User login
Adjuvant S-1 plus endocrine therapy improves invasive DFS in ER+/HER2− breast cancer
Key clinical point: In patients with estrogen receptor-positive/human epidermal growth factor receptor 2-negative breast cancer (ER+/HER2− BC), adjuvant S-1 plus standard endocrine therapy (ET) significantly reduced invasive disease-free survival (IDFS) events compared with ET alone.
Major finding: IDFS events were observed in 16% vs. 11% of patients in ET-only vs. ET+S-1 groups (hazard ratio [HR], 0.63; P = .0003), respectively. The estimated 5-year IDFS was 87% in the ET+S-1 group vs. 82% in the ET-only group. However, adverse events were higher in patients receiving ET+S-1 therapy (99%) vs. ET alone (79%) but were majorly manageable.
Study details: Phase 3 trial included 1,930 women with ER+/HER− BC with intermediate to high risk for recurrence who were randomly allocated to receive either ET alone (n=973) or ET+S-1 (n=957) for a median follow-up of 52.2 months.
Disclosures: This study was sponsored by the Public Health Research Foundation, Japan, and funded by Taiho Pharmaceuticals. Masakazu Toi and other study authors reported funding and ties with various pharmaceutical companies including Taiho during and outside the conduct of the study.
Source: Toi M et al. Lancet Oncol. 2021 Jan 1. doi: 10.1016/S1470-2045(20)30534-9.
Key clinical point: In patients with estrogen receptor-positive/human epidermal growth factor receptor 2-negative breast cancer (ER+/HER2− BC), adjuvant S-1 plus standard endocrine therapy (ET) significantly reduced invasive disease-free survival (IDFS) events compared with ET alone.
Major finding: IDFS events were observed in 16% vs. 11% of patients in ET-only vs. ET+S-1 groups (hazard ratio [HR], 0.63; P = .0003), respectively. The estimated 5-year IDFS was 87% in the ET+S-1 group vs. 82% in the ET-only group. However, adverse events were higher in patients receiving ET+S-1 therapy (99%) vs. ET alone (79%) but were majorly manageable.
Study details: Phase 3 trial included 1,930 women with ER+/HER− BC with intermediate to high risk for recurrence who were randomly allocated to receive either ET alone (n=973) or ET+S-1 (n=957) for a median follow-up of 52.2 months.
Disclosures: This study was sponsored by the Public Health Research Foundation, Japan, and funded by Taiho Pharmaceuticals. Masakazu Toi and other study authors reported funding and ties with various pharmaceutical companies including Taiho during and outside the conduct of the study.
Source: Toi M et al. Lancet Oncol. 2021 Jan 1. doi: 10.1016/S1470-2045(20)30534-9.
Key clinical point: In patients with estrogen receptor-positive/human epidermal growth factor receptor 2-negative breast cancer (ER+/HER2− BC), adjuvant S-1 plus standard endocrine therapy (ET) significantly reduced invasive disease-free survival (IDFS) events compared with ET alone.
Major finding: IDFS events were observed in 16% vs. 11% of patients in ET-only vs. ET+S-1 groups (hazard ratio [HR], 0.63; P = .0003), respectively. The estimated 5-year IDFS was 87% in the ET+S-1 group vs. 82% in the ET-only group. However, adverse events were higher in patients receiving ET+S-1 therapy (99%) vs. ET alone (79%) but were majorly manageable.
Study details: Phase 3 trial included 1,930 women with ER+/HER− BC with intermediate to high risk for recurrence who were randomly allocated to receive either ET alone (n=973) or ET+S-1 (n=957) for a median follow-up of 52.2 months.
Disclosures: This study was sponsored by the Public Health Research Foundation, Japan, and funded by Taiho Pharmaceuticals. Masakazu Toi and other study authors reported funding and ties with various pharmaceutical companies including Taiho during and outside the conduct of the study.
Source: Toi M et al. Lancet Oncol. 2021 Jan 1. doi: 10.1016/S1470-2045(20)30534-9.
Does metabolic syndrome influence outcomes in triple-negative breast cancer?
Key clinical point: Metabolic syndrome (MS) is associated with poorer disease-free survival (DFS), but not with overall survival (OS) in patients with triple-negative breast cancer (TNBC). Hypertension was the only MS component associated with poor DFS and OS.
Major finding: MS was significantly associated with poor DFS (adjusted HR [aHR], 2.24; P = .030), but not OS (aHR, 1.92; P = .103). Hypertension was significantly associated with worse DFS (aHR, 3.63; P = .006) and OS (aHR, 3.45; P = .035).
Study details: Retrospective study of 177 patients with TNBC with (n=48) or without (n=129) MS who were followed-up for at least 5 years.
Disclosures: The study was supported by the Sharpe Strumia Foundation. The authors declared no conflicts of interest.
Source: Kennard K et al. Breast Cancer Res Treat. 2021 Jan 3. doi: 10.1007/s10549-020-06034-1.
Key clinical point: Metabolic syndrome (MS) is associated with poorer disease-free survival (DFS), but not with overall survival (OS) in patients with triple-negative breast cancer (TNBC). Hypertension was the only MS component associated with poor DFS and OS.
Major finding: MS was significantly associated with poor DFS (adjusted HR [aHR], 2.24; P = .030), but not OS (aHR, 1.92; P = .103). Hypertension was significantly associated with worse DFS (aHR, 3.63; P = .006) and OS (aHR, 3.45; P = .035).
Study details: Retrospective study of 177 patients with TNBC with (n=48) or without (n=129) MS who were followed-up for at least 5 years.
Disclosures: The study was supported by the Sharpe Strumia Foundation. The authors declared no conflicts of interest.
Source: Kennard K et al. Breast Cancer Res Treat. 2021 Jan 3. doi: 10.1007/s10549-020-06034-1.
Key clinical point: Metabolic syndrome (MS) is associated with poorer disease-free survival (DFS), but not with overall survival (OS) in patients with triple-negative breast cancer (TNBC). Hypertension was the only MS component associated with poor DFS and OS.
Major finding: MS was significantly associated with poor DFS (adjusted HR [aHR], 2.24; P = .030), but not OS (aHR, 1.92; P = .103). Hypertension was significantly associated with worse DFS (aHR, 3.63; P = .006) and OS (aHR, 3.45; P = .035).
Study details: Retrospective study of 177 patients with TNBC with (n=48) or without (n=129) MS who were followed-up for at least 5 years.
Disclosures: The study was supported by the Sharpe Strumia Foundation. The authors declared no conflicts of interest.
Source: Kennard K et al. Breast Cancer Res Treat. 2021 Jan 3. doi: 10.1007/s10549-020-06034-1.
Palbociclib+ET fails to prolong PFS in AI-resistant metastatic breast cancer
Key clinical point: Palbociclib+endocrine therapy (ET) did not yield superior progression-free survival (PFS) vs. capecitabine in patients with aromatase inhibitor-resistant metastatic breast cancer (AIR-MBC) but was well tolerated and improved quality of life (QoL).
Major finding: Median PFS with palbociclib+fulvestrant vs. capecitabine was 7.5 vs. 10.0 months (adjusted hazard ratio [aHR], 1.13; P = .398). Palbociclib+ET did not show PFS superiority vs. capecitabine in patients with wild-type estrogen receptor-1 AIR-MBC (8.0 vs. 10.6 months; aHR, 1.11; P = .404). Palbociclib+ET was better tolerated and improved QoL (aHR, 0.67; P = .001).
Study details: Phase 3 PEARL trial randomly allocated postmenopausal women with AIR-MBC to receive either palbociclib+exemestane or capecitabine (n=296) or palbociclib+fulvestrant or capecitabine (n=305).
Disclosures: This study was supported by Pfizer and AstraZeneca and sponsored by GEICAM Spanish Breast Cancer Group. The lead author reported ties with AstraZeneca, Pfizer, Amgen, Taiho Oncology, Roche/Genentech, Novartis, PharmaMar, Eli Lilly, PUMA, and Daiichi-Sankyo. Other investigators reported owning stocks, being an employee, receiving support, and/or consulting for various pharmaceutical companies including Pfizer and AstraZeneca.
Source: Martin M et al. Ann Oncol. 2020 Dec 29. doi: 10.1016/j.annonc.2020.12.013.
Key clinical point: Palbociclib+endocrine therapy (ET) did not yield superior progression-free survival (PFS) vs. capecitabine in patients with aromatase inhibitor-resistant metastatic breast cancer (AIR-MBC) but was well tolerated and improved quality of life (QoL).
Major finding: Median PFS with palbociclib+fulvestrant vs. capecitabine was 7.5 vs. 10.0 months (adjusted hazard ratio [aHR], 1.13; P = .398). Palbociclib+ET did not show PFS superiority vs. capecitabine in patients with wild-type estrogen receptor-1 AIR-MBC (8.0 vs. 10.6 months; aHR, 1.11; P = .404). Palbociclib+ET was better tolerated and improved QoL (aHR, 0.67; P = .001).
Study details: Phase 3 PEARL trial randomly allocated postmenopausal women with AIR-MBC to receive either palbociclib+exemestane or capecitabine (n=296) or palbociclib+fulvestrant or capecitabine (n=305).
Disclosures: This study was supported by Pfizer and AstraZeneca and sponsored by GEICAM Spanish Breast Cancer Group. The lead author reported ties with AstraZeneca, Pfizer, Amgen, Taiho Oncology, Roche/Genentech, Novartis, PharmaMar, Eli Lilly, PUMA, and Daiichi-Sankyo. Other investigators reported owning stocks, being an employee, receiving support, and/or consulting for various pharmaceutical companies including Pfizer and AstraZeneca.
Source: Martin M et al. Ann Oncol. 2020 Dec 29. doi: 10.1016/j.annonc.2020.12.013.
Key clinical point: Palbociclib+endocrine therapy (ET) did not yield superior progression-free survival (PFS) vs. capecitabine in patients with aromatase inhibitor-resistant metastatic breast cancer (AIR-MBC) but was well tolerated and improved quality of life (QoL).
Major finding: Median PFS with palbociclib+fulvestrant vs. capecitabine was 7.5 vs. 10.0 months (adjusted hazard ratio [aHR], 1.13; P = .398). Palbociclib+ET did not show PFS superiority vs. capecitabine in patients with wild-type estrogen receptor-1 AIR-MBC (8.0 vs. 10.6 months; aHR, 1.11; P = .404). Palbociclib+ET was better tolerated and improved QoL (aHR, 0.67; P = .001).
Study details: Phase 3 PEARL trial randomly allocated postmenopausal women with AIR-MBC to receive either palbociclib+exemestane or capecitabine (n=296) or palbociclib+fulvestrant or capecitabine (n=305).
Disclosures: This study was supported by Pfizer and AstraZeneca and sponsored by GEICAM Spanish Breast Cancer Group. The lead author reported ties with AstraZeneca, Pfizer, Amgen, Taiho Oncology, Roche/Genentech, Novartis, PharmaMar, Eli Lilly, PUMA, and Daiichi-Sankyo. Other investigators reported owning stocks, being an employee, receiving support, and/or consulting for various pharmaceutical companies including Pfizer and AstraZeneca.
Source: Martin M et al. Ann Oncol. 2020 Dec 29. doi: 10.1016/j.annonc.2020.12.013.
One-third of HER2+ and triple-negative metastatic breast cancer patients develop brain metastases
Key clinical point: Incidence of brain metastases is high in patients with human epidermal growth factor receptor 2-positive (HER2+) and triple-negative metastatic breast cancer (MBC), highlighting the need for relevant screening in these high-risk populations.
Major finding: The pooled cumulative incidence of brain metastases in patients with HER2+ and triple-negative MBC was 31% (interquartile range [IQR], 24.0-34.0) and 32% (IQR,18.5-40.6) during a median follow-up of 30.7 and 32.8 months, respectively. The incidence of brain metastases per patient-year was 13% for HER2+ and triple negative MBC.
Study details: Meta-analysis of 25 studies including patients with HER2+ MBC (n=5,971), triple-negative (n=4,102), and hormone receptor+/HER2− MBC (n=14,656).
Disclosures: This study did not receive any funding. The lead author reported no disclosures. Some of his coinvestigators reported ties with various pharmaceutical companies.
Source: Kuksis M et al. Neuro Oncol. 2020 Dec 23. doi: 10.1093/neuonc/noaa285.
Key clinical point: Incidence of brain metastases is high in patients with human epidermal growth factor receptor 2-positive (HER2+) and triple-negative metastatic breast cancer (MBC), highlighting the need for relevant screening in these high-risk populations.
Major finding: The pooled cumulative incidence of brain metastases in patients with HER2+ and triple-negative MBC was 31% (interquartile range [IQR], 24.0-34.0) and 32% (IQR,18.5-40.6) during a median follow-up of 30.7 and 32.8 months, respectively. The incidence of brain metastases per patient-year was 13% for HER2+ and triple negative MBC.
Study details: Meta-analysis of 25 studies including patients with HER2+ MBC (n=5,971), triple-negative (n=4,102), and hormone receptor+/HER2− MBC (n=14,656).
Disclosures: This study did not receive any funding. The lead author reported no disclosures. Some of his coinvestigators reported ties with various pharmaceutical companies.
Source: Kuksis M et al. Neuro Oncol. 2020 Dec 23. doi: 10.1093/neuonc/noaa285.
Key clinical point: Incidence of brain metastases is high in patients with human epidermal growth factor receptor 2-positive (HER2+) and triple-negative metastatic breast cancer (MBC), highlighting the need for relevant screening in these high-risk populations.
Major finding: The pooled cumulative incidence of brain metastases in patients with HER2+ and triple-negative MBC was 31% (interquartile range [IQR], 24.0-34.0) and 32% (IQR,18.5-40.6) during a median follow-up of 30.7 and 32.8 months, respectively. The incidence of brain metastases per patient-year was 13% for HER2+ and triple negative MBC.
Study details: Meta-analysis of 25 studies including patients with HER2+ MBC (n=5,971), triple-negative (n=4,102), and hormone receptor+/HER2− MBC (n=14,656).
Disclosures: This study did not receive any funding. The lead author reported no disclosures. Some of his coinvestigators reported ties with various pharmaceutical companies.
Source: Kuksis M et al. Neuro Oncol. 2020 Dec 23. doi: 10.1093/neuonc/noaa285.
Partial breast irradiation vs. whole breast irradiation for early breast cancer
Key clinical point: Rates of ipsilateral breast tumor recurrence (IBTR) were similar between partial breast irradiation (PBI) and whole breast irradiation (WBI). Acute skin toxicities were lower with PBI.
Major finding: The rate of IBTR at 5 years was not different between PBI (1.8%; 95% highest posterior density [HPD], 0.68%-3.2%) and WBI (1.7%; 95% HPD, 0.92%-2.4%). Rates of grade 2 or greater acute skin toxicity were lower with PBI (7.1%; 95% HPD, 0%-63.4%) vs. WBI (47.5%; 95% HPD, 0%-93.4%).
Study details: Meta-analysis of 7 randomized trials including 9,758 patients receiving either WBI (n=4,840) or PBI (n=4,918).
Disclosures: No funding source was identified. The lead author reported ties with Impedimed, PreludeDX, Varian Medical Systems, and Vision RT. Some other coinvestigators reported receiving grants or consultation fees from various pharmaceutical companies.
Source: Shah C et al. Ann Surg Oncol. 2021 Jan 3. doi: 10.1245/s10434-020-09447-w.
Key clinical point: Rates of ipsilateral breast tumor recurrence (IBTR) were similar between partial breast irradiation (PBI) and whole breast irradiation (WBI). Acute skin toxicities were lower with PBI.
Major finding: The rate of IBTR at 5 years was not different between PBI (1.8%; 95% highest posterior density [HPD], 0.68%-3.2%) and WBI (1.7%; 95% HPD, 0.92%-2.4%). Rates of grade 2 or greater acute skin toxicity were lower with PBI (7.1%; 95% HPD, 0%-63.4%) vs. WBI (47.5%; 95% HPD, 0%-93.4%).
Study details: Meta-analysis of 7 randomized trials including 9,758 patients receiving either WBI (n=4,840) or PBI (n=4,918).
Disclosures: No funding source was identified. The lead author reported ties with Impedimed, PreludeDX, Varian Medical Systems, and Vision RT. Some other coinvestigators reported receiving grants or consultation fees from various pharmaceutical companies.
Source: Shah C et al. Ann Surg Oncol. 2021 Jan 3. doi: 10.1245/s10434-020-09447-w.
Key clinical point: Rates of ipsilateral breast tumor recurrence (IBTR) were similar between partial breast irradiation (PBI) and whole breast irradiation (WBI). Acute skin toxicities were lower with PBI.
Major finding: The rate of IBTR at 5 years was not different between PBI (1.8%; 95% highest posterior density [HPD], 0.68%-3.2%) and WBI (1.7%; 95% HPD, 0.92%-2.4%). Rates of grade 2 or greater acute skin toxicity were lower with PBI (7.1%; 95% HPD, 0%-63.4%) vs. WBI (47.5%; 95% HPD, 0%-93.4%).
Study details: Meta-analysis of 7 randomized trials including 9,758 patients receiving either WBI (n=4,840) or PBI (n=4,918).
Disclosures: No funding source was identified. The lead author reported ties with Impedimed, PreludeDX, Varian Medical Systems, and Vision RT. Some other coinvestigators reported receiving grants or consultation fees from various pharmaceutical companies.
Source: Shah C et al. Ann Surg Oncol. 2021 Jan 3. doi: 10.1245/s10434-020-09447-w.
MS bears no effect on certain pregnancy complications, stillbirth, or congenital deformation
, according to a new study published online Feb. 3 in Neurology Clinical Practice. While pregnancy and childbirth are not regarded as conditions that engender high-risk pregnancy in the MS population, previous studies evaluating the effects of MS on pregnancy and parturition have yet to fully elucidate some outcomes for pregnant women and their babies in multiple sclerosis.
“Women with multiple sclerosis may be understandably concerned about the risk of pregnancy,” said Melinda Magyari, MD, PhD, a consultant at the University of Copenhagen. “While previous research has shown there is no higher risk of birth defect for babies born to women with MS, we wanted to find out if women with MS are at risk for a variety of pregnancy complications.”
MS is regarded as a progressive, neurological disease mediated by the immune system that demands careful consideration of numerous situations and life changes including family planning. The MS population is overwhelmingly female, as women account for three out of every four cases of MS. The majority of these women range from 20 to 40 years of age at the time of being diagnosed with MS. Despite the unknown risks of pregnancy-related complications and various perinatal complications in this patient population, women who have MS are not discouraged from conceiving.
Assessing pregnancy outcomes
This nationwide, population-based, cross-sectional study evaluated the pregnancies of 2,930 women with MS between Jan. 1, 1997, and Dec. 31, 2016, registered in the Danish Multiple Sclerosis Registry. The researchers compared pregnancy-related and prenatal outcomes to a 5% random sample of 56,958 randomly-selected pregnant women from Denmark’s general population who did not have MS. They found no differences in the risks associated with several pregnancy-related complications (e.g., preeclampsia, gestational diabetes, or placental complications), emergency Cesarean section (C-section), instrumental delivery, stillbirth, preterm birth, or congenital malformation. Apgar scores were low in both groups. A composite of various biometrics in newborns such as reflexes, muscle tone, and heart rate immediately following birth, the Apgar score is used to help assess the neonatal health, with a value of less than 7 considered low. Here, preterm birth is defined as delivery occurring before 37 weeks of gestation, and stillbirth describes a fetus born dead after 22 weeks of gestation.
Women in the MS cohort were more likely to have elective C-sections (odds ratio, 2.89 [95% confidence interval, 1.65-2.16]), induced labor (OR, 1.15 [95%CI, 1.01-1.31]) and have babies with low birth weight based on their gestational age (OR, 1.29 [95% CI, 1.04-1.60]). Nearly 30% of babies born in the cohort (n = 851) were born to mothers who had received disease-modifying therapy (DMT). Neonates exposed to DMT weighed an average of 116 g less than babies born to mothers who had not received DMT (3,378 g vs. 3,494 g) with a slightly lower gestational age (39 weeks as opposed to 40 weeks). However, babies born to mothers with MS were less likely to show signs of asphyxia (OR, 0.87 [95% CI, 0.78-0.97]) than the comparison cohort.
“We found overall, their pregnancies were just as healthy as those of the moms without MS,” Dr. Magyari said.
Comprehensive data
Denmark’s health care system has two key features that make it an attractive setting in which to conduct such a study – the first being its universal health care. The second advantage is that the country enacted several health registries in the 1970s and 1980s that enable the collection of more comprehensive data. For example, the Danish National Patient Register is a population-based registry that spans the entire nation, facilitating epidemiological research with what the study’s authors describe as “high generalizability.” Providing additional insights regarding the patient story helps add context to pregnancy and outcomes. Among the data collected on the women studied were demographics, contact information, and abortions, both spontaneous and medically induced. The country uses other databases and registries to capture additional data. For example, the Register of Legally Induced Abortions provides data regarding the context of medically induced abortions. In contrast, the Danish Medical Birth Registry provides context regarding specified variables regarding women’s pregnancies, delivery, and perinatal outcomes. Finally, the population’s education register offers information regarding patients’ educational history.
A key strength of this study is that the long duration of follow-up data from the Danish Medical Birth Registry, along with its comprehensive data collection, eliminates recall bias. Universal access to health care also improves the generalizability of data. A limitation of the study is its lack of data on maternal smoking and its effects on low gestational weights. The study also has some data gaps, including body mass index information missing from a large portion of the cohort. Finally, the sample size of newborns born to mothers who had received DMT therapy within the last 6 months of gestation was too underpowered to stratify based on first on first-line or second-line treatment.
Dr. Magyari served on scientific advisory boards for Biogen, Sanofi, Teva, Roche, Novartis, and Merck. She has also received honoraria for lecturing from Biogen, Merck, Novartis, Sanofi, Genzyme, and has received research support and support for congress participation from Biogen, Genzyme, Teva, Roche, Merck, and Novartis. Coauthors disclosed various fees received from Merck, Novartis, Biogen, Roche, Sanofi Genzyme, and Teva.
, according to a new study published online Feb. 3 in Neurology Clinical Practice. While pregnancy and childbirth are not regarded as conditions that engender high-risk pregnancy in the MS population, previous studies evaluating the effects of MS on pregnancy and parturition have yet to fully elucidate some outcomes for pregnant women and their babies in multiple sclerosis.
“Women with multiple sclerosis may be understandably concerned about the risk of pregnancy,” said Melinda Magyari, MD, PhD, a consultant at the University of Copenhagen. “While previous research has shown there is no higher risk of birth defect for babies born to women with MS, we wanted to find out if women with MS are at risk for a variety of pregnancy complications.”
MS is regarded as a progressive, neurological disease mediated by the immune system that demands careful consideration of numerous situations and life changes including family planning. The MS population is overwhelmingly female, as women account for three out of every four cases of MS. The majority of these women range from 20 to 40 years of age at the time of being diagnosed with MS. Despite the unknown risks of pregnancy-related complications and various perinatal complications in this patient population, women who have MS are not discouraged from conceiving.
Assessing pregnancy outcomes
This nationwide, population-based, cross-sectional study evaluated the pregnancies of 2,930 women with MS between Jan. 1, 1997, and Dec. 31, 2016, registered in the Danish Multiple Sclerosis Registry. The researchers compared pregnancy-related and prenatal outcomes to a 5% random sample of 56,958 randomly-selected pregnant women from Denmark’s general population who did not have MS. They found no differences in the risks associated with several pregnancy-related complications (e.g., preeclampsia, gestational diabetes, or placental complications), emergency Cesarean section (C-section), instrumental delivery, stillbirth, preterm birth, or congenital malformation. Apgar scores were low in both groups. A composite of various biometrics in newborns such as reflexes, muscle tone, and heart rate immediately following birth, the Apgar score is used to help assess the neonatal health, with a value of less than 7 considered low. Here, preterm birth is defined as delivery occurring before 37 weeks of gestation, and stillbirth describes a fetus born dead after 22 weeks of gestation.
Women in the MS cohort were more likely to have elective C-sections (odds ratio, 2.89 [95% confidence interval, 1.65-2.16]), induced labor (OR, 1.15 [95%CI, 1.01-1.31]) and have babies with low birth weight based on their gestational age (OR, 1.29 [95% CI, 1.04-1.60]). Nearly 30% of babies born in the cohort (n = 851) were born to mothers who had received disease-modifying therapy (DMT). Neonates exposed to DMT weighed an average of 116 g less than babies born to mothers who had not received DMT (3,378 g vs. 3,494 g) with a slightly lower gestational age (39 weeks as opposed to 40 weeks). However, babies born to mothers with MS were less likely to show signs of asphyxia (OR, 0.87 [95% CI, 0.78-0.97]) than the comparison cohort.
“We found overall, their pregnancies were just as healthy as those of the moms without MS,” Dr. Magyari said.
Comprehensive data
Denmark’s health care system has two key features that make it an attractive setting in which to conduct such a study – the first being its universal health care. The second advantage is that the country enacted several health registries in the 1970s and 1980s that enable the collection of more comprehensive data. For example, the Danish National Patient Register is a population-based registry that spans the entire nation, facilitating epidemiological research with what the study’s authors describe as “high generalizability.” Providing additional insights regarding the patient story helps add context to pregnancy and outcomes. Among the data collected on the women studied were demographics, contact information, and abortions, both spontaneous and medically induced. The country uses other databases and registries to capture additional data. For example, the Register of Legally Induced Abortions provides data regarding the context of medically induced abortions. In contrast, the Danish Medical Birth Registry provides context regarding specified variables regarding women’s pregnancies, delivery, and perinatal outcomes. Finally, the population’s education register offers information regarding patients’ educational history.
A key strength of this study is that the long duration of follow-up data from the Danish Medical Birth Registry, along with its comprehensive data collection, eliminates recall bias. Universal access to health care also improves the generalizability of data. A limitation of the study is its lack of data on maternal smoking and its effects on low gestational weights. The study also has some data gaps, including body mass index information missing from a large portion of the cohort. Finally, the sample size of newborns born to mothers who had received DMT therapy within the last 6 months of gestation was too underpowered to stratify based on first on first-line or second-line treatment.
Dr. Magyari served on scientific advisory boards for Biogen, Sanofi, Teva, Roche, Novartis, and Merck. She has also received honoraria for lecturing from Biogen, Merck, Novartis, Sanofi, Genzyme, and has received research support and support for congress participation from Biogen, Genzyme, Teva, Roche, Merck, and Novartis. Coauthors disclosed various fees received from Merck, Novartis, Biogen, Roche, Sanofi Genzyme, and Teva.
, according to a new study published online Feb. 3 in Neurology Clinical Practice. While pregnancy and childbirth are not regarded as conditions that engender high-risk pregnancy in the MS population, previous studies evaluating the effects of MS on pregnancy and parturition have yet to fully elucidate some outcomes for pregnant women and their babies in multiple sclerosis.
“Women with multiple sclerosis may be understandably concerned about the risk of pregnancy,” said Melinda Magyari, MD, PhD, a consultant at the University of Copenhagen. “While previous research has shown there is no higher risk of birth defect for babies born to women with MS, we wanted to find out if women with MS are at risk for a variety of pregnancy complications.”
MS is regarded as a progressive, neurological disease mediated by the immune system that demands careful consideration of numerous situations and life changes including family planning. The MS population is overwhelmingly female, as women account for three out of every four cases of MS. The majority of these women range from 20 to 40 years of age at the time of being diagnosed with MS. Despite the unknown risks of pregnancy-related complications and various perinatal complications in this patient population, women who have MS are not discouraged from conceiving.
Assessing pregnancy outcomes
This nationwide, population-based, cross-sectional study evaluated the pregnancies of 2,930 women with MS between Jan. 1, 1997, and Dec. 31, 2016, registered in the Danish Multiple Sclerosis Registry. The researchers compared pregnancy-related and prenatal outcomes to a 5% random sample of 56,958 randomly-selected pregnant women from Denmark’s general population who did not have MS. They found no differences in the risks associated with several pregnancy-related complications (e.g., preeclampsia, gestational diabetes, or placental complications), emergency Cesarean section (C-section), instrumental delivery, stillbirth, preterm birth, or congenital malformation. Apgar scores were low in both groups. A composite of various biometrics in newborns such as reflexes, muscle tone, and heart rate immediately following birth, the Apgar score is used to help assess the neonatal health, with a value of less than 7 considered low. Here, preterm birth is defined as delivery occurring before 37 weeks of gestation, and stillbirth describes a fetus born dead after 22 weeks of gestation.
Women in the MS cohort were more likely to have elective C-sections (odds ratio, 2.89 [95% confidence interval, 1.65-2.16]), induced labor (OR, 1.15 [95%CI, 1.01-1.31]) and have babies with low birth weight based on their gestational age (OR, 1.29 [95% CI, 1.04-1.60]). Nearly 30% of babies born in the cohort (n = 851) were born to mothers who had received disease-modifying therapy (DMT). Neonates exposed to DMT weighed an average of 116 g less than babies born to mothers who had not received DMT (3,378 g vs. 3,494 g) with a slightly lower gestational age (39 weeks as opposed to 40 weeks). However, babies born to mothers with MS were less likely to show signs of asphyxia (OR, 0.87 [95% CI, 0.78-0.97]) than the comparison cohort.
“We found overall, their pregnancies were just as healthy as those of the moms without MS,” Dr. Magyari said.
Comprehensive data
Denmark’s health care system has two key features that make it an attractive setting in which to conduct such a study – the first being its universal health care. The second advantage is that the country enacted several health registries in the 1970s and 1980s that enable the collection of more comprehensive data. For example, the Danish National Patient Register is a population-based registry that spans the entire nation, facilitating epidemiological research with what the study’s authors describe as “high generalizability.” Providing additional insights regarding the patient story helps add context to pregnancy and outcomes. Among the data collected on the women studied were demographics, contact information, and abortions, both spontaneous and medically induced. The country uses other databases and registries to capture additional data. For example, the Register of Legally Induced Abortions provides data regarding the context of medically induced abortions. In contrast, the Danish Medical Birth Registry provides context regarding specified variables regarding women’s pregnancies, delivery, and perinatal outcomes. Finally, the population’s education register offers information regarding patients’ educational history.
A key strength of this study is that the long duration of follow-up data from the Danish Medical Birth Registry, along with its comprehensive data collection, eliminates recall bias. Universal access to health care also improves the generalizability of data. A limitation of the study is its lack of data on maternal smoking and its effects on low gestational weights. The study also has some data gaps, including body mass index information missing from a large portion of the cohort. Finally, the sample size of newborns born to mothers who had received DMT therapy within the last 6 months of gestation was too underpowered to stratify based on first on first-line or second-line treatment.
Dr. Magyari served on scientific advisory boards for Biogen, Sanofi, Teva, Roche, Novartis, and Merck. She has also received honoraria for lecturing from Biogen, Merck, Novartis, Sanofi, Genzyme, and has received research support and support for congress participation from Biogen, Genzyme, Teva, Roche, Merck, and Novartis. Coauthors disclosed various fees received from Merck, Novartis, Biogen, Roche, Sanofi Genzyme, and Teva.
FROM NEUROLOGY CLINICAL PRACTICE
Clinical Edge Commentary: Breast Cancer February 2021
Systemic therapy advances for metastatic breast cancer (MBC) have led to improved overall survival; however, the CNS sanctuary site can be challenging to treat and cause significant neurologic symptoms. Kuksis and colleagues reported that HER2-positive and triple-negative breast MBC subtypes have high incidence of brain metastases, with pooled cumulative incidences of 31% and 32%, respectively. The incidence per patient-year was 13% for both subtypes. The treatment landscape for HER2-positive MBC is rapidly evolving. The combination of tucatinib/capecitabine/trastuzumab demonstrated reduced risk of intracranial progression or death (HR 0.32), improvement in intracranial objective response rate (47.3% versus 20.0%) and reduced risk of death (0.58) in the HER2CLIMB study. An MRI screening program may enhance early detection and study prevention of brain metastases.
Trastuzumab and pertuzumab have significantly improved outcomes in early-stage and advanced HER2-positive breast cancer. These monoclonal antibodies are given with chemotherapy initially, followed by dual antibody therapy to complete one year for curative treatment and until disease progression in the metastatic setting. The FeDeriCa randomized phase 3 study demonstrated that fixed-dose subcutaneous combination of pertuzumab and trastuzumab was non-inferior to intravenous formulations with a geometric mean ratio of cycle 7 pertuzumab serum trough concentration of 1.22 (90% CI 1.14-1.31). Furthermore, efficacy appeared similar with pathologic complete response rates of 60% for both groups. Trastuzumab and pertuzumab administration in an infusion center requires hospital resources, cost, and patient’s time. The MetasphHER study showed that patients preferred subcutaneous trastuzumab compared to IV, and further investigation into long-term outcomes and impact on quality of life and resource allocation is desired.
The phase 3 CLEOPATRA study demonstrated improvement in overall survival with the addition of pertuzumab to docetaxel and trastuzumab in patients with HER2-positive MBC. Rash and diarrhea are well-recognized potential side effects related to pertuzumab. Ferreira et al demonstrated that rash was prognostic for PFS and OS both during and after docetaxel discontinuation, while diarrhea was only prognostic after docetaxel. Neither rash nor diarrhea were predictive of pertuzumab benefit. Studies have shown that patients who developed immune-related adverse events during anti-PD-1 treatment had PFS and OS benefit compared to those who did not. These results may help inform and encourage patients and providers regarding benefit of various treatments in the setting of toxicities. Future research exploring biomarker prognostic and predictive capability is certainly warranted.
Endocrine therapy plus a CDK 4/6 inhibitor is standard first-line treatment for hormone receptor-positive MBC. Progression that occurs rapidly or through multiple lines of endocrine therapy combinations, may indicate endocrine resistance. The phase 3 PEARL trial did not demonstrate superior PFS among patients with wild type ESR1 AI-resistant MBC with palbociclib/endocrine therapy compared to capecitabine (mPFS 8.0 vs 10.6 months, adjusted HR 1.11; p=0.404); similar PFS and aHR were seen in palbociclib/fulvestrant versus capecitabine cohort. Therapy sequencing in HR+/HER2-negative MBC is indeed relevant, and a previous meta-analysis demonstrated chemotherapy was not better than endocrine therapy plus CDK 4/6 inhibitor in first- or second-line setting. Importantly, palbociclib plus endocrine therapy was associated with better tolerability and quality of life, which should be considered when selecting treatment with similar PFS outcomes.
References:
Lin NU, Borges V, Anders C, Murthy RK, Paplomata E, Hamilton E, Hurvitz S, Loi S, Okines A, Abramson V, Bedard PL, Oliveira M, Mueller V, Zelnak A, DiGiovanna MP, Bachelot T, Chien AJ, O'Regan R, Wardley A, Conlin A, Cameron D, Carey L, Curigliano G, Gelmon K, Loibl S, Mayor J, McGoldrick S, An X, Winer EP. Intracranial efficacy and survival with tucatinib plus trastuzumab and capecitabine for previously treated HER2-positive breast cancer with brain metastases in the HER2CLIMB trial. J Clin Oncol. 2020;38:2610-2619.
Pivot X, Spano JP, Espie M, Cottu P, Jouannaud C, Pottier V, Moreau L, Extra JM, Lortholary A, Rivera P, Spaeth D, Attar-Rabia H, Benkanoun C, Dima-Martinez L, Esposito N, Gligorov J. Patients' preference of trastuzumab administration (subcutaneous versus intravenous) in HER2-positive metastatic breast cancer: Results of the randomised MetaspHer study. Eur J Cancer. 2017;82:230-236.
Das S, Ciombor KK, Haraldsdottir S, Pumpalova Y, Sahin IH, Pineda G, Shyr Y, Lin EP, Hsu CY, Chu SK, Goff LW, Cardin DB, Bilen MA, Fisher GA, Wu C, Berlin J. Immune-related adverse events and immune checkpoint inhibitor efficacy in patients with gastrointestinal cancer with Food and Drug Administration-approved indications for immunotherapy. Oncologist. 2020;25:669-679.
Giuliano M, Schettini F, Rognoni C, Milani M, Jerusalem G, Bachelot T, De Laurentiis M, Thomas G, De Placido P, Arpino G, De Placido S, Cristofanilli M, Giordano A, Puglisi F, Pistilli B, Prat A, Del Mastro L, Venturini S, Generali D. Endocrine treatment versus chemotherapy in postmenopausal women with hormone receptor-positive, HER2-negative, metastatic breast cancer: a systematic review and network meta-analysis. Lancet Oncol. 2019;20:1360-1369.
Systemic therapy advances for metastatic breast cancer (MBC) have led to improved overall survival; however, the CNS sanctuary site can be challenging to treat and cause significant neurologic symptoms. Kuksis and colleagues reported that HER2-positive and triple-negative breast MBC subtypes have high incidence of brain metastases, with pooled cumulative incidences of 31% and 32%, respectively. The incidence per patient-year was 13% for both subtypes. The treatment landscape for HER2-positive MBC is rapidly evolving. The combination of tucatinib/capecitabine/trastuzumab demonstrated reduced risk of intracranial progression or death (HR 0.32), improvement in intracranial objective response rate (47.3% versus 20.0%) and reduced risk of death (0.58) in the HER2CLIMB study. An MRI screening program may enhance early detection and study prevention of brain metastases.
Trastuzumab and pertuzumab have significantly improved outcomes in early-stage and advanced HER2-positive breast cancer. These monoclonal antibodies are given with chemotherapy initially, followed by dual antibody therapy to complete one year for curative treatment and until disease progression in the metastatic setting. The FeDeriCa randomized phase 3 study demonstrated that fixed-dose subcutaneous combination of pertuzumab and trastuzumab was non-inferior to intravenous formulations with a geometric mean ratio of cycle 7 pertuzumab serum trough concentration of 1.22 (90% CI 1.14-1.31). Furthermore, efficacy appeared similar with pathologic complete response rates of 60% for both groups. Trastuzumab and pertuzumab administration in an infusion center requires hospital resources, cost, and patient’s time. The MetasphHER study showed that patients preferred subcutaneous trastuzumab compared to IV, and further investigation into long-term outcomes and impact on quality of life and resource allocation is desired.
The phase 3 CLEOPATRA study demonstrated improvement in overall survival with the addition of pertuzumab to docetaxel and trastuzumab in patients with HER2-positive MBC. Rash and diarrhea are well-recognized potential side effects related to pertuzumab. Ferreira et al demonstrated that rash was prognostic for PFS and OS both during and after docetaxel discontinuation, while diarrhea was only prognostic after docetaxel. Neither rash nor diarrhea were predictive of pertuzumab benefit. Studies have shown that patients who developed immune-related adverse events during anti-PD-1 treatment had PFS and OS benefit compared to those who did not. These results may help inform and encourage patients and providers regarding benefit of various treatments in the setting of toxicities. Future research exploring biomarker prognostic and predictive capability is certainly warranted.
Endocrine therapy plus a CDK 4/6 inhibitor is standard first-line treatment for hormone receptor-positive MBC. Progression that occurs rapidly or through multiple lines of endocrine therapy combinations, may indicate endocrine resistance. The phase 3 PEARL trial did not demonstrate superior PFS among patients with wild type ESR1 AI-resistant MBC with palbociclib/endocrine therapy compared to capecitabine (mPFS 8.0 vs 10.6 months, adjusted HR 1.11; p=0.404); similar PFS and aHR were seen in palbociclib/fulvestrant versus capecitabine cohort. Therapy sequencing in HR+/HER2-negative MBC is indeed relevant, and a previous meta-analysis demonstrated chemotherapy was not better than endocrine therapy plus CDK 4/6 inhibitor in first- or second-line setting. Importantly, palbociclib plus endocrine therapy was associated with better tolerability and quality of life, which should be considered when selecting treatment with similar PFS outcomes.
References:
Lin NU, Borges V, Anders C, Murthy RK, Paplomata E, Hamilton E, Hurvitz S, Loi S, Okines A, Abramson V, Bedard PL, Oliveira M, Mueller V, Zelnak A, DiGiovanna MP, Bachelot T, Chien AJ, O'Regan R, Wardley A, Conlin A, Cameron D, Carey L, Curigliano G, Gelmon K, Loibl S, Mayor J, McGoldrick S, An X, Winer EP. Intracranial efficacy and survival with tucatinib plus trastuzumab and capecitabine for previously treated HER2-positive breast cancer with brain metastases in the HER2CLIMB trial. J Clin Oncol. 2020;38:2610-2619.
Pivot X, Spano JP, Espie M, Cottu P, Jouannaud C, Pottier V, Moreau L, Extra JM, Lortholary A, Rivera P, Spaeth D, Attar-Rabia H, Benkanoun C, Dima-Martinez L, Esposito N, Gligorov J. Patients' preference of trastuzumab administration (subcutaneous versus intravenous) in HER2-positive metastatic breast cancer: Results of the randomised MetaspHer study. Eur J Cancer. 2017;82:230-236.
Das S, Ciombor KK, Haraldsdottir S, Pumpalova Y, Sahin IH, Pineda G, Shyr Y, Lin EP, Hsu CY, Chu SK, Goff LW, Cardin DB, Bilen MA, Fisher GA, Wu C, Berlin J. Immune-related adverse events and immune checkpoint inhibitor efficacy in patients with gastrointestinal cancer with Food and Drug Administration-approved indications for immunotherapy. Oncologist. 2020;25:669-679.
Giuliano M, Schettini F, Rognoni C, Milani M, Jerusalem G, Bachelot T, De Laurentiis M, Thomas G, De Placido P, Arpino G, De Placido S, Cristofanilli M, Giordano A, Puglisi F, Pistilli B, Prat A, Del Mastro L, Venturini S, Generali D. Endocrine treatment versus chemotherapy in postmenopausal women with hormone receptor-positive, HER2-negative, metastatic breast cancer: a systematic review and network meta-analysis. Lancet Oncol. 2019;20:1360-1369.
Systemic therapy advances for metastatic breast cancer (MBC) have led to improved overall survival; however, the CNS sanctuary site can be challenging to treat and cause significant neurologic symptoms. Kuksis and colleagues reported that HER2-positive and triple-negative breast MBC subtypes have high incidence of brain metastases, with pooled cumulative incidences of 31% and 32%, respectively. The incidence per patient-year was 13% for both subtypes. The treatment landscape for HER2-positive MBC is rapidly evolving. The combination of tucatinib/capecitabine/trastuzumab demonstrated reduced risk of intracranial progression or death (HR 0.32), improvement in intracranial objective response rate (47.3% versus 20.0%) and reduced risk of death (0.58) in the HER2CLIMB study. An MRI screening program may enhance early detection and study prevention of brain metastases.
Trastuzumab and pertuzumab have significantly improved outcomes in early-stage and advanced HER2-positive breast cancer. These monoclonal antibodies are given with chemotherapy initially, followed by dual antibody therapy to complete one year for curative treatment and until disease progression in the metastatic setting. The FeDeriCa randomized phase 3 study demonstrated that fixed-dose subcutaneous combination of pertuzumab and trastuzumab was non-inferior to intravenous formulations with a geometric mean ratio of cycle 7 pertuzumab serum trough concentration of 1.22 (90% CI 1.14-1.31). Furthermore, efficacy appeared similar with pathologic complete response rates of 60% for both groups. Trastuzumab and pertuzumab administration in an infusion center requires hospital resources, cost, and patient’s time. The MetasphHER study showed that patients preferred subcutaneous trastuzumab compared to IV, and further investigation into long-term outcomes and impact on quality of life and resource allocation is desired.
The phase 3 CLEOPATRA study demonstrated improvement in overall survival with the addition of pertuzumab to docetaxel and trastuzumab in patients with HER2-positive MBC. Rash and diarrhea are well-recognized potential side effects related to pertuzumab. Ferreira et al demonstrated that rash was prognostic for PFS and OS both during and after docetaxel discontinuation, while diarrhea was only prognostic after docetaxel. Neither rash nor diarrhea were predictive of pertuzumab benefit. Studies have shown that patients who developed immune-related adverse events during anti-PD-1 treatment had PFS and OS benefit compared to those who did not. These results may help inform and encourage patients and providers regarding benefit of various treatments in the setting of toxicities. Future research exploring biomarker prognostic and predictive capability is certainly warranted.
Endocrine therapy plus a CDK 4/6 inhibitor is standard first-line treatment for hormone receptor-positive MBC. Progression that occurs rapidly or through multiple lines of endocrine therapy combinations, may indicate endocrine resistance. The phase 3 PEARL trial did not demonstrate superior PFS among patients with wild type ESR1 AI-resistant MBC with palbociclib/endocrine therapy compared to capecitabine (mPFS 8.0 vs 10.6 months, adjusted HR 1.11; p=0.404); similar PFS and aHR were seen in palbociclib/fulvestrant versus capecitabine cohort. Therapy sequencing in HR+/HER2-negative MBC is indeed relevant, and a previous meta-analysis demonstrated chemotherapy was not better than endocrine therapy plus CDK 4/6 inhibitor in first- or second-line setting. Importantly, palbociclib plus endocrine therapy was associated with better tolerability and quality of life, which should be considered when selecting treatment with similar PFS outcomes.
References:
Lin NU, Borges V, Anders C, Murthy RK, Paplomata E, Hamilton E, Hurvitz S, Loi S, Okines A, Abramson V, Bedard PL, Oliveira M, Mueller V, Zelnak A, DiGiovanna MP, Bachelot T, Chien AJ, O'Regan R, Wardley A, Conlin A, Cameron D, Carey L, Curigliano G, Gelmon K, Loibl S, Mayor J, McGoldrick S, An X, Winer EP. Intracranial efficacy and survival with tucatinib plus trastuzumab and capecitabine for previously treated HER2-positive breast cancer with brain metastases in the HER2CLIMB trial. J Clin Oncol. 2020;38:2610-2619.
Pivot X, Spano JP, Espie M, Cottu P, Jouannaud C, Pottier V, Moreau L, Extra JM, Lortholary A, Rivera P, Spaeth D, Attar-Rabia H, Benkanoun C, Dima-Martinez L, Esposito N, Gligorov J. Patients' preference of trastuzumab administration (subcutaneous versus intravenous) in HER2-positive metastatic breast cancer: Results of the randomised MetaspHer study. Eur J Cancer. 2017;82:230-236.
Das S, Ciombor KK, Haraldsdottir S, Pumpalova Y, Sahin IH, Pineda G, Shyr Y, Lin EP, Hsu CY, Chu SK, Goff LW, Cardin DB, Bilen MA, Fisher GA, Wu C, Berlin J. Immune-related adverse events and immune checkpoint inhibitor efficacy in patients with gastrointestinal cancer with Food and Drug Administration-approved indications for immunotherapy. Oncologist. 2020;25:669-679.
Giuliano M, Schettini F, Rognoni C, Milani M, Jerusalem G, Bachelot T, De Laurentiis M, Thomas G, De Placido P, Arpino G, De Placido S, Cristofanilli M, Giordano A, Puglisi F, Pistilli B, Prat A, Del Mastro L, Venturini S, Generali D. Endocrine treatment versus chemotherapy in postmenopausal women with hormone receptor-positive, HER2-negative, metastatic breast cancer: a systematic review and network meta-analysis. Lancet Oncol. 2019;20:1360-1369.
How does long-term OC use affect breast, ovarian, and endometrial cancer risk?
Karlsson T, Johansson T, Hoguland J, et al. Time-dependent effects of oral contraceptive use on breast, ovarian and endometrial cancers. Cancer Research. 2020;canres.2476.2020. doi:10.1158/0008-5472.CAN-20-2476.
EXPERT COMMENTARY
The long-term effects of OC use on gynecologic and breast cancers has been uncertain, with different reports yielding conflicting findings. To assess the time-dependent and long-term associations between OC use and the risk of breast, ovarian, and endometrial cancer in women born between 1939 and 1970, Karlsson and colleagues used data from the UK Biobank (which includes a large cross-sectional cohort of individuals recruited between 2006 and 2010) and national databases.
Details of the study
A total of 256,661 women were included in this study. Of these, 82% (210,443) had used or were currently using OC (ever-users) and 18% (46,218) had never used OC (never-users). There were 17,739; 1,966; and 2,462 cases of breast, ovarian, and endometrial cancer, respectively, identified.
In analyses adjusted for 10 parameters, the ORs for ovarian (OR, 0.72) and endometrial cancer (OR, 0.68) were lower among ever-users of OC compared with never-users (P<.05). However, the OR for breast cancer (OR, 1.02) was similar among ever-users and never-users of OC (P>.05).
Among women followed to age 55, results were similar for the 2 gynecologic cancers but were significantly higher for breast cancer (OR, 1.10; P<.05). With 20 or more years of OC use, greater prevention of ovarian (OR, 0.60) and, particularly, endometrial cancer (OR, 0.36) was observed (P<.05). However, the risk of breast cancer was similar in never-users and long-term users of OC.
Study strengths and limitations
A strength of this study is that, compared with most previous studies, it had a much longer follow-up period.
The authors noted, however, that among the potential limitations in the study design was the fact that only 6% of participants invited to the UK Biobank volunteered to participate in the study. This may have resulted in participation bias within the cohort, reflecting a healthier cohort that is not representative of the overall population. ●
These study findings from a large cross-sectional cohort by Karlsson and colleagues suggest that controversy regarding the association of breast cancer with OC use may reflect different study methodologies with respect to timing. The authors note that while the lifetime risk of breast cancer may not differ between OC ever-users and never-users, there appears to be a transient elevated risk associated with OC use. By contrast, OC use, particularly when used long-term, appears to “dramatically” reduce the risk of ovarian and endometrial cancer, according to the study authors.
ANDREW M. KAUNITZ, MD
Karlsson T, Johansson T, Hoguland J, et al. Time-dependent effects of oral contraceptive use on breast, ovarian and endometrial cancers. Cancer Research. 2020;canres.2476.2020. doi:10.1158/0008-5472.CAN-20-2476.
EXPERT COMMENTARY
The long-term effects of OC use on gynecologic and breast cancers has been uncertain, with different reports yielding conflicting findings. To assess the time-dependent and long-term associations between OC use and the risk of breast, ovarian, and endometrial cancer in women born between 1939 and 1970, Karlsson and colleagues used data from the UK Biobank (which includes a large cross-sectional cohort of individuals recruited between 2006 and 2010) and national databases.
Details of the study
A total of 256,661 women were included in this study. Of these, 82% (210,443) had used or were currently using OC (ever-users) and 18% (46,218) had never used OC (never-users). There were 17,739; 1,966; and 2,462 cases of breast, ovarian, and endometrial cancer, respectively, identified.
In analyses adjusted for 10 parameters, the ORs for ovarian (OR, 0.72) and endometrial cancer (OR, 0.68) were lower among ever-users of OC compared with never-users (P<.05). However, the OR for breast cancer (OR, 1.02) was similar among ever-users and never-users of OC (P>.05).
Among women followed to age 55, results were similar for the 2 gynecologic cancers but were significantly higher for breast cancer (OR, 1.10; P<.05). With 20 or more years of OC use, greater prevention of ovarian (OR, 0.60) and, particularly, endometrial cancer (OR, 0.36) was observed (P<.05). However, the risk of breast cancer was similar in never-users and long-term users of OC.
Study strengths and limitations
A strength of this study is that, compared with most previous studies, it had a much longer follow-up period.
The authors noted, however, that among the potential limitations in the study design was the fact that only 6% of participants invited to the UK Biobank volunteered to participate in the study. This may have resulted in participation bias within the cohort, reflecting a healthier cohort that is not representative of the overall population. ●
These study findings from a large cross-sectional cohort by Karlsson and colleagues suggest that controversy regarding the association of breast cancer with OC use may reflect different study methodologies with respect to timing. The authors note that while the lifetime risk of breast cancer may not differ between OC ever-users and never-users, there appears to be a transient elevated risk associated with OC use. By contrast, OC use, particularly when used long-term, appears to “dramatically” reduce the risk of ovarian and endometrial cancer, according to the study authors.
ANDREW M. KAUNITZ, MD
Karlsson T, Johansson T, Hoguland J, et al. Time-dependent effects of oral contraceptive use on breast, ovarian and endometrial cancers. Cancer Research. 2020;canres.2476.2020. doi:10.1158/0008-5472.CAN-20-2476.
EXPERT COMMENTARY
The long-term effects of OC use on gynecologic and breast cancers has been uncertain, with different reports yielding conflicting findings. To assess the time-dependent and long-term associations between OC use and the risk of breast, ovarian, and endometrial cancer in women born between 1939 and 1970, Karlsson and colleagues used data from the UK Biobank (which includes a large cross-sectional cohort of individuals recruited between 2006 and 2010) and national databases.
Details of the study
A total of 256,661 women were included in this study. Of these, 82% (210,443) had used or were currently using OC (ever-users) and 18% (46,218) had never used OC (never-users). There were 17,739; 1,966; and 2,462 cases of breast, ovarian, and endometrial cancer, respectively, identified.
In analyses adjusted for 10 parameters, the ORs for ovarian (OR, 0.72) and endometrial cancer (OR, 0.68) were lower among ever-users of OC compared with never-users (P<.05). However, the OR for breast cancer (OR, 1.02) was similar among ever-users and never-users of OC (P>.05).
Among women followed to age 55, results were similar for the 2 gynecologic cancers but were significantly higher for breast cancer (OR, 1.10; P<.05). With 20 or more years of OC use, greater prevention of ovarian (OR, 0.60) and, particularly, endometrial cancer (OR, 0.36) was observed (P<.05). However, the risk of breast cancer was similar in never-users and long-term users of OC.
Study strengths and limitations
A strength of this study is that, compared with most previous studies, it had a much longer follow-up period.
The authors noted, however, that among the potential limitations in the study design was the fact that only 6% of participants invited to the UK Biobank volunteered to participate in the study. This may have resulted in participation bias within the cohort, reflecting a healthier cohort that is not representative of the overall population. ●
These study findings from a large cross-sectional cohort by Karlsson and colleagues suggest that controversy regarding the association of breast cancer with OC use may reflect different study methodologies with respect to timing. The authors note that while the lifetime risk of breast cancer may not differ between OC ever-users and never-users, there appears to be a transient elevated risk associated with OC use. By contrast, OC use, particularly when used long-term, appears to “dramatically” reduce the risk of ovarian and endometrial cancer, according to the study authors.
ANDREW M. KAUNITZ, MD
Product update: Breast biopsy system, tamponade mini-sponge, ovulation prediction device and app
Updated option for breast biopsy
Hologic announces updates to its Brevera® Breast Biopsy System with CorLumina® Imaging Technology. The Brevera system is designed for use with the manufacturer’s Affirm® Prone biopsy guidance system.
For more information, visit https://www.hologic.com.
“Mini-sponge” device shows potential to treat PPH
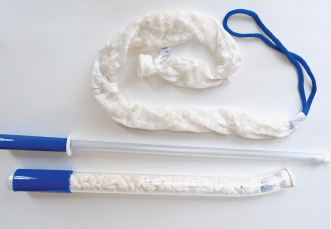
During a pilot study, reports Obstetrx, 9 patients, treated at the University Teaching Hospital in Lusaka, Zambia, did not respond to conventional PPH management options after vaginal birth but did respond, with bleeding resolved in 60 seconds and no adverse events, to the XSTAT device. The device was left in place for a mean time of 1 hour, and none of the patients required further surgical procedures or blood transfusions. The initial placement time of XSTAT (mean time to placement, 62 seconds) was faster than times reported for balloon uterine tamponade devices. The pilot study results were published in Obstetrics & Gynecology.
XSTAT is US Food and Drug Administration–approved to treat high-flow arterial bleeding in prehospital trauma settings, and Obstetrx is planning to submit for 510k clearance in 2022, after the conclusion of a follow-up PPH trial in 2021.
For more information, visit: https://www.obstetrx.com/.
Continue to: AI and ovulation prediction...
AI and ovulation prediction
A woman’s fertility window is typically the 5 days leading up to ovulation, with peak fertility in the 2 to 3 days before ovulation. There are other options for measuring that fertile window, including luteinizing hormone (LH) tests; however, Prima-Temp reports that Priya predicts the fertile window an average of 2.6 days before tests for LH. Utilizing continuous core body temperature measurement, Priya detects subtle changes in temperature patterns that occur prior to ovulation. The app portion of the technology stores and analyzes the temperature measurements, for a high-tech fertility alert system that also offers clinical diagnostic support. Potential users of the Priya system are able to sign up to receive it through the product’s website.
For more information, visit: https://www.priyafertility.com.
Updated option for breast biopsy
Hologic announces updates to its Brevera® Breast Biopsy System with CorLumina® Imaging Technology. The Brevera system is designed for use with the manufacturer’s Affirm® Prone biopsy guidance system.
For more information, visit https://www.hologic.com.
“Mini-sponge” device shows potential to treat PPH
During a pilot study, reports Obstetrx, 9 patients, treated at the University Teaching Hospital in Lusaka, Zambia, did not respond to conventional PPH management options after vaginal birth but did respond, with bleeding resolved in 60 seconds and no adverse events, to the XSTAT device. The device was left in place for a mean time of 1 hour, and none of the patients required further surgical procedures or blood transfusions. The initial placement time of XSTAT (mean time to placement, 62 seconds) was faster than times reported for balloon uterine tamponade devices. The pilot study results were published in Obstetrics & Gynecology.
XSTAT is US Food and Drug Administration–approved to treat high-flow arterial bleeding in prehospital trauma settings, and Obstetrx is planning to submit for 510k clearance in 2022, after the conclusion of a follow-up PPH trial in 2021.
For more information, visit: https://www.obstetrx.com/.
Continue to: AI and ovulation prediction...
AI and ovulation prediction
A woman’s fertility window is typically the 5 days leading up to ovulation, with peak fertility in the 2 to 3 days before ovulation. There are other options for measuring that fertile window, including luteinizing hormone (LH) tests; however, Prima-Temp reports that Priya predicts the fertile window an average of 2.6 days before tests for LH. Utilizing continuous core body temperature measurement, Priya detects subtle changes in temperature patterns that occur prior to ovulation. The app portion of the technology stores and analyzes the temperature measurements, for a high-tech fertility alert system that also offers clinical diagnostic support. Potential users of the Priya system are able to sign up to receive it through the product’s website.
For more information, visit: https://www.priyafertility.com.
Updated option for breast biopsy
Hologic announces updates to its Brevera® Breast Biopsy System with CorLumina® Imaging Technology. The Brevera system is designed for use with the manufacturer’s Affirm® Prone biopsy guidance system.
For more information, visit https://www.hologic.com.
“Mini-sponge” device shows potential to treat PPH
During a pilot study, reports Obstetrx, 9 patients, treated at the University Teaching Hospital in Lusaka, Zambia, did not respond to conventional PPH management options after vaginal birth but did respond, with bleeding resolved in 60 seconds and no adverse events, to the XSTAT device. The device was left in place for a mean time of 1 hour, and none of the patients required further surgical procedures or blood transfusions. The initial placement time of XSTAT (mean time to placement, 62 seconds) was faster than times reported for balloon uterine tamponade devices. The pilot study results were published in Obstetrics & Gynecology.
XSTAT is US Food and Drug Administration–approved to treat high-flow arterial bleeding in prehospital trauma settings, and Obstetrx is planning to submit for 510k clearance in 2022, after the conclusion of a follow-up PPH trial in 2021.
For more information, visit: https://www.obstetrx.com/.
Continue to: AI and ovulation prediction...
AI and ovulation prediction
A woman’s fertility window is typically the 5 days leading up to ovulation, with peak fertility in the 2 to 3 days before ovulation. There are other options for measuring that fertile window, including luteinizing hormone (LH) tests; however, Prima-Temp reports that Priya predicts the fertile window an average of 2.6 days before tests for LH. Utilizing continuous core body temperature measurement, Priya detects subtle changes in temperature patterns that occur prior to ovulation. The app portion of the technology stores and analyzes the temperature measurements, for a high-tech fertility alert system that also offers clinical diagnostic support. Potential users of the Priya system are able to sign up to receive it through the product’s website.
For more information, visit: https://www.priyafertility.com.
FDA approves intramuscular administration for peginterferon beta-1a in MS
“The new IM administration offers people living with relapsing MS the well-characterized efficacy and safety of Plegridy with the potential for significantly reduced injection site reactions,” Biogen said in a news release announcing the FDA action.
Plegridy is a pegylated version of interferon beta-1a, which prolongs the circulation time of the molecule in the body by increasing its size. The process extends the drug’s half-life, allowing for a less-frequent dosing schedule.
Peginterferon beta-1a administered subcutaneously was first approved by the FDA in 2014 based on data showing it significantly reduces MS relapses, disability progression, and brain lesions.
The FDA approved IM administration for peginterferon beta-1a based on data evaluating bioequivalence and adverse reactions associated with IM administration compared with subcutaneous (SC) administration in healthy volunteers.
Bioequivalence of the IM and SC dosing regimens was confirmed and volunteers receiving the drug through IM administration experienced fewer injection site reactions relative to those receiving SC administration (14.4% vs. 32.1%), the company said.
The overall safety profiles of IM and SC administration were generally similar, with no new safety signals.
The European Commission allowed marketing authorization for IM administration of peginterferon beta-1a in December 2020.
A version of this article first appeared on Medscape.com.
“The new IM administration offers people living with relapsing MS the well-characterized efficacy and safety of Plegridy with the potential for significantly reduced injection site reactions,” Biogen said in a news release announcing the FDA action.
Plegridy is a pegylated version of interferon beta-1a, which prolongs the circulation time of the molecule in the body by increasing its size. The process extends the drug’s half-life, allowing for a less-frequent dosing schedule.
Peginterferon beta-1a administered subcutaneously was first approved by the FDA in 2014 based on data showing it significantly reduces MS relapses, disability progression, and brain lesions.
The FDA approved IM administration for peginterferon beta-1a based on data evaluating bioequivalence and adverse reactions associated with IM administration compared with subcutaneous (SC) administration in healthy volunteers.
Bioequivalence of the IM and SC dosing regimens was confirmed and volunteers receiving the drug through IM administration experienced fewer injection site reactions relative to those receiving SC administration (14.4% vs. 32.1%), the company said.
The overall safety profiles of IM and SC administration were generally similar, with no new safety signals.
The European Commission allowed marketing authorization for IM administration of peginterferon beta-1a in December 2020.
A version of this article first appeared on Medscape.com.
“The new IM administration offers people living with relapsing MS the well-characterized efficacy and safety of Plegridy with the potential for significantly reduced injection site reactions,” Biogen said in a news release announcing the FDA action.
Plegridy is a pegylated version of interferon beta-1a, which prolongs the circulation time of the molecule in the body by increasing its size. The process extends the drug’s half-life, allowing for a less-frequent dosing schedule.
Peginterferon beta-1a administered subcutaneously was first approved by the FDA in 2014 based on data showing it significantly reduces MS relapses, disability progression, and brain lesions.
The FDA approved IM administration for peginterferon beta-1a based on data evaluating bioequivalence and adverse reactions associated with IM administration compared with subcutaneous (SC) administration in healthy volunteers.
Bioequivalence of the IM and SC dosing regimens was confirmed and volunteers receiving the drug through IM administration experienced fewer injection site reactions relative to those receiving SC administration (14.4% vs. 32.1%), the company said.
The overall safety profiles of IM and SC administration were generally similar, with no new safety signals.
The European Commission allowed marketing authorization for IM administration of peginterferon beta-1a in December 2020.
A version of this article first appeared on Medscape.com.