User login
Adjuvant abemaciclib+ET shows treatment benefit regardless of menopausal status in HR+/HER2− early BC
Key clinical point: The addition of abemaciclib to endocrine therapy (ET) demonstrated robust treatment benefits regardless of menopausal status in women with hormone receptor-positive (HR+), human epidermal growth factor receptor 2-negative (HER2−), high-risk early breast cancer (BC).
Major finding: Abemaciclib+ET significantly improved invasive disease-free survival (iDFS; hazard ratio 0.785; nominal P = .0268) in postmenopausal women with HR+/HER2− BC and led to even greater iDFS improvement (hazard ratio 0.578; P < .0001) in premenopausal women. No new safety events were reported.
Study details: Findings are from an exploratory analysis of the monarchE trial including 5637 patients with HR+/HER2−, node-positive, high-risk early BC who were randomly assigned to receive standard of care ET with/without adjuvant abemaciclib, of which 43.5% and 56.4% of patients were premenopausal and postmenopausal, respectively.
Disclosures: This study was funded by Eli Lilly and Company. Four authors declared being employees and shareholders of Eli Lilly. The other authors reported ties with several sources, including Eli Lilly.
Source: Paluch-Shimon S et al, on behalf of the monarchE investigators. Efficacy and safety results by menopausal status in monarchE: adjuvant abemaciclib combined with endocrine therapy in patients with HR+, HER2−, node-positive, high-risk early breast cancer. Ther Adv Med Oncol. 2023 (Feb 3). Doi: 10.1177/17588359231151840
Key clinical point: The addition of abemaciclib to endocrine therapy (ET) demonstrated robust treatment benefits regardless of menopausal status in women with hormone receptor-positive (HR+), human epidermal growth factor receptor 2-negative (HER2−), high-risk early breast cancer (BC).
Major finding: Abemaciclib+ET significantly improved invasive disease-free survival (iDFS; hazard ratio 0.785; nominal P = .0268) in postmenopausal women with HR+/HER2− BC and led to even greater iDFS improvement (hazard ratio 0.578; P < .0001) in premenopausal women. No new safety events were reported.
Study details: Findings are from an exploratory analysis of the monarchE trial including 5637 patients with HR+/HER2−, node-positive, high-risk early BC who were randomly assigned to receive standard of care ET with/without adjuvant abemaciclib, of which 43.5% and 56.4% of patients were premenopausal and postmenopausal, respectively.
Disclosures: This study was funded by Eli Lilly and Company. Four authors declared being employees and shareholders of Eli Lilly. The other authors reported ties with several sources, including Eli Lilly.
Source: Paluch-Shimon S et al, on behalf of the monarchE investigators. Efficacy and safety results by menopausal status in monarchE: adjuvant abemaciclib combined with endocrine therapy in patients with HR+, HER2−, node-positive, high-risk early breast cancer. Ther Adv Med Oncol. 2023 (Feb 3). Doi: 10.1177/17588359231151840
Key clinical point: The addition of abemaciclib to endocrine therapy (ET) demonstrated robust treatment benefits regardless of menopausal status in women with hormone receptor-positive (HR+), human epidermal growth factor receptor 2-negative (HER2−), high-risk early breast cancer (BC).
Major finding: Abemaciclib+ET significantly improved invasive disease-free survival (iDFS; hazard ratio 0.785; nominal P = .0268) in postmenopausal women with HR+/HER2− BC and led to even greater iDFS improvement (hazard ratio 0.578; P < .0001) in premenopausal women. No new safety events were reported.
Study details: Findings are from an exploratory analysis of the monarchE trial including 5637 patients with HR+/HER2−, node-positive, high-risk early BC who were randomly assigned to receive standard of care ET with/without adjuvant abemaciclib, of which 43.5% and 56.4% of patients were premenopausal and postmenopausal, respectively.
Disclosures: This study was funded by Eli Lilly and Company. Four authors declared being employees and shareholders of Eli Lilly. The other authors reported ties with several sources, including Eli Lilly.
Source: Paluch-Shimon S et al, on behalf of the monarchE investigators. Efficacy and safety results by menopausal status in monarchE: adjuvant abemaciclib combined with endocrine therapy in patients with HR+, HER2−, node-positive, high-risk early breast cancer. Ther Adv Med Oncol. 2023 (Feb 3). Doi: 10.1177/17588359231151840
Adding ET to dual anti-HER2 targeted therapy beneficial in HER2+/HR+ metastatic BC
Key clinical point: In patients with hormone receptor-positive (HR+)/human epidermal growth factor receptor 2-positive (HER2+) metastatic breast cancer (BC), adding endocrine therapy (ET) to dual anti-HER2 targeted therapy after chemotherapy improved progression-free survival (PFS) without increasing the rate of adverse events.
Major finding: There was a significant improvement in 5-year PFS with vs without the addition of ET (hazard ratio 0.59; P = .031). Nausea and vomiting were more common in patients who did not vs did receive ET (21% vs 7%; P = .010).
Study details: This study analyzed the real-world data of 147 patients with HER2+/HR+ metastatic BC from a prospective registry who received first-line chemotherapy plus trastuzumab and pertuzumab with (n = 91) or without (n = 56) concurrent ET.
Disclosures: This study did not receive any funding. Some authors declared serving on the advisory board for or receiving research funding, speaker honoraria, or travel grants from several sources.
Source: Loft M et al. Addition of endocrine therapy to dual anti-HER2 targeted therapy in initial treatment of HER2 + /HR + metastatic breast cancer. Breast Cancer Res Treat. 2023;198:67-74 (Jan 9). Doi: 10.1007/s10549-022-06856-1
Key clinical point: In patients with hormone receptor-positive (HR+)/human epidermal growth factor receptor 2-positive (HER2+) metastatic breast cancer (BC), adding endocrine therapy (ET) to dual anti-HER2 targeted therapy after chemotherapy improved progression-free survival (PFS) without increasing the rate of adverse events.
Major finding: There was a significant improvement in 5-year PFS with vs without the addition of ET (hazard ratio 0.59; P = .031). Nausea and vomiting were more common in patients who did not vs did receive ET (21% vs 7%; P = .010).
Study details: This study analyzed the real-world data of 147 patients with HER2+/HR+ metastatic BC from a prospective registry who received first-line chemotherapy plus trastuzumab and pertuzumab with (n = 91) or without (n = 56) concurrent ET.
Disclosures: This study did not receive any funding. Some authors declared serving on the advisory board for or receiving research funding, speaker honoraria, or travel grants from several sources.
Source: Loft M et al. Addition of endocrine therapy to dual anti-HER2 targeted therapy in initial treatment of HER2 + /HR + metastatic breast cancer. Breast Cancer Res Treat. 2023;198:67-74 (Jan 9). Doi: 10.1007/s10549-022-06856-1
Key clinical point: In patients with hormone receptor-positive (HR+)/human epidermal growth factor receptor 2-positive (HER2+) metastatic breast cancer (BC), adding endocrine therapy (ET) to dual anti-HER2 targeted therapy after chemotherapy improved progression-free survival (PFS) without increasing the rate of adverse events.
Major finding: There was a significant improvement in 5-year PFS with vs without the addition of ET (hazard ratio 0.59; P = .031). Nausea and vomiting were more common in patients who did not vs did receive ET (21% vs 7%; P = .010).
Study details: This study analyzed the real-world data of 147 patients with HER2+/HR+ metastatic BC from a prospective registry who received first-line chemotherapy plus trastuzumab and pertuzumab with (n = 91) or without (n = 56) concurrent ET.
Disclosures: This study did not receive any funding. Some authors declared serving on the advisory board for or receiving research funding, speaker honoraria, or travel grants from several sources.
Source: Loft M et al. Addition of endocrine therapy to dual anti-HER2 targeted therapy in initial treatment of HER2 + /HR + metastatic breast cancer. Breast Cancer Res Treat. 2023;198:67-74 (Jan 9). Doi: 10.1007/s10549-022-06856-1
SPIO can help avoid upfront sentinel lymph node dissection in ductal carcinoma in situ of the breast
Key clinical point: Marking sentinel lymph nodes (SLN) with superparamagnetic iron oxide (SPIO) nanoparticles led to a substantial proportion of patients with ductal carcinoma in situ (DCIS) of the breast avoiding an upfront SLN dissection (SLND).
Major finding: Upfront SLND could be avoided in 78.3% of patients after marking SLN with SPIO nanoparticles. Among patients receiving delayed SLND, the detection rate was significantly better with SPIO vs radioisotope (99Tc), both with (98.2% vs 63.6%) and without the concomitant use of blue dye (92.7% vs 50.9%; both P < .001).
Study details: Findings are from a prospective, multicenter cohort study including 254 patients with DCIS.
Disclosures: This study was funded by the Uppsala University. One author declared serving on an advisory board and receiving institutional grants and speaker honoraria from several sources.
Source: Karakatsanis A et al. Delayed sentinel lymph node dissection in patients with a preoperative diagnosis of ductal cancer in situ by preoperative injection with superparamagnetic iron oxide (SPIO) nanoparticles: The SentiNot study. Ann Surg Oncol. 2023 (Jan 31). Doi: 10.1245/s10434-022-13064-0
Key clinical point: Marking sentinel lymph nodes (SLN) with superparamagnetic iron oxide (SPIO) nanoparticles led to a substantial proportion of patients with ductal carcinoma in situ (DCIS) of the breast avoiding an upfront SLN dissection (SLND).
Major finding: Upfront SLND could be avoided in 78.3% of patients after marking SLN with SPIO nanoparticles. Among patients receiving delayed SLND, the detection rate was significantly better with SPIO vs radioisotope (99Tc), both with (98.2% vs 63.6%) and without the concomitant use of blue dye (92.7% vs 50.9%; both P < .001).
Study details: Findings are from a prospective, multicenter cohort study including 254 patients with DCIS.
Disclosures: This study was funded by the Uppsala University. One author declared serving on an advisory board and receiving institutional grants and speaker honoraria from several sources.
Source: Karakatsanis A et al. Delayed sentinel lymph node dissection in patients with a preoperative diagnosis of ductal cancer in situ by preoperative injection with superparamagnetic iron oxide (SPIO) nanoparticles: The SentiNot study. Ann Surg Oncol. 2023 (Jan 31). Doi: 10.1245/s10434-022-13064-0
Key clinical point: Marking sentinel lymph nodes (SLN) with superparamagnetic iron oxide (SPIO) nanoparticles led to a substantial proportion of patients with ductal carcinoma in situ (DCIS) of the breast avoiding an upfront SLN dissection (SLND).
Major finding: Upfront SLND could be avoided in 78.3% of patients after marking SLN with SPIO nanoparticles. Among patients receiving delayed SLND, the detection rate was significantly better with SPIO vs radioisotope (99Tc), both with (98.2% vs 63.6%) and without the concomitant use of blue dye (92.7% vs 50.9%; both P < .001).
Study details: Findings are from a prospective, multicenter cohort study including 254 patients with DCIS.
Disclosures: This study was funded by the Uppsala University. One author declared serving on an advisory board and receiving institutional grants and speaker honoraria from several sources.
Source: Karakatsanis A et al. Delayed sentinel lymph node dissection in patients with a preoperative diagnosis of ductal cancer in situ by preoperative injection with superparamagnetic iron oxide (SPIO) nanoparticles: The SentiNot study. Ann Surg Oncol. 2023 (Jan 31). Doi: 10.1245/s10434-022-13064-0
Contralateral prophylactic mastectomy offers no survival advantage in TNBC
Key clinical point: Contralateral prophylactic mastectomy (CPM) did not offer any survival benefit to patients with triple-negative breast cancer (TNBC) irrespective of the presence of BRCA mutations.
Major finding: The 5-year overall survival did not significantly improve with vs without CPM in the entire population of patients with TNBC (P = .05) and in the subgroups of patients with (P = .35) and without (P = .12) BRCA mutations.
Study details: Findings are from a multi-institutional database study including 796 patients with TNBC, of which 15.5% of patients underwent CPM.
Disclosures: This study did not receive any funding. The authors declared no conflicts of interest.
Source: Fasano GA et al. Survival outcomes in women with unilateral, triple-negative, breast cancer correlated with contralateral prophylactic mastectomy. Ann Surg Oncol. 2023 (Jan 21). Doi: 10.1245/s10434-022-13056-0
Key clinical point: Contralateral prophylactic mastectomy (CPM) did not offer any survival benefit to patients with triple-negative breast cancer (TNBC) irrespective of the presence of BRCA mutations.
Major finding: The 5-year overall survival did not significantly improve with vs without CPM in the entire population of patients with TNBC (P = .05) and in the subgroups of patients with (P = .35) and without (P = .12) BRCA mutations.
Study details: Findings are from a multi-institutional database study including 796 patients with TNBC, of which 15.5% of patients underwent CPM.
Disclosures: This study did not receive any funding. The authors declared no conflicts of interest.
Source: Fasano GA et al. Survival outcomes in women with unilateral, triple-negative, breast cancer correlated with contralateral prophylactic mastectomy. Ann Surg Oncol. 2023 (Jan 21). Doi: 10.1245/s10434-022-13056-0
Key clinical point: Contralateral prophylactic mastectomy (CPM) did not offer any survival benefit to patients with triple-negative breast cancer (TNBC) irrespective of the presence of BRCA mutations.
Major finding: The 5-year overall survival did not significantly improve with vs without CPM in the entire population of patients with TNBC (P = .05) and in the subgroups of patients with (P = .35) and without (P = .12) BRCA mutations.
Study details: Findings are from a multi-institutional database study including 796 patients with TNBC, of which 15.5% of patients underwent CPM.
Disclosures: This study did not receive any funding. The authors declared no conflicts of interest.
Source: Fasano GA et al. Survival outcomes in women with unilateral, triple-negative, breast cancer correlated with contralateral prophylactic mastectomy. Ann Surg Oncol. 2023 (Jan 21). Doi: 10.1245/s10434-022-13056-0
Adjuvant AI yields better survival outcomes than tamoxifen or tamoxifen+AI in HR+/HER2+ BC
Key clinical point: Adjuvant endocrine therapy (ET) with an aromatase inhibitor (AI) was associated with better disease-free survival (DFS) than tamoxifen-only or tamoxifen+AI in patients with hormone receptor-positive (HR+)/human epidermal growth factor receptor2-positive (HER2+) breast cancer (BC).
Major finding: Adjuvant ET with tamoxifen or tamoxifen+AI was associated with significantly worse DFS rates than AI in the entire population of women with HR+/HER2+ BC (hazard ratio 1.64; P = .025), with gonadotropin-releasing hormone being associated with improved DFS rates in premenopausal patients aged ≤45 years (hazard ratio 0.41; P = .023).
Study details: Findings are from a post hoc analysis of the ShortHER trial including 784 patients with HR+/HER2+ early BC who received adjuvant anthracycline/taxane-based chemotherapy plus trastuzumab.
Disclosures: This study was supported by Agenzia Italiana del Farmaco and other sources. Some authors declared receiving personal fees from several sources.
Source: Dieci MV et al. Type of adjuvant endocrine therapy and disease-free survival in patients with early HR-positive/HER2-positive BC: Analysis from the phase III randomized ShortHER trial. NPJ Breast Cancer. 2023;9(1):6 (Feb 4). Doi: 10.1038/s41523-023-00509-2
Key clinical point: Adjuvant endocrine therapy (ET) with an aromatase inhibitor (AI) was associated with better disease-free survival (DFS) than tamoxifen-only or tamoxifen+AI in patients with hormone receptor-positive (HR+)/human epidermal growth factor receptor2-positive (HER2+) breast cancer (BC).
Major finding: Adjuvant ET with tamoxifen or tamoxifen+AI was associated with significantly worse DFS rates than AI in the entire population of women with HR+/HER2+ BC (hazard ratio 1.64; P = .025), with gonadotropin-releasing hormone being associated with improved DFS rates in premenopausal patients aged ≤45 years (hazard ratio 0.41; P = .023).
Study details: Findings are from a post hoc analysis of the ShortHER trial including 784 patients with HR+/HER2+ early BC who received adjuvant anthracycline/taxane-based chemotherapy plus trastuzumab.
Disclosures: This study was supported by Agenzia Italiana del Farmaco and other sources. Some authors declared receiving personal fees from several sources.
Source: Dieci MV et al. Type of adjuvant endocrine therapy and disease-free survival in patients with early HR-positive/HER2-positive BC: Analysis from the phase III randomized ShortHER trial. NPJ Breast Cancer. 2023;9(1):6 (Feb 4). Doi: 10.1038/s41523-023-00509-2
Key clinical point: Adjuvant endocrine therapy (ET) with an aromatase inhibitor (AI) was associated with better disease-free survival (DFS) than tamoxifen-only or tamoxifen+AI in patients with hormone receptor-positive (HR+)/human epidermal growth factor receptor2-positive (HER2+) breast cancer (BC).
Major finding: Adjuvant ET with tamoxifen or tamoxifen+AI was associated with significantly worse DFS rates than AI in the entire population of women with HR+/HER2+ BC (hazard ratio 1.64; P = .025), with gonadotropin-releasing hormone being associated with improved DFS rates in premenopausal patients aged ≤45 years (hazard ratio 0.41; P = .023).
Study details: Findings are from a post hoc analysis of the ShortHER trial including 784 patients with HR+/HER2+ early BC who received adjuvant anthracycline/taxane-based chemotherapy plus trastuzumab.
Disclosures: This study was supported by Agenzia Italiana del Farmaco and other sources. Some authors declared receiving personal fees from several sources.
Source: Dieci MV et al. Type of adjuvant endocrine therapy and disease-free survival in patients with early HR-positive/HER2-positive BC: Analysis from the phase III randomized ShortHER trial. NPJ Breast Cancer. 2023;9(1):6 (Feb 4). Doi: 10.1038/s41523-023-00509-2
Real-world survival benefit with CDK4/6 inhibitors in MBC
(MBC).
Three years after starting first-line treatment, overall survival rates were 49% with endocrine therapy alone versus 73% with endocrine therapy plus a CDK4/6 inhibitor – findings largely consistent with clinical trial data.
Treatment with endocrine therapy alone “should have a limited role in early lines of therapy for patients with HR-positive/HER2-negative MBC,” the researchers concluded.
The study was published online in the journal Cancer.
The Food and Drug Administration has approved the CDK4/6 inhibitors palbociclib, abemaciclib, and ribociclib for HR-positive/HER2-negative MBC in first and advanced therapy lines. The approval was based on phase 3, randomized, controlled trial data.
Yet gaps in evidence remain regarding survival outcomes in real-world settings for patients starting treatment with a CDK4/6 inhibitor, particularly for patients aged 65 and older, who typically aren’t included in randomized clinical trials.
To address these gaps, Ravi Goyal, PhD, with the University of Houston, and colleagues conducted a retrospective cohort study using the Survey Epidemiology and End Results Medicare database.
Dr. Goyal and coauthors identified 630 Medicare patients with HR-positive/HER2-negative MBC for whom first-line treatment was initiated within 12 months of diagnosis. Patients received either endocrine therapy alone (461 patients) or endocrine therapy plus a CDK4/6 inhibitor (169 patients), most commonly palbociclib.
The median duration of follow-up from the start of treatment was 24 months in the endocrine therapy–alone group and 30 months in the combination therapy group.
In a Kaplan-Meier analysis, the overall survival rate at 3 years after starting first-line treatment was 73% for the combination therapy group versus49% for the endocrine therapy group (P < .0001). Median overall survival from first-line therapy was not estimable in the endocrine therapy plus CDK4/6 inhibitor group; it was 34.8 months in the endocrine therapy–only group.
In multivariable Cox regression models, first-line dual therapy was independently associated with 41% lower rate of death in comparison with endocrine therapy alone (adjusted hazard ratio, 0.59).
Dr. Goyal and colleagues also performed a separate analysis of 206 patients for whom treatment was initiated in the second line; 88 received endocrine therapy alone, and 118 received endocrine therapy plus CDK4/6 inhibitor therapy.
In this setting, a similar benefit of dual therapy was observed. The 3-year overall survival rate was 68% for the combination group versus 50% for endocrine therapy alone (P = .0051). Median overall survival with second-line therapy was not estimable for the combination group; it was 30.9 months for the endocrine therapy group.
In the second line, multivariable Cox regression analysis showed that combination therapy was associated with a nearly 58% lower rate of death in comparison with endocrine therapy alone (aHR, 0.42).
The coauthors of an editorial in Cancer explain that the combination of a CDK4/6 inhibitor and endocrine therapy has become the “preferred first-line approach” in the management of HR-positive/HER2-negative MBC.
Dario Trapani, MD, and Erica Mayer, MD, with Dana-Farber Cancer Institute, Boston, noted that an overall survival benefit of similar magnitude from endocrine therapy plus a CDK4/6 inhibitor has also been reported in a recent real-world study of palbociclib and letrozole.
The work of Dr. Goyal and colleagues provides “confirmatory real-world evidence observed in clinical trials for endocrine sensitive, de novo, metastatic [breast cancer] in an older population,” Dr. Trapani and Dr. Mayer wrote.
The study had no specific funding. The authors and Dr. Trapani disclosed no relevant financial relaitonships. Dr. Mayer has consulted for Lilly, Novartis, Gilead, AstraZeneca, and Diaccurate.
A version of this article first appeared on Medscape.com.
(MBC).
Three years after starting first-line treatment, overall survival rates were 49% with endocrine therapy alone versus 73% with endocrine therapy plus a CDK4/6 inhibitor – findings largely consistent with clinical trial data.
Treatment with endocrine therapy alone “should have a limited role in early lines of therapy for patients with HR-positive/HER2-negative MBC,” the researchers concluded.
The study was published online in the journal Cancer.
The Food and Drug Administration has approved the CDK4/6 inhibitors palbociclib, abemaciclib, and ribociclib for HR-positive/HER2-negative MBC in first and advanced therapy lines. The approval was based on phase 3, randomized, controlled trial data.
Yet gaps in evidence remain regarding survival outcomes in real-world settings for patients starting treatment with a CDK4/6 inhibitor, particularly for patients aged 65 and older, who typically aren’t included in randomized clinical trials.
To address these gaps, Ravi Goyal, PhD, with the University of Houston, and colleagues conducted a retrospective cohort study using the Survey Epidemiology and End Results Medicare database.
Dr. Goyal and coauthors identified 630 Medicare patients with HR-positive/HER2-negative MBC for whom first-line treatment was initiated within 12 months of diagnosis. Patients received either endocrine therapy alone (461 patients) or endocrine therapy plus a CDK4/6 inhibitor (169 patients), most commonly palbociclib.
The median duration of follow-up from the start of treatment was 24 months in the endocrine therapy–alone group and 30 months in the combination therapy group.
In a Kaplan-Meier analysis, the overall survival rate at 3 years after starting first-line treatment was 73% for the combination therapy group versus49% for the endocrine therapy group (P < .0001). Median overall survival from first-line therapy was not estimable in the endocrine therapy plus CDK4/6 inhibitor group; it was 34.8 months in the endocrine therapy–only group.
In multivariable Cox regression models, first-line dual therapy was independently associated with 41% lower rate of death in comparison with endocrine therapy alone (adjusted hazard ratio, 0.59).
Dr. Goyal and colleagues also performed a separate analysis of 206 patients for whom treatment was initiated in the second line; 88 received endocrine therapy alone, and 118 received endocrine therapy plus CDK4/6 inhibitor therapy.
In this setting, a similar benefit of dual therapy was observed. The 3-year overall survival rate was 68% for the combination group versus 50% for endocrine therapy alone (P = .0051). Median overall survival with second-line therapy was not estimable for the combination group; it was 30.9 months for the endocrine therapy group.
In the second line, multivariable Cox regression analysis showed that combination therapy was associated with a nearly 58% lower rate of death in comparison with endocrine therapy alone (aHR, 0.42).
The coauthors of an editorial in Cancer explain that the combination of a CDK4/6 inhibitor and endocrine therapy has become the “preferred first-line approach” in the management of HR-positive/HER2-negative MBC.
Dario Trapani, MD, and Erica Mayer, MD, with Dana-Farber Cancer Institute, Boston, noted that an overall survival benefit of similar magnitude from endocrine therapy plus a CDK4/6 inhibitor has also been reported in a recent real-world study of palbociclib and letrozole.
The work of Dr. Goyal and colleagues provides “confirmatory real-world evidence observed in clinical trials for endocrine sensitive, de novo, metastatic [breast cancer] in an older population,” Dr. Trapani and Dr. Mayer wrote.
The study had no specific funding. The authors and Dr. Trapani disclosed no relevant financial relaitonships. Dr. Mayer has consulted for Lilly, Novartis, Gilead, AstraZeneca, and Diaccurate.
A version of this article first appeared on Medscape.com.
(MBC).
Three years after starting first-line treatment, overall survival rates were 49% with endocrine therapy alone versus 73% with endocrine therapy plus a CDK4/6 inhibitor – findings largely consistent with clinical trial data.
Treatment with endocrine therapy alone “should have a limited role in early lines of therapy for patients with HR-positive/HER2-negative MBC,” the researchers concluded.
The study was published online in the journal Cancer.
The Food and Drug Administration has approved the CDK4/6 inhibitors palbociclib, abemaciclib, and ribociclib for HR-positive/HER2-negative MBC in first and advanced therapy lines. The approval was based on phase 3, randomized, controlled trial data.
Yet gaps in evidence remain regarding survival outcomes in real-world settings for patients starting treatment with a CDK4/6 inhibitor, particularly for patients aged 65 and older, who typically aren’t included in randomized clinical trials.
To address these gaps, Ravi Goyal, PhD, with the University of Houston, and colleagues conducted a retrospective cohort study using the Survey Epidemiology and End Results Medicare database.
Dr. Goyal and coauthors identified 630 Medicare patients with HR-positive/HER2-negative MBC for whom first-line treatment was initiated within 12 months of diagnosis. Patients received either endocrine therapy alone (461 patients) or endocrine therapy plus a CDK4/6 inhibitor (169 patients), most commonly palbociclib.
The median duration of follow-up from the start of treatment was 24 months in the endocrine therapy–alone group and 30 months in the combination therapy group.
In a Kaplan-Meier analysis, the overall survival rate at 3 years after starting first-line treatment was 73% for the combination therapy group versus49% for the endocrine therapy group (P < .0001). Median overall survival from first-line therapy was not estimable in the endocrine therapy plus CDK4/6 inhibitor group; it was 34.8 months in the endocrine therapy–only group.
In multivariable Cox regression models, first-line dual therapy was independently associated with 41% lower rate of death in comparison with endocrine therapy alone (adjusted hazard ratio, 0.59).
Dr. Goyal and colleagues also performed a separate analysis of 206 patients for whom treatment was initiated in the second line; 88 received endocrine therapy alone, and 118 received endocrine therapy plus CDK4/6 inhibitor therapy.
In this setting, a similar benefit of dual therapy was observed. The 3-year overall survival rate was 68% for the combination group versus 50% for endocrine therapy alone (P = .0051). Median overall survival with second-line therapy was not estimable for the combination group; it was 30.9 months for the endocrine therapy group.
In the second line, multivariable Cox regression analysis showed that combination therapy was associated with a nearly 58% lower rate of death in comparison with endocrine therapy alone (aHR, 0.42).
The coauthors of an editorial in Cancer explain that the combination of a CDK4/6 inhibitor and endocrine therapy has become the “preferred first-line approach” in the management of HR-positive/HER2-negative MBC.
Dario Trapani, MD, and Erica Mayer, MD, with Dana-Farber Cancer Institute, Boston, noted that an overall survival benefit of similar magnitude from endocrine therapy plus a CDK4/6 inhibitor has also been reported in a recent real-world study of palbociclib and letrozole.
The work of Dr. Goyal and colleagues provides “confirmatory real-world evidence observed in clinical trials for endocrine sensitive, de novo, metastatic [breast cancer] in an older population,” Dr. Trapani and Dr. Mayer wrote.
The study had no specific funding. The authors and Dr. Trapani disclosed no relevant financial relaitonships. Dr. Mayer has consulted for Lilly, Novartis, Gilead, AstraZeneca, and Diaccurate.
A version of this article first appeared on Medscape.com.
FROM CANCER
New cancer screen, same issues: Physicians confront Galleri test
In January 2022, Anthony Arenz, a 51-year-old living in Mesa, Ariz., breathed a small sigh of relief.
The Galleri blood test, which screens for 50 types of cancer, hadn’t detected any positive signs.
It would be welcome news to anyone but especially to a firefighter with a 9% greater risk of developing cancer and a 14% greater risk of dying from it than the average person. The Mesa unit had lost two servicemen to cancer in the past 3 years. Both were more than a decade younger than Mr. Arenz.
When the city of Mesa offered additional free screening – including a full-body MRI – to firefighters over 50, Mr. Arenz initially shrugged it off. With a negative Galleri test in hand, he didn’t want to spend more time dwelling on it.
Still, he began to feel a creeping guilt for skipping a test that many of his fallen colleagues hadn’t been offered. He tried to soothe his anxiety with research. A look through the company’s website didn’t set him at ease. According to Grail Bio, a test result of “no cancer signal detected” does not rule out cancer.
Mr. Arenz booked his free MRI.
The results left him heavy: stage I kidney cancer. The Galleri test had missed it.
Mr. Arenz received his free Galleri test through a cancer screening program funded by the city of Mesa. The program is housed at Vincere Cancer Center in Scottsdale, Ariz. Under the leadership of radiation oncologist and Vincere co-owner Vershalee Shukla, MD, the program currently screens first responders in more than 10 Arizona cities at no cost to them.
Vincere began using Galleri shortly after the test launched for consumers in June 2021. Since then, the first responder program has become the largest commercial user of the test in North America.
The Galleri test, which has not yet been approved by the Food and Drug Administration, is so new that few know what incorrect results look like in practice and how often they might occur.
After running the test on about 2,000 servicemen and servicewomen, Dr. Shukla can offer some insight about the test’s real-world value in a high-risk population.
“Cancer screening is a very complicated issue,” Dr. Shukla said in an interview. “Being honest, the tests are good but are not ready yet [for wider use].”
Mr. Arenz was not the only firefighter who got a surprise after taking a Galleri test.
In nearby Phoenix, 51-year-old firefighter Mike Curtis knew his risk for cancer was high, but he wasn’t that worried. Mr. Curtis had been running into fires since he was 17. His dad, also a firefighter, had died of cancer at age 58.
Mr. Curtis had taken the Vincere Cancer Center up on every free screening service since the program began in late 2018 – well before Dr. Shukla started using Galleri in 2021. His most recent lung CT was clear. But he underwent the Galleri test just to stay vigilant.
His result was a shock. The test detected signs of cancer.
Mr. Curtis decided to tell no one, not even his wife. He’d bear the bad news alone until he was certain.
Dr. Shukla, however, immediately doubted the blood test result. She expedited several follow-up tests. One week, a PET, and CT of the abdomen and pelvis later, her hunch was confirmed. The Galleri test result was wrong, Mr. Curtis did not have cancer.
The price of his peace of mind: an extensive workup with a $4,000 price tag. Fortunately, the bill was covered by the screening program.
Overall, in just over 18 months of using the blood test, Dr. Shukla has only encountered 1 other false positive out of about 2,000 Galleri results.
She also discovered two positive signals for cancer using Galleri that were confirmed with follow-up tests. One was a chordoma, a rare type of bone cancer, and the other was a squamous cell carcinoma of the head and neck. The Galleri test caught both remarkably early, in time for treatment.
For Dr. Shukla, however, false negatives were particularly “horrible.” Mr. Arenz’s was just 1 of 28 cancers that the blood test missed. And because 500 negative tests are yet to be validated, the 28 false negatives may be an underestimate.
In her experience, the binary test result – a simple positive or negative cancer signal – is an oversimplification of risk, she said. It “gives a false perception that you have cancer or you don’t,” although the test itself is not definitive.
Grail senior medical director Whitney Jones, MD, agreed that the test is not meant to be a stand-alone screening test for cancer. The purpose of the Galleri test is to “complement other screenings, not replace them,” Dr. Jones told this news organization.
According to an analysis of Galleri data and Dr. Shukla’s experience, the test’s specificity was over 99%. That means the test successfully minimizes false positives.
But the test’s sensitivity was much lower. From data from first responders, Dr. Shukla determined the sensitivity to be 6.7%. That means the test misses about 93 of every 100 cancers. According to Grail’s latest data from more than 6,300 people older than 50, the test’s sensitivity was 29%.
Specificity and sensitivity are metrics used to credential a test and establish confidence in its ability to detect the target disease. A test with high specificity can correctly identify patients who do not have the condition in question, while a test with high sensitivity can correctly identify patients who do have the disease. But there are trade-offs between sensitivity and specificity. One value is increased at the expense of the other.
It’s normal for a cancer screening test to prioritize specificity, according to Aparna Parikh, MD, an oncologist at Mass General Cancer Center in Boston. In a test like Galleri, which is meant to be an adjunct to other screening modalities, “at least we are seeing a good specificity, which is important, because we don’t want false positives, where the downstream impact on the patient can be high.”
Overall, Dr. Jones said, Grail Bio’s aim is to build a test that’s sensitive enough to catch the most dangerous cancers without inundating the healthcare system with false positives. In addition, Dr. Jones explained, sensitivity varies by cancer type. It tends to be lower for cancers for which other screening modalities are available, as well as for earlier-stage disease.
However, the Galleri sensitivity values are “a little bit scary,” said Ji-Hyun Lee, DrPH, professor of biostatistics at the University of Florida and director of the division of quantitative sciences at the University of Florida Health Cancer Center, both in Gainesville. Dr. Lee, who is not affiliated with Grail, reviewed the company’s publicly available data as well as Dr. Shukla’s data at the request of this news organization.
While there’s no definitive threshold for sensitivity, miss rates as high as 93% and 71% “provide little confidence in the [accuracy of the] test,” Dr. Lee said.
Positive and negative predictive values, however, are more clinically relevant measures of a screening test. These numbers indicate how likely it is that a patient’s results are true and therefore how worried they should be about a positive result and how much they should trust a negative result.
Galleri’s data in the over-50 population and Dr. Shukla’s in first responders suggest the test’s negative predictive value is very high – 98.6% and 98.1%, respectively – which means most people can trust a negative test result.
The positive predictive value, however, was less straightforward. In first responders, Dr. Shukla found that only half of positive Galleri tests were confirmed cases of cancer. And an analysis of Grail’s data found that only 38% of positive Galleri tests – 35 of 92 tests – represented a validated cancer diagnosis.
“In a clinical setting, positive predictive value is more usable for decision-making for the patient,” said Dr. Lee. “Positive predictive value isn’t always high, because everything doesn’t always transfer perfectly to the clinic.” But in the general population, if only 38% of patients with positive Galleri results truly have cancer, the test is “not quite useful to make a decision for the patient or the providers.”
Galleri may also be a costly prospect for patients, no matter the result, cautioned Electra Paskett, PhD, an epidemiologist and cancer screening expert at Ohio State University, Columbus. A positive Galleri test leads to a cascade of follow-up diagnostic tests, which payers may not cover. For a negative result, Galleri recommends that the patient undergo screening again in a year, at an annual cost of $950 plus the cost of any follow-up testing when Galleri does pick something up.
“If a provider wants to offer the Galleri test, all those things need to be made abundantly clear, in my opinion,” Dr. Paskett said.
Following the negative Galleri test, Mr. Arenz’s cancer didn’t slip through the cracks because he received other advanced imaging free of charge. But whether all doctors will go to such lengths to back up Galleri results, even for patients with negative results, is unknown.
A negative result can give patients “a huge false sense of security,” said Dr. Shukla. And if a test is positive, the workup isn’t simple. Chasing cancer, especially one that’s not really there, can be nerve-wracking and expensive.
The question, then, is why perform the Galleri test at all if results require so much validation?
Dr. Parikh explained that a high-risk group such as firefighters represents an ideal-use case for Galleri and other liquid biopsy tests. But she noted that she would be “wary of the ability of the system to manage this test en masse” were the test to be used more widely in the general population.
Dr. Shukla said it’s less about the results she’s getting today and more about making the test more effective for her patients in the future. First responders need a test such as this that can quickly identify multiple cancers. However, to improve the test, Grail needs more data from this high-risk population. That’s what she’s after.
Mr. Curtis doesn’t regret taking the Galleri test. The emotional toll of thinking he had cancer for a few days wasn’t too high a price, in his opinion. It’s part of cancer screening. But he acknowledged that it would have been a much more burdensome experience had he’d been financially responsible for the workup or if he hadn’t had Dr. Shukla to manage his case from start to finish.
Because it was free, Mr. Arenz doesn’t regret undergoing the Galleri test either. But he tells his coworkers to check the site, do their research, and get more screening.
“Any medical center that’s just doing this one test, you just have to be careful,” Dr. Shukla said. “It’s not that easy.”
A version of this article first appeared on Medscape.com.
In January 2022, Anthony Arenz, a 51-year-old living in Mesa, Ariz., breathed a small sigh of relief.
The Galleri blood test, which screens for 50 types of cancer, hadn’t detected any positive signs.
It would be welcome news to anyone but especially to a firefighter with a 9% greater risk of developing cancer and a 14% greater risk of dying from it than the average person. The Mesa unit had lost two servicemen to cancer in the past 3 years. Both were more than a decade younger than Mr. Arenz.
When the city of Mesa offered additional free screening – including a full-body MRI – to firefighters over 50, Mr. Arenz initially shrugged it off. With a negative Galleri test in hand, he didn’t want to spend more time dwelling on it.
Still, he began to feel a creeping guilt for skipping a test that many of his fallen colleagues hadn’t been offered. He tried to soothe his anxiety with research. A look through the company’s website didn’t set him at ease. According to Grail Bio, a test result of “no cancer signal detected” does not rule out cancer.
Mr. Arenz booked his free MRI.
The results left him heavy: stage I kidney cancer. The Galleri test had missed it.
Mr. Arenz received his free Galleri test through a cancer screening program funded by the city of Mesa. The program is housed at Vincere Cancer Center in Scottsdale, Ariz. Under the leadership of radiation oncologist and Vincere co-owner Vershalee Shukla, MD, the program currently screens first responders in more than 10 Arizona cities at no cost to them.
Vincere began using Galleri shortly after the test launched for consumers in June 2021. Since then, the first responder program has become the largest commercial user of the test in North America.
The Galleri test, which has not yet been approved by the Food and Drug Administration, is so new that few know what incorrect results look like in practice and how often they might occur.
After running the test on about 2,000 servicemen and servicewomen, Dr. Shukla can offer some insight about the test’s real-world value in a high-risk population.
“Cancer screening is a very complicated issue,” Dr. Shukla said in an interview. “Being honest, the tests are good but are not ready yet [for wider use].”
Mr. Arenz was not the only firefighter who got a surprise after taking a Galleri test.
In nearby Phoenix, 51-year-old firefighter Mike Curtis knew his risk for cancer was high, but he wasn’t that worried. Mr. Curtis had been running into fires since he was 17. His dad, also a firefighter, had died of cancer at age 58.
Mr. Curtis had taken the Vincere Cancer Center up on every free screening service since the program began in late 2018 – well before Dr. Shukla started using Galleri in 2021. His most recent lung CT was clear. But he underwent the Galleri test just to stay vigilant.
His result was a shock. The test detected signs of cancer.
Mr. Curtis decided to tell no one, not even his wife. He’d bear the bad news alone until he was certain.
Dr. Shukla, however, immediately doubted the blood test result. She expedited several follow-up tests. One week, a PET, and CT of the abdomen and pelvis later, her hunch was confirmed. The Galleri test result was wrong, Mr. Curtis did not have cancer.
The price of his peace of mind: an extensive workup with a $4,000 price tag. Fortunately, the bill was covered by the screening program.
Overall, in just over 18 months of using the blood test, Dr. Shukla has only encountered 1 other false positive out of about 2,000 Galleri results.
She also discovered two positive signals for cancer using Galleri that were confirmed with follow-up tests. One was a chordoma, a rare type of bone cancer, and the other was a squamous cell carcinoma of the head and neck. The Galleri test caught both remarkably early, in time for treatment.
For Dr. Shukla, however, false negatives were particularly “horrible.” Mr. Arenz’s was just 1 of 28 cancers that the blood test missed. And because 500 negative tests are yet to be validated, the 28 false negatives may be an underestimate.
In her experience, the binary test result – a simple positive or negative cancer signal – is an oversimplification of risk, she said. It “gives a false perception that you have cancer or you don’t,” although the test itself is not definitive.
Grail senior medical director Whitney Jones, MD, agreed that the test is not meant to be a stand-alone screening test for cancer. The purpose of the Galleri test is to “complement other screenings, not replace them,” Dr. Jones told this news organization.
According to an analysis of Galleri data and Dr. Shukla’s experience, the test’s specificity was over 99%. That means the test successfully minimizes false positives.
But the test’s sensitivity was much lower. From data from first responders, Dr. Shukla determined the sensitivity to be 6.7%. That means the test misses about 93 of every 100 cancers. According to Grail’s latest data from more than 6,300 people older than 50, the test’s sensitivity was 29%.
Specificity and sensitivity are metrics used to credential a test and establish confidence in its ability to detect the target disease. A test with high specificity can correctly identify patients who do not have the condition in question, while a test with high sensitivity can correctly identify patients who do have the disease. But there are trade-offs between sensitivity and specificity. One value is increased at the expense of the other.
It’s normal for a cancer screening test to prioritize specificity, according to Aparna Parikh, MD, an oncologist at Mass General Cancer Center in Boston. In a test like Galleri, which is meant to be an adjunct to other screening modalities, “at least we are seeing a good specificity, which is important, because we don’t want false positives, where the downstream impact on the patient can be high.”
Overall, Dr. Jones said, Grail Bio’s aim is to build a test that’s sensitive enough to catch the most dangerous cancers without inundating the healthcare system with false positives. In addition, Dr. Jones explained, sensitivity varies by cancer type. It tends to be lower for cancers for which other screening modalities are available, as well as for earlier-stage disease.
However, the Galleri sensitivity values are “a little bit scary,” said Ji-Hyun Lee, DrPH, professor of biostatistics at the University of Florida and director of the division of quantitative sciences at the University of Florida Health Cancer Center, both in Gainesville. Dr. Lee, who is not affiliated with Grail, reviewed the company’s publicly available data as well as Dr. Shukla’s data at the request of this news organization.
While there’s no definitive threshold for sensitivity, miss rates as high as 93% and 71% “provide little confidence in the [accuracy of the] test,” Dr. Lee said.
Positive and negative predictive values, however, are more clinically relevant measures of a screening test. These numbers indicate how likely it is that a patient’s results are true and therefore how worried they should be about a positive result and how much they should trust a negative result.
Galleri’s data in the over-50 population and Dr. Shukla’s in first responders suggest the test’s negative predictive value is very high – 98.6% and 98.1%, respectively – which means most people can trust a negative test result.
The positive predictive value, however, was less straightforward. In first responders, Dr. Shukla found that only half of positive Galleri tests were confirmed cases of cancer. And an analysis of Grail’s data found that only 38% of positive Galleri tests – 35 of 92 tests – represented a validated cancer diagnosis.
“In a clinical setting, positive predictive value is more usable for decision-making for the patient,” said Dr. Lee. “Positive predictive value isn’t always high, because everything doesn’t always transfer perfectly to the clinic.” But in the general population, if only 38% of patients with positive Galleri results truly have cancer, the test is “not quite useful to make a decision for the patient or the providers.”
Galleri may also be a costly prospect for patients, no matter the result, cautioned Electra Paskett, PhD, an epidemiologist and cancer screening expert at Ohio State University, Columbus. A positive Galleri test leads to a cascade of follow-up diagnostic tests, which payers may not cover. For a negative result, Galleri recommends that the patient undergo screening again in a year, at an annual cost of $950 plus the cost of any follow-up testing when Galleri does pick something up.
“If a provider wants to offer the Galleri test, all those things need to be made abundantly clear, in my opinion,” Dr. Paskett said.
Following the negative Galleri test, Mr. Arenz’s cancer didn’t slip through the cracks because he received other advanced imaging free of charge. But whether all doctors will go to such lengths to back up Galleri results, even for patients with negative results, is unknown.
A negative result can give patients “a huge false sense of security,” said Dr. Shukla. And if a test is positive, the workup isn’t simple. Chasing cancer, especially one that’s not really there, can be nerve-wracking and expensive.
The question, then, is why perform the Galleri test at all if results require so much validation?
Dr. Parikh explained that a high-risk group such as firefighters represents an ideal-use case for Galleri and other liquid biopsy tests. But she noted that she would be “wary of the ability of the system to manage this test en masse” were the test to be used more widely in the general population.
Dr. Shukla said it’s less about the results she’s getting today and more about making the test more effective for her patients in the future. First responders need a test such as this that can quickly identify multiple cancers. However, to improve the test, Grail needs more data from this high-risk population. That’s what she’s after.
Mr. Curtis doesn’t regret taking the Galleri test. The emotional toll of thinking he had cancer for a few days wasn’t too high a price, in his opinion. It’s part of cancer screening. But he acknowledged that it would have been a much more burdensome experience had he’d been financially responsible for the workup or if he hadn’t had Dr. Shukla to manage his case from start to finish.
Because it was free, Mr. Arenz doesn’t regret undergoing the Galleri test either. But he tells his coworkers to check the site, do their research, and get more screening.
“Any medical center that’s just doing this one test, you just have to be careful,” Dr. Shukla said. “It’s not that easy.”
A version of this article first appeared on Medscape.com.
In January 2022, Anthony Arenz, a 51-year-old living in Mesa, Ariz., breathed a small sigh of relief.
The Galleri blood test, which screens for 50 types of cancer, hadn’t detected any positive signs.
It would be welcome news to anyone but especially to a firefighter with a 9% greater risk of developing cancer and a 14% greater risk of dying from it than the average person. The Mesa unit had lost two servicemen to cancer in the past 3 years. Both were more than a decade younger than Mr. Arenz.
When the city of Mesa offered additional free screening – including a full-body MRI – to firefighters over 50, Mr. Arenz initially shrugged it off. With a negative Galleri test in hand, he didn’t want to spend more time dwelling on it.
Still, he began to feel a creeping guilt for skipping a test that many of his fallen colleagues hadn’t been offered. He tried to soothe his anxiety with research. A look through the company’s website didn’t set him at ease. According to Grail Bio, a test result of “no cancer signal detected” does not rule out cancer.
Mr. Arenz booked his free MRI.
The results left him heavy: stage I kidney cancer. The Galleri test had missed it.
Mr. Arenz received his free Galleri test through a cancer screening program funded by the city of Mesa. The program is housed at Vincere Cancer Center in Scottsdale, Ariz. Under the leadership of radiation oncologist and Vincere co-owner Vershalee Shukla, MD, the program currently screens first responders in more than 10 Arizona cities at no cost to them.
Vincere began using Galleri shortly after the test launched for consumers in June 2021. Since then, the first responder program has become the largest commercial user of the test in North America.
The Galleri test, which has not yet been approved by the Food and Drug Administration, is so new that few know what incorrect results look like in practice and how often they might occur.
After running the test on about 2,000 servicemen and servicewomen, Dr. Shukla can offer some insight about the test’s real-world value in a high-risk population.
“Cancer screening is a very complicated issue,” Dr. Shukla said in an interview. “Being honest, the tests are good but are not ready yet [for wider use].”
Mr. Arenz was not the only firefighter who got a surprise after taking a Galleri test.
In nearby Phoenix, 51-year-old firefighter Mike Curtis knew his risk for cancer was high, but he wasn’t that worried. Mr. Curtis had been running into fires since he was 17. His dad, also a firefighter, had died of cancer at age 58.
Mr. Curtis had taken the Vincere Cancer Center up on every free screening service since the program began in late 2018 – well before Dr. Shukla started using Galleri in 2021. His most recent lung CT was clear. But he underwent the Galleri test just to stay vigilant.
His result was a shock. The test detected signs of cancer.
Mr. Curtis decided to tell no one, not even his wife. He’d bear the bad news alone until he was certain.
Dr. Shukla, however, immediately doubted the blood test result. She expedited several follow-up tests. One week, a PET, and CT of the abdomen and pelvis later, her hunch was confirmed. The Galleri test result was wrong, Mr. Curtis did not have cancer.
The price of his peace of mind: an extensive workup with a $4,000 price tag. Fortunately, the bill was covered by the screening program.
Overall, in just over 18 months of using the blood test, Dr. Shukla has only encountered 1 other false positive out of about 2,000 Galleri results.
She also discovered two positive signals for cancer using Galleri that were confirmed with follow-up tests. One was a chordoma, a rare type of bone cancer, and the other was a squamous cell carcinoma of the head and neck. The Galleri test caught both remarkably early, in time for treatment.
For Dr. Shukla, however, false negatives were particularly “horrible.” Mr. Arenz’s was just 1 of 28 cancers that the blood test missed. And because 500 negative tests are yet to be validated, the 28 false negatives may be an underestimate.
In her experience, the binary test result – a simple positive or negative cancer signal – is an oversimplification of risk, she said. It “gives a false perception that you have cancer or you don’t,” although the test itself is not definitive.
Grail senior medical director Whitney Jones, MD, agreed that the test is not meant to be a stand-alone screening test for cancer. The purpose of the Galleri test is to “complement other screenings, not replace them,” Dr. Jones told this news organization.
According to an analysis of Galleri data and Dr. Shukla’s experience, the test’s specificity was over 99%. That means the test successfully minimizes false positives.
But the test’s sensitivity was much lower. From data from first responders, Dr. Shukla determined the sensitivity to be 6.7%. That means the test misses about 93 of every 100 cancers. According to Grail’s latest data from more than 6,300 people older than 50, the test’s sensitivity was 29%.
Specificity and sensitivity are metrics used to credential a test and establish confidence in its ability to detect the target disease. A test with high specificity can correctly identify patients who do not have the condition in question, while a test with high sensitivity can correctly identify patients who do have the disease. But there are trade-offs between sensitivity and specificity. One value is increased at the expense of the other.
It’s normal for a cancer screening test to prioritize specificity, according to Aparna Parikh, MD, an oncologist at Mass General Cancer Center in Boston. In a test like Galleri, which is meant to be an adjunct to other screening modalities, “at least we are seeing a good specificity, which is important, because we don’t want false positives, where the downstream impact on the patient can be high.”
Overall, Dr. Jones said, Grail Bio’s aim is to build a test that’s sensitive enough to catch the most dangerous cancers without inundating the healthcare system with false positives. In addition, Dr. Jones explained, sensitivity varies by cancer type. It tends to be lower for cancers for which other screening modalities are available, as well as for earlier-stage disease.
However, the Galleri sensitivity values are “a little bit scary,” said Ji-Hyun Lee, DrPH, professor of biostatistics at the University of Florida and director of the division of quantitative sciences at the University of Florida Health Cancer Center, both in Gainesville. Dr. Lee, who is not affiliated with Grail, reviewed the company’s publicly available data as well as Dr. Shukla’s data at the request of this news organization.
While there’s no definitive threshold for sensitivity, miss rates as high as 93% and 71% “provide little confidence in the [accuracy of the] test,” Dr. Lee said.
Positive and negative predictive values, however, are more clinically relevant measures of a screening test. These numbers indicate how likely it is that a patient’s results are true and therefore how worried they should be about a positive result and how much they should trust a negative result.
Galleri’s data in the over-50 population and Dr. Shukla’s in first responders suggest the test’s negative predictive value is very high – 98.6% and 98.1%, respectively – which means most people can trust a negative test result.
The positive predictive value, however, was less straightforward. In first responders, Dr. Shukla found that only half of positive Galleri tests were confirmed cases of cancer. And an analysis of Grail’s data found that only 38% of positive Galleri tests – 35 of 92 tests – represented a validated cancer diagnosis.
“In a clinical setting, positive predictive value is more usable for decision-making for the patient,” said Dr. Lee. “Positive predictive value isn’t always high, because everything doesn’t always transfer perfectly to the clinic.” But in the general population, if only 38% of patients with positive Galleri results truly have cancer, the test is “not quite useful to make a decision for the patient or the providers.”
Galleri may also be a costly prospect for patients, no matter the result, cautioned Electra Paskett, PhD, an epidemiologist and cancer screening expert at Ohio State University, Columbus. A positive Galleri test leads to a cascade of follow-up diagnostic tests, which payers may not cover. For a negative result, Galleri recommends that the patient undergo screening again in a year, at an annual cost of $950 plus the cost of any follow-up testing when Galleri does pick something up.
“If a provider wants to offer the Galleri test, all those things need to be made abundantly clear, in my opinion,” Dr. Paskett said.
Following the negative Galleri test, Mr. Arenz’s cancer didn’t slip through the cracks because he received other advanced imaging free of charge. But whether all doctors will go to such lengths to back up Galleri results, even for patients with negative results, is unknown.
A negative result can give patients “a huge false sense of security,” said Dr. Shukla. And if a test is positive, the workup isn’t simple. Chasing cancer, especially one that’s not really there, can be nerve-wracking and expensive.
The question, then, is why perform the Galleri test at all if results require so much validation?
Dr. Parikh explained that a high-risk group such as firefighters represents an ideal-use case for Galleri and other liquid biopsy tests. But she noted that she would be “wary of the ability of the system to manage this test en masse” were the test to be used more widely in the general population.
Dr. Shukla said it’s less about the results she’s getting today and more about making the test more effective for her patients in the future. First responders need a test such as this that can quickly identify multiple cancers. However, to improve the test, Grail needs more data from this high-risk population. That’s what she’s after.
Mr. Curtis doesn’t regret taking the Galleri test. The emotional toll of thinking he had cancer for a few days wasn’t too high a price, in his opinion. It’s part of cancer screening. But he acknowledged that it would have been a much more burdensome experience had he’d been financially responsible for the workup or if he hadn’t had Dr. Shukla to manage his case from start to finish.
Because it was free, Mr. Arenz doesn’t regret undergoing the Galleri test either. But he tells his coworkers to check the site, do their research, and get more screening.
“Any medical center that’s just doing this one test, you just have to be careful,” Dr. Shukla said. “It’s not that easy.”
A version of this article first appeared on Medscape.com.
‘Financial toxicity’ from breast cancer is a worldwide phenomenon
Women across the world face high levels of financial burden from breast cancer, a new systematic review and analysis finds. While the burden of the disease is much higher in less-developed countries, about a third of women in Western nations like the United States say the disease has hurt their financial well-being.
When it comes to financial burden, patients with breast cancer are “a highly vulnerable patient population,” said study coauthor Kavitha Ranganathan, MD, of Brigham and Women’s Hospital, Boston, in an interview. “We need to be both strategic and comprehensive with our approach and use evidence-based methods to come up with these comprehensive solutions,” said Dr. Ranganathan, who noted that she’s hearing more from patients who face monetary hurdles.
The findings were published online in JAMA Network Open.
The researchers believe their analysis is the first to attempt to understand financial toxicity (FT) – excessive financial burden – in breast cancer on a global level. This turned out to be a challenge since there’s no standard way to measure FT.
One approach is to look at financial burden in terms of whether patients are suffering from “catastrophic expenditure,” Dr. Ranganathan said. “That’s what the World Bank and other top health and economic organizations have focused on. It means that the cost of care and – whatever it takes to get care – exceeds 10% of total annual household income.”
Another approach is more subjective and based on patient-reported outcomes, she said: “Are patients having to forgo basic subsistence needs like rent and food?”
For the report, researchers analyzed studies that use both approaches to measure FT from breast cancer. The studies came from high-income countries (n = 24, including 19 from the United States) and middle- and low-income countries (n = 10), and ranged in size from 5 to 2,445 subjects.
The analyzed studies were a range of cross-sectional (n = 26), prospective (n = 7), and retrospective designs (n = 1).
The authors pooled the data from 18 studies and estimated that the rate of patients with FT was 35.3% (14 studies, 27.3%-44.4%) in high-income countries and 78.8% (4 studies, 60.4%-90.0%) in the other countries.
The researchers also conducted a separate pooled analysis of only the U.S. studies (n = 11). It found that 34% (27%-43%) of subjects reported FT. The researchers also conducted a new analysis of Canada-only studies (n = 2) and found that 19% (9%-35%) reported FT.
The researchers weren’t able to provide insight into trends in FT in the United States prior to the period of the studies (2014-2021). But raw numbers suggest the percentage of patients facing financial challenges rose over that time, suggesting a possible increase in burden.
Previous research has suggested that breast cancer poses a higher financial burden than other chronic conditions. “Breast cancer care in particular may be associated with high FT given the need for screening and diagnosis, multidisciplinary care, and longitudinal follow-up,” the researchers write. They add that “notably, gender also affects financial security.”
As for limitations, the researchers report that they only analyzed studies in English, and there was a wide variation in approaches used to analyze FT. The analysis “did not account for different health care systems or control for health care–dedicated gross domestic product,” meaning that there’s no way to know for sure that rates were lower in nations with universal health care.
How could the new findings be useful? “They’re eye-opening for health policymakers. Whenever they see these numbers, they will say, ‘Wow, it is really a problem,’ and they’ll start thinking about solutions,” said study coauthor Rania A. Mekary, PhD, MSc, MSc, of Massachusetts College of Pharmacy and Health Sciences in Boston. “When you give them evidence-based data, then they will take it more seriously.”
The researchers call for interventions in several areas including education about early diagnosis and treatment of breast cancer, expansion of health care coverage, programs to help with nonmedical costs, and better resources for breast cancer care.
In an interview, Mary C. Politi, PhD, of Washington University, St. Louis, said the new report is useful “because it examines financial hardship internationally. Some people wonder whether financial hardship is a U.S. problem because of our health care system, which often relies on insurance and a lot of cost-sharing between insurance and patients. However, financial toxicity is prevalent across countries.”
And, she said, “the study is also useful because it encourages us to measure financial hardship and burden in a more uniform way so we can better compare and pool studies.”
Dr. Politi noted that there are ways to help patients now. “Most hospitals and health centers have staff who can talk to patients about their bills. Sometimes, a payment plan can be set up to space out payments,” she said. “Health care teams can try to consolidate care for patients on the same day to reduce parking expenses or time off for work or child care. Sometimes, changing to less expensive but effective generic medications is an option.”
The study authors received support from the National Cancer Institute, the United Nations Institute for Training and Research, the Global Surgery Foundation, the Harvard Global Health Institute, the Connors Center for Women’s Health and Gender Biology, the Center for Surgery and Public Health, and the National Endowment for Plastic Surgery. Dr. Ranganathan and Dr. Mekary report no disclosures. One coauthor reported a patent (BREAST-Q) and codevelopment of QPROMS, owned by Memorial Sloan Kettering Cancer Center. Another author reports salary support from Blue Cross Blue Shield of Michigan through the collaborative quality initiative known as Michigan Social Health Interventions to Eliminate Disparities. Dr. Politi has no disclosures.
Women across the world face high levels of financial burden from breast cancer, a new systematic review and analysis finds. While the burden of the disease is much higher in less-developed countries, about a third of women in Western nations like the United States say the disease has hurt their financial well-being.
When it comes to financial burden, patients with breast cancer are “a highly vulnerable patient population,” said study coauthor Kavitha Ranganathan, MD, of Brigham and Women’s Hospital, Boston, in an interview. “We need to be both strategic and comprehensive with our approach and use evidence-based methods to come up with these comprehensive solutions,” said Dr. Ranganathan, who noted that she’s hearing more from patients who face monetary hurdles.
The findings were published online in JAMA Network Open.
The researchers believe their analysis is the first to attempt to understand financial toxicity (FT) – excessive financial burden – in breast cancer on a global level. This turned out to be a challenge since there’s no standard way to measure FT.
One approach is to look at financial burden in terms of whether patients are suffering from “catastrophic expenditure,” Dr. Ranganathan said. “That’s what the World Bank and other top health and economic organizations have focused on. It means that the cost of care and – whatever it takes to get care – exceeds 10% of total annual household income.”
Another approach is more subjective and based on patient-reported outcomes, she said: “Are patients having to forgo basic subsistence needs like rent and food?”
For the report, researchers analyzed studies that use both approaches to measure FT from breast cancer. The studies came from high-income countries (n = 24, including 19 from the United States) and middle- and low-income countries (n = 10), and ranged in size from 5 to 2,445 subjects.
The analyzed studies were a range of cross-sectional (n = 26), prospective (n = 7), and retrospective designs (n = 1).
The authors pooled the data from 18 studies and estimated that the rate of patients with FT was 35.3% (14 studies, 27.3%-44.4%) in high-income countries and 78.8% (4 studies, 60.4%-90.0%) in the other countries.
The researchers also conducted a separate pooled analysis of only the U.S. studies (n = 11). It found that 34% (27%-43%) of subjects reported FT. The researchers also conducted a new analysis of Canada-only studies (n = 2) and found that 19% (9%-35%) reported FT.
The researchers weren’t able to provide insight into trends in FT in the United States prior to the period of the studies (2014-2021). But raw numbers suggest the percentage of patients facing financial challenges rose over that time, suggesting a possible increase in burden.
Previous research has suggested that breast cancer poses a higher financial burden than other chronic conditions. “Breast cancer care in particular may be associated with high FT given the need for screening and diagnosis, multidisciplinary care, and longitudinal follow-up,” the researchers write. They add that “notably, gender also affects financial security.”
As for limitations, the researchers report that they only analyzed studies in English, and there was a wide variation in approaches used to analyze FT. The analysis “did not account for different health care systems or control for health care–dedicated gross domestic product,” meaning that there’s no way to know for sure that rates were lower in nations with universal health care.
How could the new findings be useful? “They’re eye-opening for health policymakers. Whenever they see these numbers, they will say, ‘Wow, it is really a problem,’ and they’ll start thinking about solutions,” said study coauthor Rania A. Mekary, PhD, MSc, MSc, of Massachusetts College of Pharmacy and Health Sciences in Boston. “When you give them evidence-based data, then they will take it more seriously.”
The researchers call for interventions in several areas including education about early diagnosis and treatment of breast cancer, expansion of health care coverage, programs to help with nonmedical costs, and better resources for breast cancer care.
In an interview, Mary C. Politi, PhD, of Washington University, St. Louis, said the new report is useful “because it examines financial hardship internationally. Some people wonder whether financial hardship is a U.S. problem because of our health care system, which often relies on insurance and a lot of cost-sharing between insurance and patients. However, financial toxicity is prevalent across countries.”
And, she said, “the study is also useful because it encourages us to measure financial hardship and burden in a more uniform way so we can better compare and pool studies.”
Dr. Politi noted that there are ways to help patients now. “Most hospitals and health centers have staff who can talk to patients about their bills. Sometimes, a payment plan can be set up to space out payments,” she said. “Health care teams can try to consolidate care for patients on the same day to reduce parking expenses or time off for work or child care. Sometimes, changing to less expensive but effective generic medications is an option.”
The study authors received support from the National Cancer Institute, the United Nations Institute for Training and Research, the Global Surgery Foundation, the Harvard Global Health Institute, the Connors Center for Women’s Health and Gender Biology, the Center for Surgery and Public Health, and the National Endowment for Plastic Surgery. Dr. Ranganathan and Dr. Mekary report no disclosures. One coauthor reported a patent (BREAST-Q) and codevelopment of QPROMS, owned by Memorial Sloan Kettering Cancer Center. Another author reports salary support from Blue Cross Blue Shield of Michigan through the collaborative quality initiative known as Michigan Social Health Interventions to Eliminate Disparities. Dr. Politi has no disclosures.
Women across the world face high levels of financial burden from breast cancer, a new systematic review and analysis finds. While the burden of the disease is much higher in less-developed countries, about a third of women in Western nations like the United States say the disease has hurt their financial well-being.
When it comes to financial burden, patients with breast cancer are “a highly vulnerable patient population,” said study coauthor Kavitha Ranganathan, MD, of Brigham and Women’s Hospital, Boston, in an interview. “We need to be both strategic and comprehensive with our approach and use evidence-based methods to come up with these comprehensive solutions,” said Dr. Ranganathan, who noted that she’s hearing more from patients who face monetary hurdles.
The findings were published online in JAMA Network Open.
The researchers believe their analysis is the first to attempt to understand financial toxicity (FT) – excessive financial burden – in breast cancer on a global level. This turned out to be a challenge since there’s no standard way to measure FT.
One approach is to look at financial burden in terms of whether patients are suffering from “catastrophic expenditure,” Dr. Ranganathan said. “That’s what the World Bank and other top health and economic organizations have focused on. It means that the cost of care and – whatever it takes to get care – exceeds 10% of total annual household income.”
Another approach is more subjective and based on patient-reported outcomes, she said: “Are patients having to forgo basic subsistence needs like rent and food?”
For the report, researchers analyzed studies that use both approaches to measure FT from breast cancer. The studies came from high-income countries (n = 24, including 19 from the United States) and middle- and low-income countries (n = 10), and ranged in size from 5 to 2,445 subjects.
The analyzed studies were a range of cross-sectional (n = 26), prospective (n = 7), and retrospective designs (n = 1).
The authors pooled the data from 18 studies and estimated that the rate of patients with FT was 35.3% (14 studies, 27.3%-44.4%) in high-income countries and 78.8% (4 studies, 60.4%-90.0%) in the other countries.
The researchers also conducted a separate pooled analysis of only the U.S. studies (n = 11). It found that 34% (27%-43%) of subjects reported FT. The researchers also conducted a new analysis of Canada-only studies (n = 2) and found that 19% (9%-35%) reported FT.
The researchers weren’t able to provide insight into trends in FT in the United States prior to the period of the studies (2014-2021). But raw numbers suggest the percentage of patients facing financial challenges rose over that time, suggesting a possible increase in burden.
Previous research has suggested that breast cancer poses a higher financial burden than other chronic conditions. “Breast cancer care in particular may be associated with high FT given the need for screening and diagnosis, multidisciplinary care, and longitudinal follow-up,” the researchers write. They add that “notably, gender also affects financial security.”
As for limitations, the researchers report that they only analyzed studies in English, and there was a wide variation in approaches used to analyze FT. The analysis “did not account for different health care systems or control for health care–dedicated gross domestic product,” meaning that there’s no way to know for sure that rates were lower in nations with universal health care.
How could the new findings be useful? “They’re eye-opening for health policymakers. Whenever they see these numbers, they will say, ‘Wow, it is really a problem,’ and they’ll start thinking about solutions,” said study coauthor Rania A. Mekary, PhD, MSc, MSc, of Massachusetts College of Pharmacy and Health Sciences in Boston. “When you give them evidence-based data, then they will take it more seriously.”
The researchers call for interventions in several areas including education about early diagnosis and treatment of breast cancer, expansion of health care coverage, programs to help with nonmedical costs, and better resources for breast cancer care.
In an interview, Mary C. Politi, PhD, of Washington University, St. Louis, said the new report is useful “because it examines financial hardship internationally. Some people wonder whether financial hardship is a U.S. problem because of our health care system, which often relies on insurance and a lot of cost-sharing between insurance and patients. However, financial toxicity is prevalent across countries.”
And, she said, “the study is also useful because it encourages us to measure financial hardship and burden in a more uniform way so we can better compare and pool studies.”
Dr. Politi noted that there are ways to help patients now. “Most hospitals and health centers have staff who can talk to patients about their bills. Sometimes, a payment plan can be set up to space out payments,” she said. “Health care teams can try to consolidate care for patients on the same day to reduce parking expenses or time off for work or child care. Sometimes, changing to less expensive but effective generic medications is an option.”
The study authors received support from the National Cancer Institute, the United Nations Institute for Training and Research, the Global Surgery Foundation, the Harvard Global Health Institute, the Connors Center for Women’s Health and Gender Biology, the Center for Surgery and Public Health, and the National Endowment for Plastic Surgery. Dr. Ranganathan and Dr. Mekary report no disclosures. One coauthor reported a patent (BREAST-Q) and codevelopment of QPROMS, owned by Memorial Sloan Kettering Cancer Center. Another author reports salary support from Blue Cross Blue Shield of Michigan through the collaborative quality initiative known as Michigan Social Health Interventions to Eliminate Disparities. Dr. Politi has no disclosures.
FROM JAMA NETWORK OPEN
Progress in breast cancer screening over the past 50 years: A remarkable story, but still work to do
Meaningful progress has been made in reducing deaths due to breast cancer over the last half century, with a 43% decrease in mortality rate (breast cancer deaths per 100,000 population).1 Screening mammography (SM) has contributed greatly to that success, accounting for 30% to 70% of the reduced mortality rate, with the remainder due to advancements in breast cancer treatment.2 Despite these improvements, invasive breast cancer remains the highest incident cancer in the United States and in the world, is the second leading cause of cancer death in the United States, and results in more years of life lost than any other cancer (TABLE 1).1,3
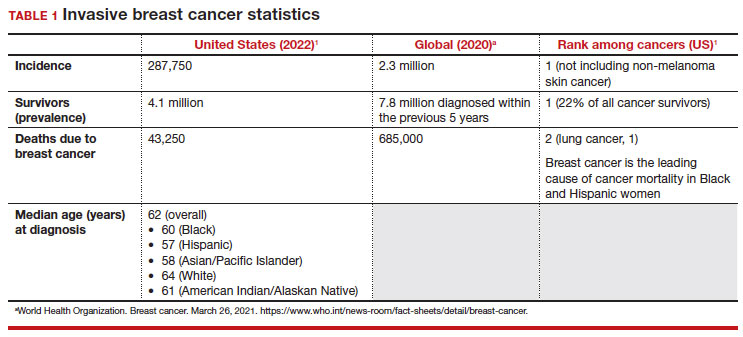
While the benefits and harms of SM are reasonably well understood, different guidelines groups have approached the relative value of the risks and benefits differently, which has led to challenges in implementation of shared decision making, particularly around the age to initiate routine screening.4-6 In this article, we will focus on the data behind the controversy, current gaps in knowledge, challenges related to breast density and screening in diverse groups, and emerging technologies to address these gaps and provide a construct for appropriate counseling of the patient across the risk spectrum.
In recognition of 35 years of publication of OBG Management, this article on breast cancer screening by Mark D. Pearlman, MD, kicks off a series that focuses on various cancer screening modalities and expert recommendations.
Stay tuned for articles on the future of cervical cancer screening and genetic testing for cancer risk beyond BRCA testing.
We look forward to continuing OBG Management’s mission of enhancing the quality of reproductive health care and the professional development of ObGyns and all women’s health care clinicians.
Breast cancer risk
Variables that affect risk
While female sex and older age are the 2 greatest risks for the development of breast cancer, many other factors can either increase or decrease breast cancer risk in a person’s lifetime. The importance of identifying risk factors is 3-fold:
- to perform risk assessment to determine if individuals would benefit from average-risk versus high-risk breast cancer surveillance
- to identify persons who might benefit from BRCA genetic counseling and screening, risk reduction medications or procedures, and
- to allow patients to determine whether any modification in their lifestyle or reproductive choices would make sense to them to reduce their future breast cancer risk.
Most of these risk variables are largely inalterable (for example, family history of breast cancer, carriage of genetic pathogenic variants such as BRCA1 and BRCA2, age of menarche and menopause), but some are potentially modifiable, such as parity, age at first birth, lactation and duration, and dietary factors, among others. TABLE 2 lists common breast cancer risk factors.
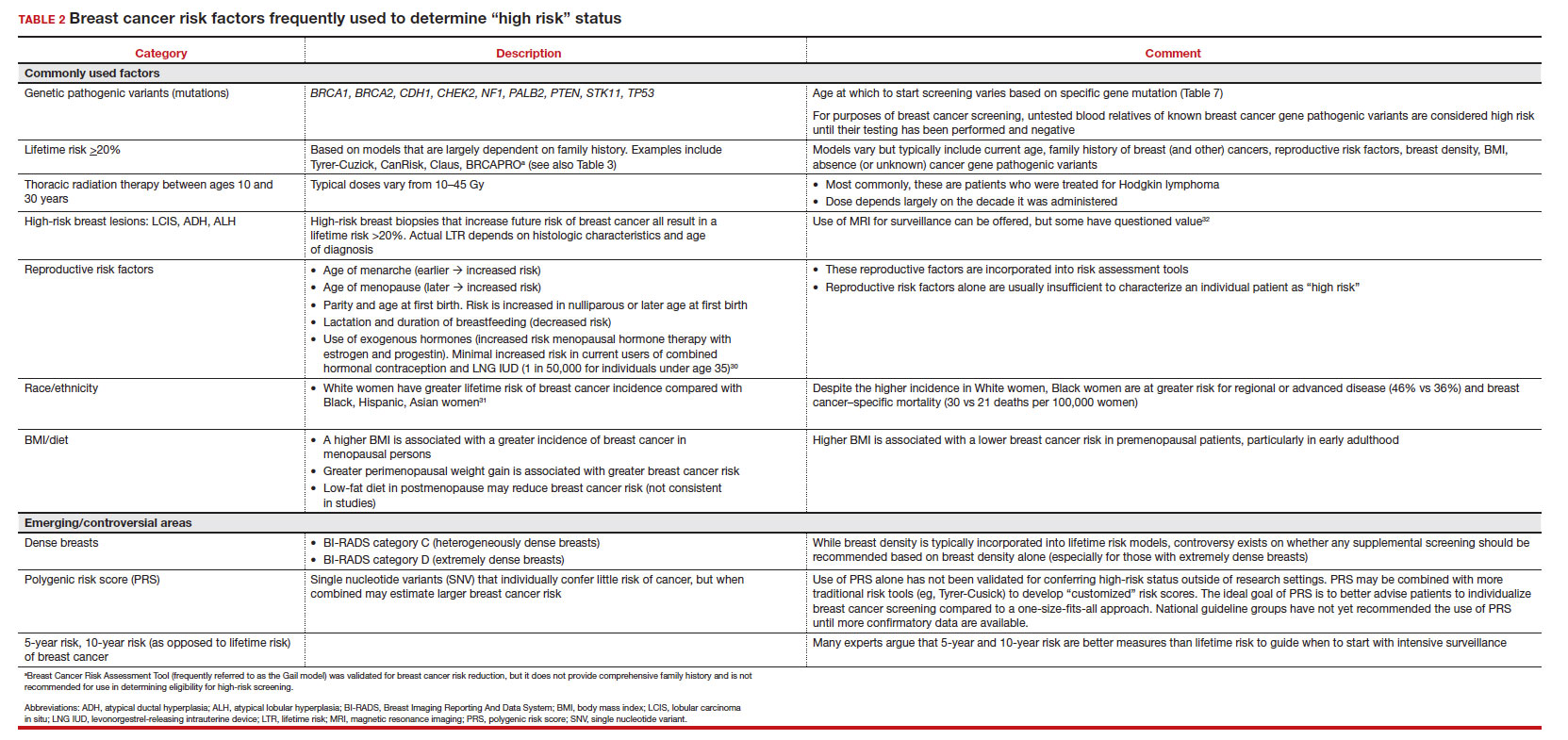
Breast cancer risk assessment
Several validated tools have been developed to estimate a person’s breast cancer risk (TABLE 3). These tools combine known risk factors and, depending on the specific tool, can provide estimates of 5-year, 10-year, or lifetime risk of breast cancer. Patients at highest risk can benefit from earlier screening, supplemental screening with breast magnetic resonance imaging (MRI), or risk reduction (see the section, “High-risk screening”). Ideally, a risk assessment should be done by age 30 so that patients at high risk can be identified for earlier or more intensive screening and for possible genetic testing in those at risk for carriage of the BRCA or other breast cancer gene pathogenic variants.5,7
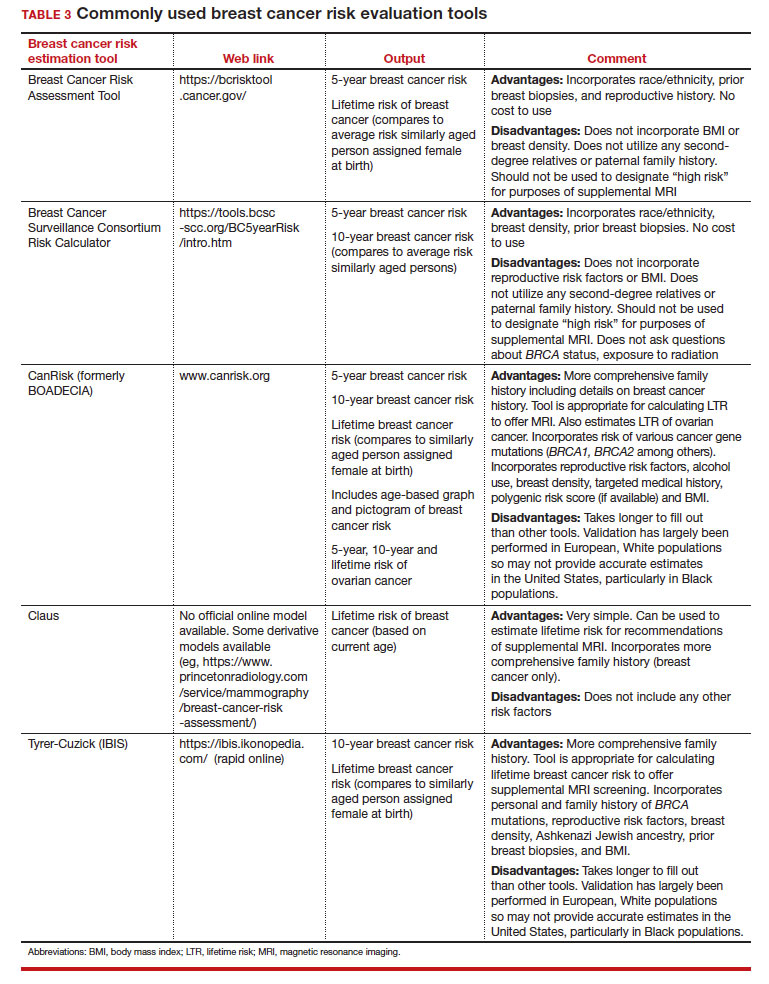
Continue to: Breast cancer screening: Efficacy and harms...
Breast cancer screening: Efficacy and harms
The earliest studies of breast cancer screening with mammography were randomized controlled trials (RCTs) that compared screened and unscreened patients aged 40 to 74. Nearly all the RCTs and numerous well-designed incidence-based and case-control studies have demonstrated that SM results in a clinically and statistically significant reduction in breast cancer mortality (TABLE 4).4,6,8 Since the mid-1980s and continuing to the current day, SM programs are routinely recommended in the United States. In addition to the mortality benefit outlined in TABLE 4, SM also is associated with a need for less invasive treatments if breast cancer is diagnosed.9,10
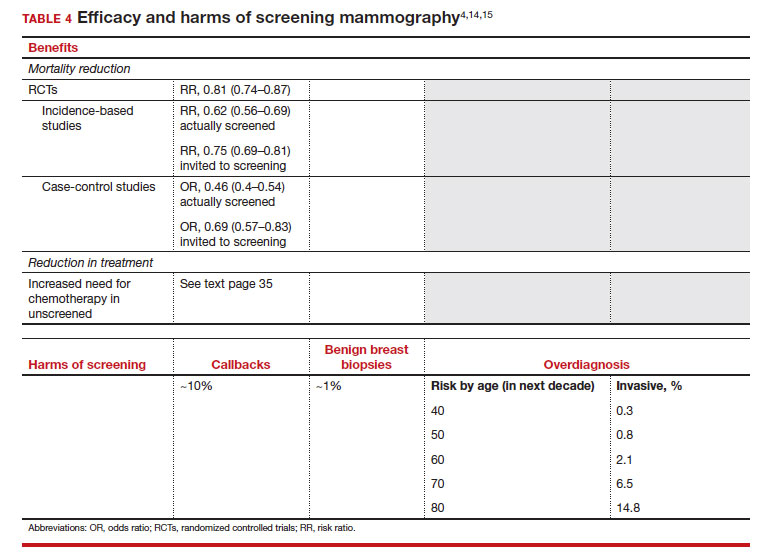
With several decades of experience, SM programs have demonstrated that multiple harms are associated with SM, including callbacks, false-positive mammograms that result in a benign biopsy, and overdiagnosis of breast cancer (TABLE 4). Overdiagnosis is a mammographic detection of a breast cancer that would not have harmed that woman in her lifetime. Overdiagnosis leads to overtreatment of breast cancers with its attendant side effects, the emotional harms of a breast cancer diagnosis, and the substantial financial cost of cancer treatment. Estimates of overdiagnosis range from 0% to 50%, with the most likely estimate of invasive breast cancer overdiagnosis from SM between 5% and 15%.11-13 Some of these overdiagnosed cancers are due to very slow growing cancers or breast cancers that may even regress. However, the higher rates of overdiagnosis occur in older persons who are screened and in whom competing causes of mortality become more prevalent. It is estimated that overdiagnosis of invasive breast cancer in patients younger than age 60 is less than 1%, but it exceeds 14% in those older than age 80 (TABLE 4).14
A structured approach is needed to counsel patients about SM so that they understand both the substantial benefit (earlier-stage diagnosis, reduced need for treatment, reduced breast cancer and all-cause mortality) and the potential harms (callback, false-positive results, and overdiagnosis). Moreover, the relative balance of the benefits and harms are influenced throughout their lifetime by both aging and changes in their personal and family medical history.
Counseling should consider factors beyond just the performance of mammography (sensitivity and specificity), such as the patient’s current health and age (competing causes of mortality), likelihood of developing breast cancer based on risk assessment (more benefit in higher-risk persons), and the individual patient’s values on the importance of the benefits and harms. The differing emphases on mammography performance and the relative value of the benefits and harms have led experts to produce disparate national guideline recommendations (TABLE 5).
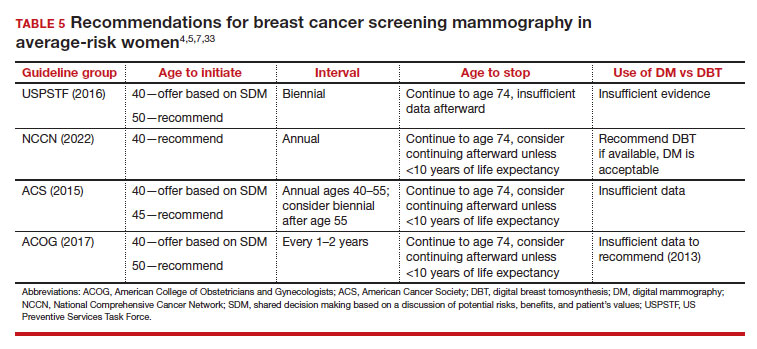
Should SM start at age 40, 45, or 50 in average-risk persons?
There is not clear consensus about the age at which to begin to recommend routine SM in patients at average risk. The National Comprehensive Cancer Network (NCCN),7 American Cancer Society (ACS),4 and the US Preventive Services Task Force (USPSTF)5 recommend that those at average risk start SM at age 40, 45, and 50, respectively (TABLE 5). While the guideline groups listed in TABLE 5 agree that there is level 1 evidence that SM reduces breast cancer mortality in the general population for persons starting at age 40, because the incidence of breast cancer is lower in younger persons (TABLE 6),4 the net population-based screening benefit is lower in this group, and the number needed to invite to screening to save a single life due to breast cancer varies.
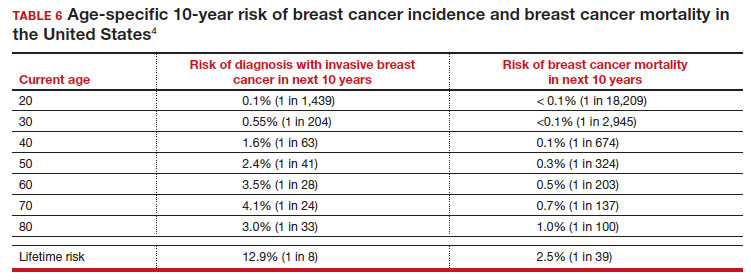
For patients in their 40s, it is estimated that 1,904 individuals need to be invited to SM to save 1 life, whereas for patients in their 50s, it is 1,339.15 However, for patients in their 40s, the number needed to screen to save 1 life due to breast cancer decreases from 1 in 1,904 if invited to be screened to 1 in 588 if they are actually screened.16 Furthermore, if a patient is diagnosed with breast cancer at age 40–50, the likelihood of dying is reduced at least 22% and perhaps as high as 48% if her cancer was diagnosed on SM compared with an unscreened individual with a symptomatic presentation (for example, palpable mass).4,15,17,18 Another benefit of SM in the fifth decade of life (40s) is the decreased need for more extensive treatment, including a higher risk of need for chemotherapy (odds ratio [OR], 2.81; 95% confidence interval [CI], 1.16–6.84); need for mastectomy (OR, 3.41; 95% CI, 1.36–8.52); and need for axillary lymph node dissection (OR, 5.76; 95% CI, 2.40–13.82) in unscreened (compared with screened) patients diagnosed with breast cancer.10
The harms associated with SM are not inconsequential and include callbacks (approximately 1 in 10), false-positive biopsy (approximately 1 in 100), and overdiagnosis (likely <1% of all breast cancers in persons younger than age 50). Because most patients in their 40s will not develop breast cancer (TABLE 6), the benefit of reduced breast cancer mortality will not be experienced by most in this decade of life, but they are still just as likely to experience a callback, false-positive biopsy, or the possibility of overdiagnosis. Interpretation of this balance on a population level is the crux of the various guideline groups’ development of differing recommendations as to when screening should start. Despite this seeming disagreement, all the guideline groups listed in TABLE 5 concur that persons at average risk for breast cancer should be offered SM if they desire starting at age 40 after a shared decision-making conversation that incorporates the patient’s view on the relative value of the benefits and risks.
Continue to: High-risk screening...
High-risk screening
Unlike in screening average-risk patients, there is less disagreement about screening in high-risk groups. TABLE 7 outlines the various categories and recommended strategies that qualify for screening at younger ages or more intensive screening. Adding breast MRI to SM in high-risk individuals results in both higher cancer detection rates and less interval breast cancers (cancers diagnosed between screening rounds) diagnosed compared with SM alone.19,20 Interval breast cancer tends to be more aggressive and is used as a surrogate marker for more recognized factors, such as breast cancer mortality. In addition to less interval breast cancers, high-risk patients are more likely to be diagnosed with node-negative disease if screening breast MRI is added to SM.
Long-term mortality benefit studies using MRI have not been conducted due to the prolonged follow-up times needed. Expense, lower specificity compared with mammography (that is, more false-positive results), and need for the use of gadolinium limit more widespread use of breast MRI screening in average-risk persons.
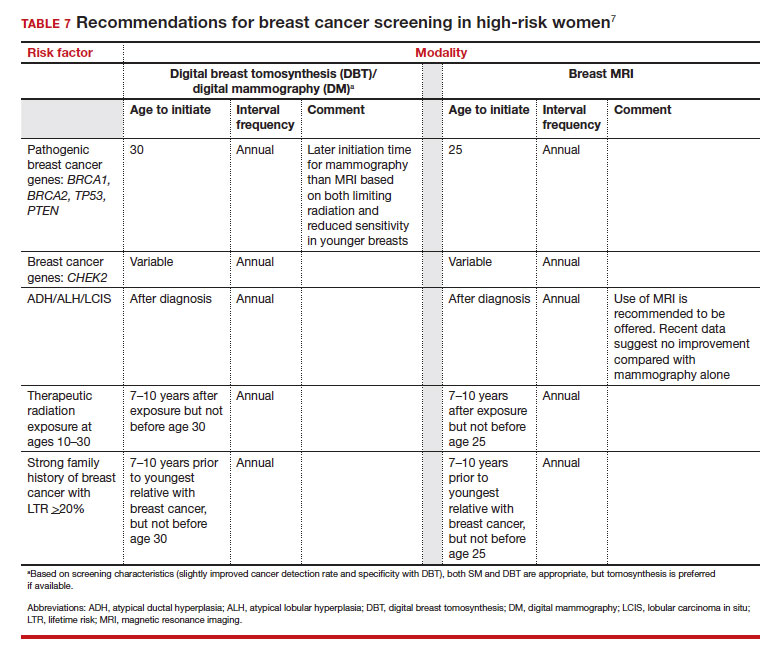
Screening in patients with dense breasts
Half of patients undergoing SM in the United States have dense breasts (heterogeneously dense breasts, 40%; extremely dense breasts, 10%). Importantly, increasing breast density is associated with a lower cancer detection rate with SM and is an independent risk factor for developing breast cancer. While most states already require patients to be notified if they have dense breasts identified on SM, the US Food and Drug Administration will soon make breast density patient notification a national standard (see: https://delauro.house.gov/media-center/press-releases/delauro-secures-timeline-fda-rollout-breast-density-notification-rule).
Most of the risk assessment tools listed in TABLE 3 incorporate breast density into their calculation of breast cancer risk. If that calculation places a patient into one of the highest-risk groups (based on additional factors like strong family history of breast cancer, reproductive risk factors, BRCA carriage, and so on), more intensive surveillance should be recommended (TABLE 7).7 However, once these risk calculations are done, most persons with dense breasts will remain in an average-risk category.
Because of the frequency and risks associated with dense breasts, different and alternative strategies have been recommended for screening persons who are at average risk with dense breasts. Supplemental screening with MRI, ultrasonography, contrast-enhanced mammography, and molecular breast imaging are all being considered but have not been studied sufficiently to demonstrate mortality benefit or cost-effectiveness.
Of all the supplemental modalities used to screen patients with dense breasts, MRI has been the best studied. A large RCT in the Netherlands evaluated supplemental MRI screening in persons with extremely dense breasts after a negative mammogram.21 Compared with no supplemental screening, the MRI group had 17 additional cancers detected per 1,000 screened and a 50% reduction in interval breast cancers; in addition, MRI was associated with a positive predictive value of 26% for biopsies. At present, high cost and limited access to standard breast MRI has not allowed its routine use for persons with dense breasts in the United States, but this may change with more experience and more widespread introduction and experience with abbreviated (or rapid) breast MRI in the future (TABLE 8).
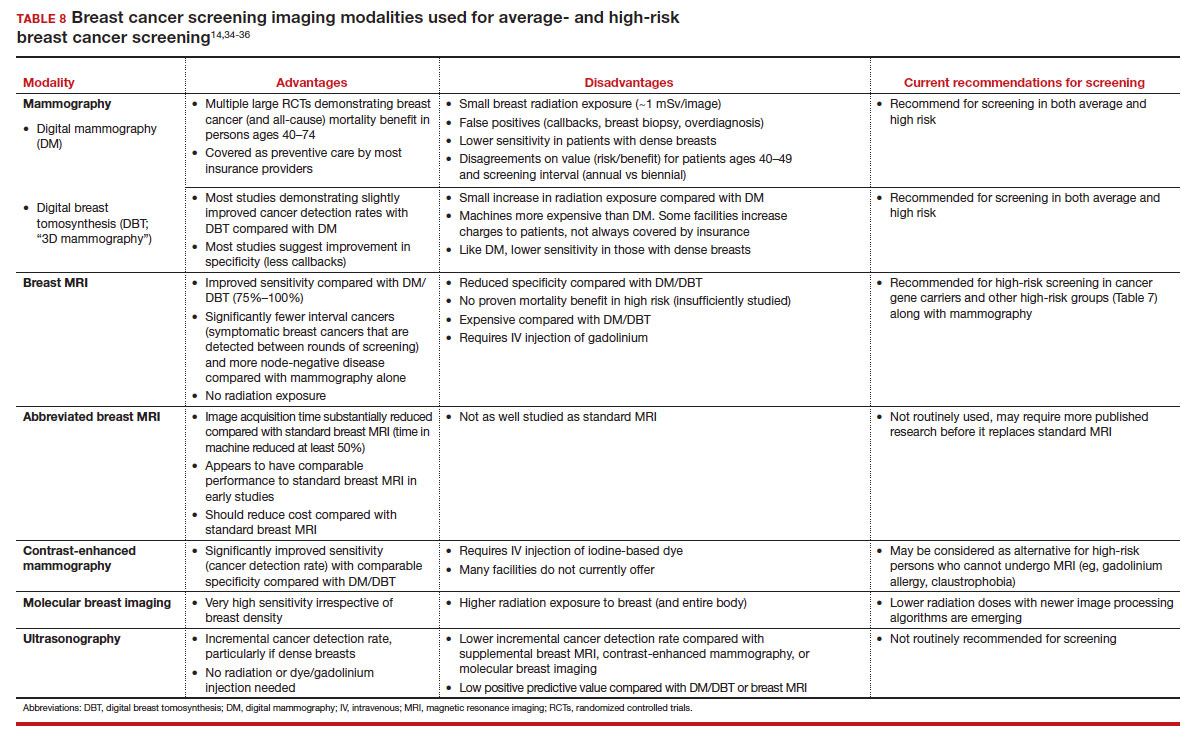
Equitable screening
Black persons who are diagnosed with breast cancer have a 40% higher risk of dying than White patients due to multiple factors, including systemic racial factors (implicit and unconscious bias), reduced access to care, and a lower likelihood of receiving standard of care once diagnosed.22-24 In addition, Black patients have twice the likelihood of being diagnosed with triple-negative breast cancers, a biologically more aggressive tumor.22-24 Among Black, Asian, and Hispanic persons diagnosed with breast cancer, one-third are diagnosed younger than age 50, which is higher than for non-Hispanic White persons. Prior to the age of 50, Black, Asian, and Hispanic patients also have a 72% more likelihood of being diagnosed with invasive breast cancer, have a 58% greater risk of advanced-stage disease, and have a 127% higher risk of dying from breast cancer compared with White patients.25,26 Based on all of these factors, delaying SM until age 50 may adversely affect the Black, Asian, and Hispanic populations.
Persons in the LGBTQ+ community do not present for SM as frequently as the general population, often because they feel threatened or unwelcome.27 Clinicians and breast imaging units should review their inclusivity policies and training to provide a welcoming and respectful environment to all persons in an effort to reduce these barriers. While data are limited and largely depend on expert opinion, current recommendations for screening in the transgender patient depend on sex assigned at birth, the type and duration of hormone use, and surgical history. In patients assigned female sex at birth, average-risk and high-risk screening recommendations are similar to those for the general population unless bilateral mastectomy has been performed.28 In transfeminine patients who have used hormones for longer than 5 years, some groups recommend annual screening starting at age 40, although well-designed studies are lacking.29
Continue to: We have done well, can we do better?...
We have done well, can we do better?
Screening mammography clearly has been an important and effective tool in the effort to reduce breast cancer mortality, but there are clear limitations. These include moderate sensitivity of mammography, particularly in patients with dense breasts, and a specificity that results in either callbacks (10%), breast biopsies for benign disease (1%), or the reality of overdiagnosis, which becomes increasingly important in older patients.
With the introduction of mammography in the mid-1980s, a one-size-fits-all approach has proved challenging more recently due to an increased recognition of the harms of screening. As a result of this evolving understanding, different recommendations for average-risk screening have emerged. With the advent of breast MRI, risk-based screening is an important but underutilized tool to identify highest-risk individuals, which is associated with improved cancer detection rates, reduced node-positive disease, and fewer diagnosed interval breast cancers. Assuring that nearly all of this highest-risk group is identified through routine breast cancer risk assessment remains a challenge for clinicians.
But what SM recommendations should be offered to persons who fall into an intermediate-risk group (15%–20%), very low-risk groups (<5%), or patients with dense breasts? These are challenges that could be met through novel and individualized approaches (for example, polygenic risk scoring, further research on newer modalities of screening [TABLE 8]), improved screening algorithms for persons with dense breasts, and enhanced clinician engagement to achieve universal breast cancer and BRCA risk assessment of patients by age 25 to 30.
In 2023, best practice and consensus guidelines for intermediate- and low-risk breast cancer groups remain unclear, and one of the many ongoing challenges is to further reduce the impact of breast cancer on the lives of persons affected and the recognized harms of SM.
In the meantime, there is consensus in average-risk patients to provide counseling about SM by age 40. My approach has been to counsel all average-risk patients on the risks and benefits of mammography using the acronym TIP-V:
- Use a Tool to calculate breast cancer risk (TABLE 3). If they are at high risk, provide recommendations for high-risk management (TABLE 7).7
- For average-risk patients, counsel that their Incidence of developing breast cancer in the next decade is approximately 1 in 70 (TABLE 6).4
- Provide data and guidance on the benefits of SM for patients in their 40s (mortality improvement, decreased treatment) and the likelihood of harm from breast cancer screening (10% callback, 1% benign biopsy, and <1% likelihood of overdiagnosis [TABLE 4]).4,14,15
- Engage the patient to better understand their relative Values of the benefits and harms and make a shared decision on screening starting at age 40, 45, or 50.
Looking forward
In summary, SM remains an important tool in the effort to decrease the risk of mortality due to breast cancer. Given the limitations of SM, however, newer tools and methods—abbreviated MRI, contrast-enhanced mammography, molecular breast imaging, customized screening intervals depending on individual risk/polygenic risk score, and customized counseling and screening based on risk factors (TABLES 2 and 7)—will play an increased role in recommendations for breast cancer screening in the future. ●
- Giaquinto AN, Sung H, Miller KD, et al. Breast cancer statistics, 2022. CA Cancer J Clin. 2022;72:524-541.
- Berry DA, Cronin KA, Plevritis SK, et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med. 2005;353:1784-1792.
- Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209-249.
- Oeffinger KC, Fontham ET, Etzioni R, et al; American Cancer Society. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314:1599-1614.
- US Preventive Services Task Force; Owens DK, Davidson KW, Drist AH, et al. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer: US Preventive Services Task Force Recommendation statement. JAMA. 2019;322:652-665.
- Nelson HD, Cantor A, Humphrey L, et al. Screening for breast cancer: a systematic review to update the 2009 US Preventive Services Task Force recommendation. Evidence synthesis no 124. AHRQ publication no 14-05201-EF-1. Rockville, MD: Agency for Healthcare Research and Quality; 2016.
- Bevers TB, Helvie M, Bonaccio E, et al. Breast cancer screening and diagnosis, version 3.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2018;16:1362-1389.
- Duffy SW, Vulkan D, Cuckle H, et al. Effect of mammographic screening from age 40 years on breast cancer mortality (UK Age trial): final results of a randomised, controlled trial. Lancet Oncol. 2020;21:1165-1172.
- Karzai S, Port E, Siderides C, et al. Impact of screening mammography on treatment in young women diagnosed with breast cancer. Ann Surg Oncol. 2022. doi:10.1245/ s10434-022-11581-6.
- Ahn S, Wooster M, Valente C, et al. Impact of screening mammography on treatment in women diagnosed with breast cancer. Ann Surg Oncol. 2018;25:2979-2986.
- Coldman A, Phillips N. Incidence of breast cancer and estimates of overdiagnosis after the initiation of a population-based mammography screening program. CMAJ. 2013;185:E492-E498.
- Etzioni R, Gulati R, Mallinger L, et al. Influence of study features and methods on overdiagnosis estimates in breast and prostate cancer screening. Ann Internal Med. 2013;158:831-838.
- Ryser MD, Lange J, Inoue LY, et al. Estimation of breast cancer overdiagnosis in a US breast screening cohort. Ann Intern Med. 2022;175:471-478.
- Monticciolo DL, Malak SF, Friedewald SM, et al. Breast cancer screening recommendations inclusive of all women at average risk: update from the ACR and Society of Breast Imaging. J Am Coll Radiol. 2021;18:1280-1288.
- Nelson HD, Fu R, Cantor A, Pappas M, et al. Effectiveness of breast cancer screening: systematic review and meta-analysis to update the 2009 US Preventive Services Task Force recommendation. Ann Internal Med. 2016;164:244-255.
- Hendrick RE, Helvie MA, Hardesty LA. Implications of CISNET modeling on number needed to screen and mortality reduction with digital mammography in women 40–49 years old. Am J Roentgenol. 2014;203:1379-1381.
- Broeders M, Moss S, Nyström L, et al; EUROSCREEN Working Group. The impact of mammographic screening on breast cancer mortality in Europe: a review of observational studies. J Med Screen. 2012;19(suppl 1):14-25.
- Tabár L, Yen AMF, Wu WYY, et al. Insights from the breast cancer screening trials: how screening affects the natural history of breast cancer and implications for evaluating service screening programs. Breast J. 2015;21:13-20.
- Kriege M, Brekelmans CTM, Boetes C, et al; Magnetic Resonance Imaging Screening Study Group. Efficacy of MRI and mammography for breast-cancer screening in women with a familial or genetic predisposition. N Engl J Med. 2004;351:427-437.
- Vreemann S, Gubern-Merida A, Lardenoije S, et al. The frequency of missed breast cancers in women participating in a high-risk MRI screening program. Breast Cancer Res Treat. 2018;169:323-331.
- Bakker MF, de Lange SV, Pijnappel RM, et al. Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med. 2019;381:2091-2102.
- Amirikia KC, Mills P, Bush J, et al. Higher population‐based incidence rates of triple‐negative breast cancer among young African‐American women: implications for breast cancer screening recommendations. Cancer. 2011;117:2747-2753.
- Kohler BA, Sherman RL, Howlader N, et al. Annual report to the nation on the status of cancer, 1975-2011, featuring incidence of breast cancer subtypes by race/ethnicity, poverty, and state. J Natl Cancer Inst. 2015;107:djv048.
- Newman LA, Kaljee LM. Health disparities and triple-negative breast cancer in African American women: a review. JAMA Surg. 2017;152:485-493.
- Stapleton SM, Oseni TO, Bababekov YJ, et al. Race/ethnicity and age distribution of breast cancer diagnosis in the United States. JAMA Surg. 2018;153:594-595.
- Hendrick RE, Monticciolo DL, Biggs KW, et al. Age distributions of breast cancer diagnosis and mortality by race and ethnicity in US women. Cancer. 2021;127:4384-4392.
- Perry H, Fang AJ, Tsai EM, et al. Imaging health and radiology care of transgender patients: a call to build evidence-based best practices. J Am Coll Radiol. 2021;18(3 pt B):475-480.
- Lockhart R, Kamaya A. Patient-friendly summary of the ACR Appropriateness Criteria: transgender breast cancer screening. J Am Coll Radiol. 2022;19:e19.
- Expert Panel on Breast Imaging; Brown A, Lourenco AP, Niell BL, et al. ACR Appropriateness Criteria transgender breast cancer screening. J Am Coll Radiol. 2021;18:S502-S515.
- Mørch LS, Skovlund CW, Hannaford PC, et al. Contemporary hormonal contraception and the risk of breast cancer. N Engl J Med. 2017;377:2228-2239.
- Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7-33.
- Laws A, Katlin F, Hans M, et al. Screening MRI does not increase cancer detection or result in an earlier stage at diagnosis for patients with high-risk breast lesions: a propensity score analysis. Ann Surg Oncol. 2023;30;68-77.
- American College of Obstetricians and Gynecologists. Practice bulletin no 179: Breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130:e1-e16.
- Grimm LJ, Mango VL, Harvey JA, et al. Implementation of abbreviated breast MRI for screening: AJR expert panel narrative review. AJR Am J Roentgenol. 2022;218:202-212.
- Potsch N, Vatteroini G, Clauser P, et al. Contrast-enhanced mammography versus contrast-enhanced breast MRI: a systematic review and meta-analysis. Radiology. 2022;305:94-103.
- Covington MF, Parent EE, Dibble EH, et al. Advances and future directions in molecular breast imaging. J Nucl Med. 2022;63:17-21.
Meaningful progress has been made in reducing deaths due to breast cancer over the last half century, with a 43% decrease in mortality rate (breast cancer deaths per 100,000 population).1 Screening mammography (SM) has contributed greatly to that success, accounting for 30% to 70% of the reduced mortality rate, with the remainder due to advancements in breast cancer treatment.2 Despite these improvements, invasive breast cancer remains the highest incident cancer in the United States and in the world, is the second leading cause of cancer death in the United States, and results in more years of life lost than any other cancer (TABLE 1).1,3

While the benefits and harms of SM are reasonably well understood, different guidelines groups have approached the relative value of the risks and benefits differently, which has led to challenges in implementation of shared decision making, particularly around the age to initiate routine screening.4-6 In this article, we will focus on the data behind the controversy, current gaps in knowledge, challenges related to breast density and screening in diverse groups, and emerging technologies to address these gaps and provide a construct for appropriate counseling of the patient across the risk spectrum.
In recognition of 35 years of publication of OBG Management, this article on breast cancer screening by Mark D. Pearlman, MD, kicks off a series that focuses on various cancer screening modalities and expert recommendations.
Stay tuned for articles on the future of cervical cancer screening and genetic testing for cancer risk beyond BRCA testing.
We look forward to continuing OBG Management’s mission of enhancing the quality of reproductive health care and the professional development of ObGyns and all women’s health care clinicians.
Breast cancer risk
Variables that affect risk
While female sex and older age are the 2 greatest risks for the development of breast cancer, many other factors can either increase or decrease breast cancer risk in a person’s lifetime. The importance of identifying risk factors is 3-fold:
- to perform risk assessment to determine if individuals would benefit from average-risk versus high-risk breast cancer surveillance
- to identify persons who might benefit from BRCA genetic counseling and screening, risk reduction medications or procedures, and
- to allow patients to determine whether any modification in their lifestyle or reproductive choices would make sense to them to reduce their future breast cancer risk.
Most of these risk variables are largely inalterable (for example, family history of breast cancer, carriage of genetic pathogenic variants such as BRCA1 and BRCA2, age of menarche and menopause), but some are potentially modifiable, such as parity, age at first birth, lactation and duration, and dietary factors, among others. TABLE 2 lists common breast cancer risk factors.

Breast cancer risk assessment
Several validated tools have been developed to estimate a person’s breast cancer risk (TABLE 3). These tools combine known risk factors and, depending on the specific tool, can provide estimates of 5-year, 10-year, or lifetime risk of breast cancer. Patients at highest risk can benefit from earlier screening, supplemental screening with breast magnetic resonance imaging (MRI), or risk reduction (see the section, “High-risk screening”). Ideally, a risk assessment should be done by age 30 so that patients at high risk can be identified for earlier or more intensive screening and for possible genetic testing in those at risk for carriage of the BRCA or other breast cancer gene pathogenic variants.5,7

Continue to: Breast cancer screening: Efficacy and harms...
Breast cancer screening: Efficacy and harms
The earliest studies of breast cancer screening with mammography were randomized controlled trials (RCTs) that compared screened and unscreened patients aged 40 to 74. Nearly all the RCTs and numerous well-designed incidence-based and case-control studies have demonstrated that SM results in a clinically and statistically significant reduction in breast cancer mortality (TABLE 4).4,6,8 Since the mid-1980s and continuing to the current day, SM programs are routinely recommended in the United States. In addition to the mortality benefit outlined in TABLE 4, SM also is associated with a need for less invasive treatments if breast cancer is diagnosed.9,10

With several decades of experience, SM programs have demonstrated that multiple harms are associated with SM, including callbacks, false-positive mammograms that result in a benign biopsy, and overdiagnosis of breast cancer (TABLE 4). Overdiagnosis is a mammographic detection of a breast cancer that would not have harmed that woman in her lifetime. Overdiagnosis leads to overtreatment of breast cancers with its attendant side effects, the emotional harms of a breast cancer diagnosis, and the substantial financial cost of cancer treatment. Estimates of overdiagnosis range from 0% to 50%, with the most likely estimate of invasive breast cancer overdiagnosis from SM between 5% and 15%.11-13 Some of these overdiagnosed cancers are due to very slow growing cancers or breast cancers that may even regress. However, the higher rates of overdiagnosis occur in older persons who are screened and in whom competing causes of mortality become more prevalent. It is estimated that overdiagnosis of invasive breast cancer in patients younger than age 60 is less than 1%, but it exceeds 14% in those older than age 80 (TABLE 4).14
A structured approach is needed to counsel patients about SM so that they understand both the substantial benefit (earlier-stage diagnosis, reduced need for treatment, reduced breast cancer and all-cause mortality) and the potential harms (callback, false-positive results, and overdiagnosis). Moreover, the relative balance of the benefits and harms are influenced throughout their lifetime by both aging and changes in their personal and family medical history.
Counseling should consider factors beyond just the performance of mammography (sensitivity and specificity), such as the patient’s current health and age (competing causes of mortality), likelihood of developing breast cancer based on risk assessment (more benefit in higher-risk persons), and the individual patient’s values on the importance of the benefits and harms. The differing emphases on mammography performance and the relative value of the benefits and harms have led experts to produce disparate national guideline recommendations (TABLE 5).

Should SM start at age 40, 45, or 50 in average-risk persons?
There is not clear consensus about the age at which to begin to recommend routine SM in patients at average risk. The National Comprehensive Cancer Network (NCCN),7 American Cancer Society (ACS),4 and the US Preventive Services Task Force (USPSTF)5 recommend that those at average risk start SM at age 40, 45, and 50, respectively (TABLE 5). While the guideline groups listed in TABLE 5 agree that there is level 1 evidence that SM reduces breast cancer mortality in the general population for persons starting at age 40, because the incidence of breast cancer is lower in younger persons (TABLE 6),4 the net population-based screening benefit is lower in this group, and the number needed to invite to screening to save a single life due to breast cancer varies.

For patients in their 40s, it is estimated that 1,904 individuals need to be invited to SM to save 1 life, whereas for patients in their 50s, it is 1,339.15 However, for patients in their 40s, the number needed to screen to save 1 life due to breast cancer decreases from 1 in 1,904 if invited to be screened to 1 in 588 if they are actually screened.16 Furthermore, if a patient is diagnosed with breast cancer at age 40–50, the likelihood of dying is reduced at least 22% and perhaps as high as 48% if her cancer was diagnosed on SM compared with an unscreened individual with a symptomatic presentation (for example, palpable mass).4,15,17,18 Another benefit of SM in the fifth decade of life (40s) is the decreased need for more extensive treatment, including a higher risk of need for chemotherapy (odds ratio [OR], 2.81; 95% confidence interval [CI], 1.16–6.84); need for mastectomy (OR, 3.41; 95% CI, 1.36–8.52); and need for axillary lymph node dissection (OR, 5.76; 95% CI, 2.40–13.82) in unscreened (compared with screened) patients diagnosed with breast cancer.10
The harms associated with SM are not inconsequential and include callbacks (approximately 1 in 10), false-positive biopsy (approximately 1 in 100), and overdiagnosis (likely <1% of all breast cancers in persons younger than age 50). Because most patients in their 40s will not develop breast cancer (TABLE 6), the benefit of reduced breast cancer mortality will not be experienced by most in this decade of life, but they are still just as likely to experience a callback, false-positive biopsy, or the possibility of overdiagnosis. Interpretation of this balance on a population level is the crux of the various guideline groups’ development of differing recommendations as to when screening should start. Despite this seeming disagreement, all the guideline groups listed in TABLE 5 concur that persons at average risk for breast cancer should be offered SM if they desire starting at age 40 after a shared decision-making conversation that incorporates the patient’s view on the relative value of the benefits and risks.
Continue to: High-risk screening...
High-risk screening
Unlike in screening average-risk patients, there is less disagreement about screening in high-risk groups. TABLE 7 outlines the various categories and recommended strategies that qualify for screening at younger ages or more intensive screening. Adding breast MRI to SM in high-risk individuals results in both higher cancer detection rates and less interval breast cancers (cancers diagnosed between screening rounds) diagnosed compared with SM alone.19,20 Interval breast cancer tends to be more aggressive and is used as a surrogate marker for more recognized factors, such as breast cancer mortality. In addition to less interval breast cancers, high-risk patients are more likely to be diagnosed with node-negative disease if screening breast MRI is added to SM.
Long-term mortality benefit studies using MRI have not been conducted due to the prolonged follow-up times needed. Expense, lower specificity compared with mammography (that is, more false-positive results), and need for the use of gadolinium limit more widespread use of breast MRI screening in average-risk persons.

Screening in patients with dense breasts
Half of patients undergoing SM in the United States have dense breasts (heterogeneously dense breasts, 40%; extremely dense breasts, 10%). Importantly, increasing breast density is associated with a lower cancer detection rate with SM and is an independent risk factor for developing breast cancer. While most states already require patients to be notified if they have dense breasts identified on SM, the US Food and Drug Administration will soon make breast density patient notification a national standard (see: https://delauro.house.gov/media-center/press-releases/delauro-secures-timeline-fda-rollout-breast-density-notification-rule).
Most of the risk assessment tools listed in TABLE 3 incorporate breast density into their calculation of breast cancer risk. If that calculation places a patient into one of the highest-risk groups (based on additional factors like strong family history of breast cancer, reproductive risk factors, BRCA carriage, and so on), more intensive surveillance should be recommended (TABLE 7).7 However, once these risk calculations are done, most persons with dense breasts will remain in an average-risk category.
Because of the frequency and risks associated with dense breasts, different and alternative strategies have been recommended for screening persons who are at average risk with dense breasts. Supplemental screening with MRI, ultrasonography, contrast-enhanced mammography, and molecular breast imaging are all being considered but have not been studied sufficiently to demonstrate mortality benefit or cost-effectiveness.
Of all the supplemental modalities used to screen patients with dense breasts, MRI has been the best studied. A large RCT in the Netherlands evaluated supplemental MRI screening in persons with extremely dense breasts after a negative mammogram.21 Compared with no supplemental screening, the MRI group had 17 additional cancers detected per 1,000 screened and a 50% reduction in interval breast cancers; in addition, MRI was associated with a positive predictive value of 26% for biopsies. At present, high cost and limited access to standard breast MRI has not allowed its routine use for persons with dense breasts in the United States, but this may change with more experience and more widespread introduction and experience with abbreviated (or rapid) breast MRI in the future (TABLE 8).

Equitable screening
Black persons who are diagnosed with breast cancer have a 40% higher risk of dying than White patients due to multiple factors, including systemic racial factors (implicit and unconscious bias), reduced access to care, and a lower likelihood of receiving standard of care once diagnosed.22-24 In addition, Black patients have twice the likelihood of being diagnosed with triple-negative breast cancers, a biologically more aggressive tumor.22-24 Among Black, Asian, and Hispanic persons diagnosed with breast cancer, one-third are diagnosed younger than age 50, which is higher than for non-Hispanic White persons. Prior to the age of 50, Black, Asian, and Hispanic patients also have a 72% more likelihood of being diagnosed with invasive breast cancer, have a 58% greater risk of advanced-stage disease, and have a 127% higher risk of dying from breast cancer compared with White patients.25,26 Based on all of these factors, delaying SM until age 50 may adversely affect the Black, Asian, and Hispanic populations.
Persons in the LGBTQ+ community do not present for SM as frequently as the general population, often because they feel threatened or unwelcome.27 Clinicians and breast imaging units should review their inclusivity policies and training to provide a welcoming and respectful environment to all persons in an effort to reduce these barriers. While data are limited and largely depend on expert opinion, current recommendations for screening in the transgender patient depend on sex assigned at birth, the type and duration of hormone use, and surgical history. In patients assigned female sex at birth, average-risk and high-risk screening recommendations are similar to those for the general population unless bilateral mastectomy has been performed.28 In transfeminine patients who have used hormones for longer than 5 years, some groups recommend annual screening starting at age 40, although well-designed studies are lacking.29
Continue to: We have done well, can we do better?...
We have done well, can we do better?
Screening mammography clearly has been an important and effective tool in the effort to reduce breast cancer mortality, but there are clear limitations. These include moderate sensitivity of mammography, particularly in patients with dense breasts, and a specificity that results in either callbacks (10%), breast biopsies for benign disease (1%), or the reality of overdiagnosis, which becomes increasingly important in older patients.
With the introduction of mammography in the mid-1980s, a one-size-fits-all approach has proved challenging more recently due to an increased recognition of the harms of screening. As a result of this evolving understanding, different recommendations for average-risk screening have emerged. With the advent of breast MRI, risk-based screening is an important but underutilized tool to identify highest-risk individuals, which is associated with improved cancer detection rates, reduced node-positive disease, and fewer diagnosed interval breast cancers. Assuring that nearly all of this highest-risk group is identified through routine breast cancer risk assessment remains a challenge for clinicians.
But what SM recommendations should be offered to persons who fall into an intermediate-risk group (15%–20%), very low-risk groups (<5%), or patients with dense breasts? These are challenges that could be met through novel and individualized approaches (for example, polygenic risk scoring, further research on newer modalities of screening [TABLE 8]), improved screening algorithms for persons with dense breasts, and enhanced clinician engagement to achieve universal breast cancer and BRCA risk assessment of patients by age 25 to 30.
In 2023, best practice and consensus guidelines for intermediate- and low-risk breast cancer groups remain unclear, and one of the many ongoing challenges is to further reduce the impact of breast cancer on the lives of persons affected and the recognized harms of SM.
In the meantime, there is consensus in average-risk patients to provide counseling about SM by age 40. My approach has been to counsel all average-risk patients on the risks and benefits of mammography using the acronym TIP-V:
- Use a Tool to calculate breast cancer risk (TABLE 3). If they are at high risk, provide recommendations for high-risk management (TABLE 7).7
- For average-risk patients, counsel that their Incidence of developing breast cancer in the next decade is approximately 1 in 70 (TABLE 6).4
- Provide data and guidance on the benefits of SM for patients in their 40s (mortality improvement, decreased treatment) and the likelihood of harm from breast cancer screening (10% callback, 1% benign biopsy, and <1% likelihood of overdiagnosis [TABLE 4]).4,14,15
- Engage the patient to better understand their relative Values of the benefits and harms and make a shared decision on screening starting at age 40, 45, or 50.
Looking forward
In summary, SM remains an important tool in the effort to decrease the risk of mortality due to breast cancer. Given the limitations of SM, however, newer tools and methods—abbreviated MRI, contrast-enhanced mammography, molecular breast imaging, customized screening intervals depending on individual risk/polygenic risk score, and customized counseling and screening based on risk factors (TABLES 2 and 7)—will play an increased role in recommendations for breast cancer screening in the future. ●
Meaningful progress has been made in reducing deaths due to breast cancer over the last half century, with a 43% decrease in mortality rate (breast cancer deaths per 100,000 population).1 Screening mammography (SM) has contributed greatly to that success, accounting for 30% to 70% of the reduced mortality rate, with the remainder due to advancements in breast cancer treatment.2 Despite these improvements, invasive breast cancer remains the highest incident cancer in the United States and in the world, is the second leading cause of cancer death in the United States, and results in more years of life lost than any other cancer (TABLE 1).1,3

While the benefits and harms of SM are reasonably well understood, different guidelines groups have approached the relative value of the risks and benefits differently, which has led to challenges in implementation of shared decision making, particularly around the age to initiate routine screening.4-6 In this article, we will focus on the data behind the controversy, current gaps in knowledge, challenges related to breast density and screening in diverse groups, and emerging technologies to address these gaps and provide a construct for appropriate counseling of the patient across the risk spectrum.
In recognition of 35 years of publication of OBG Management, this article on breast cancer screening by Mark D. Pearlman, MD, kicks off a series that focuses on various cancer screening modalities and expert recommendations.
Stay tuned for articles on the future of cervical cancer screening and genetic testing for cancer risk beyond BRCA testing.
We look forward to continuing OBG Management’s mission of enhancing the quality of reproductive health care and the professional development of ObGyns and all women’s health care clinicians.
Breast cancer risk
Variables that affect risk
While female sex and older age are the 2 greatest risks for the development of breast cancer, many other factors can either increase or decrease breast cancer risk in a person’s lifetime. The importance of identifying risk factors is 3-fold:
- to perform risk assessment to determine if individuals would benefit from average-risk versus high-risk breast cancer surveillance
- to identify persons who might benefit from BRCA genetic counseling and screening, risk reduction medications or procedures, and
- to allow patients to determine whether any modification in their lifestyle or reproductive choices would make sense to them to reduce their future breast cancer risk.
Most of these risk variables are largely inalterable (for example, family history of breast cancer, carriage of genetic pathogenic variants such as BRCA1 and BRCA2, age of menarche and menopause), but some are potentially modifiable, such as parity, age at first birth, lactation and duration, and dietary factors, among others. TABLE 2 lists common breast cancer risk factors.

Breast cancer risk assessment
Several validated tools have been developed to estimate a person’s breast cancer risk (TABLE 3). These tools combine known risk factors and, depending on the specific tool, can provide estimates of 5-year, 10-year, or lifetime risk of breast cancer. Patients at highest risk can benefit from earlier screening, supplemental screening with breast magnetic resonance imaging (MRI), or risk reduction (see the section, “High-risk screening”). Ideally, a risk assessment should be done by age 30 so that patients at high risk can be identified for earlier or more intensive screening and for possible genetic testing in those at risk for carriage of the BRCA or other breast cancer gene pathogenic variants.5,7

Continue to: Breast cancer screening: Efficacy and harms...
Breast cancer screening: Efficacy and harms
The earliest studies of breast cancer screening with mammography were randomized controlled trials (RCTs) that compared screened and unscreened patients aged 40 to 74. Nearly all the RCTs and numerous well-designed incidence-based and case-control studies have demonstrated that SM results in a clinically and statistically significant reduction in breast cancer mortality (TABLE 4).4,6,8 Since the mid-1980s and continuing to the current day, SM programs are routinely recommended in the United States. In addition to the mortality benefit outlined in TABLE 4, SM also is associated with a need for less invasive treatments if breast cancer is diagnosed.9,10

With several decades of experience, SM programs have demonstrated that multiple harms are associated with SM, including callbacks, false-positive mammograms that result in a benign biopsy, and overdiagnosis of breast cancer (TABLE 4). Overdiagnosis is a mammographic detection of a breast cancer that would not have harmed that woman in her lifetime. Overdiagnosis leads to overtreatment of breast cancers with its attendant side effects, the emotional harms of a breast cancer diagnosis, and the substantial financial cost of cancer treatment. Estimates of overdiagnosis range from 0% to 50%, with the most likely estimate of invasive breast cancer overdiagnosis from SM between 5% and 15%.11-13 Some of these overdiagnosed cancers are due to very slow growing cancers or breast cancers that may even regress. However, the higher rates of overdiagnosis occur in older persons who are screened and in whom competing causes of mortality become more prevalent. It is estimated that overdiagnosis of invasive breast cancer in patients younger than age 60 is less than 1%, but it exceeds 14% in those older than age 80 (TABLE 4).14
A structured approach is needed to counsel patients about SM so that they understand both the substantial benefit (earlier-stage diagnosis, reduced need for treatment, reduced breast cancer and all-cause mortality) and the potential harms (callback, false-positive results, and overdiagnosis). Moreover, the relative balance of the benefits and harms are influenced throughout their lifetime by both aging and changes in their personal and family medical history.
Counseling should consider factors beyond just the performance of mammography (sensitivity and specificity), such as the patient’s current health and age (competing causes of mortality), likelihood of developing breast cancer based on risk assessment (more benefit in higher-risk persons), and the individual patient’s values on the importance of the benefits and harms. The differing emphases on mammography performance and the relative value of the benefits and harms have led experts to produce disparate national guideline recommendations (TABLE 5).

Should SM start at age 40, 45, or 50 in average-risk persons?
There is not clear consensus about the age at which to begin to recommend routine SM in patients at average risk. The National Comprehensive Cancer Network (NCCN),7 American Cancer Society (ACS),4 and the US Preventive Services Task Force (USPSTF)5 recommend that those at average risk start SM at age 40, 45, and 50, respectively (TABLE 5). While the guideline groups listed in TABLE 5 agree that there is level 1 evidence that SM reduces breast cancer mortality in the general population for persons starting at age 40, because the incidence of breast cancer is lower in younger persons (TABLE 6),4 the net population-based screening benefit is lower in this group, and the number needed to invite to screening to save a single life due to breast cancer varies.

For patients in their 40s, it is estimated that 1,904 individuals need to be invited to SM to save 1 life, whereas for patients in their 50s, it is 1,339.15 However, for patients in their 40s, the number needed to screen to save 1 life due to breast cancer decreases from 1 in 1,904 if invited to be screened to 1 in 588 if they are actually screened.16 Furthermore, if a patient is diagnosed with breast cancer at age 40–50, the likelihood of dying is reduced at least 22% and perhaps as high as 48% if her cancer was diagnosed on SM compared with an unscreened individual with a symptomatic presentation (for example, palpable mass).4,15,17,18 Another benefit of SM in the fifth decade of life (40s) is the decreased need for more extensive treatment, including a higher risk of need for chemotherapy (odds ratio [OR], 2.81; 95% confidence interval [CI], 1.16–6.84); need for mastectomy (OR, 3.41; 95% CI, 1.36–8.52); and need for axillary lymph node dissection (OR, 5.76; 95% CI, 2.40–13.82) in unscreened (compared with screened) patients diagnosed with breast cancer.10
The harms associated with SM are not inconsequential and include callbacks (approximately 1 in 10), false-positive biopsy (approximately 1 in 100), and overdiagnosis (likely <1% of all breast cancers in persons younger than age 50). Because most patients in their 40s will not develop breast cancer (TABLE 6), the benefit of reduced breast cancer mortality will not be experienced by most in this decade of life, but they are still just as likely to experience a callback, false-positive biopsy, or the possibility of overdiagnosis. Interpretation of this balance on a population level is the crux of the various guideline groups’ development of differing recommendations as to when screening should start. Despite this seeming disagreement, all the guideline groups listed in TABLE 5 concur that persons at average risk for breast cancer should be offered SM if they desire starting at age 40 after a shared decision-making conversation that incorporates the patient’s view on the relative value of the benefits and risks.
Continue to: High-risk screening...
High-risk screening
Unlike in screening average-risk patients, there is less disagreement about screening in high-risk groups. TABLE 7 outlines the various categories and recommended strategies that qualify for screening at younger ages or more intensive screening. Adding breast MRI to SM in high-risk individuals results in both higher cancer detection rates and less interval breast cancers (cancers diagnosed between screening rounds) diagnosed compared with SM alone.19,20 Interval breast cancer tends to be more aggressive and is used as a surrogate marker for more recognized factors, such as breast cancer mortality. In addition to less interval breast cancers, high-risk patients are more likely to be diagnosed with node-negative disease if screening breast MRI is added to SM.
Long-term mortality benefit studies using MRI have not been conducted due to the prolonged follow-up times needed. Expense, lower specificity compared with mammography (that is, more false-positive results), and need for the use of gadolinium limit more widespread use of breast MRI screening in average-risk persons.

Screening in patients with dense breasts
Half of patients undergoing SM in the United States have dense breasts (heterogeneously dense breasts, 40%; extremely dense breasts, 10%). Importantly, increasing breast density is associated with a lower cancer detection rate with SM and is an independent risk factor for developing breast cancer. While most states already require patients to be notified if they have dense breasts identified on SM, the US Food and Drug Administration will soon make breast density patient notification a national standard (see: https://delauro.house.gov/media-center/press-releases/delauro-secures-timeline-fda-rollout-breast-density-notification-rule).
Most of the risk assessment tools listed in TABLE 3 incorporate breast density into their calculation of breast cancer risk. If that calculation places a patient into one of the highest-risk groups (based on additional factors like strong family history of breast cancer, reproductive risk factors, BRCA carriage, and so on), more intensive surveillance should be recommended (TABLE 7).7 However, once these risk calculations are done, most persons with dense breasts will remain in an average-risk category.
Because of the frequency and risks associated with dense breasts, different and alternative strategies have been recommended for screening persons who are at average risk with dense breasts. Supplemental screening with MRI, ultrasonography, contrast-enhanced mammography, and molecular breast imaging are all being considered but have not been studied sufficiently to demonstrate mortality benefit or cost-effectiveness.
Of all the supplemental modalities used to screen patients with dense breasts, MRI has been the best studied. A large RCT in the Netherlands evaluated supplemental MRI screening in persons with extremely dense breasts after a negative mammogram.21 Compared with no supplemental screening, the MRI group had 17 additional cancers detected per 1,000 screened and a 50% reduction in interval breast cancers; in addition, MRI was associated with a positive predictive value of 26% for biopsies. At present, high cost and limited access to standard breast MRI has not allowed its routine use for persons with dense breasts in the United States, but this may change with more experience and more widespread introduction and experience with abbreviated (or rapid) breast MRI in the future (TABLE 8).

Equitable screening
Black persons who are diagnosed with breast cancer have a 40% higher risk of dying than White patients due to multiple factors, including systemic racial factors (implicit and unconscious bias), reduced access to care, and a lower likelihood of receiving standard of care once diagnosed.22-24 In addition, Black patients have twice the likelihood of being diagnosed with triple-negative breast cancers, a biologically more aggressive tumor.22-24 Among Black, Asian, and Hispanic persons diagnosed with breast cancer, one-third are diagnosed younger than age 50, which is higher than for non-Hispanic White persons. Prior to the age of 50, Black, Asian, and Hispanic patients also have a 72% more likelihood of being diagnosed with invasive breast cancer, have a 58% greater risk of advanced-stage disease, and have a 127% higher risk of dying from breast cancer compared with White patients.25,26 Based on all of these factors, delaying SM until age 50 may adversely affect the Black, Asian, and Hispanic populations.
Persons in the LGBTQ+ community do not present for SM as frequently as the general population, often because they feel threatened or unwelcome.27 Clinicians and breast imaging units should review their inclusivity policies and training to provide a welcoming and respectful environment to all persons in an effort to reduce these barriers. While data are limited and largely depend on expert opinion, current recommendations for screening in the transgender patient depend on sex assigned at birth, the type and duration of hormone use, and surgical history. In patients assigned female sex at birth, average-risk and high-risk screening recommendations are similar to those for the general population unless bilateral mastectomy has been performed.28 In transfeminine patients who have used hormones for longer than 5 years, some groups recommend annual screening starting at age 40, although well-designed studies are lacking.29
Continue to: We have done well, can we do better?...
We have done well, can we do better?
Screening mammography clearly has been an important and effective tool in the effort to reduce breast cancer mortality, but there are clear limitations. These include moderate sensitivity of mammography, particularly in patients with dense breasts, and a specificity that results in either callbacks (10%), breast biopsies for benign disease (1%), or the reality of overdiagnosis, which becomes increasingly important in older patients.
With the introduction of mammography in the mid-1980s, a one-size-fits-all approach has proved challenging more recently due to an increased recognition of the harms of screening. As a result of this evolving understanding, different recommendations for average-risk screening have emerged. With the advent of breast MRI, risk-based screening is an important but underutilized tool to identify highest-risk individuals, which is associated with improved cancer detection rates, reduced node-positive disease, and fewer diagnosed interval breast cancers. Assuring that nearly all of this highest-risk group is identified through routine breast cancer risk assessment remains a challenge for clinicians.
But what SM recommendations should be offered to persons who fall into an intermediate-risk group (15%–20%), very low-risk groups (<5%), or patients with dense breasts? These are challenges that could be met through novel and individualized approaches (for example, polygenic risk scoring, further research on newer modalities of screening [TABLE 8]), improved screening algorithms for persons with dense breasts, and enhanced clinician engagement to achieve universal breast cancer and BRCA risk assessment of patients by age 25 to 30.
In 2023, best practice and consensus guidelines for intermediate- and low-risk breast cancer groups remain unclear, and one of the many ongoing challenges is to further reduce the impact of breast cancer on the lives of persons affected and the recognized harms of SM.
In the meantime, there is consensus in average-risk patients to provide counseling about SM by age 40. My approach has been to counsel all average-risk patients on the risks and benefits of mammography using the acronym TIP-V:
- Use a Tool to calculate breast cancer risk (TABLE 3). If they are at high risk, provide recommendations for high-risk management (TABLE 7).7
- For average-risk patients, counsel that their Incidence of developing breast cancer in the next decade is approximately 1 in 70 (TABLE 6).4
- Provide data and guidance on the benefits of SM for patients in their 40s (mortality improvement, decreased treatment) and the likelihood of harm from breast cancer screening (10% callback, 1% benign biopsy, and <1% likelihood of overdiagnosis [TABLE 4]).4,14,15
- Engage the patient to better understand their relative Values of the benefits and harms and make a shared decision on screening starting at age 40, 45, or 50.
Looking forward
In summary, SM remains an important tool in the effort to decrease the risk of mortality due to breast cancer. Given the limitations of SM, however, newer tools and methods—abbreviated MRI, contrast-enhanced mammography, molecular breast imaging, customized screening intervals depending on individual risk/polygenic risk score, and customized counseling and screening based on risk factors (TABLES 2 and 7)—will play an increased role in recommendations for breast cancer screening in the future. ●
- Giaquinto AN, Sung H, Miller KD, et al. Breast cancer statistics, 2022. CA Cancer J Clin. 2022;72:524-541.
- Berry DA, Cronin KA, Plevritis SK, et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med. 2005;353:1784-1792.
- Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209-249.
- Oeffinger KC, Fontham ET, Etzioni R, et al; American Cancer Society. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314:1599-1614.
- US Preventive Services Task Force; Owens DK, Davidson KW, Drist AH, et al. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer: US Preventive Services Task Force Recommendation statement. JAMA. 2019;322:652-665.
- Nelson HD, Cantor A, Humphrey L, et al. Screening for breast cancer: a systematic review to update the 2009 US Preventive Services Task Force recommendation. Evidence synthesis no 124. AHRQ publication no 14-05201-EF-1. Rockville, MD: Agency for Healthcare Research and Quality; 2016.
- Bevers TB, Helvie M, Bonaccio E, et al. Breast cancer screening and diagnosis, version 3.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2018;16:1362-1389.
- Duffy SW, Vulkan D, Cuckle H, et al. Effect of mammographic screening from age 40 years on breast cancer mortality (UK Age trial): final results of a randomised, controlled trial. Lancet Oncol. 2020;21:1165-1172.
- Karzai S, Port E, Siderides C, et al. Impact of screening mammography on treatment in young women diagnosed with breast cancer. Ann Surg Oncol. 2022. doi:10.1245/ s10434-022-11581-6.
- Ahn S, Wooster M, Valente C, et al. Impact of screening mammography on treatment in women diagnosed with breast cancer. Ann Surg Oncol. 2018;25:2979-2986.
- Coldman A, Phillips N. Incidence of breast cancer and estimates of overdiagnosis after the initiation of a population-based mammography screening program. CMAJ. 2013;185:E492-E498.
- Etzioni R, Gulati R, Mallinger L, et al. Influence of study features and methods on overdiagnosis estimates in breast and prostate cancer screening. Ann Internal Med. 2013;158:831-838.
- Ryser MD, Lange J, Inoue LY, et al. Estimation of breast cancer overdiagnosis in a US breast screening cohort. Ann Intern Med. 2022;175:471-478.
- Monticciolo DL, Malak SF, Friedewald SM, et al. Breast cancer screening recommendations inclusive of all women at average risk: update from the ACR and Society of Breast Imaging. J Am Coll Radiol. 2021;18:1280-1288.
- Nelson HD, Fu R, Cantor A, Pappas M, et al. Effectiveness of breast cancer screening: systematic review and meta-analysis to update the 2009 US Preventive Services Task Force recommendation. Ann Internal Med. 2016;164:244-255.
- Hendrick RE, Helvie MA, Hardesty LA. Implications of CISNET modeling on number needed to screen and mortality reduction with digital mammography in women 40–49 years old. Am J Roentgenol. 2014;203:1379-1381.
- Broeders M, Moss S, Nyström L, et al; EUROSCREEN Working Group. The impact of mammographic screening on breast cancer mortality in Europe: a review of observational studies. J Med Screen. 2012;19(suppl 1):14-25.
- Tabár L, Yen AMF, Wu WYY, et al. Insights from the breast cancer screening trials: how screening affects the natural history of breast cancer and implications for evaluating service screening programs. Breast J. 2015;21:13-20.
- Kriege M, Brekelmans CTM, Boetes C, et al; Magnetic Resonance Imaging Screening Study Group. Efficacy of MRI and mammography for breast-cancer screening in women with a familial or genetic predisposition. N Engl J Med. 2004;351:427-437.
- Vreemann S, Gubern-Merida A, Lardenoije S, et al. The frequency of missed breast cancers in women participating in a high-risk MRI screening program. Breast Cancer Res Treat. 2018;169:323-331.
- Bakker MF, de Lange SV, Pijnappel RM, et al. Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med. 2019;381:2091-2102.
- Amirikia KC, Mills P, Bush J, et al. Higher population‐based incidence rates of triple‐negative breast cancer among young African‐American women: implications for breast cancer screening recommendations. Cancer. 2011;117:2747-2753.
- Kohler BA, Sherman RL, Howlader N, et al. Annual report to the nation on the status of cancer, 1975-2011, featuring incidence of breast cancer subtypes by race/ethnicity, poverty, and state. J Natl Cancer Inst. 2015;107:djv048.
- Newman LA, Kaljee LM. Health disparities and triple-negative breast cancer in African American women: a review. JAMA Surg. 2017;152:485-493.
- Stapleton SM, Oseni TO, Bababekov YJ, et al. Race/ethnicity and age distribution of breast cancer diagnosis in the United States. JAMA Surg. 2018;153:594-595.
- Hendrick RE, Monticciolo DL, Biggs KW, et al. Age distributions of breast cancer diagnosis and mortality by race and ethnicity in US women. Cancer. 2021;127:4384-4392.
- Perry H, Fang AJ, Tsai EM, et al. Imaging health and radiology care of transgender patients: a call to build evidence-based best practices. J Am Coll Radiol. 2021;18(3 pt B):475-480.
- Lockhart R, Kamaya A. Patient-friendly summary of the ACR Appropriateness Criteria: transgender breast cancer screening. J Am Coll Radiol. 2022;19:e19.
- Expert Panel on Breast Imaging; Brown A, Lourenco AP, Niell BL, et al. ACR Appropriateness Criteria transgender breast cancer screening. J Am Coll Radiol. 2021;18:S502-S515.
- Mørch LS, Skovlund CW, Hannaford PC, et al. Contemporary hormonal contraception and the risk of breast cancer. N Engl J Med. 2017;377:2228-2239.
- Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7-33.
- Laws A, Katlin F, Hans M, et al. Screening MRI does not increase cancer detection or result in an earlier stage at diagnosis for patients with high-risk breast lesions: a propensity score analysis. Ann Surg Oncol. 2023;30;68-77.
- American College of Obstetricians and Gynecologists. Practice bulletin no 179: Breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130:e1-e16.
- Grimm LJ, Mango VL, Harvey JA, et al. Implementation of abbreviated breast MRI for screening: AJR expert panel narrative review. AJR Am J Roentgenol. 2022;218:202-212.
- Potsch N, Vatteroini G, Clauser P, et al. Contrast-enhanced mammography versus contrast-enhanced breast MRI: a systematic review and meta-analysis. Radiology. 2022;305:94-103.
- Covington MF, Parent EE, Dibble EH, et al. Advances and future directions in molecular breast imaging. J Nucl Med. 2022;63:17-21.
- Giaquinto AN, Sung H, Miller KD, et al. Breast cancer statistics, 2022. CA Cancer J Clin. 2022;72:524-541.
- Berry DA, Cronin KA, Plevritis SK, et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med. 2005;353:1784-1792.
- Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209-249.
- Oeffinger KC, Fontham ET, Etzioni R, et al; American Cancer Society. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314:1599-1614.
- US Preventive Services Task Force; Owens DK, Davidson KW, Drist AH, et al. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer: US Preventive Services Task Force Recommendation statement. JAMA. 2019;322:652-665.
- Nelson HD, Cantor A, Humphrey L, et al. Screening for breast cancer: a systematic review to update the 2009 US Preventive Services Task Force recommendation. Evidence synthesis no 124. AHRQ publication no 14-05201-EF-1. Rockville, MD: Agency for Healthcare Research and Quality; 2016.
- Bevers TB, Helvie M, Bonaccio E, et al. Breast cancer screening and diagnosis, version 3.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2018;16:1362-1389.
- Duffy SW, Vulkan D, Cuckle H, et al. Effect of mammographic screening from age 40 years on breast cancer mortality (UK Age trial): final results of a randomised, controlled trial. Lancet Oncol. 2020;21:1165-1172.
- Karzai S, Port E, Siderides C, et al. Impact of screening mammography on treatment in young women diagnosed with breast cancer. Ann Surg Oncol. 2022. doi:10.1245/ s10434-022-11581-6.
- Ahn S, Wooster M, Valente C, et al. Impact of screening mammography on treatment in women diagnosed with breast cancer. Ann Surg Oncol. 2018;25:2979-2986.
- Coldman A, Phillips N. Incidence of breast cancer and estimates of overdiagnosis after the initiation of a population-based mammography screening program. CMAJ. 2013;185:E492-E498.
- Etzioni R, Gulati R, Mallinger L, et al. Influence of study features and methods on overdiagnosis estimates in breast and prostate cancer screening. Ann Internal Med. 2013;158:831-838.
- Ryser MD, Lange J, Inoue LY, et al. Estimation of breast cancer overdiagnosis in a US breast screening cohort. Ann Intern Med. 2022;175:471-478.
- Monticciolo DL, Malak SF, Friedewald SM, et al. Breast cancer screening recommendations inclusive of all women at average risk: update from the ACR and Society of Breast Imaging. J Am Coll Radiol. 2021;18:1280-1288.
- Nelson HD, Fu R, Cantor A, Pappas M, et al. Effectiveness of breast cancer screening: systematic review and meta-analysis to update the 2009 US Preventive Services Task Force recommendation. Ann Internal Med. 2016;164:244-255.
- Hendrick RE, Helvie MA, Hardesty LA. Implications of CISNET modeling on number needed to screen and mortality reduction with digital mammography in women 40–49 years old. Am J Roentgenol. 2014;203:1379-1381.
- Broeders M, Moss S, Nyström L, et al; EUROSCREEN Working Group. The impact of mammographic screening on breast cancer mortality in Europe: a review of observational studies. J Med Screen. 2012;19(suppl 1):14-25.
- Tabár L, Yen AMF, Wu WYY, et al. Insights from the breast cancer screening trials: how screening affects the natural history of breast cancer and implications for evaluating service screening programs. Breast J. 2015;21:13-20.
- Kriege M, Brekelmans CTM, Boetes C, et al; Magnetic Resonance Imaging Screening Study Group. Efficacy of MRI and mammography for breast-cancer screening in women with a familial or genetic predisposition. N Engl J Med. 2004;351:427-437.
- Vreemann S, Gubern-Merida A, Lardenoije S, et al. The frequency of missed breast cancers in women participating in a high-risk MRI screening program. Breast Cancer Res Treat. 2018;169:323-331.
- Bakker MF, de Lange SV, Pijnappel RM, et al. Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med. 2019;381:2091-2102.
- Amirikia KC, Mills P, Bush J, et al. Higher population‐based incidence rates of triple‐negative breast cancer among young African‐American women: implications for breast cancer screening recommendations. Cancer. 2011;117:2747-2753.
- Kohler BA, Sherman RL, Howlader N, et al. Annual report to the nation on the status of cancer, 1975-2011, featuring incidence of breast cancer subtypes by race/ethnicity, poverty, and state. J Natl Cancer Inst. 2015;107:djv048.
- Newman LA, Kaljee LM. Health disparities and triple-negative breast cancer in African American women: a review. JAMA Surg. 2017;152:485-493.
- Stapleton SM, Oseni TO, Bababekov YJ, et al. Race/ethnicity and age distribution of breast cancer diagnosis in the United States. JAMA Surg. 2018;153:594-595.
- Hendrick RE, Monticciolo DL, Biggs KW, et al. Age distributions of breast cancer diagnosis and mortality by race and ethnicity in US women. Cancer. 2021;127:4384-4392.
- Perry H, Fang AJ, Tsai EM, et al. Imaging health and radiology care of transgender patients: a call to build evidence-based best practices. J Am Coll Radiol. 2021;18(3 pt B):475-480.
- Lockhart R, Kamaya A. Patient-friendly summary of the ACR Appropriateness Criteria: transgender breast cancer screening. J Am Coll Radiol. 2022;19:e19.
- Expert Panel on Breast Imaging; Brown A, Lourenco AP, Niell BL, et al. ACR Appropriateness Criteria transgender breast cancer screening. J Am Coll Radiol. 2021;18:S502-S515.
- Mørch LS, Skovlund CW, Hannaford PC, et al. Contemporary hormonal contraception and the risk of breast cancer. N Engl J Med. 2017;377:2228-2239.
- Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7-33.
- Laws A, Katlin F, Hans M, et al. Screening MRI does not increase cancer detection or result in an earlier stage at diagnosis for patients with high-risk breast lesions: a propensity score analysis. Ann Surg Oncol. 2023;30;68-77.
- American College of Obstetricians and Gynecologists. Practice bulletin no 179: Breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130:e1-e16.
- Grimm LJ, Mango VL, Harvey JA, et al. Implementation of abbreviated breast MRI for screening: AJR expert panel narrative review. AJR Am J Roentgenol. 2022;218:202-212.
- Potsch N, Vatteroini G, Clauser P, et al. Contrast-enhanced mammography versus contrast-enhanced breast MRI: a systematic review and meta-analysis. Radiology. 2022;305:94-103.
- Covington MF, Parent EE, Dibble EH, et al. Advances and future directions in molecular breast imaging. J Nucl Med. 2022;63:17-21.
Omit radiation in older women with low-risk, ER+ breast cancer
say researchers reporting 10-year outcomes from the large phase 3 trial known as PRIME II.
“Our trial provides robust evidence indicating that irradiation can be safely omitted in women 65 years of age or older who have grade 1 or 2 ER-high cancers treated by breast-conserving therapy, provided that they receive 5 years of adjuvant endocrine therapy,” concluded investigators led by Ian Kunkler, MB, a clinical oncology professor at the University of Edinburgh.
The trial randomly assigned 1,326 women who had undergone a lumpectomy to either whole-breast irradiation or no radiation on a background of tamoxifen.
The incidence of local recurrence was lower with radiation (0.9% vs. 9.5%), but there was no significant difference in distant metastases or breast cancer–specific or overall survival.
The findings will “help clinicians guide older patients on whether this particular aspect of early breast cancer treatment can be omitted,” Dr. Kunkler said in a press release. Radiation carries risks of heart and lung damage, and these results show that skipping it does not increase the odds of dying from breast cancer.
The new study was published in the New England Journal of Medicine.
“Any doubt that radiotherapy cannot be omitted in women” who meet the criteria “can be put to rest,” commented breast radiation oncologists Alice Ho, MD, of Duke University in Durham, N.C., and Jennifer Bellon, MD, of Harvard Medical School, Boston, in an accompanying editorial.
Clinical guidelines already support omitting radiation therapy in older women with low-risk tumors treated with lumpectomy and endocrine therapy, but the move has been controversial owing to a lack of long-term data, and use of radiation for such women remains common in the United States, the investigators explain.
The “highly anticipated” results for 10-year outcomes from this trial should help address that issue, as well as “the long-standing problem of overtreatment in older women with low-risk breast cancer,” the editorialists comment.
Study details
PRIME II was conducted from 2003 to 2009 mainly in the United Kingdom. Participants were aged 65 years or older and had T1 or T2 ER-positive tumors no larger than 3 cm and were without nodal involvement.
Following lumpectomies with clear margins, the women underwent endocrine therapy; the investigators recommended tamoxifen at 20 mg/day for 5 years.
Women who were randomly assigned to radiation also received 40-50 Gy of whole-breast irradiation in 20-25 fractions over 3-5 weeks.
At 10 years, 1.6% of women in the no-radiation arm had distant metastases as their first recurrence vs. 3% of women who underwent radiation.
Ten-year breast cancer–specific survival was 97.9% with radiation and 97.4% with no radiation. Ten-year overall survival was 80.7% in the radiotherapy arm vs. 80.8% in the no-radiotherapy group.
In addition, the recurrence rate was lower after radiation. The investigators suggest that lower adherence to endocrine therapy and lower levels of ER positivity increased the risk of local recurrence among women who didn’t receive radiation.
Almost 10% of the women who did not receive radiation had local recurrences by 10 years, but the investigators note that if tumors do recur locally, women still have the option of a second lumpectomy, and if they so choose, they can then receive radiation, so local recurrence “does not necessarily mean loss of the breast.”
PRIME II was funded by the Scottish Government’s chief scientist office and the Breast Cancer Institute at Western General Hospital, Edinburgh. Dr. Kunkler reported no conflicts of interest. A coauthor has acted as a speaker, adviser, and/or researcher for many companies, including Hoffmann-La Roche, Exact Sciences, and Eli Lilly. Dr. Ho reported grants from and/or being a consultant for GlaxoSmithKline, Roche, Merck, and others. Dr. Bellon reported ties to Varian Medical Systems and Veracyte.
A version of this article originally appeared on Medscape.com.
say researchers reporting 10-year outcomes from the large phase 3 trial known as PRIME II.
“Our trial provides robust evidence indicating that irradiation can be safely omitted in women 65 years of age or older who have grade 1 or 2 ER-high cancers treated by breast-conserving therapy, provided that they receive 5 years of adjuvant endocrine therapy,” concluded investigators led by Ian Kunkler, MB, a clinical oncology professor at the University of Edinburgh.
The trial randomly assigned 1,326 women who had undergone a lumpectomy to either whole-breast irradiation or no radiation on a background of tamoxifen.
The incidence of local recurrence was lower with radiation (0.9% vs. 9.5%), but there was no significant difference in distant metastases or breast cancer–specific or overall survival.
The findings will “help clinicians guide older patients on whether this particular aspect of early breast cancer treatment can be omitted,” Dr. Kunkler said in a press release. Radiation carries risks of heart and lung damage, and these results show that skipping it does not increase the odds of dying from breast cancer.
The new study was published in the New England Journal of Medicine.
“Any doubt that radiotherapy cannot be omitted in women” who meet the criteria “can be put to rest,” commented breast radiation oncologists Alice Ho, MD, of Duke University in Durham, N.C., and Jennifer Bellon, MD, of Harvard Medical School, Boston, in an accompanying editorial.
Clinical guidelines already support omitting radiation therapy in older women with low-risk tumors treated with lumpectomy and endocrine therapy, but the move has been controversial owing to a lack of long-term data, and use of radiation for such women remains common in the United States, the investigators explain.
The “highly anticipated” results for 10-year outcomes from this trial should help address that issue, as well as “the long-standing problem of overtreatment in older women with low-risk breast cancer,” the editorialists comment.
Study details
PRIME II was conducted from 2003 to 2009 mainly in the United Kingdom. Participants were aged 65 years or older and had T1 or T2 ER-positive tumors no larger than 3 cm and were without nodal involvement.
Following lumpectomies with clear margins, the women underwent endocrine therapy; the investigators recommended tamoxifen at 20 mg/day for 5 years.
Women who were randomly assigned to radiation also received 40-50 Gy of whole-breast irradiation in 20-25 fractions over 3-5 weeks.
At 10 years, 1.6% of women in the no-radiation arm had distant metastases as their first recurrence vs. 3% of women who underwent radiation.
Ten-year breast cancer–specific survival was 97.9% with radiation and 97.4% with no radiation. Ten-year overall survival was 80.7% in the radiotherapy arm vs. 80.8% in the no-radiotherapy group.
In addition, the recurrence rate was lower after radiation. The investigators suggest that lower adherence to endocrine therapy and lower levels of ER positivity increased the risk of local recurrence among women who didn’t receive radiation.
Almost 10% of the women who did not receive radiation had local recurrences by 10 years, but the investigators note that if tumors do recur locally, women still have the option of a second lumpectomy, and if they so choose, they can then receive radiation, so local recurrence “does not necessarily mean loss of the breast.”
PRIME II was funded by the Scottish Government’s chief scientist office and the Breast Cancer Institute at Western General Hospital, Edinburgh. Dr. Kunkler reported no conflicts of interest. A coauthor has acted as a speaker, adviser, and/or researcher for many companies, including Hoffmann-La Roche, Exact Sciences, and Eli Lilly. Dr. Ho reported grants from and/or being a consultant for GlaxoSmithKline, Roche, Merck, and others. Dr. Bellon reported ties to Varian Medical Systems and Veracyte.
A version of this article originally appeared on Medscape.com.
say researchers reporting 10-year outcomes from the large phase 3 trial known as PRIME II.
“Our trial provides robust evidence indicating that irradiation can be safely omitted in women 65 years of age or older who have grade 1 or 2 ER-high cancers treated by breast-conserving therapy, provided that they receive 5 years of adjuvant endocrine therapy,” concluded investigators led by Ian Kunkler, MB, a clinical oncology professor at the University of Edinburgh.
The trial randomly assigned 1,326 women who had undergone a lumpectomy to either whole-breast irradiation or no radiation on a background of tamoxifen.
The incidence of local recurrence was lower with radiation (0.9% vs. 9.5%), but there was no significant difference in distant metastases or breast cancer–specific or overall survival.
The findings will “help clinicians guide older patients on whether this particular aspect of early breast cancer treatment can be omitted,” Dr. Kunkler said in a press release. Radiation carries risks of heart and lung damage, and these results show that skipping it does not increase the odds of dying from breast cancer.
The new study was published in the New England Journal of Medicine.
“Any doubt that radiotherapy cannot be omitted in women” who meet the criteria “can be put to rest,” commented breast radiation oncologists Alice Ho, MD, of Duke University in Durham, N.C., and Jennifer Bellon, MD, of Harvard Medical School, Boston, in an accompanying editorial.
Clinical guidelines already support omitting radiation therapy in older women with low-risk tumors treated with lumpectomy and endocrine therapy, but the move has been controversial owing to a lack of long-term data, and use of radiation for such women remains common in the United States, the investigators explain.
The “highly anticipated” results for 10-year outcomes from this trial should help address that issue, as well as “the long-standing problem of overtreatment in older women with low-risk breast cancer,” the editorialists comment.
Study details
PRIME II was conducted from 2003 to 2009 mainly in the United Kingdom. Participants were aged 65 years or older and had T1 or T2 ER-positive tumors no larger than 3 cm and were without nodal involvement.
Following lumpectomies with clear margins, the women underwent endocrine therapy; the investigators recommended tamoxifen at 20 mg/day for 5 years.
Women who were randomly assigned to radiation also received 40-50 Gy of whole-breast irradiation in 20-25 fractions over 3-5 weeks.
At 10 years, 1.6% of women in the no-radiation arm had distant metastases as their first recurrence vs. 3% of women who underwent radiation.
Ten-year breast cancer–specific survival was 97.9% with radiation and 97.4% with no radiation. Ten-year overall survival was 80.7% in the radiotherapy arm vs. 80.8% in the no-radiotherapy group.
In addition, the recurrence rate was lower after radiation. The investigators suggest that lower adherence to endocrine therapy and lower levels of ER positivity increased the risk of local recurrence among women who didn’t receive radiation.
Almost 10% of the women who did not receive radiation had local recurrences by 10 years, but the investigators note that if tumors do recur locally, women still have the option of a second lumpectomy, and if they so choose, they can then receive radiation, so local recurrence “does not necessarily mean loss of the breast.”
PRIME II was funded by the Scottish Government’s chief scientist office and the Breast Cancer Institute at Western General Hospital, Edinburgh. Dr. Kunkler reported no conflicts of interest. A coauthor has acted as a speaker, adviser, and/or researcher for many companies, including Hoffmann-La Roche, Exact Sciences, and Eli Lilly. Dr. Ho reported grants from and/or being a consultant for GlaxoSmithKline, Roche, Merck, and others. Dr. Bellon reported ties to Varian Medical Systems and Veracyte.
A version of this article originally appeared on Medscape.com.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE