significant.
Regardless, “the risk for poor glycemic control is lower in quitters than in active smokers,” said Dr. Rouland.
Quitting and diabetes risk
Will a smoker’s increased risk for diabetes drop when he or she stops smoking? “This is essentially what happens,” Dr. Rouland confirmed, “and his or her increased risk for metabolic syndrome also drops. One meta-analysis revealed a time-based effect.
“Patients who had stopped smoking less than 5 years previously had an increased relative risk for type 2 diabetes, and this risk dropped to 1.11 after more than 10 years of not smoking. Moreover, this relative risk for type 2 diabetes remained lower than that of active smokers, at between 1.19 and 1.60, depending on tobacco use.”
In regard to the risk for metabolic syndrome, those who quit smoking seem to have an increased risk of 10%, compared with nonsmokers (RR = 1.10, 1.08-1.11; P < .001). “But yet again, this increased risk is much lower than that of active smokers, whose risk is between 37% (less than 20 cigarettes/day) and 71% (more than 20 cigarettes/day).”
Women with diabetes
“The benefits of quitting appear identical, regardless of the sex of the diabetic person,” said Dr. Rouland. “As in the general population, weight gain after smoking cessation is greater in women. Furthermore, while smoking increases the risk for gestational diabetes (RR, 1.4-1.9) and for the use of insulin in this context, stopping smoking reduces these risks.
“Moreover, smoking during pregnancy not only increases the risk for pregnancy-related complications (early miscarriage, ectopic pregnancy, birth defects, placental abruption, premature birth, intrauterine fetal demise, cesarean birth, low birth weight), but it also increases the risk of type 2 diabetes in the newborn. The risk to the newborn is said to be around 34% in cases in which the mother smokes during pregnancy and 22% in cases in which the mother is a passive smoker, thereby justifying the use of measures to help the mother’s family members to stop smoking.”
Dr. Rouland reports no relevant financial relationships.
This article was translated from the Medscape French edition. A version appeared on Medscape.com.
MONTPELLIER, FRANCE – The first expert consensus on smoking and diabetes, coauthored by the Francophone Diabetes Society (SFD) and the French Society for the Study of Nicotine Addiction (SFT), was presented at the SFD’s annual conference.
Alexia Rouland, MD, an endocrinologist at Dijon Bourgogne University Hospital, Dijon, France, took the conference as an opportunity to list the many benefits of smoking cessation for patients with diabetes, despite the “slight and temporary” risk for blood sugar imbalance.
Societies target smoking
Diabetes societies around Europe have set their sights on the topic of smoking. Indeed, the guidelines published in 2019 by the European Association for the Study of Diabetes and the European Society of Cardiology state that “smoking cessation is obligatory for all prediabetic and diabetic patients” (class I, level A).
This year, the France-based SFD and SFT dedicated an expert consensus to the major problem of smoking in patients with diabetes. The aim was to provide health care professionals with convincing, well-supported arguments in favor of smoking cessation in their patients with type 1 and type 2 diabetes.
“Before anything else, diabetic patients need to be made aware of the risks of smoking,” said Dr. Rouland. “It’s not just about the fear factor, though. It’s also about providing a positive incentive – they need to be told about the ways they’ll benefit from quitting smoking. For example, you have all-cause mortality, macro- and microangiopathic complications, and so on.”
Duration of abstinence
“Diabetic patients who have stopped smoking have a relative risk for all-cause mortality of 1.28 (1.09-1.51), which is less than what you see in active smokers (relative risk = 1.58; 1.42-1.77), but still above that of nonsmokers,” said Dr. Rouland.
A previous study revealed that although the risk does indeed go down after stopping smoking, it is linked to how long ago the person stopped. Patients who stopped smoking less than 10 years ago still had a slightly raised all-cause mortality risk, and this was even higher if they had smoked for 20 years or more.
After 10 years of not smoking, however, the greater all-cause mortality risk was no longer significant in any of the groups monitored (smoking duration, number of cigarettes/day). Concrete evidence of the link between all-cause mortality and the length of time since a person stopped smoking also emerged from the large cohort in the American Nurses’ Health Study.
The relative risk for all-cause mortality in women who stopped smoking less than 5 years ago remained high (RR = 1.96, 1.47-2.67), then decreased over time. After 10 years, it was no longer significant (RR = 1.11, 0.92-1.35).
Macro- and microangiopathic risks
Smoking cessation also has a real benefit in terms of the increased macro- and microangiopathic risks. In type 2 diabetes, a study found an increased relative risk for macro- and microalbuminuria of 1.86 (95% confidence interval, 1.37-2.52) in former smokers, compared with an increased relative risk of 2.61 (95% CI, 1.86-2.64) in current smokers.
In type 1 diabetes, the cumulative risk for microalbuminuria in former smokers was 15.1% vs. 18.9% in smokers and 10% in nonsmokers.
A 2019 meta-analysis of prospective cohort studies determined that smoking is an independent risk factor for diabetic nephropathy, especially in patients with type 1 diabetes.
Yet, most of the data for this condition come from subjects with type 2 diabetes. One publication estimated its prevalence after a 1-year follow-up of the smoking cessation program as 10.9% in former smokers and 15% in those who continued smoking.
In regard to macroangiopathy in the context of type 2 diabetes, the aforementioned 2019 meta-analysis focused on coronary artery disease, cerebrovascular accident (CVA), cardiovascular mortality, and myocardial infarction (MI). It found that smokers face an increased risk for all these outcomes.
The relative risks wavered between 1.53 and 1.66 and decreased after smoking cessation. For coronary artery disease and MI, they became insignificant. There was still a risk for CVA (RR = 1.34; 1.07-1.67) and fatal cardiovascular events (RR = 1.19; 0.02-1.39).
The data are slightly more heterogeneous for type 1 diabetes, where, despite smoking cessation, the increased risk for heart failure and CVA persists in men, yet the same risk for coronary heart disease and CVA drops in women.
Risk for weight gain
Dr. Rouland tried to reassure patients about the risk for gaining weight. “Weight gain is not inevitable. There is a risk for this, but it’s temporary. And, even with some weight gain, the cardiovascular benefits are still indisputable.”
A study carried out in 2013 focused specifically on this point, with an average post-cessation weight gain of 3.8 kg (8.4 lb) seen in diabetic individuals in the first 4 years after stopping smoking and of 0.1 kg (0.2 lb) thereafter. A time-based effect was observed with regulation of excess weight post-cessation over time, as seen in the general population (3 kg [6.6 lb] on average in nondiabetic individuals).
Weight gain tends to occur mainly in the immediate post-cessation period, essentially in the first 3 months, and there is a large variation in weight change. Some people gain a lot (from 5 to 10 kg [11 to 22 lb], or even more than 10 kg); others lose weight (20% of diabetic former smokers in the first month, 7% after 12 months), and 25% gain less than 5 kg (11 lb).
Blood glucose imbalance
“A risk for blood glucose imbalance has been reported after smoking cessation, although this is very slight and only temporary,” said Dr. Rouland.
A British retrospective study examined this question, focusing on glycated hemoglobin in patients with type 2 diabetes. Hemoglobin A1c increased by 0.21% (95% CI, 0.17-0.25; P < .001) within the first year after quitting. A1c decreased as abstinence continued and became comparable to that of continual smokers after 3 years. This increase in A1c was not mediated by weight change.
Another study published in 2018 on the topic of type 2 diabetes also reported on the risk for poor glycemic control (defined as A1c > 7%) persisting for 10 years after smoking cessation (odds ratio, 1.23; 95% CI, 1.06-1.42). Thereafter, between 10 and 19 years post-cessation, the OR decreased to 0.97 (95% CI, 0.80-1.19, NS). Beyond 20 years post-cessation, the OR was 1.14 (95% CI, 0.89-1.44, NS) and was therefore no longer significant.
Regardless, “the risk for poor glycemic control is lower in quitters than in active smokers,” said Dr. Rouland.
Quitting and diabetes risk
Will a smoker’s increased risk for diabetes drop when he or she stops smoking? “This is essentially what happens,” Dr. Rouland confirmed, “and his or her increased risk for metabolic syndrome also drops. One meta-analysis revealed a time-based effect.
“Patients who had stopped smoking less than 5 years previously had an increased relative risk for type 2 diabetes, and this risk dropped to 1.11 after more than 10 years of not smoking. Moreover, this relative risk for type 2 diabetes remained lower than that of active smokers, at between 1.19 and 1.60, depending on tobacco use.”
In regard to the risk for metabolic syndrome, those who quit smoking seem to have an increased risk of 10%, compared with nonsmokers (RR = 1.10, 1.08-1.11; P < .001). “But yet again, this increased risk is much lower than that of active smokers, whose risk is between 37% (less than 20 cigarettes/day) and 71% (more than 20 cigarettes/day).”
Women with diabetes
“The benefits of quitting appear identical, regardless of the sex of the diabetic person,” said Dr. Rouland. “As in the general population, weight gain after smoking cessation is greater in women. Furthermore, while smoking increases the risk for gestational diabetes (RR, 1.4-1.9) and for the use of insulin in this context, stopping smoking reduces these risks.
“Moreover, smoking during pregnancy not only increases the risk for pregnancy-related complications (early miscarriage, ectopic pregnancy, birth defects, placental abruption, premature birth, intrauterine fetal demise, cesarean birth, low birth weight), but it also increases the risk of type 2 diabetes in the newborn. The risk to the newborn is said to be around 34% in cases in which the mother smokes during pregnancy and 22% in cases in which the mother is a passive smoker, thereby justifying the use of measures to help the mother’s family members to stop smoking.”
Dr. Rouland reports no relevant financial relationships.
This article was translated from the Medscape French edition. A version appeared on Medscape.com.
MONTPELLIER, FRANCE – The first expert consensus on smoking and diabetes, coauthored by the Francophone Diabetes Society (SFD) and the French Society for the Study of Nicotine Addiction (SFT), was presented at the SFD’s annual conference.
Alexia Rouland, MD, an endocrinologist at Dijon Bourgogne University Hospital, Dijon, France, took the conference as an opportunity to list the many benefits of smoking cessation for patients with diabetes, despite the “slight and temporary” risk for blood sugar imbalance.
Societies target smoking
Diabetes societies around Europe have set their sights on the topic of smoking. Indeed, the guidelines published in 2019 by the European Association for the Study of Diabetes and the European Society of Cardiology state that “smoking cessation is obligatory for all prediabetic and diabetic patients” (class I, level A).
This year, the France-based SFD and SFT dedicated an expert consensus to the major problem of smoking in patients with diabetes. The aim was to provide health care professionals with convincing, well-supported arguments in favor of smoking cessation in their patients with type 1 and type 2 diabetes.
“Before anything else, diabetic patients need to be made aware of the risks of smoking,” said Dr. Rouland. “It’s not just about the fear factor, though. It’s also about providing a positive incentive – they need to be told about the ways they’ll benefit from quitting smoking. For example, you have all-cause mortality, macro- and microangiopathic complications, and so on.”
Duration of abstinence
“Diabetic patients who have stopped smoking have a relative risk for all-cause mortality of 1.28 (1.09-1.51), which is less than what you see in active smokers (relative risk = 1.58; 1.42-1.77), but still above that of nonsmokers,” said Dr. Rouland.
A previous study revealed that although the risk does indeed go down after stopping smoking, it is linked to how long ago the person stopped. Patients who stopped smoking less than 10 years ago still had a slightly raised all-cause mortality risk, and this was even higher if they had smoked for 20 years or more.
After 10 years of not smoking, however, the greater all-cause mortality risk was no longer significant in any of the groups monitored (smoking duration, number of cigarettes/day). Concrete evidence of the link between all-cause mortality and the length of time since a person stopped smoking also emerged from the large cohort in the American Nurses’ Health Study.
The relative risk for all-cause mortality in women who stopped smoking less than 5 years ago remained high (RR = 1.96, 1.47-2.67), then decreased over time. After 10 years, it was no longer significant (RR = 1.11, 0.92-1.35).
Macro- and microangiopathic risks
Smoking cessation also has a real benefit in terms of the increased macro- and microangiopathic risks. In type 2 diabetes, a study found an increased relative risk for macro- and microalbuminuria of 1.86 (95% confidence interval, 1.37-2.52) in former smokers, compared with an increased relative risk of 2.61 (95% CI, 1.86-2.64) in current smokers.
In type 1 diabetes, the cumulative risk for microalbuminuria in former smokers was 15.1% vs. 18.9% in smokers and 10% in nonsmokers.
A 2019 meta-analysis of prospective cohort studies determined that smoking is an independent risk factor for diabetic nephropathy, especially in patients with type 1 diabetes.
Yet, most of the data for this condition come from subjects with type 2 diabetes. One publication estimated its prevalence after a 1-year follow-up of the smoking cessation program as 10.9% in former smokers and 15% in those who continued smoking.
In regard to macroangiopathy in the context of type 2 diabetes, the aforementioned 2019 meta-analysis focused on coronary artery disease, cerebrovascular accident (CVA), cardiovascular mortality, and myocardial infarction (MI). It found that smokers face an increased risk for all these outcomes.
The relative risks wavered between 1.53 and 1.66 and decreased after smoking cessation. For coronary artery disease and MI, they became insignificant. There was still a risk for CVA (RR = 1.34; 1.07-1.67) and fatal cardiovascular events (RR = 1.19; 0.02-1.39).
The data are slightly more heterogeneous for type 1 diabetes, where, despite smoking cessation, the increased risk for heart failure and CVA persists in men, yet the same risk for coronary heart disease and CVA drops in women.
Risk for weight gain
Dr. Rouland tried to reassure patients about the risk for gaining weight. “Weight gain is not inevitable. There is a risk for this, but it’s temporary. And, even with some weight gain, the cardiovascular benefits are still indisputable.”
A study carried out in 2013 focused specifically on this point, with an average post-cessation weight gain of 3.8 kg (8.4 lb) seen in diabetic individuals in the first 4 years after stopping smoking and of 0.1 kg (0.2 lb) thereafter. A time-based effect was observed with regulation of excess weight post-cessation over time, as seen in the general population (3 kg [6.6 lb] on average in nondiabetic individuals).
Weight gain tends to occur mainly in the immediate post-cessation period, essentially in the first 3 months, and there is a large variation in weight change. Some people gain a lot (from 5 to 10 kg [11 to 22 lb], or even more than 10 kg); others lose weight (20% of diabetic former smokers in the first month, 7% after 12 months), and 25% gain less than 5 kg (11 lb).
Blood glucose imbalance
“A risk for blood glucose imbalance has been reported after smoking cessation, although this is very slight and only temporary,” said Dr. Rouland.
A British retrospective study examined this question, focusing on glycated hemoglobin in patients with type 2 diabetes. Hemoglobin A1c increased by 0.21% (95% CI, 0.17-0.25; P < .001) within the first year after quitting. A1c decreased as abstinence continued and became comparable to that of continual smokers after 3 years. This increase in A1c was not mediated by weight change.
Another study published in 2018 on the topic of type 2 diabetes also reported on the risk for poor glycemic control (defined as A1c > 7%) persisting for 10 years after smoking cessation (odds ratio, 1.23; 95% CI, 1.06-1.42). Thereafter, between 10 and 19 years post-cessation, the OR decreased to 0.97 (95% CI, 0.80-1.19, NS). Beyond 20 years post-cessation, the OR was 1.14 (95% CI, 0.89-1.44, NS) and was therefore no longer significant.
Regardless, “the risk for poor glycemic control is lower in quitters than in active smokers,” said Dr. Rouland.
Quitting and diabetes risk
Will a smoker’s increased risk for diabetes drop when he or she stops smoking? “This is essentially what happens,” Dr. Rouland confirmed, “and his or her increased risk for metabolic syndrome also drops. One meta-analysis revealed a time-based effect.
“Patients who had stopped smoking less than 5 years previously had an increased relative risk for type 2 diabetes, and this risk dropped to 1.11 after more than 10 years of not smoking. Moreover, this relative risk for type 2 diabetes remained lower than that of active smokers, at between 1.19 and 1.60, depending on tobacco use.”
In regard to the risk for metabolic syndrome, those who quit smoking seem to have an increased risk of 10%, compared with nonsmokers (RR = 1.10, 1.08-1.11; P < .001). “But yet again, this increased risk is much lower than that of active smokers, whose risk is between 37% (less than 20 cigarettes/day) and 71% (more than 20 cigarettes/day).”
Women with diabetes
“The benefits of quitting appear identical, regardless of the sex of the diabetic person,” said Dr. Rouland. “As in the general population, weight gain after smoking cessation is greater in women. Furthermore, while smoking increases the risk for gestational diabetes (RR, 1.4-1.9) and for the use of insulin in this context, stopping smoking reduces these risks.
“Moreover, smoking during pregnancy not only increases the risk for pregnancy-related complications (early miscarriage, ectopic pregnancy, birth defects, placental abruption, premature birth, intrauterine fetal demise, cesarean birth, low birth weight), but it also increases the risk of type 2 diabetes in the newborn. The risk to the newborn is said to be around 34% in cases in which the mother smokes during pregnancy and 22% in cases in which the mother is a passive smoker, thereby justifying the use of measures to help the mother’s family members to stop smoking.”
Dr. Rouland reports no relevant financial relationships.
This article was translated from the Medscape French edition. A version appeared on Medscape.com.
Should youth with type 1 diabetes use closed-loop systems?
Would closed-loop systems be a good option for young patients with type 1 diabetes?
International and French recommendations on closed-loop systems state that the use of an “artificial pancreas” should be reserved for adults who are fully engaged with their treatment. This means that young patients, especially adolescents, who are less likely to comply with treatment and are more likely to experience suboptimal blood glucose control, are often excluded from the use of such systems for managing their diabetes.
Several recent studies seem to call this approach into question.
One such study, which was presented at a Francophone Diabetes Society conference and was published in Nature Communications, showed that adolescents with poorly controlled diabetes who were equipped with closed-loop systems gained IQ points and reasoning capacity and experienced a reduction in edematous tissue in the brain cortex. Furthermore, with the closed-loop system, patients spent 13% more time in a target range, and there was a significant reduction in time spent in hyperglycemia.
In the same vein, a small prospective study published in Diabetes Care showed that the closed-loop system with the Minimed 780G pump improved glycemic control for 20 young patients with type 1 diabetes aged 13-25 years whose diabetes was poorly controlled (hemoglobin A1c ≥ 8.5%). At the end of the 3-month study period, the average A1c had decreased from 10.5% (±2.1%) to 7.6% (±1.1%), an average decrease of 2.9%. The time spent in target A1c, which was set from 0.70 g/L to 1.80 g/L, was increased by almost 40%.
With respect to very young children, a study published in The New England Journal of Medicine also showed a favorable risk-benefit ratio for closed-loop systems. The trial, which enrolled 102 children aged 2 years to less than 6 years who had type 1 diabetes, showed that the amount of time that the glucose level was within the target range during the 13-week study period was higher (+3 hours) for those who had been randomly assigned to receive the hybrid closed-loop system (n = 68) than for those who had received the standard treatment (n = 34), either with an insulin pump or multiple daily injections or a Dexcom G6 continuous glucose monitoring device.
A previous study carried out by the Paris Public Hospital System had already shown that the French Diabeloop system could reduce episodes of hypoglycemia and achieve good glycemic control for prepubescent children (n = 21; aged 6-12 years) with type 1 diabetes in real-life conditions.
Eric Renard, MD, PhD, head of the department of endocrinology and diabetes at Lapeyronie Hospital in Montpellier, France, was not surprised at the findings from the study, especially in adolescents with poorly controlled diabetes.
“We have already seen studies in which those patients who had the most poorly controlled diabetes at the start were the ones who improved the most with the closed-loop system, by at least 20% in terms of time in target. These findings resonate with what I see in my clinic,” said Dr. Renard in an interview.
“In my experience, these young adolescents, who neglected their diabetes when they had no devices to help control it, when they had to inject themselves, et cetera ... well, they’re just not the same people when they’re put on a closed-loop system,” he added. “They rise to the challenge, and for the first time, they succeed without making a huge effort, since the algorithm does what they weren’t doing. It’s astonishing to see near-total engagement in these young people when explaining the technology to them and saying, ‘Let’s give it a go.’ These are the very same youngsters who didn’t want to hear about their diabetes in the past. They are delighted and once again involved in managing their condition.”
That’s why Dr. Renard recommends keeping an open mind when considering treatment options for young patients with poorly controlled type 1 diabetes.
“When young people have very poorly controlled diabetes, they risk having cardiovascular complications and damaging their retinas and kidneys,” he said. “If we can get them from 25% to 45% time in target, even if that hasn’t been easy to achieve, this will help save their blood vessels! The only thing we have to be careful of is that we don’t set up a closed-loop system in someone who doesn’t want one. But, if it can manage to spark the interest of a young patient, in most cases, it’s beneficial.”
This article was translated from the Medscape French edition. A version appeared on Medscape.com.
Would closed-loop systems be a good option for young patients with type 1 diabetes?
International and French recommendations on closed-loop systems state that the use of an “artificial pancreas” should be reserved for adults who are fully engaged with their treatment. This means that young patients, especially adolescents, who are less likely to comply with treatment and are more likely to experience suboptimal blood glucose control, are often excluded from the use of such systems for managing their diabetes.
Several recent studies seem to call this approach into question.
One such study, which was presented at a Francophone Diabetes Society conference and was published in Nature Communications, showed that adolescents with poorly controlled diabetes who were equipped with closed-loop systems gained IQ points and reasoning capacity and experienced a reduction in edematous tissue in the brain cortex. Furthermore, with the closed-loop system, patients spent 13% more time in a target range, and there was a significant reduction in time spent in hyperglycemia.
In the same vein, a small prospective study published in Diabetes Care showed that the closed-loop system with the Minimed 780G pump improved glycemic control for 20 young patients with type 1 diabetes aged 13-25 years whose diabetes was poorly controlled (hemoglobin A1c ≥ 8.5%). At the end of the 3-month study period, the average A1c had decreased from 10.5% (±2.1%) to 7.6% (±1.1%), an average decrease of 2.9%. The time spent in target A1c, which was set from 0.70 g/L to 1.80 g/L, was increased by almost 40%.
With respect to very young children, a study published in The New England Journal of Medicine also showed a favorable risk-benefit ratio for closed-loop systems. The trial, which enrolled 102 children aged 2 years to less than 6 years who had type 1 diabetes, showed that the amount of time that the glucose level was within the target range during the 13-week study period was higher (+3 hours) for those who had been randomly assigned to receive the hybrid closed-loop system (n = 68) than for those who had received the standard treatment (n = 34), either with an insulin pump or multiple daily injections or a Dexcom G6 continuous glucose monitoring device.
A previous study carried out by the Paris Public Hospital System had already shown that the French Diabeloop system could reduce episodes of hypoglycemia and achieve good glycemic control for prepubescent children (n = 21; aged 6-12 years) with type 1 diabetes in real-life conditions.
Eric Renard, MD, PhD, head of the department of endocrinology and diabetes at Lapeyronie Hospital in Montpellier, France, was not surprised at the findings from the study, especially in adolescents with poorly controlled diabetes.
“We have already seen studies in which those patients who had the most poorly controlled diabetes at the start were the ones who improved the most with the closed-loop system, by at least 20% in terms of time in target. These findings resonate with what I see in my clinic,” said Dr. Renard in an interview.
“In my experience, these young adolescents, who neglected their diabetes when they had no devices to help control it, when they had to inject themselves, et cetera ... well, they’re just not the same people when they’re put on a closed-loop system,” he added. “They rise to the challenge, and for the first time, they succeed without making a huge effort, since the algorithm does what they weren’t doing. It’s astonishing to see near-total engagement in these young people when explaining the technology to them and saying, ‘Let’s give it a go.’ These are the very same youngsters who didn’t want to hear about their diabetes in the past. They are delighted and once again involved in managing their condition.”
That’s why Dr. Renard recommends keeping an open mind when considering treatment options for young patients with poorly controlled type 1 diabetes.
“When young people have very poorly controlled diabetes, they risk having cardiovascular complications and damaging their retinas and kidneys,” he said. “If we can get them from 25% to 45% time in target, even if that hasn’t been easy to achieve, this will help save their blood vessels! The only thing we have to be careful of is that we don’t set up a closed-loop system in someone who doesn’t want one. But, if it can manage to spark the interest of a young patient, in most cases, it’s beneficial.”
This article was translated from the Medscape French edition. A version appeared on Medscape.com.
Would closed-loop systems be a good option for young patients with type 1 diabetes?
International and French recommendations on closed-loop systems state that the use of an “artificial pancreas” should be reserved for adults who are fully engaged with their treatment. This means that young patients, especially adolescents, who are less likely to comply with treatment and are more likely to experience suboptimal blood glucose control, are often excluded from the use of such systems for managing their diabetes.
Several recent studies seem to call this approach into question.
One such study, which was presented at a Francophone Diabetes Society conference and was published in Nature Communications, showed that adolescents with poorly controlled diabetes who were equipped with closed-loop systems gained IQ points and reasoning capacity and experienced a reduction in edematous tissue in the brain cortex. Furthermore, with the closed-loop system, patients spent 13% more time in a target range, and there was a significant reduction in time spent in hyperglycemia.
In the same vein, a small prospective study published in Diabetes Care showed that the closed-loop system with the Minimed 780G pump improved glycemic control for 20 young patients with type 1 diabetes aged 13-25 years whose diabetes was poorly controlled (hemoglobin A1c ≥ 8.5%). At the end of the 3-month study period, the average A1c had decreased from 10.5% (±2.1%) to 7.6% (±1.1%), an average decrease of 2.9%. The time spent in target A1c, which was set from 0.70 g/L to 1.80 g/L, was increased by almost 40%.
With respect to very young children, a study published in The New England Journal of Medicine also showed a favorable risk-benefit ratio for closed-loop systems. The trial, which enrolled 102 children aged 2 years to less than 6 years who had type 1 diabetes, showed that the amount of time that the glucose level was within the target range during the 13-week study period was higher (+3 hours) for those who had been randomly assigned to receive the hybrid closed-loop system (n = 68) than for those who had received the standard treatment (n = 34), either with an insulin pump or multiple daily injections or a Dexcom G6 continuous glucose monitoring device.
A previous study carried out by the Paris Public Hospital System had already shown that the French Diabeloop system could reduce episodes of hypoglycemia and achieve good glycemic control for prepubescent children (n = 21; aged 6-12 years) with type 1 diabetes in real-life conditions.
Eric Renard, MD, PhD, head of the department of endocrinology and diabetes at Lapeyronie Hospital in Montpellier, France, was not surprised at the findings from the study, especially in adolescents with poorly controlled diabetes.
“We have already seen studies in which those patients who had the most poorly controlled diabetes at the start were the ones who improved the most with the closed-loop system, by at least 20% in terms of time in target. These findings resonate with what I see in my clinic,” said Dr. Renard in an interview.
“In my experience, these young adolescents, who neglected their diabetes when they had no devices to help control it, when they had to inject themselves, et cetera ... well, they’re just not the same people when they’re put on a closed-loop system,” he added. “They rise to the challenge, and for the first time, they succeed without making a huge effort, since the algorithm does what they weren’t doing. It’s astonishing to see near-total engagement in these young people when explaining the technology to them and saying, ‘Let’s give it a go.’ These are the very same youngsters who didn’t want to hear about their diabetes in the past. They are delighted and once again involved in managing their condition.”
That’s why Dr. Renard recommends keeping an open mind when considering treatment options for young patients with poorly controlled type 1 diabetes.
“When young people have very poorly controlled diabetes, they risk having cardiovascular complications and damaging their retinas and kidneys,” he said. “If we can get them from 25% to 45% time in target, even if that hasn’t been easy to achieve, this will help save their blood vessels! The only thing we have to be careful of is that we don’t set up a closed-loop system in someone who doesn’t want one. But, if it can manage to spark the interest of a young patient, in most cases, it’s beneficial.”
This article was translated from the Medscape French edition. A version appeared on Medscape.com.
Obesity drugs overpriced, change needed to tackle issue
The lowest available national prices of drugs to treat obesity are up to 20 times higher than the estimated cost of profitable generic versions of the same agents, according to a new analysis.
The findings by Jacob Levi, MBBS, and colleagues were published in Obesity.
“Our study highlights the inequality in pricing that exists for effective antiobesity medications, which are largely unaffordable in most countries,” Dr. Levi, from Royal Free Hospital NHS Trust, London, said in a press release.
“We show that these drugs can actually be produced and sold profitably for low prices,” he summarized. “A public health approach that prioritizes improving access to medications should be adopted, instead of allowing companies to maximize profits,” Dr. Levi urged.
Dr. Levi and colleagues studied the oral agents orlistat, naltrexone/bupropion, topiramate/phentermine, and semaglutide, and subcutaneous liraglutide, semaglutide, and tirzepatide (all approved by the U.S. Food and Drug Administration to treat obesity, except for oral semaglutide and subcutaneous tirzepatide, which are not yet approved to treat obesity in the absence of type 2 diabetes).
“Worldwide, more people are dying from diabetes and clinical obesity than HIV, tuberculosis, and malaria combined now,” senior author Andrew Hill, MD, department of pharmacology and therapeutics, University of Liverpool, England, pointed out.
We need to repeat the low-cost success story with obesity drugs
“Millions of lives have been saved by treating infectious diseases at low cost in poor countries,” Dr. Hill continued. “Now we need to repeat this medical success story, with mass treatment of diabetes and clinical obesity at low prices.”
However, in an accompanying editorial, Eric A. Finkelstein, MD, and Junxing Chay, PhD, Duke-NUS Medical School, Singapore, maintain that “It would be great if everyone had affordable access to all medicines that might improve their health. Yet that is simply not possible, nor will it ever be.”
“What is truly needed is a better way to ration the health care dollars currently available in efforts to maximize population health. That is the challenge ahead not just for [antiobesity medications] but for all treatments,” they say.
“Greater use of cost-effectiveness analysis and direct negotiations, while maintaining the patent system, represents an appropriate approach for allocating scarce health care resources in the United States and beyond,” they continue.
Lowest current patented drug prices vs. estimated generic drug prices
New medications for obesity were highly effective in recent clinical trials, but high prices limit the ability of patients to get these medications, Dr. Levi and colleagues write.
They analyzed prices for obesity drugs in 16 low-, middle-, and high-income countries: Australia, Bangladesh, China, France, Germany, India, Kenya, Morocco, Norway, Peru, Pakistan, South Africa, Turkey, the United Kingdom, the United States, and Vietnam.
The researchers assessed the price of a 30-day supply of each of the studied branded drugs based on the lowest available price (in 2021 U.S. dollars) from multiple online national price databases.
Then they calculated the estimated minimum price of a 30-day supply of a potential generic version of these drugs, which included the cost of the active medicinal ingredients, the excipients (nonactive ingredients), the prefilled injectable device plus needles (for subcutaneous drugs), transportation, 10% profit, and 27% tax on profit.
The national prices of the branded medications for obesity were significantly higher than the estimated minimum prices of potential generic drugs (see Table).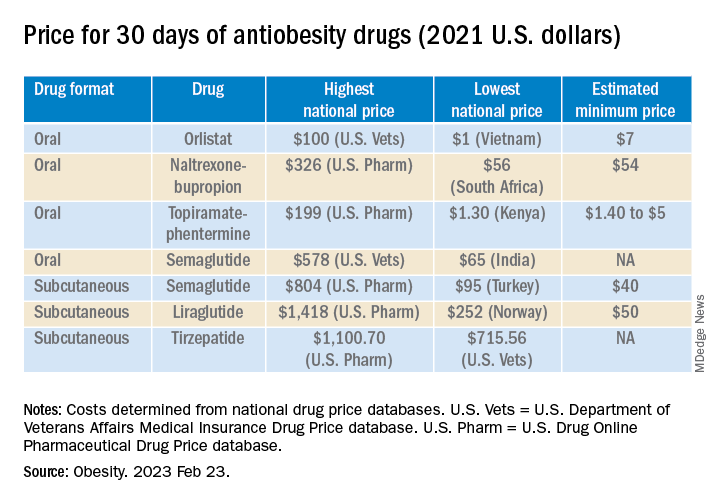
The highest national price for a branded oral drug for obesity vs. the estimated minimum price for a potential generic version was $100 vs. $7 for orlistat, $199 vs. $5 for phentermine/topiramate, and $326 vs. $54 for naltrexone/bupropion, for a 30-day supply.
There was an even greater difference between highest national branded drug price vs. estimated minimum generic drug price for the newer subcutaneously injectable drugs for obesity.
For example, the price of a 30-day course of subcutaneous semaglutide ranged from $804 (United States) to $95 (Turkey), while the estimated minimum potential generic drug price was $40 (which is 20 times lower).
The study was funded by grants from the Make Medicines Affordable/International Treatment Preparedness Coalition and from the National Heart, Lung, and Blood Institute of the National Institutes of Health. Coauthor Francois Venter has reported receiving support from the Bill and Melinda Gates Foundation, U.S. Agency for International Development, Unitaid, SA Medical Research Council, Foundation for Innovative New Diagnostics, the Children’s Investment Fund Foundation, Gilead, ViiV, Mylan, Merck, Adcock Ingram, Aspen, Abbott, Roche, Johnson & Johnson, Sanofi, Virology Education, SA HIV Clinicians Society, and Dira Sengwe. The other authors and Dr. Chay have reported no relevant financial relationships. Dr. Finkelstein has reported receiving support for serving on the WW scientific advisory board and an educational grant unrelated to the present work from Novo Nordisk.
A version of this article first appeared on Medscape.com.
The lowest available national prices of drugs to treat obesity are up to 20 times higher than the estimated cost of profitable generic versions of the same agents, according to a new analysis.
The findings by Jacob Levi, MBBS, and colleagues were published in Obesity.
“Our study highlights the inequality in pricing that exists for effective antiobesity medications, which are largely unaffordable in most countries,” Dr. Levi, from Royal Free Hospital NHS Trust, London, said in a press release.
“We show that these drugs can actually be produced and sold profitably for low prices,” he summarized. “A public health approach that prioritizes improving access to medications should be adopted, instead of allowing companies to maximize profits,” Dr. Levi urged.
Dr. Levi and colleagues studied the oral agents orlistat, naltrexone/bupropion, topiramate/phentermine, and semaglutide, and subcutaneous liraglutide, semaglutide, and tirzepatide (all approved by the U.S. Food and Drug Administration to treat obesity, except for oral semaglutide and subcutaneous tirzepatide, which are not yet approved to treat obesity in the absence of type 2 diabetes).
“Worldwide, more people are dying from diabetes and clinical obesity than HIV, tuberculosis, and malaria combined now,” senior author Andrew Hill, MD, department of pharmacology and therapeutics, University of Liverpool, England, pointed out.
We need to repeat the low-cost success story with obesity drugs
“Millions of lives have been saved by treating infectious diseases at low cost in poor countries,” Dr. Hill continued. “Now we need to repeat this medical success story, with mass treatment of diabetes and clinical obesity at low prices.”
However, in an accompanying editorial, Eric A. Finkelstein, MD, and Junxing Chay, PhD, Duke-NUS Medical School, Singapore, maintain that “It would be great if everyone had affordable access to all medicines that might improve their health. Yet that is simply not possible, nor will it ever be.”
“What is truly needed is a better way to ration the health care dollars currently available in efforts to maximize population health. That is the challenge ahead not just for [antiobesity medications] but for all treatments,” they say.
“Greater use of cost-effectiveness analysis and direct negotiations, while maintaining the patent system, represents an appropriate approach for allocating scarce health care resources in the United States and beyond,” they continue.
Lowest current patented drug prices vs. estimated generic drug prices
New medications for obesity were highly effective in recent clinical trials, but high prices limit the ability of patients to get these medications, Dr. Levi and colleagues write.
They analyzed prices for obesity drugs in 16 low-, middle-, and high-income countries: Australia, Bangladesh, China, France, Germany, India, Kenya, Morocco, Norway, Peru, Pakistan, South Africa, Turkey, the United Kingdom, the United States, and Vietnam.
The researchers assessed the price of a 30-day supply of each of the studied branded drugs based on the lowest available price (in 2021 U.S. dollars) from multiple online national price databases.
Then they calculated the estimated minimum price of a 30-day supply of a potential generic version of these drugs, which included the cost of the active medicinal ingredients, the excipients (nonactive ingredients), the prefilled injectable device plus needles (for subcutaneous drugs), transportation, 10% profit, and 27% tax on profit.
The national prices of the branded medications for obesity were significantly higher than the estimated minimum prices of potential generic drugs (see Table).
The highest national price for a branded oral drug for obesity vs. the estimated minimum price for a potential generic version was $100 vs. $7 for orlistat, $199 vs. $5 for phentermine/topiramate, and $326 vs. $54 for naltrexone/bupropion, for a 30-day supply.
There was an even greater difference between highest national branded drug price vs. estimated minimum generic drug price for the newer subcutaneously injectable drugs for obesity.
For example, the price of a 30-day course of subcutaneous semaglutide ranged from $804 (United States) to $95 (Turkey), while the estimated minimum potential generic drug price was $40 (which is 20 times lower).
The study was funded by grants from the Make Medicines Affordable/International Treatment Preparedness Coalition and from the National Heart, Lung, and Blood Institute of the National Institutes of Health. Coauthor Francois Venter has reported receiving support from the Bill and Melinda Gates Foundation, U.S. Agency for International Development, Unitaid, SA Medical Research Council, Foundation for Innovative New Diagnostics, the Children’s Investment Fund Foundation, Gilead, ViiV, Mylan, Merck, Adcock Ingram, Aspen, Abbott, Roche, Johnson & Johnson, Sanofi, Virology Education, SA HIV Clinicians Society, and Dira Sengwe. The other authors and Dr. Chay have reported no relevant financial relationships. Dr. Finkelstein has reported receiving support for serving on the WW scientific advisory board and an educational grant unrelated to the present work from Novo Nordisk.
A version of this article first appeared on Medscape.com.
The lowest available national prices of drugs to treat obesity are up to 20 times higher than the estimated cost of profitable generic versions of the same agents, according to a new analysis.
The findings by Jacob Levi, MBBS, and colleagues were published in Obesity.
“Our study highlights the inequality in pricing that exists for effective antiobesity medications, which are largely unaffordable in most countries,” Dr. Levi, from Royal Free Hospital NHS Trust, London, said in a press release.
“We show that these drugs can actually be produced and sold profitably for low prices,” he summarized. “A public health approach that prioritizes improving access to medications should be adopted, instead of allowing companies to maximize profits,” Dr. Levi urged.
Dr. Levi and colleagues studied the oral agents orlistat, naltrexone/bupropion, topiramate/phentermine, and semaglutide, and subcutaneous liraglutide, semaglutide, and tirzepatide (all approved by the U.S. Food and Drug Administration to treat obesity, except for oral semaglutide and subcutaneous tirzepatide, which are not yet approved to treat obesity in the absence of type 2 diabetes).
“Worldwide, more people are dying from diabetes and clinical obesity than HIV, tuberculosis, and malaria combined now,” senior author Andrew Hill, MD, department of pharmacology and therapeutics, University of Liverpool, England, pointed out.
We need to repeat the low-cost success story with obesity drugs
“Millions of lives have been saved by treating infectious diseases at low cost in poor countries,” Dr. Hill continued. “Now we need to repeat this medical success story, with mass treatment of diabetes and clinical obesity at low prices.”
However, in an accompanying editorial, Eric A. Finkelstein, MD, and Junxing Chay, PhD, Duke-NUS Medical School, Singapore, maintain that “It would be great if everyone had affordable access to all medicines that might improve their health. Yet that is simply not possible, nor will it ever be.”
“What is truly needed is a better way to ration the health care dollars currently available in efforts to maximize population health. That is the challenge ahead not just for [antiobesity medications] but for all treatments,” they say.
“Greater use of cost-effectiveness analysis and direct negotiations, while maintaining the patent system, represents an appropriate approach for allocating scarce health care resources in the United States and beyond,” they continue.
Lowest current patented drug prices vs. estimated generic drug prices
New medications for obesity were highly effective in recent clinical trials, but high prices limit the ability of patients to get these medications, Dr. Levi and colleagues write.
They analyzed prices for obesity drugs in 16 low-, middle-, and high-income countries: Australia, Bangladesh, China, France, Germany, India, Kenya, Morocco, Norway, Peru, Pakistan, South Africa, Turkey, the United Kingdom, the United States, and Vietnam.
The researchers assessed the price of a 30-day supply of each of the studied branded drugs based on the lowest available price (in 2021 U.S. dollars) from multiple online national price databases.
Then they calculated the estimated minimum price of a 30-day supply of a potential generic version of these drugs, which included the cost of the active medicinal ingredients, the excipients (nonactive ingredients), the prefilled injectable device plus needles (for subcutaneous drugs), transportation, 10% profit, and 27% tax on profit.
The national prices of the branded medications for obesity were significantly higher than the estimated minimum prices of potential generic drugs (see Table).
The highest national price for a branded oral drug for obesity vs. the estimated minimum price for a potential generic version was $100 vs. $7 for orlistat, $199 vs. $5 for phentermine/topiramate, and $326 vs. $54 for naltrexone/bupropion, for a 30-day supply.
There was an even greater difference between highest national branded drug price vs. estimated minimum generic drug price for the newer subcutaneously injectable drugs for obesity.
For example, the price of a 30-day course of subcutaneous semaglutide ranged from $804 (United States) to $95 (Turkey), while the estimated minimum potential generic drug price was $40 (which is 20 times lower).
The study was funded by grants from the Make Medicines Affordable/International Treatment Preparedness Coalition and from the National Heart, Lung, and Blood Institute of the National Institutes of Health. Coauthor Francois Venter has reported receiving support from the Bill and Melinda Gates Foundation, U.S. Agency for International Development, Unitaid, SA Medical Research Council, Foundation for Innovative New Diagnostics, the Children’s Investment Fund Foundation, Gilead, ViiV, Mylan, Merck, Adcock Ingram, Aspen, Abbott, Roche, Johnson & Johnson, Sanofi, Virology Education, SA HIV Clinicians Society, and Dira Sengwe. The other authors and Dr. Chay have reported no relevant financial relationships. Dr. Finkelstein has reported receiving support for serving on the WW scientific advisory board and an educational grant unrelated to the present work from Novo Nordisk.
A version of this article first appeared on Medscape.com.
Tirzepatide scores win in second obesity trial, SURMOUNT-2
The “twincretin” tirzepatide (Mounjaro) has proven successful in SURMOUNT-2, the second pivotal trial for the drug as an antiobesity agent, according to top-line results reported April 27 by tirzepatide’s manufacturer, Lilly, in a press release. The company reveals that tirzepatide achieved both of its primary endpoints in the trial, as well as all its key secondary endpoints.
The findings pave the way for tirzepatide to likely receive Food and Drug Administration approval as a treatment for obesity, perhaps before the end of 2023.
Tirzepatide received FDA approval in May 2022 for the treatment of type 2 diabetes in adults, under the brand name Mounjaro, and some people have already been using it off-label to treat obesity.
Tirzepatide is a dual glucagonlike peptide–1 (GLP-1) agonist and glucose-dependent insulinotropic polypeptide agonist. Several GLP-1 receptor agonists are already approved in the United States, including semaglutide, a once-weekly injection, which is approved as Wegovy for patients with obesity and as Ozempic for treatment of type 2 diabetes.
These agents have been incredibly popular among celebrity influencers, and with use of the #Ozempic hashtag and others on social media, this has led to unprecedented use of these products for weight loss, often among those who do not even have obesity or type 2 diabetes. Subsequently, patients with type 2 diabetes and obesity who need them have often struggled to obtain them, owing to shortages following this phenomenon.
SURMOUNT-2: Weight loss around 15%, less than seen in SURMOUNT-1
SURMOUNT-2 enrolled 938 adults with overweight or obesity and type 2 diabetes and had dual primary endpoints that both focused on weight loss, compared with placebo.
The first completed pivotal trial of tirzepatide for weight loss, SURMOUNT-1, enrolled people with overweight or obesity but no diabetes and had its main results reported in 2022. At the time, the weight loss achieved with tirzepatide, was described as “unprecedented,” with those given the highest dose in that trial (15 mg subcutaneously per week) losing an average of 20%-22% of body weight over 72 weeks, depending on the specific statistical analysis used.
For SURMOUNT-2’s first primary endpoint, 72 weeks of weekly subcutaneous injections with tirzepatide at dosages of 10 mg or 15 mg led to an average weight loss from baseline of 13.4% and 15.7%, respectively, compared with an average loss of 3.3% from baseline in the placebo-treated control arm.
For the second primary endpoint, 81.6% of people on the 10-mg dose and 86.4% on the 15-mg dose achieved at least 5% weight loss from baseline, compared with 30.5% of controls who had at least 5% weight loss from baseline.
In one key secondary endpoint, tirzepatide at dosages of 10 mg or 15 mg weekly produced at least a 15% cut in weight from baseline in 41.4% and 51.8% of participants, respectively, compared with a 2.6% rate of this endpoint in the placebo controls.
So the extent of weight loss seen in in SURMOUNT-2 was somewhat less than was reported in SURMOUNT-1, a finding consistent with many prior studies of incretin-based weight-loss agents, which seem to pack a more potent weight-loss punch in people without type 2 diabetes.
Lilly did not specifically report the treatment effect of tirzepatide on hemoglobin A1c in SURMOUNT-2, only saying that the effect was similar to what had been seen in the series of five SURPASS trials that led to the approval of tirzepatide for type 2 diabetes.
Lilly also reported that the safety profile of tirzepatide in SURMOUNT-2 generally matched what was seen in SURMOUNT-1 as well as in the SURPASS trials. The most common adverse events in SURMOUNT-2 involved gastrointestinal symptoms, such as nausea, diarrhea, and vomiting; these were generally mild to moderate in severity and clustered during the dose-escalation phase at the start of treatment. Treatment discontinuations caused by adverse effects were 3.8% on the 10-mg dosage, 7.4% on the 15-mg dosage, and 3.8% on placebo.
SURMOUNT-2 enrolled patients in the United States, Puerto Rico, and five other countries. All participants also received interventions designed to reduce their calorie intake and increase their physical activity.
More SURMOUNT-2 results at ADA in June
Lilly also announced that researchers would report more complete results from SURMOUNT-2 at the 2023 scientific sessions of the American Diabetes Association, being held in San Diego in late June, and publish the findings in a major medical journal.
Results from two additional phase 3 trials of tirzepatide in people with overweight or obesity, SURMOUNT-3 and SURMOUNT-4, are expected later in 2023.
Lilly started an application to the FDA for an indication for weight loss in October 2022 under a fast track designation by the agency, and the data collected in SURMOUNT-2 are expected to complete this application, which would then be subject to an FDA decision within about 6 months. Lilly said in its April 27 press release that it anticipates an FDA decision on this application may occur before the end of 2023.
SURMOUNT-2 and all of the other tirzepatide trials were sponsored by Lilly.
A version of this article first appeared on Medscape.com.
The “twincretin” tirzepatide (Mounjaro) has proven successful in SURMOUNT-2, the second pivotal trial for the drug as an antiobesity agent, according to top-line results reported April 27 by tirzepatide’s manufacturer, Lilly, in a press release. The company reveals that tirzepatide achieved both of its primary endpoints in the trial, as well as all its key secondary endpoints.
The findings pave the way for tirzepatide to likely receive Food and Drug Administration approval as a treatment for obesity, perhaps before the end of 2023.
Tirzepatide received FDA approval in May 2022 for the treatment of type 2 diabetes in adults, under the brand name Mounjaro, and some people have already been using it off-label to treat obesity.
Tirzepatide is a dual glucagonlike peptide–1 (GLP-1) agonist and glucose-dependent insulinotropic polypeptide agonist. Several GLP-1 receptor agonists are already approved in the United States, including semaglutide, a once-weekly injection, which is approved as Wegovy for patients with obesity and as Ozempic for treatment of type 2 diabetes.
These agents have been incredibly popular among celebrity influencers, and with use of the #Ozempic hashtag and others on social media, this has led to unprecedented use of these products for weight loss, often among those who do not even have obesity or type 2 diabetes. Subsequently, patients with type 2 diabetes and obesity who need them have often struggled to obtain them, owing to shortages following this phenomenon.
SURMOUNT-2: Weight loss around 15%, less than seen in SURMOUNT-1
SURMOUNT-2 enrolled 938 adults with overweight or obesity and type 2 diabetes and had dual primary endpoints that both focused on weight loss, compared with placebo.
The first completed pivotal trial of tirzepatide for weight loss, SURMOUNT-1, enrolled people with overweight or obesity but no diabetes and had its main results reported in 2022. At the time, the weight loss achieved with tirzepatide, was described as “unprecedented,” with those given the highest dose in that trial (15 mg subcutaneously per week) losing an average of 20%-22% of body weight over 72 weeks, depending on the specific statistical analysis used.
For SURMOUNT-2’s first primary endpoint, 72 weeks of weekly subcutaneous injections with tirzepatide at dosages of 10 mg or 15 mg led to an average weight loss from baseline of 13.4% and 15.7%, respectively, compared with an average loss of 3.3% from baseline in the placebo-treated control arm.
For the second primary endpoint, 81.6% of people on the 10-mg dose and 86.4% on the 15-mg dose achieved at least 5% weight loss from baseline, compared with 30.5% of controls who had at least 5% weight loss from baseline.
In one key secondary endpoint, tirzepatide at dosages of 10 mg or 15 mg weekly produced at least a 15% cut in weight from baseline in 41.4% and 51.8% of participants, respectively, compared with a 2.6% rate of this endpoint in the placebo controls.
So the extent of weight loss seen in in SURMOUNT-2 was somewhat less than was reported in SURMOUNT-1, a finding consistent with many prior studies of incretin-based weight-loss agents, which seem to pack a more potent weight-loss punch in people without type 2 diabetes.
Lilly did not specifically report the treatment effect of tirzepatide on hemoglobin A1c in SURMOUNT-2, only saying that the effect was similar to what had been seen in the series of five SURPASS trials that led to the approval of tirzepatide for type 2 diabetes.
Lilly also reported that the safety profile of tirzepatide in SURMOUNT-2 generally matched what was seen in SURMOUNT-1 as well as in the SURPASS trials. The most common adverse events in SURMOUNT-2 involved gastrointestinal symptoms, such as nausea, diarrhea, and vomiting; these were generally mild to moderate in severity and clustered during the dose-escalation phase at the start of treatment. Treatment discontinuations caused by adverse effects were 3.8% on the 10-mg dosage, 7.4% on the 15-mg dosage, and 3.8% on placebo.
SURMOUNT-2 enrolled patients in the United States, Puerto Rico, and five other countries. All participants also received interventions designed to reduce their calorie intake and increase their physical activity.
More SURMOUNT-2 results at ADA in June
Lilly also announced that researchers would report more complete results from SURMOUNT-2 at the 2023 scientific sessions of the American Diabetes Association, being held in San Diego in late June, and publish the findings in a major medical journal.
Results from two additional phase 3 trials of tirzepatide in people with overweight or obesity, SURMOUNT-3 and SURMOUNT-4, are expected later in 2023.
Lilly started an application to the FDA for an indication for weight loss in October 2022 under a fast track designation by the agency, and the data collected in SURMOUNT-2 are expected to complete this application, which would then be subject to an FDA decision within about 6 months. Lilly said in its April 27 press release that it anticipates an FDA decision on this application may occur before the end of 2023.
SURMOUNT-2 and all of the other tirzepatide trials were sponsored by Lilly.
A version of this article first appeared on Medscape.com.
The “twincretin” tirzepatide (Mounjaro) has proven successful in SURMOUNT-2, the second pivotal trial for the drug as an antiobesity agent, according to top-line results reported April 27 by tirzepatide’s manufacturer, Lilly, in a press release. The company reveals that tirzepatide achieved both of its primary endpoints in the trial, as well as all its key secondary endpoints.
The findings pave the way for tirzepatide to likely receive Food and Drug Administration approval as a treatment for obesity, perhaps before the end of 2023.
Tirzepatide received FDA approval in May 2022 for the treatment of type 2 diabetes in adults, under the brand name Mounjaro, and some people have already been using it off-label to treat obesity.
Tirzepatide is a dual glucagonlike peptide–1 (GLP-1) agonist and glucose-dependent insulinotropic polypeptide agonist. Several GLP-1 receptor agonists are already approved in the United States, including semaglutide, a once-weekly injection, which is approved as Wegovy for patients with obesity and as Ozempic for treatment of type 2 diabetes.
These agents have been incredibly popular among celebrity influencers, and with use of the #Ozempic hashtag and others on social media, this has led to unprecedented use of these products for weight loss, often among those who do not even have obesity or type 2 diabetes. Subsequently, patients with type 2 diabetes and obesity who need them have often struggled to obtain them, owing to shortages following this phenomenon.
SURMOUNT-2: Weight loss around 15%, less than seen in SURMOUNT-1
SURMOUNT-2 enrolled 938 adults with overweight or obesity and type 2 diabetes and had dual primary endpoints that both focused on weight loss, compared with placebo.
The first completed pivotal trial of tirzepatide for weight loss, SURMOUNT-1, enrolled people with overweight or obesity but no diabetes and had its main results reported in 2022. At the time, the weight loss achieved with tirzepatide, was described as “unprecedented,” with those given the highest dose in that trial (15 mg subcutaneously per week) losing an average of 20%-22% of body weight over 72 weeks, depending on the specific statistical analysis used.
For SURMOUNT-2’s first primary endpoint, 72 weeks of weekly subcutaneous injections with tirzepatide at dosages of 10 mg or 15 mg led to an average weight loss from baseline of 13.4% and 15.7%, respectively, compared with an average loss of 3.3% from baseline in the placebo-treated control arm.
For the second primary endpoint, 81.6% of people on the 10-mg dose and 86.4% on the 15-mg dose achieved at least 5% weight loss from baseline, compared with 30.5% of controls who had at least 5% weight loss from baseline.
In one key secondary endpoint, tirzepatide at dosages of 10 mg or 15 mg weekly produced at least a 15% cut in weight from baseline in 41.4% and 51.8% of participants, respectively, compared with a 2.6% rate of this endpoint in the placebo controls.
So the extent of weight loss seen in in SURMOUNT-2 was somewhat less than was reported in SURMOUNT-1, a finding consistent with many prior studies of incretin-based weight-loss agents, which seem to pack a more potent weight-loss punch in people without type 2 diabetes.
Lilly did not specifically report the treatment effect of tirzepatide on hemoglobin A1c in SURMOUNT-2, only saying that the effect was similar to what had been seen in the series of five SURPASS trials that led to the approval of tirzepatide for type 2 diabetes.
Lilly also reported that the safety profile of tirzepatide in SURMOUNT-2 generally matched what was seen in SURMOUNT-1 as well as in the SURPASS trials. The most common adverse events in SURMOUNT-2 involved gastrointestinal symptoms, such as nausea, diarrhea, and vomiting; these were generally mild to moderate in severity and clustered during the dose-escalation phase at the start of treatment. Treatment discontinuations caused by adverse effects were 3.8% on the 10-mg dosage, 7.4% on the 15-mg dosage, and 3.8% on placebo.
SURMOUNT-2 enrolled patients in the United States, Puerto Rico, and five other countries. All participants also received interventions designed to reduce their calorie intake and increase their physical activity.
More SURMOUNT-2 results at ADA in June
Lilly also announced that researchers would report more complete results from SURMOUNT-2 at the 2023 scientific sessions of the American Diabetes Association, being held in San Diego in late June, and publish the findings in a major medical journal.
Results from two additional phase 3 trials of tirzepatide in people with overweight or obesity, SURMOUNT-3 and SURMOUNT-4, are expected later in 2023.
Lilly started an application to the FDA for an indication for weight loss in October 2022 under a fast track designation by the agency, and the data collected in SURMOUNT-2 are expected to complete this application, which would then be subject to an FDA decision within about 6 months. Lilly said in its April 27 press release that it anticipates an FDA decision on this application may occur before the end of 2023.
SURMOUNT-2 and all of the other tirzepatide trials were sponsored by Lilly.
A version of this article first appeared on Medscape.com.
FDA okays latest artificial pancreas, the MiniMed 780G
The Food and Drug Administration has approved Medtronic Minimed’s 780G automated insulin delivery system with the Guardian 4 sensor.
The latest so-called artificial pancreas system is approved for people aged 7 years and older who have type 1 diabetes. Medtronic will begin taking preorders for the 780G on May 15, 2023. Users of the current MiniMed 770G will be eligible for no-cost remote software upgrades.
The 780G is currently available in 105 countries. It has been available in Europe since 2020 and in the United Kingdom since 2021. It is the first automated insulin delivery system to automatically administer bolus correction insulin doses every 5 minutes to correct meal-related hyperglycemia.
This so-called meal detection technology doesn’t replace manual premeal boluses but does provide extra insulin if the premeal bolus is skipped or is insufficient.
As with other automated systems, the 780G automatically adjusts basal insulin doses up or down based on glucose levels and trends and shuts off insulin delivery to prevent hypoglycemia. The insulin pump’s infusion set can be worn for 7 days, rather than 3 days as with the older system, and the glucose target level can be set as low as 100 mg/dL.
And in contrast to the older MiniMed 670G system, which tended to frequently boot users out of automated mode, with the 780G, users spent an average of 95% of the time in the automated “SmartGuard” mode.
In the pivotal U.S. trial, overall, patients who used the 780G spent 75% of the time in ideal glucose range (70-180 mg/dL) and 1.8% of the time below that range. Overnight, the figures were 82% and 1.5%, respectively. With the glucose target set at 100 mg/dL and active insulin time set to 2 hours, patients spent 78.8% of time in range without increased hyperglycemia.
In the ADAPT study, with the 780G, there was a 26% increase in time in ideal glucose range and a 1.4% reduction in A1c compared with results for patients who received multiple daily insulin injections with intermittently scanned continuous glucose monitoring, without an increase in hypoglycemia. Overnight, time in range increased 30.2%. The results were sustained at 1 year.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved Medtronic Minimed’s 780G automated insulin delivery system with the Guardian 4 sensor.
The latest so-called artificial pancreas system is approved for people aged 7 years and older who have type 1 diabetes. Medtronic will begin taking preorders for the 780G on May 15, 2023. Users of the current MiniMed 770G will be eligible for no-cost remote software upgrades.
The 780G is currently available in 105 countries. It has been available in Europe since 2020 and in the United Kingdom since 2021. It is the first automated insulin delivery system to automatically administer bolus correction insulin doses every 5 minutes to correct meal-related hyperglycemia.
This so-called meal detection technology doesn’t replace manual premeal boluses but does provide extra insulin if the premeal bolus is skipped or is insufficient.
As with other automated systems, the 780G automatically adjusts basal insulin doses up or down based on glucose levels and trends and shuts off insulin delivery to prevent hypoglycemia. The insulin pump’s infusion set can be worn for 7 days, rather than 3 days as with the older system, and the glucose target level can be set as low as 100 mg/dL.
And in contrast to the older MiniMed 670G system, which tended to frequently boot users out of automated mode, with the 780G, users spent an average of 95% of the time in the automated “SmartGuard” mode.
In the pivotal U.S. trial, overall, patients who used the 780G spent 75% of the time in ideal glucose range (70-180 mg/dL) and 1.8% of the time below that range. Overnight, the figures were 82% and 1.5%, respectively. With the glucose target set at 100 mg/dL and active insulin time set to 2 hours, patients spent 78.8% of time in range without increased hyperglycemia.
In the ADAPT study, with the 780G, there was a 26% increase in time in ideal glucose range and a 1.4% reduction in A1c compared with results for patients who received multiple daily insulin injections with intermittently scanned continuous glucose monitoring, without an increase in hypoglycemia. Overnight, time in range increased 30.2%. The results were sustained at 1 year.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved Medtronic Minimed’s 780G automated insulin delivery system with the Guardian 4 sensor.
The latest so-called artificial pancreas system is approved for people aged 7 years and older who have type 1 diabetes. Medtronic will begin taking preorders for the 780G on May 15, 2023. Users of the current MiniMed 770G will be eligible for no-cost remote software upgrades.
The 780G is currently available in 105 countries. It has been available in Europe since 2020 and in the United Kingdom since 2021. It is the first automated insulin delivery system to automatically administer bolus correction insulin doses every 5 minutes to correct meal-related hyperglycemia.
This so-called meal detection technology doesn’t replace manual premeal boluses but does provide extra insulin if the premeal bolus is skipped or is insufficient.
As with other automated systems, the 780G automatically adjusts basal insulin doses up or down based on glucose levels and trends and shuts off insulin delivery to prevent hypoglycemia. The insulin pump’s infusion set can be worn for 7 days, rather than 3 days as with the older system, and the glucose target level can be set as low as 100 mg/dL.
And in contrast to the older MiniMed 670G system, which tended to frequently boot users out of automated mode, with the 780G, users spent an average of 95% of the time in the automated “SmartGuard” mode.
In the pivotal U.S. trial, overall, patients who used the 780G spent 75% of the time in ideal glucose range (70-180 mg/dL) and 1.8% of the time below that range. Overnight, the figures were 82% and 1.5%, respectively. With the glucose target set at 100 mg/dL and active insulin time set to 2 hours, patients spent 78.8% of time in range without increased hyperglycemia.
In the ADAPT study, with the 780G, there was a 26% increase in time in ideal glucose range and a 1.4% reduction in A1c compared with results for patients who received multiple daily insulin injections with intermittently scanned continuous glucose monitoring, without an increase in hypoglycemia. Overnight, time in range increased 30.2%. The results were sustained at 1 year.
A version of this article first appeared on Medscape.com.
Poor diet causes 70% of type 2 diabetes, says new study
Poor diets account for most newly diagnosed type 2 diabetes cases worldwide, a new analysis has found.
More specifically, the modeling study showed that roughly 14 million cases of type 2 diabetes – or 70% of total type 2 diabetes diagnoses in 2018 – were linked with a poor diet, found Meghan O’Hearn, a doctoral student at the Friedman School of Nutrition Science and Policy, Tufts University, Boston, and colleagues. The study was published online in Nature Medicine.
The results also indicate that the greatest burdens of type 2 diabetes were accounted for by excess wheat intake and refined rice (24.6%), excess processed meat consumption (20.3%), and inadequate whole-grain consumption (26.1%). Factors such as drinking too much fruit juice and not eating enough nonstarchy vegetables, nuts, or seeds, had less of an impact on new cases of the disease, the researchers determined.
“These findings can help inform nutritional priorities for clinicians, policymakers, and private sector actors as they encourage healthier dietary choices that address this global epidemic,” Ms. O’Hearn said in a press release.
Prior research has suggested that poor diet contributes to about 40% of type 2 diabetes cases worldwide, the researchers note.
The team attributes their finding of a 70% contribution to the new information in their analysis, such as the first-ever inclusion of refined grains, which was one of the top contributors to diabetes burden, and updated data on dietary habits based on national individual-level dietary surveys rather than agricultural estimates.
“Our study suggests poor carbohydrate quality is a leading driver of diet-attributable type 2 diabetes globally and with important variation by nation and over time,” said senior author Dariush Mozaffarian, MD, DrPh, MPH, who is the Jean Mayer Professor of Nutrition at the Friedman School of Nutrition Science and Policy.
“These new findings reveal critical areas for national and global focus to improve nutrition and reduce devastating burdens of diabetes,” he noted.
“Left unchecked and with incidence only projected to rise, type 2 diabetes will continue to impact population health, economic productivity, [and] health care system capacity, [as well as] drive health inequities worldwide,” Ms. O’Hearn said.
It’s about reducing harmful dietary components
Ms. O’Hearn and colleagues set out to fill information gaps in knowledge about how the global burden of diet-associated type 2 diabetes is impacted by disparities and other factors known to influence risk, including dietary components.
They used information from the Global Dietary Database to study dietary intake in 184 nations from 1990 to 2018. They also studied demographics from multiple sources, estimates of type 2 diabetes incidence around the world, and data on food choices, including the effect of 11 dietary factors, from prior research.
They found that there were 8.6 million more cases of type 2 diabetes in 2018 than in 1990 because of poor diet.
Regionally, Central and Eastern Europe and Central Asia had the greatest number of type 2 diabetes cases linked to diet, particularly Poland and Russia, where diets tend to be rich in red meat, processed meat, and potatoes. Incidence was also high in Latin America and the Caribbean, especially in Colombia and Mexico, which was attributed to high consumption of sugary drinks and processed meat and low intake of whole grains.
Regions where diet had less of an impact on type 2 diabetes cases included South Asia and sub-Saharan Africa, although the largest increases in type 2 diabetes due to poor diet between 1990 and 2018 were observed in sub-Saharan Africa.
Diet-attributable type 2 diabetes was generally larger among urban versus rural residents and higher versus lower educated individuals, except in high-income countries, Central and Eastern Europe, and Central Asia, where burdens were larger in rural residents and in lower educated individuals.
Notably, women had lower proportions of diet-related type 2 diabetes, compared with men, and these proportions were inversely related to age.
Excess intake of harmful dietary factors contributed a greater percentage of the burden of type 2 diabetes globally (60.8%) than did insufficient intake of protective dietary factors (39.2%).
“Future research should address whether more complex diet–type 2 diabetes dose–response relationships exist,” the authors conclude.
Ms. O’Hearn has reported receiving research funding from the Gates Foundation, as well as the National Institutes of Health and Vail Innovative Global Research and employment with Food Systems for the Future. Dr. Mozaffarian has reported receiving funding from the National Institutes of Health, the Gates Foundation, the Rockefeller Foundation, Vail Innovative Global Research, and the Kaiser Permanente Fund at East Bay Community Foundation; personal fees from Acasti Pharma, Barilla, Danone, and Motif FoodWorks; is on the scientific advisory board for Beren Therapeutics, Brightseed, Calibrate, DiscernDx, Elysium Health, Filtricine, HumanCo, January, Perfect Day, Tiny Organics and (ended) Day Two and Season Health; has stock ownership in Calibrate and HumanCo; and receives chapter royalties from UpToDate.
A version of this article first appeared on Medscape.com.
Poor diets account for most newly diagnosed type 2 diabetes cases worldwide, a new analysis has found.
More specifically, the modeling study showed that roughly 14 million cases of type 2 diabetes – or 70% of total type 2 diabetes diagnoses in 2018 – were linked with a poor diet, found Meghan O’Hearn, a doctoral student at the Friedman School of Nutrition Science and Policy, Tufts University, Boston, and colleagues. The study was published online in Nature Medicine.
The results also indicate that the greatest burdens of type 2 diabetes were accounted for by excess wheat intake and refined rice (24.6%), excess processed meat consumption (20.3%), and inadequate whole-grain consumption (26.1%). Factors such as drinking too much fruit juice and not eating enough nonstarchy vegetables, nuts, or seeds, had less of an impact on new cases of the disease, the researchers determined.
“These findings can help inform nutritional priorities for clinicians, policymakers, and private sector actors as they encourage healthier dietary choices that address this global epidemic,” Ms. O’Hearn said in a press release.
Prior research has suggested that poor diet contributes to about 40% of type 2 diabetes cases worldwide, the researchers note.
The team attributes their finding of a 70% contribution to the new information in their analysis, such as the first-ever inclusion of refined grains, which was one of the top contributors to diabetes burden, and updated data on dietary habits based on national individual-level dietary surveys rather than agricultural estimates.
“Our study suggests poor carbohydrate quality is a leading driver of diet-attributable type 2 diabetes globally and with important variation by nation and over time,” said senior author Dariush Mozaffarian, MD, DrPh, MPH, who is the Jean Mayer Professor of Nutrition at the Friedman School of Nutrition Science and Policy.
“These new findings reveal critical areas for national and global focus to improve nutrition and reduce devastating burdens of diabetes,” he noted.
“Left unchecked and with incidence only projected to rise, type 2 diabetes will continue to impact population health, economic productivity, [and] health care system capacity, [as well as] drive health inequities worldwide,” Ms. O’Hearn said.
It’s about reducing harmful dietary components
Ms. O’Hearn and colleagues set out to fill information gaps in knowledge about how the global burden of diet-associated type 2 diabetes is impacted by disparities and other factors known to influence risk, including dietary components.
They used information from the Global Dietary Database to study dietary intake in 184 nations from 1990 to 2018. They also studied demographics from multiple sources, estimates of type 2 diabetes incidence around the world, and data on food choices, including the effect of 11 dietary factors, from prior research.
They found that there were 8.6 million more cases of type 2 diabetes in 2018 than in 1990 because of poor diet.
Regionally, Central and Eastern Europe and Central Asia had the greatest number of type 2 diabetes cases linked to diet, particularly Poland and Russia, where diets tend to be rich in red meat, processed meat, and potatoes. Incidence was also high in Latin America and the Caribbean, especially in Colombia and Mexico, which was attributed to high consumption of sugary drinks and processed meat and low intake of whole grains.
Regions where diet had less of an impact on type 2 diabetes cases included South Asia and sub-Saharan Africa, although the largest increases in type 2 diabetes due to poor diet between 1990 and 2018 were observed in sub-Saharan Africa.
Diet-attributable type 2 diabetes was generally larger among urban versus rural residents and higher versus lower educated individuals, except in high-income countries, Central and Eastern Europe, and Central Asia, where burdens were larger in rural residents and in lower educated individuals.
Notably, women had lower proportions of diet-related type 2 diabetes, compared with men, and these proportions were inversely related to age.
Excess intake of harmful dietary factors contributed a greater percentage of the burden of type 2 diabetes globally (60.8%) than did insufficient intake of protective dietary factors (39.2%).
“Future research should address whether more complex diet–type 2 diabetes dose–response relationships exist,” the authors conclude.
Ms. O’Hearn has reported receiving research funding from the Gates Foundation, as well as the National Institutes of Health and Vail Innovative Global Research and employment with Food Systems for the Future. Dr. Mozaffarian has reported receiving funding from the National Institutes of Health, the Gates Foundation, the Rockefeller Foundation, Vail Innovative Global Research, and the Kaiser Permanente Fund at East Bay Community Foundation; personal fees from Acasti Pharma, Barilla, Danone, and Motif FoodWorks; is on the scientific advisory board for Beren Therapeutics, Brightseed, Calibrate, DiscernDx, Elysium Health, Filtricine, HumanCo, January, Perfect Day, Tiny Organics and (ended) Day Two and Season Health; has stock ownership in Calibrate and HumanCo; and receives chapter royalties from UpToDate.
A version of this article first appeared on Medscape.com.
Poor diets account for most newly diagnosed type 2 diabetes cases worldwide, a new analysis has found.
More specifically, the modeling study showed that roughly 14 million cases of type 2 diabetes – or 70% of total type 2 diabetes diagnoses in 2018 – were linked with a poor diet, found Meghan O’Hearn, a doctoral student at the Friedman School of Nutrition Science and Policy, Tufts University, Boston, and colleagues. The study was published online in Nature Medicine.
The results also indicate that the greatest burdens of type 2 diabetes were accounted for by excess wheat intake and refined rice (24.6%), excess processed meat consumption (20.3%), and inadequate whole-grain consumption (26.1%). Factors such as drinking too much fruit juice and not eating enough nonstarchy vegetables, nuts, or seeds, had less of an impact on new cases of the disease, the researchers determined.
“These findings can help inform nutritional priorities for clinicians, policymakers, and private sector actors as they encourage healthier dietary choices that address this global epidemic,” Ms. O’Hearn said in a press release.
Prior research has suggested that poor diet contributes to about 40% of type 2 diabetes cases worldwide, the researchers note.
The team attributes their finding of a 70% contribution to the new information in their analysis, such as the first-ever inclusion of refined grains, which was one of the top contributors to diabetes burden, and updated data on dietary habits based on national individual-level dietary surveys rather than agricultural estimates.
“Our study suggests poor carbohydrate quality is a leading driver of diet-attributable type 2 diabetes globally and with important variation by nation and over time,” said senior author Dariush Mozaffarian, MD, DrPh, MPH, who is the Jean Mayer Professor of Nutrition at the Friedman School of Nutrition Science and Policy.
“These new findings reveal critical areas for national and global focus to improve nutrition and reduce devastating burdens of diabetes,” he noted.
“Left unchecked and with incidence only projected to rise, type 2 diabetes will continue to impact population health, economic productivity, [and] health care system capacity, [as well as] drive health inequities worldwide,” Ms. O’Hearn said.
It’s about reducing harmful dietary components
Ms. O’Hearn and colleagues set out to fill information gaps in knowledge about how the global burden of diet-associated type 2 diabetes is impacted by disparities and other factors known to influence risk, including dietary components.
They used information from the Global Dietary Database to study dietary intake in 184 nations from 1990 to 2018. They also studied demographics from multiple sources, estimates of type 2 diabetes incidence around the world, and data on food choices, including the effect of 11 dietary factors, from prior research.
They found that there were 8.6 million more cases of type 2 diabetes in 2018 than in 1990 because of poor diet.
Regionally, Central and Eastern Europe and Central Asia had the greatest number of type 2 diabetes cases linked to diet, particularly Poland and Russia, where diets tend to be rich in red meat, processed meat, and potatoes. Incidence was also high in Latin America and the Caribbean, especially in Colombia and Mexico, which was attributed to high consumption of sugary drinks and processed meat and low intake of whole grains.
Regions where diet had less of an impact on type 2 diabetes cases included South Asia and sub-Saharan Africa, although the largest increases in type 2 diabetes due to poor diet between 1990 and 2018 were observed in sub-Saharan Africa.
Diet-attributable type 2 diabetes was generally larger among urban versus rural residents and higher versus lower educated individuals, except in high-income countries, Central and Eastern Europe, and Central Asia, where burdens were larger in rural residents and in lower educated individuals.
Notably, women had lower proportions of diet-related type 2 diabetes, compared with men, and these proportions were inversely related to age.
Excess intake of harmful dietary factors contributed a greater percentage of the burden of type 2 diabetes globally (60.8%) than did insufficient intake of protective dietary factors (39.2%).
“Future research should address whether more complex diet–type 2 diabetes dose–response relationships exist,” the authors conclude.
Ms. O’Hearn has reported receiving research funding from the Gates Foundation, as well as the National Institutes of Health and Vail Innovative Global Research and employment with Food Systems for the Future. Dr. Mozaffarian has reported receiving funding from the National Institutes of Health, the Gates Foundation, the Rockefeller Foundation, Vail Innovative Global Research, and the Kaiser Permanente Fund at East Bay Community Foundation; personal fees from Acasti Pharma, Barilla, Danone, and Motif FoodWorks; is on the scientific advisory board for Beren Therapeutics, Brightseed, Calibrate, DiscernDx, Elysium Health, Filtricine, HumanCo, January, Perfect Day, Tiny Organics and (ended) Day Two and Season Health; has stock ownership in Calibrate and HumanCo; and receives chapter royalties from UpToDate.
A version of this article first appeared on Medscape.com.
FROM NATURE MEDICINE
What are the healthiest drinks for patients with type 2 diabetes?
The researchers examined data on almost 15,500 participants with type 2 diabetes from two major studies, finding that the highest level of consumption of SSBs was associated with a 20% increased risk of all-cause mortality and a 25% raised risk of cardiovascular disease, compared with consumption of the least amounts of these products.
The research, published in BMJ, also showed that drinking coffee, tea, plain water, and low-fat milk reduced the risk of all-cause death and that switching from SSBs to the other beverages was linked to lower mortality.
“Overall, these results provide additional evidence that emphasizes the importance of beverage choices in maintaining overall health among adults with diabetes,” say senior author Le Ma, PhD, department of nutrition, Harvard School of Public Health, Boston, and colleagues.
“Collectively, these findings all point in the same direction. Lower consumption of SSBs and higher consumption of coffee, tea, plain water, or low-fat milk are optimal for better health outcomes in adults with type 2 diabetes,” Nita G. Forouhi, MD, PhD, emphasizes in an accompanying editorial.
Choice of drink matters
Dr. Forouhi, from the University of Cambridge (England), warned, however, that the findings “cannot be considered cause and effect,” despite the large-scale analysis.
Moreover, “questions remain,” such as the impact of beverage consumption on coronary heart disease and stroke risk, and cancer mortality, with the current study providing “inconclusive” data on the latter.
There was also no data on the addition of sugar to tea or coffee, “so the comparative health effects of unsweetened and sweetened hot beverages remain unclear,” Dr. Forouhi points out. Also unknown is whether the type of tea consumed has a differential effect.
Despite these and other reservations, she says that overall, “Choice of beverage clearly matters.”
“The case for avoiding sugar-sweetened beverages is compelling, and it is supported by various fiscal measures in more than 45 countries. It is reasonable to shift the focus to drinks that are most likely to have positive health impacts: coffee, tea, plain water, and low-fat milk,” she notes.
Dr. Forouhi ends by underlining that the current findings tally with those seen in the general population, so “one important message is that having diabetes does not have to be especially restrictive.”
Expanding the evidence
It was estimated that 537 million adults worldwide had type 2 diabetes in 2021, a figure set to increase to 783 million by 2045, say the authors.
Individuals with type 2 diabetes have an increased risk of cardiovascular disease, among many other comorbidities, as well as premature death. Dietary interventions can play an important role in managing these risks.
Recommendations on the healthiest beverages to drink are largely based on evidence from the general population, and data are limited on the best options for adults with type 2 diabetes, who have altered metabolism, the researchers note.
To expand on this, they examined data from the Nurses’ Health Study, which enrolled female registered nurses aged 30-55 years and was initiated in 1976, and the Health Professionals Follow-Up Study, which included male health professionals aged 40-75 years and was initiated in 1996.
For the current analysis, 11,399 women and 4,087 men with type 2 diabetes were included from the two studies, of whom 2,715 were diagnosed before study entry.
Participants’ average daily beverage intake was assessed using a validated food frequency questionnaire administered every 2-4 years. SSBs included caffeinated and caffeine-free colas, other carbonated SSBs, and noncarbonated SSBs, such as fruit punches, lemonades, or other fruit drinks.
During 285,967 person-years of follow-up, there were 7,638 (49.3%) deaths, and 3,447 (22.3%) cases of incident cardiovascular disease were documented during 248,447 person-years of follow-up.
Fully adjusted multivariate analysis comparing the lowest and highest beverage intake indicated that SSBs were associated with a significant increase in all-cause mortality, at a pooled hazard ratio of 1.20, or 1.08 for each additional serving per day (P = .01).
In contrast, the associations between all-cause mortality and consumption of artificially sweetened beverages, fruit juice, and full-fat milk were not significant, whereas coffee (HR, 0.74), tea (HR, 0.79), plain water (HR, 0.77), and low-fat milk (HR, 0.88) were linked to a reduced risk.
The team reported that there were similar associations between beverage intake and cardiovascular disease incidence, at an HR of 1.25 for SSBs, as well as for cardiovascular disease mortality, at an HR of 1.29.
Participants who increased their tea, coffee, and low-fat milk consumption during the course of the study had lower all-cause mortality than those who did not. Switching from SSBs to other beverages was also associated with lower mortality.
The researchers note, however, that there are “several potential limitations” to their study, including that “individual beverage consumption may be correlated with other dietary and lifestyle risk factors for cardiovascular disease incidence and mortality among adults with [type 2] diabetes.”
The study was sponsored by the National Institutes of Health. Dr. Ma has reported no relevant financial relationships. Disclosures for the other authors are listed with the article. Dr. Forouhi has declared receiving support from the U.K. Medical Research Council Epidemiology Unit and U.K. National Institute for Health and Care Research Biomedical Research Centre Cambridge.
A version of this article first appeared on Medscape.com.
The researchers examined data on almost 15,500 participants with type 2 diabetes from two major studies, finding that the highest level of consumption of SSBs was associated with a 20% increased risk of all-cause mortality and a 25% raised risk of cardiovascular disease, compared with consumption of the least amounts of these products.
The research, published in BMJ, also showed that drinking coffee, tea, plain water, and low-fat milk reduced the risk of all-cause death and that switching from SSBs to the other beverages was linked to lower mortality.
“Overall, these results provide additional evidence that emphasizes the importance of beverage choices in maintaining overall health among adults with diabetes,” say senior author Le Ma, PhD, department of nutrition, Harvard School of Public Health, Boston, and colleagues.
“Collectively, these findings all point in the same direction. Lower consumption of SSBs and higher consumption of coffee, tea, plain water, or low-fat milk are optimal for better health outcomes in adults with type 2 diabetes,” Nita G. Forouhi, MD, PhD, emphasizes in an accompanying editorial.
Choice of drink matters
Dr. Forouhi, from the University of Cambridge (England), warned, however, that the findings “cannot be considered cause and effect,” despite the large-scale analysis.
Moreover, “questions remain,” such as the impact of beverage consumption on coronary heart disease and stroke risk, and cancer mortality, with the current study providing “inconclusive” data on the latter.
There was also no data on the addition of sugar to tea or coffee, “so the comparative health effects of unsweetened and sweetened hot beverages remain unclear,” Dr. Forouhi points out. Also unknown is whether the type of tea consumed has a differential effect.
Despite these and other reservations, she says that overall, “Choice of beverage clearly matters.”
“The case for avoiding sugar-sweetened beverages is compelling, and it is supported by various fiscal measures in more than 45 countries. It is reasonable to shift the focus to drinks that are most likely to have positive health impacts: coffee, tea, plain water, and low-fat milk,” she notes.
Dr. Forouhi ends by underlining that the current findings tally with those seen in the general population, so “one important message is that having diabetes does not have to be especially restrictive.”
Expanding the evidence
It was estimated that 537 million adults worldwide had type 2 diabetes in 2021, a figure set to increase to 783 million by 2045, say the authors.
Individuals with type 2 diabetes have an increased risk of cardiovascular disease, among many other comorbidities, as well as premature death. Dietary interventions can play an important role in managing these risks.
Recommendations on the healthiest beverages to drink are largely based on evidence from the general population, and data are limited on the best options for adults with type 2 diabetes, who have altered metabolism, the researchers note.
To expand on this, they examined data from the Nurses’ Health Study, which enrolled female registered nurses aged 30-55 years and was initiated in 1976, and the Health Professionals Follow-Up Study, which included male health professionals aged 40-75 years and was initiated in 1996.
For the current analysis, 11,399 women and 4,087 men with type 2 diabetes were included from the two studies, of whom 2,715 were diagnosed before study entry.
Participants’ average daily beverage intake was assessed using a validated food frequency questionnaire administered every 2-4 years. SSBs included caffeinated and caffeine-free colas, other carbonated SSBs, and noncarbonated SSBs, such as fruit punches, lemonades, or other fruit drinks.
During 285,967 person-years of follow-up, there were 7,638 (49.3%) deaths, and 3,447 (22.3%) cases of incident cardiovascular disease were documented during 248,447 person-years of follow-up.
Fully adjusted multivariate analysis comparing the lowest and highest beverage intake indicated that SSBs were associated with a significant increase in all-cause mortality, at a pooled hazard ratio of 1.20, or 1.08 for each additional serving per day (P = .01).
In contrast, the associations between all-cause mortality and consumption of artificially sweetened beverages, fruit juice, and full-fat milk were not significant, whereas coffee (HR, 0.74), tea (HR, 0.79), plain water (HR, 0.77), and low-fat milk (HR, 0.88) were linked to a reduced risk.
The team reported that there were similar associations between beverage intake and cardiovascular disease incidence, at an HR of 1.25 for SSBs, as well as for cardiovascular disease mortality, at an HR of 1.29.
Participants who increased their tea, coffee, and low-fat milk consumption during the course of the study had lower all-cause mortality than those who did not. Switching from SSBs to other beverages was also associated with lower mortality.
The researchers note, however, that there are “several potential limitations” to their study, including that “individual beverage consumption may be correlated with other dietary and lifestyle risk factors for cardiovascular disease incidence and mortality among adults with [type 2] diabetes.”
The study was sponsored by the National Institutes of Health. Dr. Ma has reported no relevant financial relationships. Disclosures for the other authors are listed with the article. Dr. Forouhi has declared receiving support from the U.K. Medical Research Council Epidemiology Unit and U.K. National Institute for Health and Care Research Biomedical Research Centre Cambridge.
A version of this article first appeared on Medscape.com.
The researchers examined data on almost 15,500 participants with type 2 diabetes from two major studies, finding that the highest level of consumption of SSBs was associated with a 20% increased risk of all-cause mortality and a 25% raised risk of cardiovascular disease, compared with consumption of the least amounts of these products.
The research, published in BMJ, also showed that drinking coffee, tea, plain water, and low-fat milk reduced the risk of all-cause death and that switching from SSBs to the other beverages was linked to lower mortality.
“Overall, these results provide additional evidence that emphasizes the importance of beverage choices in maintaining overall health among adults with diabetes,” say senior author Le Ma, PhD, department of nutrition, Harvard School of Public Health, Boston, and colleagues.
“Collectively, these findings all point in the same direction. Lower consumption of SSBs and higher consumption of coffee, tea, plain water, or low-fat milk are optimal for better health outcomes in adults with type 2 diabetes,” Nita G. Forouhi, MD, PhD, emphasizes in an accompanying editorial.
Choice of drink matters
Dr. Forouhi, from the University of Cambridge (England), warned, however, that the findings “cannot be considered cause and effect,” despite the large-scale analysis.
Moreover, “questions remain,” such as the impact of beverage consumption on coronary heart disease and stroke risk, and cancer mortality, with the current study providing “inconclusive” data on the latter.
There was also no data on the addition of sugar to tea or coffee, “so the comparative health effects of unsweetened and sweetened hot beverages remain unclear,” Dr. Forouhi points out. Also unknown is whether the type of tea consumed has a differential effect.
Despite these and other reservations, she says that overall, “Choice of beverage clearly matters.”
“The case for avoiding sugar-sweetened beverages is compelling, and it is supported by various fiscal measures in more than 45 countries. It is reasonable to shift the focus to drinks that are most likely to have positive health impacts: coffee, tea, plain water, and low-fat milk,” she notes.
Dr. Forouhi ends by underlining that the current findings tally with those seen in the general population, so “one important message is that having diabetes does not have to be especially restrictive.”
Expanding the evidence
It was estimated that 537 million adults worldwide had type 2 diabetes in 2021, a figure set to increase to 783 million by 2045, say the authors.
Individuals with type 2 diabetes have an increased risk of cardiovascular disease, among many other comorbidities, as well as premature death. Dietary interventions can play an important role in managing these risks.
Recommendations on the healthiest beverages to drink are largely based on evidence from the general population, and data are limited on the best options for adults with type 2 diabetes, who have altered metabolism, the researchers note.
To expand on this, they examined data from the Nurses’ Health Study, which enrolled female registered nurses aged 30-55 years and was initiated in 1976, and the Health Professionals Follow-Up Study, which included male health professionals aged 40-75 years and was initiated in 1996.
For the current analysis, 11,399 women and 4,087 men with type 2 diabetes were included from the two studies, of whom 2,715 were diagnosed before study entry.
Participants’ average daily beverage intake was assessed using a validated food frequency questionnaire administered every 2-4 years. SSBs included caffeinated and caffeine-free colas, other carbonated SSBs, and noncarbonated SSBs, such as fruit punches, lemonades, or other fruit drinks.
During 285,967 person-years of follow-up, there were 7,638 (49.3%) deaths, and 3,447 (22.3%) cases of incident cardiovascular disease were documented during 248,447 person-years of follow-up.
Fully adjusted multivariate analysis comparing the lowest and highest beverage intake indicated that SSBs were associated with a significant increase in all-cause mortality, at a pooled hazard ratio of 1.20, or 1.08 for each additional serving per day (P = .01).
In contrast, the associations between all-cause mortality and consumption of artificially sweetened beverages, fruit juice, and full-fat milk were not significant, whereas coffee (HR, 0.74), tea (HR, 0.79), plain water (HR, 0.77), and low-fat milk (HR, 0.88) were linked to a reduced risk.
The team reported that there were similar associations between beverage intake and cardiovascular disease incidence, at an HR of 1.25 for SSBs, as well as for cardiovascular disease mortality, at an HR of 1.29.
Participants who increased their tea, coffee, and low-fat milk consumption during the course of the study had lower all-cause mortality than those who did not. Switching from SSBs to other beverages was also associated with lower mortality.
The researchers note, however, that there are “several potential limitations” to their study, including that “individual beverage consumption may be correlated with other dietary and lifestyle risk factors for cardiovascular disease incidence and mortality among adults with [type 2] diabetes.”
The study was sponsored by the National Institutes of Health. Dr. Ma has reported no relevant financial relationships. Disclosures for the other authors are listed with the article. Dr. Forouhi has declared receiving support from the U.K. Medical Research Council Epidemiology Unit and U.K. National Institute for Health and Care Research Biomedical Research Centre Cambridge.
A version of this article first appeared on Medscape.com.
FROM THE BMJ
The Prediabetes Debate: Should the Diabetes Diagnostic Threshold Be Lowered, Allowing Clinicians to Intervene Earlier?
I believe that the diagnosis of type 2 diabetes mellitus (T2DM) should be broadened to match the glycemic thresholds currently used for prediabetes. This would eliminate the need for a separate category, the “prediabetes” nomenclature, and allow for earlier therapeutic intervention in patients. Current diabetes diagnostic thresholds do not reflect the latest advancements in T2DM understanding. The latest in T2DM research suggests that intervening and treating prediabetes earlier could potentially offer better clinical outcomes and enhance patients’ quality of life, as cell and tissue damage occurs early and leads to dysfunction prior to a diabetes diagnosis.1 T2DM is a highly complex disease with multifactorial causes beyond hyperglycemia that should be considered, such as hyperlipidemia, insulin resistance, hyperinsulinemia, and autoimmune inflammatory mechanisms that lead to β-cell dysfunction or failure, which results in hyperglycemia.
Prediabetes is associated with micro- and macrovascular complications that can occur early in the progression to frank disease state.1,2 This phase of diabetes also includes insulin resistance, impaired incretin action, insulin hypersecretion, increased lipolysis, and ectopic lipid storage—all of which damage β cells. These dysfunctions are also present in the frank diabetic disease state.3,4 Furthermore, diabetic retinopathy occurs in 8% to 12% of patients with prediabetes, and retinopathy begins earlier than previously thought, with neuroinflammation occurring even before vascular damage.5,6 Unfortunately, these neuro-inflammatory lesions cannot be detected with the typical instruments used in an ophthalmologist’s office.
It is believed that, through the principle of metabolic memory, even a moderate increase or episodic spikes in blood glucose can lead to negative effects in prediabetic patients who are susceptible to T2DM.1,5 Therefore, a lower diabetes diagnostic threshold could allow for earlier, more precise, and personalized therapies based on each patient’s individual risk factors and biomarkers. With a diagnosis of T2DM at the current prediabetes threshold, patients could receive treatment covered by health insurance while in the “prediabetic” state—treatment that would not have been previously approved. Patients should be treated earlier and on an individual basis with counseling on diet and lifestyle changes and antidiabetic agents to reduce glycemic levels, preserve β cells, and reduce cardiovascular [CV] or renal risk, among other complications.6,7
Moreover, the newer agents for treating diabetes such as glucagon-like pepetide-1 receptor agonists and sodium-glucose cotransporter 2 inhibitors, which are also associated with reduction in adverse CV and/or renal outcomes, could be beneficial if administered early in the prediabetic stage of disease.8,9
Early lifestyle and pharmacologic interventions can reduce the rate of progression from prediabetes to diabetes as well as complications and associated conditions, and even potentially result in remission or a full reversal of diabetes. These “side benefits” of lowering the diabetes diagnostic threshold (over and above glycemic control) make any cost-effectiveness calculations all the more advantageous to individual patients as well as to society.
Given the numerous benefits of earlier intervention for diabetes treatment, I believe a call to re-evaluate the current diabetes diagnostic threshold is in order, as it will do a great service for all patients who are currently at risk for developing T2DM.
Armato JP, DeFronzo RA, Abdul-Ghani M, Ruby RJ. Successful treatment of prediabetes in clinical practice using physiological assessment (STOP DIABETES). Lancet Diabetes Endocrinol. 2018;6:781-789.
Schwartz SS, Epstein S, Corkey BE, et al. A unified pathophysiological construct of diabetes and its complications. Trends Endocrinol Metab. 2017;28:645-655.
American Diabetes Association. Standards of Medical Care in Diabetes – 2021. Diabetes Care. 2021;44(S15–S39):S111-S124.
Brannick B, Wynn A, Dagogo-Jack S. Prediabetes as a toxic environment for the initiation of microvascular and macrovascular complications. Exp Biol Med (Maywood). 2016;241:1323-1331.
Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care. 1997;20:1183-1197.
Sinclair SH, Schwartz SS. Diabetic retinopathy–an underdiagnosed and undertreated inflammatory, neuro-vascular complication of diabetes. Front Endocrinol (Lausanne). 2019;10:843.
Edwards CM, Cusi K. Prediabetes: a worldwide epidemic. Endocrinol Metab Clin N Am. 2016;45:751-764.
Kanat M, DeFronzo RA, Abdul-Ghani MA. Treatment of prediabetes. World J Diabetes. 2015;6:1207-1222.
Dankner R, Roth J. The personalized approach for detecting prediabetes and diabetes. Curr Diabetes Rev. 2016;12:58-65.
I believe that the diagnosis of type 2 diabetes mellitus (T2DM) should be broadened to match the glycemic thresholds currently used for prediabetes. This would eliminate the need for a separate category, the “prediabetes” nomenclature, and allow for earlier therapeutic intervention in patients. Current diabetes diagnostic thresholds do not reflect the latest advancements in T2DM understanding. The latest in T2DM research suggests that intervening and treating prediabetes earlier could potentially offer better clinical outcomes and enhance patients’ quality of life, as cell and tissue damage occurs early and leads to dysfunction prior to a diabetes diagnosis.1 T2DM is a highly complex disease with multifactorial causes beyond hyperglycemia that should be considered, such as hyperlipidemia, insulin resistance, hyperinsulinemia, and autoimmune inflammatory mechanisms that lead to β-cell dysfunction or failure, which results in hyperglycemia.
Prediabetes is associated with micro- and macrovascular complications that can occur early in the progression to frank disease state.1,2 This phase of diabetes also includes insulin resistance, impaired incretin action, insulin hypersecretion, increased lipolysis, and ectopic lipid storage—all of which damage β cells. These dysfunctions are also present in the frank diabetic disease state.3,4 Furthermore, diabetic retinopathy occurs in 8% to 12% of patients with prediabetes, and retinopathy begins earlier than previously thought, with neuroinflammation occurring even before vascular damage.5,6 Unfortunately, these neuro-inflammatory lesions cannot be detected with the typical instruments used in an ophthalmologist’s office.
It is believed that, through the principle of metabolic memory, even a moderate increase or episodic spikes in blood glucose can lead to negative effects in prediabetic patients who are susceptible to T2DM.1,5 Therefore, a lower diabetes diagnostic threshold could allow for earlier, more precise, and personalized therapies based on each patient’s individual risk factors and biomarkers. With a diagnosis of T2DM at the current prediabetes threshold, patients could receive treatment covered by health insurance while in the “prediabetic” state—treatment that would not have been previously approved. Patients should be treated earlier and on an individual basis with counseling on diet and lifestyle changes and antidiabetic agents to reduce glycemic levels, preserve β cells, and reduce cardiovascular [CV] or renal risk, among other complications.6,7
Moreover, the newer agents for treating diabetes such as glucagon-like pepetide-1 receptor agonists and sodium-glucose cotransporter 2 inhibitors, which are also associated with reduction in adverse CV and/or renal outcomes, could be beneficial if administered early in the prediabetic stage of disease.8,9
Early lifestyle and pharmacologic interventions can reduce the rate of progression from prediabetes to diabetes as well as complications and associated conditions, and even potentially result in remission or a full reversal of diabetes. These “side benefits” of lowering the diabetes diagnostic threshold (over and above glycemic control) make any cost-effectiveness calculations all the more advantageous to individual patients as well as to society.
Given the numerous benefits of earlier intervention for diabetes treatment, I believe a call to re-evaluate the current diabetes diagnostic threshold is in order, as it will do a great service for all patients who are currently at risk for developing T2DM.
I believe that the diagnosis of type 2 diabetes mellitus (T2DM) should be broadened to match the glycemic thresholds currently used for prediabetes. This would eliminate the need for a separate category, the “prediabetes” nomenclature, and allow for earlier therapeutic intervention in patients. Current diabetes diagnostic thresholds do not reflect the latest advancements in T2DM understanding. The latest in T2DM research suggests that intervening and treating prediabetes earlier could potentially offer better clinical outcomes and enhance patients’ quality of life, as cell and tissue damage occurs early and leads to dysfunction prior to a diabetes diagnosis.1 T2DM is a highly complex disease with multifactorial causes beyond hyperglycemia that should be considered, such as hyperlipidemia, insulin resistance, hyperinsulinemia, and autoimmune inflammatory mechanisms that lead to β-cell dysfunction or failure, which results in hyperglycemia.
Prediabetes is associated with micro- and macrovascular complications that can occur early in the progression to frank disease state.1,2 This phase of diabetes also includes insulin resistance, impaired incretin action, insulin hypersecretion, increased lipolysis, and ectopic lipid storage—all of which damage β cells. These dysfunctions are also present in the frank diabetic disease state.3,4 Furthermore, diabetic retinopathy occurs in 8% to 12% of patients with prediabetes, and retinopathy begins earlier than previously thought, with neuroinflammation occurring even before vascular damage.5,6 Unfortunately, these neuro-inflammatory lesions cannot be detected with the typical instruments used in an ophthalmologist’s office.
It is believed that, through the principle of metabolic memory, even a moderate increase or episodic spikes in blood glucose can lead to negative effects in prediabetic patients who are susceptible to T2DM.1,5 Therefore, a lower diabetes diagnostic threshold could allow for earlier, more precise, and personalized therapies based on each patient’s individual risk factors and biomarkers. With a diagnosis of T2DM at the current prediabetes threshold, patients could receive treatment covered by health insurance while in the “prediabetic” state—treatment that would not have been previously approved. Patients should be treated earlier and on an individual basis with counseling on diet and lifestyle changes and antidiabetic agents to reduce glycemic levels, preserve β cells, and reduce cardiovascular [CV] or renal risk, among other complications.6,7
Moreover, the newer agents for treating diabetes such as glucagon-like pepetide-1 receptor agonists and sodium-glucose cotransporter 2 inhibitors, which are also associated with reduction in adverse CV and/or renal outcomes, could be beneficial if administered early in the prediabetic stage of disease.8,9
Early lifestyle and pharmacologic interventions can reduce the rate of progression from prediabetes to diabetes as well as complications and associated conditions, and even potentially result in remission or a full reversal of diabetes. These “side benefits” of lowering the diabetes diagnostic threshold (over and above glycemic control) make any cost-effectiveness calculations all the more advantageous to individual patients as well as to society.
Given the numerous benefits of earlier intervention for diabetes treatment, I believe a call to re-evaluate the current diabetes diagnostic threshold is in order, as it will do a great service for all patients who are currently at risk for developing T2DM.
Armato JP, DeFronzo RA, Abdul-Ghani M, Ruby RJ. Successful treatment of prediabetes in clinical practice using physiological assessment (STOP DIABETES). Lancet Diabetes Endocrinol. 2018;6:781-789.
Schwartz SS, Epstein S, Corkey BE, et al. A unified pathophysiological construct of diabetes and its complications. Trends Endocrinol Metab. 2017;28:645-655.
American Diabetes Association. Standards of Medical Care in Diabetes – 2021. Diabetes Care. 2021;44(S15–S39):S111-S124.
Brannick B, Wynn A, Dagogo-Jack S. Prediabetes as a toxic environment for the initiation of microvascular and macrovascular complications. Exp Biol Med (Maywood). 2016;241:1323-1331.
Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care. 1997;20:1183-1197.
Sinclair SH, Schwartz SS. Diabetic retinopathy–an underdiagnosed and undertreated inflammatory, neuro-vascular complication of diabetes. Front Endocrinol (Lausanne). 2019;10:843.
Edwards CM, Cusi K. Prediabetes: a worldwide epidemic. Endocrinol Metab Clin N Am. 2016;45:751-764.
Kanat M, DeFronzo RA, Abdul-Ghani MA. Treatment of prediabetes. World J Diabetes. 2015;6:1207-1222.
Dankner R, Roth J. The personalized approach for detecting prediabetes and diabetes. Curr Diabetes Rev. 2016;12:58-65.
Armato JP, DeFronzo RA, Abdul-Ghani M, Ruby RJ. Successful treatment of prediabetes in clinical practice using physiological assessment (STOP DIABETES). Lancet Diabetes Endocrinol. 2018;6:781-789.
Schwartz SS, Epstein S, Corkey BE, et al. A unified pathophysiological construct of diabetes and its complications. Trends Endocrinol Metab. 2017;28:645-655.
American Diabetes Association. Standards of Medical Care in Diabetes – 2021. Diabetes Care. 2021;44(S15–S39):S111-S124.
Brannick B, Wynn A, Dagogo-Jack S. Prediabetes as a toxic environment for the initiation of microvascular and macrovascular complications. Exp Biol Med (Maywood). 2016;241:1323-1331.
Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care. 1997;20:1183-1197.
Sinclair SH, Schwartz SS. Diabetic retinopathy–an underdiagnosed and undertreated inflammatory, neuro-vascular complication of diabetes. Front Endocrinol (Lausanne). 2019;10:843.
Edwards CM, Cusi K. Prediabetes: a worldwide epidemic. Endocrinol Metab Clin N Am. 2016;45:751-764.
Kanat M, DeFronzo RA, Abdul-Ghani MA. Treatment of prediabetes. World J Diabetes. 2015;6:1207-1222.
Dankner R, Roth J. The personalized approach for detecting prediabetes and diabetes. Curr Diabetes Rev. 2016;12:58-65.





