User login
CASE Strange creatures
Ms. L, age 78, is admitted to the inpatient unit for treatment of psychosis and behavioral changes. In the months before this admission, she had visited the emergency room several times for recurrent falls. CT scans of the head show no acute changes; brain and spinal MRI reveal evidence of chronic white matter disease and degenerative changes of the spine. Medical workup is unremarkable and includes evaluation for syncope and ambulation impairments related to degenerative disease of the hip joints.
Ms. L and her family are instructed to follow-up with her primary care physician and a neurologist for neuromuscular workup.
She next presents to her primary care physician, describing hallucinations of strangers walking around her house. Over a few weeks, hallucinations expand to include a fixed hallucination of creatures that she describes as having qualities of insects and plants, “piling up” around her. She describes tactile hallucinations of these creatures crawling on her skin, and she tracks their movements around her. She complains of vivid visual hallucinations of these creatures spinning webs across the room and she says she keeps the lights on at night. Ms. L becomes anxious and depressed, and her insomnia becomes worse.
She is referred for outpatient psychiatric evaluation and treatment.
Ms. L’s family notes lapses of short-term memory, disorganization, and difficulty with tasks such as cooking because she has trouble following steps. These deficits come and go, with periods when she is functional and others during which she experiences considerable confusion. The family is uncertain when these signs and symptoms first appeared, but are clear that these deficits are having an impact on her day-to-day life. She can conduct activities of daily living, but with increasing difficulty—and only with help from her husband for tasks that require complex order and movement.
Over several months, Ms. L’s gait stability decreases and she begins to rely on a walker to keep from falling. On the Montreal Cognitive Assessment screening for cognitive dysfunction, she scores 19 out of 30 (normal range >25). This suggests cognitive impairment greater than expected for her age, compared with normal controls, and, when coupled with her functional impairment, raises the possibility of a diagnosis of dementia with Lewy bodies (DLB).
a) donepezil
b) memantine
c) quetiapine
d) low-dose clozapine
The authors’ observations
Limited literature exists of placebo-controlled, large-scale studies on DLB treatment. Cholinesterase inhibitors have shown some symptomatic benefit, including for hallucinations.1-3 Memantine, an N-methyl-d-aspartate receptor blocker, shows mixed results.4 Many studies explore the use of neuroleptics for treating hallucinations in psychosis in Parkinson’s disease and Parkinson’s disease dementia (PDD) but, in DLB, the literature primarily consists of case reports.2 Much of DLB treatment is inferred and intermixed with studies on PDD.5,6
Low-dose clozapine has become a standard treatment for psychosis in Parkinson’s disease based on the findings of several trials.6 Despite its side-effect profile, clozapine has been shown to ameliorate hallucinations in PDD without exacerbating parkinsonian symptoms,7,8 and is the only medication with proven efficacy in PDD.2 The French Clozapine Parkinson Study Group demonstrated relief of psychotic symptoms of Parkinson’s disease with clozapine, 6.25 mg/d.9 The Clozapine Study Group found complete resolution of hallucinations in some patients within 1 day of initiating clozapine. Among patients in this study who did not see immediate benefit, most showed significant improvement of psychotic symptoms in 1 or 2 weeks.10
TREATMENT Few options
Ms. L’s psychiatrist and primary care physician start her on a series of medications. Donepezil is initiated for suspected dementia. We begin a trial of quetiapine to address the hallucinations, but the drug makes her movement symptoms worse. Risperidone also is tried but, again, the drugs make movement symptoms, particularly gait instability, tremor, and rigidity worse without alleviating the hallucinations. Neuroleptics seem to exacerbate confusion. Because of worsening depressive symptoms and our concern over possible pseudodementia, we try several selective serotonin reuptake inhibitors (SSRIs) and mirtazapine. Antidepressants have little effect on her depressive symptoms and do not improve hallucinations or insomnia.
Ms. L’s signs and symptoms become worse over the next few months, with more severe hallucinations, agitation, insomnia, and gait instability. Her agitation over the hallucinations increases and she begins pouring bleach around herself in bed and spraying her house with toxic bug spray. Ms. L’s family brings her to the hospital after they observe her scratching the hallucinatory creatures off of her skin with a razor blade and trying to pry them out of her mouth with a piece of metal.
In the hospital, medical and neurologic workups rule out organic causes for her symptoms and signs. MRI is consistent with imaging from 6 months earlier. Focal neurologic signs are absent. Blood work is within normal limits, failing to reveal any pathology that would suggest a cause for her symptoms and signs, such as syphilis, vitamin deficiency, and Lyme disease.
Ms. L’s symptoms were consistent with consensus guideline criteria for a clinical diagnosis of DLB (Table 1).11-18
She is started on low-dose quetiapine, which she tolerates poorly with worsening confusion, rigidity, tremor, and gait instability. Because other agents failed, Ms. L’s providers and family decide on a trial of clozapine.
Within 24 hours after the first dose of clozapine, 25 mg, sleep improves, the tactile component of hallucinations diminish, and she begins to spend increasing periods of time “observing the creatures” rather than fighting with them.
Over the next few days, Ms. L’s attitude towards the creatures changes. Now, as she sits observing them intently, the hallucinations evolve: rather than tormenting her and causing distress, the plant-creatures burst apart and a miniature knight on horseback charges out. The rest of the creatures then gather into a rank and file and the knight leads them to the nearest exit.
Clozapine is titrated to 50 mg/d, which she tolerates well without exacerbation of cognitive symptoms or movement disorder. The only notable adverse effect at the time of her discharge is sialorrhea.
with an antipsychotic?
a) start low and go slow
b) monitor her heart rate and blood pressure
c) readminister the Montreal Cognitive Assessment
d) all of the above
The authors’ observations
Ideally, in psychosis, antipsychotics eliminate positive symptoms such as hallucinations and delusions. In DLB, the aim is to alleviate the agitation and suffering brought on by the psychotic symptoms without exacerbating other motor and cognitive symptoms. The hallucinations are obstinate, and it is a well-known quality of this disorder that patients are exceptionally susceptible to a range of antipsychotic side effects including cognitive impairment, fatigue, neuroleptic malignant syndrome, and parkinsonism.19
Treatment in DLB requires trial and error, and medications with fewer associated risks should be administered first. Patients with DLB treated with neuroleptics have an increased risk of death compared with those who are not treated.19 Moreover, prescribing information for clozapine includes a black-box warning that the drug:
- is not approved for dementia-related psychosis and
- is associated with an increased risk of death in elderly patients with these conditions, similar to what is seen with other neuroleptics.20
Despite these well-known concerns, it remains difficult for clinicians not to try to treat the distress caused by these symptoms.
We chose clozapine for Ms. L because:
- other neuroleptics failed
- acetylcholinesterase inhibitors did not alleviate Ms. L’s psychosis and associated behavioral disturbance
- there is substantial evidence that the drug can be effective in Parkinson’s disease with psychosis.
There is controversy regarding use of clozapine in DLB. In one case series, clozapine trigger extreme neuroleptic reactions in some patients, similar to what occurs with other second-generation antipsychotics.21 Another case series provides examples of the drug’s efficacy in treating hallucinations and delusions with minimal adverse effects.22
It is important to emphasize that Ms. L’s hallucinations did not go away; rather, they changed to a more benign presentation that she could manage and, occasionally, found pleasant. Ultimately, her agitation—the primary target of treatment—improved markedly with the arrival of the knight in shining armor.
Treatment recommendations
If neuropsychiatric symptoms in DLB are the primary concern of the patient and family, we recommend the following:
- Begin treatment with a cholinesterase inhibitor. The best evidence exists for rivastigmine and donepezil. These drugs have a low risk of side effects, which are primarily gastrointestinal effects with some reports of worsening extrapyramidal symptoms.23-25
- If the patient obtains minimal benefit or develops a significant adverse effect from cholinesterase inhibitors, consider memantine. Its efficacy is under examination and results are mixed; it can be used in combination with cholinesterase inhibitors.26-28
- If psychotic symptoms are upsetting and refractory to other therapies, consider antipsychotics. Avoid first-generation antipsychotics. The American Psychiatric Association recommends aripiprazole or quetiapine initially, although there is little evidence comparing neuroleptics in DLB.29 Because of its risks, reserve clozapine for refractory cases. An exception might be made for patients sensitive to extrapyramidal effects, in whom clozapine could be considered earlier.
There are no formal neuroleptic dosing guidelines beyond a general urging towards minimalism. Mosimann and McKeith30 recommend clozapine, 12.5 mg/d; olanzapine, 2.5 mg/d; risperidone, 0.25 mg/d; or quetiapine, 12.5 mg/d. Such dosages might be effective while producing only minimal side effects.9,31
SSRIs and other antidepressants have not been shown to improve neuropsychiatric symptoms, and often are poorly tolerated.32
One study found efficacy with electroconvulsive therapy and transcranial magnetic stimulation in treatment-resistant patients.33
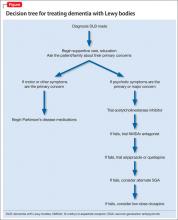
In addition to these treatments, nonpharmaceutical interventions should be employed from the earliest stages of diagnosis and treatment (Table 2). See the Figure for an algorithm for treating DLB. These include educational and behavioral interventions, social support, psychological interventions, and environmental therapies and modifications.
OUTCOME New friends
The creatures return from time to time, Ms. L reports, but are no longer upsetting because the white knight (a sort of mental deus ex machina) leads the once-terrifying things away. She describes the hallucination as a kind of zoological observation, refers to the creatures that once horrified her as “her friends,” and chuckles as she observes their natural history. This new, far more benign hallucination becomes a mainstay of her symptoms, and she is discharged to the care of her husband and family.
Soon after her discharge, her hallucinations resolved completely, but returned briefly when Ms. L resumed smoking cigarettes because smoking is known to lower clozapine serum levels.34
Bottom Line
Consider a low dosage of a neuroleptic when a patient suffers significant distress and behavioral disturbance related to psychotic symptoms in dementia with Lewy bodies and those problems are not relieved by other agents. Low-dose clozapine is an option for refractory psychotic symptoms or in patients with severe extrapyramidal sensitivity. Start low, and go slow.
Related Resources
- Bishnoi RJ, Grossberg GT, Manepalli J. Differentiating Alzheimer’s disease from dementia with Lewy bodies. Current Psychiatry. 2012;11(11):22-27.
- McKeith I, Emre M. Management of Parkinson’s disease dementia and dementia with Lewy bodies. In: Emre M, ed. Cognitive impairment and dementia in Parkinson’s disease. Oxford, United Kingdom: Oxford University Press; 2010:245-256.
Drug Brand Names
Aripiprazole • Abilify Mirtazapine • Remeron
Clozapine • Clozaril Olanzapine • Zyprexa
Donepezil • Aricept Quetiapine • Seroquel
Haloperidol • Haldol Risperidone • Risperdal
Memantine • Namenda Rivastigmine • Exelon
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Wesnes KA, McKeith IG, Ferrara R, et al. Effects of rivastigmine on cognitive function in dementia with Lewy bodies: a randomised placebo-controlled international study using the Cognitive Drug Research computerized assessment system. Dement Geriatr Cogn Disord. 2002; 13(3):183-192.
2. Weintraub D, Hurtig HI. Presentation and management of psychosis in Parkinson’s disease and dementia with Lewy bodies. Am J Psychiatry. 2007;164(10):1491-1498.
3. McKeith IG, Wesnes KA, Perry E, et al. Hallucinations predict attentional improvements with rivastigmine in dementia with Lewy bodies. Dement Geriatr Cogn Disord. 2004;18(1):94-100.
4. Emre M, Tsolaki , Bonuccelli U, et al. Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2010;9(10):969-977.
5. Aarsland D, Ballard C, Walker Z, et al. Clinical trials of dementia with Lewy bodies and Parkinson’s disease dementia. Curr Neurol Neurosci Rep. 2012;12(5):492-501.
6. Drach LM. Drug treatment of dementia with Lewy bodies and Parkinson’s disease dementia--common features and differences [in German]. Med Monatsschr Pharm. 2011; 34(2):47-52.
7. Frieling H, Hillemacher T, Ziegenbein M, et al. Treating dopamimetic psychosis in Parkinson’s disease: Structured review and meta-analysis. Eur Neuropsychopharmacol. 2007;17(3):165-171.
8. Marti MJ, Tolosa E, de la Cerda A. Dementia in Parkinson’s disease. J Neurol. 2007;254(suppl 5):41-48.
9. French Clozapine Parkinson Study Group. Clozapine in drug-induced psychosis in Parkinson’s disease. Lancet. 1999;353(9169):2041-2042.
10. Friedman JH, Factor SA. Atypical antipsychotics in the treatment of drug-induced psychosis in Parkinson’s disease. Mov Disord. 2000;15(2):201-211.
11. McKeith IG, Galasko D, Kosaka K, et al. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the consortium on DLB international workshop. Neurology. 1996;47(5):1113-1124.
12. McKeith IG, Ballard CG, Perry RH et al. Prospective validation of consensus criteria for the diagnosis of dementia with Lewy bodies. Neurology. 2000;54(5):1050-1058.
13. McKeith IG, Dickson DW, Lowe J, et al. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology. 2005;65(12):1863-1872.
14. McKeith IG. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the Consortium on DLB International Workshop. J Alzheimers Dis. 2006;9(suppl 3):417-423.
15. Geser F, Wenning GK, Poewe W, et al. How to diagnose dementia with Lewy bodies: state of the art. Mov Disord. 2005;20(suppl 12):S11-20.
16. Latoo J, Jan F. Dementia with Lewy bodies: clinical review. British Journal of Medical Practioners. 2008;1(1):10-14.
17. McKeith I. Dementia with Lewy bodies. Dialogues Clin Neurosci. 2004;6(3):333-341.
18. Litvan I, Bhatia KP, Burn DJ, et al; Movement Disorders Society Scientific Issues Committee. SIC Task Force Appraisal of clinical diagnostic criteria for parkinsonian disorders. Mov Disord. 2003;18(5):467-486.
19. McKeith I, Fairbairn A, Perry R, et al. Neuroleptic sensitivity in patients with senile dementia of Lewy body type. BMJ. 1992;305(6855):673-678.
20. Clozapine Monitoring Guidelines. 2008. http://www.clozapineregistry.com/resuming_treatment_after_interruption.pdf.ashx. Accessed October 31, 2013.
21. Burke WJ, Pfeiffer RF, McComb RD. Neuroleptic sensitivity to clozapine in dementia with Lewy bodies. J Neuropsychiatry Clin Neurosci. 1998;10(2):227-229.
22. Chacko RC, Hurley RA, Jankovic J. Clozapine use in diffuse Lewy body disease. J Neuropsychiatry Clin Neurosci. 1993;5(2):206-208.
23. McKeith I, Del Ser T, Spano P, et al. Efficacy of rivastigmine in dementia with Lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet. 2000;356(9247):2031-2036
24. Mori E, Ikeda M, Kosaka K; Donepezil-DLB Study Investiagors. Donepezil for dementia with Lewy bodies: a randomized, placebo‐controlled trial. Ann Neurol. 2012; 72(1):41-52.
25. Ukai K, Aleksic B, Ishihara R, et al. Efficacy of donepezil for the treatment of visual and multiple sensory hallucinations in dementia with Lewy bodies. Clinical Neuropsychopharmacology and Therapeutics. 2011;2:56-58.
26. Aarsland D, Ballard C, Walker Z, et al. Memantine in patients with Parkinson’s disease dementia or dementia with Lewy bodies: a double-blind, placebo-controlled, multicentre trial. Lancet Neurol. 2009;8(7):613-618.
27. Boeve BF, Silber MH, Ferman TJ. Melatonin for treatment of REM sleep behavior disorder in neurologic disorders: results in 14 patients. Sleep Med. 2003;4(4):281-284.
28. Mathys ML, McCarrell J, Sleeper RB, et al. Visual hallucinations treated with the reinitiation of memantine in a patient with Lewy body dementia. Ann Pharmacother. 2013;47(2):e10.
29. American Psychiatric Association. Practice guideline for the treatment of patients with Alzheimer’s disease and other dementias. 2nd ed. http://psychiatryonline.org/pdfaccess.ashx?ResourceID=243205&PDFSource=6. Accessed November 1, 2013.
30. Mosimann U, McKeith IG. Dementia with lewy bodies—diagnosis and treatment. Swiss Med Wkly. 2003;133:131-142.
31. Baskys A, Davis P Atypical antipsychotic quetiapine in the treatment of the psychosis associated with Lewy body dementia. Neurobiol Aging. 2002;23:S63.
32. Culo S, Mulsant BH, Rosen J, et al. Treating neuropsychiatric symptoms in dementia with Lewy bodies: a randomized controlled-trial. Alzheimer Dis Assoc Disord. 2010;24(4):306-364.
33. Takahashi S, Mizukami K, Yasuno F, et al. Depression associated with dementia with Lewy bodies (DLB) and the effect of somatotherapy. Psychogeriatrics. 2009;9(2):56-61.
34. van der Weide J, Steijns LS, van Weelden MJ. The effect of smoking and cytochrome P450 CYP1A2 genetic polymorphism on clozapine clearance and dose requirement. Pharmacogenetics. 2003;13(3):169-172.
CASE Strange creatures
Ms. L, age 78, is admitted to the inpatient unit for treatment of psychosis and behavioral changes. In the months before this admission, she had visited the emergency room several times for recurrent falls. CT scans of the head show no acute changes; brain and spinal MRI reveal evidence of chronic white matter disease and degenerative changes of the spine. Medical workup is unremarkable and includes evaluation for syncope and ambulation impairments related to degenerative disease of the hip joints.
Ms. L and her family are instructed to follow-up with her primary care physician and a neurologist for neuromuscular workup.
She next presents to her primary care physician, describing hallucinations of strangers walking around her house. Over a few weeks, hallucinations expand to include a fixed hallucination of creatures that she describes as having qualities of insects and plants, “piling up” around her. She describes tactile hallucinations of these creatures crawling on her skin, and she tracks their movements around her. She complains of vivid visual hallucinations of these creatures spinning webs across the room and she says she keeps the lights on at night. Ms. L becomes anxious and depressed, and her insomnia becomes worse.
She is referred for outpatient psychiatric evaluation and treatment.
Ms. L’s family notes lapses of short-term memory, disorganization, and difficulty with tasks such as cooking because she has trouble following steps. These deficits come and go, with periods when she is functional and others during which she experiences considerable confusion. The family is uncertain when these signs and symptoms first appeared, but are clear that these deficits are having an impact on her day-to-day life. She can conduct activities of daily living, but with increasing difficulty—and only with help from her husband for tasks that require complex order and movement.
Over several months, Ms. L’s gait stability decreases and she begins to rely on a walker to keep from falling. On the Montreal Cognitive Assessment screening for cognitive dysfunction, she scores 19 out of 30 (normal range >25). This suggests cognitive impairment greater than expected for her age, compared with normal controls, and, when coupled with her functional impairment, raises the possibility of a diagnosis of dementia with Lewy bodies (DLB).
a) donepezil
b) memantine
c) quetiapine
d) low-dose clozapine
The authors’ observations
Limited literature exists of placebo-controlled, large-scale studies on DLB treatment. Cholinesterase inhibitors have shown some symptomatic benefit, including for hallucinations.1-3 Memantine, an N-methyl-d-aspartate receptor blocker, shows mixed results.4 Many studies explore the use of neuroleptics for treating hallucinations in psychosis in Parkinson’s disease and Parkinson’s disease dementia (PDD) but, in DLB, the literature primarily consists of case reports.2 Much of DLB treatment is inferred and intermixed with studies on PDD.5,6
Low-dose clozapine has become a standard treatment for psychosis in Parkinson’s disease based on the findings of several trials.6 Despite its side-effect profile, clozapine has been shown to ameliorate hallucinations in PDD without exacerbating parkinsonian symptoms,7,8 and is the only medication with proven efficacy in PDD.2 The French Clozapine Parkinson Study Group demonstrated relief of psychotic symptoms of Parkinson’s disease with clozapine, 6.25 mg/d.9 The Clozapine Study Group found complete resolution of hallucinations in some patients within 1 day of initiating clozapine. Among patients in this study who did not see immediate benefit, most showed significant improvement of psychotic symptoms in 1 or 2 weeks.10
TREATMENT Few options
Ms. L’s psychiatrist and primary care physician start her on a series of medications. Donepezil is initiated for suspected dementia. We begin a trial of quetiapine to address the hallucinations, but the drug makes her movement symptoms worse. Risperidone also is tried but, again, the drugs make movement symptoms, particularly gait instability, tremor, and rigidity worse without alleviating the hallucinations. Neuroleptics seem to exacerbate confusion. Because of worsening depressive symptoms and our concern over possible pseudodementia, we try several selective serotonin reuptake inhibitors (SSRIs) and mirtazapine. Antidepressants have little effect on her depressive symptoms and do not improve hallucinations or insomnia.
Ms. L’s signs and symptoms become worse over the next few months, with more severe hallucinations, agitation, insomnia, and gait instability. Her agitation over the hallucinations increases and she begins pouring bleach around herself in bed and spraying her house with toxic bug spray. Ms. L’s family brings her to the hospital after they observe her scratching the hallucinatory creatures off of her skin with a razor blade and trying to pry them out of her mouth with a piece of metal.
In the hospital, medical and neurologic workups rule out organic causes for her symptoms and signs. MRI is consistent with imaging from 6 months earlier. Focal neurologic signs are absent. Blood work is within normal limits, failing to reveal any pathology that would suggest a cause for her symptoms and signs, such as syphilis, vitamin deficiency, and Lyme disease.
Ms. L’s symptoms were consistent with consensus guideline criteria for a clinical diagnosis of DLB (Table 1).11-18
She is started on low-dose quetiapine, which she tolerates poorly with worsening confusion, rigidity, tremor, and gait instability. Because other agents failed, Ms. L’s providers and family decide on a trial of clozapine.
Within 24 hours after the first dose of clozapine, 25 mg, sleep improves, the tactile component of hallucinations diminish, and she begins to spend increasing periods of time “observing the creatures” rather than fighting with them.
Over the next few days, Ms. L’s attitude towards the creatures changes. Now, as she sits observing them intently, the hallucinations evolve: rather than tormenting her and causing distress, the plant-creatures burst apart and a miniature knight on horseback charges out. The rest of the creatures then gather into a rank and file and the knight leads them to the nearest exit.
Clozapine is titrated to 50 mg/d, which she tolerates well without exacerbation of cognitive symptoms or movement disorder. The only notable adverse effect at the time of her discharge is sialorrhea.
with an antipsychotic?
a) start low and go slow
b) monitor her heart rate and blood pressure
c) readminister the Montreal Cognitive Assessment
d) all of the above
The authors’ observations
Ideally, in psychosis, antipsychotics eliminate positive symptoms such as hallucinations and delusions. In DLB, the aim is to alleviate the agitation and suffering brought on by the psychotic symptoms without exacerbating other motor and cognitive symptoms. The hallucinations are obstinate, and it is a well-known quality of this disorder that patients are exceptionally susceptible to a range of antipsychotic side effects including cognitive impairment, fatigue, neuroleptic malignant syndrome, and parkinsonism.19
Treatment in DLB requires trial and error, and medications with fewer associated risks should be administered first. Patients with DLB treated with neuroleptics have an increased risk of death compared with those who are not treated.19 Moreover, prescribing information for clozapine includes a black-box warning that the drug:
- is not approved for dementia-related psychosis and
- is associated with an increased risk of death in elderly patients with these conditions, similar to what is seen with other neuroleptics.20
Despite these well-known concerns, it remains difficult for clinicians not to try to treat the distress caused by these symptoms.
We chose clozapine for Ms. L because:
- other neuroleptics failed
- acetylcholinesterase inhibitors did not alleviate Ms. L’s psychosis and associated behavioral disturbance
- there is substantial evidence that the drug can be effective in Parkinson’s disease with psychosis.
There is controversy regarding use of clozapine in DLB. In one case series, clozapine trigger extreme neuroleptic reactions in some patients, similar to what occurs with other second-generation antipsychotics.21 Another case series provides examples of the drug’s efficacy in treating hallucinations and delusions with minimal adverse effects.22
It is important to emphasize that Ms. L’s hallucinations did not go away; rather, they changed to a more benign presentation that she could manage and, occasionally, found pleasant. Ultimately, her agitation—the primary target of treatment—improved markedly with the arrival of the knight in shining armor.
Treatment recommendations
If neuropsychiatric symptoms in DLB are the primary concern of the patient and family, we recommend the following:
- Begin treatment with a cholinesterase inhibitor. The best evidence exists for rivastigmine and donepezil. These drugs have a low risk of side effects, which are primarily gastrointestinal effects with some reports of worsening extrapyramidal symptoms.23-25
- If the patient obtains minimal benefit or develops a significant adverse effect from cholinesterase inhibitors, consider memantine. Its efficacy is under examination and results are mixed; it can be used in combination with cholinesterase inhibitors.26-28
- If psychotic symptoms are upsetting and refractory to other therapies, consider antipsychotics. Avoid first-generation antipsychotics. The American Psychiatric Association recommends aripiprazole or quetiapine initially, although there is little evidence comparing neuroleptics in DLB.29 Because of its risks, reserve clozapine for refractory cases. An exception might be made for patients sensitive to extrapyramidal effects, in whom clozapine could be considered earlier.
There are no formal neuroleptic dosing guidelines beyond a general urging towards minimalism. Mosimann and McKeith30 recommend clozapine, 12.5 mg/d; olanzapine, 2.5 mg/d; risperidone, 0.25 mg/d; or quetiapine, 12.5 mg/d. Such dosages might be effective while producing only minimal side effects.9,31
SSRIs and other antidepressants have not been shown to improve neuropsychiatric symptoms, and often are poorly tolerated.32
One study found efficacy with electroconvulsive therapy and transcranial magnetic stimulation in treatment-resistant patients.33
In addition to these treatments, nonpharmaceutical interventions should be employed from the earliest stages of diagnosis and treatment (Table 2). See the Figure for an algorithm for treating DLB. These include educational and behavioral interventions, social support, psychological interventions, and environmental therapies and modifications.
OUTCOME New friends
The creatures return from time to time, Ms. L reports, but are no longer upsetting because the white knight (a sort of mental deus ex machina) leads the once-terrifying things away. She describes the hallucination as a kind of zoological observation, refers to the creatures that once horrified her as “her friends,” and chuckles as she observes their natural history. This new, far more benign hallucination becomes a mainstay of her symptoms, and she is discharged to the care of her husband and family.
Soon after her discharge, her hallucinations resolved completely, but returned briefly when Ms. L resumed smoking cigarettes because smoking is known to lower clozapine serum levels.34
Bottom Line
Consider a low dosage of a neuroleptic when a patient suffers significant distress and behavioral disturbance related to psychotic symptoms in dementia with Lewy bodies and those problems are not relieved by other agents. Low-dose clozapine is an option for refractory psychotic symptoms or in patients with severe extrapyramidal sensitivity. Start low, and go slow.
Related Resources
- Bishnoi RJ, Grossberg GT, Manepalli J. Differentiating Alzheimer’s disease from dementia with Lewy bodies. Current Psychiatry. 2012;11(11):22-27.
- McKeith I, Emre M. Management of Parkinson’s disease dementia and dementia with Lewy bodies. In: Emre M, ed. Cognitive impairment and dementia in Parkinson’s disease. Oxford, United Kingdom: Oxford University Press; 2010:245-256.
Drug Brand Names
Aripiprazole • Abilify Mirtazapine • Remeron
Clozapine • Clozaril Olanzapine • Zyprexa
Donepezil • Aricept Quetiapine • Seroquel
Haloperidol • Haldol Risperidone • Risperdal
Memantine • Namenda Rivastigmine • Exelon
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
CASE Strange creatures
Ms. L, age 78, is admitted to the inpatient unit for treatment of psychosis and behavioral changes. In the months before this admission, she had visited the emergency room several times for recurrent falls. CT scans of the head show no acute changes; brain and spinal MRI reveal evidence of chronic white matter disease and degenerative changes of the spine. Medical workup is unremarkable and includes evaluation for syncope and ambulation impairments related to degenerative disease of the hip joints.
Ms. L and her family are instructed to follow-up with her primary care physician and a neurologist for neuromuscular workup.
She next presents to her primary care physician, describing hallucinations of strangers walking around her house. Over a few weeks, hallucinations expand to include a fixed hallucination of creatures that she describes as having qualities of insects and plants, “piling up” around her. She describes tactile hallucinations of these creatures crawling on her skin, and she tracks their movements around her. She complains of vivid visual hallucinations of these creatures spinning webs across the room and she says she keeps the lights on at night. Ms. L becomes anxious and depressed, and her insomnia becomes worse.
She is referred for outpatient psychiatric evaluation and treatment.
Ms. L’s family notes lapses of short-term memory, disorganization, and difficulty with tasks such as cooking because she has trouble following steps. These deficits come and go, with periods when she is functional and others during which she experiences considerable confusion. The family is uncertain when these signs and symptoms first appeared, but are clear that these deficits are having an impact on her day-to-day life. She can conduct activities of daily living, but with increasing difficulty—and only with help from her husband for tasks that require complex order and movement.
Over several months, Ms. L’s gait stability decreases and she begins to rely on a walker to keep from falling. On the Montreal Cognitive Assessment screening for cognitive dysfunction, she scores 19 out of 30 (normal range >25). This suggests cognitive impairment greater than expected for her age, compared with normal controls, and, when coupled with her functional impairment, raises the possibility of a diagnosis of dementia with Lewy bodies (DLB).
a) donepezil
b) memantine
c) quetiapine
d) low-dose clozapine
The authors’ observations
Limited literature exists of placebo-controlled, large-scale studies on DLB treatment. Cholinesterase inhibitors have shown some symptomatic benefit, including for hallucinations.1-3 Memantine, an N-methyl-d-aspartate receptor blocker, shows mixed results.4 Many studies explore the use of neuroleptics for treating hallucinations in psychosis in Parkinson’s disease and Parkinson’s disease dementia (PDD) but, in DLB, the literature primarily consists of case reports.2 Much of DLB treatment is inferred and intermixed with studies on PDD.5,6
Low-dose clozapine has become a standard treatment for psychosis in Parkinson’s disease based on the findings of several trials.6 Despite its side-effect profile, clozapine has been shown to ameliorate hallucinations in PDD without exacerbating parkinsonian symptoms,7,8 and is the only medication with proven efficacy in PDD.2 The French Clozapine Parkinson Study Group demonstrated relief of psychotic symptoms of Parkinson’s disease with clozapine, 6.25 mg/d.9 The Clozapine Study Group found complete resolution of hallucinations in some patients within 1 day of initiating clozapine. Among patients in this study who did not see immediate benefit, most showed significant improvement of psychotic symptoms in 1 or 2 weeks.10
TREATMENT Few options
Ms. L’s psychiatrist and primary care physician start her on a series of medications. Donepezil is initiated for suspected dementia. We begin a trial of quetiapine to address the hallucinations, but the drug makes her movement symptoms worse. Risperidone also is tried but, again, the drugs make movement symptoms, particularly gait instability, tremor, and rigidity worse without alleviating the hallucinations. Neuroleptics seem to exacerbate confusion. Because of worsening depressive symptoms and our concern over possible pseudodementia, we try several selective serotonin reuptake inhibitors (SSRIs) and mirtazapine. Antidepressants have little effect on her depressive symptoms and do not improve hallucinations or insomnia.
Ms. L’s signs and symptoms become worse over the next few months, with more severe hallucinations, agitation, insomnia, and gait instability. Her agitation over the hallucinations increases and she begins pouring bleach around herself in bed and spraying her house with toxic bug spray. Ms. L’s family brings her to the hospital after they observe her scratching the hallucinatory creatures off of her skin with a razor blade and trying to pry them out of her mouth with a piece of metal.
In the hospital, medical and neurologic workups rule out organic causes for her symptoms and signs. MRI is consistent with imaging from 6 months earlier. Focal neurologic signs are absent. Blood work is within normal limits, failing to reveal any pathology that would suggest a cause for her symptoms and signs, such as syphilis, vitamin deficiency, and Lyme disease.
Ms. L’s symptoms were consistent with consensus guideline criteria for a clinical diagnosis of DLB (Table 1).11-18
She is started on low-dose quetiapine, which she tolerates poorly with worsening confusion, rigidity, tremor, and gait instability. Because other agents failed, Ms. L’s providers and family decide on a trial of clozapine.
Within 24 hours after the first dose of clozapine, 25 mg, sleep improves, the tactile component of hallucinations diminish, and she begins to spend increasing periods of time “observing the creatures” rather than fighting with them.
Over the next few days, Ms. L’s attitude towards the creatures changes. Now, as she sits observing them intently, the hallucinations evolve: rather than tormenting her and causing distress, the plant-creatures burst apart and a miniature knight on horseback charges out. The rest of the creatures then gather into a rank and file and the knight leads them to the nearest exit.
Clozapine is titrated to 50 mg/d, which she tolerates well without exacerbation of cognitive symptoms or movement disorder. The only notable adverse effect at the time of her discharge is sialorrhea.
with an antipsychotic?
a) start low and go slow
b) monitor her heart rate and blood pressure
c) readminister the Montreal Cognitive Assessment
d) all of the above
The authors’ observations
Ideally, in psychosis, antipsychotics eliminate positive symptoms such as hallucinations and delusions. In DLB, the aim is to alleviate the agitation and suffering brought on by the psychotic symptoms without exacerbating other motor and cognitive symptoms. The hallucinations are obstinate, and it is a well-known quality of this disorder that patients are exceptionally susceptible to a range of antipsychotic side effects including cognitive impairment, fatigue, neuroleptic malignant syndrome, and parkinsonism.19
Treatment in DLB requires trial and error, and medications with fewer associated risks should be administered first. Patients with DLB treated with neuroleptics have an increased risk of death compared with those who are not treated.19 Moreover, prescribing information for clozapine includes a black-box warning that the drug:
- is not approved for dementia-related psychosis and
- is associated with an increased risk of death in elderly patients with these conditions, similar to what is seen with other neuroleptics.20
Despite these well-known concerns, it remains difficult for clinicians not to try to treat the distress caused by these symptoms.
We chose clozapine for Ms. L because:
- other neuroleptics failed
- acetylcholinesterase inhibitors did not alleviate Ms. L’s psychosis and associated behavioral disturbance
- there is substantial evidence that the drug can be effective in Parkinson’s disease with psychosis.
There is controversy regarding use of clozapine in DLB. In one case series, clozapine trigger extreme neuroleptic reactions in some patients, similar to what occurs with other second-generation antipsychotics.21 Another case series provides examples of the drug’s efficacy in treating hallucinations and delusions with minimal adverse effects.22
It is important to emphasize that Ms. L’s hallucinations did not go away; rather, they changed to a more benign presentation that she could manage and, occasionally, found pleasant. Ultimately, her agitation—the primary target of treatment—improved markedly with the arrival of the knight in shining armor.
Treatment recommendations
If neuropsychiatric symptoms in DLB are the primary concern of the patient and family, we recommend the following:
- Begin treatment with a cholinesterase inhibitor. The best evidence exists for rivastigmine and donepezil. These drugs have a low risk of side effects, which are primarily gastrointestinal effects with some reports of worsening extrapyramidal symptoms.23-25
- If the patient obtains minimal benefit or develops a significant adverse effect from cholinesterase inhibitors, consider memantine. Its efficacy is under examination and results are mixed; it can be used in combination with cholinesterase inhibitors.26-28
- If psychotic symptoms are upsetting and refractory to other therapies, consider antipsychotics. Avoid first-generation antipsychotics. The American Psychiatric Association recommends aripiprazole or quetiapine initially, although there is little evidence comparing neuroleptics in DLB.29 Because of its risks, reserve clozapine for refractory cases. An exception might be made for patients sensitive to extrapyramidal effects, in whom clozapine could be considered earlier.
There are no formal neuroleptic dosing guidelines beyond a general urging towards minimalism. Mosimann and McKeith30 recommend clozapine, 12.5 mg/d; olanzapine, 2.5 mg/d; risperidone, 0.25 mg/d; or quetiapine, 12.5 mg/d. Such dosages might be effective while producing only minimal side effects.9,31
SSRIs and other antidepressants have not been shown to improve neuropsychiatric symptoms, and often are poorly tolerated.32
One study found efficacy with electroconvulsive therapy and transcranial magnetic stimulation in treatment-resistant patients.33
In addition to these treatments, nonpharmaceutical interventions should be employed from the earliest stages of diagnosis and treatment (Table 2). See the Figure for an algorithm for treating DLB. These include educational and behavioral interventions, social support, psychological interventions, and environmental therapies and modifications.
OUTCOME New friends
The creatures return from time to time, Ms. L reports, but are no longer upsetting because the white knight (a sort of mental deus ex machina) leads the once-terrifying things away. She describes the hallucination as a kind of zoological observation, refers to the creatures that once horrified her as “her friends,” and chuckles as she observes their natural history. This new, far more benign hallucination becomes a mainstay of her symptoms, and she is discharged to the care of her husband and family.
Soon after her discharge, her hallucinations resolved completely, but returned briefly when Ms. L resumed smoking cigarettes because smoking is known to lower clozapine serum levels.34
Bottom Line
Consider a low dosage of a neuroleptic when a patient suffers significant distress and behavioral disturbance related to psychotic symptoms in dementia with Lewy bodies and those problems are not relieved by other agents. Low-dose clozapine is an option for refractory psychotic symptoms or in patients with severe extrapyramidal sensitivity. Start low, and go slow.
Related Resources
- Bishnoi RJ, Grossberg GT, Manepalli J. Differentiating Alzheimer’s disease from dementia with Lewy bodies. Current Psychiatry. 2012;11(11):22-27.
- McKeith I, Emre M. Management of Parkinson’s disease dementia and dementia with Lewy bodies. In: Emre M, ed. Cognitive impairment and dementia in Parkinson’s disease. Oxford, United Kingdom: Oxford University Press; 2010:245-256.
Drug Brand Names
Aripiprazole • Abilify Mirtazapine • Remeron
Clozapine • Clozaril Olanzapine • Zyprexa
Donepezil • Aricept Quetiapine • Seroquel
Haloperidol • Haldol Risperidone • Risperdal
Memantine • Namenda Rivastigmine • Exelon
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Wesnes KA, McKeith IG, Ferrara R, et al. Effects of rivastigmine on cognitive function in dementia with Lewy bodies: a randomised placebo-controlled international study using the Cognitive Drug Research computerized assessment system. Dement Geriatr Cogn Disord. 2002; 13(3):183-192.
2. Weintraub D, Hurtig HI. Presentation and management of psychosis in Parkinson’s disease and dementia with Lewy bodies. Am J Psychiatry. 2007;164(10):1491-1498.
3. McKeith IG, Wesnes KA, Perry E, et al. Hallucinations predict attentional improvements with rivastigmine in dementia with Lewy bodies. Dement Geriatr Cogn Disord. 2004;18(1):94-100.
4. Emre M, Tsolaki , Bonuccelli U, et al. Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2010;9(10):969-977.
5. Aarsland D, Ballard C, Walker Z, et al. Clinical trials of dementia with Lewy bodies and Parkinson’s disease dementia. Curr Neurol Neurosci Rep. 2012;12(5):492-501.
6. Drach LM. Drug treatment of dementia with Lewy bodies and Parkinson’s disease dementia--common features and differences [in German]. Med Monatsschr Pharm. 2011; 34(2):47-52.
7. Frieling H, Hillemacher T, Ziegenbein M, et al. Treating dopamimetic psychosis in Parkinson’s disease: Structured review and meta-analysis. Eur Neuropsychopharmacol. 2007;17(3):165-171.
8. Marti MJ, Tolosa E, de la Cerda A. Dementia in Parkinson’s disease. J Neurol. 2007;254(suppl 5):41-48.
9. French Clozapine Parkinson Study Group. Clozapine in drug-induced psychosis in Parkinson’s disease. Lancet. 1999;353(9169):2041-2042.
10. Friedman JH, Factor SA. Atypical antipsychotics in the treatment of drug-induced psychosis in Parkinson’s disease. Mov Disord. 2000;15(2):201-211.
11. McKeith IG, Galasko D, Kosaka K, et al. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the consortium on DLB international workshop. Neurology. 1996;47(5):1113-1124.
12. McKeith IG, Ballard CG, Perry RH et al. Prospective validation of consensus criteria for the diagnosis of dementia with Lewy bodies. Neurology. 2000;54(5):1050-1058.
13. McKeith IG, Dickson DW, Lowe J, et al. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology. 2005;65(12):1863-1872.
14. McKeith IG. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the Consortium on DLB International Workshop. J Alzheimers Dis. 2006;9(suppl 3):417-423.
15. Geser F, Wenning GK, Poewe W, et al. How to diagnose dementia with Lewy bodies: state of the art. Mov Disord. 2005;20(suppl 12):S11-20.
16. Latoo J, Jan F. Dementia with Lewy bodies: clinical review. British Journal of Medical Practioners. 2008;1(1):10-14.
17. McKeith I. Dementia with Lewy bodies. Dialogues Clin Neurosci. 2004;6(3):333-341.
18. Litvan I, Bhatia KP, Burn DJ, et al; Movement Disorders Society Scientific Issues Committee. SIC Task Force Appraisal of clinical diagnostic criteria for parkinsonian disorders. Mov Disord. 2003;18(5):467-486.
19. McKeith I, Fairbairn A, Perry R, et al. Neuroleptic sensitivity in patients with senile dementia of Lewy body type. BMJ. 1992;305(6855):673-678.
20. Clozapine Monitoring Guidelines. 2008. http://www.clozapineregistry.com/resuming_treatment_after_interruption.pdf.ashx. Accessed October 31, 2013.
21. Burke WJ, Pfeiffer RF, McComb RD. Neuroleptic sensitivity to clozapine in dementia with Lewy bodies. J Neuropsychiatry Clin Neurosci. 1998;10(2):227-229.
22. Chacko RC, Hurley RA, Jankovic J. Clozapine use in diffuse Lewy body disease. J Neuropsychiatry Clin Neurosci. 1993;5(2):206-208.
23. McKeith I, Del Ser T, Spano P, et al. Efficacy of rivastigmine in dementia with Lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet. 2000;356(9247):2031-2036
24. Mori E, Ikeda M, Kosaka K; Donepezil-DLB Study Investiagors. Donepezil for dementia with Lewy bodies: a randomized, placebo‐controlled trial. Ann Neurol. 2012; 72(1):41-52.
25. Ukai K, Aleksic B, Ishihara R, et al. Efficacy of donepezil for the treatment of visual and multiple sensory hallucinations in dementia with Lewy bodies. Clinical Neuropsychopharmacology and Therapeutics. 2011;2:56-58.
26. Aarsland D, Ballard C, Walker Z, et al. Memantine in patients with Parkinson’s disease dementia or dementia with Lewy bodies: a double-blind, placebo-controlled, multicentre trial. Lancet Neurol. 2009;8(7):613-618.
27. Boeve BF, Silber MH, Ferman TJ. Melatonin for treatment of REM sleep behavior disorder in neurologic disorders: results in 14 patients. Sleep Med. 2003;4(4):281-284.
28. Mathys ML, McCarrell J, Sleeper RB, et al. Visual hallucinations treated with the reinitiation of memantine in a patient with Lewy body dementia. Ann Pharmacother. 2013;47(2):e10.
29. American Psychiatric Association. Practice guideline for the treatment of patients with Alzheimer’s disease and other dementias. 2nd ed. http://psychiatryonline.org/pdfaccess.ashx?ResourceID=243205&PDFSource=6. Accessed November 1, 2013.
30. Mosimann U, McKeith IG. Dementia with lewy bodies—diagnosis and treatment. Swiss Med Wkly. 2003;133:131-142.
31. Baskys A, Davis P Atypical antipsychotic quetiapine in the treatment of the psychosis associated with Lewy body dementia. Neurobiol Aging. 2002;23:S63.
32. Culo S, Mulsant BH, Rosen J, et al. Treating neuropsychiatric symptoms in dementia with Lewy bodies: a randomized controlled-trial. Alzheimer Dis Assoc Disord. 2010;24(4):306-364.
33. Takahashi S, Mizukami K, Yasuno F, et al. Depression associated with dementia with Lewy bodies (DLB) and the effect of somatotherapy. Psychogeriatrics. 2009;9(2):56-61.
34. van der Weide J, Steijns LS, van Weelden MJ. The effect of smoking and cytochrome P450 CYP1A2 genetic polymorphism on clozapine clearance and dose requirement. Pharmacogenetics. 2003;13(3):169-172.
1. Wesnes KA, McKeith IG, Ferrara R, et al. Effects of rivastigmine on cognitive function in dementia with Lewy bodies: a randomised placebo-controlled international study using the Cognitive Drug Research computerized assessment system. Dement Geriatr Cogn Disord. 2002; 13(3):183-192.
2. Weintraub D, Hurtig HI. Presentation and management of psychosis in Parkinson’s disease and dementia with Lewy bodies. Am J Psychiatry. 2007;164(10):1491-1498.
3. McKeith IG, Wesnes KA, Perry E, et al. Hallucinations predict attentional improvements with rivastigmine in dementia with Lewy bodies. Dement Geriatr Cogn Disord. 2004;18(1):94-100.
4. Emre M, Tsolaki , Bonuccelli U, et al. Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2010;9(10):969-977.
5. Aarsland D, Ballard C, Walker Z, et al. Clinical trials of dementia with Lewy bodies and Parkinson’s disease dementia. Curr Neurol Neurosci Rep. 2012;12(5):492-501.
6. Drach LM. Drug treatment of dementia with Lewy bodies and Parkinson’s disease dementia--common features and differences [in German]. Med Monatsschr Pharm. 2011; 34(2):47-52.
7. Frieling H, Hillemacher T, Ziegenbein M, et al. Treating dopamimetic psychosis in Parkinson’s disease: Structured review and meta-analysis. Eur Neuropsychopharmacol. 2007;17(3):165-171.
8. Marti MJ, Tolosa E, de la Cerda A. Dementia in Parkinson’s disease. J Neurol. 2007;254(suppl 5):41-48.
9. French Clozapine Parkinson Study Group. Clozapine in drug-induced psychosis in Parkinson’s disease. Lancet. 1999;353(9169):2041-2042.
10. Friedman JH, Factor SA. Atypical antipsychotics in the treatment of drug-induced psychosis in Parkinson’s disease. Mov Disord. 2000;15(2):201-211.
11. McKeith IG, Galasko D, Kosaka K, et al. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the consortium on DLB international workshop. Neurology. 1996;47(5):1113-1124.
12. McKeith IG, Ballard CG, Perry RH et al. Prospective validation of consensus criteria for the diagnosis of dementia with Lewy bodies. Neurology. 2000;54(5):1050-1058.
13. McKeith IG, Dickson DW, Lowe J, et al. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology. 2005;65(12):1863-1872.
14. McKeith IG. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the Consortium on DLB International Workshop. J Alzheimers Dis. 2006;9(suppl 3):417-423.
15. Geser F, Wenning GK, Poewe W, et al. How to diagnose dementia with Lewy bodies: state of the art. Mov Disord. 2005;20(suppl 12):S11-20.
16. Latoo J, Jan F. Dementia with Lewy bodies: clinical review. British Journal of Medical Practioners. 2008;1(1):10-14.
17. McKeith I. Dementia with Lewy bodies. Dialogues Clin Neurosci. 2004;6(3):333-341.
18. Litvan I, Bhatia KP, Burn DJ, et al; Movement Disorders Society Scientific Issues Committee. SIC Task Force Appraisal of clinical diagnostic criteria for parkinsonian disorders. Mov Disord. 2003;18(5):467-486.
19. McKeith I, Fairbairn A, Perry R, et al. Neuroleptic sensitivity in patients with senile dementia of Lewy body type. BMJ. 1992;305(6855):673-678.
20. Clozapine Monitoring Guidelines. 2008. http://www.clozapineregistry.com/resuming_treatment_after_interruption.pdf.ashx. Accessed October 31, 2013.
21. Burke WJ, Pfeiffer RF, McComb RD. Neuroleptic sensitivity to clozapine in dementia with Lewy bodies. J Neuropsychiatry Clin Neurosci. 1998;10(2):227-229.
22. Chacko RC, Hurley RA, Jankovic J. Clozapine use in diffuse Lewy body disease. J Neuropsychiatry Clin Neurosci. 1993;5(2):206-208.
23. McKeith I, Del Ser T, Spano P, et al. Efficacy of rivastigmine in dementia with Lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet. 2000;356(9247):2031-2036
24. Mori E, Ikeda M, Kosaka K; Donepezil-DLB Study Investiagors. Donepezil for dementia with Lewy bodies: a randomized, placebo‐controlled trial. Ann Neurol. 2012; 72(1):41-52.
25. Ukai K, Aleksic B, Ishihara R, et al. Efficacy of donepezil for the treatment of visual and multiple sensory hallucinations in dementia with Lewy bodies. Clinical Neuropsychopharmacology and Therapeutics. 2011;2:56-58.
26. Aarsland D, Ballard C, Walker Z, et al. Memantine in patients with Parkinson’s disease dementia or dementia with Lewy bodies: a double-blind, placebo-controlled, multicentre trial. Lancet Neurol. 2009;8(7):613-618.
27. Boeve BF, Silber MH, Ferman TJ. Melatonin for treatment of REM sleep behavior disorder in neurologic disorders: results in 14 patients. Sleep Med. 2003;4(4):281-284.
28. Mathys ML, McCarrell J, Sleeper RB, et al. Visual hallucinations treated with the reinitiation of memantine in a patient with Lewy body dementia. Ann Pharmacother. 2013;47(2):e10.
29. American Psychiatric Association. Practice guideline for the treatment of patients with Alzheimer’s disease and other dementias. 2nd ed. http://psychiatryonline.org/pdfaccess.ashx?ResourceID=243205&PDFSource=6. Accessed November 1, 2013.
30. Mosimann U, McKeith IG. Dementia with lewy bodies—diagnosis and treatment. Swiss Med Wkly. 2003;133:131-142.
31. Baskys A, Davis P Atypical antipsychotic quetiapine in the treatment of the psychosis associated with Lewy body dementia. Neurobiol Aging. 2002;23:S63.
32. Culo S, Mulsant BH, Rosen J, et al. Treating neuropsychiatric symptoms in dementia with Lewy bodies: a randomized controlled-trial. Alzheimer Dis Assoc Disord. 2010;24(4):306-364.
33. Takahashi S, Mizukami K, Yasuno F, et al. Depression associated with dementia with Lewy bodies (DLB) and the effect of somatotherapy. Psychogeriatrics. 2009;9(2):56-61.
34. van der Weide J, Steijns LS, van Weelden MJ. The effect of smoking and cytochrome P450 CYP1A2 genetic polymorphism on clozapine clearance and dose requirement. Pharmacogenetics. 2003;13(3):169-172.