User login
Cerebral palsy (CP) embodies a collection of disorders involving permanent but nonprogressive motor dysfunction secondary to one of a variety of abnormal disturbances that can occur in the developing fetal or infantile brain.1 The motor impairment of CP classically leads to irregularities in muscle tone, posture, and/or movement, resulting in limitations of functional abilities that vary in severity.1,2 Patients with CP commonly experience dysphagia, gastroesophageal reflux disease, impaired airway clearance, chest wall and spine deformities, restrictive lung disease, and/or recurrent aspiration.1 Consequently, pulmonary disease is the leading cause of morbidity and mortality in patients with severe CP, characterized by recurrent bacterial infections.3,4
Frequent antibiotic use increases the risk of multidrug-resistant pathogen formation and hypersensitivity to antibiotics. Life-threatening allergic reactions in a patient population with impaired lung function significantly complicates patient management, often leading to suboptimal treatment with second-line agents.5 This case study describes a previously penicillin-tolerant patient with CP who developed a type I hypersensitivity reaction to ceftazidime/avibactam and was treated successfully with the antibiotic after rapid induction of temporary tolerance.
Case Presentation
A 34-year-old male with a complex medical history of severe spastic CP and atonic seizures was recently diagnosed with adenocarcinoma of the colon and admitted for ileostomy and sigmoidectomy. The surgery was complicated by spillage of intestinal contents into the peritoneal cavity 3 days postoperation. The patient was urgently taken to the operating room for exploratory laparotomy, culminating in remaining colectomy, complete abdominal washout, and wound vacuum placement. He continued to deteriorate clinically over the next few weeks, beginning with the development of feculent peritonitis and septic shock. Respiratory distress ensued, and the patient required a tracheostomy with mechanical ventilation. A computed tomography of the chest was consistent with multifocal pneumonia, and a respiratory culture of bronchioalveolar lavage fluid cultivated Klebsiella pneumoniae, a carbapenem-resistant Enterobacteriaceae.
The infectious disease service was consulted and recommended ceftazidime/avibactam as the only acceptable antibiotic to treat this organism. The patient had no history of drug hypersensitivities. However, he developed diffuse, generalized urticaria and predominately right-sided flushing immediately following the onset of the antibiotic infusion. The urticaria was pruritic. The patient did not have angioedema, and he did not experience any adverse respiratory, cardiac, gastrointestinal, or neurologic symptoms. The infusion was ceased immediately, and the patient was treated with a combination of diphenhydramine 50 mg IV and ranitidine 50 mg IV. Resolution of his hypersensitivity symptoms occurred within an hour of treatment, and vital signs remained stable with no resurgence of symptoms. At the time of his reaction, the patient also was taking pantoprazole, valproate, metoprolol, risperidone, and oxycodone as needed for pain. A tryptase level was not measured.
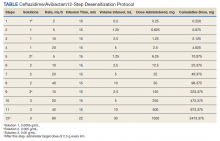
The allergy and immunology service was consulted for rapid desensitization to ceftazidime/avibactam as the culture and sensitivity test demonstrated the bacterium to be resistant to alternative antibiotics. Skin testing to ceftazidime/avibactam was deferred at the time due to the patient’s critical illness. The patient was premedicated with diphenhydramine and ranitidine 50 mg IV. Rapid IV desensitization was performed using a standard 12-step protocol developed for chemotherapeutic agents but demonstrated as safe and effective when applied to antibiotics in patients with cystic fibrosis.5 The antibiotic was administered in sequential 15-minute intervals for a total of 12 progressively doubled doses with continuous monitoring for the appearance of allergic reactions (Table). The target dose of 2.5 g was successfully achieved, and the patient tolerated a complete 14-day treatment regimen with no further adverse reactions to the medication. During the remainder of his hospital admission, the patient improved significantly without further complications.
Discussion
This is the first reported case in the literature to describe a type I hypersensitivity reaction with rapid IV induction of tolerance to ceftazidime/avibactam. We describe his reaction as type I hypersensitivity because the patient developed immediate generalized urticaria and flushing. Use of a safe desensitization protocol, demonstrated in this case report, is paramount to optimal management of infections in patient populations with severely decreased lung function, such as CP.5-7 It provides a safe and effective technique to maintain patients on first line, preferred therapy, despite their increased risk of potentially life-threatening allergic reactions.
Interestingly, this patient previously tolerated penicillins and cephalosporins without adverse reactions, suggesting the possibility of a non–IgE-mediated vs an IgE-mediated mechanism to the hypersensitivity reaction. The patient also was receiving oxycodone at the time of his reaction, and oxycodone can cause nonspecific mast cell degranulation. Additional information from skin testing to ceftazidime/avibactam could help determine whether the patient had an IgE-mediated hypersensitivity reaction. This information could help clarify the culprit agent and guide further avoidance recommendations.
Unfortunately, because the patient was critically ill, skin testing was not performed, and he underwent an urgent antibiotic desensitization with success. It was recommended that the patient follow up in the allergy and immunology clinic for further evaluation with skin testing to ceftazidime/avibactam as well as other β-lactams to determine his future risk of reaction. Unfortunately, he was lost to follow-up.
Frequent IV antibiotic use is a risk factor for the development of antibiotic allergies.8,9 This patient had received many prior courses of IV antibiotics, and this factor most likely contributed to his immediate hypersensitivity reaction to ceftazidime/avibactam. Fortunately, he tolerated a rapid induction of tolerance.
As life expectancies for patients with chronic medical conditions that involve recurrent infections increase, the associated emergence of multidrug-resistant pathogens and necessity for use of novel combination antibiotics should prompt further investigation of nonirritating doses of these drugs for skin testing in the case of drug hypersensitivities. This information would be essential for skin prick testing and determination of whether patients have a true IgE-mediated reaction to these antibiotics.
Conclusions
This is the first case report demonstrating a successful rapid induction of tolerance for the antibiotic ceftazidime/avibactam. Fortunately, the patient tolerated the desensitization procedure without further adverse reactions, and he had a resolution of his infection.
1. Rosenbaum P, Paneth N, Leviton A, et al. A report: the definition and classification of cerebral palsy April 2006. Dev Med Child Neurol. 2007;109:8-14.
2. Haak P, Lenski M, Hidecker MJ, et al. Cerebral palsy and aging. Dev Med Child Neurol. 2009;51(suppl 4):16-23. doi:10.1111/j.1469-8749.2009.03428.x
3. Duruflé-Tapin A, Colin A, Nicolas B, Lebreton C, Dauvergne F, Gallien P. Analysis of the medical causes of death in cerebral palsy. Ann Phys Rehabil Med. 2014;57(1):24-37. doi:10.1016/j.rehab.2013.11.002
4. Boel L, Pernet K, Toussaint M, et al. Respiratory morbidity in children with cerebral palsy: an overview. Dev Med Child Neurol. 2019;61(6):646-653. doi:10.1111/dmcn.14060
5. Legere HJ 3rd, Palis RI, Rodriguez Bouza T, Uluer AZ, Castells MC. A safe protocol for rapid desensitization in patients with cystic fibrosis and antibiotic hypersensitivity. J Cyst Fibros. 2009;8(6):418-424. doi:10.1016/j.jcf.2009.08.002
6. Castells M. Rapid desensitization for hypersensitivity reactions to medications. Immunol Allergy Clin North Am. 2009;29(3):585-606. doi:10.1016/j.iac.2009.04.012
7. Liu A, Fanning L, Chong H, et al. Desensitization regimens for drug allergy: state of the art in the 21st century. Clin Exp Allergy. 2011;41(12):1679-1689. doi:10.1111/j.1365-2222.2011.03825.x
8. Thong BY, Tan TC. Epidemiology and risk factors for drug allergy. Br J Clin Pharmacol. 2011;71(5):684-700. doi:10.1111/j.1365-2125.2010.03774.x
9. Adkinson NF Jr. Risk factors for drug allergy. J Allergy Clin Immunol. 1984;74(4, pt 2):567-572. doi:10.1016/0091-6749(84)90108-8
Cerebral palsy (CP) embodies a collection of disorders involving permanent but nonprogressive motor dysfunction secondary to one of a variety of abnormal disturbances that can occur in the developing fetal or infantile brain.1 The motor impairment of CP classically leads to irregularities in muscle tone, posture, and/or movement, resulting in limitations of functional abilities that vary in severity.1,2 Patients with CP commonly experience dysphagia, gastroesophageal reflux disease, impaired airway clearance, chest wall and spine deformities, restrictive lung disease, and/or recurrent aspiration.1 Consequently, pulmonary disease is the leading cause of morbidity and mortality in patients with severe CP, characterized by recurrent bacterial infections.3,4
Frequent antibiotic use increases the risk of multidrug-resistant pathogen formation and hypersensitivity to antibiotics. Life-threatening allergic reactions in a patient population with impaired lung function significantly complicates patient management, often leading to suboptimal treatment with second-line agents.5 This case study describes a previously penicillin-tolerant patient with CP who developed a type I hypersensitivity reaction to ceftazidime/avibactam and was treated successfully with the antibiotic after rapid induction of temporary tolerance.
Case Presentation
A 34-year-old male with a complex medical history of severe spastic CP and atonic seizures was recently diagnosed with adenocarcinoma of the colon and admitted for ileostomy and sigmoidectomy. The surgery was complicated by spillage of intestinal contents into the peritoneal cavity 3 days postoperation. The patient was urgently taken to the operating room for exploratory laparotomy, culminating in remaining colectomy, complete abdominal washout, and wound vacuum placement. He continued to deteriorate clinically over the next few weeks, beginning with the development of feculent peritonitis and septic shock. Respiratory distress ensued, and the patient required a tracheostomy with mechanical ventilation. A computed tomography of the chest was consistent with multifocal pneumonia, and a respiratory culture of bronchioalveolar lavage fluid cultivated Klebsiella pneumoniae, a carbapenem-resistant Enterobacteriaceae.
The infectious disease service was consulted and recommended ceftazidime/avibactam as the only acceptable antibiotic to treat this organism. The patient had no history of drug hypersensitivities. However, he developed diffuse, generalized urticaria and predominately right-sided flushing immediately following the onset of the antibiotic infusion. The urticaria was pruritic. The patient did not have angioedema, and he did not experience any adverse respiratory, cardiac, gastrointestinal, or neurologic symptoms. The infusion was ceased immediately, and the patient was treated with a combination of diphenhydramine 50 mg IV and ranitidine 50 mg IV. Resolution of his hypersensitivity symptoms occurred within an hour of treatment, and vital signs remained stable with no resurgence of symptoms. At the time of his reaction, the patient also was taking pantoprazole, valproate, metoprolol, risperidone, and oxycodone as needed for pain. A tryptase level was not measured.
The allergy and immunology service was consulted for rapid desensitization to ceftazidime/avibactam as the culture and sensitivity test demonstrated the bacterium to be resistant to alternative antibiotics. Skin testing to ceftazidime/avibactam was deferred at the time due to the patient’s critical illness. The patient was premedicated with diphenhydramine and ranitidine 50 mg IV. Rapid IV desensitization was performed using a standard 12-step protocol developed for chemotherapeutic agents but demonstrated as safe and effective when applied to antibiotics in patients with cystic fibrosis.5 The antibiotic was administered in sequential 15-minute intervals for a total of 12 progressively doubled doses with continuous monitoring for the appearance of allergic reactions (Table). The target dose of 2.5 g was successfully achieved, and the patient tolerated a complete 14-day treatment regimen with no further adverse reactions to the medication. During the remainder of his hospital admission, the patient improved significantly without further complications.
Discussion
This is the first reported case in the literature to describe a type I hypersensitivity reaction with rapid IV induction of tolerance to ceftazidime/avibactam. We describe his reaction as type I hypersensitivity because the patient developed immediate generalized urticaria and flushing. Use of a safe desensitization protocol, demonstrated in this case report, is paramount to optimal management of infections in patient populations with severely decreased lung function, such as CP.5-7 It provides a safe and effective technique to maintain patients on first line, preferred therapy, despite their increased risk of potentially life-threatening allergic reactions.
Interestingly, this patient previously tolerated penicillins and cephalosporins without adverse reactions, suggesting the possibility of a non–IgE-mediated vs an IgE-mediated mechanism to the hypersensitivity reaction. The patient also was receiving oxycodone at the time of his reaction, and oxycodone can cause nonspecific mast cell degranulation. Additional information from skin testing to ceftazidime/avibactam could help determine whether the patient had an IgE-mediated hypersensitivity reaction. This information could help clarify the culprit agent and guide further avoidance recommendations.
Unfortunately, because the patient was critically ill, skin testing was not performed, and he underwent an urgent antibiotic desensitization with success. It was recommended that the patient follow up in the allergy and immunology clinic for further evaluation with skin testing to ceftazidime/avibactam as well as other β-lactams to determine his future risk of reaction. Unfortunately, he was lost to follow-up.
Frequent IV antibiotic use is a risk factor for the development of antibiotic allergies.8,9 This patient had received many prior courses of IV antibiotics, and this factor most likely contributed to his immediate hypersensitivity reaction to ceftazidime/avibactam. Fortunately, he tolerated a rapid induction of tolerance.
As life expectancies for patients with chronic medical conditions that involve recurrent infections increase, the associated emergence of multidrug-resistant pathogens and necessity for use of novel combination antibiotics should prompt further investigation of nonirritating doses of these drugs for skin testing in the case of drug hypersensitivities. This information would be essential for skin prick testing and determination of whether patients have a true IgE-mediated reaction to these antibiotics.
Conclusions
This is the first case report demonstrating a successful rapid induction of tolerance for the antibiotic ceftazidime/avibactam. Fortunately, the patient tolerated the desensitization procedure without further adverse reactions, and he had a resolution of his infection.
Cerebral palsy (CP) embodies a collection of disorders involving permanent but nonprogressive motor dysfunction secondary to one of a variety of abnormal disturbances that can occur in the developing fetal or infantile brain.1 The motor impairment of CP classically leads to irregularities in muscle tone, posture, and/or movement, resulting in limitations of functional abilities that vary in severity.1,2 Patients with CP commonly experience dysphagia, gastroesophageal reflux disease, impaired airway clearance, chest wall and spine deformities, restrictive lung disease, and/or recurrent aspiration.1 Consequently, pulmonary disease is the leading cause of morbidity and mortality in patients with severe CP, characterized by recurrent bacterial infections.3,4
Frequent antibiotic use increases the risk of multidrug-resistant pathogen formation and hypersensitivity to antibiotics. Life-threatening allergic reactions in a patient population with impaired lung function significantly complicates patient management, often leading to suboptimal treatment with second-line agents.5 This case study describes a previously penicillin-tolerant patient with CP who developed a type I hypersensitivity reaction to ceftazidime/avibactam and was treated successfully with the antibiotic after rapid induction of temporary tolerance.
Case Presentation
A 34-year-old male with a complex medical history of severe spastic CP and atonic seizures was recently diagnosed with adenocarcinoma of the colon and admitted for ileostomy and sigmoidectomy. The surgery was complicated by spillage of intestinal contents into the peritoneal cavity 3 days postoperation. The patient was urgently taken to the operating room for exploratory laparotomy, culminating in remaining colectomy, complete abdominal washout, and wound vacuum placement. He continued to deteriorate clinically over the next few weeks, beginning with the development of feculent peritonitis and septic shock. Respiratory distress ensued, and the patient required a tracheostomy with mechanical ventilation. A computed tomography of the chest was consistent with multifocal pneumonia, and a respiratory culture of bronchioalveolar lavage fluid cultivated Klebsiella pneumoniae, a carbapenem-resistant Enterobacteriaceae.
The infectious disease service was consulted and recommended ceftazidime/avibactam as the only acceptable antibiotic to treat this organism. The patient had no history of drug hypersensitivities. However, he developed diffuse, generalized urticaria and predominately right-sided flushing immediately following the onset of the antibiotic infusion. The urticaria was pruritic. The patient did not have angioedema, and he did not experience any adverse respiratory, cardiac, gastrointestinal, or neurologic symptoms. The infusion was ceased immediately, and the patient was treated with a combination of diphenhydramine 50 mg IV and ranitidine 50 mg IV. Resolution of his hypersensitivity symptoms occurred within an hour of treatment, and vital signs remained stable with no resurgence of symptoms. At the time of his reaction, the patient also was taking pantoprazole, valproate, metoprolol, risperidone, and oxycodone as needed for pain. A tryptase level was not measured.
The allergy and immunology service was consulted for rapid desensitization to ceftazidime/avibactam as the culture and sensitivity test demonstrated the bacterium to be resistant to alternative antibiotics. Skin testing to ceftazidime/avibactam was deferred at the time due to the patient’s critical illness. The patient was premedicated with diphenhydramine and ranitidine 50 mg IV. Rapid IV desensitization was performed using a standard 12-step protocol developed for chemotherapeutic agents but demonstrated as safe and effective when applied to antibiotics in patients with cystic fibrosis.5 The antibiotic was administered in sequential 15-minute intervals for a total of 12 progressively doubled doses with continuous monitoring for the appearance of allergic reactions (Table). The target dose of 2.5 g was successfully achieved, and the patient tolerated a complete 14-day treatment regimen with no further adverse reactions to the medication. During the remainder of his hospital admission, the patient improved significantly without further complications.
Discussion
This is the first reported case in the literature to describe a type I hypersensitivity reaction with rapid IV induction of tolerance to ceftazidime/avibactam. We describe his reaction as type I hypersensitivity because the patient developed immediate generalized urticaria and flushing. Use of a safe desensitization protocol, demonstrated in this case report, is paramount to optimal management of infections in patient populations with severely decreased lung function, such as CP.5-7 It provides a safe and effective technique to maintain patients on first line, preferred therapy, despite their increased risk of potentially life-threatening allergic reactions.
Interestingly, this patient previously tolerated penicillins and cephalosporins without adverse reactions, suggesting the possibility of a non–IgE-mediated vs an IgE-mediated mechanism to the hypersensitivity reaction. The patient also was receiving oxycodone at the time of his reaction, and oxycodone can cause nonspecific mast cell degranulation. Additional information from skin testing to ceftazidime/avibactam could help determine whether the patient had an IgE-mediated hypersensitivity reaction. This information could help clarify the culprit agent and guide further avoidance recommendations.
Unfortunately, because the patient was critically ill, skin testing was not performed, and he underwent an urgent antibiotic desensitization with success. It was recommended that the patient follow up in the allergy and immunology clinic for further evaluation with skin testing to ceftazidime/avibactam as well as other β-lactams to determine his future risk of reaction. Unfortunately, he was lost to follow-up.
Frequent IV antibiotic use is a risk factor for the development of antibiotic allergies.8,9 This patient had received many prior courses of IV antibiotics, and this factor most likely contributed to his immediate hypersensitivity reaction to ceftazidime/avibactam. Fortunately, he tolerated a rapid induction of tolerance.
As life expectancies for patients with chronic medical conditions that involve recurrent infections increase, the associated emergence of multidrug-resistant pathogens and necessity for use of novel combination antibiotics should prompt further investigation of nonirritating doses of these drugs for skin testing in the case of drug hypersensitivities. This information would be essential for skin prick testing and determination of whether patients have a true IgE-mediated reaction to these antibiotics.
Conclusions
This is the first case report demonstrating a successful rapid induction of tolerance for the antibiotic ceftazidime/avibactam. Fortunately, the patient tolerated the desensitization procedure without further adverse reactions, and he had a resolution of his infection.
1. Rosenbaum P, Paneth N, Leviton A, et al. A report: the definition and classification of cerebral palsy April 2006. Dev Med Child Neurol. 2007;109:8-14.
2. Haak P, Lenski M, Hidecker MJ, et al. Cerebral palsy and aging. Dev Med Child Neurol. 2009;51(suppl 4):16-23. doi:10.1111/j.1469-8749.2009.03428.x
3. Duruflé-Tapin A, Colin A, Nicolas B, Lebreton C, Dauvergne F, Gallien P. Analysis of the medical causes of death in cerebral palsy. Ann Phys Rehabil Med. 2014;57(1):24-37. doi:10.1016/j.rehab.2013.11.002
4. Boel L, Pernet K, Toussaint M, et al. Respiratory morbidity in children with cerebral palsy: an overview. Dev Med Child Neurol. 2019;61(6):646-653. doi:10.1111/dmcn.14060
5. Legere HJ 3rd, Palis RI, Rodriguez Bouza T, Uluer AZ, Castells MC. A safe protocol for rapid desensitization in patients with cystic fibrosis and antibiotic hypersensitivity. J Cyst Fibros. 2009;8(6):418-424. doi:10.1016/j.jcf.2009.08.002
6. Castells M. Rapid desensitization for hypersensitivity reactions to medications. Immunol Allergy Clin North Am. 2009;29(3):585-606. doi:10.1016/j.iac.2009.04.012
7. Liu A, Fanning L, Chong H, et al. Desensitization regimens for drug allergy: state of the art in the 21st century. Clin Exp Allergy. 2011;41(12):1679-1689. doi:10.1111/j.1365-2222.2011.03825.x
8. Thong BY, Tan TC. Epidemiology and risk factors for drug allergy. Br J Clin Pharmacol. 2011;71(5):684-700. doi:10.1111/j.1365-2125.2010.03774.x
9. Adkinson NF Jr. Risk factors for drug allergy. J Allergy Clin Immunol. 1984;74(4, pt 2):567-572. doi:10.1016/0091-6749(84)90108-8
1. Rosenbaum P, Paneth N, Leviton A, et al. A report: the definition and classification of cerebral palsy April 2006. Dev Med Child Neurol. 2007;109:8-14.
2. Haak P, Lenski M, Hidecker MJ, et al. Cerebral palsy and aging. Dev Med Child Neurol. 2009;51(suppl 4):16-23. doi:10.1111/j.1469-8749.2009.03428.x
3. Duruflé-Tapin A, Colin A, Nicolas B, Lebreton C, Dauvergne F, Gallien P. Analysis of the medical causes of death in cerebral palsy. Ann Phys Rehabil Med. 2014;57(1):24-37. doi:10.1016/j.rehab.2013.11.002
4. Boel L, Pernet K, Toussaint M, et al. Respiratory morbidity in children with cerebral palsy: an overview. Dev Med Child Neurol. 2019;61(6):646-653. doi:10.1111/dmcn.14060
5. Legere HJ 3rd, Palis RI, Rodriguez Bouza T, Uluer AZ, Castells MC. A safe protocol for rapid desensitization in patients with cystic fibrosis and antibiotic hypersensitivity. J Cyst Fibros. 2009;8(6):418-424. doi:10.1016/j.jcf.2009.08.002
6. Castells M. Rapid desensitization for hypersensitivity reactions to medications. Immunol Allergy Clin North Am. 2009;29(3):585-606. doi:10.1016/j.iac.2009.04.012
7. Liu A, Fanning L, Chong H, et al. Desensitization regimens for drug allergy: state of the art in the 21st century. Clin Exp Allergy. 2011;41(12):1679-1689. doi:10.1111/j.1365-2222.2011.03825.x
8. Thong BY, Tan TC. Epidemiology and risk factors for drug allergy. Br J Clin Pharmacol. 2011;71(5):684-700. doi:10.1111/j.1365-2125.2010.03774.x
9. Adkinson NF Jr. Risk factors for drug allergy. J Allergy Clin Immunol. 1984;74(4, pt 2):567-572. doi:10.1016/0091-6749(84)90108-8