User login
Flurry of cancer drug endorsements from EU panel
The CHMP recommended the granting of a conditional marketing authorization for pralsetinib (Gavreto) for the treatment of non–small cell lung cancer (NSCLC).
Specifically, pralsetinib is indicated as monotherapy for the treatment of adult patients with rearranged during transfection (RET) fusion-positive advanced NSCLC not previously treated with a RET inhibitor.
Available as 100 mg capsules, pralsetinib is a RET-receptor tyrosine kinase inhibitor, targeting oncogenic RET fusion proteins (KIF5B-RET and CCDC6-RET).
Pralsetinib’s benefits are its objective response rate and response duration in patients with RET-fusion positive NSCLC, as observed in a pivotal phase 1/2, open-label, multi-cohort, single-arm study.
The most common side effects are anemia, increased aspartate aminotransferase, neutropenia, constipation, musculoskeletal pain, fatigue, leukopenia, increased alanine aminotransferase, and hypertension.
CHMP also recommended ripretinib (Qinlock) for the treatment of adult patients with advanced gastrointestinal stromal tumor (GIST) who have received prior treatment with three or more kinase inhibitors, including imatinib (Gleevec).
Available as 50 mg tablets, ripretinib is a protein kinase inhibitor designed to selectively block the oncogenic KIT and PDGFRA kinases by inhibiting their active conformation.
Ripretinib improved progression-free survival in patients with GIST.
The most common side effects are fatigue, alopecia, nausea, myalgia, constipation, diarrhea, palmar-plantar erythrodysesthesia syndrome, weight loss, and vomiting.
The third drug recommended for approval was zanubrutinib (Brukinsa) for the treatment of adult patients with Waldenström’s macroglobulinemia who have received at least one prior therapy or who are to receive the drug as first-line treatment (and are unsuitable for chemo-immunotherapy).
Available as 80 mg capsules, zanubrutinib is a Bruton’s tyrosine kinase inhibitor that blocks the activity of BTK, inactivating the pathways necessary for B-cell proliferation, trafficking, chemotaxis, and adhesion.
Zanubrutinib has demonstrated a clinically meaningful rate of very good partial response and/or complete response.
The most common side effects are neutropenia, thrombocytopenia, upper respiratory tract infection, hemorrhage/hematoma, rash, bruising, anemia, musculoskeletal pain, diarrhea, pneumonia, and cough.
Two new indications for already marketed drugs
CHMP also recommended an extension of the indications for two immunotherapies.
Pembrolizumab (Keytruda) will now also have an indication for use in combination with chemotherapy for the treatment of locally recurrent unresectable or metastatic triple negative breast cancer in adults whose tumors express PD-L1 with a CPS greater than or equal to 10 and who have not received prior chemotherapy for metastatic disease
Nivolumab (Opdivo) received an extension of indication to include use, in combination with fluoropyrimidine- and platinum-based combination chemotherapy, in the firstline treatment of adult patients with HER2 negative advanced or metastatic gastric, gastroesophageal junction, or esophageal adenocarcinoma whose tumors express PD-L1 with a combined positive score (CPS) greater than or equal to 5.
A version of this article first appeared on Medscape.com.
The CHMP recommended the granting of a conditional marketing authorization for pralsetinib (Gavreto) for the treatment of non–small cell lung cancer (NSCLC).
Specifically, pralsetinib is indicated as monotherapy for the treatment of adult patients with rearranged during transfection (RET) fusion-positive advanced NSCLC not previously treated with a RET inhibitor.
Available as 100 mg capsules, pralsetinib is a RET-receptor tyrosine kinase inhibitor, targeting oncogenic RET fusion proteins (KIF5B-RET and CCDC6-RET).
Pralsetinib’s benefits are its objective response rate and response duration in patients with RET-fusion positive NSCLC, as observed in a pivotal phase 1/2, open-label, multi-cohort, single-arm study.
The most common side effects are anemia, increased aspartate aminotransferase, neutropenia, constipation, musculoskeletal pain, fatigue, leukopenia, increased alanine aminotransferase, and hypertension.
CHMP also recommended ripretinib (Qinlock) for the treatment of adult patients with advanced gastrointestinal stromal tumor (GIST) who have received prior treatment with three or more kinase inhibitors, including imatinib (Gleevec).
Available as 50 mg tablets, ripretinib is a protein kinase inhibitor designed to selectively block the oncogenic KIT and PDGFRA kinases by inhibiting their active conformation.
Ripretinib improved progression-free survival in patients with GIST.
The most common side effects are fatigue, alopecia, nausea, myalgia, constipation, diarrhea, palmar-plantar erythrodysesthesia syndrome, weight loss, and vomiting.
The third drug recommended for approval was zanubrutinib (Brukinsa) for the treatment of adult patients with Waldenström’s macroglobulinemia who have received at least one prior therapy or who are to receive the drug as first-line treatment (and are unsuitable for chemo-immunotherapy).
Available as 80 mg capsules, zanubrutinib is a Bruton’s tyrosine kinase inhibitor that blocks the activity of BTK, inactivating the pathways necessary for B-cell proliferation, trafficking, chemotaxis, and adhesion.
Zanubrutinib has demonstrated a clinically meaningful rate of very good partial response and/or complete response.
The most common side effects are neutropenia, thrombocytopenia, upper respiratory tract infection, hemorrhage/hematoma, rash, bruising, anemia, musculoskeletal pain, diarrhea, pneumonia, and cough.
Two new indications for already marketed drugs
CHMP also recommended an extension of the indications for two immunotherapies.
Pembrolizumab (Keytruda) will now also have an indication for use in combination with chemotherapy for the treatment of locally recurrent unresectable or metastatic triple negative breast cancer in adults whose tumors express PD-L1 with a CPS greater than or equal to 10 and who have not received prior chemotherapy for metastatic disease
Nivolumab (Opdivo) received an extension of indication to include use, in combination with fluoropyrimidine- and platinum-based combination chemotherapy, in the firstline treatment of adult patients with HER2 negative advanced or metastatic gastric, gastroesophageal junction, or esophageal adenocarcinoma whose tumors express PD-L1 with a combined positive score (CPS) greater than or equal to 5.
A version of this article first appeared on Medscape.com.
The CHMP recommended the granting of a conditional marketing authorization for pralsetinib (Gavreto) for the treatment of non–small cell lung cancer (NSCLC).
Specifically, pralsetinib is indicated as monotherapy for the treatment of adult patients with rearranged during transfection (RET) fusion-positive advanced NSCLC not previously treated with a RET inhibitor.
Available as 100 mg capsules, pralsetinib is a RET-receptor tyrosine kinase inhibitor, targeting oncogenic RET fusion proteins (KIF5B-RET and CCDC6-RET).
Pralsetinib’s benefits are its objective response rate and response duration in patients with RET-fusion positive NSCLC, as observed in a pivotal phase 1/2, open-label, multi-cohort, single-arm study.
The most common side effects are anemia, increased aspartate aminotransferase, neutropenia, constipation, musculoskeletal pain, fatigue, leukopenia, increased alanine aminotransferase, and hypertension.
CHMP also recommended ripretinib (Qinlock) for the treatment of adult patients with advanced gastrointestinal stromal tumor (GIST) who have received prior treatment with three or more kinase inhibitors, including imatinib (Gleevec).
Available as 50 mg tablets, ripretinib is a protein kinase inhibitor designed to selectively block the oncogenic KIT and PDGFRA kinases by inhibiting their active conformation.
Ripretinib improved progression-free survival in patients with GIST.
The most common side effects are fatigue, alopecia, nausea, myalgia, constipation, diarrhea, palmar-plantar erythrodysesthesia syndrome, weight loss, and vomiting.
The third drug recommended for approval was zanubrutinib (Brukinsa) for the treatment of adult patients with Waldenström’s macroglobulinemia who have received at least one prior therapy or who are to receive the drug as first-line treatment (and are unsuitable for chemo-immunotherapy).
Available as 80 mg capsules, zanubrutinib is a Bruton’s tyrosine kinase inhibitor that blocks the activity of BTK, inactivating the pathways necessary for B-cell proliferation, trafficking, chemotaxis, and adhesion.
Zanubrutinib has demonstrated a clinically meaningful rate of very good partial response and/or complete response.
The most common side effects are neutropenia, thrombocytopenia, upper respiratory tract infection, hemorrhage/hematoma, rash, bruising, anemia, musculoskeletal pain, diarrhea, pneumonia, and cough.
Two new indications for already marketed drugs
CHMP also recommended an extension of the indications for two immunotherapies.
Pembrolizumab (Keytruda) will now also have an indication for use in combination with chemotherapy for the treatment of locally recurrent unresectable or metastatic triple negative breast cancer in adults whose tumors express PD-L1 with a CPS greater than or equal to 10 and who have not received prior chemotherapy for metastatic disease
Nivolumab (Opdivo) received an extension of indication to include use, in combination with fluoropyrimidine- and platinum-based combination chemotherapy, in the firstline treatment of adult patients with HER2 negative advanced or metastatic gastric, gastroesophageal junction, or esophageal adenocarcinoma whose tumors express PD-L1 with a combined positive score (CPS) greater than or equal to 5.
A version of this article first appeared on Medscape.com.
Immunotherapy for cancer patients with poor PS needs a rethink
The findings have prompted an expert to argue against the use of immunotherapy for such patients, who may have little time left and very little chance of benefiting.
“It is quite clear from clinical practice that most patients with limited PS do very poorly and do not benefit from immune check point inhibitors (ICI),” Jason Luke, MD, UPMC Hillman Cancer Center and the University of Pittsburgh, said in an email.
“So, my strong opinion is that patients should not be getting an immunotherapy just because it might not cause as many side effects as chemotherapy,” he added.
“Instead of giving an immunotherapy with little chance of success, patients and families deserve to have a direct conversation about what realistic expectations [might be] and how we as the oncology community can support them to achieve whatever their personal goals are in the time that they have left,” he emphasized.
Dr. Luke was the lead author of an editorial in which he commented on the study. Both the study and the editorial were published online in JCO Oncology Practice.
Variety of cancers
The study was conducted by Mridula Krishnan, MD, Nebraska Medicine Fred and Pamela Buffett Cancer Center, Omaha, Nebraska, and colleagues.
The team reviewed 257 patients who had been treated with either a programmed cell death protein–1 inhibitor or programmed cell death–ligand-1 inhibitor for a variety of advanced cancers. The drugs included pembrolizumab (Keytruda), nivolumab (Opdivo), atezolizumab (Tecentique), durvalumab (Imfinzi), and avelumab (Bavencio).
Most of the patients (71%) had good PS, with an Eastern Cooperative Oncology Group (ECOG) PS of 0-1 on initiation of immunotherapy; 29% of patients had poor PS, with an ECOG PS of greater than or equal to 2.
“The primary outcome was OS stratified by ECOG PS 0-1 versus ≥2,” note the authors. Across all tumor types, OS was superior for patients in the ECOG 0-1 PS group, the investigators note. The median OS was 12.6 months, compared with only 3.1 months for patients in the ECOG greater than or equal to 2 group (P < .001).
Moreover, overall response rates for patients with a poor PS were low. Only 8%, or 6 of 75 patients with an ECOG PS of greater than or equal to 2, achieved an objective response by RECIST criteria.
This compared to an overall response rate of 23% for patients with an ECOG PS of 0-1, the investigators note (P = .005).
Interestingly, the hospice referral rate for patients with a poor PS (67%) was similar to that of patients with a PS of 1-2 (61.9%), Dr. Krishnan and colleagues observe.
Those with a poor PS were more like to die in-hospital (28.6%) than were patients with a good PS (15.1%; P = .035). The authors point out that it is well known that outcomes with chemotherapy are worse among patients who experience a decline in functional reserve, owing to increased susceptibility to toxicity and complications.
“Regardless of age, patients with ECOG PS >2 usually have poor tolerability to chemotherapy, and this correlates with worse survival outcome,” they emphasize. There is as yet no clear guidance regarding the impact of PS on ICI treatment response, although “there should be,” Dr. Luke believes.
“In a patient with declining performance status, especially ECOG PS 3-4 but potentially 2 as well, there is little likelihood that the functional and immune reserve of the patient will be adequate to mount a robust antitumor response,” he elaborated.
“It’s not impossible, but trying for it should not come at the expense of engaging about end-of-life care and maximizing the palliative opportunities that many only have a short window of time in which to pursue,” he added.
Again, Dr. Luke strongly believes that just giving an ICI without engaging in a frank conversation with the patient and their families – which happens all too often, he feels – is absolutely not the way to go when treating patients with a poor PS and little time left.
“Patients and families might be better served by having a more direct and frank conversation about what the likelihood [is] that ICI therapy will actually do,” Dr. Luke stressed.
In their editorial, Dr. Luke and colleagues write: “Overall, we as an oncology community need to improve our communication with patients regarding goals of care and end-of-life considerations as opposed to reflexive treatment initiation,” he writes.
“Our duty, first and foremost, should focus on the person sitting in front of us – taking a step back may be the best way to move forward with compassionate care,” they add.
The authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The findings have prompted an expert to argue against the use of immunotherapy for such patients, who may have little time left and very little chance of benefiting.
“It is quite clear from clinical practice that most patients with limited PS do very poorly and do not benefit from immune check point inhibitors (ICI),” Jason Luke, MD, UPMC Hillman Cancer Center and the University of Pittsburgh, said in an email.
“So, my strong opinion is that patients should not be getting an immunotherapy just because it might not cause as many side effects as chemotherapy,” he added.
“Instead of giving an immunotherapy with little chance of success, patients and families deserve to have a direct conversation about what realistic expectations [might be] and how we as the oncology community can support them to achieve whatever their personal goals are in the time that they have left,” he emphasized.
Dr. Luke was the lead author of an editorial in which he commented on the study. Both the study and the editorial were published online in JCO Oncology Practice.
Variety of cancers
The study was conducted by Mridula Krishnan, MD, Nebraska Medicine Fred and Pamela Buffett Cancer Center, Omaha, Nebraska, and colleagues.
The team reviewed 257 patients who had been treated with either a programmed cell death protein–1 inhibitor or programmed cell death–ligand-1 inhibitor for a variety of advanced cancers. The drugs included pembrolizumab (Keytruda), nivolumab (Opdivo), atezolizumab (Tecentique), durvalumab (Imfinzi), and avelumab (Bavencio).
Most of the patients (71%) had good PS, with an Eastern Cooperative Oncology Group (ECOG) PS of 0-1 on initiation of immunotherapy; 29% of patients had poor PS, with an ECOG PS of greater than or equal to 2.
“The primary outcome was OS stratified by ECOG PS 0-1 versus ≥2,” note the authors. Across all tumor types, OS was superior for patients in the ECOG 0-1 PS group, the investigators note. The median OS was 12.6 months, compared with only 3.1 months for patients in the ECOG greater than or equal to 2 group (P < .001).
Moreover, overall response rates for patients with a poor PS were low. Only 8%, or 6 of 75 patients with an ECOG PS of greater than or equal to 2, achieved an objective response by RECIST criteria.
This compared to an overall response rate of 23% for patients with an ECOG PS of 0-1, the investigators note (P = .005).
Interestingly, the hospice referral rate for patients with a poor PS (67%) was similar to that of patients with a PS of 1-2 (61.9%), Dr. Krishnan and colleagues observe.
Those with a poor PS were more like to die in-hospital (28.6%) than were patients with a good PS (15.1%; P = .035). The authors point out that it is well known that outcomes with chemotherapy are worse among patients who experience a decline in functional reserve, owing to increased susceptibility to toxicity and complications.
“Regardless of age, patients with ECOG PS >2 usually have poor tolerability to chemotherapy, and this correlates with worse survival outcome,” they emphasize. There is as yet no clear guidance regarding the impact of PS on ICI treatment response, although “there should be,” Dr. Luke believes.
“In a patient with declining performance status, especially ECOG PS 3-4 but potentially 2 as well, there is little likelihood that the functional and immune reserve of the patient will be adequate to mount a robust antitumor response,” he elaborated.
“It’s not impossible, but trying for it should not come at the expense of engaging about end-of-life care and maximizing the palliative opportunities that many only have a short window of time in which to pursue,” he added.
Again, Dr. Luke strongly believes that just giving an ICI without engaging in a frank conversation with the patient and their families – which happens all too often, he feels – is absolutely not the way to go when treating patients with a poor PS and little time left.
“Patients and families might be better served by having a more direct and frank conversation about what the likelihood [is] that ICI therapy will actually do,” Dr. Luke stressed.
In their editorial, Dr. Luke and colleagues write: “Overall, we as an oncology community need to improve our communication with patients regarding goals of care and end-of-life considerations as opposed to reflexive treatment initiation,” he writes.
“Our duty, first and foremost, should focus on the person sitting in front of us – taking a step back may be the best way to move forward with compassionate care,” they add.
The authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The findings have prompted an expert to argue against the use of immunotherapy for such patients, who may have little time left and very little chance of benefiting.
“It is quite clear from clinical practice that most patients with limited PS do very poorly and do not benefit from immune check point inhibitors (ICI),” Jason Luke, MD, UPMC Hillman Cancer Center and the University of Pittsburgh, said in an email.
“So, my strong opinion is that patients should not be getting an immunotherapy just because it might not cause as many side effects as chemotherapy,” he added.
“Instead of giving an immunotherapy with little chance of success, patients and families deserve to have a direct conversation about what realistic expectations [might be] and how we as the oncology community can support them to achieve whatever their personal goals are in the time that they have left,” he emphasized.
Dr. Luke was the lead author of an editorial in which he commented on the study. Both the study and the editorial were published online in JCO Oncology Practice.
Variety of cancers
The study was conducted by Mridula Krishnan, MD, Nebraska Medicine Fred and Pamela Buffett Cancer Center, Omaha, Nebraska, and colleagues.
The team reviewed 257 patients who had been treated with either a programmed cell death protein–1 inhibitor or programmed cell death–ligand-1 inhibitor for a variety of advanced cancers. The drugs included pembrolizumab (Keytruda), nivolumab (Opdivo), atezolizumab (Tecentique), durvalumab (Imfinzi), and avelumab (Bavencio).
Most of the patients (71%) had good PS, with an Eastern Cooperative Oncology Group (ECOG) PS of 0-1 on initiation of immunotherapy; 29% of patients had poor PS, with an ECOG PS of greater than or equal to 2.
“The primary outcome was OS stratified by ECOG PS 0-1 versus ≥2,” note the authors. Across all tumor types, OS was superior for patients in the ECOG 0-1 PS group, the investigators note. The median OS was 12.6 months, compared with only 3.1 months for patients in the ECOG greater than or equal to 2 group (P < .001).
Moreover, overall response rates for patients with a poor PS were low. Only 8%, or 6 of 75 patients with an ECOG PS of greater than or equal to 2, achieved an objective response by RECIST criteria.
This compared to an overall response rate of 23% for patients with an ECOG PS of 0-1, the investigators note (P = .005).
Interestingly, the hospice referral rate for patients with a poor PS (67%) was similar to that of patients with a PS of 1-2 (61.9%), Dr. Krishnan and colleagues observe.
Those with a poor PS were more like to die in-hospital (28.6%) than were patients with a good PS (15.1%; P = .035). The authors point out that it is well known that outcomes with chemotherapy are worse among patients who experience a decline in functional reserve, owing to increased susceptibility to toxicity and complications.
“Regardless of age, patients with ECOG PS >2 usually have poor tolerability to chemotherapy, and this correlates with worse survival outcome,” they emphasize. There is as yet no clear guidance regarding the impact of PS on ICI treatment response, although “there should be,” Dr. Luke believes.
“In a patient with declining performance status, especially ECOG PS 3-4 but potentially 2 as well, there is little likelihood that the functional and immune reserve of the patient will be adequate to mount a robust antitumor response,” he elaborated.
“It’s not impossible, but trying for it should not come at the expense of engaging about end-of-life care and maximizing the palliative opportunities that many only have a short window of time in which to pursue,” he added.
Again, Dr. Luke strongly believes that just giving an ICI without engaging in a frank conversation with the patient and their families – which happens all too often, he feels – is absolutely not the way to go when treating patients with a poor PS and little time left.
“Patients and families might be better served by having a more direct and frank conversation about what the likelihood [is] that ICI therapy will actually do,” Dr. Luke stressed.
In their editorial, Dr. Luke and colleagues write: “Overall, we as an oncology community need to improve our communication with patients regarding goals of care and end-of-life considerations as opposed to reflexive treatment initiation,” he writes.
“Our duty, first and foremost, should focus on the person sitting in front of us – taking a step back may be the best way to move forward with compassionate care,” they add.
The authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
How knowledgeable are ObGyns about dense breasts?
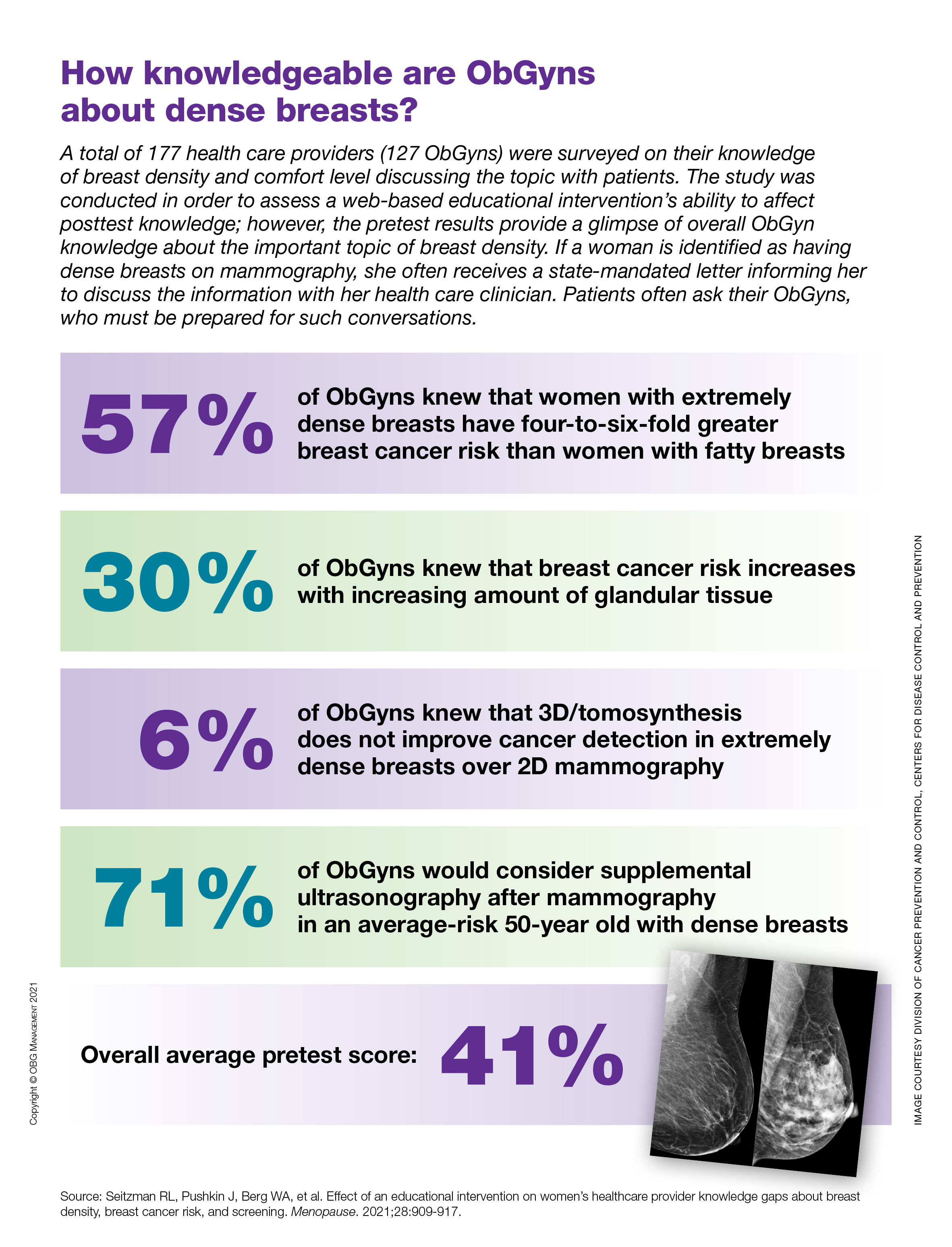


Breast density associated with increased invasive breast cancer risk after age 65
The findings, based on an analysis of Breast Cancer Surveillance Consortium data from Jan. 1, 1996, to Dec. 31, 2012, have potential implications for screening mammography decisions in older women – particularly those aged 75 years and older, for whom screening guidance is limited by a paucity of data, Dejana Braithwaite, PhD, of the University of Florida Health Cancer Center, Gainesville, and colleagues reported in JAMA Network Open.
The investigators analyzed 221,714 screening mammograms from 193,787 women aged 65 and older in the United States. About 65% of the mammograms were from women aged 65-74 years and about 35% were from women aged 75 years and older, who comprised 38% of the study population.
During a mean follow-up of 6.3 years, 5,069 invasive breast cancers were diagnosed, the authors noted.
The 5-year cumulative incidence of invasive breast cancer increased in tandem with increasing breast density among those aged 65-74 years and among those aged 75 and older: The cumulative incidence per 1,000 women aged 65-74 years was 11.3 for those with almost entirely fatty breasts, 17.2 for those with scattered fibroglandular densities, and 23.7 for those with extremely or heterogeneously dense breasts. The cumulative incidence rates for those aged 75 years and older were 13.5, 18.4, and 22.5 per 1,000 women, respectively, they found.
Extreme or heterogeneous breast density was associated with increased risk of breast cancer, compared with scattered fibroglandular breast density, in both age categories (hazard ratios, 1.39 and 1.23 for those aged 65-74 years and 75 years and older, respectively), whereas the risk of invasive breast cancer was about 30% lower among women with almost entirely fatty breasts, compared with women with scattered fibroglandular breast density (HRs, 0.66 and 0.73 for the 65-74 and 75-plus age groups, respectively).
The associations between breast density and breast cancer were statistically significant after adjustment for body mass index (BMI) and other risk factors.
However, no significant differences were seen between breast density and breast cancer risk based on BMI, noted the authors, who investigated this potential association as part of their effort to identify subpopulations of older women who might benefit from screening, “especially because the U.S. Preventive Service Task Force guidelines state that the current evidence is considered insufficient to recommend routine breast cancer screening for women aged 75 years or older,” they wrote.
Further, although breast density is important in risk assessment and could be evaluated in older women, some risk prediction models exclude women aged 75 or older in risk assessments, they noted, adding that this is concerning given “the aging of the population in the U.S. and worldwide.”
“The positive associations found in this study between breast density and breast cancer among women aged 75 years or older suggest that breast density and life expectancy should be considered together when discussing the potential benefits and harms of continued screening mammography in this population,” they concluded.
The new findings supplement those from prior studies and highlight “the intersection of ... two subjects that have garnered considerable lay public, healthy policy, and academic interest” in recent years: screening mammography in older women and the risk of breast cancer caused by breast density in older women, Catherine M. Tuite, MD, of ChristianaCare Helen F. Graham Cancer Center and Research Institute, Newark, Del., wrote in a commentary published with the study.
“Although there is a linear association between age and mammographic density, age is not a perfect surrogate for the latter, and there are meaningful numbers of older women with mammographically dense breast tissue,” she said, noting that a 75-year-old woman in the United States has a life expectancy of 12-14 additional years, and that “continuation of screening mammography in healthy women aged 75 years or older may offer a substantial opportunity to avoid morbidity and mortality from breast cancer in this age group.”
However, overdiagnosis also remains a concern, she said.
“Breast density and age are only a few of the many factors currently under investigation in the drive toward risk-based or personalized breast cancer screening,” she wrote. “We must remain cautious in the application of restrictive screening for women of any age with supposedly lower than average risk ... ultimately, the decision of when to stop screening is personal, and each woman deserves the agency to weigh her own wishes, values, and life experiences with an accurate and unbiased discussion of risks and benefits of screening mammography in making that decision.”
This study was supported by grants from the National Cancer Institute and the Breast Cancer Surveillance Consortium. Cancer and vital status data collection was supported in part by several state public health departments and cancer registries. Dr. Advani and Dr. Tuite each reported having no disclosures.
The findings, based on an analysis of Breast Cancer Surveillance Consortium data from Jan. 1, 1996, to Dec. 31, 2012, have potential implications for screening mammography decisions in older women – particularly those aged 75 years and older, for whom screening guidance is limited by a paucity of data, Dejana Braithwaite, PhD, of the University of Florida Health Cancer Center, Gainesville, and colleagues reported in JAMA Network Open.
The investigators analyzed 221,714 screening mammograms from 193,787 women aged 65 and older in the United States. About 65% of the mammograms were from women aged 65-74 years and about 35% were from women aged 75 years and older, who comprised 38% of the study population.
During a mean follow-up of 6.3 years, 5,069 invasive breast cancers were diagnosed, the authors noted.
The 5-year cumulative incidence of invasive breast cancer increased in tandem with increasing breast density among those aged 65-74 years and among those aged 75 and older: The cumulative incidence per 1,000 women aged 65-74 years was 11.3 for those with almost entirely fatty breasts, 17.2 for those with scattered fibroglandular densities, and 23.7 for those with extremely or heterogeneously dense breasts. The cumulative incidence rates for those aged 75 years and older were 13.5, 18.4, and 22.5 per 1,000 women, respectively, they found.
Extreme or heterogeneous breast density was associated with increased risk of breast cancer, compared with scattered fibroglandular breast density, in both age categories (hazard ratios, 1.39 and 1.23 for those aged 65-74 years and 75 years and older, respectively), whereas the risk of invasive breast cancer was about 30% lower among women with almost entirely fatty breasts, compared with women with scattered fibroglandular breast density (HRs, 0.66 and 0.73 for the 65-74 and 75-plus age groups, respectively).
The associations between breast density and breast cancer were statistically significant after adjustment for body mass index (BMI) and other risk factors.
However, no significant differences were seen between breast density and breast cancer risk based on BMI, noted the authors, who investigated this potential association as part of their effort to identify subpopulations of older women who might benefit from screening, “especially because the U.S. Preventive Service Task Force guidelines state that the current evidence is considered insufficient to recommend routine breast cancer screening for women aged 75 years or older,” they wrote.
Further, although breast density is important in risk assessment and could be evaluated in older women, some risk prediction models exclude women aged 75 or older in risk assessments, they noted, adding that this is concerning given “the aging of the population in the U.S. and worldwide.”
“The positive associations found in this study between breast density and breast cancer among women aged 75 years or older suggest that breast density and life expectancy should be considered together when discussing the potential benefits and harms of continued screening mammography in this population,” they concluded.
The new findings supplement those from prior studies and highlight “the intersection of ... two subjects that have garnered considerable lay public, healthy policy, and academic interest” in recent years: screening mammography in older women and the risk of breast cancer caused by breast density in older women, Catherine M. Tuite, MD, of ChristianaCare Helen F. Graham Cancer Center and Research Institute, Newark, Del., wrote in a commentary published with the study.
“Although there is a linear association between age and mammographic density, age is not a perfect surrogate for the latter, and there are meaningful numbers of older women with mammographically dense breast tissue,” she said, noting that a 75-year-old woman in the United States has a life expectancy of 12-14 additional years, and that “continuation of screening mammography in healthy women aged 75 years or older may offer a substantial opportunity to avoid morbidity and mortality from breast cancer in this age group.”
However, overdiagnosis also remains a concern, she said.
“Breast density and age are only a few of the many factors currently under investigation in the drive toward risk-based or personalized breast cancer screening,” she wrote. “We must remain cautious in the application of restrictive screening for women of any age with supposedly lower than average risk ... ultimately, the decision of when to stop screening is personal, and each woman deserves the agency to weigh her own wishes, values, and life experiences with an accurate and unbiased discussion of risks and benefits of screening mammography in making that decision.”
This study was supported by grants from the National Cancer Institute and the Breast Cancer Surveillance Consortium. Cancer and vital status data collection was supported in part by several state public health departments and cancer registries. Dr. Advani and Dr. Tuite each reported having no disclosures.
The findings, based on an analysis of Breast Cancer Surveillance Consortium data from Jan. 1, 1996, to Dec. 31, 2012, have potential implications for screening mammography decisions in older women – particularly those aged 75 years and older, for whom screening guidance is limited by a paucity of data, Dejana Braithwaite, PhD, of the University of Florida Health Cancer Center, Gainesville, and colleagues reported in JAMA Network Open.
The investigators analyzed 221,714 screening mammograms from 193,787 women aged 65 and older in the United States. About 65% of the mammograms were from women aged 65-74 years and about 35% were from women aged 75 years and older, who comprised 38% of the study population.
During a mean follow-up of 6.3 years, 5,069 invasive breast cancers were diagnosed, the authors noted.
The 5-year cumulative incidence of invasive breast cancer increased in tandem with increasing breast density among those aged 65-74 years and among those aged 75 and older: The cumulative incidence per 1,000 women aged 65-74 years was 11.3 for those with almost entirely fatty breasts, 17.2 for those with scattered fibroglandular densities, and 23.7 for those with extremely or heterogeneously dense breasts. The cumulative incidence rates for those aged 75 years and older were 13.5, 18.4, and 22.5 per 1,000 women, respectively, they found.
Extreme or heterogeneous breast density was associated with increased risk of breast cancer, compared with scattered fibroglandular breast density, in both age categories (hazard ratios, 1.39 and 1.23 for those aged 65-74 years and 75 years and older, respectively), whereas the risk of invasive breast cancer was about 30% lower among women with almost entirely fatty breasts, compared with women with scattered fibroglandular breast density (HRs, 0.66 and 0.73 for the 65-74 and 75-plus age groups, respectively).
The associations between breast density and breast cancer were statistically significant after adjustment for body mass index (BMI) and other risk factors.
However, no significant differences were seen between breast density and breast cancer risk based on BMI, noted the authors, who investigated this potential association as part of their effort to identify subpopulations of older women who might benefit from screening, “especially because the U.S. Preventive Service Task Force guidelines state that the current evidence is considered insufficient to recommend routine breast cancer screening for women aged 75 years or older,” they wrote.
Further, although breast density is important in risk assessment and could be evaluated in older women, some risk prediction models exclude women aged 75 or older in risk assessments, they noted, adding that this is concerning given “the aging of the population in the U.S. and worldwide.”
“The positive associations found in this study between breast density and breast cancer among women aged 75 years or older suggest that breast density and life expectancy should be considered together when discussing the potential benefits and harms of continued screening mammography in this population,” they concluded.
The new findings supplement those from prior studies and highlight “the intersection of ... two subjects that have garnered considerable lay public, healthy policy, and academic interest” in recent years: screening mammography in older women and the risk of breast cancer caused by breast density in older women, Catherine M. Tuite, MD, of ChristianaCare Helen F. Graham Cancer Center and Research Institute, Newark, Del., wrote in a commentary published with the study.
“Although there is a linear association between age and mammographic density, age is not a perfect surrogate for the latter, and there are meaningful numbers of older women with mammographically dense breast tissue,” she said, noting that a 75-year-old woman in the United States has a life expectancy of 12-14 additional years, and that “continuation of screening mammography in healthy women aged 75 years or older may offer a substantial opportunity to avoid morbidity and mortality from breast cancer in this age group.”
However, overdiagnosis also remains a concern, she said.
“Breast density and age are only a few of the many factors currently under investigation in the drive toward risk-based or personalized breast cancer screening,” she wrote. “We must remain cautious in the application of restrictive screening for women of any age with supposedly lower than average risk ... ultimately, the decision of when to stop screening is personal, and each woman deserves the agency to weigh her own wishes, values, and life experiences with an accurate and unbiased discussion of risks and benefits of screening mammography in making that decision.”
This study was supported by grants from the National Cancer Institute and the Breast Cancer Surveillance Consortium. Cancer and vital status data collection was supported in part by several state public health departments and cancer registries. Dr. Advani and Dr. Tuite each reported having no disclosures.
FROM JAMA NETWORK OPEN
Antipsychotics tied to increased breast cancer risk
Use of antipsychotics that increase prolactin levels is significantly associated with an increased risk for breast cancer in women with schizophrenia, new research suggests. However, at least one expert says that, at this point, clinical implications are premature.
Investigators compared data from Finnish nationwide registers on more than 30,000 women diagnosed with schizophrenia. Of those patients, 1,069 were diagnosed with breast cancer. Results showed that long-term exposure to prolactin-increasing antipsychotics was associated with a 56% increased risk of developing breast cancer in comparison with exposure of short duration. No significant association was found with cumulative exposure to prolactin-sparing antipsychotics.
“In case of planning for long-term antipsychotic [therapy], prefer non–prolactin-raising antipsychotics in females and inform patients about a potential risk to allow for informed shared decision-making,” study coauthor Christoph U. Correll, MD, professor of psychiatry and molecular medicine at Hofstra University, Hempstead, N.Y., told this news organization.
“ he said.
The study was published online Aug. 30, 2021, in The Lancet.
A ‘relevant contribution’
Breast cancer is 25% more prevalent among women with schizophrenia than among women in the general population. Antipsychotics have long been suspected as a potential culprit, but research results have been inconsistent, said Dr. Correll.
In addition, high concentrations of prolactin are associated with a higher risk of developing breast cancer, but most previous research did not distinguish between antipsychotics that increased prolactin levels those that did not.
Dr. Correll and colleagues “wanted to add to this literature by utilizing a generalizable nationwide sample with a sufficient large number of patients and sufficiently long follow-up to address the clinically very relevant question whether antipsychotic use could increase the risk of breast cancer.”
They also believed that grouping antipsychotics into prolactin-raising and non–prolactin-raising agents would be “a relevant contribution.”
The researchers drew on data from several large Finnish databases to conduct a nested case-control study of 30,785 women aged at least16 years who were diagnosed with schizophrenia between 1972 and 2014.
Of these patients, 1,069 received an initial diagnosis of invasive breast cancer (after being diagnosed with schizophrenia) between 2000 and 2017. These case patients were compared to 5,339 matched control patients. The mean age of the case patients and the control patients was 62 years. The mean time since initial diagnosis of schizophrenia was 24 years.
Antipsychotic use was divided into three periods: less than 1 year, 1-4 years, and ≥5 years. Antipsychotics were further divided into prolactin-increasing or prolactin-sparing drugs (for example, clozapine, quetiapine, or aripiprazole). Breast cancer was divided into either lobular or ductal adenocarcinoma.
In their statistical analyses, the researchers adjusted for an array of covariates, including previous diagnoses of other medical conditions, drugs that may modify the risk for breast cancer (for example, beta-blockers, calcium channel blockers, spironolactone, loop diuretics, and statins), substance misuse, suicide attempt, parity, and use of hormone replacement therapy (HRT).
‘Clinically meaningful’ risk
Ductal adenocarcinoma was more common than lobular adenocarcinoma (73% vs. 20% among case patients). A higher proportion of case patients used cardiovascular medications and HRT, compared with control patients.
A higher proportion of case patients had used prolactin-increasing antipsychotics for at least 5 years, compared with control patients (71.4% vs. 64.3%; adjusted odds ratio, 1.56; 95% CI, 1.27-1.92; P < .0001) in comparison with minimal exposure (<1 year) to prolactin-increasing antipsychotics.
On the other hand, a similar proportion of case patients and control patients used prolactin-sparing antipsychotics for at least 5 years (8.3 vs. 8.2%; aOR, 1.19; 95% CI, 0.90-1.58); the OR of 1.19 was not deemed significant.
Although exposure of ≥5 years to prolactin-increasing antipsychotics was associated with an increased risk for both types of adenocarcinoma, the risk was higher for lobular than for ductal disease (aOR, 2.36; 95% CI, 1.46-3.82 vs. aOR, 1.42; 95% CI, 1.12-1.80).
“Conservatively, if we subtract the 19% nonsignificantly increased odds with prolactin-sparing antipsychotics from the 56% significantly increased odds with prolactin-increasing antipsychotics, we obtain a 37% relative increase in odds,” the authors noted.
“Using a lifetime incidence of breast cancer in women in the general population of about 12%, with a somewhat higher lifetime incidence in patients with schizophrenia than the general population, this difference between prolactin-increasing versus prolactin-sparing antipsychotics in breast cancer risk upon exposure of 5 or more years would correspond to about a 4% (37% x 12%) increase in absolute breast cancer odds with prolactin-increasing antipsychotic treatment” – a difference the authors call “clinically meaningful.”
Correll noted that although the study was conducted in a Finnish population, the findings are generalizable to other populations.
Clinical implications premature?
Commenting on the study, Anton Pottegård, MScPharm, PhD, DMSc, professor of pharmacoepidemiology, department of public health, University of Southern Denmark, Odense, expressed concern that “this new study is fairly aggressive in its recommendation [that] we need to pay attention to hyperprolactinemia, as this seems to cause breast cancer.”
Dr. Pottegård, who is also the head of research, Hospital Pharmacy Funen, Odense University Hospital, who was not involved with the study, said he does not “think that the full body of the literature supports such a direct conclusion and/or direct inference to clinical practice.”
Although “this is an important study to further this work, I do not think we are at a place (yet) where it should lead to different action from clinicians,” Dr. Pottegård cautioned.
Also commenting on the study, Mary Seeman, MDCM, DSc, professor emeritus of neurosciences and clinical translation, department of psychiatry, University of Toronto, called the question of whether prolactin-increasing antipsychotics increase breast cancer risk “very complicated because the incidence of breast cancer ... is higher in women with schizophrenia than in other women.”
Dr. Seeman, who was not involved with the study, pointed to other reasons for the increased risk, including higher rates of obesity, substance abuse, cigarette smoking, stress, and sedentary behavior, all of which raise prolactin levels. Additionally, “protective factors such as pregnancies and breastfeeding are less frequent in women with schizophrenia than in their peers.” Women with schizophrenia also “tend not to do breast screening, see their doctors less often, follow doctors’ orders less rigorously, and obtain treatment less often.”
The take-home message “is to prescribe prolactin-sparing medication to women if at all possible – but until we know more, that is good advice, although not always possible because the illness for which the antipsychotics are prescribed may not respond to those particular medications,” Dr. Seeman said.
The study was funded by the Finnish Ministry of Social Affairs and Health through the developmental fund for Niuvanniemi Hospital. Funding was also provided to individual researchers by the Academy of Finland, the Finnish Medical Foundation, and the Emil Aaltonen foundation. Dr. Correll has been a consultant or advisor to or has received honoraria from numerous companies. He has provided expert testimony for Janssen and Otsuka; received royalties from UpToDate and is a stock option holder of LB Pharma; served on a data safety monitoring board for Lundbeck, Rovi, Supernus, and Teva; and received grant support from Janssen and Takeda. Dr. Pottegård and Dr. Seeman disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Use of antipsychotics that increase prolactin levels is significantly associated with an increased risk for breast cancer in women with schizophrenia, new research suggests. However, at least one expert says that, at this point, clinical implications are premature.
Investigators compared data from Finnish nationwide registers on more than 30,000 women diagnosed with schizophrenia. Of those patients, 1,069 were diagnosed with breast cancer. Results showed that long-term exposure to prolactin-increasing antipsychotics was associated with a 56% increased risk of developing breast cancer in comparison with exposure of short duration. No significant association was found with cumulative exposure to prolactin-sparing antipsychotics.
“In case of planning for long-term antipsychotic [therapy], prefer non–prolactin-raising antipsychotics in females and inform patients about a potential risk to allow for informed shared decision-making,” study coauthor Christoph U. Correll, MD, professor of psychiatry and molecular medicine at Hofstra University, Hempstead, N.Y., told this news organization.
“ he said.
The study was published online Aug. 30, 2021, in The Lancet.
A ‘relevant contribution’
Breast cancer is 25% more prevalent among women with schizophrenia than among women in the general population. Antipsychotics have long been suspected as a potential culprit, but research results have been inconsistent, said Dr. Correll.
In addition, high concentrations of prolactin are associated with a higher risk of developing breast cancer, but most previous research did not distinguish between antipsychotics that increased prolactin levels those that did not.
Dr. Correll and colleagues “wanted to add to this literature by utilizing a generalizable nationwide sample with a sufficient large number of patients and sufficiently long follow-up to address the clinically very relevant question whether antipsychotic use could increase the risk of breast cancer.”
They also believed that grouping antipsychotics into prolactin-raising and non–prolactin-raising agents would be “a relevant contribution.”
The researchers drew on data from several large Finnish databases to conduct a nested case-control study of 30,785 women aged at least16 years who were diagnosed with schizophrenia between 1972 and 2014.
Of these patients, 1,069 received an initial diagnosis of invasive breast cancer (after being diagnosed with schizophrenia) between 2000 and 2017. These case patients were compared to 5,339 matched control patients. The mean age of the case patients and the control patients was 62 years. The mean time since initial diagnosis of schizophrenia was 24 years.
Antipsychotic use was divided into three periods: less than 1 year, 1-4 years, and ≥5 years. Antipsychotics were further divided into prolactin-increasing or prolactin-sparing drugs (for example, clozapine, quetiapine, or aripiprazole). Breast cancer was divided into either lobular or ductal adenocarcinoma.
In their statistical analyses, the researchers adjusted for an array of covariates, including previous diagnoses of other medical conditions, drugs that may modify the risk for breast cancer (for example, beta-blockers, calcium channel blockers, spironolactone, loop diuretics, and statins), substance misuse, suicide attempt, parity, and use of hormone replacement therapy (HRT).
‘Clinically meaningful’ risk
Ductal adenocarcinoma was more common than lobular adenocarcinoma (73% vs. 20% among case patients). A higher proportion of case patients used cardiovascular medications and HRT, compared with control patients.
A higher proportion of case patients had used prolactin-increasing antipsychotics for at least 5 years, compared with control patients (71.4% vs. 64.3%; adjusted odds ratio, 1.56; 95% CI, 1.27-1.92; P < .0001) in comparison with minimal exposure (<1 year) to prolactin-increasing antipsychotics.
On the other hand, a similar proportion of case patients and control patients used prolactin-sparing antipsychotics for at least 5 years (8.3 vs. 8.2%; aOR, 1.19; 95% CI, 0.90-1.58); the OR of 1.19 was not deemed significant.
Although exposure of ≥5 years to prolactin-increasing antipsychotics was associated with an increased risk for both types of adenocarcinoma, the risk was higher for lobular than for ductal disease (aOR, 2.36; 95% CI, 1.46-3.82 vs. aOR, 1.42; 95% CI, 1.12-1.80).
“Conservatively, if we subtract the 19% nonsignificantly increased odds with prolactin-sparing antipsychotics from the 56% significantly increased odds with prolactin-increasing antipsychotics, we obtain a 37% relative increase in odds,” the authors noted.
“Using a lifetime incidence of breast cancer in women in the general population of about 12%, with a somewhat higher lifetime incidence in patients with schizophrenia than the general population, this difference between prolactin-increasing versus prolactin-sparing antipsychotics in breast cancer risk upon exposure of 5 or more years would correspond to about a 4% (37% x 12%) increase in absolute breast cancer odds with prolactin-increasing antipsychotic treatment” – a difference the authors call “clinically meaningful.”
Correll noted that although the study was conducted in a Finnish population, the findings are generalizable to other populations.
Clinical implications premature?
Commenting on the study, Anton Pottegård, MScPharm, PhD, DMSc, professor of pharmacoepidemiology, department of public health, University of Southern Denmark, Odense, expressed concern that “this new study is fairly aggressive in its recommendation [that] we need to pay attention to hyperprolactinemia, as this seems to cause breast cancer.”
Dr. Pottegård, who is also the head of research, Hospital Pharmacy Funen, Odense University Hospital, who was not involved with the study, said he does not “think that the full body of the literature supports such a direct conclusion and/or direct inference to clinical practice.”
Although “this is an important study to further this work, I do not think we are at a place (yet) where it should lead to different action from clinicians,” Dr. Pottegård cautioned.
Also commenting on the study, Mary Seeman, MDCM, DSc, professor emeritus of neurosciences and clinical translation, department of psychiatry, University of Toronto, called the question of whether prolactin-increasing antipsychotics increase breast cancer risk “very complicated because the incidence of breast cancer ... is higher in women with schizophrenia than in other women.”
Dr. Seeman, who was not involved with the study, pointed to other reasons for the increased risk, including higher rates of obesity, substance abuse, cigarette smoking, stress, and sedentary behavior, all of which raise prolactin levels. Additionally, “protective factors such as pregnancies and breastfeeding are less frequent in women with schizophrenia than in their peers.” Women with schizophrenia also “tend not to do breast screening, see their doctors less often, follow doctors’ orders less rigorously, and obtain treatment less often.”
The take-home message “is to prescribe prolactin-sparing medication to women if at all possible – but until we know more, that is good advice, although not always possible because the illness for which the antipsychotics are prescribed may not respond to those particular medications,” Dr. Seeman said.
The study was funded by the Finnish Ministry of Social Affairs and Health through the developmental fund for Niuvanniemi Hospital. Funding was also provided to individual researchers by the Academy of Finland, the Finnish Medical Foundation, and the Emil Aaltonen foundation. Dr. Correll has been a consultant or advisor to or has received honoraria from numerous companies. He has provided expert testimony for Janssen and Otsuka; received royalties from UpToDate and is a stock option holder of LB Pharma; served on a data safety monitoring board for Lundbeck, Rovi, Supernus, and Teva; and received grant support from Janssen and Takeda. Dr. Pottegård and Dr. Seeman disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Use of antipsychotics that increase prolactin levels is significantly associated with an increased risk for breast cancer in women with schizophrenia, new research suggests. However, at least one expert says that, at this point, clinical implications are premature.
Investigators compared data from Finnish nationwide registers on more than 30,000 women diagnosed with schizophrenia. Of those patients, 1,069 were diagnosed with breast cancer. Results showed that long-term exposure to prolactin-increasing antipsychotics was associated with a 56% increased risk of developing breast cancer in comparison with exposure of short duration. No significant association was found with cumulative exposure to prolactin-sparing antipsychotics.
“In case of planning for long-term antipsychotic [therapy], prefer non–prolactin-raising antipsychotics in females and inform patients about a potential risk to allow for informed shared decision-making,” study coauthor Christoph U. Correll, MD, professor of psychiatry and molecular medicine at Hofstra University, Hempstead, N.Y., told this news organization.
“ he said.
The study was published online Aug. 30, 2021, in The Lancet.
A ‘relevant contribution’
Breast cancer is 25% more prevalent among women with schizophrenia than among women in the general population. Antipsychotics have long been suspected as a potential culprit, but research results have been inconsistent, said Dr. Correll.
In addition, high concentrations of prolactin are associated with a higher risk of developing breast cancer, but most previous research did not distinguish between antipsychotics that increased prolactin levels those that did not.
Dr. Correll and colleagues “wanted to add to this literature by utilizing a generalizable nationwide sample with a sufficient large number of patients and sufficiently long follow-up to address the clinically very relevant question whether antipsychotic use could increase the risk of breast cancer.”
They also believed that grouping antipsychotics into prolactin-raising and non–prolactin-raising agents would be “a relevant contribution.”
The researchers drew on data from several large Finnish databases to conduct a nested case-control study of 30,785 women aged at least16 years who were diagnosed with schizophrenia between 1972 and 2014.
Of these patients, 1,069 received an initial diagnosis of invasive breast cancer (after being diagnosed with schizophrenia) between 2000 and 2017. These case patients were compared to 5,339 matched control patients. The mean age of the case patients and the control patients was 62 years. The mean time since initial diagnosis of schizophrenia was 24 years.
Antipsychotic use was divided into three periods: less than 1 year, 1-4 years, and ≥5 years. Antipsychotics were further divided into prolactin-increasing or prolactin-sparing drugs (for example, clozapine, quetiapine, or aripiprazole). Breast cancer was divided into either lobular or ductal adenocarcinoma.
In their statistical analyses, the researchers adjusted for an array of covariates, including previous diagnoses of other medical conditions, drugs that may modify the risk for breast cancer (for example, beta-blockers, calcium channel blockers, spironolactone, loop diuretics, and statins), substance misuse, suicide attempt, parity, and use of hormone replacement therapy (HRT).
‘Clinically meaningful’ risk
Ductal adenocarcinoma was more common than lobular adenocarcinoma (73% vs. 20% among case patients). A higher proportion of case patients used cardiovascular medications and HRT, compared with control patients.
A higher proportion of case patients had used prolactin-increasing antipsychotics for at least 5 years, compared with control patients (71.4% vs. 64.3%; adjusted odds ratio, 1.56; 95% CI, 1.27-1.92; P < .0001) in comparison with minimal exposure (<1 year) to prolactin-increasing antipsychotics.
On the other hand, a similar proportion of case patients and control patients used prolactin-sparing antipsychotics for at least 5 years (8.3 vs. 8.2%; aOR, 1.19; 95% CI, 0.90-1.58); the OR of 1.19 was not deemed significant.
Although exposure of ≥5 years to prolactin-increasing antipsychotics was associated with an increased risk for both types of adenocarcinoma, the risk was higher for lobular than for ductal disease (aOR, 2.36; 95% CI, 1.46-3.82 vs. aOR, 1.42; 95% CI, 1.12-1.80).
“Conservatively, if we subtract the 19% nonsignificantly increased odds with prolactin-sparing antipsychotics from the 56% significantly increased odds with prolactin-increasing antipsychotics, we obtain a 37% relative increase in odds,” the authors noted.
“Using a lifetime incidence of breast cancer in women in the general population of about 12%, with a somewhat higher lifetime incidence in patients with schizophrenia than the general population, this difference between prolactin-increasing versus prolactin-sparing antipsychotics in breast cancer risk upon exposure of 5 or more years would correspond to about a 4% (37% x 12%) increase in absolute breast cancer odds with prolactin-increasing antipsychotic treatment” – a difference the authors call “clinically meaningful.”
Correll noted that although the study was conducted in a Finnish population, the findings are generalizable to other populations.
Clinical implications premature?
Commenting on the study, Anton Pottegård, MScPharm, PhD, DMSc, professor of pharmacoepidemiology, department of public health, University of Southern Denmark, Odense, expressed concern that “this new study is fairly aggressive in its recommendation [that] we need to pay attention to hyperprolactinemia, as this seems to cause breast cancer.”
Dr. Pottegård, who is also the head of research, Hospital Pharmacy Funen, Odense University Hospital, who was not involved with the study, said he does not “think that the full body of the literature supports such a direct conclusion and/or direct inference to clinical practice.”
Although “this is an important study to further this work, I do not think we are at a place (yet) where it should lead to different action from clinicians,” Dr. Pottegård cautioned.
Also commenting on the study, Mary Seeman, MDCM, DSc, professor emeritus of neurosciences and clinical translation, department of psychiatry, University of Toronto, called the question of whether prolactin-increasing antipsychotics increase breast cancer risk “very complicated because the incidence of breast cancer ... is higher in women with schizophrenia than in other women.”
Dr. Seeman, who was not involved with the study, pointed to other reasons for the increased risk, including higher rates of obesity, substance abuse, cigarette smoking, stress, and sedentary behavior, all of which raise prolactin levels. Additionally, “protective factors such as pregnancies and breastfeeding are less frequent in women with schizophrenia than in their peers.” Women with schizophrenia also “tend not to do breast screening, see their doctors less often, follow doctors’ orders less rigorously, and obtain treatment less often.”
The take-home message “is to prescribe prolactin-sparing medication to women if at all possible – but until we know more, that is good advice, although not always possible because the illness for which the antipsychotics are prescribed may not respond to those particular medications,” Dr. Seeman said.
The study was funded by the Finnish Ministry of Social Affairs and Health through the developmental fund for Niuvanniemi Hospital. Funding was also provided to individual researchers by the Academy of Finland, the Finnish Medical Foundation, and the Emil Aaltonen foundation. Dr. Correll has been a consultant or advisor to or has received honoraria from numerous companies. He has provided expert testimony for Janssen and Otsuka; received royalties from UpToDate and is a stock option holder of LB Pharma; served on a data safety monitoring board for Lundbeck, Rovi, Supernus, and Teva; and received grant support from Janssen and Takeda. Dr. Pottegård and Dr. Seeman disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Data sharing to improve AI used in breast-imaging research
The curated dataset, which consists of 22,032 DBT volumes associated with 5,610 studies from 5,060 patients, was published online in JAMA Network Open. The studies were divided into types: normal studies (91.4%), actionable studies that required additional imaging but no biopsy (5.0%), benign biopsied studies (2.0%), and studies that detected cancer (1.6%).
To develop and evaluate their deep-learning model for the detection of architectural distortions and masses, the researchers used a test set of 460 studies from 418 patients with cancer. Their algorithm reached a breast-based sensitivity of two false positives per DBT volume, or 65%.
“The main focus of this publication is on the dataset, rather than on a specific hypothesis,” said principal researcher Maciej A. Mazurowski, PhD, scientific director of the Duke Center for Artificial Intelligence in Radiology in Durham, N.C.
“We have publicly shared a large dataset of digital breast tomosynthesis images, which are sometimes referred to as 3D mammograms, for more than 5,000 patients. There are two purposes for sharing data like these. One is to improve research and development of machine-learning algorithms. You can train models with these data. The other reason, maybe even more important, is to provide a benchmark to test algorithms,” he said in an interview.
The large-scale sharing of data is a key step toward transparency in science, said Dr. Mazurowski. “It is about making sure results can be easily reproduced and setting benchmarks.”
The dataset includes masses and architectural distortions that were annotated by two experienced radiologists, but does not include annotations for calcifications and/or microcalcifications.
This lack of calcifications is a limitation of the study, said Jean Seely, MD, professor of radiology at the University of Ottawa, who is president of the Canadian Society of Breast Imaging and regional lead for the Ontario Breast Screening Program.
“About 45% of invasive breast cancers are diagnosed based on calcifications,” she explained.
Still, although the sensitivity of the AI algorithm was not high (65%) – the average sensitivity of 2D mammography is 85% – the researchers should be commended for releasing such a large dataset, said Dr. Seely.
“The fact that they have made it publicly available is very, very useful,” she said, adding that the dataset can be leveraged in future breast-imaging research.
Although DBT is much better at identifying breast cancers than mammography, DBT exams take about 30% more time to read.
“There’s a lot of work being done in artificial intelligence in breast imaging to not only improve the workflow for breast radiologists, but also to help with the diagnosis and detection,” she noted. “Anything that helps improve the confidence and the accuracy of the radiologist is really what we’re aiming for right now.”
The size and the content of this dataset will contribute to breast-imaging research, said Jaron Chong, MD, of the department of medical imaging at Western University in London, Ontario, who is chair of the AI Standing Committee at the Canadian Association of Radiologists.
“The contribution could be valuable in the long term because DBT is a rare dataset in comparison to conventional 2D mammography,” said Dr. Chong. “Most existing datasets have focused on two-dimensional imaging. We might see more research papers reference this dataset in the future, iterating and improving upon this article’s algorithm performance.”
Dr. Mazurowski reports serving as an adviser to Gradient Health. Dr. Seely is an unpaid principal investigator for the Ottawa site of the Tomosynthesis Mammographic Imaging Screening Trial (TMIST). Dr. Chong has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The curated dataset, which consists of 22,032 DBT volumes associated with 5,610 studies from 5,060 patients, was published online in JAMA Network Open. The studies were divided into types: normal studies (91.4%), actionable studies that required additional imaging but no biopsy (5.0%), benign biopsied studies (2.0%), and studies that detected cancer (1.6%).
To develop and evaluate their deep-learning model for the detection of architectural distortions and masses, the researchers used a test set of 460 studies from 418 patients with cancer. Their algorithm reached a breast-based sensitivity of two false positives per DBT volume, or 65%.
“The main focus of this publication is on the dataset, rather than on a specific hypothesis,” said principal researcher Maciej A. Mazurowski, PhD, scientific director of the Duke Center for Artificial Intelligence in Radiology in Durham, N.C.
“We have publicly shared a large dataset of digital breast tomosynthesis images, which are sometimes referred to as 3D mammograms, for more than 5,000 patients. There are two purposes for sharing data like these. One is to improve research and development of machine-learning algorithms. You can train models with these data. The other reason, maybe even more important, is to provide a benchmark to test algorithms,” he said in an interview.
The large-scale sharing of data is a key step toward transparency in science, said Dr. Mazurowski. “It is about making sure results can be easily reproduced and setting benchmarks.”
The dataset includes masses and architectural distortions that were annotated by two experienced radiologists, but does not include annotations for calcifications and/or microcalcifications.
This lack of calcifications is a limitation of the study, said Jean Seely, MD, professor of radiology at the University of Ottawa, who is president of the Canadian Society of Breast Imaging and regional lead for the Ontario Breast Screening Program.
“About 45% of invasive breast cancers are diagnosed based on calcifications,” she explained.
Still, although the sensitivity of the AI algorithm was not high (65%) – the average sensitivity of 2D mammography is 85% – the researchers should be commended for releasing such a large dataset, said Dr. Seely.
“The fact that they have made it publicly available is very, very useful,” she said, adding that the dataset can be leveraged in future breast-imaging research.
Although DBT is much better at identifying breast cancers than mammography, DBT exams take about 30% more time to read.
“There’s a lot of work being done in artificial intelligence in breast imaging to not only improve the workflow for breast radiologists, but also to help with the diagnosis and detection,” she noted. “Anything that helps improve the confidence and the accuracy of the radiologist is really what we’re aiming for right now.”
The size and the content of this dataset will contribute to breast-imaging research, said Jaron Chong, MD, of the department of medical imaging at Western University in London, Ontario, who is chair of the AI Standing Committee at the Canadian Association of Radiologists.
“The contribution could be valuable in the long term because DBT is a rare dataset in comparison to conventional 2D mammography,” said Dr. Chong. “Most existing datasets have focused on two-dimensional imaging. We might see more research papers reference this dataset in the future, iterating and improving upon this article’s algorithm performance.”
Dr. Mazurowski reports serving as an adviser to Gradient Health. Dr. Seely is an unpaid principal investigator for the Ottawa site of the Tomosynthesis Mammographic Imaging Screening Trial (TMIST). Dr. Chong has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The curated dataset, which consists of 22,032 DBT volumes associated with 5,610 studies from 5,060 patients, was published online in JAMA Network Open. The studies were divided into types: normal studies (91.4%), actionable studies that required additional imaging but no biopsy (5.0%), benign biopsied studies (2.0%), and studies that detected cancer (1.6%).
To develop and evaluate their deep-learning model for the detection of architectural distortions and masses, the researchers used a test set of 460 studies from 418 patients with cancer. Their algorithm reached a breast-based sensitivity of two false positives per DBT volume, or 65%.
“The main focus of this publication is on the dataset, rather than on a specific hypothesis,” said principal researcher Maciej A. Mazurowski, PhD, scientific director of the Duke Center for Artificial Intelligence in Radiology in Durham, N.C.
“We have publicly shared a large dataset of digital breast tomosynthesis images, which are sometimes referred to as 3D mammograms, for more than 5,000 patients. There are two purposes for sharing data like these. One is to improve research and development of machine-learning algorithms. You can train models with these data. The other reason, maybe even more important, is to provide a benchmark to test algorithms,” he said in an interview.
The large-scale sharing of data is a key step toward transparency in science, said Dr. Mazurowski. “It is about making sure results can be easily reproduced and setting benchmarks.”
The dataset includes masses and architectural distortions that were annotated by two experienced radiologists, but does not include annotations for calcifications and/or microcalcifications.
This lack of calcifications is a limitation of the study, said Jean Seely, MD, professor of radiology at the University of Ottawa, who is president of the Canadian Society of Breast Imaging and regional lead for the Ontario Breast Screening Program.
“About 45% of invasive breast cancers are diagnosed based on calcifications,” she explained.
Still, although the sensitivity of the AI algorithm was not high (65%) – the average sensitivity of 2D mammography is 85% – the researchers should be commended for releasing such a large dataset, said Dr. Seely.
“The fact that they have made it publicly available is very, very useful,” she said, adding that the dataset can be leveraged in future breast-imaging research.
Although DBT is much better at identifying breast cancers than mammography, DBT exams take about 30% more time to read.
“There’s a lot of work being done in artificial intelligence in breast imaging to not only improve the workflow for breast radiologists, but also to help with the diagnosis and detection,” she noted. “Anything that helps improve the confidence and the accuracy of the radiologist is really what we’re aiming for right now.”
The size and the content of this dataset will contribute to breast-imaging research, said Jaron Chong, MD, of the department of medical imaging at Western University in London, Ontario, who is chair of the AI Standing Committee at the Canadian Association of Radiologists.
“The contribution could be valuable in the long term because DBT is a rare dataset in comparison to conventional 2D mammography,” said Dr. Chong. “Most existing datasets have focused on two-dimensional imaging. We might see more research papers reference this dataset in the future, iterating and improving upon this article’s algorithm performance.”
Dr. Mazurowski reports serving as an adviser to Gradient Health. Dr. Seely is an unpaid principal investigator for the Ottawa site of the Tomosynthesis Mammographic Imaging Screening Trial (TMIST). Dr. Chong has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA warns again about robotic mastectomy, breaks new ground
The agency also called out robotically assisted surgical (RAS) device use in the United States that lacks proper federal oversight.
“The FDA is aware of allegations that clinical studies are being conducted” with RAS devices in breast cancer “without the FDA oversight required for such significant risk studies,” the agency said.
The new advisory comes 4 weeks after a Medscape Medical News exclusive story on a set of clinical trials recently underway using RAS devices for nipple-sparing mastectomy, both prophylactically and as a breast cancer treatment.
The report found that investigators are either not collecting cancer outcomes or not doing so as a primary measure – despite a stiff warning in 2019 from the FDA that those outcomes are important.
“Congratulations to the press on doing its job well and informing government. I think this [safety communication] is a direct result of Medscape following up on this issue,” said Hooman Noorchashm, MD, PhD, a patient advocate from Philadelphia. He is a former surgeon and faculty member at the University of Pennsylvania.
In reviewing the FDA’s new warning, Dr. Noorchashm pointed out that the agency also stated unequivocally – after previously hinting – that any study of robotic mastectomy “must include monitoring of long-term clinical outcomes” such as cancer recurrence, disease-free survival, and overall survival.
That’s a change in approach – previously the FDA has typically approved/cleared RAS devices for use in cancer surgery based on 30-day complication rates (compared with standards of care) and had no requirement for cancer-related outcomes data.
“This [new] advisory reiterates the need for a clear focus on primary oncologic outcomes to, at the very least, demonstrate the noninferiority of robotic assisted surgical devices for performing mastectomy procedures,” said Dr. Noorchashm.
In a 2019 warning about robotic mastectomy, the FDA suggested that it would require oncologic measures moving forward, saying that it “anticipates” that any evaluation of new use of robotic devices in women’s cancer “would be supported” by long-term cancer outcomes. But it stopped short of publicly saying so. The new advisory changes that.
Direct comparison with traditional mastectomy needed
There are safety concerns with robotic mastectomy. Experts question whether a surgeon can easily remove a breast tumor in one piece through the small incision (a selling point of the robot). If the tissue cannot be removed in one piece, cancer fragments may be left behind.
As a result, a randomized trial with traditional open mastectomy as a comparator is needed, Dr. Noorchashm stressed. The current batch of clinical trials in the United States are all single-arm studies and as such are “totally inappropriate,” he said.
Julie Margenthaler, MD, a breast surgeon at Washington University in St Louis, also noted the importance of a randomized trial.
“I feel strongly that robotic-assisted mastectomy should only be performed in the setting of a well-designed clinical trial and that oncologic outcomes should be a primary or secondary endpoint analysis as part of that trial,” she said in an email.
Intuitive Surgical, a California-based manufacturer of robotic devices in health care and a pioneer in robot-assisted surgery, is funding one of the current clinical trials of robotic mastectomy in the United States – a single-arm, five-center trial examining use in the prophylactic setting. The two primary outcomes are conversions to open mastectomy (efficacy measure) and the incidence of adverse events during surgery to 42 days after surgery (safety measure).
This news organization previously asked the company, which manufactures the market leader da Vinci robotic surgical equipment, if it planned to conduct a randomized trial.
“Any plans for use of da Vinci Xi surgical system in nipple-sparing mastectomy will be based on these [single-arm] study results as well as other data and evidence,” said a spokesperson, who did not confirm use of a randomized trial.
The new FDA requirement for long-term oncologic outcomes also in part arises from “diminished long-term survival” that was associated with robotic surgery and other minimally invasive surgery for hysterectomy related to cervical cancer.
A version of this article first appeared on Medscape.com.
The agency also called out robotically assisted surgical (RAS) device use in the United States that lacks proper federal oversight.
“The FDA is aware of allegations that clinical studies are being conducted” with RAS devices in breast cancer “without the FDA oversight required for such significant risk studies,” the agency said.
The new advisory comes 4 weeks after a Medscape Medical News exclusive story on a set of clinical trials recently underway using RAS devices for nipple-sparing mastectomy, both prophylactically and as a breast cancer treatment.
The report found that investigators are either not collecting cancer outcomes or not doing so as a primary measure – despite a stiff warning in 2019 from the FDA that those outcomes are important.
“Congratulations to the press on doing its job well and informing government. I think this [safety communication] is a direct result of Medscape following up on this issue,” said Hooman Noorchashm, MD, PhD, a patient advocate from Philadelphia. He is a former surgeon and faculty member at the University of Pennsylvania.
In reviewing the FDA’s new warning, Dr. Noorchashm pointed out that the agency also stated unequivocally – after previously hinting – that any study of robotic mastectomy “must include monitoring of long-term clinical outcomes” such as cancer recurrence, disease-free survival, and overall survival.
That’s a change in approach – previously the FDA has typically approved/cleared RAS devices for use in cancer surgery based on 30-day complication rates (compared with standards of care) and had no requirement for cancer-related outcomes data.
“This [new] advisory reiterates the need for a clear focus on primary oncologic outcomes to, at the very least, demonstrate the noninferiority of robotic assisted surgical devices for performing mastectomy procedures,” said Dr. Noorchashm.
In a 2019 warning about robotic mastectomy, the FDA suggested that it would require oncologic measures moving forward, saying that it “anticipates” that any evaluation of new use of robotic devices in women’s cancer “would be supported” by long-term cancer outcomes. But it stopped short of publicly saying so. The new advisory changes that.
Direct comparison with traditional mastectomy needed
There are safety concerns with robotic mastectomy. Experts question whether a surgeon can easily remove a breast tumor in one piece through the small incision (a selling point of the robot). If the tissue cannot be removed in one piece, cancer fragments may be left behind.
As a result, a randomized trial with traditional open mastectomy as a comparator is needed, Dr. Noorchashm stressed. The current batch of clinical trials in the United States are all single-arm studies and as such are “totally inappropriate,” he said.
Julie Margenthaler, MD, a breast surgeon at Washington University in St Louis, also noted the importance of a randomized trial.
“I feel strongly that robotic-assisted mastectomy should only be performed in the setting of a well-designed clinical trial and that oncologic outcomes should be a primary or secondary endpoint analysis as part of that trial,” she said in an email.
Intuitive Surgical, a California-based manufacturer of robotic devices in health care and a pioneer in robot-assisted surgery, is funding one of the current clinical trials of robotic mastectomy in the United States – a single-arm, five-center trial examining use in the prophylactic setting. The two primary outcomes are conversions to open mastectomy (efficacy measure) and the incidence of adverse events during surgery to 42 days after surgery (safety measure).
This news organization previously asked the company, which manufactures the market leader da Vinci robotic surgical equipment, if it planned to conduct a randomized trial.
“Any plans for use of da Vinci Xi surgical system in nipple-sparing mastectomy will be based on these [single-arm] study results as well as other data and evidence,” said a spokesperson, who did not confirm use of a randomized trial.
The new FDA requirement for long-term oncologic outcomes also in part arises from “diminished long-term survival” that was associated with robotic surgery and other minimally invasive surgery for hysterectomy related to cervical cancer.
A version of this article first appeared on Medscape.com.
The agency also called out robotically assisted surgical (RAS) device use in the United States that lacks proper federal oversight.
“The FDA is aware of allegations that clinical studies are being conducted” with RAS devices in breast cancer “without the FDA oversight required for such significant risk studies,” the agency said.
The new advisory comes 4 weeks after a Medscape Medical News exclusive story on a set of clinical trials recently underway using RAS devices for nipple-sparing mastectomy, both prophylactically and as a breast cancer treatment.
The report found that investigators are either not collecting cancer outcomes or not doing so as a primary measure – despite a stiff warning in 2019 from the FDA that those outcomes are important.
“Congratulations to the press on doing its job well and informing government. I think this [safety communication] is a direct result of Medscape following up on this issue,” said Hooman Noorchashm, MD, PhD, a patient advocate from Philadelphia. He is a former surgeon and faculty member at the University of Pennsylvania.
In reviewing the FDA’s new warning, Dr. Noorchashm pointed out that the agency also stated unequivocally – after previously hinting – that any study of robotic mastectomy “must include monitoring of long-term clinical outcomes” such as cancer recurrence, disease-free survival, and overall survival.
That’s a change in approach – previously the FDA has typically approved/cleared RAS devices for use in cancer surgery based on 30-day complication rates (compared with standards of care) and had no requirement for cancer-related outcomes data.
“This [new] advisory reiterates the need for a clear focus on primary oncologic outcomes to, at the very least, demonstrate the noninferiority of robotic assisted surgical devices for performing mastectomy procedures,” said Dr. Noorchashm.
In a 2019 warning about robotic mastectomy, the FDA suggested that it would require oncologic measures moving forward, saying that it “anticipates” that any evaluation of new use of robotic devices in women’s cancer “would be supported” by long-term cancer outcomes. But it stopped short of publicly saying so. The new advisory changes that.
Direct comparison with traditional mastectomy needed
There are safety concerns with robotic mastectomy. Experts question whether a surgeon can easily remove a breast tumor in one piece through the small incision (a selling point of the robot). If the tissue cannot be removed in one piece, cancer fragments may be left behind.
As a result, a randomized trial with traditional open mastectomy as a comparator is needed, Dr. Noorchashm stressed. The current batch of clinical trials in the United States are all single-arm studies and as such are “totally inappropriate,” he said.
Julie Margenthaler, MD, a breast surgeon at Washington University in St Louis, also noted the importance of a randomized trial.
“I feel strongly that robotic-assisted mastectomy should only be performed in the setting of a well-designed clinical trial and that oncologic outcomes should be a primary or secondary endpoint analysis as part of that trial,” she said in an email.
Intuitive Surgical, a California-based manufacturer of robotic devices in health care and a pioneer in robot-assisted surgery, is funding one of the current clinical trials of robotic mastectomy in the United States – a single-arm, five-center trial examining use in the prophylactic setting. The two primary outcomes are conversions to open mastectomy (efficacy measure) and the incidence of adverse events during surgery to 42 days after surgery (safety measure).
This news organization previously asked the company, which manufactures the market leader da Vinci robotic surgical equipment, if it planned to conduct a randomized trial.
“Any plans for use of da Vinci Xi surgical system in nipple-sparing mastectomy will be based on these [single-arm] study results as well as other data and evidence,” said a spokesperson, who did not confirm use of a randomized trial.
The new FDA requirement for long-term oncologic outcomes also in part arises from “diminished long-term survival” that was associated with robotic surgery and other minimally invasive surgery for hysterectomy related to cervical cancer.
A version of this article first appeared on Medscape.com.
A case is building for personalized, genome-based radiation dosing
A team of researchers from the Cleveland Clinic, the Moffitt Cancer Center in Tampa, and Case Western Reserve University in Cleveland is zeroing in on a way to personalize radiation therapy for cancer patients based on genomic profile, much as genomics is used to tailor oncologic drug therapy.
It’s called “genomic-adjusted radiation dose” (GARD), a dose tailored to a person’s radiosensitivity as determined by the expression of 10 genes, known as the radiosensitivity index (RSI), combined with a linear quadratic model to yield GARD, a prediction of risk and benefit at various radiation doses for a particular patient.
A recent report in The Lancet Oncology validated GARD in 1,615 patients with seven cancer types from 11 study cohorts. If it holds up in clinical trials set to start later this year, GARD should “allow us to predict the benefit of radiation for an individual patient and adjust their treatment strategy,” wrote the authors of an editorial that accompanied the study. “The efforts need to be applauded worldwide, because radiotherapy is considerably lagging, compared with the enormous progress done in the field of personalized medicine,” Orit Kaidar-Person, MD, a radiation oncologist at Sheba Medical Center in Ramat Gan, Israel, and colleagues wrote.
GARD was associated with time to first recurrence and overall survival for patients receiving radiotherapy and predicted radiotherapy benefit, while physical dose did not. The team found a relative 2% reduction in risk of first recurrence for each unit increase of GARD (P = .0017) and a relative 3% increase in overall survival for each unit increase in GARD (P = .0007), among those who got radiotherapy. Values of GARD run from 0 to over 100, with higher scores meaning more radiation benefit.
The radiosensitivity index, which was derived from genomic studies of cancer cell lines exposed to radiation, was previously validated by the team and other groups across several tumor types.
Currently, radiation dosing is generally uniform for a given disease site and stage, based on the assumption that a given dose of radiation results in the same clinical effect across patients. In fact, the biological effect of a given dose varies widely between individual patients. “Patients we treat uniformly do not have a uniform response” which is why a more personalized approach would help, said lead investigator and Cleveland Clinic radiation oncologist Jacob Scott, MD, DPhil.
One patient with a given tumor might benefit from 2 extra fractions, while the next might need an extra 15 for the same benefit. “You need to know about [a patient’s] tumor genomics to know how hard you have to work,” he said.
Dr. Scott and colleagues are working with a genomics company to commercialize the approach. The vision for now is that physicians would ship in biopsy samples to be analyzed; RSI and GARD would be calculated, and then a decision support report would be sent back to the treatment team outlining the risks and benefits of various doses for the patient.
Dr. Scott, who holds proprietary rights on the approach, is bullish. When asked if he anticipates GARD dosing to be standard of care in 10 years, he said that “I can’t imagine another world. Everything else in cancer is personalized. Why aren’t we? It just makes sense. I know there’s a better way” to prescribe radiation, “and I’m excited for the future when I can use it.”
When asked for comment, Brian Marples, PhD, a radiation oncology professor at the University of Rochester (N.Y.), said the data so far for GARD “seem very solid. I’m very excited by the concept.”
It’s been “the holy grail” of radiation researchers to find a biologic marker that predicts what dosages patients need and what can be given safely. “This strategy is a good way of doing that. Other groups are proposing similar strategies, but I think this group is ahead. I can see [GARD] being readily applied to the clinic because patients are [already] getting their tumors genomically characterized as part of care,” Dr. Marples said.
But many questions remain. For instance, the editorial writers questioned how GARD is “affected by tumor heterogeneity, response to systemic therapy, and changes in the tumor microenvironment.” Also, the approach is based on conventional 2 Gy fractions, but other fractionation regimens are becoming more common.
For Dr. Marples, the big caveat is that most cancer patients are treated with both radiation and chemotherapy. He said he would like to see GARD validated in patients who receive both.
They seven tumor types in the study included breast cancer, head and neck cancer, non–small cell lung cancer, pancreatic cancer, endometrial cancer, melanoma, and glioma. The majority of the subjects were treated with radiation, and each had the genomic data needed to calculate GARD.
Dr. Scott, senior author and Moffitt Center radiation oncologist Javier Torres-Roca, MD, and a third author hold intellectual property rights on RSI, GARD, and prescription dose base on RSI, plus equity in Cvergenx, a company that seeks to commercialize the approach. Dr. Torres-Roca and another author are cofounders. The editorial writers and Dr. Marples did not have any relevant disclosures.
A team of researchers from the Cleveland Clinic, the Moffitt Cancer Center in Tampa, and Case Western Reserve University in Cleveland is zeroing in on a way to personalize radiation therapy for cancer patients based on genomic profile, much as genomics is used to tailor oncologic drug therapy.
It’s called “genomic-adjusted radiation dose” (GARD), a dose tailored to a person’s radiosensitivity as determined by the expression of 10 genes, known as the radiosensitivity index (RSI), combined with a linear quadratic model to yield GARD, a prediction of risk and benefit at various radiation doses for a particular patient.
A recent report in The Lancet Oncology validated GARD in 1,615 patients with seven cancer types from 11 study cohorts. If it holds up in clinical trials set to start later this year, GARD should “allow us to predict the benefit of radiation for an individual patient and adjust their treatment strategy,” wrote the authors of an editorial that accompanied the study. “The efforts need to be applauded worldwide, because radiotherapy is considerably lagging, compared with the enormous progress done in the field of personalized medicine,” Orit Kaidar-Person, MD, a radiation oncologist at Sheba Medical Center in Ramat Gan, Israel, and colleagues wrote.
GARD was associated with time to first recurrence and overall survival for patients receiving radiotherapy and predicted radiotherapy benefit, while physical dose did not. The team found a relative 2% reduction in risk of first recurrence for each unit increase of GARD (P = .0017) and a relative 3% increase in overall survival for each unit increase in GARD (P = .0007), among those who got radiotherapy. Values of GARD run from 0 to over 100, with higher scores meaning more radiation benefit.
The radiosensitivity index, which was derived from genomic studies of cancer cell lines exposed to radiation, was previously validated by the team and other groups across several tumor types.
Currently, radiation dosing is generally uniform for a given disease site and stage, based on the assumption that a given dose of radiation results in the same clinical effect across patients. In fact, the biological effect of a given dose varies widely between individual patients. “Patients we treat uniformly do not have a uniform response” which is why a more personalized approach would help, said lead investigator and Cleveland Clinic radiation oncologist Jacob Scott, MD, DPhil.
One patient with a given tumor might benefit from 2 extra fractions, while the next might need an extra 15 for the same benefit. “You need to know about [a patient’s] tumor genomics to know how hard you have to work,” he said.
Dr. Scott and colleagues are working with a genomics company to commercialize the approach. The vision for now is that physicians would ship in biopsy samples to be analyzed; RSI and GARD would be calculated, and then a decision support report would be sent back to the treatment team outlining the risks and benefits of various doses for the patient.
Dr. Scott, who holds proprietary rights on the approach, is bullish. When asked if he anticipates GARD dosing to be standard of care in 10 years, he said that “I can’t imagine another world. Everything else in cancer is personalized. Why aren’t we? It just makes sense. I know there’s a better way” to prescribe radiation, “and I’m excited for the future when I can use it.”
When asked for comment, Brian Marples, PhD, a radiation oncology professor at the University of Rochester (N.Y.), said the data so far for GARD “seem very solid. I’m very excited by the concept.”
It’s been “the holy grail” of radiation researchers to find a biologic marker that predicts what dosages patients need and what can be given safely. “This strategy is a good way of doing that. Other groups are proposing similar strategies, but I think this group is ahead. I can see [GARD] being readily applied to the clinic because patients are [already] getting their tumors genomically characterized as part of care,” Dr. Marples said.
But many questions remain. For instance, the editorial writers questioned how GARD is “affected by tumor heterogeneity, response to systemic therapy, and changes in the tumor microenvironment.” Also, the approach is based on conventional 2 Gy fractions, but other fractionation regimens are becoming more common.
For Dr. Marples, the big caveat is that most cancer patients are treated with both radiation and chemotherapy. He said he would like to see GARD validated in patients who receive both.
They seven tumor types in the study included breast cancer, head and neck cancer, non–small cell lung cancer, pancreatic cancer, endometrial cancer, melanoma, and glioma. The majority of the subjects were treated with radiation, and each had the genomic data needed to calculate GARD.
Dr. Scott, senior author and Moffitt Center radiation oncologist Javier Torres-Roca, MD, and a third author hold intellectual property rights on RSI, GARD, and prescription dose base on RSI, plus equity in Cvergenx, a company that seeks to commercialize the approach. Dr. Torres-Roca and another author are cofounders. The editorial writers and Dr. Marples did not have any relevant disclosures.
A team of researchers from the Cleveland Clinic, the Moffitt Cancer Center in Tampa, and Case Western Reserve University in Cleveland is zeroing in on a way to personalize radiation therapy for cancer patients based on genomic profile, much as genomics is used to tailor oncologic drug therapy.
It’s called “genomic-adjusted radiation dose” (GARD), a dose tailored to a person’s radiosensitivity as determined by the expression of 10 genes, known as the radiosensitivity index (RSI), combined with a linear quadratic model to yield GARD, a prediction of risk and benefit at various radiation doses for a particular patient.
A recent report in The Lancet Oncology validated GARD in 1,615 patients with seven cancer types from 11 study cohorts. If it holds up in clinical trials set to start later this year, GARD should “allow us to predict the benefit of radiation for an individual patient and adjust their treatment strategy,” wrote the authors of an editorial that accompanied the study. “The efforts need to be applauded worldwide, because radiotherapy is considerably lagging, compared with the enormous progress done in the field of personalized medicine,” Orit Kaidar-Person, MD, a radiation oncologist at Sheba Medical Center in Ramat Gan, Israel, and colleagues wrote.
GARD was associated with time to first recurrence and overall survival for patients receiving radiotherapy and predicted radiotherapy benefit, while physical dose did not. The team found a relative 2% reduction in risk of first recurrence for each unit increase of GARD (P = .0017) and a relative 3% increase in overall survival for each unit increase in GARD (P = .0007), among those who got radiotherapy. Values of GARD run from 0 to over 100, with higher scores meaning more radiation benefit.
The radiosensitivity index, which was derived from genomic studies of cancer cell lines exposed to radiation, was previously validated by the team and other groups across several tumor types.
Currently, radiation dosing is generally uniform for a given disease site and stage, based on the assumption that a given dose of radiation results in the same clinical effect across patients. In fact, the biological effect of a given dose varies widely between individual patients. “Patients we treat uniformly do not have a uniform response” which is why a more personalized approach would help, said lead investigator and Cleveland Clinic radiation oncologist Jacob Scott, MD, DPhil.
One patient with a given tumor might benefit from 2 extra fractions, while the next might need an extra 15 for the same benefit. “You need to know about [a patient’s] tumor genomics to know how hard you have to work,” he said.
Dr. Scott and colleagues are working with a genomics company to commercialize the approach. The vision for now is that physicians would ship in biopsy samples to be analyzed; RSI and GARD would be calculated, and then a decision support report would be sent back to the treatment team outlining the risks and benefits of various doses for the patient.
Dr. Scott, who holds proprietary rights on the approach, is bullish. When asked if he anticipates GARD dosing to be standard of care in 10 years, he said that “I can’t imagine another world. Everything else in cancer is personalized. Why aren’t we? It just makes sense. I know there’s a better way” to prescribe radiation, “and I’m excited for the future when I can use it.”
When asked for comment, Brian Marples, PhD, a radiation oncology professor at the University of Rochester (N.Y.), said the data so far for GARD “seem very solid. I’m very excited by the concept.”
It’s been “the holy grail” of radiation researchers to find a biologic marker that predicts what dosages patients need and what can be given safely. “This strategy is a good way of doing that. Other groups are proposing similar strategies, but I think this group is ahead. I can see [GARD] being readily applied to the clinic because patients are [already] getting their tumors genomically characterized as part of care,” Dr. Marples said.
But many questions remain. For instance, the editorial writers questioned how GARD is “affected by tumor heterogeneity, response to systemic therapy, and changes in the tumor microenvironment.” Also, the approach is based on conventional 2 Gy fractions, but other fractionation regimens are becoming more common.
For Dr. Marples, the big caveat is that most cancer patients are treated with both radiation and chemotherapy. He said he would like to see GARD validated in patients who receive both.
They seven tumor types in the study included breast cancer, head and neck cancer, non–small cell lung cancer, pancreatic cancer, endometrial cancer, melanoma, and glioma. The majority of the subjects were treated with radiation, and each had the genomic data needed to calculate GARD.
Dr. Scott, senior author and Moffitt Center radiation oncologist Javier Torres-Roca, MD, and a third author hold intellectual property rights on RSI, GARD, and prescription dose base on RSI, plus equity in Cvergenx, a company that seeks to commercialize the approach. Dr. Torres-Roca and another author are cofounders. The editorial writers and Dr. Marples did not have any relevant disclosures.
FROM LANCET ONCOLOGY
Clinical Edge Journal Scan Commentary: Breast Cancer September 2021
Metastatic triple-negative breast cancer (TNBC) is a heterogeneous, biologically complex subtype, with continuing efforts to identify therapeutic targets. The PI3K/AKT signaling pathway plays a key role in cell proliferation, survival, invasion, and metabolism. In the phase II LOTUS trial (Dent et al) including 124 patients with advanced TNBC (no prior therapy for advanced disease), addition of the oral AKT inhibitor, ipatasertib, to paclitaxel led to a numerical improvement in OS that was not statistically significant. The median OS was 25.8 vs 16.9 months for the ipatasertib-paclitaxel vs placebo-paclitaxel, respectively (HR 0.80, 95% CI 0.50-1.28). Findings from cohort A of the phase III IPATunity130 trial showed that ipatasertib-paclitaxel did not lead to a statistically significant improvement in PFS vs placebo-paclitaxel in patients with PIK3CA/AKT1/PTEN-altered advanced TNBC in the first-line metastatic setting (mPFS 7.4 vs 6.1 months, respectively; HR 1.02, p=0.9237). These findings support further investigation into predictors of response and other molecular markers that may play a role in the diversity of mTNBC.
Endocrine therapy resistance remains a significant challenge in advanced HR+/HER2- breast cancer, and initial studies with the oral histone deacetylase inhibitor, etinostat, showed promise in this space. Unfortunately, these findings were not upheld in the phase III E2112 trial (Connolly et al), which failed to show an improvement in PFS or OS with exemestane-etinostat (EE) compared to exemestane-placebo (EP) among patients who had progressed on prior non-steroidal AI. Median PFS was 3.3 vs 3.1 months (HR 0.87; p=0.30) and median OS was 23.4 vs 21.7 months (HR 0.99; p=0.94) for the EE vs EP arms, respectively. The combination of an alternative HDAC inhibitor, tucidinostat, has been approved in China in combination with exemestane based on PFS benefit (3.6 months) in the phase III ACE trial. There are notable difference between E2112 and ACE trials, including patient population and design, and importantly OS has not been reported for the latter. The relatively short mPFS and low response rate (5-6%) in E2112 argues for more efficacious therapeutics. There is also value in correlative studies to help further elucidate if there is a role for HDAC inhibitors in this space.
Although adjuvant endocrine therapy has had a significantly beneficial effect on outcomes in early-stage HR+ breast cancer, late recurrences are characteristic of the luminal subtype, and have led to trials investigating extended adjuvant endocrine therapy. The phase III SALSA trial included 3484 women with early HR+ breast cancer who had received 5 years of adjuvant endocrine therapy with randomization to anastrozole for an additional 2 vs 5 years (comparing total of 7 vs 10 years). There was no significant difference in disease-free survival (DFS) at 8 years (73.6% vs 73.9% in the 2 vs 5-year groups, respectively; HR 0.99, p=0.90). Additionally, there was a lower risk of bone fracture in the 2 vs 5-year group (4.7% vs 6.3%; HR 1.35). It is essential to balance modest benefits with toxicities of prolonged AI use, and valuable to identify high-risk patients who may benefit from extended adjuvant endocrine therapy.
References:
Chan A, Moy B, Mansi J, et al; ExteNET Study Group. Final efficacy results of neratinib in HER2-positive hormone receptor-positive early-stage breast cancer from the phase III ExteNET trial. Clin Breast Cancer. 2021;21(1):80-91.e7.
von Minckwitz G, Huang CS, Mano MS, et al; KATHERINE Investigators. Trastuzumab emtansine for residual invasive HER2-positive breast cancer. N Engl J Med. 2019;380(7):617-628.
Dent R, Kim SB, Oliveira M, et al. Double-blind placebo-controlled randomized phase III trial evaluating first-line ipatasertib combined with paclitaxel for PIK3CA/AKT1/PTEN-altered locally advanced unresectable or metastatic triple-negative breast cancer: primary results from IPATunity130 cohort A. Presented at: 2020 San Antonio Breast Cancer Symposium; December 8-11, 2020; Virtual. Oral GS3-04.
Jiang Z, Li W, Hu X, et al. Tucidinostat plus exemestane for postmenopausal patients with advanced, hormone receptor-positive breast cancer (ACE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(6):806-815.
Mamounas EP, Bandos H, Lembersky BC, et al. Use of letrozole after aromatase inhibitor-based therapy in postmenopausal breast cancer (NRG Oncology/NSABP B-42): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(1):88-99.
Metastatic triple-negative breast cancer (TNBC) is a heterogeneous, biologically complex subtype, with continuing efforts to identify therapeutic targets. The PI3K/AKT signaling pathway plays a key role in cell proliferation, survival, invasion, and metabolism. In the phase II LOTUS trial (Dent et al) including 124 patients with advanced TNBC (no prior therapy for advanced disease), addition of the oral AKT inhibitor, ipatasertib, to paclitaxel led to a numerical improvement in OS that was not statistically significant. The median OS was 25.8 vs 16.9 months for the ipatasertib-paclitaxel vs placebo-paclitaxel, respectively (HR 0.80, 95% CI 0.50-1.28). Findings from cohort A of the phase III IPATunity130 trial showed that ipatasertib-paclitaxel did not lead to a statistically significant improvement in PFS vs placebo-paclitaxel in patients with PIK3CA/AKT1/PTEN-altered advanced TNBC in the first-line metastatic setting (mPFS 7.4 vs 6.1 months, respectively; HR 1.02, p=0.9237). These findings support further investigation into predictors of response and other molecular markers that may play a role in the diversity of mTNBC.
Endocrine therapy resistance remains a significant challenge in advanced HR+/HER2- breast cancer, and initial studies with the oral histone deacetylase inhibitor, etinostat, showed promise in this space. Unfortunately, these findings were not upheld in the phase III E2112 trial (Connolly et al), which failed to show an improvement in PFS or OS with exemestane-etinostat (EE) compared to exemestane-placebo (EP) among patients who had progressed on prior non-steroidal AI. Median PFS was 3.3 vs 3.1 months (HR 0.87; p=0.30) and median OS was 23.4 vs 21.7 months (HR 0.99; p=0.94) for the EE vs EP arms, respectively. The combination of an alternative HDAC inhibitor, tucidinostat, has been approved in China in combination with exemestane based on PFS benefit (3.6 months) in the phase III ACE trial. There are notable difference between E2112 and ACE trials, including patient population and design, and importantly OS has not been reported for the latter. The relatively short mPFS and low response rate (5-6%) in E2112 argues for more efficacious therapeutics. There is also value in correlative studies to help further elucidate if there is a role for HDAC inhibitors in this space.
Although adjuvant endocrine therapy has had a significantly beneficial effect on outcomes in early-stage HR+ breast cancer, late recurrences are characteristic of the luminal subtype, and have led to trials investigating extended adjuvant endocrine therapy. The phase III SALSA trial included 3484 women with early HR+ breast cancer who had received 5 years of adjuvant endocrine therapy with randomization to anastrozole for an additional 2 vs 5 years (comparing total of 7 vs 10 years). There was no significant difference in disease-free survival (DFS) at 8 years (73.6% vs 73.9% in the 2 vs 5-year groups, respectively; HR 0.99, p=0.90). Additionally, there was a lower risk of bone fracture in the 2 vs 5-year group (4.7% vs 6.3%; HR 1.35). It is essential to balance modest benefits with toxicities of prolonged AI use, and valuable to identify high-risk patients who may benefit from extended adjuvant endocrine therapy.
References:
Chan A, Moy B, Mansi J, et al; ExteNET Study Group. Final efficacy results of neratinib in HER2-positive hormone receptor-positive early-stage breast cancer from the phase III ExteNET trial. Clin Breast Cancer. 2021;21(1):80-91.e7.
von Minckwitz G, Huang CS, Mano MS, et al; KATHERINE Investigators. Trastuzumab emtansine for residual invasive HER2-positive breast cancer. N Engl J Med. 2019;380(7):617-628.
Dent R, Kim SB, Oliveira M, et al. Double-blind placebo-controlled randomized phase III trial evaluating first-line ipatasertib combined with paclitaxel for PIK3CA/AKT1/PTEN-altered locally advanced unresectable or metastatic triple-negative breast cancer: primary results from IPATunity130 cohort A. Presented at: 2020 San Antonio Breast Cancer Symposium; December 8-11, 2020; Virtual. Oral GS3-04.
Jiang Z, Li W, Hu X, et al. Tucidinostat plus exemestane for postmenopausal patients with advanced, hormone receptor-positive breast cancer (ACE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(6):806-815.
Mamounas EP, Bandos H, Lembersky BC, et al. Use of letrozole after aromatase inhibitor-based therapy in postmenopausal breast cancer (NRG Oncology/NSABP B-42): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(1):88-99.
Metastatic triple-negative breast cancer (TNBC) is a heterogeneous, biologically complex subtype, with continuing efforts to identify therapeutic targets. The PI3K/AKT signaling pathway plays a key role in cell proliferation, survival, invasion, and metabolism. In the phase II LOTUS trial (Dent et al) including 124 patients with advanced TNBC (no prior therapy for advanced disease), addition of the oral AKT inhibitor, ipatasertib, to paclitaxel led to a numerical improvement in OS that was not statistically significant. The median OS was 25.8 vs 16.9 months for the ipatasertib-paclitaxel vs placebo-paclitaxel, respectively (HR 0.80, 95% CI 0.50-1.28). Findings from cohort A of the phase III IPATunity130 trial showed that ipatasertib-paclitaxel did not lead to a statistically significant improvement in PFS vs placebo-paclitaxel in patients with PIK3CA/AKT1/PTEN-altered advanced TNBC in the first-line metastatic setting (mPFS 7.4 vs 6.1 months, respectively; HR 1.02, p=0.9237). These findings support further investigation into predictors of response and other molecular markers that may play a role in the diversity of mTNBC.
Endocrine therapy resistance remains a significant challenge in advanced HR+/HER2- breast cancer, and initial studies with the oral histone deacetylase inhibitor, etinostat, showed promise in this space. Unfortunately, these findings were not upheld in the phase III E2112 trial (Connolly et al), which failed to show an improvement in PFS or OS with exemestane-etinostat (EE) compared to exemestane-placebo (EP) among patients who had progressed on prior non-steroidal AI. Median PFS was 3.3 vs 3.1 months (HR 0.87; p=0.30) and median OS was 23.4 vs 21.7 months (HR 0.99; p=0.94) for the EE vs EP arms, respectively. The combination of an alternative HDAC inhibitor, tucidinostat, has been approved in China in combination with exemestane based on PFS benefit (3.6 months) in the phase III ACE trial. There are notable difference between E2112 and ACE trials, including patient population and design, and importantly OS has not been reported for the latter. The relatively short mPFS and low response rate (5-6%) in E2112 argues for more efficacious therapeutics. There is also value in correlative studies to help further elucidate if there is a role for HDAC inhibitors in this space.
Although adjuvant endocrine therapy has had a significantly beneficial effect on outcomes in early-stage HR+ breast cancer, late recurrences are characteristic of the luminal subtype, and have led to trials investigating extended adjuvant endocrine therapy. The phase III SALSA trial included 3484 women with early HR+ breast cancer who had received 5 years of adjuvant endocrine therapy with randomization to anastrozole for an additional 2 vs 5 years (comparing total of 7 vs 10 years). There was no significant difference in disease-free survival (DFS) at 8 years (73.6% vs 73.9% in the 2 vs 5-year groups, respectively; HR 0.99, p=0.90). Additionally, there was a lower risk of bone fracture in the 2 vs 5-year group (4.7% vs 6.3%; HR 1.35). It is essential to balance modest benefits with toxicities of prolonged AI use, and valuable to identify high-risk patients who may benefit from extended adjuvant endocrine therapy.
References:
Chan A, Moy B, Mansi J, et al; ExteNET Study Group. Final efficacy results of neratinib in HER2-positive hormone receptor-positive early-stage breast cancer from the phase III ExteNET trial. Clin Breast Cancer. 2021;21(1):80-91.e7.
von Minckwitz G, Huang CS, Mano MS, et al; KATHERINE Investigators. Trastuzumab emtansine for residual invasive HER2-positive breast cancer. N Engl J Med. 2019;380(7):617-628.
Dent R, Kim SB, Oliveira M, et al. Double-blind placebo-controlled randomized phase III trial evaluating first-line ipatasertib combined with paclitaxel for PIK3CA/AKT1/PTEN-altered locally advanced unresectable or metastatic triple-negative breast cancer: primary results from IPATunity130 cohort A. Presented at: 2020 San Antonio Breast Cancer Symposium; December 8-11, 2020; Virtual. Oral GS3-04.
Jiang Z, Li W, Hu X, et al. Tucidinostat plus exemestane for postmenopausal patients with advanced, hormone receptor-positive breast cancer (ACE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(6):806-815.
Mamounas EP, Bandos H, Lembersky BC, et al. Use of letrozole after aromatase inhibitor-based therapy in postmenopausal breast cancer (NRG Oncology/NSABP B-42): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(1):88-99.
TNBC: Statin use is linked to survival benefit
Key clinical point: Initiation of statin treatment within 12 months of diagnosis improves survival in women with stage I-III triple-negative breast cancer (TNBC).
Major finding: Statin treatment was initiated in 2,281 patients within 12 months of diagnosis. In 1,534 patients with TNBC, statin use was associated with a significant improvement in breast cancer-specific survival (standardized hazard ratio [HR], 0.42; P = 0.022) and overall survival (HR, 0.70; P = 0.046). No significant association was observed between statin use and survival in patients without TNBC.
Study details: retrospective study of 23,192 patients with stage I-III breast cancer from the Surveillance, Epidemiology, and End Results-Medicare and Texas Cancer Registry-Medicare database between 2008 and 2015.
Disclosures: The study was supported by National Institutes of Health and Cancer Prevention Research Institute of Texas. The authors received grants, research funding, and personal fees outside this work.
Source: Nowakowska MK et al. Cancer. 2021 Aug 3. doi: 10.1002/cncr.33797.
Key clinical point: Initiation of statin treatment within 12 months of diagnosis improves survival in women with stage I-III triple-negative breast cancer (TNBC).
Major finding: Statin treatment was initiated in 2,281 patients within 12 months of diagnosis. In 1,534 patients with TNBC, statin use was associated with a significant improvement in breast cancer-specific survival (standardized hazard ratio [HR], 0.42; P = 0.022) and overall survival (HR, 0.70; P = 0.046). No significant association was observed between statin use and survival in patients without TNBC.
Study details: retrospective study of 23,192 patients with stage I-III breast cancer from the Surveillance, Epidemiology, and End Results-Medicare and Texas Cancer Registry-Medicare database between 2008 and 2015.
Disclosures: The study was supported by National Institutes of Health and Cancer Prevention Research Institute of Texas. The authors received grants, research funding, and personal fees outside this work.
Source: Nowakowska MK et al. Cancer. 2021 Aug 3. doi: 10.1002/cncr.33797.
Key clinical point: Initiation of statin treatment within 12 months of diagnosis improves survival in women with stage I-III triple-negative breast cancer (TNBC).
Major finding: Statin treatment was initiated in 2,281 patients within 12 months of diagnosis. In 1,534 patients with TNBC, statin use was associated with a significant improvement in breast cancer-specific survival (standardized hazard ratio [HR], 0.42; P = 0.022) and overall survival (HR, 0.70; P = 0.046). No significant association was observed between statin use and survival in patients without TNBC.
Study details: retrospective study of 23,192 patients with stage I-III breast cancer from the Surveillance, Epidemiology, and End Results-Medicare and Texas Cancer Registry-Medicare database between 2008 and 2015.
Disclosures: The study was supported by National Institutes of Health and Cancer Prevention Research Institute of Texas. The authors received grants, research funding, and personal fees outside this work.
Source: Nowakowska MK et al. Cancer. 2021 Aug 3. doi: 10.1002/cncr.33797.


